Chapter 108 Principles of Perforator Flaps
BASIC ANATOMY
1 Define “perforator.”
The word “perforator” is derived from the Latin per (through) and forare (to pierce or bore). Any blood vessel that passes through a defined fenestration in the deep fascia should be considered a “perforator” of the deep fascia, regardless of the origin of that perforator.
2 How do direct and indirect “perforators” differ?
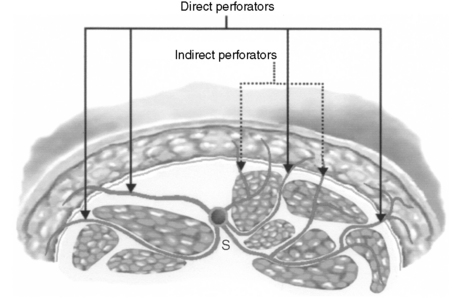
All perforators arising from the underlying source vessel to a given angiosome contribute to the latter’s “fascial plexus” either directly or indirectly. Using the terms for specific types of deep fascia perforators introduced by Nakajima et al., direct perforators (e.g., axial, septocutaneous, direct cutaneous branch of a muscular vessel) course from the source vessel to the skin without first supplying any other deep structure. Indirect perforators (e.g., musculocutaneous perforators) first will have passed via some intermediary structure (e.g., muscle) before ultimately reaching the subdermal plexus (Fig. 108-1).
3 Where is the “fascial plexus”?
The “fascial plexus” is not a discrete structure per se but represents the confluence of multiple, adjacent vascular networks and their branches that have typically emanated from any of the deep fascia “perforators,” whether direct or indirect perforators. Intercommunications between these networks exist at the subfascial, fascial, suprafascial, subcutaneous, and subdermal levels.
4 What is a “mother” vessel?
The “mother” vessel refers to the source vessel to a given angiosome, from which the perforator takes its origin.
5 What is a “perforator flap”?
Tissue receiving its vascular supply via any “perforator” of the deep fascia is considered a “perforator flap.” Because this could be by “direct” or “indirect” perforators, the corresponding flaps then would be either direct or indirect “perforator flaps.” (Further emphasis on direct perforator flaps can be found in Chapter 106.)
6 What is a “true” perforator flap?
According to Wei et al., only muscle perforator flaps are “true” perforator flaps. Only a “true” muscle perforator flap requires an intramuscular dissection of the perforator. This is considered an important point to distinguish the technical differences (and difficulties) encountered in the raising of such flaps in comparison to other forms of fasciocutaneous flaps. The muscle is also always excluded so that function is preserved (Table 108-1).
Table 108-1 Attributes and Liabilities of Perforator versus Muscle Flaps
| Perforator Flaps | Muscle Flaps | |
|---|---|---|
| Accessibility | + | − |
| Anatomic anomalies | − | + |
| Availability | + | − |
| Composite flaps | ± | + |
| Use in infected or irradiated wound | ± | + |
| Donor site morbidity | − | + |
| Dynamic transfer | − | + |
| Expendable | + | − |
| Malleability | − | + |
| Microsurgical tissue transfer | = | = |
| Reliability | = | = |
| Sensate | + | − |
| Size | + | ± |
| Thinness | ± | + |
+, Asset; −, detriment; =, no significant difference; ±, variable.
7 Could muscle perforator flaps be considered a form of fasciocutaneous flap?
Unequivocally yes! A muscle perforator flap relies on large perforating musculocutaneous branches from the source pedicle to a muscle. These are identical to the “perforating cutaneous branches of a muscular vessel,” which, according to Nakajima et al., represent one type of deep fascia perforator and is the basis of one of their corresponding types of fasciocutaneous flaps. However, this remains an extremely controversial point.
8 Name different types of indirect perforator flaps
Remember that indirect perforators first have passed along or through some intermediary structure before reaching the subdermal plexus. Musculocutaneous perforators obviously pass through muscle on their way to the deep fascia, so muscle perforator flaps are indirect perforator flaps and at the present time represent the most common clinical application of this genre.
The cutaneous sensitive nerves often pierce the deep fascia along with their accompanying circulation that has secondary cutaneous branches. Thus neurocutaneous flaps are another form of indirect perforator flap. A good example is the distal-based sural neuroadipofascial flap that today is commonly used for lower leg defects in lieu of free-tissue transfers. The medial cutaneous branch of the sural nerve is an intrinsic part of this flap. It pierces the deep fascia at the level of the triceps surae, often accompanied by the median sural artery. Sometimes the nerve can be excluded from this flap, but the lesser saphenous vein must then be retained. It too pierces the deep fascia more proximally with its own accompanying vascular system, so venous flaps may represent yet another form of indirect perforator flap. Niranjan has also shown the existence of periosteal and peritendinous indirect perforator flaps.
9 Must all perforator flaps be cutaneous flaps?
As long as the requisite perforator and its corresponding fascial plexus are maintained within the flap, the cutaneous component can be eliminated. As an adiposal or adipofascial flap, this will simplify direct donor site closure, sometimes important to eliminate the need for a skin graft.
10 Does the deep fascia have to be included with a perforator flap?
On the contrary, the deep fascia is not essential and typically is excluded from a perforator flap, an especially valuable maneuver for tension-free closure obviating the need for reinforcement of a deep inferior epigastric artery perforator (DIEAP) flap donor site. However, it can be included as a composite flap if vascularized fascia is needed at the recipient site. In addition, sometimes inclusion of the deep fascia simplifies the initial identification of the perforators at a subfascial plane.
11 In what body regions do musculocutaneous perforators practical for muscle perforator flaps predominate compared with direct perforators of the deep fascia?
Because most direct perforators of the deep fascia arise along intercompartmental or intermuscular septa, they are more prevalent wherever long, slender muscles are found, as in the extremities. In contrast, musculocutaneous perforators are more numerous over the broad, flatter muscles associated with the trunk, where muscular septa are few and far apart. However, many important musculocutaneous perforators can also be found piercing other larger muscles, especially of the lower extremity.
12 Describe the course of the perforator veins
Perforator veins usually accompany the perforator arteries at the site of penetration through the deep fascia. However, within the subcutaneous tissues they often diverge from the arteries and have an unpredictable course. Sometimes outflow from the perforator veins proceeds to nearby superficial veins rather than to the deep venous system.
13 List some methods that allow preoperative identification of perforators
Color duplex ultrasound is highly sensitive not only for identifying the site of a perforator but also for providing qualitative information such as caliber and flow characteristics. Unfortunately, it is time consuming. Multidetector-row helical computed tomograph has been used in Europe as a more rapid method. The most pragmatic method universally available, although not as precise, is use of the ubiquitous handheld audible Doppler to survey the given territory. This method also allows intraoperative localization, which may be necessary.
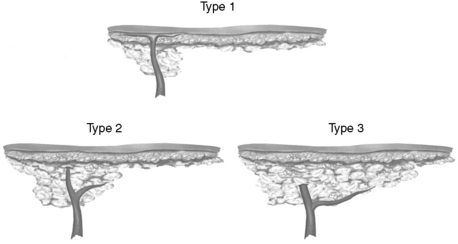
14 While thinning perforator flaps, Kimura found what three different branching patterns of musculocutaneous perforators through the subcutaneous tissues in their course to the subdermal plexus?
Type 1 perforators pass almost directly from the deep fascia to the subdermal plexus without branching. Type 2 perforators branch in the adipose tissue just before reaching the subdermal plexus, with the branches then running parallel to the flap surface. In type 3 perforators, branches follow the deep fascia for an indeterminate distance before eventually proceeding into the subcutaneous tissues (Fig. 108-2).
15 Do different donor sites have predictable suprafascial branching patterns of perforators?
An axiom in perforator flaps is that anatomic anomalies are the norm, and nothing is routine. Nevertheless, Kimura found that the lateral circumflex femoral artery-tensor fascia lata (LCFA-tfl ) and DIEA perforator flaps consistently displayed a type II pattern (see Fig. 108-2), branching just before reaching the superficial adipose layer. The LCFA-vastus lateralis (LCFA-vl, as known as the anterolateral thigh) perforator flap often had early branching or a type 3 pattern.
BASIC PHYSIOLOGY
16 When exploring the potential vessels for a perforator flap, what is the smallest size that should be chosen?
A good rule of thumb is to choose an artery that has an external diameter ≥0.5 mm or if pulsations are readily visible. This is not to say that smaller perforators cannot be chosen, but the dissection then becomes increasingly more difficult.
17 Although a single perforator could sustain an entire flap, state some good reasons to include more
Twisting of a single perforator >360 ° could go unrecognized and be disastrous because flow would be totally obstructed. This occurrence is grossly more obvious if at least two perforators are included. Inadvertent injury (it happens!) to a single perforator would likewise be disastrous. More perforators, especially if spread far apart, should logically also sustain a longer flap more safely.
18 How can the potential territory of a perforator flap be estimated?
Safe limits for the design of perforator flaps have been determined only by clinical trial and error. Deep fascia perforators are frequently anomalous in caliber and location not only among individuals but also in opposite sides of the same individual, so an exact preoperative prediction of safe flap boundaries remains impossible. Ongoing injection studies of discrete perforators can only provide estimates, as again the anatomy of each individual is different and the dynamic territory for each perforator cannot be known beforehand.
19 What is the point of rotation of a perforator flap?
Especially applicable when used as a local flap, this corresponds to the site where the flap is tethered by its vascular pedicle.
20 Describe the arc of rotation of a local perforator flap
The distance from the point of rotation to the most distant safe length of the given flap will determine the range of coverage or arc of rotation through which a local perforator flap can be transposed.
21 How can the arc of rotation of a local perforator flap be increased?
Further dissection of the requisite perforator back to its origin from its “mother” vessel or even more dissection of the source vessel itself is the only way after the flap has been elevated. The flap initially can be designed eccentrically or more distally in relation to the required perforator, rather than centered about it, to increase reach. Either maneuver can be used to increase the pedicle length if used as a free flap.
22 How can venous congestion in a perforator flap be aborted?
Because the course of the perforator veins often is anomalous, whenever possible a subcutaneous vein should be preserved while harvesting a perforator flap. This is especially true if large superficial veins are encountered during flap elevation, as a reciprocal relationship often exists with the deep system. The superficial vein then can be used to “supercharge” the venous outflow. In addition, other large perforator veins should be identified as divided and set aside to allow the same possibility of “supercharging” them later, particularly if the superficial veins are inadequate.
23 Is the immediate thinning of a perforator flap hazardous?
The arbitrary removal of fat from the bottom surface of a perforator flap, even while intentionally leaving fat around the perforator itself, could potentially interfere with maximum viability. The exact course of perforators in the subcutaneous tissues can be variable. Also, venous outflow could depend on maintaining an intact superficial system that could thereby be disrupted.
MUSCLE PERFORATOR FLAPS
24 Describe the nomenclature for muscle perforator flaps
Systems for nomenclature have been based on the name of the source vessel to the flap, its anatomic region of origin, and/or the muscle traversed. A universal system has yet to be adopted.
25 Based on the aforementioned four nomenclature systems for muscle perforator flaps, label a flap from the anterolateral thigh if based on a lateral circumflex femoral (LCF) perforator of the vastus lateralis muscle
26 Based on the aforementioned four nomenclature systems for muscle perforator flaps, label a flap if based on a superior epigastric (SE) perforator of the rectus abdominis muscle
27 Does a muscle perforator flap capture the same territory as its corresponding musculocutaneous flap?
It would seem intuitively obvious that if only a single or a few musculocutaneous perforators were kept with a perforator flap, flow would be less than to the same cutaneous territory where all perforators had been kept with a musculocutaneous flap. Indeed, using laser Doppler flowmetry in a rat perforator flap model, flow was shown to be less. Interestingly, total surface area viability nevertheless proved not to be different!
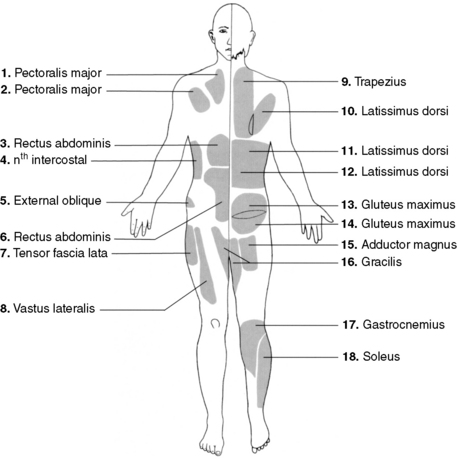
28 List the source vessels for the muscle perforator flaps identified here by the corresponding muscle (Fig. 108-3):
NONMUSCLE PERFORATOR FLAPS
29 Explain the basis of circulation to a neurocutaneous flap
The cutaneous sensitive nerves have both an intrinsic and an extrinsic neurocutaneous blood supply. The intrinsic supply, well known as the vaso nervorum, is maintained throughout the course of the nerve. It has multiple interconnections with the extrinsic system that arise either directly from a regional source vessel or secondarily from the surrounding fascial plexus. The latter forms “choke” and sometimes true anastomosis between networks of adjacent deep fascia perforators. Both not only supply the given cutaneous nerve, which is its primary intent, but also periodically are the origin of branches to the skin.
30 What is the axis of a neurocutaneous flap?
Because the paraneural vascular plexus follows the course of the nerve via either “choke” or true anastomoses, the predominant direction of blood flow or axis of a neurocutaneous flap will follow the path of the nerve itself.
31 Why were Pontén’s so-called “superflaps” so robust?
Pontén’s proximal-based and sensate fasciocutaneous flaps raised from the lower extremity, which he called “superflaps,” probably included the local cutaneous sensitive nerves. In reality, these probably were neurocutaneous flaps. As we know, any cutaneous flap oriented along the course of the sensitive nerve by definition has an axis with the same orientation that ensures maximizing blood flow to that flap and in turn greater survival length.
32 What landmarks can be used to ensure the appropriate orientation of a neurocutaneous flap in the extremities?
The major superficial venous channels in the extremities serve a dominant role as the means for outflow from the paraneural vascular plexus and thus tend to parallel the cutaneous sensory nerves. The sural flap (usually containing the lesser saphenous vein and medial sural cutaneous nerve) and cephalic flap (cephalic vein and lateral antebrachial cutaneous nerve) are two examples in which inclusion of the anatomically obvious vein will almost automatically allow incorporation of the nerve and usually its vascular plexus within the flap.
SUPERMICROSURGERY
33 Define “supermicrosurgery.”
A term coined by Koshima, supermicrosurgery is any microsurgery on structures 0.5 to 0.8 mm in diameter.
34 What is Koshima’s perforator-based flap?
Usually free-tissue transfers, in these flaps only the perforator is retained with the vascular pedicle. The “mother” vessel is not included. Because only the perforator is needed, dissection can stop at the fascial level. Microanastomoses are then made to the perforators alone, often truly requiring the skills of a “supermicrosurgeon.”
35 Describe how to design a “free-style” local or free flap
If a satisfactory, discrete perforator can be identified anywhere in the body, a flap can be designed about it. The constraints of using only described territories can be disregarded, allowing the choice of donor site to be determined by selection of the best possible available tissues as needed to match color, contour, texture, etc., at the recipient site.
36 How does “microdissection” more safely allow reduction of the thickness of a perforator flap?
In this technique developed by Kimura, an operating microscope is used, essentially for careful separation of the superficial and deep adipose tissue layers from each other. The deep adipose tissue is teased away from the perforator starting at the level of the deep fascia. This dissection continues up to the superficial adipose layer, creating a void about the intact perforator and its branches. Flap elevation is then completed at the superficial adipose level proceeding away from the requisite perforator (Fig. 108-4).
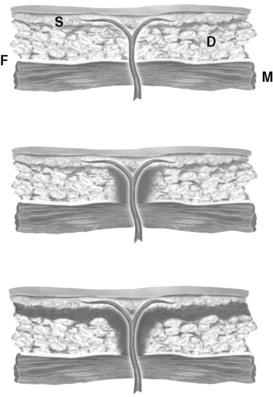
Figure 108-4 Technique of microdissection. Top, Initial course of perforator. D, Deep adipose layer; F, deep fascia; M, muscle; S, superficial adipose layer. Center, Removal of deep adipose tissue begins around the perforator starting at the level of the deep fascia to create a hole extending to the superficial adipose layer. Bottom, Completion of elevation of the cutaneous flap peripheral to the perforator, staying just beneath the superficial adipose layer.
(Courtesy Carol Varma, Multimedia Communication Manager, The Lehigh Valley Hospital, Allentown, Pennsylvania.)
37 What is a “subdermal vascular network” flap?
This is an ultrathin cutaneous flap with both the superficial and deep adipose layers removed, except carefully where retained about nutritive perforators. Both direct and indirect perforators have been used to supply this flap, often with disparate pedicles used for supercharging as necessary.
Alkureishi L.W.T., Shaw-Dunn J., Ross G.H. Effects of thinning the anterolateral thigh flap on the blood supply to the skin. Br J Plast Surg. 2003;56:401-408.
Blondeel P.N., van Landuyt K.H.I., Monstrey S.J.M., et al. The “Gent” consensus on perforator flap terminology: Preliminary definitions. Plast Reconstr Surg. 2003;112:1378-1383.
Blondeel P.N., Arnstein M., Verstraete K., Depuydt K., van Landuyt K.H., Monstrey S.J. Venous congestion and blood flow in free transverse rectus abdominis myocutaneous and deep inferior epigastric perforator flaps. Plast Reconstr Surg. 2000;106:1295-1299.
El-Mrakby H.H., Milner R.H. The vascular anatomy of the lower anterior abdominal wall: A microdissection study on the deep inferior epigastric vessels and the perforator branches. Plast Reconstr Surg. 2002;109:539-543.
Geddes C.R., Morris S.F., Neligan P.C. Perforator flaps: Evolution, classification, and applications. Ann Plast Surg. 2003;50:90-99.
Hallock G.G. Muscle perforator flaps: The name game. Ann Plast Surg. 2003;51:630-632.
Hallock G.G. Doppler Sonography and color duplex imaging for planning a perforator flap. Clin Plast Surg. 2003;30:347-357.
Hallock G.G. The superior epigastric RECTUS ABDOMINIS muscle perforator flap. Ann Plast Surg. 2005;55:430-432.
Hallock G.G., Rice D.C. Comparison of TRAM and DIEP flap physiology in a rat model. Plast Reconstr Surg. 2004;114:1179-1184.
Hallock G.G. Direct and indirect perforator flaps: The history and the controversy. Plast Reconstr Surg. 2003;111:855-866.
Kim J.T. New nomenclature concept of perforator flap. Br J Plast Surg. 2005;58:431-440.
Hyakusoku H., Gao J.H., Pennington D.G., Aoki R., Murakami M., Ogawa R. The microvascular augmented subdermal vascular network (ma-SVN) flap: Its variations and recent development in using intercostal perforators. Br J Plast Surg. 2002;55:402-411.
Kimura N., Satoh K., Hsaka Y. Microdissected thin perforator flaps: 46 cases. Plast Reconstr Surg. 2003;112:1875-1885.
Kimura N., Satoh K., Hasumi T., Ostuka T. Clinical application of the free thin anterolateral thigh flap in 31 consecutive patients. Plast Reconstr Surg. 2001;108:1197-1208.
Koshima I., Moriguchi T., Fukuda H., Yoshikawa Y., Soeda S. Free, thinned, paraumbilical perforator-based flaps. J Reconstr Microsurg. 1991;7:313-316.
Koshima I., Inagawa K., Urushibata K., Moriguchi T. Paraumbilical perforator flap without deep inferior epigastric vessels. Plast Reconstr Surg. 1998;102:1052-1057.
Koshima I., Nanba Y., Takahasi Y., Tsukino A., Kishimoto K. Future of supramicrosurgery as it relates to breast reconstruction: Free paraumbilical perforator adiposal flap. Semin Plast Surg. 2002;16:93-99.
Kroll S.S. Venous congestion and blood flow in free transverse rectus abdominis myocutaneous and deep inferior epigastric perforator flaps. Plast Reconstr Surg. 2000;106:1295-1299.
Kuo Y.R., Juo M.H., Chou W.C., Liu Y.T., Lutz B.S., Jeng S.F. One-stage reconstruction of soft tissue and Achilles tendon defects using a composite free anterolateral thigh flap with vascularized fascia lata: Clinical experience and functional assessment. Ann Plast Surg. 2003;50:149-155.
Masquelet A.C., Romana M.C., Wolf G. Skin island flaps supplied by the vascular axis of the sensitive superficial nerves: Anatomic study and clinical experience in the leg. Plast Reconstr Surg. 1992;89:1115-1121.
Nakajima H., Fujino T., Adachi S. A new concept of vascular supply to the skin and classification of skin flaps according to their vascularization. Ann Plast Surg. 1986;16:1-17.
Nakajima H., Imanishi N., Fukuzumi S., Minabe T., Aiso S., Fujino T. Accompanying arteries of the cutaneous veins and cutaneous nerves in the extremities: Anatomical study and a concept of the venoadipofascial and/or neuroadipofascial pedicled fasciocutaneous flap. Plast Reconstr Surg. 1998;102:779-791.
Niranjan N.S., Price R.D., Govilkar P. Fascial feeder and perforator-based V-Y advancement flaps in the reconstruction of lower limb defects. Br J Plast Surg. 2000;53:679-689.
Nojima K., Brown S.A., Acikel C., et al. Defining vascular supply and territory of thinned perforator flaps: Part I, anterolateral thigh perforator flap. Plast Reconstr Surg. 2005;116:182-193.
Pontén B. The fasciocutaneous flap: Its use in soft tissue defects of the lower leg. Br J Plast Surg. 1981;34:215-220.
Taylor G.I. The “Gent” consensus on perforator flap terminology: Preliminary definitions (discussion). Plast Reconstr Surg. 2003;112:1384-1387.
Villafane O., Gahankari D., Webster M. Superficial inferior epigastric vein (SIEV) “lifeboat” for DIEP/TRAM flaps. Br J Plast Surg. 1999;52:599.
Wei F.C., Jain V., Suominen S., Chen H.C. Confusion among perforator flaps: What is a true perforator flap? Plast Reconstr Surg. 2001;107:874-876.
Wei F.C., Mardini S. Free-style free flaps. Plast Reconstr Surg. 2004;114:910-916.
Wei F.C., Celik N., Jeng S.F. Application of “simplified nomenclature for compound flaps” to the anterolateral thigh flap. Plast Reconstr Surg. 2005;115:1051-1055.