Traumatic Spinal Cord Injury
SPINAL CORD INJURY
Spinal cord injury (SCI) is a catastrophic event of low incidence and high cost. It is most often highly active persons who incur the types of accidents that cause severe SCI. Within a matter of seconds, the person sustaining a traumatic SCI will become dependent on others or on assistive devices to perform even the most basic activities of self-care. SCI rarely occurs in isolation, and over 75% of these individuals have some other systemic injury. In 10% to 15%, there is an associated head injury. This concern has led to the widely quoted clinical maxim that all traumatized patients or any patient with a severe head injury should be presumed to have a spine injury or SCI until proven otherwise.30
Incidence and Risk Factors
Males account for more than 80% of all cases of traumatic SCI. Most of the injured have traditionally been young people, but the mean age of SCI increased in the 1990s and is now recorded as the early thirties.11,12 It is postulated that there is now a higher survival rate in the older population; in addition, the mean age of the general population also has increased. It is estimated that there are approximately 30 to 40 cases per 1 million persons on an annual basis, with an additional 6 to 8 deaths (per 1 million) occurring before hospitalization; however, the number of deaths occurring before hospitalization is decreasing. The number of individuals living with SCI is probably between 183,000 and 230,000 persons, with approximately 10,000 new cases each year.
The primary cause of SCI is motor vehicle accidents, accounting for 40% of SCIs. The overall incidence of SCI associated with automobile accidents has decreased in the 1990s because of mandatory seat belt laws and installation of air bags in cars manufactured after 1990. Still, drivers aged 16 to 20 have an annual rate of involvement in fatal crashes of 62 per 100,000 licensed drivers, compared with 29 per 100,000 for the general public.
In some urban areas the incidence of gunshot-related SCI is increasing. Approximately 15% of SCIs are caused by gunshot wounds; these include job-related injuries to security guards, policeman, workers shot during robberies, and others. The likelihood of SCI from a gunshot wound appears to be higher among those who have had previous gunshot wounds (30%) or who have had prior involvement in the criminal justice system (52%). Unlike sports injuries, which peak nationwide during the summer months, the incidence of penetrating wounds of the spine remains the same throughout the year, and 40% of them occur on a Saturday or Sunday.25
Sports-related injuries account for less than 7% of the total. Accidents resulting in SCI are most prevalent in the contact sports of football and wrestling, high-speed sports such as snow skiing and surfing, and sports in which injuries can involve a fall from a height, such as a trampoline or a horse. The most preventable cause of SCI is diving. The injury occurs most often in water 4 to 6 feet deep. Fig. 34-1 shows a breakdown of the typical incidence of injury types.
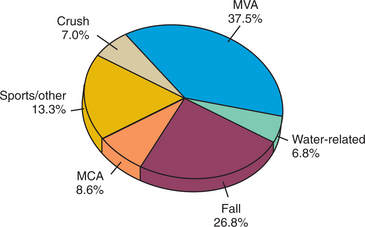
Figure 34-1 Epidemiology of spinal cord injury. MCA, Motorcycle accident; MVA, motor vehicle accident. (From Goetz CG, ed: Textbook of clinical neurology, ed 2, Philadelphia, 2003, Saunders.)
Approximately 47% of all persons with SCIs have paraplegia, and 53% have tetraplegia. Twenty-six percent of all thoracic and lumbar cord injuries are complete lesions, as are 18% of cervical injuries. For penetrating wounds of the spine, however, a significantly greater proportion are complete, and there is a much greater shift toward thoracic spinal injuries as compared with injuries of the neck or lumbar spine.72
The incidence of traumatic SCI in small children is low, and they represent less than 10% of the traumatic SCI population. In children, SCI is most often related to an automobile accident. After 45 years of age, falls become the most common cause of SCI.
In most high-income countries today SCI is the leading cause of death between the ages of 1 year and 44 years. Similar trends are seen in today’s low-and middle-income countries, with increases in many injury-related causes of death, especially road traffic. Because of recent increases in the use of motorized transport globally, road traffic crashes have now become a leading cause of death among young, working-aged adults in almost every country. In the age group 15 to 44 years, road traffic deaths are second only to human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS) as a cause of death.
All too often, injury prevention is misconstrued as merely admonitions to be careful. In reality, injury prevention is a scientific field that seeks to understand the extent and characteristics of injury through surveillance and research. There is a need to identify risk factors and target these risk factors through well-developed and scientifically based prevention efforts. Recent developments to improve road safety in the United States have included graduated licensing and strengthened efforts to combat drunk driving, including wider adoption of a blood alcohol concentration of 80 mg/dl as the criterion for drunk driving.55
Definition and Etiologic Factors
SCI is classified as concussion, contusion, or laceration. A concussion is an injury caused by a blow or violent shaking and results in temporary loss of function, similar to the cerebral concussion associated with head injury. In contusion injury the glial tissue and spinal cord surface remain intact. There may be a loss of central grey and white matter, which creates a cavity that is surrounded by a rim of intact white matter at the periphery of the spinal cord. Laceration or maceration of the cord occurs with more severe injuries in which the glia is disrupted and the spinal cord tissue may be torn. Occasionally this can result in complete transection of the cord. Gunshot wounds, knife wounds, and puncture injuries fall into this category.
Hemorrhages into the dura are common, although they rarely become large enough to compromise the spinal cord. Subarachnoid hemorrhages, caused by contusion and laceration of the cord, are frequent and can cause further compression of the cord.
The mechanism of injury influences the type and degree of the spinal cord lesion. Fig. 34-2 shows the flexion damage that is referred to as the hangman’s fracture, related to excessive flexion. Approximately 50% of injuries come from excessive flexion of the spinal column that results in a severe neurologic disorder.18 Fig. 34-3 shows how extension can cause SCI in the elderly population. Fig. 34-4 shows vascular changes that may result from displacement of spinal components. The spinal cord is often violently displaced or compressed momentarily during an injury with forceful flexion, extension, and rotation of the spine. The vertebral body can burst and cause pressure or scatter bone fragments into the spinal cord. Fig. 34-5 illustrates this phenomenon. Complete spinal cord lesions occur in about one third of flexion injuries. With crush fractures of the vertebrae, there is a 75% chance of a complete spinal cord lesion.
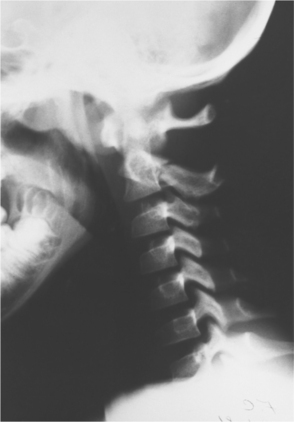
Figure 34-2 Fracture of C2 (hangman’s fracture). (From Green NB, Swiontkowski MF, eds: Skeletal trauma in children, ed 3, Philadelphia, 2003, Saunders.)
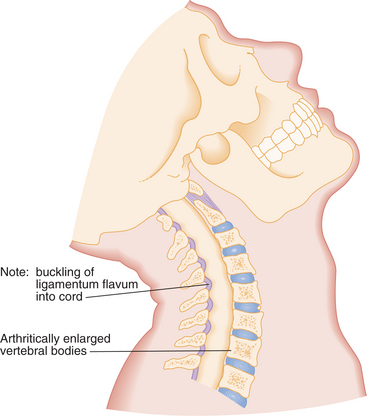
Figure 34-3 Elderly patients subjected to extension forces can sustain cervical spinal cord injury as a result of compression of the spinal cord between the posterior hypertrophic ligamentum flavum and the arthritically enlarged anterior vertebral bodies. (From Marx JA, ed: Rosen’s emergency medicine: concepts and clinical practice, ed 6, Philadelphia, 2006, Mosby.)
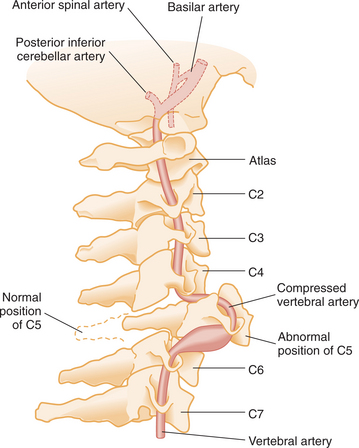
Figure 34-4 Mechanism of vascular injury of the spinal cord resulting from cervical vertebral injury. (From Marx JA, ed: Rosen’s emergency medicine: concepts and clinical practice, ed 6, Philadelphia, 2006, Mosby.)
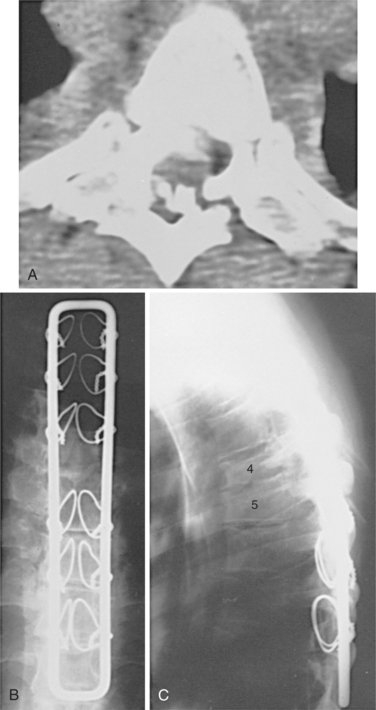
Figure 34-5 A T4-T5 fracture-dislocation resulted in a complete spinal cord injury in a 30-year-old man. A, A computed tomographic scan through the injured level demonstrates marked displacement and comminution at T4-T5, with multiple bone fragments within the canal. B, A postoperative anteroposterior radiograph shows stabilization with a Luque rectangle and sublaminar wires. This instrumentation provided rigid fixation and allowed early mobilization with minimal external support. The strength of fixation could have been improved with the use of double wires around the lamina bilaterally. C, Postoperative lateral radiograph. (From Browner BD, Jupiter JB, Levine AM, et al: Skeletal trauma: basic science, management, and reconstruction, ed 3, Philadelphia, 2003, Saunders.)
The majority of spinal cord–injured patients have at least one other system injury. Occasionally these injuries take precedence in evaluation and treatment. If one level of bony injury has been identified, it is necessary to survey the entire spine, because there is a 10% to 15% incidence of spinal injury at other levels.30
The difference between a complete and an incomplete spinal cord lesion may depend on the survival of a small fraction of the axons in the spinal cord. Evidence of axonal conduction across the lesion site has been found in individuals with clinical neurologic diagnoses of complete SCI at that level. The surviving axons may be injured and therefore have a decreased response to stimuli. The injured axon conducts slowly and fatigues rapidly.79
Pathogenesis
The development of the spinal lesion occurs over time and in a neuroanatomic distribution. In the first 18 hours, there is necrotic death of axons that were directly disrupted by the trauma. In the following weeks, there is further progression of tissue injury in both directions from the lesion. The immune system probably plays a major role during this phase. It appears that immune cells, such as monocytes and macrophages, emit chemical signals, such as cytokines and chemokines, that trigger apoptosis, or programmed cell death. This breakdown of cell function can occur away from the lesion site, often by as far as four spinal segments.17
The pathophysiology of SCI may be divided into phases. Primary injury refers to the structural damage occurring instantly after the traumatic event. Trauma to the spinal cord results in primary destruction of neurons at the level of the injury by disruption of the membrane, hemorrhage, and vascular damage. More extensive primary injury may occur, however, if an injured spine is not adequately immobilized. A critical aspect of these lesions is that even after severe injuries, a small peripheral rim of spared tissue and axons often remains. Spared descending systems play an important role in recovery. In paraplegia, the amount of spared rim correlates with the level of locomotor function.5,70
Secondary injury refers to a pathophysiologic cascade initiated shortly after injury, including such insults as ischemia, hypoxia, edema, and various harmful biochemical events. The spread of damage is thought to be due to initiation of biochemical events leading to necrosis and excitotoxic damage and can continue for hours, days, or weeks.3 Fig. 34-6 shows the changes that can result from SCI. Because it is extremely rare for the primary injury to cause transection of the spinal cord, and it has been shown that less than 10% of the cross-sectional area of the spinal cord supports locomotion, it is very important to focus clinical attention on the secondary injury process.
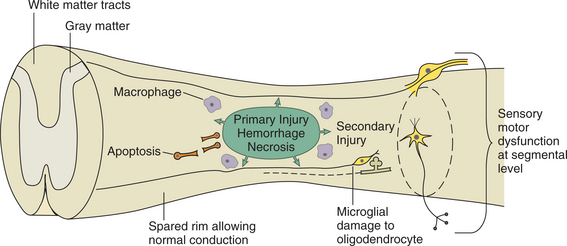
Figure 34-6 Spinal cord contusion lesions are characterized by a primary area created by hemorrhage of blood vessels causing necrosis of cells. This area eventually spreads because of secondary injury associated with apoptosis (programmed cell death), macrophages acting as immune mediators, and microglia causing damage to oligodendrocytes. The secondary damage may continue for days to weeks and move along the segmental levels, causing sensory and motor dysfunction. The spared rim may allow normal processing and preservation of function.
Electrolyte disturbances following SCI include increased intracellular calcium level, increased extracellular potassium level, and increased sodium permeability. The route of calcium entry rather than the amount may be the critical component. The influx of calcium ions in the neuronal cell can then lead to activation of various secondary processes resulting in cellular death. Excitatory neurotransmitter accumulation, arachidonic acid release, endogenous opiate activation, and prostaglandin production can cause damage as part of the postinjury cascade. Free radical production and lipid oxidation play a central role in this process. See Chapter 28 for information regarding the effects of these disturbances. This results in ischemia, edema formation, membrane destruction, cell death, and eventually permanent neurologic deficits.30
Blood Flow Changes.: Ischemia related to reduced blood flow is a very prominent feature of post-SCI events. Damage to blood vessels results in microhemorrhage in the central grey matter, which spreads radially and axially. The resulting hypoxic and ischemic events deprive grey and white matter of oxygen and nutrients necessary for neural cell survival and function. Within 2 hours of injury there is a significant reduction in spinal cord blood flow. Swelling rapidly occurs at the injury level, and because the bony spinal canal has a fixed diameter, pressure on the cord climbs higher than venous blood pressure. Ischemia in the area of injury may be due to the presence of norepinephrine, serotonin, histamine, and prostaglandins, all of which cause vasoconstriction. This ischemia may be compounded by loss of the normal autoregulatory response of the spinal cord vasculature.
Changes in blood flow, represented by small hemorrhages, begin soon after the SCI. After several hours there appear to be gross hemorrhages present, preceded by endothelial breakdown and pathologic coagulation products in the blood vessels. Ischemia and necrosis occur primarily in the grey matter, presumably because of the richer blood supply. Macrophages enter the lesion and begin to digest the necrotic debris, converting the complex myelin lipids to neutral fat. Axonal swelling and increased permeability of blood vessels result in a visibly swollen spinal cord.77 Glial cells become active after about 6 days, and astrocytic fibers form scarlike tissue that lines the cavities created by the necrosis.
Autoregulation of circulation is disabled at the injury site. Systemic pressure changes may be responsible for changes in spinal blood flow, which may cause nervous tissue damage by direct effects. The changes in blood flow may reflect rather than cause secondary injury.34
Edema.: Edema formation is another feature of the secondary injury process. Edema develops first at the injury site and subsequently spreads into adjacent and sometimes distant segments of the cord. The relationship between this edema and worsening of neurologic function is not well understood. The relationship of mechanisms of damage leading to cell death is shown in Fig. 34-7.
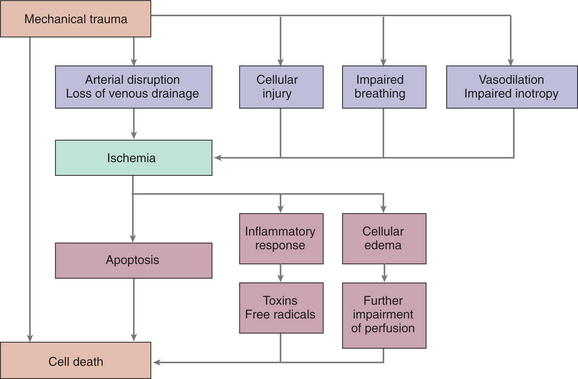
Figure 34-7 Mechanisms of spinal cord injury. Mechanical trauma to the spinal cord is exacerbated by systemic hypoperfusion or hypoxia. (From Miller RD, ed: Miller’s anesthesia, ed 6, New York, 2005, Churchill Livingstone. Redrawn from Dutton RP: Spinal cord injury, Int Anesthesiol Clin 40:109, 2002.)
Demyelination.: Demyelination results in reduced rate of firing in the injured spinal cord. The demyelination is due to direct trauma to the oligodendroglial cells that produce myelin. Unlike neuronal excitotoxicity, which is mediated predominantly by N-methyl-Daspartate (NMDA) receptors, mature oligodendrocytes are sensitive to excitotoxicity mediated by non-NMDA receptors. Lymphocytes and macrophages invade the lesion site by way of the disrupted blood-brain barrier as part of the inflammatory response. The myelin sheath becomes thin between the nodes, and this is responsible for a decrease in the peak currents along the axon. Loss of a single segment of myelin renders an axon dysfunctional; therefore, a large subset of axons crossing the lesion eventually become nonfunctional despite the axon’s remaining physically intact.
Changes in white matter begin with wallerian degeneration in the ascending posterior columns above the level of the lesion and in the descending corticospinal tracts. Wallerian degeneration may be triggered by microglial activation and by the destruction of the oligodendrocytes via the release of cytokines or other neurotropic factors.44 The immune system appears also to trigger the release of nerve growth factor, which can be neuroprotective to some cells while it is toxic to other cells in the spinal cord.
A prominent feature of subchronic SCI is the maturation of a scar around the lesion. This scar tissue forms a cellular and molecular barrier to axonal regeneration. By the chronic injury phase, the scar is well formed and consists of several cell types, such as the reactive astrocytes, fibroblasts, Schwann cells, microglia, and macrophages that have invaded the scar.
Grey Matter.: Typically, the loss of central grey matter is confined to between one and one and one-half segmental levels of the spinal cord, causing central cavitation. The result is a fluid-filled cyst or syrinx (see later), or the cord collapses around the loss of tissue in an hourglass shape with the minimal diameter located at the spinal segment of the original injury.
Dural Scarring.: Scarring of the dura can cause a permanent connection of the cord to the overlying dura. Because the cord is normally freely mobile within the spinal canal, restricted motion attributable to dural scarring produces unusual forces on the cord when the neck is bent or with normal breathing or the cardiac cycle. These forces can produce microscopic injury, which may limit optimal regeneration and recovery.
Neural Function.: Neural activity below the injury level is related to passive and active limb movements and sensory stimuli from the moving limbs. Substantial reduction of neural activity limits the body’s ability to maintain the cellular functions of the spinal cord circuitry. Slow progressive loss of function is normal in chronic SCI. It is believed that the injured central nervous system (CNS) undergoes accelerated aging, with abnormal cell production and impairments in mechanisms of cellular repair. The cellular mechanisms important for regeneration may be lost as a part of this process.
Animal models of SCI suggest that areas of the brain involved in sensorimotor control undergo atrophic changes after spinal cord transection. A significant decrease in the size and number of corticospinal neurons has been demonstrated in the rat brain. Atrophy may occur in the subacute phase (5 to 10 weeks), with cell death occurring in the chronic phase (months to years). In the somatosensory system, shrinkage of the dorsal column nuclei and thalamus has been demonstrated in primates following upper limb deafferentation. Thus, both the somatosensory and motor systems are susceptible to atrophy after nervous system damage.42
Neurapraxia.: The syndrome of neurapraxia is of special concern after athletic injury. Affected individuals experience dramatic, although transient, neurologic deficits, including tetraplegia. Transient tetraplegia most commonly occurs as a result of axial loading of the spine. In athletes with narrowing of the anteroposterior diameter of the spinal canal, both hyperextension and hyperflexion can lead to cord compression. This is referred to as the pincer mechanism. In the already stenotic canal, with hyperextension the cord is compressed between the posterior inferior margin of the superior vertebral body and the anterior superior aspect of the spinolaminar line of the subjacent vertebra. Conversely, in hyperflexion the cord is compressed between the anterior superior aspect of the spinolaminar line of the superior vertebra and the posterior superior margin of the inferior vertebra. In both cases, this sudden decrease in anteroposterior diameter of the spinal canal results in compression of the spinal cord. Many attempts have been made to quantitate the level of risk to these individuals from continued athletic participation; however, considerable controversy still exists.27
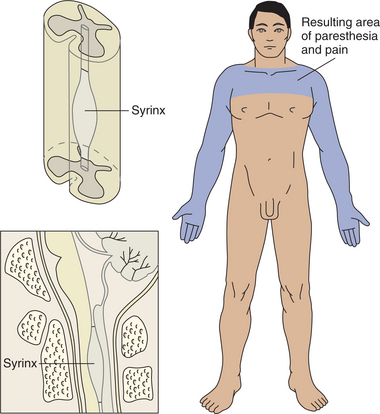
Syringomyelia.: One type of pathologic condition that can appear over time in the spinal cord related to trauma is syringomyelia. It is a clinical syndrome that results from cystic cavitation and gliosis of the spinal cord. This is reported to occur in close to 2% of persons with paraplegia and in 0.2% of quadriplegic individuals. In the chronic spinal cord lesion, the cysts may continue to develop, become tubular in shape, like that of a syrinx, and extend over several spinal levels (Fig. 34-8). Posttraumatic syringomyelia can develop up to 30 years after the initial lesion, but most commonly occurs within 4 to 9 years after trauma. One mechanism of syrinx formation is an initial hematomyelia followed by resorption and formation of a cyst cavity.30
In some cases, there are multiple cavities. The cavity may occupy almost the entire cross-sectional area of the cord, compressing the posterior columns. As the cyst develops, usually below the level of the initial lesion, there can be significant pain as a result of the compromise of the central spinal cord structures, such as the substantia gelatinosa and the posterior root entry zone.73 The spinothalamic tracts are involved, which can result in the sharp pain that is often the first presenting symptom. There can be lower motor neuron dysfunction, causing weakness, atrophy, and loss of reflex activity. Sensory loss is common, and the sympathetic nervous system can become involved, resulting in syndromes such as Horner’s syndrome.48
The thoracic area of the spine is the most common site for the syrinx to develop, with descending and ascending fibers running in the walls of the cavitated lesions. The size and extent of the syrinx are represented by the symptoms, but because of location below the site of the lesion, changes are not easily recognized. The syringomyelia may be responsible for the spasms, phantom sensations, reflex changes, and autonomic visceral phenomena that may occur. Scoliosis can result from the loss of input to the paraspinals.44 Anything that blocks the free flow of cerebrospinal fluid (CSF) can keep this fluid from moving normally in and out of the head. Pressure can build up in the syrinx, causing expansion and possible rupture, damaging normal spinal cord tissue and injuring nerve cells. Many people with posttraumatic syringomyelia do not develop any symptoms until midlife or later.32
Syringomyelia can be a very disabling condition. Spasms, phantom sensations, and autonomic visceral (organ) changes can occur. Sexual dysfunction, muscle spasticity, and loss of bowel or bladder control can develop. The first symptom may be sharp pain. Muscle atrophy, stiffness, and weakness of the neck, back, shoulders, arms, or legs, along with loss of reflexes, are common. Symptoms may be distributed like a cape over the shoulders and back. Headaches and loss of sensation (pinprick and temperature) in the hands may be reported. The symptoms may only occur on one side of the body, depending on where the syrinx develops.
Clinical Manifestations
Level of Injury.: SCIs are named according to the level of neurologic impairment. Differences may exist in the motor versus sensory levels identified. The American Spinal Injury Association (ASIA) has created standards for assessment and classification that are widely used (Figs. 34-9 and 34-10). The sensory examination consists of testing 28 dermatomes on each side of the body using pinprick and light touch, with the sensation in each then scored as follows: 0 = absent, 1 = impaired, 2 = normal. Sensation of the external anal sphincter is tested as yes or no. The level of inquiry reflects the most caudal level of the spinal cord that exhibits intact sensory and motor functioning.21,26

Figure 34-9 American Spinal Injury Association (ASIA) motor assessment form. (Courtesy American Spinal Injury Association International, Atlanta.)
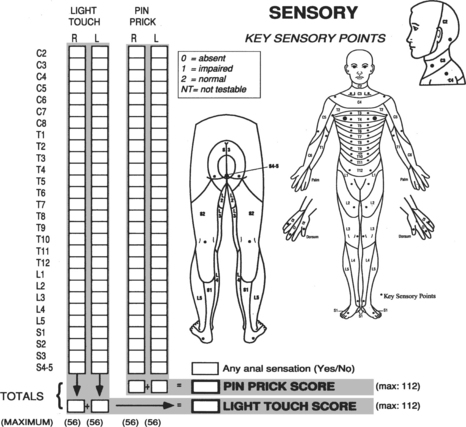
Figure 34-10 American Spinal Injury Association (ASIA) sensory assessment form. (Courtesy American Spinal Injury Association International, Atlanta.)
Identification of motor impairment is more problematic, given the dual innervation of many muscles. The strength of a given muscle is a reflection of the functioning of two or more cord segments. Loss of innervation from a spinal cord level results in weakness of the dual innervated muscle. To determine the level of innervation, the therapist looks for the muscle that has a two thirds muscle strength with a one third muscle strength in the next most rostral muscle. Volitional contraction of the anal sphincter is also noted. Asymmetric damage may result in different levels of neurologic impairment on the left versus the right side. The lesion can be then be reported as, for example, a right C5 and a left C6 lesion.
Lesions are reported as complete when there is complete loss of sensory and motor function below the level of the lesion. Complete lesions are a result of spinal cord transection, severe compression, or extensive vascular dysfunction.
Incomplete lesions are the partial loss of sensory and motor function below the level of the injury. Incomplete lesions often occur when there is contusion produced by bony fragments, soft tissue, or edema within the spinal canal. The resulting motor or sensory function is called sparing. Box 34-1 describes the ASIA method for describing impairment related to SCI.
Spinal Cord Injury Syndromes.: Within the category of incomplete spinal cord lesions are the recognizable syndromes that have been identified. Several syndromes are illustrated in Fig. 34-11.
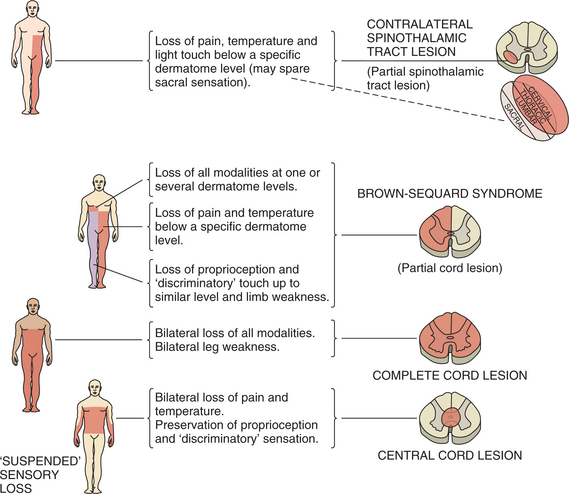
Figure 34-11 Spinal cord syndromes. Patterns of sensory loss and weakness. (From Lindsey KW, Bone I, Callander R: Neurology and neurosurgery illustrated, New York, 1986, Churchill Livingstone, p 188.)
Brown-Séquard syndrome is characterized by damage to one side of the spinal cord. The most common causes are stab and gunshot wounds. Loss of the entire hemisection of the spinal cord is rare; the natural lesion is always irregular. There is weakness ipsilateral to the lesion. Lateral column damage results in abnormal reflexes, including a positive Babinski’s sign, and clonus. Often there is ipsilateral spasticity in the muscles innervated below the lesion. As a result of dorsal column damage, there is loss of proprioception, kinesthesia, and vibratory sense. On the contralateral (opposite) side, there is pain and temperature loss starting a few levels below the lesion. The lateral spinothalamic tract ascends on the same side for several segments before crossing, giving rise to the discrepancy between the level and contralateral signs.
Anterior cord syndrome is frequently associated with flexion injuries and is often the result of loss of supply from the anterior spinal artery. Damage to the anterior and anterolateral aspect of the cord results in bilateral loss of motor function and pain and temperature sensation because of interruption of the anterior and lateral spinothalamic tracts and corticospinal tract.
Central cord syndrome is a result of damage to the central aspect of the spinal cord, often caused by hyperextension injuries in the cervical region. There is characteristically more severe neurologic involvement in the upper extremities than in the lower extremities. Peripherally located fibers are not affected, and therefore function is retained in the thoracic, lumbar, and sacral regions, including the bowel, bladder, and genitalia.
Posterior cord syndrome is extremely rare, with preservation of motor function, pain, and light touch sensation. There is loss of proprioception below the level of the lesion, leading to a wide-based steppage gait.
Conus medullaris syndrome and cauda equina syndrome reflect damage at the base of the spinal cord and generally result in lower limb paralysis, reflexive bowel, or both.
The site of spinal cord damage determines the extent of the physical impairments. Injury of the cord in the cervical region creates tetraplegia, or paralysis of all four limbs. In addition to the limbs, the trunk and muscles of respiration are involved. Damage in the thoracic or lumbar region will result in paraplegia or paraparesis involving only the lower extremities and generally the lower trunk.
Changes in Muscle Tone.: Paralysis of the voluntary musculature is the most obvious effect of SCI. Damage can involve the descending motor tracts, anterior horn cells, or spinal nerves, and it is often seen in combinations of these. Spinal shock is the loss of sensory, motor, and automatic control below the level of the lesion that occurs immediately after the trauma but resolves within a few weeks after injury.27
When the descending tracts are involved, immediate flaccidity is present and reflexes are absent. This is followed by autonomic symptoms, including sweating and reflex incontinence of bladder and rectum. Within weeks there is a gradual increase in the resting tone of the muscles innervated below the lesion, and reflexes reappear.
Spasticity is an inevitable consequence of spinal cord lesions. There is an essential or basic spasticity, which may be of some benefit to the individual when emptying the bladder or flexing the hip and knee. Excess spasticity is due to afferent stimuli. Spasticity can be made worse by the presence of constipation, infection, fracture, or a pressure sore below the level of the lesion, and it can be exacerbated by a sudden change in temperature or by physical or emotional stress. Typically, the flaccid condition lasts longer and spasticity comes later in a cervical injury compared with a thoracic injury.39,43
Autonomic Nervous System Changes.: Autonomic dysreflexia (AD) can occur with a lesion above T5 and is the result of impaired function of the autonomic nervous system (ANS) caused by simultaneous sympathetic and parasympathetic activity. The ANS regulates body functions such as heart rate, blood pressure, and gland activity. Noxious stimuli, such as elevated blood pressure, overextended bladder or bowel, or other visceral stimuli, will typically elicit a sympathetic response, resulting in vasoconstriction and an increase in blood pressure. In the non–spinal cord injured individual, the descending sympathetic output compensates for this increase in blood pressure by causing vasodilation to bring blood pressure to a more normal level. Following SCI, sensory nerves below the level of the injury continue to transmit excitatory impulses, causing similar vasoconstriction and increased blood pressure. With the lack of sympathetic inhibitory output below the lesion, however, the blood pressure keeps rising unchecked. Secretions of neurotrans- mitters, such as norepinephrine, epinephrine, and dopamine, support this sympathetic response. Control of the situation at the brainstem level leads to parasympathetic stimulation to slow the heart rate (bradycardia) through stimulation of the vagus nerve. This response is not strong enough to overcome the extreme vasoconstriction. The vasoconstriction continues above the level of the lesion and results in profuse sweating and skin flushing. A severe pounding headache follows, with sweating and chills without fever. The increase in blood pressure makes the person susceptible to subarachnoid hemorrhage, renal or retinal hemorrhage, and seizure or myocardial infarction. AD should be handled as a medical emergency.1,26 See Box 34-2 for signs and triggers of AD.80
Loss of thermoregulation below the level of the spinal cord lesion is a result of the disruption of the autonomic pathways from the hypothalamus, resulting in subnormal body temperature in a normal ambient environment. Vasoconstriction, the ability to shiver, and the ability to sweat are lost. The body temperature then is greatly influenced by the external environment, and sensory feedback from the head and neck must be used to assist in regulating body temperature. The higher the lesion is, the more severe the problem becomes.
Skeletal Changes.: Joint ankylosis caused by heterotopic ossification, or ectopic bone formation in the soft tissue such as the tendons and connective tissue, can limit range of motion, cause pain, and impair seating and posture. It often develops near the large joints, such as the anterior area of the hip, knee, shoulder, and elbow. It is always found below the level of the lesion, and it begins to develop within the first year after injury. The initial symptoms are soft tissue swelling, pain, redness, and increased temperature in the affected area. Changes in bony alignment can develop secondary to muscle imbalances caused by unopposed contractions. Scoliosis can develop over time due to lack of paraspinal support, as is evidenced in Fig. 34-12.
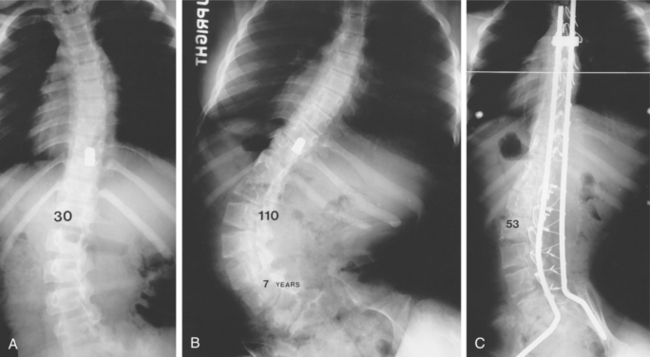
Figure 34-12 Progressive paralytic scoliosis after gunshot wound. A, Initial curve of 30 degrees. B, Seven years later, curve is 110 degrees. C, After fusion and segmental instrumentation, correction to 53 degrees. (From Canale ST, ed: Campbell’s operative orthopaedics, ed 10, St Louis, 2003, Mosby.)
Pain.: Individuals with SCI must deal with a number of secondary complications in addition to any disability caused by the injury itself. Pain, weakness, and fatigue appear to be most common and most closely linked to individual social and mental health functioning.41 The number of reported pain sites increases with time, regardless of level or completeness of injury. While physical independence, mobility, and social integration remained relatively stable despite increasing numbers of pain sites, increases in depressive symptoms are associated with increased pain. Smokers with SCI report more pain sites than their nonsmoking counterparts.61
Pain caused by irritation of the nerve root is common, especially in cauda equina injury. Dysesthesia, impairment of sensation usually perceived as pain, can occur in areas with sensory loss and is often described as burning, pins and needles, or tingling. Disturbances of proprioception are related, and the person feels that a limb is in a different position than it is.
Musculoskeletal pain can result from faulty posture and overuse of limbs. Joint, ligament, and tendon deterioration is common, and secondary injury from muscle imbalances can come from functioning on the edge. Proper strength training can reduce these injuries and lead to more independence.
Fatigue.: Complaints of fatigue, noted to be higher than in the general population, may be associated with changes in several systems. ANS changes with inadequate sweating and thermoregulation can cause activity intolerance. Psychologic well-being is associated with fatigue; depression and decreased community mobility can be predicted by increasing complaints of fatigue.
Respiratory Complications.: Respiratory complications associated with spinal cord lesions can be life-threatening. Pulmonary complications are a common cause of death in both the acute and chronic phases. Spinal shock and poor management of edema can reduce vital capacity.
Lesions above C4 result in paralysis of muscles of inspiration and generally require artificial ventilation because of loss of the phrenic nerve innervation. Pulmonary complications with lesions at C5 through T12 arise as a result of loss of innervation of the muscles of expiration, the abdominal and intercostal muscles. The position of the diaphragm is compromised, and the abdominal musculature is unable to exert pressure during forced expiration. Paralysis of the external oblique muscles also inhibits the person’s ability to cough and expel secretions.
An altered breathing pattern develops in conjunction with the loss of the diaphragmatic muscle, the intercostal muscles, and the accessory muscles of inspiration. The upper chest wall flattens and the abdominal wall expands, leading to musculoskeletal changes in the trunk.
Aspiration and pneumonia occur frequently in individuals with SCI and is usually associated with high-level injuries and complete lesions and advanced age. Pneumonia is the most common cause of death, especially in the period immediately after the injury. With no other complications, proper rehabilitation, and stable respiration, the death rate from pneumonia matches that in the general population.60
Cardiovascular Conditions.: Cardiovascular conditions, including deep vein thrombosis and pulmonary embolism, are associated with SCI because of increased coagulability of blood and decreased venous return. This may be associated with sympathetic dysfunction and unopposed vagal action. For long-term SCI, morbidity and mortality from cardiovascular causes now exceeds that caused by renal and pulmonary conditions, the primary causes of mortality in previous decades. Risk of cardiovascular involvement comes from a greater prevalence of obesity, lipid disorders, metabolic syndrome, and diabetes. Daily energy expenditure is significantly lower in individuals with SCI, not only because of a lack of motor function, but also because of a lack of accessibility and fewer opportunities to engage in physical activity. Autonomic dysfunction caused by SCI is also associated with several conditions that contribute to heightened cardiovascular risk, including abnormalities in blood pressure, heart rate variability, arrhythmias, and a blunted cardiovascular response to exercise that can limit the capacity to perform physical activity.56
Metabolic Conditions.: Persons with SCI are prone to abnormal carbohydrate metabolism and are found to develop hyperinsulinemia and insulin resistance. During the acute phase of SCI, there is significant weight loss, especially with tetraplegia, associated with increased metabolic demands, muscle atrophy, and a negative nitrogen and calcium balance. Hypoproteinemia can be caused by pressure ulcers. Over time, there is usually an increase in body fat in proportion to lean tissue in the person with chronic SCI. A more sedentary lifestyle as a result of SCI may predispose a person to some of these conditions.
Soon after SCI, bones start losing minerals and become less dense. This may be due to alteration of the ANS and circulatory system. Inactivity and lack of weight bearing also foster the development of osteoporosis. It is believed that individuals with SCI may have an earlier onset and a greater extent of osteoporosis and that it may affect different body regions. There is a greater risk of fracture with osteoporosis.
Pressure Ulcers.: Pressure ulcers are a frequent complication of SCI. They arise primarily because of the pressure associated with lack of mobility and resulting pressure in the area of a bony prominence. Moisture, poor nutrition, complete lesions, acute illness, and cigarette smoking predispose the skin to breakdown. Persons who do not follow through on self-care requirements because of depression, lack of motivation, substance abuse, or alcoholism are also prone to develop more and deeper pressure ulcers. Initially, the sacrum, heel, and scapula are the most common sites of ulcer formation because of time spent in bed. As the individual begins to use a chair for mobility, the trochanter and ischium become common sites of pressure ulcers.75
Bowel and Bladder Control.: Bowel and bladder control is always affected in the person with SCI. The spinal center for urination is the conus medullaris. Primary reflex control originates from the sacral segment. During the stage of spinal shock, the urinary bladder is flaccid. All muscle tone and bladder reflexes are absent. Lesions above the conus medullaris will cause a reflex neurogenic bladder, reflected by spasticity, voiding difficulties, detrusor muscle hypertrophy, and urethral reflux. Lesions at the conus medullaris cause nonreflex bladders, resulting in flaccidity and decreased tone of the perineal muscles and urethral sphincter. Bowel patterns mimic bladder responses in their response to spinal shock; reflex bowel occurs in lesions above the conus medullaris, and nonreflex bowel is caused by damage to the conus and cauda equina.47 Fig. 34-13 shows bladder reflex pathways.
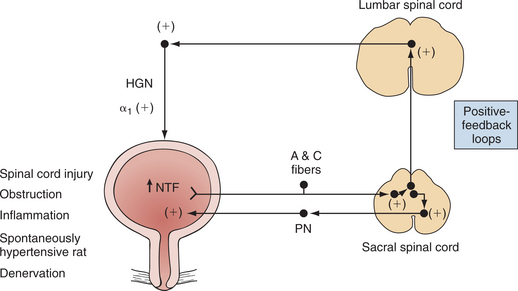
Figure 34-13 Possible mechanisms underlying spasticity in bladder reflex pathways induced by various pathologic conditions. Bladders from rats with chronic spinal cord injury exhibit increased level of neurotrophic factors (NTFs), such as nerve growth factor. NTFs can increase the excitability of C-fiber bladder afferent neurons and alter reflex mechanisms in parasympathetic excitatory pathways in the pelvic nerve (PN) as well as in sympathetic pathways in the hypogastric nerve (HGN). These reflex circuits are organized in the spinal cord as positive-feedback loops that induce involuntary bladder activity. (From Wein AJ, Kavoussi LR, et al: Campbell-Walsh urology, ed 9, Philadelphia, 2007, Saunders.)
Urinary tract infection is the most frequent secondary medical complication seen in persons with SCI. This persists despite improved catheter materials and design and use of antibiotics. In concert with this is the increased concentration of calcium in the urinary system, which leads to formation of kidney stones. Calculi in the kidney are a complication found more frequently in individuals using an indwelling catheter.14
Sexuality.: Sexual response is directly related to the level and completeness of injury. Sexual function relies on nervous pathways similar to those of the bladder and bowel and is altered as described earlier. There are two types of responses: reflexogenic, or a response to external stimulation seen in persons with upper motor neuron lesions, and psychogenic, a response that occurs through cognitive activity such as fantasy, associated with lower motor neuron lesions. Men with higher-level lesions can often achieve a reflexive erection but typically do not ejaculate. Those with lower lesions can more easily ejaculate, but achieving an erection is more difficult. With cauda equina lesions, erection and ejaculation are not usually possible. The primary reason for pursuing sexual activity is for intimacy needs, not fertility. Bladder and bowel concerns during sexual activity are not strong enough to deter the majority of the population from engaging in sexual activity. In addition, the occurrence of AD during typical bladder or bowel care is a significant variable predicting the occurrence and distress of AD during sexual activity.2 Menses are typically interrupted for approximately 3 to 6 months; when restored, they can be another cause of AD. Fertility and pregnancy are uninterrupted, but the pregnancy must be observed closely, especially in the last trimester. Labor may begin without the woman’s knowing it because of loss of sensation, and labor may initiate AD.64
Sleep Disorders.: The prevalence of obstructive sleep apnea–hypopnea syndrome (OSAHS) is high after cervical cord injury. OSAHS is characterized by repeated oxygen desaturation. OSAHS should be suspected especially in individuals with daytime sleepiness, obesity, and frequent awakenings during sleep.49 The changes in heart rhythm associated with OSAHS include sinusal arrhythmia, severe bradycardia, and ventricular and supraventricular tachycardia. The risk of sudden death, particularly of cardiovascular cause, is well known.16
MEDICAL MANAGEMENT
Delayed recognition of SCI is a significant problem in emergent care of traumatic injuries, occurring in more than 20% of cases.
Lateral film studies with plain radiographs are a rapid and effective way of evaluating cervical SCI, with the ability to detect approximately 85% of such injuries. When the open-mouth odontoid view and supine anteroposterior view are added, the accuracy rises to almost 100%. Any area that is inadequately demonstrated in the three-view spinal series is examined by computed tomography (CT). Flexion-extension studies are used primarily to evaluate instability caused by occult ligamentous injury and should not be done if there is any neurologic, bony, or soft tissue injury. CT demonstrates soft tissue structures and allows visualization of the bony limits of the spinal canal in the axial plane. CT is superior to other diagnostic procedures in demonstrating impingement on the neuronal canal.19
Fig. 34-14 shows a Jefferson fracture seen on x-ray and magnetic resonance imaging (MRI) scan. Fig. 34-15 compares fractures at time of injury and after stabilization. Myelography is indicated for optimal visualization of compression of the spinal cord after trauma. Myelography alone is rarely indicated, and it is used in conjunction with CT. In many cases, MRI has replaced myelography.
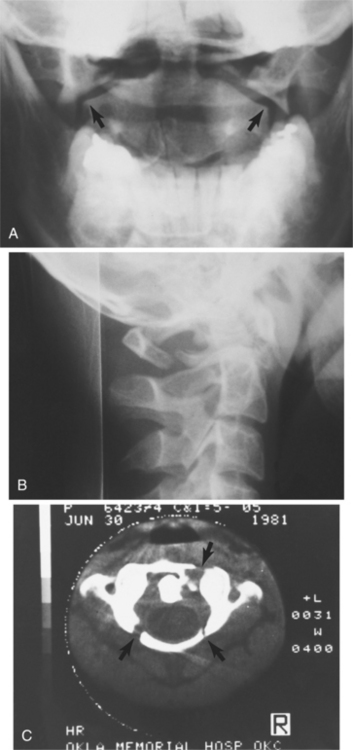
Figure 34-14 Fracture of C1 (Jefferson fracture). A, Anteroposterior view showing lateral displacement of the lateral mass and articulating facets of C1 on C2. B, Oblique view illustrating disruption of the posterior aspect of the ring of C1. C, Computed tomographic scan revealing the true extent of the injury. (From Green NE, Swiotkowski MF, eds: Skeletal trauma in children, ed 3, Philadelphia, 2003, Saunders. Courtesy of Dr. Teresa Stacy.)
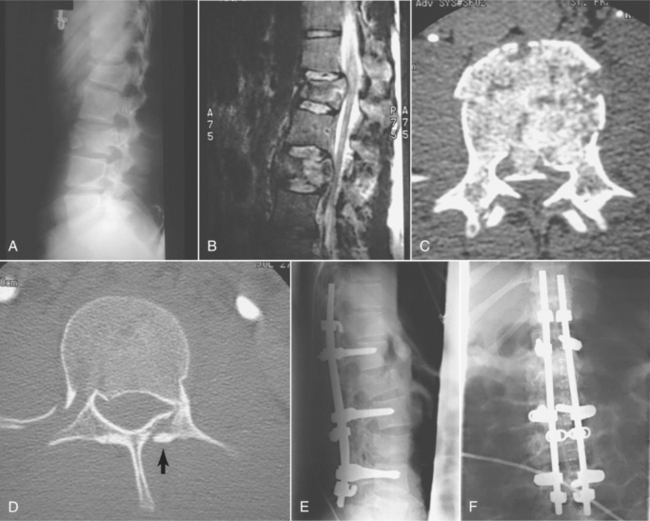
Figure 34-15 A 21-year-old man involved in a motor vehicle accident sustained a burst fracture of L1 and L3. The patient had an incomplete spinal cord injury. A, A preoperative lateral view shows loss of height predominately at L1. B, A sagittal-cut magnetic resonance image shows compression at both L1 and L3. C, An axial-cut computed tomographic (CT) scan at L3 shows a retropulsed fragment filling half the canal. D, An axial CT scan at L1 shows a fracture of the lamina and retropulsion of a fragment into canal. E, This injury was stabilized with Isola instrumentation combining both pedicle screws and laminar hooks. Sagittal alignment was maintained. F, Postoperative anteroposterior radiograph showing a cross-connection added for additional stability. (From Browner BD, Jipiter JB, Levine AM, et al: Skeletal trauma: basic science, management, and reconstruction, ed 3, Philadelphia, 2003, Saunders.)
In acute SCI, MRI is sometimes problematic because of its limited use around ferromagnetic objects such as respirators, oxygen tanks, and traction devices. When these obstacles do not exist, the extent of spinal cord damage and the possibility of disk herniation can be more readily assessed by MRI. The presence of intradural or extradural hematoma can often be demonstrated on MRI. MRI is useful in excluding spinal cord contusion or hemorrhage in persons with neurologic deficits and normal CT scans and plain films.21
MRI is insensitive to signals in the chronically injured cord because of lack of inflammation, hemorrhage, and swelling. The recent development of advanced structural imaging techniques, such as diffusion tensor imaging (DTI) and magnetization transfer (MT), is beginning to overcome these problems. In fact, lack of inflammation is advantageous for these types of images. DTI provides a unique image contrast called diffusion anisotropy. This reveals tissue organization at the microscopic level based on the average motion of water molecules.7
Advanced MRI techniques are better than conventional MRI in visualizing chronic SCI. The development of functional MRI (fMRI), currently useful in imaging the brain, and its application to the spinal cord should revolutionize the field of regeneration. To date, translation of brain fMRI techniques to the spinal cord has been hampered by the spinal cord’s small size and mobility, and artifacts induced by pulse of CSF flow. Understanding recent advances in MRI technology and imaging is important to the discussion of regeneration. Transplanted cells can now be tracked after they are placed in living organisms using MRI. Neural stem cells are labeled with paramagnetic agents before they are transplanted, and MRI tracks their distribution and migration after they are placed in the damaged spinal cord.
In spinal trauma with severe neural malfunction, neurophysiologic studies help in determining which neural elements are involved, which spinal segment is responsi- ble for mechanical or other irritation, and whether the lesion is chronic, acute progressing, or resolving. Neurophysiologic studies allow intraoperative monitoring and include somatosensory evoked potentials (SEPs), motor evoked potentials (MEPs), neurography, F-wave and H-reflex electromyography (EMG), and sympathetic skin response (SSR). SEPs and MEPs are useful in the investigation of the CNS. EMG, neurography, and F-wave and H-reflex studies are used for evaluation of the peripheral component of spinal injury.79
A unique SCI syndrome, burning hands syndrome, was first described in sports injury. This syndrome appears to be a variation of central cord syndrome associated with severe burning paresthesias and dysesthesias in the hands and/or the feet. Other signs of neurologic dysfunction are minimal or absent. Over 50% of the time there is an underlying spinal fracture-dislocation. It is important to differentiate this syndrome from the much more common and usually innocuous “burning” or “stinging” of brachial plexus origin. Box 34-3 outlines the combined evaluation of head and neck injuries that should be performed beginning at the sideline.30
Diagnosis of syringomyelia may be delayed because the early symptoms are so similar to those of other, more common neurologic disorders. The accident that caused the initial damage may have been months to years previous to the onset of symptoms. MRI is the best imaging study for diagnosing this disorder in the beginning stages. MRI will show the syrinx number, size, and location, and any other abnormalities of the spine and spinal cord. Functional MRI allows the surgeon to see the spinal fluid pulsating within the syrinx.
TREATMENT.
Interventions at several levels are required to improve mortality, morbidity, and quality of life. This begins with early surgical stabilization and with pharmacologic treatments aimed at blocking excitotoxicity and apoptosis. Prevention of the delayed wave of cell death that occurs in the weeks following injury is critical at the secondary level. Once injury is complete, the focus shifts to promoting regeneration. Pharmacologic treatments and transplantation are paired with the appropriate physical activity in order to optimize regeneration. It is now clear that spontaneous regeneration can be facilitated, and there is potential for optimizing regeneration even long after the injury and possibly extending throughout the remaining lifespan.
Emergent Care.
The emergent phase of care is crucial for the person with traumatic SCI. It can make the difference between living the rest of life with a disability or recovering with only temporary neurologic deficits. An incomplete injury can be made worse by mishandling and can be made better by prompt attention to critical procedures. Box 34-4 describes guidelines for essential trauma care.11,30
Assessment of the likelihood of SCI includes understanding the mechanics of the trauma and obtaining vital signs to determine if the individual is in neurogenic shock. Movement of the distal components of the body reflects the intactness of the spinal cord. In the case of a cervical injury, paradoxic respiration or abdominal breathing may be present, and immediate immobilization should be instituted. Use of a rigid collar and spinal board can help to prevent movement of the spinal column. Oxygen and medication should be given to control the hyperperfusion and swelling of the spinal cord. Transport should be swift, with care taken to avoid physical jarring caused by an uneven road surface and sudden stops.12,78
Monitoring in the critical care phase includes cardiac and neurologic status. Orthopedic management may begin at this phase and includes closed and open reduction of the vertebrae and decompression of the spinal cord. Insertion of a halo is common in treatment of cervical injuries, and a halo can be used without surgical intervention. In the more than 50% of individuals with SCI requiring surgery, fusion and internal fixation is the most common procedure performed. The goal is to restore spinal alignment, establish spinal stability, and prevent further neurologic deterioration, enhancing recovery. Blood pressure dysregulation in persons with SCI may reflect increased vascular nitrous oxide. Treatment of hypotension using nitrous oxide inhibition has been shown to be effective in this population.71
Pharmacologic control of edema, blood flow, and secondary neurologic sequelae shows promise in current studies. Corticosteroids such as methylprednisolone have shown good results in large doses, but the side effects must be considered. MRI findings suggest that methylprednisolone therapy in the acute phase of SCI may decrease the extent of intramedullary spinal cord hemorrhage.50 Although the concern about steroids continues, methylprednisolone should be administered to individuals with incomplete cervical SCI according to the Second National Acute Spinal Cord Injury Study protocol.69
Drugs that block opiate receptors appear to protect the spinal cord by decreasing endotoxic and hemorrhagic shock. Preservation of spinal cord function can be achieved by modulation of the neurotransmitters that are produced when there is injury to the nervous system. The dose-response curve remains narrow in most cases.76
When considering the appropriate type and dose of medication to be given to a person with an acute SCI, the degree of SCI must be assessed accurately. A moderate injury may require a different set of extracellular substances than that required in the secondary damage management for a severe injury.34
Management of Complications.
Management of complications of SCI is critical. High cervical injuries require immediate placement of ventilation equipment and maintenance of pulmonary hygiene. Therapy consists of intermittent positive pressure breathing (IPPB), bronchodilators, and mucolytics. Prevention of pulmonary infection is critical in SCI.
In electrophrenic respiration the phrenic nerve is implanted with a single electrode, and the client carries a transmitter to activate the lung. This works well in persons with high cervical lesions and sparing of C3 to C5 anterior horn cells. This method of respiration more closely mimics normal physiology than does IPPB.12
Loss of ANS control affects the function of the cardiovascular system. Acute management of blood pressure is critical. The autonomic lesion predisposes persons with high spinal cord lesions to abnormal cardiovascular responses to vasoactive agents.
Treatment of spasticity includes use of muscle relaxants and spasmolytic agents.63 Compared with oral baclofen, intrathecal baclofen infusion does not affect respiratory function and results in improved sleep continuity. Intrathecal baclofen infusion in therapeutic doses acts at the spinal level rather than at the supraspinal level.9 Sustained-release fampridine is effective in individuals with chronic SCI.13 One of the things that should be considered in the use of these medications is the fact that baclofen profoundly inhibits cell proliferation, survival, and differentiation, particularly myelination. Baclofen produces an irreversible loss of function. Baclofen is not an extremely effective oral agent, and there is now evidence that it can harm by inhibiting regeneration and recovery. Current research has shown that patterned activity, such as functional electrical stimulation (FES) bicycling (three times per week for 1 hour each time) may be a better way to control spasticity than medications.7 Peripheral nerve blocks, such as botulinum toxin (Botox), provide a temporary reduction of spasticity. If there is long-term, severe spasticity, the contractile potential of the muscle can be modified by surgery. Therefore, spasticity-related interventions need to be aimed at what matters most to the individual. It is critical for clinicians to understand individuals’ experiences to make accurate assessments, effectively evaluate treatment interventions, and select appropriate management strategies.52,53
Pain Management.
Despite the fact that SCI causes loss of sensation, there is often significant pain that develops over time. Pain in SCI is classified in many different ways associated with intrinsic or neurogenic dysfunction, such as pain associated with syringomyelia or central cord pain, peripheral nerve pain, and musculoskeletal or mechanical pain. Psychogenic pain is also addressed in some classifications.
Management of neurogenic pain in SCI is by systemic or local drug therapy and by neuroaugmentative and neurodestructive intervention. The pharmacologic approach includes nonsteroidal analgesics, opioids, antidepressants, and anticonvulsants.38 Pregabalin (Lyrica) is associated with relief of central neuropathic pain and with reduction in pain-related sleep interference and significant improvement in sleep problems. Action on centrally located calcium channels may be important in the effectiveness of pregabalin in managing central neuropathic pain.
Neuroaugmentative procedures include transcutaneous electrical nerve stimulation (TENS), epidural spinal stimulation, and central thalamic stimulation. Neurodestructive procedures include both chemical and surgical destruction of nervous structures. Procedures may include deafferentation, interruption of ascending pain systems, or destruction of cells in the dorsal horn.
Diffuse, chronic, and dysesthetic pain following SCI has been described by several authors using different terms. Dysesthetic pain syndrome is difficult to treat. This pain is distinguished by its quality and is usually described using words such as burning, stabbing, crushing, pressing, or pounding, referred to as allesthesia and allodynia, and can be called the “central Tinel” sign. Light touch or tapping over areas rostral to the level of injury can be painful, and pain can be triggered by nonnoxious stimuli such as movement of the bed. It can also be triggered by noxious stimuli, such as smoking or gastrointestinal disturbances.74
Suburothelial injection of botulinum A toxin can effectively inhibit the occurrence of neurogenic detrusor overactivity, providing increased bladder capacity and improved incontinence grade in individuals with SCI. The therapeutic effect declines gradually after 3 months, and all symptoms return within 6 months.
Decompressive surgery is performed in individuals with spinal cord syringomyelia, depending on which area is affected. Surgery to create a pseudomeningomyelocele, an artificial CSF reservoir, performed to normalize the CSF flow, has been shown to be effective. By draining the cyst, it is possible to prevent the cyst from reexpanding. Draining the fluid can relieve pain, headache, and a sensation of tightness in the head or neck. In a dural graft procedure, the space around the spinal cord is enlarged to allow free flow of fluid and reduce pressure.
Strategies for Spinal Cord Repair.
Nervous system repair is now feasible, and there is much research related to various strategies. A small degree of regeneration can result in recovery of function. A substantial loss of spinal cord tissue does not preclude function based on corticospinal tracts. There are two main regenerative approaches that are currently being studied: (1) optimizing spontaneous regeneration to restore function, and (2) transplanting stem cells. At this time there is no treatment that can clearly aid regeneration, but given the following information, primarily based on animal studies, it is likely that it will be developed in the near future.7
Embryonic stem cells are true stem cells that show unlimited capacity for self-renewal. In contrast, adult stem cells are progenitor cells or cells that are immature or undifferentiated. Their capacity for unlimited self-renewal and plasticity has not been as comprehensively demonstrated. Most progenitor cells are dormant or possess little activity in the tissue in which they reside. They exhibit slow growth, and their main role is to replace cells lost by normal attrition. Upon tissue damage or injury, progenitor cells can be activated by growth factors or cytokines, leading to increased cell division important for the repair process. Progenitor cells participate in the normal maintenance of the CNS. These mechanisms include production and replacement of cells lost to normal aging and cell turnover.
Critical components of optimal care following SCI are protection of neural tissue and limitation of secondary damage, facilitation of axonal regrowth, and control of factors that inhibit intrinsic neural repair. Because the consequences of SCI are complex, it is most likely that a hierarchy of intervention strategies will be needed to restore suprasegmental control leading to the recovery of function in the spinal cord.8,35 The phases of injury and the neurophysiologic events will create both limitations and advantages related to potential treatments administered during the different phases of injury. For example, progenitor cells transplanted during the acute injury phase are vulnerable to the same set of cell death mechanisms predominant during the secondary phase of acute injury. Understanding the function of growth-inhibiting factors within the adult CNS may lead to control of the neural destruction after SCI.9
Improving regeneration of axons after SCI has been attempted by transplantation of various cell types. Success has been achieved in creating and sustaining function in both neurons and glial cells. Two CNS areas have shown the most response, the olfactory bulb and the hippocampal dentate gyrus. However, there has been some limited success in other area of the brain.51
Astrocytes can express molecules that are both growth permissive and growth inhibitory at the same time. Reactive astrocytes appear to create an inhibitory environment within the injured spinal cord and form an astroglial scar that acts as a physical and chemical barrier to axonal regeneration. Maintaining an environment to support the growth of axons may involve the selective removal of astrocytes from the site of injury.20,67 In contrast, glial restricted precursors (GRP)-derived astrocytes (GDAs) may promote axonal regeneration via suppression of astrogliosis, realignment of host tissues, and delaying of expression of inhibitory proteoglycans. The glia, not the neurons, are the critical elements in preventing growth and in restoring it. Neurons retain the power to grow, and their sprouts only await the provision of a suitable glial pathway to be able to advance across the lesion.
Inflammatory reactions in the CNS have a dual nature; they may be neuroprotective as well as neurotoxic. Studies illustrate that the nervous and immune systems have overlapping rules of organization and intercellular communication. As a result, both systems express a host of common cytokines and neurotrophic factors that regulate cell survival and function. These shared mediators enable the two systems to engage in cross-talk and may provide a molecular explanation for neuroprotective effects of inflammation, which have been observed in animal models of CNS inflammation and trauma. T cells and natural killer (NK) cells in the spinal cord of rats can reduce the extent of neuron injury after ventral root avulsion. Hippocampal neurogenesis can be restored in mice with SCI by the transfer of CNS-reactive T cells.
Remyelination of bare axons after nervous system injury can be promoted by injecting endogenous stem cells that can be mobilized to become oligodendrocytes by exogenously delivering growth factors such as NT-3 or BDNF.37 Schwann cells have been used to facilitate a permissive environment for the injured spinal cord to regenerate. Previous experiments have shown compressive mechanical stress to be important in stimulating the regenerative behavior of Schwann cells. Transplantation of highly permissive Schwann cell–enriched peripheral nerve grafts may enhance regeneration in SCI.22 Fig. 34-16 demonstrates the neuroprotective processes that may be integral within the immunomodulation.
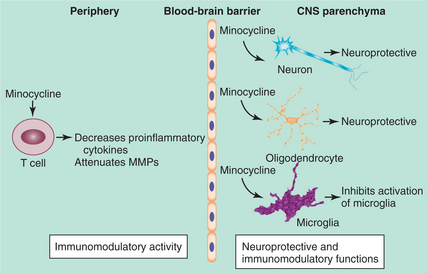
Figure 34-16 Peripheral and central functions of minocycline. Minocycline has immunomodulatory activity in the periphery and both immunomodulatory and neuroprotective capacity within the central nervous system. (From The Lancet Neurol ogy Volume 3, Issue 12, 2004, Fig. 3, Elsevier.)
Peripheral nerves may be able to provide an axonal bridge across the longer areas of spinal cord damage by activating nerve impulses carried from the brain, through intercostal nerve axons grown from implanted nerve, into the isolated distal end of the transected spinal cord, bridging the transection and connecting with neurons in the grey matter of the isolated distal segment of the spinal cord. It is possible that inferior-to-superior nerve bridging can produce return of function, just as superior-toinferior nerve bridging does. This is related to the concept that axons have a “relentless compulsion” to grow until they participate in return of function. Regenerationassociated genes (RAGs) are associated with developing neurons and as a response to CNS injury, are considered an important element for biochemical therapies driving regeneration of the axon.26 Electrical stimulation may provide an avenue for accelerated axonal outgrowth from the proximal nerve stump. (See Special Implications for the Therapist: Traumatic Spinal Cord Injury at the end of this chapter.)
A newly discovered cell line, hNT2.17, which expresses an exclusively neuronal phenotype, is a human-derived cell line that may be used to treat neuropathic pain and tactile and thermal allodynia, once it is further tested for safety and approved by the Food and Drug Administration. A dose of these human cells could be delivered with a spinal tap and affect the intrathecal spinal environment for sensory system modulation.24
Stem Cell Transplantation.
Transplantation of stem cells is being studied extensively in relation to the treatment of SCI. Processes include producing regenerative growth factors, expressing substances capable of breaking down scar tissue and modulating the immune system’s response to injury. It appears that there will be potential for reprogramming the host microenvironment; for example, embryonic stem cell transplantation reduces macrophage influx by more than 50%.
An immature CNS does not produce the same inhibitory effect on axon growth as the mature CNS does. After spinal cord lesions and transplantation of spinal cord tissue, there is extensive growth of descending axons into the transplants. A transplant of fetal spinal cord tissue may serve as a bridge to permit the regrowth of axons from spinal and supraspinal levels across the site of SCI.6 Transplants combined with neurotrophin treatment appear to have an additive effect compared with each intervention individually. Transplants and exogenous application of neurotrophic factors up-regulate cellular programs associated with regrowth and increase the extent of axonal regrowth with a favorable environment.5
Human umbilical cord blood stem cells (hUCB) hold great promise for therapeutic repair after SCI. Human embryonic stem cells (hESCs) may offer a renewable source of a wide range of cell types for use in research and cell-based therapies to treat disease. Cografted neural stem cells and NT-3 gene–modified Schwann cells promote the recovery of transected SCI and are a potential therapy for SCI. The grafted cells could enhance the survival of injured neurons in the inner pyramidal layer of sensorimotor cortex and could decrease the latency and increase the amplitude of cortical somatosensory and cortical motor evoked potentials, promoting limited structural and functional recovery.18,33 The CNS offers the benefit of transplantation into an immunologically privileged site.
PROGNOSIS.
The prognosis for recovery and repair depends on the phase of the injury and the age of the individual, with the best potential related to a younger age. The chronic consequences of SCI are related to maintenance as well as plasticity and repair. More than 90% of persons admitted to an acute care hospital for treat ment of SCI are ultimately discharged home. Morbidity and mortality during the first 4 weeks following SCI are most often related to paralysis of the respiratory muscles. The presence of either proteinuria with protein of 500 mg/day or greater is associated independently with increased mortality in the chronic SCI population.32
Long-term urinary tract infection continues to be a cause of death, but control of sepsis has improved markedly since 1970. This is primarily because of improvement in bladder training, antibiotic treatment, control of fluid intake, and surgery for obstruction of the lower urinary tract. Another common cause of death is respiratory disease, and this is the leading cause of death among high cervical injury clients. Pneumonia continues at a rate higher than in the general population, with pulmonary edema associated with injuries above T6. Heart disease is common, including myocardial infarction, cardiac arrest, myocarditis, and pulmonary embolism. However, the mortality rate is not much higher than that in the general population and is improving with increased pharmacologic control and improved medical knowledge of the cardiovascular changes accompanying SCI.
Most motor recovery occurs during the first 6 months, and strength can continue to increase with appropriate facilitation. The muscles graded 1 to 3 in the zone of partial preservation have potential to recover motor function. Overall, more than one half of the SCI population will have return of some neurologic function. Compression fractures have the most favorable prognosis for return of function, with crush fractures having the least chance for return of function.44 Skeletal complications are related to the deformity and degenerative changes associated with nonuse of extremities. These can lead to pain and further neurologic compromise.
Prognosis related to mobility is a concern to most persons, especially those with thoracic-level injury. Preservation of axonal integrity and regrowth of neural tissue will have a significant effect on the recovery of mobility after SCI. Turning this nervous system recovery into improved functional status is part of current and ongoing research in the rehabilitation field.
People with SCI experience significant problems in a number of areas of life, resulting in ongoing stress related to pain, lack of income and money problems, spasticity, stress and worries, and difficulty in their sex lives. These problems do not appear to be highly correlated with aging, suggesting that they will not necessarily become more problematic, nor are they likely to self-remediate.45
References
1. Altrice, MB, Morrison, SA, McDowell, SL, et al. Traumatic spinal cord injury. In: Umphred DA, ed. Neurological rehabilitation. ed 5. St Louis: Mosby-Year Book; 2007:605–657.
2. Anderson, KD. The impact of spinal cord injury on sexual function: concerns of the general population. Spinal Cord. 2007;45(5):328–337.
3. Ballaz, L. Acute peripheral blood flow response induced by passive leg cycle exercise in people with spinal cord injury. Arch Phys Med Rehabil. 2007;88(4):471–476.
4. Basso, DM. Neuroanatomical substrates of functional recovery after experimental spinal cord injury: implications of basic science research for human spinal cord injury. Phys Ther. 2000;80:808–817.
5. Beattie, MS, Bresnahan, JC. Cell death, repair, and recovery of function after spinal cord contusion injuries in rats. In: Kalb RG, Strittmatter SM, eds. Neurobiology of spinal cord injury. Totowa, NJ: Humana Press; 2000:1–21.
6. Behrman, AL, Bowden, MG, Nair, PM. Neuroplasticity after spinal cord injury and training: an emerging paradigm shift in rehabilitation and walking recovery. Phys Ther. 2006;86:1407–1423.
7. Belegu, V, Oudega, M, Gary, DS, et al. Restoring function after spinal cord injury: promoting spontaneous regeneration with stem cells and activity-based therapies. Neurosurg Clin North Am. 2007;18(1):143–168.
8. Bensmail, D. Effect of intrathecal baclofen on sleep and respiratory function in patients with spasticity. Neurology. 2006;67(8):1432–1436.
9. Bregman, BS. Transplants and neurotrophins modify the response of developing and mature CNS neurons to spinal cord injury. In: Kalb RG, Strittmatter SM, eds. Neurobiology of spinal cord injury. Totowa, NJ: Humana Press; 2000:169–194.
10. Brotherton, SS. Falls in individuals with incomplete spinal cord injury. Spinal Cord. 2007;45(1):37–40.
11. Buchanan, LE. Acute care: medical/surgical management. In: Buchanan LE, Nawoczenski DA, eds. Spinal cord injury. Baltimore: Williams & Wilkins; 1987:35–60.
12. Buchanan, LE. Emergency care. In: Buchanan LE, Nawoczenski DA, eds. Spinal cord injury. Baltimore: Williams & Wilkins; 1987:21–34.
13. Cardenas, DD. Phase 2 trial of sustained-release fampridine in chronic spinal cord injury. Spinal Cord. 2007;45(2):158–168.
14. Cardenas, DD, Farrell-Roberts, L, Sipski, ML, et al. Stover SL, DeLisa JA, Whiteneck GG, eds. Spinal cord injury: clinical outcomes from the model systems. Aspen Publishers: Gaithersburg, MD, 1995:120–144.
15. Carpenter, C. Community participation after spinal cord injury. Arch Phys Med Rehabil. 2007;88(4):427–433.
16. Catarino, A. Obstructive sleep apnea-hypopnea syndrome (OSAHS) and changes in heart rhythm. Rev Port Pneumol. 2003;9(5 suppl 1):50–51.
17. Chang, HT. Subacute human spinal cord contusion: few lymphocytes and many macrophages. Spinal Cord. 2007;45(2):174–182.
18. Choong, C. Human embryonic stem cells. Neurosurg Clin North Am. 2007;18(1):1–14.
19. Daffner, RH. Imaging of thoracic and lumbar vertebral fractures. In: Floman YF, Farcy JC, Argenson CA, eds. Thoracolumbar spine fractures. New York: Raven Press; 1993:69–98.
20. Dasari, VR. Axonal remyelination by cord blood stem cells after spinal cord injury. J Neurotrauma. 2007;24(2):391–410.
21. Davis, P, Rao, KC. Spine trauma. In: Rao KC, Williams JP, Lee BC, et al, eds. MRI and CT of the spine. Baltimore: Williams & Wilkins; 1994:277–346.
22. Dinh, P. Transplantation of preconditioned Schwann cells following hemisection spinal cord injury. Spine. 2007;32(9):943–949.
23. Dimitrijevic, MM, Dimitrijevic, MR. Clinical practice of functional electrical stimulation. In: Illis LS, ed. Spinal cord dysfunction vol 2, Intervention and treatment. Oxford, England: Oxford University Press; 1992:168–174.
24. Eaton, MJ. Subarachnoid transplant of a human neuronal cell line attenuates chronic allodynia and hyperalgesia after excitotoxic spinal cord injury in the rat. J Pain. 2007;8(1):33–50.
25. Eismont, FJ, Lattuga, S. Gunshot wounds of the spine. In Jupiter BD, Jupiter JB, Levine AM, et al, eds.: Skeletal trauma: basic science, management, and reconstruction, ed 3, Philadelphia: Saunders, 2003.
26. Farcy, JC, Rawlins, BA. Physiology and pathophysiology of the neural elements. In: Floman YF, Farcy JC, Argenson CA, eds. Thoracolumbar spine fractures. New York: Raven Press; 1993:35–44.
27. Fagan, K. Transient quadriplegia and return-to-play criteria. Clin Sports Med. 2004;23(3):409–419.
28. Field-Fote, EC, Lindley, SD, Sherman, AL. Locomotor training approaches for individuals with spinal cord injury; a preliminary report of walking related outcomes. J Neurol Phys Ther. 2005;29:127–167.
29. Gater, DR. Pathophysiology of obesity after spinal cord injury. Top Spinal Cord Inj Rehabil. 2007;12(4):20–34.
30. Ghafoor, AU. Caring for the patients with cervical spine injuries: what have we learned? J Clin Anesth. 2005;17(8):640–649.
31. Gorgey, AS. Skeletal muscle atrophy and increased intramuscular fat after incomplete spinal cord injury. Spinal Cord. 2007;45(4):304–309.
32. Greenwell, MW. Kidney disease as a predictor of mortality in chronic spinal cord injury. Am J Kidney Dis. 2007;49(3):383–393.
33. Guo, J. Cotransplant of neural stem cells and NT-3 gene modified Schwann cells promote the recovery of transected spinal cord injury. Spinal Cord. 2007;45(1):15–24.
34. Hansebout, RR. Spinal injury and spinal cord blood-flow: the effect of early treatment and local cooling. In: Illis LS, ed. Spinal cord dysfunction vol 2, Intervention and treatment. Oxford, England: Oxford University Press; 1992:58–78.
35. Heimburger, RF. Is there hope for return of function in lower extremities paralyzed by spinal cord injury? J Am Coll Surg. 2006;202(6):1001–1004. [discussion, 1004].
36. Hoffman, LR. Cortical reorganization following bimanual training and somatosensory stimulation in cervical spinal cord injury: a case report. Phys Ther. 2007;87(2):208–223.
37. Hohlfeld, R. Dual role of inflammation in CNS disease. Neurology. 2007;68(22 suppl 3):S58–S63.
38. Illis, LS. Central pain. In: Illis LS, ed. Spinal cord dysfunction vol 2, Intervention and treatment. Oxford, England: Oxford University Press; 1992:145–155.
39. Illis, LS. Spasticity: clinical aspects. In: Illis LS, ed. Spinal cord dysfunction vol 2, Intervention and treatment. Oxford, England: Oxford University Press; 1992:81–93.
40. Jain, NB. Factors associated with health-related quality of life in chronic spinal cord injury. Am J Phys Med Rehabil. 2007;86(5):387–396.
41. Jensen, MP. Symptom burden in persons with spinal cord injury. Arch Phys Med Rehabil. 2007;88(5):638–645.
42. Jurkiewicz, MT. Somatosensory cortical atrophy after spinal cord injury: a voxel-based morphometry study. Neurology. 2006;66(5):762–764.
43. Khan, S. Hypothermia in patients with chronic spinal cord injury. J Spinal Cord Med. 2007;30(1):27–30.
44. Kakulas, BA, Taylor, JR. Pathology of injuries of the vertebral column and spinal cord. In: Vinken PJ, Bruyn GW, Klawans HL, eds. Spinal cord trauma. Amsterdam: Elsevier Science Publishers; 1992:21–54.
45. Krause, JS. Self-reported problems after spinal cord injury: implications for rehabilitation practice. Top Spinal Cord Inj Rehabil. 2007;12(3):35–44.
46. Krause, JS. Pain interference in ambulatory spinal cord injury. Top Spinal Cord Inj Rehabil. 2007;12(3):91–96.
47. Kuo, HC. Therapeutic effects of suburothelial injection of botulinum A toxin for neurogenic detrusor overactivity due to chronic cerebrovascular accident and spinal cord lesions. Urology. 2006;67(2):232–236.
48. Lazar, RB. Posttraumatic syringomyelia. In: Yarkony GM, ed. Spinal cord injury: medical management and rehabilitation. Gaithersburg, MD: Aspen Publishers, 1994.
49. Leduc, BE. Estimated prevalence of obstructive sleep apnea-hypopnea syndrome after cervical cord injury. Arch Phys Med Rehabil. 2007;88(3):333–337.
50. Leypold, BG. The impact of methylprednisolone on lesion severity following spinal cord injury. Spine. 2007;32(3):373–378.
51. Love, R. Stem-cell derived astrocytes promote repair of spinal cord. Lancet Neurol. 2006;5(6):474–475.
52. Mahoney, JS. Spasticity experience domains in persons with spinal cord injury. Arch Phys Med Rehabil. 2007;88(3):287–294.
53. Masson, P. PDE-5 inhibitors: current status and future trends. Urol Clin North Am. 2005;32(4):511–525. [viii].
54. Matjacic, Z. Apparatus for dynamic balance training during treadmill walking. J Rehabil Med. 2007;39(1):91–94.
55. Mock, C. Advancing injury prevention and trauma care in North America and globally. Surg Clin North Am. 2007;87(1):1–19. [v].
56. Myers, J. Cardiovascular disease in spinal cord injury: an overview of prevalence, risk, evaluation, and management. Am J Phys Med Rehabil. 2007;86(2):142–152.
57. Nash, MS. Effects of circuit resistance training on fitness attributes and upper-extremity pain in middle-aged men with paraplegia. Arch Phys Med Rehabil. 2007;88(1):70–75.
58. Nawoczenski, DA, Rinehart, ME, Duncanson, P, et al. Physical management. In: Buchanan LE, Nawoczenski DA, eds. Spinal cord injury. Baltimore: Williams & Wilkins; 1987:123–184.
59. Rägnarsson, KT. Rehabilitation principles in the management of thoracolumbar spine fractures. In: Floman YF, Farcy JC, Argenson CA, eds. Thoracolumbar spine fractures. New York: Raven Press; 1993:463–485.
60. Ragnarsson, KT, Hall, KM, Wilmot, CB, et al. Management of pulmonary, cardiovascular and metabolic conditions after spinal cord injury. In: Stover SL, DeLisa JA, Whiteneck GG, eds. Spinal cord injury: clinical outcomes from the model systems. Gaithersburg, MD: Aspen Publishers, 1995.
61. Richardson, EJ. A longitudinal study of joint pain following SCI: concurrent trends in participation, depression, and the effects of smoking. Top Spinal Cord Inj Rehabil. 2007;12(3):45–55.
62. Shields, RK. Musculoskeletal adaptations in chronic spinal cord injury: effects of long-term soleus electrical stimulation training. Neurorehabil Neural Repair. 2007;21(2):169–179.
63. Schmitz, TJ. Traumatic spinal cord injury. In: Schmitz TJ, O’Sullivan SB, eds. Physical rehabilitation assessment and treatment. ed 3. Philadelphia: FA Davis; 1994:533–575.
64. Schneider, FJ. Traumatic spinal cord injury. In Umphred DA, ed.: Neurological rehabilitation, ed 2, St Louis: Mosby-Year Book, 1990.
65. Somers, MF. Spinal cord injury: functional rehabilitation. Norwalk, CT: Appleton & Lange, 1992.
66. Sprigle, S. Development of valid and reliable measures of postural stability. J Spinal Cord Med. 2007;30(1):40–49.
67. Steeves, JD. Strategies for spinal cord repair. In: Kalb RG, Strittmatter SM, eds. Neurobiology of spinal cord injury. Totowa, NJ: Humana Press; 2000:113–130.
68. Stoner, L. Electrical stimulation-evoked resistance exercise therapy improves arterial health after chronic spinal cord injury. Spinal Cord. 2007;45(1):49–56.
69. Tsutsumi, S. Effects of the Second National Acute Spinal Cord Injury Study of high-dose methylprednisolone therapy on acute cervical spinal cord injury-results in spinal injuries center. Spine. 2006;31(26):2992–2996.
70. Waxman, SG, deGroot, J. Correlative neuroanatomy, ed 22. Norwalk, CT: Appleton & Lange, 1995.
71. Wecht, JM. Normalization of supine blood pressure after nitric oxide synthase inhibition in persons with tetraplegia. J Spinal Cord Med. 2007;30(1):5–9.
72. Whalley Hammell, K. Experience of rehabilitation following spinal cord injury: a meta-synthesis of qualitative findings. Spinal Cord. 2007;45(4):260–274.
73. Williams, B. Post-traumatic syringomyelia (cystic myelopathy). In: Vinken PJ, Bruyn GW, Klawans HL, eds. Spinal cord trauma. Amsterdam: Elsevier; 1992:375–394.
74. Woodward, KG. The proximal tap or “central Tinel” sign in central dysesthetic syndrome after spinal cord injury. J Am Paraplegia Soc. 1991;14(3):136–138.
75. Yarkony, GM, Heinemann, AW. Pressure ulcers. In: Stover SL, DeLisa JA, Whiteneck GG, eds. Spinal cord injury: clinical outcomes from the model systems. Gaithersburg, MD: Aspen Publishers; 1995:100–119.
76. Young, W. Clinical trials and experimental therapies of spinal cord injury. In: Vinken PJ, Bruyn GW, Klawans HL, eds. Spinal cord trauma. Amsterdam: Elsevier; 1992:399–420.
77. Young, W. Neurophysiology of spinal cord injury. In: Errico TJ, Bauer RD, Waugh T, eds. Spinal trauma. Philadelphia: JB Lippincott; 1991:377–414.
78. Young, W. Pharmacologic therapy of acute spinal cord injury. In: Errico TJ, Bauer RD, Waugh T, eds. Spinal trauma. Philadelphia: JB Lippincott; 1991:415–434.
79. Young, W. Therapy for acute spinal cord injury. In: Illis LS, ed. Spinal cord dysfunction vol 2, Intervention and treatment. Oxford, England: Oxford University Press; 1992:28–58.
80. Zabel, RJ, Forest, B. Autonomic dysreflexia: an acute care emergency. Acute Care Perspect. 7(3), 1999.