Chapter 9 Paediatric mechanical support
INTRODUCTION
The technique of respiratory support in children needs to be informed by an understanding of age-specific pathophysiology. In contrast to adults, the most common causes of respiratory failure in children vary with age. In the newborn infant, prematurity, hyaline membrane disease, asphyxia and aspiration pneumonia are the most common causes of breathing difficulty. Under 2 years of age, bronchopneumonia, bronchiolitis, croup, status asthmaticus, foreign body inhalation and congenital heart and airway anomalies are important; compared with asthma, accidental poisoning and central nervous system infection, trauma and cerebral hypoxia/ischaemia in the over 2 year olds. Although the principles of when and how mechanical support should be undertaken in such patients are, broadly speaking, similar to those applied in adults, there are differences in epidemiology, pathophysiology and management that warrant consideration. The emphasis, therefore, of this chapter will be a paediatric perspective of respiratory supportive therapy.
EPIDEMIOLOGY OF ACUTE RESPIRATORY FAILURE IN CHILDREN
Respiratory failure develops when the rate of gas exchange between the air and the blood fails to match the body’s metabolic demands. The patient therefore loses the ability to provide sufficient oxygen to the blood and develops hypoxaemia, or the patient is unable to ventilate adequately and develops hypercarbia. Epidemiologically, there is little information in children about the incidence of acute respiratory failure. Adult definitions using blood gas parameters may be appropriate for certain age groups but in others they may not be useful. For example, in infants with acute bronchiolitis, acute respiratory failure is usually defined as:
PaCO2 = 8 kPa (60 mmHg) with PaO2 = 8 kPa (60 mmHg) when using FiO2 = 0.6
(where PaCO2 = partial pressure of carbon dioxide in arterial blood, PaO2 = partial pressure of oxygen in arterial blood, FiO2 = fractional inspired oxygen concentration) or, in the case of patients with respiratory arrest, a preceding history of severe respiratory distress accompanied by cyanosis.
However, when trying to look at large populations, in the absence of blood gases, a more pragmatic definition for acute respiratory failure is needed. For example, when using the definition of ‘acute airway management necessitating endotracheal tube intubation’ (Tasker 2000) it is possible to explore issues such as the pattern and time course of paediatric disease that have some bearing on how mechanical support should be undertaken.
Pattern of and time course of disease
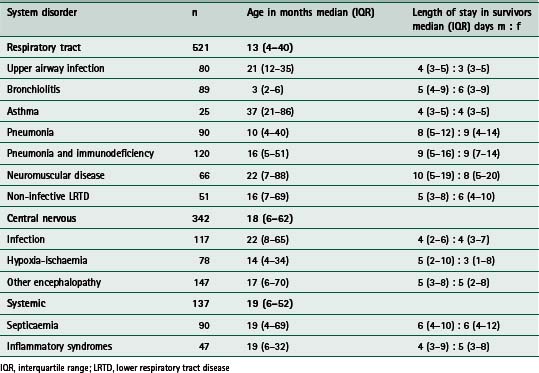
Table 9.1 summarizes a retrospective analysis of 1000 infants and children (aged older than 28 days and younger than 17 years) who required endotracheal intubation for acute respiratory failure complicating acutely acquired medical, rather than surgical, disease (Tasker 2000). The three major categories relate to the system or problem underlying respiratory failure (respiratory tract disorder, central nervous system disorder or systemic disorder) and the subcategories relate to the clinical diagnostic entities commonly encountered in intensive care. Respiratory tract problems due to infection are, not surprisingly, the most common problems seen. The time course of recovery in survivors is influenced by the site within the airways that infection has reached. This is reflected by an increase in the length of stay in the intensive care unit (ICU) with more distally affected tissues (i.e upper airway compared with lower airway). In relating such information to clinical practice one can use the expected time course to decide on an agenda for treatment or ‘care pathway’. For example, given that the expected time course for intensive care recovery in pneumonia necessitating intubation is around 8 days (interquartile range 5–12 days) one can then predict when certain targets should be met. The same applies to the other 11 distinct diagnosis-related entities. This idea will be revisited later in this section where three clinical examples are discussed.
Acute hypoxaemic respiratory failure
Acute hypoxaemic respiratory failure (AHRF) signifies respiratory failure at the more severe end of the pathophysiological spectrum, irrespective of underlying aetiology. For paediatric practice we identify this state by using diagnostic criteria that have been modified from the American-European Consensus Conference diagnostic criteria for acute respiratory distress syndrome (ARDS) (Bernard et al 1994). These criteria include:
 evidence of a severe defect in oxygenation (PaO2/FiO2 of less than 26.7 kPa, 200 mmHg) for at least 6 consecutive hours on the day of admission
evidence of a severe defect in oxygenation (PaO2/FiO2 of less than 26.7 kPa, 200 mmHg) for at least 6 consecutive hours on the day of admissionChildren meeting all the above criteria except the characteristic chest radiographic appearances of ARDS (last criterion) are described as cases of AHRF. The significance of AHRF is that it implies a certain severity of illness and risk of mortality, factors which are important when it comes to deciding which ventilatory strategy should be adopted and the use of adjunctive therapies (see section on ‘ventilation strategies for specific disease’). For example, in a prospective, epidemiological study, Peters and colleagues (1998) found that out of 850 mechanically ventilated infants and children, AHRF occurred in 118 patients (14%, 95% confidence interval (CI) 12–16%). Of these 118 patients, 52 met the criteria for ARDS (44%, 35–53%). In all 850 patients, mortality was four times higher than the mortality seen in those patients without AHRF. In the AHRF patients, mortality was three times higher for those with ARDS (Peters et al 1998). In a study from North America, the PALISI network (Pediatric Acute Lung Injury and Sepsis Investigators) reported a mortality from ARDS of 4.3% (Randolph et al 2003). This coincides with the adoption of protective modes of ventilation for this condition. Therefore, identifying these entities (i.e. AHRF and ARDS) at an early stage is important so as to institute the most appropriate method or mode of ventilation.
INDICATIONS FOR SUPPORTIVE RESPIRATORY THERAPY
For practical purposes we can consider the treatment of respiratory dysfunction in terms of treating hypoxia and hypercarbia. Appropriate management is aimed first at prevention, second at early diagnosis and third at a clear understanding of the pathophysiology and way in which the proposed treatment works to maintain or restore good lung function.
Hypoxia
Hypoxia must be treated first; give oxygen. At the same time attempts should be made to correct the underlying problem. Local processes in the lung, such as atelectasis and bronchopneumonia, can result in a portion of the pulmonary blood flow perfusing unventilated alveoli (i.e. intrapulmonary shunt), which in some cases may be effectively treated by pulmonary toilet and postural change. With a large shunt fraction – greater than 25% of pulmonary blood flow – PaO2 is not significantly improved by solely increasing the FiO2. In these cases a diffuse pulmonary process is usually present and a form of assisted positive airway pressure is required. Such assistance may also be required for severe impairment of chest wall mechanics (e.g. rib fractures, pain, weakness, etc.) even in the absence of pulmonary parenchymal disease.
In infants and children, there are several methods of administering oxygen (Table 9.2). Young patients do not usually tolerate nasal catheters and cannulae. Facemasks with a reservoir and a non-rebreathing valve can be used to increase the FiO2. Alternatively, high-flow oxygen via the appropriate Venturi-valve mask can be used. Oxygen delivered via the oxygen inlet of an incubator rarely exceeds an FiO2 of 0.4. When supplemental oxygen is delivered into an tent the concentration varies depending on any leaks in the system. Regardless of the technique, it is essential that the administered oxygen is warmed and humidified. To avoid damage to the lungs, oxygen administration should be discontinued as soon as possible (as indicated by blood gas measurements). An FiO2 below 0.6 is preferred, to minimize the risk of oxygen toxicity. Reduction in the FiO2 should be carried out cautiously in a stepwise manner. To facilitate this process both the concentration and duration of oxygen therapy must be recorded accurately. A well-calibrated oxygen analyser is used to check the inspired concentration every 2 hours when using a tent, head box or when adding oxygen into an incubator. The need for monitoring PaO2 in preterm newborn infants is related to the potential for pulmonary oxygen toxicity and the danger of retrolental fibroplasia. So, in any patient, oxygen should be administered at the lowest concentration sufficient to maintain the PaO2 between 6.7 and 13.3 kPa (50–100 mmHg). Continuous measurement or monitoring of transcutaneous oxygen or pulse oximetry arterial oxygen saturation (SpO2) are essential additions to the direct, and intermittent, measurement of arterial blood gases. In some instances, supplemental oxygen may cause respiratory depression if there has been chronic respiratory failure and the patient has a hypoxic-drive to ventilation (as opposed to the normal situation where the PaCO2 is the most important factor in the control of ventilation). This phenomenon is generally uncommon in paediatric practice, but has been encountered in children with cystic fibrosis, cerebral palsy and bronchopulmonary dysplasia.
Table 9.2 Methods of oxygen administration
| Method | Maximum achievable FiO2 at 6–10 l/min of oxygen (%) |
|---|---|
| Nasopharyngeal catheter | 50 |
| Nasal prongs | 50 |
| Mask without reservoir bag | 50 |
| Mask with reservoir bag (partial rebreathing) | 70 |
| Mask with reservoir bag (non-rebreathing) | 95 |
| Venturi | 24, 28, 35, 40 |
| Incubator | 40 |
| Canopy tent | 50 |
| Head box | 95 |
FiO2, fractional inspired oxygen concentration
In addition to oxygen, some type of positive pressure may be useful in the management of hypoxia. Mask and nasal continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP) increase lung compliance by recruiting additional areas of the lung for ventilation. Also, lung recruitment improves oxygenation by decreasing intrapulmonary shunt. These modes of non-invasive ventilation (NIV) are being used more frequently as the step before invasive mechanical ventilation. When invasive mechanical ventilation is used, the addition of some positive end-expiratory pressure (PEEP) is a common practice in maintaining adequate functional residual capacity. However, PEEP may adversely affect the patient lung mechanics if hyper-inflation occurs. This problem results in impaired pulmonary perfusion and further accentuates any ventilation–perfusion (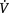 /
/ ) mismatch. Therefore PEEP above 4 cmH2O should be used judiciously if there is already regional hyperinflation, such as occurs in bronchopulmonary dysplasia (Box 9.1). In this context a strategy for treating hypoxia is outlined in Box 9.2.
) mismatch. Therefore PEEP above 4 cmH2O should be used judiciously if there is already regional hyperinflation, such as occurs in bronchopulmonary dysplasia (Box 9.1). In this context a strategy for treating hypoxia is outlined in Box 9.2.
Hypercarbia
When shallow (or ineffectual) breathing is present, the dead space (i.e. ventilated but non-perfused regions) becomes a larger fraction of each breath. This change results in a decrease in alveolar ventilation, even if the lung parenchyma is normal. When hypercarbia has been found and its cause considered, NIV can be tried a step before invasive mechanical ventilation. This may be useful when hypercarbia is secondary to neuromuscular weakness. If the patient needs invasive mechanical ventilation, then increasing tidal volume or respiratory rate can bring about increased alveolar ventilation. However, these changes may increase mean airway pressure that may generate detrimental effects on pulmonary vascular resistance and 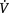 /
/ matching.
matching.
If the patient is treated with full mechanical ventilation, the first step is to make sure that the patient is receiving an appropriate tidal volume and minute ventilation. Ventilatory system leaks and loss of a portion of the tidal volume through compressive loss in the tubing, as well as abnormalities in endotracheal tube function, are common problems that need rectifying. Having excluded mechanical factors, the other causes of hypercarbia may be related to an increase in CO2 production or an increase in dead-space ventilation. An increase in dead space may be due to excessive PEEP (particularly when there is already hyperinflation or hypovolaemia) and it may be corrected by intravenous volume to increase preload to the heart.
Endotracheal intubation
There are four absolute indications for controlling the airway by endotracheal intubation:
 maintaining the patency of the airway where problems are present or anticipated (e.g. direct airway trauma, oedema or infection)
maintaining the patency of the airway where problems are present or anticipated (e.g. direct airway trauma, oedema or infection) to protect the airway from aspiration in states of altered consciousness, where airway-protective mechanisms may be lost or impaired
to protect the airway from aspiration in states of altered consciousness, where airway-protective mechanisms may be lost or impaired to facilitate pulmonary toilet and avoid airway obstruction when there is marked atelectasis and pulmonary infection – an inadequate cough might necessitate more direct access to the airways for suctioning
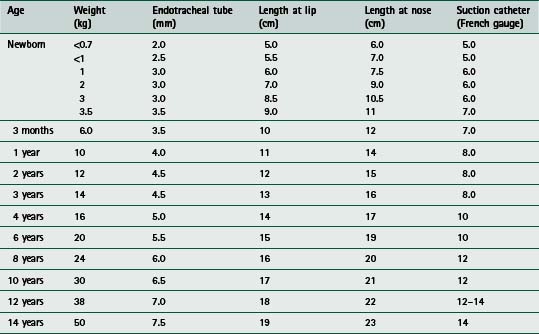
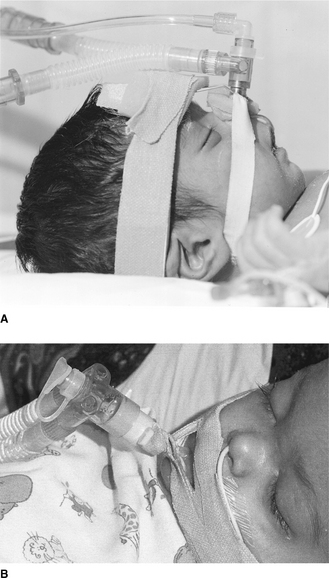
to facilitate pulmonary toilet and avoid airway obstruction when there is marked atelectasis and pulmonary infection – an inadequate cough might necessitate more direct access to the airways for suctioningIn practice, experienced staff should carry out establishing airway and respiratory support for the acutely ill child, because such patients can deteriorate rapidly, particularly at the time of inducing anaesthesia. Following preoxygenation with 100% inspired oxygen, a variety of agents are used to facilitate endotracheal intubation, including intravenous induction with drugs such as fentanyl, midazolam and suxamethonium, or inhalational induction with gases such as halothane or isoflurane. Table 9.3 provides a guide to the appropriate endotracheal tube size, length and suction catheter used in the paediatric age range and Figure 9.1 illustrates a commonly used method of endotracheal tube fixation.
MECHANICAL VENTILATION
Many of the ventilatory techniques are similar for children and adults. However, there are some differences between these two groups that are highlighted below.
General ventilatory care
A variety of ventilators can be used in paediatric mechanical ventilation (Fig. 9.2). There are specific ventilators designed for neonates and small infants that are used mainly in neonatal units to ventilate premature babies. Fortunately most modern ventilators can be used across the whole age spectrum and they can deliver different modes of pressure control ventilation (PCV) and volume control ventilation (VCV).
One of the main goals during mechanical ventilation is to minimize and optimize patient–ventilator interaction. Adequate patient comfort will potentially decrease the need for pharmacological sedation and may help to minimize the duration of mechanical ventilation. The addition of PEEP at 3–5 cmH2O above atmospheric pressure is routinely used unless there is a contraindication. This helps to avoid alveolar collapse during mechanical ventilation. The amount of PEEP may need to be increased in conditions associated with low lung volume, such as hyaline membrane disease, atelectasis, severe pneumonia (e.g. viral, Pneumocystis carinii) or ARDS.
Complications of ventilator therapy occur frequently and all intensive care staff should be continually aware of the potential hazards (Box 9.3). Aseptic technique is important for tracheal airway care because nosocomial infection constitutes a large and preventable problem. The application of PEEP, increased tidal volumes and increased airway pressure can also produce complications. Potential disruption of normal 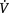 /
/ matching seen with spontaneous breathing can occur with lung over-expansion and leads to regional hypoperfusion. A decrease in venous return, an increase in pulmonary vascular resistance and a decrease in left ventricular output can impair cardiac output and oxygen delivery. The more compliant the lung or the less compliant the chest wall, the greater the transmission of positive airway pressure to the mediastinum and the greater the negative effect on cardiac function. Volume loading or inotropic support can overcome the concomitant decrease in cardiac output, in large part.
matching seen with spontaneous breathing can occur with lung over-expansion and leads to regional hypoperfusion. A decrease in venous return, an increase in pulmonary vascular resistance and a decrease in left ventricular output can impair cardiac output and oxygen delivery. The more compliant the lung or the less compliant the chest wall, the greater the transmission of positive airway pressure to the mediastinum and the greater the negative effect on cardiac function. Volume loading or inotropic support can overcome the concomitant decrease in cardiac output, in large part.
The significance of ventilator-induced lung injury (VILI) has been appreciated more during the last few years. It now seems clear that the pathogenesis of respiratory failure is greatly influenced by the way the lungs are ventilated. Barotrauma and volutrauma have been used to describe VILI. However, there is some controversy as to which of these is the more damaging. Both can produce overdistension and it is this problem that appears to cause the injury. To complicate matters, another factor appears to be ‘atelectrauma,’ i.e. where the repeated collapse and re-expansion of areas of the lung produce shearing injury that contributes to inflammation and lung damage. The best ventilator strategy should therefore aim to keep tidal volume to a minimum with optimum PEEP and, sometimes, neuromuscular blockade. Theoretically, these manoeuvres should reduce atelectasis and patient–ventilator interactions.
In regard to other problems, pulmonary interstitial emphysema, pneumomediastinum, pneumoperitoneum and subcutaneous emphysema do not require specific treatment unless there is significant haemodynamic impairment. Poor renal function, as exhibited by decreased glomerular filtration rate, urine production and sodium excretion, can be a consequence of hypoxia and hypercarbia. This may be further compounded by the effects of mechanical ventilation with PEEP on producing an antidiuretic hormone-mediated salt and water-retaining effect (probably secondary to decreased cardiac output), an increased renal vein pressure and a neural reflex from the pressure-distorted atrial wall.
Acute deterioration and ‘troubleshooting’
In mechanically ventilated patients, the adequacy of gas exchange and ventilation should be assessed frequently. Changes in therapy should then be titrated against expected parameters or targets (Table 9.5). When there is an acute deterioration during mechanical ventilation, problem solving or ‘troubleshooting’ should begin with making the patient safe. In the first instance this means disconnecting the patient from the ventilator and support breathing with bag-ventilation using FiO2 of 1.0. Easy ventilation and patient stabilization with the bag suggests a ventilator problem that should be systematically addressed (e.g. check the circuit for leaks, check ventilator function, check gas flow). However, it should be remembered that ‘hand-bagging’ might result in increased tidal volume, which can also be responsible for the patient’s improvement. Patients with stiff lungs are frequently dyspnoeic, despite adequate gas exchange. Increasing the tidal volume will correct this subjective feeling and may also account for patient improvement. Difficult bagging at the time of disconnecting the ventilator strongly suggests a problem with the endotracheal tube or the lung to chest-wall complex. A suction catheter (Table 9.3) should be passed down the endotracheal tube to check for narrowing or blockage. Chest examination, blood gases and chest radiography should be ordered. A blocked endotracheal tube should be replaced. A pneumothorax requires chest tube placement. If neither of these is the cause for deterioration, then the possibilities may include new problems such as an increased oxygen demand due to sepsis, impaired oxygen delivery due to heart failure or acute pulmonary injury due to gastric aspiration. These and other causes need to be sought and treated appropriately.
Commonly used modes of mechanical ventilation in children
Pressure versus volume control
In VCV a set tidal volume is delivered and the peak inspiratory pressure (PIP) will depend on lung compliance, inspiratory flow and airway resistance. The plateau pressure is a more accurate measurement of actual alveolar pressure and it will be mainly influenced by lung compliance. The ventilator rate and inspiratory time are set by the clinician. This mode has the advantage of guaranteeing minute ventilation, especially in conditions when optimum minute ventilation is required (e.g. head injury). However, a decrease in compliance can lead to excessively high pressures with risk of lung injury (Table 9.4).
Table 9.4 Advantages and disadvantages of pressure-limited and volume-limited ventilation
| Pressure-limited | Volume-limited | |
|---|---|---|
| Advantages | Avoids excessive inflating pressures Decreased risk of barotrauma |
Constant volume delivered High inflating pressures reflect changes in mechanics |
| Disadvantages | Variable volume delivered No signs of altered mechanics |
Capable of generating very high inflating pressures Increased risk of barotrauma |
In PCV the delivered breath is limited by pressure. The tidal volume is determined by preset pressure limit, inspiratory time and lung compliance; it may also vary with the condition of the lung. The flow is decelerating, meaning that it slows down progressively after reaching the set inspiratory pressure. As in VCV, the ventilator rate and inspiratory time are set by the clinician. PCV has the advantage of decelerating flow and it has been considered to be less injurious because lower pressures are usually achieved, compared with volume control. However, a decrease in lung compliance, such as that caused by accumulation of secretions, may be associated with a decrease in tidal volume. This situation may go unrecognized because the ventilator will continue to cycle at the preset pressure (Table 9.4).
There are few controlled studies in children comparing these various modes of ventilation. Usually the final choice of ventilator mode depends on personal experience, the availability of technology and the underlying disease. Whatever mode is chosen it is essential to ensure that the expected tidal volume, minute ventilation and pressures are achieved.
Low tidal volume ventilation
In patients with AHRF or ARDS, ventilation using low tidal volumes has been widely accepted. In 2000, the ARDS network reported a decreased mortality in adults when using tidal volumes of 6 ml/kg as compared to 12 ml/kg (Acute Respiratory Distress Syndrome Network 2000). These results are probably applicable to children. The risk of using smaller tidal volumes for mechanical ventilation is that it can lead to insufficient gas exchange and hypercarbia. Therefore, the strategy commonly adopted when using low tidal volume ventilation is to accept a higher PaCO2 provided the arterial pH is 7.25. In practice, the ventilator is set so that peak inspiratory pressures are limited below 30 cmH2O (30 cmH2O of plateau pressure when using volume control) while employing high mean airway pressures to ensure maximum lung volume recruitment via the use of PEEP.
Pressure-regulated volume control (PRVC) ventilation
This mode of ventilation is now available in some newer ventilators. This mode delivers a preset tidal volume in a pressure-limited manner using the lowest possible pressure with a decelerating flow. To guarantee the set tidal volume, the gas flow and pressure change constantly in each delivered breath, depending on lung compliance and airway resistance. This method has the advantage of guaranteeing tidal volume and using decelerating flow. As lung compliance improves during the course of pulmonary disease, the ventilator will automatically wean inspiratory pressures. Even though PRVC seems to have advantages over other modes, clinical controlled trials to evaluate its benefits are still lacking in children.
NEWER VENTILATORY SUPPORT TECHNIQUES
High-frequency oscillatory ventilation
High-frequency ventilation techniques, including high-frequency positive pressure ventilation, high-frequency jet ventilation and high-frequency oscillatory ventilation (HFOV), achieve adequate ventilation by employing tidal volumes that are often less than actual dead space and respiratory rates of 60–3000 cycles/minute (Fig. 9.3). The most widely used of these modes in children is the HFOV. The theoretical advantage of HFOV is that it keeps the lungs open by using a relatively high mean airway pressure and it delivers very low tidal volumes, thereby minimizing lung damage caused by high volumes and high inspiratory pressures. The high mean airway pressure allows lung-volume recruitment and improves oxygenation.
The most common indication for this form of ventilation is refractory hypoxaemia. However, HFOV has been used in bronchopleural fistula and some types of obstructive disease and bronchiolitis (Kneyber et al 2005, Slee-Wijffels et al 2005). Unfortunately there is a paucity of published clinical trials on the use of HFOV in the paediatric population and its benefits have not been clearly established as compared with conventional mechanical ventilation. Despite this lack of evidence, HFOV is frequently used in the treatment of hypoxemic respiratory disease. When used, early institution of this therapy seems to be more beneficial in this group of patients (Arnold et al 1994). The experience reported by Watkins and colleagues (2000) in 100 courses of such ventilation would suggest that, in the presence of AHRF or ARDS, a threshold mean airway pressure of 16 cmH2O is an appropriate indication.
Extracorporeal membrane oxygenation
Extracorporeal membrane oxygenation (ECMO) is designed to provide a variable degree of cardiopulmonary support for a predetermined period of time over which the underlying pulmonary disorder is expected to recover. Potentially, ECMO allows recovery without subjecting the lungs to the risks of VILI or oxygen toxicity. Venoarterial systems may be used to completely take over the child’s own heart and lung function (Fig. 9.4), although in practice extracorporeal flows may be limited by venous drainage (usually from the right internal jugular vein). Venovenous systems have been used for CO2 removal, although a retrospective study concluded that venovenous ECMO could effectively provide adequate oxygenation for children with severe acute respiratory failure (Pettignano et al 2003). In the complete absence of pulmonary function, venovenous ECMO will provide SpO2 of 80%. Using extracorporeal support, success has been achieved in some children with acute lung injury (Pearson et al 1993). However, appropriate patient selection is a critical and contentious issue. One method, proposed by Bartlett (1982, 1990), is to identify neonates at high risk for failing to respond to conventional therapy by applying an index of oxygenation, which is related to the mean airway pressure and the FiO2 during mechanical ventilation, and the achieved PaO2:
Oxygenation index (OI) = (mean airway pressure × FiO2 × 100)/PaO2 (mmHg)
In Bartlett’s proposal (1990), OI greater than 25 predicted 50% mortality and OI greater than 40 predicted 80% mortality. Anecdotally, in children, Goldman and colleagues (1997) have found in meningococcal sepsis that ARDS that fails to respond to HFOV may be reasonably treated with ECMO.
VENTILATION STRATEGIES FOR SPECIFIC DISEASE
In paediatric practice there are some specific diseases or problems that do require a specific ventilatory strategy. These issues are illustrated by the examples that follow.
Acute bronchiolitis
The typical features of bronchiolitis are:
 acute generalized peripheral airway obstruction (‘air trapping’) with tachypnoea, decreased breath sounds and low hemidiaphragms on chest radiography
acute generalized peripheral airway obstruction (‘air trapping’) with tachypnoea, decreased breath sounds and low hemidiaphragms on chest radiographyRespiratory syncytial virus (RSV) is the most frequent cause of bronchiolitis. Other viral causes include adenovirus, influenza and parainfluenza viruses and rhinovirus. Cytomegalovirus can produce a bronchio-litis or pneumonitis-like illness in immunocompromised children. Rare non-viral causes of the bronchiolitis syndrome include Mycoplasma pneumoniae and Bordetella pertussis infection.
Sixteen per cent of infants hospitalized for RSV have apnoea and its course is usually short-lived. Clinically these episodes are diaphragmatic or non-obstructive with complete absence of respiratory effort. In these cases, endotracheal intubation with minimal support is required until the problem resolves. In patients with worsening respiratory distress due to pulmonary parenchymal changes, mechanical support does not necessarily require endotracheal intubation. In some instances, nasopharyngeal prong CPAP, which maintains positive transpulmonary pressure during spontaneous breathing, can be used to avoid mechanical ventilation. When infants with RSV infection require mechanical ventilation, there are many similarities with mechanical ventilation of adults with status asthmaticus (Box 9.4). Clinical observation of inspiratory and expiratory chest excursion as well as regular auscultation is important if overventilation, with associated hyperinflation and barotrauma, is to be avoided. The aim is to maintain or achieve adequate arterial oxygenation and control of respiratory acidosis. This may even necessitate ventilating at slow rates with prolonged expiratory times to permit adequate CO2 clearance. Low levels of PEEP are used to decrease airway resistance and improve gas exchange, although in studies of lung mechanics this has not been verified. The presence of inadvertent or auto-PEEP requires that extrinsic PEEP be applied to the same level in order to maintain expiratory flow.
Box 9.4 Ventilation of acute bronchiolitis
| Oxygenation | ||
| Aim: | PaO2 9–10 kPa | Using PEEP and supplemental oxygen |
| If: | FiO2 ≥0.6 | Increase PEEP |
| If: | PaO2 ≥11.3 kPa | Decrease FiO2 to 0.6: —then decrease PEEP to 4 cmH2O —then decrease FiO2 |
| Carbon dioxide | ||
| Aim: | PaCO2 5.3–6.5 kPa | Using rate 10–15/min, tidal volume 15 ml/kg or whatever PIP to achieve adequate chest movement If PIP ≥30 cmH2O or if agitated then paralyse |
| If: | PaCO2 ≥7 kPa | Pressure-limited ventilation: —increase PIP before rate if chest excursion inadequate Volume ventilation: —increase minute ventilation with rate up to 20/min |
| If: | PaCO2 ≤4.7 kPa | Lower tidal volume to 10 ml/kg: —then PIP to 25 cmH2O —then rate |
| Extubation criteria | ||
| PaO2 ≥9.3 kPa | PEEP/CPAP ≤4 cmH2O FiO2 ≤0.4 |
|
| PaCO2 ≤6 kPa | Rate ≤6/min | |
| Continue with supplemental oxygen until SpO2 .0.95 in room air | ||
In patients with hyperinflation the ventilatory strategy should aim to limit ventilator-associated dynamic hyperinflation and impaired minute ventilation. The ventilator should be set at a slow rate (10–15 breaths/minute) and with a prolonged expiratory time. Time-cycled, PCV is used in this instance while aiming for an arterial pH >7.25 and a SpO2 88–92%. When indicated, neuromuscular blockade and antibiotics will need to be prescribed. All patients should receive adequate analgesia and sedation during mechanical ventilation. Bronchodilators can be administered if patients demonstrate a therapeutic response to an initial trial dose. In the acute phase of illness, fluids, electrolytes and hydration must be closely monitored while generally restricting fluid to 67–75% of maintenance requirements. In the weaning phase, patients can be removed from ventilatory support when safe to do so. In regard to blood gas parameters this means, in general, adequate oxygenation in an FiO2 <0.4 and normal pH, with good respiratory drive, in the absence of hypercarbia. Discharge from the intensive care unit can then be considered once the patient has managed at least 12–24 hours without any respiratory assistance. Overall these patients will, on average, spend about 7 days (interquartile range 4–8 days) on the intensive care unit (Tasker et al 2000).
In about one-fifth of mechanically ventilated patients with RSV more severe disease is seen (Tasker et al 2000). In this instance, more extensive pulmonary pathology results in a picture of pneumonitis with diffuse alveolar consolidation rather than bronchiolitis with lung hyperinflation. These infants have the clinical features of ARDS as they exhibit four-quadrant consolidation on chest radiograph. The aim of mechanical support should be to recruit lung volume with the addition of PEEP. Sometimes HFOV or ECMO is required if lung injury becomes more extensive with likely development of interstitial emphysema and pneumothorax. The time course of this problem is very different to the usual course of bronchiolitis and, on average, patients spend at least 2 weeks on the intensive care unit (Tasker et al 2000).
Acute hypoxaemic respiratory failure
In patients with AHRF or ARDS the low tidal volume strategy in addition to PEEP helps to minimize VILI. The level of PEEP should be adjusted so as ensure maximum lung volume recruitment. Adequate recruitment improves oxygenation and avoids repeated alveolar collapse and reopening that lead to shear injuries. If oxygenation continues being inadequate with a mean airway pressure of 16 cmH2O or greater, then HFOV should be considered, particularly if the problem is one of diffuse parenchymal changes or consolidation. A series of adjunctive therapies have been proposed for this condition.
Prone positioning
Prone positioning has been shown to improve oxygenation during hypoxaemic respiratory disease in a number of adult and paediatric studies (Curley et al 2005, Pelosi et al 2002). These observations together with the simplicity of the procedure have justified the inclusion of this technique as standard practice. The incidence of serious adverse effects has not been reported to increase during prone position and only mild complications (e.g. facial oedema and pressure ulcers) have been described. During ventilation in the supine position the dorsal areas of the lung become preferentially consolidated or collapsed. The change to prone position allows recruitment of the dorsal areas of the lung and generates a more even distribution of ventilation and improved 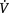 /
/ matching. Unfortunately, even though the change in position seems to improve oxygenation during acute lung injury (ALI), it does not appear to have an impact on outcome. A recent study failed to show a reduction in ventilator-free days or mortality in children with ALI ventilated in prone position as compared with supine position (Curley et al 2005).
matching. Unfortunately, even though the change in position seems to improve oxygenation during acute lung injury (ALI), it does not appear to have an impact on outcome. A recent study failed to show a reduction in ventilator-free days or mortality in children with ALI ventilated in prone position as compared with supine position (Curley et al 2005).
Recruitment manoeuvres
Recruitment manoeuvres are aimed at recovering collapsed areas of the lung during ALI, especially after specific manipulations such a suctioning and disconnection of the ventilator. A variety of manoeuvres have been described including increases in the level of PEEP while maintaining constant inspiratory pressures, prolonged increases in pressure (i.e. 40 mmHg for 40 seconds) as a single manoeuvre or using stepwise increases, and the use of high tidal volumes. There are no controlled studies and there is no consensus regarding these manoeuvres and their safety in children.
Nitric oxide
Nitric oxide (NO) is an endogenous, endothelium-derived vasodilator. Inhaled NO has the theoretical advantage of producing selective reduction in pulmonary vascular resistance. The fact that inhaled NO acts on the vessels of the aerated areas of the lung during ARDS suggests that it should improve 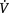 /
/ mismatch and therefore should be helpful during the management of this condition. Unfortunately, even though NO has shown to transiently improve oxygenation in ARDS, it does not seem to improve outcome (Sokol et al 2003).
mismatch and therefore should be helpful during the management of this condition. Unfortunately, even though NO has shown to transiently improve oxygenation in ARDS, it does not seem to improve outcome (Sokol et al 2003).
Surfactant
The use of exogenous surfactant in adults with ARDS has been shown to be feasible and safe; however, it has not proven to be effective in adult ALI. A recent multicentre, randomized, blinded trial of calfactant (extract of natural surfactant from calf lungs) compared with placebo in 153 infants with ALI showed improvement in oxygenation and a decrease in mortality in the patients that had received surfactant (Willson et al 2005).
Other considerations during mechanical ventilation
Position during mechanical ventilation
Changes in position during mechanical ventilation can be used to improve lung volumes, 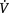 /
/ matching, the clearance of airway secretions or to improve comfort and work of breathing. Changes in position can also be used to avoid or treat pressure lesions. Elevation of the bed head may also help to reduce gastro-oesophageal reflux and pulmonary aspiration (Torres et al 1992).
matching, the clearance of airway secretions or to improve comfort and work of breathing. Changes in position can also be used to avoid or treat pressure lesions. Elevation of the bed head may also help to reduce gastro-oesophageal reflux and pulmonary aspiration (Torres et al 1992).
Humidification
During mechanical ventilation most of the upper airway is bypassed by the endotracheal tube or tracheostomy tube, and the rest of the airway may not be able to supply enough heat and moisture to the delivered gases that are dry and colder than body temperature. Humidification and heating is therefore essential to avoid complications such as hypothermia, inspissation of airway secretions, destruction of airway epithelium and atelectasis. In adults humidifying filters (which operate passively by storing heat and moisture from the patient’s exhaled gas) are frequently used for this purpose; however, in children, active heating of humidified air is necessary. In order to achieve this, a humidifier that operates to increase the heat and water vapour content of the gases is interposed in the inspiratory limb of the ventilator circuit and the temperature is usually set to deliver approximately 37°C to the distal area of the circuit.
Suctioning
Suctioning of the airway to remove secretions is part of the routine care of patients receiving artificial ventilation. However, suctioning can lead to side effects including hypoxaemia, haemodynamic instability, mucosal damage, increase in intracranial pressure and patient discomfort. The use of adequate sedation, reassurance, preoxygenation and a good technique may help to minimize these complications.
Physiotherapy
Physiotherapy is frequently used as an integral part of the management of children receiving mechanical ventilation. However, the place, frequency and techniques of physiotherapy vary widely among units. Frequent techniques used in mechanically ventilated patients are manual hyperinflation, percussion, vibration and positioning, all of which are believed to increase the clearance of secretions (Chapter 10).
Monitoring during mechanical ventilation
Adequate monitoring of the parameters affected by mechanical ventilation is essential. The information helps to characterize the pathophysiology of the underlying condition, to improve ventilation, to enhance patient comfort, and to guide weaning. Blood gas measurement is the gold standard for assessing pulmonary gas exchange, but this technique is limited by the fact that measurements are not continuous, because they require invasive techniques. The use of pulse oximeters and end-tidal carbon dioxide concentration helps to minimize the number of blood gas samples. Transcutaneous carbon dioxide measurement is another way of continuously monitoring the CO2 status.
Monitoring of the respiratory mechanics and analysis of the patient–ventilator interaction should be part of the routine care during mechanical ventilation. Most modern mechanical ventilators provide a graphical display of gas flow, airway pressures and tidal and minute volumes. In small infants and neonates, the information obtained with a pneumotachometer positioned at the end of the endotracheal tube instead of inside the mechanical ventilator can be more reliable. These data, together with measurements of pulmonary gas exchange, help to assess the adequacy of ventilation.
A new technique, electroimpedance tomography, analyses the distribution of ventilation and can be used at the bedside. This technique used 16 electrodes placed around the chest. Injection of an alternating electrical current between sequential pairs of adjacent electrodes and the repeated measurement of the differences in voltage through the array of electrodes permits the detection of changes in impedance. Since air is a poor conductor of electricity compared with other tissues, computer-assisted use of a mathematical algorithm then allows the reconstruction of a spatial image of a section of the chest. This method has the advantage of being non-invasive, radiation free, portable and dynamic. Different studies have shown its usefulness to assess regional lung ventilation (Victorino et al 2004).
Weaning from mechanical ventilation
Stopping mechanical ventilation as soon as the child is ready to be weaned from support is essential to avoid complications. Prolonged mechanical ventilation has been associated with higher risk of nosocomial pneumonia, progressive ventilator-associated lung injury, airway injury, physiological dependence on sedative and narcotic drugs, and even higher mortality. In children who need mechanical ventilation for respiratory failure, the usual approach is to wean the ventilator settings gradually, until the patient is considered to be ready for extubation. However, this gradual weaning process is now being questioned.
A randomized controlled trial comparing weaning protocols versus standard physician-guided weaning in children did not show any impact on the duration of mechanical ventilation. This study also showed that gradual weaning may not be indicated for the majority of children receiving mechanical ventilation due to respiratory failure, and that a large group of children were ready to be extubated when the physicians determined that they were ready to begin a weaning process (Randolph et al 2002). Readiness for extubation can be evaluated using a daily test as soon as the patient meets the criteria for testing (Box 9.5). Different tests for extubation readiness have been proposed. In one paediatric study, patients were extubated if they passed a trial of spontaneous breathing with either pressure support of 10 cmH2O, or T-piece for up to 2 hours. Mechanical ventilation was reinstituted if any of the following signs appeared:
Use of sedation has an impact on the duration of mechanical ventilation. Improved management of sedatives, including the use of objective sedation scores, should be part of the standard care for children receiving mechanical ventilation.
Non-invasive support
Non-invasive ventilation is the delivery of supportive ventilation without the use of an endotracheal airway. NIV is usually delivered as positive pressure ventilation using CPAP or BiPAP; however, negative pressure ventilation by mean of a cuirass is still useful for some conditions. Initially the use of NIV was mostly used in children with neuromuscular disease. Now, it is used in most types of respiratory failure.
In neonates and small infants the most frequent way of delivering non-invasive positive pressure ventilation (NPPV) is by means of nasal CPAP, although modern technology also incorporates the possibility of delivering BiPAP. The most frequent indication is bronchiolitis and other infections with apnoea. In bigger children NPPV is frequently delivered as BiPAP (Fig 9.5), although CPAP can also be used. In this age it is used for different forms of acute hypoxaemic or hypercarbic respiratory failure including asthma, neuromuscular disease, or after extubation in children that have received long periods of mechanical ventilation. The usual way of applying this form of support is via a nasal mask or a facemask. Nasal masks are better tolerated, but many patients with respiratory distress are mouth breathers and therefore a facemask might be necessary (Fig. 9.6). In order to tolerate NPPV some sedation is needed in the young infant. In older children, explanation, reassurance and familiarity with the therapy should suffice. To increase the tolerance, NPPV can be started at low-pressure settings that are then increased progressively until the target pressures are obtained. Nasogastric tubes can be used when the accumulation of air in the stomach is a concern, especially in children with facemasks. However, many clinicians prefer not to use nasogastric tubes as their placement with tapes can reduce the tightness of the seal between the mask and the face.
A review of clinical studies using NPPV has shown that improvement usually occurs after the first 3 hours of therapy (Akingbola & Hopkins 2001). NPPV is not recommended in ARDS (Essouri et al 2006). The contraindications for NPPV include respiratory arrest, inability to use the mask because of trauma or surgery, difficult secretion management, haemodynamic instability, altered mental state, risk of aspiration, intolerance to the therapy and life-threatening refractory hypoxaemia or hypercarbia.
Finally, one technique that has historically been used effectively in children with neuromuscular disease since the poliomyelitis epidemics in the 1950s is extrathoracic negative pressure ventilation with a cuirass (Lassen 1953). In the past decade there has been renewed interest in this form of ventilation (Meessen et al 1994). There are many physiological reasons why negative pressure support should be beneficial, such as its ability to increase tonic activity in the diaphragm and intercostal muscles. In children with neuromuscular disease, who are on positive pressure mechanical support, it has been found that, when it comes to weaning, extubation and the introduction of negative pressure support means that analgesia and sedation can be discontinued quite quickly (Chisakuta & Tasker 1998). This approach should limit the unavoidable iatrogenic worsening of respiratory drive that results from the co-administration of analgesia and sedation (which is invariably necessary for children in order that they may tolerate the endotracheal tube). In myasthenic patients the time course of mechanical ventilatory support can be more than halved using this technique. Regarding bronchiolitis-related apnoeas, a retrospective analysis comparing two centres, one using negative pressure ventilation and the other using conventional therapies, concluded that the use of NPV was associated with a reduced rate of endotracheal intubation, and shorter paediatric intensive care unit (PICU) stay (Al-balkhi et al 2005). As more portable and easier-to-use ventilators become available, this form of respiratory support may be used more frequently.
Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. New England Journal of Medicine. 2000;342(18):1301-1308.
Akingbola OA, Hopkins RL. Pediatric noninvasive positive pressure ventilation. Pediatric Critical Care Medicine. 2001;2(2):164-169.
Al-balkhi A, Klonin H, Marinaki K, et al. Review of treatment of bronchiolitis related apnoea in two centres. Archives of Disease in Childhood. 2005;90(3):288-291.
Arnold JH, Hanson JH, Toro-Figuero LO, et al. Prospective, randomized comparison of high-frequency oscillatory ventilation and conventional mechanical ventilation in pediatric respiratory failure. Critical Care Medicine. 1994;22(10):1530-1539.
Bartlett RH. Extracorporeal life support for cardiopulmonary failure. Current Problems in Surgery. 1990;27(10):621-705.
Bartlett RH, Andrews AF, Toomasian JM, et al. Extracorporeal membrane oxygenation for newborn respiratory failure: forty-five cases. Surgery. 1982;92(2):425-433.
Bernard GR, Artigas A, Brigham KL, et al. The American-European consensus conference on ARDS: definitions, mechanisms, relevant outcomes and clinical trial coordination. American Journal of Respiratory and Critical Care Medicine. 1994;149(3 Pt 1):818-824.
Chisakuta A, Tasker RC. Respiratory failure in myasthenia gravis and negative pressure support. Pediatric Neurology. 1998;19(3):225-226.
Curley MA, Hibberd PL, Fineman LD, et al. Effect of prone positioning on clinical outcomes in children with acute lung injury: a randomized controlled trial. Journal of the American Medical Association. 2005;294(2):229-237.
Essouri S, Chevret L, Durand P, et al. Non-invasive positive pressure ventilation: five years of experience in a pediatric intensive care unit. Pediatric Critical Care Medicine. 2006;7(4):329-334.
Farias JA, Alias I, Retta A, et al. An evaluation of extubation failure predictors in mechanically ventilated infants and children. Intensive Care Medicine. 2002;28(6):752-757.
Goldman AP, Kerr SJ, Butt W, et al. Extracorporeal support for intractable cardiorespiratory failure due to meningococcal disease. Lancet. 1997;349(9050):466-469.
Kneyber MC, Plötz FB, Sibarani-Ponsen RD, Markhorst DG. High-frequency oscillatory ventilation (HFOV) facilitates CO2 elimination in small airway disease: the open airway concept. Respiratory Medicine. 2005;99(11):1459-1461.
Lassen HC. A preliminary report on the 1952 epidemic of poliomyelitis in Copenhagen with special reference to the treatment of acute respiratory insufficiency. Lancet. 1953;1(1):37-41.
Meessen NE, van der Grinten CP, Luijendijk SC, et al. Continuous negative airway pressure increases tonic activity in diaphragm and intercostal muscles in humans. Journal of Applied Physiology. 1994;77(3):1256-1262.
Pearson GA, Grant J, Field D, et al. Extracorporeal life support in paediatrics. Archives of Disease in Childhood. 1993;68(1):94-96.
Pelosi P, Brazzi L, Gattinoni L. Prone position in acute respiratory distress syndrome. European Respiratory Journal. 2002;20(4):1017-1028.
Peters MJ, Tasker RC, Kiff KM, Yates R, Hatch DJ. Acute hypoxemic respiratory failure in children: case mix and the utility of respiratory severity indices. Intensive Care Medicine. 1998;24(7):699-705.
Pettignano R, Fortenberry JD, Heard ML, et al. Primary use of the venovenous approach for extracorporeal membrane oxygenation in pediatric acute respiratory failure. Pediatric Critical Care Medicine. 2003;4(3):291-298.
Randolph AG, Meert KL, O’Neil ME, et al. (Pediatric Acute Injury and Sepsis Investigators Network). The feasibility of conducting clinical trials in infants and children with acute respiratory failure. American Journal of Respiratory and Critical Care Medicine. 2003;167(10):1334-1340.
Randolph AG, Wypij D, Venkataraman ST, et al. (Pediatric Acute Lung Injury and Sepsis Investigators (PALISI) Network). Effect of mechanical ventilator weaning protocols on respiratory outcomes in infants and children: a randomized controlled trial. Journal of the American Medical Association. 2002;288(20):2561-2568.
Slee-Wijffels FYAM, van der Vaart KRM, Twisk JWR, et al. High-frequency ventilation in children: a single-center experience of 53 cases. Critical Care. 2005;9(3):R274-279.
Sokol J, Jacobs SE, Bohn D, 2003 Inhaled nitric oxide for acute hypoxemic respiratory failure in children and adults. Cochrane Database of Systematic Reviews Issue 1. Art. No.: CD002787. DOI: 10.1002/14651858.CD002787
Tasker RC. Gender differences and critical medical illness. Acta Paediatrica. 2000;89(5):621-623.
Tasker RC, Gordon I, Kiff K. Time course of severe respiratory syncytial virus infection in mechanically ventilated infants. Acta Paediatrica. 2000;89(8):938-941.
Torres A, Serra-Batlles J, Ros E, et al. Pulmonary aspiration of gastric contents in patients receiving mechanical ventilation: the effect of body position. Annals of Internal Medicine. 1992;116(7):540-543.
Victorino JA, Borges JB, Okamoto VN, et al. Imbalances in regional lung ventilation: a validation study on electrical impedance tomography. American Journal of Respiratory and Critical Care Medicine. 2004;169(7):777-778.
Watkins SJ, Peters MJ, Tasker RC. One hundred courses of high frequency oscillatory ventilation: what have we learned? European Journal of Pediatrics. 2000;159(1-2):134.
Willson DF, Thomas NJ, Markovitz BP, et alPediatric Acute Lung Injury and Sepsis Investigators. Effect of exogenous surfactant (calfactant) in pediatric acute lung injury: a randomized controlled trial. Journal of the American Medical Association. 2005;293(4):470-476.