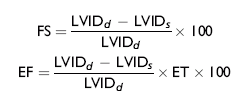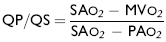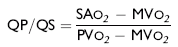Chapter 30 Diseases of the Cardiovascular System
PERFORMING THE ELECTROCARDIOGRAM
No single electrocardiographic lead system has been universally accepted for use in large animals. Bipolar leads (I, II, III, base-apex, X, Y, and Z of the orthogonal lead system) and unipolar leads (aVF, aVR, aVL, thoracic) have been described, but the amplitude, duration, and configuration of the different waveforms vary widely, depending on an animal’s breed, size, body type, and sex. In addition, there is lability of certain waveforms within each animal depending on the level of exercise, excitement, or organic heart disease. Large animals have a deeply penetrating Purkinje system, and depolarization from ventricular endocardium to epicardium occurs explosively and in many directions at once. This period of ventricular activation is responsible for the electrocardiographic criteria indicative of ventricular enlargement in small animals but contributes little to the generation of the QRS complex of large animals. Thus establishing specific diagnostic criteria for chamber enlargement in large animal species has been difficult because changes in the QRS complex are not sensitive or specific for ventricular enlargement.
Therefore the electrocardiogram (ECG) is used primarily to detect cardiac arrhythmias. For this purpose a single-channel machine can be used, and the lead system chosen can be any that generates distinctive P, QRS, and T complexes. If an arrhythmia is detected, another lead can then be obtained to further characterize the QRS and T complexes and confirm their origin. The lead system should be easy to apply, and the tracing free of artifacts created by muscle tremors, skin movement, shifting of weight, and changes in limb position. Two such leads commonly used for the diagnosis of cardiac arrhythmias are the base-apex lead1 and the Y lead of the orthogonal lead system.2 The base-apex lead is attached by placing the positive electrode from one of three standard bipolar leads (lead I, II, or III) on the left thorax in the fifth intercostal space at the level of the elbow or at the location where the apex beat is most readily palpable. The negative electrode is attached to the skin of the right jugular furrow two thirds of the way from the ramus of the mandible to the thoracic inlet or at the top of the right scapular spine. The ground electrode can be attached to any site remote from the heart. Electrical contact is improved by clipping hair or wetting the skin with alcohol. The base-apex lead ECG is recorded by switching the machine to the bipolar lead that has been attached to the horse and recording the ECG (Table 30-1). Lead Y is attached by placing the positive electrode over the xiphoid and the negative electrode cranially to the front of the chest.
Table 30-1 Standard Bipolar Electrocardiographic Leads
| Lead | Positive Electrode | Negative Electrode |
|---|---|---|
| I | Left arm | Right arm |
| II | Left leg | Right arm |
| III | Left leg | Left arm |
Continuous electrocardiographic recording over a 24-hour period (Holter monitoring) or with radiotelemetry is also useful for evaluating horses with arrhythmias. Continuous recording of the ECG can be performed with contact electrodes, electrode patches that are held against the skin with a surcingle, or electrode patches attached to shaved skin with a cyanoacrylate adhesive and protected underneath a surcingle.3,4 The contact electrodes or electrode patches held against the skin with a tight surcingle appear to work best for obtaining a continuous ECG recording. With bipolar contact electrodes, the positive electrode is placed over the left cardiac silhouette or over the sternum and the negative electrode is placed over the dorsum to the left of the withers where the electrode will lie flat and remain in contact with the skin.3 The electrodes are kept moist with alcohol. The electrodes are then covered with moist sponges to maintain contact and are held in position with a tight surcingle. With electrode patches the electrodes are taped to a small square of cardboard to provide them with some rigidity. The best recording is usually obtained with the left arm electrode placed on the sternum, the right leg electrode on the right side in the fifth intercostal space at the level of the point of the shoulder, and the right arm electrode placed on the left side in the fifth intercostal space at the level of the point of the shoulder. These electrodes are then held in position with a tight surcingle after removal of the plastic that covers the conducting material overlying the electrode. The electrodes are connected to a recorder (reel-to-reel, cassette, or digital) that records the animal’s heart rhythm for the entire monitoring period (Holter monitor) or a telemetry device that sends the ECG signal back to the receiver to be displayed on a monitor. The continuous 24-hour Holter monitor is useful for diagnosing arrhythmias that occur intermittently or for monitoring cardiac rhythm during exercise. Radiotelemetry electrocardiography is useful for monitoring cardiac rhythm during treatment or during exercise.
In the base-apex lead the P wave is positive in most horses and ruminants. The P wave is most frequently bifid in horses. In many horses a Ta wave, indicative of atrial repolarization, occurs as a negative deflection after the P wave (Fig. 30-1). The QRS complex begins with a small positive deflection (rS) and is followed by a large negative deflection, which terminates in the ST segment. The T wave is variable and can be positive, negative, or biphasic in horses and ruminants. Frequently the appearance of the T wave is variable within one recording. Fig. 30-2 illustrates a typical base-apex ECG recorded from a cow and a horse at a 25-mm/sec paper speed with the gain set at 10 mm/mV.
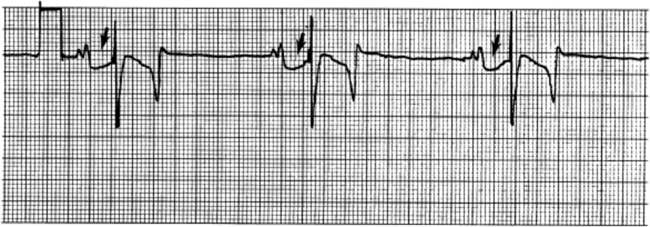
Fig. 30-1 Base-apex lead ECG recorded from a horse. Arrows point to the atrial repolarization wave (Ta) frequently seen in normal horses. It follows the notched P wave and precedes the QRS complex. Paper speed 25 mm/sec, calibration 1 cm/mV.
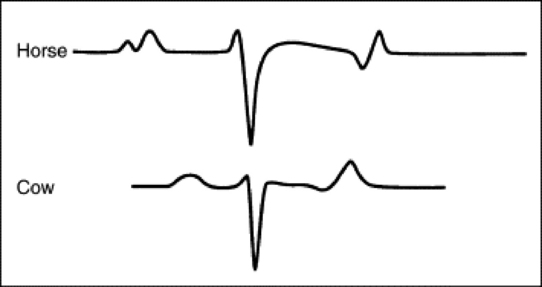
Fig. 30-2 Schematic representation of a typical base-apex lead ECG recorded from a cow and horse. In horses the P and T waves may be variable in appearance.
When a systematic approach is used to analyze the ECG, diagnosing arrhythmias is not difficult. The following step-by-step approach can be used:
USE OF ECHOCARDIOGRAPHY IN LARGE ANIMALS
Echocardiography is a noninvasive diagnostic tool that uses sound waves in the range of 1.5 to 10 MHz to visualize the heart in motion, using either a single icepick (M-mode) or a two-dimensional (B-mode) image. Noninvasive evaluation of blood flow in the heart and great vessels is performed with pulsed wave, color flow, and continuous wave Doppler echocardiography. Precise localization of abnormal flow within the heart and great vessels is performed with pulsed wave and color flow Doppler echocardiography, whereas continuous wave Doppler echocardiography is used to determine the peak velocity of blood flow and to noninvasively estimate pressure gradients. In contrast to M-mode and two-dimensional echocardiography, in which the best image is obtained with the ultrasound beam perpendicular to the structures being imaged, optimal Doppler signals are obtained with the ultrasound beam parallel to the blood flow being evaluated. For accurate peak blood flow velocities to be recorded with continuous wave Doppler echocardiography, the ultrasound beam should be as close to parallel as possible (less than a 20-degree angle) to the direction of blood flow being measured. This alignment is difficult or impossible to achieve in most large animals with valvular heart disease; therefore accurate peak blood flow velocities often cannot be obtained from large animals with valvular heart disease. Noninvasive estimations of pressure gradients are inaccurate in these instances. Alignment of the ultrasound beam parallel to shunt flow is possible with most ventricular septal defects (VSDs) because of the typical location of the VSD in the membranous portion of the interventricular septum. A more extensive review of the theory and application of echocardiography in horses and cattle has been published.5-11
Echocardiography is particularly useful in evaluating large animals with cardiovascular disease because the examination is noninvasive and can be performed in most standing, unsedated animals in a timely fashion. Diagnostic criteria for valvular, myocardial, pericardial, and congenital lesions of the heart are well established, and the information obtained assists the practitioner in confirming a diagnosis, assessing the extent of the disease, determining the severity of cardiac dysfunction, monitoring the response to treatment, and providing an accurate prognosis. Large animal echocardiographic equipment should provide satisfactory resolution of images at depths of 26 cm or greater. Portable ultrasound machines are available that can display depths of up to 36 cm for equine and bovine cardiology. These machines are available with pulsed, continuous wave, and color flow Doppler. However, Doppler echocardiography is used mainly in specialty practices and referral institutions because performing and interpreting a complete echocardiogram, including Doppler, requires a significant amount of training and expertise and state-of-the-art color flow Doppler equipment remains fairly expensive, although it has decreased in price.
Echocardiographic examination is performed in a systematic way, using standardized images to obtain information about chamber size, wall thickness, myocardial function, valve appearance, valve function, great vessels, blood flow, and presence of abnormal structures or echodensities. The standard equine or bovine echocardiogram is performed from the right parasternal window (the right fourth intercostal space in horses and third intercostal space in ruminants) with a 2.5-MHz transducer. Higher-frequency transducers should be used to examine younger animals, South American camelids, and small ruminants. Both long- and short-axis views of all cardiac structures should be evaluated. The cardiac valves should be carefully examined for any abnormalities of structure or function (thickening, prolapse, ruptured chordae tendineae, fenestrations, flail valve leaflet, vegetative lesion, or high-frequency vibrations). The relative size, shape, and relationship of the cardiac chambers and great vessels should be assessed, and an evaluation of myocardial function and blood flow performed. Standard measurements of left ventricular internal diameter, left ventricular free wall thickness, interventricular septal thickness, and right ventricular internal diameter should all be obtained at end diastole and peak systole from the M-mode echocardiogram. The diameter of the aortic root and left atrial appendage, the distance between the interventricular septum and the peak opening of the septal leaflet of the mitral valve (septal to E point separation), and the left ventricular ejection time (ET) should also be determined. End-diastolic measurements are obtained at the Q wave of the ECG, whereas peak systolic measurements are made from the peak downward deflection of the interventricular septum. Calculations of fractional shortening (FS) and ejection fraction (EF) can then be performed to assess left ventricular function using the following formulas:
in which LVIDd is the left ventricular internal diameter at end diastole (cm), LVIDs is the left ventricular internal diameter in systole (cm), and ET is the left ventricular ejection time (sec).
Echocardiograms should also be performed from the left cardiac window when the entire heart cannot be successfully imaged from the right side; atrial fibrillation is present; abnormalities of the mitral valve, aortic valve, pulmonic valve, aorta, pulmonary artery, left atrium, left ventricle, or outflow portion of the interventricular septum are detected; or murmurs originating from the mitral, aortic, or pulmonic valves are detected or a pericardiocentesis is planned. The maximal diameter of the left atrium should be obtained from the left cardiac window at the level of the left coronary artery, parallel to the mitral valve (Fig. 30-3). The diameters of the aorta and pulmonary artery should be measured from similar locations in the vessel on the two-dimensional echocardiogram and compared. Echocardiography should be considered a useful diagnostic test to evaluate patients with the following complaints, physical examination findings, or tentative diagnoses:
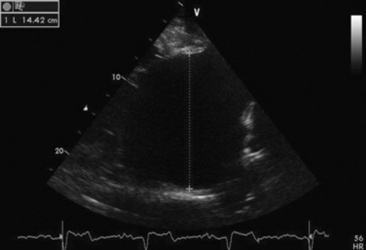
Fig. 30-3 Two-dimensional echocardiographic image of the left atrial diameter obtained from the left parasternal long axis window in a horse with mild left atrial enlargement secondary to cardiomyopathy and mitral regurgitation.
Color flow or pulsed wave Doppler echocardiography should be used to map the size and location of a turbulent jet associated with an intracardiac or extracardiac shunt, valvular regurgitation, or stenosis (rare) and to semiquantitate its severity. Continuous wave Doppler echocardiography can then be used to measure the peak velocity of blood flow in the jet, estimating (noninvasively) the pressure difference between cardiac chambers using the Bernoulli equation and assessing the hemodynamic significance of the lesion. This can be accurately performed in most patients with a VSD but is difficult to impossible to accurately perform in many patients with valvular insufficiencies because of the limited windows available for interrogating blood flow in large animals and the inability to align the ultrasound beam to within 20 degrees of the abnormal blood flow. Contrast echocardiography, a technique involving microbubble-laden injections of saline, carbon dioxide, or indocyanine green, can also be used to demonstrate valve dysfunction and the direction of intracardiac shunts (VSD and atrial septal defect [ASD]) and extracardiac shunts (patent ductus arteriosus [PDA], truncus arteriosus).
CARDIAC CATHETERIZATION IN LARGE ANIMALS
Cardiac and great vessel catheterization can be performed in standing, unsedated large animals to determine the following data:
Cardiac catheterization is also used for special diagnostic studies such as angiocardiography, nuclear angiocardiography, and indicator dilution studies. These data are used to determine the direction and size of intracardiac and extracardiac shunts, chamber size and contractility, and valvular and myocardial function. Much of this same information can now be obtained noninvasively with echocardiography and can help the practitioner establish a diagnosis, more accurately assess the prognosis, and provide a direction for therapy.
Cardiac catheterization is usually reserved for specialty practices and referral institutions because of the equipment needed and the skills required for acquiring and interpreting accurate data. Results are not always specific, but catheterization can add quantitative measurements that increase the accuracy of the diagnosis and prognosis of certain cardiac conditions.
Blood Pressure Measurements
The normal pressures for cattle and horses are listed in Table 30-2.12-15 The values for horses represent a summary of data from numerous authors as cited in the references given. The accuracy of pressure recordings is greatly influenced by the choice of catheter and the recording equipment used. The pulmonary arterial wedge pressure is an indicator of the left atrial mean pressure, as long as balloon inflation of the catheter occludes flow in the segment of the pulmonary artery that is catheterized. Pulmonary arterial wedge pressure is superior to central venous pressure as a monitor of left ventricular function and fluid therapy.
Table 30-2 Cardiac Pressure Measurements in Normal Horses and Cattle*
| Horses12 | Cattle13 | |
|---|---|---|
| RA | 12 to 28/22 to 5 (4 to 10) | (5) |
| RV | 30 to 59/24 to 14 (9 to 25) | 42 to 56/0 to 1 (19 to 28) |
| PA | 34 to 48/14 to 22 (16 to 30) | 33 to 46/19 to 21 (24 to 31) |
| PAW | 13/3 (8) | (5 to 21)11 |
| LV | 140 to 148/15 to 17 | (120 to 144)12 |
| AO | 131 to 144/86 to 100 (110 to 115) | |
| CA | 142 to 157/98 to 119 (113 to 124) | 160 to 208/110 to 147 (135 to 175) |
* Pressure ranges are reported as systolic/diastolic (mean) in mm Hg unless otherwise designated. AO, Aorta; CA, carotid artery; LV, left ventricle; PA, pulmonary artery; PAW, pulmonary arterial wedge; RA, Right atrium; RV, right ventricle.
The shape of the pressure curve in the ventricles, aorta, or pulmonary artery may have diagnostic significance in conditions such as constrictive pericardial disease (Fig. 30-4); pulmonic stenosis (giant A wave), rare as an isolated defect in large animals; or tricuspid or mitral valve regurgitation (large V wave). These conditions are more commonly assessed by echocardiography. An abnormal rise in pressure going from one chamber to the next indicates a stenotic lesion (uncommon in large animals) at the level of the pressure gradient. The size of the pressure gradient can be used to determine the severity of the lesion.
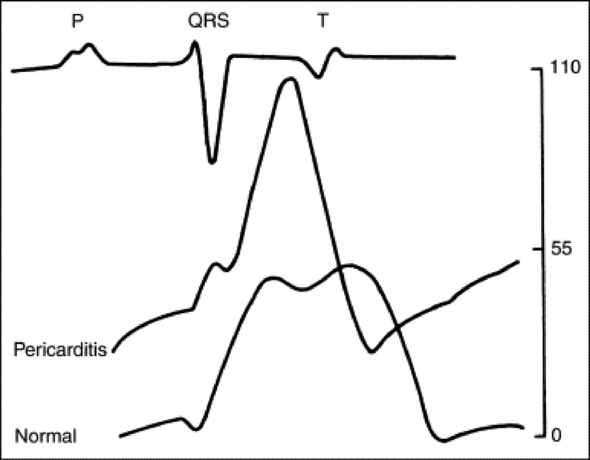
Fig. 30-4 Schematic representation of ventricular pressure curves recorded from a normal horse and a horse with pericarditis, showing the relationship between pressure changes and the ECG. With pericarditis, the ventricular end-diastolic and systolic pressures are elevated and pressure declines sharply in early diastole.
Blood Oxygen Measurements
Blood oxygen measurements are taken from the chambers on the right side of the heart to detect abnormal elevations indicative of a left-to-right shunt (ASD, VSD, PDA). Criteria for oxygen step-ups have not been established for large animals, but human guidelines have been accepted for qualitative assessment of shunts.16,17 In humans, oxygen content step-ups of the following magnitude are considered abnormal and indicative of a left-to>right shunt18:
Oxygen content depends on the hemoglobin concentration; therefore oxygen saturation, which is independent of hemoglobin concentration, may be a more accurate indicator of shunts in anemic or polycythemic patients. Changes in the animal’s physiologic status during sampling (cardiac output, ventilation, oxygen consumption), incomplete mixing of shunted blood, and variable time intervals between sampling can be potential sources of error. Several samples within a single chamber improve the reliability of results. Small shunts and shunts in animals with low systemic arterial oxygen tension may not be detected by this technique.
Shunt calculations can be made once oxygen saturation or content has been measured in each of the right heart chambers, the pulmonary artery, and a systemic artery. For a left-to-right shunt, the pulmonary flow/systemic flow ratio (QP/QS) is determined as follows:
in which SAo2 is the arterial blood oxygen content, MVo2 is the mixed venous blood oxygen content, and PAo2 is the pulmonary artery oxygen content. A 2:1 QP/QS represents a 50% left-to-right shunt, indicating that 50% of the pulmonary flow is from the left side of the heart. For a right-to-left shunt, the QP/QS is determined as follows:
in which PVo2 is the pulmonary venous oxygen content (assumed to be 98% of oxygen capacity plus 0.3 mL of dissolved oxygen).19
Cardiac Output and Ventricular Function Assessment
Cardiac output is determined by indicator dilution methods (usually by dye dilution or thermodilution or, more recently, by lithium dilution), by the Fick method, or by using two-dimensional or Doppler echocardiography. The Fick method requires the use of a face mask and simultaneous determination of mixed venous and arterial blood samples. Dye dilution and thermodilution results are comparable when 30 to 40 mL of 5% dextrose are injected rapidly at 32° F (0° C).17 Lithium dilution compares favorably with thermodilution in anesthetized horses and foals.20,21 Volumetric echocardiography using the Bullet method achieves results similar to those obtained with the lithium-dilution method in anesthetized foals.22 Transthoracic Doppler echocardiography was closely correlated with thermodilution in standing horses.23 Cardiac output values in the resting horse range from 32 to 40 L/min.24 Cardiac index is the cardiac output divided by a measure of body size (the body weight in kilograms) and is expressed in mL/kg/min.24 The normal cardiac index for the adult horse ranges from 72 to 88 mL/kg/min.24,25 Cardiac output values of 20.9 to 23.6 L/min have been reported in normal anesthetized horses.20 In cattle a cardiac index of approximately 110 mL/kg/min has been reported.15 Cardiac output measurements in clinical patients vary with heart rate, excitement, hydration, and many other factors and are best determined in the pulmonary artery. Electronic integration and computation of area under curve by means of battery-powered units that can display results instantly provide the most reliable results.26
Cardiac output results or indicator dilution curves can provide quantitative and qualitative assessment of cardiac shunts. Characteristic changes in the temperature-time curve (thermodilution methods) or dye concentration-time curve indicate the presence of a left-to-right, right-to-left, or bidirectional shunt. Calculation of the cardiac output in the chamber just proximal to the shunt and distal to it can give a quantitative estimate of the size of the shunt.17
Angiocardiography
Angiocardiography is used in neonates or animals small enough to have the entire cardiac silhouette visualized on a single radiograph cassette. The contrast medium must be injected rapidly, and in most cases this is done with a pressure injector. Specialized radiographic requirements include rapid film change capabilities, rapid image sequence acquisition, or cineradiography. Angiocardiography is used to confirm the presence of an intracardiac shunt (ASD, VSD) or extracardiac shunt (PDA, truncus arteriosus) or valve dysfunction, to visualize chamber size, or to estimate contractility. Angiocardiography is performed in anesthetized animals.
Nuclear Angiocardiography
In nuclear angiocardiography, specialized equipment captures sequential digitized images of the right side of the heart, lung, and left side of the heart after rapid injection of radiographic tracer into peripheral circulation. A more extensive review of this subject has been published.27 Nuclear angiocardiography can be used to confirm valvular dysfunction, which is manifested by chamber enlargement or prolonged washout of affected vessels or cardiac chambers and is quantitated by the regurgitant fraction. It also can reveal enlargement of chambers and prolonged washout resulting from cardiac failure. The presence of intracardiac or extracardiac shunts can be demonstrated by the simultaneous visualization of left- and right-side cardiac chambers or slow washout downstream of the shunt. In addition, nuclear angiocardiography can be used to calculate cardiac output and EF and other indices of cardiac function.
CONGENITAL CARDIOVASCULAR DISEASE
The cause of congenital cardiac defects has not been established, although hereditary factors may be responsible for some defects. In humans, additional factors such as maternal infection, age, and nutritional status have been identified. Fetal anoxia from placental insufficiency, fetal infection or metabolic dysfunction, or other causes may contribute to the development of congenital cardiac defects. The same factors may apply in animals. Congenital cardiac defects in large animals can occur alone or in combination. The most commonly reported is VSD.28-33 Multiple cardiac anomalies including PDA,34,35 tetralogy of Fallot,36,37 truncus arteriosus,38,39 total anomalous pulmonary venous connection,40 and Eisenmenger’s complex in calves41 have been reported. Congenital anomalies of the tricuspid,38,42-44 mitral,45 and pulmonic valves38,46,47 are uncommon. Congenital abnormalities of the aorta are reported in calves and foals but are also uncommon.33,48,49 ASD occurs more commonly in calves than in foals and is frequently accompanied by other defects. 33,39,50,51 Hypoplasia of the left and right ventricles has been infrequently reported in calves and foals.33,52,53
Congenital cardiovascular disease should be suspected in a young patient if examination reveals a holosystolic (pansystolic), holodiastolic, or continuous murmur or a murmur with a palpable thrill or wide radiation over the thorax. Cyanosis at rest or with exercise in a patient with a cardiac murmur warrants consideration of a right-to-left cardiac shunt, obstructive pulmonary disease, or severe stenosis of the structures of the right side of the heart. The presence of any of these findings in a young animal with a history of lethargy, weakness, or failure to thrive constitutes grounds to suspect congenital cardiovascular disease.
Ventricular Septal Defect
Definition and Etiology
A VSD is an opening in the interventricular septum that creates a communication between the left and right ventricles. In large animals most defects occur in the membranous septum and are imaged ventral to the septal leaflet of the tricuspid valve and the right and/or noncoronary leaflet of the aortic valve.29-33 VSD can occur as a single defect or as part of a complex anomaly. Many cardiac malformations such as tetralogy and pentalogy of Fallot, truncus or pseudotruncus arteriosus, common atrioventricular canal defect, tricuspid atresia, and double outlet right ventricle include a VSD. The cause of VSD is unknown, although it has been documented to be a heritable defect in Limousine33 and possibly Hereford54 cattle. The defect is thought to result either from failure of fusion of a part of the endocardial cushion and the muscular ventricular septum or failure of fusion of the truncal and conal septa.55
Clinical Signs and Differential Diagnosis
The clinical signs of an isolated VSD vary and depend on the size of the defect, the direction of the shunted blood, and the presence of concurrent valvular or myocardial disease. In isolated VSD the blood flow is shunted from the left ventricle to the right ventricle through the defect in the interventricular septum. The size of the shunt depends on the size of the defect and the pressures in the left ventricle, right ventricle, and pulmonary artery.
VSD is suspected when there is a loud, harsh, plateau-shaped pansystolic murmur with its point of maximal intensity (PMI) in the tricuspid valve area and a slightly softer, more crescendo-decrescendo holosystolic murmur that is loudest in the pulmonic valve area. The murmur on the left side has its PMI in the pulmonic valve area, associated with a relative pulmonic stenosis (increased blood flow across a normal pulmonic valve). A palpable cardiac thrill usually is present over the tricuspid valve region, and occasionally there is splitting of the second heart sound. The murmur may be the only clinical sign identified if the defect is small. On the other hand, poor growth, lethargy, dyspnea, exercise intolerance, and signs of CHF can be exhibited by animals with a moderate to large VSD. This usually develops by the time the animal is 5 years old. Occasionally there is a diastolic murmur of aortic insufficiency associated with a large VSD, the location of which compromises the support of one of the aortic valve cusps.31,32 Cardiac arrhythmias, particularly atrial fibrillation, may be associated with VSD when there is cardiac enlargement or failure.
If the systolic murmur is loudest on the left side of the thorax, a subpulmonic VSD or a complex anomaly with pulmonic stenosis (or some form of right ventricular outflow tract obstruction) should be suspected.31,32,56 The pulmonic murmur is usually louder than the tricuspid murmur in large animals with tetralogy of Fallot. Large animals with tetralogy of Fallot may have cyanosis at rest (uncommon in horses) or with exercise or exertion. Cyanosis is also a distinguishing feature of Eisenmenger’s complex, a defect in which right-sided heart resistance to blood flow causes the shunt associated with VSD to become right to left. Congenital abnormalities of the mitral and tricuspid valves cause a loud systolic murmur audible on both sides of the thorax. The PMI of the left-sided systolic murmur is more caudally located (in the mitral to aortic valve area) than the relative pulmonic stenosis murmur. Usually, the murmur of mitral regurgitation is the loudest of the 2 murmurs. In addition, congenital mitral or tricuspid valve dysplasia is rare in large animals. An innocent flow murmur of neonates can usually be distinguished from VSD by its crescendo-decrescendo shape, PMI at the left heart base, lack of radiation, and low to moderate intensity.
Clinical Pathology
Echocardiography is the diagnostic technique of choice for identifying a VSD. With two-dimensional echocardiography the VSD can be imaged directly (Fig. 30-5) and the shunt size, location, and direction demonstrated with pulsed wave Doppler, continuous wave Doppler, color flow echocardiography, or the injection of microbubbles. Careful scanning of the interventricular septum should be performed with two-dimensional echocardiography to directly image the VSD and measure its maximal diameter in two mutually perpendicular planes.31,32 The typical membranous VSD (≤2.5 cm in both planes) is missed if the long-axis view of the left ventricular outflow tract is not examined. The membranous VSD is located underneath the septal leaflet of the tricuspid valve and the right or noncoronary leaflet of the aortic valve. If a membranous defect is not found, the entire septum should be carefully scanned in all imaging planes to detect the VSD. The subpulmonic location, more common in calves, is easy to miss. This defect is usually best imaged in the short-axis view, scanning the interventricular septum between the left and right ventricular outflow tracts. With a left-to-right shunt a high-velocity turbulent jet is depicted from the right parasternal window, originating in the left ventricle, traversing through the hole in the interventricular septum into the right ventricle with color flow Doppler echocardiography (blood flow toward the transducer), whereas a negative contrast jet is imaged in the right ventricle with a right-sided injection of microbubbles.31,32 A left ventricular injection of microbubbles is necessary to visualize echo-laden blood in the right ventricle with a typical VSD. M-mode echocardiography may show septal discontinuity when traversing the ventricular septum from the apex of the heart to the aortic root (septal dropout). Moderate to large VSDs show left atrial and left ventricular enlargement, right ventricular enlargement, and pulmonary artery dilation. The left atrial—to—aortic root ratio is increased. Aortic valve prolapse and aortic regurgitation may also be detected because of loss of support of the aortic root from the VSD. Myocardial dysfunction and subsequent CHF may occur with a large VSD. Continuous wave Doppler echocardiography can be used to noninvasively assess the hemodynamic significance of the shunt (see Fig. 30-5, B). A peak shunt flow velocity of greater than or equal to 4 m/sec indicates a restrictive VSD with normal or near normal right ventricular pressures. Nuclear angiocardiography can also be used to demonstrate simultaneous opacification of the left and right ventricles. The radiographic presence of cardiomegaly and increased vascularization of the lungs provides only nonspecific evidence of VSD. If polycythemia is found, a complicated VSD should be suspected.
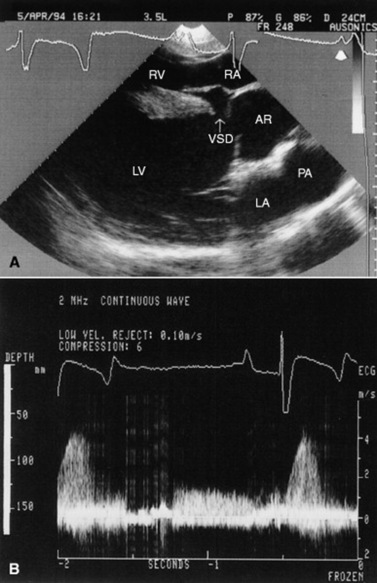
Fig. 30-5 Two-dimensional echocardiographic image (A) and continuous wave Doppler spectral tracing (B) of a ventricular septal defect (arrow) in a weanling colt. The ventricular septal defect (VSD) is located just underneath the septal leaflet of the tricuspid valve and right coronary cusp of the aortic valve. The VSD is best imaged in this left ventricular outflow tract view. There is significant left ventricular enlargement in this colt. The right atrium (RA), right ventricle (RV), left ventricle (LV), left atrium (LA), aortic root (AR), and pulmonary artery (PA) are all visible in this view. The spectral tracing shows a peak shunt velocity of slightly under 4 m/sec in systole, with some turbulent flow (spectral broadening) also detected in diastole with a peak flow velocity of approximately 1.3 m/sec.
Cardiac catheterization can be used for qualitative and quantitative assessment of the VSD but has largely been supplanted by echocardiographic diagnosis. Elevated cardiac pressures provide qualitative evidence of a VSD. Right ventricular systolic pressure elevation is most common and may equal left ventricular pressure with a large VSD. Pulmonary artery pressure can be increased as a result of increased blood flow from the left-to-right shunt or increased pulmonary vascular resistance and decreased flow (restrictive pulmonary hypertension). Elevation of left or right ventricular diastolic pressure provides evidence of cardiac failure in the patient with VSD. Oximetric data (oxygen content or oxygen saturation) can be used to locate the shunt and provide some evidence of the size of the defect. A step-up in oxygen content or saturation between the right atrium and the pulmonary artery suggests a moderate or large VSD. Because most VSDs are located high in the right ventricular outflow tract, there may be inadequate mixing of shunted blood in the right ventricle to detect the shunt in this chamber unless the catheter is directed to the outflow tract for sampling. When the oxygen step-up is detected first in the pulmonary artery, a PDA cannot be ruled out. A small shunt may be missed by oximetry as described earlier under oxygen sampling.
Through the use of indicator dilution methods described previously, the shape of the concentration-time or temperature-time curve can be used to demonstrate the left-to-right shunt of a VSD. A comparison of the cardiac output measured in the right ventricle with the cardiac output measured in the pulmonary artery can give an estimation of the percent of pulmonary blood flow coming from shunted blood and therefore an estimation of the size of the defect.57
Angiocardiography can be performed in the anesthetized neonate and can definitively demonstrate VSD if there is simultaneous opacification of the left and right ventricles when dye is injected into the left ventricle.
Pathophysiology
A small VSD may provide enough resistance to flow that the left-to-right shunt is minimal and the patient remains asymptomatic. Horses can race successfully with small VSDs (≤2.5 cm in diameter with peak shunt velocities ≥4 m/sec), although they are not usually successful as elite racehorses.32 VSDs produce a left-to-right shunt because the pressure in the left ventricle exceeds the pressure in the right ventricle. A peak shunt velocity of 3 to 4 m/sec indicates an increased right ventricular pressure and a less restrictive VSD; however, the defect is usually compatible with a normal life expectancy. A peak shunt velocity of less than 3 m/sec is indicative of a large shunt that is hemodynamically significant. These animals usually develop CHF by 5 years of age. Occasionally the VSD is so large that the pressure between the two chambers is equalized. The right ventricle, pulmonary circulation, left atrium, and left ventricle must compensate for this volume overload, which generally results in dilation of cardiac chambers and the development of pulmonary hypertension.
Pulmonary vascular resistance can increase because of simultaneous pulmonary disease or left-sided heart failure from chronic volume overload. In addition to volume overload, the right ventricle is subjected to a chronic pressure overload, which may be sufficient to reverse the direction of the shunt (Eisenmenger’s complex, more common in cattle than horses). Because of the pressure and volume overload with moderate to large VSDs, patients with this condition run a greater risk of developing CHF.
Considerable turbulence associated with the left-to-right shunts and endocardial damage increase the risk of endocarditis in patients with VSD.58 Because the VSD usually is located high in the left ventricular outflow tract, structural support of the aortic valve cusps may be lost, and aortic insufficiency may develop.32 Significant aortic regurgitation adds to the left ventricular volume overload caused by the VSD.
Epidemiology
The true incidence of VSD in large animals is unknown, although it is recognized as the most common congenital cardiac defect. In one study, 36 calves had 78 congenital cardiac defects, of which 11 were VSDs.33
Necropsy Findings
VSD is usually located high in the interventricular septum just ventral to the right or noncoronary cusp of the aortic valve in the left ventricle and underneath the septal leaflet of the tricuspid valve or caudal or ventral to the crista terminalis in the right ventricle. It can be an isolated defect or can be accompanied by other cardiac or organ anomalies. If the defect is moderate or large, there is right ventricular, left atrial, and left ventricular enlargement and pulmonary artery dilation. The lungs may be congested because of increased pulmonary blood flow, and secondary pneumonia is not uncommon. If pulmonary vascular resistance was increased, right ventricular hypertrophy may be present. There may be secondary endocarditis (infrequent in large animals)58 or endocardial lesions as a result of turbulent blood flow across the defect.
Treatment and Prognosis
There is no practical treatment for VSD in large animals. A complete echocardiographic examination is indicated to identify the presence and significance of a VSD. It is important to identify those animals with moderate to large defects, because the prognosis for normal production or function is poor. Horses with large defects (>3.5 cm) and peak shunt velocities through the defect of <3 m/sec should not be broken to ride as they will develop CHF early in life and have a shortened life expectancy. Animals with small defects may remain asymptomatic throughout life. It is important to recognize that small defects, which provide a large resistance to flow, can produce loud murmurs. Because of this, the intensity of the murmur is not a good predictor of the size of the defect.
Currently there is limited evidence that VSD is inherited in cattle or horses. However, breeding animals with VSD is not advised because of the increased risk of heart failure and other cardiac complications. As a general rule, bull studs do not accept animals with this defect into a breeding program.
Patent Ductus Arteriosus
Definition and Etiology
A PDA is the persistent patency of a vessel (normally present in the fetus) that connects the pulmonary arterial system to the aorta. The ductus arteriosus fails to close at birth when breathing begins and placental circulation is removed. Closure of the ductus arteriosus occurs in response to decreasing pulmonary vascular resistance and increased systemic vascular resistance.
A PDA can occur as a single defect (rare in large animals) or with other cardiac anomalies. In large animals the most common other defects reported with PDA are tetralogy and pentalogy of Fallot and pseudotruncus arteriosus.
Clinical Signs and Differential Diagnosis
The clinical signs of PDA depend on the length and diameter of the ductus arteriosus, the direction of the shunted blood, and the presence of other cardiac defects. A PDA should be suspected when a continuous, high-pitched murmur, frequently referred to as a “machinery murmur” because of its alternating intensity, is auscultated. The murmur may be heard on the left and right sides of the thorax but is usually loudest in the left third or fourth intercostal space at the level of the shoulder. The intensity of the murmur increases with increased heart rate, exercise, or excitement. The arterial pulses are usually bounding because of the runoff of blood from the systemic to the pulmonary circulation. Occasionally the PDA is manifested by a holosystolic murmur59 because the diastolic component is barely audible, except at the left heart base. Large PDAs can exist without producing a murmur. In the animal with increased pulmonary vascular resistance and reversal of the shunt, there may be cyanosis of the caudal parts of the body if the PDA enters the aorta caudal to the brachiocephalic trunk. Stunting of growth may also occur.
Other causes of a continuous murmur in large animals are extremely rare; however, the detection of a continuous machinery murmur should lead the veterinary clinician to suspect a complex congenital cardiac defect that includes a PDA, rather than an isolated defect. In older horses an aorticocardiac fistula should be suspected, particularly if the machinery murmur is of recent onset and is loudest on the right side of the chest. A systolic and diastolic murmur can be present in young animals with a large VSD causing aortic insufficiency. A similar murmur is possible with vegetative endocarditis of one of the atrioventricular or semilunar valves, producing insufficiency and stenosis of the affected valve. These latter conditions should not have the machinery murmur characteristic of the PDA. A loud systolic ejection murmur, which is confused with a PDA, frequently can be heard at the left heart base of foals shortly after closure of the PDA. This murmur may persist for 2 to 3 months.34
Clinical Pathology
No characteristic clinicopathologic changes are associated with a PDA. Radiography may show enlargement of the cardiac silhouette and pulmonary overcirculation in an uncomplicated PDA. Pulmonary venous congestion, interstitial pulmonary edema, and alveolar edema are evidence of a large PDA with left-sided heart failure. These signs are not specific for PDA and can be present with any congenital heart defect that results in a left-to-right shunt. No consistent electrocardiographic pattern has been identified with PDA. Echocardiographic evidence of a PDA is provided by the detection of an enlarged left atrium and left ventricle with a pattern of left ventricular volume overload and increased values for the ratio of the left atrial to aortic root dimension.31,59-61 Direct visualization of the ductus arteriosus is difficult with echocardiography but is most successful when performed from the left cardiac window. A PDA arising from the pulmonary artery has been imaged echocardiographically in an 11-month-old Friesian-Holstein heifer.35 High-velocity turbulent flow throughout the cardiac cycle in the pulmonary artery and ductus arteriosus is detected with pulsed wave, continuous wave, or color flow Doppler echocardiography.60
Cardiac angiocardiography and nuclear angiocardiography using a selective aortic angiogram provide definitive evidence of a PDA. Oximetric data show a step-up in oxygen content or saturation in the pulmonary artery that is proportional to the size of the shunt. Indicator dilution methods also provide evidence of a left-to-right shunt occurring in the pulmonary artery in cases of an uncomplicated PDA. Pulmonary arterial and right ventricular pressures may be increased with a large PDA.
Pathophysiology
Normally the ductus arteriosus narrows near term and constricts rapidly after birth in response to lowered pulmonary vascular resistance, increased systemic vascular resistance, increased blood volume, and increased left ventricular pressure when breathing begins and the placental circulation is removed. If the ductus arteriosus is large or the resistance to flow across the ductus is minimal, there is a significant left-to-right shunt, which produces a large left ventricular volume overload. The left ventricular response may be failure or, with time, dilation (primarily) and hypertrophy. Pulmonary hypertension and congestion result. The right ventricle can be affected by the pulmonary pressure load, and right ventricular hypertrophy can also develop. If the pulmonary resistance equals or exceeds the systemic vascular resistance, a right-to-left shunt occurs.
Epidemiology
Normal foals may have a PDA for a few days after birth, but closure of the ductus arteriosus is expected by 96 hours of age.59 Normal ruminants rarely have a PDA after birth, and if one is present it is considered abnormal. Functional closure may precede anatomic closure of the PDA. This defect is uncommon in older animals. Currently there is no evidence to suggest that this is an inherited defect in horses or cattle.
Necropsy Findings
The ductus arteriosus can be of variable length and diameter but is patent between the aorta and the pulmonary artery. The PDA often enters the aorta caudal to the origin of the brachiocephalic trunk. Changes in the left and right ventricles and lung and pulmonary vasculature are variable and depend on the size of the shunt. When the PDA is large, there may be cardiomegaly with left atrial and left ventricular dilation, right ventricular hypertrophy, pulmonary congestion, and edema.
Treatment and Prognosis
There is insufficient evidence on which to base a prognosis for animals with PDA. The condition can be corrected surgically in neonates, but future performance has not been documented. Animals with small defects may remain asymptomatic throughout life. The prognosis is poor if the defect is large, because the risk for left (primarily) and right ventricular failure is increased. Pharmacologic closure of the PDA using inhibitors of prostaglandin synthesis has been successful in humans but is not without risk of complications and recurrence. The efficacy of prostaglandin inhibitors has not been evaluated in large animals.
Tetralogy and Pentalogy of Fallot
Definition and Etiology
Tetralogy and pentalogy of Fallot are characterized by biventricular origin (overriding) of the aorta, VSD, right ventricular hypertrophy, and obstruction of pulmonary arterial flow. When there is an associated ASD, the anomaly is referred to as pentalogy of Fallot. The defect is caused by abnormal development of the conal septum in the embryonic heart, which leads to narrowing of the right ventricular infundibulum (pulmonic stenosis), an inability of the conal septum to participate in closure of the interventricular foramen (VSD), and overriding of the aorta. Right ventricular hypertrophy develops as a result of the pulmonary outflow obstruction.
Clinical Signs and Differential Diagnosis
Tetralogy of Fallot is one of the more common congenital cardiac defects that cause cyanosis in large animals. Resting cyanosis is rare in horses, although it may be detectable after exercise. Cyanosis of the oral and nasal mucosa, the tongue, the vaginal mucous membranes, and occasionally the nose and skin of light-colored animals is noticed when more than 5 g/dL of hemoglobin are reduced (unoxygenated). Exercise intolerance is often marked and is characterized in most cases by dyspnea or collapse. Frequently the owner complains of slow growth or small size. A loud pansystolic murmur, which is associated with a palpable thrill, is loudest in the left third to fourth intercostal space. The murmur may be a crescendo-decrescendo murmur of pulmonic stenosis or the harsh, plateau-shaped murmur of a VSD; one of these usually predominates. A harsh band-shaped pansystolic murmur is also auscultated in the tricuspid valve area but is usually one or two grades softer than the pulmonic stenosis murmur. Excitement of the animal may result in auscultation of a gallop rhythm or an early systolic ejection click. A continuous machinery murmur can be auscultated in some patients, associated with continuous shunting through the PDA.
Tetralogy and pentalogy of Fallot must be distinguished from other causes of cyanosis in young animals. Respiratory distress syndrome of neonates can be distinguished by the presence of tachypnea, dyspnea, and abnormal lung sounds in the absence of a cardiac murmur. Cyanosis caused by central nervous system disease has other neurologic manifestations. Cyanosis from congenital cardiac disease may be caused by a right-to-left shunt or by heart failure with pulmonary edema. Cyanosis resulting from heart failure or respiratory disease improves with oxygen administration, whereas the patient with a right-to-left cardiac shunt fails to improve. Right-to-left cardiac shunting does or can occur with tetralogy and pentalogy of Fallot, reverse PDA or VSD, tricuspid valve or right ventricular atresia, left ventricular hypoplasia, persistent truncus arteriosus, pseudotruncus arteriosus, and other complex congenital cardiac disease, all of which may occur with cyanosis and a cardiac murmur. A complete echocardiographic examination using a segmental approach to cardiac anatomy is needed to accurately diagnose the correct congenital cardiac malformation and has widely supplanted other methods of diagnosing complex congenital cardiac disease in large animals. Radiography and cardiac catheterization provide supplemental information that may be helpful in distinguishing among the causes of right-to-left cardiac shunting.
Clinical Pathology
Increased packed cell volume (PCV), red blood cell count, and hemoglobin concentration (polycythemia) may be present in some animals with tetralogy and pentalogy of Fallot.37 However, polycythemia is uncommon in foals with cyanotic congenital cardiac disease and is usually less than 45% in most calves. Electrocardiographic changes are usually nonspecific, but a right-axis deviation may be detected.37 Radiographs of the lungs may show decreased vascularity. The four components of tetralogy of Fallot are easily visualized echocardiographically. The VSD and overriding aorta usually are clearly visible with two-dimensional echocardiography. The malalignment VSD is usually large and located just below the right cusp of the aortic valve, separated from the pulmonic valve by the crista supraventricularis. The aortic root is usually large and overrides the septal defect. Echocardiography shows increased thickness of the right ventricular wall, ventricular septal hypertrophy, paradoxic septal motion, and similar left and right ventricular internal dimensions. Narrowing of the right ventricular outflow tract, pulmonic stenosis, or a hypoplastic pulmonary artery (most common) may be imaged as the cause of the right ventricular outflow tract obstruction. Pulsed wave and color flow Doppler echocardiography can be used to further characterize the abnormalities of blood flow associated with tetralogy of Fallot, in particular the severity of the right ventricular outflow tract obstruction. Contrast echocardiography also nicely demonstrates the path of blood flow with a peripheral venous injection. Contrast echoes are imaged entering the right ventricle from the right atrium, then simultaneous opacification of the pulmonary artery, left ventricle, and aorta occurs.
Cardiac catheterization can be used to demonstrate equalization of ventricular pressures and a pressure gradient between the right ventricle and pulmonary artery. Oximetry should demonstrate decreased oxygen content in the left ventricle compared with the pulmonary vein. Angiocardiography demonstrates simultaneous filling of the right ventricle, left ventricle, and overriding aorta with decreased pulmonary artery filling and increased right ventricular trabeculation (hypertrophy).
Pathophysiology
VSD is usually large, resulting in equalization of pressures in the two ventricles and the aorta. The degree of shunting is controlled by the resistance across the stenotic right ventricular outflow tract compared with the resistance across the aortic valve. If the right ventricular outflow tract is severely obstructed, the clinical signs of cyanosis are more marked. Excitement, drugs, or increased myocardial contractility from any cause decreases right ventricular volume and worsens clinical signs. Right ventricular failure usually is not a consequence of the pressure overload because of equalization of the ventricular pressures.
Epidemiology
The prevalence of tetralogy and pentalogy of Fallot in large animals has not been documented, but these defects seem to be more common in calves than in foals. There is no evidence that these disorders are inherited.
Necropsy Findings
Examination of the heart reveals a rounded apex caused by right ventricular enlargement. A high, usually large VSD, an overriding aorta that straddles the VSD and the left and right ventricle, right ventricular hypertrophy, and septal hypertrophy are present. There is usually right ventricular infundibular narrowing and a hypoplastic pulmonary artery, although there may be valvular pulmonic stenosis with a poststenotic dilation. The right and left atria may be enlarged.
Treatment and Prognosis
There is no practical treatment for tetralogy and pentalogy of Fallot in large animals. When cyanosis or exercise intolerance is present or growth is stunted (the latter two are common findings in affected animals), the prognosis for long-term survival, production, or performance is poor. Affected foals should not be used for performance or broken to ride if they live long enough. As with many congenital cardiac diseases, the intensity of the murmur is not a good predictor of the severity of the condition, and further diagnostic tests are indicated.
Other Congenital Cardiac Defects
ATRIAL SEPTAL DEFECT
ASD is a connection between the left and right atria at the septal level. The most common type of defect is the ostium secundum defect, of which patent foramen ovale is seen most frequently. Patent foramen ovale is relatively common in calves and is caused by the failure of the septum primum, the valve of the foramen ovale, to become adherent to the crista dividends after birth, when changes in left and right atrial pressures produce functional closure of the foramen ovale. Patent foramen ovale is frequently associated with PDA in calves.33
Animals frequently with an ASD are asymptomatic, but a holosystolic, crescendo-decrescendo murmur may be heard at the left heart base. The shunt is usually left to right, and the murmur is the result of increased volume being ejected across the pulmonic valve. If the defect is large, right atrial, right ventricular, and left atrial dilation may be present. Differential diagnostic considerations are a functional murmur, pulmonic stenosis, VSD, or PDA. A definitive diagnosis can be made by two-dimensional echocardiography in which an enlarged right atrium, right ventricle, and left atrium are imaged. Pulsed wave Doppler, color flow, or contrast echocardiography can be used to demonstrate the shunt through the ASD.
PULMONIC VALVE STENOSIS
Pulmonic valve stenosis is uncommon as a single defect but has been reported in a foal with VSD and as one of multiple defects in calves and foals.33,47 Clinical signs of cardiac murmur, cyanosis, and polycythemia are variable and depend largely on the other cardiac defects present. Characterization of the severity of the pulmonic stenosis and other associated cardiac defects can be performed with a complete echocardiographic examination.
TRICUSPID VALVE ATRESIA
Tricuspid valve atresia has been reported in foals38,42-44 in conjunction with other cardiac defects. The abnormalities associated with tricuspid atresia include patent foramen ovale, VSD, small right ventricle, large left ventricle, and large mitral valve orifice. The foals showed cyanosis and a crescendo-decrescendo or band-shaped holosystolic or pansystolic murmur audible over the left and right heart base. Tachycardia, tachypnea, and weak peripheral pulses also were present. Polycythemia was commonly reported. Echocardiographic diagnosis of tricuspid atresia in foals has been reported.38,43 A thick echo in the region of the tricuspid valve that does not separate in diastole (absent tricuspid valve), an ASD (usually patent foramen ovale), a VSD, a small right ventricle, a large left ventricle, and a large mitral valve orifice are the echocardiographic findings in tricuspid atresia. Blood flow (right to left) through the patent foramen ovale into the left atrium and left ventricle followed by simultaneous opacification of the aorta and right ventricle is detected with contrast echocardiography. Necropsy showed tricuspid atresia, along with ASD, VSD, small right ventricle, and large left ventricle. Pulmonic valve stenosis and dextropositioning of the aorta have also been reported.
MITRAL VALVE DYSPLASIA
Mitral valve dysplasia is an early developmental anomaly that has been reported in the horse.45 The affected foal had a grade 4-5/6 holosystolic decrescendo-type murmur loudest in the mitral to aortic valve area. The mitral valve leaflets appeared thickened, bright, and irregularly nodular echocardiographically. A cleft was imaged in the midportion of the free wall leaflet with small papillary muscles and shortened, unevenly thickened chordae tendineae.45
VENTRICULAR HYPOPLASIA
Ventricular hypoplasia has been reported in foals and calves.33,52 The defect may be present with other cardiac defects and is usually associated with early death. The defect was present in three closely related Holstein calves, suggesting possible genetic factors.33
TRUNCUS OR PSEUDOTRUNCUS ARTERIOSUS
Persistent truncus arteriosus refers to the condition in which one arterial vessel leaves the heart above a VSD. The coronary and pulmonary arteries and aorta arise from this vessel. Persistent truncus arteriosus has been diagnosed in foals and calves.39 Subclassifications of this condition have been applied to humans, depending on the origin of the pulmonary trunk or arteries. Pseudotruncus arteriosus has also been described in foals and a calf and is characterized by the presence of a remnant of an atretic pulmonary trunk.38,62 With pseudotruncus arteriosus, the pulmonary blood supply comes from bronchial arteries or a PDA. Clinical manifestations of these conditions include tachycardia, exercise intolerance, and a cardiac murmur. The murmur may be a continuous machinery murmur if a PDA is also present, holosystolic and crescendo-decrescendo and loudest at the left heart base, or the coarse murmur of the VSD may be auscultated, although the relative pulmonic stenosis component is absent. Cyanosis, dyspnea, or syncope may be seen with exercise or excitement. CHF and stunted growth may be noticed. Polycythemia was detected in a calf with a pseudotruncus arteriosus.62 The presence of cyanosis with the cardiac murmur helps differentiate this condition from a simple VSD or PDA. Definitive diagnosis may be made by echocardiography, angiocardiography, or nuclear angiocardiography.
AORTIC ANOMALIES
Dextropositioning or transposition of the aorta are the most common aortic anomalies of foals and calves and are seen most frequently with other defects. Other aortic anomalies of foals and calves are persistence of the right aortic arch and double aortic arch, which may cause esophageal compression. The clinical presentation is one of esophageal obstruction. Interruption of the aortic arch in two foals with VSD, ASD, and PDA has been reported.49 The foals showed weakness, lethargy, cyanosis, and tachycardia. The murmur was pansystolic and plateau-shaped, with the PMI on the right side of the thorax. Radiology showed cardiomegaly and increased vascularization of the lungs. Cardiac catheterization showed left ventricular failure. Bicuspid and quadricuspid cusps of the aortic and pulmonic valves occur in large animals and usually result in both stenosis and valvular insufficiency. An aneurysm of the sinus of Valsalva was detected in a 3-year-old thoroughbred gelding as an incidental finding.63 Occasionally a diastolic murmur may be associated with aortic insufficiency in horses with sinus of Valsalva aneurysms.63,64 These sinus of Valsalva aneurysms usually rupture later in life, and the horse develops acute distress, colic, and uniform ventricular tachycardia. A continuous machinery murmur is usually present on the right side of the thorax, associated with the presence of an aorticocardiac fistula.
EISENMENGER’S COMPLEX
Eisenmenger’s complex has been described in a stunted, 24-month-old Holstein heifer that had a loud, crescendo-decrescendo, pansystolic murmur heard best over the pulmonic valve.41 The heifer had a prominent gallop rhythm from a loud fourth heart sound and exercise intolerance without cyanosis. Polycythemia was present, however. Cardiac catheterization showed increased pressures in the right atria, right ventricle, and pulmonary artery, with normal left-sided pressures. The echocardiogram was characterized by a VSD, overriding aorta, and dilation of the pulmonary trunk, a feature that distinguished this from tetralogy of Fallot. Left ventricular function was decreased, and at necropsy the heart was enlarged and rounded with a dilated pulmonary trunk and small aorta. The right ventricle was dilated and hypertrophied, whereas the left atria and ventricle were only mildly dilated.
ECTOPIA CORDIS CERVICALIS
Ectopia cordis cervicalis is a relatively common defect of cattle.33,48 Although this defect usually results in the heart being in the cervical region, a few animals may have the heart in the pectoral region (14%) or the abdomen (3%).48 Various defects are associated with ectopia cordis cervicalis, including defects of the heart, great vessels, neck (torticollis), ribs, and sternebrae. The heart is usually contained within the pericardium under the muscles of the skin in the ventral cervical area, with the double apex of the heart pointing craniodorsally. The ligaments of the pericardium are most frequently attached to the mandibles and the parotid fascia cranially, the cervical fascia laterally, and the first rib or manubrium caudally. The lung may lack the cardiac notch and often protrudes to the base of the heart. Although the prognosis for a productive life is poor, some calves lived until approximately 1 year of age.
MISCELLANEOUS CARDIAC DEFECTS
Other cardiac defects can occur, but the significance of the lesion is questioned or the defect has been recorded infrequently. Complete atrioventricular canal defect has been reported in a foal.51 Anomalous coronary artery development has been reported at postmortem examination, but the lesion was not necessarily the cause of death in a calf.65 Anomalous origin of the coronary artery has been thought to be the cause of death in horses.66 Congenital hematomas of the atrioventricular valves also have been noted, but the significance is unknown.33 Endocardial fibroelastosis, an anomalous development of the endocardium associated with left ventricular hypertrophy, is usually a severe defect resulting in death of the animal. The frequency of this defect in large animals is not established.
VALVULAR HEART DISEASE
Definition and Etiology
In adult animals, disorders of the tricuspid, pulmonic, mitral, or aortic valves are usually acquired and most commonly result in insufficiency of the affected valve. These disorders may be the result of degenerative changes, infection (bacterial or viral endocarditis or myocarditis), inflammation (valvulitis), trauma, or unknown causes (cardiomyopathy). They are usually manifested by a cardiac murmur, most frequently of valvular regurgitation, with the PMI at the location of the affected valve or in the direction of the regurgitant blood flow. The mitral and aortic valves are the most common location of degenerative valve disease in horses.67 Predisposing causes such as microembolism or infarction have not been identified in large animals. Chronic active infection such as foot abscesses, rumenitis, reticular abscess, or other septic process may lead to sustained or recurrent bacteremia, predisposing the animal to the development of bacterial endocarditis, particularly in cattle, or a nonvegetative valvulitis, probably more common in horses. Experimentally, valvular vegetative endocarditis can be induced by intravenous administration of bacteria without preliminary damage to a valve.68 Rupture of a valve leaflet or chordae tendineae can cause valvular heart disease, as can dilation of a cardiac chamber from any cause or rupture of the aortic root or of a sinus of Valsalva aneurysm.69-71 In rare cases neoplasia, primarily lymphosarcoma of cattle, can cause valvular heart disease. Congenital valvular heart disease in adult animals is rare. The most common bacterial isolates from equine and bovine endocarditis cases are streptococci and Pasturella or Actinobacillus species and Arcanobacterium (Actinomyces) pyogenes in horses and cattle, respectively, although a wide variety of organisms has been isolated from large animals with endocarditis.72,73,75-79
Clinical Signs and Differential Diagnosis
Most animals with valvular heart disease have no clinical signs but have a cardiac murmur that is detected during a routine examination. The clinical signs vary depending on the severity of the lesion and its rate of development. Murmurs of valvular heart disease are frequently holosystolic (Fig. 30-6), pansystolic, or holodiastolic (Fig. 30-7). They radiate from the PMI in the direction of the abnormal blood flow; are coarse and band-shaped, crescendo or honking (if systolic), or decrescendo and blowing or musical (if diastolic); and are usually moderate to loud in intensity (≥grade 3/6) but may be softer if holodiastolic. All of these characteristics help distinguish these murmurs from functional or innocent murmurs, which generally occur early or late in systole or diastole but can be holosystolic; are soft and blowing or crescendo-decrescendo in quality; are localized to a small area; do not radiate; and are soft to moderate in intensity (≤3/6). The intensity of the murmur is not a reliable indicator of the severity of the lesion, except in horses with tricuspid regurgitation, in which the longer, louder murmurs are associated with a larger jet of tricuspid regurgitation.80 In cattle, in particular, severely involved valves (usually in cattle with endocarditis) commonly have faint or no audible murmurs.
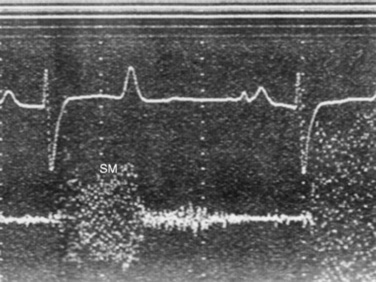
Fig. 30-6 Phonocardiogram obtained from a horse with a ruptured mitral valve chorda tendineae. A loud, plateau-shaped holosystolic murmur (SM), which is variable in intensity, occurs when the free wall leaflet of the mitral valve is prolapsing into the left atrium.
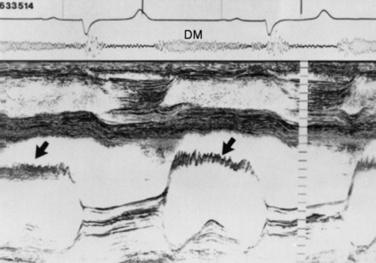
Fig. 30-7 Phonocardiogram and M-mode echocardiogram obtained from a horse with a holodiastolic murmur (DM) caused by aortic valve regurgitation. The M-mode echocardiogram shows diastolic flutter (arrows) of the septal mitral valve leaflet, characteristic of aortic valve insufficiency.
The location of the PMI of the murmur is helpful in distinguishing which valve is involved, although more than one valve can be affected in the same animal. The PMI for lesions of the mitral valve frequently is at the left apex of the heart, although murmurs of mitral regurgitation usually radiate dorsally and toward the left heart base and aortic valve area. Therefore loud systolic murmurs with the PMI in the aortic or mitral valve area in horses are usually mitral regurgitation murmurs. Disorders of the tricuspid valve commonly have the PMI on the right side of the thorax (third to fourth intercostal spaces [horses] or second to third intercostal spaces [cattle]). Infrequently, the murmur may also be heard on the left side of the thorax cranial to the pulmonic valve location in the second intercostal space. Aortic and pulmonic valve lesions produce murmurs with the PMI at the left heart base in the third or fourth intercostal space. Acquired valvular lesions of the mitral and tricuspid valves produce primarily systolic murmurs.72,73,75,80-84 Diastolic tricuspid flow murmurs have been reported, however, and may be associated with right-sided mural or valvular masses in horses85 or may be physiologic, associated with normal blood flow across the atrioventricular valves.86,87 Lesions of the aortic and pulmonic valves may produce diastolic murmurs, systolic murmurs, or both.72,88-90 However, diastolic murmurs of regurgitation are most common in large animals. Aortic regurgitation associated with degenerative valve disease is most common in horses, whereas pulmonic regurgitation associated with bacterial endocarditis is more common in cattle. Aortic valve lesions in horses have primarily holodiastolic, decrescendo, musical murmurs (see Fig. 30-7) but can also be decrescendo, soft, and blowing. Aortic regurgitation murmurs are accompanied by a water-hammer or bounding arterial pulse if the aortic regurgitation is associated with a significant left ventricular volume overload.72,91 The musical quality of the murmur (harmonic) indicates that some part of the aortic valve is vibrating during diastole. The arterial pulse quality becomes more bounding as the aortic regurgitation becomes more severe, and is a good clinical indicator of the degree of left ventricular volume overload. Ventricular premature beats and atrial fibrillation may also be detected in horses with significant aortic regurgitation.
Besides the cardiac murmur, animals with valvular heart disease may have exercise intolerance, weight loss, or signs of CHF evidenced by tachycardia, coughing, respiratory distress, jugular venous distention, subcutaneous edema, and ascites (uncommon in large animals). In adult cattle, mammary vein distention is another sign of CHF.73 Cardiac enlargement may be noted as an increased area of auscultation and/or percussion or caudal dislocation of the apical impulse of the heart. Atrial fibrillation may be present. This development is usually an indicator of atrial enlargement in animals with valvular heart disease. If tricuspid valve regurgitation is present, there may be abnormal systolic jugular venous pulsations. If mitral regurgitation is present there may be tachycardia, tachypnea, poor recovery to resting respiratory rate after exercise, coughing, and frothy pulmonary edema. Lung sounds may be harsh at rest and on deep inspiration may include rare crackles or moist bubbly sounds. Most horses with pulmonary edema have only harsh breath sounds that are detected at rest and on deep inspiration.
One of the clinical signs of bacterial endocarditis is a cardiac murmur, the PMI and timing of which depend on the valve or valves affected. Other signs may include tachycardia, arrhythmias, auscultation of prominent heart sounds, tachypnea, coughing, recurring fever, anorexia, weight loss, or signs of CHF. Evidence of disseminated sepsis such as pneumonia, hematuria, or pyuria is usually present. Shifting leg lameness and swollen joints or tendon sheaths are common but usually have an immune-mediated cause, although a horse with bacterial endocarditis and septic tenosynovitis has been reported.76 Mastitis and decreased milk production are common in cattle. The presence of weight loss, fever, and signs of recurring sepsis help distinguish bacterial endocarditis from other forms of acquired valvular disease.
The clinical signs of a major mitral valve chordal rupture (major chorda tendineae) or its characteristic murmur distinguish this disease from other mitral valve diseases. The murmur is usually a widely radiating murmur of mitral valve regurgitation (see Fig. 30-6) with a distinctive honking quality (again the honking quality is consistent with vibration of the mitral valve chorda tendineae or leaflet with blood flow in systole). However, the honking quality may be absent and replaced by a band-shaped pansystolic murmur. There may be evidence of acute hemodynamic collapse. Acute onset of respiratory distress with coughing and expectorating foamy pulmonary edema fluid (this fluid is also detected at the external nares) is a relatively consistent feature with rupture of a major chorda tendineae. Signs of right-sided heart failure (jugular venous distention, subcutaneous edema, and ascites) may develop rapidly. Atrial arrhythmias, most frequently atrial fibrillation, often develop secondary to atrial enlargement. Supraventricular arrhythmias have also been reported in foals with a ruptured mitral chorda tendineae.83 The acute onset of respiratory distress, along with a honking systolic murmur, distinguishes mitral valve chordal rupture from other causes of mitral regurgitation. The honking systolic murmur of a ruptured mitral valve chorda tendineae can also be heard in the absence of any clinical signs in horses with a minor chordal rupture. The murmur of mitral valve prolapse is also a distinctive murmur and should be suspected in horses whenever a mid- to late-crescendo systolic murmur is auscultated with the PMI over the mitral valve area.92 A similar murmur is frequently auscultated in horses with tricuspid valve prolapse. Murmurs of mitral or tricuspid valve prolapse can be detected in horses with all degrees of valvular insufficiency. Most frequently, however, only small amounts of valvular regurgitation are associated with valvular prolapse. An increased prevalence of mitral and tricuspid regurgitation has been reported in young horses in training.93
Clinical Pathology
Diagnosis of valvular disease is best performed with a complete echocardiographic examination including M-mode, two-dimensional, and Doppler echocardiography. Two-dimensional echocardiography is superior to M-mode for detection of valvular abnormalities (Figs. 30-8 and 30-9), measurement of valvular masses (Fig. 30-9, B), and the global assessment of ventricular function, but chamber enlargement, high-frequency vibrations of the valve leaflets, and shortening fraction (an indication of ventricular systolic function) can be determined by both. Pulsed wave, continuous wave, and color flow Doppler echocardiography can be used to semiquantitate the severity of valvular regurgitation.92-97 The size of the regurgitant jet detected with pulsed wave or color flow echocardiography is one indicator of the severity of the valvular insufficiency.98 The duration of the mitral regurgitation jet is also important in assessing severity. Clinically insignificant jets of regurgitation are detected only just behind the valve when it is closed. Valvular insufficiency is mild when the jet occupies one third or less of the receiving chamber, moderate when the jet occupies greater than one third but less than two thirds of the receiving chamber, and severe when the jet occupies greater than two thirds of the receiving chamber.
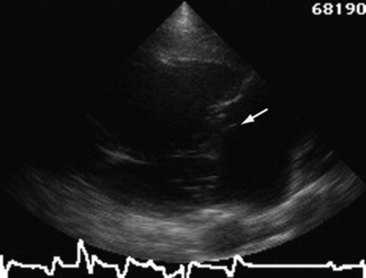
Fig. 30-8 Two-dimensional echocardiographic image obtained from the left parasternal window of a ruptured mitral chorda tendineae (arrow) in the left atrium of a gelding with mitral regurgitation and a honking pansystolic murmur.
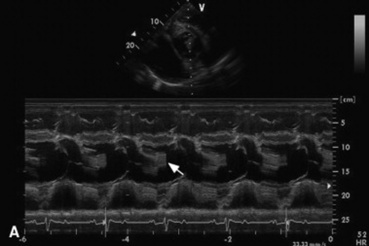
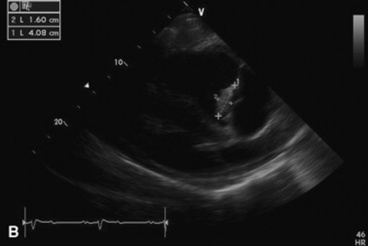
Fig. 30-9 Echocardiograms of a bacterial endocarditis lesion on the aortic valve of a horse. The lesion can be seen on the aortic valve M-mode (A) as a thick echoic band (arrow) and is measured in the two-dimensional echocardiographic image of the left parasternal long axis view (B).
Echocardiographic signs of mitral regurgitation are increased left atrial (Fig. 30-10; see also Fig. 30-3) and left ventricular dimensions and a left-sided volume overload. The cause of the valvular regurgitation can often be determined. Endocarditis (see Fig. 30-9), ruptured mitral valve chordae tendineae (see Fig. 30-8), a flail valve leaflet, valvular prolapse, or thickening of the valve leaflet are readily imaged echocardiographically.99 The regurgitant jet detected with pulsed wave or color flow Doppler echocardiography usually originates from the site of the valvular abnormalities detected with two-dimensional echocardiography. In some animals, the lesion responsible for the valvular insufficiency is not visualized with two-dimensional echocardiography but the regurgitant orifice is detected with pulsed wave or color flow Doppler echocardiography.95-97100 Left ventricular function may be normal (if the mitral regurgitation is mild) or the FS may be increased (if there is a significant left ventricular volume overload associated with moderate to severe mitral regurgitation), unless there is concomitant myocardial disease. A ruptured mitral valve chorda is diagnosed by finding a mobile linear echo everting into the left atrium or a flail leaflet that may prolapse into the left atrium during systole (see Fig. 30-8), systolic and chaotic diastolic mitral valve flutter, rapid mitral valve opening with increased excursion of the affected leaflet, and lack of coaptation of the mitral valve in systole. The asynchronous movement of any portion of the valve leaflet during any phase of the cardiac cycle indicates the presence of a flail valve leaflet.81,99 A larger than normal pulmonary artery (larger than the aortic root) is compatible with severe pulmonary hypertension and left-sided heart failure. A smaller than normal aortic root is detected echocardiographically in horses in low-output left-sided heart failure.83,98,99
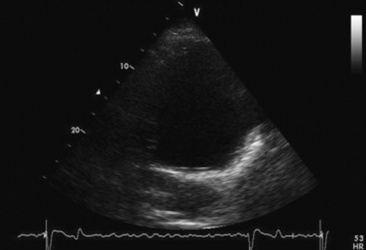
Fig. 30-10 Left parasternal echocardiographic image of the left atrium and mitral valve from a horse with left atrial enlargement (15.55 cm).
Tricuspid regurgitation may produce echocardiographic evidence of right atrial and right ventricular enlargement with paradoxic septal motion. Frequently the cause of tricuspid valve regurgitation in cattle is bacterial endocarditis, and the incompetent valve can be visualized. In cattle, neoplasia of the right atrium, tricuspid valve, or right ventricle can usually be visualized when present.101 Tricuspid regurgitation is common in horses with no obvious valvular lesion.87,93,102
Aortic valve regurgitation is diagnosed echocardiographically by observing left ventricular dilation, increased aortic root diameter (Fig. 30-11), increased left ventricular FS (if the aortic regurgitation is moderate to severe and left ventricular function is normal), diastolic fluttering of the septal mitral valve leaflet (Fig. 30-12; see also Fig. 30-7), or, less frequently, by observing high-frequency vibrations of the interventricular septum or aortic valve in diastole.89,91,96,98 Rarely, premature closure of the mitral valve is detected. Thickening of the left cusp of the aortic valve is frequently detected echocardiographically, but prolapse, fenestration, and tears of the aortic cusps also occur.
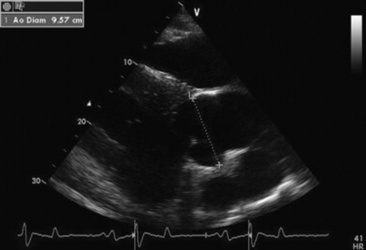
Fig. 30-11 Two-dimensional echocardiographic image of an enlarged aortic root (9.57 cm) obtained from an aged gelding with moderate aortic regurgitation that has been present for several years.
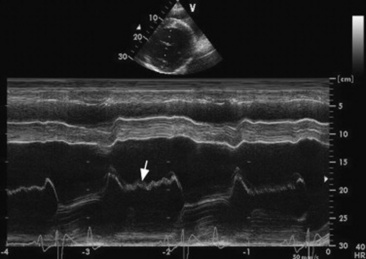
Fig. 30-12 M-mode echocardiogram obtained from a stallion with moderate chronic aortic regurgitation. Notice the high-frequency vibrations (arrow) on the septal leaflet of the mitral valve.
Acquired pulmonic valve lesions are uncommon in large animals and when present are usually associated with bacterial endocarditis.83,90,103 Diagnosis is established by finding the mass associated with the pulmonic valve. Severe pulmonic regurgitation associated with pulmonic valve rupture has been reported in one horse.104 Pulmonic regurgitation is most common in horses with pulmonary hypertension and CHF but is rarely detected clinically.
Lesions of bacterial endocarditis (see Fig. 30-9) may have a shaggy, ragged, or cystic appearance on two-dimensional echocardiography.*Occasionally the only abnormality noticed is valve thickening with ventricular hyperkinesis and enlargement of the chambers on the side of the affected valve. Acoustic reverberation and production of microbubbles have also been associated with valvular bacterial endocarditis.83,86 Other laboratory evidence of bacterial endocarditis includes anemia, neutrophilia (a left shift may be present), an increased serum globulin concentration, and hyperfibrinogenemia. Liver enzymes are frequently mildly elevated, and a urinalysis sometimes shows hematuria or pyuria. Positive blood cultures taken during febrile episodes confirm the diagnosis when associated with the above findings. However, many times the culture results are negative in large animals with bacterial endocarditis. Other laboratory evidence of disseminated sepsis includes neutrophilic response in peritoneal, tracheal wash, or joint fluid. Ventricular arrhythmias are frequently detected electrocardiographically in individuals with mitral or aortic valve endocarditis. Radiographic or sonographic evidence of disseminated pneumonia also may be found with bacterial endocarditis, usually in large animals with a right-sided lesion.
Other laboratory evidence of valvular heart disease is nonspecific. The ECG is not reliable for detecting chamber enlargement associated with valvular incompetence. The ECG is valuable for documentation of cardiac arrhythmias occurring secondary to chamber enlargement or the underlying myocardial disease. Radiographic findings of cardiac enlargement, increased pulmonary vascular pattern, or pulmonary edema are also nonspecific. Cardiac catheterization can be performed, and pressure measurements help determine the degree of cardiac dysfunction. Nuclear angiocardiography shows cardiac enlargement or prolonged washout of contrast material. Time-activity curves can be helpful in accurately documenting ventricular dysfunction and the valvular regurgitant fraction.106
Pathophysiology
Acquired valvular heart disease that is slow in onset or gradually progressive may be asymptomatic at first, but cardiac changes occur that eventually may lead to CHF. Valvular incompetence from endocarditis, degenerative changes, chordal rupture, or other causes results in volume overload of the recipient chamber. Initially the output of the chamber is increased to maintain forward output, but the increased end-diastolic volume of the recipient chamber leads to compensatory dilation and a mild elevation in end-diastolic pressure. Compensatory hypertrophy may also result. In the later stages of valvular regurgitation, contractile function of the volume-overloaded chamber may diminish, leading to further elevation of end-diastolic pressure and decreased compliance. In the case of severe aortic and mitral regurgitation, this causes elevation of left atrial pressure and eventually pulmonary venous hypertension. Pulmonary artery wedge pressure is higher at rest and during exercise in horses with moderate mitral regurgitation.107 With severe tricuspid regurgitation, right atrial or central venous pressure increases. Increased myocardial oxygen consumption is a natural sequela, and biventricular failure can ensue.
If valvular heart disease is acute in onset, as with major chordal rupture of the mitral valve, the regurgitation and volume overload are imposed on the left atrium, which cannot dilate and adapt acutely to the increased diastolic filling. The sudden hemodynamic change leads to pulmonary venous hypertension and acute pulmonary edema. However, most horses with mitral regurgitation do not have acute fulminant pulmonary edema. Instead, chronic pulmonary hypertension leading to subtle respiratory signs associated with interstitial pulmonary edema and the subsequent development of right-sided CHF are common in horses with severe mitral regurgitation.99 In one study 12 of 14 horses with CHF had jugular distentions or pulsations, 10 had abnormal lung sounds (crackles), and nine had a cough.108
Bacterial endocarditis of the mitral or tricuspid valve may lead to rupture of the chordae tendineae. Mitral chordal rupture has been reported in adult horses and foals with bacterial endocarditis, leading to acute left-sided CHF.83 In addition to the hemodynamic load placed on the heart from an incompetent valve, bacterial endocarditis also results in disseminated sepsis. The vegetations are made up of layers of fibrin, blood cells, necrotic tissue, and bacteria and are relatively resistant to short-term antimicrobial therapy. The disseminated sepsis may be the cause of death or the reason for culling of the animal. Myocardial necrosis of a papillary muscle was associated with mitral chordal rupture in one foal.83
Epidemiology
Acquired valvular heart disease is common in large animals, involving 356 of 1557 horses (22.9%) in one abattoir survey.67 Endocarditis, one form of valvular heart disease, was reported in 4% of cattle in another study.109 Bacterial endocarditis most commonly affects the tricuspid valve in cattle but has been reported on the pulmonic, mitral, and aortic valves as well.73,89,90,101 Most cases of tricuspid valve bacterial endocarditis have been reported in horses with septic jugular vein thrombophlebitis.72,75,110 Tricuspid valve endocarditis has also been reported in a horse with a VSD.85 Aortic and mitral valve endocarditis are most common in horses, occurring with nearly equal frequency.74,75 In the largest survey of acquired valvular heart disease in horses, the aortic valve was affected most commonly with degenerative valve changes, followed by the mitral valve, tricuspid valve, and, uncommonly, the pulmonic valve.67 Not all valvular lesions are associated with incompetency of the valve. Degenerative valvular changes, particularly changes involving the aortic valve, are seen more commonly in older horses.
Necropsy Findings
Acquired valvular disease is associated with finding nodular thickening, fibrous bands, valve fenestrations, rupture of mitral valve chordae tendineae, fibrinous masses typical of endocarditis, and combinations of these lesions on postmortem examination. There may be associated or secondary changes varying from enlargement of a chamber or vessel to hematomas, degeneration, inflammation, and fibrosis. Enlargement (primarily dilation) of the chamber receiving the regurgitant flow, in addition to enlargement (primarily dilation) of the chamber or vessel from which the regurgitant flow arises, is commonly detected. Jet lesions, usually found in the receiving chamber, are associated with the high-velocity turbulent regurgitant blood flow.
Subcutaneous edema; increased pericardial, pleural, or peritoneal fluid; and congestion and mottling of the liver may be present and indicate CHF. Ascites associated with CHF is uncommon in large animals, particularly in horses. A primary source of chronic or active infection, along with evidence of bacterial embolization, may be seen in cattle with endocarditis but is rare in horses.
Treatment and Prognosis
The treatment and prognosis of acquired valvular heart disease depend on the cause, onset, duration, and severity of the lesion. In general, the prognosis is guarded to poor when evidence of valvular incompetence includes tachycardia, exercise intolerance, signs of CHF, or echocardiographic evidence of severe chamber enlargement. Degenerative valve disease may be asymptomatic except for a cardiac murmur, or it may be mild; but it generally is slowly progressive and therefore historically has been given a guarded prognosis. A more accurate prognosis for horses with murmurs can now be obtained from a complete echocardiographic examination, including Doppler echocardiography. The valve affected, the lesions detected on the valve leaflets, the degree of chamber enlargement and volume overload detected, the echocardiographic assessment of myocardial function, and the severity of the regurgitation determined with Doppler echocardiography, coupled with the animal’s age and intended use, can be used to formulate a prognosis.96,98 Valvular regurgitation associated with no detectable abnormalities, valvular prolapse, and mild valvular thickening usually has a fair to good prognosis if the amount of valvular regurgitation is small. Individuals with ruptured chordae tendineae, flail valve leaflets, and marked valvular thickening usually have moderate to severe regurgitation, which is likely to progress more rapidly and usually warrants a guarded to poor prognosis. Mitral regurgitation is the most likely valvular insufficiency to be associated with clinical signs of cardiovascular disease, whereas primary aortic regurgitation and tricuspid regurgitation infrequently result in the development of CHF or the death of the animal, except in horses with severe aortic regurgitation. Bacterial endocarditis has a guarded to grave prognosis, even with long-term antibiotic therapy, and frequently results in sudden death of the animal, although bacteriologic cures have been reported.
Despite the guarded long-term prognosis, palliative therapy can be applied for most forms of acquired valvular heart disease. Bacterial endocarditis is treated with long-term administration, ideally intravenously (IV), of bacteriocidal antimicrobials, the choice of which is based on blood culture and sensitivity results. Continuous intravenous infusion of antimicrobials is the ideal initial treatment, when feasible. In cattle and horses, initial therapy is directed at the likelihood of a gram-positive infection. Combination antibiotic therapy consisting of a penicillin and an aminoglycoside can be used for gram-negative organisms. Using rifampin at a dose of 5 mg/kg twice daily orally (PO) in combination with another antibiotic with appropriate spectrum has improved the short-term outlook for large animals with bacterial endocarditis. Aspirin (100 mg/kg/day, ruminants; 17 mg/kg every other day, horses) and low-dose heparin (30 U/kg subcutaneously [SC] twice daily, ruminants and horses) may be used in patients with valvular endocarditis in an attempt to prevent platelet adhesion and increased size of the valvular mass. Early diagnosis and aggressive treatment with the appropriate antimicrobials for a prolonged period are important for successful treatment of bacterial endocarditis. The long-term outcome for successfully treated cases is still poor because the scarring that results as the endocarditis lesion heals may lead to severe valvular regurgitation and the death of the animal, particularly with left-sided bacterial endocarditis lesions.75,76,78,98,111 Compliance and economics are also major drawbacks in treating endocarditis.
The hemodynamic consequences of valvular heart disease (volume overload or CHF) may be improved by using diuretics. Furosemide has been used most commonly at a dose of 0.5 to 1 mg/kg as needed. With the low bioavailability of oral furosemide, intravenous administration is preferred to obtain the maximal diuretic effect.112 Digoxin can be used to improve contractility when CHF has occurred. Conditions such as aortic valve or mitral valve regurgitation may show little or no long-term improvement, although many individuals improve for 2 to 6 months before the CHF becomes refractory to treatment. The administration of a maintenance dose of digoxin at 2.2 μg/kg IV twice daily or 11 μg/kg PO twice daily has resulted in therapeutic plasma digoxin concentrations and clinical improvement in horses with CHF with no significant adverse effects.113 Loading doses are rarely used, because of problems with digoxin toxicity.114 An intravenous loading dose of 12 to 14 μg/kg can be used in horses but must be divided into three administrations, followed by a maintenance dose of 6 to 7 μg/kg daily.115 Digoxin can also be administered PO to horses at a dose of 34 to 70 μg/kg for loading and 17 to 35 μg/kg for maintenance.115 Digoxin is administered IV to cattle at a loading dose of 22 μg/kg followed by a maintenance dosage of 11 μg/kg three times daily or, preferably, at an infusion rate of 0.86 μg/kg/hr.116 The clinical impression is that horses, as do other species, benefit from the use of vasodilators. Clinical and echocardiographic improvement has been seen in several horses with moderate mitral or aortic regurgitation treated with angiotensin-converting enzyme (ACE) inhibitors. Quinapril at 120 mg/horse/day resulted in an increase in stroke volume and cardiac output that was statistically significant after 8 weeks of treatment in horses with mitral regurgitation.117 Clinical and echocardiographic improvement has been seen in horses with CHF. Clinical improvement has been seen with hydralazine at 0.05 to 1.5 mg/kg PO twice daily. The ACE inhibitor enalapril has also appeared to be effective in horses with moderate to severe mitral or aortic regurgitation and in horses with CHF, at a dose of 0.5 mg/kg PO twice daily. Ramipril was beneficial in the treatment of one horse with CHF at 50 μg/kg daily PO.118 Until recently the expense of these drugs limited the usefulness of enalapril and the other ACE inhibitors. However, a generic form of enalapril maleate is now available, making this treatment more affordable for most owners. Although the short-term outlook may be improved with use of cardiovascular drugs, the prognosis is guarded for long-term survival or production.
Prevention and Control
Many causes of acquired valvular heart disease cannot be controlled. Appropriate therapy for chronic active infections and careful attention to asepsis with intravenous medication and other invasive procedures may help prevent endocarditis and, possibly, nonvegetative valvulitis. Effective parasite control measures may eliminate some predisposing causes of valvular heart disease such as trauma to heart valves, microembolism, or infarction in horses.
BRISKET DISEASE: COR PULMONALE AND PULMONARY HYPERTENSION
Definition and Etiology
Cor pulmonale is a term used to refer to the effect of lung dysfunction on the heart and is therefore a secondary form of heart disease. Regardless of the cause, the underlying feature is pulmonary hypertension that leads to right ventricular hypertrophy, dilation, or failure. The primary cause of the disease (also called high-mountain disease or high-altitude disease in cattle) is hypoxic vasoconstriction from high-altitude dwelling. The disease is worsened by the ingestion of locoweed (Oxytropis and Astragalus species).119,120 Chronic pulmonary disease such as bronchopneumonia or lungworm infection in cattle also can result in cor pulmonale.121 In the horse, right-sided heart dysfunction (e.g., decreased cardiac output, increased right ventricular end-diastolic pressure, increased right atrial or central venous pressure, jugular venous distention, or subcutaneous edema) associated with chronic pulmonary disease is rare but may be more common than previously recognized. However, right-sided heart dysfunction associated with pulmonary hypertension and left-sided heart dysfunction is a common cause of CHF.
Clinical Signs and Differential Diagnosis
Frequently the primary presenting clinical sign of brisket disease is subcutaneous edema of the brisket, the ventral thorax, the submandibular area, and occasionally the limbs. Jugular venous distention or pulsations may be present. Dyspnea and tachypnea frequently are exhibited. Tachycardia is present, and a gallop rhythm may be auscultated. Splitting of the second heart sound (S2) is a variable finding. Pulmonary hypertension may accentuate the separation of the aortic and pulmonic valve closures, producing audible splitting of S2 that is most noticeable during inspiration. In some horses with moderate or severe recurrent airway obstruction (RAO) and increased vascular impedance; however, pulmonic valve closure occurs early, and only a single S2 is audible.122
A cardiac murmur may be auscultated, caused either by tricuspid insufficiency or a pulmonic valve ejection murmur. The murmur of tricuspid insufficiency, which is secondary to right ventricular dilation, is regurgitant or plateau-shaped with the PMI over the right thorax or, less frequently, the left second intercostal space. The pulmonic valve ejection murmur, found less commonly, is audible as a crescendo-decrescendo murmur at the left heart base.123 Pleural or pericardial effusion is not common with cor pulmonale.124 In horses with RAO leading to cor pulmonale, tachypnea, labored breathing, coughing, and exercise intolerance are common complaints. Wheezes are usually auscultated bilaterally in the thorax of affected horses.
These clinical signs are not specific for brisket disease (cor pulmonale) and largely reflect right-sided heart failure. Other considerations when signs of right-sided heart failure are present should be bacterial endocarditis or tricuspid insufficiency from any cause, cardiomyopathy, cardiac lymphosarcoma or other thoracic neoplasms, pericarditis, left-sided heart failure, pleuritis or pleural effusion, and congenital pulmonic valve stenosis (rare). With pericarditis, heart sounds may be muffled, or the characteristic “washing machine murmur” may be audible. Left-sided heart failure is frequently accompanied by pleural effusion, pulmonary edema, and weak peripheral pulses. In horses, however, expectoration of pulmonary edema fluid occurs only with sudden onset of severe left-sided heart failure. In horses with chronic left-sided heart failure, tachypnea, coughing, poor recovery after exercise, and harsh lung sounds may be all that is detected.
Clinical Pathology
A complete blood count may reveal a neutrophilia in cases of cor pulmonale caused by primary lung disease. Radiography of the thorax reveals primary pulmonary disease such as bronchopneumonia, bronchiectasis, or chronic bronchitis in cattle that have developed this disease at low altitudes. Radiographic findings in horses with RAO usually reveal an interstitial pattern. Arterial blood gases demonstrate the presence of hypoxia and may also show hypercapnia. Transtracheal wash fluid cytology and bacterial culture may provide evidence of the cause of the primary lung disease. Fecal sedimentation helps rule out parasitic bronchitis or pneumonia. Electrocardiographic findings are not specific. An echocardiogram may provide further evidence of pulmonary hypertension and right-sided heart dysfunction by showing right ventricular hypertrophy and dilation, increased septal thickness, and abnormal septal motion (Fig. 30-13).125,126 Dilation of the pulmonary artery is also detected with two-dimensional echocardiography and is a sensitive indicator of pulmonary hypertension or increased flow through the pulmonary artery. A dilated pulmonary artery has been reported in horses with cor pulmonale and in horses with acute pulmonary obstruction from RAO.126,127 Cardiac catheterization reveals elevated pressure in the pulmonary artery, right ventricle, and right atrium. Elevation of right ventricular end-diastolic pressure is a sign of right ventricular failure.
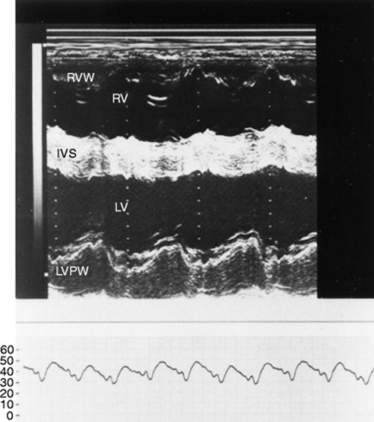
Fig. 30-13 M-mode echocardiogram and pulmonary artery pressure curve obtained from a cow with pulmonary hypertension and cor pulmonale. The echocardiogram shows mild thickening of the right ventricular wall (RVW) and interventricular septum (IVS), dilation of the right ventricle (RV), and abnormal septal motion. LV, Left ventricle; LVPW, left ventricular free wall. The pulmonary artery pressure curve demonstrates elevated systolic and diastolic pressures. (Pulmonary artery pressure curve in millimeters of mercury [mm Hg].)
Pathophysiology
Pulmonary arteriolar constriction is the response to hypoxia from high-altitude dwelling or pulmonary disease. The response to hypoxia varies, depending on the amount of smooth muscle in the pulmonary arteries. In cattle, increased pulmonary vascular resistance and pulmonary hypertension frequently develop. Chronic pulmonary artery hypertension causes a pressure overload on the right ventricle, which responds to the increased work load with hypertrophy, dilation, or failure, depending on the speed with which the condition develops. The disease is progressive, and at some stage the right ventricular myocardium is unable to compensate, dilates, and fails. With failure come the typical signs of jugular venous distention and development of subcutaneous edema. Chronic right-sided heart failure may result in diastolic dysfunction of the left ventricle.
Epidemiology
The disease is more common in cattle than in other animal species, especially when cattle are kept at altitudes over 6000 feet, in which cases the incidence has been estimated at 0.5% to 2%.128 Mainly calves are affected, and some herds with a higher prevalence are thought to have a genetic predisposition to develop pulmonary arterial constriction when subjected to hypoxia at high altitude. Ingestion of locoweed (Oxytropis and Astragalus species) also predisposes cattle to right-sided heart failure at high altitudes by causing toxic myocardial damage.119
Necropsy Findings
There is evidence of right-sided heart failure such as subcutaneous edema in the submandibular area, brisket, and ventral abdomen. Findings include dilation and hypertrophy of the right ventricle, congestion of the liver, and ascites. The lung may have changes of pneumonia, bronchitis, bronchiectasis, or emphysema. Lesions of locoweed toxicity may be found in other organs in cattle.119 Lesions of chronic interstitial pulmonary disease are found in horses with RAO.
Treatment and Prognosis
Removing the animal from high altitude, treating the primary lung disease, and administering oxygen may help eliminate the hypoxia and thus the pulmonary artery hypertension. Cor pulmonale from high altitude is potentially reversible when the animal is returned to a lower altitude. The heart failure can be treated with digoxin and diuretics. Beneficial effects of vasodilator therapy have not been documented. Once heart failure signs have developed, the prognosis is guarded even with appropriate treatment. Removing the horse from the environment inducing RAO is critical to the treatment of the horse with cor pulmonale. A more detailed discussion of the treatment and management of horses with RAO can be found in Chapter 31.
Prevention and Control
The selection of breeding stock with low or normal pulmonary arterial pressures at altitudes above 5000 feet (mean pulmonary artery pressure less than 35 mm Hg) helps to eliminate the predisposing genetic susceptibility to the effects of hypoxia of high altitude.128 The disease can also be controlled by removing susceptible animals from high altitude and preventing locoweed ingestion. Herd health practices such as avoiding crowding and poor ventilation and using appropriate vaccinations to reduce the incidence of pulmonary disease help to reduce the incidence of other causes of cor pulmonale.
MYOCARDIAL DISEASE: MYOCARDITIS AND CARDIOMYOPATHY
Definition and Etiology
Myocarditis is an inflammation in the myocardium caused by bacterial, viral, or parasitic organisms or thromboembolic disease caused by these organisms. In large animals, recognized bacterial causes include Staphylococcus aureus, Streptococcus equi, Clostridium chauvoei, and Mycobacterium species. Myocarditis also can occur after bacteremia, septicemia, pericarditis, or endocarditis, regardless of the causative agent. Known viral causes of myocarditis include foot-and-mouth disease, equine infectious anemia, equine viral arteritis, equine influenza, and African horse sickness. Parasitic causes of myocarditis may include strongylosis129 or onchocerciasis in horses, and toxoplasmosis, cysticercosis, or sarcocystic infection in ruminants.130,131 Currently it is thought that infection with the spirochete Borrelia burgdorferi may be a cause of myocarditis in domestic animals, as it is in human beings.132 Cardiomyopathy is a subacute or chronic disease of the ventricular myocardium that occurs without anatomic valvular disease, congenital malformations of the heart or vessels, or pulmonary disease. In large animals, dilated cardiomyopathy is the only cardiomyopathy of known significance. Dilated cardiomyopathy is associated with ventricular dilation, increased ventricular mass, and decreased systolic function. Although the cause is frequently undetermined,133-135 several conditions have been related to or identified with cardiomyopathy. The most common cause of cardiomyopathy in horses is probably myocarditis, although the inciting insult is usually difficult to identify. An inherited cardiomyopathy, which is thought to be linked to the red Holstein gene, has been found in Holstein-Friesian cattle in Canada, Japan, Australia, the Netherlands, and Switzerland.136-139 There is also a cardiomyopathy associated with a curly hair coat in polled Herefords.140 Cardiomyopathy has been associated with ingestion of monensin, lasalocid, salinomycin, gossypol, Cassia occidentalis, and Phalaris species. Vitamin E and selenium deficiency, copper deficiency, excessive molybdenum, or high sulfates (secondary copper deficiency) may cause cardiomyopathy.141-150 Myocardial infiltration by neoplasia such as lymphosarcoma or fibrosarcoma also may cause cardiomyopathy. A fibrofatty infiltration of the right ventricle and interventricular septum that appears to be similar to arrhythmogenic right ventricular dysplasia has been reported in horses.151
Clinical Signs and Differential Diagnosis
The manifestations of myocarditis are highly variable, depending on the extent of the disease, location of the inflammation within the myocardium, and associated systemic illness. Myocarditis can easily be missed because of the lack of specific cardiac signs or the predominance of signs related to the organ system with primary involvement (e.g., strangles in horses or mastitis in cattle). Animals with myocarditis are often febrile or have a recent history of fever and may have tachycardia. The elevated heart rate may be caused by sinus tachycardia or other paroxysmal or sustained supraventricular or ventricular arrhythmias, because cardiac arrhythmias are common. Premature beats are also commonly detected in large animals with myocarditis and may be present in animals with a normal or increased heart rate. Occasionally, acute myocarditis is associated with auscultation of a pronounced gallop rhythm or a cardiac murmur of either tricuspid or mitral valve insufficiency. Jugular venous distention, other signs of CHF such as peripheral edema, and signs of circulatory collapse may be present. Horses may show evidence of myalgia, reluctance to move, or exercise intolerance. With these signs it may be difficult to distinguish myocarditis from colic, respiratory disease, lameness, or septicemia. If signs are more clearly cardiac (e.g., murmur, arrhythmia, jugular venous pulsations), the practitioner should rule out endocarditis, cardiomyopathy, cardiac neoplasia, and CHF from another cause.
The clinical signs associated with dilated cardiomyopathy can also be variable but are usually more clearly associated with heart disease. Most animals have signs of cardiac failure such as peripheral edema and jugular venous distention or pulsations (Fig. 30-14). Auscultation frequently reveals tachycardia, gallop rhythm, muffled heart sounds, or a cardiac arrhythmia. Cardiac murmurs of tricuspid or mitral valve insufficiency are usually present secondary to the ventricular dilation. The breath sounds are increased, tachypnea is present, and, frequently, percussion of the thorax reveals signs of pleural or pericardial fluid. Occasionally, respiratory distress is evident with hyperpnea, dyspnea, coughing, and the presence of a bloody froth at the nostril. Nonspecific signs of cardiomyopathy such as exercise intolerance, syncope, diarrhea, or anorexia may also be present. In cattle, decreased milk production and abomasal displacement have been associated with dilated cardiomyopathy.136 Palpable liver enlargement has also been reported in cattle with cardiomyopathy.137 Most affected cows develop clinical signs within 3 months of calving.137
Fig. 30-14 Holstein cow with cardiomyopathy showing signs of CHF (submandibular and brisket edema, jugular venous distention, and pulsations).
Sudden death can be a feature of myocarditis or dilated cardiomyopathy, and it frequently follows stress or exercise. Recumbency, collapse, and sudden death are common presenting signs in animals with ionophore toxicosis.141-147149 In many animals with inherited cardiomyopathy, death occurs within 6 months of age.138,140 In Holstein-Friesian cattle with suspected inherited cardiomyopathy, heart failure and death may not occur until cattle are 2 to 4 years old.136,137 Other acquired causes of cardiomyopathy can occur at any age.
When animals show clinical signs of dilated cardiomyopathy, many differential diagnoses should be considered. If the animal is young, other congenital heart defects, cor pulmonale, and nutritional myodegeneration should be considered. In adults, bacterial endocarditis, cardiac neoplasia, thoracic abscess, pericarditis, pleuritis, and diaphragmatic hernia may also have similar clinical manifestations. Although the cause of dilated cardiomyopathy may be difficult to determine, nutritional (vitamin E, selenium, or copper deficiency), toxic (monensin, gossypol, salinomycin, lasalocid, Cassia species, or Phalaris), infectious (viral, bacterial, or parasitic) or drug-induced causes should be investigated.
Clinical Pathology
Depending on the cause of the myocardial disease, the clinicopathologic findings may vary. Routine complete blood count may be normal, or a neutrophilic leukocytosis may be present. No consistent changes are expected in the serum chemistry profile, but serum albumin concentration may be slightly decreased and serum urea nitrogen, creatinine, γ-glutamyltranspeptidase (GGT), sorbitol dehydrogenase (SDH), and bilirubin concentrations may be increased because of congestive liver disease when signs of CHF are present. Serum concentrations of creatine kinase (CK) and lactic dehydrogenase (LDH) may be elevated, but hepatic and skeletal muscle contribution to these elevations cannot be ruled out. A better indication of myocardial disease may be the elevation of cardiac troponin I (cTnI) or the myocardial isoenzymes of CK (myocardial bound [MB]) and LDH (LDH1). Large elevations of the myocardial isoenzymes of CK and LDH were detected in one outbreak of monensin toxicosis144 and have been detected in other horses with myocardial necrosis of unknown etiology. Marked elevations of cTnI have also been reported in a horse with multiform ventricular tachycardia and myocardial necrosis of unknown cause.152 and in a horse with ventricular tachycardia and aortic septal rupture.153 Elevations of cTnI were detected in a recent herd exposure to lethal concentrations of monensin. Mild elevations of cTnI were also reported in two Belgian horses with atypical acute monensin toxicosis.150 Analysis of pericardial or pleural fluid reveals a transudate with low protein concentration (less than 2.5 g/dL) and cellularity (white blood cell [WBC] count less than 2500/μL), with the predominant cell type being mononuclear cells, unless the myocarditis is an extension of pericarditis. Serum should be tested for serologic evidence of bovine leukosis (BLV) infection in adult cattle, α-tocopherol, glutathione peroxidase, and copper concentrations. Whole blood selenium levels should be determined. In infectious myocarditis or inherited cardiomyopathy, the results of these tests will most likely be normal. A negative BLV test result essentially eliminates lymphosarcoma as a cause of the myocardial disease; a positive test result does not confirm a causal relationship. In horses, serum should be tested for serologic evidence of a variety of equine viruses, particularly influenza, equine viral arteritis, and herpes virus. Hemoglobinuria, if present, suggests consideration of monensin, gossypol, or nutritional myodegeneration as the cause of myocardial disease. Arterial blood oxygen tension may be reduced below 80 mm Hg in animals with myocardial disease.
An ECG may demonstrate sinus tachycardia or other cardiac arrhythmias; the frequency and severity of these arrhythmias are best demonstrated with continuous electrocardiographic monitoring. There may be some evidence of conduction abnormalities in the base-apex lead, but these findings are not specific for myocardial disease. Echocardiography may be normal in animals with myocarditis, or the abnormalities detected may be caused by the arrhythmias present. A small left ventricular internal diameter, thickened left ventricular free wall and interventricular septum, small aortic root diameter, and decreased FS may be detected in animals with sustained ventricular tachycardia and decreased cardiac output (Fig. 30-15). More frequently, however, there is increased ventricular chamber size, decreased thickness of the interventricular septum and left ventricular free wall, and decreased myocardial function (decreased FS, decreased EF, and myocardial dyskinesis), especially in more severe cases of myocarditis and in animals with dilated cardiomyopathy (Fig. 30-16). In humans with myocarditis there may be evidence of abnormal left ventricular wall motion, paradoxic motion of the interventricular septum, or an echo-free pericardial space.154 Additional echocardiographic features of dilated cardiomyopathy are increased end-systolic and end-diastolic dimensions of the left and right ventricles (see Fig. 30-16), increased left atrial size, and an increased left atrial to aortic root dimension ratio. There may be abnormal mitral valve closure (increased EF slope) and increased separation of the septal mitral valve leaflet and the interventricular septum. A decreased aortic root diameter may be detected, along with a shortened ET, in animals with low-output left-sided heart failure. An enlarged pulmonary artery indicative of pulmonary hypertension (Fig. 30-17) may also be present.
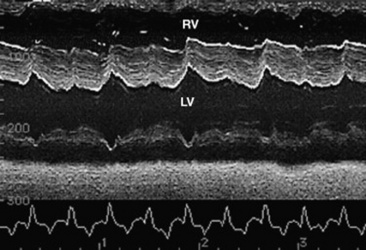
Fig. 30-15 M-mode echocardiogram of the left ventricle (LV) obtained from a mare with sustained uniform ventricular tachycardia. Note the small end-diastolic and end-systolic dimensions of the left ventricle and the little variation between systole and diastole in this dimension. RV, Right ventricle.
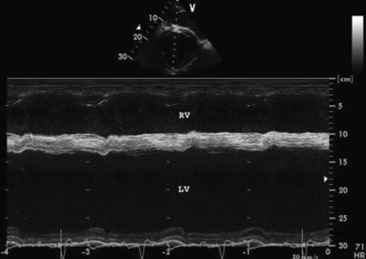
Fig. 30-16 M-mode echocardiogram of the left ventricle obtained from a mare with cardiomyopathy and severe left ventricular dysfunction. Note the large end-diastolic dimension of her left ventricle (LV), the thin interventricular septum and left ventricular free wall, and the poor left ventricular contractility. The fractional shortening in this mare varied from 2% to 8%. RV, Right ventricle.
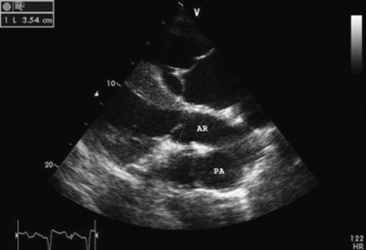
Fig. 30-17 Two-dimensional echocardiogram of the aorta (AR) and main pulmonary artery (PA) obtained from a foal with cardiomyopathy and severe mitral and tricuspid regurgitation. The pulmonary artery is larger than the aorta, indicative of pulmonary hypertension and impending left-sided CHF. The aorta measured 3.54 cm at its origin.
Cardiac catheterization may reveal elevated intracardiac pressures (right atrial, right ventricular, pulmonary artery, pulmonary capillary wedge, and left ventricular end-diastolic) in animals with dilated cardiomyopathy. Humans with acute infectious myocarditis may show hypotension and a narrow pulse pressure. Nuclear angiocardiography may be used to show decreased EF155 in animals with myocardial disease.
Pathophysiology
The pathophysiologic changes associated with myocardial disease depend on the specific nature and extent of the disease. Acute myocarditis or angiopathic myocardial lesions129 may go on to develop into idiopathic dilated cardiomyopathy. It is speculated that changes in cellular metabolism occur with either acute myocarditis or dilated cardiomyopathy, resulting in ECG abnormalities and reduced myocardial performance (reduced cardiac output). In an attempt to compensate for reduced cardiac output, circulating fluid volume is increased by activation of the renin-angiotensin-aldosterone system, and arterial resistance is increased. These compensatory mechanisms frequently lead to only minor improvement in the cardiac output of the failing heart, and the increase in ventricular preload (venous return) and afterload (arterial resistance) may cause pulmonary edema and a further reduction in cardiac contractility. Ventricular dilation, further reduction in cardiac output, and signs of heart failure occur.
Epidemiology
The prevalence of acute myocarditis is difficult to estimate, because frequently the disease goes undiagnosed, is mild, or is masked by disease in another organ system or the animal recovers spontaneously. Similarly, it is difficult to assess the clinical significance of postmortem findings of myocardial inflammation and fibrosis. The morbidity of myocarditis in large animals probably is underestimated because it is rarely the cause of mortality and is associated with viral, bacterial, and parasitic infections that manifest themselves without specific signs of cardiovascular disease.
Inherited cardiomyopathy has been reported in cattle in Japan, Australia, the Netherlands, Canada, and Switzerland, with the incidence in inbred populations reaching 3% to 5%.137 A familial form of dilated cardiomyopathy has also been reported in cattle in Sweden, Denmark, Australia, and the United Kingdom.137 These cattle may be genetically linked by the presence of the red gene in Holstein-Friesian cattle.136 Although in some cattle inherited cardiomyopathy resulted in death by 6 months of age,138,140 other cattle failed to show clinical signs until 2 to 6 years of age.134-135,137,139 No sex predilection is recognized in inherited cardiomyopathy of cattle.
The morbidity and mortality from cardiomyopathy stemming from other causes in cattle and horses is not known. From the number of clinical reports, inherited cardiomyopathy seems to be more prevalent in cattle than in horses. Idiopathic dilated cardiomyopathy or cardiomyopathy secondary to viral or bacterial infections may be more common in horses and is a common cause for the acute onset of CHF in horses of racing age.
Necropsy Findings
There may be no gross lesions associated with myocarditis. Depending on the relationship between death and the occurrence of myocarditis, microscopic examination of the myocardium may show no inflammatory cells but increased fibrous tissue in the interstitium, or there may be foci of inflammatory cells (typically mononuclear cells), variation in the cross-sectional area of the cardiac myocyte, degeneration of adjacent myocardial fibers, myocardial necrosis, and fibrosis. Focal interstitial nephritis and fibrosis were reported in eight of nine Holstein-Friesian cattle in Scotland with dilated cardiomyopathy.137 Often the inciting cause of myocardial necrosis in horses is unknown.152 Samples of gastric or ruminal contents should be obtained and submitted for analysis of ionophores if ionophore toxicosis is suspected. Feed samples should also be obtained and submitted for ionophore analysis in these cases.
The lesions of cardiomyopathy are recognized more easily grossly as enlargement of the heart, which may be rounded to globose. Biatrial, biventricular, and pulmonary artery dilation is usually present. Patchy or uniform streaks of myocardial pallor may be detected. In rare cases, one of the chambers or major vessels (usually pulmonary artery) may rupture. Generalized edema is frequently found as ascites (uncommon in horses), pericardial or pleural effusion, and edema of the mesentery and subcutaneous tissues. There may be evidence of vascular congestion of the liver, lungs, and spleen. The kidneys may be pale and swollen and have an irregular, granular, pitted surface.
Microscopically the lesions of cardiomyopathy are characterized as myocardial vacuolation and degeneration with necrosis and fibrosis. Calcification may be associated with these lesions. Occasionally there is increased vascularization with proliferative regeneration of myofibers. In some cases, no histopathologic abnormalities are found.
Treatment and Prognosis
The treatment of myocarditis includes treatment of the underlying causative agent if it is recognized and control of the complications such as arrhythmias, CHF, or shock. Thromboembolism is rarely a complication recognized in animals. Prompt administration of vitamin E may be beneficial to the survivors in cases of ionophore toxicosis. Performance animals should be rested. Corticosteroids may be beneficial in animals with severe toxemia, complicated arrhythmias, or intractable heart failure, but their use in early cases of myocarditis or in infections suspected of having a viral cause is controversial because viral recrudescence may occur. However, in many cases the corticosteroids appear to have a beneficial effect. Prognosis is good if there are no signs of heart failure and if cardiac arrhythmias are managed successfully. Prognosis is guarded to poor if signs of CHF are present.
Therapeutic strategies currently used for treatment of dilated cardiomyopathy include positive inotropic agents (digoxin), diuretics, vasodilators, rest, and in some cases removal of pleural or abdominal fluid. The advantages of vasodilators (venodilators to relieve pulmonary edema and arterial dilators to reduce preload and afterload and improve contractility) in the treatment of cardiomyopathy are recognized in humans and small animals,156 but these drugs have not been widely used in large animals because of the lack of pharmacokinetic studies and clinical trials.
Digoxin is the positive inotropic agent used almost exclusively in large animals but is contraindicated in animals with acute monensin toxicosis. A priming or loading dose can be used in horses with acute severe CHF, followed with a maintenance dose, or, more frequently, the horse is started on a maintenance dose from the onset of therapy. In horses the drug can be administered PO or parenterally. In cattle, low bioavailability limits its use to the intravenous route of administration.157 Guidelines for the use of digoxin are given in Box 30-1.158-160 In general, maintenance dosages are used because of the lowered risk of adverse reactions. A peak and trough serum digoxin concentration should be obtained 3 to 5 days after digoxin therapy is begun, and the dosage should then be adjusted accordingly. Ideally the peak and trough digoxin concentration should be 1 to 2 ng/mL.
Box 30-1 Guidelines for Digoxin Therapy in Horses and Cattle
| HORSES | CATTLE |
|---|---|
| Priming dose IV: 12 to14 μg/kg | Priming dose IV: 22 μg/kg |
| Priming dose PO: 34 (elixir)to 70 (powdered tablets in suspension) μg/kg | Maintenance dosage IV: Infusion of 0.86 μg/kg/hr or give 11 μg/kg three times daily |
| Maintenance dosage IV: 2.2 μg/kg every 12 hours (most commonly used) or 6 to 7 μg/kg/24 hr | |
| Maintenance dosage PO: 11 μg/kg every 12 hours (most commonly used) | |
| or 17 (elixir) to 35 (powdered tablets in suspension) μg/kg/24 hr |
Dehydration, acid-base imbalance, and electrolyte abnormalities should be corrected before digoxin therapy. The dose of digoxin should be decreased in animals with elevated creatinine or blood urea nitrogen (BUN) until these values return to normal. In horses the intravenous loading dose of digoxin should be administered slowly or divided and given at a rate of one third of the loading dose hourly until completion.158 Alternatively, therapy can be initiated with the maintenance dose. Close monitoring of body weight, appetite, electrolyte concentrations, creatinine or BUN concentration, and cardiac rhythm is essential during therapy. Therapeutic drug monitoring with digoxin plasma concentrations can be helpful during initial therapy, when the volume of distribution of the drug and the patient’s body weight may be in a state of flux. Samples for determination of peak serum digoxin concentrations should be obtained 1 to 2 hours after digoxin administration and should not exceed 2.5 ng/mL.160,161
The diuretic used most commonly in large animals is furosemide. It is administered parenterally at the rate of 0.5 to 1 mg/kg twice daily or as needed to control edema. Oral absorption of furosemide is poor or variable in horses.162 A continuous rate infusion of furosemide is recommended in horses when rapid profound diuresis is needed.163 The half-life and diuretic effect of bumetanide, a sulfonamide diuretic, are shorter than those of furosemide in horses.164 Electrolyte concentrations and water consumption should be monitored closely in patients receiving diuretics.
The vasodilators used most frequently in horses include hydralazine and enalapril, an ACE inhibitor. Both have resulted in clinical improvement in animals treated for CHF. Hydralazine is administered PO at a dose of 0.5 to 1.5 mg/kg twice daily. Enalapril has been used at a dose of 0.5 mg/kg PO twice daily in the management of horses with moderate to severe mitral or aortic regurgitation and in the treatment of horses with CHF. However, after a single 0.5-mg/kg oral dose of enalapril, neither enalapril or enalaprilat were detectable in the serum.165 Ramipril was reported to result in clinical and echocardiographic improvement of a horse with CHF.166 Quinipril at a dosage of 120 mg/horse/day has also been shown to increase stroke volume and cardiac output in horses with mitral regurgitation after 8 weeks of therapy.167
Control of cardiac arrhythmias should be attempted in animals that are hemodynamically unstable or threatened with the development of worsening arrhythmias. Quinidine is usually the drug of choice for control of atrial and ventricular arrhythmias in cattle and is one of the drugs of choice in horses. Procainamide, lidocaine and propafenone have also been successful in the treatment of ventricular arrhythmias in horses (Table 30-3). Although administration is limited to the intravenous route in cattle, oral or intravenous administration in horses results in adequate plasma concentrations to control arrhythmias. Quinidine is administered IV by infusion or by divided bolus injections as shown in Box 30-2.
Table 30-3 Drug Therapy for Ventricular Tachycardia
| Drug | Usual Dose |
|---|---|
| Bretylium tosylate | 0.5 mg/kg IV for life-threatening ventricular tachycardia or ventricular fibrillation |
| Dexamethasone | 0.05–0.22 mg/kg, IV or IM |
| Furosemide | 1-2 mg/kg as needed for pulmonary edema |
| Lidocaine | Equine: 0.1-0.25 mg/kg as a bolus; repeat up to total of 0.5 mg/kg in 10–15 min |
| Bovine: 0.5 mg/kg, slowly IV; can repeat in 15 min | |
| Magnesium SO4 | IV infusion at 1 g/min, to effect, up to a maximum of 25 g |
| Procainamide | 1 mg/kg/min, IV to a maximum of 20 mg/kg |
| Propafenone | 2 mg/kg PO tid |
| 0.5-1 mg/kg in 5% dextrose slowly IV over 5-8 min for refractory sustained ventricular tachycardia (not available in United States) | |
| Propranolol | 0.03 mg/kg, IV |
| Quinidine gluconate (IV) | 1-10 mg/kg, IV; total dose in 0.25- to 0.5-mg/kg boluses 5-10 min apart |
IM, Intramuscular; IV, intravenous; PO, by mouth; tid, three times per day.
Box 30-2 Quinidine Administration in Horses and Cattle
| HORSES | CATTLE |
| IV: 1.5 to 2 mg/kg every 10–20 min until conversion or desired effect but not to exceed a total dose of 12 mg/kg | IV: 48 mg/kg infused IV over a 4-hr period |
| PO: 22 mg/kg (1 g/100 lb of body weight) q2h until conversion or desired effect; not to exceed a total dose of 132 mg/kg q2h; if quinidine plasma concentrations cannot be obtained promptly, a total dose of 88 mg/kg q2h should not be exceeded |
When quinidine is used IV, concurrent intravenous administration of a balanced electrolyte solution at the rate of 3 to 4 mL/kg/hr is desirable to maintain blood pressure in animals with a severely compromised cardiovascular status.
Prognosis for animals with dilated cardiomyopathy is poor. Echocardiography is useful in determining the severity of the myocardial dysfunction and in formulating a prognosis. The results of the initial echocardiographic examination were the best prognostic indicator of survival in a recent outbreak of monensin toxicosis.144 Recovery of animals with cardiomyopathy is unusual, constant therapy is required for maintenance of animals with CHF, and sudden death can occur at any time. Some animals may have significant myocardial dysfunction without CHF and are “cardiac cripples,” comfortable at pasture but not safe to use for performance. These animals may later develop CHF.
Prevention and Control
Maintenance of a good vaccination program may limit the bacterial and viral causes of myocarditis. Parasite control may also reduce myocardial injury that predisposes to myocarditis or cardiomyopathy in horses. Toxic myocardial diseases are prevented by proper mixing of feeds containing monensin, salinomycin, or lasalocid in feed mills in which horse feed is not made, by not shipping bulk horse feed in trucks in which medicated ionophore-containing feed has been transported, and by preventing horses from ingesting these medicated feeds. Gossypol toxicity can be prevented by feeding no cottonseed meal or cottonseed meal with low gossypol content, especially when feeding preruminants.168 Feeding of cottonseed meal can be limited by providing more forage or additional protein sources in the diet of ruminants so that ingestion of gossypol is limited to concentrations of 1 to 2 g/kg or less of feed for adult cattle and 0.5 to 1 g/kg or less of feed for immature cattle.169 Nutritional myocardial disease can be prevented by adequate feeding of vitamin E, selenium, and copper with supplementation as required. Inherited cardiomyopathy is controlled by avoiding breeding of known or suspected carriers of the polled Hereford and Holstein-Friesian breeds.
PERICARDITIS
Definition and Etiology
Pericarditis is inflammation of the pericardium that results in the accumulation of fluid or exudate between the visceral and parietal pericardium. Pericarditis in large animals can be caused by trauma from penetration of ingested foreign objects or external wounds, hematogenous spread (septicemia) of infection, extension of infection from the lung or pleura, viral infections such as equine viral arteritis or equine influenza, and neoplasia. Idiopathic pericarditis, characterized by an aseptic inflammatory exudate, is not uncommon in horses and has recently been reported in cattle.170-176 Autoimmune, hereditary, and metabolic causes of pericarditis have not been documented in large animals.
Clinical Signs and Differential Diagnosis
The clinical presentation of pericarditis can vary, depending on the volume and rate of development of the pericardial effusion and the cause. Nonspecific clinical signs—fever, anorexia, depression, or weight loss—may be the chief complaints, but, more frequently, peripheral edema, jugular venous distention and pulsations, tachypnea, or dyspnea is the presenting clinical sign. Cattle may exhibit pain by an abnormal stance characterized by abducted elbows, a spontaneous or induced expiratory grunt, reluctance to move, or preference to stand with the forequarters elevated. Horses may show signs of colic or syncopal episodes.
The most consistent findings on auscultation are tachycardia, muffling of heart sounds, and absence of lung sounds in the ventral thorax. Dorsally the lung sounds are louder than normal. These findings are in contrast to pleuritis, an important diagnostic ruleout for pericarditis, in which the lung sounds are muffled ventrally but the heart sounds are not and radiation of the heart sounds occurs over a wider area than normal.177 In cattle, splashing sounds frequently are audible in the cardiac auscultation area, a sound some refer to as a “washing machine murmur.” This is attributed to the accumulation of gas and fluid in the pericardium and is indicative of the presence of gas-forming (anaerobic) organisms and a grave prognosis. In cattle this murmur distinguishes pericarditis from cardiac neoplasia or other causes of CHF. The splashing sounds are absent in horses, and muffled heart sounds are the rule, but pericardial friction rubs may be auscultated, especially after pericardial fluid accumulation has been relieved.172 A gallop rhythm may be auscultated in either horses or cattle.
Mucous membranes may be congested and have a prolonged capillary refill time. Jugular venous distention and pulsations usually are present. Arterial pulses are weak. The latter findings may help distinguish pericarditis from primary pleuritis. Percussion of the thorax reveals ventral dullness, and pleural effusion is frequently present in horses with pericarditis. A concurrent pleuropneumonia or septic pleuritis is present in some horses.174,178-181 Ascites is infrequently detected in horses with pericarditis, but concurrent peritonitis has been reported.182
Clinical Pathology
The changes shown in a complete blood count are not specific and depend on the cause of the pericarditis. There may be hemoconcentration if the animal is dehydrated or toxemic, or there may be a mild anemia associated with chronic infectious pericarditis. The WBC count may be normal or increased. There may be an absolute neutrophilia or a lymphopenia. Frequently the fibrinogen concentration is elevated. The serum chemistries are usually normal except for albumin concentration, which may be low171-172 and accompanied by an elevation in globulin concentration. Liver enzyme, bilirubin, serum urea nitrogen, and creatinine concentrations are frequently mildly elevated, consistent with the development of CHF. Laboratory evidence of dehydration (increased PCV, total protein, serum urea nitrogen, and creatinine concentrations) may be exaggerated when diuretics have been administered previously. Although total serum LDH concentration may be elevated, more specific information may come from finding an increase in the cTnI or the myocardial isoenzymes CK and LDH. An elevated cTnI was reported in one cow with hemorrhagic pericarditis.175 Electrolyte concentrations are frequently normal, but serum calcium and potassium concentrations may be low because of anorexia. Decreased sodium and chloride concentrations have been reported in horses with pericarditis.172 No consistent abnormalities in arterial or venous blood gas concentrations have been reported in large animals with pericarditis.
Radiography is not a sensitive diagnostic test for pericarditis in horses, although an enlarged rounded cardiac silhouette is detected with large pericardial effusions. In cattle, traumatic pericarditis frequently results in fluid and gas accumulation in the pericardium that is detectable radiographically and is relatively specific for this disease. A metallic foreign body is usually detected radiographically in the cranial reticulum or caudal thorax in cattle with traumatic pericarditis (Fig. 30-18). Radiographic changes may not be detected in early or uncomplicated pericarditis and, if fluid accumulation is large and there is concurrent pleural effusion, are indistinguishable from those associated with pleuritis. An enlarged, rounded cardiac silhouette is not specific for pericarditis and can be seen with other causes of generalized cardiomegaly. An obscured cardiac silhouette, vena cava, and diaphragm with dorsal displacement of the trachea may be seen with pericarditis or pleuritis. The lungs, which may be aerated only dorsally, frequently have interstitial infiltrates.
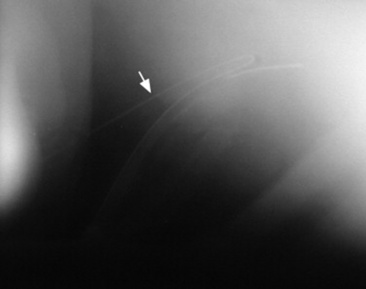
Fig. 30-18 Lateral radiograph of the caudal thorax and cranial reticulum showing a wire (arrow) in a cow with traumatic reticuloperitonitis.
The ECG changes most commonly associated with pericarditis in large animals are decreased amplitude of the QRS complexes (less than 1.5 mV in the base-apex lead)172,173,182 (Fig. 30-19), electrical alternans (altered configuration of the P, QRS, or T complexes on a regular basis), and ST-segment elevation or slurring.171,172,178,182,183 A right-axis deviation in the standard limb leads may be apparent.171,183 Cardiac arrhythmias (usually atrial or ventricular premature beats) may be detected in affected animals, suggesting a concurrent myocarditis (Fig. 30-20). In some animals, no ECG changes are evident.184
Fig. 30-19 Base-apex lead ECG taken from a cow before (upper strip) and after (lower strip) removal of 2 L of pericardial fluid. The markedly decreased amplitude (0.6 mV) of the QRS complexes is improved (1.2 mV) after pericardiocentesis.
Fig. 30-20 ECG (rhythm strip) obtained from a horse with pericarditis and junctional tachycardia. Note the P wave at the same place in the QT interval.
Echocardiography reveals findings specific for pericarditis and is used to confirm the diagnosis noninvasively. An echo-free space between the visceral and parietal pericardium is evident (Fig. 30-21) and apparent even with a minimum amount of fluid accumulation.177,178 Frequently, hypoechoic to echoic strands that correspond to fibrin are imaged in the pericardial space with a hypoechoic to echoic layer covering the epicardial surface of the heart in large animals with pericarditis. The pericardial effusion is often anechoic to hypoechoic, but more echoic pericardial fluid is occasionally seen, consistent with a more exudative fluid. Bright hyperechoic pinpoint echoes representing free gas are often imaged in cattle with pericarditis. Gas in the pericardial sac of cattle with splashy heart sounds may limit the ability to obtain an echocardiographic evaluation of all cardiac structures. The visualization of intrapericardial gas is a sensitive indicator of anaerobic pericarditis. Pericardial effusion is differentiated from pleural effusion by finding the echo-free space surrounding the right ventricle and left ventricular free wall, but pericardial effusion is rarely imaged behind the left atrium.185,186 Right ventricular diastolic collapse and right atrial collapse are common findings with large pericardial effusions and are an indication of the development of cardiac tamponade and hemodynamically significant pericardial effusion.173 Other echocardiographic findings consistent with pericarditis are decreased left ventricular chamber dimension, decreased left ventricular free wall motion, and decreased parietal pericardial motion.173,174,178,187 Increased (see Fig. 30-21) or paradoxic motion (cranial motion beginning before the QRS complex) of the interventricular septum may be apparent.186,187
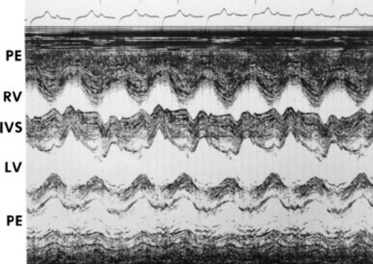
Fig. 30-21 M-mode echocardiogram obtained from a cow with traumatic pericarditis. There is a separation between the visceral and parietal pericardium cranial to the right ventricle (RV) and caudal to the left ventricle. The cranial pericardial effusion (PE) is echogenic, suggesting that fibrin or debris is present. The left ventricular (LV) dimension is reduced, and there is marked cranial motion of the interventricular septum (IVS) just before the QRS complex.
The site for pericardiocentesis should be selected echocardiographically. Pericardial fluid is obtained most commonly from the left fifth intercostal space 2.5 to 10 cm dorsal to the olecranon, above the level of the lateral thoracic vein, the safest site for pericardiocentesis. When there is a large volume of pericardial effusion, pericardiocentesis can be productive from the right at approximately the same location. If at all possible, a pericardial catheter (large-bore Argyle tube) should be inserted into the pericardial space at the time the initial pericardiocentesis is performed to enable repeated drainage and lavage of the pericardial sac (Fig. 30-22). Fluid analysis and bacterial and viral culture results depend on the cause. In cattle with traumatic pericarditis, fluid analysis usually reveals elevated protein concentration (>3.5 g/dL) and an elevated WBC count (>2500/μL), composed primarily of neutrophils. The fluid is urine colored to slightly blood tinged and foamy, and it has a foul odor. A mixed population of gram-positive and gram-negative aerobic and anaerobic bacteria (gastrointestinal flora) usually are present. Protozoa may be found in unusual circumstances. Hemorrhagic effusion has been reported in several cows with idiopathic pericarditis that responded favorably to pericardial drainage (usually with lavage) along with systemic antimicrobial therapy.175 In horses, protein concentration of the pericardial fluid is elevated (≥2.5 g/dL) and the WBC count is normal or elevated with a population predominantly of neutrophils, although red blood cells, lymphocytes, eosinophils, mesothelial cells, and histiocytes have been observed.171-173,178,182 Bacterial (aerobic and anaerobic) cultures and viral cultures may be negative. Actinobacillus organisms were frequently isolated from mares with pericarditis in association with the mare reproductive loss syndrome.188 Paired serum may support a viral cause. Analysis of pleural and peritoneal fluid (when present) usually reveals fluid characterized as a modified transudate or that is mildly inflammatory.

Fig. 30-22 Insertion of a pericardial chest tube into the left fifth intercostal space for drainage and lavage of the pericardial space. Note that the left foreleg is being pulled forward, the clinician is inserting the large-bore Argyle chest tube into the left fifth intercostal space above the lateral thoracic vein, and the cow is wearing a radiotelemetry ECG system underneath the surcingle.
Cardiac catheterization demonstrates an elevation in central venous or right atrial pressure, and the atrial and ventricular pressure curve may be abnormal in appearance. Right atrial, right ventricular, and pulmonary artery end-diastolic pressures may equilibrate.183 In combination these findings are relatively specific for pericarditis.
Pathophysiology
The accumulation of fluid in the pericardium occurs as a result of inflammation. The rate of fluid accumulation and the degree to which the pericardial pressure increases determine the pathophysiologic consequences. Generally pericarditis results in decreased distensibility (increased ventricular end-diastolic pressure) of the heart, which impairs the ability of the heart to fill during diastole. The elevation in end-diastolic pressure and impairment of ventricular filling elevate atrial pressure and reduce venous flow or venous return to the heart and diastolic perfusion of the myocardium. The result is a depression of ventricular contractility, stroke volume, and consequently cardiac output. In addition, arterial pressure and renal blood flow are decreased. Initially, compensatory mechanisms consisting of vasoconstriction, increased heart rate, and sodium retention (increased vascular volume) may maintain cardiac output. Failure to maintain cardiac output results in circulatory collapse.
Pericarditis can be classified as primarily effusive, constrictive, or a combination of both.183 The hemodynamic consequences of effusive pericarditis are primarily caused by the physical presence of pericardial fluid, whereas constrictive pericarditis is classified as such because the reduction in ventricular compliance is caused by fibrinous or fibrotic involvement of the pericardium and epicardium. Removal of pericardial fluid results in improved cardiac performance in effusive pericarditis but is of limited usefulness in constrictive pericarditis.
Epidemiology
Pericarditis is uncommon in horses; when it does occur, it is most frequently idiopathic and can occur in a horse of any age. History of a recent respiratory tract infection is not uncommon. Exposure of horses to Eastern tent caterpillars was the greatest risk factor for the development of fibrinous pericarditis during the epidemic of mare reproductive loss syndrome.189 Traumatic pericarditis has been reported in horses but is rare.190 Traumatic pericarditis is not uncommon in cattle, but it occurs in less than 10% of cattle with traumatic reticuloperitonitis.191 Most cattle are affected in late gestation or at parturition. Idiopathic pericarditis is rare in cattle.
Necropsy Findings
Gross postmortem examination shows distention of the pericardial sac with serosanguineous or urine-colored fluid that is foamy and may be malodorous (cattle). There may be organization of fibrinous exudate and fibrosis that is also evident on the epicardium (Fig. 30-23) and may infiltrate the myocardium. Pleural effusion may be present. Other signs of CHF such as pulmonary congestion, pulmonary edema, and chronic passive congestion of the liver may be present. If the cause is traumatic, the offending object may be well contained in a fibrous tract located between the reticulum and the pericardium.
Fig. 30-23 Postmortem photograph of a cow with constrictive pericarditis. The pericardium is opened and reflected, revealing the epicardium (arrow) covered in fibrinopurulent exudate.
Histopathologic examination reveals pericardial, epicardial, and occasionally myocardial fibrosis and inflammation with neutrophilic, lymphocytic, eosinophilic, or plasma cell infiltrates. Bacteria may be visible. The liver may show diffuse, centrilobular necrosis, fatty change, dilation and congestion of sinusoids, and perivenous fibrosis.
Treatment and Prognosis
Treatment of traumatic pericarditis in cattle is unrewarding and usually is addressed toward salvage or short-term survival to calving. Repeated pericardial drainage by means of pericardiocentesis or a fifth rib resection, lavage, or pericardiectomy may be useful for short-term survival, but the prognosis for return to normal function is poor, because CHF results from involvement not only of the pericardium but also of the epicardium and myocardium (see Fig. 30-23). Thoracotomy by a split-rib technique followed by pericardiectomy has been effective in treating some cattle with traumatic, restrictive pericarditis.192
Treatment of pericarditis not caused by trauma has been successful in horses, but the initial prognosis should be guarded. Aggressive treatment of horses with moderate to large pericardial effusions should include the placement of a large-bore indwelling chest tube into the pericardial sac under echocardiographic guidance and drainage and lavage of the pericardial sac, with local infusion of antibiotics, in addition to the initial broad spectrum antimicrobial therapy. Obtaining a sample for cytology and culture and sensitivity testing via a pericardiocentesis should be postponed until an indwelling catheter can be safely inserted because it is difficult to insert the catheter once the pericardiocentesis has been performed. The pericardial drainage and lavage, performed once or twice daily as needed, has been very effective in treating idiopathic or septic pericarditis in horses.173,174,178,182 The indwelling tube should remain in situ until the fluid recovered at the time of drainage is consistently less than or equal to the volume instilled in the pericardial sac with antimicrobials 12 to 24 hours earlier. The ECG should be monitored, because occasionally cardiac arrhythmias occur during the therapeutic procedure or during a routine pericardiocentesis. All horses with pericarditis should initially be treated for septic pericarditis with systemic broad-spectrum bacteriocidal antimicrobial drugs. The antimicrobial choice should be based on the most likely causative agents and modified as needed by the results of culture and sensitivity testing. Fibrinous pericarditis has been successfully treated in horses with broad-spectrum bacteriocidal antimicrobials when the amount of pericardial fluid detected echocardiographically was too small to safely obtain a sample for cytology and culture and sensitivity testing. Treatment of idiopathic pericarditis with corticosteroids should be initiated only after the results of the cytology and culture and sensitivity testing demonstrate no evidence of sepsis. Pericardiotomy or preferably pericardiectomy is the treatment of choice if signs of restrictive (constrictive) pericarditis are present. The surgical procedure is prolonged, expensive, and of considerable risk. A partial pericardiectomy was performed in one horse with constrictive pericarditis but was only transiently successful.179
In traumatic pericarditis the antibiotic selected must be capable of covering gram-positive and gram-negative aerobic and anaerobic bacteria. Nonsteroidal antiinflammatory drugs have been deemed useful as adjunctive therapy, as have corticosteroids if bacterial culture of the fluid is negative and if no evidence of sepsis is detected cytologically.
Although diuretics are effective in eliminating the severity of peripheral edema, they further reduce venous return and preload in animals with pericarditis. The result is further compromise of cardiac output and worsening of heart failure.
Prevention and Control
Traumatic pericarditis in cattle can be prevented by routine administration of magnets to heifers at the time of pregnancy diagnosis. At each subsequent pregnancy diagnosis, the cattle should be checked for the presence of the magnet in the reticulum. It is not beneficial to have more than one magnet present at one time.
Some types of infectious pericarditis in horses may be controlled by routine vaccination against the common respiratory pathogens.