28 Dermatophytosis
INTRODUCTION
Dermatophytosis is a superficial infection of the skin by pathogenic dermatophytes belonging to species Microsporum, Trichophyton and Epidermophyton, which are known to invade keratinized tissues in animals and humans. The dermatophytes are classified according to their natural habitats, i.e. animals, humans or soil. Zoophilic species such as M. canis and T. equinum are adapted to inhabit animals, although the spores can survive in the environment for long periods. Anthropophilic dermatophytes are adapted to man, and these species include Epidermophyton spp. and M. audouinii. Geophilic dermatophytes inhabit the soil and include M. gypseum and M. nanum. Dermatophytosis is a zoonotic condition and therefore vets require an understanding of the pathogenesis of the disease, not only as regards its treatment in animals, but also as regards limiting further infections in both people and animals. This case report describes the steps involved in the diagnosis and management of cats infected with M. canis.
CASE HISTORY
Although dermatophytosis can occur at any age, younger animals are more predisposed to infection. The condition is spread between animals or from environmental contamination of the hair coat. Studies have reported a higher incidence in rescue centres, in show cats and in breeding establishments. There is reported breed predisposition in Persian cats.
CLINICAL EXAMINATION
Feline dermatophytosis can present not only with a variety of different lesions, but also with different reaction patterns, which are all common to other diseases seen in this species. The alopecia may be circumscribed or diffuse, with patches of scaling, hyperpigmentation, erythema and comedone formation, which may be present on the head or on the extremities. The hairs on the margin of lesions may be broken. In some feline cases clinical reaction patterns such as miliary dermatitis, eosinophilic granuloma complex and chin acne have been associated with dermatophyte infection. Dermatophyte pseudomycetoma, a form seen in long-haired cats, is characterized by subcutaneous nodules, which may ulcerate and/or discharge.
In dogs the clinical signs, although variable, are mainly associated with hair loss. The lesion distribution may be localized or diffuse. Circular patches of alopecia, scaling, hyperpigmentation, crusts and/or follicular papules may be seen on the head and the extremities (see Chapter 9). The lesions are usually well demarcated. Kerion, an inflammatory nodule seen in dogs, is a result of concurrent fungal and bacterial infection. Symmetric nasal or facial lesions that mimic autoimmune disease in rare cases may be associated with dermatophyte infections caused by M. persicolor or by T. mentagrophytes.
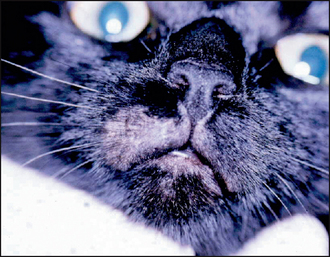
In this case the examination was performed wearing disposable gloves, because from the history dermatophytosis was high on the list of differential diagnoses. A dermatological examination in this case revealed a single expanding patch of alopecia with broken hairs at the periphery on the upper lip (Fig. 28.1). The lesion surface was scaly but non-inflammatory. The general physical examination was within normal limits.
CASE WORK-UP
A diagnosis is made on the history, clinical signs and laboratory tests. In-house screening tests may be carried out to demonstrate the presence of a dermatophyte but the services of a commercial laboratory are likely to be required to speciate the fungus. Knowing the species will help in formulating a strategy both for treatment and control of the fungus within the environment. In-house screening tests include Wood’s lamp examination (Fig. 28.2), direct microscopy and culture on dermatophyte test medium (DTM), and are discussed in detail in Chapter 2. All these tests are dependent on good technique for their success and have their pitfalls.

Figure 28.2 Example of apple green fluorescence of M. canis-infected hairs on Wood’s lamp examination.
Dermatophytosis can mimic almost any other dermatosis, particularly in cats. The differential diagnosis in this case included:
The following tests were performed:
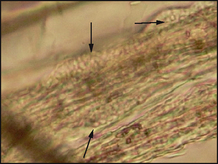
Figure 28.3 Microscopic examination of hair plucks in liquid paraffin mount showing ectothrix arthrospores (arrowed).
Definitive diagnosis is made on the cultural characteristics and on the microscopic morphology of the fungal hyphae and macroconidia. Microsporum canis appears as cotton wool-like white colonies and the reverse side of the growth medium has a yellow–orange pigment that turns brown. The morphology of the macroconidia is determined by the flag technique. Take a small square of clear adhesive tape and place it on the surface of the colony using a pair of rat tooth forceps. Then place the tape on a microscope slide on which there is a drop of lactophenol cotton blue stain (do not forget to disinfect the instrument with heat or chemical disinfection). Microsporum canis macroconidia contain six or more cells and are spindle shaped with thick walls which form a knob at one end (Fig. 28.4). This technique can also be used on DTM medium to look for macroconidia.
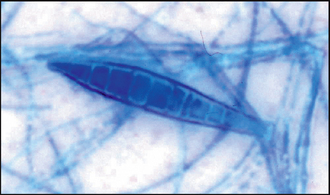
Figure 28.4 Macroconidia from M. canis showing thick walls, more than six cells and a knob at one end.
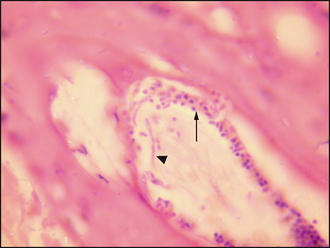
Skin biopsies may indicate the presence of fungal elements (Fig. 28.5), but do not indicate the species involved; therefore, they are of limited value as a diagnostic aid, except where fungal infections mimic autoimmune disease, or for deep bacterial infections. Some cases may require special staining with PAS to demonstrate the presence of hyphae or spores. Histopathology is a useful tool to rule out alopecia areata and pseudopelade. In this case, given the history, clinical signs and the supporting evidence from the microscopic examination of hairs, histopathological examination was not indicated.
AETIOPATHOGENESIS OF DERMATOPHYTOSIS
Dermatophytes invade keratinized tissue such as hair, stratum corneum and nails. Dermatophyte infections are acquired by contact with arthrospores from fomites, infected soil and infected animals. The first step in the process of infection is the adherence of the arthrospores to the stratum corneum. The second step is the germination of the spore and the production of hyphae, which secrete keratinase and invade the stratum corneum and anagen hair shafts.
Dermatophytosis is a contagious disease, and infections can spread between species by both direct and indirect contact if the predisposing conditions are favourable. These predisposing factors include both host and environmental factors.
Host predisposing factors
Young, older and immunosuppressed animals appear to be the most susceptible to infection. Cats with FIV or FeLV and animals receiving chemotherapeutic drugs may be at risk. Genetic susceptibility also may be an important predisposing factor, especially for Persian cats. Cats that have recovered from infections remain susceptible, but the time taken for recovery from a repeat infection is shorter and the disease is less severe. It is thought that the mechanical removal of spores by grooming in cats may provide a natural defence against infection. It follows that long-haired cats, including Persians, could be at higher risk, because the length of their coat may inhibit such removal. Matted fur, in older animals, may also trap spores and prevent their removal by grooming.
Innate immunity
Innate immunity, or non-specific immunity, is the body’s first line of defence against infection. Transferrin, complement and neutrophils are involved in protecting against infection in its early phase. Transferrin, an iron-binding protein found in the epidermis, inhibits fungal growth by limiting the availability of iron. Complement is activated via the alternative pathway and this also inhibits fungal growth. Dermatophyte penetration into tissue will recruit neutrophils to the site within hours, where they too will inhibit fungal growth.
Cell-mediated immunity
Cell-mediated immunity provides the main mechanism for protection against, and elimination of, infection. The mechanism is not fully understood, but this is a T-cell-mediated response and involves the release of cytokines that both increase keratinocyte turnover and so aid in physical expulsion of the fungus, and also result in up-regulation of the inflammatory response directed against the fungus.
Humoral immunity
Humoral immunity, which involves production of antidermatophyte antibodies, provides little protection to the individual. In fact, some dermatologists argue that high levels of antibodies may prevent the cell-mediated response and could therefore result in chronic infections.
Behavioural factors
Whatever the level of spores in the environment, breakdown of the epidermal barrier is also a prerequisite for infection to occur. This barrier may be breached by trauma either by accident or during grooming. Microtrauma, caused by self-grooming and by ectoparasites, has been suggested as a predisposing factor in cats.
Environmental predisposing factors
Temperature and humidity are important predisposing factors, which explains the higher incidence of infection in geographical locations where the climate favours fungal growth. In some species, such as horses and cattle, excessive wetting leading to maceration is also considered a predisposing factor.
Spores in the environment can be a constant source of infection, as they may remain viable for years under natural conditions and play a substantial role in spreading infection in cat colonies.
EPIDEMIOLOGY
Reports of the incidence of the disease vary throughout the world. The variation can perhaps be explained by local breed predisposition effects, and a range of environmental factors which affect the survival of the organism. Transmission of disease is either by direct contact, with an infected or a carrier animal, or by indirect contact, via a contaminated environment.
Most of the dermatophyte infections in the UK in small animals are caused by M. canis. A survey based on a study of hair samples collected over 35 years from cats suspected of having dermatophytosis found that dermatophytes were isolated from 26% of samples, of which 92% were M. canis. Studies on the isolation of M. canis from asymptomatic cats have been reported to range from 0% to 88% and such studies in the UK showed an isolation rate of 2.2% in the south-west of England and 2.16% in the south-east.
PROGNOSIS
The prognosis is good, but owners should be made aware of the disease’s zoonotic potential, and the costs and the time involved in achieving a cure. Mycological cure can be achieved even in cattery situations.
TREATMENT OPTIONS
Although dermatophytosis is a self-limiting disease, it should be treated because of its zoonotic potential, especially to children and immunocompromised persons. Treatment also reduces the time it takes to resolve the condition and, in addition, it reduces the consequent environmental contamination, an important factor in the pathogenesis of the disease. Pet animals with dermatophytosis should be treated topically and systemically. In addition, measures to reduce the environmental contamination should be taken. Many of the antifungal agents mentioned here do not have a veterinary licence in the UK and therefore their use in the UK requires the owner’s informed consent.
Systemic treatment
Systemic medication is recommended in all cases of dermatophytosis in small animals. Several different drugs are available for systemic use; itraconazole is the only one licensed for cats at present.
One of the difficulties in treating dermatophytosis is defining the end point of treatment. This is because there will be clinical resolution of lesions before the fungus has been eliminated from the skin and it is important to continue treatment until mycological cure is achieved. This is defined as two negative fungal cultures with an interval of a month between the tests.
Itraconazole: Itraconazole, a triazole antifungal agent, is the treatment of choice. It is administered with food, for improved absorption, at 5–10 mg/kg, once daily. The treatment should be pulsed, one week on and one week off, over 6 weeks or until mycological cure. Vomiting is a rare side-effect and it is generally better tolerated than ketoconazole. It exerts its effect by inhibiting the demethylation of lanosterol, a precursor sterol in the synthesis of ergosterol, which is a component of the fungal cell wall. It is available as a liquid formulation of 10 mg/ml.
Griseofulvin: Griseofulvin inhibits the growth of various species of Microsporum, Trichophyton and Epidermophyton. It is available in two formulations, a micro-size and an ultra-micro-size. The latter is commonly used for small animals at the dose of 10–15 mg/kg daily, with a fatty meal to enhance absorption. Therapy should be continued until two consecutive negative mycological cultures are obtained. Griseofulvin inhibits fungal cell mitosis by disruption of the mitotic spindle; it may also inhibit chitin synthesis. Adverse effects are not uncommon. The most common side-effects are vomiting, diarrhoea and anorexia. In cats, especially those with FIV, bone marrow suppression resulting in anaemia and leucopenia may occur. It is also teratogenic in humans and animals, and must not be used in very young or pregnant animals. Women of child-bearing age must wear gloves when handling the medication. It is no longer available as a licensed treatment for small animals in the UK.
Ketoconazole: Ketoconazole, an imidazole, is effective against dermatophyte infections. It is better absorbed at an acid pH, and so antacids and H2 receptor antagonists, such as cimetidine, should not be used concurrently. Ketoconazole is administered at the dose of 5–10 mg/kg orally s.i.d. Adverse effects include anorexia, increased liver enzymes and it may also suppress basal cortisol levels. Its mode of action is the same as itraconazole.
Terbinafine: Terbinafine is an allylamine antifungal, which is effective against dermatophytosis. The suggested dose is 20–30 mg/kg orally, once daily. Terbinafine blocks the synthesis of ergosterol by inhibiting the enzyme squalene epoxidase in the membrane. Potential side-effects include anorexia, raised liver enzymes and GIT disturbances. It is not licensed for veterinary use in the UK, and its use requires consideration of the prescribing cascade and informed written consent.
Lufenuron: The use of lufenuron, a chitin synthase inhibitor, remains controversial. Following the initial case report of good efficacy when administering lufenuron at 50–60 mg/kg, several further studies have reported much less encouraging results. Lufenuron failed to prevent infection, or alter the course of infection, in two blinded studies. In addition, lufenuron has failed to exhibit any antifungal activity in in vitro studies, and therefore at present its use for control or treatment of dermatophytosis is not recommended.
Vaccine: A killed M. canis vaccine is licensed for use in the USA and some European countries. It does not prevent infection, but is known to reduce the severity of the lesions after infection by M. canis. If the vaccine is used following infection, it hastens the resolution of clinical lesions, but not mycological cure. It may be of some value in cattery situations.
Topical treatment
Topical antifungal preparations containing miconazole, clotrimazole and enilconazole are available as shampoos, lotions, creams or rinses.
Clipping
Long-haired animals, especially Persian cats, or animals with generalized lesions should be clipped before application. Preferably this should be performed in an isolated area that can be easily decontaminated. To clip or not to clip is controversial; however, with due care, clipping removes the spores found in the hair coat and thereby reduces the load on the animal and further environmental contamination. Additionally, it makes it easier to apply topical therapies.
Environmental control
Environmental decontamination is an important adjunct to clinical treatment, since fungal spores can survive for prolonged periods away from the animal. Furthermore, spores can become airborne and are then spread by air currents. The number of animals infected, as well as the number of recurrent infections in the same animal, is directly related to the levels of spores in the environment. It is therefore easier to deal with infections in single-animal households. In multi-cat households and catteries, or kennels, additional measures are required.
The recommended procedures to control environmental contamination include:
The environmental decontamination of an infection involving kennels or a cattery is involved and expensive. The aim in such situations is long-term eradication, and detailed protocols for such situations are beyond the scope of this case report (see Further reading).
Treatment in this case
This case was treated with griseofulvin at the dose of 30 mg/kg orally given with a fatty meal. The affected area was washed in chlorhexidine/miconazole shampoo every day and a full body wash was done once a week (N.B. this cat was treated prior to the availability of itraconazole, the current licensed treatment for dermatophytosis in cats in the UK). The cat in this case was confined to the utility room to limit contamination, and it was an area that could be easily wiped with household bleach diluted 1 : 10.
The treatment was continued until 2 weeks after the second successive toothbrush sample tested negative for fungal culture. In total, the cat was treated for 7 weeks.
Barrier nursing and disinfection at the surgery premises should be paramount, as with any infectious disease, especially if the cat is clipped in the practice.