8 Innate Host Responses
The body protects itself from microbial infection in ways that are similar to those used by a country to protect itself from invasion. Barriers, such as skin, mucosal surfaces, and the acid of the stomach, prevent invasion by most microbes. The microbes that are capable of passing these barriers are bombarded with soluble antimicrobial molecules, such as defensins, complement components, and type 1 interferons. As the infection expands, troops of cells of the innate response, including neutrophils, monocyte-macrophage lineage cells, immature dendritic cells (iDCs), Langerhans cells and dendritic cells (DCs), and natural killer (NK) cells become involved. Often, these innate responses are sufficient to control the infection. Antigen-specific responses support, enhance, and control the cell-mediated innate responses (Box 8-1).
Box 8-1
Innate Host Responses
Innate protections are activated by direct contact with repetitive structures of the microbial surface or genome, termed pathogen-associated molecular patterns (PAMPs). In contrast, the antigen-specific responses sense and are activated by small structures termed epitopes.
Barriers to Infection
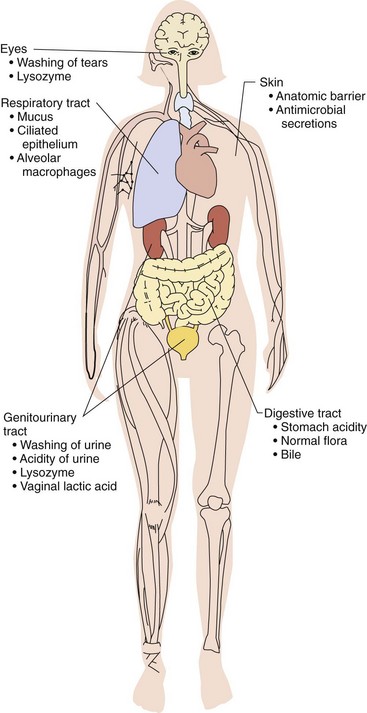
The skin and mucous membranes serve as barriers to most infectious agents (Figure 8-1), with few exceptions (e.g., papillomavirus, dermatophytes [“skin-loving” fungi]). Free fatty acids produced in sebaceous glands and by organisms on the skin surface, lactic acid in perspiration, and the low pH and relatively dry environment of the skin all form unfavorable conditions for the survival of most organisms.
The mucosal epithelium covering the orifices of the body is protected by mucus secretions and cilia. For example, pulmonary airways are coated with mucus, which is continuously transported toward the mouth by ciliated epithelial cells. Large, airborne particles get caught in the mucus, whereas small particles (0.05 to 3 microns [µm], the size of viruses or bacteria) that reach the alveoli are phagocytosed by macrophages and transported out of the airspaces. Some bacteria and viruses (e.g., Bordetella pertussis, influenza virus), cigarette smoke, or other pollutants can interfere with this clearance mechanism by damaging the ciliated epithelial cells, thus rendering the patient susceptible to secondary bacterial pneumonia. Antimicrobial substances (cationic peptides [defensins], lysozyme, lactoferrin, and secretory [IgA]) found in secretions at mucosal surfaces (e.g., tears, mucus, and saliva) also provide protection. Different defensins can disrupt bacterial, viral, and fungal membranes. Lysozyme induces lysis of bacteria by cleaving the polysaccharide backbone of the peptidoglycan of gram-positive bacteria. Lactoferrin, an iron-binding protein, deprives microbes of the free iron they need for growth (Table 8-1).
Table 8-1 Soluble Innate Defense Mediators
| Factor | Function | Source |
|---|---|---|
| Lysozyme | Catalyzes hydrolysis of bacterial peptidoglycan | Tears, saliva, nasal secretions, body fluids, lysosomal granules |
| Lactoferrin, transferrin | Bind iron and compete with microorganisms for it | Specific granules of PMNs |
| Lactoperoxidase | May be inhibitory to many microorganisms | Milk and saliva |
| β-Lysin | Is effective mainly against gram-positive bacteria | Thrombocytes, normal serum |
| Chemotactic factors | Induce directed migration of PMNs, monocytes, and other cells | Complement and chemokines |
| Properdin | Activates complement in the absence of antibody-antigen complex | Normal plasma |
| Lectins | Bind to microbial carbohydrates to promote phagocytosis | Normal plasma |
| Cationic peptides | Disrupt membranes, block cell transport activities | Polymorphonuclear granules, epithelial cells, etc. (defensins, etc.) |
PMNs, Polymorphonuclear neutrophils (leukocytes).
The acidic environment of the stomach, bladder, and kidneys and the bile of the intestines inactivate many viruses and bacteria. Urinary flow also limits the establishment of infection.
Body temperature, and especially fever, limits or prevents the growth of many microbes, especially viruses. In addition, the immune response is more efficient at elevated temperatures.
Soluble Components of Innate Responses
Antimicrobial Peptides
Defensins and cathelicidins are peptides produced by neutrophils, epithelial cells, and other cells that are toxic to many microbes. Defensins are small (approximately 30 amino acids), cationic peptides that can disrupt membranes, kill bacteria and fungi and inactivate viruses. When secreted by Paneth cells in the bowel, they limit and regulate the bacteria living in the lumen. Production of defensins may be constituitive or stimulated by microbial products or cytokines, including interleukin (IL)-17. Cathelicidins are cleaved to produce microbiocidal peptides.
Complement
The complement system is an alarm and a weapon against infection, especially bacterial infection. The complement system is activated directly by bacteria and bacterial products (alternate or properdin pathway), by lectin binding to sugars on the bacterial cell surface (mannose-binding protein), or by complexes of antibody and antigen (classical pathway) (Figure 8-2). Activation by either pathway initiates a cascade of proteolytic events that cleave the proteins into “a” and “b” subunits. The “a” subunits (C3a, C5a) attract (chemotactic factors) phagocytic and inflammatory cells to the site, allow access to soluble molecules and cells by increasing vascular permeability (anaphylactic C3a, C4a, C5a) and activate responses. The “b” subunits are bigger and bind to the agent to promote their phagocytosis (opsonization) and elimination, and build a molecular drill that can directly kill the infecting agent. The three activation pathways of complement coalesce at a common junction point, the activation of the C3 component.
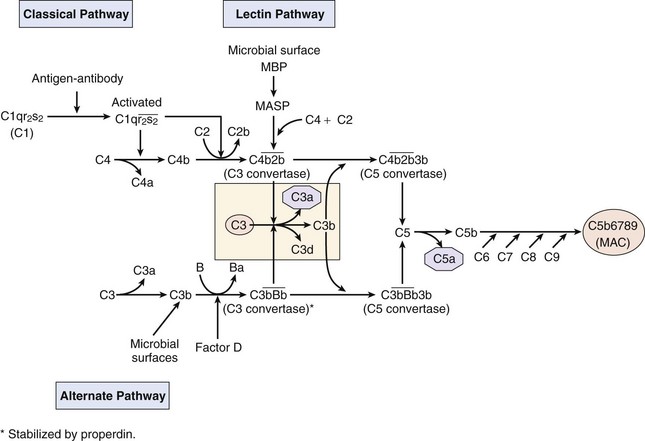
Figure 8-2 The classical, lectin, and alternate complement pathways. Despite different activators, all three pathways converge toward the cleavage of C3 and C5 to provide chemoattractants and anaphylotoxins (C3a, C5a), an opsonin (C3b) that adheres to membranes, a B-cell activator (C3d) and to initiate the membrane attack complex (MAC) to kill cells. MASP, MBP-associated serine protease; MBP, mannose-binding protein.
(Redrawn from Rosenthal KS, Tan M: Rapid review microbiology and immunology, ed 3, St Louis, 2010, Mosby.)
Alternate Pathway
The alternate pathway is activated directly by bacterial cell surfaces and their components (e.g., endotoxin, microbial polysaccharides), as well as other factors. This pathway can be activated before the establishment of an immune response to the infecting bacteria because it does not depend on antibody and does not involve the early complement components (C1, C2, and C4). The initial activation of the alternate pathway is mediated by properdin factor B binding to C3b and then with properdin factor D, which splits factor B in the complex to yield the Bb active fragment that remains linked to C3b (activation unit). The C3b sticks to the cell surface and anchors the complex. The complement cascade then continues in a manner analogous to the classical pathway.
Lectin Pathway
The lectin pathway is also a bacterial and fungal defense mechanism. Mannose-binding protein is a large serum protein that binds to nonreduced mannose, fucose, and glucosamine on bacterial, fungal, and other cell surfaces. Mannose-binding protein resembles and replaces the C1q component of the classical pathways and on binding to bacterial surfaces, activates the cleavage of the mannose binding protein–associated serine protease. Mannose binding protein–associated serine protease cleaves the C4 and C2 components to produce the C3 convertase, the junction point of the complement cascade.
Classical Pathway
The classical complement cascade is initiated by the binding of the first component, C1, to the Fc portion of antibody (IgG or IgM, not IgA or IgE) that is bound to cell surface antigens or to an immune complex with soluble antigens. C1 consists of a complex of three separate proteins designated C1q, C1r, and C1s (see Figure 8-2). One molecule each of C1q and C1s with two molecules of C1r constitutes the C1 complex or recognition unit. C1q facilitates binding of the recognition unit to cell surface antigen-antibody complexes. Binding of C1q activates C1r (referred to now as C1r*) and in turn C1s (C1s*). C1s* then cleaves C4 to C4a and C4b, and C2 to C2a and C2b. The ability of a single recognition unit to split numerous C2 and C4 molecules represents an amplification mechanism in the complement cascade. The union of C4b and C2b produces C4b2b, which is known as C3 convertase. This complex binds to the cell membrane and cleaves C3 into C3a and C3b fragments. The C3b protein has a unique thioester bond that will covalently attach C3b to a cell surface or be hydrolyzed. The C3 convertase amplifies the response by splitting many C3 molecules. The interaction of C3b with C4b2b bound to the cell membrane produces C4b3b2b, which is termed C5 convertase. This activation unit splits C5 into C5a and C5b fragments and represents yet another amplification step.
Biologic Activities of Complement Components
Cleavage of the C3 and C5 components produces important factors that enhance clearance of the infectious agent by promoting access to the infection site and attracting the cells that mediate protective inflammatory reactions. C3b is an opsonin that promotes clearance of bacteria by binding directly to the cell membrane to make the cell more attractive to phagocytic cells, such as neutrophils and macrophages, which have receptors for C3b. C3b can be cleaved further to generate C3d, which is an activator of B lymphocytes. Complement fragments C3a, C4a, and C5a serve as powerful anaphylatoxins that stimulate mast cells to release histamine and tumor necrosis factor-α (TNF-α), which enhances vascular permeability and smooth muscle contraction. C3a and C5a also act as attractants (chemotactic factors) for neutrophils and macrophages by increasing adhesion protein expression of the capillary lining near the infection. These proteins are powerful promoters of inflammatory reactions. For many infections, these responses provide the major antimicrobial function of the complement system.
The complement system also interacts with the clotting cascade. Activated coagulation factors can cleave C5a, and a protease of the lectin pathway can cleave prothrombin to result in production of fibrin and activation of the clotting cascade.
Membrane Attack Complex
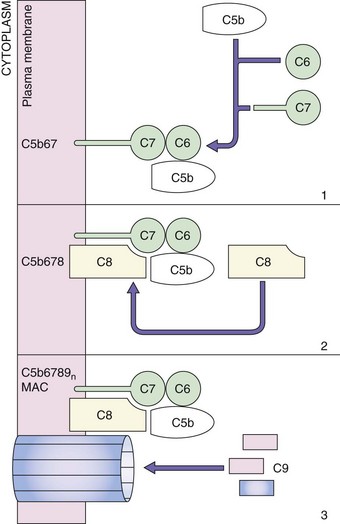
The terminal stage of the classical pathway involves creation of the membrane attack complex (MAC), which is also called the lytic unit (Figure 8-3). The five terminal complement proteins (C5 through C9) assemble into an MAC on target cell membranes to mediate injury. Initiation of the MAC assembly begins with C5 cleavage into C5a and C5b fragments. A (C5b,6,7,8)1(C9)n complex forms and drills a hole in the membrane, leading to apoptosis or the hypotonic lysis of cells. Neisseria bacteria are very sensitive to this manner of killing, while gram-positive bacteria are relatively insensitive. The C9 component is similar to perforin, which is produced by cytolytic T cells and NK cells.
Regulation of Complement Activation
Humans have several mechanisms for preventing generation of the C3 convertase to protect against inappropriate complement activation. These include C1 inhibitor, C4 binding protein, factor H, factor I, and the cell surface proteins, which are decay-accelerating factor (DAF) and membrane cofactor protein. In addition, CD59 (protectin) prevents formation of the MAC. Most infectious agents lack these protective mechanisms and remain susceptible to complement. A genetic deficiency in these protection systems can result in disease.
Interferons
Interferons are small, cytokine-like proteins that can interfere with virus replication but also have systemic effects (described in more detail in Chapter 10). The type I interferons include α and β but not γ, which is a type II interferon. The type I interferons are primarily a very early antiviral response triggered by the double-stranded RNA intermediates of virus replication and other structures that bind to Toll-like receptors (TLRs), RIG-1 (retinoic acid–inducible gene 1), and other PAMP receptors (PAMPRs). Plasmacytoid DCs produce large amounts of IFN-α in response to viral infection, especially during viremia, but other cells can also make IFN-α. IFN-β is made primarily by fibroblasts. The type I interferons promote transcription of antiviral proteins in cells that are activated upon viral infection. They also activate systemic responses, including fever and enhance T-cell activation. Type I interferons will be discussed further with respect to the response to viral infections.
IFN-γ is a type II interferon and differs in biochemical and biologic properties from type I interferons. IFN-γ is primarily a cytokine produced by NK and T cells as part of TH1 immune responses and activates macrophages and myeloid cells. IFN-γ will be discussed further with respect to T-cell responses.
Cellular Components of Innate Responses
Phagocytes
Neutrophils play a major role in antibacterial and antifungal protections and a lesser role for antiviral protections. The neutrophil surface is decorated with receptors that bind microbes, such as C-type lectin and scavenger receptors, and opsonin receptors for the Fc portion of immunoglobulin, C3b, or lectins bound to the microbial surface. These receptors promote phagocytosis of the microbe and their subsequent killing, as described later. Neutrophils have many granules that contain antimicrobial proteins and substances. These cells are terminally differentiated, spend less than 3 days in the blood, rapidly die in tissue, and become pus at the site of infection.
Cells of the Monocyte-Macrophage Lineage
Macrophages mature from blood monocytes and, like neutrophils, are decorated with opsonin receptors to promote phagtocytosis of microbes, receptors for PAMPs (see later) to initiate activation and response, cytokine receptors, to promote activation of the macrophages, and express MHC II proteins for antigen presentation to CD4 T cells (Figure 8-4). Unlike neutrophils, macrophages live longer, must be activated to kill phagocytosed microbes, can divide, and remain at the site of infection or inflammation.
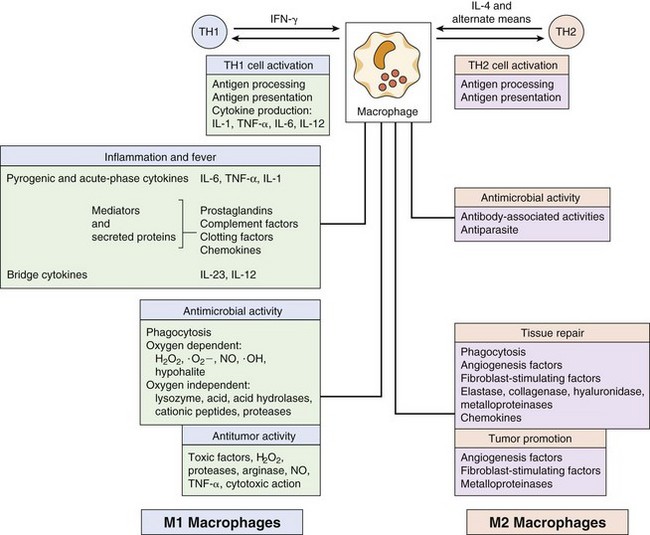
Figure 8-4 The many functions of macrophages and members of the macrophage family. H2O2, Hydrogen peroxide; IFN-γ, interferon-γ; IL, interleukin; NO, nitric oxide; ·O−, oxygen radical; ·OH, hydroxyl radical; TH, T helper (cell); TNF-α, tumor necrosis factor-α.
(From Roitt I, et al: Immunology, ed 4, St Louis, 1996, Mosby.)
Macrophages can be activated by IFN-γ (classical activation) produced by NK cells and CD4 and CD8 T cells as part of the TH1 response and are then able to kill phagocytosed bacteria. These are called M1 macrophages. Activated M1 macrophages produce cytokines, enzymes, and other molecules to promote antimicrobial function (Box 8-2). They also reinforce local inflammatory reactions by producing various chemokines to attract neutrophils, iDCs, NK cells, and activated T cells. Activation of the macrophages makes them more efficient killers of phagocytosed microbes, virally infected cells, and tumor cells. Alternatively activated macrophages (M2 macrophages) are activated by the TH2-related cytokines, IL-4 and IL-13, and support antiparasitic responses, promote tissue remodeling, and wound repair. Continuous (chronic) stimulation of macrophages by T cells, as in the case of an unresolved mycobacterial infection, promotes the fusion of macrophages into multinucleated giant cells and large macrophages called epithelioid cells that surround the infection and form a granuloma.
Box 8-2
Secreted Products of Macrophages with a Protective Effect on the Body
Acute-phase cytokines: IL-6, TNF-α, and IL-1 (endogenous pyrogens)
G-CSF, Granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN-α, interferon-α; IL, interleukin; M-CSF, macrophage colony-stimulating factor; TNF-α, tumor necrosis factor-α.
Immature Dendritic Cells and Dendritic Cells
DCs provide the bridge between the innate and the immune responses. The cytokines they produce determine the nature of the T-cell response. Monocytes and precursor myeloid DCs circulate in the blood and then differentiate into iDCs in tissue and lymphoid organs. iDCs are phagocytic, and upon activation by danger signals, they release an early cytokine-mediated warning system and then mature into DCs. Mature DCs are the ultimate antigen-presenting cell, the only antigen-presenting cell that can initiate an antigen-specific T-cell response (Box 8-3). These cells express different combinations of danger sensors that can detect tissue trauma (adenosine triphosphate [ATP], adenosine, reactive oxygen species [ROS], heat shock proteins) and infection, including Toll-like receptors and other receptors (see later).
Box 8-3
Dendritic Cells (DCs)
Natural Killer, γ/δ T Cells, and NKT Cells
NK cells are innate lymphoid cells (ILCs) that provide an early cellular response to a viral infection, have antitumor activity, and amplify inflammatory reactions after bacterial infection. NK cells are also responsible for antibody-dependent cellular cytotoxicity (ADCC), in which they bind and kill antibody-coated cells. NK cells are large granular lymphocytes (LGLs) that share many characteristics with T cells, except the mechanism for target cell recognition. NK cells do not express a T-cell receptor (TCR) or CD3 and cannot make IL-2. They neither recognize a specific antigen nor require presentation of antigen by MHC molecules. The NK system does not involve memory or require sensitization and cannot be enhanced by specific immunization.
NK cells are activated by (1) IFN-α and IFN-β (produced early in response to viral and other infections), (2) TNF-α, (3) IL-12, IL-15, and IL-18 (produced by pre-DCs and activated macrophages), and (4) IL-2 (produced by CD4 TH1 cells). The NK cells express many of the same cell surface markers as T cells (e.g., CD2, CD7, IL-2 receptor [IL-2R], and FasL [Fas ligand]) but also the Fc receptor for IgG (CD16), complement receptors for ADCC, and NK-specific inhibitory receptors and activating receptors (including NK immunoglobulin-like receptors [KIR]). Activated NK cells produce IFN-γ, IL-1, and granulocyte-macrophage colony-stimulating factor (GM-CSF). The granules in an NK cell contain perforin, a pore-forming protein, and granzymes (esterases), which are similar to the contents of the granules of a CD8 cytotoxic T lymphocyte (CTL). These molecules promote the death of the target cell.
The NK cell sees every cell as a potential victim, especially those that appear in distress, unless it receives an inhibitory signal from the target cell. NK cells interact closely with the target cell by binding to carbohydrates and surface proteins on the cell surface. The interaction of a class I MHC molecule on the target cell with a KIR inhibitory receptor is like communicating a secret password, indicating that all is normal, and this provides an inhibitory signal to prevent NK killing of the target cell. Virus-infected and tumor cells express “stress-related receptors” and are often deficient in MHC I molecules and become NK-cell targets. Binding of the NK cell to antibody-coated target cells (ADCCs) also initiates killing, but this is not controlled by an inhibitory signal. The killing mechanisms are similar to those of CTLs. A synapse (pocket) is formed between the NK and target cell, and perforin and granzymes are released to disrupt the target cell and induce apoptosis. In addition, the interaction of the FasL on the NK cell with Fas protein on the target cell can also induce apoptosis.
Other ILCs resemble CD4 T cells and produce cytokines to regulate epithelial and lymphocyte responses. ILCs line the inside of the intestinal epithelium and produce cytokines to regulate their production of defensins as well as T-cell responses to the gut microbial flora and facilitate antiparasitic worm protections. Errors in their function are associated with inflammatory bowel diseases.
NKT cells and γ/δ T cells reside in tissue and in the blood and differ from other T cells because they have a limited repertoire of T-cell receptors. Unlike other T cells, NKT and γ/δ T cells sense nonpeptide antigens, including bacterial glycolipids (mycobacteria) and phosphorylated amine metabolites from some bacteria (Escherichia coli, mycobacteria) but not others (streptococci, staphylococci). These T cells and NK cells produce IFN-γ, which activate macrophages and DCs to enforce a protective TH1 cycle of cytokines and local cellular inflammatory reactions. NKT cells also express NK-cell receptors.
Activation of Innate Cellular Responses
The cells of the innate response are activated by direct interaction with repetitive external structures and the deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) of microbes. Later, their functions are enhanced, suppressed, and regulated by T cells and T-cell–generated cytokines. These cells express different combinations of danger sensors for microbial infection and cell trauma, including the TLR family of proteins, as well as other receptors. The TLRs include at least 10 different cell surface and intracellular proteins that sense the presence of microbial infection by binding to the characteristic patterns within molecules on the outside of bacteria, fungi, and viruses, and even to forms of the DNA and RNA of these microbes; these are termed pathogen-associated molecular patterns (PAMPs) (Box 8-4; Table 8-2; Figure 8-5). These patterns are present within the endotoxin component of lipopolysaccharide (LPS) and in teichoic acid, fungal glycans, unmethylated cytosine-guanosine units of DNA (CpG oligodeoxynucleotides [ODNs]) commonly found in bacteria, double-stranded RNA produced during the replication of some viruses, and other molecules. Cytoplasmic sensors of bacterial peptidoglycan include nucleotide-binding oligomerization domain protein 1 (NOD1), NOD2, and cryopyrin and, for nucleic acids, RIG-1, melanoma differentiation–associated gene 5 (MDA5), etc. Binding of PAMPs to TLRs and other PAMPRs activates adaptor proteins that trigger cascades of protein kinases and other responses that result in the activation of the cell and production of specific cytokines. These cytokines can include IL-1 and TNF-α, IL-6, interferons α and β, and various chemokines.
Box 8-4
Pathogen Pattern Receptors (PPRs)
PPRs are receptors for microbial structures.
PPRs activate defenses against extracellular and intracellular infections.
AIM2, Absence in melanoma 2; NALP3, nacht, leucine-rich repeat and pyrin domain–containing protein 3; NOD, nucleotide-binding oligomerization domain; RIG-1, retinoic acid–inducible gene-1.
* Proteins may also bind.
Table 8-2 Pathogen Pattern Receptors
| Receptor* | Microbial Activators | Ligand |
|---|---|---|
| Cell Surface | ||
| TLR1 | Bacteria, mycobacteria Neisseria meningitidis |
Lipopeptides Soluble factors |
| TLR2 | Bacteria Fungi Cells |
LTA, LPS, PG, etc. Zymosan Necrotic cells |
| TLR4 | Bacteria, parasites, host proteins Viruses, parasites, host proteins |
LPS, fungal mannans, viral glycoproteins, parasitic phospholipids, host heat shock proteins, LDL |
| TLR5 | Bacteria | Flagellin |
| TLR6 | Bacteria Fungi |
LTA, lipopeptides, zymosan |
| Lectins | Bacteria, fungi, viruses | Specific carbohydrates (e.g., mannose) |
| N-Formyl methionine receptor | Bacteria | Bacterial proteins |
| Endosome | ||
| TLR3 | Viruses | Double-stranded RNA |
| TLR7 | Viruses | Single-stranded RNA Imidazoquinolines |
| TLR8 | Viruses | Single-stranded RNA Imidazoquinolines |
| TLR9 | Bacteria Viruses |
Unmethylated DNA (CpG) |
| Cytoplasm | ||
| NOD1, NOD2, NALP3 | Bacteria | Peptidoglycan |
| Cryopyrin | Bacteria | Peptidoglycan |
| RIG-1 | Viruses | RNA |
| MDA5 | Viruses | RNA |
| DAI | Viruses, cytoplasmic DNA | DNA |
Activators: DAI, DNA-dependent activator of interferon regulatory factors; DNA, deoxyribonucleic acid; dsRNA, double-stranded RNA; LDL, minimally modified low-density lipoprotein; LPS, lipopolysaccharide; LTA, lipoteichoic acid; MDA5, melanoma differentiation–associated gene 5; NALP3, Nacht, leucine-rich repeat and pyrin domain–containing protein 3; NOD, nucleotide-binding oligomerization domain; PG, peptidoglycan; RIG-1, retinoic acid–inducible gene-1; TLR, Toll-like receptor.
* Information about Toll-like receptors from Takeda A, Kaisho T, Akira S: Toll-like receptors, Annu Rev Immunol 21:335–376, 2003; and Akira S, Takeda K: Toll-like receptor signalling, Nat Rev Immunol 4:499–511, 2003.
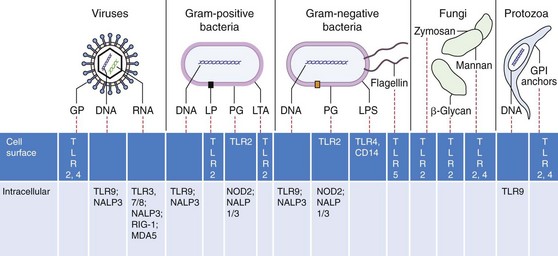
Figure 8-5 Recognition of pathogen-associated molecular patterns. Microbial structures, RNA and DNA bind to specific receptors on the cell surface, in vesicles, or in the cytoplasm to activate innate responses. FL, Flagellin; GP, glycoproteins; GPI, phosphatidylinositol glycan–anchored proteins; LP, lipoproteins; LPS, lipopolysaccharide; LTA, lipoteichoic acid; MDA5, melanoma differentiation–associated gene 5; NALP3, Nacht, leucine-rich repeat and pyrin domain–containing protein 1/3; NOD2, nucleotide-binding oligomerization domain protein 2; PG, peptidoglycan; RIG-1, retinoic acid-inducible gene protein-1; TLR9, Toll-like receptor 9.
(Modified from Mogensen TH: Pathogen recognition and inflammatory signaling in innate immune defenses, Clin Microbiol Rev 22:240–273, 2009.)
Local inflammation is also promoted by the inflammasome (Figure 8-6). The inflammasome is a multiprotein complex present in epithelial cells, DCs, macrophages, and other cells and are activated by several of the adaptor proteins induced in response to PAMPRs, tissue damage, or indications of intracellular infection. Proteases released upon uric acid crystal (gout) or asbestos puncture of phagosomes and lysosomes can also activate inflammasome formation. The inflammasome activates the caspase 1 protease, which then cleaves, activates, and promotes the release of IL-1β and IL-18. These activated cytokines promote local inflammation. The activated inflammasome can also initiate an apoptosis-like cell death for cells bearing intracellular bacterial infections.
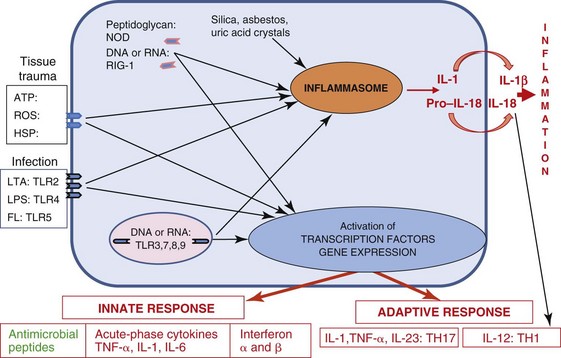
Figure 8-6 Induction of inflammatory responses. Receptors for pathogen-associated molecular patterns and danger signals (damage-associated molecular patterns receptors) at the cell surface, in vesicles, and in the cytoplasm (1) activate signal cascades that (2) produce adaptor proteins that (3) activate local inflammatory responses. The adaptor proteins initiate the assembly of the inflammasome and also trigger the transcription of cytokines. Cytokines activate innate and promote antigen-specific responses. In addition, crystalline materials lyse lysosomes, releasing proteases that cleave precursors to initiate assembly and activation of the inflammasome and promote inflammation. ATP, Adenosine triphosphate; FL, flagellin; HSP, heat shock protein; IL, interleukin; LPS, lipopolysaccharide; LTA, lipoteichoic acid; NOD, nucleotide-binding oligomerization domain protein; RIG-1, retinoic acid-inducible gene protein 1; ROS, reactive oxygen species; TLR, Toll-like receptor; TNF-α, tumor necrosis factor-α.
Chemotaxis and Leukocyte Migration
Chemotactic factors produced in response to infection and inflammatory responses, such as complement components (C3a, C5a), bacterial products (e.g., formyl-methionyl-leucyl-phenylalanine [f-met-leu-phe]), and chemokines, are powerful chemoattractants for neutrophils, macrophages, and later in the response, lymphocytes. Chemokines are small cytokine-like proteins that direct the migration of white blood cells. Most chemokines are either CC (adjacent cysteines) or CXC (cysteines separated by one amino acid) chemokines. Chemokines bind to G-protein–coupled receptors specific for structurally similar cytokines. Chemokines may recruit lymphocytes and leukocytes to the site of infection or inflammation or to different sites in the lymph node. The chemokines establish a chemically lighted “runway” to guide these cells to the site of an infection and also activate them. The chemokines, IL-1, and TNF-α cause the endothelial cells lining the capillaries (near the inflammation) and the leukocytes passing by to express complementary adhesion molecules (molecular “Velcro”). The leukocytes slow, roll, attach to the lining, and then extravasate across (i.e., pass through) the capillary wall to the site of inflammation, a process called diapedesis (Figure 8-7).
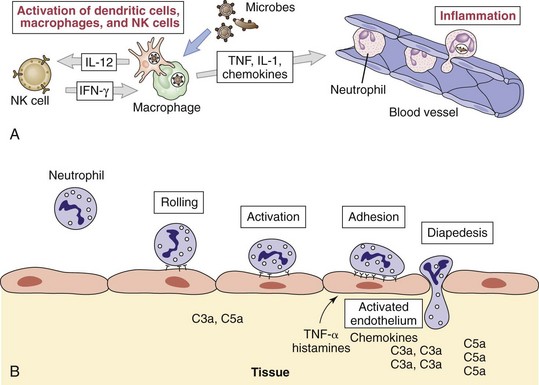
Figure 8-7 A and B, Neutrophil diapedesis in response to inflammatory signals. Tumor necrosis factor-α (TNF-α) and chemokines activate the expression of selectins and intercellular adhesion molecules on the endothelium near the inflammation and their ligands on the neutrophil: integrins, L-selectin, and leukocyte function–associated antigen-1. The neutrophil binds progressively tighter to the endothelium until it finds its way through the endothelium. Epithelial cells, Langerhans cells, and macrophages activated by microbes and interferon-γ (IFN-γ) make TNF-α and other cytokines and chemokines to enhance diapedesis. IL, Interleukin; NK, natural killer.
(A, From Abbas AK, Lichtman AH: Basic immunology: functions and disorders of the immune system, ed 3, Philadelphia, 2008, WB Saunders.)
Phagocytic Responses
Polymorphonuclear neutrophils (PMNs), monocytes, and occasionally eosinophils are the first cells to arrive at the site in response to infection; they are followed later by macrophages. Neutrophils provide a major antibacterial response and contribution to inflammation. An increased number of neutrophils in the blood, body fluids (e.g., cerebrospinal fluid), or tissue indicates a bacterial infection. The mobilization of neutrophils is accompanied by a “left shift,” an increase in the number of immature band forms released from the bone marrow (left refers to the beginning of a chart of neutrophil development).
Phagocytosis of bacteria by macrophages and neutrophils involves three steps: attachment, internalization, and digestion. Attachment of the bacteria to the macrophage is mediated by receptors for bacterial carbohydrates (lectins [specific sugar-binding proteins]), fibronectin receptors (especially for Staphylococcus aureus), and receptors for opsonins, including complement (C3b), mannose-binding protein, and the Fc portion of antibody. After attachment, a section of plasma membrane surrounds the particle, which forms a phagocytic vacuole around the microbe. This vacuole fuses with the primary lysosomes (macrophages) or granules (PMNs) to allow inactivation and digestion of the vacuole contents.
Phagocytic killing may be oxygen dependent or oxygen independent, depending on the antimicrobial chemicals produced by the granules (Figure 8-8). Neutrophils do not need special activation to kill internalized microbes, but their response is reinforced by IL-17–mediated activities. Activation of macrophages is promoted by IFN-γ (best) and GM-CSF, which are produced early in the infection by NK and NKT cells or later by CD4 T cells, and sustained by TNF-α and lymphotoxin (TNF-β). Activation of macrophages is required for macrophages to kill internalized microbes.
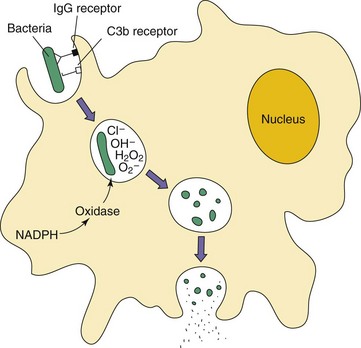
Figure 8-8 Phagocytosis and killing of bacteria. Bacteria are bound directly or are opsonized by mannose-binding protein, immunoglobulin G (IgG), and/or C3b receptors, promoting their adherence and uptake by phagocytes. Within the phagosome, oxygen-dependent and oxygen-independent mechanisms kill and degrade the bacteria. NADPH, Nicotinamide adenine dinucleotide phosphate reduced.
Oxygen-dependent killing is activated by a powerful oxidative burst that culminates in the formation of hydrogen peroxide and other antimicrobial substances (ROS) (Box 8-5). In the neutrophil, but not the macrophage, hydrogen peroxide with myeloperoxidase (released by primary granules during fusion to the phagolysosome) transforms chloride ions into hypochlorous ions that kill the microorganisms. Nitric oxide produced by neutrophils and activated macrophages has antimicrobial activity and is also a major second messenger molecule (like cyclic adenosine monophosphate [cAMP]) which enhances the inflammatory and other responses.
Box 8-5
Antibacterial Compounds of the Phagolysosome
NADH, Nicotinamide adenine dinucleotide reduced; NADPH, nicotinamide adenine dinucleotide phosphate reduced.
The neutrophil can also mediate oxygen-independent killing upon fusion of the phagosome with azurophilic granules containing cationic proteins (e.g., cathepsin G) and specific granules containing lysozyme and lactoferrin. These proteins kill gram-negative bacteria by disrupting their cell membrane integrity, but they are far less effective against gram-positive bacteria, which are killed principally through the oxygen-dependent mechanism.
The neutrophils contribute to the inflammation in several ways. Prostaglandins and leukotrienes, which increase vascular permeability, are released, causing swelling (edema) and stimulating pain receptors. In addition, during phagocytosis, the granules may leak their contents to cause tissue damage. The neutrophils have short lives, and dead neutrophils produce pus.
Resting macrophages are phagocytic and will internalize microbes but do not have the preformed granules of antimicrobial molecules to kill them. Activation of the macrophage by IFN-γ, making the macrophages “angry,” promotes production of inducible nitric oxide synthase (iNOS) and nitric oxide, other ROS, and antimicrobial enzymes to kill internalized microbes. Activated macrophages also make acute-phase cytokines (IL-1, IL-6, and TNF-α) and possibly IL-23 or IL-12. Intracellular infection can occur upon infection of a resting macrophage or if the microbe can counteract the antimicrobial activities of an activated macrophage.
In addition to the tissue macrophages, splenic macrophages are important for clearing bacteria, especially encapsulated bacteria, from blood. Asplenic (congenitally or surgically) individuals are highly susceptible to pneumonia, meningitis, and other manifestations of Streptococcus pneumoniae, Neisseria meningitidis, and other encapsulated bacteria.
Normal Flora–Associated Responses
Innate responses are constantly being stimulated by the normal flora of the skin, nares, oral region, urogenital and gastrointestinal tracts. PAMPRs on the cells of the intestine continuously see the LPS, lipoteichoic acid (LTA), flagella, and other components of the bacteria within the lumen. An equilibrium is maintained between innate, immune regulatory responses and their microbial stimuli. Disruption of the equilibrium by altering the microbial species with antimicrobial treatment or by disrupting the innate and immune responses can result in inflammatory bowel disease, autoimmune diseases, or gastroenteritis.
Inflammation
Proinflammatory Cytokines
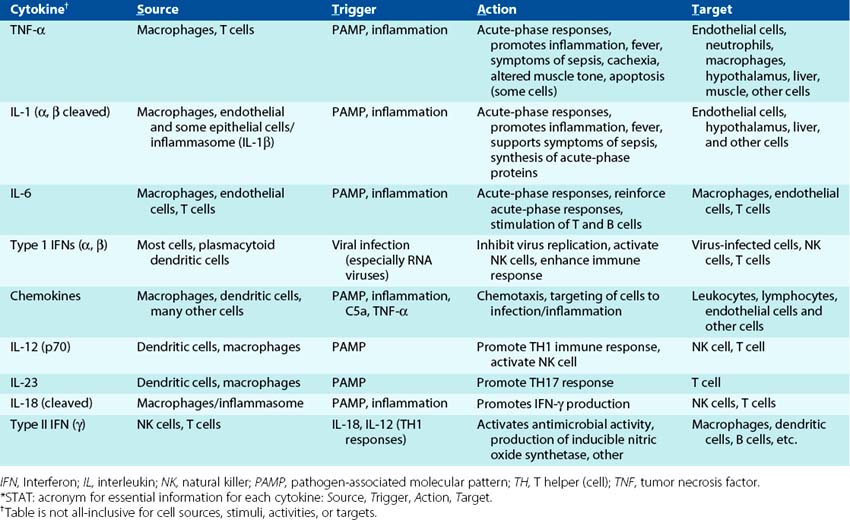
The proinflammatory cytokines, sometimes referred to as acute-phase cytokines, are IL-1, TNF-α, and IL-6 (Table 8-3). These cytokines are produced by activated macrophages and other cells. IL-1 and TNF-α share properties. Both of these cytokines are endogenous pyrogens capable of stimulating fever; they promote local inflammatory reactions and synthesis of acute-phase proteins.
TNF-α is the ultimate mediator of inflammation and the systemic effects of infection. TNF-α stimulates endothelial cells to express adhesion molecules and chemokines to attract leukocytes to the site of infection, activates neutrophils and macrophages, and promotes apoptosis of certain cell types. Systemically, TNF acts on the hypothalamus to induce fever, can cause systemic metabolic changes, weight loss (cachexia) and loss of appetite, and enhance production of IL-1, IL-6, and chemokines, and it promotes acute-phase protein synthesis by the liver. At high concentrations, TNF-α elicits all of the functions leading to septic shock.
There are two types of IL-1, IL-1α and IL-1β. IL-1 is produced mainly by activated macrophages, also neutrophils, epithelial, and endothelial cells. IL-1β must be cleaved by the inflammasome to become activated. IL-1 shares many of the activities of TNF-α to promote local and systemic inflammatory responses. Unlike TNF-α, IL-1 cannot induce apoptosis and will enhance but is not sufficient to cause septic shock. IL-6 is produced by many cell types, promotes the synthesis of acute-phase proteins in the liver, production of neutrophils in bone marrow, and the activation of T and B lymphocytes.
IL-23 and IL-12 are cytokines that bridge the innate and immune responses. Both cytokines have two subunits, a p40 subunit and a p35 subunit for IL-12 and a p19 subunit for IL-23. IL-23 promotes TH17 responses from memory T cells, which enhance neutrophil action. IL-12 promotes NK-cell function and is required to promote a TH1 immune response, which enhances macrophages and other myeloid cells functions. These cytokines will be discussed further regarding their actions on T cells. IL-18 is produced by macrophages, must be cleaved by the inflammasome to an active form, and promotes NK- and T-cell function.
Acute Inflammation
Acute inflammation is an early defense mechanism to contain an infection, prevent its spread from the initial focus, and activate subsequent immune responses. Initially, inflammation can be triggered by the response to danger signals resulting from infection and tissue damage and then may be maintained or enhanced by cytokine and T-cell stimulation of additional cellular responses.
The three major events in acute inflammation are (1) expansion of capillaries to increase blood flow (causing redness or a rash and releasing heat); (2) increase in permeability of the microvasculature structure to allow escape of fluid, plasma proteins, and leukocytes from the circulation (swelling or edema); and (3) recruitment of neutrophils and their accumulation and response to infection at the site of injury. Inflammatory responses are beneficial but are associated with pain, redness, heat, and swelling and can also cause tissue damage. The mediators of inflammation are listed in Table 8-4.
Table 8-4 Mediators of Acute and Chronic Inflammation
| Action | Mediators |
|---|---|
| Acute Inflammation | |
| Increased vascular permeability | Histamine, bradykinin, C3a, C5a, leukotrienes, PAF, substance P |
| Vasodilation | Histamine, prostaglandins, PAF, nitric oxide (NO) |
| Pain | Bradykinin, prostaglandins |
| Leukocyte adhesion | Leukotriene B4, IL-1, TNF-α, C5a |
| Leukocyte chemotaxis | C5a, C3a, IL-8, chemokines, PAF, leukotriene B4 |
| Acute-phase response | IL-1, IL-6, TNF-α |
| Tissue damage | Proteases, free radicals, NO, neutrophil granule contents |
| Fever | IL-1, TNF, prostaglandins |
| Chronic Inflammation | |
| Activation of T cells and macrophages, and acute-phase processes | T cell (TNF, IL-17, IFN-γ) and macrophages (IL-1, TNF-α, IL-23, IL-12) cytokines |
IFN-γ, Interferon-γ; IL, interleukin; PAF, platelet activating factor; TNF, tumor necrosis factor.
From Novak R: Crash course immunology, Philadelphia, 2006, Mosby.
Tissue damage is caused to some extent by complement and macrophages but mostly by neutrophils. Dead neutrophils are a major component of pus. Kinins and clotting factors induced by tissue damage (e.g., factor XII [Hageman factor], bradykinin, fibrinopeptides) are also involved in inflammation. These factors increase vascular permeability and are chemotactic for leukocytes. Products of arachidonic acid metabolism also affect inflammation. Cyclooxygenase-2 (COX-2) and 5-lipooxygenase convert arachidonic acid to prostaglandins and leukotrienes, respectively, which can mediate essentially every aspect of acute inflammation. The course of inflammation can be followed by rapid increases in acute-phase proteins, especially C-reactive protein (which can increase a thousand fold within 24 to 48 hours) and serum amyloid A.
Acute-Phase Response
The acute-phase response is triggered by infection, tissue injury, prostaglandin E2, interferons associated with viral infection, acute-phase cytokines (IL-1, IL-6, TNF-α), and inflammation (Box 8-6). The acute-phase response promotes changes that support host defenses and include fever, anorexia, sleepiness, metabolic changes, and production of proteins. IL-1 and TNF-α are also endogenous pyrogens because they promote fever production. Acute-phase proteins that are produced and released into the serum include C-reactive protein, complement components, coagulation proteins, LPS-binding proteins, transport proteins, protease inhibitors, and adherence proteins. C-reactive protein binds to the polysaccharides of numerous bacteria and fungi and activates the complement pathway, facilitating removal of these organisms from the body by enhancing phagocytosis. Hepcidin inhibits iron uptake by the gut and macrophages, and this reduces availability to microbes. The acute-phase proteins reinforce the innate defenses against infection, but their excessive production during sepsis (induced by endotoxin) can cause serious problems, such as shock.
Sepsis and Cytokine Storms
Cytokine storms are generated by an overwhelming release of cytokines in response to bacterial cell wall components, especially LPS, toxic shock toxins, and certain viral infections, especially viremias. During bacteremia, large amounts of complement C5a and cytokines are produced and distributed throughout the body (Figure 8-9). C5a promotes vascular leakage, neutrophil activation, and activation of the coagulation pathway. Plasmacytoid DCs in the blood produce large amounts of inflammatory cytokines and IL-12 in response to bacterial PAMPs. Endotoxin is an especially potent activator of cells and inducer of cytokine production and sepsis (see Figure 14-4). Cytokine storms can also occur upon the abnormal stimulation of T cells and antigen-presenting cells (DCs, macrophages, and B cells) by superantigens produced by S. aureus or Streptococcus pyogenes (see Figure 14-3). During viremia, large amounts of IFN-α and other cytokines are produced by plasmacytoid DCs and by T cells.
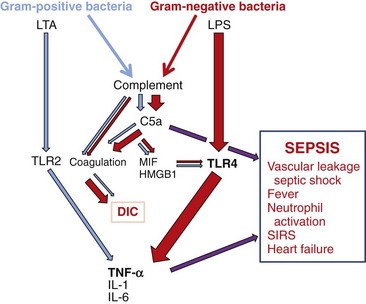
Figure 8-9 Gram-positive and gram-negative bacteria induce sepsis by shared and separate pathways. Bacterial surfaces and lipopolysaccharide (LPS) activate complement, producing C5a, which facilitates inflammation, activates coagulation, and produces macrophage migration inhibitory factor (MIF) and high–mobility group box 1 protein (HMGB1), cytokines that enhance inflammation. LPS, lipoteichoic acid (LTA), and other pathogen-associated molecular patterns interact with Toll-like receptors (TLRs) and other pathogen pattern receptors to activate inflammation and proinflammatory cytokine production. These add up to sepsis. DIC, Disseminated intravascular coagulation; IL, interleukin; SIRS, systemic inflammatory response syndrome; TNF-α, tumor necrosis factor-α.
(Modified from Rittirsch D, Flierl MA, Ward PA: Harmful molecular mechanisms in sepsis, Nat Rev Immunol 8:776–787, 2008.)
The excess cytokines in the blood can induce inflammatory trauma throughout the entire body. Most significantly, increases in vascular permeability can result in leakage of fluids from the bloodstream into tissue and cause shock. Septic shock is a form of cytokine storm and can be attributed to the systemic action of large quantities of TNF-α.
Bridge to Antigen-Specific Immune Responses
The innate response is often sufficient to control an infection but also initiates antigen-specific immunity. First, the complement components, cytokines, chemokines, and interferons produced during the acute-phase response prepare the lymphocytes, then the DCs deliver the antigen and intiate the T-cell response in the lymph node. DCs are the key to the transition and determine the nature of the subsequent response (Figure 8-10).
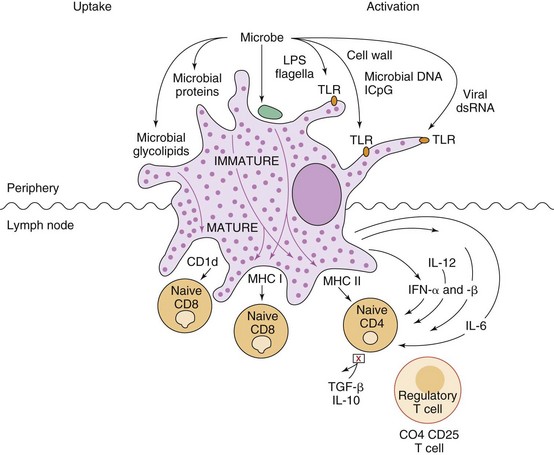
Figure 8-10 Dendritic cells (DCs) initiate immune responses. Immature DCs constantly internalize and process proteins, debris, and microbes, when present. Binding of microbial components to Toll-like receptors (TLRs) activates the maturation of the DC so that it ceases to internalize any new material; moves to the lymph node, up-regulates major histocompatibility complex (MHC) II, and co-receptors B7, and B7-1 molecules for antigen presentation; and produces cytokines to activate T cells. Release of interleukin (IL)-6 inhibits release of transforming growth factor-β (TGF-β) and IL-10 by T-regulatory cells. The cytokines produced by DCs and their interaction with TH0 cells initiate immune responses. IL-12 promotes TH1 responses, while IL-4 promotes TH2 responses. Most of the T cells divide to enlarge the response but some remain as memory cells. Memory cells can be activated by a DC-, macrophage-, or B-cell presentation of antigen for a secondary response. IFN, Interferon; LPS, lipopolysaccharide.
iDCs are constantly acquiring antigenic material by macropinocytosis, pinocytosis, or phagocytosis of apoptotic cells, debris, and proteins in normal tissue and at the site of infection or tumor. Upon activation of the iDC through a PAMPR in response to infection, acute-phase cytokines (IL-1, IL-6, and TNF-α) are released, the iDC matures into a DC, and its role changes. The DC loses its ability to phagocytize, preventing it from acquiring irrelevant antigenic material other than the microbial debris, and it progresses to the lymph node. By analogy, the iDC is like a clam, constantly surveying its environment by filter feeding the cellular and microbial debris (if present), but when triggered by a TLR signal, indicating that microbes are present, it releases a local cytokine alarm, closes its shell, and moves to the lymph node to trigger a response to the challenge. The mature DC moves to T-cell areas of lymph nodes and up-regulates its cell surface molecules for antigen presentation (class II MHC and B7-1 and B7-2 [co-stimulatory] molecules). Microbe-activated mature DCs release cytokines (e.g., IL-12), which activate responses to reinforce local host defenses (TH1 responses). DCs present antigenic material attached to MHC class I and CD1 molecules to CD8 T and NKT cells, and on MHC class II molecules to CD4 T cells. DCs are so effective at presenting antigen that 10 cells loaded with antigen are sufficient to initiate protective immunity to a lethal bacterial challenge in a mouse. T-cell responses will be described in the next chapter.
1. What are the innate soluble factors that act on microbial infections, and what are their functions?
2. What are the contributions of neutrophils, M1 and M2 macrophages, Langerhans, and DCs to antimicrobial responses?
3. A 65-year-old woman has fever and chills. A gram-negative, oxidase-negative bacillus is isolated from her blood. What triggered and is causing her symptoms?
4. A 45-year-old man has a boil on his hand. A gram-positive, catalase- and coagulase-positive coccus was isolated from the pus of the lesion. What innate responses are active on this infection?
| Factor | Action |
|---|---|
| Antimicrobial peptides | Killing of microbe |
| Complement: MAC | Kills gram-negative bacteria |
| Complement: C3b | Opsonization |
| Complement: C3d | Activates B cells |
| Complement: “a” fragments C3a, C4a, C5a | Attraction, anaphylaxis |
| Lectins | Opsonization |
| C-reactive protein | Opsonization |
| Cytokines | Activation of responses |
| Chemokines | Attraction of leukocytes |
2. Neutrophils leave the bone marrow ready to attack. Neutrophils are phagocytic and the major antibacterial response. Their granules are filled with antimicrobial substances and enzymes that are released into endosomes and leak from the cell upon phagocytosis of a microbe. They are the first to be attracted to an infection and have a very short half-life.
Macrophages enter later than neutrophils. They may be resident, or they may mature from monocytes that enter the site of infection. Macrophages must be activated by IFN-γ and TNF-α produced by NK cells or T cells to become and maintain inflammatory antimicrobial activity (M1). Macrophages have a long lifespan. M1 macrophages produce acute-phase cytokines, IL-12, and antibacterial substances, such as reactive oxygen molecules, nitric oxide, and enzymes. Macrophages are also antigen-presenting cells and use the peptides presented on MHC II molecules to recruit and activate T-cell help. M2 macrophages develop in the presence of IL-4, are also phagocytic and promote wound healing and angiogenesis. Macrophages may progress from M1 to M2, changing their contribution to resolution of the infection and its damage.
DCs are the only cells that can initiate an immune response by activating naïve T cells. iDCs are also an early warning system that release cytokines and chemokines appropriate to the microbial trigger, which will facilitate other host protections. Langerhans cells are a skin-resident DC that can also move to the lymph node to activate naïve T cells. DCs are a bridge between the innate and the immune response.
3. The lipid A (endotoxin) of the LPS from the outer membrane of the enteric (probably E. coli) bacteria in the blood binds to TLR4 on macrophages and other cells to activate the production of acute-phase cytokines (TNF-α, IL-1, and IL-6). TNF-α and IL-1 are endogenous pyrogens that promote fever production. These cytokines also induce other systemic effects. The bacteria will also activate the alternate and lectin pathways of complement, and the “a” components (C3a, C4a, and C5a) will also trigger systemic inflammatory responses.
4. The S. aureus infection triggers release of bactericidal peptides from epithelial and other cells, complement activation, release of C3a and C5a to act as chemotactic and anaphylactic substances to attract neutrophils and, later, macrophages to the site. LTA will activate TLR2 to promote TNF-α and IL-1 production by macrophages which will further promote the inflammation. Dead neutrophils produce pus.
Abbas AK, et al. Cellular and molecular immunology, ed 7. Philadelphia: WB Saunders; 2011.
Akira S, Takeda K. Toll-like receptor signaling. Nat Rev Immunol. 2004;4:499–511.
DeFranco AL, Locksley RM, Robertson M. Immunity: the immune response in infectious and inflammatory disease. Sunderland, Mass: Sinauer Associates; 2007.
Janeway CA, et al. Immunobiology: the immune system in health and disease, ed 6. New York: Garland Science; 2004.
Kindt TJ, Goldsby RA, Osborne BA. Kuby immunology, ed 7. New York: WH Freeman; 2011.
Kumar V, Abbas AK, Fausto N. Robbins and Cotran pathologic basis of disease, ed 7. Philadelphia: Elsevier; 2005.
Lamkanfi M. Emerging inflammasome effector mechanisms. Nat Rev Immunol. 2011;11:213–220.
Netea MG, van der Meer JW. Immunodeficiency and genetic defects of pattern-recognition receptors. N Engl J Med. 2011;364:60–70.
Rittirsch D, Flierl MA, Ward PA. Harmful molecular mechanisms in sepsis. Nat Rev Immunol. 2008;8:776–787.
Sompayrac L. How the immune system works, ed 2. Malden, Mass: Blackwell Scientific; 2003.
Takeda K, Kaisho T, Akira S. Toll-like receptors. Annu Rev Immunol. 2003;21:335–376.
Trends Immunol: Issues contain understandable reviews on current topics in immunology.