Hypertrichosis and Hirsutism
Definitions
• Hypertrichosis: excessive hair growth on any area of the body, beyond what is normally expected for a patient's demographic group.
• Hirsutism: excessive terminal hair growth in women or children in a pattern typically seen in adult men.
• Lanugo hair: long fine hair that is grown in utero, covers the fetus, and is normally shed either in utero or during the first few weeks of life.
• Vellus hair: short, non- or lightly pigmented hair that covers most areas of the body; occasionally so fine as to not be appreciated clinically.
• Terminal hair: thick pigmented hair that is typical of the scalp and androgen-dependent areas, such as the axillae and pubic region in both sexes, as well as the beard, trunk, and limbs in adult males.
• Hyperandrogenism: elevated serum levels of testosterone, DHEAS, and/or androstenedione; cutaneous findings include hirsutism, severe or treatment-resistant acne, androgenetic alopecia, and seborrhea.
• Virilization: in women, clinical features of hyperandrogenism plus clitoromegaly, deepened voice, increased muscle mass, breast atrophy, and increased libido.
• Depilation: removal of a portion of the hair at some point along its shaft.
Hypertrichosis
• Can be classified based on the distribution (generalized vs. localized), age of onset (congenital or early onset vs. acquired), and type of hair (lanugo vs. vellus vs. terminal).
• Three mechanisms of hypertrichosis are generally recognized:
1. Conversion of vellus to terminal hair (e.g. localized hypertrichosis in an area of chronic rubbing or scratching).
2. Changes in the hair growth cycle (e.g. intranasal and ear hairs in older males due to prolongation of anagen phase).
3. Increase in hair follicle density beyond what is normal for a given site (e.g. congenital melanocytic nevus).
• Rx: treat underlying condition, if possible, otherwise see Table 57.2.
Table 57.2
Treatment options for hypertrichosis and unwanted hair.
Epilation technique results typically last longer than those of depilation. Chemical depilatories work by dissolving hair shafts, specifically by breaking disulfide bonds.

* More effective than calcium thioglycolate but more irritating and odiferous.
** These epilatory methods are operator-dependent and have the potential for permanent hair removal results.
IPL, intense pulsed light.
• Shaving of hairs does not increase the thickness or pigmentation of the hairs, contrary to common belief.
Generalized Hypertrichosis
• The presence of lanugo hair, excess vellus hair, or terminal hair on most of the body.
• An approach to a patient with generalized hypertrichosis is presented in Fig. 57.1.
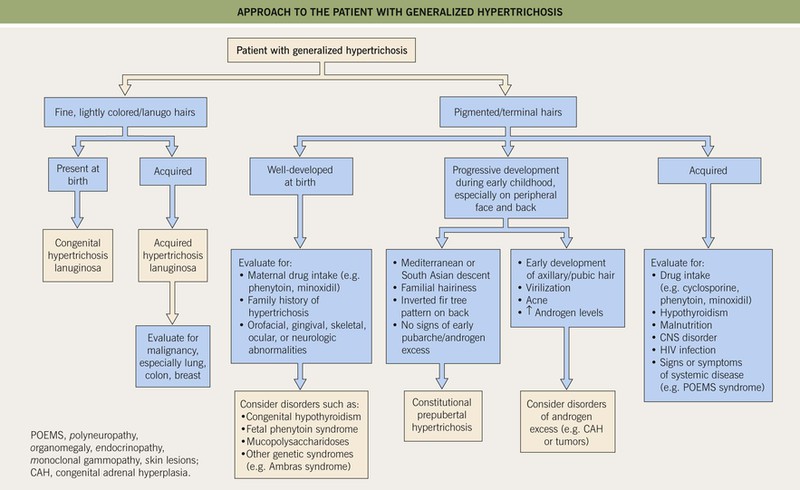
Fig. 57.1 Approach to the patient with generalized hypertrichosis. Acquired hypertrichosis lanuginosa occasionally may involve only the face.
• In young girls, constitutional prepubertal hypertrichosis is the most common form of generalized hypertrichosis.
Localized Hypertrichosis
• Most often involves a switch from vellus to terminal hair in sites that normally do not have terminal hairs.
• Congenital localized hypertrichosis is usually related to an underlying hamartoma or occurs at a specific anatomic site (Table 57.1; Figs. 57.2–57.6).
Table 57.1
Classification and key features of selected disorders of localized hypertrichosis.
LSC, lichen simplex chronicus; PCT, porphyria cutanea tarda; CSA, cyclosporine; EGFR, epidermal growth factor receptor.
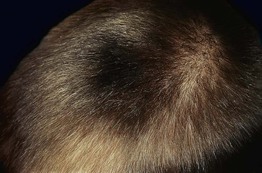
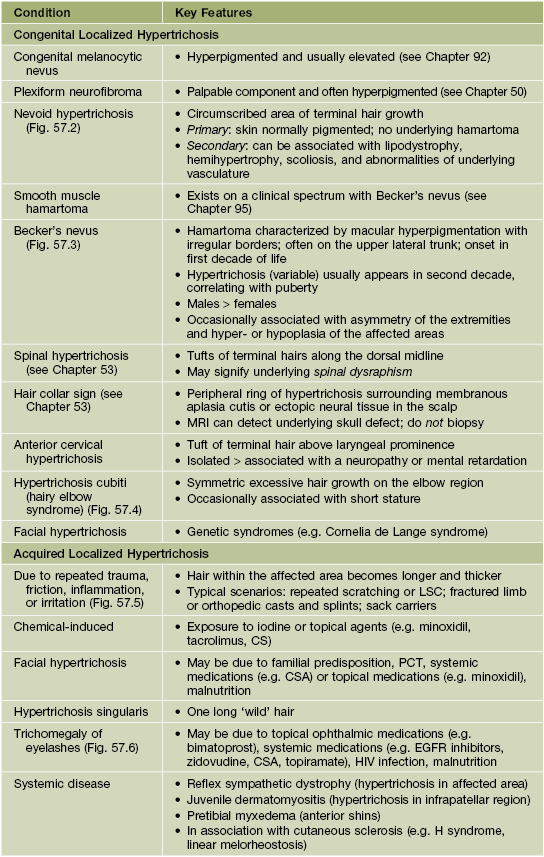
Fig. 57.2 Nevoid hypertrichosis in the scalp of a young boy. There was no underlying melanocytic nevus or hyperpigmentation. Courtesy, Jean L. Bolognia, MD.
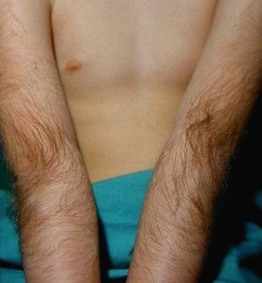
Fig. 57.4 Hypertrichosis cubiti. Multiple terminal hairs on both elbows in a child. Courtesy, Francisco M. Camacho Martinez, MD.
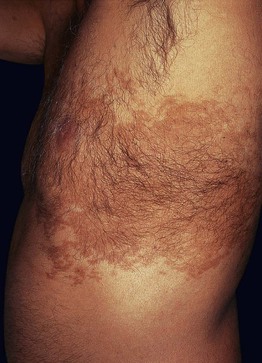
Fig. 57.5 Acquired localized hypertrichosis. Hypertrichosis, hyperpigmentation, and epidermal hyperplasia at the site of friction.
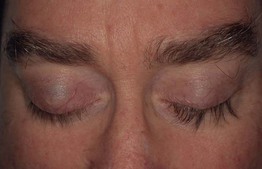
Fig. 57.6 Unilateral trichomegaly. Bimatoprost ophthalmic solution (0.03%) was applied to one eye to treat glaucoma. Courtesy, Jean L. Bolognia, MD.
• Acquired localized hypertrichosis most often develops after repeated trauma, friction, irritation, or inflammation (see Table 57.1).
Hirsutism
• Affects ~5–10% of females of reproductive age; can also affect postmenopausal women.
• Due to hyperandrogenism (exogenous or endogenous) or increased sensitivity of the hair follicle to normal androgen levels and is the most commonly used clinical criterion of androgen excess.
• Quantified using the modified Ferriman and Gallwey (mFG) method (Fig. 57.7).
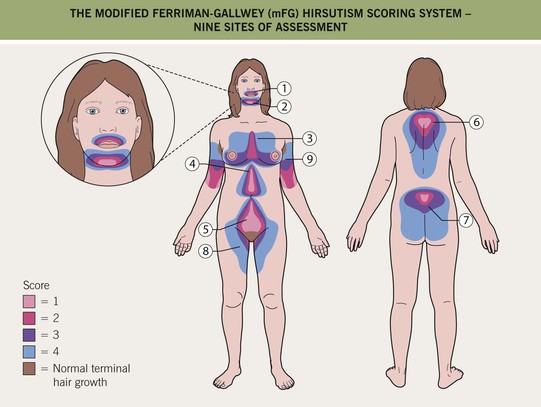
Fig. 57.7 The modified Ferriman–Gallwey (mFG) hirsutism scoring system. In this system, nine body areas are evaluated for the amount of terminal hair growth. A score of ‘0’ (no terminal hair growth) up to ‘4’ (frankly virile) is given to each of the nine areas and these are added together to compute a hormonal hirsutism score (mFG score). A total score <2–3 is considered normal for East Asian and Native American females, whereas <6–8 is considered normal in other populations.
• Defined as an mFG score of >2–3 in women from East Asia as well as Native Americans or an mFG score of ≥6–8 in other populations.
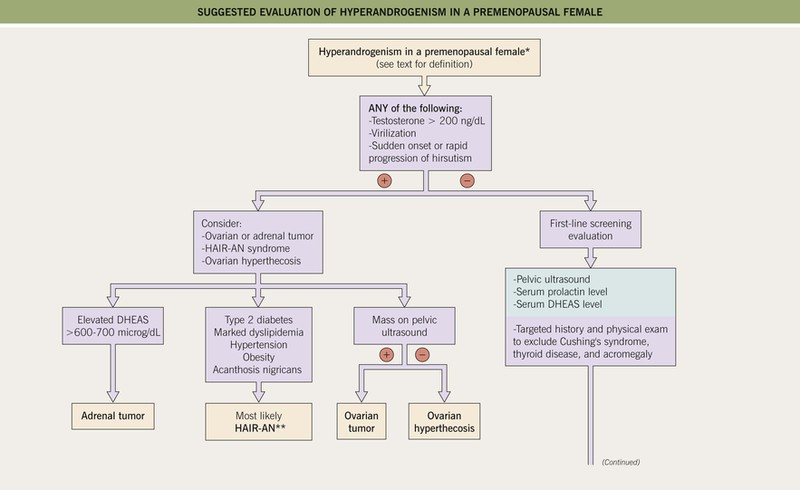
• Etiologies of hyperandrogenism and hirsutism in premenopausal females include.
– Most common: polycystic ovary syndrome (PCOS), idiopathic (end-organ sensitivity; Fig. 57.8).
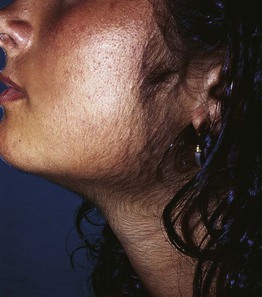
Fig. 57.8 Facial hirsutism in a young female. This can be due to hyperandrogenism or end-organ sensitivity. Courtesy, Francisco M. Camacho Martinez, MD.
– Less common: nonclassic congenital adrenal hyperplasia, ovarian hyperthecosis, tumoral.
– Must exclude: pregnancy; drugs (e.g. androgens, oral contraceptives with androgenic progestins, anabolic steroids, valproic acid).
• Etiology in postmenopausal women (new onset) is most likely ovarian hyperthecosis or tumoral hirsutism.
• Suggested algorithms for the evaluation of hirsutism and for hyperandrogenism are shown in Figs. 57.9 and 57.10, respectively.
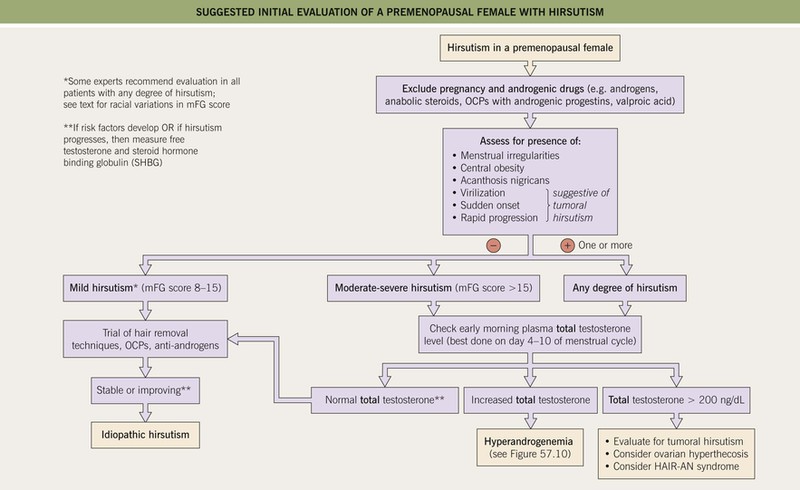
Fig. 57.9 Suggested initial evaluation of a premenopausal female with hirsutism (based on 2008 Endocrine Society clinical practice guidelines). OCP, oral contraceptive pills; mFG, modified Ferriman–Gallwey score (see Fig. 57.7); HAIR-AN, hyperandrogenism, insulin resistance, acanthosis nigricans syndrome.
Polycystic Ovary Syndrome (PCOS)
• Diagnosed by the presence of ≥2 of the following criteria and the exclusion of other possible etiologies (see Fig. 57.10):
1. Oligo- or anovulation (<8 menses/year or cycles >35 days).
2. Clinical and/or biochemical signs of hyperandrogenism (Fig. 57.11).
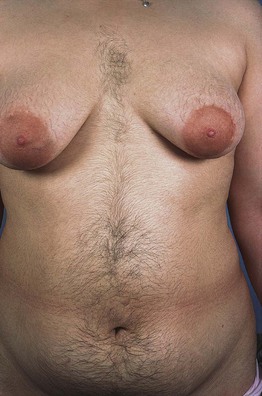
Fig. 57.11 Hirsutism in a young woman with polycystic ovarian syndrome (PCOS). Modified Ferriman–Gallwey (mFG) score of 9 based on three sites: chest, upper abdomen, and lower abdomen.
3. Polycystic ovaries (pelvic ultrasound).
• Occurs only during the reproductive years, and although the majority of patients are obese, some are of normal weight.
• Other associated findings may include acanthosis nigricans and insulin resistance; patients are at increased risk for the metabolic syndrome (see Table 45.11), infertility, obstructive sleep apnea, and possibly endometrial carcinoma.
• Rx of hirsutism: hormonal agents (Table 57.3) as well as general hair removal methods (see Table 57.2).
Table 57.3
Systemic agents and practical treatment recommendations for hirsutism and hyperandrogenism.
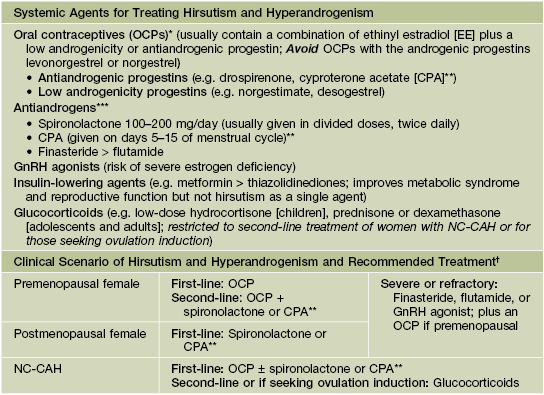
* Should avoid in females who smoke or who have risk factors for hypercoagulability and thrombosis.
** CPA is not available in the United States.
*** Requires concomitant reliable contraceptive method because of the risk of feminization of the male fetus.
† A waiting period of 6–9 months is recommended after initiating a treatment and before adding or changing medications.
NC-CAH, nonclassic congenital adrenal hyperplasia.
Idiopathic Hirsutism
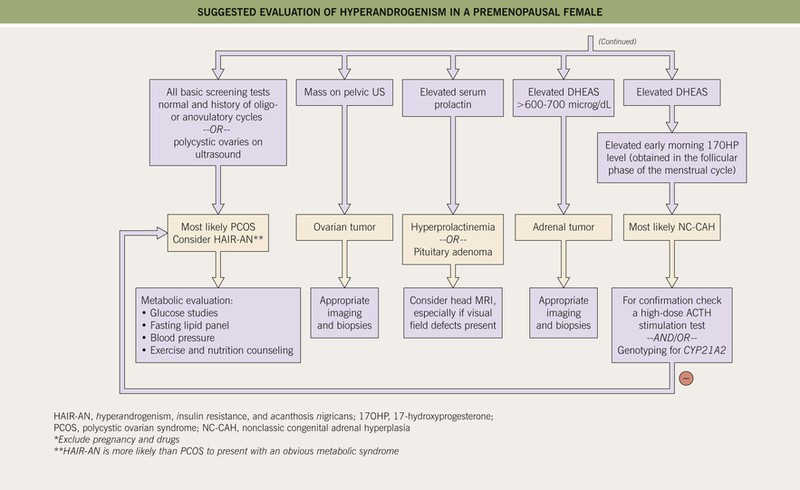
Nonclassic Congenital Adrenal Hyperplasia (NC-CAH)
• Autosomal recessive disorder due to partial deficiency in 21-hydroxylase activity (mutations in CYP21A2), leading to increased 17-hydroxyprogesterone (17-OHP) levels.
• More prevalent in Ashkenazi Jews, Hispanics, and Slavics.
• Usually presents in the peripubertal and young adult years with hirsutism, menstrual irregularities, infertility, androgenetic alopecia, and acne.
• Initial testing includes measuring an early morning 17-OHP level during the follicular phase of menstrual cycle; if equivocal, a high-dose ACTH stimulation test and/or genotyping for CYP21A2 can be performed.
• Rx of hirsutism: overlaps with that of PCOS, but may also include systemic CS (see Tables 57.2 and 57.3).
Ovarian Hyperthecosis
• Occurs in both premenopausal and postmenopausal females.
• Due to the differentiation of ovarian interstitial cells into steroidogenically active luteinized stromal cells, resulting in greater production of androgens.
• Clinical features are similar to those of PCOS but with more pronounced and long-standing hirsutism, an increased likelihood of virilization (Fig. 57.12), and the occurrence postmenopause.
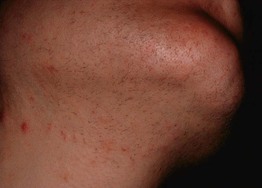
Fig. 57.12 Facial hirsutism (mFG score 4) due to ovarian hyperthecosis. Courtesy, Robert Hartman, MD.
• Testosterone levels are often quite elevated (>200 ng/dl); DHEAS can be normal or increased (see Fig. 57.10).
• Compared to tumoral hirsutism, a slower onset of symptoms and more gradual worsening over years.
• Increased risk of insulin resistance, type 2 diabetes, and endometrial carcinoma.
Tumoral Hirsutism
• Can be caused by a variety of benign or malignant ovarian or adrenal androgen-secreting tumors.
• Typically presents with either sudden onset or rapid progression of hirsutism and other clinical features of hyperandrogenism; virilization more likely.
• Diagnostic clues are extremely elevated levels of testosterone (>200 ng/dl, seen in ovarian and adrenal tumors) or DHEAS (>600–700 microg/dl, seen in adrenal tumors).
• If suspected, an appropriate imaging study is recommended (e.g. CT scan, MRI, and/or ultrasound).
HAIR-AN Syndrome
• Hyperandrogenism, insulin resistance, and acanthosis nigricans syndrome.
• Marked insulin resistance leads to secondary increased insulin levels and resultant increased ovarian androgen production.
• More likely than PCOS to have overt type 2 diabetes, hypertension, and cardiovascular disease.
• Marked hyperandrogenism and often frank virilization; total testosterone levels are often markedly increased.
• Evaluation involves excluding an androgen-secreting tumor and PCOS.
Hirsutism Associated with Other Endocrine Abnormalities
• Cushing's syndrome, hyperprolactinemia, pituitary adenoma, acromegaly, and thyroid dysfunction may present with hirsutism and other signs of hyperandrogenism, but typically manifest with other distinguishing and diagnostic features specific to the underlying disease (see Chapter 45).
For further information see Ch. 70. From Dermatology, Third Edition.