9 Dermatophytosis in a Jack Russell terrier
INTRODUCTION
Dermatophytosis is a fungal invasion of keratinized tissue of the stratum corneum, hair or claws by Trichophyton, Epidermophyton or Microsporum spp. In small animals, dermatophytosis is most frequently associated with infections caused by Trichophyton or Microsporum spp. Dermatophytosis results in variable alopecia, erythema, scaling, crusting and pruritus. This case report describes a case of pruritic dermatophytosis caused by infection with Trichophyton erinacei.
CASE HISTORY
There are aspects of the history that might alert the clinician to the possibility of dermatophytosis. These would include: breed predispositions to dermatophytosis (see ‘Epidemiology’ section); a history of exposure to a zoophilic or geophilic source of infection, such as a terrier breed hunting hedgehogs; evidence of contagion in contact animals or lesions on the owner suggestive of zoonosis; and the onset of skin disease in an animal with no previous history of skin problems. If there is pruritus, the appearance of alopecia or scaling prior to the onset of pruritus would suggest that this was not an allergic aetiology. A poor response to glucocorticoids, antibacterial or antiparasitic therapy could also be suggestive of dermatophytosis.
The relevant aspects of the history were:
CLINICAL EXAMINATION
There was mild peripheral lymphadenopathy, but there were no other abnormalities on general physical examination.
Examination of the skin revealed extensive patches of relatively well-demarcated alopecia, erythema, hyperpigmentation, and crusting and scaling over the muzzle, periorbital skin, the left dorsal trunk, the lateral thighs and the forelimbs (Figs 9.1, 9.2 and 9.3).
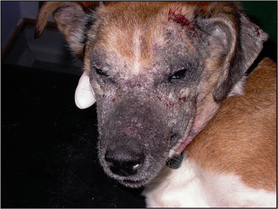
Figure 9.1 Well-demarcated alopecia, hyperpigmentation, scaling and crusting over the face. The nasal planum is unaffected.
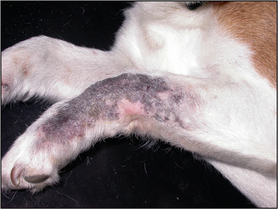
Figure 9.2 Well-demarcated patch of alopecia, erythema and hyperpigmentation over the left carpal area.
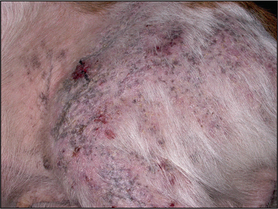
Figure 9.3 Patchy alopecia, multifocal post-inflammatory hyperpigmentation and erythema over the left hind limb.
Multifocal patches of apparently spreading alopecia are suggestive of a folliculitis. The three common causes of folliculitis are demodicosis, pyoderma and dermatophytosis. In this case, the signalment, history and clinical presentation were highly suggestive of dermatophytosis resulting from infection with a Trichophyton spp.
The list of possible differential diagnoses included:
In a case like this, hypothyroidism and hyperadrenocorticism, or another underlying immunosuppressive disease, might predispose it to the development of pyoderma, demodicosis or dermatophytosis.
CASE WORK-UP
Multiple deep skin scrapings should be performed in any case where demodicosis is suspected. In this case there were crusting lesions and cytology was also indicated to identify the presence or absence of bacterial infection.
The following diagnostic tests were performed:
DIAGNOSIS
The history, clinical signs and fungal culture were consistent with a diagnosis of dermatophytosis, due to an infection with Trichophyton erinaciei.
PROGNOSIS
In general, the prognosis for treating dermatophytosis is good, but persistent infections do occur, particularly where there is immunosuppression. In cases of dermatophytosis resulting from Trichophyton spp., scarring resulting in permanent loss of hair over severely affected areas is a common sequela.
AETIOPATHOGENESIS OF TRICHOPHYTOSIS
Depending on their adaptation to soil, animal or man, dermatophytes may be classified as geophilic, zoophilic or anthrophilic respectively. The most commonly isolated dermatophyte pathogens in dogs and cats are Microsporum canis, M. gypseum and T. mentagrophytes. Infection requires direct transmission of spores to a susceptible host and transmission varies with the species of fungi. In the case of M. canis, it is usually by direct contact with an infected cat, or via contact with infected grooming utensils, fomites or an otherwise contaminated environment. Infection may also be acquired from direct or indirect contact with an infected or carrier person. Trichophyton spp. infection usually results from contact with an infected animal (in this case a hedgehog) and M. gypseum through contact with infected soil. Fungal spores are extremely resistant and the spores of M. canis may last up to 18 months within the environment.
Dermatophytes have been isolated from the skin and hair coat of healthy cats and dogs, reflecting asymptomatic carriage of the fungus. Thus, mere exposure to the fungus does not mean infection. Factors that favour the establishment of an infection include exposure to an increasing mass of fungal spores, increased skin humidity, mechanical disruption of the stratum corneum and compromised host immunosurveillance.
In an experimental model of M. canis dermatophytosis in cats, the time from inoculation to establishment of the lesion was in the region of 7–14 days. Dermatophytes require a source of newly synthesized keratin to survive and therefore invade the hair shaft or, in the case of M. persicolor, the keratinized cells of the stratum corneum. Fungal hyphae invade the hair follicle ostia, proliferate on the surface of the hair shaft and migrate downwards towards the hair bulb. Most infections involve ectothrix invasion of hair shafts, where fungal spores accumulate on the surface of hair shafts. The fungus produces keratinases that allow invasion of the hair shaft. Invasion of the hair shaft continues up to the keratogenous zone known as Adamson’s fringe. The fungus then reaches a state of equilibrium with hair growth until the hair is eventually expelled. The initial growth and invasion of the fungus in the hair shaft must be ‘faster’ than loss of keratin tissues to prevent it being shed.
Spontaneous resolution of an infection is depen-dent on the host mounting an effective cell-mediated immune response. To check this response, some fungi produce substances (mannans) that inhibit cell-mediated immunity.
EPIDEMIOLOGY
In UK small animal practice, dermatophytosis is an uncommon skin disease. Various epidemiological factors have been recognized that tend to increase the incidence of dermatophytosis. They are:
It should be borne in mind that M. canis infections are more common in animals from urban areas, whereas T. mentagrophytes and M. gypseum infections are seen more frequently in animals from rural environments.
TREATMENT OPTIONS
Although dermatophytosis in healthy dogs and cats can undergo spontaneous remission within 3 months, a case of generalized dermatophytosis requires systemic antifungal therapy. There is little published information on the treatment of dermatophytosis caused by Trichophyton spp., but anecdote and clinical experience indicates that this disease may be more refractory to treatment, compared to infections with Microsporum canis. The current options for systemic antifungal therapy include the azoles ketoconazole, itraconazole and fluconazole, and the allylamine terbinafine. Griseofulvin is no longer available as a licensed treatment in the UK (Chapter 28).
Azoles: The azoles inhibit fungal lanosterol 14-demethylase, a cytochrome P-450 enzyme. They also inhibit intracellular triglyceride, phospholipid and cell wall chitin synthesis. Their potency and potential toxicity are related to their affinity to bind to fungal, versus mammalian, cytochrome P-450 enzymes. Ketoconazole, itraconazole and then fluconazole show increasing specificity for fungal as opposed to mammalian enzyme systems, and therefore itraconazole and fluconazole show increased antifungal potency and decreased side-effects compared to ketoconazole. Because of their effects on mammalian cytochrome P-450 enzymes, azoles have the potential to inhibit the metabolism of other medications and there is a significant potential for drug interactions. The azoles, particularly itraconazole, are lipophilic and keratinophilic and achieve persistent high concentrations within the stratum corneum and hair follicle, which makes them suitable for pulse therapy.
For the treatment of canine dermatophytosis, ketoconazole is administered at dosages of 10–20 mg/kg/day either as a single dose or divided twice daily. Absorption is enhanced by administration with food, to achieve an acid gastric pH. The most common adverse reaction is gastrointestinal disturbance with vomiting and/or diarrhoea, but higher doses may result in hepatotoxicity. Itraconazole is more expensive than ketoconazole but has less potential for toxicity. It may be administered at a dosage of 5–10 mg/kg q24 for the treatment of canine dermatophytosis.
Allylamines: Terbinafine, an allylamine, has been shown to be effective in the treatment of dermatophytosis; however, it is not licensed for this use in the UK. It inhibits the synthesis of ergosterol, an important component of fungal cell walls. It is also lipophilic and keratinophilic, and reaches high concentrations in the stratum corneum and in hard keratin structures such as nails. The dosage in dogs is 20–30 mg/kg every 24 hours. Elevation in ALT has been observed at this dosage, although there were no signs of clinical toxicity.
Topical therapy: Topical therapy is also indicated for the treatment of dermatophytosis as it may hasten the resolution of infection and prevent environmental contamination with fungal spores. The decontamination of the affected animal is also important to decrease the incidence of zoonotic infection.
Clipping, particularly of long-haired animals, should be considered as part of the treatment of dermatophytosis. Clipping reduces environmental contamination with fungal elements and is particularly helpful in the treatment of M. canis infection (see Section 3, Chapter 24).
Chosen treatment: Because there was evidence of pyoderma, and pending the results of the fungal culture, antibiotic treatment was started using cefalexin at a dosage of 25 mg/kg b.i.d., along with twice weekly chlorhexidine and miconazole shampoo therapy.
Following confirmation of the diagnosis, systemic antifungal therapy was introduced using itraconazole at a dosage of 10 mg/kg s.i.d. per os.
As this was a short-haired dog, clipping was not performed, but weekly chlorhexidine and miconazole shampoos were continued.
Dermatophytosis has a very varied clinical appearance. Historically, dermatophytosis has been over-diagnosed because the classic, ‘ringworm’ lesion consisting of a circular patch of alopecia with an erythematous margin is much more likely to be due to a pyoderma than a fungal infection. However, dermatophytosis should be considered in the differential diagnosis of any annular, papular or pustular eruption.
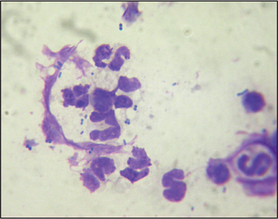
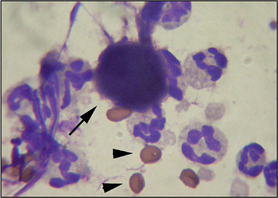
Cytology in this case showed the presence of acantholytic keratinocytes in addition to neutrophils. This cytology is suggestive of pemphigus foliaceus (see Section 2, Chapter 13). However, acantholysis is recognized to occur in some cases of dermatophytosis, particularly that caused by Trichophyton spp. infection, because of the production of fungal enzymes that result in acantholysis. The same phenomenon is seen in some cases of pyoderma and so a diagnosis of pemphigus foliaceus should never be made on the basis of cytology alone. Another clue in this case that it was a dermatophyte infection, rather than pemphigus foliaceus, was that the nasal planum was unaffected. Dermatophytosis usually results in a folliculitis and there are no hair follicles on the nasal planum.
Dermatophytosis is a contagious, zoonotic disease. The fungal spores are very resistant and will last up to 18 months in the environment. It is therefore important to take every precaution to avoid accidental inoculation with spores from infected animals, and to avoid both environmental contamination and spreading the infection to susceptible animals.
If they have to be hospitalized, infected animals should be kept in suitable isolation facilities. Nurses handling these animals should wear disposable overalls, gloves and hats that are removed and carefully disposed of before handling animals in other areas of the practice. All bedding, grooming utensils, clippers, cages and so on should be vacuumed, scrubbed and washed with hot water, detergent and a suitable disinfectant after use. Current recommendations for environmental decontamination are to use either a concentrated chlorine bleach solution (1 : 10 to 1 : 100) or Clinafarm® (enilconazole) environmental spray (licensed for cattery use in most of Europe), or the detergent–peroxide-based product Virkon-S®.
Clipping of long-haired animals is recommended as an aid in the treatment of dermatophytosis, because it speeds resolution of infection and reduces environmental contamination. Clipping should be done carefully and great care should be taken to avoid excoriating the skin with the clipper blades, because inoculation and infection of excoriated areas is highly likely.
FOLLOW-UP
Antibacterial therapy was discontinued after 4 weeks of treatment. After 2 months of systemic antifungal therapy, there was a significant clinical improvement, with resolution of all the scaling and crusting, but the alopecia and hyperpigmentation remained on the previously affected areas (Figs 9.6 and 9.7).
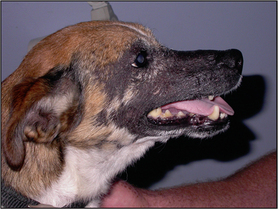
Figure 9.6 After 2 months of therapy with itraconazole. Active inflammation has resolved but hyperpigmentation and alopecia persist over the face.

Figure 9.7 After 2 months of therapy with itraconazole. Active inflammation has resolved but hyperpigmentation and alopecia persist over the hindquarters.
No dermatophytes were grown on a repeat fungal culture and treatment was continued for a further 4 weeks. Another repeat fungal culture was again negative and so systemic treatment was withdrawn. It has been shown that clinical cure occurs prior to complete elimination of fungal elements from the skin, and two negative fungal cultures at an interval of 1 month should be obtained prior to withdrawal of therapy.
Follow-up a year later revealed that there had been some further hair regrowth over the previously affected areas, but partial alopecia remained. There had been no further episodes of skin disease.