Chapter 126 Leukocytosis
Leukocytosis is an elevation in the total leukocyte, or white blood cell (WBC), count that is 2 standard deviations above the mean count for a particular age (Chapter 708). The various causes of leukocytosis are categorized by the class of leukocyte that is elevated and whether the process is acute, chronic, or lifelong. To evaluate the patient with leukocytosis, it is critical to determine which class of WBCs is elevated, and also the duration and extent of the leukocytosis. Each blood count should be evaluated with regard to the absolute number of cells/µL and the normal range for the patient’s age.
A WBC count exceeding 50,000/µL is termed a leukemoid reaction because of the similarity to features of leukemia. Leukemoid reactions are usually neutrophilic and are most frequently associated with septicemia and severe bacterial infections, including shigellosis, salmonellosis, and meningococcemia. Infection in children with leukocyte adhesion deficiency results in WBC counts approaching or exceeding 100,000/µL.
A >5% proportion of immature neutrophil cells is termed a left shift and indicates rapid release of cells from the bone marrow. This release may result in increased circulating band forms, which usually constitute 1-5% of circulating neutrophilic cells, or metamyelocytes and myelocytes, which are not usually found in the peripheral circulation. Higher degrees of left shift with more immature neutrophil precursors are indicative of serious bacterial infections but may also be encountered with trauma, burns, surgery, acute hemolysis, or hemorrhage.
Neutrophilia
Neutrophilia is an increase in the total number of blood neutrophils, which for older children and adults is >8,000/µL. During the 1st day of life, the upper limit of the normal neutrophil count ranges from 7,000 to 12,000/µL. In the 1st mo of life, the neutrophil count ranges from 1,800 to 5,400/µL, and by 1 yr of age, the range is 1,500-8,500/µL.
An increase in circulating neutrophils is a result of a disturbance of the normal equilibrium involving bone marrow neutrophil production, movement out of the marrow compartments into the circulation, and neutrophil destruction. Neutrophilia may arise either alone or in combination with enhanced mobilization into the circulating pool from either the bone marrow storage compartment or the peripheral blood marginating pool, by impaired neutrophil egress into tissues, or by expansion of the circulating neutrophil pool secondary to increased granulocytopoiesis. Myelocytes are not released to the blood except under extreme circumstances.
Acute Acquired Neutrophilia
Neutrophilia is usually an acquired, secondary finding associated with inflammation, infection, injury, or stress. Acute or chronic bacterial infections, trauma, and surgery are among the most common causes encountered in clinical practice. Neutrophilia may also be associated with heatstroke, burns, diabetic ketoacidosis, or any other acute stress. Drugs commonly associated with neutrophilia include epinephrine, corticosteroids, and recombinant growth factors such as recombinant human granulocyte colony-stimulating factor (rhG-CSF) and recombinant human granulocyte-macrophage colony-stimulating factor (rhGM-CSF).
Epinephrine causes release into the circulation of a sequestered pool of neutrophils that normally marginate along the vascular endothelium. Corticosteroids accelerate the release of neutrophils and bands from a large storage pool within the bone marrow and impair the migration of neutrophils from the circulation into tissues. Acute neutrophilia in response to inflammation and infections occurs because of release of neutrophils from the marrow storage pool. The post-mitotic marrow neutrophil pools are approximately 10 times the sizes of the blood neutrophil pool, and about half of these cells are bands and segmented neutrophils. In neutrophil production disorders, such as those associated with malignancies and cancer chemotherapy, the size of this pool may be reduced and the capacity to develop neutrophilia remains impaired. Exposure of blood to foreign substances such as hemodialysis membrane activates the complement system and causes transient neutropenia followed by neutrophilia because of release of bone marrow neutrophils. The colony-stimulating factors G-CSF and GM-CSF cause acute and chronic neutrophilia by mobilizing cells from the marrow reserves and by stimulating neutrophil production (Fig. 126-1).
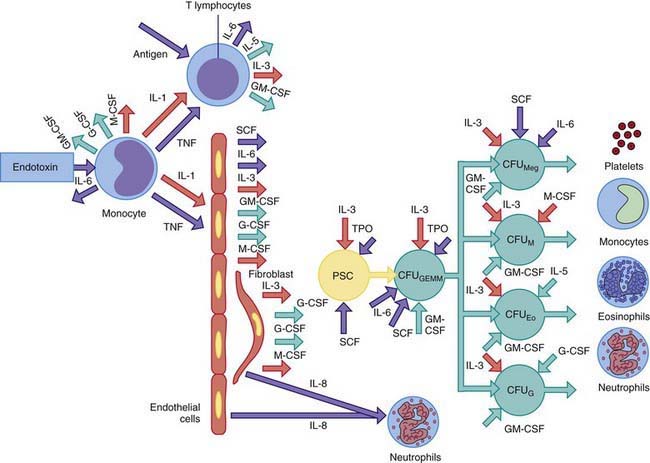
Figure 126-1 Cytokine control of the phagocyte production and activation by release of endotoxin during infection. Both endothelial cells and fibroblasts release substantial quantities of both granulocyte-macrophage colony–stimulating factor (GM-CSF) and granulocyte colony–stimulating factor (G-CSF) in response to tumor necrosis factor (TNF) and interleukin 1 (IL-1). Both TNF and IL-1 are released from endotoxin-activated monocytes and antigen-activated T lymphocytes. Stem cell factor (SCF), interleukin 3 (IL-3), GM-CSF, and interleukin 6 (IL-6) each influence the growth and differentiation of multilineage progenitor cells known as colony forming units (CFU)—granulocytes, eosinophils, monocytes, megakaryocytes (CFUGEMM)—to the committed colony forming unit macrophage (CFUM), CFU eosinophil (CFUEo), or CFU granulocyte (CFUG). Platelet production can also be augmented by expansion of CFU megakaryocyte (CFUmeg). Factors that serve as growth and differentiation factors for a specific lineage also act as activation factors for the terminally differentiated forms of the same lineage. Macrophage colony–stimulating factor (M-CSF) enhances antibody-mediated cytotoxicity and phagocyte functions of monocytes. G-CSF promotes neutrophil bactericidal capacity, and interleukin 5 (IL-5) activates eosinophil function. Within hours of an infection, cytokine primed neutrophils move to the invasion site from the circulation under the influence of interleukin 8 (IL-8), which is released from endothelial cells and fibroblasts.
Chronic Acquired Neutrophilia
Chronic neutrophilia is usually associated with continued stimulation of neutrophil production resulting from persistent inflammatory reactions or chronic infections (e.g., tuberculosis), vasculitis, postsplenectomy states, Hodgkin disease, chronic myelogenous leukemia, chronic blood loss, sickle cell disease, some chronic hemolytic anemias, and prolonged administration of corticosteroids. Chronic neutrophilia can arise after expansion of cell production secondary to stimulation of cell divisions within the mitotic precursor pool, which consists of promyelocytes and myelocytes (see Fig. 126-1). Subsequently, the size of the post-mitotic pool increases. These changes lead to an increase in the marrow reserve pool, which can be readily mobilized for release of neutrophils into the circulation. The neutrophil production rate can increase greatly in response to exogenously administered hemopoietic growth factors, such as rhG-CSF, with a maximum response taking at least 1 wk to develop.
Lifelong Neutrophilia
Congenital asplenia is associated with lifelong neutrophilia. Uncommon genetic disorders that present with neutrophilia include leukocyte adhesion deficiency, familial myeloproliferative disease, Down syndrome, and Rac-2 mutation (Chapter 124). In an autosomal dominant form of hereditary neutrophilia, patients maintain an absolute neutrophil count between 1,400 and 150,000/µL, which is associated with hepatosplenomegaly, an increased alkaline phosphatase level, and Gaucher-type histiocytes in the bone marrow.
Evaluation of persistent neutrophilia requires a careful history, physical examination, and laboratory studies to search for infectious, inflammatory, and neoplastic conditions. The leukocyte alkaline phosphatase cytochemical stain of circulating neutrophils is useful to differentiate chronic myelogenous leukemia, in which the level is uniformly near zero, from reactive or secondary neutrophilia, in which normal to elevated levels are found.
Monocytosis
The average absolute blood monocyte count varies with age, which must be considered in the assessment of monocytosis. Given the role of monocytes in antigen presentation and cytokine secretion and as effectors of ingestion of invading organisms, it is not surprising that many clinical disorders give rise to monocytosis (Table 126-1). Most commonly, monocytosis occurs in patients recovering from myelosuppressive chemotherapy and is a harbinger of the return of the neutrophil count to normal. Monocytosis is occasionally a sign of an acute bacterial, viral, protozoal, or rickettsial infection, and may also occur in some forms of chronic neutropenia and postsplenectomy states. Chronic inflammatory conditions can stimulate sustained monocytosis, including preleukemia, chronic myelogenous leukemia, lymphomas, and occasionally Hodgkin disease.
Table 126-1 CAUSES OF MONOCYTOSIS
INFECTIONS
Bacterial Infections
Tuberculosis
Brucellosis
Typhoid fever
Syphilis
Infective endocarditis
Nonbacterial Infections
Fungal infections
Rocky Mountain spotted fever
Typhus
Kala-azar
Malaria
HEMATOLOGIC DISORDERS
Postsplenectomy states
Congenital and acquired neutropenias
Hemolytic anemias
MALIGNANT DISORDERS
Preleukemia
Acute myelogenous leukemia
Chronic myelogenous leukemia
Juvenile myelomonocytic leukemia
Hodgkin disease
Non-Hodgkin lymphomas
CHRONIC INFLAMMATORY DISEASES
Systemic lupus erythematosus
Rheumatoid arthritis
Polyarteritis nodosa
Inflammatory bowel disease
Sarcoidosis
MISCELLANEOUS
Cirrhosis
Recovery from marrow suppression induced by chemotherapy
Drug reactions
Lymphocytosis
The most common cause of lymphocytosis is an acute viral illness. Lymphocytosis is a normal response to most viral infections because the majority of circulating lymphocytes are T cells. In infectious mononucleosis, the B cells are infected with the Epstein-Barr virus and the T cells react to the viral antigens present in the B cells, resulting in atypical lymphocytes with the typical large, vacuolated morphology. Other viral infections classically associated with lymphocytosis are cytomegalovirus and viral hepatitis. Chronic bacterial infections such as tuberculosis and brucellosis may lead to a sustained lymphocytosis. Pertussis is accompanied by lymphocytosis in ≈25% of infants infected before 6 mo of age. Thyrotoxicosis and Addison disease are endocrine disorders associated with lymphocytosis. Persistent or profound lymphocytosis suggests acute lymphocytic leukemia.
Basophilia
Basophilia occurs when the basophil count exceeds 100-120 cells/µL. Basophilia is a nonspecific sign of a wide variety of disorders and is usually of limited diagnostic importance. Basophilia is most often present in hypersensitivity reactions and frequently accompanies the leukocytosis of chronic myeloid leukemia.
Boxer LA. Leukocytosis. In: Sills RH, editor. Practical algorithms in pediatric hematology and oncology. New York: Karger; 2003:38-39.
Dinauer MC, Newburger PE. The phagocyte system and disorders of granulopoiesis and granulocyte function. In: Orkin SH, Ginsburg D, Nathan DG, et al, editors. Nathan and Oski’s hematology of infancy and childhood. ed 7. Philadelphia: Saunders/Elsevier; 2009:1109-1217.