14 Salivary gland disease
ASSUMED KNOWLEDGE
It is assumed that at this stage you will have knowledge/competencies in the following areas:
If you think that you are not competent in these areas, revise them before reading this chapter or cross-check with relevant texts as you read.
INTENDED LEARNING OUTCOMES
At the end of this chapter you should be able to:
APPLIED ANATOMY
Parotid gland
The parotid gland is the largest of the salivary glands. There is considerable variation in size between individuals but for any individual both sides are similar. Although embryologically the parotid consists only of a single lobe, it is convenient to speak of a superficial lobe (superficial to the plane of the branches of the facial nerve) and a deep lobe (deep to that plane). The superficial lobe of the parotid is limited laterally by the preparotid fascia and overlying fat and skin. The zygomatic arch and temporomandibular joint is superior, the masseter muscle is anterior and posteriorly are the cartilaginous external auditory meatus, the mastoid process and the sternocleidomastoid muscle. The isthmus of the parotid gland is bounded by the ramus of the mandible anteriorly and the posterior belly of the digastric behind. The deep lobe is related to the medial pterygoid anteriorly, the styloid apparatus posteromedially and the internal jugular vein. Its deep surface lies immediately superficial to the tonsillar fossa and neoplasms arising from the deep lobe often present as tonsillar masses. The trunk of the facial nerve arises from the stylomastoid foramen just behind the styloid process. It enters the posterior parotid and becomes rapidly more superficial before dividing into two divisions, the superior larger zygomaticotemporal and the smaller buccocervical division. The facial nerve then further divides into five main branches—temporal, zygomatic, buccal, mandibular and cervical—as it passes forwards in a plane separating the superficial and deep lobes. The main collecting duct arises within the superficial lobe and passes horizontally forwards, parallel to the buccal branch in a direction corresponding to a line joining the tragus to a point midway between the alar and the commissure. The duct crosses the masseter muscle horizontally, then turns at right angles to pierce the buccinator muscle and enters the oral cavity at the parotid papilla opposite the second upper molar.
Submandibular gland
The submandibular gland consists of two lobes—superficial and deep—which are folded over each other posteriorly around the posterior free border of the mylohyoid muscle. The larger superficial lobe lies within the submandibular triangle covered by deep cervical fascia, platysma, fat and skin. The mandibular branch of the facial nerve loops down below the lower border of the mandible. It usually lies on the deep surface of the fascia but may lie between the fascia and the overlying platysma. The deep lobe of the submandibular gland is ‘suspended’ from the lingual nerve and sublingual ganglion by the parasympathetic secretomotor fibres, which must be carefully divided without damaging the sensory fibres of the lingual nerve when removing the gland. The hypoglossal nerve lies in the bed of the deep lobe. The submandibular duct arises from the anterior pole of the deep lobe and runs forwards in the floor of the mouth, crossing the lingual nerve as it passes medially to innervate the tongue.
Sublingual gland
The sublingual gland is a complex of major and minor salivary glands, which occupies much of the submucosa of the floor of the mouth extending posteriorly to the second molar region. It lies partly within the sublingual fossa of the mandible above the mylohyoid line. Up to 20 small ducts open directly in the floor of the mouth. Some open directly into the submandibular duct.
When considering the many and various conditions and disorders that can affect the salivary glands, it is useful to classify the various conditions according to the standard ‘surgical sieve’ (see Ch. 2).
DEVELOPMENTAL DISORDERS
Developmental disorders such as accessory lobes, ectopic development, agenesis, duct atresia and congenital fistula are rarely a problem to the dental practitioner.
INFLAMMATORY DISORDERS
Viral
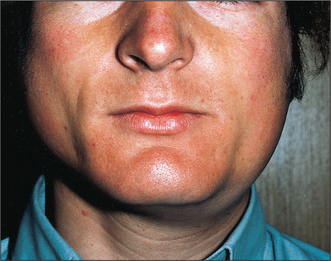
Mumps (Fig. 14.1) is the most common cause of acute painful parotid swelling affecting children. It is endemic in urban areas and spreads via airborne droplet infection of infected saliva. The disease starts with a prodromal period of 1 or 2 days, during which the child experiences feverishness, chills, nausea, anorexia and headache. This is typically followed by pain and swelling of one or both parotid glands. The parotid pain can be very severe and is exacerbated by eating or drinking. Symptoms resolve spontaneously after 5–10 days. In a classical case of mumps the diagnosis is based on the history and clinical examination: a history of recent contact with an affected patient and bilateral painful parotid swelling is sufficient. However, the presentation may be atypical or sporadic or have predominantly unilateral or even submandibular involvement. In this situation mumps-specific IgM can be identified in the serum as early as 11 days following original exposure to the virus. This antibody is also detectable in the saliva. One episode of infection confers lifelong immunity.
The treatment of mumps is symptomatic. Regular paracetamol and encouragement to maintain fluid intake is important. Complications such as orchitis, oophoritis, meningoencephalitis, pancreatitis and sensorineural deafness are uncommon in children but are more likely in adults. The incidence of mumps is becoming less common as, increasingly, children are receiving the mumps, measles and rubella vaccine at an early age. However, it is important to bear it in mind as a diagnosis whenever a febrile fretful child presents with salivary gland enlargement.
A number of other viral agents—Coxsackie A and B, parainfluenza 1 and 3, ECHO and lymphocytic choriomeningitis—can all cause identical signs and symptoms. Cytomegalic inclusion disease affects mainly newborn infants and children. It is believed to be an intrauterine infection which becomes manifest after birth.
Bacterial
Acute ascending bacterial sialadenitis affects mostly the parotid glands (Fig. 14.2). Historically, it was described in dehydrated, cachectic patients often following major abdominal surgery when the patient was on a ‘nil by mouth’ regime. The reduced salivary flow and oral sepsis resulted in bacteria colonizing the parotid duct and subsequently involving the parotid parenchyma. With current hospital practice and improved oral hygiene, patients are rarely allowed to become dehydrated and this clinical pattern is uncommon. The typical patient presenting with an acute ascending bacterial parotitis now is an otherwise fit young adult with no obvious predisposing factors.
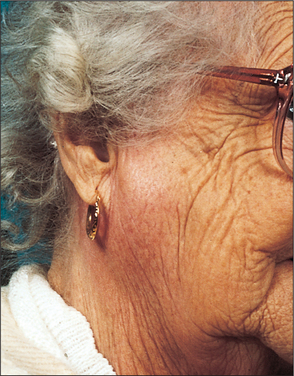
Fig. 14.2 Ascending bacterial parotitis often affects fit young patients as well as the elderly, without obvious predisposing risk factors.
Clinical presentation is of the onset of tender, red, painful parotid swelling over a few hours. There is associated malaise, pyrexia and often regional lymphadenopathy. Pain is exacerbated on attempting to eat or drink. The parotid swelling may be diffuse but often it is localized to the lower pole of the gland, presumably because the infection tends to localize under the effect of gravity.
If the gland is gently ‘milked’ by massaging the cheek cloudy turbid saliva can be expressed from the parotid duct; this should be cultured. The infecting organism is usually Staphylococcus aureus or Streptococcus viridans. Sialography must never be undertaken during the acute phase of infection as the retrograde injection of infected material into the duct system will result in bacteraemia. Ultrasound imaging shows the characteristic dilatation of the acinae.
If the patient presents at an early stage before abscess formation, the infection can usually be controlled with antibiotics. The clinician should not delay waiting for the results of bacterial culture: in a patient not allergic to penicillin a combination of a broad-spectrum penicillin (ampicillin) and a penicillinase-resistant agent (flucloxacillin) is usually effective. In patients allergic to the penicillins, clindamycin is a good alternative. This antibiotic is actively secreted in saliva. The levels in the saliva exceed those in the circulating serum. If the gland becomes fluctuant, indicating abscess formation, the pus must be drained. Occasionally it is possible to drain the abscess by aspirating the pus through a large-bore hypodermic needle, but usually it is necessary to undertake formal surgical drainage under general anaesthesia.
Care must be taken when making the incision not to damage a branch of the facial nerve. Only the skin should be incised and the operation continued with blunt dissection using sinus forceps. A small drain is inserted for 24 hours. Surprisingly, following removal of the drain, the incision heals uneventfully and a salivary fistula does not occur.
Chronic bacterial sialadenitis is far more common in the submandibular salivary gland and usually occurs secondary to chronic obstruction (see below). Unfortunately the submandibular gland has a poor capacity for recovery following infection and in most cases, following control of any acute symptoms with antibiotics, the gland itself must be removed. The operation is performed in hospital under general anaesthesia. Great care must be taken not to damage the mandibular branch of the facial nerve when making the incision, the lingual nerve when mobilizing the gland and clamping the duct and the hypoglossal nerve when separating the gland from the floor of the submandibular triangle.
Chronic sclerosing sialadenitis is a condition that can affect the submandibular gland after a long period of chronic bacterial infection. Progressive atrophy and fibrosis eventually results in a small hard mass, which may undergo dystrophic calcification. This then presents in the submandibular region as a Kuttner tumour (the word tumour being used in its literal sense as a lump and not as a neoplasm).
Recurrent parotitis of childhood
Recurrent parotitis of childhood exists as a distinct clinical entity but little is known regarding its aetiology and prognosis. It is characterized by the rapid swelling of usually one parotid gland, accompanied by pain and difficulty in chewing, as well as systemic symptoms such as fever and malaise. Although each episode of parotid swelling is usually unilateral, the opposite side may be involved in subsequent episodes. Each episode of pain and swelling lasts for 3–7 days and is followed by a quiescent period of a few weeks to several months. Occasionally episodes are so frequent that the child loses a considerable amount of schooling. The onset is usually between 3 and 6 years, although it has been reported in infants as young as 4 months. The diagnosis is based on the characteristic history and is confirmed by sialography, which shows a very characteristic punctate sialectasis often likened to a snow storm against a dark night sky (Fig. 14.3).
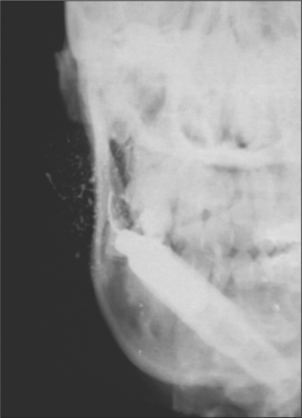
Fig. 14.3 Characteristic punctate sialectasis seen in recurrent parotitis of childhood: the ‘snow storm’ appearance.
Traditionally the episodes of parotitis have been treated with antibiotics and symptoms settle within 3–5 days on such a regimen. Occasionally, recurrent episodes are so frequent that prophylactic antibiotics are required for a period of months or years. Symptoms seem to resolve spontaneously at puberty but some of these patients go on to develop chronic bacterial parotitis later in life.
‘Specific infections’ (granulomatous sialadenitis)
Swelling of the salivary glands may occasionally be caused by mycobacterial infection, cat-scratch disease, syphilis, toxoplasmosis, mycoses, sarcoid, Wegener’s and other granulomatous disease.
Allergic sialadenitis
A variety of potential allergens causing acute parotid swelling have been identified. Some foods, drugs (most frequently chloramphenicol and tetracycline), metals such as nickel and pollens have been incriminated.
Radiation sialadenitis
Following the start of therapeutic irradiation, when the parotid glands are within the radiation field the patient develops an acute parotitis, usually after 24 hours. The glands are swollen and tender, there is a marked rise in salivary amylase and the salivary flow rate is reduced. The reaction is self-limiting and resolves after 2 or 3 days, even though the radiotherapy continues. This reaction is quite distinct from the permanent radiation atrophy that occurs with therapeutic doses above 40 Gy, which develops progressively after the radiation has been completed and results in xerostomia.
HIV-associated sialadenitis
Chronic parotitis in children is almost pathognomonic of HIV infection. In adults a sicca syndrome and lymphocytic infiltration of the salivary glands are more usual. The presentation of HIV-associated sialadenitis is very similar to classical Sjögren’s syndrome (see later). Dry mouth, dry eyes and swelling of the salivary glands together with lymphadenopathy suggests the diagnosis. Histologically the condition closely resembles Sjögren’s syndrome and differentiation may be difficult. However, autoantibodies—including antinuclear, rheumatoid factor, SS-A and SS-B—are absent unless the patient coincidentally has a connective tissue disorder. AIDS-associated lymphoma presenting as salivary gland swelling has also been described.
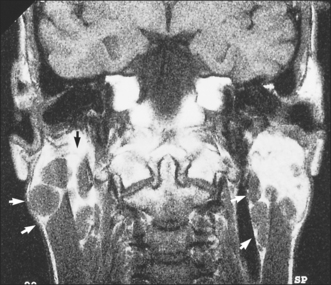
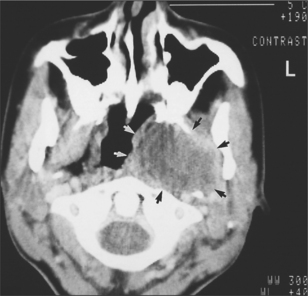
Another presentation of salivary gland disease in HIV-positive patients is multiple parotid cysts causing gross parotid swelling and significant facial disfigurement. On imaging with CT or MRI the parotids have the appearance of a Swiss cheese, with multiple large cystic lesions (Fig. 14.4). The glands are not painful and there is no reduction in salivary flow rate. Surgery may be indicated to improve the appearance.
Sialadenitis of minor salivary glands
Whenever the major salivary glands are involved, the same histological changes will be present in the minor glands. Similarly, Sjögren’s syndrome invariably involves the minor glands—and indeed the diagnosis is often confirmed on the basis of a minor salivary gland biopsy.
Stomatitis nicotina is a chronic inflammatory disorder of the minor salivary glands of the palate, seen in heavy smokers. The appearance is of multiple small target lesions in which the central opening of the gland is inflamed and bright red, but the surrounding halo of palatal mucosa is white as a result of hyperkeratosis. The condition does not carry a risk of malignant change and is reversible, if the patient can be persuaded to stop smoking.
Acute necrotizing sialometaplasia is an unusual condition, which was first described in 1973. It occurs only on the hard palate in the molar region in the vault of the palate midway between the midline and the gingival margin. It is seen only in heavy smokers. It has a characteristic appearance, which resembles a carcinoma with central ulceration and raised erythematous margins. The ulcer may be as much as 3 cm in diameter. As it so closely resembles a carcinoma the diagnosis is often made on the basis of surgical biopsy. The lesions are self-healing, but often take 10–12 weeks to resolve. As they are extremely painful it is helpful to construct a removable cover plate for the patient to wear, particularly during meal times.
OBSTRUCTION AND TRAUMA
Papillary obstruction
Occasionally a rough upper molar tooth or an overextended denture flange will irritate the parotid papilla. If this is sufficient to cause ulceration with consequent inflammation and oedema salivary flow might be obstructed, particularly at meal times when the flow rate is increased significantly. In this situation the patient suffers rapid-onset pain and swelling at meal times. Rarely, an aphthous ulcer on the parotid papilla will cause the same symptoms.
If the trauma to the parotid papilla continues there will be progressive scarring and fibrosis in the soft tissues and permanent stenosis of the papilla can occur. Once this is established even removal of the original factor causing the irritation will be unlikely to resolve the problem, and a papillotomy will be required. This is a simple procedure performed under local anaesthesia. A probe is inserted into the orifice of the papilla and with a scalpel blade the papilla is split open by incising down onto the probe. This lays open the papilla and divides the stenosis, allowing free drainage of saliva.
Stone formation (sialolithiasis)
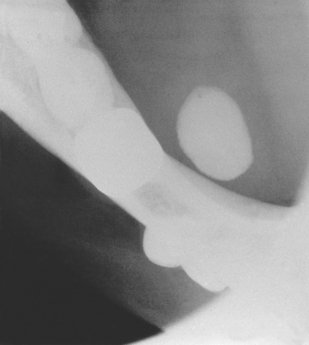
As many as 80% of all salivary stones occur in the submandibular gland or duct (Fig. 14.5), 10% occur in the parotid, 7% in the sublingual gland and the remainder occur in the minor salivary glands. Most stones occur in the submandibular glands because their secretions contain mucus and the viscosity is higher. Of submandibular stones 80% are radio-opaque and can be identified using plain radiographs. Occlusal views are particularly helpful because the image of a small stone must be projected away from other radio-opaque structures such as the teeth and jaws (Fig. 14.6). By contrast the majority of parotid stones are radiolucent and cannot be detected on plain radiography.
The typical presentation of a submandibular calculus is acute pain and swelling at meal times. Onset is rapid, within a minute of starting the meal, and the swelling resolves over a period of about 1 hour, after the meal is completed. However, this classical picture occurs only when the stone causes almost complete obstruction, often when it is impacted at the opening of the submandibular duct. More often the stone causes only partial obstruction and is lying either within the hilum of the gland or within the duct in the floor of the mouth. In this situation the patient may complain of occasional swelling, often with minimal discomfort, or of a chronically enlarged mass in the submandibular triangle with episodes of dull aching pain. This results from chronic bacterial infection, usually due to S. viridans, arising in an obstructed gland with salivary stasis and poor emptying. Often a salivary stone is totally asymptomatic and is discovered coincidentally during radiography for other reasons.
If a stone is identified on plain radiographs, no other investigation is necessary. If the symptoms suggest a diagnosis of a stone, but the stone cannot be seen on routine radiographs, sialography will be needed. However, this procedure does carry the risk of displacing the stone more proximally in the duct system, to a position where it may be difficult to remove. A radiolucent stone will show as a filling defect in the column of contrast medium and the emptying film will show retained contrast medium proximal to the site of the stone.
A submandibular stone often impacts at the papilla or lies in the floor of the mouth, where it is usually palpable. Less commonly, the stone lies partially within the substance of the gland at the hilum where the duct leaves the gland. If the stone is trapped at the duct papilla, it can often be released by gently probing and dilatation of the papilla. It may be necessary to slit the duct in order to release the stone. If the stone is lying in the submandibular duct in the floor of the mouth, anterior to the point at which the duct crosses the lingual nerve (second molar region), the stone can be released by opening the duct longitudinally. It is important to pass a large suture around the duct posterior to the stone so that during the operative procedure the stone cannot be displaced backwards in the duct. Once the calculus has been released, the wall of the duct should be sutured to the mucosa of the floor of the mouth to maintain an opening for the free drainage of saliva. No attempt should be made to repair the duct wall as this will lead to stricture formation. A parotid stone located at the confluence of the collecting ducts can be released surgically by raising a preauricular flap, exposing the parotid duct and incising it longitudinally to release the stone. Salivary stones can sometimes be retrieved endoscopically or under fluoroscopic X-ray control using small wire baskets or angioplasty balloons.
Obstruction in and around the duct wall
Scarring and fibrosis in the duct will result in stricture formation and obstruction to salivary flow. It often presents as a complication of long-standing sialolithiasis but may occur as a result of trauma, particularly to the floor of the mouth. In patients with masseteric hypertrophy the parotid duct may be stretched around the anterior border of the muscle and this may cause obstruction of salivary flow at meal times.
Salivary neoplasms usually develop within the substance of the salivary gland and grow slowly, compressing and displacing the adjacent normal gland tissue. On occasion, the enlarging tumour presses on an adjacent collecting duct and the subsequent obstruction leads to the appearance of a sudden increase in size of the tumour, suggesting malignant change. It is important to recognize that this is not necessarily so.
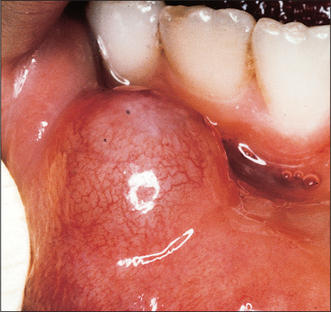
Mucoceles
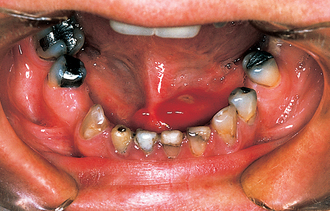
Mucous retention cysts and mucous extravasation cysts arise in the minor salivary glands as a result of mechanical damage to the gland or its duct. The common sites are on the mucosal aspect of the lower lip (Fig. 14.7), particularly in patients with a deep overbite, and in the buccal mucosa posteriorly where an upper wisdom tooth is erupting buccally. Typically the patient presents with a history of recurrent swelling that develops over days or weeks, ruptures and then recurs after a few weeks. The cysts rarely exceed 1 cm in diameter and are tense, bluish, sessile swellings. The treatment is not to the cyst alone but the underlying minor gland, which must also be excised under local anaesthesia.
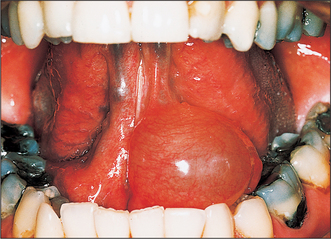
A ranula is a large mucocele arising from the sublingual gland, and typically presents as a large tense bluish swelling in the anterior floor of the mouth, often displacing the tongue (Fig. 14.8). However, the ranula may push its way through the midline mylohyoid dehiscence in the floor of the mouth and enter the submental space presenting as a midline swelling in the upper neck. This is the ‘plunging ranula’. The swelling may be entirely in the neck or the ranula may take on an hourglass shape with both submental and intraoral swelling. The treatment of a ranula must include excision of the sublingual gland.
SALIVARY NEOPLASMS
Salivary neoplasms comprise 1.2% of all neoplastic disease. The WHO classification of salivary gland tumours, published in 1991, categorizes the tumours according to the tissue of origin as being of epithelial or non-epithelial origin. In addition, there is a group of unclassified and allied conditions, which, although not strictly neoplastic, can present as a localized swelling that clinically may mimic a neoplastic condition.
Clinical presentation
Nearly all salivary neoplasms present as slowly growing masses which have often been present for several years (Fig. 14.9). Usually, even malignant salivary tumours grow slowly. Unfortunately, pain is not a reliable indication of malignancy. Certainly if a malignant salivary neoplasm is invading a sensory nerve, pain or paraesthesia can occur. Similarly, if the facial nerve is invaded, paralysis can occur. Frequently at surgery a nerve that has been functioning normally preoperatively is seen to be invaded by tumour. Furthermore, benign tumours often present with pain and aching in the affected gland, presumably due to capsular distension and possibly also due to an element of outflow obstruction. Therefore the only reliable clinical indication of malignancy is facial nerve palsy in the case of the parotid, induration and/or ulceration of the overlying skin or mucosa and regional lymphatic metastasis.
Investigation
For parotid and submandibular tumours, CT scanning or MRI are the most helpful imaging techniques (Fig. 14.10). They will confirm that the mass being investigated is indeed intrinsic to the gland; they accurately image the borders of the tumour and show if it is well circumscribed (and likely to be benign) or diffuse and invasive (and likely to be malignant). In addition, scans show the relationship of the tumour to other anatomic structures and help with the planning of subsequent surgery. Unfortunately, in the case of parotid tumours, these scanning techniques cannot reliably image the facial nerve and therefore reliably determine whether the tumour is superficial or deep to the plane of the nerve. However, this plane can usually be inferred, as the course of the facial nerve is constant from the stylomastoid foramen, becoming more superficial and passing forwards over the masseter muscle.
Conventional sialography has very little to offer in the investigation of neoplastic salivary disease. Apart from demonstrating the presence of a space-occupying lesion, it does not reliably differentiate benign from malignant tumours and is extremely poor at distinguishing between deep lobe and superficial lobe parotid tumours. Ultrasound scanning is a readily available, inexpensive and non-invasive technique that is useful in imaging the parotid. The two most common tumours have very characteristic echo patterns and often an accurate diagnosis can be suggested. Isotope imaging with technetium pertechnetate gives inconsistent results.
Open surgical biopsy of intrinsic neoplasms of the major glands is absolutely contraindicated. At least 75% of all parotid tumours and more than 50% of all submandibular gland tumours will prove to be benign pleomorphic adenomas. This tumour, which is only poorly encapsulated, is very tense and if an incision is made into it the contents escape into the surrounding tissue planes—and it is almost impossible to eradicate the microscopic spillage of tumour cells. If this happens the patient will be at risk of developing multiple local tumour recurrences over many years. However, if there is overt skin infiltration or ulceration an open biopsy is essential to establish a preoperative diagnosis upon which to plan surgery. For tumours of the minor salivary glands, particularly in the palate, there is a much higher chance of the tumour being malignant and, as it is not necessary to open up other tissue planes to gain access to the tumour, open incisional biopsy is important.
Fine-needle aspiration (see Ch. 8) is a safe alternative to open biopsy of a major gland. Evidence suggests that provided the needle gauge does not exceed 18 G there is no risk of seeding viable tumour cells. Although advocates of this technique claim high accuracy and specificity there is inevitably a high risk of sampling error. Many salivary neoplasms are not homogeneous and, following excision, the entire tumour mass often must be sectioned to find the one critical diagnostic area of the mass.
EPITHELIAL TUMOURS
Of all salivary epithelial tumours 75% arise in the parotid glands and of these only 15% are malignant; just over 10% occur in the submandibular glands and approximately one-third are malignant; about 15% of tumours occur in the minor salivary glands and nearly half of these will be malignant. Tumours arising in the sublingual glands are very rare (0.3%) but nearly all will be malignant.
Both benign tumours (adenomas) and malignant tumours (carcinomas) occur.
Clinical features of salivary tumours (WHO classification of 1991, simplified)
Adenomas
Of the variety of benign adenomas that have been described only two, the pleomorphic adenoma and Warthin tumour, arise with any frequency.
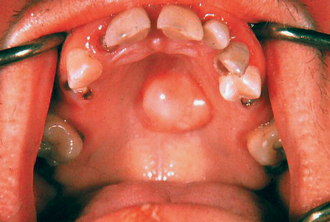
The pleomorphic adenoma occurs at any age (mean 42 years) and has an equal sex incidence. It accounts for at least 75% of parotid tumours and more than 50% of submandibular tumours but for rather less than 50% of minor gland tumours. Clinically the tumour has the texture of cartilage and has an irregular and bosselated surface. In the palate, the overlying mucosa is rarely ulcerated. Rarely, after a number of years the tumour may undergo malignant change, so all patients presenting with pleomorphic adenomas should be advised to undergo surgical removal of the tumour.
The Warthin tumour occurs only in the parotid gland, where it accounts for approximately 15% of all neoplasms. It is a disease of the elderly, with a mean age of presentation of 60 years. Historically it had a male:female ratio of 4:1 but is now becoming increasingly common in women. Recent evidence suggests that this tumour is related to cigarette smoking. It is also unusual in that in 10% of cases it arises either bilaterally in the parotids or is multi-centric in the one gland. It does not undergo malignant change.
Carcinomas
The acinic cell carcinoma and the mucoepidermoid carcinoma, although undoubtedly malignant tumours with a potential for local invasion and metastatic spread, are frequently very low grade histologically and do not require the radical treatment needed for more aggressive tumours. Together they account for only 5% of all tumours at any site.
The adenoid cystic carcinoma, adenocarcinoma, squamous cell carcinoma and undifferentiated carcinoma are all aggressive malignant tumours that carry a poor prognosis regardless of treatment. The adenoid cystic carcinoma is characterized by inevitably killing the patient, by relentless perineural spread along the cranial nerves and into the brain. However, it grows extremely slowly and, although invariably fatal, the 5- and 10-year survival figures are 70% and 40%, respectively. It is also unusual in having a predilection for distant metastasis to the lungs, where it often produces multiple ‘cannon ball’ tumours, which can remain symptomless for many years. The other carcinomas mentioned above have 5-year survival figures of around 35–40%. Carcinoma arising in a pleomorphic adenoma carries a very poor prognosis, with a 5-year survival rate of only 25%.
Management of epithelial tumours
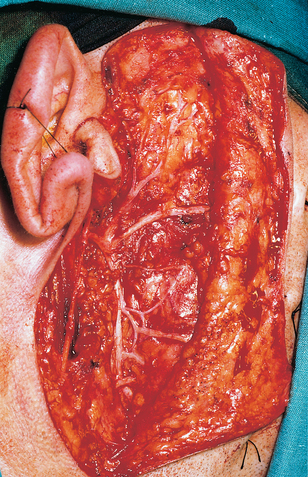
Both benign and malignant tumours arising in the parotid or submandibular glands are treated surgically, by excision with wide margins. In the parotidgland, excision is by either superficial or total parotidectomy, according to the location of the tumour. Unless the patient presents with facial nerve palsy (indicating a malignant tumour) the facial nerve should be preserved (Fig. 14.11). In the submandibular gland treatment is by excision of the gland. If the definitive pathological diagnosis is of a high-grade malignancy, then the patient should receive radical postoperative radiotherapy. In those cases when the tumour involves skin or other adjacent structures, or where there is lymphatic metastasis, the patient should undergo radical excision, including a neck dissection and sacrificing any structures invaded by tumour, and treated with postoperative radiotherapy.
Pleomorphic adenomas arising in the minor salivary glands can be treated by local excision with a 5-mm margin. They do not invade periosteum and so in the hard palate they should be excised subperiosteally without removing any underlying bone. Mucoepidermoid carcinomas and acinic cell carcinomas require rather more radical excision, with a 10-mm margin, and when they are situated in the palate palatal fenestration should be undertaken. Postoperative radiotherapy is indicated only for high-grade tumours or if the margins are not clear. For the remaining carcinomas arising in the minor salivary glands, radical surgical excision and postoperative radiotherapy is indicated. In the palate this will be by maxillectomy.
Complications of parotidectomy
In experienced hands permanent damage to the facial nerve should not occur in conservative parotidectomy when the facial nerve is being preserved. However, various degrees of transient weakness, lasting typically 6 weeks, are common, occurring in approximately 33% of cases. This is due to demyelination as a result of handling the nerve and ischaemia in the nerve following surgery. This demyelination reverses spontaneously. Surprisingly, postoperative salivary fistula formation is uncommon. More common is the formation of recurrent collections of saliva under the flap (sialoceles); although these are often aspirated to reassure the patient, they recur initially for a period of a few weeks and then resolve without active intervention.
Gustatory sweating (Frey’s syndrome) is a complication that occurs in at least a third of patients following parotid surgery. Symptoms first develop 12–18 months following surgery and the patient complains of redness of the overlying skin and sweating of the skin at mealtimes. Usually the symptoms are mild but the sweating can be so profuse that at mealtimes the patient must drape a towel over the shoulder on the affected side to absorb the perspiration. It is claimed that the condition occurs as a result of the regeneration of secretomotor parasympathetic nerve fibres along the myelin sheaths of the sympathetic nerve fibres of the sweat glands in the overlying skin. The condition is difficult to treat surgically, but subcutaneous infiltration of botulinum toxin into the affected area controls the symptoms very well.
NON-EPITHELIAL TUMOURS
A variety of non-epithelial tumours arise in the salivary glands. Haemangiomas and lymphangiomas (cystic hygromas) occur in childhood. Haemangiomas occur mostly in the parotid, appearing shortly after birth and growing progressively for several months. The majority undergo spontaneous regression by 2 years of age. Females are more frequently affected than males. Lymphangiomas are less common. They may affect any of the salivary glands, and form spongelike multicystic lesions: 50% are manifest by 12 months and 90% will be evident by the end of the second year. Lymphangiomas do not undergo spontaneous involution. They frequently extend into the neck and mediastinum and can undergo dramatically rapid growth, causing respiratory obstruction. Treatment is by complete surgical excision but this may be technically very difficult.
Neurofibromas and neurilemmomas are the most common non-epithelial tumours arising in adults. Clinically they are not distinguishable from other salivary tumours and are diagnosed only following surgery for a presumed epithelial tumour. Lipomas occur only in the parotids, particularly in adult males. They are treated by surgical excision.
MALIGNANT LYMPHOMAS
True extranodal lymphoma arising in the salivary glands, usually the parotids, is rare. It occurs as a manifestation of a mucosa-associated lymph tissue (MALT) lymphoma. More common is lymphoma arising from the lymph nodes, either on the surface of the glands or within the parenchyma of the gland. Lymphoma also arises in the salivary glands as a complication of HIV disease, in the benign lymphoepithelial lesion, and Sjögren’s syndrome (see below). The peak incidence for non-Hodgkin’s lymphoma is the sixth and seventh decades and women are twice as likely to be affected, largely due to lymphomatous change in Sjögren’s syndrome, which is more common in women. By contrast Hodgkin’s disease arises in the juxta-glandular nodes rather than within the salivary gland parenchyma. Its peak incidence is the third and fourth decades and males predominate in the radio of 4:1.
Salivary gland lymphomas usually present as firm painless swellings and more than 90% occur in the parotids. If the lymphoma is confined to the parotid, treatment is by parotidectomy with postoperative radiotherapy. If there is evidence of spread beyond the salivary gland, treatment is by polychemotherapy according to the accepted protocols, based on histo-logical characterization.
UNCLASSIFIED AND ALLIED CONDITIONS
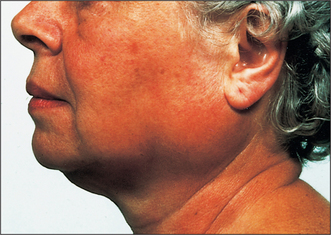
Sialosis is an uncommon, non-inflammatory cause of salivary swelling, usually affecting the parotid glands symmetrically. It is usually associated with conditions such as alcohol abuse, diabetes mellitus, pregnancy, malnutrition and some drugs (usually sympathomimetics). It typically affects middle-aged and elderly patients, who present with bilateral soft parotid swellings which are usually painless. Biopsy of the glands reveals extensive fatty replacement but otherwise normal tissues. No treatment is known to be effective but sometimes parotidectomy is required to correct the disfigurement.
Necrotizing sialometaplasia, benign lymphoepithelial lesion (see later), salivary duct cysts, Kuttner tumour and cystic lymphoid hyperplasia of HIV disease can all mimic salivary gland neoplasia. Similarly, branchial cysts and dermoids can cause diagnostic confusion on occasion.
DEGENERATIVE CONDITIONS
Sjögren’s syndrome
Sjögren’s syndrome is an autoimmune condition causing progressive destruction of the salivary and lachrymal glands. In 1933 Sjögren, a Swedish ophthalmologist, first described the association of keratoconjunctivitis sicca (dry eyes) and xerostomia (dry mouth). Shortly thereafter he noted that these symptoms frequently occurred in patients with rheumatoid arthritis. It has since been realized that Sjögren’s syndrome can occur in association with any connective tissue disorder: indeed, the association is more common in many connective tissue disorders than it is with rheumatoid arthritis. Only 15% of patients with rheumatoid arthritis develop Sjögren’s syndrome, whereas 30% of patients with systemic lupus erythematosis and nearly all patients with primary biliary cirrhosis do so. This combination of dry eyes, dry mouth and a connective tissue disorder (most often rheumatoid arthritis as this is by far the most common connective tissue disorder) is called secondary Sjögren’s syndrome. The same combination of dry eyes and dry mouth but without associated connective tissue disorder is known as primary Sjögren’s syndrome. Primary Sjögren’s syndrome also differs from secondary Sjögren’s syndrome in that xerostomia and xerophthalmia are more severe, dysfunction of other exocrine glands is more widespread, incidence of lymphoma is higher and the autoantibody profile is different.
Females are affected more often than males in the ratio of 10:1. Typically patients are middle-aged. The presenting complaint is usually of the underlying connective tissue disorder and only later does the patient become aware of a gritty feeling in the eyes (due to conjunctivitis in the dry eyes) or a dry mouth. In the primary disorder, the complaint will be related to reduced lachrymal or salivary flow. Occasionally there is enlargement of the parotid glands bilaterally and even more rarely the enlarged parotids are painful. Although total xerostomia causes the mucosa to become parchment-like and is obvious, lesser degrees of xerostomia may cause problems for the patient but may not be obvious on clinical examination. Superinfection with Candida albicans is common. Less frequently, the patient develops bacterial sialadenitis due to ascending infection from the mouth. The condition does not invariably progress to total xerostomia and for any individual patient it is not possible to predict the outcome. The characteristic feature of the condition is progressive lymphocytic infiltration, acinar destruction and proliferation of duct epithelium of all salivary and lachrymal tissue.
The diagnosis is often based on the characteristic history: no laboratory investigation is pathognomonic of either primary or secondary Sjögren’s syndrome. However, the following investigations are usually undertaken:

Fig. 14.12 Parotid sialogram showing gross destruction of parotid parenchyma in a patient with advanced Sjögren’s syndrome.
The management of Sjögren’s syndrome must be symptomatic. No known treatment modifies or reverses the xerostomia and keratoconjunctivitis sicca. Artificial tears are essential to preserve the cornea. For the dry mouth various artificial saliva preparations are available (e.g. Saliva Orthana, Glandosane, Luborant) but often the patient prefers to use frequent drinks and learns to carry a bottle of water with them at all times. If a dentate patient is to use saliva substitutes the product should not have a low pH and should contain fluoride because rampant dental caries is a frequent complication. Other complications are recurrent infections in the mouth, ascending parotitis and those relating to any underlying connective tissue disorder. There is also increased incidence of developing MALT lymphoma (most commonly monocytoid B-cell lymphoma) in patients with Sjögren’s syndrome. The risk is highest in those with primary Sjögren’s syndrome. The onset of lymphoma is often heralded by immunological changes (falling immunoglobulin levels, falling titre of rheumatoid factor, rising B2-microglobulin titre, rising serum macroglobulin titre and the appearance of monoclonal light chains in the serum and urine), lymphadenopathy and weight loss.
‘Benign’ lymphoepithelial lesion
Use of the word ‘benign’ to describe this lesion is misleading because approximately 20% of patients with benign lymphoepithelial lesion or Sjögren’s syndrome ultimately develop MALT lymphoma.
Histologically it is not possible to distinguish benign lymphoepithelial lesion from Sjögren’s syndrome. Both are characterized by lymphocytic infiltration, acinar atrophy and ductal epithelial proliferation. Indeed, they may well be manifestations of the same condition.
Clinically benign lymphoepithelial lesion presents as diffuse swelling of the parotid gland. The swelling is firm and often painful and in 20% of cases is bilateral. Most patients are female (80%) and most are over 50 years old at presentation. Parotidectomy is mostly undertaken in order to establish the diagnosis, but if any parotid remnants are left, the swelling may recur with the risk of lymphomatous change. Prolonged follow-up is essential.
Mikulicz syndrome
In 1888 Mikulicz described benign, asymptomatic, symmetrical enlargement of the lachrymal and salivary glands. His original publication presented a series of patients who clearly had a variety of different conditions: benign lymphoepithelial lesion, Sjögren’s syndrome, lymphoma, lymphocytic leukaemia, sarcoid and sialosis can all present in this way. The term Mikulicz syndrome is thus not helpful and should not be used.
Xerostomia
A complaint of dry mouth is common in oral surgery and oral medicine clinics. It seems to be particularly frequent in postmenopausal women, who also complain of a burning tongue or mouth. Normal salivary flow decreases with age in both men and women, although of course postmenopausal women are the group most likely to develop Sjögren’s syndrome.
The situation is further confused as patients with Sjögren’s syndrome are frequently unaware of having a dry mouth and patients who complain of dry mouth frequently have normal salivary flow rates. The most common causes of xerostomia, in order of frequency, are:
Xerostomia can be difficult to treat. Treatment is aimed at relief of symptoms and avoidance or control of complications. Artificial salivas are not well accepted but their lubricant properties may be particularly useful at mealtimes. Cholinergic drugs such as pilocarpine can be used but their adverse effects (diarrhoea and pupillary dilatation) often outweigh any benefit. Furthermore there must be some remaining functional salivary tissue for such drugs to have any effect.
Rampant caries and destructive periodontal disease due to oral infection are major complications. Meticulous oral hygiene and the weekly use of topical fluoride is essential. There is a high incidence of oral candidosis and antifungal drugs are necessary. Conventional tablets and lozenges to be sucked are not well tolerated as the mouth is too dry to dissolve the drug: miconazole gel or ketoconazole suspension are better tolerated. Ascending parotitis is treated with antibiotics as required. Sometimes it is necessary to perform a parotidectomy and tie off the parotid duct to prevent further episodes of infection.
Sialorrhoea
Some drugs and painful lesions in the mouth increase salivary flow rates. In normal health this is rarely noticed as the excess saliva is swallowed spontaneously. ‘False ptyalism’ is more common and is a well-recognized delusional symptom or occurs due to faulty neuromuscular control leading to drooling despite normal saliva production. Uncontrollable drooling is usually treated surgically. As the submandibular gland contributes most resting saliva attention is directed at these glands bilaterally. The submandibular ducts can be mobilized and repositioned in the base of the anterior pillars of the fauces. Alternatively, the two glands may be excised.
Cawson R.A., Gleeson M.J., Eveson J.W. Pathology and surgery of the salivary glands. Oxford, UK: ISIS, 1997.
Langdon J.D. Sublingual and submandibular gland excisio. In: Langdon J.D., Patel M.F., editors. Operative maxillofacial surgery. London: Chapman & Hall; 1998:375-380.
Langdon J.D. Parotid surgery. In: Langdon J.D., Patel M.F., editors. Operative maxillofacial surgery. London: Chapman & Hall; 1998:381-390.
Langdon J.D. Salivary gland disorders. In: Russell R.C.G., Williams N.S., Bulstrode C.J.K., editors. Bailey & Love’s short practice of surgery. 23rd edn. London: Arnold; 2000:651-668. Ch 42
Norman J.E. DeB, McGurk M. Colour atlas and text of the salivary glands. London: Mosby-Wolfe, 1995.
SELF-ASSESSMENT
Answers on page 268.