58 Rhabdoviruses, Filoviruses, and Bornaviruses
A 15-year-old girl picked up a bat and was bitten on her hand. One month later, she developed double vision, nausea, and vomiting. Over the course of 4 days, her neurologic disease developed, and she had a fever of 38.9° C. Rabies was suspected, and rabies-virus–specific antibodies were detected in the patient’s serum and cerebrospinal fluid (1 : 32 titer). The patient was put into a drug-induced coma with ventilator support and treated with intravenous ribavirin for 7 days, when cerebrospinal fluid antibody titers rose to 1 : 2048. After 3 months, she was able to walk with assistance; ride a stationary cycle for 8 minutes; and feed herself a soft, solid diet; solve math puzzles; use sign language; and was regaining the ability to speak. This is the only example of a patient surviving without having received timely postexposure rabies immunization.*
1. How is rabies infection confirmed?
2. What is the usual disease progression following a bite from a rabid animal?
3. When is antirabies antibody detected in a normal rabies disease presentation?
4. What is postexposure rabies immunization, and why does it work?
5. How does ribavirin inhibit the replication of rabies and other viruses?
Answers
1. Antirabies antibody may be present late in the infection and an ELISA can be used for detection. Cerebrospinal fluid or saliva can be analyzed by RT-PCR for viral genome. A brain biopsy can be analyzed by direct immunofluorescence detection for rabies antigen.
2. After a long incubation period, initial symptoms are fever, malaise, headache, pain or paresthesia (itching) at the site of the bite, gastrointestinal symptoms, fatigue, and anorexia. The prodrome usually lasts 2 to 10 days, after which the neurologic symptoms specific to rabies appear. Hydrophobia (fear of water), triggered by the pain associated with the patient’s attempts to swallow water, focal and generalized seizures, disorientation, and hallucinations are also common during the neurologic phase. The paralysis may lead to respiratory failure. The patient becomes comatose after the neurologic phase, which lasts from 2 to 10 days. This phase almost universally leads to death resulting from neurologic and pulmonary complications.
3. The antibody is detected late in the course of disease, after the infection has progressed to generate neurologic symptoms.
4. After being bitten by an animal suspected of carrying rabies, the bite site is washed carefully and then instilled with rabies immune globulin. The patient then receives four immunizations with rabies antigen.
5. Ribavirin is a guanosine analogue that promotes hypermutation of the viral genome, leading to the production of noninfectious viruses.
Rhabdoviruses
The members of the family Rhabdoviridae (from the Greek word rhabdos, meaning “rod”) include pathogens for a variety of mammals, fish, birds, and plants. The family contains Vesiculovirus (vesicular stomatitis viruses [VSVs]), Lyssavirus (Greek for “frenzy”) (rabies and rabies-like viruses), an unnamed genus constituting the plant rhabdovirus group, and other ungrouped rhabdoviruses of mammals, birds, fish, and arthropods.
Rabies virus is the most significant pathogen of the rhabdoviruses. Until Louis Pasteur developed the killed-rabies vaccine, a bite from a “mad” dog always led to the characteristic symptoms of hydrophobia and certain death.
Physiology, Structure, and Replication
Rhabdoviruses are simple viruses encoding only five proteins and appearing as bullet-shaped, enveloped virions with a diameter of 50 to 95 nm and length of 130 to 380 nm (Box 58-1, Figure 58-1). Spikes composed of a trimer of the glycoprotein (G) cover the surface of the virus. The viral attachment protein, G protein, generates neutralizing antibodies. The G protein of the vesicular stomatitis virus is a simple glycoprotein with N-linked glycan. This G protein has been used as the prototype for studying eukaryotic glycoprotein processing.
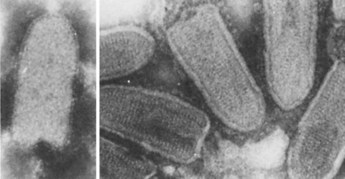
Figure 58-1 Rhabdoviridae seen by electron microscopy: rabies virus (left) and vesicular stomatitis virus (right).
(From Fields BN: Virology, New York, 1985, Raven.)
Within the envelope, the helical nucleocapsid is coiled symmetrically into a cylindric structure, giving it the appearance of striations (see Figure 58-1). The nucleocapsid is composed of one molecule of single-stranded, negative-sense ribonucleic acid (RNA) of approximately 12,000 bases and the nucleoprotein (N), large (L), and nonstructural (NS) proteins. The matrix (M) protein lies between the envelope and the nucleocapsid. The N protein is the major structural protein of the virus. It protects the RNA from ribonuclease digestion and maintains the RNA in a configuration acceptable for transcription. The L and NS proteins constitute the RNA-dependent RNA polymerase.
The replicative cycle of VSV is the prototype for the rhabdoviruses and other negative-strand RNA viruses (see Chapter 44, Figure 44-14). The viral G protein attaches to the host cell and is internalized by endocytosis. Rabies virus binds to either the nicotinic acetylcholine receptor (AChR), neural cell adhesion molecule (NCAM), or other molecules. The viral envelope then fuses with the membrane of the endosome on acidification of the vesicle. This uncoats the nucleocapsid, releasing it into the cytoplasm, where replication takes place. Endosomal vesicles may deliver whole rabies virions along the axon to neuronal cell bodies, where its replication takes place.
The RNA-dependent RNA polymerase associated with the nucleocapsid transcribes the viral genomic RNA, producing five individual messenger RNAs (mRNAs). For rabies virus, this occurs in the Negri bodies. These mRNAs are then translated into the five viral proteins. The viral genomic RNA is also transcribed into a full-length, positive-sense RNA template that is used to generate new genomes. The G protein is synthesized by membrane-bound ribosomes, processed by the Golgi apparatus, and delivered to the cell surface in membrane vesicles. The M protein associates with the G protein–modified membranes.
Assembly of the virion occurs in two phases: (1) assembly of the nucleocapsid in the cytoplasm and (2) envelopment and release at the cell plasma membrane. The genome associates with the N protein and then with the polymerase proteins L and NS to form the nucleocapsid. Association of the nucleocapsid with the M protein at the plasma membrane induces coiling into its condensed form and the characteristic bullet shape of the virion. The virus then buds through the plasma membrane and is released when the entire nucleocapsid is enveloped. Cell death and lysis occur after infection with most rhabdoviruses, with the important exception of rabies virus, which produces little discernible cell damage.
Pathogenesis and Immunity
Only the pathogenesis of rabies virus infection is discussed here (Box 58-2). Rabies infection usually results from the bite of a rabid animal. Rabies infection of the animal causes secretion of the virus in the animal’s saliva and promotes aggressive behavior (“mad” dog), which in turn promotes transmission of the virus. The virus can also be transmitted through the inhalation of aerosolized virus (as may be found in bat caves), in transplanted infected tissue (e.g., cornea), and by inoculation through intact mucosal membranes.
Box 58-2
Disease Mechanisms of Rabies Virus
Rabies is usually transmitted in saliva and is acquired from the bite of a rabid animal.
Rabies virus is not very cytolytic and seems to remain cell associated.
Virus replicates in the muscle at the site of the bite, with minimal or no symptoms (incubation phase).
The length of the incubation phase is determined by the infectious dose and the proximity of the infection site to the central nervous system (CNS) and brain.
After weeks to months, the virus infects the peripheral nerves and travels up the CNS to the brain (prodrome phase).
Infection of the brain causes classic symptoms, coma, and death (neurologic phase).
During the neurologic phase, the virus spreads to the glands, skin, and other body parts, including the salivary glands, from where it is transmitted.
Rabies infection does not elicit an antibody response until the late stages of the disease, when the virus has spread from the CNS to other sites.
Administration of antibody can block the progression of the virus and disease if given early enough.
The long incubation period allows active immunization as a postexposure treatment.
The virus replicates quietly at the site for days to months (Figure 58-2) before progressing to the central nervous system (CNS). Rabies virus travels by retrograde axoplasmic transport to the dorsal root ganglia and to the spinal cord. Once the virus gains access to the spinal cord, the brain becomes rapidly infected. The affected areas are the hippocampus, brainstem, ganglionic cells of the pontine nuclei, and Purkinje cells of the cerebellum. The virus then disseminates from the CNS via afferent neurons to highly innervated sites, such as the skin of the head and neck, salivary glands, retina, cornea, nasal mucosa, adrenal medulla, renal parenchyma, and pancreatic acinar cells. After the virus invades the brain and spinal cord, encephalitis develops and neurons degenerate. Despite the extensive CNS involvement and impairment of CNS function, little histopathologic change can be observed in the affected tissue, other than the presence of Negri bodies (see section on Laboratory Diagnosis).
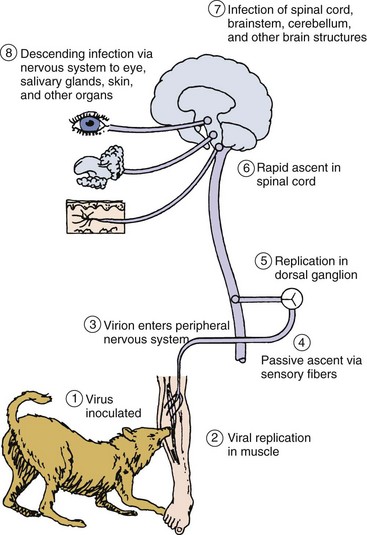
Figure 58-2 Pathogenesis of rabies virus infection. Numbered steps describe the sequence of events.
(Modified from Belshe RB: Textbook of human virology, ed 2, St Louis, 1991, Mosby.)
Rabies is fatal once clinical disease is apparent. The length of the incubation period is determined by (1) the concentration of the virus in the inoculum, (2) the proximity of the wound to the brain, (3) the severity of the wound, (4) the host’s age, and (5) the host’s immune status.
In contrast to other viral encephalitis syndromes, rabies is minimally cytolytic and rarely causes inflammatory lesions. Viral proteins inhibit apoptosis and aspects of interferon action. Neutralizing antibodies are not apparent until after the clinical disease is well established. Little antigen is released, and the infection probably remains hidden from the immune response. Cell-mediated immunity appears to play little or no role in protection against rabies virus infection.
Antibody can block the spread of virus to the CNS and brain if administered or generated during the incubation period. The incubation period is usually long enough to allow generation of a therapeutic protective antibody response after active immunization with the killed rabies vaccine.
Epidemiology
Rabies is the classic zoonotic infection spread from animals to humans (Box 58-3). It is endemic in a variety of animals worldwide, except in Australia. Rabies is maintained and spread in two ways. In urban rabies, dogs are the primary transmitter, and in sylvatic (forest) rabies, many species of wildlife can serve as transmitters. In the United States, rabies is more prevalent in cats, because they are not vaccinated. Virus-containing aerosols, bites, and scratches from infected bats also spread the disease. The principal reservoir for rabies in most of the world, however, is the dog. In Latin America and Asia, this feature is a problem because of the existence of many stray, unvaccinated dogs and the absence of rabies-control programs. These two factors are responsible for thousands of rabies cases in dogs each year in these regions. Although rare, there are cases of rabies transmission via corneal and organ transplants.
Because of the excellent vaccination program in the United States, sylvatic rabies accounts for most of the cases of animal rabies in this country. Statistics for animal rabies are collected by the Centers for Disease Control and Prevention, which in 1999 recorded more than 8000 documented cases of rabies in raccoons, skunks, bats, and farm animals, in addition to dogs and cats (Figure 58-3). Badgers and foxes are also major carriers of rabies in Western Europe. In South America, vampire bats transmit rabies to cattle, resulting in losses of millions of dollars each year.
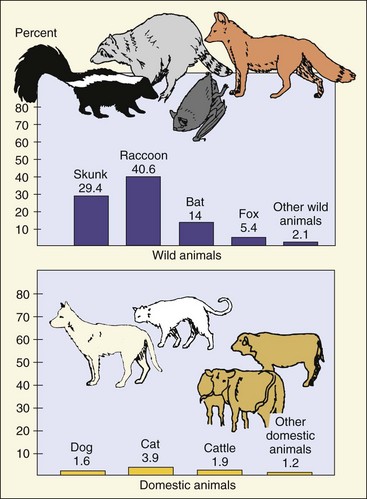
Figure 58-3 Distribution of animal rabies in the United States, 1999. The percentages relate to the total number of cases of animal rabies.
(Data from Krebs JW, Rupprecht CE, Childs JE: Rabies surveillance in the United States during 1999, J Am Vet Med Assoc 217:1799–1811, 2000.)
Although underreported, it is estimated that rabies accounts for 70,000 deaths, mostly children, annually worldwide, with at least 25,000 deaths in India, where the virus is transmitted by dogs in 96% of cases. In Latin America, cases of human rabies primarily result from contact with rabid dogs in urban areas. In Indonesia, an outbreak of more than 200 human cases of rabies in 1999 promoted the killing of more than 40,000 dogs on the islands. The incidence of human rabies in the United States is approximately one case per year, due in large part to effective dog vaccination programs and limited human contact with skunks, raccoons, and bats. Since 1990, human cases of rabies in the United States have been caused primarily by bat variants of the virus. The World Health Organization estimates that 10 million people per year receive treatment after exposure to animals suspected of being rabid.
Clinical Syndromes (Box 58-4)
Rabies is virtually always fatal unless treated by vaccination. After a long but highly variable incubation period, the prodrome phase of rabies ensues (Table 58-1). The patient has symptoms such as fever, malaise, headache, pain or paresthesia (itching) at the site of the bite, gastrointestinal symptoms, fatigue, and anorexia. The prodrome usually lasts 2 to 10 days, after which the neurologic symptoms specific to rabies appear. Hydrophobia (fear of water), the most characteristic symptom of rabies, occurs in 20% to 50% of patients. It is triggered by the pain associated with the patient’s attempts to swallow water. Focal and generalized seizures, disorientation, and hallucinations are also common during the neurologic phase. Fifteen percent to 60% of patients exhibit paralysis as the only manifestation of rabies. The paralysis may lead to respiratory failure.
Box 58-4
Clinical Summary
Rabies: A 3-year-old girl was found to have a bat flying in her bedroom. The bat apparently was there all night. There was no evidence of any bite wound or contact, and the bat was caught and released. Three weeks later, the child developed a change in behavior, becoming irritable and agitated. This state quickly progressed to confusion, uncontrollable thrashing about, and inability to handle her secretions. She eventually became comatose and died from respiratory arrest.
The patient becomes comatose after the neurologic phase, which lasts from 2 to 10 days. This phase almost universally leads to death resulting from neurologic and pulmonary complications.
Laboratory Diagnosis
The occurrence of neurologic symptoms in a person who has been bitten by an animal generally establishes the diagnosis of rabies. Unfortunately, evidence of infection, including symptoms and the detection of antibody, does not occur until it is too late for intervention. Laboratory tests are usually performed to confirm the diagnosis and to determine whether a suspected individual or animal is rabid (postmortem).
The diagnosis of rabies is made through detection of viral antigen in the CNS or skin, isolation of the virus, detection of the genome, and serologic findings. The hallmark diagnostic finding has been the detection of intracytoplasmic inclusions consisting of aggregates of viral nucleocapsids (Negri bodies) in affected neurons (see Chapter 47, Figure 47-3). Although their finding is diagnostic of rabies, Negri bodies are seen in only 70% to 90% of brain tissue from infected humans.
Antigen detection using direct immunofluorescence or genome detection using reverse transcriptase polymerase chain reaction (RT-PCR) are relatively quick and sensitive assays that are the preferred methods for diagnosing rabies. Samples of saliva are easy to test, but serum, spinal fluid, skin biopsy material from the nape of the neck, brain biopsy or autopsy material, and impression smears of corneal epithelial cells can also be examined.
Rabies can also be grown in cell culture or in intracerebrally inoculated infant mice but requires special laboratory isolation procedures and is not routinely performed. Inoculated cell cultures or brain tissues are subsequently examined with direct immunofluorescence.
Rabies antibody titers in serum and cerebrospinal fluid are usually measured by enzyme-linked immunosorbent assay (ELISA) or a rapid fluorescent focus inhibition test. Antibody usually is not detectable until late in the disease, however.
Treatment and Prophylaxis
Clinical rabies is almost always fatal unless treated with postrabies immunization. Once the symptoms have appeared, little other than supportive care can be given. There is one case of successful cessation of disease progression by postexposure ribavirin treatment (see introductory case study).
Postexposure prophylaxis is the only hope for preventing overt clinical illness in the affected person. Although human cases of rabies are rare, approximately 20,000 people receive rabies prophylaxis each year in the United States alone. Prophylaxis should be initiated for anyone exposed by bite or by contamination of an open wound or mucous membrane to the saliva or brain tissue of an animal suspected to be infected with the virus, unless the animal is tested and shown not to be rabid.
The first protective measure is local treatment of the wound. The wound should be washed immediately with soap and water or another substance that inactivates the virus. The World Health Organization Expert Committee on Rabies also recommends the instillation of antirabies serum around the wound.
Subsequently, immunization with vaccine in combination with administration of one dose of human rabies immunoglobulin (HRIG) or equine antirabies serum is recommended. Passive immunization with HRIG provides antibody until the patient produces antibody in response to the vaccine. A series of five vaccinations is then administered over the course of a month. The slow course of rabies disease allows active immunity to be generated in time to afford protection.
The rabies vaccine is a killed-virus vaccine prepared through the chemical inactivation of rabies infected–tissue culture human diploid cells (HDCV) or fetal rhesus lung cells. These vaccines cause fewer negative reactions than the older vaccines (Semple and Fermi), which were prepared in the brains of adult or suckling animals. The HDCV is administered intramuscularly on the day of exposure and then on days 3, 7, 14, and 28 or intradermally with a lower dose of vaccine to multiple sites on days 0, 3, 7, 28, and 90. Preexposure vaccination should be performed on animal workers, laboratory workers who handle potentially infected tissue, and people traveling to areas where rabies is endemic. HDCV administered intramuscularly or intradermally in three doses is recommended and provides 2 years of protection.
Ultimately the prevention of human rabies hinges on the effective control of rabies in domestic and wild animals. Its control in domestic animals depends on the removal of stray and unwanted animals and the vaccination of all dogs and cats. A variety of attenuated oral vaccines have also been used successfully to immunize foxes. A live recombinant vaccinia virus vaccine expressing the rabies virus G protein is in use in the United States. This vaccine, which is injected into bait and parachuted into the forest, successfully immunizes raccoons, foxes, and other animals. Accidental injection of a woman with this vaccinia-rabies hybrid vaccine resulted in immunization against both smallpox and rabies viruses (see Bibliography).
Filoviruses
The Marburg and Ebola viruses (Figure 58-4) were classified as members of the family Rhabdoviridae but are now classified as filoviruses (Filoviridae). They are filamentous, enveloped, negative-strand RNA viruses. These agents cause severe or fatal hemorrhagic fevers and are endemic in Africa. Awareness of the Ebola virus increased after an outbreak of the disease in Zaire in 1995, in Gabon in 1996, and also after the release of the movie “Outbreak,” based on the book by Robin Cook, and the book The Hot Zone by Richard Preston.
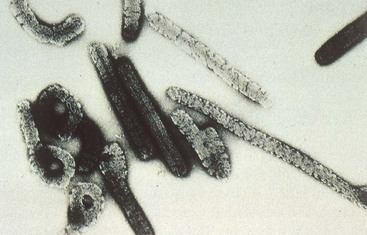
Figure 58-4 Electron micrograph of the Ebola virus.
(Courtesy Centers for Disease Control and Prevention, Atlanta.)
Structure and Replication
Filoviruses have a single-stranded RNA genome (4.5 × 106 Da) that encodes seven proteins. The virions form enveloped filaments with a diameter of 80 nm but may also assume other shapes. They vary in length from 800 nm to as long as 1400 nm. The nucleocapsid is helical and enclosed in an envelope containing one glycoprotein. The Ebola virus binds to the Niemann-Pick C1 (NPC1) protein enters the cell and replicates in the cytoplasm like the rhabdoviruses.
Pathogenesis
The filoviruses replicate efficiently, producing large amounts of virus in monocytes, macrophage, dendritic cells, and other cells. Replication in monocytes elicits a cytokine storm of proinflammatory cytokines similar to a superantigen induced cytokine storm. Viral cytopathogenesis causes extensive tissue necrosis in parenchymal cells of the liver, spleen, lymph nodes, and lungs. The breakdown of endothelial cells leading to vascular injury can be attributed to the Ebola glycoproteins. Strains with mutations in the glycoprotein gene lack the hemorrhagic component of disease. The widespread hemorrhage that occurs in affected patients causes edema and hypovolemic shock. The virus can also evade host innate and immune responses. A small soluble glycoprotein is shed, can inhibit neutrophil activation, and block antibody action. The viral proteins can also inhibit interferon production and action.
Epidemiology
Marburg virus infection was first detected among laboratory workers in Marburg, Germany, who had been exposed to tissues from apparently healthy African green monkeys. Rare cases of Marburg virus infection have been seen in Zimbabwe and Kenya.
Ebola virus was named for the river in the Democratic Republic of Congo (formerly Zaire) where it was discovered. Outbreaks of Ebola virus disease have occurred in the Democratic Republic of Congo and Sudan. During an outbreak, the Ebola virus is so lethal that it eliminates the susceptible population before it can be spread extensively. Since 1976, when the virus was discovered, approximately 1850 cases and more than 1200 deaths have occurred. However, in rural areas of central Africa, as much as 18% of the population have antibody to this virus, indicating that subclinical infections do occur.
These viruses may be endemic in bats or wild monkeys and can be spread to humans and between humans. Contact with the animal reservoir or direct contact with infected blood or secretions can spread the disease. These viruses have been transmitted by accidental injection and through the use of contaminated syringes. Health care workers tending the sick and monkey handlers may be at risk.
Clinical Syndromes
Marburg and Ebola viruses are the most severe causes of viral hemorrhagic fevers (Clinical Case 58-1). The illness usually begins with flulike symptoms, such as headache and myalgia. Nausea, vomiting, and diarrhea occur within a few days; a rash also may develop. Subsequently, hemorrhage from multiple sites (especially the gastrointestinal tract) and death occur in as many as 90% of patients with clinically evident disease. The 1995 outbreak in Kikwit, Congo, killed 245 people.
Clinical Case 58-1
Ebola
Emond and associates described the following case of Ebola infection (Br Med J 2:541–544, 1977). Within 6 days of a needle-stick accident, while handling animal liver infected with Ebola virus, a scientist complained of abdominal pain and nausea. He was transferred to a high-security infectious disease unit and placed in an isolation room. At admission (day 1), he was experiencing fatigue, anorexia, nausea, abdominal pain, and had a fever of 38° C. Interferon was administered twice a day, and it appeared to have worked, except that the next morning his fever returned (39° C). He was given heat-inactivated convalescent serum with no immediate effect. On day 4, he sweated profusely, and his temperature dropped to normal, but he had a new rash on his chest. At midday of day 4, he experienced sudden, violent shivering, fever of 40° C, nausea, vomiting, and diarrhea. These symptoms continued for 3 days, with spread of the rash across his body. On day 6, more convalescent serum and rehydration treatment were administered. The patient made a slow recovery over the next 10 weeks. Virus, as detected by electron microscopy and by inoculation of guinea pigs, was present in his blood from the first day of symptoms. (Currently, the analysis would be performed by reverse transcriptase polymerase chain reaction, with less risk to laboratory personnel.) Virus titers dropped by 1000-fold after interferon treatment and were undetectable by day 9. Treatment of the patient and handling of samples were performed under the strictest isolation conditions available at the time. Even though the scientist took precautions and soaked his hand in bleach as soon as possible, his fate was already sealed. Luckily, interferon therapy and convalescent serum were available to limit the extent of disease progression. In their absence, he would have died from a rapidly progressing hemorrhagic disease.
Laboratory Diagnosis
All specimens from patients with a suspected filovirus infection must be handled with extreme care to prevent accidental infection. Handling of these viruses requires level 4 isolation procedures that are not routinely available. Marburg virus may grow rapidly in tissue culture (Vero cells), but animal (e.g., guinea pig) inoculation may be necessary to recover Ebola virus.
Infected cells have large eosinophilic cytoplasmic inclusion bodies. Viral antigens can be detected in tissue by direct immunofluorescence analysis and in fluids by ELISA. RT-PCR amplification of the viral genome in secretions can be used to confirm the diagnosis and minimize handling of samples.
Immunoglobulin G (IgG) and IgM antibody to filovirus antigens can be detected by immunofluorescence or ELISA.
Treatment, Prevention, and Control
Antibody-containing serum and interferon therapies have been tried in patients with filovirus infections. Infected patients should be quarantined, and contaminated animals should be sacrificed. Handling of the viruses or contaminated materials requires very stringent (level 4) isolation procedures.
Borna Disease Virus
Borna disease virus (BDV) is the only member of a family of enveloped, negative-strand RNA viruses. BDV was first associated with infection of horses in Germany. The virus has received considerable recent interest because of its association with specific neuropsychiatric diseases, such as schizophrenia.
Structure and Replication
The 8910-nucleotide–long genome of BDV encodes five detectable proteins, including a polymerase (L), nucleoprotein (N), phosphoprotein (P), matrix protein (M), and envelope glycoprotein (G). Unlike most negative-strand viruses, BDV replicates in the nucleus. Although this is similar to the orthomyxoviruses, BDV differs in that its genome is unsegmented. Also unusual for an RNA virus, one of the positive-strand RNAs that is transcribed from the genome is processed to remove introns to produce three mRNAs for three different proteins.
Pathogenesis
BDV is highly neurotropic and capable of spreading throughout the CNS. BDV also infects parenchymal cells of different organs and peripheral blood mononuclear cells. The virus is not very cytolytic and establishes a persistent infection in the infected individual. T-cell immune responses are important for controlling BDV infections but also contribute to tissue damage leading to disease.
Clinical Syndromes
Although there is limited understanding of the BDV disease in humans, infection of animals can result in subtle losses of learning and memory and in fatal immune-mediated meningoencephalitis. Many of the outcomes of BDV infection of laboratory animals resemble human neuropsychiatric diseases, including depression, bipolar disorder, schizophrenia, and autism. The presence of antibodies to the virus and/or infected peripheral blood mononuclear cells in higher-than-background numbers of patients with schizophrenia, autism, and other neuropsychiatric diseases suggest that BDV either causes or exacerbates these mental illnesses.
Epidemiology
BDV is a zoonose capable of infecting many different mammalian species, including horses, sheep, and humans. Most outbreaks of the virus have occurred in central Europe, but the virus has also been detected in North America and Asia. Neither the reservoir nor the mode of transmission of BDV is known. Higher levels of infection of humans are present where outbreaks in horses have been observed.
An 11-year-old boy was brought to a hospital in California after falling; his bruises were treated, and he was released. The following day, he refused to drink water with his medicine, and he became more anxious. That night he began to act up and hallucinate. He also was salivating and had difficulty breathing. Two days later, he had a fever of 40.8° C (105.4° F) and experienced two episodes of cardiac arrest. Although rabies was suspected, no remarkable data were obtained from a computed tomographic image of the brain or cerebrospinal fluid analysis. A skin biopsy from the nape of the neck was negative for viral antigen on day 3 but was positive for rabies on day 7. The patient’s condition continued to deteriorate, and he died 11 days later. When the parents were questioned, it was learned that 6 months earlier, the boy had been bitten on the finger by a dog while on a trip to India.
1. What clinical features of this case suggested rabies?
2. Why does rabies have such a long incubation period?
3. What treatment should have been given immediately after the dog bite? What treatment should be given as soon as the diagnosis was suspected?
4. How do the clinical aspects of rabies differ from those of other neurologic viral diseases?
1. Rabies is suggested by the boy’s refusal to drink (hydrophobia), hallucinations, anxiety, salivation, difficulty breathing, and fever.
2. Rabies has a long incubation period because it is not very cytolytic, and once it enters the neuron, it is relatively hidden from immune responses. The characteristic disease signs occur only when the virus has reached the brain and started to replicate and cause damage.
3. Immediately after the dog bite, the child should have been injected with rabies-specific immune globulin as close to the site as possible. A course of immunization with the inactivated rabies vaccine should also be initiated as soon as possible.
4. Unlike other neurologic viral diseases, rabies infection is undetectable until it reaches the brain (too late for treatment), and then it infects the salivary gland, causing painful swallowing and potential infection of others.