Chapter 15 Thoracic organ transplantation
INTRODUCTION
Thoracic organ transplantation is a well-established treatment for patients with end-stage heart and lung disease. It aims to improve the quality of life and survival of those patients who are already managed optimally, often with maximal medical therapy. Physiotherapists are key members of the transplant team, providing expertise in the physical and functional assessment, respiratory management and rehabilitation of patients both before and after surgery.
This chapter discusses the physiotherapy management before and after heart, heart-lung and lung transplantation. The historical context, assessment and selection of appropriate candidates, surgical procedures, key concepts of immunosuppression, rejection, infection and denervation are also considered. Issues specific to paediatric patients are identified.
HISTORY
The first human thoracic organ transplants were performed in the 1960s. In 1963 Dr James Hardy (Jackson, USA) performed a single lung transplant in a lung cancer sufferer. Controversially, the following year he transplanted a chimpanzee heart into a human recipient. In 1967 Dr Christiaan Barnard (Cape Town, South Africa) performed the first human heart transplant. More than a hundred heart transplants were performed in 1968–69, but almost all patients died within 60 days.
The 1980s heralded a new era in thoracic organ transplantation with the first long-term survivors of heart-lung (Reitz et al 1982), single lung (Cooper et al 1987) and double lung (Patterson et al 1988) transplantation. The advent of ciclosporin (Borel 1980) as the principal immunosuppressant, with a greater ability to prevent acute rejection, was considered pivotal to improved survival outcomes.
From the late 1980s to the mid 1990s there was steady growth in both the overall numbers of transplants performed and the number of centres performing them. These numbers have now stabilized and reflect the ongoing shortage of donor organs. Internationally, over 4000 heart transplants, 60 heart-lung transplants and 1600 lung transplants are performed in adults each year (Taylor et al 2005, Trulock et al 2005). The number of paediatric transplants is much smaller, with approximately 350 heart transplants, 10 heart-lung transplants, and 80 lung transplants performed (Boucek et al 2005).
Over the last 30 years, significant advances have been made in all aspects of the care of thoracic organ transplant recipients. There have been improvements in operative techniques, organ preservation and cross matching. New, less toxic immunosuppressants have been developed. There is a greater understanding of immunology and a greater ability to bridge to transplant with mechanical support devices. Current survival outcomes for patients receiving heart transplantation are more favourable than for lung transplantation with 81%, 67% and 48% survival at 1, 5 and 10 years compared with 76%, 49% and 24%, respectively (Taylor et al 2005, Trulock et al 2005). Outcomes in children are similar to those in adults.
SELECTION OF CANDIDATES
A variety of criteria have been developed to identify patients who will live longer and function better with transplantation than with medical therapy. Through a rigorous evaluation process, the transplant team assesses the patient’s severity of organ failure, screen for comorbidities that may negatively affect survival, and assess psychosocial variables necessary for successful outcomes following transplantation.
INDICATIONS FOR TRANSPLANTATION
Thoracic organ transplantation is indicated in patients with various end-stage diseases where survival is limited and quality of life poor (Table 15.1).
Table 15.1 Indications for thoracic organ transplantation
| Heart | |
| Heart-lung | |
| Single lung | |
| Bilateral lung |
Heart transplant
The distribution of indications for cardiac transplantation has not changed significantly over the last 10 years (Taylor et al 2005). The most common indications in adults continue to be ischaemic and non-ischaemic heart failure (45% each). Valvular disease (3–4%), adult congenital heart disease (2%) and allograft failure requiring retransplantation (2%) make up the remaining indications. What has changed is the increasing number of patients bridged to transplant using intravenous inotropic support (48%) and some type of mechanical circulatory support (21%) with a left ventricular assist device (LVAD).
Lung transplant
The main indications of adult lung transplant are chronic obstructive pulmonary disease (38%), idiopathic pulmonary fibrosis (17%), cystic fibrosis (17%) and a 1-antitrypsin deficiency emphysema (9%). The most common indications in childhood differ by age group. In infants, congenital heart disease is the most common indication (47%). In older children, cystic fibrosis (72%), primary pulmonary hypertension (10%) and retransplantation (6%) are the main indications. Interstitial lung disease, pulmonary vascular disease and bronchiolitis obliterans less commonly lead to transplantation in children (Boucek et al 2005). Lung transplant is rarely appropriate in critically ill patients in desperate clinical situations (i.e. intubated in the intensive care unit).
Heart-lung transplant
Primary pulmonary hypertension and pulmonary hypertension associated with Eisenmenger’s syndrome/congenital heart disease have been the main indications for heart-lung transplantation in adults and children (see Table 15.1).
Contraindications to transplantation
Patients with comorbidities that may seriously compromise the outcome of transplantation are excluded. There are few absolute contraindications to thoracic organ transplantation, and consideration of relative contraindications is usually made on an individual basis (Box 15.1).
ASSESSMENT
Potential recipients are assessed by an experienced multidisciplinary team at a transplant centre (Box 15.2). This process involves extensive physiological, functional and psychological assessment in order to:
 evaluate the severity of cardiac and/or pulmonary dysfunction (e.g. pulmonary function testing, per-fusion scan, echocardiography, gated blood pool scan)
evaluate the severity of cardiac and/or pulmonary dysfunction (e.g. pulmonary function testing, per-fusion scan, echocardiography, gated blood pool scan) identify previous exposure to potentially complicating infection: cytomegalovirus (CMV), toxoplasmosis, hepatitis B, hepatitis C, methicillin-resistant Staphylococcus aureus (MRSA), Epstein-Barr virus (EBV) and human immunodeficiency virus (HIV)
identify previous exposure to potentially complicating infection: cytomegalovirus (CMV), toxoplasmosis, hepatitis B, hepatitis C, methicillin-resistant Staphylococcus aureus (MRSA), Epstein-Barr virus (EBV) and human immunodeficiency virus (HIV)The assessment is usually performed as an inpatient over a 2–3 day period. Once the evaluation has been compiled, all members of the transplant team meet to discuss the findings and come to a consensus regarding the patient’s appropriateness for listing.
Physiotherapy assessment
Physiotherapy assessment of the potential transplant candidate is similar to that of any cardiorespiratory medical or surgical patient. It focuses on the impact of cardiac, respiratory and musculoskeletal limitations on exercise, functional capacity and social performance. The medical history and results of relevant investigations (e.g. imaging, arterial blood gases, lung function, angiography) should be reviewed before seeing the patient so that the patient’s unique pathophysiology and clinical status is understood.
Musculoskeletal assessment
The screening assessment should include:
A more in-depth assessment is required if musculoskeletal abnormalities are found. Structural and postural thoracic kyphosis, shoulder pathologies (rotator cuff impingement syndromes), shortened calf, hamstrings and iliopsoas muscles and reduced muscle bulk are commonly seen in thoracic transplant candidates.
Exercise capacity
 six-minute walk test (6MWT)
six-minute walk test (6MWT)
—distance walked, response to exercise (i.e. SpO2, HR, BP, symptoms), limitation to exercise
—may be used as a predictor of survival in cardiac disease and also in some diagnostic groups (e.g. cystic fibrosis, idiopathic pulmonary fibrosis, pulmonary arterial hypertension)
—general guidelines: In adults a 6MWT distance <300–400 m is suggested as appropriate for listing for both heart and lung transplantation (Kadikar et al 1997, Raul et al 1998), whereas in children desaturation on 6MWT is a better predictor of survival (Aurora et al 2000).
 maximal exercise testing
maximal exercise testing
—VO2peak <14 ml kg-1 min-1 is an indication for listing for cardiac transplant (Mancini et al 1991). There is little evidence that this test predicts prognosis in pulmonary disease.
SURGICAL PROCEDURES
Heart transplantation
Orthotopic heart transplantation
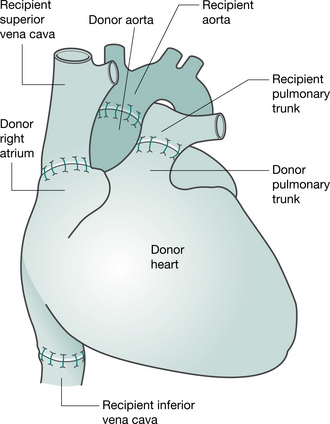
Preparation of the heart (and heart-lung) transplant recipient is similar to that for any patient undergoing cardiac surgery (anaesthesia, median sternotomy and cardiopulmonary bypass). When the donor heart is present in the recipient theatre and has passed a final inspection, the recipient heart is removed, by incising the atria, pulmonary artery and aorta. The posterior walls of both atria, including the sinoatrial node, are left intact. The donor heart is sutured in place. The anastomoses join the recipient and donor atria, the pulmonary arteries and the aortas (Keogh et al 1986) (Fig. 15.1). More recently, the bicaval anastomosis technique has been carried out at some centres. Potential advantages of this technique, which maintains the anatomic integrity of the donor right atrium, include a reduction in the incidence and severity of tricuspid regurgitation, preservation of right atrial function and facilitation of restoration of sinus rhythm (Morgan & Edwards 2005).
Heterotopic heart transplantation
Heterotopic transplantation is rarely performed, but is occasionally indicated in patients with cardiac dysfunction resulting in severe pulmonary hypertension. In this ‘piggyback’ procedure the recipient heart is left in place and the donor heart is positioned in the right chest. The donor heart is connected to the recipient’s in parallel by anastomoses made between the two hearts at the atria, pulmonary arteries and aortas (Newcomb et al 2004). Both hearts contribute to the cardiac output and share the work required to overcome the increased pulmonary pressures (Newcomb et al 2004, Novitzky et al 1983). Inherent problems with this procedure include pulmonary compression of the recipient’s right lung, difficulty obtaining endomyocardial biopsy and need for anticoagulation.
Lung transplantation
Single lung transplantation
In single lung transplantation, the native lung with the poorest pulmonary function according to the preoperative quantitative perfusion scan is excised. If both the lungs have similar function, then the right side is preferred as surgical exposure and the institution of cardiopulmonary bypass is easier.
The operation is performed via a posterolateral thoracotomy through the fourth or fifth intercostal space. The recipient’s lung is removed and the donor lung positioned in the chest. The bronchial anastomosis is performed first, followed by the pulmonary artery anastomosis. After completion of these anastomoses, the lung is reinflated and perfusion is re-established. Following resumption of ventilation to the donor lung, haemostasis is obtained, two intercostal catheters are placed (apical and basal) and the chest is closed. Fol-lowing reintubation with a single lumen tube, flexible bronchoscopy is performed to inspect the bronchial anastomosis and clear the airway of blood or residual secretions. Cardiopulmonary bypass is rarely needed during this operation.
Double lung/bilateral sequential lung transplantation
The most commonly performed double lung transplantation (DLT) procedure is bilateral sequential lung transplantation. The early experiences of double lung transplantation involved the implantation ‘en bloc’ of both lungs via a median sternotomy, utilizing an omental wrap to secure the tracheal anastomosis (Patterson et al 1988). In an effort to avoid the high incidence of airway complications associated with the original procedure, the technique of bilateral sequential lung transplantation via bilateral anterolateral thoracotomies through the fourth or fifth intercostal space connected with a transverse sternotomy or ‘clamshell incision’ is now preferred. Mobilization and pneumonectomy of the native lung and the implantation of the lung graft are conducted in the same manner as described for single lung transplantation.
Heart-lung transplantation
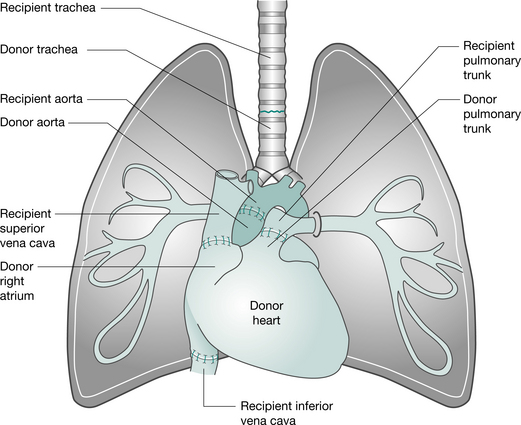
This operation can be performed via a median sternotomy or a clamshell incision. Following the institution of cardiopulmonary bypass, the heart and lungs are excised separately, allowing identification and protection of the phrenic, recurrent laryngeal and vagus nerves. The heart is removed, leaving the posterior wall of the right atrium. The left and then right lungs are removed (following stapling of the bronchi, to minimize the risk of contaminating the area) and the trachea is divided. The donor heart-lung block is implanted, starting with the tracheal, then the atrial and aortic anastomoses (Fig. 15.2). Ventilation is established (ensuring the patency of the airway anastomosis) and the heart resuscitated (Jamieson et al 1984). To improve donor availability, recipients with primary lung disease who receive heart-lung blocks (e.g. patients with cystic fibrosis) may be asked to donate their hearts to a cardiac patient. This is termed the ‘domino’ procedure.
Other lung techniques
The scarcity of donor lungs, especially for small and paediatric recipients, has led to the development of operations that allow larger lungs to be downsized. These techniques are not considered standard practice.
The split-lung technique (Couetil et al 1997) utilizes individual lobes from the donor. It may be indicated if there is localized pathology in one lobe of the donor lung or if the donor organ is larger than expected.
Living donor lobar lung transplantation (Date et al 2003, Starnes et al 1999) involves two donors (usually relatives) each donating a single lobe (usually lower lobe) for bilateral lung transplantation. The recipient is usually critically ill and cannot wait for cadaveric transplantation. It is most often performed in adolescents or young adults with cystic fibrosis.
KEY CONCEPTS
Organ donation
Organ donation for transplantation is performed in the setting of brain death. Brain death is defined as a complete and irreversible cessation of brain activity. The main causes are severe head injury from physical trauma, often from road traffic accidents and from subarachnoid haemorrhage. In most countries, consent from family members or next-of-kin is required. It is normal practice for consent to be sought even if the brain-dead individual had expressed the wish to donate. In some countries (e.g. Spain, Belgium, Poland, France) potential donors are presumed to have given consent, although some jurisdictions allow opting out from the system. Once consent is obtained, the non-living donor is kept on ventilatory support until the organs have been surgically removed. The donor is given expert medical and nursing care to optimize organ performance. Physiotherapists sometimes assist in the removal of retained lung secretions and help to optimize ventilation (Gabbay et al 1999).
A very small number of living and non-heart beating donors are used for lung transplantation internationally. Living donor lobar transplantation remains a second-line treatment due to the inevitable risk of lobectomy to the donors. Non-heart-beating donors are individuals who do not meet brain death criteria but for whom further medical intervention is futile. Organs can be rapidly retrieved after certification of death following withdrawal of support and asystole.
Timing of organ retrieval and implantation is important and necessitates a high level of coordination between the donor and recipient transplant teams. Organ procurement occurs at the hospital where the donor is managed. The organ is kept in preservation solution while it is transported to the transplant centre for implantation into the selected recipient. Most cardiac teams aim for an ischaemic time of less than 4 hours, from the time of cross-clamping the aorta in the donor to reperfusing the organ in the recipient. Lung teams aim for less than 8 hours. Longer ischaemic times are associated with poorer early graft function (Del Rizzo et al 1999, Thabut et al 2005).
The recipient team selects the appropriate recipient based upon:
Organ allocation systems vary worldwide. In Australia and the UK, severity of illness and medical urgency are taken into account. In the United States of America, the system for allocation of heart and lungs has recently been changed, in an attempt to direct scarce organs to individuals who will derive the most benefit from them.
Immunosuppression and rejection
Rejection is a specific immune response to the donor tissue (allograft) and is part of the normal host defence system against foreign antigens. The response can occur by humoral (B lymphocyte) or cell-mediated (T lymphocyte) immune mechanisms. Immunosuppression is required to manage rejection.
The majority of thoracic organ transplant recipients remain on two or three lifelong maintenance immunosuppressive agents. Typically, one of these will be a calcineurin inhibitor (e.g. ciclosporin, tacrolimus), one will be a cell-cycle inhibitor (e.g. mycophenolate mofetil, azathioprine), and the third will be a glucocorticoid (e.g. prednisone). Immunosuppression protocols vary widely from centre to centre.
Immunosuppressants have a number of specific side effects, which are listed in Table 15.2. It is important that physiotherapists working with thoracic organ transplant recipients are familiar with these side effects. Many agents impact on the musculoskeletal system and may cause bone morbidities such as avascular necrosis, osteoporosis and reduced tissue healing. Side effects of some agents affect the patient’s ability to participate in exercise training (e.g. hypertension, nausea) or have practical implications (e.g. fine hand tremor affecting writing ability and difficulty fitting into footwear due to fluid retention).
Table 15.2 Immunosuppression and side effects
| Immunosuppressant | Side effects |
|---|---|
| Calcineurin inhibitors | |
| Ciclosporin | Nephrotoxicity, hepatic dysfunction, hirsutism, tremor, hypertension, susceptibility to malignant neoplasms |
| Tacrolimus | Hypertension, diabetes, nephrotoxicity, increased risk of malignancy, neurotoxicity, tremor, headache, diarrhoea, nausea |
| Cell cycle inhibitors | |
| Azathioprine | Bone marrow suppression, hepatic dysfunction, nausea, anorexia |
| Mycophenolate mofetil (MMF) | Diarrhoea, bone marrow suppression, opportunistic infection (especially invasive CMV) |
| Sirolimus | Diarrhoea, haematological disturbances, hyperlipidaemia, arthralgia, opportunistic infection (PCP, CMV), epistaxis, rash, abnormal healing, hepatic dysfunction, renal impairment, increases malignancy especially skin, bone necrosis |
| Everolimus | Increased malignancy especially skin, haematological disturbances, opportunistic infection (PCP, CMV), gastrointestinal upset, hepatic dysfunction, hyperlipidaemia, myalgia, hypertension |
| Corticosteroids | |
| Prednisolone | Sodium and fluid retention, hypokalaemia, hyperglycaemia, gastrointestinal ulceration, osteoporosis, skin fragility, increased appetite, mood changes |
| Methylprednisolone | As above |
PCP, Pneumocystis carinii pneumonia; CMV, cytomegalovirus
Immunosuppressive therapy must be carefully balanced to prevent rejection without the development of serious adverse effects of the immunotherapy itself. Excessive immunosuppression increases the risk of infection, kidney and liver dysfunction and malignancies. Inadequate immunosuppression may result in rejection.
Graft rejection can be divided into three subcategories: hyperacute, acute and chronic rejection.
Hyperacute rejection
Hyperacute, or primary graft failure, generally occurs within the first 72 hours postoperatively. Primary graft failure generally results from ischemia-reperfusion injury. The presenting features of the heart transplant recipient may be similar to a patient in cardiogenic shock, while the features of the lung transplant recipient may be similar to a patient with acute respiratory distress syndrome. Both may require total mechanical support in the form of extracorporeal membrane oxygenation (ECMO).
Acute rejection
Acute rejection (Fig. 15.3) is characterized by a host T-cell response toward the transplanted organ and is a common complication in the first weeks to months following transplant. The diagnosis is based on both clinical and histological criteria. Often, mild to moderate rejection is not associated with any reliable signs or symptoms.
In lung transplant recipients, rejection often mimics an upper respiratory tract infection or bronchitis. The spectrum of clinical features is non-specific and includes dyspnoea, fever, non-productive cough, hypoxaemia, or malaise. The chest radiograph may show new opacifications or pleural effusions. There may be a drop in lung function. As it is difficult to differentiate rejection and infection on the basis of clinical, radiological or physiological criteria, bronchoscopy with bronchoalveolar lavage (BAL) and transbronchial biopsy (TBB) are often required. Acute lung rejection is graded from A0 (none) to A4 (severe) based on a standardized grading system (Yousem et al 1996). In addition, the presence of airway inflammation is graded from B0 (no airway inflammation) to B4 (severe airway inflammation). Grade A2 and above is generally treated with augmented immunosuppression, usually intravenous pulsed methylprednisolone. A1 rejection is usually not treated.
Heart transplant recipients with moderate to severe rejection may present with arrhythmias, hypotension, fever, increased weight/fluid retention, malaise or dysp-noea. Routine endomyocardial biopsies via right heart catheter are the mainstay for monitoring rejection in these patients. Criteria for grading these biopsies have recently been modified, but the principles of grading and treatment are similar to those for lung transplantation.
Chronic graft impairment
Chronic graft impairment, sometimes called chronic rejection, manifests as airway disease in lung transplant recipients, and cardiac allograft vasculopathy in cardiac recipients. In both cases the causes are not completely understood, but are likely to be a result of scarring following acute rejection, infection, and other triggers, combined with chronic low-grade inflammation.
In the lung, chronic graft dysfunction is characterized histologically by bronchiolitis obliterans and physiologically by airflow limitation (Cooper et al 1993). High-resolution computed tomography typically shows traction bronchiectasis, decreased peripheral vascular markings and gas trapping (Moorish et al 1991). Transbronchial biopsy rarely produces enough tissue to confirm the diagnosis, so the surrogate diagnosis of bronchiolitis obliterans syndrome (BOS) is used instead. BOS is defined by an irreversible fall in lung function when other causes have been excluded, and is graded from BOS0 to BOS3.
Until recently, BOS was described as untreatable and irreversible. However, it is now believed that some patients who were previously diagnosed with BOS had some degree of reversible airway dysfunction caused by non-immune insults such as gastro-oesophageal reflux or airway infection. Specifically, a number of centres have now reported a very high prevalence of gastro-oesophageal reflux in transplant recipients, with a gratifying improvement in outcomes following surgical treatment of this reflux (Button et al 2005, Cantu et al 2004, Davis et al 2003). In addition, reports from more than one centre have described how low-dose macrolide therapy can improve graft function in some patients with BOS. The mechanism is not fully understood, but the observation that patients who respond are usually those who have evidence of neutrophilic airway inflammation suggests that the anti-inflammatory properties of macrolides are responsible for the improvement (Crowley & Egan 2005).
Patients who have established BOS, which does not respond to such measures, are of considerable concern, as by definition there is no effective treatment. Some of these patients stabilize, and can continue for many years with limited lung function. Others develop steadily worsening graft dysfunction and progress to respiratory failure. BOS is the most common cause of late death following lung transplantation and is the main reason why improvements in long-term outcome have been slow.
Cardiac allograft vasculopathy is the main cause of late graft failure and death in heart transplant recipients (Taylor et al 2005). The coronary arteries develop a progressive concentric hyperplasia leading to vessel lumen obliteration. This can develop as early as 3 months after transplantation and is detected by angiography or increasingly, by intravascular ultrasound. The cause of the process is unclear, though long graft ischaemic time and recurrent rejection episodes have been identified as contributing factors. A recent study from a paediatric centre suggests that chronic low-grade CMV infection or reactivation may be a major cause (Hussain et al 2006). Treatment options are limited, although statin therapy may be of some benefit (Grigioni et al 2006) and is therefore now prescribed to all heart transplant recipients, including those with no evidence of vasculopathy. Angioplasty may be undertaken for diseased vessels if there is a discrete lesion, but is considered palliative and has not been shown to alter the natural history of cardiac allograft vasculopathy (Jonas et al 2006). The only definitive therapy is retransplantation.
Infection
Chronic immunosuppression renders transplant recipients more susceptible to infections (Box 15.3). The frequency of bacterial infections tends to peak within the first 3 months, while immunosuppressive therapy is often at a maximum level. The most frequent sites of infection are the lungs and blood. Infection is the major cause of morbidity and mortality in both the early and late post-transplant periods.
Denervation
Cardiac denervation
The pathophysiology of the transplanted heart is unique. The denervation of the organ makes it dependent on its intrinsic rate. It will therefore have a higher than normal resting heart rate secondary to the lack of inhibitory vagal influence. An alteration in the physiological response to exercise is also seen. In the normally innervated heart it is predominantly changes in heart rate that account for the increase in cardiac output in response to dynamic exercise. In contrast, the denervated heart increases cardiac output by increasing stroke volume (based on the Frank-Starling mechanism). The heart rate rises more gradually following the start of exercise, does not reach a similar peak and slows more gradually once exercise is stopped. This pattern of heart rate response is primarily the result of changing levels of circulating catecholamines (i.e. adrenaline and noradrenaline). In addition, transmission of ischaemic pain is prevented in the denervated heart (Weber 1990). Although partial reinnervation occurs in some recipients, the majority of patients will not experience anginal symptoms and should be advised against unsupervised exercise at high intensities for long periods. This is especially important if angiography indicates the presence of coronary artery disease (Hosenpud 1999, Kavanagh 1996).
Lung denervation
Similarly, lung and heart-lung transplantation involves denervation of the lungs below the airway anastomosis, with associated loss of all pulmonary innervation except post-ganglionic efferent nerves. Although the laryngeal expiration reflex is preserved, the cough response is severely impaired (Higenbottam et al 1989). In addition, ciliary function in the graft is abnormal, possibly because of the loss of the normal blood supply. Taken together, these two defects predispose the recipient to retained airway secretions and lower respiratory tract infection, particularly in the early postoperative period. Some clinicians believe that loss of afferent feedback results in loss of the Hering-Breuer reflex. This may lead to an erratic breathing pattern soon after extubation in some patients (Mattila et al 1987).
PREOPERATIVE REHABILITATION
Pre-transplant rehabilitation for heart and lung transplant candidates closely follows cardiac and pulmonary rehabilitation models of care, respectively. It involves the active collaboration between the patient, his family/supports and the multidisciplinary team. Exercise training, education, nutritional intervention and psychosocial support are key components. As cardiac and pulmonary rehabilitation programmes are now widely recognized as part of the routine management of patients with chronic cardiac and respiratory disease, most patients assessed for transplant will have participated in a rehabilitation programme before transplant assessment (Lloyd-Williams et al 2002).
Pre-transplant rehabilitation aims to optimize physical and functional performance and quality of life. Addressing the deconditioning that results from the preoperative disease state is considered essential for survival to transplant and recovery afterwards.
Exercise training
The heterogeneous nature of transplant candidates means that the individual patient’s unique pathophysiology, time in clinical course, symptoms, needs, goals and response to exercise must be taken into consideration when undertaking exercise training. All patients waiting for heart, heart-lung and lung transplants are encouraged to remain as active as possible. A careful assessment is required to ensure that each patient is safe to exercise. Physiotherapists working in this area may be exposed to conditions rarely seen. Caution must be taken with all patients. Patients who demonstrate profound desaturation despite oxygen therapy, hypotension or any other markedly abnormal response may not be able to exercise, or will be restricted to gentle stretches or activities of daily living only. Overall, the vast majority of patients are safe to participate in an exercise training programme.
General guidelines for exercise training in cardiac rehabilitation and pulmonary rehabilitation have been discussed (Chapters 13 & 14). It is important to note that currently there are no formal evidence-based guidelines regarding exercise training in lung disease populations other than COPD. Expert opinion based on knowledge of the underlying pathophysiology and clinical experience underpins clinical practice in this area.
Some disease groups have unique features that must be taken into consideration when planning and conducting exercise training programmes. Patients who experience dynamic hyperinflation may benefit from using a four-wheeled frame to support their upper limbs during lower limb training (Solway et al 2002). The prevention of cross-infection and ensuring an adequate salt intake is essential for patients with cystic fibrosis (Lands & Coates 1999, Saiman & Siegel 2004). Patients with interstitial lung disease often experience dyspnoea but may be able to perform interval training regimens with high-flow oxygen. High-intensity exercise is generally not recommended for patients with pulmonary arterial hypertension (Nici et al 2006).
Cardiac transplant candidates are generally able to exercise within the limits of standard haemodynamic guidelines. Close monitoring of symptoms, in particular dyspnoea, dizziness, light-headedness and chest pain, in addition to heart rate and blood pressure, is required. If they have an inappropriate response to exercise (e.g. a decrease in systolic blood pressure >20 mmHg), exercise should be ceased (Fletcher et al 2001).
Exercise tolerance in heart failure patients is poorly correlated with the degree of cardiac dysfunction (Myers et al 1998). The major limitation to exercise capacity stems from secondary, peripheral adaptations. These include impaired muscle structure and function, vascular and metabolic abnormalities (Clark et al 1996). Recent studies suggest that VO2peak, a measure of cardiopulmonary exercise capacity, strongly predicts prognosis in heart failure, exhibiting a higher correlation with mortality than measures of left ventricular function (Myers et al 1998). The positive effect of endurance training on VO2peak in patients with heart failure is well recognized (Lloyd-Williams et al 2002); however, it is known that endurance training alone does not enhance skeletal muscle strength (Delagardelle et al 2002). Resistance training has been shown to be safe and is recommended for patients with stable heart failure, as it directly targets the peripheral impairments demonstrated by this patient population (King et al 2000, Werber-Zion et al 2004). Studies have shown that 1-repetition maximum (RM) testing should be performed to ensure effective and safe dosage of resistance training (Werber-Zion et al 2004). A combined model of training would appear to be superior to either isolated endurance or resistance training, with improvements in VO2peak, muscle strength, endurance, vascular function and quality of life (Maiorana et al 2000).
In lung transplant candidates, many factors con-tribute to exercise limitation, including ventilatory limitations, gas exchange abnormalities, skeletal muscle dysfunction and respiratory muscle dysfunction. It has been shown that patients with severe chronic respiratory disease can sustain the necessary training intensity and duration for skeletal muscle adaptation (Maltais et al 1996, Whittom et al 1998).
Close monitoring of symptoms, particularly shortness of breath, in addition to oxygen saturation and heart rate is required. An appropriate target for oxygen saturation during exercise for each individual should be determined before starting exercise. Supplemental oxygen is often required. It is clinical practice to maintain oxygen saturation at greater than 90% during exercise where possible. Non-invasive ventilation may help achieve a greater training intensity by unloading the respiratory muscles in some patients (Hoo 2003, van ‘t Hul et al 2006).
Most adult transplant centres offer dedicated preoperative exercise training classes for transplant candidates. Patients living close to the transplant centre attend the gymnasium 2-3 times a week and continue elements of the programme in a home routine. Patients who regularly attend other centres for ongoing outpatient treatment should be supervised by their local physiotherapist. Patient progress should be reported back to the transplant team. Patients who live beyond the reach of regular hospital attendance often benefit from regular telephone contact. The use of a diary to maintain motivation and for adequate monitoring is often beneficial.
Experience suggests that an ongoing, supervised exercise programme performed with other transplant candidates is ideal. Supervision by an experienced physiotherapist allows for alterations in exercise prescription as physical performance and symptoms change. Patient behavioural patterns, particularly adherence to therapies and the ability to cope with stressors, can be observed and strategies to facilitate modification put in place where needed. In a supportive, encouraging environment, patient motivation and self-management can be fostered and maintained during the often long waiting period (Craven et al 1990). Exercise training classes are often a key referral point for care from other members of the multidisciplinary team.
Pre-transplant exercise guidelines
 Training intensity:
Training intensity:
— lung patients: aim for Borg 4 for dyspnoea or fatigue
— cardiac patients: aim for Borg rate of perceived exertion (RPE) 9-12 (Chapter 14).
Education
Pre-transplant education is facilitated by various members of the multidisciplinary team. Some units have a structured, group education programme while others provide one-to-one (individual patients and carer) education.
 practical aspects of being on the waiting lists (notification, what to bring to hospital, how to get to hospital)
practical aspects of being on the waiting lists (notification, what to bring to hospital, how to get to hospital)Timing to transplantation is variable, with more hearts being available than lungs. Time on the waiting list can vary from weeks to years. Once on the waiting list, patients are monitored closely by the transplant team to ensure that candidates continue to meet selection criteria. Periodic review also allows for fine-tuning of patient management.
Bridge to transplant
In an effort to optimize therapy and improve survival while on the waiting list, a number of manage-ment strategies including pharmacological therapies, device therapies and surgical intervention are considered.
Device therapies have been shown to decrease mortality and improve cardiac function in patients with end-stage heart failure. Device therapies used in cardiac transplant candidates include implantable cardioverter-defibrillators (ICDs), cardiac resynchronization therapy (CRT) and ventricular assist devices (VADs).
The left ventricular assist device (LVAD) has been increasingly utilized as a bridge to cardiac transplantation and is now being used as destination therapy (Peterzen et al 2002). The first bridge to transplant with a LVAD was performed more than 20 years ago. Since that time, devices have continued to evolve. Increased knowledge about patient selection, the timing of implantation and improved patient management has resulted in improved outcomes with decreasing adverse events.
LVAD implantation has been shown to have a significant impact on exercise capacity, with improvements in haemodynamic, ventilatory and neurohormonal measures (Mancini et al 1998, Peterzen et al 2002). Both submaximal and peak exercise capacity have been shown to improve. It seems logical therefore, that if the general health and fitness of these patients can be improved before transplantation, this may improve outcome following surgery.
Exercise prescription for LVAD patients is a particular challenge given the nature and position of the equipment and the often very poor functional capacity of these patients prior to implantation. Several studies (Morrone et al 1996, Reedy et al 1992) have demonstrated that early submaximal exercise is not only safe in this population but improves morbidity and mortality. A structured training programme has been shown to improve exercise capacity (Mettauer et al 2001); however, more work is needed in this area to determine the optimum type and intensity of exercise.
In lung transplant candidates, surgery, pharmaceutical agents and mechanical support are also used. Lung volume reduction surgery may be used to delay the need for transplant in highly selected patients with emphysema. (Cordova & Criner 2002). Inhaled nitric oxide may be used in the setting of severe pulmonary hypertension (Olsson et al 2005, Yung et al 2001) and non-invasive ventilation for those with hypercapnic respiratory failure (British Thoracic Society 2002) and cystic fibrosis (Madden et al 2002).
POSTOPERATIVE MANAGEMENT
The aim of medical management in the early transplant period is to initiate effective immunosuppression, to minimize the risk of infection and to protect other organ systems, such as the renal system.
Post-transplant physiotherapy management is similar to that of other thoracic surgery patients. All physiotherapy interventions must be based on thorough assessment and treatment individualized for each patient. Subjective and objective assessment findings must be reviewed along with the latest microbiology results, arterial blood gases, chest radiograph, cardiovascular measures and oxygen saturation. Treatment choice, frequency and duration will depend on the individual patient presentation. Patients vary according to a number of factors, including:
Physiotherapy treatment in the postoperative period aims to:
Management in the intensive care unit
The physiotherapy programme is initiated in the intensive care unit (ICU) as early as the first postoperative day. Acutely, the goals are to prevent perioperative complications of bedrest. Airway clearance techniques, joint range of motion and positioning are implemented as indicated. Once the cardiovascular and respiratory systems are stabilized, the patient is rapidly weaned from the ventilator and extubated. Delay in extubation can occur for a number of reasons. Primary graft failure is one of the most common reasons. As the intubation period increases, so too does the risk of nosocomial pneumonia. Non-invasive ventilatory support may be useful in assisting patients who have experienced difficulties in weaning and extubation.
It is important for patients to have sufficient analgesia to allow effective huffing, coughing and for early mobilization. Patients are commonly taught thoracic expansion exercises, huffing and coughing routines to detect and clear secretions. It is common for lung and heart-lung transplant recipients to expectorate old blood and slough from the bronchial/tracheal anastomosis during the first few days to a week. Secretions may also originate from the donor (donor-acquired infection) or recipient’s native airways (common in cystic fibrosis).
Lung transplant patients often have a poor ability to perceive the presence of secretions and this may persist in the long term. The loss of neural innervation and abnormal mucociliary function are amongst a number of factors that predispose the lung transplant recipient to lung infections (Chaparro & Kesten 1997). If sputum retention becomes a problem for a recipient, inhalation therapy and an appropriate airway clearance technique should be instituted. Positioning, combined with an airway clearance technique (e.g. active cycle of breathing techniques), are the primary forms of initial treatment. If other airway clearance techniques are deemed necessary (i.e. treatments involving positive pressure), this is usually undertaken following consultation with the medical team. The place of regular airway clearance in the absence of excessive secretions has not yet been established but is currently under investigation.
Once extubated and stable, the emphasis is on early mobilization, with patients assisted to sit out of bed often within 24 hours of surgery (Fig. 15.4). Ambulation is initiated in the ICU when the patient shows adequate muscle strength, testing a minimum of grade 3 out of 5 in selective lower limb muscles (Clarkson & Gilewich 1989). The length of stay in ICU can be as short as 1 day or extend to prolonged periods depending on complications.
Ward management
On the ward, physiotherapy treatment focuses on achieving independence with activities of daily living, increasing endurance (walking, stationary cycling, stair climbing), and exercises addressing any specific musculoskeletal deficits. Stationary cycling in the patient’s room is particularly useful in the early stages following lung and heart-lung transplant when intercostals drains are on suction. Full range of upper limb elevation should be encouraged once drain tubes are removed. Postural re-education is also often required. Motivation and self-management may be enhanced by using charts to provide a measure of improvement in function.
Regular airway clearance may be necessary only in the presence of infection and retained secretions. Routine bronchoscopy is used to assess airway anastomotic healing and patency, the presence of infection (from bronchoalveolar lavage) and rejection (from transbronchial biopsy). Bronchoscopic findings are very useful to the physiotherapist and assist in clinical decision making and in determining appropriate treatments.
The average length of hospital stay following heart or lung transplantation is 2-3 weeks. Some patients are discharged as early as 1 week postoperatively, while others may spend weeks to months in hospital secondary to complications.
Before discharge from hospital, patients must have a good understanding of the signs and symptoms of rejection and infection, their medications and chest wall/sternal precautions. Patients are advised not to lift greater than 5 kg for 10-12 weeks. Rehabilitation usually continues on an outpatient basis at the transplant centre.
OUTPATIENT REHABILITATION
Many patients attend a formal outpatient rehabilitation programme comprising exercise training and education at the transplant centre. The primary goals in the rehabilitation phase after transplantation are to:
 improve the patient’s confidence in becoming involved in a full range of activities of daily living and appropriate exercise activities
improve the patient’s confidence in becoming involved in a full range of activities of daily living and appropriate exercise activitiesIdeally patients would attend the gymnasium three times per week for supervised exercise training, commonly in a group setting with other transplant recipients (Fig. 15.5). By approximately 12 weeks, most patients have achieved a good level of fitness and function and are able to be discharged from physiotherapy with a maintenance home exercise programme to be undertaken independently. Patients are encouraged to maintain an active lifestyle (Fig. 15.6) and those who require further rehabilitation are referred closer to their local area where access to services is more convenient.
Exercise training programmes post-transplant are similar to those previously described pre-transplant. Special considerations for exercise training are outlined in Box 15.5.
Box 15.5 Special considerations for exercise training post-transplant
Closely monitor signs and symptoms
 Transplant complications (e.g. rejection, infection) often manifest as a reduction in exercise tolerance or abnormal exercise response. Concerns should be reported immediately to the transplant unit.
Transplant complications (e.g. rejection, infection) often manifest as a reduction in exercise tolerance or abnormal exercise response. Concerns should be reported immediately to the transplant unit.Heart transplant recipients have an abnormal response to exercise due to denervation
 Ensure adequate warm up and cool down to allow adequate time for the effects of catecholamine levels on cardiac output (Niset et al 1991).
Ensure adequate warm up and cool down to allow adequate time for the effects of catecholamine levels on cardiac output (Niset et al 1991).Consider the musculoskeletal side effects of immunosuppressant drugs
(see Table 15.2)
Observe chest wall/sternal precautions
Bone morbidity — osteoporosis, pathological fracture and avascular necrosis are common
 Progressive resistance training should be included in post-transplant exercise programmes to optimize bone mineral density (Braith et al 1996, Mitchell et al 2003).
Progressive resistance training should be included in post-transplant exercise programmes to optimize bone mineral density (Braith et al 1996, Mitchell et al 2003). Pathological fracture (particularly vertebral compression) and avascular necrosis (commonly head of femur) should be considered as potential causes of pain. Imaging techniques such as bone scan may be required so that appropriate management is undertaken (Henderson et al 1997).
Pathological fracture (particularly vertebral compression) and avascular necrosis (commonly head of femur) should be considered as potential causes of pain. Imaging techniques such as bone scan may be required so that appropriate management is undertaken (Henderson et al 1997).Care must be taken when progressing activity
 Most patients are unaccustomed to exercise and are at risk of overuse injuries if activity is progressed too rapidly (e.g. achilles tendinitis, shin splints).
Most patients are unaccustomed to exercise and are at risk of overuse injuries if activity is progressed too rapidly (e.g. achilles tendinitis, shin splints).Modify exercise during episodes of acute rejection
 Moderate rejection — patients should not exercise during the short period in which they are receiving high-dose steroids. Exercise should be gradually increased and symptom-limited when recommenced.
Moderate rejection — patients should not exercise during the short period in which they are receiving high-dose steroids. Exercise should be gradually increased and symptom-limited when recommenced.HR, heart rate; BP, blood pressure; SOB, shortness of breath; RPE, rate of perceived exertion
Education continues for the patient and carer in the outpatient setting facilitated by the multidisciplinary team. Common topics include:
Regular medical follow-up continues until most patients can be reviewed on a 6-12 monthly basis. Improvements in functional exercise capacity and quality of life reflect the multimodal nature of the management of the transplant recipient (e.g. surgery, rehabilitation, lifestyle changes). Specific outcome measures for physiotherapy interventions should relate to each physiotherapy goal. Current clinical practice is variable and there is no consensus as to which key outcomes should be used (Maher & Williams 2005). It has been suggested that functional exercise capacity and level of activity should be measured before and after postoperative rehabilitation and then yearly as a minimum.
FUNCTIONAL OUTCOMES POST-TRANSPLANT
Exercise capacity improves significantly after heart, heart-lung and lung transplantation; however, it remains below normal predicted values.
Heart transplant recipients demonstrate an improvement in VO2peak for up to 2 years post-transplant; however, this value is only approximately 50% of normal values. The main determinant of this lower exercise capacity appears to be chronotropic incompetence of the denervated heart (Mandak et al 1995, Quigg et al 1988).
Substantial exercise limitation is seen almost universally in lung transplant recipients with a VO2peak of 40-60% predicted normal (Howard et al 1994, Levy et al 1993 , Theodore et al 1987, Williams et al 1992). Patients demonstrate an adequate heart rate reserve, no significant gas exchange abnormalities and a normal breathing reserve. Peripheral muscle limitation, with leg tiredness the predominant symptom at exercise termination, is thought to be due to a number of factors including skeletal muscle deconditioning, dysfunction and atrophy (Wang et al 1999) and myotoxic effects of immunosuppressants (Mercier et al 1995).
Perceived functional status also improves following thoracic organ transplantation (Grady & Lanuza 2005, Lanuza et al 2000). Eighty to ninety per cent of paediatric transplant recipients report no limitation to activity levels for at least 5 years following transplant (Boucek et al 2005). Adult recipients demonstrate variable return to work rates (12-74%), which appear to increase over time. Patients report high global quality of life and these perceptions improve over pre-transplant levels (Cupples et al 2006).
LONG-TERM MANAGEMENT
Improved survival outcomes have led to significant growth in the demand for physiotherapy management of long-term morbidity. Long-term survivors experience a range of problems requiring hospital admission or outpatient follow-up. These include:
Many transplant recipients maintain near-normal function for many years post-transplant. Others develop complications that require long-term medical or physiotherapy input. Physiotherapists commonly address problems such as:
In such cases there is a need to set realistic goals and to assist the patient and family to accept the decline in functional performance. Optimizing quality of life remains the primary goal. Inevitably, the care of these patients shifts away from acute medical management towards chronic disease management and palliative care. This transition is often difficult for both the patient and the health care team.
SPECIFIC CONSIDERATIONS FOR PAEDIATRIC PATIENTS
Most of the information presented above applies to both paediatric and adult heart and lung transplant recipients. However, there are a few additional issues that are specific to children. These are listed in Table 15.3.
Table 15.3 Specific considerations for children undergoing heart or lung transplantation
| Lung function testing | All lung transplant recipients use spirometry to monitor lung function. This is possible in all children aged 4 years or more, though techniques may need to be modified. Different lung function techniques are needed for children less than 4 years |
| Biopsy | Endocardial and transbronchial biopsies are essential for detecting graft rejection. In all children these procedures are usually performed under general anaesthetic, rather than sedation. They are technically more difficult in younger children |
| Infections | Many children will not have had previous exposure to common viruses, and the incidence of primary infection is much higher than for adults. It is therefore essential that immunization status is optimized prior to listing for transplantation |
| Malignancy | Post-transplant lymphoproliferative disease (PTLD) is far more common in children than in adults. Other tumours, especially skin and lung, are rarer in children |
| Psychosocial issues | Many children coming to transplant are physically and emotionally immature because of their chronic illness. A successful transplant allows a child to transform their life and to catch up on many of the activities that were previously denied to them. Some children find this change in lifestyle difficult, particularly if it coincides with puberty. Non-adherence to therapy is an important cause of poor outcome, especially in teenage patients, and appears worst in those who have a chronic illness like cystic fibrosis. Most centres stress that adolescents should steadily take more responsibility for their own care and are given practical assistance to boost adherence to therapy |
CONCLUSION
Over the past two decades, thoracic organ transplantation has changed from a highly experimental surgical procedure to a widely accepted therapeutic option for end-stage cardiac and pulmonary failure. Continuing advances in all aspects of pre- and post-transplant management (e.g. immunosuppressive therapy, medical management, operative techniques) have led to significant improvements in morbidity and mortality. As long-term survival improves, there is greater recognition of the importance of optimizing the physical condition of potential candidates pre-transplant and the need to actively manage long-term comorbidities. Physiotherapists have a key and enduring role in the management of these patients that extends from pre-transplant assessment through to the palliative care of long-term survivors. Patients undergoing thoracic organ transplantation present with a broad range of complex medical issues, which necessitate management by a highly specialized team. Physiotherapists are an integral part of that team, and as such require a high level of knowledge and a range of specialist skills in the management of cardiorespiratory, orthopaedic and neurological pathologies.
Aurora P, Wade A, Whitmore P, Whitehead B. A model for predicting life expectancy of children with cystic fibrosis. European Respiratory Journal. 2000;16(6):1056-1060.
Borel JF. Immunosuppressive properties of ciclosporin A (CY-A). Transplantation Proceedings. 1980;12(2):233.
Boucek MM, Edwards LB, Keck BM, et al. Registry of the International Society for Heart and Lung Transplantation: Eighth Official Pediatric Report - 2005. Journal of Heart and Lung Transplantation. 2005;24(8):968-982.
Braith RW, Mills RM, Welsch MA, Keller JW, Pollock ML. Resistance exercise training restores bone mineral density in heart transplant recipients. Journal of the American College of Cardiology. 1996;28(6):1471-1477.
British Thoracic Society Standards of Care Committee. Non-invasive ventilation in acute respiratory failure. Thorax. 2002;57(3):192-211.
Button BM, Roberts S, Kotsimbos TC, et al. Gastroesophageal reflux (symptomatic and silent): a potentially significant problem in patients with cystic fibrosis before and after lung transplantation. Journal of Heart and Lung Transplantation. 2005;24(10):1522-1529.
Cantu E3rd, Appel JZ3rd, Hartwig MG, et al. J. Maxwell Chamberlain Memorial Paper. Early fundoplication prevents chronic allograft dysfunction in patients with gastroesophageal reflux disease. Annals of Thoracic Surgery. 2004;78(4):1142-1151.
Chaparro C, Kesten S. Infections in lung transplant recipients. Clinics in Chest Medicine. 1997;18(2):339-351.
Clark A, Poole-Wilson P, Coats A. Exercise limitation in chronic heart failure: central role of the periphery. Journal of the American College of Cardiology. 1996;28:1092-1102.
Clarkson H, Gilewich G. Musculoskeletal assessment. In: Joint range of motion and manual muscle strength. Baltimore: Williams & Wilkins; 1989.
Cooper JD, Billingham M, Egan T, et al. A working formulation for the standardization of nomenclature and for clinical staging of chronic dysfunction in lung allografts. International Society for Heart and Lung Transplantation. Journal of Heart and Lung Transplantation. 1993;12(5):713-716.
Cooper JD, Pearson FG, Patterson GA, et al. Technique of successful lung transplantation in humans. Journal of Thoracic and Cardiovascular Surgery. 1987;93(2):173-181.
Cordova FC, Criner GJ. Lung volume reduction surgery as a bridge to lung transplantation. American Journal of Respiratory Medicine. 2002;1(5):313-324.
Couetil JP, Tolan MJ, Loulmet DF, et al. Pulmonary bipartitioning and lobar transplantation: a new approach to donor organ shortage. Journal of Thoracic and Cardiovascular Surgery. 1997;113(3):529-537.
Craven JL, Bright J, Dear CL. Psychiatric, psychosocial, and rehabilitative aspects of lung transplantation. Clinics in Chest Medicine. 1990;11(2):247-257.
Crowley S, Egan JJ. Macrolide antibiotics and bronchiolitis obliterans following lung transplantation. Expert Review of Anti-infective Therapy. 2005;3(6):923-930.
Cupples S, Dew MA, Grady KL, et al. Report of the Psychosocial Outcomes Workgroup of the Nursing and Social Sciences Council of the International Society for Heart and Lung Transplantation: present status of research on psychosocial outcomes in cardiothoracic transplantation: review and recommendations for the field. Journal of Heart and Lung Transplantation. 2006;25(6):716-725.
Date H, Aoe M, Nagahiro I, et al. Living-donor lobar lung transplantation for various lung diseases. Journal of Thoracic and Cardiovascular Surgery. 2003;126(2):476-481.
Davis RDJ, Lau CL, Eubanks S, et al. Improved lung allograft function after fundoplication in patients with gastroesophageal reflux disease undergoing lung transplantation. Journal of Thoracic and Cardiovascular Surgery. 2003;125(3):533-542.
Del Rizzo DF, Menkis AH, Pflugfelder PW, et al. The role of donor age and ischemic time on survival following orthotopic heart transplantation. Journal of Heart and Lung Transplantation. 1999;18(4):310-319.
Delagardelle C, Feiereisen P, Autier P, et al. Strength/endurance training versus endurance training in congestive heart failure. Medicine and Science in Sports and Exercise. 2002;34(12):1868-1872.
Fletcher G, Balady G, Amsterdam E, et al. Exercise standards for testing and training: a statement for healthcare professionals from the American Heart Association. Circulation. 2001;104:1694-1740.
Gabbay E, Williams TJ, Griffiths AP, et al. Maximizing the utilization of donor organs offered for lung transplantation. American Journal of Respiratory and Critical Care Medicine. 1999;160(1):265-271.
Grady KL, Lanuza DM. Physical functional outcomes after cardiothoracic transplantation. Journal of Cardiovascular Nursing. 2005;20(5 Suppl):S43-50.
Grigioni F, Carigi S, Potena L, et al. Long-term safety and effectiveness of statins for heart transplant recipients in routine clinical practice. Transplant Proceedings. 2006;38(5):1507-1510.
Henderson K, Marshall G, Sambrook P, Keogh A, Eisman J. Two cases of hip pain in patients with heart transplantation. Australian Journal of Physiotherapy. 1997;43(2):131-133.
Higenbottam T, Jackson M, Woolman P, Lowry R, Wallwork J. The cough response to ultrasonically nebulized distilled water in heart-lung transplantation patients. American Review of Respiratory Disease. 1989;140(1):58-61.
Hoo GWS. Nonpharmacologic adjuncts to training during pulmonary rehabilitation: the role of supplemental oxygen and noninvasive ventilation. Journal of Rehabilitation Research and Development. 2003;40(5 Suppl 2):81-97.
Hosenpud J. Coronary artery disease after heart transplantation. American Heart Journal. 1999;138(5 part 2):469-472.
Howard DK, Iademarco EJ, Trulock EP. The role of cardiopulmonary exercise testing in lung and heart-lung transplantation. Clinics in Chest Medicine. 1994;15(2):405-420.
Hussain T, Fenton MJ, Burch M, et al. Pre-transplant cytomegalovirus serology is a risk factor for cardiac allograft vasculopathy in children. Journal of Heart and Lung Transplantation. 2006;25(2):85. (Suppl)
Jamieson S, Stinson E, Oyer PE, Baldwin JC, Shumway NE. Operative technique for heart-lung transplantation. Journal of Thoracic and Cardiovascular Surgery. 1984;87:930-935.
Jonas M, Fang JC, Wang JC, et al. In-stent restenosis and remote coronary lesion progression are coupled in cardiac transplant vasculopathy but not in native coronary artery disease. Journal of the American College of Cardiology. 2006;48(3):453-461.
Kadikar A, Maurer J, Kesten S. The six-minute walk test: a guide to assessment for lung transplantation. Journal of Heart and Lung Transplantation. 1997;16(3):313-319.
Kavanagh T. Physical training in heart transplant recipients. Journal of Cardiovascular Risk. 1996;3:154-159.
Keogh A, Baron D, Spratt P, Esmore DS, Chang V. Cardiac transplantation in Australia. Australian Family Physician. 1986;15(11):1474-1481.
King M, Dracup K, Fonarow G, Woo MA. The hemodynamic effects of isotonic exercise using hand-held weights in patients with heart failure. Journal of Heart and Lung Transplantation. 2000;19:1209-1218.
Lands L, Coates A. Cardiopulmonary and skeletal muscle function and their effects on exercise limitation. In: Yankaskas J, Knowles M, editors. Cystic fibrosis in adults. New York: Lippincott-Raven; 1999:365-379.
Lanuza DM, Lefaiver CA, Farcas GA. Research on the quality of life of lung transplant candidates and recipients: an integrative review. Heart and Lung. 2000;29(3):180-195.
Levy RD, Ernst P, Levine SM, et al. Exercise performance after lung transplantation. Journal of Heart and Lung Transplantation. 1993;12(1, Pt 1):27-33.
Lloyd-Williams F, Mair FS, Leitner M. Exercise training and heart failure: a systematic review of current evidence. British Journal of General Practice. 2002;52(474):47-55.
Madden BP, Kariyawasam H, Siddiqi AJ, et al. Noninvasive ventilation in cystic fibrosis patients with acute or chronic respiratory failure. European Respiratory Journal. 2002;19(2):310-313.
Maher C, Williams M. Factors influencing the use of outcome measures in physiotherapy management of lung transplant patients in Australia and New Zealand. Physiotherapy Theory and Practice. 2005;21(4):201-217.
Maiorana A, O’Driscoll G, Cheetham C, et al. Combined aerobic and resistance exercise training improves functional capacity and strength in chronic heart failure. Journal of Applied Physiology. 2000;88:1565-1570.
Maltais F, LeBlanc P, Simard C, et al. Skeletal muscle adaptation to endurance training in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine. 1996;154(2 Pt 1):442-447.
Mancini D, Goldsmith R, Levin H, et al. Comparison of exercise performance in patients with chronic severe heart failure versus left ventricular assist devices. Circulation. 1998;98(12):1178-1183.
Mancini DM, Eisen H, Kussmaul W, et al. Value of peak exercise oxygen consumption for optimal timing of cardiac transplantation in ambulatory patients with heart failure. Circulation. 1991;83(3):778-786.
Mandak J, Aaransen K, Mancini D. Serial assessment of exercise capacity after heart transplantation. Journal of Heart and Lung Transplantation. 1995;14(3):468-478.
Mattila S, Mattila I, Viljanen B, Viljanen A. Reappearance of Hering-Breuer reflex after bilateral autotransplantation of the lungs. Scandinavian Journal of Thoracic and Cardiovascular Surgery. 1987;21(1):15-20.
Mercier JG, Hokanson JF, Brooks GA. Effects of ciclosporine A on skeletal muscle mitochondrial respiration and endurance time in rats. American Journal of Respiratory and Critical Care Medicine. 1995;151(5):1532-1536.
Mettauer B, Geny B, Lonsdorfer-Wolf E, et al. Exercise training with a heart device: a hemodynamic, metabolic, and hormonal study. Medicine and Science in Sports and Exercise. 2001;33(1):2-8.
Mitchell MJ, Baz MA, Fulton MN, Lisor CF, Braith RW. Resistance training prevents vertebral osteoporosis in lung transplant recipients. Transplantation. 2003;76(3):557-562.
Moorish W, Herman S, Weisbrod G, Chamberlain D. Bronchiolitis obliterans after lung transplantation: findings at chest radiography and high-resolution CT. Radiology. 1991;179:487-490.
Morgan JA, Edwards NM. Orthotopic cardiac transplantation: comparison of outcome using biatrial, bicaval and total techniques. Journal of Cardiac Surgery. 2005;20:102-106.
Morrone T, Buck L, Catanese K, et al. Early progressive mobilization of patients with left ventricular assist devices is safe and optimizes recovery before heart transplantation. Journal of Heart and Lung Transplantation. 1996;15:423-429.
Myers J, Gullestad L, Vagelos R, et al. Clinical, hemodynamic, and cardiopulmonary exercise test determinants of survival in patients referred for evaluation of heart failure. Annals of Internal Medicine. 1998;129:286-293.
Newcomb AE, Esmore DS, Rosenfeldt FL, Richardson M, Marasco SF. Heterotopic heart transplantation: an expanding role in the twenty-first century? Annals of Thoracic Surgery. 2004;78(4):1345-1350.
Nici L, Donner C, Wouters E, et al. American Thoracic Society/European Respiratory Society statement on pulmonary rehabilitation. American Journal of Respiratory and Critical Care Medicine. 2006;173(12):1390-1413.
Niset G, Hermans L, Depelchin P. Exercise and heart transplantation. A review. Sports Medicine. 1991;12(6):359-379.
Novitzky D, Cooper DK, Barnard CN. The surgical technique of heterotopic heart transplantation. Annals of Thoracic Surgery. 1983;36(4):476-482.
Olsson JK, Zamanian RT, Feinstein JA, Doyle RL. Surgical and interventional therapies for pulmonary arterial hypertension. Seminars in Respiratory and Critical Care Medicine. 2005;26(4):417-428.
Patterson GA, Cooper JD, Goldman B, et al. Technique of successful clinical double-lung transplantation. Annals of Thoracic Surgery. 1988;45(6):626-633.
Peterzen B, Lonn U, Jansson K, et al. Long-term follow-up of patients treated with an implantable left ventricular assist device as an extended bridge to heart transplantation. Journal of Heart Transplantation. 2002;21:604-607.
Quigg R, Salyer J, Mohanty P, Simpsen P. Impaired exercise capacity late after cardiac transplantation: influence of chronotropic incompetence, hypertension, and calcium channel blockers. American Heart Journal. 1988;136(3):465-473.
Raul G, Germain P, Bareiss P. Does the 6-minute walk test predict prognosis in patients with NYHA class II or III chronic heart failure? American Heart Journal. 1998;136(3):449-457.
Reedy JE, Swartz MT, Lohmann DP, et al. The importance of patient mobility with ventricular assist device support. American Society for Artificial Internal Organs Journal. 1992;38(3):M151-153.
Reitz BA, Wallwork JL, Hunt SA, et al. Heart-lung transplantation: successful therapy for patients with pulmonary vascular disease. New England Journal of Medicine. 1982;306(10):557-564.
Saiman L, Siegel J. Infection control in cystic fibrosis. Clinical Microbiology Review. 2004;17(1):57-71.
Solway S, Brooks D, Lau L, Goldstein R. The short-term effect of a rollator on functional exercise capacity among individuals with severe COPD. Chest. 2002;122(1):56-65.
Starnes VA, Woo MS, MacLaughlin EF, et al. Comparison of outcomes between living donor and cadaveric lung transplantation in children. Annals of Thoracic Surgery. 1999;68(6):2279-2283. ; discussion 2283-2274
Taylor DO, Edwards LB, Boucek MM, et al. Registry of the International Society for Heart and Lung Transplantation: Twenty-second Official Adult Heart Transplant Report - 2005. Journal of Heart and Lung Transplantation. 2005;24(8):945-955.
Thabut G, Mal H, Cerrina J, et al. Graft ischemic time and outcome of lung transplantation: a multicenter analysis. American Journal of Respiratory and Critical Care Medicine. 2005;171(7):786-791.
Theodore J, Morris AJ, Burke CM, et al. Cardiopulmonary function at maximum tolerable constant work rate exercise following human heart-lung transplantation. Chest. 1987;92(3):433-439.
Trulock EP, Edwards LB, Taylor DO, et al. Registry of the International Society for Heart and Lung Transplantation: Twenty-second Official Adult Lung and Heart-lung Transplant Report -, 2005. Journal of Heart and Lung Transplantation. 2005;24(8):956-967.
van’t Hul A, Gosselink R, Hollander P, Postmus P, Kwakkel G. Training with inspiratory pressure support in patients with severe COPD. European Respiratory Journal. 2006;27(1):65-72.
Wang XN, Williams TJ, McKenna MJ, et al. Skeletal muscle oxidative capacity, fiber type, and metabolites after lung transplantation. American Journal of Respiratory and Critical Care Medicine. 1999;160(1):57-63.
Weber B. Cardiac surgery and heart transplantation. In Hudak C, Gallo B, Benz J, editors: Critical care nursing: a holistic approach, 5th edn., Philadelphia, PA: JB Lippincott, 1990.
Werber-Zion G, Goldhammer E, Shaar A, Pollock M. Left ventricular function during strength testing and resistance exercise in patients with left ventricular dysfunction. Journal of Cardiopulmonary Rehabilitation. 2004;24:100-109.
Whittom F, Jobin J, Simard PM, et al. Histochemical and morphological characteristics of the vastus lateralis muscle in patients with chronic obstructive pulmonary disease. Medicine and Science in Sports and Exercise. 1998;30(10):1467-1474.
Williams TJ, Patterson GA, McClean PA, Zamel N, Maurer JR. Maximal exercise testing in single and double lung transplant recipients. American Review of Respiratory Disease. 1992;145(1):101-105.
Yousem SA, Berry GJ, Cagle PT, et al. Revision of the 1990 working formulation for the classification of pulmonary allograft rejection: Lung Rejection Study Group. Journal of Heart and Lung Transplantation. 1996;15(1 Pt 1):1-15.
Yung GL, Kriett JM, Jamieson SW, et al. Outpatient inhaled nitric oxide in a patient with idiopathic pulmonary fibrosis: a bridge to lung transplantation. Journal of Heart and Lung Transplantation. 2001;20(11):1224-1227.