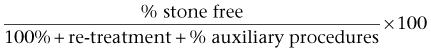chapter 48 Surgical Management of Upper Urinary Tract Calculi
Historical Overview
Kidney Calculi
Although calculi in the kidney were rare before the Industrial Revolution (Shah and Whitfield, 2002), the existence of nephrolithiasis was known to Hippocrates, who described the symptoms of renal colic: “An acute pain is felt in the kidney, the loins, the flank and the testis of the affected side; the patient passes urine frequently; gradually the urine is suppressed. With the urine, sand is passed.” It is not certain whether Hippocrates actually performed surgery on patients with renal calculi, but he did describe the following operations: the drainage of tuberculous and nontuberculous pyelonephritic abscesses, the incision of swelling in the loin due to renal tumefaction resulting from stone, and the drainage of kidneys presenting with acute congestion caused by pyelonephritis (Wershub, 1970). In the centuries that followed Hippocrates there was little scientific progress in the surgical therapy for patients with renal calculi. The alleged first account of a surgical attempt to remove a stone from a patient’s kidney is the case of the French archer of Bagnolet. Little is known of the authenticity of this tale of a condemned man afflicted with a renal calculus who agreed to allow surgery on the affected kidney with the condition that if he survived he would be freed. According to the anecdote the man survived the open surgical stone removal and was freed in 1474 (Herman, 1973). Unfortunately there are no first-hand records of this event so the veracity of this claim is uncertain. The first verifiable account of renal stone surgery was in 1550, when Cardan of Milan opened a lumbar abscess on a young girl and removed 18 calculi (Desnos, 1972). For the following 2 centuries most surgeons were in agreement that the only indication for open renal surgery was the infected calculous kidney, distended by the accumulation of purulent matter, or those kidneys in which the calculus could be palpated in the organ itself.
In 1734 Lafite incised a swelling in a patient’s loin and drained considerable purulence. Twenty-two days later the pus reaccumulated; he probed the incision and found a stone in the region of the kidney. Lafite widened the prior incision and removed two calculi; the patient recovered well. Four years later Lafite again removed stones from a man who had undergone drainage of a lumbar swelling 11 years before and who had a persistent urinary fistula. Lafite concluded that it was possible to remove the stones at the time of the first surgical intervention rather than subject the patient to multiple procedures (Ballenger et al, 1933). In 1872, William Ingalls of Boston City Hospital removed a large calculus from the right kidney of a 31-year-old woman with a persistent pyelocutaneous fistula (Spirnak and Resnick, 1983). Ingalls incised the sinus tract of the fistula and extracted the stone with forceps, thus performing the first recorded nephrolithotomy in America. In 1880, Henry Morris of England was the first to remove a stone from an otherwise healthy kidney by nephrolithotomy, extracting a 31-g mulberry calculus from the kidney of a young woman (Dudley, 1973).
As the surgical techniques of nephrolithotomy evolved, renal parenchymal incisions were made in a variety of different ways in an effort to reduce hemorrhagic morbidity. Heineke, in 1879, first described a pyelotomy incision for the extraction of calculi. The operation rapidly found favor and was employed by many surgeons, although it was not possible to extend the incision to permit extraction of large renal calculi without damaging the retropelvic renal artery (Wershub, 1970). Josef Hyrtl, in 1882, and Max Brödel, in 1902, described a relatively avascular plane near the midline (5 mm posterior) of the convex border of the kidney through which the collecting system of the kidney could be entered. In continental Europe, credit for the plane was given to Hyrtl; but in England and the United States it was called the Brödel bloodless line or the Brödel white line (Schultheiss et al, 2000). Although the existence of this avascular plane was an important discovery, surgeons continued to find that bleeding during nephrolithotomy was a considerable problem. Zuckerkandl described an inferior pyelonephrolithotomy in which a pyelotomy incision was extended into the lower pole of the kidney. Partner recommended a V-shaped incision with two limbs radiating toward the poles of the kidney. Other attempts were made to control the persistent problem of bleeding, including compression of the hilar vessels and various methods of suturing. In 1887, Czerny was the first to approximate the cut edges of the incised kidney with suture to control hemorrhage and to prevent fistula formation. In the same year, Guyon reported that nephrectomy, although efficacious in curing patients suffering from calculous pyonephrosis, was more dangerous than nephrolithotomy because lithiasis was often bilateral (Wershub, 1970). In 1889 Kümmell was the first surgeon to perform a partial nephrectomy for calculous pyonephrosis (Redman, 1983). Lower, in 1913, revived interest in pyelolithotomy when he suggested that this technique may be a safer and easier method of removing renal calculi than nephrolithotomy. Although several small series of cases indicated that there might be a higher incidence of stone recurrence after pyelolithotomy, other studies showed that recurrence was no more common than it was after nephrolithotomy (Murphy, 1972). These findings, in conjunction with rapid advancements in the field of radiography, brought about a decided preference for pyelolithotomy (Gil-Vernet and Culla, 1981). In 1943 Dees and Fox reported the first use of coagulum to remove small stones and stone fragments from the renal pelvis and calyces (Marshall, 1983). Fibrinogen and thrombin were used to make a coagulum that was injected into the renal pelvis and produced a flexible cast of the pelvis and calyces. The use of this technique was limited initially owing to the scarcity of materials and the risk of blood-borne disease transmission. However, interest in coagulum pyelolithotomy was renewed when cryoprecipitate was found to be a safe and readily available source of concentrated fibrinogen (Fischer et al, 1980).
An important advance in the open surgical approach to the kidney was the intrasinusally extended pyelolithotomy, pioneered by Gil-Vernet in 1965. Because of its wide applicability and minimal morbidity this approach to the renal collecting system became the procedure of choice for treatment of the majority of renal pelvic calculi. Patients harboring large or complex calculi could be effectively treated with extended pyelolithotomy combined with multiple radial nephrotomies (Wickham et al, 1974). In 1968 Smith and Boyce described anatrophic nephrolithotomy, a procedure that derived its name from the technique of incising the renal parenchyma along the avascular plane between the anterior and posterior vascular distributions. Because an incision in this plane does not interrupt the blood supply to the renal parenchyma it does not result in atrophy, hence the term anatrophic. This procedure permits a relatively bloodless operation that encompasses stone removal, reconstruction of the calyceal system, and closure of the renal capsule with preservation of renal function. Although stone-free rates of these modern surgical techniques were excellent, morbidity was significant, and the search for new techniques and technologies continued.
Ureteral Calculi
Ambroise Paré is credited with the first account of a ureteral calculus, when, in 1564, he described “the cruel pain [that] tormented the patient in that place where the stone lodged.” Paré also stated that death was the consequence of having calculi impacted in both ureters (Murphy, 1972). Morris recounted that surgical intervention was an option in the treatment of ureteral stones when he reported in 1898 that “operations on the ureter are an advance of the last few years, but not many have been recorded up to the present time” (Ballenger et al, 1933). Thomas Emmet, of New York, published an account in 1879 of three female patients with stones impacted at the distal aspect of the ureter. In one patient Emmet opened the bladder and removed the stone with forceps; in a second patient he removed a stone by cutting down on it through the vaginal wall. These procedures were the first records of a surgeon making a definite diagnosis of ureteral calculus and deliberately and successfully performing a ureterolithotomy. In the years that followed, intraperitoneal, perineal, sacral, transrectal, and transvaginal approaches were used. In 1910 Gibson, of New York, described an incision parallel to and just above the Poupart ligament, wholly extraperitoneal, by which the lower ureter, even down to its entrance into the bladder, could be readily exposed. This safe and comparatively easy approach to the ureter gave open ureterolithotomy a solid basis for success.
The Rise of Endourology
Before the development of endoscopy attempts to blindly extract calculi were not uncommon. In 1889, Gustav Kolisher performed the first successful stone manipulation, reporting that he “located the stone with a metal-tipped catheter several inches above the ureteric orifice and through it injected 30 cc of sterile oil,” displacing the stone (Murphy, 1972).
The development of minimally invasive surgical techniques for the treatment of patients suffering from urinary lithiasis has been greatly dependent on technologic advances in the fields of fiberoptics, radiographic imaging, and lithotripsy (shockwave, ultrasonic, electrohydraulic, and laser). These advancements have accelerated the evolution of modern techniques of calculus removal, including ureteroscopy, percutaneous nephrolithotomy (PNL), and extracorporeal shockwave lithotripsy (SWL). In 1979 Arthur Smith defined the term endourology as closed controlled manipulation within the genitourinary tract (Smith et al, 1979).
Ureteroscopy
The practice of ureteroscopy began by happenstance when, in 1912, Hugh Hampton Young introduced a pediatric cystoscope into the massively dilated ureter of a child with posterior urethral valves (Young and McKay, 1929). Aided by the child’s secondary ureteral dilation, Young was able to advance the cystoscope to the level of the renal pelvis, thus becoming the first urologist to view the intrarenal collecting system endoscopically. Unfortunately the following 3 decades held few significant advances in ureteroscopic technology until knowledge of fiberoptics could be put to clinical use. By 1957 Curtiss and Hirschowitz combined a large number of glass fibers into a coherent bundle and fused the fibers at their ends to allow them to move individually along their length, thus creating the first flexible endoscope (Hirschowitz et al, 1957). In 1964 Marshall reported the first urologic use of this new type of flexible endoscope when he passed the scope through an open ureterotomy to the level of the renal pelvis, thereby performing the first flexible ureteroscopy. Subsequently, two of his associates, McGovern and Walzak, performed the first transurethral flexible ureteroscopy when they passed the same 9-Fr flexible endoscope to inspect a ureteral calculus. Since then, developments in optics and mechanics have greatly improved the design of flexible ureteroscopes.
Currently available ureteroscopes range from 54 to 70 cm in length and have a tapered shaft diameter that increases proximally. As the tip of the ureteroscope is inserted into the ureter and passed retrograde, the ureter is slowly dilated. Initial ureteroscopes had neither a working nor an irrigating channel. Most modern ureteroscopes have a single working channel, and some have a second irrigation channel that serves to distend the ureter and maintain visualization.
Early flexible ureteroscopes were passive, and the subsequent incorporation of active tip deflection has greatly increased their utility. Active deflection refers to deflection of the tip of the endoscope, which is controlled by the surgeon through a lever mechanism on the handle of the endoscope. Flexible ureteroscopes have been introduced with two segments of active deflection, with the active primary site of deflection providing 170 to 180 degrees of up and down movement; the secondary active deflection, located several centimeters proximal to the primary deflection, is a 130-degree one-way downward deflection. This design greatly facilitates entry into the lower pole infundibulum. Other manufacturers have designed flexible ureteroscopes with 270 degrees of deflection, which also facilitates entry into the lower pole infundibulum.
More recently, efforts have been devoted to advancing the imaging capability of the flexible ureteroscope. Digital endoscopes, which incorporate an optical chip [complementary metal oxide semiconductor [CMOS] or charge-coupled device [CCD]) at the tip have been introduced. Although the initial generation of these chips were quite large, further refinements have reduced their size so that they can be applied to flexible ureteroscopes. Advantages associated with this technology include improved optical characteristics, obviating focus and white-balancing issues, and decreased surgeon fatigue, because cumbersome proximal camera and light cord attachments are not required. Image processing software permits digital zoom capability as well. In comparing digital flexible ureteroscopy to conventional fiberoptic flexible ureteroscopy, investigators have noted that the digital devices are associated with superior imaging characteristics (Humphreys et al, 2008). Several disadvantages of the digital device must be recognized, however: digital ureteroscopes are larger in diameter compared with their fiberoptic counterparts, and the technology also is more costly.
Interestingly, the first reports of rigid ureteroscopy trailed those of flexible ureteroscopy by almost 10 years. In 1977 Goodman reported on three cases in which a pediatric cystoscope was used to treat patients with ureteral maladies. These initial rigid ureteroscopes employed a rod-lens system that was large (10 to 13 Fr) and inflexible. Most rigid ureteroscope designs have replaced this rod-lens system of image transmission with fiberoptics, which allows significant reduction in the size of the endoscope. In addition, the flexibility of the fiberoptic bundles allows the shaft of the endoscope to become somewhat bendable along its vertical axis, hence the term semirigid ureteroscope.
Parallel to improvements in rigid and flexible ureteroscopes were advances in intracorporeal lithotripters, including ultrasonic, electrohydraulic, pneumatic, and laser probes, allowing efficient stone fragmentation through the miniaturized modern ureteroscopic equipment. Many new stone retrieval devices, designed to pass through the working channel of a ureteroscope, have been introduced with the capability for manipulation and deflection.
Percutaneous Stone Removal
The first description of percutaneous stone removal was that of Rupel and Brown (1941) of Indianapolis, who removed a stone through a previously established surgical nephrostomy. It was not until 1955, however, that Goodwin and associates described the first placement of a percutaneous nephrostomy tube to drain a grossly hydronephrotic kidney. These researchers did not have the benefit of radiographic guidance, and so the drainage tube was placed without imaging. In 1976, Fernstrom and Johannson first reported the establishment of percutaneous access with the specific intention of removing a renal stone. Subsequent advances in endoscopes, imaging equipment, and intracorporeal lithotripters allowed urologists and radiologists to refine these percutaneous techniques through the late 1970s and early 1980s into well-established methods for removal of upper urinary tract calculi.
Extracorporeal Shockwave Lithotripsy
The phenomenon that sound waves can be focused has been known since antiquity. The ancient Greeks, as taught by Dionysius, used this knowledge to construct vaults that allowed them to overhear the conversations of their imprisoned enemies. In the 18th and 19th centuries, cabinets constructed with echo or sound mirrors were capable of transmitting the ticking of a pocket watch over a distance exceeding 60 feet.
High-energy shockwaves, too, have been recognized for many years. Examples of high-energy shockwaves include the blast effect associated with explosions, as well as the potentially window-shattering sonic boom created when aircraft pass beyond the speed of sound. Engineers at Dornier Medical Systems in what was then West Germany, during research on the effects of shockwaves on military hardware, demonstrated that these shockwaves are reflectable and therefore focusable. The possibility of applying shockwave energy to human tissue was discovered when, by chance, a test engineer touched a target body at the very moment of impact of a high-velocity projectile. The engineer felt a sensation similar to an electric shock, although the contact point at the skin showed no damage at all (Hepp, 1984). This observation and its potential military applications led Dornier to pursue a method of generating a reproducible shockwave.
Beginning in 1969 and funded by the German Ministry of Defense, Dornier began a study of the effects of shockwaves on tissue. Specifically, the study was to determine if the shockwaves generated by a projectile striking the wall of a military tank would damage the lungs of a crew member leaning against the same wall. During the study, Dornier engineers developed techniques to reproducibly generate shockwaves. In the course of this effort the engineers discovered that shockwaves generated in water could pass through living tissue (except for the lung) without discernible damage to the tissue but that brittle materials in the path of the shockwaves would be fragmented.
At some point a possible medical application of shockwaves became apparent: if shockwaves could safely pass through tissue but fragment brittle materials, perhaps they could be used to break up kidney stones. Dornier engineers found that lower-energy shockwaves, which would be appropriate for medical applications, could be generated in a predictable and reproducible manner by an underwater electrical spark discharge.
In 1972, on the basis of preliminary studies performed by Dornier Medical Systems, an agreement was reached with Egbert Schmiedt, director of the urologic clinic at the University of Munich, to proceed with further investigation of the therapeutic potential of this technology (Chaussy and Fuchs, 1986). This research was supported by the West German Federal Ministry of Research and Technology, and the development of the Dornier lithotripter progressed through several prototypes, ultimately culminating in February 1980 with the first treatment of a human by SWL. The production and distribution of the Dornier HM3 lithotripter began in late 1983, and SWL was approved by the U.S. Food and Drug Administration in 1984. Since Dornier’s pioneering work, numerous other companies have demonstrated that shockwaves capable of stone fragmentation may be generated by electromagnetic induction, microexplosions, focused lasers, and piezoelectric crystals. To date, more than 3000 lithotripters of all types have been placed worldwide, and more than 1 million patients are treated annually with SWL.
Renal Calculi
The primary goal of surgical stone management is to achieve maximal stone clearance with minimal morbidity to the patient. The introduction of SWL as well as continuing advancements in the field of endourology have allowed most patients with renal stones to be treated in a minimally invasive fashion. However, as the armamentarium of treatment modalities available to the urologist has increased, new controversies regarding the indications for these therapies have developed. Currently, urologists face the challenge of selecting the optimal treatment modality on the basis of the patient’s and the stone’s characteristics. Four minimally invasive treatment modalities are available for the treatment of patients with kidney stones and are discussed in this chapter: SWL, PNL, ureteroscopy, and laparoscopic stone surgery. SWL has also been used in combination with PNL (“sandwich technique”) as well as with ureteroscopic lithotripsy. Advances in endoscopic technology and surgical technique have dramatically reduced the need for open surgical procedures to treat patients with renal and ureteral calculi.
Most patients harboring “simple” renal calculi can be treated satisfactorily with SWL. However, there are other patients who are unlikely to achieve a successful outcome with SWL; factors associated with poor stone clearance rates after SWL include large renal calculi, stones within dependent or obstructed portions of the collecting system, stones of certain composition (cystine, calcium oxalate monohydrate, and brushite), and obesity or a body habitus that inhibits imaging and targeting of the stone. For patients with these clinical characteristics, alternative treatment modalities, such as ureteroscopy or PNL, should be considered. The urologist, then, when treating a patient with a renal calculus, must ask: Is the patient an appropriate candidate for SWL, or should other treatment modalities be used?
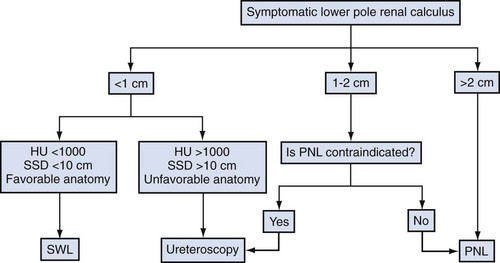
Stone-related factors (size, number, location, composition), renal anatomy, and clinical factors of the patient (Table 48–1) as well as the morbidity inherent in each treatment modality and the availability of the requisite equipment should all be considered before selecting the optimal therapy. Although treatment decisions are ultimately the result of an integrated analysis of a multiplicity of factors, for the sake of simplicity these factors and considerations in management of kidney calculi are reviewed separately (Fig. 48–1).
Table 48–1 Factors Affecting Management of Renal Stones
| STONE FACTORS | RENAL ANATOMIC FACTORS | CLINICAL (PATIENT) FACTORS |
|---|---|---|
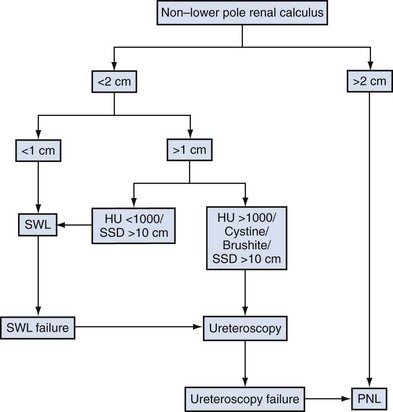
Figure 48–1 Treatment algorithm. HU, Hounsfield unit; PNL, percutaneous nephrolithotomy; SSD, skin-to-stone distance: SWL, shockwave lithotripsy.
(Adapted from Wen CC, Nakada SI. Treatment selection and outcomes: renal calculi. Urol Clin North Am 2007;34[3]:409–19.)
Preoperative Evaluation
The evaluation of patients with urolithiasis in the current era of minimally invasive therapies has not changed substantially from that of the previous era of open stone surgery. Although standard imaging of urinary tract calculi has historically relied on plain abdominal radiography and intravenous urography, recent evidence suggests that unenhanced helical computed tomography (CT) has gained widespread acceptance (Heidenreich et al, 2002). Nephrotomography, radionuclide studies, and retrograde or contrast-enhanced studies are only occasionally necessary to obtain more detailed anatomic and functional information.
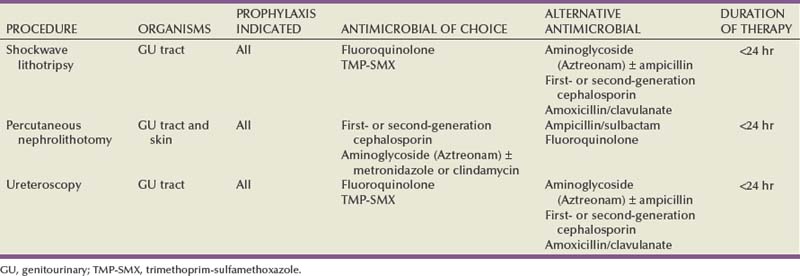
Bacteriologic evaluation of the urine is mandatory for all patients. The composition of any previous stone material passed or removed from the patient is extremely important. If previous stones have contained significant amounts of calcium oxalate monohydrate (whewellite) or brushite, fragmentation with SWL may be expected to be more difficult. If a stone of such composition is of a large size, the patient may achieve a better outcome with a percutaneous or ureteroscopic procedure rather than with SWL. Cystinuria may be revealed by previous stone analysis or by the characteristic cystine crystals on urinalysis. Any patient whose stone or stones have radiographic features suggestive of cystine (low radiodensity, ground-glass appearance, smooth edges, bilateral stones) should be screened for cystinuria before treatment because these stones are often not well fragmented by SWL. Many stones may harbor bacteria even though bacteriuria is only intermittently present. This is particularly true in the patient who has received antibiotics in the past. Mariappan and associates (2005) have reported that the best predictor of post-PNL urosepsis is stone culture or renal pelvic urine culture results rather than bladder urine culture results. The fragmentation of stones, despite sterile urine, may release preformed bacterial endotoxins and viable bacteria that place the patient at risk for septic complications (Scherz and Parsons, 1987; McAleer et al, 2002, 2003; Paterson et al, 2003). In a review of a large series of patients undergoing PNL, de la Rosette and coworkers (2008) also confirmed that a positive preoperative urine culture was a significant predictor of postoperative morbidity. Therefore, patients who have radiographic or clinical features suggestive of struvite or in whom infection is suspected should receive a regimen of broad-spectrum antibiotics before surgery to reduce the risk of sepsis. Parenteral antibiotics should be administered perioperatively in any patient in whom urinary infection is suspected.
Natural History
Calyceal Stones
Before the introduction of minimally invasive therapies, urologists were often reluctant to remove incidentally found, asymptomatic, or minimally symptomatic calyceal stones because of the high morbidity associated with open surgery. Although the traditional indications for the treatment of a renal calculus, including pain, infection, and obstruction, have not changed, the introduction of minimally invasive procedures has allowed patients with mild or even no symptoms to consider these less morbid treatments. However, the decision for intervention should be based on a thorough understanding of the natural history of calyceal stones.
Hubner and Porpaczy (1990) reviewed the natural history of calyceal stones followed for an average of 7.4 years. During the observation period, 45% of the stones increased in size, 68% of the patients experienced symptoms of infection, and 51% of the patients experienced pain. Inci and associates (2007) also found that for patients with asymptomatic lower pole calculi, one third of the stones progressed in size and 11% ultimately required surgical intervention. Thus, most calyceal stones, in the absence of intervention, are likely to increase in size and cause symptoms of pain or infection. Furthermore, as time progresses and stone size increases the likelihood of spontaneous stone passage becomes further reduced. Burgher and associates (2004) reported a series of 300 patients initially presenting with asymptomatic renal calculi who were observed for a mean of 3.26 years. Seventy-seven percent of patients experienced progression of calculi, with 26% requiring surgical intervention. Those patients who initially presented with calculi larger than 4 mm were more likely to fail observation than were patients with smaller solitary calculi. Keeley and colleagues (2001) reported the results of a randomized prospective trial of SWL versus observation for 200 patients with small asymptomatic calyceal calculi. Although the authors found little difference in the number of patients in each group requiring additional treatment, the interventions in the observation group were more invasive. Patients in the SWL group required no invasive treatment on follow-up and could be adequately managed with analgesia or antibiotics. However, there was no evidence of a difference in stone-specific symptoms, quality of life, or renal function tests between the two groups at study’s end. The authors concluded that SWL does not appear to improve the clinical outcome of patients with small, asymptomatic renal calculi. Considering the entirety of the literature to the present time, the necessity of treating patients with small (<5 mm), nonobstructive, asymptomatic stones in a prophylactic fashion remains undetermined. However, should asymptomatic stones not be treated, patients must be advised about the need for regular follow-up; a significant proportion of these calculi will eventually become symptomatic and require intervention. Treatment decisions in these situations should be based on the individual patient’s risk factors and the patient’s preference. In several groups of patients, including pediatric patients, patients with a solitary kidney, patients in high-risk professions (e.g., pilots), and women considering pregnancy, treatment of asymptomatic calyceal stones may be indicated. Importantly, if asymptomatic stones are to be managed expectantly, a metabolic evaluation should be performed, to direct a regimen that will prevent stone growth and thereby reduce the need for future surgery (Galvin and Pearle, 2006).
Historically, simple calyceal stones documented to be immobile and not causing obstruction have been thought to be unlikely causes of flank pain. However, it has been reported that the pain induced by nonobstructive calyceal stones is characterized by a dull, deep ache, different from the classic pain of renal colic (Coury et al, 1988). It has been reported that endoscopic treatment of calyceal calcifications is associated with improved pain relief for many patients (Taub et al, 2006). Therefore, a patient who is thought to be symptomatic from a calyceal stone should be treated.
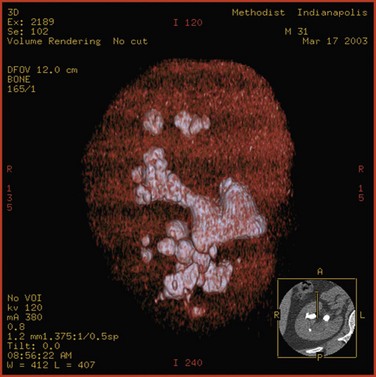
Staghorn Calculi
Staghorn calculi are those stones that fill the major part of the renal collecting system. Typically, they occupy the renal pelvis and branch into most of the calyces, mimicking the horns of a deer or stag (Fig. 48–2). Most staghorn stones are composed of struvite (Segura et al, 1994). Until the early 1970s some physicians believed that patients harboring staghorn calculi should not be treated (Segura, 1997). However, a better understanding of the natural history of staghorn stones has evolved. It is now generally accepted that, if left untreated, a staghorn calculus is associated with progressive deterioration of renal function. Additionally, morbidities associated with an untreated staghorn stone include pain, recurrent urinary tract infection, and sepsis events. Furthermore, patients with untreated staghorn calculi face an increased risk of death.
Thus, untreated struvite staghorn calculi eventually destroy the kidney and pose a significant risk to the patient’s life. The American Urological Association (AUA) Nephrolithiasis Clinical Guidelines, released in 2005, recommended that in otherwise healthy individuals, newly diagnosed struvite staghorn calculi should be treated surgically (Preminger et al, 2005). Moreover, struvite stones must be removed completely to minimize the risk of continued urea-splitting bacteriuria.
Stone Factors
Stone factors to be considered in the treatment of patients with kidney calculi include stone burden (size and number), stone composition, and stone location. Because a significant issue in renal stone location is the treatment of a lower pole stone and the influence of renal anatomy on the treatment results of such calculi, further discussion of this topic is included in the section on renal anatomic factors.
Treatment Decisions by Stone Burden
Stone burden (size and number) is perhaps the single most important factor in determining the appropriate treatment modality for a patient with renal calculi. However, cutoff points for stone size that would categorize those who should be treated by SWL and those who should undergo other treatment modalities have not been definitively determined. For now, kidney calculi are generally divided into the categories of nonstaghorn and staghorn stones. Most controversy related to surgical treatment focuses on the subject of nonstaghorn stones.
Nonstaghorn Calculi
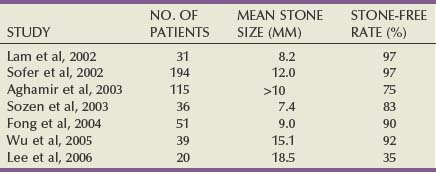
In examining the efficacy of SWL in the treatment of patients with renal calculi, passage of stone debris rather than fragmentation of the stone is the primary limiting factor (Renner et al, 1999). There is general agreement that stone free is the most rigorous definition of successful outcome of any stone removal procedure and that complete stone clearance should be the preferred goal of any intervention (Psihramis et al, 1992). However, because SWL outcome is dependent on spontaneous stone clearance, treatment results are often reported in terms of “success rates,” which may be defined as patients who are either stone free or who have asymptomatic, small, residual fragments. Various cutoff points between 2 and 5 mm are used in the literature to define the size of these fragments, making study comparisons difficult. In many cases, failure of SWL is not due to a failure of stone fragmentation but rather a failure to clear the resulting stone fragments. Failure to clear stone fragments is a concern, because it results in a higher re-treatment rate as well as a higher number of ancillary procedures. Clayman and associates (1989) suggested that in comparing the results of SWL and PNL or in comparing different lithotripters, the parameters of stone-free rate, re-treatment rate, and number of auxiliary procedures should be combined into an effectiveness quotient that may better express treatment results and allow one to compare different treatment modalities:
For example, Netto and associates (1991), in a study comparing PNL and SWL for patients with lower pole calculi, reported overall stone-free rates of 93.6% and 79.2% for PNL and SWL, respectively; these values were not significantly different. However, the effectiveness quotients of 93.7% and 55.9% for PNL and SWL did differ significantly because this calculation incorporated the 41% re-treatment rate for the SWL group.
The negative effect of an increasing stone burden (size and number) on the results of SWL has been described by a number of groups, dating from the initial reports of SWL to the present generation of lithotripters (Drach et al, 1986; Lingeman et al, 1986a; El-Assmy et al, 2006a; Tan et al, 2006). A now-axiomatic principle of SWL is that as stone burden increases, the stone-free rate declines and the need for ancillary procedures and re-treatment rises. Importantly, stone burden is not defined solely on the basis of the largest stone present in the kidney but also it takes into account the overall number of stones present. Furthermore, larger stone burdens are associated with a higher rate of residual stones, a point of particular concern in the treatment of patients with struvite calculi (Preminger et al, 2005).
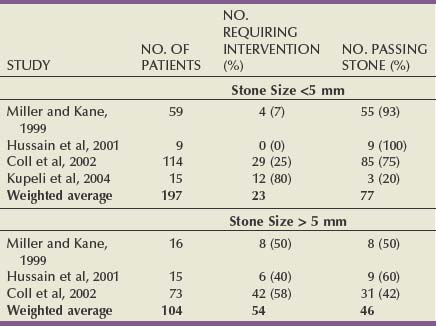
Figure 48–3 illustrates the effect of the size of solitary renal stones on the results of SWL. PNL, although more invasive and often associated with higher morbidity, achieves better stone-free rates than does SWL and is not affected by stone size (Lingeman et al, 1987a). Ureteroscopy, an alternative treatment for patients with renal calculi, is also negatively affected by increasing stone burden, although to a lesser degree than is SWL, because stone fragments are often removed or vaporized. Thus, as stone burden increases, PNL becomes more efficient than either SWL or ureteroscopy.
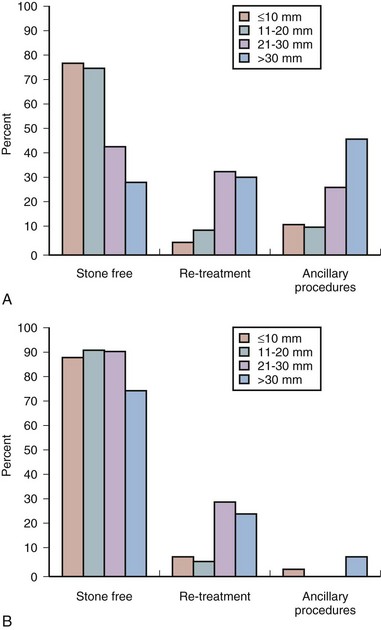
Figure 48–3 A, Solitary nonstaghorn calculi treated by shockwave lithotripsy, stratified by size. B, Solitary nonstaghorn calculi treated by percutaneous nephrolithotomy, stratified by size.
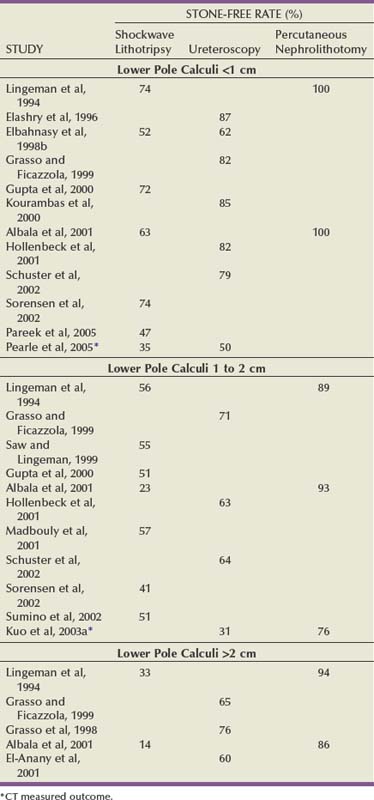
Importantly, 50% to 60% of all solitary renal calculi are less than 10 mm in diameter (Cass, 1995; Renner and Rassweiler, 1999; Logarakis et al, 2000). Treatment results of SWL for this substantial group of patients are generally satisfactory and independent of stone location or composition. Although better results can be achieved with PNL or ureteroscopy for patients with stones smaller than 10 mm, these procedures are more invasive, are associated with greater morbidity, and may be reserved for special circumstances (e.g., anatomic malformation causing obstruction, SWL failure).
Patients with calculi between 10 and 20 mm are often treated with SWL as first-line management. However, stone location and composition can meaningfully affect the results of SWL for patients with calculi in this size range and should be carefully considered. For example, SWL results for patients with 10- to 20-mm stones in the lower pole are inferior (55%) to SWL results for patients with stones in the upper and middle pole calyces (71.8% and 76.5%, respectively) (Saw and Lingeman, 1999). A prospective, randomized controlled trial compared SWL and PNL for patients with lower pole renal calculi; the stone-free rate for PNL was 95%, versus 37% for SWL (Albala et al, 2001). Stone composition merits consideration when evaluating treatment alternatives for patients with stones larger than 10 mm, as cystine calculi and brushite calculi both respond poorly to SWL treatment. This effect is particularly pronounced for stones larger than 15 to 20 mm. Therefore, patients with renal stones of 10 to 20 mm and factors predicting poor treatment outcomes with SWL should be advised about alternative therapeutic modalities. Both PNL and ureteroscopy are less affected by stone location and composition, and good results may be attained with these modalities for patients with 10- to 20-mm renal stones.
Patients with renal calculi greater than 2 cm who are treated with SWL monotherapy commonly experience poor treatment outcomes, a fact that was first recognized over two decades ago in an NIH Consensus Conference. Interestingly, the 2 cm threshold for SWL first noted in that conference document is still valid in the present day (Consensus Conference, 1988). Murray and coworkers (1995) reported that SWL monotherapy for renal calculi greater than 3 cm yielded an overall success rate of 27% at 3 months follow-up. The best stone-free rate (60%) was obtained for stones smaller than 500 mm2 that were located primarily within the renal pelvis; the stone-free rate for stones with surface areas larger than 1000 mm2 was a dismal 8%. Notably, steinstrasse occurred in 23% of patients. El-Assmy and colleagues (2006a) reported on patients with large-volume renal calculi treated with SWL monotherapy. Long-term follow-up demonstrated a stone-free rate of 59%; significant complications occurred in 13%, and unplanned secondary procedures were required in 18.4% of cases.
As an alternative to SWL for large-volume calculi, ureteroscopy emerged in the 1990s as a viable treatment option. Grasso and associates (1998) provided one of the earliest series of patients with large (>2 cm) upper urinary tract stones treated by ureteroscopy. One third of patients with renal stones required a second-look endoscopy; and in three patients with renal calculi, conversion to PNL was necessary. The overall success rate, defined as pulverization of the stone to dust or fragments smaller than 2 mm, after the second ureteroscopy procedure was 91%, which is comparable to PNL results. However, the 6-month follow-up data, which were available for 25 patients, demonstrated that only 60% of patients were stone free, whereas 24% had small lower pole debris and 16% had new stone growth. As surgical techniques and technology have evolved, ureteroscopy has been applied to patients with progressively larger stone burdens with acceptable results and morbidity (Mariani, 2007; Ricchiuti et al, 2007; Breda et al, 2008). In general, these treatment approaches have relied on a staged approach to achieve a successful outcome.
In summary, for patients harboring nonstaghorn stones smaller than 10 mm, SWL is usually the primary approach. For patients with stones between 10 and 20 mm, SWL can still be considered a first-line treatment unless factors of stone composition, location, or renal anatomy suggest that a more optimal outcome may be achieved with a more invasive treatment modality (PNL or ureteroscopy). Patients with stones larger than 20 mm should primarily be treated by PNL unless specific indications for ureteroscopy are present (e.g., bleeding diathesis, obesity).
Staghorn Calculi
Patients suffering from staghorn calculi remain a challenging problem for the practicing urologist. Most staghorn stones are composed of struvite, and factors that predispose to urinary tract infection and retained urine increase the likelihood of struvite stone formation (Gettman and Segura, 1999). However, other crystals, including cystine, calcium oxalate monohydrate, and uric acid, can assume a staghorn configuration.
The conservative treatment of patients with staghorn calculi exposes the patient to an increased risk of renal loss as well as a mortality rate of up to 30% (Blandy and Singh, 1976; Rous and Turner, 1977; Koga et al, 1991). Therefore the ideal management of patients with staghorn calculi is composed of three stages. First, complete surgical removal of the entire stone burden is essential. If all of the infected stone debris is not evacuated, urea-splitting bacteriuria may persist, which can ultimately lead to eventual stone regrowth. The urologist should select the procedure or combination of procedures most likely to render the patient free of stone material while minimizing the risk of morbidity and mortality. Second, any metabolic abnormalities must be identified and appropriately treated. It has been reported that metabolic abnormalities are not uncommon in patients with infected stones (Segura et al, 1981). However, others have found that stone recurrence after complete elimination of calculi is uncommon (Silverman and Stamey, 1983). Different definitions of the term infection stone, and in particular the inclusion of mixed struvite and calcium oxalate stones in studies, probably explain these contradictory reports. In a group of patients with infection stones, those with pure struvite stones were significantly less likely to have metabolic abnormalities than were patients who had stones composed of a mixture of struvite and calcium oxalate (Lingeman, 1995; Lingeman et al, 1995c). Finally, anatomic abnormalities that may contribute to stasis within the urinary tract should be addressed.
Classification of Staghorn Calculi
The traditional definition of staghorn calculus is a renal pelvic stone with extension into the renal calyces. Historically, staghorn calculi were classified as partial or complete. Partial staghorn calculi were defined as renal pelvic calculi extending into two calyceal groups, and complete staghorn stones were defined as renal pelvic calculi extending into all major calyceal groups, filling at least 80% of the collecting system. At the time this definition was popularized there was only one treatment option for patients with these calculi: open surgery. With the introduction of newer and less invasive treatment modalities that are highly sensitive to stone burden there is a greater need to classify staghorn calculi according to reproducible, quantitative criteria that allow clinicians to better evaluate treatment outcomes.
The morphologic classification into partial and complete staghorn calculi is inadequate, as demonstrated by Lam and colleagues (1992c), who reported considerable overlap in stone burdens of calculi grouped as partial or complete staghorn stones. When stone burden was assessed by stone surface area as measured on a kidney-ureter-bladder (KUB) radiographic image, for stones between 501 and 1500 mm2 an overlap between partial and complete staghorn calculi commonly occurs. To remedy this limitation several groups have proposed new classification schemes to better define staghorn calculi (Rocco et al, 1984; Griffith and Valiquette, 1987; Ackermann et al, 1989; Di Silverio et al, 1990). However, the cumbersome and subjective nature of these classification approaches has resulted in limited clinical use.
Therefore, at present, the most accurate method to estimate the volume of a staghorn calculus is CT with three-dimensional reconstruction. This technique permits highly accurate determination of stone volume as well as the three linear dimensions of renal calculi and correlates well with the actual volume of the stone, as measured by water displacement (Lam et al, 1992c). Thiruchelvam and coworkers (2005) reported that an added benefit of three-dimensional image reconstruction is in planning for subsequent percutaneous stone removal. However, three-dimensional CT reconstructions are costly, time consuming, and not widely available, thus limiting the utility of this technology. Nadler and associates (2004) have reported the use of coronal reconstructions of axial CT images to calculate the craniocaudal length of stones, which can facilitate more economical stone volume calculations.
Surgical Management of Staghorn Calculi
Historically, patients with staghorn calculi were subjected to open surgical stone removal procedures. Overall, the stone-free rate after open surgery for patients with struvite staghorn stones was reported to be about 85%, with a 30% stone recurrence rate (Griffith et al, 1978). With the rise of endourology, however, minimally invasive procedures proved competitive with open surgery. In a comparison of PNL and anatrophic nephrolithotomy, Lingeman and associates (1987a) demonstrated that the stone-free rates of the two interventions were similar. Additionally, the convalescence, hospital stay, and blood transfusions were less for PNL. More recently, Al-Kohlani (2005) performed a randomized controlled trial comparing PNL and open surgery. Similar to the previous work of Lingeman they found that the morbidity, hospital stay, and operating room time all favored PNL, and the patients treated with PNL all returned to work sooner.
For patients with staghorn calculi, SWL monotherapy has a limited role in the management of this type of stone burden. A number of groups have reported that SWL monotherapy for staghorn calculi is associated with poor stone-free rates. Furthermore, patients treated with SWL require a greater number of secondary procedures. Postoperative complications are more common and include morbidities such as steinstrasse, renal colic, sepsis, and perinephric hematoma. The success rate of PNL as initial therapy for staghorn renal calculi is independent of stone composition and superior to SWL for all types of stone material. When treating struvite stones, SWL monotherapy may be particularly problematic because residual, “clinically insignificant” fragments can prevent the sterilization of urine, increasing the risk of stone regrowth.
The use of multiple endourologic techniques for the treatment of patients with staghorn stones is referred to as combination therapy or “sandwich therapy.” The most frequently used multimodal regimen was described by Streem and colleagues (1997) and consisted of a primary percutaneous stone debulking followed by SWL of any inaccessible, residual infundibulocalyceal stone extensions or fragments. After SWL a secondary percutaneous procedure was performed. These various stages are usually separated by 1 or 2 days. Stone-free rates for combined therapy are similar to those obtained by PNL alone or by open surgery (Lam et al, 1992b). The management of patients with staghorn stones by a combined approach must be viewed as primarily percutaneous in nature, with SWL being used only as an adjunct to minimize the number of access points required. Improved PNL techniques, incorporating the increasing use of flexible nephroscopy and providing complete or nearly complete clearance of stone material at the time of the primary procedure, may have decreased or eliminated the need for additional SWL treatment (Preminger et al, 2005).
The 2005 AUA Nephrolithiasis Committee has published recommendations for the management of patients with staghorn calculi based on a meta-analysis of outcome data from published, peer-reviewed articles. According to the committee, all treatment options (SWL, PNL, combined PNL and SWL, open surgery) must be discussed with the patient (Preminger et al, 2005). As a guideline, however, PNL, followed by either SWL or repeated PNL, should be used for most patients with struvite staghorn calculi, with PNL being the initial element of the combination therapy. SWL and open surgery should not be used for most of these patients as a first-line treatment. PNL and SWL are equally effective in treating patients with small-volume staghorn stones when the renal anatomy is normal or nearly normal. Open surgery may be an option in unusual situations in which a staghorn stone is not expected to be removed by a reasonable number of PNL or SWL procedures. Nephrectomy is an option for the patient with a poorly functioning kidney harboring a staghorn stone. Although there are limited data for the treatment of pediatric patients with staghorn calculi, PNL is a safe and effective therapy. Pediatric patients often experience better stone-free rates than do adults with treatment by SWL. The stone-free rate with SWL monotherapy, reported by studies including only pediatric patients, is 78%; although more than one procedure is generally required to achieve this outcome; fortunately, complications are infrequent (Preminger et al, 2005). However, the developing kidney may be more susceptible to the bioeffects of SWL (Connors et al, 2006).
Treatment Decisions by Stone Composition
Dretler (1988) first introduced the concept that stone fragility, defined as the readiness with which a stone is fragmented by SWL, is variable among stones of different composition. Saw and Lingeman (1999) subsequently reported that, when adjusted for size, cystine and brushite calculi are the most resistant to SWL, followed by calcium oxalate monohydrate; following, in descending order of resistance to fragmentation, are struvite, calcium oxalate dihydrate, and uric acid stones (Pittomvils et al, 1994; Saw and Lingeman, 1999). Stone composition affects not just resistance to fragmentation but also the type of fragments produced. Cystine and calcium oxalate monohydrate, in addition to being difficult to fragment, tend to produce relatively large pieces that may be difficult to clear from the collecting system (Pittomvils et al, 1994; Rutchik and Resnick, 1998). In general, patients with such stones (i.e., brushite, cystine, calcium oxalate monohydrate) should be treated by SWL only when the stone burden is small (i.e., <1.5 cm). Those patients with larger stones should preferentially be treated with PNL or ureteroscopy. Interestingly, the outcome of intracorporeal lithotripsy is also affected by stone composition. Teichman and colleagues (1998c) reported that the holmium laser was the most effective lithotrite for fragmenting struvite stones and the least effective lithotrite for fragmenting calcium oxalate monohydrate stones. These results are consistent with the known thermal threshold for each stone composition.
Patients suffering from cystinuria present a unique challenge to the urologist. Assimos and associates (2002) reported that cystinuric patients have higher serum creatinine levels compared with a cohort of calcium oxalate stone formers. Importantly, they also reported that cystinuric patients are at greater risk for renal loss than are calcium oxalate stone formers and that open stone surgery for these patients is associated with higher serum creatinine concentration and potential renal loss. Furthermore, patients with cystinuria have been reported to be poorly compliant with medical therapy, increasing the likelihood of recurrent stone events (Pietrow et al, 2003a). Chow and Streem (1998) analyzed 31 cystinuric patients who underwent selected intervention for 61 stone events and reported that the probability of stone recurrence at 1 and 5 years was 27% and 73%, respectively. Achieving stone-free status prolonged the time to stone recurrence compared with patients left with residual fragments, a finding confirmed by Knoll and associates (1988). The high likelihood of repeated procedures underlines the need to select not just the least invasive treatment modality but also the most effective treatment modality. Treatment efficacy is of particular importance, as Barbey and associates (2000) reported that decreased renal function was more pronounced in cystinuric patients subjected to more stone removal procedures. Evan and associates (2006) have also reported that patients with cystinuria demonstrate anatomic findings, such as collecting tubule crystal plugging, as well as glomerular changes consistent with medical renal disease. These findings were more severe in subjects with extensive stone-forming histories, confirming the unique nature of this population.
Therefore, when stone removal is required, a minimally invasive approach is preferred. SWL, when it is used unselectively to treat patients with cystine stones, yields poor results. Hockley and colleagues (1989) found that stone-free rates when applying SWL to calculi less than 20 mm or 20 mm or greater were 70.5% and 41%, respectively, whereas the stone-free rates for those who underwent PNL were 100% and 92%, respectively. Kachel and associates (1991) suggested the following treatment algorithm for patients with cystine stones: SWL monotherapy for cystine renal calculi 15 mm or smaller and PNL for stones larger than 15 mm in diameter. Rudnick and colleagues (1999) reported success with retrograde ureterorenoscopic fragmentation in patients with 1.5- to 3.0-cm renal calculi. This approach is especially appealing because it has an inherently low morbidity. Both Ahmed and associates (2008) and Trinchieri and associates (2007) have confirmed that endourologic approaches to patients with cystine calculi yield superior results, with minimal morbidity.
Brushite calculi have a resistance to fragmentation that is surpassed only by that of cystine calculi (Dretler, 1988). Klee and associates (1991) described 30 patients with a total of 46 brushite stones. The overall success rate for patients treated by SWL monotherapy was 65% (with success defined as fragments < 4 mm), with a mean of 1.5 SWL sessions required per stone. However, only 11% of patients became stone free. PNL and ureteroscopy achieved 100% success rates and stone-free rates of 100% and 66%, respectively. Of 20 kidneys with residual fragments smaller than 4 mm, 12 had rapid regrowth to significant size within 3 to 12 months. Parks and associates (2004) found that SWL use was more frequent among brushite stone formers than among a similar cohort of calcium oxalate stone formers. Therefore, when brushite calculi are suspected or confirmed, a surgical treatment algorithm similar to that for cystine stones should be applied.
Although resistance to SWL is generally common for very hard stones, it also characterizes the rare and very soft matrix calculi that are composed of as much as 65% organic matter (compared with 2% to 3% organic matter in most noninfected urinary calculi). Matrix stones are radiolucent and are often associated with urea-splitting bacteriuria. SWL is not an effective treatment modality for patients with these stones. These situations are usually best treated with PNL (O’Connor et al, 1990). SWL is usually ineffective because of the stone’s gelatinous nature, and ureteroscopy may be compromised by the large volume of stone material present (Bani-Hani et al, 2005).
Another soft radiolucent stone is composed of indinavir, a protease inhibitor commonly used in the treatment of human immunodeficiency virus infection (Daudon et al, 1997). A significant number of patients who receive this drug develop symptoms or signs of indinavir nephrolithiasis (Saltel et al, 2000; Nadler et al, 2003). Reiter and coworkers (1999) reported an incidence of symptomatic stone episodes in 12.4% of 105 patients treated with indinavir. The mean time from the initiation of indinavir therapy until the acute stone episode was 21.5 weeks. Twelve of 16 stones were passed spontaneously. Kohan and colleagues (1999) described 13 symptomatic patients with indinavir stones; conservative therapy was successful in 11 patients, and 2 patients were treated by stent placement. Pure indinavir stones are not detectable with standard radiography or CT. However, some patients form indinavir stones that contain a calcium component, which may be radiographically visible (Sundaram and Saltzman, 1999). Hydration and analgesic therapy are recommended for the initial treatment of patients with indinavir stones. Indinavir therapy may need to be temporarily or permanently discontinued, in which case another protease inhibitor may be substituted. Invasive intervention may be necessary for patients with prolonged renal obstruction, signs of sepsis, or unremitting symptoms.
The ability to predict stone composition and, consequently, the number of shockwaves required for complete stone fragmentation would be of great benefit in selecting appropriate treatment of patients with stone disease. If a stone is of a type not amenable to treatment by SWL, other modes of treatment could be pursued. However, except for cystinuric patients and patients who have had previous stone analysis, accurate prediction of stone composition based on imaging and the patient’s history is difficult. The ability of plain radiography to differentiate subtypes of calcium oxalate stones and possible relationships to stone fragility was first suggested by Dretler (Dretler, 1988; Dretler and Polykoff, 1996). A number of investigators subsequently examined the utility of x-ray patterns to predict stone fragility. In general, smooth edged stones with a uniform and homogenous structure require more shockwaves to fragment than do reticulated, spiculated stones with an irregular margin or structure. Additionally, stones that were more dense than bone (using either the 12th rib or a transverse process as a reference point) responded more poorly to SWL.
Non–contrast-enhanced helical CT, which is presently the most commonly utilized method of evaluating patients with suspected renal colic, may be useful when trying to identify stone composition. Several in-vitro investigations of this technology have been reported. Mostafavi and associates (1998) first performed an in-vitro study that used the attenuation levels acquired by CT to accurately predict the chemical composition of pure urinary calculi. Similarly, Saw and associates (2000) found that, in an in-vitro investigation, CT was able to differentiate between stone groups (each containing at least 60% of one stone constituent) on the basis of absolute attenuation values.
Joseph and associates (2002) have provided an in-vivo corollary when they reported that SWL success rates were significantly lower for those calculi with attenuation values greater than 1000 Hounsfield units (HU) than for those calculi with attenuation values less than 1000 HU. Gupta and associates (2005) have confirmed that there is a linear relationship between Hounsfield unit measurement of a stone and the likelihood of that stone fragmenting in a single SWL session. Other investigators have further advanced knowledge in this area, performing prospective studies to evaluate the utility of Hounsfield unit measurement in predicting stone fragmentation. Wang and associates (2005) confirmed that stone density greater than 900 HU (along with stone burden greater than 700 mm3 and irregular stone shape) is a predictor of a poor SWL outcome. El-Nahas and coworkers (2007), too, found that a stone density greater than 1000 HU predicted failure.
Although CT attenuation values can distinguish some stone types in vivo, such as uric acid from calcium stones, the use of attenuation values alone results in considerable overlap; the range of values for calcium oxalate monohydrate and struvite stones does not allow these types to be confidently distinguished. Furthermore, it is not certain that the ease with which a stone is fragmented by SWL can be predicted by knowing only the major mineral composition of the stone. For example, cystine stones, which are considered difficult to break, have been shown in certain cases to break easily (Bhatta et al, 1989). Williams and associates (2003), too, reported that the variability in stone fragility to shockwaves is large, even within groups defined by mineral composition. It is likely that this variability in fragility could be due to variation in stone composition or structure, including variable amounts of secondary mineral in the stone, variation in the spatial arrangement of the secondary mineral within the stone, and variation in the layer structures of the primary and secondary minerals within a stone. Williams and associates (2002) also reported that displaying the data acquired by helical CT with use of bone windows can reveal remarkable internal structural detail of kidney calculi (Fig. 48–4 on the Expert Consult website![]() ). It is clear that CT provides a wealth of information about stone characteristics. However, additional work is needed to determine the utility of this powerful imaging tool in determining the susceptibility of a given stone to SWL.
). It is clear that CT provides a wealth of information about stone characteristics. However, additional work is needed to determine the utility of this powerful imaging tool in determining the susceptibility of a given stone to SWL.

Figure 48–4 Photographic and helical CT images show structural variability in stones of the same type. Note that although all stones depicted are calcium oxalate in type, some have a mottled structure and others have a lamellar structure.
Dual-source CT is a recent innovation in CT technology; it incorporates two unique x-ray sources, rather than a single x-ray source as in conventional CT. In addition to improved temporal resolution, dual-source CT scanners have the ability to operate the two x-ray sources simultaneously, which has the potential to differentiate materials on the basis of their unique energy-dependent profiles. Dual-source CT technology has been reported to reliably differentiate calcium stones from uric acid stones (Flohr et al, 2006) as well as calcium phosphate stones from calcium oxalate stones (Matlaga et al, 2008; Boll et al, 2009).
Renal Anatomic Factors
There are certain anatomic factors, either congenital or acquired, that can hinder stone clearance after SWL. Congenital anomalies manifest not uncommonly in the upper urinary tract, and almost all that affect the drainage of the kidney are associated with an increased incidence of calculous disease. Such abnormalities include ureteropelvic junction (UPJ) obstruction, horseshoe kidney, and other ectopic or fusion anomalies as well as calyceal diverticula. Hydronephrosis, too, is associated with a failure to clear stone fragments after SWL. Although not, strictly speaking, an anatomic abnormality, the dependent position of lower pole calyces may also affect stone clearance after SWL. In addition, stone-free rates after SWL for patients with hydronephrosis or obstruction are poor. Any patient with obstruction distal to the targeted stone should not undergo SWL treatment. If both obstruction and infection are present, SWL may result in life-threatening urosepsis. Furthermore, the patient is unlikely to clear stone fragments unless the concomitant obstruction is alleviated.
Ureteropelvic Junction Obstruction
UPJ obstruction in adults is commonly associated with urinary calculi. Furthermore, a stone at the UPJ can exacerbate the degree of preexisting obstruction and further compromise the renal unit (Rutchik and Resnick, 1998). The role of anatomic obstruction and associated urinary stasis in stone formation is not clearly established. Husmann and colleagues (1995) reviewed the records of patients with simultaneous UPJ obstruction and renal calculi and found that 71% of patients with nonstruvite stones had significant metabolic abnormalities. Matin and Streem (2000) have also reported that patients with UPJ obstruction are at increased risk for lithogenic factors associated with metabolic stone disease. Thus, in addition to anatomic obstruction, underlying metabolic abnormalities are commonly present in patients with UPJ obstruction.
Although patients with stones and concomitant UPJ obstruction have traditionally been treated by open pyeloplasty and stone extraction, PNL with concomitant endopyelotomy can achieve good results with less morbidity. Endopyelotomy, defined as incision of the UPJ obstruction intraluminally, has an overall success rate between 67% and 88% (Ramsay et al, 1984; Van Cangh et al, 1989, 1994; Motola et al, 1993; Kletscher et al, 1995; Nadler et al, 1996; Albani et al, 2004; Knudsen et al, 2004). Percutaneous endopyelotomy can be combined with PNL, which permits efficient stone removal as well as careful endoscopic inspection of the UPJ. Success rates for combined PNL and endopyelotomy are comparable to endopyelotomy alone; a large, long-term review of endopyelotomy from the Mayo Clinic found that the presence of a stone did not adversely affect treatment outcome (Dimarco et al, 2006). Although retrograde endopyelotomy is another technique for the treatment of UPJ obstruction, the antegrade approach is preferable when renal calculi are present because it simplifies the stone removal aspect of the procedure.
Laparoscopic pyeloplasty has emerged as a commonly applied treatment for patients requiring surgical repair of UPJ obstruction. Because stones are commonly present in such situations there has been interest in laparoscopic stone removal at the time of laparoscopic pyeloplasty. Ramakumar and colleagues (2002) initially reported the feasibility of laparoscopic pyeloplasty with concomitant pyelolithotomy. Ball and associates (2004) subsequently noted, however, that laparoscopic pyeloplasty with concomitant pyelolithotomy is most efficacious when it is applied to patients with limited stone burdens. As surgical instrumentation had advanced, Stein and associates (2008) have subsequently reported that increasing stone burdens can be efficiently treated, using laparoscopic, rather than endoscopic, instrumentation. Atug and associates (2005) have also reported the application of robotic technology, specifically robotic graspers, to accomplish pyelolithotomy at the time of a robotic pyeloplasty; this technique, too, permits the efficient removal of a stone burden.
Calyceal Diverticula
Calyceal diverticula are congenitally derived, nonsecretory, urothelium-lined eventrations of the renal collecting system that are filled with urine. A narrow neck communicating with the collecting system is typically present, which permits the diverticulum to fill passively with urine. Calyceal diverticula are uncommon, having been reported as incidental findings in 0.2% to 0.6% of individuals undergoing renal imaging (Middleton and Pfister, 1974; Timmons et al, 1975; Wulfsohn, 1980; Michel et al, 1985). Stones have been reported to form in 9.5% to 50% of these cavities and can cause pain and hematuria or harbor bacteria (Yow and Bunts, 1955; Williams et al, 1969; Middleton and Pfister, 1974). The role of metabolic factors versus urinary stasis in the pathogenesis of stone formation in calyceal diverticula is controversial. Burns and coworkers (1984) suggested that particle retention time, especially in the setting of a diverticulum, could be the cause of stone formation. However, several studies examining metabolic data have drawn conflicting conclusions. Although Hsu and Streem (1998) have reported metabolic abnormalities in 50% of 14 patients with calyceal diverticular calculi, Liatsikos and associates (2000) found a low incidence of metabolic abnormalities in 49 patients with calyceal diverticular stones. Matlaga and associates (2007) reported that it is likely a combination of both urinary stasis and metabolic factors that incite stone formation in these structures.
Historically, patients with calyceal diverticula were treated by open surgical nephrotomy with closure of the infundibulum, marsupialization and fulguration of the diverticular cavity, or partial nephrectomy. However, with the development of minimally invasive surgical therapies, including PNL, ureteroscopy, SWL, and laparoscopic surgery, open surgical treatments are now uncommon. The use of SWL for the treatment of patients with diverticular calculi is controversial. Most investigators agree that to prevent stone recurrence, eradication of the diverticulum should accompany stone removal, a goal that is not achieved with SWL (Cohen and Preminger, 1997). Furthermore, the stone-free rate for patients with calculi within calyceal diverticula who are treated by SWL averages only 21% (range, 4% to 58%) (Renner and Rassweiler, 1999). However, an average of 60% (range, 36% to 86%) of patients will become symptom free after SWL. The follow-up in series showing the highest symptom-free rates is relatively short (3 to 6 months); on extended follow-up, Jones and associates (1991b) demonstrated that some of the patients initially rendered symptom free will subsequently become symptomatic and require re-treatment. The highest success rate with SWL was reported by Streem and Yost (1992), who treated a series of 19 patients harboring calyceal diverticular calculi smaller than 1.5 cm and a functionally patent diverticular neck. An initial stone-free rate of 58% and a symptom-free rate of 86% were achieved, probably as a result of careful selection of patients; all of the diverticula filled with contrast material on preoperative intravenous pyelography, and the ostia were well visualized. In addition, the aggregate stone size was smaller than 1.5 cm. However, recurrent stone growth was demonstrated in 1 patient during the follow-up period, and 6 of 9 patients with infection presumed to be secondary to the diverticulum developed recurrent infections.
The percutaneous approach for the management of subjects with diverticular stones provides the patient with the best chance of becoming stone and symptom free (Al-Basam et al, 2000; Monga et al, 2000; Auge et al, 2002b). Furthermore, there is an excellent chance of ablating the diverticular cavity by this approach. A direct percutaneous approach to the calyceal diverticulum is preferable because it allows use of a rigid nephroscope for stone extraction, dilation and incision of the diverticular neck (if desired), and fulguration of the diverticular epithelium. Kim and associates (2005a) reported a novel, single-stage procedure that does not require ureteral catheter placement or entrance into the renal collecting system. Percutaneous access is directed onto the diverticular cavity harboring stone, two movable core J wires are coiled in the diverticular cavity, and the track is balloon dilated. Once the stone material is removed, the cavity is fulgurated.
Retrograde ureteroscopic management is a reasonable option for certain patients with diverticula in the upper and middle portions of the kidney when the stone burden is less than 2 cm and the diverticular neck is short and accessible (Grasso et al, 1995b). Ureteroscopy may avoid some of the morbidity of a percutaneous approach, while being more effective than SWL. With the ureteroscope the surgeon may use a laser fiber to incise the stenotic infundibulum, fragment the stone, and ablate the diverticular lining. Fuchs and David (1989) reported a stone-free rate of 73% and a symptom-free rate of 86% with this approach, and Batter and Dretler (1997) reported that of 18 patients in whom they could access the diverticulum, all were symptom free at a mean follow-up of 39 months. It should be noted, however, that many subjects in these reports required multiple procedures to achieve a successful outcome; the price for the minimally invasive nature of ureteroscopy may be an increased acceptance of a staged treatment approach. Auge and associates (2002b), however, reported a symptom-free rate of 35% and stone-free rate of 19% for this technique, leading them to conclude that a percutaneous approach should be considered the primary treatment modality for calyceal diverticula. Turna and associates (2007) have also reported the results of a long-term study, finding that PNL is associated with superior stone-free rates relative to ureteroscopy when applied to calyceal diverticula.
Laparoscopic management of patients with symptomatic calyceal diverticula has been described in several small series and case reports (Gluckman et al, 1993; Ruckle and Segura, 1994; Harewood et al, 1996; Hoznek et al, 1998; Curran et al, 1999). Miller and associates (2002) reported a series of five patients with symptomatic, stone-bearing calyceal diverticula who underwent retroperitoneoscopic surgical therapy. The authors concluded that the optimal candidates for this approach are those with a symptomatic calyceal diverticulum with thin overlying renal parenchyma, those with a large stone burden, and those with an anterior lesion inaccessible to or unsuccessfully managed by other approaches. Wyler and associates (2005) and Terai and associates (2004) have both reported the treatment of calyceal diverticula from a retroperitoneoscopic approach as well. It should be noted, however, that the laparoscopic approach is more invasive than PNL and therefore is usually limited to treatment of diverticula in anterior calyces, which are otherwise difficult to access with a percutaneous approach. Surgeons undertaking a laparoscopic approach should be facile with intraoperative laparoscopic ultrasound and laparoscopic suturing techniques.
Horseshoe Kidney and Renal Ectopia
Horseshoe kidney is the most common congenital renal anomaly, and patients with this condition are often affected by urolithiasis (Evans and Resnick, 1981). A horseshoe kidney is considered the result of a median fusion of metanephric tissue during early gestation. Subsequent entrapment of the fused lower pole isthmus by the inferior mesenteric artery results in an incomplete cephalad migration and an associated malrotation of the kidney (Hohenfellner et al, 1992). In many cases, the UPJ is anomalous because of a high ureteral insertion into an elongated renal pelvis. This can cause impaired urine drainage, a likely reason that up to two thirds of such patients are found on evaluation to have hydronephrosis, infection, or urolithiasis (Lampel et al, 1996). Although SWL can be used to treat patients with calyceal stones in a horseshoe kidney, the anomalous orientation of the kidney makes localization of the calculi more difficult, especially for stones in the anteromedial calyces. Placement of the patient in a prone position may facilitate localization of the stone (Jenkins and Gillenwater, 1988). Alternatively, a “blast path” technique, which uses the observation that sufficient acoustic pressure for stone fragmentation exists beyond F2 along the axis of the shockwave in many lithotripters, may be employed (Locke et al, 1990). Reported results of SWL treatment of patients with horseshoe kidney stones are variable; stone-free rates between 28% and 78% have been reported (Esuvaranathan et al, 1991; Vandeursen and Baert, 1992; Theiss et al, 1993; Kirkali et al, 1996; Lampel et al, 1996; Kupeli et al, 1999). Much of the wide range of results can be explained by variability in stone size and location among the different studies. Theiss and colleagues (1993) stratified stones by location and found, as expected, that the clearance rate for lower calyceal stones was inferior to that of middle and upper calyceal stones (a stone-free rate of 53.8% vs. 100%). Kirkali and associates (1996) studied a series of 18 patients with calculi larger than 10 mm and found a stone-free rate of 28%. Patients with renal calculi in horseshoe kidneys treated by SWL required a higher number of shockwaves per treatment and also experienced a higher re-treatment rate (30% vs. 10%) than did patients with similar stones in orthotopic renal units (Chaussy and Schmiedt, 1984; Drach et al, 1986; Lingeman et al, 1986a). Lampel and associates (1996) reported a recurrence rate of 86% for patients harboring persistent fragments after SWL compared with a 14% recurrence rate for patients who were stone free. Although stone-free rates, in aggregate, are low for patients with horseshoe kidneys, with proper selection these rates can be improved. Favorable factors for SWL of horseshoe kidneys include a stone burden smaller than 1.5 cm and nonobstructed collecting system drainage.
Several groups have applied ureteroscopic techniques to the treatment of calculi residing in horseshoe kidneys. Andreoni and associates (2000) treated three patients in this manner, with successful outcomes. Weizer and colleagues (2005), too, reported a similar experience. In both series, ureteral access sheaths, holmium laser energy, and nitinol grasping devices were used. Symons and colleagues (2008) also reported successful ureteroscopic treatment of stone burdens within horseshoe kidneys, although a majority of the patients required a staged approach with more than one treatment session. However, taken all together, these studies confirm that ureteroscopy may be an effective treatment modality for a stone burden less than 1.5 cm in horseshoe kidneys; flexible ureteroscopes, and devices such as the holmium laser, nitinol graspers, and ureteral access sheaths should be available when this approach is selected. Because spontaneous expulsion of stone fragments may be variable in these situations, meticulous attention should be given to the extraction of stone material.
For patients with larger calculi or for those who demonstrate evidence of poor urine drainage, PNL should be the primary approach. The results of PNL for patients with calculi in a horseshoe kidney are generally superior to those achieved with SWL, with an average stone-free rate of 84% (range, 75% to 100%) (Esuvaranathan et al, 1991; Jones et al, 1991a; Al-Otaibi and Hosking, 1999; Raj et al, 2003; Shokeir et al, 2004). The optimal point of entry for these kidneys is through a posterior calyx, which is typically more medial than in the normal kidney because of the altered renal axis and rotation associated with the midline fusion (Fig. 48–5). Except in the isthmus of the horseshoe kidney, where the vascular supply can vary, vessels tend to enter the kidney anteriorly. An upper pole collecting system puncture is often appealing because the entire kidney is usually subcostal. In most cases the lower pole calyces are anterior and inaccessible percutaneously. During PNL the flexible nephroscope often aids in accessing stones within anteromedial calyces, which may be difficult to reach with a rigid nephroscope.
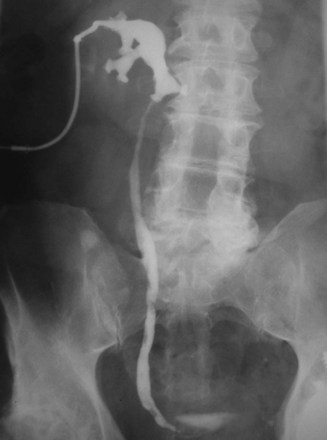
Figure 48–5 Antegrade nephrostogram performed after percutaneous nephrolithotomy of a horseshoe kidney via an upper pole access. Note the subcostal nature of the access and the unique calyceal orientation inherent in a horseshoe kidney.
An ectopic kidney can be found in a pelvic, iliac, abdominal, thoracic, or crossed position. The most common site of ectopia is the pelvis, and the incidence of pelvic kidney is estimated to be 1 in 2200 to 1 in 3000 patients. Although the ectopic location of the kidney can cause positioning problems during SWL treatment, calculi in these kidneys can often be approached with SWL. Although stone-free rates have been reported to be in the range of 66% to 71%, in some cases multiple treatment sessions may be necessary (Sheir et al, 2003; Tunc et al, 2004). Certain maneuvers may be required to adequately target the stone. If the bony pelvis shields the stone from the shockwave of the lithotripter, a prone position may be necessary. In general, stone clearance rates may be maximized by minimizing the stone burden treated.
Overall, stone-free rates for PNL of ectopic kidneys are better than those achieved with SWL. Likely, these superior success rates are due to the use of flexible nephroscopy and nitinol grasping devices. Percutaneous access for ectopic renal units is generally obtained with the patient in the supine position. Although Desai (2009) and others have reported using only ultrasound to guide the access portion of the procedure, such an imaging modality may increase the risk for bowel injury. Alternatively, laparoscopic assistance can be used to guide access and minimize the likelihood of bowel injury (Fig. 48–6). Holman and Toth (1998) and Maheshwari and associates (2004) have described laparoscopy-assisted PNL, reporting good results and no major complications. Matlaga and associates (2006) have also described unique cases that required transhepatic and transiliac access, maneuvers that highlight the need to tailor the intervention to the patient’s clinical situation. Alternatively, CT guidance can permit percutaneous access into the ectopic renal collecting system without injury to adjacent organs (Matlaga et al, 2003). Flexible ureteroscopy can also be an attractive alternative for this unusual problem if the stone burden is not large. Weizer and associates (2005) reported a series of patients with renal calculi in an ectopic kidney who were treated successfully with ureteroscopy.
Lower Pole Calculi
Although SWL is the preferred method for treatment of most patients with symptomatic upper tract calculi, there is considerable controversy concerning the management of patients with lower pole renal calculi (Tolley and Downey, 1999). The limitations of SWL for treatment of patients with lower pole stones were first noted in a meta-analytic study by Lingeman and associates (1994). These investigators reported that the stone-free results achieved with PNL were superior to those of SWL: the overall stone-free rate for SWL applied to lower pole calculi was 60% compared with 90% for PNL. Stratified according to stone size, patients with stones smaller than 10 mm had stone-free rates of 74% when they were treated by SWL compared with 100% by PNL. Among patients with stones of 10 to 20 mm, stone-free rates were 56% for the SWL group compared with 89% for the PNL group. A regression analysis demonstrated that increasing stone burden was associated with progressively less successful stone-free outcomes for patients treated with SWL, whereas for PNL-treated patients stone-free rates were not different across categories of stone size. This study has prompted a number of groups to assess the impact of lower calyceal anatomy (Table 48–2). Furthermore, since the mid 1980s, a significant change in renal stone distribution has been noted, with an increase in the percentage of SWL treatments for patients with renal calculi being performed for lower calyceal stones (2% in 1984 to 48% in 1991) (Lingeman et al, 1994). This change in stone distribution may be explained by the tendency of small, radiographically undetectable fragments to gravitate to more dependent calyces after SWL therapy and to act as a nidus for new stone growth (Carr et al, 1996).
The reasons for poor clearance of fragments from the lower pole after SWL are unclear. Intuitively, the gravity-dependent position of the lower pole calyx may impede the passage of stone fragments (Elbahnasy et al, 1998b). However, several investigators have proposed that in addition to the gravity-dependent positioning of the lower pole calyces, anatomic features may play an important role in the evacuation of stone fragments from this location. Anatomic factors were first suggested by Sampaio and Aragao (1992, 1994), who described the anatomy of the lower pole by use of polyester resin endocasts of the intrarenal collecting system obtained from adult cadavers (Fig. 48–7). The authors hypothesized that a lower pole with multiple infundibula might have poor drainage and consequently less possibility of eliminating stone fragments than would an inferior pole drained by a single infundibulum receiving fused calyces. They also hypothesized that a small diameter of the lower pole infundibulum might hinder passage of stone fragments. Finally, the authors examined the angle formed between the lower infundibulum and the renal pelvis and hypothesized that an angle greater than 90 degrees should facilitate drainage of fragments from the lower pole. Other investigators have analyzed the impact of lower pole anatomy on SWL stone clearance and have come to divergent conclusions (Albala et al, 2001). Although many have reported that certain anatomic factors alone, or in combination, may promote stone clearance, others have found no effect on lower pole anatomy in predicting treatment outcome.
Figure 48–7 Measurement scheme for lower pole anatomy. LPIL (lower pole infundibular length): measure A to B; LPIW (lower pole infundibular width): measure C to D; LPIP (lower pole infundibulopelvic) angle: measure QRS angle.
(Reprinted with permission from Albala DM, Assimos DG, Clayman RV, et al. Lower pole I: a prospective randomized trial of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy for lower pole nephrolithiasis—initial results. J Urol 2001;166:2072–80.)
One of the confounding issues in examining lower pole outcomes is the reproducibility of lower pole anatomic measurements. Pace and colleagues (2000) reported that there is wide variability in lower calyceal infundibular width between different intravenous pyelography phases. They found that the width was greatest on the compression film and smallest on the postvoid film, which prompted them to suggest the use of standardized timing when infundibular width is measured from an intravenous pyelogram. Although some of the disparity regarding the lower pole anatomic influences on stone-free status may be due to the techniques in which the parameters were measured, other sources of variability may be due to the type of lithotripter employed. In addition, stone composition, an important variable, is often neglected in these studies. However, it is likely that an obtuse versus an acute lower pole infundibulopelvic angle is important in fragment clearance after SWL. The most appropriate method for measuring lower pole infundibulopelvic angle and its relevant cutoff values as well as the significance of other lower pole anatomic features as predictive factors of stone clearance are yet to be determined. It may be that, in the future, computer modeling may be of benefit in these cases. Poulakis and associates (2003) developed an artificial neural network, incorporating both anatomic factors and dynamic measurements, and were able to accurately predict a successful outcome for patients undergoing SWL in over 90% of cases.
Despite the lower stone-free rates achieved with SWL therapy, a number of clinicians advocate this modality for the treatment of patients with lower pole calculi. In fact, a survey performed by Gerber (2003) found that 65% of urologists would use SWL for lower pole stones 1 to 2 cm in size and 2% would treat stones greater than 2 cm with SWL. Interestingly, however, success rates for SWL of lower pole calculi are generally poor. In trials comparing SWL to PNL, such as those performed by Elbahnasy and associates (1998a) and Cass (1996), the stone-free rate for PNL is superior. Similarly, in series just addressing patients treated with SWL, stone-free rates for lower pole calculi greater than 2 cm are less than 50% (Obek et al, 2001; Chen and Streem, 1996).
The poor stone clearance rates for patients with lower pole calculi treated by SWL have prompted investigators to examine techniques to facilitate fragment passage. McCullough (1989) anecdotally reported that postural drainage may assist in the elimination of retained fragments from dependent calyces. Brownlee and associates (1990) subsequently treated patients with residual lower pole fragments with controlled inversion therapy, using intravenous hydration, inversion, and percussion. D’a Honey and associates (2000) reported a pilot study to determine whether mechanical percussion with inversion therapy and furosemide-induced diuresis can move stone fragments out of the lower pole of the kidney. At a mean time of 63 days after SWL, this group reported an 83% stone passage rate. In a subsequent study, Pace and associates (2001) compared the effectiveness of mechanical percussion, inversion, and furosemide-induced diuresis with observation for elimination of lower calyceal fragments after SWL. They reported that 40% of patients with residual lower pole fragments treated with this regimen became stone free compared with 3% in the observation group; the observation group was then treated with this regimen as part of a crossover design, and 43% were rendered stone free. Other authors have reported lower pole irrigation techniques as adjuncts to SWL (Nicely et al, 1992; Graham and Nelson, 1994). More recently, pharmacotherapy with potassium citrate and thiazide diuretics has been described (Soygur et al, 2002; Arrabal-Martin et al, 2006). However, at this point in time none of these techniques has gained widespread acceptance.
As previously described, retrospective studies have indicated the results of SWL of lower pole calculi are inferior to those of PNL and depend on stone size. Albala and associates (2001) performed a multicenter, randomized, prospective study (Lower Pole I) that compared the treatment outcomes for patients with lower pole calculi after either PNL or SWL. The stone-free rate at 3 months after treatment, as measured by nephrotomograms, was 95% for those undergoing PNL and 37% for those undergoing SWL. Of note, stone clearance from the lower pole after SWL was especially poor as stone size increased above 10 mm. For patients with stones smaller than 10 mm, 20 of 20 (100%) patients treated with PNL were stone free whereas 12 of 19 (63%) treated with SWL were stone free. For those patients with calculi of 11 to 20 mm, 26 of 28 (93%) treated with PNL were stone free, whereas 6 of 26 (23%) treated with SWL were stone free. Finally, for patients with calculi of 21 to 30 mm, 6 of 7 (86%) treated with PNL were stone free whereas 1 of 7 (14%) treated with SWL was stone free. The main advantage of SWL was the lower associated morbidity.
In the decade since Grasso and Ficazzola (1999) first reported on a series of patients with lower pole calculi who underwent ureteroscopic treatment, the field of endourology has witnessed a number of advancements in instrumentation, technique, and technology. Consequently, ureteroscopy is now routinely applied to stones in the lower pole calyces, and a number of groups have reported success with this approach. Hollenbeck and associates (2001) reviewed 61 patients who underwent 68 ureteroscopic procedures for the treatment of lower pole calculi. Laser lithotripsy was performed in situ, without displacement of the stone from the lower pole. After a single procedure, 62% were stone free, and after a second procedure, 69% were stone free. Stratified by size, 71% of patients with stones smaller than 10 mm and 75% of patients with calculi of 10 to 20 mm were stone free.
There are certain maneuvers that may facilitate ureteroscopy for lower pole stones. Any device passed through the working channel of a flexible ureteroscope will have an adverse effect on the scope’s deflectability, potentially hindering access to the lower pole. However, nitinol baskets and flexible grasping devices have less of a deleterious effect than do laser fibers and may be used to displace a lower pole stone to a mid or upper calyceal location. Kourambas and associates (2000) found that the stone-free rate of patients who were treated with stone displacement before fragmentation was 90% compared with a stone-free rate of 83% for those patients who underwent in-situ fragmentation. Schuster and associates (2002) similarly reported a 77% stone-free rate for patients with lower pole calculi smaller than 1 cm treated in situ versus an 89% stone-free rate for those treated with displacement first. For patients with calculi larger than 1 cm, 100% of those undergoing displacement first were treated successfully compared with 29% of those treated in situ. The ureteral access sheath also may improve stone-free rates for this clinical scenario. Portis and associates (2006) performed a prospective study of patients with intrarenal calculi between 5 and 15 mm and found that with an access sheath and stone displacement a stone-free outcome, by strict CT criteria, was achieved in 59%.
Recognizing the technologic advances in ureteroscopy, the Lower Pole Stone Study Group compared ureteroscopy and PNL for patients with 10- to 25-mm lower pole stones (Kuo et al, 2003a). The primary outcome was a stone-free state, as measured by CT. The results of the study favored PNL, with a 71% stone-free rate, compared with a 37% rate for ureteroscopy. Although the length of hospitalization was shorter for the ureteroscopy cohort, interestingly the convalescence was not different, possibly owing to the morbidity of the ureteral stent among those undergoing ureteroscopy. The definition of stone free was strict and based on CT; if the definition is broadened to include fragments smaller than 4 mm, the stone-free rate improved to 80% for the ureteroscopy cohort and to 100% for the PNL cohort (Kuo et al, 2003a). Thus, ureteroscopy for patients with a large renal stone burden is probably inferior to PNL and should be used only for those patients who are poor candidates for PNL. The Lower Pole Stone Study Group also performed a prospective, randomized study of SWL and ureteroscopy for lower pole stones smaller than 10 mm. Although there was a 15% difference in stone-free rates between SWL (35%) and ureteroscopy (50%), the difference was not statistically significant. The recovery time, however, was shorter for SWL, and more SWL patients would prefer to be re-treated by that modality (Pearle et al, 2005).
The optimal approach for management of patients with lower pole stones continues to evolve (Fig. 48–8). SWL is a consideration for individuals with lower pole stones of 1 cm or less in aggregate size, because there is a reasonable chance of achieving a stone-free state with minimal attendant morbidity. Patients with lower pole stones of 2 cm or more are still best served with PNL because this offers them the best chance of being rendered stone free with one procedure. Much of the controversy regarding treatment of lower pole stones is limited to stones of 10 to 20 mm in diameter. PNL, ureteroscopy, and SWL are all acceptable options. Stone composition and lower pole anatomy should be considered in recommending a treatment modality for these patients. Patients with an acute lower pole infundibulopelvic angle (with or without other unfavorable anatomic features), patients whose SWL treatment has failed, and patients known to have stones resistant to SWL should be treated with PNL or ureteroscopy. In the future it may be possible to predict which patients among this group will have good results with the different treatment modalities.
Clinical Factors
The treatment of urinary tract calculi must be viewed in the context of the patient as a whole. Any coexisting clinical factors that may affect the safety and the efficacy of the selected treatment must be considered.
Urinary Tract Infection
Urinary tract infection, in the presence of renal calculi, can be difficult to eradicate unless the offending stones are completely removed. For these patients, then, PNL or ureteroscopy, both of which permit the complete removal of stone fragments, may be preferable to SWL. The reported incidence of sepsis after SWL is less than 1%, although a staghorn calculus increases this rate substantially to 2.7% to 56% (Lam et al, 1992a; Meretyk et al, 1997). The risk of sepsis increases if the urine culture demonstrates bacterial growth before SWL (Zink, 1988). Furthermore, the risk of sepsis is also greater in the presence of obstruction (Meretyk et al, 1992). Therefore, SWL should be performed only if the urine is sterile and there is no distal obstruction. The American Urological Association guideline for antibiotic therapy (Wolf et al, 2008) has provided specific recommendations for endourologic procedures (Table 48–3).
Morbid Obesity
Morbid obesity, which may be defined as more than 100 pounds overweight, greater than 200% of ideal body weight, or a body mass index (BMI) greater than 40, poses a number of physiologic and technical challenges to the successful treatment of kidney stones (Giblin et al, 1995; Freedman et al, 2002). The respiratory and cardiovascular morbidities inherent in a morbidly obese state can significantly increase the perioperative risk associated with even a minimally invasive procedure. Morbid obesity may make SWL impractical or technically impossible because of weight limitations on the lithotripter table or gantry, of the inability to target the stone radiographically, or the distance of the stone to the skin exceeds the maximum allowable focal distance of the lithotripter. When the increased distance from the skin surface to the stone renders positioning of the stone at the focus of the shockwave impossible, a blast path technique that relies on high pressures produced at a point located coaxially beyond F2 may be required (Whelan et al, 1988; Locke et al, 1990). Although the successful treatment of obese patients with SWL has been reported, high-energy settings of the lithotripter may be required. Should the urologist treating a morbidly obese patient have a choice among different SWL machines, the machine with the greatest focal length and highest peak pressure should be selected.
Despite these potential maneuvers, the morbidly obese may still achieve a suboptimal outcome after SWL. Several groups have acknowledged that BMI is a significant negative predictor of a stone free outcome after SWL (Ackermann et al, 1994; Portis et al, 2003). Because many patients undergo preoperative CT, the concept of a skin-to-stone distance (SSD), quantified on CT, has been popularized. More recently, this concept has been evaluated by the SSD metric, defined on preoperative axial imaging. Perks and associates (2008) reported that success rates were 79% versus 57% for SSD less than or greater than 9 cm, respectively. Pareek and associates (2005) similarly found that only 20% of stone-free patients had an SSD greater than 10 cm, whereas 85% of patients with residual stone patients had an SSD of 10 cm or greater; the SSD was a stronger predictor of treatment failure than the BMI.
Although obesity may have an adverse effect on SWL outcome, endoscopic interventions may be less sensitive to this parameter. In many cases PNL is the only effective treatment option for the morbidly obese patient with a complex renal calculus. The judicious use of an extralong working sheath, along with extralong rigid nephroscopes, flexible nephroscopes, and longer instruments, can overcome the increased SSD in these patients. Pearle and associates (1998) described 57 obese patients (BMI > 30) who underwent PNL. The overall stone-free rate (88.3%) and the complication and transfusion rates (12.8% and 5%, respectively) were comparable to the results of PNL in an unselected population of patients. Koo and associates (2004) similarly reported that the outcome of PNL is independent of the patient’s BMI.
Significant alterations in technique are not required when a ureteroscopic approach is selected for the morbidly obese patient with a renal calculus. For morbidly obese patients who do not harbor an excessively large stone burden the ureteroscopic approach may be preferable (Dash et al, 2002; Natalin et al, 2009). In fact, when compared with subjects of normal weight, ureteroscopy of morbidly obese confers no additional morbidity (Preminger et al, 2007).
Spinal Deformity or Limb Contractures
Patients with spinal deformity or limb contractures may also be difficult to position within the lithotripter. Alternative treatment modalities, including PNL and ureteroscopy with flexible instruments, may be preferable in these patients (Rubenstein et al, 2004). Ureteral access sheaths may facilitate repeated re-entry of the renal collecting systems in these situations as well. Owing to the unusual anatomy that is often present a preoperative CT scan can help in planning percutaneous access. In certain cases CT guidance may be necessary to safely obtain percutaneous access into the renal collecting system (Matlaga et al, 2003).
Uncorrected Coagulopathy
Although SWL or PNL treatment of patients with uncorrected coagulopathy can result in life-threatening hemorrhage, such patients can be safely treated once the bleeding diathesis has been corrected (Streem and Yost, 1990). However, if the patient’s coagulopathy is the result of a pharmacologic therapy that cannot be safely discontinued, ureteroscopy with holmium:yttrium-aluminum-garnet (holmium:YAG) laser lithotripsy is the preferable approach. Since Grasso and Chalik (1998) first reported that this was a safe and effective treatment method for patients with uncorrected coagulopathies, several other investigators have published similar findings. Watterson and associates (2002) as well as Turna and associates (2008) have reported safely managing patients with this approach.
Other Groups
The management of renal calculi in children is discussed in Chapter 135. However, when children as well as the elderly, the hypertensive, and those with impaired renal function are treated with SWL any adverse effects of the shockwaves may be enhanced. Care should be taken to limit the number and energy of shockwaves applied in such circumstances (Janetschek et al, 1997; Evan et al, 1998; Lifshitz et al, 1998).
Assessment and Fate of Residual Fragments
In the era of open stone surgery residual fragments of any size suggested a failed procedure. However, the increasing popularity of SWL as the treatment of choice for most patients with upper tract urinary calculi has changed the definition of a successful outcome and introduced the concept of clinically insignificant residual fragments (Newman et al, 1988). Newer definitions of treatment success were required after the observation that although many patients had successful stone fragmentation with SWL not all patients completely and spontaneously passed all of the stone fragments. Furthermore, the clearance of stone fragments produced by shockwaves is not immediate; as many as 85% of patients have radiologic evidence of residual fragments in the kidney several days after SWL (Drach et al, 1986). Although most fragments will pass spontaneously during the first 3 months after SWL, continued clearance of fragments can occur for more than 24 months after treatment (Chaussy and Schmiedt, 1984; Graff et al, 1988; Kohrmann et al, 1993). Therefore, since the introduction of SWL, treatment outcomes for patients with renal calculi have been reported by two different terms: stone-free rate and success rate. The stone-free rate is self-explanatory, but the success rate includes patients who are stone free as well as those with clinically insignificant residual fragments. These different methods of reporting treatment results, the lack of consensus regarding the definition of clinically insignificant residual fragments, and the various modalities used for assessing postprocedural stone-free status (KUB radiography, nephrotomography, ultrasonography, CT) make the comparison of endourologic stone removal procedures with SWL difficult.
Clinically insignificant residual fragments were initially defined as residual fragments 4 mm or smaller in diameter associated with sterile urine in an otherwise asymptomatic patient (Newman et al, 1988). However, small residual stones often become clinically symptomatic (Streem et al, 1996; Zanetti et al, 1997; Candau et al, 2000). Streem and colleagues (1996) first reported on the prospective, long-term follow-up study of post-SWL residual fragments; in a series of 160 patients with small, residual, asymptomatic calcium oxalate or calcium phosphate stones (≤4 mm), 43% had suffered a significant symptomatic episode at a mean follow-up of 23 months.
Residual fragments not only may become symptomatic but also can act as a nidus for new stone growth. Complete stone removal will decrease the risk of stone recurrence and regrowth (Singh et al, 1975; Patterson et al, 1987; Newman et al, 1988). Stone recurrence rates of 6% to 15% have been reported for patients who were rendered stone free after SWL, compared with rates of 17% to 80% when residual fragments were present (Graff et al, 1988; Newman et al, 1988; Nijman et al, 1989; Beck and Riehle, 1991; Fuchs et al, 1991; Zanetti et al, 1991; Nakamoto et al, 1993). In the long-term prospective study of Streem and associates (1997), residual fragments (<4 mm) after SWL were found to pass spontaneously in 24% of patients, although 18% of patients experienced stone growth. Similarly, a retrospective study of 83 patients with residual fragments (<4 mm) after SWL found complete stone clearance occurred in 33% of subjects but 37% of subjects developed increased stone burden (Candau et al, 2000). In this study the size of the residual fragment influenced the likelihood of its becoming symptomatic.
Many investigators have noted that, after SWL, residual fragments are commonly localized to lower pole calyces no matter where the stone was treated in the kidney (Drach et al, 1986; Graff et al, 1988; Liedle et al, 1988; Zanetti et al, 1991; Kohrmann et al, 1993). As well, the incidence of stone recurrence is greater in the lower pole calyces after SWL (Zanetti et al, 1991; Kohrmann et al, 1993; Carr et al, 1996) as opposed to PNL. Furthermore, at 1-year follow-up there is a significantly greater rate of new stone formation in those treated with SWL and the recurrent stones are more likely to be in the lower calyces. A plausible explanation for these results is that fine debris, undetectable by KUB radiography, persists after SWL and, because of gravity, tends to settle in the most dependent calyces, serving as a nidus for new stone formation.
In patients with infection-related calculi, the consequence of residual fragments is particularly harmful. Residual fragments may harbor the offending bacteria and thus perpetuate postoperative bacteriuria and persistent infection. Furthermore, stone regrowth has been reported in up to 75% of such patients after SWL, compared with 10% of patients who experienced complete stone removal (Beck and Riehle, 1991; Zanetti et al, 1991).
For patients with metabolic stone disease (e.g., those with cystinuria), complete stone removal does not prevent stone recurrence but it does prolong treatment intervals (Chow and Streem, 1998). Thus, residual stones, including small stones, may not have an immediate clinical relevance but are likely to affect the patient’s well-being in the long term. In these situations it is important to select a treatment approach that is most likely to render the patient stone free.
The sensitivity of the method used to detect residual stones after a stone removal procedure has important effects on the reported incidence and size of residual fragments. In early studies investigating stone clearance after SWL, plain radiography was commonly used to determine stone-free status. A plain radiograph can detect an opaque stone fragment as small as 2 mm (Thornbury and Parker, 1982). However, Denstedt and coworkers (1991) reported that for patients with large renal calculi treated by a combination of PNL and SWL, plain radiography overestimated stone-free rates by 35% and 17%, respectively, compared with flexible nephroscopy. Several groups have reported that nephrotomography is superior to plain radiography in evaluating stone-free status; additional residual stone fragments are detected in 12% to 47% of patients (Hjollund Madsen, 1972; Schwartz et al, 1984; Goldwasser et al, 1989). Ultrasonography is inferior to plain radiography and is particularly inadequate in detecting ureteral stones, although this modality is very effective in diagnosing hydronephrosis (Yilmaz et al, 1998; Older and Jenkins, 2000). In fact, ultrasonography should be considered after all ureteroscopic procedures because silent obstruction has been reported to occur in certain, albeit rare, instances (Weizer et al, 2002). A prospective study comparing the relative efficacy of abdominal radiography and renal ultrasonography with excretory urography for the evaluation of asymptomatic patients 1 month after SWL treatment demonstrated that the combination of ultrasonography and abdominal radiography was as good as or better than intravenous urography in identifying residual stone fragments and renal abnormalities, including hydronephrosis (Coughlin et al, 1989). The authors suggested that routine radiologic evaluation of asymptomatic patients after SWL could be limited to abdominal radiography and ultrasonography.
Although flexible nephroscopy may be considered the “gold standard” for assessment of residual stones after PNL, its routine use has been challenged by studies showing the high sensitivity of CT in detecting residual stones after PNL. Pearle and coworkers (1999) noted that CT had 100% sensitivity for detecting residual stones after PNL in 36 patients evaluated with both CT and flexible nephroscopy. Selective use of flexible nephroscopy based on positive CT findings would have avoided an unnecessary procedure in 20% of patients. In a retrospective study of 121 patients who underwent CT after PNL (including 59% stone-free patients and 16% patients with fragments of 1 to 3 mm), Waldmann and associates (1999) reported that routine nephroscopy would not have been required in 75% of cases. Given its wide availability and high sensitivity, non–contrast-enhanced CT has become the primary method for evaluation of patients after PNL.
Adjunctive Medical Treatment
Any patient who requires surgical stone removal should be offered a metabolic evaluation and stone analysis. Appropriate medical therapy can decrease the risk of stone recurrence and stone regrowth (Lifshitz et al, 1999a). Mardis and associates (2004) emphasized the importance of the administration of validated medical therapies, guided by metabolic evaluation, in reducing stone recurrence rates after treatment. Others (Lee et al, 1999; Cicerello et al, 1994) have reported similar experiences, describing reduced stone recurrence rates after a metabolic evaluation and medical management. Although recurrence rates are lower in a stone-free state, medical management is also effective in the presence of residual fragments (Fine et al, 1995). Kang and associates (2007) and Raman and associates (2009) have both reported that, after PNL, appropriate medical therapy will reduce the risk of stone recurrence or progression of any residual fragments. Any postprocedural medical management should not supercede the choice of a treatment approach that gives the patient the best chance to achieve a stone-free status with minimal morbidity. However, it is reasonable to begin a metabolic work-up, such as 24-hour urinalysis, while the patient is waiting to undergo a scheduled procedure. In this way the time from diagnosis of a stone to the treatment of a metabolic abnormality may be minimized and medical therapy may be begun expeditiously.
Key Points: Renal Calculi
Ureteral Calculi
The goal of the surgical treatment of patients suffering from ureteral calculi is to achieve complete stone clearance with minimal attendant morbidity. Improvements in surgical technology, such as SWL, rigid and flexible ureteroscopes, the holmium:YAG laser, and basket devices, have greatly augmented the urologist’s ability to efficiently treat such patients, regardless of the size or location of the ureteral calculus. Although the treatment options available to the urologist are greater now than they have ever been, most patients with ureteral calculi do not require intervention. Ureteral calculi 4 mm or smaller will usually pass spontaneously, although in some cases with discomfort and expense to the patient. Ureteral calculi of any size may be associated with renal obstruction, and care must be taken to prevent irreversible damage to the kidney, whether the patient selects expectant or active treatment.
Perhaps the greatest dilemma facing the urologist today is “to blast or not to blast” (i.e., to choose between the two most frequently used modalities in ureteral stone treatment—SWL and ureteroscopy). PNL is a less commonly used treatment option and is usually reserved for a limited group of patients with large, proximal ureteral stones. Open ureterolithotomy is rarely indicated, although it may be an option as a salvage procedure. Laparoscopic ureterolithotomy has been described both as a salvage procedure in lieu of an open ureterolithotomy as well as a first-line therapy. The ultimate role of this procedure in the urologic armamentarium is yet to be determined, although it is effective for a select group of patients.
The factors that the urologist must consider when recommending treatment to patients with ureteral calculi may be grouped into three broad categories: stone-related factors (location, size, composition, duration of presence, and degree of obstruction), clinical factors (the patient’s tolerance of symptomatic events, the patient’s expectations regarding treatment outcome, associated urinary tract infection, solitary kidney, and abnormal ureteral anatomy), and technical factors (equipment available for treatment, costs) (Table 48–4). These factors may be thought of as treatment modifiers; the presence or absence of one or more of these factors may shift the balance toward certain treatment modalities or away from other treatment modalities.
Table 48–4 Factors Affecting Management of Ureteral Stones
| STONE FACTORS | CLINICAL FACTORS | TECHNICAL FACTORS |
|---|---|---|
Natural History
The indications for intervention in the management of patients with ureteral calculi have clearly been affected by the increased efficiency and reduced morbidity of minimally invasive treatment modalities. Although the traditional indications for intervention (intolerable or intractable symptoms, infection, obstruction, and a stone that is unlikely to pass spontaneously) have not changed, the array of technologies currently available allows almost any symptomatic patient to be considered a candidate for stone removal. An early cost analysis in the era of minimally invasive surgery (Lingeman et al, 1986b) reported that when a patient requires hospitalization it is less costly to remove the patient’s stone with either SWL or ureteroscopy than to attempt to control the patient’s symptoms with pharmacotherapy only. However, many patients will pass their stone spontaneously, making a surgical intervention unnecessary. A thorough knowledge, then, of the natural history of ureteral stones permits a well-informed judgment of when conservative measures (e.g., observation), rather than intervention, are indicated. Furthermore, such data help the patient consider the spectrum of options and decide whether to try to endure further symptoms or to elect immediate stone removal.
In the absence of external ureteral compression or internal narrowing, the width of the stone is the most significant measurement affecting the likelihood of stone passage (Ueno et al, 1977). The 5-mm reference size has emerged as a breakpoint; when evaluating for passage, ureteral stones are generally categorized as greater than or less than 5 mm in greatest diameter (Table 48–5). There have been four studies, including 197 patients, that have evaluated the likelihood of a 5-mm stone passing spontaneously. A meta-analysis of the studies’ data found that the likelihood of a stone less than 5 mm passing spontaneously was 77%. Three studies, including 104 patients, have evaluated the likelihood of stones greater than 5 mm passing spontaneously. A similarly performed meta-analysis found that the likelihood of stones greater than 5 mm passing spontaneously was 46%. Morse and Resnick (1991) reported that the rate of spontaneous passage is greater for distal ureteral stones (71%) than for proximal ureteral stones (22%). Hubner and associates (1993) also reported that the likelihood of spontaneous stone passage was directly related to stone location at the time of presentation, because the rate of spontaneous passage for stones in the distal ureter was 45%, compared with 12% for the proximal ureter. These rates have been affirmed by a more recent review of CT of ureteral calculi (Coll et al, 2002). For patients with stones of 5 mm or less, conservative management should be considered, whereas the chance of spontaneous passage for larger stones and more proximal stones diminishes considerably and thus intervention should be more readily contemplated.
Miller and Kane (1999) analyzed the interval from presentation to passage to develop a time course for counseling patients. They found that the interval to stone passage was highly variable and dependent on both stone size and location: for 95% of stones to pass, 31 days were required for stones 2 mm or less, and about 40 days were required for stones 2 to 6 mm. Furthermore, only 4.8% of patients with stones smaller than 2 mm required intervention compared with 50% of patients with stones 4 to 6 mm. Cummings and coworkers (2000) trained an artificial neural network to predict outcome in patients with ureteral stones with 76% accuracy. The duration of symptoms before initial presentation was the most influential factor, followed by degree of hydronephrosis. The importance of symptom duration was reported by several other authors, who concluded that if significant progress has not occurred after 1 month of observation, intervention is usually required (Ibrahim et al, 1991; Hubner et al, 1993; Singal and Denstedt, 1997).
A number of investigators have reported on the use of pharmacologic means to promote ureteral stone passage, a practice that has been termed medical expulsive therapy. Both calcium-channel blockers and α-adrenergic blockers have demonstrated effects on ureteral smooth muscle, making them the logical target for clinical evaluations. Borghi and associates (1994) first reported a randomized, double-blind, placebo-controlled study that compared the effectiveness of nifedipine plus methylprednisolone with the effectiveness of the corticosteroid alone in hastening stone passage. They found that the calcium-channel blocker plus corticosteroid group experienced a greater rate of passage than the group treated with the corticosteroid alone. Since this report there have been other publications detailing varied combinations of medical treatments, including corticosteroid, nonsteroidal anti-inflammatory drug, calcium-channel blocker, and α-adrenergic blocker medications. These studies are difficult to interpret in aggregate because there is no uniformity in the medication studied, duration of treatment, adjunctive therapy, and inclusion criteria. The common theme across these studies, however, was that medical treatment with either α-adrenergic blockers or calcium-channel blockers increased the likelihood of spontaneous stone passage. The benefit of corticosteroids is small, however. Hollingsworth and associates (2006) performed a meta-analysis of these pharmacologic therapies, defining their efficacy in promoting the spontaneous stone passage. They found that there was a significant benefit for both calcium-channel blockers and α-adrenergic blockers in improving stone passage rates. However, the question of an optimum medical regimen was not definitively answered. A subsequent meta-analysis, performed by the AUA/European Association of Urology (EAU) Ureteral Stones Guidelines Panel, found that α-adrenergic blockers were superior to nifedipine and may be the preferred agent for medical expulsive therapy (Preminger et al, 2007). Importantly, if medical expulsive therapy is to be selected by the patient and physician, the patient must be reasonably comfortable with the therapeutic approach and counseled on the attendant risks, including drug side effects and that the drug is being administered for an “off label” use.
Stone Factors
Location
The location at which the passage of a ureteral stone is arrested is an important factor in assessing the likelihood of spontaneous passage as well as in determining the optimal treatment options and their relative successes. The statistical probability of spontaneous ureteral stone passage is directly related to the distance of the ureter to be traversed and inversely related to stone size.
Ureteral stones usually become impacted at three distinct sites where the caliber of the ureter narrows: the UPJ, the iliac vessels, and the ureterovesical junction. Historically the ureter was divided into thirds: the proximal ureter, from the UPJ to the upper edge of the sacrum; the middle ureter, from the superior to the inferior margin of the sacroiliac joint; and the distal ureter, from the inferior margin of the sacrum to the ureterovesical junction. This three-part division was based on the different open surgical approaches to stones in the ureter. However, with the development of minimally invasive techniques, open surgery is rarely indicated and the ureter is more often now divided into two segments, proximal and distal; the point of division is the narrow part of the ureter where it crosses the iliac vessels, reflecting a technical impediment for rigid ureteroscopy (Segura et al, 1997). Although in the early experience with endourology this impediment was reflected in inferior treatment outcomes for proximal ureteral calculi, as skill and equipment have evolved this effect has lessened (Tables 48-6 and 48-7).
Historically SWL was the preferred treatment for patients with proximal ureteral calculi. The 1997 AUA Nephrolithiasis Guideline Panel documented an 83% stone-free rate for SWL of proximal ureteral stones, a result that required 1.4 procedures per patient to achieve. In the distal ureter the results of SWL were 85%, requiring 1.3 treatments per patient. The updated guidelines of 2007 reported that SWL resulted in a stone-free outcome for 82% of patients treated for a proximal ureteral stone and 74% of patients treated for a distal ureteral stone. Interestingly, a statistical comparison of the two analyses confirms that the decline in efficacy for SWL of distal ureteral stones was statistically significant. There is no clear explanation for this change over the intervening decade. For those patients with proximal ureteral stones there was no difference in outcome if SWL was performed in situ or using a pushback technique.
With the increased dissemination of flexible ureteroscopy, the anatomic location of the targeted stone has had progressively less effect on treatment outcome. In 1997 the Guideline Panel recommended SWL for small stones in the proximal ureter and either SWL or ureteroscopy for large proximal ureteral stones. However, in the 2007 document, ureteroscopy is now deemed appropriate for stones of any size in the ureter. The stone-free rate for proximal ureteral stones was 81%, and for distal ureteral stones the stone-free rate was 91%. Stone-free rates were better for ureteroscopy than for SWL when treating distal ureteral stones of any size, as well as for proximal ureteral stones greater than 10 mm. The results for ureteroscopy with a flexible ureteroscope for proximal ureteral stones were better than those achieved with a rigid device, although this difference was not statistically significant.
In select cases with impacted stones in the upper ureter, which have failed treatment with SWL or ureteroscopy, percutaneous stone removal is a reasonable option (Maheshwari et al, 1999). In this way, rather than standard retrograde access, percutaneous antegrade access to the stone is established for subsequent endoscopic removal. Other cases in which a percutaneous approach for an upper ureteral stone may be appropriate would be in combination with renal stone removal and in cases of ureteral stones that occur after urinary diversion.
Stone Burden
The stone burden, in terms of both size and number, may affect what form of therapy will be the most efficient and confer the highest stone-free rate. The likelihood of a successful outcome with SWL is known to be adversely affected by an increasing stone burden for not just renal calculi but also ureteral calculi. In the 2007 AUA/EAU guideline, ureteral stones were classified as either less than or greater than 10 mm. At all locations in the ureter the treatment outcome for stones less than 10 mm was superior to that for stones greater than 10 mm. Specifically, for stones in the distal ureter the stone-free rate for stones less than 10 mm treated by SWL was 86%, compared with a 74% stone-free rate for those greater than 10 mm. For proximal ureteral stones, the trend held true, as 90% of patients with stones less than 10 mm achieved a stone-free outcome, compared with 68% with stones greater than 10 mm. Ureteroscopy is also affected by stone burden, but perhaps to a lesser extent. For distal ureteral stones treated with ureteroscopy, stone-free outcomes occurred in 97% of cases when the burden was less than 10 mm and in 93% of cases when the burden was greater than 10 mm. For stones in the proximal ureter the outcomes stratified by stone size were almost identical: 80% of patients were stone free for stones less than 10 mm and 79% were stone free for stones greater than 10 mm.
Stone Composition
Stone composition, if it is known, is useful information to help discern which treatment strategy may be favored (e.g., SWL for fragile calcium oxalate dihydrate stones or ureteroscopy and intracorporeal lithotripsy for cystine or brushite stones that are relatively resistant to SWL). In general, most ureteral stones are resistant to medical dissolution therapy. However, in the moderately symptomatic or asymptomatic patient with an incomplete obstruction caused by a uric acid stone, urine pH manipulation may be attempted as part of a conservative treatment approach. Urine pH manipulation should also be considered for the patient who does not desire or cannot medically tolerate anesthesia for SWL or ureteroscopy and who is thought to harbor a uric acid stone.
Duration of Stone Presence
The management of patients with ureteral stones may be affected by the duration of a stone’s presence or the patient’s symptoms. The length of time a stone has been in the ureter is significant because of the potential for irreversible loss of renal function. However, even with complete ureteral obstruction, irreversible loss of renal function does not occur before 2 weeks, although it can progress to total renal unit loss at up to 6 weeks (Vaughan and Gillenwater, 1971).
Andren-Sandberg (1983) reported that in 358 patients with ureteral stones, nuclear renography detected impaired renal function in 27% of patients who were asymptomatic. Furthermore, 7% of these patients had persistent renal impairment up to 17 months after stone passage. Holm-Nielsen and associates (1981) reported that of 134 patients with unilateral ureteral stones, one third of the patients with obstruction lasting more than 4 weeks suffered irreversible renal damage. Similarly, Kelleher and associates (1991) found that sequential renal scintigraphy performed on 76 patients with obstructive ureteral calculi demonstrated an 18% incidence of reduced renal function (defined as a decrease in relative function greater than 7%). Two of the 14 patients had persistent loss of renal function after treatment. These two patients had undergone previous unsuccessful attempts at stone removal, emphasizing the importance of achieving stone-free status at the time of the initial procedure. Irving and associates (2000) studied 54 patients with ureteral calculi and reported that 28% of patients had silent loss of renal function at presentation. Interestingly, small calculi were as likely to be associated with diminished renal function as were larger stones, and patients who underwent early intervention (before 7 days) for diminished renal function had a better outcome than did patients with delayed intervention. Because the patient’s symptoms and stone size do not predict loss of renal function, and because there is no clear time threshold for irreversible damage, intervention should be considered in any patient with ureteral obstruction unless the ability to closely monitor renal function is available.
Clinical Factors
Pain
Pain (renal or ureteral colic) is the primary presenting symptom of most patients with ureteral obstruction and is the source of considerable morbidity. The pain of ureteral colic is mediated by prostaglandins released by the ureter in response to obstruction. Prostaglandins act to increase ureteral peristalsis to aid in stone passage and also to sensitize nociceptors to stimuli such as bradykinins that induce pain and visceral responses (nausea, vomiting) (Selmy et al, 1994). For these reasons, prostaglandin inhibitors such as indomethacin and ketorolac are particularly effective in relieving the pain and nausea of ureteral obstruction and are commonly used for this purpose in the emergency department setting (Brown, 2006). However, prostaglandin inhibitors must be used with caution or avoided entirely in patients with compromised renal function; infusion of ketorolac in an acute unilateral ureteral obstruction model decreased ipsilateral renal blood flow by 35% (Perlmutter et al, 1993).
Management of ureteral obstruction on the basis of symptoms should be tailored to the amount of time the symptoms have persisted. Patients presenting with early symptoms of renal colic may be managed expectantly for stone passage as long as their symptoms are controllable with oral medical therapy. Patients with symptoms of longer duration may be more appropriately managed by relieving the ureteral obstruction through either the placement of a ureteral stent or definitive stone treatment. Patients with ureteral stones causing severe symptoms refractory to conservative and medical therapy require prompt treatment.
Infection
Infection associated with ureteral stones, or obstructed pyelonephritis, is a not uncommon and potentially life-threatening urologic emergency. Such patients are typically febrile and may present with signs of septic shock, such as hypotension. Urgent drainage of the obstructed portion of the urinary tract by either ureteral catheter or percutaneous nephrostomy is essential. A urine culture specimen from the obstructed segment will aid in directing antibiotic therapy. Definitive stone therapy should be delayed until urine cultures are negative and the patient has recovered completely.
The Patient’s Expectations
The patient’s expectations must be considered in recommending different treatment modalities. For ureteral stones with low probability of spontaneous passage the patient must be informed of the available treatments, including the relative benefits and risks associated with each. Although ureteroscopy is more invasive than SWL, the patient may achieve immediate or nearly immediate stone-free status with a single procedure. Although SWL is less invasive, a patient may be reluctant to select this modality because it entails an often lengthy follow-up until fragment clearance as well as the risk of unplanned additional invasive procedures and re-treatments. Conversely, patients may select SWL as a consequence of apprehension of the anesthesia associated with ureteroscopy or the possibility of a temporary ureteral stent.
Solitary Kidney
Patients with ureteral stones in either surgically or functionally solitary kidneys require a modification of standard treatment algorithms. A ureteral stone obstructing a solitary kidney demands prompt attention, usually with internal drainage and definitive stone treatment (SWL or ureteroscopy).
Aberrant Anatomy
Ureteral stones in patients with abnormal anatomy (ureteral ectopia, ureteroceles, megaureters) may have impaired egress because of obstructive or functional factors (Kajikawa et al, 1985; Diamond et al, 1994; Dretler, 1995). These patients may not respond to therapies such as SWL with the same level of success as do those with normal ureteral anatomy. The anatomic abnormality may need to be corrected or circumvented to permit successful treatment of ureteral stones. For example, patients with recurrent stones that become lodged and obstruct the ureter in a ureterocele should have the ureterocele treated (by either endoscopic incision or open excision with reimplantation) simultaneously with ureteral stone removal. Ureteroscopy, with modern endoscopes and implements, has been reported to be a successful technique with which to treat patients with unusual renal or ureteral anatomy (Weizer et al, 2005). One anatomic situation in which retrograde ureteroscopic treatment is likely not to be successful is that of the patient who has previously undergone a cross-trigonal ureteral reimplant. Krambeck and associates (2007) reported on a series of patients who had previously undergone ureteral reimplantation, finding that a cross trigonal reimplant was associated with a need for more invasive stone removal procedures and that ureteroscopy often failed in this population, because the kidney could not be accessed in a retrograde fashion.
Technical Factors
The treatment of patients with ureteral calculi depends on multiple surgical technologies, and the availability of certain equipment will affect the possible options for treatment. Few operating environments will have all possible lithotripters, ureteroscopes, lithotrites, or stone retrieval devices immediately available. In addition, the surgeon’s preference and technical expertise will also affect the technique chosen. The majority of urologists and patients with stone disease in the United States do not have direct access to a fixed lithotripter on an unlimited basis. Therefore, treatment decisions may need to be modified according to lithotripter availability. Patients with symptomatic ureteral stones and no immediate lithotripter access have several options: they may be clinically observed with pain and emetic control; they can undergo placement of an internal ureteral stent to relieve the symptoms of renal colic (but then may suffer stent-related morbidities); or they may have primary ureteroscopic removal of the stone, provided the requisite endoscopic equipment is available.
Because of recent pressures to decrease resource use there is increasing emphasis in the modern medical environment on the reduction of cost. Such economic pressures have promoted the movement toward less invasive, more cost-effective therapy for patients with ureteral stones. Most studies performed in the United States have found that ureteroscopy is a more cost-effective intervention than SWL, a differential that is likely due to the higher re-treatment rate associated with SWL. Grasso and colleagues (1995a) analyzed the cost of ureteroscopy and SWL for patients with ureteral calculi. When they compared outpatient ureteroscopic lithotripsy with SWL monotherapy, treatment costs were similar. However, the addition of re-treatments and auxiliary procedures after SWL more than doubled the costs and weighed heavily against this modality’s cost-effectiveness. Kapoor and associates (1992) compared the treatment costs of SWL and ureteroscopy: as a consequence of the significantly higher success rates (97% vs. 75%), ureteroscopy was the more cost-effective treatment approach. Parker and associates (2004) studied the cost associated with treating proximal ureteral stones and found that ureteroscopy was less costly, again as a consequence of its superior initial treatment success. Several investigators have approached the question of cost-effectiveness treatment for ureteral calculi by constructing decision analysis models. Wolf and associates (1995) used a literature-based probability decision tree to determine the more effective and cost-efficient therapy for patients with distal ureteral calculi and found that although initial SWL was only slightly more expensive than ureteroscopy the cost differential increased when additional complications and re-treatments were calculated. Lotan and associates (2002) also constructed a decision analysis model to determine the most cost-effective treatment for ureteral stones. Although observation was the least costly intervention, ureteroscopy was a less costly intervention than SWL for the treatment of stones at all locations in the ureter. Thus taken all together, ureteroscopy is the most cost-effective treatment strategy for ureteral stones at all locations, after observation fails.
Laparoscopic Ureterolithotomy
Laparoscopy should be considered whenever one contemplates the open surgical removal of a ureteral calculus. Although the indications for this surgery are not common there are certain patients for whom this may be an acceptable approach, such as those with concomitant upper tract abnormalities (e.g., ureteral stricture, UPJ obstruction) requiring surgical repair.
Both transperitoneal and retroperitoneal approaches have been described for laparoscopic ureterolithotomy (Wuernschimmel and Lipsky, 1993; Gaur et al, 1994). Keeley and associates (1999a) utilized laparoscopic ureterolithotomy in only 1.1% of patients treated in a 5-year period, with successful results in all cases. The indications for a laparoscopic procedure included stones that could not be accessed ureteroscopically, stones that did not fragment with other treatment modalities, and large (>1.5 cm) proximal ureteral stones. Confirming the superior morbidity of the procedure, Goel and Hemal (2001) reported the results of a prospective comparison of laparoscopic and open ureterolithotomy. A number of other centers have reported their successful experiences with the application of this technique (Turk et al, 1998; Gaur et al, 2002; El-Feel et al, 2007; El-Moula et al, 2008). Among these groups the techniques were varied; some describe suturing and stenting the ureter, whereas others leave the ureter unsutured and unstented. Further advancing laparoscopic stone surgery, Rane and associates (2008) reported the successful case of a single-port laparoscopic ureterolithotomy. Although many of the aforementioned authors suggest that laparoscopic ureterolithotomy may be used as a primary procedure, it is more likely that this operation will be reserved for those patients who require a salvage procedure after failed ureteroscopy or SWL or who have another indication for a laparoscopic procedure.
Key Points: Ureteral Calculi
Urinary Calculi During Pregnancy
Incidence
Urolithiasis is an infrequent complication of pregnancy. However, pain from renal colic is the most common nonobstetric reason for hospital admission during pregnancy (Rodriguez and Klein, 1988). Furthermore, the occurrence of urinary calculi during pregnancy presents danger not only to the mother but also to the fetus because renal colic, infection, and obstruction are all associated with premature labor (Maikranz et al, 1987; Hendricks et al, 1991). The reported incidence of symptomatic urinary calculi during pregnancy ranges from 1 in 200 to 1 in 2500 pregnancies; the wide variation in reported incidence may be due to the small numbers of patients in these studies (Gorton and Whitfield, 1997). However, the incidence of symptomatic urinary calculi has been calculated to be the same for pregnant women as for nonpregnant women of childbearing age (Coe et al, 1978; Hendricks et al, 1991). Multiparous women have been reported to be affected more often than primiparous women, in some cases by a ratio of about 3 : 1 (Horowitz and Schmidt, 1985; Rodriguez and Klein, 1988). However, when it is adjusted for age, the incidence for multiparous women is no greater than that for primiparous women (Swanson et al, 1995). Calculi present with equal frequency on the left and right sides, although ureteral calculi occur almost twice as frequently as renal calculi (Stothers and Lee, 1992; Parulkar et al, 1998). The majority of patients with symptomatic calculi present during the second or third trimesters but rarely during the first trimester (Denstedt and Razvi, 1992; Stothers and Lee, 1992; Swanson et al, 1995).
Although symptomatic urolithiasis may be an uncommon complication of pregnancy, renal colic during pregnancy is a serious concern, because such an event may be dangerous to both the mother as well as the fetus. Swartz and associates (2007) analyzed the hospital discharge data from 1987 through 2003 in the state of Washington and found that women admitted for nephrolithiasis had a significantly greater (adjusted odds ratio 1.8) risk of preterm delivery compared with women without stones. Lewis and associates (2003) also reviewed a large database comprised of over 21,000 deliveries and found that of the 86 patients diagnosed with a stone during pregnancy there was an increased risk of preterm premature rupture of membranes (2.9% in non-stone patients vs. 7% in stone patients). Preterm premature rupture of membranes, as the authors note, carries with it an increased risk of morbidity and mortality to the newborn.
Etiology
Pregnancy induces significant physiologic alterations, some of which affect the urinary system. The most remarkable anatomic change is the dilation of the renal calyces, pelvis, and ureters, which is usually evident by the first 6 to 10 weeks of gestation. Pregnancy-induced hydronephrosis is the most common cause of dilation of the urinary tract in pregnancy and may cause flank discomfort or even mimic renal colic. Upper tract dilation is seen in up to 90% of pregnant women by the third trimester and may persist for as long as 12 weeks postpartum (Boridy et al, 1996). The right ureter tends to be more dilated than the left, and the dilation rarely is observed distal to the pelvic brim (Schulman and Herlinger, 1975). Rarely, spontaneous rupture of the kidney may occur, and if it does, it more commonly happens on the right side (MacNeily et al, 1991; Loughlin, 1994). Both humoral as well as mechanical factors have been implicated in the etiology of hydronephrosis in pregnant women. Circulating progesterone, a humoral factor that is increased in pregnancy, causes relaxation of ureteral smooth muscle, reducing ureteral peristalsis. Paller and Ferris (1996) have reported that dilation of the urinary collecting system can be reproduced in an animal model by the administration of estrogen and progesterone. However, recent evidence suggests that mechanical factors, in particular the gravid uterus directly compressing the ureters, are likely to be primary in the pathogenesis of this condition; women with an altered upper urinary tract in whom the ureter does not cross the pelvic brim, such as those with ileal conduit or renal ectopia, do not experience hydronephrosis during pregnancy (Rasmussen and Nielsen, 1988; Dafnis and Sabatini, 1992; Swanson et al, 1995). Although the exact etiology of hydronephrosis of pregnancy is not yet well defined, most would agree that both mechanical and humoral factors play a role in the pathogenesis of this condition.
Other important physiologic changes in pregnancy include an increase in renal plasma flow, which induces a 30% to 50% increase in glomerular filtration rate. As a result of this physiologic alteration the normal ranges of serum creatinine and blood urea nitrogen are approximately 25% lower for the pregnant patient. Importantly then, a serum creatinine value that is in the normal range for the nonpregnant population may actually represent a decrease in renal function for the pregnant patient (Paller and Ferris, 1996). The increase in renal plasma flow and glomerular filtration rate also increases the filtered loads of sodium, calcium, and uric acid, causing a state of hypercalciuria and hyperuricosuria (Boyle et al, 1966; Howarth et al, 1977; Gertner et al, 1986). Hypercalciuria is further exacerbated by the suppression of parathyroid hormone and the increase in circulating 1,25-dihydroxycholecalciferol produced by the placenta, which increases intestinal absorption of calcium. Twenty-four-hour urine chemistries performed among pregnant women have also demonstrated that urinary pH is elevated in the course of pregnancy, most dramatically during the second trimester (Resim et al, 2006). However, these potentially lithogenic physiologic changes are offset by an increase in the excretion of urinary inhibitors, such as citrate and magnesium, as well as an increase in urine output (Biyani and Joyce, 2002). It has been postulated that metabolic alterations in the urine may contribute to the accelerated encrustation of ureteral stents during pregnancy (Denstedt and Razvi, 1992; Loughlin, 1994). Ross and associates (2008) have reported that stones that occur during pregnancy are most commonly calcium phosphate in composition, a finding that may be explained by the relatively elevated urinary pH and hypercalciuria that occur in the pregnant state.
Evaluation
Although renal colic is the most common nonobstetric cause of abdominal pain in hospitalized pregnant women, the diagnosis of urolithiasis in the pregnant patient can be challenging; many of the usual presenting signs and symptoms may be masked by the patient’s gravid status. As gestation progresses, the perception and localization of pain may be altered. Stothers and Lee (1992) reported that 28% of pregnant patients ultimately diagnosed with an obstructing stone were initially, and incorrectly, diagnosed with appendicitis, diverticulitis, or placental abruption. For most patients, however, the most common presenting symptom is flank pain, usually accompanied by either macroscopic or microscopic hematuria and, in some cases, urinary tract infection (Stothers and Lee, 1992). Hematuria can occasionally occur in the normal course of pregnancy; however, hematuria without discomfort is unusual in a patient with stone disease (Swanson et al, 1995). It is particularly important to obtain a urine specimen for culture from these patients because pyuria may commonly be seen in the urinalysis of a pregnant patient, which diminishes the sensitivity of this test in detecting urinary tract infection (Hendricks et al, 1991; Houshiar and Ercole, 1996; Parulkar et al, 1998). A diagnosis of urinary calculi should be considered in evaluation of a pregnant patient who suffers from persistent urinary tract infection or infection with a urea-splitting organism. Other symptoms that may indicate urolithiasis include irritative voiding symptoms, chills, nausea, and vomiting. However, these symptoms may also occur with other intra-abdominal conditions so the urologist must maintain a high index of suspicion when examining these patients.
An important factor in the radiographic evaluation of pregnant patients suffering from stone disease is the risk of ionizing radiation exposure to the fetus. The principal effects of irradiation on the fetus include teratogenesis, carcinogenesis, and mutagenesis. However, the risk associated with radiation depends critically on the gestational age and the amount of radiation delivered (Biyani and Joyce, 2002). During the first trimester, the period of early organogenesis and rapid cell division, the embryo is sensitive to the effects of radiation (Swartz and Reichling, 1978). Although the fetus has diminished sensitivity to the teratogenic effects of radiation in the second and third trimesters, such exposure may increase the risk for development of childhood malignant neoplasia (Harvey et al, 1985).
Because the radiation dose below which no deleterious effects on the fetus may occur has not been defined with certainty, it may be presumed that exposure to any level of radiation will carry some degree of risk. For this reason ultrasonography has become the standard initial study in evaluation of the pregnant patient thought to be experiencing renal colic. Unfortunately, it can be difficult to adequately visualize the ureter with ultrasound examination as well as to distinguish dilation of the ureter that may be associated with a normal pregnancy from ureteral obstruction due to calculus. Stothers and Lee (1992) reported that renal ultrasonography for the detection of calculi had a sensitivity of 34% and a specificity of 86%. Butler and associates (2000) similarly reported that ultrasonography diagnosed 60% of 35 women who were later proven to have nephrolithiasis. Several techniques have been recommended to improve the diagnostic capability of this technology. Color Doppler imaging allows the sonographer to differentiate the iliac artery and vein from the dilated ureter. MacNeily and associates (1991) reported that the use of this technique can distinguish a dilated infrailiac ureter, which was strongly correlated with ureteral obstruction. Color Doppler imaging can also demonstrate jets of urine expelled from the ureter into the bladder. Deyoe and associates (1995) reported that if there are no ureteral jets on the suspected side of obstruction, ureteral obstruction can be diagnosed with a sensitivity of 100% and a specificity of 91%. However, Burke and Washowich (1998) reported that there is variation in ureteral jet symmetry in later pregnancy and recommended the use of this technique with caution. Renal vascular resistance increases in the presence of acute obstruction, and duplex ultrasonography allows the quantification of this alteration by calculating the kidney’s resistive index (Ulrich et al, 1995). Shokeir and Abdulmaaboud (1999) prospectively evaluated 117 nonpregnant patients with ultrasonography; they reported that resistive index measurements had 77% sensitivity and 83% specificity in diagnosis of ureteral calculi and that change in resistive index had 88% sensitivity and 98% specificity. Horrigan and associates (1996) reported that renal resistive index remains unchanged from the nonpregnant state throughout the course of pregnancy and also is unaffected by the physiologic hydronephrosis of pregnancy, which suggests that this imaging modality may be useful in detecting acute obstruction in this population. Shokeir and associates (2000) evaluated pregnant women in a manner similar to their initial study and found that resistive index had a sensitivity of 45% and a specificity of 91% in detecting an obstructing ureteral calculus; change in resistive index had a sensitivity of 95% and a specificity of 100%. If an obstructing calculus cannot be visualized by conventional renal sonography, transvaginal ultrasonography can provide imaging of the distal ureter. In a series of 13 women, 6 of whom were pregnant, Laing and associates (1994) identified distal ureteral stones in all 13 patients with transvaginal ultrasonography and in only 2 by transabdominal ultrasonography.
If the clinician determines that ultrasound evaluation is inadequate, other imaging studies may be considered. If intravenous pyelography is required, a limited study is recommended. Stothers and Lee (1992) were able to visualize calculi in 16 of 17 pregnant patients with a three-film study, obtaining scout, 30-second, and 20-minute films. Nuclear renography is a technique that can provide a functional assessment of pregnant patients with suspected ureteral obstruction while exposing them to a limited amount of radiation. However, the radioisotope is excreted in the urine, and the bladder reservoir can provide a significant source of radiation exposure to the fetus, necessitating high fluid intake and frequent voiding for these patients (Biyani and Joyce, 2002). This radiographic technique unfortunately does not provide good anatomic detail or visualization of calculi. Magnetic resonance imaging (MRI) does not rely on ionizing radiation or contrast medium, making it a potentially attractive tool to evaluate pregnant patients. Because MRI does not visualize calcium, stones are seen as filling defects overlying the high signal intensity of urine (Fig. 48–9). The visualization of smaller stones with this technique is difficult (Hattery and King, 1995; Roy et al, 1995). Spencer and associates (2004) reported on the use of MRI to evaluate pregnant women with hydronephrosis and flank pain and found that this technique enabled the accurate distinguishing of physiologic hydronephrosis of pregnancy from hydronephrosis due to an obstructing ureteral calculus. White and associates (2007) have reported on a technique of low-dose radiation exposure CT for the evaluation of pregnant women; however, at present the reliability of low-dose CT in the diagnosis of urinary calculi is unresolved. Conventional CT should be avoided during pregnancy because the radiation dose is particularly high.
Treatment
Fifty to 80 percent of pregnant patients with symptomatic calculi will pass their stones spontaneously when treated conservatively with hydration and analgesia (Denstedt and Razvi, 1992; Stothers and Lee, 1992; Gorton and Whitfield, 1997; Parulkar et al, 1998). Intervention is required in approximately one third of patients, usually for pain uncontrolled by analgesia or signs of persistent obstruction and infection. When treatment is selected, it should be recognized that there is some controversy regarding the most appropriate method of intervention. Some have maintained that ureteral stents are the optimal treatment of such patients. Although ureteral stents do effectively drain an obstructed collecting system, they are by no means the perfect solution to this problem. The changes in urinary chemistry that occur during pregnancy, in particular the hypercalciuria and hyperuricosuria, have been implicated in the accelerated encrustation of ureteral stents that is encountered in this population. As a consequence of this phenomenon it has been recommended that ureteral stents placed in pregnant women be exchanged every 4 to 6 weeks. Ostensibly, then, for a woman in an early gestational stage, multiple stent changes will be required over the course of the pregnancy. An indwelling stent places these women at an increased risk for bacteriuria, urinary tract infection, as well as stent migration, all of which are serious morbidities that may have an adverse effect on the pregnancy. Ureteral stents themselves are associated with pain, which can have a negative impact on a patient’s quality of life.
Percutaneous nephrostomy drains are an alternative treatment option for pregnant women with obstructing renal calculi. Just as with ureteral stents, nephrostomy tubes will effectively drain an obstructed collecting system. However, many of the same limitations that apply to ureteral stents also apply to nephrostomy drains. Khoo and associates reported that of 29 pregnant women managed with nephrostomy drainage, over half required tube exchanges, replacements, or flushings that were required due to either dislodgement or obstruction. Kavoussi and associates (1992) also reported that the majority of pregnant patients managed with nephrostomy drainage will require exchange of the tube due to occlusion from debris. One third of the patients in the Kavoussi and associates’ series ultimately required nephrostomy removal due to recurrent drain obstruction, fever, or pain.
Both ureteral stent placement and nephrostomy drain placement are temporizing procedures that do not remove the obstructing symptomatic calculus. Therefore, both of these interventions imply that in the postpartum period the mother will require a definitive procedure to remove the calculus. It may be hypothesized that one of the advantages of both ureteral stent and nephrostomy drain placement is that neither of these procedures requires a general anesthetic. However, many of the reports of ureteroscopy in pregnancy have described local anesthesia, regional anesthesia, or sedoanalgesia, all approaches that obviate the need for general anesthesia. It is likely that recent improvements in surgical technology may be responsible for the increased utilization of ureteroscopy in the treatment of pregnant women. In recent years there have been great advances in both semirigid and flexible ureteroscopes. As recently as a decade ago, standard ureteroscope diameter ranged up to 11 Fr, in contrast to modern endoscopes that typically have a diameter of 6 to 8 Fr. Consequently, accessing all aspects of the renal collecting system in a safe and expedient manner is now a straightforward endeavor that generally does not require ureteral dilatation or other extraordinary maneuvers. The widespread utilization of intracorporeal lithotrites such as the holmium laser permits the safe and atraumatic fragmentation of calculi at any location. Improvements in flexible grasping devices have enhanced the efficiency of stone extraction.
Overall, complications in pregnant women undergoing ureteroscopy are uncommon. Semins and associates (2009) performed a meta-analysis of all reports of ureteroscopy of pregnant women to define the rate of complications in this population. They then compared the complication rate to the AUA/EAU Ureteral Stones Guidelines and found that there was no difference in the complication rates among pregnant and nonpregnant women undergoing ureteroscopy. Other treatment modalities that are effective in the nonpregnant patient are not appropriate for this population. Although there have been reports of the inadvertent treatment of pregnant patients with SWL, with no adverse sequelae to the fetus, pregnancy remains a contraindication to this treatment modality (Chaussy and Fuchs, 1989; Frankenschmidt and Sommerkamp, 1998). PNL should be deferred until after birth because this procedure often requires prolonged anesthesia and radiation exposure.
Stone Removal: Surgical Techniques and Technology
Intracorporeal Lithotripters
Ureteroscopy and PNL occupy an essential place in the treatment of urinary calculi as increasing technologic advancements allow easier access to stones in all parts of the kidney and ureter. In particular, improvements in ureteroscopic equipment emphasize the need for appropriate and effective miniaturized intracorporeal lithotripsy devices. Smaller ureteral stones can be extracted intact with endoscopic baskets or grasping devices after ureteral dilation, if necessary. However, larger ureteral stones require lithotripsy to permit the safe extraction of calculus fragments. The fragmentation of renal stones during PNL requires an approach different from that applied to ureteral intracorporeal lithotripsy. Although small and flexible endoscopic lithotrites are essential for the occasional difficult-to-approach kidney stone, renal stones can be visualized with a rigid nephroscope in most cases. In these situations, with a large kidney stone burden, the efficiency of the lithotrite is the most important requirement and size and flexibility are of secondary importance. The urologist who treats patients suffering from urolithiasis thus requires an armamentarium of intracorporeal lithotripsy devices, each maximizing a different quality (e.g., size, flexibility, efficiency).
Four techniques are available for intracorporeal lithotripsy: electrohydraulic lithotripsy (EHL), laser lithotripsy, ultrasonic lithotripsy, and ballistic lithotripsy. These techniques can be divided into those lithotrites that are flexible (laser lithotripsy and EHL) and those that are rigid (ultrasonic and ballistic lithotripsy). Here the mechanisms, advantages, disadvantages, and surgical techniques of the various flexible and rigid intracorporeal lithotripters are reviewed.
Flexible Lithotripters
Electrohydraulic Lithotripsy
EHL was invented in 1955 by Yutkin, an engineer at the University of Kiev, and was the first technique developed for intracorporeal lithotripsy (Grocela and Dretler, 1997). The first reported use of EHL outside the Eastern bloc was in 1960, when a modified version of Yutkin’s invention, the Urat-1, was used to fragment bladder calculi (Rouvalis, 1970). EHL was first applied to renal calculi during an open surgical lithotomy in 1975 (Raney and Handler, 1975). In 1985, Lytton reported the first experience treating patients with ureteral stones with a rigid ureteroscope and a 5-Fr EHL probe; no immediate or long-term complications were encountered (Green and Lytton, 1985). The use of smaller EHL probes through a flexible ureteroscope was first reported in 1988 (Begun et al, 1988).
The EHL probe is essentially an underwater spark plug composed of two concentric electrodes of different voltage polarities separated by insulation. When a current sufficient to overcome the insulative gap is applied, a spark is produced. The spark discharge causes the explosive formation of a plasma channel and vaporization of the water surrounding the electrode. The rapidly expanding plasma causes a hydraulic shockwave followed by formation of a cavitation bubble (Fig. 48–10). Depending on the proximity of the probe to the stone surface, the collapse of the cavitation bubble may be symmetrical (at a distance of about 1 mm from the stone), resulting in a strong secondary shockwave, or asymmetrical (at a distance equivalent to a maximum bubble radius of about 3 mm), leading to the formation of high-speed microjets (Vorreuther et al, 1995; Zhong et al, 1997). Unlike in SWL the shockwave is not focused, so the stone must be placed where the shockwave is generated. The first EHL probes developed were of larger diameters (9 Fr), which, because of their size, had a narrow margin of safety. Later improvements in technology allowed the development of smaller probes, from 1.6 to 5 Fr, that were safer and had the ability to be passed through small-diameter, flexible ureteroscopes without occluding the irrigation or working channel. There is little difference in fragmentation ability among the different-sized probes, but the larger probes tended to be more durable (Segura, 1999). Subsequent improvements in the EHL generator allowed the surgeon more control over energy discharge, pulse, and duration. Although it was originally hypothesized to function optimally in a  to
to 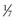 normal saline solution, Denstedt and Clayman (1990) demonstrated that EHL works equally well in a normal saline solution, eliminating the hazard of irrigating the upper urinary tract with a hypotonic solution.
normal saline solution, Denstedt and Clayman (1990) demonstrated that EHL works equally well in a normal saline solution, eliminating the hazard of irrigating the upper urinary tract with a hypotonic solution.
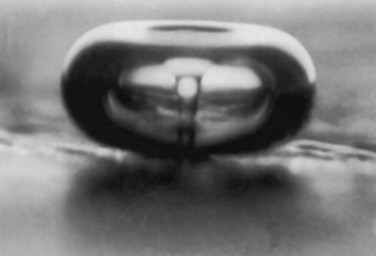
Figure 48–10 Photograph of liquid microjet produced by an asymmetrically collapsing cavitation bubble.
(Courtesy of Dr. Larry Crum.)
Advantages and Disadvantages
The major disadvantage of EHL is its propensity to damage the ureteral mucosa and its association with ureteral perforation. Raney (1978) reported that with a 9-Fr probe, 90% of ureteral stones could be successfully fragmented, but there was a 40% incidence of ureteral extravasation. Ureteral perforation remained an issue of concern with EHL in the ureter despite advancements in technology and technique. Hofbauer and coworkers (1995), in a prospective study of 72 patients, reported a perforation rate of 17.6% with EHL versus 2.6% with pneumatic lithotripsy. However, others have reported a lower rate of perforation, with a mean incidence of 8.5% recorded. Vorreuther and associates (1995) suggested that the mechanism of damage is the expansion of the cavitation bubble, and thus injury may occur even when the probe is not in direct contact with the mucosa. The diameter of the cavitation bubble depends on the energy used and can expand to more than 1.5 cm when energies greater than 1300 mJ are employed. Therefore the risk of perforation is greater with higher energies, such as in treatment of a hard stone. Even with smaller probes and lower energy settings, perforation may occur if repeated pulses are applied close to the mucosa. Santa-Cruz and colleagues (1998), in a comparative in-vitro study, reported that the holmium laser and EHL were associated with a higher risk of perforation compared with the coumarin pulsed-dye laser and pneumatic lithotripter. When the authors placed a 3-Fr probe 0.5 mm from the ureteral wall, perforation was induced with an average of 24 pulses. The risk of perforation may be higher for impacted stones associated with significant mucosal edema or if vision is impaired by the minor hemorrhage that commonly occurs during EHL (Hofbauer et al, 1995).
As with most lithotrites, retrograde propulsion of calculi and fragments can occur during EHL and is more pronounced than with holmium:YAG lithotripsy (Teichman et al, 1997). In a series of 43 patients treated for proximal ureteral calculi, 14% required subsequent SWL for stones that migrated into the kidney (Yang and Hong, 1996). Placement of a basket or other retention device above the stone may prevent stone retropulsion. However, care should be taken not to activate the EHL device directly on the basket wires or the guidewire. Another disadvantage of EHL compared with holmium:YAG lithotripsy is the larger number and size of fragments produced, especially for stones larger than 15 mm. The repeated passage of the ureteroscope to extract the multiple stone fragments produced during EHL may exacerbate mucosal irritation (Teichman et al, 1997).
EHL will successfully fragment 90% of stones. However, treatment failures may be due to a variety of stone compositions. Stone surface characteristics may also play a role in fragmentation efficiency; rough calculi have been reported to fragment more readily than smooth calculi (Basar et al, 1997). Although EHL successfully fragments most ureteral stones, the average 3-month stone-free rate is only 84% because some of the fragments created during lithotripsy and not removed may be retained in the ureter. Stone-free rates decrease with ureteral stones larger than 15 mm and are significantly lower than those reported for holmium:YAG lithotripsy (67% vs. 100%) (Teichman et al, 1997).
The advantages of EHL include probe flexibility, especially the smaller probes such as the 1.9 Fr, which allows intracorporeal lithotripsy throughout the entire upper urinary tract through rigid or flexible ureteroscopes. Only the holmium:YAG laser, configured with the 200-µm fiber, offers comparable size and flexibility advantages (Elashry et al, 1996). The 1.6-Fr EHL probe may be even more flexible than the 200-µm laser fiber (Poon et al, 1997).
EHL is also the least costly intracorporeal device, requiring the purchase of a comparatively inexpensive generator and probes. An average of 1 to 1.3 probes are used per case, except in instances of harder stones (e.g., calcium oxalate monohydrate stones), when two or more EHL probes may be needed (Elashry et al, 1996; Huang et al, 1998).
Technique
For intraureteral lithotripsy, the smaller 1.6- and 1.9-Fr probes should be used. The EHL fiber tip should be positioned 2 to 5 mm distal to the end of the ureteroscope to protect the lens system from being damaged when the probe is discharged. Before the EHL generator is activated the stone must be clearly visible. The probe is placed about 1 mm from the stone surface, a distance allowing maximum shockwave emission (Zhong et al, 1997). Initially, low voltage (50 to 60 V) and short intermittent or single pulses are used to enhance safety. The generator output is increased as needed to fragment the stone. However, it is recommended that the treating physician limit the maximum output used in treating ureteral stones to minimize the risk of perforation. The goal of the treatment is to create fragments that can be removed with grasping forceps or a basket device or fragments that are likely to pass spontaneously. Attempts to reduce the stone to fragments smaller than 2 mm are not recommended because damage to the urothelium may occur (Denstedt and Clayman, 1990). After 50 to 60 seconds of firing, the insulation at the tip of the probe may peel away and at this time a new probe should be used (Segura, 1999).
Laser Lithotripsy
Laser is an acronym for light amplification by stimulated emission of radiation, which is a concise description of how a laser works. Laser energy is produced when an atom is stimulated by an external energy source, which creates a population of electrons in an excited state. These excited or higher-energy electrons can release their excess energy in the form of photons or light energy. Laser light differs from natural light in that it is coherent (all photons are in phase with one another), collimated (photons travel parallel to each other), and monochromatic (all photons have the same wavelength (Floratos and de la Rosette, 1999). These unique features of laser light allow considerable energy to be transmitted in a highly concentrated manner. Lasers are named after the medium that generates their specific wavelength of light; for example, the laser was developed in 1960 and the first medium used was the ruby. In 1968, Mulvaney and Beck reported that although the ruby laser could effectively fragment urinary calculi it generated excessive heat and was not appropriate for clinical use. This continuous-wave laser simply heats the stone until vaporization occurs, which requires the laser to generate heat greater than the melting point of the stone. A solution for this problem came with the development of pulsed lasers: the application of pulsed energy results in high-power density at the stone’s surface but little heat dissipation. The first widely available laser lithotrite was the pulsed-dye laser, which employed a coumarin green dye as the liquid laser medium. Although the coumarin pulsed-dye laser represented a major advancement in intracorporeal lithotripsy there were a number of significant drawbacks to this technology: stones of certain composition (calcium oxalate monohydrate, cystine) would not fragment well or even at all, coumarin dye is a toxic agent and required cumbersome disposal procedures, and the required eye protection made visualization of the stone and fiber difficult.
Continued technologic advancements eventually led to the development of the holmium:YAG laser. The holmium laser is a solid-state laser system that operates at a wavelength of 2140 nm in the pulsed mode. Pulse duration of the holmium laser ranges from 250 to 350 µsec and is substantially longer than the pulse duration in pulsed-dye lasers. The holmium laser is highly absorbed by water; and because tissues are composed mainly of water the majority of the holmium laser energy is absorbed superficially, which results in superficial cutting or ablation. The zone of thermal injury associated with laser ablation ranges from 0.5 to 1.0 mm (Wollin and Denstedt, 1998). The mechanism of stone fragmentation of the holmium:YAG laser is different from that of the pulsed-dye lasers. The long holmium:YAG pulse duration produces an elongated cavitation bubble that generates only a weak shockwave, in contradistinction to the strong shockwave produced by short-pulse lasers. Vassar and associates (1999) demonstrated that during holmium lithotripsy, stone fragmentation began before bubble collapse and shockwave production. Furthermore, no stone fragmentation occurred when the fiber was discharged at an incident angle of 90 degrees. Lithotripsy was more efficient for dry stones in air, indicating that the holmium laser requires direct absorption of laser energy. These data as well as the presence of thermal products after holmium irradiation, such as glowing hot stone fragments, indicate that holmium laser lithotripsy occurs primarily through a photothermal mechanism that causes stone vaporization (Dushinski and Lingeman, 1998; Wollin and Denstedt, 1998; Vassar et al, 1999).
Advantages and Disadvantages
The holmium:YAG laser can transmit its energy through a flexible fiber, which facilitates intracorporeal lithotripsy throughout the entire collecting system. However, compared with EHL, the holmium:YAG laser is safer and more efficient. Whereas EHL may cause injury to the ureter even when the probe is activated several millimeters away from the ureteral wall, the holmium laser may be safely activated at a distance of 0.5 to 1 mm from the ureteral wall (Santa-Cruz et al, 1998). The ability of the holmium laser to fragment all stones regardless of composition is a clear advantage over the coumarin pulsed-dye laser. Successful fragmentation of ureteral stones of all compositions has been reported, and mean perforation and stricture rates are generally in the range of 1% to 2%. During PNL the holmium laser is most helpful in clearing smaller stones (<2 cm) when the use of flexible instruments is required for access to stones in a calyx remote from the nephrostomy site (Wollin and Denstedt, 1998). The holmium laser is one of the safest, most effective, and most versatile intracorporeal lithotripters. Further advantages of the holmium laser include its production of significantly smaller fragments as compared with other lithotrites. These small fragments are easily irrigated out of the collecting system, which reduces the need for extraction of the fragments with basket or grasping devices (Teichman et al, 1998b). The holmium laser produces a weak shockwave, which reduces the likelihood of retropulsion of the stone or stone fragments compared with EHL or pneumatic lithotrites (Teichman et al, 1998b; Vassar et al, 1999; Sofer and Denstedt, 2000). However, the 365- and 550-µm laser fibers will cause significantly more retropulsion than the 200-µm fibers (White et al, 1998). Of note, Kang and associates (2006) have demonstrated that not just the laser fiber size, but also laser settings, such as the pulse duration, will also affect stone retropulsion. The authors found that stone retropulsion could be significantly reduced by increasing the laser pulse duration.
The holmium laser has several distinct operating advantages compared with the coumarin pulsed-dye laser. The required eye protection for the holmium laser does not compromise the ureteroscopic view of the stone or the fiber (Segura, 1999). In fact, the holmium laser properties are such that with use of energy levels applied for stone disease (i.e., less than 15 watts) the operator’s cornea would be damaged only if it were positioned at a distance of 10 cm or less from the fiber (Scarpa et al, 1999). The holmium laser is more compact than the coumarin laser, requires minimal maintenance, and is ready for use 1 minute after it is turned on.
The major disadvantage of the holmium laser is the initial high cost of the device and the cost of the laser fibers. Elashry and coworkers (1996) noted an advantage of EHL over holmium laser lithotripsy in capital and service contract cost and lithotripter cost per case. However, the holmium laser has multiple soft tissue applications and can be used to treat patients with benign prostatic hyperplasia, strictures, and urothelial tumors. In addition, the laser fibers are reusable, so that the effective cost of the holmium laser device and reusable fibers may be lower than that of EHL (Teichman et al, 1998b). The most significant improvement in holmium laser lithotripsy will likely come from improved delivery fibers. At present, the smallest fiber in widespread use, the 200-µm fiber, impedes deflection of a flexible ureteroscope by up to 20 degrees. As smaller laser fibers, such as those of 150-µm diameter and smaller, are produced, it is likely that this effect on endoscope deflection will be further reduced. The fracture of a laser fiber inside of an endoscope can result in a catastrophic failure of the scope, as when this occurs the fiberoptic bundles that transmit images and light are generally destroyed. Future efforts toward maximizing fiber durability may reduce these events.
A potential side effect of holmium laser lithotripsy is the production of cyanide when uric acid stones are treated, which has been reported in vitro. However, a review of clinical experience suggests no significant cyanide toxicity from holmium laser lithotripsy (Teichman et al, 1998a).
Technique
The technique of holmium laser lithotripsy is relatively straightforward and involves placement of the fiber on the stone surface before the laser is activated (Table 48–8). Clear vision is essential at all times to avoid mucosal perforation. After initiation of holmium laser lithotripsy, a short pause is often required because of the “snowstorm effect” created by the scattering of minute stone fragments, which can be cleared by endoscopic irrigation (Scarpa et al, 1999). Caution must be exercised in operating the holmium laser near a guidewire or a basket because the holmium laser is capable of cutting through metal (Freiha et al, 1997; Lane et al, 2005). Furthermore, the laser fiber should extend at least 2 mm beyond the tip of the endoscope to avoid destroying the lens system or the working channel of the endoscope. Baskets used to stabilize calculi during laser lithotripsy should be the preformed type and not the type manufactured by bending of the wire, so if they are inadvertently transected by the holmium laser they will retain the basket shape and will not cause a sharp barbed effect (Grasso and Chalik, 1998).
Table 48–8 Holmium Laser Lithotripsy: Points of Technique
| Do |
| Don’t |
Holmium laser fibers are available in 200-, 365-, 550-, and 1000-µm diameters as well as end- or side-firing fibers. However, only the 200- and 365-µm fibers are used for flexible intracorporeal lithotripsy. Teichman and colleagues (1998c) reported that the 550-µm side-firing fiber is more effective than the end-firing fiber during PNL, suggesting that the more nearly normal (perpendicular) laser-to-calculus incident angle provided by the side-firing fiber enhances lithotripsy. However, in treatment of ureteral stones, the end-firing fiber may produce a better angle of attack. Lithotripsy with the holmium laser depends on the pulse energy output and the diameter of the optical delivery fiber, suggesting that lithotripsy efficiency correlates with energy density (Vassar et al, 1998). Energy density increases with decreasing fiber diameter, although Calvano and associates (1999) demonstrated, in vitro, that peak lithotripsy occurred with 365- and 550-µm fibers, whereas the 200-µm fiber can act as a fine drill, which is less effective. Compared with some of the soft tissue applications of the holmium laser, the power used for stone fragmentation is considerably lower. In general, pulse energies of 0.6 to 1.2 J and pulse rates of 5 to 15 Hz are used (Wollin and Denstedt, 1998; Spore et al, 1999). Because high-pulse energy narrows the safety margin and may increase stone retropulsion as well as fiber damage, it is recommended that treatment be begun with low-pulse energy (e.g., 0.6 J) with a pulse rate of 6 Hz and that pulse frequency be increased (in preference to increasing pulse energy) as needed to speed fragmentation (Spore et al, 1999). To maximize lithotripsy efficiency, the treating physician should move the laser fiber over the stone surface in a “painting” fashion, vaporizing the stone rather than fragmenting it, and avoid drilling into the stone, fracturing the fiber tip, or drilling past the stone, damaging the urothelium. The laser fiber should be kept at least 1 mm from the urothelium, and lithotripsy should proceed until the stone fragments are small enough to be passed spontaneously or can be safely retrieved with a basket or grasping device.
Rigid Lithotripters
Ballistic Lithotripsy
Ballistic lithotripsy relies on energy generated by the movement of a projectile (Fig. 48–11). The initial movement of the projectile can be induced by a variety of stimuli, but once the projectile is in contact with another object the ballistic energy is transferred to the object. Flexible objects preserve the momentum of the energy, but inflexible objects, such as a stone, fragment on impact (a “jackhammer” effect).
Figure 48–11 Schematic illustration of the LithoClast (Electromedical Systems, Kaufering, Germany) handpiece mechanism. An oscillating pellet provides ballistic energy to the probe, resulting in a jackhammer-like effect on calculi.
(Courtesy of Dr. John Denstedt.)
Several manufactures have introduced ballistic lithotrites. The Swiss LithoClast, introduced in the early 1990s, was the first ballistic lithotrite. The metal projectile in the handpiece of the LithoClast is propelled by measured bursts of compressed air against the head of a metal probe at a frequency of 12 cycles per second. The probe tip is placed against the stone, and the LithoClast is activated by a foot pedal (Denstedt et al, 1992). Following the introduction of the LithoClast, the electrokinetic lithotripter was introduced in the mid 1990s (Menezes et al, 2000). The electrokinetic lithotripter consists of a rheostat and a handset containing an electric coil that generates an electromagnetic field, which then vibrates the probe at 15 to 30 cycles/sec. Whereas the LithoClast is connected to the hospital central air supply or to a compressed air tank, the electrokinetic lithotripter requires electrical power. In both instruments the discharged probe is brought back into its former position by a rubber bushing around the base of the probe (Vorreuther et al, 1998). Studies comparing the LithoClast and the electrokinetic lithotripter, using the same stone fragmentation mechanism, showed no significant difference in stone fragmentation, proximal stone migration, and safety margin. However, the handpiece of the electrokinetic lithotripter is significantly heavier than that of the LithoClast (Vorreuther et al, 1998; Menezes et al, 2000). Rane and associates (2008) first reported on a novel, hand-held ballistic lithotripter, the StoneBreaker. This portable device utilizes a small cylinder-based air supply, rather than hospital air, simplifying its ergonomic profile.
Advantages and Disadvantages
The ballistic lithotrites provide an effective means for stone fragmentation in the entire urinary tract, with a wide margin of safety. Successful fragmentation of ureteral stones of all compositions has been reported in 73% to 100% of cases, a success rate similar to that of EHL. The lower success rate of 73.7% reported by Knispel and associates (1998) suggests reduced efficiency of the LithoClast when it is applied through the deflected working channel (30 degrees) of the 6.9-Fr semirigid ureteroscope. As well, a significant decrease in the maximum tip displacement and velocity of the LithoClast 0.89-mm flexible probe occurs when it is used through a flexible ureteroscope deflected more than 24 degrees (Zhu et al, 2000). Grocela and Dretler (1997) also reported that for the current ballistic devices, bowing of the probe during lithotripsy results in significant power loss. Ballistic devices may be especially advantageous when large or hard stones are encountered during PNL or endoscopic lithotripsy of bladder calculi. In contrast to ureteral stones, kidney stones are easily “pinned down” against the urothelium during ballistic lithotripsy, allowing a rapid and more efficient fragmentation method than ultrasonic lithotripsy. Once the bulk of the calculus is fragmented, lithotripsy can be completed with the ultrasonic lithotripter, which can also aspirate minute stone fragments (Denstedt, 1993; Teh et al, 1998; Yavascaoglu et al, 1999). Compared with EHL, ultrasonic lithotripsy, and laser lithotripsy, ballistic devices have a significantly lower risk of ureteral perforation (Piergiovanni et al, 1994). In an animal model, despite 6 minutes of activation in direct contact with the ureteral wall, a ballistic lithotripter was unable to cause perforation (Santa-Cruz et al, 1998). Furthermore, because no heat is produced during lithotripsy, the risk of thermal injury to the urothelium is eliminated.
One of the advantages of ballistic lithotrites is their relatively low cost and low maintenance. Although the devices are more expensive than EHL, in terms of capital equipment purchasing, there are no disposable costs and the probes have an extremely long life span (Hofbauer et al, 1995).
Disadvantages of ballistic devices include the rigid nature of the technology, which requires ureteroscopes or nephroscopes with straight working channels. In addition, ballistic lithotripsy is associated with a relatively high rate of stone retropulsion, reported in 2% to 17% of ureteral stone treatments. Often, failure to fragment a stone is related to an inability to trap a ureteral stone in a capacious ureter (Denstedt et al, 1992). The migration rate depends on the initial stone location; there is a higher chance of stone migration for proximal ureteral stones compared with distal ureteral stones (Knispel et al, 1998). Limited data are available on the beneficial effects of suction devices, such as the LithoVac, in limiting stone migration. Delvecchio and colleagues (2003) reported the use of the 0.8-mm pneumatic lithotripsy probe placed through a 4.8-Fr hollow LithoVac suction probe in 21 patients with ureteral stones. Overall stone-free rate at 3 months was 95%, and the suction device reportedly facilitated lithotripsy by preventing stone migration and maintaining a clear endoscopic view.
Teichman and associates (1998b) reported that fragments larger than 4 mm are produced by all types of endoscopic lithotrites, with the exception of the holmium:YAG laser. Fragmenting a stone into pieces smaller than 4 mm with a ballistic lithotripter can be challenging, especially a hard stone in a dilated ureter. Fragments larger than 4 mm are associated with a higher rate of repeated ureteroscopy and therefore should be removed with baskets or stone graspers during the initial procedure (Keeley et al, 1999b).
Technique
Like other lithotrites, the ballistic lithotripter should be activated only when there is a clear view of the stone and the probe position can be identified. Fixation of the stone is rarely difficult in the kidney or the bladder but may be a problem in the ureter. Fixation of ureteral stones with a basket or proximal placement of a ureteral occlusion balloon is sometimes necessary. The goal of ballistic lithotripsy in the ureter is to generate fragments that are small enough to permit spontaneous passage (<2 mm). However, more often, larger fragments have to be removed with a basket or grasping device. The relatively atraumatic nature of ballistic lithotripsy may allow the avoidance of stent placement after ureteroscopy. Tan and colleagues (1998) reported the use of a stent in only 9 of 68 patients undergoing ballistic lithotripsy. In this series, difficult ureteral access and severe edema and trauma at the site of stone impaction were indications for stent placement.
Ultrasonic Lithotripsy
Mulvaney first reported the use of ultrasound vibrations to break renal calculi in 1953. Since then, ultrasonic lithotripsy has become a commonly used modality for the treatment of renal calculi during PNL as well as for the fragmentation of bladder and ureteral stones. The ultrasound probe works by applying electrical energy to excite a piezoceramic plate in the ultrasound transducer (Fig. 48–12). The plate resonates at a specific frequency and generates ultrasonic waves at a frequency of 23,000 to 25,000 Hz. At operating frequencies there is no audible sound, although 98 dB of ultrasonic inaudible noise levels have been measured (Segura and LeRoy, 1984).
Ultrasound energy is transformed into longitudinal and transverse vibrations of the hollow steel probe, which then transmits the energy to the calculus. The probe tip causes the stone to resonate at high frequency and to break; but when the probe is placed on compliant tissue, such as urothelium, damage is minimal because the tissue does not resonate with the vibrational energy (Grocela and Dretler, 1997). Although some heat may develop at the end of the probe during lithotripsy, with an irrigation rate of 30 mL/min the temperature increase at the tip of the probe can be reduced to a maximum of 1.4° C (Marberger, 1983). Because irrigation may be limited during ureteroscopy, ultrasonic lithotripsy is more efficient during PNL, owing to the greater flow of irrigant through the larger diameter ultrasonic probes that can be used. The ultrasonic lithotripter system is connected to suction so that debris from the stone is removed continuously with the irrigating fluid during lithotripsy. In addition, the flow of fluid through the hollow probe serves to cool the instrument. Heating of the ultrasound transducer should alert the surgeon to possible occlusion in the probe lumen, an occurrence more commonly encountered with small-diameter probes that are used in the ureter. Although many manufacturers provide an integrated power and suction foot switch for the ultrasonic unit, wall suction with intermittent clamping of the suction tubing by an assistant is a simple and inexpensive alternative. In general, suction is applied only when the ultrasonic lithotripter is activated, and suction pressures in the range of 60 to 80 cm H2O are sufficient to maintain adequate flow of irrigant during lithotripsy. Higher suction pressures tend to draw air bubbles into the system, impeding vision. Ultrasonic probes are available at sizes ranging from 2.5 to 12 Fr. The 2.5-Fr probe is solid and contains no hollow center for suction. Therefore, when it is used in the ureter, heat dissipation is slow. Bending the probe results in energy loss at the convexity of the bend, with the energy being transformed to heat (Marberger, 1983).
Stones vary in their susceptibility to destruction with ultrasound. Although the chemical composition of the stone influences the time required for complete disintegration (cystine, calcium oxalate monohydrate, and uric acid being the most resistant to fragmentation), the size, density, and surface structure of the calculus appear to be more important. Smaller stones are more rapidly destroyed, as are rough stones. Smooth-surfaced large stones may be more difficult to fragment (Marberger, 1983; Segura and LeRoy, 1984).
Auge and associates (2002a) have reported on a new combination intracorporeal lithotripter that incorporates ballistic lithotripsy and ultrasonic lithotripsy modalities (LithoClast Ultra). In their in-vitro study of this device it was found to more efficiently fragment and clear phantom stones compared with a standard ultrasonic or ballistic device. These findings were confirmed in a subsequent in-vitro study by Kuo and associates (2004).
Advantages and Disadvantages
The major advantage of ultrasonic lithotripsy is the efficient combination of stone fragmentation and simultaneous fragment removal. Fragments smaller than 2 mm are aspirated through the hollow lithotrite along with the irrigation fluid. Larger fragments may be removed with forceps or baskets. The efficiency of this technique coupled with the minimal risk of serious tissue damage has made this technology popular. Ultrasonic lithotripsy is often the first modality used for stone fragmentation during PNL.
However, the rigid nature of ultrasonic probes and their small diameter limit the appeal of this technology in treatment of ureteral stones. A ureteroscope with a straight working channel is required. Furthermore, a relatively large 5-Fr working channel is needed to accommodate the 4.5-Fr hollow probe. However, success rates between 69% and 100% have been reported (Denstedt, 1996; Gur et al, 2004). The technology may be particularly useful for patients with large ureteral stones as well as for those suffering from steinstrasse because removal of stone debris is facilitated. Excellent results have also been reported for distal ureteral stones easily accessible to the rigid ureteroscope (Grocela and Dretler, 1997; Segura, 1999). Chaussy and colleagues (1987) reported a 96.6% complete fragmentation rate in 118 patients with a 2.5-Fr solid probe that can be used with smaller ureteroscopes, and Fuchs (1988) has reported similar results. However, in a later report, Murthy and associates (1997) compared a group of 25 patients treated by a rigid ureteroscope and the 3-Fr ultrasonic solid probe with a group of 122 patients treated by the LithoClast ballistic device, and the overall success rate was significantly higher for the LithoClast group than for the ultrasonic group (97.3% vs. 84%, respectively).
Technique
When ultrasonic lithotripsy is applied during PNL the stone should first be trapped between the probe and the urothelium. The application of gentle pressure to the stone enhances fragmentation, but the temptation to push too hard should be avoided because calculi can easily be pushed through the urothelium. The risk of perforation increases with smaller or more ruggedly surfaced stones as the force applied to the stone is transferred to a smaller surface area of the urothelium. The risk of perforation is particularly high in the thin-walled renal pelvis or ureter rather than in a calyx that is backed by renal parenchyma.
When ureteral stones are treated the ureter may need to be dilated to allow passage of the offset rigid ureteroscope. The ultrasonic probe is passed through the working channel and placed directly on the stone. If necessary, the stone can be engaged in a stone basket to prevent proximal migration. As with other intracorporeal lithotripsy devices the goal of treatment is either to fragment the stone completely or to generate fragments that are small enough to be extracted or passed spontaneously.
Combination Ballistic and Ultrasonic Devices
Several manufacturers have introduced combined ultrasonic and pneumatic devices that aim to combine the superior fragmentation ability of the pneumatic component with the ability of the ultrasonic modality to simultaneously evacuate stone fragments. The first combination device brought to the clinical market was the Lithoclast Ultra (Boston Scientific, Natick, MA), which relied on a combination handpiece (actually, two separate handpieces connected together) to join the ultrasonic and pneumatic components. The first portion of the combination handpiece was a traditionally designed pneumatic handle, with a smaller-diameter solid probe. The ultrasonic handle, driven by a standard piezoelectric mechanism, was modified to allow the coaxial insertion of the pneumatic probe. Each modality can be activated separately or in unison; when operated in unison, the ballistic fragmentation of the stone is accomplished with the pneumatic component and the ultrasonic component then removes the resulting debris.
Given the varied types of rigid intracorporeal devices (stand-alone ballistic and ultrasonic as well as combination ballistic and ultrasonic), a rigorous and impartial evaluation of intracorporeal lithotripters is a subject of importance to urologists. Each device may have certain unique properties that make it more suitable for particular applications, and manufacturer’s claims may contain elements of bias that make it difficult for the urologist to ascertain which device may be most suitable to purchase. Therefore, a number of investigators have devised testing methods to compare intracorporeal lithotrites. Liatsikos and associates (2001) first reported an in-vitro testing system designed to measure the efficiency of ultrasonic lithotrites in which stone phantoms were fragmented in a nephroscope-guided manner. The inherent weakness in this study design was that stone fragmentation was directed by hand, which could introduce significant operator bias. Haupt and Haupt (2003) subsequently reported an in-vitro system that relied on an elaborate weight and fulcrum to bring a stone phantom into contact with the probe tip at a constant force. Although operator bias was no longer present, this system was complex and cumbersome, making replication challenging. Kuo and associates (2003b) have presented a novel and simple hands-free testing system in which the ultrasonic handpieces were secured upright and the stone phantom placed into contact with the probe by a weight mechanism (Fig. 48–13). This design system was first utilized to test the efficiency of pure ultrasonic lithotrites and measured the time it took for the probe to penetrate the stone phantom. In this study the Olympus LUS-2 (Olympus, Melville, NY) produced the fastest overall stone penetration time.
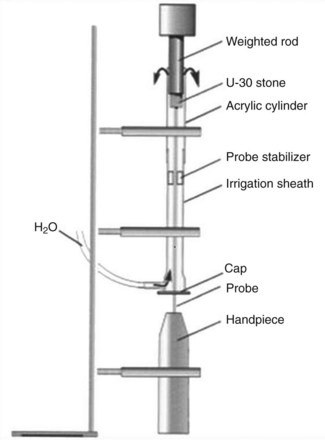
Figure 48–13 In-vitro testing apparatus for a “hands free” testing approach to the evaluation of intracorporeal lithotrites.
Following the introduction of the combination ultrasonic and pneumatic devices, the same testing apparatus previously used by Kuo and associates (2004) to evaluate the ultrasonic devices was used to evaluate the LithoClast Ultra. Because of the wide variety of ultrasonic power and pneumatic frequency settings available the testing apparatus was used to assess the efficiency of various setting combinations. The end point was stone penetration time, and the fastest stone penetration times were achieved at settings of 100% ultrasonic power and 12-Hz pneumatic frequency. Pietrow and associates (2003b) have evaluated the efficiency of the Lithoclast Ultra combination device in a clinical setting, performing a prospective, randomized trial comparing the combination device with standard ultrasonic lithotrites in patients undergoing PNL. The stone clearance times were significantly better for the combination device than for the conventional ultrasonic lithotripters.
The Cyberwand (Gyrus ACMI, Southborough, MA) is an intracorporeal lithotripter that relies on a dual ultrasonic probe design that incorporates coaxial high-frequency and low-frequency probes. The dual probe design creates a synergistic effect, which enables efficient stone fragmentation while still allowing the suction evacuation of small fragments just as other ultrasonic devices do. Kim and associates (2007) used the aforementioned hands-free testing design previously described by Kuo and associates to find that the stone penetration time for the Cyberwand was almost twice as rapid as it was for the Lithoclast Ultra.
Conclusion
The current technology of intracorporeal lithotripsy provides the urologist with several effective options for stone fragmentation, depending on the type of endoscope used (rigid or flexible) and the location and accessibility of the stone. The holmium laser has become the mainstay of ureterorenoscopic lithotripsy by virtue of its ability to fragment all stones. As well, the use of small-diameter fibers allows access to all areas of the ureter and intrarenal collecting system. However, for patients with complex, large-volume calculi undergoing PNL, the combination devices will permit more efficient fragmentation of the stone. When selecting a lithotripter for purchase, the institution must take into account the number and nature of stone-related procedures performed to maximize the utility and cost-effectiveness of the device.