BOVINE VIRUS DIARRHEA, MUCOSAL DISEASE. BOVINE PESTIVIRUS DISEASE COMPLEX
Etiology
Bovine virus diarrhea virus. Type 1 and type 2 genotypes and subtypes. Noncytopathic and cytopathic biotypes. Antigenic diversity and cross-reactivity among strains of virus
Epidemiology
Occurs worldwide and major economic importance. Prevalence of infection high in cattle population. Persistently-infected (PI) calves are major source of virus. Young and unvaccinated cattle in herd most susceptible
Pathogenesis
Virus causes subacute infections, peracute infections and thrombocytopenia and hemorrhagic syndrome, immunosuppression, fetal infections which persist in the fetus until and after birth in persistently-infected cattle which are also immunotolerant and may develop mucosal disease
Signs
Inapparent subclinical bovine virus diarrhea acute mucosal disease, in persistently infected cattle 6–24 months of age with fever, diarrhea, oral erosions and high case–fatality rate, peracute bovine virus diarrhea in cattle of all ages including adults with severe diarrhea, fever, agalactia and rapid death in few days, thrombocytopenia and hemorrhagic disease in veal calves; reproductive failure (decreased conception rate, abortion, stillbirth, weak neonates, congenital defects)
Clinical Pathology
Leukopenia in acute mucosal disease. Virus isolation from persistently infected animals and from cattle with acute viremia, serology for serum neutralizing antibodies
Lesions
Erosive stomatitis and gastroenteritis, depletion of Peyer’s patches. Widespread hemorrhages in peracute form. Abortions. Congenital defects of calves (cerebellar hypoplasia, ocular defects)
Diagnostic confirmation
Virus isolation from blood and tissues. Antigen detection (antigen capture ELISAs and immunohistochemical tests). Polymerase chain reaction amplification of RNA. Viral neutralization serum antibody and ELISA tests
Differential diagnosis list
Diseases with oral erosions and diarrhea (rinderpest, bovine malignant catarrh). Diseases with oral lesions and no diarrhea (foot-and-mouth disease, vesicular stomatitis, bluetongue, bovine papular stomatitis, necrotic stomatitis), disease with diarrhea and no oral lesions (salmonellosis, winter dysentery, Johne’s disease, copper deficiency, ostertagiasis, coccidiosis, arsenic poisoning, carbohydrate engorgement)
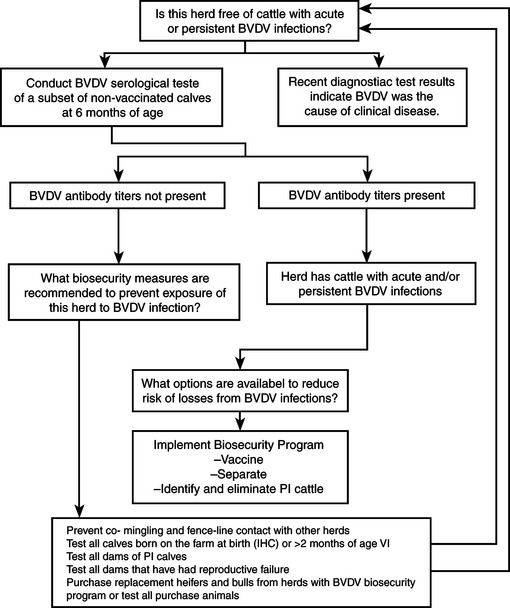
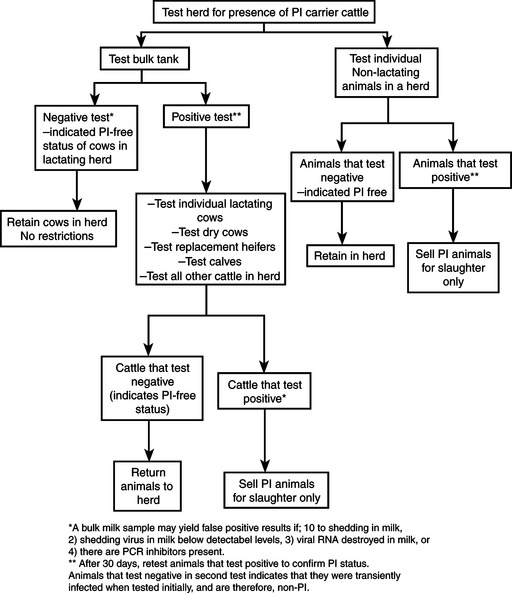
Control
Detection and elimination of PI animals from the herd. Prevention of introduction of infection into herd. Vaccination of breeding females to prevent fetal infection. Eradication by detection and elimination of persistently-infected animals, no vaccination and strict biosecurity measures to prevent introduction of PI animals into the herd
ETIOLOGY
The bovine virus diarrhea virus (BVDV) is one of three pestiviruses:
1. Bovine virus diarrhea virus (also known as mucosal disease virus or bovine pestivirus)
2. Border disease virus of sheep
3. Hog cholera virus (also called European or classical swine fever virus).
The viruses are classified in the virus family Flaviviridae and are members of the genus Pestivirus.1 Cross-infection between species can be achieved experimentally and has been demonstrated in field infections.1 The molecular biology of the BVDV has been reviewed.2
Pestiviruses are nonsegmented, single, sense stranded (positive polarity (+)) RNA viruses. The genomic structure has been described.1 Phenotypic diversity, such as antigenic variation, infectivity and replication rates, which can affect viral virulence, can be attributed to genomic reassortments, mutations or recombinations.1
Among the ruminant pestiviruses, particularly BVDV, there are two biotypes designated as non-cytopathic (NCP) and cytopathic (CP) depending on their effect on tissue culture cells. The non-cytopathic type is the most common and most important. Only the non-cytopathic type crosses the placenta, invades the fetus and establishes persistent infection in the fetus, which is crucial for spread of the virus. It is the cause of a wide range of congenital, enteric and reproductive diseases. In contrast, the cytopathic biotype of the virus is usually associated with only mucosal disease in animals already persistently infected with the non-cytopathic biotype. Both biotypes can be isolated from animals dying of mucosal disease and there is evidence that the cytopathic biotype evolves by mutation from the non-cytopathic biotype within PI animals. There is considerable antigenic diversity and antigenic cross-reactivity among isolates of BVDV which has implications for diagnostic testing and for control by vaccination.2 Antigenic and genetic differences have divided the BVDV into type I and type II genotypes. Each genotype has been subdivided into subgenotypes. BVDV-1 isolates are grouped by phylogenetic analysis into at least 11 genetic groups.3
Two subgenotypes of BVDV-1 are designated 1a and 1b. BVDV 1a strains are considered to be American in origin, whereas BVDV 1b is considered to have originated in Europe. However, genetic typing of bovine pestiviruses from both Northern Ireland and the Republic of Ireland were found to be BVDV 1a.4 Most strains in England and Wales are BVDV 1b. It is suggested that the widespread importation of Holstein cattle from North America may have contributed to the predominance of BVDV 1a in the British Isles. Import restrictions which limited importation of livestock from continental Europe may explain the prevention of introduction of BVDV 1b strains to Great Britain and Ireland which will change with increased levels of animal movement following the introduction of the Single European Market.
Genomic recombination can occur in noncytopathic viruses from either genotype giving resulting in cytopathic viruses.5 Only noncytopathic BVDVs cause severe acute bovine virus diarrhea. Genetic drift results in genotype; genomic recombination is associated with changes in phenotype (biotype). Variation in genotype is more significant to detection and control than variation in biotype.5
BVDV-2 genotypes are antigenically distinct and some isolates cause severe disease outbreaks.6 Not all BVDV-2 isolates cause clinically severe disease; avirulent strains do exist. Virulent BVDV-2 strains inoculated into calves produce disease characterized by fever, diarrhea, leucopenia, lymphopenia, neutropenia, thrombocytopenia, and death. Infection with avirulent BVDV-2 strains causes leucopenia and low-grade fever.6
EPIDEMIOLOGY
Occurrence
Diseases associated with the BVDV have been recorded in most countries where cattle are raised and in some countries may be the single most important virus infection of cattle. The prevalence of infection is high but the incidence of clinical mucosal disease is low.
The BVDV causes several different diseases including:
• Benign bovine virus diarrhea, which is usually subclinical
• Fatal mucosal disease which occurs in persistently viremic animals and those specifically immunotolerant as a result of an infection acquired in early fetal life
• Peracute, highly fatal diarrhea
• Thrombocytopenia and hemorrhagic disease
• Congenital abnormalities in calves as a result of fetal infection in mid-gestation.
The virus may also be immunosuppressive.
Mucosal disease was first recognized in 1946 and for the next 35 years it was assumed that the disease was the result of an infection prior to the onset of illness. It is now clear mucosal disease occurs only in PI animals as a result of a congenital infection with a non-cytopathic strain of the virus acquired in early fetal life. These animals remain specifically immunotolerant to the homologous strain of the BVDV throughout postnatal life, and fatal mucosal disease is precipitated by a superinfection with a cytopathic strain of the virus occurring usually at 6–24 months of age or older.
In the late 1980s and early 1990s, a peracute form of the enteric form of the disease and thrombocytopenia in young and adult immunocompetent animals infected with highly virulent strains of the virus were recognized. This was the first indication that mucosal disease could occur in immunocompetent animals as a result of postnatal infection.
Prevalence of infection
Of cattle over 1 year of age, 60–80% have serum neutralizing antibodies to the virus.7 Vaccination programs and the existence of persistently viremic animals which excrete the virus are responsible for the serologically positive animals in a herd.7 In a survey of 256 beef breeding herds in the United States, over 90% of herds and more than 68% of cattle have been exposed to the BVDV either through vaccination or natural exposure.8 Based on serum neutralization assays of type 1 and type 2 BVDV in a diagnostic laboratory in the Upper Midwest United States over a 7-year period there has been a progressive increase in the number of cases with high SN titers.9 The increase is due in part to more extensive use of vaccination but probably also related to a rise in field infections. However, the lack of standardization of the serum neutralization testing between laboratories may affect the interpretation of such surveys.
Young cattle which are persistently infected with a non-cytopathic strain of the virus are the major source of infection in a herd. Conversely, the absence of PI animals in a herd could result in a serologically negative herd. Seroepidemiological surveys of feedlot cattle also reveal that animals seroconvert to the BVDV during the first several weeks following the arrival in the feedlot due to presence of PI animals.
The mean prevalence of PI animals in herds is about 1–2%. A survey of all cattle in 19 Danish dairy herds with unknown status of BVDV infection revealed that 1.4% of the animals were PI. The prevalence of seropositive animals in herds with one or more PI animals was 87%; in herds without PI animals the prevalence was 43%. The prevalence is also much higher in animals under 1 year of age compared to the older animals in the herd. The theoretical risk of fetal infections occurring during the first 3 months of pregnancy was estimated to be 3.3%.10 The age distribution of PI animals may be clustered into two separate groups based on the introduction of infected animals followed by the birth of infected animals.10 In 66 selected herds in the United States, the mean frequency of persistent infection was 1.9% from six herds and nil from 60 herds.11 However, in two of the six herds the prevalence of PI animals was 27% and 18%, and in one herd all PI animals died of mucosal disease within 5 months of the initial sampling.11 Of randomly selected beef herds in the United States, about 3% had calves with persistent BVDV infection, and 19% of herds suspected by the herd veterinarian to be a BVDV infected herd had calves with persistent infection.12 Most persistently infected calves survived to weaning and could provide a constant source of virus throughout the breeding season and early gestation.
Serological surveys in Norway revealed a prevalence rate of 18.5% in cattle, 4.5% in sheep and 2.2% in pigs.11 In all three species, the prevalence rate varied considerably according to herd and geographical location.
The prevalence of PI cattle may vary between countries due to vaccination practices, the population densities of cattle and housing compared to pastured animals, the selling of male calves after birth, and housing young animals on different premises.11
The prevalence of BVDV infection in a population of feedlot calves in western Canada was 27% according to the ELISA test and it varied from 0–63%; according to the virus neutralization test the seroconversion risk was 40% and it varied from 0–100%.13 In the same survey, the prevalence of PI calves was <0.1%, which is unusually low suggesting that few PI calves were purchased. The prevalence of acute viremia in the calves treated in the feedlot hospital was low at 4%, thus the prevalence of persistent infection was low but serological tests suggested a high risk of seroconversion to BVDV.
The prevalence of PI calves born in a beef herd in 1 year ranged from 9.1–12.7%.14 When isolation of the calves on a commercial feedlot to mimic normal management conditions in western Canada, was compared to BVDV-negative herdmates, persistently infected calves were ‘poor doers’ and had poor survivability to 1 year of age.
A high rate of BVDV infections in replacement dairy heifers between weaning and 9 months of age has been observed.15 The risk of BVDV infection increased with age from 150 to 260 days of age and coincided with removal from the relative isolation in hutches, diminished expected protection from colostral antibodies, and increased exposure to a large number of cattle, some of which possibly shed the virus.
Congenital infection (not persistent infection) with the BVDV in apparently healthy dairy calves, at a rate of about 10% has been reported.16 Calves with congenital infection had a 2-fold higher risk of a severe illness, compared to calves without congenital infection.
Over a 20-year period in the northwestern United States from 1980 to 2000, there was a shift in disease profiles associated with the BVDV infection and in the age of animal at onset of disease.17 In 1980, data indicated a low fetal infection rate (<5%), followed by steady increases of clinical cases and peaking at 6 months of age (30%). By 2000, the shift of BVDV cases was noticeable and indicated a biphasic occurrence of disease. The first phase was fetal infections, which increased to >25%, followed by a second phase at 6 months(>35%). The changing patterns may have been due to increased susceptibility of pregnant cattle to BVDV infection and the emergence of type 2 BVDV. The second phase at 6 months of age may be associated with increased susceptibility following decline of passive immunity. Over a 2-year period (1998–2000) type 2 BVDV isolates were most common and associated with abortion-open cows. BVDV type 1a was associated least with early infection (<100 days gestation) and most with late infections (>100 days); BVDV 1b was intermediate, followed by BVDV type 2 which was associated more with early infection (45%) and less with late infections (55%) when compared with BVDV 1a and BVDV 1b.
Pestiviruses also infect a wide range of other domestic animals, captive and free-living ruminants.18 There is serological evidence of BVDV infection in exotic ruminants, but outbreaks of disease are recorded only occasionally and usually as single fatal cases. The clinical and pathological findings in some animals are similar to those of mucosal disease in cattle. Serological surveys indicate that many species of free-living ruminants in North America, Europe and Africa have varying prevalence rates. BVD antibody has been found in some free-living red deer in various districts in Denmark probably as a result of infection from cattle19 The virus has been isolated from a free-ranging mule deer in Wyoming and 60% of mule deer in the same area had antibody to type 1a BVDV suggesting the virus circulates in the mule deer population.20 Experimental infection of New World Camelids, llamas, results in no clinical signs, fetal infection or persistent infection of crias.21
Pestiviruses have also been associated with outbreaks of disease among captive ruminants in zoological collections.18
Serological surveys of some populations of sheep and goats revealed that 11% of sheep and 16% of goats were seropositive.18 A pestivirus which cross-reacts with the BVDV causes border disease in lambs following in utero infection of pregnant ewes.18 Sheep can be infected naturally and transmission of the virus from cattle to sheep has been demonstrated. In Sweden, cattle infected with the BVDV are considered responsible for the majority of pestivirus infections in Swedish sheep.22 Experimental pestivirus infections in pregnant goats causes reproductive failure, abortions, stillbirths, and persistent infection in newborn kids with a disease similar to border disease in lambs.18 Experimental infection of newborn kids may result in minor lesions of the central nervous system but no clinical evidence of disease.18
Experimental ruminant pestivirus infection in pigs causes reproductive disease and growth retardation in piglets.18 Some strains of the BVDV inoculated into pigs cause false-positive reactions to tests for swine fever antibodies and may protect against subsequent challenge with swine fever virus.18 The importance of these and other species as a source of infection for cattle is unknown.
Morbidity and case–fatality rates
Mucosal disease in PI immunotolerant seronegative animals occurs in all classes of cattle mostly between 6 and 24 months of age, rarely in calves as young as 4 months of age, or cattle older than 2 years of age. The incidence of mucosal disease in a herd is usually less than 5% of the animals up to 2 years of age. Occasionally, epidemics have been observed in which up to 25% or more of the animals of the most commonly affected age group will develop mucosal disease.
Outbreaks of the recently recognized peracute BVD occurs in immunocompetent non-PI animals are characterized by a high case rate among all clinically affected animals. Morbidity rates may reach 40% and population mortality rates 20%. Herd outbreaks of acute disease associated with BVDV in veal calves caused population mortality rates ranging between herds from 10–25%.7
Methods of transmission
The major source of infection is the PI viremic animal. The virus can be isolated from nasal discharge, saliva, semen, feces, urine, tears and milk, each of which would allow wide dissemination of the virus.
Direct contact
The virus is transmitted by direct contact between animals, and by transplacental transmission to the fetus. Discharges from the reproductive tract of an infected cow, either PI or systemically immune, including aborted fetuses, can be potent sources of the virus. Nose-to-nose contact is an effective method of transmitting the virus from PI to susceptible animals.11,23 Thus PI animals may introduce the infection into a herd, or when infected animals are mixed with susceptible animals at the time of breeding or under conditions requiring emergency movement because of drought, flood or fire. The accidental mixing of a PI bull with susceptible breeding females during the breeding season in a beef herd may result in a major herd outbreak of mucosal disease. A pestivirus has been transmitted by contact from a PI bullock to pregnant sheep, resulting in the birth of PI lambs, one of which was able to transmit the virus by contact to pregnant cattle.24 Two of these animals gave birth to PI calves, one of which transmitted the virus again by contact with pregnant sheep, leading to another generation of PI lambs.
Transmission of the virus between healthy immunocompetent animals is probably insignificant because they produce antibodies and eliminate the virus. However, the spread of transmission from transiently viremic cattle to seronegative animals in a dairy herd was slow, requiring about 30 months to spread to about 83% of the susceptible group. Primary infected animals are not effective transmitters of the virus.23 Animals with primary infection even when co-infected with the bovine coronavirus do not transmit the virus to susceptible animals in close contact.25 Susceptible animals introduced into a herd, typically heifers, become infected by contact with persistently viremic animals and major economic losses can occur if they are at a vulnerable stage of pregnancy. The introduction of an unknown persistently infected cow or heifer into a susceptible herd can also cause major economic losses.11
The fetus can be infected by transplacental transmission of the virus from the infected dam, whether the dam is transiently or persistently infected.11 Fetal infection has been produced by inoculation of non-immune pregnant dams.7 Epidemics of abortion and congenital defects of calves have occurred when transplacental virus infection of the fetuses of cows in the first trimester, in previously virus-free herds, has followed the introduction of BVDV-infected animals.7
PI females can remain clinically normal for several years, during which time they may breed successfully and their progeny may also be apparently normal but are invariably also PI. In this way a maternal viremic family can be established which can persist for several generations and provides one of the major mechanisms for maintenance of the virus as endemic in the herd.7
PI bulls may also introduce the virus into artificial breeding units. A PI bull can shed the virus in his semen for a long period,26 and if introduced into a susceptible herd could have immediate undesirable effects on reproductive performance. However, PI bulls may have acceptable semen quality and fertility.26 Previously unexposed heifers have been shown not to conceive to service by a PI bull until they have seroconverted. The virus may be transmitted in cattle by artificial insemination with semen from a PI bull.26 The use of semen from a transiently infected bull has the potential to introduce pestivirus infection into a group or herd of susceptible animals but the conception rates are usually within normal ranges.26 However, once the infection is established in such a herd there is the potential for its amplification through a secondary cycle of transmission from heifers which were infected from the semen. The virus can persist in the semen of acutely infected bulls for several months after experimental exposure of immunocompetent, seronegative postpubertal animals.27 There is also a record of a post-pubertal bull in an artificial insemination unit which was shedding the virus in semen over a period of 11 months while not demonstrating any evidence of viremia but with a high level of serum antibodies.28 The virus could not be isolated from numerous blood samples and somatic organ tissues but at necropsy the virus was sequestered in the testes. It hypothesized that infection occurred shortly before the blood-testes barrier became fully functional thus allowing the virus to enter the seminiferous tubules but excluding the ensuing high levels of antibody from the site.
Indirect contact
Indirect airborne transmission of the virus can occur in calves housed near a PI calf for one week-at distances of 1.5 and 10 m – without having direct contact with a PI calf.23 Infection can also occur in calves housed in a pen directly after removal of a PI calf, but without the pen being cleaned and disinfected.
The virus has been experimentally transmitted by allowing blood feeding flies to feed on a PI animal followed by feeding on BVDV-free seronegative recipients. The virus was isolated from some of the flies, and from the recipient animals which also seroconverted.11
The BVDV has been transmitted from a PI animal to susceptible heifers which were examined per rectum using the same glove.29 Reusing a hypodermic needle on susceptible animals 3 min after the needle had been used on a PI animal or reusing a nose tong within 3 min after it had been used on a PI animal could also transmit the infection. The virus can be spread by hypodermic needles used on vaccine bottles contaminated by the nasal discharge of PI calves.23
Risk factors
Animal risk factors
In general, young cattle are most susceptible to BVDV infection but adult cattle may develop severe disease if infected with the highly virulent genotypes of the virus. Persistent infection can be established only in approximately the first half of fetal life. Compared to many other pathogens, fetal survival following early intrauterine infection with non-cytopathic BVDV is common and can be as high as 70%. Unvaccinated animals, improperly vaccinated animals, or animals whose immune status has waned are most susceptible to infection and the potential for clinical disease. Vaccinated animals may be susceptible if field isolates of the virus are distinct from those used in the vaccine. PI animals are susceptible to the development of mucosal disease following superinfection with the cytopathic virus. They are also susceptible to other infectious diseases such as pneumonia.
Immune mechanisms
The literature on the immune response to the BVDV has been reviewed.30 The interaction of the virus with both innate and adaptive immunity has been made much clearer.
Transient immunosuppression occurs in acutely infected animals. The virus infects cells pivotal in control of the innate and acquired immune response. The effects on the innate immune system have been examined in granulocytes, monocytes, and natural killer cells. The virus infects cells of the innate immune system affecting the function of neutrophils, monocytes, macrophages, and dendritic cells. Neutrophils are impaired in microbicidal, chemotactic, and antibody-dependent cell-mediated cytotoxicity.
In vitro or in vivo infection with BVDV, either cytopathic or noncytopathic biotype, depresses various aspects of macrophage function which can adversely affect normal defense mechanisms of the lung which can facilitate bacterial colonization.30 The effect of the different biotypes of the virus on interferon activity are variable.
In the acquired or adaptive immune response, BVDV infections have their major effect on thymic and follicular T-lymphocytes.30 The effect on the number of circulating T-lymphocytes is strain dependent and varies from a mild to severe lymphopenia with highly virulent strains. The virus also affects T-helper lymphocyte and cytotoxic T-lymphocyte responses. BVDV infections have their major effect on follicular B-lymphocytes. The effect on the number of circulating B-lymphocytes varies but depletion of B-cells occurs in the lymphoid follicles of the lymph nodes with highly virulent NCP BVDV and in Peyer’s patches with both mucosal disease and highly virulent BVDV infections.
The BVDV humoral antigens have been examined.30 There are four major structural antigenic polypeptides. Glycoprotein E2 is the major glycoprotein and antigenic target for antibodies. E2 is highly antigenic and elicits the production of neutralizing antibodies in the host after infection or vaccination with live or killed vaccines. The ability of BVDV antibodies to protect against BVDV infection and the development of long-term virus infection is dependent on the virus strain and the level and isotype of antibodies produced. BVDV antibodies are indicators of the presence of a particular immune response rather than an indicator of a protective immune response. High levels of neutralizing antibodies prevent disease following homologous challenge. However, animals with neutralizing antibodies may develop viremia. Shedding of the virus in nasal secretions may occur in the presence of serum neutralizing antibodies.
In vitro cross protection studies with serum from cattle vaccinated with either modified live virus or inactivated vaccine demonstrated wide crossneutralization against 12 to 22 different BVDV strains but not field studies do not show this extensive crossneutralization.
The immune response in the calf is influenced by two factors: the development of active immunity and the decay of maternal or passive immunity. Young calves at 10 to 14 days of age seronegative to BVDV can develop a protective immune response following vaccination with MLV vaccine.31,32 However, the presence of maternal antibody in calves interfered with the immune response and the animals were not protected from a challenge 4.5 months later. A predictive study estimated that calves must be 142 days of age to become seronegative for BVDV type 1 antibodies and 114 days for type2 antibodies.33
The immunology of BVDV persistence has been examined and reviewed.30 The interaction of NCP BVDV in pregnant animals demonstrated that heifers carrying PI calves develop BVDV antibody titers 5 to 10 times higher than heifers carrying non-PI calves. The inability of NCP BVDV to induce IFN-alpha in the fetus is one of the major immune evasion mechanisms that allows BVDV to establish persistence. The major mechanism for persistence is tolerance of the CD4+ cells. The specificity is very high, as PI animals can respond to homologous virus changes as small as a single amino acid. This explains why some PI animals can develop an antibody response to the homologous virus from the multiple BVDV quasispecies that will arise as the PI animals mature. Experimental infection of PI animals with antigenically related CP BVDV resulted in 50% developing mucosal disease. Those which did not develop mucosal disease had higher levels of circulating gamma-delta T-cells before the challenge with CP BVDV. Vaccination of BVDV type 1 b tolerant PI animals with vaccines containing either CP BVDV type 1 a or a NCP type 1 b and also Mannheimia haemolytica resulted in an BVDV antibody response only in those animals receiving vaccines containing the heterologous type 1a. All of the PI animals had lower M. haemolytica antibody response.
Following natural infection of seronegative immunocompetent cattle with most of the strains of BVDV which do not cause severe disease, there is a transient viremia; serum neutralizing antibodies develop within 2–3 weeks, peak at 8–10 weeks, and remain detectable for many months. The humoral immune response after natural BVDV infection in cattle is considered to be lifelong, and includes antibodies to a range of virus-encoded proteins, including the immunodominant surface glycoprotein gp53 and the highly immunogenic, non-structural, catalytic serine protease NS2-3.34 Vaccination of naïve cattle with inactivated BVDV vaccines, results in virus neutralization peak titers at about 5 weeks after the second vaccination with a return to seronegativity within 12 weeks of vaccination.35 This pattern of response is typical of inactivated vaccines. Experimentally and naturally infected animals may have moderate to high levels of serum neutralizing antibodies to the virus for three years after being infected.34 The ability to cross the placenta of susceptible pregnant animals and cause a variety of fetal infections is the most important evidence of the success of the BVDV in the evasion of the host immune system.30
The high percentage of animals which are seropositive in the cattle population or in herds which have experienced the disease is due to the presence of PI animals in the herd. Vaccination of immunocompetent cattle with the live virus vaccines induces a broad spectrum and durable immunity. It is generally accepted that cattle respond to natural infections or vaccination with modified live-virus vaccines with a long-lasting immunity, and it is likely that the immune response includes cell-mediated immunity. Immunization with an inactivated virus vaccine may result in an only very short-lived immunity with a narrow antigenic spectrum. Existence of neutralizing antibodies is generally considered to be the most significant predictor of an effective immune response.36 The presence of neutralizing antibodies in breeding females will protect the fetus against BVDV infection during pregnancy. Passively acquired antibodies, usually IgG, protect against nasopharyngeal shedding of the virus, and reduce viremia in challenge-inoculated calves. There is considerable antigenic variation among strains of the virus but there is also considerable cross-protection against heterotypic strains of the virus. Recent outbreaks of severe disease due to type II BVDV infections occurred primarily in herds which were not properly vaccinated. Current vaccines derived from type I strains appear to protect against infections with type II strains.36
Colostral antibody in calves lasts until 4–6 months depending on the initial level achieved after the ingestion of colostrums.37 The half-life of the antibody is 21 d in normal calves but in persistently-infected calves titers decline more rapidly and by 4–8 weeks no antibodies may be detectable.
The predicted ages of dairy calves when colostrum-derived BVDV antibodies would no longer provide protection against disease or interfere with vaccination has examined.33 About 50% of calves become seronegative for BVDV type 2 by 114 days of age. Rate of antibody decay was significantly associated with antibody titer at 1 to 3 days of age and with whether calves were congenitally infected with the virus. Three-month old calves were predicted to have a mean BVDV type-1 antibody titer of 1:32 and mean BVDV type-2 antibody titer of 1:16. These data can be used to estimate the age by which a group of calves would be expected to lose passive protection.
Passively acquired antibodies can prevent clinical disease in experimentally inoculated calves at 2 to 5 weeks of age and protection continues to exist after the decay of passive antibodies which implies the existence of additional immune mechanisms other than serum antibodies.38
PI calves are infected during early fetal life and are born seronegative and immunotolerant to the specific strain of virus in their tissues. Most PI calves remain seronegative to specific virus but will respond immunologically to other pathogens.
The immunosuppressive effects of the BVDV are discussed later under that heading.
Environmental and management risk factors
The major management risk factors are the introduction of PI animals into a susceptible herd and the failure of a vaccination program or an inadequate vaccination program. In the recent outbreaks of severe disease in cattle herds in Ontario and Quebec, failure to vaccinate or failure to use the vaccine properly was a common historical finding.39
Pathogen risk factors
The bovine pestivirus is one of the most widespread and important virus infections in cattle throughout the world. While only one serotype of BVDV is recognized, isolates of these viruses vary genomically, antigenically and biotypically. These pathogen characteristics are important in the pathogenesis of the various diseases associated with the virus, the immune response of animals to different isolates of the virus, and the laboratory diagnosis.
Positive-strand RNA viruses, like BVDV, are subject to genomic modifications that involve point mutations or recombination of RNA and thus are highly mutable.40 The genetic diversity among isolates of the virus is characteristic of RNA viruses which exist in nature as quasispecies (a swarm of viral mutants). The high frequency of mutation, propensity for recombination, and selective pressure from immune responses stimulated by natural infection or vaccination has led to the creation of a large assortment of genetic and antigenic variants of the virus. The consequences of diversity include diversity of clinical disease, diagnostic difficulties, and vaccination failures.2,40
Several isolates have been identified that are antigenically related, but there may be antigenic variants that are immunologically distinct. In addition to antigenic diversity among strains, there are major molecular differences between the same strains. Differences in neutralizing antibody titers against specific isolates of BVDV are detectable in polyclonal serum from convalescent cattle. Monoclonal antibodies that have neutralizing activity differentiate BVD viruses into several groups. The antigenic variability of this virus may also explain the wide range of lesions and disease complexes which have been observed. Outbreaks of disease associated with the virus in commercial beef herds in Argentina were characterized by different clinical manifestations such as mucosal disease, enteritis and generalized dermatitis each caused by different genotypes of the virus.41 This requires further virology and molecular studies which are necessary to improve diagnostic methods and formulate effective vaccines. The practical consequences of antigenic diversity are that neither natural infection nor vaccination can provide complete protection against most of the naturally occurring strains. There is also considerable cross-reactivity between isolates of the virus, which explains why properly vaccinated animals have considerable immunity.
Phylogenetic analysis of the viruses from persistently infected cattle on a number of farms in Sweden found a strict herd-specific clustering of the virus.42
The BVDV can be segregated into two subgroups termed BVDV I and BVDV II.1,2 Type 2 isolates are those commonly used in vaccine production, diagnostic tests and research. Type II isolates are predominantly from fetal bovine sera, persistently infected calves born to dams vaccinated against BVDV, and cattle which have died from an acute form of BVDV termed ‘hemorrhagic disease’. The epidemic of BVDV-associated disease in cattle in Quebec in 1993, associated with morbidity rates of up to 80% in some herds, and mortality rates up to 30%, was associated with isolates belonging to type 2.43 Experimentally, type 2 BVDV induces the highest degree of viremia and more pronounced lesions and more extensive distribution of viral antigen compared to Type 1 BVDV which induced the lowest.44,45
Non-cytopathic and cytopathic BVD virus biotypes.
Bovine viral diarrhea viruses exist as non-cytopathic or cytopathic biotypes.1,2 Non-cytopathic biotypes produce little if any visible cytopathic change in cell cultures, and infected cells generally appear normal. Cytopathic biotypes, in contrast, cause cellular vacuolation and cell death. The biotypes are classified by their ability or lack of ability to cause overt cytopathic change in cell cultures without reference to their ability to cause disease in the animal. The two biotypes of BVDV are not distinguishable serologically. However, at the molecular level, cytopathic viruses produce one additional protein known as p80. There is strong evidence that cytopathic viruses are derived from non-cytopathic viruses by mutation. In most cases RNA recombination is responsible for the generation of the cytopathic viruses.39 A second method is based on the introduction of a set of point mutations within the NS2 gene. The non-cytopathic virus is thus the natural ‘wild-type’ virus.
The BVDV is maintained in the environment by PI animals which were infected by non-cytopathic virus in utero from 42–125 d of gestation, when their immune system does not recognize the persisting virus as foreign and the animal is said to be immunotolerant. In general, PI animals lack neutralizing and non-neutralizing antibodies to the BVDV. The immune tolerance is highly specific, as these animals can mount an immune response to superinfections from antigenically dissimilar BVDV. PI animals with BVDV antibodies, other than calves with passively acquired colostral antibody, probably acquire them because they have encountered a BVDV, via natural exposure or vaccination that is dissimilar antigenically. There is no evidence that cytopathic viruses can produce persistent infections.
The non-cytopathic BVDV, the NADL-A strain, infects the bovine fetus following oronasal exposure of the pregnant dam, whereas the closely related cytopathic NADL-A are incapable of fetal infection.46 The inability of the CP BVDV to establish fetal infections following oronasal exposure explains the preponderance of NCP BVDV in the cattle population. The extraordinary ability of the NCP BVDV to establish fetal infections leading to the birth of PI calves provides a robust viral reservoir. Shedding and horizontal transmission maintains a high degree of viral infections in cattle herds.
In some PI animals, viral antigen may be widespread in many tissues, including the cerebellum and other parts of the brain, spleen, kidney, lungs and parts of the intestine. Yet, the PI calf is often born normal and vigorous, and reaches adulthood due to the non-pathogenicity of the virus. But the virus continues to replicate lifelong in the tissues of these animals and virus is shed continually into the environment.
Mucosal disease is the result of PI animals being superinfected with a cytopathic virus that is antigenically similar to the persisting non-cytopathic virus and both types are isolated from cattle with mucosal disease. Because of the antigenic similarity of the viruses, it is thought that the immune system of the PI animal does not recognize the cytopathic virus as foreign and thus fails to protect the animal from severe disease. The uncontrolled infection with cytopathic virus causes the severe lesions in mucosal disease.
Until recently, it was thought that postnatal infections of non-pregnant cattle with BVDV were benign, and only sometimes produced clinical disease. Following infection of immunocompetent animals, there was viremia, mild fever and diarrhea and recovery. The animals seroconverted, which accounted for the high percentage of normal animals in the cattle population which were serologically positive. In the late 1980s and early 1990s in the northeastern United States, Ontario, and Quebec, virulent BVD non-cytopathic viruses emerged which caused severe acute disease in both calves and adult cattle.43 The majority of viral isolates were non-cytopathic and were typed as BVDV 2.43 Thrombocytopenia and hemorrhagic disease associated with non-cytopathic BVDV has been recognized in adult dairy cattle and weaned beef calves. The disease occurred in veal calves in the same geographical area. In addition, highly virulent BVDV are causing severe diarrhea and death in adult cattle with clinical findings and lesions similar to those of acute mucosal disease. There are now reports of severe illness resembling acute mucosal disease in adult cattle in Britain, attributed to infections with non-cytopathic BVDV. Type 2 viruses have been isolated in British cattle.47 Only non-cytopathic BVDV have been isolated from these animals. All of the available evidence suggests that these animals are immunocompetent and not persistently infected but they may be non-immune because of lack of vaccination or acquired antibody. Type 2 BVDV has been isolated in Slovakia which means that this genotype is now in central Europe.48 Experimentally, differences in virulence between two non-cytopathic bovine viral diarrhea viruses in calves have been described. Type 2 BVDV strains have been isolated in Brazil affecting young cattle with severe gastrointestinal and respiratory disease.49
Antigenic similarity between the biotypes is a consistent finding in animals dying from fatal mucosal disease, which suggests that cytopathic strains may arise by mutation from non-cytopathic strains. The literature on the molecular biology of the virus has been reviewed.6 Analysis of viral proteins with monoclonal antibodies has yielded detailed information about the antigenic composition of both structural and non-structural proteins.
Economic importance
In Canada, the BVDV disease complex is considered as one of four infectious diseases known as production limiting diseases in dairy herds.50 The other three are Johne’s disease, enzootic bovine leukosis, and neosporosis. These diseases are present on many Canadian dairy farms and have significant economic loss due to disease and lowered productivity. The direct production losses (milk loss, premature voluntary culling and reduced slaughter value, mortality loss, and abortion and reproductive loss) and treatment costs (veterinary services, medication cost, an extra farm labor cost) due to four infectious diseases in the Maritime provinces of Canada were determined.51 Total annual costs for an average, infected, 50 cow herd in 1997 were: Johne’s disease $2472; BVDV $2421; neosporosis $2304; enzootic bovine leukosis $806. The largest effect on costs was due to milk yield effects.
Calculation of the losses associated with BVD outbreaks in dairy herds vary widely.52 In most cases the estimated losses only include those due to abortion and dead animals whereas indirect effects such as increasing the risk to other diseases are not included and therefore are considered as conservative estimates. The economic losses associated with outbreaks of the various forms BVDV infection in herds vary from a few thousand up to $100 000.52 Most estimations of losses at the national level range from $10 and 40 million per million calvings. National eradication schemes as done in Scandinavian countries has been cost-effective.52
The economic losses associated with the introduction of the BVDV into a susceptible herd of pregnant cattle are due to abortion, congenital defects, stillbirths, increased neonatal mortality, increased occurrence of other infectious diseases, prenatal and postnatal growth retardation, suboptimal reproductive performance due to infertility, deaths from mucosal disease, and the early disposal of PI animals. Large losses due to fetal infection occur during the first 2–3 years following introduction of infection to a susceptible herd.10 The economic losses are high when epidemics of fatal mucosal disease occur. While the incidence of mucosal disease is usually below 5%, it can be as high as 22%, which is costly because of veterinary visits, the submission of animals to the diagnostic laboratory, the death of animals, and the anxiety the epidemic creates in the mind of the producer who wonders if the entire herd will become affected.
Economic losses due to BVDV infection vary depending on the immune status of the population and the pathogenicity of the infecting virus strains. Introduction of the infection into a totally susceptible population invariably causes extensive losses until a state of equilibrium is reached. Infection with highly virulent strains cause severe clinical disease and death.
The magnitude of the losses in an infected herd may be expected to fluctuate. They may be relatively large with the occurrence of disease on an epidemic scale after initial horizontal transmission to non-immune pregnant cows, but considerably lower when endemic infection is maintained in the herd through the presence of viremic families. However, a further phase of high losses may occur should management allow heifers to reach breeding age without being exposed to infection or vaccinated. A linear programming model to estimate the economic impact of bovine viral diarrhea at the whole-farm level has been described.53
Using the output from an epidemiological model of an outbreak of BVD in a Scottish beef suckler herd, the estimated losses associated with an outbreak of BVD were 59 Euros mean loss per cow per annum without taking into account any financial premiums associated with disease-free status of the herd.54 Two highly significant areas of loss often ignored in the field are immunosuppression and reproductive failure.54
PATHOGENESIS
The pathogenesis depends on multiple interactive factors. Host factors which influence the clinical outcome of BVDV infection include:
• Whether the host is immunocompetent or immunotolerant to the virus
• Transplacental infection and gestational age of the fetus
• Induction of immune tolerance in the fetus and the emergence of fetal immune competence at about 180 d of gestation
• Immune status (passively derived or actively derived from previous infection or vaccination)
• Presence of stressors. Genetic diversity among isolates may account for differences in the clinical response to infection. Differences in virulence between non-cytopathic isolates and between genotypes have been described.7 Apart from those infected with the virus in utero, most cattle are immunocompetent to the virus and will successfully control a natural infection, develop antibodies and eliminate the virus so that latency and shedding does not occur.
The consequences of infection with the BVDV are divided into the following different categories based on the status of the animal. There is a spectrum of clinical responses based on the host factors and the virulence of the isolates involved.
Immunocompetent non-pregnant cattle
Subclinical BVDV
This is a subacute infection in seronegative, immunocompetent cattle usually after colostral immunity has waned and it occurs in both sexes and any class of cattle. It is usually a clinically unrecognizable infection with the development of serum-neutralizing (SN) antibodies and elimination of the virus from normal immunocompetent animals. This accounts for the high percentage of normal animals that are serologically positive to the virus. A mild transient clinical disease characterized by inappetence for a few days, depression, fever, mild diarrhea, transient leukopenia and recovery in a few days may occasionally occur.
In some cases, outbreaks of diarrhea occur in animals ranging from 6 months to 1 year of age, characterized by high morbidity and low or no mortality. The most likely source of infection is PI animals in the herd.
Peracute bovine virus diarrhea
A severe and highly fatal form of bovine virus diarrhea associated with non-cytopathic Type II isolates of the BVD virus is recognized.43 Outbreaks were most common in dairy herds with inadequate vaccination programs and which recently introduced animals into the herd.
Thromboctyopenia and hemorrhagic syndrome
Thrombocytopenia and the hemorrhagic syndrome occurs in adult cattle and veal calves affected with the peracute form of BVDV infection.55 Platelet counts are reduced to below 25 000/μL, and clinically are bloody diarrhea, petechial and ecchymotic hemorrhages of the sclera of the eyes, epistaxis and abnormal bleeding from injection sites. Hyphema may also occur. Thrombocytopenia due to destruction of platelets has been reproduced experimentally in young calves by inoculation of the BVDV. type 2 BVDV isolates are most commonly associated with the hemorrhagic syndrome. Experimentally, altered platelet function occurs in calves with type 2 BVDV isolates but not with type 1 BVDV strains.56 In calves experimentally infected with ncp type 2 BVDV isolates of different virulence induced clinical signs and cytopenia which appeared to be proportional to infection severity.57 The important virulence characteristics are duration of neutropenia, severity of thrombocytopenia, delayed increase in proliferating myeloid cells, and the presence of virus in bone marrow percursor cells. Infection of bone marrow megakaryocytes myeloid cells may also be involved. The North American hypervirulent type 2 BVDV induces severe thrombocytopenia, profuse diarrhea and pneumonia in all experimentally infected calves, while none of the European strains tested, all belonging to genotype 1, induced significant pathological signs even though isolated from cases of hemorrhagic syndrome.57 It is suggested that induction of sporadic hemorrhagic syndrome by BVDV type 1 requires the presence of other co-factors.
Osteopetrosis, anemia, thrombocytopenia and bone marrow necrosis can occur in beef calves naturally infected with type 2 strains of the virus. Experimental infection of calves with the non-cytopathic virus causes thrombocytopenia whereas cytopathic virus did not.
Diarrhea of neonatal calves
The role of the virus causing diarrhea in calves under a few weeks of age is uncertain. Naturally occurring cases of acute neonatal diarrhea due to infection with the virus in immunocompetent calves under 6 weeks of age have been reported only rarely. Calves born with PI status may be unthrifty and be affected with chronic diarrhea and pneumonia as young calves. However, if the virus causes diarrhea in calves the pathogenesis is not clear. Calves born from cows free of the infection are not likely to be exposed to the infection. Immunocompetent dams provide colostral immunity to their calves, which should protect them against viremia due to BVDV for 6 months or longer.59 Fatal enteritis has been reproduced experimentally by infecting colostrum-fed and colostrum-deprived neonatal calves with the virus.7 In older colostrum-fed calves, experimental infection resulted in mild disease with rapid recovery. Experimentally, calves from 7–50 d of age with colostral virus neutralizing (VN) antibody titers below 256 or lower, developed a fever and systemic spread of the virus when challenged with the virus.59 Calves with titers lower than 16 developed severe clinical disease characterized by fever, leukopenia, thrombocytopenia and diarrhea. The severity and duration of clinical signs decreased as titers of passively acquired viral neutralizing antibody increased. Another requirement for effective protection, is that the colostral antibody must be specific for the virulent virus. Experimentally, the intranasal inoculation of healthy BVDV-free calves 6 months of age with either the non-cytopathic or cytopathic BVDV results in a mild form of entercolitis, and mild follicular lymphocytic depletion.
Meningoencephalitis
A type 2 BVDV strain has been isolated from the brain tissue of 15-month-old heifer with neurological clinical findings and pathologic evidence of multifocal meningoencephalitis.60 This suggests a neurovirulent strain of the virus.
Immunosuppression
There is evidence that postnatal BVDV infections of cattle can cause immunosuppression and enhance the development of other infectious diseases.36 However, the evidence is controversial and must take into account the immune mechanisms of PI animals compared to animals with primary infections. There is circumstantial evidence that BVDV infections may be a major factor in multiple etiological diseases such as pneumonia and enteritis. Cytopathogenic isolates from genetic cluster 1 d of BVDV type 1 experimentally induced a primary respiratory disease in previously seronegative and immunocompetent calves.61 All infected calves seroconverted and contact calves also developed a respiratory infection following exposure to infected calves.
The lesions of BVDV in cattle suggest immunosuppression because of lymphoid depletion and neutropenia.36 Similarly, some of the modified live-BVDV vaccines are considered immunosuppressive in calves or may potentiate intercurrent infections.
In vitro evidence of immunosuppression.
This indicates that the virus interferes with lymphocyte and macrophage function. BVDV infection of peripheral monocytes in vitro causes a significant decrease in their random locomotion and chemotactic response to a lymphokine. These abnormalities could impair the ability of the host to localize monocytes and macrophages in the vicinity of other infections. Neutrophils from BVDV-infected cattle may have their bactericidal, fungicidal and virucidal mechanisms impaired, which could increase susceptibility of BVDV-infected cattle to secondary infections. Alveolar macrophage function is reduced in calves experimentally infected by the respiratory route with a cytopathic biotype of the virus and with the other immunosuppressive attributes of the virus, could favor a predisposing role for the virus in the pathogenesis of respiratory disease in calves.22
Experimentally, the virus can:
• Alter neutrophil function36
• Impair immunoglobulin secretion by peripheral lymphocytes
• Allow the infectious bovine rhinotracheitis (IBR) virus to be more widely distributed in various tissues and infect tissue culture cells
• Cause the release of substances which can suppress the proliferative response of bovine mononuclear cells to blastogenic substances.
Impairment of neutrophil function in cattle persistently infected with the BVDV differs from impairment of neutrophil function in healthy cattle mounting an immune response to the infection.36
In vivo evidence of immunosuppression.
Primary postnatal infections cause a transient reduction in the absolute number of T- and B-lymphocytes and in the percentage of T-lymphocytes. The evidence incriminating the virus as a predisposing pathogen in naturally occurring cases of bovine respiratory disease is largely circumstantial. The presence of the virus in the respiratory tract tissues of cattle affected with pneumonia is difficult to interpret. Several different viruses have been incriminated in the cause of acute bovine respiratory disease but experimental evidence to support their involvement has centered on the IBR and PI-3 viruses.
In outbreaks of respiratory disease in calves and adult cattle associated with multiple viral infections, the BVDV is often the most frequent viral agent. This could indicate that the virus is an important contributory pathogen in respiratory disease of cattle.
Experimentally the BVDV can facilitate the colonization of P. haemolytica in the lungs, resulting in severe pulmonary lesions.36 Severe fibrinopurulent bronchopneumonia and pleuritis involving 40–75% of lung volume developed in calves experimentally inoculated sequentially with the BVDV and P. haemolytica. However, in some experiments the BVDV has no effect. BVDV may be present with other pathogens, such as those viruses or Pasteurella sp. and this may indicate that synergism occurs. However, it is also possible that the virus may be coincidentally present in some animals and have no adverse effect. It has been argued that there is no substantive evidence to implicate the BVDV, as it occurs in the benign form of the disease, in the pathogenesis of naturally occurring acute undifferentiated respiratory disease of cattle. Field observations suggest that following the introduction of BVDV infection into a susceptible herd, there may be an increased incidence of viral and bacterial pneumonia in the calves, which may continue for up to 2 years.
BVDV types 1 and 2, along with the PI-3 virus, and the BRSV, have been isolated from lung tissue in recently weaned beef calves with acute respiratory disease soon after arrival in the feedlot.62 The BVDV1b was the predominant subtype identified in recently weaned beef calves affected with pneumonia due to Mannheimia haemolytica and Pasteurella multocida.63
Bovine viral diarrhea virus in the feedlot
There is epidemiological evidence that the BVDV may be one of the most economically important infectious pathogens of feedlot cattle.64 The Academy of Veterinary Consultants has proposed that the beef and dairy cattle industries adopt measures to control and target eventual eradication of BVDV from North America.65 The immunosuppressive potential of the virus or its synergistic effects with other pathogens are considered to be associated with bovine respiratory disease in feedlot cattle. Individual, unknown persistently infected animals which are purchased and introduced into the feedlot, serve as reservoirs of the virus for naïve cattle which are subsequently co-mingled in the feedlot. BVDV has been incriminated in bovine respiratory disease in feedlot cattle from which pathogens such Mannheimia haemolytica, Mycoplasma bovis, Histophilus somni, infectious bovine rhinotracheitis virus, have been isolated from lung lesions.64 Chronic, antibiotic-resistant pneumonia, often with concurrent polyarthritis, occurs in feedlot cattle in western Canada. The prevalence of M. bovis and the BVDV in the tissues of affected animals suggests there may be synergism between the BVDV and M. bovis.66
There is considerable seroepidemiological evidence that the BVDV titers of feedlot cattle on arrival are associated with subsequent respiratory disease. Cattle arriving with a titer were at decreased risk of respiratory disease; those cattle which seroconverted after arrival were associated with increased risk of disease. Seroepidemiological studies of undifferentiated fever in recently weaned beef calves arriving in the feedlot indicates that animals arriving with a higher BVDV antibody titer were associated with a decreased risk of undifferentiated fever compared to those with lower levels on arrival.67 Persistently-infected calves introduced into the feedlot usually are unthrifty at the time of weaning, and most will die during the feeding period.10 Cross-sectional and cohort studies a large number of cattle arriving in a feedlot determined the prevalence and effect of PI animals on subsequent disease. The prevalence of PI cattle on arrival was 0.3%, 2.6% in chronically ill feedlot cattle, and 2.5% in dead cattle.68 The risk of initial treatment for respiratory disease was 43% greater in cattle exposed to a PI animal compared with those not exposed to a PI animals. Overall, 15.9% of initial respiratory tract disease events were attributable to exposure to a PI animal.68
Primary BVDV infections occur in feedlot cattle which are not persistently-infected and may be the inapparent or subclinical form or the peracute form of the disease. The thrombocytopenic form of BVDV infection has also been described in feedlot cattle.64
Ovarian dysfunction
Ovarian dynamics may be changed in cattle infected with BVDV.26 Ova exposed to the virus in vitro can have virus particles attached to the zona pellucida but the intact zona pellucida protects the developing embryonic cells from infection. However, following removal of the zona pellucida, cytopathic BVDV may have detrimental effects on survivability of blastocyts. Bovine follicular cells and oocytes are permissive to BVDV at all stages of follicular development69 and there may be a transient fall in estradiol secretion following acute infection; both may reduce fertility. Infection during the critical period of growth of preovulatory follicles causes varying degrees of necrosis of the granulosa cells which can result in a spectrum of ovarian dysfunction including retarded follicular growth, delayed ovulation and anovulation.70 Return to ovarian function following BVDV infection may take several months in some cases.
Immunocompetent pregnant cattle and fetal infections
The BVDV can cause significant early reproductive loss in non-immune pregnant cattle including fertilization failure, embryonic mortality and abortion.71 In addition, infection of the fetus between 42 and 125 d of gestation may result in persistently-infected fetuses which are carried to term and the calf may be born alive and thrive normally or be unthrifty.
The experimental and clinical observations of the effects of the virus on early parts of the reproductive cycle are conflicting. The virus can be transmitted by natural service or artificial insemination with the possibility of fertilization failure or early embryonic mortality, which in turn, leads to repeat breeding. The principal determinant of the outcome of in utero infection in cattle is the age of the conceptus and fetus when infection occurs. The BVDV can cause reproductive failure in susceptible cattle during the following stages of the reproductive cycle:
1. Infection prior to insemination. Exposure of cattle to the virus during the estrus cycle prior to insemination can result in a decrease in conception rate due to failure of ovulation or delayed ovulation. BVDV has been associated with ovaritis in infertile heifers.26 PI cows may have morphologic changes in their ovaries, suggesting a reduction in normal ovarian activities. It is not known if similar findings occur in cows acutely infected with the virus.
2. Insemination of cattle with semen containing bovine pestivirus. The insemination of seronegative and virus-free heifers with BVDV-contaminated semen can result in poor conception rates initially, followed by normal conception following seroconversion to the virus, and the birth of normal calves with no evidence of infection with the virus.26 Experimentally, the intrauterine infusion of the virus into cattle at the time of insemination has prevented conception and has been attributed to prevention of fertilization or simply recognized as an empty uterus at 5 weeks after breeding. It seems that intrauterine infection at the time of breeding may have some effects on the very early stages of reproduction, in addition to those that could be attributed to infection by other contact routes. Infection of susceptible cows either 9 d before or soon after insemination can result in a marked reduction in conception rates and significant embryo–fetal loss.26
The BVDV can be present in the semen of bulls either due to a persistent infection or an acute postnatal transient infection of the bull. The semen of an immunotolerant PI bull may be normal and the pregnancy rates of heifers bred by him may be normal thus the spermatozoa from an infected bull do not necessarily carry the virus. In other situations the quality of the semen of PI bulls may be abnormal. Acute infection of immunocompetent BVDV seronegative bulls with the virus can result in transient shedding of the virus in semen and to a marked deterioration of semen quality.26 The amount of virus in the semen of acutely infected bulls is much less than that found in the semen of PI bulls. Experimental acute infection of bulls with the BVDV can result in shedding of the virus in raw, unprocessed, and diluted and extended semen during and after the end of the period of viremia.26 The most productive sites of virus replication are in the seminal vehicles and prostate gland.26
The economic losses can be considerable if the virus is introduced into herds undertaking an embryo transfer and artificial insemination program.26 Infection may occur following the use of infected semen for artificial insemination or through the use of infected bovine serum as a transport medium or diluent for embryo transfer.
3. Infection during embryonic period: 0–45 d gestation. Natural BVDV infection of seronegative heifers 4 days after insemination results in viremia between 8 and 17 d and a decrease in conception rate and pregnancy rate compared to uninfected heifers.26 Infected heifers which fail to conceive return to estrus approximately 20 d later. Experimentally there is no indication of impairment of in vitro development of bovine embryos when they are exposed to the BVDV.72 The zona pellucida appears to prevent the virus from gaining access to the embryonic cells.73
Fetal infections
4. Infection during late embryonic–early fetal period: 45–125 d gestation. Following the infection of a non-immune pregnant animal the virus is capable of crossing the placental barrier and invading the fetus. In experimental infection of pregnant heifers with a noncytopathic strain of the virus at 85 days of gestation, fetal infection can occur 14 days post-infection without preceding or simultaneous high concentration of the virus in uterus or placenta.74 This supports the proposition that the passage of virus can occur via the vasculature and not via local cell-to-cell spread and that fetal infection can occur in the absence of significant levels of virus in the placenta.
Fetal infection can result in a wide spectrum of abnormalities from death of the fetus to congenital defects, to a persistent infection of the fetus until term and birth of a calf with lifelong infection without clinical signs. The results are mainly dependent on the stage of fetal development at which infection takes place. In general, the risk for the fetus is highest during early pregnancy. Infection of the fetus from 50–100 d of gestation may result in fetal death and expulsion of the fetus (abortion) from days to several months after fetal infection, or mummification. However, fetal survival following infection is common and can be as high as 70%.
Persistent viremia and mucosal disease. One of the most important effects of BVDV infections of the fetus is the development of PI animals. Following infection of the fetus with a non-cytopathic isolate of the virus from about 45–125 d of gestation, it will not develop serum virus-neutralizing antibodies and may be carried normally to term and be born with a persistent infection.75 Mucosal disease occurs in a proportion of these, and only in these, PI animals. From birth to the time of clinical disease, which usually occurs between 6 and 24 months of age, and rarely up to 3 years of age, these animals are persistently viremic and specifically immunotolerant to the homologous strain of the persisting virus.76 They may appear clinically normal or unthrifty and small for their age. Occasionally, PI cattle may survive and remain healthy for up to 5 years during which time they shed the virus in their mucous secretions and may be seropositive to a range of BVDV strains, including their own persisting strain. Calves with either transient or persistent infections with BVDV have lower than normal serum concentrations of thyroid hormones which may be associated with the retarded growth.77 The heart girth of PI calves is smaller than controls. PI animals are infected only with the non-cytopathic biotype of the virus and they excrete large quantities of the virus into the environment and serve as the major source of the non-cytopathic virus in a herd.
During the postnatal period, superinfection with a cytopathic isolate of the virus may precipitate fatal clinical mucosal disease in these animals. PI calves have been reproduced experimentally by the inoculation of fetuses with a non-cytopathic isolate of the BVDV from 42–125 d of gestation. Fatal mucosal disease has been reproduced by the inoculation of persistently viremic specifically immunotolerant calves with a homologous cytopathic isolate of the virus. It is suggested that the cytopathic virus has a qualitative preference for intestinal lymphoid tissue in older postnatal calves, which may not be sufficiently developed for the cytopathic virus to establish a persistent viremia in the young fetus. Following the experimental production of mucosal disease, the cytopathic biotype of the virus can be found in lesions of the lymphoid tissue of the small and large intestines, in Peyer’s patches, in intramural ganglia and in duodenal glands. Severe tissue damage is also related to the presence of the cytopathic virus. Both biotypes of the virus are present in animals which develop fatal mucosal disease.
It is now likely that mutation of the non-cytopathic virus to the cytopathic virus occurs within the animal rather than the introduction of a cytopathic virus from an infected animal introduced into the herd.75 The non-cytopathic biotype could mutate to a cytopathic biotype by taking up cellular sequences during a recombination event. Once a homologous cytopathic virus has arisen, it can quickly spread to other PI animals within the same group and this may explain the rapid development of an outbreak of fatal mucosal disease. Recombination between a non-cytopathic BVDV-1 virus and a cytopathic BVDV-1 vaccine virus causing mucosal disease 3 months after vaccination has been described but is probably rare.78
Typical mucosal disease occurs within 2–3 weeks following development of the antigenically homologous cytopathic virus in the PI animal. The affected cattle do not respond serologically to the homologous cytopathic virus. Superinfection with an antigenically heterologous cytopathic virus does not result in typical, but rather atypical, mucosal disease several months later, or not at all, and infected animal respond serologically to the heterologous cytopathic virus.75
The pathogenesis of the lesions of mucosal disease remains obscure.75 The viral antigen can be detected in many tissues including:
• Ileum and lymphoid tissue in the proximal colon
• Bronchiolar epithelial cells
The pathological changes which characterize the disease involve the integument and the epithelia of the respiratory and alimentary tracts as well as lymphoid tissues.
The basic lesion is a small vesicle ulcer which affects only epithelial cells. The erosions occur throughout the:
Vascular injury leading to vasculitis is a characteristic feature of the disease due to the pestiviruses, which may explain the type and distribution of the lesions which occur in fatal mucosal disease. The vascular injury may be initiated by degenerative changes of the endothelial cells; this may lead to formation of a thrombus, which can detach and circulate as emboli, and resulting in generalized vasculitis.
Death from acute mucosal disease usually occurs within 2 weeks of the onset of clinical signs and both cytopathic and non-cytopathic isolates of the virus can be recovered from the tissues of affected cattle.75
Animals that are immunotolerant to the BVDV are immunocompetent to other antigens.79 They develop neutralizing serotiters to the IBR and PI-3 viruses and agglutinating titers to P. haemolytica.36 They will also produce VN antibody titers, following the administration of commercial live-BVDV vaccine, against the vaccine virus as well as other laboratory strains. Furthermore, in spite of this antibody formation, the original virus will persist.
Spontaneous insulin-dependent diabetes mellitus associated with persistent BVDV infection in young cattle has been described.80,81 Lesions were present in the pancreatic islet cells.
5. Infection during fetal period: 125–175 d gestation. Congenital defects. Transplacental infection of the fetus approximately between 125–175 d of gestation can result in numerous congenital defects. This period of development corresponds to the final stages of organogenesis of the nervous system and the development of the fetal immune system, which can result in the generation of an inflammatory response to the virus.
Cerebellar hypoplasia occurs and ocular abnormalities consist of retinal atrophy, optic neuritis, cataract, and microphthalmia with retinal dysplasia.26 Calves with cerebellar hypoplasia are unable to stand and walk normally immediately after birth. Defects of the eyes result in varying degrees of blindness; the cataracts are obvious when they occur. Calves may be smaller than normal and have a curly hair coat.26
Congenital morphological defects follow infections which occur somewhat later in gestation than do infections resulting in persistent viremia, and may be due in part to the emerging immunological capability. The presence of either persistent infection or antibody is variable.
6. Infection during fetal period between 180 d gestation and term. Infection of the fetus with the BVDV after approximately 150 d gestation results in a fully competent immune response and elimination of the virus. At birth, the fetus has antibody to the virus but is virus free. The effects of late-gestation infections are not well documented but abortions, stillbirths, and weak calves are reported.
Border disease
Border disease of sheep is associated with an in utero infection with a related pestivirus which cross-reacts with the BVDV.18 Ewes are clinically normal, but affected newborn lambs have a hairy fleece, clonic rhythmic tremors and are unthrifty. Hypomyelination and abnormal cells occur in the central nervous system. The hairy birthcoats have been attributed to hypertrophy of primary follicles and medullation of wool fibers. Surviving lambs are also PI with the virus. The virus can be isolated in cell culture and detected by immunofluorescent staining of the peripheral leukocytes, cellular debris in urine and cerebrospinal fluid of lambs up to 1 year of age. Affected lambs, like calves, have no detectable serum-neutralizing antibody. Adult sheep, after recovery from infection by the virus, have no detectable virus in the leukocytes and have serum neutralizing antibodies. Pestivirus of sheep and cattle will readily infect the alternative species, both naturally and experimentally, but the role that such cross-reaction plays in causing the respective diseases has not been determined. More details are available under border disease.
Sheep are an excellent animal model for studying the pathogenesis of congenital BVDV-2 infection. Experimental infection of pregnant ewes with a Brazilian non-cytopathic BVDV-2 isolate results in many features of BVDV-1 infection of pregnant sheep and cattle.82 Transplacental transmission of the virus to the fetuses is very efficient and results in fetal and perinatal deaths and the production of persistently-infected viremic lambs. The bovine pestivirus can spread to the ovine fetus within 4 days following intranasal inoculation of ewes in early pregnancy in the absence of maternal immunity.83 An enteric disease characterized by diarrhea and unthriftiness in 6- to 12-month-old lambs, and death in 3 to 14 days was similar to mucosal disease in cattle.84 At necropsy, the changes resembled mucosal disease in cattle and the Border disease virus was isolated.
CLINICAL FINDINGS
Inapparent or subclinical infection (bovine virus diarrhea)
The most frequent form of BVDV infection in cattle is non-clinical or a mild disease of high morbidity and low case fatality characterized by a mild fever, leukopenia, inappetence and mild diarrhea followed by rapid recovery in a few days and the production of virus-neutralizing antibodies. This form occurs in immunocompetent seronegative cattle which are infected in postnatal life, accounting for the high proportion of adult animals which possess SN antibodies.7 The literature commonly refers to this subclinical infection as bovine virus diarrhea. A similar infection, with no long-term consequences other than the development of antibody, can occur in fetuses over about 150–180 d gestation.
Acute mucosal disease
The acute mucosal form of the disease is characterized by the sudden onset of clinical disease in animals from 6–24 months of age which were infected during early fetal life. The morbidity is low and the case–fatality rate is high (over 90%). Within herds, 5–25% of animals in this age group may develop the disease over a period of several days or sporadic cases may occur over several weeks or months. Morbidity rates of 44% and case–fatality rates of 100% have been reported in isolated herds. Well-nourished, thrifty and previously clinically normal animals can be affected. In severe outbreaks, deaths from mucosal disease may account for only a proportion of the actual number of PI animals in the herd; some of the PI animals may have been culled, died for other reasons or were slaughtered. Following outbreaks of mucosal disease in a herd, there may be a rapid decline in the number of PI animals born in the subsequent few years because of spread of the infection and development of acquired immunity in the breeding females.
Affected animals are depressed, anorexic and drool saliva, wetting hair around the mouth. Fever 40–41°C (104–105°F), tachycardia and polypnea are common. Ruminal contractions are usually absent and a profuse and watery diarrhea occurs 2–4 d after the onset of clinical illness. The feces are foul smelling and may contain mucus and variable quantities of blood. Occasionally, small tags of fibrinous intestinal casts are present. Straining at defecation is common and the perineum is usually stained and smeared with feces.
Lesions of the oral cavity mucosa consist of discrete, shallow erosions which become confluent, resulting in large areas of necrotic epithelium becoming separated from the mucosa. These erosions occur:
The entire oral cavity may have a ‘cooked’ appearance with the grayish colored necrotic epithelium covering the deep-pink, raw base. Similar lesions occur on the muzzle and may become confluent and covered with scabs and debris. Although the oral lesions are significant in the identification of the disease, they may be absent or difficult to see in up to 20% of the affected animals. Esophagoscopy has been used for visualization of the typical erosions of the mucosa of the esophagus.85
There is usually a mucopurulent nasal discharge associated with some minor erosions on the external nares and similar lesions in the pharynx. Lacrimation and corneal edema are sometimes observed. Lameness occurs in some animals and appears to be due to laminitis, coronitis and erosive lesions of the skin of the interdigital cleft, which commonly affect all four feet.
Dehydration and weakness are progressive and death occurs 5–7 d after the onset of signs. In peracute cases, which die within a few days after the onset of illness, the diarrhea may not be evident even though the intestines are distended with fluid. Presumably, there is paralytic ileus and the intestinal fluid is not moved down the intestinal tract.
Chronic mucosal disease
Some acute cases of mucosal disease do not die within the expected time of several days and become chronically ill. There may be intermittent bouts of:
Shallow erosive lesions covered with scabs can be found on the perineum, around the scrotum, preputial orifice and vulva, between the legs and at the skin– horn junction around the dew claws, in the interdigital cleft and at the heels, and there may be extensive scurfiness of the skin. The failure of these skin lesions to heal is an important clinical finding suggesting chronic mucosal disease. Chronic cases will sometimes survive for up to 18 months during which time they are unthrifty and ultimately die from chronic inanition.
The chronic clinical form of the disease described above must be distinguished from the unthrifty persistently viremic animal described next.
Unthrifty PI calves
Calves which are born PI may be smaller in body size than their contemporaries and may fail to grow normally. A curly haired coat is characteristic of some PI calves.13,14 They may survive and appear unthrifty for several months or more until they develop fatal mucosal disease or some other infectious disease such as pneumonia. While these calves are stunted and unthrifty in appearance they do not have detectable clinical evidence of mucosal disease and they are seronegative to the BVDV. The birth of a high percentage of PI calves may result in a high incidence of fatal respiratory disease when the calves are 7–9 months of age.
Peracute bovine virus diarrhea
This is a severe form of the enteric form of the disease; it is often highly fatal, occurs in immunocompetent seronegative cattle in postnatal life and is associated with highly virulent isolates of the Type II virus.21,39 Dairy herds, beef breeding herds and beef feedlots have been affected in the outbreaks recorded in Ontario,43 Quebec and Pennsylvania (in the United States), and in the United Kingdom in the early 1990s. Inadequate biosecurity of animals imported into the herd, and the failure to vaccinate for BVDV or an inadequate BVDV vaccination program were common risk factors in affected herds. In affected herds, all ages of cattle are affected including calves, yearlings and adults. Mortality was highest in the young-age groups.
The most common complaint given by the owners was the presence of respiratory disease in affected animals. The outbreaks were slowly progressive and lasted for several weeks. Severe depression, respiratory distress, anorexia, profuse watery diarrhea, dysentery, conjunctivitis, fever up to 42.0°C, and agalactia in adult lactating dairy cows were common. Oral erosions were inconsistent. Abortion, usually in late gestation, was a common but inconsistent occurrence. Morbidity rates may be up to 40% and crude mortality rates may reach 25%. Many animals may die within a few days after the onset of clinical signs, and persistently infected calves were commonly born several months following the outbreaks.
Thrombocytopenia and hemorrhagic disease
Thrombocytopenia and hemorrhagic disease have been associated with BVDV infection but whether or not the affected animals were previously PI is uncertain; only non-cytopathic BVDVs have been isolated.55,58 Bloody diarrhea, petechial and ecchymotic hemorrhages of the visible mucosae, hyphema, epistaxis and prolonged bleeding from injection sites or insect bites have been observed. Cattle have platelet counts of less than 25 000/fl and clinically there is bloody diarrhea. Fever, diarrhea, rumen stasis and dehydration are also common. The case– fatality rate is approximately 25%; survivors can recover and thrive normally or remain unthrifty. A similar syndrome of thrombocytopenia has been described in weaned beef calves but the virus could not be isolated from affected calves.64
Reproductive failure and neonatal disease
The introduction of BVDV infection into groups of susceptible breeding females around the time of insemination and during the embryonic early- to mid-fetal period can result in conception failure, increased embryonic mortality, fetal mummification, abortion, premature births, stillbirths, congenital defects, the birth of stunted weak calves, and the birth of PI calves which subsequently may develop mucosal disease.26 Following introduction of infection into a beef herd, losses may be insidious and characterized by reduced pregnancy rates, abortions, excessive postnatal calf losses and the premature culling of young cows because of their failure to wean a well-grown calf.86 These losses including those due to mucosal disease in PI animals may continue for 2–4 years.10 In dairy herds in Norway, time to first calving was increased by 14 to 16 days in the second year after seroconversion, with the effect restricted to young stock.55 Studies in dairy herds in Switzerland indicate that infection with the virus during the first 45 days of gestation did not influence the rate of return to estrus.87 By contrast, there was an increase in the abortion rate in mid-term gestation (days 46 to 210) while no such effect occurred in animals which seroconverted later stages of gestation. At least 7% of fetal deaths were attributable to infection with the virus.
A large scale assessment of the effect associated with BVDV infection on fertility of dairy cows in 6149 herds in France, found that the virus was associated with an increase in the risk of embryonic death and fetal death.88
Under field conditions, the effect of subclinical BVDV infection on subsequent dairy heifer fertility may be due to a complex interrelationship among multiple BVDV infections dependent on the type of and timing of infection relative to reproductive performance. A high BVDV type 2 antibody titer (1:4096) in dairy heifers at 10 months of age was associated with 32 more days to conceive, compared with a low titer (1:128).89 Conversely, infection with BVDV by 5 to 6 months of age and a high BVDV type 2 titers one month before conception or breeding was associated with improved fertility. Heifers with evidence of congenital BVDV infection had lower fertility than non-infected heifers (15–42 days longer time-to-first AI), which depended on BVDV type 2 titers at 10 months of age.89
In beef herds, although abortions due to BVDV may occur at any time during gestation, typically several cows in a herd abort during a short period of time before the start of the calving season. At the beginning of the calving season, premature births and stillbirths occur. Some calves are born alive, take a few breaths and die. Weak calves are generally born during the first 2–4 weeks of the calving season. Affected calves are weak and flaccid at birth, and may appear small or normal. Death usually occurs within several hours despite intensive care and the feeding of colostrum. The prevalence of fetal infection with BVDV can be as high as 21%.90 In a study of health and performance in 213 dairy herds in Sweden, the risks for clinical mastitis, retained placenta, and estrus-stimulating treatments were higher and the calving intervals were longer in those herds with an increasing or maintained high prevalence of BVDV antibody-positive cows.91 A persistent BVDV infection in a dairy herd severely affects reproductive performance of heifers and cows at risk and can have an adverse effect on calf health over a period of time. In multiparous cows giving birth to PI calves there may be increased gestation lengths and retained placentas.
Congenital defects in calves
A number of congenital defects in calves are present26 including: microencephalopathy, hydrocephalus, hydranencephaly, porencephaly, cerebellar hypoplasia, and hypomyelination. Cerebellar hypoplasia was the first recognized teratogenic effects of the virus and has been well documented. At birth, affected calves exhibit varying degrees of severity of ataxia, wide-based stance, stumbling gait, and falling backwards when attempting to walk. Mildly affected calves may survive if hand-fed and managed carefully but severely affected cases usually die or are euthanized. Other congenital defects include cataracts, microphthalmia, optic neuritis, retinal degeneration, thymic hypoplasia, hypotrichosis and alopecia, curly hair coat, hyena disease, deranged osteogenesis, mandibular brachygnathism, and growth retardation.
CLINICAL PATHOLOGY
The clinical diagnosis of mucosal disease is usually made on the basis of the presence of characteristic clinical and pathological findings. A severe leukopenia is characteristic of acute mucosal disease. The decrease is commonly to below 50% of normal and total leukocyte counts of 1000–3000/μL are common and may persist for weeks.
The laboratory diagnosis of BVDV infections relies on the isolation or detection of the virus or components and/or the demonstration of a serological response to the virus. The literature on the laboratory diagnosis of bovine virus infections has been reviewed.92
The type of samples to be submitted depends on the clinical and herd history, whether acute or persistent infections are suspected, and the vaccination history is needed to interpret serology.
Virus isolation
Despite recent advances in BVDV diagnostic science, culture and identification of the BVDV from clinical specimens remains the ‘gold standard’ diagnostic technique.92 Strains of virus can be characterized in vitro as cytopathic or non-cytopathic biotypes. Cytopathic strains cause characteristic in vitro cell changes that are evident in inoculated cell cultures within 48 h. Most BVDV isolates obtained from field cases are non-cytopathic in cell culture. The isolated virus is recognized by identifying viral antigen in positive cell cultures by immunofluorescence or immunoenzyme staining. Virus isolation can be attempted by inoculation of nasopharyngeal and ocular swabs, semen, intestinal tissues, spleen, or most other tissues, or the buffy coat or serum of blood to cell cultures. In the live animal, the best sample for BVDV isolation is whole blood from which blood (buffy coat) cells are extracted and used as inoculum. Both cytopathic and non-cytopathic viruses have been isolated from the spleen or blood of individual cattle with mucosal disease. Whole blood or serum from PI animals is used for the isolation of the virus. The virus can be isolated successfully from blood which has been stored at room temperatures for up to 5 d before being processed.
For handling of large numbers of samples such as in a whole herd screening for PI animals, a microtiter virus isolation method, the immunoperoxidase monolayer assay (IPMA), using serum as the diagnostic specimen is widely used. The assay requires approximately 5–7 working days which allows for two passages to be completed. The main limitation of IPMA in PI testing is its nonapplicability on sera from animals under 3 months of age in which maternal antibodies can interfere with growth of the virus in cell cultures. Some adult PI cattle have been IPMA negative on serum but virus can still be isolated from the buffy coat cells.93 However, the prevalence of such animals is extremely low, and IIPMA is widely accepted as a reliable test for detecting PI cattle of 3 months or older.
The indirect immunoperoxidase staining technique is recommended for certification of BVDV-free bovine semen for artificial insemination units when the immunofluorescent test is not available.
Antigen detection
The BVDV antigen can be identified rapidly in tissue samples using immunohistochemical methods such as immunofluorescence or immunoenzyme staining in sections of frozen tissue.92,94,95 A monoclonal antibody 15C5, which reacts with the E0 protein, has been shown to react broadly with most strains of BVDV and can be used to detect BVDV antigen in formalin-fixed, paraffin-embedded tissues. This has broad diagnostic and research applications. Using these methods, the identification of BVDV antigen in fixed tissues can be used as positive laboratory confirmation of BVDV infection without positive virus isolation and is useful when investigating disease syndromes such as enteric disease, respiratory tract disease and reproductive tract disease.
Immunohistochemical (IHC) staining for BVDV in formalin-fixed, paraffin embedded skin biopsies is an effective method for the diagnosis of persistently-infected cattle.95 The technique is easy, accurate, and a less expensive antemortem diagnostic test for the detection of PI animals compared to virus isolation. It is suitable for herd screening because samples can be taken from cattle of any age, sample collection is simple, the samples are stable for transport and handling, and the test is both sensitive and specific for BVDV PI cattle. Positive IHC staining is most pronounced in the keratinocytes and in the hair follicle epithelium, hair matrix cells of the hair bulb, and the dermal papilla. IHC on skin samples is an effective method for screening neonatal calves for persistent infection.97 Skin samples from cattle with acute BVDV infection or transient infection may stain positive with IHC but the distribution of staining is confined to the epidermal keratinocytes and follicular ostia, in contrast to PI animals with antigen-positive staining cells in all layers of the epidermis. Uncertain cases should be retested a few weeks after the first test.
A monoclonal antigen-capture ELISA test is capable of rapidly and accurately detecting pestivirus-specific antigens in peripheral blood leukocytes, blood clots, and tissue samples of PI cattle.12,92,94 It has demonstrated good agreement with conventional virus isolation procedures and is suitable for routine diagnostic and certification testing. Monoclonal antibody techniques have also been used to detect the virus antigen in the central nervous system of PI cattle. Four commercially available ELISAs for the detection of BVDV antigen in the blood of PI cattle have been compared and are highly sensitive and specific and considered valuable in eradication programs when monitoring large numbers of animals.98
Herd Chek BVDV Antigen Serum Test Kit (IDEXX).
This is an enzyme immunoassay for the detection of BVDV antigen in serum, plasma and whole blood. It is based on the detection of the Ems (gp44–48) glycoprotein of the BVD virus. The Ems protein is known to be secreted in serum, plasma or whole blood samples, which can be tested without laborious sample presentation. It has the following attributes: detects types 1 and 2 isolates; designed for identification of PI animals; can be used on serum, plasma or blood; no sample preparation; rapid testing method-quick (<2 hours) and overnight protocols; ready-to-use reagents; same substrate, wash and stop solution as other IDEXX BVDV family assays; compatible with xChek software.
Polymerase chain reaction (PCR) amplification
PCR amplification of an RNA genome involves the binding of specific DNA oligonucleotides to cDNA target sequences, resulting in amplification of size-specific DNA fragments that are detectable by gel elecrophoresis.
The PCR test is able to detect small amounts of viral nucleic-acid from samples of blood and tissues including preserved material.92,94 Factors such as cost, technical expertise, equipment and automation, and RNA extraction methods are considerations in comparing with the standard methods of virus isolation. The PCR is 10–50 times more sensitive than the dot blot hybridization technique.
The reverse transcriptase-polymerase chain reaction (RT-PCR) amplification has gained widespread use as a routine diagnostic method for BVDV.99 The high analytical sensitivity of the RT-PCR allows for pooling of specimens to reduce unit cost test. Pooling is especially applicable for persistent infection testing whereby a single positive specimen can still be detected in a pool of several dozen samples.
The RT-PCR test has been used to detect the presence of the BVDV in somatic cells from bulk milk samples.100 Compared to existing methods, RT-PCR test showed 100% specificity and sensitivity in detecting PI lactating cattle in milking herds.
The reverse transcriptase-nested polymerase chain reaction (RT-nPCR) assay is a rapid and sensitive method to detect BVDV in extended semen samples.101 While the prevalence of persistent testicular infection with the BVDV is very low, use of the rapid, sensitive RT-nPCR assay on extended semen samples can be used to ensure that the virus is not transmitted in cryopreserved semen.
Serology
Serological techniques are used to detect and measure antibodies. The serum neutralization (SN) test has been the standard test to determine the occurrence of a rising BVDV titer between acute and convalescent sera. The test carried out in microtiter plates, takes 3–5 d to complete and is relatively simple to interpret. A cytopathic virus is used in order to easily detect the neutralization of the virus. Because of the antigenic differences that exist within the BVD viruses, the reported antibody titer for a specific serum sample may vary greatly between laboratories depending on the strain of virus used in the test.
Following acute infection, serum antibody is first detectable at 2–3 weeks and peak antibody levels occur 8–10 weeks later. Following successful vaccination, SN titers will be high for many months.
PI animals are seronegative, except if they have colostral antibody for the first several weeks after birth. Antibodies are usually not detectable in the sera of most cattle with mucosal disease. The specific immune tolerance of the persisting virus is also not broken by the cytopathic virus if it is antigenically similar or identical to the persisting virus and results in fatal mucosal disease. PI cattle exposed to other isolates of cytopathic viruses that do not immediately induce mucosal disease may produce highly specific serum-neutralizing antibodies.75
Precolostral serum from calves infected in utero as immunocompetent fetuses may have virus-specific neutralizing antibodies and their demonstration is meaningful for the diagnosis of past infection. A short 3-day incubation serum neutralization test is now available which is an improvement over the 5-day test.
Antibody ELISA
ELISAs are available to measure serum antibodies to BVDV and are a rapid and economical alternative to the serum neutralization test. However, because of laboratory requirements for extensive purification of antigen for the BVDV ELISA, it has not gained wide acceptance, even though ELISA titers correlate well with SN titers. The Cedist blocking ELISA for BVDV antibodies is a simple, rapid and reliable test for the detection of specific antibodies in serum, plasma, or bulk tank milk.102 Test results correlate well with virus neutralization test results and may be useful for large-scale screening and eradication programs.
Using a blocking ELISA test, the level of antibodies in bulk milk is a valuable aid to indicate the infectious status of a dairy herd and for identifying herds suspected of harboring an active infection.103 A herd with two consecutive bulk milk results four months apart of 60% (percentage inhibition) is more likely to have a very high percentage of infection. Testing of bulk tank milk for antibody using the ELISA can be used to determine the prevalence of dairy herds with antibodies, the relationships between the ELISA values in bulk milk and the location, milk yields and somatic cell counts of the herds, the annual incidence risk of new infections, and combined with the RT-PCR to detect viral RNA, to obtain an estimate of the herd prevalence of lactating persistently-infected animals.104
Use of laboratory tests in the herd
Because of the complex nature of BVDV infections, it is often difficult to obtain a definitive etiological diagnosis. The type of samples to be submitted to the laboratory and the interpretation of the results depends on clinical and herd history, and the vaccination status of the herd. The testing strategies to be used will depend on the specific disease history of the herd, the age of animals to be tested, the cost of the test, the needs of the owner of the herd, and the reasons for doing the testing.92
Acute infections
The diagnosis of acute infections must be done as early as 3 d after infection to 8–10 d after infection. A whole-blood sample is the best sample for virus isolation to identify acutely infected animals. In herd outbreaks, blood samples from normal animals should also be submitted. For serology, paired acute and convalescent samples collected 30 d apart are required to identify a four-fold increase in serum antibody titers. In abortions, the dam may have already seroconverted before the abortion and there may be no seroconversion between acute and convalescent sera. However, some aborted fetuses may be serologically positive, which confirms intrauterine infection in the later part of fetal life. If the dam is negative, BVDV can be ruled out as a cause of abortion. Calves born with congenital defects may have antibody but blood samples must be taken before the ingestion of colostrum.
Persistently infected animals
Persistently infected animals in a herd can be identified using any or a combination of the following testing procedures:
1. Collect whole blood from all animals in the herd including calves. An RT-PCR can be done on every sample which is very expensive but highly sensitive.93 The test can be done pools of whole blood. The number of samples in the pool is a function of the sensitivity of the test a particular laboratory.
2. Collect serum on all animals over 3 months of age. Test younger calves as they age or use an alternative test. With serum testing colostral antibodies may interfere with the test or eliminating detectable virus from the fluid fraction of blood for some variable period of time. Virus in mononuclear cells is unaffected by colostral antibodies. Tests that can be done on serum include the microplate virus isolation, antigen-capture ELISA, or RT-PCR.
3. Collect skin biopsies (ear notches) from all animals in the herd including calves. The tests of choice are the IHC on formalin-fixed tissues or antigen-capture ELISA using fresh samples. Use of fresh tissue samples eliminates the need for formalin.
4. For dairy herds, collect composite milk samples from lactating cows and screen remainder of herd using procedure 1 or 3 above. Somatic cells from the milk are screened for the virus by RT-PCR or virus isolation.
5. Test calves as they are born with 1 or 3. If the producer has accurate calving records, the determination that the calf is not PI automatically defines the status of the dam as not being PI. Using this protocol, ongoing surveillance is maintained with a single test defining the status of two animals. For dairy herds, bull calves must be tested as well as the heifer calves to achieve a complete herd screening program.
Before the development of the PCR test, identification of PI animals depended on virus isolation from sequential samples collected 30 d apart. By testing 30 d apart, it is also possible to test for a four-fold increase in antibody titer should the first virus isolation be the result of an acute infection. In most cases, serum is adequate for virus isolation. In young calves under 3 months of age, colostral antibody may decrease the level of free virus in the serum and may result in a false-negative test. For this reason, the use of whole blood which allows isolation of the virus from the buffy coat is recommended in calves under 3 months of age.
Most PI animals are seronegative after the colostral immunity has waned, but they may develop SN antibodies to heterologous strains of the virus.
Prenatal diagnosis of persistent infection
Pregnant dams with PI fetuses (PI carriers) have exceptionally high antibody titers.105,106 Testing pregnant dams can be used to identify and exclude PI carriers from livestock markets without completely blocking the trade with pregnant seropositive cattle. Testing is most reliable when done in late pregnancy (not before the 7th month of gestation). In dams carrying PI fetuses, the immune response was markedly higher (13 811 ± 1273U ELISA) than those in dams carrying uninfected fetuses (2542± 588U ELISA).106
The BVDV has also been detected in amniotic and/or allantoic fluid from both cattle and sheep with infected fetuses.107 A blind puncture technique for collection of fetal fluid in late pregnancy is used to collect fetal fluid. The site of collection is over the right ventral abdominal wall approximately 10 cm cranial of the udder and 10 cm medial of the flank. A nested PCR test is used to amplify the viral RNA.
Herd screening
When a diagnosis of BVDV infection has been made in a herd, for example in the case of mucosal disease in a yearling, then further investigation for the detection of infected animals at the herd level is indicated. The most common strategy for herd screening is to submit serum samples from all animals over 3 months of age and whole blood samples from calves under 3 months of age. All animals in the herd should be tested. The samples may be tested for SN antibodies and/or the presence of the virus. Virus isolation using the microtiter immunoperoxidase test is the most common method used for large numbers of samples. Calves born for the next 9 months should also be tested to detect any additional PI animals that are born which may have been infected in utero at the time of the herd infection. The goal is to insure that no additional PI animals appear and that the maternal–fetal transmission cycle is broken. In herds in which cases of mucosal disease have been recognized, most of the normal animals will have high levels of SN titers.
During the 9–12 month period of testing, management strategies should ensure that all young stock and breeding females are not in contact with infected animals. However, in some countries where vaccines are unavailable, breeding females are placed in direct contact with persistently-infected animals prior to the breeding season as a method of natural vaccination.
The serologic evaluation of unvaccinated heifers 6 to 12 months of age is an accurate method for identifying herds containing PI animals.108 Both type 1 and type 2 BVDV antibody titers should be determined to prevent misclassification. The sensitivity and specificity of serologic evaluation of five heifers for identifying these herds were 66 and 100%, respectively, in herds which contained PI cattle. Pooled-sample testing using PCR/probe testing, can be used as a herd screening test for detection of BVDV persistently infected cattle.109 However, random serologic testing of a small sample size of beef calves or cows did not satisfactorily predict the presence of a PI in a extensively managed beef cow-calf herd.110 Whole-herd screening by use of one of the methods for detecting virus or viral antigens such as IHC of skin (ear notch) specimens is required for detection and elimination of animals persistently infected with BVDV in a herd.110
NECROPSY FINDINGS
Acute mucosal disease
The gross abnormalities are usually confined to the alimentary tract.111 Characteristic shallow erosions with very little inflammation around them and with a raw, red base are present on the muzzle, in the mouth, and to a lesser extent in the pharynx, larynx and posterior nares. In the esophagus these erosions are linear in shape and lie in the direction of the folds of the esophageal mucosa. Erosive lesions may be present in the forestomachs, but are usually confined to the pillars of the rumen and the leaves of the omasum. Histologically, the lesions of the squamous mucosa of the alimentary tract begin with necrosis of individual cells and groups of cells. These foci enlarge and result in areas of necrosis with little or no inflammation of the lamina propria. If the necrotic foci are abraded, erosions and ulcers develop. In the abomasum, there is often a marked erythema of the mucosa accompanied by multiple submucosal hemorrhages and gross edema of the wall. Erosions and ulcers are common on the sides of the rugae of the abomasum and may be punctuate or more than 1 cm in diameter. The lesions have raised margins with a distinct pale halo. Histologically, there is epithelial necrosis of the deep parts of the glandular epithelium.
The mucosa of the small intestine often appears normal except for patchy or diffuse congestion and edema in some cases. In cases with a short clinical course it is common to find coagulated blood and fibrin overlying and outlining the mucosal aspect of Peyer’s patches, which are also eroded. This is a very distinctive lesion which is paralleled only by rinderpest and sometimes bovine malignant catarrh. Severely affected Peyer’s patches may be obvious through the serosa as red-black oval areas up to 10–12 cm long on the antimesenteric border of the intestine. In the large intestine the mucosa may be congested, often in a ‘tiger stripe’ pattern following colonic folds. The characteristic microscopic lesion in the intestinal mucosa is destruction of the epithelial lining of the crypts of Lieberkuhn. In Peyer’s patches, there is lysis of the follicular lymphoid tissue, collapse of the lamina propria and often consequent downgrowth of cryptal epithelium. A less frequently noted microscopic finding is a vasculitis with fibrinoid necrosis of the media; this change may also be observed in a variety of other organs.
Non-alimentary tract lesions which may be seen on occasion include ulceration of the muzzle, interdigitial skin and conjunctival membranes. Growth arrest lines in the long bones may be seen and secondary bacterial bronchopneumonia can also occur.14
Peracute BVD
The lesions of this form of the infection are similar to acute mucosal disease and it is usually not possible to differentiate between the two forms based on gross and histopathological findings. There may be an absence of gross enteric lesions, especially in animals which die within 24 h after the onset of clinical signs and in calves less than 6 months of age.6 In these peracute cases, pneumonia may be the most obvious lesion. Cases in which there is widespread hemorrhage attributable to thrombocytopenia may also be a form of peracute infection. Experimental infections with non-cytopathic type 2 strains have resulted in viral infection and necrosis of marrow precursor cells, especially megakaryocytes, as well as peripheral thrombocytopenia and leucopenia.57
Chronic mucosal disease
The necrotic epithelium may not be eroded by alimentary movements but instead remain in situ as slightly elevated, yellow, friable plaques, especially on the tongue and in the rumen. Subacute cases with a very prolonged course may show very few gross lesions in the mouth, some in the esophagus and no lesions in the stomachs and intestines. Peyer’s patches may be difficult to locate in these animals and when examined histologically the lymphoid follicles are hypocellular.
In naturally occurring mucosal disease, non-cytopathic and cytopathic viruses can be found in the spleen, intestines and esophagus. Viral antigens are also detectable in mucosal cells of the nares, rumen, abomasum, gallbladder and salivary glands.33 In PI animals, viral antigen can be found in myenteric ganglion cells, cells within crypts, mononuclear cells of gut-associated lymphoid tissue, and mononuclear cells of mesenteric lymph nodes. Viral antigen can also be found in adrenocortical cells, cerebral neurons, endocrine cells of the pituitary gland, thyroid follicles, and pancreatic islets.
The virus can be demonstrated in sections from formalin-fixed, paraffin-embedded tissue using various immunohistochemical techniques, including a method utilizing a monoclonal antibody96 and the detection of viral antigen in formalin-fixed sections of skin collected at post mortem remains strongly indicative persistent infection. Such IHC techniques have also enabled the demonstration of viral antigen in association with specific lesions, such as within the Purkinje fibers and conduction system of the myocardium of a 4-month-old calf,112 pancreatic islet cells of diabetic cattle81 and various cells of the central nervous system in a heifer with meningoencephalitis.60 This virus is also recognized as a cause of myocarditis113 which may include a mild lymphoplasmacytic myocardial arteritis with or without fibrinoid necrosis. It must be remembered that the demonstration of BVDV antigen, or the isolation of the virus from necropsy material, does not mean that the animal suffered from mucosal disease or the peracute form of infection unless supportive lesions are observed. The virus is often found in animals dying as a consequence of other disease processes, such as pneumonia.66,114 Confirmation of the presence of the virus is nevertheless significant. In terms of the individual animal, the virus may have caused a degree of immunosuppression. For the herd, the presence of circulating virus has important implications for the animals of breeding age.
Abortion
The pathological criteria for the diagnosis of BVDV as a cause of abortion have not been established.85 Finding antibody in a fetus, as in an unsuckled neonate, indicates that intrauterine infection has occurred but its diagnostic significance in regard to the abortion is not clear. The recovery of virus from the fetus, or demonstration of viral antigen within fetal tissues is only suggestive of a diagnosis of pestiviral abortion. Recognized BVDV-associated congenital defects in calves, including cerebellar hypoplasia, cataracts, retinal degeneration and dysplasia, hypoplasia and neuritis of the optic nerves, and musculoskeletal deformities52 are clear-cut indicators of compromised fetal health. However, microscopic lesions associated with BVD abortion have been described in fetal eyelid, lung and myocardium yet at the present time their diagnostic value is still controversial.87 Growth arrest lines are sometimes noted in the long bones of aborted fetuses infected with the BVDV and in utero the infection may also produce osteopetrosis. Osteopetrotic lesions, as well as anemia, thrombocytopenia and marrow necrosis have been described in 2-month-old beef calves infected with a non-cytopathic strain of BVDV.115 Infection of megakaryocytes with non-cytopathic strains of BVDV has been confirmed experimentally.116 Immunohistochemical analysis of cryostat sections of brain, skin, thyroid gland, abomasum and placenta is a rapid, sensitive method for detecting pestiviruses in bovine and ovine fetuses.117 However, in most bovine fetuses, immunohistochemical testing of formalin-fixed, paraffin-embedded tissues is recommended, as the detection of BVDV antigen in formalin-fixed fetal tissues appears to be superior to traditional virus isolation techniques and fluorescent antibody techniques.107
Samples for confirmation of diagnosis
• Histology – formalin-fixed oral/esophageal lesions, thymus, Peyer’s patches, colon, abomasum, rumen, mesenteric lymph node, heart, ear. Abortions – eyelid, lung, thymus, spleen, intestine, liver, kidney, heart, brain, eye (LM, IHC)
• Virology – thymus, thyroid, Peyer’s patch, spleen, lung, mesenteric lymph node (ISO, FAT, PCR).
The differential diagnosis of diseases associated with BVDV infection must be considered according to the many different subclinical and clinical forms of the disease affecting many body systems. Each manifestation of clinical disease must be differentiated clinically and pathologically from similar diseases. The distinguishing features of each manifestation and the diseases they resemble are summarized here.
Mucosal disease.
The differentiation of the diseases causing erosive lesions of the oral cavity mucosa can be perplexing both clinically and at necropsy. The similarity between them is the more important because rinderpest and foot-and-mouth disease are major plague diseases. The situation is so dangerous that if there is any doubt as to the identity of the disease under examination, samples should be submitted for laboratory examination.
There are many diseases of the alimentary tract of cattle which can be grouped according to the presence or absence of oral lesions with or without diarrhea. These have been summarized in Table 21.4. An erosive stomatitis and gastroenteritis are characteristic of rinderpest, bovine virus diarrhea and bovine malignant catarrh. The stomatitis and hyperemia are remarkably severe in bovine malignant catarrh along with a corneoscleral opacity, lymph node enlargement, hematuria and terminal encephalitis. Rinderpest is characterized by a high morbidity and mortality and knowledge of the disease in the area.
Table 21.4 Differential diagnosis of diseases of cattle in which there are either oral lesions or diarrhea alone or together in the same animal


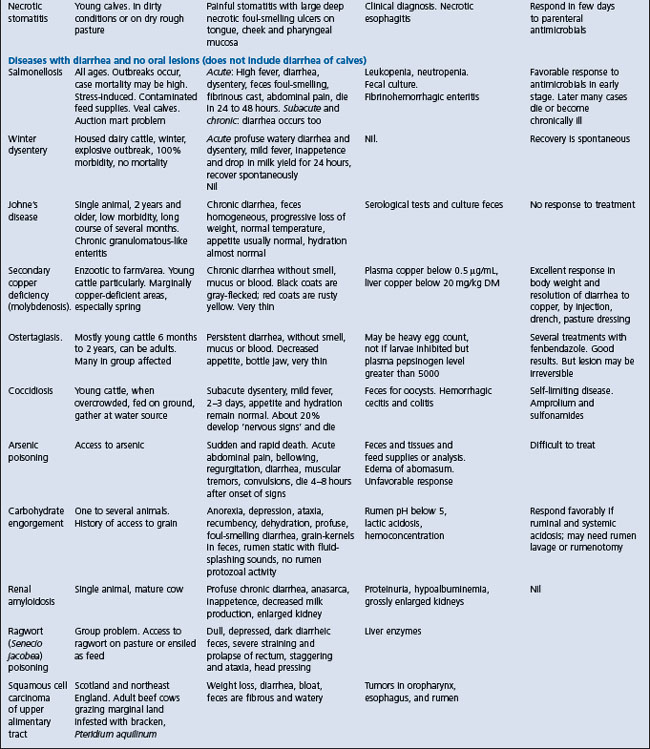
The vesicular diseases, foot and mouth disease and vesicular stomatitis, are characterized by the presence of vesicles on the tongue and buccal mucosa, teats and coronets and should be distinguishable from erosions.
Diseases causing diarrhea with no oral lesions include winter dysentery, salmonellosis, Johne’s disease, molybdenum poisoning (conditioned copper deficiency), parasitism (ostertagiasis), and arsenic poisoning.
A definitive diagnosis depends on isolation of BVDV from the buffy coat or serum of blood and other tissues. Calves with congenital defects can be provisionally identified as bovine virus diarrhea by detection of specific antibodies in calves which have not sucked; this is not an easy specimen to obtain in beef cattle running at pasture.
Although bovine virus diarrhea is not a disease of the respiratory tract it is not uncommon for respiratory signs to be evident and confusion in diagnosis between it and infectious bovine rhinotracheitis (IBR), and even pneumonic pasteurellosis, does arise. It is necessary to depend on a careful clinical examination of oral and nasal mucosae to ensure that there are no mucosal lesions. It is also necessary to include bovine virus diarrhea in the list of diagnostic possibilities when considering the causes of abortion and stillbirth in cattle. Immunoglobulin determinations in aborted fetuses may be of diagnostic value.
The definitive diagnosis of chronic mucosal disease presents problems because often the affected animal has no specific neutralizing antibody because of immunosuppression or the inability to secrete antibody. A presumptive diagnosis can be made on the basis of the clinical characteristics of the acute disease, the absence of other lesions to account for the chronic form of the disease and the presence of pancytopenia. Virus isolation must be attempted along with detailed pathological examination.
• Inapparent subclinical BVDV infection. Common diseases include acute undifferentiated fever, acute undifferentiated bovine respiratory disease
• Peracute bovine virus diarrhea. Malignant catarrhal fever. Acute salmonellosis
• Respiratory disease. All common causes of bovine respiratory disease. See Table 18.5
• Thrombocytopenia and hemorrhagic disease. Malignant catarrhal fever. Moldy sweet clover poisoning
• Unthrifty persistently infected calves. General malnutrition. Copper deficiency. Chronic pneumonia
• Reproductive failure. Common causes of reproductive failure in dairy and beef cattle herds characterized by anestrus, failure to breed, unsatisfactory semen, failure of fertilization, embryonic mortality, fetal resorption, fetal mummification, abortion, stillbirth, and perinatal mortality
• Neonatal calf diarrhea. All common causes of acute undifferentiated diarrhea of calves under 30 days of age.
• Congenital defects of calves. All inherited defects of the nervous system of calves manifested clinically at birth, and diseases of uncertain etiology characterized by nervous system involvement
TREATMENT
There is no specific treatment for any of the diseases associated with the BVDV.
The prognosis for severe cases of mucosal disease with profuse watery diarrhea and marked oral lesions is unfavorable and slaughter for salvage or euthanasia should be considered. Animals with chronic BVD should be culled and destroyed.
CONTROL AND PREVENTION
The continued presence of the BVDV in the bovine population is not due to the lack of quality diagnostic tests. BVDV continues to cause significant economic losses because of failures in implementing a sound immunization program, failures in establishing herd-monitoring programs, and failures in developing effective biosecurity and biocontainment programs.
The ultimate goal of BVDV prevention and control measures is to eliminate the potential for the birth of calves persistently infected with the virus.118 ‘Persistence is the key’.
A combination of biosecurity, vaccination, and biocontainment strategies are necessary to control and prevent BVDV infection and its consequences in a herd and country.119-121
Biosecurity is the action taken to prevent the introduction of a disease agent into a herd or region. The goal of a BVDV biosecurity program is to prevent the introduction of the virus into the cattle herd and preventing transmission of the virus to susceptible animals. Biocontainment includes the strategies to control an already existing disease in a herd or region. The goal of biocontainment is to minimize the occurrence or severity of disease associated with BVDV infection, or to completely eliminate the virus from the herd. Biocontainment includes actions to increase host immunity, remove PI cattle from the herd, and prevent effective contact between BVDV-infected and BVDV-susceptible animals. The most important subpopulation to protect from exposure it pregnant cattle, especially those in early gestation. The herd must be protected from direct exposure to cattle from other herds that may be BVDV transiently infected or persistently infected. Examples of these exposures include fenceline contact, movement to and from fairs and exhibitions, and new herd additions. Quarantine of new additions for 2 to 3 weeks after arrival prevents exposure of the native herd to unknown infected animals. Each addition must be tested for BVDV PI while in quarantine or before arrival so that these primary reservoirs of virus can be removed before they are commingled with the native herd. New additions that arrive pregnant should not calve in the presence of pregnant cattle from the native herd. The calves born to pregnant new additions must be isolated from the native herd until their BVDV status can be determined.
Beef feedlots and heifer rearing operations present a special biosecurity challenge because the opportunity to introduce BVDV PI animals into these systems is increased by the frequent introduction of cattle usually co-mingled from multiple sources. The introduction of PI cattle may affect the health and performance of pen-mates and dairy and beef heifers exposed to BVDV during gestation at a heifer development facility may later give birth to PI calves in destination herds. BVDV exposure could be minimized in these facilities by testing all new arrivals and removing PI cattle during a quarantine period of 2 to 3 weeks and before entering into the primary facilities.
Elimination of BVDV PI cattle early in the production system, such as at the cow-calf herd level, benefit the cattle industry at subsequent points, such as at the feedlot and heifer development enterprises. Ideally, procurement of animals from biosecure herds and animals previously tested negative for BVDV PI would eliminate the risk for virus exposure from PI animals in these types of operations65,122 (Figs 21.3-21.5).
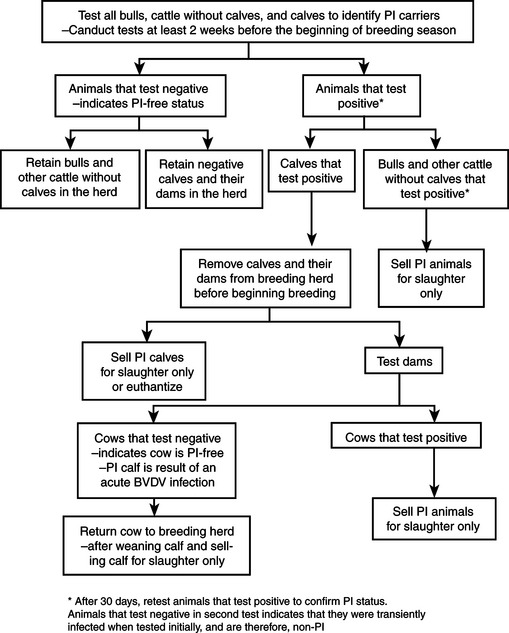
Fig. 21.4 Flow chart for testing a beef herd pre-breeding to detect and eliminate BVDV carrier cattle.
In response to significant biologic and economic loss due to the bovine virus diarrhea complex, the Academy of Veterinary Consultants drafted and approved a position statement in 2001, The position states: ‘The beef and dairy industries suffer loss due to effects of the bovine viral diarrhea virus infection. The highly mutable nature of the BVDV and the emergence of highly virulent strains of the BVDV contribute to limited success of present control programs. Also, persistently infected cattle are the primary source of infection and effective testing procedures are available to identify those infected carriers.65 Therefore, it is the resolve of the Academy of Veterinary Consultants that the beef and dairy industries adopt measures to control and target eventual eradication of BVDV from North America.’ The ‘BVD Decision/Management Guidelines for Beef Cattle Veterinarians’ are available65 and were adopted July 2003.
The successful control and prevention of the bovine virus diarrhea–mucosal disease complex depends on:
• Identification and elimination of PI animals from the herd
• Prevention of introduction of infection into the herd (biosecurity)
Identification and elimination of PI animals from the herd
Identification and elimination of PI cattle is an essential component of a control program in an infected herd.119 Elimination of such animals, also known as ‘clearance of infection’ will result in the improved health of the herd. The testing procedures to detect PI animals are described under Clinical pathology.
In beef herds, to prevent contact with pregnant cows, PI animals should be identified and removed prior to the start of the breeding season. All calves, all replacement heifers, all bulls, and all non-pregnant dams without calves must be tested for PI status. Any female pregnant at the time the herd is tested should be isolated from the breeding herd and kept isolated until her calf is tested and found to be negative. In most whole herd testing situations, IHC testing of skin samples is the test of choice because it can be accurately performed on animals of any age, and a single sample is all that is usually required.
Herd monitoring for PI animals can be done with pooled whole blood or serum samples for PCR testing. By pooling samples, the expense of screening herds with a low prevalence of PI animals is minimized. The PCR test is ideal for pooled sample testing for PI animals because it is sensitive enough to detect minute amounts of virus. A single PI animal can be detected in pools of 200 to 250 negative samples. If the initial pool is PCR-positive, it must be split and retested to differentiate viremic and non-viremic animals. Once the viremic animals are identified, they must be classified as transiently infected or PI with either a subsequent PCR, virus isolation, or IMPA test in three weeks, or using the IHC testing of a skin sample.122 Using a two-test strategy to screen feeder calves with a PCR assay of pooled samples and the second of immunohistochemical testing only of those animals represented in pooled samples with positive assay, will reduce the cost of screening incoming feedlot cattle, compared with immunohistochemical testing of all animals.123
Following the successful detection and removal of PI animals, ‘self-clearance’ or elimination of all evidence of the infection from the herd will occur. Transient infections which occur in non-pregnant animals are inefficient in transmitting the virus. The main route of transmission within a herd is from PI animals to susceptible animals. The virus is commonly maintained in a herd when seronegative animals in early pregnancy are exposed to PI animals. Self-clearance is also more likely in small herds compared to large herds which commonly have rearing conditions which increase the risk of exposure of PI animals to susceptible seronegative animals in early pregnancy.
Prevention of introduction of infection into herd (biosecurity)
After identification and elimination of the PI animals, the new virus-free status of the herd should be maintained by a program of testing of all introduced animals for freedom from infection. In many cases introductions can be guaranteed, as far as is reasonably possible, to be free of infection by selecting animals which have convincing titers of serum antibody or are negative and are derived from a totally negative herd or stable subherd. According to the period over which the herd of origin has been established and has been free from introductions, its free status may be established by testing an adequate sample of animals. In other cases, antibody-negative introductions should be examined for virus or held for a period of on-property quarantine in close contact with a few serologically negative test animals which are subsequently examined for antibody.
Significant reproductive wastage due to BVDV infection can be prevented by the testing of introductions to the herd or management of the herd to maximize immunity prior to breeding. Cattle producers purchasing pregnant heifers to expand their herds must be aware of the possibility their fetuses may already be PI. At that stage there is no simple test which will identify those heifers which are pregnant with a PI fetus. Calves from these purchased heifers of unknown vaccination history should be considered infected until proven otherwise.
Artificial insemination units are now adopting comprehensive testing programs to identify PI bulls and immunocompetent bulls with the transient acute BVDV infection. PI bulls are detected by virus isolation from blood and not by serological testing. The semen of PI bulls will usually contain the virus but the quality of the semen will not necessarily be abnormal. This emphasizes the need for virological surveillance of breeding herds, and artificial insemination and embryo transfer centers. It is also important to prevent contamination by this virus of the fluids used for recovery, in vitro manipulation and transfer of bovine embryos.
Immunization programs and biocontainment
Infectious disease models demonstrate that after BVDV is eliminated, the cattle become increasingly susceptible to new infections and the possibility increases of an outbreak with severe clinical signs following a new BVDV exposure. Thus, in the absence of strict biosecurity, recurring patterns of reinfection with severe clinical signs are expected every few years following actions which eliminate the virus. In North America and other regions where BVDV is endemic and reexposure is likely, it remains prudent to continue vaccination after eliminating the virus from the herd.119 Considering the high prevalence of BVDV infection that causes high economic losses, vaccination of cattle herds is certainly indicated, provided efficacious and safe vaccines are available. The literature on vaccination of cattle against bovine virus diarrhea124 and the evolution of the BVDV vaccines have been reviewed.120
To be effective, vaccination against BVDV infection should protect against viremia, block infection of target cells of the reproductive and lymphatic systems to avoid occurrence of fetal infection and immunosuppression, respectively.120 Antibodies present in the systemic circulation effectively neutralized viral infectivity, promote clearance of the virus, and prevent seeding of target organs such as the fetus. The goal of immunization is to stimulate both the B and T-cell arms of the immune systems. The B-cell arm of the immune response has the major responsibility for inactivating free virus. This is achieved primarily by immunoglobulin, which neutralizes the BVDV infectivity and secondarily aggregates BVDV and enhances clearance. Cell-mediated immunity, particularly CD4+ cells, which is type-2 like is important for the resolution of acute infection with noncytopathic BVDV.
An important strategy for successful control is vaccination of the breeding female at least several weeks before breeding.119 The vaccination program must be vigilant. Experimental exposure of pubertal heifers to the virus 6 weeks before breeding stimulates the production of SN antibodies which protects against transplacental infection of the fetuses when the pregnant dams are challenged with homologous virus at 100 d of gestation. A high incidence of fetal death and intrauterine growth retardation occurs in the non-immune dams which are challenged with the virus. Thus the presence of maternal immunity protects the fetus from infection. These observations provide justification for the use of BVDV vaccines in females before breeding in an attempt to stimulate maternal immunity to provide protection for the fetus. However, immunization in terms of protecting the fetus may not be effective against strains which are different from that contained in the vaccine and the ultimate precaution is to prevent cows or heifers from making new contacts shortly before or during the first half of pregnancy. It should also be emphasized that control of the infection, and of mucosal disease, depends entirely on control among the breeding stock. Infection among non-breeders is of no long-term consequence, except insofar as they may be a source of infection to breeders and compromise the continuing freedom from infection of that group.
The emphasis must be on the vaccination of immunocompetent animals which do not have persistent viral infection. This should provide at least partial, if not complete, protection against fetal infection, abortion, stillbirth, intrauterine growth retardation, congenital defects and persistent viral infection of the newborn calf. The aim of a vaccination program is therefore to ensure that all breeding females have antibodies to the virus before they become pregnant. It is important to emphasize that vaccination should be done at least 3 weeks before breeding so that the breeding females become seropositive to the virus before conception. This is necessary regardless of the type of vaccine used. The suppliers of inactivated virus vaccines commonly promote their vaccines on the basis that they can be given safely to pregnant cows. While it is true that the inactivated virus vaccines are not fetopathogenic, only successful vaccination before conception will protect the fetus from natural infection for the entire gestational period.
BVDV vaccines
Both modified live-virus (MLV) and inactivated-virus vaccines are available. Currently there are many BVDV vaccines which are federally licensed in North America alone and all meet or exceed requirements for purity, potency and safety. These requirements ensure that vaccines elicit an immune response, are free from extraneous agents and do not induce disease.
The important variables to consider when selecting a vaccine for use in different production systems include: immune response; cross reactivity; fetal protection; duration of immunity; immunosuppression; reversion to virulence; effect of maternal antibody on immune responses, and purity.
The MLV vaccines usually contain a single strain of attenuated cytopathic BVDV. The strains most commonly are BVDV–NADL, BVDV–Singer and BVDV–C24C. Several inactivated vaccines contain both cytopathic and non-cytopathic strains of the virus.
MLV–BVDV vaccines
MLV–BVDV vaccines are attenuated so that replication of the virus is restricted, reducing both viral virulence and shedding of the vaccine virus by the vaccinate.120 The advantages of MLV-BVDV vaccines are numerous and significant. Because antigen is amplified by replication in the animal, only small numbers of viral particles are necessary and thus the vaccines are inexpensive and only a single vaccination is necessary for adequate immunization. Within 3 weeks after vaccination, antibody is detectable which will neutralize an antigenically wide range of BVDV strains. It is suggested that the duration of antibody following vaccination is similar to that following natural infection which persists for more than 1 year and for several years in most cattle. However, in some cattle, detectable levels of neutralizing antibody which are distinct from the vaccine virus wane within 2 years. Immunization of calves with MLV vaccine is not inhibited by colostral antibody at titers up to 1:32. Assuming both a half-life of 21 d for colostral antibody and an initial viral neutralizing titer of 1:2000 after ingestion of colostrum, immunization should be successful in most calves that are 4–6 months of age. Revaccination before the first breeding is recommended.
The disadvantages of MLV vaccines include failure of immunization if the vaccine is not stored or handled properly. MLV vaccines may cause disease if the vaccine virus regains virulence. Adventitious agents may also be present, which are capable of causing disease but these are rare. Outbreaks of mucosal disease sometimes occurred about 10–14 d following vaccination. The possible causes postulated for these so-called vaccination breaks included the following:
1. The vaccine virus may not have been sufficiently attenuated and actually caused the disease.
2. Calves may have been incubating the disease when vaccinated.
3. Some calves were immunologically tolerant because of infection during fetal life, allowing the vaccine virus to cause the disease. These vaccination breaks gave modified live vaccines a poor reputation and as a result they have not been used on a regular basis. Also, veterinarians began to make regular reports that the vaccine was ineffective against mucosal disease, but the reasons were unknown.
4. MLV vaccines are also potentially fetopathogenic and should not be used in pregnant cows. The possible effects of such vaccination are variable and dependent on the stage of gestation when the vaccination occurs. The vaccination of pregnant cattle, without detectable neutralizing antibodies to the virus, between 51 and 190 d of gestation with a commercial modified BVDV live vaccine can result in transplacental transmission of the vaccine virus and is not recommended. Abortions, congenital abnormalities of the nervous and musculoskeletal systems, perinatal deaths, growth retardation, persistent viral infection and the late onset of mucosal disease are all possible outcomes of vaccinating pregnant cattle with a MLV before 120 d of gestation. Between 120 and 190 d of gestation, the fetus can be expected to become immuno-competent and produce serum-neutralizing antibodies which can be detected in the precolostral serum of the calf at birth. The vaccination of pregnant cattle without neutralizing antibodies to the virus between 190 and 265 d of gestation will also result in transplacental transmission of the virus and the presence of neutralizing antibodies in the precolostral serum of calves at birth. Calves derived from dams vaccinated between 90 and 118 d of gestation may exhibit ataxia, torticollis, opisthotonos and/or growth retardation.
A temperature-sensitive BVDV vaccine will cause seroconversion, produces no clinical signs of disease or leukopenia and, when used experimentally in pregnant cows, does not result in fetal infection as evidenced by lack of virus isolation and absence of precolostral antibodies in the calves which are born healthy.
Immunosuppression and genetic recombination are other potential risks associated with MLV vaccines. Vaccine strains retain some immunosuppressive properties which may predispose animals to the development of other infectious diseases.
Inactivated BVDV vaccines
The disadvantages of the MLV vaccines stimulated the development of inactivated-virus vaccines.
The advantages of inactivated BVDV vaccines include:
The disadvantages of inactivated BVDV vaccines include:
Adverse reactions at the site of vaccination may occur and are associated with the adjuvant in the vaccine. Maternal antibody may interfere with inactivated vaccines and calves may need to be revaccinated periodically from 6 months of age to just before breeding.
Combination vaccines
BVDV vaccines are often incorporated in multivalent vaccines to prevent respiratory diseases of cattle. These vaccines include combinations of the live and inactivated antigens of bovine herpes virus-1, parainfluenza-3 virus, bovine respiratory disease virus, and Mannheimia haemolytica, and Histophilus somni for administration all at the same vaccination time.
Immune response to BVDV vaccines
Efficacy of BVDV vaccines
It is not possible, on the basis of scientific information available, to recommend the use of one form of the vaccine over another. The lack of comparative efficacy data on different vaccines makes it difficult for veterinarians to recommend which vaccines to use. Ideally, the selection of vaccines should be based on efficacy data from controlled field trials. To be effective, vaccination against BVDV infection should protect against viremia to prevent dissemination of virus throughout the host following infection, to block infection of target cells of the reproductive and lymphatic systems to avoid fetal infection, and to prevent immunosuppression.
The efficacy of BVDV vaccines for the prevention of fetal infection has been uncertain. Most BVDV vaccines have not been tested for efficacy against protection of infection of the fetus. The criteria used for the evaluation of BVDV vaccines have been assessment of the magnitude of the serological response, safety and purity following vaccination of young calves 4–6 months of age. There are a limited number of studies available which have critically evaluated the efficacy of BVDV vaccines to protect the fetus.
A single dose of a modified live NADL-derived BVDV type 1 vaccine will confer protection to dams and their fetuses against challenge exposure to heterologous BVDV type 1 virus.124 A single dose of a modified live type 1 isolate of BVDV vaccine protects young calves vaccinated at 10–14 d of age and experimentally challenged intranasally 21 d later with a virulent type 2 isolate of BVDV.31
An inactivated vaccine has been evaluated to protect the fetus against experimental challenge.126 Heifers were vaccinated two or three times before breeding and challenged when between 25 and 80 d of pregnancy. There was no evidence of a viremia in the vaccinated heifers after challenge, or of infection of the fetuses. The challenge strain of the virus was fetopathogenic in control unvaccinated animals. The inactivated vaccine also protected calves against experimental respiratory challenge with the same live strain of the virus as evaluated by an absence of nasal shedding, viremia and leukopenia in vaccinated calves.127 Because of the high costs of evaluating BVDV vaccines for the protection of the bovine fetus, sheep can be used in initial BVDV vaccination-challenge experiments to evaluate vaccine efficacy, to determine optimal composition of the vaccine, protective dose, method of immunization and duration of protection.128 A EU licensed inactivated BVDV vaccine provided fetal protection when heifers vaccinated 6 months previously were challenged at about 87 days of gestation by close contact with three persistently infected carrier heifers.129 However, in another study heifers vaccinated with two inactivated vaccines were challenged at 82 days of gestation with three different Dutch field strains and all calves born from unvaccinated control animals were persistently infected. Calves born from dams vaccinated with two different inactivated BVDV vaccines were persistently infected in 78% and 60%, respectively.130
Vaccination of heifers with a non-cytopathic type 1 MLV BVDV vaccine prior to breeding protected fetuses from infection with a heterologous type 1 BVDV challenge at 55 to 100 days of gestation.131 A MLV vaccine containing types 1 and 2 BVDV provided fetal protection in 91% of heifers challenged with type 1 virus and 100% heifers challenged with type 2 virus at 60 to 90 days of gestation.132 A commercial MLV combination vaccine containing type 1 and type 2 BVDV given prebreeding protected 100% of fetuses against type 1 BVDV infection and 95% of fetuses against type 2 BVDV infection.133 In summary, there is considerable fetal protection provided by vaccines administered prior to breeding but the protection is not 100%.
Postnatal protection is necessary to provide immunity against the forms of BVDV infection which occur after birth including subclinical infection, acute infections, peracute bovine virus diarrhea, and immunosuppression associated with respiratory disease and other infections predisposed by the BVDV.
The pathogenesis of mucosal disease in PI animals explains why vaccination of calves at about 6 months of age may not provide protection in all cases. If mucosal disease is a late sequel to fetal infection, then some calves will be PI, specifically immunotolerant, and may eventually develop mucosal disease whether they respond to the vaccine virus or not. Thus they could become infected with clinical disease regardless of vaccination. Also, new cases of mucosal disease may occur in 5–10% of calves within a few weeks following vaccination with a live-virus vaccine. It has been postulated that this is likely to be due to the vaccine virus fulfilling the role of a superinfecting virus to precipitate clinical disease. However, it is a relatively infrequent occurrence and it did not occur on one occasion when known persistently viremic cattle were experimentally vaccinated with a cytopathic live-virus vaccine. Clinical disease was subsequently induced following experimental inoculation of persistently infected animals with a different cytopathic strain.
Cross-reactivity of vaccines
While there is considerable antigenic similarity between the biotypes of the virus and among isolates of either biotype, there is also antigenic diversity among the isolates and neutralizing antibodies induced by vaccination might not react with certain isolates of the virus. There are significant antigenic differences between certain strains of the virus. Vaccination of yearling cattle with either of two commercially available monovalent modified live BVD vaccines stimulated the production of SN antibodies to each of 10 cytopathic and 10 non-cytopathic isolates of the virus by one or more of the animals by 14 d after vaccination. No animal produced detectable SN antibodies to all 20 viruses.
The use of three different inactivated vaccines in cows stimulated the production of SN antibodies in all animals to each of 10 non-cytopathic and 10 cytopathic isolates of the virus when measured 14 d after the second vaccination. The reciprocals of SN antibody titers varied from 4–256, thus both the attenuated and inactivated vaccines induced antibodies to a broad range of BVD viruses.
The administration of a single dose of a MLV-BVDV vaccine to seronegative cattle, 3–8 years of age, induced an antibody response detectable for 18 months and the antibodies were able to cross-neutralize 12 antigenically diverse stains of the virus.134 A commercial vaccines containing two inactivated strains of Type 1 BVDV, an inactivated strain of bovine herpesvirus-1, and modified-live strains of bovine respiratory syncytial virus and parainfluenza-3 virus provided virus neutralizing antibodies against 22 BVDV isolates in vaccinated calves.135
The sera from a beef herd of 5725 cows which had been vaccinated annually for 7 years with an inactivated vaccine was tested for SN antibodies against several isolates of cytopathic and non-cytopathic viruses.136 Approximately 96% of the cattle did not have detectable antibody titers to the virus used in the vaccine but did have titers to other isolates of the virus. Non-cytopathic virus was isolated from three of 448 samples of sera which had titers of 64 or less against a particular isolate of the virus. The failure to detect antibody titers against certain isolates of the virus may have been due to antigenic diversity and natural decay of antibodies. Other possibilities include the use of excessive concentrations of virus in the neutralization test, and the failure of the inactivated vaccine to induce antibodies.
Cross protection of genotypes.
The cross-protective efficacy of BVDV vaccines according to genotypes has been examined with contradictory results. In some cases, prior exposure of cattle to type 1 virus (as either a MLV or inactivated vaccine) does not always provide protection against infection with type 2 virus.124 A type 1 vaccine provided protection against type 2 challenge.137 A single vaccination with a type 1 strain was efficacious against a type 2 strain challenge for at least 231 days. Following challenge, most vaccinates were free of febrile response and leukopenia; in contrast challenge virus was isolated from all control calves. A commercial inactivated type 1 BVDV vaccine provided significant but not complete clinical and virological protection against challenge a heterologous type 2 strain of BVDV.138 A single dose of MLV type 1 vaccine given at 10 to 14 days of age can protect susceptible calves from virulent type 2 BVDV for at least 4 months but high concentrations of BVDV-specific maternally derived antibodies can block the induction of the response.32 Properly used vaccines containing type 1 virus may reduce the incidence and severity of disease associated with type 2 virus. The recent occurrence of type 2 virus in North America and the UK warrants a review of current laboratory testing to ensure adequate procedures are in place to detect type 2 virus, or antibody to it.47
Commercially available vaccines
Most of the commercially available vaccines for the BVDV are combined with other antigens such as the IBR, PI-3 and BRS viruses. In one study, the serological responses of beef calves 6–8 months of age were compared following vaccination with eight commercial vaccines containing IBR, PI-3, BRSV, and BVDV.139 In general, the serological responses to the viruses varied among different commercial vaccines, between and within MLV and killed-virus vaccines, and routes of administration. All vaccinated calves developed higher antibody titers to the antigens than unvaccinated controls. The serological responses to the BVDV were low; only 20% of the calves had a four-fold seroconversion to the virus after two vaccinations. There are wide variations in onset of antibody responses and duration dependent on vaccine type and virus involved.140 The possibility that multiple antigen vaccines may not contain sufficient antigenic mass of individual antigens to stimulate an adequate level of each specific antibody has not been explored.
Field observations indicate that vaccine potency may vary considerably. Some lots of vaccines have failed to induce seroconversion in calves following carefully controlled vaccination. Unpublished observations by some clinicians found a wide variation in the amount of virus present in vaccines, and manufacturing processes may vary considerably resulting in destruction of live virus. Thus part of the vaccination program may necessarily include evaluation of the vaccine by SN testing before and after vaccination, and submitting a sample of the vaccine to a laboratory for PCR testing or viral isolation.
The use of a multivalent MLV vaccine containing bovine herpes virus-1, BVDV1 and BVDV2 in beef calves in close contact with pregnant control cows did not result in any shedding of the viruses over a period of 103 days following vaccination.141
Strategies for BVDV vaccination programs
The strategies for effective vaccination against BVDV infections are: prevention of fetal infection, and control and prevention of postnatal infections.
Prevention of fetal infection in dairy and beef herds.
With the present state of knowledge, a rational vaccination program to prevent fetal infection, for both beef-breeding herds and dairy herds, consists of vaccinating all calves at 4–6 months of age with an MLV vaccine. The emphasis must be on immunization of the heifers before breeding so that the virus does not reach the fetus before 120 d of gestation. All heifer replacements and cows are vaccinated 3–6 weeks before breeding with an MLV vaccine. Such vaccination of immunocompetent animals should result in a lifetime of protective immunity. However, to insure a level of herd immunity, all breeding females are revaccinated annually 3–6 weeks before breeding. All bulls are revaccinated annually.
Colostral immunity is present for up to 6 months of age in calves born from immune cows. Calves with even higher titers of colostral BVDV antibody may have an active response to vaccination but it is questionable whether this is of any useful purpose. If vaccination of the dam before conception is the vital part of the program, the vaccination of calves born from immune cows may be unnecessary until they approach breeding age.
There is currently no information available on the efficacy of the available BVDV vaccines for the control of the acute and peracute forms of the disease, including the hemorrhagic form, which have been recognized in the early 1990s and described earlier. A rational vaccination program for the control of the new BVDV infections occurring in immunocompetent animals would be similar to the earlier mentioned program in dairy and beef breeding herds. However, to date there is insufficient information based on clinical field trials or experimental challenge to make recommendations. However, in herds experiencing outbreaks of bovine virus diarrhea infection due to the highly virulent strains of the virus, it would seem rational to vaccinate all animals in the herd with the precaution that pregnant animals will have to be vaccinated with the inactivated virus vaccine.
Vaccination schedules
Strategic vaccination schedules for the various situations should emphasize induction of maximal protective responses to correspond with the stage of the production cycle when the risk and consequences of BVDV infections are greatest. This means into well-timed administration of vaccines prebreeding and preweaning to protect against reproductive losses and respiratory tract disease, respectively.120 Recommendations for vaccination schedules for beef and dairy cattle herds are outlined here.
Beef cow–calf herd
All beef heifer replacements should be vaccinated with a MLV BVDV vaccine at least three weeks before breeding. Cows should be vaccinated annually, at least three before breeding.
Beef calves should be vaccinated at least three weeks before weaning in order to have maximum protection during subsequent periods of high risk at and after weaning.
Beef feedlot
There is no indication for vaccination of feedlot cattle for mucosal disease in PI animals. In a population of feedlot cattle originating from several sources, the PI animals will likely develop mucosal disease regardless of any BVDV vaccination. However, if there is a risk of the postnatal forms of BVDV such as the peracute BVD associated with the highly virulent strains of BVDV, the thrombocytopenia, and the immunosuppressive effects of benign BVDV infection, feedlot cattle should be vaccinated on arrival with a MLV vaccine. A review of bovine respiratory disease vaccine efficacy concluded that there were no reliable reports of field trials evaluating the clinical effects of BVDV vaccines in North American feedlot cattle.64
The use of multivalent MLV viral vaccines containing IBR, PI-3, BVDV, and BRSV have been evaluated in fall-placed, auction market derived, feedlot calves in western Canada.142 Those cattle receiving the multivalent vaccine had significantly lower treatment rates than those in the univalent vaccine group. Cattle receiving the multivalent vaccine had higher carcass weights, weight gain, and average daily gain throughout the feeding period. There was a net economic advantage when the multivalent vaccine was used compared to a univalent IBR vaccine. However, it is not possible to determine which of the viral antigens of the vaccine were responsible for the advantage.
Dairy herd
Dairy heifer calves should be vaccinated at about 4 months of age, and with a booster at 5 to 6 months of age. MLV BVDV vaccines containing both type 1 and type 2 genotypes should be used.
Heifer replacements are vaccinated with a MLV BVDV vaccine about 45 days before being bred for the first time. This will boost serum neutralizing titers as much as possible to prevent fetal infection in the first 140 days of gestation. Dairy bulls are vaccinated at 8 to 12 months of age.
In situations where pneumonia in calves is associated with the BVDV, it is recommended that calves be vaccinated with an inactivated vaccine containing types 1a and 1b and type 2. Vaccination of dairy calves with a killed BVDV type 1 vaccine at 15 days of age, and MLV BVDV type 1 vaccine at 40 to 45 days of age provided an estimated overall protective effect of 48% against type 1 through 4 to 9 months of age.143 However, the type 1 vaccine did not affect transmission of type 2 virus.
Recently calved cows are vaccinated with MLV BVDV vaccine at about 30 days before breeding. This will ensure high SN titers to prevent fetal infection, reduce transmission of homologous viruses to older fetuses thus preventing some congenital infections, abortions, and stillbirths; and to stimulate high colostral antibody titers so that calves receive a large mass of BVDV antibody.
Inactivated BVDV vaccines can be used in pregnant cows when BVDV abortions are occurring in the herd. Two vaccinations, 2 to 3 weeks apart beginning at the time of pregnancy diagnosis or one month before the estimated time of abortion. Vaccines containing both type 1 and type 2 genotype are recommended. Inactivated vaccines have also been used at drying off and 3 to 4 weeks later to enhance colostral antibody titers. The strains of the virus used in inactivated vaccines tend to be different than those used in MLV vaccines, so using inactivated products and alternating manufacturers may provide greater cross protection and cross-react with as many field strains as possible.
Booster vaccination of dairy cows 35 days after calving with a MLV BVDV vaccine greatly increased the antibody response compared with saline controls and cows vaccinated with inactivated vaccines containing BVDV, IBR, BRSV, and PI-3 viral antigens.144
Vaccination of pregnant cows and heifers with a multivalent vaccine containing MLV BHV-1, BVDV, PI-3, and BRSV during all three trimesters of pregnancy is safe provided the animals have been previously vaccinated prior to breeding with the same MLV components.145
Veal calves should be vaccinated after arrival with a MLV vaccine containing types 1 and 2 genotypes.
Current vaccination practices
Surveys of livestock producers indicate that about 40% of dairy producers in the United States do not vaccinate dairy heifers against BVDV, and the proportion of improperly vaccinated herds is unknown. In the 1993 outbreaks of peracute/acute forms of BVD in the United States and Canada, initial field reports indicated that affected herds had not been vaccinated or had been vaccinated improperly. When the inactivated vaccine was used, cattle had not been given the second dose of vaccine 2–4 weeks after the primary vaccination as recommended by the manufacturers. Surveys in Pennsylvania indicate that many producers did not vaccinate all susceptible groups of cattle in the herd.146 Many producers did not administer the secondary vaccination of the inactivated vaccine. While 82% of dairy producers indicated they routinely vaccinated their herds, only 27% of the herds were found to be adequately vaccinated. A survey of vaccination practices in Saskatchewan dairy herds indicated that only 34% of dairy herds were vaccinated against BVDV.147 In addition, only 25% of producers who vaccinate follow the label directions for administering inactivated virus vaccines, and more specifically, the requirement to give two doses at the recommended interval. The three most common practices were annual vaccination (50%), vaccination prior to breeding (19.5%) and biannual vaccination (7.3%).
Producers may not vaccinate for a number of reasons:
1. They may not believe in the efficacy and cost-effectiveness of the vaccine
2. They may forget to vaccinate on a regular basis
3. They may have vaccinated only part of the herd when they thought the entire herd was vaccinated.
As dairy herds increase in size by importing animals from dispersed herds, the owners may not consider the necessity to vaccinate and significant numbers of animals in the herd are susceptible. Inadequate vaccination practices can be minimized by the veterinarian who can play an important role in clearly outlining in written form the vaccination program for individual herds. Constant surveillance of the health management strategies are necessary. Good and reliable records which keep track of vaccinations, when they were given, which animals were vaccinated, and which vaccines were used, are vital. Veterinarians must work with their clients to develop a specific vaccination and biosecurity protocol for each herd. The specific details of the vaccination program must be very clear (preferably in written form) including:
• Which vaccines are to be used
• Needles and syringes to be used
Vaccine failures may occur because of improper use and storage of the vaccine. Syringes must be not washed with water or solutions containing chemicals or ingredients which will readily kill any live virus in the vaccine.
Veterinarians providing a health management service should also follow up their recommendations to insure client compliance. Veterinarians can also assist producers in developing methods to handle livestock and purchased replacements by designing protocols for importing animals into the herd.
Immunization without vaccines
In Australia, where no BVDV vaccine is available, controlled exposure of non-pregnant heifers to a PI carrier animal for 12 d on pasture resulted in seroconversion by about 20 d.148 The subcutaneous, intranasal, or conjunctival inoculation of blood from PI animals into yearling heifers also resulted in seroconversion. Neither inoculation nor contact infection produced any clinical illness. The highest dilutions of serum at which seroconversion occurred were conjunctival undiluted; intranasal 101 and subcutaneous 105. With the subcutaneous route all heifers seroconverted at 103.
Eradication of BVDV infection without vaccination
The bovine virus diarrhea disease complex has been known since the late 1940s and early 1950s. Since about 1985, veterinarians have attempted to control the disease by culling PI animals, vaccination and certain levels of biosecurity. The diverse and vague clinical signs of the infection have made diagnosis difficult, costly and often elusive and frustrating. Several diagnostic tests have been developed to aid in diagnosis of BVDV infections, and most importantly for the detection of PI animals. Many vaccines have been developed since about 1960 which have reduced losses but not adequately enough because none of the vaccines will provide complete protection given the antigenic diversity of BVDV isolates. Anything less then absolute fetal protection by vaccines will still allow some PI animals to be present in the herd. Because of these difficulties and the high economic losses associated with BVDV, total eradication of the virus from herds of cattle and from countries has now become a reality.
A control and eradication program against BVDV without vaccination has been initiated in the Scandinavian countries with very promising results.49 The literature on eradication of BVDV from cattle herds and countries has been reviewed.121,149,150 The basic strategy, achievements, and status of ‘test and cull’ control programs implemented in Scandinavian countries and some other European countries are summarized here.
Concepts which are considered universal for developing control and eradication programs for BVDV include: (i) a herd is not infected until one or more persistent infections have been established; (ii) the high incidence of self-clearance will reduce the prevalence of BVDV infections in cattle populations even without active disease clearance, provided virus is not re-introduced; and (iii) BVDV cannot persist within a herd when contacts between PI animals and susceptible animals in early pregnancy do not occur.149 Thus, the ‘test and cull’ strategy is the major principle for effective eradication.
Before considering an eradication program in a region or country, an overall assessment of the economic importance of the BVDV disease complex should be estimated. Cost is an important factor in determining whether any measures against the infection should be initiated. Ideally, the overall cost of organized an eradication program should be administered by dairy and beef cattle associations, and animal health organizations. Diagnostic laboratories must be able to assist with the planning of sampling and providing information on the epidemiology of the infection to cattle producers in general, ensuring that known risk factors are identified and minimized.
Success of any eradication program is dependent on: making farmers aware of the improvements in animal health which can be gained from disease clearance as well as losses anticipated if disease occurs; making them realize that they themselves are responsible for the herd’s biosecurity; and, providing them with the information to do so.
The components of an organized eradication program based on test and cull include several factors including the following:121
• Population dynamics. In the region of concern, basic cattle population data such as average size of herd, production type (dairy, beef or others) and population density. Basic knowledge of the dynamics of the cattle industry such as movement patterns, restocking of breeding herds, vaccination programs, livestock markets, community pastures, and cattle exhibitions and sales
• Prevalence monitoring. A comprehensive knowledge of the prevalence of infection is necessary to identify herds with an ongoing infection with BVDV as well as those susceptible to infection
• Diagnostic tests. Reliable diagnostic tests for test and cull programs are necessary. The tests must be as sensitive and specific as possible, and they must be easy to use, reproducible, suitable for large-scale testing, and of reasonable cost
• Education. All those involved in the actual program must be fully informed with the latest information about the various aspects of the BVDV disease complex, including how the virus is transmitted, diagnostic testing and interpretation of results, and the strategies to be used
• Biosecurity. Biosecurity measures to prevent introduction of infection into virus free herds must be given high priority. This includes consideration of the possibilities of direct and indirect contact with infectious animals outside of the herd, and ensuring that all replacement animals imported into herd cleared of BVDV are kept in quarantine facilities until they (and their fetuses) are verified free of the virus. If different herds share common pastures, rules should be set out to ensure only BVDV free animals are allowed onto the pastures. Other means of reinfection are by biologic products, including semen, embryos, colostrum, vaccines, and other veterinary drugs, which should be verified free from BVDV before being used
• Logistics. The overall plans for sampling and testing should be outlined by an advisory body with access to all available data on epidemiology and laboratory capacity. On a regional basis, organization of testing, actions after initial screening of herds, as well as follow-up testing could be organized advantageously by a district veterinary officer or someone with similar experience in surveillance for notifiable animal diseases
• Animal identification. Individual animal identification with easily read ear tags or electronic identification is a strict requirement
• Legislation. Organized efforts to control BVD on a national level, requires legislation or some means of regulating free movement of potentially viremic animals. Initially, this may become a requirement of herd managers who have successfully cleared their animals of BVDV or livestock trade or breeding companies who want to promote a specific health status of their animals. At later stages, test certificates documenting freedom from BVDV issued by district veterinary officers engaged in organization of BVD control activities may evolve as mandatory documentation to allow access to livestock auctions, exhibitions, or communal grazing land.
Scandinavian countries
The Scandinavian countries, and some other regions in Europe, have successfully achieved control of BVDV without the use of vaccines, and are aiming towards complete eradication. The seroprevalences of BVDV in the cattle populations of these countries ranged from very high to low. In the early 1990s, the herd-level seroprevalence of BVDV in Denmark was 100%, 40% in Denmark, 25–40% in Norway, and 1% in Finland. No vaccines against BVDV have been licensed or used in the Scandinavian countries and thus the seroprevalence was due to natural infection.
The basic elements of the control programs in all Scandinavian countries are similar. Three different activity levels can be distinguished. The first level included screening of all cattle herds with the principal aim to identify BVDV-free herds and maintain them free. Bulk milk samples collected for milk quality monitoring or sera from a limited number of animals representing all epidemiologic groups of the herd are tested for antibodies to BVDV. Next they are scored to indicate freedom from BVDV or a more or less likely ongoing infection with BVDV. This population-wide screening is repeated annually to monitor the spread of BVDV or the effect of the control program. The second level of activity aims to identify herds with an active infection among those positive for BVDV, for example, those with one or more PI animals. By limiting the number of herds which require a full herd screening, efforts can be focused where needed and the overall cost minimized. The aim of the third level activity is to identify all PI individuals in herds with active infection. This involves an initial sampling of all cattle on the farm, plus a follow-up phase to test calves born to BVDV antibody positive dams which were pregnant during or shortly after the initial testing. After the herd clearing is completed, surveillance at level 2 serves to verify success and eventually to certify that cleared herds are free from BVDV, despite still strongly positive by level 1 antibody surveillance results.
Sweden launched its eradication program in 1993 as a voluntary scheme operated by farmers’ organizations with advisory input from veterinary virologists in diagnostic laboratories and animal health authorities. Upon joining the scheme, herd owners agreed to comply with a set of rules restricting free movement of animals of unknown BVDV status, including specific hygienic measures designed to minimize the risk of virus spread. The farmers subscribing to scheme benefited by regular screening to determine BVDV status and by further guidelines on how to identify PI animals from BVDV-positive herds. When cleared of BVDV, herds were also certified as BVDV free and thus allowed farmers to engage in livestock trade with other affiliated herds. Except for some grants from the Board of Agriculture to assist with testing in high prevalence areas, the farmers covered the expenses of the control program. The program was designed for both dairy and beef herds. In dairy cattle herds, prevalence surveillance could easily be done using antibody testing of bulk milk samples. In beef cattle herds, the ‘spot test’ strategy was used with either pooled milk from primiparous cows or sera from young animals, using the same antibody ELISA as used for bulk milk. For virus detection, a cell culture-based microplate immunoperoxidase assay was used. By 2002, 99% of the beef herds were affiliated and the percentages of certified BVD-free dairy and beef herds had increased to 93 and 88%, respectively. To address the problem of nonsubscribing herds which acted as potential sources of reinfection, the Board of Agriculture declared in 2002, that subscription to the control program was mandatory for all cattle holdings. In 1998, it was predicted that Sweden would become free of BVDV in 2002 if the rate herd clearing continued as planned. Based on the currently available figures, it might be reasonable to assume that national BVDV free status can be achieved in 2004.
Norway launched a nation wide control program in 1992 with the long term aim of eradication. Most cattle were dairy production herds. Soon after the program was begun, BVD was designated a notifiable disease by the Animal Health Authority. This included legislative power to impose movement restrictions on cattle from infected herds to limit further spread of BVD as much as possible. The diagnostic activities included annual bulk milk antibody testing of dairy herds and spot test serology of beef herds with both sample categories analyzed by ELISA. Herds with high bulk milk antibody levels were selected for further testing of milk from primiparous cows or sera from young animals. Herds with antibody positive second-level samples were considered infected, and movement restrictions were imposed. Samples negative for or with low levels of antibodies to BVDV were tested for BVDV antigen by ELISA. Calves born to antibody-positive dams in recently cleared were not sampled for antigen testing until they reached 3 months of age to avoid analytic interference of maternal antibodies.
The cost of surveillance testing at levels 1 and 2 was covered by the control scheme, whereas the cost of sampling and testing to clear individual herds for BVDV was paid for by the farmers. Remarkable progress was made and eradication is expected in 2004.
In Denmark, the high prevalence of infection, the animal density and the structure and management of cattle herds required a specific approach but using the same principles outlined above. A nation-wide control program was begun in 1994. Participation initially was voluntary but since 1996, legislation was introduced to prohibiting transportation of viremic animals. By early 1999, 9% of the dairy and 5% of the beef herds were still registered with PI status. These were attributed to reinfection of previously cleared herds, in part due to noncompliance with biosecurity recommendations on how to avoid infection. There was no legislative requirement of for owners of herds with PI status to get them cleared.
In Finland, the herd seroprevalence of BVDV infection has always been much lower at about 1%. In 1999, a voluntary control and eradication program was begun. Bulk milk samples from dairy cattle herds were tested by ELISA, and sera from beef cows tested by microneutralization. Between 1994 and 1997, the prevalence of BVD positive herds declined from 1% to 0.4% of dairy herds, and from 30% to 3.2% in beef herds. The prevalence continued to drop but remained at a low level analogous to the tailing pattern also seen in antibody prevalence in Scandinavian countries. The program was not mandatory and some farmers did not join.
In 1999, in the UK, the cattle industry established Cattle Health Certification Standards (Checs) as a non-trading organization to promote and regulate voluntary schemes for the control of BVDV and other pathogens. The basis is the identification and removal of PI animals from herds, combined with changes in husbandry procedures to prevent infection from being reintroduced. There are three programs, allowing the farmer to work with the veterinarian to formulate a BVDV health strategy to meet the particular needs of that farm. The Accreditation Program demonstrates the herd is free from BVDV, to maintain freedom from the virus, and to allow the sale of animals as accredited free of the virus. In the screening and eradication program the objective is to implement a control program to reduce the detrimental effects on the herd productivity associated with the BVDV and to allow sale of animals of known status. The program applies where there is already evidence of recent BVDV infection in the herd or where positive results have been found in the course of an accreditation program. In the Vaccination Monitored Free Program the objective is to control BVDV infection through vaccination of the breeding herd and, by regular monitoring of young animals, to demonstrate that the control is effective and exposure of young animals to the virus has not occurred. The goal is to allow the sale of animals that are accredited as being from a vaccinated herd and monitored free of active BVDV infection. This program is considered appropriate for commercial herds selling animals for finishing. The status of these herds is lower than that of BVDV Accredited Herds.
A scheme to control and eradicate BVDV was initiated in 1994 in the Shetland Islands.151 Over a three-period every bovine animal on the islands was blood-sampled and laboratory tested using Mab-based ELISAs for BVD virus antibody and antigen detection for evidence of disease. Virus positive cattle were culled from infected herds and compensation was paid as part of the control program. The pilot scheme indicated that it is possible to control and eradicate BVDV in herds in a defined geographical area. If pregnant replacement breeding stock are purchased and imported into a herd, even if they may be antibody positive and virus negative, they may be carriers of persistently-infected fetuses, and their calves when born must be tested before colostrum ingestion for persistent infection and culled if positive.
No countrywide BVDV control schemes involving entire cattle populations have been launched on the European mainland. There is some interest in control of the disease complex using the test and cull procedures in individual herds, in groups of herds supervised by animal health organizations, or even regions within certain countries. Programs have been developed in Lower Saxony in Germany152 and in Austria.153
In Austria, cattle herds are identified as infected or not by using herd-level antibody tests on bulk milk or on groups of young animals. PI animals are detected and eliminated to prevent spread of infection between animals which are commonly grazed on communal pastures.152 A modified commercially available antigen-capture ELISA, and a RT-PCR tests were used and both were confirmed as 100% when compared to the Ag-ELISA kit (Herd Chek, IDEXX).
In Lower Saxony, because of the high seroprevalence of infection due to vaccination, control and eradication will involve a combination of identification of PI animals, systematic vaccination programs, biosecurity measures to prevent the introduction of infection into BVDV free herds, followed by eventual cessation of the use of vaccines.
In response to significant biologic and economic loss due to the bovine virus diarrhea complex, the Academy of Veterinary Consultants drafted and approved a position statement in 2001, The position states: ‘The beef and dairy industries suffer loss due to effects of the bovine viral diarrhea virus infection. The highly mutable nature of the BVDV and the emergence of highly virulent strains of the BVDV contribute to limited success of present control programs. Also, persistently infected cattle are the primary source of infection and effective testing procedures are available to identify those infected carriers.65 Therefore, it is the resolve of the Academy of Veterinary Consultants that the beef and dairy industries adopt measures to control and target eventual eradication of BVDV from North America.’ The ‘BVD Decision/Management Guidelines for Beef Cattle Veterinarians’ are available65 and were adopted July 2003.
There are no plans to eradicate BVDV from North America as is being done in the Nordic Countries. However, it is very realistic and possible that the virus could be eradicated on a herd by herd basis using detection and elimination of PI animals, the judicious use of effective vaccines, regular diagnostic testing for PI animals, and implementation of biosecurity measures to ensure that re-infection of the herd of does not occur.
Donis RO. Molecular biology of bovine viral diarrhea virus and its interactions with the host. In: Epidemiology of bovine virus diarrhea. Bovine viral diarrhea virus. Vet Clin North Am Food Anim Pract. 1995;113:393.
Houe H. Epidemiological features and economical importance of bovine virus diarrhea virus (BVDV) infections. Vet Microbiol. 1999;64:89-107.
Lindberg ALE, Alenius S. Principles for eradication of bovine viral diarrhea virus (BVDV) infections in cattle populations. Vet Microbiol. 1999;64:197-222.
Sandvik T. Laboratory diagnostic investigation for bovine viral diarrhea virus infections in cattle. Vet Microbiol. 1999;64:123-134.
Hamers C, et al. Diversity among bovine pestiviruses. Vet J. 2001;161:112-122.
Van Oirschot JT. Vaccination of cattle against bovine virus diarrhea. Vet Microbiol. 1999;64:169-183.
Goens SD. The evolution of bovine viral diarrhea. Can Vet J. 2002;43:946-954.
Lindberg A. Bovine viral diarrhea virus infections and its control: A review. Vet Quart. 2003;25:1-16.
Moczygemba L. A review of the relationship between persistent infection of cattle with bovine viral diarrhea virus and feedlot morbidity and gain. Bovine Pract. 2003;37:155-161.
Peterhans E, Jungi TW, Schweizer M. BVDV and innate immunity. Biologicals. 2003;31:107-111.
Slenning BD. Bovine viral diarrhea virus testing options. Proc Am Assoc Bov Pract. 2003;36:45-51.
Brock KV. The many faces of bovine viral diarrhea virus. Vet Clin North Am Food Anim Pract. 2004;20:1-3.
Bolin SR, Grooms DL. Origination and consequences of bovine viral diarrhea virus diversity. Vet Clin North Am Food Anim Pract. 2004;20:51-68.
Brock KV. Strategies for the control and prevention of bovine viral diarrheal virus. Vet Clin North Am Food Anim Pract. 2004;20:171-180.
Brodersen BW. Immunohistochemistry used as a screening method for persistent bovine viral diarrhea virus infection. Vet Clin North Am Food Anim Pract. 2004;20:85-93.
Campbell JR. Effect of bovine viral diarrhea virus in the feedlot. Vet Clin North Am Food Anim Pract. 2004;20:39-50.
Chase CCL, Elmowalid G, Yousif AAA. The immune response to bovine viral diarrhea virus: a constantly changing picture. Vet Clin North Am Food Anim Pract. 2004;20:95-114.
Grooms DL. Reproductive consequences of infection with bovine viral diarrhea virus. Vet Clin North Am Food Anim Pract. 2004;20:205-219.
Givens MD, Waldrop JG. Bovine viral diarrhea virus in embryo and semen production systems. Vet Clin North Am Food Anim Pract. 2004;20:21-38.
Kelling CL. Evolution of bovine viral diarrhea virus vaccines. Vet Clin North Am Food Anim Pract. 2004;20:115-129.
Larson RL, et al. Bovine viral diarrhea (BVD): Review for beef cattle veterinarians. Bovine Pract. 2004;38:93-102.
Larson RL, et al. BVD decision/management guidelines for beef veterinarians. Bovine Pract. 2004;38:103-112.
Lindberg A. The Nordic bovine viral diarrhea virus eradication programs; their success and future. Cattle Pract. 2004;12:3-5.
Saliki JT, Dubovi EJ. Laboratory diagnosis of bovine viral diarrhea virus infections. Vet Clin North Am Food Anim Pract. 2004;20:69-83.
Sandvik T. Progress of control and prevention programs for bovine viral diarrhea virus in Europe. Vet Clin North Am Food Anim Pract. 2004;20:151-169.
Smith DR, Grotelueschen DM. Biosecurity and biocontainment of bovine viral diarrhea virus. Vet Clin North Am Food Anim Pract. 2004;20:131-149.
1 Goens SD. Can Vet J. 2002;43:946-954.
2 Hamers C, et al. Vet J. 2001;161:112.
3 Vilcek S, et al. Arch Virol. 2001;146:99-115.
4 Graham DA, et al. Res Vet Sci. 2001;71:127-134.
5 Ridpath JF. Biologicals. 2003;31:127-131.
6 Ridpath JF, et al. Vet Microbiol. 2000;77:145.
7 Baker JC. Vet Clin North Am Food Anim Pract. 1995;113:425.
8 Paisley LG, et al. Therigenol. 1996;46:1313.
9 Chase CCL, et al. Biologicals. 2003;31:145.
10 Houe H. Vet Microbiol. 1999;64:89-107.
11 Houe H. Vet Clin North Am Food Anim Pract. 1995;113:521.
12 Wittum TE, et al. Prev Vet Med. 2001;49:83.
13 Taylor LF, et al. Can J Vet Res. 1995;59:87.
14 Taylor LF, et al. Can Vet J. 1997;38:29.
15 Rush DM, et al. J Am Vet Med Assoc. 2001;219:1426-1431.
16 Munoz-Zanzi CA, et al. Am J Vet Res. 2003;64:358-365.
17 Evermann JF, Ridpath JE. Vet Microbiol. 2002:129.
18 Loken T. Vet Clin North Am Food Anim Pract. 1995;113:597.
19 Nielsen SS, et al. J Wildlife Dis 36:584.
20 Van Campen H, et al. J Wildlife Dis. 2000;37:306.
21 Wentz PA, et al. J Am Vet Med Assoc. 2003;223:223.
22 Welsh MD, et al. Vet Immunol Immunopathol. 1995;46:195.
23 Niskanen R, Lindberg A. Vet J. 2003;165:125-130.
24 Paton D, et al. Arch Virol. 1997;142:929.
25 Niskanen R, et al. Vet J. 2002;163:251-259.
26 Grooms DL. Vet Clin Food Anim. 2004;20:5.
27 Givens MD, et al. Am J Vet Res. 2003;64:428.
28 Voges H, et al. Vet Microbiol. 1998;61:165.
29 Lang-Ree JR, et al. Vet Rec. 1994;135:412.
30 Chase CCL, Elmowalid G, Yousif AAA. Vet Clin North Am Food Anim Pract. 2004;20:95-114.
31 Cortese VS, et al. J Am Vet Med Assoc. 1998;213:1312.
32 Ellis J, et al. J Am Vet Med Assoc. 2001;219:351.
33 Munoz-Zanzi CA, et al. J Am Vet Med Assoc. 2002;221:676-685.
34 Fredriksen B, et al. Vet Rec. 1999;144:111.
35 Graham DA, et al. Vet Rec. 2003;152:795.
36 Potgieter LND. Vet Clin North Am Food Anim Pract. 1995;113:501.
37 Bolin S. Vet Clin North Am Food Anim Pract. 1995;113:615.
38 Ridpath JF, et al. Am J Vet Res. 2003;64:65.
39 Kummer BM, et al. Vet Microbiol. 2001;77:117.
40 Bolin SR, Grooms DL. Vet Clin Food Anim. 2004;20:51-68.
41 Odeon AC, et al. Vet Microbiol. 2003;96:133.
42 Vilcek S, et al. Vet J. 1999;158:33.
43 Carman S, et al. J Vet Diagn Invest. 1998;10:27.
44 Walz PH, et al. Am J Vet Res. 2001;62:1095.
45 Kelling CL, et al. Am J Vet Res. 2003;63:1379.
46 Harding MJ, et al. Am J Vet Res. 2002;63:1458.
47 Wakeley PR, et al. Vet Microbiol. 2004;102:19.
48 Vilcek S, et al. Vet Rec. 2002;151:150.
49 Flores EF, et al. Vet Microbiol. 2000;77:175.
50 Van Leeuwen JA, et al. Can Vet J. 2001;42:193.
51 Chi J, et al. Prev Vet Med. 2002;55:137.
52 Houe H. Biologicals. 2003;31:137-143.
53 Stott AW, et al. Prev Vet Med. 2003;59:51-66.
54 Gunn GJ, Stott AW, Humphrey RW. Vet J. 2004;167:143-149.
55 Valle PS, et al. Prev Vet Med. 2001;51:17.
56 Walz PH, et al. Can J Vet Res. 2001;65:241.
57 Wood RD, et al. Can J Vet Res. 2004;68:42.
58 Hamers C, et al. Vet J. 2000;160:250.
59 Bolin SR, Ridpath JF. Am J Vet Res. 1995;56:755.
60 Blas-Machado U, et al. Vet Pathol. 2004;41:190.
61 Baule C, et al. J Clin Microbiol. 2001;39:146.
62 Fulton RW, et al. Can J Vet Res. 2000;64:151.
63 Fulton RW, et al. Can J Vet Res. 2002;66:181.
64 Campbell JR. Vet Clin North Am Food Anim Pract. 2004;20:39-50.
65 Larson RL, et al. Bovine Pract. 2004;38:103.
66 Shahriar FM, et al. Can Vet J. 2002;43:863.
67 Booker CW, et al. Can Vet J. 1999;40.
68 Loneragan GH, et al. J Am Vet Med Assoc. 2005;226:595.
69 Fray MD, et al. Vet Microbiol. 2000;77:185.
70 McGowan MR, et al. Theriogenol. 2001;59:1051.
71 Moenning V, et al. Vet Clin North Am Food Anim Pract. 1995;113:477.
72 Bielanski A, Loewen K. Anim Reprod Sci. 1994;35:183.
73 Brock KV, et al. Theriogenol. 1997;47:837.
74 Frederiksen B, et al. Vet Pathol. 1999;36:267.
75 Bolin SR. Vet Clin North Am Food Anim Pract. 1995;113:489.
76 Peterhans E, et al. Biologicals. 2003;31:107-111.
77 Larsson B, et al. Res Vet Sci. 1995;58:186.
78 Ridpath JF, Bolin SR. Virol. 1995;212:259.
79 Houe H, Heron I. Acta Vet Scand. 1993;34:305.
80 Murondoti A, et al. Vet Rec. 1999;144:1999.
81 Taniyama H, et al. J Comp Path. 1999;121:149.
82 Scherer CFC, et al. Vet Microbiol. 2001;79:285.
83 Swaspidan S, et al. Vet Pathol. 2001;38:275.
84 Monies RJ, et al. Vet Rec. 2004;155:765.
85 Franz S, Baumgartner W. Vet J. 2002;165:205.
86 Taylor LF, Rodwell BJ. Aust Vet J. 2001;79:682.
87 Rufenacht J, et al. Theriogenol. 2001;56:199.
88 Robert A, et al. Therigenol. 2004;61:117.
89 Munoz-Zanzi CA. Theriogenol. 2004;61:1085.
90 Munoz DP, et al. Br Vet J. 1996;152:175.
91 Niskanen R, et al. Prev Vet Med. 1995;23:229.
92 Saliki JT, Dubovi EJ. Vet Clin Food Anim. 2004;20:69.
93 Grooms DL, et al. J Am Vet Med Assoc. 2001;219:629.
94 Brock KV. Vet Clin North Am Food Anim Pract. 1995;113:549.
95 Brodersen BW. Vet Clin North Am Food Anim Pract. 2004;20:85-93.
96 Njaa BL, et al. J Vet Diagn Invest. 2000;12:393.
97 Grooms DL, Keilen ED. Clin Diagn Lab Immunol. 2002;9:898.
98 Brinkoff J, et al. Vet Microbiol. 1996;50:1.
99 Ridpath JF, et al. J Vet Diagn Invest. 2002;14:303.
100 Drew TW, et al. Vet Microbiol. 1999;64:145.
101 Givens MD, et al. Vet Microbiol. 2003;96:145.
102 Kramps JA, et al. Vet Microbiol. 1999;64:135.
103 Beaudeau F, et al. Vet Rec. 2001;149:236.
104 Graham DA, et al. Vet Rec. 2001;149:261.
105 Lindberg A, et al. Prev Vet Med. 2001;51:199.
106 Brownlie J, et al. Clin Diagn Virol. 1998;10:141.
107 Lindberg A, et al. Vet J. 2002;164:151.
108 Pillars RB, Grooms DL. Am J Vet Res. 2002;63:499.
109 Munoz-Zanzi CA, et al. J Vet Diagn Invest. 2000;12:195.
110 Waldner CL, Campbell JR. Am J Vet Res. 2005;66:825.
111 Bielefeldt-Ohmann H. Vet Clin North Am Food Anim Pract. 1995;113:447.
112 Piero FD, et al. Vet Rec. 1997;140:407.
113 Haines DM, et al. Can Vet J. 2004;45:231.
114 Haines DM, et al. Can Vet J. 2001;42:857.
115 Scruggs DW, et al. J Vet Diagn Invest. 1995;7:555.
116 Marshal DJ, et al. Vet Pathol. 1996;33:311.
117 Thur B, et al. Am J Vet Res. 1997;58:1371.
118 Brock KV. Vet Clin North Am Food Anim Pract. 2004;20:171.
119 Smith DR, Grotelueschen DM. Vet Clin North Am Food Anim Pract. 2004;20:131.
120 Kelling CL. Vet Clin North Am Food Anim Pract. 2004;20:115.
121 Sandvik T. Vet Clin North Am Food Anim Pract. 2004;20:151.
122 Larson RL, et al. Bovine Pract. 2004;38:93.
123 Larson RL, et al. J Am Vet Med Assoc. 2005;226:249.
124 van Oirshot JT, et al. Vet Microbiol. 1999;64:199.
125 Cortese VS, et al. Am J Vet Res. 1998;59:1409.
126 Brownlie J, et al. Vet Rec. 1995;137:58.
127 Howard CJ. Vet Microbiol. 1994;42:171.
128 Bruschke CJM, et al. Vet Microbiol. 1996;50:33.
129 Patel JR, et al. Arch Virol. 2002;147:2453.
130 Zimmer GM, et al. Vet Microbiol. 2002;89:255.
131 Dean HJ, et al. Am J Vet Res. 2003;64:530.
132 Kovacs F, et al. Vet Microbiol. 2003;96:117.
133 Fairbanks KK, et al. J Am Vet Med Assoc. 2004;225:1898.
134 Cortese VS, et al. Am J Vet Res. 1998;59:848.
135 Hamers C, et al. Vet J. 2002;163:61.
136 Bolin SR, et al. Am J Vet Res. 1991;52:1033.
137 Dean HJ, Leyh R. Vaccine. 1999;17:1117.
138 Hamers C, et al. Vet Rec. 2003;153:236.
139 Van Donkersgoed J, et al. Can Vet J. 1991;32:727.
140 Fulton RW, et al. Vaccine. 1995;13:725.
141 Kleibocker SB, et al. J Am Vet Med Assoc. 2003;222:1399.
142 Schunicht OC, et al. Can Vet J. 2003;44:43.
143 Thurmond MC, et al. J Am Vet Med Assoc. 2001;219:968.
144 Dubovi EJ, et al. Vet Ther. 2000;1:49.
145 Ellsworth MA, et al. Vet Therap. 2003;4:120.
146 Rauff Y, et al. J Am Vet Med Assoc. 1996;209:1618.
147 Carruthers TD, Petrie L. Aust Vet J. 1996;37:621.
148 Cook IG, et al. Aust Vet J. 1990;67:393.
149 Lindberg ALE, Alenius S. Vet Microbiol. 1999;64:197.
150 Lindberg ALE. Vet Quarterly. 2003;25:1.
151 Synge BA, et al. Vet Microbiol. 1999;64:223.