Antifungal and Antiviral Agents
1 Name several types of antifungal agents and discuss their indications in dentistry and potential adverse reactions.
2 Discuss the treatment of herpes simplex.
3 Describe the various drugs and drug combinations used to treat acquired immunodeficiency syndrome.
The antibiotics and antiinfectives discussed in Chapter 7 are effective against a certain spectrum of organisms: bacteria, protozoa, rickettsia, trichomonads, amoebas, and spirochetes. These agents are not effective against either fungal or viral infections. This chapter discusses treatment and management of fungal and viral infections commonly encountered in the dental office: the fungus Candida albicans (candidiasis or thrush) and the herpes simplex virus. The viral infection, acquired immunodeficiency syndrome (AIDS), and the different types of hepatitis (A, B, C, D, Ex) are discussed with reference to factors that might affect dental treatment. Other fungal and viral infections that are common in the population are briefly mentioned.
ANTIFUNGAL AGENTS
Although fungal infections are not often encountered in dental practice, when they are present, they are often difficult to treat. Unlike bacterial infections, fungal infections are more insidious. Fungal infections are more likely to occur in patients who are immunocompromised, and these infections can become chronic. Fungal infections can be divided into those that affect primarily the skin or mucosa (mucocutaneous) and those that affect the whole body (systemic). The dental health care worker usually treats skin or mucosal lesions, most commonly within the mouth. These mucosal lesions may be treated with a topical or systemic antifungal agent.
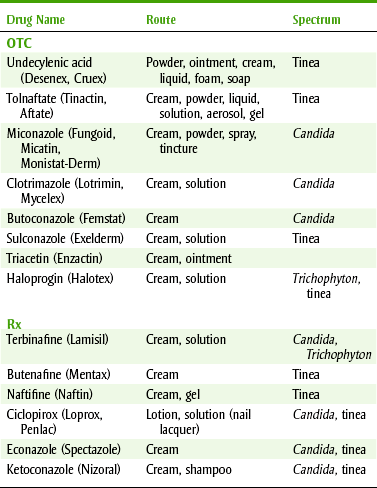
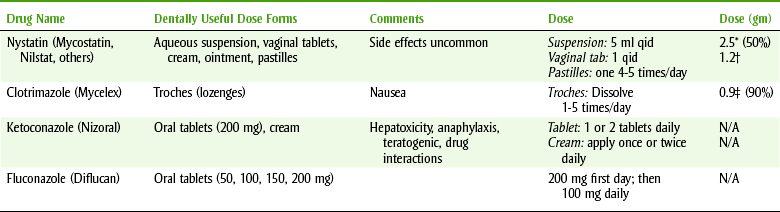
Although there are different groups of fungi, two common groups are candida-like and tinea. Dental health care workers manage mucocutaneous candidal infections, primarily caused by C. albicans, with nystatin, clotrimazole, ketoconazole, or fluconazole (Table 8-1). Infections with tinea affect the skin and produce athlete’s foot, “jock itch,” and ringworm. Both over-the-counter (OTC) and prescription products are used to manage these conditions topically (Table 8-2).
TABLE 8-1
DENTALLY USEFUL ANTIFUNGAL AGENTS FOR ORAL CANDIDIASIS
N/A, Not available; qid, four times a day.
*Sucrose.
†0.4 gm sucrose + 0.8 gm glucose.
‡Glucose.
Mucocutaneous candidal infections commonly occur in the vaginal canal. If the patient can recognize the symptoms (by having had a previous infection), an antifungal OTC product can be purchased and used.
Systemic mycoses produced by fungi include aspergillosis, blastomycosis, coccidioidomycosis, cryptococcosis, histoplasmosis, mucormycosis, and paracoccidioidomycosis. Chromomycosis, mycetoma, and sporotrichosis may progress to deep mycotic infections. These serious infections are medical management situations beyond the scope of this chapter. Figure 8-1 provides comparative acquisition.
Nystatin
Nystatin (nye-STAT-in) (Mycostatin, Nilstat) is a prescription antifungal agent that is produced by Streptomyces noursei. Its mechanism of action involves binding to sterols in the fungal cell membrane. This produces an increase in membrane permeability and allows leakage of potassium and other essential cellular constituents. Because bacteria do not contain sterols in their cell membranes, nystatin is not active against these organisms.
Nystatin is not absorbed from the mucous membranes or through intact skin; taken orally, it is poorly absorbed from the gastrointestinal tract. In usual therapeutic doses, blood levels are not detectable. When administered orally, it is not absorbed but is excreted unchanged in the feces. Nystatin is fungicidal and fungistatic against a variety of yeasts and fungi. In vitro, nystatin inhibits C. albicans and some other species of Candida.
The adverse reactions associated with nystatin are minor and infrequent. Applied topically or taken orally (through the gastrointestinal tract), there is little if any absorption. When higher doses have been used, nausea, vomiting, and diarrhea have occasionally occurred. Rarely, hypersensitivity reactions have been reported.
Nystatin is used for both the treatment and the prevention of oral candidiasis in susceptible cases. Although C. albicans is a frequent inhabitant of the oral cavity, only under unusual conditions does it produce disease. Often, patients affected are immunocompromised.
For the treatment of oral candidiasis, nystatin is available (see Table 8-1) in the form of an aqueous suspension (100,000 U/ml) containing 50% sucrose. The directions to the patient are to swish, swirl, and spit or swallow 5 ml (1 tsp) four times daily. The suspension should remain in the mouth 2 minutes for the best effect. For infants and small children with thrush, half of a dropperful (2.5 ml) is placed in each side of the mouth and rubbed into the recesses of the mouth and on the lesions. If swallowed, diabetics using this suspension must take the sugar content into account (2.5 gm sucrose per tsp) when planning their meals and insulin use.
Nystatin pastilles are licorice-flavored, are rubbery, and also contain sugar. Informal feedback has indicated that patients with xerostomia might not find this dose form acceptable. The advantage of this preparation is that it takes 15 minutes for the lozenge to dissolve in the mouth, thus bathing lesions in the antifungal agent for a longer period. It is allowed to dissolve in the mouth four times daily. The dental health care worker must discuss patients’ oral health habits, especially when patients are chronically ingesting these cariogenic agents.
Nystatin is available in vaginal tablets for use in vaginal infections. Occasionally, these vaginal tablets can be used orally. They are dissolved in the mouth four times daily. The advantages of the vaginal tablet used as a lozenge are that the drug remains in contact with the infected oral mucosa longer than it does when in the suspension form and it contains no sugar. The disadvantage is that it is not flavored for oral use.
Patients should be instructed to use the nystatin product for 10 to 14 days, depending on the severity of the infection, or for 48 hours after the symptoms have subsided and cultures have returned negative. Cultures are typically not performed. Some patients, especially if immunocompromised, may require long-term prophylactic antifungal agents to control candidiasis.
Imidazoles
Imidazoles useful in dentistry include clotrimazole, miconazole, and ketoconazole.
 CLOTRIMAZOLE
CLOTRIMAZOLE
Clotrimazole (kloe-TRIM-a-zole) (Mycelex) is a synthetic antifungal agent available in the form of a slowly dissolving, sugar-containing lozenge for oral use. It is also available as an OTC cream for topical application to the skin or vaginal canal.
Clotrimazole’s mechanism of action involves alteration of cell membrane permeability. It binds with the phospholipids in the cell membrane of the fungus. As a result of the alteration in permeability, the cell membrane loses its function and the cellular constituents are lost.
Clotrimazole oral lozenges dissolve in approximately 15 to 30 minutes. Patients with xerostomia may have difficulty dissolving this product. Saliva concentrations that are sufficient to inhibit most Candida species are maintained in the mouth for about 3 hours. The drug is bound to the oral mucosa, from which it is slowly released. The amount of clotrimazole absorbed systemically by this route is unknown, but some absorption occurs. Each lozenge also contains 0.9 gm of glucose. The spectrum of action of clotrimazole is primarily against the Candida species.
The most common adverse reactions associated with clotrimazole involve the gastrointestinal tract, including abdominal pain, diarrhea, and nausea. Clotrimazole has been reported to produce elevated liver enzyme (aspartate aminotransferase) levels in approximately 15% of patients.
Systemic clotrimazole has been assigned to Food and Drug Administration (FDA) pregnancy category C. Very high doses have been embryotoxic in rats and mice. High doses have caused impaired mating and a decrease in both the number and survival of the young. No teratogenic effects have been found in several other species tested. No carcinogenicity has been demonstrated in rats.
Clotrimazole is indicated for the local treatment of oropharyngeal candidiasis. Patients should be instructed to dissolve the lozenge in the mouth slowly, like a cough drop, to minimize gastrointestinal discomfort. They should also be told to take all of the medication prescribed to minimize relapse. The usual adult dosage is 1 lozenge (10 mg) five times daily for 10 to 14 days (or longer for immunosuppressed patients) or for 48 hours after the symptoms have cleared. Some clinicians advocate dissolving one 100-mg clotrimazole (Mycelex) vaginal tablet four times daily in the oral cavity, like a lozenge or troche. The advantages of the vaginal tablet used as a lozenge are that the drug remains in contact with the infected oral mucosa longer than it does when in the suspension form and it contains no sugar. The disadvantage is that it is not flavored for oral use.
 KETOCONAZOLE
KETOCONAZOLE
Ketoconazole (kee-toe-KON-a-zole) (Nizoral), another imidazole used in dentistry, alters cellular membranes and interferes with intracellular enzymes. By interfering with the synthesis of ergosterol, a cellular component of fungi, membrane permeability is altered and purine transport inhibited. The imidazoles inhibit the C-14 demethylation of lanosterol, an ergosterol precursor. It also inhibits sex steroid biosynthesis, including testosterone, perhaps by blocking several P-450 enzyme steps.
Pharmacokinetics: For the adequate systemic absorption of ketoconazole, an acidic environment is required. Patients with achlorhydria should take ketoconazole with hydrochloride acid (use straw to minimize damage to teeth). Medications that interfere with the normal production of stomach acid, such as H2-blockers or H2-receptor antagonists (H2-RA) and proton pump inhibitors, reduce the absorption of ketoconazole. If ketoconazole must be taken with drugs that reduce the stomach acid, the ketoconazole should be taken as long a time as possible before or after the acid-reducing drug. All imidazole antifungal agents require an acidic environment for optimal absorption. With the exception of the cerebrospinal fluid (CSF), it is well distributed in humans. It crosses the placenta and is excreted in breast milk. The peak serum concentration occurs between 1 and 4 hours after administration. Ketoconazole is metabolized in the liver, and approximately 13% is excreted by the kidney, with a half-life between 2 and 8 hours. Because of the small contribution of the kidney to the excretion of ketoconazole, patients with renal impairment do not generally require a reduction in their dose. Because the primary route of excretion for ketoconazole is biliary, patients with hepatic impairment may require a lower dose.
Spectrum: Ketoconazole is effective against a wide variety of fungal infections. It is indicated in many systemic fungal infections, including blastomycosis, candidiasis, coccidioidomycosis, and histoplasmosis. Although effective against organisms causing tinea, it is not the drug of choice unless traditional agents have failed.
Gastrointestinal Effects: The most frequent adverse reactions (3% to 10%) associated with ketoconazole are nausea and vomiting, which can be minimized by taking ketoconazole with food.
Hepatotoxicity: The most serious adverse reaction associated with ketoconazole is hepatotoxicity. Its incidence is at least 1 : 10,000. It is usually reversible on discontinuation of the drug, but occasionally it has been fatal. It is thought to be an idiosyncratic reaction that can happen at any time. With extended use, the patient should have periodic liver function tests (LFTs). Patients taking other hepatotoxic agents, those with liver disease (e.g., alcoholic hepatitis), or those on prolonged therapy should be watched closely because they may be more susceptible to this hepatotoxicity.
Ketoconazole, in higher doses, inhibits the secretion of the corticosteroids and lowers the serum levels of testosterone. In men, this effect can produce gynecomastia and impotence. This adverse effect is specific to ketoconazole.
Other Effects: Other adverse reactions reported include headache, dizziness, drowsiness, photophobia, skin rash or pruritus, and insomnia. Fever, chills, dyspnea, tinnitus, arthralgias, and thrombocytopenia have occurred in a few patients. When ketoconazole is applied topically, irritation, pruritus, and stinging are the most commonly reported side effects.
Drug Interactions: Ketoconazole has many drug interactions that have been reported in the literature. Because an acidic environment is required for dissolution and absorption of ketoconazole, agents that alter the amount of stomach acid could theoretically reduce the absorption of ketoconazole (H2-RA, H+-pump inhibitors, anticholinergic agents, and antacids). At least 2 hours should elapse between the ingestion of these agents and ketoconazole’s administration.
Ketoconazole inhibits the cytochrome P (CYP)-450 3A4 hepatic microsomal isoenzyme, which can produce drug interactions with many other drugs also metabolized by this isoenzyme. Ketoconazole can increase the blood levels of cyclosporine, warfarin, corticosteroids, phenytoin, digoxin, lovastatin, and simvastatin, to name a few.
Isoniazid, phenytoin, and theophylline can decrease ketoconazole serum levels. Ketoconazole should not be used with rifampin because rifampin renders its blood level undetectable. Ketoconazole may decrease the effect of oral contraceptives; an alternative method of birth control should be suggested. Ketoconazole may produce a disulfiram-like reaction or enhance hepatotoxicity with alcohol.
Dental: Ketoconazole is indicated in the treatment and management of mucocutaneous and oropharyngeal candidiasis (oral thrush). It can be used prophylactically in chronic mucocutaneous candidiasis. Because of its adverse reaction profile, ketoconazole should be used only after topical antifungal agents have been ineffective or if there is reason to believe that they will be ineffective.
Dose: The usual adult dose of ketoconazole for the treatment of Candida species is 200 to 400 mg orally (PO) daily (qd). It should be used for at least 2 weeks, and 6 to 12 months may be required for chronic mucocutaneous candidiasis. Maintenance therapy may be necessary for certain patients. Ketoconazole is available for topical administration in a 2% aqueous vehicle (cream) for tinea or candidal infections. It is applied once or twice daily for at least 2 weeks. Ketoconazole (Nizoral) shampoo is used twice weekly to treat dandruff, a condition caused by the fungus.
 OTHER IMIDAZOLES
OTHER IMIDAZOLES
Other imidazoles, such as fluconazole (floo-KON-a-zole) (Diflucan), an oral triazole antifungal agent, are used to treat certain fungal infections. Fluconazole prevents the synthesis of ergosterol in the fungal cell membranes by inhibiting fungal CYP-450 enzymes. Phospholipids and unsaturated fatty acids accumulate in the fungal cells.
Fluconazole is indicated for treatment of oropharyngeal and esophageal candidiasis and serious systemic candidal infections. One tablet of fluconazole is now indicated to treat vaginal candidiasis. Fluconazole is used prophylactically against candidiasis in immunocompromised patients or for the treatment of candidal infections that do not respond to other agents.
Itraconazole (it-ra-KON-a-zole) (Sporanox), another systemic imidazole, is used for blastomycosis, histoplasmosis, and aspergillosis. It is the first antifungal agent to be effective in the treatment of onychomycosis of the toenail or fingernail (pulse therapy is used, i.e., on-off-on-off).
Other Antifungal Agents
An important agent in the treatment of many serious systemic fungal infections is amphotericin B (am-foe-TER-i-sin) (Fungizone). Because of its side effects, it has earned the nickname “amphoterrible.” It must be administered parenterally because it is poorly absorbed from the intestinal tract.
Amphotericin is produced by Streptomyces nodosus. It binds to the sterols in the fungus cell membrane, altering membrane permeability and allowing the loss of potassium and small molecules from the cells.
The spectrum of amphotericin includes many fungi, such as certain strains of Aspergillus, Paracoccidioides, Coccidioides, Cryptococcus, Histoplasma, Mucor, and Candida organisms. It is also effective against the protozoa Leishmania.
The adverse reactions associated with amphotericin are wide ranging and potentially serious, but it is often the only effective treatment for certain serious systemic fungal infections. Most patients experience hypokalemia, headache, chills (50%), fever, malaise, muscle and joint pain, gastric complaints, and nephrotoxicity (80%). Amphotericin has many potentially serious drug interactions.
Topical amphotericin has produced burning, itching, and in rare cases an allergic contact dermatitis. It is available as a 3% cream or ointment. The parenteral forms of amphotericin include liposomal and cholesteryl forms.
 GRISEOFULVIN
GRISEOFULVIN
Griseofulvin (gri-see-oh-FUL-vin) (Fulvicin P/G, Grisactin Ultra, Gris-PEG) is an antibiotic produced by Penicillium griseofulvum. Its antifungal action is produced by disrupting the cell’s mitotic spindle structure and arresting cell division in metaphase (Figure 8-2). Unlike many drugs, griseofulvin’s absorption is enhanced by taking it with a fatty meal. It is tightly bound and preferentially deposited in diseased keratin precursors (hair, nails, and skin). Its spectrum includes tinea (e.g., ringworm), Trichophyton, Microsporum, and Epidermophyton species but does not include Candida organisms.
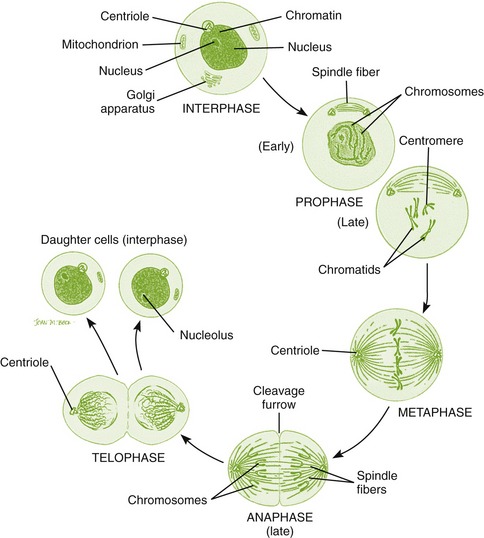
FIGURE 8-2 The process of mitosis. Note the mitotic spindle structure formed during cell division. The drug griseofulvin disrupts this structure and arrests cell division to produce its antifungal action. (From Thibodeau GA, Patton KT: Structure & function of the body, ed 13, St Louis, 2008, Mosby.)
The adverse reactions of griseofulvin include headache, gastrointestinal complaints, and overgrowth of Candida organisms in the oral cavity (thrush). Hypersensitivity reactions include urticaria, photosensitivity, and lupuslike reactions. The possibility of some cross-sensitivity with penicillins should be considered because the organism that makes griseofulvin is in a family related to penicillin. Depression of hematopoietic functions and carcinogenicity in animals have been demonstrated. It can also produce a disulfiram-like reaction.
Griseofulvin is indicated in the treatment of susceptible infections of the skin, hair, and nails. Because the drug is deposited only in the growing tissues, the duration of treatment depends on the time it takes for the affected area to completely grow out, which may be from 2 weeks to 8 months. Although there is no known use in dentistry, the side effects of griseofulvin, hematopoietic suppression and oral candida infection, must be considered when a dental patient is taking this drug.
ANTIVIRAL AGENTS
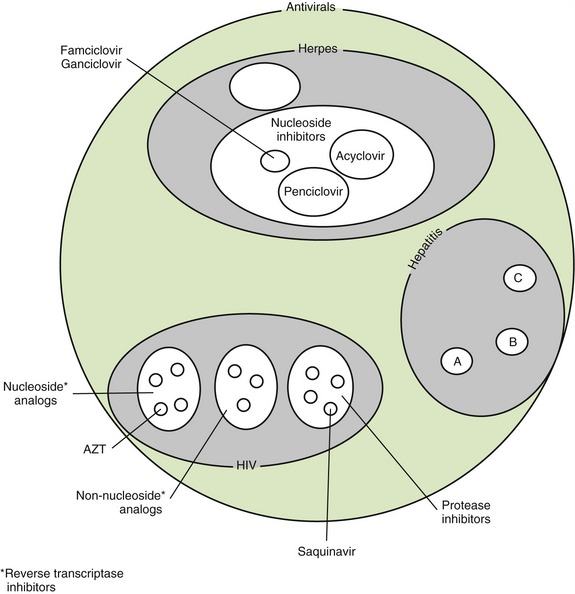
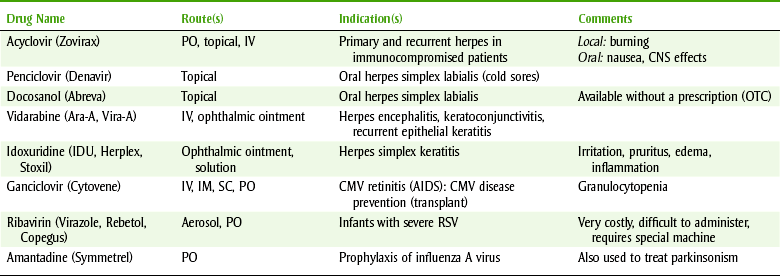
The search for drugs useful in the treatment of viral infections has posed the greatest problem of all infectious organisms. This is because viruses are obligate intracellular organisms that require cooperation from their host’s cells. Therefore to kill the virus, often the host’s cell must also be harmed. The herpes virus, because of the location of the lesions around the oral cavity or in some cases on the dentist’s or hygienist’s finger (herpetic whitlow), has been of the most interest to the dental health care worker. Now, with the symptoms of AIDS being seen clinically in the mouth, the treatment of this virus takes on more importance. Table 8-3 lists some antiviral agents along with their routes of administration and indications. Figure 8-3 separates the antivirals into related groups.
TABLE 8-3
ANTIVIRAL AGENTS, EXCLUDING HUMAN IMMUNODEFICIENCY VIRUS DRUGS
AIDS, Acquired immunodeficiency syndrome; CMV, cytomegalovirus; CNS, central nervous system; IM, intramuscular; IV, intravenous; OTC, over-the-counter; PO, orally; RSV, respiratory syncytial virus; SC, subcutaneous.
Herpes Simplex
Herpes viruses are associated with “cold sores,” and dental practitioners are asked for “something to help.” Most antiviral agents are either purine or pyrimidine analogs that inhibit DNA synthesis (Figure 8-4 and Color Plates 1 and 2).
 ACYCLOVIR
ACYCLOVIR
Acyclovir (ay-SYE-kloe-veer) (Zovirax) is a purine nucleoside that works by inhibiting deoxyribonucleic acid (DNA) replication. It is much less toxic to normal uninfected cells because it is preferentially taken up by infected cells. In the host’s cells, acyclovir is only minimally phosphorylated. This explains its excellent adverse reaction profile.
Pharmacokinetics: When acyclovir is taken orally, between 15% and 30% is absorbed. Peak concentrations occur within about 2 hours. Food does not affect the drug’s absorption. Acyclovir is distributed widely throughout the body. Approximately 10% of a dose of acyclovir is metabolized in the liver.
Spectrum: The antiviral action of acyclovir includes various herpesviruses, including herpes simplex types 1 and 2 (HSV-1 and HSV-2), varicella-zoster, Epstein-Barr, Herpesvirus simiae (B virus), and cytomegalovirus. Several mechanisms of resistance to acyclovir have been found.
Adverse Reactions: The type and extent of the adverse reactions experienced depend on the route of administration of acyclovir.
Topical Administration: When administered topically, acyclovir produces burning, stinging, or mild pain in about one-third of patients. Itching and skin rash have also been reported.
Oral Administration: One of the most common adverse effects associated with oral acyclovir is headache (13%). Other central nervous system (CNS) effects include vertigo, dizziness, fatigue, insomnia, irritability, and mental depression. Oral acyclovir also commonly produces gastrointestinal adverse reactions, including nausea, vomiting, and diarrhea. Anorexia and a funny taste in the mouth have also been reported rarely. Other side effects associated with oral acyclovir include acne, accelerated hair loss, arthralgia, fever, menstrual abnormalities, sore throat, lymphadenopathy, thrombophlebitis, edema, muscle cramps, leg pain, and palpitation.
Parenteral Administration: With parenteral administration, local reactions at the injection site are the most common side effects reported; these include irritation, erythema, pain, and phlebitis. Because acyclovir can precipitate in the renal tubules, it can occasionally affect the blood urea nitrogen or serum creatine levels. Symptoms of encephalopathy, including lethargy, obtundation, tremors, confusion, hallucination, agitation, seizures, and coma, have been reported in about 1% of patients given parenteral acyclovir.
Topical: The indications for topical acyclovir include initial herpes genitalis and limited non–life-threatening initial and recurrent mucocutaneous herpes simplex (HSV-1 and HSV-2) in immunocompromised patients. Topical acyclovir has not been effective in the treatment of recurrent herpes genitalis or herpes labialis infections in nonimmunocompromised patients. It does produce a limited shortening in the duration of viral shedding in males by a few hours. It does not prevent the transmission of infection, and it does not prevent recurrence. Although available literature does not support the use of topical acyclovir for management of herpes labialis in dentistry, it is used extensively. No acyclovir products are approved for the treatment of recurrent herpes labialis in the immunocompetent patient.
Oral: The oral form of acyclovir is indicated in the treatment of initial herpes genitalis and management of recurrent herpes genitalis infections in both immunocompromised and nonimmunocompromised patients. It is effective in the prophylaxis of recurrent herpes genitalis infections in both patient groups. It is not indicated for the suppression of recurrent herpes genitalis in patients with mild infections. In the treatment of herpes labialis, oral acyclovir has shown unimpressive results. Even with hundreds of patients in some studies, only a small difference in a few measured parameters is seen. Higher doses may prove to be effective.
Injectable: The parenteral form of acyclovir is used for severe initial herpes genitalis infections in the nonimmunocompromised patient. It is also indicated for the treatment of initial and recurrent mucocutaneous herpes simplex infections in the immunocompromised patient. Other uses include herpes zoster and varicella-zoster treatment.
Dose: The usual oral adult dosage of acyclovir for the treatment of initial genital herpes is 200 mg every 4 hours (q4h) while the patient is awake five times daily for 10 days. Treatment should be started as soon as the prodromal stage is noticed. The prophylactic dosage for recurrent episodes is 400 mg twice a day (bid) not to exceed 12 months. Some patients may need up to 200 mg five times daily. For intermittent therapy of recurrent episodes, the dose is 200 mg q4h five times a day for 5 days. This is the common dose that has been studied in herpes labialis. Acyclovir has not been shown to effectively treat herpes labialis in topical, tablet, or capsule forms. The slight decrease in crusting or pain does not warrant its use. A larger dose is used in the treatment of herpes zoster (shingles) or chickenpox. It does not prevent the postherpetic neuralgia produced by shingles.
 DOCOSANOL 10%
DOCOSANOL 10%
Docosanol 10% (Abreva), available topically and without a prescription, has been shown to decrease healing time by about a half day in patients with recurrent orolabial herpes when started within 12 hours of the appearance of prodromal symptoms. The advantage of docosanol 10% is that it is available without a prescription.
 PENCICLOVIR
PENCICLOVIR
Penciclovir (pen-CY-klo-veer) (Denavir), available topically, has been shown to reduce both the duration of the lesion and the pain of the lesions on the lips and face associated with both primary and recurrent herpes simplex. The advantages of penciclovir over acyclovir are that it can achieve a higher concentration within the cell and that the drug remains in the cells longer.
 FAMCICLOVIR
FAMCICLOVIR
Famciclovir (fam-CY-klo-veer) and valacyclovir (val-a-CY-klo-veer) are prodrugs converted to penciclovir and acyclovir, respectively, as they pass through the intestinal wall. They are indicated in the treatment of recurrent episodes of genital herpes. They have not been studied for use in herpes labialis. Organisms that have resistance to acyclovir often have cross-resistance with these other agents. Famciclovir and valacyclovir are indicated for acute localized varicella-zoster infections. Intravenous ganciclovir is indicated for serious cytomegalovirus retinitis in immunocompromised patients.
Acquired Immunodeficiency Syndrome
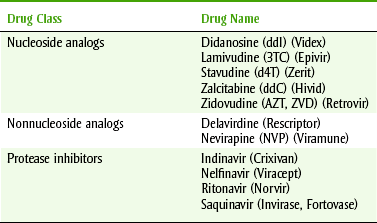
AIDS is the disease produced by infection with the retrovirus human immunodeficiency virus (HIV). Antiretroviral agents are used in combinations called “cocktails” to manage AIDS. Nucleoside reverse transcriptase inhibitors, nonnucleoside reverse transcriptase inhibitors, and protease inhibitors are the groups discussed in this chapter. Table 8-4 lists examples of drugs used to treat HIV. Opportunistic infections often occur in patients with AIDS, so they may be taking various antiinfective agents to prevent diseases such as tuberculosis, Pneumocystis carinii pneumonia, herpes infections, and candidiasis (Table 8-5).
TABLE 8-5
OPPORTUNISTIC INFECTIONS IN PATIENTS WITH HUMAN IMMUNODEFICIENCY VIRUS (HIV) AND DRUGS OF CHOICE
| Organism | Effect | Treatment |
| Cryptococcus neoformans | Meningitis | Amphotericin B |
| Candida | Esophagitis | Clotrimazole, ketoconazole |
| Pneumocystis carinii | Pneumonia | TMP-SMZ, pentamidine |
| CMV | Lungs, pneumonitis | Ganciclovir |
| Mycobacterium tuberculosis | Lungs | INH + rifampin + pyrazinamide |
| Toxoplasma gondii | Encephalitis | Pyrimethamine-sulfadiazine |
CMV, Cytomegalovirus; INH, isoniazid; TMP-SMZ, trimethoprim-sulfamethoxazole.
 NUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITORS
NUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITORS
Zidovudine (zye-DOE-vue-deen) (AZT, Retrovir), a thymidine analog, is converted into zidovudine triphosphate by cellular enzymes. This AZT derivative is then integrated into DNA polymerase (reverse transcriptase) so that synthesis of viral DNA is terminated. The reverse transcriptase of HIV is 100 times more susceptible to inhibition than are normal human cells. The nucleoside antiretroviral agents block viral replication and conversion into a form that can get into an uninfected host cell. It has no effect on cells already containing HIV. AZT is well absorbed orally, metabolized by the liver, and excreted by the kidneys with a half-life of about 1 hour. It is distributed to most body tissue, including CSF. AZT inhibits HIV synthesis and reduces the morbidity and mortality from AIDS and AIDS-related complex. Opportunistic infections are reduced in both number and frequency.
The toxicity of AZT is related to bone marrow depression, which can lead to anemia, granulocytopenia, and thrombocytopenia. Transfusions are often required. CNS effects include headache, agitation, and insomnia. Nausea occurs in almost half the patients. A causal relationship to AZT has not been established, but oral manifestations reported include altered sense of taste, edema of the tongue, bleeding gums, and mouth ulcers. Adverse reactions sometimes limit treatment with AZT.
Acetaminophen, indomethacin, and aspirin can inhibit AZT’s metabolism and potentiate the toxicity of both drugs. Other nonsteroidal antiinflammatory agents have not been implicated, but current literature should be consulted. A higher incidence of granulocytopenia was reported when acetaminophen was used with AZT.
 NONNUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITORS
NONNUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITORS
Nevirapine (ne-VYE-ra-peen) (VP, Viramune), a nonnucleoside reverse transcriptase inhibitor, is specific for HIV-1. HIV-2 is different from HIV-1 in that it is only transmitted from a woman to her child while it is in the womb. This particular form of the virus appears to be centralized in western Africa. HIV-1 is transmitted via sexual intercourse, sharing of bodily fluids, and intravenous substance abuse, and during pregnancy and childbirth. These agents inhibit the same enzymes as the nucleoside analogs, but they do not require bioactivation. Adverse reactions include CNS effects (headache, drowsiness), rash, gastrointestinal effects (diarrhea, nausea), and elevated LFTs. When these agents are used alone, resistance to them develops quickly. This group is combined with the nucleoside analogs and the protease inhibitors.
 PROTEASE INHIBITORS
PROTEASE INHIBITORS
Saquinavir (sa-KWIN-a-veer) (Invirase), a protease inhibitor, prevents the cleavage of viral protein precursors needed to generate functional structural proteins in and modulation of reverse transcriptase activity, preventing the maturation of HIV-infected cells. The difference between the protease inhibitors and the other two groups is that the protease inhibitors can interfere with the action of the HIV-infected cells. Its adverse reactions include rash, hyperglycemia, and paresthesias. Gastrointestinal adverse reactions include pain, diarrhea, and vomiting. Oral adverse reactions involve buccal mucosal ulceration. Ketoconazole significantly increases the levels of saquinavir. Patients taking saquinavir should take it 2 hours after a full meal and avoid sunlight. Although adverse reactions can occur, they are generally less serious than with the older agents. The discovery of the protease inhibitors has made a substantial difference in both the mortality and morbidity of AIDS patients.
 COMBINATIONS
COMBINATIONS
In the management of HIV and AIDS, drugs are combined to produce an improved effect. The combinations of drugs, called “cocktails,” used to manage HIV or AIDS are changing constantly. Rapid changes in retroviral drug therapy make it impossible to predict what specific agents will be used in a few years. Within each group, the agents chosen are often the newest agents, which have quickly been brought to the market. Because the drugs that patients with AIDS will be taking will be the newest agents, it is important to obtain information about these drugs before planning dental treatment. Normally, patients with HIV will be taking three drugs: a nucleoside, a nonnucleoside reverse transcriptase inhibitor, and a protease inhibitor. An example of this combination is lamivudine, nevirapine, and saquinavir.
Other Antiviral Agents
Amantadine (a-MAN-ta-deen) (Symmetrel) inhibits the penetration of the adsorbed virus into the host’s cells or inhibits the uncoating of the influenza A viruses. Its common side effects include nausea, dizziness, lightheadedness, and insomnia. It can be used prophylactically for prevention of an influenza A viral infection or for treatment to reduce the symptoms of the infection. It is used in institutional patients (e.g., nursing homes) to prevent the spread of infection during outbreaks. It also has antiparkinsonian action. A new relative of amantadine with similar action is rimantadine (Flumadine). Resistance to both drugs is widespread, and as a result, the use of both drugs is limited.
 INTERFERONS
INTERFERONS
The interferons (in-ter-FEER-ons) are a large group of endogenous proteins that have antiviral, cytotoxic, and immunomodulating action. Recombinant DNA technology now produces interferons. The FDA has approved several interferons currently classified as alfa* (α), beta (β), and gamma (γ) for certain indications, but they are used for other, unapproved indications. Many more are available that have not been marketed; for example, there are 16 known subtypes of α-interferons. The types of interferons available and their indications are listed in Table 8-6. All currently available interferons are parenteral. The most common uses of the interferons are for the treatment of hepatitis C and multiple sclerosis.
TABLE 8-6
| Interferon Type | Trade Name | Indications, Selected |
| Alfa-2a | Roferon-A | Hairy cell leukemia, chronic hepatitis B or C |
| Peginterferon alfa-2a | Pegasys | Hepatitis B or C |
| With ribavirin | Copegus | |
| Alfa-2b | Intron-A | Hairy cell leukemia, chronic non-A, non-B/C hepatitis AIDS-related Kaposi’s sarcoma, acute hepatitis, chronic hepatitis B |
| Peginterferon alfa-2b | PEG-Intron | Chronic hepatitis C |
| Plus ribavirin | Rebetol | |
| Alfa-n3 | Alferon N | Condyloma acuminatum |
| Beta-1a | Avonex | Multiple sclerosis, relapsing |
| Beta-1b | Betaseron | Multiple sclerosis, relapsing |
Interferons are used parenterally, and injection site reactions, such as necrosis, can occur. The interferons interact with cells through cell surface receptors. Activation of these receptors produces the following effects: induction of gene transcription, inhibition of cellular growth, alteration of the state of cellular differentiation, and interference with oncogene expression. Other effects include altering cell surface antigen expression, increasing phagocytic activity of macrophages, and augmenting the cytotoxicity of lymphocytes. With so many actions, it is no wonder that these agents are being explored for other indications.
Adverse reactions vary, depending on the interferon, but some can be serious and even require discontinuation of the drug. A flulike syndrome, consisting of myalgias, fatigue, headache, and arthralgia, occurs in many patients. Other side effects include CNS effects (fatigue, fever, headache, depression, and chills), gastrointestinal tract effects (nausea, vomiting, and diarrhea), and rash. Oral effects include taste changes, reactivation of herpes labialis, and excessive salivation.
CLINICAL SKILLS ASSESSMENT
1. Name the least toxic antifungal agent useful in the treatment of oral candidiasis. State three dose forms useful in dentistry and describe their pros and cons.
2. State two agents (other than those from question 1) used to treat oral candidiasis. State one problem with each agent. Explain when administration of each is appropriate.
3. Describe the reason for the difficulty associated with the treatment of herpes simplex labialis with antiviral agents. Describe any useful clinically proved effect of either topical or systemic use in dentistry.
4. Explain the significance of a patient taking AZT, ddI, ddC, d4T, NVP, and ritonavir.
5. Describe the serious adverse reactions associated with AZT.
6. Describe how drug combinations are used to manage HIV.
7. List the therapeutic use of two different interferons.
8. Describe the mechanism of action of the different groups of antiviral drugs.
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
*Spanish spelling is used for global consistency.