FUNGAL INFECTIONS OF THE EQUINE RESPIRATORY TRACT
ALLISON J. STEWART
Definition and Etiology
Fungi are eukaryotic organisms with a definitive cell wall made up of chitins, glucans, and mannans. Within the fungal cell wall, the plasma membrane contains ergosterol, a cell membrane sterol that is frequently targeted by antifungal agents. There are over 70,000 species of fungi, but only 50 species are known to cause disease in people or animals. Pathogenic fungi can be divided into three groups: multinucleate septate filamentous fungi, nonseptate filamentous fungi, and yeasts. Dimorphic fungi are able to interchange between forms depending on environmental conditions. Examples of dimorphic organisms include Blastomyces dermatitidis, Histoplasma capsulatum, and Coccidioides immitis, which exist in yeast form in vertebrate host tissue and in hyphal or mycelial form in vitro. In soil and decaying matter, the mycelial form usually is present and is composed of a collection of hyphae. The mycelia produce infective spores that are capable of inoculating vertebrate tissue.
Fungal infections in horses are relatively uncommon, although geographic prevalence is highly variable. Fungi are ubiquitous, and their constant aerosol exposure to respiratory tissue is inevitable. Upper respiratory and pulmonary disease caused by fungi is frequently acquired by the inhalation route, with the sporular diameter small enough to allow penetration into the distal airways and alveoli. In most samples of stable air, more than 90% of particles visible under a light microscope are spores of fungi or actinomycetes.309 One study found that if a horse stood quietly in its stable without access to hay, the mean concentration of dust was very low (approximately 12 particles per cubic centimeter [of particles less than 5 mm in diameter]). When the bedding was disturbed during the normal “bedding down” operation, the concentration of respirable dust increased sixfold.310 Systemic fungal infections and some cases of fungal pneumonia are thought to arise through a compromised GI tract, by inhalation, or via open wounds.
Tissue invasion of pulmonary tissue usually occurs in the immunocompromised host, although on occasion the normal individual may be affected. Important predisposing factors for fungal pneumonia include (1) qualitative and especially quantitative granulocyte abnormalities and (2) the presence of devitalized tissue. In most cases of upper respiratory tract fungal granuloma, there are no obvious predisposing causes.
Primary pathogenic fungi such as B. dermatitidis, H. capsulatum, C. immitis, Cryptococcus neoformans, and Conidiobolus coronatus usually infect immunologically normal horses. However, in a report of cryptococcosis in seven horses, five had a history of illness that may have predisposed them to cryptococcosis.311 A separate group of fungal pathogens tend to infect only those equine patients with abnormal host defenses. Opportunistic fungi including Aspergillus species, Candida species, Fusarium species, Emmonsia crescens, and P. carinii have caused fungal disease in horses that are immunocompromised or neutropenic; have neoplasia, colitis, enteritis, or bacterial pneumonia; or have been treated with corticosteroids.312-323 In vitro studies support the critical role of phagocytic cells in host defense against opportunistic fungi.
Clinical Signs
Mycotic granulomas have been found in the nasal passages, paranasal sinuses, nasopharynx, guttural pouch, trachea, bronchioles, lungs, and mediastinum of infected horses. The most common clinical signs of upper respiratory fungal infection include unilateral or bilateral serosanguineous or mucopurulent nasal discharge, as well as inspiratory or expiratory noise. Other clinical signs include coughing, facial deformation, and dyspnea caused by partial blockage of nasal passages by granulomatous masses. Differential diagnoses for fungal granulomas of the respiratory tract include ethmoidal hematoma, squamous cell carcinoma, amyloidosis, and exuberant granulation tissue.
Fungal plaques in the guttural pouch often are located over the arterial blood supply. Horses with guttural pouch mycosis usually have episodic serosanguineous nasal discharge that may progress to potentially fatal epistaxis if there is erosion into an artery. The duration of clinical signs can vary from days to many months. Guttural pouch mycosis is discussed in detail elsewhere.
Pulmonary fungal infections causing granulomas, diffuse pneumonia, or pleuropneumonia can manifest with signs similar to those of bacterial infection. There may be cough, nasal discharge, tachypnea, respiratory distress, hemoptysis, and, if chronic, weight loss. Radiographic appearance of fungal pneumonia may reveal virtually any infiltrative pattern. Although miliary patterns are occasionally seen, the most common initial finding is a patchy bronchopneumonia. Multiple focal sites are common, and lesions tend to be peripheral in distribution. Differential diagnoses include bacterial pneumonia, recurrent airway obstructive disease, silicosis, granulomatous disease complex, or neoplasia.
Systemic infections can have variable clinical signs dependent on the location and extent of the infection. Fungal infections can affect multiple organ systems and body cavities. Weight loss, colic, or diarrhea can often occur with infection within the abdominal cavity.
Diagnostic Sampling
Lesions in the nasal passages, nasopharynx, guttural pouch, trachea, and bronchioles usually can be observed directly during endoscopic examination. Masses in lungs and paranasal sinuses may be observed radiographically. CT or MRI provides detailed imaging of the equine skull and can be used to determine the extent of lesions and bony invasion. An 8- to 20-mm trephine* can be drilled into the nasal or maxillary sinus; a sterile rigid arthroscope or flexible endoscope can then be passed through the trephine to directly view some lesions within the paranasal sinuses.
For nasal and nasopharyngeal lesions, specimens for cytology, histopathology, and culture can be obtained by use of endoscopically guided biopsy instruments; however, these samples tend to be small, superficial, and often nondiagnostic. Mucosal contaminants may overgrow the organism of interest. Larger biopsy samples from the nasal passages or nasopharynx often can be obtained by use of a uterine biopsy instrument† passed nasally with visual guidance from a flexible endoscope. Excisional biopsy or surgical debulking may be performed through a sinus flap or via laryngotomy.
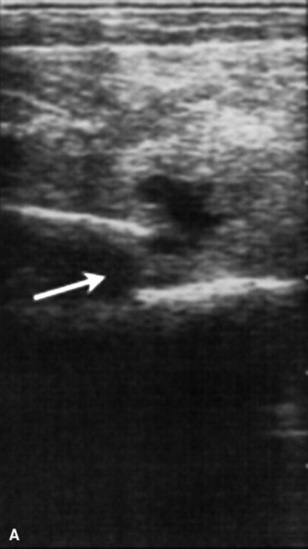
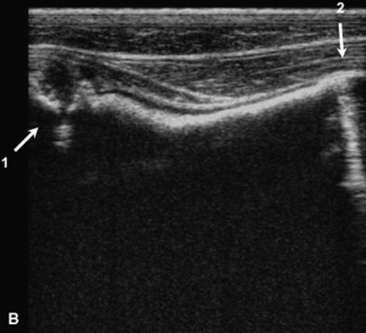
Fungal pneumonia may be diagnosed from samples obtained by tracheal wash or BAL or via a lung biopsy (Fig. 31-17). Lung biopsy is associated with significant risk if a pulmonary vessel is accidentally biopsied. The biopsy should be performed ideally after radiographic evaluation or with concurrent ultrasound guidance, and the sample should be obtained from the periphery of the lung; however, some horses have experienced fatal hemorrhage associated with biopsy of a vessel only 2 cm from the periphery. The lung is rich in plasminogen, so bleeding complications may be severe. Spring-loaded biopsy needles‡ are safer for lung biopsy than Tru-Cut biopsy instruments.324 Ultrasound evaluation can be used to monitor for bleeding after the procedure.
CYTOLOGY
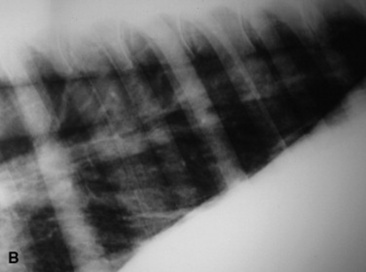
Fungal hyphae may be identified in airway fluid or in impression smears obtained from biopsied masses. Clinicians must be careful in attributing significance to the presence of fungal elements in a TTA. Fungal hyphae are often present either free or in large mononuclear cells in tracheal aspirates from healthy horses.90 A study of healthy thoroughbred racehorses showed that 70% had fungal elements detected in their tracheal aspirates.92 None of the horses from either study had other evidence of fungal pneumonia. Barn fungus such as Alternaria species are nonpathogenic and rarely incite an inflammatory response in the host. The organisms often have a blocklike appearance and may be colored (Fig. 31-18, A and B). A normal predominance of macrophages, lymphocytes, and nondegenerate neutrophils (<5% to 10%) would be expected.
To be significant cytologically, large numbers of fungi should be involved in the inflammatory process within the lung. With fungal pneumonia, aspirates may contain predominately neutrophils that often are degenerate and may contain intracellular fungal hyphae. If processing of the sample is delayed, extracellular fungi may be phagocytized, which confuses the interpretation. Some fungi have characteristic morphologic features that can permit an early presumptive identification (Table 31-5).
HISTOPATHOLOGY
Hyphae of certain fungi may be poorly visualized when routine hemotoxylin and eosin (H&E) stains are used; therefore special stains such as periodic acid Schiff (PAS), Gridley’s fungus, and Grocott-Gomori methenamine-silver nitrate (GMS) can be useful to stain histopathologic specimens. With chronicity, there is often evidence of extensive fibrosis.
MICROBIOLOGIC CULTURE
Fungi can be found in tracheal aspirates from healthy horses. Sixteen percent of healthy horses are reported to have fungal growth on the tracheal aspirate bacterial culture plates; therefore results of fungal culture must be interpreted with caution.91 Some fungi have fastidious growth requirements. They may be outcompeted by contaminant bacteria and may take up to several weeks to grow on culture media. For transport of tissue for microbiologic culture, the sample should be placed in a prepared culture medium* and transported at room temperature. Specific culture media such as Sabouraud’s dextrose agar,† inhibitory mold agar, and Mycobiotic agar containing cycloheximide and chloramphenicol are useful.
MOLECULAR TECHNIQUES
Serologic tests that use immunodiffusion, radioimmunoassays, complement fixation, and ELISA are available to detect circulating antibodies against several fungal organisms (Table 31-6). These titers often decrease with resolution of disease; therefore repeated measurements can be used to help monitor response to treatment. IHC,318,325-327 fluorescent in situ hybridization,326 and DNA probes328 can be used to positively diagnose fungal organisms in histopathogy sections. A panfungal real-time PCR assay‡ can be used to detect a variety of fungal organisms in body fluids and fungal isolates, followed by species-specific real-time PCR assay to positively identify the organism.313
IMMUNE FUNCTION TESTING
Several fungal infections have been associated with host immunosuppression caused by severe malnutrition, congenital immunodeficiency, or acquired immunodeficiency.321-323 Blood can be tested for immunoglobulin quantification by radial immunodiffusion§ and lymphocyte subpopulation phenotyping via flow cytometry. ,323,329
,323,329
Treatment
Treatment of fungal granulomas of the upper respiratory tract may involve surgical options (debulking, laser therapy, or cryotherapy) and/or medical therapy (systemic, topical, or intralesional). Treatment of Aspergillis species pneumonia is usually frustrating, as diagnosis is often made late in the course of disease and the horse frequently has severe underlying illness. Successful therapy has been reported for fungal pneumonia with other causes.313,330-333 Several pharmacokinetic studies on antifungal drugs have been recently performed,334-337 and as these medications become more affordable, the success rate of therapy is likely to increase. The drug of choice depends on the site of infection, the fungus involved, and the financial resources of the owner. Specific antifungal agents used in horses have been recently reviewed.338 Therapy may not be attempted because of the severity of the primary disease, expense, or poor prognosis.
Prevention of invasive fungal pneumonia is difficult. It is impossible for the horse to avoid large inhaled inocula, given its environmental conditions. Improving ventilation and minimizing exposure to inspired spores are most beneficial in immunocompromised patients. At present the most important methods of disease prevention are treating predisposing illnesses promptly and effectively and judiciously avoiding overuse of corticosteroids and broad-spectrum antibiotics.
ANTIFUNGAL THERAPEUTICS (Table 31-7)
Amphotericin B
Amphotericin B deoxycholate is a polyene antibiotic that combines with ergosterol in the fungal cell membrane, resulting in an increase in cell permeability. IV amphotericin B can cause nephrotoxicity and phlebitis. Other possible side effects include anorexia, anemia, cardiac arrhythmias, hepatic and renal dysfunction, and hypersensitivity reactions.339 Amphotericin B also been used successfully to treat histoplasmosis and pulmonary aspergillosis and cryptococcosis.330,331,340 A high dose of oral amphotericin B successfully treated mucormycosis caused by Absidia corymbifera.341 Topical amphotericin B has been successful in treatment of nasopharyngeal C. coronatus.342-345
Azoles
Benzimidazole derivatives in the class azoles, such as miconazole, enilconazole, ketoconazole, itraconazole, fluconazole, and voriconazole, destroy fungi by inhibition of ergosterol biosynthesis in the fungal cell membrane. Azole antifungals inhibit cytochrome P450-dependent 14α–sterol demethylase, which is essential for the formation of ergosterol. Topical 2% miconazole was used in the resolution of four cases of guttural pouch mycosis346 and as part of successful multimodal therapy against nasopharyngeal Pseudallescheria boydii.347 Enilconazole is not commercially available in the United States but has been used topically in the successful treatment of guttural pouch mycosis.348-350 Aerosolization of 1.2 mg of enilconazole per kilogram q12h in 125 mL of saline resolved Scopulariopsis pneumonia.333 Ketoconazole is absorbed poorly in the nonacidified form334 but can be acidified for better absorption (30 mg/kg via NGT q12h mixed with 0.2 NHCl).333
Itraconazole (Sporanox solution) is absorbed well orally (bioavailability 60%). A dose of 5 mg/kg PO q24h maintains concentrations above MIC for susceptible yeasts (Histoplasma and Blastomyces species) and Aspergillus species, with no detectable side effects.336 The use of compounded itraconazole is not recommended. It is very unstable (requires low pH), and owing to its lipophilic nature it is difficult to formulate in aqueous solution.
Oral fluconazole at a loading dose of 14 mg/kg followed by 5 mg/kg every 24 hours yields concentrations in plasma, cerebrospinal fluid (CSF), synovial fluid, aqueous humor, and urine above the MIC reported for several equine fungal pathogens.335 Fluconazole, however, reportedly has minimal activity against filamentous fungi (Aspergillus and Fusarium species). Low-dose oral fluconazole (1 mg/kg PO q24h) for at least 10 to 15 days anecdotally has been successful in treatment of fungal keratitis. Compounded fluconazole formulations are very stable. The cost of fluconazole has been markedly reduced since generic products became available.
Voriconazole, a new broad-spectrum triazole antifungal agent, was approved for use in human medicine in 2002. It is now considered the drug of choice for initial treatment of invasive aspergillosis, candidiasis, cryptococcosis, and serious fungal infections caused by Scedosporium apiospermum and Fusarium species in patients that are unable to tolerate or are refractory to other therapeutic agents.351,352 An initial single-dose pharmacokinetic study in horses recommended a dose of 4 mg/kg PO q24h.337 Multiple dosing studies are still required owing to the potential for drug accumulation.
SYSTEMIC IODIDE THERAPY
The exact mode of action of iodides is unknown; however, they seem to have a beneficial effect on the granulomatous inflammatory process. Iodides have very little, if any, direct in vitro antibiotic effect.353 Although several successful cases in which iodides were used as primary or adjunctive therapy have been reported, overall efficacy is considered limited at best. Treatment is inexpensive, but toxicity and resistance can occur. Iodide toxicity is characterized by excessive lacrimation, nonproductive cough, increased respiratory secretions, and dermatitis.354 The recommended dose of 20% sodium iodide is 20 to 40 mg/kg/day IV for 7 to 10 days.342,345,354,355 Orally administered iodine is available in two forms. Inorganic potassium iodide (10 to 40 mg/kg/day) is available only as a chemical grade and is unstable in the presence of light, heat, and excessive humidity.345,355 Organic ethylenediamine dihydriodide (EDDI) (0.86 to 1.72 mg of EDDI per kilogram, equivalent to 20 to 40 mg/kg/day of the 4.57% dextrose powdered form*) is commercially available.355 Administration of iodine in the diet of pregnant mares may cause congenital hypothyroidism in foals and should be avoided.
ETIOLOGIC AGENTS
Conidiobolomycosis
C. coronatus is a saprophytic fungus that causes granulomatous lesions of the upper respiratory tract in horses. Single to multiple granulomatous lesions in the nasal passages, trachea, or soft palate can be observed endoscopically (Fig. 31-19). Histologic appearance of conidiobolomycosis is similar to that of pythiosis and basidiobolomycosis. Hyphae of C. coronatus are thin-walled, highly septate, and irregularly branched (see Table 31-5).356 The lesions typically have large numbers of eosinophils and fewer macrophages, neutrophils, plasma cells, and lymphocytes surrounding hyphae. Definitive diagnosis is based on microbiologic culture, immunodiffusion, or PCR.357 Detection of serum antibodies by immunodiffusion is considered highly sensitive and specific354,358 and can be used to monitor response to treatment.358 A nested PCR Pythium assay has also been used to identify C. coronatus.359
Conidiobolomycosis lesions can be treated with surgical excision, laser therapy, cryotherapy, or long-term administration of iodides or antifungals.342,354,357 Amphotericin B has been administered intralesionally or topically in combination with dimethyl sulfoxide (DMSO) to treat C. coronatus.342-345 It is important to remember that long-term therapy and reevaluation are essential, as recurrence can occur.345 Oral fluconazole was successful in treating two pregnant mares with nasal conidiobolomycosis.360 A vaccine using C. coronatus antigen from broth cultures was unsuccessful in treating seven horses with conidiobolomycosis.357
Cryptococcosis
Cryptococcosis is caused by C. neoformans (var. neoformans and var. gattii) and is a ubiquitous, saprophytic, round, basidiomycetous yeastlike fungus with a large heteropolysaccharide capsule that does not take up common cytologic stains (see Table 31-5, Fig. 31-20). The capsule forms a clear halo when stained with India ink. The capsule is immunosuppressive and antiphagocytic. A relatively high frequency of equine cryptococcosis occurs in Western Australia.311 There is an epidemiologic relationship between C. neoformans var. gattii and the Australian river redgum tree (Eucalyptus camaldulensis), whereas C. neoformans var. neoformans has historically been associated with bird (particularly pigeon) excreta.311 Cytologic or histopathologic identification is very reliable for diagnosis because of the characteristic morphology.361 Serologic testing with latex agglutination to identify cryptococcal capsular antigen is useful with resolution of lesions correlated with declining serum titers.331
Cryptococcosis in horses is associated primarily with pneumonia, rhinitis (Fig. 31-21), meningitis, and abortion. Successful medical treatment, however, has been reported rarely. Surgical removal of a localized jejunal lesion was successful in one horse.362 A pony with multiple pulmonary cryptococcomas, from which Cryptococcus gattii was cultured from both transtracheal washings and lung mass aspirates, was treated successfully with daily infusions of amphotericin B over 1 month. One year after cessation of treatment, clinical signs had resolved and the cryptococcal antigen titer had decreased from 4096 to 256.331
Cryptococcus should be treated with amphotericin B, fluconazole, or voriconazole (see Table 31-7).
Pseudallescheriosis
P. boydii is a saprophytic ascomycete. Infection most commonly involves the extremities, and in human patients it is known as Madura foot. Hyphae within tissue cannot be differentiated from Fusarium or Aspergillus species unless cultured. P. boydii cultured from the nasal cavity and sinus of a horse with chronic, malodorous nasal discharge was susceptible in vitro to miconazole, ketoconazole, natamycin, and clotrimazole. After debridement and flushing of the plaque, miconazole cream was infused twice daily for 4 weeks through lavage tubing that had been passed into the nasal passage through a hole drilled in the frontal bone and sinuses. Adjunctive iodide therapy was also administered, and the lesions resolved (Fig. 31-22).347
Nasal mycosis caused by P. boydii has been reported in two other horses, both of which were euthanized.363,364 P. boydii has also been isolated from the pharynx of 2 of 60 normal donkeys and from horses with chronic uterine infection.365,366
Aspergillosis
Aspergillus species have broad septate hyphae with parallel sides and acute right-angled branching (see Table 31-5). They have a propensity for vascular invasion. Definitive diagnosis is by culture or staining by IHC or immunofluorescence. Aspergillus species are very common in the environment, especially in moldy feed and bedding.340 They are opportunistic pathogens and often cause disease in horses that are immunosuppressed from debilitating disease such as enterocolitis, septicemia, neoplasia, Cushing’s disease, or equine protozoal myeloencephalitis or because of surgery or that have been treated with immunosuppressive drugs312,314,316-318 (Fig. 31-23).
Infection is by inhalation of an overwhelming number of spores resulting in fungal proliferation and invasion of the small airways, or by translocation of organisms across an inflamed GI tract leading to lesions centered around large blood vessels as a result of hematogenous spread. In two retrospective studies of invasive pulmonary aspergillosis, 41 of 49 cases were associated with enterocolitis.312,314 Aspergillus species pneumonia is almost uniformly fatal, often with no or mild respiratory signs. Antemortem diagnosis of pulmonary aspergillosis is made rarely. In a retrospective study of 30 cases of Aspergillus species pneumonia, only two cases were diagnosed or suspected antemortem.314 TTA or BAL may not be helpful because hyphae and spores are often present extracellularly or within macrophages in aspirates and lavages from healthy animals.312 False-negative results also can occur. Cytologic examination of a transtracheal wash specimen obtained from the foal shown in Fig. 31-23, A, 2 days before euthanasia failed to identify Aspergillus species hyphae. Serologic diagnosis occasionally has been useful367 but is often unreliable because many horses have titers to Aspergillus species. Development of a commercially available ELISA is promising.316,340
In human medicine 50% to 90% of patients with invasive aspergillosis die despite treatment. Amphotericin B has been the mainstay of treatment for invasive aspergillus for decades but is associated with nephrotoxicity in about 50% of human patients. Nephrotoxicity is reduced using liposomal amphotericin B. Voriconazole, a new azole antifungal, is now considered the drug of choice against human aspergillosis, whereas caspofungin (in the new class of echinocandin antifungals) shows promising results in patients with refractory infections.368 Oral itraconazole is currently the treatment of choice for aspergillosis in horses owing to a lack of pharmacokinetic analysis and safety testing and the costs associated with the newer antifungal agents.336 In human medicine itraconazole has shown comparable response rates to amphotericin B.368 There are limited reports of horses surviving pulmonary aspergillosis.340
Treatment of mycotic aspergillus rhinitis and sinusitis in horses has been more successful. It is, however, important to rule out concomitant invasive pulmonary aspergillosis. Oral itraconazole,369 topical natamycin (flushed via an endoscope or indwelling catheter placed into the sinus), and nystatin powder (insufflated up the nostril) have been curative.370
Blastomycosis
Blastomycosis is caused by inhalation of conidiae of the thermally dimorphic saprophytic fungus B. dermatitidis. Blastomyces yeasts can be identified on cytologic examination, often within multinucleated giant cells. They are spheric, with basophilic protoplasm and unstained, uniformly shaped refractile walls. Unilateral, broad-based budding is characteristic (see Table 31-5).
Blastomycosis was reported to cause pyogranulomatous pleuropneumonia, pulmonary abscessation, peritonitis, and abscesses in a 5-year-old horse.328B. dermatitidis was positively identified from transtracheal wash fluid by use of a DNA probe, and serology was strongly positive. The horse was euthanized without treatment.328 Disseminated blastomycosis was diagnosed in a miniature horse with SC infections associated with a chronic pectoral wound. The pony had pulmonary consolidation and pleural effusion. Yeasts were observed histologically in many tissues, and B. dermatitidis was cultured after 6 weeks.371
Treatment with amphotericin B, itraconazole, or fluconazole is recommended (see Table 31-7).
Histoplasmosis
Histoplasmosis is caused by the saprophytic, dimorphic fungus H. capsulatum, which is most prevalent in moist soil containing bird or bat waste. Yeast organisms are 2 to 4 μm in diameter, with a thin clear halo surrounding a round or crescent-shaped basophilic cytoplasm (Fig. 31-24). Histoplasmosis has been reported in less than 10 equine cases,330 and thus horses are considered to be relatively resistant to disease. H. capsulatum may occur in an enteric, pulmonary, or disseminated form.330H. capsulatum was identified in pulmonary granulomas in a horse dying of chronic Yersinia colitis372 and in another horse with intestinal salmonellosis.373 It has also been associated with abortions and severe granulomatous pneumonia in neonatal foals (Fig. 31-25) and a yearling.374 Successful treatment with amphotericin B was reported in a filly with pulmonary histoplasmosis diagnosed by cytologic identification of the organism on a tracheal wash smear and from a lung aspirate.330
Treatment with amphotericin B or itraconazole is recommended (see Table 31-7).
Coccidiomycosis
C. immitis is a soil saprophyte that grows in semiarid areas with sandy, alkaline soils.375 Inhaled arthroconidia enlarge to form nonbudding spherules, which incite an inflammatory reaction in the lungs and lymph nodes.375 Horses have weight loss, fever, abdominal pain, and signs of respiratory disease (Fig. 31-26). Localized, recurring nasal granulomas also have been reported.376 Diffuse infections with granulomas in the lungs, liver, kidney, or spleen carry a grave prognosis.375 Przewalskii horses may be more susceptible.377
C. immitis is difficult to culture, and spherules may not be observed histologically from antemortem lung biopsies. However, serology is very useful to diagnose infection, and decreasing titers are associated with clinical improvement.332,375 Serum antibodies are detected rarely in healthy horses.378 Antifungal agents successful in treatment of infected horses include itraconazole and fluconazole.332,379
Scopulariopsis
Scopulariopsis pneumonia was diagnosed by culture of BALF in a 2-year-old quarter horse filly with pleuropneumonia. The infection resolved after multimodal therapy with ketoconazole and aerosolization of enilconazole.333
Adiaspiromycosis
Adiaspiromycotic miliary fungal pneumonia caused by the saprophytic soil mold E. crescens was diagnosed in a horse by percutaneous lung biopsy. Euthanasia was performed without treatment.319
Acremonium Strictum
A diagnosis of interstitial fungal pneumonia caused by Acremonium strictum was made based on cytologic evaluation, culture, and PCR testing of BALF in a 10-year-old horse. The horse made an uneventful recovery with supportive treatment, which included 1 month of fluconazole.313 In general, fluconazole has been shown to have poor activity against A. strictum in vitro.380 The isolate cultured from the horse was found to be resistant to fluconazole based on in vitro sensitivity testing that was performed after the course of treatment. It is therefore uncertain if the fluconazole assisted in disease resolution.313
Candidiasis
In human medicine, candidemia is the most common fungal infection in burn patients, neutropenic patients with malignancies, patients undergoing complex abdominal surgery, and patients receiving total parenteral nutrition and long-term corticosteroid therapy.368 In humans, Candida species account for 8% to 10% of all blood culture isolates and rank fourth in the list of most frequently isolated pathogens from blood cultures.368 The mortality in patients with candidemia is 40% to 75%.368 Over 60% of isolates are Candida albicans.368 Fluconazole is generally considered the drug of choice against Candida species, although Candida krusei is resistant to fluconazole. Itraconazole, amphotericin B, caspofungin, and voraconazole are alternative antifungal agents (see Table 31-7).368
Systemic candidiasis was diagnosed and successfully treated in four neonatal foals. Each foal had prior sepsis attributable to gram-negative bacteria that had been aggressively treated with numerous antibiotics and parenteral nutrition. C. albicans was cultured from a transtracheal wash from one of the foals. Three of the foals had Candida glossitis, and one had panophthalmitis and fungal keratitis. Two of the foals were treated with IV amphotericin B, and oral fluconazole was used on the other two foals.320
Superficial Candida species infections of the mucous membranes (thrush)381 can occur in isolation or as part of a systemic infection, and further microbiologic culturing of the blood, tracheal wash, urine, or joint fluid may be indicated to rule out systemic infection.320Candida species glossitis can be treated by rinsing the mouth either with potassium permanganate (0.025% q24h) or nystatin (0.3 g in 10 mL of water q8h).
Pneumocystosis
P. carinii has been reclassified from a protozoan to a saprophytic fungus based on the DNA sequence of its 16S-like RNA subunit, but some researchers even consider it to be a plant because it lacks ergosterol, the major fungal sterol.382 It exists as an ameboid yeast or as a cystic sporangia. P. carinii cannot be cultured, and diagnosis is based on the cytologic identification of characteristic morphologic features using specimens obtained by BAL rather than tracheal wash. A fluorescent in situ hybridization method with an oligonucleotide probe that targets the 18S ribosomal RNA has been developed recently to detect P. carinii in histologic sections.326 IHC also can be used.325
Three quarters of human AIDS patients are infected with P. carinii, and people undergoing immunosuppressive therapy after organ transplantation are predisposed. P. carinii causes diffuse interstitial pneumonia, especially in immunocompromised patients such as Arabian foals with SCID.321 It also has been diagnosed in immunocompromised adult horses,322,323 as well as an immunocompetent foal.325 TMS (25 to 30 mg/kg PO q12h) is the treatment of choice (see Table 31-5).383,384 Dapsone* (3 mg/kg PO daily for 2 months) was used to successfully treat a foal that developed Salmonella enterocolitis after treatment with TMS.329
STREPTOCOCCUS EQUI INFECTION (STRANGLES)
Corinne R. Sweeney
S. equi infections in horses, commonly referred to as strangles, was described in early veterinary science literature. Since described by Jordanus Ruffus in 1251, much has been learned about the disease and the organism that causes it.387,388
Clinical Signs
Strangles is characterized by sudden onset of fever followed by upper respiratory tract catarrh, as evidenced by mucopurulent nasal discharge (Fig. 31-27) and acute swelling with subsequent abscess formation in submandibular and retropharyngeal lymph nodes. The name strangles was coined because affected horses sometimes suffocated as the lymph nodes became enlarged and obstructed the airway. Clinical severity varies greatly depending on the immune status of the animal.
Fever is the first clinical sign and is maintained as lymphadenopathy develops and abscesses mature. Pharyngitis makes swallowing painful, and affected animals may become anorectic or reluctant to eat and stand with the neck extended.
Lymphadenopathy is a major clinical sign. The submandibular and retropharyngeal lymph nodes are about equally involved in S. equi infections. They usually become swollen and painful approximately 1 week after infection. Other lymph nodes of the head region (parotid, cranial cervical, and retropharyngeal) are also frequently involved and may abscess (Fig. 31-28). Retropharyngeal lymph nodes may drain into and cause empyema of the guttural pouch (Fig. 31-29). Periorbital abscesses can cause marked swelling of the eyelids. Abscesses of the lymph nodes at the thoracic inlet can cause severe tracheal compression, asphyxia, and death.
Pathogenesis
S. equi enters via the mouth or nose and attaches to cells in the crypt of the lingual and palatine tonsils and to the follicular associated epithelium of the pharyngeal and tubal tonsils. There is no evidence of colonization before penetration. After a few hours the organism is difficult to detect on the mucosal surface but is visible within cells of the epithelium and subepithelial follicles. Translocation occurs in a few hours to the mandibular and suprapharyngeal lymph nodes that drain the pharyngeal and tonsillar region.
Complement-derived chemotactic factors attract large numbers of polymorphonuclear neutrophils (PMNs), although gross evidence of abscessation is not visible for 3 to 5 days after S. equi enters the lymph node. Failure of neutrophils to phagocytose and kill the streptococci appears to be a result of a combination of the hyaluronic acid capsule, antiphagocytic SeM protein, Mac protein, and other undetermined antiphagocytic factors released by the organism.
Although strangles predominantly involves the upper airways including the guttural pouches and associated lymph nodes, metastasis to other locations occasionally occurs. Spread may be hematogenous or via lymphatic channels, which results in abscesses in lymph nodes and other organs of the thorax and abdomen (Fig. 31-30). This form of the disease is known as bastard strangles.
Nasal shedding of S. equi usually begins 2 to 3 days after onset of fever and persists for 2 to 3 weeks in most animals. Some animals never shed. In others, shedding may persist much longer, should infection persist in the guttural pouch.389,390 Systemic and mucosal immune responses are evident 2 to 3 weeks after infection and coincide with mucosal clearance.
Approximately 75% of horses develop a solid enduring immunity to strangles after recovery from the disease. A small percentage of these horses become susceptible to a second attack of the disease within months, which probably represents a failure to produce or maintain an adequate level of the appropriate mucosal and systemic antibodies. Older horses with residual immunity have limited susceptibility and develop a mild form of strangles often termed catarrhal strangles. These animals shed virulent S. equi that will produce severe disease in more susceptible, often younger horses. Colostral antibodies ingested during the first 24 hours of life have also been shown to recirculate to the nasopharyngeal mucosa, thus providing an additional source of protection to the foal during its first weeks. Foals that suckle immune mares are usually resistant to S. equi infection until weaning.
Epidemiology
TRANSMISSION
Purulent discharges from horses with active strangles and those that are recovering are an important and easily recognizable source of new S. equi infections among susceptible horses. Transmission of infection occurs when there is either direct or indirect transfer of S. equi within these purulent discharges between affected and susceptible horses. Direct transmission refers to horse-to-horse contacts, particularly through normal equine social behavior involving mutual head contact. Indirect transmission occurs through the sharing of contaminated housing, water sources, feed or feeding utensils, twitches, tack, and other less obvious fomites such as the clothing and equipment of handlers, caretakers, farriers, and veterinarians unless appropriate barrier precautions are undertaken to prevent spread of S. equi.
It is increasingly recognized that there may be transmission that originates from outwardly healthy-appearing animals, and in this situation the source of infection may not be readily recognized and clinical signs may appear in in-contact animals without warning. S. equi may originate from outwardly healthy horses as follows:

Horses that are incubating the disease and go on to develop signs themselves

Horses that are recovering from recent disease but that continue to harbor the organism after full clinical recovery for some weeks

Horses that are fully recovered from the disease but continue to be potentially infectious for prolonged periods through periodic shedding of
S. equi
ENVIRONMENTAL PERSISTENCE OF STREPTOCOCCUS EQUI
Currently there is a lack of definitive field-based proof for prolonged environmental persistence of S. equi. S. equi is known to be sensitive to bacteriocins from environmental bacteria and does not readily survive in the presence of other soil-borne flora.
Diagnosis
CULTURE
Culture of nasal swabs, nasal washes, or pus aspirated from abscesses remains the gold standard for detection of S. equi. Nasal washes are more effective than swabs in detection of small numbers of S. equi because a greater surface area within the internal nares is sampled. Culture may, however, be unsuccessful during the incubation and early clinical phases. S. equi is normally not present on the mucosa until 24 to 48 hours after the onset of fever, so infection in horses monitored by daily measuring of rectal temperatures during an outbreak may be recognized early and isolated to limit transmission of S. equi.
POLYMERASE CHAIN REACTION
The PCR is designed to detect DNA sequence of SeM, the gene for the antiphagocytic M protein of S. equi. However, PCR does not distinguish between dead and live organisms, so a positive test result must be considered presumptive until confirmed by culture. In addition, clinical samples that contain polymerase inhibitors or abundant S. equi may give negative PCR results although culture of the same sample confirms the presence of S. equi. PCR accompanying culture on a nasal swab or wash may be used in a control program to select animals for guttural pouch endoscopy.389,391
SEROLOGY
Fifteen or more surface exposed or secreted proteins of S. equi elicit strong serum antibody responses during infection and convalescence. The most reactive and best studied of these is SeM, a major virulence factor and protective immunogen. A proprietary ELISA for measuring SeM specific antibody is commercially available (EB1, IDEXX, Lexington, Ky.) and is useful for diagnosing recent (but not necessarily current) S. equi infection, determining the need for booster vaccination, and aiding in the diagnosis of purpura hemorrhagica and metastatic abscesses. It does not distinguish between vaccine and infection response. That considerable variation exists in the responses of individual horses should be kept in mind when interpreting results.
Vaccination
Most horses develop a solid immunity during recovery from strangles, which persists in over 75% of animals for 5 years or longer. This indicates that stimulation of a high level of immunity is biologically feasible given appropriate presentation of protective immunogen(s). There is evidence that immunity in horses resistant to reinfection is mediated at the mucosal level and functions to block entry of S. equi. However, systemic immunity after parenteral inoculation of avirulent live S. equi is also protective. Together, these findings indicate that optimum immunity may require both systemic and mucosal responses.
It is likely that more effective and safer vaccines will eventually be developed based on genomic sequence information from S. equi and S. zooepidemicus. Protective immunogens must be identified for systemic and mucosal responses, and the appropriate modes of presentation elucidated by experiment. Because it is likely that different immunogens function at the tonsillar and lymphatic levels, the appropriate combination of these components will also have to be identified in multiple experiments with ponies and horses.
USE OF EXTRACT VACCINES
Extract vaccines are given intramuscularly or subcutaneously and elicit serum antibody responses 7 to 10 days later. Naïve horses and foals require a schedule of two or three doses at an interval of 2 weeks. Booster doses are given once annually. Pregnant mares may be boostered a month before expected date of foaling. Horses known to have had strangles within the previous year should not be vaccinated. Horses with signs of strangles should not be vaccinated. During an outbreak, only horses with no known contact with animals with strangles should be promptly vaccinated.
USE OF ATTENUATED LIVE INTRANASAL VACCINE
Live vaccine should be administered only to healthy nonfebrile animals free of nasal discharges. Vaccine is given in a schedule of two doses at 2- to 3-week intervals. Annual booster doses are recommended. Live vaccine should not be used during an outbreak, except in horses with no known contact with infected or exposed animals. The mode of application should be such that an adequate amount of vaccine reaches the pharyngeal and lingual tonsils.
Control of Outbreaks
A practical disease-control strategy should then be agreed on and implemented. The general aims and measures for such a strategy are outlined in Table 31-8.387
Table 31-8 Aims and Measures Used To Control Transmission of S. equi on Affected Premises
| Aim |
Measure |
| 1. |
To prevent the spread of S. equi infection to horses on other premises and to new arrivals on the affected premises |
Stop all movement of horses on and off the affected premises immediately and until further notice.
Horses with strangles and their contacts should be maintained in well-demarcated “dirty” (i.e., S. equi positive) quarantine areas.
Clustering of cases in groups should allow parts of the premises to be easily allocated as “dirty” and “clean” areas. |
| 2. |
To establish whether convalescing horses are infectious after clinical recovery |
At least three nasopharyngeal swabs or lavages are performed at approximately weekly intervals in all recovered horses and their contacts, and the samples are tested for S. equi by culture and PCR assay.
Horses that are consistently negative are returned to the “clean” area. |
| 3. |
To investigate all outwardly healthy horses in which S. equi is detected by either culture or PCR assay |
Perform endoscopy of the upper respiratory tract and guttural pouches.
|
| 4. |
To eliminate S. equi infection from the guttural pouches |
Remove pathology through a combination of flushing and aspiration with saline, and remove chondroids using endoscopically guided instruments.
Perform topical and systemic administration of antimicrobials to eliminate S. equi infection. |
| 5. |
To prevent indirect cross-infection by S. equi from horses in the “dirty” area to those in the “clean” area of the premises |
Personnel should use dedicated protective clothing when dealing with infectious animals and should not deal simultaneously with susceptible animals.
If this is unavoidable, infectious horses should be dealt with after susceptible animals.
Strict hygiene measures are introduced, including provision of dedicated clothing and equipment for each area and disinfection facilities for personnel and use of thorough stable cleaning and disinfection methods.
When cost is not a factor, consideration should be given to destruction of the dedicated equipment after eradication of the infection.
After removal of organic material from stables, all surfaces should be thoroughly soaked in an appropriate liquid disinfectant or steam-treated and allowed to dry. This should be repeated if possible.
Manure and waste feed from infectious animals should be composted (disinfected by heat) in an isolated location.
Pastures used to hold infectious animals should be rested for 4 weeks.
Care should be taken to disinfect water troughs at least once daily during an outbreak.
Horse vans should be hosed clean and disinfected after each use. |
PCR, Polymerase chain reaction.
Treatment
Appropriate treatment of horses with strangles usually depends on the stage and severity of the disease, and veterinary opinion as to whether or not to use antibiotic treatment remains markedly divided. However, the majority of strangles patients require no treatment other than proper rest, a dry, warm stall, and soft, moist, and palatable food of good quality while the disease is allowed to run its course. Food and water should be easily accessible to the horse.
HORSES WITH EARLY CLINICAL SIGNS
During an outbreak, immediate antibiotic therapy for new cases in the early acute phase with fever and depression may be curative and may prevent focal abscessation. Because abscesses have not developed at this early stage, the antibiotics have adequate access to the bacteria. Immediate treatment of horses that show the earliest clinical sign, fever, could be an effective way of controlling strangles outbreaks in racing stables or riding barns, although the disadvantages of treatment should be weighed.
HORSES WITH LYMPH NODE ABSCESSATION
Once an external lymphadenopathy is detected in an otherwise alert and healthy horse, antibiotic therapy is probably contraindicated. Although it provides temporary clinical improvement in fever and lethargy, it only prolongs the inevitable enlargement and eventual rupture of lymph node abscess. Therapy should be directed toward enhancing maturation and drainage of the abscesses. Topical treatments such as Ichthammol or a hot pack may be applied to promote maturation of the lymph node abscess, although objective controlled data supporting the use of these techniques are lacking. Surgical drainage of lymphadenopathies is sometimes indicated if abscesses do not rupture spontaneously; however, it is critical to wait until the abscess has matured and thinned out ventrally. The use of NSAIDs such as phenylbutazone may improve the horse’s demeanor by reducing fever, pain, and inflammatory swelling at the site of the abscesses. This may in turn encourage eating and drinking.
Even in the face of detectable lymphadenopathy, if the horse is febrile, depressed, anorectic, and especially manifesting dyspnea as result of partial upper airway obstruction, antibiotic therapy is indicated to decrease abscess size and prevent complete airway obstruction. Rarely, affected horses may require intensive supportive therapy, including IV fluids, feeding by nasogastric tube, and tracheostomy. An animal requiring a tracheostomy should be given systemic antimicrobial drugs to prevent secondary bacterial infections of the lower respiratory tract.
DRUGS OF CHOICE FOR THERAPY
Penicillin is generally considered the drug of choice for the treatment of nonpneumococcal streptococcal disease, with alternative drugs used depending on ease of administration or the site of infection. Other agents for therapy include cephalosporins and macrolides. Although there is evidence that trimethoprim-sulfadiazine did not eliminate S. zooepidemicus infection in tissue chambers implanted subcutaneously in ponies, the study did not determine its effectiveness against S. equi. Anecdotally, many veterinarians believe that trimethoprim-sulfadiazine is effective in treating horses with strangles.
Complications
The overall complication rate associated with S. equi infection is approximately 20%. The occurrence of complications can significantly increase the mortality rate.
A variety of complications can occur as a result of strangles. These can be generally grouped as:

Those associated with the spread of infection to other locations (bastard strangles)

Immune-mediated processes, including purpura hemorrhagica and myopathies

Other complications
COMPLICATIONS ASSOCIATED WITH METASTATIC SPREAD OF INFECTION
S. equi may potentially infect any site. The term bastard strangles is often used to describe metastatic abscessation. Spread of the organism may occur through several routes, including hematogenous spread, lymphatic migration, or spread via close association with a septic focus or direct aspiration of purulent material.
Common sites of infection include the lung, mesentery, liver, spleen, kidneys, and brain. Respiratory distress may occur owing to tracheal compression resulting from enlargement of the cranial mediastinal lymph nodes. Suppurative bronchopneumonia is one important sequela of strangles.
Another important complication of strangles is extension of infection to the sinuses or guttural pouches. Infection of the guttural pouch is of particular importance, as the guttural pouch is the most common site for prolonged carriage of the organism.389 Other reported conditions associated with S. equi infection include myocarditis, endocarditis, panophthalmitis, periorbital abscesses, ulcerative keratitis, paravertebral abscesses, septic arthritis, and tenosynovitis.
IMMUNE-MEDIATED COMPLICATIONS
Purpura Hemorrhagica
Purpura hemorrhagica is an aseptic necrotizing vasculitis characterized primarily by edema and petechial or ecchymotic hemorrhage. Although the exact pathogenesis of purpura hemorrhagica is not fully understood, it appears to a vasculitis caused by the deposition of immune complexes in blood vessel walls. The risk of developing purpura hemorrhagica after exposure to S. equi through infection or vaccination is not known. A preexisting high serum antibody titer to S. equi antigens may predispose horses to the development of purpura hemorrhagica.
Myositis
Both muscle infarctions and rhabdomyolysis with progressive atrophy have been documented in horses after exposure to S. equi.

Muscle infarctions: This syndrome is most likely a manifestation of purpura hemorrhagica. On histopathology, there is acute coagulative necrosis of muscle with infarctions.

Rhabdomyolysis with progressive atrophy: Significant rhabdomyolysis has been identified in some quarter horses after exposure to
S. equi.
OTHER COMPLICATIONS
Streptococcal antigens have been suggested as a trigger for development of myocarditis and proliferative glomerulonephritis. Agalactia has been reported in broodmares with strangles. Although infection of the mammary glands is possible, the mammary glands are usually normal and the agalactia is thought to be secondary to the fever, anorexia, and lethargy associated with infection.
ACUTE RESPIRATORY DISTRESS SYNDROME AND ACUTE LUNG INJURY (ACUTE BRONCHOINTERSTITIAL PNEUMONIA)
BETTINA DUNKEL
PAMELA A. WILKINS
Definition and Pathophysiology
ALI and ARDS comprise a syndrome of severe pulmonary dysfunction and respiratory failure caused by physical or chemical injury or an exaggerated pulmonary immune response. In human medicine ALI and ARDS are defined by an acute onset, bilateral pulmonary infiltrates, a ratio of pulmonary arterial oxygen pressure (PaO2) to fraction of inspired oxygen (FiO2) of <300 for the less severe form, ALI, and <200 for ARDS, and no clinical evidence of left atrial hypertension or a pulmonary arterial occlusion pressure of <18 mm Hg.392 The definition acknowledges ALI and ARDS as clinically recognized conditions, regardless of the inciting cause of the respiratory failure.393 No consensus with regard to a uniformly accepted definition in veterinary medicine has been published as of this writing.
ALI or ARDS arises as a complication after major infectious or noninfectious bodily injury. During infections, such as pneumonia, inflammatory cells and mediators localize and destroy the infectious agent while fibrin generated by a locally confined procoagulant state immobilizes the source of infection and minimizes spread of the pathogens.394 In ALI and ARDS an imbalance of proinflammatory and antiinflammatory factors combined with activation of the pulmonary endothelium and derangement of the coagulation cascade favors the development of a fulminant, uncontrolled pulmonary inflammatory response and a widespread procoagulant environment in alveoli and the pulmonary microcirculation.393 The triggering event may be an intrapulmonary (viral or bacterial pneumonia, smoke inhalation, near drowning, or food aspiration) or extrapulmonary (trauma, multiple transfusions, systemic inflammatory response, or sepsis) insult.393,395
Although not temporally distinct and largely overlapping, the pathophysiologic events can be divided into an initial exudative phase, a fibroproliferative phase, and, if the patient survives, a recovery phase.396 The exudative phase is characterized by uncontrolled released of inflammatory mediators such as IL-1, IL-6, IL-8, and TNF-α and influx of inflammatory cells into the pulmonary tissue. Activated neutrophils and alveolar macrophages marginate in the microcirculation and extravasate into the pulmonary tissue, where they release oxidants, reactive oxygen species, proteases, and cytokines. These in turn attract and activate further inflammatory cells, thereby amplifying and perpetuating the inflammatory cascade. The inflammatory process ultimately damages or destroys the alveolar endothelial and epithelial barrier, causing flooding of the alveoli once the edema safety factors are exhausted. The barrier tends to collapse suddenly, resulting in rapid filling of the alveoli with proteinaceous exudate, leukocytes, and red blood cells and destruction of the surfactant layer.393 The fast onset of pulmonary edema may correspond with the clinically observed acute onset of respiratory distress. Endotoxin and inflammatory cytokines such as TNF, IL-1, and IL-6 activate the coagulation cascade by tissue factor release while fibrinolysis is inhibited by increased plasminogen activator inhibitor (PAI)–1 activity. The result is formation of thrombi in the pulmonary microvasculature and deposition of intraalveolar fibrin-rich hyaline membranes.394,396 The fibroproliferative response, which can begin within 24 hours of the onset of ALI and ARDS, is characterized by type I pneumocyte necrosis and proliferation of the more resistant type II pneumocytes in an attempt to restore the epithelial surface.393,396 Fibroblasts invade pulmonary interstitium, alveolar walls, and deposited collagen. The resultant fibrosis reduces pulmonary compliance and increases the work of breathing, and alveolar obliteration and interstitial thickening lead to poor gas exchange.396
The effect on the patient is severe hypoxemia with hypocapnia or hypercapnia caused by ventilation-perfusion (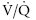 ) mismatch and decreased pulmonary compliance. Alveolar edema causes hypoxemia by creating areas of a low
) mismatch and decreased pulmonary compliance. Alveolar edema causes hypoxemia by creating areas of a low 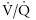 ratio (adequate perfusion but no ventilation), whereas areas with deceased perfusion generate hypoxemia and eventually hypercapnia by increasing dead space ventilation (high
ratio (adequate perfusion but no ventilation), whereas areas with deceased perfusion generate hypoxemia and eventually hypercapnia by increasing dead space ventilation (high 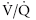 ratio; adequate ventilation with minimal perfusion).393 The coexistence of areas with high and low
ratio; adequate ventilation with minimal perfusion).393 The coexistence of areas with high and low 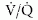 ratios adjacent to normal lung tissue makes mechanical ventilation of patients with ALI and ARDS extremely challenging.
ratios adjacent to normal lung tissue makes mechanical ventilation of patients with ALI and ARDS extremely challenging.
The resolution of ALI and ARDS depends on a functional distal lung epithelium. For edema to resolve, endothelial and epithelial leaks must be sealed before active salt and water transport can clear the edema fluid followed by cellular protein clearance.393 Sodium and chloride ions are actively transported via ion channels (epithelial sodium channel ENaC and cystic fibrosis transmembrane conductance regulator CFTR, a chloride transporter) on the apical surface and Na/K-ATPase on the basolateral membrane of alveolar epithelial cells. Water follows the created osmotic gradient through aquaporin-5 channels located on type I epithelial cells.393,397 If damage to the pulmonary epithelium is minor, pulmonary edema can be cleared swiftly, pulmonary function improves rapidly, and chances of survival increase. If large areas of alveolar epithelium are destroyed, repair must occur before the edema can be cleared.393 Severe damage to the epithelium corresponds with prolonged respiratory failure, slow recovery by gradual regeneration of the respiratory epithelium, and high mortality.397 Although the mortality of human patients with ALI and ARDS is still high, less than 5% of patients die of refractory hypoxemia. Most patients succumb to their primary disease process and multiorgan failure.396 Recovering patients may have complete resolution of pulmonary compromise, whereas others suffer from residual functional impairment such as muscle weakness, decreased pulmonary function, restrictive or obstructive changes, and low diffusion capacities.396
An ALI-ARDS-like syndrome has been described in 1- to 7-month-old foals and occasionally in neonatal foals.398-400 In earlier reports the condition was named acute interstitial or bronchointerstitial pneumonia. Extensive searches for a common pathogen were conducted until with increasing understanding of the pathophysiology it became evident that multiple infectious and noninfectious agents are able to initiate the inflammatory response.398,400-402 In foals the most common underlying condition appears to be bacterial pneumonia, and in rare cases the underlying condition is viral pneumonia.398-402 There are no reports of ALI or ARDS in adult horses, although based on few clinical case reports and small studies one can suspect that a similar form of acute pulmonary injury exists. Buergelt and colleagues398 reported not only on 14 foals with acute respiratory distress, but also on six adult horses. However, the onset of respiratory disease in the adults was more prolonged (11 days to 2 months), and the description better fit the heterogenous group of interstitial pneumonia than ALI and ARDS. Two adult horses with respiratory distress after near drowning met the criteria for ALI,403,404 and two horses affected by smoke inhalation experienced fulminant pulmonary inflammation and most likely ALI.405 Acute respiratory distress in ponies after experimentally induced gram-negative sepsis has been reported, and although some of the ponies may have suffered from ALI and ARDS, most animals were agonal, making interpretation of the observed pulmonary changes difficult.406
Clinical, Diagnostic, and Postmortem Findings
Clinically it is almost impossible to differentiate foals with ALI and ARDS from foals with severe bacterial, particularly advanced R. equi, pneumonia, and in a number of cases ALI or ARDS has been triggered by an underlying bacterial infection. Clinical signs include profound depression, fever, and respiratory distress with tachycardia, tachypnea, nostril flare, and cyanotic mucous membranes. Auscultation of the lungs can reveal a variety of abnormal sounds or even complete silence over nonventilated areas. Laboratory findings include severe hypoxemia with hypocapnia or hypercapnia and, depending on the underlying disease process, leukocytosis or leukopenia and hyperfibrinogenemia. Signs of disseminated intravascular coagulation were present in two foals with ALI and ARDS in one report,401 and an abnormal coagulation panel may be observed. Transtracheal lavage fluid demonstrates neutrophilic inflammation with or without signs of infection, depending on the underlying cause. Thoracic radiographs demonstrate a diffuse, dense bronchointerstitial pattern often coalescing to a focal or diffuse alveolar pattern with prominent airbronchograms. The initial radiographic appearance of the lungs may worsen dramatically within hours because of rapid progression of pulmonary edema. On the other hand, absorption of the alveolar fluid may result in impressive improvement of the radiographic appearance within days.401,402 A thoracic sonogram is useful in foals that cannot be safely transported to a radiography unit and reveals multiple coalescing comet-tail artifacts predominately in the caudodorsal lung field.
On gross pathologic examination, the affected lungs are wet and firm and fail to collapse, and airways contain variable amounts of pink, foamy liquid. Histologically necrosis of alveolar and terminal bronchial epithelium is observed, with extensive filling of the alveolar spaces with neutrophils, macrophages, hemorrhage, protein-rich edema fluid, or hyaline membranes. Occasionally, microthrombi can be detected in interstitial capillaries. In more chronic cases diffuse proliferation of cuboidal pneumocytes and fibroblasts and beginning fibrosis may be present.398,400-402
Diagnosis
The diagnosis can be established if the onset of the respiratory distress is acute, PaO2/FiO2 is <300 (PaO2 of <63 mm Hg on room air) with little response to O2 supplementation, the correspondent radiographic or ultrasonographic changes are observed, clinical signs of cardiac disease are absent, and the extent of the respiratory distress cannot solely be attributed to a primary disease process (e.g., almost complete destruction of the pulmonary parenchyma by R. equi abscesses). Presence of a primary disease process such as bacterial or viral pneumonia does not rule out the concurrent presence of ALI or ARDS.
Treatment and Prognosis
In human medicine, mechanical ventilation using low tidal volumes (6 mL/kg) and some form of positive end-expiratory pressure is the treatment of choice.407 Because mechanical ventilation, with the exception of rare circumstances,408 is possible only in neonatal equine patients, the most important treatment remains intranasal insufflation of humidified oxygen via unilateral or bilateral intranasal cannulas at high flow rates (10 to 30 L/min). Large-bore tubing systems minimize the resistance to airflow, and FiO2 as high as 70% has been measured at the carina in adult horses using bilateral flow rates of 15 L/min (combined 30 L/min).409 Alternatively, use of intratracheal O2 insufflation has been described, achieving results similar to high bilateral intranasal flow rates.410 Antiinflammatory treatment is also essential; although the use of corticosteroids in human ALI and ARDS is controversial; a recent small study reported reduced mortality and decreased need for ventilation in the group treated with low doses of corticosteroids (2 mg of methylprednisolone per kilogram IV loading dose followed by 2 mg/kg/day IV divided in four treatments).411 Considering that ventilation is rarely an option and that in two reports all but two foal surviving ALI and ARDS received corticosteroids, anecdotal evidence is in favor of steroid administration. IV prednisolone sodium succinate or methylprednisolone at 1 to 2 mg/kg/day divided in two to four doses may be useful. Antimicrobial treatment, if required, should be directed against the underlying disease process. Bronchoconstriction is not a prominent feature of ALI and ARDS, and bronchodilators are therefore of limited use but may be beneficial in individual cases. As described earlier, clearance of edema fluid from the lungs depends on active transepithelial ion transport, a process that can be stimulated by β2-agonists such as salmeterol, terbutaline, and epinephrine. There is some evidence in experimental models of ALI that β2-agonists delivered into the airways reduce pulmonary edema by attenuating pulmonary vascular injury and upregulating fluid clearance from the air spaces.412 Care should be taken when administering bronchodilators as they can induce sudden worsening of ventilation-perfusion mismatch and can lead to acute decompensation of the severely hypoxic patient. Judicious IV fluid therapy may be necessary in dehydrated patients. The increased permeability of the pulmonary capillaries makes capillary hydrostatic pressure the main determinant of pulmonary edema formation, and overhydration or rapid changes in circulating blood volume should be avoided. On the other hand, suboptimal hydration status decreases cardiac output and worsens oxygen delivery to the peripheral tissue. Measurement of central venous pressure may aid in determination of the patient’s hydration status. If foals are too depressed to nurse or eat, nutritional support is essential.
Several other treatments have been suggested in human medicine, including vasodilators such as inhaled nitric oxide or prostacycline, surfactant to reduce surface tension, and scavengers of reactive oxygen species (tocopherol, ascorbic acid); however, none has been proven to increase survival despite the fact that individual treatments improved pulmonary function.396 Considering that, in contrast to human medicine, most equine patients die as a direct result of respiratory failure and severe hypoxemia, some of those therapeutic options may benefit the equine patient.
Based on the limited information available, the prognosis for survival and future athletic performance is guarded, with survival rates ranging from 60% to 69% in two clinical reports.401,402 Nevertheless, survival and successful careers as racehorses have been reported even in severely affected animals.402
INTERSTITIAL PNEUMONIA
PAMELA A. WILKINS
KURT WILLIAMS
FABIO DEL PIERO
Interstitial pneumonia is an uncommon cause of acute or chronic disorders of the lower respiratory tract of horses.413-419 However, because of the severity of the process, it becomes important to recognize and definitively diagnose this entity as early as possible in its clinical course. The term interstitial pneumonia defines a number of diseases that are chronic and progress to pulmonary fibrosis. The course is insidious and morphologically characterized by alveolar structural derangements that lead to loss of functional gas exchange units of the lung and altered mechanical properties of the lung, characterizing the pneumonia as a restrictive lung problem.
Etiology
Multiple agents have been implicated in the genesis of interstitial pneumonia in animals, but fewer than 20 have been confirmed in horses (Box 31-2). Chief among these are infectious agents and ingested toxins. Frequently the causative agent cannot be identified, because of the insidious nature of the process, and the final diagnosis is idiopathic interstitial pneumonia. The lung responds in a rather stereotypic manner to injury, and our limited ability to identify infectious, toxic, and immunologic causes frequently hinders our current ability to make an accurate identification of a specific cause. All efforts should be made to identify a causative agent early in the course of the disease, but the practitioner needs to be aware that treatment frequently will be nonspecific and supportive.
Box 31-2 Causes of Interstitial Pneumonia in Horses
ACUTE
Infections (systemic viral, bacterial, parasitic)
Ingested toxins or precursors
Perilla mint
(Perilla frutescens), crofton weed
(Eupatorium adenophorum), Crotalaria species,
Senecio species
Hypersensitivity
Acute hypersensitivity pneumonitis
Endogenous metabolic or toxic conditions
Shock—particularly endotoxic (ARDS)
Disseminated intravascular coagulation (DIC)
Idiopathic or cryptogenic
CHRONIC
Infections (systemic viral, bacterial, parasitic)
Inhaled inorganic dust (pneumonoconioses)
Hypersensitivity
Hypersensitivity pneumonitis
Ingested toxins or precursors
Perilla mint
(Perilla frutescens), crofton weed
(Eupatorium adenophorum), Crotalaria species,
Senecio species
Collagen-vascular disorders
Idiopathic or cryptogenic
INFECTIOUS AGENTS
Infectious causes of interstitial pneumonia in horses and foals include viral, bacterial, parasitic, protozoal, and fungal agents. Typically the pneumonia is acute and severe, characterized by severe damage to the lung parenchyma (alveolar region).
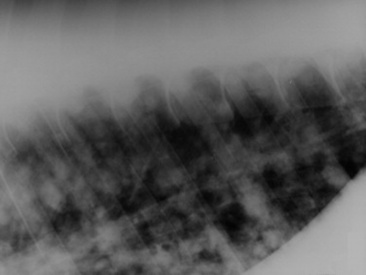
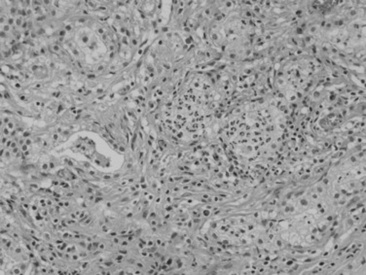
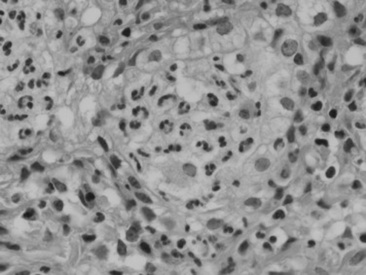
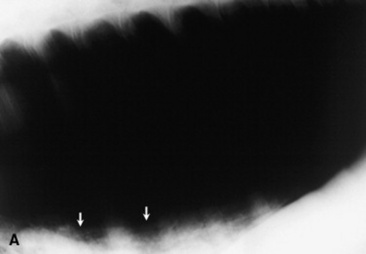
Viral agents are frequently implicated or suspected but rarely identified by the usual serologic, histopathologic, and virus isolation methods. The advent of more sensitive and specific techniques, such as in situ PCR and monoclonal antibody IHC, may partially resolve the current diagnostic challenge. Most recently, a novel gamma-EHV, EHV type 5 (EHV-5), has been found associated with a nodular form of interstitial pneumonia of horses characterized by pulmonary interstitial fibrosis, suggesting that these rather unusual cases may have an underlying infectious cause.420 This disease has been termed equine multinodular pulmonary fibrosis by the authors describing the pathology of the disease. Horses affected generally have a history of fever, cough, and weight loss, accompanied by exercise intolerance and finally respiratory distress. They can be acutely affected or have a more chronic progressive problem. Typical radiographs reveal multiple discreet to coalescing nodular densities overlying a diffuse interstitial pattern (Figs. 31-31 and 31-32). Histologic appearance is diagnostic of this disease (Figs. 31-33 and 31-34). EHV-5 has been identified in both BALF and lung biopsy samples from affected patients by both PCR assay and IHC.
In some horses with bronchointerstitial pneumonia of unknown cause, bacterial agents have been isolated from the lung. The usual distribution of bacterial bronchopneumonia in the horse is cranioventral, whereas the distribution in interstitial pneumonia is diffuse. In the latter cases the bacteria are most likely opportunistic pathogens and do not represent the primary causative agent. An exception is R. equi pneumonia of foals, which can cause an ARDS in older foals. R. equi has been cultured from foals with severe, acute bronchointerstitial pneumonia with a diffuse pulmonary distribution.416 Interstitial pneumonia associated with P. carinii has been described in the foal, and Mycoplasma species has been isolated from the respiratory tract of adult horses.421 The significance of the Mycoplasma isolates remains a matter of debate. P. carinii pneumonia is thought to occur primarily in immunocompromised foals as a complication of some other serious disease, such as infectious pneumonia or SCID. It is characterized by plasmacytic lymphocytic interstitial pneumonia with flooding of alveoli with foamy acidophilic material.
Parasitic pneumonia, an uncommon cause of chronic bronchointerstitial pneumonia, usually occurs in young foals secondary to migration of Parascaris equorum larvae through the pulmonary parenchyma.
INGESTED CHEMICALS
Ingested chemicals rank second only to infectious agents as potential causes of interstitial pneumonia in horses. Ingestion of pyrrolizidine alkaloids from a variety of plants (mostly genera Crotalaria, Trichodesma, and Senecio) can cause interstitial pneumonia in horses.422 This toxicity is associated with production of a toxic metabolite that is activated in the liver then circulates to the lung. The toxic alkylating agents damage capillary endothelial cells, although the amount of alkaloid required to damage the lung is generally less than that required for hepatotoxicity. Crofton weed (Eupatorium adenophorum), a poisonous plant found primarily in Australia and Hawaii, produces interstitial pneumonia in horses. Toxicity is associated with ingestion of the flowering plant, but the nature of the toxin is not known. Perilla ketone, derived from the plant Perilla fructans, produces acute respiratory distress with a week of ingestion in ponies. The lesions include diffuse alveolitis and type II pneumocyte proliferation with sparing of the bronchioles. Toxicity depends on additional metabolism of the 3-substituted furan by the mixed function oxidase system, which occurs directly in the lung of the horse.
INHALED CHEMICALS
Direct pulmonary injury by inhaled chemicals is an uncommon cause of interstitial pneumonia in horses. In people this type of pneumonia is primarily related to occupational exposure. Smoke inhalation causes acute, diffuse interstitial pneumonia in horses, frequently followed within a few days by opportunistic bacterial pneumonia. Oxygen toxicity can theoretically produce interstitial pneumonia and alveolar type II cell proliferation. This problem is more likely to be seen in neonatal foals mechanically ventilated with increased levels of oxygen (FiO2 > 50%) for several days, although this may in fact be a form of ventilator-associated lung injury (VALI) associated with mechanical stretch of the airspaces. Damage is thought to be due to production of reactive oxygen metabolites, which attack a lung that may already have been injured by barotrauma resulting from a ventilator-driven increase in airway pressure. Agrichemicals or herbicides, such as paraquat, may cause acute interstitial pneumonia (AIP) in horses and should be considered in horses with a history of possible exposure. Silicosis is a specific chronic granulomatous pneumonia of horses associated with inhalation of silicon dioxide crystals. This syndrome has been described in horses originating from the Carmel Valley region of California. The inhaled particles are ingested by alveolar macrophages and result in lysis of the macrophage, chronic alveolitis, and fibrosis. Multiple granulomas are present, and submicron intracytoplasmic crystalline particles can be identified in macrophages.
HYPERSENSITIVITY REACTIONS
In the most specific sense, hypersensitivity pneumonitis refers to pulmonary disease caused by inhalation of organic antigens. Lymphocytic, plasmacytic bronchitis and bronchiolitis, combined with lymphocytic interstitial pneumonia, characterize the disease in horse lung. Granuloma formation and fibrosis can be observed. Chicken dust and fungi have been implicated as causes of severe, chronic bronchointerstitial pneumonia, but the syndrome itself is quite rare.
ENDOGENOUS METABOLIC AND TOXIC CONDITIONS
Various conditions cause acute pulmonary injury with inflammatory edema or severe alveolar wall damage and serofibrinous exudation similar to that described for AIP. Acute uremia, shock, burns, and trauma can produce an acute pulmonary injury termed acute lung injury or acute respiratory distress syndrome, depending on severity. Although endotoxin does not directly injure the lung, endotoxemia in the horse initiates inflammatory and metabolic cascades that can lead to pulmonary injury. Activation of these pathways produces vasoactive and chemoattractant molecules that increase vascular permeability, activate complement, produce proinflammatory cytokines, and release neutrophil enzymes that can adversely affect the lungs of horses. Horses as a species are quite sensitive to the negative effects of endotoxemia, and their lungs are particularly sensitive, perhaps because of the presence of intravascular macrophages, which further amplify the inflammatory cascade. Both ALI and ARDS have recently been defined for veterinary patients, although the definition has not been published as of the date of this writing.
Pathophysiology
Interstitial pneumonia progresses through four phases. During the first, the initial insult causes parenchymal injury and alveolitis. This is followed by a proliferative phase characterized by cellular and parenchymal alterations in tissues of the lung. Chronic cases progress to the development of interstitial fibrosis, and the final stage results in end-stage irreparable fibrosis of the lung.
The structural changes that occur in the lung reduce the number of functional alveoli, adversely affecting ventilatory function of the lung and altering ventilation-perfusion relationships. Reduced lung compliance is associated with the loss of distensible alveoli, the presence of pulmonary edema, and fibrosis. Total and vital lung capacity are decreased in association with the loss of functional gas exchange units and reduced lung compliance. The work of breathing is increased, resulting in exercise intolerance and difficulty in breathing. Pulmonary hypertension and cor pulmonale may be complications of interstitial pneumonia and fibrosis. Although the origin of pulmonary hypertension is unclear, hypoxic vasoconstriction and generation of vasoactive compounds (such as endothelin-1) that alter pulmonary vascular resistance acutely, and vessel anatomy chronically, may play a role.
Clinical Signs
Horses affected with interstitial pneumonia frequently have fever, cough, weight loss, nasal discharge, exercise intolerance, severe dyspnea, cyanosis, and a restrictive breathing pattern. A “heave line” is frequently present; nostril flare and an anxious expression are usual. The history can be acute or chronic. Although affected foals are frequently depressed and anorectic, adults may be bright and alert with a variable appetite. The disease proceeds toward death in many cases, with progressive respiratory compromise, although some patients may also slowly improve with time. More than one foal at a farm may be affected.
Diagnosis
In older horses the primary differential diagnosis of heaves may be excluded by the leukocytosis and hyperfibrinogenemia that commonly occur in horses with interstitial pneumonia and fibrosis but do not occur in horses with heaves. However, these abnormal features are common in horses with infectious bronchopneumonia, and thoracic radiography is of paramount importance in the establishment of a definitive diagnosis. Typically, thoracic radiographs reveal extensive interstitial and bronchointerstitial pulmonary patterns.423 Nodular infiltrates may be present, either large or miliary, but always diffusely distributed.
Culture of transtracheal or BAL aspirates often yields no significant growth of bacterial or fungal pathogens. Cytologic evaluation of tracheal or BALF shows increased numbers of neutrophils and macrophages. If P. carinii is involved, BALF may reveal trophozoites or intracystic bodies with special stains, such as toluidine blue or methenamine silver. Using PCR, EHV-5 has been found in BALF from horses with EMPF.
Histologic examination of a transthoracic lung biopsy specimen is the definitive diagnostic test for chronic interstitial pneumonia and fibrosis and for EMPF. Care must be taken to ensure the biopsy is obtained from a representative area, and we have found ultrasound guidance useful. Clinically important complications from this technique are uncommon but can occur. Biopsy can both define the causative agent and confirm the clinical diagnosis in EMPF when the sample is evaluated for EHV-5.
Additional diagnostic procedures could include arterial blood gas analysis, abdominocentesis and thoracocentesis to rule out metastatic neoplastic disease, PFT, viral isolation, serologic testing for antibody to fungi and chicken serum if hypersensitivity pneumonitis is suspected, and immunohistochemical evaluation of lung tissue for suspected infectious agents. A complete cardiac evaluation should also be conducted, as pulmonary hypertension and cor pulmonale are not uncommon sequelae.
Treatment
Treatment of these cases is often unrewarding. Therapeutic goals are treatment of any underlying or secondary infection; suppression of inflammation; maintenance of tissue oxygen delivery within appropriate limits; relief of any associated bronchoconstriction; and prevention or treatment of complications. Environmental control, with appropriate temperature and humidity control and good ventilation, is beneficial.
Parenteral corticosteroid therapy is the mainstay of treatment, with early and aggressive therapy providing the best long-term outcome, particularly in foals. In one report of 23 foals affected with acute bronchointerstitial pneumonia, 9 of 10 treated with corticosteroids survived, but none of those not receiving steroid treatment lived.
Broad-spectrum antimicrobial treatment should be instituted initially, particularly in foals, as described for the treatment of infectious bronchopneumonia. The choice of antimicrobial agent and duration of therapy should be dictated finally by the culture and sensitivity results from the TTA and by the patient’s clinical course. Anecdotally, horses with EMPF have responded to treatment with acyclovir and corticosteroids. The course of therapy is generally prolonged (6 to 12 weeks); clinical and radiographic improvement has been noted in a few cases to date.
Foals, in particular, and adults with severe respiratory distress may benefit from nasal insufflation of humidified oxygen, with flow rates of 10 L/min for foals and 15 L/min in adults. If necessary, as determined by persistent hypoxemia in the face of intranasal insufflation at the rates given, a second nasal canula can be placed in the opposite nostril to increase the FiO2. Care must be taken to avoid obstruction of the nasal passages. Alternatively, intratracheal or transtracheal insufflation can be considered to further increase FiO2 and improve oxygenation.
Systemic bronchodilator therapy may or may not be indicated in these cases. If used, bronchodilators may worsen ventilation-perfusion inequalities. Therefore bronchodilator therapy should be accompanied by supplemental oxygen, and the effects should be monitored with serial blood gas measurements and discontinued if hypoxemia worsens. Nebulized or aerosolized bronchodilator therapy may be more judicious, and beneficial effects are evident in some foals with respiratory distress. Aminophylline and theophylline should not be used, owing to their narrow therapeutic range. Furosemide may be appropriate for its bronchodilator effect and its effect on reducing pulmonary artery pressure, particularly if cor pulmonale develops.
Prognosis
The prognosis for horses with interstitial pneumonia is uniformly poor to guarded. Affected foals, treated early and aggressively with corticosteroid and antimicrobial therapy, have the best outlook for life. The disease is usually progressive in adults and eventually results in the demise of the horse, although the occasional horse recovers sufficiently to return to previous performance levels. A fair number of adult horses, with continuous intense management, will live for a period of time but will be severely compromised, which limits their usefulness. Exceptions to the poor prognosis may be seen in cases of P. carinii pneumonia in foals if they are treated early and aggressively and possibly also in cases of idiopathic interstitial pneumonia or EMPF in adult horses that are treated early with corticosteroids and antiviral drugs. A trial of treatment for peracute interstitial disease for 48 hours is warranted, and patients with chronic interstitial pneumonia should be treated for a minimum of 6 weeks before the possibility of recovery is discarded.
EQUINE RESPIRATORY VIRUSES
GABRIELE A. LANDOLT
D. Paul Lunn
Viral respiratory infections in horses are major causes of respiratory disease, resulting in impaired pulmonary function and reduced performance. Although vaccines are available for many of the causative agents, the respiratory viruses have remained a major concern in the horse industry. Infection with equine respiratory viruses can lead to serious respiratory illness with potentially fatal consequences. Yet the incidence and severity of viral respiratory disease may vary from year to year and are dependent on the attack rate, virulence of the circulating virus, infectious dose, management and environmental conditions, and degree of immunity of individual animals in the population.
With the exception of HEV infection, in a group of susceptible horses equine viral respiratory infection generally is characterized by the rapid spread of an acute, febrile respiratory disease accompanied by a dry cough and nasal discharge. Therefore a presumptive diagnosis of viral respiratory disease can often be made based on history and clinical signs. However, to confirm and differentiate among the potential causative agents, laboratory diagnosis is required.
Diagnosis of Equine Respiratory Viruses
The basic approaches for the laboratory diagnosis of equine viral respiratory disease are the isolation of the causative agent (virus isolation), demonstration of the virus or viral product in clinical specimens (detection of virus particles by electron microscopy; detection of viral antigens by immunofluorescence, ELISA, or immunoperoxidase; detection of the viral genome), and the detection of virus-specific antibodies (serology). Because each of these methods has its merits as well as disadvantages (e.g., lack of sensitivity, long turnaround time, high costs), it may be necessary to combine several of them to identify the causative agent accurately and rapidly. The World Organisation for Animal Health (OIE, www.oie.int) publishes the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, which provides an in-depth review of current diagnostic testing strategies.
VIRUS ISOLATION
Virus isolation from clinical samples is crucial for epidemiologic investigation and for vaccine production and generally is carried out in cell culture. The notable exception is equine influenza virus, for which embryonated chicken eggs have remained the culture system of choice. As virus shedding in secretions of the respiratory tract is greatest within the first few days after infection, the best results for virus isolation are often achieved by culturing nasopharyngeal swabs collected in the first 24 to 48 hours after onset of clinical disease. The duration of nasal virus shedding is often brief in partially immune animals, so it may prove useful to sample immunologically less protected individuals in a group (e.g., younger horses, poorly vaccinated or unvaccinated animals) to increase the likelihood of demonstrating infectious virus. Nasopharyngeal or nasal passage samples are best collected using polyester-tipped swabs instead of cotton swabs because certain viruses (e.g., influenza A virus) have been found to adhere to the cotton fibers. The swabs should be placed in a sterile viral transport medium and kept on ice until further analysis.
ANTIGEN DETECTION
The most important advantages of diagnostic tests aimed at the detection of viral antigen over traditional virus isolation are the faster turnaround time as well as the ability to detect virions that have lost their infectivity. Immunofluorescence (fluorescent antibody [FA] test) is based on the detection of virus-infected cells employing virus-specific fluorochrome-labeled antibodies. The technique is used to detect viral antigen in a broad range of clinical samples (e.g., frozen sections of tissues, 132 tissue imprints, cells obtained from nasal scrapings or tracheal washes) and has been shown to be highly sensitive. A number of antigen-capture ELISA-based assays have been developed for the detection of equine respiratory viruses.424-428 Frequently a solid-phase system is used for virus detection. Specific antibody is first adsorbed to the surface of a microtiter plate, and the sample containing the suspected virus is then added. If the virus is present, it binds to the adsorbed antibody. After washing, specific enzyme-labeled antibodies are added. Detection of virus in the sample is achieved by addition of the enzyme substrate, which results in a color reaction. Commercially available ELISA-based test kits (e.g., Flu OIA assay, Biostar, Boulder, Colo.; Directigen Flu-A assay, Becton Dickinson Microbiology Systems, Cockeysville, Md.) have been validated for the rapid, stallside detection of equine influenza virus. Many investigators have found these commercial diagnostic kits to be highly specific and rapid, and their sensitivity approaches that of egg isolation.429-433 Since the discovery of the PCR the technique has found wide application in diagnostic laboratories. Through the choice of appropriate oligonucleotide primers, a selected region of the viral genome can be amplified. PCR-based assays are extremely sensitive and theoretically can detect a single copy of the target nucleic acid in a sample.434-436 Yet, owing to the assay’s high sensitivity, the greatest challenge facing the diagnostic application of PCR is the production of false-positive results. Nevertheless, PCR-based techniques offer a more sensitive tool for the diagnosis of equine respiratory viruses than conventional techniques such as culture or immunoassays. To increase the diagnostic capacity of PCR, multiplex PCR assays have been developed. By including more than one pair of primers in the reaction mixture, these assays are designed to simultaneously detect a panel of respiratory pathogens. For example, multiplex PCR assays designed to detect nucleic acid of influenza A and B viruses, respiratory syncytial virus (RSV), adenovirus, and parainfluenza virus have been developed for screening of human clinical samples.437-440 In equine diagnostics, assays capable of simultaneously detecting and differentiating EHV-1 and EHV-4441-443 as well as EHV-2 and EHV-5444 have been published. More recently, real-time PCR–based assays have been used for the detection and quantification of equine respiratory pathogens.445-447 By employing a target-specific fluorescent probe, real-time PCR–based assays eliminate the need to distinguish the target by PCR fragment size on electrophoresis or hybridization with probes post-PCR. The advantages of real-time PCR include high specificity, sensitivity, and a rapid turnaround time.
ANTIBODY DETECTION
Detection of virus-specific antibodies has been and continues to be a cornerstone in the diagnosis of equine viral respiratory disease. Detection of specific antibody can provide support that an animal has been exposed to a given antigen. Often, this is used as circumstantial evidence of infectious disease. Commonly used techniques for antibody detection include ELISAs, serum neutralization tests, single radial hemolysis, and hemagglutination inhibition assays. Because most serologic assays are fairly easy to perform and cost-effective and because large numbers of samples can be collected and tested simultaneously, serologic testing is particularly useful for large-scale herd surveillance. Although useful, there are limitations to serologic testing. For example, the presence of antibody indicates exposure to antigen but does not necessarily indicate active infection or disease. If horses have been vaccinated, a diagnosis of acute infection can often be made only by testing paired samples (acute and convalescent) and by demonstrating at least a fourfold rise in antibody titers over a period of several weeks (seroconversion). Because of the inherent variability of immunoassays, paired samples always should be run by the same laboratory. In addition, antibody is not always detected in the presence of active infection. The most common reason for a negative result despite infection is that insufficient time has passed between infection and measurement of an antibody response. Alternatively, immunocompromised horses may fail to produce a detectable antibody response despite active infection.
Treatment
The treatment of horses with viral respiratory disease is primarily symptomatic. It is important to ensure that sick animals are kept in a clean, well-ventilated, and stress-free environment. Morbidity caused by pyrexia and myalgia may be reduced by administering NSAIDs. It is important to monitor affected horses for development of complications such as pneumonia and myocarditis, and animals exhibiting signs of respiratory disease after 10 days should be reevaluated to rule out secondary bacterial infections.
Antiviral drugs, such as amantadine, rimantadine, and acyclovir, have been used for prophylaxis and treatment of horses during outbreaks of equine influenza and EHV-1, respectively. Clinical trials with the influenza virus M2 ion channel blockers amantadine and rimantadine have shown that both drugs appear to be equally effective in reducing the severity and duration of clinical illness.448,449 In addition, oral administration of rimantadine (30 mg/kg q12h) was shown to reduce the virus load in nasal secretion, although the duration of nasal virus shedding was similar to that in the untreated controls.448 However, the therapeutic and prophylactic benefits of M2 ion blocker antiviral therapy during outbreaks of equine influenza may be limited owing to the potential for the rapid development of drug resistance. In vivo and in vitro studies have demonstrated that drug-resistant influenza virus mutants can develop as early as 1 day after the start of treatment.450 Subsequent isolation of the drug-resistant virus from susceptible contacts showing signs of clinical disease indicates that acquisition of drug resistance is not associated with attenuation of the virus.451,452 Finally, treatment of horses with amantadine has also been associated with adverse effects and poor oral bioavailablity.453
Acyclovir (9-[2-hydroxyethoxymethyl]guanine), a chain-terminating nucleoside analog, has been shown to be highly efficacious against herpes simples virus and, to some extent, varicella-zoster virus. EHV-1 was also found to be sensitive to inhibition by acyclovir in vitro and in animal models.454,455 Although there have been no controlled studies reporting on the efficacy of acyclovir in the treatment and prevention of EHV-1 infection in horses, anecdotal reports suggest a potential benefit of acyclovir administration (10 mg/kg PO five times daily) in horses with clinical signs of EHV-1 infection (e.g., myelitis, neonatal infection).456-458 In humans, acyclovir can be administered by injection, as a topical cream, or as an ingestible capsule. It is interesting to note that evaluation of the pharmacokinetics of a single PO or IV dose of acyclovir in adult horses revealed that intragastric administration of the drug was associated with high variability in serum drug concentrations as well as poor bioavailability.459
EQUINE INFLUENZA VIRUS
Equine influenza is the most frequently diagnosed cause of viral respiratory disease of the horse.460,461 It is a highly infectious disease that affects horses, donkeys, mules, and zebras. Outbreaks of a disease resembling influenza were reported as early as 1751; however, the causative agent was not isolated until 1956.462,463
Etiology
Equine influenza viruses are members of the family Orthomyxoviridae, which contains enveloped viruses with segmented, single-stranded, negative-sense RNA genomes.464 The Orthomyxoviridae include five genera: influenza A, B, and C viruses, thogotoviruses, and isaviruses.464 Equine influenza viruses are influenza A viruses. In contrast to influenza B and C viruses, which appear to have a fairly narrow host range, influenza A viruses can infect a wide variety of species (e.g., horses, humans, wild and domestic birds, pigs, sea mammals, dogs, and cats). It has long been recognized that influenza A viruses exhibit partial restriction of their host range,463 indicating that viruses from one host species occasionally can transmit to infect another host. Undoubtedly, the most prominent examples for direct transmission of influenza A viruses among species are the recent infections of humans and cats with the highly pathogenic avian H5N1 viruses.465-468 Although horses have often been regarded as isolated or “dead-end” hosts for influenza,463 events of recent years clearly indicate that the species barrier for viruses jumping either to or from horses is not absolute. For example, the severe influenza epidemic in horses occurring in 1989 in the Jilin and Heilongjiang provinces in the northeast of the People’s Republic of China was caused by a virus with avian-like genetic features, suggesting that the virus had spread directly to horses from birds.469 Another more recent example is the transmission of an equine-lineage virus to dogs in the United States.470 First isolated from greyhound dogs at a racing kennel in Florida in 2004, the equine-lineage virus spread quickly though a substantial portion of the dog population in the United States over the course of 2 years.
Although the viral and host factors that determine the host range of influenza viruses, and the mechanisms by which species barriers are overcome, are not completely understood, a large body of research highlights the importance of the viral hemagglutinin (HA) glycoprotein in determining host specificity.463,471,472 HA serves as the viral receptor-binding protein and is embedded, along with the neuraminidase (NA) and the ion channel M2 protein, in a host cell—derived lipid envelope. Mediated by binding of the HA to sialic acid residues on the host cells, the virion envelope and the host cell membrane fuse. This interaction subsequently promotes the release of the viral genome into the cytoplasm of the host cell. NA is responsible for the cleavage of the α-ketosidic linkage between a sialic acid molecule and an adjacent sugar D-galactose or D-galactosamine. Biologically, NA facilitates the mobility of the influenza virus virion by assisting in the release of the budding virus particle from the host cells.473,474 Both HA and NA are major surface antigens of the influenza virus virion. Partly because of the host immune pressure, these proteins can undergo substantial antigenic variation (antigenic drift), which allows the mutant viruses to escape neutralizing antibodies.
Based on antigenic properties of HA and NA, influenza A viruses are divided into subtypes. To date, 16 HA subtypes (H1 to H16) and nine NA subtypes (N1 to N9) have been described. Although viruses of all 16 HA and nine NA subtypes of influenza A viruses have been isolated from wild waterfowl,475-477 only a limited number of subtypes have been associated with infection of mammals. For example, in humans, only viruses of H1, N2, H3, N1, and N2 subtypes have circulated widely in the population,463,478 and only H1, H3, N1, and N2 subtypes have been consistently isolated from pigs.479-481 In horses, apart from isolated reports of infection with viruses of H1N1, H2N2, and H3N2 viruses (usually associated with human infections),482 equine influenza infections have been caused by viruses of H7N7 (A/equine/1) and H3N8 (A/equine/2) subtypes.462,483,484 Although large-scale serologic surveillance indicates that H7N7 viruses may still be circulating at low levels in the equine population of Central Asia463 and Eastern Europe,462,485 these viruses have not been isolated from horses since the late 1970s.433,475,486 In contrast, despite intensive vaccination programs, equine H3N8 viruses continue to circulate in large parts of the world. Since the 1980s a number of severe, widespread influenza outbreaks in horses have occurred in South Africa,487 India,488 the People’s Republic of China,469,489 and most recently the United Kingdom.490 The causative agent of the South African outbreak was identified as an H3N8 virus, which was most likely introduced by importation of infected horses from the United States or Europe.433,491
Since the early to mid 1980s the equine lineage H3N8 viruses have diverged into two distinct evolutionary lineages: European and American. Although the circulation of both lineages initially centered largely on their geographic origin,492 both lineages now appear to be cocirculating in Europe and, to a considerably lesser extent, in the United States.433,493,494 Continued genetic divergence appears to have resulted in the formation of three American-like lineages with distinct antigenic characteristics (a South American lineage, a Kentucky lineage, and a Florida lineage).493 The rate of genetic diversion of equine viruses is small compared with human and swine influenza viruses, but the sustained genetic evolution continues to have considerable impact in terms of immunization.
Epidemiology
Experimentally, horses of all age-groups are susceptible to infection.495 However, although sporadic infection in foals has been described, there exists only one published report of a widespread outbreak in foals less than 6 months of age.496 The lower incidence of disease in young foals is most likely a result of the presence of maternally derived antibodies. Influenza-specific serum antibody concentration is a highly accurate correlate to protection against infection and disease, and animals with high concentrations of homologous antibody are almost always protected against experimental challenge.497,498 After natural infection, ponies were reported to be protected from infection for 8 months, with partial protection persisting for more than 1 year.499 Although vaccination does not always provide full protection against clinical disease, immunization of horses against influenza has reduced the frequency of disease outbreaks and the frequency and severity of clinical signs when epizootic disease occurs among vaccinated animals.495,500,501
Outbreaks of equine influenza occur most often when susceptible animals are congregated and housed in close contact with one another (e.g., horse shows, racetracks, sale barns). Virus transmission occurs through direct contact with infected animals or via droplet or airborne transmission.502 The virus has been shown to be able to remain infectious for up to 48 hours on dry surfaces (equipment, countertops), but it is capable of surviving for as long as 72 hours in wet environments.503 Anecdotal evidence suggests that the disease can spread in a matter of hours to days among immunologically naïve animals that are kept in close confinement. Such effective spread is enhanced by a short incubation period (approximately 2 days), high concentrations of virus shed in nasal secretions, an explosive cough, and the practice of housing horses in confined spaces.503 In partially immune animals, the spread of disease is often considerably slower and outbreaks may last as long as 3 to 4 weeks.504 After experimental infection, horses typically shed virus for 6 to 7 days.505-508
Pathogenesis
Influenza A viruses replicate and induce pathologic changes throughout the entire respiratory tract, with the most significant lesions occurring in the lower airways. After inhalation of the virus, the virion attaches to and enters the respiratory epithelial cells. After replication and release from the host cell, the virus spreads quickly throughout the respiratory tract. Largely through apoptosis,509-511 influenza virus replication leads to cell death and subsequent loss of the ciliated respiratory epithelium in the trachea and bronchial tree.512 Consequently, tracheal mucociliary clearance is impaired, predisposing affected horses to the development of secondary bacterial infections.483,513,514 In addition, the disruption of the superficial cell layers allows opportunistic bacteria to invade the respiratory epithelium of both the upper and the lower respiratory tract. The combination of reduced airway clearance and erosion of the respiratory epithelium can result in bacterial bronchopneumonia and other complications.512,515 In uncomplicated cases, regeneration of the respiratory epithelium begins approximately 3 to 5 days after onset of illness,515 but resolution of the epithelial damage takes a minimum of 3 weeks.514,516
Clinical Presentation
Clinical signs of influenza virus infection in horses are often seen 48 hours after exposure to the virus. In vaccinated animals the disease is rarely fatal, but deaths have been reported during some epidemics, particularly in donkeys and rarely in neonatal foals.462,517 The typical clinical presentation of equine influenza consists of pyrexia, anorexia, lethargy, nasal discharge, and cough. Pyrexia is often the first symptom present, with body temperatures peaking somewhere between 48 to 96 hours after infection. In some cases a second peak of pyrexia may occur around 7 days after infection. Nasal discharge is often serous in the first few days of illness but may become mucopurulent by 72 to 96 hours after infection. Concurrently a dry, hacking cough develops. Coughing typically persists long after pyrexia and nasal discharge have resolved and may still be present 3 weeks postinfection. Most affected animals are anorectic during the initial pyrexia, and weight loss has consistently been documented in experimental infections. In severe infections, lung sounds may become increased in amplitude and adventitial sounds may sometimes be detected. Uncomplicated cases of equine influenza typically resolve within 7 to 14 days postinfection, but complications of equine influenza virus infections can be severe and include secondary bacterial pneumonia, myositis, myocarditis, and limb edema.484,513,514 It has also been speculated that influenza infection may predispose horses to the development of IAD, RAO, and EIPH.484,512,513 Furthermore, the effects of influenza virus infection can be significantly exacerbated by even moderate exercise, resulting in increased weight loss and other clinical signs.518
Hemographic abnormalities associated with equine influenza infection can include moderate normocytic, normochromic anemia and leukopenia, which may result from both neutropenia and lymphopenia. Monocytosis during early convalescences is a variable finding.
Control
Vaccination against equine influenza virus infection is the foundation of influenza control. Although a wide variety of vaccine formulations, including modified live and recombinant vaccines, are commercially available, inactivated vaccines are the most common type of vaccine used. Many challenge studies have demonstrated the ability of inactivated vaccines to protect against homologous virus challenge.497,498,519,520 However, the value of inactivated vaccines is critically dependent on the quality, quantity, and strain of virus used for vaccine preparation.521
Based on the finding that the H7N7 viruses have not been isolated from horses since the late 1070s,486 the inclusion of A/equine/1 virus is no longer thought to be necessary for equine influenza vaccines. In contrast, the divergence of the H3N8 equine viruses in two distinct lineages has made the inclusion of strains representative of both the American and European lineages a desirable feature of vaccines.433 Sustained genetic evolution of the A/equine/2 viruses will continue to result in failure of killed vaccines, and therefore inclusion of viral antigens representative of contemporary circulating viruses has to remain a priority.
An intranasal, cold-adapted, modified live virus vaccine (FluAvert I.N., Intervet, Millsboro, Del.) has become available in North America. Whereas studies have demonstrated that the vaccine was able to confer protection for up to 12 months after a single administration, only a 6-month claim is made on the product data sheet.508,522 Despite the fact that the vaccine contains only one modified live virus strain, which is based on the H3N8 A/Eq/KY/91 isolate, the vaccine appears to protect against both the European and the American lineages.523 A recombinant canary pox vector-based vaccine (ProteqFlu, Merial) is also available on the veterinary market and has shown excellent performance against recent influenza virus strains.524
Because of the presence of maternal antibodies, recommendations for vaccination generally state not to vaccinate foals younger than 6 months of age.525-527 For inactivated vaccines, initial vaccination should include three doses (even when the data sheet recommends only two initial doses). The timing of the first three doses is critical. The recommended interval between the first and second dose is 3 to 4 weeks. The third dose should be administered when the antibody response to the second dose has waned, which is thought to occur at 3 to 4 months after the administration of the second dose. Regardless of the recommendations provided by the manufacturer, this vaccination regimen is preferred, and contemporary inactivated vaccines are likely to perform well if this approach is taken. After the initial vaccination series, subsequent booster vaccinations should be given at 6-month intervals in high-risk populations. The modified live cold-adapted intranasal vaccine requires only a single initial dose, followed by booster doses at 6-month intervals. If a higher risk of infection is anticipated (e.g., horse show), booster doses may be administered even to well-vaccinated horses 1 to 2 weeks before potential exposure.
EQUINE HERPES VIRUSES
EHV infection is a common infection of equids throughout the world, with at least nine herpesviruses identified to date and with more likely to be identified in future.528,529 The most important EHVs are the Alphaherpesvirinae EHV-1 and EHV-4, which both cause respiratory disease. EHV-1 commonly infects horses, donkeys, and mules and occasionally infects domestic cattle, captive camelids, cervids, and zebras; EHV-4 is restricted to horses.528 Both EHV-2 and EHV-5, which are Gammaherpesvirinae, can be associated with respiratory disease, although their pathogenicity is low.530 EHV-2 is distributed throughout the world and is a ubiquitous infection of the horse; this increasingly appears to be also the case for EHV-5,531-533 which previously had been identified only in Australia and New Zealand.530
Etiology
Both EHV-1 and EHV-4 are members of the Alphaherpesvirinae subfamily of herpesviruses, in the genus Varicellovirus. These two viruses have very similar antigenic, genetic, and pathogenetic characteristics, although they are quite distinct from the major equine gammaherpesviruses (EHV-2, EHV-3, and EHV-5).528 Both viruses are easily destroyed by solvents, detergents, disinfectants, and other common veterinary means of decontamination, but under favorable conditions they can persist in the environment for as long as a month, and in refrigerated liquids for much longer.
Both EHV-1 and EHV-4 have a linear double-stranded DNA genome of 145 and 150 kbp encoding 79 and 80 ORFs, respectively,534-536 which can code for 77 proteins. Amino acid homology of EHV-1 and EHV-4 is 55% to 96%.534 The gene products of EHV-1 and EHV-4 are classified as immediate early, early, or late (which include the major glycoproteins).537-540 One genetic factor that has been identified as being associated with the occurrence of EHV myeloencephalopathy (EHM) is a mutation in the DNApol gene.541 This mutation is present in the large majority, but not all, of the EHM isolates of EHV-1. Infection with viruses with this mutation generated higher levels of viremia in experimental infection,542 but beyond this there is still a limited understanding of the relationship between the mutation and the pathology that results in EHM.
Epidemiology
EHV-1 infection causes significant suffering in horses and extensive economic losses through frequent outbreaks of a range of diseases including outbreaks of respiratory disease, abortion, neonatal foal death, myeloencephalopathy, and chorioretinopathy.528 EHV-4 can cause respiratory disease outbreaks, but only sporadic cases of abortion rather than abortion storms. Infections caused by EHV-1 and EHV-4 are particularly common in young performance horses and typically result in establishment of latent infection within the first weeks or months of life,543 with subsequent viral reactivation causing clinical disease and viral shedding during periods of stress.528 As more sophisticated methods of detecting latent infection with EHV-1 and EHV-4 are developed,544 the detectable prevalence of infection continues to rise, with the implication that EHV-free horses are very rare.
Pathogenesis
The pathogenesis of EHV-1 infection has been comprehensively reviewed.528,545,546 Primary EHV-1 infection occurs at the respiratory epithelium, resulting in erosion of the upper respiratory mucosal surface and viral shedding for 4 to 7 days postinfection; shedding can occur for longer at lower levels, and possibly beyond 14 days. Cell-to-cell spread results in the presence of virus in respiratory tract lymph nodes within 12 to 24 hours postinfection. A lymphocyte-associated viremia is then established and is responsible for the delivery of EHV-1 to the pregnant uterus and other tissues. This viremia can persist for up to 21 days and is a prerequisite for abortion and EHM. In pregnant horses, EHV-1 infection can result in abortion, typically in the last 4 months of pregnancy. There are usually no signs of impending abortion, and the foal is typically expelled in the amniotic membrane having just died from asphyxia owing to placental separation. Some fetuses survive initially but then die as a result of respiratory failure caused by infection of the lungs. The pathology is of infection of endothelial cells of the pregnant uterus causing a vasculitis affecting the small arteriolar branches of the glandular layer of the endometrium at the base of microcotyledons.528 EHV-1 myeloencephalitis results from widespread vascular injury after damage to the vascular endothelium of the blood-brain barrier resulting from viral infection of the endothelium. Vasculitis develops, with or without local hemorrhage and thromboischemic necrosis, in the brain and spinal cord. It is possible that the immunopathologic response to EHV-1 infection of the vascular endothelium of the CNS may play a part in the cause of the thromboischemic necrosis, thereby contributing to the disease process.
Latency is established in both the lymphoreticular system and in the trigeminal ganglion,528 although lymphoid tissues are likely to be the primary home of latent virus.544 Reactivation of latent virus frequently results in reinfection of the upper respiratory tract and further shedding of virus. Latency and reactivation are critical features of the epidemiology of EHV-1 infection, allowing the virus to persist for lengthy periods, and in all likelihood as a life-long infection, in the majority of previously infected horses before reactivating and infecting new susceptible horses or resulting in disease in the latently infected horse itself.
Immunity to EHV-1 and EHV-4 has been extensively reviewed.546 The protective immune response after infection is short lived (3 to 6 months), although this may increase with age and repeated exposure. High titers of virus-neutralizing (VN) antibody before infection can reduce the amount and duration of virus shed from the nasopharynx547 but fail to prevent infection.548 The underlying reason for this failure to prevent infection is likely to be related to the rapid intracellular location of EHV-1,549,550 which permits escape from the effects of VN antibody. However, intracellular virus is susceptible to cytotoxic T lymphocytes (CTLs), which can lyse virus-infected cells. This CTL activity is major histocompatibility complex (MHC) class I restricted and mediated by CD8+ lymphocytes.551 Ponies with high frequencies of CD8+ CTLs show a reduction in the clinical and virologic outcome of infection552 and are protected against abortion.553 Control of EHV-1 infection requires an immune response that not only limits primary infection but also controls reactivation of latent infections.548 Although circulating and mucosal antibodies each play some role in protection,547,554 the most important component of a protective immune response to EHV-1 is the CTL response.553,555,556
An important aspect of EHV-1 pathogenesis and immunity is the capacity of the virus to evade the immune response.557 This may be mediated by downregulation of expression of viral proteins in infected cells558,559 or by downregulation of host MHC-1 expression, thereby reducing antigen presentation to the critical CTL population.560
Clinical Presentation
Both EHV-1 and EHV-4 cause respiratory signs, but signs are more severe for EHV-1, as is the likelihood of viremia. Clinical respiratory disease in susceptible horses has an incubation period of 1 to 3 days. Signs include pyrexia, which is typically biphasic, with a first peak at 24 to 48 hours associated with upper respiratory tract infection and a second peak at 4 to 8 days associated with the development of viremia. Depression and anorexia are often mild, and progressive nasal discharge becomes mucopurulent by day 5 to 7. Conjunctivitis, lymphadenopathy of the submandibular lymph nodes, and edema or vasculitis of the distal limbs are variable findings. Respiratory disease, particularly as an outbreak, is uncommon in horses over 2 years of age, presumably because of recurrent exposure to the virus and development of sufficient immunity to suppress clinical signs of disease.
In neonatal foals infected with EHV-1 in utero, foals may be normal at birth but quickly become affected by weakness, lethargy, and profound respiratory distress. Prognosis is poor for survival. The abortion and EHM forms of EHV-1 are discussed elsewhere under the appropriate system.
The clinical signs resulting from infection with EHV-2 remain poorly described and established, although it may be a cause of keratoconjunctivitis and possibly upper respiratory tract disease in foals and weanlings.530
Prevention
Because of the ubiquitous nature of EHV-1 and EHV-4 infection, elimination of these pathogens from the population is impossible. Strategies must be used to prevent dissemination of the virus from horses undergoing an active infection. Vaccines are an attractive tool for this, but currently there are no products that can provide comprehensive protection against infection and its sequelae. Inactivated EHV-1 vaccines are capable of eliciting circulating VN antibody responses, which can limit respiratory disease and nasal viral shedding, and the use of some of these vaccines has been associated with a reduction in the incidence of abortion storms.546-548561 Vaccines containing higher antigen loads, and typically those licensed for control of abortion, can generate high titered VN antibody responses and even EHV-1-specific IFN-γ responses.561 Such vaccines are likely to outperform other inactivated products with lower antigen loads. The one commercially available modified live EHV-1 vaccine (MLV) generates limited EHV-specific cellular immunity562 and has been demonstrated to reduce respiratory disease and viral shedding.563 The use of current commercial vaccines of proven efficacy is recommended at 6-month intervals for control of respiratory disease and shedding of EHV-1 and EHV-4. Generally, vaccines effective against EHV-1 will cross-protect against EHV-4. There is no evidence that any commercial or experimental vaccine can prevent the occurrence of EHM.
No attempts are made to control infection caused by EHV-2 or EHV-5 with vaccination. However, the proposal that concurrent EHV-2 infection may play a role in the cause of R. equi pneumonia has led some clinicians to attempt vaccination of foals with killed EHV-2 vaccines.564
Biosecurity measures after detection of outbreaks of EHV-1 and EHV-4 should include isolation of affected and in-contact horses for 21 to 28 days. The virus is thought to spread primarily through contact, with a limited aerosol spread compared with influenza virus. Extensive codes of practice have been described for control of spread of EHV-1 in breeding facilities, both in publications528 and in online resources (www.hblb.org.uk).
EQUINE ARTERITIS VIRUS
EVA is a contagious disease of horses caused by equine arteritis virus (EAV).565 The disease was probably recognized for two centuries before the isolation of EAV in 1953.565 Although the vast majority of EAV infections are inapparent or subclinical, occasional outbreaks occur that result in respiratory disease in adults, abortion, and fatal pneumonia in foals. These disease outbreaks have significant costs in terms of animal suffering and economic loss, and the virus now appears to occur worldwide, infecting horses, donkeys, and mules.
Etiology
EAV is an enveloped RNA virus and is the prototype virus of the genus Arterivirus, family Arteriviridae, order Nidovirales.566 The genome has only nine ORFs, encoding five structural proteins, a viral replicase, and two known nonstructural membrane glycoproteins. The GL envelope glycoprotein expresses the major neutralization determinants of the virus.567 Only one serotype has been identified, which is the Bucyrus strain derived from the 1953 disease outbreak of abortion and respiratory disease in standardbreds that first led to the definition of EVA as a clinical viral disease of horses and the identification of EAV as the causative agent.565 Although there is only one serotype, there are extensive variations in antigenic and pathogenic properties in different isolates of the virus, which are accompanied by differences in structural protein genes. The virus is readily inactivated by lipid solvents and disinfectants, although it can survive for years at subfreezing temperatures.
Epidemiology
EAV is spread by the respiratory and venereal routes, and the persistently infected carrier stallion is central to its persistence and epidemiology.565 Outbreaks of EVA frequently follow the importation of carrier stallions, and this has been identified as an increasingly important factor in the spread of this infection throughout the world in recent years. Similarly, virus can be transported and transmitted through infected semen.
Stallions infected by the respiratory route can become carriers in 30% to 50% of cases, shedding for months to years, although some of these horses do spontaneously clear the infection. The carrier state is testosterone dependent, as castration resolves infection. In the carrier stallion the virus spontaneously generates diverse populations of viral variants, which are found in the semen of infected stallions.568 When one of these variants is transmitted to a mare through breeding, or occasionally through semen-contaminated fomites, a new outbreak can occur if the population is susceptible.565 Transmission by the venereal route is associated with infection rates in mares as high as 85% to 100%.566 In this circumstance subsequent spread of the virus is by aerosol and the respiratory route, with large quantities of virus shed for 7 to 16 days.566 During these outbreaks the EAV strain remains stable with minimal genetic change.565 Seroprevalence varies among breeds and among countries and geographic regions; up to 85% of standardbreds but <5% of thoroughbreds are infected in the United States.569 Because there is no variation in breed susceptibility to EVA, this difference is probably a consequence of the number of actively shedding carrier stallions in different breeds. The virulence of strains in specific breeds can vary, however, and the strains shed by standardbreds are typically highly attenuated, causing minimal disease.565
The immune response to infection is strong, leading to protective immunity with a duration of several years.566 Foals born to immunized mares are protected by colostral antibodies for 2 to 6 months.
Pathogenesis
Both venereal and inhalation transmission of the virus occurs in EVA. After respiratory infection the virus invades the respiratory epithelium and infects bronchial and alveolar macrophages, reaching bronchial and other regional lymph nodes by 48 hours postinfection.566 A leukocyte-associated viremia then leads to general distribution of the virus and further replication in the adrenals, thyroid, liver, and testes. The virus localizes to the endothelium and medial myocytes of blood vessels by 6 to 8 days postinfection, with endothelial damage, lymphocytic infiltration, and damage to the elastica interna of arterioles in the respiratory tract and throughout the body. This results in both hemorrhage and edema in affected organs and tissues, which is most severe approximately 10 days postinfection. Virus is eliminated by 28 days postinfection, except in carrier stallions, in which it persists particularly in the ampulla of the vas deferens.566
Clinical Presentation
The great majority of infected horses are asymptomatic. When disease does occur, the clinical signs of EVA can vary from fever and leukopenia to severe disease and death. The incubation period is 3 to 7 days. Each individual may show some of the following signs. Typically the affected animals are pyrexic, with temperatures reaching 40.5° C (105° F) for 1 to 5 days. Anorexia, depression, serous nasal discharge, lacrimation, and coughing may also occur. Edema of the limbs, palpebra, and scrotum has been reported.566
Abortion occurs in the acute or early convalescent phases of infection and can be the only sign of infection. Abortion can occur at 2 to 10 months of gestation, with abortion rates of 10% to 70%. Comingling of mares recently bred by an infected stallion with pregnant mares is a common scenario leading to abortion.
Neonatal foals infected with EAV show severe respiratory signs and high mortality. In foals that survive more than 24 hours, a fever and leukopenia or thrombocytopenia are present. Interstitial pneumonia, lymphocytic arteritis or periarteritis, renal tubular necrosis, and fibrinoid necrosis of the tunic media are seen on pathologic examination. Old, debilitated, or immunosuppressed horse can be similarly predisposed to severe EVA.565
Prevention
Both a modified live and a killed vaccine against EVA have been developed in recent years.565,566 The modified live vaccine provides complete or partial protection against the clinical signs of EVA, but viral replication still occurs after challenge. The duration of vaccinal protection appears to be as long as 2 years, although annual revaccination of breeding stallions is recommended before the start of the breeding season. The modified live vaccine has been used successfully to limit outbreaks of infe ction in race courses after initial diagnosis.566
The most important steps in EVA control are the identification of carrier stallions, the prevention of the development of the carrier status in future stallions through vaccination of all colts with breeding potential in the first year of life, and annual revaccination thereafter.566 Mares being bred to carrier stallions should be vaccinated against EVA. Fresh and frozen semen from carrier stallions is also infective, and EAV-negative mares receiving infected semen should be vaccinated. Extensive codes of practice have been produced by both the USDA (www.aphis.usda.gov) and the Horserace Betting Levy Board (www.hblb.org.uk) to assist in control of EVA.
EQUINE RHINITIS VIRUS
Etiology
Equine rhinitis viruses (formerly equine rhinoviruses) are a known cause of clinical and subclinical respiratory infections in horses worldwide. The viruses are classified as members of the family Picornaviridae, which includes small (“pico”), nonenveloped, single-stranded RNA viruses. Among the equine rhinitis viruses, at least two serotypes have been isolated from horses: equine rhinitis A virus (ERAV) (formerly equine rhinovirus 1 [ERV-1]) and equine rhinitis B virus (ERBV) (formerly equine rhinovirus 2 [ERV-2]).570,571
Considerable uncertainty has surrounded the classification of the equine rhinitis viruses. Because the illness caused by these viruses resembles the common cold brought about by rhinoviruses in humans, they initially were included in the genus Rhinovirus. However, subsequent studies demonstrated that ERAV differed from other members of the Rhinovirus genus as well as from ERBV.571-574 For example, whereas rhinoviruses generally demonstrate a narrow host cell range in vitro and in vivo, ERAV is able to infect and replicate in a broad range of cell types and host species. For instance, intranasal inoculation of a human volunteer with ERAV resulted in pharyngitis, lymphadenitis, fever, and viremia.575 Furthermore, the presence of systemic signs and viremia during ERAV infection, the occurrence of persistent infections, and the physical properties of the virus are all more reminiscent of foot-and-mouth disease virus (FMDV). Based on these characteristics, along with similarities in protein sequences, ERAV was placed alongside FMDV in the Aphthovirus genus of the picornavirus family. Subsequently, ERBV has been reclassified as the sole member of the new genus Erbovirus.
EPIDEMIOLOGY
First isolated from horses in the United Kingdom,576 ERAV infection occurs worldwide and is a recognized cause of acute respiratory disease.570,577-579 The incidence of neutralizing antibody to ERAV varies according to the age of the horses, and most horses appear to become infected during their second year of life. In a study by Studdert and Gleeson, only 16% of horses between the ages of 6 and 12 months were seropositive.573 However, studies conducted in older horses found that a high percentage of 5-year-old horses have antibodies to equine rhinitis virus.577,580 As with many other respiratory pathogens, young horses entering a large population and in frequent contact with other horses are most susceptible to infection.577 In addition, there appears to be a seasonal distribution, with most of the ERAV infections occurring in late winter and spring.581,582 Stress (e.g., transportation and training) and concurrent disease resulting in immunosuppression may predispose to infection.583 The virus is fairly resistant to inactivation and is thought to survive in the environment for a considerable amount of time. As for human rhinoviruses, virus spread occurs most likely through direct and indirect contact (fomites).584
In contrast to ERAV, the prevalence and clinical significance of ERBV are less defined. A recent study found that 24% of 258 horse sera tested were seropositive for ERBV, indicating that this virus may also be prevalent in the horse population.585 In fact, the results of a recent study suggest that ERBV may play an important role as a causative agent of clinical respiratory disease.586 However, although ERBV was recovered from a large number of horses with acute respiratory disease, only a small percentage of animals demonstrated a concurrent rise in antibody titers for paired sera.
Clinical Presentation
Although ERAV is generally considered a respiratory pathogen, the clinical signs associated with infection may be quite variable. After experimental infection with ERAV, horses may develop pyrexia, anorexia, nasal discharge, pharyngitis, and lymphadenitis involving at least the submandibular and pharyngeal lymph nodes.587 On rare occasions laryngitis or mild bronchitis can develop.580 Based on serologic studies, subclinical ERAV infection may also occur and may be important in the dissemination of the disease.573,581,584,587,588
An important distinction of ERAV compared with other members of the genus Rhinovirus (e.g., human or bovine rhinoviruses) is the finding that horses infected with ERAV may develop viremia. On average, viremia lasts 4 to 5 days and the disappearance of virus from the blood coincides with the appearance of neutralizing antibodies.587 Although the relationship between antibody levels and protection against disease has not been established, immunity is thought to be solid, and antibodies persist for a long time.584
Infection with ERAV may be accompanied by long-term urinary and fecal shedding as well as viral persistence.587 The recognition of the potential for ERAV persistence has important implications for disease diagnosis. Highly sensitive diagnostic methods (e.g., IFA or RT-PCR) may be able to detect persistent ERAV in horses suffering from respiratory disease, which may not be associated with the observed illness. This notion is underlined by the recent finding that the ERAV genome was detected by RT-PCR in nasopharyngeal swab samples obtained from two clinically healthy horses 1 year after acute ERAV infection.571
Control
There is currently no vaccine available to control ERAV infection. A recent study by Warner and co-workers demonstrated that the capsid protein VP1 participates directly in viral receptor binding.589 VP1 is a major surface antigen and therefore may serve as a potential target for vaccines.
EQUINE ADENOVIRUS
Etiology
Adenoviruses (family Adenoviridae) are a group of nonenveloped, icosahedral DNA viruses. Whereas most vertebrate species harbor an adenovirus, the viruses are highly species-specific. In humans, adenoviruses are extremely prevalent and represent a significant cause of acute respiratory, ocular, and GI disease. Infections of immunocompetent individuals usually stay localized and often take a mild or subclinical course.590 Similarly, equine adenoviruses have been isolated from clinically healthy adult horses591 and foals.592,593 Equine adenovirus (EAdV)–1 has been implicated as the cause of acute respiratory tract disease594,595 and conjunctivitis, as well as a progressive, fatal pneumonia in Arabian foals with SCID.596-598 A second serotype, EAdV-2, has been incriminated as a cause of intestinal infection599,600 and diarrhea in immunocompetent foals.601,602
Epidemiology
First isolated in 1969 from a foal with respiratory disease,603 EAdVs have a worldwide distribution.604 Although serologic evidence from Australia, the United Kingdom, the United States, the Netherlands, and Nigeria indicates that adenovirus infection occurs at a high frequency in the general horse population,605-608 the role of EadV in respiratory disease in adult horses remains unclear. In this regard it is interesting that a number of studies found no apparent association between clinically apparent respiratory disease in young adult horses and infection with adenovirus, as diagnosed by subsequent seroconverison.588,609,610
In contrast, fatal and nonfatal adenovirus pneumonia has been well documented in foals, particularly of the Arabian breed. Fatal respiratory disease is predominantly seen in foals with SCID. Other foals may develop respiratory disease but usually do not succumb to it.
In general, adenovirus transmission is thought to occur via direct contact with secretions from infected animals or indirectly via fomites or infected premises. In adult horses the virus can persist in the upper respiratory tract, and these animals can act as a reservoir host for the virus. Adenoviruses are highly stable and have been found to remain infectious at 4° C for up to 1 year.
Clinical Presentation
The clinical signs recorded after experimental intranasal infection of a thoroughbred yearling were confined to a serous nasal discharge that lasted from 4 to 12 days postinoculation. Peak levels of serum antibodies were found at 13 days after inoculation and dropped by approximately 2 months after experimental infection.611
In foals, clinical signs generally occur at 10 to 35 days of age.612 After experimental inoculation at 24 hours of age, clinical signs appeared 3 to 5 days after infection and consisted of intermittent pyrexia, nasal and ocular discharge, tachypnea, and cough. All foals recovered by day 10 postinoculation. The majority of foals developed a mild and transient lymphopenia that was followed by lymphocytosis. Typically, about 25% of infected foals also develop diarrhea.612 Gross pulmonary lesions found in animals euthanized at 14 to 21 days postinoculation included atelectasis involving up to 25% of the pulmonary parenchyma and suppurative bronchopneumonia. Histologically, swelling and hyperplasia of the airway epithelium, as well as intranuclear inclusion bodies in the respiratory epithelial cells, were consistent findings up to 11 days postinfection.613
Foals with SCID show a progressive decline in health despite intensive therapy. At necropsy, conjunctivitis, rhinitis, tracheitis, and bronchopneumonia are present. Intranuclear inclusion bodies are readily demonstrated in the respiratory epithelial cells. Areas of interstitial pneumonia surround affected bronchi. Acute pancreatitis characterized by inclusion bodies in acinus and ductal cells, as well as adenoviral sialoadenitis involving the labial and parotid salivary gland, can often be demonstrated.600,614
Control
No vaccine is available to control adenovirus infection in horses. However, except in immunocompromised animals, the virus appears to be of little clinical importance.
HENDRA VIRUS
In August 1994, two horses died suddenly on a property in Mackay, Queensland, Australia. Ten days after the death of the second horse, the owner was admitted to the hospital with meningitis. One month after the incident in Mackay, 13 horses housed at a stable in Hendra, Queensland, died or were euthanized owing to severe respiratory illness. Subsequently the trainer and one of the stable workers developed an influenza-like illness, to which the trainer succumbed 6 days later. Potential diagnoses included African horse sickness and equine influenza, although they were exotic to Australia. Yet, during postmortem examination, a novel paramyxo-like virus was isolated from tissues of the affected horses and the trainer.615 The virus, first referred to as equine morbillivirus, was subsequently named Hendra virus.
Etiology
HeV is a member of the genus Henipavirus (which also contains Nipah and Menangle viruses) within the Paramyxoviridae virus family. Paramyxoviruses, like orthomyxoviruses (see discussion of equine influenza), are enveloped viruses that contain a negative-sense, single-stranded RNA genome. HeV has unique morphologic characteristics that are useful for diagnostic purposes.616 Electron microscopy reveals pleomorphic enveloped particles that contain a helical nucleocapsid that is characteristic for paramyxoviruses. The envelope is covered with spikes of 10 and 18 nm length, which gives the particle a unique “double-fringed” appearance.617 Owing to the fact that HeV contains a host cell—derived lipid envelope, inactivation of the virus is possible using compounds that disrupt the lipid envelope (e.g., lysol, sodium dodecyl sulphate [SDS], acetone, methanol).618
Epidemiology
HeV is able to cause natural disease in humans and horses615 and experimentally induced disease in cats, guinea pigs, and mice.619,620 The virus naturally infects bats of the suborder Megachiroptera, in which infection does not seem to cause clinical disease.621,622 These bats are considered to be the natural reservoir of HeV, yet the method of transmission from bats to horses is unknown. Although the virus transmits readily between bats, it does not appear to spread easily between horses or from horses to humans. This notion is highlighted by the finding that no disease spread occurred to other nearby stables during the outbreaks in 1994.623 In addition, not all horses on the premises became infected, and horses placed in uncleaned stalls that previously contained animals that succumbed to the disease did not become infected. Under experimental conditions, transmission did not occur between infected and in-contact horses.624 However, natural transmission among mammalian species can occur but seems to require very close contact.619 Despite the fact that intermammalian transmission of HeV appears to be relatively rare, the virus has the potential to cause lethal zoonotic infection and therefore poses a serious public health and occupational safety risk for veterinarians.
Pathogenesis
Experimentally, horses can be infected after parenteral and oronasal challenge.615,624 Infection is characterized by the systemic distribution of the agent. After experimental infection of horses and cats, virus was isolated from the buccal cavity, blood, brain, spleen, lungs, bronchial lymph nodes, kidneys, and urine.619,620,624 Based on the finding that virus is shed in the urine of experimentally infected animals, it has been speculated that consumption of food contaminated by urine of infected horses may serve as a method of transmission under natural conditions. However, although virus was present in the urine of horses that had died of natural HeV infection, investigators failed to recover HeV from the floors of the stalls in which the animals had been housed.624
Clinical Presentation
In horses, HeV infection is characterized by an acute, febrile respiratory illness that is sometimes accompanied by facial swelling, ataxia, head pressing, and recumbency. Frothy nasal discharge and tachycardia are considered to be indicators of imminent death. The course of disease in infected horses is short, with animals dying within 36 hours after onset of clinical signs. Experimentally the incubation period is 6 to 12 days. Although only one of eight experimentally infected horses recovered, seven infected horses survived during the field outbreak at Hendra. However, two of these horses subsequently developed neurologic signs, which consisted of tonic spasms of the muscles of the neck and hind limbs.625
At necropsy the most significant lesions are the severely congested, firm, and fluid-filled lungs. In some cases fibrin tags are present on the pleural surface. Histologically, HeV infection is associated with interstitial pneumonia with proteinaceous alveolar edema and hemorrhage. Possibly the most remarkable histologic finding is the presence of syncytial giant cells in the endothelium of pulmonary blood vessels. If examined under the electron microscope, these giant cells contain cytoplasmic inclusion bodies containing the characteristic nucleocapsid structure of paramyxoviruses.618
As only three cases of human HeV infection have been reported, the disease syndrome associated with infection is poorly defined. One patient developed severe, fatal respiratory illness, and a second patient developed a self-limiting influenza-like illness.626 The third patient succumbed to severe HeV encephalitis more than a year after initial infection.627
Control
To date, no vaccine or antiviral therapy is available to control HeV infection. The outbreak in Australia was controlled by a stamping-out program involving slaughter of all known infected horses, quarantine of premises, control of animal movement in defined zones, and serologic surveillance.
EQUINE LUNGWORM
JULIE ROSS
The nematode Dictyocaulus arnfieldi can affect donkeys, horses, ponies, and zebras and is found throughout the world.628 Donkeys are believed to be the natural hosts of D. arnfieldi, with a reported prevalence of infection of 51% to 73%.629,630 Clinical signs of D. arnfieldi infection are rarely seen in donkeys, and individual animals have been found to maintain patent, nonclinical infection for over 5 years in the absence of effective anthelmintic treatment.631 Horses (and ponies) are abnormal D. arnfieldi hosts. The prevalence of D. arnfieldi infection in horses is difficult to establish, as infection is usually nonpatent owing to retardation of larval maturation in the lungs.632 Clayton and colleagues629 found that 5.88% of horses living in contact with donkeys had patent D. arnfieldi infection. A study of thoroughbred horses in Kentucky by Lyons and co-workers633 found D. arnfieldi to be present in the lungs of 11% of horses at postmortem examination (on individual farms, a prevalence of up to 83% was found). In horses with documented parasite infection, only 30% had patent D. arnfieldi infection (documented via a modified Baermann’s technique). Most parasites identified in the pulmonary tissues were larval stage 5 (L5), with few sexually mature adults being found. A study of 5437 live equids revealed a prevalence of patent D. arnfieldi infection of 2% and 0% in horses and ponies, respectively.634 In contrast, patent D. arnfieldi infection was identified in 68% of donkeys and 30% of mules. These studies highlight the difficulty of identifying D. arnfieldi infection in horses and ponies. Patent D. arnfieldi infection can be more easily established in pony foals than in adults. Necropsy examination of these foals revealed no evidence of retardation of larval development, with sexually mature adult parasites frequently being identified.629
D. arnfieldi has a direct life-cycle. L1 hatches shortly after eggs are passed in the feces, and the larvae undergo two molts before they enter a free-living, infective stage (L3). The development of L3 can occur in 4 days. Pilobolus fungi may play a role in the dissemination of D. arnfieldi larvae from feces.635 L3 is ingested by the host and penetrates the intestinal mucosa before passing to the mesenteric lymph nodes, where molting occurs. L4 organisms travel, via lymph and blood, to the lungs and break out of the capillaries into the alveoli. Molting from L4 to L5 occurs in the bronchioles, and maturation to the adult form occurs in the small bronchi.628 Eggs are laid in the bronchi and then transported via the mucociliary clearance apparatus to the pharynx, where they are swallowed and eventually passed in the manure. The prepatent period of D. arnfieldi is 2 to 4 months.635 Clinical disease can be seen as early as 12 days after ingestion of infective larvae.628 The parasite has a limited survival time in the environment (49 days in warm soil) and cannot overwinter on pasture.632 Fecal larval production in donkeys increases through the spring and summer months,636 and environmental conditions favor development of patent infections in late summer and autumn.
Transmission from donkeys to horses is thought to be the main source of infection in horses, but horse-to-horse transmission does occur, thus clinical disease cannot be ruled out based on lack of contact with donkeys.634,629 In one study 67% of farms on which horses with patent D. arnfieldi infection were stabled had no history of occupancy by donkeys.634
Clinical disease associated with D. arnfieldi was identified in 2.6% of horses presented to a referral clinic for evaluation of suspected pulmonary disease.637 The average age of the population identified as being infected with D. arnfieldi was 5 years. Clinical signs of D. arnfieldi infection in horses have been described. One study identified coughing in 100% of cases, with nasal discharge (42.9%), abnormal tracheal auscultation (42.9%), dyspnea (33.3%), and abnormal thoracic auscultation (28.5%) being identified less frequently.637 Other clinical signs that have been described include paroxysmal coughing, increased expiratory effort, and audible wheezes and crackles on auscultation.632 Many reports of lungworm infection describe a chronic disease process; however, in a study involving seven D. arnfieldi cases, three cases were of acute onset. It is possible that these animals had concomitant bacterial or viral disease or that the initial lungworm challenge resulted in acute clinical signs.637 Although clinical disease is rare in donkeys, D. arnfieldi infection has been associated with increased susceptibility to respiratory viruses (e.g., influenza).637
As discussed earlier, identification of D. arnfieldi infection using a modified Baermann’s technique frequently results in false-negative results.629 Clinical signs of nonpyrexic, chronic respiratory disease, possibly with a history of contact with donkeys, may be suggestive of D. arnfieldi infection if other causes of respiratory disease have been ruled out. Dixon and colleagues637 associated a marked increased in eosinophil ratio in BALF and tracheal secretions with D. arnfieldi infection if there was concurrent evidence of lungworm larvae in the airways or respiratory secretions or if the eosinophilic nature of the respiratory secretions resolved with appropriate anthelmintic treatment. Respiratory secretion eosinophilia may be a useful indicator of lungworm infection; however, this finding can also be seen in eosinophilic pulmonary disease637,638 and during transient migration of P. equorum.628 Clinical disease with P. equorum is most commonly seen in young horses and ponies. Tracheobronchial aspirates and BALF obtained from normal horses are expected to contain few (<3%) eosinophils639; however, significant respiratory secretion eosinophilia has been identified in horses with no apparent respiratory disease.640 Eosinophilia in respiratory secretions is therefore suggestive of but not pathognomic for D. arnfieldi infection. Repeat evaluation of respiratory secretions after D. arnfieldi treatment should reveal a marked improvement in eosinophilia ratio in lungworm cases.640 D. arnfieldi larvae have been identified during microscopic examination of centrifugated respiratory mucus before fixing and staining have been performed, and it is suggested that such evaluation is an important diagnostic tool.632 Peripheral eosinophilia has not been definitively correlated with lungworm infection.641 At necropsy, a modified Baermann’s technique can be applied to diced lung tissue to aid in the identification of larvae.
Treatment for D. arnfieldi infection includes effective anthelmintic treatment, removal from contact with donkeys, and moving horses and ponies to a pasture not grazed by donkeys since the previous autumn. Effective anthelmintics include moxidectin,642 ivermectin (active against mature and immature parasite stages),629,643 and mebendazole (16–20 mg/kg for 5 days).644 Fenbendazole has been shown to improve clinical signs of presumed D. arnfieldi infection; however, suppression of fecal shedding was maintained for less than 4 weeks with doses up to 30 mg/kg.641 Treatment with 200 μg of ivermectin per kilogram, along with a pasture change after treatment, resulted in complete improvement in six of seven D. arnfieldi cases and partial improvement in one case.645 Treatment of donkeys should be instituted to decrease transmission to horses and ponies and potentially to decrease the donkey’s susceptibility to other respiratory diseases.636
Necropsy findings in donkeys and horses with D. arnfieldi infection are similar.646,647 Gross findings include circumscribed areas of raised pulmonary tissue (3 to 5 cm), more commonly found in the caudal lung lobes. Sectioning of these areas reveals overinflated pulmonary tissue surrounding small bronchi that are packed with mature D. arnfieldi. Small airways in these areas tend to be occluded with exudate. Histologic examination of the pulmonary tissue reveals diffuse eosinophilia throughout the pulmonary tissues, especially around the airways. A marked inflammatory reaction is found around parasitized bronchi, with heavy lymphoid cell infiltration of epithelium, lamina propria, and peribronchial tissue. Epithelium becomes hyperplastic, and mucus-secreting cells increase in size and number. Little cellular or mucoid reaction is seen in bronchial lumen around adult parasites; however, free larvae in bronchial lumen result in intense mucopurulent reaction. Bronchioles in affected areas of the lung usually show evidence of bronchiolitis, with free mucus present in the lumen of bronchioles and hyperplastic, columnar epithelium. Bronchioles are most commonly surrounded by discrete lymphoid nodules. Pulmonary pathology in donkeys tends to be very localized with much of the lung appearing normal on histologic examination.646
Prevention of D. arnfieldi infection in horses and ponies involves effective anthelmintic treatment and pasturing horses and ponies separately from donkeys or treating donkeys with appropriate anthelmintic treatment to avoid patent infection. Anthelmintic treatment is especially important in the spring. Regular removal of manure from pasture can also help to decrease the parasite load in the environment.
THORACIC TRAUMA
JANE E. AXON
Thoracic trauma results from blunt or penetrating injuries to the chest wall. Trauma can result in thoracic wounds, hemothorax, pneumothorax, fractured ribs, and diaphragmatic hernia, and a combination can occur in the same horse. Prompt assessment and implementation of emergency procedures in the injured horse with respiratory distress in the field is vital to optimize the horse’s chances for survival.648
Clinical signs associated with injury to the thorax can vary from mild musculoskeletal pain and a stiff gait to severe respiratory distress and hypovolemic shock. Signs of respiratory distress include anxiety, tachypnea, nostril flaring, cyanotic mucous membranes, tachycardia, and an altered respiratory pattern. Auscultation and percussion of the thorax may assist with locating and identifying the lesions. Percussion and palpation will need to be performed carefully in horses in pain. Palpation of SC emphysema in the absence of an external wound may be associated with a lacerated lung or tracheal trauma and in a dyspneic horse should raise the suspicion of a concurrent pneumothorax.649 The injured horse may also have weak pulses, pale mucous membranes, and tachycardia resulting from acute hemorrhage from lacerated intercostal or pulmonary parenchymal vessels. Evaluation of the abdomen should also be undertaken. On expiration the cranial portion of the diaphragm extends to the sixth rib; therefore an abdominocentesis should be performed on a horse with a wound caudal to the sixth rib or with a deep penetrating wound to assess abdominal involvement.648 Further evaluation of the injured horse will include thoracic radiographs, ultrasonographic examination, and exploration of the wound. Arterial blood gas analysis will assist in determining the extent of hypoventilation.
Open thoracic wounds should be covered immediately to prevent additional air from entering the chest cavity.649 Plastic food wrap wrapped around the thorax or a gauze roll or towel sutured in place can be used. A tension pneumothorax, which may occur with sucking thoracic or axillary wounds, can be alleviated by inserting a trocar into the dorsal pleural space so pleural and atmospheric pressures can equilibrate and a less compromising pneumothorax is created.648 Intranasal oxygen therapy and removal of air from the thorax may be necessary to improve alveolar ventilation and oxygenation in a horse with pneumothorax before further evaluation. Circulatory support may be necessary until hemorrhage has stabilized.648 Sedatives and tranquilizers should be used judiciously because most cause respiratory depression; however, they may have to be used if the horse’s anxiety precludes instituting treatment.
Hemothorax
Thoracic trauma resulting from lacerations to pleural or pulmonary vessels or rupture of large thoracic vessels may result in hemothorax. Other causes of hemothorax include rupture of lung parenchymal bullae, vessel erosion by severe lung abscessation or neoplasia, hemangiosarcoma, spontaneous hemorrhage at maximal excursion, and coagulopathy.650,651 Iatrogenic causes include tube thoracostomy and lung biopsy. Hemothorax can be unilateral or bilateral depending on the cause and whether the mediastinum is complete.
Diagnosis is based on clinical signs, radiographic and ultrasonographic findings, and laboratory evaluation of the fluid obtained from thoracocentesis.650 The horse may be dyspneic and tachycardic, depending on the volume of blood loss into the pleural cavity. On auscultation there is a decrease in normal breath sounds ventrally, and heart sounds are often muffled and radiate over a wider area.650 Percussion reveals a change from the normal resonance of aerated lung to dullness over the hemothorax.650 Thoracic radiographs show opacity of ventral lung fields, with an associated horizontal fluid line and a loss of diaphragmatic and cardiac silhouettes.652 Ultrasonography shows fluid within the pleural cavity with a characteristic hypoechoic to echogenic smoke swirling pattern.653 Fluid collected by thoracocentesis should be submitted for cell count, packed cell volume, and total protein. A cytologic evaluation should also be performed to determine whether there is evidence of infection or neoplasia, and the sample should be cultured if an infection is suspected. A clotting profile and platelet count should be performed if a coagulopathy is suspected. Hematology and biochemistry should also be performed, as the horse may develop anemia and hypoproteinemia associated with the blood loss.
Treatment is aimed at stabilizing cardiopulmonary function and treating the underlying cause. Horses with hemothorax from nonpenetrating thoracic trauma or a noninfectious cause can be managed conservatively with intranasal oxygen insufflation, analgesics, IV fluids, or whole blood transfusion if the blood loss is severe. If the horse is not hypoxemic or in respiratory distress, rest alone and close monitoring may suffice. Even if respiratory distress is evident, a tube thoracostomy and drainage of blood may not be necessary.651 Drainage of the thorax is not recommended in horses with a coagulopathy unless severe respiratory distress is present.651 If there is a penetrating wound or evidence of infection, drainage should be attempted by ultrasound-guided ventral placement of chest drains. This, however, should not be undertaken until the hemorrhage has stopped and the circulating blood volume has been restored.654 All horses, even without evidence of infection, should have broad-spectrum antimicrobial coverage, as blood is an excellent medium for bacterial growth.650
The prognosis for horses with uncomplicated trauma and hemothorax is good. If the hemothorax is the result of a penetrating wound, the prognosis is poor.
Pneumothorax
Traumatic causes of pneumothorax include puncture or laceration of the trachea, ruptured esophagus, penetration of foreign objects into the thoracic cavity, external wounds resulting in SC emphysema and pneumomediastinum, and direct penetrating or blunt trauma to the lung parenchyma.650,655-657 Pleuropneumonia is also an important cause of pneumothorax ; in one study 17 of 40 horses had pneumothorax resulting from pleuropneumonia.655 Air can escape into the pleural cavity through slow leaks from necrotic lung or through formation of bronchopleural fistulas. Additional causes of pneumothorax include ruptured emphysematous lung bullae, maximal exercise, and iatrogenic causes such as tube thoracostomy, lung biopsy, mechanical ventilation, and transtracheal aspiration.650,651
Pneumothorax can be described as closed, in which air from the lung is trapped in the pleural space, or open, in which there is free communication between the pleural space and external environment and air is sucked into the pleural space with inspiration. A tension pneumothorax occurs when air accumulates in the thorax until intrapleural pressure exceeds atmospheric pressure. This is often seen with a sucking pleurocutaneous wound as air moves into the chest cavity with inspiration and, owing to a valve effect of the wound, cannot escape during expiration.649 Intrapleural pressure may increase to such an extent that the thorax becomes fixed in maximal extension. This leads to severe cardiopulmonary effects with decreased venous return to the heart because of vena cava compression and hypoxemia.658,658a Pneumothorax is usually bilateral because of the incomplete mediastinum of horses; however, it may be unilateral if the fenestrations in the mediastinum are blocked as a result of inflammation or a collapsed lung.
Diagnosis is based on clinical signs, aspiration of air from the pleural cavity, and radiographic and ultrasonographic findings.650 Clinical signs that can be associated with pneumothorax are dyspnea, tachypnea, cyanosis, and evidence of trauma.650 Characteristic findings on auscultation are a decrease or lack of normal breath sounds dorsally and tympany and hyperresonance on percussion; however, these clinical signs can vary and are not reliable.655 Concurrent SC emphysema may complicate interpretation. However, if a horse with SC emphysema is dyspneic or distressed, pneumothorax should be suspected unless the emphysema is causing airway compression.650 Thoracic radiographs show lack of pulmonary vasculature in the dorsal aspect of the caudal lung fields and ventral displacement of the dorsal lung margin (Fig. 31-35). On ultrasonographic examination there are parallel horizontal lines of air artifact reverberation without the pattern of pleura and “comet tail” artifacts. The dorsal air image also moves ventrally with inspiration over underlying collapsed lung or pleural fluid, creating a “curtain” image.653 SC emphysema in tissue and fascial planes may prevent accurate sonographic imaging. Aspiration of air from the thorax is also diagnostic, as is the associated improvement in clinical signs.
Treatment is aimed at improving alveolar ventilation by correcting the pneumothorax, if the horse is showing signs of respiratory distress, and treating the underlying cause. Treatment of simple pneumothorax requires rest and close observation while gradual reabsorption of air occurs. If hypoxemia (PaO2 < 80 mm Hg, percent oxygen saturation [%O2sat] < 90%) or dyspnea is present, nasal insufflation of oxygen (15 L/min) should be administered.654 An open sucking wound should be occluded. Air is removed from the pleural space by inserting a teat cannula or thoracostomy tube, with a suction device attached, into the dorsal thoracic cavity. If a suction machine is not available, repeated aspiration with a 60-ml syringe through a three-way stopcock valve and or tubing attached to the teat cannula and run into a container of water can be used.649 The latter acts as a simple water trap, allowing air to be expelled during inspiration while preventing air movement back into the chest with expiration.649 If the pneumothorax reoccurs or continues, tubes should be left in place to allow constant air removal. A Heimlich chest drainage valve* provides continual drainage and if correctly placed is effective for long periods. In long-standing cases, gradual reexpansion of the lungs is recommended to avoid reexpansion pulmonary edema.650 Prophylactic broad-spectrum antimicrobial therapy is recommended as long as the tube is in place. If the cause of pneumothorax requires correction under general anesthesia, ventilation should be improved before induction by removing air from the pleural cavity and correcting lung atelectasis.
The prognosis for horses with pneumothorax is good, providing infections can be successfully treated and air leaks are sealed. Pneumothorax associated with pleuropneumonia and parenchymal lesions resulting in air leakage carry a poor prognosis and with an esophageal rupture carry a very poor prognosis.650,655
Rib Fractures
Fractured ribs are most often a result of an accident or kick; however, in neonates fractured ribs are commonly associated with birth trauma.
ADULTS
Fractured ribs can be detected either through exploration of the wound or by eliciting a pain reaction on palpation over the affected area. Crepitus associated with a closed fracture is often not detected. Shallow breaths and guarded thoracic movements may be seen as a result of pain from the fracture. Soft-tissue swelling and SC emphysema may also be present. Fractured ribs in adults usually heal without fixation; however, if pain and excessive movement of rib ends are present, surgical correction may be warranted. Stabilization of a flail thorax is usually not necessary unless there is respiratory compromise.649 NSAIDs, circumferential bandaging, and intercostal nerve blocks assist with providing pain relief. Open fractures should be explored and debrided, and soft tissue repaired.649 The horse should be closely monitored for respiratory distress or hemorrhage, as a closed pneumothorax or hemothorax may result if a fractured rib end lacerates underlying lung parenchyma or vessels.648
FOALS
Blunt thoracic trauma during parturition resulting in costochondral dislocation or rib fracture is more common in foals from primiparous mares and dystocias.658a The majority of fractures are subclinical; however, sudden death, hemothorax, hemopericardium, lacerated myocardium, pneumothorax, diaphragmatic hernia, and severe respiratory distress can occur.658a,659 The incidence of fractured ribs in newborn foals on one farm was 21% (55 of 263), and none had clinical signs associated with the fracture after a 6-month period.658a However, in other reports rib fractures accounted for 13% of life-threatening fractures in foals less than 7 days of age, and in a necropsy study 19 of 67 foals with fractured ribs (28%) died as a direct result of rib fracture.659,660 The most common site of injury is the cranioventral half of the thorax at the costochondral junction or adjacent dorsal area.659 Clinical signs are variable and include pain on rib palpation, “clicking” or crepitation on auscultation, edema localized over or ventral to the fractured ribs, and signs of pain including grunting or groaning when the patient is manipulated or lying on the affected side. Palpation of crepitus over the fractured site is not a consistent finding. If the foal is systemically stable, asymmetry of the thoracic cage can be evaluated if the foal is positioned in dorsal recumbency.658a In severe cases involving multiple consecutive ribs, the foal can be in respiratory distress with a flail thorax. Foals may also exhibit respiratory distress with pneumothorax. Pale mucous membranes can be indicative of internal hemorrhage and abdominal hemorrhage, and diaphragmatic hernia should be considered if the diaphragm has been lacerated. Radiographs for detection of fractured ribs in neonates is not reliable.659,661 Ultrasonographic examination is more reliable and also assists in evaluating the adjacent structures and presence of hemothorax or pneumothorax (Fig. 31-36). Conservative treatment with box rest and supportive care is successful in the majority of uncomplicated rib fractures; however, surgical repair has been performed in patients with multiple rib fractures or those with potential for severe internal injury.661,662 Specific treatments based on the internal injuries should also be implemented.
Diaphragmatic Hernia
Diaphragmatic hernias occur sporadically in horses.663,664 Occurrence is usually associated with a history of trauma or sudden increase in abdominal pressure and exertion, as may be experienced by stallions during breeding or mares during parturition.664 In one study, 48% of 50 horses diagnosed with diaphragmatic hernia had a history of recent trauma.663 In some cases no history of injury or predisposing cause can be identified.664 Congenital diaphragmatic hernia can occur owing to incomplete fusion of any of the four embryonic components of the diaphragm or as a result of abdominal compression during parturition.665 A congenital peritoneopericardial hernia has been reported.666 Congenital hernias are typically located in the most ventral portion of the diaphragm, whereas diaphragmatic defects from trauma are usually located at the junction of muscular and tendinous portions of the diaphragm.665 The size and position of the defect are important in determining whether herniation of abdominal viscera occurs.664,667 Any segment of intestine can be involved, although small intestine is the most frequent.665
Clinical signs usually relate to intestinal obstruction or respiratory compromise; however, less specific signs of exercise intolerance and weight loss have been reported.664 A diaphragmatic hernia may also initially be clinically silent if there is minimal pulmonary or bowel compromise. GI-related clinical signs are variable and depend on the segment and amount of intestine herniated and whether it is simply displaced, incarcerated, or strangulated.664 Frequently reported clinical signs are colic, moderate to severe tachypnea, and endotoxic shock. Respiratory signs of tachypnea can be attributed to pain at the site of injury or GI tract, shock, and lung compression and decreased ventilation.664,667,668 Acute tearing of the diaphragm may be associated with hemorrhage into both thoracic and abdominal cavities.
Diagnosis can be challenging, and thoracic radiographs showing abdominal organs within the thoracic cavity are considered most diagnostic.664,667 Ultrasonographic examination may show abdominal organs in the thoracic cavity; however, viscera may not be seen if covered by aerated lung or located away from the thoracic wall.653 Rectal examination often suggests a relative lack of normal abdominal viscera. Auscultation of the thorax may reveal GI sounds, but it is usually not possible to differentiate from sounds referred from the abdomen. There may also be an absence of breath sounds ventrally and associated dullness on percussion, although these findings are not reliable. Abdominal paracentesis is usually normal and can be misleading because the compromised intestine is in the thorax, and therefore thoracocentesis may be more representative.664 Thoracoscopy may also assist visualization of the hernia; however, partial deflation of the lungs is necessary to visualize the diaphragm, which may further compromise the patient.658,667
Surgical repair of diaphragmatic hernias has been reported but is fraught with complications associated with anesthesia, resection of devitalized bowel, anatomic location, and repair of the defect and possible fractured rib repair.664 In chronic cases with few or no clinical signs, repair does not appear to be necessary, although complications associated with herniated viscera may eventually occur.665,669
PULMONARY EDEMA
PAMELA A. WILKINS
Pulmonary edema rarely occurs as a primary event in the horse and, when present, is usually secondary to some other pathologic process. Extravascular fluid accumulates within the lung after events that alter hydrostatic and colloid osmotic interstitial and vascular forces, change the surface area and pore size of the blood gas barrier, or diminish lymphatics drainage.670 Pulmonary edema can be classified as cardiogenic or noncardiogenic. Pulmonary capillary pressure can be increased by any increase in left atrial or pulmonary artery pressure. In the horse this can occur secondary to acute renal failure, left ventricular failure, or very high cardiac output conditions, such as extreme exercise. Increases in microvascular permeability may occur with sepsis, disseminated intravascular coagulation, hypoxic acidosis, or primary pulmonary pathology, resulting in the release of mediators of inflammation that increase vascular endothelial or alveolar epithelial permeability. Pulmonary edema associated with airway obstruction has been termed negative-pressure pulmonary edema (NPPE) and has been reported in horses.671,672 NPPE occurs secondary to inspiratory efforts against a closed glottis that result in a precipitous fall in intrathoracic pressure. The large decrease in intrathoracic pressure increases the transmural pressure gradient for all intrathoracic vascular structures, favoring movement of water into the extravascular space.
Diagnosis is based on clinical examination, a history of predisposing causes, and radiographs. Horses have a shallow rapid respiratory pattern and may be dyspneic. Arterial blood gas analysis may reveal hypoxemia and hypercapnia. Fine crackles or wheezes may be audible on auscultation. Patients with volume overload (associated with renal failure or, rarely, too-rapid fluid administration) or primary cardiac problems may have an increased central venous pressure with pronounced venous distention. Fluid (clear or slightly yellow or pink-tinged) may drip from the nostrils and can increase in volume without necessarily becoming frothy. Progression to this stage warrants a very grave prognosis. Radiographic findings are nonspecific but include peribronchial and perivascular cuffing, increased prominence of vessels, and a hazy reticular interstitial pattern. Underlying pulmonary disease may obscure signs of edema, and radiographs of sufficiently high quality to show relatively subtle changes may not be obtainable in mature horses. Noncardiogenic pulmonary edema (capillary leak) is a component of the definition for ALI and ARDS.
Treatment
Treatment consists of correcting the cause, reversing hypoxemia, decreasing plasma volume and left atrial pressure, and increasing plasma colloid osmotic pressure. Intranasal oxygen and even mechanical ventilation may be needed in severe cases. Improvement in oxygenation can be monitored by sequential arterial blood gas analysis or by using transcutaneous oxygen saturation monitoring equipment applied to the nasal mucosa, tongue, or other available nonpigmented mucous membrane. In cases of NPPE, maintaining an adequate, low-resistance airway is very important to prevent further damage, and tracheostomy may be necessary. IV fluid therapy should be guided by the patient’s needs and monitored by serial measurement of central venous pressure if necessary. Furosemide may be given intravenously or intramuscularly at a dose of 1 to 2 mg/kg and repeated in 1 hour. If helpful, the dose can be titrated for each patient. At a dose of 1 mg/kg, approximately 8 L of urine is produced in approximately 1 hour.673 A few studies have been conducted on the effects of furosemide on pulmonary hemodynamics in the horse.674-677 The effects in horses with pulmonary edema have not been reported. Colloid solutions should be administered cautiously or in conjunction with use of diuretics because they can initially increase vascular pressure. Plasma may be safer than other colloid preparations such as dextrans or hetastarch. Colloid solutions may be of little benefit in raising intravascular osmotic pressure in patients with increased microvascular permeability.
Antiprostaglandin drugs (flunixin meglumine, phenylbutazone) and antihistamines may help. Bronchodilators may be of benefit. The use of corticosteroids remains controversial; if they are used, antimicrobial coverage is advisable because pulmonary edema has been shown to impair pulmonary bacterial defense mechanisms.
SMOKE INHALATION
PEGGY S. MARSH
Smoke inhalation injury is typically associated with exposure to fires, and there are often concurrent problems in other body systems from thermal injury. Extensive or severe burns can magnify the severity of the injuries. Thermal injury along with smoke inhalation leads to both local and diffuse lesions. Massive tissue edema may occur; it can be a challenge to manage and can create organ dysfunction. To further complicate the problem, severely affected patients may develop a wide variety of problems including life-threatening sepsis and/or hypermetabolism. Depending on the severity of the lesions, smoke and fire injuries often lead to systemic inflammatory response syndrome and in some cases multiple organ system failure.
Insult to the respiratory system by smoke inhalation depends on the fuels that burned, the completeness of combustion, and the generated heat intensity. In general, lesions are initiated by three mechanisms. The first is direct thermal injury, which can be limited to the upper respiratory tract by laryngeal reflexes and efficient heat exchange within the nasal passages. Toxic chemicals in the smoke can cause damage, both directly and indirectly, through inflammatory mediators. Carbon monoxide intoxication is commonly associated with human injuries from smoke and is a product of incomplete combustion.678 Finally, with combustion there is consumption of oxygen, and the resulting low PAO2 can lead to pulmonary vasoconstriction as well as generalized hypoxia.
Three phases of pulmonary dysfunction have been described in the horse.679,680 The first stage is acute pulmonary insufficiency caused by several mechanisms. Carbon monoxide may be present in sufficiently high concentration to cause toxicity within a short time after exposure. Carbon monoxide combines with hemoglobin to form carboxyhemoglobin. Hemoglobin has a 200- to 250-times greater affinity for carbon monoxide as compared with its affinity for oxygen.678 High levels of circulating carboxyhemoglobin result in a shift of the oxyhemoglobin dissociation curve to the left, thereby decreasing oxygen release at the tissue level and leading to tissue hypoxia. Other processes occurring during this acute phase include progressive edema and necrosis in the upper respiratory tract, leading to airway obstruction, bronchoconstriction in the lower respiratory tract from the irritating effects of noxious products, and altered pulmonary blood flow.681
These insults produce the second stage: formation of pulmonary edema, lower airway obstruction, and pulmonary parenchymal lesions. Within 48 to 72 hours after exposure, driven by pulmonary macrophages, neutrophils are called into the area of insult. They release cytokines, proteolytic enzymes, and oxygen-derived free radicals. Expression of the inflammatory cascade in excess of balance causes microvascular damage, leading to increased extravascular lung water. Local insult also results in the release of tissue factor initiating the coagulation cascade to produce fibrin. Debris from the inflammatory cascade along with fibrin and material directly deposited from smoke inhalation create pseudomembranous casts, which may obstruct the small airways. Widespread plugging of the airways may significantly increase airway pressure, causing barotrauma and alveolar damage.681 Bronchopneumonia is the last stage and occurs as a result of the impaired host immune system, both locally and systemically. This phase may occur up to 1 to 2 weeks after the initial injury.
As the saying goes, “Where there’s smoke, there’s fire“; commonly horses with smoke inhalation injury will also have burn injury. A complete description of this process in horses has been reported.682 Thermal injury causes a local response that includes microvascular insult and direct tissue coagulation leading to inflammation, local edema, and finally necrosis. Extensive local injuries will drive a systemic response. Initially there is a decrease in systemic organ blood flow. This is followed by the formation of generalized edema. The pathophysiology of edema formation is complex and involves protein shifts, endothelial damage, and alteration of the interstitial architecture, all leading to a net accumulation of fluid in the interstitial spaces.683 Over time major thermal injury is characterized by high cardiac output, increased oxygen consumption, and protein and fat wasting, all of which may create a hypermetabolic state.684,685 Loss of skin as a barrier, release of inflammatory mediators, and hypermetabolism play a role in the development of immunosuppression. Other features noted with burn injury in the horse include acute hemolysis, renal failure, laminitis, and myositis.686
Horses exposed to fire with smoke will have a variety of clinical signs depending on the duration and type of exposure and the length of time from the insult. The extent of damage to the skin may be very difficult to ascertain initially. Acutely, within the first 6 hours, signs of carbon monoxide toxicity and shock may occur. The patient shows signs of severe hypoxemia and may be depressed, disoriented, irritable, ataxic, or even moribund and comatose. As edema and necrosis progress in the upper respiratory tract, dyspnea and stridor may develop. Auscultation of the thorax may reveal decreased air movement, crackles, or wheezes, but these may not become apparent for 12 to 24 hours. If edema of the airways is sufficiently severe, airflow may be severely restricted. Edema fluid may be visible at the nostrils and, later, may be replaced by inflammatory exudate. Concurrent edema formation may be occurring systemically. Hypoxia and generalized edema may lead to dysfunction of distant organs such as the kidneys and muscles. Signs of infection may be difficult to ascertain from other signs. All that may be noticed is a fever and a worsening of respiratory signs after initial improvement.
Diagnosis is typically based on history and physical examination. A normal initial examination does not rule out exposure because the onset of clinical signs may be delayed several days. Within a short time after exposure, carboxyhemoglobin concentration in venous blood can be measured. A level above 10% is consistent with carbon monoxide toxicity.679 Various diagnostic tests are useful in determining the extent of respiratory injury. These include endoscopy of the upper respiratory tract and tracheobronchial tree, thoracic radiographs, blood gas analysis, hematology, and cytologic evaluation of tracheal aspirates. Any or all of these tests can be performed on a serial basis as prognostic aids. Diagnostic tools for other organ injury include blood pressure monitoring, measurement of central venous pressure, serial hematologic and serum chemistry analyses, and monitoring of urine production.
Treatment depends on stage of injury. Initially, oxygen support is of benefit. It is a treatment for CO toxicity and helps reduce hypoxemia.687 Humidified oxygen can be supplied by nasal insufflation or via transtracheal catheter. Upper respiratory tract obstruction may necessitate a tracheostomy. Attention should be paid to keeping the airways clear, and nebulization may be useful, especially when pseudomembranous casts are suspected. Bronchodilators may be useful in counteracting reflex bronchoconstriction. Decreasing inflammation and pulmonary edema may necessitate the use of diuretics and NSAIDs. Use of corticosteroids is controversial because of potential for immunosuppression and laminitis.679 Novel therapies may include use of hyperbaric oxygen chambers and inhalation treatment with medication to inhibit inflammatory mediators, coagulation factors, or oxidative stress.688
Secondary complications require attention. Cardiovascular compromise and burn-induced edema may necessitate the judicious use of IV fluids. To prevent infection, strict hygiene, meticulous nursing care, and optimal nutritional support should be provided.682,688 Prophylactic antimicrobial use is not recommended in human patients. Documented infection should be treated with appropriate antimicrobial agents based on results of culture and sensitivity patterns.682
RECURRENT AIRWAY OBSTRUCTION
DOROTHY M. AINSWORTH
Definition and Etiology
RAO is an inflammatory condition of the lower respiratory tract of mature horses that is characterized by excessive mucus production, neutrophil accumulation, bronchial hyperreactivity, and (in most cases) reversible bronchospasm. Because the equine disease differs substantially in its etiopathogenesis from chronic obstructive pulmonary disease (COPD) of humans, it is recommended that the equine disorder no longer be called COPD. The preferred name is RAO or “heaves.”689
RAO typically develops in adult horses residing in the northern hemisphere, for example in the Northeast and Midwest regions of the United States as well as in Great Britain and Switzerland, where the climate is wet and cool and horses are stabled and fed hay. RAO rarely occurs in horses maintained in warm dry climates such as those found in California or in Australia.690 In North America the prevalence of RAO diagnosed at veterinary teaching hospitals has been positively correlated with rainfall, minimum temperatures, and total pollen and mold counts occurring 1 to 3 months previously.691 These findings suggest that environmental conditions are integral to the development of the disease in susceptible horses.
RAO is considered to be a hypersensitivity to inhaled molds and organic dusts contained in the feeds hay and straw. In this regard, RAO is similar to occupational asthma that develops in human workers exposed to organic dusts.692 Identifying the specific causative agents within the hay dust that induce the disease has been a challenge, as stable dust contains over 50 species of molds, large numbers of forage mites, endotoxin, and other inorganic components.693 Inhalation of aqueous extracts of Aspergillus fumigatus, Faenia rectivirgula (Micropolyspora faeni), or Thermoactinomyces vulgaris partially but not fully induces RAO in susceptible horses.694-696 Similarly, inhalation of endotoxin, a component of hay dust, has been found to produce airway neutrophilia but not all of the changes in pulmonary function that are characteristic of RAO.697,698 To fully induce RAO in susceptible horses requires inhalation of hay dust particulates either by natural challenge (stabling and feeding hay) or by nebulization of a hay dust solution.699-702
ROLE OF THE INNATE IMMUNE SYSTEM
After inhalation of the organic dust, it appears that both adaptive and innate immunologic responses contribute to the pulmonary inflammation. Within hours of hay dust exposure, neutrophilia and excessive mucus production are evident endoscopically and cytologically.703,704 Pulmonary macrophages isolated from affected horses at this time exhibit an upregulation in the gene expression of TNF-α, IL-1β and IL-8.705 The transcription of these proinflammatory genes may reflect involvement of TLR2 and/or TLR4 signaling pathways (stimulated by fungal wall components and endotoxin) with subsequent nuclear factor (NF)–κB activation and gene transcription.706-708 However, the importance of these pathways needs to be confirmed because many different mediators activate NF-κB and enhance gene expression. The initial upregulation of these proinflammatory genes is transient, with mRNA levels returning to preexposure levels 24 hours after hay dust challenge.705 With continuous hay dust exposure, both the protein and the gene expression levels of IL-8 in the airway cells and bronchial epithelium, as well as the gene expression levels of IL-1β and TNF-α in the bronchoalveolar lavage fluid (BALF) cells are upregulated in chronically affected horses.709-712 However, this gene upregulation may reflect contributions from both macrophages and extravasated neutrophils accumulating in the airways. In addition to IL-8, IL-1β, and TNF-α, another chemokine that has the potential to propagate the airway neutrophilia is IL-17.712,713 Secreted by lymphocytes and neutrophils, IL-17 promotes maturation, chemotaxis, and activation of neutrophils. In horses continuously exposed to hay dust for periods exceeding 2 weeks, IL-17 also upregulates the gene expression of IL-8 in the airway epithelium.712
ROLE OF THE ADAPTIVE IMMUNE SYSTEM
The role of the adaptive immune system in the development of RAO is currently controversial. Data that support the hypothesis that RAO is an IgE-mediated (T-helper 2 [Th2]) disorder include the findings of elevated allergen-specific IgE concentrations in the serum714 and in the BALF of RAO-susceptible or affected horses715,716 and increased numbers of IL-4 and IL-5 (mRNA positive) BALF cells—detected by in situ hybridization techniques—in RAO-affected horses.717,718 Although Th2 disorders are typically characterized by eosinophilic infiltrates, it has been hypothesized that IL-4 activates its receptor on neutrophils to inhibit neutrophil apoptosis, to increase protein synthesis, and to upregulate the expression of the IL-9 receptor.719 It has been further hypothesized that activation of the IL-9 neutrophil receptor then enhances IL-8 production and neutrophil influx.719,720 Although the gene expressions of IL-4 and IL-9 receptors are indeed upregulated in peripheral blood neutrophils isolated from RAO-prone and RAO-affected horses, the existence of this pathway, linking the Th2 limb with the development of neutrophilia, requires additional investigation and confirmation.
In contrast, other data suggest that RAO is not simply a Th2 immune response. For example, Van der Haegen and colleagues were unable to demonstrate a significant difference in IgE-protein—positive cells in the lung tissue samples of RAO-affected as compared with control horses.721 Second, cytokine profiles of BAL cells710,711 or of pulmonary CD4+ and CD8+ cells722 isolated from RAO-affected horses during the first 24 or 48 hours after hay dust exposure fail to demonstrate an upregulation of IL-4, IL-5, or IL-13. Furthermore, chronic (3 week) hay dust exposure is associated with an increase in the gene expression of IFN-γ in the BALF cells,710,711 although in one trial but not in the second trial of the same chronically affected horses the gene expression of IL-4 was upregulated.710 Fourth, GATA-3, the transcription factor required for IL-4 gene expression, is not upregulated in the bronchial cells of RAO-affected horses.723 Nevertheless, in evaluation of these studies caution should be exercised in categorizing RAO as a Th1 immune disorder. This is because (1) concomitant increases in the gene expression of t-bet, the transcriptional factor for IFN-γ expression in CD4+ cells, is not upregulated concurrently with IFN-γ,711 and (2) multiple signaling pathways may stimulate the production of IFN-γ. In other species, TLR stimulation leads to the production of proinflammatory factors including IL-12 and subsequent IFN-γ synthesis.724,725
OTHER INFLAMMATORY MEDIATORS
With the accumulation of intraluminal neutrophils and peribronchiolar lymphocytes and with the activation of alveolar macrophages, it is not surprising that numerous other inflammatory mediators are released and may contribute to the inflammatory response of RAO. For example, histamine that is released by degranulated mast cells increases vascular permeability and produces bronchoconstriction. Although histamine concentrations in the BALF increase in RAO-susceptible horses after exposure to dusty hay, histamine-1 receptor antagonists provide little therapeutic relief for affected horses. This finding suggests that histamine may not be the predominant mediator responsible for the airway inflammatory response.726 The potential role of prostaglandins, with their bronchoconstrictive (PGF2α, PGD2, TXA2) and/or bronchodilatory capabilities (PGE2, PGI2), have also been examined. In RAO-affected horses, increases in BALF PGE2 and PGF727,728 and in serum TXB2 (a metabolite of TXA2) are found.729 However, although administration of a cyclooxygenase inhibitor prevents the increase in the serum TXB2 in RAO-affected horses, it does not inhibit the development of airway hyperresponsiveness or bronchospasm in susceptible horses. This finding suggests that the eicosanoids are not the major contributor to the disease.729 The contribution of lipoxygenase metabolites to the airway disease has also been investigated in RAO-affected horses. In humans, leukotrienes have multiple actions: LTB4, LTC4, and LTD4 are bronchoconstrictors; LTC4 and LTD4 are mucus secretagogues; and LTB4 (through its action on NF-κB) is a chemotactin, promoting the upregulation of adhesion molecules and cytokines that facilitate neutrophil extravasation.730 In horses, inhalation of LTB4 or LTD4 induces airway neutrophilia and bronchoconstriction, respectively, in normal and RAO-affected horses.731 BALF cells isolated from RAO-affected horses during the first 48 hours of hay dust exposure and stimulated ex vivo produce increased concentrations of LTB4 and LTC4.730 However, actual BALF concentrations of LTB4 and LTC4 are not elevated in chronically affected horses.728 Furthermore, pretreatment of RAO-susceptible horses with either a 5-lipoxygenase inhibitor or a leukotriene receptor antagonist before hay dust exposure does not prevent the development of airway neutrophilia or alterations in lung mechanics.732,733 Treatment of clinically affected horses with a leukotriene receptor antagonist also fails to improve pulmonary function test results, clinical scores, or arterial blood gas tensions.734
There is some evidence that reactive oxygen species, derived from pulmonary macrophages and granulocytes, contribute to the inflammatory process of RAO.735 An “oxidant stress” has been proposed to develop, based on the findings of increased levels of oxidized glutathione; increased glutathione redox ratios (the ratio of oxidized glutathione to total glutathione); and decreased ascorbic acid concentrations in the pulmonary epithelial lining fluid of RAO-affected horses.735,736 Changes in these indices of oxidant stress are also correlated with lung dysfunction parameters—total lung resistance, dynamic lung compliance, and arterial oxygen tensions—in RAO-susceptible and affected horses.737 Reactive oxygen species are well recognized for their microbicidal activities. They also function as messengers in intracellular signaling pathways, upregulating proinflammatory gene expression by activating NF-κB.738 Thus it is possible that reactive oxygen species generated during the inflammatory process of RAO also contribute to the NF-κB activation that has been detected in bronchial epithelial cells and in BALF cells of RAO-affected horses.739,740
Proteases derived from airway phagocytes, lymphocytes, and epithelial cells may also propagate the inflammatory reaction of RAO. Although many matrix metalloproteinases (MMPs) with gelatinolytic and collagenolytic activities have been identified in the respiratory tract secretions of RAO-affected horses,741,742 the gelatinase MMP-9 is markedly increased in BALF and in tracheal epithelial lining fluid of diseased horses.743 It has been suggested that the degradative effect of MMP-9 on the basement membrane and on the extracellular matrix components facilitates the extravascular movement of neutrophils into the airways. In vitro, tetracycline derivates effectively inhibit MMP activity detected in tracheal epithelial lining fluid,744 but the efficacy of these compounds in ameliorating or preventing the inflammatory response of RAO-susceptible horses exposed to hay dust remains to be determined.
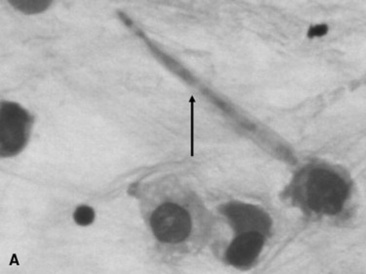
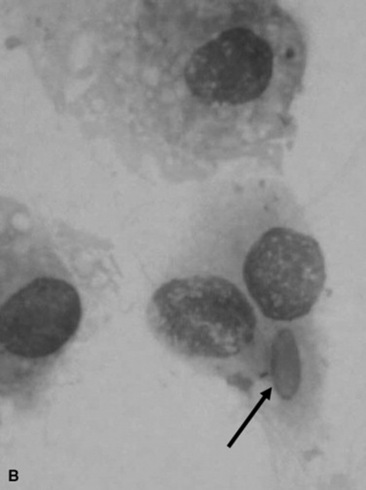
 ,323,329
,323,329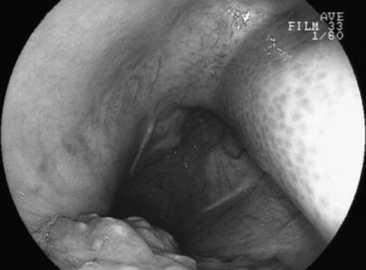
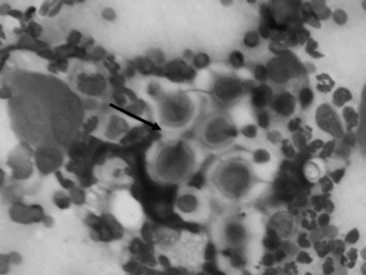
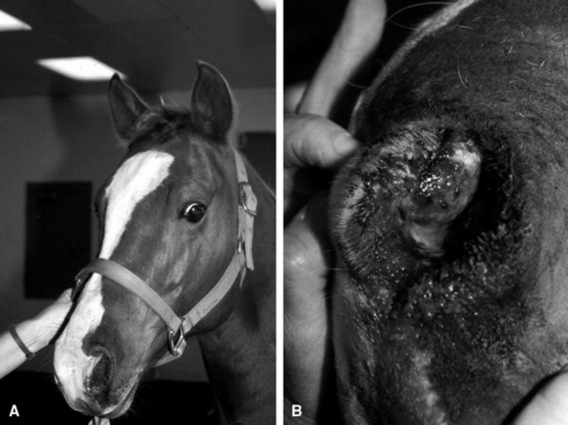
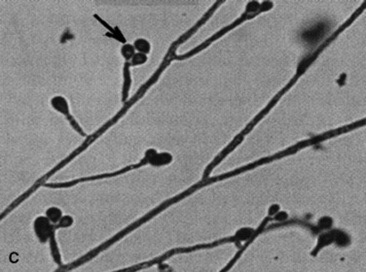
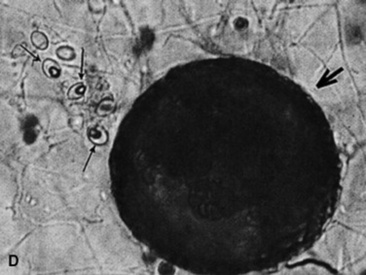
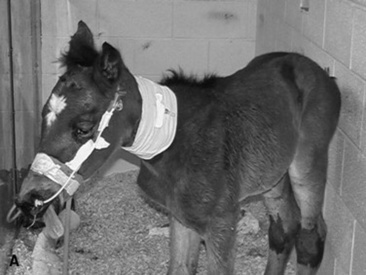


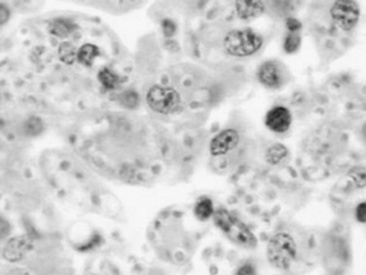
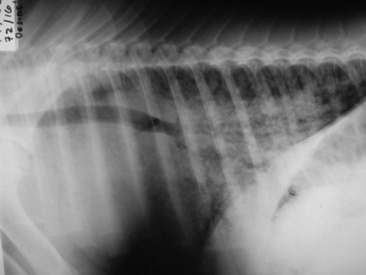
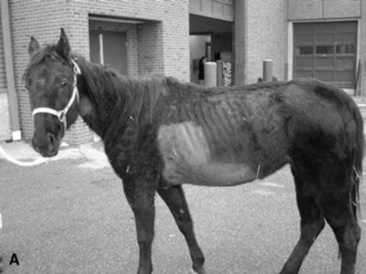



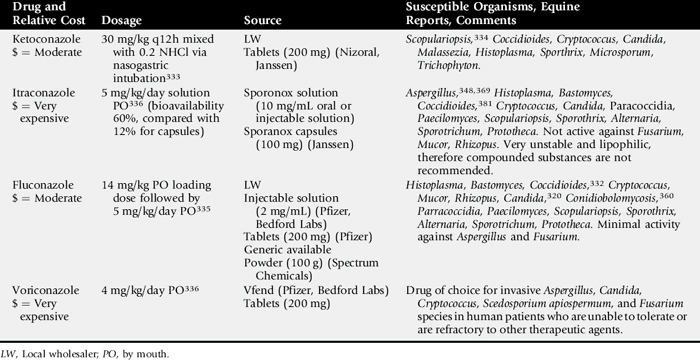


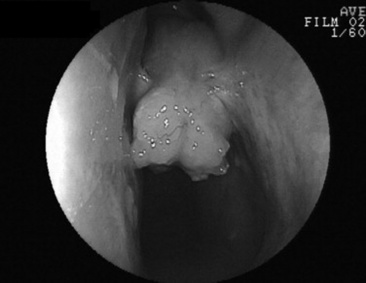
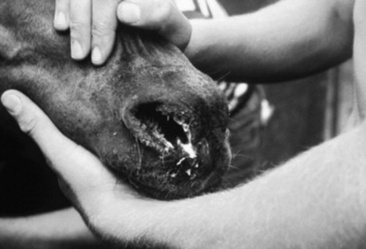
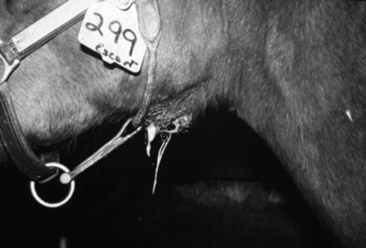

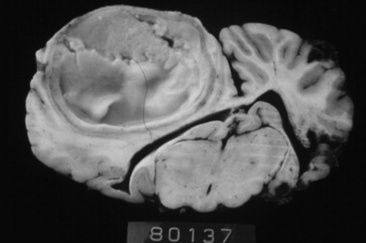

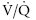 ) mismatch and decreased pulmonary compliance. Alveolar edema causes hypoxemia by creating areas of a low
) mismatch and decreased pulmonary compliance. Alveolar edema causes hypoxemia by creating areas of a low 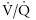 ratio (adequate perfusion but no ventilation), whereas areas with deceased perfusion generate hypoxemia and eventually hypercapnia by increasing dead space ventilation (high
ratio (adequate perfusion but no ventilation), whereas areas with deceased perfusion generate hypoxemia and eventually hypercapnia by increasing dead space ventilation (high 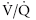 ratio; adequate ventilation with minimal perfusion).
ratio; adequate ventilation with minimal perfusion).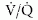 ratios adjacent to normal lung tissue makes mechanical ventilation of patients with ALI and ARDS extremely challenging.
ratios adjacent to normal lung tissue makes mechanical ventilation of patients with ALI and ARDS extremely challenging.