chapter 90 Development, Molecular Biology, and Physiology of the Prostate
The focus of this chapter is on the development, anatomy, histology, and physiology of the prostate and seminal vesicles, the male sex accessory glands that contribute to seminal fluid. It is worth noting that these roles are deduced by anatomic and physiologic observation but that male infertility is infrequently traced to disorders of these organs. Therefore, their physiologic roles in reproduction are either unimportant or, as the authors believe, exceedingly resistant to malfunction. In the specific case of the prostate, its physiologic role is overshadowed by a striking vulnerability toward other types of disease—pathologic prostate growth represents an especially important cause of morbidity and mortality in men. Because pathologic prostate growth in adults involves reactivation of physiologic growth and differentiation pathways that act in embryonic and perinatal development (Marker, 2008; Schaeffer et al, 2008; Pritchard et al, 2009), regulation of these events is a major scientific and clinical question and comprises the main discussion in this chapter. Our understanding of prostate growth control is based on phenotypic evaluation humans with genetic diseases affecting the androgen receptor (AR) signaling axis and on analysis of laboratory animals or cultured cells subjected to genetic, surgical, or pharmacologic manipulation. The experimental data represent the broadest and deepest vein of information on prostate growth control and are the major source for the discussion of this topic. Possible physiologic roles of cellular constituents of the prostate are described in the final section of this chapter, and this discussion may be useful for students of prostate biomarkers.
Developmental and Cell Biology
Seminal Vesicles
The sex accessory tissues include the epididymis, ampullae, seminal vesicles, prostate, Cowper (bulbourethral) gland, and glands of Littre. All of these glands have reproductive roles, but the seminal vesicles work in tandem with the prostate and provide an important counterpoint to its biology and pathologic processes. The seminal vesicles are two saccular glands that pair with the vasa deferentia to form the ejaculatory ducts that empty into the craniodorsal aspects of the prostate. Together with the prostate the seminal vesicles produce seminal fluid that nurtures, protects, and facilitates sperm transport for mammalian reproduction. The division of labor between the two glands is surprisingly variable. At one of end of the spectrum is the dog, a species in which seminal vesicles are absent and the prostate must therefore carry out the functions that are divided between the two glands in other species. Most mammals, including humans, rats, and mice, occupy the other end of the spectrum, in which the seminal vesicles produce most of the seminal fluid, with the prostate playing a minor role. In species with both glands, physiologic cooperation between the two glands is also appreciable at the molecular level. For example, the major secretory protein product of the seminal vesicles is semenogelin, a 52-kD protein that serves as a substrate for proteolytic enzymes produced by the prostate, including prostate-specific antigen (PSA). Proteolysis of semenogelin yields a variety of peptide byproducts that are believed to serve reproductive and antimicrobial functions in humans (Curry and Atherton, 1990). In mice and rats, seminal vesicle and prostate products cooperate to coagulate the ejaculate into a firm copulatory plug in the vagina during mating. The plug serves as a temporary barrier to further mating by the female, potentially blocking impregnation by competing males.
Embryonic Development of the Seminal Vesicles
Seminal vesicles develop from the mesonephric (wolffian) ducts shortly before the onset of prostate development. Seminal vesicle development is strictly dependent on an intact AR signaling pathway, including the ligand testosterone (reviewed in Wilson et al, 1981). This requirement contrasts to development of the human prostate (see later), which, in addition to an intact AR pathway, requires conversion of testosterone into the more potent 5α-reduced androgen, dihydrotestosterone (DHT) (Andersson et al, 1991; Mahendroo and Russell, 1999).
Thick smooth muscle layers constitute the muscular stroma of the seminal vesicles, which surrounds a short columnar-to-cuboidal epithelium. The epithelium has distinct basal and luminal layers that are notable for unusually variable nuclear size and shape, a feature that is also found in prostate cancer (Epstein and Netto, 2007). Another notable feature of the epithelium of the seminal vesicles, the almost invariable appearance of gold-colored intracytoplasmic pigment, is usually absent in prostate cancer and helps to clarify any confusion between the two entities. Seminal vesical pigment is thought to derive from cellular byproducts of nonviable sperm ingested by the seminal vesical epithelium (spermatophagy).
The seminal vesicles are extremely resistant to disease. Given their proximity, shared functions, and similar endocrine requirements to the prostate, it is striking that diseases of the seminal vesicles in humans are vanishingly rare. In contrast, prostate disease, at least in Western cultures, is a nearly universal rite of passage into old age (see subsequent chapters on benign prostatic hyperplasia and prostate cancer). Accordingly, contrasting gene expression between seminal vesicles and prostate has been used as a strategy to discover the molecular basis of prostate cancer risk (Thompson et al, 2008).
The Prostate
Regional Differentiation of the Lower Urinary Tract
The prostate is a derivative of the primitive endoderm (gut tube). Regional differentiation of the primitive gut tube into foregut, midgut, and hindgut is followed by a swelling at the caudal end that creates the cloaca (see Chapter 111). The cloaca, a Latin term meaning “sewer,” receives output from both the urinary and intestinal tracts and represents the fully differentiated state in birds, reptiles, amphibians, marsupials, and monotrenes. In placental mammals, however, the cloaca is divided by the urorectal septum during embryogenesis to create separate urinary and digestive outlets. The ventral urinary compartment is called the primitive urogenital sinus, which further segments into the urinary bladder at its cranial end and the urethra at its caudal terminus.
Prostate Budding
In males, the prostate develops just caudal to the bladder neck via the proliferation of epithelial buds extending out from the urogenital sinus epithelium. Prostate buds invade at stereotyped locations that pattern the future development of distinct prostate lobes in the rodent and, potentially, zones in the human. These regions prepare for epithelial bud invasion by “mesenchymal condensation,” a process in which urogenital sinus mesenchymal cells (cells constituting loose connective tissue that will differentiate into stromal elements) become closely packed together (reviewed in Thomson, 2008). This condensation occurs in both males and females and is therefore androgen independent. In contrast, epithelial budding is strictly androgen-dependent and represents the first events in prostate development that are identifiable at the light microscopic level. Prostate budding requires intricate epithelial-mesenchymal interactions (Fig. 90–1). In humans, prostate budding occurs during the 10th week of gestation. In mice, prostate budding occurs on the 17th gestational day, 2 days before birth. Importantly, androgen exposure is not only necessary but also sufficient to drive prostatic differentiation and growth in the embryo. This fact, along with the ability to easily manipulate androgen levels in experimental animals, makes the prostate a particularly appealing subject for the study of epithelial cell fate determination (Cunha et al, 1987; Schaeffer et al, 2008). Prostate buds initially grow as solid epithelial cords that subsequently (postnatal days 1 to 14 in mice) branch and canalize (Sugimura et al, 1986) as part of a sophisticated branching morphogenesis program.
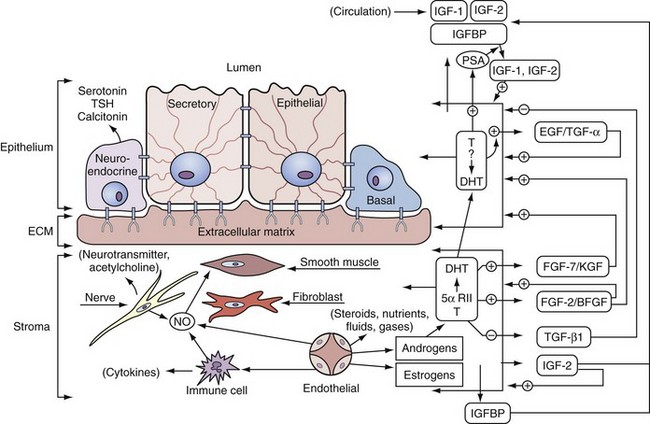
Figure 90–1 Stromal-epithelial interactions. Shown is a schematic of the types of stromal-epithelial interactions in information transfer and regulation within the prostate. Testosterone and growth factors interact on and between stromal and epithelial cells. The production of growth factors is either stimulated or inhibited by androgens. The growth factors can function on the same cell (autocrine) or on distant cells (paracrine). Nitric oxide (NO) is formed from nerve cells, endothelial cells, or macrophages and affects smooth muscle contraction (see text for details). Important features in this schematic are (1) three types of prostate epithelial cells—neuroendocrine, secretory, and basal; (2) five important prostatic stromal cells—smooth muscle, fibroblast, immune cells, endothelial cells, and nerve cells; (3) testosterone converted to dihydrotestosterone (DHT) by 5α-reductase in the stromal compartment; (4) three sources of NO production in the prostate—nerve, immune cells (e.g., macrophages), and endothelial cells; and (5) stromal-epithelial interactions mediated through various growth factors (see text). ECM, extracellular matrix.
Cytodifferentiation
In the mouse, the urogenital sinus epithelium begins as a homogeneous cell compartment that differentiates (after birth in mice) into distinct basal (adjacent to stroma) and luminal layers (Wang et al, 2001). Intervening epithelial cells, called “intermediate cells,” are present that have features of both basal and luminal cells. A fourth cell type, the neuroendocrine cell, is present in large numbers before prostate epithelial budding and decreases during embryonic development (Aumuller et al, 2001). The development of this cell type during mouse embryogenesis has not been well characterized, and the source of these cells has been variously proposed to be neural crest or urogenital sinus endoderm (Aumuller et al, 2001; Goldstein et al, 2008), illustrating the need to further delineate lineage commitment events in prostate epithelium.
Molecular Features of Prostate Development
Induction of Prostate Budding
Although AR signaling through DHT is the primary motivating force behind prostate development, it specifies only the timing of events not their location. The AR signaling machinery is present diffusely throughout the lower genitourinary tract (Takeda et al, 1985; Berman et al, 1995). Prostate epithelial buds form at precise locations through mechanisms that are not understood. This spatial control may involve paralogous homeobox (Hox) genes, which are transcriptional regulators that govern differential gene expression along the craniocaudal (head to tail) and proximodistal (e.g., shoulder to fingertip) axes in a variety of tissues, including the genitourinary tract (reviewed in Beck et al, 2000; Kmita and Duboule, 2003). In vertebrates, the paralogous Hox genes exist as four similar clusters (clusters A, B, C, and D), each of which resides on a separate chromosome and encodes genes whose chromosomal position from 3′ to 5′ mirrors their expression pattern in the embryo. The paralogous genes are distinct from other more distantly related transcription factor families that also contain DNA-binding homeobox motifs, such as the NK family whose members are expressed in a more discrete, organ-specific manner (e.g., see Nkx3.1, later). Paralagous Hox genes are sequentially numbered from 1 to 13, with the higher numbers in the 5′ position showing the most distal or caudal expression patterns. Accordingly, Hoxa13, Hoxb13, and Hoxd13 are paralogues on chromosomes 7, 17, and 2, respectively, that have overlapping expression patterns and functions in distal genitourinary tract development. Hoxb13 regulatory elements have been characterized that restrict its function to the caudal end of the genitourinary and digestive tracts and can be used to engineer androgen-independent prostatic expression of genes of interest (McMullin et al, 2009). Homozygous mutations in individual Hox genes result in subtle changes in prostatic branching patterns (Podlasek et al, 1997) and/or defective epithelial maturation (Economides and Capecchi, 2003). Mutations involving more than one of these genes results in significantly more severe urogenital phenotypes, such as significant prostate hypoplasia in Hoxd13/Hoxb13 compound mutant mice or failure of separate urinary and gastrointestinal tract outlets to form in Hoxa13/Hoxd13 compound mutants (Kondo et al, 1997; Warot et al, 1997). These multigene experiments are technically challenging and have not yet revealed the effects of complete loss of Hox gene function in the prostate.
Mesenchymal condensation occurs in both males and females, so it is not sufficient to drive prostate development but may be necessary. Condensation of the ventral mesenchymal pad is defective in mice lacking the gene for Noggin (see TGF-β family section later for further discussion of the roles of Noggin in prostate development), which antagonizes binding of bone morphogenetic protein (BMP) ligands to their receptors (Cook et al, 2007). This observation suggests that BMP signaling enhances mesenchymal condensation, either by direct action on mesenchyme or through regulation of epithelial-derived factors important in this process. Condensed mesenchyme is highly enriched for expression of fibroblast growth factors (FGFs) that are essential for epithelial bud outgrowth. For example, mice with engineered mutations of the mesenchyme-specific growth factor Fgf10 gene generate small abortive epithelial buds and fail to grow prostates (Donjacour et al, 2003).
Epithelial Budding
Epithelial expression of the NK homeobox transcription family member Nkx3.1 is the earliest indicator of prostate development at the molecular level. This transcription factor influences the degree of branching in the mature mouse prostate, where it can act as a tumor suppressor (Bieberich et al, 1996; Bhatia-Gaur et al, 1999; Abate-Shen et al, 2008). Nkx3.1 (Bhatia-Gaur et al, 1999), Sox9 (Lupien et al, 2008; Schaeffer et al, 2008; Thomsen et al, 2008), and sonic hedgehog (Podlasek et al, 1999) are expressed in emerging prostate buds, but prostate buds can emerge in the absence of these factors (see later).
Genes Required for Prostate Epithelial Budding
Mutations in the transcriptional regulator p63 (TP63) (Signoretti et al, 2000) or in the AR signaling axis (reviewed in Cunha et al, 1987) can completely abolish prostate induction. Noggin mutations selectively impair budding of the ventral lobes of the prostate, leaving anterior and dorsolateral budding unimpaired (Cook et al, 2007). Overall, however, the process appears to be a very robust one, with evidence of prostate epithelial bud formation persisting in the presence of a variety of genetic mutations that affect future steps in prostate ductal morphogenesis, particularly branching morphogenesis. TP63 has transcriptional repressor and activator activities that balance differentiation and stem/progenitor cell functions in epithelia (McKeon, 2004). The transcriptional targets of TP63 in prostate epithelial cells remain to be elucidated (Grisanzio and Signoretti, 2008).
Andromedins
One of the most striking aspects of the induction of prostate epithelial budding is the finding by Cunha and Lung (1978) that AR signaling is required in the mesenchyme but dispensable in the epithelium. Thus the action of androgens in this process appears to be indirect. This has led to the hypothesis that mesenchymal cells secrete inductive factors in response to androgens called “andromedins” (Yan et al, 1992). Andromedins have proven to be elusive quarry. Indeed, androgen-regulated mesenchymal factors have been difficult to identify much less characterize functionally (reviewed in Thomson, 2008), leading to alternative hypotheses involving androgen-mediated suppression of soluble factors (Tenniswood, 1986) or of smooth muscle cell barriers (Thomson et al, 2002) that inhibit induction of prostate epithelial budding. Studies to unravel the mechanism by which AR can indirectly induce prostate lineage commitment and growth are ongoing and are likely to yield important insights into the basic underpinnings of both physiologic and pathologic prostate growth.
Molecular Control of Branching Morphogenesis
Once set in motion, prostate growth and homeostasis continues to require androgens throughout life and this requirement appears to continue to be indirect, through mesenchymal or stromal AR signaling. Epithelial branching morphogenesis occurs through signaling cascades that inhibit further outgrowth along the long axis of an extending epithelial bud while stimulating lateral growth at its tip (Hogan, 1999). Through engineered deletion of genes in transgenic mice, several individual genes and components of classic morphogenetic pathways have been shown to be required for branching morphogenesis. Indeed, morphologic aberrations seen on interruption of a cellular pathway may be the most sensitive measure of a role for that pathway in regulation of prostate growth. As such, a wide variety of genes and pathways have been strongly implicated in prostate branching morphogenesis, only a few of which are covered here. For a more comprehensive perspective, including additional pathways such as those centered around Notch and Forkhead proteins, the reader is referred to recent reviews (Leong and Gao, 2008; Matusik et al, 2008).
The transcription factor Nkx3.1 (see earlier) helps determine the branching pattern of the prostate, as demonstrated by the reduced numbers of duct tips seen in mice with engineered Nkx3.1 deletion (Bhatia-Gaur et al, 1999). This relatively subtle phenotypic change may be important, however, as indicated by a dramatic decrease in the ability of Nkx3.1-mutant prostates to manufacture mature secretory proteins (Bhatia-Gaur et al, 1999). The regulatory machinery that promotes Nkx3.1 expression early in prostate induction has been used to drive expression of Cre recombinase to specifically inactivate other genes in prostate epithelial cells (Lin et al, 2007; Thomsen et al, 2008; Zhang et al, 2008).
Nkx3.1-Cre–mediated deletion was used to show that the sex-determining region Y–box 9 (Sox9) transcription factor is required for the development of the ventral lobe of the mouse prostate. Ventral lobe buds appeared at the normal embryonic age but failed to grow out and branch. The specificity of this proliferative defect for the ventral lobe, rather than other lobes, could be due to either a lobe-specific role for Sox9 or to a delay in Cre-mediated Sox9 excision found in the other prostate lobes. The latter possibility would be consistent with a transient requirement for the protein in ductal outgrowth (Thomsen et al, 2008).
Fibroblast Growth Factors
The fibroblast growth factor (FGF) family of related secreted peptides promote growth in recipient cells by binding to cell surface receptors and activating intracellular second messenger cascades. Epithelial branching morphogenesis, be it in the lung, salivary gland, mammary gland, or prostate, requires such signals to proceed. Of the FGFs, Fgf-7 (keratinocyte growth factor) and Fgf-10 have been studied most extensively in prostate development. Both of these ligands preferentially bind to Fgfr-2 over the three other family members (FGFRs 1, 3, and 4) (reviewed in Thomson, 2001, 2008). Ligand binding activates the intracellular microtubule-associated protein kinase (MAPK) pathway, leading to enhanced activity of growth-promoting transcription factors and increased proliferation.
Fgfr-2 is expressed on developing prostate epithelial cells where it can interact with its coreceptor Frs-2α. Fgf-7 and Fgf-10, in contrast, are secreted by prostate mesenchyme. This arrangement, along with androgen-independent growth of prostate organ cultures exposed to these ligands, have led to the proposal that they act as andromedins (Yan et al, 1992; Lu et al, 1999). However, Thomson and associates (2002) make a compelling argument to the contrary, pointing out that Fgf-7 and Fgf-10 expression patterns are equivalent in male and female rodent embryos and are therefore unlikely to mediate AR signaling from mesenchyme to epithelium. Instead, FGF signaling appears to have a critical role in epithelial outgrowth that is downstream of as yet undetermined prostate-inductive events. This role was demonstrated most dramatically by almost complete failure of prostate development in Fgf-10–deficient mice (Donjacour et al, 2003) and further supported by prostate hypoplasia and decreased epithelial branching in mice with prostate-targeted deletion of the genes encoding Fgfr-2 or Frs-2α.
The Hedgehog (Hh) Signaling Pathway
Across a variety of organs, elaboration of secreted hedgehog ligands (Sonic hedgehog, Indian hedgehog, and Desert hedgehog) by epithelial cells and reception in adjacent mesenchyme coordinates the activities of the Gli family proteins in regulating hedgehog pathway target genes. In the mesenchyme of developing prostate, several Hh target genes have been identified (Yu et al, 2009), including the cytokine Cxcl14, the insulin-like growth factor–binding protein Igfbp3, and the delta/notch-like epidermal growth factor–related receptor Dner. The roles of these particular genes in prostate development have yet to be ascertained, but, as a whole, Hh pathway target genes have been implicated in placement of prostate epithelial buds and in subsequent ductal branching and outgrowth. In particular, buds form in the absence of the dominant Hh ligand in the prostate (Berman et al, 2004) but are mislocalized in prostates of mice bearing mutations of downstream effectors of the pathway, Gli proteins (Doles et al, 2006). Later on in development, Hh ligands enhance epithelial outgrowth and branching (Freestone et al, 2003), which proceed abnormally in prostate organ cultures treated with Hh pathway antagonists (Lamm et al, 2002; Freestone et al, 2003; Berman et al, 2004). In adult animals the pathway may play a role in homeostasis, as indicated in a failure of prostates to regenerate after castration of animals treated with antibodies or small molecules that block Hh signaling (Berman et al, 2004). Taken together, these data indicate a growth-promoting role for the pathway in prostate epithelium, one that may have clinical relevance in pathologic prostate growth (reviewed in Shaw and Bushman, 2007).
The Transforming Growth Factor β (TGF-β) Superfamily
TGF-β superfamily members include TGF-β itself, as well as members of the growth and differentiation factors (GDFs) and the bone morphogenetic proteins (BMPs). These factors act through transmembrane receptors and the SMAD family of intracellular signal transducing proteins (Schmierer and Hill, 2007). Little is known about GDFs in the prostate, but both TGFs and BMPs are likely to play important roles. In organogenesis this superfamily is best known as a mesenchymal mediator of epithelial growth suppression, but (less frequently) they can also stimulate growth and/or be produced by epithelial cells. TGF-β1 inhibits net growth of the prostate but can stimulate growth in certain regions of the gland, particularly in the distal tips of the ventral prostate (Tomlinson et al, 2004a). Although the mechanism for growth promotion by TGF-β1 is unclear, the growth suppressive effect could quite reasonably relate to its ability to suppress levels of another mesenchymal growth factor FGF-10 (Tomlinson et al, 2004b) (see FGF section, earlier). A similar mechanism could remain in place in mature males, where TGF-β signaling in proximal ducts is believed to help to maintain prostate epithelial stem cells in a quiescent (growth-suppressed) state (Salm et al, 2005). BMP4 and BMP7 exert important and highly localized growth-suppressive activities in prostate development that help guide branching morphogenesis and prevent overproduction and disorganized epithelial growth. Like TGF-β, BMPs are most active during epithelial budding and subsequent prostate branching (Lamm et al, 2001; Tomlinson et al, 2004a; Grishina et al, 2005) (embryonic day 17 through postnatal day 5 in the mouse). Activation of BMP signaling suppresses branching morphogenesis, as indicated by experimental addition of exogenous BMP4 or BMP7 protein to prostate organ cultures (Lamm et al, 2002; Grishina et al, 2005) or by genetic deletion of the BMP inhibitor Noggin (Cook et al, 2007). BMP inactivation can show the opposite effect, as epithelial overgrowth of prostates with genetic deletion of BMP7 (Grishina et al, 2005).
Prostate Zonal and Lobar Anatomy
The rodent prostate is divided into paired anterior, dorsolateral, and ventral lobes. Each empties into to the urethra separately at its proximal extreme, with the distal end floating freely in the pelvic cavity. In contrast, the human prostate, like that of most primates and canine species, grows as a single organ encircling the urethra. In man and in dog, the latter arrangement is coupled with a propensity to grow in old age (benign prostatic hyperplasia), making males vulnerable to urinary obstruction (see Chapter 91). In rodents, the anterior, ventral, and dorsolateral lobes are named for the distinct locations of the urethra from which they originate. Each lobe also has a different branching pattern with a distinctive histologic appearance. These differences reviewed by Timms (2008) have been likened to different zones of the human prostate, histologically (Price, 1963), molecularly (Berquin et al, 2005; Thielen et al, 2007), and in terms of propensities to be affected by disease. In mice, mRNA transcripts for spermine-binding protein, probasin, and renin-1 are specific for ventral, dorsolateral, and anterior lobes, respectively (Cook et al, 2007), whereas human zone specific gene expression has not been as well characterized. Of particular interest, the mouse dorsolateral prostate shares molecular and histologic similarities to the peripheral zone of the human prostate, the zone most prone to the development of cancer.
Key Points: Embryonic Development
Prostate Cell Types
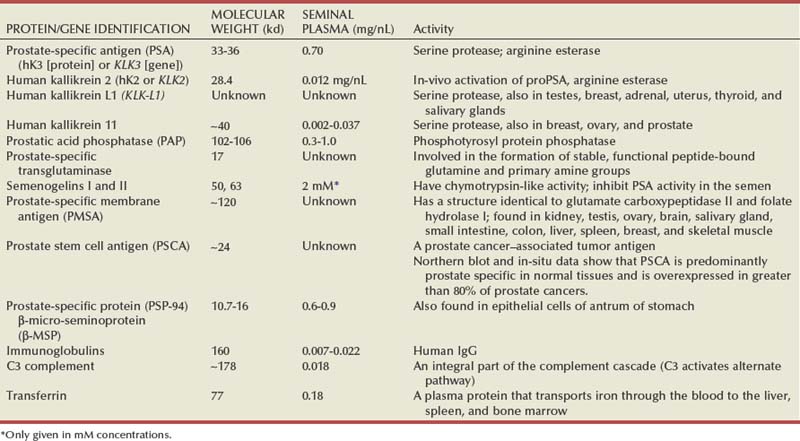
The prostatic epithelium in the human is composed of two major cellular compartments: epithelial cells and stromal cells (Table 90–1). The prostate epithelial compartment consists of basal epithelial cells, intermediate cells, neuroendocrine cells, and luminal secretory epithelial cells (reviewed by De Marzo et al, 1998a). The stromal compartment architecturally serves as structural support and consists predominantly of connective tissue, smooth muscle cells, and fibroblasts. Most prostate cell types have been characterized in vitro (Peehl, 2005).
Table 90–1 Summary of the Anatomy and Cell Biology of the Prostate Gland
| COMPONENTS | PROPERTIES |
|---|---|
| Development | |
| Seminal vesicles | From wolffian ducts through testosterone stimulation |
| Prostate | From urogenital sinus through dihydrotestosterone stimulation |
| Prostate Zones | |
| Anterior fibromuscular | 30% of prostate mass, no glandular elements, smooth muscle |
| Peripheral | Largest zone, 75% of prostate glandular elements, site of carcinomas |
| Central | 25% of prostate glandular elements; surrounds ejaculatory ducts |
| Transitional | Smallest zone, surrounds upper urethra complex, sphincter |
| 5% of prostate glandular elements, site of benign prostatic hyperplasia | |
| 15%-30% of prostate volume | |
| Epithelial Cells | |
| Basal | Small and flattened undifferentiated, nonsecretory cells with a low proliferative index (<1%) that express keratins 5, 14, and 18 |
| Intermediate | Proliferating cell type that has characteristics intermediate between basal and secretory cells, including production of basal and secretory cell keratins |
| Columnar secretory | Terminally differentiated, nondividing, rich in acid phosphatase and prostate-specific antigen; 20-µm tall, most abundant cell, keratins 5, and 18 |
| Epithelial Cells cont’d | |
| Neuroendocrine | Terminally differentiated, nonproliferating cells that express serotonin, chromogranin-A, neuron-specific enolase, and synaptophysin proteins |
| Stromal Cells | |
| Smooth muscle | Rich in α-actin, myosin, and desmin |
| Fibroblast | Vimentin rich and associated with fibronectin |
| Endothelial | Associated with fibronectin; alkaline phosphatase positive |
| Tissue Matrix | |
| Extracellular matrix | |
| Basement membrane | Type IV and V collagen meshwork that is laminin rich and supports basal cells, stem cells, transit-amplifying cells, and secretory epithelium |
| Connective tissue | Type I and type III fibrillar collagen; elastin |
| Glycosaminoglycans | Sulfates of dermatan, chondroitin, and heparin; hyaluronic acid |
| Cytomatrix | Tubulin, α-actin, and intermediate filaments of keratin |
| Nuclear matrix | Dynamic structure of the nucleus that directs the functional organization of DNA into loop domains; contains ribonuclear proteins |
In most glands with renewing cell populations there is a steady-state flow of cells from mostly quiescent stem cells to a more rapidly dividing pool of transient proliferating cells. This proliferating population finally reaches terminal differentiation, characterized by metabolically active secretory epithelium. In the prostate, cell lineage has not been rigorously determined but has been inferred from a variety of sources. A hypothetical differentiation scheme for prostate epithelium is presented in Figure 90–2. As in most multilayered epithelia, stem cells reside in the basal compartment and appear to give rise to all of the other epithelial cell types, as well as neuroendocrine cells. These include fully differentiated secretory cells that line glandular lumina (luminal cells), neuroendocrine cells that secrete bioactive peptides, and intermediate cells that show phenotypic features that are intermediate between basal cells and luminal cells.
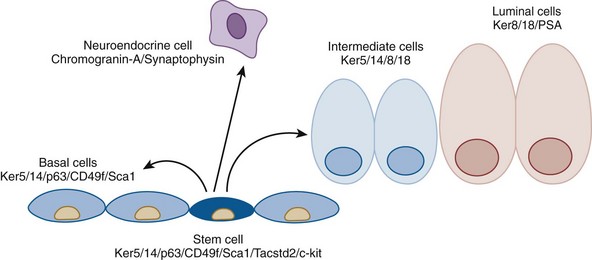
Figure 90–2 Hypothetical cell differentiation in adult prostate. Basal cells (medium blue) express basal cell proteins, including cytokeratin (CK) proteins 5, and 14, p63, CD49f, and Sca1. Stem cells within the basal cell compartment (dark blue) express basal cell proteins, as well as Tacstd2 and c-kit. Basal stem cells populate the basal cell compartment (medium blue) and, eventually, intermediate cells (light blue). Intermediate cells proliferate and differentiate into quiescent luminal cells (orange). Neuroendocrine cells (purple) are also believed to derive from epithelial stem cells. Formal lineage tracing has not been performed in all of these cell types, thus the true differentiation pathway remains to be determined.
(Modified from Wang Y, Hayward S, et al. Cell differentiation lineage in the prostate. Differentiation 2001;68[4-5]:270–9.)
Luminal Epithelial Cells
The luminal epithelial cell is the “workhorse” of the prostate gland, responsible for epithelial barrier integrity and production of prostatic secretion. Luminal cells constitute most of the prostate epithelium. These tall (10 to 20 µm) columnar secretory epithelial cells are terminally differentiated and have a low proliferative index (De Marzo et al, 1998a); they are easily distinguished by their morphologic features and abundant secretory granules and enzymes. Secretory cells produce a variety of proteins that characterize prostatic differentiation, including PSA, acid phosphatase, AR, leucine amino peptidase, and 15-lipoxygenase-2 (Shappell et al, 1999; Bhatia et al, 2003). They are also rich in keratin filaments (subtypes 8 and 18) (van Leenders and Schalken, 2003). These tall, columnar secretory cells appear in rows like a picket fence with each cell connected to the next by cell adhesion molecules; the apical aspect of these cells projects into the lumen, with the base attached to a basement membrane through integrin receptors (Knox et al, 1994). The nucleus is at the base just below a clear zone (2 to 8 µm) of abundant Golgi apparatus, and the upper cellular periphery is rich in secretory granules and enzymes. The apical plasma membrane facing the lumen possesses microvilli, and secretions move into the open collecting spaces of the acinus. These epithelial cells ring the periphery of the acinus and produce secretions into the acini that drain into ducts connected to the urethra.
Neuroendocrine Cells
Neuroendocrine cells are cells that release hormones in response to neural stimulation. In the prostate, neuroendocrine cells reside among the more abundant secretory epithelial cells in the normal prostate gland as well as in the urothelium of the prostatic urethra (Aumuller et al, 2001). There are two types of neuroendocrine cells: the first is open and possesses specialized microvilli that protrude into the lumen; the second is closed with long dendrite-like processes that extend to nearby epithelial cells and basal cells close to afferent and efferent nerves (diSant-Agnese and deMesy-Jensen, 1984; diSant-Agnese et al, 1985; Abrahamsson, 1999; Vashchenko and Abrahamsson, 2005).
Thinking on the origin of prostatic neuroendocrine cells has evolved. Aumuller and associates (2001) demonstrated that neuroendocrine cells are readily identified in male and female urogenital sinus epithelium before human prostate development, suggesting that these might represent a separate lineage that is independent of prostate epithelium. More recently, Goldstein and coworkers (2008) showed that neuroendocrine, basal, and secretory luminal cells can all originate from a common pluripotent Trop2-expressing prostate epithelial stem cell precursor.
Current evidence suggests that neuroendocrine cells can influence growth, differentiation, and secretory activity of the prostate epithelium through paracrine and autocrine mechanisms (Abrahamsson, 1999; Vashchenko and Abrahamsson, 2005). Neuroendocrine cells bring about their regulatory activity by the secretion of hormonal polypeptides or biogenic amines such as serotonin. High-pressure liquid chromatography measurements have shown that normal human prostate tissue contains approximately 1400 ng of serotonin per gram of tissue, and this would certainly emphasize the importance of these cells (Davis, 1987). Higgins and Gosling (1989) have studied the structure and intrinsic innervations of the normal human prostate and have observed acetylcholinesterase-containing nerves associated with smooth muscle in both the peripheral and the central parts of the prostate. In addition, they have shown that the majority of the acini in the peripheral and central regions possess a rich plexus of autonomic nerves and that vasoactive intestinal peptide-positive nerve fibers are found in relation to the epithelial lining acini in the central and peripheral regions of the gland. Lepor and Kuhar (1984) characterized and studied the location of the muscarinic cholinergic receptor in human prostate tissue and localized it to the epithelial cells. This is consistent with the neuropharmacology of muscarinic cholinergic agonist, which has a marked effect on increasing prostatic secretion. However, the α1-adrenergic receptor has its effect in the human prostatic stromal compartment. This is of clinical importance because of the use of selective α1-adrenergic antagonists to alleviate bladder outlet obstruction secondary to benign prostatic hyperplasia (Lepor, 1993). Recent work has demonstrated three subtypes of the α1-adrenergic receptor (α1A, α1B, and α1D). Of these, the α1A receptor appears to be linked to smooth muscle contraction of the prostate (Lepor et al, 1993).
Neuroendocrine cells are terminally differentiated (i.e., nonproliferating) and do not express detectable AR, PSA, or Bcl-2. These cells release peptide hormones or prohormones by fusion of intracellular granules with the cell membrane and exocytosis of their contents. In addition to serotonin, neuroendocrine cells produce numerous bioactive macromolecules (bombesin, neuron-specific enolase, calcitonin gene family members, thyroid-stimulating hormone–like peptide, somatostatin, synaptophysin, and parathyroid hormone–like peptide are major examples). Neuroendocrine factors appear likely to influence the growth, differentiation, and secretion of epithelium of the prostate in both normal and malignant conditions (Vashchenko and Abrahamsson, 2005).
Basal Cells
Basal cells (reviewed by De Marzo et al, 1998a) are the smallest of epithelial cells. They have a low mitotic index and are a minor population, accounting for less than 10% of the total cell number. Basal cells express a distinct keratin subtype profile (subtypes 5 and 14) compared with the columnar epithelial cells (subtypes 8 and 18). These cells are typically pyramid shaped with relatively little cytoplasm and condensed chromatin. Basal cells rest on the basement membrane wedged between the bases of adjacent, tall, columnar epithelial cells. The basal cell compartment has long been considered the likely source of the epithelial stem cells of the prostate because they are relatively undifferentiated with a low proliferative index (~1%) and almost devoid of secretory products, such as PSA and prostatic acid phosphatase (see Fig. 90–2).
Prostate Epithelial Stem Cells
Androgen deprivation does not affect stem cells but rather enriches them, because the more fully differentiated cells initiate apoptosis. Experiments in rodents have shown after castration that testosterone supplementation can restore prostate growth by stimulating stem cell proliferation. When mice are castrated after implantation with human prostate primary xenografts and then restimulated with testosterone, the basal cell population is highly overrepresented, consistent with the concept that the human basal compartment also contains prostate epithelial stem cells (Huss et al, 2004).
Recent experimental work in the mouse has provided powerful functional evidence for stem cell populations in the prostate, localizing them to the basal compartment, particularly in the proximal portions of prostatic ducts. These experiments used in vivo grafting assays to demonstrate critical stem cell characteristics including the ability of long-lived cells to proliferate indefinitely and to give rise to more differentiated phenotypes. Tsujimura and colleagues demonstrated DNA label-retaining prostate epithelial cells with long-term proliferative potential that were preferentially localized to the proximal segments of prostatic ducts of adult males (Tsujimura et al, 2002). Further studies have mapped the stem cell properties of proximal duct cells to those expressing the mouse stem cell antigen Sca1, the basal cell integrin α6 (Itga6 or CD49f), the tumor-associated calcium signal transducer Tacstd2 (also known as Trop2), and the stem cell factor receptor, c-kit (Burger et al, 2005; Xin et al, 2005; Lawson et al, 2007; Goldstein et al, 2008; Leong and Gao, 2008).
Intermediate Cells
Intermediate cells are so named because they possess phenotypic characteristics intermediate between basal and luminal cells. Similarities of these cells to prostate cancer cells have marked them as hypothetical substrates for neoplastic transformation (Verhagen et al, 1992; De Marzo et al, 1998b), although their susceptibility to carcinogenesis is unknown. These investigators proposed that intermediate cells fulfill a transient amplifying function, providing a short-term amplification function for the long-term proliferative capabilities of basal stem cells. Intermediate cells produce basal cell keratins (5 and 14) and the secretory cell keratins 8 and 18 (De Marzo et al, 1998b; Schalken and van Leenders, 2003). Uzgare and colleagues (2004) reported transient amplifying properties of human intermediate cells in culture: a high proliferative fraction with the ability to proliferate for a limited number of generations. Survival of terminally differentiated secretory luminal cells and proliferation of intermediate cells require androgens potentially acting indirectly through the secretion of androgen-regulated growth factors by the stromal compartment (andromedins) (Uzgare et al, 2004).
Key Points: Prostate Epithelial Cell Types
The Stroma and Tissue Matrix
The noncellular stroma and connective tissue of the prostate make up what is termed the ground substance and the extracellular matrix in what was first suggested by Arcadi (1954) to play an important role in prostate function and disease. The extracellular matrix has long been recognized as one of the important inductive components during normal development of many different types of cells (Cunha, 1976; Hay, 1981; Bissell et al, 1982; Getzenberg et al, 1990; Risbridger et al, 2005). Classic tissue recombination experiments by Cunha and colleagues (1987) have clearly shown the direct importance of the isolated embryonic mesenchyme to the induction of differentiation of normal prostatic epithelial cells (see earlier discussion).
The epithelial cells rest on the basement lamina or membrane, which is a complex structure containing, in part, collagen types IV and V, glycosaminoglycans, complex polysaccharides, and glycolipids. This layer forms an interface to the stromal compartment that provides structural support for the basal cells and their progeny. It consists of an extracellular matrix, ground substance, and a variety of stromal cells, including the fibroblasts, capillary and lymphatic endothelial cells, smooth muscle cells, neuroendocrine cells, and axons (reviewed in Taylor and Risbridger, 2008).
The cytomatrix (cytoplasmic skeleton) terminates in the center of the cell by direct attachment to the nuclear matrix (Fig. 90–3). The prostatic epithelial cell therefore has direct structural linkage via the matrix system from the DNA to the plasma membrane. The cytomatrix then makes direct contact with the basement membrane, extracellular matrix, and ground substance of the stroma. This entire interlocking tissue scaffolding or superstructure is termed the tissue matrix and may have dynamic properties in ordering and controlling biologic processes as well as in the transport of secretions from the sex accessory tissues (Getzenberg et al, 1990; Konety and Getzenberg, 1999; Etienne-Manneville, 2004; Miner and Yurchenco, 2004; Hallmann et al, 2005).
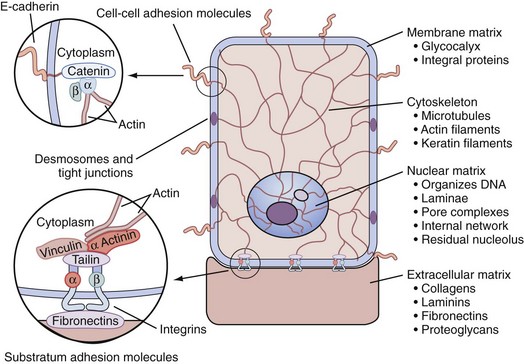
Figure 90–3 The tissue matrix system, a superstructure scaffold network that connects the extracellular matrix components to the cell through interactions with cell adhesion molecules (e.g., integrins). Cell adhesion molecules extend through the plasma membrane and connect directly or indirectly to the cytomatrix structures. The cytomatrix directly couples to the nuclear matrix, which attaches and organizes the cellular DNA. Cell adhesion molecules and desmosomes connect neighboring cells.
(From Getzenberg RH, Pienta KJ, Coffey DS. The tissue matrix: cell dynamics and hormone action. Endocr Rev 1990;11:399–416.)
Understanding the biologic components of the tissue matrix system within sex accessory tissues is of paramount importance to understanding its physiology. The laminin proteins are glycoproteins of the extracellular matrix that mediate attachment of cells to the type IV collagen of the basement membrane (Miner and Yurchenco, 2004; Yurchenco et al, 2004; Hallmann et al, 2005). Laminin is produced by epithelial cells but not by fibroblasts; it is a large molecule (approximately 800 kD) with molecular domains that interact with the type IV collagen of the basement membrane and with integrin-type receptors within the cell surface glycocalyx of the epithelial cell (Aumailley et al, 2005). Laminins are the major anchor filaments in the basement membranes of epithelial cells that stabilize attachment of hemidesmosomes via α6β4 integrin (Brar et al, 2003; Miner and Yurchenco, 2004). The key functional properties of the laminins are cell adhesion, proliferation, differentiation, growth, and migration. Laminin surrounds the basement membrane of prostate acinar epithelial cells, capillaries, smooth muscle, and nerve fibers but not lymphatics, lymphocytes, or fibroblasts; the laminin structure and its distribution are disrupted in BPH and higher-grade prostatic interepithelial neoplasia and higher-grade prostate neoplasms (Sinha et al, 1989; Brar et al, 2003; Miner and Yurchenco, 2004).
In summary, the development and maintenance of the prostate occurs through androgen-dependent and highly regulated tissue morphogenesis in processes involving epithelial cell differentiation, proliferation, and apoptosis (Cunha et al, 2004). Communication through numerous extracellular interactions is directed to the intracellular cytoskeleton and then to the nuclear matrix, which ultimately regulates a variety of transcriptional cell functions that control such critical phenotypic qualities as cell size and shape, cell motility, epithelial cell turnover, proliferation, and differentiation (Getzenberg et al, 1990; Pienta, 1993; Miner and Yurchenco, 2004).
Key Points: Laminin Proteins
Endocrine Control of Prostate Growth
The prostate, like other sex accessory tissues, is stimulated in its growth, maintenance, and secretory function by the continued presence of certain hormones and growth factors. Foremost among these is testosterone, which is converted within the prostate into the more active androgen DHT. Testosterone is synthesized in the Leydig cells of the testes from pregnenolone by a series of reversible reactions. However, once testosterone is converted by 5α-reductase into DHT or converted by aromatase into estrogens the process is irreversible: testosterone can be converted to DHT or estrogens, but estrogens and DHT cannot be converted to testosterone. Androgens, estrogens, and adrenal steroids are believed to have strong effects on different cells and tissues in the body that can vary with development and age. This varies from embryonic development to puberty and on into adult maintenance and aging. Therefore, androgen ablation or androgen treatments have a wide variety of physiologic effects that merit consideration.
The generalized endocrine physiology of the prostate is depicted in Figure 90–4. The hypothalamus releases a small 10-residue polypeptide (decapeptide) referred to as luteinizing hormone–releasing hormone (LHRH), also called gonadotropin-releasing hormone (GnRH). Under the stimulation of LHRH the pituitary releases luteinizing hormone that is transported to the testes and acts directly on the Leydig cells to stimulate de novo steroid synthesis and release of testosterone, the major serum androgen of the body. Most of the estrogen in the male is derived from peripheral conversion of androgens to estrogens through aromatization. Exogenous estrogens, such as diethylstilbestrol, block androgen action not primarily by direct effects on the prostate but indirectly through blocking pituitary function. The estrogen causes a negative feedback on luteinizing hormone release that reduces the serum signal for testicular testosterone production; therefore, estrogen acts as an effective “chemical castration.”
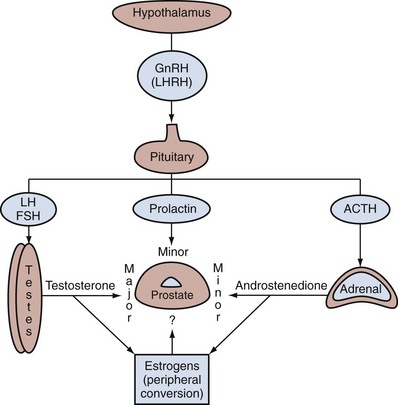
Figure 90–4 Simplified endocrinology of the prostate. Luteinizing hormone–releasing hormone (LHRH), also known as gonadotropin-releasing hormone (GnRH), stimulates the pituitary to release the gonadotropins luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which stimulate the Leydig cells of the testes to synthesize testosterone. Testosterone is the major serum androgen stimulating prostate growth. Peripheral conversion of testosterone by aromatization forms the estrogens in the male. The adrenal gland is under stimulation by adrenocorticotropic hormone (ACTH) and releases the minor androgens, such as androstenedione, which is also converted peripherally to estrogens. Prolactin has also been shown to have a minor effect in stimulating androgen-induced prostate growth. The prostate can produce its own growth factors (autocrine or paracrine) or respond to circulating growth factors.
In addition to testosterone, the adrenal secretes a weak androgen, androstenedione; however, this is not a major influence on prostate physiology because castration leads to almost complete involution of the prostate, meaning that insufficient adrenal androgens are present to stimulate any meaningful growth of the normal prostate. Similar to serum testosterone, androstenedione can undergo aromatization to estrone. Overproduction of androstenedione, such as occurs in certain forms of congenital adrenal hyperplasia, may stimulate prostate growth; however, again, the role of adrenal androgens in regulating prostate growth is minor.
Androgen Production by the Testes
Because the testes produce the major serum androgen supporting prostate and sex accessory tissue growth, it is important to briefly review this function. In the normal human male the major circulating serum androgen is testosterone, which is almost exclusively (~95%) of testicular origin. Under normal physiologic conditions the Leydig cells of the testis are the major source of the testicular androgens. The Leydig cells are stimulated by the gonadotropins (primarily the luteinizing hormone) to synthesize testosterone from acetate and cholesterol. The spermatic vein concentration of testosterone is 40 to 50 µg/dL, approximately 75 times more concentrated than the level detected in the peripheral venous serum (Hammond, 1978), which is approximately 600 ng/dL. Other androgens also leave the testes by the spermatic vein, and these include androstanediol, androstenedione (3 µg/dL), dehydroepiandrosterone (7 µg/dL), and DHT (0.4 µg/dL). The concentrations of these androgens are much lower in the spermatic vein than the concentration of testosterone, with all being less than 15% of the concentration of testosterone.
The total testosterone that enters the plasma is referred to as the testosterone blood production rate and is 6 to 7 mg/day in the human. Although other steroids, such as androstenedione from the adrenals, can be converted by peripheral metabolism to testosterone they probably account for less than 5% of the overall production of plasma testosterone. The plasma half-life of testosterone is only 10 to 20 minutes, which means that a man undergoing bilateral simple orchiectomy is functionally castrated within 1 to 2 hours of surgery.
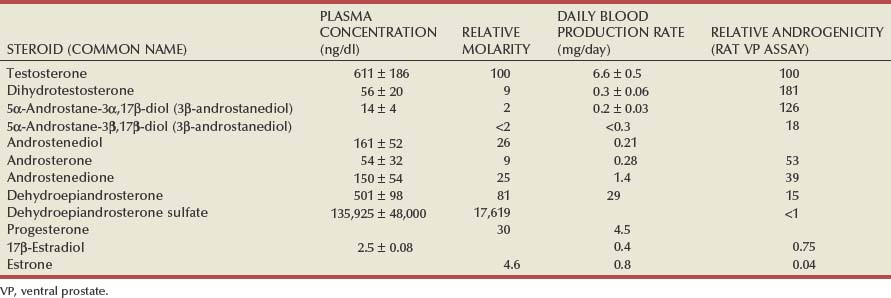
The average testosterone concentration in the adult human male plasma is approximately 611 ng/dL ± 186 with a normal range of 300 to 1000 that is equal to 10.4 to 34.7 nmol/L in SI units (Table 90–2). Serum testosterone level is not remarkably related to age between 25 and 70 years, although it does decline gradually to approximately 500 ng/dL after 70 years of age. It is recognized that plasma concentrations of testosterone can vary widely in an individual on any one day and may reflect both episodic and diurnal variations in the production rate.
Metabolic androgens such as 17-ketosteroids are then secreted into the urine as water-soluble glucuronide or sulfate conjugates. The total 17-ketosteroid level in the urine in adult men is 4 to 25 mg/24 hr and is not an accurate index of testosterone production, because other steroids from the adrenals as well as nonandrogenic steroids can be metabolized to 17-ketosteroids. Only small (25 to 160 µg/day) amounts of testosterone enter the urine without metabolism, and this urinary testosterone represents less than 2% of the daily testosterone production.
Although testosterone is the primary plasma androgen inducing growth of the prostate gland and other sex accessory tissues, it appears to function as a prohormone in that the most active form of the androgen in the prostate is not testosterone but rather DHT (Farnsworth and Brown, 1963; Anderson and Liao, 1968; Bruchovsky and Wilson, 1968) (Fig. 90–5). The formation of DHT involves the reduction of the double bond in the A ring of testosterone through the enzymatic action of the enzyme 5α-reductase (Fig. 90–6). There are at least two isoforms of this enzyme (type 1 and type 2). Type 2 5α-reductase expression predominates in human accessory sex tissues and is localized to the fibromuscular stromal compartment (Silver et al, 1994). The type 1 isoform predominates in skin, in prostatic epithelia, and to a lesser extent in prostatic fibromuscular stroma. Inhibition of 5α-reductase by finasteride appears to be largely selective for the type 2 isoform (Iehle et al, 1995; Habib et al, 1997); the newer agent dutasteride inhibits both type 1 and type 2 5α-reductase. Both drugs appear to exert similar effects in terms of reduction in prostatic volume and serum PSA concentration, suggesting that the type 2 isoform is the only clinically significant isoform present in the prostate. DHT concentration in the plasma of normal men is low, 56 ± 20 ng/dL, in comparison to testosterone, which is 11-fold higher at approximately 611 ng/dL (see Table 90–2). In summary, although DHT is a potent androgen (2 to 10 times as potent as testosterone in many bioassay systems), its low plasma concentration and tight binding to plasma proteins diminishes its direct importance as a circulating androgen affecting prostate and seminal vesicle growth. In contrast, DHT is of paramount importance within the prostate, where it is formed from testosterone. DHT is the major form of androgen found within the prostate gland (5 ng/g tissue wet weight) and is fivefold higher than testosterone. In the prostate, DHT binds to ARs and activates the receptors to regulate a variety of cellular processes. In summary, DHT becomes the major androgen regulating the cellular events of growth, differentiation, and function in the prostate.
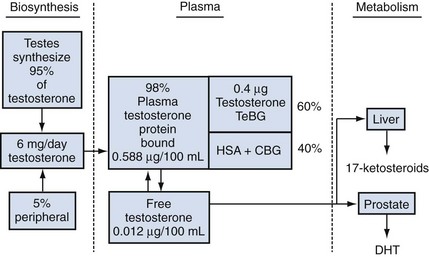
Figure 90–5 Quantitative assessment of the testicular biosynthesis, plasma transport, and metabolism of testosterone. Plasma testosterone is bound to testosterone-binding globulin (TeBG), human serum albumin (HSA), and cortisol-binding globulin (CBG). All numbers are average values for the normal adult male. DHT, dihydrotestosterone.
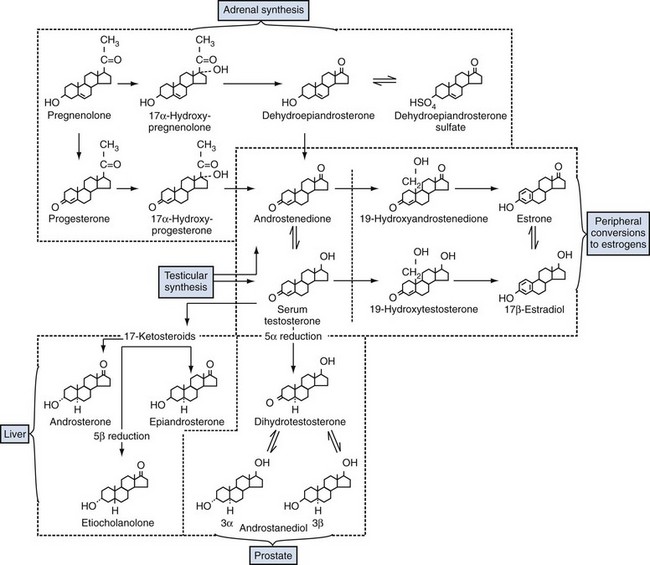
Figure 90–6 Overview of the synthesis and metabolism of testosterone in four main body compartments: adrenal synthesis of androstenedione; peripheral conversion of androgens (androstenedione and testosterone) to estrogens; formation of active androgen (DHT) within the prostate; and inactivation in the liver of testosterone to three types of 17-ketosteroids.
The normal adult male plasma levels of some important steroids are summarized in Table 90–2. These values are derived as averages from numerous studies. Individual values can fluctuate with age, time of day, medications, stress, hospitalization, and environmental changes.
Adrenal Androgens
There is evidence that overproduction of adrenal steroids can stimulate growth of the prostate gland. For example, in humans, abnormal virilism has been observed in immature males with a hyperfunctioning adrenal cortex. In rodents, overstimulation of the adrenals can also induce limited prostate growth even in the absence of testicular androgens. For example, administration of exogenous adrenocorticotropic hormone to castrated animals does significantly increase the growth of sex accessory glands (Tullner, 1963; Tisell, 1970; Walsh and Gittes, 1970). However, the effect of normal levels of adrenal androgens on the prostate in noncastrated humans and adult male rats does not appear to be significant because adrenalectomy has little effect on prostate size, DNA, or morphologic characteristics of the sex accessory tissue (Mobbs et al, 1973; Oesterling et al, 1986). Furthermore, after castration in animals, with the adrenals intact, the prostate will finally diminish to a very small size (90% reduction in total cell mass). Finally, the small involuted ventral prostate in the castrated rat cannot be significantly reduced further by additional adrenalectomy or hypophysectomy (Kyprianou, 1987). In castrated rats, the DHT level in the prostatic tissue is approximately 20% of that in normal intact animals. Adrenalectomy lowers the DHT to nondetectable levels without further diminution in prostate growth. This indicates that a threshold level of DHT is required in the prostate to stimulate growth and the castrate level is below this threshold. It has also been concluded similarly that the prostate of man does not restore itself after castration, indicating that adrenal androgens are insufficient to compensate for the loss of testicular function. Quantitative morphometry of the human prostate (Oesterling et al, 1986) also confirms that the adrenal gland has little effect on the epithelial cell size of the normal prostate.
The adrenal steroids dehydroepiandrosterone (DHEA) and the conjugate dehydroepiandrosterone sulfate (DHEAS) as well as androstenedione are androgens synthesized from acetate and cholesterol (see Fig. 90–6) that are secreted by the normal human adrenal glands. Essentially all of the DHEA in the male plasma is of adrenal cortex origin, and the production rate in man is 10 to 30 mg/day. Less than 1% of the total testosterone in the plasma is derived from DHEA (Horton, 1976; MacDonald, 1976). The prostate and seminal vesicles of the rat and the human prostate can slowly hydrolyze DHEAS to free steroids through a prostate sulfatase enzymatic activity, but the degree of conversion is low; hence, DHEAS is not a potent androgen.
A second adrenal androgen is androstenedione, and the plasma concentration in adult men is approximately 150 ± 54 ng/dL (see Table 90–2). The blood production rate of androstenedione in human males is 2 to 6 mg/day, with approximately 20% of the androstenedione being generated by peripheral metabolism of other steroids. Androstenedione cannot be converted directly to DHT. An important role for androstenedione in the male may be its peripheral conversion to estrogens through the aromatase reaction (see Fig. 90–6).
The adrenal gland also produces C21 steroids (e.g., progesterone). The plasma production rate at 0.75 mg/day is low, producing a low plasma progesterone concentration of 30 ng/dL. Although progesterone is weakly androgenic it does not exert a significant effect on the prostate at the low concentrations present in normal male plasma. In summary, under normal conditions, the adrenals do not support significant growth of prostatic tissue.
Estrogens in the Male
The estrogen receptors (ERs) are differentially expressed in the prostate. In the mouse, ER-α is expressed early (1 week) in the stroma of the ventral prostate but by 2 weeks is preferentially expressed in the epithelia, and ER-α is absent altogether in the ventral prostate by 4 weeks. In contrast, ER-β exists in the epithelial compartment as the dominant ER by the fourth week. Interestingly, however, knockout mice for ER (both isoforms) are able to form grossly normal prostates, although fertility may be limited in the ER-α (Couse et al, 2001). Only small amounts of estrogen are produced directly by the testes. In the plasma of young healthy human males 75% to 90% of the estrogens are derived from the peripheral conversion of androstenedione and testosterone to estrone and estradiol through the aromatase reaction (see Fig. 90–6) (Horton, 1976; MacDonald, 1976). The androgenic C19 steroids (testosterone and androstenedione) are converted to the estrogenic C18 steroids first by removal of the 19-methyl group and followed by the formation of an aromatic or phenolic steroid A ring (aromatase reaction), present in both estradiol and estrone. Estradiol is formed from testosterone and estrone from androstenedione; these two estrogens are interconvertible. The daily production of estradiol in the human male is 40 to 50 µg, and only 5 to 10 µg (10% to 25%) can be accounted for by direct testicular secretion (see Table 90–2).
Androgen-Binding Proteins in the Plasma
Less than 2% of the total testosterone in human plasma is free or unbound; the remaining 98% is bound to several different types of plasma proteins (see Fig. 90–5). The plasma proteins that bind steroids include human serum albumin, sex hormone–binding globulin (SHBG or testosterone-estrogen–binding globulin [TeBG]), corticosteroid-binding globulin (also named transcortin), progesterone-binding globulin, and, to a lesser extent, α1-acid glycoprotein. Under normal conditions, the total amount of testosterone bound to progesterone-binding globulin and α1-acid glycoprotein is nominal and is usually ignored.
The regulation of the amount of androgen that is free is an important physiologic variable and varies in different species. The total amount of steroid bound depends on two factors: (1) the affinity of the steroid to bind to a specific protein and (2) the capacity, which is the maximal potential binding when all of a binding protein is saturated with bound steroid; the capacity is governed by the amount of binding protein in the plasma. Serum albumin has a relatively low affinity for testosterone, but, given its abundance, it has a high capacity. In contrast, SHBG has a high affinity for binding steroids but the protein is present in relatively low concentrations; however, the plasma molarity of each binding protein exceeds the plasma molarity for total testosterone concentration. The majority of testosterone bound to plasma protein is associated with SHBG. For example, Vermeulen (1973) has calculated that in the normal human male 57% of testosterone in the plasma is bound to SHBG and 40% is bound to human serum albumin. Less than 1% is bound to corticosteroid-binding globulin, and only 2% of the total testosterone is free (see Fig. 90–5). The normal plasma free testosterone level is therefore 12.1 ± 3.7 ng/dL or 0.42 nM; this non–protein-bound “free testosterone” is bioavailable to diffuse into the sex accessory tissue and into liver cells for metabolism. In addition, a large percentage of the SHBG is saturated whereas only a small fraction of the total capacity of corticosteroid-binding globulin and albumin is used under normal conditions. As testosterone levels increase in the plasma, the order of increasing saturation of the plasma proteins proceeds from SHBG to corticosteroid-binding globulin to albumin. Therefore, the binding of androgen is a dynamic equilibrium between various serum proteins.
The total plasma levels of SHBG can be altered by hormone therapy. Administration of testosterone decreases SHBG levels in the plasma, whereas estrogen therapy stimulates SHBG levels (Forest et al, 1968; Vermeulen, 1969; Burton and Westphal, 1972). Estrogen also competes with testosterone for binding to SHBG, but estrogen has only one third the binding affinity of testosterone. Therefore, administration of small amounts of estrogen increases the total concentration of SHBG, and this effectively increases the binding of testosterone and thus lowers the free testosterone plasma concentration.
Because only free testosterone is bioavailable, the binding of testosterone to plasma proteins inhibits net testosterone uptake into the prostate (Lasnitzki and Franklin, 1972). It is apparent that androgenic activity is regulated in part by the extent of binding of an androgen to the steroid-binding proteins in the plasma.
Regulation of Prostate Growth by Steroids and Protein Growth Factors
There are multiple levels of prostate growth regulation that include steroid hormone action, growth factors, and direct cell-cell communication and interactions with the extracellular matrix. These interactive types of growth control are accomplished by several generalized systems, as depicted in the schematic in Figure 90–7. They include the following:
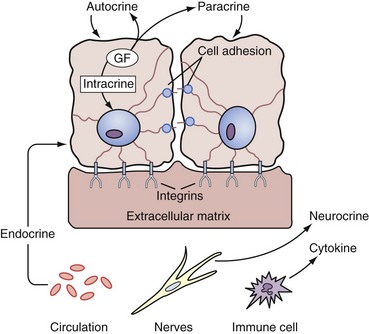
Figure 90–7 Types of growth control. Endocrine signals are carried through the circulation from distant organs. Paracrine signals are produced in proximity by neighboring cells. Autocrine signals feedback on the same cells from which they are produced. Intracrine signals are a special subset of autocrine signals that never leave the cell but rather act locally within that cell. Cytokines are paracrine-like factors (typically) that are made by immune cells. Neurocrine factors are released by nerves. Cell adhesion molecules directly link neighboring cells, often through association with cognate adhesion molecules. Cells are also bound to the extracellular matrix through interactions with other cell adhesion molecules (e.g., integrins). GF, growth factor.
Of these seven growth control systems, the first extensively studied on the prostate was the endocrine effect of androgenic steroids, such as testosterone, in the regulation of prostate growth through changes in serum testosterone levels and conversion to DHT. However, androgens alone are not sufficient for full prostate growth. In the past 2 decades, extensive progress has been made in the understanding of the other systems, particularly the interactive role of growth factors and their receptors. At present, the roles of these receptors in cell signaling to the nucleus and of the structural elements in cellular control involving the tissue matrix are being developed. These mechanisms are reviewed next, starting with androgen action at the cell level beginning with the arrival of testosterone in the serum.
Androgen Action at the Cellular Level
Testosterone in the serum arrives at the prostate bound to albumin and to the steroid-binding globulins. Free testosterone enters the prostate cell by diffusion, where it is then subjected to a variety of steroid metabolic steps that appear to regulate the activity of the steroid hormone and its downstream effectors. A simplified schematic of the temporal sequence of intracellular events is depicted in Figure 90–8 and includes the following:
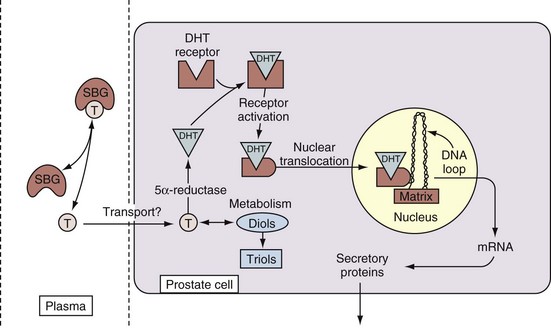
Figure 90–8 Simplified schematic of the effects of testosterone in activating transcriptional targets in an epithelial cell. In the plasma, testosterone (T) is bound to serum-binding globulins (SBG), such as testosterone-binding globulin and albumin. Unbound testosterone is transported by passive diffusion into the prostate, where it is enzymatically converted to dihydrotestosterone (DHT) by 5α-reductase (type 2) and further metabolized to diols (3α or 3β) and irreversibly metabolized into the more water-soluble triols (6α or 7α). DHT binds to a cytoplasmic receptor (androgen receptor) that is activated and translocated to the nucleus. There the androgen receptor localizes in matrix acceptor sites and subsequently activates or represses certain target genes by regulating production of their mRNA. The RNA is then transported to the cytoplasm, where it is translated into a variety of proteins (e.g., secretory proteins such as PSA).
The epithelial cell is the primary unit in secretion, but specific genes are also activated in the stromal cells, and these events are also regulated by testosterone, estrogens, and growth factors in a similar chain of events. However, not all cells respond the same to androgens or estrogens. For simplicity, these steps are discussed in relation to the epithelial cells. Androgens and estrogens, both together and separately, can affect prostate cells through interaction with receptors, and it appears that estrogens might have their primary effect on the stromal cells.
5α-Reductase and Androgen Metabolism within the Prostate
After the free testosterone in the plasma has entered the prostatic cells through diffusion it is rapidly metabolized to other steroids by a series of prostatic enzymes (Isaacs et al, 1981, 1983; Isaacs and Coffey, 1981; Bruchovsky and Dunstan-Adams, 1985). More than 90% of testosterone is irreversibly converted to the main prostatic androgen DHT (Fig. 90–9) through the action of the reduced form of nicotinamide-adenine dinucleotide phosphate (NADP) and the enzyme 5α-reductase located on the endoplasmic reticulum and on the nuclear membrane. The enzyme 5α-reductase reduces the unsaturated bond in testosterone between the 4 and 5 positions to form the 5α-reduced product DHT. The Km for testosterone is 8.3 nM, and the serum level of testosterone is only in the range of 0.5 to 3.0 nM, indicating that the enzyme cannot be saturated because the testosterone substrate would be less than the Km value. Bruchovsky and Dunstan-Adams (1985) reported a 10-fold increase in the maximal velocity in the stromal tissue compared with the epithelium. They observed 262 pmol of DHT formed in 30 minutes per milligram of protein from testosterone measured in the stroma and less than 10% of that amount with a maximal velocity of 19 for the epithelium. The stromal Km was 76 nM and the epithelial Km was 13 nM. These differences between stromal and epithelial kinetics were used to deduce the existence of two different isoenzymes of the 5α-reductase (Andersson et al, 1991).
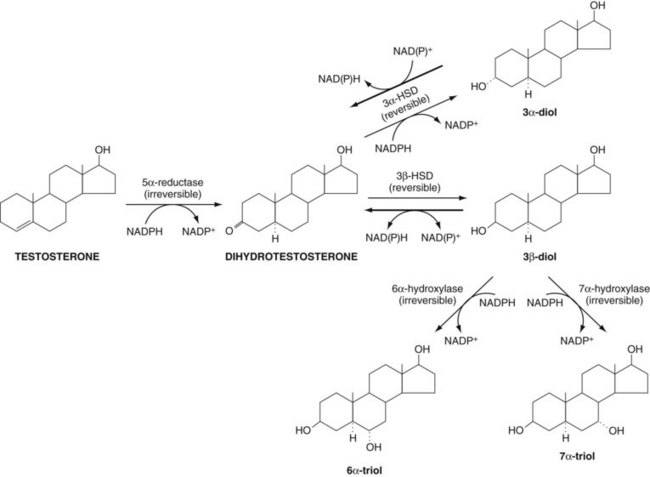
Figure 90–9 Metabolic pathways for testosterone within the prostate. Testosterone is irreversibly metabolized by 5α-reductase to dihydrotestosterone (DHT), which is then reversibly converted into 3α-diol and 3β-diol. The 3β-diol is irreversibly inactivated to the more soluble 6α-triol and 7α-triol. 3α-HSD, 3α-hydroxysteroid dehydrogenase.
In the human, rat, and monkey there are two isozymes of 5α-reductase (Table 90–3). The human and rat 5α-reductase isozymes are composed of 254 to 260 amino acids with a molecular weight of 28 to 29 kD. These enzymes are N- and O-glycosylated and have a high percentage of hydrophobic amino acids that are distributed throughout the enzyme. The chromosomal localization of the human 5α-reductase isozyme genes has been reported; the type 1 enzyme is at the extreme tip of the short arm of chromosome 5, and the human type 2 gene is on the short arm of chromosome 2. There is a 49% homology between type 1 and 2 enzymes in the human. The properties of these enzymes have been reviewed in detail by Russell and Wilson (1994) and for the effects on prostate growth by McConnell (1995). The effect of finasteride on 5α-reductase activity has been reviewed by Rittmaster (1994). The type 1 enzyme is in the skin and in the adult scalp and is believed to be involved in hair formation. It is present to lesser degrees in the prostate epithelium and stroma. This isoform is found in normal levels in men with congenital 5α-reductase deficiency. The type 2 enzyme is mutated in 5α-reductase deficiency and is the dominant isoform present in the prostate gland. The type 2 enzyme appears in the basal cells of the epithelium and in the stromal cells but is absent in the secretory epithelial cells. This has raised the possibility that DHT stimulation of epithelial cells is derived from DHT converted within the stromal or basal cells. Silver and coworkers (1994) have studied the cell type–specific expression of these reductases as well as their regulation. It appears that the 5α-reductase type 2 in the prostate does not change dramatically in individuals undergoing short-term androgen ablation.
Table 90–3 Properties and Distribution of 5α-Reductase Types 1 and 2
| TYPE 1 | TYPE 2 | |
|---|---|---|
| Chromosome | 5p15 | 2p23 |
| Molecular weight | 29,000 | 28,000 |
| Amino acids | 259 | 254 |
| Exons | 4 | 4 |
| Introns | 5 | 5 |
| Homology | 49% | 49% |
| pH optima | Alkaline (6-8.5) | Acidic (5.0) |
| Km testosterone (µM) | 1.5 | 0.1-1.0 |
| Ki finasteride (nM) | 325 | 12 |
| Half-life (hr) | 20-30 | 20-30 |
| 5α-Reductase deficiency | Normal | Mutated |
| Prostate Cells | ||
| HUMAN | ||
| Luminal epithelial | ± | − |
| Basal epithelial | − | + |
| Stromal | ± | + |
| Skin | + | − |
| RAT | ||
| Prostate cells | ||
| Luminal epithelial | − | − |
| Basal epithelial | + | − |
| Stromal | − | + |
Berman and associates (1995) have studied the distribution of the two 5α-reductase isozymes in the urogenital tract of the fetal rat. At 17 to 21 days of development the expression of type 1 gene predominated in the epithelial cells; the type 2 gene was limited to the mesenchymal cells. This is true in both the testosterone-dependent and DHT-dependent anlagen of the urogenital tract. These investigators observed that androgens could stimulate the expression of the type 2 gene in the urogenital tract but not of the type 1 gene. They suggested that the type 2 5α-reductase gene exhibits positive feedback control in that the product of the enzyme, DHT, can stimulate expression of the gene; however, no evidence for such regulation of either 5α-reductase gene was detected in the fetus.
In summary, 5α-reductase is of great importance because the product DHT is important in the differentiation of the prostate during fetal development and because mutations in 5α-reductase give rise to a rare form of pseudohermaphroditism. In prostate physiology, expression of the 5α-reductase gene is regulated by androgens in both the prostate and liver. It is also believed that 5α-reductase is involved in male pattern baldness, acne, and hirsutism as well as in BPH. The 5α-reductase inhibitors finasteride (type 2 inhibitor) and dutasteride (type 1 and 2 inhibitor) are clinically useful drugs in the treatment of BPH and male pattern baldness when they are given to appropriate populations of patients.
After DHT is formed from testosterone in the prostate it is then subjected to a series of reversible metabolic reactions to form 3α-diol (5α-androstane-3α,17β-diol) and 3β-diol (5α-androstane-3β,17β-diol) (see Fig. 90–9). The enzymes that perform this transformation of DHT are 3α- or 3β-hydroxysteroid oxidoreductases. These enzymes use NADP as a cofactor, but in contrast to 5α-reductase they can also use nicotinamide-adenine dinucleotide (NAD). The equilibrium for the metabolism of DHT favors the formation of DHT, that is, the 3-hydroxy group of 3α-diol and 3β-diol is oxidized to the 3-ketone that is present in DHT. It is known that administration of 3α-diol to an animal is a strong androgen through its rapid conversion to the effective DHT. On the other hand, 3β-diol is not effective as an androgen because it is rapidly and irreversibly converted to the triol form by hydroxylation in the 6a or 7a position (see Fig. 90–9). The triols are dead-end products of testosterone metabolism but are water soluble and inactive as androgens because they cannot re-form DHT. Steroids also can form glucuronide or sulfate conjugates and be secreted in a more soluble form. In summary, testosterone is irreversibly metabolized to DHT that is in equilibrium with other reduced steroids primarily through oxidation and reduction at the 3 position. The steroids are inactivated by being irreversibly hydroxylated to the inactive triols.
Androgen Regulation of Stromal-Epithelial Interactions
It is now apparent that there is a dynamic and reciprocal interaction between the functions of epithelial cells and those of stromal cells (Steiner, 1993; Cunha, 1994; Sikes, 1995; Cunha et al, 2003, 2004). These crosstalk interactions are mediated through the spatial organization of extracellular matrix elements that form the basement membrane linkage. This linkage presents, filters, and organizes the two-way paracrine signals and the flow of information between those two cellular compartments. For example, fluids, gases, nutrients, hormones, and many growth factors arriving in the prostate through the circulation must first pass through the stromal ground substance, the extracellular matrix, and the basement membrane before reaching the base of the secretory epithelial cells. Early in development, the functions of the epithelial and mesenchymal (stromal) elements vary as to their cell types, compositions, properties, and interactions. It is the integrated system biology of these two tissue elements as well as their dynamics during aging that play a vital role in the prostate’s functions as a unit and gland. Indeed, it is the breakdown of these tissue interactions that is one of the hallmarks of the abnormal growth of the prostate that starts very early in life and is initiated at some time just after maximum virilization, at approximately 25 years of age. The prostate is extremely susceptible to permanent early alterations in form and structure as a consequence of genetic, environmental, dietary, or metabolic factors with aging (Risbridger et al, 2005). In fact, it is essential to establish the link between hormonal (androgens and estrogens) changes that occur during the fetal or neonatal period that imprint and hence may result in onset of late-life disease. With aging, during a period of 50 to 60 years, this organ slowly progresses through the transition from normal zonal histologic anatomy and function to the early signs of BPH, to prostatic inflammatory atrophy, to prostatic interepithelial neoplasia, and finally to various types of prostatic adenocarcinoma. This concept has been thoroughly documented in several rodent models (Rajfer and Coffey, 1978, 1979; Naslund and Coffey, 1986, 1987; Prins and Birch, 1995; Singh et al, 1999; Prins et al, 2001; Risbridger et al, 2005).
Cell Adhesion Molecules
Cell-cell and extracellular matrix interactions are becoming major targets for understanding how the phenotype of a cell is regulated. Transmembrane receptors on the cell surface extend out through the plasma membrane and form a bridge directly connecting the cytoskeleton with proteins and receptors located within the extracellular matrix or on neighboring cells. The cell adhesion molecules (CAMs) are divided into four major types: (1) integrins, which link the cell to the basement membrane and extracellular matrix components through heterodimer interactions; (2) cadherins, which link the cell to neighboring cells through homotypic polymers; (3) selectins, which link the cell to carbohydrate moieties primarily on the vascular system; and (4) immunoglobulin superfamily adhesion molecules. The most extensively studied of these cell adhesion molecules in the prostate in order of interest have been E-cadherins that bind prostate epithelial cells to each other and CD71 that binds to transferrin, as well as several of the integrin molecules. These bindings have been surveyed in prostate tumor cell lines in vitro (Rokhlin and Cohen, 1995), but more extensive work needs to be carried out in vivo in the normal developing prostate and in prostate cancer.
The integrins are made up of two covalently linked heterodimers termed α and β subunits. These integrins serve externally to contact the extracellular matrix receptors of fibronectin, fibrinogens, collagen, and laminin as well as glycosaminoglycans in the proteoglycans of the extracellular matrix. The integrin receptor domains inside the cell compartment serve as focal points for determining the structure and organization of the cytoskeleton. Approximately eight subunits of α and β can interact in different heterodimers that are tissue specific and can be of several types even on one cell. Different combinations may have varying degrees of binding activity for extracellular matrix components. For example, α3β1 binds to laminin, collagen, and fibronectin and does so by recognizing a triplet of amino acids in those proteins made up of arginine, glycine, and aspartic acid (RGD).
Other types of transmembrane receptors also extend out of the cell to make direct cell-cell contact with the neighboring cell by recognizing similar receptors and forming homodimer bonds. Certain homophilic dimers that require calcium for interactions to form cell-cell bonds with neighboring cells are termed cadherins. Four of the cadherin types have been cloned. They contain between 723 and 748 amino acids that are composed of a single peptide, an extracellular region with three repeated domains, a hydrophobic transmembrane region, and a long cytoplasmic tail. There is approximately 50% homology across species and between integrins. The cadherins are classified into three subtypes: E-cadherins found in adult epithelial cells (also earlier termed uvomorulin, cell CAM 120/80, ARC-1, or L-CAM); N-cadherins found in neural tissues of muscle (also termed A-CAM); and P-cadherins found primarily in placenta and epithelium (Albelva, 1994). In the prostate cell, for example, E-cadherins extending out of the surface membrane make contact with the neighboring cell and form a homodimer, and the E-cadherin extending inside the cell by passing through the membrane would form as an organizing center that binds a complex of three cytoplasmic proteins termed catenins α, β, and γ. This complex is localized to the zonula adherins of the cell and participates in junction formation and stabilization of the cytoskeleton. These interlocking matrix systems interact to form a structural network extending externally from cell-cell contact and the extracellular matrix interactions and then internally to cytoskeleton organization and centrally, terminating by direct contact with the nuclear matrix that forms tissue-specific DNA organization.
The interactions of the nonhistone tissue matrix regulate many aspects of DNA functions involved in growth and differentiation (Getzenberg et al, 1990; Boccardo et al, 2003). The nonhistone proteins such as high-mobility group (HMG) proteins include HMGI/Y (HMGA) that participate in numerous cellular processes, such as regulation of inducible gene transcription, integration of retroviruses into chromosomes, and induction of malignant transformation (Reeves and Beckerbauer, 2003). Through protein-DNA and protein-protein interactions the members of the HMGA family can influence growth, cell proliferation, differentiation, and cell death; they influence chromosome dynamics by acting as architectural transcription factors affecting several genes that have an impact on tissue structure and organization. This class of genes is often upregulated in cancer (Reeves and Beckerbauer, 2003). These types of tissue matrix interactions are essential to the understanding of stromal-epithelial interactions because they form direct structural linkages and communications between the stroma and the epithelial nuclear DNA. In summary, under the influence of hormones (estrogens and androgens) and diet, it is the regulation of chromatin structure and organization through the histone and nonhistone pathways that replaces and maintains the tissue organization as well as interactions in health and disease.
The discussion, to this point, has concerned primarily insoluble elements in inducing stromal-epithelial interactions, but soluble hormones such as steroids, vitamins, and growth factors are also important (Sikes, 1995). The prostatic stromal cells contain steroid receptors and respond to both androgens and estrogens (see earlier discussion). Androgens and estrogens can alter the formation of collagen (Coffey and Walsh, 1990) and other extracellular matrix components, such as glycosaminoglycans, in the prostate (DeKlerk et al, 1984; DeKlerk and Human, 1985; Kofoed et al, 1990; Horsfall et al, 1994).
In summary, different components of matrix interactions can have either an inhibitory role in negative regulation of normal prostate growth or a positive role in establishing tumor growth. There have been many hypotheses concerning the mechanism of these epithelial-stromal interactions, but they have yet to be fully resolved.
Key Points: Cell Adhesion Molecules
Regulation of Prostate Growth at The Molecular Level: Steroid Receptors
In almost all cells in the body, steroids can enter the nucleus, but only a few cells can retain this steroid within the nucleus for any length of time. The cells that retain the steroid have receptors that are steroid specific, which can regulate specific steroid-sensitive genes within the nucleus to alter the expression of certain proteins. The AR’s affinity for the nuclear acceptor site to which it binds in the nucleus is probably a compilation of binding to specific sequences on DNA (androgen response elements) as well as tissue-specific binding to coregulatory factors. The uptake and binding of the AR in the nucleus are regulated by the presence of the androgen ligand bound to the receptor, resulting in receptor activation. When androgens are not present, the receptor decreases its affinity for nuclear binding and can easily be removed and, indeed, under castrate conditions, receptors may leak back out into the cytoplasm (Husmann et al, 1990). Immunohistochemical techniques indicate that in the presence of androgen the AR localizes primarily to the nucleus.
The prostate and seminal vesicles contain steroid-specific and high-affinity (10−9 to 10−10 M Kd) saturable (100 to 1000 fmol of receptor per milligram of DNA equivalents of tissue) ARs that were first described by Liao and Fang in 1969. There are 5,000 to 20,000 molecules of these receptors per cell, far more than can bind to the androgen response element sites, which are probably fewer than 400. Classic AR function has been characterized as a genomic process wherein certain transgenes are regulated by the activated AR. More recently, however, attention has been focusing on the nongenomic mechanisms of androgen action (Benten et al, 1997; Jones et al, 2004). Nongenomic androgen action is characterized by extremely rapid changes in cellular physiology, typically measured in seconds or minutes, as opposed to the longer times required for target gene regulation to be translated into protein changes. Whether nongenomic androgen action occurs through the same ARs involved in genomic regulation still remains to be determined. The properties and hormonal regulation of ARs as well as their uses have been reviewed in detail (Gelmann, 2002; Black and Paschal, 2004).
Key Points: Androgen Receptor
Androgen Receptor
The cloning of the human AR and its expression was a hallmark in the study of the mechanism of androgen action (Chang et al, 1988b; Lubahn et al, 1988). This led to the study of the sequence of the gene and its protein product—and how this is altered in inherited androgen insensitivity syndromes—as well as receptor function (Chang et al, 1995).
The AR gene is on the long arm of the X chromosome at position Xq11.2-q12. Because there is only one X chromosome in a male, it is a single copy gene. The coding sequence on this gene is divided into 8 exons that are transcribed and processed into mRNA and then subsequently translated into protein. The total genomic DNA spans a minimum of 80 kilobases (Marcelli et al, 1990) but forms a final message of only 10.6 kilobases, that is, only 17% of the total gene, with an open reading frame of 2757 base pairs. This is similar to the organization of many other steroid receptors that also contain information from 8 exons, such as the progesterone and estrogen receptors. The AR is a member of the nuclear receptor superfamily, which is a group of ligand-inducible transcription factors. The nuclear receptor superfamily has more than 200 members at present (Escriva et al, 2004). All of these receptors share certain structural features that allow them to regulate gene expression, although the ligands for many of these receptors have yet to be identified (so-called orphan receptors). Such receptors include the glucocorticoid receptor, the retinoic acid receptors (RXR and RAR), the vitamin D receptor, the estrogen and progesterone receptors, the peroxisome proliferator-activated receptor (PPAR-γ), and many orphan receptors. Like other steroid receptors, the AR is divided into three distinct, modular domains: the amino-terminal domain, the DNA-binding domain, and the carboxyl-terminal ligand-binding domain. Despite the similarity in structural organization of all of the nuclear receptors, activation of different receptors results in markedly different cellular responses. Mutational analyses of the human AR have allowed a detailed mapping of a variety of different functions, which are diagrammed schematically in Figure 90–10.
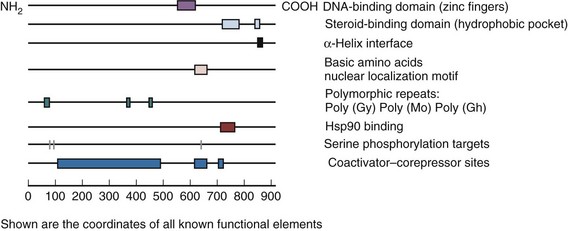
Figure 90–10 Structure of the human androgen receptor protein. The androgen receptor is divided into several functional domains including the DNA-binding domain (consisting of two zinc fingers), a steroid-binding domain (consisting of a hydrophobic pocket), a nuclear localization motif, and several coactivator/corepressor binding sites. There are three polymorphic repeats of glycine, proline, and glutamine, with varying sizes among different populations. The relative positions of the functional elements are shown to scale.
Upstream (5′ direction) of the transcriptional initiation site is the regulatory element of the gene that controls its expression. It is unusual in that it contains the GC box rather than the classic TATA and CCAAT that are commonly found in promoters of polymerase II–dependent genes. Closer to the initiation site located only 70 base pairs upstream is a 50-base pair purine-rich region that is a cis-acting element for AR transcription. There are other cis-acting elements, including an AP-1 (which is bound by a heterodimer of c-Fos and c-Jun) and a RARE (retinoic acid response element) as well as a cyclic adenosine monophosphate (AMP) response element (AR/CRE1). This suggests that the regulation of expression of the AR gene may involve cyclic AMP, activation of c-Fos/c-Jun, or retinoids (Kuiper et al, 1989; Faber et al, 1993; Mizokami et al, 1994; Young et al, 1994). Activation of the AR appears to be a function of multiple steps including initial complex formation with certain chaperonins, binding of ligand, post-translational modifications, dimerization, nuclear localization, and binding of the receptor to certain transcriptional coactivator complexes that remodel chromatin, target the initiation site, and stabilize the RNA polymerase II machinery for repeated rounds of transcription. Each of these features is discussed in the context of known structural features of the receptor as diagrammed in Figure 90–11.
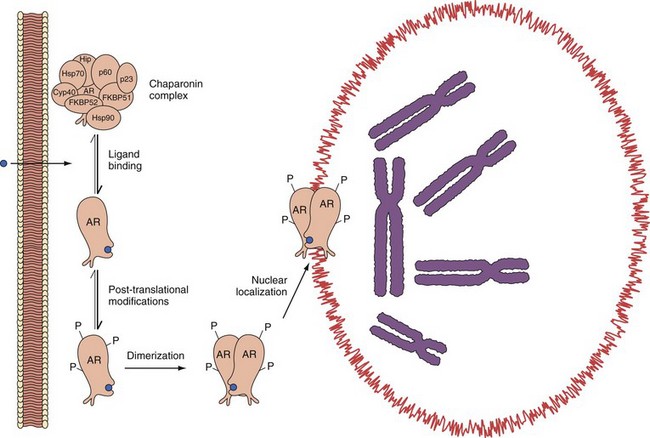
Figure 90–11 Mechanism of androgen receptor (AR) activation by ligand. Androgen enters the cell membrane by passive diffusion and binds the androgen receptor in the cytoplasm. The androgen receptor exists in equilibrium with the chaperonin complex, consisting of at least eight different components, including Hsp90, Hsp70, Hip, p60, p23, FKBP51, FKBP52, and Cyp40. Once activated by binding ligand, post-translational modifications occur, such as phosphorylation. Contemporaneously, dimerization occurs, and the activated modified androgen receptor is translocated to the nucleus by active transport.
Chaperonin Binding
Immediately after nascent production of the protein in the ribosome, the receptor forms complexes with several other proteins, referred to as chaperonins. These chaperonins form an aggregate complex, which is known as the 8S complex, in reference to the size of the complex on sucrose gradient sedimentation analysis. This chaperonin complex includes at least eight known components (Hsp90, Hsp70, Hip, p60, p23, FKBP51, FKBP52, and Cyp40), which serve to sequester the receptor into an inactive pool (see Fig. 90–11). By analogy to the progesterone receptor, which has had the most detailed scrutiny in regard to the molecular biology of the chaperonin complex (Nair et al, 1996; Pratt and Toft, 1997; Smith, 2000), the AR can dissociate into a monomeric form (4S on sucrose gradient centrifugation) that is in equilibrium with the 8S form, with the preponderant species existing in the chaperonin complex. This larger complex may be particularly favored by virtue of mass action alone, because the heat shock proteins tend to be among the most abundant proteins in the cell. Although the AR is uncomplexed, it is susceptible to various different post-translational processing steps, including phosphorylation or glycosylation. Such interactions may then inhibit reassociation with the chaperonins, leading to ligand-dependent activation, ligand-independent activation, or receptor inactivation with proteasome-mediated degradation. Evidence for such a mechanism includes a PEST sequence similar to one in the vitamin D receptor that is present in the hinge region of all known mammalian ARs, suggesting that it may function in proteasome-mediated AR turnover. Moreover, proteasome inhibition leads to a significant increase in AR isoforms (Sheflin et al, 2000).
DNA-Binding Domain
Near the end of exon 1 and extending into exon 3 is the coding sequence for the DNA-binding domain. The DNA-binding domain of the AR consists of 72 amino acids rich in cysteine and encoding two zinc finger motifs, which allows specific recognition of certain DNA sequences referred to as androgen response elements. Such elements typically consist of a palindromic repeat separated by a three-nucleotide spacer, for example, GG(A/T)ACAnnnTGTTCT (Roche et al, 1992). X-ray crystallography of certain steroid receptors (glucocorticoid receptor and progesterone receptor) has shown that the first zinc finger directs sequence specificity of binding by directly contacting the DNA bases in the major groove; the second zinc finger functions to stabilize the protein-DNA complex by contacting the sugar-phosphate backbone. Although the protein-DNA interaction appears to be largely limited to the zinc finger motifs, sequences in the amino terminus appear to be important in stabilizing these structures because mutations in this region result in a mildly diminished DNA-binding affinity. This DNA-binding domain of the zinc fingers in the steroid receptor molecule is highly conserved. In this region of exon 2-3, there is a 79% homology with the progesterone receptor, a 76% homology with the glucocorticoid receptor, and a 56% homology with the ER (Chang et al, 1988a, 1988b). The closest homology of the AR is with the progesterone receptor (Lubahn et al, 1988; Marcelli et al, 1990). Mutations of amino acids in this area of the AR can make the receptor unable to activate androgen-sensitive genes (Govindan, 1990), which is the basis of one of the inherited androgen-insensitivity syndromes—testicular feminization.
The DNA-binding domain binds to its cognate DNA-regulatory site, referred to as a hormone response element. Hormone response elements can be divided into different groups on the basis of common structural features for which whole groups of receptors are capable of binding. Class I hormone response elements include the glucocorticoid receptor, the progesterone receptor, and the mineralocorticoid receptor and are characterized by a half-site consensus sequence of TGTTCT. Class II hormone response elements include the ER, whose prototype half-site sequence is TGACC. The hormone response elements to which ARs have been shown to bind belong to the class I subgroup (Tan, 1990). A consensus sequence for the androgen response element has been determined by an RNA-binding site selection assay with an AR fusion protein to be GG(A/T)ACAnnnTGTTCT (Roche et al, 1992). Such binding sites are characterized by an inverted palindromic repeat with a dyad axis of symmetry, indicating that the receptors are binding in a head-to-head fashion. Recently, however, the androgen response element of the rat probasin promoter has been found to be a direct repeat (Schoenmakers et al, 2000). Surprisingly, x-ray crystallography data reveal that AR dimers bind to direct repeat target sequences in a head-to-head fashion, maintaining the normal orientation of an inverted repeat target sequence (Shaffer et al, 2004). To date, only the AR has been found to bind to a direct repeat target sequence with an orientation normally expected for an inverted repeat. This difference may represent one way ARs maintain specificity of target gene regulation.
Ligand-Binding Domain
Ligand-dependent activation is characterized by ligand-receptor dimerization, post-translational modifications (e.g., phosphorylation), nuclear translocation, and subsequent target gene activation (or repression). It is believed that binding of either DHT or testosterone to the ligand-binding domain can facilitate these processes, although the binding affinity for DHT is significantly higher than for testosterone. Binding of the androgen to the carboxyl-terminal ligand-binding domain is required for activation; however, deletion of the ligand-binding domain can lead to a constitutively active AR. Thus, at least part of the interaction with the chaperonin complex involves the carboxyl portion of the receptor (Marcelli et al, 1990). However, small point mutations in the ligand-binding region can lead to significant changes in the characteristics of AR action. For instance, a single point mutation in the ligand-binding domain of the AR (codon 877, Thr->Ala) identified in the LNCaP cell line of prostate cancer renders it weakly inducible by inappropriate steroids such as progesterone while retaining the ability to be stimulated by androgens. Marcelli and coworkers (1990) reported that mutations in the AR at amino acid 587 or 794 are inactive in the assay for androgen binding and for transcriptional activation. However, the removal of amino acids from 708 to the carboxyl end at 917 (i.e., the entire ligand-binding domain) leads to the synthesis of a receptor protein that does not bind androgens but is still constitutively active in activating transgenes.
Dimerization
The identification of the palindromic structure of certain hormone response elements for all steroid receptors led to the proposal that these transcription factors bind to the DNA as a dimer. Subsequent analysis of the receptor-DNA interactions has confirmed this hypothesis, and dimerization is now thought to represent an important step in the regulation of steroid receptor action. A hydrophobic heptad repeat within the ligand-binding domain at codons 859 to 880 is conserved among all steroid receptors and is thought to be necessary for high-affinity dimerization. Removal of these sequences leads to low-affinity dimerization, presumably through the action of the DNA-binding zinc fingers on the palindromic androgen response elements. Abolishment of the DNA-binding domain does not inhibit the strong affinity dimerization present in the ligand-binding domain. The strong dimerization signal appears to be related to a hydrophobic α-helix interface formed by the conserved heptad (Centenera et al, 2008).
Post-translational Modifications
Once the AR has been bound to steroid ligand and dissociated from the chaperonin complex, it is susceptible to a variety of post-translational modifications, any one of which may significantly affect the function and turnover of the receptor. For instance, the AR can be acetylated (Fu et al, 2004) or phosphorylated (Goueli et al, 1984). In the rat ventral prostate it has been reported that this occurs through a nuclear cyclic AMP–dependent protein kinase (Kemppainen et al, 1992). Receptor phosphorylation may be an important mechanism in the nuclear translocation of steroid receptors as well as in DNA binding and transcriptional regulation. The stimulation for phosphorylation appears to be optimal with the binding of an androgen agonist, because antagonists such as flutamide appear to favor the dephosphorylated state, suggesting that the phosphorylation status may be associated with the ultimate activity of the receptor (Wang et al, 1999). Both serine and tyrosine residues have been found to be phosphorylated in other steroid receptors (Landers and Spelsberg, 1992; Sadar et al, 1999). In addition to phosphorylation by protein kinase A, ARs also appear to stimulate mitogen-activated protein kinases, which may provide a different level of regulation of gene activity because such kinases often modulate other transcription factors, such as Elk-1 (Peterziel et al, 1999). The prostate is a rich source of acid phosphatases, and some have suggested that these enzymes may be active in regulating the phosphotyrosyl residues of the AR, thus playing a role in dephosphorylation and inactivation of ARs (Goldsteyn, 1989), although this relationship is certainly not causal.
Nuclear Localization
After activation by the binding of steroid ligand the AR is transported to the nucleus across the nuclear pore complex by a process involving at least two nuclear localization signals, one for import and one for nuclear export. Evidence for nuclear localization signal-dependent nuclear translocation is well established and can be found in a variety of nuclear proteins, including the SV40 large T antigen. In most cases it consists of a stretch of basic amino acids. The prototype nuclear localization signal from the SV40 large T antigen is PKKKRKV, although various other basic sequences have also been implicated in nuclear localization signaling. The nuclear localization of the AR appears to involve multiple steps, including the binding of the basic amino acid nuclear localization signal to importins α and β, docking of an importin-cargo complex to the nuclear pore, translocation to the nucleus, and Ran-GTP–mediated release of the cargo (Rao et al, 2002). Two regions of steroid receptors have gained most attention as regulators of receptor trafficking. The first region is the second DNA-binding zinc finger region together with the flanking hinge region (NL1) consisting of a bipartite signal including flanking leucines and the core signal 628RKLKKLGN (Kemppainen et al, 1992; Ylikomi et al, 1992; Poukka et al, 2000). However, this one putative nuclear signal peptide is not sufficient by itself for high-efficiency translocation; and by analogy to other steroid receptors, additional nuclear localization signals may exist in the steroid-binding domain (Kemppainen et al, 1992). NL1 acts constitutively and participates in rapid nuclear import that is facilitated by importin-α binding (Savory et al, 1999). A number of coregulators of steroid receptor–mediated transactivation interact with regions that encompass NL1 (Jackson et al, 1997; Moilanen et al, 1998; Powers et al, 1998; McKenna et al, 1999). Some of these proteins, such as SNURF and Ubc9, lose their ability to interact with AR when the region overlapping with the bipartite NL1 is destroyed (Moilanen et al, 1998; Poukka et al, 2000). A second signal, NESAR, is located in the ligand binding domain (Saporita et al, 2003) and is a nuclear export signal that facilitates export of AR when it is not complexed to ligand. Between the NL1 and NESAR, the AR is actively shuttled between the cytoplasm and nucleus, presumably to tightly regulate its intrinsic ability to activate or repress gene expression.
Transcriptional Activation Domains
Once the AR has achieved active localization into the nucleus it must find and bind to its target sequences in the genomic DNA. The precise mechanism by which receptor localization occurs to target genes is still unknown; however, there is emerging evidence indicating it is a highly orchestrated process (O’Malley, 2008). For instance, it is now known that so-called pioneer factors such as FOXA1 are targeted to specific chromosomal sites by epigenetic signaling, and then the ARs bind to these sites to subsequently regulate the target genes (Lupien et al, 2008). Once the AR has properly localized within the target chromatin sites it must coordinate binding to a number of associated factors referred to as coactivators and corepressors that subsequently regulate gene expression (Fig. 90–12). A list of recently identified coactivators is provided in Table 90–4. Most of these factors interact promiscuously with many steroid receptors, although more AR-specific factors are routinely being discovered. Because the number of potential coregulators clearly exceeds the capacity for direct interaction by a single receptor, the most likely mechanism is that transcriptional activation by AR involves multiple factors that act in both a sequential and combinatorial manner to reorganize chromatin templates (Pollard and Peterson, 1998). The precise timing and sequence of binding of these factors remain to be elucidated; however, one can generally break down the processes empirically into chromatin/nucleosomal remodeling (an energy-dependent process), histone acetyltransferase activity, and subsequent recruitment of TATA-binding protein–associated factors, all of which promote an increased rate of gene transcription by RNA polymerase II. Under certain conditions, such as binding of an AR antagonist (e.g., flutamide), the histone acetyltransferase activity is actually inhibited and transrepression may occur. Such inhibition of gene expression appears to involve the nuclear corepressor proteins N-CoR and SMRT (Glass and Rosenfeld, 2000). Other proteins may play a similar role, such as the HBO1 gene (Sharma et al, 2000). Amino-terminal deletions in region 46-408 result in dominant negative suppression of hormone-inducible transgene activation, indicating that the coactivator functions require an interaction within that site and that in the absence of this region the receptor forms dysfunctional complexes in the chromatin (Palvimo et al, 1993).
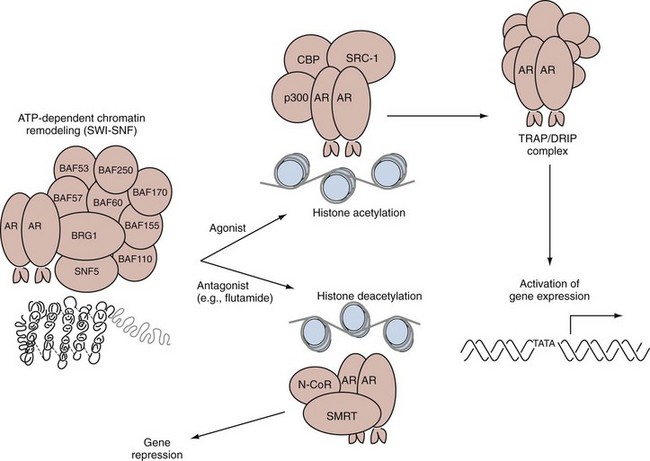
Figure 90–12 Mechanism of action of the nuclear-activated androgen receptor (AR). Once the androgen receptor has been translocated to the nucleus it undergoes several steps (many of which may occur contemporaneously): (1) chromatin remodeling in an ATP-dependent fashion via the SWI-SNF complex; and (2) agonist (i.e., DHT)-mediated histone acetylation in a process involving multiple transmission factors including p300, CBP, and SRC1. In the case of a certain antagonist, histone deacetylation may be favored, and the activated nuclear receptors complex with repressors of gene expression such as N-CoR and SMRT. (3) The activated androgen receptor complexes then associate with other γ-trans-acting factors via the TRAP/DRIP complex at sites typically upstream of the target gene known as androgen response elements. This complex then leads to androgen-regulated activation of gene expression.
Table 90–4 Abbreviated List of Putative Androgen Receptor Coactivators
The transcription domain of the AR is coded in exon 1, which is the largest of the exons, containing 1607 base pairs. Analysis of this region reveals three homopolymeric repeated regions, including a repeat of approximately 20 glutamines, followed by a space containing 8 repetitive prolines and 23 repetitive glycine units (see Fig. 90–12). The glutamine repeats form a β sheet that helps form a polar zipper, which favors certain protein-protein interactions. Fusion of this type of polymeric glutamine repeat with the DNA-binding domain of GAL4 in yeast results in a GAL4-directed increase in transcriptional activity, demonstrating the importance of this region in promoting transgene activation (Gerber et al, 1994).
Studies demonstrate that these polyglutamine repeats appear to interact directly with the carboxyl terminus of the transcription factor p160 (Irvine et al, 2000). In the normal population this repeat varies over a length of 11 to 31 residues, resulting in a true allelic polymorphism. This means that different people have alleles of variant polyglutamine repeat units. This variation is racially defined, and it has been suggested that this may be related to the differences in the incidence rates of prostate cancer in different ethnic groups. The most common CAG repeat length in whites is a modal value of 21; in African-Americans, it is shorter at 18; and in Asians, it is longer at a mean of 23. The longer the glutamine repeats, the lower the activity of the AR in activating target genes. Patients with X-linked spinal and bulbar muscular atrophy, termed Kennedy disease, possess a larger glutamine repeat in the range of 40 to 60. The ARs in Kennedy disease exhibit markedly less transactivation activity (Laspada, 1991). Moreover, men with male factor infertility are found to have longer than average polymorphisms of the AR than in normal controls (Tut et al, 1997). Genetic studies of inherited diseases of androgen insensitivity and overvirilization as well as changes in the AR mutation associated with prostate cancer and its biologic properties will be of great help in unraveling the role of the human AR in relation to its structure.
Androgen Receptor–Dependent Chromatin Remodeling
Part of the tissue and gene specificity in the recognition of receptors and DNA may depend on the organization of the DNA within the nucleus (Getzenberg et al, 1990). The steroid-receptor complex can interact only with genes that are in regions that are “open” or in the transcriptionally active form. Studies show that these open regions of chromatin (euchromatin) can extend up to 100,000 base pairs in length, or more than 10-fold the size of a typical gene, which usually ranges from 1000 to 10,000 base pairs. It is unknown how such a large range of DNA is altered in conformation, but it may be through binding to structures like the nuclear matrix, which can order large loop domains in the region of 60,000 to 120,000 base pairs. Nuclear receptors are thought to interact with chromatin remodeling complexes in an ATP-dependent fashion, in a process directed in part by “pioneer factors” including FOXA1 (Lupien et al, 2008), and this may be among the earliest steps in the ultimate regulation of certain target genes (Glass and Rosenfeld, 2000).
During cell division, chromosomal organization is spatially regulated at each of the critical phases of mitosis (Williams and Fisher, 2003). Epigenetic regulation of chromosome structure and function is highly ordered during cell division, differentiation, and development (Lam et al, 2005; Margueron et al, 2005). In fact, chromosomal proteins are required to maintain such ordered structure for euchromatin, heterochromatin, and centromeric chromatin to sustain normal cell and tissue functions. To achieve such coordinate regulation, the protective packaging of DNA is engineered through an elegant system of tightly wound DNA around an eight-component histone core called a nucleosome. This core consists of dimers of H2A, H2B, H3, and H4, whose ability to compact DNA is directly regulated by post-translational modifications. The selective regulation of such post-translational histone modification constitutes a major regulatory mechanism for gene expression and is referred to as the histone code (see Fig. 90–12). Histone modifications include acetylation, phosphorylation, ubiquination, and methylation (Downs and Jackson, 2003; He et al, 2003; Cosgrove et al, 2004; Cosgrove and Wolberger, 2005; Lam et al, 2005).
ARs are known to interact with structural components of the chromatin organizing complexes. Such complexes include the multisubunit human SWI-SNF complex, which has been shown to remodel mononucleosome and polynucleosome templates in an ATP-dependent manner (Peterson and Tamkun, 1995). The isolated hSWI-SNF ATPase subunits BRG1 and hBRM also have these activities (Phelan et al, 2000).
The transcriptional activation of nuclear receptors (neuroendocrine cells) requires multiple factors including SWI-SNF complex, CPB/p300, and SRC family members, which are large and contain numerous subunits, many of which make contact with a variety of nucleosome components and the nuclear matrix (Huang et al, 2003). Such components include BAF53a, BAF57, BAF60, BAF110, BAF155, BAF170, BAF250, BRG1, BRM, and SNF5. Because condensed chromatin renders genes inaccessible for transcription, the combination of a steroid receptor along with the SWI-SNF complex formation appears to be critical for appropriate nucleosomal remodeling to allow appropriate target genes to be accessible for gene regulation (Sudarsanam and Winston, 2000; Huang et al, 2003). Once the receptor SWI-SNF complex, CPB/p300, and other mediators have successfully “opened” the structure of the chromatin to allow transcriptional regulation, the AR must interact with a distinctly different set of cofactors. Post-translational histone modification clearly appears to be requisite for chromosome remodeling and optimal gene expression (Ewen, 2000; He et al, 2003). In most models tested the rates of gene transcription actually correlate with the degree of modification of histones through acetylation, phosphorylation, ubiquination, and methylation. In other words, hyperacetylated histone regions correspond to the highest gene-transcriptional regions, whereas the hypoacetylated histone regions correspond to the lowest gene-transcriptional regions (Pazin and Kadonaga, 1997). A number of histone acetyltransferase complexes have been found to be associated with nuclear receptors, including the AR. These complexes include p/CAF, a homologue to the yeast GCN5, which participates in the yeast SAGA complex. This complex includes factors that possess HAT activity but also the TATA-binding protein (TBP) and a number of TBP-associated factors. The p/CAF protein has been found to be associated with the retinoic acid receptor and may be involved with multiple nuclear receptors. It is also known to bind to other HAT proteins, including CBP/p300, which is known to acetylate not only histones but other transcription factors as well. The CBP/p300 complex is an essential coactivator for many genes and may actually serve as a molecular scaffold in stimulating gene transcription (McKenna et al, 1999; Huang et al, 2003; Marshall et al, 2003). Such complexes include the SRC1 coactivator, among others. More recently, the modifiers of the histone code have expanded considerably to include sophisticated enzyme-mediated alterations in the histones H2A, H2B, H3, and H4 by phosphorylation, ubiquination, and methylation that open the chromatin, allowing the recruitment of important transcription factors that allow normal cell functions (Lam et al, 2005; Margueron et al, 2005). The list of coactivators associated with nuclear receptors and the AR in particular is extensive and almost certainly incomplete. A short list of factors found to be associated with the receptor at this level of gene regulation is given in Table 90–4. Among the most important factors is SRC1 (steroid receptor coactivator 1), which has mild HAT activity and seems to be required for optimal stimulation of steroid-dependent transcription. Additional factors include SRA, a structural RNA necessary for the coactivator complex to function optimally (McKenna et al, 1999), and p160 coactivator, which appears to be required for hormone-dependent activation and to directly interact with the polyglutamine repeats found in the amino-terminal transactivation domain (Irvine et al, 2000).
Most genes have a regulatory region immediately upstream of the transcriptional start site. The regulatory region is divided into a core promoter element that is present in all genes as well as other upstream elements that serve to regulate the overall gene expression pattern. This promoter element specifies the site to which RNA polymerase II will attach to the DNA and will determine the actual point for the initiation of transcription. The RNA polymerase will copy or transcribe the DNA code into mRNA, a process termed transcription. This promoter area starts at −16 nucleotides to +32 upstream from the gene initiation site. This region of −32 to +16 was originally referred to as the Goldberg-Hogness box or TATA box and has a consensus sequence of TATAAAAG. The RNA polymerase II enzyme binds to this TATA box as one of the initial steps in transcription. Farther upstream from the TATA box is a second gene control element termed generically the hormone response element, which has been identified in many genes regulated by steroid hormones and is one of multiple sites where the receptor binds to the DNA. As stated earlier, in androgen-regulated genes, this area is termed the androgen response element; in estrogen, the estrogen response element; and in glucocorticoid, the glucocorticoid response element. This hormone response element area may contain several discrete sequences, but its overall role is to modulate the frequency of transcription initiation vis-à-vis interactions with transcriptional factors. In independent analysis, thyroid hormone receptors were found to be associated with affinity purified proteins, which were found to markedly enhance ligand-dependent cell-free transcription. This complex was referred to as TRAP, for thyroid receptor-associated proteins (Fondell et al, 1996). In a similar set of experiments, the same type of complex was isolated for the vitamin D receptor and was called DRIP for D receptor–interacting proteins (Rachez et al, 1998). Subsequent analysis revealed that they shared at least nine proteins, and this activator-recruited cofactor complex (TRAP/DRIP/ARC) is part of a large composite coactivator complex used by a variety of transcription factors for the regulation of certain target genes. Such transcription factors include SREBP, NFκB, and VP16 (Sun et al, 1998; Gu et al, 1999; Ito et al, 1999; Naar et al, 1999; Ryu et al, 1999).
In summary, the TATA box tells where RNA polymerase binds and where the initiation of transcription is to start and the hormone response element regulates how frequently it is to be transcribed when it is bound to a hormone receptor. This is accomplished by the presence of certain cofactors in the TRAP/DRIP complex. Because the hormone response element of DNA sequence has been shown to be independent of its position or its orientation, it resembles what has been called the transcription enhancer element that has been found in many other types of genes. The hormone response element section can vary in its location upstream from the initiation of the gene from −20 to −6000 for various different types of hormones. With the steroid hormones, it appears to reside about −140 nucleotides upstream from the initiation site. For example, in the glucocorticoid receptor recognition element the site for glucocorticoid receptor recognition is approximately −140 and contains a sequence of nucleotides of AAAATGGAC. Deletion mapping experiments have indicated that the receptor-binding domain located in the hormone response element is indeed required for receptor binding and is necessary for steroid-mediated control of transcription.
Once the DNA is transcribed into mRNA, a series of adenine units are added to the end (called the poly-A tail) and then the mRNA is cut and spliced on small nuclear particles (called splicesomes) located on the nuclear matrix, and this splicing removes the intron portion of the message. The final mRNA is shipped out of the nucleus, believed to occur on the structural components of the nuclear matrix, and passes through the pore complexes of the nucleus and out to the ribosomes where the mRNA is then translated into protein product, a step termed translation. The proteins have specific amino acid sequences that instruct the cell where to ship the protein in relation to secretory granules or to the membrane area. The protein can also be modified after translation by the subsequent addition of carbohydrates to become glycoproteins or to be phosphorylated by kinases. Under appropriate signals, such as neurologic control, secretory proteins can then be excreted into the lumen of the prostate. This is a process that occurs when secretory proteins of the prostate and seminal vesicles are formed into the ejaculate. A schematic example of this process is shown in Figure 90–8, and such a system would include PSA and acid phosphatase as well as many other protein products that are regulated in their synthesis by androgens through receptor interactions.
During embryonic development, the AR appears first in the mesenchyme of both the rat ventral prostate and seminal vesicles and a few days later in the epithelial cells, but it is unknown what regulates this timing. In the development of the seminal vesicles and the wolffian duct, testosterone appears to be the primary androgen in glandular development, and in the ventral prostate that forms from the urogenital sinus the androgen is primarily DHT. Both testosterone and DHT can bind to the AR; however, on a molar basis, DHT is 3 to 10 times more potent. This decrease in potency of testosterone is believed to be due to the rapid off rates of the testosterone once it is bound so that equilibrium results in less receptor occupancy compared with DHT at similar tissue levels. A report indicates that in some cases, AR-mediated transcriptional regulation can occur even in the absence of direct interaction of the AR with the specific androgen response elements (Kallio et al, 1995). The report stated that the ARs are able to elicit both transrepression and transactivation without interacting directly with the specific DNA elements. This may indicate that the AR can bind to regulatory units within the transcription factors and thus alter their properties even in the absence of direct DNA binding to an androgen response element.
The next part of this discussion is focused on the structure of the nucleus, where the genetic information of the genes, the AR interactions, and the mRNA processing occur and are integrated. This is within a highly ordered structure of the nucleus that is determined by a residual scaffolding framework, called the nuclear matrix, which provides three-dimensional organization to both the nucleus and the DNA.
Role of the Nuclear Matrix in Androgen Action
The DNA may be identical in every cell of different tissues in the body, but it appears to be organized in a different three-dimensional array in different cell types. This spatial organization of DNA appears to be determined in part by nuclear architecture and structure dictated by the scaffolding element termed the nuclear matrix. Therefore, more may be required than just a steroid receptor and a DNA sequence with an androgen response element to determine the high tissue specificity of androgen hormone action. It may require regulation of DNA conformation and three-dimensional structure. There is strong evidence to support the belief that structural components of the nucleus may organize the DNA into different topologic constraints that permit specific steroid receptor interactions themselves. It is also believed that these structural modifications of DNA topology may be an integral part of differentiation. The nuclear matrix has been proposed to be an important structural element in this type of DNA organization (Getzenberg et al, 1990; Boccardo et al, 2003). The matrix facilitates the location of target genes and their conformation and facilitates their cointeraction with steroid receptors. Barrack and Coffey first showed that the nuclear matrix is a major target for androgen and ER binding (Barrack and Coffey, 1980; Barrack, 1987). Because the matrix has been implicated in many important nuclear events, it would provide an ideal target for androgen action. The nuclear matrix has been defined as the dynamic structural subcomponent of the nucleus that directs the functional organization of DNA into loop domains and provides sites for the specific control of nucleic acids (Nelson et al, 1986; Getzenberg et al, 1990). Conceptually, it can be viewed as the nuclear equivalent to the cytomatrix or cytoskeleton. The nuclear matrix contains residual nuclear elements, including the pore complex lamina, the residual nucleolus, and an internal ribonucleoprotein particle network attached to a dynamic fibrous protein mesh (Berezney and Coffey, 1977). The nuclear matrix may be isolated by sequential extractions employing nonionic detergent, brief digestion with DNase I, and a hypertonic salt buffer wash. The residual nuclear matrix structures represent only 15% or less of the original total nuclear mass. More than 98% of the DNA, 70% of the RNA, and 90% of the nuclear proteins have been extracted, and the remaining structure is essentially devoid of histones and lipids.
The nuclear matrix has been implicated as an important structural component in a wide variety of biologic functions. There are approximately 50,000 DNA loop domains in a nucleus, each containing about 60 kilobase pairs of DNA, and these loops are attached at their bases to the nuclear matrix (Pardoll et al, 1980; Vogelstein et al, 1980; Luke and Coffey, 1994). This loop organization is maintained during interphase and throughout metaphase (Nelson et al, 1986). Topoisomerase II, an enzyme that modulates DNA twisting and topology, is associated with the nuclear matrix and the mitotic chromosome scaffold. Many studies with a wide variety of systems have demonstrated that active genes are associated with the nuclear matrix, whereas transcriptionally inactive genes are not close to the matrix. This location of active genes on the matrix provides evidence that the matrix plays an important organizing role in differentiation, placing genes in different configuration.
Androgens can activate DNA synthesis and cell replication in target tissues. The nuclear matrix also serves an important role in DNA replication. The matrix contains fixed sites for DNA synthesis (Pardoll et al, 1980) located at the base of the DNA loop. During DNA synthesis the DNA loop domains are reeled down through the attached replicating complex that is fixed on the matrix. Therefore, the DNA replication fork, DNA polymerase, and newly replicated DNA have been shown to be associated with the nuclear matrix. It is easy to visualize how hormone action and alteration in the nuclear matrix structures could impinge on the androgen regulation of DNA synthesis and growth in a prostate cell.
The nuclear matrix is also associated with mRNA synthesis during transcription. Ciejek and colleagues (1982) observed that more than 95% of the unprocessed mRNA precursor for ovalbumin is associated with nuclear matrix of the chick oviduct. When the intron portions of the RNA were spliced out, the mature mRNA was released from the nuclear matrix. This led them to suggest that the nuclear matrix is involved in RNA processing. Marriman and van Venrooij (1985) have reported that all RNA cleavage products and RNA processing intermediates are firmly bound to nuclear matrix. Once again, alterations in nuclear matrix structures with steroid receptor interactions could alter important steps in transcription and RNA processing. The nuclear matrix contains the attachment sites for the small nuclear ribonucleoprotein particles that are part of the nuclear splicesome system central to the nuclear processing of RNA to the final mRNA, which is transported out to the cytoplasm to be translated.
Ahmed and colleagues have carried out an extensive series of studies of the phosphorylation of the nuclear matrix and related proteins in the ventral prostate of the rat after androgen stimulation and withdrawal (Ahmed, 1987; Ahmed et al, 1993; Tawfic et al, 1993, 1994). They have shown that the nuclear matrix can be phosphorylated by casein kinase 2 (CK-2). One of the targets of this phosphorylation is nucleolin, which is an abundant nucleolar phosphoprotein involved in the synthesis of ribosomal DNA and exquisitely regulated by androgens (Tawfic et al, 1994). Another important protein in nucleolar function that is required for growth is B23, which is also regulated by CK-2 (Tawfic et al, 1993).
In summary, the nuclear matrix is an important structural modulator of nuclear regulation and is an ideal target for hormonal regulation. Indeed, the nuclear matrix is a major site of steroid hormone receptor binding (Barrack and Coffey, 1982; Donnelly et al, 1983; Wilson and Colvard, 1984; Alexander et al, 1987; Barrack, 1987; Metzger and Korach, 1990; Luke and Coffey, 1994). In the prostate, more than 60% of all nuclear ARs are associated with the nuclear matrix (Barrack and Coffey, 1982). The matrix is also a target for many other types of regulatory interactions, including the nuclear products of oncogenes and viral proteins that can also induce growth regulation similar to hormone-induced growth. For example, the nuclear matrix is reported to be a cellular target for the retrovirus Myc oncogene protein and the polyoma large T antigen. All of these transformation proteins that bind to the nucleus are believed to be early molecular events in carcinogenesis or transformation. Therefore, the observation that ARs interact with the matrix has precedence with the matrix as a common target in factors that regulate cell structure and function.
Key Points: Androgen Receptor Structure and Function
Prominent, Nonpeptide Components of Prostatic Secretions
The seminal plasma is formed primarily from the secretions of the sex accessory tissues, which provide a suitable environment for survival and function of spermatozoa. The sex accessory tissues include the epididymis, ampullae, seminal vesicles, prostate, Cowper (bulbourethral) gland, and glands of Littre. The average volume of the normal human ejaculate is approximately 3 mL, ranging from 2 to 6 mL, and it has two components: spermatozoa and seminal plasma. Spermatozoa, which represent less than 1% of the total ejaculate, are present in the range of 100 million/mL. The major contribution to the volume of seminal plasma (average 3 mL) comes from the seminal vesicles (1.5 to 2 mL), from the prostate (0.5 mL), and from the Cowper gland and glands of Littre (0.1 to 0.2 mL). During ejaculation the secretions of these glands are released in a sequential manner (Amelar, 1962; Amelar and Hotchkiss, 1965; Tauber and Zaneveld, 1976; Zaneveld, 1981). The first fraction of the human ejaculate is rich in sperm and prostatic secretions, such as citric acid. The level of fructose, which represents a major secretory product of the seminal vesicles, is elevated in the later fraction of ejaculate. More recently, seminal albumin was measured in seminal plasma and the authors demonstrated an association with sperm morphology but not several other semen parameters (Elzanaty et al, 2007). The overall chemical composition of normal human and rodent prostatic secretions and seminal plasma has been widely studied, and the results have been summarized in excellent reviews (Mann, 1981; Zaneveld, 1981; Aumuller and Seitz, 1990; Daniels, 1990; Chow et al, 1993; Gonzalez, 1993; Elzanaty et al, 2007). Recently, an analysis of expressed prostatic secretions (EPS) revealed that citrate, myoinositol, and spermine metabolite measurements could significantly differentiate controls from men with prostate cancer (Serkova et al, 2008). Control levels of citrate were 353.2 (125.9 to 764.5) µmol/mL, those of myoinositol were 21.2 (7.7 to 41.9) µmol/mL, and those of spermine were 58.0 (18.9 to 168.2) µmol/mL. In cancer, citrate levels were significantly reduced whereas those of myoinositol and spermine were significantly increased.
In relation to other body fluids the seminal plasma is unusual because of its high concentrations of potassium, zinc, citric acid, fructose, phosphorylcholine, spermine, free amino acids, prostaglandins, and enzymes (most notably acid phosphatase, diamine oxidase, β-glucuronidase, lactate dehydrogenase, α-amylase, PSA, and seminal proteinase).
Citric Acid
One of the major anions in human seminal plasma is citrate (mean, 376 mg/dL), which is present in the range of 20 mM or 60 mEq/L. This is compared with the chloride ion (155 mg/dL) at 40 mM. Citrate is a potent binder of metal ions, and the seminal plasma concentration of citrate, 20 mM, is comparable to that of the total divalent metals at 13.6 mM (calcium, 7 mM; magnesium, 4.5 mM; zinc, 2.1 mM). Prostatic citrate levels approximate 15.8 mg/mL (Zaneveld, 1981), and the values for seminal vesicle citric acid secretions are almost 100-fold less, being only 0.2 mg/mL. Citric acid is formed in the prostate at 100 times higher concentration than is seen in other soft tissues (e.g., prostate tissue, 30,000 nmol/g; other tissues, range of 150 to 450 nmol/g). The concentration of citrate in the ejaculate is 500 to 1000 times higher than that in the plasma. Prostate secretory epithelial cells form citrate from aspartic acid and glucose. The high concentrations within the prostate result partly from the inability of the prostate cell mitochondria to oxidize citrate readily once it is formed; therefore, the rate of citrate synthesis far exceeds the rate of citrate oxidation (Costello and Franklin, 1989, 1994). Kavanagh (1994) measured citrate and isocitrate levels, which are catalyzed by aconitase, and demonstrated ratios of 33 : 1 in the prostate, whereas other tissues demonstrate ratios of 10 : 1. This decreased activity of aconitase might explain the high levels of citrate within the prostate.
Diamine oxidase, an enzyme that degrades polyamines within the prostate, has been linked to citric acid concentrations and, indirectly, to sperm motility and fertility (Gonzalez, 1994; Le Calvé et al, 1995). Yacoe and associates (1991) used magnetic resonance spectroscopy to investigate the relationship between citrate metabolism and prostate cancer and demonstrated small but statistically insignificant differences between normal epithelium and prostate cancer cell lines. The relationship between prostatic inflammatory disease and citric acid has also been investigated (Wolff et al, 1991). In addition, it is now possible to accurately quantify the concentration of citrate within the prostate by proton magnetic resonance spectroscopy (Liney et al, 1996, 1997; Lowry et al, 1996; Lynch and Nicholson, 1997). Most recently, the ratio of citrate to other components of prostatic secretions has been used to diagnose prostate cancer (Costello and Franklin, 1989; Kim et al, 1998; Pucar et al, 2005).
Fructose
The source of fructose in human seminal plasma is the seminal vesicles (Mann, 1981). Patients with congenital absence of the seminal vesicles also have an associated absence of fructose in their ejaculates (Phadke et al, 1973). The seminal vesicle secretion contains smaller amounts of other free sugars such as glucose, sorbitol, ribose, and fructose, and these sugars usually amount to less than 10 mg/dL. In comparison, the concentration of the reducing sugar fructose is approximately 300 mg/dL in human seminal secretion, and it has a level of 200 mg/dL in seminal plasma.
Fructose levels are under androgenic regulation, but many factors, such as storage, frequency of ejaculation, blood glucose levels, and nutritional status, can also affect seminal plasma concentration (Mann, 1981); these considerations may account for the wide variations encountered in different semen samples from the same patient. Furthermore, plasma levels of androgens do not always correlate with seminal plasma fructose levels; therefore these levels are not a reliable index of the androgenic state of the subject. Seminal fructose levels have also been proposed to be under sympathetic control (Lamano-Carvalho et al, 1993; Kempinas et al, 1995). The physiologic role of fructose in seminal vesicle secretion has been indirectly linked to forward sperm motility through prostasome function (Fabiani et al, 1995) and to seminal viscosity (Gonzalez, 1993).
The source of fructose in seminal vesicles appears to proceed from glucose by aldose reduction to sorbitol and a subsequent ketone reduction to form fructose. The fructose of the seminal plasma appears to provide an anaerobic and an aerobic source of energy for the spermatozoa (Mann, 1981). The cervical mucus has high concentrations of glucose and very low levels of fructose, and the sperm are capable of using both types of sugars. In an epidemiologic study conducted among nearly 50,000 participants in the Health Professionals Follow-up Study of Cancer in 1986, fructose consumption (70 vs. 40 g/day) and high fruit intake were identified as protective against the development of advanced prostate cancer (Giovannucci et al, 1998).
Polyamines
Polyamines are the most basic (positively charged) small organic molecules in nature. They occur ubiquitously in tissues at high concentrations and are believed to be involved in diverse physiologic processes that share a relationship to cell proliferation and growth. Indeed, polyamines can serve as growth factors for cultured mammalian cells and bacteria and as inhibitors of enzymes, including protein kinases.
The exact role of polyamines at the molecular level still eludes science, but they represent important biologic compounds and are found at high levels in the ejaculate. Polyamines may affect the gating and transport of substances through membrane channels. From a clinical perspective, polyamines (spermidine and spermine) have been investigated as markers of androgen deprivation therapy among men with advanced-stage prostate cancer by Cipolla and colleagues (Cipolla et al, 1994). Other researchers (Heston, 1991; Kadmon, 1992; Madhubala and Pegg, 1992; Love et al, 1993) have investigated the role of polyamines in the pathophysiology of prostate cancer. The first and rate-limiting step in polyamine synthesis within the prostate is controlled by the enzyme ornithine decarboxylase (ODC). ODC gene expression has been demonstrated to be increased in BPH tissue (Liu et al, 2002). ODC can be inhibited by difluoromethylornithine (DMFO), which in turn inhibits polyamine synthesis. DMFO has been proposed as an agent for chemoprevention of prostate cancer (Kadmon, 1992).
Spermine levels in normal human seminal plasma range from 50 to 350 mg/dL and originate primarily from the prostate gland, which is the richest source of spermine in the body. Spermine [NH2-(CH2)3-NH-(CH2)4-(CH2)4-NH-(CH2)3-NH2] is a basic aliphatic polyamine and, because of its four positive charges, binds strongly to acidic or negatively charged molecules such as phosphate ions, nucleic acid, and phospholipids. When semen is allowed to stand at room temperature, acid phosphatase enzymatically hydrolyzes seminal phosphorylcholine to form free inorganic phosphate ions, which then interact with the positively charged spermine and precipitate as large, translucent salt crystals of spermine phosphate. Polyamines can also form amide bonds and make their covalent addition to protein carboxylic groups (Williams-Ashman, 1975), and this modification may be involved in regulatory function.
There has been much interest in spermine and other related polyamines, such as spermidine and putrescine, because of the rapid and dramatic changes in levels and ratios associated with many types of cells that have been induced into growth. Williams-Ashman and colleagues have investigated in detail the biosynthesis and regulation of polyamines in the male reproductive tract and have characterized the enzymatic reactions that progress from ornithine to putrescine to spermidine to spermine (Williams-Ashman et al, 1969, 1972; Williams-Ashman, 1975). Polyamines are oxidized enzymatically by diamine oxidase (present in the seminal plasma) to form highly reactive aldehyde compounds that can be toxic to both sperm and bacteria (Le Calvé et al, 1995). The formation of these aldehyde products produces the characteristic odor of semen. It is also possible that these aldehydes or polyamines may, themselves, protect the genitourinary tract from infective agents. Relationships between spermine levels in seminal plasma and sperm count and motility have also been suggested (Stamey et al, 1968; Fair et al, 1993; Fair and Parrish, 1981; Le Calvé et al, 1995). Like citrate, spermine can also be quantified within prostate tissue by magnetic resonance spectroscopy (van der Graaf et al, 2000).
Phosphorylcholine
Other positively charged amines are at high concentrations in the ejaculate, including choline and phosphorylcholine, which are usually found as components of lipid or as lipotropic factors. The semen of mammals is rich in choline [(CH3)3-N+-(CH2)2-OH]. In humans, phosphorylcholine predominates, whereas in most other species much higher levels of α-glycerylphosphorylcholine are present, often exceeding 1 g/dL of seminal plasma. Seligman and associates (1975) have demonstrated that phosphorylcholine is a highly specific substrate for prostatic acid phosphatase, which is also active in seminal plasma. The result of this enzymatic activity is the rapid formation of free choline in the first ejaculate. In contrast, α-glycerylphosphorylcholine is secreted primarily in the epididymis and is not readily hydrolyzed by acid phosphatase. For these reasons, Mann and Mann (1981) have suggested that the level of α-glycerylphosphorylcholine can be used as an index for assessing the contribution of the epididymal secretion to the ejaculate. The secretion from the epididymis is also under androgenic control. The function of these choline compounds is unknown; it appears that they are not metabolized by spermatozoa, nor do they affect the respiration of the sperm (Dawson et al, 1957).
Prostaglandins
The richest sources of prostaglandins in the human are the seminal vesicles (Pourian et al, 1995). Prostaglandins are present in seminal plasma at a total concentration of 100 to 300 µg/mL. Von Euler (1934) proposed the name prostaglandins for the active components in seminal plasma in the belief that they originated from the prostate gland, but Eliasson (1959) established that the primary source of prostaglandin is the seminal vesicles not the prostate; however, the original name has survived. Prostaglandins have a wide distribution in mammalian tissues but at much lower concentrations than in the seminal vesicles (Vane and Botting, 1995).
There are more than 90 different prostaglandins present in the human, with 15 prostaglandins present in human semen, and they are all 20-carbon hydroxy fatty acids with a cyclopentane ring having two side chains; as such, they are derivatives of prostanoic acid. The 15 types of prostaglandins within the prostate are divided into four major groups, designated A, B, E, and F according to the structure of the five-membered cyclopentane ring. Each of these groups is further subdivided according to the position and number of double bonds in the side chain (therefore, PGE3 indicates prostaglandins of E type with three double bonds in the side chain). The E group of prostaglandins is the major component in the male reproductive tract, whereas the F group predominates in the female system. Fuchs and Chantharaski (1976) have summarized the reported levels of human seminal plasma prostaglandins and report the following mean values (µg/mL): PGE1, 20; PGE2, 15; (PGE1 + E2) − 19-OH, 100; PGA1 + A2, 9; (PGA1 + A2) − 19-OH1, 31; PGB1 + B2, 18; (PGB1 + B2) − 19-OH, 13; PGF1α, 3; and PGF2α, 4.
These compounds are potent pharmacologic agents that have been implicated in a wide variety of biologic events in the male, including erection, ejaculation, and sperm motility and transport, as well as in testicular and penile contractions. In addition, prostaglandins from seminal fluid deposited in the vagina have been reported to affect cervical mucus, vaginal secretion, and sperm transport in the female genital tract. Chaudry and colleagues (1994) investigated the relationship between prostaglandin metabolism and both benign prostatic tissue and prostate cancer tissue. Prostaglandin E has been related to the immunosuppressive effects of seminal plasma mediated through the extracellular organelles, or “prostasomes” (Kelly et al, 1991). Finally, Olin and associates (1993) have investigated the effect of prostaglandins on fertility.
Cholesterol and Lipids
Scott (1945) reported that human seminal plasma contains 185 mg/dL of total lipids, 103 mg/dL of cholesterol, and 83 mg/dL of phospholipids (Vignon et al, 1992). In comparison, human prostatic secretion contains the following: total lipids, 186 mg/dL; cholesterol, 80 mg/dL; and phospholipids, 180 mg/dL. The lipids of semen have been further described (White, 1976), and the phospholipids of seminal plasma are composed of 44% sphingomyelin, 12.3% ethanolamine plasmalogen, and 11.2% phosphatidylserine (Poulos et al, 1973).
The reported levels of cholesterol in seminal plasma have varied considerably from 11 to 103 mg/dL (Scott, 1945; Eliasson, 1959; Poulos et al, 1973). White (1975) believes that the ratio of cholesterol to phospholipid in seminal plasma stabilizes the sperm against temperature and environmental shock. Thompson and associates (1987), Rohan and coworkers (1995), and Rose and Connolly (1992) have reviewed the role of dietary lipids in the pathogenesis of prostate cancer.
Zinc
The high level of zinc in human seminal plasma (140 µg/mL) appears to originate primarily from secretions of the prostate gland (488 ± 18 µg/mL) (Bedwal and Bahuguna, 1994). The prostate has the highest concentration of zinc (50 mg/100 g dry weight) of any organ. Mackenzie and colleagues (1962) reported that human seminal plasma contained 310 mg of zinc/100 g dry weight and that spermatozoa contained 200 mg/100 g dry weight. In comparison, prostatic secretions from eight normal subjects had 720 mg zinc/100 g dry weight. Byar (1974) has reviewed many of the early experiments and concepts related to zinc in the reproductive tract. Zinc levels are elevated or stable in BPH, whereas there is a marked decrease in zinc content associated with prostatic adenocarcinoma. The localization of zinc-65 in the human prostate by radioautography appears to be within the epithelial cells; however, in the lateral prostate of the rat, large quantities of zinc were also associated with the stroma and particularly with the basal membrane and the elastin protein component (Chandler et al, 1977). Oral intake of zinc does not alter zinc levels in prostatic fluid.
Many physiologic roles have been postulated for zinc since the classic studies of Gunn and associates (1956, 1965), who correlated endocrine effects on zinc uptake and concentration in the prostate of the rodent. There are many important zinc-containing metalloenzymes, but the concentration of zinc in the prostate probably exceeds that present in zinc-associated enzymes. Zinc is known to bind many proteins (Sansone et al, 1991). Johnson and associates (1969) characterized zinc-binding proteins in the prostatic secretion of the dog, on hydrolysis, as containing only eight types of amino acids. Heathcote and Washington (1973) described a zinc-binding protein in human BPH that was rich in histidine and alanine. Jonsson and colleagues (2005) suggested that one possible role of zinc in semen may be to regulate the activity of PSA by binding to semenogelins I and II and fragments thereof. There have been other studies on zinc-binding proteins from the prostate (Reed and Stitch, 1973; Fair et al, 1976), and additional information on these interesting proteins is needed.
An important role for zinc in prostatic secretion has been postulated in the studies of Fair and associates (1976), which suggest the direct role of zinc as a prostatic antibacterial factor. In the study of 36 normal men free from bacterial prostatic infections, the mean value of zinc in the prostatic secretion was approximately 350 µg/mL, with a wide range of 150 to 1000 µg/mL. In comparison, the prostatic fluid obtained from 61 specimens collected from 15 patients with documented chronic bacterial prostatitis had a reduction of more than 80% and averaged only 50 µg/mL, with a range of 0 to 139 µg/mL. The authors proposed a lower limit of normal at 150 µg/mL. In addition, in-vitro studies of free zinc ions at concentrations normally found in prostatic fluid have confirmed the bactericidal activity of zinc against a variety of gram-positive and gram-negative bacteria. However, a considerable portion of the zinc in the prostate appears to be bound to unique proteins, such as metallothionein, and it is not certain how this might alter the biologic properties of zinc (Suzuki et al, 1994, 1995). Yan and colleagues (2008) reported that assessment of decreased zinc in a normal human prostate epithelial cell (PrEC) in vitro resulted in increased single-strand DNA breaks (Comet assay) and differential expression of several genes (Affymetrix HG-U133A gene chips) involved in cell cycle progression, apoptosis, transcription, and DNA damage response and repair. Hence, a zinc deficiency may compromise DNA integrity in the prostate. In prostate cancer the ability of prostate cells to accumulate zinc is lost during disease progression and may be in part due to the genetic and epigenetic alterations produced by zinc depletion.
Prostatic Secretory Proteins
The predominant secretory proteins of the sex accessory tissues have been reviewed (Lilja and Abrahamsson, 1988; Aumuller and Seitz, 1990; Aumuller et al, 1990; Lilja, 1993a, 1993b; Rittenhouse et al, 1998; Saedi et al, 2001; Diamandis and Yousef, 2002; Yousef and Diamandis, 2002). High-resolution, two-dimensional electrophoresis profiles of the major secretory protein markers from human ejaculate, seminal plasma, and prostatic secretions have been reported (Edwards et al, 1981; Carter and Resnick, 1982; Rui et al, 1984; Tsai et al, 1984; Dube et al, 1987; Aumuller and Seitz, 1990); however, several are found in abundance and have clinical significance. These include PSA (human kallikrein 3 [hK3, protein; or KLK3, gene]); human kallikrein 2 (hK2 or KLK2); prostase/KLK-L1 (Yousef et al, 1999; Lwaleed et al, 2004; Clements, 2008); prostatic acid phosphatase; and prostate-specific protein (PSP-94), also called β-microseminoprotein (β-MSP). Table 90–5 lists some characteristics of the major secretory proteins in the sex accessory tissues.
Table 90–6 illustrates the number of sample, median, and ranges of components of prostatic secretions among normal male reproductive parameters.
Prostate-Specific Antigen
Major changes in the use of PSA have occurred since the 1980s; for a general overview, see the work of Polascik and coworkers (1999). A serine protease, PSA-like molecule was first demonstrated in human prostatic tissue in 1970 (Ablin et al, 1970), found in seminal plasma in 1971 (Hara et al, 1971), purified from prostatic tissue in 1979 (Wang et al, 1979), measured in the serum of men in 1980 (Kuriyama et al, 1980), and widely used as a clinical marker of prostate cancer by 1988 (Seamonds et al, 1986; Chan et al, 1987; Stamey et al, 1987; Oesterling et al, 1988). The historical information, molecular characteristics, physiologic properties, and immunoassay for PSA have been reviewed elsewhere (Oesterling, 1991, 1993; Shellhammer, 1993; Vessella and Lange, 1993; Partin and Oesterling, 1994; McCormack et al, 1995; Polascik et al, 1999).
The discovery of PSA resulted from a search of the ejaculate and prostatic fluid by immunoprecipitation to find specific proteins for forensic use. In 1971, Japanese workers isolated, from the seminal plasma, a protein that was proved to be antigenically specific for semen; they reported its chemical and physical characteristics and termed it γ-seminoprotein (Hara et al, 1971). Several years later, in an attempt to develop this protein further as a forensic marker for semen identification, γ-seminoprotein was purified from human seminal plasma. These seminal proteins, initially called γ-seminoproteins, have now been shown by sequence to be the same as PSA. Lilja and Abrahamsson (1988) reported the same proteolytic activity and site of glycosylation, the same molecular weight protein and gene sequence, and identical immunohistochemical characteristics and serum characteristics in these proteins.
Wang and coworkers (1979) first reported this human PSA that has now proved to be an important marker of the prostate and prostate disease. PSA is a glycoprotein acting as a serine protease of molecular weight 33 kD that contains 7% carbohydrate (Watt et al, 1986) and is found almost exclusively in the epithelial cells of the prostate (Armbruster, 1993; Rittenhouse et al, 1998). PSA was measured in the serum and was demonstrated to be a clinically important assay for monitoring prostate cancer (Kuriyama et al, 1980, 1981). For a detailed description of the use of PSA and its limitations, both clinically and in laboratory medicine analysis, see the work of McCormack and associates (McCormack et al, 1995; Rittenhouse et al, 1998; Polascik et al, 1999).
Watt and coworkers (1986) have studied PSA extensively and were the first to report its complete amino acid sequence. The single polypeptide chain contains 240 amino acids and an O-linked carbohydrate side chain attached to a serine residue. Lundwall and Lilja (1987) cloned the complementary DNA (cDNA) that encodes the PSA gene. Their study indicates that the mRNA of PSA in the prostate is approximately 1.5 kilobases.
PSA acts physiologically like a serine protease and an arginine esterase with both chymotrypsin-like and trypsin-like activity. The sequence of the protein is similar to that of other kallikreins (Rittenhouse et al, 1998) involved in prostatic cell regulatory mechanisms. Lilja (1985) and Watt and coworkers (1986) reported that one of the structural proteins of the seminal fluid, semenogelin, causes the ejaculate to clot. Semenogelin is the predominant seminal vesicle-secreted protein and one of the physiologic substrates for PSA. One possible biologic role of PSA is to lyse the clot in the ejaculate, but it is at present unknown why this clotting and lysing mechanism is important to reproductive physiology.
The PSA gene (hKLK3) is a member of a human tissue kallikrein gene family that includes hKLK1, hKLK2, hKLK3, and KLK-L1 (Lundwall, 1989; McMullen et al, 1991; Berg et al, 1992; Carbini et al, 1993; Clements, 1994; McCormack et al, 1995; Rittenhouse et al, 1998; Nelson et al, 1999; Yousef and Diamandis, 2003). To date there are more than 15 different human kallikreins, with expression noted in prostate, breast, ovarian, and testicular cancers (Obiezu and Diamandis, 2005). These genes are all located on chromosome 19 (Reigman, 1992; Yousef et al, 1999; Yousef and Diamandis, 2003). The ectopic expression of PSA has been reported in smaller concentrations in the tissue of malignant breast tumors (Yu et al, 1994a, 1994b, 1994c), normal breast tissue, breast milk, female serum, and adrenal and renal carcinomas; however, for practical and clinical purposes, PSA is an organ-specific but not a cancer-specific marker. A limitation of PSA as a tumor marker is demonstrated in the substantial overlap in values between benign and malignant prostate disease (Oesterling et al, 1988; Partin et al, 1990).
Most work regarding the molecular biology and biochemistry of PSA is based on extensive study of purified protein from seminal fluid in which the concentration of PSA is nearly a million-fold higher than that found routinely in serum (McCormack et al, 1995). The concentrations found in seminal plasma range from 0.5 to 5.0 mg/mL, whereas normal serum concentrations in men aged 50 to 80 years without prostatic disease range between 1.0 and 4.0 ng/mL (Catalona et al, 1991). Pre-pro-PSA (261 amino acids) is processed in the endoplasmic reticulum of prostatic epithelial cells, where a 17-peptide pre-region residue is cleaved. Seven more peptides are then cleaved from the propeptide to form the active PSA peptide (Rittenhouse et al, 1998). This pro-PSA represents an inactive (zymogen) precursor of PSA and is secreted and cleaved by hK2 (Lilja, 1985; Villoutreix et al, 1994; Rittenhouse et al, 1998).
The most exciting and clinically useful discovery in this area is the demonstration of different molecular forms of PSA (free [unbound] and complexed [bound]) circulating within the blood (Lilja et al, 1991; Stenman, 1991; Christensson et al, 1993; Lilja, 1993a; McCormack et al, 1995; Partin and Carter, 1996; Polascik et al, 1999). Complexed PSA is found in the serum irreversibly and covalently bound to α1-antichymotrypsin (ACT), an endogenous serine protease inhibitor. This form of complexed PSA (PSA-ACT) is enzymatically inactive yet has immunoreactivity. In addition, an unknown quantity of PSA is also complexed to α2-macroglobulin (PSA-A2M). Free PSA, found in lower concentrations than complexed PSA (PSA-ACT), is also enzymatically inactive, yet it is also immunoreactive, whereas PSA-A2M is not. Depending on the monoclonal antibodies used to measure serum PSA, various amounts of free and complexed PSA are recognized and contribute to the total (measurable) amount. Development of new monoclonal antibodies specific for free PSA and complexed PSA has allowed accurate measurement of the different molecular forms of PSA and their ratios. This has the potential of increasing the sensitivity and specificity of PSA for diagnosis of prostate cancer (Catalona et al, 1998).
Human Kallikrein 2
Human kallikrein 2 (hK2 [protein] or KLK2 [gene]) is a prostate-specific serine protease closely related to PSA and has largely been overlooked thus far (Rittenhouse et al, 1998). hK2 was first demonstrated from a low-stringency hybridization screen of a human liver genomic library in 1992, and the amino acid sequence is predicted to have 80% homology with PSA (hK3, KLK3) (Young et al, 1992). The striking homology between these two “prostate-specific” proteins suggested a close physiologic relationship. More recently, recombinant hK2 has been expressed and purified (Kumar et al, 1996; Mikolajczyk et al, 1998). Unlike PSA, hK2 is shown to be trypsin-like with selective cleavage at arginine residues and has a more potent (20,000-fold greater than PSA) protease activity (Mikolajczyk et al, 1998). Monoclonal antibodies to hK2 have been developed and have a low incidence of cross-reacting with PSA (Finlay et al, 1998). An exciting finding has been the independent demonstration that hK2 cleaves pro-PSA to generate the enzymatically active form of PSA (Kumar et al, 1996). Immunohistochemical studies have shown hK2 to be prostate localized and to increase in expression from normal to metastatic, poorly differentiated prostatic epithelium (Darson et al, 1997). Preliminary studies of hK2 in the serum of men with prostate cancer have suggested clinical utility for early detection of prostate cancer (Partin et al, 1999).
Human Kallikrein L1
Attempts to find other novel human kallikrein-like genes on chromosome 19 have identified yet another member of the human kallikrein gene family, KLK-L1 (Nelson et al, 1999; Yousef et al, 1999). Nelson and associates (1999) constructed a cDNA library enriched through subtraction with the cDNAs from four other normal tissues to yield an expressed sequence tag identifying a gene that they have called prostase. The sequence of prostase exhibits features similar to the other members of the kallikrein family. Yousef and coworkers (1999) also found KLK-L1 in breast tissue and demonstrated that it is hormonally regulated. Although the clinical utility of the members of the kallikrein gene family has not yet been determined, it is under investigation.
Human Kallikrein 11
Human kallikrein 11 (hK11) is a serine protease that shares similarities to human kallikrein 3 (hK3) or PSA with significant homologies at the levels of nucleotide and protein structure (Diamandis and Yousef, 2002). Localization of hK11 in epithelial cells of various organs has been demonstrated immunohistochemically and hK11 has been further detected in amniotic fluid, milk of lactating women, cerebrospinal fluid, follicular fluid, and breast cancer cytosols. The highest levels of hK11 were observed in prostatic tissue extracts and seminal plasma, in which it was present at 300-fold lower levels than PSA. Elevated serum levels of hK11 were found in 60% of men with prostate cancer; the ratio of hK11 to total PSA was able to reduce the number of biopsies required, and the data were similar to those gleaned from free PSA assays (Diamandis and Yousef, 2002; Nakamura et al, 2003).
Human Kallikrein 14
Human kallikrein 14, a trypsin-like human kallikrein-related peptidase (KLK), has been shown to exert a significant and dose-dependent effect on semen liquefaction (Emami et al, 2008; Emami and Diamandis, 2008). Liquefaction of human semen involves proteolytic degradation of the seminal coagulum and release of motile spermatozoa. Several members of human kallikrein–related peptidases are implicated in semen liquefaction, functioning through highly regulated proteolytic cascades. Among these, KLK3 (also known as prostate-specific antigen) is the main enzyme responsible for processing of the primary components of semen coagulum. KLK14 recently has been identified as a potential activator of KLK3 and other KLKs (Emami et al, 2008; Emami and Diamandis, 2008). Borgono and associates (2003) measured seminal plasma levels of KLK14 by an enzyme-linked immunosorbent immunoassay (ELISA) in 36 human semen samples and found KLK14 to be 0.6 to 23.6 µg/L (mean 10.8; median 10.7 µg/L). Semenogelin I and semenogelin II are degraded by PSA to form various biologically active peptides involved in semen liquefaction and release of motile spermatozoa. Semenogelins I and II, through chelation with the excess of free zinc, are also directly cleaved by KLK14, in the same manner as PSA. In addition, KLK14 has also been demonstrated to be a potential biomarker for ovarian and breast cancer (Borgono et al, 2003). KLK14 should be considered as another active molecule involved in semen liquefaction.
Prostate-Specific Transglutaminases
Human prostate-specific transglutaminase 4 belongs to a family of enzymes that irreversibly cross-link peptide-bound glutamine residues through reactions with either lysines or primary amines such as polyamines (Dubbink et al, 1998). Transglutaminases are located throughout the body, but they are highly tissue specific. Dubbink and coworkers (1998) described a new prostate-specific transglutaminase with 35-kilobase genomic DNA and consisting of 13 exons and 12 introns. The main transcription initiation site is located 52 base pairs upstream of the translational start code. At least one splice variant was described, and a transglutaminase 4 gene (TGM4) promoter was analyzed by sequencing and transfection experiments and found at −1276 to −563. Subsequently, an Sp1 binding site (promoter) required for basal activity of TGM4 was identified (Dubbink et al, 1999). The TGM4 promoter was characterized by deletion mapping and mutational analysis. These researchers determined that positions between −113 and −87 were essential for core activity of the promoter. The sequences identified are binding sites for the Sp1 and Sp3 transcription factors; however, their precise role in TGM4 regulation was not deduced from experiments described (Dubbink et al, 1999). Of importance is the fact that the major gel-forming proteins in semen, semenogelins I and II, are substrates for transglutaminase 4 (Peter et al, 1998). Esposito and Caputo (2005) reviewed the range of substrates for transglutaminases in detail and characterized the molecular basis of transglutaminase-catalyzed reactions and also assessed possible physiologic function and pathophysiologic processes due to such interactions. The transglutaminase for the prostate is transglutaminase 4; it weighs 77 kD, is androgen regulated, and is found extracellularly. Transglutaminases catalyze the post-translational modification of proteins by formation of polymerized cross-linkages between the γ-carboxamide group of protein-bound glutamine residues and the ε-amino group of protein-bound lysine residues, which results in a stabilized molecular complex. There is evidence to suggest that the biochemical affinity of transglutaminase 4 for acyl-type substrates such as kinesin proteins in protein secretions of the semen may be important for correct extrusion of transglutaminase 4 from the coagulating gland (Esposito and Caputo, 2005).
An and coworkers (1999) also described cloning of TGM4 (human prostate-specific transglutaminase) and its promoter in the elements of −1 to −500 and also at −520 to −1400. In addition, this group applied Northern blot hybridization and reverse-transcription polymerase chain reaction (RT-PCR) analysis to confirm prostate specificity and Gleason grade-specific expression by RT-PCR and noted significant downregulation in high Gleason grade as well as in metastatic tissue extracts. From a protein perspective, Birckbichler and colleagues (2000) revealed by quantitative immunofluorescence that prostate cancer was significantly decreased compared with normal prostate and prostatitis cases, but this is in contrast to what was observed with RT-PCR results (An et al, 1999), where the higher Gleason grade tumors tended to be significantly decreased. This discrepancy of results needs to be rectified in larger experiments comparing RT-PCR with protein expression to determine whether this is a technical problem or rather one of translation of transglutaminase 4 mRNA versus protein in the malignant disease process.
Semenogelins I and II
Semenogelin I and semenogelin II are dominant proteins in human semen coagulum that are degraded by PSA to form various biologically active peptides, which in combination with fibronectin give rise to the gel-like coagulum of newly ejaculated semen (Lilja, 1985; Malm et al, 1996; de Lamirande et al, 1997). The genes encoding semenogelins I and II are located in separate regions 11.5 kilobase pairs apart on chromosome 20. The major biologic function of semenogelin involves capacitation, which is defined as a series of changes in cell membranes, enzyme activities, and ion fluxes that sperm undergo as they traverse the female urogenital tract to reach the zona pellucida and fertilize the egg (de Lamirande et al, 1997). It has been demonstrated that biologically active peptides from semenogelin I and semenogelin II proteolysis scavenge superoxide anion and may affect sperm oxidase to serve as natural regulators of sperm capacitation (de Lamirande et al, 2001; de Lamirande, 2007). Semenogelins from the seminal vesicles and zinc ions from the prostate play a significant role in semen aggregation at the time of sperm ejaculation and also in sperm motility by binding to the sperm and then interacting with zinc (de Lamirande, 2007; Yoshida et al, 2008). It is of physiologic and possibly pathophysiologic importance that these major gel-forming proteins in semen, semenogelins I and II, are substrates for transglutaminase 4 (Peter et al, 1998). Both of these proteins originate from the glandular epithelium of the seminal vesicles and are produced in high concentrations; however, in the epididymis, only semenogelin I is expressed. There is evidence by immunohistochemistry that other cell types including the vas deferens, prostate, and trachea demonstrate strong signals for semenogelins I and II, and weaker but positive signals were seen in skeletal muscle cells and in the central nervous system (Lundwall et al, 2002).
Key Points: Secretory Proteins
Prostate-Specific Membrane Antigen
Recent reviews of the biochemistry and biology of the prostate-specific membrane antigen (PSMA) in human tissues and prostate cancer surveys describe the differential regulation of the molecule, its enzymatic functions, and its potential as a biomarker for in-vivo imaging and immunotherapy (Elgamal et al, 2000; Ghosh and Heston, 2004). The gene encoding PSMA is located on chromosome 11p11-12 and codes for a type II membrane glycoprotein (MW: ~100,000 daltons) with intracellular (1 to 18 amino acids), transmembrane (19 to 43 amino acids), and large extracellular (44 to 750 amino acids) domains (Israeli et al, 1994; Ghosh and Heston, 2004; Davis et al, 2005). The cDNA (2.65 kb GenBank Accession M99487) encoding PSMA was first reported by Israeli and colleagues in 1993 and its deduced amino acid sequence determined (Israeli et al, 1994). It encodes a 750-amino acid protein with a predicted molecular mass of 84 kD (excluding carbohydrates). The hydrophobic amino acids found on amino acid residues 20 to 43 suggested that this protein is a type II integral membrane protein with a small intracellular domain and a large extracellular domain (Fair et al, 1997). The promoter for PSMA has been cloned (Good et al, 1999), and PSMA has been expressed and purified from a baculovirus expression system (Lodge et al, 1999). A portion of the transmembrane domain of this protein (amino acid residues 1250 to 1700) shares 57% homology with the human transferrin receptor mRNA (Mahadevan and Saldanha, 1999). Alternative splicing variants of PSMA (PSM′-PSA′ extracellular domain protein) are under investigation to better understand the clinical significance of this important membrane protein found within the prostate (Liu et al, 1997; Grauer et al, 1998; Murphy et al, 1998; Ghosh and Heston, 2004; Rajasekaran et al, 2005). PSMA has been crystallized and its structure deduced at 3.5-Å resolution. These analyses reveal a homodimer with structural similarity to the transferrin receptor, a receptor for iron-loaded transferrin that lacks protease activity (Davis et al, 2005). However, unlike the transferrin receptor, the protease domain of PSMA (glutamate carboxypeptidase II) contains a binuclear zinc site, catalytic residues, and a proposed substrate-binding arginine patch.
PSMA in the central nervous system metabolizes the brain neurotransmitter N-acetyl-aspartyl-glutamate or NAAG (named NAALADase). In the intestine, PSMA is found in the proximal small intestine, where it removes γ-linked glutamates from poly-γ-glutamated folate (folate hydrolase 1), or as a carboxypeptidase, glutamate carboxypeptidase II. Another interesting potential targeting feature of PSMA expression in the prostate is that it is overexpressed in neovasculature of other tumors (Silver et al, 1997; Chang et al, 1999b, 2001). A PSM-like molecule has been located at 11q14.3, but only PSMA is overexpressed in prostate cancer. In the prostate there are three alternatively spliced variants of PSMA. However, only one of these isoforms (PSM′ located at the 5′ end of PSMA cDNA) is known to be differentially expressed in normal tissue, BPH, and prostate cancer (Elgamal et al, 2000; Rajasekaran et al, 2005). PSMA mRNA expression within prostate cancers is highest in the hormone-deprived state, contrary to PSA mRNA, which often demonstrates lower, even absent expression in the hormone-deprived state (Henttu et al, 1992; Israeli et al, 1994; Wright, 1995; Rajasekaran et al, 2005).
PSMA is hormonally controlled by steroids similar to PSA (Israeli et al, 1994; Elgamal et al, 2000). A study by Su and colleagues (Su et al, 1995) using RT-PCR on peripheral blood demonstrated that the PSMA : PSM′ ratio is threefold to sixfold upregulated in prostate cancer compared with BPH (0.76 to 1.6) and normal (0.075 to 0.45). Also, Elgamal and associates (2000) assessed the overall RT-PCR assay results described for six separate RT-PCR peripheral blood studies; the data yielded a pooled sensitivity of 66% for RT-PCR PSMA versus 62% for RT-PCR PSA, suggesting inadequate sensitivity for routine clinical use.
Today, only a few studies have investigated PSMA protein levels in the blood of patients with prostate disease in spite of numerous publications regarding the production of numerous new monoclonal antibodies made to PSMA (Chang et al, 1999a; Tino et al, 2000). The existing assays can be characterized as research assays only, and the clinical data represent preliminary testing and not validation for a new clinical assay for assessment of diagnostic clinical performance. Reports to date used Western slot blot, a competitive ELISA, and more immuno–surface-enhanced laser desorption/ionization (immuno-SELDI). A competitive ELISA developed by Horoszewicz and colleagues (1987) using 9h10-A4 and 7E11-C5 monoclonal antibodies found an increased PSMA level in 47% of patients (20 of 43) with prostate cancer versus only 5% of patients (3 of 66) without prostate cancer, and the result was negative in 30 normal blood donors. In addition, PSMA expression within cancerous lesions appears to correlate directly with the degree of differentiation and not with tumor stage (Wright, 1995). Other investigators had employed a competitive ELISA and Western blotting and confirmed increasing expression with higher grade and stage, thus implicating PSMA in recurrence and progression (Rochon et al, 1994; Douglas et al, 1997; Murphy et al, 1997). A significant effort has been made to demonstrate tissue expression by quantitative immunoassays in the LNCaP cell line, human prostate cancer, and normal or BPH tissues as well as in metastatic tissue, seminal fluid, and urine (Su et al, 1995; Troyer et al, 1995; Sokoloff et al, 2000; Ross et al, 2003), but none of these assays was developed into serum immunoassays. However, Xiao and colleagues (2001) reported the use of an immuno-SELDI assay for PSMA. The immuno-SELDI assay used the 7E11-C5 immunoglobulin G1 monoclonal antibody developed by Horoszewicz and colleagues (1987) and employed in the clinically available ProstaScint scans. In that study, the ProteinChip array was coated with 1 µg of G protein and then residual active sites were blocked with 1 M ethanolamine, washed, and treated with 1.5 µg 7E11-C5 monoclonal antibody (Xiao et al, 2001). The assay was formatted to create a 96-well array, and the same clinical samples were processed that were run with Western blotting (Beckett et al, 1999; Elgamal et al, 2000). The results of the immuno-SELDI assay clearly revealed that serum PSMA with the 7E11-C5 antibody differentiated prostate cancer (623.1 ng/mL; n = 17) from BPH (117.1 ng/mL; n = 10, P < .001). The authors showed considerable age-related overlap in serum activity in normal subjects and patients with BPH older than age 50 years. The knowledge gained from these efforts about PSMA molecular characterization and diagnostics has been very valuable and has set the stage to apply new and innovative methods to manage prostate cancer. The potential use of PSMA as a novel prostate-specific targeting medical device based on PSMA-substrates, PSMA-peptides, PSMA-RNA aptamers, and anti-PSMA antibodies is being tested for targeted drug delivery and medical imaging (Lupold and Rodriguez, 2004; Davis et al, 2005; Chandran et al, 2008).
Finally, a current review of the multifunctional potential of the PSMA molecule by Rajasekaran and colleagues (2005) indicates that PSMA (type II membrane glycoprotein) dimerization is similar to transferrin receptor and may function as a receptor for internalizing a putative ligand. PSMA enzyme activities (NAALADase and folate reductase) are consistent with a role in nutrient uptake. The PSMA peptidase activity may be involved in signal transduction in prostate epithelial cells and may activate cascades that result in cell survival, cell proliferation, and cell migration functions. This multifunctional molecule not only has numerous physiologic benefits but also has several diagnostic and therapeutic potential benefits to manage prostate cancer as well.
Prostate Stem Cell Antigen
Reiter and coworkers (1998) identified prostate stem cell antigen (PSCA), a cell surface antigen that is expressed in the prostate (among other tissues including bladder). The PSCA gene encodes a 123-amino acid glycoprotein, with 30% homology to stem cell antigen 2 (Sca-2). Like Sca-2, PSCA is a member of the Thy-1/Ly-6 family and is anchored by a glycosylphosphatidylinositol linkage. By use of mRNA in-situ hybridization, PSCA expression was localized in normal prostate to the basal cell epithelium, the putative stem cell compartment of prostatic epithelium; hence, PSCA may be a marker of prostate stem/progenitor cells. Hara and associates (2002) performed an analysis of PSA, PSMA, and PSCA mRNA level on peripheral blood by RT-PCR in 58 cases of prostate cancer and 71 cases of nonmalignant disorders. The results were 7 of 58 (12.1%) for PSA, 12 of 58 (20.7%) for PSMA, and 8 of 58 (13.8%) for PSCA; zero samples were positive for nonmalignant diseases. A summary of prognostic value for the three biomarkers was a hierarchy of PSCA > PSA > PSMA for RT-PCR of the 58 patients with prostate cancer. Note that in this group of patients, when the RT-PCR result was positive for PSCA the patients had a lower disease progression-free survival than with the other two biomarkers. The PSCA expression increased with higher Gleason score and cancer stage as well as with progression to metastasis and may be a useful biomarker for staging of prostate cancer (Hara et al, 2002). Han and associates (2004) performed immunohistochemistry analysis of PSCA by a 246-patient tissue microarray; the results revealed that a PSCA staining intensity of 3.0 correlated with adverse prognostic features including Gleason score of 7.0 (P = .001), seminal vesicle invasion (P = .005), and capsular involvement (P = .033). However, after multivariate analysis, PSCA did not hold up as an independent predictor of PSA recurrence. Zhigang and Wenlv (2004) studied BPH, low-grade prostatic interepithelial neoplasia (LGPIN), high-grade prostatic interepithelial neoplasia (HGPIN), and prostate cancer at the tissue level by immunohistochemistry and at the mRNA level by in-situ hybridization. In BPH and LGPIN the staining of PSCA protein and mRNA was weak or negative and less intense and uniform than in HGPIN and prostate cancer. There was moderate to strong PSCA protein as well as mRNA expression in 8 of 11 (72.7%) HGPIN and in 40 of 48 (83.4%) prostate cancer specimens that were examined by immunohistochemistry and in-situ hybridization analyses. When the prostate cancer specimens examined by immunohistochemistry and in-situ hybridization analyses were compared with BPH (20%) and LGPIN (22.2%) samples, the results were statistically significant (P < .05, respectively). The expression level of PSCA increased with high Gleason grade, advanced stage, and progression to androgen independence (P < .05, respectively). In addition, in this study, protein immunostaining and in-situ hybridization mRNA stain showed a high degree of correlation between PSCA protein and mRNA overexpression in prostate cancer, supporting the potential of PSCA as a prognostic biomarker. Clearly, the value of this protein to the biology of prostate epithelial tissue morphogenesis and also as a new biomarker for diagnosis and treatment of prostate cancer is yet to be realized.
The potential use of PSCA, a membrane surface antigen that is highly expressed in the prostate, as a novel prostate targeting medical device for diagnostics (blood immunoassays or medical imaging) and therapy (vaccines or immunotherapy) is currently being actively studied (Olafsen et al, 2007; Raff et al, 2009). Here is another unique opportunity to utilize prostate developmental knowledge as a possible tool to manage prostate cancer because of its amplification during carcinogenesis.
Prostatic Acid Phosphatase
Acid phosphatase activity is more than 200 times more abundant in prostate tissue than in any other tissue and is the source of the high levels of acid phosphatase in ejaculate. Phosphatase enzymes hydrolyze many types of organic monophosphate esters to yield inorganic phosphate and alcohol. Many phosphatase enzymes exhibit optimal activity in vitro in the acid (pH 4 to 6) or alkaline (pH 8 to 11) ranges and are thus classified broadly as either acid or alkaline phosphatase.
Acid phosphatase activity may be further defined by factors that inhibit its enzymatic activity. For example, erythrocyte acid phosphatase is particularly sensitive to inhibition by 0.5% formaldehyde or copper ions (0.2 mM) whereas prostatic acid phosphatase (PAP) activity is far more sensitive to inhibition by fluoride ions (1 mM) or L-tartrate (1 mM).
Osteoclasts are also a rich source of tartrate-insensitive acid phosphatase. Minor elevations in serum acid phosphatase levels can accompany Paget disease, osteoporosis, nonprostatic bone metastasis, and other conditions of increased bone resorption as well as metastatic prostate cancer. All acid phosphatases hydrolyze a wide range of natural and synthetic phosphomonoesters, and this has provided a wide variety of assay systems and the expression of different units of activity, depending on the assay. These synthetic substrates include, in part, phenylphosphate (Gutman, 1938); phenolphthalein phosphate; paranitrophenyl phosphate, also called Sigma 104; and thymolphthalein phosphate (Roy et al, 1971). The specificity of these substrates varies with the type and source of acid phosphatase; it appears that thymolphthalein phosphate may be the most specific substrate for assaying serum levels of prostate-specific acid phosphatase, but specific antibodies are now available for immunoassays. Interest in acid phosphatase assays in serum as a measure of prostatic cancer metastasis before definitive therapy has decreased with the availability of the more sensitive and specific PSA assay (Burnett et al, 1962).
The natural substrate for PAP may be phosphorylcholine phosphate, which is rapidly hydrolyzed in the semen (Seligman et al, 1951). The biologic functions of this enzyme and its reactions are not known, but it is of interest that PAP can hydrolyze protein tyrosine phosphate esters, natural products of many oncogene protein tyrosine kinases (Li et al, 1984; Lin and Clinton, 1986). By magnetic resonance spectroscopic techniques, it has been shown that the ratio of intracellular choline to citrate levels within the prostate can help differentiate normal from cancerous prostate tissue (Scheidler et al, 1999). Further clinical testing is required before this finding will influence clinical practice. It is unknown whether acid phosphatase is a regulatory factor in the tyrosyl protein kinase systems so essential as signaling mechanisms in growth factor function.
Human PAP is a glycoprotein dimer of molecular weight 102,000 and contains about 7% carbohydrate by weight, composed of 15 residues per mole of neutral sugars (fructose, galactose, and mannose), 6 residues per mole of sialic acid, and 13 residues of N-acetylglucosamine (Chu et al, 1977). The protein can be dissociated into two subunits of 50 kD. The activity of the purified human enzyme is 723 U/mg with α-naphthyl phosphate, and the seminal plasma contains 0.3 to 1 g/L or 177 to 760 U/mL. The high enzymatic activity of PAP is not characteristic of accessory tissues in many other species; the level is 1000 times higher per gram of tissue in the human prostate than in the rat prostate. The clinical aspects of PAP were reviewed by Romas and Kwan (Lowe and Trauzzi, 1993; Romas and Kwan, 1993).
Prostate-Specific Protein 94 (β-Microseminoprotein and β-Inhibin)
A major, cysteine-rich, nonglycosylated 16-kD protein that contains 94 amino acids has been found in prostatic secretions and named prostate-specific protein 94 (PSP-94); it is one of the three predominant proteins secreted in the prostate glands and found in seminal fluids along with PSA and PAP. This protein had previously been designated β-inhibin and also β-microseminoprotein (Dube et al, 1987; Ulvsback et al, 1989). Transcripts of mRNA for this protein have also been identified in nongenital tissues (Ulvsback et al, 1989). The human gene for PSP-94 has been mapped to chromosome 10 (q11.2) and there are three glucocorticoid response elements and one estrogen response element in the promoter region of the first intron. Based on these observations the gene is likely regulated by hormones in humans (Nolet et al, 1991; Ochiai et al, 1995) because this also was reported in studies of the rat lateral prostate (Kwong et al, 2000). Also, Valtonen-Andre and associates (2008) demonstrated that in young, healthy males the PSP-94 levels in the serum correlate well with those in the seminal plasma (r = 0.50, P < .001). An automated immunoassay was performed with an Autodelphia 1235 (Wallac) and produced median values of PSP-94 in 205 young men that were 12 mg/L (2.5 to 97.5 percentile, 4.9 to 26 mg/L) in serum and 0.53 g/L (2.5 to 97.5 percentile, 0.13 to 2.0 g/L) or 1.8 mg (2.5 to 97.5 percentile, 0.32 to 6.6 mg) in seminal plasma. These data provide a solid basis for evaluation of this biomarker both in healthy men and those with prostate cancer.
One of the main biologic functions of PSP-94 is the inhibition of follicle-stimulating hormone (Garde et al, 1999). Whereas follicle-stimulating hormone is made by the pituitary gland, the prostate has been shown to be an extrapituitary source of follicle-stimulating hormone. There are follicle-stimulating hormone receptors in the prostate, and it appears that an autocrine or paracrine regulation of this hormone influences prostate epithelial proliferation (Ben-Josef et al, 1999; Porter et al, 2001). Also, Chan and colleagues (1999) used in-situ hybridization to study expression of PSP-94 in human prostates. They found that fetal prostate at 6 to 7 months synthesizes PSA and PAP but not PSP-94, and this observation appears to relate to the development of the prostate gland. Zonal anatomic distribution of PSP-94 in the adult prostate demonstrated that the protein is expressed mostly in the acini of the peripheral zone rather than the central or transitional zones. Recently, Anahi Franchi and associates (2008) studied the PSP-94 and its potential interaction with human spermatozoa and its possible role in fertility. Using purified PSP-94, they demonstrated a specific interaction at the sperm surface. Also, employing a dual antibody ELISA technology the authors noted that of 62 patients being assessed for fertility fertile men had a lower concentration of the protein than subfertile men (OT, ST, and AS) and suggested that semen quality may be affected by the concentrations of the PSP-94. Another function of the PSP-94 may be to interact directly with spermatozoa in a manner that can impact the quality of sperm structure and function.
In the area of cancer, Chan and coworkers (1999) found that PSP-94 expression is markedly downregulated with increasing Gleason grade of prostate cancer. Furthermore, Shukeir and coworkers (2003) demonstrated a significant decrease in growth of the highly metastatic Dunning R3327 subline MatLyLu rat prostate model transfected with parathyroid hormone–related protein by treatment with varying doses of commercial PSP-94 purified from human seminal plasma (0, 0.1, 1.0, and 10 µg/kg/day). Serum levels of parathyroid hormone–related protein and calcium were used to monitor the efficacy of treatment with PSP-94. Hence, PSP-94 is an effective inhibitor of hormone-independent, late-stage prostate cancer metastasis in this Dunning MatLyLu animal model. The PSP-94 molecule has not yet been crystallized; however, Joshi and Jyothi (2002), in a computer-simulated molecular model, have predicted its structure and calculated its binding activity and related biologic activity (follicle-stimulating hormone inhibition) and immunogenic properties. Using three-dimensional structure constructed by nuclear magnetic resonance (NMR), Ghasriani and associates (2006, 2009) have shown the PSP-94 molecule to consist of two distinct domains that form a rather extended structure. The two domains are connected to each other by the peptide backbone, one disulfide bond, and interactions between the amino and carboxyl termini and are oriented to give the molecule a rather extended structure. In addition, Ghasriani and associates (2009) have demonstrated the specific molecular interactions of the PSP-94 with cysteine-rich secretory protein 3 (CRISP-3) applying multidimensional NMR. The CRISP proteins are ubiquitous among organisms and snake venoms, and they are reported to be calcium ion channel blockers; however, the relevance of these observations of protein-protein interactions to seminal plasma is yet to be determined.
Protein C Inhibitor
Human semen contains several enzymes and inhibitors of the hemostatic coagulation system (Lwaleed et al, 2004; Fernandez and Heeb, 2007). In human semen PSA exists as a molecular complex with protein C inhibitor (PCI), and the latter provides some inhibitory consequences for actions of PSA. The predominant structural proteins of coagulated semen are those proteins secreted by the seminal vesicle including semenogelins I and II and fibronectin, and these proteins remain stable in the seminal vesicle’s secretions for up to 20 hours at 37°C but rapidly cleave into small peptides on mixing with the proteases (e.g., PAP, hKLK2 [PSA], hKLK3, hKLK14) of prostatic secretion (Lwaleed et al, 2004; Fernandez and Heeb, 2007). The human PCI gene is located in chromosome 14q32.1 and is a serine protease inhibitor that corresponds to a region containing the genes of related serpins (SERPINA5) (Suzuki et al, 1987; Fernandez and Heeb, 2007; Suzuki et al, 2007). PCI is a heparin-dependent inhibitor of activated protein C (APC) that is immunologically and functionally identical to a heparin-dependent urokinase inhibitor (plasminogen activator inhibitor type 3). PCI also inhibits several other blood coagulation and fibrinolytic factors (e.g., FXa, FXI, plasma kallikrein) (Lwaleed et al, 2004; Espana et al, 2007; Fernandez and Heeb, 2007; Suzuki et al, 2007). Suzuki and associates (2007) also demonstrated that digestion of human seminal coagula with PSA releases PCI and PSA-PCI complex from the coagula into the soluble phase, suggesting the presence of active PCI within the semen coagula. PCI then forms a “ternary protein complex” with PSA and semenogelin II in the seminal plasma. The binding of semenogelin II to PSA and PCI is influenced by the molecular microenvironment, including pH, ionic strength, heparin, negatively charged dextran sulfate, divalent cations, and particularly by zinc. These observations suggest that binding of PCI to semenogelins in seminal vesicles regulates the PSA-catalyzed degradation of semenogelins in seminal plasma; the complex formation among PCI, PSA, and semenogelins is modulated by several factors in seminal plasma. Espana and colleagues (2007) determined that PCI is secreted at very high levels in the seminal vesicles in an active form and also occurs in high concentrations in the seminal plasma. The concentration of PCI in 40 seminal plasma samples ranged from 2.2 to 3.7 mM (i.e., about 220 mg/L), and 45% of the seminal PCI was functionally active when assayed immediately after ejaculation. Notably, infertile men had significantly decreased seminal PCI levels (0.6 to 3.2 mM). However, the concentration of PSA in seminal plasma far exceeds the capacity of PCI to inhibit this molecule and hence the biologic role of PCI in seminal plasma. Espana and colleagues (2007) utilized purified PCI to assess several functional aspects of PCI, and evidence indicated that PCI is involved in human reproduction at several key steps, including fertilization. Hence, PCI is abundant in the seminal fluid and it plays a key role in the interaction among semenogelins, PSA, and likely other proteins in semen, resulting in protein-protein interactions critical to semen coagulation and liquefaction. A balance of seminal fluid coagulative proteins, active enzymes, and metabolites is required to effect sperm motility and successful fertilization (Lwaleed et al, 2004; Espana et al, 2007; Fernandez and Heeb, 2007; Suzuki et al, 2007).
Leucine Aminopeptidase
Aminopeptidases hydrolyze the amino-terminal amino acid from small polypeptides. Leucine aminopeptidases are particularly active against the substrate L-leucyl-glycine, and some of these enzymes are referred to as arylamidases because the optimal substrate is L-leucyl-β-naphthylamine. The human prostate is rich in the arylamidase type of leucine aminopeptidase, with a presence in prostatic fluid of 30,000 units/mL.
Leucine aminopeptidase is a product of the epithelial cells of the prostate (Niemi et al, 1963) and is secreted into the lumen of the acini (Kirchheim et al, 1964; Vafa et al, 1993). Rackley and associates (1991) demonstrated that extracts from prostatic carcinoma contained less leucine aminopeptidase activity than did tissue obtained from BPH.
Lactate Dehydrogenase
The isoenzyme ratios of lactate dehydrogenase (LDH) in human semen may be altered in a patient with prostate cancer (Oliver et al, 1970; Grayhack et al, 1977). LDH (MW: 150 kD) is composed of four subunits (each of 35 kD) of only two different types of proteins, denoted M and H. The LDH of muscle has four M units, and that of heart has four H units. Five isoenzymes of LDH can be found in tissues with a four-subunit composition as follows: LDH I, MMMM; LDH II, MMMH; LDH III, MMHH; LDH IV, MHHH; and LDH V, HHHH. The M and H subunits appear to be the same in all tissues, but the amounts of LDH I to V can vary. Denis and Prout (1963) observed increased levels of LDH IV and V in prostatic cancer tissue. Several investigators have observed elevated ratios of LDH V/LDH I in human prostatic cancer (Elhilali, 1968; Oliver et al, 1970; Flocks and Schmidt, 1972).
Immunoglobulins, C3 Complement, and Transferrin
There are many reports establishing the presence of immunoglobulins (Igs) in human seminal plasma (Liang et al, 1981; Gahankari and Golhar, 1993). It is possible to measure levels of IgG from 7 to 22 mg/dL and those of IgA from 0 to 6 mg/dL; however, IgM is at low, often undetectable levels (Friberg, 1976). The complete source of these antibodies is not known, although they are found in expressed prostatic fluid (Grayhack et al, 1979) and may be related to infections (Fowler et al, 1982). They are usually found at lower levels in seminal plasma than in blood, but the possibility of diffusion across the “blood–seminal plasma barrier” has not been eliminated (see discussion by Friberg, 1976).
Expressed prostatic fluid contains considerable amounts of the C3 component of complement, present at 1.82 mg/dL, and this increases nearly 10-fold in fluid collected from patients with prostatic adenocarcinoma to levels of 16.9 mg/dL (Grayhack and Lee, 1981). Prostatitis has also been shown to be related to C3 among men with chronic prostatitis (Blenk and Hofstetter, 1991). Prostatitis and BPH increase the level only approximately twofold. In the same manner, transferrin, an iron-carrying protein, is increased, going from levels of 5.3 mg/dL in normal prostatic fluid to 42.4 mg/dL in prostatic carcinoma (Grayhack and Lee, 1981).
John and colleagues (2003) conducted a prospective study of the ejaculate of 88 patients with chronic prostatitis by surveying IgG, IgA, and IgM and interleukin-1α, soluble interleukin-2 receptor, and interleukin-6. The control group consisted of 96 normal ejaculates according to the World Health Organization criteria. Ejaculates of patients with chronic prostatitis increased during symptoms and subsided when clinical symptoms decreased. The authors observed that a combination of the humoral immune (IgA and interleukin-6) changes and T-cell–rich infiltrates is suggestive of an autoimmune component of the disease. Alexander and coworkers (2004) are studying a group of patients with chronic granulomatous prostatitis consisting of histologically diffuse nonspecific inflammatory changes that include epithelioid histiocytes and occasional multinucleate giant cells admixed with lymphocytes and plasma cells. They have identified an association between the major histocompatibility locus antigen HLA-DRB2*1501 and granulomatous prostatitis and have suggested the possibility that it may be an autoimmune disease.
Zinc α2-Glycoprotein
In the seminal plasma, zinc α2-glycoprotein (ZAG) is synthesized by prostate epithelial cells and secreted into seminal fluid (Ding et al, 2007), and it constitutes about 30% of the proteins present in the seminal fluid (Poortmans and Schmid, 1968). The ZAG glycoprotein is found in many body fluids with a molecular mass of 41 kD, and the crystal structure is quite similar to that of a class I major histocompatibility complex (Burgi and Schmid, 1961; Burgi et al, 1989; Sanchez et al, 1999; Delker et al, 2004; Hassan et al, 2008a, 2008b). Additionally, ZAG is assigned to the chromosome 7q22.1 based on fluorescent hybridization karyotyping (Hassan et al, 2008a). The crystal structure of ZAG consists of a large groove analogous to class I major histocompatibility complex peptide-binding grooves and the structure and environment of the groove reflect its role in immunoregulation and in lipid catabolism (Sanchez et al, 1999; Hassan et al, 2008b). ZAG appears naturally in blood, sweat, seminal fluid, breast cyst fluid, cerebrospinal fluid, and urine and is also found in secretory epithelial cells of the liver and the gastrointestinal tract (Tada et al, 1991; Hassan et al, 2008a, 2008b). Biochemically, ZAG stimulates lipid degeneration in adipocytes and appears to be involved in cachexia, a wasting syndrome that can affect people with cancer, acquired immunodeficiency syndrome, and other terminal illnesses (Hirai et al, 1998; Bing et al, 2004; Russell and Tisdale, 2005; Hassan et al, 2008b). The purification and characterization of ZAG from human seminal plasma revealed that it was bound to prolactin-inducible complex (PIP) (Hassan et al, 2008a). With the use of a ZAG tryptic peptide as a standard and a high-flow liquid chromatography–tandem mass spectrometry assay, serum levels in six healthy men were calculated as 3.65 (0.71) mg/L (Bondar et al, 2007). Furthermore, the concentration of ZAG and PIP has been reported to increase dramatically in carcinomas; therefore it has been considered as a good biomarker for prostate, breast, oral, and epidermal carcinomas (Hassan et al, 2008b). Thus ZAG is a protein regulated by glucocorticoids and has the ability to impact fertilization and lipid mobilization (adipokine).
Seminal Vesicle Secretory Proteins
Williams-Ashman (1983) presented a classic review on regulatory features of development and function of the seminal vesicles. The secretory proteins of the seminal vesicles are major proteins and enzymes involved in the rapid clotting of the ejaculate (Cunha et al, 1992). The major clotting protein has been termed semenogelin (Lilja and Abrahamsson, 1988). It has been shown to be the seminal vesicle–specific antigen. These clotted proteins from the seminal vesicles serve as substrates for PSA that enzymatically lyse the clot through their protease activity (Lilja, 1985; Aumuller and Seitz, 1990). Beyond the coagulation reaction it is not known what role these seminal vesicle proteins play, but their effects on fertility and uterine sperm motility have been studied in the mouse (Peitz and Olds-Clarke, 1986). Many of the proteins secreted by the seminal vesicles are under androgen regulation (Higgins and Hemingway, 1991; Hagstrom et al, 1992). More recent work (Harvey et al, 1995) has identified an androgen-regulated protease with elastase-like activity within seminal vesicle secretions. The semenogelins I and II are secreted in abundance by the seminal vesicles; and in addition to having coagulum-forming functions and being cleaved by kallikrein-like peptidases to generate biologically active products the semenogelins are assumed to activate sperm hyaluronidase, affect sperm motility, possess antimicrobial activity, serve as substrates for transglutaminase, and have amyloid properties (Jonsson et al, 2006; de Lamirande, 2007; Hassan et al, 2008b).
Additionally, cholesterol and sphingomyelin-rich small, lipid membrane–confined exosome-like vesicles (prostasomes) have been isolated from human semen and these structures provide an additional source of several hundred proteins that are quite important to fully understanding the biology of reproduction as well as improving our knowledge of the semen coagulation and liquefaction system (Ronquist and Brody, 1985; Arienti Saccardi et al, 1999; Poliakov et al, 2009). Prostasomes contain numerous proteins that can affect fertility, promote sperm motility, and stabilize the acrosome reaction (Delves et al, 2007). Sucrose gradient purified prostasomes have been observed by electron microscopy, and their composition has been surveyed after trypsin digestion by liquid chromatography/mass spectroscopy (Poliakov et al, 2009). A diversity of structural and functional proteins involved in fertilization, cell adhesion, apoptosis, immunity, metabolism, signal transduction, transport, angiogenesis, and so on, have been identified in prostasomes and have opened a new source of urologic scientific investigation to pursue new biomarkers of disease and elucidate mechanisms of fertility (Delves et al, 2007; Poliakov et al, 2009).
Coagulation and Liquefaction of Semen
Within 5 minutes after ejaculation, human semen coagulates into a semisolid gel. On further standing for a 5- to 20-minute period, the clot spontaneously liquefies to form a viscous liquid (Huggins, 1942; Tauber and Zaneveld, 1976; Mann, 1981). Calcium-binding substances, such as sodium citrate and heparin, do not inhibit the coagulation process, nor are prothrombin, fibrinogen, or factor XII required because they are absent in seminal plasma (Mann, 1981). The seminal clot is formed of fibers 0.15 to 10 nm in width, and its morphologic appearance differs from that of a blood fibrin clot (Huggins, 1942; Tauber and Zaneveld, 1976; Mann, 1981). Factors affecting blood coagulation do not regulate semen viscosity (Amelar, 1962). From these observations, and others, it appears that the coagulation of human semen is different from that of blood.
Examination of split human ejaculates indicates that the first fraction, originating primarily from the Cowper gland and the prostate, contains the liquefaction factors. The final fraction of the ejaculate, enriched in seminal vesicle secretions, is responsible for the coagulation of the ejaculate (Lilja et al, 1987).
It has long been known that prostatic fluid has a dramatic, fibrinolytic-like activity and that 2 mL of this secretion can liquefy 100 mL of clotted blood in 18 hours at 37°C (Huggins, 1942; Mann, 1981). The factors involved in such proteolytic activity in semen have been resolved (Huggins, 1942; Syner et al, 1975; Tauber et al, 1975, 1976; Tauber and Zaneveld, 1976; Mann, 1981; Zaneveld, 1982; Lilja et al, 1987). Two types of seminal plasma proteolytic enzymes appear to be major factors in the liquefaction process: plasminogen activators and PSA. Two plasminogen activators have been isolated from seminal plasma; they have molecular weights of 70 and 74 kD and appear to be related to urokinase (Propping, 1974). It is believed that the plasminogen activators originate from the prostatic secretions.
The seminal plasma contains a variety of other proteolytic enzymes, including pepsinogen, lysozyme, α-amylase, and hyaluronidase. In addition, human semen inhibits the activity of the proteolytic enzyme trypsin, and this is the result of the presence in the seminal plasma of such proteinase inhibitors as α1-antitrypsin and α1-antichymotrypsin. Coagulation and liquefaction vary in different species. For example, the semen of the bull or dog does not coagulate whereas rodents, such as the rat and guinea pig, ejaculate a firm pellet that does not appear to liquefy (Tauber et al, 1975, 1976; Tauber and Zaneveld, 1976). In rodents the plugs form through the action of an enzyme called vesiculase, which comes from the anterior lobe of the prostate and reacts with seminal vesicle secretions. Because of this action, the anterior lobe of the rodent prostate is also called the coagulating gland. Vesiculase is not identical to thrombin because it does not coagulate fibrinogen, nor does thrombin clot the secretions of the seminal vesicles. Williams-Ashman and associates (1977) have established that vesiculase has transamidase activity, catalyzing the formation of γ-glutamyl-ε-lysine cross links in a clottable protein derived from the seminal vesicles. This seminal vesicle protein, which serves as a substrate for vesiculase, is a basic substance with a molecular weight of 17.9 kD; it has been characterized as to its physical properties.
In summary, it appears that seminal plasma coagulation and liquefaction are under enzymatic control but the biologic purpose of this process has not been resolved. Several key enzymes (e.g., hKLK2 [PSA], hKLK3, hKLK14, PAP) and proteins (e.g., semenogelins, PSP-94, ZAG) of the seminal vesicles and prostate gland are involved in this coagulation and liquefaction system. There have been reports that some infertile men may have impairment of the liquefaction process (Bunge and Sherman, 1954; Bunge, 1970; Eliasson, 1973; Amelar et al, 1977; Jonsson et al, 2006; de Lamirande, 2007; Anahi Franchi et al, 2008; Hassan et al, 2008b; Poliakov et al, 2009).