Urinalysis
Urinalysis is a relatively simple, rapid, and inexpensive laboratory procedure. It evaluates the physical and chemical properties of urine, as well as the sediment. A urinalysis provides information to the veterinarian on the status of the urinary system, metabolic and endocrine systems, and electrolyte and hydration status. Therefore the veterinarian may request that the owner bring a urine sample for initial testing.
SPECIMEN COLLECTION
The first step in performing a urinalysis is proper collection of a urine sample, which must be carefully obtained to ensure accurate results. Analysis of urine samples should be performed only on samples taken before administration of therapeutic agents. Urine specimens may be obtained by natural voiding of urine, bladder expression, catheterization, or cystocentesis. The two preferred methods are cystocentesis and catheterization; these methods provide optimal samples for all aspects of urinalysis by avoiding contamination from the distal genital tract and external areas. Collecting samples by voiding or expression of the bladder may be easier, but urine collected in these ways may be of limited diagnostic value. Except for cytologic examination, performing a urinalysis on preprandial morning samples is best. Morning samples tend to be the most concentrated and least affected by dietary factors, which increases the chances of finding formed elements.
Voided or “Free Catch” Sample
The easiest sample to obtain is the voided sample, which is collected as the animal urinates. A sample collected in this manner is not satisfactory for bacteriologic examination because it is often contaminated during urination. Occasionally voided samples contain increased white blood cell (WBC) counts because of contamination from inflammatory lesions of the distal genital tract. Results of other evaluations are usually unaffected.
A voided sample is collected in a clean, although not necessarily sterile, container. If possible, the vulva or prepuce should be washed to decrease contamination of the sample before collection. Animals’ owners may not be able to do this because they may be asked to collect a sample when the animal spontaneously urinates. Furthermore, the cleansed tissue in the external orifice area does not remain clean for long. A midurination (midstream) sample is best because it is less likely to be contaminated. However, the initially voided urine is sometimes collected as a precaution against not being able to collect a midurination sample.
Dogs may begin to urinate and then stop when collection is attempted. Chances of successfully obtaining urine may be increased by attaching a paper cup to a long pole and collecting the sample without disturbing the animal. A voided sample from a cat is difficult to obtain. Occasionally cats urinate in an empty litter box. Veterinarians prefer to give cat owners nonabsorbent granules to use in the litter box. Cows may be stimulated to urinate by rubbing a hand or dry hay ventral to the vulva in a circular fashion. Sheep may be stimulated to urinate by occluding their nostrils. Horses may be stimulated to urinate by rubbing a warm, wet cloth on their ventral abdomen or by placing them in a clean stall with fresh hay.
Bladder Expression
Urine may be collected in small animals by manual compression of the bladder. Samples obtained in this manner are also unsatisfactory for bacteriologic culturing. As with collection of a voided sample, the external genitalia should be cleansed before bladder expression. With the animal standing or in lateral recumbency, the bladder is palpated in the caudal abdomen and gentle, steady pressure is applied. Care must be taken not to exert too much pressure and injure or rupture the bladder. Relaxation of the bladder sphincters often takes a few minutes. Occasionally an increase in red blood cells (RBCs) will be found because of pressure applied to the bladder and an increase in WBCs resulting from contamination originating in the distal genital tract. If bacteria are in the urine, the kidneys may become infected from these bacteria. This method should never be used on animals whose urethra may be obstructed or whose bladder wall may be fragile because excessive pressure can cause the bladder to rupture. In large animals, urination may be stimulated by maintaining pressure on the bladder through the rectal wall while performing rectal palpation.
Catheterization
Catheterization is the insertion of a polypropylene or rubber catheter into the bladder by way of the urethra. A variety of catheters exist for different species and sexes. As with the previous two methods of collection, the external genitalia should be cleansed before the procedure. Sterile catheters should always be used and sterile gloves worn. Care must be taken to maintain sterility and prevent trauma to the urinary tract. This method may be used for culture and sensitivity if a cystocentesis cannot be performed. For female animals a speculum improves visualization of the urethral orifice and thus facilitates the catheterization. The catheter should pass easily through the urethra. A small amount of sterile, water-soluble lubricating jelly, such as K-Y Jelly (Johnson & Johnson, Arlington, TX), should be placed on the tip of the catheter. Care must be taken to avoid trauma to the sensitive urethral mucosa. The distal end of many catheters is designed for attachment to a syringe so that urine can be collected with gentle aspiration. Collection into a sterile syringe is especially advantageous if bacteriologic culture is anticipated. Often the first portion of the sample obtained is discarded because of possible contamination as the catheter was advanced through the distal urethra. Occasionally an increase in RBCs and epithelial cells may be seen in the sample because of urethral mucosa damage from the catheter.
Cystocentesis
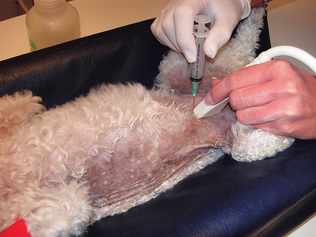
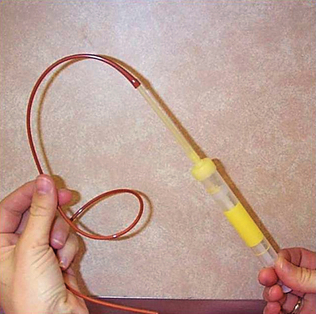
Cystocentesis is used often to collect sterile urine samples from dogs and cats only when the bladder is sufficiently distended so that it can be easily isolated. This procedure should be performed only on calm, easily restrained patients (Fig. 5-1). An ultrasound-guided cystocentesis may also be performed. The bladder must be palpated before the procedure to avoid damage to other internal organs. When performing a cystocentesis, a 22- or 20-gauge needle by 1 inch or 1½ inches and a 10-ml syringe should be used. Once the needle is through the skin, it should never be redirected because of the potential for damage to other internal organs. With the animal in lateral recumbency or ventral recumbency or standing, the bladder is gently palpated and immobilized and the needle is inserted into the caudal abdomen. For male dogs, insert the needle caudal to the umbilicus and to the side of the sheath. For female dogs and for cats, insert the needle on the ventral midline caudal to the umbilicus. Gently aspirate urine into the syringe and properly label it with the patient information. This sample can also be used for culture and sensitivity testing. Occasionally samples contain an increase in RBCs caused by bladder trauma. The urine S-Monovette system (Sarstedt AG & Co., Nümbrecht, Germany) is a commercially available urine collection device that consists of a sterile, individually wrapped syringe with a disposable tube (Fig. 5-2). This system can simplify cystocentesis collection and minimizes the potential for contamination when samples are transferred between a syringe and collection tube. The sample is drawn directly into the tube. The tube can also be centrifuged for urine sediment examination. A special tip is also included to collect catheter specimens.
All the aforementioned methods of collection are satisfactory for qualitative analysis. However, for quantitative analysis, a 24-hour sample must be collected. Ratios of certain urine constituents, such as protein/creatinine, have recently been used to obtain data that may be interpreted in a similar manner to 24-hour urinary excretions.
QUALITY ASSURANCE
Quality assurance begins with proper specimen identification and handling. All samples should be labeled immediately after collection, and urinalysis should be performed as soon as possible. Reagent strips and tablets must be kept in tightly sealed bottles, and outdated reagents must be replaced with fresh reagents. Reactions for most constituents in urine may be checked against available controls (e.g., Chek-Stix, Bayer Corporation, Leverkusen, Germany; Uritrol, YD Diagnostics, Seoul, Korea; Liquid Urine Control, Kenlor Industries, Inc., Santa Ana, CA). In addition, urine samples with distinct reactions for certain constituents sometimes may be preserved and used as positive controls. The results obtained from control samples and made-up controls should be plotted to determine whether observer drift or reagent decomposition is occurring. The urinalysis laboratory report should include patient information, collection technique, date and time collected, method of preservation (if used), complete urinalysis results, including the results of microscopic examination results. Precision and accuracy need to be maintained by the veterinary technician for proper interpretation of results.
SPECIMEN STORAGE AND HANDLING
Ideally, samples should be analyzed within 30 minutes to 1 hour of collection to avoid postcollection artifacts and degenerative changes. If immediate analysis is not possible, refrigeration preserves most urine constituents for an additional 6 to 12 hours. Refrigeration may have an impact on urine specific gravity, so this test should be done before refrigeration. If a urine sample is going to be refrigerated, it should have a tight-fitting lid to prevent evaporation and contamination. Crystals may form when the urine cools. Decreased glucose and bilirubin concentrations, increased pH resulting from bacterial breakdown of urea to ammonia, crystal formation with increased sample turbidity, breakdown of casts and RBCs (especially in dilute or alkaline urine), and bacterial proliferation may occur in samples allowed to stand for long periods at room temperature. Many crystals may form in refrigerated samples. Refrigerated urine should be warmed to room temperature before evaluation, and crystals that formed during cooling may not dissolve when the sample is brought to room temperature. The urine sample should be mixed by gentle inversion before evaluation so that formed elements are evenly distributed. Cells tend to break down rapidly in urine, so if cytologic evaluation is to be performed, the urine should be centrifuged soon after collection and 1 to 2 drops of the patient’s serum or bovine albumin added to the sediment to preserve cell morphologic characteristics.
Samples to be transported to an outside laboratory or held for longer than 6 to 12 hours may be preserved by adding one of the following: 1 drop of 40% formalin in 1 oz of urine; toluene sufficient to form a layer on top of the sample; a single thymol crystal; or one part 5% phenol to nine parts urine. If formalin is used as a preservative, chemical tests should be performed before the addition of formalin because it interferes with some chemical analyses, especially that for glucose. Formalin, however, is the best preservative for formed elements in urine.
PHYSICAL PROPERTIES
Physical properties of urine include all the observations that may be made without the aid of a microscope or chemical reagents. Volume, color, odor, transparency, and specific gravity of urine are evaluated. (Procedure 5-1 describes the procedure for a routine urinalysis.)
Urine Volume
The animal’s owner often provides information concerning the amount of urine passed. However, owners may mistake frequent urination, or pollakiuria, for increased urine production, or polyuria. Therefore obtaining an estimate of the amount of urine an animal is producing is important. Many factors unrelated to disease influence the amount of urine produced. Some of these factors include fluid intake, external losses (especially through the respiratory system and the intestinal tract), environmental temperature and humidity, amount and type of food, level of physical activity, size of the animal, and species. Observing a single urination is not reliable for estimating urine output of an animal. Ideally, 24-hour urine volume should be determined, although this is often impractical. Observing an animal in its cage or outdoors may provide a rough estimate of the volume of urine being produced. Table 5-1 lists the approximate daily urine production for common domestic species. The amount of urine produced per day is variable. Normal urine output for adult dogs and cats is approximately 20 to 40 ml/kg body weight per day.
An increase in daily urine output or production is termed polyuria and is usually accompanied by polydipsia. Polydipsia is defined as an increase in water consumption. With polyuria the urine is usually pale or light yellow in color and has a low specific gravity. Polyuria occurs in many diseases, including nephritis, diabetes mellitus, diabetes insipidus, pyometra in dogs and cats, and liver disease. It also is seen after administration of diuretics, corticosteroids, or fluids.
Oliguria, a decrease in daily urine output, may occur when an animal has restricted access to water. It can be severe when the environmental temperature increases and causes excess water loss through the respiratory system. With oliguria the urine is usually concentrated and has a high specific gravity. Oliguria also occurs with acute nephritis, fever, shock, heart disease, and dehydration. Anuria, the absence of urine production, may be seen in complete urethral obstruction, urinary bladder rupture, and renal shut down.
Color
Normal urine color is light yellow to amber as a result of the presence of pigments called urochromes. The magnitude of yellow color in urine varies with the degree of urine concentration or dilution (Table 5-2). Colorless urine usually has a low specific gravity and is often associated with polyuria. Dark yellow to yellow-brown urine generally has a high specific gravity and may be associated with oliguria. Yellow-brown or green urine that produces a greenish-yellow foam when shaken is likely to contain bile pigments. Red or red-brown urine indicates the presence of RBCs (referred to as hematuria) or hemoglobin (referred to as hemoglobinuria). Urine that is brown when voided may contain myoglobin (referred to as myoglobinuria) excreted during conditions that cause muscle cell lysis, such as rhabdomyolysis in horses. Some drugs may alter the color of urine; red, green, or blue urine may be observed. When observing urine it should be in a clear plastic or glass container against a white background to be properly evaluated for color.
Clarity/Transparency
In most species freshly voided urine is transparent or clear. Normal equine urine is cloudy because of a high concentration of calcium carbonate crystals and mucus secreted by glands in the renal pelvis. Normal rabbit urine also has high concentrations of calcium carbonate crystals and appears milky. When observing urine for the degree of transparency, it should be placed against a letter-print background. Transparency is noted as clear, slightly cloudy, cloudy, or turbid (flocculent) depending on how well the letters can be read through the sample. Clear samples usually do not have much sediment on centrifugation. Cloudy samples usually contain large particles and often yield a significant amount of sediment on centrifugation. Urine may become cloudy while standing because of bacterial multiplication or crystal formation. Substances that cause urine to be cloudy include RBCs, WBCs, epithelial cells, casts, crystals, mucus, fat, and bacteria. Other causes of turbidity can include contaminants from the collection container or surface and contamination with feces. Flocculent samples contain suspended particles that are sometimes large enough to be seen with the naked eye.
Odor
The odor of urine is not highly diagnostic but sometimes may be helpful. Normal urine has a distinctive odor that varies among species. The urine of male cats, goats, and pigs has a strong odor. An ammonia odor may occur with cystitis caused by bacteria that produce urease (Proteus spp. or Staphylococcus spp.) that has metabolized urea to ammonia. Samples left standing at room temperature may occasionally develop an ammonia odor as a result of bacterial growth. A characteristic sweet or fruity odor to urine indicates ketones and is most commonly found with diabetes mellitus, ketosis in cows, and pregnancy disease in ewes.
Specific Gravity
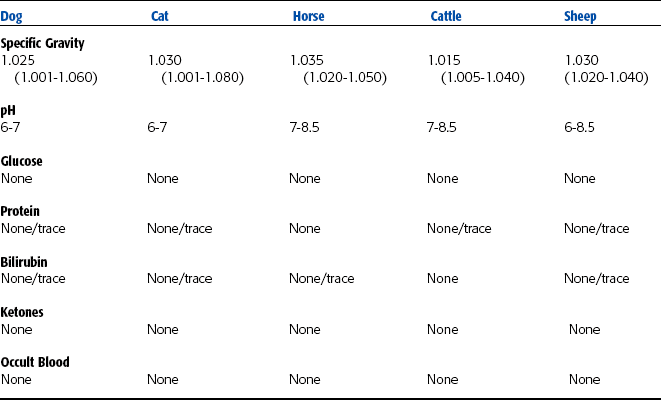
Specific gravity is defined as the weight (density) of a quantity of liquid compared with that of an equal amount of distilled water. The number and molecular weight of dissolved solutes determine the specific gravity of urine. Specific gravity may be determined before or after centrifugation because the particles that settle during centrifugation have little or no effect on specific gravity. Whichever method is used to perform the specific gravity in a specific clinic, whether before or after centrifugation, the same method must be consistently performed by all clinic personnel. If the urine is turbid, the sample should be centrifuged and the supernatant used to determine the specific gravity. The specific gravity of urine from polyuric patients tends to be low, and urine from oliguric patients tends to be high. The specific gravity of normal urine depends on eating and drinking habits, environmental temperature, and when the sample was collected. An early morning, midurination sample tends to be the most concentrated. Interpretation of urine specific gravity yields information on the hydration status and the ability of the kidneys to concentrate or dilute urine. Specific gravity of normal animals is extremely variable and fluctuates throughout the day. Table 5-3 lists the urine specific gravity ranges for normal domestic animals. In normal dogs the urine specific gravity may range from 1.001 to 1.060 and in normal cats from 1.001 to 1.080.
To determine specific gravity of urine, the refractometer, urinometer, or reagent strips can be used. Urine specific gravity is less frequently determined by use of a urinometer. This instrument requires a large amount of urine (approximately 10 ml) and generally provides less-reproducible results than a refractometer.
Refractometer
Specific gravity is most commonly determined by a refractometer. More information on the principle of the refractometer is presented in Chapter 1. Urine contains substances that absorb various wavelengths of light. The light waves bend as they pass through the medium, and this bend is measured by the refractometer. The refractive index of a fluid is influenced by the same factors that determine specific gravity and therefore provides an estimate of urine specific gravity.
Reagent Strips
Several reagent strips have been developed to determine urine specific gravity. Reagent strip specific gravity is the least reliable method for determining urine specific gravity in animals.
Causes of Altered Urine Specific Gravity
Increased urine specific gravity is seen with decreased water intake, increased fluid loss through sources other than urination (e.g., sweating, panting, diarrhea), and increased excretion of urine solutes. Decreased water intake in animals with normal renal function rapidly causes increased urine specific gravity. Increased urine specific gravity may occur in acute renal failure, dehydration, and shock.
Decreased urine specific gravity is seen in diseases in which the kidneys cannot resorb water and with increased fluid intake, such as with polydipsia or excessive fluid administration. Pyometra, diabetes insipidus, psychogenic polydipsia, some liver diseases, certain types of renal disease, and diuretic therapy may also cause decreased urine specific gravity.
Isosthenuria occurs when the urine specific gravity approaches that of glomerular filtrate (1.008 to 1.012). In other words, urine with this specific gravity range has not been concentrated or diluted by the kidneys. Animals with chronic renal disease frequently produce isosthenuric urine. In animals with kidney disease, the closer the specific gravity is to isosthenuric, the greater the amount of kidney function that has been lost. When these animals are deprived of water, their urine specific gravity usually remains in the isosthenuric range. Animals with decreased renal function are often slightly to moderately dehydrated and have urine specific gravity slightly greater than isosthenuric (1.015 to 1.020).
CHEMICAL PROPERTIES
Testing for various chemical constituents of urine is usually performed with reagent strips that are impregnated with appropriate chemicals or reagent tablets. The container of reagent strips must be stored at room temperature with the lid tightly closed. The expiration date should also be noted. Some reagent strips simultaneously test for numerous constituents, and other strips exist for individual tests. Urine is added to the reagent strip from a pipette, or the strips are dipped in the urine sample and the color changes are noted at specific time intervals. The concentration of various constituents is determined by comparing the colors on the strip with the color chart on the label of the strip container (Fig. 5-3). The manufacturer’s directions must be carefully followed.
pH
The pH expresses the hydrogen ion (H+) concentration. Essentially, pH is a measure of the degree of acidity or alkalinity of urine. A pH above 7.0 is alkaline, whereas a pH below 7.0 is acidic. Proper technique must be used to obtain accurate results. The urine sample must be fresh to obtain accurate results. The pH of samples left standing open at room temperature tends to increase resulting from a loss of carbon dioxide, whereas delays in reading the reaction may lead to color changes and false readings. If samples containing urease-producing bacteria (Proteus spp. or Staphylococcus spp.) are left standing, the pH is usually increased.
The kidneys play a role in acid-base regulation of the body. Kidneys must vary the pH of urine to compensate for diet and products of metabolism. The pH of a healthy animal’s urine depends largely on its diet. Alkaline urine usually is found in animals on plant diets, whereas high-protein cereal diets or diets of animal origin cause acidic urine. Therefore herbivores normally have alkaline urine, carnivores have acidic urine, and omnivores have either acidic or alkaline urine depending on what was ingested. Many dog foods contain substantial amounts of plant material that may cause the urine to be slightly alkaline. Nursing herbivores have acidic urine from the consumption of milk. Other factors such as stress and excitement, especially in cats, increase the urine pH and may create a transient glucosuria. Table 5-3 lists the normal urine constituents and characteristics, including pH, for common domestic species.
Urine pH is usually measured with reagent strips or a pH meter. Factors that may decrease the pH (acidity) include fever, starvation, high-protein diet, acidosis, excessive muscular activity, or administration of certain drugs. Increased pH (alkalinity) may be caused by alkalosis, high-fiber diets (plants), infection of the urinary tract with urease bacteria, use of certain drugs, or urine retention such as occurs with urethral obstruction or bladder paralysis. If the pH of the urine is too acidic or too alkaline specific crystals or uroliths can form. The pH can be manipulated with diet to help dissolve the solids or prevent them from forming.
Protein
Protein is usually absent or present in only trace amounts in normal urine obtained by catheterization or cystocentesis. In healthy animals, plasma proteins that pass into the glomerular filtrate are resorbed in the renal tubules before the filtrate reaches the renal pelvis. However, voided samples or those obtained by expressing the bladder may contain a small amount of protein from secretions that may contaminate urine during its passage along the urinary tract. Trauma to the urinary tract that results from cystocentesis, catheterization, or bladder expression may occasionally cause sufficient bleeding that results in a trace of protein in the urine. Urine protein measurements are interpreted in light of the collection method, urine specific gravity, rate of urine formation, and contributions from any hemorrhage or inflammation noted by sample analysis. Protein levels in the urine may be measured by several methods, including reagent test strips, sulfosalicylic acid turbidity test, and urine protein/creatinine ratio.
Protein Determination by Reagent Test Strips
Urine dipsticks allow semiquantitative measurement of protein in urine by progressive color changes on the reaction pad. Reagent strip analysis is a rapid, convenient, and reasonably accurate method of determining urinary protein levels. The accuracy of these methods is variable. Reagent strips primarily detect albumin (protein soluble in water) and are much less sensitive to globulins (proteins insoluble in water). False-positive results may occur in alkaline urine depending on factors such as diet, urinary tract infection, or urine retention (urethral obstruction). Protein measurements considered excessive or pathologic should be confirmed by sulfosalicylic acid turbidity test or specific biochemical analysis. Microalbuminuria is the presence of an abnormal amount of albumin (1 to 30 mg/dl) in urine that is not detected by the reagent strip method. The reagent strip method detects urine protein concentrations that are greater than 30 mg/dl. The albumin-capture enzyme linked immunosorbent assay (ELISA) method is used to measure albumin levels 1 to 30 mg/dl in the urine (Fig. 5-4). See Chapter 8 for more information on the principles of ELISA tests.
Protein Determination by Sulfosalicylic Acid Turbidity Test
Sulfosalicylic acid turbidity determines urine protein levels by acid precipitation. The resultant turbidity is proportional to the concentration of protein. Results are compared with levels in prepared standards and thus may be reported in semiquantitative units. The advantage of this method is that it is equally sensitive to albumin and globulins and is quite useful to confirm strip methods, especially in alkaline urine. This test also measures Bence Jones proteins (light chain proteins that can pass through the glomerulus). Components of extremely alkaline urine may interact with the acid and decrease the amount of protein precipitated.
Urine Protein/Creatinine Ratio
This test is used to help confirm significant amounts of protein in the urine. To determine its significance, urine protein concentration can be compared with that of creatinine. The sample is centrifuged to separate particulate matter (cells) from dissolved substances (protein) and the creatinine and protein concentration of the supernatant is used. The ratio is obtained by dividing the protein concentration by the creatinine concentration. The ratio is not affected by urine concentration and volume and therefore aids in the accurate assessment of urine protein loss in patients with low specific gravity.
Interpretation of Protein in Urine
Very dilute urine may yield a false-negative result because the protein concentration may be below the sensitivity of the testing method. A trace amount of protein in a very dilute sample may be clinically significant because dilute urine often occurs when a large volume of urine is being produced, such as in a patient with chronic renal failure.
Occasionally a small amount of protein is found in the urine of normal animals. Transient proteinuria may result from a temporary increase in glomerular permeability, allowing excessive protein to enter the filtrate. This condition is caused by increased pressure in the glomerular capillaries and may be found with muscle exertion, emotional stress, or convulsions. Occasionally a small amount of urine protein is found after parturition, during the first few days of life, and during estrus. The presence of protein in the urine is usually abnormal and is primarily attributable to disease of the urinary tract (or possibly the genital system).
In most cases proteinuria indicates disease of the urinary tract, especially of the kidneys. Both acute and chronic renal diseases lead to proteinuria. Acute nephritis is characterized by marked proteinuria with WBCs and casts in the urine, whereas in chronic renal disease the degree of proteinuria is qualitatively less. However, in chronic renal disease urine output is usually excessive with low specific gravity; therefore the total protein excreted is actually quite significant. The ratio of urine protein to creatinine is used to determine the degree of protein loss in chronic renal disease.
Multiple myeloma, a cancer of plasma cells, may produce large quantities of light chain proteins (Bence Jones protein) that may leak through the glomerulus. In patients with myeloma, proteins may be passed in the urine because they have damaged the glomerulus or because they are the “light chains” that freely pass through the glomerulus. Because these proteins do not react with the protein pads on the reagent strips, the sulfosalicylic acid method is necessary to detect and quantify them.
Mild proteinuria is seen with passive congestion of the kidneys, as in congestive heart failure or any other impediment of blood flow from the kidneys. Proteinuria of renal origin may also be caused by trauma; tumors; renal infarcts; or necrosis resulting from drugs and chemicals such as sulfonamides, lead, mercury, arsenic, and ether.
Inflammation of the urinary or genital tract may cause proteinuria of postrenal origin. Proteinuria also may be seen with traumatic catheterization or bladder expression.
Glucose
The presence of glucose in urine is known as glucosuria or glycosuria. Glucose is filtered through the glomerulus and resorbed by the kidney tubules. The amount of glucose in the urine depends on blood glucose levels and the rates of glomerular filtration and tubular resorption. Glucosuria usually does not occur in normal animals unless the blood glucose level exceeds the renal threshold (approximately 170 to 180 mg/dl for dogs). At this concentration tubular resorption cannot keep up with the glomerular filtration of glucose, and glucose passes into the urine.
Glucosuria occurs in diabetes mellitus as a result of a deficiency of insulin or an inability of insulin to function. Insulin is necessary to transport glucose into body cells, and a deficiency causes hyperglycemia and spilling of glucose into the urine. A high-carbohydrate meal may lead to blood glucose levels exceeding the renal threshold and thus glucosuria. Because of this, a period of fasting is recommended before urine glucose concentration is determined. Fear, excitement, or restraint, especially in cats, often causes hyperglycemia and glucosuria as a result of epinephrine release. Glucosuria often occurs after intravenous administration of fluids containing glucose and occasionally after general anesthesia. Rarely glucosuria is found in hyperthyroidism, Cushing’s disease, and chronic liver disease. A rare condition called renal glucosuria may occur when the blood glucose concentration is in the normal range. Renal glucosuria is caused by reduced resorption of glucose in the renal tubules. Glucosuria may occur in some cats with chronic disease, possibly as a result of altered proximal renal tubular function.
False-positive results for glucose may be seen after the use of various drugs, including ascorbic acid (vitamin C), morphine, salicylates (e.g., aspirin), cephalosporins, and penicillin.
Various reagent test strips are available to detect glucose in urine. Clinitest Reagent Tablets (Bayer Corporation) are also available. These tablets detect any sugar in the urine, whereas most reagent test strips detect only glucose.
Ketones
Ketones include acetone, acetoacetic acid, and β-hydroxybutyric acid. Ketone bodies are formed during incomplete catabolism of fatty acids. Normal animals may have small amounts of ketones in the blood. Conditions characterized by altered carbohydrate metabolism may result in excessive amounts of fat catabolism to provide energy. When fatty acid metabolism is not accompanied by sufficient carbohydrate metabolism, excess ketones are present in the urine, a condition called ketonuria.
A common cause of ketonuria is ketonemia, or ketosis, in lactating cows and pregnant ewes and cows. Ketosis usually occurs early in lactation (3 to 6 weeks after freshing), when the energy for milk production exceeds the capacity of the cow to ingest sufficient feed to meet its energy requirements. In ewes, this condition is called pregnancy toxemia and is seen when the ewe is carrying twins or triplets. Ketosis is associated with hypoglycemia and is caused by carbohydrate intake insufficient to meet energy requirements. Body fat is then rapidly metabolized, resulting in ketonemia and ketonuria.
Ketonuria frequently occurs in animals with diabetes mellitus. Because the animal lacks the insulin necessary for carbohydrate metabolism, fat is broken down to meet the animal’s energy needs and excess ketones are excreted in the urine. Ketones are important sources of energy and are normally produced during fat metabolism. Problems develop, however, when excessive ketones are produced. Ketones are toxic, causing central nervous system depression and acidosis. Acidosis resulting from ketonemia is termed ketoacidosis.
Ketonemia with ketonuria also occurs with high-fat diets, starvation, fasting, long-term anorexia, and impaired liver function. With a high-fat diet, carbohydrates meet a relatively low percentage of energy needs, so a great amount of fat is used to meet energy needs. In the fasting, starved, or anorexic animal, body fat is used to meet energy needs, producing a greater than normal amount of ketones. With liver damage, impaired carbohydrate metabolism leads to fat serving as the main energy source, especially when the damaged liver cannot store adequate amounts of glycogen.
Measurement of Urine Ketone Content
Urinary ketones are detected by using urinary reagent strips with a ketone reagent pad. The color intensity is roughly proportional to the concentration of urine ketones. These methods are most sensitive to acetoacetic acid and less sensitive to acetone and do not detect β-hydroxybutyric acid. β-Hydroxybutyric acid is the first ketone produced by the body in any condition that causes ketosis. Urine reagent test strips may not adequately identify these patients until the ketosis has been present for some time.
Bile Pigments
Bile pigments commonly detected in urine are bilirubin and urobilinogen. Only conjugated bilirubin (water soluble) is found in urine because unconjugated bilirubin does not pass through the glomerulus into the renal filtrate; it is bound to albumin and is not water soluble. Normal dogs, especially males, occasionally have bilirubin in their urine because of a low renal threshold for conjugated bilirubin and the ability of their kidneys to conjugate bilirubin. Many normal cattle also have small amounts of bilirubin in their urine. Bilirubin is usually not found in the urine of cats, pigs, sheep, or horses. In cats the renal threshold is many times that of dogs, and any amount of bilirubin in the urine is considered abnormal and suggests disease.
Bilirubinuria is seen in a number of diseases, including obstruction of bile flow from the liver to the small intestine and in liver disease. Bilirubinuria results from accumulation in hepatic cells of conjugated bilirubin that is released into the blood and excreted in the urine. Conditions causing biliary obstruction include calculi in the bile duct, tumors in the area of the bile duct, acute enteritis, pancreatitis, and obstruction of the upper intestinal tract. When conjugated, bilirubin enters the bloodstream after being released from damaged liver cells and passes into the urine.
Hemolytic anemia (RBC destruction) may also cause bilirubinuria, especially in dogs. In hemolytic anemia, the liver’s ability to metabolize the excess bilirubin may be exceeded, resulting in release of conjugated bilirubin into the blood and ultimately bilirubinuria. In dogs, unconjugated bilirubin from hemoglobin catabolism in the mononuclear phagocytic system can be conjugated in the kidney and passed in the urine.
Bilirubinuria is detected with the Ictotest (Bayer Corporation). A diazo compound in reagent the tablet reacts with bilirubin to produce a blue or purple color. The speed with which the color change occurs and the degree of color change indicates the amount of bilirubin present. Reagent strips are less sensitive than Ictotest tablets. Urine to be tested for bilirubin must not be exposed to light because bilirubin is broken down by short- wave light. False-negative results for bilirubin occur in urine that is exposed to sunlight or artificial light.
In the intestines, bacteria convert bilirubin to stercobilinogen and urobilinogen. The bulk of these products are excreted in the feces, but some are resorbed into the bloodstream and excreted by the liver into the intestinal tract. A small amount of resorbed urobilinogen is excreted by the kidneys into the urine. Urobilinogen in a urine sample is considered normal. The reliability of screening tests for detection of urobilinogen is questionable because of the instability of urobilinogen.
Blood (Hemoprotein)
Tests for blood in urine detect hematuria, the presence of intact RBCs in urine; hemoglobinuria, the presence of free hemoglobin in urine; and myoglobinuria, the presence of myoglobin in the urine. Hematuria, hemoglobinuria, and myoglobinuria may occur simultaneously. The presence of one does not rule out the others. The urine sediment should also be examined for intact RBCs.
Hematuria
Hematuria usually is a sign of disease causing bleeding somewhere in the urogenital tract, whereas hemoglobinuria usually indicates intravascular hemolysis. Some systemic conditions may also cause hematuria. In very dilute or highly alkaline urine, RBCs often lyse to yield hemoglobin. Therefore in dilute or highly alkaline urine, hemoglobinuria may not be the result of hemoglobin entering the urine through the glomerulus. Ghost cells (the shells of lysed RBCs) may be seen on microscopic examination of sediment if the source of hemoglobin is lysis of RBCs within the excretory pathway or in vitro.
Moderate to large amounts of blood impart a cloudy red, brown, or wine color to urine. Similar colors, but with a transparent appearance that remains after centrifugation, indicate hemoglobinuria. With minute amounts of blood in the urine, a visible color change usually is not evident. Occult, or hidden, blood occurs when the urine is not obviously discolored by blood but blood is detected by chemical analysis. More information on hematuria is found in the section on microscopic examination of urinary sediment.
Hemoglobinuria
Hemoglobinuria is usually the result of intravascular hemolysis. Hemoglobin from RBCs broken down intravascularly is normally bound to the plasma protein haptoglobin. When hemoglobin is bound to haptoglobin, it does not pass through glomeruli. If intravascular hemolysis overwhelms the binding ability of haptoglobin, hemoglobinemia leads to hemoglobinuria because free hemoglobin filters through glomeruli. Hemoglobinuria is indicated by a positive test for hemoglobin without RBCs in the urine sediment, or the degree of the test reaction is often greater than may be accounted for by the numbers of RBCs in the urine sediment. When hemoglobin concentration is sufficiently high in the urine to impart red discoloration, the urine remains red after centrifugation. If the discoloration is from intact RBCs, the urine is clear above the pellet after centrifugation. Partial clearing after centrifugation indicates both hemoglobinuria and hematuria. The presence of hemoglobin (either as free hemoglobin or in RBCs) must be confirmed by urine dipstick test and further evaluation by microscopic examination.
Hemoglobinuria may be seen with many conditions that cause intravascular hemolysis. Conditions that can cause intravascular hemolysis include immune-mediated hemolytic anemia, isoimmune hemolytic disease of neonates, incompatible blood transfusions, leptospirosis, babesiosis, certain heavy metals (e.g., copper), and ingestion of certain poisonous plants. Other conditions that cause hemoglobinuria include severe hypophosphatemia, postparturient hemoglobinemia in cattle, and hemolysis that occurs when cattle drink large quantities of water after being unable to obtain water (e.g., after a long period of low temperatures has frozen their usual water source).
If the urine is dilute or very alkaline, hemoglobinuria can originate from lysis of RBCs in the urine. This condition must be considered hematuria because intact RBCs were initially present. Often ghost RBCs may be found when hemoglobinuria is caused by release of hemoglobin from RBCs in vitro.
Because the test for blood in the urine detects hemoglobinuria, hematuria, and myoglobinuria, other considerations include sediment examination, history, physical examination findings, and additional laboratory procedures to determine the cause of the positive test for blood in the urine.
Myoglobinuria
Myoglobin is a protein found in muscle. Severe muscle damage causes myoglobin to leak from muscle cells into the blood. Myoglobin passes through the glomeruli and is excreted in the urine. Urine containing myoglobin is usually very dark brown to almost black, but at low concentrations the urine may have a similar color to that seen with hemoglobin. Distinguishing myoglobinuria from hemoglobinuria may be difficult. History and clinical findings that suggest muscle damage help determine whether a positive hemoglobin test is due to the presence of myoglobin. Myoglobinuria is frequently seen in horses with exertional rhabdomyolysis.
Several methods have been used to try to distinguish hemoglobin from myoglobin. None of the methods is completely reliable. They sometimes may be differentiated on the basis of their different molecular weights and different solubility in ammonium sulfate.
Leukocytes
Presumptive evidence of leukocytes (WBCs) in urine may be obtained with the leukocyte reaction of certain reagent strips. However, many false-negative reactions occur with animal species, and microscopic evaluation is necessary to confirm a positive result. The leukocyte reagent strip test is not valid for cats because of false-positive results.
MICROSCOPIC EXAMINATION OF URINE SEDIMENT
Microscopic examination of urine sediment is an important part of a complete urinalysis, especially for recognizing diseases of the urinary tract. Many abnormalities in a urine sample cannot be detected with reagent test strips or tablets, but often more specific information may be obtained by observation of the urine sediment. In addition, urine sediment examination is occasionally an aid in diagnosing systemic disease. In human medicine, microscopic analysis of urine sediment is usually performed only when patients are symptomatic or abnormalities are evident on the physical and chemical urine examinations. However, many veterinary practitioners routinely request a urine sediment examination on every urine sample.
With the exception of horse and rabbit urine, normal urine of domestic animals does not contain a large amount of sediment. Small numbers of epithelial cells, mucus threads, RBCs, WBCs, hyaline casts, and crystals of various types can be found in the urine of normal animals. The urine of horses and rabbits usually has large amounts of calcium carbonate crystals. Urine must be collected cleanly because bacteria and aberrant substances may be present in a urine sample that has been contaminated during collection.
The best samples for sediment examination are morning samples or samples collected after several hours of water deprivation. Because such samples are more concentrated, the chances of finding formed elements are increased. Sediment should be examined while the urine is fresh because bacteria will multiply if allowed to stand at room temperature for a period of time. Also, as previously discussed, other changes may occur in a sample as it ages. Crystals may form as the sample cools and casts may dissolve in alkaline urine. If a voided sample is collected, a midstream sample is preferred because it is less likely to be contaminated by cells, bacteria, and debris from the external genital surfaces. Urine collected by cystocentesis is the best sample for microscopic examination. If the sample cannot be examined within 1 hour of collection, it should be refrigerated or preserved.
For semiquantitative measurements of the formed elements in urine, the volume of urine used and the volume of sediment obtained should be recorded. If a sufficient volume has been obtained, 5 to 10 ml of a well-mixed sample should be placed in a graduated, conical centrifuge tube and centrifuged for 3 to 5 minutes at approximately 1000 to 2000 rpm depending on the radius of the centrifuge. Excessive force compacts the sediment and may distort or disrupt formed elements. The procedure should be standardized for a particular centrifuge to yield uniform results. After centrifugation, the volume of sediment is recorded, and the supernatant is gently poured off, leaving approximately 0.5 ml of urine in the bottom of the tube. The sediment is resuspended by gently flicking the bottom of the centrifuge tube with the fingers or by mixing gently with a pipette (Procedure 5-2).
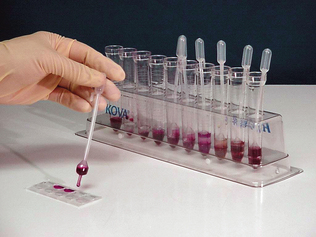
The Kova urine sediment system (Hycor Biomedical Inc., Garden Grove, CA) provides a method for standardization of initial sample volume, the volume of sample used to resuspend the packed sediment, and the distribution of elements on the slide. Each specimen is processed in a specially shaped conical plastic tube with a flared opening for easy filling. When the supernatant is poured off after centrifuging, a fixed volume is retained along with the sediment. The specially designed pipette is then used to dispense a fixed volume of the resuspended sediment into a special chambered slide for microscopic examination (Fig. 5-5). This unique system provides an even distribution of microscopic elements that improves visualization.
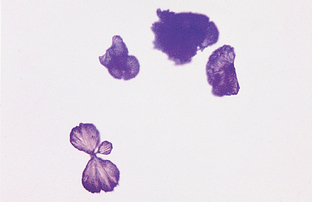
The sediment may be examined stained or unstained. Examining the sediment unstained first allows better evaluation of the specimen. To exam ine unstained sediment, a small drop of the suspended sediment is placed on a clean glass slide, covered with a cover slip, and examined immediately. Subdued light that partially refracts the elements must be used to examine unstained urine sediment. This is achieved by partially closing the diaphragm and adjusting the condenser downward until optimal contrast is achieved. If too much light is present, some structures may be missed. The fine adjustment knob of the microscope should be continuously adjusted to see the depth of the object, as well as other structures. The use of stain in the sediment may help identify different cell types. However, stains often introduce artifacts into the sediment, particularly precipitate material and bacteria. Available urine sediment stains include Sternheimer-Malbin stain (Sedi-Stain, Becton, Dickinson, Franklin Lakes, NJ) (Fig. 5-6) or 0.5% new methylene blue containing a small amount of formalin. One drop of stain is mixed with the suspended sediment before placing a drop of sediment on a microscope slide. A cover slip is placed over the drop of stained sediment. The amount of illumination is less critical when examining a stained specimen than with an unstained one, although reduced illumination also aids visualization of substances by providing contrast. Quantifying elements in the sediment should never be done with a stained slide because the stain dilutes the sample significantly. One method that may simplify the urinalysis procedure is to prepare two drops of urine sediment side by side on the same microscope slide (Fig. 5-7). One drop has stain added and can be used to identify cells while the unstained side is used to quantify elements in the urine.
The specimen must be initially scanned under low power (10× objective) to evaluate the overall quality of the preparation and identify larger elements, such as crystals or aggregates of cells. The entire area under the cover slip should be examined because casts tend to migrate toward the edge of the cover slip. Casts and crystals are identified and reported as the number observed per low power field (lpf). The high power lens (40× objective) is necessary to identify most objects accurately, detect bacteria, and differentiate cell types. A minimum of 10 microscopic fields with a high-power lens should be observed. Epithelial cells, RBCs, and WBCs are reported as the average number observed per high power field (hpf). Bacteria are reported as few, moderate, or many, and their morphologic characteristics (cocci, bacilli) are noted. Alternatively, elements can be reported as a range seen. For example, 1 to 4 cells per high power field would indicate that nearly every microscopic field examined had at least one cell and some had as many as four. Bacteria and crystals may also be semi-quantified using a scale of +1 to +4.
CONSTITUENTS OF URINE SEDIMENT
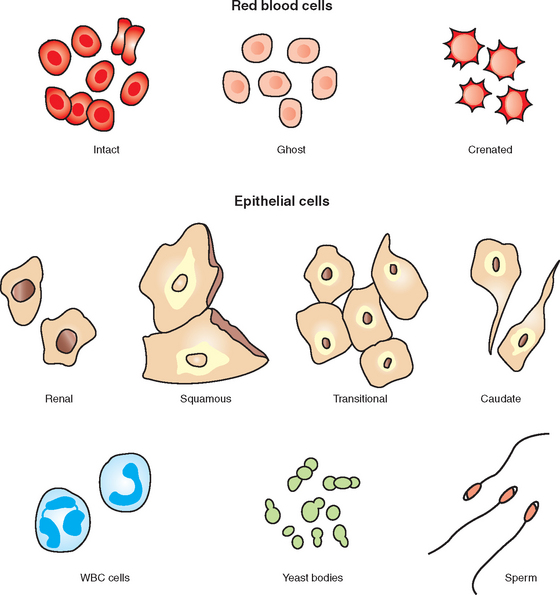
Normal urine sediment in healthy animals may contain a few casts; crystals; epithelial cells; RBCs; WBCs; mucus threads and, in males or recently bred females, spermatozoa. Fat droplets, artifacts, and contaminants may also be seen. If more than a few erythrocytes, leukocytes, hyperplastic and/or neoplastic epithelial cells, casts, crystals, parasite ova, bacteria, and yeast are identified in urine sediment, it is considered abnormal and further diagnostic tests should be performed.
Erythrocytes
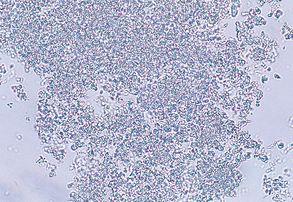
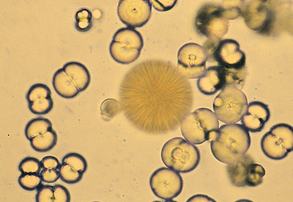
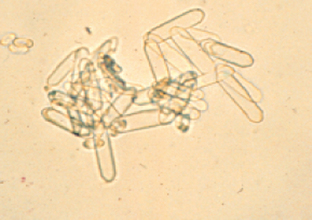
Erythrocytes (RBCs) may have several different appearances depending on the urine concentration, pH, and time elapsed between collection and examination. In a fresh sample, RBCs are small, round, usually smooth edged, somewhat refractile, and yellow or orange, but they may be colorless if their hemoglobin has diffused during standing (Fig. 5-8 and Fig. 5-9). RBCs are smaller than WBCs and may have a smooth, biconcave disk shape (Fig. 5-10). In concentrated urine, RBCs shrink and crenate. Crenated RBCs have ruffled edges, are slightly darker, and may even appear granular as a result of membrane irregularities (see Fig. 5-10). In dilute or alkaline urine, RBCs swell and may lyse. Swollen RBCs have smooth edges and are pale yellow or orange. Lysed RBCs may appear as colorless rings (shadow cells or ghost cells) that vary in size (see Fig. 5-10). However, lysed RBCs, especially when resulting from marked alkalinity, often dissolve and cannot be found on microscopic examination. Normally, urine sediment contains fewer than 2 to 3 RBCs per high power field.
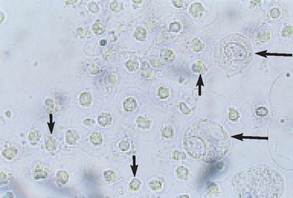
Figure 5-8 Unstained urine showing crenated RBCs (short arrows) and two epithelial cells (long arrows). (Reprinted from Raskin RE, Meyer DJ: Atlas of canine and feline cytology, St Louis, 2001, Saunders.)
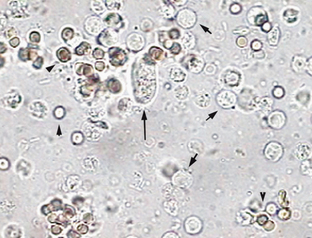
Figure 5-9 Urine sediment with a cast (long arrow) and several RBCs (arrowheads) and WBCs (short arrows). (Reprinted from VanSteenhouse JL: Clinical pathology. In McCurnin DM, Bassert JM, editors: Clinical textbook for veterinary technicians, ed. 6, St Louis, 2006, Saunders.)
Because mammalian RBCs contain no nucleus, they may be confused with fat globules and yeast. However, their light yellow or orange color usually allows them to be differentiated from these other elements. Furthermore, variation in RBC size is minimal, whereas fat globules vary in size. Erythrocytes in urine usually indicate bleeding somewhere in the urogenital tract or occasionally in the genital system. A voided sample from a female in proestrus or estrus or after parturition may be contaminated with RBCs. Both females and males with inflammatory conditions in the genital system may have RBCs in urine collected by free catch or expression of the bladder. Urine collected by catheterization from females with inflammatory lesions in the genital tract usually is not contaminated, but urine from males with genital tract inflammation may be contaminated. Even the slight trauma that occurs from catheterization, cystocentesis, and manual expression of the bladder may slightly increase the number of RBCs in the sediment. Generally, cystocentesis does not cause much increase in RBC numbers. The veterinary technician should note the method of urine collection on the laboratory report to help determine the significance of RBCs in urine.
Leukocytes
Leukocytes (WBCs) are larger than erythrocytes and smaller than renal epithelial cells. Leukocytes are spherical and can appear as a dull gray or greenish-yellow color. They are identified in urine sediment by their characteristic granules or by lobulations of the nucleus (Fig. 5-11). Their appearance is attributable to the fact that most WBCs in urine are neutrophils, which contain a large number of granules. Few leukocytes are found in the urine of animals without urinary or genital tract disease. WBCs shrink in concentrated urine and swell in dilute urine. Leukocytes are usually in low numbers in urine (0 to 1/hpf). Finding more than 2 to 3 per high power field indicates an inflammatory process somewhere in the urinary or genital tracts. The term for excessive WBCs in the urine is pyuria. Pyuria is indicative of an inflammatory or infectious process such as nephritis, pyelonephritis, cystitis, urethritis, or ureteritis. Urine with increased numbers of leukocytes should be cultured for bacteria even if organisms are not observed by microscopic examination.
Epithelial Cells
A few epithelial cells in urine are considered normal and occur as a result of normal sloughing of old cells. A marked increase indicates inflammation. The three types of epithelial cells found in urinary sediment are squamous, transitional, and renal (see Fig. 5-10). Differentiation of transitional from renal epithelial cells is often difficult. In this case, reporting the cells as nonsquamous epithelial cells is acceptable.
Squamous Epithelial Cells
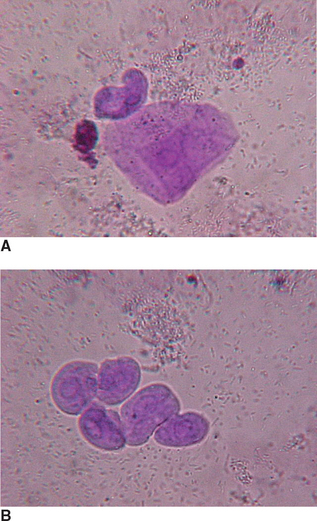
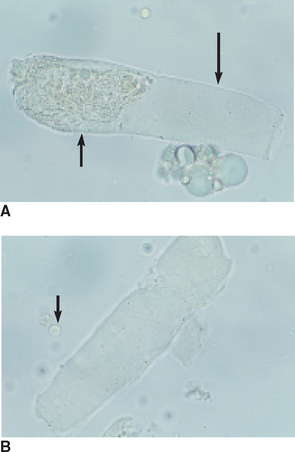
Squamous epithelial cells, derived from the distal urethra, vagina, vulva, or prepuce, are occasionally found in voided samples. Their presence usually is not considered significant. These flat, thin cells with a homogeneous appearance are the largest cells found in urine sediment. They often have straight edges and distinct corners, which sometimes curl or fold (Fig. 5-12, A). They may show a small, round nucleus. Squamous epithelial cells are not normally found in samples obtained by cystocentesis or catheterization.
Transitional Epithelial Cells
Transitional epithelial cells come from the bladder, ureters, renal pelvis, and proximal urethra. They are usually round, but they may be pear shaped or caudate. They are granular, have a small nucleus, and are larger than WBCs (Fig. 5-12, B). Low numbers of transitional cells (0 to 1/hpf) may be found in urinary sediment as a result of sloughing of old cells, but an increased number suggests cystitis or pyelonephritis. Increased numbers also may be seen if catheterization was used to obtain the sample.
Renal Epithelial Cells
Renal epithelial cells are the smallest epithelial cells observed in urine. They originate in the renal tubules, are only slightly larger than WBCs, and are often confused with WBCs. Renal epithelial cells are generally round and contain a large nucleus and nongranular or finely granular cytoplasm (see Fig. 5-10). They are rarely found (0 to 1/hpf). Increased numbers of these cells occur in diseases of the kidney parenchyma.
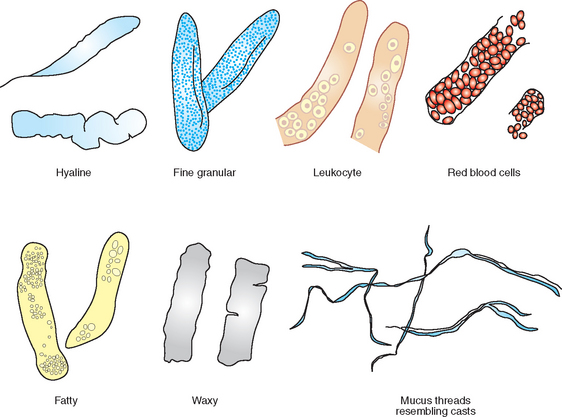
Casts
Casts are formed in the lumen of the distal and collecting tubules of the kidney, where the concentration and acidity of urine are greatest. In the renal tubules, secreted protein precipitates in acidic conditions and forms casts shaped like the tubules in which they form. They are composed of a matrix of protein from plasma and mucoprotein secreted by the tubules. They are commonly classified on the basis of appearance as hyaline, epithelial, cellular (RBCs and/or WBCs), granular, waxy, fatty, and mixed casts. Which cast type is present depends in part on how quickly the filtrate is moving through the tubules and how much tubular damage is present. Faster moving filtrate with minor tubule damage is usually evident as a hyaline cast. Slower moving filtrate allows time for the cells to be incorporated within the cast. If the filtrate is moving very slowly, the cells will degenerate as the cast continues through the tubules; the cast will then appear as a granular cast.
All casts are cylindrical structures, with parallel sides, and their width is determined by the width of the lumen in which they are formed. Their ends may be tapered, irregular, or round. Any cells or structures in the area may also be incorporated into casts, imparting the morphologic features that allow them to be specifically identified (Fig. 5-13). Casts dissolve while in alkaline urine, so cast identification should be performed in fresh samples that have not become alkaline with standing. Because casts dissolve quickly in alkaline urine they are rarely seen in the sediment of herbivores, which characteristically have alkaline urine. Casts may be disrupted with high-speed centrifugation and rough sample handling. A few hyaline casts or granular casts (0 to 1/hpf) may be seen in normal urine, but larger numbers of casts indicate a lesion in the renal tubules. The number of casts observed is not a reliable indicator of the severity of the urinary disease.
Hyaline Casts
Hyaline casts are clear, colorless, and somewhat transparent structures composed only of protein. They are difficult to see and usually are identified only in dim light. Hyaline casts are cylindrical, with parallel sides and usually rounded ends (Fig. 5-14). They are easier to identify in stained sediment than in unstained sediment. Increased numbers of hyaline casts indicate the mildest form of renal irritation. Their numbers also are increased with fever, poor renal perfusion, strenuous exercise, or general anesthesia.
Granular Casts
Granular casts, which are hyaline casts containing granules, are the most common type of cast seen in animals (Fig. 5-15). The granules are from tubular epithelial cells, RBCs, or WBCs that became incorporated in the cast and then degenerated. Cellular degeneration may occur in the tubules producing granular casts, which may be coarse or fine in appearance. Other materials released from cells in the urinary tract may also become embedded in casts. Granular casts are seen in large numbers with acute nephritis and indicate more severe kidney damage than do hyaline casts.
Epithelial Casts
Epithelial casts consist of epithelial cells from the renal tubules imbedded in a hyaline matrix (Fig. 5-16). Epithelial cells in casts are always of the renal epithelial type because this is the only epithelial cell present at the site of cast formation. These casts are formed by epithelium sloughing in the tubules. These casts are seen in acute nephritis or other conditions that cause degeneration of the renal tubular epithelium.
Leukocyte Casts
Leukocyte casts contain WBCs, predominantly neutrophils (see Fig. 5-13). These casts can be readily identified unless cellular degeneration has occurred. The presence of WBCs and leukocyte casts indicates inflammation in the renal tubules.
Erythrocyte Casts
Erythrocyte casts are deep yellow to orange in color. The RBC membranes may or may not be visible. Erythrocyte casts contain RBCs and form when RBCs aggregate within the lumen of the tubule (Fig. 5-17). Erythrocyte casts indicate renal bleeding. Bleeding may be strictly from hemorrhage resulting from trauma or bleeding disorders, or it may occur as part of an inflammatory lesion.
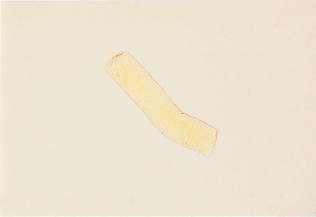
Waxy Casts
Waxy casts resemble hyaline casts but are usually wider, with square ends rather than round ends and a dull, homogeneous, waxy appearance (Fig. 5-18). They are colorless or gray and highly refractile. They indicate chronic, severe degeneration of the renal tubules.
Fatty Casts
Fatty casts contain many small droplets of fat that appear as refractile bodies (see Fig. 5-13). They are frequently seen in cats with renal disease because cats have lipid in their renal parenchyma. They are occasionally seen in dogs with diabetes mellitus. Large numbers of fatty casts suggest degeneration of the renal tubules.
Crystals
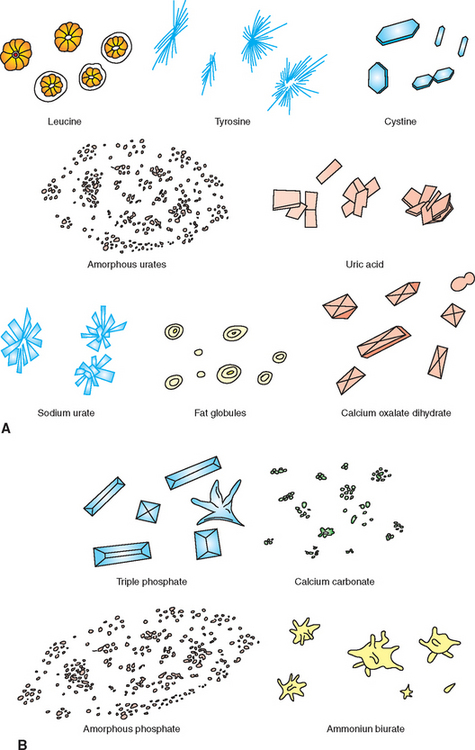
The presence of crystals in the urine is termed crystalluria. Crystalluria may or may not be of clinical significance. Certain crystals form as a consequence of their elements being secreted into the urine by normal renal activity (Fig. 5-19). Some crystals form as a consequence of metabolic diseases. Conditions that lead to crystal formation may also cause formation of urinary calculi. The type of crystals formed depends on the urine pH, concentration and temperature, and the solubility of the elements (see Table 5-4). If a urine sample is allowed to stand and cool before examination, the number of crystals in the sample increases because the materials that make up crystals are less soluble at lower temperatures. Refrigerated samples often have many more crystals than warm, fresh samples. Sometimes crystals dissolve when a refrigerated sample is warmed to room temperature. Crystals are generally reported as occasional, moderate, or many or as +1 to +4. Although crystals (and uroliths) are often identified by their morphologic characteristics, the only definitive methods to identify crystals is with x-ray diffraction or chemical analysis.
TABLE 5-4
| Crystal | pH |
| Ammonium biurate | Slightly acidic, neutral, alkaline |
| Amorphous phosphate | Neutral, alkaline |
| Amorphous urates | Acidic, neutral |
| Bilirubin | Acidic |
| Calcium carbonate | Neutral, alkaline |
| Calcium oxalate | Acidic, neutral, alkaline |
| Cystine | Acidic |
| Leucine | Acidic |
| Triple phosphate | Slightly acidic, neutral, alkaline |
| Tyrosine | Acidic |
| Uric acid | Acidic |
Struvite
Struvite crystals are sometimes referred to as triple phosphate crystals or magnesium ammonium phosphate crystals. They are found in alkaline to slightly acidic urine. Generally, struvite crystals are six-to eight-sided prisms, with tapering sides and ends (Figs. 5-20 and 5-21). Struvite crystals typically are described as resembling coffin lids, although they may take on other shapes. Occasionally they may assume a fern-leaf shape, especially when the urine contains a high concentration of ammonia.
Amorphous Phosphate
Amorphous phosphate crystals are common in alkaline urine and appear as a granular precipitate (Fig. 5-22).
Calcium Carbonate
Calcium carbonate crystals are commonly seen in the urine of horses and rabbits. They are round, with many lines radiating from their centers, or appear as large granular masses (Fig. 5-23). They also may have a “dumbbell” shape. They are of no clinical significance.
Amorphous Urates
Amorphous urates appear as a granular precipitate similar to amorphous phosphates (see Fig. 5-15 and Fig. 5-27). Amorphous urates are seen in acidic urine, whereas amorphous phosphates are found in alkaline urine.
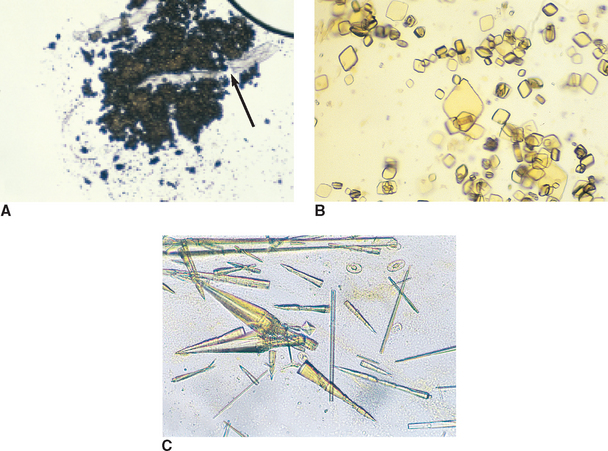
Figure 5-27 A, Amorphous urate crystals. A cotton fiber (contaminant) is trapped within the crystals (arrow). B, Uric acid crystals. These are not commonly found in small animals except for dalmatian dogs. C, Sodium urate crystals. May be found in association with ammonium biurate uroliths. A calcium oxalate dihyrate crystals is also present (center). (From Raskin RE, Meyer DJ: Atlas of canine and feline cytology, St Louis, 2001, Saunders.)
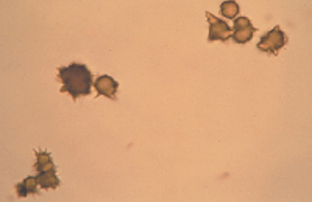
Ammonium Biurate
Ammonium biurate crystals are seen in slightly acidic, neutral, or alkaline urine. These crystals are brown in color and round with long, irregular spicules (“thorn apple” shape) (Fig. 5-24; also see Fig. 5-19). Often the spicules fracture and the remaining crystal is brown, with fine radiating lines. They are most common in animals with severe liver disease, such as portacaval shunts.
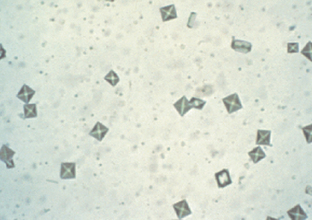
Calcium Oxalate
Calcium oxalate dihydrate crystals generally appear as small squares, containing an “X” across the crystal resembling the back of an envelope (Fig. 5-25). Calcium oxalate monohydrate crystals may be small and dumbbell shaped or they may be elongated and pointed at each end (resembling a slat from a picket fence) (Fig. 5-26). Calcium dihydrate crystals are found in acidic and neutral urine and are commonly seen in small numbers in dogs and horses. The urine of animals poisoned with ethylene glycol (antifreeze) often contains large numbers of calcium oxalate crystals, especially calcium monohydrate crystals. Animals with oxalate urolithiasis may have large numbers of calcium oxalate crystals in their urine, and large numbers of oxalate crystals may indicate predisposition to oxalate urolithiasis.
Sulfonamide
Sulfonamide crystals may be seen in animals being treated with sulfonamides. Sulfonamide crystals are round, usually dark, with individual crystals radiating from the center. They are less likely to be observed in alkaline urine because these crystals are more soluble in alkaline urine. Prevention of precipitation of these crystals in the renal tubules is assisted by maintaining alkaline urine and encouraging the animal to drink.
Uric Acid
Uric acid crystals take on a variety of shapes but are usually diamond or rhomboid (Fig. 5-27). They appear yellow or yellow-brown in color and are not commonly found in the dog and cat except in dalmatian dogs.
Leucine
Leucine crystals are wheel or “pincushion” shaped and are yellow or brown in color (see Fig. 5-15). Animals with liver disease may have leucine crystals in their urine.
Tyrosine
Tyrosine crystals are dark, have needlelike projections, and are highly refractile (Fig. 5-28). They are often found in small clusters. Animals with liver disease may have tyrosine crystals in their urine. They are not a common finding in the dog and cat.
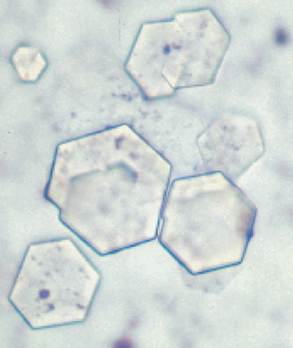
Cystine
Cystine crystals appear flat and are six-sided (hexagonal), colorless, and thin (Fig. 5-29). They can be associated with renal tubular dysfunction or cystine urolithiasis.
MICROORGANISMS
A variety of microorganisms may be found in urine sediment, including bacteria, fungi, and protozoa. Normal urine is free of bacteria but may be contaminated by bacteria residing on the epithelium of the vagina, vulva, or prepuce during urination. Normal urine collected by cystocentesis or catheterization does not contain bacteria and therefore is considered sterile. Because bacteria often proliferate in urine that has been left standing for some time, especially at room temperature, the urine must be immediately examined or refrigerated until it can be examined. Bacteria can be identified only under magnification. They may be round (cocci) or rod shaped (bacilli), usually refract light, and appear to be quivering as a result of Brownian movement (see Fig. 5-11). They are reported as few, moderate, many, or too numerous to count (TNTC). A large number of bacteria accompanied by a large number of WBCs suggests infection and inflammation of the urinary tract (e.g., cystitis, pyelonephritis) or genital tract (e.g., prostatitis, metritis, or vaginitis). Bacteria in the urine sample are most significant when they are also identified within the cytoplasm of the WBCs. These samples should be submitted for bacterial culture.
Yeasts are often confused with RBCs or lipid droplets, but they usually display characteristic budding and may have double refractile walls. Yeast usually are contaminants in urine samples because yeast infections of the urinary tract are rare in domestic animals. Yeast infection of the external genitalia may cause yeast to be present in voided samples. Fungi also may be found in urine. Fungi are filamentous and usually branching. Fungal infections of the urinary tract are uncommon but are quite serious when they occur.
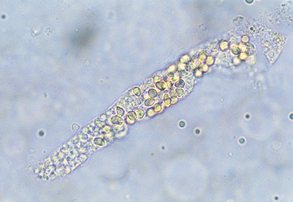
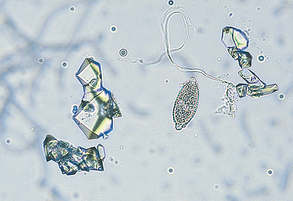
Parasite Ova, Microfilaria
Parasite ova may be seen in the urine sediment of animals with urinary parasites or because of fecal contamination at the time of collection of the urine sample. Some parasites of the urinary tract include Capillaria plica, a bladder worm of dogs and cats (Fig. 5-30), and Dioctophyma renale, a kidney worm of dogs. Microfilaria (e.g., Dirofilaria immitis) may be seen in the urine sediment of dogs with adult heartworms, and circulating microfilaria may be seen if hemorrhage into the urine occurs either from disease or as a result of trauma during collection (Fig. 5-31).
MISCELLANEOUS COMPONENTS OF URINE
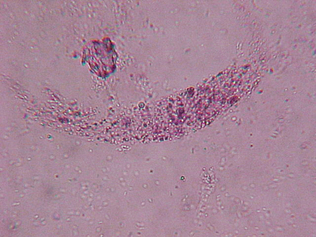
Mucus threads are often confused with casts, but they do not have the well-delineated edges of casts. They resemble a twisted ribbon more than a cast (see Fig. 5-13). A large amount of mucus is normally present in equine urine because horses have mucus glands in the renal pelvis and ureter. In other animals, mucus indicates urethral irritation or contamination of the sample with genital secretions.
Spermatozoa
Spermatozoa are occasionally seen in the urine sediment of intact male animals (see Fig. 5-10). They are easily recognized and have no clinical significance. Sperm may also be present in recently bred females. Large amounts of sperm in urine may produce false positive results for proteins.
Fat Droplets
In urine sediment, fat droplets are lightly green-tinged, highly refractile, spherical bodies of varying sizes. Because they vary in size, they can be distinguished from RBCs and yeast, which tend to be uniform in size. If a sediment smear sits for a few moments before being examined, fat droplets rise to a plane just beneath the cover slip, whereas other formed elements settle to the top of the slide. Therefore fat droplets are often not in the plane of focus of other formed elements. Small, round structures found under the cover slip are usually fat globules. Uniformly sized, round structures found in a lower plane are usually RBCs. In sediment stained with Sudan III stain, fat droplets appear orange or red in color. Frequently, fat droplets from catheter lubricants or from oily surfaces of collecting vials and pipettes may contaminate urine. Fat in the urine, called lipuria, is seen to some degree in most cats. Lipuria is also seen with obesity, diabetes mellitus, hypothyroidism, and rarely, after a high-fat meal.
Artifacts
Many artifacts may enter the urine sample during collection, transportation, or examination. Recognition of these structures as irrelevant and not a normal part of the sediment evaluation. These contaminants may be a source of great confusion.
Air bubbles, oil droplets (usually resulting from lubricated catheters), starch granules (from surgical gloves), hair, fecal material, plant spores, pollen, cotton fiber, dust, glass particles or chips, bacteria, and fungi may contaminate urine. Ova of intestinal parasites may be observed as a result of fecal contamination of the urine sample.
UROLITHIASIS
Uroliths are calculi (stones) composed of various minerals that are found anywhere in the urinary tract; their occurrence is termed urolithiasis. They may cause blockage of urine outflow from the bladder into the urethra; lodge in the urethra causing severe, acute inability to urinate; or remain in the bladder and cause inflammation and bleeding. Determining the composition of calculi is critical because their prevention and the animal’s prognosis depend on identification of their composition. Once composition is determined, proper therapy may be initiated to remove them and prevent their reoccurrence. Urolithiasis may be a particular problem in castrated male ruminants. Lodging of calculi in the urethra obstructs the outflow of urine, which is a major problem in lambs and steers, particularly those that are fed high-concentrate rations. The most common calculi in these species are composed of calcium, magnesium, and ammonium carbonate or calcium, magnesium, and ammonium phosphate.
Analysis of the mineral composition of uroliths may be determined by submitting it intact to a reference laboratory for quantitative analysis. Occasionally, a reasonable idea about the composition of a urolith may be made by its gross and radiographic appearance and the crystal types found in the sediment. Uroliths of dogs and cats are usually struvite. Cystine and oxalate uroliths may also be observed. Urate uroliths are seen mostly in Dalmatians because this breed excretes large amounts of uric acid.
Recommended Reading
Cowell, RL, Tyler, RD, Meinkoth, JH. Diagnostic cytology and hematology of the dog and cat, ed 2. St Louis: Mosby, 1999.
Graff, L. A handbook of routine urinalysis. Philadelphia: Lippincott, 1983.
Meyer, DJ, Harvey, JW. Veterinary laboratory medicine interpretation and diagnosis, ed 3. St Louis: Saunders, 2004.
Modern urine chemistry. Elkhart, IN: Miles Laboratories, 1993.
Osborne, CA, Stevens, JB. Urinalysis: a clinical guide to compassionate patient care. Shawnee Mission, KS: Bayer, 1999.
Raskin, RE, Meyer, DJ. Atlas of canine and feline cytology. St Louis: Saunders, 2001.