MODIFIED LIVE VIRUS PARENTERAL VACCINES
The modified live virus parenteral vaccines were the initial vaccines licensed for use in cattle for protection against BHV-1.238 Vaccines are attenuated by multiple passages in cell culture and often retain their ability to replicate in a susceptible animal, possibly causing a viremia. Modified live virus parenteral vaccines are relatively inexpensive, offer a convenient route of administration, and stimulate a rapid onset of immunity (i.e., within 3 days of administration).239-241 In general, one dose given to a susceptible animal stimulates protective immunity, which varies in duration depending on the clinical form of the disease challenge. Calves receiving a combination modified live virus vaccine with BHV-1 were protected for at least 126 days after vaccination as measured by protection against infection242 The modified live virus parenteral vaccines may cross the placenta and infect the fetus, causing abortion.243 Almost all modified live virus BHV-1 parenteral vaccines are not approved for use in pregnant heifers or cows nor for nursing calves.234 Recently two companies have received label claim approval for BHV-1 and BVDV modified live virus vaccine use in pregnant cows providing they vaccinated with that line of vaccines within 12 months of being vaccinated during pregnancy and to nursing calves provided their dams were vaccinated within 12 months.234
MODIFIED LIVE VIRUS INTRANASAL VACCINES
Modified live virus intranasal vaccines generally can be divided into two types, based on the attenuation process: (1) those modified by passage in a cell culture244,245 and (2) those modified by treatment such that they become “temperature sensitive”246 (i.e., they do not replicate at internal body temperature). Modified live virus intranasal vaccines stimulate protection in susceptible animals with only one dose, in contrast to the chemically altered modified live virus parenteral vaccines. The label directions for selected, but not all, modified live virus intranasal vaccines may indicate that they can be safely used in pregnant cattle.234 These vaccines induce a rapid onset of protection (within 3 days of administration), possibly through IFN in the nasal secretions.244 One benefit of modified live virus intranasal vaccines is that they stimulate immunity at the upper respiratory tract, the portal of entry of the virus. Another benefit is their potential to immunize calves that are already seropositive because of maternal (humoral) antibodies passively transferred through the colostrum.247 Animals vaccinated with modified live virus intranasal vaccines may transiently shed virus in the nasal secretions and therefore might infect susceptible contact animals.248
CHEMICALLY ALTERED LIVE VIRUS VACCINES
The chemically altered BHV-1 vaccine strain was modified by nitrous acid treatment, which caused changes in the viral genome that resulted in a strain that is temperature sensitive, meaning that it has limited replication at internal body temperature.249 Presumably, because of the limited viral replication, the vaccine requires two doses to stimulate immunity. Because it is temperature sensitive, the vaccine can be used in pregnant cattle.234,249,250 In one study, heifers received two doses of the vaccine and were challenged with BHV-1 7 months later (at 6 months’ gestation). These heifers showed a significant reduction in the number of abortions and stillbirths compared with controls.250
INACTIVATED VIRAL VACCINES
Inactivated viral vaccines are prepared by growing virus in cell cultures and then inactivating them with chemicals. An adjuvant is added to the inactivated strain to help stimulate an immune response. Inactivated BHV-1 vaccines require two doses (14 to 28 days apart) when used for the initial vaccination of susceptible cattle. Historically it has been thought that inactivated vaccines against viruses did not induce as long a DOI as the modified live virus vaccines, nor did they confer protection against mucosal infections. Controlled studies should be performed to determine the DOI induced by inactivated BHV-1 vaccines and modified live virus vaccines, both for respiratory disease and for fetal infections. A disadvantage of inactivated vaccines is that the onset of protection may not be as rapid as with modified live virus parenteral or modified live virus intranasal vaccines. An advantage of the inactivated vaccines is that they can be used in pregnant cows and nursing calves.
Use of Vaccines to Prevent and Control Bovine Herpesvirus Type 1 Diseases
Many vaccines are available for preventing and controlling the different forms of BHV-1 disease, and each vaccine has certain characteristics that should be considered when designing a vaccination program. Each vaccine also has both benefits and limitations. Probably more important is the management of the cattle for which the vaccines are used.
PREGNANT ANIMALS OR ANIMALS NEARING BREEDING SEASON
The modified live virus parenteral vaccines may infect the fetus if pregnant susceptible heifers or cows are vaccinated. Abortions have been reported subsequent to vaccination with modified live virus parenteral vaccines.243 The modified live virus vaccine virus may also result in corpus luteum infection or disease.251,252 Experimental studies have indicated a reduced conception rate in susceptible cattle that received a modified live virus parenteral vaccine 3 to 4 days before or 14 days after breeding.251,252 It has been reported that pregnant cattle raised in contact with calves recently vaccinated with modified live virus parenteral vaccines had a greater incidence of BHV-1 abortion than those that did not have contact with vaccinates.253 Consequently, the labels of modified live virus parenteral vaccines have usually stated that the vaccine should not be used in calves nursing pregnant cows. Recent studies have shown that calves given a modified live virus parenteral vaccine did not shed virus in their nasal secretions, nor did contact animals become infected with the vaccine virus.254-256 One company received label claim of prior vaccination for the modified live virus vaccine containing BHV-1 and BVDV for pregnant cows, provided the cows had received the same line of vaccines with the modified live virus BHV-1 and BVDV within 12 months of being vaccinated during pregnancy.234 Likewise, that vaccine could be used in nursing calves if the cows had been previously vaccinated with that line of vaccines within 12 months of prior vaccination.234 Another concern is that the modified live vaccine virus may recrudesce, with resulting shedding of virus in cattle either stressed or receiving corticosteroids.257 Realistically, concern about transmission of BHV-1 to animals in contact with those receiving modified live virus parenteral vaccines would be negligible if the contact animals were properly immunized and immune to BHV-1.
Until the vaccine labels on most modified live virus parenteral vaccines are changed, modified live virus intranasal vaccines or the inactivated or chemically altered live virus vaccines usually are recommended for pregnant cattle or those near breeding. The one exception is the approved vaccine cited previously. Vaccine recommendations should be weighed using the benefits of vaccination as a guide and especially with the realization that properly vaccinated cattle are better protected when exposed to either field (virulent) or vaccine strains shed by vaccinated animals.
RAPID ONSET OF IMMUNITY
Cattle that are susceptible and likely to be exposed to BHV-1 should receive either a modified live virus parenteral vaccine or a modified live virus intranasal vaccine because both types induce immunity within 3 days of the initial dose. Rapid onset of immunity is desirable in such situations as stocker calf and feedlot operations, in which calves are transported long distances to pastures or feedlots, which stresses the animals and makes them more susceptible to infection. Such calves also are exposed to infection with BHV-1 from contact cattle in the markets. The drawback to inactivated vaccines is that two doses are required to obtain good immunity.
DURATION OF IMMUNITY
Controlled studies on the DOI are limited. A degree of protection against challenge existed at 6 to 9 months after vaccination with a modified live virus intranasal vaccine or an inactivated vaccine.258,259 A parenteral modified live virus BHV-1 vaccine provided protection up to 126 days after vaccination.242 A second parental modified live virus BHV-1 vaccine provided protection against BHV-1—induced abortions for 12 months after vaccination.234 Challenge studies for licensure usually are performed on calves within days of vaccination, at the time of peak immunity. Also, the challenge may be for only one form of disease, usually the respiratory type. Such challenges may detect only protection against a severe form of the respiratory disease. BHV-1 manifests itself in other forms, such as abortions, neonatal disease, genital disease (male and female), and conjunctivitis. Yet little or no data are available regarding the efficacy of vaccines against these other forms of disease. For example, in one case the genital form of BHV-1 disease (infectious pustular vulvovaginitis) occurred in heifers that had received a modified live virus parenteral vaccine 5 months earlier.260 Given the lack of DOI studies for all BHV-1 vaccines individually and the cost of vaccines, breeding animals usually are vaccinated at least annually. In some feedyard situations the animals may be revaccinated during the feeding period. It is industry practice that feedlot cattle receive a monovalent BHV-1 modified live virus parenteral vaccine at reimplant time at approximately 100 days after arrival. There have been field reports of BHV-1 respiratory disease (IBR) in feedlot cattle a few months after entry or processing, at which time they received modified live virus vaccines containing BHV-1.
VACCINATION OF CALVES WITH MATERNAL ANTIBODY
The possibility exists that maternal BHV-1 antibodies acquired by the calf through ingestion and absorption of colostrum may interfere with vaccination. The level of these serum BHV-1 antibodies in the calf depend on the amount in the colostrum, the amount absorbed, and the half-life of the particular antibody; for BHV-1, it is 21.2 days.236 Some calves receive no BHV-1 antibodies through the colostrum, or they may lose them within 1 month. Some calves, however, may have serum BHV-1 antibodies for up to 6 months after birth.257
Vaccination recommendations for neonatal calves include use of multiple doses of a modified live virus parenteral, an inactivated, or a chemically altered live virus vaccine or administration of a modified live virus intranasal vaccine. The maternal antibodies may block the parenterally administered modified live virus or inactivated vaccine. However, the modified live virus intranasal vaccine may induce BHV-1 antibody immunity.247 Calves often are revaccinated at 6 to 8 months of age regardless of their prior vaccination history.
ADVANCES IN VACCINES
Molecular techniques of biotechnology have been applied to the study of vaccines and the response to vaccination (vaccinology). These advances are especially noted for herpesviruses, including BHV-1. In addition to conventional vaccines manufactured via propagation of modified live virus and inactivated BHV-1 strains, current and future technologies offer opportunities for other vaccines.261,262 These include subunit vaccines with a portion of the virus, deletion mutants with specific viral genomic fragments deleted, live vectored strains, DNA vaccines using plasmids, and plant-based vaccines. Deletion mutant BHV-1 vaccines as marker vaccines with selected glycoprotein genes deleted along with diagnostic tests for the deleted genes permit identification of vaccinates under control programs.261 Recently, needle-free delivery of vaccines has been developed and implemented.261 By high pressure gas delivery, vaccines may penetrate the skin and be administered intradermally, subcutaneously, or intramuscularly.261 Such delivery is designed to minimize damage resulting from intramuscular injections. Two studies compared needle-free intramuscular injection of multivalent modified live virus vaccine containing BHV-1 with conventional subcutaneous injection via syringe in dairy calves and feedlot cattle. In both studies, antibody titers to BHV-1 were higher at day 21 postvaccination than after conventional needle injection.263,264
Vaccination Programs
The best possible vaccine provides protective immunity in the host against infection (viral replication) when challenged; protects the animal against all forms of disease, including multiple organ and systemic forms; and provides lifelong mucosal and systemic immunity. Ideally the vaccine recommendations would incorporate the results of field trials that are carefully designed to show the efficacy of the vaccine against a pathogen. Unfortunately little information is available, as can be seen by a review of the literature, for evaluating the field efficacy of the respiratory disease vaccines.265 The summary of results was mixed for BHV-1 vaccines and for other respiratory viral and bacterial vaccines.
Veterinarians therefore must make recommendations based on (1) experimental studies of vaccination followed by challenge under controlled laboratory conditions apart from the field conditions of normal cattle management and (2) clinical experience with vaccines. Data from challenge studies often are under the control of universities, the federal government, or a biologic manufacturer and may not be published. Government licensure studies require efficacy and safety evaluations, but these studies may not be available in scientific publications for review by those making recommendations. For these reasons the veterinarian may not have access to all the data needed to make a good decision on vaccines.
The veterinarian’s dilemma is confounded by other factors. First, licensure may be granted for vaccination efficacy that demonstrated protection against one form of disease, such as respiratory type. However, the virus may be just as important a pathogen of other organs, such as the developing fetus, as is the case with BHV-1. Second, information about the DOI induced by each commercially available BHV-1 viral vaccine is not available or is limited. Licensure studies may use challenge of vaccinated animals within 2 to 4 weeks of initial vaccination, yet cattle may be in feedyards (months) or breeding herds (years) after vaccination. Third, vaccines may induce a strong parenteral immunity, yet the surface mucosal defenses at the portal of entry may still be susceptible to infection even in presumably well-vaccinated animals. Thus it is entirely possible that natural infections could still occur in these vaccinated animals. Ecologically this point is reinforced, because viruses for which there are good immunization products are still circulating in cattle populations after years of vaccination.
The veterinarian therefore must weigh both the benefits provided by vaccines and their limitations. This probably is best done by focusing on the real, economic effects of certain disease manifestations, including morbidity, mortality, and treatment and prevention costs. Historically this approach has been applied to two important forms of BHV-1 disease: the respiratory form, singularly or in combination with pneumonic bacterial diseases, and the fetal disease (abortions). As a result, most vaccine regimens focus on preventing respiratory disease in both young or adult animals and on protecting the pregnant breeding herd of cows and heifers against abortions. Another fact to be considered is that many vaccines may have multiple viral or bacterial components (or both), which may require multiple doses for an immunogen mixed with one that requires only one dose.
GUIDELINES
Calves
Calves may be vaccinated at weaning or 30 days before weaning. Calves vaccinated before 6 months of age should be revaccinated because the earlier vaccination may have been blocked by maternal antibodies. The modified live virus parenteral and intranasal vaccines require only one dose in susceptible calves, whereas the chemically altered live virus or inactivated vaccines require two doses. Although the labels for most modified live virus parenteral vaccines state that the vaccine should not be used if the calf is nursing a pregnant cow, the likelihood of infection of the pregnant cow may be minimal, especially if she is already immune. Yet as described earlier, modified live virus parenteral vaccines are available for use in pregnant cows and nursing calves.
Breeding Cows and Heifers
Yearling heifers (12 to 14 months of age) should be vaccinated at least 1 month before breeding. Any of the vaccines may be used, but if two doses are required, the second dose should be given at least 1 month before breeding.
Pregnant cows may be vaccinated with a vaccine that has a label description warranting such use; these include modified live virus intranasal vaccines, chemically altered live virus vaccines, inactivated vaccines, and approved modified live virus parenteral vaccines. Generally one dose is used, primarily because of management considerations. Administering booster doses of the BHV-1 vaccines may have two conflicting outcomes as a result of booster dose stimulation of an increase in colostral BHV-1 antibodies, which are transferred to the newborn calf in the colostrums: (1) it may be beneficial to the calf to have increased BHV-1 serum antibodies for protection against BHV-1 disease, or (2) the calf may have longer duration of BHV-1 antibodies, which may block BHV-1 immunization. There are no published multiyear DOI studies in vaccinated cattle challenged with virulent BHV-1. Because of the relatively low cost of BHV-1 vaccines and the need to vaccinate against other pathogens, many breeding cows are given a BHV-1 vaccine annually.
Stocker and Feeder Cattle
Cattle to be shipped to forage pasture after weaning (wheat pasture or native grass) or to feedyards should be vaccinated 2 to 3 weeks before shipment. However, management practices and marketing may permit vaccination only at initial collection point, market site, or stocker or feedlot delivery. All the major types of BHV-1 vaccines may be used, but those that require only one dose have two advantages: onset of immunity is rapid, and less handling is required (one dose versus two).
Cattle presented for purchase immediately before shipment, with no known vaccination history, pose a challenge. Presumably healthy cattle may be candidates for the one-dose modified live virus parenteral or modified live virus intranasal vaccines because these calves may benefit from rapid immunity. Cattle already infected with BHV-1 may not be protected by vaccination.
Cattle entering the feedyard usually receive either the modified live virus parenteral or modified live virus intranasal vaccine, particularly for the rapid onset of immunity. Cattle sometimes are revaccinated during the feeding period to ensure protection against BHV-1 disease later in the feedyard.
Breeding Services
Veterinarians should consult the breeding bull center for vaccination requirements of bulls, especially relating to export shipment and collection for artificial insemination. Modified live virus intranasal vaccines have been used in artificial insemination bulls because these vaccines are less likely to cause latent infections than modified live virus parenteral vaccines.266,267 As mentioned previously, modified live virus parenteral vaccines cause latent infections that may recrudesce with stress or the administration of corticosteroids, which means the virus would be present in the semen.257,267
BOVINE VIRUS DIARRHEA VIRUS VACCINES
Victor S. Cortese
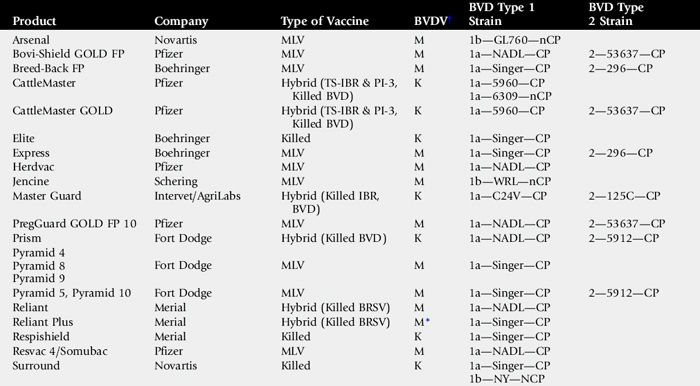
Our knowledge of the different vaccines’ ability to protect against infection with BVDV is increasing rapidly. Currently, three kinds of BVDV vaccines are available: modified live virus vaccines, temperature-sensitive vaccines (available only in Europe), and inactivated virus vaccines (Table 48-12). Modified live virus BVDV vaccines have demonstrated advantages over the newer inactivated vaccines. Most of these advantages are common to all modified live virus vaccines268,269: (1) modified live virus vaccines are less expensive; (2) postvaccination anaphylactic reactions occur less often than after administration of some inactivated vaccines; (3) immunity is achieved more rapidly after administration of a single dose (within 7 to 10 days); (4) modified live virus vaccines produce much higher levels of serum neutralizing antibodies270; (5) immunity lasts longer271-273; and (6) modified live virus vaccines are effective against a broader spectrum of viral strains.270
On the other hand, modified live BVDV vaccines have certain disadvantages compared with inactivated BVDV vaccines; these disadvantages are that (1) some modified live virus vaccines can produce mild immunosuppression for brief periods after administration272; (2) some have been associated with what is usually a rather low incidence of a highly fatal disease syndrome, postvaccinal mucosal disease (MD)273,274 (see later); and (3) until recently, modified live virus vaccines were not ordinarily recommended for routine use in pregnant cattle.
Studies have shown that the duration of cross-neutralizing antibodies stimulated by inactivated BVDV vaccines depends on the antigenic similarity between the vaccine strain and the wild type virus to which the cow is exposed.275,276 If there are few common proteins, the ability to neutralize can be as short as 4 months; if there are many common antigenic sites, neutralization may last a year. Modified live BVDV vaccines stimulate cross-neutralizing antibodies that can still be detected 18 months after vaccination.270 This longer duration was further demonstrated when a modified live BVDV vaccine received a USDA label describing a 1-year duration of immunity against the birth of both type 1 and type 2 persistently infected calves.277
Recent studies have demonstrated the ability of both modified live278,279 and inactivated type 1 vaccines280 to -provide protection against type 2 BVDV strains, although the protection afforded by the modified live virus vaccine was more complete. USDA/APHIS has established a licensing protocol for BVDV vaccines to obtain a type 2 BVDV protection claim. Most of the vaccines licensed against both BVDV type 1 and type 2 contain BVDV type 1 and type 2 isolates (see Table 48-10). The question of current vaccines’ability to cross-protect against BVDV type 1b strains has been raised,281 and further studies are needed to determine the level of protection afforded by current vaccine strains.
VACCINATION AND MUCOSAL DISEASE
MD is seen when an animal that is persistently infected is exposed to another closely related cytopathic strain of BVDV. Also, theoretically a noncytopathic BVDV strain can mutate spontaneously into a cytopathic strain, resulting in MD without any subsequent exposure. High stress and immunodepression may be involved in this mutation.282,283
A major concern with the modified live virus vaccines is whether they can cause MD.275,284,285 For MD to occur, the cytopathic strain in the modified live virus vaccine must be closely related to the noncytopathic strain in the persistently infected animal. With the degree of attenuation of modified live virus vaccines today, an animal must be nutritionally deficient or severely stressed, or both, to face an increased likelihood of developing MD from the vaccine. This suggests that a specific set of circumstances is required and that MD caused by vaccination, when it occurs, is rare.
BOVINE VIRUS DIARRHEA VIRUS VACCINES AND REPRODUCTIVE CONTROL
Control of BVDV centers on prevention of persistent infection and elimination of persistently infected cattle; this means that identifying and removing persistently infected animals and continued vaccination to prevent persistent infection are necessary for effective control. Persistent infections occur through in utero infection of the fetus (up to approximately 125 days’ gestation) with a noncytopathic strain of BVDV.270,282 The mechanism of transplacental transfer of BVDV is unknown; however, small amounts of virus in the dam’s bloodstream appear to be sufficient to produce these immunotolerant cattle. Protection of the dam may or may not correlate with protection of the fetus from subsequent persistent infection if viremia of the dam occurs. To break the vicious cycle of in utero infection and persistent infection, it is essential that vaccination provide fetal protection.
Several studies have been performed to assess the ability of vaccines to protect the fetus against either natural or artificial challenge. The results of these studies showed that most inactivated vaccines failed to provide much fetal protection,275,286-291 except for one experimental vaccine, which is reported to give a high level of fetal protection. With the experimental vaccine, the lack of virus isolation from offspring of vaccinated animals indicated good protection.292 However, the challenge of controls resulted in only approximately a 50% rate of persistent infection. Recently, the new inactivated vaccine has demonstrated a high level of fetal protection and was given the appropriate label by the USDA (see Table 48-9). Other published reports demonstrated that modified live virus BVDV vaccines were more effective at protecting the fetus293-295 and that inclusion of a type 2 vaccine broadened the strains against which the vaccine protects.296 To date, vaccines licensed in the United States have not been required to provide fetal protection. However, label indications are now being granted by the USDA to vaccines that have demonstrated the ability to prevent the development of persistently infected calves. Several vaccines have achieved this label (see Table 48-9).
Bovine Virus Diarrhea Virus Vaccination Programs
Because BVDV infections can cause severe death loss and immunosuppression, all herds of cattle should be vaccinated against BVDV. Although it was once thought that BVDV vaccines would not immunize calves that were passively immune to BVDV,297,298 recent studies have shown that immunization can occur with certain inactivated vaccines299 and with modified live virus vaccines (when the passive antibody titer against BVDV is 1:64 or below).278,300 When required, BVDV vaccines can be administered to young calves, with the possibility of gaining some degree of protection. In most calves the maternal antibodies against BVDV drop below the 1:64 level by 5 to 6 months of age. If an early BVDV problem does not exist, waiting to administer the first BVDV vaccine until at least 6 months of age increases the number of animals that respond to vaccination.
Among cows given modified live BVDV vaccines in the last trimester of pregnancy, 86% of the calves from seronegative cows301 and some calves (0%301 to 52%302) from seropositive dams were actively immune at birth. In addition, the calves’ level of passive immunity to BVDV was enhanced, in that seronegative cows seroconverted, and serum antibody titers were boostered in 52% of the seropositive cows. The rate of occurrence of BVD in neonatal calves was reduced.303 Administration of a modified live BVDV vaccine to seronegative cows after day 118 of gestation did not result in adverse effects.303,304 However, vaccination with a modified live noncytopathic virus vaccine before day 118 resulted in fetal resorption, abortion, congenital defects, and the birth of undersized, weak, persistently infected calves.305 Several authors have recommended vaccination of cows with a modified live BVDV vaccine during the last trimester of pregnancy.283 Two modified live BVDV vaccines are now labeled for use in pregnant animals. However, the label directions must be adhered to in order to ensure the safety of these vaccination schedules.
It is important to consider the epidemiologic, nonresponse rate to any vaccine when designing a BVDV vaccination program. Therefore even though modified live BVDV vaccines do not require a booster dose, a second dose may be advised to stimulate protection in animals that did not respond to the initial vaccination. BVDV vaccination programs should include the following features:
1
A virus isolation and cull program should be instituted, along with a vaccination program that includes administration of at least one dose of a modified live BVDV vaccine to all replacement animals.
2
Vaccination with killed vaccines should be increased to two or three times a year,
or a modified live BVDV vaccine should be given to open cows 3 weeks before breeding or turning in the bull.
3
The vaccines used should have been proved to stimulate protection against BVDV type 1 and type 2.
BOVINE RESPIRATORY SYNCYTIAL VIRUS VACCINES
John A. Ellis
BRSV is a prevalent paramyxovirus that can cause disease in cattle of all ages but that primarily affects calves in recurrent seasonal outbreaks.306-308 Clinical disease is characterized by pyrexia, coughing, and tachypnea, which can progress rapidly to severe expiratory dyspnea.307,309 BRSV is also considered one of the viral agents that predisposes animals to secondary bacterial infections in the bovine respiratory disease (BRD) complex; however, secondary infections often are absent in fatal BRSV-associated respiratory disease.310,311 As well, subclinical BRSV infection or mild respiratory disease resulting from BRSV infection in dairy cattle may have a negative impact on milk production.312
PARENTERAL VACCINES
Modified live virus and inactivated parenteral BRSV vaccines have been commercially available since the 1980s. Most of these vaccines are formulated in combination with other viral respiratory pathogens, including PI-3, BHV-1, and BVDV.168 The efficacy of commercial modified live virus nonadjuvanted and adjuvanted combination BRSV vaccines in protecting calves from severe clinical disease subsequent to experimental infection with a virulent field isolate has been demonstrated.313 Most vaccinated calves shed virus, but the peak virus titer was suppressed compared with unvaccinated controls. Viral clearance was coincident with the simultaneous appearance of mucosal antibody, cytotoxic T cells in the lungs, and anamnestic or primary serum antibody responses. In contrast, virus clearance in unvaccinated calves was coincident with the appearance of BRSV-specific cytotoxic cells before mucosal antibody was detected. Although administration of modified live virus BRSV vaccines by the intramuscular route to passively immune calves reportedly did not elicit mucosal memory IgA or serum antibody responses, or even prime for such responses,308 T cell responses have been demonstrated after parenteral vaccination in passively immune calves.314 Whether these responses correlate to a protective cell-mediated memory response when maternal antibodies decline has not been determined.
More recent investigations using the same challenge model in BRSV-seronegative calves have documented a similar efficacy of at least two combination inactivated vaccines containing BRSV formulated with different adjuvants.315,316 Clinical protection was associated with serum concentrations of BRSV-specific IgG as determined by ELISA315,316 and in one case315 the presence of IFN-γ—secreting BRSV-specific CD4+ T lymphocytes in the blood of vaccinated calves. There are conflicting data concerning the ability of commercial inactivated BRSV vaccines to override maternal antibodies and stimulate protective responses after parenteral immunization, which may be related to the vaccine formulation and adjuvant.317,318
Most experimental studies concerning the efficacy of commercial BRSV vaccines are consistent with previous field trials, demonstrating the safety and efficacy of parenterally administered vaccines.319,320 In addition, one study suggested the usefulness of combination vaccines in reducing the impact of subclinical BRSV infections.313
Protection against BRSV infection and disease is associated, at least in part, with an IgA response306-308; however, serum IgG acquired from either passive or active immunization can significantly reduce the severity of clinical respiratory disease that results from BRSV infection. Several studies have reported that the incidence and severity of disease in calves were inversely related to the maternal antibody titers.308,321,322 In the case of herd immunity, passive antibodies were detectable in 50% of the calves for 3 months after birth and were present in some calves until 7 months of age.323 Therefore administration of BRSV vaccines to cows in late gestation to booster colostral antibody titers is a rational strategy to deal with BRSV-induced respiratory disease in young calves (1 to 3 months of age) in problem herds, without having to be concerned about the immunizing potential of particular parenteral vaccines in young seropositive calves.
INTRANASAL VACCINES
Because BRSV is an endemic infection in most cow herds, most young calves will have BRSV-specific maternal antibodies, unless there is failure of passive transfer. As well, given the endemic nature of BRSV, it is likely that many calves are exposed to BRSV early in life and will contract BRSV-associated respiratory disease if their passive protection is poor or has waned. Considering the mixed results, to date, with parenteral administration of BRSV vaccines to young seropositive calves, the availability of a safe, effective intranasal modified live virus BRSV vaccine may represent the most effective means of immunizing young calves. In fact, it was demonstrated in the late 1980s that intranasal inoculation of tissue-culture—attenuated BRSV to passively immune calves primed for mucosal memory and an anamnestic IgA response subsequent to challenge.324 More recent similar studies with culture-attenuated BRSV confirmed this phenomenon.325 In addition, two recent studies suggest that currently available parenteral combination modified live virus vaccines can be efficacious if administered intranasally.326,327 Single intranasal administration of these vaccines to young calves primed for protective responses to subsequent experimental challenge. In one of these studies,326 results demonstrated that effective priming was achieved in BRSV-seropositive calves, confirming the previous observations with experimental single component BRSV vaccines.322,323 Recently an intranasal BRSV vaccine was licensed in Europe and the United Kingdom.
ADVERSE REACTIONS TO BOVINE RESPIRATORY SYNCYTIAL VIRUS VACCINES
The major factor hampering the development of a vaccine for human respiratory syncytial virus (HRSV) is the dramatic HRSV vaccine failure in the 1960s, in which vaccination with a formalin-activated (FI), alum-adjuvanted HRSV predisposed children to more severe disease after subsequent HRSV infection.328 Although this same disease-enhancing phenomenon has been demonstrated in some studies of experimental FI-BRSV vaccines in cattle,329,330 it is unlikely that modern manufacturing practices would use formalin inactivation for BRSV or other viral vaccines. Nevertheless, there are documented cases of apparent enhancement of BRSV-associated respiratory disease in cattle that received either inactivated or modified live virus vaccine in the field, indicating that some commercial vaccine formulations may stimulate potentially pathogenic immune responses in some cattle.329,330 The immunologic mechanisms responsible for vaccine-associated disease enhancement are not completely understood. The current hypothesis is that some BRSV vaccines, notably FI-BRSV, mainly prime Th2-like responses involving eosinophil influx into the lung and production of high concentrations of BRSV-specific IgE.329,330 Several studies have consistently documented a disparity in the type of antibody responses induced in cattle by modified live virus and inactivated BRSV vaccines.330-335 Parenterally administered modified live virus BRSV vaccines generally stimulate moderate to high concentrations of VN antibody in the serum, whereas inactivated vaccines stimulate high concentrations of partially neutralizing or nonneutralizing antibody. The data are conflicting concerning the prophylactic or disease-enhancing properties of these different types of vaccine-induced antibody responses.329,333 Alternatively, as was suggested in the case of disease enhancement after parenteral administration of a combination modified live virus BRSV vaccine,331 the timing between vaccination and infection could be a critical factor. This may be related to the stage of the immune response—specifically, a predominance of BRSV-specific IgM at the time of infection may somehow predispose to enhanced disease.
Currently there is little information about the duration of the protective response after vaccination; however, cattle can be experimentally reinfected by 35 days after initial infection with virulent BRSV even in the presence of circulating antibody.331 Nevertheless, as the epidemiology of BRSV-associated respiratory disease in the field indicates, disease caused by reinfection usually is less severe than that after the initial infection.306-308 These observations indicate that most cattle do not develop an allergic (IgE) response to BRSV from naturally acquired infections, or to most BRSV vaccines, in the vast majority of cases.
PARAINFLUENZA TYPE 3 VIRUS VACCINES
John A. Ellis
Parainfluenza virus type 3 (PI-3V) is a ubiquitous paramyxovirus in cattle populations worldwide.336,337 In uncomplicated experimental PI-3V infections, clinical signs of coughing, tachypnea, and fever have been observed from 4 to 12 days after infection.336 Although respiratory disease has been experimentally reproduced in calves infected with PI-3V, seroconversion has been demonstrated after outbreaks of respiratory disease,338 and PI-3V has been identified in the lesions of BRD complex at postmortem examination, the importance of this agent in the BRD complex remains controversial.336 Generally PI-3V is viewed as a potentiating agent in mixed infections, predisposing the animal to bacterial pneumonia by altering bacterial clearance in the upper and lower airways and by infecting both respiratory epithelia and alveolar macrophages.336
Currently five types of PI-3V vaccines are available commercially: (1) modified live virus intramuscular vaccines; (2) modified live virus, temperature-sensitive, intramuscular vaccines; (3) modified live virus intranasal vaccines; (4) modified live virus, temperature-sensitive, intranasal vaccines; and (5) inactivated virus vaccines. All PI-3V vaccines available in North America are combined at least with a BHV-1 vaccine. Currently, a single-antigen, modified live virus PI-3V intranasal vaccine is available in Europe. The efficacy of this formulation recently was demonstrated in a severe challenge model.339
Opinions are divided as to the relative importance of mucosal versus systemic immune responses in achieving protection from PI-3V—associated respiratory disease and, by extension, the comparative efficacy of intranasal and intramuscular vaccines. Some comparative studies339,340 reported that intranasal vaccination resulted in better protection against experimental challenge; others341 were unable to demonstrate any advantage to the use of one vaccine or route of administration over the other. A notable exception, however, was young calves with maternal antibodies, in which intranasal administration was thought to produce a more effective immune response. Passive antibodies may persist in calves until 8 months of age and may interfere with active immunization.342 Consequently, calves vaccinated parenterally before 6 months of age should be revaccinated after reaching 8 or 9 months of age.342
Although not experimentally documented, as in the case of BRSV, it is likely that, given the similar biology of PI-3V infection, a mucosal (IgA) response is necessary to prevent PI-3V infection but that passively (maternal) or actively acquired serum IgG is likely to mediate significant sparing of clinical disease subsequent to infection. The cell-mediated (cytotoxic T cell) response in the clearance of PI-3V is a poorly documented but probably important effector mechanism stimulated by modified live virus vaccines, as is the case with BRSV.
There is debate about the overall utility and economic benefit of using PI-3V (and BRSV) vaccines in the field.343,344 Much of the uncertainty undoubtedly is related to the difficulties involved in determining the relative importance of a particular agent in a multifactorial disease process such as BRD complex. Few studies344,345 address the economic impact of subclinical paramyxovirus infections in cattle. No recent studies (in the last 5 years) directly address the economic impact of inclusion of PI-3V in combination vaccines. One large study346 conducted under commercial feedlot conditions demonstrated an economic benefit to using a four-way combination modified live virus vaccine containing PI-3V together with BHV-1, BRSV, and BVDV versus a single component BHV-1 vaccine; however, it was not possible to determine which antigen(s) were responsible for disease sparing.
MANNHEIMIA (PASTEURELLA) HAEMOLYTICA, PASTEURELLA MULTOCIDA, AND HISTOPHILUS SOMNI (HAEMOPHILUS SOMNUS)
Anthony W. Confer
MANNHEIMIA HAEMOLYTICA VACCINES
M. haemolytica serotype 1 is the main species of bacteria responsible for the clinical signs and lesions of severe bovine fibrinous pleuropneumonia (shipping fever).347,348 The bacterium is a gram-negative commensal in the bovine nasopharynx, and after stress or viral infections the bacterium proliferates and is inhaled into the lungs where it stimulates a series of pathologic events leading to acute, severe fibrinopurulent inflammation and necrosis. The bacterium was renamed Mannheimia haemolytica because of substantial genomic differences between it and other members of the Pasteurella genus; however, it is still often referred to in commercial vaccines by its previous name, Pasteurella haemolytica.349 In this discussion the organism’s current and correct scientific name, M. haemolytica, will be used.
Important Mannheimia haemolytica Immunogens
M. haemolytica has numerous potential immunogens. Those with the greatest potential for stimulating immunity include capsular polysaccharide, lipopolysaccharide (LPS), outer membrane proteins (OMPs), iron-regulated OMPs, a secreted leukotoxin (LKT), a serotype-specific antigen, and several other secreted enzymes including neuraminidase, a sialoglycoprotease, and a bovine IgG1 protease.350,351 The central dogma of M. haemolytica vaccination is that immunity to the organism requires stimulation of antibodies that neutralize LKT and antibodies that bind to surface antigens allowing for complement-mediated killing and/or phagocytosis of the bacterium.352 There is no agreement as to what are the most important surface antigens; OMPs and iron-regulated OMPs are the major candidates based on many in vitro and in vivo studies.353-357 Capsular polysaccharide is theoretically an important surface antigen because it is the first surface molecule encountered by cellular and humoral components of the immune system, and its presence enhances M. haemolytica resistance to phagocytosis and complement-mediated killing.358 However, antibody responses to M. haemolytica capsular polysaccharide do not always correlate with resistance, and vaccination with purified capsular polysaccharide failed to protect against challenge.359,360 LPS is also a surface antigen; however, antibodies to M. haemolytica LPS failed to correlate with resistance to experimental challenge, and passive antibodies to M. haemolytica LPS were not protective in experimentally challenged calves.361,362
Commercial Vaccines
When a vaccination program for prevention of BRD is designed, four questions should be addressed:

Should
M. haemolytica vaccine be used?

What type of
M. haemolytica vaccine should be used?

How many doses of
M. haemolytica vaccine should be given?

When should
M. haemolytica vaccine be given?
The practicing veterinarian must answer these questions based on the cattle production situation; stocker, dairy, or feedlot management; interpretation of published literature; consultations with colleagues; and personal experience.
Numerous commercially available bovine biologics contain M. haemolytica antigens.363 M. haemolytica vaccines are often in combination with viral vaccines, H. somni or P. multocida bacterins, and occasionally Clostridium species biologics. Despite the various licensed M. haemolytica biologics available, formulations of M. haemolytica vaccines fall into one of eight categories, seven of which are nonliving vaccines. These are described by their manufacturers as follows: (1) bacterin with aluminum hydroxide adjuvant; (2) bacterin with water-in-oil adjuvant; (3) outer membrane extract; (4) bacterin-toxoid (LKT toxoid); (5) toxoid—cell-associated antigen; (6) adjuvanted toxoid (culture supernatant); (7) autogenous (herd-specific) bacterins produced from isolates submitted by practicing veterinarians; and (8) live streptomycin-dependent mutant. The last vaccine is the only currently licensed live M. haemolytica biologic. In the past, several live M. haemolytica vaccines were commercially available, and those vaccines showed potential efficacy; however, untoward side effects such as severe local and systemic reactions often occurred after vaccination.
Experimental Studies
Conventional formalin-inactivated, whole-cell, aluminum hydroxide—adsorbed M. haemolytica bacterins were the industry standard for many years; however, they stimulate low antibody titers to surface antigens, do not stimulate antibodies to LKT, and in experimental challenges or field trials were either ineffective in substantially enhancing resistance to pneumonic pasteurellosis or associated with increased disease and/or lesions.364 In contrast, experimental studies with M. haemolytica bacterins in water-in-oil adjuvants, outer membrane extracts, and recombinant iron-regulated OMP vaccines significantly enhanced resistance against experimental challenge even though they did not stimulate antibodies to LKT, indicating that the adjuvant used is probably of importance in M. haemolytica immunity.365-367 The other commercial M. haemolytica vaccines stimulate antibodies to LKT and to various surface antigens.368,369
Vaccine efficacy has been demonstrated primarily with experimental models of pneumonia using one of several challenge methods including direct M. haemolytica challenge via intratracheal, intrabronchial, or transthoracic routes or using a combination viral (usually BHV-1) and M. haemolytica challenge.350 The majority of published reports of experimental vaccination and challenge studies have used experimental vaccines and not commercial ones. There are few published reports of efficacy of individual commercial vaccines against experimental M. haemolytica challenge. For example, in one experiment, cattle were vaccinated with a commercial bacterin toxoid and compared with unvaccinated controls after a transthoracic M. haemolytica challenge.366 In a second experiment in the same manuscript, cattle were vaccinated with a commercial outer membrane extract vaccine and compared with control cattle after experimental challenge.366 Both vaccines significantly enhanced resistance against experimental challenge. In recent years, several studies have demonstrated that although vaccination with commercial vaccines can enhance resistance against experimental M. haemolytica challenge, addition of one of several M. haemolytica recombinant proteins, including LKT, sialoglycoprotease, or outer membrane lipoprotein, PlpE, enhanced efficacy of the commercial product.353,354,369,370 Direct comparisons of two or more commercial M. haemolytica vaccines after experimental challenge have rarely been published. In one such comparison between a commercial M. haemolytica bacterin toxoid and the live streptomycin-dependent mutant vaccines, the bacterin toxoid elicited the greatest serologic responses and significantly reduced lung lesions after experimental challenge.367 Calves receiving the live mutant vaccine had lesions that were not significantly lower than in control cattle. Demonstration of protection against experimental challenge, however, may not necessarily indicate that the vaccine will be efficacious against natural disease under field conditions.
Field Studies
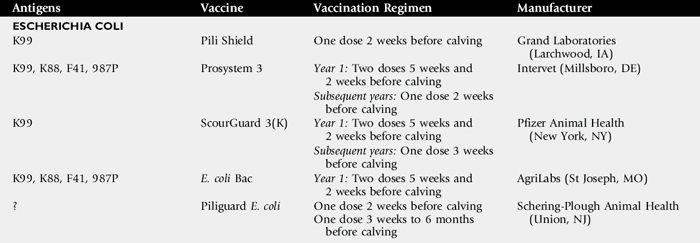
The number of published field studies using commercial M. haemolytica vaccines is limited. I am unaware of any published studies using autogenous M. haemolytica bacterins. In both dairy and beef cattle, maternal antibodies to M. haemolytica and P. multocida decline to undetectable levels between 30 and 90 days of age.369 Most calves subsequently spontaneously develop antibodies to these bacteria owing to natural exposure. There is substantial evidence that cattle entering a feedlot with preexisting serum antibody titers to M. haemolytica have less respiratory disease and fewer deaths than do those without serum antibodies.371 Therefore vaccination of cattle before shipment so that they can develop appropriate immunity is ideal, and determination of the appropriate time to vaccinate cattle with an M. haemolytica vaccine becomes critical.372-374 Manufacturers of M. haemolytica biologics usually recommend vaccination between 15 and 21 days before “weaning, shipping or exposure.”363 Although many of the currently available M. haemolytica biologics are licensed for only one injection, manufacturers recommend a booster if possible. However, administration of two doses of an M. haemolytica vaccine may not be practical for beef cattle. Shewen375 demonstrated that one of the reasons that one dose of M. haemolytica vaccine often stimulates adequate antibody response is because most cattle carry M. haemolytica in their nasopharynx and have a primed immune system that can produce a rapid anamnestic response to vaccination.
To determine the best time to vaccinate before shipping cattle, several studies have looked at how rapidly various commercial M. haemolytica vaccines induce antibody response and how long after vaccination do detectable antibodies remain. Two studies followed antibody responses to LKT and surface antigens in beef calves vaccinated with various commercial M. haemolytica vaccines and found marked differences both in rapidity and persistence of antibodies.354,376 With few exceptions, antibody responses reached their maximum 14 days after vaccination and had markedly waned by day 42. In another study, long-term antibody responses of cattle were followed after vaccination with three nonliving commercial M. haemolytica vaccines, a bacterin toxoid, outer membrane extract, and adjuvanted toxoid.377 Serum antibody responses to M. haemolytica surface antigens (all tested vaccines) and LKT antigens (bacterin toxoid and adjuvanted toxoid only) were at a maximum 2 to 3 weeks after vaccination, but most antibody responses had returned to normal by 6 weeks after vaccination. Revaccination 140 days after the initial vaccination resulted in rapid anamnestic responses that were usually higher than the initial responses. These data support manufacturer recommendations and indicate that if cattle are to be vaccinated with one of these M. haemolytica vaccines before shipment, vaccines should be given within 2 to 3 weeks of shipment to maximize antibodies at the time of shipment stress. If vaccination was performed before that time, a booster should be given before shipment.
Vaccination of cattle against pneumonic pasteurellosis on arrival at the feedlot is somewhat controversial because it may not allow enough time for development of solid protection before the period of highest morbidity.364 In addition, if cattle were vaccinated 2 to 3 weeks before shipment and had adequate antibody responses, antibody titers may be adequate, and revaccination may not be cost-effective. However, the vaccination history is not always known for beef cattle, and vaccination on entry to the feedlot is often practiced.372-374 Results in several field trials indicate that this practice can often afford some protection against shipping fever during the first 14 days in the feedlot.373 Selective use of M. haemolytica vaccines has also been advocated. Some feedlots designate cattle as high risk or low risk for respiratory disease. Managers may be more willing to vaccinate low-risk cattle, because high-risk cattle are either sick on arrival or can develop disease soon after entry into the feedlot.374 Therefore there would not be sufficient time for vaccination of high-risk cattle to stimulate immunity. However, low-risk cattle are less likely to develop disease soon after entry into the feedlot, and when they are vaccinated there is often adequate time for immunity to develop before a respiratory outbreak occurs.374
Perino and Hunsaker378 reviewed 10 published studies of several commercial live and subunit M. haemolytica vaccines with respect to their efficacies in field studies of feedlot cattle. Their report confirms that vaccination of cattle with newer generation M. haemolytica vaccines does not consistently reduce morbidity or mortality or increase weight gains. Of those studies, five showed positive outcomes based on reduced morbidity, mortality, or increased weight gain, whereas five studies demonstrated no positive outcome. Three of those studies demonstrating positive outcomes involved the same M. haemolytica bacterin toxoid given at arrival in the feedlot; however, two clinical trials with the same vaccine showed no significant differences when given at arrival and/or 3 weeks before shipment. In several field studies in which a positive outcome was demonstrated using a new generation M. haemolytica vaccine, economic benefits ranged from approximately $10 to $34 per head. Morbidity and mortality rates have been reduced by approximately 30% to 45% and 84% to 100%, respectively.379
Dairy and Veal Calves
Studies of M. haemolytica vaccinations in dairy and veal calves have been published less frequently than those in beef calves. In one study, dairy calves vaccinated at around 10 weeks of age with M. haemolytica toxoid failed to produce significant antibody responses to LKT or OMPs.380 In another study, vaccination of Holstein calves with adjuvanted toxoid (culture supernatant) at 2 to 4 weeks of age resulted in 50% or less of the calves seroconverting to M. haemolytica surface antigens, and those antibodies were in the IgM class.381 None of those vaccinates developed LKT neutralizing antibodies. Furthermore, many unvaccinated calves developed anti—M. haemolytica antibodies after 5 weeks of age, suggesting natural exposure to the organism. Low antibody responses in M. haemolytica—vaccinated young dairy calves probably indicate interference of vaccination by colostral antibodies.
With respect to protection afforded young calves by M. haemolytica vaccines, M. haemolytica toxoid— or live-mutant—vaccinated calves had incidences of respiratory disease similar to those of unvaccinated controls.382,383 In another study, an M. haemolytica bacterin toxoid was found to be less effective than a commercial streptomycin-dependent mutant M. haemolytica and P. multocida vaccine in reducing respiratory disease in veal calves.384 However, antibody responses to M. haemolytica and the causes of respiratory disease were not determined in the veal calf study. Failure of M. haemolytica vaccines to provide protection in dairy and veal calves could occur because P. multocida is usually the most common isolate from dairy calf pneumonia.
PASTEURELLA MULTOCIDA VACCINES
P. multocida, particularly serogroup A, serotype 3 isolates, is the second most common bacterium associated with pneumonia in beef cattle and the most common isolated from pneumonia in dairy calves.347 Although M. haemolytica has traditionally been the major pathogenic bacterium associated with shipping fever, recent studies suggest that the incidence of P. multocida in this disease has increased in beef cattle.348,372
The pneumonia produced by P. multocida is less acute and severe than M. haemolytica—associated pneumonia. The antigenic makeup of nonliving P. multocida vaccines presently available are proprietary and are described in the Compendium of Veterinary Products as bacterial extracts, cell-associated antigens, soluble antigens, and/or bacterins. Conventional formalin-inactivated, whole-cell, aluminum hydroxide—adsorbed bacterins have been the industry standard and are not considered highly efficacious.363 A commercial live streptomycin-dependent mutant P. multocida and M. haemolytica vaccine is available. Its efficacy is not well documented in the literature.
The immune mechanisms involved in resistance to P. multocida lung infections in cattle are poorly understood. There is more published work related to vaccines against P. multocida serogroups B and E, which cause hemorrhagic septicemia in cattle and water buffalo.385 Recent studies suggest that P. multocida OMPs or iron-regulated OMPs could be important immunogens for protecting cattle against pneumonia, and vaccination of calves with outer membrane preparations substantially enhanced resistance against experimental challenge.350,386,387 Although the P. multocida toxin, which is produced primarily by serogroup D isolates, is an important virulence factor and immunogen for P. multocida in atrophic rhinitis of swine, there is no evidence that this toxin is important in pneumonia of cattle, nor would it be beneficial to include the toxin in a vaccine for cattle.350
HISTOPHILUS SOMNI (HAEMOPHILUS SOMNUS) VACCINES
Haemophilus somnus’ name was recently changed to H. somni, which we will use in this discussion. H. somni is the cause of thrombotic meningoencephalitis (TME), septicemia, and reproductive disorders in cattle. In addition, it is the third most common bacterial isolate from beef cattle pneumonia in most epidemiologic surveys.347,348 Cases of H. somni—induced pneumonia are often associated with concurrent myocardial necrosis. Potential immunogens have been experimentally studied in H. somni and consist of lipooligosaccharide (LOS),388 several OMPs—including a surface protein that binds the Fc receptor of bovine immunoglobulin and is associated with serum resistance of pathogenic strains389—and iron-regulated OMPs.390 As with most gram-negative bacteria, H. somni LOS is a dominant antigen that stimulates an antibody response to the polysaccharide moiety after natural or experimental exposure. It is interesting to note that H. somni LOS has been demonstrated to exhibit phase variation in its epitopes (i.e., antigenic drift), thereby allowing the bacterium to escape the immune response.388 Currently there is no evidence that those anti-LOS antibodies are protective.391 Likewise, several OMPs and iron-regulated OMPs have been shown to be immunogenic in cattle. The major H. somni OMP is weakly immunogenic and shows strain antigenic variability.392 Their role in stimulating immunity is not known.
Several approved H. somni biologics are available, often in combination with respiratory viruses and Pasteurella species. All of the currently licensed H. somni biologics are formalin-killed bacterins with aluminum hydroxide as an adjuvant. Efficacy of H. somni bacterins has been generally favorable in stimulating protection against experimental pneumonia, against intravenous and intracisternal H. somni challenge as a model of TME, and against natural TME.354 Overall, vaccine-induced immunity has been best against experimental and natural TME.364,380,393 Using an experimental challenge model of H. somni—induced pneumonia, significant protection was afforded calves vaccinated twice with an H. somni bacterin.394 Resistance correlated with a high serum antibody response to the bacterium. One study demonstrated a reduced risk for respiratory disease in cattle that had high antibodies titers to H. somni on arrival in a feedlot.395 Therefore the potential exists for stimulating resistance to H. somni—associated pneumonia. In addition, commercial H. somni vaccines can stimulate IgE antibodies and thus potentially increase the risk for type I hypersensitivity.396
Under field conditions, commercial H. somni bacterins have had limited success in inducing protection against respiratory disease. Published reports have shown conflicting results. Perino and Hunsaker378 reassessed the results of three published commercial H. somni bacterin field trials and reaffirmed that this is the case. In one trial H. somni vaccination resulted in a reduced treatment rate when vaccinations were given at arrival and 21 days later.397 In another study, vaccination once with a commercial bacterin was associated with significantly more animals being treated for respiratory disease compared with unvaccinated cattle or those vaccinated twice at 21 day intervals.398,399 In another study, vaccination with a commercial H. somni bacterin on arrival at the feedlot was associated with no significant differences between the number of animals treated for respiratory disease compared with unvaccinated controls.400 In a recent study, partial reduction in feedlot respiratory disease was associated with vaccinating for H. somni, whereas a significant reduction in respiratory disease was associated with H. somni vaccine in combination with M. haemolytica or M. haemolytica vaccine alone.401 Ribble and colleagues,402 however, demonstrated reduced steer mortality after H. somni vaccination, but not heifer mortality.
BOVINE REPRODUCTIVE DISEASE VACCINES
Victor S. Cortese
Carol A. Bolin
As explained earlier, cows have a multilayered placenta, which leaves the fetus susceptible to infection. Infection of the placenta, inflammation of the ovary, death of the fetus, or disruption of the cervical plug all may cause abortion. Reproductive disease therefore is the most difficult disorder against which to achieve protection. Vaccination must minimize the amount or duration (or both) of the viremia or septicemia, or it must prevent the pathogen from moving through the cervix or crossing the placenta.
The reproductive diseases and protection against them through vaccination are areas of active research. With current research a vaccination program can be designed to aid in the control of reproductive diseases. Unfortunately, there is little or no research on the efficacy of many vaccines currently used to prevent reproductive disease. Because the causes of reproductive failure are so numerous (infectious agents account for only a small percentage), vaccination to prevent infectious reproductive losses many not appear to be effective. This often is a result of the fact that diagnostic testing has not been attempted or has not determined the cause of reproductive inefficiencies. A vaccination program may be inappropriately instituted when the cause is not infectious, or the current program may unfairly be deemed ineffective. A Neospora vaccine against Neospora-induced reproductive disease in cattle has been granted a license. Little published information is available on this product. Although safety has been shown, the efficacy is questionable.
The use of viral vaccines to help prevent reproductive diseases was discussed earlier in the chapter.
BRUCELLA ABORTUS VACCINE
Brucella vaccination has best shown the effectiveness of vaccination in controlling a reproductive disease. The successful control or even eradication of B. abortus in many areas of North America is a testament to the ability of a program involving testing, culling, and vaccination to control a reproductive disease. Vaccination with either strain 19 or strain RB51 Brucella has proved to be effective; however, many herd owners have stopped vaccinating against this disease as states have been declared Brucella free.
Abortions caused by B. abortus usually are seen after 5 months of gestation. Retained placentae and subsequent metritis usually follow. The abortion is caused by severe placentitis. Brucella infections have also been associated with a decrease in conception rates and an increase in services per conception. A higher number of dead and weak calves has also been demonstrated in infected herds. Orchitis or seminal vesiculitis or both may characterize infections in bulls.
Only heifer calves can be vaccinated for brucellosis. Both of the two licensed B. abortus vaccines are modified live bacterins, and vaccination of bulls may lead to orchitis.403 Legal use of the vaccines usually is confined to heifer calves 4 to 12 months of age, because vaccination of older animals with the strain 19 vaccine may lead to false-positive results on routine Brucella screening tests. Because the strain 19 vaccine may cause septicemia, clinical illness, and occasionally death,404 sick, unhealthy, or stressed cattle should not be vaccinated. The RB51 strain vaccine is an O antigen—deficient mutant of B. abortus strain 2308. The RB51 vaccine has three primary advantages:

Antibodies induced by this vaccine do not react with the serologic tests routinely done to diagnose
Brucella infections.

The vaccine can be used in adult cattle at a lower dosage under special circumstances and with the permission of the USDA.

The vaccine tends to cause less postvaccination fever and stress than the traditional strain 19 vaccines.
The long-term immunity conferred by Brucella vaccination is the cell-mediated type.405,406 Calfhood vaccination does not prevent a herd of cattle from becoming infected with B. abortus. However, it does largely prevent abortions and protects 65% to 75% of the cattle in the herd from infection while infected reactors are identified and slaughtered.407 For these reasons, in addition to vaccination, a control program should include testing and culling of all animals that test positive.
LEPTOSPIRA BACTERINS
Leptospirosis occurs worldwide and is caused by infection with the spirochete Leptospira. The pathogenic leptospires were formerly classified as members of the species L. interrogans; the genus has recently been reorganized, and pathogenic leptospires are now identified in seven species of Leptospira.408 As part of this reclassification the serovar names have remained the same, but some of the common leptospiral pathogens of cattle have different species names than before. The key changes for this discussion include the following: (1) L. interrogans serovar Grippotyphosa is now L. kirschneri serovar Grippotyphosa, and (2) the two types of serovar Hardjo have been formally split into two species; serovar Hardjo type hardjo-bovis (found in the United States and much of the world) is now L. borgpetersenii serovar Hardjo, and the less common serovar Hardjo type hardjo-prajitno (found primarily in the United Kingdom) is now L. interrogans serovar Hardjo.
Although traditionally associated with abortions, infection with various serovars of Leptospira are associated with a variety of clinical signs including severe systemic disease most often in young animals, decreased milk production, birth of weak calves, and infertility. In addition, infected cattle are known to present a risk of zoonotic transmission of the infection to humans. Many different serovars of Leptospira have been shown to cause reproductive failure and abortions in cattle. Of these serovars, Hardjo, Pomona, and Grippotyphosa409-412 are more common, with serovars Canicola, Icterohaemorrhagiae, and Bratislava occasionally implicated. The epidemiology of infection of cattle with these serovars differs, with cattle serving as the reservoir or maintenance host for serovar Hardjo and as an incidental host for the other serovars. In general, maintenance host infections are associated with a high prevalence of infection, a poor immune response, and long-term infection and shedding, whereas incidental host infections are characterized by low overall prevalence of infection with epidemics recognized, a vigorous immune response, and short-term infection and shedding. These differences in the epidemiology of leptospirosis caused by different serovars of Leptospira require different strategies for prevention.
Leptospirosis can cause abortion storms in which a high number of cattle may abort within a short period. There may also be an increased number of stillbirths and births of premature and weak calves during these periods.413 Although serovars Pomona and Grippotyphosa tend to cause abortions in the last trimester of pregnancy, serovar Hardjo can cause abortions at any stage of pregnancy. Abortions usually are caused by fetal infection and subsequent death of the fetus, although placentitis may also occur. Serovar Hardjo can also colonize the oviducts and uterus,410,414,415 diminishing fertility. After an initial serovar Hardjo infection, cattle may remain infected and shed the spirochete for long periods,416,417 whereas infection and shedding of the others serovars is relatively brief.
Current bacterins generally contain combinations of leptospiral serovars Pomona, Grippotyphosa, Canicola, Icterohaemorrhagiae, and either L. interrogans serovar Hardjo (hardjo-prajitno) or L. borgpetersenii serovar Hardjo (hardjo-bovis). At the time of this writing a monovalent L. borgpetersenii serovar Hardjo (hardjo-bovis) vaccine is also available. There has been considerable debate in recent years regarding the efficacy of leptospiral bacterins for cattle. The bacterins have label claims that indicate they are to be used as an “aid in the prevention of disease.” Therefore these bacterins should be expected to decrease the severity of clinical signs, including abortion, associated with such infection. In general, the evidence supports such claims for serovars for which cattle are an incidental host, that is, serovars Pomona, Grippotyphosa, Canicola, and Icterohaemorrhagiae. Protection mediated by these bacterins for these serovars is thought to occur because of induction of antibodies directed against the LPS on the surface of the Leptospira.418 However, vaccination does not always prevent infection and leptospiruria caused by serovar Pomona.419-421
The efficacy of bacterins for prevention of infection, leptospiruria, and clinical signs associated with serovar Hardjo infection is significantly more controversial. The evidence that the efficacy of traditional serovar Hardjo vaccines is less that optimal includes induction of a relatively poor antibody response in vaccinated animals, the common presence of Hardjo infection in herds despite routine vaccination, and experimental trials that did not demonstrate protection afforded by these traditional vaccines for prevention of infection, colonization of the renal or genital tract, or transplacental infection on challenge with Leptospira borgpetersenii serovar Hardjo (hardjo-bovis).422-424 In these studies, cattle were not protected from infection despite the induction of antibody directed against serovar Hardjo LPS. Further investigation and evaluation of other serovar Hardjo bacterins has led to a hypothesis that CMI may play a role in protective immunity against serovar Hardjo in cattle.425-429 Newer bacterins for serovar Hardjo have been introduced in monovalent and polyvalent formats, and there is evidence that these bacterins provide significant protection of cattle against infection, tissue colonization, shedding, and transplacental infection.424,428-430 Other new bacterins for serovar Hardjo are also entering the marketplace, but as of this writing extensive data regarding the performance of these products are not available in the literature.
Some Leptospira bacterins are labeled as single initial dose products, but a booster dose is recommended approximately 1 month after the first dose.418Leptospira bacterins must be administered by intramuscular or subcutaneous injection. Although some manufacturers specify revaccination at 12-month intervals, this DOI has been questioned, and more frequent revaccination often is needed to control Leptospira abortions.419,420,422 One of the newer serovar Hardjo bacterins has documented a 1-year DOI for this component, but this DOI has not been documented for other Hardjo vaccines or for the other serovars, making vaccination every 6 months a reasonable recommendation in many circumstances. Nevertheless, because leptospiral abortions are uncommon during the first half of pregnancy, it may be possible to use an annual vaccination schedule (serovar Hardjo excepted) in seasonally calving herds such as beef herds. Cattle in such herds can be vaccinated when they are 2 to 4 months pregnant, usually at the time that pregnancy is diagnosed, and protected through the balance of the pregnancy with a single annual dose.419
Prevention of leptospirosis caused by serovar Hardjo requires a somewhat different approach. Prebreeding vaccination of heifers that is effective in managing reproductive sequelae of other types of leptospirosis may be too late to prevent the consequences of serovar Hardjo infection. Heifers exposed very early in life may remain infected well into the time of breeding. Therefore efforts to control serovar Hardjo infection should be targeted at preventing the initial infection and is best done by vaccinating young stock well before the time when they are mixed with older animals. In addition, bulls can carry serovar Hardjo and transmit the infection quite readily during breeding.431 Therefore bulls should be fully included in efforts to control serovar Hardjo infection by vaccination.
BOVINE GENITAL CAMPYLOBACTERIOSIS VACCINES
Originally classified as Vibrio, Campylobacter fetus subsp. veneralis causes a venereal infection of cattle. The bacteria are introduced during natural breeding by infected bulls or by artificial insemination (AI) with infected semen. Bulls usually are infected by breeding with infected cows, but contact with infected bedding may also be a cause. Older bulls (over 4 years of age) are more likely to be infected. After deposition in the vagina, the bacteria rapidly colonize the vagina and cervix, and in 25% of these cows the bacteria are found in the oviducts. The organism can persist for months after infection of these sites. It has been shown that fertility never returns to normal in some infected animals, and some animals may be permanently sterile because of the damage caused by salpingitis.
Vaccination with Campylobacter vaccines has been shown to be effective in protecting heifers even when vaginal cultures test positive for the bacteria.432 It appears that the uterus is very resistant to the bacteria after vaccination. Studies have demonstrated improved breeding efficiency in vaccinated herds.432 Vaccination of bulls with oil-adjuvant vaccines not only prevents infection of bulls for up to 1 year433 but also aids in prevention of mechanical transfer of organisms during natural service.434 Furthermore, vaccination with two doses has been shown to be effective at clearing infections from carrier bulls.435,436
Vaccination
Use of C. fetus bacterins is recommended in all breeding herds that use bulls, even if only on selected cows. In heifer herds using virgin bulls or in 100% AI-bred herds, vaccination against Vibrio organisms is not necessary.
Several different C. fetus vaccines are available, including oil-adjuvanted and aluminum hydroxide—adsorbed types. Oil-adjuvant C. fetus bacterins have proved to be more effective437 and to provide longer lasting protection after a single dose.438 Unfortunately, oil-adjuvant vaccines cause localized granuloma formation and fibrosis at the site of injection. This may cause visible blemishes, which may be objectionable in registered stock or show cattle. Administration no earlier than 4 months before the breeding season is preferred.438 When aluminum hydroxide—adsorbed C. fetus bacterins are used, a priming dose should be administered at least 6 weeks before the immunizing (booster) dose, and the booster should be administered 10 days before the beginning of the breeding season.437 After administration of an aluminum hydroxide—adsorbed bacterin, serum antibody concentrations peak rapidly and decline precipitously, falling to susceptible levels by 4 weeks after one dose or 11 weeks after two doses.439 Some aluminum hydroxide—adsorbed bacterins do not require an initial booster.
Campylobacteriosis (vibriosis) is most effectively controlled when all breeding-age animals, including bulls, are included in the vaccination program.434 Vibrin* is the only C. fetus bacterin available in the United States that has been evaluated in bulls.432 Two 5-mL doses are administered to breeding bulls at 4-week intervals beginning 8 weeks before the start of the breeding season.432 In subsequent years a single booster dose is administered 4 weeks before the start of the breeding season.433 This dosage is 2½ times that recommended for vaccination of cows.
BOVINE TRICHOMONIASIS VACCINES
Bovine trichomoniasis is a venereal infection of cattle caused by the protozoal agent Tritrichomonas foetus. Early in the course of the disease, abortions with pyometra may be seen in 5% of infected cows. These abortions occur early in gestation.440 However, infertility is the most common sign, with long interservice intervals.441,442 Early embryonic death is followed by a period of conception failure. Some natural resistance develops after infection, but carrier cows may be an important component of the epidemiology of this disease. In rare cases a cow may be left sterile after an infection because of uterine destruction.443
The efficacy of Tritrichomonas vaccines is questionable, 444-446 but the vaccines do appear to reduce actual reproductive losses.447 Heifers, cows, and breeding bulls should be vaccinated twice at 2- to 4-week intervals, the second dose given 4 weeks before the beginning of the breeding season.448 Subcutaneous administration is recommended. In subsequent years a single annual booster vaccination should be given 4 weeks before the beginning of the breeding season.
In a problem herd, trichomoniasis vaccination must be coupled with other control measures, such as culturing, culling, and treatment to effectively control the disease.
NEONATAL CALF ENTERIC DISEASE VACCINES
Gerald E. Duhamel
Neonatal calf enteric diseases (NCEDs) can have a devastating impact on the profitability of beef cow-calf and dairy operations. In addition to mortality, medical, and labor costs, NCED can significantly reduce body weight of beef calves at weaning and performance of replacement dairy heifers.449
Several well-characterized infectious causes of NCED have been described.450 Some of the more common infectious agents are group A RVs, coronavirus (CV), enterotoxigenic E. coli, salmonellae, and Cryptosporidium species.451-457 Sporadic outbreaks of necrohemorrhagic enteritis affecting 3- to 10-day-old calves have been associated with infection with C. perfringens type C.458 Other less well-characterized viruses (group B RV, calicivirus, torovirus, astrovirus), bacteria (enterotoxigenic Bacteroides fragilis, attaching-effacing enteropathogenic and enterohemorrhagic E. coli, Enterococcus durans), and protozoans (Giardia duodenalis) also have been associated with NCED.
Because of the continuous calving and constant introduction of replacement heifers on dairy farms and the continuous flow of susceptible calves on veal calf operations, infections caused by more than one agent should be suspected in these types of operations.451-457,459-461 In addition to different infectious agents, mixed infections with different groups and serotypes or genotypes of viruses and bacteria also can occur. As a consequence, the severity, duration, and spread of clinical disease associated with complicated infections are usually greater than with a single agent, and control may be more challenging. When scours occurs in a herd that has been vaccinated against enteric diseases, possible contributing factors should be considered, including concurrent infection with pathogens not present in the vaccine (e.g., Cryptosporidium species, salmonellae, nongroup A RVs, other pathogens) and suboptimal management, including poor control of environmental contamination. Under these conditions, strategic improvement in husbandry practices might be sufficient to reduce the infectious threshold for NCED and obtain the full benefit of pathogen-specific vaccination. In any situation, thorough laboratory diagnostic investigation is essential in order to correctly identify the primary cause of NCED and any other possible contributing factors and to serve as a basis for appropriate control strategies for the present and future calf crops.
Management practices and risk factors associated with the development of NCED are sufficiently different between dairy and beef herds that control measures can be tailored according to the type of production. For example, in dairy herds, high turnover of older cows and continuous introduction of replacement heifers pose a higher risk for introducing new strains of enteric disease pathogens in a susceptible population than in closed beef herds. Similarly, the continuous calving and the proximity of susceptible calves on dairy farms create an ideal environment for recirculation of enteric disease agents back into the adult population. This in turn may increase and broaden herd immunity such that the efficacy of a vaccine in these herds might be enhanced, partly because of the booster effect vaccine can have on preexisting immunity. Beef cattle herds, in contrast, are generally relatively closed, and a susceptible calf population is present only for a relatively short period annually during the calving season. Introduction of new pathogens or new strains of enteric disease agents, particularly during the calving season, can have a devastating effect because of the potential for exposure of a high number of susceptible animals over a relatively short period of time. Conversely, after an outbreak, herd immunity might be more stable and the occurrence of NCED might be reduced during the following calving seasons.
Under both types of production systems, vaccination for NCED is rarely successful without reasonably good management programs and sanitation aimed at providing adequate intake of protective colostrum and minimizing environmental contamination. General herd health management steps that can affect the success of a vaccination program for NCED should include a calving cow-calf care plan aimed at minimizing exposure of newborn calves to infectious agents that can overwhelm innate or passively acquired immunity. This is accomplished by eliminating all potential means of NCED transmission and by reducing the load and duration of exposure to pathogens present in the calving environment by following eight simple recommendations:
1
Establish a sound biosecurity program, including purchase of replacement animals only from reputable sources and a complete ban on the introduction of foster calves, which pose a risk of introducing new NCED agents.
2
Provide adequate protein and energy feed, including proper concentrations of micronutrients (e.g., copper and selenium), and abundant clean drinking water throughout gestation and calving so that heifers and cows are within acceptable body condition score at calving and therefore produce an adequate volume of high-quality colostrum and milk, and calves are healthy and vigorous at birth, with a fully developed and functional immune system.
4623
Avoid crowding, and keep calving area and equipment clean, dry, and protected from elements, particularly winter wind. Separate feeding and bedding grounds should be available in the calving area. Similarly, to minimize infectious disease transmission, separate feeding and manure handling equipments should be used.
4
Limit the spread of infectious disease agents from younger, more susceptible, and potentially shedding pairs to the main herd by segregating first-calf heifer pairs from the mature cow herd during the calving season until the youngest calf is at least 4 weeks old. Also, because of potential problems associated with nursing in first-calf heifers, newborn calves in this group should be monitored closely for colostrum intake and early detection of failure of passive transfer (e.g., total serum protein concentration below 5 at 12 hours of age) so that rapid intervention can be implemented.
462-464 Because first-calf heifers are at a competitive disadvantage with mature cows, segregation of pregnant first-calf heifers also ensures free access to feed and water at all times.
5
Ensure that each calf ingest approximately 10% of its body weight in colostrum within the first 6 hours of birth, and if a calf is unable to nurse naturally or is at risk for failure of passive transfer of maternal immunity, then force-feed fresh or frozen colostrum from the calf dam’s or from a mature cow in the same herd or from a neighboring herd.
6
Disinfect all equipment used for feeding colostrum and treating sick calves routinely; this includes such items as nipple bottles, esophageal probes, and balling guns.
7
Move cow-calf pairs away from the calving area as soon as possible after birth in dairy herds, whereas in beef herds, segregate calves by age by scheduled movement of dams that have not calved to new calving pastures—the so-called “Sandhills Calving System” (
http://vetext.unl.edu/stories/200703050.shtml).
8
Quickly isolate scouring calves from healthy calves in dairy herds, or sick cow-calf pairs from healthy pairs in beef herds.
462Giving calves a healthy start maximizes their genetic production potential while reducing the costs and labor associated with treatment of sick animals.
ROTAVIRUS AND CORONAVIRUS VACCINES
RVs and CVs are ubiquitous in the cattle population; most adult cattle have VN serum antibodies.465-469 RV infections are widespread in both dairy and beef cow-calf productions, whereas infection with CV most often occurs as sporadic outbreaks of severe diarrhea in beef cow-calf herds or chronic low-grade diarrhea in dairy and veal calf operations. In addition to causing NCED, CV has been associated with winter dysentery in adult cattle470 and respiratory tract infections in calves471 and as a contributing infectious cause of BRD complex in feedlot cattle.472 Fecal shedding of RV and CV is common among adult cattle,473-476 which provides an immediate source of virus challenge for naive newborn calves and allows persistence of these agents at the herd level.
Although only one type of CV is known to cause NCED,477 different subtypes of CVs can be identified on the basis of minor genomic and antigenic differences.477-481 On a herd basis, however, affected calves can display a range of clinical signs from enteric only to mixed enteric and respiratory signs. A recent molecular epidemiologic investigation based on comparative analyses of the gene encoding the S glycoprotein, a major structural protein of bovine CV, revealed (1) identical CV strains in different animals from the same herd and from paired nasal and fecal samples from the same animals, suggesting herd outbreaks are associated with a single strain circulating among susceptible cattle, (2) identical CV strains in affected cattle from different herds in the same region, suggesting transmission between herds, and (3) different CV strains in cattle affected during different outbreaks occurring over several years in the same herd, suggesting herd outbreaks are associated with the introduction of new strains in a recovered herd.482
Currently RVs are classified into at least seven distinct groups, A through G.483 Although RVs that belong to groups A, B, and C have been found to naturally infect cattle,476,483-487 group A RVs are by far the prevalent type.485,487 Members of the group A RVs are further classified according to antigenic and genetic differences in their outer capsid proteins, G and P.488-490 Because both of these proteins are involved in neutralization of infectivity in vitro and protection in vivo,491-493 consideration of the G-P configuration of RVs is critical to development of an effective vaccination program for prevention of NCED.
Although a good correlation has been found between the antigenicity of the G proteins and their corresponding gene sequences, a similar relationship has not been found for the P proteins. Therefore 14 different G serotypes, which correspond to 14 G genotypes, have been identified among group A RVs, whereas 10 serotypes identified on the basis of the antigenicity of the P proteins have been assigned to 20 genotypes when compared on the basis of the nucleotide sequence of the P protein genes.490 At least eight distinct G serotypes or genotypes (G1, G2, G3, G6, G8, G10, G11, and untypeable) are known to infect cattle in the United States494,495; G6 is the most widespread, and G8 and G10 account for lower percentages of infections.469,495-500 Conversely, RVs with any of four P serotypes or genotypes—P6(1), P7(5), P8(11), and untypeable—are known to infect catte, with P7(5) being the prevalent type.495,498,501
The genome of RVs is composed of 11 gene segments that can be exchanged among RV isolates when animals are infected by more than one virus type at the same time.489 Therefore mixed infections produce virus with a genetic makeup derived from either parental strain by a mechanism called gene reassortment. Because the G and P proteins independently are involved in generation of specific VN antibodies, reassortment of these genes during mixed infections can generate new progeny viruses that can evade what once was a protective immune response, thus allowing persistence of RVs in susceptible populations.500 Therefore the potential occurrence of RVs carrying any of 36 possible antigenic configurations (4 Ps × 8 Gs = 32 potential types) underscores the limitation of vaccination programs and also the importance of sound management practices designed to minimize exposure by limiting environmental contamination.
Vaccination Programs
Two approaches are widely used in an attempt to protect calves against RV and CV infection and diarrhea. The most common approach involves passive protection of the suckling neonate by transfer of high levels of specific VN colostral antibodies induced by parenteral vaccination of the pregnant dam.467,502-510 The mechanism of passive protection with this approach is attributable to the continuing presence of an amount of specific VN antibodies in the intestinal lumen sufficient to neutralize infectious virus before infection of the intestinal villous enterocytes.465,507,511-515 The continuous presence of VN antibodies in the gut lumen also may reduce the severity of the disease if infection has already developed. In addition, some of the VN colostral antibodies that are absorbed into the bloodstream are secreted onto mucous membranes and may provide local immunity later in life.516 However, colostral transfer of immunity is less efficient in ruminants than in other species; high concentrations of maternal antibodies are present for less than 3 days postpartum, and these concentrations often fall below protective levels within 1 week of parturition.*
A delicate balance exists between passive protection afforded by lactogenic immunity and development of the calf’s own innate and adaptive local immunity to RV and CV. Because most calves’ level of resistance to the adverse clinical effects of RV and CV infection increases with age, disease caused by these agents can be controlled under certain circumstances by continuous hand-feeding of fresh, frozen, or fermented colostrum from vaccinated cows throughout the first 3 to 4 weeks of life.† The goal with this approach is to allow development of subclinical infection while the milk VN antibody concentration is partially protective.504,515,521 Conversely, lactogenic immunity might interfere with development of adaptive immunity, or some calves might not be exposed to RV and CV infection until after milk antibody concentrations have fallen below protective levels, leaving the calf fully susceptible to infection.502,504,522
An RV and CV vaccine for parenteral administration to pregnant cows (Calf-Guarda) first became commercially available in 1979. This vaccine contained a single live attenuated G6:P6(1) strain of group A RV. However, because VN antibody concentrations in the colostrum and milk of vaccinated cows were low,467,468,504,518 this product later was replaced by a formulation containing inactivated RV and CV (ScourGuard 3 [K]b). In early 2000 an inactivated RV and CV vaccine (Scour Bosd), which contains three strains of group A RV, was licensed for use in pregnant cattle in the United States. More recently, in 2005 an inactivated RV and CV vaccine (Guardiane) containing two strains of group A RV and two strains of CV was licenced for subcutaneous administration in pregnant cattle in the United States. A significant advantage of the later vaccine for dairy cattle is the administration schedule, with the second booster dose administered before the dry period. In late 2006 the single G6 RV ScourGuard 3[K]/Cb vaccine was upgraded to contain G6 and G10 strains of group A RV, and the window for administration of both formulations to pregnant cows was extended (ScourGuard 4[K]/Cc).
Vaccination of pregnant cows with inactivated RV and CV vaccine can raise the level of VN antibodies in the colostrum.467,502-511,523 Although there are reports of successful protection of calves against NCED by parenteral vaccination of pregnant cows with RV and CV,* negative results also have been reported.531 Failures of this vaccination strategy generally are attributable to failure of passive transfer of colostral antibodies to the calves or overwhelming infectious virus challenge. An alternative explanation might be that the serotypic specificity of the passively transferred maternal antibodies may affect the efficacy of the RV vaccine.532
Information on protective immunity against infection of calves with group A RVs having the same or a different G-P serotype configurations as the vaccine strain is incomplete.496,523,533-537 Although immunity to RV appears to be G-P serotype specific with some strains,496,523,533,536 there is some indication that immunity directed against certain strains can neutralize in vitro496,537 and protect challenge-exposed calves in vivo496,536 against RV strains with different G-P configurations. Also, parenteral vaccination of seropositive cows with a single strain of RV can elicit serum VN antibodies to a broad spectrum of RV serotypes and genotypes, suggesting that this strategy may provide a means of enhancing passive protection against a range of potential RV serotype and genotype challenges.524,534,535 However, a difference in the P protein between the vaccine RV and the infecting RV was suggested as the basis for failure of the inactivated monovalent RV and CV vaccine in beef cow-calf herds.538,539
Although antigenic and genotypic variation has been documented among bovine CV infections,477-481 reduced protection of nursing calves after challenge with a strain of bovine CV different from the vaccine strain has not been demonstrated. Consequently, presence of more than one strain of bovine CV in certain vaccine might not be justified at this time.
Another approach for prevention of NCED caused by RV and CV involves oral vaccination of calves with a modified live virus vaccine (Calf-Guarda), which contains an attenuated G6:P6(1) strain of group A RV.540-552 The mechanism of disease prevention with this vaccine is unknown, but interference with infection by virulent virus followed by development of secretory IgM and IgA and/or CMI in the intestinal mucosa have been proposed.522,543 To achieve adequate protection, the manufacturers recommend that the vaccine be given immediately after birth, before the calf has nursed. This regimen might be applicable to calves whose dams have not been vaccinated. However, because the colostrum of most heifers and cows contains some level of VN antibodies arising from natural exposure,‡ administration of colostrum should be delayed for several hours after vaccination to avoid inactivation of the vaccine virus. Under commercial conditions, it is nearly impossible to administer vaccine within minutes of birth or to effectively regulate the intake of colostrum in relation to the time of vaccination. Therefore infection before vaccination, neutralization of the vaccine virus by colostral antibodies, and overwhelming challenge with infectious virus shed by unvaccinated, diseased calves might explain a lack of efficacy of this approach.505,545-548 This is evident from the data obtained in vaccine efficacy evaluation studies in which only a portion of the calves on a farm or a ranch were vaccinated in double-blind or odd-even—day vaccination trials.517,549,550 When all calves were either vaccinated or not vaccinated in sequential comparisons, morbidity and mortality rates from NCED were significantly reduced by this vaccination strategy,511,540-542,549,540 but the design and statistical validity of the latter kinds of trials have been questioned.531,546
Although not recommended by the manufacturer, the oral attenuated vaccine has been administered to calves with unknown immune status that were raised as veal calves or replacements in heifer development operations.551 Under these circumstances, vaccination might provide active immunity and protection against potential virus challenge when calves from several different sources are commingled.
ROTAVIRUS AND CORONAVIRUS VACCINATION PRODUCTS
SCOURGUARD 4(K)/C
ScourGuard 4(K)Cc consists of inactivated bovine G6 and G10 strains of group A RV and a bovine CV strain combined with K99 E. coli bacterin and C. perfringens type C toxoid. Two doses of vaccine should be administered by intramuscular injection to pregnant cows and heifers 6 to 9 weeks precalving. A second dose should be given 3 to 6 weeks before the expected calving date to first-calf heifers and mature cows not previously vaccinated. A single annual booster dose should be administered 3 to 6 weeks before each subsequent calving.
SCOURGUARD 3(K)/C
ScourGuard 3(K)/Cb consists of inactivated bovine G6:P6(1) strain of group A RV and a bovine CV strain combined with K99 E. coli bacterin and C. perfringens type C toxoid. Two doses of vaccine should be administered by intramuscular injection to pregnant cows and heifers 6 to 9 weeks precalving. A second dose should be given 3 to 6 weeks before the expected calving date to first-calf heifers and mature cows not previously vaccinated. Cows that have not calved within 40 days after administration of the last vaccine dose should be revaccinated. A single annual booster dose should be administered 3 to 6 weeks before each subsequent calving.
Scour Bos
Scour Bosd consists of inactivated bovine group A RV strains G6:P6(1), G8, and G10 and a CV strain combined with bacterins from four E. coli K99 serotypes and C. perfringens type C toxoid. The vaccine should be administered by deep intramuscular injection into the neck of pregnant cows and heifers up to 10 weeks before the expected calving date. A booster dose of Scour Bos RV and CV vaccine must be given approximately 6 weeks later during the first year. Only one annual dose up to 10 weeks before calving is required thereafter.
Guardian
Guardiane consists of inactivated bovine group A RV G6 and G10 strains together with “type 1” and “type 3” bovine CV strains combined with enriched K99 pili from E. coli and type C and D toxoids of C. perfringens. The vaccine should be administered by subcutaneous injection of pregnant cows and heifers up to 12 weeks before the expected calving date. A booster dose of vaccine must be given approximately 3 to 6 weeks later during the first year. Only one annual dose up to 5 to 7 weeks before calving is required thereafter.
Calf-Guard
Calf-Guarda is a modified live bovine G6:P6(1) strain of group A RV and bovine CV recommended for oral vaccination of newborn calves. The vaccine should be administered immediately after birth, before the calf has nursed. As indicated earlier, interference by maternal antibodies may limit the efficacy of the vaccine.
BACTERIAL SCOURS VACCINES
Victor S. Cortese
Charles A. Hjerpe
ENTEROTOXIGENIC ESCHERICHIA COLI (CALF SCOURS) BACTERINS
Most cases of scours caused by E. coli occur within the first 72 hours of life. More than 90% of these cases are caused by E. coli containing the K99 pilus attachment fimbriae.552-554 These strains also may have other fimbriae types, such as F41 and F1 (type 1).552,555 Villous attachment and colonization by strains of enterotoxigenic E. coli having multiple fimbriae types appear to be effectively prevented by vaccination with bacterins that have only a single pilus antigen in common with challenge strains.552,555 However, disease occasionally may be caused by non-K99 E. coli.556 The attaching and effacing E. coli types, which may cause disease at 7 to 21 days of age, often do not produce K99 pilus and are not protected by current E. coli K99 bacterins.557,558 This apparent lack of efficacy may be seen with other non-K99 E. coli; therefore typing of E. coli isolates from scours cases may be important in determining which vaccine to use.
Because E. coli scours occurs so early in life, the newborn calf does not have enough time to derive protection from vaccination. Therefore control of E. coli infection has been aimed at controlling calf exposure to the pathogens and vaccinating the cow to increase the colostral antibody levels against this pathogen (i.e., usually against the K99 pilus antigen).554 Cows are vaccinated in late gestation to ensure high concentrations of anti-K99 colostral antibodies. When colostrum from vaccinated cows is fed to newborn calves, the antibodies act in the small intestine to block the pili from binding to specific receptor sites on the brush border of small intestinal villous enterocytes.552,559 E. coli bacteria that are prevented from attaching to the jejunal and ileal villi are carried into the large bowel by peristalsis. In this way, colonization of villi and production of enterotoxin are avoided. By the time they are 48 to 96 hours old, most calves are highly resistant to infection.552,560 Thus feeding calves colostrum with a high concentration of antibodies against K99 antigen, even though restricted to the first day of life, often is sufficient to prevent the disease.555 Passive circulating humoral antibodies, which are absorbed into the bloodstream from the calf’s gut, are thought to play little or no role in immunity to neonatal enteric disease caused by enterotoxigenic E. coli.552 In dairies these vaccines are only as good as the colostrum program that is in place.
Nearly all strains of enterotoxigenic E. coli that have been isolated from neonatal calves have K99 pili.561 Currently there is no evidence that bacterins with multiple pilus antigen types are more effective than those with only one pilus antigen type, as long as the vaccine and the challenge strains share a common pilus antigen.552 However, vaccines with multiple pilus antigens are more likely to have at least one of the antigens found on the virulent challenge strain of E. coli.
Escherichia coli Vaccination Programs
The general recommendations for use of E. coli bacterins are summarized in Table 48-13. E. coli bacterins are offered as single-antigen vaccines and in combination with other antigens. Oil-adjuvant E. coli bacterins are administered by intramuscular injection in a single dose 2 weeks to 6 months before calving, and administration is repeated -annually.561 Non—oil-adjuvant E. coli bacterins are recommended for intramuscular or subcutaneous injection in two doses administered at a 2- to 4-week interval, with the second dose given 3 to 6 weeks before calving. In subsequent years a single booster dose should be administered 3 to 6 weeks before calving. E. coli bacterins do not protect calves that do not ingest sufficient amounts of colostrum sufficiently soon after birth. Also, because this protection is based on passive immunity, high challenge levels may overwhelm the finite amount of antibodies present.
SALMONELLA VACCINES
Salmonella outbreaks can be caused by myriad Salmonella serotypes and strains (see Chapter 32). These bacteria may cause diarrhea or septicemia or both. Most outbreaks in cattle have historically involved either Salmonella Dublin or Salmonella Typhimurium; therefore all but one of the commercial Salmonella vaccines contain either Salmonella Typhimurium alone or Salmonella Typhimurium and Salmonella Dublin. More recently, S. Newport and other group C Salmonella have greatly increased in importance. All currently licensed products are formalin-inactivated, whole-cell, aluminum hydroxide—absorbed bacterins, except for a modified live S. Dublin vaccine,* and the newest vaccine against bovine Salmonella, the siderophore receptor vaccine.† The gram-negative core antigen vaccines may also provide some protection from morbidity and mortality associated with salmonellosis.
A modified live, genetically altered (via deletion) S. Dublin vaccine is marketed as Entervene-d for parenteral use in young calves. Research indicates that it is effective in calves 2 weeks of age and provides some cross-protection to challenge with virulent S. Typhimurium.561a,561b As with many gram-negative vaccines, adverse reactions to this vaccine have been reported. Anecdotal reports of effective oral use in calves younger than 2 weeks of age indicate that this route may help avoid some adverse reactions associated with injection of an endotoxin-containing product.
Killed Salmonella bacterins can produce measurable antibody responses to bacterial proteins in calves and mature cattle. However, calves vaccinated with a killed bacterin are not able to produce anti-LPS antibodies until 12 weeks of age,562 and optimum responsiveness does not occur until 1 year of age.563 Most controlled studies in which calves were vaccinated with a killed Salmonella bacterin and orally challenged reported lack of protection.564-566 One small study that reported good protection after vaccination of 3- to 6-week-old calves with two doses of killed bacterin used intramuscular challenge.567 Another study in 6-month-old cattle found that a single intradermal dose of heat-killed Salmonella Dublin protected against intravenous challenge.568 Vaccination of cattle 3 months of age or older with two doses of killed Salmonella bacterins is likely to be useful for preventing salmonellosis. A newer Salmonella vaccine (SRP) using subunit technology has been conditionally licensed. Although little has been published on this vaccine, the clinical impression is that it has been successful in controlling disease caused by multiple different serotypes of Salmonella in some cases.
Vaccination of adult cows, with passive transfer of antibody to calves through colostrum, frequently is used in dairies to control calfhood salmonellosis. Controlled trials evaluating passive protection have produced mixed results, with some indicating lack of protection566,569 and others demonstrating some protection in very young calves (5 days old).570 Vaccination of dry cows may be useful for helping to control salmonellosis in calves younger than 3 weeks of age but is probably minimally effective for controlling salmonellosis in calves older than 3 or 4 weeks. Anecdotal reports exist of protection against salmonellosis from vaccinating young calves several times with a gram-negative core antigen vaccine (Endovac-Bovi*). Another gram-negative core antigen vaccine, a J5 E. coli bacterin (J Vac†, J-5‡), reduced the mortality rate from naturally occurring salmonellosis in dairy calves vaccinated at 3 and 17 days of age.571
GRAM-NEGATIVE, CORE ANTIGEN BACTERINS
All genera and species of gram-negative bacteria contain a common set of gram-negative core antigens, which are present in the deeper layers of the bacterial cell wall.572 Endotoxin from gram-negative bacteria is thought to play an important role in the production of the clinical signs, biochemical and hematologic alterations, and pathologic lesions associated with a wide variety of bovine diseases caused by gram-negative bacteria,572 including coliform mastitis (caused by E. coli, Klebsiella species, or Enterobacter aerogenes), Pasteurella bronchopneumonia and fibrinous pneumonia, and salmonellosis. Gram-negative core antigen vaccines are designed to reduce the severity of clinical signs associated with gram-negative sepsis and endotoxemia. They offer breadth against all gram-negative infections.
General Considerations
Three gram-negative, core antigen, oil-adjuvant vaccines are currently marketed. Two use the J5 Rc-mutant strain of E. coli. This strain of E. coli lacks the serotype-specific O-chain surface antigens that ordinarily prevent deeper cell wall antigens from contacting the host immune system and stimulating production of antibodies against gram-negative core antigens.572 The third vaccine uses an Re-mutant strain of Salmonella Typhimurium.
Theoretically, gram-negative core antigen vaccines would be expected to reduce the severity of disease manifestations but not the rate of occurrence of disease. In four different clinical trials, however, the incidence of clinical coliform mastitis in dairy cows was reduced by 69%, 72%, 80%, and 82%, respectively, by vaccination with a J5 E. coli vaccine.573-575 These data are consistent with the hypothesis that antibodies induced against gram-negative core antigens assist in both the destruction and the removal of intact bacteria and in the neutralization of endotoxin. In the trial in which clinical coliform mastitis was reduced by 82%, however, vaccination did not reduce the incidence of subclinical intramammary coliform infections that were present at the time of calving.575
Vaccination Programs
Recommendations for use of these bacterins to prevent coliform mastitis and scours are summarized in Table 48-13. Cows should not receive any other vaccine containing gram-negative organisms (Pasteurella, Salmonella, Brucella, Campylobacter [Vibrio], H. somnus, E. coli, or M. bovis bacterins) within 5 days of vaccination with J5 bacterin.
A J5 E. coli bacterin also has successfully reduced morbidity and mortality in experimental Salmonella Typhimurium -infections in calves.576 However, because of the short half-life of passively acquired antibodies stimulated by gram-negative core antigen vaccines,577 vaccination of dry cows is unlikely to be an effective control measure for salmonellosis in calves except when calves are exposed to and infected with salmonellosis in the first week or two after birth. Salmonellosis in dairy calves often occurs after 2 weeks of age. However, when vaccinated at 3 and 10 days of age with an oil-adjuvant J5 E. coli bacterin, calves do develop strong antibody responses by 17 days of age; this strong antibody response occurs even in the presence of passively acquired antibodies to gram-negative core antigens.578 Consequently, a combined program in which both dry cows and neonatal calves are immunized against gram-negative core antigens appears to be a promising approach for reducing the severity of salmonellosis in neonatal dairy calves.
In a field study, vaccination of healthy dairy calves with an oil-adjuvant J5 E. coli bacterin at 3 and 17 days of age reduced the mortality rate from salmonellosis.571 (In this study, the dams were not vaccinated.) In addition, the morbidity rate from undifferentiated respiratory disease was significantly reduced by 9%.576 In a parallel study in poorly nourished calves, however, vaccination actually increased the cumulative 60-day mortality by 113%.571 These kinds of vaccines may prove more effective for reducing the severity of Pasteurella infections of the lung when used in older calves.
CLOSTRIDIAL VACCINES
J. Glenn Songer
Clostridia produce acute and frequently fatal disease, with pathogenesis often mediated by toxic proteins.579 Prevention is often based on immunoprophylactic amelioration of the effects of these molecules. However, the ready availability of inexpensive, efficacious bacterins, toxoids, and bacterin-toxoids has not eliminated clostridial infections. Accurate diagnosis remains an important component in management of clostridial diseases.580,581
Immunization against clostridial diseases can be complicated by the development of “site reactions,” leading to trimming at slaughter.582 These problems are exacerbated by the multivalent nature of many modern products and have stimulated the biologics industry to seek a new paradigm for preparation and delivery of immunoprophylactic products. Approaches have included concentration of the antigen into a smaller dose and use of alternate adjuvants; recombinant proteins, delivered by conventional means, by application of “slow-release” media, or by in vivo expression from attenuated bacterial delivery systems, will likely be a major focus of effort.
CLOSTRIDIUM CHAUVOEI (BLACKLEG) BACTERINS
Blackleg is not uncommon, in spite of the long-term availability of generally effective bacterins. Ingestion is probably the most common route of exposure in cattle, and dormant spores seeded to skeletal muscle germinate when muscle damage provides appropriate conditions. Affected animals have fever, anorexia, depression, and lameness, with extensive dry and emphysematous to edematous, hemorrhagic, and necrotic lesions. Diagnostically, it is important to distinguish between blackleg and malignant edema.
As with other histotoxic clostridial infections, vaccination against blackleg is universally advocated, especially in cattle under the age of 2 years. Dogma is that protection arises from the immune response to a heat-labile soluble antigen, but C. chauvoei produces alpha-toxin and several other toxic factors, which may be equally important targets.583,584 Recommendations for immunization are summarized in Table 48-14.
Table 48-14 General Considerations for Use of Clostridial Seven-Way and Eight-Way Bacterin-Toxoids*
| |
Coverage Designation* |
| Protection, by Species |
Seven-Way |
Eight-Way |
| Clostridium chauvoei |
x |
x |
| Clostridium septicum |
x |
x |
| Clostridium novyi B |
x |
x |
| Clostridium sordellii |
x |
x |
| Clostridium perfringens type C |
x |
x |
| Clostridium perfringens type D |
x |
x |
| Clostridium perfringens type B† |
x |
x |
| Clostridium haemolyticum (novyi type D) |
|
x |
| Dose (mL) |
2–5 |
5 |
| Recommended boosters |
After 2–4 weeks for C. sordellii and C. perfringens C and D; after 5–6 months for C. novyi type B and C. haemolyticum; calves vaccinated initially at <3 months of age should be revaccinated at 4–6 months (or at weaning) |
| Comments |
No use <21 days preslaughter |
CLOSTRIDIUM SEPTICUM (MALIGNANT EDEMA) BACTERINS
Wound infections caused by C. septicum (malignant edema)585 usually follow direct contamination of a traumatic wound, including genital tract infections after mismanaged deliveries. Infection spreads along fascial planes, and lesions proceed from warm and pitting to crepitant and cold. Death commonly occurs in less than 24 hours. Braxy is a form of enteric infection that occurs not uncommonly in calves.586 Diagnosis is often by use of a fluorescent antibody test.587
A single immunizing dose of C. septicum bacterin yields adequate protection, but annual booster vaccination is recommended in high-risk situations588 (see Table 48-14). Vaccines elicit antibody responses to both somatic and toxin antigens, and recent findings suggest a central role for alpha-toxin.589,590
CLOSTRIDIUM NOVYI TYPES A AND B (BIGHEAD AND INFECTIOUS NECROTIC HEPATITIS) BACTERINS
Clostridium haemolyticum (Clostridium novyi type D) (Bacillary Hemoglobinuria) Bacterins
C. novyi toxigenic types A and B cause myonecrosis in humans (gas gangrene) and domestic animals (bighead of sheep) and infectious necrotic hepatitis (black disease of sheep and cattle).591 The hallmark lesion is edema, likely resulting from vascular damage caused by alpha-toxin. Dormant spores are often found in Kupffer cells but germinate when liver injury provides appropriate conditions. Type C strains are nontoxigenic and therefore nonpathogenic.592
Clostridium haemolyticum (C. novyi type D) beta-toxin mediates the pathogenesis of redwater, usually in well-nourished animals at least 1 year of age. Liver damage caused by migrating flukes encourages germination of dormant spores in Kupffer cells. Dissemination of beta-toxin via the bloodstream results in intravascular hemolysis, hemorrhage, and hernoglobinuria,593,594 and death ultimately results from anoxia. A vaccination program is essential for herds pastured in endemic areas. When light exposure to fluke metacercariae is expected, a single annual dose of bacterin should be administered to all cattle over the age of 6 months, before they are pastured in the spring. With heavy exposure to flukes, a booster dose should be administered in season. The prominent roles of alpha- and beta-toxins suggest that they may find use in second-generation immunoprophylactic products.
CLOSTRIDIUM BOTULINUM (BOTULISM) AND CLOSTRIDIUM TETANI (TETANUS) TOXOIDS
Botulism is caused by C. botulinum neurotoxins, which block acetylcholine release from cholinergic nerve endings.595,596 Type C is most common in cattle in the United States. Direct contamination of feeds by the organism sometimes leads to intoxication, but it is more commonly associated with an animal carcass in the feed. Clinical signs include incoordination, flaccid paralysis, and difficulty in swallowing; respiratory paralysis eventually causes death.597,598
Toxoids of botulinum toxins can be employed for immunoprophylaxis, but vaccination is usually practiced only in populations at immediate risk, such as beef cattle grazed on phosphorus-deficient range land.599 Feeding of poultry litter poses as similar problem, in that it may contain animal remains.
Spores of C. tetani originate in soil and are usually introduced traumatically to animal hosts, where they germinate and produce tetanus neurotoxin.600 Tetanus can develop in dairy cows as a postparturient complication and in calves after castration by the elastrator method.601 Toxin moves retrograde, binding to presynaptic axonal terminals and resulting in muscular tremor and increased stimulus response; continued motor neuron hyperactivity causes sustained tetanic spasms in the innervated muscles and then permanent rigidity. Death is due to respiratory failure.
Acquired resistance to tetanus is based on circulating antitoxin, and widespread vaccination with toxoid has dramatically lessened the impact of tetanus on animal production. Neonatal passive immunity is followed by active immunization with toxoid after 2 to 3 months. Boosters are commonly recommended at 1- to 5-year intervals. Passive immunotherapy is directed toward neutralization of preformed toxin, although it is much more effective when used prophylactically than therapeutically. Universal vaccination is not usually recommended as a cost-effective means for control of tetanus.
CLOSTRIDIUM PERFRINGENS TOXOIDS
C. perfringens causes a wide variety of diseases in domestic animals, and those of greatest importance affect the gastrointestinal tract602 (Table 48-15) (see Chapter 32). Type B infections are apparently extraordinarily rare in the United States, but it has been speculated that their pathogenesis can be explained by additive or synergistic effects of beta- and epsilon-toxins. Type C strains multiply rapidly in the gut of neonates, and in this relatively trypsin-free environment, beta-toxin produces local hemorrhage and necrosis, as well as systemic effects.603 Type D strains fill intestinal niches opened by sudden dietary changes, and epsilon-toxin in circulation damages the central nervous system and other systems distant from the gut.604 Type E causes hemorrhagic enteritis in calves, and its virulence is based apparently on the action of iota-toxin.605
Table 48-15 Diseases of Cattle Caused by Clostridium perfringens
| C. perfringens Type |
Diseases In Domestic Animals |
Major Toxins |
| A |
Myonecrosis, enterotoxemia, abomasitis, possible sudden death |
Alpha |
| B |
Neonatal hemorrhagic enteritis |
Alpha, beta, epsilon |
| C |
Neonatal hemorrhagic or necrotic enterotoxemia |
Alpha, beta |
| D |
Enterotoxemia |
Alpha, epsilon |
| E |
Enterotoxemia |
Alpha, iota |
The current enigma is type A infections. Although long accepted as causes of lamb enterotoxemia,606 fowl necrotic enteritis, and enteritis in dogs and horses, they are increasingly recognized as causes of enteritis in piglets and calves. Little is known about the pathogenesis of type A enteric infections, but type A strains are commonly found in cases of tympany, abomasitis, and abomasal hemorrhage and ulceration in calves.607,608
Most agree that routine vaccination against type C enterotoxemia is required only in herds in which the disease has been documented. The usual practice is to vaccinate the dam, providing passive immunity via colostrum. Initial immunization should be followed by a booster after 3 to 4 weeks, with the second dose (and subsequent annual boosters) administered approximately 2 weeks before calving. Type D enterotoxemia occurs sufficiently infrequently in cattle that many believe vaccination to not be cost-effective. No commercial products are licensed in the United States for use against infections by strains of types A and E, and production of autogenous toxoids or bacterin-toxoids has become quite common; anecdotal evidence suggests remarkable efficacy in many cases. Similar products have been produced from strains of type E. These should be used with the awareness that, unlike beta- and epsilon-toxin concentrations in commercial products, alpha- and iota-toxin concentrations in autogenous toxoids may not be optimal.
CLOSTRIDIUM SORDELLII BACTERINS
C. sordellii is commonly found in feces of domestic animals, as well as in the soil, and is occasionally isolated from fatal myositis, liver disease, and sudden death in cattle. Edema in the subcutaneous tissues and along fascial planes of muscles and subendocardial hemorrhage are common signs. The organism produces numerous toxic or putatively toxic substances, foremost of which is a toxin that resembles toxins A and B of Clostridium difficile.609 Immunization is achieved by administration of multiway bacterin-toxoids (see Table 48-13).
MISCELLANEOUS BOVINE RICKETTSIAL, BACTERIAL, AND VIRAL DISEASE VACCINES
Derek A. Mosier
ANAPLASMOSIS
Anaplasmosis is a vector-borne or mechanically transmitted disease caused by the rickettsia Anaplasma marginale.610 The disease occurs worldwide and is most prevalent in tropical and semitropical areas. Anaplasmosis is the only major tick-borne disease of cattle in North America, being enzootic in the southeastern and some midwestern and western states and sporadic in the northern states and Canada.610-612 In enzootic areas with adequate numbers of arthropod vectors, most adult cattle become naturally immune through repeated exposure. Maternal antibodies protect calves until they also become subclinically infected and develop immunity. Disease is more severe in older cattle than in calves, and nonimmune older cattle are particularly at risk when they are moved into an endemic area.610 Susceptibility to disease occurs when there is a lack of arthropod vectors to maintain natural infection and immunity or when a nonimmune adult is introduced into an enzootic area. In these situations or when vector or environmental conditions suggest an increased risk of disease, vaccination can be beneficial.
No widely marketed commercial vaccines against anaplasmosis were available as of 2006 in North America. A killed vaccine* is available in some states including Florida, Louisiana, Texas, Oklahoma, Arkansas, California, Oregon, Nevada, Tennessee, Mississippi, Indiana, Iowa, Illinois, and Kansas and in Puerto Rico. The vaccine employs the same A. marginale antigens and purification procedure that was used for the discontinued Plazvax† vaccine. The killed vaccine is not USDA licensed, but it is USDA approved for use as an experimental vaccine. The vaccine has reportedly been used successfully in cows at all stages of pregnancy without an episode of neonatal isoerythrolysis. In endemic areas the vaccine is recommended for use just before the onset of the vector season. Vaccine-induced immunity does not generally occur until 2 weeks after administration of the second dose of an initial series or 2 weeks after a booster dose in previously immunized cattle.612 Vaccination does not prevent infection or clinical disease and does not eliminate A. marginale from a herd, but it does reduce the severity and incidence of disease.610,611 Inactivated vaccines could be used in conjunction with oxytetracycline in the face of outbreaks to provide both temporary and more prolonged protection.611
A sheep-passaged, modified live vaccine‡ has been used in California and Latin America.613 Because this vaccine causes mild clinical disease, it has limited use for vaccination of mature susceptible cattle.610,613 If the vaccine is administered to cattle over 2 years of age, anemia, severe clinical disease, and death may occur, especially in bulls and heavily lactating cows.613 The vaccine is recommended for use in healthy cattle between 1 month and 2 years of age and is most commonly administered to 7- to 24-month-old cattle in herds in endemic areas. Concurrent use of certain antibiotics or other live or modified live virus vaccines is contraindicated.
A live vaccine* derived from A. marginale subsp. centrale, a less pathogenic species or subspecies of A. marginale, is used in some countries but not in North America.614,615 This vaccine consists of standardized and frozen red blood cells from splenectomized cattle that were infected with A. marginale subsp. centrale. The vaccine is recommended for use in 4- to 9-month-old cattle; older cattle have an increased risk of severe vaccine-induced disease. The vaccine produces mild disease but protects against subsequent severe disease caused by A. marginale. Immunity is considered long term, or possibly for life if subsequent natural exposure occurs to ensure the development of durable immunity.
Immunity to A. marginale is proposed to involve humoral responses to a variety of major surface proteins and enhanced macrophage phagocytosis and killing, both mediated by IFN-γ—producing CD4+ T lymphocytes.610,612,616-618 Inadequate protection from vaccines during field use can result from antigenic variability of the organism, geographic differences in the organism that result in a lack of cross-reactivity between A. marginale strains, and weak immune responses to protective A. marginale antigens.610,615,617,618 Purified native, recombinant, and tick culture—derived A. marginale immunogens and DNA vaccines are being investigated for possible commercial use in the future.615,617,619-622
INFECTIOUS BOVINE KERATOCONJUNCTIVITIS
The most common infectious agent associated with infectious bovine keratoconjunctivitis (IBK) is M. bovis.623 Vaccination is most effective when done before fly season in herds with a history of problems. Certain breeds, such as Herefords and Hereford crosses, are particularly susceptible and may benefit from vaccination.623 Commercial vaccines used to help prevent the disease consist of inactivated cultures of various strains of M. bovis.624 Some products recommend two doses given 3 weeks apart for initial vaccination, beginning as early as 3 weeks of age to no earlier than 5 months of age. Other products recommend a single dose administered 3 to 6 weeks before the predicted onset of the disease season, with annual vaccination thereafter. M. bovis bacterins are also available in combination with seven-way clostridial bacterin-toxoids.625 Although multivalent M. bovis bacterins can provide some protection in field use, efficacy varies depending on the M. bovis strains present in the bacterin and those responsible for disease.623,626-630 Seven different disease-producing serogroups of M. bovis are recognized based on differences in pili, and there is variable cross-reactivity between serogroups.624 Furthermore, pilin gene rearrangements and pilin-type switching can allow M. bovis to switch from expression of one type of pilus antigen to another, making it difficult to predict what serogroup(s) may be necessary for protection.624,627,631 Therefore monovalent bacterins are generally ineffective in field use, and multivalent bacterins provide neither consistent nor reliable protection.624,626,627,629 Vaccines must incorporate pili from all major serogroups or conserved, immunogenic portions of all serogroups to provide optimum protection.624,627-630,632 Experimental recombinant vaccines containing cloned pili of various serogroups have demonstrated some promise for future vaccines.624,626,628 Another immunogen considered important for protection is M. bovis hemolysin/cytolysin.623,624 Experimental vaccines containing hemolysin/cytolysin preparations have shown some efficacy in experimental trials.623,624,633,634 Other potential immunogens include iron-regulated OMPs, proteases, fibrinolysins, and phospholipases.623,624 Vaccine-induced protection is correlated predominately with a suitable lacrimal (IgA) mucosal immune response and not with serum antibody levels to M. bovis antigens.623,624 Therefore an antigen delivery system that enhances mucosal immunity is an important feature of an effective vaccine. The presence of other agents contributing to IBK (e.g., Moraxella [Branhamella] ovis, Mycoplasma bovoculi, or BHV-1) should also be considered when there is poor M. bovis vaccine efficacy.635 Autogenous M. bovis vaccines have not been consistently effective against the disease.623,636 Use of modified live virus vaccines for IBR is contraindicated in the presence of an outbreak of IBK because it may exacerbate the IBK.623
STAPHYLOCOCCAL MASTITIS
Staphylococcus aureus is considered one of the most important causative agents of bovine mastitis.637 Vaccination against S. aureus may be beneficial in dairy herds that have an existing mastitis problem.637,638 However, vaccination in well-managed dairy herds with a low level of staphylococcal mastitis may not provide much economic benefit.638 Staphylococcal bacterins contain antigens from multiple strains or serotypes of S. aureus.639,640 The recommended vaccination protocol is two doses given 2 weeks apart followed by revaccination at 6-month intervals. Vaccination can start at 6 months of age, and one of the semiannual doses should be given 3 to 4 weeks before calving. Vaccination with S. aureus bacterins does not generally eliminate disease but can substantially reduce clinical mastitis and the incidence of subclinical and chronic staphylococcal infection.638,641-644 Vaccination may be more effective in heifers because of their initial lower basal immunity compared with older cows.642 The benefits of immunity induced early in life include the abilities to clear the organism and to resist chronic infection on initial natural exposure.645 Vaccination during the dry period may be more effective than vaccination during lactation.637 In some but not all studies, vaccination has reduced somatic cell counts in milk.642,643,645 Vaccination in combination with antimicrobial therapy has been successfully used to eliminate chronic staphylococcal mastitis.646
In considering the use of staphylococcal vaccines, the prevalence of various pathogens that can cause mastitis must be considered. For mastitis caused by S. aureus, differences between the S. aureus strains in vaccines and the strains specifically responsible for the disease may diminish the efficacy of the vaccine.637,640,644 Experimental trials suggest that more effective vaccines may be derived by stimulating immune responses to combinations of S. aureus capsular polysaccharide.647 A vaccine based on technologies used for human staphylococcal vaccines that incorporates capsular polysaccharide from the three S. aureus serotypes most commonly associated with mastitis stimulated immunologic parameters necessary for protection.648 These studies may form the basis for more effective vaccines in the future.
In herds in which other pathogens are a major cause of mastitis, S. aureus vaccines may be of minimal benefit.638,641 Other important causes of bovine mastitis include Streptococcus species (e.g., Streptococcus uberis, Streptococcus dysgalactiae, and Streptococcus agalactiae) and coliform bacteria (e.g., E. coli).637,649 Streptococcus agalactiae vaccines generally are not protective, but other Streptococcus species are responsive to vaccination.649 Experimental vaccines composed of bacterial proteins derived from S. uberis and S. dysgalactiae reduced somatic cell counts compared with controls after challenge.650,651 E. coli J5 vaccines have also been used to successfully reduce the severity of mastitis (reviewed elsewhere in this section).
ANTHRAX
Anthrax is an acute, highly fatal disease caused by B. anthracis.652 Vaccination has proven to be an effective means of controlling the disease in endemic areas and in the face of outbreaks.653,654 Bovine anthrax vaccines are derived from the live toxigenic, nonencapsulated spore vaccine developed by Sterne and consist of spores suspended in a diluent containing saponin and glycerin.655 Annual vaccination of livestock in areas of endemic anthrax is recommended 4 weeks before outbreaks are expected. A single dose generally provides adequate immunity, but a second dose given 2 to 4 weeks after the first is often recommended.656 Cattle should not be vaccinated within 42 days of slaughter. Antibiotics should not be administered within 7 days of vaccination to avoid interference with in vivo growth of the vaccine organism. Vaccination in the face of an outbreak does not protect all cattle, but the spread of infection and the number of new cases generally declines within 10 days.654,657 Localized subcutaneous edema commonly develops at the injection site within 24 hours; it may last for several days and is sometimes severe.653 Since the intentional release of B. anthracis via the mail system in 2001, there has been increased interest in technologies for the development of an efficacious, long-acting human vaccine for anthrax.653,658-662 Although some of these technologies could hold promise to reduce some of the localized side effects of the current bovine vaccine, it is unlikely that a new bovine vaccine will match the safety and efficacy of the Sterne strain vaccine in the foreseeable future. All anthrax outbreaks should be reported to local regulatory and public health officials, and appropriate guidelines for vaccination should be followed, including quarantine and vaccination of all susceptible livestock on affected and surrounding premises.
INTERDIGITAL NECROBACILLOSIS (FOOT ROT)
Interdigital necrobacillosis (foot rot) in cattle results from interdigital infection with Fusobacterium necrophorum with lesser contributions from Prevotella (Bacterioides) melaninogenica and sometimes Dichelobacter (Bacterioides) nodosus and other bacteria.663-665 Commercial F. necrophorum bacterins to aid in the prevention of foot rot (and hepatic necrobacillosis) are available for use in cattle. Recommendations for initial vaccination are two doses given 3 to 4 weeks apart, followed by annual revaccination. Vaccination is also recommended when endemic conditions exist or when exposure is imminent. The efficacy of F. necrophorum vaccines is not clear, but some benefit has been demonstrated in experimental studies and field trials.666,667 Vaccination is especially recommended in herds that have a high incidence of disease.666 Protective immunity most closely correlates with the level of anti-LKT antibodies.668,669 A leukotoxoid vaccine composed of cell-free supernatant from a high LKT—producing strain of F. necrophorum was effective in reducing experimental hepatic necrobacillosis669 and presumably would have some benefit against interdigital F. necrophorum infection. An autogenous vaccine containing D. nodosus reduced the severity of interdigital dermatitis but not of necrobacillosis.670
PAPILLOMATOUS DIGITAL DERMATITIS (FOOTWARTS)
Papillomatous digital dermatitis, or footwarts, can be a serious problem in dairy cattle.671,672 The disease is characterized by ulcerative to proliferative digital lesions that most often occur in replacement heifers and younger cows after introduction into a milking herd.671-673 Risk is greatest in larger dairy breeds in herds of greater than 500 head. The cause of the disease is uncertain, but Treponema species—like spirochetes and flexible, gram-negative rods (Serpens species) have been incriminated.673-676 Commercial bacterins are available for use as preventatives and/or aids to treatment and consist of killed cultures of Serpens species or Treponema species organisms. The recommendations are for three doses administered subcutaneously at 3- to 4-week intervals, followed by revaccination every 4 to 6 months. Company field trials report reduced onset of new infections and sometimes more rapid resolution of existing infections in vaccinated cattle. Another study of clinically affected cattle in which vaccination was combined with treatment with topical lincomycin showed no significant improvement in vaccinated cows compared with unvaccinated ones.677 The high recurrence rates of natural infection suggest that immunity to the disease is short-lived or weak.673
RABIES
Rabies is a highly fatal, zoonotic neurologic disease caused by a rhabdovirus.678 Routine vaccination of cattle is not common in most situations. However, vaccination may be cost-effective in rural areas of Latin America, where vampire bats are important sylvatic vectors.679,680 In endemic areas vaccination of valuable cattle or herds may be a reasonable precautionary measure.678 This is particularly true in situations in which cattle are in frequent contact with human beings, in order to reduce the anxiety of animal workers and minimize the likelihood of human exposure. Currently licensed rabies vaccines for cattle contain inactivated, cell culture—derived virus.681 The recommended regimen is initial vaccination at 3 months of age followed by annual vaccination thereafter. The duration of protective neutralizing antibody levels after initial vaccination can vary.682 Therefore some experts have suggested that a second booster dose be given either 1 month after initial vaccination or at 6 months of age.682,683 Subsequent annual revaccination induces strong anamnestic responses that persist for 1 year or longer.682,683 In Latin America, modified live vaccines are sometimes used.680 However, these do not stimulate the same level of immunity as do the inactivated virus vaccines. A Capripoxvirus vector expressing rabies virus glycoprotein has shown promise in providing long-term protection against rabies (and lumpy skin disease) and may be a cost-effective mechanism for rabies control in cattle in some developing countries.684
FIBROPAPILLOMAS (WARTS)
Fibropapillomas (warts) are manifested in a variety of forms and locations, each caused by a specific bovine papillomavirus (BPV).685,686 Lesions associated with papillomaviruses can occur in the epidermis of the head, face, neck, and legs (BPV-1 and BPV-22), upper alimentary and urinary tracts (BPV-4), teats and udder (BPV-1, BPV-3, BPV-5, and BPV-6), and genital epithelium (BPV-1).685-688 Immunity after infection or vaccination is virus type specific and is induced by viral structural proteins.685,687-690 Therefore the efficacy of both autogenous and commercial vaccines depends on which viral antigens are incorporated into the vaccine and which virus type is responsible for the disease. Vaccines containing BPV-1 and BPV-2 are generally effective for prevention but not treatment of disease caused by the homologous virus.687 Vaccines usually are ineffective for treatment or prevention of disease caused by BPV-3 and BPV-5.687 Vaccination with recombinant capsid proteins of BPV-4 was effective in preventing papillomas after experimental challenge with BPV-4.691 The interpretation of the response to vaccination against fibropapilloma can be complicated by spontaneous regression of some lesions.685 Lesions associated with BPV-1 and BPV-2 usually spontaneously regress within 1 to 12 months, whereas lesions caused by BPV-3 and BPV-5 do not normally spontaneously regress.687,692
Commercial vaccines consist of inactivated, virus-laden tissue extracts derived from bovine papillomas.693 The recommended regimen is an initial dose divided and given in at least two different sites, followed by a second dose in 3 to 5 weeks. Vaccination should continue for at least 1 year after elimination of disease from the herd. Autogenous vaccines can be made by homogenization and inactivation (0.3% formalin) of excised wart tissue, followed by dilution of the homogenate in physiologic saline and filtration through gauze. Three 1- to 5-mL intradermal injections given at 1-week intervals are recommended. Vaccination is most commonly used with valuable animals destined for competitive shows or for overseas sale.687 Vaccination can also be helpful as a preventive measure in herds with a high incidence of cutaneous fibropapillomas or to reduce the risk of penile fibropapillomas in groups of young bull calves.685 Recombinant BPV proteins have shown promise in experimental studies and could form the basis for a vaccine that protects against all BPV types.691,694 Vaccination with avian Newcastle disease virus vaccine has also enhanced clinical recovery from disease.695
REFERENCES
1 Wilson WD, Kanara EW, Spensley MS, et al. American Association of Equine Practitioners’ Vaccination Guidelines Subcommittee of the AVMA Council on Biologic and Therapeutic Agents: guidelines for vaccination of horses. J Am Vet Med Assoc. 1995;207:426.
2 Jorm LR. Laboratory studies on the survival of Streptococcus equi subspecies equi on surfaces. Plowright W, Rossdale PD, Wade JF, . Equine Infectious Diseases VI: Proceedings of the Sixth International Conference. Newmarket, England: R & W Publications; 1991:39.
3 Horohov DW, Lunn DP, Townsend HG, Wilson D. Equine vaccination. J Vet Intern Med. 2000;14:221.
4 Lunn DP, Townsend HG. Equine vaccination. Vet Clin North Am Equine Pract. 2000;16:199.
5 Townsend HGG. The role of vaccines and their efficacy in the control of infectious respiratory disease of the horse. Proceedings of the 46th Annual Convention of the American Association of Equine Practitioners, San Antonio. 2000:21.
6 Chambers TM, Holland RE, Tudor LR, et al. A new modified live equine influenza virus vaccine: phenotypic stability, restricted spread and efficacy against heterologous virus challenge. Equine Vet J. 2001;33:630.
7 Lunn DP, Hussey S, Sebing R, et al. Safety, efficacy, and immunogenicity of a modified-live equine influenza virus vaccine in ponies after induction of exercise-induced immunosuppression. J Am Vet Med Assoc. 2001;218:900.
8 Townsend HG, Penner SJ, Watts TC, et al. Efficacy of a cold-adapted, intranasal, equine influenza vaccine: challenge trials. Equine Vet J. 2001;33:637.
9 Ng T, Hathaway D, Jennings N, et al. Equine vaccine for West Nile virus. Dev Biol (Basel). 2003;114:221.
10 Grosenbaugh DA, Backus CS, Karaca K, et al. The anamnestic serologic response to vaccination with a canarypox virus—vectored recombinant West Nile virus (WNV) vaccine in horses previously vaccinated with an inactivated WNV vaccine. Vet Ther. 2004;5:251.
11 Minke JM, Siger L, Karaca K, et al. Recombinant canarypoxvirus vaccine carrying the prM/E genes of West Nile virus protects horses against a West Nile virus—mosquito challenge. Arch Virol Suppl. 2004;18:221.
12 Siger L, Bowen RA, Karaca K, et al. Assessment of the efficacy of a single dose of a recombinant vaccine against West Nile virus in response to natural challenge with West Nile virus—infected mosquitoes in horses. Am J Vet Res. 2004;65:1459.
13 Chiang YW, Jensen C, Holt T, et al. Demonstration of the efficacy of a West Nile virus DNA vaccine in foals. Proceedings of the 51st Annual Convention of the American Association of Equine Practitioners, Seattle. 2005:183.
14 Edlund Toulemonde C, Daly J, Sindle T, et al. Efficacy of a recombinant equine influenza vaccine against challenge with an American lineage H3N8 influenza virus responsible for the 2003 outbreak in the United Kingdom. Vet Rec. 2005;156:367.
15 Long MT, Gibbs EP, Seino KK, et al. Safety and efficacy of a live attenuated West Nile virus chimers vaccine in horses with experimentally induced West Nile virus clinical disease. Proceedings of the 51st Annual Convention of the American Association of Equine Practitioners, Seattle. 2005:177.
16 Long MT, Seino KK, Gibbs EP, et al. Duration of immunity against experimentally induced West Nile virus encephalomyelitis in horses using a West Nile virus chimera vaccine. Proceedings of the 52nd Annual Convention of the American Association of Equine Practitioners, San Antonio. 2006:235.
17 Siger L, Bowen R, Karaca K, et al. Evaluation of the efficacy provided by a recombinant canarypox-vectored equine West Nile virus vaccine against an experimental West Nile virus intrathecal challenge in horses. Vet Ther. 2006;7:249.
18 Minke JM, Toulemonde CE, Coupier H, et al. Efficacy of a canarypox-vectored recombinant vaccine expressing the hemagglutinin gene of equine influenza H3N8 virus in the protection of ponies from viral challenge. Am J Vet Res. 2007;68:213.
19 Morley PS, Townsend HG, Bogdan JR, Haines DM. Efficacy of a commercial vaccine for preventing disease caused by influenza virus infection in horses. J Am Vet Med Assoc. 1999;215:61.
20 Holmes MA, Townsend HG, Kohler AK, et al. Immune responses to commercial equine vaccines against equine herpesvirus-1, equine influenza virus, eastern equine encephalomyelitis, and tetanus. Vet Immunol Immunopathol. 2006;111:67.
21 Lunn DP. Immunological basis of vaccination. Proceedings of the 46th Annual Convention of the American Association of Equine Practitioners, San Antonio. 2000:1.
22 Slater J. Immunological control of viral and bacterial pathogens. Proceedings of the 46th Annual Convention of the American Association of Equine Practitioners, San Antonio. 2000:10.
23 Wilson WD, Mihalyi JE, Hussey S, Lunn DP. Passive transfer of maternal immunoglobulin isotype antibodies against tetanus and influenza and their effect on the response of foals to vaccination. Equine Vet J. 2001;33:644.
24 van Maanen C, Bruin G, de Boer-Luijtze E, et al. Interference of maternal antibodies with the immune response of foals after vaccination against equine influenza. Vet Q. 1992;14:13.
25 Van Oirschot JT, Bruin G, de Boer-Luytze E, Smolders G. Maternal antibodies against equine influenza virus in foals and their interference with vaccination. Zentralbl Veterinarmed B. 1991;38:391.
26 Cullinane A, Weld J, Nelly M, McBride C. The interference of maternal antibodies with the immune response of thoroughbred foals and yearlings to vaccination against equine influenza. Hakijama H, Plowright W, . Proceedings of the Seventh International Conference on Equine Infectious Diseases, Tokyo. 1994:52.
27 Cullinane A, Weld J, Osborne M, et al. Field studies on equine influenza vaccination regimes in thoroughbred foals and yearlings. Vet J. 2001;161:174.
28 Conboy HS, Berry DB, Fallon EH, et al. Failure of foal seroconversion following equine influenza vaccination. Proceedings of the 43rd Annual Convention of the American Association of Equine Practitioners, Phoenix. 1997:22.
29 Holland RE, Conboy HS, Berry DB, et al. Age dependence on foal vaccination for equine influenza: new evidence from the USA. Wernery U, et al. Equine Infectious Diseases VIII: Proceedings of the Eighth International Conference. Newmarket, England: R & W Publications; 1999:547.
30 Wilson JH, et al. Investigation of vaccine-induced tolerance to Eastern equine encephalitis virus in foals. Proceedings of the 41st Annual Conference of the American Association of Equine Practitioners, Lexington, Ky. 1995:178.
31 Holznagel DL, Hussey S, Mihalyi JE, et al. Onset of immunoglobulin production in foals. Equine Vet J. 2003;35:620.
32 Bürki F, Nowotny N, Rossmanith W, et al. Training of the immune system of foals against ERP virus infections by frequent vaccination with presently available commercial vaccines. Dtsch Tierarztl Wochenschr. 1989;96:162.
33 Breathnach CC, Allen GP, Holland RE, et al. Problems associated with vaccination of foals against equine herpesvirus-4 and the role of anti—EHV-4 maternal antibodies. Wernery U, et al. Equine Infectious Diseases VIII: Proceedings of the Eighth International Conference. Newmarket, England: R & W Publications; 1999:426.
34 Ferguson JA, Reeves WC, Hardy JL. Studies on immunity to alphaviruses in foals. Am J Vet Res. 1979;40:5.
35 Wilson WD. Vaccination programs for foals and weanlings. Proceedings of the 45th Annual Convention of the American Association of Equine Practitioners. 1999:254.
36 Wilson WD, Rossdale PD. Effect of age on the serological responses of thoroughbred foals to vaccination with an inactivated EHV-1/EHV-4 vaccine. Wernery U, et al. Equine Infectious Diseases VIII: Proceedings of the Eighth International Conference. Newmarket, England: R & W Publications; 1999:428.
37 Wilson WD. Strategies for vaccinating mares, foals, and weanlings. Proceedings of the 51st Annual Convention of the American Association of Equine Practitioners, Seattle. 2005:421.
38 Heldens JG, Kersten AJ, Weststrate MW, van den Hoven R. Duration of immunity induced by an adjuvanted and inactivated equine influenza, tetanus and equine herpesvirus 1 and 4 combination vaccine. Vet Q. 2001;23:210.
39 Mair TS. Adverse reactions to equine vaccinations: a preliminary survey. Vet Rec. 1988;122:396.
40 Taylor FRG. Speculations on the cause of adverse reactions to equine influenza vaccination. Equine Vet Educ. 1989;1:79.
41 Timoney PJ, McCollum WH. Equine viral arteritis. Vet Clin North Am Equine Pract. 1993;9:295.
42 Lohrer J, Radvila P. Active tetanus protection in the horses and the duration of immunity. Schweiz Arch Tierheilkd. 1970;112:307.
43 Liefman CE. Active immunisation of horses against tetanus including the booster dose and its application. Aust Vet J. 1981;57:57.
44 Jansen BC, Knoetze PC. The immune response of horses to tetanus toxoid. Onderstepoort J Vet Res. 1979;46:211.
45 Heinig A. Experimentelle untersuchungen uber den eintritt der immunitat nach einmaliger tetanue-schutzimpfung. Arch Exper Vet Med. 1954;8:394.
46 Green SL, Little CB, Baird JD, et al. Tetanus in the horse: a review of 20 cases (1970 to 1990). J Vet Intern Med. 1994;8:128.
47 Liefman CE. Combined active-passive immunisation of horses against tetanus. Aust Vet J. 1980;56:119.
48 Panciera RJ. Serum hepatitis in the horse. J Am Vet Med Assoc. 1969;155:408.
49 Messer NT, Johnson PJ. Idiopathic acute hepatic disease in horses: 12 cases (1982–1992). J Am Vet Med Assoc. 1994;204:1934.
50 Hays MB. Definitive efficacy and safety testing for equine encephalomyelitis vaccine. J Am Vet Med Assoc. 1969;155:374.
51 Barber TL, Walton TE, Lewi KJ. Efficacy of trivalent inactivated encephalomyelitis virus vaccine in horses. Am J Vet Res. 1978;39:621.
52 Eisner RJ, Nusbaum SR. A study to determine the optimum time for vaccination of foals against eastern and western encephalitis viruses. Proceedings of the American Association of Veterinary Laboratory Diagnosticians. 1979:435.
53 Gibbs EPJ, Wilson JH, All BPIII, Powell DG. Studies on passive immunity and the vaccination of foals against eastern equine encephalitis in Florida. Powell DG, . Equine Infectious Diseases V: Proceedings of the Fifth International Conference on Equine Infectious Diseases. Lexington, Ky: University Press of Kentucky; 1988:201.
54 Liu IKM. Duration of maternally derived antibodies in neonatal foals. Mod Vet Pract. 1986;67:454.
55 Wilson JH, Rubin HL, Lane TJ, Gibbs EPJ. A survey of eastern equine encephalomyelitis in Florida horses: prevalence, economic impact, and management practices, 1982–1983. Prev Vet Med. 1986;4:261.
56 Wilson JH, Davis A, Bender JB, Minicucci LA. Residual effects of West Nile viral encephalomyelitis in horses. Proceedings of the 49th Annual Convention of the American Association of Equine Practitioners, New Orleans. 2003:399.
57 Bunning ML, Bowen RA, Cropp CB, et al. Experimental infection of horses with West Nile virus. Emerg Infect Dis. 2002;8:380.
58 Arroyo J, Miller C, Catalan J, et al. ChimeriVax—West Nile virus live-attenuated vaccine: preclinical evaluation of safety, immunogenicity, and efficacy. J Virol. 2004;78:12497.
59 Seino KK, Long MT, Gibbs EP. Investigation into the comparative efficacy of three West Nile virus vaccines in experimentally induced West Nile clinical disease in horses. Proceedings of the 52nd Annual Convention of the American Association of Equine Practitioners, San Antonio. 2006:233.
60 Salazar P, Traub-Dargatz JL, Morley PS, et al. Outcome of equids with clinical signs of West Nile virus infection and factors associated with death. J Am Vet Med Assoc. 2004;225:267.
61 Schuler LA, Khaitsa ML, Dyer NW, Stoltenow CL. Evaluation of an outbreak of West Nile virus infection in horses: 569 cases (2002). J Am Vet Med Assoc. 2004;225:1084.
62 Gardner IA, Wong SJ, Ferraro GL, et al. Incidence and effects of West Nile virus infection in vaccinated and unvaccinated horses in California. Vet Res. 2007;38:109.
63 Vest DJ, Cohen ND, Berezowski CJ, et al. Evaluation of administration of West Nile virus vaccine to pregnant broodmares. J Am Vet Med Assoc. 2004;225:1894.
64 Long MT. Flavivirus infection. In: Sellon DC, Long MT, editors. Equine infectious diseases. St Louis: Saunders; 2007:198.
65 Tizard I, Y Ni. Use of serologic testing to assess immune status of companion animals. J Am Vet Med Assoc. 1998;213:54.
66 Green SL, Smith LL, Vernau W, Beacock SM. Rabies in horses: 21 cases (1970–1990). J Am Vet Med Assoc. 1992;200:1133.
67 Morley PS, Townsend HG, Bogdan JR, Haines DM. Risk factors for disease associated with influenza virus infections during three epidemics in horses. J Am Vet Med Assoc. 2000;216:545.
68 Wilson WD. Equine influenza. Vet Clin North Am Equine Pract. 1993;9:257.
69 Nelson KM, Schram BR, McGregor MW, et al. Local and systemic isotype-specific antibody responses to equine influenza virus infection versus conventional vaccination. Vaccine. 1998;16:1306.
70 Hannant D, Mumford JA. Cell mediated immune responses in ponies following infection with equine influenza virus (H3N8): the influence of induction culture conditions on the properties of cytotoxic effector cells. Vet Immunol Immunopathol. 1989;21:327.
71 Hannant D, Jessett DM, O’Neill T, Mumford JA. Antibody isotype responses in the serum and respiratory tract to primary and secondary infections with equine influenza virus (H3N8). Vet Microbiol. 1989;19:293.
72 Hannant D, Jessett DM, O’Neill T, et al. Duration of circulating antibody and immunity following infection with equine influenza virus. Vet Rec. 1988;122:125.
73 van Maanen C, Cullinane A. Equine influenza virus infections: an update. Vet Q. 2002;24:79.
74 Hannant D, et al. Cellular immune responses stimulated by inactivated virus vaccines and infection with equine influenza virus (H3N8). Equine Infectious Diseases VII: Proceedings of the Seventh International Conference. Newmarket UK: R & W Publications; 1994:169.