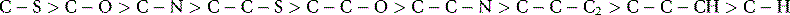Chapter 2 Hydrocarbons
alkanes, alkenes, aromatic and alkylhalides
Lipophilicity
The activity of a drug within the body depends on its pharmacokinetics and its pharmacodynamics. Pharmacodynamics describes how the drug affects the body, i.e. how quickly it diffuses into tissues and how strongly it binds to proteins and receptors. Pharmacokinetics describes how a drug is distributed by the body which tissues it accumulates in and how quickly it is eliminated from those tissues. Pharmacokinetics is a relatively slow process; when a drug reaches its site of action the process of binding to its receptor site is relatively rapid. One of the most important physicochemical properties governing drug distribution within the body is its lipophilicity/partition coefficient.
n-Alkanes
n-Alkanes are the simplest organic molecules. They are devoid of polarity and their physical properties are governed by van der Waals interactions between the molecules, which increase with the number of CH2 atoms in the structure of the molecule (Table 2.1). Thus the boiling points of the hydrocarbons increase with the addition of each CH2 group. The increments in b.p. for the addition of a CH2 get less as the molecular weights of the hydrocarbons get larger.
| Hydrocarbon | Formula | b.p °C |
|---|---|---|
| Methane | CH4 | −162 |
| Ethane | C2H6 | −87 |
| Propane | C3H8 | −42 |
| Octane | C8H18 | 126 |
| Nonane | C9H20 | 150 |
| Decane | C10H22 | 173 |
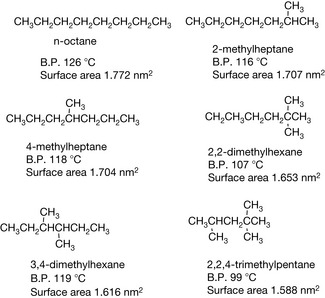
Since boiling point gives an indication of the strength of van der Waals/lipophilic interactions from Table 2.1 it is evident that, when incorporated in a drug, the larger the alkyl group the more it is going to promote interactions with lipophilic structures in the body such as membranes and hydrophobic pockets within receptor proteins. If the alkene is branched, the boiling point is generally less than that of the straight-chain alkane since the molecules present a lower surface area to each other and hence van der Waals interactions are lower. This is exemplified by a series of octanes shown in Figure 2.1, all of which have a lower boiling point than octane. Thus, branching in an alkane substituent in a drug reduces van der Waals interactions as the shape of a molecule gets closer to that of a sphere, which has the lowest surface area to volume of any shape.
Alkanes are biologically fairly inert and are not readily metabolised by the body. For example, liquid paraffin, which is a mixture of long-chain alkanes and is used as a laxative, is not absorbed by the bowel and is excreted unchanged. Similarly, soft paraffins which are applied to the skin in greasy ointments are not appreciably absorbed by the skin. However, volatile hydrocarbons such as methane, which is present in natural gas, are readily absorbed into the bloodstream via the lungs. They thus become concentrated in lipophilic membranes and act as general anaesthetics, producing unconsciousness. Like all anaesthetics, the effects are reversible if an unconscious individual having accidently breathed in methane is found in time. Cyclopropane, a simple alkane, was actually used as a volatile anaesthetic. Although alkanes, particularly the higher boiling point hydrocarbons, are not readily absorbed by the body, the small amounts that are absorbed by the body are hydroxylated via the action of cytochrome P450 (cyp450) in the liver.
Stereochemical considerations
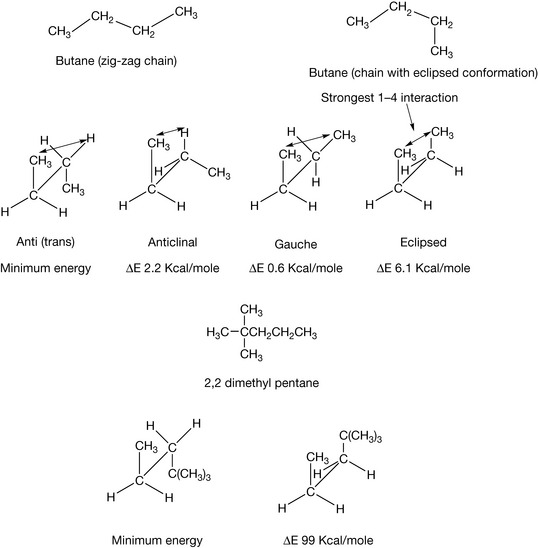
As well as the surface area of hydrocarbons, another consideration is their stereochemistry, that is the shape that the molecules make in three dimensions. This is determined by the most favourable orientation of groups with respect to each other; in unfavourable orientations groups are brought too close and push against each other. One of the simplest examples for conformational analysis is butane. All bonds within a molecule can be rotated to give a higher or a lower energy arrangement of the atoms in space. The lowest energy state for butane is with the chain in the zigzag pattern where the central bond in the chain is rotated to give an anti or trans conformation (Fig. 2.2). Rotation about the central bond can give any number of conformational states with a range of energies between the lowest energy anti state and the highest energy eclipsed state which is at an energy level 6.1 Kcal/mole higher. At room temperature the thermal energy in the environment is great enough to allow the molecule to rotate freely about its central bond, passing through the higher energy state. The reason for the increase in energy in the eclipsed conformation can be viewed as being mainly due to steric repulsion between the two methyl groups. However, the total conformational energy includes tension within the bonds of the molecules which is either caused by them being stretched or twisted (torsional) and deformation of the tetrahedral angle of the bonds between the atoms of the molecule. When the groups involved in the eclipsed conformation are very large, free rotation about the bond under consideration is not possible. For example, the energy of the eclipsed conformation of 2,2 dimethyl pentane is 99 Kcal above the energy of the minimal energy trans configuration (see Fig. 2.2). As temperature increases even conformationally restricted hydrocarbon chains are freer to rotate and adopt a variety of conformations. This phenomenon is important in the behaviour of membrane lipids where the formation of kinks in the lipid chain, which at low temperatures exists in the completely zigzag form, causes the membrane to get thinner and spread out at the same time as temperature rises.
Cycloalkanes
The smallest cycloalkane is cyclopropane which has a very strained three-membered ring and the carbon bonds within the molecule are bent in order to compensate for the fact that they cannot adopt their natural tetrahedral structure. As the number of carbon atoms in the ring increases the rings become less strained. There is still considerable strain within cyclobutane but in cyclopentane the strain is minimal and cyclohexane is an unstrained ring. Apart from having been used as a volatile anaesthetic, the cyclopropyl ring has been used as an alkyl group in some drugs, e.g. ciprofloxacin (Fig 2.3), buprenorphine and prazepam.
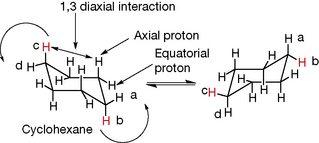
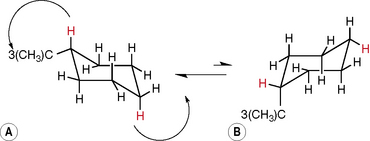
Cyclohexane is free from strain since its ring is able to pucker, thus allowing the carbons to adopt their natural tetrahedral conformation (Fig. 2.4). The cyclohexane ring contains two types of protons, axial protons and equatorial protons, and the ring can flip between two forms so that the axial protons become equatorial and vice versa, e.g. protons a and b and c and d in the figure. The two forms of cyclohexane are indistinguishable but, since the axial positions are more sterically crowded, a large substituent on the ring will lock it into one configuration so that, for instance, tertiary butyl cyclohexane will exist almost completely in form A (Fig. 2.5).
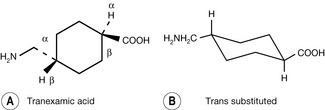
Stereochemistry is very important in determining the biological activity of drugs and naturally occurring biologically active compounds. Among commonly used drug molecules the steroids are most prominent in having a rigidly defined polycyclic stereochemistry. A simple example of a drug with stereochemistry defined by bulky substituents in its ring is the haemostatic drug tranexamic acid. It can be drawn either viewed as a hexagon with substituents attached to it (Fig. 2.6A) or in a way indicating more directly that the substituents attached to it are in equatorial positions (Fig. 2.6B). When it is drawn in form A the wedge-shaped bonds indicate substituents which are closer to the observer relative to the substituents which are further away, which are indicated by dashed bonds. The substituents with the wedge-shaped bonds are said to be β and those with the dashed bonds are said to be α. When the structure is drawn in form B it is possible to see that the two substituents are equatorial, which is the least sterically strained form for the molecule, and that they are on opposite sides of the ring. Substituents on opposite sides of the ring are said to be trans to each other. Where the substituents are on the same side of the ring they are said to be cis.
For instance, if the methylamine group were substituted so that it was cis relative to the carboxylic acid group, a stereoisomer of tanexamic acid would be produced (Fig. 2.7). This molecule has a completely different shape from tanexamic acid and hence completely different biological properties.
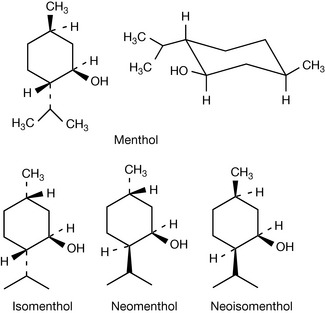
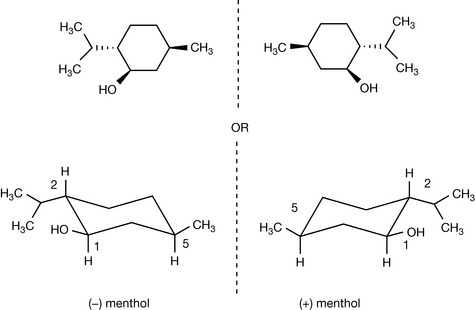
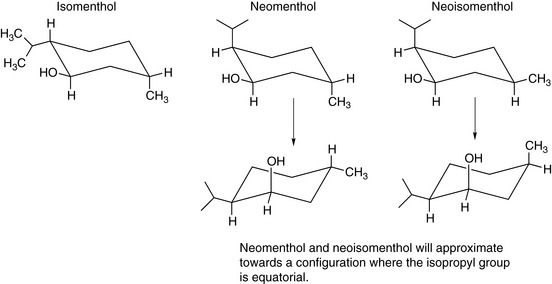
As the number of substituents on the ring increases, the number of possible isomers increases. Menthol, a commonly used pharmaceutical excipient, has three isomers. Menthol itself has all substituents in equatorial positions. Assignment of the stereochemistry of substitution is made relative to the hydroxyl group that, by convention, is given highest priority since the oxygen atom is the heaviest atom attached to the structure. Thus the methyl substituent is cis relative to the hydroxyl and the isopropyl group is trans. If all the possible permutations of this are taken into account three additional chemical compounds which are isomers are apparent. These are called isomenthol, neomenthol and neoisomenthol (Fig. 2.8). They have different properties from menthol, e.g. different melting points and boiling points, and it is possible to simply verify that they have different biological properties from menthol since, although all the isomers smell of mint, their exact smells are rather different.
![]() Self Test 2.2
Self Test 2.2
Draw the structures of the isomers of menthol, as shown in Figure 2.8, so that it is possible to see whether the substituents are axial or equatorial. Which of the structures which you have drawn do you think might rearrange to give less sterically strained forms?
Optical isomerism
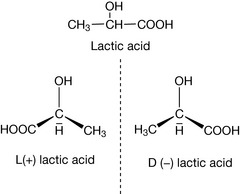
In considering stereoisomerism above, we have ignored an additional form of isomerism which is of great importance in biologically active molecules. Optical isomerism is a specific form of stereoisomerism where the isomers do not differ in their physical properties such as melting point, boiling point or partition coefficient. There is only one physically measurable difference between them, which is that they tilt plane polarised light in opposite directions. Lactic acid, a by-product of glucose metabolism, can exist in two forms or enantiomers which are mirror images of each other (Fig. 2.9). This occurs when the groups attached to a central carbon atom, the chiral centre, are all different. The mirror images, like right and left hands, cannot be superimposed on each other.
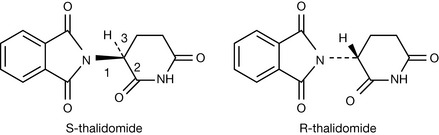
Although enantiomers always have identical physical properties their biological properties can be quite different. The most notable example in pharmacy is thalidomide (Fig. 2.10), which was given as a sedative in the 1960s and was responsible for causing birth defects. The drug has a single chiral centre and is chemically synthesised as a 1:1 mixture of the two enantiomers (racemate). Because of their identical physical properties, enantiomers are difficult to separate and the drug was given as the racemate (1:1 mixture of the two enantiomers). It was presumed that both isomers had identical biological properties; however, subsequently it was found that the S-isomer was teratogenic. However, administration of the S-isomer alone would not necessarily have improved matters since the two isomers interconvert in vivo. Thalidomide is still used as the racemic drug but for treating diseases that produce immunosuppression such as leprosy, in the absence of pregnancy.
The terms (+) and (−), L and D and R and S have been introduced without a complete explanation. The terms (+) and (−) simply refer to the direction in which a pure enantiomer rotates plane polarised light, i.e. to the right or to the left. In older literature (+) and (−) are often referred to as d and l where d stands for dextrorotatory and l for laevorotatory. This type of nomenclature persists where levodopa (LDOPA) is the (−) amino acid used to treat Parkinson’s disease. Similarly, the terms D and L are the same as R and S that are the modern equivalents. R and S are a convention for describing the absolute configuration of a chiral centre. To assign the absolute configuration, the group of lowest priority, often a hydrogen atom, is placed behind the plane of the paper. Assignment of the configuration is then made on the basis of precedence rules based on the atomic weights of the atoms attached to the chiral carbon atom. For example, if C is the chiral carbon atom then the bonds attached would have the following precedence:
It is difficult to be comprehensive in the precedence order but this order will apply to the majority of drug molecules. If we examine S and R thalidomide we find for S thalidomide (as drawn) that the group of lowest priority is placed behind the plane of the paper and the precedence order for the groups attached to the chiral centre are N > C–O > C–C giving an anticlockwise, hence S, progression. Ignoring the fact that R thalidomide has the hydrogen above the plane of the paper we can make the same assignment as for S thalidomide but reverse it, giving us an R configuration. Alternatively, the figure could be redrawn with the hydrogen behind the plane of the paper. Some molecules have multiple chiral centres; an example we used earlier is menthol (Fig. 2.11). The fact that menthol has a mirror image was overlooked and thus menthol structure has a total of eight isomers (23) not four; however, these isomers are comprised of four enantiomeric pairs. Thus menthol, isomenthol, neomenthol and neoisomenthol all have mirror image isomers which have identical physical properties but all differ in properties from the non-mirror image isomers. For example (−) neomenthol has the same physical properties as (+) neomenthol but different physical properties from both (+) and (−) isomenthol, which in turn have the same physical properties. The enantiomeric pairs all have different biological properties and thus (−) menthol has a slightly different smell from (+) menthol. The classic smell difference in this respect is between S (+) and R (−) carvone, the S (+) isomer smells of caraway and the R (−) isomer smells of dill. The (−) menthol has the following chiral centres: 1 (R), 2 (S), 5 (R) and it follows that (+) menthol has the configuration 1 (S), 2 (R), 5 (S). Although it is possible to draw the absolute configuration of a molecule on paper easily, it is only possible to relate this configuration to the direction in which a molecule will rotate plane polarised light by experiment. The principal method for determining the absolute configuration of a chiral centre or chiral centres within a molecule is single-crystal X-ray crystallography, which is a relatively complex technique. In contrast, polarimetry, which is the method used to determine relative configuration (whether a molecule is [+] or [−]) is an easy technique to use.
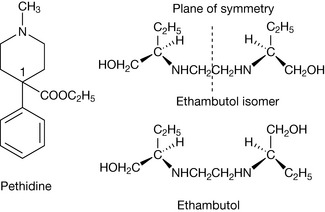
Care has to be taken in recognising a chiral centre; for example, pethidine (Fig. 2.12) may look as if it has a chiral centre at 1 but, in fact, it is symmetrically substituted by the heterocyclic ring. The isomer of ethambutol (see Fig. 2.11) is not a chiral molecule but is called a meso compound since it has a plane of symmetry running through its centre which means its mirror image is superimposable. Ethambutol itself, however, is a chiral molecule since its mirror image is not superimposable.
Many drug molecules contain several chiral centres and these types of drug molecules are generally derived from natural sources where they have been stereospecifically synthesised by plants or microorganisms. Examples include: penicillins, steroids, alkaloids, tetracyclines, aminoglycosides and amino acids. Where a drug has been wholly chemically synthesised it may often contain no chiral centres or have only a single chiral centre and be produced as a racemate.
![]() Self Test 2.3
Self Test 2.3
Assign the absolute configurations of the numbered centres in the following molecules and indicate how many isomers are possible for each structure.
Many drugs have a single chiral centre but are administered as a racemate. Table 2.2 shows some examples of drugs where the enantiomers of the drug have different biological properties.
Table 2.2 Enantiomeric drugs and their biological properties
| Drug | Properties |
|---|---|
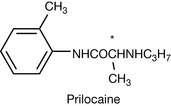 |
Both isomers are local anaesthetics but the R isomer is enzymatically hydrolysed much more quickly than the S isomer. The hydrolysis product, toluidine, affects the oxygen-carrying capacity of red blood cells. |
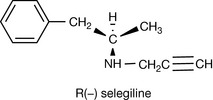 |
Selegeline, a monoamine oxidase inhibitor, is dealkylated by metabolism to amfetamine. The R isomer forms R amfetamine which has little effect on the CNS whereas the S isomer forms S amfetamine which has strong CNS effects. The drug is given as the pure R isomer. |
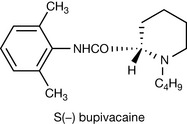 |
Levobupivacaine (S (−)) has been recently introduced as an anaesthetic since there is some evidence that it is less cardiotoxic than racemic mixtures of bupivacaine. |
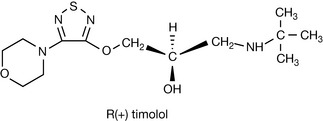 |
Timolol is used to treat both hypertension and glaucoma in the form of its S (−) isomer. However, there would be a reduction in side effects if the R(+) isomer were used to treat glaucoma since it is effective in treating glaucoma but has very weak β-adrenergic blocking activity. |
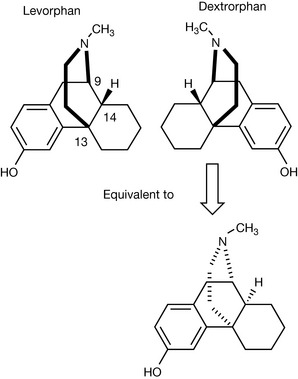 |
Most commonly, drugs which are administered as racemates have only one chiral centre. Levorphan and dextrorphan (based on the morphine structure) are unusual in that they have the opposite configurations at three chiral centres. Levorphan ([−] isomer 9 R, 13 R 14 R) is a narcotic analgesic which is potentially addictive whereas dextrorphan ([+] isomer 9S, 13 S, 14S) is used as an antitussive drug in the form of its O-methylether dextromethorphan. |
There are a number of drugs such as dextrorophan and levorphan which are prefixed by dextro- or levo- to indicate that they rotate plane polarised light either in the (+) or the (−) direction, but remember that the prefixes L and D or levo- and dextro- only refer to the direction in which the compound rotates plane polarised light. It does not tell us whether the compound is R or S or, if it has more than one chiral centre, what the configurations of these centres are.
![]() Self Test 2.4
Self Test 2.4
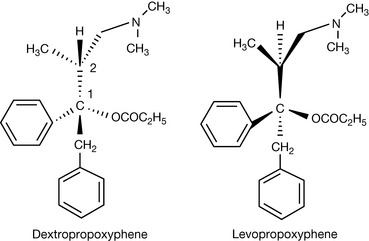
Assign the absolute configurations of the chiral centres of dextropropoxyphene and levopropoxyphene. These drugs are morphine analogues. Can you guess which is the antitussive and which is the narcotic analgesic? List all (+) (dextro-) and (−) (levo-) drugs which you can find in the BNF?
Double bonds
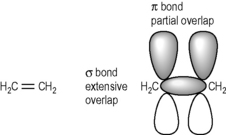
Stereochemistry is most complex where carbon substitution is tetrahedral. Where double bonds are present in the molecule it is flattened and thus takes up less space. Ethylene is the simplest compound which contains a double bond. The σ bond is a strong bond produced by extensive overlap of orbitals whereas the π bond is produced by partial overlap of orbitals (Fig. 2.13) and hence is a weaker bond which is rich in electron density, which is available for sharing with a suitable reactive chemical species. Thus, in biological systems, reactive double bonds can prove problematical since they can form unwanted covalent bonds with biological structures.
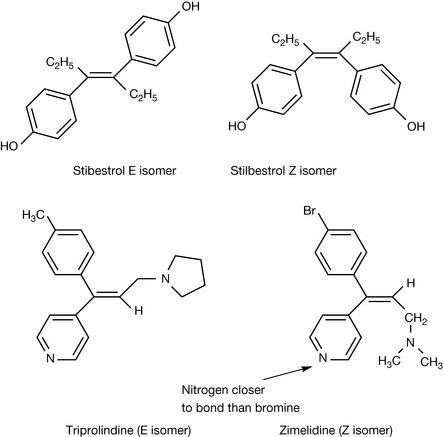
The presence of a double bond, because of the lack of free rotation about the bond, within a molecule can give rise to a form of isomerism known as geometrical isomerism. There are not many drugs which exhibit geometrical isomerism since, unlike enantiomers, geometrical isomers can be more easily separated. One example is the E (trans) and Z (cis) isomers of the oestrogen analogue diethyl stilbestrol (Fig. 2.14). The E isomer is the therapeutic agent used and it is a much more potent oestrogen than the Z isomer. The letter E indicates that the highest priority substituents are on opposite ends of the double bond and are on opposite sides of the bond, whereas Z indicates that the highest priority substituents are on opposite ends of the bond and are on the same side of the bond. The priority rules are the same as those used in assigning absolute configurations. Thus the antihistamine triprolidine is used in the form of its E isomer since, working back from the point where they are attached to the bond, the two nitrogen-containing substituents have the highest priority. Similarly, the antidepressant zimelidine has the Z configuration.
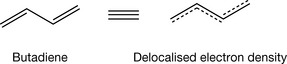
When two or more double bonds are separated from each other by single bonds, a conjugated electronic system is produced where the electrons in the double bonds are delocalised onto all the atoms in the conjugated system. The simplest conjugated systems can be seen in butadiene (Fig. 2.15).
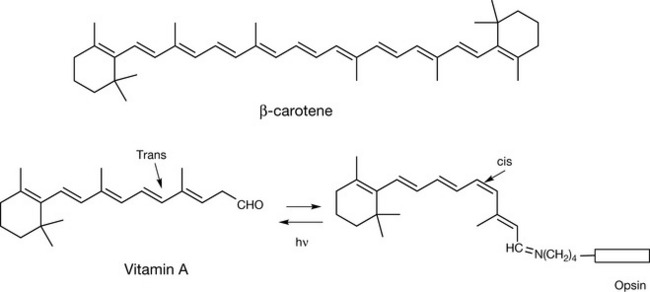
As a system of conjugated double bonds gets longer, the electrons become more loosely held within a molecule, producing a compound that can be excited by visible light and thus appears coloured. This is the case with beta-carotene (Fig. 2.16,) which occurs as an orange pigment in many plants, e.g. carrots, and may assist in the harvesting of light by plants. Beta-carotene can be converted by the human body into vitamin A, which is yellow in colour. Vitamin A is involved in the visual process as shown in Figure 2.16 where it binds to the visual protein opsin in a form where one of its double bonds is Z (cis). Absorbance of light energy causes the bond to convert from Z to E (trans), thus altering the 3-D structure of the protein to which it is bound and triggering off events such as ion channel opening and formation of secondary messengers. The interconversion between cis and trans forms causes mechanical energy to be generated from light energy.
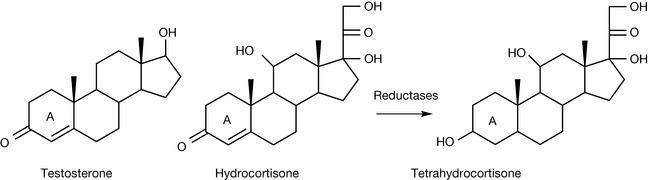
There are not many drugs which contain extended double-bond systems. Steroids such as testosterone and hydrocortisone (Fig. 2.17) have an enone double-bond system in their A ring. This is crucial for their biological activity and enzymatic reduction of the double bonds results in loss of the activity of the steroid. Tetrahydrocortisone is inactive and is the main urinary metabolite of hydrocortisone.
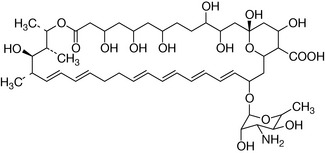
Nystatin (Fig. 2.18), a macrocyclic polyene antibiotic, is an example of a drug with an extended system of double bonds. The double bonds interact with membrane lipids, disrupting the cell membrane and thus killing the pathogen. Amphotericin B, another antifungal compound, works by a similar mechanism.
Membrane lipids
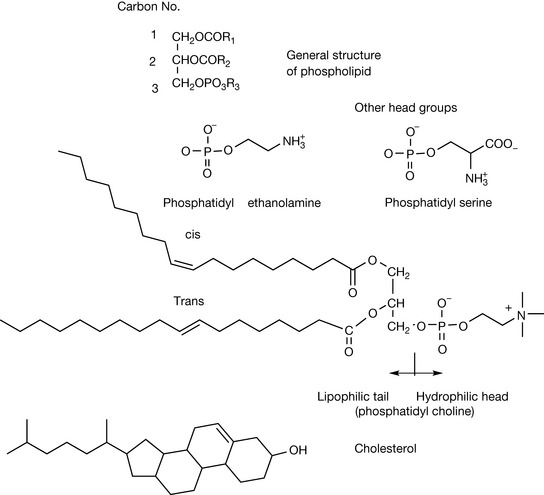
Double bonds are found in the lipids that compose cell membranes. Membranes are composed of complex mixtures of lipids and the complexity of the mixture enables membranes to produce their responsive properties. Membrane lipids are all amphiphilic and have a polar head group in combination with a lipophilic tail which causes in an aqueous medium the typical lipid double layer. Figure 2.19 shows some typical membrane lipids. The phospholipids are triesters of glycerol where R1 may be a saturated or unsaturated fatty acid, R2 is usually an unsaturated fatty acid and R3 is one of a number of polar head groups. The lipophilic fatty acid tails of the molecules are composed of saturated and unsaturated hydrocarbon chains between 14 and 24 carbons long. Unsaturated lipids may contain cis or trans double bonds. Unsaturated lipids tend to orientate themselves as shown for the trans monounsaturated fatty acid shown in Figure 2.19.
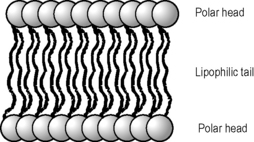
Figure 2.20 shows a lipid bilayer. Such bilayers are regarded as being in a lyotropic liquid crystal form where they are highly ordered as in a crystal but the ordering of the molecules is very sensitive to temperature and other environmental factors. The bilayer is structured so that the polar head groups of the lipids are in contact with the surrounding water while the lipophilic chains are hidden inside the layer.
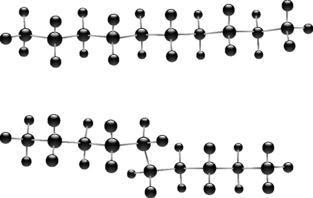
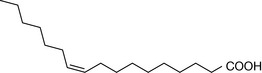
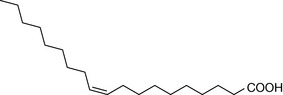
The composition of the fatty acids making up the membranes of cells varies from organ to organ and from species to species. The composition of the fatty acids making up the phosphatidylcholine lipid fraction from membranes of human red blood cells is shown in Table 2.3. As is the case with all membranes, the saturated fatty acid component predominates (in this case palmitic and stearic acids). The saturated fatty acids pack together closely in the membrane, producing a rigid membrane. The introduction of unsaturated fatty acids into the membrane (particularly cis fatty acids with their awkward shapes) prevents the membrane fatty acids from packing closely, thus producing a more fluid membrane. The fatty acid chains become increasingly bent as the number of cis double bonds in the hydrocarbon chain increases. The saturated fatty acids themselves can also adopt less linear configurations, with kinks in their chains like the cis unsaturated fatty acids. In the liquid crystalline state the saturated fatty acids in lipids typically adopt a gauche configuration (Fig. 2.21) where the chain bends and thus shortens so that the membrane gets thinner but spreads out over a larger area.
Table 2.3 The most abundant fatty acids within the phospholipids of red blood cells
| Fatty acid | molar % | |
|---|---|---|
| Palmitic acid | 33 |
 |
| Palmitoleic acid | 1 |
 |
| Stearic acid | 11.7 |
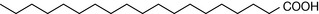 |
| Oleic acid | 20.6 |
 |
| Linoleic acid | 18.2 |
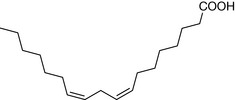 |
| Eicosatetraenoic acid | 5 |
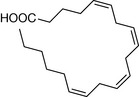 |
| Docosapentenoic acid | 5.4 |
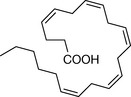 |
Lipid oxidation
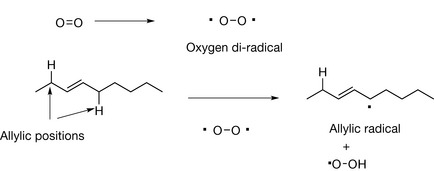
The presence of a double bond within a hydrocarbon chain increases its susceptibility to oxidation. The allylic position is particularly susceptible to attack by oxygen which can behave as a di-radical having two reactive unpaired electrons. The oxygen di-radical abstracts a hydrogen atom from an allylic position to form an allylic radical (Fig. 2.22), this process is favoured by the presence of the double bond which stabilises the allylic radical.
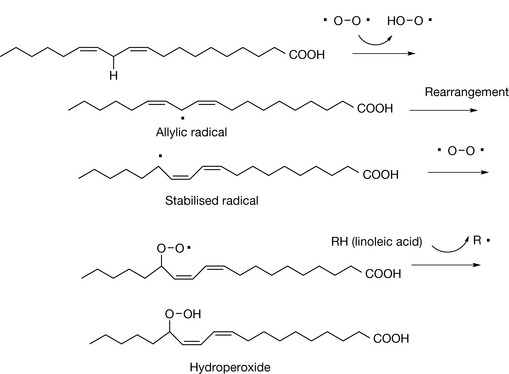
These types of radical are formed even more rapidly where more than one double bond is involved, e.g. in linoleic acid (Fig. 2.23).
The formation of a hydroperoxide generates another allylic radical so that a chain reaction occurs resulting in formation of further molecules of hydroperoxide. These hydroperoxides are unstable and decompose to give a mixture of degradation products which include a range of aldehydes, and hydroxyaldehydes such as 4-hydroxynonenal (4-HNE) which is a degradation product of arachidonic acid. Molecules such as 4-HNE may have a role in promoting apoptosis.
Prostaglandins
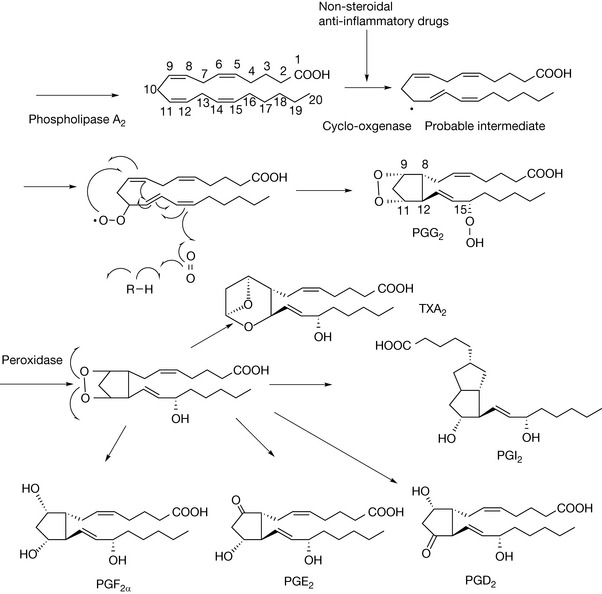
The susceptibility of unsaturated membrane lipids to oxidation by atmospheric oxygen is to an extent mirrored by the ease with which they can be oxidised via enzymatic action. Eicosatetraenoic acid (arachidonic acid, AA) provides the precursor for the prostanoid class of locally acting hormones (Fig. 2.24). Prostanoids are important mediators of inflammation and a wide range of other processes. The first step in the enzymatic conversion of AA to prostanoids involves enzymatic cleavage of the ester bond between AA and the glycerol backbone of a phospholipid into which it is incorporated and this reaction is carried out by phospholipase A2. The next step is, probably, essentially the same as the allylic oxidation of polyunsaturated fatty acids carried out by atmospheric oxygen and this is followed by structural rearrangement within the molecule, giving rise to PGG2. The main difference from a chemical process, of course, is that the stereochemistry of the reaction is enzymatically controlled and thus chiral centres are generated at positions 8 (R), 9 (R), 11 (S), 12 (R) and 15 (S). This would not occur with a purely chemical process. Nonetheless, lipid peroxidation may occur in cell membranes via reaction with reactive oxygen species forming prostaglandin-like structures and may be a component in autoimmune diseases. The oxygen bridge in PGG2 can be simply modified to give rise to a range of prostanoids, the most important of which are: PGE2, PGD2, PGF2α, PGI2 and TXA2. AA also gives rise to another series of mediators called the leukotrienes which are also involved in inflammatory and immunological processes. Leukotrienes are also formed by peroxidation of the AA structure but lack the five-membered ring which gives rise to the hairpin structure of the prostaglandins.
Molecular shape and structure activity relationships
Drug action depends on the drug interacting effectively with its site of action which may be a receptor protein, an enzyme or DNA. The effectiveness of its interaction depends on how well the shape of the drug matches the shape of the target site. In the case of a receptor binding site there is usually a natural ligand which fits it and drugs are designed either to block the binding of the drug to the receptor without triggering of the receptor activity (antagonists) or to bind to the receptor more strongly than the natural ligand stimulating the same actions as the drug (agonists).
Lipophilic groups in proteins
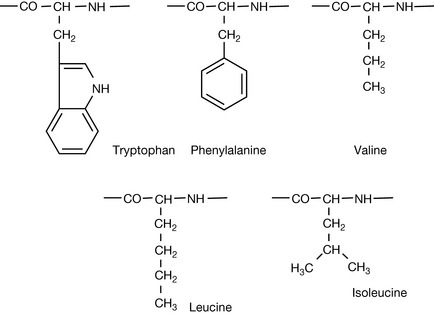
The binding of a drug to a protein depends on the intermolecular forces described in Chapter 1. Thus electrostatic, dipole–dipole, hydrogen bonding, π–π interaction and van der Waals interactions, and even covalent bonding all play a part. The strength of van der Waals interaction depends on the availability of lipophilic groups within the protein. The amino acids with high lipophilicity, and thus potential for van der Waals interaction, are shown in Figure 2.25.
Proteins with a high content of these amino acids are lipophilic; thus ApoE, a protein which is responsible for carrying lipids around the body, contains about 12.6% leucine residues where the average abundance of an amino acid is closer to 5%.
The benzene ring
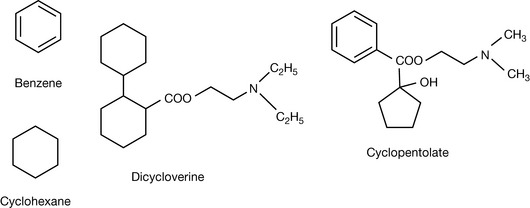
One or more benzene rings are found in a majority of drug molecules. Most of the time, it can be considered that it just contributes to the lipophilicity of a drug, although it does have chemical properties which are different from saturated hydrocarbons. The surface area of benzene is 1.01 nm2 compared with 1.20 nm2 for cyclohexane. Although the chemistry of cyclohexane and benzene is quite different, in most cases they can be regarded as being similar in biological terms so that dicycloverine and cyclopentolate, for example, are both anticholinergic drugs with similar structures (Fig. 2.26). The presence of the benzene ring in cyclopentolate does not dramatically alter its properties compared with dicycloverine. The benzene ring is often a convenient starting point for the synthesis of a drug molecule since it is chemically reactive and in addition it is found in many products which are either drugs or have been developed into drugs by chemical modification. Extensive discussion of the chemistry of the benzene ring can be found in organic chemistry textbooks.
Alkylhalides
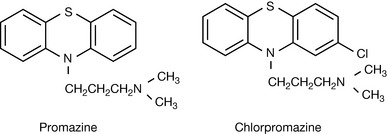
Halogen atoms have been used extensively modify the activity of drug molecules. Often, the increase in the potency resulting from addition of a halogen atom to a drug molecule can be attributed to increased partition coefficient. Thus chlorpromazine (log P 3.4 at pH 7.4) is more potent than promazine (log P 2.5 at pH 7.4) as a tranquilliser (Fig. 2.27). The presence of the chlorine atom in its structure increases its lipophilicity and this means that it is more effective in crossing the blood–brain barrier. In the case of many drugs there is an optimum lipophilicity and, despite there being many analogues of chlorpromazine where the chlorine is replaced by another group, none of these offer significant advantages over chlorpromazine. There are a number of other examples where addition of a highly lipophilic chlorine to a structure increases the potency, e.g. pheniramine and chlorphenamine, mefenamic acid and diclofenac.