Heart and Neck Vessels
Outline
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
• Physical Examination Summary Checklist
• Quick Assessment for Common Conditions
Structure and Function
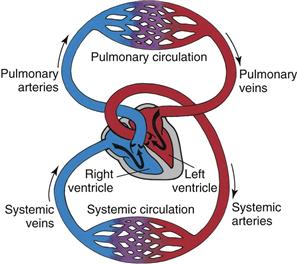
The cardiovascular system consists of the heart (a muscular pump) and the blood vessels. The blood vessels are arranged in two continuous loops, the pulmonary circulation and the systemic circulation (Fig. 19-1). When the heart contracts, it pumps blood simultaneously into both loops.
Position and Surface Landmarks
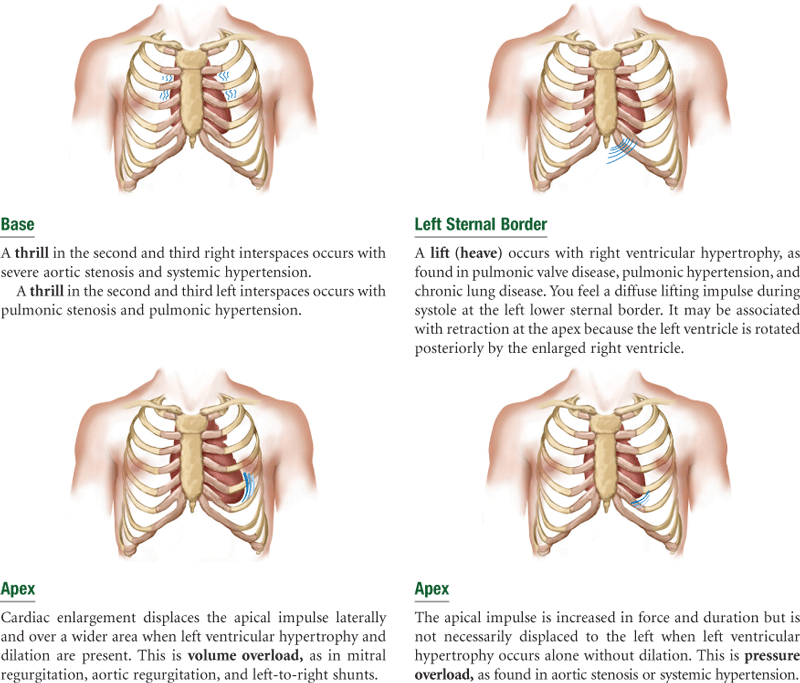
The precordium is the area on the anterior chest directly overlying the heart and great vessels (Fig. 19-2). The great vessels are the major arteries and veins connected to the heart. The heart and great vessels are located between the lungs in the middle third of the thoracic cage (mediastinum). The heart extends from the 2nd to 5th intercostal space and from the right border of the sternum to the left midclavicular line.
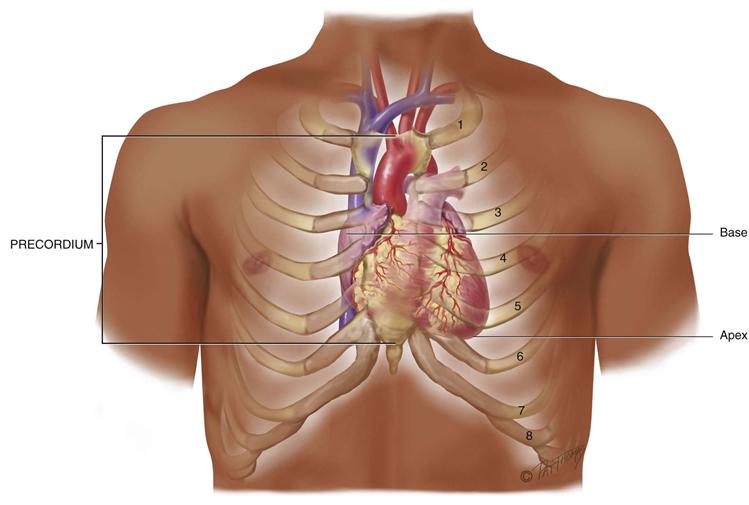
19-2 Copyright © (2006) © Pat Thomas, 2006.
Think of the heart as an upside-down triangle in the chest. The “top” of the heart is the broader base, and the “bottom” is the apex, which points down and to the left (Fig. 19-3). During contraction, the apex beats against the chest wall, producing an apical impulse. This is palpable in most people, normally at the fifth intercostal space, 7 to 9 cm from the midsternal line.
19-3 Copyright © (2006) © Pat Thomas, 2006.
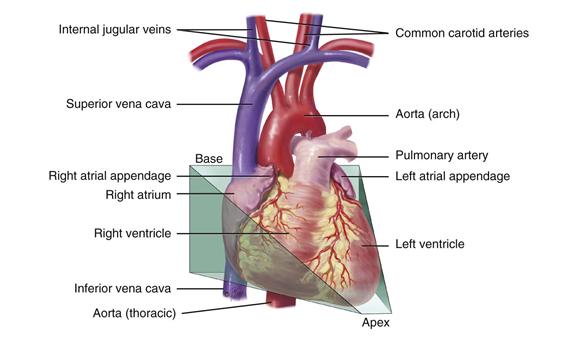
Inside the body, the heart is rotated so that its right side is anterior and its left side is mostly posterior. Of the heart’s four chambers, the right ventricle forms the greatest area of anterior cardiac surface. The left ventricle lies behind the right ventricle and forms the apex and slender area of the left border. The right atrium lies to the right and above the right ventricle and forms the right border. The left atrium is located posteriorly, with only a small portion, the left atrial appendage, showing anteriorly.
The great vessels lie bunched above the base of the heart. The superior and inferior vena cava return unoxygenated venous blood to the right side of the heart. The pulmonary artery leaves the right ventricle, bifurcates, and carries the venous blood to the lungs. The pulmonary veins return the freshly oxygenated blood to the left side of the heart, and the aorta carries it out to the body. The aorta ascends from the left ventricle, arches back at the level of the sternal angle, and descends behind the heart.
Heart Wall, Chambers, and Valves
The heart wall has numerous layers. The pericardium is a tough, fibrous, double-walled sac that surrounds and protects the heart (see its cut edge in Fig. 19-4). It has two layers that contain a few milliliters of serous pericardial fluid. This ensures smooth, friction-free movement of the heart muscle. The pericardium is adherent to the great vessels, esophagus, sternum, and pleurae and is anchored to the diaphragm. The myocardium is the muscular wall of the heart; it does the pumping. The endocardium is the thin layer of endothelial tissue that lines the inner surface of the heart chambers and valves.
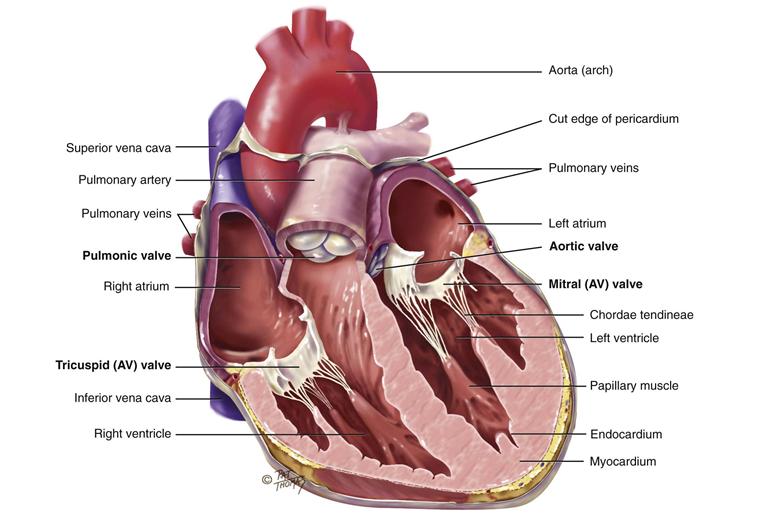
19-4 Copyright © (2006) © Pat Thomas, 2006.
The common metaphor is to think of the heart as a pump. But consider that the heart is actually two pumps; the right side of the heart pumps blood into the lungs, and the left side of the heart simultaneously pumps blood into the body. The two pumps are separated by an impermeable wall, the septum. Each side has an atrium and a ventricle. The atrium (Latin for “anteroom”) is a thin-walled reservoir for holding blood, and the thick-walled ventricle is the muscular pumping chamber. (It is common to use the following abbreviations to refer to the chambers: RA, right atrium; RV, right ventricle; LA, left atrium; and LV, left ventricle.)
The four chambers are separated by swinging-door–like structures, called valves, whose main purpose is to prevent backflow of blood. The valves are unidirectional; they can open only one way. The valves open and close passively in response to pressure gradients in the moving blood.
There are four valves in the heart (see Fig. 19-4). The two atrioventricular (AV) valves separate the atria and the ventricles. The right AV valve is the tricuspid, and the left AV valve is the bicuspid or mitral valve (so named because it resembles a bishop’s mitred cap). The valves’ thin leaflets are anchored by collagenous fibers (chordae tendineae) to papillary muscles embedded in the ventricle floor. The AV valves open during the heart’s filling phase, or diastole, to allow the ventricles to fill with blood. During the pumping phase, or systole, the AV valves close to prevent regurgitation of blood back up into the atria. The papillary muscles contract at this time, so that the valve leaflets meet and unite to form a perfect seal without turning themselves inside out.
The semilunar (SL) valves are set between the ventricles and the arteries. Each valve has three cusps that look like half moons. The SL valves are the pulmonic valve in the right side of the heart and the aortic valve in the left side of the heart. They open during pumping, or systole, to allow blood to be ejected from the heart.
Note: There are no valves between the vena cava and the right atrium nor between the pulmonary veins and the left atrium. For this reason, abnormally high pressure in the left side of the heart gives a person symptoms of pulmonary congestion, and abnormally high pressure in the right side of the heart shows in the neck veins and abdomen.
Direction of Blood Flow
Think of an unoxygenated red blood cell being drained downstream into the vena cava. It is swept along with the flow of venous blood and follows the route illustrated in Fig. 19-5.
1. From liver to right atrium (RA) through inferior vena cava
Superior vena cava drains venous blood from the head and upper extremities
From RA, venous blood travels through tricuspid valve to right ventricle (RV)
2. From RV, venous blood flows through pulmonic valve to pulmonary artery
Pulmonary artery delivers unoxygenated blood to lungs
Pulmonary veins return fresh blood to left atrium (LA)
4. From LA, arterial blood travels through mitral valve to left ventricle (LV)
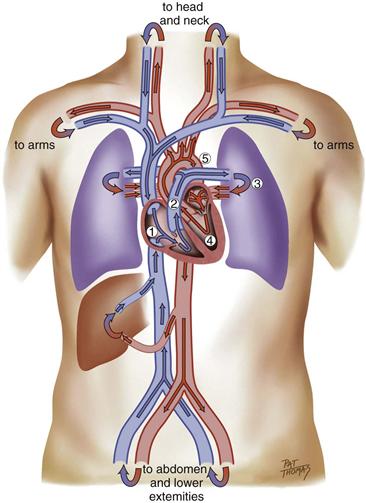
19-5
Remember that the circulation is a continuous loop. The blood is kept moving along by continually shifting pressure gradients. The blood flows from an area of higher pressure to one of lower pressure.
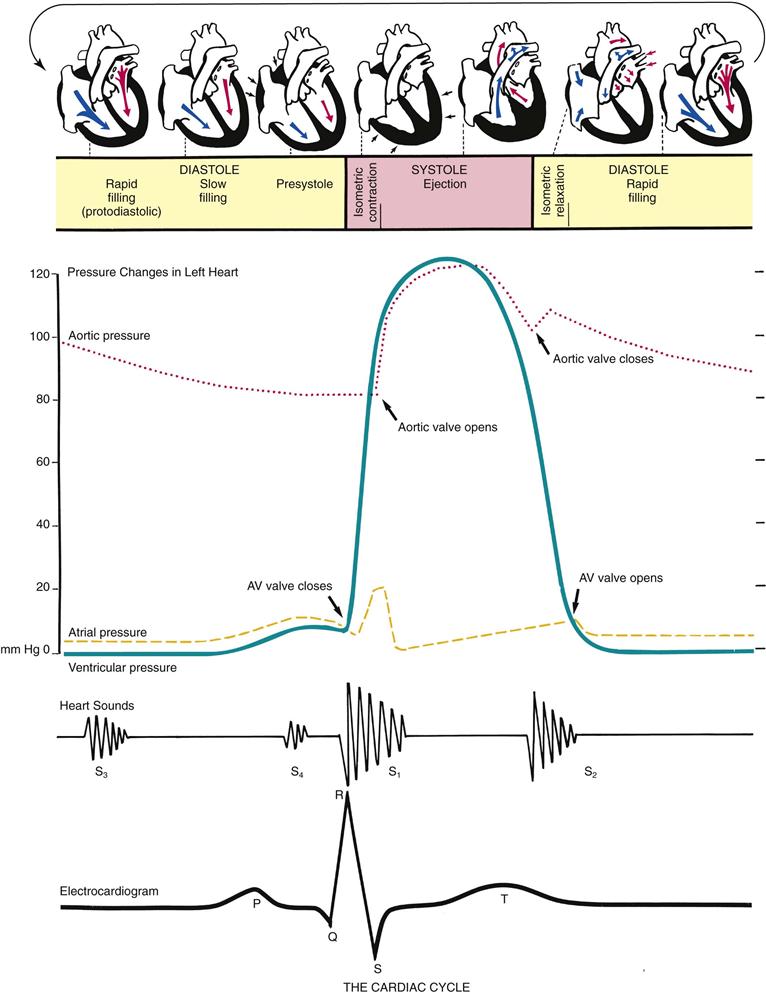
Cardiac Cycle
The rhythmic movement of blood through the heart is the cardiac cycle. It has two phases, diastole and systole. In diastole, the ventricles relax and fill with blood. This takes up two thirds of the cardiac cycle. The heart’s contraction is systole. During systole, blood is pumped from the ventricles and fills the pulmonary and systemic arteries. This is one third of the cardiac cycle.
Diastole
In diastole, the ventricles are relaxed and the AV valves (i.e., the tricuspid and mitral) are open (Fig. 19-6). (Opening of the normal valve is acoustically silent.) The pressure in the atria is higher than that in the ventricles, so blood pours rapidly into the ventricles. This first passive filling phase is called early or protodiastolic filling.
19-6
Toward the end of diastole, the atria contract and push the last amount of blood (about 25% of stroke volume) into the ventricles. This active filling phase is called presystole, or atrial systole, or sometimes the “atrial kick.” It causes a small rise in left ventricular pressure. (Note that atrial systole occurs during ventricular diastole, a confusing but important point.)
Systole
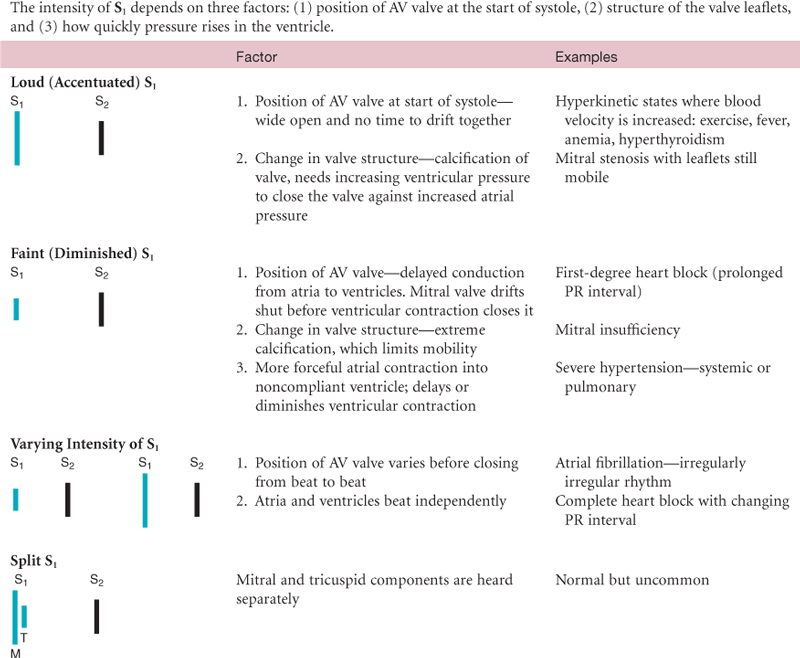
Now so much blood has been pumped into the ventricles that ventricular pressure is finally higher than that in the atria, so the mitral and tricuspid valves swing shut. The closure of the AV valves contributes to the first heart sound (S1) and signals the beginning of systole. The AV valves close to prevent any regurgitation of blood back up into the atria during contraction.
For a very brief moment, all four valves are closed. The ventricular walls contract. This contraction against a closed system works to build pressure inside the ventricles to a high level (isometric contraction). Consider first the left side of the heart. When the pressure in the ventricle finally exceeds pressure in the aorta, the aortic valve opens and blood is ejected rapidly.
After the ventricle’s contents are ejected, its pressure falls. When pressure falls below pressure in the aorta, some blood flows backward toward the ventricle, causing the aortic valve to swing shut. This closure of the semilunar valves causes the second heart sound (S2) and signals the end of systole.
Diastole Again
Now all four valves are closed and the ventricles relax (called isometric or isovolumic relaxation). Meanwhile, the atria have been filling with blood delivered from the lungs. Atrial pressure is now higher than the relaxed ventricular pressure. The mitral valve drifts open, and diastolic filling begins again.
Events in the Right and Left Sides
The same events are happening at the same time in the right side of the heart, but pressures in the right side of the heart are much lower than those of the left side because less energy is needed to pump blood to its destination, the pulmonary circulation. Also, events occur just slightly later in the right side of the heart because of the route of myocardial depolarization. As a result, two distinct components to each of the heart sounds exist, and sometimes you can hear them separately. In the first heart sound, the mitral component (M1) closes just before the tricuspid component (T1). And with S2, aortic closure (A2) occurs slightly before pulmonic closure (P2).
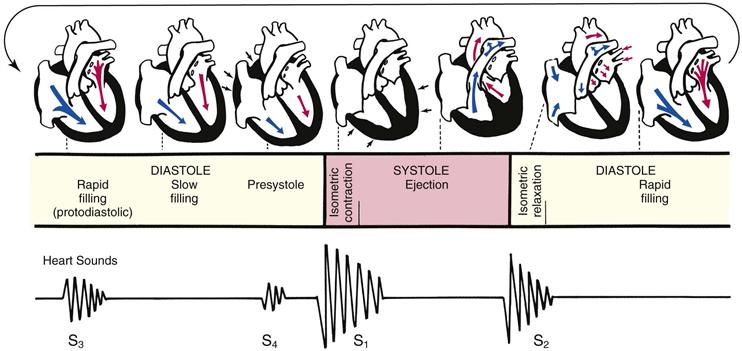
Heart Sounds
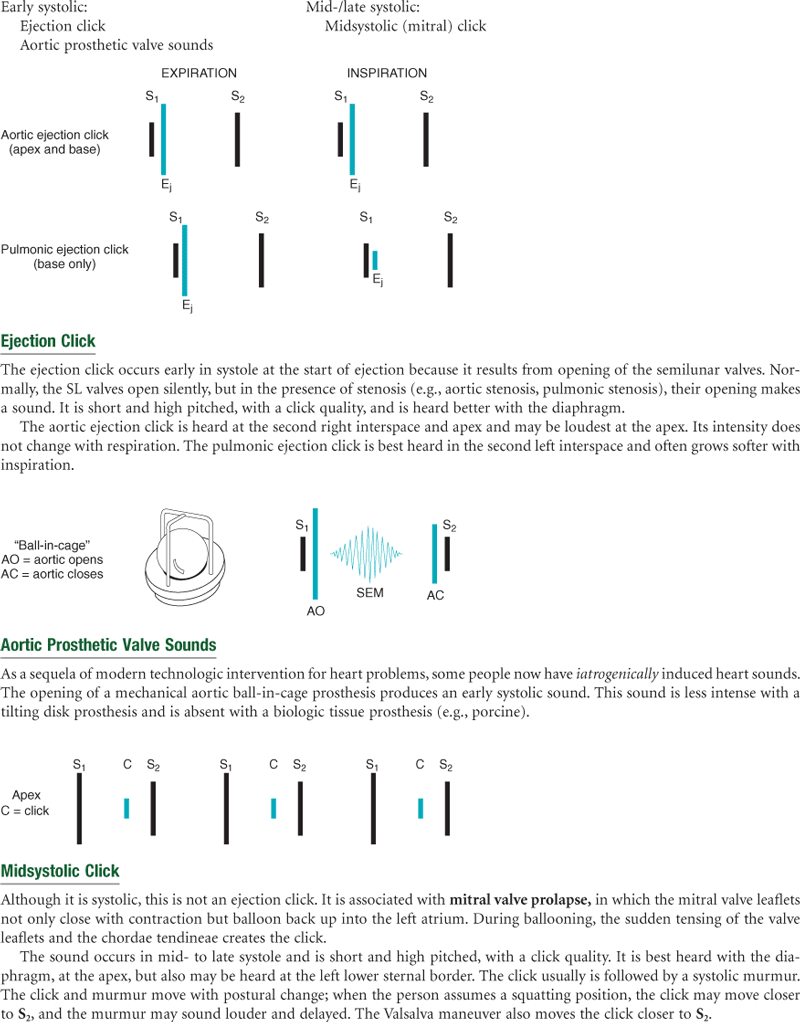
Events in the cardiac cycle generate sounds that can be heard through a stethoscope over the chest wall. These include normal heart sounds and, occasionally, extra heart sounds and murmurs (Fig. 19-7).
19-7
Normal Heart Sounds
The first heart sound (S1) occurs with closure of the AV valves and thus signals the beginning of systole. The mitral component of the first sound (M1) slightly precedes the tricuspid component (T1), but you usually hear these two components fused as one sound. You can hear S1 over all the precordium, but usually it is loudest at the apex.
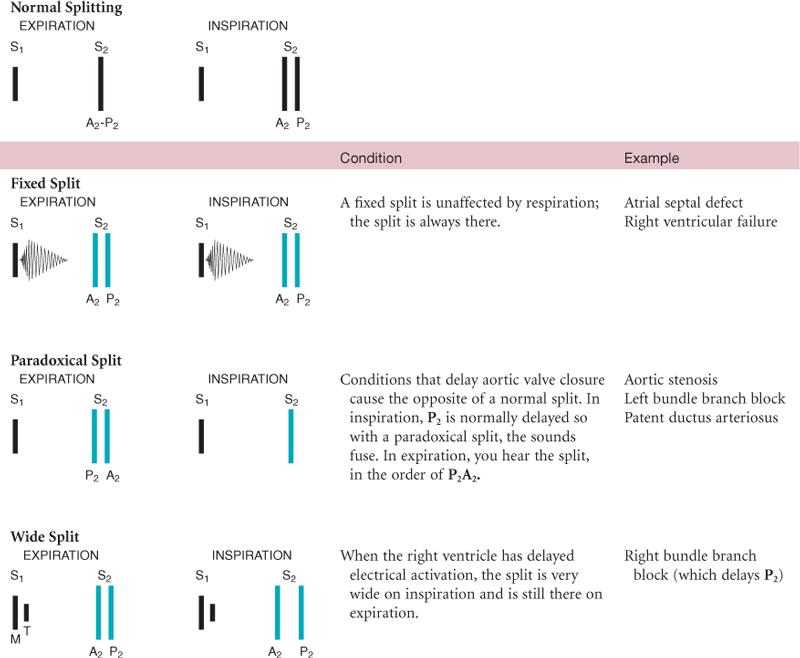
The second heart sound (S2) occurs with closure of the semilunar valves and signals the end of systole. The aortic component of the second sound (A2) slightly precedes the pulmonic component (P2). Although it is heard over all the precordium, S2 is loudest at the base.
Effect of Respiration
The volume of right and left ventricular systole is just about equal, but this can be affected by respiration. To learn this, consider the phrase:

That means that during inspiration, intrathoracic pressure is decreased. This pushes more blood into the vena cava, increasing venous return to the right side of the heart, which increases right ventricular stroke volume. The increased volume prolongs right ventricular systole and delays pulmonic valve closure.
Meanwhile, on the left side, a greater amount of blood is sequestered in the lungs during inspiration. This momentarily decreases the amount returned to the left side of the heart, decreasing left ventricular stroke volume. The decreased volume shortens left ventricular systole and allows the aortic valve to close a bit earlier. When the aortic valve closes significantly earlier than the pulmonic valve, you can hear the two components separately. This is a split S2.
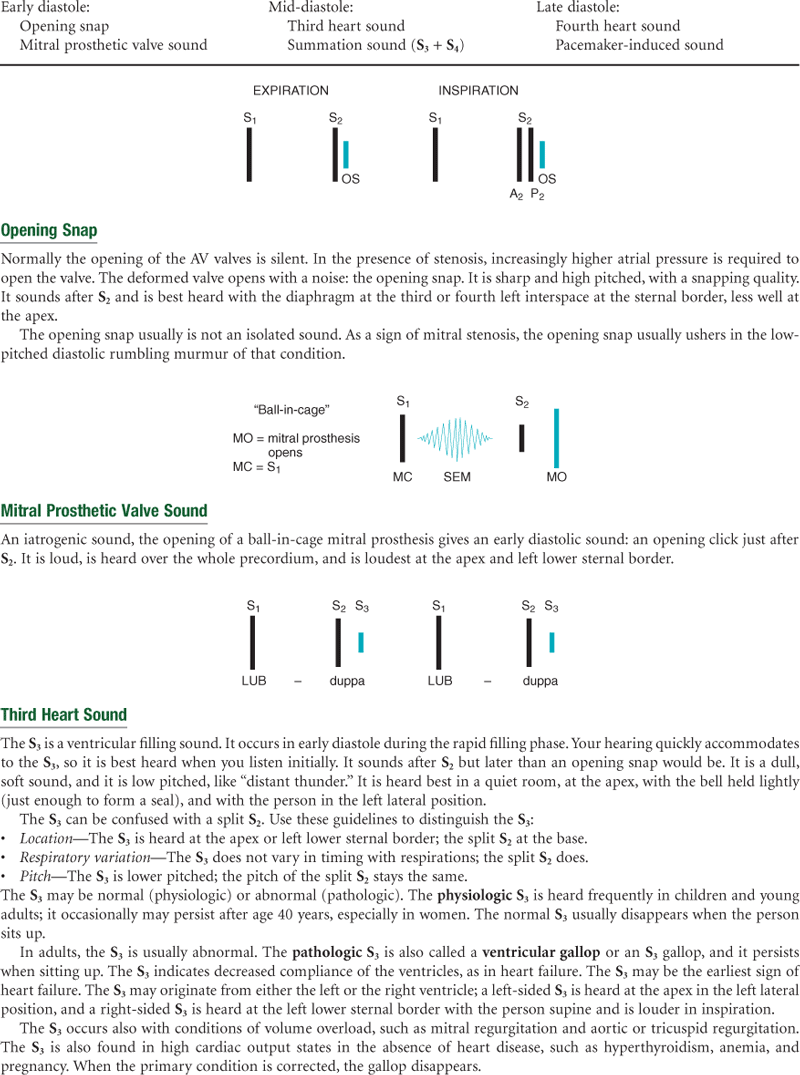
Extra Heart Sounds
Third Heart Sound (S3)
Normally, diastole is a silent event. However, in some conditions, ventricular filling creates vibrations that can be heard over the chest. These vibrations are S3. S3 occurs when the ventricles are resistant to filling during the early rapid filling phase (protodiastole). This occurs immediately after S2, when the AV valves open and atrial blood first pours into the ventricles. (See a complete discussion of S3 in Table 19-7 on p. 490.)
Murmurs
Blood circulating through normal cardiac chambers and valves usually makes no noise. However, some conditions create turbulent blood flow and collision currents. These result in a murmur, much like a pile of stones or a sharp turn in a stream creates a noisy water flow. A murmur is a gentle, blowing, swooshing sound that can be heard on the chest wall. Conditions resulting in a murmur are as follows:
Characteristics of Sound
All heart sounds are described by:
1. Frequency (pitch)—heart sounds are described as high pitched or low pitched, although these terms are relative because all are low-frequency sounds, and you need a good stethoscope to hear them
2. Intensity (loudness)—loud or soft
3. Duration—very short for heart sounds; silent periods are longer
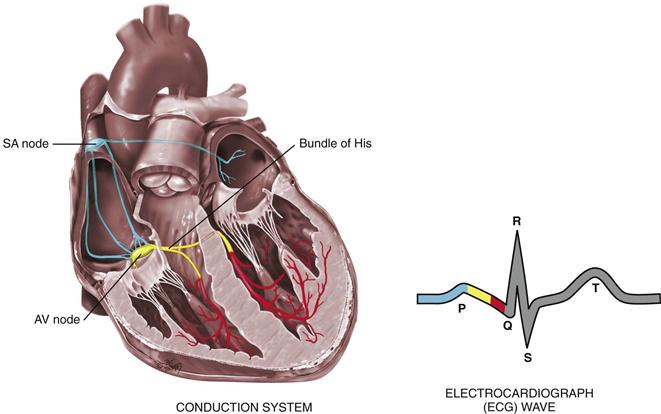
Conduction
Of all organs, the heart has a unique ability—automaticity. The heart can contract by itself, independent of any signals or stimulation from the body. The heart contracts in response to an electrical current conveyed by a conduction system (Fig. 19-8). Specialized cells in the sinoatrial (SA) node near the superior vena cava initiate an electrical impulse. (Because the SA node has an intrinsic rhythm, it is the “pacemaker.”) The current flows in an orderly sequence, first across the atria to the AV node low in the atrial septum. There it is delayed slightly so that the atria have time to contract before the ventricles are stimulated. Then the impulse travels to the bundle of His, the right and left bundle branches, and then through the ventricles.
19-8 Copyright © (2006) © Pat Thomas, 2006.
The electrical impulse stimulates the heart to do its work, which is to contract. A small amount of electricity spreads to the body surface, where it can be measured and recorded on the electrocardiograph (ECG). The ECG waves are arbitrarily labeled PQRST, which stand for the following elements:
P wave—depolarization of the atria
PR interval—from the beginning of the P wave to the beginning of the QRS complex (the time necessary for atrial depolarization plus time for the impulse to travel through the AV node to the ventricles)
Electrical events slightly precede the mechanical events in the heart. The ECG juxtaposed on the cardiac cycle is illustrated in Fig. 19-6.
Pumping Ability
In the resting adult, the heart normally pumps between 4 and 6 L of blood per minute throughout the body. This cardiac output equals the volume of blood in each systole (called the stroke volume) times the number of beats per minute (rate). This is described as:
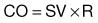
The heart can alter its cardiac output to adapt to the metabolic needs of the body. Preload and afterload affect the heart’s ability to increase cardiac output.
Preload is the venous return that builds during diastole. It is the length to which the ventricular muscle is stretched at the end of diastole just before contraction (Fig. 19-9).
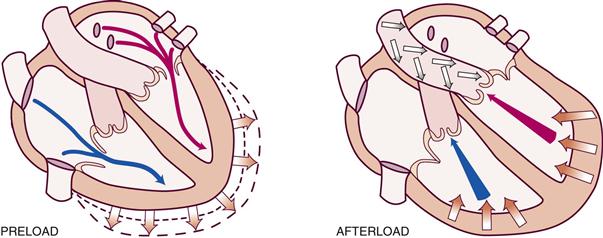
19-9 Copyright © (2006) © Pat Thomas, 2006.
When the volume of blood returned to the ventricles is increased (as when exercise stimulates skeletal muscles to contract and force more blood back to the heart), the muscle bundles are stretched beyond their normal resting state to accommodate. The force of this switch is the preload. According to the Frank-Starling law, the greater the stretch, the stronger is the heart’s contraction. This increased contractility results in an increased volume of blood ejected (increased stroke volume).
Afterload is the opposing pressure the ventricle must generate to open the aortic valve against the higher aortic pressure. It is the resistance against which the ventricle must pump its blood. Once the ventricle is filled with blood, the ventricular end diastolic pressure is 5 to 10 mm Hg, whereas that in the aorta is 70 to 80 mm Hg. To overcome this difference, the ventricular muscle tenses (isovolumic contraction). After the aortic valve opens, rapid ejection occurs.
The Neck Vessels
Cardiovascular assessment includes the survey of vascular structures in the neck—the carotid artery and the jugular veins (Fig. 19-10). These vessels reflect the efficiency of cardiac function.
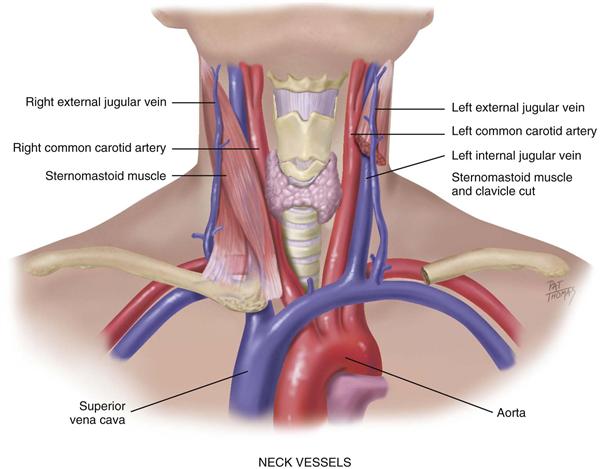
19-10
The Carotid Artery Pulse
Chapter 9 describes the pulse as a pressure wave generated by each systole pumping blood into the aorta. The carotid artery is a central artery—that is, it is close to the heart. Its timing closely coincides with ventricular systole. (Assessment of the peripheral pulses is found in Chapter 20, and blood pressure assessment is found in Chapter 9.)
The carotid artery is located in the groove between the trachea and the sternomastoid muscle, medial to and alongside that muscle. Note the characteristics of its waveform (Fig. 19-11): a smooth rapid upstroke, a summit that is rounded and smooth, and a downstroke that is more gradual and that has a dicrotic notch caused by closure of the aortic valve (marked D in the figure).
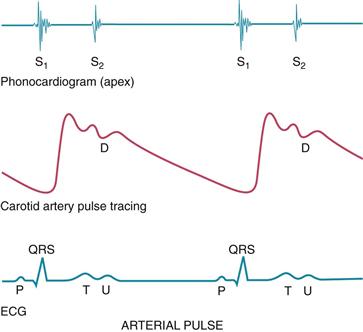
19-11
Jugular Venous Pulse and Pressure
The jugular veins empty unoxygenated blood directly into the superior vena cava. Because no cardiac valve exists to separate the superior vena cava from the right atrium, the jugular veins give information about activity on the right side of the heart. Specifically, they reflect filling pressure and volume changes. Because volume and pressure increase when the right side of the heart fails to pump efficiently, the jugular veins expose this.
Two jugular veins are present in each side of the neck (see Fig. 19-10). The larger internal jugular lies deep and medial to the sternomastoid muscle. It is usually not visible, although its diffuse pulsations may be seen in the sternal notch when the person is supine. The external jugular vein is more superficial; it lies lateral to the sternomastoid muscle, above the clavicle.
Although an arterial pulse is caused by a forward propulsion of blood, the jugular pulse is different. The jugular pulse results from a backwash, a waveform moving backward caused by events upstream. The jugular pulse has five components, as shown in Fig 19-12.
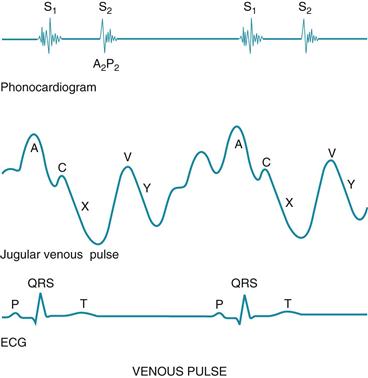
19-12
The five components of the jugular venous pulse occur because of events in the right side of the heart. The A wave reflects atrial contraction because some blood flows backward to the vena cava during right atrial contraction. The C wave, or ventricular contraction, is backflow from the bulging upward of the tricuspid valve when it closes at the beginning of ventricular systole (not from the neighboring carotid artery pulsation). Next, the X descent shows atrial relaxation when the right ventricle contracts during systole and pulls the bottom of the atria downward. The V wave occurs with passive atrial filling because of the increasing volume in the right atria and increased pressure. Finally, the Y descent reflects passive ventricular filling when the tricuspid valve opens and blood flows from the RA to the RV.
 Developmental Competence
Developmental Competence
Infants and Children
The fetal heart functions early; it begins to beat at the end of 3 weeks’ gestation. The lungs are nonfunctional, but the fetal circulation compensates for this (Fig. 19-13). Oxygenation takes place at the placenta, and the arterial blood is returned to the right side of the fetal heart. There is no point in pumping all this freshly oxygenated blood through the lungs, so it is rerouted in two ways. First, about two thirds of it is shunted through an opening in the atrial septum, the foramen ovale, into the left side of the heart, where it is pumped out through the aorta. Second, the rest of the oxygenated blood is pumped by the right side of the heart out through the pulmonary artery, but it is detoured through the ductus arteriosus to the aorta. Because they are both pumping into the systemic circulation, the right and left ventricles are equal in weight and muscle wall thickness.
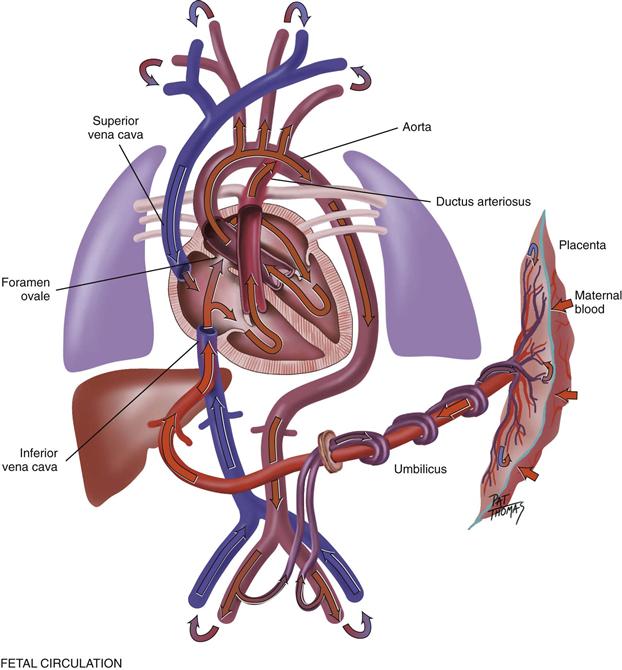
19-13
Inflation and aeration of the lungs at birth produces circulatory changes. Now the blood is oxygenated through the lungs rather than through the placenta. The foramen ovale closes within the first hour because of the new lower pressure in the right side of the heart than in the left side. The ductus arteriosus closes later, usually within 10 to 15 hours of birth. Now the left ventricle has the greater workload of pumping into the systemic circulation, so that when the baby has reached 1 year of age, the left ventricle’s mass increases to reach the adult ratio of 2 : 1, left ventricle to right ventricle.
The heart’s position in the chest is more horizontal in the infant than in the adult; thus the apex is higher, located at the fourth left intercostal space (Fig. 19-14). It reaches the adult position when the child reaches age 7 years.
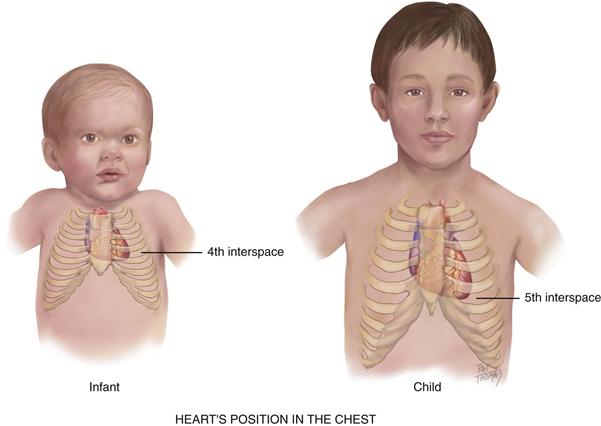
19-14
The Pregnant Woman
Blood volume increases by 30% to 40% during pregnancy, with the most rapid expansion occurring during the second trimester. This creates an increase in stroke volume and cardiac output and an increased pulse rate of 10 to 15 beats per minute. Despite the increased cardiac output, arterial blood pressure decreases in pregnancy as a result of peripheral vasodilation. The blood pressure drops to its lowest point during the second trimester and then rises after that. The blood pressure varies with the person’s position, as described on p. 483.
The Aging Adult
It is difficult to isolate the “aging process” of the cardiovascular system per se because it is so closely interrelated with lifestyle, habits, and diseases. We now know that lifestyle is a modifying factor in the development of cardiovascular disease; smoking, diet, alcohol use, exercise patterns, and stress have an influence on coronary artery disease. Lifestyle also affects the aging process; cardiac changes once thought to be due to aging are partially due to the sedentary lifestyle accompanying aging (Fig. 19-15). What is left to be attributed to the aging process alone?
19-15
Hemodynamic Changes with Aging
• With aging, there is an increase in systolic blood pressure (BP).6 This is due to stiffening of the large arteries, which in turn is due to calcification of vessel walls (arteriosclerosis). This stiffening creates an increase in pulse wave velocity because the less compliant arteries cannot store the volume ejected.
• The overall size of the heart does not increase with age, but left ventricular wall thickness increases. This is an adaptive mechanism to accommodate the vascular stiffening mentioned earlier that creates an increased workload on the heart.
• No significant change in diastolic pressure occurs with age. A rising systolic pressure with a relatively constant diastolic pressure increases the pulse pressure (the difference between the two).
• No change in resting heart rate occurs with aging.
• Cardiac output at rest is not changed with aging.
• There is a decreased ability of the heart to augment cardiac output with exercise. This is shown by a decreased maximum heart rate with exercise and diminished sympathetic response. Noncardiac factors also cause a decrease in maximum work performance with aging: decrease in skeletal muscle performance, increase in muscle fatigue, increased sense of dyspnea. Chronic exercise conditioning will modify many of the aging changes in cardiovascular function.32
Dysrhythmias
The presence of supraventricular and ventricular dysrhythmias increases with age. Ectopic beats are common in aging people; although these are usually asymptomatic in healthy older people, they may compromise cardiac output and blood pressure when disease is present.
Tachydysrhythmias may not be tolerated as well in older people. The myocardium is thicker and less compliant, and early diastolic filling is impaired at rest. Thus it may not tolerate a tachycardia as well because of shortened diastole. Also, tachydysrhythmias may further compromise a vital organ whose function has already been affected by aging or disease. For example, a ventricular tachycardia produces a 40% to 70% decrease in cerebral blood flow. Although a younger person may tolerate this, an older person with cerebrovascular disease may experience syncope.24a
ECG
Age-related changes in the ECG occur as a result of histologic changes in the conduction system. These changes include:
• Prolonged P-R interval (first-degree AV block) and prolonged Q-T interval, but the QRS interval is unchanged
• Left axis deviation from age-related mild LV hypertrophy and fibrosis in left bundle branch
Although the hemodynamic changes associated with aging alone do not seem severe or portentous, the fact remains that the incidence of cardiovascular disease increases with age. The incidence of coronary artery disease increases sharply with advancing age and accounts for about half of the deaths of older people. Hypertension (systolic >140 mm Hg and/or diastolic >90 mm Hg) and heart failure also increase with age. Certainly, lifestyle habits (smoking, chronic alcohol use, lack of exercise, diet) play a significant role in the acquisition of heart disease. Also, increasing the physical activity of older adults—even at a moderate level—is associated with a reduced risk of death from cardiovascular diseases and respiratory illnesses. Both points underscore the need for health teaching as an important treatment parameter.
 culture and genetics
culture and genetics
Prevalence is an estimate of how many people in a stated geographic location have a disease at a given point in time. In the United States, an estimated 81 million people (more than 1 in 3) have one or more forms of cardiovascular heart disease (CVD).3 The annual rates of first CVD event increase with age. For women, comparable rates occur 10 years later in life than for men, but this gap narrows with advancing age.
Causes of CVD include an interaction of genetic, environmental, and lifestyle factors. However, evidence shows potentially modifiable risk factors attribute to the overwhelming majority of cardiac risk. For example, myocardial infarction (MI) is an important type of CVD. The INTERHEART study covering 52 countries indicated that nine potentially modifiable risk factors accounted for 90% of the population attributable risk for MI in men and 94% in women!47 These nine modifiable risk factors include abnormal lipids, smoking, hypertension, diabetes, abdominal obesity, psychosocial factors, consumption of fruits and vegetables, alcohol use, and regular physical activity.
High Blood Pressure (HBP)
Although all adults have some potential CVD risk, some groups (defined by race, ethnicity, gender, socioeconomic status, educational level) carry an excess burden of CVD. Hypertension is a systolic blood pressure (SBP) of ≥140 mm Hg or diastolic blood pressure (DBP) of ≥90 mm Hg or currently taking antihypertensive medicine. A higher percentage of men than women have hypertension until age 45 years. From age 45 to 64 years, the percentages are similar; after age 64 years, women have a much higher percentage of hypertension than men have.3 Also, hypertension is 2 to 3 times more common among women taking oral contraceptives (especially among obese and older women) than in women who do not take them. Among racial groups, the prevalence of hypertension in Blacks is among the highest in the world and it is rising. The prevalence of hypertension is 31.8% for African Americans, 25.3% for American Indians or Alaska natives, 23.3% for whites, and 21% for Hispanics and Asians.3 Compared with whites, African Americans develop HBP earlier in life and their average BPs are much higher. This results in African Americans having a greater rate of stroke, death due to heart disease, and end-stage kidney disease.
Smoking
In the 40+ years from 1965 to 2004, U.S. smoking rates declined by 50.4% among adults 18 years of age and older.33 This results in 2008 with 23.1% of men and 18.3% of women being smokers. Nicotine increases the risk of MI and stroke by causing the following: increase in oxygen demand with a concomitant decrease in oxygen supply; an activation of platelets, activation of fibrinogen; and an adverse change in the lipid profile.
Serum Cholesterol
High levels of low-density lipoprotein gradually add to the lipid core of thrombus formation in arteries, which results in MI and stroke. The current cutpoints for cholesterol risk in adults are the following: total cholesterol levels of ≥240 mg/dL are high risk; and levels from 200 to 239 mg/dL are borderline–high risk. The age-adjusted prevalence of total cholesterol levels over 200 mg/dL are as follows: 51.1% of Mexican-American men and 49% of Mexican-American women; 45% of white men and 48.7% of white women; and 40.2% of African American men and 41.8% of African American women.3
Obesity
The epidemic of obesity in the United States is well known and is referenced in many chapters of this text. Among Americans ages 20 years and older, the prevalence of overweight or obesity (body mass index [BMI] of ≥25 kg/m2 for overweight and ≥30.0 for obesity) is as follows: 74.8% of Mexican-American men and 73% of Mexican-American women; 73.7% of African American men and 77.7% of African American women; and 72.4% of white men and 57.5% of white women.
Type 2 Diabetes Mellitus
The risk of CVD is twofold greater among persons with diabetes mellitus (DM) than without DM. The increased prevalence of DM in the United States is being followed by an increasing prevalence of CVD morbidity and mortality.3 Diabetes causes damage to the large blood vessels that nourish the brain, heart, and extremities; this results in stroke, coronary artery disease, and peripheral vascular disease.
About 13% of African Americans 20 years of age and older have DM. Between 11.8% and 13.1% of Mexican Americans have DM, compared with 6.4% of whites.3 The most powerful predictor of type 2 DM is obesity, with abdominal (visceral) fat posing a greater risk than lower body obesity poses. Evidence from epidemiologic studies shows a strong genetic factor for DM, but no specific antigen type has yet been identified. In the past, type 2 DM was diagnosed in adults 40 years of age and older, but now we are finding more children with type 2 DM. These children are usually overweight or obese, have a family history of DM, and identify with American Indian, African American, Hispanic, or Asian groups.3
Subjective Data
| Examiner Asks | Rationale |
| 1.Chest pain. Any chest pain or tightness? •Onset: When did it start? How long have you had it this time? Had this type of pain before? How often? •Location: Where did the pain start? Does the pain radiate to any other spot? •Character: How would you describe it? Crushing, stabbing, burning, viselike? (Allow the person to offer adjectives before you suggest them.) (Note if uses clenched fist to describe pain.) | Angina, an important cardiac symptom, occurs when the heart’s own blood supply cannot keep up with metabolic demand. Chest pain also may have pulmonary, musculoskeletal, or gastrointestinal origin; it is important to differentiate. A squeezing “clenched fist” sign is characteristic of angina, but the symptoms below may be anginal equivalents in the absence of chest pain.37a |
| •Pain brought on by: Activity—what type; rest; emotional upset; after eating; during sexual intercourse; with cold weather? | |
| •Any associated symptoms: Sweating, ashen gray or pale skin, heart skips beat, shortness of breath, nausea or vomiting, racing of heart? | Diaphoresis, cold sweats, pallor, grayness. Palpitations, dyspnea, nausea, tachycardia, fatigue. |
| •Pain made worse by moving the arms or neck, breathing, lying flat? | Try to differentiate pain of cardiac versus noncardiac origin. |
| •Pain relieved by rest or nitroglycerin? How many tablets? | |
| 2.Dyspnea. Any shortness of breath? •What type of activity and how much brings on shortness of breath? How much activity brought it on 6 months ago? •Onset: Does the shortness of breath come on unexpectedly? •Duration: Constant or does it come and go? •Seem to be affected by position: Lying down? •Awaken you from sleep at night? |
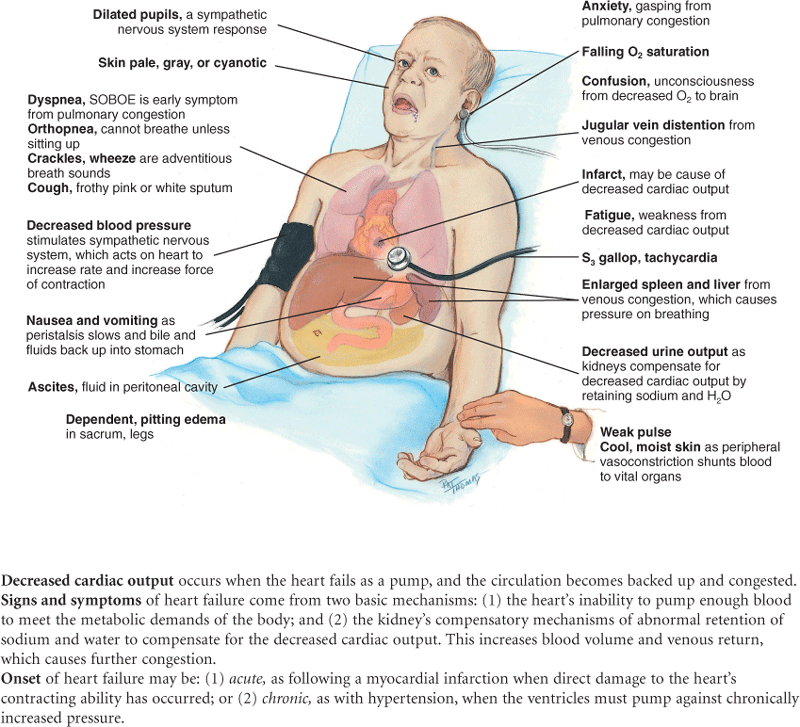
Dyspnea on exertion (DOE)—quantify exactly (e.g., DOE after walking two level blocks). Paroxysmal. Constant or intermittent. Recumbent. Paroxysmal nocturnal dyspnea (PND) occurs with heart failure. Lying down increases volume of intrathoracic blood, and the weakened heart cannot accommodate the increased load. Typically, the person awakens after 2 hours of sleep with the perception of needing fresh air. |
| •Does the shortness of breath interfere with activities of daily living? | |
| 3.Orthopnea. How many pillows do you use when sleeping or lying down? |
Orthopnea is the need to assume a more upright position to breathe. Note the exact number of pillows used. |
| 4.Cough. Do you have a cough? |
|
| •Duration: How long have you had it? | |
| •Frequency: Is it related to time of day? | |
| •Type: Dry, hacking, barky, hoarse, or congested? | |
| •Do you cough up mucus? Color? Any odor? Blood tinged? | Sputum production, mucoid or purulent. Hemoptysis is often a pulmonary disorder but also occurs with mitral stenosis. |
| •Associated with: Activity, position (lying down), anxiety, talking? | |
| •Does activity make it better or worse (sit, walk, exercise)? | |
| •Relieved by rest or medication? | |
| 5.Fatigue. Do you seem to tire easily? Able to keep up with your family and co-workers? |
|
| •Onset: When did fatigue start? Sudden or gradual? Has any recent change occurred in energy level? | |
| •Fatigue related to time of day: All day, morning, evening? | Fatigue from decreased cardiac output is worse in the evening, whereas fatigue from anxiety or depression occurs all day or is worse in the morning. |
| 6.Cyanosis or pallor. Ever noted your facial skin turn blue or ashen? |
Cyanosis or pallor occurs with myocardial infarction or low cardiac output states as a result of decreased tissue perfusion. |
| 7.Edema. Any swelling of your feet and legs? •Onset: When did you first notice this? •Any recent change? |
Edema is dependent when caused by heart failure. |
| •What time of day does the swelling occur? Do your shoes feel tight at the end of day? | Cardiac edema is worse at evening and better in morning after elevating legs all night. |
| •How much swelling would you say there is? Are both legs equally swollen? | Cardiac edema is bilateral; unilateral swelling has a local vein cause. |
| •Does the swelling go away with: Rest, elevation, after a night’s sleep? | |
| •Any associated symptoms, such as shortness of breath? If so, does the shortness of breath occur before leg swelling or after? | |
| 8.Nocturia. Do you awaken at night with an urgent need to urinate? How long has this been occurring? Any recent change? |
Nocturia—Recumbency at night promotes fluid reabsorption and excretion; this occurs with heart failure in the person who is ambulatory during the day. |
| 9.Cardiac history. Any past history of: Hypertension, elevated cholesterol or triglycerides, heart murmur, congenital heart disease, rheumatic fever or unexplained joint pains as child or youth, recurrent tonsillitis, anemia? |
|
| •Ever had heart disease? When was this? Treated by medication or heart surgery? | |
| •Last ECG, stress ECG, serum cholesterol measurement, other heart tests? | |
| 10. Family cardiac history. Any family history of: Hypertension, obesity, diabetes, coronary artery disease (CAD), sudden death at younger age? |
|
| 11.Personal habits (cardiac risk factors). •Nutrition: Please describe your usual daily diet. (Note if this diet is representative of the basic food groups, the amount of calories, cholesterol, |
|
| and any additives such as salt.) What is your usual weight? Has there been any recent change? •Smoking: Do you smoke cigarettes or other tobacco? At what age did you start? How many packs per day? For how many years have you smoked this amount? Have you ever tried to quit? If so, how did this go? •Alcohol: How much alcohol do you usually drink each week, or each day? When was your last drink? What was the number of drinks during that episode? Have you ever been told you had a drinking problem? |
Risk factors for CAD—Collect data regarding elevated cholesterol, elevated blood pressure, blood sugar levels above 130 mg/dL or known diabetes mellitus, obesity, cigarette smoking, low activity level, and length of any hormone replacement therapy for postmenopausal women. |
| •Exercise: What is your usual amount of exercise each day or week? What type of exercise (state type or sport)? If a sport, what is your usual amount (light, moderate, heavy)? | |
| •Drugs: Do you take any antihypertensives, beta-blockers, calcium channel blockers, digoxin, diuretics, aspirin/anticoagulants, over-the-counter or street drugs? | |
| Additional History for Infants | |
| 1. How was the mother’s health during pregnancy: Any unexplained fever, rubella first trimester, other infection, hypertension, drugs taken? |
|
| 2. Have you noted any cyanosis while nursing, crying? Is the baby able to eat, nurse, or finish bottle without tiring? |
To screen for heart disease in infant, note fatigue during feeding. Infant with heart failure takes fewer ounces each feeding; becomes dyspneic with sucking; may be diaphoretic, then falls into exhausted sleep; awakens after a short time hungry again. |
| 3. Growth: Has this baby grown as expected by growth charts and about the same as siblings or peers? |
Poor weight gain. |
| 4. Activity: Were this baby’s motor milestones achieved as expected? Is the baby able to play without tiring? How many naps does the baby take each day? How long does a nap last? |
|
| Additional History for Children | |
| 1. Growth: Has this child grown as expected by growth charts? |
Poor weight gain. |
| 2. Activity: Is this child able to keep up with siblings or age mates? Is the child willing or reluctant to go out to play? Is the child able to climb stairs, ride a bike, walk a few blocks? Does the child squat to rest during play or to watch television, or assume a knee-chest position while sleeping? Have you noted “blue spells” during exercise? |
Fatigue. Record specific limitations. Cyanosis. |
| 3. Has the child had any unexplained joint pains or unexplained fever? |
|
| 4. Does the child have frequent headaches, nosebleeds? |
|
| 5. Does the child have frequent respiratory infections? How many per year? How are they treated? Have any of these proved to be streptococcal infections? |
|
| 6. Family history: Does the child have a sibling with heart defect? Is anyone in the child’s family known to have chromosomal abnormalities, such as Down syndrome? |
|
| Additional History for the Pregnant Woman | |
| 1. Have you had any high blood pressure during this or earlier pregnancies? |
|
| •What was your usual blood pressure level before pregnancy? How has your blood pressure been monitored during the pregnancy? | |
| •If high blood pressure, what treatment has been started? | |
| •Any associated symptoms: Weight gain, protein in urine, swelling in feet, legs, or face? | |
| 2. Have you had any faintness or dizziness with this pregnancy? |
|
| Additional History for the Aging Adult | |
| 1. Do you have any known heart or lung disease: Hypertension, CAD, chronic emphysema, or bronchitis? |
|
| •What efforts to treat this have been started? | |
| •Usual symptoms changed recently? Does your illness interfere with activities of daily living? | |
| 2. Do you take any medications for your illness such as digitalis? Aware of side effects? Have you recently stopped taking your medication? Why? |
Noncompliance may be related to side effects or lack of finances. |
| 3. Environment: Does your home have any stairs? How often do you need to climb them? Does this have any effect on activities of daily living? |
Objective Data


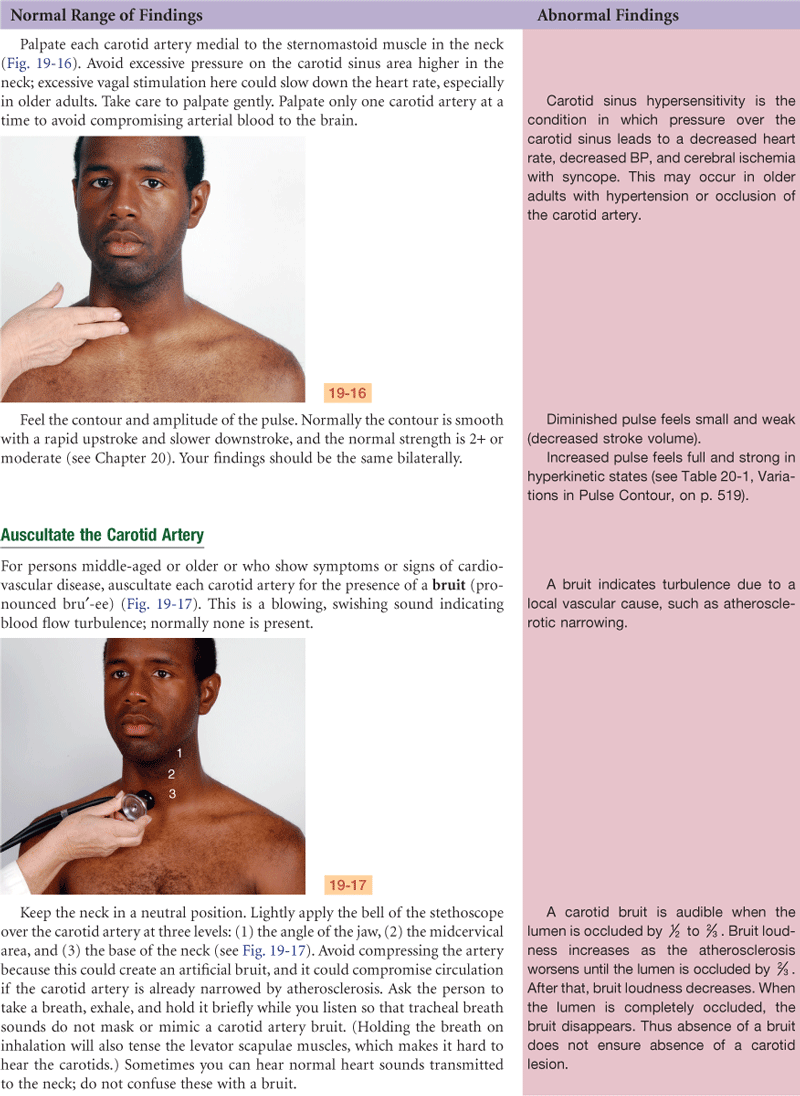

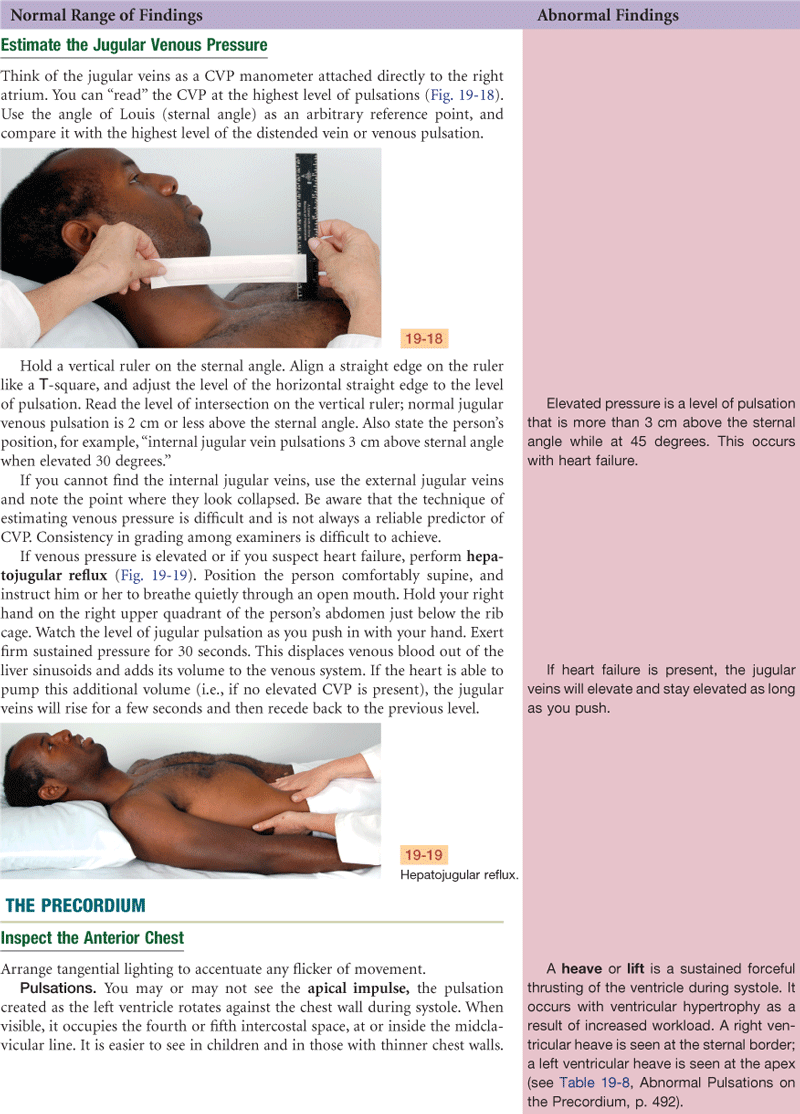
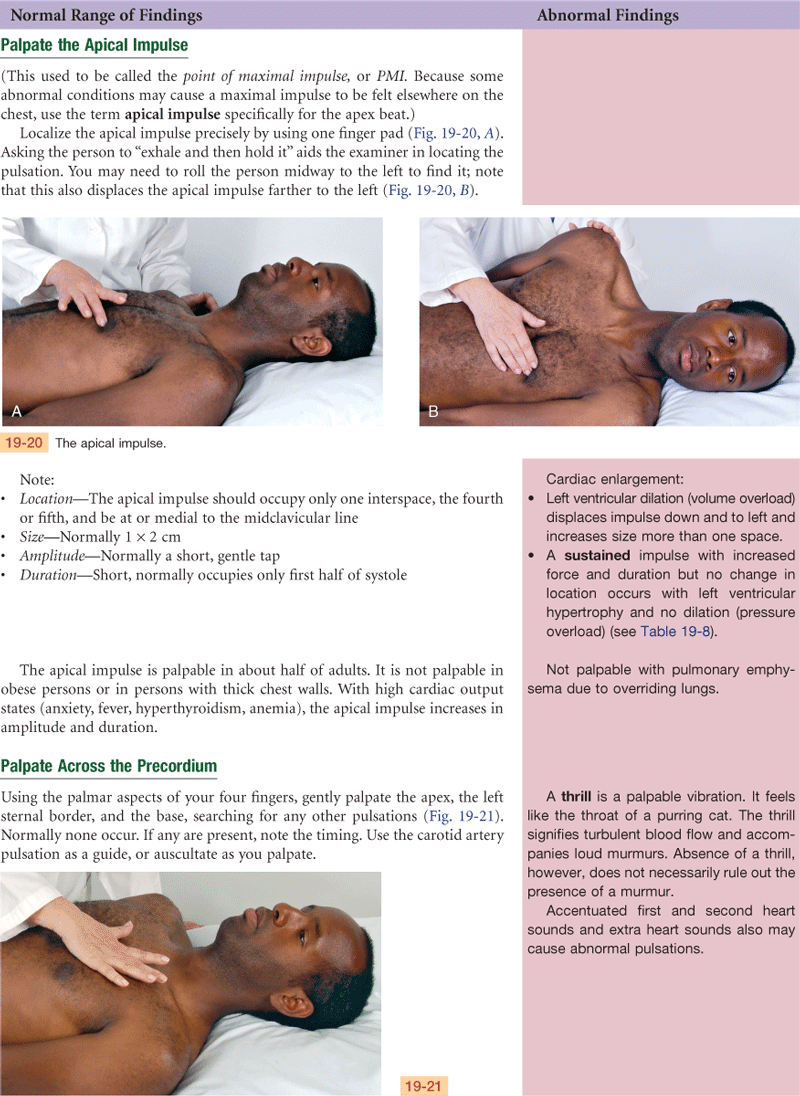
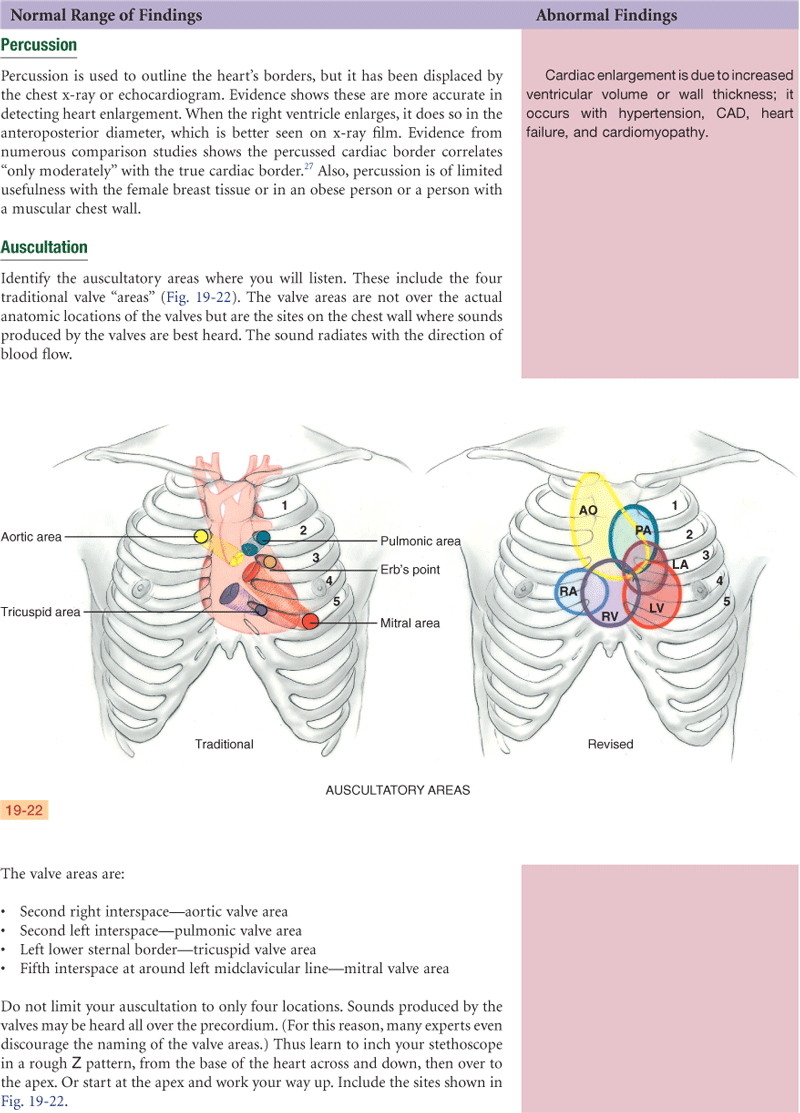


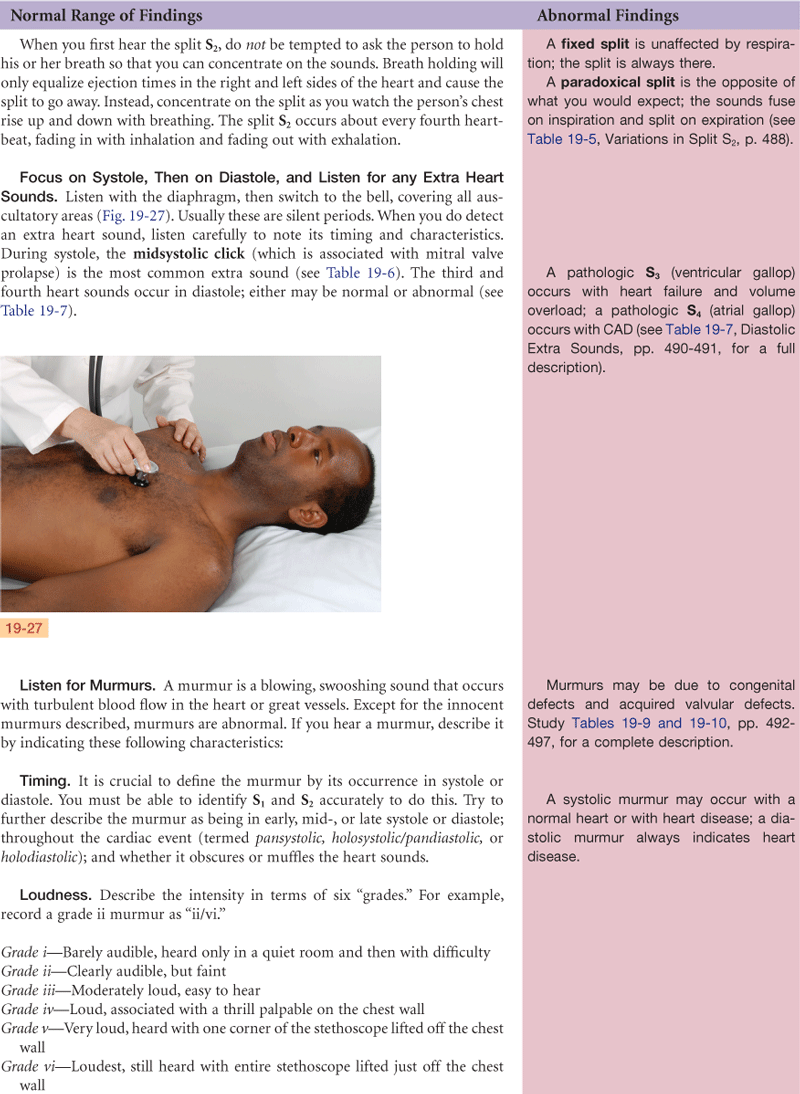

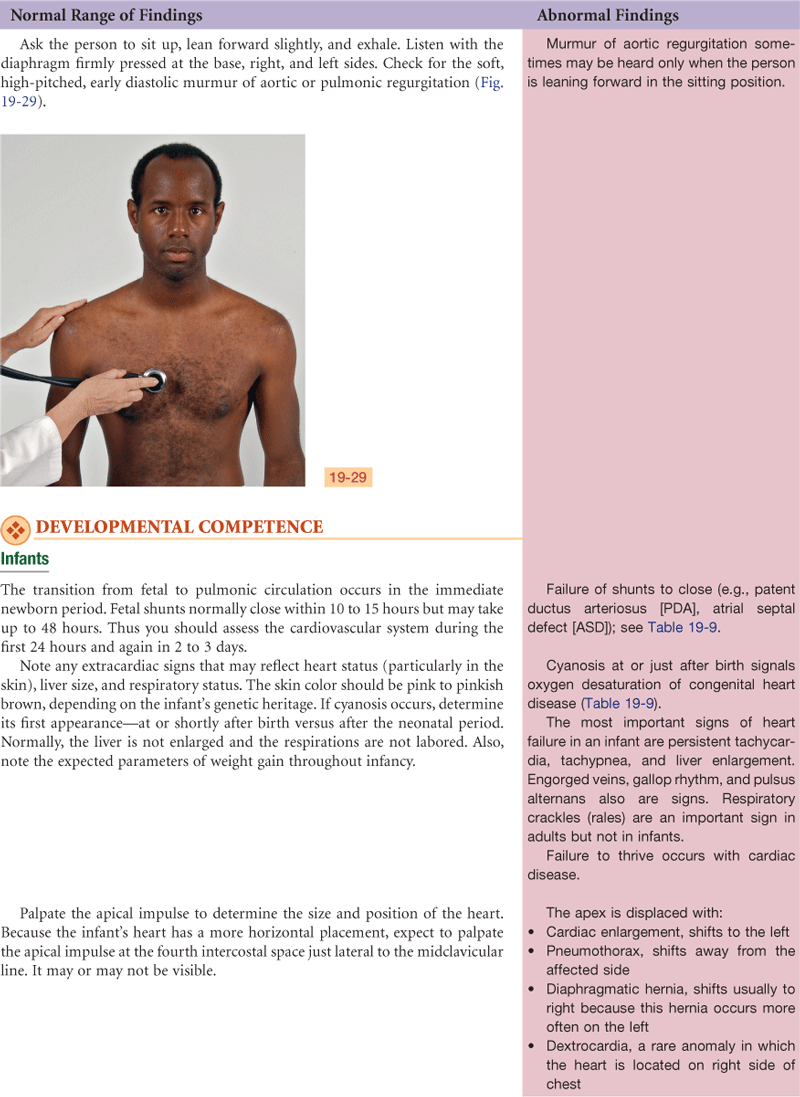

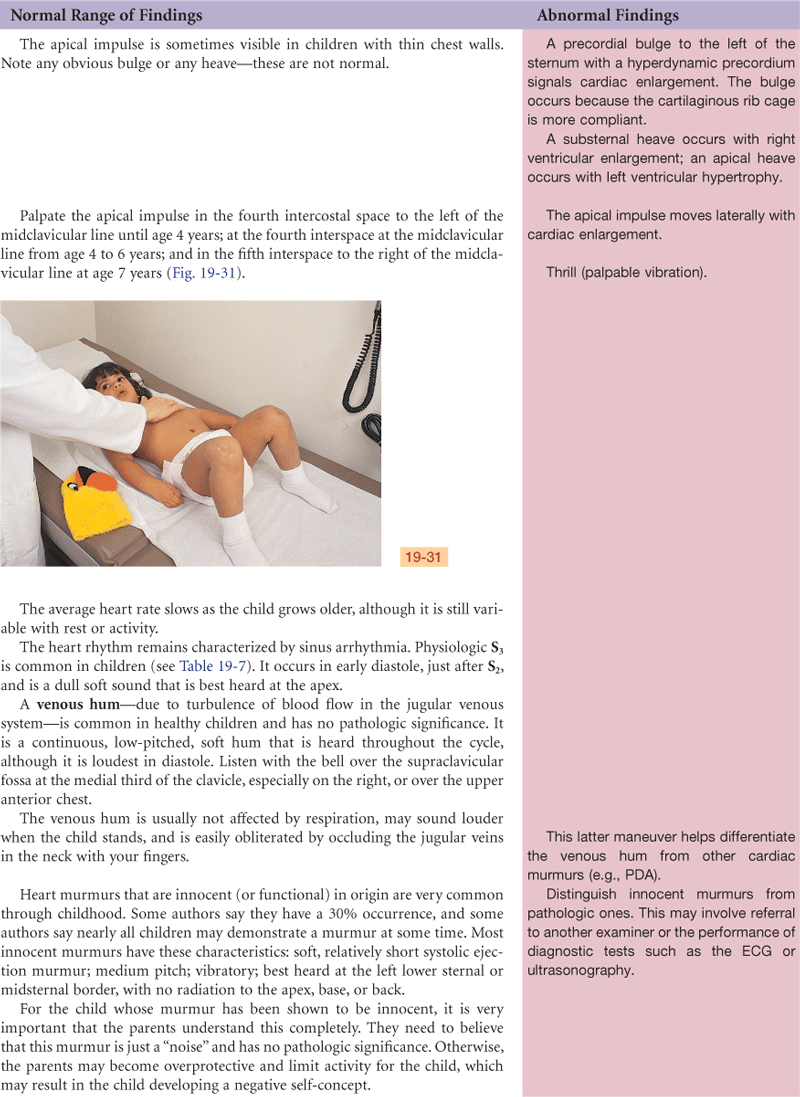

TABLE 19-1
Characteristics of Jugular Versus Carotid Pulsations
| Internal Jugular Pulse | Carotid Pulse | |
| 1. Location | Lower, more lateral, under or behind the sternomastoid muscle | Higher and medial to this muscle |
| 2. Quality | Undulant and diffuse, two visible waves per cycle | Brisk and localized, one wave per cycle |
| 3. Respiration | Varies with respiration; its level descends during inspiration when intrathoracic pressure is decreased | Does not vary |
| 4. Palpable | No | Yes |
| 5. Pressure | Light pressure at the base of the neck easily obliterates | No change |
| 6. Position of person | Level of pulse drops and disappears as the person is brought to a sitting position | Unaffected |
 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
No chest pain, dyspnea, orthopnea, cough, fatigue, or edema. No history of hypertension, abnormal blood tests, heart murmur, or rheumatic fever in self. Last ECG 2 yrs. PTA, result normal. No stress ECG or other heart tests.
Family history—Father with obesity, smoking, and hypertension, treated  diuretic medication. No other family history significant for cardiovascular disease.
diuretic medication. No other family history significant for cardiovascular disease.
Personal habits—Diet balanced in 4 food groups, 2 to 3 c. regular coffee/day; no smoking; alcohol, 1 to 2 beers occasionally on weekend; exercise, runs 2 miles, 3 to 4 ×/week; no prescription or OTC medications or street drugs.
Objective
Neck: Carotids 2+ and = bilaterally. Internal jugular vein pulsations present when supine and disappear when elevated to a 45° position.
Precordium: Inspection. No visible pulsations, no heave or lift.
Palpation: Apical impulse in 5th ics at left midclavicular line, no thrill.
Auscultation: Rate 68 beats per minute, rhythm regular, S1-S2 are normal, not diminished or accentuated, no S3, no S4 or other extra sounds, no murmurs.
Assessment
Neck vessels healthy by inspection and auscultation
Heart sounds normal
Focused Assessment: Clinical Case Study
Mr. N.V. is a 53-year-old white male woodcutter admitted to the CCU at University Medical Center (UMC) with chest pain.
Subjective
1 year PTA—N.V. admitted to UMC with crushing substernal chest pain, radiating to L shoulder, accompanied by nausea, vomiting, diaphoresis.
Diagnosed as MI, hospitalized 7 days, discharged with nitroglycerin prn for anginal pain.
Did not return to work. Activity included walking 1 mile/day, hunting. Had occasional episodes of chest pain with exercise, relieved by rest.
1 day PTA—had increasing frequency of chest pain, about every 2 hours, lasting few minutes, saw pain as warning to go to MD.
Day of admission—severe substernal chest pain (“like someone sitting on my chest”) unrelieved by rest. Saw personal MD, while in office had episode of chest pain similar to last year’s, accompanied by diaphoresis, no N & V or SOB, relieved by 1 nitroglycerin. Transferred to UMC by paramedics. No further pain since admission 2 hours ago.
Family hx—mother died of MI at age 57.
Personal habits—smokes 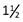 pack cigarettes daily × 34 years, no alcohol, diet—trying to limit fat and fried food, still high in added salt.
pack cigarettes daily × 34 years, no alcohol, diet—trying to limit fat and fried food, still high in added salt.
Objective
Extremities: Skin pink, no cyanosis. Upper extrem.—capillary refill sluggish, no clubbing. Lower extrem.—no edema, no hair growth 10 cm below knee bilaterally.
Pulses—
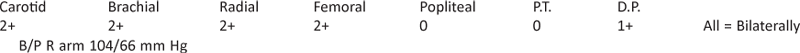
B/P R arm 104/66 mm Hg
Neck: External jugulars flat. Internal jugular pulsations present when supine and absent when elevated to 45°.
Precordium: Inspection. Apical impulse visible 5th ics, 7 cm left of midsternal line, no heave.
Palpation: Apical impulse palpable in 5th and 6th ics. No thrill.
Auscultation: Apical rate 92 bpm regular, S1-S2 are normal, not diminished or accentuated, no S3 or S4, grade iii/vi systolic murmur present at left lower sternal border.
Assessment
Substernal chest pain
Systolic murmur
Ineffective tissue perfusion R/T interruption in flow
Decreased cardiac output R/T reduction in stroke volume
Abnormal Findings for Advanced Practice
TABLE 19-10
Murmurs Due to Valvular Defects
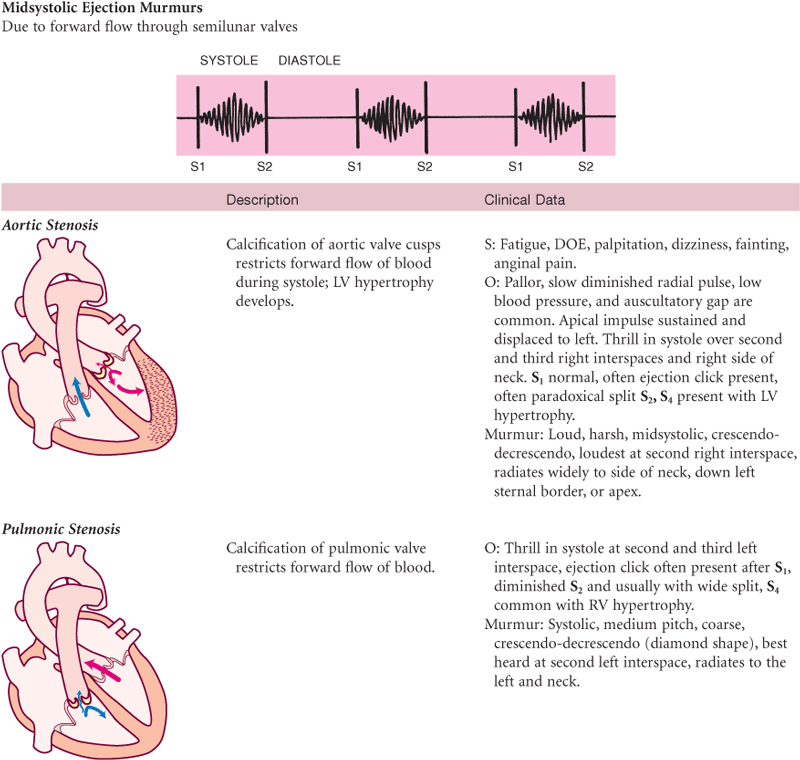
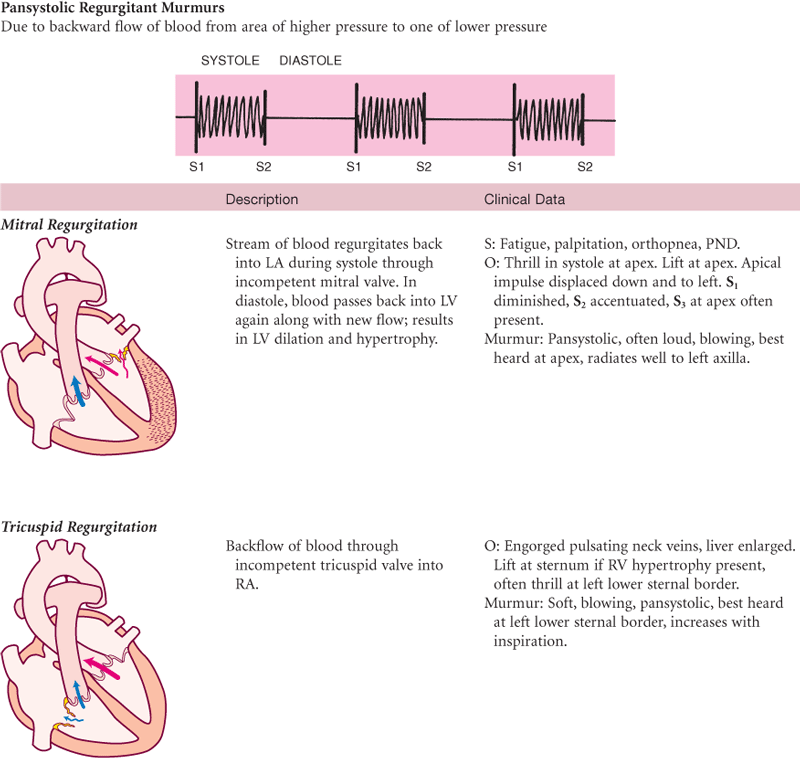
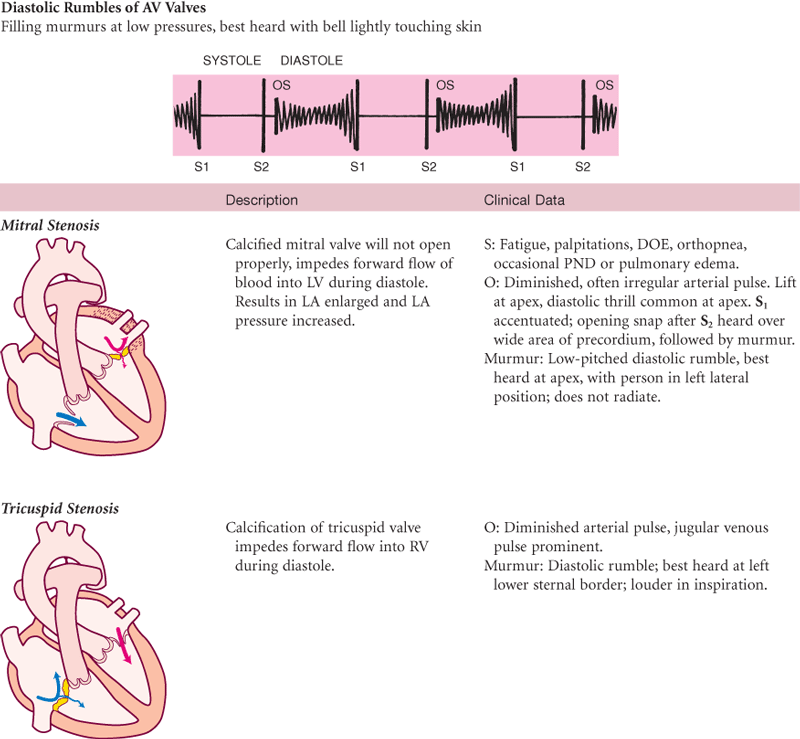
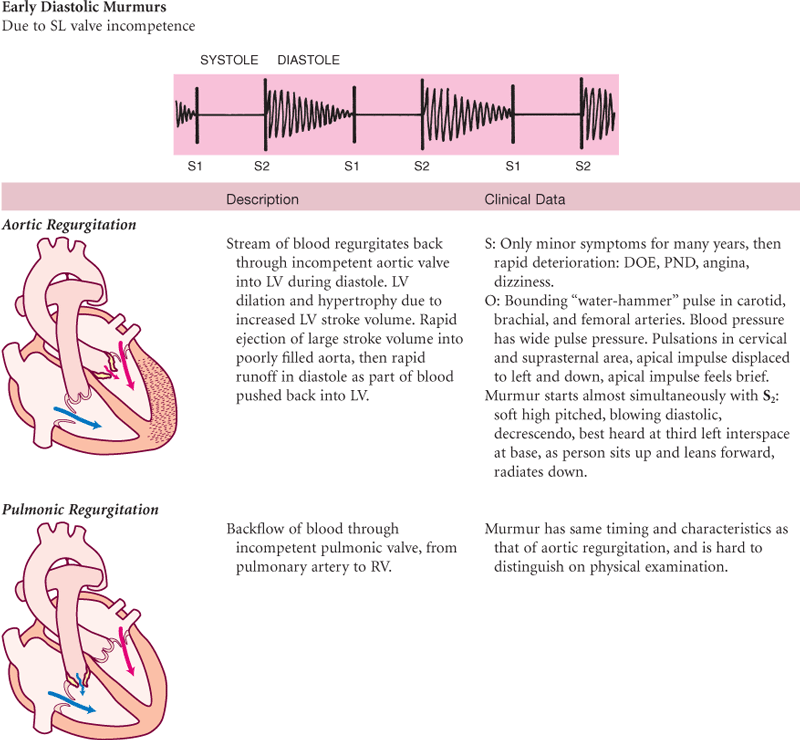
S, Subjective data; O, objective data.
Images © Pat Thomas, 2006.
Summary Checklist: Heart and Neck Vessels Exam
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
1. Carotid pulse—Observe and palpate
2. Observe jugular venous pulse
3. Estimate jugular venous pressure
1. Describe location of apical impulse
2. Note any heave (lift) or thrill
1. Identify anatomic areas where you listen
2. Note rate and rhythm of heartbeat
3. Identify S1 and S2 and note any variation
4. Listen in systole and diastole for any extra heart sounds
5. Listen in systole and diastole for any murmurs
Bibliography
1. Acelajado MC, Oparil S. Hypertension in the elderly. Clinics in Geriatric Medicine. 2009;25(3):391–412.
2. Allman E, Berry D, Nasir L. Depression and coping in heart failure patients. Journal of Cardiovascular Nursing. 2009;24(2):106–117.
3. American Heart Association. Heart disease and stroke statistics, 2010. Retrieved May 2010, from www.americanheart.org/downloadable/heart; 2010.
4. Andrews TD, Cook SS, Baumeister M, et al. ARVC: help prevent sudden death. Nurse Practitioner. 2010;35(2):26–33.
5. Armbrister KA. Self-management: improving heart failure outcomes. Nurse Practitioner. 2008;33(11):20–28.
6. Aronow WS. Heart disease and aging. Medical Clinics of North America. 2006;90(1):849–862.
7. Braveman PA, Cubbin C, Egerter S, et al. Socioeconomic disparities in health in the United States: what the patterns tell us. American Journal of Public Health. 2010;100(Suppl. 1):S186–S196.
8. Brown DW, Giles WH, Croft JB. Association of cardiac auscultatory findings with coronary heart disease mortality. North American Journal of Medical Sciences. 2009;1:327–332.
9. Conn RD, O’Keefe JH. Cardiac physical diagnosis in the digital age: an important but increasingly neglected skill (from stethoscopes to microchips). American Journal of Cardiology. 2009;104(4):590–595.
10. Cunningham FG, Leveno KJ, Bloom SL, et al. Williams obstetrics. 23rd ed. New York: McGraw-Hill; 2010.
11. Dakin CL. New approaches to heart failure in the ED. American Journal of Nursing. 2008;108(3):68–71.
12. Dimeff RJ. High school athlete with family history of sudden cardiac death. Consultant. 2009;49(2):79–80.
13. Dracup K, McKinley S, Doering LV, et al. Acute coronary syndrome: what do patients know? Archives of Internal Medicine. 2008;168(10):1049–1054.
14. Dressler DK. Death by clot: acute coronary syndromes, ischemic stroke, pulmonary embolism, and disseminated intravascular coagulation. AACN Advanced Critical Care. 2009;20(2):166–176.
15. Eslick GD. Usefulness of chest pain character and location as diagnostic indicators of an acute coronary syndrome. American Journal of Cardiology. 2005;95(1):1228–1231.
15a. Fleg JL. Diagnostic evaluations. In: Abrams WB, Berkow R, eds. The Merck manual of geriatrics. Rahway, NJ: Merck, Sharp, & Dohme; 1990.
16a. Fleiner S. Recognition and stabilization of neonates with congenital heart disease. Newborn and Infant Nursing Reviews. 2006;6(3):137–150.
17. Grossman VGA, McGowan BA. Postural orthostatic tachycardia syndrome. American Journal of Nursing. 2008;108(8):58–60.
18. Hartas G, Tsounias E, Gupta-Malhotra M. Approach to diagnosing congenital cardiac disorders. Critical Care Nursing Clinics of North America. 2009;21(1):27–36.
19. Hayman LL, Kamau MW, Stuart-Shor EM. The heart of the matter: reducing CVD risk. Nurse Practitioner. 2009;34(5):31–35.
20. Klein DG. Thoracic aortic aneurysms. Journal of Cardiovascular Nursing. 2005;20:245–250.
21. Kliegman RM, Behrman RE, Jenson HB, et al. Nelson textbook of pediatrics. 18th ed. Philadelphia: Saunders; 2007.
22. Klieman L, Hyde S, Berra K. Cardiovascular disease risk reduction in older adults. Journal of Cardiovascular Nursing. 2006;21:527–539.
23. Kumar A, Cannon CP. Acute coronary syndromes. Mayo Clinic Proceedings. 2009;84(10):917–938.
24. Lembo NJ, Dell’Italia LJ, Crawford MH, et al. Bedside diagnosis of systolic murmurs. New England Journal of Medicine. 1988;318(24):1572–1578.
24a. Libby P, Bonow RO, Mann DL, et al. Braunwald’s heart disease: a textbook of cardiovascular medicine. 8th ed. Philadelphia: Saunders; 2008.
25. Lloyd-Jones DM, Hong Y, Labarthe D, et al. Defining and setting national goals for cardiovascular health promotion and disease reduction. Circulation. 2010;121(1):586–613.
26. Matthews KA, Crawford SL, Chae CU, et al. Are changes in cardiovascular disease risk factors in midlife women due to chronological aging or to the menopausal transition? Journal of the American College of Cardiology. 2009;54:2366–2373.
27. McGee S. Evidence based physical diagnosis. 2nd ed. Philadelphia: Saunders; 2007.
28. Moe GW, Tu J. Heart failure in the ethnic minorities. Current Opinion in Cardiology. 2010;2:124–130.
29. Mosack V, Steinke EE. Trends in sexual concerns after myocardial infarction. Journal of Cardiovascular Nursing. 2009;24(2):162–170.
30. Mosley W, Lloyd-Jones DM. Epidemiology of hypertension in the elderly. Clinics in Geriatric Medicine. 2009;25(2):179–189.
31. Moulton SA. Hypertension in African Americans and its related chronic diseases. Journal of Cultural Diversity. 2009;16(4):165–170.
32. Muster AJ, Kim H, Kane B, et al. Ten-year echo/Doppler determination of the benefits of aerobic exercise after the age of 65 years. Echocardiography. 2009;27(1):5–10.
33. National Center for Health Statistics. Health, United States, 2008 with chartbook. Hyattsville, MD: Author; 2009; Retrieved May 20, 2010, from www.cdc.gov/nchs/data/hus/hus08.pdf; 2009.
34. Overbaugh KJ. Acute coronary syndrome. American Journal of Nursing. 2009;109(5):42–52.
35. Perloff JK. Physical examination of the heart and circulation. 3rd ed. Philadelphia: Saunders; 2000.
36. Poletti JM. Carotid bruit on an orthopedic preoperative exam. Nurse Practitioner. 2009;34(3):8–11.
37. Ramani GV, Uber PA, Mehra MR. Chronic heart failure. Mayo Clinic Proceedings. 2010;85(2):180–195.
37a. Reigle J. Evaluating the patient with chest pain. Journal of Cardiovascular Nursing. 2005;20:226–231.
38. Rich MW. Heart failure in older adults. Medical Clinics of North America. 2006;90(5):863–885.
39. Riegel B, Dickson VV, Cameron J, et al. Symptom recognition in elders with heart failure. Journal of Nursing Scholarship. 2010;42(1):92–100.
40. Sen B, McNab A, Burdess C. Identifying and managing patients with acute coronary conditions. Emergency Nurse. 2009;17(7):18–23.
41. Stewart D, Casida J. Diagnosis and management of an adult patient with atrial septal defect. Nurse Practitioner. 2010;35(2):8–11.
42. Tanaka H. Habitual exercise for the elderly. Family & Community Health. 2009;32(Suppl. 1):S57–S65.
43. Thompson J. Psychological and physical etiologies of heart palpitations. Nurse Practitioner. 2006;31(2):14–25.
44. Tilkian AG, Conover MB. Understanding heart sounds and murmurs with an introduction to lung sounds. 4th ed. Philadelphia: Saunders; 2001.
45. Turris SA. Women’s decisions to seek treatment for the symptoms of potential cardiac illness. Journal of Nursing Scholarship. 2009;41(1):5–12.
46. Wei JY. Age and the cardiovascular system. New England Journal of Medicine. 1992;327:1735–1739.
47. Yusuf S, Hawken S, Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study). Lancet. 2004;364:937–952.