EXUDATIVE EPIDERMITIS (GREASY PIG DISEASE)
ETIOLOGY
The condition of exudative dermatitis is similar to staphylococcal scalded skin syndrome in humans associated with S. aureus.1
Staphylococcus hyicus is the cause of exudative epidermitis in suckling and weaned piglets.2 It also causes several other diseases sporadically in different animal species; bacteriuria in pigs, polyarthritis in pigs,3 abortion in pigs, flank biting and necrotic ear lesions,4 and pneumonia.5
In other species it has been associated with skin infections in horses, donkeys, and cattle, subclinical mastitis in cows and osteomyelitis in heifers.2
A second species, Staphylococcus chromogenes, is part of the normal skin flora of pigs,6,7 cattle, and poultry. It had been considered nonpathogenic until exudative epidermitis was associated with it in 2005.8 These strains also produced exfoliative toxin type B (ExLB), which was identified by PCR.
Etiology Staphylococcus hyicus
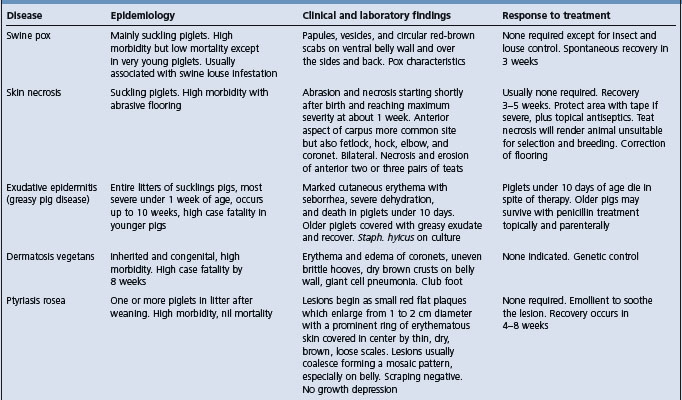
Epidemiology Affects suckling and weanling piglets under 6 weeks of age; peak incidence under 1 week of age. Morbidity 20–100%; case fatality 50–75%. Organism carried by sow
Signs Marked cutaneous erythema and pain, dehydration, extensive greasy exudate; peracute cases die; less severe cases may survive
Clinical pathology Bacterial culture of skin
Necropsy findings Exudative epidermitis; degenerative changes in kidney
Diagnostic confirmation Culture of organism
Differential diagnosis See Table 16.3
Treatment Penicillin parenterally
Control Sanitation and hygiene of pens. In outbreaks, isolate affected piglets and sows
EPIDEMIOLOGY
Occurrence
Most cases of exudative epidermitis occur in suckling and weaned piglets under 6 weeks of age, with a peak incidence in piglets under 1 week. Occasionally groups of pigs up to 3 months of age may be affected. Within litters the incidence is high, often all piglets being affected. The morbidity will vary from 20–100% and the case fatality rate from 50–75%. The organism has been isolated from the joint fluid of lame pigs affected with arthritis.3 In 28% and 26% of studies of cases of exudative epidermitis9,10 no cases of toxigenic S. hyicus could be detected. In a recent study of 314 cases in Denmark it was shown that 20% had exfoliatum toxin A, 33% had B, 18% had C, and 22% had D in 60% of cases of exudative epidermitis investigated.
Method of transmission
The source of the organism is unknown but the gilt or sow is probably an inapparent carrier. It can be isolated from the skin of healthy in-contact piglets and healthy sows.11 It can be frequently isolated from the vagina of prepubertal gilts and the majority of the litters from the same gilts may be colonized by the organism within 24 hours after farrowing.12 The maternal strains of S. hyicus persisted on the skin of the offspring piglets for the first 3 weeks of the piglets’ life – the critical period for outbreaks of exudative epidermitis.12 The organism has also been isolated from the atmosphere of buildings housing affected pigs. Bacteriophage typing of S. hyicus subsp. hyicus isolated from pigs with or without exudative epidermitis revealed two or more phage patterns in the isolates from each pig with the disease and a single-phage pattern in isolates from healthy pigs.13,14
Risk factors
Animal risk factors
Field evidence suggests that environmental stress of various kinds, including agalactia in the sow and intercurrent infection, predisposes to the disease. Lesions commonly develop first over the head, apparently in association with bite wounds, which occur when the needle teeth have not been cut or have been cut badly. Other factors include fighting following mixing of litters, excessive humidity over 70%, and following sarcoptic mange. The presence of the disease in a swine herd can account for a 35% reduction in the margin of output over feed and veterinary costs over a 2-month period.
Pathogen risk factors
Strains of S. hyicus can be divided into virulent and avirulent strains with regard to ability to produce exudative epidermitis in experimental piglets; both types of strain can be isolated simultaneously from diseased piglets.4 It has been shown that different types of S. hyicus expressing different types of toxin may be present in the same diseased pig.10
S. hyicus produces an exfoliative toxin that can be used to reproduce the disease.15,16 There are several toxins, including ExLA, ExLB, ExLC, ExLD, SHETa, and SHETb.17 The exfoliative toxins from isolates from different countries have recently been described.18 Strains of the organism isolated from a large number of Danish pig herds indicated different electrophoretic motility and plasma-mediated antibiotic resistance patterns.19 The antibiotic and plasmid profiles of strains isolated from pig herds may be a reflection of the use of antibiotics in those herds.20 Different types of toxin are produced.21-23
Recently the genes encoding for the exfoliative toxins SHETb, ExLA, ExLB, ExLC, and ExLD have been identified24 and sequenced.
The condition has been seen more frequently in cases of PRRS and PCV2 infections.25,26
The organism has been found as a frequent inhabitant of the skin of cattle and has been isolated from cattle with skin lesions.27,28 Naturally occurring lesions of dermatitis of the lower limbs of horses29 and similar lesions over the neck and back of donkeys30 have been recorded. Experimentally, the organism can cause lesions in horses similar to those of exudative epidermitis. A concurrent infection with Dermatophilus congolensis has also been reported.31
PATHOGENESIS
S. hyicus has cytotoxic activity for porcine keratinocyte cells in culture, which may indicate one of its virulence factors.31
The exfoliative toxins are actually epidermolysins32 that are active against desmoglein-1, which is a desmosomal cadherin-like molecule33 involved in cell-to-cell adhesion.34 The ExLs can cause blister formation in the porcine skin by digesting porcine desmoglein-1 in a similar way to exfoliative toxins of S. aureus.
The earliest lesion is a subcorneal pustular dermatitis involving the interfollicular epidermis. Exfoliation follows with sebaceous exudation and formation of a crust. In the well-developed case there is a thick surface crust composed of orthokeratotic and parakeratotic hyperkeratosis and neutrophilic microabscesses with numerous colonies of Gram-positive cocci.35
Although the principal lesion is an inflammatory–exudative reaction in the corium and upper layers of the epidermis, the disease is probably a systemic rather than a local one. Experimental infection of gnotobiotic pigs leads to dermatitis of the snout and ears, then the medial aspect of the thighs, the abdominal wall and the coronets. The lesions can be produced experimentally by using crude extracellular products and a partially purified exfoliative toxin.36
CLINICAL FINDINGS
The morbidity varies from 10–100% and the mortality from 5–90%, with an average of 25%.35
In the peracute form, which occurs most commonly in piglets only a few days of age, there is a sudden onset of marked cutaneous erythema, with severe pain on palpation, evidenced by squealing. Anorexia, severe dehydration, and weakness are present and death occurs in 24–48 hours. The entire skin coat appears wrinkled and reddened and is covered with a greasy, gray-brown exudate that accumulates in thick clumps around the eyes, behind the ears, and over the abdominal wall. In the less acute form, seen in older pigs 3–10 weeks of age, the greasy exudate becomes thickened and brown and peels off in scabs, leaving a deep-pink-colored to normal skin surface. There is no irritation or pruritus. In the subacute form, the exudate dries into brown scales that are most prominent on the face, around the eyes, and behind the ears. In a small percentage of pigs the chronic form occurs and the course is much longer; there is thickening with wrinkling of the skin and thick scabs that crack along flexion lines, forming deep fissures. Most peracute cases die, while piglets with the less severe forms will survive if treated. Some pigs are affected with ulcerative glossitis and stomatitis.
Abortion in a sow has been attributed to the organism.37
CLINICAL PATHOLOGY
Bacterial examination of skin swabs may reveal the presence of S. hyicus. A phage typing system can be used to determine the presence of virulent strains and to distinguish them from less virulent strains.14
NECROPSY FINDINGS
Necropsy of these dehydrated, unthrifty piglets often reveals a white precipitate in the renal papillae and pelvis. Occasionally this cellular debris causes ureteral blockage. Some piglets also have a mild ulcerative glossitis and stomatitis. Microscopically, there is separation of the cells of the epidermis in the upper stratum spinosum, exfoliation of the skin, erythema, and serous exudation. The crusting dermatitis features a superficial folliculitis and a hyperkeratotic perivascular dermatitis with intracorneal pustules and prominent bacterial colonies. Degenerative changes are visible in the renal tubular epithelium.
Samples for confirmation of diagnosis
• Bacteriology – samples of acute skin lesions (CULT). The organism forms 3–4 mm white, nonhemolytic colonies on blood agar. It is catalase- and mannitol-negative but hyaluronidase-positive
• Histology – formalin-fixed skin (multiple sites), kidney (LM).
A PCR is available but requires a pure culture and large numbers of organisms to be successful.
Exudative epidermitis may resemble several skin diseases of pigs of all age groups (Table 16.3). However, in exudative epidermitis there is no pruritus or fever. Careful gross examination of the lesions, particularly their distribution, the state of the hair shaft, the character of the exudate and the presence or absence of pruritus must be considered, along with skin scrapings and biopsies.
TREATMENT
Experimentally infected piglets respond favorably to a topical application of cloxacillin 10000 IU/g of lanolin base and 1% hydrocortisone combined with parenteral cloxacillin. Treatment must be administered as soon as the lesions are visible. Procaine benzylpenicillin at a dose of 20000 IU/kg BW intramuscularly daily for 3 days is also recommended. The antimicrobial sensitivities determined in one field investigation revealed that all isolates were sensitive to novobiocin, neomycin, and cloxacillin. Novobiocin may be the antimicrobial of choice since staphylococci are universally sensitive to this antibiotic.38 However, there is no available information on the efficacy of antimicrobials for naturally occurring cases of exudative epidermitis. A study has suggested that erythromycin, sulfathiazole, and trimethoprim may be the most useful drugs, whereas penicillin and tetracyclines may not be very useful.39 Naturally occurring cases in piglets under 10 days of age respond poorly, while older pigs recover with a skin wash using a suitable disinfectant soap. The most successful treatment is antibiotics and skin washing for a period of at least 5–7 days. It is also essential to make sure that there is sufficient dietary provision of zinc, biotin, fat, selenium, and vitamin E in the diet.
CONTROL
The infected accommodation should be cleaned, disinfected and left vacant before another farrowing sow is placed in the pen. Strict isolation of the affected piglets and their dam is necessary to prevent spread throughout the herd. Dead piglets should be removed promptly from the premises and in-contact sows should be washed with a suitable disinfectant soap. Maternal antibodies will protect piglets in the first few weeks of life.
Autogenous vaccines have been used with varying degrees of success. It is important to use a strain that produces the exfoliative toxin, so the recent development of PCRs that identify the genes for toxin development will ensure that the right isolate is used for the autogenous vaccine. It will also facilitate the development of a commercial vaccine.
A novel approach to the control is bacterial interference. Experimentally, the precolonization of the skin of gnotobiotic piglets with an avirulent strain of S. hyicus will prevent the experimental reproduction of the disease with the virulent strain of the organism.
1 Ladhani S, et al. Clin Microbiol Rev. 1999;12:224.
2 Wegener HC, et al. Can J Vet Res. 1993;57:119.
3 Hill BD, et al. Aust Vet J. 1996;73:179.
4 Mirt D. Vet Rec. 1999;144:92.
5 Teuscher E, Higgins R. Med Vet Quebec. 1990;20:64.
6 Devriese LA, et al. Int J Syst Bacteriol. 1978;28:482.
7 Saito K, et al. J Vet Med Sci. 1996;58:711.
8 Andresen LO, et al. Vet Microbiol. 2005;105:291.
9 Andresen LO, et al. FEMS Immunol Med Microbiol. 1998;20:301.
10 Andresen LO, et al. Vet Microbiol. 1999;68:285.
11 Bara MR, et al. Aust Vet J. 1993;70:256.
12 Wegener HC, et al. Epidemiol Infect. 1992;109:433.
13 Wegener HC, et al. Res Microbiol. 1993;144:237.
14 Wegener HC. J Vet Med Series B. 1993;40:13.
15 Tanabe T, et al. Vet Microbiol. 1996;48:9.
16 Andresen LO, et al. Microb Pathog. 1997;22:113.
17 Fudaba Y, et al. Microb Pathog. 2005;39:171.
18 Andresen LO. Vet Rec. 2005;157:376.
19 Wegener HC, Schwarz S. Vet Microbiol. 1993;34:363.
20 Noble WC, Allaker RP. Vet Rec. 1992;130:466.
21 Tanabe T, et al. Infect Immun. 1993;61:2973.
22 Sato H, et al. J Bacteriol. 2000;182:4096.
23 Andresen LO, et al. FEMS Immunol Med Microbiol. 1999;23:295.
24 Ahrens P, Andresen LO. J Bacteriol. 2004;186:1833.
25 Whitaker HK, et al. J Vet Diagn Invest. 1990;2:244.
26 Wattrang E, et al. Vet Microbiol. 2002;86:281.
27 Hazirika RA, et al. Res Vet Sci. 1991;50:374.
28 Hazirika RA, et al. Indian Vet J. 1991;68:163.
29 Devriese LA, et al. Equine Vet J. 1983;15:263.
30 Devriese LA, Thelisen M. Vet Rec. 1986;118:1091.
31 Allaker RP, et al. Vet Microbiol. 1991;26:161.
32 Prevost G, et al. Curr Opin Infect Dis. 2003;16:71.
33 Amagai M, et al. Nat Med. 2000;6:1275.
34 Hankawa YNM, et al. J Clin Invest. 2002;110:53.
35 Kim J, Chae CJ. Vet J. 2004;167:104.
36 Andresen LO, et al. Microb Pathog. 1993;15:217.
37 Onet GE, Pommer JL. J Am Vet Med Assoc. 1991;199:362.