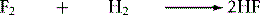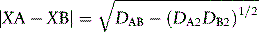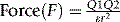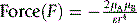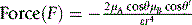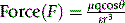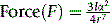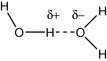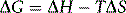Chapter 1 Bond type and bond strength
Introduction and synopsis
Of the 110 or so elements there are only about 22 which occur naturally in biological systems. The use of therapeutic agents introduces about another half dozen. In earlier centuries before the advent of selective antimicrobial agents the use of the toxic elements mercury and antimony was very popular to treat the then, more or less, incurable diseases of syphilis and malaria. If a cure was effective it was often a pretty close run thing with its opposite.
The structure of living systems is largely based on four elements: carbon, hydrogen, oxygen and nitrogen, the greatest part of any organism being hydrogen and oxygen in the form of water (ca. 70% of the human body). The predominant type of chemical bond which holds the structure of an organism together is the covalent bond which is formed when two atoms share their electrons. Covalent bonds vary in strength and properties according to the elements involved in the bond. One method of classifying the elements, which join together to form bonds, is according to their electronegativity. The electronegativity value for an element provides a measure of its affinity for electrons and hence its ability to attract a negative charge or conversely to give up some electron density and hold a positive charge. The electronegativity of some of the more common elements in biology is shown in Table 1.1; the derivation of electronegativity values is discussed later in the chapter.
Table 1.1 Electronegativities of the biological elements
| Metals | Non-metals |
|---|---|
| Na 0.9 | H 2.1 |
| Mg 1.2 | C 2.5 |
| Si 1.8 | N 3.0 |
| K 0.8 | O 3.5 |
| Ca 1.0 | F 4.0 |
| Mn 1.5 | P 2.1 |
| Fe 1.8 | S 2.5 |
| Co 1.9 | Cl 3.0 |
| Cu 1.9 | Se 2.4 |
| Zn 1.6 | I 2.5 |
Covalent bonds are usually formed between non-metals, with the more electronegative element in the bond attracting negative charge and creating a dipole (see Box 1.1).
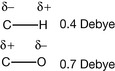
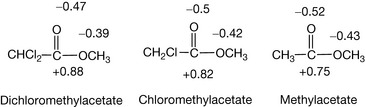
A bond between two different atoms always has a dipole moment in which the negative end of the dipole is centred on the electronegative atom. When two dipoles are in different molecules they can interact to form a weak reversible bond and these types of bonds are very important in drug action. A dipole moment is conveniently expressed in Debye (units in charge/metre). The dipole moments for C–H and C–O are shown above; the dipole–dipole interactions between positively charged atoms and oxygen in drug molecules are of more significance than those with carbon.
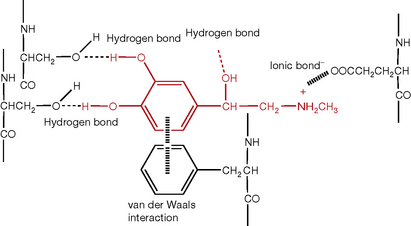
Bonds formed between metals and non-metals are ionic because of their large differences in electronegativity. In an ionic bond the electrons are not shared, the less electronegative element gives the more electronegative element its electron(s) and then they are held together in the bond by the attraction of the resultant opposite charges. A third type of bond is due to weak van der Waals forces which are strongest between bulky chemical groups. Figure 1.1 shows some examples of bond types using an example which approximates to the binding of adrenaline to a β2-adrenergic receptor where it interacts with several amino acid side chains on the G-protein.
Collectively, the making and breaking of various bond types drives the machinery of living organisms. Thus, for instance, the binding of calcium ions to certain proteins causes a change in their shape which results in muscle contraction. Similarly, the binding of a biologically active molecule such as acetylcholine to its receptor protein on muscle cell changes the shape of the pores in the muscle cell membrane, allowing sodium ions to enter the cell. This initiates a chain of events which results in entry of calcium ions into the cell and hence muscle contraction. Drug molecules act by altering the natural functioning of the machinery of the cells within the body (or pathogen), ideally, in order to repair an imbalance that is causing disease. Pharmaceutical chemistry concerns itself with which components within the structure of a molecule cause it to affect cells in a particular way. This brief introduction mentions several concepts relating to bond type which we will now discuss in more detail.
Covalent bonds
Covalent bonds are the strongest type of bond and are not usually involved directly in drug action. In fact, in most circumstances the formation of such bonds with a drug would give cause for concern because of the potential for alteration DNA and which could be potentially carcinogenic. However, there are circumstances in which the formation of a covalent bond is the therapeutic target for a drug. This is most notably the case for the alkylating agents used in cancer chemotherapy which are designed to form covalent bonds with the DNA of the cancerous cells and prevent them growing. As one might imagine, this process is also damaging to healthy cells within the body.
What is a covalent bond?
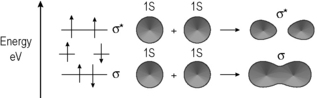
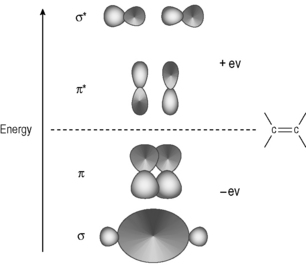
The theory of chemical bonding can be very complex and is worthy of several volumes in itself, but a basic knowledge will be sufficient for our purposes (see Box 1.2). The modern understanding of chemical bonding derives from quantum mechanics. A bond is formed when an atom shares its electrons with another atom. According to quantum mechanics, the electron behaves like a light wave and when electrons are shared they can either reinforce each other or cancel each other out in the way that light waves do when they destructively interfere. The waveform of an electron is known as a molecular orbital. The simplest atom is the hydrogen atom and when two hydrogen atoms join together they provide the simplest situation in which two molecular orbitals combine. Figure 1.2 shows the overlap between in-phase and out-of-phase hydrogen orbitals to form a bond.
An electron volt is a measure of force, albeit a very tiny amount of force.
1 eV = 1.602176487(40)×10−19 Joules. One Joule is approximately the energy required to lift a small apple quickly up 1 metre, or is the amount of energy required to heat 800 mL of dry, cool air through 1 degree Celsius.
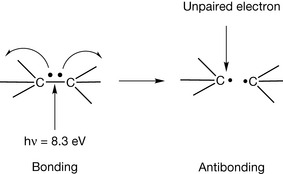
The in-phase orbitals produce a viable σ−bond which holds the atoms together and the out-of-phase orbitals produce an σ* anti-bonding orbital in which there is no overlap between the atomic orbitals. Since the bonding between the two hydrogen atoms, or indeed any pair of atoms, is due to an electronic wave phenomenon one way of examining bonds is by observing their interaction with electromagnetic radiation.
A pair of hydrogen atoms requires high energy/short wavelength radiation at about 150 nm (8.3 eV) to break the bond between them. Formation of the anti-bonding orbital can be represented as shown in Figure 1.3 where the two electrons making the bond are moved apart. The use of curly arrows to represent movement of electrons is shown in more detail in Box 1.3. Because bonding energy is quantised, the energy required is an exact figure for a given bond. The anti-bonding state is unstable and short lived so either the bond will re-form or a new bond will form with some suitable chemical species in the environment.
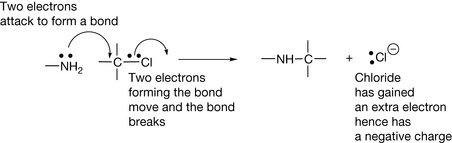
Throughout the book, extensive use is made of curly arrows to represent the movement of electrons. This convention is quite simple to understand.
Unpaired electrons (free radicals) usually have a very short time of independence since they will rapidly pair with another electron to form a bond. This is why free radicals are very damaging to living tissues.
Weaker bonds such as those found in DNA bases only require radiation around 260 nm (4.8 eV) to break them. This radiation is present in sunlight and hence the potentially damaging effects of overexposure to sunlight. A simple example of a weaker type of covalent bond is found in ethylene (Fig. 1.4). In this molecule the molecular orbitals have a more complex (hybrid) waveform than the simple 1S orbital of the hydrogen atom. The most important point to note is that the π bond in ethylene is formed by only partial overlap of the orbitals that form it. The stronger σ-bond in the molecule results from more complete overlap of the orbitals involved in it. Thus the energy required to excite the electrons in the π bond in ethylene to their excited (anti-bonding) state is lower than that required to excite those in the σ bond.
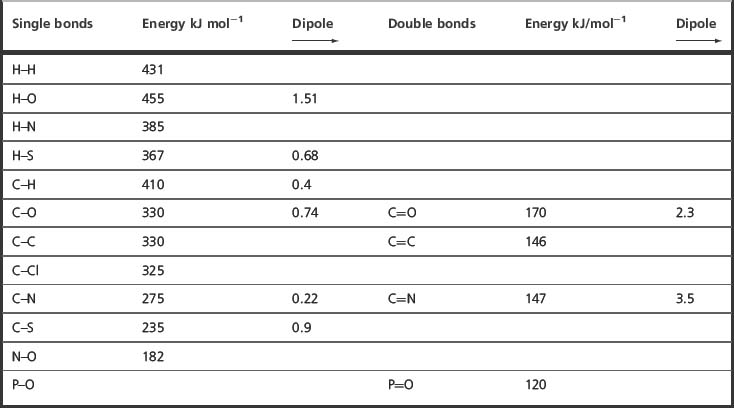
Table 1.2 shows the strength of some covalent bonds commonly found within drug molecules and within the biomolecules composing the human body, derived from calorimetric measurements.
A method for estimating the strength of a bond is by using calorimetry, which measures the heat of a reaction. For example, the reaction between fluorine and hydrogen
shown above in theory generates 542 kJ mol−1 of heat. Using the values shown in Table 1.2, on the deficit side, the hydrogen–hydrogen bond (431 kJ mol−1 ) and the fluorine–fluorine bond (155 kJ mol−1) are broken but on the credit side two H–F bonds (2 × 564 kJ mol−1) are formed. Thus the total energy derived from the reaction is 1128−586 = 542 kJ mol−1. The heat given out by a reaction is called the enthalpy of reaction and given the symbol Δ H, it is also given a minus sign, indicating that heat is produced by the reaction; thus the theoretical Δ H for the above reaction is −542 kJ mol−1. The heat of such a reaction can be measured using a calorimeter, which measures the amount of heat released by measuring the increase in temperature of water surrounding the sample being reacted. In this case, if 2 moles (40 g) of hydrogen fluoride were formed from hydrogen and fluorine the heat generated would be enough to raise the temperature of 1 litre of water from 0°C to beyond its boiling point (1 mole = 18 g of water requires 75.4 J to raise its temperature by 1°C). To break the bonds between H and F in 2 moles of HF, 1128 kJ of heat energy would be required. Thermal dissociation of bonds within molecules occurs, for example, when the analytical technique of inductively coupled plasma emission spectroscopy is used. In this technique temperatures of ca. 6000°C, provided by a heated plasma of argon gas, are used to disrupt the bonds in molecules so that the elements making them up can be analysed by exciting their electrons.
Dipole moments of bonds
The strengths of covalent bonds (Box 1.4) are less important in appreciating the relationship between drug structure and activity than the weaker interactions which occur between molecules as opposed to the forces within molecules. An example of a weak bonding force is that generated by the dipole moment of a bond. Linus Pauling produced a method for estimating the electronegativity of elements using equation 1 shown below.
Box 1.4 How strong is a chemical bond?
Often, bond energy and the force holding two atoms together are used interchangeably. In fact, the energy is equal to force × distance since the force has to be applied in order to stretch the bond for a certain distance before it will break. Chemical bonds can be broken by applying a force in the same way as one might apply weights to a spring until it snapped. Of course, one cannot literally suspend a weight from a bond in order to break it but one can stretch it to snapping point using appropriate chemicals. If it were possible to stretch a bond by attaching a weight, it would require a weight of 5–10 micrograms to break a single covalent bond (a microgram is about the size of a speck of dust). This might not sound like much but one has to remember that 1 mole of a compound contains 6.022 × 1023 molecules.
X is the electronegativity of elements A and B.
DAB, DA2 and DB2 are the bond dissociation energies between AB, A2 and B2.
By arbitrarily defining the most electronegative element, fluorine, as having an electronegativity value of 4.0 the values for all the other elements can be defined and this led to the values shown in Table 1.1.
Many covalent bonds have dipoles and dipole moments within drug molecules are important for their interaction with the biomolecules within cells which transmit the drug's action. The larger the dipole, the stronger the dipolar interaction with proteins and receptors within the cell. In addition, the strength of the dipole moment gives an indication of how likely the drug is to be degraded by reactants with an affinity for positive or negative charge. As indicated above, in most cases drug action is not based on covalent bond formation. There are exceptions (see Box 1.5), and thus the importance of covalent bond strength in pharmaceutical chemistry relates mainly to the stability of drugs in formulations, although it usually relates to the lifetime of the drug in vivo as well. The likelihood of a drug degrading depends on a combination of bond strength and the ease with which it is attacked at either positive or negative centres of charge. Thus the carbon–carbon bond within biomolecules is one of the least susceptible to degradation, certainly in vitro and to some extent in vivo, since it does not have a large dipole despite the fact that it stores a large amount of energy.
Box 1.5 Covalent bond formation in drug therapy
Covalent bond formation is important in the action of the following classes of drugs:
In Table 1.2 above it can be seen that the carbon–carbon double bond is stronger than the carbon–carbon single bond but listing its additive strength in this way belies the fact that one of the carbon bonds, the π bond, within the double bond is relatively weak and thus is quite susceptible to chemical attack.
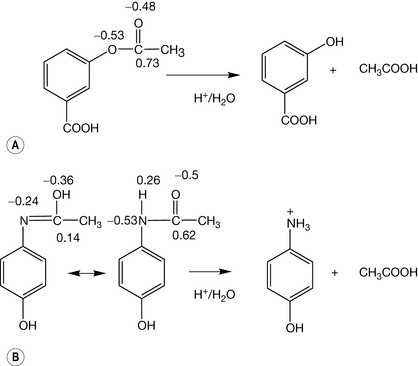
Some types of degradation reaction are more important with regard to drug molecules than others. Most often it is the polarised bonds within drug molecules which are most susceptible to chemical degradation, i.e. have the lowest activation energies in this respect. Figure 1.5 shows the partial charge distribution within aspirin and paracetamol.
Under conditions of acidic hydrolysis of an ester or amide the positively charged protons in the acidic medium will attack the most negative point within the group undergoing hydrolysis. In the case of aspirin the greatest negative charge is on the two oxygens within the ester group and protonation can occur at either of these positions, thus promoting attack by water at the positively charged carbon, which results in the breakdown of aspirin into acetic acid and salicylic acid. The activation energy for this reaction is low and hydrolysis occurs slowly at room temperature. It can also be seen from the polarised distribution of charge that the ester bond in aspirin is also susceptible to attack by HO− which will attach itself to the positively charged carbon and thus promote hydrolysis. The hydrolysis of esters is covered in Chapter 2. As pharmaceutical chemists, we are interested in what conditions are likely to cause our medicine to degrade. Obviously, aspirin should be kept dry.
The amide bond in paracetamol is also polarised and the charges on oxygen and carbon are not very different from those on the oxygens in the ester group in aspirin. However, amides hydrolyse much less readily than esters. One way of explaining this is that the carbonyl carbon is not as positively charged in the amide as in the ester and thus is less liable to attack by water or by the hydroxyl ion. However, calculation of charge distribution by chemical modelling software has its limitations and sometimes a less sophisticated view yields useful information. Without changing the charge on the amide group it is possible to write a resonance form for the amide where the proton is on the oxygen. The concept of resonance can be applied throughout organic chemistry and in simple terms all it means is that electron density is shared more evenly throughout a structure or group within a structure. As was discussed at the beginning of this section, the most stable bonds are those where electron density is distributed evenly and thus there is likelihood of degradative attack. If the charge distribution is calculated for the resonance form of paracetamol the charge distribution across the oxygen, carbon and nitrogen is evened out. Thus, activation energy required for hydrolysis of an amide is greater than that required for hydrolysis of an ester. It is important to note that once the activation energy for hydrolysis has been overcome the acidic hydrolysis of an amide proceeds rapidly and irreversibly, i.e. it is thermodynamically favourable and releases more energy than hydrolysis of the equivalent ester. In this case a non-polar molecule activation energy can be high but this is followed by a high release of energy (Box 1.6).
Box 1.6 Activation energy versus thermodynamic energy release
The absence of a dipole moment means that the activation energy required for degradation of a carbon–carbon bond to occur is high; for example, propane gas is a rich source of thermal energy (3671 kJ mol−1 is enough to boil several kettles of water when camping under the most arctic conditions). However, it has to be heated to a high temperature, e.g. with a match, gently warming it does not work, before it ignites and reacts with oxygen. Thermodynamically, this reaction lies far in the direction (to the right) of the formation of carbon dioxide from the carbon atoms and water from the hydrogen atoms in the propane. In contrast, the energy required to activate the degradation of an ester by acidic hydrolysis described below is minimal but the energy released by the reaction is small and thermodynamically the reaction lies only slightly in the direction of the reaction products.
Thus in chemical reactions we have two ideas: the ease with which the reaction can be triggered and the amount of energy released by the reaction.
![]() Self Test 1.2
Self Test 1.2
From the charge distribution in the following esters, arrange them in order of increasing rate of hydrolysis caused by HO−.
How much do you think the rate of acid hydrolysis might vary?
Under acidic conditions (pH 1) at room temperature, aspirin hydrolyses in a few hours whereas paracetamol can be stored at room temperature under acidic conditions (pH 1) for many days without much hydrolysis occurring. We will return to the properties of esters and amides in a little more detail in Chapters 3 and 4.
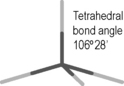
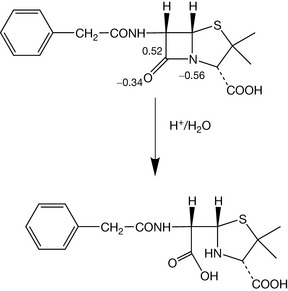
As can be seen in Figure 1.5 aspirin and paracetamol have many bonds in their structure but it is only the bonds where the charge distribution is polarised which are susceptible to hydrolytic attack. Ester bonds are among the most labile bonds which are found in drug molecules. Another type of bond which limits shelf life is found in the lactam ring of penicillins. This bond may look like an amide bond, and calculation of charge density on the C, N and O atoms in the bond does not reveal any great difference from the atoms in the amide bond of paracetamol. At this point an additional concept is required, which is that ring strain renders the lactam ring much more susceptible to nucleophilic attack (i.e. where a negative species attacks a positive) or electrophilic attack (where a positive species attacks a negative) and this is indeed also responsible for its biological activity (Ch. 22). Ring strain arises from the fact that carbon atoms have preferred angles between the bonds attached to them. The preferred angles for the four bonded carbons in the ring (A and B) is 109°28′ (Box 1.7) and for the three bonded carbonyl carbons 120°; in the lactam ring the carbons are forced to have angles approximating to 90°. The strained ring reduces the activation energy required for the hydrolysis reaction to proceed (Fig. 1.6).
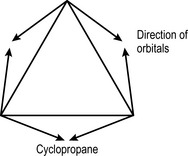
Four-bonded carbon prefers tetrahedral geometry. Any deviation from the preferred angle produces strain. Rings with six atoms or more are not strained. In five-membered rings the strain is fairly minimal but four- and three-membered rings are quite strained. Cyclopropane is the simplest three-membered carbon structure. In fact, the orbitals of the carbon atoms in this ring can be represented as being bent away from the ring with an angle of 9.4°, indicating that they do not overlap as extensively as in an unstrained structure.
Nevertheless, cyclopropane is quite stable compared to the lactam ring in a penicillin because its even charge distribution presents no point for nucleophilic or electrophilic attack and thus the activation energy for its reaction is high. Indeed, it was even used as a volatile anaesthetic, although it has fallen out of favour since a spark overcoming the activation energy for the molecular reaction results in a large thermodynamic change, i.e. an explosion.
The distortion of the ring geometry is further reflected in the IR absorption of the carbonyl carbon within the lactam ring which is at high energy, ca. 1770 cm−1. This is due to the lack of interaction between the nitrogen and the carbonyl oxygen, which normally occurs in amides, resulting from the distorted geometry. IR provides a simple method for examining bond strength (Box 1.8).
Box 1.8 Resonance and infrared spectrophotometry as a simple tool for observing bond strength
The concept of resonance was first applied to the benzene ring where all the bonds were found to be identical. In the structure drawn on the left it looks as if there are two different types of bond and the structure is more correctly drawn as on the right with the electron density from the double bonds evenly distributed throughout the molecule.
There are a number of ways of observing the electron density within double bonds. One simple method is infrared spectrophotometry (IR). IR observes interaction of IR radiation (heat radiation) with bonds in molecules. IR radiation causes bonds to stretch: the lower the wavenumber (1/λ) of the IR radiation required to stretch a bond the weaker a bond. The energy of the radiation used in IR is reported as 1/wavelength in centimetres. The higher the number, the higher the energy. Carbonyl groups absorb IR radiation between 1650 cm−1 and 1800 cm−1. The carbonyl group of the ester in aspirin has an absorption at 1760 cm−1 whereas the carbonyl group in the amide of paracetamol absorbs at 1650 cm−1. This indicates that the carbonyl in paracetamol has lost some of its electron density and thus some of its strength relative to the carbonyl in aspirin due to the resonance shown in Figure 1.5.
Intermolecular or inter-atomic forces
Introduction
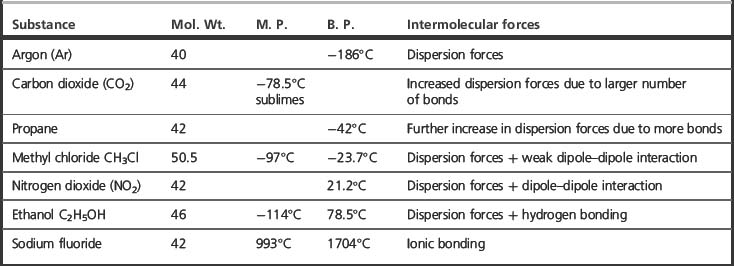
The actions of drugs on biological molecules involve intermolecular forces. There are a number of types of interactions between molecules, ranging from strong ionic interactions to weak but additively strong van der Waals interactions. The different strength of the different types of intermolecular force can be observed in the melting points of the series of compounds shown in Table 1.3, all of which have similar molecular weights but which exhibit different types of intermolecular interactions. The high degree of precision with which a naturally occurring ligand binds to its site of action depends on a delicate balance of the different intermolecular forces (see also Box 1.9). Drugs usually do not fit a given receptor with the same degree of precision as a natural ligand but have actions which originate in strong binding to parts of the receptor. With a knowledge of receptor structure, the intermolecular forces required for binding can be used to model ideal drug structures which will bind strongly.
Box 1.9 A digression on the matter of force versus energy
It is quite easy to use the terms force and energy interchangeably. However, there is a difference. Intermolecular force decreases as the distance between two atoms or molecules increases. Thus, the spring analogy used to describe a bond is not strictly correct since the resistance produced by a spring when it is stretched beyond the point where it will spring back becomes high. Two better analogies are: moving two strong magnets apart, where the force decreases with distance until it is too weak to move them back together; or pulling a fence post out of some sticky mud where initially the resistance is high but, as it becomes less deeply buried, it moves more easily. In the two activities described, the energy required is the sum of the effort expended in overcoming the force of resistance (analogous to bond energy) which decreases with the distance moved from the starting point.
Ionic bonds
When two atoms which are widely different in electronegativity are brought into contact, rather than a covalent bond forming, the more electronegative atom will take an electron or electrons from the less electronegative atom, thus becoming negatively charged while the less electronegative atom becomes positively charged. A simple example is the reaction between a sodium atom (a soft metal) and a chlorine atom (gas). Having gained positive and negative charges, the two atoms are now attracted to each other, and if they are considered to be point charges the magnitude of the energy of the attraction would be 694 kJ mol−1 if they were separated by a distance of 200 picometres in a vacuum. Such an energy is greater than the strength of most covalent bonds. However, in practice many ionic bonds can be readily broken using water as a solvent because of the strength of the bonds formed between the water and the positive and negative ions.
When a salt is dissolved in a solvent, the force of attraction between its ions is governed largely by the dielectric constant of the medium in which it is dissolved and the distance of separation between the two ions. The force of attraction between two ions can be calculated according to equation 2.
Where Q1 and Q2 are the charges on the ions, r is the distance of separation and ε is the dielectric constant of the medium.
The dielectric constant of water is defined as 80 relative to that of a vacuum. Thus energy of the bond between two point charges separated by two picometres in water would be 80 times less than the energy of the bond in a vacuum and have a value of ca. 8 kJ mol−1. Water is the best solvent for ionic compounds because of its high dielectric constant. Table 1.4 shows the dielectric constants of some common solvents. Sodium chloride dissolves in water at 1 g in 2.8 mL and in glycerol, which has half the dielectric constant of water; 1 g dissolves in 10 mL. Sodium chloride is almost insoluble in ethanol; in this case the dielectric constant is too low for the inter-ionic attraction to be overcome.
Table 1.4 Dielectric constants of some common solvents
| SOLVENT | DIELECTRIC CONSTANT ε |
|---|---|
| Water | 78.5 |
| Glycerol C3H5(OH)3 | 42.5 |
| Acetonitrile CH3CN | 36.2 |
| Methanol CH3OH | 32.6 |
| Ethanol CH3 CH2OH | 24.3 |
| Benzene C6H6 | 4.6 |
In living systems, ionic associations take place in aqueous media and the energies involved in these types of interaction are between 4 and 40 kJ mol−1. The reason for the wide range in energies of ionic interactions is that the strength of the interaction depends on the dielectric constant of the environment and on how closely the two ions approach. Strong interactions between ions may occur because within cell membranes and in the hydrophobic cavities of proteins the amounts of water present may be greatly reduced, resulting in a decrease in the dielectric constant of the environment. The binding of an ionic drug with hydrophobic groups attached may further reduce the water present in a binding site. Since the force of attraction between ions varies as the square of the distance of their separation ionic forces act over a greater distance than the other intermolecular forces described below. The long-range attraction of ions and the consequent speed of association between oppositely charged ions means that ionic forces are important in driving much of the mechanical work of the cell. Table 1.5 shows the principal ionic groups which are found in proteins, along with their pKa values and percentage ionisation of these groups at the physiological pH of 7.4. These charged groups within protein structures provide sites for the binding of ionic drugs through formation of ion pairs and non-ionic drugs through ion–dipole interactions.
Table 1.5 Ionised groups found on proteins
| Acidic groups | pKa | % Ionisation at pH 7.4 |
|---|---|---|
| Terminal carboxyl (–COO−) | 1.8–2.4 | 100 |
| Aspartic acid side chain (–CH2CH2COO−) | 3.7 | 99.98 |
| Glutamic acid side chain(–CH2CH2CH2 COO−) | 4.3 | 99.9 |
| Basic Groups | ||
| Terminal ammonium (–NH3+) | 7.5–10.3 | 55.7–99.9 |
| Arginine (–NH−(NH2)C=NH2+) | 12.5 | 100 |
| Lysine (–CH2CH2CH2CH2NH3+) | 10.5 | 99.9 |
| Histidine | 6.0 | 4 |
Where ionic interaction is an important component in the interaction of a drug with a receptor or G-protein, other forces may come into play in reinforcing the ionic interaction and these include particularly dipole–dipole interactions, hydrogen bonding and van der Waals forces (as illustrated in Fig. 1.1 for adrenaline). Thus the thermodynamics of drug action is based on the interplay between several forces which are relatively weak compared to the covalent bonds which make up the scaffolding of living systems. That is not to say that these weaker forces do not result indirectly in the breaking or formation of covalent bonds, but this is a consequence of the initial interaction of the drug with its receptor, which may trigger off enzyme action which results the making or breaking of covalent bonds.
Dipole–dipole interactions
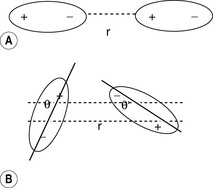
The strength of a dipole (Fig 1.7) within a bond depends on the relative electronegativities of the atoms composing the bond. Where a molecule contains a dipole, it can interact with other molecules containing a dipole. The force between two dipoles is give by equation 3.
Where μA and μB are the dipole moments of the molecules involved in the interaction, ε is the dielectric constant on the medium in which the interaction is taking place. r is the distance between the two dipoles.
Equation 3 holds if the dipoles interact in a linear fashion as shown in Figure 1.7A: in this case the interaction is maximal. Where the interaction is at an angle as in Figure 1.7B, the interaction is given by equation 4 (cos θ is <1).
Dipolar interactions are very dependent on temperature since an increase in temperature promotes random motion in molecules, which makes the linear orientation of the dipoles less likely. The interaction between dipoles falls off with the fourth power of distance and thus acts over a much smaller distance than ionic interactions, which are dependent of r2. The strength of these interactions is within an energy range of 0.4–4 kJ mol−1. When brought into close contact within a receptor, for example, ionic interactions, dipolar interactions with a drug become significant since contact is both close and at the optimum angle. If dipole–dipole interactions are important for activity at a receptor, any slight spatial adjustment in order to accommodate of a molecule could result in loss of activity.
Ion–dipole interactions
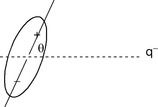
The interaction between an ion and a dipole (Fig. 1.8) depends on the cube of the distance between them (equation 5). Like dipole–dipole interactions, these interactions depend on the orientation of the dipole with respect to the ion.
Charged dipole interactions tend to be stronger than dipole–dipole interactions and have energies in the range 1–10 kJ mol−1. These interactions are responsible for the solvation of ions in solution and are responsible for promoting the dissolution of ionic solids.
Van der Waals forces
Van der Waals forces (or London dispersion forces) arise in molecules where the charge appears to be evenly distributed because vibrations within molecules cause transient dipoles. These transient dipoles induce transient attractive dipoles in adjacent molecules with energies in the range 0.3–1.9 kJ mol−1. As can be seen from equation 6, these forces only operate at very close range and fall off at the rate of the seventh power of the separation of the interacting species.
Where I is the first ionisation potential of the atom or molecule involved in the interaction, α is its polarisabliity and r is the separation distance.
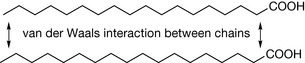
However, at close range the additive effect of the interactions can be high and, for instance, it has been estimated that the additive effect of attraction between the atoms involved in the hydrocarbon chain of stearic acid is 33.5 kJ mol−1 (Fig. 1.9).
The strength of the forces involved depends on the polarisablility of the structural component in which the transient dipole is induced. The increased polarisability is related to increased molecular weight and large atoms within a structure increase the van der Waals interactions considerably because of their high polarisabilities. This can be seen in the case of C2H5Cl (B.P. 12°C), C2H5Br (B.P. 38°C) and C2H5I (B.P. 72°C). The dipole moment of these molecules increases from iodide to chloride but this is more than offset by the increasing van der Waals interactions from chloride to iodide.
Charge transfer interactions
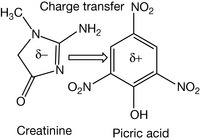
Charge transfer interactions are also thought to be important in the interaction between drugs and proteins. These interactions are quite weak being <1 eV. A commonplace example is the complex formed between picric acid and creatinine (Fig. 1.10) in solution which is used as a clinical assay to normalise the concentration of urine samples. Charge transfer requires a π-base, in this case creatinine which is electron rich and a π-acid, in this case picric acid which is electron deficient due to electrons being withdrawn from the aromatic ring. The formation of the complex results in a shift in the ultraviolet spectrum, which can be quantitatively measured.
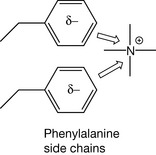
Such interactions are also possible between ions and electron-rich systems. In some cases, charge transfer interactions may be involved in receptor binding, e.g. interactions between a quaternary ammonium centre in a drug and phenylalanine side chains in a protein (Fig. 1.11).
The hydrogen bond, solvation and entropy of mixing
The hydrogen bond occurs whenever hydrogen, which is bonded to an electronegative atom such as N or O, interacts with another electronegative atom. The most immediately familiar type of hydrogen bond is that which occurs between water molecules but the hydrogen bond is also very important in the structure of DNA and proteins. Compared to other hydrides of electronegative elements, the boiling point of water is high, e.g. compare water 100°C, HF 20°C and HCl −33°C. The boiling point of water is thus elevated above that which would be predicted on the basis of its molecular weight (MW). For instance H2S has a boiling point of −61°C although its MW is 34 compared with a MW of 18 for water. The elevation of boiling point in the case of water is due to the strong hydrogen bonds between its molecules. Hydrogen bonds are specialised dipole–dipole interactions. The strength of the hydrogen bonds in water is partly due to the high electronegativity of oxygen and partly due to the favourable geometry of the water molecule that allows the formation of two hydrogen bonds per molecule and the formation of complex networks of water molecules. The hydrogen bond in water is expressed as shown in Figure 1.12.
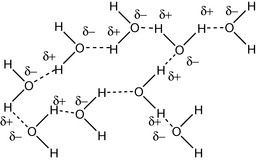
The weaker hydrogen bond is represented by the dotted line. Hydrogen bond strength in water is around 20 kJ mol−1. The bulk structure of water consists of cluster of hydrogen-bonded molecules (Fig. 1.13). If a water-soluble alcohol such as methanol is mixed with water, the temperature rises upon mixing. This is due to the thermodynamics of mixing.
We have touched on the ideas of thermodynamics in the process of discussing the release of energy when bonds are broken and reformed, e.g. propane burns in air, and because of the large release of energy resulting from the formation of H–O and C–O bonds. If we examine the preparation of a solution containing 20% w/w methanol in water by mixing the two liquids, it is not immediately apparent why the solution should get warmer.
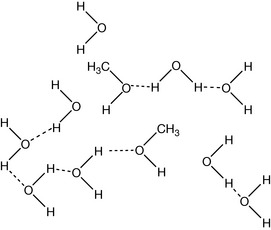
On closer examination, it can be seen that the methanol can only hydrogen bond at one end of the molecule (Fig. 1.14) and thus it causes many of the hydrogen bonds between water molecules to break, and this process would be expected to require an investment of energy rather than generating heat. This simple, easily observable example necessitates the introduction of another concept into the generation of energy by chemical change. We have observed earlier when weaker bonds are replaced with stronger bonds energy is released. In the current example, it is the second law of thermodynamics that explains the warming of the methanol–water mixture. The change in energy during a process according to the second law of thermodynamics can be stated as follows:
Where ΔG is the Gibbs free energy of a process, which is a measure of how thermodynamically favourable it is. We have already encountered ΔH in the form of the heat generated or by a chemical change, but heat alone does not fully describe a process. For instance, when ice melts the heat invested in breaking the bonds between the water molecules in the ice cannot be readily recovered in order to reverse the process; although the water remains at 0°C when it melts it does not spontaneously turn back into ice. The reason that processes are not readily reversible is because some of the energy generated or invested during the change takes the form of entropy, ΔS (Box 1.10). Entropy concerns the behaviour of molecules during a chemical or physical change. When ice melts, the molecules in the water produced have greater freedom to move about (kinetic energy) and greater freedom to vibrate and rotate (internal molecular energy) than they did in the ice. Such energy processes, which change the way in which molecules behave in space, are termed entropy and are not reversible without investing energy. For example, if we want to refreeze water to produce ice using a freezer we have to use electrical heating to evaporate the refrigerant in the freezer in order to produce the cooling required in the same way as eau de cologne cools the skin as it evaporates. The greater the freedom of molecules due to being able to move, and for complex molecules this includes vibration, rotation etc., as well as translational movement, the greater the entropy of a system. The heat generated when methanol is mixed with water indicates that mixing in this case is energetically favourable despite the loss of hydrogen bonding shown in Figure 1.14. The processes occurring during the mixing are complex but at least in part some of the energy emitted is due to a loss of vibrational energy in the methyl group, possibly due to compression by water molecules. This can be seen in the IR spectrum of the methyl group where the C–H stretching occurs at higher energy in methanol–water mixtures compared to pure methanol (Watson et al. unpublished). Such events at the molecular level may seem a little abstract but what we observe is that the methanol–water mixture emits heat radiation upon mixing. Indeed, our perception of the heat is due to transfer of some of the vibrational entropy to the molecules making up our skin surface from the warm molecules that our skin is in contact with. The concept of entropy or energy change accompanying a change in the order of molecules is important in relation to the binding of drugs to their sites of action. The binding of a drug to a receptor can be extremely complex with regard to the effect on other molecules in solution since it can: displace bound water, displace ions which can have an effect both on the structure of the water surrounding the binding site, and change the position of hydrophobic or hydrophilic groups which can affect the structure of the protein to which it is binding and also the structure of the water in the environment. However, overall binding has to be thermodynamically favourable and for many drugs binding has to be more favourable than for the natural ligand at the binding site.
Box 1.10 Explanations of entropy
Entropy is a troublesome concept, so it is worth trying to define it in a number of different ways.
Solvation of ions
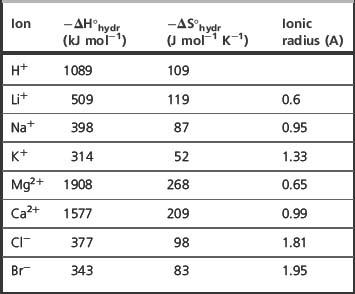
The medium for solvation of greatest interest in biological systems is water. As was seen in the case of hydrogen bonding, water, because of its strong dipole, has an affinity for both positive and negative centres of charge. This makes it an excellent solvent for ionic materials. For instance, sodium chloride will dissolve quite readily in water despite the high energy of the ionic bonds holding the ions together because when it dissolves there is a large gain in energy in the form of the dipolar bonds formed between water and the free ions in solution. Table 1.6 shows the hydration enthalpies and entropies for a number of ions. In water, ions are ‘structure making’ and the heat produced is due to favourable enthalpy where bonds are formed. The negative entropy values in the table indicate that entropy decreases when solvation occurs.
In general, the higher the charge density (charge/ionic radius) of an ion, the greater its enthalpy and entropy of hydration. When taking into account ionic radius, the degree of salvation of the ion has to be considered; thus, for instance, the sodium ion is too large for potassium ion channels since, although its ionic radius is smaller overall, it has a larger radius due to the fact that it is in association with more water molecules than potassium. Most organic cations have relatively low charge densities compared to inorganic cations and hence relatively low levels of hydration. As can be seen from Table 1.6, cations tend to be more extensively hydrated than anions. The mechanics of living systems are essentially driven by electrostatic interactions between ions and proteins and water.
![]() Self Test 1.1
Self Test 1.1
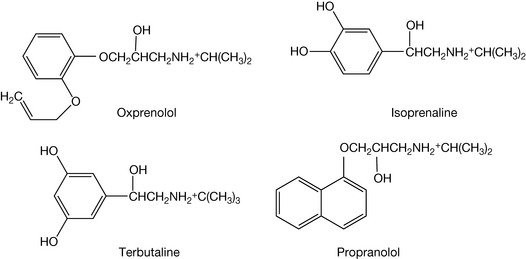
Mimic: isoprenaline and terbutaline. Block: oxprenolol and propranolol.
The blocking agents will bind strongly to the carboxylic group via their amine groups and also interact where the side chain hydroxyl of adrenaline binds.