Introduction to Central Nervous System Disorders
OVERVIEW
The central nervous system (CNS) controls and regulates all mental and physical functions. The nervous system is unparalleled among organ systems in terms of diversity of cellular constituents. It is composed of a network of neural tissue that includes both receptors and transmitters. There is a complex interaction among the areas that control different functions. A variety of neurons provide transmission of specific information throughout the nervous system. Disease or trauma of the CNS may affect the nervous system through damage to several types of tissues in a local area, such as in stroke, or it may cause dysfunction in one type of tissue throughout many areas of the CNS such as in multiple sclerosis. Dysfunction of the neurons in an area of the brain can disrupt the complex organization of firing, resulting in abnormal perception of the environment, uncoordinated movement, loss of force production, and decreases in cognition.
Behavior, including thought and movement, is shaped by the interplay between genes and the environment. There are genes that control entry into the cell cycle where cells synthesize DNA and undergo mitosis. Proliferation can be triggered by internal signals or in response to external growth factor stimulation. There is a complex spectrum of alterations produced by aging, disease, and neoplastic transformation. Neoplastic transformation, which is the basis of cancer, is characterized by mutations of genes regulating cell growth, differentiation, and death. A set of genes appears to inhibit cellular proliferation; these genes are the “brakes” of the cell cycle, and loss of these genes may lead to tumor growth.16
Inherited patterns of deoxyribonucleic acid (DNA) expression appear to cause a predisposition for neurologic disease and affect the ability to repair damage from an insult in the nervous system. Genetic information is stored in the chromosomes within each individual cell in the body; about 80,000 genes are represented and arranged in a precise order. More than one-third of the genes are expressed as messenger ribonucleic acid (RNA) in the brain, more than in any other part of the body. An anomaly or alternative gene version is referred to as an allele. Single-gene mutations or alleles have been identified and can be associated with degenerative neurologic disease such as Huntington’s disease. However, for most chronic disorders, there appear to be multiple abnormalities, and it is clear that environmental conditions have an effect on how the abnormality is manifested.2
Pathologic derangements of normal cellular processes are a way of looking at possible causes of disease. Diseases in which cells are lost are characterized as necrotic and apoptotic. Both necrosis and apoptosis underlie diseases as diverse as stroke, trauma, demyelinating disorders, infections, and neurodegenerative disorders.
PATHOGENESIS
Neuronal cell death is a hallmark of many disorders of the nervous system through the processes of necrosis and apoptosis. When cell death is caused by necrosis, there is cellular swelling, fragmentation, and cell disintegration. Necrosis causes the internal structure of the cell to swell as water enters the cell through osmosis and cell membranes to rupture. Lymphocytes and polynuclear cells can cause inflammatory cells to surround the necrotic debris, resulting in release of cytotoxic compounds and destruction of neighboring cells. Excitotoxicity results from the inappropriate activation of excitatory amino acid receptors leading to the entry of calcium ions into the cell. The calcium activates intracellular function. Damaged cells release excitotoxins that damage surrounding cells.16
Apoptosis is programmed cell death, or a type of cellular suicide, but apoptosis does not cause inflammatory responses. It is a more organized process with fragmentation of the cells and degradation of the DNA. It is common during the development of cells to eliminate the overproduction of one cell type. The biochemical pathway is present in all cells of the body and is used normally in the maturation and regulation of the nervous system with systematic removal of neurons from the brain. In apoptosis, the cell is removed by macrophages and leaves no residual damage to other components of the CNS. If the cell sustains genetic damage through neurodegenerative disease or injury and cannot be repaired by the system, the cell dies. Damage to the CNS can cause excessive apoptosis through the process of trophic factor withdrawal, oxidative insults, metabolic compromise, overactivation of glutamate receptors, and exposure to bacterial toxins.36 These processes are described in the next paragraphs.
The intensity of cellular injury determines whether the cell dies or is able to survive. Very severe injury leads to the passive process of necrosis, less severe but irreparable injury leads to the active process of apoptosis, and survivable injury leads to reactive changes such as gliosis or scarring.
Free radical formation is a by-product of excitotoxicity. Free radicals are capable of destroying cellular components and triggering apoptosis. Free radicals are molecules with an odd number of electrons. The odd, or unpaired, electron is highly reactive as it seeks to pair with another free electron. Free radicals are generated during oxidative metabolism and energy production in the body. Free radicals are related to normal metabolism but can be the cause of oxidative stress in brain injury and disease. Oxidative stress refers to cells and tissues that have been altered by exposure to oxidants. Oxidation of lipids, proteins, and DNA leads to tissue injury. Nitrogen monoxide (nitric oxide [NO]) is a free radical generated by NO synthase (NOS). This enzyme modulates physiologic responses, such as vasodilation or signaling, in the brain. Oxidative stress, rather than being the primary cause, appears to be a secondary complication in many progressive disorders, such as Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis (ALS), as well as disorders of mental status. An enhanced antioxidant status is associated with reduced risk of several diseases.44
The blood-brain barrier is made from endothelial cells and its tight junctions, so that substances can pass only through the cell and not in between cells. Drug entry into the CNS is determined by the drug’s lipid solubility. Glucose and amino acid cross the endothelial cell barrier via protein transporters.
The ependyma cells line the ventricles and spinal canal and regulate metabolism between the channels of the extracellular space and the ventricles. Ependyma forms the basis of the cerebral spinal fluid barrier. There is movement of molecules through the extracellular space with the possibility of long range and relatively diffuse actions of neurotransmitters released into the extracellular space. This type of signaling is known as volume transmission and may have a major role in setting large-scale neuronal excitability or inhibition.16
Dysfunction within the nervous system can affect either or both of the two main classes of cells: the glial cells and the neurons. Stem cells create new glia and new neurons. The region immediately beneath the ependymal cell layer produces new cells at a very low rate in the adult compared to the amount created during neurogenesis in early development. They migrate widely through the brain and conform phenotypically to the regions where they end up.
Glial Cells
Aside from neurons, macroglia and microglia are the two primary cell types located throughout the CNS. The macroglia are derived from a nerve cell lineage and are classified into three distinct subtypes: astrocytes, oligodendrocytes, and Schwann cells. These macroglia are the most populous cells of the CNS and support and maintain neuronal plasticity throughout the CNS. Glial cells are often implicated in the disease process that affects brain tissue.26
Microglia are the resident immune cells of the brain. Microglia also are interspersed throughout the brain and represent approximately 10% of the CNS population. Microglia differ from the macroglia because they are derived from a monocyte cell lineage. Microglia respond to CNS insult both by diffuse proliferation and infiltration of CNS tissue. Microglia are pivotal in innate immune activation and function to modulate neuroinflammatory signals throughout the brain. In the absence of stimulus, microglia are dormant.
During an immune response, microglia are activated. Inflammatory cytokines produced within the CNS target neuronal substrates, triggering a response of fever, increased sleep, reduced appetite, and lethargy. Collectively, these behavioral symptoms of sickness are evolutionarily conserved and function to increase the metabolic demand for clearance of pathogens via the microglia.20 Active microglia show macrophage-like activities, including scavenging, phagocytosis, antigen presentation, and inflammatory cytokine production. Microglia recruit and activate astrocytes to propagate these inflammatory signals further. Normally, these neuroinflammatory changes are transient and beneficial, with microglia returning to the dormant state after the resolution of the immune challenge. Aging, however, may provide a brain environment in which microglia activation is not resolved, leading to a heightened sensitivity to immune activation; this lack of resolution may contribute to the pathogenesis of neurologic disease.55
Activated microglia and monocytes coming in from the bloodstream can assume the form of macrophages, or giant multinucleated cells filled with ingested debris. Nearby neurons may be damaged by toxins released from activated macrophages and microglia.
Astrocytes are named because they look like star cells. They are the most numerous cells in the brain and outnumber neurons 10: 1. Fig. 28-1 shows the relationship of the glial cell to the neuron. The glial cells provide support and structure for the CNS and play the role that connective tissue performs in other parts of the body. The glial cells are active in the system but are not involved in signaling information. The neurons communicate information to one another in order to process sensory information, program motor and emotional responses, and store information through memory.
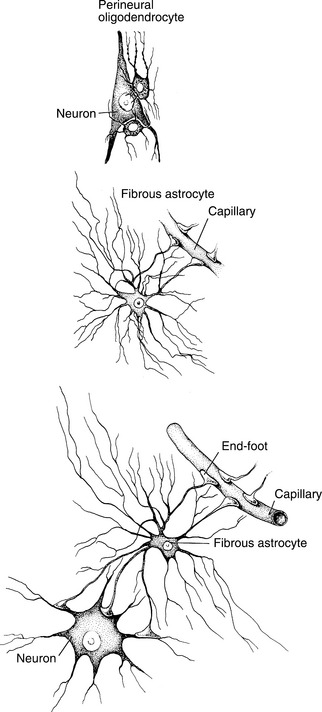
Figure 28-1 The relationship of the glial cells (astrocytes, oligodendrocytes) to the neurons and capillaries. (From Kandel ER, Schwartz JH: Principles of neural science, ed 2, New York, 1985, Elsevier.)
In addition to their support function, the cells serve a nutritive function, since they connect to the capillary wall and to the nerve cell. They may be responsible for the release of nerve growth factor. Astrocytes are permeable to potassium and therefore are involved in maintaining the correct potassium balance in the extracellular space. Astroglia have the ability to monitor and remove extracellular glutamate and other residual neuronal debris after brain injury and can seal off damaged brain tissue.26,52 When the astroglial cells become dysfunctional as part of an injury or degenerative process, it may reinforce neuronal damage. Astroglial changes are widely recognized to be one of the earliest and most remarkable cellular responses to CNS injury.42 Astrocyte swelling is a common pathologic finding and is often seen at the interface with the vascular system. The swelling may be a factor in gliosis, a reaction of the glial cells that produces tissue that is laid down in a scarlike manner, producing glial scarring.41 Astroglial cell tissue can be the site of neoplastic disorders that disrupt nerve cell function by compressing the neurons and blood supply in the surrounding area (see Chapter 30).
The two other glial cell types, the oligodendrocyte, a part of the CNS, and the Schwann cell found in the peripheral nervous system are responsible for the production of the myelin sheath, which surrounds the axon. (See Chapter 39 for information on the peripheral disorders that are associated.) Demyelinating disorders that target the CNS, such as multiple sclerosis, are often the result of disrupted function of the oligodendrocyte.1 This process is further described in the section on multiple sclerosis in Chapter 31. Figs. 28-2 and 28-3 show the oligodendrocyte and describe the process of myelination.41
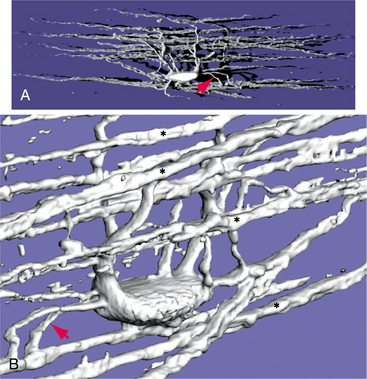
Figure 28-2 A, Single oligodendrocyte from a rat. B, More magnified view showing the process as they emerge from the cell body. (From Nolte J: The human brain: An introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby. Courtesy Dr. Peter S. Eggli, Institute of Anatomy, University of Bern, Bern, Switzerland.)
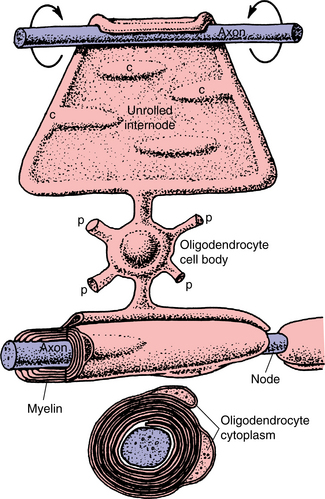
Figure 28-3 Schematic diagram of the formation of myelin in the central nervous system (CNS). (From Nolte J: The human brain: An introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby. Redrawn from Krsti é RV: Illustrated encyclopedia of human histology, Berlin, 1984, Springer-Verlag.)
Pain was classically viewed as being mediated solely by neurons, as are other sensory phenomena. Spinal cord glia amplify pain and are activated by certain sensory signals arriving from the periphery. These glia express characteristics in common with immune cells in that they respond to viruses and bacteria, releasing proinflammatory cytokines, which create pathologic pain. (See Chapter 7 for more information about interactions between the immune system and the CNS.) Altering glial function has become a new approach to pain control.64
Nerve Cells
There are many mechanisms of communication between nerve cells related to the structure and function of each cell type. The location in the nervous system, the input cells, and the target cells will determine how a cell communicates. The cell body size and the shape and configuration of dendrites and axons will also affect the method of communication. However, almost all neurons will typically fire through a manner that can be described schematically. Essentially, the chemical information encoded by a gene within one nerve cell is delivered to the appropriate postsynaptic genome through a series of molecular reactions.56 Information is transferred via electrical signals that travel along the neuron and is carried to the next neuron through a series of biochemical events that will influence the behavior of the second-order neuron.
The cell body of the neuron is the metabolic center of the neuron and includes the nucleus where the genetic material is located. The gene expressed in a cell directs the manufacture of proteins that determine the structure, function, and regulation of the neural circuits. Mutation, or changes in the structure of the DNA, can lead to the production of abnormal proteins that can be associated with vulnerability to neurologic disease. Abnormalities within the gene structure leading to predisposition for mutations can be inherited. Toxicity or abuse of drugs can also affect the ability of the DNA to replicate in a normal manner and can cause long-term dysfunction of the nervous system. Cell body inclusions are growths that occur with in the cell body as a part of aging, such as Lewy bodies, but can also be a part of the disease process and can cause loss of function of the cell as a result of the obliteration of the nucleus of the cell.44
The cell body generates electrical activity through action potentials. A transient increase in sodium perme- ability is the molecular foundation of the action potential. The increase in sodium permeability causes this ion to be dominant and establishes the membrane potential as +40 mV, or action potential. This is transient and soon closed as the potassium channels open and resting potential is restored.
Ion channels are proteins that span the cell membrane and are able to conduct ions through the membrane. The ion channels recognize and select specific ions for transfer. They are able to open and close in response to specific electrical, mechanical, or chemical signals. Fig. 28-4 describes the gating properties.41 Sodium channel blockers bind the outer axonal surface of the channel and prevent the flux of sodium. The nerve cells sequentially generate four different signals at different sites within the cell: an input signal, a trigger signal, a conducting signal, and an output signal. The input signal depolarizes the cell membrane. Dendrites are typically the site for receiving incoming signals from other neurons. It is in the trigger zone on the initial segment of the axon that the receptor signals are summed, and the neuron then fires an action potential through the length of the axon. The intensity of the conducting signals is determined by the frequency of individual action potentials. As the action potential reaches the neuron’s terminal, it stimulates the release of a chemical neurotransmitter cell through the presynaptic terminals.26 Fig. 28-5 shows the processes related to transmitter release.41
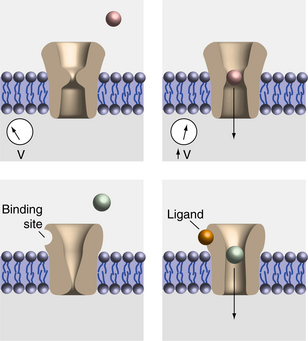
Figure 28-4 Ion channels respond to the changes in voltage. A represents the closed state, and B represents the open state that allows neurotransmitters to gain entry into the cell. C and D represent the opening based on the ligand attaching to the protein that causes the channel to open. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
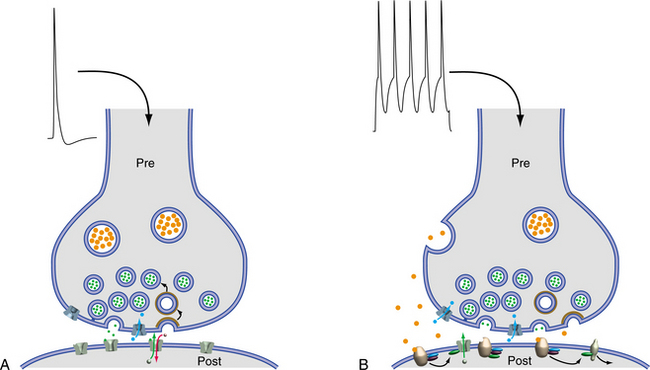
Figure 28-5 A, Depolarization of the terminal causes sodium influx and opening of the channels in the postsynaptic neuron. B, Release of transmitters from large and small vesicles, the status of the postsynaptic proteins will affect the binding capability. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
The axon serves as the entry route of a number of pathogens and toxins and presents a large target as a result of its large volume. Excitatory synapses are distributed distally in the dendritic receptive field, and inhibitory synapses exist in the proximal dendritic field or on the cell body. The combined firing creates modulation of input.
The axon of the nerve can selectively be damaged, without destruction of the cell body, causing a decrease or loss of presynaptic activity. The stretch damage to the axon is responsible for the abnormal or delayed firing associated with damage to the brainstem in head trauma. Axonal spheroid formation is a reaction to injury resulting in formation of axon retraction balls and can be seen in radiation necrosis and traumatic brain injury. Axon degeneration plays a part in multiple sclerosis.16
Neurotransmission
By means of its axonal terminals, one neuron contacts and transmits information to the receptive surface of another neuron. The release of neurotransmitter from the presynaptic terminal and the uptake of that substance in the postsynaptic receptor are known as a synapse. A simplified diagram is shown Fig. 28-6. Virtually all communication between neurons occurs via chemicals. The chemical communication involved in this process is universally known as either neurotransmission or neuromodulation.27 Changes in neurotransmitter substances in the space surrounding the neurons have been implicated in many nervous system disease processes.
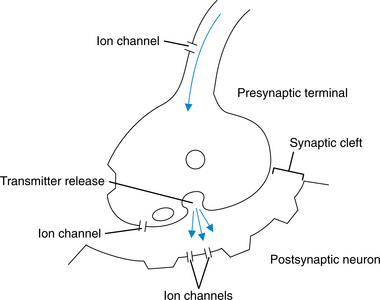
Figure 28-6 Schematic representation of the postsynaptic neuron and the presynaptic terminal. Transmitter substances are synthesized in presynaptic terminals, released into the synaptic cleft, and occupied in the postsynaptic terminal.
Neurotransmitters are synthesized within each neuron, stored in presynaptic vesicles, and released from depolarized nerve terminals. They bind specifically to presynaptic or postsynaptic receptors, which recognize the neurotransmitter’s chemical conformation. A single neuron can release several different neurotransmitter substances, and a single neuron can be selectively receptive to differ- ent types of neurotransmitters because of the differences in ion channels.57 Activation of a receptor in response to neurotransmitter can cause changes in a variety of molecules. Modification, or modulation, of the system can take place presynaptically, postsynaptically, or within the cell body.
Changes in the target cell can cause abnormal responses to normal levels of transmitters. The amount of neurotransmitter released in the synaptic cleft is determined by the neuronal firing rate, the quantity of transmitter in the nerve terminal, and the cumulative regulatory actions of excitatory and inhibitory neurotransmitters. These biochemical actions alter the electrical activity of the postsynaptic neurons.
One aspect of chemical transmission that is extremely important in signaling is the time course of transmitter in the synaptic cleft. The breakdown of a transmitter is an important variable and can change the concentration of the substance in the synaptic cleft.22 Control of the neurotransmitter in the synaptic cleft is the basis for pharmacologic treatment in degenerative neurologic disease. Fig. 28-7 diagrams the various ways that the substances in the synaptic cleft can be removed.41
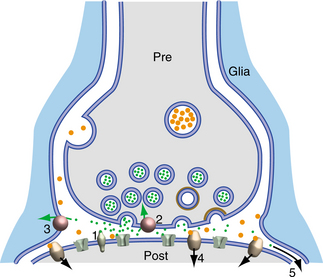
Figure 28-7 The transmitter substances can be removed by (1) enzymatic inactivation of neurotransmitter, (2) reuptake of the neurotransmitter by the presynaptic terminal, (3) removal by the nearby glial cells, (4) uptake by the postsynaptic terminal, or (5) it may just move out of the synaptic space into adjoining spaces. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
An important concept for all neurotransmitters is that the final result of either hyperpolarization or depolarization depends on both the transmitter and its receptor. The concept of an inhibitory transmitter should be abandoned for the more accurate concept of an inhibitory interaction between neurotransmitter and receptor.
A wide range of substances makes up the neurotransmitter substances used by the nervous system. In some cases, they can coexist in the same neuron. Box 28-1 represents some typical substances that can be used as neurotransmitters. These substances can be used by neurons in different ways, according to the function of the specific neuron. To be used as a neurotransmitter, these substances are packaged in vesicles within the neuron and respond to the particular enzymes that are specific to that neuron.57
Amino Acids.: One of the small-molecule neurotransmitters, glutamate, is an excitatory amino acid transmitter used throughout the brain and spinal column. It is an intermediate transmitter in cellular metabolism, so the presence of glutamate in a cell does not necessarily suggest neurologic activity. Glutamate functions with its receptors in an excitatory or depolarizing system at primary afferent nerve endings, the granule cells of the cerebellum, the dentate gyrus, and the corticostriatal and subthalamopallidal pathways important to basal ganglia function. When the levels of glutamate rise above normal, it can become neurotoxic and cause cell death. Glutamate opens ion channels to bring calcium into the cell. In the case of excess glutamate, too much calcium is allowed into the cell, and the calcium eventually destroys the cell. Excess glutamate can be an effect of neuronal injury, as in stroke, brain, or spinal cord injury. It appears that the genes in the nerve cell body may trigger this excitotoxic mechanism, resulting in release of excess glutamate that may lead to the degenerative processes associated with diseases such as ALS, Alzheimer’s, Huntington’s, and Parkinson’s.43 Part of the activation of seizure is due to glutamate receptors. Toxins or drug abuse can also trigger an excitotoxic level of glutamate.56
γ-Aminobutyric acid (GABA) is a tiny amino acid that serves both as a neurotransmitter and as an intermediate metabolite in the normal function of cells. GABA is synthesized from glutamate by way of the vitamin B6-dependent enzyme, glutamate decarboxylase. GABA is the major transmitter for brief inhibitory synapses. GABAergic cells have a dense representation within the basal ganglia.21 Loss of GABAergic neurons that inhibit glutamate results in increased excitation. Glycine is another amino acid neurotransmitter that is the transmitter at some inhibitory CNS synapses. The distributions of GABA and glycine synapses overlap, but glycine is more prominent in the spinal cord.41
The N-methyl-D-aspartate (NMDA) receptor has a complex process using glutamate and glycine activation at the same time but also requiring membrane polarization to remove magnesium from inside the cell, so that the cell can allow sodium to be active within the cell. NMDA receptors are widely distributed throughout the neocortex, hippocampus, and anterior horn motor neurons. The NMDA response thus works when the membrane bearing the receptor has already been depolarized by another stimulus, so it prolongs or augments the initial depolarization. This activity supports the activities of learning and memorization. During cellular energy failure induced by ischemia, there is collapse of membrane potentials (depolarization) and uncontrolled synaptic and transmembrane release of excitatory amino acids into the extracellular space. NMDA receptors will open and allow calcium into the intracellular space causing damage to the mitochondria, limiting the production of adenosine triphosphate (ATP). Drugs that are NMDA receptor antagonists include ketamine and eliprodil. Antiepileptic drugs, such as felbamate and lamotrigine, block the glutamate and glycine activity at the NMDA receptors.16
Amines.: Cholinergic neurons play two different roles in the nervous system. Acetylcholine was the first neurotransmitter discovered and has primary activity at the level of the peripheral nervous system. It is the transmitter released by the motor neurons at neuromuscular junctions and within the autonomic nervous system. Disorders related to acetylcholine are discussed in Chapter 39. The role of the cholinergic neurons in the CNS is quite different because it is involved with the regulation of the general level of activity. Cholinergic systems can be mapped to the medial cortex and to the areas responsible for information flow to the hypothalamus and amygdala through the reticular formation. They also constitute a major element of the autonomic nervous system as preganglionic neurons of sympathetic ganglia and postganglionic parasympathetic neurons. The cholinergic and biogenic amine systems appear to establish the activity set point of the cortex and basal ganglia rather than point to point neural firing.16 Biogenic amines are synthesized from amino acid precursors, dopamine, serotonin, and norepinephrine. Two transmitters known as catecholamines are dopamine and norepinephrine.
Dopamine is synthesized in four major CNS pathways. The most important and most widely understood involves the nigrostriatal pathway of the basal ganglia. Dopaminergic function is decreased in individuals with Parkinson’s disease and attention disorders affecting the frontal lobe. The synthetic pathway for dopamine is tyrosine to dopa to dopamine.
Norepinephrine is a neurotransmitter found in the hypothalamus and the locus ceruleus in the brainstem. It is synthesized from dopamine and therefore shares the same enzymes, including the rate-limiting tyrosine hydroxylase. Like dopamine, norepinephrine is removed from the synapse by active reuptake into the presynaptic cell and then is metabolized by two enzymes, monoamine oxidase (MAO) and catechol O-methyltransferase (COMT).21 Dopamine and norepinephrine are the primary neurotransmitters associated with the task of attending. Both need to be enhanced to achieve sustained clinical benefit.
The catecholamines appear to have an important role in working memory. The cholinergic system appears to be critical for the acquisition of long-term declarative memories. Cholinergic function decreases somewhat with age and greatly in individuals with Alzheimer’s disease, and these changes may contribute importantly to corresponding reductions in declarative memory ability. Newer centrally acting cholinesterase inhibitors that can be administered orally have been developed that have been marketed for improvement of memory.
Serotonin has its main cell bodies in the dorsal raphe nucleus of the brain stem as well as the spinal cord, hippocampus, and cerebellum. In parallel to dopamine, it is synthesized by a two-step process, first with a rate-limiting enzyme and then a general enzyme. The first step takes tryptophan to 5-hydroxytryptophan (5-HTP) with the rate-limiting enzyme, tryptophan hydroxylase. The second step takes this intermediate to serotonin (5-hydroxytryptamine [5-HT]) by aromatic amino acid decarboxylase, which is the same enzyme involved in dopamine synthesis. There are several types of serotonin receptors spread throughout the brain. Serotonin is metabolized like the catecholamines by active reuptake into the presynaptic cell and then metabolism by MAO. Serotonin is removed from the synaptic cleft by reuptake pumps rather than by degradation. Tricyclic antidepressants work by inhibiting this reuptake.16
Neuropeptides.: Neurons can secrete hormones, or neuropeptides, and most or all of them can function as neurotransmitters. Neuropeptides are metabolically difficult for cells to make and transport, and can be effective at very low concentrations. Synthesis of neuropeptides begins in the nucleus of the cell, where the gene is transcribed into RNA. In the endoplasmic reticulum the RNA is translated into the neuropeptide transmitter. When they have been activated and used to produce a signal, a new supply must be produced in the cell body. Neuropeptides coexist in neurons with both amino acid and amine neurotransmitters. Neuroactive peptides are involved in modulating sensibility and emotions.
Gaseous Neurotransmitters and Others.: NO and carbon monoxide (CO) are gases than can diffuse easily through neuronal membranes and can influence subsequent transmitter release. Astrocytes may be the target, mediating cell to cell communication between vessel endothelium and smooth muscle and are critical in vasomotor control, inflammation and neuronal communication. NO sets the functional state of adjacent cells and has a short half-life. NO released from endothelial cells acts on vascular smooth muscle causing vasodilatation. NO released from inflammatory cells occurs in high concentrations and kills cells. NO may play a role in neurodegeneration, and acute elevations may contribute to damage in ischemia and trauma. NO synthesis is augmented by NMDA receptor activation by glutamate, therefore NO may synergize excitotoxity.27
Neurotrophic factors are essential to maintenance and survival of neurons and their terminals but are produced by the body in a limited supply. Four major neurotrophins have been identified in humans: nerve growth factor, brain-derived neurotrophic factor, neurotrophin 3, and neurotrophin 4/5. Neurotrophins interact with receptor cells to prolong the life of the neuron. Although this class of substances is also not fully understood, it is clear that it plays a role in the development of the nervous system. It appears to work by suppressing the pathway that leads to apoptosis.26
CLINICAL MANIFESTATIONS
The skin, muscles, and joints contain a variety of receptors that create electrical activity as described previously8,54 (see Chapter 39). The electrical input is carried to the CNS through the afferent axons via the spinal cord. The cell bodies rest in the ganglion of the dorsal root that lies adjacent to the spinal cord. The afferent fibers are arranged somatotopically in the spinal column and ascend to the brainstem and the sensory cortex. Fig. 28-8 shows the simplified synapse.41 A characteristic of the fibers that run in the dorsal column of the spinal cord is that they synapse at the level of the brainstem nuclei, where they cross over to the contralateral (opposite) hemisphere of the brain. This phenomenon is illustrated in Fig. 28-9.41 When there is a disorder of the brain that affects the afferent system above the level of the brainstem, symptoms occur on the side contralateral to the lesion.26,41
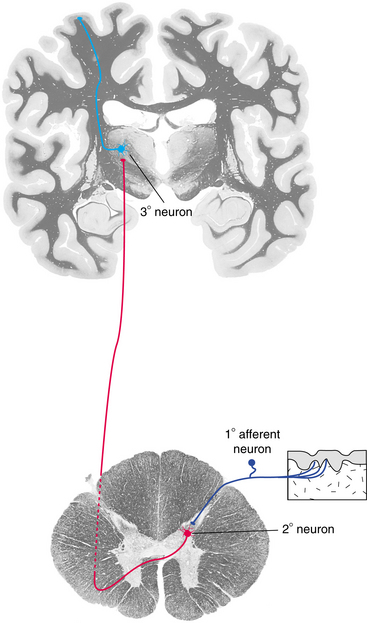
Figure 28-8 The minimum sensory pathway from the periphery to the cerebral cortex. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
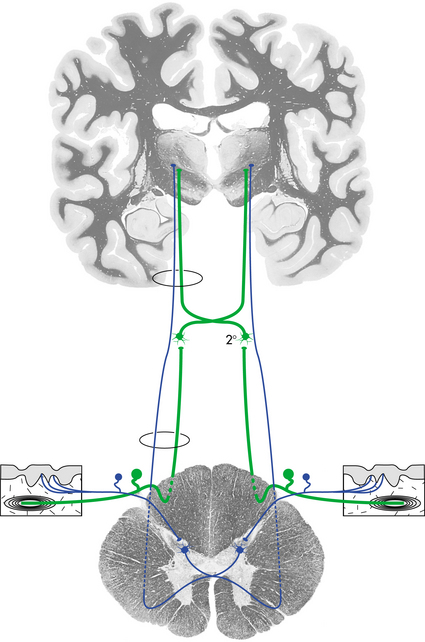
Figure 28-9 A, In the spinal cord, a lesion would result in decreased touch on the same side of the lesion and decreased pain sensation on the contralateral side. B, A lesion above the medulla would cause decreased touch and pain on the contralateral side. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
The brainstem receives information from specialized senses. For example, vestibular information is received via cranial nerve VIII and integrated through the brainstem nuclei, contributing to postural control and locomotion. Disorders of the afferent nerve, dorsal columns of the spinal cord, and brainstem result in changes in the sensory input available. This can manifest as lack of cutaneous sensation, numbness, tingling, paresthesias, or dysesthesias in the distribution of the nerves affected. Sensory input from the joints and muscles is known as proprioception. When this sensory function is lost or disturbed, the person will have difficulty maintaining the body in the appropriate position for the voluntary and involuntary movements necessary for most functional activities, especially those required for postural control. Movements become ataxic or uncoordinated because of the loss of feedback on position from the joints.3
The nervous system has several pain-control pathways available, some of which suppress and some of which facilitate the experience of pain. Modulation of noxious stimuli is directed by the reticular formation. Noxious stimuli can be experienced as more or less painful, depending on the individual’s circumstances. If an individual is focused on a task during an injury, such as a soldier or athlete, the pain of the injury may be suppressed until the task is completed. When a lesion affects the midbrain areas that modulate and interpret sensory input, such as the thalamus, the result can cause exaggeration of sensory stimuli.
Disruption of the sensory input provided by the optic nerve is evident in some disorders of the brain and will result in loss of vision in some or all of a field of view. Visual-field cuts are common with stroke (see Chapter 32). Visual hallucinations can also be part of a CNS disorder when the optic radiations or occipital lobe is disrupted, which may also be caused by stroke or a degenerative disease such as multiple sclerosis.
Brainstem Dysfunction
The brainstem contains the lower motor neurons for the muscles of the head and does the initial processing of general afferent information concerning the head. The cranial nerves enter the system at the brainstem through the respective nuclei and provide sensation and motor control of the head and neck. An anatomic view of the cranial nerves and the relationship of the nuclei to central structures is provided in Fig. 28-10.41 The sensory and motor functions of the cranial nerves are outlined in Table 28-1. A working knowledge of the attributes of the cranial nerves assists in the understanding of the level and impact of lesions within the CNS.
Table 28-1
The Cranial Nerves and Their Functions
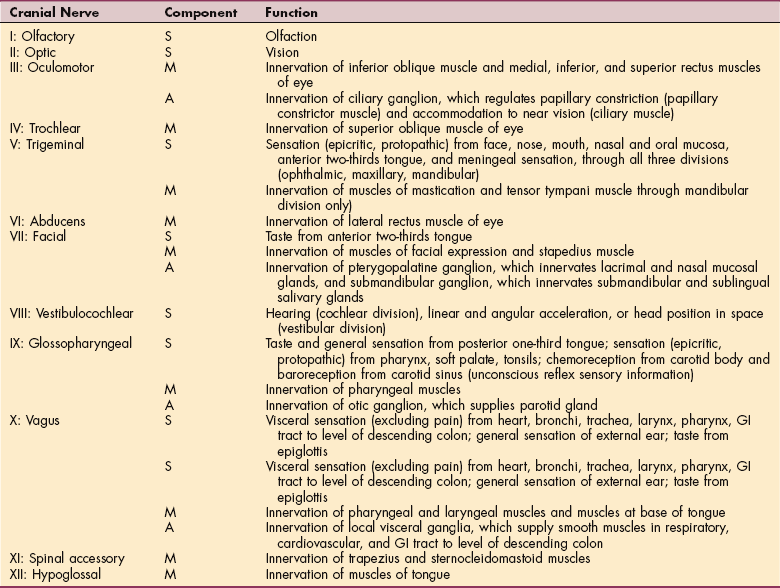
S, Sensory nervous system; M, motor nervous system; A, autonomic nervous system; GI, gastrointestinal.
From Felten DL, Felten SY: A regional and systemic overview of functional neuroanatomy. In Farber SD: Neurorehabilitation: a multisensory approach, Philadelphia, 1982, Saunders, pp 53-54.
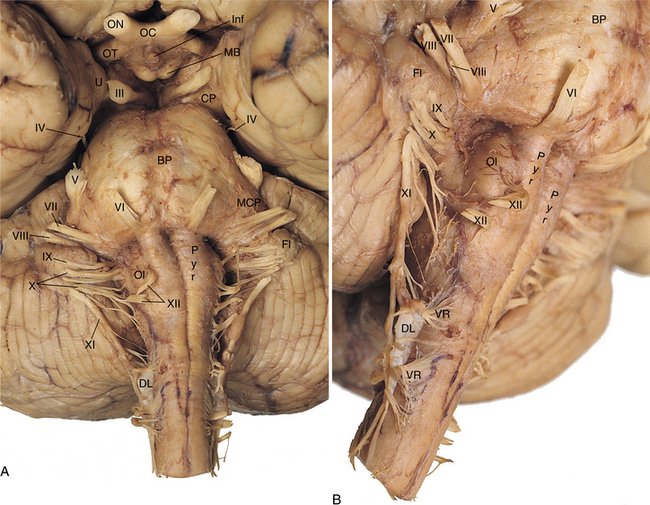
Figure 28-10 Representation of the cranial nerves III through XII. ON, Optic nerve; OC, optic chiasm; OT, optic tract; U, uncus; BP, basal pons; CP, cerebral peduncle; DL, dentate ligament (supports spinal cord); FL, flocculus of cerebellum; Pyr, (pyramids); OL, olive; MCP, middle cerebellar peduncle. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby. Dissection by Dr. Norman Koeling, The University of Arizona College of Medicine, Tucson, AZ.)
Distinctive brainstem functions include a conduit for spinal cord activity in both ascending sensory tracts and descending motor tracts. The nuclei in the brainstem provide relay functions to divert the information to the appropriate higher level structures for further modification.
The brainstem has been divided into three major subdivisions related to a characteristic set of features. The medulla, attached directly to the spinal cord, houses the inferior olivary nucleus that has direct output connections to the cerebellum and gets direct input from the spinal cord and cerebellum. The pons extends from the medulla and is attached to the cerebellum through both the middle and superior cerebellar peduncles receiving major outflow from the cerebellum. Vestibular nuclei sit within the pons, making it the center for integration of vestibular input.
The third level of the brainstem, the midbrain, contains the red nucleus with fibers that connect the cerebellum to the thalamus. The substantia nigra found here connects to the basal ganglia structures and shares the dopamine pathway related to the initiation and control of movement. It is also connected to the cortex through the cerebral peduncle containing descending fibers.
The reticular formation is a diffuse network of neurons, extending through the brainstem to higher levels, and is important in influencing movement. The reticular regions are closely related to the cerebellum, basal ganglia, vestibular nuclei, and substantia nigra and involved with complex movement patterns. It is through the reticular formation that there is inhibition of flexor reflexes, so that only noxious stimulus can evoke the flexor response such as the reflexive pulling a hand away from a hot stove.
This is a brief reflection of the complexity of the brainstem and is not intended to be comprehensive. However, it is clear that advanced knowledge of the interface and connections of the brainstem helps the therapist understand the functions that are described throughout this text.
Movement Disorders
Control of movement is accomplished by the cooperative effort of many brain structures.8,54 Activity initiated in the cerebral cortex triggers interneurons that regulate interaction of the lower motor neurons. The parietal and premotor areas of the cerebral cortex are involved in identifying targets in space, determining a course of action, and creating the motor program. The cortex determines strategies for movement. The brainstem and spinal cord are responsible for the execution of the task. The same signal may be processed simultaneously by many different brain structures for different purposes, showing parallel distributed processing. Various areas of the brain, such as the cerebellum and basal ganglia, interact to establish a motor program that modifies the hierarchic information going from the cortex to the spinal cord.
Abnormal movement patterns in neurologic disorders can result from lesions of the CNS at many levels. A simplified representation of the typical synaptic flow of neurons and interneurons is seen in Fig. 28-11. It is important to recognize that there are many synapses not represented here in the levels of the brainstem and central modulation centers of the basal ganglia and limbic lobes.
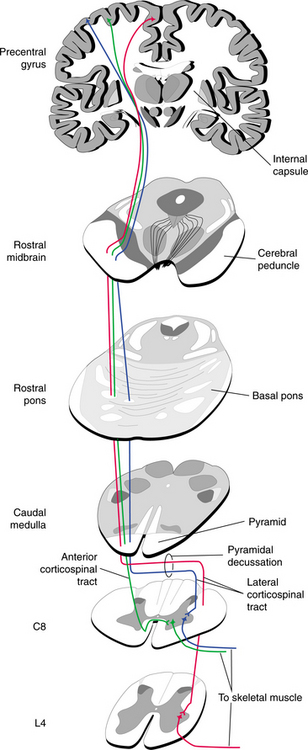
Figure 28-11 Pathway of the motor system from the cortex to the skeletal muscle as it courses through the brainstem structures and spinal cord. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Damage at any level brings about the movement disorders that are related to the pathologies in the next chapters. Motor output is critically evaluated by the therapist, thus the knowledge of patterns of abnormal movement associated with disorders of the brain is part of the clinical expertise necessary to practice.
Disorders of Coordinated Movement
Lack of coordinated movement known as ataxia can occur with damage to a variety of structures of the nervous system, including sensory neuropathies, but is most commonly associated with cerebellar dysfunction. Damage to the input and output structures of the cerebellum, such as the thalamus and vestibular nuclei, can also cause ataxic movements.
Input regarding the position of the head, trunk, and extremities comes from the spinal cord in order to compare the resulting activity with the intended motor command. This input comes in rapidly because the relay involves only a few synapses. The input comes through the climbing fibers that connect the inferior olive to the Purkinje’s cell or from mossy fibers that relay the remaining information.5 The deep cerebellar nuclei are the structures that communicate information from the Purkinje’s cell to the various nuclei of the brainstem and thalamus.26 The cerebellum has no direct synapse with the spinal cord but exerts its influence through the action on interneurons within the nuclei of the brainstem.
The medial region known as the vestibulocerebellum connects with the cortex and brainstem through both its ascending and descending projections. The cerebellum has influence on movement through the vestibulospinal and reticulospinal tracts. Lesions result in the inability to coordinate eye and head movement, postural sway, and delayed equilibrium responses.5 Postural tremor is present in some individuals with vestibulocerebellar lesions.
The spinocerebellum connects to the somatosensory tracts of the spinal cord. It receives input from the cortex regarding the ongoing motor command. Control of proximal musculature is achieved via the connections to the motor cortex. Lesions of the spinocerebellum can cause hypotonia and disruption of rhythmic patterns associated with walking. Precision of voluntary movements is lost when this area is dysfunctional.38
The anterior lobe of the cerebellum is implicated in disorders of gait with loss of balance noted in stance. Proprioception may give inaccurate cues because the cerebellar relays become disrupted. Long loop reflexes lose adaptability and are unable to trigger appropriate responses in the lower leg to maintain balance when the body sways or the surface is moving. The ability to modify reflexes is lost even when there are repeated trials.60
In the cerebrocerebellum, or posterior lobes, connections are made to the cortex through the pons. The posterior lobes are involved in complex motor, perceptual, and cognitive tasks. Lesions of the cerebrocerebellum lead to a decomposition of movement and timing.
Hypotonicity, or decreased muscle tone, can occur on the side of the lesion or bilaterally if the lesion is central and is seen primarily in the proximal muscle groups. The person with hypotonicity is unable to fixate the limb posturally, leading to incoordination with movement. Asthenia, or generalized weakness, is sometimes seen in the person with cerebellar lesions. Hypotonicity and asthenia, however, do not always occur together. It is believed that both disorders represent loss of input from the cerebellum to the cerebral cortex, but they may represent loss of input to different areas of the cortex.
Dysmetria, the underestimation or overestimation of a necessary movement toward a target, is commonly seen with cerebellar disorders. There is an error in the production of force necessary to perform an intended movement. The initiation of movement is prolonged compared to normal, and the ability to change directions rapidly is impaired. The resulting overshoot and undershoot during movement are known as an intention tremor. Dysdiadochokinesia, the inability to perform rapidly alternating movements, is related to the inability to stop ongoing movement. The movement becomes slow, without rhythm or consistency.
Decomposition of movement is seen in persons with cerebellar dysfunction. Instead of performing a movement in one smooth motion, the person will move in distinct sequences to accomplish the motion. Multijoint movements are more affected than single-joint movements. Disruption in force and extent of movement will result in difficulty with grip control and maintaining static hold against resistance. When the resistance is removed, for example, the extremity will oscillate because of lack of feedback regarding position and force needed to maintain static hold.
Scanning speech is a component of cerebellar dysfunction representing complexity of the motor activity. Word selection is not affected, but the words are pronounced slowly and without melody, tone, or rhythm. This reflects the incoordination or hypotonicity of the muscles of the larynx in controlling the voice.
Eye movements are disrupted in the person with cerebellar dysfunction, in both a static head and eye position and with movement of the head. Gaze-evoked nystagmus, or nonvoluntary rhythmic oscillation of the eye, occurs when the cerebellum is unable to hold the gaze on an object, especially a lateral position. When looking at a lateral target, the eyes drift back toward midline and then immediately back to the target. Eyes flickering on and off the target, eyes fluttering around the target, or spastic bursts of eye oscillations may be present when there is brainstem or midline cerebellar lesions.
Ocular dysmetria is similar to the dysmetria seen in the extremities. This dysmetria is seen in cerebellar lesions when the eyes are moving from one target to another (known as saccadic movement) or when attempting to follow a target (known as smooth pursuit).
Vestibuloocular function is disrupted in medial lesions, and the ability to maintain eye stability during head movement is affected. See the section on Vestibular Dysfunction in Chapter 38 for more information on vestibuloocular dysfunction.
Gait disturbance is another disorder related to dysfunction of the cerebellum. The gait becomes wide based and staggering without typical arm swing. The step length is uneven, the step widths are inconsistent, and the feet are often lifted higher than necessary. Stance and swing become irregular, and there is loss of adaptation to changes in terrain. It becomes difficult to perform heel-to-toe walking or walking a straight line, which is the standard sobriety test. In some persons, there is a surprising ability to avoid a fall, although the standing balance is abnormal. When the person is able to perform compensatory movements of the upper body and limbs, falls can be avoided.19
The cerebellum plays a major role in motor learning. The cerebellum is vital in anticipatory, or feed-forward, activity and modification of response.30 The cerebellum learns or memorizes small movements that are integrated into complex activity. During the acquisition phase of motor learning, the cerebellum is active.24 Increased activity has also been noted during mental imagery or mental rehearsal of a motor program.46 The cerebellum is active during cognitive and emotional processes, and lesions can cause difficulty in shifting attention from one sensory or thought domain to another.
Deficits of Higher Brain Function
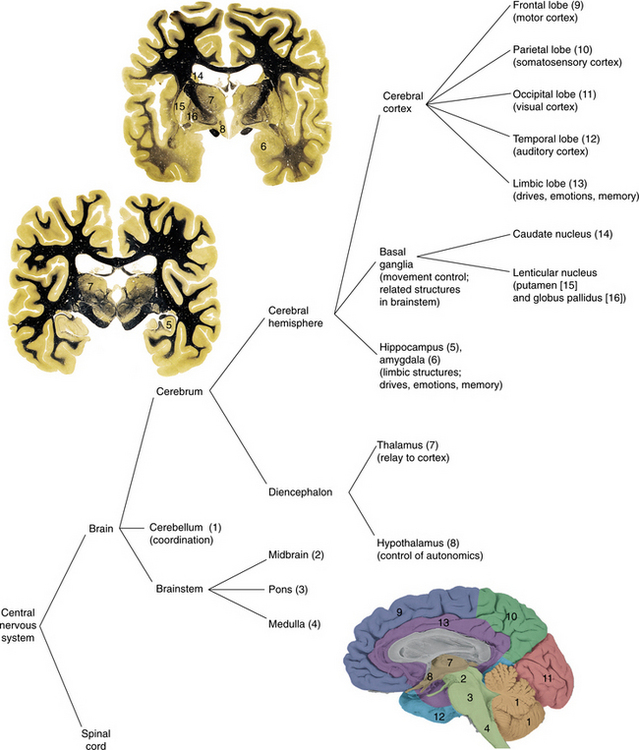
The cortex has a great deal to do with the abilities and activities that are a part of the highest development in humans, including language and abstract thinking. Perception, movement, and adaptive response to the outside world depends on an intact cerebral cortex. As with other parts of the CNS, it is subdivided for ease of understanding the separate functions, although the structure and function is full of overlap. Fig. 28-12 represents some of the functional specialization of the brain. Fig. 28-13 describes the lobar relationship to the cerebellum and brainstem.
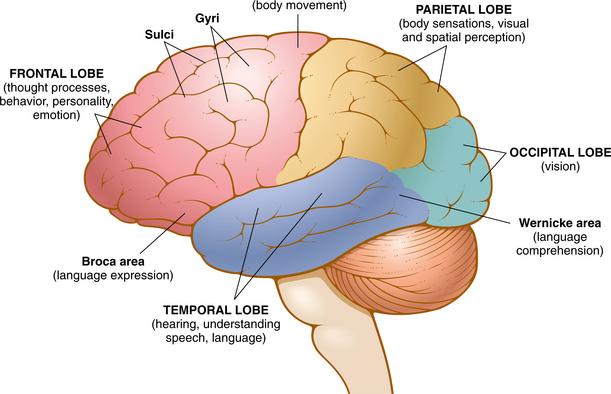
Figure 28-12 Schematic representation of functional specialization in the cortex. (From Chabner DE: The language of medicine, ed 8, Philadelphia, 2007, Saunders.)
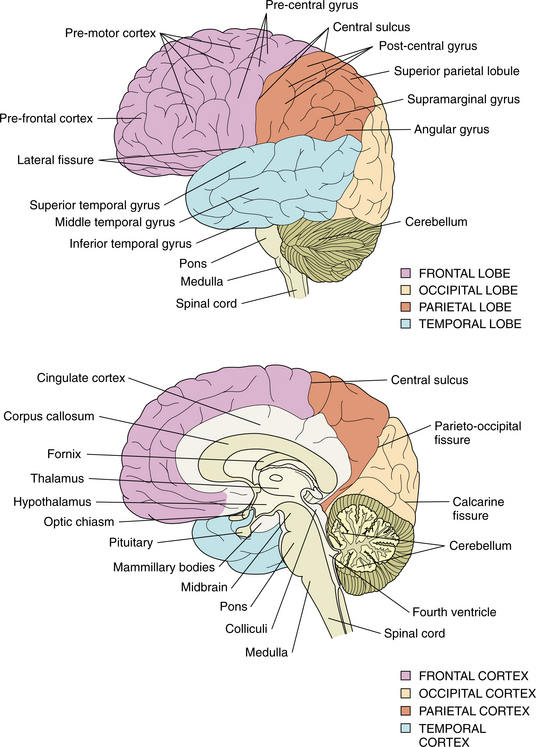
Figure 28-13 The lobes of the cortex and their relationship to the cerebellum, midbrain, and brainstem. (From Farber SD: Neurorehabilitation: a multisensory approach, Philadelphia, 1982, WB Saunders.)
The frontal lobe is the largest single area of the brain, constituting nearly one-third of the brain’s cortical surface. It is phylogenetically the youngest area of the brain and has major connections with all other areas of the brain. The frontal lobe is responsible for the highest levels of cognitive processing, control of emotion, and behavior. An individual’s personality is established as a frontal lobe function, and one of the most disturbing deficits seen with lesions affecting the frontal lobe is change from the person’s premorbid personality. A person’s character and temperament are changed by damage to the frontal lobe. Slow processing of information, lack of judgment based on known consequences, withdrawal, and irritability can be the result of an insult to the frontal lobe. Lack of inhibition and apathy are common clinical problems related to frontal lobe damage. The person with a frontal lobe disorder may lack insight into the deficits, and therefore behavior can be difficult to control.
The right hemisphere syndrome represents the inability to orient the body within external space and generate the appropriate motor responses. Hemineglect is one of the most common deficits seen with right hemisphere lesions. The individual does not respond to sensory stimuli on the left side of the body and does not respond to the environment surrounding the left side. Hemineglect is evident in the involved extremities and trunk during mobility and self-care activities. The ability to draw in two and three dimensions is lost along with other drawing skills, such as perspective and accurate copying. Spatial disorientation can result, with the person losing familiarity with the environment and becoming lost in areas that should be familiar. Inability to read and follow a map can be an indication of right hemisphere deficit.13
Disorders of emotional adjustment often follow a lesion in the right hemisphere. These disorders are primarily in the affective domain of interpersonal relationships and socialization. Cortical control of the limbic system is believed to be responsible, but the exact mechanism of control of more complex emotional behavior is not completely understood at this time. There appears to be hemispheric lateralization of emotions with suggestions that the right hemisphere is the dominant hemisphere in controlling emotions.
Language is one of the higher functions of the brain that is affected in many disorders of the CNS. Speech is a more elementary capacity than language and refers to the mechanical act of uttering words using the neuromuscular structures responsible for articulation. Dysarthria, a disturbance in articulation, and anarthria, the lack of ability to produce speech, are disorders of speech not language. One common language disorder is expressive aphasia, a deficit in speech production or language output, accompanied by a deficit in communication, in which speech comes out as garbled or inappropriate words.
Localization of speech production in the left frontal lobe and impaired language comprehension in the temporal lobe demonstrate how higher functions can be related to brain regions. However, language control may be in different areas for different persons, and therefore damage to the same area of the brain may produce aphasia in some individuals whereas others may be spared. Left hand–dominant people may have right hemisphere dominance for language.31
Alexia is another symptom of higher brain dysfunction. It is the acquired inability to read. Alexia is typically caused by lesions in the left occipital lobe and the corpus callosum that prevent incoming visual information from reaching the angular gyrus for linguistic interpretation. Agraphia can be caused by lesions located anywhere in the cerebrum. Because writing is a motor skill, lesions of the corticospinal tract, basal ganglia, and cerebellum; myopathies; and peripheral nerve injuries can all cause abnormal or clumsy writing. These disorders may be seen in addition to neurobehavioral syndromes. Typically the features of agraphia tend to parallel the characteristics of aphasia.
Apraxia is an acquired disorder of skilled purposeful movement that is not a result of paresis, akinesia, ataxia, sensory loss, or comprehension. Ideomotor apraxia is the most common type and represents the inability to carry out a motor act on verbal command. Ideomotor apraxia appears to be caused by a lesion in the arcuate fasciculus. The anterior connection from the left parietal lobe may be disrupted, preventing the motor system from receiving the command to act. A lesion in the left premotor area can cause apraxia by directly interrupting the motor act. Damage to the anterior corpus callosum can lead to apraxia that is evident in the left hand only. Ideational apraxia is failure to perform a sequential act even though each part of the act can be performed individually. The lesion causing ideational apraxia appears to be in the left parietal lobe, as in hemiparesis, or in the frontal lobe, as in Alzheimer’s disease. The syndrome is seen as well with diffuse cortical damage associated with degenerative dementia.
Agnosia is the inability to recognize an object; the previously acquired meaning of an object is no longer attached to it. Agnosia is associated with lesions of the sensory cortices involved with seeing, hearing, and feeling and with the loss of one sensory modality. It is difficult to assess because the person is often easily able to compensate. Although the ability to recognize an object by vision is gone, the ability to recognize that same object by hearing or feeling is retained.
Altered States of Consciousness
Alteration of consciousness is not considered an independent disease entity but a reflection of some underlying disease or abnormal state of brain function. The human brain possesses a mechanism that allows a waking and sleeping state (arousal), as well as a separate ability to focus awareness on relevant environmental stimuli (attention).13,37 To achieve a state of consciousness the cerebral cortex must be activated by the ascending reticular formation fibers in the brainstem. The fibers extend to the thalamus, limbic system, and cortex. The upper part of this system acts as an on/off switch for consciousness and controls the sleep-wake cycle. The lower part controls respiration.
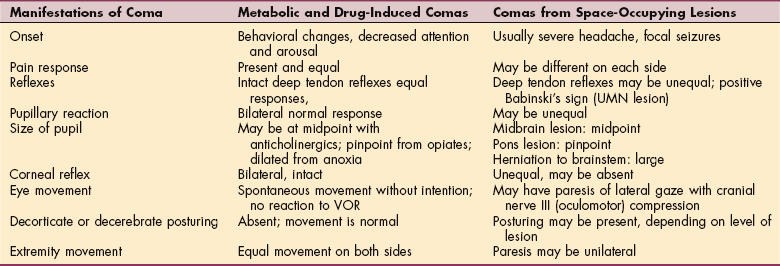
Disturbances of arousal and attention can range from coma after brainstem injury (see Chapter 33) to confusional states caused by drug intoxication. Metabolic or systemic disorders generally cause depressed consciousness without focal neurologic findings.59 CNS disorders may or may not have concomitant focal signs. Table 28-2 compares metabolic and drug-induced coma with coma caused by space-occupying lesions.
Clinical disorders of arousal may result in hyperaroused states and can appear as restlessness, agitation, or delirium. This is presumably a result of the loss of hemispheric inhibition of brainstem function. Hypoarousal can be described on a spectrum ranging from drowsiness to stupor and coma. Stupor is a state of unresponsiveness that requires vigorous stimulation to bring about arousal. Coma is a state of unarousable unresponsiveness. Small and restricted lesions of the brainstem can result in stupor and coma. Massive bilateral hemispheric lesions are necessary to cause coma.
Damage to the cerebral cortex can be caused by loss of blood flow, subarachnoid hemorrhage, anesthetic toxicity, hypoglycemia, hypothermia, or status epilepticus (see the section on Epilepsy in Chapter 36). If the link to the brainstem is destroyed, the person will remain in a persistent vegetative state (PVS). Although the person may make random movements and the eyes may open, mentation remains absent. Akinetic mutism, similar to PVS, reflects damage to the mediofrontal lobe and results in lack of motivation to perform any motor or mental activity (abulia). In the locked-in syndrome, there is damage to the pons resulting most often from thrombosis of the basilar artery. This is a remarkable impairment, involving no mental deficit at all but resulting in inability to move anything but the eyes. It is in essence the opposite of PVS.
Supratentorial lesions that cause increased pressure, such as hemorrhage, cerebral edema, or neoplasm, can cause coma by producing tentorial herniation and subsequent compression of the brainstem. There is usually a hemiparesis with a dilated pupil on the side of the lesion because of central compression involving the third cranial nerve by the herniation.
In infratentorial lesions, brainstem damage can be related to drugs, hemorrhage, infarction, or compression from the posterior fossa. Disruption of ocular movements is an early sign of brainstem involvement.65 There is loss of the pupillary reaction to light while the corneal reflex remains intact.
Brain death relates to destruction of both the upper and lower parts of the reticular formation in the brainstem, which will eventually lead to death. Cortical electrical activity and spinal reflexes may be preserved, but these are of no consequence because they are unable to be used for thought or movement.
Attention is more difficult to relate to specific brain structure than arousal. However, the acute confusional state is one of the most common neurologic disorders encountered. Although there is not a clear understanding of the mechanism of attention from the neuroanatomic perspective, there appears to be a major role played by the parietal and frontal lobes. Frontal and prefrontal areas of the brain are responsible for mental control, concentration, vigilance, and performance of meaningful activity. Cognition and emotional control are established by extensive white matter connections between the frontal lobes and the remainder of the cerebrum.48 Diseases that affect the white matter, such as multiple sclerosis, can affect the level of attention without decreasing arousal. Psychiatric disease has an effect on both arousal and attention37. The acute confusional state may be the result of a number of causes. Intoxicants, metabolic disorders, infections, epilepsy, blood flow disorders, traumatic injuries, and neoplasms can all be responsible for the change in orientation or attention.
Emotional Instability
The orbital prefrontal region is especially expanded in the right cortex and is dominant for selectively attending to facial expressions. It has extensive connections with limbic and subcortical regions, important in regulation of emotional information and mediation of pleasure and pain. Primitive emotions that serve fundamental motivational and social communication functions and nonverbal affects are spontaneously expressed on the face. Psychic systems process unconscious information. Empathetic cognition and the perception of the emotional states of other human beings are developed within the first 3 years of life. Control of vital functions enable the individual to cope actively with stress and external challenge. Self-regulation functions are learned through this region.49
Although the limbic system defies exact definition, it is recognized as the area of control of human behavior and is widely studied in behavioral neurology. The limbic system, sometimes referred to as the limbic lobe, is generally considered to encompass part of the cortical, diencephalon, and brainstem structures. This system includes the orbitofrontal cortex, hippocampus, parahippocampal gyrus, cingulate gyrus, dentate gyrus, amygdala, septal area, hypothalamus, and portions of the thalamus. The limbic lobe structures are seen in Fig. 28-14.41 Working together, these structures provide the essential, need-directed motor activity necessary for survival. This is the area that integrates the motivation and intentional drive to trigger a motor act. Both the automatic and somatic systems are influenced by the limbic system.62 Limbic syndromes involve the primary emotions, which are those associated with pain, pleasure, anger, and fear. The processing of the limbic system is responsible for the fact that emotionally charged experiences will be more easily remembered than those with less emotional stimulation.13 In lower animals, the limbic system is concerned primarily with the sense of smell, and it is a common observation that smells can trigger a strong emotional response in humans.
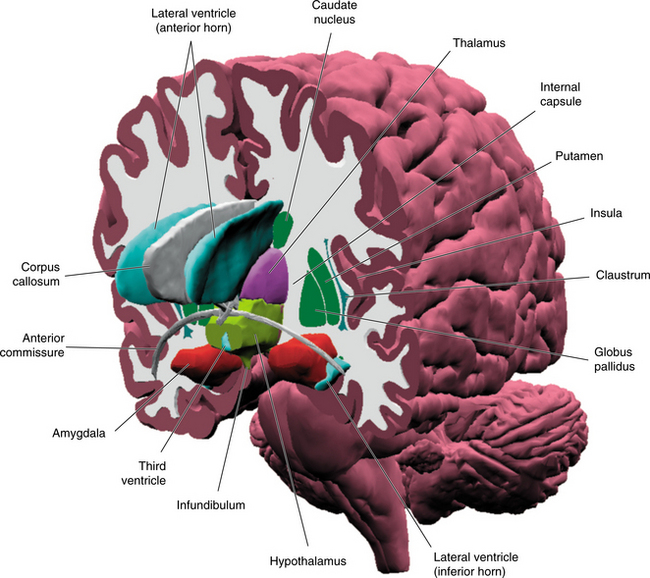
Figure 28-14 Three-dimensional representation of the structures of, and surrounding the limbic lobe. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
The amygdala are nuclei located in the medial temporal lobe anterior to the hippocampus as illustrated in Fig. 28-15. The amygdala is involved in sensory processing and determining the value of the information received. The patterns of emotional memories are formed here, and this is the area that establishes the anxiety and panic or the pleasure that is unconsciously related to an experience that may or may not be remembered.3 The amygdala is richly connected with the prefrontal cortex, the thalamus, hypothalamus, and brainstem as seen in Fig. 28-16.41 Not represented here is the influence that the prefrontal cortex has on the amygdala, which is thought to be inhibitory. The amygdala is the central structure associated with the learning of fear, fearful responding, and associated autonomic and behavior responses.
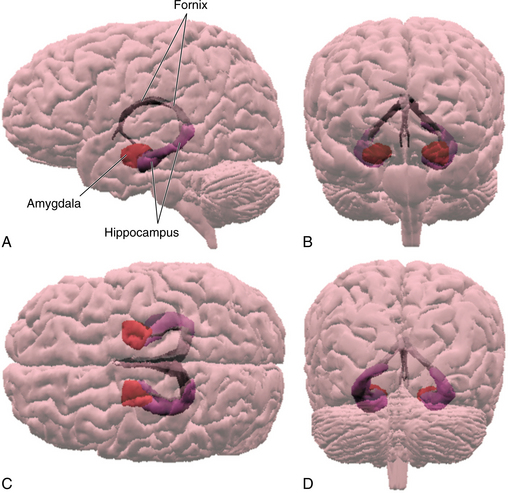
Figure 28-15 Placement of the main structures of the limbic lobe as seen from left (A), back (B), above (C), and behind (D). (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
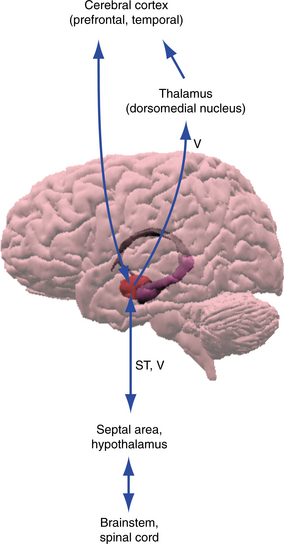
Figure 28-16 The limbic lobe gives input directed toward the cerebral cortex and the hypothalamus, brainstem, and spinal cord. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Kindling is a term originally used to describe how subthreshold seizure activity becomes increasingly active and severe with successive seizures. Partial kindling also can occur in the amygdala, and it appears related to increased defensive responses and anxiety-like behavior in animals. In humans the amygdala is involved in integrating emotional and contextual information that are parts of the human stress response. Amygdaloidal lesions result in hampered fear conditioning, whereas amygdaloidal stimulation results in classic fear responses such as defensive and aggressive behavior and autonomic reactivity. It has been demonstrated that the amygdala plays a role in conditioned fear responding in general and the startle response in particular.
Perhaps the best example of an anxiety disorder that seems to follow the classic fear-conditioning model is posttraumatic stress disorder (PTSD). Greater amygdala activations were identified in persons with PTSD than in healthy controls. Activations in the amygdala correlated with weakened activation of the medial prefrontal cortex and hippocampus.39 The prefrontal cortex is thought to be hypoactivated in PTSD, particularly during trauma memory activation. It is possible that such prefrontal cortex hypoactivation may be related to startle responses that are larger in PTSD when exposed to contexts in which they know they will be exposed to reminders of their trauma. Recent positron emission tomography (PET) studies have found that the amygdala is clearly active when individuals are thinking about their trauma.28 The amygdala and the hippocampus play a crucial role in the pathophysiology of social phobia.
The thalamus is a two-lobed medial structure that that sits just above the brainstem and is bounded on its dorsal surfaces by the lateral ventricles. The thalamus consists of multiple nuclei receiving input from sensory receptors and brainstem arousal systems and then relays this information to the frontal cortex, the cingulate gyrus, the amygdala, and the hippocampus. With the exception of olfaction, all sensory input goes through thalamic nuclei before being sent onto the cortex. The thalamus affects the quantity and quality of sensory processing. The fact that basic sensory information and arousal signals converge in the thalamus explains why even basic sensory signals can be distorted under conditions of high arousal. Although moderate arousal may facilitate transmission, conditions of high stress likely will distort or hinder transmissions to target structures throughout the brain.
Dissociative symptoms are common among survivors of trauma, and maladaptive levels of dissociation can develop alongside other pathologic responses to trauma. Dissociation is defined as a disruption in the usually integrated functions of consciousness, memory, identity, or perception of the environment, and the term dissociative symptoms in the literature have been used to capture a range of symptoms that can include changes in time perception, altered sensory perception, flashbacks, psychogenic amnesia, reduction in awareness, affective blunting, feelings of detachment, depersonalization, multiple identities, and derealization.15,28
Although severe disturbances in sensory processing may occur under conditions of extreme stress, more subtle changes may occur even at baseline. The thalamus has rich bidirectional connections with the cingulate gyrus and the frontal cortex, two of the structures responsible for the prioritization and shifting of attention. During the extreme stress of an actual trauma, it is likely that the thalamus impairs rather than facilitates the processing of environmental stimuli. In this sense, the thalamus has a role to play in amnesia for traumatic events. For example, disruption in the relay of contextual and traumatic information could contribute to the fragmentation and inaccuracies associated with traumatic memory. Unlike the role the hippocampus might play in fragmenting sensory elements of the memory, however, thalamic interference would result in an initial interference with basic stimulus encoding.28
Levels of emotion that are generated and advanced by the limbic system, or more specifically the amygdala, can be described on a continuum. An emotion can be triggered as fear or frustration, which when heightened can manifest as anger. If the neurochemical activity continues to build and leads to internal chaos or conflict, it becomes rage. The motor response will become violent if there is a sufficient trigger. Genetics and environmental history will lead to differences in how a person moves from fear to violence. When there is damage to the area of the limbic lobe that results from injury or disease, there can be an increase in rage and easy progression to violence. The diffuse axonal damage of head injury can cause a tendency to become easily frustrated or to have unsubstantiated fears.
The different symptom dimensions of obsessive compulsive disorder and other anxiety disorders are likely to share common neural substrates dedicated to general threat detection and emotional arousal because these reactions are adaptive and useful to deal with different kinds of threat. Research suggests that syndrome-specific neural substrates may have evolved to deal with specific threats. In evolutionary terms, general anxiety, which is common to individuals who have obsessive compulsive disorder and other anxiety disorders, may have evolved to deal with nonspecific threats, for example, cleanliness is important for protection against infections; harming obsessions and checking rituals keep people safe; and hoarding helps people survive periods of scarcity The neuroimaging findings, showing increased activation of limbic and ventral frontal-striatal regions in obsessive compulsive disorder, could reflect exaggerations of normal emotional responses to biologically relevant stimuli rather than fundamentally abnormal neuronal responses.35
Borderline personality disorder (BPD), including affective dysregulation, identity disturbance, and self-mutilating behaviors, were originally thought to involve a disorder of character. In recent studies, it appears that BPD individuals are compromised significantly in executive skill and/or other frontal lobe functions, visuomotor speed, attention, and verbal memory. These fairly consistent neuropsychologic findings in adults are supported by developmental studies of children with borderline features. These children appear to have greater difficulty with executive skills, including planning, organizing, and sequencing; perceptual motor functioning; and memory proficiency.39
Fear conditioning is a fast process, with a long-lasting effect, but repeated exposure to the conditioned stimulus in the absence of the unconditioned stimulus can lead to extinction. Extinction reduces the likelihood that the conditioned stimulus will elicit the fear response. The medial prefrontal and anterior cingulate cortices have been implicated in extinction learning. Understanding how learned fears are diminished and how extinction learning is changed in individuals who have anxiety disorders might be an important step in translating neurobiologic research to diagnosis and treatment of these individuals.
Memory Problems
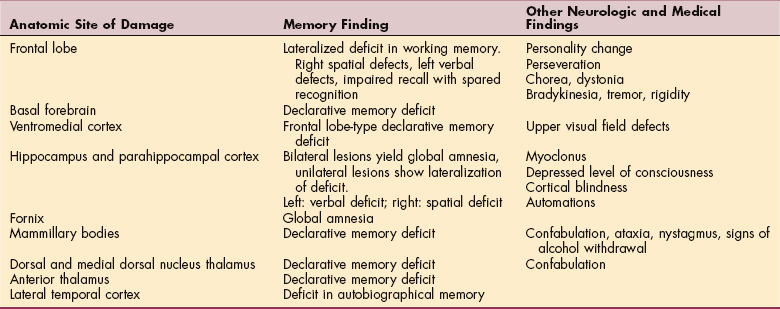
Memory is associated with various areas of the brain, and a particular area may be responsible for different aspects of memory. The hippocampus, the thalamus, and the basal forebrain are critical to the performance of recent memory (Table 28-3). Fig. 28-17 shows the relationship of learning strategies and brain regions. For immediate auditory memory, left and right temporal-parietal cortices mediate auditory verbal and nonverbal material. Neurogenesis has been observed in the dentate gyrus of the hippocampus throughout the lives of many species, including humans. Not all newly generated hippocampal neurons survive, but hippocampal-dependent memory tasks can enhance the survival of these neurons.21 Inflammatory cytokines reduce hippocampal neurogenesis and impair the ability to maintain long-term potentiation in the hippocampus, which is a critical physiologic process involved in memory consolidation.20
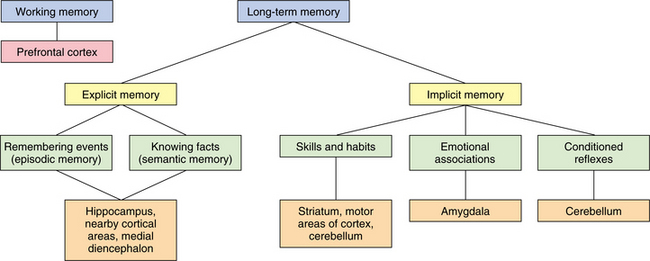
Figure 28-17 Anatomic correlates for explicit and implicit learning. (From Nolte J: The human brain: an introduction to its functional anatomy, ed 5, St Louis, 2002, Mosby.)
Working memory, the ability to hold information in short-term storage while permitting other cognitive operations to take place, appears to depend on the prefrontal cortex. Keeping a spatial location in mind may involve a right frontal area that directs the maintenance of that information in a right parietal area, whereas keeping a word in mind may involve a left frontal area that directs the maintenance of that information in a left temporal or parietal area. Specific basal ganglia and cerebellar areas appear to support the working memory capacity of particular frontal regions.
Disorders of recent memory, known as amnesia, are a significant neurobehavioral phenomenon and common in persons after traumatic brain injury. Declarative memory is retention of facts and events of a prior experience or the memory of what has occurred and is related to explicit learning. Procedural memory describes the learning of skills and habits, or how something is done. Implicit learning is based on procedural memory. The relationship to memory and relearning motor skills is discussed in the Special Implications for the Physical Therapist: Motor Learning Strategies in this chapter.
Anterograde amnesia is the failure of new learning or formation of new memory. Retrograde amnesia is the loss of ability to recall events. The inability to acquire new learning is often accompanied by confabulation, the fabrication of information in response to questioning. Traumatic amnesia refers to an individual’s inability to recall significant aspects of their traumatic experience. Traumatic memories are reported to be fragmented, compartmentalized, and disintegrated, suggesting that the hippocampus still may have a role to play in the phenomena of traumatic amnesia. Dysregulation in the hippocampal system has the potential to generate narratives of traumatic events that are spotty and unreliable.
Neuromodulators, such as norepinephrine, have the potential to affect hippocampal functioning in a more dynamic fashion. The locus ceruleus, located in the brainstem, for example, projects directly to the hippocampus and modulates its functioning through norepinephrine release. The effects of such a network are unclear, although the implications are that stress-related memory alterations might occur on a split-second basis and deficient or extreme locus ceruleus input may disrupt normal hippocampal processing severely. Given that the hippocampus plays a role in integrating input from diverse sources when encoding memory, disruptions in its functioning may lead to memories that seem fragmented and nonlinear. Over time, fragments of the memory may become consolidated, vivid, and easily recalled, while other fragments rarely are accessed.28
The role of stress in neurologic disease often is overlooked. Several chronic neurologic disease states, such as Alzheimer’s disease, are associated with elevated secretion of stress hormones, in particular cortisol, which results from overactivity of the hypothalamic-pituitary-adrenal (HPA) axis. The stress or perceived threat activates the hypothalamus, triggering release of hormones that cause increased adrenal output of cortisol.47 Stress also can trigger or exacerbate symptom onset and perhaps progression of chronic illness such as Parkinson’s disease. Stress hormones also can mitigate the impact of acute neurotrauma; for example, there is a positive correlation between cortisol levels and mortality after head injury. Thus neurologic disease states can occur within a context of elevated glucocorticoids, which may have profound influences on recovery and neuroplasticity. In addition, abnormal regulation of glucocorticoid release is associated with many affective disorders, such as depression and PTSD, that are overrepresented in populations with neurologic disease; Parkinson’s disease is a prime example. Acute and sustained glucocorticoid release also can precipitate changes in peripheral and central immune signaling, resulting in cytokine profiles that may be deleterious for functional recovery in the face of neurologic challenge.23
Accumulation of risk is another concept that plays a pivotal role in the life-course model of chronic diseases. Allostasis is defined as the ability to achieve stability through change. The price of this accommodation to stress has been defined as the allostatic load. It follows that acute stress (the “fight, flight, or freeze” response) and chronic stress resulting from the cumulative load of minor day-to-day stresses can add to the allostatic load and have long-term consequences. Subacute stress is defined as an accumulation of stressful life events over a duration of months and includes emotional factors, such as hostility and anger, as well as affective disorders such as major depression and anxiety disorders. Chronic stressors include factors, such as low social support, work stress, marital stress, and caregiver strain, and present as feelings of fatigue, lack of energy, irritability, and demoralization.
The link between chronic psychologic distress and adverse behavior, such as overeating, may be centrally mediated. Normally, glucocorticoids help end acute stress responses by exerting negative feedback on the HPA axis. The combination of chronic stress and high glucocorticoid levels seems to stimulate a preferential desire to ingest sweet and fatty foods, presumably by affecting dopaminergic transmission in areas of the brain associated with motivation and reward.17
Brain areas associated with reward are linked with those that sense physical pain. Chronic pain can cause depression, and depression can increase pain. Most individuals who have depression also present with physical symptoms. Studies using functional magnetic resonance imaging (fMRI) have shown that social rejection lights up brain areas that are also key regions in the response to physical pain. The area of the anterior cingulate cortex that is activated by visceral pain also is activated in cases of social rejection.
Disturbances of neurologic function can result in behavioral disturbances that mimic disturbances of mental function in psychiatric disorders. Delusions, or fixed false beliefs, have been reported in a great variety of neurologic conditions and appear to be associated with the limbic system. Paranoid delusions are common in disorders of the medial temporal lobe or a combination of the frontal and right parietal lobes. Hallucinations are sensory experiences without external stimulation. Visual hallucinations generally suggest neurologic involvement; auditory hallucinations imply psychiatric disease. Midbrain lesions in the cerebral peduncles can cause hallucinations involving animals. Temporal lesions can cause recurrent auditory experiences.13
Rapid-eye movement (REM) sleep behavior disorder is characterized by loss of muscular atonia and prominent motor behaviors during REM sleep. Sleep behavior disorder can cause sleep disruption. The disorder is strongly associated with neurodegenerative diseases such as multiple-system atrophy, Parkinson’s disease, dementia with Lewy bodies, and progressive supranuclear palsy. The symptoms of sleep behavior disorder precede other symptoms of these neurodegenerative disorders by several years. Furthermore, several recent studies have shown that sleep behavior disorder is associated with abnormalities of electroencephalographic (EEG) activity; cerebral blood flow; and cognitive, perceptual, and autonomic functions. Sleep behavior disorder might be a stage in the development of neurodegenerative disorder. Box 28-2 includes the areas of the brain that play a role in sleep behavior.18
Lesions of the hemispheres or lobes may cause loss of the functions that each hemisphere controls. Because diseases and damage caused by trauma will often affect one area of the brain, the associated syndromes for the main areas of the brain are described.33
Autonomic Nervous System
The term autonomic nervous system was introduced to describe the system of nerves that controls the unstriated tissue, the cardiac muscle, and the glandular tissue of mammals involved in the control of autonomic function. The autonomic CNS neurons are located at many levels from the cerebral cortex to the spinal cord. Efferent autonomic pathways are organized in two major outflows: the sympathetic and parasympathetic. Finally, the enteric nervous system, which is considered a separate and independent division of the autonomic nervous system, is located in the walls of the gut. The schematic diagram of the autonomic nervous system is seen in Fig. 28-18.
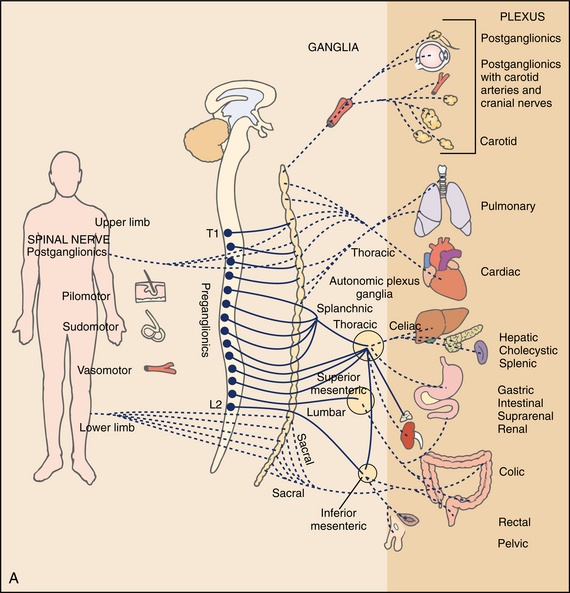
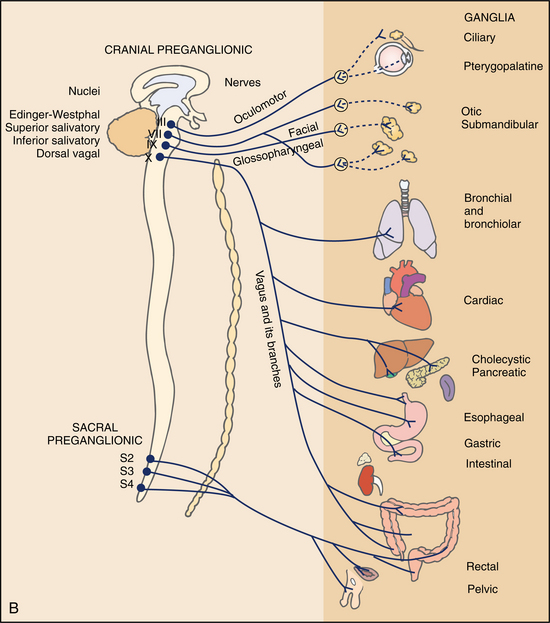
Figure 28-18 Sympathetic (A) and parasympathetic (B) divisions of the autonomic nervous system: efferent systems. (From Levy MN, Koeppen BM: Berne and Levy principles of physiology, ed 4, St. Louis, 2006, Mosby.)
Neurons in the cerebral cortex, basal forebrain, hypothalamus, midbrain, pons, and medulla participate in autonomic control. The central autonomic network integrates visceral, humoral, and environmental information to produce coordinated autonomic, neuroendocrine, and behavioral responses to external or internal stimuli. A coordinated response is generated through interconnections among the amygdala and the neocortex, forebrain, hypothalamus, and autonomic and somatic motor nuclei of the brainstem. The insular and medial prefrontal cortices (paralimbic areas) and nuclei of the amygdala are the higher centers involved in the processing of visceral information and the initiation of integrated autonomic responses. The central nucleus of the amygdala projects to the hypothalamus, periaqueductal gray (PAG), and autonomic nuclei of the brainstem to integrate autonomic, endocrine, and motor responses to emotionally relevant stimuli.
The hypothalamus integrates the autonomic and endocrine responses that are critical for homeostasis. The PAG matter of the midbrain is the site of integrated autonomic, behavioral, and antinociceptive stress responses. It is organized into separate columns that control specific patterns of response to stress. The lateral PAG mediates sympathoexcitation, opioid-independent analgesia, and motor responses consistent with the fight-or-flight reaction. The ventrolateral PAG produces sympathoinhibition, opioid-dependent analgesia, and motor inhibition.
Neurons in the medulla are critical for the control of cardiovascular, respiratory, and gastrointestinal functions. The medullary nucleus of the solitary tract is the first relay station for the arterial baroreceptors and chemoreceptors, as well as cardiopulmonary and gastrointestinal afferents.
Preganglionic sympathetic neurons are organized into different functional units that control blood flow to the skin and muscles, secretion of sweat glands, skin hair follicles, systemic blood flow, as well as the function of viscera. Selectivity is refined by the release of different neurotransmitters. Acetylcholine is the neurotransmitter of the sympathetic and parasympathetic preganglionic neurons. The main postganglionic sympathetic neurotransmitter is norepinephrine.
Visceral afferents transmit conscious sensations (e.g., gut distention and cardiac ischemia) and unconscious visceral sensations (e.g., blood pressure and chemical composition of the blood). Their most important function is to initiate autonomic reflexes at the local, ganglion, spinal, and supraspinal levels. Visceral sensation is carried primarily by the spinothalamic and spinoreticular pathways, which transmit visceral pain and sexual sensations. Brainstem visceral afferents are carried by the vagus and glossopharyngeal nerves. Brainstem visceral afferents are important in complex automatic motor acts such as swallowing, vomiting, and coughing.21
Traumatic spinal cord injury, particularly injury above the T5 level, is associated with severe and disabling cardiovascular, gastrointestinal, bladder, and sexual dysfunction. These individuals have both supine and orthostatic hypotension and are at risk of developing bradycardia and cardiac arrest during tracheal suction or other maneuvers that activate the vagovagal reflexes.
Pure autonomic failure with no other neurologic deficits is rare. More often, autonomic failure occurs in combination with other neurologic disorders, such as Parkinson’s disease, and multiple system atrophy In these individuals, it is important for the examiner to inquire about abnormalities in gait, changes in facial expression, the presence of dysarthria, difficulty in swallowing, and balance problems. Autonomic failure also occurs in individuals with some peripheral neuropathies such as those associated with diabetes. Because autonomic failure may be caused by lesions at different levels of the nervous system, a history of secondary trauma, cerebrovascular disease, tumors, infections, or demyelinating diseases should be established. Additionally, because the most frequent type of autonomic dysfunction encountered in medical practice is pharmacologic, there should be a thorough review of medication use, especially antihypertensive and psychotropic drugs.
Some conditions may be confused with autonomic failure, including neurally mediated syncope, which is referred to as vasovagal, vasodepressor, or reflex syncope. This condition is caused by a paroxysmal reversal of the normal pattern of autonomic activation that maintains blood pressure in the standing position; these individuals do not have autonomic failure. A detailed history is important to differentiate this disorder. In contrast to individuals with chronic autonomic failure in whom syncope appears as a gradual fading of vision and loss of awareness, individuals with neurally mediated syncope often have signs and symptoms of autonomic overactivity such as diaphoresis and nausea before the event. This distinction and the episodic nature of neurally mediated syncope should be part of a thorough clinical history.
The complexity of the nervous system cannot be overstated. The information provided in this overview of the components of the CNS is meant to illuminate the many facets of the system that can be affected by pathologic processes. Fig. 28-19 shows the relationship of the areas that have been described here.41 Familiarity with this relationship is the basis of attempting to understand the pathologies that will be considered in the next chapters. To understand pathology, one must have a good working knowledge of brain structure and function. The Whole Brain Atlas web site provides dynamic images of the brain integrating imaging techniques that link anatomy and pathology.66
Aging and the Central Nervous System
Senescence, or aging, results from changes in DNA, RNA, and proteins. Errors in the duplication of DNA increase with age because of random damage over time. There may be a specific genetic program for senescence. Fibroblasts from an older individual double fewer times than those of an embryo.
Age-related reduction in adult brain weight represents loss of brain tissue. There is highly selective atrophy of brain tissue in the aging CNS. It is not clear how much of the change represents actual loss of nerve cells, since the changes in vascular tissue and glial cells may represent some of the loss. Simple loss of cells is common, Nerve cell shrinking, causing possible changes in functional efficiency, may be a more important effect of old age than cell loss. Nerve conduction velocity decreases with age in both the motor and sensory systems. By the eighth decade there is an average loss of 15% of the velocity in the myelinated fibers.32
The inner structure of the nerve cell changes with aging. The presence of lipofuscin, or wear and tear pigment, a pigmented lipid found in the cytoplasm, may interfere with normal cell function via pressure on the cell nucleus. The pigmented nuclei of the brainstem catecholaminergic neurotransmitter accumulates with age. Damage to an axon close to the neuronal cell body results in changes in the area of the cell body and is referred to as an axonal reaction. The mechanism and relationship to dysfunction are still not clearly understood. The deposition of amyloid-β protein creating plaques in the cerebral cortex is found in many but not all older people. Neuritic, or senile, plaques are found outside the neuron filled with degenerating axons, dendrites, astrocytes, and amyloid. They represent damage to brain tissue. The neuritic plaques are thought to occur most often in the cortex and hippocampus and have been associated with dementia and Alzheimer’s disease.14
Neurofibrillary tangles, or abnormal neurologic fibers that displace and distort the cell body, are found in higher concentrations in the older brain. Neurofibrillary tangles and amyloid are also found in higher concentrations in people with Alzheimer’s disease (see Chapter 31).
The blood supply also diminishes during aging, with a net reduction of 10% to 15%. The relationship of cerebral blood flow, the resultant decrease in the glucose supply to the brain, and decreased metabolism are not well-established as to cause and effect. All three are noted in the aging brain.44
Morphologic changes in the aging brain are accompanied by neurochemical changes. Changes in neurotransmitter activity are seen with aging and are an area of great interest at this time. There is a decrease in the number of some of the receptors, as well as decreases in synthesis of some of the neurotransmitters. Enzymes involved in synthesis of transmitters, such as dopamine, norepinephrine, and acetylcholine, are decreased with age. Changes in these neurotransmitters may be reflected in decreased control over visceral functions, emotions, and attention. Serotonin, involved in central regulatory activities of respiration, thermoregulation, sleep, and memory, appears to be reduced in the older brain. Depression in the older adult may be related to increased production of MAO, which breaks down catecholamines and results in a loss of the feeling of well-being.27
Other changes in the brain related to neurotransmission, such as Parkinson’s and Alzheimer’s disease, are described in the chapters that follow. Mood and depressive symptoms also are common in elderly individuals who are ill and are associated with increased morbidity and mortality. There is a relationship between inflammatory cytokines and depressive disorders. Age-associated alterations in immunity are apparent in the innate immune cells of the brain. There is an elevated inflammatory profile in the aging brain consisting of an increased population of reactive glia. A potential consequence of a reactive glial cell population in the brain is an exaggerated inflammatory response to innate immune activation. Even in the absence of detectable disease, the glia population undergoes an age-related transformation that creates a more sensitive brain environment.
An amplified and prolonged inflammatory response in the aged brain promotes protracted behavioral and cognitive impairments and the behavioral consequences of illness and infection in the elderly, if prolonged, can have deleterious affects on mental health. There is an increased prevalence of delirium in elderly individuals who present to the emergency department as a result of infections unrelated to the CNS. Viral or bacterial pneumonia in the aged frequently presents clinically as delirium, even in the absence of classic pneumonia symptoms.20
The central mechanisms that are involved in the control of balance do not appear to change excessively with age but are more likely to be affected by degenerative neurologic diseases such as Parkinson’s or Alzheimer’s disease. Age-related changes in the peripheral vestibular system include hair cell receptors that begin to decrease at the age of 30 years, and by 55 to 60 years, there is a loss of the vestibular receptor ganglion cells. The myelinated nerve cells of the vestibular system show up to a 40% loss. Partial loss of vestibular function in the older population can lead to complaints of dizziness, with less ability of the nervous system to accommodate the loss compared with younger persons.
In addition to vestibular losses, there is concomitant loss of other sensory inputs relating to balance and mobility: vision and somatosensation. Maintaining equilibrium, or balance, requires a multimodal system integrating vestibular, visual, and somatosensory signals. The integration of these signals in the CNS coordinates multiple output responses: eye movement, postural correction, motor skill, and conscious awareness of spatial orientation. There are longer response latencies and delayed reaction times. Vision changes include loss of acuity, decreased peripheral fields, and loss of depth perception. The loss of input from this combination is slow, with compensation developing through the years.11 Eventually a loss of functional reserve, or redundant function, that is normally present in virtually all physiologic systems is seen with aging. There is an apparent decrease in the ability to integrate conflicting sensory information to determine appropriate postural responses. Changes occur as well in motor output that may contribute to the loss of balance and mobility.10 Although the response patterns are the same in young and old people, with responses being activated in the stretched ankle muscle and radiating up to the thigh, in some older people, this response is disrupted with the proximal muscles being activated before the distal muscles. In the older person, there appears to be more co-contraction of muscles around the ankle as a result of perturbation.53
Neurologic disease is more prevalent in older persons, as is the risk of neurologic sequelae as a result of intracranial hemorrhage, subdural hematoma, and neoplasms. Awareness of the signs and symptoms of these disorders is essential. The therapist may be the person able to identify a disease or the potential for a disorder that may manifest during a treatment session.
DIAGNOSIS
Clinical localization is the first step to differential diagnosis for a individual with neurologic disease. Coupling the time course of the illness with the clinical localization is the essence of neurology. The history of the onset and nature of the symptoms is critical to establish the diagnosis related to the neurologic disorder. In many cases, based on the history and symptoms, the clinician is able to generate a hypothesis regarding the site in the nervous system that has been affected and the nature of the lesion. A complete history of the nature of the symptoms is also critical to determining which diagnostic tools will provide the most accurate differential diagnosis and best determine the cause.
The examination of the client with neurologic dysfunction often begins with mental status changes. Alterations of consciousness and disturbances of higher brain function give the clinician clues about the nature of the disease process and the location of damage within the brain59 (Table 28-4).
Table 28-4
Useful Studies in the Evaluation of Disorder of Level of Consciousness
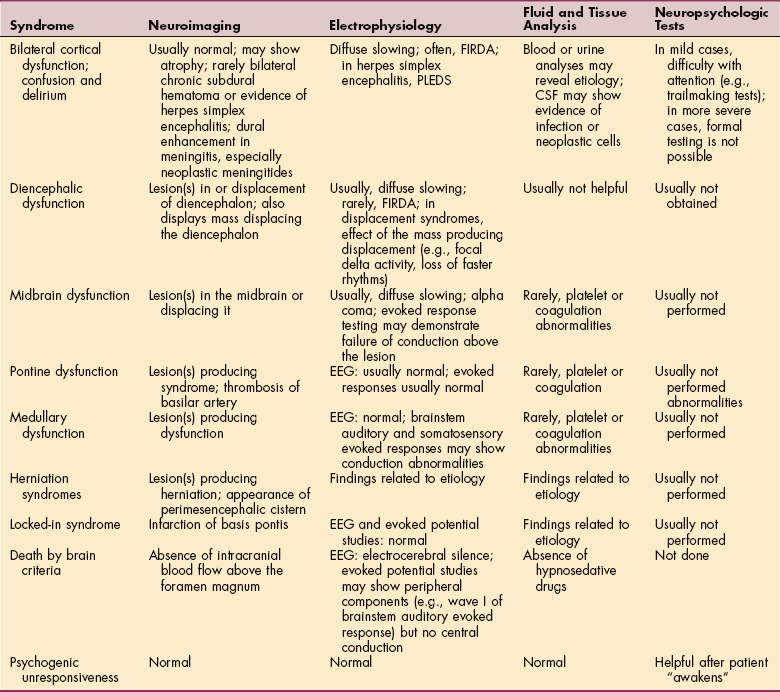
FIRDA, Frontally predominant intermittent rhythmic delta activity; PLEDS, periodic lateralized epileptiform activity; CSF, cerebrospinal fluid; EEG, electroencephalogram.
From Koenigsberg RA, Faro SH, Hershey BL, et al: Neuroimaging. In Goetz CG: Textbook of clinical neurology, ed 2, Philadelphia, 2003, WB Saunders.
Motor and sensory changes will also reflect the type, level, and extent of damage to the system in the case of both disease and trauma. Understanding the typical motor and sensory changes associated with a particular disease or disorder leads the evaluation. For example, knowing that ALS involves both upper and lower motor signs may help the clinician when this otherwise perplexing condition presents in the clinic. Understanding the functional deficits related to each condition can also lead the clinician to a diagnosis. Gait disorders are often representative of the level or location of damage within the nervous system.
The diagnosis of neurologic disorders remains a clinical specialty, although the use of sophisticated imaging and measurement of neural function have provided insight into the pathologic state of the nervous system. The following are examples of diagnostic test currently performed.
Computed Tomography
Computed tomography (CT) scans allows a snapshot of the CNS, and damage within tissue can be identified. Disorders affecting blood flow, multiple sclerosis, neoplasm, and infection can be identified with these scans. CT is an excellent study to evaluate for acute intracranial hemorrhage, particularly in the subarachnoid space. Active bleeding may be detected in either epidural or subdural hemorrhages as a relative lucency, which is commonly referred to as the “swirl sign.” Edema from excitotoxic damage associated with infarct or diffuse anoxia can be seen representing intracellular fluid, and vasogenic edema is the abnormal accumulation of extracellular fluid in the white matter that looks like fingers following the white matter tracts. Evaluation of ventricular size can be done by CT. Enlargement of the temporal horns out of proportion to the lateral ventricular bodies is helpful in recognizing early hydrocephalus. CT can be helpful to follow ventricular size after shunting.
CT is very useful for detecting intracranial calcifications such as those seen in congenital infections, vascular lesions, and metabolic disease. The location and distribution of calcifications is helpful in differentiating these various causes. The identification of calcification in a neoplasm aids in differential diagnosis.21
Magnetic Resonance Imaging
MRI signal patterns are recognizable with common diseases such as cerebral edema, neoplasm, abscess, infarcts, or demyelinating processes. MRI is the study of choice to evaluate all lesions in the brain and spine. CT, however, is more sensitive than MRI for the evaluation of calcifications, subtle fractures, and remains pivotal in the diagnosis of acute subarachnoid hemorrhage. Additionally, MRI cannot be performed in individuals who have intraorbital foreign bodies, pacemakers, or non-MRIcompatible implants, such as artificial heart valves, vascular clips, cochlear implants, or ventilators. The use of MRI is the modality of choice for detecting congenital malformations. Infection of the spine is better evaluated by MRI.
Functional Magnetic Resonance Imaging
fMRI is based on blood oxygenation level–dependent (BOLD) imaging of the brain and provides functional data of cerebral activation during any given task (e.g., motor, visual, or cognitive). This method allows for a quantitative nonclinical method for assessing changes in cerebral function as related to cerebral activities (i.e., performance of physical or cognitive tasks). The BOLD fMRI technique allows for detection of minute changes in cerebral oxygenation. fMRI studies of various pathways of the brain, including the language, memory, and visual pathways, will enhance our ability to connect activity to changes in the brain. For example, frontal and lateral three-dimensional (3D) fMRI renderings show functional activity in the occipital lobe secondary to a visual stimulus.
Positron Emission Tomography
Positron emission tomography (PET) and single-photon emission CT (SPECT) scanning can show cellular activity via regional blood flow in the brain and are now used to monitor changes in the brain with functional activity. Both techniques can be used to depict the regional density of a number of neurotransmitters, allowing researchers to better understand the role of different parts of the brain during activity.
Electroencephalography
Cerebral ischemia produces neuronal dysfunction, leading to slowing of frequencies or reduced amplitude in the EEG tracing. These changes may be generalized (global ischemia) or regional (focal ischemia). The depth of ischemia is associated with the severity of EEG changes. EEG cannot assess the whole cerebral cortex, however, and is less reliable at assessing subcortical structures.
Brainstem Auditory Evoked Potentials
Potentials generated in the auditory nerve and in different regions of the auditory pathways in the brainstem can be recorded. The attention of the subject is not required. Because the brainstem auditory evoked potential (BAEP) is of very low voltage, between 1000 and 2000 responses are generally recorded so that the BAEP can be extracted by averaging from the background noise. Wave III probably arises in the region of the superior olive, whereas waves IV and V arise in the midbrain and inferior colliculus. Waves VI and VII are of uncertain origin and little clinical utility because of their inconsistency in normal subjects. The most consistent components are waves I, III, and V, and it is to these that attention is directed when BAEPs are evaluated for clinical purposes.
The BAEP is an important means of evaluating function of the cranial nerve VIII and the central auditory pathways in the brainstem. In infants, young children, and adults who are unable to cooperate for behavioral testing, BAEPs can be used to evaluate hearing. The wave V component of the response is generated by auditory stimuli that are too weak to generate other components. The BAEP is also useful in assessing the integrity of the brainstem. The presence of normal BAEPs in comatose individuals suggests either that the coma is due to bihemispheric disease or that it relates to metabolic or toxic factors; abnormal BAEPs in this context imply brainstem pathology and a poorer prognosis than otherwise. When coma is due to brainstem pathology, the BAEP findings help in localizing the lesion.
BAEPs have been used to detect subclinical brainstem pathology in individuals with suspected multiple sclerosis. However, the yield in this circumstance is less than with the visual or somatosensory evoked potentials, possibly because the auditory pathway is relatively short or is more likely to be spared. Cerebral ischemia results in delay in the arrival of or reduction in amplitude of evoked responses.21
Transcranial Doppler Ultrasonography
Transcranial Doppler ultrasonography uniquely measures local blood flow velocity in the proximal portions of large intracranial arteries. Hemodynamic compromise is inferred when there is reduction in mean flow velocities or when there is slow flow acceleration. In addition, transcranial Doppler ultrasonography can detect cerebral microembolic signals, reflecting the presence of gaseous or particulate matter in the cerebral artery. Solid, fat, gas, or air materials in flowing blood are larger and of different composition and, thus, have different acoustic impedance than surrounding red blood cells. Thus the Doppler ultrasound beam is both reflected and scattered at the interface between the embolus and blood, resulting in an increased intensity of the received Doppler signal. A completely accurate and reliable characterization of embolus size and composition, however, is not yet possible with current technology.
Near-Infrared Spectroscopy
In brain tissue, the venous oxygen saturation predominates (70% to 80%), and cerebral oximetry relies on this fact. Near-infrared spectroscopy (NIRS) uses light optical spectroscopy in the near-infrared range to evaluate brain oxygen saturation by measuring regional cerebral venous oxygen saturation. Table 28-5 describes the use of various imaging techniques correlated to anatomic site.
Table 28-5
Neuroimaging Applications in Diagnosis and Therapy
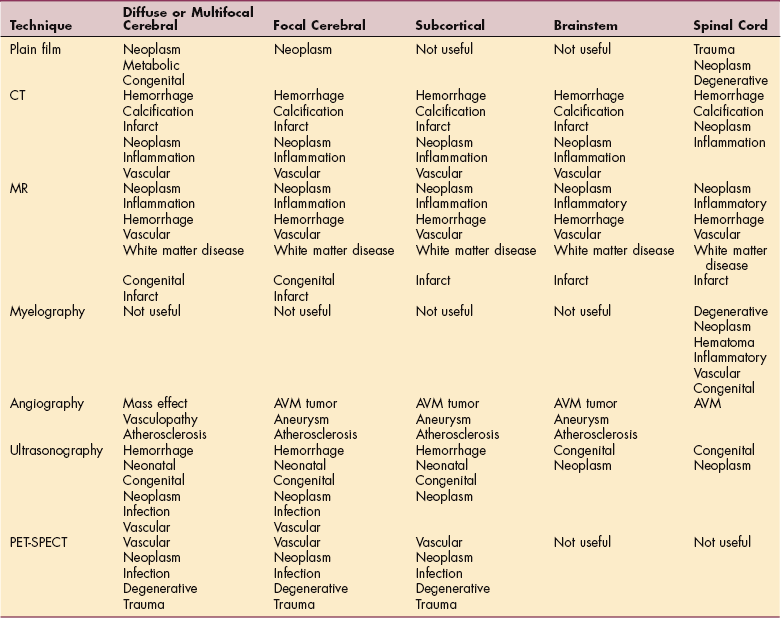
From Koenigsberg RA, Faro SH, Hershey BL, et al: Neuroimaging. In Goetz CG: Textbook of clinical neurology, ed 2, Philadelphia, 2003, WB Saunders. AV M, Arteriovenous malformation; PET-SPECT, positron emission tomography-single-photon emission computed tomography.
TREATMENT
Treatment is based on an understanding of the level and type of neuronal dysfunction. Treatment of neurologic disorders has been a frustrating science in the past, but with better understanding of the cellular processes and changes related to disease, treatment holds more promise.
Methods to Control Central Nervous System Damage
Damage or disease of the nervous system often results in changes in the production and uptake of neurotransmitters. Many important drugs that alter nervous system function act by selective interaction with neurotransmitter receptors. Drugs that act at synapses either enhance or block the action of these neurotransmitters. Most neurotransmitters with a prominent role in brain function produce very brief receptor-mediated actions at specific groups of synapses. A few neurotransmitters are more prolonged and act more widely throughout the extracellular space. The combined action of both a briefly acting and a more enduring neurotransmitter produces a modulation of postsynaptic neuronal activity. Pharmacologic strategies are currently aimed at modulation of neurotransmitter synthesis, release, reuptake, and degradation.25 Some drugs mediate inhibition of neurotransmitter release by acting at presynaptic receptors. Opiates are one group of drugs that act by the inhibition of neurotransmitter release. Drugs used to control excessive tone in specific muscle groups often work by inhibiting neurotransmitter release. Anesthetic drugs modify the actions of neurotransmitter receptors by changing the membranes of cells on or within which the receptors are located.
Drug therapy can stimulate neurotransmitter release. Drugs aimed at maintaining neurotransmitter activity in the synaptic cleft can be useful in neuromuscular junction diseases. Another way to regulate the level of neurotransmitters is to influence the rate of chemical degradation. Drugs can inhibit the breakdown of certain elements that may be broken down by natural processes such as oxidation. One action of these drugs is to prolong the efficacy of released neurotransmitters by inhibiting their degradation.61 An example of this process is the regulation dopamine. Dopamine activity can be increased by four mechanisms:
Synthesis of the neurotransmitter can be increased by giving dopa because it is the product beyond the rate-limiting enzyme and there is ordinarily an abundant amount of aromatic amino acid decarboxylase in the CNS. When dopa is combined with a peripherally active decarboxylase inhibitor, more dopa is delivered across the blood-brain barrier and can be used to synthesize central dopamine. Drugs, such as cocaine, amphetamine, and methylphenidate, can increase release.
The normal metabolism of dopamine involves reuptake of dopamine into the presynaptic cell, with subsequent metabolism by two enzymes, MAO and COMT. Prolongation of dopamine activity can be effected by blocking reuptake or altering enzyme activity. Amantadine and possibly some tricyclic antidepressant medications operate on the dopaminergic system through blockade of reuptake. MAO inhibitors and COMT inhibitors for human use also increase dopaminergic activity. Finally, direct activation of the dopamine receptors on the striatal cell can be induced by agonists like bromocriptine, pergolide, and other drugs. Importantly, orally administered dopamine itself has no place in altering the CNS dopamine levels, because, being a positively charged molecule, it cannot cross the blood-brain barrier.21
Other drugs protect the cell membrane in the presence of toxins that act on the membrane such as the toxic effects of the free radicals produced in brain tissue after hypoxia, ischemia, and seizures. Damage to the neuron occurs when the free radical is allowed to penetrate the membrane.45 The best defense is to prevent penetrance. Antioxidant therapies are being examined for a variety of neurodegenerative disorders and the sequelae of stroke and spinal cord injury. It is believed that the toxicity of glutamate can be blocked by various antioxidants. Vitamin E, a free radical scavenger, is an antioxidant that has been tested widely. However, it does not pass easily through the blood-brain barrier, and as a fat-soluble vitamin, it can be toxic in large doses. Estrogen can work as an antioxidant through intrinsic neurotrophic activities. Ongoing studies are looking at the natural substances noted above as well as manufactured substances that will provide antioxidant or free radical scavenging. There is great hope that substances that will slow down the destruction related to oxidative stress will prove to be curative for progressive diseases of the CNS, as well as other degenerative processes associated with connective tissue, neoplasm, and aging.7 Specific drugs used for primary neurologic disorders are described in the next chapters.
Exciting new advancements in the field of molecular genetics have begun to identify novel candidate genes that may be involved in many inherited or acquired neurologic disorders. The basic principle of gene therapy is to transfer exogenous genes into specific cell types within the human body to correct a pathologic disorder. Currently, gene therapies are based on simple nucleic acid sequences or derived from unique viruses. Researchers have attempted to use the power of viruses to essentially hijack cells for gene expression of appropriate therapeutic molecules.
Stem cells are unspecialized living cells that have the capacity to renew themselves for long periods of time through cell division. Under certain physiologic or experimental conditions, they can be induced to become cells with special functions such as the beating cells of the heart muscle or the insulin-producing cells of the pancreas. Embryonic stem cells are derived from embryos that develop from eggs that have been fertilized in vitro and then donated for research purposes with the informed consent of the donors. The embryos from which human embryonic stem cells are derived are typically 4 or 5 days old and consist of a hollow microscopic collection of cells called the blastocyst. An adult stem cell is an undifferentiated cell found among differentiated cells in a tissue or organ. The primary roles of adult stem cells in a living organism are to maintain and repair the tissue in which they are found. Some researchers now use the term somatic stem cell instead of adult stem cell. Unlike embryonic stem cells, which are defined by their origin, the origin of adult stem cells in mature tissues is unknown. A single adult stem cell could have the ability generate a line of genetically identical cells, or clones.2
There is evidence for the impact of immune system dysfunction in these diseases despite the blood-brain barrier protection of the CNS from the direct effects of autoimmune responses. Identification of immune system elements is leading researchers toward an understanding of the role of the immune system in diseases such as multiple sclerosis, ALS, Parkinson’s disease, and Alzheimer’s disease.
Use of catheters to deliver drugs directly into the cerebrospinal fluid or brain tissue has enhanced the ability to deliver drugs that act directly on the neuron. Although the catheters have been made more sophisticated and can deliver the drugs in measured doses, complications of administration and uneven levels of absorption continue to be limiting factors.
Treatment of Nonneural Dysfunction
Many drugs used to treat neurologic disorders influence nonneuronal tissue, including cerebral blood vessels and glia. Cerebral edema can increase the permeability of the blood-brain barrier, causing an increase in fluid within the brain. The resulting compression of brain tissue can be life-threatening. Drugs, such as mannitol, that control cerebral edema, or drugs that provide diuresis can help preserve neuronal function. In demyelinating disease, antiinflammatory and immunosuppressive drugs are used to preserve the function of the glial cells that produce the myelin sheath.
For some of the viruses that invade the CNS there is replication of cells in nonneural tissue. Use of drugs that inhibit RNA or DNA synthesis can prevent viral replication without disrupting neuronal integrity. Acyclovir, used in the treatment of herpes encephalitis, is an example of this type of drug.
In infants and children, there is an altered drug metabolism that should be considered whenever administering drugs that act on the nervous system. Concomitant illness and fever will further alter drug metabolism. An immature blood-brain barrier can also affect the absorption of drugs into brain tissue. When anticonvulsants are administered, close monitoring of blood levels is necessary.
PROGNOSIS
Prognosis is the keystone to management of neurologic disorders because it links diagnosis to outcomes and identifies need for treatment. Prognostic studies can also identify if available treatment is ineffective. In addition, these studies can indicate which diseases have an important impact on function or disability.34
Disability resulting from neurologic disease and trauma can be extensive, and care of these clients often requires use of limited resources: time and money. With the tremendous advances made in the emergent medical care of trauma victims and people with significant neurologic disease, the number of people living with neurologic disorder is increasing at a steady rate.6
Permanent or progressive impairments can be demoralizing to clients and their families. Clients must reorganize their perspectives in order to learn alternative ways of regaining as much control as possible over life activities. Success builds a sense of efficacy, and failure undermines self-worth. Tackling challenges in successive attainable steps will lead to further competencies in associated tasks. When individuals see others with similar disabilities perform successfully, they may have increased confidence in their own abilities. The persuasion of health care providers and caregivers can boost effort but must be realistic. Perceived self-efficacy can influence the course of health outcomes and functional status. The prognosis for an individual should consider both the social and cognitive status of the individual in relationship to the diagnosis.4
The economic evaluation of health care reflects the complexity of the disease treatment process and the value of health effects. Policy makers are demanding information about the economic outcomes of diseases and their treatments. Research methodologies oriented toward cost-of-illness and cost-benefit analysis have emerged. Clinicians should be involved in this analysis to maintain perspective, especially in catastrophic and degenerative processes.30
Several chronic neurologic disease states, such as Alzheimer’s disease, are associated with elevated secretion of stress hormones such as cortisol. Stress also can trigger or exacerbate symptom onset and perhaps progression of chronic illness such as Parkinson’s disease. Stress hormones also can mitigate the impact of acute neurotrauma; for example, there is a positive correlation between cortisol levels and mortality after head injury. Thus neurologic disease states can occur within a context of elevated glucocorticoids, which may have profound influences on recovery and neuroplasticity. In addition, abnormal regulation of glucocorticoid release is associated with many affective disorders, such as depression and PTSD, that are overrepresented in populations with neurologic disease. Release of glucocorticoids, however, also can occur in anticipation of adverse events, which is a mechanism particularly relevant to neurologists and psychoneuroimmunologists. Anticipatory release of glucocorticoids occurs in the absence of a frank physical stimulus, keyed by memories or instinctual predispositions.
Measures of health-related quality of life address the impact of health on physical, social, and psychologic aspects of life. The particular scale may address issues related to a specific population or may be sensitive to a clinical intervention. The therapist should be familiar with the measurement tools typically used during intervention for a condition or disease. As much as possible, these tools are described in the appropriate chapter in this section.6
Physiologic Basis for the Recovery of Function
After injury to the nervous system, there are changes in the structure and function of the neurons. In some instances the changes can lead to further damage, whereas other changes facilitate recovery.
Diaschisis, or neural shock, occurs when there is injury to a nerve and disruption of the neural pathway that extends a distance from the site of injury. When the neurons distal to the injury regain function, which may be soon after the injury, partial function may return.
Injury may be secondary to either swelling of the axon or edema in the surrounding tissue that blocks synaptic activity in the injured neurons, as well as that in the surrounding area. With reduction of the edema, function may return. This is the reason that medications that reduce edema are often given in the context of diffuse brain swelling.
When there is a loss of presynaptic function in one area, the postsynaptic target cells for that area may become more sensitive to neurotransmitters that are now produced in lower concentrations. The compensatory mechanism is known as denervation supersensitivity. Regenerative synaptogenesis occurs when injured axons begin sprouting. Collateral sprouting is the process of neighboring axons sprouting to connect with sites that were previously innervated by the injured axon.
Suppression of a response to a stimulus is considered habituation, whereas sensitization is an increased response to a stimulus, usually related to noxious stimuli or pain. Adaptation is the ability to modify a motor response based on changes in the sensory environment or input received.
Long-term potentiation occurs when a weak input and a strong input arrive at a dendrite at the same time. The weak stimulus is enhanced by the strong stimulus. With repeated activation of combined stimuli, there is an increase in the presynaptic transmitter associated with the stimulus. After the long-term potentiation has been established, the weak input will elicit a stronger response than it had initially.31
The characteristics of a lesion will have a profound effect on recovery from a brain injury. Small lesions of the brainstem may in fact be as devastating as large lesions of the cerebral cortex. Cerebellar damage can affect both learning and memory of movements. Lesions that occur gradually appear to cause less disruption of function than lesions that occur all at once such as with strokes. Advanced age will adversely affect the return of function. Studies show that a person’s prior level of activity and environment will affect the rate and extent of recovery. An enriched environment will positively affect recovery when it is available either before the insult or during the recovery period.
Redistribution of cortical mapping is seen after a lesion in the brain.9 These changes may involve unmasking of previously nonfunctional synaptic connections from adjacent areas, or the ability of the neighboring inputs may take over. It is clear that both sensory and motor maps in the cortex are constantly changing according to input from the environment. In addition, the brain appears to increase use of ipsilateral pathways after a lesion that affects one side of the brain.
Recovery of function by strict definition is the return to original processes for an activity. When alternative processes are used to complete the task, it is considered compensation. Neural modifiability may be seen as a change in the organization of connections among neurons and is often referred to as plasticity. Some of the recovery after CNS damage is considered spontaneous. Forced recovery is the result of specific interventions that create change in the neural structure.53 Constraint-induced training makes use of this action.
Physiologic studies suggest that motor relearning and recovery of function may be accomplished through the same neural mechanisms and reflect the plasticity of the brain. Learning alters our capability to perform the appropriate motor act by changing both the effectiveness of the neural pathways used and the anatomic connections.
Learning involves storage of memory and can occur in all parts of the brain with both parallel and hierarchic processing. The area of representation within the brain becomes specialized for both inputs and outputs. Areas of the brain used during the early phase of learning movement are different from those used once a skill is learned. Initially, more areas of the brain are active, since skill develops both the number of neurons firing and location of activity change. The use of sensory input is increased in the early stages of learning. The prefrontal areas are also more active in the learning phase and become less active during automatic movements.9 The stimuli repeatedly excite cortical neuron populations, and the neurons progressively grow in numbers. Repetition will lead to greater specificity, and the responses become stronger. With skill acquisition, sensory feedback appears to be less critical.
Control of learning comes from many areas of the CNS working together. The area involved may depend on a number of variables associated with the type of learning taking place and is influenced by the environment. The cortex is involved in learning through sensorimotor integration. It is postulated that there are widely distributed groups of neurons acting as a cortical engram, composed of multiple functional groupings. Thus, when an activity is repeated and stored in memory, the engrams are available to trigger groups of cells that fire synchronously during movement. The engrams appear to influence the precision, speed, and accuracy of movement.31
The limbic system is critical to the learning phase because it generates need-directed motor activity and communicates the intent to the rest of the brain. The limbic system is a critical part of the neural representation necessary for memory that includes the cortex and thalamus.
The cerebellum appears to be active during procedural learning. A possible mechanism is through the influence of the climbing fibers on the mossy fibers with eventual change in the output fibers, the Purkinje fibers.58 The lateral cerebellum affects cognition through its relationship to the frontal areas active during cognitive processes.46
The basal ganglia appear to be highly involved in the cognitive aspects of motor behavior although the level of contribution remains unclear. Habit formation appears to be associated with functions of the basal ganglia, and the control of internally generated movement here appears to be a part of the motor learning continuum.31
References
1. American Physical Therapy Association (APTA) III STEP: linking movement science and intervention. APTA Conference held July 12-15, 2005, Salt Lake City, UT
2. Asbury, AK, McKhann, GM, McDonald, WI. Diseases of the nervous system: clinical neurobiology. Philadelphia: Saunders, 1986.
3. Bach-y-Rita, P. Applications of principles of brain plasticity and training to restore function. In: Young RR, Delwaide PJ, eds. Principles and practice of restorative neurology. Oxford, UK: Butterworth-Heinemann, 1992.
4. Bandura, A. Psychological aspects of prognostic judgments. In Evans RW, Baskin DS, Yatsu FM, eds.: Prognosis of neurological disorders, ed 2, New York: Oxford University Press, 2000.
5. Bastian, AJ. Ataxia: “If only I felt steadier.”. PT Magazine. 1998:62–68. [Continuing Education Series in PT].
6. Batchelor, T, Cook, FE. Clinimetric methods. In: Batchelor T, Cudkowicz C, eds. Principles of neuroepidemiology. Boston: Butterworth Heinemann, 2001.
7. Behl, C, Moosmann, B. Estrogens and other antioxidants in neuroprotection: implications for Alzheimer’s disease. In: Poli G, Cadenas E, Packer L, eds. Free radicals in brain pathology. New York: Marcel Dekker, 2000.
8. Burt, AM. Textbook of neuroanatomy. Philadelphia: Saunders, 1993.
9. Byl, N, Merzneich, MM, Cheung, S, et al. A primate model for studying focal dystonia and repetitive strain injury: effects on the primary somatosensory cortex. Phys Ther. 1999;77:269–284.
10. Chandler, JM, Duncan, PW. Balance and falls in the elderly: issues in evaluation and treatment. In: Guccione AA, ed. Geriatric physical therapy. St Louis: Mosby-Year Book, 1993.
11. Craik, RL. Recovery processes: maximizing function. In: Lister MJ, ed. Contemporary management of motor control problems. Alexandria, VA: American Physical Therapy Association, 1992. [Proceedings of the II Step Conference].
12. Craik, RL. Sensorimotor changes and adaptation in the older adult. In: Guccione AA, ed. Geriatric physical therapy. St Louis: Mosby-Year Book, 1993.
13. Filley, CM. Neurobehavioral anatomy. Niwot, CO: University Press of Colorado, 1995.
14. Fretwell, MD. Aging changes in structure and function. In Carnevale DL, Patrick M, eds.: Nursing management for the elderly, ed 3, Philadelphia: Lippincott, 1993.
15. Frewen, PA. Neurobiology of dissociation: unity and disunity in mind-body-brain. Psychiatr Clin North Am. 2006.
16. Fuller, GN, Goodman, JC. Practical review of neuropathology. Philadelphia: Lippincott Williams & Wilkins, 2001.
17. Gans, RO. The metabolic syndrome, depression, and cardiovascular disease: interrelated conditions that share pathophysiologic mechanisms. Med Clin North Am. 2006;90(4):573–591.
18. Gagnon, JF. Rapid-eye-movement sleep behaviour disorder and neurodegenerative diseases. Neurology. 2006;5(5):424–432.
19. Gill-Body, KM, et al. Rehabilitation of balance in two patients with cerebellar dysfunction. Phys Ther. 1997;77:534–552.
20. Godbout, JP. Age and neuroinflammation: a lifetime of psychoneuroimmune consequences. Neurol Clin. 2006;24(3):521–538.
21. Goetz, CG. Textbook of clinical neurology, ed 2. Philadelphia: WB Saunders, 2003.
22. Hansson, H, Muyderman, H, Leonova, J, et al. Astroglia and glutamate in physiology and pathology: aspects on glutamate transport, glutamate-induced cell swelling and gap-junction communication. Neurochem Int. 2000;37:317–329.
23. Herman, JP. Hypothalamic-pituitary-adrenal axis, glucocorticoids, and neurological disease. Neurol Clin. 2006;24(3):461–468.
24. Hikosaka, O, et al. Parallel neural networks for learning sequential procedures. Trends Neurosci. 1999;22:464.
25. Johnston, MV, Silverstein, FS. Fundamentals of drug therapy in neurology. In: Johnston MV, Macdonald RL, Young AB, eds. Principles of drug therapy in neurology. Philadelphia: Davis, 1992.
26. Kandel, ER. Nerve cells and behavior. In Kandel ER, Schwartz JH, Jessell TM, eds.: Principles of neural science, ed 4, New York: McGraw-Hill Health Professions Division, 2000.
27. Katz, PS. What are we talking about? Modes of neuronal communication. In: Katz PS, ed. Beyond neurotransmission: neuromodulation and its importance for information processing. New York: Oxford University Press, 1999.
28. Kimble, M. Clinical correlates of neurological change in posttraumatic stress disorder an overview of critical systems. Psychiatr Clin North Am. 2004;27(1):49–65.
29. Lang, CE, Bastian, AJ. Cerebellar subjects show impaired adaptation of anticipatory EMG during catching. J Neurophysiol. 1999;82:2108.
30. Langfitt, JT, Holloway, RC. Economic prognosis: evaluating economic outcomes of health care. In Evans RW, Baskin DS, Yatsu FM, eds.: Prognosis of neurological disorders, ed 2, New York: Oxford University Press, 2000.
31. Leonard, CL. The neuroscience of human movement. St Louis: Mosby, 1998.
32. Lewis, CB, Jordan, KS. Clinical implications of neurological changes with age. In: Lewis CB, ed. Aging: the health care challenge. Philadelphia: Davis, 1985.
33. Lindsay, KW, Bone, I, Callandar, R. Neurology and neurosurgery illustrated. New York: Churchill Livingstone, 1986.
34. Longstreth, WT, Koepsell, TD, Nelson, LM, et al. Prognosis: keystone of clinical neurology. In Evans RW, Baskin DS, Yatsu FM, eds.: Prognosis of neurological disorders, ed 2, New York: Oxford University Press, 2000.
35. Mataix, M, Coles, D. Common and distinct neural correlates of obsessive-compulsive and related disorders. Psychiatr Clin North Am. 2006;29(2):391–410.
36. Mattson, MP, Duan, W, Chan, SL, et al. Apoptotic and antiapoptotic signaling at the synapse: from adaptive plasticity to neurodegenerative disorders. In: Shaw CA, McEachern JC, eds. Toward a theory of neuroplasticity. Lillington, NC: Taylor & Francis, 2001.
37. McDonough-Means, SI, Cohen, MW. Attention deficit disorder. In: Rakel D, ed. Integrative medicine. Philadelphia: WB Saunders, 2003.
38. Melnick, ME, Omreland, B. Movement dysfunction associated with cerebellar problems. In Umphred DA, ed.: Neurological rehabilitation, ed 4, St Louis: Mosby, 2001.
39. Monarch, ES. Neuropsychological impairment in borderline personality disorder. Psychiatr Clin North Am. 2004;27(1):67–82.
40. Newton, RA. Contemporary issues and theories of motor control: assessment of movement and posture. In Umphred DA, ed.: Neurological rehabilitation, ed 4, St Louis: Mosby, 2001.
41. Nolte, J. The human brain: an introduction to its functional anatomy, ed 5. St Louis: Mosby, 2002.
42. Norenberg, MD. Astrocyte responses to CNS injury. J Neuropathol Exp Neurol. 1994;53:213–220.
43. Panter, SS, Faden, AI. Biochemical changes and secondary injury from stroke and trauma. In: Young RR, Delwaide PJ, eds. Principles and practice of restorative neurology. Oxford, UK: Butterworth-Heinemann, 1992.
44. Poirier, J, Finch, CE. Neurochemistry of the aging human brain. In Hazzard WR, Andres R, Bierman EL, et al, eds.: Principles of geriatric medicine and gerontology, ed 2, New York: McGraw-Hill, 1990.
45. Poli, G, Cadenas, E, Packer, L. Free radicals in brain pathology. New York: Marcel Dekker, 2000.
46. Ryding, E, Decety, J, Sjöholm, H, et al. Motor imaging activates the cerebellum regionally. A SPECT rCBF study with 99mTc-HMPAO. Brain Res Cogn Brain Res. 1993;1:94–99.
47. Scaer, RC. The body bears the burden: trauma, dissociation, and disease. New York: Haworth Medical Press, 2001.
48. Siegel, DJ. The developing mind: toward a neurobiology of interpersonal experience. New York: The Gilford Press, 1999.
49. Shore, AN. Affect regulation and repair of the self. New York: WW Norton & Company, 2003.
50. Schmidt, RA. Motor control and learning: a behavioral emphasis, ed 2. Champaign, IL: Human Kinetics, 1988.
51. Schmidt, RA. Motor learning principles for physical therapy. In: Lister MJ, ed. Contemporary management of motor control problems. Proceedings of the II Step Conference. Alexandria, VA: American Physical Therapy Association, 1992.
52. Seigelbaum, SA, Koester, J. Ion channels. In Kandel ER, Schwartz JH, Jessell JH, eds.: Principles of neural science, ed 4, New York: McGraw-Hill, 2000.
53. Shumway-Cook, A, Woollacott, MH. Motor control: theory and function. Philadelphia: Lippincott, Williams & Wilkins, 2001.
54. Singer, C, Weiner, WJ. The neurological examination. In Weiner WJ, Goetz CG, eds.: Neurology for the non-neurologist, ed 3, Philadelphia: Lippincott, 1994.
55. Smith, CU. Elements of molecular neurobiology. London: John Wiley & Sons Ltd, 2002.
56. Stahl, SM. Essential psychopharmacology: neuroscientific basis and practical applications, ed 2. New York: Cambridge University Press, 2000.
57. Swartz, JH. Neurotransmitters. In Kandel ER, Schwartz JH, Jessell JH, eds.: Principles of neural science, ed 4, New York: McGraw-Hill, 2000.
58. Thach, WT. A cerebellar role in acquisition of novel static and dynamic muscle activities in holding, pointing, throwing, and reaching. In: Bloedel JR, Ebner TJ, Wise SP, eds. The acquisition of motor behavior in vertebrates. Cambridge, MA: MIT Press, 1996.
59. Topel, JL, Lewis, SL. Examination of the comatose patient. In Weiner WJ, Goetz CG, eds.: Neurology for the non-neurologist, ed 3, Philadelphia: Lippincott, 1994.
60. Topka, H. Motor skill learning in patients with cerebellar degeneration. J Neurol Sci. 1998;158:164.
61. Traub, M, Haigh, JR, Marsden, CD. Recent developments in the pharmacotherapy of major neurological dysfunction. In: Young RR, Delwaide PJ, eds. Principles and practice of restorative neurology. Oxford: Butterworth-Heinemann, 1992.
62. Umphred, DA. The limbic system: influence over motor control and learning. In Umphred DA, ed.: Neurological rehabilitation, ed 4, St Louis: Mosby, 2001.
63. Umphred, DA, Byl, N, Lazaro, RT, et al. Interventions for neurological disabilities. In Umphred DA, ed.: Neurological rehabilitation, ed 4, St Louis: Mosby, 2001.
64. Watkins, LR, Milligan, ED, Maier, SF. Glial activation: a driving force for pathological pain. Boulder, CO: Department of Psychology & the Center for Neurosciences, University of Colorado at Boulder, 2000.
65. Waxman, SG, deGroot, J. Correlative neuroanatomy. East Norwalk, CT: Appleton and Lange, 1995.
66. Whole Brain Atlas http://www.med.harvard.edu/AANLIB/home.html
67. Winstein, CJ. Knowledge of results and motor learning: implications for physical therapy. Phys Ther. 1991;71:140–149.