INFECTION
Enteritis caused by infection is frequently a result of Salmonella species, Clostridia species, Neorickettsia risticii, or virulent E. coli. Viruses such as rotavirus can also cause infection in foals with subsequent clinical signs. These agents adhere to and attack mucosal cells. When bacteria bind to the mucosal cells, the release of endotoxin or exotoxins causes the mucosal cells to release cytokines, which signal immunocytes and neutrophils of impending invasion. The gut has a large number of lymphocytes and macrophages in the lamina propria, suggesting constant stimulus by agents or substances.183,256 Although the mucosal cell has an innate ability to resist infection, the communication between the immunocytes and the mucosa is likely the chief defense. The infection can change mucosal function without destroying the mucosal barrier; increased TNF-α concentration has been shown to impair barrier function at the tight junctions.257 The change can cause lack of absorption and/or massive secretion of fluid into the bowel, causing diarrhea.
Interaction of neutrophils with the apical membrane of epithelial membrane after paracellular migration stimulates adenosine receptors by production of 5′-AMP.258 Subsequent functional secretion is caused by stimulation of adenyl cyclase and guanyl cyclase, which catalyze formation of cAMP or cGMP respectively. These cyclic nucleotides subsequently activate protein kinases, which block the NaCl absorptive process in the absorptive mucosal cell and stimulate Cl secretion in the crypt cell.259 Inflammation with release of eicosanoids and bradykinin can also initiate the secretory process. The enteric nervous system can also induce inflammation and secretion in the bowel, which can be inhibited by nerve-blocking agents or indomethacin.259,260 Neuropeptides from efferent nerves are hypothesized to initiate this response by affecting mucosal cells, fibroblasts, and endothelial cells.178,261
When bacteria cause injury by adhering to the mucosal surface and invading the mucosa, the response of the mucosal cells stimulates both the afferent nervous system and an immediate local immune reaction. Cytokines from local macrophages or injured epithelial cells serve as the messengers of recognition, which stimulate macrophages, neutrophils, and eosinophils to migrate to the region of invasion. Lymphocytes are also activated, releasing cytokines including IFN-γ. After an initial delay in mucosal cell apoptosis during bacterial adhesion and invasion, apoptosis is increased, theoretically to increase cell turnover and healing. TNF-α and nitric oxide appear to control mucosal cell apoptosis.218
The inflammatory response to invasion often causes massive mucosal necrosis, with loss of mucosal cells and a massive infiltrate of neutrophils and lymphoid cells. Fibrin exudate creates a cast on the mucosal surface of the bowel. The vasculature is exposed to the bowel lumen, allowing subsequent invasion of the bacteria, both pathogens and others, as well as bacterial toxins. The remaining mucosal cells are stimulated to secrete water via the stimulation of cAMP within the crypt cells. Water is also retained in the bowel because of the lack of absorptive cells; the result is diarrhea.
The infection can involve the other layers of the intestine, although it rarely involves the serosa. Still, some diseases such as hemorrhagic fibrinonecrotic duodenitis—proximal jejunitis (DPJ) affect all layers of the intestine. A causative organism has never been discovered to explain DPJ, but the lesion is similar to that seen with clostridial disease in young swine.262 Similar to reperfusion injury, the inflammatory process creates dysfunction that can last for days. Although long-term complications from DPJ have not been reported, animals with severe cases have not survived because of the lack of healing and severe residual inflammation in the bowel wall.262
PERITONEAL INFLAMMATION
The serosal layer is made of a single-cell mesothelial layer mounted on a layer of connective tissue. This layer is important for maintenance of a lubricated barrier at the bowel surface, necessary for normal intestinal motility and peritoneal cavity fluid exchange. The mesothelial layer attaches to a basement membrane, which is adjacent to an elastic layer. The mesothelial cells vary in type. Some are short and have channels linking the peritoneal surface to the serosa. Others have long microvilli, which appear to help trap fluid on the surface of the peritoneum, providing the chief mechanism of lubrication on the bowel surface. Mesothelial cells react to circulating or intraperitoneal lipopolysaccharide (LPS), infection, and surgery by releasing TNF-α, IL-1β, IL-6, and macrophage inflammatory protein (MIP).263 The response in the serosa is attraction and migration of neutrophils into the serosal connective tissue.
The initial response to serosal injury has been studied in laboratory animals using predominantly scarification of organs or fecal contamination of the peritoneal surface. The response to ischemia or distention of the equine small intestine is similar, though often more severe. During ischemia the mesothelium is rapidly lost, with subsequent serosal swelling with edema. During reperfusion the serosal vasculature becomes more permeable, and polymorphonuclear cells and mononuclear cells migrate through capillaries or venules and infiltrate into the serosal connective tissue layer. Neutrophils accumulate at the basement membrane around vessels and within lymphatics. Fibrin accumulates within the serosa and on the surface. WBCs release oxygen radicals and proteolytic enzymes, resulting in disruption of collagen, the primary ground substance of the serosa. The denuded serosal response includes increased vascular permeability, which allows the surface to be covered with a fibrin clot. After 24 to 48 hours there is massive accumulation of cells, which are predominately neutrophils, within the serosa and at the new surface.
Cytokines are involved in the response to serosal injury and the subsequent healing.264,265 After intestinal anastomosis in rabbits, macrophages increased in number until about day four. Superoxide levels in these cells are high in the first 24 hours. Prostaglandins, cytokine secretion, and plasminogen activator inhibitor activity are known to increase during the first 3 days after peritoneal injury.264 IL-1 and TNF-α are secreted by peritoneal macrophages after injury and appear to modulate peritoneal healing.266 Peritoneal macrophages also secrete plasminogen activator. The secretion of both plasminogen activator and plasminogen activator inhibitor is stimulated by IL-1. After intestinal anastomosis there is a decrease in fibrinolytic activity for the first 5 days. Thereafter, plasminogen activator returns to the normal preoperative level. Normally fibrin will be dissolved by plasmin after plasminogen is activated by tissue plasminogen activator (TPA). The severity of the serosal injury is also related to the reduction of TPA and to the suppression of plasminogen activator normally produced by macrophages. If plasminogen is not activated or is absent, adhesions have a greater likelihood of becoming fibrous and permanent.
As healing progresses, fibroblasts migrate into the fibrin and a layer of granulation tissue forms both beneath and on top of the original basement membrane (Fig. 32-39). Mesothelial cells produce connective tissue growth factor in response to IL-1β, which simulates fibroblast proliferation.190 During this stage IL-1 stimulates and prostaglandin E2 inhibits fibroblast activity in the injured serosa. Primordial stem cells migrate to the surface and change to form a new mesothelium, a metaplasia likely under the control of growth factor from fibroblasts. The greater the inflammatory reaction within the serosa and on the surface, the more fibroplasia occurs, delaying mesothelium resurfacing and increasing the chance for adhesion formation or bowel scarring. Experimentally the severity of adhesions is correlated with increasing concentrations of TNF-α in peritoneal fluid, and antibodies against TNF-α can decrease adhesion formation.266,267 Healing of the serosa may not result in bowel-to-bowel adhesions but can still cause bowel and mesenteric scarring, which can cause luminal narrowing or kinking. The serosa also becomes thickened, which may result in bowel dysfunction or may interrupt the vascular supply, resulting in chronic obstruction.
In the horse the serosal injury is frequently caused by bowel distention. The cellular injury is similar to an inflammatory model except for the vascular sequelae. After small intestinal ischemia there is an initial vascular hyperemia in most of the bowel but a reduction of perfusion to the serosa.231 This same effect occurs during bowel distention and is even greater after alleviation of distention. The edema formation in distended bowel takes place immediately and increases serosal tissue pressure, which exerts extravascular pressure and closes capillaries and venules.231 This continues after bowel decompression, resulting in ischemic injury during reperfusion of the serosa. Reperfusion after decreasing bowel distention also causes serosal endothelial cell swelling and capillary plugging. This helps to explain the adhesions seen in bowel that was distended only proximal to an obstruction or strangulating lesion but was otherwise not involved in an ischemic lesion.
Adhesions resulting from septic peritonitis occur in response to a massive inflammatory response in the serosa. Similar to the response to ischemia, there is neutrophil migration and fibrin deposition in and on the serosa.265 The inflammatory response may be so great that the proteolytic enzymes may prevent adhesion between bowel loops by breaking down fibrin. However, in most cases there is massive fibrin production, and bowel-to-bowel adhesions occur frequently.
BOWEL HEALING
After ischemic damage the mucosa heals rapidly.268-270 Enterocytes migrate along the lamina propria, covering the surface within 24 to 38 hours. In the mucosa the crypt cells are responsible for rapid multiplication and physically forcing the older cells to the mucosal surface. Delayed healing is associated with a massive inflammatory response, which can stimulate delayed epithelial apoptosis and lack of cell replacement.182,271 Connective tissue growth factor stimulates mucosal growth and may be responsible for excessive fibrosis in chronic inflammatory disease.272
Common use of nonsteroidal antiinflammatory drugs (NSAIDs), in particular flunixin meglumine, delays mucosal cell function during reperfusion.270 Although replacement of the mucosal cell lining after ischemia is rapid, in vitro application of flunixin meglumine delays normal return of mucosal barrier function. Increased permeability to endotoxin in flunixin-treated bowel compared with the control intestine may lead to shock or delayed healing of intestine.273
Similarly, the serosal mesothelium also heals rapidly after loss from abrasion, though it takes longer than the mucosa to heal with a functional cell boundary. The mesothelial cells come from multipotent stem cells in the serosal connective tissues. These migrate to the serosal surface and form an initial layer of cuboidal cells before transforming to the more characteristic flattened mesothelial cells. The healing time appears to be dependent on the amount of inflammation and subsequent production of fibrous tissue on the serosal surface.
Although healing is thought to be successful when clinical signs of colic, obstruction, or peritonitis are no longer observed, the latent effect of serosal fibrosis, residual mucosal inflammation, and ganglionitis with loss of neurons may increase the risk of future colic episodes. Horses that have had a colic episode or previous abdominal surgery are three to four times more likely to have a second colic episode than horses that have never had colic.234 Whether chronic inflammation is responsible for recurrent colic has not been determined.
ENDOTOXEMIA
Robert J. Mackay
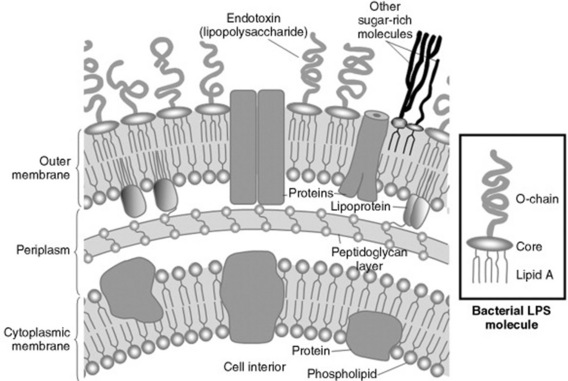
The heat-stable endotoxic activity of Vibrio cholerae identified by Pfeiffer more than 100 years ago resides in LPS, the principal component of the outer leaflet of the outer membrane of all gram-negative bacteria (Fig. 32-40).274 Each LPS molecule has three structural domains: a polar polysaccharide O-region, which projects into the aqueous extracellular environment; a hydrophobic lipid A region, which is largely buried in the bacterial outer membrane; and a core acidic oligosaccharide region connecting the two. The O-region is highly variable, consisting of repeating units each of one to eight glycosyl residues, and contains antigens specific for each bacterial strain; the core glycolipid region is relatively constant among bacteria and mediates most of the toxic effects of endotoxin. On bacterial death or during bacterial proliferation, large (>106-dalton molecular mass) aggregates of LPS and membrane protein are released. It is these protein-lipid micelles that constitute native endotoxin and are to be found circulating in naturally acquired cases of endotoxemia. In recognition of the fact that most endotoxic activity resides in the LPS component, the terms endotoxin (the activity) and lipopolysaccharide (the molecule) are used interchangeably except when a specific or purified LPS is being referenced.275
Endotoxemia literally is the presence of endotoxin in the blood. When the term is used clinically, it implies only the presence of clinical signs typically caused by circulating endotoxin. A survey of diplomates of the American College of Veterinary Internal Medicine and the American College of Veterinary Surgeons found that neutropenia, leukopenia, hyperemic mucous membranes, tachycardia, and fever were the clinical and laboratory values most characteristic of horses with presumptive endotoxemia.276,277 This term should not be confused with either bacteremia, which refers only to the presence of viable circulating bacteria, or septicemia, which is an older term referring to systemic disease caused by circulating microorganisms and/or their products.
The ability to respond to minute local concentrations of endotoxin by mounting vigorous inflammatory responses is well conserved across species.278 Endotoxemia as a clinical syndrome in equine patients was first recognized more than 40 years ago.279 The potential importance of endotoxemia was then shown by reports that intravenous infusion of LPS into horses reproduced many of the adverse clinical signs of diseases such as colitis, metritis, and strangulating intestinal obstruction.280-288 Further evidence for the importance of endotoxemia was the detection of circulating endotoxin in some horses with experimentally induced laminitis289 or intestinal strangulation obstruction290,291 and in horses with naturally occurring gastrointestinal diseases or septicemia,291-296 hemostatic disorders,297 and exhaustion associated with endurance298 or racing299 events. Since the original case descriptions, a large body of review literature has documented the efforts that have been made to understand and, more importantly, treat equine endotoxemia.300-310
Within the last decade, rapid advances have been made in deciphering the molecular pathologies of sepsis in human beings and experimental animals. Much existing dogma has been swept away by this new information, and the pivotal events of endotoxemia in horses with sepsis can now be discerned. Unfortunately, over the same period it has become distressingly clear that the frequent promise of “silver bullet” treatments thrown up by experimental models and preclinical studies has not been realized in large, controlled, multicentered clinical trials in human patients. Therefore it is conceivable that some of the cherished but largely unscrutinized mainstays of equine endotoxemia treatment are of questionable value (at least in the life-saving sense). This section explores the new information that identifies endotoxin as but one of many pathogen- or host-derived signals that elicit global and destructive host responses and focus on treatment strategies that have evidence-based support (at least in human medicine).
ENDOTOXEMIA AND SEPSIS
The extraordinary ferocity of the host response to endotoxin was nicely captured by Thomas311:
“The gram-negative bacteria … display lipopolysaccharide … in their walls and these macromolecules are read by our tissues as the very worst of bad news. When we sense lipopolysaccharides we are likely to turn on every defense at our disposal; we will bomb, defoliate, blockade, seal off, and destroy all tissues in the area …. Cells believe that it signifies the presence of gram-negative bacteria, and they will stop at nothing to avoid this threat.”
Although this picture still accurately describes modern concepts of the early responses to endotoxin, it has become clear that a variety of other pathogen-derived molecules set off similar or identical host responses. For example, toxic shock syndrome resulting from Staphylococcus aureus infection312 and streptococcal toxic shock313 are examples of hyperinflammatory septic syndromes in horses that resemble diseases characterized by endotoxemia. In severe sepsis (including putative endotoxemia in horses), it is likely that the clinical presentation is an aggregate of responses to multiple microbial signals and certain “danger” signals generated by the host itself.
DANGER SIGNALS AND INNATE IMMUNITY
Animals have the ability to recognize distinctive patterns on molecules that signal potential danger. As a group, these molecules that express danger motifs are aptly termed damage-associated molecular patterns (DAMPs).314 Included among DAMPs are microbial signals like endotoxin, collectively termed pathogen-associated molecular patterns (PAMPs), and alarmins, endogenously produced molecules that originate from damaged or inflamed tissues (Table 32-2).315 A limited number of germ-encoded receptor types, both soluble and cell-associated, are dispersed throughout the body to detect potential threats (both septic and nonseptic). These are pattern-recognition receptors (PRRs) and are exemplified by Toll-like receptors (TLRs) on (and in) cells and complement receptor proteins and Hageman factor in plasma.316,317 A list of these and other PRRs is given inTable 32-3. The interaction between DAMPs and PRRs is the initial event in the innate immune responses that result in signs of endotoxemia or other forms of sepsis.
Table 32-2 Partial List of Damage-Associated Molecular Patterns314,315,368
| PAMPs |
Alarmins |
| LPS (endotoxin) |
HMGB1 |
| Lipoprotein |
S-100 proteins* |
| Peptidoglycan |
HSPs |
| Flagellin |
Defensins |
| Lipoteichoic acid |
Cathelicidins |
| Zymosan |
|
| Viral double-stranded RNA |
|
| N-acetyl glucosamine |
|
HMGB1, High-mobility group box 1 protein; HSPs, heat shock proteins; LPS, lipopolysaccharide; PAMPs, pathogen-associated molecular patterns.
Table 32-3 Partial Listing of Pattern Recognition Receptors316,317,368
| Cell-Associated |
Soluble |
| TLR (1-11) |
Hageman factor |
| RAGE |
MBL |
| Nod1/Nod2 |
C3b, Bb |
| CD14 |
Ficolins |
MBL, Mannan-binding lectin; Nod, nucleotide-binding oligomerization domain; RAGE, receptor for advanced glycation end products; TLR, toll-like receptor.
CLINICAL SEPSIS SYNDROMES
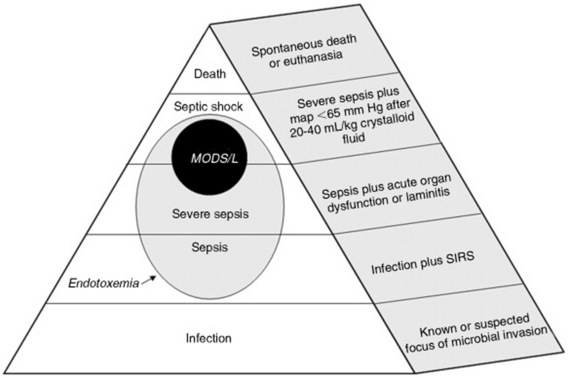
A scheme showing increasingly severe stages of sepsis, with definitions, from local infection to death is shown in Fig. 32-41. The lethality of each grade increases from the base to the apex of the figure. In humans, reported mortality rates for severe sepsis are 25% to 30%318 and for septic shock are 40% to 70%.319 Endotoxemia is shown as a subset of sepsis (at all stages) in acknowledgement of the common pathogenesis of all sepsis syndromes. Dysfunction of two or more organs is termed multiple organ dysfunction syndrome (MODS) and carries an additional increment of lethality in human beings.320 Comparable data are not available for the horse. Definitions for SIRS, organ dysfunction, and laminitis are given inTable 32-4. Note that sepsis is defined as suspected infection plus SIRS; therefore a horse with a strangulating intestinal obstruction and SIRS but no definable infection is still classified as septic or endotoxemic. Horses with signs of mild endotoxemia—leukopenia and fever—fit the definition for sepsis. With the exception of laminitis, these definitions are not validated for the horse but are reasonable extrapolations from accepted human criteria.
Table 32-4 Criteria for Systemic Inflammatory Response Syndrome, Organ Dysfunction, and Laminitis
| SIRS CRITERIA (2 OR MORE OF THE FOLLOWING) |
| Hypothermia |
<98° F or hyperthermia >101.5° F |
| Leukopenia |
<5000/μL or leukocytosis >14,500/μL |
| Tachycardia |
>50 beats/min |
| Tachypnea |
>25 breaths/min or PaCO2 <32 mm Hg |
| ORGAN DYSFUNCTION AND LAMINITIS CRITERIA |
| Neurologic |
Severe obtundation (stupor, semicoma, coma) |
| Renal |
Creatinine >2 mg/dL after ≥20 mL/kg IV crystalloid fluids, or increase of ≥0.5 mm Hg since last measurement |
| Hemostatic |
Platelet count <100,000/μL or aPTT >70 seconds |
| Respiratory |
PaO2 <65 mm Hg, or <75 mm Hg with oxygen supplementation or mechanical ventilation |
| Intestinal |
Absent gut sounds, or absent motility on ultrasound examination |
| Hemodynamic |
Mean arterial pressure <65 mm Hg after ≥20 mL/kg IV crystalloid fluids |
| Hepatic |
Bilirubin concentration >6 mg/dL; GGT >60 U/L with no other explanation |
| Laminitis |
Bounding digital pulses, sensitivity to digital pressure over the coronary band, sensitivity to hoof tester pressure over the sole, Obel grade >1 |
aPTT, Activated partial thromboplastin time; GGT, γ-glutamyltransferase; IV, intravenous; SIRS, systemic inflammatory response syndrome.
The fundamental difference between serious sepsis in humans and the syndromes seen in equids is the propensity for the latter to be associated with laminitis. In the context of sepsis, laminitis is often life-threatening. Because an organ is defined here as “a dispersed or solid tissue that performs a specialized function,” it is probably inappropriate to classify the hoof as an organ as part of an equine MODS definition. It is clear from recent data, however, that the same types of global inflammatory and coagulation disorders that lead to MODS in patients with sepsis also are involved in the pathogenesis of both carbohydrate- and black walnut—induced laminitis.321-325 In light of their likely common pathogenesis, laminitis and MODS are presented together inFig. 32-41 (as MODS/L).
MOLECULAR BASIS FOR ENDOTOXEMIA AND SEPSIS
Endotoxin Enters the Circulation
Although endotoxin is ubiquitous in the environment, both free and as a component of gram-negative bacteria, it normally is excluded from the body by the skin and mucous membranes. If the protective integument or mucosae are subjected to gram-negative bacterial infection or otherwise damaged, endotoxin may reach the blood in sufficient amount (<1 μg of purified LPS in experimental situations) to cause clinical signs. Gram-negative bacterial enterocolitis (e.g., salmonellosis), metritis, pleuropneumonia, wound infection, and neonatal septicemia are common examples. Because gram amounts of free endotoxin normally are safely sequestered within the intestine of the adult horse, damage to the gut wall as a result of local (e.g., intestinal volvulus, infarction, incarceration) or systemic (e.g., hypovolemic shock) causes of tissue hypoxia, inflammation (e.g., DPJ or clostridial enteritis), mechanical trauma (e.g., rectal perforation, prolonged exercise), or intraluminal acidification (e.g., grain overload) is particularly likely to result in endotoxemia. In highly contaminated environments, potentially harmful amounts of endotoxin can be introduced into the lungs via inhalation.326 Endotoxin may even be delivered directly into the blood via parenteral solutions (e.g., homemade intravenous fluids).
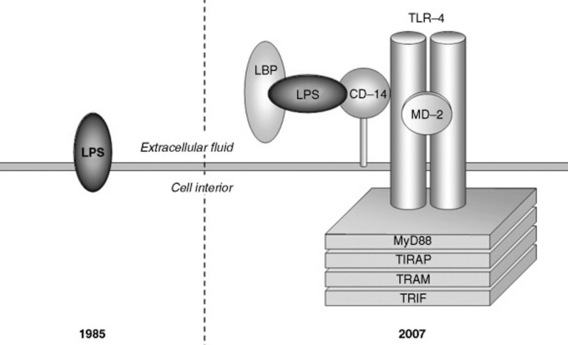
Endotoxin Interacts with Pattern Recognition Receptors
In 1985 almost nothing was known about the way in which LPS molecules interacted with cells of the immune system. In fact, endotoxin was widely believed to enter cells not via engagement of cell-surface receptors but by hydrophobic insertion into the membrane (Fig. 32-42). Since then, sequential discoveries of a plasma LPS-binding protein (LBP; 1989 for humans,327 2005 for horses328), CD14 (1990 for humans,329 2003 for horses330), and TLRs (1998 for humans,331 2005 for horses332) have elucidated the molecular processes of endotoxin binding and signaling (see Fig. 32-42). LPS first interacts with LBP, a normal plasma acute-phase protein; this interaction facilitates binding to the soluble or cell-associated co-receptor CD14. The LPS/LBP/CD14 complex recruits and activates TLR4 dimer and an accessory component MD-2 in preparation for LPS signaling. On ligation of the receptor, the conserved intracellular domain of TLR4 (Toll-IL-1 receptor [TIR]) initiates multiple downstream pathways that culminate in translocation to the nucleus of inducible transcription factors including nuclear factor (NF)–κB and activator protein 1 (AP-1).333 NF-κB binds to consensus sequences on the promoter or enhancer regions of an array of genes whose products are involved in the inflammatory response.333 It has recently been argued that the global activation of TLR4 typical of sepsis requires the actions of endogenous agonists (e.g., proteases).335 Endotoxin also may activate NF-κB via TLR4-independent interactions with β2-integrins, heat-shock proteins, and the intracellular Nod-1 receptor.336 Although endotoxin binds predominantly to TLR4, a still-growing family of TLRs (12 in rodents, 11 in humans, unknown in horses) is available to bind to other DAMPs (Table 32-5).336 During sepsis, this diversity of TLRs allows redundant signaling of inflammatory cells. For example, non-LPS components of gram-negative bacteria may bind other TLRs (e.g., lamellin binds to TLR5, lipoprotein and peptidoglycan to TLR2), whereas PAMPs from gram-positive bacteria or fungi bind different TLRs in the course of polymicrobial sepsis. Tissues subject to attack by mediators produced after the initial round of TLR binding can feed back and amplify the inflammatory response by releasing alarmins (e.g., high-mobility group box 1 [HMGB1]338), which in turn can bind to TLR or other PRRs.
Table 32-5 Ligands for Human Toll-like Receptors316,317
| TLR1 |
Triacyl lipopeptides |
| TLR2 |
Lipoprotein |
| |
Peptidoglycan (gram-positive bacteria) |
| |
Lipoteichoic acid (gram-positive bacteria) |
| |
Zymosan (yeast) |
| Lipoarabinomannan (mycobacteria) |
| TLR3 |
Viral double-stranded RNA |
| TLR4 |
LPS |
| |
Respiratory syncytial virus fusion protein |
| |
HSP70 |
| TLR5 |
Flagellin (Salmonella Typhimurium) |
| TLR6 |
Diacyl lipopeptides |
| |
Zymosan |
| TLR7/8 |
Viral single-stranded RNA |
| TLR9 |
Unmyethylated CpG-containing DNA |
| |
Herpes virus DNA |
| TLR10 |
Not determined |
| TR11 |
Uropathogenic bacteria |
LPS, Lipopolysaccharide; TLR, Toll-like receptor.
Simultaneous with cellular activation, endotoxin interacts with soluble PRRs normally present in plasma. Of particular importance, endotoxin binds to complement proteins to initiate the lectin-dependent and alternative pathways of complement activation and activates coagulation factor XII (Hageman factor) to set off the “contact” system of coagulation.
Mediators are Released
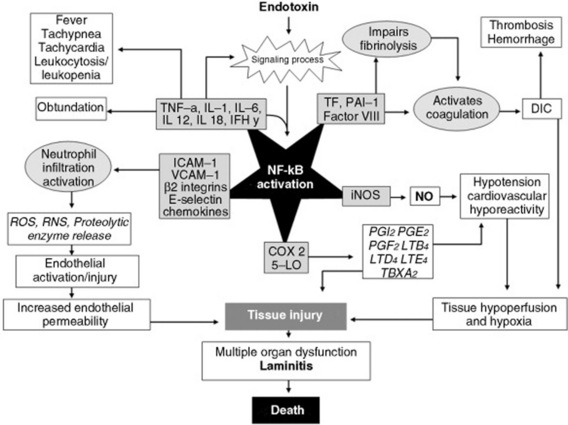
As described earlier, endotoxin engages TLR4 on cells of the innate and adaptive immune systems, especially mononuclear phagocytes (monocytes and macrophages), neutrophils, endothelial cells, and dendritic cells. Pulmonary intravascular macrophages339 and Kupffer cells likely are the most important mononuclear phagocytes in this regard. Endotoxin causes NF-κB activation in these and many other cell types via multiple signaling pathways resulting in the expression of more than 200 genes, many of which are involved in the pathogenesis of sepsis.333 These include genes for proinflammatory cytokines (e.g., TNF, IL-1β, IL-6, IL-8, IL-12, IL-18), chemokines (e.g., IL-8, MIP), type 1 IFNs, procoagulants, adhesion molecules, immunoreceptors (e.g., TNF receptors), enzymes (e.g., elastase), and acute-phase proteins (e.g., fibrinogen).341 NF-κB activity is further amplified by the paracrine actions of these proinflammatory cytokines, and by other DAMPs, cellular hypoxia, cellular necrosis, and chemical stress (including oxidant stress). Two of the cytokines secreted by macrophages, IL-12 and IL-18, stimulate IFN-γ synthesis and secretion from NK (natural killer) and other cells.342 Because IFN-γ is a potent stimulator of both innate and acquired immune responses, it is considered to be a principal link between the two systems.
Endotoxin activates coagulation factor XII (Hageman factor) leading both to liberation of bradykinin and to initiation of intravascular coagulation. Even more important, complement is activated by alternative, lectin-mediated, and classical pathways to yield numerous active peptide products.
THE EARLY (HOT) PHASE OF SEPSIS
The early (hot) phase of sepsis is characterized by inflammation, coagulation, and necrosis. The principal NF-κB events during early sepsis are summarized in Fig. 32-43. This phase has been described as a “cytokine storm,” during which there is flooding of inflammatory, procoagulant, and vasoactive mediators throughout the body. The net effects of these mediators are to promote microvascular injury and hypotension. The singular contributions of many mediators to sepsis is demonstrated by experiments using sepsis models in which blocking or deleting a single mediator has had a positive effect on outcome.
THE LATE (COLD) PHASE OF SEPSIS
It recently has become apparent that many patients with sepsis are profoundly immunosuppressed as shown by lymphopenia, anergy, and susceptibility to opportunistic infections (e.g., pulmonary aspergillosis in horses with enteric salmonellosis).343,344 This immunosuppression has been explained by the reactive production of antiinflammatory mediators in response to the cytokine storm and termed the compensatory antiinflammatory response syndrome (CARS).345 Although many such mediators are produced and actually can have favorable antiinflammatory effects when used as therapy in models of sepsis, it is now clear that widespread apoptotic death of lymphocytes (particularly B-cells and CD4-positive T-helper cells) and dendritic cells secondary to activation of intracellular caspases is largely responsible for sepsis-associated immunosuppression.346,347 During experimental endotoxemia in cats, there also is apoptosis of intestinal epithelial cells,348 raising the possibility that apoptotic processes may affect intestinal permeability in endotoxemic horses. It is interesting to note that macrophages and neutrophils are spared premature apoptotic death; neutrophils actually are prevented from physiologic apoptosis during sepsis and remain viable in sequestered sites.349 The relationship between the two “phases” of sepsis is not clear, and it cannot necessarily be inferred that hot and cold phases occur in the same patient.
THE EFFECTS OF ENDOTOXEMIA AND SEPSIS
During sepsis, large numbers of neutrophils accumulate on the endothelial surfaces of organs undergoing failure, and insult to one organ can trigger the widespread recruitment and sequestration of neutrophils in others. Such a scenario likely underlies the association of laminitis with severe intestinal disease.350,351
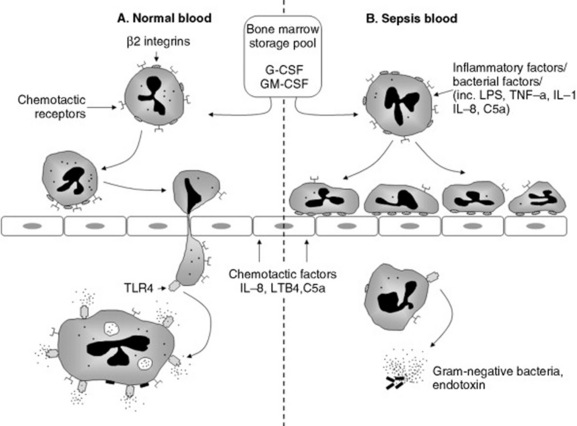
In response to PAMPs (including LPS), inflammatory cytokines produced by macrophages and other cells, and complement peptides, endothelial cells, and neutrophils express selectins.349 Selectins on endothelial cells (E and P) and neutrophils (L) reciprocally engage glycoprotein ligands to “tether” the neutrophil to the endothelial surface. A series of these transient interactions between ligands and receptors allows neutrophils to roll along the endothelial surface (Fig. 32-44). Neutrophil capture is most efficient in areas of low shear force such as the walls of postcapillary venules and in pulmonary capillaries. During rolling, neutrophils are activated or “triggered” by selectins, chemokines, and PAF expressed on endothelial cells. The firm attachment or arrest step of the cascade is mediated by the avid interaction of neutrophil integrins with adhesion molecules of the immunoglobulin superfamily expressed on endothelial cells. During firm attachment the activated neutrophil spreads out and, in the healthy animal, squeezes between the intercellular junctions of adjacent endothelial cells and migrates into tissues up a gradient of chemotactic factors such as microbial chemotaxins, LTB4, IL-8, or C5a. By contrast, when compared with normal neutrophils, those found in septic animals have defective chemotactic responses but bind with greater avidity to the endothelium and to other neutrophils. When cultured, macrophages and neutrophils from patients with gram-negative sepsis are hyporesponsive to LPS, suggesting a functional switch to LPS tolerance during the early stages of endotoxemia (see Fig. 32-44).352 Sequestration of neutrophils on activated endothelium and in neutrophil aggregates accounts for the neutropenia found in most horses with endotoxemia. It is interesting to note that the lifespan of these sequestered neutrophils is prolonged during sepsis because normal apoptosis is prevented.349
Tightly adherent neutrophil-endothelial conjugates formed during sepsis seal off microscopic pockets between the juxtaposed cells into which cellular products can be concentrated. Of particular significance are the reactive oxygen species (ROS) produced as a result of the activation of NAD(P)H-oxidase in neutrophils (respiratory burst) and xanthine oxidase in endothelial cells.354 Digital laminae of horses may be particularly vulnerable to the effects of ROSs because of low content of the endogenous oxidant scavenger superoxide dismutase.355 In the presence of neutrophil granule myeloperoxidase and H2O2, highly toxic hypochlorous acid is formed on the endothelial surface.356 Superoxide anion generated as part of the neutrophil respiratory burst reacts with nitric oxide (NO) from endothelial cells to yield reactive peroxynitrite radicals. Other potentially corrosive substances are contributed by neutrophil granules and include elastase, serine proteases, matrix metalloproteinases (MMPs), and defensins.357 In addition to direct damage caused by membrane lipid peroxidation, ROSs indirectly stimulate the expression of multiple inflammatory, procoagulant, and vasoactive mediators via activation of NF-κB in both neutrophils and endothelial cells. Mediators such as bradykinin, PAF, C3a, C5a, and leukotriene B4 (LTB4) directly increase vascular permeability by promoting active retraction of endothelial cells via phosphorylation of the light chain of nonmuscle myosin.358 Vascular leak facilitates the movement of potentially harmful substances into tissues.
In health the antithrombotic phenotype of endothelial cells is maintained by the presence of low amounts of prostacyclin (prostaglandin I2 [PGI2]) and NO, and surface expression of thrombomodulin, protein S, and protein C complex and TPA.359 During endotoxemia, endothelium supports extrinsic pathway activation because of leukocyte-induced physical damage, expression of the procoagulant tissue factor,360 downregulation of antithrombin-III and protein C, and inhibition of fibrinolysis through expression of plasminogen activator inhibitor 1 (PAI-1).361,362 Additional procoagulant effect may be provided by deposition on the endothelial surface of all of the components of the intravascular coagulation system. Microvascular perfusion is further compromised by sepsis-associated increase in “stiffness” of both RBCs and WBCs.363 Such cells are unable to deform and squeeze through narrow capillaries.
The effect of endotoxemia on vascular tone depends on the stage and severity of disease and the particular organ (vascular bed) considered. Neuroendocrine responses to sepsis lead to the upregulation of predominantly pressor mediators including arginine vasopressin, angiotensin II, serotonin, epinephrine, and norepinephrine. Inflammatory mediators are a mix of vasoconstrictors (thromboxane A2 [TXA2], endothelin, C3a, C4a, C5a) and vasodilators (PGE2, PGI2, adenosine, bradykinin, NO). In animals with serious sepsis, balances of constricting and dilating influences unique to each vascular bed, loss of vasoregulatory tone, and refractoriness of damaged endothelium to vasoactive substances causes maldistribution of blood flow among organs and systemic hypotension.
Because of poor perfusion pressure, direct microvascular injury, thrombosis, and loss of endothelial integrity (capillary leak), ischemia and hypoxia of organs and tissues occur.364
DEVELOPMENT OF GLOBAL TISSUE HYPOXIA
The fundamental event in serious sepsis is the development of global tissue hypoxia. During serious sepsis, widespread microvascular and mitochondrial injury365 decrease oxygen delivery and consumption at the cell, tissue, and organ levels. Oxygen delivery to tissues is a product of cardiac output and oxygen content (which itself is a product of hemoglobin oxygen saturation and hemoglobin concentration). The product of systemic oxygen delivery and the percentage of oxygen extracted (normally ≤25%) by the tissues is the systemic oxygen consumption. The balance between systemic oxygen delivery and consumption is reflected by the mixed venous hemoglobin oxygen saturation (SVO2). SVO2 has been shown in other species to be a useful surrogate for cardiac index as a target for goal-directed therapy.366 Central venous oxygen saturation (ScVO2), obtained through a central venous line, is a reasonable substitute for SVO2 (which must be measured via a Swan-Ganz catheter). Global tissue hypoxia results when systemic oxygen delivery fails to meet the oxygen requirements of tissues.
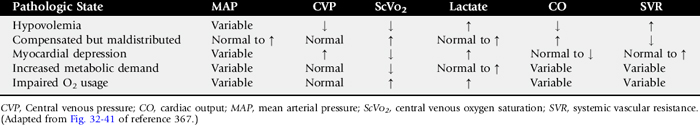
Global tissue hypoxia resulting from cardiovascular insufficiency is the sine qua non of serious sepsis. Various hemodynamic combinations may create a systemic imbalance between tissue oxygen supply and demand367:
 Hypovolemia.
Hypovolemia. Because of decreased preload caused by hypovolemia, concomitant left ventricular dysfunction, and reflex systemic arterial vasoconstriction, early endotoxemia is often characterized by low cardiac output (i.e., hypodynamic circulatory insufficiency).
 Compensated but maldistributed perfusion.
Compensated but maldistributed perfusion. After fluid-electrolyte resuscitation, compensatory mechanisms and low afterload drive transition to a hyperdynamic state. Even with normal or increased cardiac output, perfusion abnormalities may persist owing to regional hypoperfusion associated with derangements in blood flow distribution and loss of vasoregulatory control to vascular beds. This state is often described as
distributive shock. Myocardial depression
Myocardial depression secondary to effects of inflammatory mediators and apoptosis of cardiomyocytes is the primary cause of low cardiac output in 15% of human patients with serious sepsis or septic shock.
368 Increased metabolic demands.
Increased metabolic demands. SIRS increases metabolic demands, as evidenced by increased splanchnic and total body oxygen consumption.
 Impaired oxygen utilization.
Impaired oxygen utilization. The bioenergetics of cellular extraction and use or respiration may be abnormal at least partially because of mitochondrial dysfunction.
365,369These derangements may occur independently of measured hemodynamic parameters. The theoretic components of each of these hemodynamic states are shown inTable 32-6.
SIGNS OF ENDOTOXEMIA
Clinical Signs
The clinical signs of horses given intravenous endotoxin experimentally may range from fever without obvious malaise to multiple organ failure and death. Obviously, signs that are not due exclusively to endotoxin (e.g., severe pain caused by intestinal strangulation or diarrhea in horses with Salmonella colitis) may greatly influence the overall clinical presentation of horses with naturally acquired endotoxemia.
Typically in an adult horse given a moderate sublethal dose of endotoxin (e.g., 0.1 to 1 μg of LPS per kilogram of body weight), an early period of mild tachypnea peaks within 30 minutes and resolves within 2 hours. During this period, mucous membranes are pale. Beginning within 90 minutes of LPS injection, depression, restlessness, and inappetence are present and rectal temperature begins to rise. Auscultable intestinal sounds usually cease during this period and remain depressed for several hours. Intermittent signs of colic usually are seen, including recumbency (usually without rolling). Small amounts of loose feces usually are passed. Heart rate peaks during the stage of maximal abdominal discomfort (approximately 2 hours after administration of endotoxin), then temporarily declines. During this time, mucous membranes become congested, the capillary refill time is prolonged, and a dark “toxic” line may become apparent around the gingival margins of the teeth. Beginning at 4 to 6 hours after endotoxin administration, there is a secondary phase of tachycardia and tachypnea that likely is related to development of systemic hypotension and fever. This secondary phase persists for several hours. Horses presented clinically with mild to moderate endotoxemia usually resemble experimental animals during the period 2 to 6 hours after intravenous endotoxin administration.
At higher LPS doses (e.g., 100 μg/kg) in experimental animals or in patients with severe endotoxemia, signs of circulatory failure and disordered hemostasis dominate the clinical picture. Usually these horses are stuporous and totally anorectic. Signs of dehydration such as reduced skin turgor, dry mucous membranes, and sunken eyes are obvious. As systemic blood flow becomes more compromised, rectal temperature may drop into or below the normal range. Urine output is reduced or nonexistent. There are dark, congested mucous membranes, rapid and weak peripheral pulses, cold extremities, and sweating, and the horse may have muscle tremors and become recumbent.
Vascular damage may be seen as petechial and ecchymotic hemorrhages on mucous membranes. A poor prognostic sign is the development of a hypercoagulation syndrome, during which routine venipuncture or catheter placement initiates thrombosis along the entire visible length of the jugular veins (or other superficial veins). If both jugular veins are thus occluded, there usually is massive swelling of the soft tissues of the head, and associated laryngeal edema may cause signs of upper respiratory tract obstruction. In some horses, thrombosed superficial vessels can easily be palpated through the skin of the legs and abdomen. Infarction of bowel segments or lungs may cause severe clinical signs that are unresponsive to treatment. A rare syndrome of thrombosis of major limb arteries found in young foals is likely a result of sepsis syndrome.370,371 At the time that hypercoagulation syndrome is recognized clinically, there is often evidence of a secondary bleeding tendency (a consequence of platelet and clotting factor depletion and uncontrolled activation of fibrinolysis), seen as prolonged hemorrhage from venipuncture sites and widespread mucosal petechiation. In cases with a severe pulmonary component, there may be hemorrhage into the respiratory tract with progressive tachypnea and dyspnea.
If moderately to severely affected animals survive for more than 24 hours, there usually is visible edema of the ventral abdomen and limbs. Signs of laminitis may first become apparent at this stage and may progress in severity even while the other systemic signs of endotoxemia improve.
Clinicopathologic Signs
Although the measured concentration of endotoxin in blood does not correlate well with severity of clinical signs, demonstration of circulating endotoxin obviously is definitive proof of endotoxemia. In one study, 12% of horses with acute gastrointestinal disease had detectable plasma endotoxin.372 Reported concentrations in these horses were 0 to 30,400 pg/mL with a mean of 218 pg/mL. Experimentally, plasma endotoxin usually is assayed by some variant of the Limulus amebocyte lysate assay. A simple horse-side test for endotoxin that was marketed for use in clinical practice is no longer commercially available.
There is early and profound leukopenia principally caused by neutropenia (usually accompanied by left shift and a toxic appearance of stained cells). Lymphopenia (<1000/μL) is found in the most severe cases and likely reflects sepsis-induced apoptosis and immunosuppression. Adult horses often are hyperglycemic at presentation, whereas neonates with sepsis are usually hypoglycemic. Other abnormalities are nonspecific and reflect altered tissue perfusion and organ dysfunction (see Table 32-4).
In moderate and severe cases of endotoxemia, there also may be evidence of disordered hemostasis; values affected in blood may include any to all of the following: reduction in the circulating platelet count (<100,000/μL), reduction in plasma fibrinogen concentration, prolongation of the activated partial thromboplastin, prothrombin, or thrombin time, increased activity of PAI-1, and increased concentration of fibrin degradation products. In horses with acute gastrointestinal diseases, there also is increased activity in peritoneal fluid of many of the elements of the fibrinolytic system, including TPA. Depletion of key clotting factors can most simply be detected as prolongation of plasma recalcification time.373
NO “SILVER BULLET”
There is no “silver bullet” for treatment of endotoxemia. It has to be admitted that, almost without exception, potentially novel treatments for gram-negative sepsis that have been promising at the experimental level have failed when applied in clinical settings in human beings (and to a limited extent horses). A short list of such trials includes anti-TNF-α,374 IL-1-receptor antagonist,375 ibuprofen,376 PAF antagonist,377 elastase inhibitor,378 nitric oxide synthase (NOS) inhibitor,379 antithrombin III (AT-III),380 and tissue factor pathway inhibitor.381 It is clear that most experimental sepsis models do not replicate naturally occurring sepsis, at least as it occurs in humans. Several of many likely explanations for the disparity between the results of experimental models and large clinical studies are (1) heterogeneity of presentations among enrollees in clinical studies; (2) diversity in genetic susceptibility to sepsis among outbred populations; (3) differences between experimental and clinical study species; (4) differences in timing of potential treatments relative to the onset of sepsis between the two study populations; (5) preponderance of cold sepsis and immunosuppression states in naturally occurring sepsis compared with the “cytokine storm” usually recreated in experimental models.
Even among sepsis models, results for the same candidate treatment are often inconsistent. For example, neutralization and inhibition of inflammatory cytokines usually have salutary effects in LPS-infusion experiments; however, equivalent studies involving the cecal ligation-puncture model in mice (widely considered to be one of the best sepsis models) usually have shown either neutral or negative effects on mortality.382
It is sobering to review the most pertinent available data for the three antiendotoxic drugs that are most commonly used to treat horses with suspected endotoxemia—namely, flunixin meglumine, polymyxin B, and pentoxifylline. Although various NSAIDs have been shown to effectively prevent the signs of LPS infusion in horses,383-392 there is no convincing study that shows NSAIDs actually save lives in patients with naturally occurring sepsis. In contrast, a large multicentered, controlled, masked, prospective study of ibuprofen in humans with sepsis syndrome showed no effect of this drug on the development of shock or the acute respiratory distress syndrome and did not improve survival.393 Polymyxin B and the nontoxic polymyxin B-dextran 70 conjugate PMX622 also safely prevented signs of endotoxemia when given before LPS to otherwise healthy horses394-399; however, the drug has not advanced beyond Phase 1 trials in human patients because experimental studies showed that it did not protect mice if given after intraperitoneal endotoxin.400 Finally, intravenous pentoxifylline significantly, albeit only partially, reduced adverse signs in horses given LPS.401 The usefulness of this finding is called into question by the observation that the lethality-sparing effect of pentoxifylline in endotoxemic mice was removed if the drug was given in combination with indomethacin, a potent NSAID.402,403 It was concluded that upregulation of prostacyclin production by pentoxifylline, which was prevented by concurrent indomethacin, reduced mortality in subject mice by preventing endotoxin-induced leukopenia. It is interesting to note that pentoxifylline alone increased WBC counts in horses. This effect was prevented by flunixin401,405; thus, a potential beneficial effect of pentoxifylline in endotoxemic horses may be neutralized by concurrent NSAID administration.
Evidence-based medicine so far acknowledges only one agent, drotrecogin alfa (activated protein C),406 as able to reduce mortality in patients with sepsis, although there has been some recent controversy regarding the side effects of the drug and the analyses performed in the original study.407,408 Despite theoretic potential in some patients with endotoxemia, drotrecogin is much too expensive to be considered for equine use.
Fortunately, there is powerful evidence to support the value of early aggressive cardiovascular resuscitation. Use of oxygen, fluids, pressors, inotropes, and packed RBCs during the first 6 hours after admission to achieve a sequence of physiologic goals reduced in-hospital mortality of human patients with serious sepsis from 46.5% in the group that received standard therapy to 30.5% in patients given early goal-directed therapy (EGDT).409 Similar protocols now are widely used in emergency rooms throughout the United States.367 Many of the underlying principles of EGDT can be applied to resuscitation of septic horses, especially neonates (see later). It is reasonable to conclude that improvements in cardiovascular support of horses with sepsis will continue to be of much more value than any single “magic bullet” currently available or even on the horizon.
ASSESSMENT OF THE STAGE OF ENDOTOXEMIA OR SEPSIS
Horses with endotoxemia should be staged according to the criteria found in Fig. 32-41 andTable 32-4. It is especially important to recognize that horses with serious sepsis need aggressive intervention in order to survive.
TREATMENT OF ENDOTOXEMIA AND SEPSIS
A prioritized strategy for management of horses with endotoxemia is as follows: (1) cardiovascular resuscitation; (2) laminitis prevention; (3) removal of the cause(s) of endotoxemia; (4) neutralization of circulating endotoxin; and (5) inhibition of endotoxin-induced inflammation. These approaches are most applicable to horses with serious endotoxemia or sepsis.
Cardiovascular Resuscitation
Expansion of blood volume remains the cornerstone of treatment for horses with serious sepsis or endotoxemia. Most cases (e.g., adults with blood lactate of 2 to 4 mmoles/L) can be treated successfully with intravenous balanced polyionic solution according to guidelines for estimating water deficits provided in the fluid therapy section of this chapter. Urination should begin during the rapid replacement of estimated losses. Ideally the fine control of fluid replacement should be based on serial measurements of packed cell volume (PCV) or plasma protein concentration and further guided by following blood lactate concentration. Some clinicians prefer the early use of compatible plasma (5 L for a 450-kg horse, 1 to 2 L for a neonate) or other colloid solutions such as 6% hydroxyethyl starch solution (6% hetastarch in 0.9% saline; Abbott Laboratories, North Chicago, IL) to replace the extravasated colloid lost as a result of capillary leak. Plasma has the advantage of providing immunoglobulin, acute-phase proteins, and anticoagulants (see Laminitis Prevention).
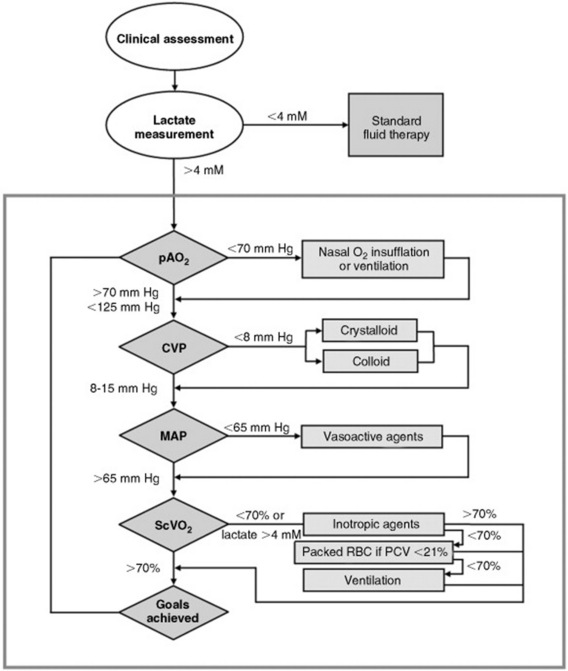
In horses with the most life-threatening forms of endotoxemia and sepsis (lactate >4 mmoles/L [>5 mmoles/L in neonates <24 hours old], MODS/L, septic shock), aggressive hemodynamic monitoring and EGDT are indicated. Because such treatment still carries at best a guarded prognosis for survival, financial commitment often in the range or $5000 to $20,000, and transfer to a referral center, a decision to continue treatment requires that a very clear and realistic discussion of these issues take place with the horse’s owner. Early intervention is essential in sepsis therapy, in critical care parlance, terms such as the “golden 6 hours” and the “silver 24 hours” exemplify this concept.410
Relatively simple equipment and supplies but time-intensive monitoring and treatment are needed for effective EGDT. A central venous catheter (e.g., for 50-kg neonates, two-lumen indwelling catheter, 7 Fr × 30 cm [ES-14702], Arrow International, Reading, PA; 500-kg adults, two-lumen Hickman 9 Fr × 90 cm, Bard Access Systems, Salt Lake City, UT), a blood gas and lactate analyzer, an indirect arterial blood pressure monitor, and a central venous blood pressure monitor (transducer and data display and recorder or water monometer) are required for the full EGDT bundle; however, much can be achieved with a jugular catheter, water manometer, and lactate analyzer. The details of an intensive approach to EGDT for the first 6 hours after admission are shown in Fig. 32-45. ScVO2 has been shown to correlate well in this context with cardiac index. Unfortunately, although measurements obtained via a typical jugular intravenous catheter can be used to approximate arterial lactate concentration and CVP in humans412,413 (although not in dogs and cats414), jugular blood SO2 did not correlate with central measurements in endotoxemic pigs415 and probably should not be used for that purpose. Although labor-intensive, none of monitoring techniques is technically difficult. One advantage of being able to measure both lactate and ScVO2 is that inferences can be made as to the particular hemodynamic derangement responsible for signs of global tissue hypoxia (see Table 32-6).
When the goals of EGDT are met, fluid management should continue according to standard protocols (see Fluid Therapy section) but with adjustments made according to the results of continued monitoring (PaO2, CvO2, lactate, CVP, mean arterial pressure [MAP]). If the blood bicarbonate concentration is <16 mmol/L after EGDT goals are met (or plasma total CO2 concentration is <17 mmol/L), sodium bicarbonate should be given IV to replace calculated deficits. During correction of acidemia, intravenous fluids should be supplemented with potassium (10 to 20 mmol/L) to prevent correction-induced hypokalemia. A maintenance-rate infusion of glucose should be given to neonates with sepsis (4 mL of 5% dextrose—containing fluids per kilogram per hour) and hypoglycemic adults (2 mL of 5% or 0.2 mL of 50% dextrose per kilogram per hour). Glucose concentration should be regularly monitored, and the use of concurrent insulin infusion should be considered, especially if blood glucose is normal or high (see Laminitis Prevention section).
Laminitis Prevention
Adult horses with serious sepsis are at high risk for the development of laminitis. Among the clinical risk factors likely operative in the setting of sepsis are body condition score ≥ 5, being a pony, ≥24 hours since onset of signs of endotoxemia or sepsis, rectal temperature >101.5° F, CRT >2 seconds, or cold extremities. The digital laminae are injured early in the period of hot sepsis, perhaps irreversibly, by processes associated with cytokine storm and global tissue hypoxia. Insulin resistance, hyperglycemia, microvascular injury and thrombosis, and protease activation all may be involved in sepsis-associated laminitis. The laminitis prevention bundle provided in Box 32-3 provides a reasonable preventative strategy, but it must be implemented very early (preferably before increased digital pulses are detected). Global tissue hypoxia is addressed with standard or EGDT fluid resuscitation, small vessel plugging with flunixin (reduced TXA2), pentoxifylline (vasodilation with PGI2, suppression of inflammatory cytokines, increased RBC deformability), and plasma and heparin (increased active AT-III), and hyperglycemia and insulin resistance with continuous-rate infusions of regular insulin and glucose (prevention of the damaging effects of hyperglycemia; possible salutary, glucose-independent effects of insulin). If laminitis is already present, or if it develops during the course of treatment, it should be treated as described in Chapter 38.
Box 32-3 Laminitis Prevention Bundle
Flunixin (0.25 mg/kg every 8 hours)
Pentoxifylline (10 mg/kg PO bid)
Plasma (10 mL/kg)/heparin (4 U/mL plasma)
Insulin (0.01-0.05 U/kg/h)/glucose (0.2 mL 50% dextrose per kilogram per hour) with 2- to 4-hourly measurement of blood glucose concentration
Removal of the Cause(s) of Endotoxemia and Sepsis
Removal of the cause of endotoxemia usually involves both removal of the source of endotoxin and correction of the abnormality that allows access of endotoxin to blood. In some cases a source of endotoxin can be mechanically removed; for example, gram-negative bacteria and associated inflammatory effusion can be drained from pleural or peritoneal cavities or carefully siphoned from the postpartum uterus. Antimicrobial therapy for gram-negative infection also is essential. In general, broad-spectrum bactericidal drugs should be selected because endotoxemic horses may be immunosuppressed. In horses in which endotoxemia is associated with diarrhea or other signs of colitis or typhlitis, the use of antimicrobial drugs is controversial because of their causal association with severe colitis. They probably should be given only in the following situations: (1) the horse is <3 months old; (2) there is suspicion of clostridial or antimicrobial-associated enteritis (metronidazole or vancomycin); (3) there is degenerative left shift or total neutrophil or lymphocyte count of <1000 μg/mL; or (4) there is clinical evidence of dyshemostasis (e.g., jugular thrombosis or abnormal coagulogram). It should be noted that effective antimicrobial therapy could temporarily worsen clinical signs by causing the release of endotoxin from killed bacteria. This possibility should be anticipated and minimized by the timely use of NSAID or other antiendotoxic therapy (see following paragraphs).
When intestinal strangulation is the cause of endotoxemia, surgical correction obviously is of paramount importance. For the purposes of perioperative management, however, it should be noted that resumption of intestinal blood flow could worsen endotoxemia: sequestered endotoxin may be flushed into the circulation through compromised intestinal walls. At least in the case of small intestinal ischemia, the mucosal barrier to endotoxin may be further compromised by ischemia-reperfusion injury when full blood flow is restored by luminal decompression or other manipulation. Again, prophylactic use of NSAIDs and/or ROS scavengers may be warranted.
Neutralization of Circulating Endotoxin
HYPERIMMUNE PLASMA AND SERUM
An antiserum (Endoserum; Immvac Inc., Columbia, MO) and several hyperimmune plasmas (e.g., Polymune J; Veterinary Dynamics, Templeton, CA) produced by immunization of horses against R-mutant endotoxins are used in horses with suspected endotoxemia (in some cases, this is an off-label use). As is the case with studies in human beings and small experimental animals, use of cross-reactive endotoxin antibodies in horses with either experimentally or naturally acquired endotoxemia has yielded conflicting results. In several studies there was impressive reduction of mortality rate or improvement in clinical signs when antiendotoxin serum or plasma was given to horses416-418; however, in other studies no improvement was demonstrated.419,420 Pretreatment of foals with antiserum was associated in one report with significant worsening of clinical response to IV administered endotoxin compared with foals that received no pretreatment.421 These disparate results probably reflect, at least in part, variation in the quality of antisera and experimental conditions; therefore no blanket recommendation can be made as to the clinical use of such products. As evidence of the potential general value of hyperimmune plasmas, it is worth noting the reduction in mortality achieved when antiendotoxin plasma raised against the E. coli mutant J5 was given to bacteremic humans (39% for controls versus 22% for those give antiendotoxin plasma) in a masked, well-controlled study at a single hospital.422 In contrast, subsequent large multicenter studies of two different antiendotoxin monoclonals failed to show beneficial effects.423,424 Hyperimmune plasmas (raised against any antigen[s]) also contain colloid, anticoagulant, and increased amounts of substances such as acute-phase proteins, which might have nonspecific beneficial effect in the setting of endotoxemia and sepsis. Therefore the use of 10 to 40 mL of hyperimmune plasma (of any specificity) per kilogram can be justified in treatment of serious endotoxemia or sepsis.
POLYMYXIN B
Polymyxin B is a broad-spectrum cyclic peptide antibiotic with potent endotoxin-binding activity. Potentially lethal side effects of respiratory paralysis and nephrotoxicity have precluded use of this agent as a systemic antimicrobial drug; however, polymyxin B retains endotoxin-neutralizing capacity at nontoxic dosages. Pretreatment of foals with polymyxin B at a dosage rate of 6000 U (1 mg)/kg significantly suppressed clinical and cytokine responses to intravenous endotoxin without causing toxic side effects.425 Repeated administration to ponies of 15,000 U/kg also produced no sign of toxicity.426 At dose of 5000 U/kg, polymyxin B protected even when given 30 minutes after the start of LPS infusion.427 The results of a pharmacokinetic and pharmacodynamic study of polymyxin B in horses suggested that the drug could safely be given at 6000 U/kg every 8 hours to maintain continuous endotoxin neutralization.428 Horses given polymyxin B at 5 mg/kg as a polymyxin B–dextran 70 conjugate (also known as PMX622) were fully protected from the effects of endotoxin but had a transient hypertensive response to treatment infusion.429 This side effect was prevented by the use of an NSAID. In horses with moderate or severe endotoxemia, consideration should be given to the cautious use of polymyxin B (Polymyxin B sulfate; Bedford Laboratories, Bedford OH) given IV two or three times daily at a dosage rate of 6000 U/kg. Each treatment should be given over at least 15 minutes.
Inhibition of Endotoxin-Induced Inflammation
NONSTEROIDAL ANTIINFLAMMATORY DRUGS
Through inhibition of cyclooxygenase (COX), NSAIDs reduce the formation of prostanoid metabolites (e.g., thromboxanes and prostaglandins) from arachidonic acid and thereby attenuate much of the adverse effect of endotoxin. As stated earlier, it has not yet been established whether or not NSAIDs actually reduce mortality in patients with sepsis. Flunixin meglumine, phenylbutazone, ketoprofen, eltenac, and aspirin are examples of this class of drugs used in horses. When flunixin is administered at 0.25 mg/kg every 6 to 8 hours, endotoxin-induced prostanoid production is prevented, and maximal antiendotoxic effects are produced in experimental situations without obscuring the signs of colic or risking toxic side effects of the drug.430 It should be noted that flunixin does not reduce endotoxin-induced leukopenia. Because there is evidence that aspirin does not prevent endotoxin-induced aggregation of platelets,431 there appears to be no rationale for the common practice of adding aspirin to the NSAID regimen. Most NSAIDs inhibit constitutive COX-1 activity (in addition to endotoxin-induced COX-2 activity), so there is some morbidity associated with their use. There may be gastric ulceration, right dorsal colitis, renal papillary necrosis, and possibly impairment of intestinal motility.432,433 In light of this toxic potential of equine NSAID use, it has been suggested that use of COX-2 selective drugs may minimize side effects while maintaining efficacy. Two NSAIDs with documented analgesic effect in horses, carprofen and meloxicam, have been shown to be COX-2-selective.434 Etodolac, a COX-2–specific drug in dogs and humans, is not COX-2 selective in horses when used at analgesic doses (23 mg/kg PO once or twice daily).435 Also, COX-2 activity does have potentially beneficial effects in horses with sepsis: COX-2 products (e.g., PGE2, PGI2) mediate epithelial restitution in damaged equine colon436 and are thought to be important in maintaining the antithrombotic phenotype of normal endothelium. NSAIDs in the coxib class, which are potently COX-2 specific in humans, have been shown to increase the risk of atherosclerotic cardiovascular disease in humans.437
METHYL XANTHINE DERIVATIVES
Inflammatory cytokine production by macrophages is suppressed in dose-dependent fashion by methyl xanthine derivatives. This effect appears to be caused by phosphodiesterase inhibition and consequent elevation of intracellular cAMP. Pentoxifylline, a drug that is in widespread use in human beings as a hemorheologic agent, has also been shown to increase RBC deformability in horses.438 Pentoxifylline also inhibits TNF production in horse blood and in cultured equine macrophages while increasing secretion of prostacyclin.405,439 Studies in other species suggest that pentoxifylline stimulates production of the antiinflammatory cytokine IL-10, suppresses neutrophil activation, and inhibits activation of NF-κB.441 A pharmacokinetic study in horses has indicated that administration at 10 mg/kg PO two times daily provides serum concentrations equivalent to those used therapeutically in humans.442 The potential for flunixin to antagonize the potential beneficial effects of pentoxifylline was discussed earlier in this section. In light of the strong conceptual arguments for its use, pentoxifylline therapy (10 mg/kg PO bid) in endotoxemia is reasonable.
CORTICOSTEROIDS
The corticosteroid class of drugs theoretically has many useful actions in combating the effects of endotoxemia. These include reduced production of cytokines, inhibition of TNF production by macrophages, stabilization of cell membranes, and prevention of neutrophil activation. It is surprising, however, that neutral or negative effects of steroid use were found in large, multicenter studies of humans with gram-negative sepsis.443 Corticosteroids also are widely believed to increase susceptibility to laminitis in endotoxemic horses, perhaps by increasing the sensitivity of digital vessels to the constrictive actions of circulating catecholamines or by inducing insulin resistance and hyperglycemia.444,445 Use of high-dose corticosteroids is contraindicated in the treatment of endotoxemia in adult horses.
Some human patients with sepsis appear to respond to “physiologic” doses of hydrocortisone.446 Most of these patients had high baseline cortisol concentrations but were thought to be in a state of adrenal insufficiency. In one study, low-dose hydrocortisone was associated with reduced vasopressor use and lower mortality rates.446 This concept is not universally accepted, and the use of low-dose hydrocortisone therapy has not yet been reported in equine patients.448
HEPARIN
The use of heparin in horses with endotoxemia is somewhat controversial. It prevents microvascular thrombosis principally by promoting the anticoagulant activity of AT-III. Unfortunately, heparin cannot reverse existing thrombosis, and because AT-III is consumed during severe coagulopathy, it may not prevent additional intravascular coagulation in such cases. Fresh and fresh-frozen plasma are good sources of AT-III but also provide clotting factors that could potentiate intravascular coagulation. When given at the recommended intravenous or subcutaneous dose of 40 U/kg tid or 150 U/kg bid, respectively, unfractionated heparin causes intravascular agglutination of equine RBCs.449 Therefore it could be argued that the use of heparin might actually exacerbate intravascular cellular plugging. This side effect can be avoided by using low—molecular weight heparin, which is nonagglutinating but retains anticoagulant activity, principally via inhibition of factor Xa.450 The use of heparin should be considered in horses that are at high risk for laminitis (e.g., DPJ or grain overload) or hypercoagulation syndrome (early evidence of dyshemostasis such as abnormal coagulogram or spontaneous venous thrombosis). In the latter setting, heparin should be given with plasma (10 to 40 mL/kg) at a dose of either 200 to 300 IU/kg/day for unfractionated heparin (either divided bid SC or as a continuous intravenous infusion) or 50 anti-Xa IU/kg for low—molecular-weight heparin (SC sid).
SCAVENGERS OF REACTIVE OXYGEN SPECIES
ROSs are thought to cause corrosive tissue damage during endotoxemia and potentiate the production of inflammatory cytokines via activation of NF-κB. Surgical deflation of distended small intestine is thought to lead to ischemia-reperfusion injury, a process that generates ROSs from epithelial xanthine oxidase. The life-saving process of fluid resuscitation in horses with hypovolemic shock may even lead to whole body ischemia-reperfusion. Despite these presumed associations between oxidant stress and the signs of endotoxemia, little effort has been made to intervene therapeutically at this level. There is some evidence that allopurinol, a hydroxyl radical scavenger and inhibitor of xanthine oxidase activity, has positive clinical effect during sublethal endotoxin infusion.451 A recommended dose for allopurinol is 5 mg/kg IV. Because dimethyl sulfoxide (DMSO) has been shown to be a potent scavenger of hydroxyl radicals with efficacy in rodent sepsis models,452 it seems reasonable to use this agent in the treatment of equine endotoxemia. Like allopurinol, DMSO may reduce intestinal mucosal injury after ischemia-reperfusion; to date, evidence for efficacy in this setting has been mixed. DMSO can be given by rapid intravenous infusion (or by nasogastric tube) as a 10% to 20% solution in saline at dose of 0.02 to 1 g/kg every 6 to 12 hours.
Other antioxidants that are used in equine medicine, including vitamin C, vitamin E, and N-acetylcysteine, have shown benefit in rodent sepsis models but have not been evaluated in equine endotoxemia.
Ethyl pyruvate, a stable analog of pyruvate, has been shown to have remarkable protective efficacy in a variety of models of septic and nonseptic shock in rodents and other species.453 Because this agent is inexpensive and can be given in intravenous crystalloid fluids, it would appear to have potential for the treatment of endotoxemia and sepsis. The beneficial actions of this agent have been ascribed to its antioxidant actions.
Miscellaneous Treatments
Naloxone, a narcotic antagonist, at a dose of 0.2 mg/kg, blunted some of the cardiovascular effects of high-dose endotoxin in one study,455 but a dose of 1 mg/kg had no effect in another.456 Of importance, 0.75 mg/kg naloxone caused signs of colic in conscious horses,457 probably by blocking the actions of endogenous β-endorphins at the high affinity μ-receptor. The detergent tyloxapol was remarkably effective in preventing the effects of endotoxin in anesthetized horses.458 The mechanism of antiendotoxic action of tyloxapol is unknown, but the detergent has been shown to have wide-ranging effects on cells and proteins, some of which may preclude its use in clinical cases. For example, the detergent has been shown to inhibit cellular phagocytosis, an important event in innate immunity. Also, this agent induces marked hyperlipidemia (up to 100-fold higher than controls) in horses because of interference with lipoprotein metabolism. Similarly, a phospholipid emulsion effectively prevented adverse effects of subsequent endotoxemia; however, the treatment induced hemolysis sufficient to preclude its use in clinical cases.459 A published report460 on the use of the sulfonyl analog of the alpha-phenyl-N-tert-butyl-nitrone spin trap molecule suggests that this agent was effective in reducing clinical signs in horses given endotoxin. A cautionary note was the observation that some rodents given the same agent at high doses actually suffered enhanced endotoxin-induced mortality.
PAF inhibitors have been effective antiendotoxic agents in some species but have not yet shown much positive clinical effect in horses or humans.461 In dogs and other experimental animals, inhibitors of NO production such as NG-monomethyl arginine reverse endotoxin— or TNF-induced hypotension462; however, NOS inhibitors generally have no protective effect in sepsis models. Furthermore, NO production may not be increased in horses with endotoxemia.463
A promising method of treatment may be the use of ketamine CRI. Ketamine has been shown in vitro to suppress the production of inflammatory mediators by LPS-stimulated equine peritoneal macrophages.464 Constant-rate infusion of ketamine at 1.5 mg/kg/h for 320 minutes achieves blood concentrations compatible with this inflammatory effect and has been shown to be safe and nonsedating.465 The antiinflammatory actions of ketamine appear to be mediated by the actions of adenosine on the adenosine A2A receptor.466 The equine adenosine A2A receptor was recently cloned and characterized pharmacologically and is itself a potential direct target for antiinflammatory drugs.467 Because ketamine inhibits inducible macrophage-type nitric oxide synthase and thus potentially causes vasoconstriction,468 this approach should be used with caution.
Future Treatment Considerations
Current research in horses and other experimental animals suggests that magic bullets will be very hard to find. Most antiinflammatory approaches, even if they are aimed at the “root and trunk” of the inflammatory cascade (e.g., NF-κB activation) do not work consistently in severe sepsis models (e.g., cecal-ligation puncture) or in phase III clinical trials of human patients. It is becoming increasingly clear that much of the morbidity and mortality associated with sepsis results from “cold” sepsis, the state characterized by profound immunosuppression rather than cytokine storm. Affected patients are likely to be injured further by antiinflammatory therapy. There is some indication that IFN-γ, a cytokine that is pivotal in both innate and acquired immunity, can improve survival in immunosuppressed septic mice by preventing apoptosis of lymphocytes.469 The issue of cold versus hot sepsis raises the issue of the need for accurate recognition of the stages of sepsis. Plasma procalcitonin concentration apparently is able to discriminate levels of sepsis and septic versus nonseptic SIRS.470,471 Similarly, HMGB1 levels have been used to define sepsis categories in humans and to provide prognostic information.472 These or equivalent markers need to be introduced into equine sepsis diagnosis.
There remains enthusiasm for strategies aimed at effective means to suppress or scavenge ROSs. In this regard, the remarkable effects of ethyl pyruvate in multiple models of inflammation, which likely mediates via its antioxidant effects, offer considerable promise.453
On the horizon are some different approaches that have the potential to be both effective and affordable. One of the most exciting possibilities is that gene therapy might be used to transfect host cells transiently in a targeted way with genes encoding antiinflammatory mediators (e.g., IL-10, TGF-β) or antisense RNA or ribozymes directed against mRNA of proinflammatory or even antiinflammatory or apoptotic mediators.
MEDICAL DISORDERS OF THE SMALL INTESTINE
Jennifer L. Davis
ULCERATIVE DUODENITIS
Pathophysiology
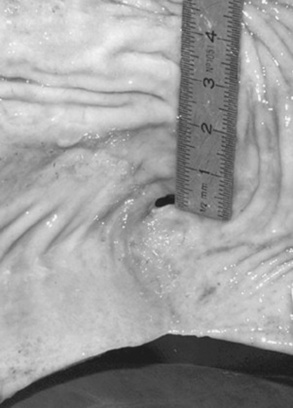
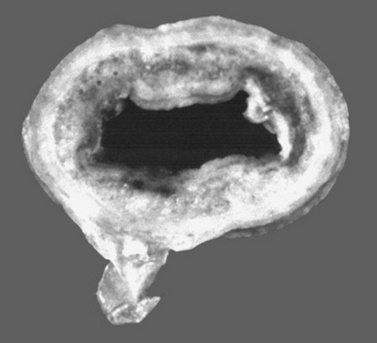
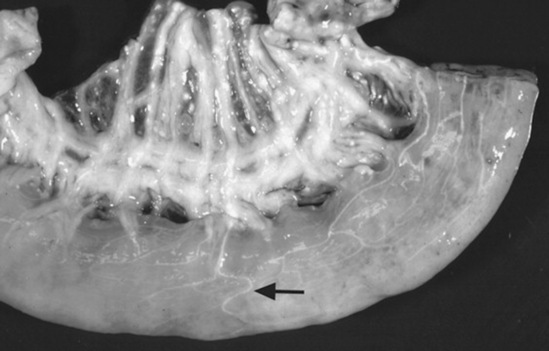
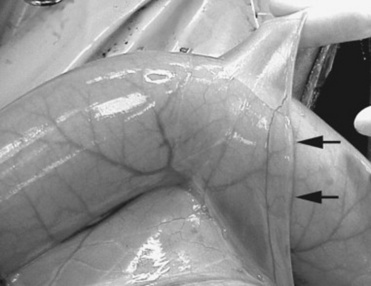
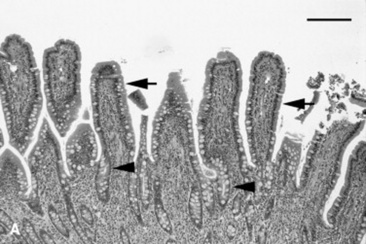
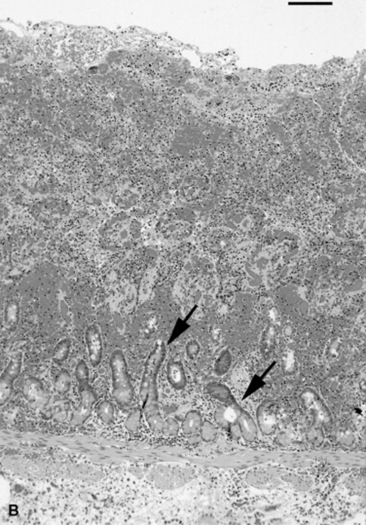
Ulcerative duodenitis most often affects foals and, to a lesser degree, yearling horses. Older horses are rarely affected. Lesions occur primarily in the proximal duodenum and may include erosions, focal ulceration, and diffuse inflammation with or without ulceration. The terms duodenal ulceration and ulcerative duodenitis may refer to differing clinical manifestations of the same problem, and the terms are used interchangeably in this section.
The pathophysiology of duodenal ulcer disease in foals is less well understood than gastric ulcer disease. The disorder is classically considered to be a peptic disease, one in which damage to the duodenal mucosa results from excessive exposure to hydrochloric acid and pepsin. This concept may require revision. Equine duodenal ulcer disease has been presumed to be similar to the disorder in humans, but most cases of duodenal ulcer disease in people are associated with H. pylori infection.480H. pylori bacteria have not been reported in equine gastrointestinal tissues; however, H. pylori colonize only gastric (glandular) mucosa, and infection of the duodenum must be preceded by metaplasia of areas of duodenal mucosa to gastric mucosa. This is thought to occur from chronic peptic injury. In humans the incidence of duodenal ulcer disease increases with age,481 which contrasts with horses, in which duodenal ulcer occurs primarily in animals less than 1 year old.482
We have recognized occurrences of duodenal ulceration and inflammation in which cases were clustered geographically (same farm) and temporally. These foals all had moderate to severe gastric ulceration, and they had extensive inflammation with varying degrees of erosion or ulceration in the proximal duodenum (see Plates 10, 11). A similar temporal and geographic association was reported in two cases of ulcerative duodenitis in yearlings.483 These findings seem inconsistent with a purely peptic insult as the cause for the ulcerative duodenitis. In one report of seven foals with ulcerative duodenitis,484 lesions typically extended into a large area of the proximal duodenum, were characterized by mucosal necrosis, and often had a sharp line of demarcation between affected and more normal-appearing mucosa. In the foals of that report, no common microbial organism other than E. coli was identified, and a cause for the ulcerative duodenitis was not determined. In most foals with duodenal disease the lesions were not focal ulcers but rather appeared as more generalized inflammation. An infectious cause seems likely but has not been identified. In the 1980s rotavirus infection was thought to be associated with gastroduodenal ulcer disease in foals, but most foals with duodenal ulcer disease do not have rotavirus infection.
Duodenal ulcer disease in foals may have a component of peptic injury. The duodenal mucosa possesses some intrinsic properties that are protective against peptic injury, although these are not as elaborate as in the gastric glandular mucosa. The most important factor that protects the duodenal mucosa from acidic gastric secretions may be the sodium- and bicarbonate-rich secretions that probably originate from the pancreas and that will neutralize acid entering the duodenum from the stomach.485
Clinical Signs
The signs of duodenal ulceration or ulcerative duodenitis have been classically described as being the severe forms of gastric ulcer signs,486 and in many cases duodenal and gastric ulcers occur simultaneously. However, many foals with ulcerative duodenitis will not have signs similar to those of gastric ulceration until severe gastric ulceration has occurred. Thus the primary signs of duodenitis can be nonspecific; they include fever, mild to moderate abdominal discomfort, mild obtundation, and diarrhea. A CBC will often reveal peripheral blood leukocytosis and hyperfibrinogenemia.
Gastric ulceration frequently occurs along with duodenal ulceration and may be secondary to physiologic or anatomic obstruction to gastric emptying. The gastric ulceration tends to be severe (see Plate 12), often leading to gastroesophageal reflux and esophagitis. Foals with esophagitis often exhibit ptyalism. In general the sequelae of duodenal ulceration are more severe than those of primary gastric ulceration. Complications of duodenal ulceration include duodenal perforation with peritonitis or adhesions, duodenal stricture with complete or partial obstruction (Figs. 32-46 and 32-47), ascending cholangitis and hepatitis, and ascending pancreatitis.
Diagnosis
Duodenoscopy is the most specific means of diagnosis. It requires an endoscope with at least a 200-cm working length in foals up to 6 months of age, and a longer endoscope is required in older foals to examine the duodenal mucosa. Because of the size of the stomach and the anatomic configuration of the duodenum in foals, it is usually not possible to advance the endoscope past the duodenal ampulla. Occasionally the endoscope can be advanced into the descending duodenum.
A diagnosis is most readily made in cases in which lesions are diffuse or located within the ampulla. Excessive enterogastric reflux of bile through the pylorus is consistent with duodenal dysfunction. Ulceration at the pylorus or pyloric antrum may accompany duodenal ulceration and thus provide an indication of potential duodenal involvement. Severe gastric ulceration in foals should alert the endoscopist to the potential for duodenal involvement. In such cases, oral H2 receptor antagonist therapy may be less effective in resolving gastric lesions than in cases of primary gastric ulceration, because of delayed gastric emptying secondary to duodenal ulceration. Therefore if a foal has received such treatment before endoscopy and if gastric ulceration is severe, suspicion of duodenal ulceration should increase.
Other diagnostic procedures that may be helpful include evaluation of peritoneal fluid; serum liver enzymes, particularly biliary-associated enzymes (γ-glutamyltransferase [GGT], ALP); serum bile acids; and radiography. With severe duodenal ulceration, survey radiographs of the cranial abdomen may reveal accumulation of fluid within the stomach and gas ascending the biliary ducts.487 If barium contrast medium is placed into the stomach, complete emptying is usually delayed (>2 hours) and an irregular mucosal border may be noted in the descending duodenum. It should be recognized that in most cases radiography would not contribute to a diagnosis of duodenal ulcer per se, although duodenal stricture may be noted. If the descending duodenum is to be imaged, the volume of contrast medium placed in the stomach should not exceed 0.5 to 1 L in a foal and 1 to 2 L in a weanling or yearling, or the proximal descending duodenum will be obscured by contrast medium within the stomach.
Treatment
The effectiveness of treatment of duodenal ulceration or ulcerative duodenitis depends on the extent and severity of ulceration and the absence of complications, particularly perforation and stricture of the duodenum. Treatment objectives are to decrease duodenal inflammation, treat secondary gastric and esophageal ulceration, promote gastric emptying, and treat related problems such as peritonitis. If duodenal ulceration is confirmed or even suspected on the basis of clinical signs, treatment should be aggressive.
In acute cases of ulcerative duodenitis there usually is a pronounced lymphocytic infiltration of the mucosa. In more chronic cases there is a mixture of neutrophils, macrophages, fibroblasts, and fibrinonecrotic exudate. Definitive antiinflammatory therapy has not been described for these cases, and the use of corticosteroids or nonsteroidal antiinflammatory medications is controversial because they may worsen gastric ulcer disease through inhibition of protective prostaglandin synthesis.
Suppression of gastric acid secretion is still an important objective in the treatment of ulcerative duodenitis in foals, because most affected foals have gastric ulcers. Initially, acid suppression should be accomplished via parenteral administration of H2 antagonist (cimetidine, 7 mg/kg IV q6h, or ranitidine, 1.5 mg/kg IV q8h). Oral medications are unlikely to be adequately delivered to and absorbed from the small intestine in the first days of treatment. Gastric emptying can be enhanced with bethanechol (0.02 mg/kg SC q6–8h or 0.35 mg/kg PO q8h when the foal can consume orally). In foals that have severe duodenal disease or that have required surgery, bethanechol has been given for up to 3 months. Once the foal can accept oral medication, it should be treated with the proton pump inhibitor omeprazole at a dose of 4 mg/kg once daily for the paste formulation. It should be noted that in the 24 hours after the first dose, acid suppression is incomplete, and maximal suppression of acid secretion is achieved between days 1 and 5.488 Therefore a common practice is to administer an H2 antagonist IV for the initial 2 days of treatment with omeprazole in foals with duodenitis.
Misoprostol is a synthetic prostaglandin E1 analogue that has been successfully used in treating duodenal ulcerations in humans. Doses of 2 to 5 μg/kg q8-12h can be used in horses, although side effects may include abdominal pain and diarrhea. Sucralfate promotes duodenal mucosal healing in humans.489 The dose of sucralfate that is effective in humans with duodenal ulceration ranges from 1 to 2 g two to four times daily. Foals treated for duodenal ulceration that do not have impaired gastric emptying should be administered 2 to 4 g of sucralfate three times daily. Sucralfate should not be used as the sole therapeutic agent for duodenal ulceration.
Foals with duodenitis must usually be prevented from nursing or eating for 1 to 3 days. During this time, parenteral feeding should be considered. Depending on the age of the foal, administration of parenteral nutrition should provide 40 to 60 kcal/kg/day.
If medical therapy is ineffective or if sequelae of duodenal ulceration cause complications, surgical intervention may be required. Gastroenterostomy has been reported to be effective in some cases through bypassing the affected portion of duodenum and allowing for an alternative route for gastric emptying.487 However, short-term survival and long-term quality of life and use are often unsatisfactory. Patients that have a successful surgical outcome require long-term aftercare and usually require long-term maintenance acid suppression and treatment with a prokinetic drug until gastric emptying is normalized. Therefore an owner should be prepared to make a significant time and financial commitment before surgery is considered.
DUODENITIS–PROXIMAL JEJUNITIS
DPJ, also known as anterior enteritis or proximal enteritis, describes a clinical syndrome that is characterized by inflammation and edema of the duodenum and proximal jejunum, excessive fluid and electrolyte secretion into the small intestine, and, consequently, large volumes of enterogastric reflux. The syndrome of DPJ was first described in 1982490 and was more fully characterized in 1987.491 A subsequent report492 described clinical and clinicopathologic parameters in horses with DPJ that differed somewhat from cases in the 1982 report, suggesting that either the cases were of similar etiopathogenesis but of different severity or that the cases were of different etiopathogenesis with the only similarity being the segment of bowel affected. A diagnosis of DPJ is often applied to cases in which there is abdominal discomfort, small intestinal distention, and excessive enterogastric reflux without obstruction, yet it is unclear whether all of these cases lie along a spectrum of severity of DPJ or whether there are several disease entities that affect the proximal small intestine and that share clinical features. The latter seems more likely.
Pathophysiology
In horses with DPJ, lesions are consistently found in the duodenum, but the severity and frequency of lesions in the jejunum are variable. Serositis is a consistent finding, characterized by bright red to dark red petechial and ecchymotic hemorrhages on the serosal surface.491 Histologic lesions include hyperemia and edema of the mucosa and submucosa, villous epithelial degeneration, epithelial cell sloughing, neutrophilic pleocytosis, hemorrhages in the muscular layers, and fibrinopurulent exudation on the serosa.
With DPJ there is an increased volume of duodenogastric reflux, typically 50 to 100 mL/min. This reflux has been considered to result from increased intestinal fluid secretion and decreased motility. Mechanisms of intestinal fluid secretion include passive transmucosal exudation, secondary to mucosal and submucosal inflammation and characterized by a protein-rich fluid secretion, and active fluid secretion, caused by increased cyclic nucleotides and characterized by fluid with a high electrolyte and low protein content. The components of fluid in the intestines of horses with DPJ have not been characterized, but it is likely to result from a combination of passive and active secretion. In some horses the hemorrhagic nature of the gastric reflux implies increased capillary permeability of the duodenal mucosa, whereas in other horses the watery nature of the reflux, the presence of serum electrolyte disturbances,493 and the absence of peripheral hypoproteinemia are most consistent with an active secretory process.
Another potential source of the large volume of fluid secreted into the proximal small intestine and refluxed into the stomach is the pancreas. Normally there is periodic orad movement of duodenal contents into the stomach, which has been observed endoscopically and has been documented by collecting gastric contents with and without pyloric obstruction.485 The duodenal contents have a large component of water, sodium, and bicarbonate, as well as bile salts from the liver. These secretions are presumed to originate from the pancreas, as well as the liver, and pathology of either of those organs may contribute to the pathophysiology in cases of DPJ.
Suppurative cholangiohepatits has been reported in cases of small intestinal inflammation secondary to DPJ.494,495 The pathophysiology behind this observation may be related to an increased luminal pressure in horses with DPJ, increasing the likelihood of intestinal content regurgitation into the bile ducts. It is also possible that horses with DPJ absorb inflammatory mediators or bacterial products from the small intestine via the portal blood flow or systemic circulation.
Although the exact cause of DPJ is not known, several bacteria and toxins have been implicated. C. difficile has been frequently implicated in causing the disease. One prospective study cultured toxigenic species of C. difficile from the reflux of 10 out of 10 horses diagnosed with DPJ, and only 1 of 16 horses diagnosed with other causes of nasogastric reflux. Of the strains cultured from these horses, 8 of 10 produced both A and B toxins, whereas the remaining two produced only toxin B.496 This is significant in that toxin B as been shown to cause inhibitory electromechanical disturbances to smooth muscle in the small intestine, which may be a possible cause of ileus in these horses.497 Toxin A has also been shown to promote inflammatory cell infiltration into the smooth muscle layers.498 Influx of neutrophils and release of inflammatory mediators in the intestinal wall have been shown to activate nitric oxide pathways, which results in inhibition of the enteric nervous system, increases in sympathetic tone, and a subsequent reduction of contractile activity in the gut.499C. perfringens and Salmonella species have also infrequently been associated with DPJ, but the significance of these pathogens remains unknown.
Fusarium moniliforme has been cultured from the feed of horses with naturally occurring DPJ. Under experimental conditions, F. moniliforme producing fumonosin B1 mycotoxins caused lesions consistent with DPJ.500 Neurologic lesions were also present in the horses with DPJ in that study, however, and both of those horses died with lesions consistent with equine leukoencephalomalacia. Cantharidin toxins can also cause reddening of the mucosa in the small intestine, as well as excessive gastric reflux501 and may be a cause in some cases. It is possible that this one syndrome has multiple initiating causes and that no one causative agent will ever be definitively identified.
Clinical Signs and Differential Diagnosis
The major differential diagnoses for DPJ include simple or strangulating small intestinal obstructions. Differentiation can be extremely difficult in some cases and may delay surgical intervention in cases of small intestinal obstruction, to the detriment of the patient. The criteria used to discriminate between DPJ and obstructive lesions include degree of pain, presence of fever, and changes in hematologic parameters and abdominal fluid.
Horses with DPJ have a history of an acute onset of moderate to severe abdominal pain that often is followed by varying degrees of depression. Nasogastric intubation yields a large volume of enterogastric reflux, which is frequently orange-brown in color, with a fetid odor. Palpation per rectum reveals multiple loops of mild to moderately distended small intestine. The initial volume of reflux may range from as little as 4 to 5 L to up to 32 L. The duration of the reflux may be as short as 24 to 48 hours, but it usually lasts 3 to 7 days. Horses are often febrile (rectal temperature greater than 38° C [101° F]) and dehydrated and have injected mucous membranes, prolonged capillary refill time, diminished intestinal sounds, tachycardia (>60 beats/min), and tachypnea.490-493,502,503 Although abdominal pain usually abates after gastric decompression, most horses remain depressed, which perhaps is the most consistent and characteristic clinical sign of the disease. If the fluid that accumulates in the proximal intestinal tract is not removed periodically, signs of abdominal pain recur.
Assessment of the degree of small intestinal distention and the thickness of the intestinal wall can be useful indicators. Often, horses with DPJ have generalized distention of small intestine, but when palpated per rectum the intestine does not feel taut. In many cases of small intestinal obstruction the bowel will feel tightly distended, but this is not universally true. Ultrasonography can be used, both transrectal and transabdominal, to determine the diameter of the small intestine, evaluate contractions, and measure the thickness of the wall of the intestine. With acute obstruction one can see several segments of small intestine that are 6 to 10 cm in diameter, have no contraction, and have a wall diameter of 3 to 5 mm. With DPJ small intestinal diameter may be less, and the thickness of the intestinal wall may exceed 6 mm.
Culture of the reflux for Clostridium and Salmonella species can be attempted. This can be difficult, however, given the special conditions required for anaerobic cultures, as well as the intermittent shedding of Salmonella species into the gastrointestinal tract of normal horses. Also, the large volume of reflux in these cases may dilute the bacterial population to the point where isolating low numbers of bacteria is difficult.
Clinicopathologic Findings
Clinical laboratory findings include increased PCV and total plasma proteins (hemoconcentration) and a metabolic acidosis in longstanding or severe cases. Abdominocentesis often reveals an elevated peritoneal fluid total protein concentration and a mild to moderate increase in the peritoneal WBC count (>5000 cells/μL).502 The peritoneal fluid is usually yellow and turbid, but in severe cases diapedesis occurs, resulting in a serosanguineous color. An abdominal fluid total protein concentration ≥3.5 g/dL is associated with a poorer prognosis.502 The WBC count in the peripheral blood may be normal or increased.491,492 In addition, hypocalcemia, hyponatremia, hypochloremia, hypokalemia, and acid-base alterations have been reported in horses with DPJ.493 An elevated anion gap may be present, secondary to decreased calcium and magnesium or increased lactate or albumin concentrations.502 Increases in the anion gap to ≥15 mEq/L have also been associated with a poor prognosis.
Elevations in liver enzymes, particularly GGT, may be seen in horses with DPJ and may be a useful way to help differentiate between DPJ and strangulating lesions of the small intestine.494,495 A study examining a large series of DPJ cases retrospectively to determine the prevalence of hepatic damage in horses with small intestinal inflammation showed that horses with DPJ had significantly higher hepatic enzyme activities than the control group of horses with small intestinal strangulating obstruction (SISO).495 Over 50% of horses with PE had biochemical evidence of hepatic disease (high GGT, aspartate aminotransferase [AST], or ALP activity). Horses with DPJ had a 12.1-fold higher risk of having a high GGT activity and a 1.8-fold risk of having a high AST activity than horses with SISO. AST activity in horses with PE ranged from 133 to 2994 IU/L (reference range 215.8 to 365 IU/L), GGT ranged from 7 to 117 IU/L (reference range 6.2 to 19.1 IU/L), and ALP ranged from 86 to 1103 IU/L (reference range 69.4 to 293.7 IU/L). Bile acid concentrations were rarely abnormal, indicating that hepatic failure was uncommon. Histopathologic evidence of liver pathology was a common feature in the horses with PE that had either biopsies or necropsies performed. Centrilobular necrosis and inflammation were noted in some cases.
Treatment
Because the causative agent(s) of DPJ are unknown, treatment remains empiric and consists of aggressive supportive therapy. The continuous production of enterogastric reflux requires gastric decompression every 1 to 2 hours to relieve pain and to prevent gastric rupture. Approximately 4 to 8 L of malodorous gastric fluid can be collected during decompression. The stomach should be decompressed frequently, regardless of whether or not the horse is showing signs of abdominal pain, as these signs may be masked by severe depression or the administration of analgesic or antiinflammatory medications. Horses should receive nothing by mouth until small intestinal function has returned, recognized clinically by cessation or reduction of the nasogastric reflux to 1 to 2 L over a 4-hour period and increased frequency of borborygmi. The time necessary for gastric decompression varies with each individual patient. Repeated rectal examinations after the first day of therapy will inconsistently reveal distended loops of small intestine, depending on the frequency of removal of the reflux and the severity of the initial lesion. Ultrasonography may reveal fluid-filled small intestine when such bowel is not discernible by rectal palpation. Loops of small intestine are most frequently visualized in the ventral flank area, near the udder or prepuce; therefore this area should be examined in all cases of suspected small intestinal distention.
Intravenous administration of a balanced electrolyte solution is necessary to maintain intravascular fluid volume and cardiovascular performance. In some horses even rapid administration of fluid fails to adequately restore and maintain intravascular volume because of enteric fluid losses that can be as great as 8 L hour. In addition, the very large volume of isotonic crystalloid fluid that must be given IV to keep pace with enteric fluid losses with DPJ may accelerate the flux of fluid from the vasculature into the intestinal lumen because of reduced intravascular oncotic pressure, increased capillary perfusion pressure, and increased capillary permeability in the inflamed intestine. Consequently the balance between adequate hydration and the volume of enterogastric reflux obtained requires careful and frequent monitoring.
Administration of colloid solutions may be of benefit in preserving intravascular volume without promoting enterogastric reflux. The most frequently used colloids in the horse include hyperimmune plasma, and hydroxyethyl starch solutions. Plasma products require a large volume of administration to exert a colloidal effect, which is cost prohibitive in many cases. Smaller doses, however, may have a beneficial effect in horses with DPJ, particularly in animals showing signs of sepsis or endotoxemia. Hydroxyethyl starch solutions have been shown to significantly increase plasma oncotic pressure in ponies administered 10 mL/kg504 and represent a reasonably priced alternative to plasma. These solutions have been associated with changes in the hemostatic profile in normal ponies at higher doses of 20 mL/kg505 and should be used only with caution in horses already at risk for coagulopathies.
During the initial hours of therapy, even aggressive intravenous fluid administration may result in only moderate clinical improvement. A positive clinical response, as evidenced by improved hydration status, decreased heart rate, decreased enterogastric reflux, improved attitude, and improvement in parameters reflecting kidney function (decreased blood urea nitrogen [BUN] and serum creatinine), correlates with resolving intestinal inflammation.
NSAIDs should be used judiciously to avoid masking the clinical signs of a potential surgical lesion. Flunixin meglumine can be used at a dose of 0.25 to 0.5 mg/kg every 6 hours to reduce the untoward effects of arachidonic acid metabolites.
Antimicrobial agents are typically administered to horses with DPJ, although the necessity for antimicrobial treatment in horses with DPJ is uncertain. Given the association of DPJ with C. difficile, administration of intravenous penicillin (22,000 to 44,000 IU/kg q6h) is warranted. Metronidazole also has excellent activity against C. difficile; however, administration is difficult because nothing can be administered per os. Rectal administration of metronidazole has been studied506 and can be used in these cases. The dose and frequency of administration should be increased because bioavailability after rectal administration is much less than after oral administration (30% vs. 74%, respectively). Broad-spectrum antimicrobial treatment may be indicated in horses with low WBC counts, but care must be taken in selecting an antimicrobial to avoid potential adverse effects, particularly nephrotoxicosis with aminoglycosides in a dehydrated patient with compromised renal function.
Horses with DPJ may have to be kept from eating for several days and are often in a hypermetabolic state; therefore they rapidly develop a negative energy and nitrogen balance. In these horses parenteral nutritional support should be considered. Parenterally administered solutions containing glucose, balanced amino acid solutions, lipid emulsions, balanced electrolyte and trace minerals, and vitamins have been administered to adult horses with a variety of intestinal disorders, including DPJ. Providing for part of the horse’s nutritional requirements (8000 to 12,000 kcal/day) is possible with glucose—amino acid solutions that are of moderate cost. The rationale for this treatment is that through provision of nutritional support to an anorectic, severely ill horse, the healing process will be facilitated, complications will be reduced, and the duration of hospitalization may be shortened. Thus the overall cost of providing nutritional supplementation, enteral or parenteral, to horses with DPJ may well be offset by quicker recoveries and diminished requirements for other costly treatments.
Prokinetic agents may also be useful in cases of DPJ. Of the available prokinetics, lidocaine is used most frequently.507 A loading dose of 1.3 mg/kg slow intravenous bolus followed by a continuous infusion of 0.05 mg/kg/hr has been shown to shorten the time of reflux and decrease the hospital stay in horses with DPJ.508 It may do this by decreasing sympathetic tone, acting as an analgesic agent, or decreasing granulocyte infiltration in the intestinal wall. Its use should be reserved for horses in which a surgical lesion has been ruled out, as it can very effectively mask intestinal pain. Horses should be refluxed frequently during the infusion and checked for other signs of complications, such as laminitis, routinely. Metoclopramide, erythromycin lactobionate, and cisapride are also used in cases of DPJ.507 The efficacy of prokinetics in this disease is debated, mainly because they require a healthy intestine in order to exert an effect. A more in-depth discussion of prokinetic agents can be found in the section on gastrointestinal ileus in this chapter.
Medical therapy is sufficient in most cases of DPJ. In patients with prolonged nasogastric reflux (>7 days), excessive fluid losses that cannot be corrected with conventional fluid therapy, or clinical and laboratory findings strongly suggestive of an intestinal obstruction, surgery should be considered. Animals with severe cases of DPJ may develop infarction of a segment of the small intestine that requires surgical removal. Surgery is used to make the diagnosis and potentially to alleviate enterogastric reflux by providing an alternative route for fluid that accumulates in the small intestine. On entrance into the abdominal cavity, dilated small intestine is immediately apparent. After the extent of the diseased intestine is determined, a segment of normal distal jejunum is laid side to side to the proximal diseased intestine in an isoperistaltic fashion, as far proximal on the affected bowel as possible without extending to bowel that cannot be removed from the abdominal cavity. A small 1- to 1.5-cm hand-sewn anastomosis can then be made between the two segments of intestine.509 This provides an adequate stoma for direct intestinal decompression while minimally compromising the digestive and absorptive capacity of the small intestine. Potential complications of this procedure include development of an intestinal incarceration through the loop that is formed and the development of small intestinal adhesions.
Complications
Complications of DPJ include septic peritonitis, myocardial and renal infarction, aspiration pneumonia, adhesions of the proximal small intestine, and laminitis. The prognosis for surviving the initial intestinal insult is good in cases of DPJ. The death and function losses from this disease are more commonly related to the secondary complications such as laminitis and intraabdominal adhesions. In one report, laminitis occurred in 28% of horses with DPJ, and associated factors were high body weight and hemorrhagic gastric reflux.510 Laminitis prophylaxis is routinely incorporated into the medical therapy and can consist of a variety of treatments, none of which is proven to be effective. These include NSAIDs, topical glyceryl trinitrate, and DMSO (200 mg/kg given as a 10% solution in normal saline). Horses that received heparin as a prophylactic treatment for laminitis were less likely to develop clinical laminitis than horses that did not receive heparin in one study.510
PROLIFERATIVE ENTEROPATHY
PE is an infrequently diagnosed disorder of the small intestine of weanling foals or yearlings caused by Lawsonia intracellularis, an obligate intracellular pathogen. Alternative names for this condition include proliferative enteritis, proliferative ileitis, and intestinal adenomatosis. The hallmarks of PE are chronic wasting with severe hypoproteinemia accompanied by grossly thickened small intestine with mucosal ulceration. The disease can occur in individual animals or as a herd outbreak among young animals.511-515
Pathophysiology
The mechanism of enteritis after infection with L. intracellularis involves invasion of the proliferating crypt cells in the ileum, causing excessive mitotic division and severe hyperplasia.516 The hyperplastic mucosa becomes grossly thickened and develops a corrugated appearance. As would be expected, this thickening of the mucosa, along with the proliferation of immature crypt cells rather than mature villous cells, leads to a limited brush border development and a decreased absorptive capacity, which results in the weight loss and hypoproteinemia present in these cases. The organism can divide within the infected cells and migrate up to the mucosal layers as the cells proliferate and advance. The main differential diagnosis in these cases is R. equi enteritis, which can also cause ulceration in the areas of Peyer’s patches throughout the small intestine, cecum, and colon.
A genetic predisposition to L. intracellularis infection has been proposed. A recent study showed that polymorphisms in several important immune response genes in foals were related to the fecal shedding of L. intracellularis organisms.517 None of these foals had clinical disease consistent with PE, however, and the significance of these gene polymorphisms is still unknown.
Clinical and Laboratory Findings
PE is a chronic, progressive disorder, and therefore affected animals are typically not presented until the disease is advanced. Horses with PE have a variety of clinical problems, most notably chronic weight loss, intermittent abdominal discomfort, or diarrhea. Some affected animals are erroneously treated for primary gastric ulceration, and indeed there may appear to be temporary improvement in attitude and appetite. This may reflect successful treatment of gastric ulcers that develop as a secondary problem. Many affected animals are diminished in stature, reflecting retarded growth resulting from a chronic intestinal disorder that probably affects absorption of nutrients. Affected animals typically appear lethargic with a rough haircoat and may have concurrent respiratory infection, dermatitis, and intestinal parasitism.
Ventral edema often is present as a result of hypoproteinemia. Some animals have tachycardia and tachypnea. Fever is an inconsistent finding. Abdominal ultrasonography is often useful to identify thickened small intestine. The entire ventral abdomen should be examined, but the affected intestine is most often in the distal jejunum and ileum, which can be most commonly seen along the midline caudally. If transrectal ultrasonography is possible, better detail of the intestinal wall will be appreciated. In affected animals the intestinal wall diameter will be 6 to 12 mm. More normal surrounding small intestine may be seen, and in contrast the affected segment of bowel will appear rigid with a corrugated appearance to the mucosa, and the diameter of the lumen will be decreased in size because of mucosal proliferation (Fig. 32-48).
CBC findings in horses with PE vary. Leukocytosis is a frequent finding, and this may be characterized by a lymphocytosis. Profound hypoproteinemia (serum protein <3 g/dL, albumin <1.5 g/dL) is a consistent finding. A foal in one report had mildly decreased total serum protein with markedly low albumin (0.6 g/dL) and polyclonal gammopathy (4.6 g/dL). Hyperfibrinogenemia occurs in some cases. Other clinicopathologic findings are variable and depend on the chronicity and whether there are excessive fluid and electrolyte losses through diarrhea.
For definitive diagnosis of PE caused by L. intracellularis, isolation of the organism is considered the gold standard. However, culture of this organism is difficult and requires specialized culture media; therefore the sensitivity of this test is questionable. Specific tests for L. intracellularis infections in foals have been developed. A PCR can be performed on tissues or feces of PE-suspect foals. Use of this test in tissues is sensitive and specific; however, fecal PCR has a higher incidence of false-negative results.515 A more useful antemortem test is the indirect fluorescent antibody (IFA) test that detects antibodies against L. intracellularis in serum.515 Titers ≥1:30 were found to be diagnostic of infection in foals.
Pathologic Findings
Lesions are most frequently found in the distal jejunum and ileum, although diffuse thickening of the small intestine may occur. Classically there is pronounced mucosal thickening with varying severity of ulceration and transmural edema. The affected bowel appears to be stiff, and the mucosal surface has a corrugated appearance. Mucosal pleocytosis is a common feature, but in different cases the predominating inflammatory cell type will differ. A lymphocytic or plasmacytic cellular infiltration may be present. There is crypt proliferation, accompanied by crypt elongation and epithelial hyperplasia. The villi are blunted and may become fused. Silver staining with Warthin-Starry stain reveals elongated, curved bacilli in the apical zone of the crypt epithelial cells.514
Treatment
Treatment objectives are to eliminate the infection, reduce intestinal inflammation, and maintain hydration and plasma colloid oncotic pressure. Several antibiotics have been reported to be useful for the treatment of L. intracellularis infection. Lipophilic drugs with high intracellular penetration are most effective because the bacterium resides within the cells. Ampicillin has good activity against the bacterium in vitro, but because it does not penetrate intracellularly, it is unlikely to be effective in vivo. The most common antibiotic used in PE is erythromycin with or without rifampin for approximately 6 weeks.515 Other drugs in this class, including azithromycin, may be useful for treatment of this disease owing to their high intracellular concentrations.518 Chloramphenicol has also been reported to be effective and is a good alternative in foals that develop a worsening of the diarrhea while on erythromycin. The tetracyclines, particularly oral doxycycline, are a cheap and effective alternative to other antibiotics. A recent report looked at 11 cases of PE in foals treated with tetracycline therapy.519 In this report, 9 of the 11 survived to discharge. The average time to resolution of the diarrhea was 3.5 days after initiation of therapy, and the average treatment time was 3 weeks. The most common administration schedule used was oxytetracycline 6.6 mg/kg IV q12h for 3 to 7 days followed by oral doxycycline 10 mg/kg PO q12h for up to 17 days.517
Supportive therapy with crystalloid fluids to correct dehydration, electrolyte imbalances, and azotemia secondary to fluid losses from profuse diarrhea is warranted. Colloidal support with plasma or hydroxyethyl starch solutions can help correct the edema and decreased colloid oncotic pressure. NSAIDs should be used with caution in foals showing signs of dehydration.
Prognosis in these cases is generally good with early and correct diagnosis of the problem. Therapy can be prolonged and should be continued until the diarrhea and hypoproteinemia have resolved and there is no longer evidence of thickened small intestine on ultrasound.
RHODOCOCCUS EQUI ENTERITIS
R. equi bacteria most frequently cause a severe pyogranulomatous pneumonia in foals 2 to 3 months of age. Extrapulmonary disorders have been associated with R. equi infection in foals, however, and include several intestinal manifestations. Enterocolitis and typhlitis were reported in 10 of 61 foals in one study.520 Diarrhea (13 of 61) and abdominal lymphadenitis (17 of 61) were also reported in the same study.
Pathophysiology
Intestinal infection with R. equi can occur through either fecal-oral transmission or swallowing of infected sputum. The organism invades and reproduces within the macrophages, causing a pyogranulomatous disease. In a survey of normal foals on two different farms, R. equi was shed in the feces of 16 of 17 and 19 of 26 foals, respectively.521 Only two foals developed clinical signs of intestinal disease, and the shedding of bacteria in the feces increased threefold to fourfold during those times. Such foals, as well as adults, are likely the source of repeated contamination on endemic farms. Whether or not the organism causes disease is based on the presence of virulence factors, particularly VapA, which has been most commonly associated with disease in pneumonic foals.
Clinical and Laboratory Findings
Foals diagnosed with R. equi enteritis typically manifest the signs of pneumonia first, although foals in which the enteric form is the major pathology have been reported.522 With the enteric form of the disease, diarrhea, weight loss, and colic are present. Foals are often febrile, anorectic, and depressed. Typical clinicopathologic findings include leukocytosis with neutrophilia and severe hyperfibrinogenemia. Ultrasound of the abdomen may reveal pyogranulomatous abscesses in the lymph nodes.
A diagnosis of R. equi enteritis can be presumed in pneumonic foals showing signs of gastrointestinal disease that culture positive for R. equi in transtracheal wash fluid. R. equi can also be cultured from the feces, small intestinal luminal contents, or occasionally the peritoneal fluid of affected animals. A PCR based on the vapA gene has been developed and can be used as an adjunct diagnostic in these cases.523
Pathologic Findings
The most common small intestinal lesion is multifocal ulcerative enteritis in the area of the Peyer’s patches of the ileum. Other parts of the small intestine may be affected, with lesions present throughout the entire small intestine.523 These lesions frequently extend into the cecum and colon as well. A suppurative exudate is also present, along with pyogranulomatous inflammation of the mesenteric lymph nodes.
Treatment
Treatment of R. equi enteritis is similar to treatment of R. equi pneumonia. Erythromycin estolate (25 mg/kg PO q6-8h) or erythromycin phosphate (37.5 mg/kg PO q12h) combined with rifampin (5 to 7.5 mg/kg PO q12h) has been the traditional treatment for these foals, and it is still effective but requires a long duration of treatment and is difficult for owners to administer because of the frequency of the treatments. Alternative treatments include azithromycin (10 mg/kg PO q24h for 5 days, followed by q48h) and clarithromycin (7.5 mg/kg PO q12h). It is very important to continue treatment until the hematologic abnormalities and radiographic examinations have returned to normal, in order to prevent relapses.
Complications
Complications associated with R. equi enteritis include septic peritonitis and the development of intestinal adhesions. These may lead to death of the animal, or chronic abdominal problems in those that survive. Therapy is often prolonged in these cases, and owners should be warned that foals with extrapulmonary disorders associated with R. equi pneumonia have a more guarded prognosis.
ENTERIC PYTHIOSIS
Pythium species are protistal organisms that belong to a group of phycomycotic organisms that also includes Conidiobolus and Basidiobolus species. They are a frequent cause of severely pruritic cutaneous granulomas in horses along the Gulf Coast and southern United States. Much less frequently, the disease can cause granulomatous lesions in the small intestine.
Pathophysiology
Pythium species are presumed to be transmitted via contact with contaminated water. Once ingested the organisms are thought to penetrate the intestinal mucosa through an existing lesion, because necrotic tissue is considered chemotactic.524Pythium species may be able to penetrate healthy tissue, however, because some cases in dogs have been reported to have mesenteric lymph node involvement without any mucosal lesions.525
Clinical and Laboratory Findings
Four reports of enteric pythiosis can be found in the literature.526-529 In all cases, masses occurred in the mid to distal jejunum. One horse died suddenly, one was euthanized during surgery, and two were successfully treated with a jejunal resection. Organic iodide therapy was instituted in one horse postoperatively for 30 days. In three cases clinical signs of intestinal disease had been present for several months before diagnosis. Chronic colic, weight loss, inappetence, and ill thrift have all been reported and could be related to the intestinal lesion. None of the reported animals had skin lesions. Hematologic evaluation was nondiagnostic in the three cases in which it was performed.
Grossly, lesions are caseous with discrete yellow foci (“kunkers”), and the intestinal wall is thickened because of a pyogranulomatous inflammation. Microscopically, a diffuse, mixed inflammatory infiltrate, along with granulation tissue, is found in the submucosa, tunica muscularis, and mesenteric attachments. Culture of the organism is difficult in these lesions; however, an indirect immunoperoxidase technique to stain for Pythium-positive hyphae is available in some laboratories.528
Treatment
Treatment of phycomycetes is often difficult. They are not true fungi; therefore they are resistant to many antifungal drugs. Systemic amphotericin B is only rarely effective in treating the cutaneous disorder. Organic iodides are inexpensive and safe when administered orally, but the mechanism of action as an antimicrobial drug is not understood, and the efficacy of this compound has not been proven when treating this disease. A vaccine can be formulated against the organism, and this has been shown to shrink the lesions in horses with cutaneous disease. Whether or not it would work in enteric disease is unknown at this time. In addition, premortem or presurgical diagnosis of the disease is extremely difficult, making surgical resection and biopsy the most effective treatment.
INFLAMMATORY BOWEL DISEASE
Several intestinal disorders characterized by inflammatory cell infiltration have been placed under the umbrella of inflammatory bowel disease (IBD), including granulomatous enteritis, multisystemic eosinophilic epitheliotropic enterocolitis, eosinophilic enterocolitis, lymphocytic-plasmacytic enteritis, and basophilic enterocolitis.530-536 IBD in humans is typically characterized by a neutrophilic inflammation, but neutrophils are just the effector cell in a highly complex disease process.537 The syndromes described for equine inflammatory intestinal disorders therefore appear to differ from human IBD; given the different types of cellular infiltrates found in affected horses, these disorders presumably reflect different pathophysiologic mechanisms. Thus use of the term inflammatory bowel disease is not intended to imply either a similarity to the condition described in human beings or similarity among the various syndromes described in horses.
Clinical and Laboratory Findings
Horses with IBD typically have progressive weight loss despite a good appetite and have intermittent abdominal discomfort. If the disease predominates in the small intestine, diarrhea will not be a feature. In some cases there will be associated dermatitis.538 Horses often present with peripheral edema secondary to hypoproteinemia from enteric protein losses. Ultrasonography may reveal thickened small intestine (>5 mm wall diameter).
Clinicopathologic abnormalities may include anemia, hypoalbuminemia, hypoproteinemia, and malabsorption of glucose and D-xylose. Hypoalbuminemia in the absence of proteinuria or severe liver dysfunction is consistent with protein-losing enteropathy. Some horses may have a relative gammopathy. Serum electrolyte concentrations and total CO2 are usually normal. Subclinical disseminated intravascular coagulation with thrombocytopenia and increased fibrinogen degradation products have been identified in horses with chronic enteritis.533,539
In many horses inflammatory cells infiltrate throughout the intestinal tract. Therefore rectal mucosal biopsy may be useful to identify cases of IBD in horses.540 A definitive diagnosis often requires biopsy of the small and/or large intestine. With appropriate instruments this can be done by laparoscopy, although an exploration via a ventral midline approach permits a more thorough evaluation of the abdomen. In most horses cellular infiltration can be found to varying degrees throughout the intestinal tract.
Histopathologic evaluation of biopsy specimens can be used to differentiate among the various IBDs reported in horses, based on the following criteria.534 A diagnosis of granulomatous enteritis is made when aggregates of macrophages and epithelioid cells are found in the mucosa and/or submucosa, along with villous atrophy. In lymphocytic-plasmacytic enterocolitis, lymphocytes and plasmacytes are present in the lamina propria, and villous atrophy usually occurs. For multisystemic eosinophilic epitheliotropic disease, eosinophils, lymphocytes, and macrophages are found in the mucosa and submucosa. Rarely basophils have also been reported.
A new syndrome has recently been discovered involving focal areas of eosinophilic inflammatory infiltrates within the small intestine, termed idiopathic focal eosinophilic enteritis (IFEE).536,541-544 Affected horses typically are presented not because of chronic weight loss or diarrhea, but rather as acute colic cases. Hypoproteinemia and malabsorption are not present. The lesions are intramural masses or circumferential mural bands. Eosinophils with or without lymphocytes are seen infiltrating all layers of the intestine, with varying degrees of fibrosis.534 No underlying cause of the disease has been found, although food allergy, parasitism, and Pythium species have all been suggested.544 The incidence, or at least the diagnosis, of this disease appears to be increasing.
Treatment and Prognosis
Because the specific diseases included under the general category of IBD are quite different, a generalized treatment recommendation cannot be made. Most reported cases of IBD in horses have been fatal, even with aggressive treatment with corticosteroids. Classically reported cases of eosinophilic, lymphocytic, and basophilic enteritis have failed to respond to treatment. If treatment is attempted, immunosuppressive doses of dexamethasone, up to 0.2 mg/kg, once daily are recommended. Successful remission of granulomatous enteritis was reported in one patient that was treated with dexamethasone545; however, in the vast majority of cases treatment is unsuccessful. Cases of IFEE should be differentiated from other causes of IBD with regard to treatment and prognosis. These cases frequently respond to surgical decompression without resection if circumferential mural bands are the only lesion present.544 In cases of intramural masses, surgical resection of the lesions usually resolves the problem.543
NEOPLASIA
Primary and secondary neoplasia involving the alimentary tract of horses is relatively uncommon, although several cases have been reported.546 Typical signs associated with, but not diagnostic for, small intestinal neoplasia include colic and weight loss. In most horses with focal intestinal neoplasia the problem becomes apparent when only lumen obstruction develops. Lymphosarcoma can be disseminated throughout a large portion of intestine, eventually resulting in a malabsorption and weight loss syndrome. Lymphosarcoma affects horses of all ages and can be manifested as an enteric disorder, as well as affecting other systems.547 The diagnosis of enteric lymphosarcoma can occasionally be made on the basis of cytologic examination of fluid obtained by abdominocentesis. In other cases intestinal biopsy is required to diagnose the neoplastic disorder. The prognosis is best if the tumor is discrete and can be removed surgically. A combination chemotherapy protocol of cytarabine (170 mg/m2 IM), cyclophosphamide (142 mg/m2 IV), and prednisolone (86 mg/m2 PO) has been reported to be effective in the treatment of mixed-cell thoracic lymphoma.548 In other cases, corticosteroid therapy may induce clinical remission for several months, although it is not curative.
Other neoplasms affecting the small intestine are unusual, typically arising from the wall of the bowel, and include adenocarcinoma,549 leiomyosarcoma,550 and neurofibroma.551 These tumors often result in intestinal obstruction and signs of abdominal discomfort. Discrete tumors may be surgically removed.
Paraneoplastic syndromes may the first indication of neoplastic processes in the horse. Pemphigus, hypoglycemia, hypercalcemia, erythrocytosis, and neuropathies have all been associated with tumors in horses.552 Paraneoplastic processes may also lead to intestinal tract dysfunction in horses with extraintestinal tumors. Amyloid deposition secondary to multiple myeloma in a horse has been described.553
SMALL INTESTINAL FIBROSIS
Small intestinal fibrosis is a rare syndrome that causes weight loss, chronic colic, and progressive debility in horses and ponies. Clusters of affected animals are reported in Colorado554,555; however newer reports have been published in horses from Missouri.556 The pathophysiology behind the fibrosis is not known, although ingestion of toxins (Convolvulus arvensis or common bindweed), inhibitors of 11β-hydroxysteroid dehydrogenase, or compounds with mineralocorticoid activity has been suggested.
Palpation per rectum typically reveals thickening of the small intestinal wall, which may be confirmed by ultrasound.555 Grossly, the overall length of the intestine may be shortened by as much as 50%.554,555 The histologic lesions include arteriosclerosis, capillary endothelial hypertrophy, extensive fibrosis of the submucosa, and hypertrophy of the muscularis mucosae and tunica muscularis.554 The prognosis for the disease is determined by the length of the intestine that is involved. Surgical resection may be attempted.
LYMPHANGIECTASIA AND CHYLOABDOMEN
There are few reports of lymphangiectasia and chyloabdomen in the literature. Lymphangiectasia is dilation of the lymphatics of the small or large intestine (Fig. 32-49), usually caused by an abscess557 or neoplasia. There are reports of chyloabdomen resulting from congenital lymphatic defects in a neonatal foal, intraabdominal abscesses in a foal, and abdominal adhesions in a miniature horse.557-559 Typically, thickening of the intestinal wall and leakage of chyle into the peritoneal cavity results from obstruction or rupture of the lymphatics. Animals can have signs that include abdominal discomfort, diarrhea, and chronic weight loss. Diagnosis is made on the basis of abdominal fluid analysis and ultrasonography. Chylous abdominal fluid appears milky and may contain a high percentage of lymphocytes. Ultrasonography may reveal segments of thickened small intestine. Diagnosis is confirmed at surgery or postmortem examination, and treatment, if possible, is usually surgical, although in my experience medical resolution of the effusion has been achieved.
SURGICAL DISORDERS OF THE SMALL INTESTINE
Anthony T. Blikslager
SIMPLE OBSTRUCTION
Simple intestinal obstruction is a physical obstruction of the lumen without obstruction of vascular flow. The most common causes are intraluminal masses composed of feed material (e.g., ileal impaction) or accumulation of parasites (e.g., ascarid impactions). There are other instances in which the bowel is obstructed without associated compromise of blood supply, most commonly by extraluminal compression by a mass or band of tissue in horses with intraabdominal adhesions. Because a large volume of fluid enters the small intestinal lumen on a daily basis,560,561 the obstructed intestine becomes distended. The volume of fluid on a daily basis in a hindgut fermenter such as the horse is approximately one extracellular fluid volume, or approximately one third of the horse’s body weight.560 Therefore, although the blood supply is not directly involved in simple obstructions, progressive and marked distention can result in decreased mural blood flow562 and eventual necrosis of tissues.563
Ascarid Impactions
Impactions caused by Parascaris equorum typically occur in weanling foals (median age, 5 months; range, 4 to 24 months) that have been on a poor deworming program and that are administered an anthelmintic when they have a heavy parasite burden.564 Products that cause sudden ascarid paralysis or death, including piperazine, organophosphates, and pyrantel pamoate, have been incriminated.565 However, it is likely that any effective broad-spectrum anthelmintic, such as the avermectins, will have the same effect. Clinical signs include variable onset of colic after administration of an anthelmintic (usually within 1 to 5 days) and signs compatible with small intestinal obstruction, including nasogastric reflux.564 The onset of the disease varies according to the degree of obstruction.565 Diagnosis may be tentatively based on the history in a foal that appears unthrifty, has been recently dewormed, and has signs referable to small intestinal obstruction. The presence of dead ascarids in nasogastric reflux would raise the index of suspicion of this particular form of obstruction.564 Abdominal radiographs or ultrasound will likely indicate the presence of multiple loops of distended small intestine but are not needed if clinical signs indicate the need for immediate surgery. Surgical treatment typically involves an enterotomy made over the intraluminal impaction and removal of ascarids.
Although simple intestinal obstruction tends to carry a favorable prognosis for survival, ascarid impaction is a notable exception. The mortality rate in these cases is high (up to 92% in one study) as a result of severe intestinal compromise, peritonitis, and development of adhesions. The severe intestinal compromise is almost entirely attributable to the duration of impaction, which for unknown reasons is not as readily recognized in foals as it is in adults. Reasons for this could include failure of the owner or farm manager to recognize colic in foals, or failure on the part of the owner or veterinarian to recognize the implications of colic in foals. Although foals readily display signs of colic, other medical causes of colic such as gastric ulcer disease or enteritis might be higher on the list of differential diagnoses than for adults. Second, the size of a foal may make episodes of colic appear more manageable than in adults, in which colic-induced trauma, especially to the head, may force the issue of referral and surgery at an earlier stage. This is speculative, but early intervention in foals with ascarid impaction would likely result in dramatic reductions in the reported mortality rate.564
Ileal Impaction
Ileal impactions occur most commonly in adult horses in the southeastern United States. Although feeding of coastal Bermuda hay has been implicated in the regional distribution of this disease, it has been difficult to separate geographic location from regional hay sources as risk factors.566 However, a recent study from the Southeastern United States showed that feeding coastal Bermuda hay and failing to deworm with an anthelmintic with efficacy against tapeworms are significant risk factors for ileal impaction.567 Furthermore, in a study performed in the United Kingdom, horses with evidence of tapeworm infection were at risk for developing ileal impaction.568 Signs are typical for an adult horse with small intestinal obstruction, including onset of moderate to severe colic and palpable loops of distended small intestine per rectum as the condition progresses. Because the ileum is the distalmost aspect of the small intestinal tract, nasogastric reflux may take a considerable time to develop and is found in only approximately 50% of horses requiring surgical correction of ileal impaction.569,570 The diagnosis is usually made at surgery, although an impacted ileum may be palpated per rectum.571 However, multiple loops of distended small intestine frequently make the impaction difficult to palpate. Nonetheless, clinicians working in the southeastern United States who evaluate horses with cases of mild or moderate colic that have a history of eating coastal Bermuda hay, and in which distended loops of small intestine can be detected adjacent to the cecum, should have ileal impaction high on the list of differential diagnoses. Ileal impactions may resolve with medical treatment. In particular, horses that have clinical signs compatible with obstruction of the small intestine but also have normal abdominal fluid should be treated medically unless pain is unmanageable or subsequent abdominal fluid samples indicate intestinal compromise. In those cases in which surgery becomes necessary, fluids (2 to 3 L) can be directly infused into the mass, allowing the surgeon to breakdown the impaction. However, extensive small intestinal distention and intraoperative manipulation of the ileum frequently leads to POI.572 Therefore some surgeons now elect to perform an enterotomy and to flush the contents from the intestinal lumen with minimal manipulation. Although early studies indicated a guarded prognosis,570 more recent studies indicate that the prognosis is good.569,571
Ileal Hypertrophy
Ileal hypertrophy is a disorder in which the muscular layers (both circular and longitudinal) of the ileum hypertrophy for unknown reasons. Parasitism has been implicated, particularly for those parasites that tend to localize to the ileum (such as tapeworms), but this has never been proven. In some cases the jejunum may also be hypertrophied, either alone or in combination with the ileum.573 It is clear from these findings that initial functional obstruction initiates this syndrome, causing the musculature of the intestine to hypertrophy in order to push intestinal contents aborally, but there is no evidence as to the mechanisms of this disease. Clinical signs include chronic intermittent colic as the ileum hypertrophies and gradually occludes the lumen. In one study partial anorexia and chronic weight loss (1 to 6 months) were documented in 45% of the horses.573 The diagnosis is usually made at surgery, although in some cases the hypertrophied ileum may be palpated per rectum or seen on ultrasonographic evaluation of the abdomen. For treatment, an ileocecal or jejunocecal anastomosis to bypass the hypertrophied ileum is performed. Without surgical bypass, intermittent colic persists, and the thickened ileum may ultimately rupture. According to one recent report of 11 horses with hypertrophy of the ileum, only one horse survived, indicating a poor prognosis. The most common reason for euthanasia was spontaneous ileal rupture.573
Meckel’s Diverticulum
Meckel’s diverticulum is an embryonic remnant that may become impacted. The diverticulum arises from the vitelloumbilical duct, which fails to completely atrophy, and becomes a blind pouch projecting from the antimesenteric border of the ileum.574,575 Occasionally an associated mesodiverticular band may extend from the diverticulum to the umbilical remnant and serve as a point around which small intestine may become strangulated. Mesodiverticular bands may also originate from the embryonic ventral mesentery and attach to the antimesenteric surface of the bowel, thereby forming a potential space within which intestine may become entrapped (Fig. 32-50).576 Clinical signs range from chronic colic for an impacted Meckel’s diverticulum to acute severe colic if a mesodiverticular band strangulates intestine. The diagnosis is made at surgery, and treatment requires resection of the diverticulum and any associated bands.
STRANGULATING OBSTRUCTION
Strangulation obstruction of the intestine is characterized by simultaneous occlusion of the intestinal lumen and its blood supply. Although strangulation of the intestinal lumen results in clinical signs similar to those of simple obstruction, occlusion of the blood supply results in a more rapid deterioration of the intestinal mucosa and subsequent onset of sepsis. Therefore clinical signs (including pain, heart rate, mucous membrane color, and capillary refill time) are typically more severe, and the prognosis is less favorable.
A great deal of work has been done to characterize mucosal injury that occurs during strangulation577,578 and, more recently, during reperfusion.579 In general, the lesion that develops during strangulation is severe, leaving little viable bowel for further injury during reperfusion.580 The severity of the ischemic lesion is partly attributable to the fact that in most cases initial occlusion of veins and partial occlusion of arterial blood supply during strangulation induces a hemorrhagic lesion. This results in extensive congestion and mucosal degeneration (Fig. 32-51).577 Bowel peripheral to the strangulating lesion may also become injured as a result of distention.581 In addition, distended small intestine that remains viable after surgical correction of strangulation may be subject to reperfusion injury after surgical correction of the lesion.582 Unfortunately, attempts at reducing mucosal injury in the horse with antioxidants, which would be expected to inhibit injury attributable to reperfusion injury and associated reactive oxygen metabolites, have been unsuccessful.583
The prognosis for survival in horses with small intestinal strangulating lesions is generally lower than with most types of colic. In a large multicenter study, SISO had a fatality rate of 67% compared with that of small intestinal simple obstruction, which had a fatality rate of 48%.584 Although recent studies indicate higher survival rates in general, the relative fatality of strangulating obstruction compared with simple obstruction is probably similar. One study indicated that in excess of 70% of horses were discharged from the hospital after surgical correction of SISO.585 These figures should be interpreted cautiously because they are based on the number of horses that are recovered from surgery rather than the number of horses initially presented for evaluation of severe colic. In addition, owners should be warned that the long-term survival rate is reduced by long-term complications.586 The principal reason for reduced long-term survival in patients that have had small intestinal surgery is intraabdominal adhesions (Fig. 32-52). For example, in one study 22% of horses that had small intestinal surgery required further surgery or were euthanized because of adhesions, and only 25% of horses that had a clinical problem with adhesions survived.587
Epiploic Foramen Entrapment
The epiploic foramen is a potential opening (because the walls of the foramen are usually in contact) to the omental bursa located within the right cranial quadrant of the abdomen. It is bounded dorsally by the caudate process of the liver and caudal vena cava and ventrally by the pancreas, the hepatoduodenal ligament, and the portal vein.588,589 Clinical signs include acute onset of severe colic with examination findings compatible with small intestinal obstruction. Although the condition was reportedly more prevalent in older horses, recent studies have not detected an age predilection.590 However, studies have detected a seasonal pattern for this disease. Specifically, more horses appear to suffer from epiploic foramen entrapment during the fall and winter months.591 An additional factor associated with epiploic foramen entrapment is crib-biting. One potential reason for this association is the development of negative intrathoracic pressure during cribbing, which might then result in cranial movement of intestine into the vicinity of the epiploic foramen. The association between crib-biting and epiploic foramen entrapment has now been shown in separate studies performed in the United Kingdom and the United States.592
The diagnosis is definitively made at surgery, although ultrasonographic findings of distended loops of edematous small intestine adjacent to the right middle body wall are suggestive of epiploic foramen entrapment.588 Entrapped small intestine may enter the foramen from the visceral surface of the liver toward the right body wall or in the opposite direction. Reports differ as to which is the more common form. Entrapped small intestine may be limited to a portion of the intestinal wall (parietal hernia).593 In addition, the large colon may become entrapped within the epiploic foramen.594 In treating epiploic foramen entrapment, the epiploic foramen must not be enlarged either by blunt force or with a sharp instrument, because rupture of the vena cava or portal vein and fatal hemorrhage may occur. In addition, excessive force used to extract entrapped bowel during surgery may also result in rupture of a mesenteric branch of the cranial mesenteric artery. The finding of intraabdominal hemorrhage in some cases of epiploic foramen entrapment before surgery may result from compromise of the mesenteric blood supply rather than the major blood vessels that border the epiploic foramen. Prognosis has substantially improved over the last decade, with current short-term survival rates (discharge from the hospital) ranging from 74% to 79%.588,595
Strangulation by Mesenteric Pedunculated Lipoma
Lipomas form between the leaves of the mesentery as horses age, and mesenteric stalks develop as the weight of the lipoma tugs on the mesentery. The stalk of the lipoma and a loop of small intestine or small colon may become intertwined, causing strangulation (Fig. 32-53). Strangulating lipomas should be suspected in mature horses (>10 years old) with acute colic referable to the small intestinal tract.596 In addition, geldings and ponies appear to be at risk for developing strangulating lipomas,597 possibly because of differences in fat metabolism. One recent study documented a case of strangulating lipoma in a much younger patient, indicating that although the aging process is associated with development of lipomas, this disease cannot be ruled out preoperatively purely on the basis of age.598,599 The diagnosis is usually made at surgery, although on rare occasions a lipoma can be palpated per rectum.600
Treatment involves surgical resection of the lipoma and strangulated bowel, although strangulated intestine is on occasion viable. Studies indicate that approximately 50% to 78% of horses are discharged from the hospital after surgical treatment.585
Small Intestinal Volvulus
A volvulus is a twist along the axis of the mesentery, whereas torsion is a twist along the longitudinal axis of the intestine. Small intestinal volvulus is theoretically initiated by a change in local peristalsis, but the mechanisms of this disease are unclear.601 It is reportedly one of the most commonly diagnosed causes of small intestinal obstruction in foals.602,603 It has been theorized that young foals may be at risk for small intestinal volvulus because of changing feed habits as they adapt to a forage-based adult diet. Onset of acute, severe colic, a distended abdomen, and radiographic or ultrasonographic evidence of multiple loops of distended small intestine in a young foal would be suggestive of small intestinal volvulus. However, volvulus is not possible to differentiate from other causes of small intestinal obstruction preoperatively.
In adult horses, volvulus frequently occurs in association with another disease process, during which small intestinal obstruction results in distention and subsequent rotation of the small intestinal around the root of the mesentery (e.g., in horses that have strangulation caused initially by a pedunculated lipoma). Although any segment of the small intestine may be involved, the distal jejunum and ileum are most frequently affected, most likely because the mesentery is longer in the more distal bowel. The diagnosis is made by palpating a twist at the origin of the cranial mesenteric artery during surgical exploration. Treatment includes resection of devitalized bowel, which may not be an option because of the extent of small intestinal involvement. For example, if complete volvulus of the small intestinal tract occurs, the only option would be to untwist the intestine and hope that the bowel was sufficiently viable to survive the disease process. This would in turn depend on the duration of disease and the extent of intestinal ischemia (i.e., how tightly the intestine twisted as the volvulus developed). Prognosis is based on the extent of small intestine involved and its appearance after surgical correction of the lesion.604 For example, the degree of sepsis may relate to the surface area of devitalized tissue. In addition, the length of the time it takes to complete the surgery can become problematic, particularly in patients with sepsis, in which it is difficult to maintain adequate arterial blood pressure. Nonetheless, survival in patients with extensive small intestinal resection may not relate to the absolute length of intestine resected.605 A recent study indicated that the prognosis for survival is good (80% of horses recovered from surgery and were discharged).606
Inguinal Hernia
Inguinal hernias are more common in standardbred and Tennessee Walking horses, which tend to have congenitally large inguinal canals. Inguinal hernias may also occur in neonatal foals, but they differ from hernias in mature horses in that they are typically nonstrangulating. Typical historical findings in mature horses with inguinal hernias include acute onset of colic in a stallion that has recently been used for breeding. A cardinal sign of inguinal herniation is a cool, enlarged testicle on one side of the scrotum.607,608 This is attributable to occlusion of the testicular blood supply in most cases rather than strangulated intestine within the scrotum itself. Inguinal hernias can also be detected on rectal palpation, and ultrasound of the testicle is very helpful in determining the diagnosis, particularly considering the differential diagnosis of testicular torsion. The nature of the hernia (direct versus indirect) is determined by the integrity of the parietal vaginal tunic. In horses in which the bowel remains within the parietal vaginal tunic, the hernia is said to be indirect, because strictly speaking the bowel remains within the peritoneal cavity. Direct hernias are those in which bowel ruptures through the parietal vaginal tunic and occupies a subcutaneous location, followed by strangulation. Direct hernias most commonly occur in foals and should be suspected when a congenital inguinal hernia is associated with colic, swelling that extends from the inguinal region of the prepuce, and intestine that may be palpated subcutaneously.609,610 Although most congenital indirect inguinal hernias resolve with repeated manual reduction and/or application of a diaper, surgical intervention is recommended in foals with congenital direct hernias in order to reduce the hernia as well as repair the parietal vaginal tunic.
In stallions with indirect inguinal hernias, manipulation of herniated bowel per rectum can be used to reduce a hernia but is not generally recommended because of the risk of a rectal tear. In addition, the inability to visually assess the integrity of the bowel may well be problematic. However, in many cases taken to surgery, the short segment of herniated intestine will markedly improve in appearance once it has been surgically reduced, and in some cases the affected intestine can be left unresected. The affected testicle will be congested because of vascular compromise within the spermatic cord, and although it may remain viable, it is recommended that it be resected. Furthermore, the inguinal canal should be partially closed at surgery to prevent recurrence, and the risk that this procedure will reduce spermatic cord vasculature is a further problem. The prognosis in adult horses is good, with up to 75% of horses surviving to 6 months of age.608 Horses that have been treated for inguinal hernias may be used for breeding. In these horses, the remaining testicle will have increased sperm production, although an increased number of sperm abnormalities will be noticed after surgery because of edema and increased temperature of the scrotum.
Strangulating Umbilical Hernias
Although umbilical hernias are common in foals, strangulation of herniated bowel is rare. In one study, 6 of 147 horses with umbilical hernias (4%) had incarcerated intestine.611 Clinical signs include a warm, swollen, firm, and painful hernia sac associated with signs of colic. The affected segment of bowel is usually small intestine, but herniation of cecum or large colon has also been reported.612 In rare cases a hernia that involves only part of the intestinal wall may be found and is referred to as Richter’s hernia. In foals that have Richter’s hernia, an enterocutaneous fistula may develop. In one study, 13 of 13 foals with strangulating umbilical hernias survived to discharge, although at least three horses died because of long-term complications.612
Diaphragmatic Hernias
Herniation of intestine through a rent in the diaphragm is uncommon in the horse, accounting for 0.3% of all cases of colic in a large multicenter study.584 Any segment of bowel may be involved, although small intestine is most frequently herniated.613 Diaphragmatic rents may be congenital or acquired, but acquired hernias are more common. Congenital rents may result from incomplete fusion of any of the four embryonic components of the diaphragm: pleuroperitoneal membranes, transverse septum, and esophageal mesentery. In addition, abdominal compression of the foal at parturition may result in a congenital hernia. Acquired hernias are presumed to result from trauma to the chest or a sudden increase in intraabdominal pressure such as might occur during parturition, distention of the abdomen, a sudden fall, or strenuous exercise.614 In one study, 19 of 40 horses diagnosed with diaphragmatic hernia (48%) had a history of recent trauma.615 Hernias have been located in a number of different locations, although large congenital hernias are typically present at the most ventral aspect of the diaphragm and most acquired hernias are located at the junction of the muscular and tendinous portions of the diaphragm. In addition, a peritoneal-pericardial hernia has been documented in at least one horse.616
The clinical signs are usually associated with intestinal obstruction rather than respiratory embarrassment. However, careful auscultation may reveal an area of decreased lung sounds associated with obstructed intestine and increased fluid within the chest cavity. Such signs may prompt thoracic radiography or ultrasound, both of which can be used to make a diagnosis. In one review, 7 of 40 horses reported in the literature with diaphragmatic hernia (18%) had dyspnea.615 Auscultation may also reveal thoracic intestinal sounds, but it is typically not possible to differentiate these from sounds referred from the abdomen. In one report, two of three horses diagnosed with small intestinal strangulation by diaphragmatic hernia had respiratory acidemia, attributable to decreased ventilation.617
Intussusceptions
An intussusception involves a segment of bowel (intussusceptum) that invaginates into an adjacent aboral segment of bowel (intussuscipiens). The reason for such invagination is not clear, but it may involve a lesion at the leading edge of the intussusception, including small masses, foreign bodies, or parasites. In particular, tapeworms (Anoplocephala perfoliata) have been implicated.618 Ileocecal intussusceptions are the most common intestinal intussusceptions in the horse and typically affect young animals. For example, in one study evaluating 26 cases of ileocecal intussusception, the median age of the horses was 1 year old.619 Acute ileocecal intussusceptions are those in which the horse has a duration of colic of less than 24 hours and involve variable lengths of intestine that ranged in one study from 6 cm to 457 cm long. In acute cases the involved segment of ileum typically has a compromised blood supply. Chronic ileocecal intussusceptions typically involve short segments of ileum (up to 10 cm long), and the ileal blood supply is frequently intact. Abdominocentesis results are variable because strangulated bowel is contained within the adjacent bowel. There is often evidence of obstruction of the small intestine, including nasogastric reflux and multiple distended loops of small intestine on rectal palpation. Horses with chronic ileocecal intussusceptions have mild, intermittent colic, often without evidence of small intestinal obstruction. A mass may be palpated in the region of the cecal base in approximately 50% of cases. Transabdominal ultrasound may be helpful in discerning the nature of the mass. The intussusception has a characteristic target appearance on cross-section.620
Other segments of the small intestine may also be intussuscepted, including the jejunum. In one study on 11 jejunojejunal intussusceptions, the length of bowel involved ranged from 0.4 to 9.1 m.621 Attempts at reducing intussusceptions at surgery are usually futile because of intramural swelling of affected bowel. Jejunojejunal intussusceptions should be resected. For acute ileocecal intussusceptions the small intestine should be transected as far distally as possible, and a jejunocecal anastomosis performed. In cases with particularly long intussusceptions (length up to 10 m has been reported), an intracecal resection may be attempted. For horses with chronic ileocecal intussusceptions, a jejunocecal by-pass without small intestinal transection should be performed.
In one study that evaluated the survival of horses with ileocecal intussusceptions, seven of seven horses with chronic intussusceptions survived long term (>4 months), whereas only 5 of 12 horses with acute intussusceptions (42%) survived long term.619 In a separate study that evaluated survival in horses with jejunojejunal intussusceptions, 6 of 11 horses (54%) were discharged from the hospital after surgery, and 4 of 11 horses (36%) survived long term (>16 months).621
Other Small Intestinal Strangulations
Entrapment of the small intestine may occur through rents in the mesentery, internal ligaments such as the gastrosplenic ligament,622 the broad ligament, and the proximal aspect of the cecocolic ligament.623 Entrapments may also occur through trauma-induced body wall hernias. For all of these conditions it is often necessary to enlarge the rent or hernia to allow reduction of entrapped small intestine. In the case of body wall hernias, the defect should be closed with suture, or patched using mesh. Entrapment of small intestine within mesenteric rents appears to be particularly problematic because they are difficult to reduce and have an unfavorable prognosis. The latter may be a result of the fact that large lengths of intestine may become strangulated, and because the mesenteric vasculature is frequently compromised to the extent that intraabdominal hemorrhage has been noted in some cases.624
NONSTRANGULATING INFARCTION
Nonstrangulating infarction occurs secondary to cranial mesenteric arteritis caused by migration of S. vulgaris625 and has become a rare surgical disorder since the advent of broad-spectrum anthelmintics. Although thromboemboli have been implicated in the pathogenesis of this disease, careful dissection of naturally occurring lesions has not revealed the presence of thrombi at the site of intestinal infarctions in most cases. These findings suggest that vasospasm plays an important role in this disease.625 Clinical signs are highly variable, depending on the extent to which arterial flow is reduced and the segment of intestine affected. Any segment of intestine supplied by the cranial mesenteric artery or one of its major branches may be affected, but the distal small intestine and large colon are more commonly involved. There are no clinical variables that can be used to reliably predict or differentiate this disease from strangulating obstruction. In some cases massive infarction results in acute, severe colic. Other cases may have intermittent colic as smaller emboli are released into the colonic blood supply. Occasionally an abnormal mass and fremitus may be detected on palpation of the root of the cranial mesenteric artery per rectum. This disease should be considered a differential diagnosis in horses with a history of inadequate anthelmintic treatment and the presence of intermittent colic that is difficult to localize. Although fecal parasite egg counts should be performed, they are neither indicative of the degree nor specific for the type of parasitic infestation.
In addition to routine treatment of colic, dehydration, and endotoxemia, medical treatment may include aspirin (20 mg/kg q24h) to decrease thrombosis. Definitive diagnosis requires surgical exploration. Surgical treatment depends on the distribution of infarction. Unfortunately, these cases are difficult to treat because of the patchy distribution of the lesions and the possibility of lesions extending beyond the limits of surgical resection. In addition, further infarction may occur after surgery. The prognosis is fair for horses with intermittent mild episodes of colic that may be amenable to medical therapy, but horses that require surgical intervention have a poor prognosis.625
GASTROINTESTINAL ILEUS
Guy D. Lester
A number of factors are responsible for movement of feed through the gastrointestinal tract, and disturbance to any of these could result in ineffective transit and signs of abdominal pain. Gastrointestinal motility involves an extremely complex interaction among the enteric nervous system, the wall of the intestine, and the luminal contents. Other factors that influence the transit of digesta include gravity, the volume and viscosity of the contents, and pressure gradients created by simultaneous contraction and relaxation of adjacent segments of bowel. Several equine diseases likely involve altered gastrointestinal motility including POI, cecal emptying defect, gastroduodenal ulcer disease, intraluminal obstruction, bowel distention, strangulating obstructions, peritonitis, and primary IBDs, such as DPJ or colitis. Ineffective intestinal motility is also a feature of several neonatal diseases including prematurity, systemic sepsis, and perinatal asphyxia. Certain parasitic infections, electrolyte derangements, and endotoxemia can modify digesta transit in horses of all ages. Intestinal motility is also disturbed during general anesthesia and when specific sedatives, such as xylazine, romifidine, or detomidine, are administered.
POSTOPERATIVE ILEUS
The inhibition of propulsive bowel activity is usually referred to as ileus. Ileus is most frequently ascribed to the condition that occurs after laparotomy and is termed simple or uncomplicated postoperative ileus. When intestinal motility is disturbed for longer periods after surgery, typically for more than 72 hours, the term complicated or paralytic ileus is used.626 POI in horses is most commonly associated with surgery of the small intestine, particularly after resection and anastomosis. The true incidence of POI in horses undergoing laparotomy for gastrointestinal disease is difficult to determine because of variation in definition, surgical skill, and anesthetic technique. POI was reported as a complication in only 10% of horses that underwent laparotomy for a small intestinal lesion at a referral practice.627 However in a multicenter study 47 of 251 horses undergoing laparotomy for acute colic including small and large intestinal disease developed POI.628
POI can have a significant negative impact on short-term postoperative survival.629,630 Motility dysfunction is likely present in all horses after laparotomy, but most cases are subclinical and require minimal intervention. In symptomatic animals clinical signs are typically observed within 24 hours of recovery and include colic, tachycardia, dehydration, decreased borborygmi and fecal output, and sequestration of fluid within the stomach. Rectal examination and ultrasound may reveal small intestinal distention with rare or absent wall movement. The severity and duration of intestinal stasis is variable, lasting from minutes to days.
CECAL EMPTYING DEFECT
An important and specific motility problem involving the cecum occurs sporadically in horses.631-634 There are limited epidemiologic data about the condition, and key aspects of the pathophysiology are not known. Cecal emptying defect occurs most commonly after general anesthesia and extraabdominal surgery, particularly orthopedic and upper airway procedures, and is therefore often categorized as a form of POI. Young male performance animals appear to be at greatest risk, especially if the procedure is performed with the animal in full work. Other cases occur spontaneously, often in animals with painful primary conditions such as uveitis, orchitis, or septic tenosynovitis. Clinical signs are often subtle unless perforation has occurred. In horses with a cecal emptying defect after anesthesia, clinical signs are usually apparent by 3 to 5 days after the procedure. The earliest detectable signs include depression and a reduction in appetite and fecal output. Ineffective emptying results in overfilling of the cecum with moist contents, which is manifest by signs of mild to moderate colic. If the condition is recognized late or untreated, the cecum may rupture, resulting in fatal peritonitis.
Physiology
The control of intestinal motility is complex and involves a combination of central innervation, autonomic innervation, and the enteric nervous system (Fig. 32-54). Intestinal contractions are primarily controlled by the enteric nervous system and do not require extrinsic neural input. The inherent rhythmicity of electrical activity in the intestine is controlled by the interstitial cells of Cajal (ICCs). These are highly specialized cells that are electrically coupled to the smooth muscle syncytium via gap junctions.635 ICCs are responsible not only for generation of slow waves (cyclical electrical activity), but also for coordination of pacemaker activity and propagation of slow waves along the intestine. ICCs appear to be critically involved in a range of motility disorders in humans, including gastroparesis, pseudoobstruction, and chronic constipation.636 In horses a reduction in ICC density was demonstrated with equine dysautonomia (grass sickness) and in horses with large intestinal disease.637,638
The inherent “excitability” of smooth muscle cells in response to stimuli varies among regions of the gastrointestinal tract and among species. This variability in cell excitability is dependent on the magnitude of the membrane potential, which in turn is regulated by the number and subtype of potassium channels.635
The enteric nervous system plays an important role in control and coordination of intestinal contraction. Contractile events are influenced by central and autonomic innervation, but external neural input is not required for contraction. The parasympathetic supply to the gastrointestinal tract is via the vagus and pelvic nerves, and the sympathetic supply is through postganglionic fibers of the cranial and caudal mesenteric plexuses. A complex network of interneurones within each plexus integrates and amplifies neural input, and the intensity and frequency of resultant smooth muscle contractions are proportional to sympathetic and parasympathetic input. Additional binding sites for a number of other endogenous chemicals, including dopamine, motilin, and serotonin, can be found within the enteric nervous system and on smooth muscle cells.639 It is important to appreciate that mechanisms to slow progressive intestinal motility are also critical in order to retain feed for adequate digestion and absorption of nutrients. The terms jejunal and ileal brake have been used to describe the slowing of transit caused by mediators such as peptide YY, noradrenergic nerves, and opioid, serotonergic, and chemosensitive afferent neurons.640
Acetylcholine (ACh) is the dominant excitatory neurotransmitter in the gastrointestinal tract and exerts its action through muscarinic type 2 (M2) and M3 receptors. Sympathetic fibers innervating the gastrointestinal trace are adrenergic, postganglionic fibers with cell bodies located in the prevertebral ganglia. Activation of α2-adrenergic receptors on cholinergic neurons within enteric ganglia inhibits the release of ACh and therefore reduces intestinal contraction. The β1-, β2- and β-atypical receptors are directly inhibitory to the intestinal smooth muscle.641 Inhibitory nonadrenergic, noncholinergic (NANC) neurotransmiters include ATP, vasoactive intestinal peptide (VIP), and nitric oxide (NO).642,643 These neurotransmitters are critical for mediating descending inhibition during peristalsis and receptive relaxation. Substance P is a NANC neurotransmitter that may be involved in contraction of the small intestine and large colon.644-646 The rate and force of intestinal contractions along the small intestine and large colon of the horse are key determinants of intestinal motility. Of even greater importance to the net propulsion of digesta are the cyclical patterns of contractile activity. These patterns are known as the small intestinal and colonic migrating motility complexes (MMC).647 The colonic complex usually originates in the right ventral colon and variably traverses the ascending and descending colons. Many of these complexes are temporally related to a specialized motility event of the ileum known as the migrating action potential complex.
MEDIATORS OF GASTROINTESTINAL ILEUS
Inflammation within the intestinal muscularis and inhibitory neural events are important initiators of intestinal ileus.648,649 Intestinal inflammation is not only important in primary intestinal diseases in horses, such as DPJ and colitis, but is induced after simple intestinal handling during laparotomy. Experimental data from other species suggests that handling of the small or large intestine at the time of surgery induces an intense inflammatory response within the muscularis and a reduction in the intensity of smooth muscle contraction. Furthermore, the inflamed intestine fails to contract normally in response to putative prokinetic agents. Additional studies have identified that this inflammatory response was not restricted to segments that were manipulated at the time of surgery but also involved other regions throughout the gastrointestinal tract.649 Intestinal handling in experimental animals causes local overproduction of NO because of the upregulation of iNOS by resident macrophages.650 NO is a key inhibitory neurotransmitter of the NANC system.643 Consequently NOS inhibition could be an important target for prevention of ileus.
The inhibition of motility associated with peritoneal inflammation may also be mediated through neural reflexes. The afferent segment is partly composed of capsaicin-sensitive visceral afferent C-fibers that terminate in the dorsal horn of the spinal cord, where they can activate inhibitory sympathetic fibers or, alternatively, synapse directly on the sympathetic ganglia. Consequently the efferent limb of the reflex expresses increased sympathetic outflow, primarily mediated through stimulation of α2-adrenoreceptors, with subsequent inhibition of ACh release. This provides the rationale for alpha-2 blockade in the treatment of ileus. Intraluminal infusion of capsaicin before abdominal surgery ameliorated the severity of POI in experimental rats.651 This finding highlights the importance of visceral afferent fibers in the development of POI.
Ileus can also occur with intestinal obstruction or displacement. Mild to moderate distention of the bowel, such as that occurring in the early stages of an intraluminal obstruction, evokes an increase in local contractile activity.652,653 Excessive distention results in inhibition of motility within the distended segment of bowel. Intestinal stasis is not always detrimental and under certain conditions may be protective.
Endotoxemia is a feature of many diseases of the equine gastrointestinal tract, and endotoxins can independently exert a negative effect on intestinal motility and transit.654 Various mediators are likely involved, but activation of α2-adrenoreceptors and production of prostanoids appear to be important, as the inhibitory effects of experimental endotoxin infusion are ameliorated by pretreatment with yohimbine or NSAIDs (phenylbutazone or flunixin), respectively.655,656 Endotoxin infusion induced an inflammatory response in the intestine of rats that mimicked that induced by handling during laparotomy.657
The pathophysiology of cecal emptying defect is not known. This syndrome may best mimic POI in humans, which is generally considered a large intestinal disorder. An important difference in horses is that laparotomy is a rare predisposing factor, and most cases occur in horses undergoing routine extraabdominal surgical procedures. General anesthesia itself is a potent inhibitor of gastrointestinal motility in horses, but the effects are short-lived and reversible within hours of anesthetic withdrawal.658 The return of normal motility in horses after experimental ileus was most delayed in the cecum, suggesting that this may be a common site of ileus in horses.659 A link between routine postoperative medications, such as phenylbutazone and aminoglycoside antibiotics, has been suspected but not established. An inhibitory effect of NSAIDs on large colon contractility has been demonstrated using in vitro techniques, although the effect of selective COX-2 inhibitors was variable and not as marked as that of mixed isoform inhibitors.660,661 Primary sympathetic overstimulation could be involved, as many of the affected animals are young, male horses or animals with painful diseases.
The development of small intestinal POI, but not cecal emptying dysfunction, is influenced by the duration of surgery.628,632 Technique may have a weak influence on small intestinal POI after jejunojejunostomy. The duration of intestinal ileus was shorter in animals that received a side-to-side stapled anastomosis than in those that had a hand-sewn end-to-end procedure.629 The duration of ileus after stapled end-to-end anastomosis was not different from that after either procedure.
Reported risk factors for the development of POI in horses include age (>10 years), small intestinal resection and anastomosis, breed, and duration of anesthesia and surgery.628,662 A pelvic flexure enterotomy and emptying the colon and intraoperative lidocaine infusion had a protective effect against POI.
The inhibitory effects of α2-adrenergic agonists, such as xylazine and detomidine, on intestinal motility in horses are well described.663-671 The activation of postganglionic cholinergic nerves in the myenteric plexus by this class of drugs results in reduced ACh release from autonomic and enteric nerves. Intravenous xylazine inhibits cecal and large colon motility for 20 to 30 minutes without seriously disrupting small intestinal myoelectric activity, and detomidine can reduce large intestinal myoelectric activity for up to 3 hours. The α2-antagonist yohimbine has a weak but positive effect on cecal emptying in normal ponies, suggesting that normal motility is under constant α2-adrenergic tone.665 Atropine is a postganglionic blocking agent that binds to muscarinic receptors. When administered at 0.04 mg/kg, individual small intestinal, cecal, and colonic contractions are inhibited for about 120 minutes, but small intestinal and colonic migrating complexes are suppressed for up to 8 hours.672
Diagnosis
The diagnosis of ileus is based on history and physical examination findings. Important tests include determination of pulse rate and rhythm, auscultation and percussion of the abdomen, rectal palpation, and passage of a nasogastric tube. A CBC with fibrinogen estimation and cytologic analysis of peritoneal fluid may improve the accuracy of diagnosis. Affected animals may be colicky because of accumulation of fluid in the upper gastrointestinal tract (classical POI) or cecal contents (cecal emptying defect). Decompression of the stomach is important diagnostically and therapeutically in horses with POI after small intestinal surgery. Failure to relieve pain with gastric decompression could point toward mechanical obstruction, severe inflammation of the intestine, or peritonitis. Most animals with ileus are depressed and have reduced fecal output and intestinal borborygmi. Intestinal sounds should, however, be interpreted with caution, because the presence of borborygmi does not always equate to progressive intestinal motility and may merely reflect local, nonpropagated contractions. Rectal palpation findings in cases of persistent POI or DPJ are usually nonspecific but may reveal dilated, fluid-filled loops of small intestine. Roughened peritoneal surfaces can occasionally be palpated if there is peritonitis. Cecal distention with digesta can be palpated in horses with advanced cecal dysfunction.
It is important to distinguish functional ileus from mechanical obstruction. This can be extremely difficult, but horses with mechanical obstruction typically have sustained high volumes of gastric reflux that vary little over time.
Treatment
The management of gastrointestinal ileus is dependent on the segment of gastrointestinal tract involved. Therapy for ileus of the proximal gastrointestinal tract typically involves a combination of gastric decompression, fluid and electrolyte therapy, and antiinflammatory drugs. Nasogastric decompression is the mainstay of management to prevent overdistention of the stomach and small intestine. This often necessitates placement of an indwelling nasogastric tube, a procedure that some have speculated may in itself delay gastric emptying. The placement of an indwelling tube for 18 hours did not adversely affect the emptying of liquids, but in a recent study a delay was noted when the tube was left in place for 72 hours.673,674 Both studies used apparently healthy animals.
Electrolyte balance is important, particularly with respect to maintaining adequate extracellular concentrations of potassium, calcium, and magnesium. Calculation of the volume of fluid to be administered should include maintenance requirements (40 to 60 mL/kg/day) plus an estimate of losses, especially those lost through gastric decompression. Overhydration should be avoided in horses with small intestinal ileus. Fluid therapy is the key component in the management of cecal emptying defect, usually in combination with lubricants or laxatives, such as mineral oil or magnesium sulfate, and with careful use of antiinflammatory drugs. Horses with primary cecal impaction or impaction secondary to an emptying defect may require surgery in order to prevent rupture. The surgical management of these cases is controversial and may include typhlotomy alone, typhlotomy with a bypass procedure such as ileocolic or jejunocolic anastomosis, or a bypass without typhlotomy.675 Most horses that undergo simple typhlotomy have an uneventful recovery, although a small number will reimpact and require a second laparotomy.633
A significant clinical or economic benefit of parenteral nutrition in adult horses with gastrointestinal disease has also yet to be demonstrated.676-678 Parenteral nutrition should be considered when feed has been withheld for more than 96 hours, particularly in the horse with a surgical wound. Limited exercise, in the form of hand-walking, may provide some benefit to these animals, although there is no evidence that exercise has a direct impact on intestinal motility in either horses or humans with POI.679
Drugs that may retard normal intestinal motility should be avoided in horses with gastrointestinal ileus. These include the anticholinergics, such as atropine, and opiate receptor agonists, such as morphine and meperidine.680,681 Butorphanol appears to have little or no adverse effect on either small or large intestinal motility.682,683 α2-Adrenergic agonists should be used sparingly because of their inhibitory effects on large intestinal motility.
The pivotal role of intestinal inflammation provides a strong rationale for the use of antiinflammatory drugs in affected animals. Flunixin meglumine is widely used in equine practice as an analgesic and antiinflammatory agent, and it also ameliorates many of the adverse systemic effects of endotoxin. A potential negative effect of NSAIDs on large intestinal contractility has been suggested on the basis of in vitro studies660; however, a similar response has not been reported in whole animal studies.663,665,684 There may be a role for novel agents in the future. Carbon monoxide (CO) has prevented POI in experimental animals, a benefit not only when CO was inhaled but also when CO was dissolved in a peritoneal lavage solution, making potential administration considerably easier and safer.685 CO has potent intestinal antiinflammatory properties and is derived from the degradation of heme by the enzyme heme oxygenase. Expression of a heme oxygenase isoform (HO-1) can be induced by a variety of stimuli.
Broad-spectrum antimicrobial drugs are indicated when sepsis is suspected or if the immune system is compromised, as in cases with moderate to severe endotoxemia. Theoretic concerns have been raised regarding the use of aminoglycoside antibiotics in animals with ileus. Inhibition of intestinal contractions occurred when sections of intestine were exposed to high concentrations of aminoglycoside antimicrobial drugs, but this inhibitory effect is unlikely to occur at clinically relevant doses.686 The administration of benzyl penicillin and/or ceftiofur was associated with an increased risk of colic in the week after anesthesia for diagnostic imaging or nonabdominal surgical procedures.687
Motility-modifying drugs could play an important role in the prevention and treatment of gastrointestinal ileus. An effective prokinetic agent could shorten the length of hospitalization, thereby reducing the cost of treatment and the number of potential complications such as weight loss, thrombophlebitis, and laminitis. There is also evidence that the development of abdominal adhesions could be minimized with the use of an effective prokinetic drug.688,689 A potential negative impact of prokinetic use in the postoperative period after anastomosis is increased predisposition to dehiscence.690
Most motility-modifying drugs require a healthy intestinal wall in order to enhance intestinal contraction. There are several examples in horses in which the expected contractile response to certain drugs is blunted in the face of disease. It is reasonable to assume that many putative prokinetic drugs would be partially or totally ineffective in horses with abdominal disease. This would include intestine that has undergone prolonged or excessive distention or that is in any inflammatory condition, such as after intestinal manipulation or associated with primary inflammatory diseases such as DPJ or Salmonella enteritis. Numerous drugs have been investigated in humans with POI, but currently there is no prokinetic agent that has been found to be safe and effective.691 The financial pressures associated with equine patient management often compel attending veterinarians to “experiment” with a range of putative prokinetic drugs.
Bethanechol is a methyl derivate of carbachol and is an ACh receptor agonist. The drug acts both at the level of the myenteric plexus and directly on intestinal smooth cells through muscarinic receptors. The actions are primarily mediated through activation of M3 receptors, although M2 receptors are also involved to a lesser extent.692 There is evidence in other species that activation of M2 receptors not only facilitates intestinal contraction but also may lead to antinociception.693 Bethanechol is not degraded by the enzyme anticholinesterase. As anticipated the drug has a range of cholinergic side effects, including abdominal discomfort, sweating, and salivation, although these are minimal when the drug is administered at 0.025 to 0.05 mg/kg of body weight SC. Bethanechol is one of the most useful prokinetic agents in equine practice, as it exerts effects throughout the gastrointestinal tract; however, it does not appear to be commonly used.694 It is most commonly used in the management of delayed gastric emptying and slowed small intestinal transit. Experimentally the drug has been shown to significantly increase gastric contractility and hasten the emptying of liquid and solid phase markers from the stomach of normal horses.695,696 The drug has potent effects in the hindgut as well. Bethanechol increases both the relative strength and duration of wall contractions in the cecum and right ventral colon and consequently speeds up cecal emptying.665 When given to ponies at a dose of 0.05 mg/kg SC, the drug increased electrical activity in the large colon for approximately 80 minutes.684 There are wide ranges in reported dose rates of bethanechol, with doses up to 0.25 mg/kg recommended for oral administration in the management of delayed gastric emptying.
Neostigmine increases receptor levels of ACh by inhibiting cholinesterase. The drug (0.022 to 0.025 mg/kg IV) promotes cecal and colonic contractile activity and enhances cecal emptying in normal ponies.665 Neostigmine has been used in the management of small intestinal ileus but significantly delayed the emptying of 6-mm beads from the stomach of normal adult horses.697 A survey of prokinetic use indicated that neostigmine was more commonly used in the management of large intestinal disease.694
Metoclopramide is a moderate partial 5-hydroxytryptamine 4 (5HT-4)–receptor agonist, a moderate 5HT-3–receptor antagonist, and an antagonist of both dopamine 1 (DA1) and 2 (DA2) receptors. It has been suggested that the 5HT-4 agonist properties are primarily responsible for the prokinetic effects of metoclopramide. Antagonism of prejunctional DA2 receptors facilitates ACh release and smooth muscle contraction. The effect on dopamine receptor antagonism is absent in newer benzamides. Metoclopramide crosses the blood brain barrier, where its antagonist properties on central DA2 receptors can result in extrapyramidal signs, including seizure. These have been observed when the original reported dose of 0.25 mg/kg, given by intravenous infusion over 30 minutes, was used in practice.698 These signs were responsible for poor acceptance of the drug in equine practice. An experimental bolus of endotoxin caused a delay in gastric emptying, as assessed using the acetaminophen absorption test; this delay was partially ameliorated through pretreatment with metoclopramide (0.125 mg/kg in 1 L infused over 15 minutes), but again the investigators reported adverse side effects.699 Most investigators have failed to demonstrate significant effects of metoclopramide in experimental animals, but constant intravenous infusion (0.04 mg/kg/hr) in a population of postoperative horses significantly decreased the volume and duration of gastric reflux over control and intermittent drug infusion groups.700 Infusion was well tolerated and was superior to intermittent infusion or no treatment at all.
Cisapride is a second-generation benzamide that is commonly reported to act as a primary 5HT-4 agonist and 5HT-3–receptor antagonist. Furthermore, stimulation of 5HT-4 receptors within the enteric nervous system enhances release of ACh from the myenteric plexus, thereby promoting intestinal contraction. In vitro studies of equine jejunum concluded that the contractile actions of cisapride in the species were primarily mediated through 5HT-2 and not 5HT-4 receptors and that the response was noncholinergic.701 Several reports suggest efficacy of cisapride in the management of intestinal disease in horses, including the resolution of persistent large colon impaction, treatment of equine grass sickness, and prevention of POI in horses after small intestinal surgery (0.1 mg/kg body weight IM during postoperative period).702-705 Cisapride has the potential to cause adverse cardiac side effects mediated through blockage of the rapid component of the delayed rectifier potassium current that include lengthening of the QT interval and development of torsades de pointes, a potentially fatal arrhythmia. These adverse effects have resulted in drug withdrawal in the United States and in many other countries. Another benzamide, mosapride, resulted in increased myoelectric activity of the small intestine and cecum of horses after oral administration. The authors reported that mosapride is a selective 5HT-4 agonist; consequently the reported effects are surprising given that the actions of 5-HT on the equine jejunum were reported to be primarily mediated through activation of 5HT-2 and 5HT-3 receptors.701 Mosapride is marketed in several Asian countries and is not available in the United States.
Domperidone acts as a competitive antagonist at peripheral DA2 receptors; these receptors are inhibitory. The drug has been used to manage gastroparesis in humans for many years.706 The primary use in equine practice is in the management of mares grazing endophyte-infected tall fescue (1.1 mg/kg/day PO), principally because of drug-enhanced prolactin release from the anterior pituitary. The potential prokinetic effects of domperidone have not been extensively studied in horses, but a modest efficacy of domperidone (0.2 mg/kg IV) was demonstrated in a model of experimental ileus in two ponies.702
Erythromycin is a direct motilin receptor agonist acting directly on smooth muscle cells as well as within the enteric nervous system to facilitate the release of ACh and motilin. Erythromycin enhances gastric emptying in normal horses but in contrast to most other species has a more pronounced effect on the hindgut.695,707 This is somewhat surprising given that a higher density of motilin receptors was reported in the duodenum than in either the cecum or pelvic flexure of adult horse.708 Erythromycin lactobionate (1 mg/kg IV) hastens cecal emptying in normal animals and induces propagating colonic MMC-like activity across the colon. Administration is often associated with defecation and abdominal discomfort.
Several problems limit use of erythromycin as a prokinetic agent in equine practice. The principal use in human medicine has been restricted to management of acute exacerbations of diabetic gastroparesis, facilitation of feeding tube placement, and upper gastrointestinal endoscopy.709 Although it has been shown to enhance emptying in normal horses, it was not as effective as bethanechol.695 Given its potent prokinetic effects in the cecum, erythromycin may be helpful at preventing cecal impaction in horses after anesthesia. However, its effectiveness on cecal motility appears to be markedly reduced in the immediate postoperative period.659 High doses, constant infusion, or prolonged use of erythromycin also induces receptor tachyphylaxis and therefore reduced efficacy. There is also evidence that as little as 2 hours of intraluminal distention leads to a reduction in the total number of motilin receptors and in the amount of motilin receptor mRNA, although erythromycin binding to remaining motilin receptors is not affected.710 Erythromycin can induce diarrhea in adults; therefore administration over many days should be avoided. The diarrhea induced by erythromycin is most likely mediated by C. difficile overgrowth and toxin elaboration.711
Given that opiates have an inhibitory effect on normal intestinal motility, it is not unreasonable to assume that antagonists may have the potential for prokinetic activity. Naloxone (0.05 mg/kg IV) have been shown to induce contractile activity in the cecum and left colon.712 The administration of naloxone was often followed by defecation within 15 to 20 minutes. Naloxone has not been beneficial in preventing POI in human beings.713 An in vitro study reported a direct contractile effect of the peripherally acting opioid antagonist N-methylnaltrexone on the circular muscle layer of the jejunum and large colon.714 The authors suggested a potential role for the drug in preventing intestinal complications associated with the use of morphine for pain relief.
α2-Adrenoreceptor antagonists, such as yohimbine and telazoline, counteract increased sympathetic outflow in response to nociceptive stimulation. Yohimbine (75 μg/kg by slow intravenous infusion) hastens cecal motility and emptying in normal ponies and also attenuates the negative effects of endotoxin on motility.655,665
Lidocaine (also referred to as lignocaine in some countries) appears to be the most commonly used putative prokinetic agent in the management and prevention of POI.694 Intravenous infusion of lidocaine may suppress primary afferent neurons, thereby limiting reflex efferent inhibition of motility. It is also possible that lidocaine may block the inhibitory effect of NANC neurotransmitters on smooth muscle.715 Any positive effect of lidocaine could also be related to the drug’s significant antiinflammatory properties. These include amelioration of the cytokine response to endotoxemia, reduced neutrophil free-radical production, impaired leukocyte phagocytic function, and inhibition of leukocyte migration through suppression of chemokines.716 As discussed earlier, inflammation may be the most important initiator of intestinal stasis in the postoperative period.
A combination of intraoperative and postoperative lidocaine infusion is commonly used as a preventative strategy against POI. Postanesthetic treatment typically involves a slow intravenous bolus of 2% lidocaine (1.3 mg/kg) followed by a constant infusion at 0.05 mg/kg/min for 24 hours. The recommended target range for serum concentration of lidocaine is 1 to 2 mg/dL. In a prospective study of horses undergoing laparotomy for colic, the authors failed to demonstrate any difference between lidocaine and saline infusion with respect to return of borborygmi, time to first feces, or gastric reflux.717 The authors did, however, report some positive differences in several ultrasound parameters, including jejunal diameter and the apparent volume of peritoneal fluid. Unfortunately only several of the horses in the study had a small intestinal lesion that required resection and anastomosis, and only two animals developed significant POI, one in each group. Lidocaine infusion was also contrasted with saline infusion in a group of horses with DPJ or POI.715 Criteria for inclusion in that study included animals with net gastric reflux of more than 2 L/hr for 24 hours or a cumulative reflux volume of more than 20 L in less than 24 hours. Significant benefits of lidocaine infusion included a reduction in the duration of refluxing, hastened passage of feces, and a shorter total period of hospitalization for survivors. There were horses that did not improve in response to lidocaine infusion, and animals that continued to produce net gastric reflux throughout the 24-hour infusion period had a poorer outcome.
The rate of lidocaine infusion requires close monitoring, because infusion can be associated with reversible side effects that include muscle fasciculations, ataxia, and seizure. The drug is highly protein-bound, so hypoproteinemic horses may be more susceptible to signs of toxicity.
MEDICAL DISORDERS OF THE LARGE INTESTINE
Samuel L. Jones
ACUTE DIARRHEA
Diarrhea in the horse can be defined as the passage of fecal material that has increased water content. It can vary from soft, formed stools with a mild to moderate increase in water content to projectile fecal passages that contain little solid matter. The passage of excessive water in the feces reflects disruption of the normal balance of fluid and electrolyte secretion and absorption in the intestinal tract. In adult horses diarrhea results almost exclusively from disorders of the large intestine, although diarrhea may be a feature of some descending small colon disorders. Diarrhea can result in significant losses of water, electrolytes, and plasma protein and is often accompanied by local and systemic inflammatory responses.
Diarrhea disorders in adult horses can be divided into those characterized by inflammation of the cecum and large intestine (typhlitis, colitis) and those in which there typically is not an inflammatory response. Inflammatory disorders can be those characterized by an acute inflammatory response (salmonellosis, Potomac horse fever [PHF], and clostridiosis, described later), disorders associated with endoparasitism (small and large strongyle larval migration or encystation), disorders arising from toxicity (cantharidin toxicity, described later, and nonsteroidal antiinflammatory toxicity, described on p. 754), and disorders included under the umbrella term inflammatory bowel disease (see p. 730). Disorders that can manifest with diarrhea but that typically do not have colonic inflammation include those in which there is increased intestinal hydrostatic pressure (congestive heart failure, cirrhotic liver disease), intraluminal osmolarity, and poorly defined disorders in which fluid secretion may be stimulated by the enteric nervous system.
Colitis is active inflammation within the colon that is usually associated with myriad local and systemic pathophysiologic events. Diarrhea is an important problem in horses with colitis, but impaired cardiopulmonary function, coagulopathies, and other sequelae of activation of inflammatory mediator cascades and septicemia can be most life-threatening. Complications that occur in colitis patients, regardless of cause, include overwhelming sepsis triggered by endotoxemia, septicemia and hematogenous organ colonization by bacteria, immune suppression and susceptibility to superinfection with bacteria or fungi, cecum or colon infarction, jugular vein thrombosis, and laminitis.
A variety of inflammatory cells and mediators affect the equine colon. With acute colitis the neutrophil is the effector cell, and the cascade of activation of inflammatory mediators associated with acute colitis is designed to bring neutrophils to where chemical signals indicative of bacterial infection have been detected. Signs of sepsis associated with endotoxemia (see p. 712) frequently accompany, or even precede, diarrhea in horses. Severe tissue inflammation can result in ulceration of the mucosal epithelium, resulting in chronic malabsorption and protein-losing enteropathy. Malabsorption of volatile fatty acids (VFAs) and metabolic alterations that result from excessive production and release of inflammatory mediators can lead to an energy deficit and catabolism of body tissues.
Specific diseases affecting the equine large colon (e.g., salmonellosis, PHF, clostridial colitis, and cantharidin toxicity) may have different activators of local and systemic inflammatory responses, and they can produce unique toxins that further contribute to tissue injury. Regardless of the cause, the clinical problems of affected horses are often similar, and the clinician must consider treatments that modify inflammatory changes and replace losses of fluid, electrolytes, and plasma protein. In many cases the cause of the diarrhea is not determined. Descriptions of important causes of diarrhea in horses follow.
Salmonellosis
Salmonella bacteria possess an array of virulence factors that confer attributes of mucosal adhesion and invasion, produce enterotoxins that stimulate intestinal fluid secretion, activate local inflammation, including recruitment of inflammatory cells and the release of their mediators, cause local cytotoxic effects, and initiate systemic responses attributable to LPS.
Epidemiology
Numerous Salmonella serotypes have been associated with equine colitis, and overall more than 2500 serotypes of Salmonella have been described. Salmonella Typhimurium is the most frequently isolated serotype in horses, with dozens of other serotypes isolated sporadically. There are many reports describing clusters of cases of equine salmonellosis in which specific Salmonella serovars predominate. Nosocomial infections associated with Salmonella Krefeld,718-720Salmonella Typhimurium,719Salmonella Anatum,721 and Salmonella Infantis722 have been reported in recent years. Pertinent features of these bacteria are the ability to withstand a wide range of environmental conditions, the ability to rapidly invade and spread within the host (and thus be shed into the environment through the feces), and the range of severity of illness that results from infection.
Other key elements that influence whether clusters of cases will occur given the presence of a particular Salmonella serovar in the environment are availability and population density of susceptible hosts and the size of infective dose of the pathogen. It is for these reasons that veterinary hospitals, breeding farms, and other facilities that may have a high density of horses are most vulnerable to the development of Salmonella outbreaks. In one study at a veterinary teaching hospital, horses that were at greatest risk for developing salmonellosis were those treated with antimicrobial drugs or admitted for treatment of colic.723 Such horses will include the majority of patients in any equine referral hospital. Breeding farms are susceptible to Salmonella outbreaks (or other enteric infections) because of the concentration of large numbers of immunologically immature newborn animals. In either case the particular Salmonella organism involved in disease outbreaks may not have to be especially virulent, because the inherent susceptibility of the host provides the microbe the opportunity to colonize and invade the host. Also of importance is the ability of the organism (inherent or acquired) to persist in the environment, lying in wait for both susceptible hosts and environmental conditions that favor its propagation and dissemination.
Serovars or strains of Salmonella that have newly acquired virulence plasmids can rapidly become established and spread among farms, sales areas, veterinary clinics, and veterinary teaching hospitals. Often the origin of these “new” bacteria is undetermined, but in some cases the organism can be traced to contaminated feed,724 a specific shedder introduced into the environment, domestic or feral animals (barn cats), birds, and wildlife.
Salmonellae are ubiquitous in the environment, and the prevalence of fecal shedding varies by the group of horses sampled and the method of detection. Prevalence of fecal shedding, based on fecal culture, in asymptomatic horses admitted to teaching hospitals varied from 1% to 5%.725,726 In a nationwide survey of prevalence of Salmonella in fecal samples from horses on farms and ranches. Salmonella bacteria were cultured from less than 1% of horses.727 Use of PCR to detect Salmonella DNA in horse feces resulted in a prevalence rate of 17% in horses admitted to a teaching hospital for lameness examination and greater than 60% in hospitalized horses with gastrointestinal disease.728 In that report the PCR technique was determined to be specific for Salmonella DNA, but the test cannot differentiate between shedding of live bacteria and DNA from dead organisms.
Horses are not considered to be carriers per se of Salmonella, because no host-adapted Salmonella species have been identified in horses. Salmonella Abortus equi has now disappeared in the United States. Horses that shed Salmonella species in the feces usually do so transiently for several days to weeks; infrequently horses may shed salmonella bacteria for several months.
In acutely affected horses, large numbers of highly infective salmonellae can be shed in the diarrheic feces. Susceptible animals such as young foals, hospitalized horses receiving antimicrobial drugs, and horses under stress can become ill after becoming infected by numbers of salmonellae 100 to 1000 times less than those required to infect immunocompetent normal horses. Thus particular care should be taken in the management of horses and foals with diarrhea in environments in which there are animals at risk (e.g., hospitals, breeding farms, racetracks). Asymptomatic shedders generally pass relatively small numbers of salmonellae in the feces and do not appear to pose an important threat to healthy horses, although asymptomatic shedders have been responsible for outbreaks of salmonellosis in hospitals and on breeding farms.
Clinical Findings
Salmonellosis typically is characterized by an acute colitis that results in profuse diarrhea and occasionally abdominal pain. Horses with salmonellosis often have signs of sepsis associated with endotoxemia and suffer from cardiovascular shock, vascular leak syndrome, and coagulopathies. Horses are usually febrile, tachycardic, moderately to severely obtunded, and dehydrated. With other clinical syndromes of salmonella infection, diarrhea is not a feature. These syndromes include fever and leukopenia, colic, and proximal enteritis with gastric reflux.
Diagnosis
Confirmation of salmonellosis requires bacteriologic culture of Salmonella bacteria. Multiple fecal cultures for Salmonella species should be performed on all horses with diarrhea. It is recommended that at least three to five fecal samples collected 12 to 24 hours apart be submitted to increase the sensitivity of culture.729 Samples with little solid matter often yield negative culture results, even when the horse is infected with Salmonella. Formed fecal samples are more likely to result in a positive culture result from infected horses. A 5- to 10-g amount of feces should be submitted for culture in selective media such as tetrathionate broth or selenite broth and brilliant green agar or XLD agar. Culture of a rectal mucosa biopsy may be a useful adjunctive test for Salmonellosis. PCR is a sensitive screening tool for detection of fecal Salmonella DNA and is important for environmental monitoring and biocontrol measures in hospitals.
Treatment and Prevention
In most cases of salmonellosis, aggressive treatment facilitates resolution of the severe diarrhea and associated metabolic disorders within 7 to 10 days of the onset of illness. Intravenous administration of polyionic fluids is required to replace fluid and electrolyte losses and to augment preload in horses with poor venous return (see the Fluid Therapy for Horses with Gastrointestinal Diseases section [p. 767] and Chapter 44 for more comprehensive information about fluid therapy in horses with diarrhea). Plasma may be required to replace lost plasma proteins and increase plasma colloidal pressure. Alternative colloidal fluids, such as hetastarch, may be quite useful and more cost-effective than plasma in horses with vascular leak syndrome and hypoproteinemia. Flunixin meglumine is often used to treat inflammation in horses with signs of sepsis. Flunixin and other NSAIDs should be used with caution and at the lowest dose possible to prevent worsening of colonic mucosal damage or inhibition of mucosal repair. Other antiinflammatory strategies may be used to treat sepsis associated with salmonellosis. Total or partial parenteral nutritional support is often indicated to provide adequate calories and amino acids during the most debilitating period of the illness. Antimicrobial administration to horses with suspected or known salmonellosis is not universally practiced. Antimicrobial administration may decrease the spread of salmonella bacteria to other organs and may have some direct effect on salmonella bacteria in the colon. Traditionally, use of antimicrobial drugs such as chloramphenicol, trimethoprim-sulfa (TMS), gentamicin, and cephalosporins has not appeared to accelerate resolution of signs of colitis. Fluoroquinolones, such as enrofloxacin, although potentially arthropathic in young horses, may be more effective because of high lipid solubility and bactericidal activity against Salmonella bacteria.
Horses that have severe diarrhea and sepsis for 10 days or longer are unlikely to survive, even with intensive therapy, because they often have extensive ulceration of colonic mucosa and chronic, severe inflammation within the wall of the colon. Ulceration may be exacerbated by administration of NSAIDs. Complications such as catheter-associated thrombophlebitis, colonic infarction (Fig. 32-55), colonization of other organs by salmonellae or other enteric bacteria, and laminitis can occur.
Measures designed to prevent the spread of Salmonella in an environment with potentially susceptible hosts need not be excessively laborious or expensive. The goal is to minimize the size of the infective dose of an enteric pathogen to which a susceptible host may be exposed. Thorough cleaning of areas where fecal contamination is likely and prevention of mechanical distribution of contaminated material are the most important measures that should be taken. Extensive use of disinfectants may not be necessary if cleaning measures are adequate. Cleaning must include removal of organic debris, which can be accomplished with several products designed for that task. Areas that require particular attention are stalls, including water buckets or automatic watering systems, drains, and cracks in the floors and wall; stall implements; surgical areas, including drains; and nasogastric tubes and pumps.
If a diarrheic horse is in the environment, it should be isolated to the degree possible. Bedding material should be removed frequently to minimize accumulation of potential enteropathogens. Personnel entering the stall should be restricted to the professional staff, and they should wear disposable plastic boots. Footbaths with disinfectant often are not effective because they quickly accumulate organic material that interferes with the disinfectant activity of the footbath. Once a horse vacates a stall, the stall should be thoroughly cleaned, allowed to dry, disinfected, and determined to be negative for Salmonella by culturing selected sites in the stall (floor, drain, waterer). Personnel should use common sense when dealing with diarrheic animals. If bedding is being blown about by wind or if mechanical blowers are used to clean aisles, then potential enteric pathogens may be readily spread to other horses.
Potomac Horse Fever
PHF is an infectious enterocolonic disorder caused by N. risticii (formerly Ehrlichia risticii).730,731 The organism is an obligate intracellular parasite, infecting peripheral monocytes and macrophages, colonic and small intestinal epithelial cells, and colon mast cells.732 The pathophysiology of the disease is incompletely understood, although horses infected with N. risticii often have clinical signs and complications similar to those in horses with salmonellosis. After experimental inoculation with N. risticii horses had a mild, transient fever 2 to 4 days after infection.733 By 10 to 14 days after experimental infection horses became febrile, had a poor appetite, and exhibited mild to severe gastrointestinal signs ranging from mild colic and soft stool to profuse diarrhea.
In clinical cases of PHF, there are signs of sepsis, including fever, leukopenia, congested mucous membranes, and hypercoagulability. The early fever observed in experimental cases is usually not detected by owners, and when clinical signs are seen it is presumed that the horse was infected 10 to 14 days previously. Hypoproteinemia is a frequent finding in horses with clinical cases of PHF, which reflects loss of serum prote in through inflamed intestinal mucosa. It is interesting to note, however, that the magnitude of intestinal inflammation is typically much less than with salmonellosis, yet the magnitude of hypoproteinemia (total protein <3 g/dL) can be as severe.
Laminitis is a frequent sequela and may occur in 30% in horses with PHF. N. risticii has also been associated with abortion in mares, although this is an unusual occurrence.734 In some horses that show significant seroconversion to N. risticii, laminitis is the only clinical sign.
Mode of Transmission
Although originally described as a disease of horses living near the Potomac River in Maryland and Virginia, horses with serologic evidence of exposure to N. risticii have been reported in most states. Because early research on PHF demonstrated that infection was readily transmitted through blood,733 it was presumed that the natural mode of transmission involved an insect or arthropod vector. However, several studies have failed to demonstrate any such vectors.735,736
The association between an affected horse and proximity to a river (within 5 miles) remains strong. Recently investigators have found a possible link between this association and how the disease may be transmitted. N. risticii and Neorickettsia helmintheca were found to share a high degree of DNA homology.737N. helmintheca is transmitted to mammals via a trematode that parasitizes fish and aquatic snails.737 This prompted investigators to search for evidence that N. risticii might reside in trematodes and their hosts found in riverine inhabitants. N. risticii DNA was detected in operculate snails (Pleuroceridae: Juga species) collected from stream water in a northern California pasture in which PHF is enzootic.738 Moreover, N. risticii was detected in trematode stages found in the secretions of freshwater snails and in aquatic insects.739,740 The sequences of these genes were virtually identical to those of the genes of an equine N. risticii strain isolated from horses located on a property near the snail collection site. Further work demonstrated N. risticii DNA in two trematodes. Acanthatrium species and Lecithodendrium species, found in bats and swallows, suggesting that these animals may serve as a reservoir for the organism.741 Oral transmission of N. risticii with infected cell cultures has been produced experimentally,742 supporting a role for ingestion of N. risticii, rather than blood transmission, as the route of natural infection. Oral transmission and clinical signs of PHF were demonstrated in horses fed infected aquatic insects (caddis flies), suggesting that ingestion of infected aquatic insects may be the natural route of infection.743,744 Ingestion of water containing infected trematode stages released from aquatic snails may also be a route of infection.