Chapter 25 Alterations in the Leukogram
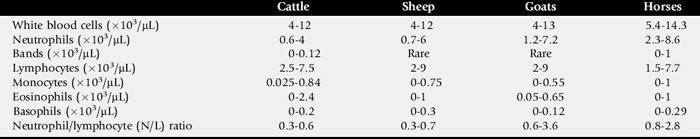
The leukogram (white blood cell [WBC] count, differential analysis, and WBC morphology) provides extremely useful laboratory data when considered with the history, clinical signs, and physical findings. To fully use the leukogram, the practitioner must know the types of WBCs, their kinetics and functions, and the pathologic and physiologic conditions that can cause deviations from normal. Normal values for leukogram data are given in Table 25-1.
Mature WBCs include neutrophils, lymphocytes, monocytes, eosinophils, and basophils. Immature leukocytes that may or may not be present in the peripheral blood include band or nonsegmented neutrophils, metamyelocytes, myelocytes, and progranulocytes. These immature forms usually are found only in the bone marrow, but they may be released in response to disease.
LEUKOCYTES
Leukocytes, or WBCs, are divided into two main categories: polymorphonuclear (PMN) leukocytes (granulocytes) and mononuclear leukocytes. PMN leukocytes include neutrophils, eosinophils, and basophils, which are produced in the bone marrow. The mononuclear leukocytes are the lymphocytes and monocytes. Lymphocytes are produced in the bone marrow (the primary source), lymphoid organs (thymus, spleen, and lymph nodes), and gut-associated lymphoid tissues (Peyer’s patches, tonsils). Monocytes, the largest of the WBCs, originate in the bone marrow.
Neutrophils
Neutrophils develop in the bone marrow as myeloblasts, progranulocytes or promyelocytes, myelocytes, metamyelocytes, band cells, and segmented neutrophils. Myeloblasts arise from bipotential committed stem cells (colony-forming unit—granulocyte, macrophage) that are derived from pluripotent hematopoietic stem cells.1
Myeloblasts, promyelocytes, and myelocytes are capable of cell division, but metamyelocytes, bands, and segmented neutrophils (about 80% of the bone marrow granulocytes) do not divide.2 The normal interval for progression from myeloblast to mature blood neutrophil is 4 to 9 days, depending on the species. A functional storage compartment of neutrophils exists in the marrow to prevent marrow depletion by the sudden imposition of a greatly increased peripheral use. The storage pool, which is limited to segmented neutrophils and some bands, varies in size among species and is relatively small in adult cattle.1 A marginal pool of neutrophils adheres to endothelium throughout the microvasculature. Neutrophils in the circulating pool are the only part of the total neutrophil population enumerated by the peripheral blood neutrophil count. In large animals the marginal neutrophil pool is approximately equal in size to the circulating pool.1
After entering the bloodstream, neutrophils have a half-life of 6 to 14 hours, depending on the species. The entire blood pool of neutrophils is replaced two to two-and-one-half times per day. Neutrophils move randomly into the tissues by diapedesis through vascular endothelium and do not return to the blood. Neutrophils migrate into tissues within 2 hours of injury, infection, or inflammation. In the absence of such lesions, the neutrophils are destroyed by macrophages of the bone marrow, liver, and spleen or are lost into body secretions and excretions within 96 hours of exiting the marrow.2
The major function of neutrophils is to phagocytize and destroy foreign material, especially pathogenic bacteria. Bacterial products and substances released from activated lymphocytes (lymphokines), macrophages, and cellular damage are responsible for attracting the neutrophils, which move by chemotaxis and bind to the foreign particle.3 Opsonins are protein components of serum that adhere to foreign particles and make them more liable to phagocytosis.4 Opsonization by specific antibodies increases the rate and magnitude of ingestion for most bacterial organisms. Once drawn into the cell, the bacteria are enclosed in a phagocytic vacuole, which fuses with lysosomes in the neutrophil cytoplasm that release hydrolytic enzymes to destroy the contents. Degranulation, which occurs within 5 seconds of a phagocytic event, can lead to release of lysosomal enzymes into the surrounding milieu and cause tissue destruction.
Gram-negative bacteria are more resistant to digestion in the neutrophil than gram-positive bacteria because of the structure of their outer wall. Brucella abortus, Mycobacterium paratuberculosis, Listeria monocytogenes, and some Salmonella serotypes are extremely resistant to lysosomal destruction and may survive and multiply within the neutrophil.
EOSINOPHILS
Eosinophils are produced in the bone marrow and follow the same maturation sequence and kinetics as neutrophils, except that eosinophils arise from the colony-forming unit—eosinophil.1 There is a large marrow reserve of eosinophils, and their circulatory half-life varies from 30 minutes to 10 hours, depending on the species. Eosinophils also are found in many body tissues, particularly the gut, subcutis, uterus, and respiratory tract, where they have a half-life of 12 days.
Eosinophils are most important in controlling parasitic infections and in regulating inflammatory and allergic reactions. Eosinophils are less efficient phagocytes than neutrophils and provide little host resistance to bacterial infection but are important in protective parasitic immunity.2 Eosinophils are believed to regulate immediate (type I) hypersensitivity and inflammatory responses by inactivating histamine, leukotrienes, platelet-activating factor, and other chemical mediators involved in these processes. Other, less characterized effects of eosinophils include tissue damage, augmentation of coagulation and fibrinolysis, and inhibition of granulopoiesis.
BASOPHILS
Basophils are relatively scarce in the blood of large animal species. The blood basophil resembles the tissue mast cell and is believed to perform similar functions. Basophils are produced in the bone marrow by mitosis of basophilic promonocytes through the same sequential stages of maturation as neutrophils. Basophils have a mean circulatory half-life of about 6 hours. They then enter tissues, where they survive for approximately 10 to 12 days.1 Mast cells, found in many body tissues, originate from undifferentiated connective tissue mesenchymal cells, especially near blood vessels. There is evidence in some species that mast cells also originate from a precursor in the bone marrow.1
Basophils and mast cells contain stored intragranular substances that mediate their function in allergic and inflammatory processes. The most important function of basophils and mast cells is to elicit an immediate hypersensitivity reaction through secretion of vasoactive mediators, including histamine, leukotrienes, platelet-activating factor, and others. Vasodilation, pooling of fluid in tissue spaces, and systemic reactions may occur, with signs of dyspnea, urticaria, coughing, and even severe anaphylactic shock. Eosinophils are attracted to these areas to neutralize the histamine and attenuate the inflammatory response.
Lymphocytes
Lymphocytes are produced in the bone marrow, lymph nodes, thymus, spleen, and Peyer’s patches. They are classically divided into two groups: T cells, or thymus-derived lymphocytes, and B cells, or bone marrow—derived lymphocytes. Null cells have been identified in humans and in some animals that lack specific markers for T or B cells.1 Both T- and B-cell populations are present in the peripheral blood of large animal species, with T cells making up the majority. B cells are few during fetal life but steadily increase to make up about 20% of circulating lymphocytes in most adult domestic animals.1 Observations in cattle indicate that various inflammatory states are associated with an increase in B cells and a decrease in T cells. Leukemic cows infected with bovine leukosis virus (BLV) and those with persistent lymphocytosis have a higher number of B cells, which constitute up to 97% of lymphocytes.1 Lymphocytes can recirculate and continue to undergo mitosis. Information on domestic animals is unavailable, but most blood lymphocytes in humans have an average lifespan of 4.3 years.1
B lymphocytes transform into plasma cells that produce antibodies under the regulation of T lymphocytes. T lymphocytes are primarily responsible for delayed-type hypersensitivity (DTH), graft and tumor rejection, autoimmunity, and resistance to certain intracellular pathogens. Some types of T cells have a direct cytotoxic function to destroy foreign cells, and null cells express natural killer activity. Lymphocytes produce important immunologic mediators called lymphokines, which include macrophage-activating factor, interferons, and interleukins.
The number of lymphocytes in the peripheral blood reflects a balance between cells leaving and entering the circulation; thus changes do not necessarily mean altered lymphopoiesis. In addition to consideration of the total number in the leukogram, lymphocytes can be evaluated by measuring the ratio of T cells to B cells (normal range is 1:1 to 3:1), antibody levels and response to vaccination, in vitro lymphocyte stimulation tests, and skin tests for DTH.
Monocytes
Monocytes are produced in the bone marrow from the colony-forming unit—granulocyte, a macrophage, which differentiates into either myeloblasts or monoblasts (precursors of monocytes).1 Monoblasts undergo mitosis to promonocytes, then divide one to two more times to produce monocytes. Once released into the blood, monocytes circulate for 1 to 3 days, then enter body cavities and tissues and transform into macrophages. Tissue macrophages survive in tissues for weeks to years. Once in tissue, these macrophages are described as “fixed” or “free.” Free macrophages are found in the peritoneal and pleural cavities, in joints, in alveolar spaces, and at areas of inflammation. Fixed macrophages include Kupffer’s cells of the liver, osteoclasts, microglial cells, and macrophages found in the spleen, bone marrow, and lymph nodes.
The blood monocytes and tissue macrophages constitute the mononuclear phagocyte (reticuloendothelial) system.1,2 The functions of tissue macrophages include sustained phagocytic activity to remove dead and damaged tissue; microbicidal action against some bacteria, viruses, fungi, and protozoa; regulation of the immune response in both afferent and efferent limbs; tumor defense; regulation of hematopoiesis; tissue repair and remodeling; and secretion of monokines, lysosomal enzymes, and other substances, such as coagulation factors, that have wide-ranging biologic importance. The tissue macrophages are better equipped than neutrophils to combat intracellular organisms and those that cause granulomatous inflammation such as fungal infections, listeriosis, brucellosis, Johne’s disease, tuberculosis, and salmonellosis.
Interleukin (IL)-1 and tumor necrosis factor are important macrophage-derived mediators of the inflammatory response.5 These and other cytokines are released when macrophages are exposed to bacterial products (especially endotoxin), antigens, and injured tissue. IL-1 stimulates bone marrow release of neutrophils and attracts them to areas of bacterial infection or inflammation. Tumor necrosis factor is responsible for many of the physiologic derangements associated with endotoxemia that end in shock, tissue injury, and death.
GENERAL PRINCIPLES OF LEUKOGRAM INTERPRETATION
When interpreting the leukogram, the practitioner must consider the established normal values for the species, the animal’s age and condition, and the species-specific WBC responses. Leukocytosis may be attributed to physiologic or pathologic causes, whereas leukopenia always is considered pathologic.1,6
Physiologic Leukocytosis
Physiologic leukocytosis occurs when epinephrine is released, as with stress, excitation, anxiety, or exercise. The elevated WBC count is transient and caused by both neutrophilia and lymphocytosis, although it is primarily the result of temporary mobilization of the marginal neutrophil pool.2 Corticosteroids, either exogenous or endogenous, cause neutrophilia and lymphopenia. Younger animals may have higher lymphocyte and total WBC counts than those considered normal for adults.1
Neutrophilia
Bacterial infection is the most common cause of pathologic neutrophilia (Boxes 25-1 and 25-2). In the acute stage of the infection, a left shift (presence of increased immature neutrophil granulocytes in the peripheral blood) may appear.1,6 Left shift with concomitant mature neutrophilia is called a regenerative left shift. Rebound neutrophilia often follows neutropenia associated with endotoxemia, and it is usually a good prognostic indicator. Neutrophilia is most pronounced while bacterial infections are being localized, especially with abscess formation. Chronic localized infections rarely are attended by a left shift, and in cattle the neutrophil count may increase only minimally or not at all. Horses with chronic bacterial infections often show only a mild mature neutrophilia attended by lymphopenia and a normal total WBC count.
Neutrophilia may also accompany inflammation caused by neoplasia or severe injury or may arise during the postoperative period. Less common causes of neutrophilia include parasitic and mycotic infections, tumors that cause secretion of endogenous corticosteroids, toxins, and some metabolic diseases such as pregnancy toxemia.
In large animals the total WBC or neutrophil count may not reflect chronic inflammation; for this reason, examination of neutrophil morphology is paramount in interpreting the leukogram. Bacterial infections, especially those caused by gram-negative organisms, often result in neutrophil cytoplasmic and nuclear alterations, which are referred to as “toxic changes.” These changes occur in the bone marrow and include cytoplasmic foaminess, vacuolation, and/or basophilia; reddish purple “toxic” granules; bluish cytoplasmic inclusions called Döhle bodies; and bizarre giant forms with or without polyploidy. The changes do not accompany other causes of neutrophilia.
Neutropenia
The usual causes of neutropenia in the large animal species are bacterial septicemia and endotoxemia caused by gastrointestinal disease, metritis, or coliform mastitis (Boxes 25-3 and 25-4). Some viral diseases and anaphylaxis also cause neutropenia.
A degenerative left shift, generally associated with neutropenia, occurs when immature neutrophils appear in the peripheral blood in greater numbers than mature neutrophils. In species other than cattle, degenerative left shift is an extremely poor prognostic sign. Because cattle have a small bone marrow reserve of neutrophils, immature neutrophils appear quickly in the blood during acute inflammatory diseases and often exceed the mature neutrophils. A marked fall in the WBC count commonly is seen in cattle during the developmental stage of an acute localizing inflammatory process such as mastitis or metritis. Once neutrophil production has intensified, the left shift disappears and mature neutrophilia intervenes. Neutropenia that persists longer than 4 days is a sign of inadequate granulopoiesis, which sometimes occurs subsequent to severe toxemia. Neutropenia apparently is rare in goats.
The severity of toxemia is reflected by the number of “toxic” neutrophils and the degree of toxic changes. In diseases that cause severe toxemia, precursor cells in the bone marrow become vacuolated and fail to divide, thereby contributing to the existing neutropenia. This bone marrow hypoplasia subsequent to severe infection and inflammation is seen most often in cattle.
In rare cases, neutropenia may develop subsequent to myelophthisic disease, idiopathic aplastic anemia, myelofibrosis, or bone marrow suppression by drugs, chemicals, or ionizing radiation. Lymphosarcoma may involve the bone marrow in rare cases.
Lymphocytosis
Pathologic lymphocytosis is uncommon, occurring occasionally with chronic viral infections and autoimmune disease processes (Boxes 25-5 and 25-6). Lymphocytic leukemia is rare in large animals. Thirty percent of cattle infected with BLV are leukemic (see Chapter 37), and lymphocytosis may persist in the absence of lymphoma or leukemia. Physiologic lymphocytosis associated with epinephrine release caused by excitement or exercise is common in horses under 2 years of age.
Lymphopenia
Causes of lymphopenia include acute viral diseases, endotoxin release, severe bacterial infection, septicemia, rickettsial diseases, malnutrition, tumors that cause increased release of corticosteroids, and immunodeficiency (Boxes 25-7 and 25-8). Persistent lymphopenia is a poor prognostic indicator. Increasing lymphocyte counts represent recovery.
Monocytosis
Monocytosis occurs with chronic inflammation (Box 25-9). Because the monocyte count is not highly responsive to inflammatory disease in large animals, it is not an especially useful part of the leukogram.6
Monocytopenia
Endotoxin release and viremia may cause monocytopenia. Monocytopenia occurs initially during stress periods associated with corticosteroid release and may be followed by monocytosis.
Eosinophilia
Eosinophilia is uncommon in large animals but may occur with diseases that involve an interaction between antigen, IgE antibody, and mast cells or basophils, such as parasitic infections, allergic respiratory diseases, and dermatoses (Boxes 25-10 and 25-11). Unlike in humans and small animals, visceral larval migrans rarely induces peripheral eosinophilia. Tissue protein breakdown (malignancies, chronic suppurative processes) may cause eosinophilia in rare cases through the release of histamine or eosinophilic chemotactic factor of anaphylaxis from mast cells. Histamine in the blood attracts bone marrow eosinophils to the circulation. For eosinophilia to occur in response to parasitism, a parasite protein must be released and processed by cells infiltrating the tissue site of parasitic lodgment.1 Thus eosinophilia is unlikely to accompany intestinal parasitism when the parasite is free-living in the lumen.
Eosinophilic granulocytic leukemia is rare in large animal species but has been reported in horses.7 The circulation of bizarre, immature eosinophils differentiates this from other causes of eosinophilia.
Eosinopenia
Eosinopenia is difficult to evaluate, because the leukograms of clinically normal animals may contain very few eosinophils. Eosinopenia may occur secondary to an increase in endogenous or exogenous corticosteroids. Eosinopenia also may be seen with active inflammatory processes.
Basophilia and Basopenia
Basophils are rarely seen in the peripheral blood of the large animal species, although they are more frequently encountered than in dogs and cats.1 Changes in the number of basophils are difficult to interpret. Stress causes a reduction in their number, whereas basophilia may be seen with allergic dermatitis and delayed hypersensitivity reactions.
APPROACH TO INTERPRETATION OF THE LEUKOGRAM IN HORSES
The equine neutrophil-to-leukocyte (N/L) ratio declines from approximately 2.8 at birth to 1.1 at 1 to 2 months of age and to 0.9 at 6 to 8 months. An N/L ratio near 1 persists through 2 years and increases with age to approximately 2 as lymphocyte numbers are reduced. The total WBC count increases from birth through 3 months of age, is slightly above adult values between 3 months and 2 years of age, and then starts to decline. Physiologic leukocytosis is quite common in horses under 2 years of age.1
During chronic and established inflammatory diseases, horses generally have a mature neutrophilia and lymphopenia that may or may not result in leukocytosis.6 The degree of leukocytosis in chronic suppurative diseases rarely exceeds 20,000/μL. Peracute diseases of the gastrointestinal tract and septicemia (especially in neonates), usually attended by endotoxemia, are characterized by leukopenia and a degenerative left shift, the severity of which is correlated with the prognosis. In the most severe cases the left shift includes myelocytes and neutrophils that show marked “toxic” changes. Survival and recovery are attended by a rebound neutrophilia (with or without left shift) and monocytosis. Neonates have a small neutrophil reserve and have more sluggish granulopoiesis in response to disease.
Lymphopenia and eosinopenia often occur readily in response to stress or corticosteroid administration. Chronic diseases commonly result in a reduction of the lymphocyte count, which may be under 1000/μL during severe systemic stress. The monocyte is not particularly responsive to disease in horses,1 but the blood monocyte generally decreases acutely and may increase above normal during chronic inflammatory diseases, especially those associated with tissue necrosis.
APPROACH TO INTERPRETATION OF THE LEUKOGRAM IN RUMINANTS
The general trend is for the WBC count to be higher in calves through 2 years of age and then decline with advancing age. Sheep and goat leukocyte counts increase through 2 to 3 months of age, then decline in adulthood to levels seen at birth. In cattle and sheep, neutrophils exceed lymphocytes at birth, but the ratio is reversed within the first week of life, and this ratio persists as a species characteristic. A reduction in the number of lymphocytes without similar changes in neutrophils produces an N/L ratio near unity in goats over 3 years of age.
Acute inflammatory disease and infection in cattle (e.g., neonatal septicemia, salmonellosis, enteritis, metritis, and coliform mastitis) cause a rapid drop in the WBC count because of migration of mature neutrophils to the site of inflammation, margination of neutrophils, and stress-induced loss of lymphocytes. The bone marrow has a small reserve of mature neutrophils, and immature forms (bands, metamyelocytes) are released into the circulation, creating a degenerative left shift during the first 2 to 3 days of acute inflammation. By the fourth day bone marrow granulopoiesis usually has increased sufficiently to meet the tissue demand for neutrophils, causing a normal leukocyte count with a left shift. If the inflammatory stimulus persists, a mature neutrophilia may develop. Generally the N/L ratio increases without leukocytosis. Severe systemic toxemia or chronic infections (or both) may cause granulopoietic depression and neutropenia.
Approximately 30% of cattle infected with BLV develop a benign persistent lymphocytosis, defined as an absolute blood lymphocyte count over 3 standard deviations above the normal mean for at least 3 months. Although these cells are BLV infected, cattle with lymphocytosis are clinically normal, and most do not develop enzootic bovine lymphosarcoma (EBL).8 Approximately 50% of cattle with EBL have a mild-to-moderate lymphocytosis, and leukemia is present in 10% to 30% of cases.1 Lymphocytosis, usually attended by neutrophilia, occurs sometimes as a result of chronic pyogenic conditions such as liver abscess, pericarditis, pulmonary abscess, and traumatic reticuloperitonitis.
The leukogram changes in sheep generally are similar to those of cattle. Parturition and adverse weather conditions induce typical corticosteroid-induced neutrophilia, lymphopenia, and eosinopenia.
Goats differ from cattle and sheep in that the leukogram typically has an equal or slightly greater number of neutrophils than lymphocytes.1 The total WBC count during inflammatory diseases may attain levels higher than 25,000/μL because of neutrophilia. A regenerative left shift is a common response to subacute or chronic inflammation. Leukopenia is rare.
An inherited syndrome characterized by marked neutrophilia and an increased susceptibility to bacterial infections has been recognized in Holstein-Friesian cattle under 2 years of age.9 Affected calves have a history of anorexia, weight loss, and failure to thrive, with signs of chronic intermittent pneumonia and diarrhea. Lymphadenopathy, periodontitis, and generalized dermatitis are also features of the syndrome. The neutrophils of these animals are dysfunctional because of a single mutation in CD18 that causes a lack of surface glycoproteins, called β2-integrins, that are important in cell adhesion processes.10 Calves that are heterozygous for the defect do not have dysfunctional leukocytes compared with those of clinically normal calves.11
1 Jain NC. Schalm’s veterinary hematology, ed 4. Philadelphia: Lea & Febiger, 1986.
2 Duncan JR, Prasse KW, Mahaffey EA. Veterinary laboratory medicine: clinical pathology, ed 3. Ames, Iowa: Iowa State University Press, 1994.
3 Becker EL. Chemotaxis. J Allergy Clin Immunol. 1980;66:97.
4 Roitt I, Brostoff J, Male D. Immunology. St Louis: Mosby, 1998.
5 Morris DD, Moore JN, Crowe N. Serum tumor necrosis factor activity in horses with colic attributable to gastrointestinal disease. Am J Vet Res. 1991;52:1565.
6 Morris DD. The haemolymphatic system. In: Higgins AJ, Wright IM, editors. The equine manual. London: Saunders, 1995.
7 Morris DD, Bloom JC, Roby KA, et al. Eosinophilic myeloproliferative disorder in a horse. J Am Vet Med Assoc. 1984;185:993.
8 Ferrer JR. Bovine lymphosarcoma. Adv Vet Med. 1980;24:1.
9 Gerardi AS. Bovine leukocyte adhesion deficiency: a review of a modern disease and its implications. Res Vet Sci. 1996;61:183.
10 Nagahata H, Nochi H, Tamoto K, et al. Expression and role of adhesion molecule CD18 on bovine neutrophils. Can J Vet Res. 1999;59:1.
11 Sipes KM, Edens HA, Kehrli MEJr, et al. Analysis of surface antigen expression and host defense functions in leukocyte from calves heterozygous or homozygous for bovine leukocyte adhesion deficiency. Am J Vet Res. 1999;60:1255.