Chapter 19 Rhinoscopy
Rhinoscopic Diagnosis and Treatment of Nasal Diseases, Transnares Curettage for Palliation of Malignant Nasal Obstruction
Rhinoscopy is an invaluable component of the nasal workup. Rhinoscopy is used in conjunction with diagnostics such as blood work, thoracic radiographs, skull radiographs, and skull computed tomographic (CT) scans or magnetic resonance imaging (MRI). It provides visualization of the nasal cavity, choanae, nasopharynx, and in some patients, the frontal sinuses. Chronic, unrelenting nasal disease is a frustrating problem in dogs and cats, and the primary disease can often be difficult to diagnose.1,2 Rhinoscopy is useful for both diagnostic and therapeutic case management and reduces or eliminates the need for rhinotomy in most instances. Rhinoscopy is the final component of the nasal workup. A sequential diagnostic approach is imperative, and history taking, signalment, and a physical examination are the building blocks. Nasal disease causes a variety of symptoms.
Indications
Rhinoscopy indications include chronic intermittent to paroxysmal sneezing, nasal congestion, epistaxis and stertor, or nasal airflow obstruction. As numerous nasal diseases will share a common constellation of symptoms, rhinoscopy serves as an effective tool for diagnosis provision and symptom relief from nasal tumors, nasal foreign bodies, fungal rhinitis, and both acquired and congenital nasal anatomic abnormalities such as nasal and nasopharyngeal stenoses. Rhinoscopy tends to be less effective therapeutically in the management of allergic or lymphoplasmacytic rhinitis, unless debridement of necrotic infected tissue is necessary for symptom relief. In the case of allergic rhinitis, rhinoscopy serves to rule out other potential diseases but rarely provides specific information about the origin or cure of allergic rhinitis.
Contraindications
Rhinoscopy is contraindicated in animals with primary or secondary hemostatic defects. Prolonged or excessive epistaxis can be seen in the hypertensive patient. Rhinoscopy should be pursued with extreme caution when a disease process has infiltrated the cribriform plate or there is evidence of increased intracranial pressure.
Equipment
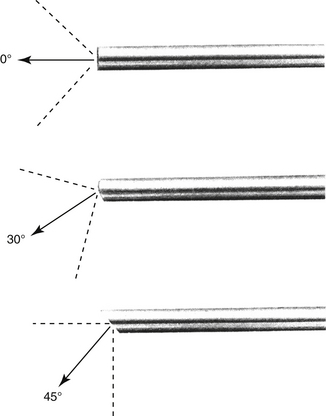
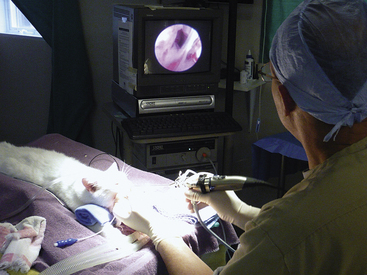
There are many equipment options available for direct visualization of the nasal cavity. The most effective tools are rigid telescopes with operating sheaths capable of irrigation and instrument passage within all nasal cavity regions. Otoscopes are only useful in nasal disease evaluation within the most rostral nasal compartment. Although preferred by some clinicians, flexible endoscopes are rarely needed and provide no additional diagnostic or therapeutic benefit to the clinician. Rigid endoscopes are the main tools used in rhinoscopy. Two rigid endoscope sizes are commonly used in small animal rhinoscopy. The 1.9- and 2.7-mm 30-degree visual field endoscopes can be used to adequately evaluate most cats and dogs the clinician will encounter in clinical practice. In cats and small dogs less than 10 kg, the 1.9-mm 30-degree integrated endoscope is preferred (Figure 19-1). In patients more than 10 kg, a 2.7-mm diameter multipurpose endoscope with a triport cystoscopy operating sheath is preferred (Figure 19-2). Some clinicians prefer using the arthroscopy sheath with the 2.7-mm scope as it has a slightly smaller working diameter but a greater flow rate when high irrigation is needed (Figure 19-3). The integrated scopes and operating sheaths generally have a parallel biopsy channel and two additional 90-degree channels with stopcocks for irrigation and suction. The arthroscopy sheath has only a single 90-degree channel for irrigation and is more than adequate in most instances because the biopsy and suction ports are seldom needed for their stated uses. The working length of these two scopes enables adequate visualization of the choanae and the nasopharynx in all but the largest of dogs. In giant breeds, a 4.0-mm diameter 30-degree visual field endoscope with a modular cystoscopy operating sheath is recommended for complete rhinoscopic examination (Figure 19-4). Angled visual field endoscopes require some clinician adaptation but when used properly allow for a much greater rotated hemispheric view with reduced traumatic leverage compared with 0-degree visual field scopes (Figure 19-5). This is a clinician preference, and either scope can be used on a daily basis to perform complete rhinoscopy safely. The chief advantages of rigid endoscopes are in their ability to provide reliable spatial orientation, high-flow irrigation, and the physical leverage used in different rhinoscopic techniques and to facilitate diagnostic biopsy.

Figure 19-1 Karl Storz model No. 27030BA 1.9-mm 30-degree visual field integrated rigid endoscope.
(Photo courtesy of Karl Storz GmbH & Co. KG, Tuttlingen, Germany.)
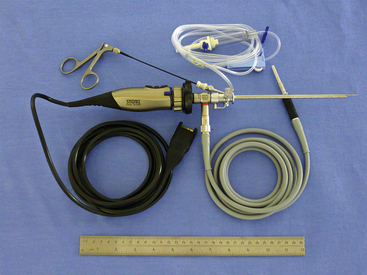
Figure 19-2 A 30-degree visual field rigid endoscope with camera, light cable, irrigation line, and biopsy instrument inserted.
(Photo courtesy of Karl Storz GmbH & Co. KG, Tuttlingen, Germany.)

Figure 19-3 Karl Storz model No. 64018B5A, 67065C, 6412BAR, 2.7-mm 30-degree visual field rigid endoscope with cystoscopy and arthroscopy sheaths.
(Photo courtesy of Karl Storz GmbH & Co. KG, Tuttlingen, Germany.)
Evaluation of the Patient with Nasal Disease
History and Signalment
A good historical evaluation of the problem along with the patient signalment can often support lesion localization before the physical examination and help guide diagnostics. Most nasal diseases can have both acute and chronic stages. Intermittent and paroxysmal sneezing tends to be seen with nasal cavity disease, whereas reverse sneezing and hacking (postnasal drip) can indicate problems in the nasopharynx or middle ear via the eustachian tube. Stertor without sneezing often suggests nasopharyngeal disease.
Nasal discharge characteristics are often nonspecific because serous, mucopurulent, and overt epistaxis can be seen with any type of nasal disease depending on the stage and duration of the problem. Primary epistaxis should suggest potential hypertension, tick-borne vasculitis, or coagulopathy. These should be investigated by the clinician before rhinoscopy. Bilateral signs occur most often with inflammatory conditions. Anatomic defects such as cleft palates can cause recurring rhinitis but should be easily identifiable without specialized diagnostics. Although rare, ciliary dyskinesia should be considered in the very young dog with recurrent bilateral mucopurulent rhinitis. Unilateral signs are often associated with foreign bodies, fungal infections, and tumors. Prior responses to an antibiotic, antiinflammatory, or antihistamine are nonspecific observations because most significant nasal cavity disease is complicated by a secondary bacterial infection, local inflammation, and reflexive mucus production. Inflammatory rhinitis, nasopharyngeal polyps, foreign bodies, and fungal infections tend to occur more in younger to middle-aged patients. Tumors tend to be more prevalent in the middle-aged to geriatric populations. Seasonal or geography-associated symptoms tend to predict allergic or inflammatory conditions (Box 19-1).
Physical Examination
The nasal cavity is surrounded by bone, which severely limits physical examination. However, with experience and attention to detail, the clinician can often localize the site of disease and narrow or prioritize the list of possible causes with a regimented physical examination. Physical examination findings are variable and can include the presence or absence of stertor, sneezing or reverse sneezing, symmetry or asymmetry of nasal airflow, nasal discharge, external nares structure, facial symmetry, and ocular retropulsion abnormalities, along with localizable pain (especially over the maxilla, sinuses, and periocular regions).
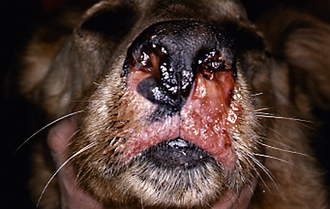
External evaluation of the nose and nasopharynx begins with examination of the nostrils and face. The external nares should be checked for the character of discharge and ulceration of the epithelium. There is a distinct appearance of the nostril in animals with fungal rhinitis that includes ulceration and hypopigmentation. Hold the mouth closed with one hand, and close off one nostril with the other. Unilateral or bilateral obstruction can be determined with this maneuver. Animals with obstruction become frantic until they can open their mouths and breathe. If air movement is normal, suspect a generalized rhinitis or possibly a systemic disorder causing nasal symptoms. Epistaxis with normal movement of air through both nostrils should raise a red flag. Epistaxis can be unilateral or bilateral and can occur without primary nasal disease. In animals without obvious obstructive nasal disease, a coagulopathy, tick-borne vasculitis, or hypertension must be ruled out before general anesthesia and rhinoscopy are pursued. Fundic examinations are recommended in all patients with epistaxis. Palpate the face to check for asymmetry or bony defects in the skull. Destruction of bone indicates a neoplastic process. Pain can be seen with erosive neoplasms but is also seen with fungal rhinitis and peracute sinusitis. If the patient allows, examine the oral cavity, teeth, gums, and hard and soft palates both visually and through palpation. Erosion of the hard palate or ventral deflection of the soft palate by a lesion is sometimes visible or palpable during oral examination. Severe dental disease can cause rhinitis or overt oronasal fistulae typically in geriatric small to miniature dog breeds. Significant gingival recession, overt cavities, and loose teeth should prompt consideration of nasal disease of dental origin. Retropulsion of both eyes should be similar. A decrease in caudal movement of one eye indicates a periorbital, orbital, or retrobulbar swelling. Evaluate the movement of air through both nostrils.
Prerhinoscopic Clinical Pathology
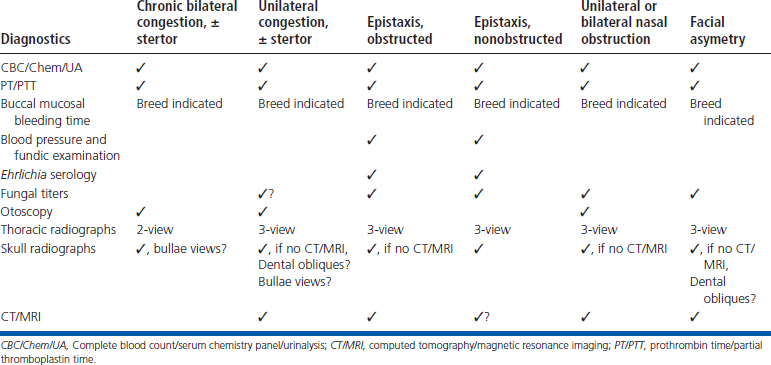
A complete blood count, serum chemistry panel, urinalysis, prothrombin time (PT) and partial thromboplastin time (PTT), and chest radiographs are recommended before rhinoscopy in all patients. In at-risk breeds, either a Von Willebrand’s factor analysis or buccal mucosal bleeding time is recommended. If epistaxis without obstruction is present, tick serologic analysis and a blood pressure measurement is indicated. Fungal serologic analysis is indicated in specific cases that are determined by geographic location and clinical suspicion. Negative fungal serologic results do not always rule out a particular fungal infection. Specific fungal serologic and polymerase chain reaction tests will have variable specificity and sensitivity with respect to the detection of specific fungal infections and should be assessed at the interpretation of results.3,4 Further steps in the diagnostic workup require general anesthesia.
Radiography
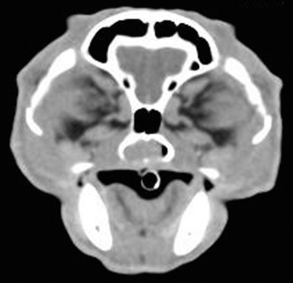
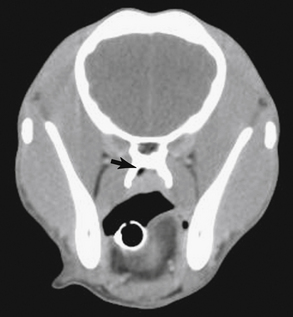
Radiographic imaging or CT is always performed before rhinoscopy. Rhinoscopic iatrogenic hemorrhaging and irrigation fluid used during the procedure will distort tissue and create fluid densities that can influence the findings in any radiographic study. Nasal radiographs can be performed in most hospitals and, with practice, can be very useful in the localization and characterization of nasal and nasopharyngeal disease.5 Nasal radiographs require general anesthesia and special patient positioning. Most commonly performed are 30-degree beam-angled open-mouth ventrodorsal and straight lateral views. The open-mouth view provides a complete, symmetrical image of the nasal cavity without superimposition of the mandible. Tumors typically show asymmetrical opacities and turbinate destruction (Figure 19-6). Turbinate destruction is also seen with fungal rhinitis. The straight lateral view allows evaluation of the nasopharynx and frontal sinuses (Figure 19-7). Further study of the frontal sinuses is achieved with a rostrocaudal tangential view (Figure 19-8). This allows comparison of the left and right frontal sinuses as the cavities are superimposed on the lateral view. Dental films of the maxillary arcade are sometimes needed to rule out tooth root abscesses and oronasal fistulae. MRI and CT scans are becoming more commonly available and provide better detail of the nasal, nasopharyngeal, and sinus cavities and cranial vault anatomy compared with standard radiography.6,7 Advanced imaging via MRI and CT, although considered state of the art, assist in description and localization of diseased tissue but have not yet produced results specific enough to eliminate the need for rhinoscopy8,9 (Figures 19-9 through 19-11) (Table 19-1).
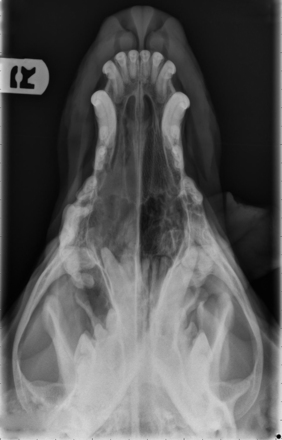
Figure 19-6 Canine open-mouth ventrodorsal view with right-sided destructive soft tissue mass; nasal osteosarcoma.
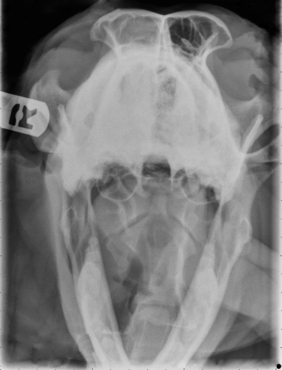
Figure 19-7 Canine tangential sinus view with right-sided frontal sinus soft tissue opacity; nasal osteosarcoma.
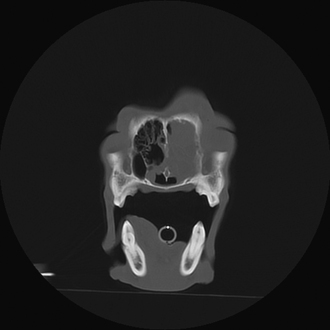
Figure 19-10 Canine computed tomographic scan; sagittal section through the caudal nasal cavity showing left-sided infiltrative soft tissue mass with septal and nasal bone osteolysis.
Anesthesia for Rhinoscopy
Although rhinoscopy is less invasive compared with rhinotomy, adequate preoperative neuroleptanalgesia, intraoperative and postoperative analgesia, and antiinflammatory treatments are recommended based on the patient’s preoperative evaluation. Preanesthetic nonsteroidal antiinflammatory drugs (NSAIDs) should be used with caution and forethought because of potential inhibitory effects of some agents on platelet aggregation, as well as other adverse primary and drug interactive secondary reactions.10 Alpha-2 agonists can cause significant hypertension11 and should be considered potentially disadvantageous when used in a procedure likely to induce significant epistaxis. Anesthetic induction and intubation should be performed promptly because patients with nasal obstruction cannot breathe when their mouths are closed following heavy sedation. Immediate airway protection via appropriate endotracheal intubation will also help to prevent aspiration of infected postnasal secretions. Anesthesia maintenance is often provided via gas inhalation because all patients are intubated. During rhinoscopy abrupt and forceful sneezing and nasopharyngeal reflexive movements can be observed even in the adequately anesthetized patient. These forceful movements can greatly affect procedural ease, as well as induce patient trauma and cause equipment damage. Bilateral nerve blockade at the level of the infraorbital foramen can greatly reduce the sneeze reflex, and a combination of lidocaine and bupivacaine is recommended in dogs and at reduced doses in cats. Intraoperative and perioperative, culture-guided antibiotic therapy is recommended because of the high incidence of secondary bacterial rhinitis in the patient with nasal disease. Endotracheal tube cuff evaluation should be rechecked before any flushing because of the potential for iatrogenic flushing or aspiration of infected nasal secretions into the lungs.
Technique: Rigid Antegrade Rhinoscopy
Patients are placed in a sternal recumbent position. The cuffed endotracheal tube is secured to the mandible. A rolled towel is placed beneath the animal’s neck to point the muzzle in a ventral direction, and an oral speculum is placed to hold the mouth open (Figure 19-12). Fluid and tissue will then tend to run out of the nostrils and mouth rather than collect around the larynx. No gauze is stuffed in the oropharnx. This impedes the movement of fluid from the nasopharynx into the mouth. The animal’s front legs are pulled laterally or covered with plastic to prevent soiling. Again, endotracheal tube cuff evaluation should be rechecked before any flushing because of the potential for iatrogenic flushing or aspiration of infected nasal secretions into the lungs.
Some clinicians prefer to collect samples for culture before rhinoscopy begins via blind insertion of the culturette through the nares. In some instances this can result in significant hemorrhage that will impair rhinoscopic visualization. We recommend tissue submission from biopsy samples collected after the rhinoscopic examination has been completed.
The scope is inserted at a slightly dorsal angle through the dorsal medial aspect of the nostril. Some prefer entering the ventral medial aspect of the nostril. Once past the nostril and the alar cartilage, the scope is straightened and advanced in a caudal direction. The nasal septum provides a source of anatomic reference. The three primary meatus should be examined. Mucosal changes, turbinate structure, and the nature of discharge are evaluated. Advance the scope slowly and passively to diminish bleeding. Allow the scope to follow the meatus with gentle pressure. When resistance is met or the field is obscured, pull the scope back gradually until visibility is restored. The goal is to examine as much of the nasal cavity as possible. Ease of examination is influenced by the size of the animal, breed, skull conformation, and the underlying disease.
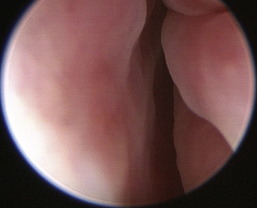
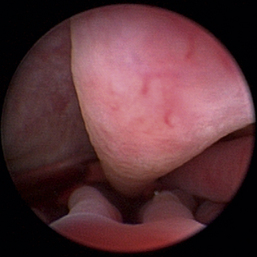
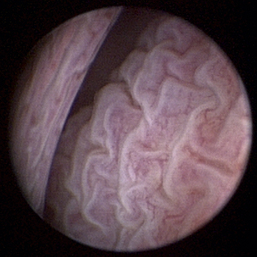
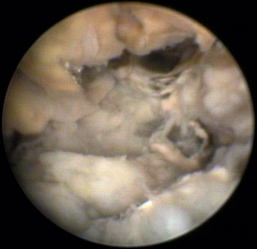
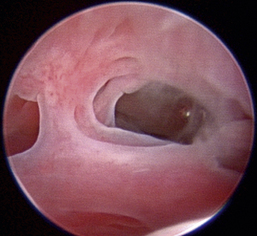
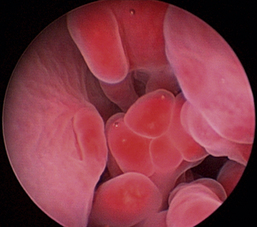
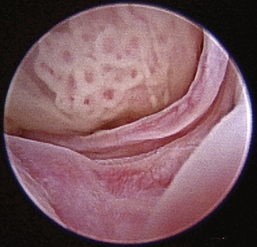
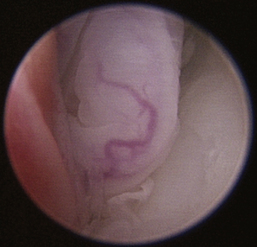
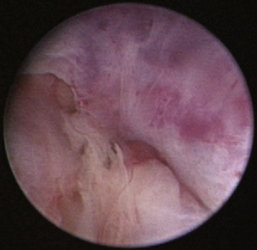
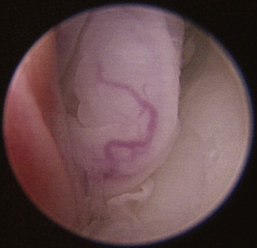
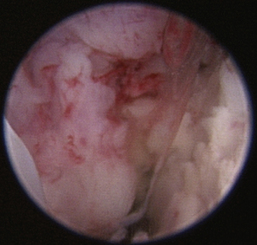
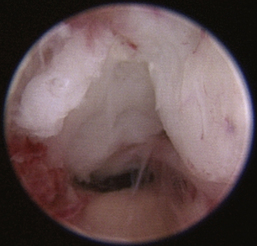
Normal turbinates have a smooth, pink-to-white surface and a spatial alignment that provides channels for the passage of air. The color varies and appears tan in the caudal nasal cavity. Turbinates come in different shapes and sizes, but the surface should be smooth. Ethmoid turbinates in the caudal nasal cavity will have a characteristic stippled or corrugated appearance (Figures 19-13 through 19-16). Ulcerations or various proliferations of the mucosa are indicative of disease. These changes are typically diffuse and accompanied by a lot of mucus. Mucus can be thin to purulent. Copious amounts can be flushed out of the nasal cavity to allow better visibility. The specific disease is determined by biopsy. After full examination of the nasal cavity, run the scope along the floor of the nasal sinus to the level of the choanae. Keeping the scope pointed in a ventral medial direction prevents inadvertent trauma or penetration of the cribriform plate. The index finger of the free hand can be used to follow the scope as it moves caudally over the hard palate; the surgeon can then palpate the scope through the soft palate when it moves into the nasopharynx. Some force is needed to complete this procedure and may result in increased bleeding.
Flexible fiberoptic endoscopes are preferred and utilized effectively by some clinicians, but we do not routinely use them for primary evaluations. Retrograde placement of these scopes behind the soft palate allows visualization of the nasopharynx, choanae, and caudal segments of the nasal passages. Flexible endoscopes can also be used in an antegrade fashion through the nostrils in some patients but are limited by the scope and patient size. Irrigation is limited with small fiberoptic scopes; biopsy instruments are diminutive at best, and the reliability of representative tissue collection should be questioned when this technique is used. The 2.5-mm fiberscope has a single channel used for both irrigation and instrument passage. When an instrument is passed, the irrigation flow is markedly reduced, drastically affecting visualization during biopsy collection. The spatial orientation estimation and direct visualization provided by flexible endoscopes is inferior when compared with rigid telescopes. In most cases, rigid telescopes can be passed through the nasal cavity, choanae, and into the nasopharynx, eliminating the need for flexible retrograde rhinoscopy.
After the visual examination is completed, tissue samples are collected for culture and biopsy evaluation. Specialized endoscopic instruments are available for collecting samples through the biopsy channel of all scopes. Depending on the scope size and instrument channel, tissue samples may be very small, and the reliability of such minuscule samples should be questioned by the clinician (Figure 19-17). If endoscopic instruments are utilized, multiple (>12) samples should be collected to increase the accuracy of diagnosis. We recommend an alternate technique in which tissue samples are collected for biopsy evaluation by passing larger instruments parallel to the scope or retrieving samples without direct visualization based on lesion spatial approximation. With practice, the clinician should be able to spatially localize the diseased tissue (on the basis of the depth of penetration of the rigid scope, the angle of insertion, and sometimes visible transnasal illumination) and insert an instrument to the site large enough to attain grossly substantial tissues. The sample site is then reexamined with the scope after biopsy, and the process is repeated to the clinicians’ satisfaction. Even foreign bodies can be removed in this fashion. Focal, discrete lesions are uncommon in the patient with grossly symptomatic nasal disease. Direct visualization is recommended when these sites are sampled. Pass an alligator forceps or a cup biopsy forceps outside the scope until the instrument can be seen. This affords visualization of the biopsy site. It can be a tedious process, but the samples are adequate. Blind biopsy techniques do not diminish the value of rhinoscopy, and adequate tissue submission should be the ultimate diagnostic goal. Tissue samples are submitted for histopathologic analysis and aerobic bacterial culture in most cases. Mild-to-moderate, spontaneously resolving hemorrhaging is expected after the collection of samples for culture and biopsy evaluation.
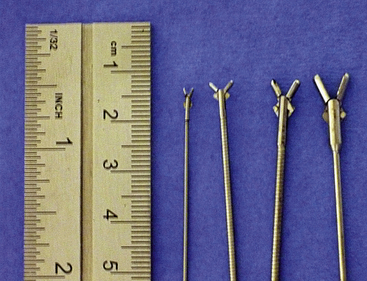
Figure 19-17 Cup forceps comparison left to right, 2.5-mm fiberoptic and 1.9-mm integrated cup, 2.7-mm cystoscopy sheath cup, 4.0-mm cystoscopy sheath cup, free cup.
Transnares curettage (TNC) is performed as described later in Chapter 19 for obstructive neoplastic and hyperplastic states and to debride fungal plaques before local administration of antifungal medications. After sample collection or TNC the nasal cavity is copiously flushed to remove loose tissue fragments, blood clots, nasal secretions, and debris. The oropharynx is carefully suctioned. Extubation is performed as late as the patient will tolerate to allow for maximal recovery of gag responses and oral respiratory compensation with persistent nasal obstruction. Some patients require makeshift temporary oral speculum placement to maintain oral airflow. Alternatively, a hanging maxilla technique can be used to provide the same effect.
Of note, some clinicians prefer the use of an air interface when performing rhinoscopy. Saline irrigation will reduce visible hyperemia immediately via reflexive mucosal vasoconstriction. We have found an air interface to be less advantageous over a saline interface because most patients with significant nasal disease require lavage of the nasal mucosa for full visualization of nasal anatomy. Preexisting mucus, hemorrhages, and debris will greatly reduce the productivity of the rhinoscopic examination unless properly managed.
Rhinoscopy Complications
Rhinoscopic-induced epistaxis is expected to persist for 1 to 7 days and up to 2 weeks after aggressive TNC. The client should be made aware of this in advance to make any appropriate arrangements at home to accommodate for this temporary side effect.
Significant rhinoscopic complications are rare with appropriate patient selection, preparation, and technique. Significant complications include severe epistaxis with infiltrative malignancy presence, encephalitis with cribriform plate defects, and pneumonia or pneumonitis after aspiration of contaminated irrigation fluids or preexisting infected mucus. Aspiration is often seen with improper endotracheal intubation, suboptimal cuff insufflation, or in the immediate anesthetic recovery period after extubation while the patient is heavily sedated and coughing is suppressed from opioid analgesic administration.
Specific Rhinoscopic Diagnoses
Nasopharyngeal Diseases
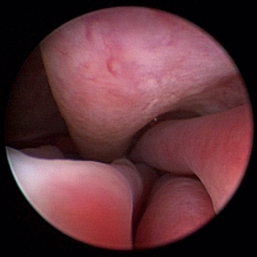
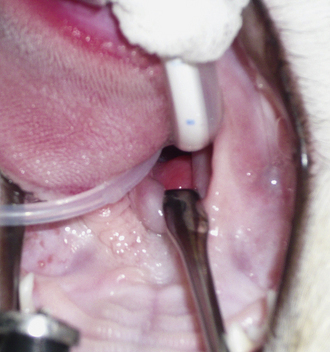
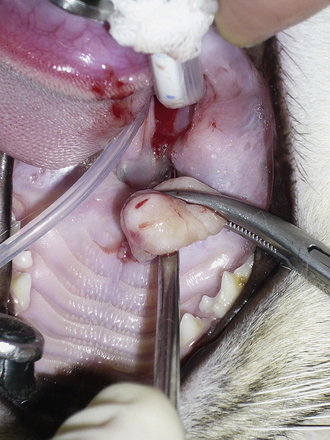
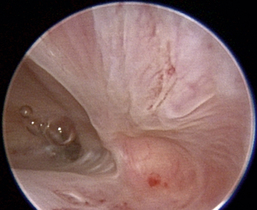
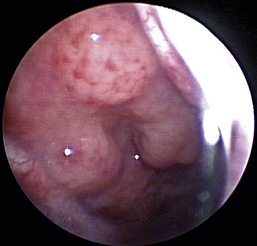
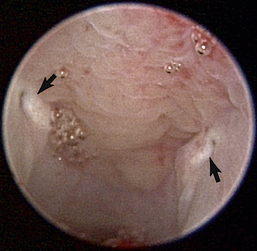
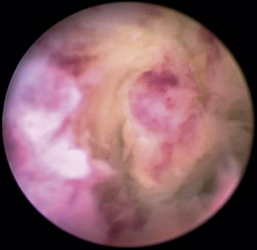
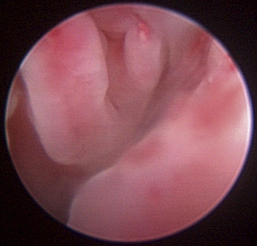
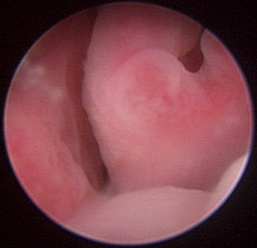
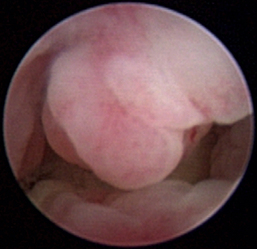
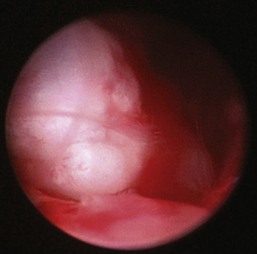
The hallmark symptom of nasopharyngeal disease is stertor. Discharge, sneezing, reverse sneezing, and hacking may be present variably. If the main complaint is noisy breathing and not productive sneezing with nasal discharge, the nasopharynx may be the site of primary disease. The most common primary problems in the nasopharynx in cats are inflammatory polyps and tumors.12,13 Cats with polyps are generally young and have a history of noisy breathing. Sneezing and mucopurulent nasal discharge may be present, but the chief complaint is stertor and increased respiratory effort. This is a problem that often requires otoscopic as well as rhinoscopic intervention. With the patient placed in dorsal recumbency, the soft palate is reflected rostrally with the use of a spay hook, and the polyp is removed via traction and avulsion (Figures 19-18 and 19-19). The eustachian tube orifices can generally be directly visualized and any remaining tissue removed, if present. Both ears should be carefully examined, and transtympanic bulla curettage should be performed to remove any middle ear proliferative tissue (see Chapter 20). In older cats with similar symptoms, a neoplasm in the nasopharynx is more likely, and rhinoscopy is often much more rewarding (Figure 19-20). Foreign bodies can become lodged in this area after vomiting or pharyngeal nasopharyngeal spasms induced by attempted swallowing of the foreign object. Nasal and nasopharyngeal stenosis after trauma, nasogastric reflux,14 and chronic rhinitis can occur in dogs and cats (Figures 19-21 through 19-23). Reflux can occur during general anesthesia and can be a helpful clue if postoperative symptoms arise and persist. Congenital choanal atresia and nasopharyngeal dysgenesis have also been reported in dogs.15,16 Nonspecific chronic inflammation of the nasopharynx causes a cobblestone appearance of the mucosa due to the formation of hyperplastic lymphoid follicles (Figures 19-24 and 19-25). Chronic inflammation may be the result of a lymphoplasmacytic rhinitis complex or sometimes may be secondary to otitis media and middle ear drainage of infected secretions via the eustachian tube. Otoscopy is recommended in any patient with nasopharyngitis of unknown origin. CT studies performed in a population of 45 cats with chronic sinonasal discharge revealed effusive bulla disease in 28% of symptomatic cats.17 Unfortunately, we are unable to differentiate primary and secondary pathology based on the concurrence of nasal and middle ear disease. Regardless of the knowledge of the primary cause, both inflammatory foci should be treated to achieve symptom relief.
Neoplasia
Foreign Bodies
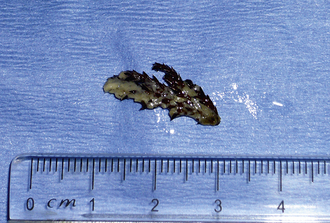
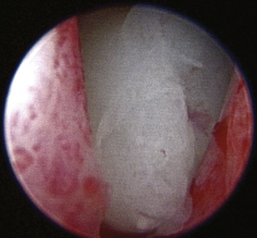
Nasal foreign bodies can present with extreme variability depending on the foreign material, amount, and skull conformation of the patient. Unilateral to bilateral mucopurulent to mucohemorrhagic discharges can be present. Most patients’ histories will include a response to medication and symptom recurrence on treatment cessation. Foreign bodies can be either radiodense or radiolucent. Because of extensive regional soft tissue changes, nasal foreign bodies can be mistaken for tumors, even with advanced imaging.18 Foreign bodies generally require rhinoscopy for both definitive diagnosis and removal (Figures 19-26 through 19-28).
Parasitic Rhinitis
Pneumonyssoides caninum infection has been well characterized in dogs.19 Diagnosis requires direct examination and observation of the mite within the nares and rostral nasal compartment. Infection most often causes reverse sneezing and has been considered to be an etiologic factor of gastric dilatation volvulus complex in breed-predisposed canines.20 Successful treatment has been reported using ivermectin,19 milbemycin oxime,21 and selamectin.22
Fungal Rhinitis
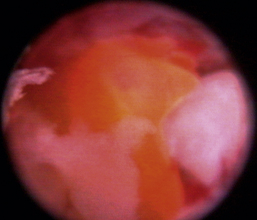
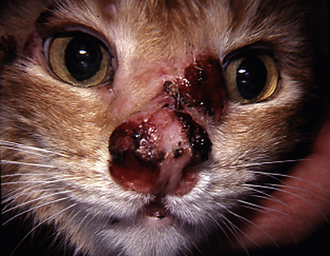
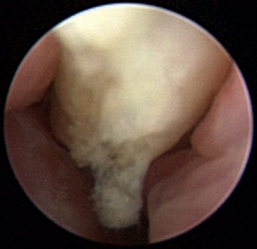
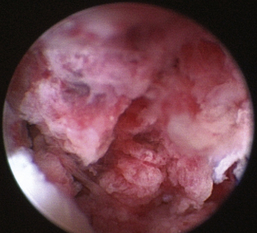
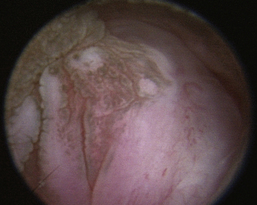
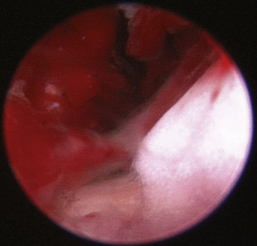
The hallmark presentation for the patient with fungal rhinitis varies somewhat with the fungus in question and domestic species affected4,23-25 (Figures 19-29 and 19-30). No response or a transient improvement can be seen with antibiotic therapy because secondary bacterial infections can be present.
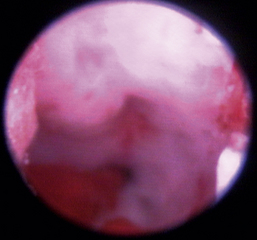
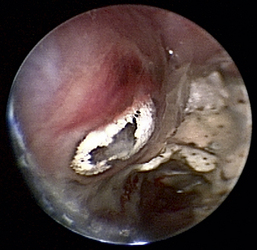
Aspergillus fumigatus is the most common canine fungal rhinitis.26 Chronic, unilateral mucopurulent to mucohemorrhagic nasal discharge is often reported. Serologic,3 CT, and MRI27 findings lack sensitivity and specificity, respectively, for reliable diagnosis of sinonasal aspergillosis. Rhinoscopic visualization and histopathologic evaluation are necessary for diagnosis confirmation. The classic fungal rhinitis lesion is a green-gray, whitish, necrotic plaque in the midcaudal nasal cavity. Regional turbinate reactive hyperplasia can be present, making the visual examination findings heterogeneous at best (Figures 19-31 through 19-33). The frontal sinuses should be examined in patients with suspected fungal rhinitis because treatment of all fungal mats is necessary for disease resolution. Sinusoscopy, sinusotomy, or both are often recommended to access and debride frontal sinus fungal mats and for antifungal instillation.28 Several treatment protocols have been described employing either single-agent sinonasal lavages or systemic antifungal therapy, depending on the fungus. We believe rhinoscopic debridement and flushing to remove fungal plaques improves the response to local treatment.
Cryptococcus neoformans is the most common feline fungal rhinitis.26 This fungus often infects the rostral nasal compartment, causing planum deformation, ulceration, and a mass effect. It has been reported to proliferate in the nasopharynx of cats and dogs.29 This fungus can also be part of a localized facial dermatitis or more systemic infection. Serologic analysis is sensitive and specific enough to provide a presumptive diagnosis.
Other fungal infections reported to cause rhinitis in the dog and cat include Rhinosporidium, Exophiala, Alternaria, Trichosporon, Blastomyces, and Histoplasma and the algae Prototheca.26
”Allergic” or Inflammatory Rhinitis
The hallmark presentation of the patient with an “allergic” rhinitis is chronic, bilateral, serous to mucopurulent or mucohemorrhagic discharge. Occasionally discharge will be unilateral, but changes in the nasal cavity are bilateral.
Allergic rhinitis is a broad term often interchanged with chronic rhinitis, chronic hyperplastic rhinitis, lymphoplasmacytic rhinitis (LPR), and eosinophilic rhinitis (ER). LPR and ER are classified based on the predominant cell type on histopathologic examination. Many chronic nasal conditions show a mix of inflammatory cell types and varying degrees of proliferative changes in the nasal mucosa. Eosinophils are rarely the predominant cell type, and ER is uncommon. Lymphocytes and plasma cells are common cell types seen on histopathologic examination, and varying numbers of neutrophils and eosinophils are present.30,31 LPR is the most common form of chronic noninfectious inflammatory rhinitis in dogs and cats. A number of studies have been performed to attempt specific cytokine profiling without uniform results. T-helper II cytokine profiles may predominate in dogs,32,33 whereas T-helper I profiles may predominate in cats.34 This information has yet to have proven helpful in the specific treatment and elimination of symptoms. Multiple infective agents have been identified in association with chronic canine and feline rhinosinusitis, but none has been significantly reproducible enough to be deemed causative.35-37
Medical management employing various forms of corticosteroids, antibiotics, and nebulization therapies is generally temporarily successful, but symptoms return with complete treatment cessation. Secondary bacterial infections commonly compound symptoms. We have had significant success using fluocinolone acetonide with dimethyl-sulfoxide (Synotic) mixed with various culture-guided parenteral antibiotics as a direct nose drop. This technique has the benefit of achieving high local steroid, antioxidant, and antibiotic concentration with minimal systemic absorption. The dimethylsulfoxide component may theoretically help medication penetration into significantly hyperplastic nasal tissues. Lastly, a liquid nose drop helps to moisturize productive nasal secretions that add to symptomatic obstruction common in these patients.
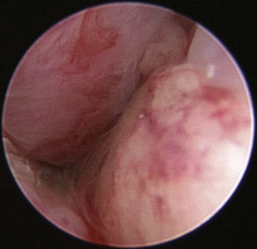
There are many nonspecific changes seen with rhinoscopic evaluation that vary from mild to severe (Figures 19-34 through 19-41). Mucosal hyperplasia, hyperemia, and accumulations of mucoid to mucopurulent secretions are typical findings. Turbinate mucosa can have a classic clubbed and hyperplastic appearance. Progression can result in generalized or polypoid mucosal proliferation or hyperplasia. In severe, chronic cases, infected and necrotic turbinate can be seen. The combination of mucosal hyperplasia and abundant accumulation of mucus compromises the air passageways and can limit the movement of the scope, especially in cats and small dogs. Inflamed tissue bleeds easily and can further decrease visibility. Site selection for biopsy is often nonspecific because most of the nasal mucosa is involved. Bacterial cultures collected at the time of examination may reveal a current secondary bacterial infection, unlikely the primary cause but nonetheless creating pathologic symptoms. Mycoplasma may be of concern in cats with recurring rhinitis.35 Biopsy and appropriate culture submission are critical for developing a specific medical plan to manage the patients’ current symptoms.
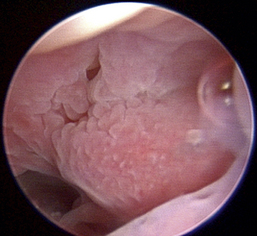
Figure 19-36 Canine severe and chronic lymphoplasmacytic rhinitis with polypoid mucosal hyperplasia.
1. Meler E., Dunn M., Lecuyer M. A retrospective study of canine persistent nasal disease: 80 cases (1998–2003). Can Vet J. 2008;49(1):71-76.
2. Demko J.L., Cohn L.A. Chronic nasal discharge in cats: 75 cases (1993-2004). J Am Vet Med Assoc. 2007;230(7):1032-1037.
3. Pomrantz J.S., Johnson L.R., Nelson R.W., et al. Comparison of serologic evaluation via agar gel immunodiffusion and fungal culture of tissue for diagnosis of nasal aspergillosis in dogs. J Am Vet Med Assoc. 2007;230(9):1319-1323.
4. Kerl M.E. Update on canine and feline fungal diseases. Vet Clin North Am Small Anim Pract. 2003;33(4):721-747.
5. Pownder S., Rose M., Crawford J. Radiographic techniques of the nasal cavity and sinuses. Clin Tech Small Anim Pract. 2006;21(2):46-54.
6. Petite A.F.B., Dennis R. Comparison of radiography and magnetic resonance imaging for evaluating the extent of nasal neoplasia in dogs. J Small Anim Pract. 2006;47(9):529-536.
7. Johnson E.G., Wisner E.R. Advances in respiratory imaging. Vet Clin North Am Small Anim Pract. 2007;37(5):879-900.
8. Miles M.S., Dhaliwal R.S., Moore M.P., et al. Association of magnetic resonance imaging findings and histologic diagnosis in dogs with nasal disease: 78 cases (2001-2004). J Am Vet Med Assoc. 2008;232(12):1844-1849.
9. Lefebvre J., Kuehn N.F., Wortinger A. Computed tomography as an aid in the diagnosis of chronic nasal disease in dogs. J Small Anim Pract. 2005;46(6):280-285.
10. Curry S.L., Cogar S.M., Cook J.L. Nonsteroidal antiinflammatory drugs: a review. J Am Anim Hosp Assoc. 2005;41:298-309.
11. Sinclair M.D. A review of the physiological effects of alpha2-agonists related to the clinical use of medetomidine in small animal practice. Can Vet J. 2003;44:885-897.
12. Allen H.S., Broussard J., Noone K. Nasopharyngeal diseases in cats: a retrospective study of 53 cases (1991-1998). J Am Anim Hosp Assoc. 1999;35(6):457-461.
13. Hunt G., Perkins C., Foster S. Nasopharyngeal disorders of dogs and cats: a review and retrospective study. Compend Contin Educ Vet. 2002;24(3):184-200.
14. Berent A.C., Kinns J., Weisse C. Balloon dilatation of nasopharyngeal stenosis in a dog. J Am Vet Med Assoc. 2006;229(3):385.
15. Coolman B.R., Marretta S.M., McKiernan B.C., et al. Choanal atresia and secondary nasopharyngeal stenosis in a dog. J Am Anim Hosp Assoc. 1998;34(6):497-501.
16. Kirberger R.M., Steenkamp G., Spotswood T.C., et al. Stenotic nasopharyngeal dysgenesis in the Dachshund: seven cases (2002-2004). J Am Anim Hosp Assoc. 2006;42(4):290-297.
17. Detweiler D.A., Johnson L.R., Kass P.H., et al. Computed tomographic evidence of bulla effusion in cats with sinonasal disease: 2001-2004. J Vet Intern Med. 2006;20(5):1080-1084.
18. Jones J.C., Ober C.P. Computed tomographic diagnosis of nongastrointestinal foreign bodies in dogs. J Am Anim Hosp Assoc. 2007;43(2):99-111.
19. Gunnarsson L., Zakrisson G., Lilliehook I., et al. Experimental infection of dogs with the nasal mite. Pneumonyssoides caninum, Vet Parasitol. 1998;77(2-3):179-186.
20. Bredal W.P. Pneumonyssoides caninum infection—a risk factor for gastric dilatation-volvulus in dogs. Vet Res Commun. 1998;22(4):225-231.
21. Gunnarsson L.K., Moller L.C., Einarsson A.M., et al. Clinical efficacy of milbemycin oxime in the treatment of nasal mite infection in dogs. J Am Anim Hosp Assoc. 1999;35:81-84.
22. Gunnarsson L., Zakrisson G., Christensson D., et al. Efficacy of selamectin in the treatment of nasal mite (Pneumonyssoides caninum) infection in dogs. J Am Anim Hosp Assoc. 2004;40(5):400-404.
23. Wehner A., Crochik S., Howerth E.W., et al. Diagnosis and treatment of blastomycosis affecting the nose and nasopharynx of a dog. J Am Vet Med Assoc. 2008;233(7):1112-1126.
24. Malik R., Dill-Macky E., Martin P., et al. Cryptococcosis in dogs: a retrospective study of 20 consecutive cases. J Med Vet Mycol. 1995;33(5):291-297.
25. Benitah N. Canine nasal aspergillosis. Clin Tech Small Anim Pract. 2006;21(2):82-88.
26. Wolf A.M. Fungal diseases of the nasal cavity of the dog and cat. Vet Clin North Am Small Anim Pract. 1992;22(5):1119-1132.
27. Saunders J.H., Clercx C., Snaps F.R., et al. Radiographic, magnetic resonance imaging, computed tomographic, and rhinoscopic features of nasal aspergillosis in dogs. J Am Vet Med Assoc. 2004;225(11):1703-1712.
28. Peeters D., Clercx C. Update on canine sinonasal aspergillosis. Vet Clin North Am Small Anim Pract. 2007;37(5):901-916.
29. Malik R., Martin P., Wigney D.I., et al. Nasopharyngeal cryptococcosis. Aust Vet J. 1997;75(7):483-488.
30. Windsor R.C., Johnson L.R. Canine chronic inflammatory rhinitis. Clin Tech Small Anim Pract. 2006;21(2):76-81.
31. Michiels L., Day M.J., Snaps F., et al. A retrospective study of non-specific rhinitis in 22 cats and the value of nasal cytology and histopathology. J Feline Med Surg. 2003;5(5):279-285.
32. Kurata K., Maeda S., Yasunaga S., et al. Immunological findings in 3 dogs clinically diagnosed with allergic rhinitis. J Vet Med Sci. 2004;66(1):25-29.
33. Peeters D., Peters I.R., Helps C.R., et al. Distinct tissue cytokine and chemokine mRNA expression in canine sinonasal aspergillosis and idiopathic lymphoplasmacytic rhinitis. Vet Immunol Immunopathol. 2007;117(1-2):95-105.
34. Veir J.K., Lappin M.R., Dow S.W. Evaluation of a novel immunotherapy for treatment of chronic rhinitis in cats. J Feline Med Surg. 2006;8(6):400-411.
35. Johnson L.R., Foley J.E., De Cock H.E., et al. Assessment of infectious organisms associated with chronic rhinosinusitis in cats. J Am Vet Med Assoc. 2005;227(4):579-585.
36. Windsor R.C., Johnson L.R., Sykes J.E., et al. Molecular detection of microbes in nasal tissue of dogs with idiopathic lymphoplasmacytic rhinitis. J Vet Intern Med. 2006;20(2):250-256.
37. Hawkins E.C., Johnson L.R., Guptill L., et al. Failure to identify an association between serologic or molecular evidence of Bartonella infection and idiopathic rhinitis in dogs. J Am Vet Med Assoc. 2008;233(4):597-599.
Palliative Treatment of Nasal Tumors Using Rhinoscopic-Assisted Transnares Curettage
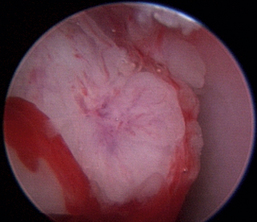
Tumors of the nasal cavity and nasopharynx account for approximately 1% to 2% of all canine and feline neoplasms.1 Benign masses are uncommon. The majority of nasal tumors are malignant, and a variety of germinal origins are reported2,3 (Figures 19-42 through 19-49). Most nasal tumors are primary,2 but metastatic nasal tumors have been reported.3,4 Local infiltration is common. Rostral cerebral metastasis has been reported with a variety of sinonasal tumors.5 Distant metastases of primary nasal tumors are uncommon but have been reported and generally observed later in the disease process.6 Numerous causes have been proposed. Etiologic factors shown to be associated with sinonasal tumor development include exposure to household tobacco smoke,7 coal and kerosene heater exhausts,8 mesaticephaly, and dolichocephaly.9 Generally, complete primary removal of a symptomatic tumor is not possible due to space limitations, sensitivity of local anatomy, and local infiltration by the tumor.9 The prognosis associated with malignant nasal tumors is often poor due to the tumor behavior, inaccessibility of the site to primary treatment, and the significant impact of nasal airflow obstruction on patient quality of life.
Because of the characteristics of nasal tumors, treatment options are often limited by equipment specialization, cost, and technical skill. Current treatment options include rhinotomy, radiation therapy (RT), rhinoscopic shaver debridement, rhinoscopic laser ablation, and chemotherapy. Rhinotomy has been described by both ventral and dorsal nasal cavity approaches,10 is considered traumatic, and has a relatively high complication rate. Rhinotomy alone has been shown to marginally increase survival times in dogs with sinonasal tumors when compared with untreated tumors.11,12 Rhinotomy with full turbinate exenteration and RT has been shown to significantly prolong survival but carries a high risk of procedural complications.13 RT in various applications has been shown to be an effective treatment in numerous studies. RT with or without cytoreduction has been shown to provide a longer disease-free interval and is the treatment of choice.6,13-17 RT without surgical cytoreduction does not relieve nasal obstruction immediately. There are numerous documented short- and long-term side effects associated with this treatment.13-17 Stereotactic radiosurgery is considered to be at the forefront of RT.18 These machines are able to deliver a very high dose of radiation to a specific site with minimal collateral tissue damage.18 Unfortunately, the limited availability and expense of such technology removes it from common practice, even in most major metropolitan areas. Rhinoscopic debridement of sinonasal tumors with the use of an arthroscopic power shaver has been described.10 Rhinoscopic diode laser ablation of nasal tumors is limited by availability and technical expertise and is a lengthy procedure.19,20 The beneficial effect of systemic chemotherapy has been reported with variable results.21-23 There may be a significant difference in predicted chemotherapy response among the nasal tumor germinal origins.22 No single chemotherapeutic regimen has currently been proven to be reliably effective with all nasal tumors. Palliative treatment with NSAIDs has been advocated after the identification of cyclooxygenase enzyme expression by some nasal tumors.23-25 There are currently no controlled studies identifying a benefit in survival of patients with nasal tumors treated with long-term NSAIDs only. NSAIDs in combination with chemotherapy have been shown to have a beneficial effect on survival in dogs.22 Because of factors such as treatment availability, cost, efficacy, invasiveness, and incident treatment complications, many clients desire an option other than RT, rhinotomy, or rhinoscopic-assisted laser ablation.
Transnares curettage (TNC) helps provide relief of nasal obstruction and tumor-associated epistaxis and provides generous tissue samples for histopathologic confirmation in undiagnosed cases. With the use of a variable combination of radiographic, rhinoscopic, and nasal flushing localization techniques, tumor tissue is identified and removed through the nostrils with a combination of common surgical instruments. Abnormal tissue is removed via traction avulsion directly through the nostrils or traumatically loosened and flushed through the nasopharynx out of the mouth. The procedure was adapted from two articles written by Withrow et al in the 1970s and 1980s and has been used by one of the authors for 20 years.26-28 The introduction of rhinoscopy to the procedure has improved visualization of abnormal tissues and has enabled more thorough tumor removal. Removal of the abnormal tissue restores nasal breathing, decompresses sinus congestion, and eliminates significant ongoing paraneoplastic epistaxis. The quality of life is improved, and the invasiveness is minimal.
Preoperative evaluation of the patient with a nasal tumor consists of a complete blood count, serum biochemistry profile, urinalysis, PT and PTT, three-view thoracic radiographs, and nasal imaging of choice (e.g., rhinoscopy, radiographs, CT, or MRI). Von Willebrand’s factor analysis or buccal mucosal bleeding times should be performed in predisposed breeds. TNC is contraindicated in the coagulopathic or thrombocytopenic or thrombocytopathic patient.
Although TNC is less invasive compared with rhinotomy, adequate preoperative neuroleptanalgesia, intraoperative and postoperative analgesia, and antiinflammatory treatments are recommended based on the patient’s preoperative evaluation. Abrupt and forceful sneezing and nasopharyngeal reflexive movements can be observed even in the adequately anesthetized patient. These forceful movements can greatly affect procedural ease, as well as induce patient trauma and cause equipment damage. Bilateral nerve blockade at the level of the infraorbital foramen can greatly reduce the sneeze reflex, and a combination of lidocaine and bupivacaine is recommended in dogs and at reduced doses in cats. Intraoperative and perioperative, culture-guided antibiotic therapy is recommended because of the high incidence of secondary bacterial rhinitis in the patient with a nasal tumor. Endotracheal tube cuff evaluation should be rechecked before any flushing because of the potential for iatrogenic flushing or aspiration of infected nasal secretions into the lungs.
Rigid, integrated endoscopes are recommended for rhinoscopy. A 1.9-mm rigid endoscope is used in cats and small dogs, and a 2.7-mm rigid endoscope is used in dogs that weigh more than 15 kg. The length should be at least 17 cm for large dogs. A 4.0-mm rigid endoscope may be needed in some dogs more than 45 kg. The integrated scopes provide a port for constant fluid infusion. This is necessary for flushing secretions and blood away from the tip of the scope and nasal lumen so that adequate anatomic visualization is provided.
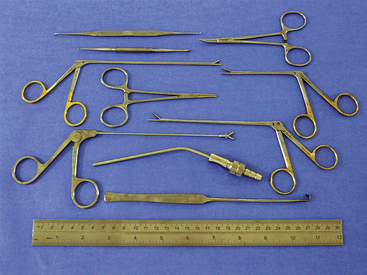
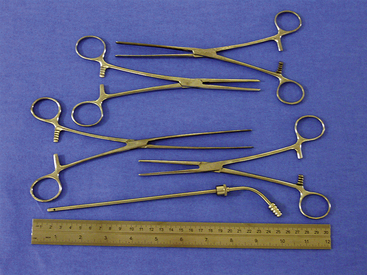
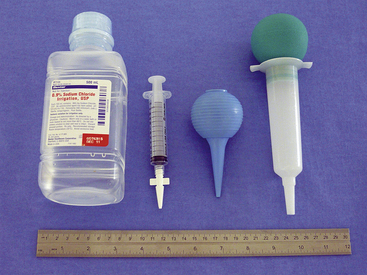
Instruments used for TNC vary depending on the size of the animal and conformation of the nasal cavity (Figures 19-50 and 19-51). Kelly, mosquito, and Carmalt hemostatic forceps and various sized rongeurs work well for grasping and retrieving tissue. Curettes and a Freer periosteal elevator are used to loosen tissues attached to bone. Straight Doyen tissue forceps (dogs) and Noyes alligator forceps (small dogs and cats) are used to clear the choanae. Frazier suction tips are used for curettage as well as for suction. The largest diameter suction tip catheter the nasal cavity will accommodate should be used to limit clogging of the catheter with tissue and debris. Flushing is accomplished in larger animals with the use of a bulb syringe or a 60-mL catheter-tipped syringe. In cats and small dogs, a 12-mL syringe with a Christmas tree adapter works well (Figure 19-52).
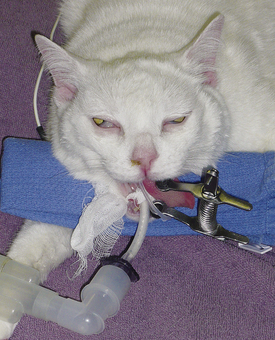
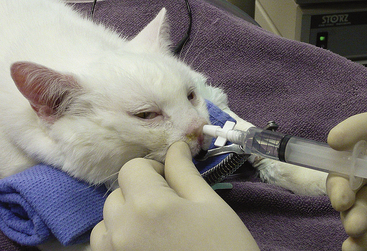
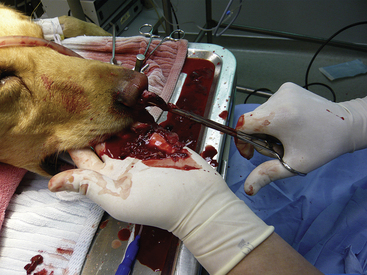
Patients are placed in a sternal recumbent position. The cuffed endotracheal tube is secured to the mandible. A rolled towel is placed beneath the animal’s neck to point the muzzle in a ventral direction, and an oral speculum is placed to hold the mouth open (Figure 19-53). Fluid and tissue will then tend to run out of the nostrils and mouth rather than collect around the larynx. No gauze is stuffed in the oropharynx. This impedes the movement of fluid from the nasopharynx into the mouth. The animal’s front legs are pulled laterally or covered with plastic to prevent soiling. Again, endotracheal tube cuff evaluation should be rechecked before any flushing because of the potential for iatrogenic flushing or aspiration of infected nasal secretions into the lungs.
The first step is to flush each nostril (Figure 19-54). This gives a good indication of the degree of obstruction. A bulb syringe or syringe with adapter is seated in the nostril, and the nasal planum is squeezed on each side to provide an effective seal. Normally, fluid should move easily through the nasal cavity into the nasopharynx and roll out the mouth. Resistance varies with the degree of obstruction. The nasal cavity is then evaluated with the endoscope. Nasal tumors typically are unilateral, but both passages should be carefully examined to find any tumor extending across the midline. Always evaluate the patency of the choanae, and pass the scope into the nasopharynx. The presence of neoplastic tissue may prevent visualization of the choanae on one or both sides.
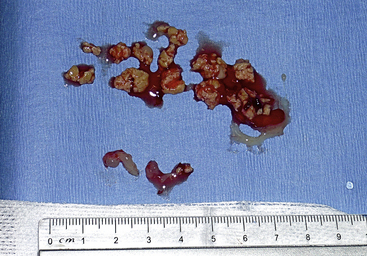
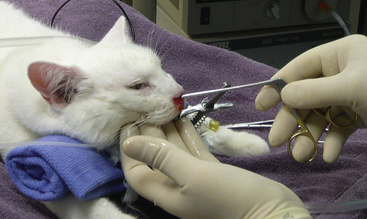
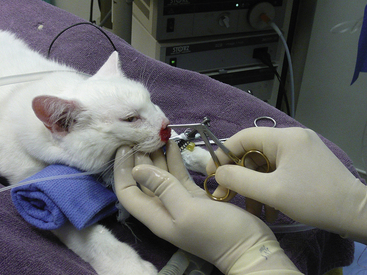
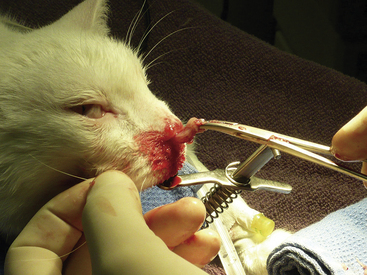
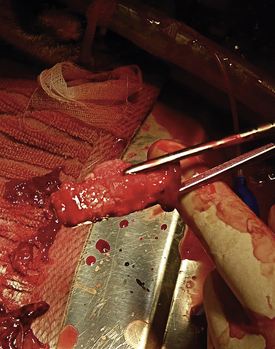
Curettage is performed after complete rhinoscopic evaluation. Combining rhinoscopic findings with the results of the radiographs, CT, or MRI gives the surgeon good localization of the mass. Curettage is a blind procedure done by feel and spatial approximation estimates supported by imaging results. Knowledge of nasal anatomy is essential. A blunt instrument is passed through the nostril on the affected side (Figure 19-55). Slide the instrument along the hard palate near the midline until it can be palpated sitting above the soft palate (Figure 19-56). A Doyen forcep in dogs or a Noyes alligator forcep in cats is used for this step. Moving the instrument along the floor of the nasal cavity prevents inadvertent puncturing of the cribriform plate. Vigorously move the probe vertically, horizontally, and circularly at the level of the choanae. This maneuver loosens obstructive tissue. Once the probe moves easily into the nasopharynx, flushing is performed to expel dislodged tissues. Loose fragments of tissue will be flushed around the soft palate and out of the mouth and can often provide adequate histopathologic specimens in cases with open diagnoses. Once the choanae are clear, curettage continues in the nasal passage. Tissue is grasped with hemostats or rongeurs and is pulled out through the nostril (Figure 19-57). Abnormal tissue is removed with minimal force; traction on healthy tissue is met with significant resistance and should signal the surgeon to proceed further with caution in that location. Continue flushing as tissue is being retrieved. This also provides excellent biopsy samples. Check the progress of tissue debridement with rhinoscopy. Continue curettage, flushing, and rhinoscopic inspection until the bulk of abnormal tissue has been removed. Rhinoscopic assistance is optimal, but this procedure can be performed with only the benefit of serial flushing to mark reestablishment of nasal patency (Figures 19-58 and 19-59). Flushing resistance should be minimal after adequate curettage has been completed. This predicts restoration of nasal breathing in the postoperative period. Remove any tissue associated with the cribriform plate with extreme caution. Use a curette or angled Frazier suction tip to debride the maxillary recess. Visualization of the frontal sinus is difficult. Direct trephination of the frontal sinus may be necessary to remove abnormal tissue that extends into this space. The need for this intervention may be determined in advance based on the results of radiographic imaging. Large amounts of tumor tissue can be debrided by this process and will immediately restore nasal airflow patency and dramatically cytoreduce the tumor burden before follow-up therapies (Figures 19-60 through 19-62).
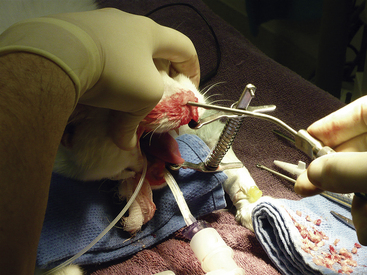
Figure 19-58 Periodic flushing and suctioning are performed to clear the nasal passage and determine the end of debridement.
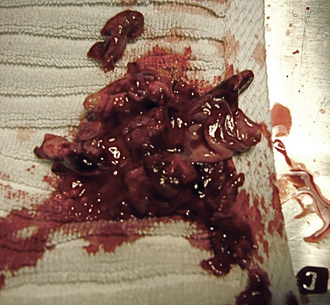
Figure 19-62 Large amounts of tumor can be safely removed, which reestablishes nasal airflow and dramatically improves quality of life.
Rhinoscopic sequence imaging of TNC debridement of a feline caudal nasal and nasopharyngeal adenocarcinoma is depicted in Figures 19-63 through 19-69.
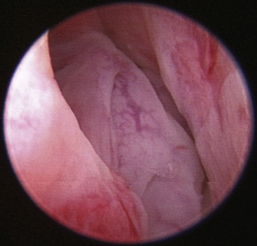
Figure 19-63 Transnares curettage sequence: feline nasal adenocarcinoma of the right caudal nasal cavity and nasopharynx, evaluation of the tumor extent, tumor debridement, and reevaluation of lumen patency after the procedure.
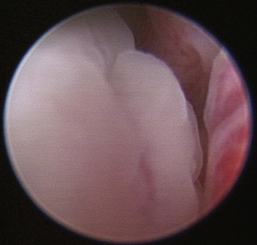
Figure 19-65 Full mass obstruction is present, filling the nasopharynx and preventing air flow bilaterally.
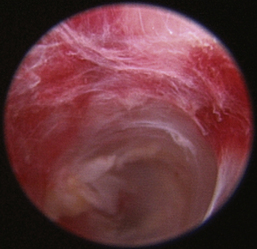
Figure 19-69 The nasal passage and nasopharynx is clear; restoration of patient nasal breathing has been accomplished with minimal trauma.
Small fragments of visually abnormal tissue may persist despite repeated efforts to remove them. Running a separate instrument along the side of the scope or passing a diode laser down the biopsy port of the scope allows visualization and removal of this tissue. Vigorous curettage and flushing may dislodge these fragments. Considering the failure of rhinotomy and complete extirpation of the tumor to increase the disease-free interval, removal of every fragment of tumor is a moot point. This is a palliative procedure. Removing the majority of the tumor eliminates bleeding, and clearance of the choanae and nasopharynx restores nasal breathing.
Epistaxis will appear significant throughout this procedure. This is expected and is rarely enough to result in anemia requiring transfusion. It is extremely rare to encounter life-threatening hemorrhaging. We have observed three cases in a 10-year period in which hemorrhaging was acutely massive and life-threatening. Retrospectively, maxilla infiltration with compromise of the maxillary artery and rostral branches was suspected. In the event acute severe hemorrhaging is observed, stabilization maneuvers such as unilateral carotid compression, unilateral carotid ligation, Foley catheter or small endotracheal tube intranasal insertion, and bulb expansion can be implemented. Most tumor-associated epistaxis will resolve spontaneously with appropriate debridement of the neoplastic tissue. Postoperative epistaxis is expected for 5 to 7 days. We have found no need for or benefit of nasal instillation of vasoconstrictors. Most patients are discharged with a presumptive antibiotic (pending culture results, if applicable), opioid analgesic, and steroid or NSAID (depending on their preoperative assessment and drug safety profiles) within 24 to 48 hours of the procedure.
We have observed surgical remission times of single-digit months (1 to 9 months) for 80% of patients receiving TNC only (unpublished data). The procedure can be repeated as needed based on the patient disease progression. The disease of patients with bone infiltration tends to be less responsive than does the disease in patients with primarily intraluminal recurrence. We have observed multiple dogs and cats with low-grade adenocarcinoma and low-grade chondrosarcoma experience survival times of 2 to 4 years treated with intermittent TNC and palliative medications alone (generally NSAIDs and antibiotics, as clinically indicated). We have combined and routinely recommend follow-up RT in all patients that are able to pursue additional treatment. Survival times are in tabulation after a 5-year retrospective of both dogs and cats having TNC, with and without follow-up RT.
1. Madewell B.R., Priester W.A., Gillette E.L., et al. Neoplasms of the nasal passages and paranasal sinuses in domestic animals as reported by 13 veterinary colleges. Am J Vet Res. 1976;137:851-856.
2. Cotchin E. Spontaneous neoplasms of the upper respiratory tract in animals. In: Muir C.S., Shanmugaratnam K., editors. Cancer of the nasopharynx. Flushing, N.Y.: Medical Examination Publishing, 1967.
3. Patniak A.K. Canine sinonasal neoplasms: clinicopathological study of 285 cases. J Am Anim Hosp Assoc. 1989;25:103-114.
4. Affolter V.K., Moore P.F. Localized and disseminated histiocytic sarcoma of dendritic cell origin in dogs. Vet Pathol. 2002;39(1):74-83.
5. Snyder J.M., Lipitz L., Skorupski K.A., et al. Secondary intracranial neoplasia in the dog: 177 cases (1986-2003). J Vet Intern Med. 2008;22(1):172-177.
6. McEntee M.C., Page R.L., Heidner G.L., et al. A retrospective study of 27 dogs with intranasal neoplasms treated with cobalt radiation. Vet Radiol. 1991;32(3):135-139.
7. Reif J.S., Bruns C., Lower K.S. Cancer of the nasal cavity and paranasal sinuses and exposure to environmental tobacco smoke in pet dogs. Am J Epidemiol. 1998;147(5):488-492.
8. Bukowski J.A., Wartenberg D., Goldschmidt M. Environmental causes for sinonasal cancers in pet dogs, and their usefulness as sentinels of indoor cancer risk. J Toxicol Environ Health A. 1998;54(7):579-591.
9. Morgan J.P., Suter P.F., O’Brien T.R., et al. Tumors in the nasal cavity of the dog: a radiographic study. Vet Radiol Ultrasound. 1972;13(1):18-26.
10. Degner D.A. Surgical considerations of nasal discharge. Clin Tech Small Anim Pract. 2006;21(2):64-68.
11. Laing E.J., Binnington A.G. Surgical therapy of canine nasal tumors: a retrospective study (1982-1986). Can Vet J. 1988;29(10):809-813.
12. Henry C.J., Brewer W.G.Jr., Tyler J.W., et al. Survival in dogs with nasal adenocarcinoma: 64 cases (1981-1995). J Vet Intern Med. 1998;12(6):436-439.
13. Adams W.M., Bjorling D.E., McAnulty J.E., et al. Outcome of accelerated radiotherapy alone or accelerated radiotherapy followed by exenteration of the nasal cavity in dogs with intranasal neoplasia: 53 cases (1990-2002). J Am Vet Med Assoc. 2005;227(6):936-941.
14. LaDue T.A., Dodge R., Page R.L., et al. Factors influencing survival after radiotherapy of nasal tumors in 130 dogs. Vet Radiol Ultrasound. 1999;40(3):312-317.
15. Mellanby R.J., Stevenson R.K., Herrtage M.E., et al. Long-term outcome of 56 dogs with nasal tumours treated with four doses of radiation at intervals of seven days. Vet Rec. 2002;151(9):253-257.
16. Gieger T., Rassnick K., Siegel S., et al. Palliation of clinical signs in 48 dogs with nasal carcinomas treated with coarse-fraction radiation therapy. J Am Anim Hosp Assoc. 2008;44(3):116-123.
17. Northrup N.C., Etue S.M., Ruslander D.M., et al. Retrospective study of orthovoltage radiation therapy for nasal tumors in 42 dogs. J Vet Intern Med. 2001;15(3):183-189.
18. Gordon I.K., Kent M.S. Veterinary radiation oncology: technology, imaging, intervention and future applications. Cancer Ther. 2008;6(1):167-176.
19. Berger N., Eeg P.H. Veterinary laser surgery. Malden, Mass: Blackwell Publishing; 2006.
20. McCarthy Timothy. Veterinary endoscopy for the small animal practitioner. St Louis: Saunders; 2004.
21. Henry C.J., Brewer W.G.Jr., Tyler J.W., et al. Survival in dogs with nasal adenocarcinoma: 64 cases (1981-1995). J Vet Intern Med. 1998;12(6):436-439.
22. Langova V., Mutsaers A.J., Phillips B., et al. Treatment of eight dogs with nasal tumours with alternating doses of doxorubicin and carboplatin in conjunction with oral piroxicam. Aust Vet J. 2004;82(11):676-680.
23. Hahn K.A., Knapp D.W., Richardson R.C., et al. Clinical response of nasal adenocarcinoma to cisplatin chemotherapy in 11 dogs. J Am Vet Med Assoc. 1992;200:355-357.
24. Borzacchiello G., Paciello O., Papparella S. Expression of cyclooxygenase-1 and -2 in canine nasal carcinomas. J Comp Pathol. 2004;131(1):70-76.
25. Kleiter M., Malarkey D.E., Ruslander D.E., et al. Expression of cyclooxygenase-2 in canine epithelial nasal tumors. Vet Radiol Ultrasound. 2004;45(3):255-260.
26. MacEwen G., Withrow S., Patnaik A.K. Nasal tumors in the dog: retrospective evaluation of diagnosis, prognosis and treatment. J Am Vet Med Assoc. 1977;170(1):45-48.
27. Withrow S.J., Susaneck S.J., Macy D.W., et al. Aspiration and punch biopsy techniques for nasal tumors. J Am Anim Hosp Assoc. 1985;21:551-554.
28. Withrow S.J. Diagnostic and therapeutic nasal flush in small animals. J Am Anim Hosp Assoc. 1977;13:704-707.