Immunology, Serology, and Molecular Diagnostics
This chapter reviews basic principles of the immune system; some of its disorders; the practical applications of immunology, including vaccinations and serology tests; and molecular diagnostic tests.
THE IMMUNE RESPONSE
Vertebrate species have two major internal defense systems: the innate, or nonspecific, immune system and the adaptive, or specific, immune system (also called acquired immunity).
Innate Immune System
Foreign bodies, such as bacteria, viruses, and fungi, first encounter barriers of the innate immune system. These barriers include the skin; physical and biochemical components in the nasopharynx, gut, lungs, and genitourinary tract; and populations of commensal bacteria that compete with invading pathogens and the body’s inflammatory response. The inflammatory response is a response to infection or tissue injury. Alerted by chemicals released from the infected site, blood vessels dilate and allow neutrophils to pass into tissue where they phagocytize infectious agents and kill the pathogens with chemicals stored in their cytoplasm. The classic signs of inflammation are pain, heat, redness, swelling, and loss of function. Each of these is related to physiologic mechanisms taking place in the inflammatory process. Inflammation is a protective mechanism of the innate immune system, but it can overreact and actually cause tissue damage itself.
Monocytes also follow neutrophils to inflammatory sites. Here, like neutrophils, they ingest and destroy inert particles, viruses, bacteria, and cellular debris by phagocytosis. In the blood they are called monocytes, but when they migrate to various tissues and organs they become macrophages and are given specialized names. They locate in connective tissue, liver, brain, lung, spleen, bone marrow, and lymph nodes and together make up the mononuclear phagocytic system.
In addition to the phagocytic cells, natural killer (NK) cells, interferons, and the complement system are important components of the innate immune system. NK cells are not T or B lymphocytes, but rather a small subset of lymphocytes found in the blood and peripheral lymphoid organs. NK cells recognize and destroy host cells that are infected with microbes, such as viruses. They also activate phagocytes by releasing interferon-γ. Interferons are cytokines (soluble proteins secreted by cells to mediate immune responses) that elicit other cellular reactions, such as prevention of viral replication, and influence the actions of NK cells. They are also active in the adaptive immune response.
The complement system consists of a group of proteins found in the blood. Collectively they are referred to as complement and are integral in both the innate and adaptive systems. When activated, a series of chemical reactions, known as the complement cascade, occurs. The system can be activated through one of three pathways, but the later steps are the same for all pathways. The components of the complement system are numbered C1 through C9, with some having several subunits designated by letters.
The classical pathway, a mechanism of the adaptive immune system, is activated when C1 is bound to an antigen-antibody complex. The other pathways of complement activation are part of the innate immune system and are triggered by microbial surfaces and plasma lectins that bind to microbes. All three initial pathways catalyze a series of reactions of other complement molecules that have numerous physiologic effects. These include opsonization of the microbes to promote phagocytosis, the stimulation of inflammation and cell lysis by the formation of a membrane attack complex on the surface of the antigen.
Adaptive Immune System
If foreign bodies evade the innate immune system, they then encounter the adaptive immune system, which is more sophisticated. The adaptive immune system is divided into two components: the humoral immune system and the cell-mediated immune system. The adaptive immune system has the ability to respond specifically to foreign substances. These substances, or antigens, may be bacterial, viral, fungal, or altered endogenous cells of the host’s body. Their presence initiates humoral and cellular responses that neutralize, detoxify, and eliminate these foreign materials from the host.
Lymphocytes and their progeny are the cell types largely responsible for the adaptive immune system. This line of defense is not, however, divorced from the innate immune system. Macrophages process antigens and present them to antigen-committed lymphocytes. That is, they act as antigen-presenting cells.
Lymphoid stem cells develop first in the yolk sac and then in the fetal liver. The bone marrow assumes this responsibility near parturition and serves as the source of these cells throughout postnatal life. The lymphoid stem cells are destined to further mature in one of two places: the bone marrow or the thymus. B lymphocytes mature in the bone marrow, whereas T lymphocytes mature in the thymus (Fig. 8-1).
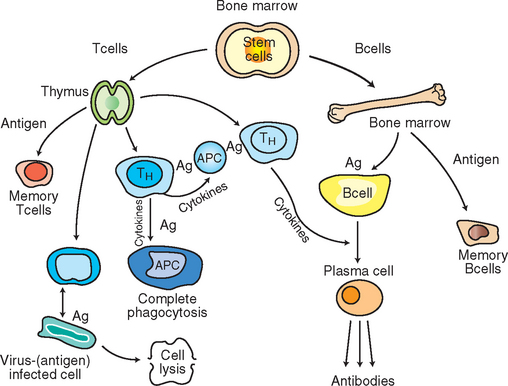
Figure 8-1 Pathways of lymphoid cells in the immune response. Stem cells in the bonemarrow give rise to T and B lymphocytes. Lymphocytes mature in the thymus and bone marrow. Exposure to antigens causes lymphocytes to differentiate and proliferate into memory and effector cells. Ag, Antigen; TH, T-helper lymphocytes; TC, T-cytotoxic lymphocyte.
Humoral Immune System
Lymphocytes that mature in the bone marrow (B cells) are concerned chiefly with production and secretion of immunoglobulin (Ig) molecules, which are also known as antibodies. This is referred to as humoral immunity because the antibodies are secreted into the body’s fluids or “humors”. Their maturation process consists of three stages: the lymphoblast, the prolymphocyte, and the mature lymphocyte. The mature cells leave the bone marrow to seed secondary lymphoid organs, chiefly the spleen and lymph nodes, where they encounter antigens. The humoral immune system can recognize billions of different antigens because as B cells mature, each B cell develops a specific receptor molecule to a specific antigen. When an antigen enters the body, a mature B cell that is committed to that particular antigen will react with it. Stimulation of that B cell to produce antibodies is a complex process, requiring the help of specialized T lymphocytes called helper T cells. Helper T cells produce cytokines that activate the B cells. The antigen-stimulated B cell then quickly divides and differentiates, producing a clone of identical B cells that all produce the same type of antigen-specific antibody. These antibody-secreting B cells are now called plasma cells, a type of effector cell. An effector cell is a cell of the immune system that performs specific functions to destroy foreign antigen. Some of the antigen-stimulated B cells differentiate into memory B cells, which respond faster to a second exposure of that antigen (see Fig. 8-1).
Antibodies (immunoglobulins) are protein molecules consisting of two pairs of polypeptide chains configured in a Y shape (Fig. 8-2). Each immunoglobulin (Ig) molecule contains two variable regions and one constant region. The variable regions (Fab) bind to the antigen, and the constant region (Fc) is responsible for the unique functions of the different antibody classes.
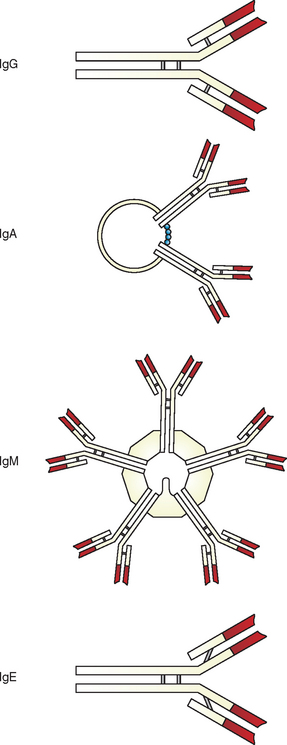
Figure 8-2 Schematic representation of IgM (pentamer), IgG and IgE (monomers), and IgA (dimer). (From Gershwin L, et al: Immunology and immunopathology of domestic animals, ed 2, St Louis, 1995, Mosby.)
Five distinct classes of immunoglobulins are produced: IgM, IgG, IgE, IgA, and IgD. The first antibody type produced in response to an antigen is IgM. IgM is a pentameric molecule (i.e., contains five monomers) and comprises approximately 5% of circulating immunoglobulin. IgM is relatively large and is therefore unable to enter tissue spaces. The most abundant circulating immunoglobulin is IgG. It comprises approximately 75% of circulating immunoglobulin and remains in circulation longest. IgG is a relatively small monomer that is capable of entering tissue spaces and is usually produced during a secondary immune response. IgE is usu-ally present in very small amounts and is similar in structure to IgG. IgA comprises approximately 20% of circulating antibody, and IgD is a monomer that, when present, is in very low abundance.
The following are the functions of the different classes:
IgG: neutralization of microbes and toxins; opsonization of microbes for phagocytosis by macrophages and neutrophils; activation of complement; fetal and neonatal immunity by passive transfer across placenta and in colostrums
IgE: immediate hypersensitivity reactions, such as allergies and anaphylactic shock; coating of helminth parasites for destruction by eosinophils
IgA: mucosal immunity; protection of respiratory, intestinal, and urogenital tracts
Antibodies interact with antigens in different ways to prevent antigenic attachment or invasion of body cells. A neutralization antibody reaction occurs when antibody binds directly with the antigen. For example, if a foreign microbe or microbial toxin is bound by antibody, it cannot infect or damage body cells. This essentially neutralizes the potential effect of the antigen. Sometimes antibodies coat microbes. The Fab region of the antibody attaches to receptors on the microbial surface. The Fc region of the antibody then binds to macrophages or neutrophils and the microbe is phagocytized. If the antigenic material is a helminth parasite, IgE antibodies opsonize the worms; but instead of phagocytosis, which would be ineffectual against the large worms, eosinophils bind to and destroy the parasites. When complement is activated by some antibodies the end result is antigenic cell lysis.
Precipitation reactions occur when antigens bind with antibodies and form an insoluble complex. This precipitate forms on surfaces and the precipitant itself may cause pathology. For exam-ple, precipitation of bacterial fragments in the glomerular membrane can result in glomerular nephritis, described later as a type III hypersensitivity reaction.
Cell-Mediated Immune System
Lymphoid stem cells that mature in the thymus develop into T-cell lymphocytes. Like that of B cells, their maturation process consists of three morphologically distinct stages: lymphoblast, prolymphocyte, and lymphocyte. As these cells mature, they also develop receptors to specific antigens and become immunocompetent or antigen-committed T lymphocytes. Some references refer to T and B cells at this stage as naive lymphocytes. Then, after contact with their specific antigens, these cells proliferate and differentiate into either clones of memory cells or clones of effector cells against those antigens.
Memory cells recognize antigens to which they have previously been exposed. On a subsequent encounter, they elicit a more rapid immune response.
Different types of T-effector cells exist, such as helper T cells (CD4+) and cytolytic T cells (CD8+). CD4+ and CD8+ refer to surface molecules, or markers, found on helper T and cytolytic T lymphocytes, respectively. Cytolytic T lymphocytes are also called cytotoxic T lymphocytes. HIV (human immunodeficiency virus), the AIDS virus, has a special affinity for the helper T lymphocytes.
Helper T lymphocytes recognize antigen that has been phagocytized by an antigen-presenting cell (APC), such as a macrophage. The APC displays a portion of the antigen on its surface and presents it to the helper T lymphocyte. This stimulates the helper lymphocyte to release cytokines. These cytokines then in turn are the chemical signal that helps the APC to further phagocytize the ingested microbe. T helpers, when stimulated, also release cytokines that help B cells differentiate into antibody-producing cells.
Cytolytic T lymphocytes recognize antigen particles that are on the surface of infected body cells and are able to lyse and kill the infected cells. Microbe infected cells, tumor cells, and cells of foreign tissue graft all may be eliminated in this manner (see Fig. 8-1).
Immunologic Tolerance
One of the most important features of an animal’s immune system is that it does not destroy its own cells. This may seem obvious, but it can actually happen. Maturing lymphocytes develop antigen receptors for foreign antigens but also for the animal’s antigens on its own cells. Therefore these self-reactive lymphocytes could attack the self-antigens. However, in the healthy animal, mechanisms normally are in place that prevent this self-destruction. The immune system can discriminate between self and non-self, which results in immunologic tolerance.
Invading microbes typically are immunogenic; that is, they will interact with their specific naive lymphocytes, which then proliferate and differentiate into effector cells that destroy the foreign microbes. However, to tolerate self-antigens, the animal relies on mechanisms such as antigenic tolerance and ignorance. Self-antigens are normally tolerogenic; the lymphocytes are either unable to respond when they encounter self-antigens (anergy) or they die when they encounter self-antigens (apoptosis). Self-antigens may also be ignored by the naive lymphocytes, in which case the self-antigens are called nonimmunogenic.
These mechanisms are elaborate. When naive lymphocytes are destroyed by apoptosis, the immune system is in effect selecting for the beneficial lymphocytes that have receptors for foreign antigens and eliminating the self-lymphocytes that would cause self-destruction. This is called negative selection and takes place in the bone marrow, thymus, and peripheral lymphoid tissues.
Another mechanism of immunologic tolerance is through the activity of regulatory lymphocytes. Some T lymphocytes, formerly called “suppressor T cells,” become regulatory lymphocytes. Regulatory T cells prevent self-reactive lymphocytes from differentiating into effector cells. They are unable to destroy self-antigens.
This is an oversimplification of the intricacies involved in how immunologic tolerance is maintained. When these mechanisms fail, autoimmune disease results and the animal’s immune system is directed against itself.
DISORDERS OF THE IMMUNE SYSTEM
Some immune responses have an adverse effect on the host animal. The immune responses are uncontrolled, or hypersensitive, and cause tissue injury. Four types of hypersensitivity diseases have been categorized. Type I hypersensitivity is an immediate hypersensitivity that occurs when chemical mediators from mast cells are released. Allergies (atopy) and anaphylactic shock, a severe reaction that may occur within seconds after an antigen enters the circulation, are type I hypersensitivity diseases.
Autoimmune hemolytic anemia, a condition causing destruction of red blood cells (RBCs) by the host itself, is a type II hypersensitivity. These are antibody-mediated diseases in which the antibodies are directed against the animal’s own cells.
Immune complex disease, or type III hypersensitivity, occurs when antibodies and antigens form complexes that deposit in various blood vessels. Glomerulonephritis, caused by deposition of antibody-antigen complexes in the kidney, is an example of type III hypersensitivity.
Type IV hypersensitivity is T cell–mediated disease caused by the reaction of T lymphocytes against self-antigens in tissues. Contact hypersensitivity reactions, such as those that may occur in dogs from contact with plastic in food dishes and collars or in human beings from contact with poison ivy, cause tissue injury in a delayed response. The chemicals from these substances react with skin proteins, and the immune system recognizes this chemical-protein complex as foreign, resulting in dermatitis.
In addition to hypersensitivity reactions, the immune system also may show deficiencies. A deficiency may exist in phagocytes or in immunoglobulins. A condition called combined immunodeficiency affects animals in early life, after serum levels of maternally derived antibodies have declined. Arabian foals with this disease often die from opportunistic infection resulting from an absence or deficiency of immunoglobulins.
Lymphoma, a type of tumor characterized by uncontrolled proliferation of lymphocytes, is another abnormality of the immune system. The immune system normally recognizes and destroys cancer cells before they become established in the body, but sometimes the cancer seems to become resistant and escapes the immune defense mechanisms.
Passive Immunity
Animals become passively resistant to disease by receiving maternal antibodies in the colostrum or by receiving preformed antibodies by injection. These antibodies have been produced in a donor animal. A donor animal is vaccinated with a pathogen. When its serum antibodies reach a high concentration, the animal is bled and the globulin portion containing the antibodies is separated and purified. The protection that an animal receives from an injection of this immunoglobulin is short-lived but immediate.
Immunization
Animals become actively resistant to disease by having the disease and developing antibodies, or by being vaccinated or immunized, in which case they also develop their own antibodies. Immunization is accomplished by injecting a suspension of microorganisms into an animal for the purpose of eliciting an antibody response but not causing the disease. The microorganisms may be either attenuated (weakened but still alive) or inactivated (killed). Attenuated vaccines normally cause a longer-lasting, more potent immune response. Inactivated vaccines are generally safer and have less ability to cause disease, although vaccine-associated sarcomas in cats have been an issue. An adjuvant may be added to the vaccine to enhance the normal immune response. Some adjuvants do this by simply slowing the rate of antigen elimination from the body so that the antigen is present longer to stimulate antibody production. Killed vaccines require more adjuvant; the increased adjuvant is thought to cause the sarcomas.
Effective DNA vaccines are now being developed by using molecular genetics. They are expected to be safer than traditional vaccines and can be made more quickly. Once the technology is in place, more DNA vaccines should be developed quickly.
Vaccines may be given subcutaneously or intramuscularly depending on the vaccine. Other vaccines are aerosolized and given intranasally. Some vaccines are put in the feed or drinking water. Veterinary technicians working at fish hatcheries may vaccinate fish by putting the vaccine in their water.
TESTS OF HUMORAL IMMUNITY
The science of detection and measurement of antibodies or antigens is called serology. Detection depends on the binding of antibodies and antigens. Unfortunately, this binding phenomenon is ordinarily invisible. Visualization, and thus detection, of the antigen-antibody reaction depends on secondary events by which the union is easily detected and therefore of diagnostic use in the veterinary practice.
Commercial production of monoclonal antibodies to many different antigens has resulted in a variety of test kits for use in the veterinary laboratory. These specific antibodies to many different antigens can be produced and used in the laboratory for rapid identification of disease-producing organisms.
Immunization with viruses, bacteria, or other entities stimulates antibody production in an animal. The antibody-secreting, transformed lymphocytes (plasma cells) may be isolated from the animal and chemically fused with a type of “immortal” cell that propagates indefinitely, such as mouse myeloma cells. The antibodies these hybrid cells produce, called monoclonal antibodies, are collected. Because each monoclonal antibody attaches to only one specific part of one type of molecule (antigen), use of these antibodies in diagnostic kits makes the tests specific and greatly reduces interpretation problems of the result. For example, the feline leukemia virus antigen reacts with only the feline leukemia virus antibody. A specific reaction is diagnostically significant for this complicated disease. In addition to their specificity, these procedures allow rapid identification of the pathogen.
Many serologic tests use monoclonal antibodies. Enzyme immunoassay, latex agglutination, immunodiffusion, and rapid immunomigration are methods used in veterinary laboratories. Other methods, such as complement fixation, immunofluorescence, immunoelectron microscopy, virus neutralization, and polymerase chain reaction (PCR) DNA amplification are used in veterinary reference laboratories and research facilities and are not included in this discussion, except for a brief explanation of PCR. Many of the principles are the same, however, and understanding a few procedures gives readers a good basis for understanding others.
Reference laboratories offer myriad serologic tests specifically developed for veterinary samples. Tests for blood types, allergies, bovine leukemia virus, reproductive hormones, Lyme disease, and brucellosis are a few of the diagnostic tests available (Table 8-1).
TABLE 8-1
Commercially Available Immunologic Test Kits

DMS Laboratories, Inc., 2 Darts Mill Road, Flemington, NJ 08822. Synbiotics Corporation, 11011 Via Frontera, San Diego, CA 92127. IDEXX Laboratories, One Idexx Drive, Westbrook, ME 04092. Heska Corporation, 3760 Rocky Mountain Ave., Loveland, CO 80538. ImmuCell Corporation, 56 Evergreen Drive, Portland, ME 04103. Intervet Inc., 405 State Street, P.O. Box 318, Millsboro, DE 19966.
Enzyme-Linked Immunosorbent Assay
The enzyme-linked immunosorbent assay (ELISA) has been adapted to many agents commonly tested for in the veterinary laboratory (Figs. 8-3 to 8-7). With monoclonal antibodies, the specificity of ELISA is high; that is, little cross-reactivity occurs with other agents. This phenomenon makes ELISA an accurate way to detect specific antigens such as viruses, bacteria, parasites, or hormones in serum. ELISA also may be used to test for an antibody in the serum, in which case the test kit contains the specific antigen. Some of the available ELISA kits detect heartworms, feline leukemia virus, feline immunodeficiency virus, canine parvovirus, and progesterone. For the ELISA antigen detection system, monoclonal antibody is bound to the walls of wells in a test tray, to a membrane, or to a plastic wand (Boxes 8-1 to 8-3). Antigen, if present in the sample, binds to this antibody and to a second enzyme-labeled antibody that is added to aid in detection of the antigen. This is followed by rinsing. When a chromogenic (color-producing) substrate is added to the mixture, it reacts with the enzyme to develop a specific color, indicating the presence of antigen in the sample. If the sample contained no antigen, the entire enzyme-labeled antibody was washed away in the rinsing process and no color reaction develops.
Figure 8-3 A critical step in the Microwell enzyme immunoassay is washing away the unbound enzyme-labeled antibodies. (Pet Check Heartworm PF Antigen Test Kit, courtesy IDEXX Laboratories, Westbrook, ME.)
Figure 8-4 A positive test result indicated on the ELISA membrane format (SNAP Test for Ehrlichia antibodies, IDEXX). A positive control spot is also seen. (Courtesy IDEXX Laboratories, Westbrook, ME.)
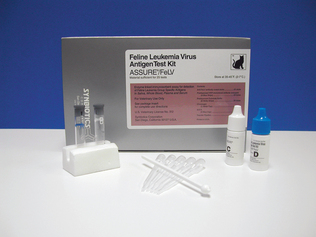
Figure 8-5 Kit to detect feline leukemia antigens in cat serum (Assure/FeLV, Synbiotics) based on the ELISA wand format. (Courtesy Synbiotics Corporation, San Diego, CA.)

Figure 8-6 With the ELISA wells format for the determination of progesterone in canine serum (Ovuchek Premate, Synbiotics), a positive reaction is indicated by color development. (Courtesy Synbiotics Corporation, San Diego, CA.)
Figure 8-7 Clumped latex particles representing antigen-antibody complexes. Samples 1 and 2 indicate a positive reaction. Samples 3 and 4, showing no clumping, indicate a negative reaction.
A similar procedure is used for ELISA antibody detection. In this procedure, antigen is bound to the wells, membrane, or wand, and the patient sample is assayed for the presence of a specific antibody.
Competitive Enzyme-Linked Immunosorbent Assay
The competitive ELISA (CELISA), when used to test for patient antigen, uses an enzyme-labeled antigen, as well as monoclonal antibodies. Patient antigen, if present, competes with enzyme-labeled antigens for the antibodies coating the test wells. Color developer reacts with the enzyme to produce a color. The intensity of the color produced varies with the concentration of the patient antigen (Box 8-4). Equine infectious anemia antibodies may be detected in horse serum with a CELISA test.
Latex Agglutination
The latex agglutination test uses small, spherical latex particles coated with antigen suspended in water. If serum containing the corresponding antibody is added to the mixture, formation of antibody-antigen complexes causes agglutination (clumping). Agglutination changes the appearance of the latex suspension from smooth and milky to clumpy because the latex particles have clustered together. If no antibody is present in the sample, the mixture of latex and serum remains evenly dispersed. Bovine serum may be tested for brucellosis antibodies with this method (Fig. 8-8 and Box 8-5; also see Table 8-1).
Rapid Immunomigration
Rapid immunomigration (RIM) is a technology that historically has been called immunochromatography or lateral flow immunoassay. The signal-generating component of this test is colloidal gold. Its counterparts in other technologies are enzymes and color reagent or agglutinated latex particles. All three types of components create a positive result.
In this type of procedure colloidal gold is conjugated to antibodies specific for the antigen being tested. These conjugated antibodies are present in the membrane of the test cassette where the patient sample is applied. If antigens are present in the patient sample, they bind to the conjugated antibodies and the antibody-antigen complexes migrate along the membrane to another area of the cassette, where the results are read. Buffer may be added to aid the migrational flow of the antibody-antigen complexes. In the reading area a second antibody is present in the membrane. If antigen is in the sample it is captured, along with the first antibody and the colloidal gold, by the second antibody. The accumulation of colloidal gold in that area causes a color change. To ensure quality results, control antigen is present in another area of the membrane strip. The conjugated first antibody binds to the antigen in the control area. Its accumulation also causes a color change and occurs whether or not antigen is present in the patient sample. A positive patient result shows two areas of color change, one for the patient and one for the quality control antigen. If the control antigen area does not change color, the test is considered invalid regardless of color change in the patient area. Feline leukemia test kits (e.g., WITNESS FeLV, Merial UK, Essex, England; Synbiotics, Lyons, France) that use rapid immunomigration are available (Fig. 8-8 and Box 8-6; also see Table 8-1).
Immunodiffusion
In immunodiffusion, patient serum samples (possibly containing antibodies) and the antigen to this antibody (supplied in the test kit) are placed in separate wells in an agar gel plate. Both components diffuse into the agar and form a visible band of precipitation when they combine. If no band forms, no antibody exists in the patient’s serum sample or the patient’s antibody levels are insufficient to cause precipitation in the gel. Diseases that may be detected by immunodiffusion are equine infectious anemia and Johne’s disease (Fig. 8-9 and Box 8-7; also see Table 8-1).
Radioimmunoassay
A competitive form of radioimmunoassay has primarily been used in research and diagnostic laboratories for many years. The test principle is similar to the CELISA technique except that a radioisotope is used in place of the enzyme. The assay typically consists of an antigen labeled with a radioisotope and an antibody. When combined with patient serum that contains the same antigen, both antigens compete for the antibody. With increasing amounts of patient antigen, more labeled antigen is displaced from the antibody. The remaining amount of radioactivity is measured and compared with a standard curve to determine the concentration of antigen in the patient’s serum.
Fluorescent Antibody Testing
Although not commonly performed in veterinary practices, fluorescent testing is available at most veterinary reference laboratories. These test procedures are frequently used to verify a tentative diagnosis made by the veterinarian. Two methods are available—direct and indirect antibody testing—both of which detect the presence of specific antibody in a sample (see Fig. 8-10). In the direct procedure, patient sample is added to a test slide that has been precoated with a fluorescent dye-conjugated antigen. The dye combines with a specific antibody if present in the patient sample. The slide is then examined with a special microscope designed for fluorescent microscopy. For cellular antigens, the cell will appear outlined with fluorescent material.
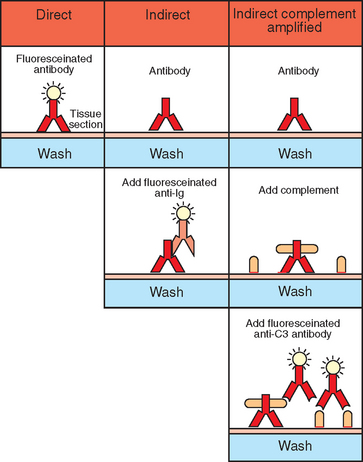
Figure 8-10 Flourescent antibody technique. (Redrawn from Roitt I, Brostoff J, Male D: Immunology, ed. 3, London, 1993, Mosby–Year Book Europe.)
With an indirect fluorescent antibody (IFA) technique, the patient sample is incubated on a slide that contains the specific test antigen. The slide is then washed to remove any unbound antibody. Fluorescent-labeled antiantibody is added to the system. The slide is then microscopically examined. Any fluorescence indicates a positive test result. Fluorescent techniques exist for antigen detection as well.
Coombs Testing
The presence of inappropriate antibodies (i.e., antibodies against the body’s own tissues) are detected with the Coombs test (see Fig. 8-11). The direct Coombs reaction is used to detect antibody that has attacked the body’s own erythrocytes.
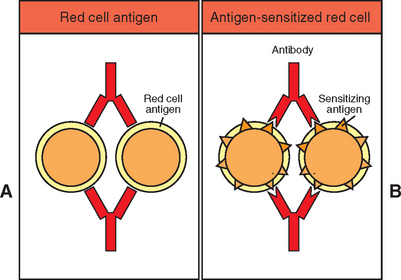
Figure 8-11 Principles of the Coombs reaction. (A) direct Coombs test; (B) indirect Coombs test. (Redrawn from Roitt I, Brostoff J, Male D: Immunology, ed. 3, London, 1993, Mosby–Year Book Europe.)
A positive direct Coombs test provides evidence of immune-mediated hemolytic disease. The procedure involves incubating the suspect sample with antisera, which reacts with the species’ immunoglobulins. If the erythrocytes in the sample are coated with immunoglobulin (self-antibody), the antisera and immunoglobulin on the erythrocytes will react and result in visible agglutination of the erythrocytes.
Indirect Coombs testing detects circulating antibody. A positive indirect Coombs test result indicates the presence of circulating antibodies against the body’s own tissues. To visualize the reaction, patient serum is incubated with erythrocytes from a normal animal of the same species. If antibody is present in the patient serum, it will bind to these erythrocytes just like it would to its own. Subsequent addition of an anti–gamma globulin for the species being tested results in hemagglutination.
Intradermal Tests
Skin tests are used to diagnose various allergies to allergens in the environment. Allergies are mediated by IgE antibody molecules and can be detected by using allergenic extracts of grasses, trees, weed pollens, molds, dust, insects, and other possibly offending antigens. The extracts are injected intradermally, and the injection sites are monitored for allergic reactions. A positive reaction appears as a raised welt, meaning that the animal is allergic to that antigen.
Antibody Titers
Although not routinely performed in the veterinary practice laboratory, antibody titer tests may be needed by the clinician to distinguish between active infection and prior exposure to certain antigens. This is particularly important when no reliable antigen test is available. Titer refers to the greatest dilution at which a patient sample no longer yields a positive result for the presence of a specific antibody.
The test requires making serial dilutions of a sample. Each dilution is then examined for the presence of the antibody. The reciprocal of the greatest dilution that still elicits a positive test result is the titer. A high titer often indicates active infection. Low titers usually indicate previous exposure to the specific antigen.
BLOOD GROUPS AND IMMUNITY
Red cell antigens are structures on the surface of RBCs from one animal that may react with antibodies in the plasma of another animal. The specific surface markers present in an individual animal are genetically determined and are referred to as blood group antigens. The number of blood groups varies among species. Antigen-antibody reactions can occur with blood transfusions as a result of variation in blood group antigens between the recipient and donor. This usually results in clumping or agglutination of RBCs. However, in some species the antigen-antibody reaction is more likely to result in RBC lysis.
Although erythrocytes of common domestic animals possess antigens, naturally occurring antibodies (alloantibodies) present in human beings are less common in animals. However, they do occur in cats, cattle, sheep, and pigs. Once a transfusion has been given to an animal, antibodies against the RBC antigen (immune antibodies) are formed. Breeding females, especially mares, should always be given properly matched blood to avoid sensitization that results in destruction of the foal’s RBCs.
Blood Types
More than a dozen different canine blood groups have been identified. Nomenclature for the blood group systems is designated by the letters DEA (for dog erythrocyte antigen) followed by a number. For DEA systems other than DEA 1, the erythrocytes are designated as positive or negative for that specific antigen. The DEA 1 group has three subgroups designated as DEA 1.1 (A1), DEA 1.2 (A2), and DEA 1.2 (A3). Canine erythrocytes may be positive or negative for each of the DEA 1 subgroups. DEA-3 (B), DEA-4 (C), DEA-5 (D), and DEA-7 (Tr) also designate major blood groups. The only groups considered to be clinically significant are DEA-1.1 and DEA-7. The DEA1.1 subgroup elicits the greatest antigen response and causes the most serious transfusion reactions. Approximately 50% of all dogs are positive for the DEA 1.1 antigen. Transfusion reactions to the other blood groups are not known to cause clinical signs. Because naturally occurring anti-A antibodies do not exist, the first transfusion of A-positive blood into an A-negative recipient may not result in an immediate reaction. However, antibodies can develop and result in a delayed transfusion reaction in as little as a week after the original mismatched transfusion. If a previously immunized A-negative dog receives A-positive blood, severe reactions occur in less than 1 hour.
Cats
One blood group system has been identified in the cat, designated the AB system. Blood groups of cats include A, B, and AB. Few cats have group AB. The majority of cats have group A, which probably explains the low incidence of transfusion reactions in cats. Type B occurs in certain purebred breeds and certain geographic areas. Unlike dogs, cats do possess naturally occurring antibodies to the erythrocyte antigen they are lacking. Type B cats have strong anti-A antibodies, whereas type A cats have weak anti-B antibodies. Transfusing type B cats with type A blood may result in serious transfusion reactions and death. Thus blood for transfusion of purebred cats should be selected by typing or crossmatching.
Neonatal isoerythrolysis has been documented in type A and type AB kittens born of type B queens with naturally occurring anti-A antibodies.
Cattle
Eleven blood groups have been described in cattle, designated A, B, C, F, J, L, M, R, S, T, and Z. Group B is polymorphic, with more than 60 different antigens. Anti-J antibodies are the only common natural antibodies in cattle. J-negative donors may be used to minimize transfusion reactions.
Sheep and Goats
Seven blood group systems have been identified in sheep, designated A, B, C, D, M, R, and X. Similar to cattle, the B system is highly polymorphic. Naturally occurring R antibodies may be present. Neonatal isoerythrolysis may occur in lambs administered bovine colostrum. This is caused by the presence of antibodies to sheep erythrocytes in bovine colostrum. Five major systems have been identified in goats, designated A, B, C, M, and J. Naturally occurring J antibodies may be present.
Horses
More than 30 blood groups have been described in eight major blood group systems in horses, designated A, C, D, K, P, Q, T, and U. Naturally occurring antibodies do exist, but antibodies may be present as a result of vaccinations containing equine tissue or transplacental immunization. Crossmatching should be done before the first transfusion in a horse. Transfusion reactions in horses are commonly fatal.
The mare-foal incompatibility test is a crossmatch procedure that detects the presence of antibodies in mare serum (or colostrum) to foal erythrocytes to confirm or prevent neonatal isoerythrolysis.
Typing and crossmatching of blood
Methods for identification of some canine and feline blood groups are available for use in the veterinary practice (Fig. 8-12). Detailed determination of blood type requires the use of antisera, which consist of antibodies specific for each possible blood type of a given species. Commercial antisera for canine and feline group testing are available. The procedure requires a whole blood sample collected with ethylenediamine tetraacetic acid (EDTA), heparin, or acid-citrate-dextrose (ACD) anticoagulant. The blood is centrifuged at 1000g for 10 minutes. After removal of the plasma and buffy coat, the erythrocytes are washed three times in a saline solution and resuspended. The red cell suspension is distributed among as many tubes as required for the number of blood type antisera being tested. A small amount (usually 0.1 ml) of the antisera in question is added to the appropriately labeled tube. The tubes are incubated for 15 minutes at room temperature and then recentrifuged for 15 seconds at 1000g. Each tube is examined for evidence of hemolysis or agglutination, both macroscopically and microscopically. Weak positive results may require additional testing.
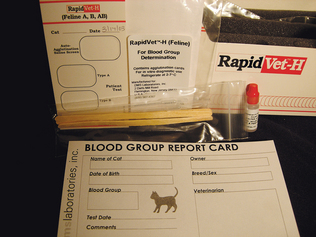
Figure 8-12 Methods for identification of some canine and feline blood groups are available for use in the veterinary practice.
Blood typing of large animals is impractical for routine analysis before transfusion. Literally thousands of different antisera would be required because of the large number of different blood groups in the sheep, cow, and horse.
In the absence of commercial antisera, crossmatching of a blood donor and recipient animal reduces the possibility of a transfusion reaction. The procedure for crossmatching requires both a serum and a whole blood sample and is divided into two parts. Red cell suspensions, collected as for the blood typing procedure, are prepared. The major crossmatch procedure involves the addition of a few drops of serum from the recipient animal to a few drops of washed cells from the donor. The mixture is incubated and then centrifuged. The presence of hemolysis or agglutination, either macroscopically or microscopically, indicates a blood type mismatch. The minor crossmatch procedure is similar except that donor serum and recipient cells are used.
Both procedures should be performed on all animals with unknown blood types that require transfusion. Two controls are used for the test, which consists of running the procedure using donor cells together with donor serum and recipient cells together with recipient serum.
TESTS OF CELL-MEDIATED IMMUNITY
Tests of humoral immunity involve detection of circulating antibodies; evaluation of cell-mediated immunity is much more difficult.
Tuberculin Skin Test
The tuberculin skin test correlates with a specific cell-mediated immune reaction. Animals infected with Mycobacterium tuberculosis, M. bovis, and M. avium bacteria develop characteristic delayed hypersensitivity reactions when exposed to purified derivatives of the organism called tuberculin. In the tuberculin skin test, tuberculin is injected intradermally at a site in the cervical region or in a skin fold at the base of the tail in large animals (see Table 8-1). A delayed local inflammatory reaction is observed if the animal has been exposed to Mycobacteria. The reaction to injection is delayed because a day or more passes before the T lymphocytes migrate to the foreign antigen injected into the dermis.
COLLECTING SAMPLES FOR SEROLOGIC TESTING
Nearly all serologic tests require serum or plasma as the sample. Whole blood should not be sent to the diagnostic laboratory when serum or plasma is specified. The most practical method of collection is the Vacutainer System (Becton Dickinson, Franklin Lakes, NJ), commonly available from many veterinary and medical supply companies. A red-topped vacuum tube is used when serum is required and a lavender-topped tube is used to collect plasma unless heparinized plasma (green-topped tube) is specifically requested.
Reference laboratories have strict requirements concerning specimen type, quality, and handling. If any uncertainty exists, the laboratory should be contacted for specific details. For each test, the requirements should be read carefully and exactly what is requested should be submitted. If a blood sample is to be collected in a syringe, a 5-ml syringe and 20-gauge needle combination should be used because it causes the least hemolysis.
Handling Serologic Samples
When serum is to be submitted, the blood sample is allowed to clot for 20 to 30 minutes at room temperature and then is centrifuged for 10 minutes at a speed no faster than 1500 rpm. If little serum has separated after centrifuging, “rimming” the tube with a wooden applicator stick to loosen the clot may help; however, this also may cause hemolysis. If plasma is desired, the sample may be centrifuged immediately after collection.
After centrifugation, a small pipette is used to aspirate the serum or plasma (upper layer) off the packed erythrocytes. The aspirate is placed into a transfer tube or other sealable test tube and clearly labeled. The serum or plasma may be tested immediately or frozen or refrigerated for later use. See Chapter 3 for more details on collection of serum samples.
Samples for most serologic tests need not be frozen but should be shipped cold, especially during hot weather. The major problem with shipping tubes is breakage. The tubes must be packed firmly in place with packing material so they do not move around when the package is jarred. Each sample must be clearly and correctly labeled and the pertinent paperwork enclosed to facilitate proper reporting of the results from the laboratory.
MOLECULAR DIAGNOSTICS
Leptospira spp., slow-growing bacteria on a culture plate, are one of many bacteria that can now be identified by using molecular diagnostic testing. Its DNA molecule, which contains its genetic information, is the molecule of interest in the test. Molecular diagnostics is based on analyzing DNA or RNA. Although too sophisticated for use in veterinary practices, veterinarians can send samples out to be tested in a short amount of time. Many of the state veterinary diagnostic laboratories now offer several molecular tests. The obvious use for the veterinarian is to identify the presence of pathogens such as viruses, fungi, or bacteria, but there are many other uses for this technology (Table 8-2 and 8-3).
TABLE 8-2
Molecular Diagnostic Tests for Veterinary Pathogens
| Organism | Suggested Samples |
| Bacillus anthracis | Blood |
| BVD1, BVD2 | Lymph nodes, spleen, serum |
| Chlamydia spp. | Placenta, liver |
| Clostridium perfringens | Isolated colony from bacterial culture |
| Escherichia coli virulence typing panel | Isolated colony from bacterial culture |
| Leptospira spp. | Urine, liver, kidney |
| Mycobacterium paratuberculosis | Intestinal mucosa, mesenteric lymph nodes |
| PPRS virus | Serum, spleen, lung |
| Salmonella spp. | Intestinal mucosa, feces, other tissues |
| West Nile virus | Kidney, heart, brain, liver, spleen |
TABLE 8-3
| Animal | Test | Test Sample |
| Avian | Bird sexing | Blood or freshly plucked feathers |
| Canine | DNA banking and profiling | Buccal swab (for animal identification) |
| Canine | Inherited disease screening | Buccal swab |
| Canine | Parentage verification | Buccal swab |
| Equine | DNA banking (for animal identification) | 15-20 hairs pulled from mane, including hair root bulbs |
| Equine | Hyperkalemic periodic paralysis screening | 15-20 hairs pulled from mane, including hair root bulbs |
| Feline | DNA banking and profiling | Buccal swab (for animal identification) |
| Feline | Parentage verification | Buccal swab |
| Feline | Polycystic kidney disease | Buccal swab |
The branches of medicine and science that use these types of DNA tests include microbiology, genetics, immunology, pharmacology, forensics, biology, food science, agriculture, archaeology, and ecology. DNA tests are available to classify cancers, detect genetic defects, verify animal pedigrees, and determine bacterial contaminants in food science applications, to name a few uses.
The advantages of these kinds of tests are increased sensitivity (degree to which a test can detect and consistently measure small amounts of sample) and increased specificity (ability of a test to detect and measure only the desired sample and not cross-react with other substances). The amount of specimen needed for the test can be exceedingly small, the tests are safe, and many factors that influence other procedures—such as age and condition of sample, fastidious growth requirements, and viability of the organism—are not as crucial with molecular diagnostic tests. The newer techniques also have faster turnaround times. Whereas traditional identification of a bacterium may take 2 to 3 days or more, molecular diagnostic testing can be accomplished in a matter of hours depending upon the test.
Disadvantages have been contamination leading to false-positive results, a high level of technical expertise needed to run the tests, the need for more than one room in which to perform the tests, and high costs. Many of these problems are being solved, and commercial kits and automated instruments are making these tests available to clinical diagnostic laboratories.
Many varieties of molecular diagnostic tests are available, but perhaps the most familiar, if not most widely used, is the polymerase chain reaction (PCR). This test detects the DNA segment of interest in the specimen submitted and amplifies its amount (Box 8-8).
Reverse Transcriptase Polymerase Chain Reaction
Sometimes RNA is the nucleic acid used for the molecular test, such as when testing for RNA viruses. The process used is called reverse transcriptase PCR (RT-PCR). It is similar to PCR, but the single-stranded RNA must first be converted to double-stranded DNA before the PCR process can continue.
Real-Time Polymerase Chain Reaction
Another significant test is real-time PCR. Compared with PCR, this method decreases the risk of contamination, is more easily automated, and is generally faster and easier to run. A fluorescent probe is added to the sample mix; it attaches to the DNA segments and, as the quantity of segments is amplified, fluorescence increases. At a set amount of fluorescence, the sample is considered positive.
Polymerase Chain Reaction
Polymerase chain reaction is called an amplification assay because a small amount of a DNA segment detected in the sample is amplified to run the test better and determine the results. That is, a PCR test produces many copies of a small, select region of the DNA molecule. Before performing the test, the nucleotide sequence of this section of DNA must be known so that the proper reagents are used. The region of the DNA that will identify the virus or bacterium is predetermined.
The amplification process consists of three basic steps: denaturation, annealing, and extension (Fig. 8-13). After amplification, the DNA segments are separated on an electrophoretic gel for identification. The sample mixture contains the specimen with the original DNA in question (if present), primers, nucleotides, and Taq DNA polymerase (the enzyme that can read the DNA code and assemble the complimentary nucleotide bases).
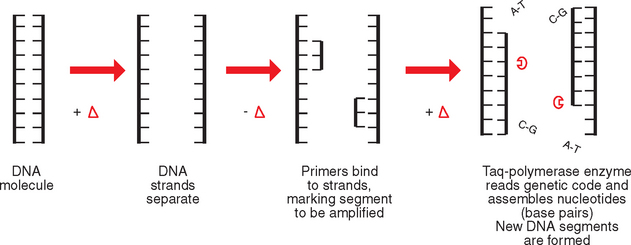
Figure 8-13 PCR showing denaturation of DNA into two strands, annealing of primers to single strands, and extension of DNA molecule with nucleotides by enzymatic action of Taq polymerase.
Denaturation
The sample is heated to break apart the double-stranded DNA molecule into two separate strands. Each strand serves as a template on which new nucleotides will attach.
Annealing
The temperature is lowered to cause the primers to bind (anneal) to the separated strands. Primers mark the beginning and the end of the section of DNA to be copied. This will happen only if DNA is present in the sample that is complementary to the primers.
Extension
Once more the temperature is raised and Taq DNA polymerase causes new complementary DNA segments to be produced (extended). Portions of two DNA molecules have been obtained, each with two strands. They are not the complete DNA molecule but do contain the desired segment.
This process is repeated 25 to 30 times in an automated thermal cycler (Fig. 8-14). The timing, temperature, and number of cycles are regulated by the instrument. The amount of DNA segments produced is far greater than the original amount of DNA in the specimen. This is why PCR is useful for detecting minute quantities of the unknown in a mixed specimen.

Figure 8-14 Thermal cycler for PCR. Instrument automatically controls the temperature and timing. (Courtesy Bio-Rad Laboratories, Inc., Hercules, CA.)
Finally, to see if the DNA segment was present in the specimen, agarose gel electrophoresis is used. The DNA segments are negatively charged particles and will move along the gel toward the positive electrode when a current is applied. The segments separate according to size, appearing as separate bands on the gel. Controls are run at the same time as the test samples. By knowing the identity of the control bands, the test bands can be compared and identified.
Interpretation of PCR tests must be done carefully. A microbe may be present in the sample, but may not cause disease. As with any laboratory test, the results must be evaluated along with all the information from the clinical case.
Recommended Reading
Birchard SJ, Sherding RG, eds. Saunders manual of small animal practice, ed 3, St Louis: Elsevier, 2006.
Sirois, M. Clinical chemistry and serology. In: Principles and practice of veterinary technology. St Louis: Mosby; 2004.
Tizard, IR, Schubot, RM. Veterinary immunology: an introduction. St Louis: Elsevier, 2004.