Chapter 6 Alterations in Cardiovascular and Hemolymphatic Systems
PERIPHERAL EDEMA, PLEURAL EFFUSION, ASCITES
Edema is an abnormal accumulation of extracellular fluid in the interstitial spaces of the tissues or in body cavities that can be generalized or localized. If the fluid accumulation occurs in the pleural cavity, it is referred to as pleural effusion or hydrothorax; if the fluid accumulation is in the abdominal cavity, it is referred to as peritoneal effusion or ascites.
Fluid accumulates more easily in those parts of the body where the connective tissue structure is relatively loose. The accumulated fluid tends to gravitate to the dependent areas of the body. In the cow, generalized edema is detected externally by swelling of the submandibular tissue, the brisket, the ventral abdomen, and occasionally the limbs (Fig. 6-1). External manifestation of generalized edema in the horse is frequently in the pectoral region between the front limbs, along the ventral abdomen, in the prepuce in stallions and geldings (Fig. 6-2), in the limbs, and sometimes in the head. Stocking up, or limb edema restricted to the lower limbs, is commonly detected in stabled horses with no underlying disease. Large amounts of fluid may accumulate before clinical signs become evident. External evidence of pulmonary edema (i.e., a frothy, possibly blood-tinged fluid in the nares or expectorated) is rarely detected in large animals (Fig. 6-3). There are numerous causes of edema, including congestive heart failure (CHF) (Boxes 6-1 and 6-2). Edema is a late sign of CHF; other subtle signs of failure may be present before edema appears.
Box 6-1 Causes of Peripheral Edema, Pleural Effusion, and Ascites in Horses
Box 6-2 Causes of Peripheral Edema, Pleural Effusion, and Ascites in Ruminants
Mechanisms of Edema
Edema is caused by an alteration in the equilibrium between capillary permeability and the forces that govern fluid movements at the capillary level. These forces are as follows:
Activation of complement and liberation of cytotoxic agents such as oxygen radicals, leukotrienes, hydrogen peroxide, platelet-activating factor, and lysosomal enzymes contribute to the endothelial and epithelial damage, causing permeability edema. Subsequent increase in colloid osmotic pressure causes fluid accumulation in the interstitial space. The most common causes of increased capillary permeability are trauma, infection, endotoxemia, and hypersensitivity (allergic) vasculitis. Topical administration of counterirritants can also cause local increase in capillary permeability. Equine purpura hemorrhagica, the most common vasculitic disease in horses, may in its mildest form have symptoms of mucosal petechiae and plaques of edema or, in severe cases, serum exudation from and necrosis of skin surfaces.
Increased hydrostatic pressure can cause either localized or generalized edema. In horses and ruminants the most common causes of increased hydrostatic pressure are CHF, venous thrombosis, liver disease causing obstruction of the portal venous system, lymphadenopathy, a cranial mediastinal mass, compression bandages, limb immobilization, and topical administration of counterirritants. CHF occurs when there is concomitant pulmonary and systemic vascular congestion. The compensatory salt and water retention increases ventricular diastolic, venous, and capillary pressures, which can result in the formation of generalized edema. Arteriolar vasodilation, caused by release of tissue mediators of inflammation or increased venous pressure resulting from obstruction to venous outflow, can also elevate capillary hydrostatic pressure and result in edema formation.
When the plasma protein concentration decreases from normal to values less than 5 g/dL or albumin concentration is less than 1.5 g/dL, generalized edema may occur. Hypoproteinemia can result from (1) decreased production of plasma proteins with starvation, liver disease, or severe heart failure, or (2) augmented loss of plasma proteins resulting from kidney disease, protein-losing enteropathies (Johne’s disease, chronic inflammatory bowel disease), peritonitis, or pleuritis. Hemodilution as a result of overzealous administration of fluids or decreased elimination of fluid can cause edema. Failure to excrete adequate water to maintain fluid balance can result from decreased glomerular filtration as a result of kidney disease or heart failure.
Increased tissue colloid osmotic pressure is rarely a cause of edema in horses and ruminants. Interstitial fluid has a lower plasma protein concentration than plasma. When capillary permeability is increased or when abnormal protein-like material is present in the interstitial space, edema can develop by this mechanism. The latter may occur with infection or after administration of topical counterirritants.
Lymphedema occurs when lymphatics are absent or obstructed. Congenital absence of lymphatics is extremely rare. Obstruction to lymphatic drainage can be caused by tumor, local inflammation (lymphangitis or lymphadenitis), or elevated central venous pressure as in heart failure.
Approach to Diagnosis of Peripheral Edema, Pleural Effusion, and Ascites
CARDIAC ARRHYTHMIAS
Cardiac arrhythmias are abnormalities in the normal heart rate, rhythm, or conduction pattern. Arrhythmias result from abnormalities of impulse generation or impulse conduction or a combination of both. In the normal heart the impulse is generated in the sinus node because it has the highest rate of spontaneous depolarization. Atrial contraction is followed shortly by ventricular contraction. There is variability in reported normal ranges for heart rate in the large adult animal species, but there is general acceptance of the following ranges:
Arrhythmias are more common in horses than in other domestic animal species. As many as 25% of horses that have no other signs of heart disease have cardiac arrhythmias during routine examination or electrocardiography.1 During continuous 24-hour electrocardiography, 44% of normal horses had second-degree atrioventricular (AV) block, 10% had sinus arrhythmia, 3% had sinoatrial (SA) block, 27% had occasional supraventricular extrasystoles, and 15% had occasional ventricular arrhythmias.2 Cardiac arrhythmias may be present in 40% of horses that have other signs of cardiac disease.1 Unlike other species, the horse has arrhythmias at rest that are considered benign or functional. Benign, physiologic, or functional arrhythmias are usually bradyarrhythmias and are thought to be the result of increased vagal tone. These arrhythmias disappear at high heart rates (exercise or excitement) or with the administration of atropine (0.02 to 0.05 mg/kg subcutaneously [SC] or intramuscularly [IM]) or glycopyrrolate (0.003 to 0.006 mg/kg SC or IM). Some examples of benign or functional arrhythmias are as follows:
Other arrhythmias are usually considered to be pathologic, even if there are no other overt signs of cardiac disease. Some examples of pathologic arrhythmias are:
The most effective method of identifying the specific arrhythmia is by performing an ECG. Arrhythmias that are transient or intermittent may not be detected with resting electrocardiography. Radiotelemetry or continuous 24-hour ECG recordings are useful to characterize the type, frequency, and severity of arrhythmias. Exercising electrocardiography may identify arrhythmias that are absent or clinically insignificant at rest but that may impair performance.
In general, cattle do not have benign arrhythmias like horses, but they are frequently found to have sinus bradycardia and sinus arrhythmia associated with lack of feed intake. These arrhythmias were previously thought to be abnormal and associated with vagal indigestion but have been shown to occur in normal cattle held off feed for 12 to 48 hours.3 Cattle with gastrointestinal disease seem to have increased susceptibility to cardiac arrhythmias, especially atrial premature depolarizations and fibrillation. Although the reason for the susceptibility is not established, abnormal electrolyte concentrations, acid-base disturbances, and aberrations in autonomic nervous system balance have been proposed.4,5 Sinus arrhythmia in goats is considered to be a benign arrhythmia and is present in many normal animals. Normal camelids also frequently have sinus arrhythmia.
Mechanisms of Cardiac Arrhythmias
Arrhythmias result from abnormalities of impulse generation or impulse conduction or a combination of both. A variety of mechanisms can cause abnormal impulse generation or conduction (Boxes 6-3 and 6-4). Abnormal impulse generation occurs because of localized changes in ionic currents that flow across the membranes of single cells or groups of cells. Abnormal impulse generation can be seen as automaticity (normal and abnormal) or triggered activity.
Automaticity, the ability to initiate action potentials spontaneously, is a property of cells in the sinus node, some parts of the atria, the AV junction, and the His-Purkinje system. Cardiac disease can be responsible for the development of automaticity in cells that normally do not have this property. Normal automaticity develops when the membrane potential slowly falls (i.e., becomes less negative) during diastole. When the membrane reaches its threshold potential, an impulse is initiated. The most common clinical arrhythmias that are thought to be caused by the automaticity mechanism are sinus tachycardia and sinus bradycardia, which are the result of alterations in autonomic nervous system tone. Enhanced automaticity in another area of the heart that is capable of automaticity (spontaneous depolarization) may be responsible for atrial or ventricular premature beats. It is not clear what clinical arrhythmias are caused by triggered activity.
Under certain circumstances, conduction abnormalities allow a propagating impulse, which has already excited the heart, to persist and reexcite the atria or ventricles after the end of the refractory period. This can occur in an ordered or random fashion. Random reentry occurs over reentrant pathways that continuously change in size and location with time, whereas ordered reentry occurs over a relatively fixed reentrant pathway. Impulse propagation may be slow enough that reentrant circuits can be established in very small areas of myocardium. In large animals the size of the myocardial circuit is large enough that relatively mild alterations in impulse propagation may make reentry feasible and may account for the relatively greater frequency of arrhythmias in these species. Although it is not possible to precisely define the mechanism of clinical arrhythmias, it is believed that atrial and ventricular fibrillation may be caused by random reentry.
Under clinical conditions, cardiac arrhythmias may be associated with disturbances in electrolyte concentrations, especially potassium and calcium, in acid-base balance, and in autonomic nervous system balance. These conditions can precipitate cellular changes conducive to the development of arrhythmias by any of the above mechanisms.
Approach to Diagnosis of Cardiac Arrhythmias
It is important to distinguish between abnormal arrhythmias that are primary and those that are secondary. Most abnormal arrhythmias of horses and cattle are tachyarrhythmias. Primary arrhythmias are caused by pathologic conditions of the heart (myocarditis, valvular disease, conduction system abnormalities, and pericarditis). Secondary arrhythmias develop in the absence of heart disease and can be caused by excitement, fever, sepsis, hypoxemia, metabolic or electrolyte imbalances, gastrointestinal disturbances, anesthesia, ionophores, other drugs, or toxemia. The treatment and prognosis for the two types of arrhythmias can be very different, and examination and laboratory tests are used to assist in making the distinction.
CARDIAC MURMURS
Throughout the cardiovascular system, blood has a laminar or streamlined flow, except in the heart and sometimes in the aorta. Occasionally conditions occur that cause turbulent flow that is sufficient to cause resonance in adjacent structures. This resonance may be heard as a murmur when a critical level of turbulence is reached (Boxes 6-5 and 6-6). The factors that determine whether blood flow is laminar or turbulent are related by the Reynolds number, which is the ratio of the inertial to viscous forces. When the Reynolds number exceeds a critical value (about 2000 in large vessels), turbulence occurs. Increased flow velocity or reduced blood viscosity (e.g., anemia) predisposes to murmur development. The characteristics of the murmur depend on the velocity of the blood flow and the nature of the structures that are caused to vibrate.
It is useful to characterize murmurs with regard to timing in the cardiac cycle (systolic, diastolic, or continuous), duration in the cardiac cycle (early, mid, late, holo-, pan-), intensity (loudness), shape and quality or frequency, PMI, and radiation of the murmur. Systolic murmurs occur anytime between the first and second heart sound. Diastolic murmurs occur between the second and first heart sounds. Continuous murmurs occur throughout the cardiac cycle (Fig. 6-4). The intensity of murmurs is frequently graded on a scale of 1 to 67:
 Grade 6 is a loud murmur that is audible with the entire stethoscope chest piece held away from the chest wall.
Grade 6 is a loud murmur that is audible with the entire stethoscope chest piece held away from the chest wall.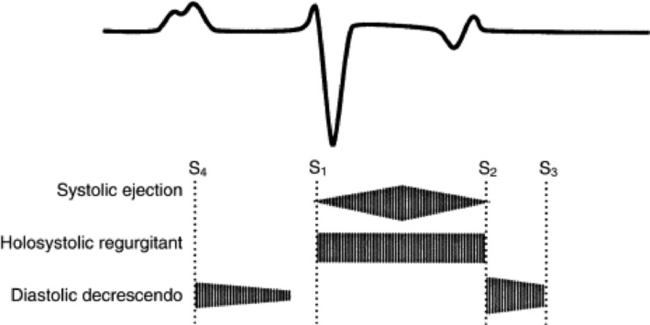
Fig. 6-4 Phonocardiographic characteristics of systolic ejection, holosystolic (pansystolic) regurgitant, and diastolic decrescendo cardiac murmurs.
The PMI of a murmur usually corresponds to the location of one of the heart valves. Murmurs associated with the mitral valve will frequently be heard best in the left fifth intercostal space just dorsal to the level of the elbow. These murmurs usually radiate dorsally or toward the aortic valve area. Pulmonic valve and aortic valve murmurs are best heard at the base of the heart. For this area to be accessed, the hand is moved under the left triceps muscle to the third and fourth intercostal spaces just below the level of the shoulder. Aortic valve murmurs are located just dorsal and caudal to the pulmonic valve. Murmurs associated with the tricuspid valve are frequently located in the right third or fourth intercostal space between the shoulder and elbow.
Most systolic murmurs fall into one of two categories: ejection or regurgitant (see Fig. 6-4). Systolic ejection murmurs are caused by obstructed, increased, or turbulent blood flow across normal or damaged semilunar valves. Valvular obstruction is rare in large animals, but functional ejection murmurs are commonly found in healthy horses. The PMI of the functional murmur is typically at the pulmonic or aortic valve or just dorsal to them over the great vessels. It is a crescendo-decrescendo murmur that is audible in early-to-mid systole. The diagnostic considerations for systolic ejection murmurs are given in Box 6-7. The innocent or functional murmur may be distinguished from a pathologic murmur by being of lower and variable intensity, peaking in early-to-mid systole, ending well before the second heart sound, and having no radiation. The physiologic systolic ejection murmur may disappear or become louder after exercise. Diagnostic considerations for systolic regurgitant murmurs are listed in Box 6-7.
Regurgitant murmurs typically begin with AV valve closure and end after pulmonic and aortic valve closure, making the second heart sound inaudible. They can be variable in duration, however, and occur in early, mid, or late systole or can be pansystolic or holosystolic. Location of the PMI and the direction of radiation of the systolic murmur distinguish mitral or tricuspid regurgitation from a ventricular septal defect (VSD). Systolic clicks are rare in horses and cattle but may indicate abnormalities of the chordae tendineae, AV valve prolapse, or dilation of the aorta.
Diastolic murmurs can occur between S4 and S1 (atrial systolic murmurs), between S2 and S3 (ventricular filling murmurs), or from S2 to S1 (aortic regurgitation or rarely, pulmonic regurgitation). Both the atrial systolic and ventricular filling murmurs are usually functional, can be heard over the left or right hemithorax, and can vary in intensity. The aortic regurgitation murmur is typically a decrescendo murmur with its PMI over the aortic valve that begins immediately after S2 (see Fig. 6-4). Some aortic regurgitation murmurs can be harsh or musical, associated with high-frequency vibrations of an aortic valve leaflet, and they may be audible over the right thorax.
Continuous murmurs are uncommon in horses and ruminants. Patent ductus arteriosus, a finding in normal foals for a short time after delivery, can be heard in the left third intercostal space. This murmur can be continuous, but more frequently only a residual systolic murmur is audible.8 Continuous machinery murmurs are most frequently reported in adult horses with an aortic cardiac fistula secondary to rupture of the aortic root or of a sinus of Valsalva aneurysm. A continuous “washing machine” murmur, which is most easily heard over the left cardiac area, is associated with traumatic pericarditis in cattle and is caused by the accumulation of fluid, gas, and fibrin within the pericardium. Acquired systolic and diastolic murmurs in adult horses or cattle are usually the result of separate murmurs.
Approach to Diagnosis of Cardiac Murmurs
MUFFLED HEART SOUNDS
Auscultation of heart sounds requires that the vibrations generated by the heart be transmitted through the tissues of the thorax to the outer chest wall with sufficient amplitude to be heard. Blood transmits sound very well, whereas lung tissue strongly attenuates sound waves. The chest wall itself causes attenuation of the sound that is most significant at the interface between bone and muscle. Therefore physical factors in a normal patient, such as a large, thick chest or obesity, can cause heart sounds to be muffled. If the environment for auscultation is conducive to hearing heart sounds, other factors such as stethoscope quality may cause muffling of heart sounds in a normal patient. One should strive to have a stethoscope with comfortably fitting earpieces, thicker and shorter tubing, a rigid diaphragm to hear S1, S2, and higher-frequency sounds, and a bell piece for auscultation of S3, S4, low-frequency sounds, and murmurs.
Heart sounds are muffled primarily because of displacement of the heart from the thoracic wall by fluid (pericardial effusion), a soft-tissue mass (abscess or tumor), or air (pneumothorax, pneumomediastinum, or emphysema) (Boxes 6-8 and 6-9). Rarely is muffling of heart sounds attributed to weak cardiac contractions alone, although this may be a finding in recumbent cows with marked hypocalcemia.
Approach to Diagnosis of Muffled Heart Sounds
CARDIOVASCULAR EXERCISE INTOLERANCE, WEAKNESS, AND SYNCOPE
Exercise intolerance, weakness, or syncope can be a clinical sign associated with disease in many body systems. Exercise intolerance can be manifested as sudden deceleration or stopping, failure to perform at an expected level, a sudden change in the level of performance or production, lowered enthusiasm for work, cough on exertion, evidence of respiratory distress, or excessive sweating. Weakness can be manifested as recumbency, difficulty in rising from recumbency, muscle tremors or fasciculations, reluctance to move, or toe dragging. Syncope is a sudden collapse and loss of consciousness (fainting).
Mechanisms of Cardiovascular Exercise Intolerance, Weakness, and Syncope
The clinical signs of exercise intolerance, syncope, or weakness can be caused by cardiovascular disease (Boxes 6-10 and 6-11). They are the result of failure to maintain cardiac output, caused by inability to regulate either heart rate or stroke volume. A normal horse increases cardiac output at submaximum heart rates (less than 210 beats/min in horses) primarily by tachycardia. At maximum heart rates (approximately 210 to 240 beats/min in horses), subsequent increments in cardiac output occur by increased stroke volume.9 The maximum heart rate for cattle and small ruminants has not been published.
Box 6-10 Causes of Exercise Intolerance, Weakness, and Syncope in Horses*
* See Chapters 8 and 13 for additional noncardiac causes.
Box 6-11 Causes of Exercise Intolerance, Weakness, and Syncope in Ruminants*
* See Chapters 8 and 13 for additional noncardiac causes.
Supraventricular cardiac arrhythmias, primarily atrial fibrillation in horses, can lead to heart rates greater than 240 beats/min with submaximum exercise.10,11 Heart rates exceeding the maximum rate may limit cardiac output by decreasing the time for diastolic perfusion of the myocardium or by limiting stroke volume because the short diastolic intervals leave inadequate time for ventricular filling. The ability to maintain cardiac output can also be compromised by other cardiac arrhythmias such as ventricular premature systoles. The frequency of extrasystoles can increase with exercise, and the timing of the abnormal beats can reduce cardiac output even at submaximum heart rates.12 Horses with cardiac arrhythmias can have abnormal elevations in lactate concentration in response to exercise, indicating a lower anaerobic threshold and leading to exercise intolerance.13,14
Cardiac output maintenance may also be compromised in animals by diseases affecting myocardial contractility or diseases that result in increased end-systolic volume despite a submaximum heart rate.15 Diseases that result in decreased venous return (peripheral vascular disease) can also reduce cardiac output and cause signs of exercise intolerance, weakness, or syncope.
Exercise intolerance or weakness can also be caused by painful peripheral vascular conditions or conditions causing peripheral hypoxia or lactic acid accumulation. In horses such conditions may exist with aortoiliac thrombosis. Sudden episodes of weakness and collapse without change in consciousness are associated with hyperkalemic period paralysis in horses (see Chapter 42).
Syncope may be associated with epilepsy or other central nervous system (CNS) disturbance. If cardiovascular and pulmonary function appears normal, the nervous system should be examined in detail (see Chapter 8). Collapse during exercise is most commonly caused by cardiovascular disease, whereas collapse at rest is usually associated with neurologic disease.
Approach to Diagnosis of Exercise Intolerance, Weakness, and Syncope
VENOUS DISTENTION AND PULSATIONS
The jugular venous pulsations observed in the neck are primarily a reflection of right atrial and right ventricular activity. There may be some small contribution from carotid arterial impact.16 The jugular venous pulse reflects the right atrial or central venous pressure, which is influenced by blood volume, right ventricular cardiac output, and right atrial contractility (Boxes 6-12 and 6-13). Jugular venous pulsations are observed in normal animals, but the pulse seldom radiates more than one third of the distance from the thoracic inlet to the ramus of the mandible when the head is held in a normal, upright position.
Mechanisms of Venous Distention and Pulsations
The normal jugular venous pulse consists of three positive and two negative deflections (Fig. 6-5). The first and dominant positive wave is the A wave, produced by atrial contraction. During atrial relaxation the pressure declines until ventricular systole. The second positive deflection is the C wave, which is produced by the bulging of the tricuspid valve leaflets into the right atrium during early (isovolumetric) right ventricular systole. Carotid arterial impact on the jugular vein may also contribute to the C wave.16 As the ventricle contracts, the plane of the tricuspid valve is pulled toward the apex of the heart and the atrial pressure declines, producing the X descent. The X descent is terminated by the V wave, which is associated with venous return, subsequent atrial filling, and a closed tricuspid valve. At the end of ventricular systole, the atrial pressure falls again as a result of tricuspid valve opening and rapid right ventricular filling. This is the Y descent. The Y descent is terminated as the pressure gradually rises with right-sided heart filling.
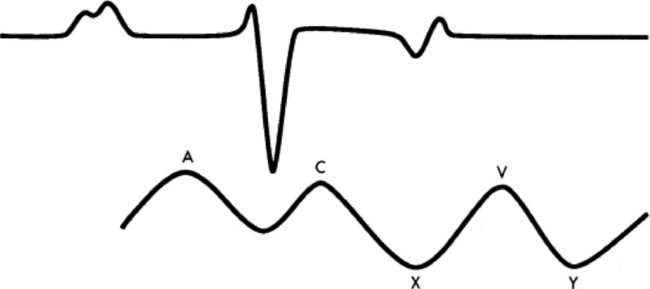
Fig. 6-5 Schematic illustration of a venous (jugular or atrial) pressure curve and its relationship to events of the electrocardiogram. A, Positive wave produced by atrial contraction; C, second positive deflection caused by bulging of the tricuspid valve during isovolumetric systole; X, first negative wave produced by the plane of the AV valve being pulled toward the apex of the heart during systole; V, positive pressure wave caused by venous return; Y, negative wave produced by AV valve opening.
Abnormal pulsations occur with increased resistance to right ventricular filling, regardless of the cause. Distention and pulsations in the jugular vein are usually associated with an elevated right ventricular pressure, such as occurs in right-sided heart failure, constrictive pericarditis, or, more rarely, cardiomyopathy. Prominent jugular pulsations are noted with tricuspid regurgitation and certain cardiac arrhythmias, especially those arrhythmias associated with atrial contraction against a closed AV valve. The carotid arterial pulse can mimic jugular venous pulsations. To distinguish among the causes of jugular venous pulsations, lightly compress but do not occlude the jugular vein at the thoracic inlet. The jugular vein will distend enough to eliminate carotid arterial pulsations. If pulsations are still present, tricuspid regurgitation, atrial arrhythmias, or right-sided heart failure should be considered. If the jugular vein is compressed near the ramus of the mandible and massaged toward the thoracic inlet, refilling is indicative of tricuspid regurgitation. Jugular venous distention without pulsations can occur with compression of the cranial vena cava from a cranial thoracic or mediastinal mass or from occlusion of the jugular vein with a thrombus.
Approach to Diagnosis of VenousDistention and Pulsations
PAINFUL PERIPHERAL SWELLINGS
Close inspection of the skin and extremities of patients can reveal evidence of peripheral vascular or lymphatic system disease. These diseases can be manifested by diffuse swelling, localized swelling (papules, nodules, macules, or wheals), or subcutaneous edema of the extremities. Frequently there is necrosis, ulceration of the skin, and exudation as the disease progresses. Animals may be lame or dyspneic, have heat in the involved area, or exhibit a painful response to palpation of the area (Boxes 6-14 and 6-15), which helps to differentiate these conditions from nonpainful peripheral edema.
Approach to Diagnosis of PainfulPeripheral Swellings
ENLARGED LYMPH NODES
Diffuse or single lymph node enlargement occurs with infectious (bacterial, viral, fungal) conditions, neoplasia, and, rarely, immune-mediated causes in large animals (Boxes 6-16 and 6-17). Lymphadenopathy may cause obstruction to lymphatic drainage, leading to peripheral edema, pleural effusion, or ascites. The peripheral lymph nodes that are most readily accessible for examination are the submandibular (horses), superficial cervical (ruminants), and superficial inguinal (ruminants) lymph nodes. When there is generalized lymphadenopathy, internal lymph nodes may be enlarged, causing clinical signs such as dyspnea, esophageal obstruction, diarrhea, or other signs of organ dysfunction.
Approach to Diagnosis of Enlarged Lymph Nodes
ABNORMAL PERIPHERAL PULSE
Palpation of the arterial pulse is an important aspect of the examination of the patient with cardiovascular disease. Arterial pulse strength and contour (how fast pressure rises and falls) are the objectives of the examination and are determined by the cardiac output, heart rate, and vascular impedance. The arterial pressure pulse begins with the opening of the aortic valve and ventricular ejection and rises rapidly in early systole. The pulse pressure reaches a peak then declines as ventricular ejection slows. During isovolumic relaxation (before AV valve opening), there is a transient reversal of flow in the arterial system, and an incisura or dicrotic notch (Fig. 6-6) is inscribed on the descending limb of the pressure curve. Following the incisura, there is a small positive wave that is attributed to elastic recoil of the aorta and the aortic valve and the summation of reflected waves from more distal arteries.15 After the positive wave, the pulse pressure declines because there is peripheral runoff of blood in diastole. The incisura and secondary positive wave are not usually palpable. Palpation of peripheral arteries (facial, transverse facial, and digital arteries in the horse and median and coccygeal arteries in ruminants) normally reveals a smooth, rapid upstroke, a dome-shaped summit, and a downstroke that is slightly more prolonged than the upstroke.
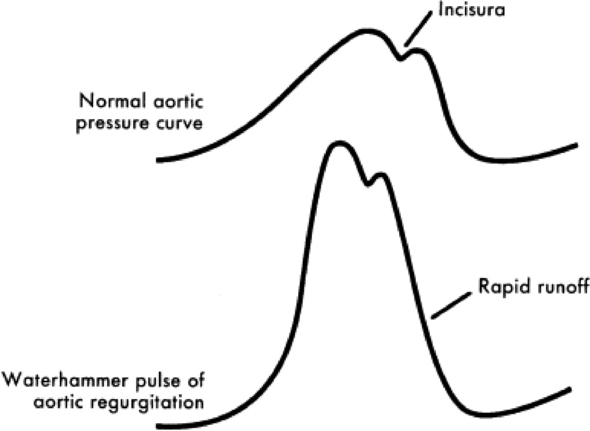
Fig. 6-6 Schematic illustration of the normal arterial pressure pulse. The incisura that occurs during the descending limb is caused by a transient reversal in flow during isovolumic relaxation. Compared with the normal arterial pressure pulse, the waterhammer pulse of aortic regurgitation builds rapidly and has a rapid runoff.
Pressure values and pulse wave configurations are altered as the pressure waves are transmitted through the peripheral arterial tree. With increasing distance from the heart, the dicrotic notch and second positive wave disappear, the systolic pressure gets higher (loss of distensibility in the distal arteries and summation of reflected pulse waves from the distal vascular bed), and the diastolic pressure gets lower. The difference between the systolic and diastolic pressure determines pulse pressure and can be evaluated by an impression of pulse strength. Pulse pressure increases as one moves to more peripheral arterial sites. The mean arterial pressure changes very little but decreases slightly as one moves downstream in the arterial system from the pressure source. Systolic blood pressure, as measured indirectly at the tail or on a limb, is higher than that measured in the ascending aorta. In smaller arterial beds (e.g., arteries of the ear), the pulse wave is gradually dampened and pulsatile characteristics are lost on the capillaries and small veins.
Mechanisms of Abnormal Peripheral Pulse
Hyperkinetic arterial pulses occur in patients with increased cardiac output (e.g., fever, exercise, excitement), increased stroke volume, or bradycardia (Boxes 6-18 and 6-19). It may also occur when there is rapid runoff of blood in the arterial system, as occurs with aortic valve regurgitation, patent ductus arteriosus, or aortic cardiac fistulas. In aortic valve regurgitation the rapidly rising, hyperdynamic pulse is caused by increased stroke volume (regurgitated blood in the left ventricle), followed by a rapid runoff of pressure later in systole as a result of regurgitation (see Fig. 6-6).
Hypokinetic pulses are present in patients with diminished stroke volume caused by hypovolemia, left ventricular failure, or, rarely, in large animals, mitral or aortic valve stenosis.
Abnormal peripheral pulses are detected in patients with cardiac arrhythmias. With premature ventricular contractions (PVCs), the compensatory pause that occurs after the PVC allows a longer period of time for ventricular filling, which results in a greater end-diastolic volume, increased contractile force, and a stronger pulse in the beat that follows the PVC. The strength of the peripheral pulse is variable in arrhythmias such as atrial fibrillation because the irregular rhythm is associated with variable time for ventricular filling. Certain arrhythmias, particularly tachyarrhythmias, allow inadequate ventricular filling to generate a peripheral arterial pulse, and a pulse deficit is palpated.
Approach to Diagnosis of Abnormal Peripheral Pulse
1 Detweiler DK, Patterson DF. The cardiovascular system. In: Catcott EJ, Smithcors JF, editors. Equine medicine and surgery. ed 2. Santa Barbara, Calif: American Veterinary Publications; 1982:645.
2 Reef VB. Frequency of cardiac arrhythmias and their significance in normal horses. Proc Am Coll Vet Intern Med. 1989;7:506.
3 McGuirk SM, Bednarski RM. Bradycardia associated with fasting in cattle. Proc Fourth Ann Vet Med Forum. 1986;2:10.
4 Goetze L, Vörös K, Scholz H, Lattmann J. Respiratory mechanics and ECG findings in experimentally induced metabolic alkalosis in adult cattle. Dtsch Tierarztl Wschr. 1984;91:307.
5 McGuirk SM, Muir WW, Sams RA, Rings DM. Atrial fibrillation in cows: clinical findings and therapeutic considerations. J Am Vet Med Assoc. 1983;182:1380.
6 Brien PJ, Landt Y, Landenson JH. Differential reactivity of cardiac and skeletal muscle from various species in a cardiac troponin I immunoassay. Clin Chem. 1997;43:2333.
7 Freeman AR, Levine SA. The clinical significance of the systolic murmur: a study of 1000 consecutive “noncardiac” cases. Ann Intern Med. 1933;6:1371.
8 Reef VB. Cardiovascular disease in the equine neonate. Vet Clin North Am (Equine Pract). 1985;1:117.
9 Evans EL. Cardiovascular adaptations to exercise and training. Vet Clin North Am Equine Pract. 1985;1:513.
10 Amada A, et al. Atrial fibrillation in the horse: clinical and histopathological studies of two cases. Exp Rep Equine Health Lab. 1974;11:51.
11 Deegen E, Buntenkotter S. Behaviour of the heart rate of horses with auricular fibrillation during exercise and after treatment. Equine Vet J. 1976;8:26.
12 Holmes JR, Alps BJ. The effect of exercise on rhythm irregularities in the horse. Vet Rec. 1966;78:672.
13 Maier-Bock H, Ehrlein H-J. Heart rate during a defined exercise test in horses with heart and lung diseases. Equine Vet J. 1978;10:235.
14 Persson SGB. Analysis of fitness and state of training. In: Snow DH, Persson SGB, Rose RJ, editors. Equine exercise physiology. Cambridge, England: Granta Editions; 1983:p 441.
15 Miller RJ, Holmes JR. Effect of cardiac arrhythmia on left ventricular and aortic blood pressure parameters in the horse. Res Vet Sci. 1983;35:190.
16 O’Rourke RA. Physical examination of the arteries and veins (including blood pressure determination). Hurst JW, editor. The heart, ed 6, vol 1. New York: McGraw-Hill, 1986;138.
17 Parry BW. Practical assessment of the circulatory status of equine colic cases. Compend Cont Educ (Pract Vet). 1986;8:S236.
18 Fregin GF. The cardiovascular system. In: Mansmann RA, McAllister ES, editors. Equine medicine and surgery. Santa Barbara, Calif: American Veterinary Publications; 1982:645.