Chapter 14 Cardiac rehabilitation and secondary prevention
INTRODUCTION
Cardiac rehabilitation is an accepted form of management for people with cardiac disease. Initially rehabilitation was offered mainly to people recovering from a myocardial infarction (MI), but now encompasses a wide range of cardiac problems. More than 275 000 people have a MI each year in England and, while more than 40% of these are fatal, the survival rate is improving (Department of Health 2006). With more people surviving the peri-infarct period, the incidence of people with chronic heart failure (CHF) is increasing. These changes in mortality and morbidity present challenges for health professionals involved in organizing and delivering rehabilitation programmes. The aim of cardiac rehabilitation is to enable people to regain full physical, psychological and social status and, in order to optimize long-term prognosis, to promote and implement coronary heart disease (CHD) secondary prevention measures. Cardiac rehabilitation should be an integral part of both acute care and long-term follow-up, but provision and uptake remains inconsistent (Beswick et al 2004, Bittner & Sanderson 2006, Brodie et al 2006). In the United Kingdom (UK), the National Service Framework for CHD (Department of Health 2000) was written to provide guidance on the management of people with coronary heart disease. One of the standards of recommended care is the provision of a multidisciplinary programme of cardiac rehabilitation and secondary prevention. There is large variation in the format and organization of rehabilitation programmes, both within and between countries, but traditionally most encompass a period of exercise training, education sessions, psychosocial support and advice/counselling for both the patient and their family. In addition to people with a MI or CHF, participants may include those with an implanted cardioverter defibrillator (ICD), pacemaker and following coronary artery bypass graft surgery (CABG) and transplantation. Demographic changes mean there is greater involvement of elderly people, which is both a reflection of an ageing population and expansion of health care, and there is also a gradual increase in the uptake of cardiac rehabilitation amongst women. The secondary prevention aspect of the programmes has an important place in the reduction of risk factors. Physically inactive people have approximately double the risk of CHD compared with active people and the implementation of regular exercise is considered to decrease their risk factor profile by the same magnitude as smoking cessation (Albu et al 2006, Department of Health 2004).
This chapter describes the exercise component of cardiac rehabilitation which is the aspect in which physiotherapists are mainly involved and is divided into five parts:
EVIDENCE BASE FOR CARDIAC REHABILITATION
With the increased evidence base for the benefits of cardiac rehabilitation, and particularly exercise training, many clinical guidelines for the management of people with cardiac problems recommend participation in a rehabilitation programme (National Institute for Clinical Excellence 2001, 2003, Scottish Intercollegiate Guidelines Network 2002). While there is a plethora of research supporting the benefits of cardiac rehabilitation programmes, the era in which the information was obtained has to be taken into consideration. Early studies concentrated on the benefits of exercise for post-MI patients in whom traditional management had restricted early activity – which differs from the early resumption of activity practised today. Early studies did not include the benefits of revascularization, thrombolysis and protective drug therapy (Franklin 2004, Kovoor et al 2006, Zwisler et al 2005). The restriction of studies to exercise training also limits their applicability to comprehensive cardiac rehabilitation programmes, as the sum of a programme may be greater than its individual parts. Additionally, women and the elderly were often excluded from research (Witt et al 2004). Consequently, most of the research findings in the 1960s and 1970s are no longer directly applicable to the management of people following a MI. The meta-analyses of Oldridge et al (1988) and O’Connor et al (1989) which have been used extensively to promote cardiac rehabilitation are now of limited usefulness and have been superseded by more recent studies (Jolliffe et al 2000, Rees et al 2004, Taylor et al 2004).
Current research continues to suggest that rehabilitation programmes confer benefit to participants in all domains of their lives, with a focus on patient selection and organization of the programme. Unfortunately the methodology of some research studies is of low quality (i.e. small sample number, restricted age/gender, lack of randomization), which limits extrapolation of their findings to clinical practice. Another problematic area for interpreting research findings is the choice of outcome measures (Sanderson et al 2004). The importance of improvement in health-related quality of life is recognized in many studies, but there is lack of consensus as to the most appropriate measuring tool (Shephard & Franklin 2001, Taylor et al 1998). The SF-36 and SF-12 Health Status Questionnaires have been widely used, but concern has been expressed as to their ability to detect change when used for serial measurements (Ni et al 2000, Smith et al 2000). Some studies suggest anxiety and depression improve after a period of rehabilitation and have been commonly measured by the Hospital and Depression Scale (Zigmond & Snaith 1983), but this is thought to be relatively insensitive to change in this group of patients (McGee et al 1999). With the difficulties in selecting robust and appropriate outcome measures for quality of life issues, studies frequently use physiological or exercise endurance indices to measure responses to exercise training. However, these also have methodological issues concerning the influence of motivation and relevance of results (Ingle et al 2005). It has been suggested that a suitable measure for people with ICDs is the confidence to exert themselves physically (Sears et al 2004). Measurements of general activity, using pedometers, are considered to demonstrate a close relationship with changes in functional status (Evangelista et al 2005) and may be a more appropriate outcome for determining the response to rehabilitation programmes.
The organization of cardiac rehabilitation programmes is varied, with some focusing primarily on education and psychosocial intervention (Mendes de Leon et al 2006) and others offering only exercise training. The World Health Organization (WHO) (Frye 1993) definition of cardiac rehabilitation encompasses the concept of comprehensive cardiac rehabilitation programmes. Research suggests the inclusion of exercise training is a key element in eliciting benefits both in facilitating return to function and secondary prevention (Leon et al 1991, National Institute for Clinical Excellence 2003, Taylor et al 2004). There is considerable variation across and between patient groups in the type and setting of exercise training (Hansen et al 2005). Many studies have concentrated on aerobic exercise owing to concerns regarding the effects of resistance training on cardiac function, but there is growing evidence that a mix of endurance and strength exercises are both safe and optimize the benefits (Adams et al 2006, Delagardelle et al 2002, Jonsdottir et al 2005, Levinger et al 2005). Concern that some patients may develop myocardial remodelling, as a result of participating in early activity (Jugdutt et al 1988, Kloner & Kloner 1983), has been alleviated by other work indicating that exercise does not contribute to the onset of remodelling (Cannistra et al 1999, DuBach et al 1997, Giannuzzi et al 1993, Myers et al 2000). The benefits of low- intensity exercise in the early stages of a programme are thought be similar to those of higher intensity (Blumenthal et al 1988, Goble et al 1991, Worcester et al 1993) and adoption of low-intensity exercise would facilitate both adherence and safety. Women have previously been under-represented in cardiac rehabilitation and may require specific programmes, based in the community or at home, to improve their uptake and adherence (Sanderson & Bittner 2005, Todaro et al 2004, Witt et al 2004). Some studies recommend inspiratory muscle training in people with chronic heart failure (Dall’Ago et al 2006).
The duration of the period of exercise training is influenced by many factors, such as physiological and psychological state (Kovoor et al 2006), objectives and adherence. Secondary prevention requires a prolonged period of training (Brubaker et al 2000) and adherence and uptake to a programme may be influenced by social support (Husak et al 2004) age, gender (Todaro et al 2004) and location of training (Grace et al 2005). The lack of consensus on an optimal period of exercise training for different groups of patients and an absence of a robust prediction model to determine the magnitude of benefit each patient may receive from a rehabilitation programme (Pierson et al 2004, Shen et al 2006) places an emphasis on assessment, joint goal setting and regular re-evaluation of response. Few studies have included a prolonged follow-up period, but any benefits appear to be quickly lost on cessation of regular exercise training.
The diversity of programmes and client groups makes it difficult to determine the cost-effectiveness of cardiac rehabilitation and this is compounded by differences in health provision among countries where this has been investigated. A survey within the UK calculated the cost per patient to be of the order of £490 (Beswick et al 2004) while an American review had higher average costs of $2054 and determined that participation prolonged survival by an additional 1.82 years at a cost of $1773 per/life-year saved (Georgiou et al 2001). It has been suggested that cardiac rehabilitation is more cost-effective than thrombolytic therapy and coronary bypass surgery (Ades et al 1997).
There has not been any reported increase in adverse events during either supervised or unsupervised exercise training following the inclusion of people who were previously considered a high risk: i.e. CHF and ICDs (Davids et al 2005, Fitchet et al 2003, Pashkow et al 1997) However, there is a paucity of information on the occurrence of adverse events outside research studies, which usually include highly selected patients who undergo formal exercise tests.
PROVISION OF CARDIAC REHABILITATION
Goals
Cardiac rehabilitation should meet the emotional, educational and physical needs of cardiac patients and their families in the acute hospital phase, through outpatient care and long-term follow-up in the community. It should be an integral part of cardiological management, with common goals to:
Cardiological management involves assessment, risk stratification (the ‘risk’ of further cardiac events), diagnostic testing, drug therapy and revascularization interventions, e.g. percutaneous coronary intervention (PCI) and CABG. There should be close collaboration between cardiology and rehabilitation professionals on risk stratification, levels of prescribed exercise and discussion of treatment plans for symptomatic patients.
Rehabilitation should combine exercise with patient education and counselling, which provide:
Patients should be offered an individually tailored package from this menu of care, according to their needs and preferences (Scottish Intercollegiate Guidelines Network 2002). Individual packages are usually described as spanning four phases of care (Coats et al 1995):
Phase I in-hospital period (average 3–5 days)
Phase II immediate post-discharge/convalescence stage (2–6 weeks)
Phase III supervised outpatient programme (6–12 weeks)
Phase IV long-term maintenance programme in the community.
However, there is now less emphasis on a fixed time scale; patients are increasingly offered a more seamless transition of care between outpatient follow-up and long-term lifestyle maintenance in the community.
Considerable variation exists in the model, timing, content and delivery of outpatient cardiac rehabilitation. Rehabilitation may be offered in supervised groups, within a hospital or community setting, or as part of a home-based package. Some programmes include education, psychosocial and exercise components while others provide only exercise training; some programmes are confined to 4–6 weeks’ duration while others last for up to a year. In order to maximize benefit and adherence, the rehabilitation professional must match the appropriate model of care to each patient’s individual needs. Increasing use of national clinical guidelines and audit tools have led to increased standardization, quality and effectiveness of cardiac rehabilitation services (American Association of Cardiovascular and Pulmonary Rehabilitation 2004, American College of Sports Medicine 2006, Fletcher et al 2001, Leon et al 2005, Scottish Intercollegiate Guidelines Network 2002).
To whom should cardiac rehabilitation be made available?
Cardiac rehabilitation should be offered to all cardiac patients who would benefit. Traditionally programmes have been targeted at post-MI and CABG patients with limited and variable service provision for patients following PCI, with angina, CHF or those who have undergone cardiac transplantation. Uptake of service is often poor among women, multi-pathology patients, some ethnic groups and the elderly. As a consequence, issues relating to access, distance, timing and flexibility of cardiac rehabilitation programmes are crucial considerations when trying to optimize service provision for such under-represented groups.
By whom should cardiac rehabilitation be delivered?
A broad spectrum of care requires the combined skills and close collaboration of a multidisciplinary team of professionals. The team should be led by a cardiologist and include nursing, physiotherapy, dietetic, occupational therapy, pharmacy and psychology staff, who have specialist training in cardiology and rehabilitation. Additional input may be required from social services and vocational guidance staff. Continuation of care in the community includes the primary healthcare team (principally general practitioner and practice nurse), Phase IV exercise instructor and possibly attendance at a cardiac patient support group. Long-term risk factor monitoring and management, coupled with a regular activity programme, is promoted at Phase IV to reinforce the need for ongoing secondary prevention.
Exercise rehabilitation should be delivered by a team of clinical and exercise specialists who are skilled in cardiovascular assessment, risk stratification, patient monitoring, exercise prescription, goal setting and behavioural management. The team needs to be able to deal with concurrent medical and psychosocial issues and combine the art and science of exercise prescription and delivery, i.e. the art of integrating strategies for behaviour change in order to enhance exercise compliance and long-term adherence, with the science of exercise prescription (American College of Sports Medicine 2006). There should also be joint working and close liaison with British Association of Cardiac Rehabilitation (BACR) Phase IV instructors (or their equivalent) who accept patients for long-term exercise in the community. Physiotherapists are key members of the team as they have specialist skills in the assessment, exercise prescription and rehabilitation management of multi-pathology patients, as well as being health educators and exercise advisors (Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006, Jolliffe et al 2000). The Association of Chartered Physiotherapists Interested in Cardiac Rehabilitation (ACPICR) in the UK has developed guidelines for practice (Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006) and a competency document outlining the physiotherapist’s role and required knowledge, skills and standard of performance in cardiac rehabilitation.
BENEFITS OF EXERCISE TRAINING
Improved exercise capacity
The development of cardiovascular endurance is the primary objective for CHD patients. Endurance training, defined as any activity which uses large muscle groups, can be sustained for a prolonged period and is rhythmic and aerobic in nature, results in an increase in maximal oxygen uptake (VO2max), i.e. the highest rate of oxygen consumption attainable during maximal exercise. Maximal oxygen uptake is limited:
 peripherally, in particular by the capacity of skeletal muscle to extract oxygen from the blood. This is represented as the difference between the oxygen content of arterial blood and mixed venous blood (arteriovenous oxygen difference
peripherally, in particular by the capacity of skeletal muscle to extract oxygen from the blood. This is represented as the difference between the oxygen content of arterial blood and mixed venous blood (arteriovenous oxygen difference 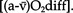 Consequently an increase in
Consequently an increase in  O2max depends on the potential for inducing central and/or peripheral adaptations.
O2max depends on the potential for inducing central and/or peripheral adaptations.Peripheral changes
Training-induced changes within skeletal muscle, which contribute to increased extraction and utilization of oxygen, include:
In cardiac patients, the increase in VO2max is attributed predominantly to peripheral adaptation. Central changes are associated with prolonged periods of high-intensity training and although in selected patients central changes have been provoked (Ehsani et al 1986, Schuler et al 1992), the high intensity of the training regimen would be inappropriate for the heterogene- ous group of patients eligible for cardiac rehabilitation programmes.
Consequences of an increase in maximal oxygen uptake
The significance of an increase in VO2max for cardiac patients is not that it permits a higher level of maximal effort (as this is rarely demanded in everyday life) but that repeated submaximal activities of daily living constitute a smaller percentage of the increased maximal capacity and therefore impose relatively less physiological stress. This is reflected in a reduction in heart rate (attributed to both increased vagal tone and reduced sympathetic outflow), blood pressure and plasma catecholamine concentrations at rest and at submaximal workloads. Since myocardial oxygen consumption (MVO2) is determined by heart rate and systolic blood pressure (referred to as rate pressure product (RPP) or double product), a reduction in either or both delays the onset of ischaemia and lessens the potential for arrhythmias. A further benefit of the training-induced bradycardia is that, at any reference submaximal workload, the period of diastole is extended and, since 80% of coronary blood flow occurs during the relaxation phase of the cardiac cycle, myocardial perfusion is significantly enhanced.
Risk factor modification
In cardiac patients exercise may have an important secondary prevention role. The rationale for aggressive risk factor modification, as part of optimal care of coronary heart disease patients, is based on the premise that the factors that contribute to initial development of disease will also influence the progression of established disease and the likelihood of future events. The ‘acute’ effects of each bout of exercise in healthy people include:
 changes in lipoprotein metabolism with consequent increased synthesis of high-density lipoprotein (HDL)
changes in lipoprotein metabolism with consequent increased synthesis of high-density lipoprotein (HDL)These effects all relate to local changes in the previously exercised muscle and are evident even after light to moderate exercise, suggesting that a general increase in physical activity is likely to contribute to the patient’s continued well-being and to the reduced mortality and morbidity associated with exercise rehabilitation.
In addition, exercise training is known to reduce known triggers for cardiac events including:
EXERCISE PRESCRIPTION
Principles of exercise prescription
When individual bouts of exercise are repeated regularly, and in accordance with established principles of training, a series of longer-term cardiovascular and metabolic adaptations occur; for instance, as described earlier, an increase in VO2max results from aerobic endurance training.
The principles of exercise training are:
Individuality – heredity plays a major part in how quickly and to what extent an individual’s body adapts to a training programme: i.e. no two individuals (other than identical twins) will exhibit the same adaptations in response to the same training programme.
Progressive overload – overload refers to placing greater demands on the body than it is accustomed to, thereby provoking adaptations. In order to continue to stimulate training adaptations, the overload must be progressively increased.
Regression or reversibility – this principle is often referred to as the principle of ‘use it or lose it’: i.e. if the stimulus to change (the overload) is withdrawn, the adaptations conferred will diminish until the level of functional capacity is once again sufficient to meet only the demands imposed by general activities of daily life.
Specificity – the adaptations conferred by training are highly specific to:
The effectiveness and appropriateness of all exercise prescriptions will depend upon manipulation of the FITT principle: i.e. the frequency, intensity, time and type of training.
Intensity of exercise
The intensity of exercise is a critical issue because vigorous activity carries a greatly increased risk of precipitating adverse events such as myocardial infarction or arrhythmias (Willich et al 1993). Frequent, moderate-intensity exercise is recommended for CHD patients since it will optimize benefits without increasing the risk of adverse events (Dafoe & Huston 1997). For individuals with greatly diminished functional capacity, several short bouts (as little as 5–10 minutes) throughout the day may be advisable. There are a number of established methods for prescribing and monitoring intensity, which may be used separately or in combination with one another.
Use of heart rate
Ideally, training heart rate is based on information derived from a maximal or symptom-limited exercise electrocardiogram test (ETT) (Box 14.1). Where a maximal test has been achieved, training heart rate should be set at 60–75% of maximal heart rate (HRmax). If the test was symptom limited, training intensity should be set at 10–20 beats per minute (bpm) below the heart rate at which symptoms were apparent and the patient’s heart rate should be monitored throughout each exercise session. ECG test information is, however, not always available to health professionals. In the absence of test data or if, for diagnostic purposes, a patient performs the exercise test ‘off-medication’, other methods for establishing appropriate training intensity have to be used.
Box 14.1 Exercise tolerance testing
In the cardiology setting, an exercise ECG using an incremental protocol is the most common method for determining cardiac perfusion and function. Its major applications are:
 measurement of functional capacity – on which advice about activities of daily living and a formal exercise prescription may be based
measurement of functional capacity – on which advice about activities of daily living and a formal exercise prescription may be based measurement of acute exercise responses including blood pressure, heart rate, ventilatory responses and detection of exercise-induced arrhythmias.
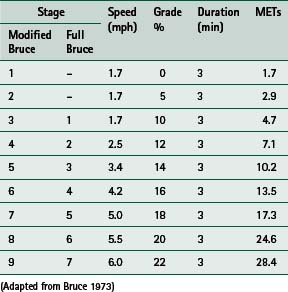
measurement of acute exercise responses including blood pressure, heart rate, ventilatory responses and detection of exercise-induced arrhythmias.Numerous exercise protocols have been developed which utilize a variety of different exercise modes, but in the United Kingdom an incremental treadmill protocol is the traditional test mode. Before acceptance into a Phase III programme, usually 2–6 weeks post MI, a symptom-limited test (i.e. the patient continues until signs or symptoms, which necessitate test termination, are evident) is customary; the Bruce protocol is the most common. Submaximal tests that use a predetermined endpoint such as an age-predicted maximum heart rate are usual before discharge. The modified Bruce protocol is the most commonly used since it introduces two preliminary, less strenuous stages (Table 14.1).
Criteria for terminating an exercise test include:
 marked drop in systolic blood pressure (>20 mmHg) indicating poor left ventricular function or severe coronary disease
marked drop in systolic blood pressure (>20 mmHg) indicating poor left ventricular function or severe coronary disease patient fatigue and/or excessive breathlessness at low workloads, which may simply indicate poor functional capacity but may also be suggestive of serious problems such as heart failure.
patient fatigue and/or excessive breathlessness at low workloads, which may simply indicate poor functional capacity but may also be suggestive of serious problems such as heart failure.In general an exercise ECG test is considered to be negative if haemodynamic responses to the increasing workload are normal and the patient satisfactorily completes a workload equivalent to the second stage of the full Bruce protocol (7 METs). The test is considered positive if the patient is symptomatic at low workloads, if there are significant ECG changes or there is an inappropriate heart rate/blood pressure response to the incremental workload.
Age-adjusted predicted maximal rates can be used (220 bpm minus age in years is one formula) and the training heart rate set at 60–75% of the predicted maximum; this is equivalent to 40–65% VO2max. However, the standard deviation (SD) for maximal heart rate during exercise is ± 10 bpm and some individuals will, therefore, have an actual maximum heart rate 20 bpm higher or lower (2 SD above or below the population mean) than predicted. An alternative approach is to prescribe training at 40–65% of heart rate reserve (HRR – the difference between resting and maximal heart rate). This HRR approach (also known as the Karvonen method) is convenient since it is known that 40–65% of heart rate reserve is equivalent to about 40–65% of VO2max (60–75% of maximal heart rate) although, across the entire range of fitness levels, it is more closely linked to the percentage of oxygen uptake reserve (VO2R): i.e. the difference between resting oxygen consumption and maximal oxygen consumption (American College of Sports Medicine 2006). An example of the HRR method for calculating training heart rate is shown below for an individual with a resting heart rate of 60 bpm and maximal heart rate of 150 bpm.
150 − 60 = 90 (heart rate reserve, HRR)
Training heart rate = 40−65% of heart rate reserve + resting heart rate (RHR)
90 × 0.40 = 36 + 60 (RHR) = 96 bpm (40%)
90 × 0.65 = 59 + 60 (RHR) = 119 bpm (65%)
Note: The formula is intended for use with known maximal heart rates. In the absence of these data, the substitution of age-adjusted predicted maxima introduces the same potential for error as previously mentioned. Consequently, any prescription that is based on predicted maximal heart rates should be used in conjunction with a rating of perceived exertion scale (RPE).
Since the relationship between exercise intensity and the per cent of maximal heart rate is preserved in patients on beta-blockers, the above formula can be adopted for calculating a training heart rate for this group but the maximal or peak heart rate must be established from an exercise test performed on medication.
Use of rating of perceived exertion
Cardiorespiratory and metabolic variables are strongly related to perceived exertion, which is accepted as a valid and reproducible indicator of the intensity of steady-state exercise. Physiotherapists and exercise specialists working in a cardiac rehabilitation setting are recommended to familiarize themselves with the scales of perceived exertion developed by Borg (1998). The Borg 15-point scale and the Borg CR10 scale of perceived exertion, together with patient instructions, are published in the Appendix of Borg’s Perceived Exertion and Pain Scales (Borg 1998). In order to preserve the validity and reproducibility of these scales, their format should not be altered and the patient instructions should be closely followed.
On the 15-point scale a rating of 12–13 (equivalent to 3–4 on the Borg CR10 scale) corresponds to approximately 40% of heart rate reserve or VO2max (60% HRmax). A rating of 15 on the 15-point scale (equivalent to 5–6 on the Borg CR10 scale) corresponds to approximately 65% of heart rate reserve or VO2max (75% HRmax).
Use of metabolic equivalent values (METs)
Exercise may also be regulated by choice of activities according to their known MET (metabolic equivalent) values (for which tables are available in most exercise physiology texts). If an individual assesses walking at 3 miles per hour (mph) as 12–13 on the Borg RPE scale (corresponding to 60% of VO2max), then activities of comparable MET value can be prescribed in the knowledge that they will present an appropriate training stimulus. Knowledge of MET values is also important in terms of excluding those activities that might pose a risk to certain individuals. Skipping (8–12 METs) or freestyle swimming (9–10 METs), for example, would be entirely inappropriate for someone with a peak capacity of 7 METs.
Some activities have a wide range of MET values, while others are relatively constant between individuals, mainly because they permit little variation in individual execution, e.g. there is very little difference in the way individuals walk or cycle. In contrast, there can be great variation in the way ‘free-moving’ activities such as dancing, skipping or rebounding on a mini-trampoline are executed. Because precise control of the exercise prescription in a cardiac population is necessary (particularly in early recovery post-event and for stable angina patients) activities that can be maintained at prescribed workloads and which permit uniform modification, e.g. altering the speed of walking or jogging or the resistance on a cycle ergometer, are preferred to those that are not amenable to standardized prescription.
Regardless of the objective method used for monitoring intensity, it is important to observe individuals for signs of excessive breathlessness, loss of quality of movement, unusual pallor or excessive sweating, all of which are inappropriate responses to moderate levels of exertion. Indications for ceasing exercise and contraindications to initiating exercise are included in the section on programme implementation.
Type of training
The inclusion of a variety of training modes within the individual prescription or the class format will minimize the incidence of overuse injuries, maximize peripheral adaptation (as, for example, when activities which require a contribution from both upper and lower body musculature are included) and increase patient motivation and adherence.
It is well documented that CHD patients who expend about 250–300 kcal per session and 1000–1500 kcal per week in additional physical activity will improve their aerobic capacity by 15–30% over a 4–6-month period (Balady et al 1994). There appears to be a continuous gradient in the benefits conferred and there is evidence that a minimum of 1600 kcal per week may halt the progression of CHD, whereas atherosclerotic regression may be achieved with a weekly energy expenditure of about 2200 kcal (Hambrecht et al 1994). Within the recommended ranges of frequency, intensity and time (or duration) of training, similar conditioning effects can be expected from any programme that realizes comparable weekly energy expenditure. Consequently the FITT components may be adjusted to provide an optimal prescription for individuals of varying cardiovascular and general medical status.
Programme format
Warm-up
Preparation for activity in older adults and especially in the cardiac population must be more gradual than for apparently healthy individuals. Fifteen minutes devoted to the warm-up component is recommended (Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006). Low-impact, dynamic movements which use large muscle groups and which take all major joint complexes through their normal range of motion should be incorporated. A gradual increase in the size and range of movements performed will delay the onset of ischaemia by allowing adequate time for coronary blood flow to increase in response to the greater myocardial demand. Gradual increments in myocardial workload will also lessen the risk of arrhythmias, which can be a consequence of abrupt increases in demand and concomitant elevated sympathetic activity. As a guideline, individuals should be within 20 bpm below the lower end of their prescribed training heart rate range at the end of the warm-up or, if RPE is used in place of heart rate monitoring, a rating no higher than 3 on the Borg CR10 scale or 10–11 on the original scale.
Although evidence of the benefit is equivocal, preparing for exercise has traditionally included static stretches, which are performed after the pulse raising and mobility phase. Because static stretches are used, the need to maintain pulse rate and body temperature during this time must be addressed.
Aerobic conditioning
The type of aerobic activity used for conditioning may adopt a continuous or interval approach. Continuous training, as the name implies, involves uninterrupted activity usually performed at a constant submaximal intensity. Its advantage is the ease with which intensity may be prescribed and monitored. Walking, jogging, cycling, rowing, bench stepping and swimming all lend themselves to a continuous approach. Interval training entails bouts of relatively intense work separated by periods of rest or less intense activity. Its main advantage is that, especially for debilitated patients, the total volume of work accomplished is generally greater than when exercise is continuous; consequently the stimulus to physiological change is greater. In an older cardiac population, the transition from one activity to another also provides a time for social interaction and support, which probably aids long-term compliance.
In group rehabilitation programmes, various ap- proaches to circuit training have proved popular; an activity is undertaken for a fixed period of time after which participants all move onto a different activity. Depending on cardiac status and individual functional capacity, participants may be prescribed an interval approach in which relatively intense periods of activity are followed by a period of less intense exercise, or alternatively, they may undertake activities all of which are of similar intensity. Circuits can be performed using little or no equipment or, if sufficient equipment is available to accommodate the whole group, cycles, rowing machines, treadmills, etc., can be used throughout the exercise session. Two approaches to group training are provided; Figure 14.1 presents a circuit design that requires only minimal equipment and Box 14.2 provides an example of how a group circuit might operate when extensive equipment is available.
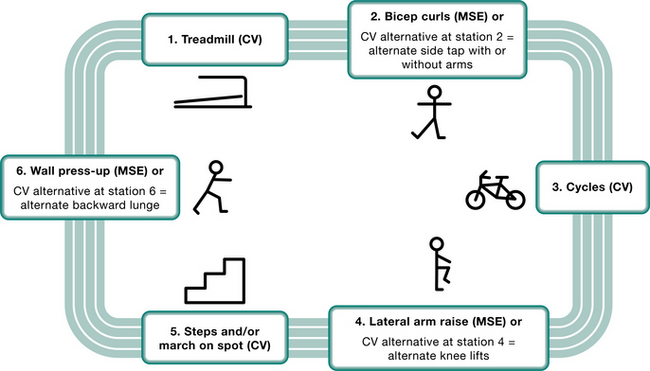
Figure 14.1 An example of interval-style circuit training suitable for a phase III cardiac rehabilitation programme. CV, cardiovascular work; MSE, muscle strength and endurance work.
Patients spend 2 minutes on stations 1, 3 and 5. At stations 2, 4 and 6, 1 minute is spent on the MSE work followed by 1 minute walking round the outside of the circuit. The patient’s attention needs to be drawn to the start of each 2-minute activity period and (for the benefit of those at the even MSE stations) when the first minute has passed. One full circuit constitutes 12 minutes and 2 circuits, 24 minutes.
Individualization and progression
The emphasis should be on improving cardiovascular endurance. Greater duration of cardiovascular work may be achieved if individuals (as and when appropriate) are encouraged to adopt some of the CV alternatives at even station numbers.
The intensity of the cardiovascular component may also be progressed at:
Progression on the alternative CV stations is achieved at:
The intensity of the MSE component may be progressed by introducing dumbbells or resistance bands or, in the case of station 6, taking the feet further from the wall or introducing backward extension of the arm at the elbow (triceps ‘kick-back’) using a dumbbell.
Determining appropriate workloads using fixed equipment
Although individuals will vary considerably in the amount of cardiovascular work they can achieve, it is suggested that for:
 the treadmill – a walking speed of 2.5–3.0 miles per hour (mph) or 4.0–4.8 kilometres per hour (kph) is prescribed with the gradient altered to elicit a heart rate response within the target training heart rate range
the treadmill – a walking speed of 2.5–3.0 miles per hour (mph) or 4.0–4.8 kilometres per hour (kph) is prescribed with the gradient altered to elicit a heart rate response within the target training heart rate range the cycle – 50–55 revolutions per minute (rpm) is prescribed with the resistance altered to elicit a heart rate response within the target heart rate range
the cycle – 50–55 revolutions per minute (rpm) is prescribed with the resistance altered to elicit a heart rate response within the target heart rate range the steps – a stepping speed to between 18 and 24 cycles per minute is prescribed (1 cycle – 4 footfalls; i.e. up, up, down, down) and the step height altered to elicit a heart rate response within the target training heart rate range.
the steps – a stepping speed to between 18 and 24 cycles per minute is prescribed (1 cycle – 4 footfalls; i.e. up, up, down, down) and the step height altered to elicit a heart rate response within the target training heart rate range.The circuit when completed twice provides a minimum of 12 minutes CV work and a maximum of 24 minutes (if all CV alternatives at even numbered stations are used).
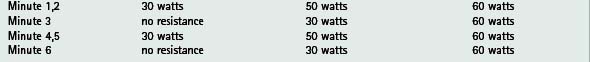
Box 14.2 An example of circuit training using gym equipment in which both interval and continuous training may be incorporated according to individual need
Treadmill for 6 minutes – speed (kph) and gradient (% grade) is individually prescribed
Cycle for 6 minutes @ 50–60 rpm – resistance (watts) individually prescribed
Stepping for 3 minutes – height of step (m)and rate of stepping (rpm) individually prescribed
Note: a cycle is 1st foot up, 2nd foot up, 1st foot down, 2nd foot down
Repeat each activity to achieve a total conditioning period of 30 minutes or repeat selected activities in accordance with individual needs and abilities.
Note: On a treadmill and during stepping, relative VO2 (ml O2/min) may be estimated on the basis of speed and gradient or speed and height of stepping, respectively, because the individual is carrying his own bodyweight and this contributes to the amount of work performed. On a cycle body weight has no influence on the cycle workload. It is important to start exercise at a relatively low level and to adjust the prescription in accordance with training heart rates and rating of perceived exertion.
kg, kilograms; kph, kilometres per hour; m, metres; rpm, rate per minute in cycles (1 cycle of stepping is 4 footfalls, i.e. up, up, down, down)
In the design shown in Figure 14.1, participants spend a fixed time (ranging from 30 seconds to 2 minutes) at ‘aerobic/cardiovascular (CV) stations’ and either rest or perform a lower intensity activity before moving on to the next aerobic station. The lower intensity or ‘active recovery’ stations are usually designed to increase the strength and endurance of specific muscle groups (MSE), e.g. triceps, pectorals, trapezius, used in activities of daily living. Individualization of the cardiovascular component of the programme is achieved through variation in:
In general the duration of activity is extended before increasing the intensity.
Exercises involving a recumbent position are discouraged because:
 following vigorous activity, the increase in venous return on lying down enhances preload and thereby myocardial workload, which increases the risk of arrhythmias and angina in some individuals
following vigorous activity, the increase in venous return on lying down enhances preload and thereby myocardial workload, which increases the risk of arrhythmias and angina in some individuals on return to an upright position the potential for orthostatic hypotensive episodes is dramatically increased in cardiac patients, most of whom are on medication that lowers blood pressure, e.g. beta-blockers and angiotensin-converting enzyme (ACE) inhibitors. Consequently, it is recommended that any recumbent work (e.g. for the abdominals or erector spinae) should be performed after completion of the circuit and a cool-down period.
on return to an upright position the potential for orthostatic hypotensive episodes is dramatically increased in cardiac patients, most of whom are on medication that lowers blood pressure, e.g. beta-blockers and angiotensin-converting enzyme (ACE) inhibitors. Consequently, it is recommended that any recumbent work (e.g. for the abdominals or erector spinae) should be performed after completion of the circuit and a cool-down period.Resistance training
Traditionally, training to increase strength (as opposed to endurance) of specific muscle groups was considered to be inappropriate for individuals with established heart disease. This was because resistance training is associated with an increase in arterial blood pressure, which increases myocardial workload. Some early studies suggested that the isometric component caused reduced ejection fraction, left ventricle wall motion abnormalities and increased incidence of arrhythmias. Further studies (Squires et al 1991, Williams 1994) have generally reported that cardiovascular and haemodynamic responses to resistance training in CHD patients and in normal subjects are similar and, because of increased diastolic pressure, may even enhance myocardial perfusion. However, in the United Kingdom, it is rare to incorporate strength training into clinically supervised programmes unless it is indicated for vocational reasons.
The American College of Sports Medicine (2006) advocates a single set of 10–15 repetitions to ‘moderate fatigue’ using 8–10 exercises. This is based on evidence that strength gains derived from one set are very similar to those reported when several are performed and adherence to programmes that are less time consuming is increased.
Cool-down
A period of 10 minutes is recommended for cool-down at the end of the cardiovascular component. This is because:
 there is an increased risk of hypotension in this group – for some this is a specific side effect of their medication. In addition, there is an age-related slowing of baroreceptor responsiveness which increases the risk of venous pooling following sustained exercise
there is an increased risk of hypotension in this group – for some this is a specific side effect of their medication. In addition, there is an age-related slowing of baroreceptor responsiveness which increases the risk of venous pooling following sustained exercise raised sympathetic activity during vigorous exercise increases the risk of arrhythmias during the immediate period following cessation of exercise.
raised sympathetic activity during vigorous exercise increases the risk of arrhythmias during the immediate period following cessation of exercise.The cool-down should incorporate movements of diminishing intensity and passive stretching of the major muscle groups used during the conditioning phase. Patient observation for up to 30 minutes after the exercise session is recommended. Many programmes follow the exercise session with an education or relaxation component, which affords the opportunity for extended observation and supervision of participants.
Progression of training
The duration, frequency or intensity of training can be increased in order to maintain the training stimulus. Ideally serial exercise testing will form the basis on which the prescription is modified, in order to ensure that it provokes physiological adaptation. In the absence of exercise testing, heart rate monitoring and rating of perceived exertion, at reference workloads, may be used to establish the appropriateness of increasing any of the three variables, either singly or in combination with one another. The way in which exercise prescription is progressed and the rate at which it is progressed will be highly variable between individuals with CHD and will be a function of many factors including age, severity of disease, motivation, dual pathology and compliance.
PROGRAMME IMPLEMENTATION
Initial convalescence phase
In-hospital activity component
Graduated mobilization of cardiac patients following acute MI, coronary artery bypass graft or unstable heart failure is initiated by nursing or physiotherapy staff on acute units as part of overall patient care. Activities in the first 24–48 hours are usually restricted to breathing exercises, simple arm/leg range of movement exercises and limited self-care activities. Patients are encouraged to sit out of bed, take short walks, shower and dress over the next 2–3 days, depending on their cardiac status. Although the physiotherapist may be involved in the earlier stages with a multi-pathology patient, their customary role is to supervise the patient’s mobilization plan, e.g. pre-discharge walk or stair assessments to determine individual exercise capacity, symptoms and/or limitations. By discharge, all patients should be conversant with the signs and symptoms of excessive exertion and be able to rate level of effort using, for example, the validated Borg scale (Borg 1998) or a locally developed scale. Both the patient and family should be advised on how to manage chest pain, symptoms of overexertion and encouraged to keep symptom diaries to discuss with staff at follow-up appointments.
A home exercise programme, which gives guidance on convalescence and household activities during the first 6 weeks and written advice on specific ‘do and don’t activities’, should be provided. Walking is recommended as the main mode of exercise and means of increasing functional capacity. An individual schedule suggesting distance/speed ratios and progressions and a home activity diary should be incorporated. The British Heart Foundation provides a wide range of professionally produced written material if none is available locally.
Although the starting level and progression of activity is always dependent on individual clinical status, symptoms and medical history, the following exercise prescription may be used as a guideline at discharge:
Type/mode: an interval rather than a continuous approach may be required initially; encourage walking and performance of sitting/standing functional activities, including active, non-resisted general arm and leg exercises
Frequency and Timing: initially, short intermittent bouts of 5–10 minutes of activity interspersed with rest periods, 2–3 times per day. Overall duration of activity may be progressed from 5 to 20 minutes, 1–2 times per day
Intensity: RPE <11 (6–20 Borg scale) or to individual tolerance, i.e. symptom limited by breathlessness/angina/fatigue at or below these suggested targets:
Immediate post-discharge convalescence phase
This home-based period may typically last from 2 to 6 weeks, depending on local protocols and resources as well as the patient’s fitness to attend the supervised outpatient programme. The immediate post-discharge phase is a time of high anxiety for patients and families and unfortunately rehabilitation services vary considerably and range from either no or limited contact to regular phone follow-up and home visits in some areas. A home programme for MI and CABG patients in the form of a workbook or exercise videos, The Heart Manual (Lewin et al 1992) and Road to Recovery – a British Heart Foundation publication (www.bhf.org.uk) – may be used as either a complete rehabilitation package or as an interim measure or adjunct to a programme. Contact from rehabilitation staff at this stage provides the opportunity to answer questions, discuss symptoms, reinforce home exercises and daily walking as appropriate: e.g. un- complicated post-MI and CABG patients should have increased the duration and frequency of activities and may be achieving up to 30 minutes of walking once or twice daily. Telephone contact also facilitates the review of risk factor modification goals and achievements.
Supervised outpatient exercise programme
The onset and duration of outpatient rehabilitation programmes vary considerably and are usually dependent on local resources. Programmes typically start any time from 3 to 6 weeks post-event and last for up to 6–12 weeks and include patients at different stages in their recovery. They may be hospital based, but there are increasing numbers of clinically supervised programmes in the community. For the purposes of this chapter, supervised exercise programmes are used as a model of exercise training.
The challenge for the exercise professional is to devise a safe and effective training programme that enables a patient to achieve a gradual transition from low-level convalescence activity through to an incremental exercise prescription. Although there is a perception that exercise training could be dangerous, the evidence is that rehabilitation programmes result in very few complications and the incidence of death is one per 1.3 million exercise hours (Franklin et al 1998, Van Camp & Peterson 1986, Vongvanich et al 1996). Indeed, if agreed guidelines and protocols for exercise training that relate to assessment, risk stratification, health and safety, patient induction, management and exercise prescription are implemented, then a safe, systematic approach, which minimizes risk and maximizes benefit, may be achieved (American Association of Cardiovascular and Pulmonary Rehabilitation 2004, American College of Sports Medicine 2006, Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006).
Assessment and risk stratification
Local protocols referring patients for exercise training should include appropriate screening and consent from a hospital physician or cardiologist. The following information should be available to the rehabilitation health professional as part of the assessment and risk stratification process:
 current cardiac status, e.g. site and size of the infarct, revascularization details, results of investigations, e.g. exercise tolerance test (see Box 14.1), echocardiogram
current cardiac status, e.g. site and size of the infarct, revascularization details, results of investigations, e.g. exercise tolerance test (see Box 14.1), echocardiogram cardiac history and relevant previous medical history, e.g. musculoskeletal problems, respiratory or neurological conditions
cardiac history and relevant previous medical history, e.g. musculoskeletal problems, respiratory or neurological conditionsClinical risk stratification, i.e. determining the prognosis and relative risk of future cardiac events and complications, as well as the risk of complications during exercise, should be carried out in accordance with AACVPR guidelines (American Association of Cardiovascular and Pulmonary Rehabilitation 2004; Fig. 5.1, page 63). The stratification of patients into low-, medium- or high-risk groups for the occurrence of events during exercise depends on their current cardiac status, including cardiac damage, complications and associated signs and symptoms. Patients who have a low ejection fraction consistent with impaired left ventricular function, serious arrhythmias or left ventricular failure are at greater risk of complications and future cardiac events and would be classified as high risk. Also the prognosis for these patients is much poorer than for the general cardiac patient; 50% of heart failure patients die within 4 years and in patients with severe heart failure 50% of those will die within 1 year (Swedberg et al 2005) compared with the mortality for patients in the first 4 weeks post-MI of 10–15% and 5% annually thereafter.
Patient safety during exercise is the main consideration for health professionals. The main risk to cardiac patients attending an exercise programme is ventricular fibrillation. When ‘predicting’ risk from clinical evidence and exercise ECG assessment, the patients who have suffered extensive cardiac damage, have residual ischaemia and demonstrate ventricular arrhythmias on exercise, for example, a patient with a significantly positive exercise ECG test (ETT) or poor functional capacity and low VO2 max, would be considered to be at higher risk of cardiac events and consequently prescribed a lower level, more cautious rehabilitation exercise programme. Limits for exercise prescription may be determined by onset of symptoms during the ETT, e.g. breathlessness or fatigue, and very importantly by evidence of ischaemia, with or without the presence of angina (silent ischaemia). Peak exercise prescription should always be set at least 10 bpm below the ischaemic threshold (American Association of Cardiovascular and Pulmonary Rehabilitation 2004, American College of Sports Medicine 2006). Distance achieved during a 6-minute walk test (6MWT) has also been used in risk assessment and exercise prescription, with a walking distance of less than 300m associated with poorer short-term survival.
Rehabilitation assessment and outcome measures
Assessment and outcome measures are essential, not only to determine risk stratification and functional capacity but also to set and evaluate the effectiveness of an exercise training programme, provide objective feedback to the patient and facilitate evidence-based practice. These measures can be used both as a baseline, entry assessment tool and as an exit outcome measure. They may include heart rate and blood pressure at rest, during exercise and at recovery, comparative ratings of perceived exertion (RPE) at given fixed workloads, bodyweight, body mass index, waist circumference measures, as well as measures of functional capacity. Direct measures of maximum functional capacity may be determined from VO2max derived from a cardiopulmonary exercise test (CPEX) and/or ETT. Indirect submaximal measures of functional capacity may be derived from the 6MWT (Steele 1994), shuttle walk test (Singh et al 1992) or Chester step test (Sykes & Roberts 2004). The measure of functional capacity used to set exercise prescription should then be successively monitored and adjusted to maintain the progressive, overload principle of exercise training. When using the walking distance achieved during a 6MWT to set an exercise prescription, the outcome may be used to calculate walk distance and speed for a home programme, the related METs level of daily activities and the general exercises which may be undertaken in a circuit training programme: e.g. 400 m walked in 6MWT is equivalent to 24 min per mile i.e. 2.5 mph pace equivalent to 2.9 METs.
Assessment
In addition to the initial screening and risk stratification on entry to the programme, there should be ongoing clinical assessment before each exercise session. Patients should be screened by the rehabilitation professional for changes and/or compliance with medication, symptoms and home activity levels. It is recommended that patients should not exercise if they are generally unwell, symptomatic or clinically unstable on arrival (Fletcher et al 2001), e.g. if they present with:
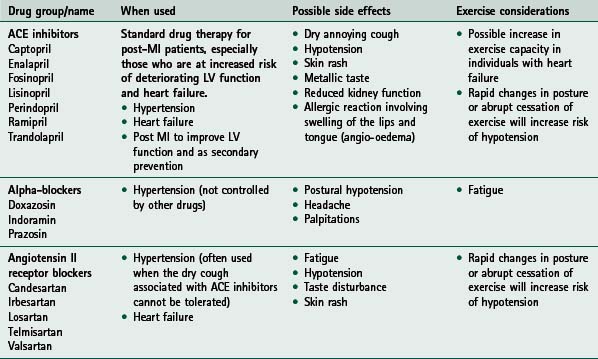
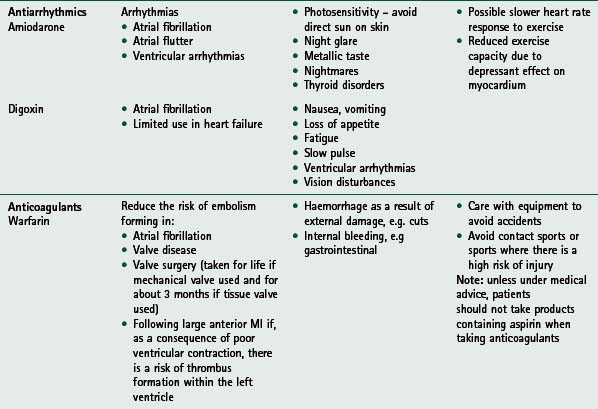
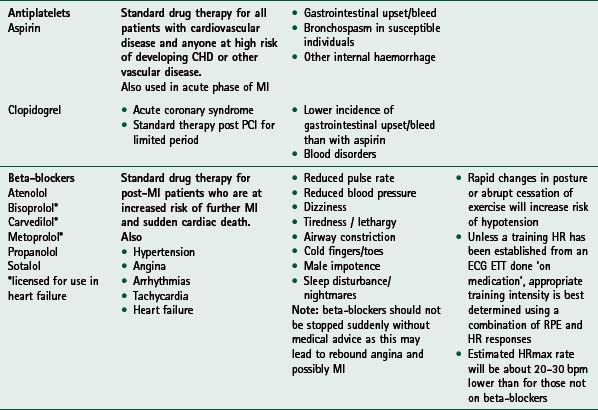
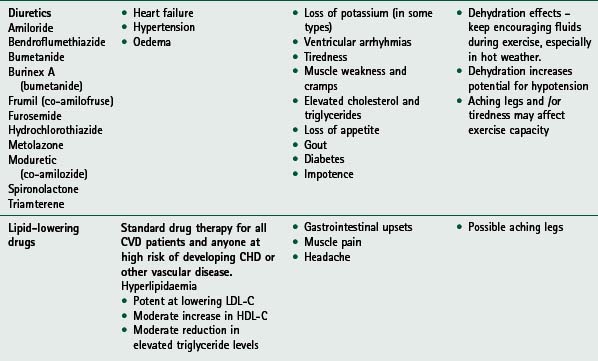
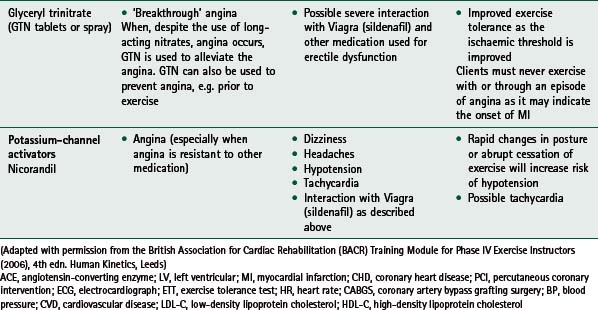
If any of these signs or symptoms is present, the patient should be seen by their general practitioner and/or cardiologist. Home activity and exercise goals should be adjusted appropriately and reviewed by rehabilitation staff before the patient restarts the exercise programme. For guidance on medication commonly used and prescribed for cardiac patients, and its associated relevance to exercise, see Table 14.2.
Programme delivery and management
Safe delivery of exercise also depends on patient supervision, appropriate education, induction and observance of health and safety protocols.
Patient supervision and staffing
 There should be an appropriate skill mix of staff with specialist training to lead and supervise exercise and deal with medical problems and emergencies.
There should be an appropriate skill mix of staff with specialist training to lead and supervise exercise and deal with medical problems and emergencies. There should be a minimum of two trained staff present with staff : patient ratio dependent on the risk stratification of patients in the group. Current recommended ratio to ensure safe monitoring/management of patients is 1 : 5; inclusion of higher-risk patients may require a higher ratio (Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006, Coats et al 1995, Scottish Intercollegiate Guidelines Network 2002).
There should be a minimum of two trained staff present with staff : patient ratio dependent on the risk stratification of patients in the group. Current recommended ratio to ensure safe monitoring/management of patients is 1 : 5; inclusion of higher-risk patients may require a higher ratio (Association of Chartered Physiotherapists in Cardiac Rehabilitation 2006, Coats et al 1995, Scottish Intercollegiate Guidelines Network 2002).Patient induction
An induction and education session should be conducted with each patient and cover:
Patients and their families should also be advised on educational issues such as:
Health and safety protocols
 There should be a local written policy, clearly displayed, for managing emergency situations, e.g. collapse of patient, and management of medical problems, e.g. chest pain, hypoglycaemic episode.
There should be a local written policy, clearly displayed, for managing emergency situations, e.g. collapse of patient, and management of medical problems, e.g. chest pain, hypoglycaemic episode. There should be rapid access to an emergency team either in hospital or via ambulance, with access to a telephone for raising emergency help.
There should be rapid access to an emergency team either in hospital or via ambulance, with access to a telephone for raising emergency help. Appropriate emergency equipment should be available. It should be regularly maintained in accordance with local protocols and staff should have regular practice in emergency drills and procedures.
Appropriate emergency equipment should be available. It should be regularly maintained in accordance with local protocols and staff should have regular practice in emergency drills and procedures. Venue access, emergency exits, toilet and changing facilities, lighting, floor surface and room space should be checked by staff to be safe and appropriate. There should be adequate space for a free exercise area and patient traffic around exercise room, and safe placement of equipment meeting recommended requirements, e.g. floor space for aerobic exercise of 1.8–2.3 m2 per patient and 0.6 m2 of space per individual using equipment (American Association of Cardiovascular and Pulmonary Rehabilitation 2004, Tharrett & Peterson 1997).
Venue access, emergency exits, toilet and changing facilities, lighting, floor surface and room space should be checked by staff to be safe and appropriate. There should be adequate space for a free exercise area and patient traffic around exercise room, and safe placement of equipment meeting recommended requirements, e.g. floor space for aerobic exercise of 1.8–2.3 m2 per patient and 0.6 m2 of space per individual using equipment (American Association of Cardiovascular and Pulmonary Rehabilitation 2004, Tharrett & Peterson 1997). Exercise class management
A skilled clinical exercise team will be able to create a safe, positive and non-intimidating environment in which a wide spectrum of patients are encouraged to participate and benefit from exercise. Supervised gym-based work or group exercise in a circuit training format are popular modes of delivering training. Each format requires rehabilitation professionals with excellent interpersonal and behaviour management skills to engage patients in exercise and develop their trust, confidence and participation. The exercise professional needs to establish an empathetic relationship with the patient and deal with emotional and psychological responses, which may vary from fearful, depressed, overdependent to aggressive, cavalier or in denial. Good interactive class management and group dynamics can create a positive atmosphere of social support and camaraderie which can result in a rewarding ‘care of the group by the group’ ethos and provide an opportunity to introduce exercise-related teaching points.
Exercise prescription and progression
The main objective of the exercise programme is to increase cardiorespiratory fitness through a cardiovascular and muscular endurance training programme. Patients report that the consequent improvement in functional capacity and strength enables them to perform activities with less effort, fewer symptoms, more confidence and an enhanced quality of life. The evidence and related pathophysiology, for endurance training in higher-risk patients with poor functional capacity, is that this is best achieved by increasing peripheral stimulus while minimizing cardiovascular stress (Swedberg et al 2005).
The principles of exercise training applied to a clinically supervised programme may be summarized as follows:
Type or mode
 Aerobic and muscular strength and endurance training that involves large muscle groups in dynamic movement, e.g. walk, cycle, circuit training.
Aerobic and muscular strength and endurance training that involves large muscle groups in dynamic movement, e.g. walk, cycle, circuit training. Initial adoption of an interval approach with eventual progression to low/moderate intensity continuous aerobic exercise.
Initial adoption of an interval approach with eventual progression to low/moderate intensity continuous aerobic exercise. Caution should be exercised over:
Caution should be exercised over:
Timing and progression
Intensity
 Aerobic exercise: 40–65% HRR or 60–75% maximal HR derived from an agreed age-estimated formula). A lower intensity prescription at 40–50% of functional capacity may be necessary for debilitated patients.
Aerobic exercise: 40–65% HRR or 60–75% maximal HR derived from an agreed age-estimated formula). A lower intensity prescription at 40–50% of functional capacity may be necessary for debilitated patients. Resistance training: 1 set of between 3–20 repetitions (e.g. 10–15) of 8–10 major muscle groups 2–3 times per week @ RPE 15–16 (stop 2–3 repetitions before volitional fatigue, while maintaining good technique).
Resistance training: 1 set of between 3–20 repetitions (e.g. 10–15) of 8–10 major muscle groups 2–3 times per week @ RPE 15–16 (stop 2–3 repetitions before volitional fatigue, while maintaining good technique).There should be close observation of all cardiac patients for up to 30 minutes post-exercise.
Exercise considerations for special cardiac groups
Considerations for prescribing and delivering exercise for various groups within the CHD population, e.g. patients with diabetes, hypertension and peripheral vascular disease, are summarized in Table 14.3. Exercise issues for specific cardiac patient groups, i.e. patients with heart failure and implantable cardioverter defibrillators, are described below.
Table 14.3 Considerations for prescribing exercise for special groups within the coronary heart disease population
g, gram; mmol/l, millimoles per litre; HR, heart rate; BP, blood pressure; SBP, systolic blood pressure; mmHg, millimetres of mercury; DBP, diastolic blood pressure; FITT, frequency, intensity, time and type of training; CV, cardiovascular; l, litre; PVD, peripheral vascular disease; ACSM, American College of Sports Medicine
Heart failure
Heart failure patients are among those deemed at highest risk of further cardiac events during exercise according to the AACVPR stratification criteria (American Association of Cardiovascular and Pulmonary Rehabilitation 2004), fulfilling one of the essential criteria for high-risk patients of an ejection fraction of less than 40%, but may additionally present with other criteria, e.g. significant symptoms at low levels of activity of less than 5 METs or the presence of abnormal haemodynamics with exercise testing. Quantifying functional capacity further stratifies relative risk; a peak VO2 of less than 10 millilitres of oxygen per kilogram of bodyweight per minute (<10 ml kg-1 min-1) is associated with a relatively higher risk, a peak VO2 of >18 ml kg-1 min-1 categorizes a patient at lower risk, while a walking distance of <300 m in the 6MWT is associated with poorer short-term survival (Cahalin et al 1996, Likoff et al 1987, Lipkin et al 1986, Swedberg et al 2005). When undertaking exercise training with higher-risk patients, rigorous patient assessment, individual exercise prescription and monitoring, coupled with safe management and delivery of exercise, are paramount (Hunt et al 2005, Pina et al 2003).
In addition to the previously discussed exercise contraindications and cautions that apply to cardiac patients, heart failure patients should report:
Issues relating to the monitoring and safety of exercise include:
 staff : patient supervision ratio should be reviewed dependent on higher risk stratification; consider 3 : 1
staff : patient supervision ratio should be reviewed dependent on higher risk stratification; consider 3 : 1 rigorous monitoring of individual patient’s heart rate, blood pressure and use of RPE (i.e. Borg CR 0-10 scale) and dyspnoea scales
rigorous monitoring of individual patient’s heart rate, blood pressure and use of RPE (i.e. Borg CR 0-10 scale) and dyspnoea scalesAdditional training considerations include:
 adopt an interval training approach with initial work phases of 1–6 minutes of activity and rest phases of 1–2 minutes (American College of Sports Medicine 2006)
adopt an interval training approach with initial work phases of 1–6 minutes of activity and rest phases of 1–2 minutes (American College of Sports Medicine 2006)Exercise issues following implantation of a cardioverter defibrillator
Exercise rehabilitation plays an important part in enabling patients to regain the confidence to resume activity following implantation of an ICD device (Lampman & Knight 2000). Significant benefits in cardiorespiratory fitness, confidence and psychological well-being have been demonstrated in patients participating in rehabilitation following implantation of an ICD device (Fitchet et al 2003). The following specific points should be considered when prescribing and delivering exercise for ICD patients:
Transition of patients to long-term community-based exercise provision
When medically and psychologically stable, CHD patients should progress from the clinically supervised rehabilitation environment to a community-based, long-term Phase IV exercise programme. They should demonstrate:
 the foundation of behavioural and lifestyle changes required for continued risk factor modification.
the foundation of behavioural and lifestyle changes required for continued risk factor modification.Rehabilitation staff discharging the patient to their general practitioner and Phase IV instructor should be satisfied that the patient is able to:
 recognize warning signs and symptoms and take appropriate action (e.g. stop/reduce exercise level, take glyceryl trinitrate)
recognize warning signs and symptoms and take appropriate action (e.g. stop/reduce exercise level, take glyceryl trinitrate) identify specific goals for long-term maintenance of lifestyle change and risk factor reduction, relating to own personal history
identify specific goals for long-term maintenance of lifestyle change and risk factor reduction, relating to own personal history take responsibility to monitor risk factors (i.e. smoking, blood pressure, cholesterol and diabetes) with their general practitioner and the practice nurse
take responsibility to monitor risk factors (i.e. smoking, blood pressure, cholesterol and diabetes) with their general practitioner and the practice nurse report results of any ongoing investigations and possible implications for exercise prescription to the Phase IV instructor.
report results of any ongoing investigations and possible implications for exercise prescription to the Phase IV instructor.A discharge communication should be provided to the patient’s general practitioner, summarizing rehabilitation outcomes, CHD risk factor status and future plans regarding exercise follow-up before the patient moves on to Phase IV. Either a similar version of this discharge summary or the recommended British Association of Cardiac Rehabilitation Information Sheet (www.bacrphaseiv.co.uk), for transition between Phases III and IV, should be completed and given to the patient so that he may take this information to the proposed Phase IV instructor.
CONCLUSION
Cardiac rehabilitation, and specifically exercise training, is an effective form of management for people with cardiac disease and is endorsed by numerous clinical guidelines. It has a large and increasing evidence base to support its implementation in a variety of settings and for a variety of cardiac conditions. Challenges remain to increase the uptake of all people who would benefit from participation in programmes and to ensure programmes and research evidence reflect changes in the medical management of this group of people.
Adams J, Cline MJ, Hibbard M, McCullough T, Hartman J. A new paradigm for post-cardiac event resistance exercise guidelines. American Journal of Cardiology. 2006;97:281-286.
Ades PA, Pashkow FJ, Nestor JR. Cost-effectiveness of cardiac rehabilitation after myocardial infarction. Journal of Cardiopulmonary Rehabilitation. 1997;17:222-231.
Albu J, Gottlieb SH, August P, et al. Modifications of coronary risk factors. American Journal of Cardiology. 2006;97(12):41-52.
American Association of Cardiovascular and Pulmonary Rehabilitation. Guidelines for cardiac rehabilitation and secondary prevention programmes, 4th edn., Champaign, IL: Human Kinetics, 2004.
American College of Sports Medicine. ACSMs guidelines for exercise testing and prescription, 7th edn., Baltimore: Williams and Wilkins, 2006.
Association of Chartered Physiotherapists in Cardiac Rehabilitation. Standards for the exercise component of the phase III cardiac rehabilitation. London: CSP, 2006.
Balady GJ, Fletcher BJ, Froelicher ES, et al. Cardiac rehabilitation programs. A statement for healthcare professionals from the American Heart Association. Circulation. 1994;90:1602-1610.
Beswick AD, Rees K, Griebsch I, et al. Provision, uptake and cost of cardiac rehabilitation programmes: improving services to under-represented groups. Health Technology Assessment. 2004;8:1-152.
Bittner V, Sanderson B. Cardiac rehabilitation as a secondary prevention centre. Coronary Artery Disease. 2006;17:211-218.
Blumenthal JA, Rejeski WJ, Walsh-Riddle M, et al. Comparison of high- and low-intensity exercise training early after acute myocardial infarction. American Journal of Cardiology. 1988;61:26-30.
Borg G. Borg’s perceived exertion and pain scales. Champaign, IL: Human Kinetics, 1998.
Brodie D, Bethell H, Bren S. Cardiac rehabilitation in England: a detailed national survey. European Journal of Cardiovascular Prevention and Rehabilitation. 2006;13:122-128.
Brubaker PH, Rejeski WJ, Smith MJ, et al. A home-based maintenance exercise program after centre-based cardiac rehabilitation: effects on blood lipids, body composition and functional capacity. Journal of Cardiopulmonary Rehabilitation. 2000;20:50-56.
Bruce RA. Principles in exercise testing. In: Naughton JP, Heuerstein HK, editors. Exercise testing and exercise training in coronary heart disease. New York: Academic Press, 1973.
Cahalin LP, Mathier MA, Semigran MJ, et al. The six minute walk test predicts peak oxygen uptake and survival in patients with advanced heart failure. Chest. 1996;110:325-332.
Cannistra LB, Davidoff R, Picard MH, Balady GJ. Moderate-high intensity exercise training after myocardial infarction: effect on left ventricular remodeling. Journal of Cardiopulmonary Rehabilitation. 1999;19:373-380.
Coats A, McGee H, Stokes H, Thompson D. BACR Guidelines for cardiac rehabilitation. Oxford: Blackwell Science, 1995.
Dafoe W, Huston P. Current trends in cardiac rehabilitation. Canadian Medical Association Journal. 1997;156:527-532.
Dall’Ago P, Chiappa GRS, Guths H, et al. Inspiratory muscle training on patients with heart failure and inspiratory muscle weakness. Journal of American College of Cardiology. 2006;47:757-763.
Davids JS, McPherson CA, Early C, et al. Benefits of cardiac rehabilitation in patients with implantable cardioverter-defibrillators: a patient survey. Archives of Physical Medicine Rehabilitation. 2005;86:1924-1928.
Delagardelle C, Feiereisen P, Autier P, et al. Strength/endurance training versus endurance training in congestive heart failure. Medicine and Science in Sports and Exercise. 2002;34:1868-1872.
Department of Health, 2000 National Service Framework: Coronary Heart Disease. Modern standards and service models. www.dh.gov.uk/en/Policyandguidance/Healthandsocialcaretopics/Coronaryheartdisease/DH_4108602 (last updated 14 April 2005, accessed 17 July 2007)
Department of Health, 2004 At least five a week: evidence on the impact of physical activity and its relationship to health
Department of Health, 2006 Coronary Heart Disease. www.dh.gov.uk/PolicyAndGuidance/HealthAndSocialCareTopics/CoronaryHeartDisease/fs/en (Accessed 17 July 2007)
DuBach P, Myers J, Dziekan G, et al. Effect of exercise training on myocardial remodeling in patients with reduced left ventricular function after myocardial infarction. Circulation. 1997;95:2060-2067.
Ehsani AA, Biello DR, Schultz J, et al. Improvement of left ventricular contractile function in patients with coronary artery disease. Circulation. 1986;74:350-388.
Evangelista LS, Dracup K, Erickson V, et al. Validity of pedometers for measuring exercise adherence in heart failure patients. Journal of Cardiac Failure. 2005;11:366-371.
Fitchet A, Doherty PJ, Bundy C, et al. Comprehensive cardiac rehabilitation programme for ICD patients - a randomized controlled trial. Heart. 2003;89:155-160.
Fletcher GF, Balady GJ, Amsterdam EA, et al. Exercise standards for testing and training: a statement for healthcare professionals from the American Heart Association. Circulation. 2001;104:1694-1781.
Franklin B. An alternative approach to the delivery of cardiac rehabilitation services: a ‘Hybrid’ model for patient care. Journal of Cardiopulmonary Rehabilitation. 2004;2:383-386.
Franklin BA, Bonzheim K, Gordon S, Timmis GC. Safety of medically supervised outpatient cardiac rehabilitation exercise therapy: a 16 year follow-up. Chest. 1998;114:902-906.
Frye BA. Review of the World Health Organization’s report on disability prevention and rehabilitation. Rehabilitation Nursing. 1993;18:43-44.
Georgiou D, Chen Y, Appadoo S, et al. Cost-effectiveness analysis of long-term moderate exercise training in chronic heart failure. American Journal of Cardiology. 2001;87:984-988.
Giannuzzi I, Tavazzi L, Temporelli PL, et al. Long-term physical training and left ventricular remodeling after anterior myocardial infarction: results of the Exercise in Anterior MI (EAMI) trial. Journal of the American College of Cardiology. 1993;22:1821-1829. for EAMI
Goble AJ, Hare DL, MacDonald PS, et al. Effect of early programmes of high and low intensity exercise on physical performance after transmural acute myocardial infarction. British Heart Journal. 1991;65:126-131.
Grace SL, McDonald J, Fishman D, Caruso V. Patient preferences for home-based versus hospital-based cardiac rehabilitation. Journal of Cardiopulmonary Rehabilitation. 2005;25:24-29.
Hambrecht R, Niebauer J, Marburger C, et al. Various intensities of leisure time physical activity in patients with coronary atherosclerotic lesions. Journal of Cardiopulmonary Rehabilitation. 1994;14:167-168.
Hansen D, Dendale P, Berger J, Meeusen R. Rehabilitation in cardiac patients: what do we know about training modalities. Sports Medicine. 2005;35:1063-1084.
Hunt SA, Abraham WT, Chin MH, et al. A report of the American College of Cardiology and American Heart Association (ACC/AHA) Task Force on Practice Guidelines ‘Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult - Summary Article’. Journal of American College of Cardiology. 2005;46:1116-1143.
Husak L, Krumholz HM, Qiutin Z, et al. Social support as a predictor of participation in cardiac rehabilitation after coronary artery bypass surgery. Journal of Cardiopulmonary Rehabilitation. 2004;24:19-25.
Ingle L, Shelton RJ, Rigby AS, et al. The reproducibility and sensitivity of the 6 minute walk test in elderly patients with chronic heart failure. European Heart Journal. 2005;26:742-751.
Jolliffe JA, Rees K, Taylor RS et al 2000 Exercise-based rehabilitation for coronary heart disease. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD001800. DOI: 10.1002/14651858.CD001800
Jonsdottir S, Anderson KK, Sigurosson AF, Sigurosson SB. The effect of physical training in chronic heart failure. European Journal of Heart Failure. 2005;8:97-101.
Jugdutt BI, Michorowski BL, Kappagoda CT. Exercise training after anterior Q wave myocardial infarction: importance of regional left ventricular function and topography. Journal of the American College of Cardiology. 1988;12:362-372.
Kloner RA, Kloner JA. The effect of early exercise on myocardial infarct scar formation. American Heart Journal. 1983;106(5, part 1):1009-1013.
Kovoor P, Lee AK, Carrozzi F, et al. Return to full normal activities including work after acute myocardial infarction. American Journal of Cardiology. 2006;97:952-958.
Lampman RM, Knight BP. Prescribing exercise training for patients with defibrillators. American Journal of Physical Medicine and Rehabilitation. 2000;79(3):292-297.
Leon AS, Certo C, Comoss P, et al. Scientific evidence of the value of cardiac rehabilitation services with emphasis on patients following myocardial infarction. Journal of Cardiopulmonary Rehabilitation. 1991;10:79-87.
Leon AS, Franklin BA, Costa F, et al. Cardiac Rehabilitation and Secondary Prevention of CHD: American Heart Association Scientific Statement. Circulation. 2005;111:369-376.
Levinger I, Bronks R, Cody DV, et al. Resistance training for chronic heart failure patients on beta-blocker medications. International Journal of Cardiology. 2005;102:493-499.
Lewin B, Robertson IH, Cay EL, et al. Effects of self-help post-myocardial infarction rehabilitation on psychological adjustment and use of health services. Lancet. 1992;339:1036-1040.
Likoff MJ, Chandler SL, Kay HR. Clinical determinants of mortality in chronic congestive heart failure secondary to idiopathic dilated or ischaemic cardiomyopathy. American Journal of Cardiology. 1987;59:634-638.
Lipkin DP, Scriven AJ, Crake T, Poole-Wilson PA. Six minute walking test for assessing exercise capacity in chronic heart failure patients. British Medical Journal. 1986;292:653-655.
McGee HM, Hevey D, Horgan JH. Psychological outcome assessments for use in cardiac rehabilitation service evaluation: a 10 year systematic review. Social Science and Medicine. 1999;48:1373-1393.
Mendes de Leon CF, Czajkowski SM, Freeland KE, Bang H, for ENRICHD Investigators. The effect of a psychosocial intervention and quality of life after an acute myocardial infarction. Journal of Cardiopulmonary Rehabilitation. 2006;26:9-13.
Myers J, Goebbels U, Dzeikan G. Exercise training and myocardial remodelling in patients with reduced ventricular function: one-year follow-up with magnetic resonance imaging. American Heart Journal. 2000;139:252-261.
National Institute for Clinical Excellence (NICE), 2001 Prophylaxis for patients who have experienced a myocardial infarction: drug treatment, cardiac rehabilitation and dietary manipulation. London
National Institute for Clinical Excellence (NICE), 2003 Chronic Heart Failure, Guideline No 5, Royal College of Physicians, London
Ni H, Toy W, Burgess D, et al. Comparative responsiveness of Short Form 12 and Minnesota Living With Heart Failure Questionnaire in patients With heart failure. Journal of Cardiac Failure. 2000;6:83-91.
O’Connor GT, Buring JE, Yusuf S, et al. An overview of randomized trials of rehabilitation with exercise after myocardial infarction. Circulation. 1989;80:234-244.
Oldridge NB, Guyatt GH, Fischer ME, Rimm AA. Cardiac rehabilitation after myocardial infarction. Combined experience of randomized clinical trials. Journal of the American Medical Association. 1988;260:945-950.
Pashkow FJ, Schweikert RA, Wilkoff BL. Exercise testing and training in patients with malignant arrhythmias. Exercise and Sport Sciences Reviews. 1997;25:235-269.
Pierson LM, Miller LE, Herbert WG. Predicting exercise training outcome from cardiac rehabilitation. Journal of Cardiopulmonary Rehabilitation. 2004;24:113-118.
Pina IL, Apstein CS, Balady GJ, et al. Exercise and heart failure - a statement from the American Heart Association Committee on Exercise, Rehabilitation and Prevention. Circulation. 2003;107(8):1210-1229.
Rees K, Taylor RS, Singh S, et al. Exercise based rehabilitation for heart failure. Cochrane Database of Systematic Reviews, Issue. 3, 2004. : CDOO3331
Sanderson KB, Bittner V. Women in cardiac rehabilitation: outcomes and identifying risk for dropout. American Heart Journal. 2005;150:1052-1058.
Sanderson KB, Southard D, Oldridge N. Outcomes evaluation in cardiac rehabilitation/secondary prevention programmes: improving patient care and programme effectiveness. Journal of Cardiopulmonary Rehabilitation. 2004;24:68-79.
Schuler G, Hambrecht R, Schlierf G, et al. Regular physical exercise and low-fat diet. Effects on progression of coronary artery disease. Circulation. 1992;86:1-11.
Scottish Intercollegiate Guidelines Network (SIGN), 2002 Cardiac rehabilitation: Guideline 57 www.sign.ac.uk/guidelines/fulltext/57/index.html (Accessed 17 July 2007)
Sears SF, Kovacs AH, Conti JB, Handberg E. Expanding the scope of practice for cardiac rehabilitation: managing patients with implantable cardioverter defibrillators. Journal of Cardiopulmonary Rehabilitation. 2004;24:209-215.
Shen BJ, Myers HF, McCreary CP. Psychological predictors of cardiac rehabilitation quality-of-life outcomes. Journal of Psychosomatic Research. 2006;60:3-11.
Shephard RJ, Franklin B. Changes in the quality of life: a major goal of cardiac rehabilitation. Journal of Cardiopulmonary Rehabilitation. 2001;21:189-200.
Singh SJ, et al. Developments of a shuttle walking test of disability in patients with chronic airways obstruction. Thorax. 1992;47:1019-1024.
Smith HJ, Taylor R, Mitchell A. A comparison of four quality of life instruments in cardiac patients: SF-36, QLI, QLMI and SEIQoL. Heart. 2000;84:390-394.
Squires RW, Muri AJ, Anderson LJ, et al. Weight training during phase II (early outpatient) cardiac rehabilitation: heart rate and blood pressure responses. Journal of Cardiac Rehabilitation. 1991;11:360-364.
Steele B, 1994 The six minute walk. In AACVPR Proceedings 9th Annual Meeting Portland, OR. AACVPR, Chicago, 383-388
Swedberg K, Cleland J, Dargie H. Task Force for the Diagnosis and Treatment of Chronic Heart Failure of the European Society of Cardiology Guidelines for the diagnosis and treatment of chronic heart failure: full text (update 2005). European Heart Journal. 2005;26:1115-1140.
Sykes K, Roberts A. The Chester step test - a simple yet effective tool for the prediction of aerobic capacity. Physiotherapy. 2004;90:183-188.
Taylor R, Kirby B, Burdon D, Caves R. The assessment of recovery in patients after myocardial infarction using three generic quality-of-life measures. Journal of Cardiopulmonary Rehabilitation. 1998;18:139-144.
Taylor RS, Brown A, Ebrahim S, et al. Exercise-based rehabilitation for patients with coronary heart disease: a systematic review and meta-analysis of randomized controlled trials. American Journal of Medicine. 2004;11:682-692.
Tharrett SJ, Peterson JA. ACSM’s health/fitness facility standards and guidelines. Champaign, IL: Human Kinetics, 1997.
Todaro JF, Shen BJ, Niaura R, et al. Do men and women achieve similar benefits from cardiac rehabilitation. Journal of Cardiopulmonary Rehabilitation. 2004;24:45-50.
Van Camp SP, Peterson RA. Cardiovascular complications of outpatient cardiac rehabilitation programmes. Journal of American Medical Association. 1986;256:1160-1163.
Vongvanich P, Paul-Labrador MJ, Merz C. Safety of medically supervised exercise in a cardiac rehabilitation centre. American Journal of Cardiology. 1996;77:1383-1385.
Williams MA. Exercise testing and training in the elderly cardiac patient. In: Current issues in cardiac rehabilitation series. Champaign, IL: Human Kinetics; 1994.
Willich SN, Lewis M, Lowel H, et al. Physical exertion as a trigger of acute myocardial infarction. New England Journal of Medicine. 1993;329:1684-1690.
Witt BJ, Jacobsen SJ, Weston SA, et al. Cardiac rehabilitation after myocardial infarction in the community. Journal of American College of Cardiology. 2004;44:988-996.
Worcester MC, Hare DL, Oliver RG, et al. Early programmes of high and low intensity exercise and quality of life after acute myocardial infarction. British Medical Journal. 1993;307:1244-1247.
Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatrica Scandinavica. 1983;67:361-370.
Zwisler A-DO, Schoul L, Soja AMB, et al. A randomized clinical trial of hospital-based, comprehensive cardiac rehabilitation versus usual care for patients with congestive heart failure, ischaemic heart disease or high risk ischaemic heart disease. American Heart Journal. 2005;150:899e7-899e16.