Peripheral Vascular System and Lymphatic System
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
• Physical Examination Summary Checklist
Structure and Function
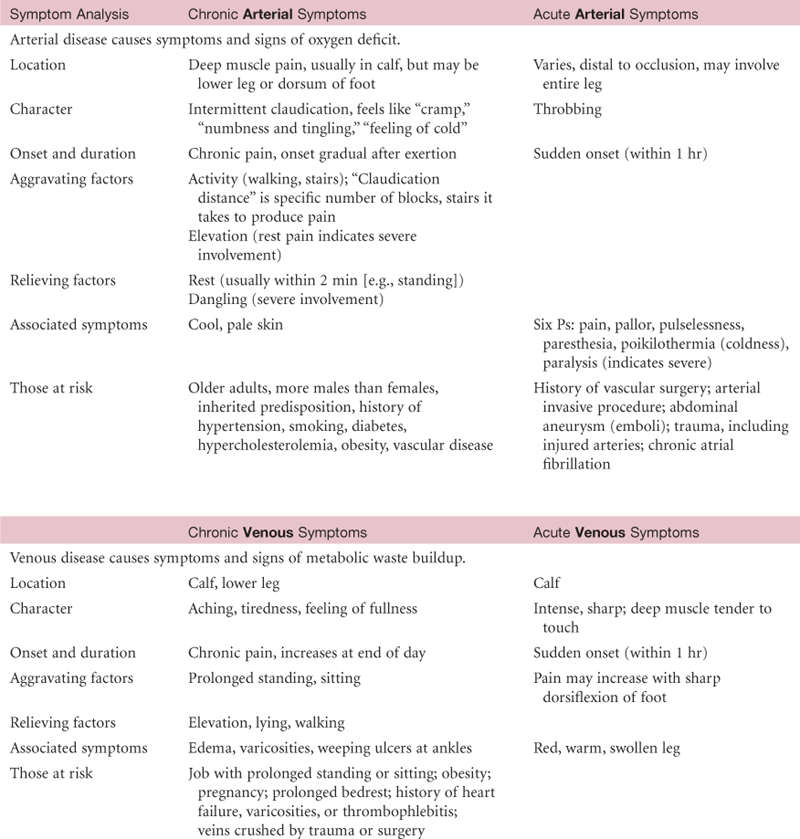
The vascular system consists of the vessels of the body. Vessels are tubes for transporting fluid, such as the blood or lymph. Any disease in the vascular system creates problems with delivery of oxygen and nutrients to the tissues or elimination of carbon dioxide and waste products from cellular metabolism.
Arteries
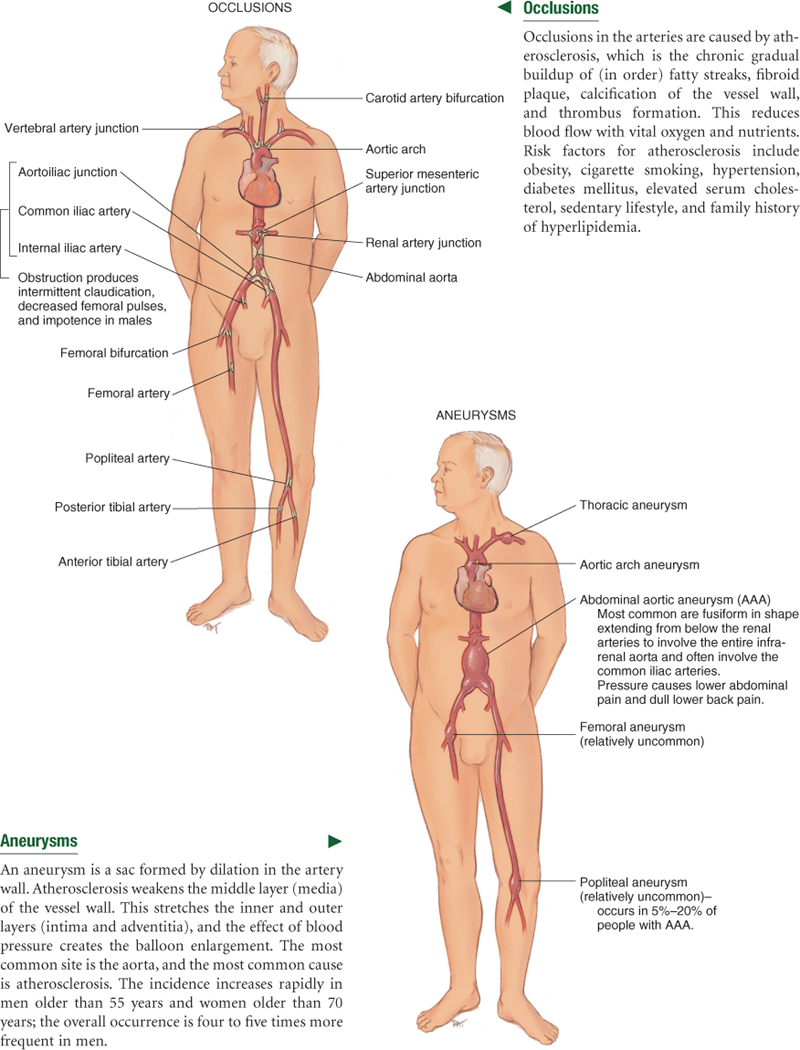
The heart pumps freshly oxygenated blood through the arteries to all body tissues. The pumping heart makes this a high-pressure system. The artery walls are strong, tough, and tense to withstand pressure demands. Arteries contain elastic fibers, which allow their walls to stretch with systole and recoil with diastole. Arteries also contain muscle fibers (vascular smooth muscle, or VSM), which control the amount of blood delivered to the tissues. The VSM contracts or dilates, which changes the diameter of the arteries to control the rate of blood flow.
Each heartbeat creates a pressure wave, which makes the arteries expand and then recoil. It is the recoil that propels blood through like a wave. All arteries have this pressure wave, or pulse, throughout their length, but you can feel it only at body sites where the artery lies close to the skin and over a bone. The following arteries are accessible to examination.
Temporal Artery
The temporal artery is palpated in front of the ear, as discussed in Chapter 13.
Carotid Artery
The carotid artery is palpated in the groove between the sternomastoid muscle and the trachea and is discussed in Chapter 19.
Arteries in the Arm
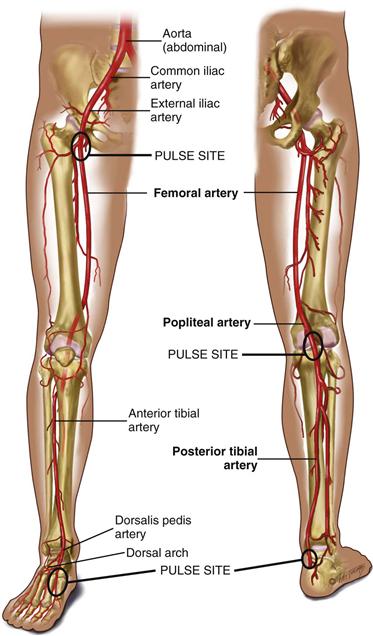
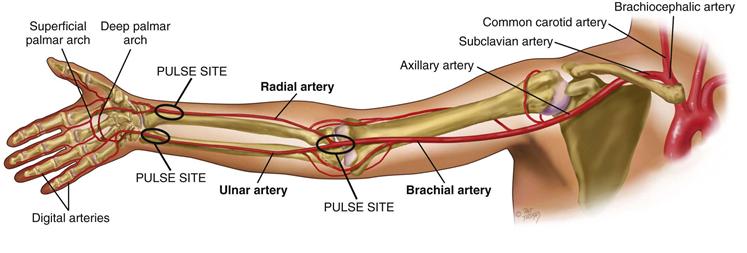
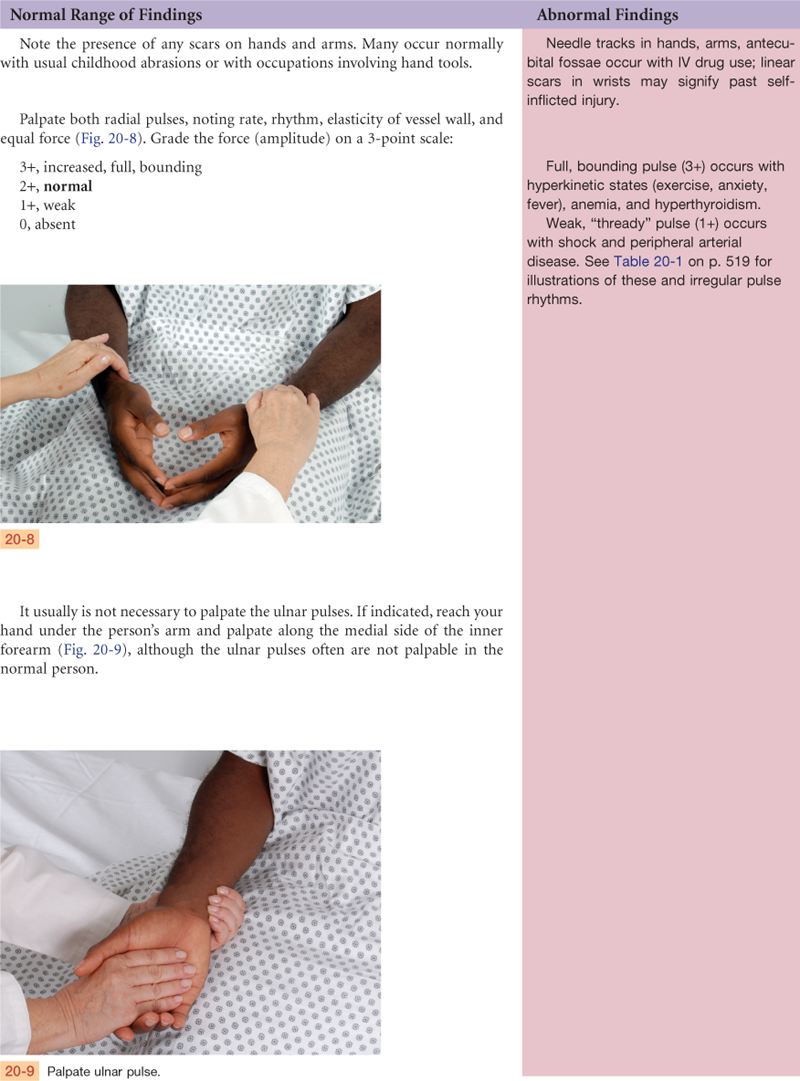
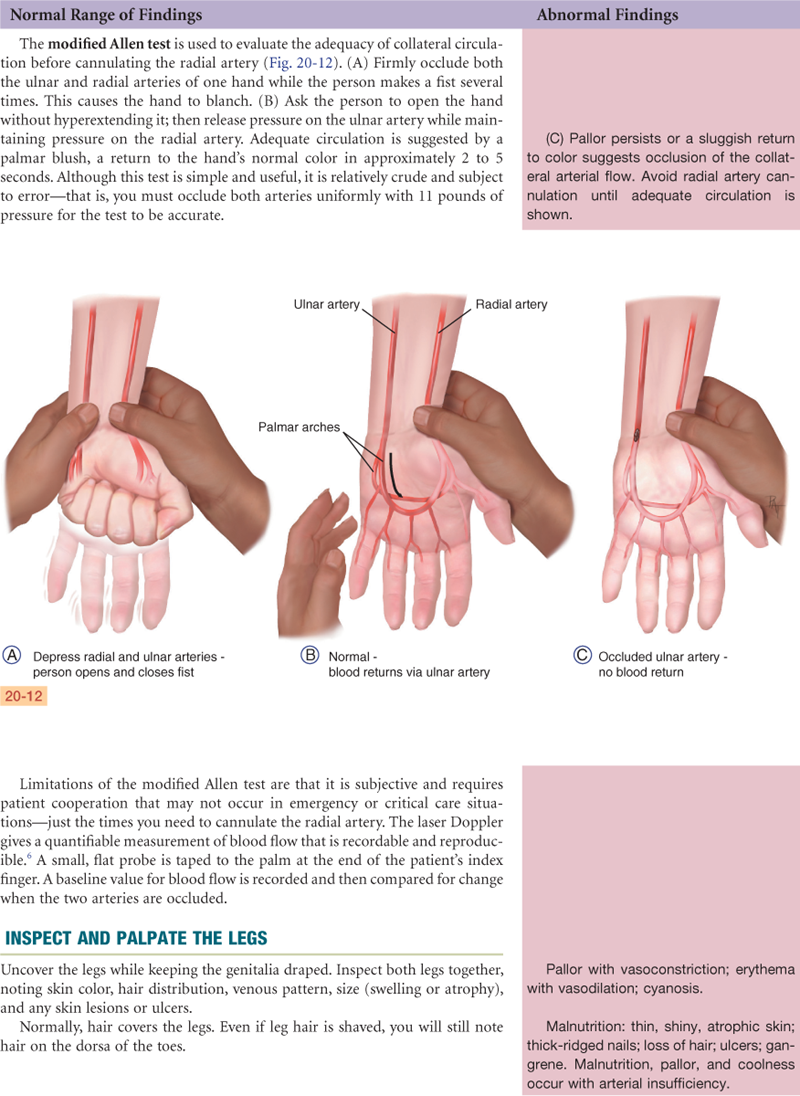
The major artery supplying the arm is the brachial artery, which runs in the biceps-triceps furrow of the upper arm and surfaces at the antecubital fossa in the elbow medial to the biceps tendon (Fig. 20-1). Immediately below the elbow, the brachial artery bifurcates into the ulnar and radial arteries. These run distally and form two arches supplying the hand; these are called the superficial and deep palmar arches. The radial pulse lies just medial to the radius at the wrist; the ulnar artery is in the same relation to the ulna, but it is deeper and often difficult to feel.
20-1 Copyright © (2010) © Pat Thomas, 2010.
Arteries in the Leg
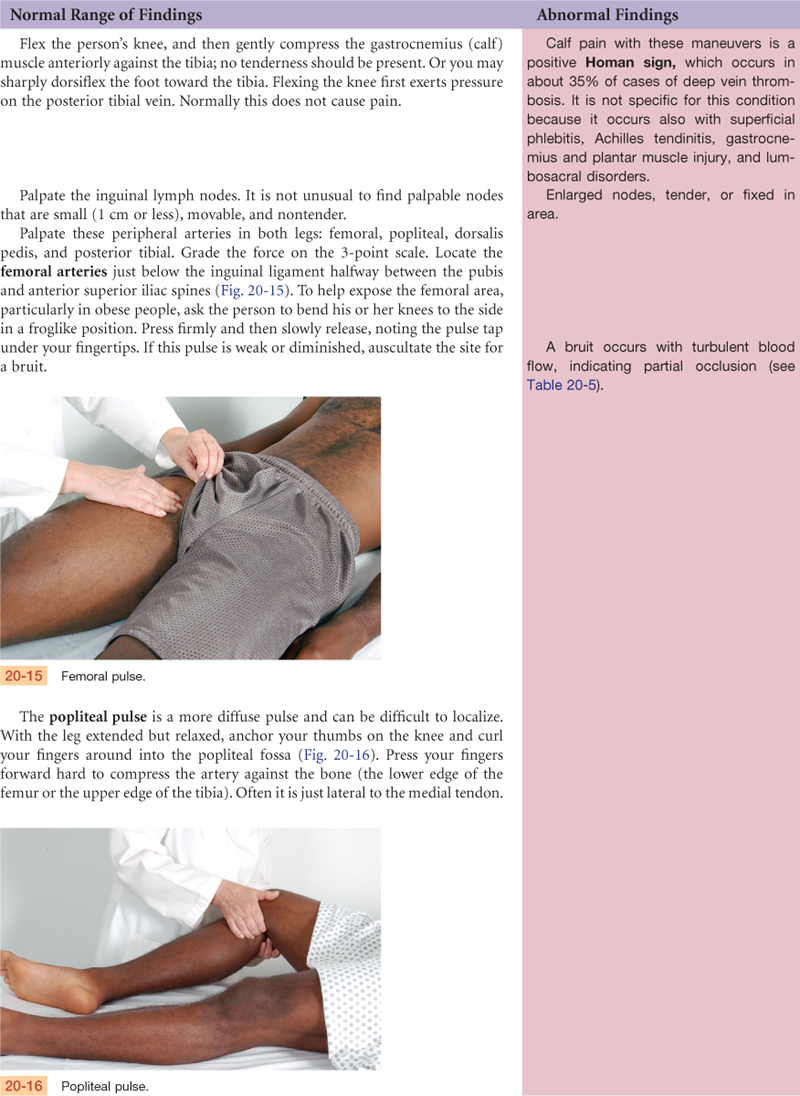
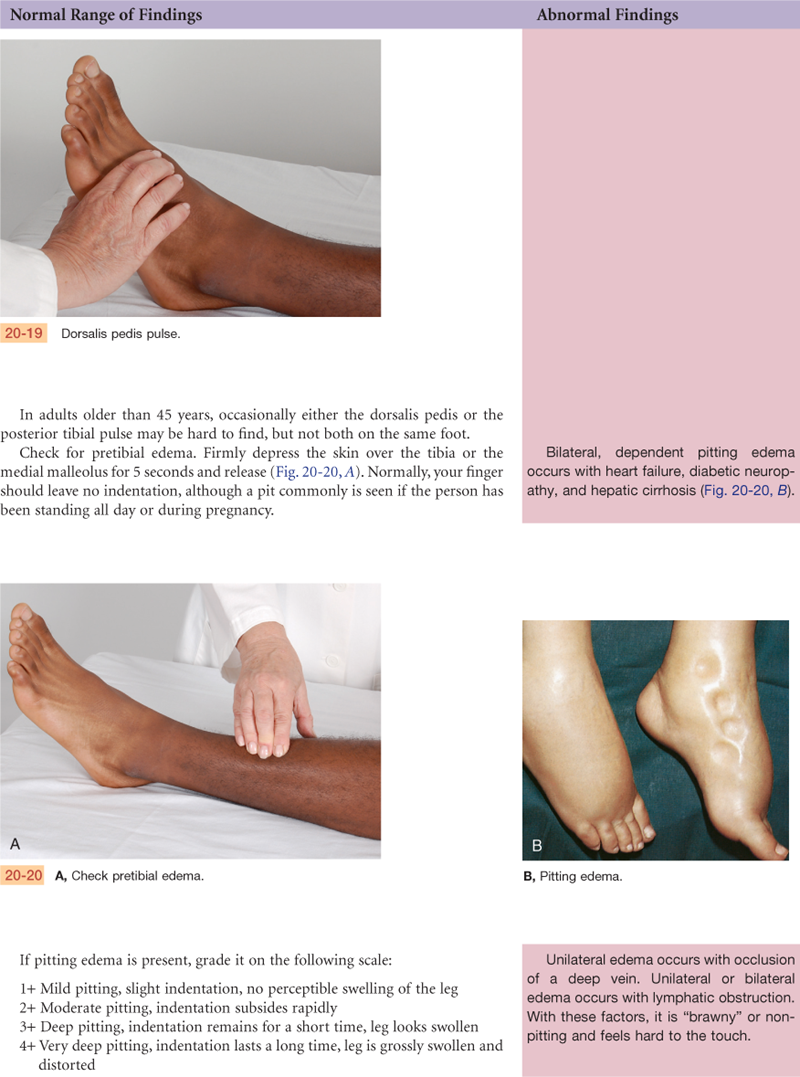
The major artery to the leg is the femoral artery, which passes under the inguinal ligament (Fig. 20-2). The femoral artery travels down the thigh. At the lower thigh, it courses posteriorly; then it is termed the popliteal artery. Below the knee, the popliteal artery divides. The anterior tibial artery travels down the front of the leg on to the dorsum of the foot, where it becomes the dorsalis pedis. In back of the leg, the posterior tibial artery travels down behind the medial malleolus and in the foot forms the plantar arteries.
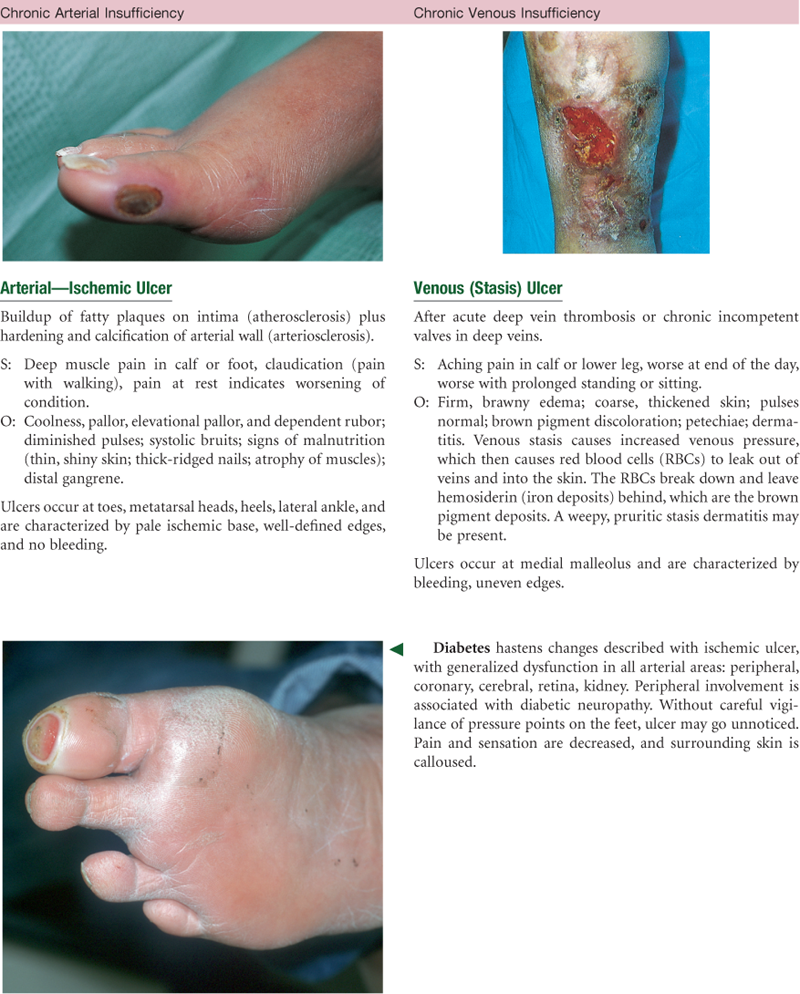
The function of the arteries is to supply oxygen and essential nutrients to the tissues. Ischemia is a deficient supply of oxygenated arterial blood to a tissue caused by obstruction of a blood vessel. A complete blockage leads to death of the distal tissue. A partial blockage creates an insufficient supply, and the ischemia may be apparent only at exercise when oxygen needs increase.
Veins
The course of veins parallels that of arteries, but the body has more veins and they lie closer to the skin surface. The following veins are accessible to examination.
Jugular Veins
Assessment of the jugular veins is presented in Chapter 19.
Veins in the Arm
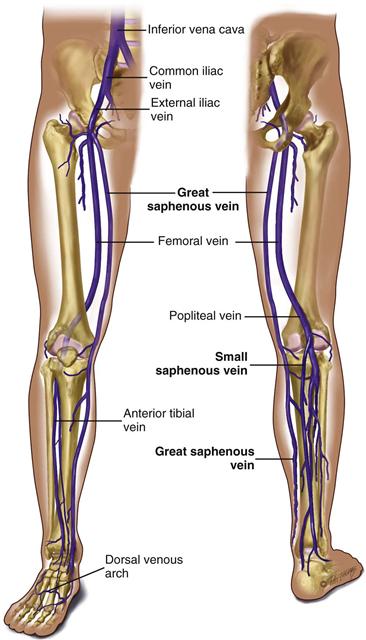
Each arm has two sets of veins: superficial and deep. The superficial veins are in the subcutaneous tissue and are responsible for most of the venous return.
Veins in the Leg
The legs have three types of veins (Fig. 20-3):
1. The deep veins run alongside the deep arteries and conduct most of the venous return from the legs. These are the femoral and popliteal veins. As long as these veins remain intact, the superficial veins can be excised without harming the circulation.
2. The superficial veins are the great and small saphenous veins. The great saphenous vein, inside the leg, starts at the medial side of the dorsum of the foot. You can see it ascend in front of the medial malleolus; then it crosses the tibia obliquely and ascends along the medial side of the thigh. The small saphenous vein, outside the leg, starts on the lateral side of the dorsum of the foot, ascends behind the lateral malleolus, up the back of the leg, where it joins the popliteal vein.
3. Perforators (not illustrated) are connecting veins that join the two sets. They also have one-way valves that route blood from the superficial into the deep veins.
Venous Flow
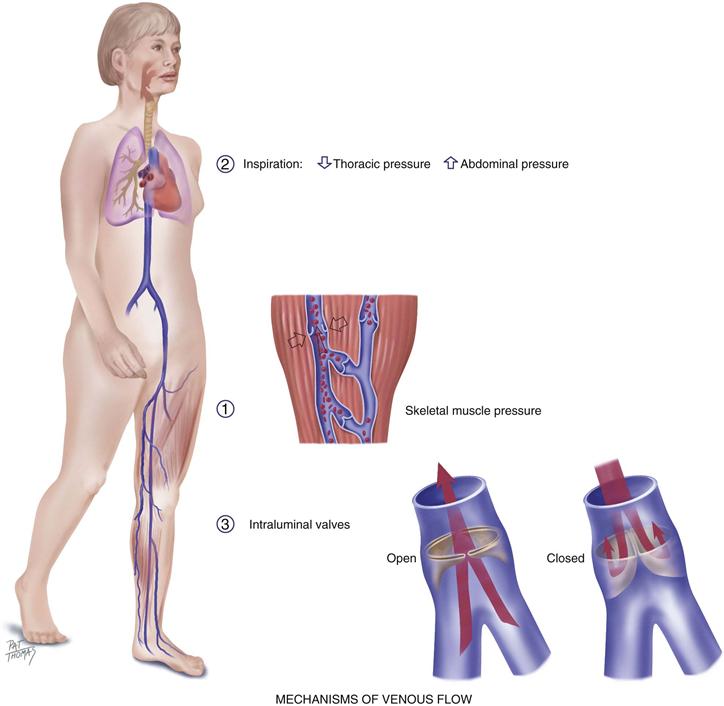
Veins drain the deoxygenated blood and its waste products from the tissues and return it to the heart. Unlike the arteries, veins are a low-pressure system. Because veins do not have a pump to generate their blood flow, the veins need a mechanism to keep blood moving (Fig. 20-4). This is accomplished by (1) the contracting skeletal muscles that milk the blood proximally, back toward the heart; (2) the pressure gradient caused by breathing, in which inspiration makes the thoracic pressure decrease and the abdominal pressure increase; and (3) the intraluminal valves, which ensure unidirectional flow. Each valve is a paired semilunar pocket that opens toward the heart and closes tightly when filled to prevent backflow of blood.
20-4
In the legs, this mechanism is called the “calf pump,” or “peripheral heart.” While walking, the calf muscles alternately contract (systole) and relax (diastole). In the contraction phase, the gastrocnemius and soleus muscles squeeze the veins and direct the blood flow proximally. Because of the valves, venous blood flows just one way—toward the heart.
Besides the presence of intraluminal valves, venous structure differs from arterial structure. Because venous pressure is lower, walls of the veins are thinner than those of the arteries. Veins have a larger diameter and are more distensible; they can expand and hold more blood when blood volume increases. This is a compensatory mechanism to reduce stress on the heart. Because of this ability to stretch, veins are called capacitance vessels.
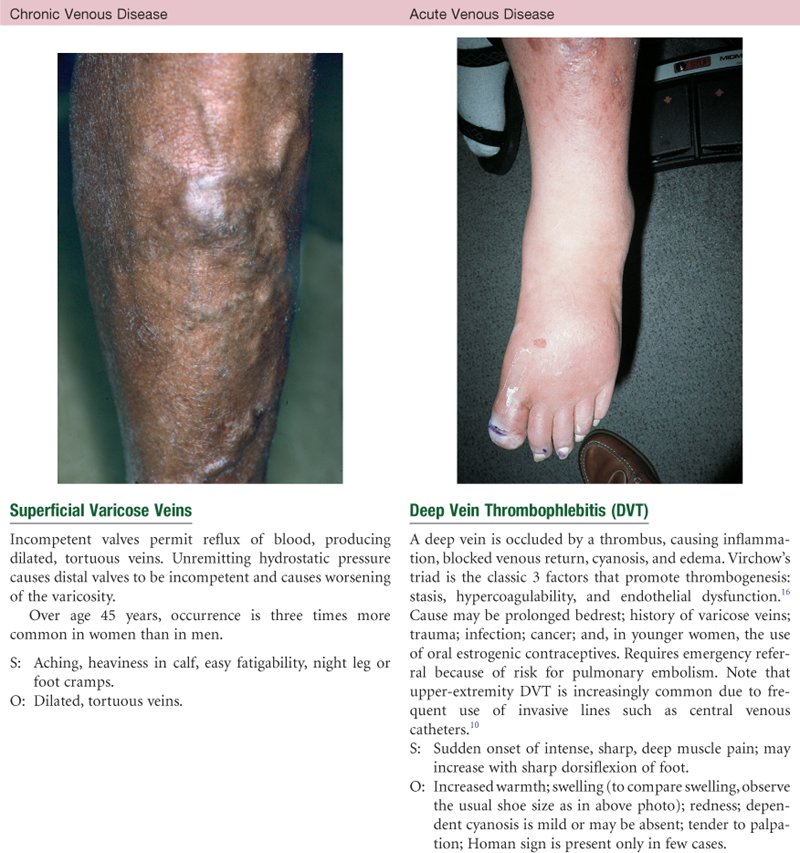
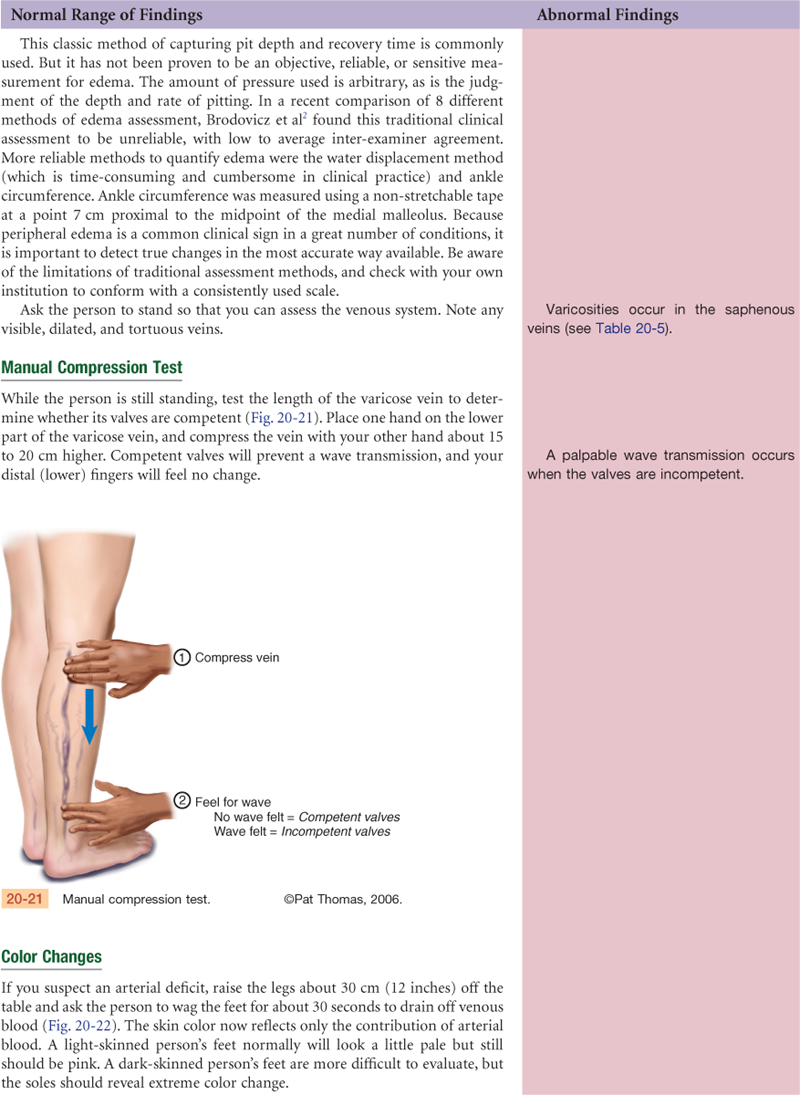
Efficient venous return depends on contracting skeletal muscles, competent valves in the veins, and a patent lumen. Problems with any of these three elements lead to venous stasis. At risk for venous disease are people who undergo prolonged standing, sitting, or bedrest, because they do not benefit from the milking action that walking accomplishes. Hypercoagulable states and vein wall trauma are other factors that increase risk for venous disease. Also, dilated and tortuous (varicose) veins create incompetent valves, wherein the lumen is so wide the valve cusps cannot approximate. This condition increases venous pressure, which further dilates the vein. Some people have a genetic predisposition to varicose veins, but obesity and pregnancy are increased risk factors.
Lymphatics
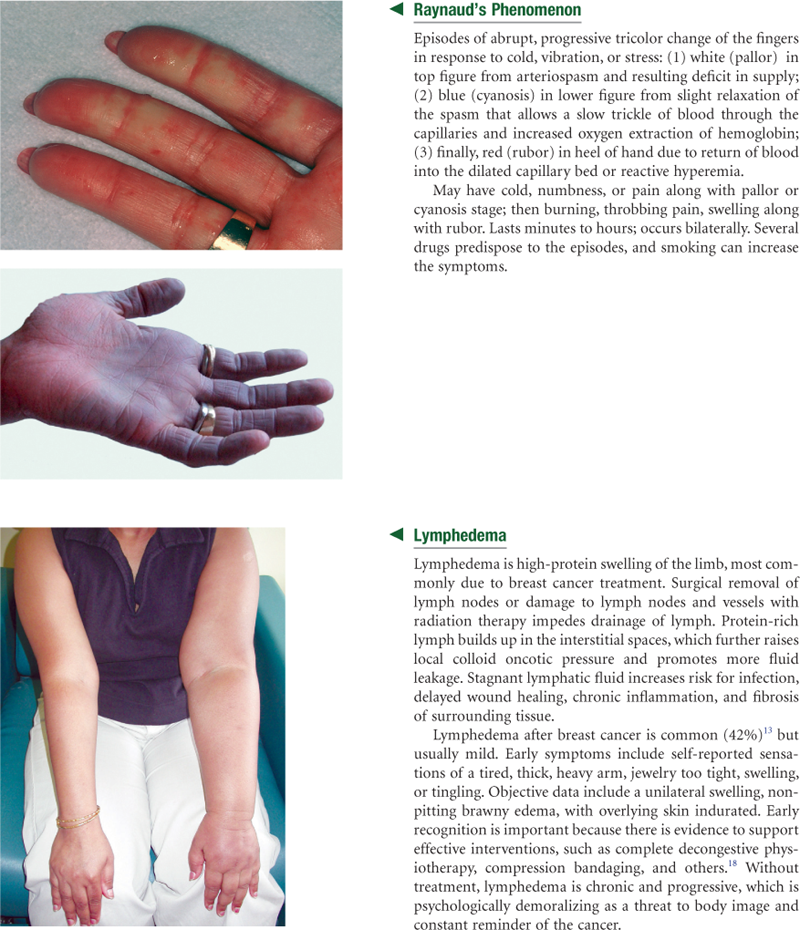
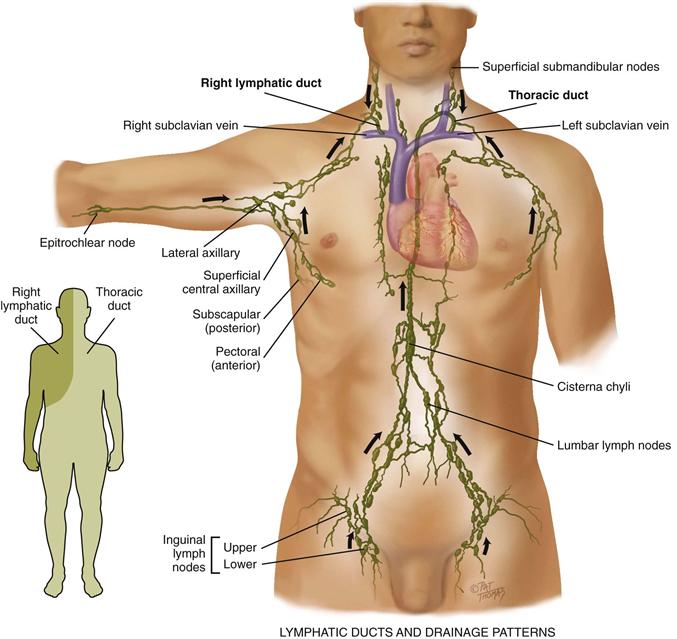
The lymphatics form a completely separate vessel system, which retrieves excess fluid from the tissue spaces and returns it to the bloodstream (Fig. 20-5). During circulation, the blood pressure pushes somewhat more fluid out of the capillaries than the veins can absorb. Without lymphatic drainage, fluid would build up in the interstitial spaces and produce edema.
20-5 Copyright © (2010) © Pat Thomas, 2010.
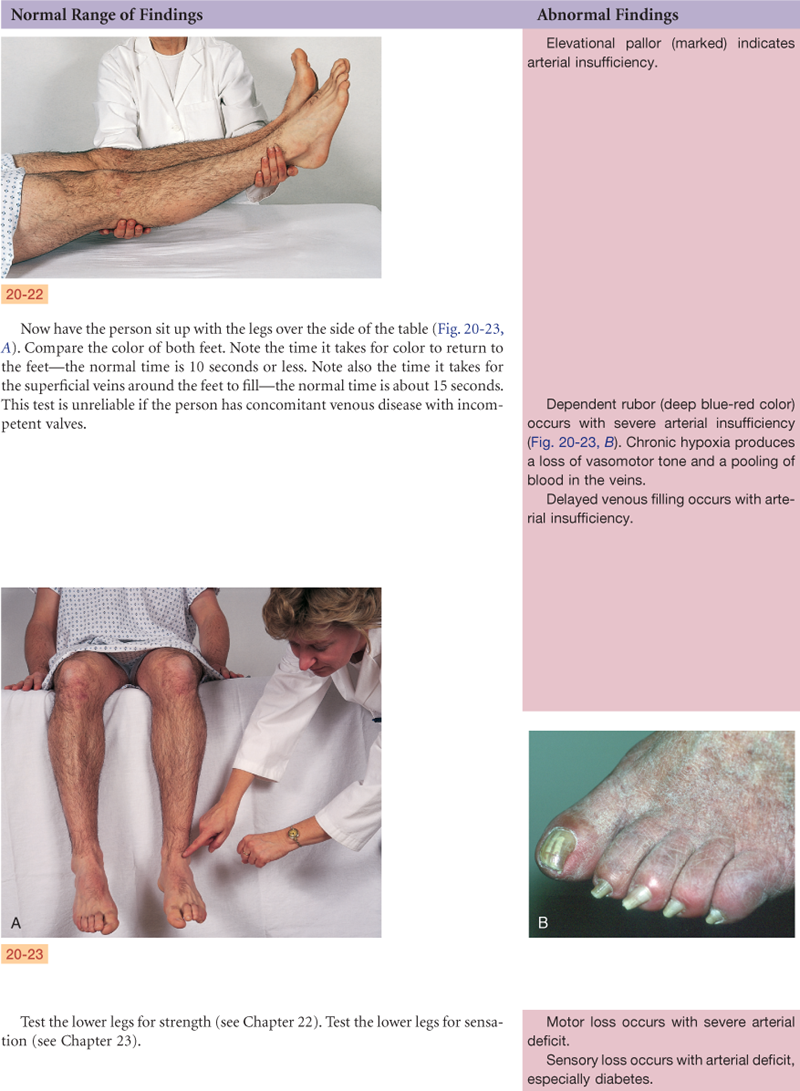
The vessels converge and drain into two main trunks, which empty into the venous system at the subclavian veins (see Fig. 20-5):
1. The right lymphatic duct empties into the right subclavian vein. It drains the right side of the head and neck, right arm, right side of the thorax, right lung and pleura, right side of the heart, and right upper section of the liver.
2. The thoracic duct drains the rest of the body. It empties into the left subclavian vein.
The functions of the lymphatic system are (1) to conserve fluid and plasma proteins that leak out of the capillaries, (2) to form a major part of the immune system that defends the body against disease, and (3) to absorb lipids from the intestinal tract.
The immune system is a complicated network of organs and cells that work together to protect the body. The immune system detects and eliminates foreign pathogens, both those that come in from the environment and those arising from inside (abnormal or mutant cells). It accomplishes this by phagocytosis (digestion) of the substances by neutrophils and monocytes/macrophages and by production of specific antibodies or specific immune responses by the lymphocytes.
The lymphatic vessels have a unique structure. Lymphatic capillaries start as microscopic open-ended tubes, which siphon interstitial fluid. The capillaries converge to form vessels. The vessels, like veins, drain into larger ones. The vessels have valves, so flow is one way from the tissue spaces into the bloodstream. The many valves make the vessels look beaded. The flow of lymph is slow compared with that of the blood. Lymph flow is propelled by contracting skeletal muscles, by pressure changes secondary to breathing, and by contraction of the vessel walls themselves.
Lymph nodes are small, oval clumps of lymphatic tissue located at intervals along the vessels. Most nodes are arranged in groups, both deep and superficial, in the body. Nodes filter the fluid before it is returned to the bloodstream and filter out microorganisms that could be harmful to the body. The pathogens are exposed to B and T lymphocytes in the lymph nodes. The lymphocytes mount an antigen-specific response to eliminate the pathogens. With local inflammation, the nodes in that area become swollen and tender.
The superficial groups of nodes are accessible to inspection and palpation and give clues to the status of the lymphatic system:
• Cervical nodes drain the head and neck and are described in Chapter 13.
• Axillary nodes drain the breast and upper arm. They are described in Chapter 17.
• The epitrochlear node is in the antecubital fossa and drains the hand and lower arm.
• The inguinal nodes in the groin drain most of the lymph of the lower extremity, the external genitalia, and the anterior abdominal wall.
Related Organs
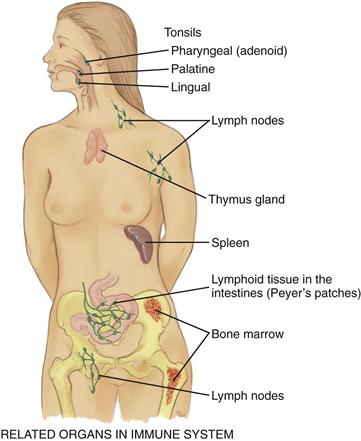
The spleen, tonsils, and thymus aid the lymphatic system (Fig. 20-6). The spleen is located in the left upper quadrant of the abdomen. It has four functions: (1) to destroy old red blood cells; (2) to produce antibodies; (3) to store red blood cells; and (4) to filter microorganisms from the blood.
20-6
The tonsils (palatine, pharyngeal, and lingual) are located at the entrance to the respiratory and gastrointestinal tracts and respond to local inflammation.
The thymus is the flat, pink-gray gland located in the superior mediastinum behind the sternum and in front of the aorta. It is relatively large in the fetus and young child and atrophies after puberty. It is important in developing the T lymphocytes of the immune system in children. The B lymphocytes originate in the bone marrow and mature in the lymphoid tissue.
 Developmental Competence
Developmental Competence
Infants and Children
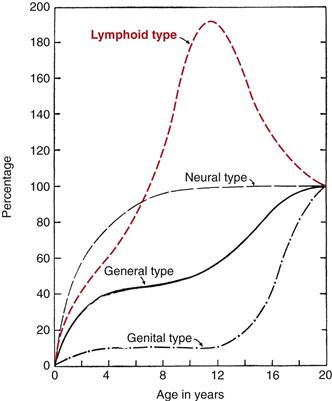
The lymphatic system has the same function in children as in adults. Lymphoid tissue has a unique growth pattern compared with other body systems (Fig. 20-7). It is well developed at birth and grows rapidly until age 10 or 11 years. By age 6 years, the lymphoid tissue reaches adult size; it surpasses adult size by puberty, and then it slowly atrophies. It is possible that the excessive antigen stimulation in children causes the early rapid growth.
Lymph nodes are relatively large in children, and the superficial ones often are palpable even when the child is healthy. With infection, excessive swelling and hyperplasia occur. Enlarged tonsils are familiar signs in respiratory infections. The excessive lymphoid response also may account for the common childhood symptom of abdominal pain with seemingly unrelated problems such as upper respiratory infections. Possibly the inflammation of mesenteric lymph nodes produces the abdominal pain.
The Pregnant Woman
Hormonal changes cause vasodilation and the resulting drop in blood pressure described in Chapter 19. The growing uterus obstructs drainage of the iliac veins and the inferior vena cava. This condition causes low blood flow and increases venous pressure. This, in turn, causes dependent edema, varicosities in the legs and vulva, and hemorrhoids.
The Aging Adult
Peripheral blood vessels grow more rigid with age, resulting in a condition called arteriosclerosis. This condition produces the rise in systolic blood pressure discussed in Chapter 9. Do not confuse this process with another one, atherosclerosis, or the deposition of fatty plaques on the intima of the arteries.
Aging produces a progressive enlargement of the intramuscular calf veins. Prolonged bedrest, prolonged immobilization, and heart failure increase the risk for deep venous thrombosis and subsequent pulmonary embolism. These conditions are common in aging and also with malignancy and myocardial infarction (MI). Low-dose anticoagulant medication reduces the risk for venous thromboembolism.
Loss of lymphatic tissue leads to fewer numbers of lymph nodes in older people and to a decrease in the size of remaining nodes.
Objective Data
| Preparation | Equipment needed |
| During a complete physical examination, examine the arms at the very beginning when you are checking the vital signs—the person is sitting. Examine the legs directly after the abdominal examination while the person is still supine. Then stand the person up to evaluate the leg veins. Examination of the arms and legs includes peripheral vascular characteristics (following here), the skin (see Chapter 12), musculoskeletal findings (see Chapter 22), and neurologic findings (see Chapter 23). A method of integrating these steps is discussed in Chapter 27. Room temperature should be about 22° C (72° F) and draftless to prevent vasodilation or vasoconstriction. Use inspection and palpation. Compare your findings with the opposite extremity. |
Occasionally need: Paper tape measure Tourniquet or blood pressure cuff Stethoscope Doppler ultrasonic stethoscope |






 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
No leg pain, no skin changes, no swelling or lymph node enlargement. No history of heart or vascular problems, diabetes, or obesity. Does not smoke. On no medications.
Objective
Inspection: Extremities have pink-tan color without redness, cyanosis, or any skin lesions. Extremity size is symmetric without swelling or atrophy.
Palpation: Temperature is warm and = bilaterally. All pulses present, 2+ and = bilaterally. No lymphadenopathy.
Assessment
Healthy tissue integrity
Effective tissue perfusion
Focused Assessment: Clinical Case Study
James K. is a 43-year-old, married, white, male city sanitation worker, admitted to University Medical Center today for “bypass surgery tomorrow to fix my aorta and these black toes.”
Subjective
6 years PTA—motorcycle accident with handlebars jammed into groin. Treated and released at local hospital. No apparent injury, although M.D. now thinks accident may have precipitated present stenosis of aorta.
1 year PTA—radiating pain in right calf on walking 1 mile. Pain relieved by stopping walking.
3 months PTA—problems with sex; unable to maintain erection during intercourse.
1 month PTA—leg pain present after walking 2 blocks. Numbness and tingling in right foot and calf. Tips of three toes on right foot look black. Saw M.D. Diagnostic studies showed stenosis of aorta “below vessels that go to my kidneys.”
Present—leg pain at rest, constant and severe, worse at night, partially relieved by dangling leg over side of bed.
Past History—no history of heart or vessel disease, hypertension, diabetes, obesity.
Personal habits—smokes cigarettes, 3 packs per day (PPD) × 23 years. Now cut down to 1 PPD.
Walking is part of occupation, although has been driving city truck past 3 months due to leg pain. On no medications.
Objective
Inspection: Lower extremity size = bilaterally with no swelling or atrophy. No varicosities. Color L leg pink-tan, R leg pink-tan when supine, but marked pallor to R foot on elevation. Black gangrene at tips of R 2nd, 3rd, 4th toes. Leg hair present but absent on involved toes.
Palpation: R foot cool and temperature gradually warms as proceed palpating up R leg.
Pulses: Femorals, both 1+; popliteals, both 0; posterior tibial, both 0 but present with Doppler; dorsalis pedis 0, but left dorsalis pedis is present with Doppler, and right is not present with Doppler.
Assessment
Ischemic rest pain R leg
Ineffective tissue perfusion R/T interruption of flow
Impaired tissue integrity R/T altered circulation
Activity intolerance R/T leg pain
Sexual dysfunction R/T effects of disease
Summary Checklist: Peripheral Vascular Examination
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
Bibliography
1. Aydin A, Shenbagamurthi S, Brem H. Lower extremity ulcers: venous, arterial, or diabetic? Emergency Medicine. 2009;41(8):18–24.
2. Brodovicz KG, McNaugton K, Uemura N, et al. Reliability and feasibility of methods to quantitatively assess peripheral edema. Clinical Medicine & Research. 2009;7(1/2):21–31.
3. Fahey VA. Vascular nursing. 4th ed. Philadelphia: Saunders; 2004.
4. Fu MR, Ridner SH, Armer J. Post–breast cancer lymphedema, Part 1. American Journal of Nursing. 2009;109(7):48–55 2009.
5. Fu MR, Ridner SH, Armer J. Post–breast cancer lymphedema, Part 2. American Journal of Nursing. 2009;109(8):34–42.
6. Fuhrman TM, McSweeney E. Noninvasive evaluation of the collateral circulation to the hand. Academic Emergency Medicine. 1995;2(3):195–199.
7. Goss DE, de Trafford J, Roberts VC, et al. Raised ankle/brachial pressure index in insulin-treated diabetic patients. Diabetic Medicine. 1989;6(7):576–578.
8. Khan NA, Rahim SA, Anand SS, et al. Does the clinical examination predict lower extremity peripheral arterial disease? Journal of the American Medical Association. 2006;295:536–545.
9. Koo LW, Reedy S, Smith JK. Patient history key to diagnosing peripheral edema. Nurse Practitioner. 2010;35(3):44–52.
10. Lancaster SL, Owens A, Bryant AS, et al. Upper-extremity deep vein thrombosis. American Journal of Nursing. 2010;110(5):48–52.
11. Meier AP, Cather JC. Blanched fingers. Proceedings (Baylor University Medical Center). 2010;23(1):73–75.
12. Muir RL. Peripheral arterial disease: pathophysiology, risk factors, diagnosis, treatment, and prevention. Journal of Vascular Nursing. 2009;27(2):26–30.
13. Norman SA, Localio R, Potashnik SL, et al. Lymphedema in breast cancer survivors: incidence, degree, time course, treatment, and symptoms. Journal of Clinical Oncology. 2009;27(3):390–397.
14. Oka RK. Peripheral arterial disease in older adults. Journal of Cardiovascular Nursing. 2006;21:515–520.
15. Ondo WG. Restless legs syndrome. Neurologic Clinics. 2009;27(3):779–799.
16. Osinbowale O, Ali L, Chi YW. Venous thromboembolism: a clinical review. Postgraduate Medicine. 2010;122(2):54–65.
17. Pearson TL. Ankle brachial index as a prognostic tool for women with coronary artery disease. Journal of Cardiovascular Nursing. 2010;25(1):20–24.
18. Poage E, Singer M, Armer J, et al. Demystifying lymphedema: development of the lymphedema putting evidence into practice card. Clinical Journal of Oncology Nursing. 2008;12(6):951–964.
19. Scully MF, Lee AYY. The challenge of cancer and thromboembolic disease: current trends and recommendations. Oncology Exchange. 2009;8(3):6–10.
20. Sieggreen M. A contemporary approach to peripheral arterial disease. Nurse Practitioner. 2006;31:14–26.