Chapter 10 Paediatrics
INTRODUCTION
The respiratory system in children differs significantly from adults, both anatomically and physiologically. These differences have important consequences for the physiotherapy care of children in terms of respiratory assessment, treatment and choice of techniques.
The principal reason for hospital admissions in children aged 0–4 years is respiratory illness and the management of children with acute or chronic respiratory disorders has become a specialized area of respiratory physiotherapy. The inexperienced physiotherapist working with children will require the support and mentorship of an experienced paediatric physiotherapist in order to develop the necessary skills.
Assessment and treatment of children requires skillful age-appropriate communication with the child, the family and within the multidisciplinary team. It is essential to include parents, relatives and carers as part of the care team and children and their parents should have a full explanation of why treatment is required and what it involves. Treating children can be difficult and challenging and these sessions are easier when children are cooperative and compliant. Cooperation can often be obtained by persuasion, distraction with games, television, cassette tapes or reading books suited to the child’s age and interest. It may be helpful in some situations to reward good behaviour or bravery, but occasionally children do refuse treatment. In these cases, if the benefits of treatment are considered to outweigh the risks, treatment must be given after thorough and careful explanation to the child and their carers.
Parents are able to refuse physiotherapy treatment for their child but this rarely occurs in practice. Parents of sick children often feel extremely vulnerable and anxious. Therapists should ensure their communication is always professional, empathetic and understanding. Parental stress may manifest in different ways, including apparent lack of concern or anger. Some parents may need special help to cope with their feelings of fear and anxiety and the regular contact between the physiotherapist and family is often an important source of support.
Children’s awareness of the implications of chronic illness and treatment develop as they grow older and they should be encouraged to take on more responsibility for their treatment. Teenagers, particularly, have a more sophisticated understanding and may be beginning to think about the future and the impact of illness on school, social life and body image.
DEVELOPMENT OF THE LUNGS
The development of the lung can be divided into four stages (Inselman & Mellins 1981):
Embryonic period (weeks 3–5).
The lung bud starts as an endodermal outgrowth of fetal foregut. The single tube thus formed soon branches into two, forming the major bronchi. By cell division, the process of growth continues until, at the end of this period, the major lung branches are formed.
Pseudoglandular period (weeks 6–16).
During this period the airways grow by dichotomous branching so that by week 16 all generations of the airway from trachea to terminal bronchioles (i.e. the preacinus) are formed. During this period the pulmonary circulation also develops, cartilage and lymphatic formation occur and cilia appear (week 10 onwards) (Langman 1977).
Canalicular period (weeks 17–24).
The respiratory bronchioles, alveolar ducts and alveoli (i.e. the acinus) start to develop during this time, simultaneously with the lung capillaries, thus preparing the lungs for their future role in gas exchange (Hislop & Reid 1974). The air–blood barrier first appears at week 19 and towards the end of this period surfactant synthesis begins.
Terminal sac period (week 24–term).
Development of the pulmonary circulation continues and the respiratory bronchioles subdivide to form air spaces. Two different cell types (types I and II pneumocytes) line the air spaces. Type I pneumocytes flatten and elongate to cover the majority of the surface area of the saccular air spaces. Type II cells only occupy approximately 2% of the surface and are responsible for surfactant synthesis and storage (Greenough 1996). Surfactant is a phospholipid, which stabilizes surface tension in the alveolus and prevents alveolar collapse on expiration. Small quantities of surfactant are present at weeks 23–24 of gestation and the amount present gradually increases until a surge at about week 30. Birth itself and the onset of respiration stimulate surfactant production.
Towards the end of the terminal sac period, the air spaces have developed into primitive multilocular alveoli. After birth, alveoli increase in size and number. The average number of alveoli in the newborn is 150 million. By the age of 3–4 years, the adult number of 300–400 million alveoli has been reached, but alveolar growth continues for the first 7 years (Hislop et al 1986). More recent estimations of mean alveolar number in adulthood have been 480 million (range 274–790 million), with alveolar number closely related to lung volume (Ochs 2004).
RESPIRATORY SYSTEM: ANATOMICAL AND PHYSIOLOGICAL DIFFERENCES BETWEEN CHILDREN AND ADULTS
The respiratory anatomy and physiology of infants and children is very different from that of adults. The principles of adult cardiorespiratory physiotherapy management cannot be transposed directly to an infant with pulmonary pathology.
Anatomical differences
Rib cage and chest shape
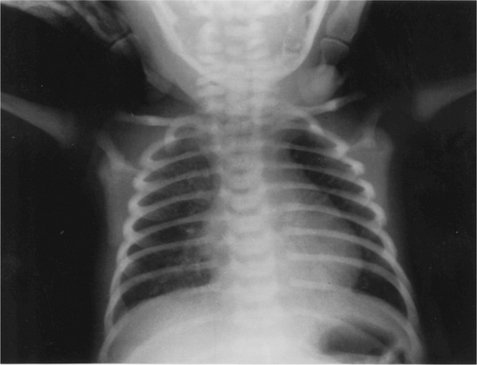
The cross-sectional shape of the infant thorax is cylindrical and not elliptical as in adolescents or adults. The ribs of the newborn infant are relatively soft and cartilaginous compared with the more rigid chest wall of older children and adults. They are also placed horizontally in relation to the sternum and vertebral column compared with the more oblique rib angle of adults (Fig. 10.1). The bucket handle rib movement seen in older children and adults is therefore not possible. As the infant grows, and begins to develop an upright posture, the ribs develop a more oblique angle and the transverse diameter of the rib cage increases. The adult chest shape is achieved by 3 years of age (Openshaw et al 1984).
The intercostal muscles are poorly developed in infancy and contraction of the intercostal muscles is inefficient at improving thoracic volumes either by increasing the anteroposterior or transverse diameters of the chest. Increased ventilatory requirements have to be met by increasing the respiratory rate rather than depth (Konno & Mead 1967).
Diaphragm
The angle of insertion of the infant diaphragm is horizontal compared with older children or adults, placing it at a mechanical disadvantage. The infant diaphragm has a lower relative muscle mass and a lower content of high-endurance muscle fibres, and thus is much more vulnerable to fatigue.
Maximal diaphragmatic activity during severe respiratory distress or respiratory obstruction leads to an inward movement of the lower rib cage instead of a downward movement of the diaphragm, as well as intercostal and sternal recession (Muller & Bryan 1979). Despite these disadvantages, the diaphragm is the main muscle of inspiration in the infant, since the intercostals are poorly developed. Ventilation in the infant is also more affected by impaired diaphragmatic function, for example by abdominal distension, hepatomegaly or phrenic nerve damage.
Preferential nasal breathing
The shape and orientation of head and neck in babies (large head, prominent occiput, short neck, large tongue, smaller retracted lower jaw, high larynx) mean that the airway is prone to obstruction in young infants. Young infants up to about 6 months of age are preferential nasal breathers and studies suggest that up to half of all neonates are unable to breathe through their mouths, except when crying, for the first few weeks of life (King & Booker 2004) The small nasal passages account for between 30% and 50% of the total airway resistance in neonates. The narrowest portion of the nasal airway has a cross-sectional area of about 20 mm2. Therefore, even a small amount of swelling or obstruction of the nasal passages of infants compromises breathing considerably and causes a disproportionate and detrimental effect on the work of breathing. Some young infants with upper respiratory tract infections and partial obstruction of their nasal passages can develop respiratory distress.
Position of larynx
In the newborn infant, the larynx and hyoid cartilage are higher in the neck and closer to the base of the epiglottis, being at the level of C3 in a premature infant and C4 in a child compared with C5–6 in the adult. The larynx descends with age, but its high position enables the infant to feed and breathe simultaneously for approximately the first 4 months of age.
This high position also provides some protection of the airway in infants younger than 4–6 months because it acts as a valve, which helps keep food in the mouth until the pharyngeal swallow is initiated. The airway has less anatomical protection as the larynx assumes its lower position in the neck and is not as directly protected by the epiglottis. Then, poor closure of the airway or partial paralysis of the vocal folds may become more evident and coughing, choking or aspiration may occur.
Airway diameter
The neonatal trachea is short (4–9 cm) and directed downward and posteriorly. The diameter of the trachea in the newborn is 4–5 mm and the diameter of an infant trachea is only about one-third that of an adult. This makes respiratory resistance higher and the work of breathing greater. Since the resistance to airflow through a tube is directly related to the tube length and inversely related to the fourth power of the radius of the tube, halving the radius of the trachea will increase its resistance (reduce flow) 16 times. Tracheal swelling as a result of endotracheal intubation or suction can therefore dramatically increase resistance to breathing. These factors give the lungs less reserve, so that a well-oxygenated infant with upper airway obstruction can become cyanotic in a matter of seconds.
In contrast to adolescents and adults, the narrowest part of the infant’s airway is not the vocal cords, but the cricoid ring. Thus an uncuffed endotracheal tube provides a larger internal diameter compared with a cuffed tube and in children will successfully seal against in the circular subglottic ring. However, the inflexible cricoid ring also leaves children more vulnerable to mucosal oedema and post-extubation stridor. The right main bronchus is less angled than the left, making right mainstem intubation more likely.
At birth there is no further increase in the number of airways formed but there is growth and development in their size. In the first few years of life there is a significant increase in the diameter of the larger, more proximal airways (Hislop & Reid 1974). The smaller, more distal airways do not increase in diameter until nearer 5 years of age. This higher peripheral airways resistance is exacerbated by respiratory infections, which cause inflammation of the airways, for example in bronchiolitis, or in the presence of secretions.
Bronchial walls
The bronchial walls are supported by cartilage, which begins to develop from 12 weeks’ gestation and continues throughout childhood. The cartilaginous support of an infant’s airways is much less than that of an adult, and predisposes the airways to collapse. The bronchial walls contain proportionally more cartilage, connective tissue and mucous glands than do those of adults, but less smooth muscle; this makes the lung tissue less compliant. The lack of bronchial smooth muscle, particularly in the smaller bronchioles, may be one reason for the lack of response to bronchodilators under the age of 12 months. The β-receptors in infants are also immature, which further reduces any response to β-adrenergic bronchodilator therapy (Reid 1984). The high proportion of mucous glands in the major bronchi of infants makes the airways more susceptible to mucus obstruction.
Alveoli and surfactant
The respiratory system is not fully developed at birth, even in the term neonate, and postnatal maturation continues for a significant time. Although by 20–27 weeks’ gestation lung acinar have formed, several types of epithelial cells can be differentiated, and the air–blood barrier is thin enough to support gas exchange; true alveoli develop only after about 36 weeks’ gestation. A term newborn has an average of 150 million alveoli. The remainder of the eventual average of 400 million alveoli develop after birth, the vast majority within the first 2 years of life. Both the number and size of alveoli continue to increase postnatally until the chest wall stops growing. By 4 years of age, the adult number of 300 million may exist, although growth can continue until 7 years of age. The smaller alveolar size of an infant makes the infant more susceptible to alveolar collapse, and the smaller number of alveoli reduces the area available for gaseous exchange (Reid 1984).
Pulmonary surfactant is a mixture of phospholipids (90%) and apoproteins (10%), which act to reduce surface tension at the air–liquid interface in the alveolus, thereby preventing collapse of lung parenchyma at the end of expiration. Type II alveolar cells synthesize and secrete surfactant from 23 to 24 weeks’ gestation. In preterm newborns, a deficiency of surfactant is a major factor in the development of neonatal respiratory distress syndrome (RDS). Male gender is a risk factor for neonatal RDS, bronchopulmonary dysplasia (BPD) and mortality. Boys with neonatal RDS seem to have more health problems than girls during the neonatal period.
Collateral ventilation
Collateral ventilation is the means by which a distal lung unit can be ventilated, despite blockage of its main airway. Collateral ventilatory pathways are achieved by a network of interconnecting pathways linking different structures. Respiratory bronchioles are linked by channels of Martin. Canals of Lambert connect respiratory and terminal bronchioles with alveoli and their ducts; and adjacent alveoli are joined by openings in the alveolar wall, called pores of Kohn (Menkes & Traystman 1977). However, none of these pathways exists at birth. The pores of Kohn develop between years 1 and 2, and the canals of Lambert do not appear until about 6 years of age. The collateral ventilatory channels between alveoli, respiratory bronchioles and terminal bronchioles are poorly developed until 2 and 3 years of age, predisposing towards alveolar collapse.
Internal organs and lymphatic tissue
The lymphatic tissue (adenoids and tonsils) may be enlarged in the infant and the tongue is also relatively large. These factors may contribute to upper airway obstruction. The heart and other organs are also relatively large in infants, leaving less space for lung expansion. The heart can occupy up to half the transverse diameter of the chest in chest radiographs.
Height and exposure to air pollution
Because children breathe more rapidly compared with adults and because they spend more time outdoors being physically active, they tend to be more exposed to outdoor air pollution and allergens than do adults and have greater deposition of particulate matter. Their reduced height means they are also more exposed to vehicle exhausts and heavier pollutants that concentrate at lower levels in the air. There is substantial evidence linking air pollution with respiratory health problems and children are more vulnerable (Brauer et al 2007, Pénard-Morand et al 2005).
Physiological differences
Respiratory compliance
Respiratory compliance is a measure of the pressure required to increase the volume of air in the lungs and reflects a combination of lung and chest wall compliance. The lung compliance of a child is comparable to that of an adult, being directly proportional to the child’s size. However, compliance is reduced in the infant because of the high proportion of cartilage in the airways. The premature infant, who lacks surfactant, demonstrates a further significant decrease in compliance. The chest wall of an infant is cartilaginous and therefore very soft and compliant in comparison with the more calcified and rigid adult structure. The intercostal muscles are also less well equipped to stabilize the rib cage during diaphragmatic contraction. Neonates therefore have an imbalance between a relatively low outward recoil of their chest wall and normal inward elastic recoil, which means that they are prone to airway collapse. An awake, spontaneously breathing neonate will maintain its functional residual capacity (FRC) by active measures including laryngeal braking, the initiation of inspiration before the end of passive expiration (intrinsic PEEP) and persistent inspiratory muscle activity throughout the respiratory cycle. These active mechanisms are lost during anaesthesia and result in a fall in FRC, airway closure, atelectasis and ventilation/perfusion mismatch.
Closing volume
The closing volume is the lung volume at which closure of the small airways occurs. This volume plus the residual volume (the volume of gas left in the lungs following maximum expiration) is known as the closing capacity (CC). In the adult, CC is less than FRC, i.e. the volume of gas left in the lungs following tidal expiration, whereas in the infant it is greater than FRC. The higher closing volumes apparent in infants are due to greater chest wall compliance and reduced elastic recoil of the lungs than in the adult. Therefore, airway closure may occur before the end of expiration, e.g. during expiratory chest vibrations, putting the infant at a much greater risk of developing widespread atelectasis, especially in the presence of lung disease, where lung volume is further reduced. In the event of respiratory distress, the infant grunts on expiration, adducting the vocal cords in an attempt to reduce the amount of gas expired, thus maintaining a higher FRC and minimizing alveolar collapse (Pang & Mellins 1975). Re-inflation of alveoli, once collapsed, is more difficult in the infant, who has to work considerably harder to overcome the effects of the compliant chest wall.
Ventilation and perfusion
In the adult, both ventilation and perfusion are preferentially distributed to the dependent lung. The best gas exchange and ventilation/perfusion match will therefore be in the dependent region of the lung (Zack et al 1974). In the infant, however, ventilation is preferentially distributed to the uppermost lung (Davies et al 1985), whereas the perfusion remains best in the dependent regions. This leads to greater gas exchange in the uppermost lung (Heaf et al 1983) but an imbalance between ventilation and perfusion (Bhuyan et al 1989). In acutely ill children with unilateral lung disease, oxygenation may be optimized by placing the ‘good’ lung uppermost. However, this is contrary to the goal of improving ventilation to the diseased lung and facilitating secretion clearance, in which positioning and postural drainage would require the diseased lung to be uppermost. The therapist would have to balance their decision based on the stability, tolerance and current therapeutic priorities.
The difference in ventilation distribution between infants and adults is most likely due to the more compliant rib cage of the infant, which compresses the dependent areas of lung. In addition, while in the adult the weight of the abdominal contents provides a preferential load on the dependent diaphragm and therefore improves its contractility, in the infant this does not happen. The effect on both hemidiaphragms is similar, due to the abdomen being so much smaller and narrower (Davies et al 1985). It has been shown in adults that, when the diaphragm is inactivated, e.g. when ventilated under anaesthetic, the ventilation distribution changes to that of an infant (Rehder et al 1972). It is not yet known exactly when the ventilation distribution in the infant changes to that of an adult, but it may be as late as 10 years of age.
Oxygen consumption, cardiac output and response to hypoxia
Infants have a higher resting metabolic rate than adults and consequently have a higher oxygen requirement. Children have a higher cardiac output and oxygen consumption per kilogram than adults; in infants this may exceed 6 ml/kg/min, twice that of adults. They support this higher output with a higher baseline heart rate but lower blood pressure than adults.
Neonatal myocardium has a large supply of mitochondria, nuclei and endoplasmic reticulum to support cell growth and protein synthesis, but these are non-contractile tissues, which render the myocardium stiff and non-compliant. This may impair filling of the left ventricle and limit the ability to increase the cardiac output by increasing stroke volume (Frank Starling mechanism). Stroke volume in infants is therefore relatively fixed and the only way of increasing cardiac output is by increasing heart rate.
The sympathetic nervous system is not well developed predisposing the neonatal heart to bradycardia. An infant responds to hypoxia with bradycardia and pulmonary vasoconstriction, whereas the adult becomes tachycardic with systemic vasodilation. The bradycardic response in infants is probably due to myocardial hypoxia and acidosis, but leads to an immediate reduction in cardiac output and the development of further hypoxia.
Although anatomical closure of the foramen ovale can occur as early as 3 months of age, the channel remains ‘probe patent’ in 50% of children up to 5 years of age, and persists in about 30% of adults. Similarly, anatomical closure of the ductus arteriosus usually occurs between 4 and 8 weeks of age. Any stimulus, such as hypoxia or acidosis, that causes an increase in pulmonary vascular resistance during the neonatal period may allow these two potential channels to reopen, resulting in right-to-left shunting and increasing hypoxia (King & Booker 2004).
Muscle fatigue
The respiratory muscles of infants tire more quickly than those of adults due to a much smaller proportion of fatigue-resistant muscle fibre (Keens & Ianuzzo 1979). There are two main muscle fibre types, type I and type II. Type I muscle fibres are slow twitch, high oxidative and slow to fatigue. Type II fibres are fast twitch, slow oxidative and tire quickly. Of the muscle fibres in the adult diaphragm, 55% are type I compared with only 30% in the infant. Premature infants tire even more easily as, at 24 weeks’ gestation, only 10% of their muscle fibres are fatigue resistant (Muller & Bryan 1979). Excessive muscle fatigue results in apnoea. By 12 months of age the number of type I fibres equals that of an adult.
Breathing pattern and rapid eye movement sleep
Irregular breathing patterns and episodes of apnoea are relatively common in neonates, especially if premature, and are related to immature cardiorespiratory control. Short spells of apnoea can be considered normal in these circumstances, but need careful monitoring as they may reflect hypoxic conditions.
During rapid eye movement (REM) sleep there is a reduction in postural tone and tonic inhibition of the infant’s intercostal muscles such that the rib cage is even less well equipped to counteract the contraction of the diaphragm during inspiration (Muller & Bryan 1979). This reduces the efficiency of respiration, causes a drop in functional residual capacity and increases the work of breathing, predisposing the infant to apnoeic episodes (Muller & Bryan 1979). The premature infant is most at risk, spending up to 20 hours a day asleep, 80% of which may be in active REM sleep compared with 20% in adult sleep.
Response to cold
Paediatric patients have an increased surface area per kilogram and lose heat to the environment more readily than adults. This is compounded by cold intravenous fluids, dry anaesthetic gases and exposure. Non-shivering thermogenesis in brown adipose tissue is the major mechanism of heat production during the first few months of life. Brown fat is specialized tissue located in the posterior of the neck, along the interscapular and vertebral areas, and surrounding the kidneys and adrenal glands. Metabolic heat production can increase up to two and a half times during cold stress. Shivering is a less economical form of heat production but does occur in severely hypothermic neonates. Hypothermia is a serious problem that can result in increased oxygen consumption, cardiac irritability and respiratory depression (King & Booker 2004).
RESPIRATORY ASSESSMENT OF THE INFANT AND CHILD
Careful assessment is essential to identify problems requiring physiotherapy intervention. Many aspects of assessment will be the same as in adults (Chapter 1), but specific differences are listed below.
Medical notes
Information can be extracted from the medical notes relating to present and past medical history. When assessing a neonate, history of pregnancy, labour and delivery are relevant as well as gestational age and weight. In addition, the Apgar score at birth should be noted. This score relates to heart rate, respiratory effort, muscle tone, reflex irritability and colour and gives an indication of the degree of asphyxiation suffered by the infant at birth.
Discussion with the relevant carers
Discussion with medical staff, nursing staff and the parent/carer is essential to obtain correct information about recent changes. In chronically ill children who require home physiotherapy, liaison with the primary healthcare team is essential.
When assessing the hospitalized child, information should be obtained about:
Observation charts and investigations
Pyrexia may indicate a possible respiratory infection. The core-to-peripheral temperature gradient should be noted, particularly in the critically ill patient as it is a reflection of peripheral vasoconstriction which can occur as a response to cold, hypovolaemia, sepsis or low cardiac output.
Tachycardia may be due to sepsis or shock. It may also be caused by inadequate levels of sedation or analgesia. In preterm infants, bradycardias may be due to many causes, including retention of secretions.
Apnoeic spells in the infant may indicate respiratory distress, sepsis or presence of secretions in the upper or lower respiratory tract.
The trend of arterial gases and their relationship to oxygen saturation and transcutaneous oxygen should be noted, together with the degree and type of respiratory support.
Results of investigations and other relevant observations should be referred to as appropriate.
Examination
Examination of the older child is similar to that of the adult (Chapter 1). The following specific factors should be considered in younger children.
Clinical signs
Clinical signs of respiratory distress are listed in Box 10.1.
Recession occurs when high negative intrathoracic pressure during inspiration pulls the soft, compliant chest wall inward. It may be sternal, subcostal or intercostal. Mild recession may be normal in preterm infants but in older infants is a sign of increased respiratory effort.
Nasal flaring is a dilatation of the nostrils by the dilatores naris muscles and is a sign of respiratory distress in the infant. It may be a primitive response attempting to decrease airway resistance.
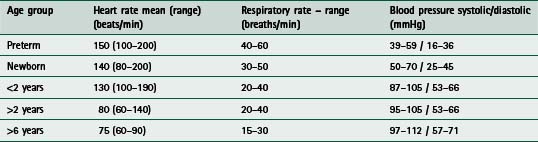
Tachypnoea (respiratory rate greater than 60 breaths/min) may indicate respiratory distress in infants. Normal values are listed in Table 10.1.
Grunting occurs when an infant expires against a partially closed glottis. This is an automatic response which increases functional residual capacity in an attempt to improve ventilation.
Stridor is heard in the presence of a narrowing of the upper trachea and/or larynx. This may be due to collapse of the floppy tracheal wall, inflammation or an inhaled foreign body. It is most commonly heard during inspiration, but in cases of severe narrowing it may be heard during both inspiration and expiration.
Cyanosis refers to the bluish colour of the skin and mucous membranes caused by hypoxaemia. In infants and young children it is an unreliable sign of respiratory distress as it depends on the relative amount and type of haemoglobin in the blood and the adequacy of the peripheral circulation. For the first 3–4 weeks of life, the newborn infant has an increased amount of fetal haemoglobin, which has a higher affinity for oxygen than adult haemoglobin. The result is a shift of the oxygen saturation curve to the left in infants.
Auscultation of the infant and young child is sometimes complicated by the easy transmission of sounds. In the infant who is ventilated, referred sounds such as water in the ventilator tubing may be transmitted to the chest. In the older child, secretions in the nose or throat may lead to referred sounds in both lung fields. Wheezing in the younger child or infant may be due to bronchospasm, but could also be due to retained secretions partially occluding smaller airways. It is sometimes very difficult to hear breath sounds in the spontaneously breathing preterm infant.
Cardiac manifestations of respiratory distress include an initial tachycardia and possible increase in systemic blood pressure. This changes with worsening hypoxia to bradycardia and hypotension.
Neck extension in an infant with respiratory distress may represent an attempt to reduce airway resistance.
Head bobbing occurs when infants attempt to use the sternocleidomastoid and the scalene muscles as accessory muscles of respiration. It is seen because the relatively weak neck extensors of infants are unable to stabilize the head.
Pallor is commonly seen in infants with respiratory distress and may be a sign of hypoxaemia or other problems, including anaemia.
Reluctance to feed is often associated with respiratory distress and infants may need to take frequent pauses from sucking when tachypnoeic.
Alterations in levels of consciousness should be noted. A reduction in activity may be due to neurological deficit or as a result of opiate analgesia but may also be due to hypoxia. It may be accompanied by an inability to feed or cry. Irritability and restlessness may also be indicative of a hypoxic state.
Other relevant observations
The behaviour of a child can often give important clues about their respiratory status. Agitation or irritability may be a sign of hypoxia, while the child in severe respiratory distress may be withdrawn and lie completely still.
It is important to note muscle tone in the infant or child with respiratory distress. A hypotonic child may have increased difficulty with breathing, coughing and expectorating, while hypertonia may also be associated with difficulty in clearing secretions.
Abdominal distension can cause or exacerbate respiratory distress, because the diaphragm is placed at a mechanical disadvantage. In infants this is of greater concern as the diaghragm is the primary muscle of respiration.
PHYSIOTHERAPY TECHNIQUES IN INFANTS AND CHILDREN
Most physiotherapy techniques used in adults can be applied in children and the same contraindications apply (Chapters 5 & 6). Treatment should never be performed routinely as it may have potentially detrimental effects (Horiuchi et al 1997, Krause & Hoehn 2000, Stiller 2000). Ideally treatment should occur before feeds or adequate time allowed following a feed to avoid problems associated with vomiting and aspiration.
Chest percussion
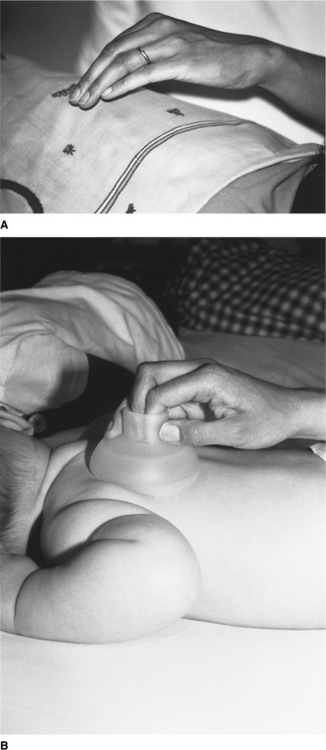
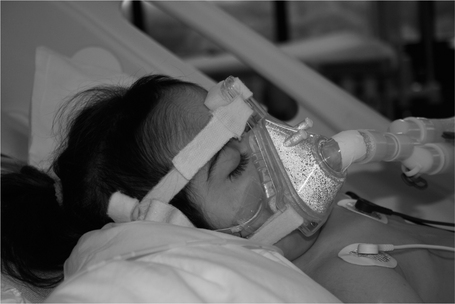
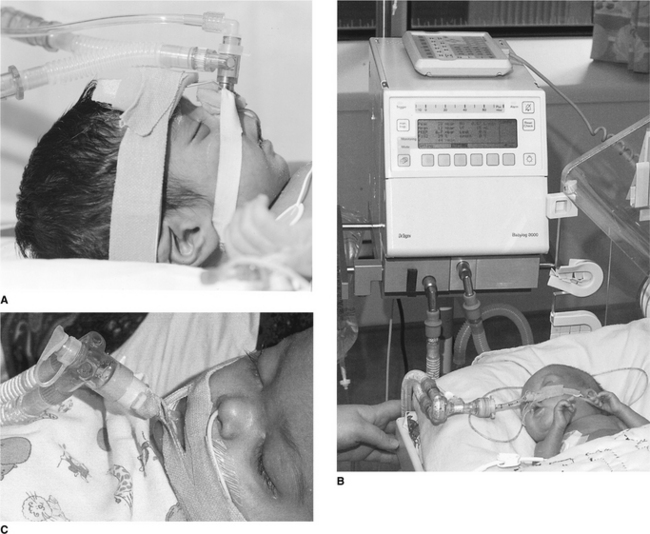
Chest percussion (sometimes referred to as chest clapping) using the hand, fingers or a facemask is generally well tolerated and widely used in children. Percussion with one hand is used in small children and babies (Fig. 10.2A). In neonates and preterm infants ‘tenting’ (using the first three or four fingers of one hand with slight elevation of the middle finger) or the use of a soft plastic cup-shaped object such as a facemask may be more appropriate (Fig. 10.2B) (Tudehope & Bagley 1980).
Vibrations and shaking
Chest wall vibrations involve the application of a rapid extrathoracic compressive force at the beginning of expiration, followed by oscillatory compressions until expiration is complete. The compressions and oscillations applied during chest wall vibrations are believed to aid secretion clearance via a number of physiological mechanisms, including increasing peak expiratory flow to move secretions towards the large airways for removal by suction or cough (Kim et al 1987, King 1998, McCarren et al 2006, Ntoumenopoulos 2005, van der Schans et al 1999, Wanner 1984).
Chest wall vibrations remain objectively undefined and may vary considerably between practitioners and units. The terms chest vibrations, compressions, shaking and expiratory flow increase techniques have been used variously in the literature (Almeida et al 2005, Sutton et al 1985, Wong et al 2003).
Chest wall vibrations appear to be used more frequently in ventilated children than percussion, probably because the glottis is held open by the endotracheal tube, facilitating rapid expiratory flow during vibrations that improve mucus clearance. There is a strong linear relationship between the maximum force applied during chest wall vibrations and the age of the child, most likely reflecting modification of techniques to accommodate changes in chest wall compliance (Gregson et al 2007a). Maximum force applied during physiotherapy can vary substantially between physiotherapists. Similarly there is marked variability in the pattern of force–time profiles between physiotherapists with respect to the duration of vibration, and amplitude, number and frequency of oscillations. Figure 10.3 illustrates the style of force profiles delivered to four infants, all aged between 5 and 14 months by four different physiotherapists. However, there is remarkable consistency within and between each physiotherapist’s treatment sessions (Gregson et al 2007b). The clinical consequences for such variation in treatment profiles remain unclear.
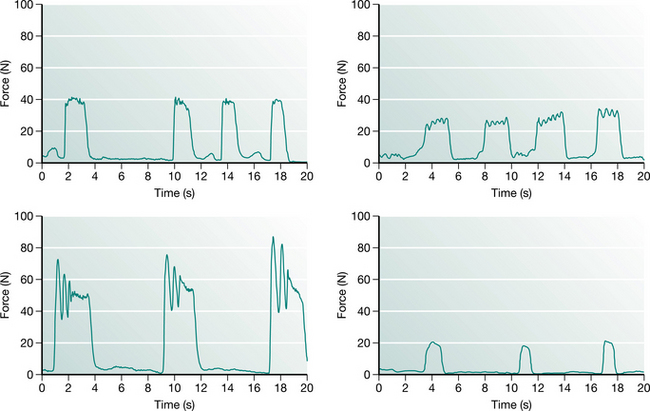
Figure 10.3 Force–time profiles of chest wall vibrations delivered by four different physiotherapists to four infants (5–14 months). The patterns are repeatable within each treatment but vary considerably between therapists with respect to magnitude and duration of vibration, and amplitude, number and frequency of oscillations.
In children who are not intubated, vibrations can be applied effectively when reflex glottic closure does not occur and when the respiratory rate is normal or near normal (30–40 breaths/min). If infants are breathing very rapidly, the expiratory phase is so short that vibrations are more difficult to perform.
Precautions for chest percussion and vibratory techniques
In children with dietary deficiencies, liver disease, bone mineral deficiency (e.g. rickets) or coagulopathies, manual techniques should be applied with caution. Manual techniques may not be appropriate in extremely premature infants and specific issues related to this group of patients are discussed later.
Chest percussion has been reported to cause an increase in bronchospasm in adults with chronic lung disease (Campbell et al 1975, Wollmer et al 1985). Premedication with bronchodilator therapy may reduce this effect but in severe cases percussion should be avoided.
Postural drainage (gravity-assisted positioning)
The use of gravity-assisted positioning, including a head-down tip, has traditionally been a component of airway clearance in babies and children. However, the use of the head-down tipped position has been the focus of considerable debate in recent years. Very few studies have examined specifically the efficacy of gravity-assisted positioning in infants and children. An Australian study of 20 babies with cystic fibrosis reported an increase in gastro-oesophageal reflux in those receiving postural drainage (PD) using a head-down tipped position compared with modified PD without a head-down tilt (Button et al 1997). Another study also undertaken in babies with cystic fibrosis (CF) (Phillips 1996) reported no adverse effect of the head-down tipped position on gastroesophageal reflux. This discrepancy could be attributed to the differences between the two study populations. Despite the inconsistency between these two studies, the concerns raised have led to a significant change in practice in many CF centres. This has to some extent been extrapolated to other paediatric respiratory disorders with the result that the head-down tipped position is now used much less in paediatric practice. A head-down tip should never be used in children with raised intracranial pressure or in preterm infants because of the risk of periventricular haemorrhage. Abdominal distension places the diaphragm at a mechanical disadvantage and a head-down tilt is likely to exacerbate this further.
Where appropriate, modified gravity-assisted positions can be used in children to assist clearance of bronchial secretions. The upper lobes, particularly the right side, are more frequently affected by respiratory problems and appropriate positioning may be helpful.
Positioning
Positioning may be used to optimize respiratory function. The supine position has been shown to be the least beneficial, while prone positioning has been shown to improve respiratory function (Chapter 4), decrease gastro-oesophageal reflux (Blumenthal & Lealman 1982) and reduce energy expenditure (Brackbill et al 1973). It is often used in closely monitored infants with respiratory problems in a hospital setting, but parents should be advised against using this position when babies are sleeping unattended because of its association with sudden infant death (Southall & Samuels 1992).
Patterns of regional ventilation in infants differ significantly from adults (Davies et al 1985), with ventilation in infants and small children being preferentially distributed to the uppermost regions of the lungs. In acutely ill children with unilateral lung disease, care should be taken if positioning the child with the affected lung uppermost as this may cause rapid deterioration of respiratory status. Spontaneously breathing newborn infants are better oxygenated when tilted slightly head up (Thoresen et al 1988) and show a drop in PaO2 if placed flat or tilted head down.
It is suggested that the redistribution of ventilation, which occurs with a change in body position, results in optimized ventilation to specific lung regions and localized improvement in airway patency. This may result in enhanced secretion clearance from these regions, which are not necessarily those positioned in such a way to allow gravitational drainage (Lannefors & Wollmer 1992).
Manual ventilation
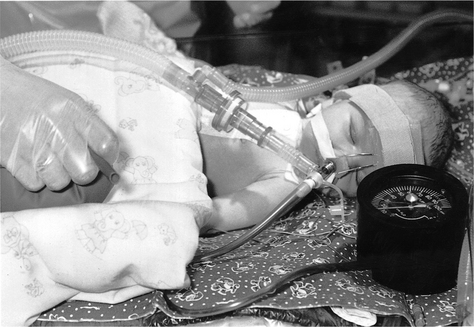
Manual lung inflation involves disconnection of the patient from mechanical ventilation to provide temporary manual ventilation. The same contraindications apply for children and adults (Chapters 5 & 8). However, special consideration should be applied in preterm infants whose lung tissue is easily damaged by high inflation pressures and in children with hyperinflated lungs (e.g. asthma and bronchiolitis) in whom there is a greater risk of pneumothorax. For infants, 500 ml bags should be used and 1 litre bags for older children. They may be valved or open-ended, so that expulsion of excess pressure is controlled by the operator’s fingers. A manometer should be placed in the circuit whenever possible to monitor the inflation pressures (Fig. 10.4). As a general guideline, manual ventilation pressures during physiotherapy should not exceed 10 cmH2O above the ventilator pressure. In order to prevent airway collapse, some positive end-expiratory pressure (PEEP) should be maintained in the bag. Self-inflating bags are used in some units. The flow rate of gas is adjusted according to the size of the child: 4 l/min for infants increasing to 8 l/min for children.
In paediatric patients manual ventilation is used to achieve the following:
Hyperinflation – a long inspiration with an inspiratory pause followed by rapid release of the bag. The aim of this technique is to recruit lung units by improving collateral ventilation and increasing lung volume. However in acute respiratory distress, the proportion of recruitable lung may be extremely variable (Gattinoni et al 2006). Following hyperinflation, a high expiratory flow may assist in mobilizing secretions towards central airways. Some studies support the use of hyperinflation for improving respiratory mechanics (Choi & Jones 2005, Marcus et al 2002). However there remains some controversy over the safety and effectiveness of manual lung hyperinflation as the volumes, pressures and FiO2 are not always controlled and there are inherent dangers of barotrauma (Berney & Denehy 2002, Gattinoni et al 1993, Savian et al 2006). In children with compromised cardiac output, the long inspiratory phase with pause may be contraindicated.
Hyperoxygenation – may be used before suction in order to reduce suction-induced hypoxia or pulmonary hypertension. A review of the efficacy of ventilator versus manual hyperinflation in delivering hyperoxygenation or hyperinflation breaths before, during and/or after endotracheal suctioning found that hyperoxygenation or hyperinflation breaths at 100% oxygen delivered via the ventilator were either superior or equivalent to manually delivered breaths in preventing suction-induced hypoxaemia. However, delivery of manual hyperinflation breaths resulted in increased airway pressure and increased haemodynamic consequences (Stone 1990, Stone & Turner 1989). In the presence of pulmonary hypertension, it is generally not advisable to use an FiO2 of 1.0 during manual hyperinflation as this may further increase blood flow to the lungs.
Hyperventilation – in order to reduce the carbon dioxide in patients with head injury, so that physiotherapy can be safely undertaken, the carbon dioxide should not be allowed to drop too low as this may lead to excessive reduction in cerebral blood flow. In those patients with a large cardiac shunt, hyperventilation may be contraindicated.
Independently performed airway clearance techniques
Over the past two decades, several modalities of airway clearance have been developed. The aim of all of these techniques is to effectively enhance clearance of bronchial secretions and at the same time to facilitate independence with treatment. The majority of techniques were developed for chronic lung disease, in particular cystic fibrosis, but their use has become widespread in both acute and chronic disorders and they are commonly used in paediatric practice (Fig. 10.5). The various techniques are described in detail in Chapter 5 and include:

Figure 10.5 Airway clearance techniques in babies and children. (A) PEP in an infant. (B) Flutter. (C) and (D) Physical activity.
Airway clearance for children with neurological and neuromuscular impairment
Impaired cough, as a consequence of weakness from neuromuscular disease such as Duchenne muscular dystrophy and spinal muscular atrophy or neurological impairment, can cause serious respiratory complications including atelectasis, pneumonia, airway obstruction and acidosis (Miske et al 2004). Chronic respiratory insufficiency and respiratory failure will ultimately result from chronic weakness of respiratory muscles, shallow breathing and ineffective cough. For these children, independently performed airway clearance techniques are not usually feasible, but options such as the ‘cough assist’ (mechanical insufflation/exsufflation device) and other non-invasive forms of positive pressure ventilation are safe and well tolerated in this client group, with growing evidence to support their efficacy (Chatwin et al 2003, Panitch 2006, Vianello et al 2005). They are discussed more comprehensively in Chapters 5 &11. Not all patients with neuromuscular disease are good candidates for the use of non-invasive respiratory aids. Potential contraindications include an inability to manage oropharyngeal secretions, mental status changes or cognitive impairment, and cardiovascular instability. For some patients, including those with the most severe spinal muscular atrophy, sole reliance on non-invasive methods of assisted cough and ventilation is inadequate, and they may require repeated episodes of intubation and mechanical ventilation in the intensive care unit to prolong survival (Birnkrant 2002).
Breathing exercises
It is possible to encourage children to deep breathe from about 2 years of age by using games such as bubbles, paper windmills or incentive spirometers, although the efficacy of these treatments is unproven. Laughing is a very effective means of lung expansion in infants. As children get older, they are able to play a more active role in their treatment and appropriate airway clearance techniques can be introduced (Chapter 5).
Coughing
Younger children are not able to cough to command and although children from about 18 months of age often mimic coughing if asked to do so, the cough is often ineffective. Tracheal compression is occasionally used to try and stimulate a cough in babies. Gentle pressure applied briefly to the trachea, below the thyroid cartilage, causes apposition of the soft and pliant tracheal walls and may stimulate the cough reflex. This technique must be used with care in small infants as this can potentially trigger a vagal response and bradycardia. If a cough cannot be stimulated or if it is ineffective, airway suction may be necessary to remove thick or copious secretions. Changing position or physical activity can be very effective in mobilizing secretions and stimulating a cough reflex in toddlers and older children. Children under the age of 4 or 5 do not usually have the ability to expectorate voluntarily and usually swallow secretions. Even older children can find it difficult to mobilize secretions far enough in to the mouth to expectorate.
Airway suction
Airway suction is discussed in Chapters 5 and 8. Suction techniques may be either naso- or oropharyngeal or endotracheal, depending on whether there is an artificial airway in situ. Adverse effects have frequently been reported and include hypoxaemia, mechanical trauma, apnoea, bronchospasm, pneumothorax, atelec-tasis, cardiac arrhythmias and even death on rare occasions (Clark et al 1990, Clarke et al 1999, Czarnik et al 1991, Kerem et al 1990, Shah et al 1992, Singer et al 1994, Stone & Turner 1989, Wood 1998). Practice varies widely among centres and where available local guidelines should be taken in to consideration (Sole et al 2003).
Complications associated with suction may be reduced by:
 Preoxygenation before suction using ventilator or manually delivered breaths with a higher FiO2 (Chulay & Graeber 1988, Goodnough 1985). Preoxygenation with ventilator breaths has been recommended in preference to disconnection and manual hyperinflation because of the reduced risk of barotrauma, loss of PEEP and FiO2 (Glass et al 1993, McCabe & Smeltzer 1993, Stone et al 1991). Particular care should be taken in preterm infants to avoid hyperoxia, as this is associated with retinopathy of prematurity (Roberton 1996).
Preoxygenation before suction using ventilator or manually delivered breaths with a higher FiO2 (Chulay & Graeber 1988, Goodnough 1985). Preoxygenation with ventilator breaths has been recommended in preference to disconnection and manual hyperinflation because of the reduced risk of barotrauma, loss of PEEP and FiO2 (Glass et al 1993, McCabe & Smeltzer 1993, Stone et al 1991). Particular care should be taken in preterm infants to avoid hyperoxia, as this is associated with retinopathy of prematurity (Roberton 1996). Suctioning via a port adapter or closed suction systems in patients who require maintenance of PEEP and/or positive pressure ventilation during suction (Harshbarger et al 1992).
Suctioning via a port adapter or closed suction systems in patients who require maintenance of PEEP and/or positive pressure ventilation during suction (Harshbarger et al 1992). Avoiding cross-infection, particularly in vulnerable infants, by meticulous hand washing and adherence to local infection control policies.
Avoiding cross-infection, particularly in vulnerable infants, by meticulous hand washing and adherence to local infection control policies. Keeping suction pressures as low as possible, without compromising the efficacy of secretion clearance. High vacuum pressures have been associated with mechanical trauma of the tracheal mucous membranes (Kleiber et al 1988).
Keeping suction pressures as low as possible, without compromising the efficacy of secretion clearance. High vacuum pressures have been associated with mechanical trauma of the tracheal mucous membranes (Kleiber et al 1988). Selecting a suction catheter with an external diameter which does not exceed 50% of the internal diameter of the airway (Imle & Klemic 1989). Most commonly used catheters are 6 and 8 French gauge (FG). Size 5 FG and below are usually ineffective in removing thick secretions. Size 10 FG and above should be reserved for use with older children.
Selecting a suction catheter with an external diameter which does not exceed 50% of the internal diameter of the airway (Imle & Klemic 1989). Most commonly used catheters are 6 and 8 French gauge (FG). Size 5 FG and below are usually ineffective in removing thick secretions. Size 10 FG and above should be reserved for use with older children. Using graduated catheters with centimetre markings to gauge how far the catheter has been passed. Pneumothorax due to direct perforation of a segmental bronchus by a suction catheter has been reported in intubated preterm infants (Vaughan et al 1978).
Using graduated catheters with centimetre markings to gauge how far the catheter has been passed. Pneumothorax due to direct perforation of a segmental bronchus by a suction catheter has been reported in intubated preterm infants (Vaughan et al 1978). Positioning in side lying and restraining the non-intubated child who requires nasopharyngeal suction, to avoid potential aspiration of gastric contents (Fig. 10.6). Constant reassurance should be given throughout the procedure. Supplemental oxygenation and resuscitation equipment should be available. Naso- pharyngeal suction of neonates may cause reflex bradycardia and apnoea.
Positioning in side lying and restraining the non-intubated child who requires nasopharyngeal suction, to avoid potential aspiration of gastric contents (Fig. 10.6). Constant reassurance should be given throughout the procedure. Supplemental oxygenation and resuscitation equipment should be available. Naso- pharyngeal suction of neonates may cause reflex bradycardia and apnoea. Avoiding nasopharyngeal suction if the child has stridor or has recently been extubated, as this may precipitate laryngospasm.
Avoiding nasopharyngeal suction if the child has stridor or has recently been extubated, as this may precipitate laryngospasm.Saline instillation
Saline instillation into the tracheal tube of ventilated patients aims to loosen thick or sticky secretions to facilitate easy removal with suction (Schreuder & Jones 2004). Evidence for the practice is variable and therefore saline should be used only where there is a clear indication. Some suggest that saline instillation at best is not effective and at worst is harmful (Blackwood 1999, Hagler & Traver 1994, Kinloch 1999, McKelvie 1998, Ridling et al 2003), while others suggest it is well tolerated even in infants and may be helpful in removing secretions adherent to the chest wall (Shorten et al 1991). Other mucolytics (N-acetylcysteine) in aliquots of 0.5–5 ml may be used to enhance secretion clearance. Larger quantities of irrigants are sometimes used as part of bronchoalveolar lavage procedures.
Passive movements
Passive movements and two-joint muscle stretches should be considered in older children in intensive care, although they are at less risk of developing joint stiffness than adults. Care should be taken when handling children and infants who are hypotonic in order to avoid soft tissue damage. Preterm infants are hypotonic and require minimal handling, so passive movements are not usually indicated.
RESPIRATORY DISEASE IN CHILDHOOD
Respiratory disease in childhood is very common and is one of the major causes of morbidity and mortality in children worldwide. Outside of the developing countries, most illnesses are mild; only a small proportion are more serious, involving the lower respiratory tract. The overall mortality rate per 100 000 children aged between 1–16 years due to respiratory illness in England and Wales has declined from 8.6 in 1968 to 1.3 in 2000. Asthma, pneumonia and cystic fibrosis (CF) together accounted for 73% of respiratory deaths in this age group (Panickar et al 2005). Respiratory disease is more common in children: from a poor socioeconomic background; with a family history of respiratory disease; from an urban rather than country environment; with a school-age sibling; or with a mother who smokes during pregnancy. The highest morbidity and mortality from lower respiratory tract disease occur in the first year of life. Respiratory disease is more severe in infants with congenital heart or lung abnormalities, immunodeficiency, cystic fibrosis or chronic lung disease.
Asthma
There is considerable global variation in the prevalence rates of asthma, with the highest rates reported in America, Australasia and the United Kingdom. Much lower rates are reported in prevalence studies from Africa and Asia. Prevalence also varies considerably within countries regionally. In the 1980s to early 1990s, several cross-sectional studies from widely varying regions of the world reported an increase in the prevalence of asthma. Although many of these studies relied on self-reported symptoms, there were also reports of a parallel increase in hospitalizations and mortality rates. However, repeat cross-sectional studies over the past decade have suggested a leveling off or even a decrease in prevalance (Toelle & Marks 2005). Atopic (allergic) disease in general has increased over the past few decades and possible explanations for this rise include outdoor pollution, social deprivation/socioeconomic status, dietary factors and passive smoking (particularly maternal smoking during pregnancy). In addition, modern Westernized homes, which tend to be highly insulated (e.g. double glazing) and have increased humidity, have been recognized to be ‘dust mite-friendly’ environments. Thick pile carpets, heavily padded furniture and conventional bedding are all potential sites for dust mite activity, a known trigger for allergic reaction.
The main pathophysiological mechanism of asthma in children is inflammation within the airway, resulting in recurrent episodes of wheezing, breathlessness and cough. There is an increased responsiveness of the smooth muscle in the bronchial wall to various stimuli. Hypertrophy of the mucous glands may lead to mucus plugging. These changes cause variable airway obstruction, which may become chronic and severe.
Aetiology
Children are more likely to develop asthma if parents or close relatives are asthmatic or atopic. There is an important link between atopy and bronchial hyperreactivity, and children with asthma often have other atopic features such as eczema, food allergy, hay fever or urticaria. Exposure to specific allergens such as house dust mite, pollen and animal dander can precipitate bronchospasm and wheeze. Exercise, particularly running, can precipitate an acute attack (exercise-induced asthma, EIA), as can emotional upset or upper respiratory tract infections.
Management
The mainstay of asthma treatment is drug therapy. There are agreed guidelines on the management of asthma (British Thoracic Society & Scottish Intercollegiate Guidelines Network (SIGN) 2003, National Asthma Education and Prevention Programme (NAEPP) 2002). The aims of therapy are to obtain optimal asthma control with few or no symptoms, undisturbed sleep, normal lung function with no limitation to daily activity and no severe, acute exacerbations. Poor asthma control has been attributed to suboptimal adherence to treatment guidelines both by physicians and families (Rabe et al 2004).
Short-acting inhaled β2-agonists may be all that is required in children who have mild intermittent asthma, but inhaled corticosteroids are the mainstay of asthma therapy in those with persistent symptoms and are given in addition to short-acting β2-agonists. Administration of corticosteroids by the inhaled route is safer and results in fewer systemic effects. It is important when using inhaled corticosteroids in children that growth is carefully monitored. In more severe asthma, long-acting β2-agonists should be added to the treatment regimen. Leukotriene receptor antagonists may also be useful in a proportion of cases. Higher doses of inhaled corticosteroids may be needed. The use of continuous (preferably alternate day) oral steroids for prophylaxis is rarely needed nowadays. More severely affected children may require them intermittently on a continuous daily basis, for short periods, during acute exacerbations.
Inhalation of asthma medications provides effective topical therapy, which usually requires smaller doses and has fewer systemic effects. However, the method of drug delivery is very important and has been extensively reviewed (O′Callaghan 2000).
The choice of device depends both on the drug to be delivered and the patient, particularly in relation to age. In children the preferred method for delivery of both inhaled corticosteroids and β2-agonists is by metered dose inhaler (MDI) along with a spacer device. Metered dose inhalers can be manually or breath-actuated and contain a mixture of propellant and drug which is emitted at a high velocity. Breath-actuated devices require an adequate inspiratory flow to trigger the device and the manual devices require coordination of the actuation of the device with inspiration. This makes them inherently difficult to use in children and therefore a valved spacer device should be incorporated into the system. The spacer allows the infant or child to inhale from a reservoir of drug within a chamber.
In babies, a facemask is required and should be held gently over the nose and mouth with the device held upright, at an angle greater than 45°, to ensure the valve is open. The drug can then pass effectively through the open valve to be inhaled (Fig. 10.7A & B). Once the child is older (usually from the age of 2 or 3), the spacer device can be used conventionally with a mouthpiece (Fig. 10.7C). The click of the valve opening will be heard with each breath. It should be noted that different spacer devices have been shown to deliver varying drug doses (Barry & O′Callaghan 1996).
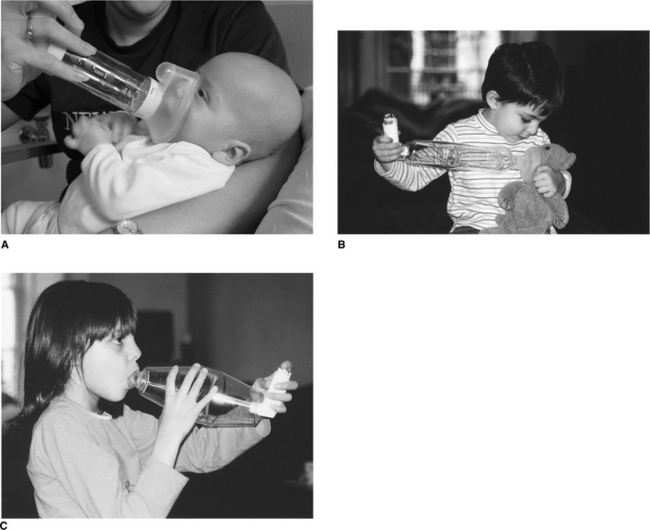
Figure 10.7 Administration of bronchodilator by spacer device to (A) an infant; (B) a teddy bear, to familiarize a young child with the device; (C) a young child.
Nebulized drug delivery systems for asthma are now rarely used in the home setting. They may be used in circumstances where medication cannot be delivered effectively using an MDI and spacer and in severe cases or during an acute exacerbation. It is preferable to use a mouthpiece (if the child is able) so as to avoid drug deposition on the face.
Children with a severe asthma attack usually display signs of acute respiratory distress; they may not be able to complete a sentence in one breath or may not be able to talk, and infants show difficulty in feeding due to breathlessness. The respiratory rate is usually high (>30/minute age 5 years and above, >50/minute - age 2–5 years) and the child is tachycardic. Obvious wheezing may not necessarily be present. In life-threatening attacks, when airway obstruction in the presence of hyperinflation is severe, the airflow may be so low that wheezing is not heard, the respiratory rate is lower than expected and the chest is ‘silent’. The child may be cyanotic and is often exhausted. Children with either severe or life-threatening asthma require immediate admission to hospital. It is important to note that if nebulized bronchodilator therapy is given during an acute attack it should be oxygen driven to avoid hypoxaemia (Inwald et al 2001).
Exercise-induced asthma
Exercise-induced asthma (EIA) is a common symptom associated with childhood asthma. However there is considerable controversy around the subject of EIA. Seear et al (2005) in a study of 52 asthmatic children concluded that the clinical diagnosis of EIA was often inaccurate mainly due to the unreliability of children’s initial reporting of symptoms.
Physiotherapy
A crucial part of the management of asthma is education of the child and parents about the condition and its treatment. Often much of this is undertaken by the primary care team. The role of specialist nurses has also increased greatly in this field, although physiotherapists are sometimes still involved in teaching children how to take their medication.
Physiotherapists should also be able to advise on exercise, which is important in the asthmatic child to maintain general fitness. Improvements in aerobic capacity following exercise programmes have been documented in asthmatic patients (Bingol Karakoc et al 2000, Matsumoto et al 1999, Neder et al 1999), but there is no clear evidence to suggest that exercise training can influence the dose of medication required or improve asthma control in some other way (Carrol & Sly 1999). A systematic review of physical training in asthma concluded that physical training improved cardiopulmonary fitness, although it had no effect on lung function. No adverse effects of exercise were found and the authors stated that there are no reasons why those with asthma should not participate in regular physical activity (Ram et al 2005). A ‘warm-up’ should be recommended before starting vigorous activity (such as football, hockey, running), particularly in children with EIA. The use of a pre-exercise inhaled β-agonist may also be helpful.
A systematic review of the use of breathing exercises was also not conclusive as to the efficacy of this form of intervention in asthma, primarily because the studies included used a wide variety of treatment interventions and outcome measures (Holloway & Ram 2004).
The child with acute asthma may need to be admitted to hospital and in severe cases may require mechanical ventilation (Chapter 9). Often the situation will resolve with careful medical management and appropriate respiratory support. Physiotherapy intervention is not always necessary. However, if problems arise, due to mucus plugging or retained secretions, chest physiotherapy may be of benefit. It is essential that bronchospasm is adequately controlled before physiotherapy techniques are started. Treatment should proceed cautiously and if bronchospasm increases, treatment should be discontinued until bronchospasm can be controlled. Although there is no routine indication for chest physiotherapy in asthma (Hondras et al 2000), children with persistent areas of lung collapse following an acute attack may respond well to an appropriate airway clearance technique. Parents may need to continue physiotherapy at home if bronchial hypersecretion persists.
Bronchiolitis
Bronchiolitis caused by human respiratory syncytial virus (RSV) is the most common severe lower respiratory tract disease in infancy. It is a seasonal disorder, occurring most frequently in the winter months and mainly affects infants under 2 years of age. The cause is viral, with RSV being the main agent in more than 70% of cases. As many as 1–2% of infants require hospital admission for management of RSV infection (Hodge & Chetcuti 2000) and of these 90% are under 12 months. Bronchiolar inflammation occurs with necrosis and destruction of cilia and epithelial cells, leading to obstruction of the small airways. Ventilation/perfusion mismatch may cause hypoxia and hypercapnia. Guidelines for the diagnosis and management of bronchiolitis have been published by the Scottish Intercollegiate Guidelines Network (2006).
Clinical features
The initial presenting symptoms are coryzal, such as the common cold. The infant develops a dry irritating cough and has difficulty in feeding. As the disease progresses, the infant becomes tachypnoeic and wheezy with signs of respiratory distress. The chest radiograph shows hyperinflation and patchy areas of collapse or pneumonic consolidation. Widespread inspiratory cre-pitations and expiratory wheezes can be heard on auscultation.
Management
Management of this condition is mainly supportive. The infant is given humidified oxygen via a head box as required. In those with severe respiratory distress, blood gas monitoring and even ventilatory support may be necessary. Intensive care management of the infant with acute bronchiolitis is discussed in Chapter 9.
Most infants have difficulty with feeding due to respiratory distress. Milder cases may tolerate small, frequent nasogastric feeds, although the nasogastric tube causes obstruction of one nostril and may itself significantly increase the work of breathing. For this reason some centres prefer to use orogastric tubes. Small-volume feeds lessen the risk of vomiting and aspiration. More severely affected infants may require intravenous nutrition.
Antibiotics are not required as the cause of the illness is viral, although they are often used if there is suspicion of secondary bacterial infection. The risk of this is increased if the infant is ventilated and many centres would use intravenous antibiotics for those requiring mechanical ventilation. Bronchodilators or inhaled corticosteroids have not been proven to be of any value in the treatment of acute bronchiolitis (Scottish Intercollegiate Guidelines Network 2006).
Ribavirin is an antiviral agent, which may be effective in reducing severity and duration of the disease. It is delivered as an aerosol by a small particle aerosol generator for long periods (> 3–5 days). The drug is expensive and its efficacy has not been proven and it is therefore not currently recommended for use in acute bronchiolitis in infants (Scottish Intercollegiate Guidelines Network 2006).
Physiotherapy
Physiotherapy is not indicated in the acute stage of bronchiolitis when the infant has signs of respiratory distress. Studies that have examined the efficacy of physiotherapy intervention compared to no treatment in these patients have not shown any benefit in terms of the course of the disease (Nicholas et al 1999, Webb et al 1985). A systematic review based on the results of three randomized controlled trials concluded that chest physiotherapy using vibration and percussion techniques does not reduce length of hospital stay, oxygen requirements, or improve the clinical severity score in infants with acute bronchiolitis who are not under mechanical ventilation and who do not have any other comorbidity (Perrotta et al 2005). The ventilated infant with bronchiolitis needs careful assessment, and physiotherapy techniques should be applied only when sputum retention or mucus plugging is a problem.
Pertussis
Pertussis, commonly called ′whooping cough′, is caused by the organism Bordetella pertussis. It occurs in epidemics every 3–4 years and is largely preventable by immunization, although immunity may not be lifelong (Raguckas et al 2007), with the highest incidence of pertussis since 1959 being reported in 2004. Pertussis is particularly dangerous in infants less than 6 months of age and in children with underlying cardiopulmonary problems, for example congenital heart disease, asthma, chronic lung disease and cystic fibrosis.
Clinical features
The disease starts with coryza lasting 7–10 days during which the child is most infectious. The cough then becomes paroxysmal and can be provoked by crying, feeding or any other disturbance. It is particularly bad at night. The spasms of coughing may cause hypoxia and apnoea, especially in infants, and may lead to further problems such as convulsions, intracranial bleeding and encephalopathy.
At the end of the coughing spasm, the inspiratory whoop may occur followed by vomiting, when thick, tenacious sputum can be expectorated. This phase of paroxysmal coughing may last for 6–8 weeks and is exhausting for the child and parents. The Chinese call pertussis the ‘100-day cough’.
Bronchopneumonia is the most common complication, particularly in infants, and is due to the primary disease itself or to secondary bacterial infection with other organisms such as Staphylococcus, Haemophilus or Pneumococcus. The chest radiograph in severe cases shows hyperinflation and patchy areas of collapse and consolidation.
Management
Most children with pertussis will be managed at home. Infants and children with pneumonia may need admission to hospital. Treatment is supportive. Minimal handling in a quiet environment is essential for the infant with pertussis in order to reduce disturbance, which may precipitate coughing spasms. Nutritional and fluid support should be given throughout the stage of paroxysmal coughing. Antibiotics do not affect the course of the disease, but erythromycin may reduce infectivity and it can also be given prophylactically to close contacts. A small number of cases, particularly infants who have had frequent apnoeic attacks or hypoxic convulsions, will need intensive care and mechanical ventilation.
Physiotherapy
Any physiotherapy manoeuvre, during the acute phase, can precipitate the paroxysmal cough with its complications. Treatment is therefore contraindicated in children during this stage.
If the child or infant requires ventilation, physiotherapy is very important to remove the extremely tenacious secretions, which easily block large and small airways and endotracheal tubes. The paroxysmal cough is not a problem when the child is paralysed in order to be ventilated.
When the stage of paroxysmal coughing is over, there may occasionally be persistent lobar collapse. This lung pathology often responds to an appropriate airway clearance technique. Parents can be taught how to treat the child at home.
Pneumonia
The most common cause of pneumonia in the neonate is Staphylococcus aureus; in the infant, RSV or Mycoplasma pneumoniae and in the child Mycoplasma, Streptococcus pneumoniae or Haemophilus influenzae. However, in a significant number of cases no pathogen is identified (British Thoracic Society 2002).
Clinical features
Presenting signs are pyrexia, dry cough, tachypnoea and not infrequently recession of the ribs and sternum. The chest radiograph shows areas of consolidation. Chest signs are often minimal compared with the degree of illness. Children with underlying pulmonary disease are particularly at risk from pneumonia.
Physiotherapy
In many cases of pneumonia there is consolidation of lung tissue with no excess secretions and there is no evidence that physiotherapy is of benefit (Stiller 2000). Where sputum retention is a problem, an appropriate airway clearance technique may be used. Copious amounts of sputum may be cleared in one treatment, following which the pyrexia may settle and the child will feel better. Reassessment of the child is often necessary, as retention of secretions may become a recurrent problem as the pneumonia resolves.
Pleural infection
Pleural infections, although relatively uncommon, have become more prevalent in the United Kingdom and the United States of America in recent years. Empyemas are a significant cause of morbidity in children, but differ from pleural infections in adults in that the final outcome is usually very good (Balfour-Lynn et al 2005). A pleural effusion in a relatively well child is usually a secondary occurrence to an acute bacterial pneumonia. The effusion is usually unilateral. Very occasionally pleural effusions in children represent an underlying malignancy; otherwise, most effusions are associated with an underlying infection. Once the presence of an effusion has been confirmed by chest radiograph or chest ultrasound and other causes ruled out, most children are started on intravenous antibiotics. A loculated effusion is treated either locally, with chest drain insertion and intrapleural fibrinolytics, or surgically with video-assisted thoracic surgery (VATS) or mini-thoracotomy.
Physiotherapy
Although these children do not always have a primary problem with bronchial secretions, immobility and the presence of a chest drain can result in retained secretions and a weak cough. Airway clearance may be necessary, using an appropriate technique. Breathing exercises and advice on coughing are also important parts of treatment. As soon as the clinical condition allows, the child should be encouraged to mobilize as much as possible.
Acute laryngotracheobronchitis (croup)
Croup is a common problem occurring between the ages of 6 months and 4 years. The illness is usually viral and produces acute inflammation and oedema of the upper airways.
Clinical features
The presenting symptoms are coryzal and later include fever, a harsh barking cough and a hoarse voice. Stridor, initially inspiratory only, is much worse at night and may become inspiratory and expiratory. Signs of respiratory obstruction are seen and the severely affected child may develop respiratory failure. The acute stage of respiratory obstruction may only last 1–2 days but the stridor and cough may continue for 7–10 days. Some children have recurrent bouts of croup.
Management
Mild cases can be managed at home. Extra humidity is often given, for example by sitting with the child in a warm steamy bathroom, although there is no objective evidence of benefit from inhaled moist air in emergency settings (Moore & Little 2006). More severely affected infants will be admitted to hospital and given humidified oxygen if hypoxic or distressed. Treatment is supportive, but with minimal handling as any disturbance that upsets the child will increase the laryngeal obstruction.
Glucocorticoids (dexamethasone and budesonide) have rapid beneficial effects on symptoms (Russell et al 2004). Nebulized adrenaline may be given with careful observation, in case of rebound and an acute collapse, and has been shown to provide short-term relief, but is probably not useful in the long term. Antibiotics are not usually required unless there is more specific evidence of a bacterial cause, for example purulent secretions.
Very few children with croup who are admitted to hospital go on to require intubation to maintain the airway due to severe respiratory obstruction. A few of these, particularly infants, may also require some additional form of respiratory support, e.g. intermittent positive pressure ventilation or continuous positive airway pressure.
Acute epiglottitis
Epiglottitis is caused by Haemophilus influenzae but is now rarely seen due to the introduction of the Hib (Haemophilus influenzae) vaccine. It is, however, a very dangerous condition, which occurs between the ages of 1 and 7 years.
Management
The child with suspected epiglottitis should not be disturbed in any way. No attempt should be made to examine the throat, as this may precipitate acute life-threatening obstruction. Usual management is intubation with a nasotracheal tube. In extreme circumstances tracheostomy may be necessary, but should only be required for 3–4 days, following which there is usually complete recovery.
Bronchopulmonary dysplasia
Infants who remain oxygen-dependent and have abnormal findings on chest radiograph are described as having bronchopulmonary dysplasia (BPD). BPD covers a broad range of disease and a variety of terminology have been used to describe this disorder, including chronic lung disease (CLD). Although both CLD and BPD are both still commonly used, it is felt that BPD distinguishes this disorder as a neonatal lung process rather than other chronic respiratory diseases (Jobe & Bancalari 2001, Ryan 2006). The classification of BPD into mild, moderate and severe, depending on oxygen and positive pressure requirement, may offer a better description of underlying pulmonary disease and has been reported to correlate with the infant’s maturity, growth and overall severity of illness (Ehrenkranz et al 2005). BPD is seen in extremely low birthweight infants and is inversely related to gestational age (Johnson et al 2002). Reported incidence of BPD varies from 15–50%, although this is likely to be related to the difference in populations (i.e number of very premature infants) among the studies.
The pathology of BPD has changed considerably over the past few decades, since the use of newer modalities of mechanical ventilation, introduction of new treatments (such as surfactant) and also due to improved survival of extremely premature infants. The pathology of BPD used to be associated with fibrosis and airway obstruction but in the present population of BPD babies, the problem is one of abnormal lung growth (in particular a marked reduction in alveolar numbers) (Kotecha 2000). This pathological picture is often termed ‘new’ BPD (Greenough et al 2006).
In addition to prematurity and low birthweight there are several other risk factors for BPD, in particular the requirement of mechanical ventilation and oxygen therapy. High peak pressures in positive pressure ventilation cause barotrauma and high inspired oxygen concentrations cause an acute inflammatory response leading to local tissue damage. Other factors that also influence the pathogenesis of BPD include the presence of a persistent arterial duct – patent ductus arteriosus (PDA) and infection.
Despite several studies, the optimum ventilation mode whereby BPD can be prevented has not been identified. Preventative strategies aim to mimimize lung injury. These include using less mechanical ventilatory support, refining the methods of mechanical ventilation and using alternative techniques: permissive hypercapnia, minimal peak pressures, rapid ventilatory rates, early use of continuous positive airway pressure (CPAP) and rapid weaning and extubation (Ambalavanan & Carlo 2006). High-frequency ventilation, in particular high-frequency oscillation, may have a place in preventing BPD but this is as yet unclear (Greenough et al 2006). The infant with BPD shows an increased oxygen requirement and carbon dioxide retention and has decreased lung compliance with increased airway resistance. Tachypnoea and persistent sternal and costal recession are often present. The condition may be progressive, requiring more ventilatory support and eventually leading to respiratory and cardiac failure. Radiographic appearance can vary but in classic BPD shows interstitial fibrosis and cystic abnormalities. The radiographic appearance in ‘new’ BPD is often of small volume and hazy lung fields.
Supplementary oxygen is the mainstay of the baby with BPD. The most appropriate target for the oxygen saturation level requires further study (Greenough et al 2006). Good nutrition is essential and the infant may require fluid restriction and diuretics. Some infants respond to bronchodilators and steroids, although the effect of long-term steroids on lung and brain growth is an issue of concern. Antibiotics may be required as these infants are prone to recurrent chest infections. Babies with a chronic oxygen requirement but who have a reasonable growth rate and do not have frequent episodes of desaturation can be discharged with home oxygen. These families require appropriate community support.
The long-term prognosis for those who survive the first 2 years is good, although infants with BPD have significant pulmonary sequelae during childhood and adolescence (Bhandari & Panitch 2006).
Physiotherapy
Infants with CLD are particularly prone to chest infections and have an increased rate of hospital admission in the first 2 years of life. Physiotherapy may be indicated if secretion retention is a problem. However these infants often wheeze and may have airway collapse. Detailed assessment is important before any intervention. If wheezing is not too severe, careful treatment may be possible. Inhaled β2-agonists may temporarily improve lung function in these babies (Ng et al 2001) and may be useful as a premedication before physiotherapy. Modified gravity-assisted positions with chest percussion may be useful in infants, but nasopharyngeal suction may be required if retained secretions are a cause for concern. In older children an appropriate airway clearance technique should be used, either during episodes of infection or if retained secretions are a persistent problem. Children, particularly infants in whom supplemental oxygen is delivered via nasal cannulae, often have a problem with thick, dry nasal secretions and may need humidification (Chapter 5).
Inhaled foreign body
Aspiration of a foreign body into the respiratory tract can occur at all ages, but is most common between the ages of 1 and 3 years. All types of foodstuffs may be aspirated, for example peanuts, pieces of fruit and vegetables, as well as small plastic or metal toys. Objects are most commonly aspirated into the right main bronchus. The left main bronchus and trachea are the next most common, and smaller objects may be inhaled into right middle and lower lobe bronchi or occasionally into the left lower lobe bronchus.
When aspiration has been witnessed by parents or carers, the child should be taken immediately to hospital. On examination there may be wheeze and some signs of respiratory distress. Breath sounds may be reduced over the affected lung. The chest radiograph, taken on expiration, may show gas trapping in the area distal to the blockage.
In some cases, the aspiration is not witnessed and the acute changes just described may be assumed to be the onset of a respiratory infection. The bronchial wall becomes oedematous, especially if the inhaled object is vegetable matter. Total obstruction of the bronchus gradually occurs and secondary pneumonic changes develop in the area distal to the obstruction. After a few days the child may become unwell with a persistent cough. The longer the obstruction remains, the more permanent the lung damage, eventually leading to bronchiectasis (Dinwiddie 1997). An inhaled foreign body should be suspected in any child with a pneumonia that does not respond to conventional treatment.
Management
All children who have aspirated a foreign body into the airway should have an urgent rigid bronchoscopy for removal of the foreign body. If symptoms persist, a repeat bronchoscopy may be necessary to ensure complete removal. Rarely bronchoscopic removal may fail and thoracotomy may be required.
Physiotherapy
Physiotherapy is not indicated to attempt to remove the object before bronchoscopy. Usually physiotherapy is ineffective as the object is firmly wedged in the bronchus. However, if the object is dislodged by physiotherapy manoeuvres, it may travel up the bronchial tree and obstruct the trachea, leading to respiratory arrest.
Following bronchoscopy, gravity-assisted positioning and chest clapping may be necessary to clear excess secretions, particularly if the object has been aspirated for some time and secondary bacterial infection has occurred.
Primary ciliary dyskinesia
Primary ciliary dyskinesia (PCD) is a rare, genetic disorder in which cilial motility is severely reduced because of structural defects within the cilia (Chapter 18), leading to disease of the upper and lower respiratory tract. Disorders of ciliary structure or function result in recurrent sinusitis and bronchiectasis due to decreased clearance of secretions (Cowan et al 2001). Males may be infertile because of reduced cilial motility of the sperm tails. Visceral mirror image arrangement occurs in approximately 50% of patients. Cilia can be examined for motility using nasal epithelial brushings.
Infants with this condition may present in the neonatal period with persistent rhinitis or pneumonia, but many children present later with chronic upper and lower respiratory tract infection. This condition is not curable, so treatment is directed towards preventing infection and chronic lung damage (Ferkol et al 2006). Appropriate antibiotic therapy is required during periods of infection. Children often require daily physiotherapy to clear bronchial secretions. An individualized programme of airway clearance should be formulated using an appropriate airway clearance technique (Chapter 5). There has been very little work published on chest physiotherapy in this condition, but it has been suggested that airway clearance techniques may be useful (Gremmo & Guenza 1999) (Chapter 18).
Cystic fibrosis
Cystic fibrosis (CF) is the most common inherited recessive condition in Caucasians, occurring in about 1 in 2500 births. The disorder results from a defective gene on chromosome 7, which encodes for the CF transmembrane conductance regulator (CFTR) protein. One of the functions of this protein (as a chloride channel) is to regulate ion transport. The absence or dysfunction of CFTR leads to defective ion transport and may result in either low volume or abnormal salt concentration of the airway surface fluid (Boucher 2004) (Chapter 18). This results in increased mucus viscosity and impaired mucociliary clearance, contributing to recurrent bacterial infection with gradual lung destruction. The survival of patients with CF has increased dramatically over the past five decades and this improvement has been attributed largely to improved nutrition and early aggressive treatment of the chest (Jaffe & Bush 2001). The primary areas affected are the respiratory and digestive tracts, but CF is a multisystem disorder with complications such as liver disease, diabetes and low bone mineral density.
CF may be identified in some babies before birth when echogenic bowel is seen on routine antenatal ultrasound scan or where there is a family history of CF and parents choose to have antenatal diagnosis. Others are identified as having CF through regional or national neonatal screening programmes. Twelve to fifteen per cent of children present at birth with meconium ileus, where thickened inspissated meconium causes blockage of the colon and ileum. The infant may present in the first day or two of life with abdominal distension, vomiting and failure to pass meconium. The obstruction can often be managed conservatively by the use of Gastrografin enema but occasionally laparotomy may be required. Other modes of presentation include recurrent chest infections and/or failure to thrive. Diagnosis of CF is confirmed with blood sampling for identification of two CF mutations and/or a sweat test.
Much focus has been placed on the early detection of lung disease in babies with CF. Traditionally the assessment of clinical status involved relying on symptoms, physical signs and investigations such as chest radiography, until the child was old enough to undertake respiratory function testing (approximately age 5–6 years). However, it is now possible to measure infant lung function and these studies along with early bronchoscopy and high-resolution computed tomography show clearly that infants with CF, who have no overtly detectable signs of respiratory disease, do have abnormalities in airway function and structure, and evidence of inflammation and even infection, within the early months of life (Armstrong et al 1995, Lum et al 2007, Martinez et al 2005, Ranganathan et al 2001). It is therefore important that these changes are detected and appropriate treatment instigated early. This may also be important in the future, when novel therapies such as gene therapy, manipulation of CFTR function and perhaps stem cell therapy are likely to work best at an early stage of the disease process, before irreversible lung damage has occurred.
Physiotherapy
Improvements in survival in CF have been attributed to regular monitoring, attention to nutrition and early, aggressive multidisciplinary treatment of CF lung disease. Most babies and young children attending cystic fibrosis centres now have good nutritional status, with body mass index (BMI) within normal limits, and do not display any recognizable signs of respiratory disease. Traditionally, airway clearance (usually in the form of postural drainage (PD) and percussion) was instigated on diagnosis. This approach is still taken by many physiotherapists, although many centres no longer use the head-down tipped position in these babies, as a consequence of a small study which suggested that a head-down tip may exacerbate gastro-oesophageal reflux and may have both short- and long-term consequences to the child’s respiratory status (Button et al 2003, 2004). In addition, these babies often have very little in the way of secretions and justifi-cation for postural drainage with a head-down tip is questionable.
Although there is considerable evidence that inflammation and infection are present from an early stage in CF and that changes in lung function and structure occur long before the onset of obvious clinical signs, the early pathophysiological picture is not associated with copious secretions or other symptoms which respond to airway clearance therapy. This has led many to question the appropriateness of a routine daily airway clearance regimen in these babies and it remains unclear whether airway clearance treatments are effective in asymptomatic babies with CF or whether a routine regimen of chest physiotherapy should be instigated regardless of clinical status (Prasad & Main 2006). An alternative approach is that these babies are very carefully monitored and active treatment applied only when symptoms warrant it. Parents and carers should always be taught an appropriate airway clearance technique soon after diagnosis and this should be practised and expertise maintained. They should also be taught how to check the child’s chest and when to instigate treatment (Chapter 18). The importance of physical activity should be emphasized from the time of diagnosis. Even if the child is not receiving a daily regimen of airway clearance, parents should be encouraged to engage in some sort of physical activity on a daily basis, even in infancy. In addition to modified postural drainage and percussion (Fig. 10.8), the use of other airway clearance techniques such as positive expiratory pressure, assisted autogenic drainage and the use of physical activity (e.g. bouncing on a gym ball) are becoming more widely used in these babies (Chapter 5).
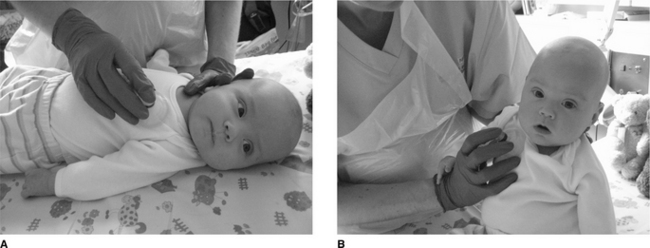
Figure 10.8 Modified postural drainage in an infant, using a facemask for percussion (A) in supine and (B) in sitting for the upper lobes.
As children grow older, they can begin to play a more active role in their treatment. Many different airway clearance modalities are now available and the treatment regimen should be individualized depending on clinical status, age and social circumstances. The overall physiotherapy treatment and the various airway clearance techniques are discussed in detail in Chapters 5 and 18.
MANAGEMENT OF THE ACUTELY ILL INFANT OR CHILD
Babies and children with a wide variety of medical and surgical problems are admitted to neonatal, general paediatric or cardiac intensive care units. Respiratory failure in acutely ill infants and children may have various aetiologies. In the neonate the most common causes are prematurity, respiratory distress syndrome, asphyxia and aspiration pneumonia. Under 2 years of age bronchopneumonia, bronchiolitis, asthma, croup, foreign body inhalation and congenital heart or pulmonary anomalies are more common aetiologies. In children over 2 years’ asthma, central nervous system infection (e.g. meningitis) and trauma are more frequent.
The effects of surgery, anaesthesia and immobility are the same in infants and children as in adults (Chapter 12). However, anatomical and physiological differences between these populations make children more vulnerable to respiratory complications. Infants and children undergoing major surgery should therefore be regularly assessed by a physiotherapist.
Physiotherapy treatments for ventilated children involving manual techniques have been shown to have an advantage over routine suction in reducing respiratory resistance, which may be of substantial benefit in patients with evidence of acute atelectasis (Main et al 2004). Within individuals, physiotherapy treatments are also more likely to produce improvements in tidal volume, respiratory compliance and resistance than suction alone, but both physiotherapy and suction procedures can produce short-term deterioration in almost one-third of children. Sensitive tools still need to be identified for selection of patients most likely to benefit from physiotherapy (Main et al 2004).
Physiotherapists working in an intensive care unit should be familiar with the equipment used on that unit (Fig. 10.9). They should be able to respond when a problem is indicated by the monitors and be able to ascertain whether the problem is patient or equipment related. Details of oxygen delivery and paediatric mechanical support are described in Chapter 9. Standard monitoring equipment is often similar to those used on adult intensive care units (Chapter 8), although normal values vary according to age (see Table 10.1).
Figure 10.9 Equipment used in a paediatric intensive care unit. Figure shows an infant undergoing high-frequency oscillatory ventilation.
Children can have either nasal or oral endotracheal tubes (Fig. 10.10). The narrowest part of the upper airways in babies and small children is the circular cricoid ring. Thus, perfectly sized uncuffed tubes can be passed nasally and form a good seal in the cricoid and reduce the risk of damage to the tracheal mucosa from larger cuffed tubes (Deakers et al 1994, Khine et al 1997). Another advantage to nasal intubation is that the mouth is free to suck a sponge or pacifier during ventilation, so that normal feeding can be started as soon after extubation as possible. An important disadvantage to ill-fitting uncuffed tubes is the potential risk of endotracheal tube leak, in terms of both inconsistent delivery of ventilation and inaccurate monitoring of respiratory function (Kuo et al 1996, Main et al 2001).
Neonatal intensive care
The reasons for admission to a neonatal intensive care unit (NICU) include the following:
Preterm delivery: defined as less than 37 completed weeks of gestation (full term is defined as 38–42 weeks). Preterm infants who require admission to a NICU are usually less than 32 weeks of gestation with a birthweight less than 2500 g. Some infants are born as early as 23 weeks’ gestation and are considered at the edge of viability. Causes of preterm birth include antepartum haemorrhage, cervical incompetence, multiple pregnancies or infection. There is also an association with deprived socioeconomic circumstances and in some cases the cause of preterm delivery is unknown.
Low birth weight is often due to prematurity but more mature infants may also be of low birth weight due to intrauterine growth retardation. Causes include placental dysfunction, smoking and intrauterine infection, e.g. rubella.
Perinatal problems such as birth asphyxia or meconium aspiration.
Congenital abnormalities include congenital heart disease and diaphragmatic hernia.
General problems of infants in the NICU
Infection
The preterm infant is particularly vulnerable to infection. Early-onset sepsis can occur during the first days of life. Infection can occur at any time during a NICU stay and may occur as a complication of invasive therapies in immature immune systems. The most important means of preventing and reducing cross-infection is by meticulous attention to hygiene by both staff and visitors.
Physiological jaundice
Physiological jaundice is common in the normal full-term infant owing to the breakdown of fetal haemoglobin causing a raised level of unconjugated bilirubin in the blood. It usually begins 2 days after birth and disappears after 1 week to 10 days. High levels of unconjugated bilirubin may diffuse into the basal ganglia and lead to a condition called kernicterus, characterized by athetoid cerebral palsy, deafness and mental retardation. Preterm infants are particularly prone to developing jaundice and run an increased risk of subsequent kernicterus, though this condition is now extremely rare. Serum bilirubin levels are closely monitored and phototherapy may be required. Phototherapy units consist of white or blue lamps, which emit light of wavelength 400–500 nm. Light of these wavelengths oxidizes unconjugated bilirubin into harmless derivatives. Infants receiving phototherapy have to be nursed naked, which can cause problems of temperature control. There is also increased insensible fluid loss and a theoretical risk of eye damage, so eye shields are placed on the infant. Advances in technology have led to a new phototherapy system that enables effective treatment to be given without the inconveniences of conventional phototherapy. The BiliBlanket® system uses fibre optics to provide therapeutic light for the treatment of physiological jaundice, filtering out the more harmful ultraviolet and infrared light. The fibre optics are covered with a pad of woven fibres and the pad is placed directly against the baby. In severe cases of jaundice an exchange transfusion (where small amounts of blood are replaced by donor blood until twice the infant’s blood volume has been exchanged) may be required.
Nutrition
Adequate calorie intake and weight gain are important in preterm and low birthweight infants to avoid hypoglycaemia, persistent jaundice and delayed recovery from respiratory distress syndrome. Feeding should be started as soon as possible, either enterally in those who can tolerate it, or intravenously. Preterm infants have poor sucking, gag and cough reflexes so will be fed nasogastrically until these develop. Continuous infusion of milk may be preferable to bolus feeds, which can increase respiratory distress, regurgitation and aspiration because of abdominal distension. Feeds are often better tolerated when the infant is lying in the prone position. Orogastric tubes may be used rather than nasogastric ones in order to avoid blockage of the nostril in spontaneously breathing infants with respiratory distress.
Temperature control
Preterm and low birthweight infants have difficulty in maintaining their body temperature because they have a large surface area relative to their body mass and easily lose heat through the skin by evaporation and radiation. They also have a smaller proportion of brown fat in comparison with full-term infants. Hypothermia may cause acidosis, hypoglycaemia, increased oxygen consumption and decreased surfactant production. Infants should therefore be kept in a thermoneutral environment (incubators or under radiant warmers) to maintain body temperature. Heat shields may be used to reduce radiant heat loss and the ambient room temperature is kept high at 27–28°C. A core temperature of less than 36.5°C in preterm infants indicates that non-essential handling should be delayed until the infant’s temperature has risen. Infants, especially preterm babies, may have difficulty maintaining their temperature and are therefore nursed in incubators or under radiant warmers.
Pulmonary haemorrhage
Pulmonary haemorrhage is defined as acute intrapulmonary bleeding. It is relatively uncommon but may be a life-threatening event. Physiotherapy is contraindicated, although regular suctioning may be required to keep the airway clear. When fresh blood is no longer being aspirated, physiotherapy techniques may assist removal of residual blood. Prognosis is often poor.
Patent ductus arteriosus
Patent ductus arteriosus (PDA) occurs in up to a third of all preterm infants of less than 1500 g at birth (Zahka & Patel 2002). In the full-term infant the duct, which is a fetal circulatory vessel, closes within the first 24 hours of life. A persistent patent duct in the preterm infant may lead to increased pulmonary blood flow. If symptomatic, a PDA can be treated medically (using indomethacin) or surgically (with ligation), depending on the infant’s clinical status.
Periventricular haemorrhage and periventricular leucomalacia
The incidence of intraventricular bleeding in preterm infants is inversely proportional to birth weight, occurring most frequently and severely in the smallest and least mature infant. Haemorrhage in the capillaries in the floor of the lateral ventricles is common in very low birthweight infants. The bleeding may extend into the ventricles and subarachnoid space. Several factors contribute to the risk of bleeding. These include hypoxia, fluctuation in blood pressure and cerebral blood flow and venous congestion. Cerebral ultrasound scanning is used to diagnose periventricular bleeds and is often the only way that small bleeds are detected. Periventricular haemorrhage is graded according to the extent of the bleeding seen on the ultrasound scan:
 Grade I Grade I |
bleeding into the floor of the ventricle |
 Grade II Grade II |
bleeding into the ventricle (intraventricular haemorrhage (IVH)) |
 Grade III–IVH Grade III–IVH |
with dilatation of the ventricle |
 Grade IV–IVH Grade IV–IVH |
and bleeding into the cerebral cortex causing areas of ischaemia. |
The smaller bleeds (grades I and II) have a good prognosis. They usually require no treatment and have no long-term sequaelae. Neurological development following a grade I or II bleed seems to be similar to that of an infant with a comparable gestation. Larger bleeds may need treatment with shunting and the outcome is dependent on the grade of the bleed. More severe bleeds are associated with ischaemic brain damage and therefore have a high mortality and morbidity.
Periventricular leucomalacia (PVL) may occur on its own or associated with PVH. Ischaemia of cerebral tissue adjacent to the ventricles causes formation of cystic lesions. There is an association with neurological problems, particularly diplegia.
Respiratory distress
Respiratory distress syndrome (RDS) is a common complication of preterm infants, primarily caused by structural and biochemical immaturity of the lung, in particular a lack of surfactant. The incidence of RDS increases with decreasing gestational age. Symptoms can develop within 4 hours of delivery with sternal and costal recession, nasal flaring, grunting and tachypnoea. Steroids are usually administered to women in preterm labour in an attempt to enhance lung maturation (Roberts & Dalziel 2006).
Pulmonary surfactant production usually begins 36–48 hours after birth, regardless of gestational age. The more mature infant will start to recover at this time. Very preterm infants who have other problems compounding their respiratory distress or infants who have developed complications of treatment may require ventilatory support for much longer. Surfactant therapy has significantly altered the treatment of RDS. Both prophylactic and early surfactant replacement therapy have been shown to reduce mortality and pulmonary complications in ventilated infants with RDS. A systematic review has reported advantages of early surfactant replacement therapy with extubation to nasal continuous positive airway pressure (CPAP) (Stevens et al 2004).
As lung collapse in RDS is primarily caused by lack of surfactant, physiotherapy is not required for this condition. Secretions may become a problem after the infant has been intubated for more than 48 hours, owing to irritation of the tracheal mucosa by the endotracheal tube. These secretions may be cleared easily by suction alone. Physiotherapy may be indicated if suction is not adequately clearing secretions.
Respiratory distress in the preterm infant can also be caused by pneumonia. Organisms causing pneumonia may be bacterial, viral or fungal and may be acquired before, during or after birth. The most serious bacterial cause is group B streptococcus. The presenting features of this pneumonia are similar to RDS with an indistinguishable chest radiograph. Group B streptococcal pneumonia can be rapidly fatal unless antibiotic therapy is started early. For this reason all infants presenting with respiratory distress are given antibiotics.
Parent-infant bonding
Infants who have been resuscitated and require immediate admission to a NICU shortly after birth will not have had the chance for physical contact with their parents. Incubators and other equipment may be a further barrier to contact. Parents will need to be supported by the NICU team through this difficult period and should be encouraged to have as close contact as possible with their baby and help with routine care.
Neonatal mechanical ventilation
Prophylactic ventilation is often started from birth in infants <1000 g, although wherever possible CPAP delivered via nasal prongs would be the intervention of choice. Other indications for ventilation are: deteriorating blood gases (hypoxaemia or hypercapnia) despite a high FiO2, recurrent or major apnoea, major surgery pre- or postoperatively for congenital anomalies.
The goals of mechanical ventilation in the NICU are to achieve and maintain adequate gas exchange, to reduce the work of breathing and to minimize the risk of secondary lung injury. Achieving synchrony between the baby’s breathing pattern and the ventilator is important, as asynchrony is associated with alterations in arterial and cerebral blood flow and an increased incidence of pneumothorax. Attempts to achieve synchrony include fast ventilatory rates and paralysing agents to suppress the infant’s respiratory drive. Time-cycled, pressure-limited devices have largely been replaced by newer ventilators which offer pressure control or volume limitation and patient triggering (Donn & Sinha 2003). Volume-limited ventilation enables the measurement of very small tidal volumes and the provision of very low flows.
The use of high-frequency ventilation has also increased over the past decade. Whether high-frequency oscillation (Chapter 9) can reduce the incidence of bronchopulmonary dysplasia remains unclear, but it is regarded as a useful rescue technique.
Complications of mechanical support in neonates
Pneumothorax may be caused by many factors including high peak inspiratory pressures, high positive end-expiratory pressure and long inflation times or ventilator asynchrony. Predisposing factors include hyperinflation of alveoli occurring in conditions such as meconium aspiration and respiratory distress syndrome (RDS). A tension pneumothorax is likely to cause a sudden deterioration and usually requires immediate insertion of an intercostal drain. Very small pneumothoraces may not require drainage.
Pulmonary interstitial emphysema used to be common in preterm infants, with the incidence being inversely proportional to gestational age. It has been seen less frequently since the introduction of treatment of preterm infants with exogenous surfactant, which improves lung compliance. Pulmonary interstitial emphysema is still seen occasionally in infants who require long-term ventilation and who have uneven aeration and gas trapping.
Retinopathy of prematurity is a condition of preterm infants seen when the capillaries in the retina proliferate, leading to haemorrhage, fibrosis and scarring. In the most severe form, this may result in permanent visual impairment. The cause is unknown, but periods of hyperoxia (exact length of time unknown) with a PaO2 of above 12 kPa are thought to be a major predisposing factor (Roberton 1996). Careful oxygen monitoring is essential to attempt to prevent this condition.
Prolonged ventilatory support and oxygen dependency may result in bronchopulmonary dysplasia (BPD), discussed earlier in this chapter.
Perinatal problems and congenital anomalies
Meconium aspiration
Meconium aspiration usually occurs in full-term infants who become hypoxic due to a prolonged and difficult labour. Hypoxia causes the infant to pass meconium into the amniotic fluid and to make gasping movements, thereby drawing meconium into the pharynx. The irritant properties of meconium can cause a chemical pneumonitis and meconium aspiration can also lead to significant gas trapping and thoracic air leak. In severe cases it can result in secondary persistent pulmonary hypertension of the newborn (PPHN). A severely affected infant is likely to require mechanical venti-lation and may require extracorporeal membrane oxygenation.
Physiotherapy is very important when meconium aspiration has occurred in order to remove the extremely thick and tenacious green secretions. In milder cases treatment consists of gravity-assisted positioning, as tolerated, with chest percussion. Treatment is usually well tolerated soon after aspiration. In more severe cases where pneumonitis develops, these babies are often very sick, require significant respiratory support, do not tolerate handling and should only be treated with caution.
Congenital diaphragmatic hernia
Diaphragmatic herniation occurs when abnormal fetal development of the diaphragm weakens the muscular barrier. The abdominal contents (usually stomach or small bowel) are displaced into the thoracic cavity, posteriorly and most commonly on the left side. The incidence is approximately 1 in 3000 births with mortality rates up to 50% (Schultz et al 2007). The abnormality may be diagnosed antenatally by ultrasound or postnatally in significant defects when the infant presents with neonatal respiratory distress. A chest radiograph will show abdominal viscera in the thoracic cavity. Unless the herniation has occurred late in pregnancy, which is very unusual, there will be associated pulmonary hypoplasia on the affected side as the abdominal viscera occupy the space normally available for the growing lung. Pulmonary hypoplasia is the main determinant of survival. The contralateral lung is also smaller than expected because of compression due to mediastinal shift during fetal development. There are also commonly other associated anomalies such as persistent fetal circulation and abnormalities of the pulmonary vasculature.
The infant with diaphragmatic hernia is often very unwell, particularly as the bowel in the chest distends with air and further compresses the lungs, and requires immediate gastric decompression with simultaneous intubation and ventilation. Surgery is not usually carried out until the infant’s condition is stabilized and extracorporeal membrane oxygenation may be required to support the infant until surgery is possible. A systematic review has reported a reduction in early mortality with extracorporeal membrane oxygenation but no overall long-term benefit (Morini et al 2006). Surgical correction is via a laparotomy. The abdominal viscera are carefully returned to the abdominal cavity and the defect in the diaphragm is closed. High-frequency oscillation has been reported as a beneficial elective ventilation strategy but as yet there have been no large randomized controlled trials (Smith et al 2005).
Postoperatively, the infant may require ventilation for some time, depending on the amount of pulmonary hypoplasia. Prognosis is variable and mortality for isolated hernias is about 45% (Wenstrom et al 1991). Physiotherapy may be indicated if retention of secretions is a problem. Manual hyperinflation techniques should avoid generating excess pressures within the hypo-plastic lungs.
Congenital anomalies of the lung
Congenital conditions of the lung such as lobar emphysema, lung cysts and adenomata are relatively rare. They may be diagnosed by ultrasound antenatally or by chest radiography postnatally. Treatment may involve surgical resection (lobectomy) if the condition is severe, but in some cases the lesions appear to resolve spontaneously in infancy.
Acquired lobar emphysema and lung cysts are more common as complications of respiratory distress syndrome and its treatment. Many cases resolve with medical management, although some do require resection. Physiotherapy may be indicated postoperatively if there is sputum retention, but manual hyperinflation is contraindicated if cysts are present.
Oesophageal atresia and tracheo-oesophageal fistula
There are five recognized types of this anomaly. In the most common variety the oesophagus ends in a blind proximal pouch (atresia) and there is a fistula between the trachea and the lower section of the oesophagus. About 10% of affected infants have oesophageal atresia with a tracheal fistula. The incidence is approximately 1 in 3000 births (Depaepe et al 1993).
The infant presents postnatally with episodes of choking, coughing and respiratory distress due to an inability to swallow saliva or feeds and consequent aspiration into the larynx or trachea. It is often difficult to pass a nasogastric tube, which on chest radiograph appears curled in the upper oesophagus.
Surgical correction is usually attempted as soon as possible and involves division of the fistula and anastomosis of the ends of the oesophagus. Some anastomoses may have to be performed under tension and the infant has to be electively ventilated and paralysed with the neck kept in flexion postoperatively. In a few cases, where the gap between the two ends of the oesophagus is too large, primary anastomosis is not possible and a feeding gastrostomy is performed. Oesophageal anastomosis or replacement by colonic, jejunal or gastric interposition is delayed.
If recurrent or continuous aspiration occurs before corrective surgery, physiotherapy (in the head-up position) may be indicated to clear excess secretions or treat lung collapse due to reflux of gastric contents. Preoperatively the airway is often kept clear by continuous suction of the upper pouch and the infant should be nursed head-up to prevent reflux of gastric contents through the fistula.
Postoperatively, head-down postural drainage is contraindicated and patients are often nursed in the head-up position for the first few days, to reduce the risk of reflux. Care must be taken not to extend the neck, especially in patients with a tight oesophageal anastomosis. Naso- or oropharyngeal suction should not in general exceed the external distance between the nasal cavity and the ear. This distance is effective at producing cough and inadvertent damage to the oesophageal anastomosis is avoided.
Gastroschisis and exomphalos (omphalocele)
These conditions are relatively rare abdominal wall defects, occurring in approximately 1 : 5000 births (Baird & MacDonald 1982). Gastroschisis refers to a full thickness abdominal wall defect next to the umbilical opening, through which the small and large bowel herniate, not usually covered by a membrane. Exomphalos occurs when the anterior abdominal wall fails to close at the base of the umbilical cord, allowing the abdominal contents and sometimes the liver to herniate through the umbilical ring and develop externally in utero. A translucent membranous sac encloses the hernial contents. The defect is usually diagnosed antenatally by ultrasound and is classified as major or minor depending on whether the defect is bigger or smaller than 5 cm. Affected infants often have other major associated anomalies.
Immediately after birth, the abdominal contents are covered to prevent heat and fluid loss until corrective surgery can be undertaken. In most cases primary repair is possible but where the defect is large a staged procedure is required, with gradual reduction of the bowels into the abdominal cavity.
Postoperatively the infant may require ventilation as the tightly packed, rigid abdomen causes respiratory embarrassment and compromises venous return. Where a staged procedure is necessary, prolonged ventilation may be required. Some infants have impaired antenatal lung growth and a proportion continue to have abnormal lung function during infancy.
These infants are particularly at risk from retention of secretions and lobar collapse due to the distended abdomen and predominantly supine nursing position (with the abdominal contents suspended above the abdomen). If treatment is required, techniques that increase intrathoracic pressure and consequently intra-abdominal pressure, such as vibrations or manual hyperinflation, should be used very cautiously. Postoperative respiratory compromise, if related to increased abdominal pressure, is unlikely to respond to physiotherapy. A slightly head-up position may relieve the thorax of some of the weight of the abdominal contents and reduce the work of breathing.
Cardiac intensive care
Congenital heart problems in infants and children
Congenital heart disease is the most common congenital anomaly with the incidence of moderate and severe forms about 6 in 1000 live births, and of all forms about 75 in 1000 live births (Hoffman & Kaplan 2002). Roughly one-third of these will require surgical intervention, with the rest either resolving spontaneously or being haemodynamically insignificant. Major congenital cardiac defects can often be detected antenatally by ultrasound examination, while more minor defects may not be detected until the postnatal period. Diagnosis is usually confirmed by echocardiography. Postnatally most cardiac defects are amenable to surgery and overall mortality has fallen to less than 5% in the best units (Elliott & Hussey 1995, Stark et al 2000). Early complete repair is attempted whenever possible, with the majority of operations being performed in the first year of life.
Management of congenital heart defects must involve agreement between cardiologist, surgeon, family and the child, if he is old enough. Each aspect of the child’s care is an integrated process requiring the skills of a multidisciplinary team before, during and after surgery.
Common paediatric cardiac surgery procedures
The normal anatomy of the heart is shown in Figure 10.11.
Figure 10.11 Anatomy of the normal heart: AO, aorta; PA, pulmonary artery; SVC, superior vena cava; IVC, inferior vena cava; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; LPA, left pulmonary artery; PV, pulmonary veins; LBV, left brachiocephalic vein.
Palliative procedures
When a primary repair is not possible, palliative or staging procedures will provide temporary or extended relief of symptoms. They are usually indicated to deal with excessive pulmonary blood flow, inadequate pulmonary blood flow or inadequate mixing between oxygenated and deoxygenated blood in the heart.
Pulmonary artery band.
The pulmonary artery band is designed to restrict excessive blood flow to the lungs by reducing the diameter of the pulmonary artery with a constricting tape. A child with excessive pulmonary blood flow (ventricular and atrioventricular septal defects or truncus arteriosus) may present with poor feeding, heart failure, tachypnoea and, if uncorrected, pulmonary hypertension. If a corrective procedure is not possible, pulmonary artery banding may be performed via a left thoracotomy, to protect the lungs from the progression of pulmonary vascular disease. The pulmonary artery pressure is reduced to approximately one-third of the systemic pressure.
The modified Blalock–Taussig shunt.
The modified Blalock–Taussig shunt (MBTS) is the most common palliative procedure used to improve pulmonary blood flow by placing a conduit between the subclavian artery and the pulmonary artery via sternotomy or thoracotomy. Inadequate pulmonary blood flow will result in poorly oxygenated blood and central cyanosis (e.g. tetralogy of Fallot, pulmonary or tricuspid atresia). If primary repair is not possible, the MBTS temporarily improves pulmonary perfusion, thereby significantly improving oxygen saturation (80–85%). The shunt is usually ligated at the time of definitive repair.
Septostomy.
In defects such as transposition of the great arteries, where there is inadequate mixing of oxygenated and deoxygenated blood within the heart, the foramen ovale may be enlarged using either a balloon atrial septostomy in neonates or surgically in older children via a Blalock–Hanlon septectomy.
Corrective surgery: closed procedures
Patent ductus arteriosus.
The ductus arteriosus is the fetal vascular connection between the main pulmonary trunk and the aorta (usually distal to the origin of the left subclavian artery), which normally closes soon after birth. If it remains open, excessive blood shunts from the aorta to the lungs causing pulmonary oedema and, in the long term, pulmonary vascular disease. Symptoms may be mild or severe, depending on the magnitude of the left-to-right shunt. This defect occurs very commonly in premature infants and may cause difficulty weaning from ventilation or congestive cardiac failure.
In some circumstances (for instance, neonates with transposition of the great arteries) it is desirable to delay closure of the ductus arteriosus and this may be achieved by the administration of prostaglandin.
It may also be possible to induce closure of the duct in preterm infants with indomethacin. Surgical correction involves a left thoracotomy and ligation. In older infants closure may be achieved via cardiac catheterization using a double umbrella device.
Coarctation of the aorta.
This is a congenital narrowing of the aorta. It usually occurs proximal to the junction of the ductus arteriosus and distal to the left subclavian artery origin. Neonatal presentation with symptoms of congestive heart failure requires early surgical repair. This is usually performed by resection of the stenosis and end-to-end anastomosis. If the aortic arch is extensively hypoplastic, aortic arch angioplasty may be necessary. Repair of simple coarctation carries almost zero mortality. For severe forms of coarctation such as interrupted aortic arch (where upper and lower aortic arches are separated) mortality is higher. Paraplegia is an extremely rare complication specific to correction of this defect (Brewer et al 1972) and may be associated with longer cross-clamping times.
Vascular ring.
This defect is caused when malformations of the aorta or pulmonary artery compresses the trachea, oesophagus or both (examples include double aortic arch, abnormally positioned innominate artery or abnormal course of the left pulmonary artery crossing behind the trachea). Symptoms include stridor, respiratory difficulties, repeated chest infections or feeding problems. Surgical decompression of the vascular ring will often improve symptoms, but tracheal stenosis or malacia are frequently associated with vascular rings and further surgery may be required to repair tracheal or bronchial obstruction.
Corrective surgery: open procedures
Open procedures require cardiopulmonary bypass, modified for children in terms of size, flow rate, perfusion, temperature and drugs (Elliott & Hussey 1995).
Atrial septal defect (ASD).
Atrial septal defect is one of the most common congenital cardiac anomalies, characterized by a hole in the septum that separates the left and right atria. Types of ASD include ostium primum defects, also referred to as partial atrioventricular sep-tal defects (AVSDs), discussed below, and ostium secundum defects due to failure of fusion of the two atrial septa and patency of the foramen ovale. Ostium secundum ASD may be associated with one or more of the superior pulmonary veins draining into the superior vena cava.
Children with ASD are generally asymptomatic and diagnosis is usually made after a murmur is detected at routine examination. If undiagnosed, slow development of symptoms may occur with rising pulmonary artery pressure and pulmonary vascular disease. If pulmonary vascular disease becomes severe and pulmonary hypertension is irreversible, then corrective surgery is not possible and heart-lung transplantation is the only pal- liative option. Because of the severe late consequences of pulmonary hypertension, repair is usually undertaken before the age of 5 years via median sternotomy or right anterior thoracotomy. Late diagnosis and surgical intervention are rare in developed countries. The septal defect is usually closed by direct suture, pericardial or synthetic patch. Umbrella or balloon devices have also been used successfully to close small, round defects via cardiac catheterization.
Ventricular septal defect (VSD).
Ventricular septal defects are the most common congenital cardiac lesions, defined by a hole in the septum that separates left and right ventricles. VSDs are often found in conjunction with other cardiac defects and the clinical presentation will depend on the size of the VSD and the presence or absence of other cardiac anomalies. Infants may present with congestive cardiac failure, recurrent chest infections and failure to thrive. More than half of all VSDs close spontaneously and do not require surgery (Elliott & Hussey 1995). However, as with ASDs, undiagnosed larger defects can lead ultimately to severe irreversible pulmonary hypertension.
VSDs (Fig. 10.12) are defined according to their position in either the perimembranous inlet, the trabecular portion or the muscular outlet of the ventricular septum. Primary repair is usually performed using synthetic or bovine pericardial patches via median sternotomy, with the cardiac approach varying according to the position of the defect. Conduction disturbances are common following surgery.
Figure 10.12 Ventricular septal defect, showing mixing of blood between the left and right ventricle: AO, aorta; PA, pulmonary artery; SVC, superior vena cava; IVC, inferior vena cava; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; LPA, left pulmonary artery; PV, pulmonary veins; LBV, left brachiocephalic vein.
Although operative mortality approaches zero for isolated septal defects, multiple VSDs or ‘Swiss cheese’ defects carry a higher risk (De Leval 1994a).
Atrioventicular septal defect (AVSD).
Incomplete development of the inferior atrial septum, superior ventricular septum and atrioventricular valves results in a spectrum of anomalies termed atrioventricular septal defects. Symptoms vary in severity according to the magnitude and direction of the shunt and the extent of the ASD, VSD, valve incompetence or combination of these. They may be associated with other cardiac defects (transposition of the great arteries, tetralogy of Fallot) and are also strongly associated with chromosomal abnormalities such as Down’s syndrome. Some patients may be asymptomatic despite high pulmonary vascular resistance, but a high left-to-right shunt causes dysp-noea, recurrent chest infection and congestive cardiac failure.
Partial AVSD refers to an ostium primum type of ASD above the mitral and tricuspid valves that are displaced into the ventricles and may be incompetent. The development of pulmonary vascular disease is uncommon.
Complete AVSD is distinguished by a single six-leafed atrioventricular valve between the right and left atrioventricular chambers and continuous with the ASD above and VSD below. Over 50% of infants with this defect will die within the first year of life because of pulmonary vascular disease if left untreated. The remaining children will almost all have died within 5 years.
Both types of AVSD are repaired with patches on cardiopulmonary bypass via a median sternotomy. Hospital mortality is usually less than 10% but may be greater in patients with major associated anomalies. Early complete repair is preferred so that irreversible development of pulmonary vascular disease may be avoided but conduction problems and valve incompetence are relatively common postoperatively.
Tetralogy of Fallot.
The four components of Fallot’s tetralogy are classically described as a large VSD, right ventricular (infundibular) outflow or valve obstruction, overriding aorta and right ventricular hypertrophy (Fig. 10.13).
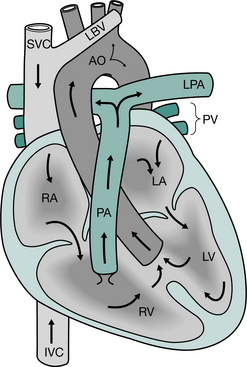
Figure 10.13 Tetralogy of Fallot, showing VSD, right ventricular hypertrophy, aorta overriding both ventricles and stenosis of the pulmonary artery: AO, aorta; PA, pulmonary artery; SVC, superior vena cava; IVC, inferior vena cava; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; LPA, left pulmonary artery; PV, pulmonary veins; LBV, left brachiocephalic vein.
Inadequate blood flow to the pulmonary circulation and preferential flow of deoxygenated blood to the aorta may cause cyanosis, but severity of symptoms will depend on the degree of obstructed pulmonary blood flow. The majority of infants are pink at birth but become progressively cyanosed as they grow. Periodic spasm of the infundibulum prevents blood flow to the lungs and may cause ‘spelling’ episodes in which infants become irritable. Continued crying leads to increasing cyanosis and eventual loss of consciousness. The spasm then relaxes and the child gradually recovers. These episodes are dangerous and may lead to death or cerebral anoxia. Older undiagnosed children may intuitively squat following exercise, which reduces blood flow to and from the lower extremities in an effort to compensate for the large oxygen debt accrued during physical activity. In the presence of cyanosis, this behaviour may suggest diagnosis of this defect.
Some controversy exists about whether it is better to do primary repair or palliative shunt with repair when the child is older. Corrective surgery will involve closure of the VSD, resection of the hypertrophied infundibulum and reconstruction of the pulmonary arteries. Long-term results are good with actuarial survival of 93% at 15 years and good quality of life (Castenda 1994).
Pulmonary atresia.
The infant with pulmonary atresia may be cyanosed at birth and this may become rapidly worse as the ductus arteriosus closes. Palliation in the form of a modified Blalock–Taussig shunt is the immediate treatment of choice so that adequate blood supply to the lungs can be established. Prostaglandins may be used to delay closure of the ductus arteriosus until surgery. In the absence of right ventricular hypoplasia and coronary artery abnormalities, mortality is very low. However, this defect can occur with a VSD, in which case the right ventricle may be hypertrophied or hypoplastic and the pulmonary valve atretic. Sometimes the coronary arteries are supplied with desaturated blood from the right ventricle and major aortopulmonary collateral arteries (MAPCAs) can augment pulmonary blood flow. The technique used for definitive surgical repair is variable depending on the size of the right ventricle.
Transposition of the great arteries (TGA).
This defect is characterized by the aorta originating from the right ventricle and the pulmonary artery from the left (Fig. 10.14). Oxygenated pulmonary blood recirculates through the lungs without reaching the body and deoxy-genated blood recirculates through the body without reaching the lungs. The two closed circulations would quickly lead to death but there is usually a degree of mixing through the PDA and, if present, associated anomalies such as ASD or VSD. Babies therefore present soon after birth with cyanosis and immediate treatment aims include keeping the ductus arteriosus open with prostaglandins until surgery. Before corrective surgery, cardiac catheterization and balloon atrial septostomy may also be necessary.
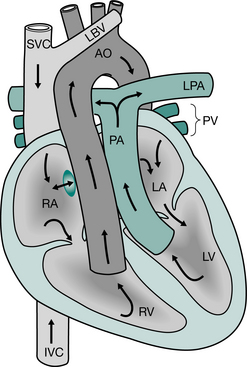
Figure 10.14 Transposition of the great arteries. Shaded area shows either position of a patent foramen ovale or site of balloon septostomy allowing some mixing of oxygenated and deoxygenated blood between the systemic and pulmonary circulations: AO, aorta; PA, pulmonary artery; SVC, superior vena cava; IVC, inferior vena cava; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; LPA, left pulmonary artery; PV, pulmonary veins; LBV, left brachiocephalic vein.
The arterial switch operation has been performed with good results since 1985 and is the preferred option for simple TGA or for TGA with VSD. It is generally performed in the first 2–3 weeks of life, while the pulmonary vascular resistance is high and the left ventricle is ‘trained’ to receive the systemic workload. The aorta and pulmonary arteries (above the level of the coronary vessels) are transected and transferred to their correct anatomical positions. The coronary arteries are also transferred to their appropriate positions. Operative mortality is low (< 2%) and long-term results appear to be far superior to the earlier Mustard or Senning operations, which redirected blood flow via intra-atrial tunnels (Freed et al 2006).
Interrupted aortic arch.
This rare condition is characterized by a discontinuous aortic arch and will result in death within the first month if left untreated. The most common site for interruption is distal to the left carotid artery. A VSD is almost always present, as is a PDA through which blood flows to the distal aorta. Soon after birth, when the ductus arteriosus begins to close, the pulmonary vascular resistance increases and severe congestive cardiac failure develops. Early surgical repair is the treatment of choice, but is technically difficult and the postoperative course is often prolonged.
Partial or total anomalous pulmonary venous connection (PAPVC or TAPVC).
These anomalies are rare and involve some or all four pulmonary veins connecting to the systemic venous circulation. Supracardiac connections (50%) involve blood draining to the innominate vein or the superior vena cava. Cardiac connections (20%) involve blood draining into the coronary sinus or directly into right atrium. Infradiaphragmatic connections (20%) drain into the portal or hepatic veins. The final 10% are mixed connections. The reduced left atrial pressure keeps the foramen ovale open postnatally and mixed arterial and venous blood is transported systemically. Thus symptoms in the first few days of life will include congestive cardiac failure and cyanosis. There are often associated cardiac anomalies and surgical repair will depend on the nature of these, if present.
Truncus arteriosus.
Truncus arteriosus is characterized by a single arterial trunk arising from both ventricles and from which the aorta and pulmonary arteries originate via a single semilunar valve. A VSD permits flow up the common trunk. Congestive cardiac failure and irreversible pulmonary vascular disease rapidly develop in early infancy and untreated infants rarely survive beyond their first year. Surgical treatment involves the closure of the ventricular septal defect with a patch to divert the left ventricular flow up the aorta. The pulmonary arteries are then detached from the common artery (truncus arteriosus) and connected to the right ventricle using a conduit.
Cardiac valve abnormalities
Aortic stenosis.
Obstruction to left ventricular outflow may occur in isolation or in combination with other cardiac defects. They may be found at valvular, subvalvular, supravalvular or combined levels. Critical stenoses present neonatally with congestive cardiac failure and reduced peripheral pulses and require immediate intervention. Relief of aortic stenosis may be obtained with aortic valvotomy, aortic valve replacement, homograft insertion or balloon dilatation. However, mortality is high (10%) and reoperation common (Elliott & Hussey 1995). Aortic stenosis may not cause problems until adulthood, although, by then, a degree of left ventricular hypertrophy may have developed.
Pulmonary stenosis.
The neonate with pulmonary stenosis may become progressively more cyanosed as the PDA closes and this may be reversed or delayed by the use of prostaglandins to keep the PDA patent. Management is similar to that of pulmonary atresia, with surgery dependent on the nature and extent, if any, of associated cardiac anomalies. Homograft valve replacement may be required at a later stage. Less critical pulmonary stenosis may present later in life with breathlessness on exertion and fatigue.
Tricuspid valve.
Tricuspid valve disease is rare in childhood but is seen in Ebstein’s anomaly. Patients present with severe cardiac failure, cyanosis and dysrhythmia. Neonatal surgery carries a high mortality and a palliative approach with a later Fontan procedure may be preferred (De Leval 1994b). In the older child it is possible to perform a more complex repair of the anomaly.
Mitral valve.
Mitral valve problems present either as stenosis or incompetence, usually associated with other cardiac anomalies. Repair is the preferred option though replacement may be the only option. Early replacement is associated with a high mortality (20%) (Carpenter 1994).
Hypoplastic left heart syndrome.
This defect is defined by aortic valve stenosis or atresia associated with severe left ventricular hypoplasia. Early mortality in untreated patients is high. The systemic blood flow derives almost entirely from the right ventricle through the ductus arteriosus and, depending on the size of it, peripheral pulses may be normal, reduced or absent. Immediate management involves keeping the ductus arteriosus open with prostaglandins. Surgical management options include early heart-lung transplantation or a three-staged surgical procedure that makes the child’s circulation function with only two of the heart’s four chambers. The first step (Norwood procedure) makes the right ventricle pump blood to the whole body and to the lungs. The second stage (Glenn procedure) allows greater blood flow to the upper body and reduces some of the workload from the right ventricle. The final procedure (Fontan procedure or total cavopulmonary connection) allows blood to return passively to the lungs (rather than being pumped there) and allows the right ventricle to only pump blood out to the body (Norwood & Jacobs 1994). The Norwood procedure is generally performed within a week of birth, the second stage at 3–6 months of age, and the Fontan at 18 months to 4 years of age. Success of these types of surgery depends on the lungs being free of pulmonary vascular disease and a good balance between pulmonary and systemic circulations.
Transplantation surgery in children
The problems of cardiac, lung and heart-lung transplant surgery in children are similar to those in adults, and are discussed in detail in Chapter 15.
General paediatric intensive care
Trauma & head injury
Accidents are the most common cause of child death after the first year of life and 50% are road traffic accidents. Children who have been severely injured may require intensive care and mechanical ventilation, particularly after head injury.
In the acutely head-injured child the primary injury refers to the damage sustained during trauma caused by bleeding, contusion or neuronal shearing. Secondary injury is due to the resultant complicating events. These may be intracranial factors such as bleeding, swelling, seizures and raised intracranial pressure (ICP) or systemic factors such as hypoxia, hypercarbia, hyper- or hypotension, hyper- or hypoglycaemia and fever. In the United Kingdom, 90–95% of injuries are managed without the need for neurosurgical intervention (Tasker et al 2006). When required, for example in the presence of an acute subdural bleed, surgical evaluation should be facilitated immediately.
The most common presentation of acute, severe head injury in children is coma. Clinical scores, such as the paediatric modifications of the Glasgow Coma Scale (GCS) (Tasker 2000, Teasdale & Jennett 1974), allow bedside assessment of neurological function and the degree of impairment of consciousness in children. Such scores are designed to allow early identification of pathology when it is still potentially reversible by medical or surgical intervention.
Coma in children may present after a longer interval than in adults. Continued extradural bleeding following a relatively minor injury may lead to a deteriorating level of consciousness. Cerebral oedema may be focal or generalized; the latter may result in an increase in intracranial pressure and cause a more rapid deterioration.
Raised intracranial pressure
Raised intracranial pressure (ICP) represents an increase in the volume of the intracranial contents. In addition to trauma, it can be caused by space-occupying lesions or encephalopathy. Normal values for ICP fall below 15 mmHg. The cerebral perfusion pressure (CPP) is the driving pressure for cerebral blood flow and is defined as the difference between mean arterial blood pressure and ICP. It is a crucial parameter, which lies within the range of 50–70 mmHg. A variety of methods to monitor ICP can be used including intraventricular catheters, subdural or subarachnoid monitors and cerebral intraparenchymal catheters.
Once the child is stabilized, medical management aims to avoid or minimize secondary brain injury. Factors that may precipitate a rise in ICP, resulting in a potential fall in CPP, should be avoided. Intubation and mechanical hyperventilation have been sometimes been used to reduce ICP. This has been controversial as hyperventilation (and hypocapnia) may induce cerebral ischaemia. In general, this is still sometimes used for short periods during acute deterioration or when intracranial hypertension is unresponsive to other therapy.
The management of children with acute head injury has been reviewed extensively (Tasker 2001). Many of the strategies for management are similar to adults and discussed in Chapter 8.
Pulmonary problems following liver transplantation
Liver transplantation is used for chronic end-stage liver disease and fulminant hepatic failure. Shortage of paediatric donors means that more and more grafts are reductions of adult livers. In some situations one donor liver can be used for two patients. Postoperative complications include bleeding and splinting of the right side of the diaphragm. Patients invariably develop a pleural effusion which is usually right-sided but may be bilateral. Acute rejection is common 5–7 days post- transplant. Some patients develop chronic rejection and require retransplantation (Salt et al 1992).
Physiotherapists may have the opportunity to assess these patients preoperatively, but often patients with fulminant hepatic failure are operated on as an emergency or are too ill to be seen preoperatively.
Postoperatively, the risk of bleeding in some patients means that handling is kept to a minimum. Patients are assessed regularly and treated as appropriate. Following extubation, ambulation is encouraged as soon as possible. Large pleural effusions coupled with ascites mean patients are often very breathless and unable to mobilize.
KEY ISSUES FOR PHYSIOTHERAPISTS TREATING ACUTELY ILL CHILDREN
Paediatric intensive care units are often stressful and demanding environments. When children are critically ill, intubated and ventilated, therapists often have to use complex clinical reasoning to make difficult decisions. Classic contraindications to certain treatment techniques may sometimes be superceded by more compelling clinical needs, for example a small or moderate pneumothorax may be considered less important than acute lobar atelectasis.
Thorough assessment before to any physiotherapy intervention in ventilated children is essential. A report from the nurse in charge of the baby will provide important information about the current haemodynamic status and stability of the child. A recent chest radiograph will be helpful in identifying any focal areas of respiratory compromise and should be used in conjunction with thorough auscultation in order to decide on the appropriateness of intervention. Evaluation of fluid status, urine output, heart rate and rhythm, blood pressure, platelets, bleeding, inotropic support, level of sedation, will all contribute to a decision on how any treatment should be performed.
Successful treatments depend on accurate assessment and then thorough and continuous evaluation of clinical data during treatment from a multitude of different sources, including haemodynamic and res-piratory information, observation and auscultation. Experience and competence are essential in the management of the most critically ill babies, as is a complete knowledge of underlying anatomical and physiological processes likely to influence the outcome of treatment. For example, treating a child with a univentricular pulmonary and systemic circulation with high bagging pressures or high oxygen concentration would dramatically influence the flow of blood to the lungs with potentially life-threatening consequences. Similarly, treating a child with head injury without appreciating the relationship between intracranial pressure, mean arterial pressure and cerebral perfusion pressure could have serious immediate and long-term consequences for their recovery.
Technological advances in recent years have meant that modern ventilators often incorporate pressure and flow sensors which allow continuous monitoring and calculation of tidal breathing parameters or respiratory mechanics from which an assessment of respiratory function can be made (MacNaughton & Evans 1999). It is imperative that physiotherapists familiarize themselves with these devices, the interpretation of data generated from them and their limitations in the clinical environment (Castle et al 2002). There is great potential for such equipment to provide objective feedback about efficacy and tolerance of treatments in individual patients and to provide excellent tools for systematic evaluation of physiotherapy treatment in mechanically ventilated infants.
Physiotherapy management of surgical patients
Preoperative management
In some hospitals preoperative visits and handbooks are available which help to reduce some of the fear of being in hospital. Except in emergency admissions, it is desirable for children and their parents to be seen by a physiotherapist before their surgery. Appropriate explanation of postoperative procedures should be given at the level of the child’s age and understanding, but overloading the child with information they do not understand may increase stress and anxiety. It is important that parents understand the need for postoperative physiotherapy intervention as they can play an important role in encouraging postoperative mobility.
Physiotherapy assessment should include cardiores-piratory status and physical and motor development. The assessment of respiratory status provides an opportunity to evaluate postoperative risks and the need for preoperative treatment. If indicated, older children may be taught an airway clearance technique. Incentive spirometry can be useful in reducing atelectasis in children after cardiac surgery, especially those techniques specifically designed for children (Krastins et al 1982).
When a child has pre-existing pulmonary disease, for example cystic fibrosis, they may need to be admitted some time before surgery for prophylactic antibiotics and for effective airway clearance. Such children may require physiotherapy and suction in the anaesthetic room following intubation and before surgery (Tannenbaum et al 2007).
Several congenital cardiac anomalies are associated with a broader spectrum of embryological malformations, some of which result in developmental delay. These children may require long-term developmental follow-up. Any preoperative neurological problems or developmental delay should be documented and appropriate management plans formulated.
Postoperative management
In addition to the altered pulmonary dynamics and respiratory insufficiency seen after general anaesthesia, open heart surgery with cardiopulmonary bypass leads to further changes in respiratory function. The lungs may be compressed during surgery, contributing to atelectasis, loss of perfusion and diminished surfactant production, all of which contribute to poor respiratory compliance postoperatively. Children and infants should be regularly reviewed and treated as required.
As sedation is reduced and children are able to take a more active role in their treatment, effective pain relief is essential. Pain due to the incision and presence of intercostal drains may cause splinting of the chest wall and reduced excursion. Adequate pain relief can be provided through continuous infusion or patient-controlled systems in older children. It may be difficult to assess the severity of pain in children, although the development of specific paediatric pain scales has made it easier in recent years (Razmus & Wilson 2006). Children in pain can be withdrawn and immobile and infants in pain may be tachycardic and tachypnoeic. Some children who have a fear of needles will deny pain in order to avoid injections.
Treatment is directed towards early extubation and mobilization. When in bed, children should be comfortably positioned in alternate side lying or sitting upright and the ‘slumped posture’ should be avoided. As soon as possible, children should be sat out of bed and walking encouraged when appropriate. Drips, drains and catheters can all be carried to allow early ambulation. Attention to posture is important, particularly following thoracotomy when arm and shoulder exercises to the affected side are also essential.
If sputum retention is a problem postoperatively, airway clearance techniques may be required. A child may prefer not to have his wound supported or to support his own wound when coughing.
Prematurity
Preterm and critically ill neonates tolerate handling poorly and should therefore be handled as little as possible. The skin of a preterm baby is very thin and easily damaged. Manual techniques should be applied with care. Physiotherapy and suction should only be carried out when indicated and careful assessment is essential before any intervention.
The positioning of infants receiving mechanical ventilation in NICU may have an impact on clinical outcome. The lateral decubitus positions, prone and supine are all used, although there is a tendency to nurse ventilated infants in the prone position. A systematic review of 11 trials involving 206 infants reported that prone positioning did have some advantage in terms of improved oxygenation but that there was no evidence to suggest that any one position during mechanical ventilation was more effective in producing a clinically relevant and sustained improvement (Balaguer et al 2006).
An active programme of pre- and post-extubation chest physiotherapy may result in a lower incidence of lobar collapse and reintubation within 24 hours (Flenady & Gray 2002). However a large randomized controlled trial testing the effects of a neonatal post-extubation programme on the incidence of post-extubation collapse found no differences between the physiotherapy and control group in terms of the rate of post-extubation collapse, adverse events (apnoea or bradycardia), duration of requirement of supplemental oxygen or the need for re-intubation within 24 hours (Bagley et al 2005).
An association between chest physiotherapy and encephaloclastic porencephaly in extremely preterm infants was reported by Harding et al in 1998. This study involved a retrospective analysis of 454 infants with birth weights less than 1500 g delivered between 24 and 27 weeks’ gestation. Affected subjects received two to three times as many chest physiotherapy treatments as did the control group, but the group also had more prolonged and severe episodes of hypotension in the first week than controls and were less likely to have had a cephalic presentation at delivery. The lesions were considered to be caused by impact of the brain with the skull during shaking movements, which could occur during chest physiotherapy with percussion (Harding et al 1998). Since this publication, however, several authors have disputed the association between encephaloclastic porencephaly and chest physiotherapy (Beby et al 1998, Gray et al 1999, Vincon 1999). The significant methodological errors in this work have been highlighted and it is possible that these lesions occurred only in the sickest infants and the fact that they had more chest physiotherapy may be a reflection of their degree of illness (Gray et al 1999). No cases of encephaloclastic porencephaly were reported over the same 3-year period in two separate studies, despite similar criteria for initiation of chest physiotherapy (Beby et al 1998, Gray et al 1999). Follow-up data from the centre in New Zealand which first reported this problem later suggested that identification of encephaloclastic porencephaly emerged at a time when the use of chest physiotherapy had already decreased and that the cluster of cases seen between 1992 and 1994, although associated with the number of chest physiotherapy treatments given, may have been due to some other factor (Knight et al 2001). Although an association between chest physiotherapy and encephaloclastic porencephaly seems unlikely, it highlights the need for very careful assessment of preterm infants and a judicious approach to treatment. If chest physiotherapy is indicated and chest percussion thought appropriate, the baby should be kept in a stable position, with the head and shoulders well sup-ported, and vital signs carefully monitored throughout treatment.
Head injury
Immobility, impaired cough, depression of the respiratory centre and pulmonary dysfunction due to anaesthetic and paralysing agents predispose patients to pulmonary complications. The frequency of pneumonia in severely head-injured patients requiring prolonged mechanical ventilation has been reported to be as high as 70% (Demling & Riessen 1993).
Safe and effective treatment should be based on careful assessment and judicious use of appropriate physiotherapy techniques (Prasad & Tasker 1990). The use of bolus doses of analgesics and sedatives or, in more unstable cases, thiopental before an intervention can help reduce acute swings in ICP. Length of treatment time is an important factor, with longer treatment more likely to produce larger elevations of ICP. Sustained increases in ICP during cumulative interventions should be avoided by allowing a return to baseline values between procedures. Careful monitoring of CPP during treatment is essential and treatment should be withheld or abandoned if levels fall below 50 mmHg.
A head-down position is generally contraindicated and any change in position should maintain the head midline in relation to body position. A 30° head-up tilt has been shown to significantly reduce ICP in the majority of patients (Feldman et al 1992). The presence of a bone flap from decompressive craniotomy may limit options for positioning. Chest clapping may be better tolerated than vibrations and manual hyperinflation may be used with careful monitoring (Prasad & Tasker 1995). Endotracheal suctioning may have severe prolonged effects on ICP (Gemma et al 2002) and great care must be taken to avoid hypoxia. A protocol for physiotherapy management is shown in Figure 10.15.
Figure 10.15 Flow diagram of an approach to chest physiotherapy in children with raised intracranial pressure.
(Reproduced with permission from Prasad and Tasker 1990)
Passive movements to maintain joint mobility may be felt necessary and it has been shown that these can be undertaken without detrimental effect on ICP in adults, provided that Valsalva-like manoeuvres are avoided (Brimioulle et al 1997).
Extracorporeal membrane oxygenation
Extracorporeal membrane oxygenation (ECMO) provides complete or partial cardiopulmonary bypass support for the heart and lungs or lungs alone when extremely ill children have severe but potentially reversible cardiac or respiratory failure (Huang et al 2007) (Chapter 9). It has been reported to be beneficial in both neonates and children with acute lung injury and respiratory failure (Pearson et al 1993, Swaniker et al 2000, UK Collaborative ECMO Trial Group 1996).
In some respects, physiotherapy treatments for children on ECMO may seem relatively less stressful for the physiotherapist. Children on full cardiopulmonary ECMO support are not reliant on mechanical ventilation for adequate oxygenation and it may not be as important to connect ventilation circuits quickly or complete suction cycles quickly. However, children on ECMO are heparinized and thus very vulnerable to bleeding, with potentially devastating consequences for survival or long-term outcome. Too much movement or coughing during physiotherapy may raise intracranial pressure and cause brain or pulmonary bleeding. If the nursing report suggests active haemorrhage, therapists should consider whether it is appropriate to apply manual techniques or treat at all. Adequate blood flow through the ECMO circuit is often dependent upon body position and the therapist should ensure that turning during treatments does not compromise flow through the cannulae.
Frequently children on ECMO for respiratory failure demonstrate a complete ‘whiteout’ of the lung fields on chest radiograph within the first day of ECMO support. During this time respiratory compliance is extremely low and little chest movement is seen or achieved during treatments. Children are often on ‘resting’ ventilation with low respiratory pressures and rates. During this time physiotherapy treatments may only consist of a quick assessment during the day, until it becomes apparent that the chest wall is beginning to move and air bronchograms are beginning to appear on chest radiograph. At this time it is appropriate to start more regular treatments in order to recruit functional airways and to facilitate early weaning from ECMO.
High-frequency oscillatory ventilation
High-frequency oscillatory ventilation (HFOV) theoretically provides gentle ventilatory support by employing very small tidal volumes and high respiratory rates (1–15 Hz) with high mean airway pressures to achieve adequate ventilation. It has been shown to be safe and effective in the treatment of respiratory failure in paediatric practice (Arnold 1996).
In theory, disconnecting infants from such ventilation for physiotherapy treatments, including manual inflation techniques, would contradict the principles of reducing volume loss, bulk flow of air and the shearing forces associated with such ventilation (Lindgren et al 2007). However many experienced physiotherapists find that unless children are disconnected for brief and effective treatments, these children are vulnerable to airway obstruction and atelectasis. The loss of volume after disconnection and suction is significant but transient (Tingay et al 2007). The key to successful physiotherapy treatments of children on HFOV lies in adequate preoxygenation, proper assessment and quick, competent and effective treatments, which do not permit de-recruitment within any treatment cycle.
Tracheal repair: tracheal stenosis and slide tracheoplasty
Subglottic stenosis occurs in some infants following prolonged intubation and leads to upper airway obstruction. It could be avoided by attention to tracheal tube placement and fixation and care with suction (Albert 1995). Acquired neonatal tracheobronchial stenosis (particularly in preterm infants) has a poor outcome. Stridor is often present and may respond to adrenaline via a nebulizer. In more severe cases a tracheostomy may be necessary until the airway has increased sufficiently in size to allow adequate ventilation. Some patients will also require surgical laryngotracheoplasty before successful decannulation of the tracheostomy can be achieved. More recently, primary repair of subglottic stenosis with laryngotracheal reconstruction has been successfully developed.
Children born with tracheal stenosis have had a high mortality. Now the development of various surgical tracheal reconstructions, replacements and slide tracheoplasty procedures has provided a new hope for such patients (Beierlein & Elliott 2006). ECMO may be required pre- or postoperatively to provide ventilatory support for patients undergoing critical tracheobronchial reconstruction. Manual hyperinflation following tracheal repair may be associated with greater risk of pneumothorax, because of the tracheal anastomosis. In addition, the tracheal anastomosis may extend distally to the tracheal tube and suction procedures should avoid traumatizing the site.
Some degree of tracheomalacia is often present and the fact that children are either paralysed or very well sedated in the early postoperative period means there is often air trapping and impaired airway clearance. It is thus essential that treatments are quick, competent and effective and include decompression, if necessary, of any trapped air. In the long term, such children may need follow-up in the community and may continue to have noisy breathing and problems with airway clearance.
Pulmonary hypertensive crisis
Pulmonary hypertensive crisis is described as an acute elevation of the pulmonary artery (PA) pressure (owing to contraction of the pulmonary arteriolar musculature), which restricts blood flow through the lungs. It is associated with a fall in left atrial pressure and a dramatic fall in cardiac output. PA pressure may approach or even exceed systemic pressure. It is seen in the presence of hypertrophic reactive arteriolar muscle in the lungs and is therefore common in those patients who have had significant left-to-right shunts (VSD, AVSD, truncus arteriosus). This phenomenon is a critical, life-threatening event and prevention of such an incident is desirable. Airway suction and chest physiotherapy have the potential both for precipitating a hypertensive crisis (by creating an imbalance in the pulmonary/systemic flow ratio) and for correcting an imbalance (caused by excess secretions). The partial pressures of blood oxygen and carbon dioxide relative to each other will determine the ratio of systemic–pulmonary blood flow. Low oxygen and high carbon dioxide will increase pulmonary vascular resistance and reduce pulmonary blood flow. High oxygen and low carbon dioxide cause an increase in pulmonary blood flow.
In children prone to pulmonary hypertensive crisis, treatment should be undertaken with great caution. Inspired oxygen should be increased during chest physiotherapy (but not too much if a child is dependent on a univentricular circulation) and treatment times kept to a minimum. Particular attention should be paid to oxygen saturation and the PA pressure in relation to systemic blood pressure. Often children will require a bolus of sedation before treatment and if there is already nitric oxide entrained in the ventilator circuit, care should be taken to ensure this supply is maintained during manual ventilation with a bag. Nitric oxide gas (NO) has a potent pulmonary vasodilatory effect and can be delivered directly to the lungs via the ventilator circuit, for the effective relief of pulmonary hypertension in infants and children. Very small doses are used to reduce pulmonary arterial pressures, while systemic blood pressure is not affected (doses larger than 80 parts per million can be toxic) (Cheifetz 2000, Haddad et al 2000, Kinsella & Abman 2000).
Delayed sternal closure
Occasionally postoperative closure of the sternum is impeded by pulmonary, myocardial or chest wall oedema (due either to prolonged bypass times or particularly complicated intracardiac repairs). If sternal closure is likely to constrict cardiopulmonary function, closure may be delayed for days or even weeks. During this period children are paralysed or very well sedated and are preferentially nursed in supine. They are therefore at much greater risk of pulmonary complications. However, if stable and if the sternum is stented (to keep its edges separate), the child can with care be quarter turned from supine. Manual hyperinflation is usually well tolerated and gentle posterior and posterolateral vibrations may be applied. When the sternum is finally closed, there is often short-term deterioration in respiratory function and it may be useful to suggest an increase in ventilatory support if treatments are to be undertaken soon after sternal closure or to delay physiotherapy treatments until respiratory function has stabilized again (Main et al 2001).
Phrenic nerve damage
Damage to the phrenic nerve is a well-documented complication of paediatric cardiac surgery (Mok et al 1991). It occurs most commonly where dissection is required close to the mediastinal vessels and pericardium with which its course is closely associated. The result may be difficulty weaning from mechanical ventilation or severe respiratory compromise once extubated. Paradoxical movement during inspiration may compress the ipsilateral lung and cause mediastinal shift to the contralateral side, causing a further loss in lung volume. Physiotherapy intervention will depend on clinical symptoms, but it is important that the patient is positioned head up to relieve the pressure from the abdominal viscera and reduce the work of breathing. It is sometimes necessary to surgically plicate the affected diaphragm.
Albert D. Management of suspected tracheobronchial stenosis in ventilated neonates. Archives of Disease in Childhood. 1995;72:1-2.
Almeida CC, Ribeiro JD, Almeida-Junior AA, Zeferino AM. Effect of expiratory flow increase technique on pulmonary function of infants on mechanical ventilation. Physiotherapy Research International. 2005;10(4):213-221.
Ambalavanan N, Carlo WA. Ventilatory strategies in the prevention and management of bronchopulmonary dysplasia. Seminars in Perinatology. 2006;30(4):192-199.
Armstrong DS, Grimwood K, Carzino R, et al. Lower respiratory tract infection and inflammation in infants with newly diagnosed cystic fibrosis. British Medical Journal. 1995;310:1571-1572.
Arnold JH. High frequency oscillatory ventilation: theory and practice in paediatric patients. Paediatric Anaesthesia. 1996;6:437-441.
Bagley CE, Gray PH, Tudehope DI, et al. Routine neonatal post-extubation chest physiotherapy: a randomized controlled trial. Journal of Paediatric and Child Health. 2005;41(11):592-597.
Baird PA, MacDonald EC. An epidemiologic study of congenital malformations of the anterior abdominal wall in more than half a million consecutive live births. American Journal of Human Genetics. 1982;34:517-521.
Balaguer A, Escribano J, Roque M, 2006 Infant position in neonates receiving mechanical ventilation. Cochrane Database of Systematic Reviews Issue 4 Art No: CD003668 DOI: 10.1002/14651858 CD003668.pub2
Balfour-Lynn IM, Abrahamson E, Cohen G, et al. BTS Guidelines for the management of pleural infection in children (on behalf of the Paediatric Pleural Diseases Subcommittee of the BTS Standards of Care Committee). Thorax. 2005;60:1-21.
Barry P, O’Callaghan C. Inhalational drug delivery from seven different spacer devices. Thorax. 1996;51:835-840.
Bauer M, Hoek G, Smit HA, et al. Air pollution and development of asthma, allergy and infections in a birth cohort. European Respiratory Journal. 2007;29:879-888.
Beby PJ, Henderson-Smart DJ, Lacey JL, Rieger I. Short and long term neurological outcomes following neonatal chest physiotherapy. Journal of Paediatric and Child Health. 1998;34:60-62.
Beierlein W, Elliott MJ. Variations in the technique of slide tracheoplasty to repair complex forms of long-segment congenital tracheal stenoses. Annals of Thoracic Surgery. 2006;82:1540-1542.
Berney S, Denehy L. A comparison of the effects of manual and ventilator hyperinflation on static lung compliance and sputum production in intubated and ventilated intensive care patients. Physiotherapy Research International. 2002;7(2):100-108.
Bhandari A, Panitch HB. Pulmonary outcomes in bronchopulmonary dysplasia. Seminars in Perinatology. 2006;30(4):219-226.
Bhuyan U, Peters AM, Gordon I, Helms P. Effect of posture on the distribution of pulmonary ventilation and perfusion in children and adults. Thorax. 1989;44:480-484.
Bingol Karakoc G, Yilmaz M, Sur S, et al. The effects of daily pulmonary rehabilitation program at home on childhood asthma. Allergology Immunopathology (Madr). 2000;28:12-14.
Birnkrant DJ. The assessment and management of the respiratory complications of pediatric neuromuscular diseases. Clinical Pediatrics (Philadelphia). 2002;41(5):301-308.
Blackwood B. Normal saline instillation with endotracheal suctioning: primum non nocere (first do no harm). Journal of Advanced Nursing. 1999;29:928-934.
Blumenthal I, Lealman GT. Effects of posture on gastro-oesophageal reflux in the newborn. Archives of Disease in Childhood. 1982;57:555-556.
Boucher RC. New concepts of the pathogenesis of cystic fibrosis lung disease. European Respiratory Journal. 2004;23:146-158.
Brackbill Y, Douthitt TC, West H. Psychophysiological effects in the neonate of prone versus supine placement. Journal of Pediatrics. 1973;82:82-83.
Brewer LA, Fosburg RG, Mulder GA, Verska JJ. Spinal cord complications following surgery for coarctation of the aorta - a study of 66 cases. Journal of Thoracic and Cardiovascular Surgery. 1972;64:368.
Brimioulle S, Moraine JJ, Norrenberg K, Kahn RJ. Effect of positioning and exercise on intracranial pressure in a neurosurgical intensive care unit. Physical Therapy. 1997;77:1682-1689.
British Thoracic Society Standards of Care Committee. Guidelines for the management of community acquired pneumonia in childhood. Thorax. 2002;57:1-24.
British Thoracic Society (BTS), Scottish Intercollegiate Guidelines Network (SIGN). British guideline on the management of asthma. Thorax. 2003;58(Suppl I):i1-i94.
Button BM, Heine RG, Catto-Smith AG, Phelan PD, Olinsky A. Postural drainage and gastro-oesophageal reflux in infants with cystic fibrosis. Archives of Disease in Childhood. 1997;76:148-150.
Button BM, Heine RG, Catto-Smith AG, et al. Chest physiotherapy in infants with cystic fibrosis: to tip or not? A five-year study. Pediatric Pulmonology. 2003;35(3):208-213.
Button BM, Heine RG, Catto-Smith AG, et al. Chest physiotherapy, gastro-oesophageal reflux, and arousal in infants with cystic fibrosis. Archives of Disease in Childhood. 2004;89(5):435-439.
Campbell AH, O’Connell JM, Wilson F. The effect of chest physiotherapy upon the FEV1 in chronic bronchitis. Medical Journal of Australia. 1975;1:33-35.
Carpenter A. Congenital malformation of the mitral valve. In: Stark J, De Leval M, editors. Surgery for congenital heart defects. 2nd edn. Philadelphia: WB Saunders; 1994:599-614.
Carrol N, Sly P. Exercise training as an adjunct to asthma management? Thorax. 1999;54:190-191.
Castenda AR. Tetralogy of Fallot. In: Stark J, De Leval M, editors. Surgery for congenital heart defects. 2nd edn. Philadelphia: WB Saunders; 1994:405-416.
Castle RA, Dunne CJ, Mok Q, et al. Accuracy of displayed values of tidal volume in the pediatric intensive care unit. Critical Care Medicine. 2002;30(11):2566-2574.
Chatwin M, Ross E, Hart N, et al. Cough augmentation with mechanical insufflation/exsufflation in patients with neuromuscular weakness. European Respiratory Journal. 2003;21(3):502-508.
Cheifetz IM. Inhaled nitric oxide: plenty of data, no consensus. Critical Care Medicine. 2000;28:902-903.
Choi JS, Jones AY. Effects of manual hyperinflation and suctioning in respiratory mechanics in mechanically ventilated patients with ventilator-associated pneumonia. Australian Journal of Physiotherapy. 2005;51:25-30.
Chulay M, Graeber GM. Efficacy of a hyperinflation and hyperoxygenation suctioning intervention. Heart and Lung. 1988;17:15-22.
Clark AP, Winslow EH, Tyler DO, White KM. Effects of endotracheal suctioning on mixed venous oxygen saturation and heart rate in critically ill adults. Heart and Lung. 1990;19:552-557.
Clarke RC, Kelly BE, Convery PN, Fee JP. Ventilatory characteristics in mechanically ventilated patients during manual hyperventilation for chest physiotherapy. Anaesthesia. 1999;54:936-940.
Cowan MJ, Gladwin MT, Shelhamer JH. Disorders of ciliary motility. American Journal of Medical Science. 2001;321:3-10.
Czarnik RE, Stone KS, Everhart CJ, Preusser BA. Differential effects of continuous versus intermittent suction on tracheal tissue. Heart and Lung. 1991;20:144-151.
Davies H, Kitchman R, Gordon G, Helms P. Regional ventilation in infancy. Reversal of the adult pattern. New England Journal of Medicine. 1985;313:1627-1628.
De Leval M. Ventricular septal defects. In: Stark J, De Leval M, editors. Surgery for congenital heart defects. 2nd edn. Philadelphia: WB Saunders; 1994:55-371.
De Leval M. Tricuspid valve. In: Stark J, De Leval M, editors. Surgery for congenital heart defects. 2nd edn. Philadelphia: WB Saunders; 1994:453-466., Ch 23
Deakers TW, Reynolds G, Stretton M, Newth CJ. Cuffed endotracheal tubes in pediatric intensive care. Journal of Pediatrics. 1994;125:57-62.
Demling RH, Riessen R. Respiratory failure after cerebral injury. Critical Care Medicine. 1993;1:440-446.
Depaepe A, Dolk A, Lechat MF. The epidemiology of tracheo-oesophageal fistula and oesophageal atresia in Europe. Archives of Disease in Childhood. 1993;68:743-748.
Dinwiddie R. Aspiration syndromes. In The diagnosis and management of paediatric respiratory disease, 2nd edn., New York: Churchill Livingstone; 1997:247-260.
Donn SM, Sinha SK. Invasive and non-invasive neonatal mechanical ventilation. Respiratory Care. 2003;48(4):426-441.
Ehrenkranz RA, Walsh MC, Vohr BR, et al. National Institutes of Child Health and Human Development Neonatal Research Network. Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116(6):1353-1360.
Elliott M, Hussey J. Paediatric cardiac surgery. In: Prasad SA, Hussey J, editors. Paediatric respiratory care. London: Chapman & Hall; 1995:122-141.
Feldman Z, Kanter MJ, Robertson CS, et al. Effect of head elevation on intracranial pressure and cerebral blood flow in head injured patients. Journal of Neurosurgery. 1992;59:206-211.
Ferkol T, Mitchison HM, O’Callaghan C et al. 2006 Current issues in the basic mechanisms, pathophysiology, diagnosis and management of primary ciliary dyskinesia. European Respiratory Monograph 37 11: 291-313
Flenady VJ, Gray PH. Chest physiotherapy for preventing morbidity in babies being extubated from mechanical ventilation. Cochrane Database of Systematic Reviews. (2):2002., article CD000283
Freed DH, Robertson CM, Sauve RS, et al. Intermediate-term outcomes of the arterial switch operation for transposition of great arteries in neonates: alive but well? Journal of Thoracic and Cardiovascular Surgery. 2006;132(4):845-852.
Gattinoni L, Caironi P, Cressoni M, et al. Lung recruitment in patients with the acute respiratory distress syndrome. New England Journal of Medicine. 2006;354(17):1775-1786.
Gattinoni L, Pesenti A, Bombino M, Pelosi P, Brazzi L. Role of extracorporeal circulation in adult respiratory distress syndrome management. New Horizons. 1993;1:603-612.
Gemma M, Tommasino C, Cerri M, et al. Intracranial effects of endotracheal suctioning in the acute phase of head injury. Journal of Neurological Anesthesiology. 2002;14:50-54.
Glass C, Grap MJ, Corley MC, Wallace D. Nurses’ ability to achieve hyperinflation and hyperoxygenation with a manual resuscitation bag during endotracheal suctioning. Heart and Lung. 1993;22:158-165.
Goodnough SK. The effects of oxygen and hyperinflation on arterial oxygen tension after endotracheal suctioning. Heart and Lung. 1985;14:11-17.
Gray PH, Flenady VJ, Blackwell L. Potential risks of chest physiotherapy in preterm infants. Journal of Pediatrics. 1999;135:131.
Greenough A. Lung maturation. In: Greenough A, Roberton NRC, Milner A, editors. Neonatal respiratory disorders. London: Arnold; 1996:13-26.
Greenough A, Kotecha S, Vrijlandt E, 2006 Bronchopulmonary dysplasia: current models and concepts. In: Frey U, Gerritsen JRespiratory diseases in infants and children. European Respiratory Monograph 11: 217-229
Gregson R, Shannon H, Main E, et al. The relationship between age and forces applied during chest physiotherapy in mechanically ventilated children. Physiotherapy. 2007;93:S517.
Gregson RK, Stocks J, Petley GW, et al. Simultaneous measurement of force and respiratory profiles during chest physiotherapy in ventilated children. Physiological Measurement. 2007;28:1017-1028.
Gremmo ML, Guenza MC. Positive expiratory pressure in the physiotherapeutic management of primary ciliary dyskinesia in the paediatric age. Monaldi Archives of Chest Disease. 1999;54:255-257.
Haddad E, Lowson SM, Johns RA, Rich GF. Use of inhaled nitric oxide perioperatively and in intensive care patients. Anesthesiology. 2000;92:1821-1825.
Hagler DA, Traver GA. Endotracheal saline and suction catheters: sources of lower airway contamination. American Journal of Critical Care. 1994;3:444-447.
Harding JE, Miles FK, Becroft DM, Allen BC, Knight DB. Chest physiotherapy may be associated with brain damage in extremely premature infants. Journal of Pediatrics. 1998;132:440-444.
Harshbarger SA, Hoffman LA, Zullo TG, Pinsky MR. Effects of a closed tracheal suction system on ventilatory and cardiovascular parameters. American Journal of Critical Care. 1992;1:57-61.
Heaf DP, Helms P, Gordon I, Turner HM. Postural effects on gas exchange in infants. New England Journal of Medicine. 1983;308(25):1505-1508.
Hislop A, Reid L. Development of the acinus in the human lung. Thorax. 1974;29:90-94.
Hislop A, Wigglesworth JS, Desai R. Alveolar development in the human fetus and infant. Early Human Development. 1986;13:1-11.
Hodge D, Chetcuti PAJ. RSV: management of the acute episode. Paediatric Respiratory Reviews. 2000;1:215-220.
Hoffman JIE, Kaplan S. The incidence of congenital heart disease. Journal of the American College of Cardiology. 2002;39(12):1890-1900.
Holloway E, Ram FSF. Breathing exercises for asthma. Cochrane Database of Systematic Reviews. (1):2004. : CD001277, pub 2
Hondras MA, Linde K, Jones AP. Manual therapy for asthma (Cochrane Review). In: The Cochrane Library. Oxford: Update Software; 2000:4.
Horiuchi K, Jordan D, Cohen D, Kemper MC, Weissman C. Insights into the increased oxygen demand during chest physiotherapy. Critical Care Medicine. 1997;25:1347-1351.
Huang SC, Wu ET, Chi NH et al. 2007 Perioperative extracorporeal membrane oxygenation support for critical pediatric airway surgery. European Journal of Pediatrics E-pub DOI 10.1007/500431-006-0390-y
Imle PC, Klemic N. Methods of airway clearance: coughing and suctioning. In: Mackenzie CF, Imle PC, Ciesla N, editors. Chest physiotherapy in the intensive care unit. Baltimore: Williams and Wilkins; 1989:153-187.
Inselman LS, Mellins RB. Growth and development of the lung. Journal of Pediatrics. 1981;98:1-15.
Inwald D, Roland M, Kuitert L, McKenzie S, Petros A. Oxygen for all in acute severe asthma. British Medical Journal. 2001;27:722-729.
Jaffe A, Bush A. Cystic fibrosis: review of the decade. Monaldi Archives of Chest Disease. 2001;56:240-247.
Jobe AH, Bancalari E. Bronchopulmonary dysplasia. American Journal of Respiratory and Critical Care Medicine. 2001;163:1723-1729.
Johnson AH, Peacock JL, Greenough A, et al. United Kingdom Oscillation Study Group. High frequency oscillatory ventilation for the prevention of chronic lung disease of prematurity. New England Journal of Medicine. 2002;347:633-642.
Keens TG, Ianuzzo CD. Development of fatigue-resistant muscle fibers in human ventilatory muscles. American Review of Respiratory Disease. 1979;119(2):139-141.
Kerem E, Yatsiv I, Goitein KJ. Effect of endotracheal suctioning on arterial blood gases in children. Intensive Care Medicine. 1990;16:95-99.
Khine HH, Corddry DH, Kettrick RG, et al. Comparison of cuffed and uncuffed endotracheal tubes in young children during general anesthesia. Anesthesiology. 1997;86:627-631.
Kim CS, Iglesias AJ, Sackner MA. Mucus clearance by 2-phase gas-liquid flow mechanism - asymmetric periodic-flow model. Journal of Applied Physiology. 1987;62(3):959-971.
King H, Booker PD. General principles of neonatal anaesthesia. Current Anaesthesia and Critical Care. 2004;15:302-308.
King M. Experimental models for studying mucociliary clearance. European Respiratory Journal. 1998;11(1):222-228.
Kinloch D. Instillation of normal saline during endotracheal suctioning: effects on mixed venous oxygen saturation. American Journal of Critical Care. 1999;8:231-240.
Kinsella JP, Abman SH. Clinical approach to inhaled nitric oxide therapy in the newborn with hypoxemia. Journal of Pediatrics. 2000;136:717-726.
Kleiber C, Krutzfield N, Rose EF. Acute histologic changes in the tracheobronchial tree associated with different suction catheter insertion techniques. Heart and Lung. 1988;17:10-14.
Knight DB, Bevan CJ, Harding JE, et al. Chest physiotherapy and porencephalic brain lesions in very preterm infants. Journal of Paediatric and Child Health. 2001;37(6):554-558.
Konno K, Mead J. Measurement of the separate volume changes of rib cage and abdomen during breathing. Journal of Applied Physiology. 1967;22:407-422.
Kotecha S. Lung growth: implications for the newborn infant. Archives of Disease in Childhood Fetal & Neonatal Edition. 2000;82:F69-F74.
Krastins I, Corey ML, McLeod A, et al. An evaluation of incentive spirometry in the management of pulmonary complications after cardiac surgery in a pediatric population. Critical Care Medicine. 1982;10:525-528.
Krause MF, Hoehn T. Chest physiotherapy in mechanically ventilated children: a review. Critical Care Medicine. 2000;28:1648-1651.
Kuo CY, Gerhardt T, Bolivar J, Claure N, Bancalari E. Effect of leak around the endotracheal tube on measurements of pulmonary compliance and resistance during mechanical ventilation: a lung model study. Pediatric Pulmonology. 1996;22:35-43.
Langman J. Medical embryology. Baltimore: Williams and Wilkins, 1977.
Lannefors L, Wollmer P. Mucus clearance with three chest physiotherapy regimes in cystic fibrosis: a comparison between postural drainage, PEP, and physical exercise. European Respiratory Journal. 1992;5:748-753.
Lindgren S, Odenstedt H, Olegard C, et al. Regional lung derecruitment after endotracheal suction during volume- or pressure-controlled ventilation: a study using electric impedance tomography. Intensive Care Medicine. 2007;33(1):172-180.
Lum S, Gustafsson P, Ljungberg H, et al. Early detection of cystic fibrosis lung disease: multiple-breath washout versus raised volume tests. Thorax. 2007;62(4):341-347.
MacNaughton PD, Evans TW. Pulmonary function in the intensive care unit. In: Hughes JMB, Pride NB, editors. Lung function tests: physiological principles and clinical applications. London: WB Saunders; 1999:185-199.
Main E, Castle R, Newham D, Stocks J. Respiratory physiotherapy vs. suction: the effects on respiratory function in ventilated infants and children. Intensive Care Medicine. 2004;30(6):1144-1151.
Main E, Castle R, Stocks J, et al. The influence of endotracheal tube leak on the assessment of respiratory function in ventilated children. Intensive Care Medicine. 2001;27(11):1788-1797.
Main E, Elliott MJ, Schindler M, Stocks J. Effect of delayed sternal closure after cardiac surgery on respiratory function in ventilated infants. Critical Care Medicine. 2001;29(9):1798-1802.
Marcus RJ, van der Walt JH, Pettifer RJ. Pulmonary volume recruitment restores pulmonary compliance and resistance in anaesthetized young children. Paediatric Anaesthesia. 2002;12:579-584.
Martinez TM, Llapur CJ, Williams TH, et al. High-resolution computed tomography imaging of airway disease in infants with cystic fibrosis. American Journal of Respiratory and Critical Care Medicine. 2005;172:1133-1138.
Matsumoto I, Araki H, Tsuda K, et al. Effects of swimming training on aerobic capacity and exercise induced bronchoconstriction in children with bronchial asthma. Thorax. 1999;54:196-201.
McCabe SM, Smeltzer SC. Comparison of tidal volumes obtained by one-handed and two-handed ventilation techniques. American Journal of Critical Care. 1993;2:467-473.
McCarren B, Alison JA, Herbert RD. Vibration and its effect on the respiratory system. Australian Journal of Physiotherapy. 2006;52(1):39-43.
McKelvie S. Endotracheal suctioning. Nursing in Critical Care. 1998;3:244-248.
Menkes HA, Traystman RJ. Collateral ventilation. American Review of Respiratory Disease. 1977;116:287-309.
Miske LJ, Hickey EM, Kolb SM, et al. Use of the mechanical in-exsufflator in pediatric patients with neuromuscular disease and impaired cough. Chest. 2004;105(3):741-747.
Mok Q, Ross-Russell R, Mulvey D, et al. Phrenic nerve injury in infants and children undergoing cardiac surgery. British Heart Journal. 1991;65(5):287-292.
Moore M, Little P, 2006 Humidified air inhalation for treating croup. Cochrane Database of Systematic Reviews, Issue 3. Art. No: CD002870. DOI: 10.1002/14651858.CD002870.pub2.
Morini F, Goldman A, Pierro A. Extracorporeal membrane oxygenation in infants with congenital diaghragmatic hernia. European Journal of Pediatric Surgery. 2006;16:385-391.
Muller NL, Bryan AC. Chest wall mechanics and respiratory muscles in infants. Pediatric Clinics of North America. 1979;26(3):503-516.
National Asthma Education and Prevention Programme (NAEPP) Expert Panel, 2002 Guidelines for the diagnosis and management of asthma - update on selected topics. NIH publication 02-5075, National Institutes of Health, Bethesda, MD
Neder JA, Nery LE, Silva AC, Cabral ALB, Fernandes ALG. Short-term effects of aerobic training in the clinical management of moderate to severe asthma in children. Thorax. 1999;54:202-206.
Ng GYT, da Silva O, Ohlsson A. Bronchodilation for the prevention and treatment of chronic lung disease in preterm infants. Cochrane Database of Systematic Reviews. 23, 2001. : CD003214
Nicholas KJ, Dhouibe MO, Marchall TG, Edmunds AT, Grant MB. Physiotherapy in patients with bronchiolitis. Physiotherapy. 1999;85(12):669-674.
Norwood WI, Jacobs ML. Hypoplastic left heart syndrome. In: Stark J, De Leval M, editors. Surgery for congenital heart defects. 2nd edn. Philadelphia: WB Saunders; 1994:587-598.
Ntoumenopoulos G. Indications for manual lung hyperinflation (MHI) in the mechanically ventilated patient with chronic obstructive pulmonary disease. Chronic Respiratory Disease. 2005;2(4):199-207.
O’Callaghan C. How to choose delivery devices for asthma. Archives of Disease in Childhood. 2000;82:185-191.
Ochs M, Nyengaard J, Jung A, et al. Number of alveoli in the human lung. American Journal of Respiratory and Critical Care Medicine. 2004;169:120-124.
Openshaw P, Edwards S, Helms P. Changes in rib cage geometry during childhood. Thorax. 1984;39:624-627.
Pang LM, Mellins RB. Neonatal cardiorespiratory physiology. Anesthesiology. 1975;43(2):171-196.
Panickar JR, Dodd SR, Smyth RL, Couriel JM. Trends in deaths from respiratory illness in children in England and Wales from, 1968-2000. Thorax. 2005;60:1035-1038.
Panitch HB. Airway clearance in children with neuromuscular weakness. Current Opinion in Pediatrics. 2006;18(3):277-281.
Pearson GA, Grant J, Field D, Sosnowski A, Firmin RK. Extracorporeal life support in paediatrics. Archives of Disease in Childhood. 1993;68:94.
Pénard-Morand C, Charpin D, Raherison C, et al. Long-term exposure to background air pollution related to respiratory and allergic health in schoolchildren. Clinical and Experimental Allergy. 2005;35(10):1279-1287.
Perrotta C, Ortiz Z, Roque M, 2005 Chest physiotherapy for acute bronchiolitis in paediatric patients between 0 and 24 months old. Cochrane Database of systematic Reviews, Issue 2. Art. No: CD004873. pub2
Phillips G. To tip or not to tip? Physiotherapy Research International. 1996;1(1):1-6.
Prasad SA, Main E. Routine airway clearance in asymptomatic infants and babies with cystic fibrosis: obligatory or obsolete? Physical Therapy Reviews. 2006;11:11-20.
Prasad SA, Tasker RC. Guidelines for physiotherapy management of critically ill children with acutely raised intracranial pressure. Physiotherapy. 1990;76(4):248-250.
Prasad SA, Tasker RC. Neurological intensive care. In: Prasad SA, Hussey J, editors. Paediatric respiratory care. London: Chapman & Hall; 1995:142-149.
Rabe KF, Adachi M, Lai CK, et al. Worldwide severity and control of asthma in children and adults: the global asthma insights and reality surveys. Journal of Allergy and Clinical Immunology. 2004;114:40-47.
Raguckas SE, Vandenbussche HL, Jacobs C, Klepser ME. Pertussis resurgence: diagnosis, treatment, prevention, and beyond. Pharmacotherapy. 2007;27(1):41-52.
Ram FSF, Robinson SM, Black PN, Picot J, 2005 Physical training for asthma. Cochrane Database of Systematic Reviews, Issue 4. Art. No.: CD001116. DOI: 10.1002/14651858.CD001116.pub2
Ranganathan SC, Dezateux C, Bush A, et al. Airway function in infants newly diagnosed with cystic fibrosis (London Collaborative Cystic Fibrosis Group). Lancet. 2001;358(9297):1964-1965.
Razmus I, Wilson D. Current trends in the development of sedation/ analgesia scales for the pediatric critical care patient. Pediatric Nursing. 2006;32:435-441.
Rehder K, Hatch DJ, Sessler AD, Fowler WS. The function of each lung of anesthetized and paralyzed man during mechanical ventilation. Anesthesiology. 1972;37(1):16-26.
Reid L., Lung growth in health and disease. British Journal of Diseases of the Chest. 1984;78:113-132.
Ridling DA, Martin LD, Bratton SL. Endotracheal suctioning with or without instillation of isotonic sodium chloride solution in critically ill children. American Journal of Critical Care. 2003;12(3):212-219.
Roberton NRC. Intensive care. In: Greenough A, Roberton NRC, Milner A, editors. Neonatal respiratory disorders. London: Arnold; 1996:174-195.
Roberts D, Dalziel S, 2006 Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database of Systematic Reviews 2006, Issue 3. Art. No: CD004454. DOI: 10.1002/14651858.CD004454.pub2
Russell K, Wiebe N, Saenz A 2004 Glucocorticoids for croup. Cochrane Database of Systematic Reviews Issue 1. Art. No: CD001955. DOI: 10.1002/14651858.CD001955.pub2
Ryan RM. A new look at bronchopulmonary dysplasia classification. Journal of Perinatology. 2006;26:207-209.
Salt A, Noble-Jameson G, Barnes ND, et al. Liver transplantation in 100 children: Cambridge and King’s College Hospital series. British Medical Journal. 1992;304:416-421.
Savian C, Paratz J, Davies A. Comparison of the effectiveness of manual and ventilator hyperinflation at different levels of positive end-expiratory pressure in artificially ventilated and intubated intensive care patients. Heart Lung. 2006;35(5):334-341.
Schreuder FM, Jones UF. The effect of saline instillation on sputum yield and oxygen saturation measurement in adult intubated patients: single subject design. Physiotherapy. 2004;90:109.
Schultz CM, Digeronimo RJ, Yoder BA, Congenital Diaphragmatic Hernia Study Group. Congenital diaphragmatic hernia: a simplified postnatal predictor of outcome. Journal of Pediatric Surgery. 2007;42:510-516.
Scottish Intercollegiate Guidelines Network. Bronchiolitis in children: a national clinical guideline. Edinburgh: Scottish Intercollegiate Guidelines Network, 2006.
Seear M, Wensley D, West N. How accurate is the diagnosis of exercise induced asthma among Vancouver schoolchildren? Archives of Disease in Childhood. 2005;90:898-902.
Shah AR, Kurth CD, Gwiazdowski SG, Chance B, Delivoria-Papadopoulos M. Fluctuations in cerebral oxygenation and blood volume during endotracheal suctioning in premature infants. Journal of Pediatrics. 1992;120:769-774.
Shorten DR, Byrne PJ, Jones RL. Infant responses to saline instillations and endotracheal suctioning. Journal of Obstetric, Gynecological and Neonatal Nursing. 1991;20:464-469.
Singer M, Vermaat J, Hall G, Latter G, Patel M. Hemodynamic effects of manual hyperinflation in critically ill mechanically ventilated patients. Chest. 1994;106:1182-1187.
Smith NP, Jesudason EC, Featherstone NC, et al. Recent advances in congenital diaghragmatic hernia. Archives of Disease in Childhood. 2005;90:426-428.
Sole ML, Byers JF, Ludy JE, et al. A multisite survey of suctioning techniques and airway management practices. American Journal of Critical Care. 2003;12(3):220-230.
Southall DP, Samuels MP. Reducing risks in the sudden infant death syndrome. British Medical Journal. 1992;304:260-265.
Stark J, Gallivan S, Lovegrove J, et al. Mortality rates after surgery for congenital heart defects in children and surgeons’ performance. Lancet. 2000;355(9208):1004-1007.
Stevens TP, Blennow M, Soll RF, 2004 Early surfactant administration with brief ventilation vs selective surfactant and continued mechanical ventilation for preterm infants with or at risk for respiratory distress syndrome. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD003063. DOI: 10.1002/14651858.CD003063.pub2
Stiller K. Physiotherapy in intensive care: towards an evidence-based practice. Chest. 2000;118:1801-1813.
Stone KS. Ventilator versus manual resuscitation bag as the method for delivering hyperoxygenation before endotracheal suctioning. AACN: Clinical Issues in Critical Care Nursing. 1990;1:289-299.
Stone KS, Talaganis SA, Preusser B, Gonyon DS. Effect of lung hyperinflation and endotracheal suctioning on heart rate and rhythm in patients after coronary artery bypass graft surgery. Heart and Lung. 1991;20:443-450.
Stone KS, Turner B. Endotracheal suctioning. Annual Review of Nursing Research. 1989;7:27-49.
Sutton PP, Lopezvidriero MT, Pavia D, et al. Assessment of percussion, vibratory-shaking and breathing exercises in chest physiotherapy. European Journal of Respiratory Diseases. 1985;66:147-152.
Swaniker F, Kolla S, Moler F, et al. Extracorporeal life support outcome for 128 pediatric patients with respiratory failure. Journal of Pediatric Surgery. 2000;35:197-202.
Tannenbaum E, Prasad SA, Dinwiddie R, Main E. Chest physiotherapy during anaesthesia for children with cystic fibrosis: effects on respiratory function. Pediatric Pulmonology. 2007;42(12):1152-1158.
Tasker RC. Neurological critical care. Current Opinion in Pediatrics. 2000;12(3):222-226.
Tasker RC. Neurocritical care and traumatic brain injury. Indian Journal of Paediatrics. 2001;68:257-266.
Tasker RC, Morris KP, Forsyth RJ, et al. Severe head injury in children: emergency access to neurosurgery in the United Kingdom. Emergency Medicine Journal. 2006;23(7):519-522.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness: a practical scale. Lancet. 1974;2:81-84.
Thoresen M, Cavan F, Whitelaw A. Effect of tilting on oxygenation in newborn infants. Archives of Disease in Childhood. 1988;63:315-317.
Tingay DG, Copnell B, Mills JF, et al. Effects of open endotracheal suction on lung volume in infants receiving HFOV. Intensive Care Medicine. 2007;33(4):689-693.
Toelle BG, Marks GB. The ebb and flow of asthma. Thorax. 2005;60(2):87-88.
Tudehope DI, Bagley C. Techniques of physiotherapy in intubated babies with RDS. Australian Paediatric Journal. 1980;16:226-228.
UK Collaborative ECMO Trial Group. UK collaborative randomised trial of neonatal extracorporeal membrane oxygenation. Lancet. 1996;348:75-81.
van der Schans CP, Postma DS, Koeter GH, Rubin BK. Physiotherapy and bronchial mucus transport. European Respiratory Journal. 1999;13(6):1477-1486.
Vaughan RS, Menke JA, Giacoia GP. Pneumothorax: a complication of endotracheal suctioning. Journal of Pediatrics. 1978;92:633-634.
Vianello A, Corrado A, Arcaro G, et al. Mechanical insufflation-exsufflation improves outcomes for neuromuscular disease patients with respiratory tract infections. American Journal of Physical Medicine and Rehabilitation. 2005;84(2):83-88.
Vincon C. Potential risks of chest physiotherapy in preterm infants. Journal of Pediatrics. 1999;135:131-132.
Wanner A. Does chest physical therapy move airway secretions? American Review of Respiratory Disease. 1984;130:701-702.
Webb MSC, Martin JA, Cartlidge PHT, Ng YK, Wright NA. Chest physiotherapy in acute bronchiolitis. Archives of Disease in Childhood. 1985;6:1078-1079.
Wenstrom KD, Weiner CP, Hanson JW. A five-year statewide experience with congenital diaphragmatic hernia. American Journal of Obstetrics and Gynecology. 1991;165:838-842.
Wollmer P, Ursing K, Midgren B, Eriksson L. Inefficiency of chest percussion in the physical therapy of chronic bronchitis. European Journal of Respiratory Disease. 1985;66:233-239.
Wong WP, Paratz JD, Wilson K, Burns YR. Hemodynamic and ventilatory effects of manual respiratory physiotherapy techniques of chest clapping, vibration, and shaking in an animal model. Journal of Applied Physiology. 2003;95(3):991-998.
Wood CJ. Endotracheal suctioning: a literature review. Intensive Critical Care Nursing. 1998;14:124-136.
Zack MB, Pontoppidan H, Kazemi H. The effect of lateral positions on gas exchange in pulmonary disease: a prospective evaluation. American Review of Respiratory Disease. 1974;110:49-55.
Zahka KG, Patel CR. Congenital defects. In Fanaroff A, Martin RJ, editors: Neonatal-perinatal medicine: diseases of the fetus and infant, 7th edn., St Louis: Mosby, 2002.