CONTAGIOUS ACNE OF HORSES (CANADIAN HORSE POX, CONTAGIOUS PUSTULAR DERMATITIS)
Contagious acne of horses is characterized by the development of pustules, particularly where the skin comes in contact with harness.
EPIDEMIOLOGY
The disease is spread from animal to animal by means of contaminated grooming utensils or harness. An existing seborrhea or folliculitis due to blockage of sebaceous gland ducts by pressure from harness probably predisposes to infection. Inefficient grooming may also be a contributing cause.
Contagious acne is of limited occurrence and causes temporary inconvenience when affected horses are unable to work.
PATHOGENESIS
Infection of the hair follicle leads to local suppuration and the formation of pustules which rupture and contaminate surrounding skin areas. Occasional lesions penetrate deeply and develop into indolent ulcers.
CLINICAL FINDINGS
The skin lesions usually develop in groups in areas which come into contact with harness. The lesions take the form of papules which develop into pustules varying in diameter from 1–2.5 cm. There is no pruritus but the lesions may be painful to touch. Rupture of the pustules leads to crust formation over an accumulation of greenish-tinged pus. Healing of lesions occurs in about 1 week but the disease may persist for 4 or more weeks if successive crops of lesions develop.
CLINICAL PATHOLOGY
Swabs of the lesions can be taken to determine the presence of C. pseudotuberculosis.
TREATMENT
Affected animals should be rested until all lesions are healed. Frequent washing with a mild skin disinfectant solution followed by the application of antibacterial ointments to the lesions should facilitate healing and prevent the development of further lesions. Parenteral administration of antibiotics may be advisable in severe cases.
RHODOCOCCUS EQUI PNEUMONIA OF FOALS
Etiology Virulent strains of Rhodococcus equi
Epidemiology Sporadic disease of 1–5-month-old foals that is endemic on some farms. Foals are infected by ingestion or inhalation during first weeks of life
Clinical signs Pneumonia, fever, respiratory distress, cough, lack of nasal discharge, failure to thrive, multiple distended joints and uveitis. Occasionally diarrhea or septic osteomyelitis
Clinical pathology Leukocytosis, hyperfibrinogenemia, inflammatory cells in tracheal aspirate
Necropsy lesions Pulmonary consolidation and abscessation. Nonseptic polyarthritis
Diagnostic confirmation Culture of R. equi from tracheal aspirate
Treatment Erythromycin estolate or microencapsulated base 25 mg/kg every 8 hours orally, in combination with rifampin 5 mg/kg every 12 hours orally. Clarithromycin (7.5 mg/kg every 12 h orally) may be substituted for erythromycin
Control Insure adequate transfer of passive immunity. Decrease stocking density. Decrease environmental contamination by virulent strains of R. equi
ETIOLOGY
Rhodococcus equi is a Gram-positive, pleomorphic rod. The most important manifestation of R. equi infection is pneumonia in foals. It also causes pleuropneumonia, pneumonia, osteomyelitis, and abortion in immunocompromised and normal adult horses, abscesses that must be differentiated from tuberculosis in pigs and ruminants, and pneumonia in immunosuppressed humans.1-5 The organism is a natural inhabitant of soil, grows well at temperatures ranging from 10–40°C, and is readily isolated from the feces of herbivores and their environment. However, isolates of R. equi vary in virulence, with many isolates obtained from feces or soil not being pathogenic.6
There are a large number of virulent strains of R. equi, based on pulsed-field gel electrophoresis of chromosomal DNA.7,8 While there is evidence of clustering of strains on farms and on most farms 1 or 2 strains predominate, there is little evidence of marked regional variations in prevalence of strains of virulent R. equi.7,8 Only rarely will it be possible to link infections to a given site or region on the basis of analysis of chromosomal DNA.7
Virulence of R. equi is dependent upon the ability of the organism to enter, survive in and replicate in macrophages. Virulence is associated with the presence of highly immunogenic virulence associated proteins (Vap A, C, D, E, G, H), of which Vap A is apparently the most important, although the role of the other Vap proteins has not been determined.9,10 Vap A is a surface-expressed, lipid-modified protein that elicits an intense humoral response by foals.10 Expression of Vap A, C, D, and E is upregulated by incubation at 37°C, consistent with their role as virulence factors. Other genes probably involved in virulence are also upregulated by conditions that mimic those in vivo.11 The presence of the virulence proteins is associated with enhanced ability of virulent R. equi to survive and replicate within macrophages, whereas avirulent strains replicate poorly or not at all.12 However, the exact virulence mechanisms of R. equi are not known.13
The genes for Vaps A and C–H are present in a pathogenicity island in an 80–85 kb plasmid that is present in 98% of isolates of R. equi from foals with pneumonia.14 Most isolates from the environment, feces, pigs, cattle, and human patients with R. equi infection do not contain either of the two identified virulence plasmids.4,15,16 Virulence is associated with the presence of the plasmid, and loss of the plasmid by a strain of R. equi results in loss of virulence. There is geographical variation both within and between countries in the prevalence of each of the virulence plasmids in isolates from foals with pneumonia.15,17,18
EPIDEMIOLOGY
Occurrence
R. equi pneumonia in foals has a worldwide distribution. Clinical disease is often sporadic but on farms where the disease is endemic annual morbidity can be as high and can vary widely from year to year.19 The median percentage of foals that developed R. equi at farms on which the disease was endemic was 6.6%, with 38% of farms having more than 10% of foals affected.20 Case fatality rates for foals on farms, as opposed to those treated at veterinary teaching hospitals, is reported as 29–42% (for 113 and 19 affected foals, respectively).20,21 The median case fatality rate for 32 farms in Texas was 25% and the case fatality rate was more than 50% for 22% of farms. The case fatality rate among foals treated at veterinary teaching hospitals is approximately 28%.22 The detection of R. equi pneumonia in one foal on a farm should prompt an examination of all other foals on that farm.
Current evidence supports the hypothesis that foals are exposed and infected within the first several days of life.23 The age at onset of clinical signs of disease associated with R. equi varies between 2 weeks and 6 months but the peak prevalence for pneumonic disease is between 1 and 3 months.24 The disease is rare in adult horses. Risk factors in foals for development of R. equi pneumonia have not been determined although a large number of factors have been examined.25 The month in which the foal was born, gestational age, dam’s parity, antimicrobial administration during the first week of life, exposure to pasture at less than 2 weeks of age, need for treatment to correct inadequate transfer of passive immunity, and size of mare/foal groups were not associated with risk of disease on farms in Texas.26
The prevalence of virulent R. equi in isolates from the environment does not appear to be greater on farms where the disease is endemic.18,27 Morbidity varies widely among geographical areas and individual farms, probably because of environmental factors that affect the number of virulent R. equi and the ease of infection. Because aerosol infection by virulent R. equi in dust is thought to be the most important route of infection of foals, factors that favor the accumulation and persistence of R. equi in soil and its ability to become aerosolized most probably increase the risk of infection. Such factors might include:24
• Hot and dry weather, favoring formation of dust
• Crowding of pastures with young horses
However, empirical demonstration of the importance of these risk factors has not been reported, with several exceptions. Soil pH, salinity and concentrations of various elements including iron, zinc, and copper are not associated with the risk of foals developing R. equi pneumonia on farms in Texas.28 These soil-associated risk factors were examined because R. equi is a normal inhabitant of the soil and of the intestine of ruminants, horses, and pigs. It is not highly resistant but it has been found to survive in moist soil for periods of longer than 12 months. The infection is considered to be soil associated and to be maintained through a soil–horse cycle.29 The number of organisms in the soil and stable areas on horse farms increases with the time that the farms have housed horses, although there is not a strong correlation between R. equi concentration in soil and prevalence of pneumonia in foals.18,30
Farms of larger size, with more resident mares, greater numbers of foals (≥15), and greater foal density per acre, and the presence of mares brought on to the farm for breeding, are all associated with greater risk of foals developing R. equi pneumonia.20,31 R. equi pneumonia does not appear to be associated with poor farm management or lack of preventative health practices such as vaccination, deworming, or administration of hyperimmune plasma.25 The practice of testing for failure of transfer of passive immunity is associated with an increased likelihood of the disease on a farm.32 However, this association probably reflects the facts that the disease is more likely on larger farms, which are more likely to perform this test, and that farms that have had the disease are more likely to institute preventive care procedures.
Transmission
Most foals are exposed to infection, as demonstrated by seroconversion, but only a few develop disease. The organism colonizes the intestine of the normal foal during the first 2 months of life and has been detected in the feces as early as 5 days.33 Inhalation of the organism in dust is probably the most important route of transmission for pneumonic disease.34,35 Intestinal disease, which may be clinically inapparent, usually occurs with pulmonary disease but the source of the infection is unclear, although it may be ingestion of contaminated material or swallowing of infected respiratory secretions. Foals over 5 weeks of age have generally been resistant to experimental challenge.
PATHOGENESIS
Exposure of foals to R. equi is common, based on rate of seroconversion, yet the development of respiratory infection and clinical disease is much less common. The reason for this is not fully understood, although development of the disease probably depends on exposure to an infectious dose of organism and the susceptibility of the foal. Recent epidemiology studies suggest that exposure occurs within the first few days of life, before waning of maternally derived passive immunity, and that affected foals have lower CD4+:CD8+ ratios before development of the clinically apparent disease than foals that do not subsequently develop the disease.36 The differences between groups of foals was largest during the first 2 weeks of life, suggesting that foals that subsequently develop disease associated with R. equi may have impaired immune function during early life.36 In adult horses, in which the disease is rare, protective immunity is associated with both cellular and humoral immune responses characterized by enhanced immunoproliferative responses of CD4+ and CD8+ cells and presence of IgGa and IgGb antibodies to Vap A.37 Opsonizing antibody to R. equi is an important defense mechanism in experimentally infected foals and administration of R. equi hyperimmune plasma or plasma rich in anti-Vap A and C antibodies protects experimentally infected foals from developing pneumonia.38 Overall, these results suggest that foals that develop R. equi pneumonia have a T helper cell (Th)2-like response to infection, rather than a Th1-like response. Th1-like responses, which are associated with enhanced CD4+ and CD8+ responses, are believed to be important in resistance to the disease.13 Whether the switch to a Th2-like response to infection is a function of virulent R. equi or an attribute of susceptible foals has not been determined.
Experimental and clinical studies indicate that the foal is infected several weeks or months before clinical signs are observed. Virulent strains of R. equi are facultative intracellular parasites of macrophages, which they ultimately destroy. Neutrophils are bactericidal for R. equi but the organism can survive by inclusion in macrophages. Opsonization of R. equi by specific antibodies results in enhanced lysosome–phagosome fusion and greater killing of R. equi by equine macrophages and monocytes,13 whereas entry of R. equi into macrophages by nonimmune phagocytosis is not associated with enhanced killing. Its survival in the macrophage is associated with absence of phagosome– lysosome fusion.39 Nonvirulent strains do not proliferate in macrophages and monocytes. The combined action of humoral and cellular immune systems is important in preventing development of the disease after inhalation of bacteria. Without opsonization, the capacity of the pulmonary macrophage of foals to kill R. equi is impaired and the organism has been shown to be able to persist in the pulmonary macrophage of infected foals. The inability of the pulmonary macrophages to destroy R. equi leads to persistent infection in the lung and a chronic bronchopneumonia with extensive abscessation and an associated suppurative lymphadenitis.
Intestinal infection is common in foals with R. equi pneumonia,40 although clinical manifestations of the intestinal infection, such as diarrhea, are uncommon. Gastrointestinal tract infection is characterized by ulcerative lesions of the mucosa of the large intestine and cecum. In rare cases bacteremia and subsequent suppurative foci may develop in many organs, including bones and joints, liver, kidneys, and subcutis.
CLINICAL FINDINGS
R. equi pneumonia of foals may present as an acute onset of inappetence, fever, depression and tachypnea or as a more chronic disease characterized by cough and failure to thrive. The former presentation is more common and usually occurs in foals less than 3–4 months of age, with younger foals being more severely affected. It is important to realize that the acute onset of the disease is preceded by a long incubation period during which clinical signs are minimal and that the development of clinical signs is associated with severe and extensive lung lesions. The foal is often in respiratory distress and is reluctant to move and to suckle. Cyanosis may be present in severe cases. Auscultation of the chest may reveal crackles and wheezes, but abnormal lung sounds are often much less apparent than the severity of the respiratory disease suggests they should be. Foals with R. equi abscesses may not have abnormal lung sounds. There is usually minimal nasal discharge.
Subcutaneous abscesses, osteomyelitis and septic arthritis may be present or develop.41 Many foals (20%) have an aseptic polyarthritis, evident as nonpainful distension of joints, at the time they develop signs of respiratory disease. Immune-mediated uveitis occurs in approximately 10% of severely affected foals.42
In older foals the disease assumes a characteristic clinical syndrome marked by development of severe lesions without clinical signs in the foal. A subacute pneumonia develops slowly with coughing, an increase in the depth of respiration with respiratory distress developing in the late stages, and characteristic loud, moist crackles or ‘rattles’ on auscultation. The foal continues to suck and the temperature is sometimes normal but the foal becomes emaciated. Severe diarrhea may follow or accompany the respiratory signs. Nasal discharge and lymph node enlargement in the throat regions are absent. Severely affected animals die in 1–2 weeks. Thoracic auscultation may not reveal any abnormalities during the preclinical stage of the disease, although careful auscultation when tidal volume of the foal is increased by exertion or use of a rebreathing bag can reveal localized wheezes and crackles or changes in the intensity of normal breath sounds (indicative of areas of lung consolidation).
Radiographic examination is a valuable aid in diagnosis and in monitoring progress in hospitalized foals.43 Affected animals show evidence of consolidation of lung tissue, lymphadenopathy, and cavitating lesions in the lungs.
Ultrasonographic examination of the chest may reveal the presence of pulmonary consolidation before clinical signs are apparent and is useful means of detecting subclinical disease.
When lesions are confined to the intestinal wall the predominant clinical sign will be diarrhea, which may be acute or chronic and intermittent.
Intra-abdominal abscesses are associated with ill-thrift, weight loss, variable abdominal distension, fever, depression and in some cases colic. Ultrasonographic examination can reveal the abscess provided that it is within the field of the ultrasound probe.
PROGNOSIS
R. equi infection in Thoroughbred and Standardbred foals is associated with a reduced chance of racing as an adult compared with the overall population of foals, but affected foals that survive have a similar racing performance as adults to horses that did not have R. equi pneumonia.22,44 The morbidity and case fatality rates are provide above under Epidemiology.
CLINICAL PATHOLOGY
Hematological evaluation usually reveals leukocytosis with neutrophilia and monocytosis, and elevation in the concentrations of acute phase proteins including plasma fibrinogen and serum amyloid A – changes characteristic, but not diagnostic, of R. equi infection.45 Monitoring of blood white cell concentration and plasma fibrinogen concentration are useful in foals from farms on which the disease is endemic. White blood cell concentrations above 13.0 × 109/L (13000 cells/μL) have a sensitivity and specificity of 95% and 61% respectively for R. equi pneumonia.46 The high sensitivity means that few foals with the disease will be missed, while the moderate specificity means that a number of foals will be incorrectly suspected as having the disease. Because a high white cell count can be caused by a number of diseases other than R. equi pneumonia, foals with high white cell counts from farms on which the disease is endemic should be further examined for evidence of disease, including detailed clinical examination possibly including ultrasonographic examination, culture or PCR of tracheal aspirates, or thoracic radiography. Measurement of plasma fibrinogen concentration is less useful for detecting foals with R. equi pneumonia. Plasma fibrinogen concentrations of 400 mg/dL (0.4 g/L) have sensitivity and specificity of 91% and 51%, respectively, whereas concentrations of 600 mg/dL (0.6 g/L) have sensitivity and specificity of 38% and 96%, respectively.46 The positive and negative predictive values of the tests depends on the prevalence of the disease among the group of foals examined, being low for farms on which the disease is rare and increasing as the prevalence of the disease increases. Serial measurement of serum amyloid A concentrations is not useful for detecting foals with clinically inapparent R. equi pneumonia, nor do foals with pneumonia reliably have higher serum amyloid A concentrations than normal foals.47
Transtracheal aspirates from affected foals reveal a neutrophilic leukocytosis. Intracellular, Gram-positive pleomorphic rods characteristic of R. equi may be present in tracheal aspirates but the sensitivity of this observation has not been determined and all tracheal aspirates should be cultured.
Although numerous serological tests have been developed, including agar gel immunodiffusion, synergistic hemolysis inhibition, radial immunodiffusion and various ELISAs, none has demonstrated value in the diagnosis of the disease in individual animals. Currently available serological tests, either as single or paired samples, are not reliable in confirming or excluding the presence of R. equi pneumonia in foals.28
Culture of tracheal aspirates is the gold standard for antemortem diagnosis of the disease, although sensitivity of culture is less than that of PCR examination of tracheal aspirates. Culture of tracheal aspirates has a sensitivity of approximately 86%, based on diagnosis of R. equi pneumonia at necropsy.46 However, R. equi can be cultured from 35% of clinical normal foals in populations in which the disease is endemic.48 A PCR test for the rapid detection of R. equi in tracheal aspirates has a sensitivity of 100% and a specificity of 91% in foals with a clinical diagnosis of R. equi pneumonia.49 PCR examination of nasal swabs for presence of R. equi has a sensitivity of 15%, which is too low to be clinically useful.50 More recent quantitative real-time PCR assays permit the rapid detection and quantification of virulent (VapA-gene-positive) strains of R. equi in tracheobronchial aspirates.51 This assay detects R. equi at concentrations as low as 20cfu/mL of tracheobronchial fluid, providing a specific and highly sensitive test for the presence of this organism. A multiplex PCR test simultaneously detects R. equi and the presence of virulence factors, thereby permitting rapid differentiation of pathogenic from nonpathogenic strains of R. equi in biological samples.52
NECROPSY FINDINGS
The predominant lesions are a pyogranulomatous pneumonia plus lymphadenitis of the bronchial lymph nodes.12 Grossly, the firm, raised lung nodules may reach several centimeters in diameter and be located anywhere in the lung field, especially in the cranioventral quadrant. If several nodules coalesce, the lesion may be misinterpreted as a suppurative bronchopneumonia. Histologically, organisms are easily demonstrated within the macrophages and giant cells comprising these lesions. Many cases also have ulcerative enterocolitis, with abscessation of mesenteric or cecocolic lymph nodes. Although necropsy may reveal widespread infection, many cases are subclinical.
DIAGNOSTIC CONFIRMATION
Antemortem diagnosis is by culture of R. equi from aspirates of tracheal fluid. Currently available serological tests do not provide confirmation of disease in individual animals.
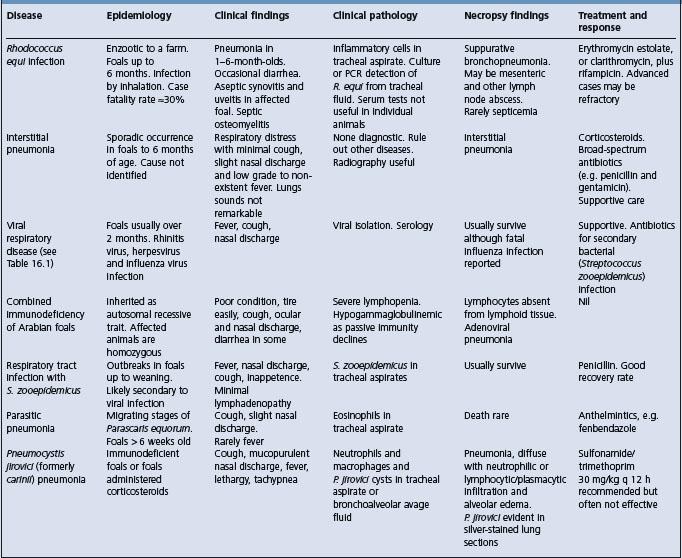
The pneumonic form of the disease may be confused with other causes of pneumonia in foals (Table 16.4). Other causes of diarrhea in this age group include parasitism due to cyathostomes, infection by Salmonella sp. and antibiotic-induced diarrhea.
The aseptic synovitis and joint effusion that frequently accompanies R. equi pneumonia should be differentiated from septic arthritis due to S. zooepidemicus, Salmonella spp., R. equi or other bacteria.
TREATMENT
The principles of treatment are cure of R. equi infection, relief of respiratory distress and correction of associated immune-mediated diseases.
Elimination of infection requires the administration of antimicrobial agents that are both effective against the organism and able to penetrate infected macrophages to gain access to the organism. In vitro antibiotic sensitivity testing has not been demonstrated to be useful in predicting the clinical efficacy of treatment. R. equi isolates from ill foals are frequently sensitive in vitro to a variety of antibiotics, including the aminoglycosides gentamicin and neomycin, tetracycline, sulfonamides and chloramphenicol, while most are resistant to cephalosporins and penicillin.53-55 However, treatment with antibiotics other than erythromycin and rifampin is associated with a lower recovery rate. Treatment with penicillin, with or without gentamicin, chloramphenicol or tetracycline, is not effective.53 Trimethoprim–sulfadiazine combinations might be effective in some foals but are not the preferred treatment.53 Neomycin has been recommended for treatment of R. equi pneumonia56 but the risk of nephrotoxicosis, need for parenteral administration and lack of demonstration of clinical efficacy do not support its use at this time.
The treatment of R. equi pneumonia in foals is achieved by administration of macrolide antibiotics in combination with rifampin. Conventional treatment is administration of the combination of an acid-stable erythromycin (preferably estolate) at a dose of 25 mg/kg orally every 12 hours and rifampin at a dose of either 5 mg/kg every 12 hours or 10 mg/kg every 24 hours. Other esters or preparations of erythromycin are less well absorbed or have shorter elimination half-lives than the estolate ester and must be administered more frequently.32 Erythromycin ethylsuccinate does not provide optimal therapy for R. equi pneumonia in foals because of poor absorption after oral administration.55,57 The macrolide antibiotics azithromycin and clarithromycin have also been used to treat foals with R. equi pneumonia. Treatment of foals with a combination of clarithromycin (7.5 mg/kg orally every 12 h) and rifampin results in improved survival over foals treated with azithromycin (10 mg/kg orally every 24 h) and rifampin or erythromycin and rifampin in a veterinary teaching hospital.58
Therapy should be continued until the foal is clinically normal and has a normal plasma fibrinogen concentration and white blood cell count, which can require treatment for at least 1 month and often longer. Radiographic or ultrasonographic demonstration of resolution of the pulmonary consolidation and abscessation is useful in the decision to stop therapy. The case fatality rate is approximately 30% (see Epidemiology, above) even with appropriate treatment.
Side-effects of erythromycin–rifampin therapy include the development of diarrhea in some foals and their dams.59 Administration of erythromycin to foals is associated with an eightfold increase in the risk of diarrhea.50 Antibiotic therapy should be temporarily discontinued in foals that develop diarrhea.
During hot weather, some foals treated with erythromycin become hyperthermic (40–41°C, 104–105.5°F) and tachypneic and occasional deaths result from this syndrome.50,60 The basis for this hyperthermic event, which may occur in healthy foals administered erythromycin, is unknown. Affected foals should be treated urgently with antipyretics, cold water bathing and housing in a cooler environment.
The emergence of R. equi isolates resistant to rifampin during therapy of foals with R. equi pneumonia has been reported.61 However, one case had an atypical clinical course, in that the foal was 10 months of age at presentation and may have been immunosuppressed, and in the other instance the foal was treated with only rifampin. The development of resistance during monotherapy with rifampin is a recognized contraindication to the use of this drug alone.
Ancillary therapy with NSAIDs, bronchodilators, and mucolytics might be of value.53 Foals in severe respiratory distress require intranasal or intratracheal administration of oxygen.
CONTROL
Control measures are designed to maximize the resistance of the foal to infection and to reduce the infection pressure on the foal by decreasing contamination of the foal’s environment with virulent R. equi. Insuring adequate transfer of colostral immunoglobulins in all foals through routine monitoring of serum immunoglobulin concentrations in 1-day-old foals is an essential part of any control program. To decrease environmental contamination with virulent R. equi, efforts should be made to reduce fecal contamination of pastures and to reduce or eliminate dusty or sandy areas.24,62 These efforts should include grassing or paving of bare areas, removal and composting of fecal material on a regular basis, reduction of stocking density and reduction in the size of mare/foal bands.24
On farms with endemic disease, regular physical examination, including auscultation of foals and once-daily monitoring of rectal temperature, can permit early identification and treatment of affected foals.63 Measurement of blood white cell count, as detailed above, can be useful in early identification of affected foals. Identification of one foal affected with R. equi pneumonia on a farm should prompt an examination of all other foals on the farm.
Ultrasonographic examination of the thorax of foals may permit identification of foals with clinically inapparent pulmonary abscesses.
The administration to foals of a hyperimmune serum, obtained from mares vaccinated with an autogenous vaccine, limits the severity of disease produced by experimental challenge but has not been consistently useful in preventing or decreasing the prevalence of naturally occurring disease.19,34,64,65 There are no vaccines effective in prevention of R. equi pneumonia in foals.
Giguere S, Prescott JF. Clinical manifestations, diagnosis, treatment, and prevention of Rhodococcus equi infections in foals. Vet Microbiol. 1997;56:313-334.
Takai S. Epidemiology of Rhodococcus equi infections: a review. Vet Microbiol. 1997;56:167-176.
Cohen ND, et al. Control and prevention of Rhodococcus equi pneumonia in foals. Compend Contin Educ Pract Vet. 2000;22:1062-1069.
Meijer WG, Prescott JF. Rhodococcus equi. Vet Res. 2004;35:383-396.
1 Davis WP, et al. Vet Pathol. 1999;36:336.
2 Verdonck F, et al. Vet Rec. 2004;154:149.
3 Vengust M, et al. Can Vet J. 2002;43:706.
4 Flynn O, et al. Vet Microbiol. 2001;78:221.
5 Cornish N, Washington JA. Curr Clin Top Infect Dis. 1999;19:198.
6 Bowles PM, et al. Vet Microbiol. 1987;14:259.
7 Cohen ND, et al. Am J Vet Res. 2003;64:153.
8 Morton AC, et al. Appl Environ Microbiol. 2001;67:2167.
9 Takai S, et al. Infect Immunol. 1992;60:2995.
10 Tan C, et al. Can J Vet Res. 1995;59:51.
11 Ren J, Prescott JF. Vet Microbiol. 2003;94:167.
12 Hondalus MK, et al. Infect Immun. 1994;62:4167.
13 Meijer WG, Prescott JF. Vet Res. 2004;35:383.
14 Haites RE, et al. J Clin Microbiol. 1997;35:1642.
15 Nicholson VM, et al. J Clin Microbiol. 1997;35:738.
16 Makrai L, et al. Vet Microbiol. 2002;88:377.
17 Yuyama T, et al. J Vet Med Sci. 2002;64:715.
18 Takai S, et al. J Vet Diagn Invest. 2001;13:489.
19 Hurley JR, et al. Aust Vet J. 1995;72:418.
20 Chaffin MK, et al. J Am Vet Med Assoc. 2003;222:467.
21 Raidal SL. Aust Vet J. 1996;73:201.
22 Ainsworth DM, et al. J Am Vet Med Assoc. 1998;213:510.
23 Horowitz ML, et al. J Vet Intern Med. 2001;15:171.
24 Prescott JF, Hoffman AM. Vet Clin North Am Equine Pract. 1993;9:375.
25 Chaffin MK, et al. J Am Vet Med Assoc. 2003;222:476.
26 Chaffin MK, et al. J Am Vet Med Assoc. 2003;223:1791.
27 Martens RJ, et al. J Am Vet Med Assoc. 2000;217:220.
28 Martens RJ, et al. J Am Vet Med Assoc. 2002;221:825.
29 Takai S, et al. Vet Microbiol. 1986;12:169.
30 Prescott JF. Vet Microbiol. 1987;14:211.
31 Cohen ND, et al. J Am Vet Med Assoc. 2005;226:404.
32 Lakritz J, et al. Compend Contin Educ Pract Vet. March: 2002:256.
33 Takai S. Vet Microbiol. 1997;52:63.
34 Martens RJ, et al. Proc Am Assoc Equine Pract. 1990;35:199.
35 Martens RJ, et al. Equine Vet J. 1982;14:111.
36 Chaffin MK, et al. Vet Immunol Immunopathol. 2004;100:33.
37 Hines MT, et al. Vet Immunol Immunopathol. 2001;79:101.
38 Hooper-McGrevy KE, et al. Am J Vet Res. 2001;62:1307.
39 Brumbaugh GW, et al. Am J Vet Res. 1990;51:766.
40 Zinc MC, et al. Can Vet J. 1986;27:213.
41 Giguere S, et al. Aust Vet J. 1994;26:74.
42 Chaffin MK, Martens RJ. Proc Am Assoc Equine Pract. 1997;43:79.
43 Prescott JF, et al. Equine Vet J. 1996;28:344.
44 Ainsworth DM, et al. Am J Vet Res. 1993;54:2115.
45 Hulten C, Demmers S. Equine Vet J. 2002;34:693.
46 Giguere S, et al. J Am Vet Med Assoc. 2003;222:775.
47 Cohen ND, et al. Equine Vet J. 2005;37:212.
48 Ardans AA, et al. Proc Am Assoc Equine Pract. 1986;32:129.
49 Sellon DC, et al. J Clin Microbiol. 2001;39:1289.
50 Stratton-Phelps M, et al. J Am Vet Med Assoc. 2000;217:68.
51 Harrington JR, et al. Am J Vet Res. 2005;66:755.
52 Halbert ND, et al. Am J Vet Res. 2005;66:1380.
53 Sweeney CR, et al. Vet Microbiol. 1987;14:329.
54 Barton MD, Hughes KL. Vet Bull. 1980;50:65.
55 Jacks SS, et al. Antimicrob Agents Chemother. 2003;47:1742.
56 Barton MD. Aust Vet J. 1986;63:163.
57 Lakritz J, et al. Vet Ther. 2002;2:189.
58 Giguere S, et al. J Vet Intern Med. 2004;18:568.
59 Gustafsson A, et al. Equine Vet J. 1997;29:314.
60 Traub-Dargatz J, et al. Proc Am Assoc Equine Pract. 1996;42:243.
61 Fines M, et al. J Clin Microbiol. 2001;39:2784.
62 Giguere S, Prescott JF. Proc Am Assoc Equine Pract. 1997;43:65.
63 Higuchi T, et al. J Am Vet Med Assoc. 1998;212:976.