Antiinfective Agents
1 Outline the history and basic principles of infection and its relevance to dentistry.
2 Summarize the principal indications for the use of antimicrobial agents.
3 Name and describe the major adverse reactions and disadvantages associated with the use of antiinfective agents.
4 Discuss penicillin, macrolides, tetracyclines, cephalosporins—their chemical makeup, properties, mechanisms of action, uses, and potential adverse reactions—and name several specific types.
5 Name and describe several other types of antibiotics and antiinfectives.
6 Discuss the use of antiinfective agents in dentistry.
7 Describe the drugs used to treat tuberculosis and the difficulties this disease presents.
8 Summarize the concept and practice of antibiotic prophylaxis in dentistry.
Antiinfective agents play an important role in dentistry because infection, after pain management, is the dental problem for which drugs are most often prescribed. As the knowledge about the etiology of dental diseases is continually increasing and the involvement of microorganisms becoming better understood, dental health care workers continue to better understand the proper place of antibiotics and their effect on microorganisms. Another important piece of the puzzle of infection is the immunologic response of the host. This puzzle piece has not yet led to therapeutic intervention strategies.
Dental infections can be divided into several types as follows:
• Caries: Caries, produced by Streptococcus mutans, is the first important dental infection of the newly erupting teeth of the young patient. At present, traditional antiinfective agents have not been useful for this problem in the general population. The treatments of choice involve the use of fluoridated water, local physical removal of bacterial plaque from teeth on a regular basis (good oral hygiene, dental prophylaxis), and appropriately placed sealants.
• Periodontal disease: In the adult patient, the dental health care team’s biggest dental problem is periodontal disease. With an increase in knowledge about antiinfective agents, dental teams will be better able to understand and properly administer new treatments for this disease such as tetracycline fibers. Because it is now known that microorganisms, such as Actinobacillus actinomycetemcomitans, black-pigmented bacteroides, motile rods, and spirochetes, are involved in periodontal disease, development of a more rational approach to treatment of periodontal disease may be possible. Table 7-1 lists the common organisms that are involved in periodontal infections and the sensitivity or resistance to the antibiotics tested. Treatments that use localized methods of drug delivery (e.g., tetracycline fibers) hold promise for the future management of periodontal disease.
TABLE 7-1
PERIODONTAL MICROBES, THEIR PRESENCE, AND IN VITRO SUSCEPTIBILITY TO CERTAIN ANTIMICROBIAL AGENTS (BY MINIMUM INHIBITORY CONCENTRATION)
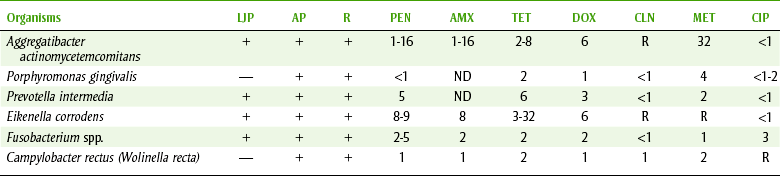
+, Elevated proportions of bacteria; —, regular proportions or not detected or studied; AMX, amoxicillin; AP, adult periodontitis; CIP, ciprofloxacin; CLN, clindamycin; DOX, doxycycline; LJP, localized juvenile periodontitis; MET, metronidazole; MIC, minimal inhibitory concentrations for 90% of strains (µg/m), except penicillin G, which is U/ml; ND, not determined; PEN, penicillin G; R, “refractory” adult periodontitis; TET, tetracycline.
Data from Slots J, Rams TE: Antibiotics in periodontal therapy: advantages and disadvantages, J Clin Periodontol 17:479, 1990.
• Localized dental infections: Most localized dental infections are extensions that arise from either periodontic- or endodontic-related sources. For most localized dental infections, if adequate drainage can be obtained, antiinfective agents are not indicated unless the patient is immunocompromised (Box 7-1). In the occasional situation in which antibiotics are indicated, the antibiotic of choice is determined by the organisms likely to be present.
• Systemic infections: Systemic dental infections can be identified because they produce systemic symptoms such as fever, malaise, and tachycardia. Lesions associated with infections producing these types of symptoms should be drained, but if this is not possible, antibiotics should be given. The duration of therapy should include the number of days for the signs and symptoms to be totally gone plus 2 or 3 days. If the dental infection has systemic symptoms, the use of antiinfective agents is indicated and may even be critical.
DENTAL INFECTION “EVOLUTION”
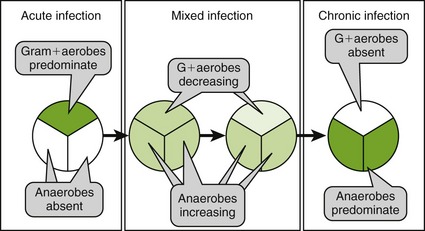
Dental infections often follow similar pathways of evolution from their beginning to their end. In the beginning, the organisms responsible for a dental infection are primarily gram-positive cocci, such as Streptococcus viridans, or α-hemolytic streptococci. After a short time, the gram-positive infection begins to include a variety of both gram-positive and gram-negative anaerobic organisms, such as Peptostreptococcus (Peptococcus) and Bacteroides (Porphyromonas and Prevotella species). At this point, the infection is termed a mixed infection. Over time, the proportion of organisms that are anaerobic increases. With additional time and no treatment, the infection progresses until it consists of predominantly anaerobic flora. At this point, the anaerobic organisms coalesce into an abscess, often visible on radiograph (x-ray).
The choice of antibiotics for a dental patient’s infection depends on where it is in its evolution. If the infection is just beginning, the organisms most likely to be present are gram-positive cocci. Penicillin is the drug of choice, unless the patient has a penicillin allergy. Amoxicillin is most often used because it is less irritating to the stomach and can be taken with food or milk. In patients allergic to penicillin, alternatives might include erythromycin or clindamycin. When the infection is at the mixed stage, agents effective against either gram-positive organisms or anaerobic organisms may be successful. Treating gram-positive organisms is easier, and the drug of choice is penicillin/amoxicillin or with a penicillin allergy, a macrolide antibiotic. For anaerobic organisms, metronidazole is effective. By eradicating one group of organisms, the balance between the two types of organisms is altered and the body can then resolve the infection. Clindamycin affects both gram-positive cocci and gram-positive and -negative anaerobes. Historically, oral surgeons have been comfortable using clindamycin, but other dentists have avoided it because of the association with pseudomembranous colitis (bloody diarrhea).
To treat a dental infection, it is critical to know what organism(s) are likely to be involved and the sensitivity of those organisms to antibiotics. Decisions are based on the likelihood of certain infections and their sensitivities.
HISTORY
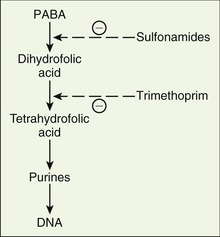
In 1932, Gerhard Domagk of Germany observed that Prontosil protected mice against infection by streptococcal bacteria. This milestone in medical history led to the development of the sulfonamides and marked the beginning of systemic antimicrobial therapy.
In 1940, Chain and Florey of England observed that interest had been focused on the sulfonamides and that other possibilities, notably those connected with naturally occurring substances, should be considered.
In 1928, Fleming of England observed that a mold, Penicillium notatum, produced a substance that inhibited the growth of certain bacteria. He named this substance “penicillin” and suggested that it might be useful for application to infected wounds. In their classic paper, Chain and coworkers reported the low toxicity and systemic antibacterial effectiveness of penicillin. The excitement that began with the sulfonamides was transferred to the penicillins. As each new antibiotic is marketed, this excitement is transferred to the newest antibiotic developed. For years, scientists have been concerned about the indiscriminate use of antibiotics. Recent developments, such as totally resistant strains of bacteria, have made this concern even more important.
DEFINITIONS
A discussion of individual antimicrobial agents is preceded by definitions of the following terms:
• Antiinfective agents: Substances that act against or destroy infections.
• Antibacterial agents: Substances that destroy or suppress the growth or multiplication of bacteria.
• Antibiotic agents: Chemical substances produced by microorganisms that have the capacity, in dilute solutions, to destroy or suppress the growth or multiplication of organisms or prevent their action. The difference among the terms antibiotic, antiinfective, and antibacterial is that antibiotics are produced by microorganisms, whereas the other agents may be developed in a chemistry laboratory (not from a living organism). Antibacterial refers to a substance from any source that inhibits or kills bacteria. The term antiinfective refers to a substance from any source that inhibits or kills organisms that can produce infection, such as bacteria, protozoa, viruses, and so forth. This difference is largely ignored in general conversation, and antiinfectives are often referred to as “antibiotics.”
• Antimicrobial agents: Substances that destroy or suppress the growth or multiplication of microorganisms.
• Antifungal agents: Substances that destroy or suppress the growth or multiplication of fungi.
• Antiviral agents: Substances that destroy or suppress the growth or multiplication of viruses.
The following are definitions of commonly used terms:
• Bactericidal: The ability to kill bacteria. This effect is irreversible, that is, if the bacteria are removed from the drug, they do not live.
• Bacteriostatic: The ability to inhibit or retard the multiplication or growth of bacteria. This is a reversible process because if the bacteria are removed from the agent, they are able to grow and multiply. Whether an antibacterial agent is labeled bactericidal or bacteriostatic depends on variables such as the dose used or the organism being treated. Box 7-2 lists the most common antimicrobial agents and classifies them as bacteriostatic or bactericidal.
• Blood (serum) level: Concentration of the antiinfective agent present in the blood or serum. The importance of the serum level is that certain levels of an antibiotic are required to produce an effect on various types of organisms. For an antibiotic to be effective, the dose given must produce this concentration in the blood.
• Infection: Infection is not only an invasion of the body by pathogenic microorganisms but also a reaction of the tissues to their presence. The presence of a pathogen does not constitute “invasion.” In fact, many potential oral pathogens are part of the normal floral in the mouth, but they only cause infection if their relative numbers rise.
• Minimum inhibitory concentration (MIC): Lowest concentration needed to inhibit visible growth of an organism on media after 18 to 24 hours of incubation. This in vitro test is more reliable and quantitative than the disk tests.
• Spectrum: Range of activity of a drug. The spectrum of activity of an antibacterial agent may be narrow, intermediate, or broad. A narrow-spectrum agent acts primarily against a smaller group of bacteria such as gram-positive cocci, gram-negative rods, gram-positive or gram-negative anaerobes, or viruses.
• Superinfection, suprainfection: Infection caused by the proliferation of microorganisms different from those causing the original infection. When antiinfectives disturb the normal flora of the body, the emergence of organisms unaffected by or resistant to the antibiotic used can occur. Superinfection is more often caused by broad-spectrum antibiotics such as tetracycline and increases when taken for a longer period. In this case, a reduction in the number of gram-positive and gram-negative bacteria allows the overgrowth of the fungus, Candida albicans. The pathogenic organisms emerging in a superinfection generally are more difficult to eradicate than the original organism and are more likely to exhibit resistance. The fact that the practitioner can cause and eliminate infections emphasizes the importance of determining a definite need before these drugs are used. (Dental practitioners should stamp out, or at least reduce, health care–acquired infections.)
• Synergism: Synergism occurs when the combination of two antibiotics produces more effect than would be expected if their individual effects were added (in other words, 1 + 1 > 2). Combinations of antibiotics that are bactericidal are generally synergistic. Combinations of those that are bacteriostatic are merely additive (1 + 1 = 2).
• Antagonism: Antagonism occurs when a combination of two agents produces less effect than either agent alone (see Box 7-2).
INFECTION
The factors that determine the likelihood of a microorganism causing an infection are the following:
• Disease-producing power of the microorganism (virulence)
• Number of organisms present (inoculum)
• Resistance of the host (immunologic response): Host resistance should be considered as having both local and systemic components. Systemically, both drugs (steroids and antineoplastic agents) and diseases (acquired immunodeficiency syndrome [AIDS] and insulin-dependent diabetes mellitus [IDDM]) may reduce a patient’s immunity (see Box 7-1) and increase the chance of an infection. Sleep deprivation and anxiety can also reduce a patient’s immunologic response to infection.
CULTURE AND SENSITIVITY
Ideally, all infections requiring antimicrobial therapy would be cultured, and sensitivity tests would be performed. Culturing involves growing the bacteria from a sample of infective exudate, and sensitivity testing involves exposing the organism to certain test antibiotics and determining whether the organism is sensitive or resistant. Today, in part at least because of the inappropriate use of antiinfective agents, organisms are more quickly becoming resistant to antibiotics.
Culture and sensitivity is the only way to be sure that a drug will kill or inhibit the growth of the infecting microorganisms in a patient-specific infection. In practice, this is often difficult. In dentistry, the need for anaerobic culturing makes obtaining a sample and culturing it more difficult. Another problem is that many dental infections are often of mixed origin so that the results of the cultures are difficult to interpret. In cases of a serious infection, an infection in a compromised patient, or an infection that is not responding to treatment, it is imperative that a culture be taken.
Culture
When a culture is taken, proper collection materials (tubes or vials with the correct media) and methods must be used to obtain reliable results. Dental professionals need to communicate to the laboratory personnel the nature of the appropriate cultures to be taken. The laboratory personnel should perform a Gram stain so that they may report all of the bacteria that are present in high numbers. Both obligate and facultative anaerobes should be preserved.
Depending on the site, the collection method varies. Examples of methods include the following: for an abscess, aspiration with a needle; for a draining lesion, a swab from an anaerobic pack; and for endodontic treatment, properly handled absorbent points. Collection methods should be adapted to keep the anaerobes alive. For the periodontal pocket, the sterile paper point or the explorer can be used to sample the pocket.
Care must be taken not to contaminate the sample with supragingival plaque, which has a different microbial constitution. In at-risk patients, a culture should be taken before antibiotics are administered because antibiotics can alter the nature of the microbes so that identification is more difficult. Infections in problem sites and in problem patients should be cultured.
Sensitivity
After the organism is identified, it is grown on culture medium (Figure 7-1). Observing whether the organisms are sensitive or resistant to certain test antibiotics assists in determining which antibiotic to use in difficult infections. One to 2 days are required before the results of this test are available. Although therapy can start before this time, it may be changed after the results are available. If clinical response has been adequate, often the original antibiotic is continued despite sensitivity results.
RESISTANCE
Resistance (related to antibiotics) is the natural or acquired ability of an organism to be immune to or to resist the effects of an antiinfective agent. Natural resistance occurs when an organism has always been resistant to an antimicrobial agent because of the bacteria’s normal properties such as lipid structures in the cell wall. Acquired resistance occurs when an organism that was previously sensitive to an antimicrobial agent develops resistance. This can occur by natural selection of a spontaneous mutation (“survival of the fittest”). An increase in the use of an antibiotic in a given population (e.g., a hospital) increases the proportion of resistant organisms in that population. Conversely, a decrease in the use of an antibiotic decreases the proportion of organisms resistant to that antibiotic in that given population. Another method by which resistance develops is by the transfer of deoxyribonucleic acid (DNA) genetic material from one organism to another via transduction, transformation, or bacterial conjugation. The first organism, which is resistant to one or more antibiotics, transfers its genetic material to a second organism. The second organism, which was not previously resistant, thus becomes resistant to the same antibiotic as the first organism without ever having been exposed to that antibiotic. This transfer of genetic material from one organism to another may occur among very different microorganisms, including transfer from a nonpathogenic bacteria to a pathogenic bacteria. The three most common mechanisms of acquired resistance are a decrease in bacterial permeability, the production of bacterial enzymes, and an alteration in the target site.
INDICATIONS FOR ANTIMICROBIAL AGENTS
Considerable controversy exists regarding the need for antimicrobial agents in various situations. The two categories of indications are prophylactic and therapeutic.
Therapeutic Indications
Although there is no simple rule to determine whether antimicrobial therapy is needed in dentistry, many infections do not require it. Most patients without immune function deficiencies, in whom drainage can be obtained, need no antibiotics to manage their dental infections. (Patients with acne are not prescribed an antibiotic every time a pimple is pinched.) Table 7-2 lists the indications for treatment of dental infections and the antibiotics of choice and their alternatives. If local resistance patterns vary from those found in the table, antibiotic choice should be based on that information. However, before a decision is made, there are several factors that must be considered.
TABLE 7-2
ANTIMICROBIAL USE IN DENTISTRY*
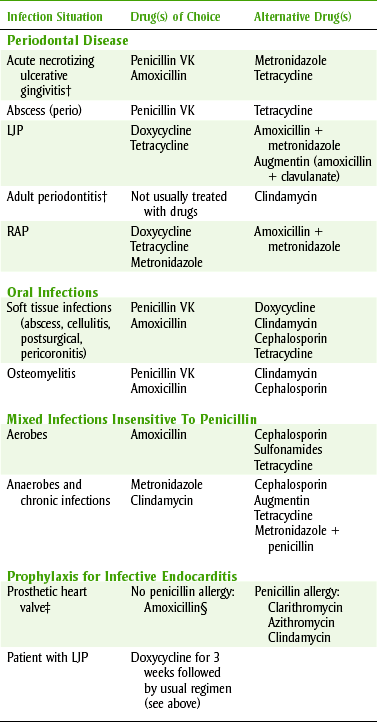
LJP, Localized juvenile periodontitis; RAP, rapid advancing periodontitis.
*Clinical conditions may alter drug therapy.
†No antimicrobial agents are usually required for these conditions.
 PATIENT
PATIENT
The best defense against a pathogen is the host response. A properly functioning defense mechanism is of primary importance. When this defense is lacking, the need for antimicrobial agents is more pressing.
 INFECTION
INFECTION
The virulence and invasiveness of the microorganism are important in deciding the acuteness, severity, and spreading tendency of an infection. An acute, severe, rapidly spreading infection should generally be treated with antimicrobial agents, whereas a mild, localized infection in which drainage can be established need not be treated. If the periodontal pocket (site) remains active despite repeated root planing, then the use of antibiotics to alter the flora may be considered.
When antimicrobial agents are to be used in the treatment of dental infections, the organisms likely to produce the infection and their susceptibility to antimicrobial agents must be considered. Table 7-2 lists the antimicrobials of choice for various dental situations (when culture and sensitivity tests are unavailable) and alternatives if the drug of choice cannot be used. When two antimicrobial agents have approximately the same therapeutic effect and their cost to the patient is very different, the cost of therapy is another consideration.
To answer the question as to whether an antibiotic is effective in a certain dental infection, one needs many patients with similar infections in which half are given active antibiotics and half are given a placebo. This study would help to define the proper use of antibiotics in dental infections. This slow testing has begun, using antibiotics in periodontal situations, but there is still much to discover.
Prophylactic Indications
Few situations arise for which a definite indication for prophylactic antibiotic coverage exists. One clear-cut use of antibiotics for prophylaxis before a dental procedure (recommended by the American Heart Association and the American Dental Association) is a history of infective endocarditis, presence of a heart valve prosthesis, or congenital heart disease. The most current guidelines regarding antibiotic prophylaxis are discussed in detail at the end of this chapter.
GENERAL ADVERSE REACTIONS AND DISADVANTAGES ASSOCIATED WITH ANTIINFECTIVE AGENTS
Superinfection (Suprainfection)
All antiinfective agents can produce an overgrowth of an organism that is different from the original infecting organism and resistant to the agent being used. The wider the spectrum of the antiinfective agent and the longer the agent is administered, the greater the chance of superinfection occurring. This side effect can be minimized by use of the most specific antiinfective agent, the shortest effective course of therapy, and adequate doses.
Allergic Reactions
All antiinfective agents, just like all drugs, have the potential to produce a variety of allergic reactions, ranging from a mild rash to fatal anaphylaxis. Some antiinfective agents, such as the penicillins and the cephalosporins, are more allergenic than other agents. Many antiinfective agents, such as erythromycin and clindamycin, have a low allergenic potential.
Drug Interactions
Antiinfective agents can interact with oral contraceptives, oral anticoagulants, and other antiinfectives (a bacteriostatic agent interferes with a bactericidal agent).
 ORAL CONTRACEPTIVES
ORAL CONTRACEPTIVES
Some antibiotics have been found to decrease oral contraceptive efficacy by increasing their clearance from the body. This drug interaction, although unlikely, should be discussed with the patient whenever a patient using oral contraceptives receives a prescription for an antibiotic. Of those antibiotics used in dentistry, ampicillin and the tetracyclines are the most likely to produce this effect. In certain patients, additional birth control measures should be used during antibiotic administration.
 ORAL ANTICOAGULANTS
ORAL ANTICOAGULANTS
Antiinfective agents can potentiate the effect of oral anticoagulants. Oral anticoagulants are vitamin K inhibitors, so interfering with the production of vitamin K could increase the anticoagulant effect. Bacterial flora in the intestine produce most of the vitamin K in human bodies. Antiinfective agents (e.g., tetracycline) reduce the bacterial flora that produce vitamin K. With the vitamin K reduced, the oral anticoagulant’s effect is increased. Erythromycin inhibits the enzymes that metabolize warfarin, leading to an increase in warfarin levels. Prolongation of the international normalized ratio (INR) leading to bleeding or hemorrhage may result. INR should be monitored more closely in patients on antiinfective therapy. Antiinfective agents interact to varying degrees depending on the antibiotic.
 OTHER ANTIINFECTIVES
OTHER ANTIINFECTIVES
Antibiotics that act at the same receptor may compete for that receptor and should not be given together (e.g., erythromycin and clindamycin). An antibiotic that has bacteriostatic properties stops the bacteria from growing, thereby inhibiting the action of a bactericidal agent (requires growing and actively dividing cells to work). Except in a few unusual, nondental cases, one antibiotic should be chosen and used alone.
Gastrointestinal Complaints
All antiinfective drugs can produce a variety of gastrointestinal complaints. The complaints include stomach pain, increased motility, and diarrhea. The incidence varies greatly, depending on the particular agent used, the dose of that agent, and whether the patient takes the drug with food. Erythromycin has the highest incidence of gastrointestinal complaints of any of the antibiotics. More serious gastrointestinal complaints, such as pseudomembranous colitis, which has been historically linked with clindamycin, are now known to occur not only with a wide variety of antiinfective agents (cephalosporins, amoxicillin) but also in the absence of any antimicrobial agents.
Pregnancy Considerations
The antimicrobial agents that can be used during pregnancy to treat infections are limited. Although the risk-to-benefit ratio must be considered whenever pregnant women are given any medications, penicillin and erythromycin have not been associated with teratogenicity and are often used. The use of clindamycin is probably also acceptable, but before any antibiotics are used in the pregnant dental patient, the patient’s obstetrician should be contacted (this procedure also helps prevent medical-legal problems). Metronidazole is not usually used during pregnancy, but exceptions exist. The tetracyclines are contraindicated during pregnancy because of their effect on developing teeth and skeleton.
Dose Forms
Adult dose forms of antibiotics are commonly tablets and capsules. Children’s dose forms, including liquid and chewable antibiotic dose forms, contain sugar as their sweetening agent. After the dentition has erupted, the dental health care worker should encourage the parent or child to brush the child’s teeth after the use of these agents. The chewable tablets can stick to the teeth, especially in the pits. Long-term administration of antibiotics could increase the child’s caries rate.
Cost
Cost is an important factor in choosing an antibiotic for a patient. If the perfect antibiotic is chosen and prescribed but the patient does not purchase the medication because it is too expensive, then poor results are likely. The best inexpensive antibiotic that can be taken will be more effective than an expensive one that cannot be purchased. Figure 7-2 compares the cost of various antiinfective agents.
PENICILLINS
The penicillins (pen-i-SILL-ins) can be divided into four major groups (see Table 7-3). The first group contains penicillin G and V, the second group is composed of the penicillinase-resistant penicillins, the third group contains amoxicillin, and the fourth group consists of extended-spectrum penicillins. Because the penicillins have many properties in common, their similarities are discussed first. In dentistry, the first and third groups are commonly used.
Source and Chemistry
The mold Penicillium notatum and related species produce the naturally occurring penicillins. The semisynthetic penicillins are produced by chemically altering the naturally produced penicillins. The penicillin structure includes a β-lactam ring fused to a five-member, S-containing thiazolidine ring. Neither of these rings has much antimicrobial action alone. Active penicillins are produced by adding different functional groups to position 6. Cleaving these functional groups from their two-ring structure results in a loss of antimicrobial activity. When the β-lactam ring is broken, such as in the presence of penicillinase, the antimicrobial activity of the compound is lost (Figure 7-3).
The addition of organic groups at the R position confers antibacterial activity to the compounds formed from 6-aminopenicillanic acid. These R groups create the various penicillins that were originally designated with letters, for example, penicillin G and penicillin V. The penicillins can be inactivated by any reaction that removes the R group or in the case of penicillinase, breaks the β-lactam ring (B). Salts of the penicillins are made by reactions at the thiazolidine (T) carboxyl ( COOH) group.
COOH) group.
Although many naturally occurring penicillins have been produced, only penicillin G (Na+/K+ penicillin) is of use today. The various semisynthetic penicillins are formed by substituting other groups at the R position.
Pharmacokinetics
Penicillin can be administered either orally or parenterally but should not be applied topically because its allergenicity is greatest by that route. When penicillin is administered orally, the amount absorbed depends on the type of penicillin. The percentage can vary from 0% to more than 90% (see Table 7-3). When the percentage absorbed is too low, as with methicillin, the penicillin is available only for injection. Penicillin V is better absorbed orally than penicillin G, so penicillin V is used for administration of oral penicillin.
The oral route provides the advantages of convenience and less likelihood of a life-threatening allergic reaction. The disadvantages of using the oral rather than the parenteral route are that the blood levels rise slower, the blood levels are less predictable because of variable absorption or lack of patient compliance (biggest problem), and some penicillins are degraded by gastric acid. The highest blood levels are obtained if the patient takes the penicillin orally at least 1 hour before or 2 hours after meals, but penicillin V and amoxicillin can be taken without regard to meals.
After absorption, penicillin is distributed throughout the body, with the exception of cerebrospinal fluid, bone, and abscesses. This includes the tissue, saliva, and kidneys. Penicillin crosses the placenta and appears in breast milk.
Penicillin is metabolized by hydrolysis in the liver and undergoes tubular secretion in the kidney. The elimination half-life for both penicillin G and penicillin V is about 0.5 hour. In five half-lives, about 2.5 hours, these penicillins are virtually eliminated from the body.
Mechanism of Action
Penicillin is a very potent bactericidal agent that attaches to penicillin-binding proteins (PBPs) on the bacterial cell membrane. The PBPs are enzymes that are involved in the synthesis of the cell wall and the maintenance of the cell’s structural integrity. Penicillin acts as the structural analog of acyl-d-alanyl-d-alanine, inhibiting the formation of cross-linkages (transpeptidases). This destroys cell wall integrity and leads to lysis. The penicillins are more effective against rapidly growing organisms. Table 7-3 summarizes the types, routes of ingestion, and other properties of the types of penicillins.
Spectrum
Penicillin G and V’s narrow spectrum of activity includes gram-positive cocci, such as Staphylococcus aureus, Staphylococcus pneumoniae, Streptococcus pyogenes, Streptococcus viridans, and certain gram-negative cocci, such as Neisseria gonorrhoeae (produces gonorrhea) and N. meningitidis. Penicillin is also effective against spirochetes and anaerobes such as Actinomyces, Peptococcus, Peptostreptococcus, Bacteroides, Corynebacterium, and Clostridium species. The spectrum of action of the penicillins matches the microbes responsible for many periodontal conditions. The other penicillins have a somewhat different spectrum that is discussed in each section.
The antibacterial activity of penicillin is standardized in international units (IU). One international unit has the activity of 0.6 mg of the master standard of sodium penicillin G, so 1 mg of pure sodium penicillin G equals 1667 IU. About 400,000 IU of penicillin V is equivalent to 250 mg. Penicillin G is usually measured in international units, whereas other penicillins are expressed in milligrams.
Resistance
Resistance to penicillin can occur by several different mechanisms. Penicillinase-producing staphylococci are resistant because their enzymes destroy some penicillins. These penicillinases inactivate the penicillin moiety by cleaving the β-lactam ring.
In hospital environments, more than 95% of the population of staphylococci are penicillinase-producing organisms. Clavulanic acid serves as an inhibitor, which allows the use of amoxicillin to treat penicillinase-producing organisms. Certain bacteria have an outer cell membrane that prevents penicillin from reaching the PBPs.
Although most oral strains of S. viridans are sensitive to penicillin, an increasing number of strains are becoming resistant. The amount of bacterial resistance is proportional to the clinical use of the antibiotic; frequent use leads to increased resistance (and vice versa).
Adverse Reactions
The untoward reactions to the penicillins can be divided into toxic reactions and allergic or hypersensitivity reactions. The penicillins are the most common cause of drug allergies.
 TOXICITY
TOXICITY
Because penicillin’s toxicity is almost nonexistent, large doses have been tolerated without adverse effects. For this reason, there is a large margin of safety when penicillin is administered. With massive intravenous (IV) doses, direct central nervous system (CNS) irritation can result in convulsions. Large doses of penicillin G have been associated with renal damage manifested as fever, eosinophilia, rashes, albuminuria, and a rise in blood urea nitrogen (BUN). Hemolytic anemia and bone marrow depression have also been produced by penicillin. The penicillinase-resistant penicillins are significantly more toxic than penicillin G. Gastrointestinal irritation can manifest itself as nausea with or without vomiting. The irritation caused by injection of penicillin can produce sterile abscesses if given intramuscularly or thrombophlebitis if given intravenously.
 ALLERGY AND HYPERSENSITIVITY
ALLERGY AND HYPERSENSITIVITY
Allergic reactions to penicillin always should be considered when penicillin is prescribed. Some studies indicate that 5% to 10% of patients receiving penicillin will have a reaction. Allergic reactions to oral penicillin are less common than with parenteral penicillin. Anaphylactic reactions are more frequent in patients pretreated with β-blockers and subsequently given oral penicillin. Anaphylactic reactions in these patients have been reported to be difficult to treat.
The following are types of allergic reactions associated with the penicillins:
• Anaphylactic reactions: Anaphylactic shock, an acute allergic reaction, occurs within minutes after the administration of penicillin and presents the most serious danger to patients. It is characterized by smooth muscle contraction (e.g., bronchoconstriction), capillary dilation (shock), and urticaria caused by the release of histamine and bradykinin. If treatment does not begin immediately, death can result. The treatment of anaphylaxis is the immediate administration of parenteral epinephrine.
• Rash: All types of skin rashes have been reported in association with the administration of penicillin. This type of reaction accounts for 80% to 90% of allergic reactions to the penicillins. These rashes are usually mild and self-limiting but can occasionally be severe. Even contact dermatitis has occurred as a result of topical exposure, for example, while preparing an injectable solution (type IV).
• Delayed serum sickness: Serum sickness is manifested as fever, skin rash, and eosinophilia or severely as arthritis, purpura, lymphadenopathy, splenomegaly, mental changes, an abnormal electrocardiogram, and edema. It usually takes at least 6 days to develop and can occur during treatment or up to 2 weeks after treatment has ceased.
• Oral lesions: Delayed reactions to penicillin can exhibit themselves in the oral cavity. These include severe stomatitis, furred tongue, black tongue, acute glossitis, and cheilosis. These oral lesions can occur most commonly with topical application but have been reported from other routes.
• Other reactions: Interstitial nephritis, hemolytic anemia, and eosinophilia are types of allergic reactions occasionally reported during penicillin therapy.
When reactions to penicillin occur, the consequences are often serious. It is estimated that an anaphylactic reaction occurs in up to 0.05% of penicillin-treated patients, with a mortality of 5% to 10%. It is estimated that 100 to 300 deaths occur annually in the United States because of an allergic reaction to penicillin. Although the chance of a serious allergic reaction to penicillin is greater after parenteral administration, anaphylactic shock and death after oral use have also been reported. Patients who have a history of any allergy are more likely to be allergic to penicillin.
Allergic reactions to penicillin of any type may be followed by more serious allergic reactions on subsequent exposure. Any history of an allergic reaction to penicillin contraindicates its use, and another antibiotic should be substituted. However, a negative history does not guarantee the lack of a penicillin allergy. If a penicillin is prescribed and any question of a reaction remains, one should make sure that, after the first dose is taken, the patient is somewhere where help can be summoned if necessary.
Uses
Penicillin is an important antibiotic in medical and dental practice. Its use in dentistry results from its bactericidal potency, lack of toxicity, and spectrum of action, which includes many oral flora. It is often used for the treatment of dental infections. Table 7-2 demonstrates the dental infections for which penicillin is the drug of choice if patients are not allergic to it. Amoxicillin, a close penicillin relative, is also used for specific prophylactic indications. It is the agent of choice for the prophylaxis of infective endocarditis in nonallergic patients who have a history of rheumatic heart disease or valve damage (see the discussion on antibiotic prophylaxis of infective endocarditis at the end of this chapter). Penicillin’s effectiveness in the treatment of dental infections is explained by its effectiveness against many aerobic and anaerobic bacteria.
Specific Penicillins
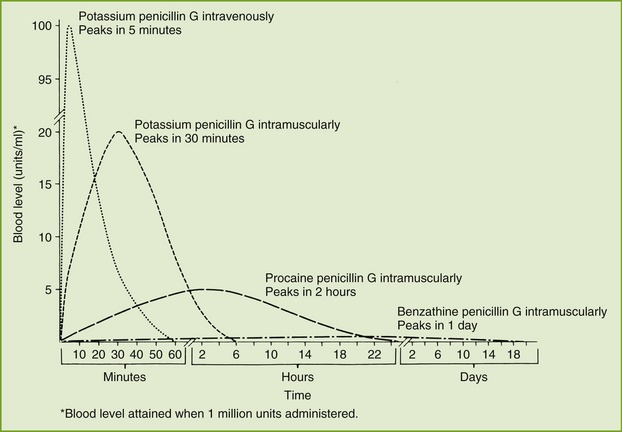
Penicillin G, the prototype penicillin, is available as sodium, potassium, procaine, or benzathine salts. These salts differ in their onset and duration of action and the plasma levels attained. Figure 7-4 compares the blood levels attained by the IV administration of the potassium salt and the intramuscular (IM) administration of the potassium, procaine, and benzathine salts. One should note that the potassium salt given intravenously produces the most rapid and highest blood level, whereas the benzathine salt given intramuscularly produces the lowest and most sustained blood level. The potassium and procaine salts, given intramuscularly, produce intermediate blood levels and durations of action. The penicillin’s duration of action is inversely proportional to the solubility of the penicillin form: the least soluble is the longest acting.
The sodium salts of penicillin should be avoided in patients with a limited sodium intake such as cardiovascular patients. Renal patients should not be given potassium salts, which can result in hyperkalemia. Patients may be allergic to the procaine moiety in procaine penicillin G. Both procaine and benzathine penicillins are suspensions given intramuscularly, from which the penicillin is slowly released.
 PENICILLIN V
PENICILLIN V
Penicillin V has a spectrum of action very similar to that of penicillin G. The potassium salt of penicillin V (K penicillin V or penicillin VK) is more soluble than the free acid and therefore is better absorbed when taken orally. Table 7-2 lists some situations in which penicillin is the drug of first choice if the patient is not allergic to it. The usual adult dose is 500 mg four times a day (qid) for treatment of an infection for a minimum of 5 days and preferably 7 to 10 days.
 PENICILLINASE-RESISTANT PENICILLINS
PENICILLINASE-RESISTANT PENICILLINS
Penicillinase-resistant penicillins should be reserved for use against only penicillinase-producing staphylococci. Compared with penicillin G, the penicillinase-resistant penicillins are less effective against penicillin G–sensitive organisms. They also produce more side effects such as gastrointestinal discomfort, bone marrow depression, and abnormal renal and hepatic function. Patients allergic to penicillin are also allergic to the penicillinase-resistant penicillins.
Because cloxacillin and dicloxacillin are better absorbed than the other penicillinase-resistant penicillins, they are the drugs of choice.
 AMPICILLINS
AMPICILLINS
Ampicillin (am-pi-SILL-in) and amoxicillin (a-mox-i-SILL-in) are most often used in medicine. These penicillinase-susceptible penicillins have a spectrum of action that includes gram-positive cocci, Haemophilus influenzae, and enterococci such as Escherichia coli, Proteus mirabilis, and Salmonella and Shigella species.
Amoxicillin, a relative of ampicillin, is most often used to treat infections because it produces higher blood levels, is better absorbed, requires less frequent dosing (three times daily versus four times daily for penicillin VK or ampicillin), and its absorption is not impaired by food. Amoxicillin is the drug of choice for prophylaxis for bacterial endocarditis before a dental procedure. Amoxicillin is used to treat upper respiratory tract infections (H. influenzae), urinary tract infection (E. coli), and meningitis (H. influenzae). Otitis media in children is often treated with amoxicillin. Amoxicillin is also available mixed with clavulanic acid, a β-lactamase inhibitor (Augmentin). Clavulanic acid combines with and inhibits the β-lactamases produced by bacteria. Therefore the amoxicillin is protected from enzymatic inactivation. This combination can be used with penicillin-producing organisms. It has had some use in the management of certain periodontal conditions (see Table 7-2).
Both ampicillin and amoxicillin can produce a variety of allergic reactions. Ampicillin is much more likely to produce rashes than other penicillins. Most agree that the ampicillin rash is not of an allergic or immunologic nature. This unusual ampicillin-related rash is much more common in patients with mononucleosis (almost 100%) or those taking allopurinol. Cross-allergenicity between penicillin VK, amoxicillin, and ampicillin is complete (omitting the “weird” ampicillin rash).
MACROLIDES
The macrolide antibiotics consist of erythromycin, clarithromycin, and azithromycin (Table 7-4).
TABLE 7-4
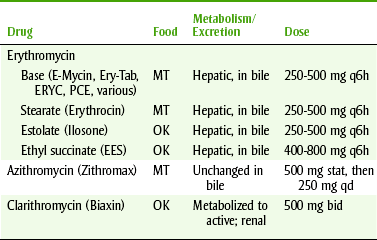
bid, Twice a day; MT, take on an empty stomach (1 hour before or 2 hours after eating); OK, may take without regard to meals; qd, daily.
Erythromycin
Erythromycin is usually bacteriostatic and interferes with protein synthesis by inhibiting the enzyme peptidyl transferase at the 50S ribosomal subunit. Its spectrum of action closely resembles that of penicillin against gram-positive bacteria. It is also the drug of choice for Bordetella, Legionella, and Actinomyces organisms, Mycoplasma pneumoniae, Entamoeba histolytica, some Chlamydia species, and diphtheria. It is also indicated for streptococcal and staphylococcal infections.
 PHARMACOKINETICS
PHARMACOKINETICS
Erythromycin is administered orally as tablets and capsules, oral suspensions in IV and IM forms, and in topical preparations. Because erythromycin is broken down in the gastric fluid, it is formulated as an enteric-coated tablet, capsule, or insoluble ester to reduce degradation by stomach acid. It should be administered 2 hours before or 2 hours after meals (see Table 7-4). The peak blood level varies between 1 and 6 hours. Although food reduces the absorption of erythromycin, it may be necessary to administer it with food to minimize its adverse gastrointestinal effects. Its half-life is 2 hours.
 ADVERSE REACTIONS
ADVERSE REACTIONS
With usual therapeutic doses of erythromycin, side effects other than gastrointestinal are usually minimal. Allergic reactions to erythromycin are uncommon.
Gastrointestinal Effects: The side effects most often associated with erythromycin administration are gastrointestinal and include stomatitis, abdominal cramps, nausea, vomiting, and diarrhea. These effects occur more often in four times daily versus twice daily dosing and with higher (2 gm/day) versus lower (1 gm/day) doses. In one study, at least one gastrointestinal side effect occurred in an average of about 50% of patients, with about 20% discontinuing their medication because of side effects.
Cholestatic Jaundice: Cholestatic jaundice has been reported primarily with the estolate form but has also been reported with the ethylsuccinate form. Erythromycin base has not been associated with this reaction. Symptoms include nausea, vomiting, and abdominal cramps followed by jaundice and elevated liver enzyme levels. Patients with a history of hepatitis should be given erythromycin base or stearate. The mechanism of this adverse effect is believed to be a hypersensitivity reaction.
 DRUG INTERACTIONS
DRUG INTERACTIONS
Erythromycin can increase the serum concentrations of theophylline, digoxin, triazolam, warfarin, carbamazepine, and cyclosporine. This effect may produce toxicity, depending on the doses of each drug. The mechanism by which erythromycin produces these drug interactions may involve inhibition of hepatic metabolism of these drugs. Table 7-5 lists some drug interactions of the macrolides.
TABLE 7-5
ERYTHROMYCIN DRUG INTERACTIONS
| Drug Interacting | Mechanism | Management |
| Antibiotics (clindamycin, penicillin) | Interferes with action of other antibiotics | Choose one antibiotic for both purposes; stop one while administering the other |
| Carbamazepine (Tegretol) | Increased serum levels of carbamazepine | Monitor |
| OC, BCP | Decreased effectiveness of OC | Use alternative method of birth control (e.g., condoms) until end of that cycle (rest of the month) |
| Warfarin (Coumadin) | Increased warfarin effect | Bleeding increased |
| Theophylline (Theo-Dur, Slo-bid) | Increased theophylline toxicity | OK to give 2 doses |
 USES
USES
Because erythromycin is active against essentially the same aerobic microorganisms as penicillin, it is the drug of first choice against these infections in penicillin-allergic patients. Erythromycin is not effective against the anaerobic Bacteroides species implicated in many dental infections.
Azithromycin and Clarithromycin
Both azithromycin (ay-ZITH-roe-my-sin) (Zithromax, Z-Pak) and clarithromycin (klare-ITH-roe-my-sin) (Biaxin) are newer macrolide antibiotics like erythromycin. They inhibit RNA-dependent protein synthesis by binding to the 50S ribosomal subunit They have activity against aerobic gram-positive cocci, such as Staphylococcus and Streptococcus organisms, and gram-negative aerobes. In contrast to erythromycin, azithromycin and clarithromycin have variable action against some anaerobes. They are bacteriostatic and can be taken without regard to meals.
The incidence of adverse reactions is lower with azithromycin and clarithromycin as compared to erythromycin. Adverse reactions relate to the gastrointestinal tract, including dyspepsia, diarrhea, nausea, and abdominal pain. Azithromycin has been reported to elevate liver function tests (LFTs) and should be used with caution in patients with hepatic impairment. Clarithromycin can produce an abnormal or metallic taste.
Several drug interactions can occur with both agents because of their reduction in the metabolism of certain drugs metabolized in the liver. Azithromycin can increase the levels of astemizole, loratadine, carbamazepine, digoxin, and triazolam but does not affect either warfarin or theophylline. The peak of azithromycin is reduced by cations, such as magnesium and aluminum, but the total drug absorbed is not affected. Clarithromycin increases the levels of drugs metabolized in liver such as theophylline, carbamazepine, digoxin, omeprazole, and astemizole. Like the other macrolides, clarithromycin inhibits the cytochrome P (CYP)-450 liver microsomal enzymes.
Azithromycin and clarithromycin are indicated as alternative antibiotics in the treatment of common orofacial infections caused by aerobic gram-positive cocci and susceptible anaerobes. The dose for azithromycin consists of 5 days of therapy: first day, 250 mg twice a day (bid), and then 250 mg/day for 4 more days; for clarithromycin, the dose is 500 mg bid for 7 to 10 days. When amoxicillin and clindamycin cannot be used for the prophylaxis of endocarditis and prosthetic joint infections, these macrolides can be used as alternative antibiotics. The dose for prevention of bacterial endocarditis or joint prosthesis is 500 mg 1 hour before the dental procedure.
TETRACYCLINES
The tetracyclines (te-tra-SYE-kleens) are broad-spectrum antibiotics affecting a wide range of microorganisms (Table 7-6). Their adverse effects on developing teeth are well known.
TABLE 7-6
ORAL AND TOPICAL TETRACYCLINES
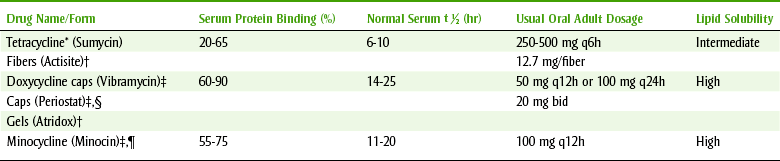
*Avoid concomitant administration with food or divalent or trivalent cations.
†Used topically in sulcus.
‡May be taken with food or milk but not high concentration of divalent or trivalent cations.
§Systemic very low doses; effect because of collagenase, not antibacterial action.
|Vestibular side effects, blue oral lesions.
The first tetracycline was isolated from a Streptomyces strain in 1948. Since then, other tetracyclines have been derived from different species of Streptomyces, and the rest have been produced semisynthetically. The tetracyclines are closely related chemically and clinically.
Pharmacokinetics
The tetracyclines are most commonly given by mouth (PO). Absorption after oral administration varies but is fairly rapid. There is wide tissue distribution, and tetracyclines are secreted in the saliva and in the milk of lactating mothers (one-half plasma concentration). Tetracyclines are concentrated by the liver and excreted into the intestines via the bile. Enterohepatic circulation prolongs the action of the tetracyclines after they have been discontinued. The tetracyclines are also stored in the dentine and enamel of unerupted teeth and are concentrated in the gingival crevicular fluid. The long-acting agents are concentrated to at least four times serum levels.
The various tetracyclines differ clinically in their duration of action, percent absorbed when taken orally, half-lives, and mechanism of elimination. Doxycycline is excreted in the feces, whereas tetracycline is eliminated essentially unchanged by glomerular filtration and minocycline is metabolized in the liver and excreted in the urine. Both doxycycline and minocycline may be given safely to patients with renal dysfunction. All tetracyclines cross the placenta and enter the fetal circulation.
Spectrum
The tetracyclines are bacteriostatic and interfere with the synthesis of bacterial protein by binding at the 30S subunit of bacterial ribosomes. As broad-spectrum antibiotics, they are effective against a wide variety of gram-positive and gram-negative bacteria (both aerobes and anaerobes), Rickettsia, spirochetes (Treponema pallidum), some protozoa (Entamoeba histolytica), and Chlamydia and Mycoplasma organisms.
Bacterial resistance to the tetracyclines develops slowly in a stepwise fashion. Cross-resistance among tetracyclines is probably complete. This resistance is caused by a decreased uptake of the tetracycline by the organism. In the study of sensitivity of organisms isolated from dental infections, one-fifth to three-fifths of S. viridans and one-fifth to two-fifths of S. aureus were found to be resistant to tetracycline. The advantage of penicillin over tetracycline in these aerobic gram-positive infections is clear.
Adverse Reactions
Although most adverse reactions to the tetracyclines occur infrequently, gastrointestinal distress is not uncommon.
 GASTROINTESTINAL EFFECTS
GASTROINTESTINAL EFFECTS
The gastrointestinal adverse effects include anorexia, nausea, vomiting, diarrhea, gastroenteritis, glossitis, stomatitis, xerostomia, and superinfection (moniliasis). The side effects are largely related to local irritation from alteration of the oral, gastric, and enteric flora.
If diarrhea occurs in a patient receiving tetracycline, the possibility of infectious enteritis, such as staphylococcal enterocolitis, intestinal candidiasis, and pseudomembranous colitis (secondary to Clostridium difficile overgrowth), must be ruled out. Some patients taking tetracyclines have developed a yellowish-brown discoloration of the tongue. This can occur with either topical or systemic administration. Patients with ill-fitting dentures are likely to have candidiasis (moniliasis) caused by superinfection associated with the areas of the oral mucosa tissue where breakdown has occurred.
 EFFECTS ON TEETH AND BONES
EFFECTS ON TEETH AND BONES
Tetracyclines are incorporated into calcifying structures. If they are used during the period of enamel calcification, they can produce permanent discoloration of the teeth and enamel hypoplasia (see Plate 15). Consequently, they should not be used during the last half of pregnancy or in children younger than 9 years. Tetracycline will affect the primary teeth if given to the mother during the last half of pregnancy or to the infant during the first 4 to 6 months of life. If tetracycline is administered between 2 months and 7 or 8 years of age, the permanent teeth will be affected. The mechanism involves the deposition of tetracycline in the enamel of the forming teeth. These stains are permanent and darken with age and exposure to light. They begin as a yellow fluorescence and progress with time to brown. This process is accelerated by exposure to light. The permanent discoloration ranges from light gray to yellow to tan. With large doses of tetracyclines, a decrease in the growth rate of bones has been demonstrated in the fetus and infants.
Minocycline can cause black pigmentation of mandibular and maxillary alveolar bone and the hard palate. When viewed through the mucosa, the pigment appears bluish. Other cases of oral pigmentation have been said to involve the crowns of the permanent teeth (half of incisal surface) and the gingival mucosa. The incidence of this oral pigmentation in adults is 10% after 1 year and 20% after 4 years of therapy. With discontinuation of minocycline, the pigmentation becomes less intense but is usually not completely reversible.
 HEPATOTOXICITY
HEPATOTOXICITY
The incidence of liver damage increases with the IV use of tetracyclines. Deaths have occurred, especially in pregnant women. Renal impairment leads to accumulation of tetracycline and may increase the likelihood of hepatic damage.
 NEPHROTOXICITY
NEPHROTOXICITY
Toxic renal effects with characteristic disorders of renal tubular function, producing Fanconi’s syndrome, have been reported after the use of old (degraded) tetracycline. Old or outdated tetracycline should be discarded to prevent future use. Because the nephrotoxic effect of the tetracyclines is additive with that of other drugs, tetracyclines should not be used concomitantly with other nephrotoxic drugs.
 HEMATOLOGIC EFFECTS
HEMATOLOGIC EFFECTS
Although uncommon, the hematologic changes hemolytic anemia, leukocytosis, and thrombocytopenic purpura have been reported after tetracycline therapy.
 SUPERINFECTION
SUPERINFECTION
With superinfection, resistant organisms multiply and may cause disease. One common situation, especially prevalent in the compromised host, is an overgrowth of C. albicans. Oral or vaginal candidiasis can result from the administration of oral tetracycline.
 PHOTOSENSITIVITY
PHOTOSENSITIVITY
Patients taking tetracyclines who are exposed to the sunlight sometimes react with an exaggerated sunburn. Although the incidence seems to vary with the different tetracyclines, patients receiving a prescription for a tetracycline should be told to use a sunscreen before exposure to the sun.
 OTHER EFFECTS
OTHER EFFECTS
Minocycline (min-oh-SYE-kleen) has been associated with CNS side effects, including lightheadedness, dizziness, and vertigo. Patients who will be driving a car should be warned about this reaction.
 ALLERGY
ALLERGY
Anaphylactic and various dermatologic reactions to the tetracyclines have occasionally occurred, but the overall allergenicity of these drugs is low. Glossitis and cheilosis have also been attributed to a hypersensitive reaction to tetracycline. A patient who is allergic to one tetracycline is almost certain to be allergic to all tetracyclines.
Drug Interactions
Divalent (Ca+2, Mg+2, Fe+2, Zn+2) and trivalent (Al+3) cations reduce the intestinal absorption of tetracyclines by forming nonabsorbable chelates of tetracycline with, for example, calcium. Dairy products containing calcium, antacids (Ca+2, Mg+2, Al+3), and mineral supplements (iron, calcium, zinc, or fortified foods) should not be taken within 2 hours of ingesting tetracycline. Reasonable quantities of dairy products can be taken with doxycycline and minocycline because there is less interference with absorption, but concomitant administration with antacids or mineral supplements should be avoided.
 ENHANCED EFFECT OF OTHER DRUGS
ENHANCED EFFECT OF OTHER DRUGS
Tetracycline enhances the effect of the oral sulfonylureas, which has the potential to result in hypoglycemia. The effects of digoxin, lithium, and theophylline may also be enhanced, which leads to toxicity from these agents with narrow therapeutic indices. Furosemide’s toxicity may also be increased by tetracycline.
 REDUCED DOXYCYCLINE EFFECT
REDUCED DOXYCYCLINE EFFECT
The barbiturates and phenytoin can reduce the action of doxycycline. The mechanism is stimulation of hepatic microsomal enzymes so that doxycycline is metabolized more rapidly.
 GENERAL ANTIBIOTIC INTERACTIONS
GENERAL ANTIBIOTIC INTERACTIONS
Like all the antibiotics, tetracyclines may reduce the effectiveness of oral contraceptives or increase the effectiveness of oral anticoagulants. Also, in most instances, mixing tetracyclines with another antibiotic results in antagonism, especially if the other antibiotic is bactericidal.
Uses
Tetracyclines, including both tetracycline and doxycycline, have extensive medical and dental use.
 MEDICAL
MEDICAL
Although active against a wide variety of microorganisms, tetracyclines are rarely the drug of choice for a specific infection. Occasionally, they are alternative drugs to treat chlamydial and rickettsial infections. They are used to treat acne (topically and systemically), pulmonary infections in patients with chronic obstructive pulmonary disease (COPD), and traveler’s diarrhea. Tetracyclines should not be used for prophylaxis against infective endocarditis except in one unusual situation in dentistry, which is discussed in the next section.
 DENTAL
DENTAL
Tetracyclines are not indicated as the drug of choice or the alternative drug of choice for dental infections unrelated to periodontal disease. They are often used for certain periodontal conditions. Conventional treatment with local measures should have failed before tetracycline therapy is initiated. A potential advantage of the tetracyclines in treatment of certain periodontal situations relates to their ability to concentrate in the gingival crevicular fluid. Because long-acting tetracyclines are concentrated to a greater extent in the gingival fluid and they require once-daily dosing, they may have some advantage over tetracycline itself. The ideal tetracycline therapy would be delivered directly to the gingival crevice, thereby greatly reducing the systemic dose. A variety of plastic strips, hollow fibers, or collars to deliver the tetracycline directly to the sulcus are being used but continue to be evaluated.
CLINDAMYCIN
Clindamycin (klin-da-MYE-sin) (Cleocin) is a bacteriostatic antibiotic effective primarily against gram-positive organisms and anaerobic Bacteroides species. Clindamycin is produced by adding a −Cl group to lincomycin, which is elaborated by Streptomyces lincolnensis, found in a soil sample taken near Lincoln, Nebraska. Clindamycin is structurally unrelated to any other antimicrobial agent other than lincomycin, which is not used.
Pharmacokinetics
Clindamycin may be administered orally, topically, intramuscularly, intravenously, or vaginally. Oral clindamycin is well absorbed, and food does not interfere with its absorption. It reaches its peak concentration in 45 minutes with a half-life of about 2.5 hours. Clindamycin is distributed throughout most body tissues, including bone, but not to the cerebrospinal fluid. Concentration in the bone can approximate that in the plasma. It crosses the placental barrier, and it is more than 90% bound to plasma proteins. Only about 10% of the active drug is eliminated in the urine. The majority of clindamycin is excreted as inactive metabolites in the urine and feces (via the bile).
Spectrum
The antibacterial spectrum of clindamycin includes many gram-positive organisms and some gram-negative organisms. The antibacterial action results from interference with bacterial protein synthesis. Clindamycin is bacteriostatic in most cases, although occasionally it can be bactericidal at higher blood levels.
Similar to erythromycin, clindamycin’s activity includes S. pyogenes and S. viridans, pneumococci, and S. aureus. In contrast to erythromycin, clindamycin is very active against several anaerobes, including Bacteroides fragilis and Bacteroides melaninogenicus, Fusobacterium species, Peptostreptococcus (anaerobic streptococci) and Peptococcus species, and Actinomyces israelii.
Bacterial resistance to clindamycin develops in a slow, stepwise manner. It occurs by mutations in the bacterial ribosomes that result in a decrease in affinity and binding capacity of these drugs. Cross-resistance between clindamycin and erythromycin is often noted. An antagonistic relationship has been observed between clindamycin and erythromycin because of competition for the same binding site (50S subunit) on the bacteria.
Adverse Reactions
The most commonly observed side effects of clindamycin are gastrointestinal, including diarrhea, nausea, vomiting, enterocolitis, and abdominal cramps. Glossitis and stomatitis have also been reported with these agents. The incidence of diarrhea with clindamycin is approximately 10%.
The development of pseudomembranous colitis (PMC), also known as antibiotic-associated colitis (AAC), has been a more serious consequence associated with clindamycin. It is characterized by severe, persistent diarrhea and the passage of blood and mucus. This colitis, which can be fatal, is caused by a toxin produced by the bacterium Clostridium difficile. It is associated not only with clindamycin but also with other antibiotics such as tetracycline, ampicillin, and the cephalosporins. Treatment of colitis includes discontinuation of the drug, vancomycin or cholestyramine administered orally, and fluid and electrolyte replacement. Systemically administered corticosteroids have sometimes proved helpful. Opioid-like agents, such as diphenoxylate and atropine (Lomotil), may exacerbate the condition and should not be used. PMC may occur during treatment, several weeks after cessation of antibiotic therapy, or without any antibiotic use.
 SUPERINFECTION
SUPERINFECTION
As with other antibiotics, superinfection by C. albicans is sometimes associated with the use of clindamycin.
Uses
Although clindamycin is effective against many gram-positive organisms, other agents are available that are at least as effective as clindamycin and do not usually cause PMC. The indications for treatment with clindamycin are limited to a number of infections caused by anaerobic organisms, especially Bacteroides species and some staphylococcal infections, when the patient is allergic to penicillin.
Many oral infections have been shown to contain a predominance of anaerobic organisms. Many of these anaerobes, such as Bacteroides oralis, Peptostreptococcus, Fusobacterium, and Veillonella species and clostridia, are sensitive to oral penicillin V. Clindamycin is the drug of choice for some Bacteroides species and other anaerobes, endocarditis prophylaxis with penicillin allergy, and some pelvic infections.
Mixed gram-positive and gram-negative anaerobic infections may be treated with clindamycin. The use of clindamycin when anaerobic osteomyelitis is suspected is indicated if the organism is susceptible. It is important to emphasize that clindamycin should be used only when specifically indicated, not indiscriminately, and the patient should be warned of the potential for PMC and informed about its symptoms (bloody diarrhea mixed with mucus). The dose of clindamycin is 150 to 300 mg q6h (qid).
METRONIDAZOLE
Metronidazole (me-troe-NI-da-zole) (Flagyl) is an antiinfective that is a synthetic nitroimidazole with trichomonacidal (Trichomonas vaginalis), amebicidal (Entamoeba histolytica species), and bactericidal action. It has exceptional action against most obligate anaerobes such as Bacteroides species. As with all antibiotics, resistance to this agent is increasing. It freely enters cells and is reduced into unknown polar compounds that do not contain the nitro group. This short-lived product is cytotoxic, but it causes DNA to lose its cyclic structure and inhibits nucleic acid synthesis, leading to death of the organisms. It affects cells whether they are or are not dividing.
In addition to its antiinfective effects, metronidazole also has antiinflammatory effects. It affects neutrophil motility, lymphocyte action, and cell-mediated immunity. What therapeutic purpose these actions might serve is yet to be identified.
Pharmacokinetics
Taken orally, metronidazole is well absorbed, with a peak level occurring between 1 and 2 hours after administration. Between 60% and 80% of a dose is excreted in the urine. Metabolites account for about 20% of the dose. Its half-life averages 8 hours, but with alcoholic liver disease it averages 18 hours. It is less than 20% protein bound. Metronidazole is somewhat concentrated in the gingival crevicular fluid, producing concentrations that are bactericidal to pathogenic periodontal organisms. Metronidazole is distributed into the cerebrospinal fluid, saliva, and breast milk in levels approximating that of the serum.
Spectrum
Metronidazole is bactericidal and penetrates all bacterial cells. The spectrum of action of metronidazole includes the protozoa T. vaginalis and E. histolytica. Metronidazole is active against obligate anaerobic bacteria such as Bacteroides, Fusobacterium, Veillonella, Treponema, Clostridium, Peptococcus, Campylobacter, and Peptostreptococcus organisms. The increased use of antibiotics is resulting in a continuing rise in the incidence of resistance. One should compare the spectrum of action of metronidazole with the bacteria responsible for periodontal conditions and the concentration effective against those bacteria to minimize resistance.
Adverse Reactions
Metronidazole’s most common adverse reactions involve the gastrointestinal tract. This side effect occurs in 12% of patients taking metronidazole. It includes nausea, anorexia, diarrhea, and vomiting. Epigastric distress and abdominal cramping have also been reported.
 CENTRAL NERVOUS SYSTEM EFFECTS
CENTRAL NERVOUS SYSTEM EFFECTS
Headache, dizziness, vertigo, and ataxia have been reported. Confusion, depression, weakness, insomnia, and serious convulsive seizures are rarely associated with metronidazole use.
 RENAL TOXICITY
RENAL TOXICITY
Cystitis, polyuria, dysuria, and incontinence can occur with metronidazole. Rarely, darkening of the urine as a result of a metabolite has been reported.
 ORAL EFFECTS
ORAL EFFECTS
Another effect that has been reported is a dry mouth. Often, an unpleasant or sharp metallic taste has been reported. Altered taste of alcohol has been noted. Glossitis, stomatitis, and a black-furred tongue are side effects the dental health care worker might observe. These side effects may be related to monilial overgrowth. Appendix E discusses xerostomia in more detail.
 OTHER EFFECTS
OTHER EFFECTS
Transient neutropenia in humans and carcinogenicity, mutagenicity, and tumorigenicity in lower life forms have been reported. Metronidazole is in Food and Drug Administration (FDA) pregnancy category B because administration to pregnant mice caused fetal toxicity. Administration of metronidazole for dental infections during pregnancy is contraindicated. Nursing mothers should not be given metronidazole unless milk is expressed and discarded beginning when the metronidazole is taken and continuing for 48 hours after discontinuing the drug.
Drug Interactions
When alcohol is ingested with metronidazole, a disulfiram-like reaction can occur. Disulfiram (Antabuse) is a drug used to treat persons with alcohol problems (see Chapter 25). Symptoms include nausea, abdominal cramps, flushing, vomiting, or headache. Alcohol should be avoided during metronidazole administration and for 1 day after therapy has ceased. Products, such as mouthwashes or elixirs, that contain alcohol should not be used during this period.
Metronidazole can potentiate the effect of warfarin. The combination of metronidazole and disulfiram has led to confusion and should be avoided. Drugs that stimulate liver microsomal enzymes, such as phenobarbital and phenytoin, can reduce the plasma levels of metronidazole. Before metronidazole is administered to patients, the possibility of a drug interaction should be checked.
Uses
Metronidazole is used for the treatment of infections caused by susceptible organisms in both medical and dental conditions. It has special usefulness because of its anaerobic spectrum.
 MEDICAL
MEDICAL
The medical uses of metronidazole include treatment of trichomoniasis, giardiasis, amebiasis, and susceptible anaerobic bacterial infections. It is effective against serious anaerobic infections of the abdomen, skeleton, and female genital tract. Endocarditis and lower respiratory tract infections caused by Bacteroides species are treated with metronidazole. It is available as oral tablets and capsules; vaginal cream and gel for vaginal infections; topical cream, gel, and lotions for the treatment of rosacea; and IV solution for anaerobic infections.
 DENTAL
DENTAL
Because of its anaerobic efficacy, metronidazole is useful in the treatment of many periodontal infections. One notable exception is that it has no action against A. actinomycetemcomitans. One advantage of metronidazole is that when prescribed generically it is inexpensive (see Figure 7-3).
CEPHALOSPORINS
The cephalosporin (sef-a-loe-SPOR-in) group of antibiotics is structurally related to the penicillins. Cephalosporins are active against a wide variety of both gram-positive and gram-negative organisms. The oral cephalosporin products, listed in Box 7-3, are divided into first-, second-, third-, and fourth-generation agents. Most third-generation cephalosporins are available for parenteral use. The orally active cephalosporins are discussed.
The source of the original cephalosporins was Cephalosporium acremonium, which was isolated from a sewer outlet near Sardinia in Italy. Because cephalosporins are true antibiotics, they were originally produced by organisms. Those available for oral use are relatively acid stable and highly resistant to penicillinase, but they are destroyed by cephalosporinase, an enzyme elaborated by some microorganisms.
Pharmacokinetics
The cephalosporins can be administered orally, intramuscularly, or intravenously. The agents that cannot be used orally are too poorly absorbed to provide adequate blood levels. The cephalosporins used orally are well absorbed. They are bound between 10% and 65% to the plasma proteins (see Box 7-3). After absorption, they are widely distributed throughout the tissues. Like penicillin, the cephalosporins are excreted by glomerular filtration and tubular secretion into the urine. Their half-lives vary between 50 and 240 minutes.
Spectrum
The cephalosporins, which are bactericidal, are active against most gram-positive cocci, penicillinase-producing staphylococci, and some gram-negative bacteria. They inhibit most Salmonella and Klebsiella organisms, some paracolon strains, and E. coli. Serratia and Enterobacter species, H. influenzae, indole-positive Proteus, methicillin-resistant staphylococci, and most Pseudomonas strains are unaffected. The generation of the cephalosporin (first, second, or third) designates the width of antimicrobial action; the first-generation width is narrower (gram-positive, few gram-negative) than the second-generation width (gram-positive, more gram-negative and anaerobes), and the third-generation agents (gram-positive weaker, many gram-negative and anaerobes) have the broadest spectrum of action.
Mechanism of Action
The mechanism of action of the cephalosporins is like that of the penicillins: inhibition of cell wall synthesis. They bind to enzymes in the cell membrane involved in cell wall synthesis. The cephalosporin acts as an analog of acyl-d-alanyl-d-alanine to produce a deficiency in the cell walls, leading to lysis. They are more effective against rapidly growing organisms (which explains the potential drug interaction between bacteriostatic and bactericidal antibiotics).
Adverse Reactions
In general, the cephalosporins have a low incidence of adverse reactions (excluding allergic reactions) and are well tolerated. They have more adverse reactions than penicillin VK. The following adverse reactions may occur.
 GASTROINTESTINAL EFFECTS
GASTROINTESTINAL EFFECTS
The most common adverse reaction associated with the cephalosporins is gastrointestinal, including diarrhea, nausea, vomiting, abdominal pain, anorexia, dyspepsia, and stomatitis.
 NEPHROTOXICITY
NEPHROTOXICITY
Evidence suggests that the cephalosporins may produce nephrotoxic effects under certain conditions. Although some have suggested that this is a toxic reaction, it may be an allergic reaction.
 SUPERINFECTION
SUPERINFECTION
As with all antibiotics, especially those with a broader spectrum of action, superinfection has been reported. Resistant gram-negative organisms are often the culprits.
 LOCAL REACTION
LOCAL REACTION
As with penicillin, the irritating nature of the cephalosporins can produce localized pain, induration, and swelling when given intramuscularly and abscess and thrombophlebitis when given intravenously.
 HEMOSTASIS AND DISULFIRAM-LIKE REACTION
HEMOSTASIS AND DISULFIRAM-LIKE REACTION
Certain parenteral cephalosporins can impair hemostasis or produce a disulfiram-like reaction. Dental health care workers do not use parenteral cephalosporins, and therefore this side effect is of no concern to dentistry.
 ALLERGY
ALLERGY
Various types of hypersensitivity reactions have been reported in approximately 5% of patients receiving cephalosporins. These reactions include fever, eosinophilia, serum sickness, rashes, and anaphylaxis. Large doses often produce a direct positive Coombs’ reaction (immune mechanism is attacking the patient’s own red blood cells). This can lead to a significant degree of hemolysis.
The cephalosporins and penicillin have similar structures; some cross-hypersensitivity can occur. Clinically, the incidence of hypersensitivity reactions to the cephalosporins is higher in patients with a history of penicillin allergy. The degree of cross-hypersensitivity reported is about 10%. Cephalosporins are often given to patients with a history of penicillin allergy, especially if the reaction was mild and in the distant past.
Uses
The cephalosporin antibiotics are indicated for the treatment of gram-positive organisms. The cephalosporins are indicated for infections that are sensitive to these agents but resistant to penicillin. They are especially useful in certain infections caused by gram-negative organisms such as Klebsiella. Their dental use includes prophylaxis for patients with “at-risk” joints who are undergoing dental procedures likely to produce bleeding. They are also used to treat infections with sensitive organisms when other agents are ineffective or cannot be used.
RATIONAL USE OF ANTIINFECTIVE AGENTS IN DENTISTRY
Figure 7-5 shows the progression of most dental infections. The early phase, stage 1, is primarily gram-positive organisms; the mixed stage, stage 2, has both aerobes and anaerobes; and the last stage, stage 3, is exclusively anaerobes. If incision and drainage is possible, most dental infections in patients with normal immunity, whether the infection is in stage 1, 2, or 3, do not need antiinfective agents.
Stage 1
Acute abscess and cellulitis are primarily the result of gram-positive organisms. The drug of choice in patients without a penicillin allergy is penicillin V 500 mg q6h for 5 to 7 days (actually the patient must take the antibiotic every day as long as symptoms persist plus 2 or 3 days). For those with an allergy to penicillin, erythromycin ethylsuccinate or clindamycin may be used (see Appendix D for a flowchart).
Stage 2
During stage 2, the infection is mixed. This can be handled by attacking either the gram-positive organisms or the anaerobes. The gram-positive organisms can be managed with the same drugs as in stage 1. To attack the anaerobes, an antiinfective with good anaerobic coverage is needed. The two antibiotics with the most anaerobic coverage are clindamycin and metronidazole. Penicillin V also has anaerobic coverage. If drainage can be established, antiinfective agents are not indicated in the immunocompetent patient.
Stage 3
In stage 3, the organisms have coalesced into one area and are almost solely anaerobic. Most often incision and drainage is sufficient. In fact, this sometimes happens spontaneously and the patient is “cured” (in his or her mind because he or she does not have pain). If chronic infection persists or if the patient is immunocompromised, use of an antibiotic with anaerobic coverage is warranted.
When a prescribed antiinfective is not effective, there may be several reasons for this outcome. If an antibiotic failure occurs, the patient must be reevaluated taking into account the following reasons why an antibiotic may be ineffective:
• Patient compliance: The patient may not be taking the antibiotic.
• Did not get the prescription filled: Was the patient informed about the benefit that the medicine would have? Was the patient informed about the risk of not taking the medicine? Consequences?
• Tried and failed to get the prescription filled: There was a long wait at the pharmacy. The children have to be picked up from school. The checkbook was forgotten. When the patient was told the price of the prescription, he or she was not able to pay for it. The patient was told that antibiotics can interfere with the effectiveness of birth control pills and she did not want to get pregnant.
• Got prescription filled but … : The patient noticed that the tablets “smelled bad” or were “hard to swallow” or “decided to take an herbal product.”
• Did not complete prescription: Patients state that they “began to feel better,” “forgot to take some pills,” “took a few but then quit,” “saved them for the next time I have a toothache,” and so on.
• Ineffective antibiotic: The antibiotic chosen may not be effective against the organism producing the infection. If antibiotics do not spawn a response after 2 or 3 days, consideration should be given to changing the antibiotic (check compliance first).
• Poor debridement: Dead tissue, purulent exudate, or foreign bodies were not completely removed from the site of infection.
• Resistant organism: The antibiotic may not be effective because the organism is resistant to the antibiotic chosen. Knowledge about the resistance patterns in the dental area is important to consider before prescribing an antibiotic.
• Concentration did not reach the site of the infection: There are several mechanisms by which an adequate concentration of the antibiotic does not reach the site of the infection. Lack of penetration may occur because of decreased vascularity, an isolated location or “walled off” area, or a drug interaction inactivating the antibiotic before absorption. Microvascular disease, often seen in diabetics, further reduces the blood flow and the amount of antibiotic sent to the area.
• Host defenses inadequate: The ability of the host’s immune system to fight the infection is very important in ridding the body of the infection.
ANTIMICROBIAL AGENTS FOR NONDENTAL USE
Vancomycin (van-koe-MYE-sin) (Vancocin) is an antibiotic elaborated by Streptomyces orientalis, an actinomycete found in soil samples from India and Indonesia. It is unrelated to any other antibiotic currently marketed. Because it has very poor gastrointestinal absorption and it causes irritation when used intramuscularly, it is usually administered only intravenously for a systemic effect. When given PO, it is being used to eradicate organisms within the gastrointestinal tract.
 SPECTRUM
SPECTRUM
Vancomycin is bactericidal and has a narrow spectrum of activity against many gram-positive cocci, including both staphylococci and streptococci. It acts by inhibition of bacterial cell wall synthesis. In the past, resistance did not develop readily, but recently vancomycin-resistant organisms have appeared. When resistance was uncommon, vancomycin was rarely used. After resistance to other organisms increased, the use of vancomycin increased. This led, predictably, to an increase in resistance to vancomycin. Cross-resistance with other antibiotics is not believed to occur because it has a different structure from other antibiotics.
 ADVERSE REACTIONS
ADVERSE REACTIONS
Except when vancomycin is given in large doses, significant toxic reactions are infrequent. With oral use, nausea, vomiting, and a bitter taste may be experienced. With IV use, an erythematous rash on the face and upper body has been reported (red man syndrome). Hypotension accompanied by flushing, chills, and drug fever are also associated with vancomycin.
Aminoglycosides
As the name implies, the aminoglycoside (a-mee-noe-GLYE-koe-side) antibiotics are made up of amino sugars in glycosidic linkage. In 1943, a strain of Streptomyces griseus was isolated that elaborated streptomycin. Further strains of Streptomyces species furnished neomycin, kanamycin, tobramycin, and amikacin, and Micromonospora organisms produced gentamicin and netilmicin. They are bactericidal and appear to inhibit protein synthesis and to act directly on the 30S subunit of the ribosome. The aminoglycosides are as follows:
 PHARMACOKINETICS
PHARMACOKINETICS
Because aminoglycosides are poorly absorbed after oral administration, they must be administered intramuscularly or intravenously for a systemic effect. Aminoglycosides are used orally for their local effect within the intestines. Before gastrointestinal surgery, aminoglycosides reduce the intestinal bacterial flora.
 SPECTRUM
SPECTRUM
The aminoglycosides are bactericidal and have a broad antibacterial spectrum. They are used primarily to treat aerobic gram-negative infections when other agents are ineffective. They have little action against gram-positive anaerobic or facultative bacteria.
 ADVERSE REACTIONS
ADVERSE REACTIONS
The adverse reactions of the aminoglycoside antibiotics seriously limit their use in clinical practice. Their major adverse effects include the following.
Ototoxicity: The aminoglycosides are toxic to the eighth cranial nerve, which can lead to auditory and vestibular (in ear) disturbances, or ototoxicity. Patients may have difficulty maintaining equilibrium and can develop vertigo. Hearing impairment and deafness, which can be permanent, have resulted from the administration of these agents. This side effect is more common in patients with renal failure because the drug accumulates in the body. Elderly patients are also more susceptible.
Chloramphenicol
Chloramphenicol (klor-am-FEN-i-kole) (Chloromycetin), a broad-spectrum, bacteriostatic antibiotic, inhibits bacterial protein synthesis by acting primarily on the 50S ribosomal unit. It is active against a large number of gram-positive and gram-negative organisms, Rickettsiae, and some Chlamydia organisms. It is particularly active against Salmonella typhi.
Chloramphenicol has fallen into disuse primarily because of its serious adverse effects, which include fatal blood dyscrasias such as aplastic anemia, agranulocytosis, hypoplastic anemia, and thrombocytopenia. Chloramphenicol can produce bone marrow suppression with pancytopenia. Although the incidence is low (1:40,000), this condition is often fatal. Chloramphenicol has no use in dentistry.
Sulfonamides
Sulfonamides were the first antibiotics that went on to pave the way for the antibiotic revolution.
 MECHANISM OF ACTION
MECHANISM OF ACTION
The structural similarity between the sulfonamide agents and p-aminobenzoic acid (PABA) is the basis for most of their antibacterial activity. Unlike humans, many bacteria are unable to use preformed folic acid, which is essential for their growth. They must synthesize folic acid from PABA. Because of their structural similarity to PABA, the sulfonamides competitively inhibit dihydropteroate synthetase, the bacterial enzyme that incorporates PABA into dihydrofolic acid, an immediate precursor of folic acid (Figure 7-6). Drugs that are metabolized to PABA (e.g., ester local anesthetics) could theoretically interfere with the action of the sulfonamides.
 SPECTRUM
SPECTRUM
The sulfonamides are bacteriostatic against many gram-positive and some gram-negative bacteria. They are often used in medicine to treat acute otitis media in children (H. influenzae), acute exacerbations of chronic bronchitis in adults (S. pneumoniae), and urinary tract infections (Klebsiella and Enterobacter organisms and E. coli). They are ineffective against S. viridans but are active against some Chlamydia organisms. Sulfonamides are also used for prophylaxis of Pneumocystis carinii pneumonitis and for traveler’s diarrhea caused by enterotoxigenic E. coli or Cyclospora organisms.
The readily absorbed sulfonamides are used for their systemic effects and are distributed throughout the body. Some poorly soluble sulfonamides, when given orally, act locally in the treatment of ulcerative colitis or before surgical procedures on the bowel.
 ADVERSE REACTIONS
ADVERSE REACTIONS
The most common adverse reaction to the sulfonamides is an allergic skin reaction. Patients with an allergy to “sulfa” drugs may exhibit some cross-hypersensitivity with thiazide diuretics and the sulfonylureas (used orally to treat diabetes). There is no cross-hypersensitivity between sulfa drugs and sulfites, sulfates, or sulfur.
The allergic reactions may manifest as rash, urticaria, pruritus, fever, a fatal exfoliative dermatitis, or periarteritis nodosa (serious blood vessel disease in which small- and medium-sized arteries become swollen and damaged). Other cutaneous allergic reactions include erythema nodosum, erythema multiforme, Stevens-Johnson syndrome, and epidermal necrolysis.
Other relatively common side effects include nausea, vomiting, abdominal discomfort, headache, and dizziness. Liver damage, depressed renal function, blood dyscrasias (agranulocytosis, thrombocytopenia, aplastic and hemolytic anemia), and precipitation of lupus erythematosus are seen less often.
Patients with human immunodeficiency virus (HIV) are much more likely to exhibit adverse effects (65%), such as rash, fever, or leukopenia, and to discontinue therapy. In patients with HIV, the sulfisoxazole-trimethoprim combination is used prophylactically to prevent P. carinii pneumonia.
The possibility of renal crystallization (crystalluria) must always be kept in mind with the sulfonamides. The earlier sulfonamides had low solubility in the urine, and there was danger of crystallization in the kidney. The new sulfonamides are more soluble and therefore less likely to precipitate in the kidney. This is the reason patients taking sulfonamides are encouraged to drink plenty of water.
Sulfamethoxazole-Trimethoprim
Trimethoprim (trye-METH-oh-prim), an antibacterial and antimalarial agent, and sulfamethoxazole (sul-fa-meth-OX-a-zole), a sulfonamide, are commonly used in combination (co-trimoxazole, SMX-TMP, Bactrim, or Septra). Because sulfamethoxazole (SMX) inhibits the incorporation of PABA into folic acid and trimethoprim (TMP) inhibits the reduction of dihydrofolate to tetrahydrofolate (see Figure 7-6), this combination inhibits two separate steps in the essential metabolic pathway of the bacteria, thus delaying resistance and leading to a synergistic effect.
SMX-TMP (Bactrim, Septra) is bacteriostatic against a wide variety of gram-positive bacteria and some gram-negative bacteria. Its adverse effects are similar to those of the sulfonamides. Approximately 75% of the adverse reactions associated with this combination involve skin disorders.
SMX-TMP is indicated in the treatment of selected urinary tract infections and selected respiratory and gastrointestinal infections. It is used extensively to treat acute otitis media in children, which is often caused by H. influenzae. A combination of erythromycin and sulfisoxazole (Pediazole) is also used to treat otitis media in children. SMX-TMP is used prophylactically to prevent P. carinii pneumonia in patients with AIDS, Serratia sepsis, and systemic Salmonella (ampicillin-resistant) and Shigella infections. SMX-TMP has no documented use in dentistry, but pediatric patients coming to the dental office may be taking it prophylactically for prevention of chronic ear infections.
Nitrofurantoin
Nitrofurantoin (nye-troe-fyoor-AN-toyn) (Macrodantin) possesses a wide antibacterial spectrum including both gram-positive and gram-negative bacteria. It is bacteriostatic against many common urinary tract pathogens, including E. coli. Many strains of Klebsiella and Enterobacter and all strains of P. aeruginosa are resistant. The most common adverse reactions are nausea, vomiting, and diarrhea, but taking the drug with food decreases these effects. Nitrofurantoin can also cause a brownish discoloration of urine. Many hypersensitivity reactions are associated with nitrofurantoin. Nitrofurantoin is used in the treatment or prophylaxis of certain urinary tract infections.
Quinolones (Fluoroquinolones)
A group of orally effective antibacterial agents, called the quinolones (KWIN-a-lones), are chemically related to nalidixic (nal-i-DIX-ik) acid (NegGram). The number of agents in this group of drugs has risen exponentially. This group may have potential use in dentistry because of their spectrum of action. As with all antibiotics, their overuse produces resistance. They are bactericidal against most gram-negative organisms and many gram-positive organisms. They are the first orally active agents against certain Pseudomonas species. There is no cross-resistance with other antimicrobial agents.
The mechanism of action of the quinolones is unique and involves antagonism of the A subunit of DNA gyrase, which is an enzyme involved in DNA synthesis. The interference with DNA gyrase results in cell death and resistance is not transferred from a resistant bacteria to an unexposed bacteria. Examples of the fluoroquinolones are listed in Box 7-4. The discussion concentrates on ciprofloxacin (sip-roe-FLOKS-a-sin) (Cipro), a prototype of the quinolones.
 PHARMACOKINETICS
PHARMACOKINETICS
Ciprofloxacin is well absorbed orally and is eliminated with a half-life of 4 hours. Both antacids and probenecid interfere with ciprofloxacin’s absorption and serum concentration. Patients should be well hydrated to prevent any possibility of crystalluria (drink water while taking).
 SPECTRUM
SPECTRUM
Ciprofloxacin is bactericidal against a wide range of gram-negative organisms, including Klebsiella and Enterobacter species, E. coli, P. aeruginosa, and gram-positive organisms such as S. aureus. Its special spectrum is against Pseudomonas organisms. Unlike other antiinfective agents, an additive action may result when ciprofloxacin is combined with other antimicrobial agents. The emergence of organisms resistant to the fluoroquinolones and the cross-resistance among the fluoroquinolones is increasing. Use of fluoroquinolones in chickens may be partially responsible for increasing resistance.
Ofloxacin (o-FLOKS-a-sin) (Floxin) is the quinolone with activity against organisms present in dental infections. When the spectrums of ciprofloxacin and ofloxacin are compared, ofloxacin’s spectrum of action most closely parallels the spectrum of microbes found in dental infections. After consulting many sources for information about the spectrum of action of the fluoroquinolones, the conclusion reached is that intraspecies variation is at least as great as interspecies variation. In other words, spectrum of action is difficult to predict and depends on which sample of microorganisms is tested. Because of this action, ciprofloxacin or other quinolones may be used in dentistry in the future.
 ADVERSE REACTIONS
ADVERSE REACTIONS
Gastrointestinal Effects: Nausea, diarrhea, vomiting, painful oral mucosa, bad taste, and oral candidiasis have been reported. Pseudomembranous colitis has been seen in patients taking quinolones.
Central Nervous System Effects: CNS adverse reactions include headache, restlessness, lightheadedness, and insomnia. CNS stimulation has been noted.
Hypersensitivity: Rash, pruritus, urticaria, hyperpigmentation, and edema of the lips have been noted. Fluoroquinolones are associated with photosensitivity reactions when the patient is exposed to the sun. The patient should be advised to use sunscreen or wear clothes that cover the whole body. A few anaphylactic reactions have been reported.
Other Effects: Disturbed vision, joint pain, renal problems, and palpitations have rarely been reported. An unusual reaction affecting the Achilles tendon has become fairly common in patients taking quinolones. These agents can produce tendonitis or tendon rupture in the Achilles tendon, so exercise should be limited. Hepatotoxicity has rarely been attributed to the quinolones.
 USES
USES
Ciprofloxacin is indicated for lower respiratory tract, skin, bone and joint, and urinary tract infections caused by susceptible organisms. Because of the spectrum of the quinolones, future use may include dental periodontal disease. In summary, the new quinolones have an advantage over other antimicrobial agents because of their unique mechanism of action, making the development and transfer of resistance more difficult. Their gram-negative spectrum coupled with their oral efficacy and bactericidal action makes these agents a welcome addition to the antimicrobial armamentarium. Many more members of this group of agents currently have been released, and more wait in the wings. Overuse of these agents has already begun, and it is hoped that they do not become much less useful as a result of overprescribing.
ANTITUBERCULOSIS AGENTS
The treatment of tuberculosis (TB), a disease caused by the acid-fast bacterium Mycobacterium tuberculosis, is difficult for several reasons.
• First, patients with TB often have inadequate defense mechanisms (e.g., AIDS).
• Second, tubercle bacilli develop resistant strains easily and possess unusual metabolic characteristics, including long periods of inactivity (they become dormant) when they are resistant to treatment.
• Third, most of the drugs available are not bactericidal and because of their toxicity often cannot be used in sufficient doses.
• Finally, people using antituberculosis agents often do not take them as prescribed.
For these reasons, the development of multidrug-resistant TB (MDR TB) has continued to increase because of its spread in patients with HIV, the homeless, and in other countries. (On an airline flight, a patient with active TB has been reported to have infected several other passengers. The latest report of the spread of TB infection occurred in a fast-food line.) The newest development in the worldwide problem with TB is the practice of manufacturing and selling drugs used for treatment of TB containing only a fraction of the labeled amount (counterfeit tablets).
Treatment of TB relies almost entirely on chemotherapy (Table 7-7). Because of the problem of resistance, at least three drugs are administered concurrently in all active cases. Isoniazid (INH), rifampin, and pyrazinamide are combined for the treatment of pulmonary TB. INH and rifampin are continued every day for 9 to 12 months. Pyrazinamide is continued for 2 months. With susceptible organisms, a patient, if compliant, usually becomes noninfective within 2 to 3 weeks. This compliance presents a problem because patients often stop taking their medication before the designated time.
TABLE 7-7
TB, Tuberculosis; CNS, central nervous system; GI, gastrointestinal; IM, intramuscular; INH, isonicotinyl hydrazine; MDR TB, multidrug-resistant tuberculosis; PZA, pyrazinamide.
*Cyclopentyl rifamycin.
The Centers for Disease Control and Prevention (CDC) recommends that the following patients receive INH because they are at risk of developing TB: close contacts of recently diagnosed patients, patients with a positive skin test and radiographic findings consistent with nonprogressive TB, patients whose skin test has become positive (converted), and immunosuppressed patients. If patients have been vaccinated against TB (with Bacillus Calmette-Guérin [BCG]), skin tests (purified protein derivative [PPD]) will always be positive, and chest x-rays are required for screening. Because many people have TB, it is not unlikely that a TB-positive person may come in and request treatment. If a person with active TB seeks oral health care, contact that person’s physician and delay health care until the disease is no longer in the active state.
Isoniazid
Isoniazid (eye-soe-NYE-a-zid) (Laniazid), or INH, is bactericidal only against actively growing tubercle bacilli. The mechanism of action may relate to inhibition of mycolic acid synthesis resulting in disruption of the bacterial cell wall. “Resting” bacilli exposed to the drug are able to resume normal growth when the drug is removed. Within a few weeks after beginning therapy, resistant strains develop.
 PHARMACOKINETICS
PHARMACOKINETICS
INH is readily absorbed from the gastrointestinal tract and is distributed throughout the body. Its metabolism varies by race. Most Eskimos and Japanese are fast acetylators, whereas whites and blacks in the United States are split 50-50 between fast and slow acetylators. The ability to acetylate rapidly is inherited as an autosomal dominant trait. Whether this ability to metabolize INH rapidly is related to the chance of developing INH-induced hepatitis is unknown. The half-life for fast acetylators is 1.5 hours and for slow acetylators is 3 hours.
 ADVERSE REACTIONS
ADVERSE REACTIONS
The incidence of all adverse reactions to INH is approximately 5%. The most common adverse reaction, occurring in about 20% of patients, involves the nervous system, which may be a result of INH causing vitamin B6 depletion. Peripheral and optic neuritis, muscle twitching, toxic encephalopathy, insomnia, restlessness, sedation, incoordination, convulsions, and even psychoses have been reported. These neurotoxic symptoms can be prevented by coadministration of pyridoxine (vitamin B6).
The other major adverse effect associated with INH is hepatotoxicity. Approximately 1% of patients taking INH exhibit clinical hepatitis and up to 10% develop abnormal laboratory values. Some cases of hepatitis have been fatal. The risk for developing this adverse effect is age related: it rarely occurs in patients younger than 20 years, whereas 2.5% of patients older than 50 develop hepatitis. This differential in incidence of hepatitis by age may modify the treatment plan for an individual patient. Other side effects include hematologic effects, gastrointestinal effects, dryness of the mouth, and a lupuslike reaction or rheumatic syndrome with arthralgia. Urinary retention and gynecomastia have been noted in males. Hypersensitivity reactions, including rashes, hepatitis, lymphadenopathy, and fever, are occasionally reported. The choice of whether to use INH depends on many factors such as patient age, presence of renal or hepatic deficiency, history of seizures, gastrointestinal disturbances, alcoholism, or history of neurotoxicity.
INH is both an inhibitor and an inducer of cytochrome P-450 2E isoenzymes. The benzodiazepines that are oxidized in the liver, such as diazepam and midazolam, may have an increased effect in patients taking INH. Foods (e.g., cheese and fish) and drugs that are contraindicated with monoamine oxidase (MAO) inhibitors may also react with INH.
Rifampin
Rifampin (RIF-am-pin) (Rifadin, Rimactane) is a semisynthetic derivative of rifamycin, an antibiotic produced by Streptomyces mediterranei. Its mechanism of action involves inhibition of DNA-dependent ribonucleic acid (RNA) polymerase, which then suppresses the initiation of chain formation. It is active against M. tuberculosis and many gram-positive and some gram-negative bacteria. Rifampin’s spectrum also includes S. aureus, N. meningitidis, H. influenzae, and Legionella species. In TB, resistance quickly develops to rifampin administered alone in a one-step process as a result of a change in the RNA polymerase. Administering rifampin with other antituberculosis agents reduces the development of resistance.
 PHARMACOKINETICS
PHARMACOKINETICS
Rifampin is absorbed from the gastrointestinal tract and eliminated in the bile, where enterohepatic circulation occurs. Its half-life is 1.5 to 5 hours and is increased in hepatic disease but unaltered by renal disease. The half-life is reduced by INH coadministration because of enzyme induction. By blocking the hepatic uptake of rifampin, probenecid increases the concentration of rifampin in the serum.
 ADVERSE REACTIONS
ADVERSE REACTIONS
The most common adverse reactions are gastrointestinal, including anorexia, stomach distress, nausea, vomiting, abdominal cramps, and diarrhea. Occasionally, rashes, thrombocytopenia, nephritis, and impairment of liver function are seen. A flulike reaction can occur with infrequent administration. Rifampin gives a red-orange color to body fluids, including tears (affecting contact lenses), urine, feces, saliva, and sweat.
Pyrazinamide
Pyrazinamide (peer-a-ZIN-a-mide) (PZA), a relative of nicotinamide, is well absorbed and widely distributed throughout the body. It is hepatotoxic and can produce rash, hyperuricemia, and gastrointestinal disturbances. The CDC currently recommends PZA for use during the first 2 months with INH and rifampin to treat TB. PZA, which used to be a tertiary drug, now plays a much more important role than it had in the past. One treatment regimen includes the use of INH and rifampin every day for 9 to 12 months. PZA is continued for 2 months (Figure 7-7).
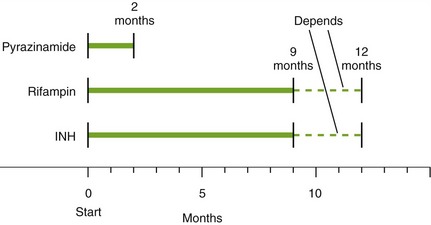
FIGURE 7-7 Treatment and time course of tuberculosis with a minimum of three drugs used. Depending on the recommended regimen, treatment with rifampin and isoniazid (INH) may last between 9 and 12 months.
If a patient is compliant and the organisms susceptible, he or she usually becomes noninfective within 2 to 3 weeks to 2 to 3 months. A negative sputum sample is required to ensure that the patient is noninfective.
Ethambutol
Ethambutol (e-THAM-byoo-tole) (Myambutol) is a synthetic tuberculostatic agent effective against M. tuberculosis. Resistance among tubercle bacilli develops very rapidly when this drug is used alone.
The most important side effect is optic neuritis, resulting in a decrease in visual acuity and loss of ability to perceive red and green. Periodic ophthalmologic examinations are recommended. Other side effects include rash, joint pain, gastrointestinal upset, malaise, headache, and dizziness. This drug is used when other antituberculosis agents cannot be used or resistance is encountered.
TOPICAL ANTIBIOTICS
In general, the use of topical antibiotics is discouraged. Systemic administration is superior in most cases. If an agent is used topically, it should be one that cannot be used systemically. One old and one newer product are mentioned briefly.
Neomycin, Polymyxin, and Bacitracin
The combination of an aminoglycoside, neomycin (nee-oh-MYE-sin), and two polypeptide antibiotics, polymyxin (pol-i-MIX-in) and bacitracin (bass-i-TRAY-sin), is available in ointment form (Neosporin, triple antibiotic ointment).
Neomycin affects gram-negative organisms, and polymyxin and bacitracin affect gram-positive organisms. This combination product is used topically on scratches; if the wound is infected, systemic antibiotics are indicated.
Mupirocin
Mupirocin (myoo-PEER-oh-sin) (Bactroban) is a topical antibacterial produced by Pseudomonas fluorescens. Mupirocin inhibits protein synthesis by binding to bacterial isoleucyl transfer-RNA synthetase. It shows no cross-resistance with other antibiotics. It is active versus certain Streptococcus and Staphylococcus organisms and is indicated for the topical treatment of impetigo. Local itching and stinging have been reported. Mupirocin is as effective as the usual systemic treatments (penicillinase-resistant penicillins) and has fewer side effects.
In dentistry, mupirocin can be used to treat the bacterial infection with streptococci or staphylococci that are occasionally present with angular cheilitis (chronic inflammatory condition at the corners of the mouth). The secondary infection can be determined by its clinical presentation. Because angular cheilitis most commonly is a fungal infection, topical antifungal agents should be used first.
ANTIBIOTIC PROPHYLAXIS USED IN DENTISTRY
Infective endocarditis is caused by an infection of the heart valves or endocardium with an organism. Infective endocarditis often begins with sterile vegetative cardiac lesions consisting of amalgamations of platelets, fibrin, and bacteria. When bacteria are introduced into the bloodstream, they may infect the damaged valves. Infective endocarditis can also occur in patients without predisposing cardiac factors. The difficult question, to which there is currently no proved answer, is this: “Which factors will be predictive in identifying patients in whom appropriate antibiotics will prevent infective endocarditis when specific dental procedures are performed?”
Prevention of Infective Endocarditis
Prophylaxis for infective endocarditis is based on the concept (which may not be true) that giving certain antibiotics to certain patients before certain procedures can prevent these patients from developing infective endocarditis. In March 2007, the journal Circulation published the most current guidelines from the American Heart Association (AHA) for antibiotic prophylaxis before dental procedures to prevent infective endocarditis. The newest guidelines recommend that only those people who are at highest risk for bad outcomes from infective endocarditis receive short-term preventive antibiotics before select, common, and routine dental procedures.
According to the AHA, these newest guidelines are based on a comprehensive review of published studies that suggest that infective endocarditis is more likely to occur from bacteria entering the bloodstream as a result of daily activities than from a dental procedure. There was also no compelling evidence in the medical and dental literature that antibiotic prophylaxis before a dental procedure would prevent infective endocarditis in those at risk for developing it. Also, the antibiotics used to prevent infective endocarditis carry risks, including adverse effects, risk of fatal allergic reactions, and the possibility of bacterial resistance.
For every situation in which it may be appropriate to use prophylactic antibiotics, the following factors should be considered:
• The specific dental procedure being performed
• The cardiac and medical condition of the patient
The updated guidelines also emphasize that maintaining optimal oral health and practicing daily oral hygiene are more important in reducing the risk for infective endocarditis than taking an antibiotic before a dental procedure.
 DENTAL PROCEDURES
DENTAL PROCEDURES
When dental treatment is rendered (including periodontal probing), organisms are more likely to enter the blood supply, producing bacteremia. Bacteremia is also produced when eating potato chips, brushing teeth, or chewing wax. The organisms can then produce infective endocarditis.
To determine whether prophylactic antibiotic coverage is needed before a dental procedure, see the infective endocarditis decision tree in Appendix D. According to the 2007 guidelines from the AHA regarding prophylactic antibiotic coverage, one should ask the following:
• Does the cardiac/medical condition warrant prophylaxis? Is the patient at highest risk for bad outcomes from infective endocarditis?
• Will the dental procedure to be performed involve manipulation of gingival tissue or the periapical region of the teeth or perforation of the oral mucosa?
Only if both of these questions are answered in the affirmative would prophylaxis be indicated.
Depending on the dental procedure being performed, patients may or may not need prophylactic antibiotic coverage. Box 7-5 divides dental procedures into those procedures that involve manipulation of the gingival mucosa or the periapical region of the teeth or perforation of the oral mucosa and those that do not require prophylaxis. Clinical judgment will determine the need either for or against antibiotic coverage for each patient.

BOX 7-5 DENTAL PROCEDURES FOR WHICH ENDOCARDITIS PROPHYLAXIS IS REASONABLE FOR PATIENTS WITH SPECIFIC CARDIAC CONDITIONS Rights were not granted to include this box in electronic media. Please refer to the printed book. From Wilson W, Taubert KA, Gewitz M et al: Prevention of infective endocarditis: guidelines from the American Heart Association, Circulation 116:1736, 2007. American Heart Association
 CARDIAC CONDITIONS
CARDIAC CONDITIONS
Patients with cardiac conditions can be divided into groups based on the cardiac condition (Box 7-6). The first group contains patients at highest risk for developing infective endocarditis (e.g., prosthetic cardiac valve, previous infective endocarditis) and who suffer the worst outcomes. Oral antibiotic prophylaxis is required for these patients if the dental procedure warrants it. The second cardiac group includes those conditions that do not require prophylactic antibiotic coverage (e.g., coronary bypass surgery after 6 months). Several new additions to this group include mitral valve prolapse and rheumatic heart disease. Although these people still have a lifelong risk of infective endocarditis, they have a much higher risk from a random blood-borne bacterial infection from day-to-day activities than from a dental or medical procedure. These changes were made based on the most current recommended guidelines from the AHA for preventing infective endocarditis.

BOX 7-6 CARDIAC CONDITIONS ASSOCIATED WITH THE HIGHEST RISK OF ADVERSE OUTCOMES FROM ENDOCARDITIS Rights were not granted to include this box in electronic media. Please refer to the printed book. From Wilson W, Taubert KA, Gewitz M et al: Prevention of infective endocarditis: guidelines from the American Heart Association, Circulation 116:1736, 2007. American Dental Association
 ANTIBIOTIC REGIMENS FOR DENTAL PROCEDURES
ANTIBIOTIC REGIMENS FOR DENTAL PROCEDURES
Table 7-8 lists the antibiotic regimens for prophylaxis of endocarditis before dental procedures. These situations include treating those patients with no allergies and those patients allergic to penicillin antibiotics or ampicillin.

TABLE 7-8 PROPHYLACTIC ANTIBIOTIC DRUG REGIMENS FOR DENTAL PROCEDURES Rights were not granted to include this table in electronic media. Please refer to the printed book. Modified from Wilson W et al: Prevention of infective endocarditis; guidelines from the American Heart Association, Circulation 116:1736, 2007. American Heart Association
When a patient’s physician is contacted concerning the patient, the current medical condition of the patient should be explored. Based on the patient’s medical status and the current recommendations, the dental health care worker determines whether antibiotics are indicated. Explain to the medical provider that the choice of therapy will be determined based on the patient’s medical condition and the dental treatment being rendered. An agreement between the dentist and medical provider should be reached, but the dentist should not agree to practice outside the recommendation. This will minimize suggestions of inappropriate antibiotics or regimens. When no recommendations exist and the literature is contradictory, the choice should be based on a consensus between the dental health care worker and the patient’s physician. If the patient is at highest risk for bad outcomes from infective endocarditis, then the usual regimens should be administered before providing dental treatment.
Prosthetic Joint Prophylaxis
Both the American Dental Association (ADA) and the American Academy of Orthopaedic Surgeons (AAOS) continue to recommend that all patients receive antibiotic prophylaxis before certain dental procedures during the first 2 years after total joint replacement (Box 7-7). Like other preprocedure prophylaxis situations, the joint condition, the dental procedures, and the appropriate drugs must be considered.
Appendix D provides a flowchart to determine when prophylaxis is needed in a patient with a joint prosthesis. Box 7-5 may be consulted to determine which dental procedures require antibiotic prophylaxis.
Whether the use of antibiotics is indicated after the patient is 2 years postreplacement surgery should be determined by those involved most closely with the patient’s condition. The ADA and the AAOS recommend that antibiotic prophylaxis before dental procedures, greater than 2 years after joint replacement, be reserved for those people that are immunocompromised or have certain comorbid conditions (see Box 7-7). Appendix D includes a decision tree on total joint replacement.
Noncardiac Medical Conditions
Patients with noncardiac medical conditions may also require prophylactic antibiotic coverage before dental procedures, but lack of agreement among practitioners for these situations causes confusion. For some conditions in this group, there is consensus that antibiotics are indicated or that antibiotics are not indicated. For other conditions there is little consensus. One should consult with the most current guidelines established by the AHA to determine if antibiotic prophylaxis is necessary.
American Dental Association, American Academy of Orthopaedic Surgeons. Advisory statement: Antibiotic prophylaxis for dental patients with total joint replacements. J Am Dent Assoc. 2003;134:895.
Berbari, EF, Hanssen, AD, Duffy, MC, et al. Risk factors for prosthetic joint infection: case-control study. Clin Infect Dis. 1998;27:1247.
Brause, BD. Infections associated with prosthetic joints. J Antimicrob Chemother. 1989;23:676.
Ching, DW, Gould, IM, Rennie, JA, et al. Prevention of late haematogenous infection in major prosthetic joints. J Antimicrob Chemother. 1989;23:676.
Fitzgerald, RH, Jacobson, JJ, Luck, JV, et al. Antibiotic prophylaxis for dental patients with total joint replacements. J Am Dent Assoc. 1997;128(7):1004.
Garvin, J, Antibiotic prophylaxis: making sense of new AHA guidelines. ADA News June 2007. www.ada.org/prof/resources/pub/adanews/adanewsarticle.asp?articleid=2567. Accessed September 20, 2007
Hersh, EV. Adverse drug interactions in dental practice: interactions involving antibiotics. J Am Dent Assoc. 1999;130(2):236.
Jacobson, JJ, Millard, HD, Plezia, R, et al. Dental treatment and late prosthetic joint infections. Oral Surg Oral Med Oral Pathol. 1986;61:413.
Jacobson, JJ, Patel, B, Asher, G, et al. Oral Staphylococcus in elderly subjects with rheumatoid arthritis. J Am Geriatr Soc. 1997;45:1–5.
Johnson, DP, Bannister, GG. The outcome of infected arthroplasty of the knee. J Bone Joint Surg Br. 1986;68(2):289.
Murray, RP, Bourne, MH, Fitzgerald, RH, Jr. Metachronous infection in patients who have had more than one total joint arthroplasty. J Bone Joint Surg Am. 1991;73(10):1469.
Poss, R, Thornhill, TS, Ewald, FC, et al. Factors influencing the incidence and outcome of infection following total joint arthroplasty. Clin Orthop. 1984;182:117.
Wilson, W, Taubert, KA, Gewitz, M, et al. Prevention of infective endocarditis: guidelines from the American Heart Association. Circulation. 2007;116:1736.
CLINICAL SKILLS ASSESSMENT
1. Under what conditions is antibiotic prophylaxis necessary?
2. Should antibiotic prophylaxis be given to a patient with prosthetic joint before dental procedures? Describe the factors, if any, that would influence the decision.
3. What antibiotic dose and dosing regimen should be used?
4. What should a patient be told about an antibiotic?
5. What are the adverse reactions associated with penicillin? What should a patient be told about them?
6. What are some reasons that a patient might be noncompliant with medication?
7. A patient has called and complained of nausea, itching, and a rash after taking a prescription for penicillin. What is happening to this patient?
8. What should the patient do?
9. Is nausea caused by an allergic reaction or is it a side effect of penicillin?
10. Can a patient allergic to penicillin receive other types of penicillin antibiotics?
11. What is the role of tetracycline antibiotics in the treatment of periodontitis?
12. What are the different dose forms available and how do they differ from one another?
13. What are the adverse reactions associated with tetracycline, and what should a patient be told about them?
14. Who is tetracycline contraindicated in and why?
15. What is the role of metronidazole in dentistry, and what are its adverse effects? Are there any drug/food interactions?
16. Compare and contrast antituberculosis drugs in terms of mechanism of action and adverse effects.
17. What are the dental concerns associated with tuberculosis and when is it okay to treat a person with tuberculosis?
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
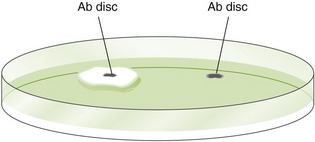
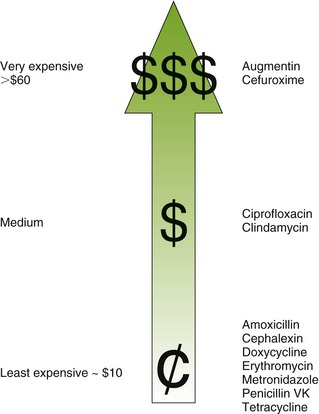
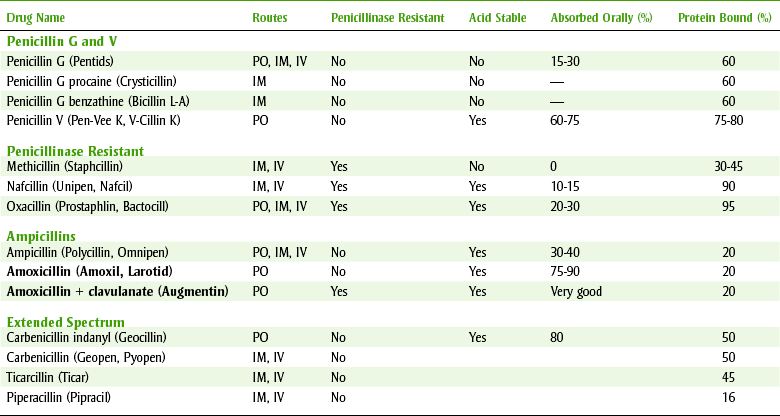
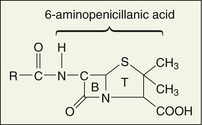
 hour
hour