8 Oral lesions: differential diagnosis and biopsy techniques
ASSUMED KNOWLEDGE
It is assumed that at this stage you will have knowledge/competencies in the following areas:
If you feel you are not competent in these areas, revise them before reading this chapter or cross-check with relevant texts as you read.
INTENDED LEARNING OUTCOMES
At the end of this chapter you should be able to:
OVERVIEW
Many oral lesions are first noticed by the patient or clinician as changes in the colour, texture or disruption of the mucosal surface, or as a swelling or asymmetry of the orofacial structures. When involving or arising from the underlying bone, these lesions may produce a variety of radiological changes, including radiolucencies with or without opacities, evidence of expansion and bone loss in the form of erosion or resorption.
Securing the diagnosis is fundamental to safe and successful management of the patient and will depend on a clear and concise history, careful clinical examination and the use of additional special investigations including radiography, biopsy and other laboratory and diagnostic techniques. Within this spectrum, biopsy is a commonly performed test and is detailed later in this chapter. Should the lesion appear malignant, biopsy should not be performed in general practice, but the patient should be referred immediately for a specialist opinion (see Ch. 10). This approach avoids the need for the general practitioner to break unexpected/distressing news with the consequent requirement to explore the management options with a now anxious patient.
All referrals require clear and concise details, but where malignancy is suspected, direct contact should be made with the specialist by telephone or fax.
SOFT-TISSUE LESIONS
Diagnosis
Soft-tissue oral lesions are common, often symptomless, slow-growing lumps and are first noticed by the clinician at a routine examination so the clinical history might not contribute greatly to making the diagnosis. Pain is unusual and would indicate infection in a lesion such as a cyst (secondary infection, see Ch. 9). On rare occasions it may indicate aggressive behaviour, e.g. malignant tumour of minor salivary gland (Ch. 14).
Clinical examination, however, based on inspection and palpation referenced to site, colour and consistency provide the main pointers to diagnosis.
Site
The posterior palate, lateral third of hard palate and upper lip are particularly high-risk sites for minor salivary gland tumours. The lower lip is a common site for mucoceles, whilst the buccal mucosa is a common site for fibroepithelial polyps particularly when associated with missing teeth.
Colour
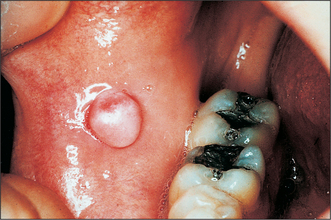
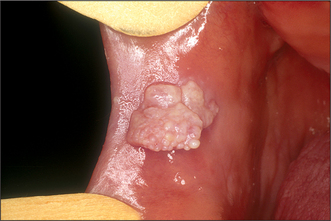
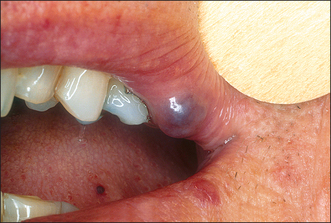
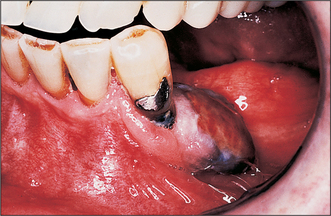
Colour will distinguish between fibroepithelial polyps (pink) (Fig. 8.1) and the white rough cauliflower presentation of viral papillomas (warts) (Fig. 8.2). Mucous cysts tend to be translucent with a bluish colour (see Figs 14.7, 14.8) and haemangiomas and ‘venous lakes’ (Fig. 8.3) dark blue. Pyogenic granulomas (Fig. 8.4) and giant-cell epulides (Fig. 8.5) normally present as maroon/red.
Anatomical relationships and consistency
Lesions may also be distinguished by their depth within the oral tissues and their relationship to adjacent structures. This will differentiate superficial lesions such as fibroepithelial polyps, viral papillomas and epulides (gingival swellings) from submucosal lesions. Palpation is essential to define the lesion’s consistency and distinguish between discrete and diffuse swellings, or whether the lesion is mobile or attached to deeper structures. Solid submucosal lesions can be reactive (e.g. lymph nodes) or neoplastic arising from any of the submucosal structures (e.g. adenomata of minor salivary glands and neurilemmomas). All submucosal swellings that are solid have the potential to be neoplastic and must be investigated.
Modifying influences
The intraoral environment having given rise to lesions may in turn alter their morphology, depending on site. For example, fibroepithelial polyps can vary in presentation from ‘leaf fibromas’ in the palate to denture-induced hyperplasia at the periphery of a denture (see Figs 11.5, 11.6). Immature or developing polyps, particularly when arising from the gingivae, are described as pyogenic granulomas and the pregnancy epulis is a variant. They present as red, acutely inflamed lesions that are soft and bleed readily. Similar in presentation is the peripheral giant-cell granuloma although this may be more purple.
Single and multiple lesions
Almost all the lesions described so far present as single and discrete swellings. The presence of multiple lesions is of significance and indicates systemic disorders. Examples include the mucosal tags and polyps in oral Crohn’s disease/orofacial granulomatosis and multiple viral papillomas of HIV infection.
White/red and pigmented lesions
White and red patches are discussed in detail in Chapter 10 (see Figs 10.1, 10.2), but it should be noted that a wide array of white patches that have no significant malignant potential can occur. The difficulty is that interpretation of this risk almost always requires a histological diagnosis. Biopsy is therefore mandatory for all unexplained lesions and the decision not to biopsy requires clinical experience in the management of these types of lesions.
A variety of pigmented lesions may present within the oral cavity; they may be blue, brown or black in colour, flat (macule), raised or granular (papule) and single or multiple. Common single macular lesions include amalgam tattoos and benign naevi. Multiple macular lesions include racial deposits and lesions secondary to chronic inflammatory damage to the epithelium such as lichen planus and tobacco-induced melanosis. However, in common with white/red lesions, biopsy may be indicated to confirm the diagnosis. It is essential that, as with leukoplakia, any recent history of ulceration, haemorrhage or change in the type (becomes papular) or size of a lesion is to be regarded as serious when urgent referral is mandatory.
Indication for biopsy
Where the diagnosis is clinically evident as in the case of fibroepithelial polyps, viral papillomas or mucous cysts, biopsy is not required for diagnosis although where treatment involves excision, the specimen should always be submitted for histological confirmation. If there is doubt or the lesion is arising deep to the mucosa then histological diagnosis is essential.
TREATMENT OF SOFT-TISSUE LESIONS
General points
The primary management objective is to establish the diagnosis. This may require nothing other than clinical examination as for a squamous papilloma or mucous cyst, but often and particularly for solid/firm submucosal lesions does require biopsy. Subsequent treatment will depend not only on the clinical significance of the diagnosis but on the presence of related symptoms and the site and size of the lesion.
Lesions causing problems, or which can be anticipated to do so, will require surgery or referral. Small, innocent lesions can be left, assuming they are not causing symptoms, increasing in size or of concern to the patient in terms of function or appearance.
In general, the size and site of the lesion do not change the technique but do require careful assessment of the patient and the level of experience of the operator. Larger lesions (greater than 2 cm), particularly submucosal ones, are more difficult to manage surgically. This is due to the access required and the risks to adjacent blood vessels and nerves. In addition they will be more difficult to repair and consequently they should not be attempted unless the operator is skilled in soft-tissue surgery and in dealing with the intraoperative and postoperative complications.
Fibroepithelial polyps, squamous papillomas and epulides
Surgical excision is the most common treatment for small lesions (fibroepithelial polyps, squamous papillomas and epulides) and is often completed as part of the biopsy (see below). It must, however, be combined with appropriate management of precipitating or causative factors. These include the elimination of chronic trauma for polyps or the removal of localized deposits of calculus for pyogenic granulomas. Peripheral giant-cell lesions will require a similar approach including curettage of the underlying bone. Epulides related to pregnancy are, unless they are large and causing distress, best left alone and removed if they persist postpartum, usually with an appropriate adjunctive periodontal therapy. Scaling and improved oral hygiene during pregnancy may reduce further growth and lessen the likelihood of surgery.
Surgical management of denture-induced hyperplasia must be combined with appropriate modification to the prosthesis such as temporary relining (Ch. 11). This is essential to retain sulcus form and prevent recurrence and is similarly so for ‘leaf-fibroma’ in the palate. Denture-induced hyperplasia can be extensive and, when affecting the lower jaw, is often related to other significant anatomical structures. This is particularly so in the anterior region where the main trunk of the mental nerve can be superficial due to resorption of the alveolus. The lingual nerve can also be at risk when the lingual side is operated on. In these circumstances it is important to carefully identify the edge of the hyperplastic tissue, to keep the excision superficial and to use blunt dissection to free the tissue from underlying structures (see section on biopsy).
Mucous cysts
Mucous cysts divide between the common extravasation cyst and the rarer retention cyst and present as tense bluish swellings often in the lower lip (see Fig. 14.7). These conditions are discussed in Chapter 14.
Vascular lesions
Vascular lesions are best considered as two groups: the rare haemangiomas, which can be extensive and multiple, and the common smaller degenerative malformations (varicosities) often seen on the lip in older patients (Fig. 8.3). For the former it is essential that the type, site and extent are fully evaluated, which normally requires referral to a specialist centre. For extensive and deep lesions or where there is any possibility that the haemangioma extends into bone, extractions or other surgery must be avoided until the type and extent of the vascular abnormality have been established. Conversely, discrete small lesions are easily dealt with either by excision with appropriate management of the vascular source or by cryotherapy. Indications for surgery are cosmetic or lesions that are repeatedly traumatized and bleed.
Submucosal solid lesions
All submucosal solid lesions require investigation as the probability of them being neoplastic is high. Treatment selection is very much a balance between incisional biopsy to determine the histological diagnosis first and removal of the lesion in its entirety to avoid compromise of the site in terms of further surgery by potential seeding of tumour cells (e.g. pleomorphic salivary adenoma). This is often decided by the site and size of the lesion along with the results of specialist imaging such as CT or MRI scanning. For small lesions the careful combination of sharp and blunt dissection as described below should only be undertaken by clinicians skilled in soft-tissue surgery. As stated below, the excision of lesions arising under mucoperiosteum such as the palate requires the defect to be closed either by dressing the site and allowing repair by secondary intention or by primary repair with a mucosal flap or graft. Surgery in the soft palate area can be difficult, particularly if the lesion is deep where there is a possibility of perforation through to the nasal side.
DIAGNOSIS AND TREATMENT OF HARD-TISSUE LESIONS
Overview
Hard-tissue lesions can arise from odontogenic tissue or from bone. They present a range of diagnoses from developmental lesions such as palatal or mandibular tori or odontomas (see Figs 8.6, 11.13, 11.14) and bony exostoses, to the rarer benign and locally invasive lesions such as ossifying fibroma, ameloblastoma and cementoblastoma.
This is a complex group of lesions and conditions. Readers are referred to relevant pathology and radiology texts for more detailed descriptions of the various types and their histological and radiological findings. From the clinical standpoint they can be considered according to presentation: the common single discrete lesions (odontomes, odontogenic tumours and osteomata); the rare, large and diffuse lesions (fibrous dysplasia and Paget’s disease); and rare conditions with multiple lesions (Gardner’s syndrome and cemento-ossifying lesions).
Often they first come to attention as a bone-hard swelling beneath the overlying mucosa, but in many cases presentation is an incidental finding on routine radiography. As the symptoms and signs have so much overlap, diagnosis is largely dependent on radiological findings and, more importantly, histology. As described below, site can be an important sign in establishing the diagnosis: not only are the lesions within this group difficult to separate, they are often clinically and radiographically similar to cysts (see Ch. 9). As with soft-tissue lesions, there will be malignant counterparts, e.g. osteosarcoma and secondary tumours, that can present in similar fashion (see below).
Odontogenic lesions
These arise as abnormalities of tooth-forming structures and form a group ranging from odontomes (which are hamartomas, i.e. developmental lumps, with progression limited to the normal growth period) to benign and locally invasive tumours. They can present as discrete single lesions and are often extensive. Radiographically they can be radiolucent with the characteristics of cysts (Ch. 9) or be radio-opaque or of mixed radiodensity.
Odontomes
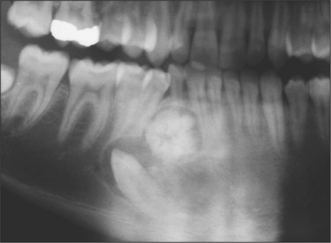
These hamartomas (see pathology texts for subclassification) do not continue to grow. They commonly present as a chance radiographic finding, related to delayed eruption of teeth (Fig. 8.6), or as a palpable mass in the alveolar region. Occasionally they cause pain if infected. Radiographs show them to be well-circumscribed, radio-opaque lesions often with a radiolucent margin akin to the periodontal ligament of a tooth.
Clinically they can be considered as malformed supernumerary teeth and treatment should reflect this, with removal recommended if they interfere with tooth eruption or orthodontic tooth movement, or are infected. In the absence of such problems, particularly if there are risks to adjacent structures or the patient has significant co-morbidity, they may be left alone.
Benign and locally invasive odontogenic tumours
Most odontogenic tumours are essentially benign and, depending on the cellular bias, can be composed entirely of soft tissue presenting as cysts or produce, to varying degrees, radio-opaque, odontogenic hard tissue. They have similar presenting symptoms and signs to odontomata but as they are tumours they will continue to grow and present a significant challenge in terms of management. This stems from their similar presenting symptoms and signs weighed against a spectrum of clinical behaviour. The potential for local recurrence is high and in some cases such as ameloblastomas, there is a very small but significant potential for metastatic spread. Very few are suited to simple enucleation. Conservative excision (removal with aggressive curettage of the bony margins) or formal resection with a margin of sound bone are the treatment options and must be referenced to the risks of local recurrence or spread and the size and anatomical relationships of the lesion. With the extensive overlap in presenting symptoms and clinical and radiographic signs between cysts and tumours it is essential that the histological diagnosis be established prior to definitive surgery. This is particularly so with large lesions and would take the form of an exploratory procedure with an incisional biopsy.
Ameloblastomas
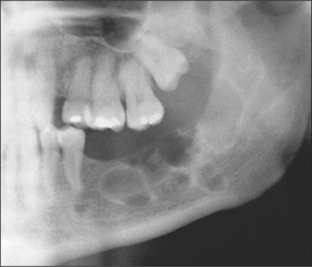
These are the most common of the odontogenic tumours with many, including malignant, variants (see pathology texts for subclassification). They almost always present as a radiolucency (Fig. 8.7), often at the angle of the mandible and can, in some instances, be confused with the odontogenic keratocyst or dentigerous cyst. When they are small they can be unilocular but large lesions usually take on a multilocular appearance with a less well-defined border. Resorption of cortical bone and roots of adjacent teeth are common findings. Interestingly, despite the moderately aggressive growth pattern of these tumours, they rarely cause damage to the neurovascular bundle, in the mandible. They tend to displace it as they enlarge, so that numbness of mental nerve distribution is a rare clinical feature compared with osteogenic malignancy or intraosseous secondary malignancies.
The most appropriate treatment is to resect the tumour with a margin of surrounding bone. For the ameloblastic fibroma conservative excision and for the unicystic ameloblastoma, enucleation alone may provide adequate treatment, but as with all this group, radiographic follow-up should be maintained for an indefinite period.
Odontogenic myxoma
This rare tumour can present with radiographic signs similar to ameloblastoma: sometimes it may display a ‘soap bubble’ appearance. It is benign but tends to extensively infiltrate the surrounding bone. Excision with extensive curettage may prevent recurrence but resection with a margin of bone may be required.
Adenomatoid odontogenic tumour and odontogenic fibroma
These uncommon tumours can be associated with unerupted teeth and may have tiny calcified areas. They are benign with a low potential for recurrence. Careful enucleation is usually curative.
Calcifying epithelial odontogenic tumour
This is a rare tumour usually presenting as a radiolucent area in the molar region of the mandible, but as its name suggests will have a radio-opaque element. In common with many other odontogenic tumours it has a malignant counterpart. It is locally invasive and resection with a margin of bone is recommended.
Cementifying lesions
Cementoblastomas are rare tumours and densely radio-opaque (not always homogeneous), and show a radiolucent rim in continuity with the periodontal space. They are attached to roots of vital teeth that may be resorbed and almost always occur in the first molar, or premolar region of the mandible. In contrast to most other odontogenic tumours they may present with pain.
They are benign and if the offending tooth is extracted and the lesion completely resected there should be no recurrence.
Cemento-osseous dysplasias form another group and as their name suggests are mixed and not neoplastic. They occur both as single and multiple lesions with a rare familial variant. It is important to note that they relate to apices of vital teeth and in early stages are radiolucent, mineralizing gradually as they mature. It is important therefore not to confuse them with dental cysts and apical granulomas.
Treatment of cemento-osseous dysplasia can be difficult and to be curative needs to be more extensive than would normally be associated with a reactive lesion. Indeed infection following attempts at surgical extraction may supervene, which can result in sequestration. Regular review is normally all that is required.
Osseous lesions
In common with the odontogenic lesions there is a wide variety of bony lesions ranging from discrete simple exostoses including tori (hamartomas) to rare benign growths, such as ossifying fibroma, to malignancies.
With the exception of tori, they are all considerably less common than odontogenic lesions but can present with the same signs including swellings of the jaw or displaced teeth. Radiographic findings are also similar with lesions presenting as radiolucencies, opacities or mixed appearance. Some, such as ossifying fibroma, have reasonably well-defined and corticated margins; others such as the giant-cell lesions are well defined but not corticated. Resorption of roots of adjacent teeth is associated with aggressive growth behaviour, but can occur with benign lesions and with giant cell lesions. Radiological features will be a major factor in making the diagnosis, but for most histological confirmation is necessary.
Also of note are discrete areas of dense bone within the cancellous architecture that are almost always a chance radiological finding. These are sometimes called enostoses or solitary bone islands and are most common in the mandible, though not related to the roots of teeth and do not exhibit a radiolucent rim. They are sometimes considered as the bone’s response to low-grade infection but perhaps better seen as a variant of normal and therefore should be left alone. The larger and more diffuse conditions of Paget’s disease, fibrous dysplasia and cherubism are rare and require specialist investigation and management (see below).
There are other intrabony lesions such as acute and chronic osteomyelitis, primary (osteogenic sarcoma) and secondary malignancies (myeloma, lymphoma and metastatic deposits from breast, bronchus, kidney, prostate or thyroid) and Langerhans cell histiocytosis, which can all present within the described spectrum of clinical and radiographic signs. It is important to be able to recognize the symptoms and signs that would warrant their inclusion in a differential diagnosis (see below).
The overlap of clinical and radiological signs makes diagnosis of this varied, complex and potentially dangerous group of lesions difficult. It is dependent on histology and sometimes other special investigations such as bone chemistry and specialist imaging such as CT and bone scans. In view of this and the often difficult decisions on treatment, and need for long-term follow-up, it is important to refer patients with these types of lesion.
Tori and exostoses
Tori and exostoses are common and non-neoplastic. They can occur singly or as multiple lesions in the midline of the palate or on the lingual aspects of the lower premolar region. In many situations they do not require any active treatment unless they are repeatedly traumatized or cause interference with the fit of dentures (see Ch. 11).
Osteomata and other benign tumours
Osteomata are benign tumours and as such will be progressive, usually as peripheral growths and should be excised. If multiple they may be a feature of Gardner’s syndrome. They can present centrally with a radiographic appearance similar to that of solitary bone islands or even cementifying lesions. A rare variant is the osteoblastoma, a lesion of variable radiodensity that, like the cementoblastoma, can be painful and is also best excised. Larger lesions such as the ossifying fibroma can be treated with local excision and curettage but require long-term follow-up as recurrence is relatively common.
Giant-cell lesions
This group of lesions extend from what are considered non-neoplastic lesions such as the peripheral giant-cell granuloma (see above) and its central counterpart, to systemic lesions associated with hyperparathyroidism to rare tumours both benign and malignant.
The central giant-cell granuloma is a radiolucent soft-tissue mass arising within bone and notably has a well-defined margin that is not corticated. This derives from its osteoclastic origin, which in turn can result in resorption of adjacent teeth. It is indistinguishable histologically from the central bony masses of hyperparathyroidism. For that reason, if a histopathology report suggests a giant-cell lesion then plasma calcium, phosphate and parathormone levels should be checked to exclude hyperparathyroidism. Otherwise these are treated by local excision and curettage and this is usually successful. The true giant-cell tumour of bone is rare in the jaws; it requires a wide resection and reconstruction.
Cherubism
Cherubism is an inherited (autosomal dominant) disorder characterized by the development of bilateral symmetrical facial swellings. The swellings are composed mainly of fibrous tissue and giant cells. Cherubism is characteristically more common in males and presents in early childhood. Radiological findings are similar to those of multilocular cysts. Treatment is conservative as the lesions usually resolve as the bony tissues mature.
Fibrous dysplasia
Fibrous dysplasia, as its name suggests, is a non-neoplastic condition where bone is replaced by fibro-osseous tissue. It is of unknown aetiology but recent studies indicate a link with activating mutations affecting the alpha subunit of the g-protein GNAS1. The association is strongest with McCune-Albright syndrome but also is detected with polyostotic and monostotic fibrous dysplasia.
Fibrous dysplasia usually presents in young adults as a unilateral painless progressive swelling often affecting the maxilla. It can become disfiguring and may displace teeth and on occasion cause malocclusion. Both clinically and radiographically the margins are poorly defined and maxillary lesions can invade and obliterate the air sinus. Radiographic findings are slightly granular, medium radiodensity with loss of trabecular pattern (‘ground glass’ or ‘orange peel’ appearance) that blends into the surrounding bone. Although growth can be rapid, it is usual to defer treatment until bodily growth has ceased. At this stage the bone may be surgically reshaped for aesthetic purposes. Surgical interference during growth tends to be ineffective with recurrence almost inevitable. Uncommonly it may involve multiple sites (polyostotic). This is more common in females and can be associated with skin pigmentation and sexual precocity (McCune-Albright syndrome). There is a small risk of malignant (sarcomatous) change particularly in the polyostotic variant.
Paget’s disease
Paget’s disease of bone is a non-neoplastic disorder that may have a viral aetiology. It occurs in old age and whereas it may start in a single bone (monostotic), it almost always develops to involve multiple sites (polyostotic). Although most common in the lumbar spine, skull involvement is common, usually calvarium, with maxilla less common and mandible rare. Initially there is bone resorption and at this stage lesions are highly vascular. Later the bone becomes extremely dense. The level of activity of the disease may be measured with the plasma alkaline phosphatase level.
There are significant implications of Paget’s disease for the dentist. In the early phase protracted bleeding may complicate extractions but later the increased bone density and the commonly associated hypercementosis can lead to difficult extractions. Deformity of bone may be evident extraorally and may interfere with the fit of dental prostheses. Bone pain can be a major problem and in extreme cases the high blood flow through active lesions can lead to ‘high-output’ cardiac failure. Surgical reduction is an option if there is significant oral deformity or functional impairment. The treatment of Paget’s can include the use of bisphosphonate drugs, which have a high propensity to induce bony necrosis that is difficult if not impossible to treat (see Ch. 4).
Haemangioma
Intrabony haemangiomas are very rare and may present as solitary radiolucent lesions, which can in some cases be multilocular. They should always be considered in the differential diagnosis of large radiolucent areas in the jaws. They mimic cysts, giant-cell lesions and odontogenic tumours such as ameloblastomas. Aspiration should be carried out before definitive surgery to exclude haemangioma as surgery could, in high-flow lesions, lead to catastrophic haemorrhage. If a haemangioma is suspected (for instance, if there were a soft-tissue haemangioma close to the site of a radiolucency of bone), specialized imaging such as CT and angiography should be considered to define the extent of the lesion and the blood supply. Treatment, if appropriate, may extend from interventional radiology techniques such as embolization to formal surgical excision.
Intrabony and other malignancies
The most common malignant diseases in the mouth are oral squamous cell carcinoma (see Ch. 10) and salivary gland tumours (see Ch. 14).
As with any other part of the skeleton, the facial bones can be the origin of primary malignant disease such as sarcomas arising from bone or cartilage, or the site of a metastatic deposit or systemic malignant disease such as myeloma or lymphoma. In addition there is a group of diseases characterized by proliferation of antigen-presenting cells (Langerhans cell histiocytosis), which although not defined as malignant, in many ways behave as malignant disease. Readers are advised to refer to relevant pathological texts.
Because early diagnosis gives the best chance of curative treatment, it is essential that clinicians recognize the symptoms and signs that would depict malignancies of this type. Invasion with destruction of adjacent tissues is the hallmark of malignancy and is usually coupled with rapidly developing swelling. The classical clinical presentation is swelling of relatively short duration, loose and/or displaced teeth and dysaesthesia of the mental or other branches of the trigeminal nerve. Radiological signs also reflect this with irregular, ‘moth-eaten’ margins typical of sarcoma and secondary deposits or the circumscribed, but ‘punched out’ lesions suggesting multiple myeloma, all without cortication of the margin. The radiodensity will vary from totally radiolucent tumours such as myeloma, through mixed radiodensity seen with some osteosarcomas and chondrosarcomas, to the radio-opaque lesion typical of a secondary deposit from the prostate. It is important to remember that ‘moth-eaten’ margins with mixed radiodensity are also typical of osteomyelitis, osteoradionecrosis and bisphosphonate-induced bone necrosis. Nasopharyngeal tumours can present with painless hard enlarged cervical lymph nodes, unilateral deafness, mandibular/lingual nerve dysaesthesia and chronic progressive trismus (Trotter’s syndrome). Prompt diagnosis is paramount and all of the above require urgent referral.
Even though these diseases are very rare, clinicians must maintain an index of suspicion and either arrange urgent referral or, if in doubt, contact specialists directly and discuss the clinical findings. Diagnosis of this group of diseases is complex and usually requires specialist biopsy and imaging techniques and therefore they should not be biopsied in general practice.
BIOPSY
Biopsy is defined as the complete or partial removal of a lesion for laboratory examination to aid definitive diagnosis. Whereas biopsy usually establishes the definitive diagnosis, there are occasions when the result is not clear and in these circumstances the available histological data must be measured against clinical findings.
Biopsy techniques
Excisional biopsy
This is the ideal approach for small superficial lesions less than 1 cm in diameter, where the clinical appearance suggests that it is benign. Most if not all of these procedures can be performed using local anaesthesia (LA). Where regional blocks are not performed, the local anaesthetic should be injected at a distance from the lesion to avoid distortion of the tissue at the operating site.
Excisional biopsy of superficial lesions
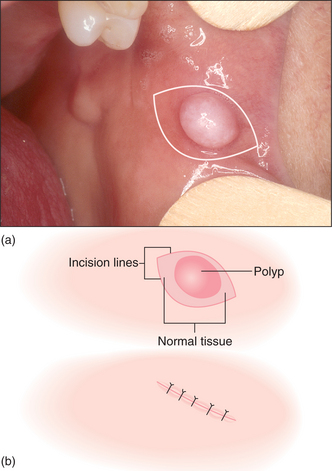
Superficial lesions such as fibroepithelial polyps and squamous papillomas that are pedunculated (arising from a narrow stalk) can be excised by simple division of the base of the pedicle where it arises from the mucosa. For papillomas that have a viral aetiology some surgeons like to use electrosurgery. For larger and sessile lesions (broad-based) excision should be based on an elliptical incision (Fig. 8.8a). This is made around the base of the lesion (No. 15 blade) and should include a 1- to 2-mm margin of normal tissue. Choice of such an incision is aimed at creating a wound that will appose (Fig. 8.8b) without redundant tissue (dog ears) and close without tension, because the ellipse needs to be at least twice as long as it is broad. A suture may be placed at the edge of the ellipse to hold the tissue. This will avoid crushing the specimen with tissue forceps. It can be used to retract the specimen during dissection with scissors or scalpel. When dissecting the specimen free it is important to avoid damaging underlying structures such as blood vessels and nerves by using ‘blunt dissection’ (described below).
Excisional biopsy of submucosal lesions
To excise submucosal lesions the covering mucosa needs to be divided to develop access and establish the plane of dissection. This is therefore more complicated than excision of superficial lesions. The orientation of the incision will be along the longest dimension of the lesion but must also take into account anatomical relationships such as the opening of the parotid duct or the mental nerve. For large or superficial lesions such as mucous cysts that have thinned the mucosa this technique can be modified to an elliptical incision to include a portion of the covering mucosa. This will reduce the risk of rupture, or perforation of the lesion at the superficial aspect, but will mean sacrificing the overlying mucosa.
To avoid distortion the outline of the incision can be marked using a surgical pen before the introduction of LA. The initial incision should divide the mucosa but not penetrate the specimen, so the depth will vary depending on the site and depth of the lesion. Superficial lesions covered by thin mucosa, as found in the floor of the mouth or lower lip, will require very careful work to avoid related anatomical structures. Following the incision, a combination of sharp and blunt dissection is required to define the plane of dissection and then deliver the lesion. In the main, blunt dissection is advised as it will define the tissue plane around the lesion and avoid damage to adjacent structures such as blood vessels and nerves.
In this technique blunt-ended scissors are introduced into the tissues at the margin of the lesion with the points together. The points are then separated, forcing the tissues apart along their natural planes and the instrument is withdrawn with the blades open so there is no cutting action. The process is best started away from the ‘thin’ covering mucosa or superficial part of the lesion as it is easier to get the plane identified and started. Dissection is then repeated from various angles to ‘develop’ the plane all around the lesion. As required, sharp dissection either with a scalpel or by cutting with scissors may also be used to finally deliver the specimen. During this stage of the process great care must be taken to prevent damage to adjacent and underlying structures, and to avoid perforating the lesion.
Incisional biopsy
This technique is indicated in cases where the diagnosis is in doubt (i.e. possible malignancy or potential for recurrence) and complete excision, in one stage under LA, is impractical in terms of size, complexity and/or the patient’s ability to cope with the surgery.
Incisional biopsy of superficial lesions
In the hospital context, if there is a suspicion of malignancy, the biopsy must be taken from a representative part of the lesion and include a suitable edge of normal tissue. This allows the sample to be held at the normal tissue margin and thereby avoids introducing surgical artifact. It also gives some idea of the pattern of invasion at the interface. An elliptical incision is suitable, with a sharp dissection technique using a scalpel and scissors.
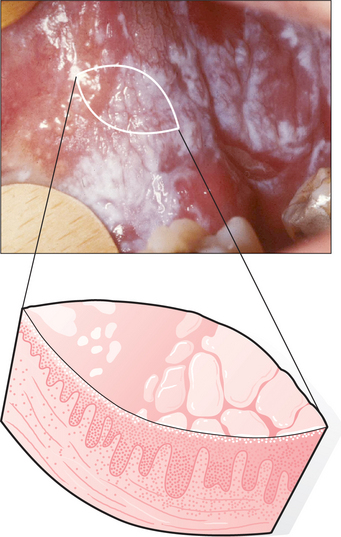
If the biopsy is of a mucosal lesion such as leukoplakia, it is important to take a deep enough specimen to get into connective tissue (approx. 6 mm) (Fig. 8.9) and to take a broad enough piece, so that after processing there will still be sufficient for a number of complete sections to be cut from it in the laboratory (at least 4 mm). Where the field of mucosal abnormality is extensive, it may be necessary to carry out multiple incisional biopsies. If this technique is carried out, it is important to draw a diagram of the lesion to make orientation clear for the pathologist.
Incisional biopsy of submucosal lesions
For large, solid submucosal swellings, particularly in the palate where the risk of minor salivary gland malignancy is high, incisional biopsy is often the procedure of choice but this decision carries significant implications for further management and therefore should be left to specialists.
Soft-tissue repair
Haemostasis is the essential first step and a period of pressure followed by suturing will achieve this in most cases. If necessary, electrocoagulation can be used but with care in areas such as the lips where nerves are superficial. Careful wound closure is important and choice of suture depends on the thickness of the mucosa. In most cases interrupted sutures using a 3/0 resorbable material on a 22-mm half-circle cutting needle is indicated but for thin mucosa such as the inside of the lip or floor of the mouth a 4/0 suture is advised.
The tension of the sutures is important and they need to be tight enough to appose the wound edges firmly but not so tight as to impair the blood supply (wound blanching). Deep sutures are only required if there is a second layer, such as tongue muscle, to repair. When repairing lip incisions avoid placing the suture through the deeper tissue as this could damage adjacent structures such as other minor salivary glands and nerves. Similarly avoid multiple or tight sutures in the floor of the mouth close to the opening of the sublingual and submandibular glands to avoid strictures.
Repairing biopsy wounds involving mucoperiosteum such as hard palate mucosa is problematic in that the wound edges cannot be approximated. They require a pack of haemostatic gauze, or other dressing such as a Whitehead’s varnish (Compound Iodoform Paint) pack or a periodontal wound dressing placed over the defect, possibly supported by a healing plate. In some cases it may be preferable to cover the defect either by an advancement or transposition flap of adjacent mucosa.
Other biopsy techniques
Punch biopsy
A circular blade can be used to remove a cylindrical core of tissue as a form of incisional biopsy: this punch biopsy technique is more commonly used on skin.
Trephines
In this technique a core of tissue is trephined from the lesion. It may be used for soft-tissue swellings or bone. This provides sufficient material, retaining tissue architecture, for histological diagnosis and is useful for lesions where access may be difficult.
Cytology
This technique is based on microscopic sampling of tissue either by fine needle aspiration (FNA) or by using an abrasive technique such as brush or swab (exfoliative cytology). Both aim to remove cells representative of the lesion, which are then transferred to microscope slides and prepared for histo-cytological assay. These are investigations that require special equipment and skill in both taking the sample and preparing the slides and, as such, are best referred to operators with the appropriate experience. All such methods lack the advantage of maintaining tissue architecture, but offer a relatively quick and minimally invasive approach to biopsy.
Aspiration biopsy
Fine-needle aspiration cytology is a useful method for sampling soft-tissue masses in the head and neck, such as lymph nodes and salivary glands. A 10- or 20-mL syringe with a 21-gauge needle is installed into a specially designed holder which facilitates the safe and controlled introduction of the needle, and allows aspiration at variable pressures. The needle is inserted into the centre of the deep tissue mass and the plunger pulled back, drawing a small ‘core’ tissue sample into the needle. The needle tip is re-sited within the lesion and the procedure repeated several times. The sample is then delivered directly onto a microscope slide, spread and fixed. This is a valuable method but technique sensitive. Accurate sampling is clearly essential and on occasions where the lesion is deep it can be combined with ultrasound investigation to ensure the needle is positioned within the lesion (ultrasound-guided FNA).
Smears
Cytological smears can be used to sample the surface of epithelial lesions, especially where the lesion is not heavily keratinized, in which case it is possible to harvest cells deeper within the epithelium. The technique could be used as an adjunct to biopsy for monitoring widespread lesions, which may undergo malignant transformation. A diagnosis of malignancy based on cytology alone would be considered insufficient by most pathologists and formal histological sampling would be required before proceeding to cancer treatment.
Care of the specimen
The key to a successful biopsy is the presentation of either the entire lesion or a representative sample of tissue uncontaminated and of good quality. The sample must not be allowed to dry out and should be placed immediately in a tissue fixative (usually 10% neutral buffered formalin). These are prerequisites for the pathologist to have tissue adequately prepared for histological interpretation. However, if special immunohistochemical techniques such as immunofluorescence are required, the tissue sample requires special processing and needs to be referred to a hospital department. Some pathologists prefer to receive all specimens fresh, on saline-soaked gauze, provided the specimen reaches the laboratory within one hour of being taken.
In the UK there are strict regulations governing transport of the specimen to the laboratory if sent by post: all the appropriate regulations should be adhered to.
Biopsy in general dental practice
For the practitioner to proceed with surgery he/she must have the appropriate skills to be able to complete the procedure and to deal with intraoperative and postoperative complications.
Generally excisional biopsy of superficial benign lesions is suitable for general dental practice with the more complicated submucosal excisional biopsies dependent on the training and experience of the individual. Solid submucosal lesions have a high probability of being a tumour and are best referred.
Incisional procedures by definition are aimed at establishing diagnosis. This will then raise issues with continuing care, the interpretation of potential for malignant change and the advisability of further treatment including major surgery. These points and the possibility of a diagnosis of malignancy would suggest that patients requiring this type of biopsy would be best referred.
As with all proposed treatment the patient needs to be able to give informed consent and with surgery this should be written consent. It requires clear explanation of the nature and significance of the problem, possible treatment options and the risks and benefits of surgery in contrast to no active treatment. Should the option be to refer the patient then the letter must be clear and concise and if there are concerns about a possible malignancy the specialist should be contacted by phone.
Barnes L., editor. Pathology and genetics of head and neck tumours. Lyon, France: IARC Press, 2005.
Cawson R.A., Binnie W.H., Barrett A.W., Wright J.M. Oral disease clinical and pathological correlations, 3rd edn. St Louis, MO: Mosby, 2001. Chs. 6, 7, 8 & 10.
Kramer I.R.H., Pindborg J.J., Shear M. Histological typing of odontogenic tumours. Heidelberg, Germany: Springer Verlag, 1992.
Peterson L.J., Ellis E., Hupp J.R., Tucker M.R., editors. Contemporary oral and maxillofacial surgery, 3rd edn. Mosby, St Louis, MO, 1997;512-532, Ch. 23
Waites E. Essentials of dental radiography and radiology, 3rd edn. Edinburgh: Harcourt Health Sciences, 2002.
SELF-ASSESSMENT
Answers on page 265.