LAMINITIS (FOUNDER)
Definition
Laminitis (“inflammation of the laminae”) is a disease that causes degeneration, necrosis, and inflammation of the dermal and epidermal laminae in the hoof wall of horses and ruminants.
Etiology and Pathogenesis
Because the epidermal laminae suspend the distal phalanx and therefore the body weight of a horse, laminar degeneration destroys the suspension mechanism and permits weight-bearing forces to push the distal phalanx ventrally. Failure of the laminar suspending mechanism causes a painful and potentially crippling lameness. Laminitis is often a sequela of digestive disturbances and other disorders that cause endotoxemia and elaboration of inflammatory mediators. Unless preventive measures are taken, laminitis often occurs after colonic torsion, proximal enteritis, colitis, grain overload, pleuropneumonia, and septic metritis (i.e., postparturient retention of the placenta).211-215
In horses, laminitis is sometimes seen following changes in feed, excess intake of cold water after strenuous exercise, grazing on lush spring grasses containing highly available carbohydrates, or persistent feeding of a high-concentrate ration.211,212 Laminitis may also be precipitated in horses by administration of high levels of corticosteroids,216 which decrease protein synthesis and potentiate digital vasoconstriction and microthrombosis.217 Excessive weight bearing in the support limb during severe lameness of the contralateral limb can produce laminitis, as can work on hard ground or extreme exhaustion and dehydration.211,214,215 A water-soluble toxin in black walnut shavings also has been shown to induce laminitis in horses.218
In cattle, laminitis is most often seen immediately after calving in fat heifers that have been fed excess concentrates and kept on concrete surfaces.219
Pathophysiology
The pathophysiology of laminitis has not been totally elucidated; however, laminitis is often considered a local manifestation of a variety of disorders that cause a generalized metabolic disturbance. Several factors may produce laminar degeneration. The integrity of the laminar suspending mechanism depends on maintenance of proteins in the cytoskeletal networks, intercellular junctions, and basement membrane of the epidermal laminar cells. This process is energy dependent, and disorders that decrease laminar perfusion or decrease protein synthesis have the potential to initiate laminar degeneration. In addition, laminar degeneration may be initiated by disorders that cause the elaboration of factors cytotoxic to the epidermal laminae, by disorders that activate metalloproteinases, or by disorders that increase the tension on the laminae. Because the laminae and their sustaining vasculature are confined within the rigid hoof wall, factors that cause tissue swelling (e.g., inflammation, edema) can theoretically increase the interstitial tissue pressure beyond critical capillary closing pressure, producing a compartment syndrome and functional ischemia of the corium. Opening of arteriovenous shunts within the corium occurs during carbohydrate-overload laminitis, but such shunting has not conclusively been shown to be the major factor producing laminar degeneration.
Laminitis is often a sequela of diseases producing gram-negative sepsis and endotoxemia, but experimental administration of endotoxin has failed to produce laminitis. However, overingestion of grain or other feeds containing large amounts of highly available carbohydrates is thought to produce endotoxemia and is the most common cause of acute laminitis. Carbohydrate overload results in bacterial overgrowth in the colon, lactic acidosis, decreased colonic pH, colonic mucosal slough, and death of colonic bacteria with concomitant liberation of endotoxin. Degeneration of the colonic mucosa is thought to allow endotoxin to gain access to the portal circulation. The mechanistic link between endotoxemia and laminar degeneration is not totally understood; however, endotoxin that crosses a compromised bowel wall into the portal circulation is removed by reticuloendothelial cells in the liver, where it likely triggers activation of leukocytes and upregulation of proinflammatory cytokines observed during colitis and grain overload. Hyperimmune serum to gram-negative core antigens has a strong protective effect on horses at high risk of developing laminitis as a result of intestinal crises or carbohydrate overload.220
Sequential biopsies of the epidermal laminae and corium during the development of grain-overload laminitis indicated that initial laminar degeneration was most compatible with ischemic or cytotoxic injury, and that a major influx of inflammatory cells, edema, and microthrombosis did not precede laminar degeneration but occurred later and was likely to accelerate the degeneration.221
Recent evidence indicates that proinflammatory cytokine expression is increased222 and leukocytes are activated223 and begin to emigrate into the perivenular interstitium of the laminar dermis224,225 during the developmental stages of black walnut extract—induced laminitis. Levels of latent metalloproteinase also increase in the plasma and vascular and perivascular tissues of the dermal laminae during the developmental stages of black walnut extract—induced226 and carbohydrate-overload laminitis.227 Matrix metalloproteinases (MMPs) in the laminar region are normally located in the laminar epidermis, where they are thought to play a major role in laminar epidermal remodeling to facilitate hoof wall growth and migration.228 Early histologic lesions during the developmental stages of laminitis are compatible with excess activation of MMPs and disruption of cell-cell and cell—basement membrane linkages.221,227 Increased expression of proinflammatory cytokines and activation and emigration of leukocytes during the developmental stage of laminitis may alter the balance between normal levels of activation and inhibition of constitutive MMPs in the laminar epidermis, leading to disruption of the laminar suspending mechanism. Continuous 48-hour application of an ice-water bath to the distal limb is thought to decrease MMP enzyme activity and was recently shown to protect the chilled digit while the contralateral nonchilled digit developed laminitis after carbohydrate overload.229
Because perfusion of the most dorsal laminae depends on vessels that course through vascular canals in the distal phalanx, distal migration of the distal phalanx caused by laminar degeneration may compromise laminar perfusion and result in a cycle that intensifies the laminar lesion. It is also theorized that the pain associated with laminar degeneration may cause release of catecholamines that potentiate peripheral vasoconstriction and further diminish laminar perfusion.
Clinical Signs
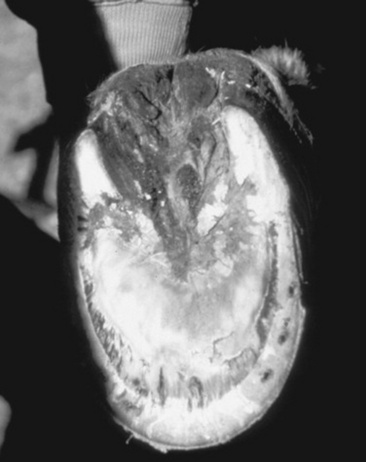
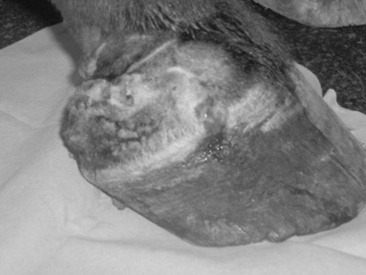
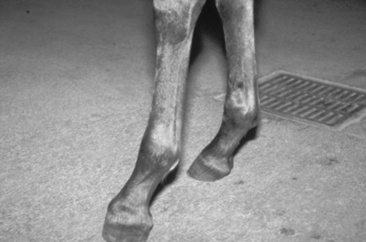
The signs of acute laminitis are lameness, depression, anorexia, and reluctance to move. Early in the disease, affected animals often paddle or shift weight from one foot to the other. Increased pulsations can be palpated and sometimes visualized in the digital arteries. Hoof-tester examinations reveal sensitivity over the sole at the toe, and tapping on the hoof wall at the toe may elicit pain. Severely affected animals may be unwilling to pick up a forefoot or hindfoot because they are reluctant to bear full weight on the contralateral foot (Obel grade III lameness230). The forefeet are usually affected more often and more severely than the hindfeet in horses, and the most dorsal laminae are more severely involved than laminae in the heel regions. Therefore, horses with laminitis typically draw the hindlimbs under the body and place the forelimbs forward to shift weight to the hindquarters and load the heels more than the toes. In ruminants the hindlimbs are most often involved, and affected animals characteristically become recumbent. In severe cases, when laminar degeneration circumferentially involves the foot, a noticeable depression can be palpated along the coronary band. In such cases, exudation is sometimes noted in the coronary region, and the skin may separate from the hoof wall. These signs indicate that the distal phalanx has shifted distally with respect to the hoof wall (i.e., severe rotation or sinking of the distal phalanx) and suggest a poor prognosis. With dislocation of the distal phalanx, the sole loses its normal cupped appearance and is flat or bulges between the toe and apex of the frog. Pulse and respiratory rates are usually increased, and other clinical signs reflect underlying disease processes.
Signs of chronic laminitis are lameness and abnormal conformation of the foot. The sole is flat or dropped, the white line is widened, and the hoof wall shows signs of uneven growth. Irregular rings of horn, closely spaced at the toe and more widely spaced near the heels, encircle the hoof wall. In ruminants the sole softens and assumes a light-yellow discoloration. Hemorrhages can often be identified in the abaxial white line region, and fissures parallel to the coronary band may be seen in the hoof wall. The signs of subsolar abscessation sometimes mimic those of laminitis; however, abscesses most often involve only one foot and rarely cause anorexia, depression, or increased pulse and respiratory rates.
Clinical Pathology and Radiology
Clinical pathologic findings during the development of acute laminitis most frequently represent alterations associated with underlying disease processes, such as enteritis, colitis, or metritis, and are not pathognomonic for laminitis. During the onset of alimentary laminitis, packed cell volume, total plasma protein, heart rate, respiratory rate, rectal temperature, and blood glucose level are often elevated. Arterial blood pressure is usually elevated in horses but depressed in ruminants.231 Neutropenia often precedes laminitis caused by disorders that produce endotoxemia; neutrophilia and eosinopenia are often seen later. Changes are thought to reflect compartmental fluid shifts and a stress response consistent with release of glucocorticoids and catecholamines. Horses with chronic severe laminitis, in which euthanasia was deemed necessary, had total WBC counts that were significantly elevated (5,000 to 18,000/μL) compared with control horses and horses that recovered from less severe bouts of laminitis.232 The persistent neutrophilia was presumably a response to infection and was thought to signify an unfavorable prognosis.
Radiographic examinations should be performed on the affected digits of horses suspected to be developing laminitis. The initial examinations should include lateromedial and 65-degree dorsoproximal-palmarodistal projections. These views should be taken to assess the appearance of the distal phalanx, the soft tissues of the hoof wall and corium, and their relationship. Lateromedial examinations are periodically repeated to check the progression of the disease. Radiographic signs of laminitis include ventral displacement of the extensor process with respect to the coronary groove of the hoof wall, increased distance between the dorsal cortex of the distal phalanx and the surface of the hoof wall, and ventral rotation of the tip of the distal phalanx. Linear radiolucencies are noted interior to the hoof wall in cases where the corium has separated from the epidermal laminae. Increasing degrees of rotation of the distal phalanx and increases in the distance between the dorsal surface of the distal phalanx and the hoof wall indicate progression of the disease (Fig. 38-30).
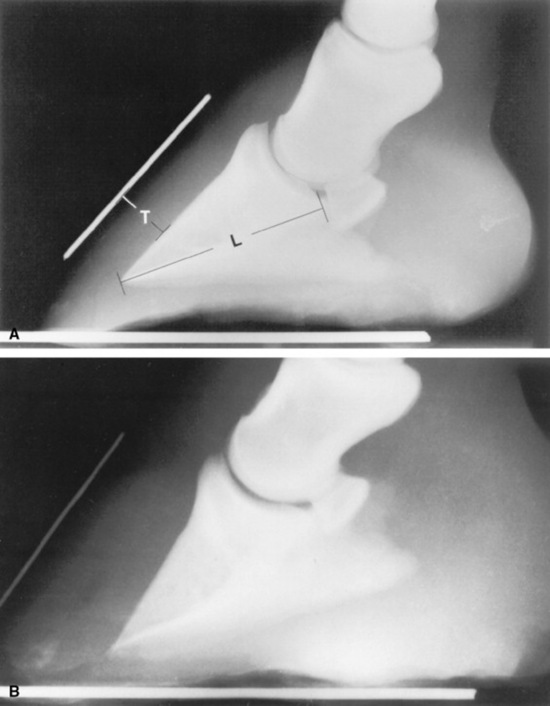

Fig. 38-30 A, Lateromedial radiograph of a normal digit. Two radiopaque markers can be seen. One has been placed on the block below the foot to mark the bearing surface of the wall, and the other marker identifies the location of the dorsal surface of the hoof wall. Notice that the dorsal surface of the hoof wall and the dorsal cortex of the distal phalanx are parallel, and that the distance between them, the soft tissue thickness (T), is approximately 25% of the distance from the tip of the distal phalanx to the articulation of the distal phalanx and the navicular bone, that is, the length of the distal phalanx (L). B, Lateromedial radiograph of a digit from a horse with severe laminitis. The distal phalanx has dropped ventrally without rotating. This phenomenon is seen in some horses with laminitis. The most consistent radiographic manifestation in such cases is an increased distance between the dorsal cortex of the distal phalanx and the dorsal surface of the hoof wall. The soft tissue thickness, as measured between the dorsal cortex and the dorsal surface of the hoof wall, in this case is 45% the length of the distal phalanx. The soft tissue thickness is normally less than 28% of the distal phalanx length for thoroughbred racehorses. C, Lateromedial radiograph of a digit from a horse with severe laminitis. Note the linear radiolucency dorsal to the distal phalanx (arrowhead). This lucency indicates a separation between the corium and primary epidermal laminae and marks the inner aspect of the hoof wall (arrows). The dorsal cortex of the distal phalanx is rotated approximately 14 degrees with respect to the inner surface of the hoof wall. Note that the dorsal and inner surfaces of the hoof wall are not parallel. This is the result of rasping along the distal portion of the dorsal surface of the hoof wall. The soft tissue thickness in this case is greatly increased to almost 42% of the distal phalanx length.
Because variations in technique affect subsequent radiographic distance and angle measurements, it is essential to standardize the radiographic procedure to detect small changes between examinations. For the lateromedial radiograph, the foot is cleaned and placed on a wooden block approximately 3 inches (7.5 cm) thick. A radiopaque marker can be embedded in the dorsal surface of the block and along the dorsal surface of the hoof wall to aid in determining the amount of rotation of the distal phalanx. However, marking the surface of the dorsal hoof wall is generally not necessary when digital radiographs are available. A small section of metal wire or a groove can be placed in the proximodorsal hoof wall as a reference for measuring vertical displacement of the distal phalanx in repeated radiographs, and a thumbtack is often useful in marking the apex of the frog for radiographic and anatomic correlation before therapeutic shoeing.
The radiographic beam should be perpendicular to a sagittal plane through the digit and should be centered midway between the toe and heels, about 1 inch (2 to 3 cm) above the bearing surface of the wall. The radiographic cassette should be parallel to the sagittal plane through the digit and should be placed as close to the foot as possible. Using a consistent technique and performing the examination in a standardized manner permit straight lateral radiographs to be produced and allow accurate quantification of radiographic parameters so that subtle changes may be identified early.
One of the earliest and most reliable radiographic signs of laminar deformity is an increase in distance between the dorsal surface of the hoof wall and the dorsal cortex of the distal phalanx. When the laminar suspending mechanism fails, weight-bearing forces cause the distal phalanx to displace distally or rotate away from the dorsal hoof wall, and the increased distance between the structures can be quantitated radiographically. Increased distance between the dorsal hoof surface and the dorsal cortex of the distal phalanx was significantly associated with increased laminar deformity during laminitis.221
A laminar index measurement has been developed to reduce the need to account for differences in radiographic magnification when comparing radiographs from different hospitals, from different breeds, or from different sizes of horses. It is useful to calculate the laminar index adjacent to the proximal and distal aspects of the dorsal cortex of the distal phalanx. The proximal laminar measurement is taken as the shortest distance between the linear portion of the dorsal cortex of the distal phalanx and the dorsal surface of the hoof wall immediately distal to the extensor process of the distal phalanx; the distal laminar measurement is taken in the same way, 5 to 6 mm proximal to the tip of the distal phalanx (see Fig. 38-30). The proximal and distal measurements are used to produce proximal and distal laminar indices by expressing them as a proportion of the length of the palmar cortex of the distal phalanx, as measured from the tip of the distal phalanx to its articulation with the navicular bone. The palmar cortex measurement serves as an index of foot size, and if the proximal or distal measurements spanning the laminae are increased in relation to the length of the palmar cortex, laminar deformity has occurred. Both the proximal and the distal laminar index measurements should be less than 30% of the palmar cortex length. The index measurements ranged between 20% and 28% for nonlame racing thoroughbreds221,233 and were greater than 30% in horses with laminitis,234 ranging up to 50% to 55% in those with severe laminar deformity.221 If the proximal and distal laminar indices are almost equal and both are greater than 30%, the distal phalanx has sunk in relation to the hoof capsule, without rotation (see Fig. 38-30). When both indices are greater than 30% and the distal index is greater than the proximal index, sinking and rotation of the distal phalanx have both occurred. Sinking generally indicates that laminar degeneration involves more than the dorsal wall laminae and carries a worse prognosis than for horses with rotation alone.
Epidemiology
A survey of the risk factors associated with laminitis indicated that intact mares and stallions were at greater risk of developing laminitis than geldings. Ponies also accounted for a significantly greater number of laminitis cases than expected based on their proportion of the caseload. The peak incidence of new cases also corresponded with growth of lush spring grasses, suggesting that ingestion of large quantities of fresh grass is also a significant risk factor for pastured horses.235
Other risk factors include diseases that cause excess weight bearing or trauma in the digit and diseases that produce endotoxemia. Persistent feeding of a high-concentrate ration, stabling on concrete surfaces, long van trips, and exposure to or ingestion of black walnut wood products are also thought to be associated with an increased risk of laminitis. In addition, horses that previously had laminitis are at greater risk than other horses.
Necropsy Findings
Peracute cases may have total degeneration of the secondary epidermal laminae, which causes a separation between the primary epidermal laminae of the hoof wall and the collagen fibers of the corium. Abscessation may occur in the necrotic laminae or subsolar tissues. The distal phalanx may sink or may be rotated ventrally with respect to the hoof capsule, and the tip may penetrate the sole (Fig. 38-31). Severe cases are accompanied by fractures of the solar margin, osteomyelitis, or severe resorption of the distal phalanx. The necropsy findings generally demonstrate a variable degree of elongation of the epidermal laminae, which depends on the severity and duration of the problem (Fig. 38-32).
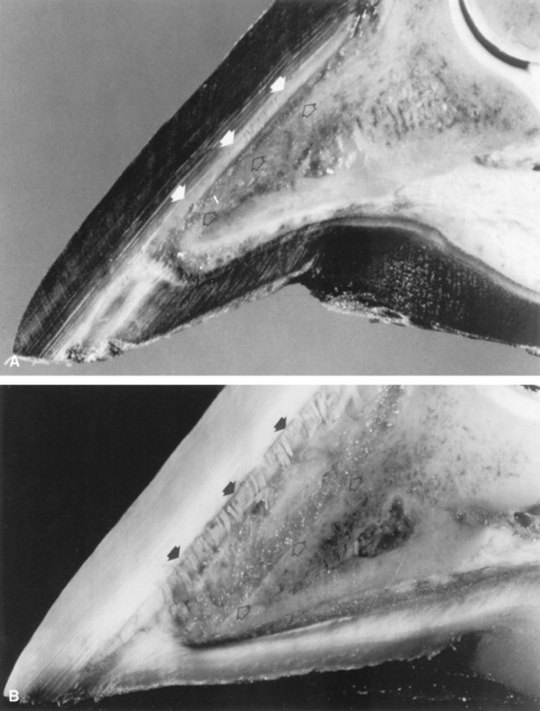
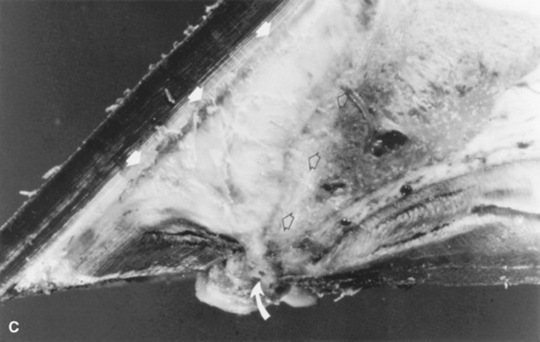
Fig. 38-31 A, Midsagittal section from the foot of a horse with a normal digit. Note the distance between the dorsal surface of the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows). The dorsal surface of the hoof wall and dorsal cortex of the distal phalanx are parallel. Compare with Figure 38-30, A. B, Midsagittal section from the foot of a horse with severe laminitis, a “sinker.” Note the increased distance between the dorsal surface of the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows). Also note that the distal phalanx has not rotated with respect to the hoof wall. Compare with Figure 38-30, B. C, Midsagittal section from the foot of a horse with severe laminitis. There is approximately an 18-degree rotation of the distal phalanx, and its tip has penetrated the sole (curved arrow). Note the increased distance between the dorsal surface of the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows). Compare with Figure 38–30, C.
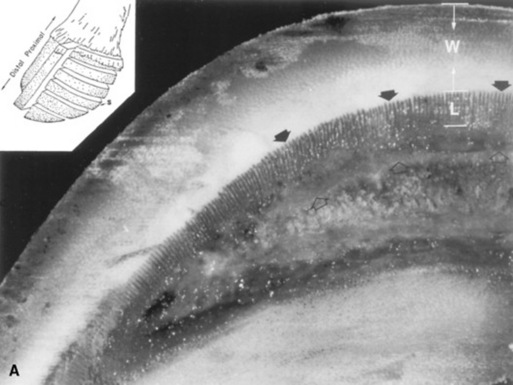
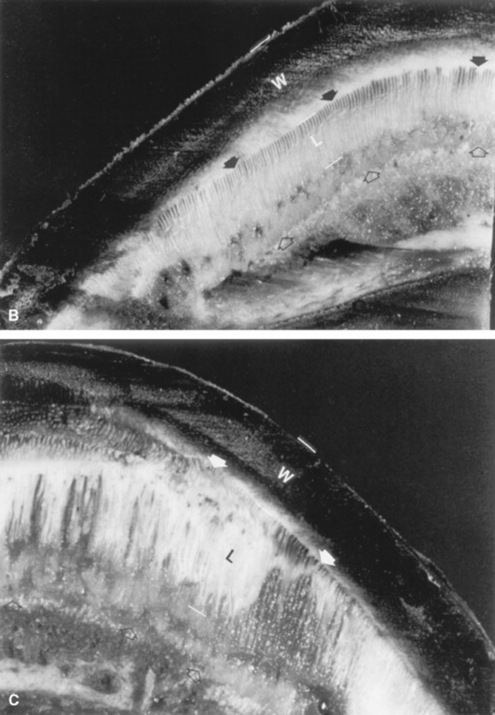
Fig. 38-32 A, Section of a healthy foot. The section was cut parallel to the coronary band, midway between the coronary band and the bearing surface of the hoof wall (inset, S). The length of the epidermal laminae (L) is approximately 33% of the thickness of the hoof wall (W) in normal horses. The distance between the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows) is normally less than 75% of the thickness of the hoof wall. B, Foot section, cut in a manner similar to that of A, from a foot of a horse with moderate laminitis. Note the increased length of the epidermal laminae (L). The increase in epidermal laminar length has allowed the distance between the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows) to become almost as large as the thickness of the hoof wall (W). C, Foot section, cut in a manner similar to that of A, from a foot of a horse with severe laminitis. Note the marked increase in length of the epidermal lamina (L). The distance between the dorsal cortex of the distal phalanx (open arrows) and the inner surface of the hoof wall (arrows) is abnormally increased to almost three times the thickness of the hoof wall (W).
Treatment
Treatment of animals developing acute laminitis should be considered an emergency. Laminar degeneration is underway by the time clinical signs of lameness appear, and even a few hours of delay in treatment can mean the difference between success and failure. Therapy should be initiated before development of clinical signs when the untreated animal is at high risk of developing laminitis (e.g., animals that recently ingested a large quantity of grain; mares with retention of placenta; horses with enteritis, colitis, or strangulating intestinal lesions).
General principles of therapy are aimed at eliminating the cause, promoting digital circulation, reducing tension on the laminae, reducing platelet activation and coagulation, and administering NSAIDs and free-radical scavenging agents to minimize digital inflammation and necrosis and to relieve pain.
ELIMINATING CAUSE
A laxative or purgative should be administered to animals that have ingested a large quantity of grain. In such cases, 3 to 4 L of mineral oil is usually given through a nasogastric tube. Intravenous administration of balanced electrolyte solution is indicated for horses with laminitis resulting from exhaustion, dehydration, and hypovolemia. Retained placentas should be treated appropriately if the placenta has not been expelled within 3 hours after parturition in mares. Antiendotoxin hyperimmune serum may be indicated for horses at risk of developing endotoxemia as a result of colon torsion, toxic diarrhea, toxic proximal enteritis, septic metritis, grain overload, or other disorders.
ADMINISTERING NONSTEROIDAL ANTIINFLAMMATORY DRUGS
Phenylbutazone is recommended, and at the onset of the syndrome it may be given once at a dose of up to 8.8 mg/kg intravenously (IV), usually followed by 4.4 mg/kg orally (PO) twice daily for several days. The dose should be tapered to 2.2 mg/kg (PO twice daily) as soon as possible. For horses with endotoxemia, flunixin meglumine* (1.1 mg/kg IV twice daily) is often used instead of phenylbutazone. Dimethyl sulfoxide (DMSO) also may be given daily (0.2 to 1.0 g/kg) for 2 or 3 days. To administer it IV in a 450-kg horse, 250 mL of the 90% solution is mixed in 3 L of balanced electrolyte solution and given slowly. DMSO should be diluted to a concentration that is less than 20% to avoid hemolysis when given IV. The use of aspirin (10 mg/kg IV or PO once daily) is sometimes advocated for its antiinflammatory and antiplatelet activities. Corticosteroids and adrenocorticotropic hormone (ACTH) are contraindicated because they decrease protein synthesis and may potentiate peripheral vasoconstriction and microthrombosis.
REDUCING TENSION ON LAMINAE
The force related to suspending the weight of the horse by the attachment between the hoof wall and the distal phalanx is likely to be a major factor producing laminar deformity in horses with laminitis. Reduction of laminar tension may be achieved by focusing the forces of weight bearing more on the frog and sole and reducing the amount of weight taken by the hoof wall. This can be accomplished by using frog support bandages or shoes, sole casts, or sand stalls. Elevation of the heel with an 18-degree wedge has been advocated to reduce the pull of the deep flexor tendon and decrease the tension on the laminae.236 This elevation can be achieved with a plastic-cuff shoe* that is bandaged or glued to the hoof wall and used with a frog support cushion† and an 18-degree heel wedge. An elevated-heel hoof cast has also been advocated and shown to reduce strain on the dorsal hoof wall laminae.237
For horses with severe acute laminitis, do not lower the heel in the acute stage, and avoid shoes that require the horse to bear full weight on one foot for a prolonged period while the shoe is being nailed on the other. Avoid using shoes that increase laminar tension by transferring more weight-bearing forces to the hoof wall. A plastic-cuff heel-wedge shoe can be temporarily taped to the hoof with minimal trauma and effort. If it makes the horse more comfortable, the shoe can be glued in place. Frog support shoes continue to put pressure on the frog when the horse is recumbent and may predispose to subsolar necrosis. Frog support bandages or Lilly Pads‡ provide satisfactory support and avoid the complications associated with shoeing. The toe should be dubbed off to decrease the lever arm effect that a long toe has on prying the wall away from the distal phalanx during breakover.
Affected horses should be encouraged to lie down to reduce laminar tension. This goal can usually be accomplished with sedation. The stall should be heavily bedded with straw and pine chip shavings to a depth of 1 to 2 feet (30 to 60 cm) for comfort and to reduce the risk of pressure sores.
PROMOTING DIGITAL CIRCULATION
Walking with frog supports in soft ground for 5 to 10 minutes every 3 to 4 hours is beneficial for nonlame horses during the developmental stages of laminitis. It may increase the amount of laminar deformity when used in horses with lameness or in nonlame horses that have a depression at the coronary band indicating that laminar degeneration has already begun. Alpha-adrenergic blocking drugs such as acetylpromazine, phenoxybenzamine, and prazosin have also been advocated to decrease peripheral vasoconstriction and promote digital circulation. Acetylpromazine (0.02 to 0.04 mg/kg intramuscularly four times daily) may be given for its theoretic effect on digital circulation and for the sedative effect that encourages the horse to lie down and reduce laminar tension. Before lameness develops, heparin may be administered (40 to 100 units/kg subcutaneously two or three times daily) to provide laminitis prophylaxis by attenuating potential microthrombosis. This therapy significantly reduced the proportion of horses developing laminitis after proximal enteritis when given before onset of lameness.238
OTHER TREATMENTS
Antibiotics may be indicated in severe cases to reduce the risk of secondary sepsis in the foot. Methionine (20 to 60 mg/kg PO once daily) and biotin (0.03 to 0.2 mg/kg PO once daily) have been used for their effect on keratinization. Recently, a continuous (48-hour) ice-water bath has been advocated to chill the distal limbs of horses at risk for developing laminitis. Other methods of cooling the distal limbs include using bandages that continuously circulate ice water and simply adding crushed ice to a rectal sleeve and tying it to the pastern to chill the dorsal hoof wall, replacing it several times daily as needed. Rectal sleeves with ice are economical, well tolerated by horses, and simple to use, but are likely to be less effective than continuous ice-water baths or bandages.
Prognosis
Owners should be advised that it is often difficult to arrive at an accurate prognosis for up to 6 weeks after the original insult. Redden239 has suggested the following general guidelines regarding prognosis.
Horses that become sound within 24 to 48 hours of the onset of treatment, remain sound, demonstrate no radiographic changes, and have no palpable increased pulsation of the digital arteries after cessation of all medications for 5 days have a good prognosis. They should be given 10 additional days of stall rest, after which they can be vanned or put back to regular work.
Horses that develop 2 to 5 degrees of rotation or a laminar index measurement of 30% to 35% within the first 30 days of the onset but then become sound, remain sound, and show no further radiographic progression after an additional 45 days without treatment have a good prognosis. They may resume light exercise, but they should not be shipped long distances for several months, and they should be considered to have an increased risk of recurrence.
Horses that develop 5 to 10 degrees of rotation in the first 6 weeks but then have no further radiographic progression should receive an additional 90 days of stall rest. If they remain sound without medication, they may resume light exercise with caution after they have been turned to pasture for an additional 12 months. Such horses will not return to their previous level of performance and are not suited for racing or endurance, but they may function as pleasure horses.
Horses that develop 10 to 15 degrees of rotation within the first 4 to 6 weeks have a poor prognosis. The tip of the distal phalanx often penetrates the sole. Necrosis of the dermal and epidermal laminae and subsolar tissues usually occurs. Drainage often is noted at the coronary band or heels and is an indication of subcapsular abscessation. Gas or fluid pockets may develop between the hoof wall and the dorsal surface of the distal phalanx. Such cases require drainage and debridement of the necrotic tissue, which may be accomplished through an anterior hoof wall resection. If the keratinized sole is underrun, it is thinned enough to be elevated off the underrun areas so that necrotic debris can be curetted and the area flushed with antiseptic solution. Daily bandage changes and antiseptic flushing or soaking are required. If all necrotic subsolar tissue can be accessed without removing the keratinized sole, a thin layer of keratinized tissue should be left in place. Horses with a thin layer of keratinized sole are usually more comfortable than those in which the sole has been completely removed. Leaving a thin layer of keratinized tissue reduces the potential for exuberant granulation and seems to increase the rate of reepithelialization across granulating wounds in the sole.
Horses with this degree of laminitis require several months of stall rest and will be chronically crippled, at best. They will require several thousand dollars of care and bandaging just to stabilize the foot. The foot usually remains chronically painful, and, if so, euthanasia is justified on humane grounds. Tenotomy of the deep digital flexor tendon is beneficial in these cases. It seems to permit such severely affected horses to become more comfortable, enhances reepithelialization of defects in the sole, and permits the dorsal hoof wall to grow better.
Horses that have circumferential laminar necrosis in which the distal phalanx drops 2 cm or rotates 15 to 20 degrees with respect to the hoof capsule, or horses that develop a laminar index greater than 50% within the first 4 to 6 weeks of onset, carry a grave prognosis.
Prevention and Control
Prevention should be aimed at controlling as many risk factors as possible. Unrestricted grazing on lush spring grasses should be avoided, especially in areas where horses have developed laminitis in preceding years, and especially for horses with a history of laminitis. Horses should not be allowed to have unrestricted access to grain or concentrates, nor should they be fed a ration that primarily consists of concentrates. Factors that cause gastrointestinal upsets should be avoided; for example, changes in the ration should be made slowly, and overheated horses should not be allowed to engorge on cold water. Retained placenta in the mare should be treated within 3 hours after parturition.
Preventive measures should be instituted before clinical signs develop for horses that are at high risk of developing laminitis from conditions such as metritis, torsion of the colon, pleuropneumonia, proximal enteritis, or colitis. Preventive therapy should include frequent walking, frog support bandages and stabling on soft surfaces, cooling the distal limb, and administration of NSAIDs and antiendotoxin hyperimmune serum.
FLUOROSIS
Definition and Etiology
Ingestion of excessive fluoride by cattle, sheep, and horses can result in toxicosis. Acute fluoride toxicosis is relatively rare and is the result of accidental massive ingestion of fluoride compounds such as sodium fluoride or sodium fluorosilicate. Signs of acute fluoride toxicosis include restlessness, stiffness, anorexia, agalactia, salivation, vomiting or regurgitation, urinary incontinence, diarrhea, clonic convulsions, hyperemia, weakness, severe depression, and cardiac failure. Necrosis of the gastrointestinal mucosa and high concentrations of fluoride in plasma and urine are present in acute fluoride toxicosis. Chronic fluoride toxicosis is most often referred to as fluorosis, a general term that includes osteofluorosis and dental fluorosis. The most common sources of excess fluorides in the diet are (1) water with a naturally high fluoride content, (2) forages contaminated with fluorides from nearby (upwind) industrial plants (e.g., phosphate-processing plants, aluminum plants, smelters), (3) mineral (nondefluorinated rock phosphorus) and feed supplements with excessive fluoride content, (4) forages contaminated by soil or water (particularly sprinkler irrigation water) with a high fluoride content, and (5) volcanic activity, which can deposit fluoride-containing ash on soil, plants, or in water used for agriculture.
Clinical Signs, Differential Diagnosis, and Patho-physiology
Clinical signs of fluorosis are usually first recognized as either dental fluorosis or osteofluorosis. Developing teeth are very sensitive to the ingestion of excess fluorides. The deciduous teeth rarely show signs of dental fluorosis because a partial placental barrier to the accumulation of fluorides appears to be present in the fetus. During tooth development, excess fluorides cause ameloblasts to reduce in size prematurely and the enamel epithelium to form an irregular matrix. This matrix does not calcify normally, producing defects in the mature teeth. Cattle are susceptible to dental fluorosis during enamel matrix formation, from approximately 6 months to 3 years of age. Excess fluoride intake after 3 years of age does not result in the typical fluoride-induced dental lesions. Changes in incisor teeth are observed most frequently and include chalkiness, mottling (striations or patches in enamel), hypoplasia (defective enamel), and hypocalcification. Clinical lesions can be graded from normal to excessive.240 Factors that influence dental fluorosis include the amount of fluoride ingested, the animal’s age, the duration and consistency (intermittent vs. continuous) of exposure to fluoride, and the source and chemical form of fluoride ingested. Although diagnostically useful, dental lesions should not be used as the sole criterion to determine the degree of fluorosis.
Fluoride accumulation in bone occurs over a prolonged time; osteofluorosis can eventually develop if excessive fluoride is ingested. In cattle the first palpable lesions occur on the medial surface of the proximal third of the metatarsal bones. Later, lesions can be palpated on the mandible, metacarpal bones, and ribs. Radiographically, osteofluorotic bones are thickened with a chalky, roughened, and irregular periosteal surface.
The presence of osteoporosis, osteosclerosis, hyperostosis, osteophytosis, or osteomalacia depends on the amount of fluoride ingested and the duration of exposure to fluorides. The articular surfaces are not involved in osteofluorosis and can be used to differentiate osteofluorosis from osteomyelitis, osteoarthritis, and septic arthritis. The osseous lesions eventually cause intermittent lameness and stiffness, which may affect feed intake, body condition, milk production, and reproduction. Severe dental fluorosis causes reduced feed intake and efficiency, and affected animals are sometimes reluctant to drink cold water.
Clinical Pathology and Diagnosis
Diagnosis of fluorosis is difficult and complicated by many factors that affect fluoride intake and deposition. Fluorosis may be suspected by history, clinical signs, and physical examination. Radiographic findings of bone disease without evidence of joint involvement are highly suggestive of fluorosis in the live animal. The fluoride concentration in the urine of cattle may help to approximate recent fluoride exposure. The diagnostic value of urine fluoride analysis increases as the duration of excess fluoride ingestion increases. Normal cattle have a urine fluoride concentration of about 2 to 6 parts per million (ppm). Cattle exhibiting moderate fluorosis have urine fluoride concentrations of about 15 to 20 ppm. Cattle with urine fluoride concentrations of 40 ppm or greater or a urinary fluoride/creatinine ratio of 0.025:1 or greater could be suspected of ingesting a diet with a fluoride concentration of 60 ppm or greater.240,241 The concentration of fluoride in bone is quite helpful in the diagnosis of fluorosis (Table 38-3). Fluoride content in cancellous bone (e.g., rib, pelvis) is greater than in cortical bone. In addition, fluoride concentrations may vary in different areas of the same bone.
Fluoride concentration of bone is usually expressed as ppm (mg/kg) of dry, fat-free bone; however, some bone samples are ashed before fluoride determination. Therefore, it is critical to note precisely which bone was sampled, how it was prepared, and what part of the bone was analyzed. The metatarsus and metacarpus are typically analyzed for fluoride content. For all practical purposes, fluoride concentration is equal in either of these bones from the same patient.170 Using sawdust from a longitudinal section of bovine metatarsus (dividing the bone into lateral and medial halves or dorsal and palmar halves) yields virtually the same fluoride concentration as the whole bone.242 The fluoride concentration of the fourteenth coccygeal vertebrae (ash basis) is approximately twice that of the metacarpus (dry, fat-free weight basis).243 This is a practical tool for clinical diagnosis.
Analysis of dietary fluoride is a valuable adjunct to the diagnosis of fluorosis. The upper safe limit of fluoride in water for livestock is 2 mg/L (ppm).244,245 This safe limit may not protect against fluorosis in all field situations because of the large number of variables involved in the pathogenesis of fluorosis. Table 38-4 lists the long-term dietary tolerances for cattle. Under field conditions, the clinician must consider all possible sources of fluoride ingestion in evaluating total intake. In addition, because fluoride intake may be intermittent, a low dietary intake at one time may not necessarily eliminate a diagnosis of fluorosis.
Table 38-4 Long-Term Tolerances of Dietary Fluoride for Cattle
| Animal | Dietary Fluoride (ppm, Dry Basis) |
|---|---|
| Dairy or beef heifers | 30-40 |
| Mature dairy cattle | 40 |
| Mature beef cattle | 40-50 |
| Fattening cattle | 100 |
Treatment and Prognosis
No specific treatment is known for ruminants with severe fluorosis, and the prognosis is poor for cattle lame from extensive bony lesions. Animals removed from the offending diet or water may lose 50% of the fluoride from bone within 2 to 5 years;246 however, severe dental damage is irreversible.
Prevention and Control
Prevention involves avoiding feeds, water, and supplements with excessive fluoride concentrations. Feeding aluminum sulfate at 0.5% of the total diet reduces bone fluoride storage by 30% to 40%. However, additional phosphorus must be supplied in the diet, or osteoporosis and possibly spontaneous fractures may occur. Aluminum chloride or calcium aluminate also can be fed to cattle to reduce fluoride absorption. Calcium carbonate added to soils high in fluorides aids in reducing fluoride in forages. Cereal grains do not accumulate fluorides and thus can be helpful in reducing overall fluoride consumption. In circumstances of high fluoride concentrations in water, use of flood irrigation rather than sprinkler irrigation decreases the fluoride content of crops such as alfalfa hay.
HYPERTROPHIC OSTEOPATHY
Definition and Etiology
Hypertrophic osteopathy (HO), also known as Marie’s disease, is an uncommon condition in horses and is characterized by symmetric proliferation of connective tissue and subperiosteal bone along the diaphyses and metaphyses of the bones of the distal extremities.247-249 The skeletal manifestations of HO are usually secondary to a primary underlying disorder elsewhere in the horse’s body, often involving a space-occupying mass.247-250 Most often, intrathoracic disorders are the primary cause;248 intraabdominal and intracranial lesions have been less frequently associated with HO.248,250,251 In addition to horses, HO has also been described in humans, dogs, cats, cattle, deer, and fowl. In humans the term hypertrophic osteoarthropathy is used because the articular surfaces are usually affected; in animals, however, HO apparently does not frequently affect the articular surfaces.247-249
In the horse, HO has been associated with a number of primary intrathoracic disorders, including mycobacterial pneumonia,248,252 lung abscess,247,248 suppurative pneumonia,247,248,253 granulomatous pneumonia, systemic granulomatous disease,248,249 fibrosing pneumonia,254 primary and metastatic lung neoplasia,253,255-258 pulmonary infarction,259 rib fracture,260 pleural adhesion,248 and pericarditis.261 Reported primary extrathoracic disorders associated with HO include ovarian neoplasia,262-264 pituitary adenoma,251 and gastric squamous cell carcinoma.250 There is one report of HO in a mare that was thought to be associated with pregnancy; signs of HO developed during three different gestational periods and regressed each time after parturition.253 In occasional cases, HO may develop in the absence of an identifiable underlying disorder.248
Pathophysiology
The pathophysiology of HO is not completely understood. Initially, blood flow to the distal limbs is increased, followed by proliferation of connective tissue, then bony proliferation along the inner aspect of the periosteum.247-249 The periosteal proliferation results in the bony enlargements seen clinically. The link between the primary lesion and the skeletal abnormalities is poorly understood. Proposed explanations include hormonal abnormalities, hypoxia, arteriovenous shunting, and neurologic mechanisms.247-249 No single theory offers a completely satisfactory explanation.
Horses of any age or breed and either gender can be affected with HO. Some evidence suggests that HO may be more common in male horses and large-breed horses.248 Ponies, donkeys, cattle, and sheep can also be affected.253,256 The condition appears to be most common in mature horses.253
Clinical Signs
Common clinical signs of HO include lameness, stiff gait, and reluctance to move or trot.247-249 Firm bony enlargements are present on the distal extremities. In some cases, there may be soft tissue swelling or edema adjacent to the bony enlargements. In some horses the limb swelling is warm and painful to palpation, whereas in others the swelling is cold and painless.248 Pain can frequently be elicited by forced flexion of the major joints. Bony enlargements are usually bilateral and symmetric and often affect the cranial, lateral, and medial aspects of the affected bones.247,248 All four limbs are usually affected.248 The metacarpal and metatarsal bones are most often affected; other sites that can be affected include the phalanges, carpus, tarsus, radius, and tibia, as well as the maxilla, mandible, and nasal bones.248
Affected horses may or may not show signs related to the primary underlying disorder, such as cough, fever, weight loss, ventral edema, tachypnea, or colic.248 Clinicopathologic features are highly variable and depend on the underlying primary disease process. Generally, there is increased serum activity of alkaline phosphatase, associated with increased osteoblastic activity at the sites of periosteal proliferation.248,249
Radiographically, the bony enlargements are characterized by periosteal proliferative, new bone formation of the diaphysis and metaphysis of affected bones.248 The periosteal reaction often exhibits an irregular, palisade pattern of osteophyte formation.247,248 The bony reaction may extend to the chondrosynovial junctions; the articular surfaces are usually not affected.249 In a few cases, nuclear scintigraphy revealed focal areas of intense uptake of radiopharmaceutical at the sites of bony enlargement.247,261
Treatment and Prognosis
The prognosis for horses with HO is guarded and depends on the underlying disorder. In one study, 71% of horses with HO were euthanized.248 Gradually progressive limb swelling and pain is the typical clinical course in most affected horses for which an underlying cause is either not identified or effectively managed. Thus, management of HO should be directed at identification and treatment of the underlying primary disorder, if possible. Common methods to aid in identification of the underlying disease include hematology, serum chemistries, fibrinogen, thoracic radiography, thoracic and abdominal ultrasonography, abdominocentesis, thoracocentesis, gastroscopy, and rectal examination. Successful management of the primary underlying disorder has resulted in partial or complete regression of the skeletal disease in a limited number of equine cases.247,248,262 Regression of HO is characterized by a decrease in limb swelling and lameness, and a return to athletic performance is possible if bony lesions are not advanced.247 In a few idiopathic cases in which an underlying disorder was not identified, bony reactions decreased with rest and phenylbutazone therapy.248
FESCUE FOOT
Definition and Etiology
Fescue foot is a toxicosis of cattle grazing pastures that contain tall fescue grass (Festuca arundinaceae Schreb). The condition is characterized by lameness, particularly of the rear limbs, progressing to dry gangrene of the feet and lower legs. The end of the tail and the ear tips occasionally may be affected.
Fescue pastures also cause three other syndromes. “Summer slump” is characterized by reduced gain and milk production, dull hair coat, and poor heat tolerance. Abdominal fat necrosis also has been reported in cattle on fescue pasture.265 Equine reproductive problems, including prolonged gestation and agalactia, as a result of grazing tall fescue late in gestations have also been described.266
The earliest description of “fescue foot” appeared in 1949 in Australia. It has been reported in Europe, New Zealand, and the United States, particularly in the Southeast. The etiologic agents in fescue have not been definitely established, although several toxins have been identified.265,267-271 Chemicals produced by the grass itself (e.g., loline, perloline, several organic acids) could be toxic agents contributing to fescue foot and other syndromes, but mycotoxins produced by endophytic fungi, which infect tall fescue, are generally accepted as being the most important agents. The endophytic fungus of tall fescue, Acremonium coenophialum (formerly Epichloe typhina), produces ergovaline, ergonine, ergosine, and lysergamide.265,267,271 All these toxins are capable of producing vasoconstriction similar to that caused by ingestion of the ergot fungus Claviceps purpurea. In fact, the symptoms of fescue foot are identical to gangrenous ergotism. Pastures not infected with Acremonium do not produce any of the symptoms of fescue toxicosis in animals.271
Environmental factors also play a role in the development of fescue foot. Although the disease can occur over a range of seasons, symptoms usually occur during colder months. Another factor is the level of pasture fertilization. High levels of nitrogen in soil, regardless of the form applied, increase pasture toxicity. Certain strains of fescue, particularly Kj-31, seem to be more toxic.272 Finally, because the endophyte imparts a selective advantage on the infected plant by increasing growth rate and disease resistance, the number of toxic plants in a pasture increases. The infestation rate of the fescue endophyte is high; more than half the forage samples from states in which fescue foot occurs are infected with A. coenophialum.273
Clinical Signs and Differential Diagnosis
Clinical signs of fescue foot usually begin as hindlimb lameness. Affected cattle also are underweight and have a dull, “rough” hair coat. The feet and pasterns become cold to palpation, and the coronary bands become reddened and swollen. Hair may be rubbed from the pastern area with the fingers, and limb edema may be present. As the condition progresses, the classic signs of fescue foot appear, including a sharp line of demarcation at the level of the pastern or fetlock, distal to which the skin becomes dry and gangrenous and eventually sloughs. The tips of the ears and tail also may necrose. Affected animals lose condition initially and eventually are unable to stand or to walk. Susceptibility to the toxin in animals on a given pasture seems to vary considerably. Generally, morbidity is low, although 20% to 30% of the herd may be affected in some circumstances.274
The diagnosis is made on the basis of characteristic signs and gross lesions, as well as the presence of tall fescue in the pasture. As mentioned, gangrenous ergotism is identical clinically, except that it occurs in the presence of the easily recognizable “ergot” fungus C. purpurea, which grows on rye grass. Similarly, chronic selenium toxicosis (“alkali disease”) mimics fescue foot, except that this disease does not occur on fescue pastures, and affected animals have elevated tissue selenium concentrations. Both ergot and selenium poisoning affect animals other than cattle, whereas fescue foot has been described only in the bovine.
Early in the course of the disease, mechanical foot injury, foot rot, or laminitis can resemble fescue foot, especially because only a few animals in a herd are affected. Close inspection of the foot, however, reveals lesions that are typical of these diseases. Necrosis as a result of freezing may be difficult to distinguish from fescue toxicosis because both occur at the same time of year.
Pathophysiology
Pastures that contain tall fescue infected with A. coenophialum produce toxins responsible for vasoconstriction. Peripheral vasoconstriction causes blood stasis, endothelial damage, and thrombosis in peripheral vessels. As a result of impaired circulation, tissues of the distal extremities become ischemic and gangrenous. By decreasing peripheral circulation, cold ambient temperatures exacerbate the condition.
Necropsy Findings
At necropsy the principal finding is a characteristic line of demarcation between normal and gangrenous tissues. Generalized loss of condition also is found because of the animal’s inability to ambulate and eat. Vascular thrombosis and necrosis of the tissues of the lower limbs are found microscopically.
Treatment and Prognosis
When animals with fescue foot are recognized early in the disease course, they should be removed from pasture as soon as possible. Antibiotic treatment to prevent bacterial invasion of injured skin and hooves is valuable; recovery can occur in 2 weeks. Once the extremities have necrosed, however, treatment is unsuccessful and euthanasia is recommended.
Control
Unlike C. purpurea, which grows only on the seed head of grasses, the toxins causing fescue foot are contained in the leaves and stems. As a result, mowing contaminated pastures is not an effective control measure. Growing strains of tall fescue with low toxicity, mixed with legume forage plants, seems to be the best management technique for controlling fescue foot. In cold weather, feeding hay to cattle when the pasture contains tall fescue decreases the ingestion of toxins.
INTERDIGITAL NECROBACILLOSIS (FOOT ROT) IN CATTLE
Definition and Etiology
Interdigital necrobacillosis is an infectious disease of cattle that is a leading cause of lameness in feedlots and confinement dairies. The condition can affect cattle at any age, although most cases are in mature or weaned animals. Sporadic cases are encountered with pastured beef and dairy animals. A variety of names have been used to describe this condition, including “foul-in-the-foot,” foot rot, and interdigital phlegmon (phlegmona interdigitalis). The condition is characterized by inflammation and tissue necrosis of the soft tissues of the interdigital space.275 Deep structures (e.g., tendons, ligaments, synovium, bone) may be involved in severe cases. Fusobacterium necrophorum, a gram-negative anaerobic rod, has classically been considered the etiologic agent associated with foot rot; however, multiple anaerobic organisms are likely to be synergistically involved. Typical foot rot lesions could be induced 5 days after inoculation of the interdigital cleft with F. necrophorum alone.276 Prevotella melaninogenicus and more recently Porphyromonas levii (both previously classified in the genus Bacteroides) have been reported to play an important role.275 The presence of Dichelobacter nodosus, the agent associated with contagious foot rot in small ruminants and interdigital dermatitis in cattle, may facilitate penetration of the skin with F. necrophorum. Wet, unsanitary conditions and rough environmental flooring are important factors in precipitating infection.
Clinical Signs
Early in the course of foot rot, symmetric swelling and heat of the interdigital space are noted, progressing to the coronary band and possibly extending proximally to the level of the fetlock. The claws begin to spread as a result of severe cellulitis, leaving a widened interdigital space. Soft tissue swelling leads to necrosis and fissure formation within a few days,277 starting at the dorsal interdigital space and spreading toward the heels. The edematous skin margins protrude and roll outward (Fig. 38-33). Exudation and pseudomembrane formation are seen, but significant purulent drainage is not evident. A characteristic foul odor accompanies the necrotic lesion. Acute and progressive lameness in one limb is typical, with the hindlimbs more frequently involved than the forelimbs.278,279 Septic synovitis, osteomyelitis, or tendon involvement is more likely to result in non-weight-bearing lameness and limb carriage. Mild to moderate elevation of the body temperature (39.4° C to 40° C [103° F to 104° F]) may be recorded. The associated pain leads to reduced ambulation, feed intake, and milk production as well as weight gain. Affected animals may spend a significant amount of time lying down, predisposing them to injury from herdmates. Systemic effects of inflammation and infection can result in reduced fertility in breeding bulls. Differential diagnoses for lameness of the foot include trauma, interdigital dermatitis, verrucose dermatitis, and laminitis.280

Fig. 38-33 Foot rot in a mature Angus cow. The interdigital skin is cracked and swollen.
Photo courtesy of Dr. Kevin Washburn, Texas A&M University, College of Veterinary Medicine & Biomedical Sciences, Large Animal Clinical Sciences.
A severe form of foot rot described more recently is peracute in onset and refractory to conventional treatments.278,281 Severe interdigital swelling is noted in multiple limbs and most often in the hindlimbs. The condition is rapidly progressive, leading to recumbency and a rapid extension to deeper structures. Euthanasia may be warranted on animal welfare grounds.282 Penicillin/sulfonamide-resistant F. necrophorum has been isolated from several cases.281 Concurrent infection with bovine viral diarrhea (BVD) virus and the resultant immunosuppression have been suggested as an etiology.283
Clinical Pathology
Laboratory analysis of blood is not typically done, although a normal or an inflammatory leukogram may be encountered. Collection of samples for microbial culture is seldom done because of the ability to diagnose the condition through typical clinical findings. Lesional swabs or biopsy samples may be submitted for bacterial culture. A mixed population of environmental and fecal contaminants is likely; however, isolation of F. necrophorum and other anaerobes suggests true foot rot. Microscopic evaluation of biopsy samples may identify spirochetes, although this is not a consistent finding.
Pathogenesis
The mechanisms of interdigital necrobacillosis are complex and incompletely understood. Compromise of the skin barrier is critical for invasion of the offending pathogens. Chronic exposure of feet to wet and dirty conditions leads to softening and maceration of the skin. Exposure to sharp gravel and stones, excessive stubble, irregular concrete, and other forms of mechanical trauma can result in significant abrasions and damage to the interdigital skin, particularly in wet environments. The combination of these two factors can result in penetration of the offending pathogens and resultant infection. Initial microbial flora may reduce tissue oxygen tension, allowing anaerobes an ideal environment to proliferate.280,284 Once the organisms colonize the subcutaneous tissues, multiple mechanisms likely promote growth and evasion of the host defense systems.
The two subspecies of F. necrophorum currently recognized are biotypes A and B. Biotype A appears to be more virulent and is more frequently isolated.285,286 Others have described differentiation of virulent strains by colony morphology,287 and PCR differentiation of strains is available.288 Although F. necrophorum is also a human pathogen, differences apparently exist between human and animal strains.285 F. necrophorum is a normal inhabitant of the gastrointestinal tract of ruminants, although isolation from feces is generally rare.285,289 Oral administration of some antimicrobials has been shown to increase fecal shedding of F. necrophorum.290 The increased number of organisms in exudate from necrotic lesions may provide an important source of transmission to other animals. Major virulence factors associated with this organism are a high—molecular-weight leukotoxin and lipopolysaccharide (LPS).291,292 The leukotoxin is cytotoxic to ruminant neutrophils and, when combined with LPS, may protect the organism from phagocytosis.285
Recent findings suggest that P. levii did not stimulate a significant chemotactic response in bovine macrophages in vitro, and suppression of phagocytosis occurred when the organism was present in low numbers.275 These properties of P. levii may facilitate a local tissue environment that allows other anaerobic organisms, including F. necrophorum, to colonize and evade host defense mechanisms.
Epidemiology
Interdigital necrobacillosis has been recognized worldwide for centuries.293 Wet and humid climates predispose to disease, and foot rot has been associated with rainy seasons.284,294 The risk of disease is higher in management systems that expose animals to traumatizing ground underfoot, such as concrete confinement with water and excrement accumulation, irrigated pastures, muddy lots, high stocking densities, and stony walkways. As noted, cattle of any age are susceptible, but mature animals account for most cases. Bos indicus breeds appear to have a lower incidence of foot rot than Bos taurus, and Jersey cattle may be overrepresented in the dairy breeds.294
Necropsy Findings
Postmortem examination is rarely performed. Characterization of the lesions reveals subcutaneous and soft tissue necrosis, with suppurative inflammation of the foot. In severe cases, evidence of osteomyelitis, tenosynovitis, and septic arthritis may be identified.
Treatment and Prevention
Most animals are responsive to parenteral antimicrobial therapy. Local treatment (e.g., topical preparations, wound care, bandaging) is probably not necessary for minor cases identified early, although local therapy may reduce the number of infectious organisms spread to the environment.284 The organisms are typically susceptible to a variety of antimicrobials.278 Procaine G penicillin (22,000 to 44,000 IU/kg intramuscularly [IM] once or twice daily) has been used successfully when infection is detected early.284 These dosages of penicillin are considered extralabel, and the clinician must take appropriate steps to ensure adequate meat- and milk-withdrawal periods. Currently, a number of drugs have been approved for the treatment of foot rot in the United States.278,282 Amoxicillin (6.6 to 11 mg/kg IM or subcutaneously [SC] for 5 days), oxytetracyline (10 mg/kg IM or SC), sulfadimethoxine (55 mg/kg PO or IV loading, then 27.5 mg/kg PO or IV daily for 5 days), erythromycin (2.2 to 4.4 mg/kg IM daily for 3 to 5 days), ceftiofur (1.1 to 2.2 mg/kg SC daily for 3 to 5 days), tylosin (18 mg/kg IM daily for up to 5 days), sulfamethazine (30 g/100 kg PO, repeated in 72 hours), and florfenicol (40 mg/kg SC once).278,280,282,284
Early recognition and treatment can produce clinical improvement in 2 to 4 days.278 The antimicrobial of choice can be tailored to suit the needs of the farm in terms of withdrawal times, number of treatments, route of administration, and familiarity with the product. Investigators found that 5 mg/kg of extralabel tilmicosin SC at the onset of lameness resulted in a 74% cure rate, significantly higher than with placebo.295 Antiinflammatory therapy, including flunixin meglumine or aspirin, may reduce fever and provide analgesia, resulting in improved appetite and ambulation.282
When topical therapy is warranted, cleaning the wound and curettage of the necrotic tissue are beneficial. A variety of topical antimicrobial or astringent preparations can be placed under a foot bandage. The main objectives of bandaging are to keep the wound clean and prevent further contamination from fecal organisms for several days. Moving treated animals to clean, dry areas is recommended.280 Dressing the foot to prevent further spreading of the digits helps to protect the interdigital space. Foot bandaging may also reduce the amount of exudate from infected feet, which could infect herdmates.284
Spontaneous recovery is possible, although the risk of complications is increased, and recovery may take several weeks.280 Animals with significant deep structure involvement may require prolonged therapy and repeated wound management. Animals with evidence of septic arthritis of the distal joints or other synovial structures may require amputation of the digit to allow drainage. Claw amputation has been described.282,296
Metaphylaxis, or the mass treatment of animals, may be warranted in some circumstances. Methods to mass-treat animals involve topical treatment in the form of footbaths or the addition of feed-grade antimicrobials and supplements to diets. It is prudent to inform owners that no feed additives are currently approved for the prevention or treatment of foot rot.297 Extralabel use of feed additives, as governed by the Animal Medicinal Drug Use Clarification Act, is strictly prohibited.278,297 The only antimicrobials approved for nonenteric disease as food additives are chlortetracycline and oxytetracycline.297 Ethylenediamine dihydriodide (EDDI) has been used to reduce the severity and incidence of foot rot,298 although high doses are toxic in cattle. EDDI use for this purpose is prohibited, based on the present regulation of feed additives.278,297
Footbaths are an appealing alternative for many producers, but proper use is important. Contact time and efficacious ingredients are sometimes difficult to ensure. Footbath additives often are rapidly contaminated with organic material, eventually leading to inactivation.297 Antibacterial agents and astringents have been traditionally used, including 3% formalin, 2% copper sulfate, oxytetracycline, and lincomycin-spectinomycin formulations. Dry footbaths have also been reported and include 10% copper sulfate and slaked lime.280 Practitioners must check state and local laws, which may prevent the use of some substances that could cause environmental contamination and might be unsafe for handlers.
Vaccination against F. necrophorum has been available for some time, but firm evidence of its efficacy in peer-reviewed publications is limited.278 One study evaluating the incidence and severity of hepatic abscesses and foot rot suggested a positive response to vaccination of feedlot cattle compared with controls, although the significance was evident only in the free-choice forage program. The same positive result was not evident when the analysis included all cattle, particularly the limit-fed grain program. The benefit of vaccination may not be able to overcome the challenge of diets that promote rumenitis.299
INFECTIOUS FOOT ROT IN SHEEP AND GOATS
Definition and Etiology
Infectious foot rot is a highly contagious foot disease of sheep and goats and is the leading cause of infectious lameness in these species.300,301 The essential pathogen is the anaerobic bacterium Dichelobacter nodosus.301-310 Synergistic activity with other microbes, primarily Fusobacterium necrophorum, plays an important role in the pathogenesis.300,303,304 Equally important are the roles that environmental moisture and climate contribute to the transmission from animal to animal.300,303-305,309 The infection has been reported in a variety of species, including cattle, horses, pigs, deer, and mouflon, but is generally considered a sheep and goat—specific disease.307
Epidemiology
Foot rot accounts for significant economic losses in sheep-producing countries302,304,306,307 and has often resulted in government control programs.301,305 Reduced body and fleece weight and increased costs of management, treatment, and culling make control of this disease important. Infectious foot rot is found worldwide wherever sheep and goats are reared, although it is primarily a problem in warm and humid climates.300,303-305,309 Sheep raised on improved pastures in arid regions may also develop the disease.304 Prolonged periods of moisture are required to facilitate transmission; short periods of heavy rainfall do not lead to significant increases in clinical disease.300,303,304 Consistent temperatures greater than 10° C (50° F) are considered favorable for transmission.300 Incidence rates are likely to correspond with the seasons that favor these conditions and may vary between geographic regions. Using pastures with ground cover that promotes trauma to the foot is likely to increase the risk of disease. The natural herding behavior of sheep and goats is likely to propagate the spread of the organism within a flock. Traditionally, routine hoof trimming was thought to reduce the incidence by improving general foot health; however, others have not made this association and have actually proposed trimming as a risk factor.300,301,304,309
Sheep are the primary species of concern, although other ruminants can be infected. An association with increased severity in lambs from aged ewes has been suggested.304 The merino breed appears to be the most susceptible, whereas British breeds demonstrate less severity.303,304 Clinical disease is more often diagnosed in adults, although lambs may also be affected. Age and breed have been identified as risk factors, with an increase in incidence with age. Breed genetics and heritability may play important roles in resistance to disease, and reducing matings from chronically infected sheep may be an important step in propagating less susceptible breeding lines.301,304 Introduction of the organism to a naive flock is almost exclusively by the addition of asymptomatic, chronic carrier animals, which may harbor the organism on their feet for years.303,304 Animals exposed to pastures or housing facilities immediately contaminated by infected feet are at risk of contracting the organism. D. nodosus is an obligate parasite of hooves and survives only a short time in the environment, but it may remain viable for up to 2 weeks in favorable conditions.303,304
Pathogenesis and Clinical Signs
The interdigital skin is typically an adequate barrier to infection. In prolonged wet conditions, however, the skin becomes macerated and weakened. Initially, F. necrophorum invades the skin and superficial soft tissues, resulting in ovine interdigital dermatitis (OID). This organism may produce a local tissue environment that promotes invasion of D. nodosus. These two organisms work synergistically to allow F. necrophorum to invade into deeper tissues and allow bacterial proteases produced by D. nodosus eventually to cause horn separation. Both organisms are essential for true foot rot to occur.300 The lesion associated with foot rot is characterized as exudative inflammation and necrosis of the epidermal tissues of the foot.307 A characteristic foul odor is almost always present.303,304 Both claws of multiple limbs are typically involved. The interdigital skin becomes moist and swollen, and the soft horn is pale, pitted, and may be separated from the skin.303
Disease associated with D. nodosus infection is highly dependent on the virulence of the organism, resistance of the animal, and environmental conditions. All these factors are extremely variable; relatively low virulent strains in wet conditions may contribute to more severe disease than a highly virulent strain in dry weather. Virulence is associated with the keratinolytic ability of a strain and occurrence of type IV fimbriae.300,302,304-307 The fimbrial subunit gene, fimA, appears to be essential for virulence.304 Heat-stable protease production is characteristic of virulent strains.
Infectious foot rot is categorized into two clinical syndromes, benign or virulent, based on clinical severity and the ability to transmit through the flock. Some have included a third, intermediate syndrome for cases that clinically are more involved than the benign form but are not associated with the appreciable production losses that occur with the virulent form.300 Foot rot accounted for a 10% decrease in body weight and wool production in one report.307 It may be difficult to define clearly each case early in the disease process; what appears to be benign may be the early onset of the more virulent form. Benign foot rot may progress to separation of the soft horn from the underlying hard horn; however, it does not progress beyond this point. Virulent foot rot is characterized by severe interdigital lesions with separation of the soft and hard horn from the sensitive laminae. The separation begins at the axial surfaces of the heels and progresses in a dorsolateral direction.303,304 Underrunning of the sole occurs, and in the most severe cases, the entire hoof may be sloughed. When confronting acute, multiple-limb lameness in several animals in a flock, a practitioner should strongly consider virulent foot rot.
The spread from animal to animal is generally rapid, and multiple stages of the infection are present within the flock. Inspection of the flock is indicated to categorize the severity of infection. In some flocks, only a small portion needs to be examined to determine the classification of disease, whereas evaluation of the majority of the flock is required in other cases. If uncertainty exists, 2 weeks is usually adequate to allow lesions to progress to a stage that will accurately allow classification of the clinical syndrome.300 Affected animals may graze on their knees to relieve the foot pain or may remain recumbent in severe cases. Systemic signs of infection may include fever, anorexia, and weight loss.303 Complications of the foot wounds include blowfly strike and secondary bacterial infection.303,304 Although susceptible, the clinical course in goats is often less severe. Interdigital dermatitis is a more prominent clinical finding than separation of the horn in this species.303
Differential diagnoses for foot rot include traumatic injury, laminitis, foot abscess, OID, and contagious ovine digital dermatitis (CODD). The presence of interdigital dermatitis increases the risk of the development of foot rot, and the prevalence may be high in the early stages of foot rot infection. It may be impossible to distinguish clinically between interdigital dermatitis and benign foot rot.300
Identified in the United Kingdom in the mid-1990s, CODD was initially thought to be a severe form of virulent foot rot. Clinically, the disease shows similarity to infectious foot rot, although initial involvement of the interdigital space is usually absent with CODD. Ulcerative lesions involving haired skin adjacent to the coronet are more common. This condition responds poorly to traditional foot rot treatment strategies. The complete pathogenesis of CODD is unclear, but one report isolated D. nodosus in 74% and a spirochete in 70% of affected feet. The spirochete was from the genus Treponema, which has been associated with similar lesions in cattle.310
Clinical Pathology, Diagnostic Tests, and Necropsy Findings
Samples collected by swabs or lesion material can be submitted for anaerobic microbial culture. The mixed bacterial population present and the fastidious growth of D. nodosus make isolation difficult and time-consuming.302,307 Collection of samples in anaerobic transport containers and rapid submission to the laboratory are required. One report found that D. nodosus could not be isolated from field samples if they remained longer than 3 hours in transport.302 Routine growth media found in most laboratories is usually not sufficient to grow this organism, and selective media and techniques are helpful in isolating this microbe.302 Microscopic examination of D. nodosus organisms by Gram stain reveals large, barbell-shaped, gram-negative rods.303,307 Some investigators have reported colony morphology and correlation with virulence, but others have been unable to make this correlation.302
Biochemical testing for elastase activity and thermal stability of bacterial proteases has been used to differentiate benign from virulent strains. Thermostability of bacterial proteases can be measured by the gelatin gel test,304,305,307 although false-positive results have been reported.305 Advanced molecular techniques, including PCR and DNA sequencing, have recently become available for detecting gene sequences that encode for virulence, particularly fimA.302,305,306 These advanced techniques have expanded the classification and number of strains and serotypes identified302,305,306 and allow a rapid and sensitive diagnostic tool for use in control programs.304 Serum antibody detection using an ELISA is available, but its specificity may be lacking.304 Serology may be helpful in determining flock involvement. Infectious foot rot is usually diagnosed on clinical findings, and postmortem examination is not typically necessary.
Treatment and Prevention
Once D. nodosus infection is identified in a flock, treatment to control and potentially eradicate the organism should be initiated. Eradication may not be possible in all flocks, and the severity of disease, management practices, strain virulence, and environmental factors undoubtedly play a critical role. Separation of animals confirmed to have lesions is likely to reduce the transmission to uninfected flockmates; however, this may not be possible under some management strategies. When possible, planning pasture rotation to allow 2-week dormancy periods enables producers to have infection-free zones. Improving pastures to prevent areas of standing water, excrement, and mud will theoretically reduce the potential for transmission. Routine flock foot trimming has been a mainstay in the prevention of foot rot, although supportive clinical data are lacking.300,301 Regions having consistent problems with foot rot have discontinued this practice because of the lack of evidence and the increase in time and labor.301 However, judicious foot trimming for the treatment of foot rot is probably beneficial on an individual basis and when topical treatments are employed.301,303,304 Meticulous paring of the hoof may be necessary if the disease has produced overgrown and misshapen feet that promote hiding places for D. nodosus. Disinfection of knives and shears between each animal is important to reduce iatrogenic transmission. Treatment is typically more successful during the dry periods of the year, when transmission is reduced.
Medical treatment methods for foot rot include topical therapy, including footbaths, topical medications, and parenteral antimicrobials. Although bandaging individual feet may increase the contact time of medications and reduce the spread of infectious material, this is not practical and does not allow the foot to dry. Topical 5% oxytetracycline tincture has been used successfully.304 Chemicals used in footbath solutions include 5% copper sulfate, 3% to 5% formalin, and 10% zinc sulfate.300,303,304 When used properly, these agents are usually highly effective, but caution must be used with each chemical to prevent adverse effects. All these agents are potentially toxic to varying degrees if ingested, and copper sulfate will stain the fleece. Comparison of formalin and zinc sulfate in footbath preparations yields similar results.304 Formalin is more resistant to contamination by organic material, but higher concentration and frequent application are extremely irritating to the skin. Additionally, considerable human health concerns and government regulations may prevent the use of formalin in some flocks. Efficacy and improved safety make zinc sulfate an ideal agent for use in footbaths. Improved chemical penetration of the hoof can be achieved when the surfactant sodium lauryl sulfate is combined with the footbath solution, reducing the need for foot trimming before treatment. Use of footbaths requires facilities and construction (6 to 12 m [20 to 40 ft] long) that ensure all feet on each animal are treated. The ideal contact time has not been established in clinical studies, but brief walk-through or soaking for up to 1 hour has been shown to be successful.300 The frequency of treatment is also not clearly defined and may be dictated by the contact time. Because of its irritating properties, formalin should not be used more than once a week.304 Although the response is likely multifactorial, cure rates of 50% to 80% have been reported with foot bathing. Separate footbaths for infected and noninfected animals reduce the risk of inadvertent transmission. Washing the feet by walking animals through a plain-water bath and allowing the feet to dry 2 hours before and at least 1 hour after treatment is ideal.300 Once treated, the groups should be immediately moved to clean, dry pastures or lots that have not held livestock for at least 2 weeks.300,303,304
Currently, no antimicrobial drugs in the United States are labeled for use against infectious foot rot in sheep, so use is extralabel. One-time treatment without foot trimming can produce similar results to foot bathing. Parenteral antimicrobials have produced recovery rates of 85% after one dose when sheep were kept on dry floors for 24 hours after treatment.300 Although D. nodosus has been reported to be sensitive to many antimicrobials in vitro, this gives no indication of sensitivity when used clinically.304 Medications used successfully have included procaine penicillin G (20,000 to 30,000 IU/kg IM twice daily),303 long-acting oxytetracycline (20 mg/kg SC once, or repeated in 72 hours),303,304 erythromycin (10 mg/kg once, or 3 to 5 mg/kg IM twice daily),303,304 florfenicol (20 mg/kg IM, repeated in 48 hours),303 and lincomycin (5 mg/kg SC once) in combination with spectinomycin (10 mg/kg SC once).304 Maintaining dry footing for 24 hours, yielding meat- and milk-withdrawal times, and the potential of reinfection are important considerations when using parenteral antimicrobials.300
Vaccination against D. nodosus is available, and its use in the treatment and control of foot rot has been reported.300,301,303,304,308 A reduction in disease severity and quicker response to treatment may be evident when this is used as a treatment modality. Several strategies have been used for classifying D. nodosus strains, based on fimbrial types, and at least 10 serogroups with 19 serotypes have been identified.300,302-304,306,307 Fimbrial gene variation undoubtedly plays an important role in vaccine efficacy.304 The current commercial vaccine available contains 10 serotypes, and antigenic competition is likely to impede additional serotypes.300,304 Identifying the most prominent strain or fimbrial type in a geographic region or flock may facilitate vaccine selection and development. Eight strains of D. nodosus have been reported to infect one foot.304 Response to vaccination appears to be strain specific, with poor cross-protection between strains. Successful resolution of clinical signs by the use of an autogenous vaccine, without other treatments, has been reported.308 The use of adjuvants in killed vaccines are required to produce an adequate antibody response, and the addition of adjuvants, particularly oil based, has led to a high incidence of injection site reactions.300,303,304 The response to vaccination is likely short-lived, about 8 to 12 weeks,300 and requires multiple doses. Vaccination of animals during the dry period, several weeks before the optimal transmission period, is likely beneficial in a control program.
Control and eradication can potentially be achieved in regions where the climate allows a break in the transmission period; however, the investment in labor and the cost of medical therapy may be high. A combination of topical treatment, parenteral antimicrobials, and vaccination is likely to be employed. Careful observation for early signs of infection and quarantine of any new animals are critical to controlling or preventing a flock outbreak. Using a weekly zinc sulfate footbath for 2 to 4 weeks, along with vaccination and culling of severe and unresponsive cases, has been suggested.303 Based on evidence of resistance heritability, careful breeding programs should be explored.301,304 Complete eradication from a farm can be achieved if the entire flock is removed, the facilities are left dormant for at least 2 weeks, and the flock is repopulated with noninfected animals.300
OTHER INFECTIOUS CONDITIONS OF THE FOOT
Infections involving the foot of horses and cattle are common and almost always caused by contamination from the environment gaining access to the soft tissue or bony structures of the foot within the hoof capsule. Typically the animal has an acute, progressive lameness such that the animal is almost non—weight bearing on the affected limb or will bear weight on the toe region of the foot but resist putting the heel on the ground. Other clinical signs include increased digital pulse pressure, heat and sensitivity to hoof tester pressure over the affected area. Regional nerve blocks can be used to localize the lameness to the foot but frequently are not necessary. Because the foot is encased in a shell of horny tissue, exudate from an infectious process often accumulates between the soft tissue and horn. The exudate then spreads or dissects along that plane until it reaches a soft tissue surface such as the coronary band, especially near the bulbs of the heels, where rupture and drainage can occur.
PROBLEMS ASSOCIATED WITH HORSESHOE NAILS (“NAIL PRICK”)
History and Clinical Signs
Nail prick (“quicking”) refers to penetration of the sensitive hoof structures, usually the sensitive laminae, by a driven horseshoe nail. The horse will usually react as the farrier drives or clinches the nail by jerking the foot from the farrier. In some cases, blood may appear on the nail or leak from the nail hole. It is important to realize that nail pricks occur for many reasons and are not always caused by a misdirected nail. Poorly made shoes, nails that are too large, poorly placed nail holes, and faulty nails can also result in a nail prick. Horses with poor hoof quality, thin hoof walls, or flaring hoof walls can be difficult to nail and thus are at greatest risk. Fractious horses and young horses not previously shod may lean on the farrier or repeatedly pull the foot from the farrier, making nail driving difficult. Damage from an improperly driven nail can vary from minimal to serious infection.
Diagnosis
Diagnosis of a misdirected nail warrants great diplomacy from the veterinarian because many owners become unjustifiably upset with the farrier. Some horses will repeatedly stomp the affected foot or paw the ground immediately after shoeing. Others may point the affected limb after shoeing. Lameness may not be apparent immediately after shoeing and may occur days later when the nail hole becomes infected and the trapped pus begins to exert pressure. The horse usually becomes acutely lame and worsens over time unless treated. Some infected nail holes will travel up the lamellae (white line) and create an abscess or soft spot at the hairline of the coronary band. The abscess will be directly aligned with the hoof wall tubules, leading to the infected nail hole, which is an important diagnostic aid. Hoof tester examination, paying particular attention to pressure over the driven nails, is an essential diagnostic tool to locate the offending nail. Using a hammer to strike the outside hoof wall may also elicit a painful response over the nail. Increased digital pulse and heat may be present.
Treatment
Pricks from nails can be potentially serious and require immediate treatment. If the nail prick is discovered by the farrier at the time of shoeing, the nail is removed. The nail should be examined for moisture or blood. The nail hole can be irrigated with DMSO, povidone-iodine (Betadine) solution, or hydrogen peroxide. The nail hole is packed with iodine-soaked cotton and the nail hole left open. Often the nail is redirected, and no further treatment is needed. If the horse’s immunization status is not current, a tetanus vaccine is given.
If the offending nail cannot be localized or the nail hole is infected, the shoe is removed. Hoof testers are then used to localize the painful nail hole. Frequently the pressure from the hoof testers will cause black, malodorous liquid “pus” to exit from the hole. The basis of treatment is to establish drainage. The infected nail hole often requires enlargement with a loop hoof knife or curette. Ideally a cone-shaped hole is made, with the larger opening at the bottom of the hoof. The hole is irrigated or the entire foot soaked in an Epsom salt—Betadine footbath for 20 to 30 minutes twice daily until the infection is gone. It is important to protect the foot from the environment (e.g., mud, dirt) by keeping the foot bandaged between foot soaks. Additional medications are usually not necessary unless involvement is extensive. Antiinflammatory medications may be beneficial to decrease pain. Once the infection has cleared, the shoe is replaced. The affected nail hole can be packed with iodine-soaked cotton and the horse reshod with a plastic pad covering the sole. Alternately, a hole can be drilled into the shoe over the affected nail hole and the shoe replaced, leaving access to the infected area for daily irrigation and povidone-iodine packing.
SUBSOLAR ABSCESS
Subsolar abscess is one of the most common causes of lameness in the horse and cow, but it is relatively rare in sheep and goats. In one study in cattle, 88% of cases of lameness were caused by problems in the hoof.311 Most often, lameness is caused by a hole or crack in the horny sole, which then becomes packed with dirt, and eventually contamination extends to and is trapped within the sensitive soft tissue beneath the sole. Animals that are housed in filthy or muddy conditions and that have inadequate hoof care or overgrown feet are more prone to this problem. Horses taken from a very dry to a very wet environment, and vice versa, also tend to have a higher incidence of sole abscesses, apparently because of the sudden change in the moisture content of the hoof and the resultant cracking of the sole. Cattle with “corkscrew claw” (a hereditary condition) or other growth deformity in the claw are more prone to sole abscess. Puncture wounds or any form of trauma that introduces contamination to the soft tissue of the sole also may result in abscessation.
History and Clinical Signs
Subsolar abscess is a common cause of acute lameness seen in all horses. There is no signalment or breed disposition. “Gravel” is a lay term describing the assumed etiology in which a piece of gravel migrates proximally from the white line to coronary band, leaving an infected tract. Subsolar abscesses may originate from a penetrating wound in the white line, a nail hole, or a deep subsolar bruise. Lameness is usually acute and severe (grade 3 to 4 of 5) and may worsen over time until drainage is established. The horse will often point and may not bear full weight on the affected limb. Distal limb swelling often accompanies a subsolar abscess that has not had drainage established. Systemic signs of infection (fever, lethargy) usually are not associated with subsolar abscesses unless deeper structures are involved. The infected tract may migrate and open at the coronary band. Before breaking open, a soft, painful area can be located by digital palpation of the coronary band.
Diagnosis
Digital pulses are usually increased, and the hoof capsule may radiate heat. A focal painful area can usually (but not always) be located with careful hoof tester examination. Careful paring of the sole and frog may be helpful in locating the abscess, but the clinician must be careful not to damage good, healthy tissue looking for the infection site. Foot poultices and hot-water footbaths with Epsom salts will help eventually to localize the affected area. Gray or black malodorous liquid will leak from the infected tract (Fig. 38-34). Radiographs are useful to rule out other causes, such as third phalanx and navicular bone fracture. In some cases a gas or fluid pocket can be identified on radiographs.
Treatment
Treatment is aimed at establishing adequate drainage. If an opening in the tract is present at the solar surface, it should be enlarged just enough for good irrigation and drainage. This may or may not require sedation or peripheral anesthesia of the foot. If pink tissue or blood is encountered, debridement is discontinued. Large holes should be avoided to prevent solar corium protrusion, which can be a painful sequela to overzealous hoof paring. If drainage occurs at the level of the coronary band and solar surface, through-and-through lavage is beneficial. Debridement at the coronary band level should be minimal to prevent iatrogenic coffin joint contamination. Once drainage is established, the foot is protected from the environment and from recontamination with a foot bandage. Continued foot soaks in warm-water povidone-iodine and Epsom salt footbaths should be continued until sepsis and inflammation are eliminated. The shoe is replaced when the affected are is dry and cornified. Large areas may require a plastic pad under the shoe for solar protection. Antibiotics and NSAIDs are rarely needed, except in severe cases or if penetration of deeper structures has occurred. The patient’s tetanus immunization should be checked and a booster injection administered if needed.
DEEP PENETRATING INJURIES TO THE SOLE
History and Clinical Signs
Most animals’ environment is filled with sharp objects that can penetrate the sole, causing severe damage to structures deep within the hoof capsule. All puncture wounds should be considered potentially serious, but those involving the solar white line or caudal frog area require special attention because of potential navicular bursa, digital tendon sheath, deep digital flexor (DDF) tendon, or distal phalanx involvement.
The clinical signs of a penetrating wound to the sole vary with the anatomic structure involved and chronicity of the injury. The animal may show minimal lameness initially at the time of injury, but severe lameness once sepsis is established days later. Animals with penetrating wounds to the podotrochlear bursa or DDF tendon attachment to the third phalanx are in considerable pain and are reluctant to bear weight on the heel region.
Diagnosis
If owners find a foreign body in the bottom of the animal’s foot, they usually should be instructed to leave the object in the foot unless there is danger of further penetration. A radiograph is taken immediately to determine depth of penetration. Digital pulses are increased, and the foot is usually warm to touch. Hoof testers are useful to determine the focal point of pain, but often the entire surface of the foot is reactive. Systemic sedation and peripheral anesthesia using an abaxial sesamoid nerve block may be needed to examine the penetrated foot thoroughly. Light paring of the sole and frog areas with a hoof knife may reveal a black spot indicating the penetration site. Often, however, the entry is not discovered because of the elastic nature of the hoof structures and collapsing of the penetration site. Regional intravenous anesthesia (Bier block) using a tourniquet is the method of choice for producing local anesthesia in cattle. Paring should be performed carefully so that the solar corium is not injured with the hoof knife. Because the corium represents the horn-producing epithelial covering of the foot, injuries to it heal the same way as skin injuries. If an entry wound is discovered, the foot is scrubbed and prepared for further diagnostic testing. A sterile, flexible probe can be gently inserted into the hole and a radiograph taken to determine the depth and direction of the hole. The clinician must be careful so that inadvertent force or horse movement does not cause the probe to penetrate previously unaffected structures. Alternately, a less invasive and preferred method is to catheterize or place a sterile teat cannula into the hole and inject sterile radiopaque material to determine which structures are involved (Fig. 38-35).
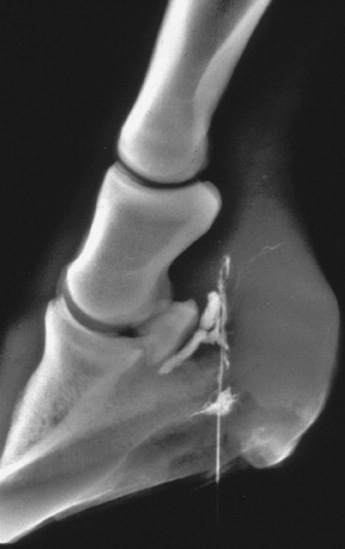
Fig. 38-35 Radiograph of a foot after injection of contrast dye to determine the path of the penetrating foreign body.
If sepsis of the podotrochlear bursa, distal interphalangeal joint, or digital tendon sheath is suspected, paracentesis and joint fluid cytology and culture and microbial sensitivity should be performed. Synovial fluid with an abnormal color, viscosity, elevated number of degenerative neutrophils, and high total protein concentration should suggest sepsis. If distal limb swelling is present, arthrocentesis should be delayed until the cellulitis subsides.
A series of radiographs should be taken to include a lateromedial, 60-degree dorsopalmar view of both the navicular bone and the third phalanx, and flexor (tangential) view of the navicular bone. Repeat examination 1 to 2 weeks after the original injury may be necessary because bony lysis takes time to develop and may not be apparent at the initial radiographic examination.
Treatment
Broad-spectrum systemic antibiotics, antiinflammatory medications, and tetanus prophylaxis are administered if penetration of deep hoof structures is suspected. Depending on the severity of the lameness, the animal may require an NSAID such as phenylbutazone (4 mg/kg twice daily in horses; 10 mg/kg once daily in cattle) to reduce pain and lameness in the initial stages. Antibiotic therapy should be continued for 2 weeks after resolution of clinical signs of infection in most cases. Preferably, antibiotics are initiated after a culture and microbial sample has been obtained. Establishment of drainage, copious lavage with sterile ionic fluid, and debridement of all necrotic tissue are indicated. Involvement of the podotrochlear bursa indicates use of navicular bursoscopy and aggressive medical therapy, consisting of daily lavaging of the bursa under general anesthesia and local treatment using antibiotics instilled within the bursa and regional limb perfusion. Surgical treatment such as the “street nail” procedure is reserved for animals not responding to aggressive medical therapy.
If the DDF tendon is involved (septic tendinitis), debridement and removal of frayed and infected tendon fibers may be performed in the standing, sedated horse using a tourniquet and peripheral anesthesia, or under general anesthesia, depending on horse temperament and owner financial constraints. After debridement, placing the horse in a 4- to 8-degree wedge shoe decreases weight-bearing forces on the DDF tendon and provides some pain relief. The wedge shoe angle is gradually decreased over several months as the DDF tendon begins to heal and strengthen. The bottom of the foot requires protection with a bandage or hospital plate until the surgical site granulates in and cornifies.
Deep penetrating wounds to the sole, especially the solar—white line junction, can result in infectious osteitis of the distal phalanx. The horse usually presents with a chronic, recurrent draining tract at the coronary band or solar surface and varying degrees of lameness. Infection of the distal phalanx often results from undetected soft tissue infection or dissecting subsolar abscesses. Septic pedal osteitis may also be a sequela to laminitis secondary to recurrent abscessation and ischemia at the toe region. As the bone infection progresses, blood supply to the area is compromised, and the area of avascular bone separates from the parent bone, forming a sequestrum. Localized third phalanx septic osteitis and sequestrum formation may not become radiographically evident for weeks after the initial penetration injury. Radiology often reveals a radiolucent area within the margins of the third phalanx, with or without sequestrum formation.
Debridement and curettage of all soft and necrotic bone can usually be performed in the standing, sedated animal if its temperament permits. The foot is desensitized with peripheral anesthesia, cleaned with a hoof knife and steel brush, then prepared for aseptic surgery using standard techniques. Hemostasis is achieved by wrapping a roll of Vetrap firmly around the fetlock joint to compress and occlude the palmar digital arteries. Access to the infected bone is made by removing sequential layers of sole using either a motorized Dremel tool or a Galt trephine with a retractable pilot bit.312 The infected bone is usually discolored and soft and should be curetted to healthy bone margins. The infected bone should be cultured and undergo microbial sensitivity testing. A postoperative radiograph should be taken to ensure complete debridement. After surgery the site is packed with sterile gauze sponges soaked in antiseptic or antimicrobial solutions, and then the foot is bandaged. Disposable diapers and duct tape are inexpensive materials for a waterproof foot bandage. The bandage is changed every 1 or 2 days for the initial few weeks. Application of a bar shoe and hospital plate provides solar protection and decreases the labor and cost of daily bandage changes (Fig. 38-36). The bolts and metal plate are removed so that the surgical site can be cleaned and treated, and then the hospital plate is bolted back in place.
After surgery the horse is confined to a small area until the hole granulates in and cornifies, usually 4 to 6 weeks. If granulation tissue becomes excessive at the surgical site, application of 2% tincture of iodine will hasten healing. If severe infection is present, the site can be packed lightly with antibiotic-impregnated beads for continued antibiotic release at the infected site. Regional limb perfusion with antibiotics is also beneficial in severe cases.
In cattle, simple sole abscesses may heal quite satisfactorily without bandaging. In cows, use of a wooden block on the unaffected claw of the affected foot elevates the affected claw to a non—weight-bearing level and greatly reduces lameness. The block is attached with an epoxy such as Technovit and allowed to wear off over a period of weeks.
Prognosis
Prognosis depends on severity of injury, structures involved, and chronicity of the problem before initiating treatment. Horses with acute penetration wounds that receive immediate and aggressive treatment have a good chance of returning to athletic use. Penetration injuries that have an established infection involving the podotrochlear bursa, digital tendon sheath, or distal interphalangeal joint have a poor prognosis. In one study, only 12 of 38 horses with sepsis of the navicular bursa had a satisfactory outcome.313 Chances of survival increased if treatment was started within a week of the penetration injury. Osteomyelitis of the navicular bone and DDF tendon rupture are common complications of a septic navicular bursa.
Prognosis for infectious osteitis of the distal phalanx is good if sepsis is not caused by laminitis. In one study, up to 24% of the distal phalanx was removed, and the horse returned to athletic soundness.314
THRUSH
Definition and Etiology
Thrush is a bacterial infection characterized by an accumulation of black, malodorous, necrotic material usually originating within the central or collateral sulci of the frogs of the hoof. This degenerative condition may spread to involve deeper structures of the foot, such as the digital cushion, hoof wall, and heel bulb region of the foot, causing inflammation and breakdown of these structures.315 Many keratolytic organisms may be present, but Fusobacterium necrophorum is often isolated. Thrush is most often caused by poor environmental conditions; animals standing in soiled stalls, deep mud, swampy land, or wet pastures are at risk for developing thrush, especially if the feet are not cleaned daily. Another predisposing factor is poor hoof conformation. Saddlebreds, Tennessee Walkers, and other gaited horses have long feet with naturally deep frog sulci and are at risk of thrush.316 Horses with sheared heels or acquired frog deformity are also predisposed. Horses shod with full pads may develop thrush secondary to moisture and dirt collection under the pad. Other, well-kept, clean horses can develop thrush for no apparent reason.
Clinical Signs and Diagnosis
Lameness may or may not be present, and the severity can vary. Diagnosis is based on the presence of black, malodorous discharge, most often within the frog sulci. The central frog sulcus is often malformed and very deep. A painful response may occur when the affected sulci are cleaned because the degenerative process may extend to sensitive structures of the foot. If structural damage has occurred, the heels may move independently of each other, causing pain on manipulation. Horses with mechanical instability of the heels may show caudal heel pain.
Treatment
The basis for treatment is removing the predisposing cause of the thrush. The horse should be moved to a clean, dry environment and have the feet cleaned daily. Any necrotic debris and undermined tissue are carefully debrided and cleaned using a hoof knife. Foot bandages may be necessary if the debridement is extensive. Systemic antibiotics may be necessary in severe cases that involve deep or more proximal tissues. Infection is usually managed by topical medication. Several caustic materials have been recommended, including a combination of phenol, tincture of iodine and 10% formalin, Kopertox solution, or methylene blue. Others have recommended soaking the foot in chlorine bleach (1 oz bleach per gallon water). If poor hoof conformation is the cause, proper farrier management should be pursued to correct existing problems. If heel instability is present, a bar shoe may be necessary to stabilize the caudal aspect of the foot. Exercise is important to strengthen the caudal aspect of the foot and will naturally clean the feet. The best treatment for thrush involves prevention by educating the client on proper hoof hygiene.
WHITE LINE DISEASE (“SEEDY TOE”)
History and Clinical Signs
The white line, visible at the sole, is created by the junction of the insensitive laminae of the hoof wall and the horn of the sole. White line disease has historically described the separation of the hoof wall from its laminar attachments. A crack or opening occurs within the white line, allowing a bacterial or fungal infection to invade the stratum medium close to the laminae, causing cavities to develop between the laminae and outer hoof wall. In environmental conditions with too much moisture (continuous wet pastures) or in drought conditions producing excessively dry feet, animals are at risk of developing a crack or opening in the white line. Horses with poor-quality hoof walls that split or crack may develop white line disease. In addition, horses with chronic laminitis having a thickened or stretched white line in the toe region (“seedy toe”) are predisposed (Fig. 38-37).
The hoof wall separation is usually a chronic condition beginning weeks or months before presentation. Horses with white line disease often show little or no lameness and therefore are not perceived as an immediate concern by many owners. Hard ground may exacerbate any lameness seen.
Diagnosis
Visual examination of the white line, assisted by a probing instrument, will reveal a cavity with separation of outer hoof wall from the laminae. Radiographic evaluation will determine the full extent of hoof wall separation. Often the cavity is either dry or filled with necrotic debris, which may involve a bacterial or fungal infection. The cavity is usually not painful to probing.
Treatment
Treatment begins with removal of the separated outer hoof wall using a hoof nippers, hoof knife, and motorized tools. It is important to remove any unattached or loose hoof wall to reveal cracks or crevices that could harbor bacteria. The Dremel tool burr is useful to smooth any cracks in the insensitive laminae that are exposed after hoof wall removal. After thorough debridement, large defects usually exist in the hoof wall and require protection (Fig. 38-38). A heart-bar shoe redistributes weight-bearing forces to the frog and caudal region of the foot and away from damaged and weakened areas. The hoof wall defects prevent normal nailing procedures, and shoe clips can help secure the shoe to the hoof. After hoof wall removal, the exposed laminae may still have an “active” infectious component. With the heart-bar shoe in place, the owner is instructed to keep the horse in a clean, dry stall and topically treat the exposed laminae with iodine or methiolate daily for 10 days or until the exposed laminae are dry and “inactive.” At this point, the horse may be a candidate for prosthetic hoof wall repair using a product such as Equilox. The plastic acrylic is trimmed and shaped to the horse’s natural hoof wall at the next shoeing. The owner should keep the hoof dry to avoid losing the acrylic patch. The horse may return to normal activity once the prosthetic patch is in place.
QUITTOR
Quittor is a term used to describe chronic infection of the medial or lateral collateral cartilage of the distal phalanx in the horse. This condition is characterized by local inflammation and necrosis of the affected cartilage, with subsequent formation of draining tracts proximal to the coronary band. The infection is usually caused by a wound, and the horse often presents with moderate to severe lameness and a history of chronic or recurrent drainage. Obtaining a dorsopalmar radiographic view after injecting contrast dye into the draining tract helps to demonstrate the infected collateral cartilage (Fig. 38-39).
Although this condition occasionally responds to medical therapy, the treatment of choice is surgical debridement with the horse under general anesthesia, the foot in full extension, and a tourniquet around the pastern. An elliptical section that includes the draining tract should be incised above the coronary band and discarded. All underlying infected soft tissue and necrotic cartilage should be excised. If the case has not become chronic and only a small amount of cartilage needs to be removed, the wound may be packed and partially sutured. The pack can be removed in 24 to 48 hours and the wound flushed twice a day until it has healed. If the condition is chronic with extensive cartilage necrosis or multiple draining tracts, the infectious process usually extends below the level of the coronary band. In this case it is best to trephine a hole in the hoof wall at the lowest point of the infection. This procedure provides better drainage, facilitates debridement of the necrotic cartilage, and allows for complete closure of the surgical incision above the coronary band. The wound should be packed through the trephine hole and treated as an open wound until it heals. In cases with extensive cartilage necrosis, care should be taken to avoid accidental opening of the coffin joint. If debridement is complete, the prognosis for the return to soundness is fair to good.317
FISTULOUS WITHERS
Definition
Fistulous withers is a chronic inflammatory condition of the supraspinous bursa and associated tissues in horses and mules. The supraspinous bursa is located dorsal to the dorsal spinous processes of the second through fifth thoracic vertebrae (T2 to T5). The normal bursa may extend laterally to the scapular cartilage and may be asymmetric. Normal bursal volumes range from 30 to 90 mL.318
Clinical Signs
Affected horses may show nonspecific signs, including lethargy, fever, and generalized muscle stiffness. These may progress to the more specific signs of pain, heat, and swelling of the withers and cervical area. Over days to weeks, these swellings mature and rupture at one or more locations, releasing serous or purulent drainage. The classically described discharge is yellowish brown and mucoid or serous with white, yellow, or red flakes (“rice bodies”).319 Fistulous tracts may extend to the dorsal spinous processes of the vertebrae, as well as to the scapula, ribs, and occasionally the thoracic cavity. Lesions and drainage may wax and wane.318
Etiology and Pathophysiology
There are two forms of fistulous withers: idiopathic and traumatic. The more common idiopathic form results from the spread of inflammation and infection of the supraspinous bursa to surrounding structures, including the nuchal ligament. Organisms reported to be the primary cause of fistulous withers include Brucella abortus, Actinomyces bovis, and Onchocerca cervicalis. B. abortus has a predilection for synovial structures and is often associated with septic supraspinous bursitis. In 1937, B. abortus was isolated from 80% of fistulous withers cases; 92% of affected horses had contact with cattle, and 65% were from farms with Brucella-positive cattle.319 Currently, the disease has some geographic variability. In New York, where the prevalence of B. abortus infection in cattle is low, B. abortus is rarely isolated from equine cases of fistulous withers. In 1988 the prevalence of B. abortus in horses presented to the New York State College of Veterinary Medicine had apparently decreased, with only 2 of 14 horses (14%) having positive titers.320 In Texas, however, where seropositive cattle are still prevalent, 9 of 28 horses (37.5%) with fistulous withers were seropositive for B. abortus, and these horses were more likely to be pastured with seropositive cattle.321 Researchers have also experimentally produced fistulous withers by injecting B. abortus and A. bovis into the supraspinous bursa.322O. cervicalis has also been proposed as an etiology for fistulous withers, especially in Australia. This parasite migrates through the nuchal ligament causing inflammation, which may extend to the supraspinous bursa. Fibrous and calcified tracts develop around dead and viable parasites, causing necrosis and degeneration of the nuchal ligament.323
The traumatic form of fistulous withers is preceded by an open wound or blunt trauma to the withers and then secondary infection. Infection may result from external contamination or from hematogenous spread to devitalized tissue. Secondary contamination of open wounds with Streptococcus equi subsp. zooepidemicus320 or environmental microorganisms is reported.
Diagnosis
A careful history must be obtained, including duration and severity of clinical signs and response to previous treatment. Physical examination with careful palpation over the withers may reveal pain, swelling, or draining tracts. Complete blood count may show leukocytosis caused by neutrophilia, as well as hyperfibrinogenemia, indicating chronic inflammation.
If the swelling is encapsulated and not draining, needle aspiration should be performed to obtain a sample for culture and sensitivity. Once tracts are open and draining, culture is of questionable validity because of secondary bacterial contamination. Negative cultures can also be misleading because B. abortus is difficult to grown in culture and may be overgrown by other bacteria. Biopsy of inflamed tissue may also be obtained for culture and histology. Blood cultures may also be diagnostic in febrile animals.
All animals with fistulous withers should have serology for Brucella performed because of the public health implications. Plate agglutination is the test of choice and is more sensitive and specific than the card test. Brucella titers are considered to be negative when they are less than 1:50. Ideally, paired titers should be performed, with any rise in titers 2 weeks apart indicative of current infection.324 Chronic infections may not show an increase, however, so a single high titer with compatible clinical signs should be considered diagnostic.
Radiographs should also be obtained in suspected cases of fistulous withers. They may show osteomyelitis and periostitis of the dorsal spinous processes of T1 to T9. There may also be swelling or granular opacity of the soft tissue in the withers region.320 Horses with positive B. abortus titers are more likely to have radiographic evidence of osteomyelitis of the dorsal spinous processes than seronegative horses.321 A positive-contrast fistulogram may help determine the extent of any draining tracts. Ultrasonography may also help delineate draining tracts.
Treatment
Medical treatment may be attempted but is often unsuccessful alone. Antimicrobial selection should be guided by culture and sensitivity results. In the absence of a culture, systemic broad-spectrum antimicrobials may be used. This can also be combined with lavage of the draining tracts. Systemic antiinflammatories are also indicated to control pain.
Brucella vaccination with the S19 strain has been reported in Brucella-confirmed cases as an effective extralabel treatment. The vaccination regimen varied from single-dose subcutaneous (SC) injection, to a series of three SC injections 10 days apart, to intravenous (IV) injection. In 1937, Duff319 reported that 95 of 134 horses that received two or three SC injections recovered after vaccination; 35 of these horses were also treated surgically. SC injection reportedly causes severe local and systemic reaction, including fever and abscessation.324 The IV route may also be used, but it resulted in death in three of four horses; the other horse fully recovered in 4 weeks.321 At this time, there are no approved vaccinations for use in horses, and the RB51 vaccine has not been used in equids. Also, the apparent decline in frequency of Brucella in fistulous withers would make vaccination of questionable value.
Medical management usually needs to be combined with aggressive surgical debridement for effective treatment. Conservative treatment alone may lead to recovery periods of 4 or more months. Reportedly, 80% of horses with traumatic fistulous withers treated conservatively will have recurrent lesions.325 Drainage and debridement with the horse standing has been performed, but general anesthesia with aggressive debridement is the preferred treatment.320 Ostectomy and curettage of the dorsal spinous processes may be necessary. Primary closure is difficult to achieve but will provide a more cosmetic result and quicker healing time. Wounds left open to heal by second intention may take 2 to 4 months to heal.320
Prevention and Control
Brucella abortus infection, if present, carries great public health implications. Horse owners and veterinary staff must be notified of the possible exposure and take appropriate precautions. Brucellosis in humans may cause acute fever with pneumonia, spondylitis, orchitis, or pyelonephritis. Chronic brucellosis causes a transient fever in people.318
Parasite control is an important aspect of prevention of fistulous withers. Horses should also be kept separate from Brucella-positive cattle. Proper saddle and harness fit will also decrease the incidence of traumatic fistulous withers.
FLEXURAL LIMB DEFORMITIES
Definition
Flexural limb deformities are defined as hyperflexion or hyperextention of a limb. Flexural deformities are described by the joint involved, typically the distal interphalangeal (DIP) joint, metacarpophalangeal (MCP) or metatarsophalangeal (MTP) joint, carpus, and rarely the tarsus. Although commonly referred to as “contracted tendons,” the tendons themselves are not truly contracted, but rather short relative to the adjacent bone. Flexural limb deformities can be classified as either congenital or acquired. Contractural deformities are very common in horses and cattle. Small ruminants and camelids are much less frequently affected.
Pathogenesis
Congenital flexural deformities (deformed at birth) are likely caused by a multitude of factors. Although intrauterine positioning has been suggested, it is probably not a common cause of flexural limb deformities. Exposure of the mare during pregnancy to diseases and teratogenic agents is a more likely cause of congenital deformities. Evidence for a definitive inciting cause is lacking, but the following have been implicated in contributing to flexural limb deformities: ingestion of locoweed (Astragalus) and hybrid Sudan grass, equine goiter, defective cross-linking of collagen, defects in elastin formation, neuromuscular disorders, and viruses.326-329 It is common for foals with congenital flexural deformities to have other anatomic abnormalities, such as spinal deformities, which may result in dystocia.
Acquired flexural deformities are seen typically from 4 weeks to 4 months of age, as well as in yearlings to 2-year-olds. These deformities have been implicated as part of the developmental orthopedic disease complex. Two general theories explain how acquired flexural deformities occur. The first theory is that the tendon unit is unable to elongate passively relative to the bone.330 In the case of the DIP joint, the rigid accessory ligament of the deep digital flexor (DDF) tendon causes a palmar rotation of the foot (club-footed) (Fig. 38-40). Flexural deformities in the MCP joint are usually caused by the accessory ligament of the superficial digital flexor (SDF) tendon. The second theory is that increased tension within the tendon induces pain, leading to the deformity.331 Acquired deformities are often noticed with an acute onset, which may add validity to the latter theory.
Diagnosis
CONGENITAL DEFORMITIES
Digital hyperextension deformities occur in neonates because of a lack of tension in the flexor muscles. Typically these deformities correct without intervention after a few weeks. In severe cases the palmar or plantar surfaces of the foal’s pastern and fetlock area may become ulcerated as a result of contact with the ground, if not protected.
Contractural deformities may involve one or a combination of the following: SDF tendon, DDF tendon, and suspensory ligament. Congenital flexural deformities are most often seen affecting the carpus, tarsus, MCP joint, and MTP joint332 (Fig. 38-41). Contracture of the DIP joint also occurs and presents with the foal walking on the toes. Depending on the severity of the deformity, if the MCP joint is affected, the foal may or may not be able to stand. Although not truly a contractural deformity, rupture of the common digital extensor (CDE) tendon results in a similar appearance. These foals have a characteristic swelling on the dorsolateral aspect of the carpus. Often these foals bow at the carpus because of the lack of support over the lateral aspect of the carpus.326,333 More importantly, foals with ruptured CDE tendons should be checked for incomplete ossification of cuboidal bones because these conditions often occur together.330
ACQUIRED DEFORMITIES
Contractural deformities are typically described by the joint affected. The most common acquired flexural deformity involves the DIP joint. The affected limb will have a steeper hoof wall angle with a shorter toe than the unaffected limb. The heel will elongate on the affected limb, and the MCP joint will concurrently begin to hyperextend. Two stages have been used to describe contractural deformities of the DIP joint: Stage I is defined as an angle of less than 90 degrees between the dorsal hoof wall and the sole; stage II is a dorsal hoof wall that exceeds the vertical angle.334
Early detection of contracture of the MCP joint is characterized by a straight fetlock angle, or upright conformation. More severe contracture of this joint is evident by buckling at the fetlock. By placing pressure in a dorsopalmar direction on the fetlock, the flexor tendons are stretched, allowing the practitioner to determine which structure is tightest, which is important for selecting an appropriate treatment.330
Carpal contractures tend to occur within the first 6 months of life. These foals tend to be fast-growing, heavier foals (Fig. 38-42).
Although not as common, contractural deformities of the proximal interphalangeal (PIP) joint occur and are usually bilateral.330 The exact mechanism is unknown, but it is thought that as the musculotendinous unit of the DDF complex shortens, the SDF tendon becomes more lax.335 This can lead to a dorsal subluxation of the joint. Often a clicking sound is appreciated when the foal walks. This contractural deformity is most often seen in weanlings with a high growth rate.330
Flexural deformities also occur after foals or horses are non—weight bearing for a time. These deformities can be permanent if the joint undergoes “contracture” or fibrodesis (fixed without osseous ankylosis).330
Treatment
CONGENITAL DEFORMITIES
Hyperextension deformities require exercise to hasten recovery. Excessive exercise is contraindicated because this leads to fatigue and possible injury. Treatments include swimming, corrective shoeing, and light bandaging when indicated. Swimming allows muscle tone to increase without bearing weight on the limbs. If the foal’s toe lifts off the ground when weight bearing, glue-on shoes with a heel extension may be indicated. Glue-on shoes must not be left on a growing foal for an extended period because they may prevent the foot from expanding normally. Bandaging is only indicated when the palmar/plantar surface of the pastern or fetlock is in contact with the ground, to prevent ulcerations of the skin. Excessive bandaging and support will only cause more laxity and should be avoided.
Treatment for congenital contractural deformities should include pain management by judicious use of NSAIDs. The use of gastroprotectants is strongly suggested to help avoid the ulcerogenic effects of NSAIDs.
Oxytetracycline at a dose of 44 mg/kg diluted in physiologic saline given slowly IV has been used for many years in foals with contractural deformities.336 Renal function of the foal should be considered before administration of oxytetracycline. Many mechanisms have been proposed to explain how oxytetracycline achieves a decrease in the angle of the MCP joint. One mechanism is the chelation of calcium resulting in muscle contraction.336 Oxytetracycline has also been shown to be a neuromuscular blocking agent, although this mechanism does not explain why other muscle groups are unaffected.336 An in vitro study showed that oxytetracycline also works through matrix metalloproteinase-1 mRNA expression to inhibit tractional organization of collagen fibrils by myofibroblasts; this interaction may cause relaxation of the musculotendinous unit.337 Regardless of the mechanism of action, foals treated with oxytetracycline should also undergo forced exercise to make the improvement permanent. Without exercise, the angle of the MCP joint tends to return to its previous value within 96 hours.338
Toe extensions benefit the foal in two ways: (1) helping to protect the toe from being overworn and (2) lengthening the breakover of the foot, adding strain to the flexor tendons, potentially helping stretch them. Toe extensions may be made using acrylic hoof products or glue-on shoes with built-in extensions. Shoes and extensions should be removed within 2 weeks because they can prevent the hoof from growing and expanding naturally in a young foal. Typically, 2 weeks is sufficient time to achieve correction. For foals with contracture of the DIP joint, frequent rasping of the heels will help stretch the DDF tendon.
Splints and casts have been used to treat flexural deformities by giving support to the limb, thereby allowing the muscle-tendon unit to relax. When using splints, it is vital to use plenty of padding on the limb and to change the padding every time the splint is changed, to ensure pressure sores are not developing. Pressure sores can be very serious and even life threatening. Splints are made from a variety of materials but most often, polyvinyl chloride (PVC) pipe cut to an appropriate length and width; commercially made splints are also available. Splints are applied by placing the splint over the padding on the palmar or plantar surface of the limb. If the contractural deformity involves the DIP joint, the splint should touch the ground. If the deformity is in the carpal region, the splint should end at the fetlock. Splints should be left in place for a period deemed adequate for the individual patient, ranging from several hours to 2 or 3 days.330,332 One major advantage over casts is that splints may be removed every few hours if desired. Casts used for the lower limb are typically half-limb casts, incorporating the foot. The exception is the carpal region, where a tube cast is more appropriate, ending at the fetlock. Casts should never be left in place longer than 2 weeks at a time because of the rapid growth rate of foals. Cast sores are a serious complication and a legitimate disadvantage to using casts.
A combination of conservative treatments should be considered when treating foals with contractural deformities. If nonsurgical management does not correct the problem, surgical intervention may be necessary; however, foals with severe congenital contractural deformities have a poor prognosis. Surgery is more often attempted for foals with carpal contracture. Other congenital contractural deformities usually respond to nonsurgical treatments. Surgical correction for contractural deformities in the carpal region involves transection of the tendons of the flexor carpi ulnaris and ulnaris lateralis, as well as transection of the palmar carpal fascia.330,332 The prognosis for mild to moderate congenital flexural deformities is good for an athletic future if the foal improves over the first 2 weeks and is able to stand and nurse.332 Contractural deformities that can be manually straightened have a much better prognosis than those that cannot be straightened.
Foals with ruptured CDE tendons are best treated nonsurgically. Affected foals should be confined to stall rest with the application of a splint.333 As with any splint, it should be well padded to protect the limb. The splint will provide support to the limb and help protect immature cuboidal bones, which often accompany this condition.330
ACQUIRED DEFORMITIES
Conservative management for acquired contractural deformities is similar to that for congenital deformities. Other conservative therapies include dietary restrictions, including early weaning. Surgical management is selected for animals unresponsive to nonsurgical treatments.
For contractural deformities of the DIP joint, desmotomy of the accessory ligament of the DDF tendon is indicated. This surgery has an excellent prognosis for full athletic potential for stage I horses. For more severe flexural deformities of this joint (stage II), a tenotomy of the DDF tendon may be necessary.339 This procedure is not performed to promote an athletic career. It is acceptable for pasture soundness, breeding animals, and for some, light riding.
Acquired flexural deformities of the MCP joint are treated nonsurgically with a combination of NSAIDs, corrective shoeing (raising the heel with or without using a vertical bar shoe with rubber dorsal tubing) and splints. Physical therapy has been used to help stretch the tendons by having the horse hop on one leg by holding the other up in a flexed position, moving in a lateral direction toward the down limb.330 If surgical correction is needed, it is important first to determine which structure is creating the tension. Most often the SDF tendon is involved, although the DDF tendon and even the suspensory ligament may be involved. For treatment of contracture of the SDF tendon, desmotomy of its accessory ligament is performed. If the DDF tendon is the most taut, desmotomy of its accessory ligament is completed. Both accessory ligaments may be cut when indicated. In severe cases the suspensory branches may be transected, but this procedure is done for salvage because it usually leads to subluxation of the PIP joint.330
Carpal contractures are treated with splints and controlled exercise. Early weaning should be considered in these foals. If surgery is necessary, a tenotomy of the flexor carpi ulnaris and ulnaris lateralis in addition to splints may yield good results.
Contracture of the PIP joint is not as common but can be a serious threat to the future athletic potential of the horse. Over time the soft tissues around the joint will fibrose, and the PIP joint may undergo degenerative joint disease. At this stage, a pastern arthrodesis may be necessary to achieve a pain-free and potentially sound limb. Desmotomy of the accessory ligament of the DDF tendon may benefit affected horses early in the disease process.330,335
TENDINITIS
Definition and Etiology
Tendon injuries can have a profound influence on the future athletic careers of horses. The superficial digital flexor (SDF) tendon is the most frequently injured tendinous structure in the performance horse.340 It occurs more frequently in the thoroughbred and standardbred racehorse, although it is typically seen in all types of performance horses. Deep digital flexor (DDF) tendon injuries in the metacarpal/metatarsal region occur infrequently340-342 and are identified in older horses (9 to 10 years)341-344 and in horses that jump.343 Western performance horses more often have hindlimb deep flexor tendinitis (or tendonitis), probably because of their sudden turns and quick stopping in roping, cutting, or reining competition. Recently, DDF tendinitis has been identified as the major contributor of lameness in horses with foot pain.343,345 Desmitis of the accessory ligament of the DDF tendon346 and the accessory ligament of the SDF tendon347 are unusual injuries in performance horses.
Tendons passively transfer muscle activity to the bone. In the equine distal limb, the palmar tendons act to support the fetlock during normal weight bearing and must withstand high—tensile stress forces during locomotion. To combat these forces, tendons contain a high proportion of type I collagen, and the tendon fibers are aligned longitudinally. When loaded, these fibers deform elastically by stretching. Normally, substantial elongation, up to a 20% increase in length,348 occurs before failure. In thoroughbred horses, normal SDF tendons have stains reaching up to 16% when galloping.349 This suggests that tendons in racehorses are close to their physiologic limit and may explain the high incidence of injury. Also, age-related and exercise-related tendon matrix deterioration may contribute to tendon weakness.350 In addition, landing after a jump increases the SDF load,351 which may explain the incidence of tendon injury in horses used for Grand Prix jumpers and eventing.
Tendon injury may occur as a single event or a cumulative fatigue failure and involves stretching of the fibers beyond physiologic limits. Response to tendon injury is similar to other tissue healing and includes inflammation, repair, and remodeling. During the acute inflammatory phase, hemorrhage and edema occur, and the degree of the response is determined by severity of the injury. Angiogenesis quickly follows, and recruited fibroblasts produce new type III collagen. A fibrinous scar of weak, haphazardly arranged collagen is formed. At this stage the tendon is prone to reinjury. Finally, the scar tissue remodels over several months, converting type III collagen to type I collagen. Controlled loading of the tendon promotes this conversation and aligns the collagen fibrils in the direction of force (longitudinally). In severely injured tendons the entire process can take 12 or more months, and tendon reinjury is common.
Clinical Signs
Clinical signs of acute SDF tendon injury include swelling and heat in the tendon and peritendinous tissues. Thickening or enlargement of the tendon is noted when it is viewed from the lateral aspect, and the classic convex or bowed profile in the metacarpal region is apparent. A painful response to direct digital palpation can be appreciated and is best performed by holding the leg and palpating the tendon between thumb and forefinger. Unless the injury is severe, most horses with tendinitis are not lame.
Horses with DDF tendinitis are moderately lame,341,342 become more lame after distal limb flexion, and have distention of the distal digital tendon sheath.341 Injuries are usually unilateral.341,342 Horses with DDF tendon injuries within the hoof capsule have unilateral or bilateral lameness without any palatable abnormalities.343,345 Horses with desmitis of the accessory ligament of the DDF tendon have obvious swelling and acutely are moderately to severely lame.346 Thickening and effusion in the carpal canal may be noted in horses with desmitis of the accessory ligament of the SDF tendon.
Diagnosis
Diagnosis of SDF tendon injury in the metacarpal region is obvious based on abnormal clinical signs. Diagnostic analgesia may assist the clinician in localization of the tendon injury if clinical signs are subtle or absent. This is particularly true for DDF tendon injury within the hoof capsule. Pain from DDF tendon injury in the distal metacarpal region can improve after intrasynovial analgesia of the distal digital tendon sheath.341 Antebrachial or proximal metacarpal analgesia alleviates pain from accessory ligament of the SDF tendon.347
Ultrasonography is the technique of choice to diagnose tendinitis, and considerable advances in instrumentation and clinical aptitude have occurred in the past decade. The entire palmar/plantar area of the metacarpus/metatarsus should be examined in transverse and longitudinal planes. The variables typically measured include cross-sectional area, echogenicity, and fiber alignment. The severity of the injury is determined by the length of the lesion, cross-sectional area of the tendon, cross-sectional area of the lesion, lesion echogenicity, and fiber alignment of the lesion.340,352,353 The severity rating score has been correlated with successful outcome. The most common type of SDF tendon injury is a central core lesion that appears as an anechoic lesion.340,352
Tendon injuries of the DDF within the hoof capsule have been identified using transcuneal ultrasonography.344 Clinician expertise and meticulous foot preparation are required. With the advent of MRI, DDF tendon injuries within the foot have been described. Injury types include core lesions, sagittal tears, and fibrillation along the dorsal border, and most lesions occur at or near the navicular bone.343,345,354
Treatment
Proper treatment of tendinitis includes assessment of the injury using ultrasonography, control of the acute inflammatory episode, controlled return to function, and adjunct medical/surgical therapy. In acute injury, a combination of systemic NSAIDs (e.g., phenylbutazone, flunixin meglumine) and topical therapy (e.g., cold-water hydrotherapy, poultice, distal limb bandage) is recommended. DMSO and antiphlogistically impregnated gauze bandages may also be beneficial. Rest is essential during the acute phases.
A variety of intratendinous and peritendinous injections have been advocated. Intralesional corticosteroids dramatically decrease the swelling and pain but cannot be recommended because of delayed tendon healing and the risk of continued injury and catastrophic injury in horses that remain in training. β-Aminoproprionitrile fumarate (BAPTEN) temporary blocks the cross-linking between collagen fibers and improves tendon healing, resulting in superior fiber alignment;355 horses with moderately to severely injured tendons were more likely to return to racing after intralesional treatment.356,357 This treatment has fallen out of favor because of complications associated with the procedure. For DDF tendon injuries, intrathecal injections of sodium hyaluronate into the distal digital tendon sheath are beneficial.358 Intralesional sodium hyaluronate for SDF tendonitis has been advocated,359 although other reports failed to document any significant benefit.357,360 Intramuscular polysulfated glycosaminoglycan (GAG) has a positive effect on tendon healing in a collagenase-induced model.361 However, no difference in return to racing or recurrence of SDF tendinitis has been demonstrated with intralesional or intramuscular polysulfated GAG.357 Newer therapies include intralesional injections of urinary bladder matrix powder and stem cells derived from fat or bone marrow. Long-term effects have yet to be determined.
Surgical treatments include tendon splitting,362 desmotomy of the superior check ligament,363-366 and desmotomy of the palmar/plantar annular ligament.367 Tendon splitting is believed to enhance revascularization and has a tendency toward improved tendon repair.362 It is currently used for in horses with core lesions to decompress areas of hemorrhage. Superior check desmotomy may improve the likelihood that horses return to racing363 and is clearly beneficial in standardbred racehorses.363,364 In thoroughbred racehorses, postoperative success was initially promising;365 however, no difference between horses treated conservatively and horses treated with desmotomy has been reported.366 In addition, desmotomy may predispose horses to desmitis of the suspensory ligament.364,366 Annular desmotomy has been successful performed in horses with signs of annular ligament constriction367,368 and is usually combined with superior check desmotomy.368 Immediate decompression and improved gliding function are presumed benefits.
Other therapies for SDF tendinitis include therapeutic ultrasound, low-power laser treatment, and extracorporeal shock wave therapy. Counterirritation (internal or external blisters) and pin firing in combination with controlled exercise or turnout in a large pasture are time-honored treatments, but their effects on tendon healing have been questionable.
Controlled exercise in combination with the previous treatments guides the repair process in hopes of producing a functional tendon or tendon substitute. If exercise is excessive, fibrous tissue is produced rather than remodeled. If no stress is applied, the tendon is more adapted to pasture exercise than performance. Therefore, the exercise regimen must be adapted to the individual patient’s needs. Depending on the severity of the tendon injury, stall rest with hand walking and gradual introduction of trot is indicated. Horses should be monitored every 2 months using ultrasonography until they have returned to their previous athletic function or have failed to do so. The exercise program is dictated by the amount and type of healing. As the tendon heals, its echogenicity increases and short, linear echoes are detected. With continued healing, there are longer linear echoes and improved fiber alignment. Decrease in the cross-sectional area is a good indication that the tendon is healing and remodeling. Ideally, complete repair will occur, and the tendon will no longer have a discretely visible area of injury because it has filled in with tissue that is isoechoic with normal tendon, and fiber alignment is parallel to normal.352
Prognosis
Prognosis for horses with SDF tendinitis depends on the severity of the lesion and the type of athletic work performed. Racehorses usually have a poorer prognosis, and 58% return to racing with severe injuries, 66% for moderate injuries, and 64% for mild tendinitis.369 Most horses treated with superior check desmotomy returned to racing; however, recurrence363-365 and suspensory desmitis364,366 may develop. Show horses have a higher likelihood of return to function,357 and many can continue to compete, although at a lower level. Horses with DDF tendon injuries have a poor prognosis.341,343,345 Prognosis for horses with desmitis of the accessory ligament of the DDF tendon is good.346 Prognosis for horses with uncomplicated desmitis of the accessory ligament of the SDF is fair, although horses with concurrent injuries are likely to have recurrent lameness.347
SUSPENSORY LIGAMENT DESMITIS
Definition and Etiology
Suspensory ligament desmitis is a common injury of performance horses and affects all breeds of horses. Injuries to the ligament occur in both the forelimbs and the hindlimbs, and bilateral desmitis may be seen. The suspensory ligament, also named the third interosseous muscle, can be divided into three regions of interest: the proximal (origin) portion, the body, and the branches. The proximal portion originates from the distal row of carpal or tarsal bones, the proximal palmar/plantar aspect of the third metacarpus/metatarsus, and the palmar carpal or tarsal ligament. The body of the suspensory ligament descends between the second and fourth metacarpal/metatarsal bones and then divides into the medial and lateral branches in the middle metacarpal/metatarsal region. The branches insert on the abaxial surface of their respective sesamoid bones. The main function of the suspensory ligament is to support the hyperextension of the metacarpophalangeal or metatarsophalangeal joint during weight bearing.370 Injury to the ligament occurs when it is overwhelmed with stress and its structural integrity is compromised. The damage induces a repair process characterized by inflammation (desmitis) and fibroplasia. It is surmised that this process is similar to tendinitis.370
Catastrophic traumatic disruption of the suspensory apparatus occurs almost exclusively in the forelimbs of thoroughbred racehorses. It is the most common cause of racetrack breakdown injuries.371 Fatigue and high speed are components of this career-ending injury.
Degenerative suspensory desmitis is a progressive degeneration primarily affecting the suspensory branches. It is most often reported in Peruvian Pasos,372 although has been identified in other breeds. The pathogenesis remains unclear, although the disease appears to run in families.373 This disorder has no gender or age predilection and can occur in the absence of athletic activity. Affected horses have continuous enlargement of the suspensory ligament because of ineffective collagen fiber repair and generalized interstitial and periligamentous fibrosis.372
Clinical Signs
Clinical signs of acute suspensory ligament desmitis include local heat, swelling, and sensitivity on palpation. In horses with proximal suspensory desmitis, swelling is in a palmar or plantar direction and frequently mild and transient because of the bony borders of the ligament. Most horses are lame, and the degree and duration of lameness depend on location and severity of the lesion. The lameness is usually worse when the horse is trotted in a circle with the affected limb on the outside of the circle. Lameness and local signs may resolve after a short period of rest and antiinflammatory treatment.
Acute, severe traumatic disruption of the suspensory apparatus results in obvious swelling, grade 4 to 5 lameness, and a hyperextended fetlock. Affected horses require first-aid treatment by application of a splint that aligns the metacarpus with the phalanges in a straight column to prevent fetlock bending.
Clinical signs of chronic suspensory desmitis include thickening of the body and branches of the ligament. Lameness is variable and may be bilateral. Chronic desmitis is typically associated with sesamoiditis, periostitis, or fractures of the small metacarpal/metatarsal bones and sesamoid bones. Straight hock conformation and hyperextension of the fetlock joints appear to be predisposing factors in proximal hindlimb suspensory desmitis, and affected horses are chronically lame despite rest.374 Degenerative suspensory desmitis is characterized by recurrent chronic lameness, marked fetlock swelling, dropped fetlocks, excessive lying down, and reluctance to move. The majority of horses will be affected in all four limbs.372
Diagnosis
If suspensory desmitis is suspected and clinical signs are not obvious, diagnostic analgesia techniques assist in the localization of the lameness to the palmar/plantar metacarpal/metatarsal region. If proximal suspensory desmitis is suspected, perineural analgesia of the lateral palmar nerve375 or deep branch of the lateral plantar nerve just distal to the tarsus376 will alleviate the lameness in the forelimb or hindlimb, respectively. Desmitis of the suspensory body and branches can be localized using low palmar/plantar analgesia.
Diagnosis of suspensory desmitis is confirmed by ultrasound examination. The severity of the lesion is assessed by obtaining cross-sectional and longitudinal views of the ligament and cross-sectional area measurements. Ultrasonographic changes include cross-sectional area enlargement, focal or diffuse areas of hypogenicity, core lesions, loss of normal fiber pattern, and periligamentous thickening. Mild injuries to the suspensory ligament may be difficult to identify because of the wide variation in its normal ultrasonographic appearance. This is especially true in the proximal region, where the suspensory ligament origin is more heterogenous than for other tendons and ligaments because of a variable amount of muscle tissue. Comparison of the affected and nonaffected limbs in the same horse is recommended because of large variation among breeds and individuals.370,377
Radiographic examination is useful for identification of the bony abnormalities usually noted with suspensory desmitis. Bony abnormalities such as subchondral sclerosis, avulsion fracture, and enthesophyte formation have been identified in horses with proximal suspensory desmitis.378,379 “Blind splints” associated with axial exostosis of the second and fourth metacarpal/metatarsal bones or fractures may also contribute to suspensory desmitis. Proximal sesamoiditis and apical sesamoid fractures may be noted in horses with desmitis of the suspensory branches. In acute traumatic disruption of the suspensory apparatus, comminuted fractures of both proximal sesamoid bones are common.
Nuclear scintigraphic findings are normal in the majority of horses with suspensory desmitis.380 Increased radiopharmaceutical uptake associated with the proximal palmar/plantar metacarpus/metatarsus can be identified in horses with avulsion fractures and other stress-related bone injuries seen in complex suspensory desmitis.378,379 Recently, MRI evaluation has been used to diagnose proximal suspensory desmitis and fibrous adhesions between the suspensory ligament and the small splint bones.381
Treatment
Treatment of acute suspensory desmitis is aimed at reducing inflammation by the administration of systemic NSAIDs, local cold hydrotherapy, limb bandage, and controlled exercise. Initial clinical signs often dissipate quickly, even though there may be significant fiber disruption of the ligament; therefore it is strongly recommended to assess the initial injury using ultrasonography. In uncomplicated desmitis, a controlled exercise program may be initiated as soon as the acute inflammation has resolved. The duration of rehabilitation will depend on the extent and severity of the desmitis. Fractures of the small metacarpal/metatarsal bones or proximal sesamoid bones should be removed before rehabilitation. Horses are confined to a stall and hand-walked. Ligamentous healing should be monitored every 60 days using ultrasound. Ideally, there should be a progressive reduction in cross-sectional area, improved echogenicity, and fiber alignment. A gradual increase in exercise can be prescribed depending on healing.
Damaged suspensory ligaments have been injected with various corticosteroids, iodine compounds, sodium hyaluronate, and polysulfated glycosaminoglycans in an effort to reduce inflammation and promote a more rapid return to function. Pin firing, blistering, and cryotherapy with liquid nitrogen have also been used. Although these therapies assist with the initial inflammation, they do not restore normal fiber architecture and can put the ligament at increased risk because of overuse of the horse based solely on reduction of clinical signs.
Recurrent or chronic proximal suspensory desmitis has been treated successfully with extracorporeal shock wave therapy.382 Other treatments, such as plantar metatarsal neurectomy,383 intralesional injection of autologous stem cells,384 desmotomy,385 and fasciotomy,383-385 have also been beneficial.
For horses with acute traumatic suspensory desmitis, it is crucial to place in the affected limb in a protective splint that aligns the metacarpus with the phalanges in a straight column to prevent fetlock bending. Successful nonsurgical management using splints, casts, and special shoes to support the fetlock has been reported.386 Surgical management involves fetlock arthrodesis using dorsal plating.
For horses with degenerative suspensory desmitis, treatment is palliative and consists of stall confinement for 6 to 12 months and extended heel or large egg-bar shoes. Humane euthanasia is recommended with continued breakdown despite treatment.
Prognosis
The prognosis for horses with suspensory desmitis depends on the location and severity of the injury. Uncomplicated cases with minimal disruption of the ligament can recover and resume preinjury athletic function; 90% of horses with acute proximal suspensory desmitis in the forelimb will return to full athletic function.387 Complicated, chronic, or recurrent desmitis has been associated with a worse prognosis; only 13% of horses with hindlimb proximal suspensory desmitis return to full work.374 Most horses with traumatic disruption of the suspensory ligament can be salvaged for breeding, although meticulous nursing care in horses treated conservatively and uncomplicated surgical repair are imperative. Prognosis for horses with degenerative suspensory desmitis is extremely poor.
FRACTURES
The ability to treat fractures in large animals depends largely on rapid and correct diagnosis, appropriate first-aid treatment and stabilization of the injured limb, and safe transportation to a referral surgical center. Emergency treatment administered by a veterinarian in the field is crucial to provide the most treatment options and to enhance the prognosis. Accurate knowledge of available treatment options and the prognosis for various fracture types is critical during discussions with clients. The surgeon receiving a patient with a fracture should be contacted early and involved in the decisions regarding limb stabilization, transportation, and owner financial commitments. Treatment options are discussed in this section; specific surgical principles related to fracture repair can be found in surgical textbooks.
Definition and Etiology
A fracture is defined as a break or rupture of a bone. In large animals, fractures are usually classified as complete or incomplete, articular or nonarticular, displaced or nondisplaced, open or closed, simple or comminuted, and transverse, oblique, or spiral. Classification by location is also common and includes terms such as physeal, metaphyseal, and diaphyseal.
Fractures in large animals are typically associated with an acute traumatic event, followed by a sudden, severe lameness. Horses may have a history of falling or running into a solid object, receiving a kick from another horse, being hit by a car, or flipping over backward (typically young animals). In another common scenario, the owner hears a loud pop or cracking sound while the horse is being longed or is participating in an athletic activity.
Stress fractures may also occur and often are a result of cyclic loading over time with corresponding fatigue, leading to bone microfracture. Intensive training schedules create a high number of loading cycles with insufficient time to remodel and repair local areas of stressed bone (e.g., racehorse training). Specific locations of stress fractures include the distal end of the scapular spine, caudoproximal humeral head, craniodistomedial aspect of the humerus, medial humeral diaphysis, caudal aspect of the radius, dorsal middiaphyseal and distodorsolateral third metacarpal, caudal border of the ilial wing and adjacent to the pubic symphysis, proximolateral tibia, and caudal diaphysis of the tibia.388
Other, less common causes of large animal fractures include nutritional deficiencies, neoplasia, and infectious causes leading to pathologic fractures. Fractures related to difficult parturition events are seen in neonatal foals and calves.
Clinical Signs and Differential Diagnosis
An animal with a long-bone fracture will typically present with an acute, non—weight-bearing lameness of the affected limb. Moderate to severe soft tissue swelling and inability to control the limb often accompany other clinical signs of distress, agitation, and pain. Because of the horse’s “fight or flight” reaction to traumatic situations, the patient will often continue attempts to place weight on the fractured limb. Fractures in locations with limited soft tissue coverage are often open (e.g., third metacarpal). Severe hemorrhage or laceration of large vessels is uncommon but may occur.
Complete, catastrophic fractures are often easily identified based on limb instability and soft tissue trauma or swelling. Proximal limb fractures may be difficult to palpate but have adjacent severe soft tissue inflammation and edema, crepitus, and difficulties in limb function. Nondisplaced or incomplete fractures may be associated with a wound or may be painful to manual palpation. Focal pain on palpation, a corresponding lameness, and a history of trauma or being kicked by another animal warrant radiographic evaluation in search of an incomplete fracture. An unrecognized incomplete fracture may progress to a complete fracture with time and exercise. Physeal fractures should be considered in young animals.
Stress fractures often present with a history of an acute, severe lameness followed by a rapid decrease in the degree of lameness. There may also be reports of intermittent lameness that improves with stall rest. Palpation of the limb often reveals a focal area of pain, heat, and swelling. The horse may resist manipulation of the limb.
Differential diagnoses should include hoof and sole abscesses, joint subluxation, joint luxation, nerve damage, soft tissue injury, and septic synovial structures.
Clinical Evaluation
A complete physical examination should be performed on initial evaluation. Animals that are not immediately identified as having a fractured limb may be in need of systemic support. Fluid therapy, pain control, antiinflammatory medications, and antimicrobials should be used as necessary.
Definitive diagnosis is made by taking multiple radiographic views; nuclear scintigraphy, ultrasonography, CT, and MRI may also be used. Information from imaging studies will provide fracture configuration details essential for determining prognosis. The bone involved, degree of comminution, articular involvement, physeal involvement, and condition of the bone fragments all affect potential repair attempts and long-term prognosis. Incomplete fractures may not be readily identified at the time of injury, and radiographs should be repeated in 10 to 14 days if a fracture is suspected.
Stress fractures also are diagnosed using radiographic evaluation and may be identified as an incomplete cortical fracture, endosteal callus, or periosteal callus.389 As with incomplete fractures, it may be difficult to identify a stress fracture by conventional radiography in the acute stages. Radiographs should be repeated in 10 to 14 days if a fracture is suspected. Other imaging modalities, especially nuclear scintigraphy or CT, may be useful in stress fracture identification.
Emergency Treatment
The goals of emergency treatment include preventing further soft tissue damage, stabilizing the injured limb to decrease the patient’s anxiety, minimizing further damage to the fractured bone ends, keeping the fracture closed, and preventing further injury and stretching to the surrounding vessels and nerves.390 A large animal with a fractured limb should be immediately restrained and calmed. Sedatives and tranquilizers should be used with caution, however, because the desired result may be difficult to achieve if the animal is agitated or in shock. The goal of sedation should be to allow manipulation of the limb and placement of external coaptation. Common sedatives should be used with caution to avoid creating additional ataxia; butorphanol should not be used if the horse has sustained a front limb fracture because it will make the horse lean onto the forehand. If a soft tissue wound is present, the clinician should assume that the fracture is open. The wound should be cleaned, topical antimicrobials used, a bandage placed to prevent further contamination, and the animal started on systemic antimicrobials. Antiinflammatory medications should be used as necessary.
Appropriate external coaptation and stabilization of the fracture for transport can greatly influence the options available for fracture repair and the prognosis for success. Table 38-5 outlines specific emergency splints for different fractures in bones.390,391 Splinting techniques are designed to be placed on the standing horse and attempt to neutralize forces acting on the fractured limb. Splints should be made of a strong, lightweight material such as polyvinyl chloride (PVC) pipe or wooden boards. Bandages over fractures should be placed tightly over the injured area to decrease soft tissue swelling and minimize slippage. A layer of cotton padding should be followed by a layer of gauze or a self-adhering bandage. Multiple layers can be placed if needed (e.g., Robert Jones—type bandage). The splint should not end at the fracture location. In general, splinting techniques can be divided into categories based on fracture location.
Table 38-5 Emergency Splinting Techniques for Fractures in Large Domestic Animals
| Location | Species/Age | Splint |
|---|---|---|
| Distal phalanx | Horses, foals Cattle, calves |
No splint needed |
| Middle phalanx | Horses, foals | Bandage with dorsal (forelimb) or plantar (hindlimb) splint to proximal third metacarpal/tarsal and cast material, Kimzey splint* |
| Proximal phalanx | Horses, foals | Bandage with dorsal (forelimb) or plantar (hindlimb) splint to proximal third metacarpal/tarsal and cast material, Kimzey splint* |
| Cattle, calves | No splint needed | |
| Distal sesamoid (navicular) | Horses, foals | No splint needed |
| Proximal sesamoids | Horses, foals | |
| Bandage with dorsal (forelimb) or plantar (hindlimb) splint and cast material, Kimzey splint* | ||
| Metacarpal/tarsal 3 | Horses, foals | Bandage with dorsal (forelimb) or plantar (hindlimb) splint and cast material, Kimzey splint* if fracture distal |
| Metacarpal/tarsal 3 & 4 | Cattle, calves | Bandage with dorsal (forelimb) or plantar (hindlimb) splint and cast, cast |
| Metacarpal/tarsal 2 & 4 | Horses, foals | No splint needed |
| Olecranon/ulna | Horses, foals | Robert Jones—type bandage with caudal splint to lock carpus in extension |
| Radius | Horses, foals Cattle, calves |
Robert Jones—type bandage with lateral splint to withers, second caudal splint if needed |
| Humerus | Horses, foals Cattle, calves |
No splint needed |
| Scapula | Horses, foals Cattle, calves |
No splint needed |
| Tibia | Horses, foals | Robert Jones—type bandage with lateral splint to tuber coxae |
| Femur | Horses, foals Cattle, calves |
No splint needed |
| Pelvis | Horses, foals Cattle, calves |
No splint needed |
* Leg-Saver splint, Kimzey Inc, 164 Kentucky Ave, Woodland, CA 95695 (530-662-9331, fax 530-662-9178); http://www.kimzeymetalproducts.com.
DISTAL LIMB FRACTURES (PHALANGES, DISTAL METACARPUS/METATARSUS)
A light cotton bandage should be placed on the affected limb from the carpus to the ground. A splint is then positioned on the dorsal aspect of a forelimb or the plantar aspect of a hindlimb. The toe is pointed toward the ground to align the dorsal cortices and to minimize stretching of the palmar/plantar vascular and nerve structures. A second splint should be placed on the medial or lateral side of the limb if any instability is present in those directions. The splint should be secured with inelastic tape. For additional stability, several rolls of fiberglass casting tape should be placed around the entire bandage and splint structure, incorporating the hoof into the cast.
MIDLIMB FRACTURES (MIDMETACARPUS TO DISTAL RADIUS, MIDMETATARSUS TO PROXIMAL METATARSUS)
A Robert Jones—type bandage should be placed on the limb from the ground to the elbow in a forelimb or the ground to the calcaneal tuber in a hindlimb. The bandage should be less extensive in the hindlimb to facilitate splint placement. In the forelimb, splints should be placed on the lateral and caudal aspects of the limb from the ground to the elbow. In the hindlimb, splints should be placed on the lateral and caudal aspects of the limb from the ground to the calcaneal tuber. The splints are secured using inelastic tape, followed by several rolls of fiberglass casting tape around the entire structure.
UPPER LIMB FRACTURES (MIDDLE AND PROXIMAL RADIUS, TARSUS, TIBIA)
A Robert Jones—type bandage should be placed on the limb from the ground and extending as proximal as possible. In the forelimb, a splint should be placed on the lateral aspect of the limb from the ground to the withers. A second splint may be placed on the caudal or cranial side, from the ground to the proximal forearm, if needed for increased stability. Horses with olecranon fractures that disable the triceps muscle benefit from reestablishment of carpal extension by placement of a caudal splint. In the hindlimb, a splint should be placed on the lateral aspect from the ground to the tuber coxae. Splints placed high in this fashion will assist in minimizing abduction of the limb.
HIGH LIMB FRACTURES (PROXIMAL TO ELBOW JOINT, FEMUR)
Fractures in these locations are covered by extensive muscle, and attempts at splinting are usually unsuccessful and not helpful to the animal. If triceps muscle function is disabled, a splint on the caudal aspect of the carpus, locking it into extension, is beneficial.
TRANSPORT
Transportation of horses with long-bone fractures can be difficult. It is best to confine the patient in the trailer so that they may lean on dividers for support. The horse should be tied loosely in the trailer to prevent turning around, but allowing the animal to use its head for balance. When possible, horses with forelimb fractures should be transported with the head facing the rear of the trailer, and those with hindlimb fractures should be transported with the head facing the front of the trailer. These positions allow the horse to control its weight most easily with the uninjured limbs during deceleration of the trailer.