PAEDIATRICS
MEASUREMENT OF LUNG FUNCTION
History and examination
Taking a history is the starting place when assessing a child’s lung function. Questions need to reflect the child’s age and must take into account symptoms at rest, and when the child is running around playing or taking part in school games or sport. For example can a baby drink a full bottle of milk or does it become breathless and start spluttering? Does a boy keep up during a game of football or is he always put in goal? Can a schoolgirl carry her books up several flights of stairs to lessons or does she have to rest at each floor? Questions are easier to answer if they relate to normal activities and information is then obtained about how the child functions in everyday life. When discussing noisy breathing, it is well to remember that many parents do not understand what is meant by terms such as wheezing (Cane et al 2000) and an ability to demonstrate these sounds may be helpful.
Physical examination is critical, although it will only give information about the child at rest. Nevertheless, inspection will soon reveal whether the patient is in respiratory distress with, for example, tachypnoea and/or dyspnoea. The normal respiratory rate is age dependent, and decreases with age (Table 3.5). It is important to determine whether the child can speak sentences without becoming breathless and whether accessory muscles are being used leading to intercostal or subcostal recession. Cyanosis, if present, ought to be fairly obvious. When reviewing a child with cystic fibrosis (CF) or bronchiectasis, it is important to inspect the sputum (colour, consistency). Palpation is good for assessing chest expansion and placing the hands on the chest will indicate, by a feeling of vibration, whether the chest is full of sputum – a method used by many parents of children with cystic fibrosis. Percussion is most useful for determining the presence of a pleural effusion (parapneumonic or empyema) indicated by dullness. Before auscultating with a stethoscope, it is worth simply listening to the child’s breathing for wheeze (an expiratory sound), stridor (an inspiratory sound) or upper respiratory tract secretions heard in the throat (harsh expiratory and inspiratory sounds that are transmitted throughout the chest). The presence of a cough and its nature (dry, moist, productive, spasmodic) should be listened for. A forceful huff may also reveal abnormal sounds not obvious with quiet breathing. Finally, listening with the stethoscope may indicate abnormal sounds that can then be located to a particular area or lobe. It is always worth asking the patient to cough before listening again, as often some of the added sounds will have disappeared.
Table 3.5 Respiratory rate by age
| Age (years) | Respiratory rate (per minute) | mean ± 2 SD |
|---|---|---|
| 1 | 30 | 18-42 |
| 2 | 29 | 19-39 |
| 4 | 26 | 16-36 |
| 8 | 23 | 13-33 |
| 12 | 21 | 13-29 |
| 16 | 20 | 12-28 |
(Adapted from Hooker et al 1992)
Dynamic lung function
In clinical practice, lung function tests may help with the management of a child with respiratory disease, for example in assessing severity, time trends, response to treatment and sometimes prognosis (Silverman & Stocks 1999). They are also useful in helping with diagnoses: for example, distinguishing between obstructive and restrictive lung disease. Further, the demonstration of obstructive lung disease that responds to a bronchodilator or worsens with exercise is an important diagnostic aid for asthma. Key abbreviations and definitions can be found in Table 3.6.
Table 3.6 Variables, abbreviations and definitions
| Variable | Abbreviation | Definition |
|---|---|---|
| Peak expiratory flow rate | PEFR | The maximal expiratory flow rate achieved during a forced expiratory manoeuvre |
| Forced expired volume in one second | FEV1 | The volume expired in the first second of maximal expiration after a maximal inspiration |
| Forced vital capacity | FVC | The maximum volume of air which can be exhaled after a maximal inspiration |
| Maximum mid-expiratory flow | MEF25-75 | The average expired flow over the middle half of the FVC manoeuvre |
| Maximum oxygen consumption |
 O2max O2max |
During maximal exercise, a point is reached where further increases in workload do not cause further increases in the maximum volume of oxygen that the body can consume |
| Peak oxygen consumption |
 O2peak O2peak |
This is highest volume of oxygen that an individual consumes during exercise when they have not reached l/O2max (for whatever reason) |
| Maximum minute ventilation |
 Emax Emax |
The maximum ventilatory capacity that can be achieved during maximal exercise |
| Anaerobic threshold | AT | This is the point at which anaerobic metabolism (marked by lactate production) supplements aerobic metabolism |
| Diffusing capacity of the lung for carbon monoxide | DLCO | This is a measure of the surface area of the lung available for gas exchange |
Peak expiratory flow rate (PEFR)
Peak expiratory flow is defined as the maximal flow achieved during an expiration delivered with a maximal force starting from maximal lung inflation. In normal subjects, PEFR is determined by:
The normal values relate to the height and gender of the child (Rosenthal et al 1993a). A child over 5–6 years ought to be able to use a peak flow meter after appropriate training and practice, but the test is very effort-dependent and easy to fake. For this reason, PEFR must be ascertained separately using a peak flow meter rather than using the value calculated by a spirometer. It is most often used in the management of asthmatic children, and inexpensive cheap peak flow meters are available on prescription for home use. It may be valuable when assessing a new therapy, but regular use with a peak flow diary is of limited value due to the poor compliance with such a regimen (Redline et al 1996). Many children simply make up the results and fill in the diary the day before their clinic appointment. However, knowledge of a child’s personal best and usual PEFR can help with asthma management, as a gradual drop in PEFR tends to precede an exacerbation and may sometimes occur before symptoms are recognized. Self-management plans can be in place whereby PEFR results can guide when to take bronchodilators, start oral corticosteroids or seek medical help. Poor control is also indicated by an increase in day-to-day variability of PEFR measurements.
Spirometry
Spirometry, which can be performed in a clinic or at the bedside, is the most valuable and reproducible lung function test used in children. A normal spirogram is shown in Fig 3.1. Most 6 year olds can perform the technique reliably, with some 5 year olds also man-aging it. Normal values take into account the child’s gender and height (Polgar & Weng 1979, Rosenthal et al 1993a) . Results are usually expressed as a ‘percent predicted’, but it is better to use standard deviation (SD) scores (z-scores) – even though these are more difficult for parents to understand. First, the SD scores are able to reflect accurately how far an individual’s spirometry result deviates from the expected norm and, secondly, SD scores will provide valuable tracking data over time. Generally, FEV1 (forced expiratory volume in one second) and FVC (forced vital capacity) >80% of the predicted mean are considered normal, while a cut-off of 60% is used for MEF25–75 (maximal expiratory flow at 25% to 75% vital capacity), as these correspond roughly to values that lie within 2 SD of the mean (Pattishall 1990). FEV1 values of 60–79% are considered to represent mild, 40–59% moderate and <40% severe dysfunction, particularly when related to CF lung disease. Puberty has a dramatic effect on lung function in that both before and after there is a linear increase of lung function with height, while during puberty the relationship is more complex (Rosenthal et al 1993a).
Spirometry is effort-dependent and it is important to watch the child’s technique; the flow–volume and volume–time curves will also indicate whether a proper effort was made. The child inhales maximally then exhales as hard as possible and for as long as possible. Expiratory flow–volume loops are obtained, along with data such as FVC and FEV1. Flow rates at smaller lung volumes e.g. maximal expiratory flow at 25% vital capacity (MEF25) or mean flows across the mid-portion can also be obtained (MEF25–75). Although these give more sensitive information about flow in the small airways, the measurements are less reliable due to their greater variability. It is best to view trends over time and FVC and FEV1 are the two measurements most often followed. The shape of the flow–volume loop can indicate the presence of airway obstruction (a scooped-out concave appearance) and is particularly useful in children when assessing for the possible diagnosis of asthma. The ratio of FEV1 to FVC, used in adult practice to differentiate restrictive from obstructive disease, is less important in children where this differentiation is usually more obvious. However, children have relatively higher flows for their size than adults, so the FEV1 / FVC ratio tends to be higher at around 90% (compared with 75–85% in adults). Inspiratory flow–volume curves can also be obtained and may be useful when consider- ing extrathoracic obstruction, e.g. from a vascular ring. Infection control is important when using spirometers, particularly in a CF clinic.
Bronchodilator responsiveness
Evidence of airway reversibility is useful both for the diagnosis of asthma and its further management. Bronchodilators should ideally be withheld for 4 hours before assessing reversibility and long-acting b2-agonists are withheld that morning. Baseline FEV1 should be measured, followed by the administration of a short-acting bronchodilator. In an effort to demonstrate reversibility, a large dose should be used, such as 1000 mg (10 puffs) of salbutamol via a metered dose inhaler with a spacer device or 5 mg via a nebulizer. FEV1 should then be measured 15 minutes later. Bronchodilator reversibility is commonly expressed as a calculated increase in FEV1 >15% of the baseline value, but this is not the best statistical method. In adults, change in FEV1 should be expressed in absolute terms with an increase of >190 ml indicating reversibility (Editorial 1992). However, in children it has been suggested that an absolute increase >9% of the predicted value (e.g. from 60% predicted to >69%) is the most appropriate way of defining reversibility (Waalkens et al 1993). In some children with genuine asthma, it may not be possible to document reversibility because they have near- normal lung function at the time of testing. In children with CF, the variability of spirometric testing is greater than in normal children, so a larger response is needed to be confident that bronchodilator responsiveness is present (Nickerson et al 1980).
Bronchial challenge
Tests of bronchial hyperreactivity (BHR) involve challenging the airway with cold air or pharmacological agents such as methacholine or histamine. Although commonly performed in adults, bronchial challenge in children tends to be reserved for research protocols and clinical studies. The principal outcome is PC20, which is the provocative concentration of bronchoconstricting agent that produces a 20% fall in FEV1 from baseline sustained for 3 minutes. Methacholine or histamine produce similar results and may be more sensitive than an exercise test at diagnosing non-specific BHR. These challenges do not produce a late-phase response. Using American Thoracic Society (ATS) guidelines (Crapo et al 2000), spirometry is performed at baseline (and the test should not proceed if FEV1 is <60% predicted), then the subject inhales the agent (diluted in 3 ml 0.9% saline) via a nebulizer. The starting dose of methacholine is 0.03 mg/ml, and it is doubled each time to a maximum of 16 mg/ml. After 2 minutes of nebulization with a mouthpiece, spirometry is performed 30 seconds and then 90 seconds after the finish. If the FEV1 falls less than 20%, the subject proceeds to the next challenge dose. If the FEV1 falls more than 20% from baseline, no further methacholine should be given. The test should be terminated and signs and symptoms should be noted. A bronchodilator should be given and spirometry repeated 10 minutes later. The subject is monitored until spirometry has returned to baseline. The dose that produced the fall in FEV1 is calculated and recorded as the PC20. This value can be used to categorize a normal, mild, moderate or severe response to the challenge (Crapo et al 2000).
Static lung volume measurements
Plethysmography or gas dilution are employed to measure static absolute lung volumes and have been discussed in the adult section. The principal difference between the two methods is that gas dilution techniques measure gas communicating with the airways while plethysmography measures all intrathoracic gas. Using these techniques, data on total lung capacity (TLC), residual volume (RV) and functional residual capacity (FRC) can be obtained in children usually over the age of 5 years although some of the younger ones are not keen on getting inside the plethysmographic box. Normative data have been published for UK children aged 4–19 years and male puberty leads to a profound change in lung function, mostly related to size of the thoracic cage, an effect not observed in girls (Rosenthal et al 1993b). The use of these techniques, however, tends to be limited to tertiary care; for example, during annual review in children with CF or as part of investigations for children with complex respiratory disorders.
Resistance and compliance
Measurements of airway resistance and conductance in children are usually measured by plethysmography, but the measurements can be quite variable. Their greatest use in children is to help in the assessment of difficult asthma as they are non-volitional, unlike spirometry, which is easy to fake. Measurements of compliance in spontaneously breathing subjects either requires the subject to relax their respiratory muscles against an occluded airway at various points during tidal breathing, or depends on the insertion of an oesophageal balloon to measure changes in intrathoracic pressure. Changes in resistance and compliance may occur in asthma.
Diffusing capacity of the lungs
Carbon monoxide diffusing capacity or transfer factor of the lung (DLCO) is a measurement of carbon monox- ide transfer from inspired gas to pulmonary capillary blood. A correction is used to take into account the patient’s size (KCO). The approaches to measuring DLCO include rebreathing, steady state and a variety of single breath techniques. In a single breath method, children need to be able to hold their breath for 10 seconds (this is usually possible over the age of 5 years) and need a minimum vital capacity of 800 ml, although rebreathing techniques can be used in smaller children. DLCO has its greatest place in assessment of lung abnormalities that impair alveolar capillary gas transport as seen in interstitial lung disease, sarcoidosis and sickle cell anaemia. Normal data have been published for UK children aged 4–19 years (Rosenthal et al 1993b). DLCO using the rebreathing technique can also be used during exercise to measure DLCO in children as young as 7 years of age (Rosenthal et al 1995, 1997, Rosenthal & Bush 1998). It is utilized as a surrogate marker of alveolar–capillary growth. Essentially DLCO may be normal at rest but may not increase appropriately during exercise, suggesting a failure of recruitment and distension of the alveolar–capillary bed as observed in chronic lung disease of prematurity (Mitchell & Teague 1998).
Preschool children
Measurement of lung function in this age group is problematic due to lack of coordination and cooperation. Over the last 10 years, studies have shown that it is possible to measure lung function in many preschool children (3–6 years) using incentive spirometry (Bridge et al 1999) or the interrupter technique (Bridge et al 1996). Other methods for assessing peripheral airway function include the multiple-breath inert gas washout (Gustafsson et al 2003).
Incentive spirometry
This method involves teaching the child the forced expired manoeuvre in a manner that is appropriate for the child’s development, with plenty of encouragement and positive reinforcement. One such method is the use of breath-activated computer animation programs showing lit candles. The procedure is explained to the child and the child is encouraged to ‘blow’ the candles out and in doing so, performs a forceful expiration. During a test, it is important to watch the child. Blows should be rejected if the tester feels the child has not tried maximally.
Resistance measured by the interruptor technique (Rint)
Rint is a method, recently applied to young children, whereby airways resistance and bronchodilator responsiveness can be measured in children as young as 2–3 years (Bridge et al 1996, 1999). The child, wearing a nose-clip, breathes quietly (tidal breathing) into the mask. Then, in response to a trigger during expiration, at the peak of a tidal flow, a shutter closes off airflow automatically for 100 milliseconds (ms). This gives a measure for airways resistance, which can then be repeated after administration of a bronchodilator or even a bronchial challenge. Normal data are becoming available and this technique may find a place in clinical practice, particularly as this is an ambulatory test that can be carried out anywhere. Rint can differentiate individual children (aged 2–5 years) with a history of wheezing, from those with recurrent cough and those with no history of respiratory symptoms (McKenzie et al 2000). Overall, the most useful application of Rint appears to be in the assessment of short-term bronchodilator responsiveness where it has been shown to be as sensitive as spirometry in asthmatic children aged 5–15 years (Bridge et al 1996, 1999).
Multiple-breath inert gas washout (MBW)
The MBW method is used to measure the efficiency of ventilation distribution in the lungs and to measure functional residual capacity (FRC). Hardware for this method includes a gas analyser (typically a respiratory mass spectrometer), flow meter, a suitable inert gas marker such as sulphur hexafluoride (SF6) and a computer for analyzing the data. Typically, a preschool child will wear a facemask with a tight seal, connected to a pneumotachometer. The child will then breathe a 4% SF6 gas mixture until this gas has equilibrated in the lung. This is called the wash-in phase. At the end of this phase, the inert gas supply is disconnected and the subject breathes room air. The cumulative expired volume for the SF6 concentration in the lung to reach less than 1/40th of the starting concentration is recorded. This is called the washout phase. The value obtained from these measurements is called the lung clearance index (LCI). This value is the cumulative expired volume to clear an inert gas from the lungs, divided by the FRC. An elevated LCI (>7.8) indicates uneven ventilation distribution, which can be the result of generalized peripheral airway obstruction. In preschool children with CF, the MBW is a more sensitive method than both airway resistance measurements and spirometry in detecting lung function abnormalities (Aurora et al 2005). Little is yet known about the usefulness of the MBW in preschool children with asthma.
Infant lung function
Infant lung function measurements are usually carried out under sedation in specialist centres, and in general are much less standardized than those in older, more cooperative children. This has made obtaining reference data difficult, although recently a multicentre group have collated data on over 400 healthy infants using similar methods in the first 2 years of life and reference data have been derived (Hoo et al 2002).
The techniques employed include the rapid thoracoabdominal compression (RTC) technique, which is used to generate partial forced expiratory manoeuvres and determine maximal flow at functional residual capacity ( maxFRC), a measure of peripheral airway function. The sleeping child breathes quietly until a jacket, placed around the chest, is suddenly inflated (squeeze technique). This produces a sharp expiration, resulting in a recorded flow–volume curve and a measurement of maximal flow at FRC.
maxFRC), a measure of peripheral airway function. The sleeping child breathes quietly until a jacket, placed around the chest, is suddenly inflated (squeeze technique). This produces a sharp expiration, resulting in a recorded flow–volume curve and a measurement of maximal flow at FRC.
In a variation, called raised volume RTC, the chest is passively inflated prior to the squeeze. This gives FEV0.4, which is similar to the information provided by the FEV1 in an older child. Normal data are available, although ideally each infant lung function laboratory will have generated its own data. Standards for infant respiratory function testing have been produced jointly by the European Respiratory Society and American Thoracic Society and published as a series of six articles (Bates et al 2000, Frey et al 2000a, 2000b, Gappa et al 2001, Sly et al 2000, Stocks et al 2001). Infant lung function testing may also include MBW techniques to measure lung clearance index, described earlier.
OXYGEN AND CARBON DIOXIDE MONITORING
Pulse oximetry
Pulse oximeters measure arterial oxygen saturation (SaO2) non-invasively, which is physiologically related to oxygen partial pressure according to the oxygen dissociation curve (see Fig. 3.3). It is immensely useful, but its shortcomings must be recognized, particularly the lack of accuracy at SaO2 <70% (Gaskin & Thomas 1995, Schnapp & Cohen 1990). Further, severe hypotension, low cardiac output, vasoconstriction and hypothermia reduce the pulsatile volume of blood in any given tissue. This reduces the signal strength and quality and causes inaccurate readings. With children, it is important that the correct-sized probe is used otherwise poor contact with the skin may result in an inaccurate reading. Finger clubbing does not seem to cause problems with finger probes.
Particular caution must be used when a pulse oximeter is the only measure of oxygenation available in a sick premature baby where too much oxygen is positively harmful. Due to the shape of the oxyhaemoglobin dissociation curve, pulse oximetry is unreliable at diagnosing hyperoxia due to the flat part of the curve. This means for example, that although a SaO2 >97% might at first sight be fine, the PaO2 may be so high as to cause toxicity (Table 3.7). Furthermore, the shape and variability of the oxyhaemoglobin dissociation curve in critically ill children means that a given SaO2 is compatible with a range of arterial oxygen tensions (Clark et al 1992). In these situations, direct measurement of the PaO2 is more appropriate. An alternative non-invasive method is transcutaneous PaO2 monitoring, although this tends to be restricted to use in neonatal units (Clark et al 1992). Particular care must be taken in the newborn not to overheat the skin, and the electrode needs changing every 3–4 hours.
Table 3.7 Arterial PO2 with corresponding SaO2 from oxyhaemoglobin dissociation curve
| Arterial PO2(kPa) | O2 saturation (%) |
|---|---|
| 13 | 97 |
| 9 | 93 |
| 8 | 89 |
| 6 | 80 |
| 5 | 75 |
| 4 | 57 |
Overnight continuous SaO2 monitoring is also an important technique to assess nocturnal hypoxia. However, it is important to state that currently there is no consistent agreed definition of what constitutes nocturnal hypoxia and this appears to be institution dependent. Monitoring is useful when assessing whether a child with CF requires nocturnal oxygen during a chest exacerbation, or when evaluating a child with difficult asthma. It is important that a continuous paper recording is produced that can be analysed the next morning, although these need to be looked at manually rather than simply relying on the computer-generated data summaries. These summaries inevitably include periods when movement artefact produces a falsely low reading which gets included in the summary data. This form of monitoring has been used in certain circumstances as a screening procedure for deciding on the need for full polysomnography in suspected cases of obstructive sleep apnoea (OSA), although a negative oximetry result will not necessarily rule out the diagnosis (Brouillette et al 2000). Although it is true that a normal overnight SaO2 is encouraging, significant hypercarbia can sometimes accompany normal oxygen levels so caution must be exercised. In addition, arousal may be so rapid in some children with OSA that there is not enough time for hypoxaemia to develop, and the diagnosis can be missed with oximetry alone. Domiciliary pulse oximetry in infants with chronic lung disease receiving home oxygen is supported by the American Thoracic Society (Allen et al 2003). However, in the UK there is no evidence that the provision of oximeters improves the outcome of babies on home oxygen, and in practice may lead to excessive adjustments of the flow rate (Balfour-Lynn et al 2005).
Non-invasive CO2 monitoring
As mentioned above, measurement of SaO2 alone gives only half the picture. This is particularly true in children with neuromuscular disease and occasionally so in cases of OSA. Ideally, continuous CO2 levels should be measured to detect alveolar hypoventilation. This is usually done using a transcutaneous monitor with an electrode that can be left on the skin for up to 8 hours. These measurements correlate reasonably well with the arterial PCO2, although the response time is too slow to pick up changes secondary to brief apnoeas or hypopnoeas (Clark et al 1992). If a transcutaneous monitor is not available, then a compromise is to measure a capillary PCO2 immediately the child awakes. Another method for continuous CO2 monitoring is measurement of airway end-tidal CO2, but usually this can only be accurately carried out on an intubated patient as ambient air dilutes the measurements if used with mask ventilation (Clark et al 1992).
Capillary blood gases
Measurement of ‘capillary gases’, i.e. pH, PO2, PCO2, bicarbonate and base excess, is very useful, especially in infancy, an age at which arterial sampling is difficult. Up to 12 months, blood is obtained by a heel prick, and in older children a finger prick is used. Although the capillary PO2 is unrepresentative of arterial levels (normally reading only about 4–5 kPa), the rest of the measurements give a reasonable guide to the child’s respiratory status. It is important that the child’s heel is warmed to ensure sufficient blood flow, and after a heel prick with a sterile lancet, 150 microlitres of free-flowing blood is collected into a heparinized capillary tube and the blood should be put into the blood gas analyser promptly. It is important that air bubbles are not sucked up into the capillary tube, otherwise the sample is spoiled.
Arterial blood gases
For a complete picture of blood gas analysis, an arterial blood sample is required. In practice, this tends to be routinely available only in neonatal and paediatric intensive care units, where an indwelling arterial or umbilical catheter has been placed. A single arterial stab is painful and difficult in a child who understandably will not cooperate. This means that the resultant analysis may not be valid, as a screaming child will have a reduced PaCO2 and possibly an altered PaO2.
EXERCISE TESTING
Exercise testing has become an important tool in the evaluation and treatment of adult and paediatric dis-orders. Exercise stresses many aspects of normal phy-siology, including ventilation and gas exchange, cardiovascular, neuromuscular and thermoregulatory functions. Cardiopulmonary limitation may not be clinically evident at rest, but exercise testing may unmask dysfunction of gas exchange and consequent functional abnormalities.
Exercise capacity
There are three major influences of exercise capacity in a normal individual:
Anatomical.
This really refers to body size. Body size influences muscle mass and the mechanical conditions under which the muscle operates. Body size is influenced by genetic and environmental factors such as diet and levels of habitual activity.
Oxygen transport.
Oxygen is transported from the atmosphere and incorporated into mitochondria of the muscle. The stages involved include ventilation of the lung, diffusion from lungs to the blood, circulation of oxyhaemoglobin, diffusion of oxygen across the capillary muscle membrane, diffusion within the muscle and finally transport to the oxidative pathways of the mitochondria. The principal limiting factor in normal circumstances is the rate of diffusion of oxygen across the capillary muscle membrane. This is greatly influenced by the mean capillary oxygen tension, which is determined by the ability of the circulation to deliver blood to the muscle capillaries.
Physiological response to exercise in children
Understanding the physiological response to exercise is necessary in order to understand the concepts of exercise testing and which exercise test is most appropriate for the situation.
During exercise cardiac output may increase five times, as a result of increases in both heart rate (HR) and stroke volume (SV), and minute ventilation ( E) may increase 25-fold in healthy individuals. As a result of these changes, DLCO increases by approximately 50% during exercise (Manier et al 1993) due to recruitment and distension of the pulmonary capillaries (particularly in the upper parts of the lung), which improve any ventilation–perfusion inequality. The red cell pulmonary capillary transit time (the time taken for a red blood cell to traverse a pulmonary capillary) falls from one second (s) at rest to 0.3 s during exercise. Additionally, during exercise, oxygen consumption (
E) may increase 25-fold in healthy individuals. As a result of these changes, DLCO increases by approximately 50% during exercise (Manier et al 1993) due to recruitment and distension of the pulmonary capillaries (particularly in the upper parts of the lung), which improve any ventilation–perfusion inequality. The red cell pulmonary capillary transit time (the time taken for a red blood cell to traverse a pulmonary capillary) falls from one second (s) at rest to 0.3 s during exercise. Additionally, during exercise, oxygen consumption ( O2) will also increase linearly with increases in workload but will eventually reach a plateau whereby any further increase in workload will not increase
O2) will also increase linearly with increases in workload but will eventually reach a plateau whereby any further increase in workload will not increase  O2. This is known as the maximum oxygen consumption (
O2. This is known as the maximum oxygen consumption ( O2max) (Editorial 2003).
O2max) (Editorial 2003).  O2max is the best index for aerobic capacity and is the gold standard marker for cardio-pulmonary fitness (Editorial 2003). It is related to oxygen availability and provides information regarding aerobic metabolism in response to exercise stress. However, when
O2max is the best index for aerobic capacity and is the gold standard marker for cardio-pulmonary fitness (Editorial 2003). It is related to oxygen availability and provides information regarding aerobic metabolism in response to exercise stress. However, when  O2max is not reached, an individual’s peak oxygen consumption (
O2max is not reached, an individual’s peak oxygen consumption ( O2peak) can also used as a marker of aerobic capacity.
O2peak) can also used as a marker of aerobic capacity.  O2max increases with age until puberty, after which the rate of increase accelerates in adolescent boys and levels off in girls.
O2max increases with age until puberty, after which the rate of increase accelerates in adolescent boys and levels off in girls.  O2max will be affected by age, gender, physical fitness, genetics, population characteristics and mode of testing.
O2max will be affected by age, gender, physical fitness, genetics, population characteristics and mode of testing.
 E increases linearly initially through an increase in the tidal volume (VT). During exercise, lactic acid is produced and this increases the ventilatory drive, which causes an increase in respiratory frequency.
E increases linearly initially through an increase in the tidal volume (VT). During exercise, lactic acid is produced and this increases the ventilatory drive, which causes an increase in respiratory frequency.  E will eventually reach a plateau with increased workload and this is termed maximum minute ventilation (
E will eventually reach a plateau with increased workload and this is termed maximum minute ventilation ( Emax). Anaerobic threshold (AT) is considered an estimator of the onset of metabolic acidosis secondary to this rise in lactic acid during exercise (Editorial 2003). AT appears to be a good indicator of physical performance capacity in that it correlates well with endurance capacity (Sullivan et al 1995, Wasserman et al 1973) and is relatively independent of maximal effort (Editorial 2003). Physical training increases the numbers of capillaries and mitochondria in muscle and will increase both
Emax). Anaerobic threshold (AT) is considered an estimator of the onset of metabolic acidosis secondary to this rise in lactic acid during exercise (Editorial 2003). AT appears to be a good indicator of physical performance capacity in that it correlates well with endurance capacity (Sullivan et al 1995, Wasserman et al 1973) and is relatively independent of maximal effort (Editorial 2003). Physical training increases the numbers of capillaries and mitochondria in muscle and will increase both  O2 and AT.
O2 and AT.
Maximal cardiac output increases with increasing body size mainly due to the increase in ventricular size and stroke volume. The maximal HR is fairly stable through childhood, then begins to decline in late teens towards adult values, whereas resting HR progressively decreases throughout childhood. A commonly used formula for predicting the maximal expected HR in children is (Nixon & Orenstein 1988):
220 – age (in years) ± 10 beats per minute
Gender differences are present throughout childhood but are more marked after puberty: for example boys have a greater  and maximal cardiac output, whereas girls have higher HR values.
and maximal cardiac output, whereas girls have higher HR values.
Indications for exercise testing
Exercise testing is valuable because clinical examination and static cardiopulmonary tests are poor at predicting exercise capacity. Guidelines have been produced by the American College of Cardiology and American Heart Association to make recommendations on the appropriate use of exercise testing in the diagnosis or treatment of patients with known or suspected cardiovascular disease, which includes a section on children (Gibbons et al 2002). The indications for exercise testing are:
Diagnostic tool
Dyspnoea assessment.
Dyspnoea is the uncomfortable sensation that breathing is difficult. Exercise can assess true dyspnoea from perceived dyspnoea (for example, due to poor fitness) by assessing appropriate physiological responses during exercise.
Diagnostic differentiation.
Exercise tests are useful for making a specific diagnosis such as exercise-induced bronchospasm and differentiating it from psychological causes. It is also useful for differentiating between cardiac and muscular pain. Further, an exercise test may identify a deficiency in a specific component of fitness: for example, differentiate a problem of muscle endurance and strength from aerobic capacity (Tomassoni 1996a).
Objective assessment of disability.
Exercise testing may help identify abnormal cardiovascular, respiratory or metabolic responses to exercise: for example, cardiac arrhythmias, hypertension, hypoxaemia or hypoglycaemia (Nixon & Orenstein 1988). The test may amplify pathophysiological changes and trigger changes not seen at rest. Similarly, exercise tests can be used to provide objective evidence that there are no functional deficits or that exercise limitation is not secondary to organic disease.
Determine severity and functional effects of known disease
An inability to perform daily physical activities can severely affect quality of life. Exercise testing can provide objective information on any reduction in physical activity. This information is often only partially obtainable from standard lung function testing performed with the child essentially at rest. Further, repeated exercise testing using reproducible outcome measures can help chart disease progression. This may be useful as part of the annual review of children with CF, or for children affected by neuromuscular disorders such as Duchenne muscular dystrophy. It can also help determine the severity of cardiac dysrhythmias, especially those that are precipitated by exercise.
Determine prognosis
In some circumstances, it may be possible to estimate prognosis from exercise ability; for example cycle ergometry in CF (Nixon et al 1992) or 6-minute walking test in severe left ventricular dysfunction (Bittner et al 1993).
Outcome measure for therapeutic interventions
Exercise testing may help in deciding the appropriateness of medical and surgical interventions. Having established a baseline, repeat testing can then be used as a measure of the effectiveness of the particular intervention. Studies in children with pulmonary hypertension treated with sildenafil were assessed by monitoring exercise performance (Humpl et al 2005, Karatza et al 2004). Exercise testing revealed an increase in exercise capacity, specifically the distance walked over this time period. Another example would be its use as one of the criteria for deciding both suitability and appropriate timing of a heart-lung transplant. Further, the functional success of surgical correction of congenital heart lesions can be monitored non-invasively with laboratory-based exercise tests (Derrick et al 2000, Rosenthal & Bush 1999, Rosenthal et al 1995).
Outcome measure in clinical trials
Exercise tests are a useful outcome measure for clinical trials (Ramsey & Boat 1994). If the test is simple and portable it may have a place in phase III multicentre trials (Pike et al 2001).
Aid to improving fitness in health and disease
Many children and their parents are anxious about allowing the child to exercise if the child has an underlying cardiac or respiratory disorder. Exercise testing, conducted with the reassurance of the supervising staff, in the protective environment of the laboratory, can be used to provide evidence that it is safe for the child to exercise. Children can then be encouraged to adopt an active lifestyle to increase their exercise tolerance and improve their level of fitness.
Types of tests available and their application in children
There are many types of exercise tests available, and choosing the appropriate test depends on the question being asked and the aspect of exercise tolerance that needs elucidating. The questions may focus on cardioventilatory variables or muscle strength. Tests may be categorized as either maximal or submaximal and their relative merits have been recently reviewed (Noonan & Dean 2000). These include:
Walking tests
Self-paced walking tests were initially developed for adults with chronic obstructive pulmonary disease and the techniques have been reviewed (Noonan & Dean 2000). The advantages of such tests are that they are cheap, easy to learn and can be used in an ambulatory setting by children as young as 5 years. However, they require adequate space and are effort-dependent so that the patient’s attitude and motivation are a major factor in determining the distance walked. One such test, the 6-minute walk test is designed such that the distance walked in 6 minutes is measured (usually walking up and down a known length of corridor). No verbal or other encouragement is given. It has been shown to be a reliable assessment of exercise performance in healthy children (Li et al 2005) and in severely ill children, the distance walked in 6 minutes correlated with  O2peak, physical work capacity and the minimum SaO2 (Nixon et al 1996). The effects of exercise training in children with congenital heart disease were monitored using a 6-minute walk test, the results of which demonstrated an increase in walking distance (Moalla et al 2005). The 6-minute walk has been further validated in children with mild and moderate CF lung disease, being compared with cycle ergometry (Gulmans et al 1996) and the 3-minute step test (Balfour-Lynn et al 1998). The 6-minute walk test is used as part of the assessment for lung transplantation in children (Aurora et al 2001).
O2peak, physical work capacity and the minimum SaO2 (Nixon et al 1996). The effects of exercise training in children with congenital heart disease were monitored using a 6-minute walk test, the results of which demonstrated an increase in walking distance (Moalla et al 2005). The 6-minute walk has been further validated in children with mild and moderate CF lung disease, being compared with cycle ergometry (Gulmans et al 1996) and the 3-minute step test (Balfour-Lynn et al 1998). The 6-minute walk test is used as part of the assessment for lung transplantation in children (Aurora et al 2001).
Stepping tests
The 3-minute step test was developed for use in children over 6 years of age as a means of assessing submaximal exercise tolerance by a simple method (Balfour-Lynn et al 1998). Adapted from an adult cardiac test used since the 1920s, it consists of stepping up and down a commercially available aerobic step for 3 minutes (Fig. 3.25).
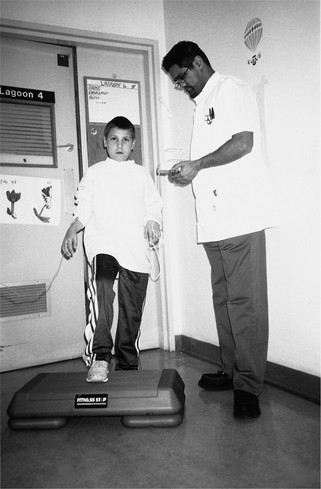
Figure 3.25 Child performing a 3-minute step test with pulse oximeter which measures oxygen saturations and heart rate.
The step test has been compared with the 6-minute walk in 54 children with cystic fibrosis; the step produced significantly greater changes in heart rate and breathlessness than the walk, with a comparable fall in SaO2 (Balfour-Lynn et al 1998). A fall in SaO2 >4% was felt to be clinically significant and could not always have been predicted from the baseline FEV1. The test was found to be simple to learn, quick to perform and required little space or expense. Importantly, motivation is excluded as a possible variable, and although patients can stop if they wish, in practice this rarely happens. The ambulatory nature of the test means it has even been used in outdoors high-altitude exercise testing of children with cystic fibrosis (Dinwiddie et al 1999). The step test has also been studied in children with severe cystic fibrosis lung disease being assessed for heart-lung transplantation (Aurora et al 2001). In this patient group it produced a greater fall in SaO2 and rise in heart rate than the 6-minute walk.
The step test has also been used to assess the effects of intravenous antibiotics on exercise tolerance in 36 children with cystic fibrosis, and all exercise outcomes were shown to improve (Pike et al 2001). It was found that it complemented spirometry and simple SaO2 monitoring, and importantly it was demonstrated that the step test was sensitive to changes after a therapeutic intervention. A modified incremental version of the original test has also been used in the evaluation of breathlessness in both normal children and children with cystic fibrosis. In this test the stepping rate rises at 2-minute intervals using rates of 20/min, 30/min and then 40/min and, although incremental, this test still remains submaximal (Prasad et al 2000). A criticism of the step test is that the workload varies with each subject. This is because the work performed in the test is a product of the stepping height and rate (both fixed) as well as the subject’s weight and height (particularly leg length). In order to keep the amount of work constant between subjects, the height or rate could be adjusted to account for differences in patient size. However, in practice this is neither feasible nor important in the context of the way the test is used, although the pubertal growth spurt might affect interpretation of longitudinal results from annual testing through puberty. A further factor to consider is that it is not shown to be a sensitive test in children with mild cystic fibrosis lung disease (Narang et al 2003).
The recommended protocol is outlined below:
 It is best for children to practise the test first with sufficient resting time before the test begins.
It is best for children to practise the test first with sufficient resting time before the test begins. Comfortable shoes should be worn (not high heels, preferably trainers) and ideally there should be a cushioned mat on the floor to lessen impact on knee joints.
Comfortable shoes should be worn (not high heels, preferably trainers) and ideally there should be a cushioned mat on the floor to lessen impact on knee joints. A pulse oximeter is used with the probe attached to the patient’s finger and the lead taped to the forearm to ensure minimal trace interference during stepping.
A pulse oximeter is used with the probe attached to the patient’s finger and the lead taped to the forearm to ensure minimal trace interference during stepping. Subjects are shown how to change the lead leg while stepping (i.e. the leg placed on the step first) so that one leg does not get overtired (muscle fatigue).
Subjects are shown how to change the lead leg while stepping (i.e. the leg placed on the step first) so that one leg does not get overtired (muscle fatigue). Standard encouragement at 1 and 2 minutes is given. This should state how far into the test the patient is and that they are doing well.
Standard encouragement at 1 and 2 minutes is given. This should state how far into the test the patient is and that they are doing well. Baseline and lowest SaO2 during the test. This may occur at any time within the 3 minutes. A fall in SaO2 over 4% should be considered abnormal.
Baseline and lowest SaO2 during the test. This may occur at any time within the 3 minutes. A fall in SaO2 over 4% should be considered abnormal. Breathlessness determined at the start and end of the test using a subjective score (10 cm visual analogue) and the objective 15-count breathlessness score (Prasad et al 2000) or ease of breathing score (Orenstein et al 2002).
Breathlessness determined at the start and end of the test using a subjective score (10 cm visual analogue) and the objective 15-count breathlessness score (Prasad et al 2000) or ease of breathing score (Orenstein et al 2002).The patients are told that they can stop the test at any time if they feel unable to continue for any reason. The investigator should stop the test if the SaO2 falls below 75%, the patient is unduly breathless or is struggling to keep pace and rhythm. If the patient stops stepping within 3 minutes, the reason why they felt unable to continue should be recorded as well as the time they stopped.
Shuttle tests
There are various forms of the shuttle test (Noonan & Dean 2000). The shuttle walk test is an incremental test of maximal capacity which is achieved by walking the subject around two cones 10 metres apart and increasing the paced walking speed every minute (see adult section). The pace is provided by an audiotape, which ‘beeps’ at progressively shorter time intervals. The result is expressed in metres. The shuttle test is akin to the standard incremental laboratory test of maximal performance. It has been used to study the effects of training on a group of asthmatic children aged 12–17 years and when compared with standard cycle ergometry, it had sufficient validity to register training effects (Ahmaidi et al 1993). The modified shuttle test has been validated in children and adolescents with cystic fibrosis (Cox et al 2006).
Maximal tests – cycle ergometry and the treadmill
The maximal exercise tests in which subjects are encouraged to reach their maximal level of tolerance remain the cornerstone of most exercise tests in adults and children.
Despite much discussion and debate over protocols, there has yet to emerge a common, standardized approach for assessing exercise capacity in children (Hebestreit 2004).
Nixon & Orenstein (1988) have reviewed the different maximal exercise protocols suitable for children. Briefly, these tests utilize a cycle ergometer or treadmill and the workload is increased progressively (usually at preset time intervals) until the subject reaches their maximal level of tolerance, with online analysis of inspired and expired gases. These tests are considered the gold standard exercise tests, with oxygen consumption,  O2peak or
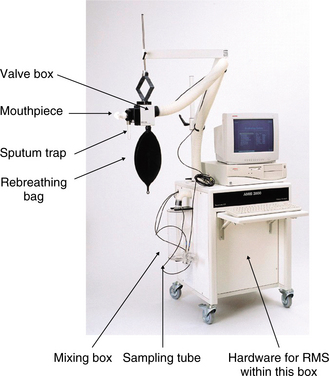
O2peak or  O2max as the principal outcome measures. One such gas analyser is the respiratory mass spectrometer (RMS), which allows measurements of both cardiac and ventilatory variables (Fig. 3.26). Normal reference ranges and ventilatory variables for children undergoing cycle ergometry using an RMS have been published (Rosenthal & Bush 1998, 2000). These data have been used to assess cardioventilatory variables during exercise in children following repair of congenital heart disease (Derrick et al 2000, Rosenthal & Bush 1999, Rosenthal et al 1995, 1997). Similar techniques have shown normal exercise capacity following congenital diaphragmatic hernia repair (Trachsel et al 2006) and abnormal ventilatory response to exercise in children with chronic lung disease following preterm birth (Jacob et al 1997, Trachsel et al 2006).
O2max as the principal outcome measures. One such gas analyser is the respiratory mass spectrometer (RMS), which allows measurements of both cardiac and ventilatory variables (Fig. 3.26). Normal reference ranges and ventilatory variables for children undergoing cycle ergometry using an RMS have been published (Rosenthal & Bush 1998, 2000). These data have been used to assess cardioventilatory variables during exercise in children following repair of congenital heart disease (Derrick et al 2000, Rosenthal & Bush 1999, Rosenthal et al 1995, 1997). Similar techniques have shown normal exercise capacity following congenital diaphragmatic hernia repair (Trachsel et al 2006) and abnormal ventilatory response to exercise in children with chronic lung disease following preterm birth (Jacob et al 1997, Trachsel et al 2006).
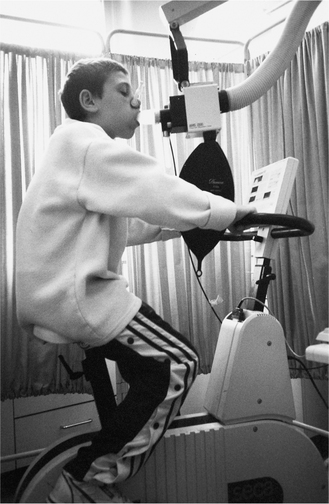
Children over 6 years of age can perform these tests although a degree of coordination and motivation are required (Fig 3.27 shows a subject attached to an RMS). Children are required to pedal or run at a particular workload for a specific period of time while simultaneously wearing a facemask connected to a gas analyser. At specific time intervals, the workload is increased again and this process will continue until the child reaches exhaustion or refuses to carry on. Gas analysis continues during the time of exercise and recovery.
Although the cycle ergometer is familiar as most children ride bicycles, some children find it easier to run than pedal and prefer the treadmill. Some children also find it difficult to maintain a constant pedalling rate. For smaller children, the speed of the treadmill must be adjusted to account for their shorter strides. Also, both cycling and running are more difficult when a tightly fitting facemask is worn. Such tests are time consuming and require significant laboratory space with highly skilled personnel needed to supervise and analyse the exercise test. The equipment is also expensive to buy, use and service.
Peak or maximal exercise tests in children do not necessarily represent their normal daily activities (Cooper 1995). Children tend to engage in very short bursts of high-intensity physical activity interspersed with variable periods of low and moderate intensity. They do not tend to perform sustained heavy exercise, which is the format of most maximal tests. Furthermore, the tests are highly effort-dependent and healthy control children can be strongly encouraged, even cajoled, to make a maximal effort. In contrast, children who have an underlying cardiorespiratory disorder may not be pushed so hard, making comparison with normal values less valid (Cooper 1995). In addition, these tests are physiologically stressful and there may be concerns over taking unnecessary risks in children with moderate to severe cardiorespiratory compromise.
Wingate Anaerobic Test (WAnT)
While some children’s daily activities are predominantly aerobic, most activities rely on both aerobic and anaerobic energy (Boas et al 1996). The intensity and duration of the activity, as well as the child’s level of fitness, determine the relative contribution of these energy sources: for example, high-intensity exercise of short duration is principally anaerobic in nature. As discussed above, this form of exercise is typical of children; hence the relevance of this form of exercise testing in paediatric practice. Anaerobic capacity is determined by the muscles’ ability to produce energy quickly from anaerobic sources, and to test this, the subject performs a 30-second all-out sprint at the highest tolerable workload on a cycle ergometer. This is designed to test anaerobic performance only and the indices of performance are peak power and mean power. A version also exists, using arm cranking, that has been used in children with neuromuscular disorders (Bar-Or 1996). Studies in children and adolescents with cystic fibrosis have shown that anaerobic exercise capacity is reduced and this mainly reflects decreased nutritional status, with pulmonary function playing a smaller part (Boas et al 1996). There is conflicting data as to whether anaerobic capacity is reduced in asthma (Welsh et al 2004).
Exercise testing and muscle strength
The main type of exercise tests performed are those assessing cardioventilatory variables. However, there is also a place for exercise tests to assess muscle strength and these usually involve lifting simple weights or mea- suring isometric muscle force using a dynamometer or myometer. Muscle strength has been shown to improve with age (Ioakimidis et al 2004) and this must be taken into consideration. These tests are particularly relevant in neuromuscular disorders where maximal aerobic power is less important, because it is seldom the limiting factor in the child’s ability to perform daily physical activities (Bar-Or 1996). However, peripheral muscle force is impaired in children with cystic fibrosis, even in the absence of reduced pulmonary or nutritional status (de Meer et al 1999). It is also possible to measure inspiratory muscle strength non-invasively, and in children with cystic fibrosis, it has been found to be impaired, even in those with good nutritional status (Hayot et al 1997). However, in one study, children with CF, admitted to hospital for treatment of chest exacerbations with intravenous antibiotics, were randomized to receive supervised exercise training (Selvadurai et al 2002). The first group received 30 minutes of aerobic exercise 5 times per week; the second group received 30 minutes of weight training (upper and lower limbs) for 30 minutes 5 times per week and the third group were encouraged to undertake their usual physical activity. An exercise test was performed on admission, discharge and 1 month post-discharge. The results showed that the group randomized to muscle training improved their FEV1 to a greater extent than the other groups. There are little data in children with cystic fibrosis that have examined the effects of both aerobic and anaerobic training on the impact on lung disease.
Outcome measures
There are many useful outcome measures during an exercise test and those employed for a specific individual really depend on the question to be answered. In children, it has been suggested that the cardiopulmonary data collected during the submaximal phases, rather than from the final single data point at peak or maximal power, should be utilized as they more likely equate to daily physical activity (Cooper 1995).
O2 consumption ( O2)
O2)
As discussed earlier,  O2 is the principal outcome measure from maximal exercise testing such as cycle ergometry or a treadmill test, and it is the single measure most commonly used to represent overall aerobic fitness in children (Nixon & Orenstein 1988). During progressively increasing exercise,
O2 is the principal outcome measure from maximal exercise testing such as cycle ergometry or a treadmill test, and it is the single measure most commonly used to represent overall aerobic fitness in children (Nixon & Orenstein 1988). During progressively increasing exercise,  O2 increases linearly with increases in work level. However,
O2 increases linearly with increases in work level. However,  O2max is observed in less than a third of children and adolescents (unlike adults) (Cooper 1995), so
O2max is observed in less than a third of children and adolescents (unlike adults) (Cooper 1995), so  O2max cannot be precisely determined in many paediatric studies. However, the value often recorded is
O2max cannot be precisely determined in many paediatric studies. However, the value often recorded is  O2peak, which is the highest oxygen consumption of an individual during a specific exercise test (Washington et al 1994). Part of the reason for this is that the test is effort-dependent so many children feel tired and stop before the true
O2peak, which is the highest oxygen consumption of an individual during a specific exercise test (Washington et al 1994). Part of the reason for this is that the test is effort-dependent so many children feel tired and stop before the true  O2max is reached. Typical
O2max is reached. Typical  O2max values in healthy children are 40–50 ml O2/kg/min, while children with cardiorespiratory disease may have values as low as 10 ml O2/kg/min (Nixon & Orenstein 1988). The intra-individual day-to-day variation in
O2max values in healthy children are 40–50 ml O2/kg/min, while children with cardiorespiratory disease may have values as low as 10 ml O2/kg/min (Nixon & Orenstein 1988). The intra-individual day-to-day variation in  O2max is around 4–6% in fit subjects, although this figure is higher in those with pulmonary disease (Noonan & Dean 2000).
O2max is around 4–6% in fit subjects, although this figure is higher in those with pulmonary disease (Noonan & Dean 2000).
As well as O2, the CO2 is measured in the inspired and expired gas collected during formal maximal testing. This gives the full picture of ventilation and pulmonary gas exchange. In addition to  O2, CO2 excretion (
O2, CO2 excretion ( CO2),
CO2),  E, respiratory gas exchange ratio (
E, respiratory gas exchange ratio ( CO2/
CO2/ O2), and the oxygen ventilatory equivalent (
O2), and the oxygen ventilatory equivalent ( E/
E/ O2) can be calculated. With the addition of flow measurements (by turbine or pneumotachograph) the average respiratory rate and tidal volume can also be measured.
O2) can be calculated. With the addition of flow measurements (by turbine or pneumotachograph) the average respiratory rate and tidal volume can also be measured.
Anaerobic threshold (AT)
Above a certain work rate threshold, the AT is the point when the oxygen demand of the exercising muscle exceeds the oxygen supply, and anaerobic metabolism is required to enable the subject to continue further exercise (Washington et al 1994). At this point, the lactate concentration of the blood increases. During the process of buffering the blood lactate levels there is an increased production of bicarbonate. This leads to a rise in PaCO2, and this in turn results in reflex compensatory hyperventilation. The AT is usually expressed as a percentage of  O2max and may vary from 40 to 60%, depending on the degree of physical conditioning. Measurement of the AT is one of the better ways of determining aerobic fitness, as the fitter the person, the higher the
O2max and may vary from 40 to 60%, depending on the degree of physical conditioning. Measurement of the AT is one of the better ways of determining aerobic fitness, as the fitter the person, the higher the  O2 before the blood lactate starts to rise. An AT less than 40% may indicate cardiac disease but also may indicate moderate deconditioning (due to lack of fitness).
O2 before the blood lactate starts to rise. An AT less than 40% may indicate cardiac disease but also may indicate moderate deconditioning (due to lack of fitness).
AT has advantages over  O2max as a primary outcome as it is more easily reproducible between occasions and between patients. The reason for this is because
O2max as a primary outcome as it is more easily reproducible between occasions and between patients. The reason for this is because  O2max requires the patient to get up to maximal exercise capacity whereas AT is a submaximal measure and is relatively independent of effort. The onset of hyperventilation beyond the anaerobic threshold is known as the ventilatory anaerobic threshold, and it can be seen as the point of inflexion on the minute ventilation /
O2max requires the patient to get up to maximal exercise capacity whereas AT is a submaximal measure and is relatively independent of effort. The onset of hyperventilation beyond the anaerobic threshold is known as the ventilatory anaerobic threshold, and it can be seen as the point of inflexion on the minute ventilation /  O2 curve (Washington et al 1994).
O2 curve (Washington et al 1994).
Minute ventilation ( E) and ventilatory limit
E) and ventilatory limit
In healthy subjects,  E increases with oxygen uptake and is not usually limiting. The increase in
E increases with oxygen uptake and is not usually limiting. The increase in  E is first achieved by a rise in tidal volume (VT), due to both a reduction in the end-expiratory lung volume and an increase an increase in end-inspired volume, until VT reaches a plateau at approximately two-thirds of vital capacity. Further increases in
E is first achieved by a rise in tidal volume (VT), due to both a reduction in the end-expiratory lung volume and an increase an increase in end-inspired volume, until VT reaches a plateau at approximately two-thirds of vital capacity. Further increases in  E are achieved by an increase in respiratory rate. The maximum minute ventilation (
E are achieved by an increase in respiratory rate. The maximum minute ventilation ( Emax) during exercise is the highest ventilation that can be achieved and further increases in workload or effort do not give rise to further increases in
Emax) during exercise is the highest ventilation that can be achieved and further increases in workload or effort do not give rise to further increases in  E. When
E. When  E is compromised by lung disease, the subject’s predicted
E is compromised by lung disease, the subject’s predicted  Emax is reached at a time when the heart rate is considerably less than its maximum; i.e. heart rate reserves greatly exceed ventilatory reserves. The predicted
Emax is reached at a time when the heart rate is considerably less than its maximum; i.e. heart rate reserves greatly exceed ventilatory reserves. The predicted  Emax on exercise can be calculated from measurements of maximum voluntary ventilation (MVV) or indirectly using (Hansen et al 1984):
Emax on exercise can be calculated from measurements of maximum voluntary ventilation (MVV) or indirectly using (Hansen et al 1984):
However, the latter method may underestimate exercise ventilation in subjects with airflow obstruction and a severely reduced FEV1.
Respiratory exchange ratio (RER)
The ratio of  CO2/
CO2/ O2 is called the respiratory exchange ratio. RER is usually measured by gas exchange at the mouth. An RER >1.0 suggests the production of lactic acidosis but it may also represent hyperventilation. An RER >1.2 usually indicates maximum effort.
O2 is called the respiratory exchange ratio. RER is usually measured by gas exchange at the mouth. An RER >1.0 suggests the production of lactic acidosis but it may also represent hyperventilation. An RER >1.2 usually indicates maximum effort.
Oxygen saturation (SaO2)
A useful measure of gas exchange that is readily available and accessible is the SaO2 determined by pulse oximetry, and desaturation <90% during exercise is an abnormal response in children. In practice, even dramatic falls in SaO2 have no untoward results, and most children gain pre-exercise levels within 5 minutes and all by 15 minutes (Henke & Orenstein 1984).
It is particularly important that movement artefact is eliminated during exercise, otherwise measurements are invalid. In our experience, a flexi-probe taped to the index finger with the lead strapped to the forearm overcomes this problem (Balfour-Lynn et al 1998). The use of a supraorbital probe (placed on the supraorbital artery) during maximal exercise has shown to be less prone to artefact (Derrick et al 2000). Ideally, traces are also recorded on to a computer program alongside the numbers, for later review. This allows elimination of false reading due to low-quality signals. The type of test may also affect the frequency of movement artefact; for example, fewer occur during the step test compared with the walking test, as the children’s arms can be kept relatively still while stepping, whereas they tend to swing their arms when walking (Balfour-Lynn et al 1998). In addition, the oximeter and its lead are kept stationary during the step test rather than being wheeled or carried up and down a corridor.
Heart rate
Measurement of heart rate is mandatory for all exercise testing. The dynamics of the heart rate response to exercise are proving to be increasingly useful to identify abnormalities in a variety of disease states. Heart rate monitoring is usually done by a pulse oximeter or cardiorater with electrodes attached to the chest; potential problems of using the former method have been discussed above. During dynamic exercise, the heart rate increases in a linear fashion with the rate of work, and increases up to 3 to 5 times from resting can be seen. Variables that affect heart rate during exercise include the type of exercise, body position during testing, gender, state of health and fitness of the subject, and environmental conditions (temperature, humidity, altitude) (Washington et al 1994). Young children compensate for their relatively small heart size (with lower stroke volume) by an increased heart rate for a given amount of work, so they attain higher maximal heart rates than adults. In pulmonary disease, the patients may not attain the expected maximal heart rate, as their exercise is ventilation-limited. Resting heart rate is also a useful measure in children with lung disease, and was shown to be significantly reduced after treatment of a chest exacerbation in children with cystic fibrosis (Pike et al 2001). Presumably, this was due to a reduction in the high metabolic rate and increased work of breathing associated with a chest infection. In addition, a high resting age-adjusted heart rate was associated with a poorer prognosis in children with cystic fibrosis (Aurora et al 2001).
Work
Maximal work capacity is the highest workload achievable on a progressive test, and is influenced by age, body size and gender. Older and larger children have a greater work capacity, as do boys (Nixon & Orenstein 1988). Obviously children with higher levels of aerobic fitness can do more work. Estimates of the amount of exercise performed are made using the following definitions (Washington et al 1994):
Distance
This is relevant in walking and shuttle tests and it is very dependent on the subject’s motivation. This is particularly true of walking tests where the subjects determine their own pace. In severe lung disease, the distance covered has been shown to correlate with survival in adults (Kadikar et al 1997). Although this is true in children with cystic fibrosis being assessed for transplantation, the minimum SaO2 during the walking test has been shown to be a stronger predictor (Aurora et al 2000).
Electrocardiogram (ECG)
ECG monitoring is particularly useful during exercise tests for children with known or suspected cardiac disease (Washington et al 1994), particularly for patients suspected of having myocardial ischaemia, e.g. aortic stenosis, ST segment changes (which may indicate mismatch of myocardial oxygen demand and supply) and for onset of dysrhythmias.
Cardiac output
Cardiac output increases with exercise to support the increasing metabolic demands of the tissues. The measurement of cardiac output is the best index of cardiac function during exercise. However, it is not routinely measured in clinical exercise laboratories because the methods used (e.g. acetylene rebreathing) are technically demanding. In healthy subjects, cardiac output is a linear function of  O2 and, during exercise, increases in cardiac output are initially accomplished by increases in stroke volume and heart rate, and then at moderate to high intensity exercise almost exclusively by increases in heart rate. The evaluation of the heart rate response yields an estimation of cardiac function during exercise. Cardiac output may rise 3–5-fold during maximal exercise. As well as the state of health, the type of exercise can determine the cardiac output; treadmill testing generally results in a higher maximal cardiac output than does cycle ergometry.
O2 and, during exercise, increases in cardiac output are initially accomplished by increases in stroke volume and heart rate, and then at moderate to high intensity exercise almost exclusively by increases in heart rate. The evaluation of the heart rate response yields an estimation of cardiac function during exercise. Cardiac output may rise 3–5-fold during maximal exercise. As well as the state of health, the type of exercise can determine the cardiac output; treadmill testing generally results in a higher maximal cardiac output than does cycle ergometry.
Blood pressure
Blood pressure changes may be monitored, although the response to exercise can be complex. In children, this is done by indirect methods using a cuff; even though motion artefact and noise can make the measurement difficult, direct arterial measurements are invasive and painful. During exercise, systolic blood pressure typically rises progressively with an increase in  O2 but rarely exceeds 200 mmHg in children, although there is no evidence of danger if it reaches 250 mmHg. Diastolic blood pressure typically remains constant or may decline slightly. If blood pressure does not increase with exercise, or if in fact it declines, this may be an ominous sign of severe cardiac dysfunction, although an exertional drop in blood pressure may also occur in normal subjects (Washington et al 1994).
O2 but rarely exceeds 200 mmHg in children, although there is no evidence of danger if it reaches 250 mmHg. Diastolic blood pressure typically remains constant or may decline slightly. If blood pressure does not increase with exercise, or if in fact it declines, this may be an ominous sign of severe cardiac dysfunction, although an exertional drop in blood pressure may also occur in normal subjects (Washington et al 1994).
Lung function
The use of spirometry, pre-, during and post-exercise can be a useful outcome measure in subjects complaining of wheeze and breathlessness during or after exertion as seen in some subjects with asthma, as discussed earlier.
Reference values
The main outcome measures for exercise tests have been discussed. However, reference values for exercise parameters have not been so well standardized. The reference values are less reliable at the extremes of height, age and body mass index. Further, variations in methodology and in the racial and socioeconomic composition of population groups necessitate validation of normal values for any particular laboratory.
Symptoms
Dyspnoea
Dyspnoea or breathlessness is the uncomfortable sensation that breathing is difficult. It is normal for breathlessness to increase during exercise. The significance of breathlessness is related to the intensity of the exercise that produces the symptoms. Clearly a child who is breathless at rest has a greater problem than one who is breathless only during intense exercise. This makes the measurement of breathlessness an important outcome of exercise testing as long as it can be quantified reliably. The usual means of assessing the subjective elements of breathlessness are the modified Borg scale of perceived breathlessness (Burdon et al 1982) and standard visual analogue scores (Fig 3.28) (Prasad et al 2000). The original Borg scale of perceived exertion (Borg 1982) modified by Burdon et al (1982) for the measurement of breathlessness has 12 points, 10 of which have accompanying verbal descriptors. It is interesting that they do give similar scores, as there is an inbuilt bias to score under 5 with the Borg scale since 5 represents ‘severe’ breathlessness, which is usually as high as patients wish to score themselves. In practice, use of the Borg scale is a problem for many children due to difficulty understanding some of the terminology (such as moderate or maximal). It is likely that many children simply use the number ratings 1 to 10 and ignore the accompanying descriptors, so that in the end the Borg scale is often used in a similar way to the visual analogue score. A theoretical advantage of the latter is that it is a continuous variable, which is supposed to be easier to use than scoring with discrete numbers. One problem, however, with the visual analogue score is that the anchor points (‘I am not at all short of breath’ or ‘The most short of breath I have ever been’) are specific to the individual, making comparisons of scores between different people less reliable. A score of 5 for one child may be very different to what another child considers 5 to represent. They often score themselves in the middle of the line, as halfway represents an average day for them, ‘I am as breathless as I usually feel after exercise,’ which of course may be very different for different individuals. These problems with the Borg and visual analogue scores highlight the difficulties of measuring a subjective feeling or symptom. However it does not invalidate their use, as it is important to know how breathless the child feels or thinks they feel, since this is the symptom that is causing them concern. Another problem is that just as in quantifying pain, what the patient says they are experiencing is not always in keeping with their appearance to others (parents and medical attendants).
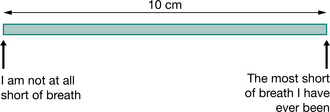
Figure 3.28 Visual analogue score. 10 cm horizontal line with two anchor points at each extreme (zero and 10 cm). The subject puts a mark through the line where they think their breathlessness fits on this scale, which is then measured (in cm) from the zero point.
Breathlessness scores.
In addition to subjective measures, there is an objective score that has been validated in children, called the 15-count breathlessness score (Prasad et al 2000). This score is determined by asking the child to take a deep breath and count out loud to 15 (taking about 8 seconds to do so). The number of breaths required to reach 15 (including the initial breath) is the score, hence 1 is the minimum score. Most children score 1 at rest, and when performing light exercise, many will still score 1, especially if they are healthy. For this reason, the 15-count score is best used in those prone to breathlessness, or in healthy subjects undergoing intense exercise. It has shown that this score can differentiate degrees of breathlessness induced by exercise of varying intensity and also discriminate between healthy children and children with cystic fibrosis in terms of their exercise-induced breathlessness. It is simple to learn and easy to demonstrate, and can be used by anyone old enough to count fluently to 15 in any language. It is important to control certain variables when using the 15-count score (Prasad et al 2000). The speed of counting is important, as a subject counting quicker is likely to take fewer breaths (hence obtain a lower score) than someone counting more slowly. A consistent speed of counting is particularly critical if a subject is repeating scores after exercise or a therapeutic intervention. In practice, it is easy to count at the correct speed and it has been found to be quite consistent. A metronome is not recommended as it can confuse children who take extra breaths since they can become out of synchrony with the metronome’s beat. Another important variable to control is how deep a breath the subject takes at the start of the count, as a low starting lung volume will make it harder to reach 15 without taking further breaths. Subjects are told to take a deep breath, which is essentially to vital capacity, and although obviously it cannot be measured, it is not difficult to see whether the child has breathed in fully.
Two other objective scores of ease of breathing have been developed. These are the count score and the maximum phonation score (Orenstein et al 2002). The maximum phonation score requires the subject to sustain the syllable ‘Ahh’ using a single breath. The time, in seconds, for which the sound can be held in one breath gives the score. The count score requires the subject to count in a single breath at one number per second. The number reached gives the score. These two measures have been validated and shown to have good test–retest reliability for both rest and exercise conditions, reflecting increasing workloads and correlating with heart rate and SaO2.
The 15-count score and the ease of breathing scores are designed to give a more objective and reproducible measure of breathlessness than the subjective modified Borg and visual analogue scores. However, they do not take into account the child’s perception of their breath- lessness, or the intensity of the ‘unpleasantness’ of the breathlessness, something only the patient can know, and are therefore best used together with one of the subjective scores.
Fatigue
A muscle is considered to fatigue when its force output decreases for a given stimulus and fatigue can be quantified formally (Fulco et al 1995). However, in practical terms, it is simply a feeling of tiredness, which is more difficult to quantify. Like breathlessness, it is somewhat subjective and motivation plays a large part in its perception. It can be a generalized body fatigue or localized leg muscle fatigue, and levels of fitness tend to determine its onset. There are measures such as the Fatigue Severity Scale and the Fatigue Scale but these are best applied to everyday fatigue. For measuring exercise-induced fatigue, a visual analogue score can be used.
Chest pain
Non-specific chest pain is not that uncommon a symptom, particularly in adolescents, and exercise testing may offer reassurance to the child and family. Often no specific diagnosis is made, but exercise testing can certainly indicate whether the pain is due to chest tightness from exercise-induced bronchospasm. The inclusion of an ECG monitor will help distinguish true cardiac pain secondary to ischaemia from non-cardiac pain, such as muscular pain. Ratings of pain measurement exist but are not relevant here, as the exercise test should be terminated if chest pain occurs.
Safety of exercise testing in children
The safety of exercise testing in children, particularly in those with chronic disease, must be considered. There is little specific work with regard to this. However, Alpert and colleagues (1983) reviewed over 1500 studies performed in their laboratory over a 9-year period in which children, including those with congenital heart disease performed cycle ergometry testing to the point of fatigue. There were no deaths, and the total complication rate was 1.8%, which included chest pain, dizziness, syncope and hypotension. Hazardous arrhythmias occurred in only 0.46% of the population. In the authors’ laboratory, where more than 500 children have undergone progressive exercise tests using the cycle ergometer and the respiratory mass spectrometer, there have been no reported major complications.
Exercise testing in clinical situations
Asthma
Exercise-induced asthma (EIA) or bronchospasm (EIB) is a common manifestation of asthma in children and adolescents. Unfortunately, pre-exercise spirometry cannot predict response to physical activity and asthmatic subjects with normal lung function at rest may have severe exercise-induced airflow limitation. EIA has been used as an epidemiological measure of asthma in many countries and it is thought that between 40 and 95% of asthmatic patients experience a degree of EIA (McFadden Jr. & Gilbert 1994). It is likely that both the increased water loss from the respiratory tract and the cooling of the airways that occur by increased venti-lation during exercise contribute to EIA and EIB (Anderson et al 1985, Gilbert & McFadden Jr. 1992, McFadden Jr. & Gilbert 1994).
EIA or EIB onset is within 5–10 minutes of starting exercise, which usually results in a variable combination of wheeze, cough, breathlessness, and chest pain or tightness. After the normal transient bronchodilation, asthmatic children experience progressive bronchoconstriction, which is usually at its worst 5–10 minutes after stopping the exercise (when symptoms are also usually at their worst). Symptoms have usually gone within 15–30 minutes of stopping, and lung function is back to normal within 30–60 minutes (Cypcar & Lemanske Jr. 1994). The response is of course variable, with some patients able to continue the exercise with spontaneous resolution of their symptoms. Rarely, a late response can be elicited with symptoms reported 4–12 hours after exercise. The severity of the response is determined by exercise intensity, climate (worst in cold dry air) and baseline airway reactivity and function. Some sports are more likely to cause problems, e.g. long-distance running, cycling, soccer, rugby and cross-country skiing, while less provoking sports include swimming, tennis and gymnastics. In fact, the incidence of EIB, as diagnosed by exercise tests, among American Olympic athletes in the 1998 Winter Olympics was reported to be 23% (Wilber et al 2000).
Exercise testing therefore has an important place in the evaluation of asthmatic children, particularly those who only develop symptoms with exercise or in whom standard treatment is not helping these exercise-induced symptoms. Further, supervised exercise testing may reassure the child and their parents that they can actually perform exercise without adverse effects, particularly if bronchodilators are taken 15–20 minutes before the start. The optimal method is to employ a standardized exercise test and measure lung function before and after the exercise. Standardization is particularly important in the follow up of EIA over time when comparing individuals and in the comparison of epidemiological studies. FEV1 or peak flow are the main outcome measures for determining EIB. There are various regimens for formally testing known or suspected asthmatic children. One example involves the child running on a treadmill to elicit a heart rate of approximately 85% of age-predicted maximum, which in practice is usually up to 8 minutes of exercise. The speed and angle of the treadmill is adjusted to ensure adequate rise in heart rate. Peak flow is measured at baseline, then every 2 minutes until completion of exercise. Measurements then continue every 1 minute for 5 minutes (to ensure an early dip is not missed), then every 5 minutes for 30 minutes. A bronchodilator is then given to demonstrate reversibility using standard methods for testing bronchodilator responsiveness (see above). SaO2 and heart rate are monitored throughout the procedure. There are two main medical treatment principles for managing EIA: prophylactic treatment (anti-inflammatory) and premedication with bronchodilators prior to physical activity.
Cystic fibrosis
In the 36 years since Godfrey and Mearns (1971) showed that exercise was limited by pulmonary mechanics in children with cystic fibrosis (CF), these children have been subjected to all forms of exercise testing and there is now a wealth of publications in this area (Orenstein & Higgins 2005). The importance lies in the fact that patients with mild disease have a normal exercise tolerance. As the disease progresses, exercise tolerance diminishes but cannot be predicted from standard spirometry (Britto et al 2000). Maximal exercise testing has been shown to predict mortality in this disease, with those with higher aerobic fitness having a three times greater increase in 8-year survival compared with those with lower fitness, even after controlling for lung function and nutritional status (Nixon et al 1992). For these reasons, it is a recommended standard of care set by the Cystic Fibrosis Trust (United Kingdom) (2001) to perform exercise testing yearly to assess disease progression. In practice, this is difficult to achieve.
CF patients tend to have high respiratory rates and low tidal volumes during exercise, with a large proportion of each breath moving in and out of an increased dead space and not participating in gas exchange. In healthy children, some ventilatory reserve remains at peak exercise, with minute ventilation remaining less than two-thirds of their maximum voluntary ventilation (MVV). In patients with CF, it is not unusual for  E to reach or even exceed MVV at peak exercise; hence exercise is restricted by a mechanical ventilatory limitation. Other limiting factors include breathlessness, muscle fatigue, nutritional status and psychological parameters. In those with severe disease, ventilation–perfusion mismatching results in O2 desaturation and even CO2 retention; however, significant desaturation is unusual in those with an FEV >50% predicted (Henke & Orenstein 1984). Anaerobic performance is also limited in CF, principally due to nutritional factors (Boas et al 1996). Cardiovascular responses to exercise are normal in CF patients with mild disease. Those with moderate to severe disease may not attain expected peak heart rates and cardiac output, since non-cardiac factors, such as those already discussed, limit their ability to exercise before their cardiovascular systems are maximally stressed.
E to reach or even exceed MVV at peak exercise; hence exercise is restricted by a mechanical ventilatory limitation. Other limiting factors include breathlessness, muscle fatigue, nutritional status and psychological parameters. In those with severe disease, ventilation–perfusion mismatching results in O2 desaturation and even CO2 retention; however, significant desaturation is unusual in those with an FEV >50% predicted (Henke & Orenstein 1984). Anaerobic performance is also limited in CF, principally due to nutritional factors (Boas et al 1996). Cardiovascular responses to exercise are normal in CF patients with mild disease. Those with moderate to severe disease may not attain expected peak heart rates and cardiac output, since non-cardiac factors, such as those already discussed, limit their ability to exercise before their cardiovascular systems are maximally stressed.
Numerous studies have suggested that exercise training improves aerobic fitness, contains the decline in pulmonary function and may even extend longevity in children with CF, suggesting an improved prognosis and quality of life in physically active CF patients (Orenstein & Higgins 2005). This benefit was more likely when the exercise programmes were supervised, included activities such as running or swimming, lasted for at least 8 weeks and patient adherence was good. Exercise has also been shown to aid airway clearance (Zach & Oberwaldner 1987), but generally it is not recommended as a substitute for the usual physiotherapy techniques. In practice, however, it is some-times difficult to persuade those children who play regular sport and are fit, to carry out regular chest physiotherapy.
Heart-lung transplant
Measurement of exercise tolerance is an important part of heart-lung transplant assessment Severe exercise intolerance may predict life expectancy and provide information about the patient’s quality of life. Various exercise tests have been employed, including the 6-minute walk test (Nixon et al 1996) and the 3-minute step test (Aurora et al 2001). Further, exercise testing has been used to assess change in cardiopulmonary performance in a group of patients both before and after heart and lung transplantation (Schwaiblmair et al 1999).
Congenital heart disease
Exercise testing is an important part of evaluation of children with cardiac disease and the American Heart Association has produced comprehensive guidelines for paediatric testing (Washington et al 1994). Exercise testing is rarely diagnostic in congenital heart disease but has its greatest value in evaluating severity. In particular, it may reveal abnormalities that are not present at rest. Tomassoni (1996b) has outlined common reasons for exercise testing in the evaluation of cardiovascular disease:
 Objective assessment of exercise-induced symptoms, e.g. chest pain, palpitations, dizziness or syncope. In particular, dysrhythmias or ischaemia can be determined.
Objective assessment of exercise-induced symptoms, e.g. chest pain, palpitations, dizziness or syncope. In particular, dysrhythmias or ischaemia can be determined. Establishment of severity of obstructive heart disease such as aortic stenosis or coarctation of the aorta. The onset of exercise-induced ischaemia can be determined.
Establishment of severity of obstructive heart disease such as aortic stenosis or coarctation of the aorta. The onset of exercise-induced ischaemia can be determined. Determination of exercise tolerance and production of recommendations on what exercise it is safe for the child to take part in.
Determination of exercise tolerance and production of recommendations on what exercise it is safe for the child to take part in.In addition, the American Heart Association suggests that exercise testing should be avoided if there is evidence of:
Musculoskeletal disorders
A recent systematic review looking at aerobic fitness in children with juvenile arthritis showed that overall, there was evidence of reduced oxygen consumption during exercise when compared with controls (Takken et al 2002). Furthermore, a more recent study showed there was impairment in peak and mean power during the WAnT test (Takken et al 2005). These results may suggest that there is a degree of deconditioning in such patients as a result of functional limitations.
Neuromuscular disease
Children with neuromuscular diseases may lead a sedentary lifestyle compared with their peers, causing secondary detrainment and markedly reduced exercise tolerance. A recent Cochrane review suggests that these subjects benefit from both targeted aerobic and muscle training exercise programmes (van der Kooi et al 2005). Muscle function, for example muscle strength and endurance, peak mechanical power and effect on oxygen status of physical movement are most commonly tested. It is important to test several muscle groups, as depending on the condition, certain muscles are affected more than others. For more severe conditions, the arm-cranking version of the Wingate Anaerobic Test may be the only test that the patients can perform, as they are too weak to walk or cycle. Children with conditions such as spastic cerebral palsy may have reasonable muscle strength, but the difficulties of incoordination make testing difficult. In addition, the inefficiency of movement means that for a given task,  O2 is much higher in these children (Nixon & Orenstein 1988). At the advanced stage of diseases such as Duchenne muscular dystrophy, the heart muscles are also affected, further limiting the severely weakened movement ability.
O2 is much higher in these children (Nixon & Orenstein 1988). At the advanced stage of diseases such as Duchenne muscular dystrophy, the heart muscles are also affected, further limiting the severely weakened movement ability.
Exercise testing in children with neuromuscular diseases can be useful for (Bar-Or 1996):
 Monitoring progression of the disease, e.g. the rate of decline in thigh muscle strength, can be useful in patients with Duchenne or Becker’s muscular dystrophy.
Monitoring progression of the disease, e.g. the rate of decline in thigh muscle strength, can be useful in patients with Duchenne or Becker’s muscular dystrophy. Measuring effect of treatment, e.g. improvement in movement seen after a child with cerebral palsy has had a procedure such as surgical release of hip adductors or lengthening of Achilles tendons.
Measuring effect of treatment, e.g. improvement in movement seen after a child with cerebral palsy has had a procedure such as surgical release of hip adductors or lengthening of Achilles tendons.Scoliosis
Children with a marked scoliosis have a deformed thoracic cage that leads to a restrictive lung disorder, often accompanied by ventilation–perfusion mismatch. They may be able to perform spirometry, and arm span is substituted for height to calculate predicted values. If a scoliosis is secondary to neuromuscular disease, the child will have the additional burden of severe muscle weakness, which may make even spirometry impossible. Although patients with mild or moderate scoliosis do not exhibit cardiopulmonary restrictions in basal static conditions, they do show a significantly lower tolerance to maximal exercise (Barrios et al 2005). Respiratory inefficiency together with lower ventilation capacity and lower  O2max may be responsible for reduced exercise tolerance in adolescents with idiopathic scoliosis. Exercise deconditioning in scoliotic patients may also contribute to exercise intolerance. With severe scoliosis, exertional dyspnoea is common (Nixon & Orenstein 1988).
O2max may be responsible for reduced exercise tolerance in adolescents with idiopathic scoliosis. Exercise deconditioning in scoliotic patients may also contribute to exercise intolerance. With severe scoliosis, exertional dyspnoea is common (Nixon & Orenstein 1988).
Chronic lung disease of prematurity (CLD)
Advances in neonatal care have led to an increasing number of extremely premature neonates surviving. Since preterm birth occurs during a vulnerable period of lung maturation, the clinical outcome is determined by the circulatory and gas exchange capacity of the lungs, as well as the medical interventions required (these include oxygen therapy and intermittent positive pressure ventilation). This may lead to considerable respiratory morbidity and preterm subjects with persistent oxygen dependency after 36 weeks corrected gestational age are defined as having CLD (Jobe & Bancalari 2001). Because cardiopulmonary limitations may not be clinically evident while the child is at rest, exercise testing may be useful in schoolage children born preterm and who developed CLD, to determine the presence and extent of any dysfunction of gas exchange secondary to alveolar growth impairment.
There are no longitudinal studies examining exercise performance in children who were preterm and had a low birthweight. In one cross-sectional study (Mitchell & Teague 1998), during treadmill exercise, the CLD and ex-preterm groups had increased wheezing and coughing. Although lung function did not change significantly during exercise, recovery was accompanied by a significant reduction in FEV1, both in the CLD and ex-preterm groups compared with the controls. The DLCO increased significantly above pre-exercise in the control and ex-preterm groups but not in CLD group, suggesting problems with recruitment and/or distension of the alveolar capillary bed. The cardiac output was also significantly lower in the CLD group during exercise. In contrast, two other groups have shown normal cardiac responses to exercise (Jacob et al 1997, Pianosi & Fisk 2000). Pianosi & Fisk (2000) showed that cardiac output was normal in CLD subjects but found that these subjects had a higher respiratory rate and maintained a lower end-tidal CO2 during submaximal exercise when compared with ex-preterm healthy term control groups. Santuz et al (1995) used an incremental protocol on a treadmill and exercised CLD subjects until exhaustion and measured expired gases throughout. The CLD group had exercise- induced bronchospasm, and a lower  O2max,
O2max,  Emax and a higher respiratory rate compared with controls, indicating both reduced aerobic capacity and ventilatory adaptation. There was no correlation between exercise performance and resting pulmonary function. Using the same exercise protocol, Bader and colleagues (1987) observed a significantly lower
Emax and a higher respiratory rate compared with controls, indicating both reduced aerobic capacity and ventilatory adaptation. There was no correlation between exercise performance and resting pulmonary function. Using the same exercise protocol, Bader and colleagues (1987) observed a significantly lower  Emax with a normal
Emax with a normal  O2max in the CLD group than in the control group.
O2max in the CLD group than in the control group.
In summary, there does appear to be sufficient evidence to suggest exercise impairment and ventilatory adaptation during exercise in subjects with CLD. The conflicting results may be in part due to the different techniques employed during exercise testing. A detailed review of this topic has been published (Narang et al 2006).
Obesity
Obesity is becoming a serious problem among an ever-increasing number of children, due to poor eating habits and a sedentary lifestyle, with approximately 15% of children in the UK being classified as obese (Owens & Gutin 1999). Paediatric obesity commonly precedes adult obesity and is associated with the development of weight-related co-morbid conditions and increased morbidity (Must 1996, Must et al 1992). Exercise is said to be a cornerstone of treating paediatric obesity along with dietary and behaviour modification. One study showed that, of 20 obese children who attended a ‘camp’ to help with weight reduction which incorporated exercise prescription, 16 showed a significant reduction in their body mass index (Deforche et al 2003); however, not all the research on the benefits gained by training has been conclusive (Epstein et al 1996). Exercise testing may be a useful way of monitoring progress, and may also be used to alter the exercise prescription with ongoing weight loss. The cardiopulmonary response of the obese child to exercise has recently been reviewed (Owens & Gutin 1999), and it was concluded that the physiological changes are similar to non-obese children. Graded exercise tests on the treadmill produced similar maximum heart rates in obese and non-obese children (Maffeis et al 1994). Similarly, cardiac output values were not different at 33% and 66% of maximal work capacity in obese and non-obese children (Davies et al 1975). However, they may have a higher oxygen demand during submaximal exercise due to the increased metabolic demand of having to move their excess weight (Norman et al 2005). Cardiac constraints may also play a role and this warrants closer, more detailed evaluation (Norman et al 2005). The current literature suggests that exercise testing can be successfully accomplished with most obese children and that the key is to be sensitive and instil confidence in these children at the time of testing as these factors alone can affect exercise performance (Owens & Gutin 1999).