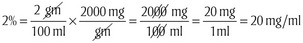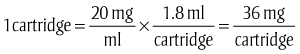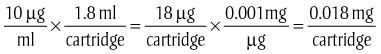Local Anesthetics
1 Discuss the history and reasons for the use of local anesthetics in dentistry.
2 Explain the mechanism of action, pharmacokinetics, pharmacologic effects, and adverse reactions of local anesthetics.
3 Describe the types and workings of each of the drugs used in local anesthetic solutions and summarize the factors involved in the choice of a local anesthetic.
4 Briefly discuss the use of and types of topical anesthetics used in dentistry.
No drugs are used more often in the dental office than the local anesthetic agents. Because their use can become routine, it is easy to forget that these agents have a potential for systemic effects in addition to the desired local effects. Dentists and, in some states, dental hygienists are responsible for the administration of local anesthetic agents in certain situations. With this duty comes the need for an in-depth knowledge of the local anesthetic agents.
HISTORY
“Painless” dentistry, through the use of a local anesthetic, is a relatively recent development. It began with the observation that the indigenous people of the South American Andes chewed certain leaves that made them feel better. The active ingredient of the leaves was cocaine, isolated by Niemann in 1860. He noted that tasting this substance produced not only the loss of taste but also of the sensation of pain (Figures 9-1 and 9-2). In 1884, Koller noted that cocaine instilled in the eye produced complete anesthesia. Its use in eye surgery was immediately adopted. During this time, Sigmund Freud was also experimenting with cocaine and its effects on the central nervous system (CNS). CNS stimulation, toxicity, and the potential for abuse were quickly recognized as major problems with the widespread use of cocaine as a local anesthetic.

FIGURE 9-1 The conduction of nerve impulses that lead a patient to experience pain can be compared to a fuse. The fuse is the “nerve,” and the dynamite is the “brain.” If the fuse is lit and the flame reaches the dynamite, an explosion occurs and the patient experiences pain. (From Malamed SF: Handbook of local anesthesia, ed 5, St Louis, 2004, Mosby.)

FIGURE 9-2 Using the same example as in Figure 9-1, local anesthetic is placed at some point between the pain stimulus and the brain (“dynamite”). The nerve impulse travels up to the point of the local anesthetic application and then “dies,” never reaching the brain. Thus the patient does not experience pain. (From Malamed SF: Handbook of local anesthesia, ed 5, St Louis, 2004, Mosby.)
The search for a more acceptable local anesthetic for dentistry continued. Einhorn synthesized procaine in 1905, but it was not until many years later that its use in dentistry became common. In 1952, the amide lidocaine (Xylocaine) was released, and mepivacaine (Carbocaine) was released in 1960. More recently, bupivacaine (Marcaine) has been made available for dental use. The search for the perfect local anesthetic agent continues.
IDEAL LOCAL ANESTHETIC
Although local anesthesia can be produced by several different agents, many are not clinically acceptable. The ideal local anesthetic should possess certain properties (Box 9-1). No local anesthetic agent in use today meets all of these requirements, although many acceptable agents are available.
CHEMISTRY
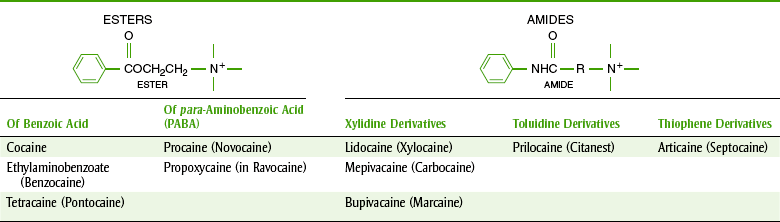
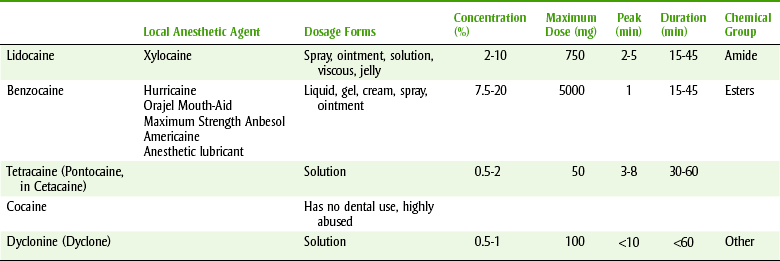
Local anesthetic agents are divided chemically into two major groups: the esters and the amides (Table 9-1). A few agents fall outside these two groups and are called other. The clinical importance of this division is associated with potential allergic reactions. A patient who has an allergy to one group is more likely to exhibit a hypersensitivity reaction to other agents within the same group. Cross-hypersensitivity between the amides and the esters is unlikely. The structure of local anesthetics is composed of the following three parts:
The aromatic nucleus (R) is lipophilic (lipid soluble), and the amino group is hydrophilic (water soluble). The esters are largely metabolized in the plasma and the amides in the liver.
MECHANISM OF ACTION
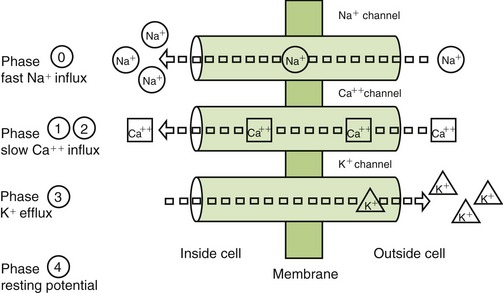
A resting nerve fiber has a large number of positive ions (cations) on the outside (electropositive) and a large number of negative ions (anions) on the inside (electronegative). The nerve action potential results in the opening of the sodium channels and an inward flux of sodium, resulting in a change from the −90-mV potential to a +40-mV potential (Figure 9-3 and Box 9-2). The outward flow of potassium ions repolarizes the membrane and closes the sodium channels. Local anesthetics attach themselves to specific receptors in the nerve membrane. After combining with the receptor, the local anesthetics block conduction of nerve impulses (thus the term nerve block) by decreasing the permeability of the nerve cell membrane to sodium ions. This then decreases the rate of depolarization of the nerve membrane, increases the threshold for excitability, and prevents the propagation of the action potential. Local anesthetics may reduce permeability by competing with calcium for the membrane binding sites and by preventing the onset of nerve conduction.
Ionization Factors
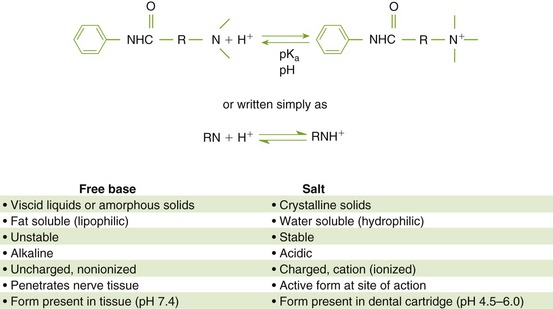
The local anesthetic agents are weak bases occurring equilibrated between their two forms, which are the fat-soluble (lipophilic) free base and the water-soluble (hydrophilic) hydrochloride salt (Figure 9-4). The proportion of drug in each form is determined by the pKa* of the local anesthetic and the pH of the environment. In the acidic pH of the dental cartridge (4.5), the proportion of the drug in the ionized form increases, thereby increasing solubility. Once injected into the tissues (pH 7.4), the amount of local anesthetic in the free-base form increases. This provides for greater tissue (lipid) penetration. In the presence of an acidic environment, such as infection or inflammation (pH lower), the amount of free base is reduced (more in ionized form), which is one reason dental anesthesia with a local anesthetic is more difficult when infection is present. Other reasons include dilution by fluid, inflammation, and vasodilation in the area. Although the free-base form is needed to penetrate the nerve membrane, it is the cationic form that exerts blocking action by binding to the specific receptor site.
PHARMACOKINETICS
The absorption of a local anesthetic depends on its route. When it is injected into the tissues, the rate of absorption depends on the vascularity of the tissues. This is a function of the degree of inflammation present, the vasodilating properties of the local anesthetic agent, the presence of heat, or the use of massage. It is important to reduce the systemic absorption of a local anesthetic when it is used in dentistry. With reduced systemic absorption, the chance of systemic toxicity is reduced. To reduce absorption, a vasoconstrictor is added to the local anesthetic. The vasoconstrictor reduces the blood supply to the area, limits systemic absorption, and reduces systemic toxicity.
With topical application, especially on the mucous membranes or if the surface is denuded, absorption can approximate that produced by intravenous (IV) injection. Absorption is also determined by the proportion of the agent present in the free-base form (nonionized).
Distribution
After absorption, local anesthetics are distributed throughout the body. Highly vascular organs have higher concentrations of anesthetics. Local anesthetics cross the placenta and blood-brain barrier. The lipid solubility of a particular anesthetic affects the potency of the agent. For example, bupivacaine, used as a 0.5% solution, is about 10 times more lipid soluble than lidocaine used as a 2% solution.
Metabolism
The local anesthetic agents are metabolized differently, depending on whether they are amides or esters. Esters are hydrolyzed by plasma pseudocholinesterases and liver esterases. Procaine is hydrolyzed to para-aminobenzoic acid (PABA), a metabolite that may be responsible for its allergic reactions. Some patients who have an atypical form of pseudocholinesterase that does not allow them to hydrolyze these esters may exhibit an increase in systemic toxicity if an ester is administered.
Amide local anesthetics are metabolized primarily by the liver. In severe liver disease or with alcoholism, amides may accumulate and produce systemic toxicity. A small amount of prilocaine is metabolized to orthotoluidine, which can produce methemoglobinemia if given in very large doses. By reducing hepatic blood flow, cimetidine can interfere with the metabolism of the amides. (This is usually unimportant in dentistry because only one dose is given. No accumulation can result if repeated doses are not administered.)
PHARMACOLOGIC EFFECTS
Peripheral Nerve Conduction (Blocker)
The main clinical effect of the local anesthetics is reversible blockage of peripheral nerve conduction. These agents inhibit the movement of the nerve impulse along the fibers, at sensory endings, at myoneural junctions, and at synapses. Therefore they may have wide-reaching effects on many kinds of nerves. Because they do not penetrate the myelin sheath, they affect the myelinated fibers only at the nodes of Ranvier. The local anesthetics affect the small, unmyelinated fibers first and the large, heavily myelinated fibers last. This is probably related to the ability of these agents to penetrate to their site of action.
The losses of nerve function are listed in Box 9-3. This listing is in the order in which the senses are typically lost, but some individual variation occurs among patients. In some patients, the pain sensation is lost before the cold sensation. The functions of the individual nerves return in reverse order.
Antiarrhythmic
Local anesthetics have a direct effect on the cardiac muscle by blocking cardiac sodium channels and depressing abnormal cardiac pacemaker activity, excitability, and conduction. They also depress the strength of cardiac contraction and produce arteriolar dilation, leading to hypotension. These properties make them useful intravenously in the treatment of arrhythmias.
ADVERSE REACTIONS
The adverse reactions and toxicity of the local anesthetics are directly related to the plasma level of drug.
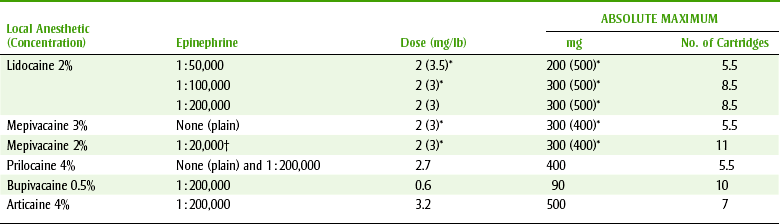
Considering the widespread use of these agents, their potential for danger must be minimal. Deaths from local anesthetics are difficult to document, but dental-related mortality is even rarer. Table 9-2 lists the maximal safe doses for common local anesthetics. Factors that influence toxicity include the following:
TABLE 9-2
MAXIMUM SAFE DOSE* (MSD) OF LOCAL ANESTHETICS
*Manufacturer’s recommendations.
†Vasoconstrictor—levonordefrin.
• Drug: Both the inherent toxicity of the particular local anesthetic and the amount of vasodilation it produces can contribute to toxicity.
• Concentration: The higher the concentration injected, the more drug that enters the systemic circulation.
• Route of administration: Inadvertent IV injection can produce extremely high blood levels. Even topical administration can produce high blood levels and lead to toxicity.
• Rate of injection: The faster the injection is made, the lower the chance that the local area can accept the volume injected. The operator, who has control over this variable, may find that counting the seconds is helpful.
• Vascularity: The presence of inflammation, infection, or vasodilation produced by the agent will increase the vascularity and therefore the systemic toxicity.
• Patient’s weight: The same dose administered to a child and an adult will produce different blood levels because of their differences in weight.
• Rate of metabolism and excretion: Amides may accumulate with liver disease; both amides and their metabolites and ester metabolites may accumulate in renal disease.
Children, elderly individuals, and debilitated persons are more susceptible to the adverse reactions of the local anesthetic agents. The symptoms of an overdose of the local anesthetic agents are directly proportional to the blood level attained.
Toxicity
The two main systems affected by local anesthetic toxicity are the CNS and the cardiovascular system.
 CENTRAL NERVOUS SYSTEM EFFECTS
CENTRAL NERVOUS SYSTEM EFFECTS
CNS stimulation may occur before CNS depression. CNS stimulation caused by depression of the inhibitory fibers results in restlessness, tremors, and convulsions. CNS depression caused by depression of both the inhibitory and facilitative fibers results in respiratory and cardiovascular depression, and coma follows.
 CARDIOVASCULAR EFFECTS
CARDIOVASCULAR EFFECTS
The local anesthetic agents can produce myocardial depression and cardiac arrest with peripheral vasodilation. The usual concentrations that are achieved with administration of dental anesthesia would not be expected to result in any of these adverse reactions, although deaths have been reported with the use of lower doses of anesthetic. It is postulated that the effect of these agents on heart conduction may produce a fatal arrhythmia.
Local Effects
Local effects can occur with the administration of local anesthetic agents. This is most commonly the result of physical injury caused by the injection technique or the administration of an excessive volume too quickly to be accepted by the tissues. Occasionally, a hematoma may be produced.
Malignant Hyperthermia
Malignant hyperthermia is an inherited disease that is transmitted as an autosomal dominant gene with reduced penetration and variable expression. Its symptoms include an acute rise in calcium, which produces muscular rigidity, metabolic acidosis, and extremely high fever. Its mortality rate is about 50%. Treatment of malignant hyperthermia includes supportive measures and the administration of dantrolene (Dantrium). In the past, it was thought that the amide local anesthetics might precipitate malignant hyperthermia, but currently they are no longer implicated. Patients with a family history of malignant hyperthermia can be given amide local anesthetic agents. Halothane, the inhalation anesthetic, and succinylcholine, the neuromuscular blocking agent, are the most common agents precipitating malignant hyperthermia.
Pregnancy and Nursing Considerations
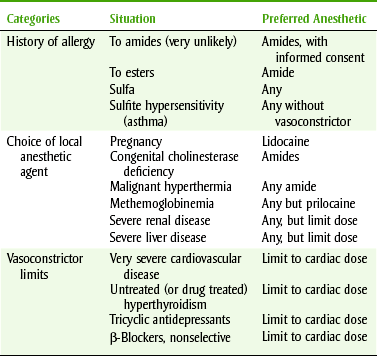
Elective dental treatment should be rendered before a patient becomes pregnant. If dental treatment is needed, however, most sources suggest that lidocaine may be administered to a pregnant woman. Fetal bradycardia has been reported when larger doses are administered to the mother near term. Both lidocaine and prilocaine are in Food and Drug Administration (FDA) pregnancy category B, whereas mepivacaine, articaine, and bupivacaine are category C drugs.
If a local anesthetic is needed, lidocaine in the smallest effective dose should be used. Usual doses of local anesthetics given to nursing mothers will not affect the health of the normal nursing infant.
Allergy
Allergic reactions that result from local anesthetics have been reported, and they range from rash to anaphylactic shock. An allergy history should be elicited from each patient before a local anesthetic agent is chosen. Esters have a much greater allergic potential; in fact, there is some question about whether amides can produce allergic reactions at all. Cross-allergenicity exists between the esters but does not seem to occur between the amides in the xylidine and toluidine groups.
Patients giving a history of allergies to all local anesthetic agents may be “tested” by giving them an amide by injection. Of course, before contemplating administering this test, trained emergency personnel, equipment, and drugs should be assembled. Use of skin testing to determine local anesthetic allergies is unreliable because it can give both false-positive and false-negative results.
Another approach to treating a patient with a history of allergies to all the local anesthetic agents is to use the antihistamine diphenhydramine (Benadryl) as a local anesthetic. Antihistamines, because of their similarity in structure to local anesthetics, have some local anesthetic action. Diphenhydramine (Benadryl) in a concentration of 1% plus 1 : 100,000 epinephrine is recommended to be given by injection to produce a block. There is no prepared product available, so this combination must be prepared from its constituents. Histories of allergic reactions to local anesthetics may have been the result of the preservative methylparaben. It is no longer present in any local anesthetic dental cartridges.
Local anesthetics with vasoconstrictors also contain a sulfite that serves as an antioxidant. In sulfite-sensitive patients, the sulfites may produce a hypersensitivity reaction that exhibits itself as an acute asthmatic attack. This reaction is the same as the “salad bar” syndrome, a hypersensitivity reaction to sulfites. In the past, certain restaurant foods offered at salad bars, such as lettuce, contained sulfites to prevent browning. Sulfites were used to help the lettuce and other greens retain their green color. Some restaurant menus still describe salad bars as “sulfite free.” Deaths of hypersensitive asthmatics who ate in restaurants have been reported. The nature of the reaction involves bronchoconstriction and anaphylactic reactions. A patient with an allergy to “sulfa” drugs does not exhibit cross-hypersensitivity with sulfites. Appendix D discusses the implications of a sulfite hypersensitivity in more detail.
COMPOSITION OF LOCAL ANESTHETIC SOLUTIONS
In addition to the local anesthetic agent, local anesthetic solutions usually contain several other ingredients such as the following:
• Vasoconstrictor: A vasoconstrictor, such as epinephrine, is added to local anesthetic solutions to retard absorption, reduce systemic toxicity, and prolong its duration of action.
• Antioxidant: An antioxidant (sodium metabisulfite, sodium bisulfite, or acetone sodium bisulfite) is included in local anesthetic solutions to retard oxidation of the epinephrine. The antioxidants, such as sodium bisulfite or metabisulfite, prolong shelf life. Asthmatic dental patients who are given local anesthetic agents with a vasoconstrictor, which also contains a sulfite agent, should be watched for symptoms of wheezing or chest tightness.
• Sodium hydroxide: Sodium hydroxide alkalinizes, or adjusts, the pH of the solution to between 6 and 7.
• Sodium chloride: Sodium chloride makes the injectable solution isotonic.
• Methylparaben and propylparaben: Methylparaben and propylparaben are preservatives added to multidose parenteral solutions to prevent bacterial growth. Unlike multidose vials, dental cartridges are single-use containers and do not contain methylparaben. In the past, this preservative was added to dental cartridges. (A question one might ask is, “Why was methylparaben added to a dental cartridge?” Perhaps the manufacturer had a big vat of solution prepared for the multidose vials, which need a preservative, and used the same solution to fill the dental cartridges.) The parabens may be responsible for some allergic reactions attributable to local anesthetic agents reported in the past. No dental cartridge currently contains methylparaben.
LOCAL ANESTHETIC AGENTS
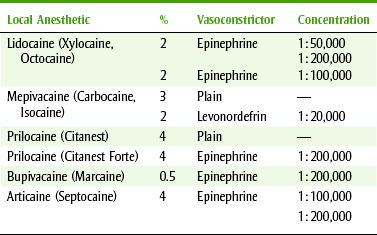
Many local anesthetic agents are available with similar pharmacologic and clinical effects and systemic toxicity. Commonly used local anesthetics are discussed next, and dental issues associated with local anesthetics are listed in Box 9-4. Table 9-3 lists the local anesthetics available in dental cartridges. For clinical applications, lidocaine with epinephrine 1 : 100,000 is the usual choice. The question to be answered is, “Under what conditions would a local anesthetic other than lidocaine with epinephrine 1 : 100,000 be indicated?”
Amides
The amide local anesthetic agents are the only class of anesthetics used parenterally. Esters are occasionally used topically. The relative lack of allergenicity of the amides is probably responsible for this use.
 LIDOCAINE
LIDOCAINE
An amide derivative of xylidine introduced in 1948, lidocaine (LYE-doe-kane) (Xylocaine, Octocaine) quickly became an anesthetic standard to which other local anesthetics were compared. It has a rapid onset, which is related to its tendency to spread well through the tissues. Lidocaine 2% with vasoconstrictor provides profound anesthesia of medium duration. It is the local anesthetic solution most commonly used in dental offices.
No cross-allergenicity between the amide lidocaine, other available amides, or esters has been documented. Some patients appear to experience some sedation with lidocaine, and in toxic reactions one is likely to observe CNS depression initially rather than the CNS stimulation characteristic of other local anesthetics (Figure 9-5).
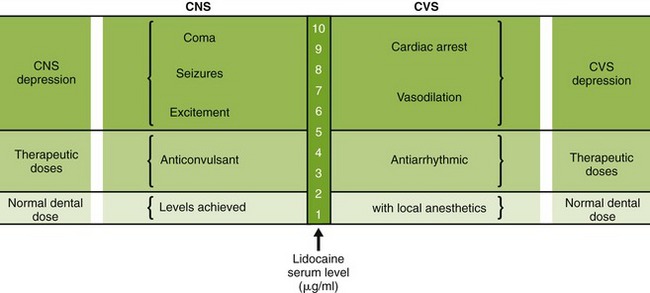
FIGURE 9-5 Relationship between levels of local anesthesia in serum and the pharmacologic and adverse effects. CNS, Central nervous system; CVS, cardiovascular system.
Adverse reactions include hypotension, positional headache, and shivering. Lidocaine is used for topical, infiltration, block, spinal, epidural, and caudal anesthesia. It is also used intravenously to treat cardiac arrhythmias during surgery.
In dentistry, lidocaine 2% with 1 : 100,000 epinephrine is used for infiltration and block anesthesia. Lidocaine is used for topical anesthesia as a 5% ointment, a 10% spray, and a 2% viscous solution. When used topically, its onset is rapid (2 to 3 minutes). Lidocaine with epinephrine 1 : 100,000 provides a 1- to 1.5-hour duration of pulpal anesthesia. Soft tissue anesthesia is maintained for 3 to 4 hours. Lidocaine with epinephrine 1 : 50,000 is used for hemostasis during surgical procedures. Rebound vasodilation (β effect) can be expected after the α effect (vasoconstriction) has occurred. A new dose form of lidocaine is a patch that is applied to the mucosal membranes for local anesthesia. It provides good anesthesia, but its maximal effect occurs after about 10 minutes, which is probably too long to wait.
 MEPIVACAINE
MEPIVACAINE
Another amide derivative of xylidine is mepivacaine (me-PIV-a-kane) (Carbocaine, Polocaine, or Isocaine). Introduced in 1960, its rate of onset, duration, potency, and toxicity are similar to those of lidocaine. No cross-allergenicity between the amide mepivacaine, other currently available amides, or the esters has been documented.
Mepivacaine is not effective topically; however, it is used for infiltration, block, spinal, epidural, and caudal anesthesia. The usual dose form in dentistry is a 2% solution with the addition of 1 : 20,000 levonordefrin (Neo-Cobefrin) as the vasoconstrictor. Because mepivacaine produces less vasodilation than lidocaine, it can be used as a 3% solution without a vasoconstrictor (called plain). It can be used for short procedures when a vasoconstrictor is contraindicated (not often). Caution should be exercised when using the increased concentrations of the local anesthetic without a vasoconstrictor because systemic toxicity is more likely. Except in unusual cases, the benefit of a shorter duration does not warrant eliminating the vasoconstrictor, especially when the concentration of the drug is increased.
 PRILOCAINE
PRILOCAINE
Prilocaine (PRILL-loh-kane) (Citanest, Citanest Forte) is related chemically and pharmacologically to both lidocaine and mepivacaine. Chemically, lidocaine and mepivacaine are xylidine derivatives, whereas prilocaine is a toluidine derivative. Prilocaine appears to be less potent and less toxic than lidocaine and has a slightly longer duration of action. It has been shown to produce satisfactory local anesthesia with low concentrations of epinephrine and without epinephrine.
Although toxicity of prilocaine is 60% of that occurring with lidocaine, several cases of methemoglobinemia have been reported after its use. Prilocaine is metabolized to ortho-toluidine and in large doses, can induce some methemoglobinemia. A very large dose (greater than the maximal safe dose) would be required to produce clinical symptoms: cyanosis of the lips and mucous membranes and occasionally respiratory or circulatory distress. Although the small doses required in dental practice are not likely to present a problem in healthy, nonpregnant adults, prilocaine should not be administered to patients with any condition in which problems of oxygenation may be especially critical. Drugs that affect the hemoglobin, such as acetaminophen, may exacerbate the adverse reaction. Methemoglobinemia can be reversed by IV methylene blue.
Prilocaine is used for infiltration, block, epidural, and caudal anesthesia. It is available in dental cartridges as a 4% concentration both with and without 1 : 200,000 epinephrine.
Prilocaine’s niche in dentistry involves situations in which the desired duration of action is somewhat longer than that obtained with mepivacaine (without and with). Prilocaine plain has a duration of action slightly longer than mepivacaine plain, and prilocaine with epinephrine has a duration of action slightly longer than lidocaine with epinephrine. The other potential advantage of prilocaine is that the concentration of epinephrine (1 : 200,000) is lower than in other local anesthetic amide combinations. Therefore, if prilocaine with epinephrine were to be used, the patient would be exposed to half of the amount of epinephrine as with lidocaine with epinephrine 1 : 100,000.
 BUPIVACAINE
BUPIVACAINE
Bupivacaine (byoo-PIV-a-kane) (Marcaine) is an amide type of local anesthetic related to lidocaine and mepivacaine. It is more potent but less toxic than the other amides. The major advantage of bupivacaine is its greatly prolonged duration of action. It is indicated in lengthy dental procedures when pulpal anesthesia of greater than 1.5 hours is needed or when postoperative pain is expected (e.g., endodontics, periodontics, or oral surgery). After sensation begins to return, a period of reduced or altered sensation (analgesia) may last several hours. Compared with lidocaine with epinephrine, the onset of bupivacaine with epinephrine is slightly longer, but its duration is at least twice that of lidocaine. It is available in dental cartridges as a 0.5% solution with 1 : 200,000 epinephrine. It should not be used in patients prone to self-mutilation (mental patients or children younger than 12 years). During its early use in anesthesiology and obstetrics, fatal unresuscitable cardiac arrests occurred. The doses used for obstetrics were much higher than those used in dentistry. After the maximal doses for obstetrics were lowered, these cardiac arrests essentially disappeared. Because much lower maximal doses are recommended for dental procedures, these adverse reactions are very unlikely to occur in dental practice. Bupivacaine has been used for infiltration, block, and peridural anesthesia.
 ARTICAINE
ARTICAINE
Articaine (Septocaine) was approved for use in the United States in 2000 and has been used in Europe since the mid-1970s. Its delay in the United States was a result of the addition of methylparaben to both multidose vials and single-dose cartridges. In the late 1990s, Septodont, a Canadian pharmaceutical company, submitted a methylparaben-free formulation for approval in the United States in 1995 and it was finally approved in 2000.
Articaine differs from other amide local anesthetics because it is derived from thiophene. This allows for greater lipid solubility and ability to cross lipid barriers such as nerve membranes. It has been suggested that this mechanism may account for its enhanced action compared with other local anesthetics. Articaine also differs from other amide local anesthetics because it has an extra ester linkage. This extra linkage causes articaine to be hydrolyzed by plasma esterase. Only 5% to 10% of articaine is metabolized by the liver, the other 90% to 95% is metabolized in the blood. Its major metabolite is articainic acid and it is unclear how active this metabolite is.
Articaine is excreted by the kidneys, 40% to 70% as articainic acid, 2% to 5% unchanged, and 4% to 15% as articainic acid glucuronide, which also appears to be inactive. The half-life of articaine is approximately 20 minutes compared with lidocaine, which is approximately 90 minutes. Articaine’s shorter half-life is the result of its metabolism by plasma esterases. Other amides are metabolized by the liver and have much longer half-lives. Because of its rapid metabolism, articaine may be a safer drug to reinject later on during a dental visit. This would be especially true if all of articaine’s metabolites were inactive.
Despite its short half-life and apparent safety, articaine is a 4% solution with a toxic dose of 7 mg/kg for the average healthy adult. Because lidocaine is only a 2% solution with the same maximal dose, the average patient can tolerate twice as much lidocaine as compared with articaine before the maximal dose is reached. In addition, articaine, like prilocaine, in very high doses may cause methemoglobinemia. It should be noted that no reported cases of methemoglobinemia have been reported with articaine in doses recommended for dental local anesthesia. Lastly, articaine rarely causes paresthesia after a mandibular block when the 4% solution is used.
Articaine is used for local, infiltrative, and conductive anesthesia. It is available as a 4% concentration with 1 : 100,000 epinephrine in a 1.7 ml dental cartridge unlike the more common 1.8 ml dental cartridge. It has become the most widely used local anesthetic in just about every country in which it has been introduced. Its relative lack of significant active metabolites makes it more desirable in patients that may need to be reinjected. This lowers the risk for toxicity. Although clinical trials have not shown that articaine is better than available local anesthetics, many of these clinical trials show that articaine slightly outperformed the local anesthetics with which it was compared.
Esters
There are currently no esters available in a dental cartridge. Esters, such as benzocaine, are commonly used topically.
 PROCAINE
PROCAINE
Procaine (PROE-kane) (Novocain) is a PABA ester. Procaine is used as an antiarrhythmic agent (procainamide) and is combined with penicillin to form procaine penicillin G. Procaine is not used in dentistry today because of the high rate of allergic reaction. The allergic reaction is usually a result of PABA and not procaine.
 PROPOXYCAINE
PROPOXYCAINE
Propoxycaine (proe-POX-i-kane) (Ravocaine), another ester of PABA, is not available in a dental cartridge.
 TETRACAINE
TETRACAINE
Tetracaine (TET-ra-kane) (Pontocaine), an ester of PABA, has a slow onset and long duration and is generally estimated to have at least 10 times the potency and toxicity of procaine. In view of this drug’s high toxicity and the rapidity with which it is absorbed from mucosal surfaces, great care must be exercised if it is used for topical anesthesia. Dermatologic reactions include contact dermatitis, burning, stinging, and angioedema. A maximal dose of 20 mg is recommended for topical administration. Tetracaine is available in various sprays, solutions, and ointments for topical application. The concentration of tetracaine in most topical preparations is 2%.
Other Local Anesthetics
Dyclonine (DYE-kloe-neen) (Dyclone) is a topical local anesthetic that is neither an ester nor an amide. Its side effects involving the cardiovascular system and CNS are similar to those of the other local anesthetics. Dyclonine may produce slight irritation and stinging when applied. Patients can exhibit allergic reactions to dyclonine, but cross-allergenicity with other local anesthetics would not be expected because of its unique structure. The onset of local anesthesia is 2 to 10 minutes, and its duration is 30 to 60 minutes. The solution and topical product are available as 0.5% and 1% concentrations.
 BENZONATATE
BENZONATATE
Benzonatate (ben-ZOE-na-tate) (Tessalon Perles) is a tetracaine congener (a near relative) indicated in the management of nonproductive cough. It is a topical anesthetic that acts on the respiratory stretch receptors, which produces its antitussive properties. Because the drug’s local anesthetic activity can reduce the patient’s gag reflex, care should be taken when working within the mouth to prevent foreign particles from entering the throat. Side effects include sedation, headache, dizziness, rash, gastrointestinal upset, and nasal congestion. It is used to treat cough.
VASOCONSTRICTORS
The vasoconstricting agents are included in local anesthetic solutions for many reasons (Box 9-5).
The vasoconstrictors are members of the autonomic nervous system drugs called the adrenergic agonists or sympathomimetics (see Chapter 4).
When a local anesthetic solution does not contain a vasoconstrictor, the anesthetic drug is more quickly removed from the injection site and distributed into systemic circulation than if the solution contained a vasoconstrictor. Plain (without vasoconstrictor) anesthetics will exhibit a shorter duration of action and result in a more rapid buildup of a systemic blood level. Therefore any anesthetic given without a vasoconstrictor is more likely to be toxic than those given with a vasoconstrictor. Any advantage gained by eliminating the vasoconstrictor (shorter duration and increased possible systemic effect of the vasoconstrictor) must be weighed against the potential for adverse effects from the epinephrine.
The decision about whether epinephrine should be used in a patient is made by weighing the risks and the benefits. Figure 9-6 shows the amount of epinephrine at rest and during mild-to-severe stress, and Figure 9-7 compares the dose for anaphylaxis and dental use.
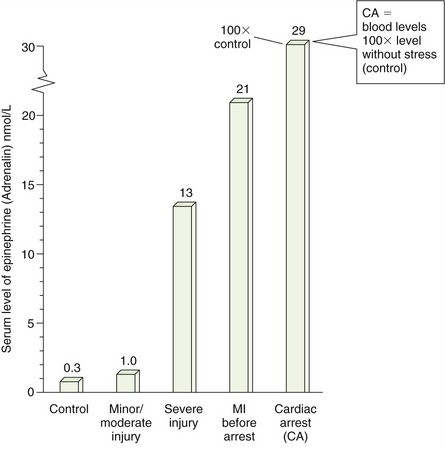
FIGURE 9-6 Blood levels of endogenous epinephrine during rest; with minor, moderate, or severe injury; myocardial infarction (MI; before arrest); and cardiac arrest (CA).
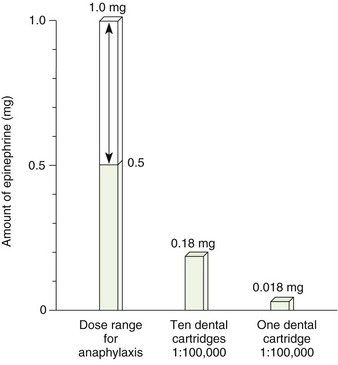
FIGURE 9-7 Histogram showing the dose range of epinephrine for anaphylaxis and the doses provided in dental cartridges.
A sufficient concentration must be used to keep the local anesthetic localized at its site of action and provide adequate depth, duration, and low systemic toxicity of the anesthetic. It has been shown that 1 : 100,000 and 1 : 200,000 produce about the same amount of vasoconstriction and the same distribution of the local anesthetics. No justification exists for the use of epinephrine in a concentration greater than 1 : 200,000, except in cases in which local hemostasis is needed (1 : 50,000 is used). Lidocaine is available with 1 : 100,000 epinephrine, although the weaker concentration has been shown to produce similar results.
In the 1940s, the literature stated that dental local anesthetics containing vasoconstrictors should not be used in patients with cardiovascular disease. This recommendation stemmed from the fear that the vasoconstrictor would elevate the blood pressure too much. It is now known that a patient can produce endogenous epinephrine far in excess of that administered in dentistry in the presence of inadequate anesthesia, which sometimes occurs when vasoconstrictors are avoided. Medical consults often recommend that epinephrine be avoided because physicians are more familiar with the doses used in medicine (0.5 to 1.0 mg) rather than the dental dose (0.018 mg/cartridge [1.8 ml] of 1 : 100,000) of epinephrine (see Figure 9-7).
Patients with uncontrolled high blood pressure, hyperthyroidism, angina pectoris, and cardiac arrhythmias and those who have had a myocardial infarction or cerebrovascular accident in the past 6 months should make an appointment for elective dental treatment after their medical condition is under control. For patients who have had a myocardial infarction or cerebrovascular accident, that would be 6 months after the cardiovascular or cerebrovascular event.. Those undergoing general anesthesia with a halogenated hydrocarbon inhalation anesthetic should be monitored for arrhythmias if epinephrine (including epinephrine-soaked retraction cords) is used for its hemostatic effect (used commonly with halothane). If arrhythmias occur, antiarrhythmic agents are administered.
Patients with cardiovascular disease who are able to withstand elective dental treatment can receive epinephrine-containing local anesthetic agents. The anesthetic should be administered in the lowest possible dose by means of the best technique, including aspiration and a very slow injection rate to minimize systemic absorption. Maximal cardiac doses should not be exceeded in patients with severe cardiovascular disease. Table 9-4 lists the maximal safe dose of epinephrine for the healthy patient (0.2 mg) and the cardiac patient (0.04 mg); the number of cartridges each of these doses represents is included. For example, the cardiac patient could be given two cartridges of 1 : 100,000 epinephrine without exceeding the cardiac dose.
TABLE 9-4
VASOCONSTRICTORS: MAXIMUM SAFE DOSE (MSD) IN NORMAL AND CARDIAC PATIENTS
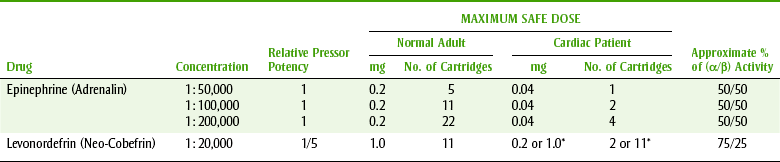
*Data from Malamed SF: Handbook of local anesthesia, ed 5, St Louis, 2004, Mosby.
Drug Interactions
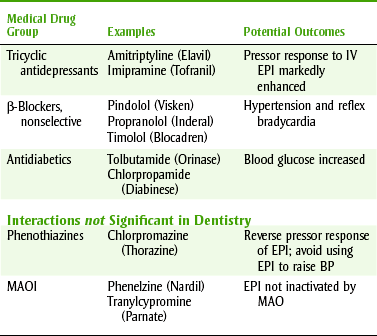
Selected drug interactions of epinephrine are listed in Table 9-5. Of the most important drug interactions with epinephrine, two are clinically significant and two are not. The two epinephrine drug interactions that are most likely to be clinically significant are those with tricyclic antidepressants and nonselective β-blockers. With tricyclic antidepressants, administration of epinephrine may produce an exaggerated increase in pressor response (increased blood pressure). With the nonselective β-blockers, hypertension and reflex bradycardia may be exhibited. These are not absolute contraindications to the use of epinephrine, but patients taking these agents should be monitored for symptoms of alterations in their blood pressure. The two drug interactions that are commonly mentioned but are not usually clinically significant are with monoamine oxidase inhibitors (MAOIs) and phenothiazines. Epinephrine can be given to patients taking MAOIs because epinephrine is eliminated primarily by reuptake and secondarily by catechol O-methyltransferase (COMT) rather than by monoamine oxidase (MAO). If any small interaction exists, it would be the result of “denervation hypersensitivity.” In contrast to epinephrine, the indirect-acting sympathomimetic agents (e.g., pseudoephedrine) should be avoided in patients taking MAOIs because they are inactivated in significant amounts by MAO. The drug interaction between epinephrine and phenothiazines occurs because the phenothiazines are α-blockers, and when an α and β agonist (epinephrine) is given, the β effects (vasodilation) predominate. Therefore, if epinephrine is used for its vasopressor effect (to raise the blood pressure), the blood pressure is likely to decrease. When epinephrine is used in a local anesthetic solution, it is not being given for its vasopressor effect, so this interaction is not clinically significant.
CHOICE OF LOCAL ANESTHETIC
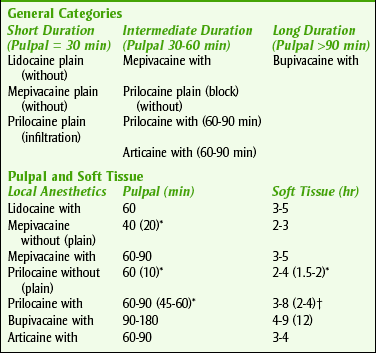
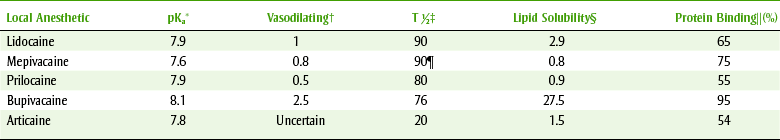
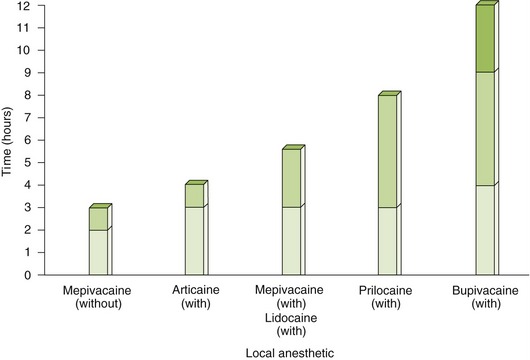
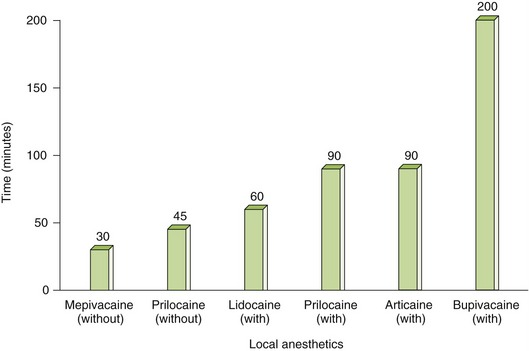
Practitioners should choose a few local anesthetic solutions to use, depending on the duration of local anesthesia desired and the side effects that must be avoided. Box 9-6 lists the local anesthetics by their durations of action, including both pulpal and soft tissue anesthesia. Figures 9-8 and 9-9 illustrate the durations of action of local anesthetic agents for soft tissue and pulpal anesthetics, respectively. Several properties of local anesthetic agents determine their differences in pharmacokinetics. Table 9-6 lists these physical properties for some local anesthetics. For example, the pKa is related to the onset of action. With a lower pKa, the local anesthetic is distributed more in the base form and so is better absorbed. The duration of action of the local anesthetic is primarily related to its protein-binding capacity. Its lipid solubility may also play some part. The duration is unrelated to the local anesthetic’s half-life because its action is terminated when the drug is removed from the receptor. The lipid solubility determines the potency of a local anesthetic agent. The vasodilating property of a local anesthetic can affect both the potency and duration of action. One should note that the vasodilating effect of lidocaine (1) is more than that of mepivacaine (0.8) and prilocaine (0.5). Because mepivacaine and prilocaine have less vasodilating effect, they can be used without vasoconstrictor. In contrast, lidocaine (1) and bupivacaine (2.5) produce too much vasodilation to be used without a vasoconstrictor. The dental practitioner should become familiar with a short-, an intermediate-, and a long-acting agent. The duration of the procedure and any patient-specific information will determine the anesthetic of choice. Table 9-7 lists some common contraindications to the use of local anesthetic agents.
TOPICAL ANESTHETICS
Benzocaine, an ester, is the most commonly used topical anesthetic; lidocaine, an amide, is the second most commonly used. Some topical anesthetics are listed in Table 9-8. Comparison among the agents should take into account their onset, duration of action, and allergenic potential. The patient should be instructed to avoid eating for 1 hour after application to oral mucosa so that the gag reflex can become fully functional.
Amides
Lidocaine (Xylocaine) is available as the base or hydrochloride salt. The base is preferred when large areas of the mucosal surfaces are ulcerated, abraded, denuded, or erythematous. The hydrochloride salt is water soluble and penetrates the tissue better. Therefore its propensity for systemic absorption is greater than with the base. Lidocaine base is available as a jelly and an oral topical solution, and hydrochloride is available as an ointment, an oral topical, and an oral aerosol. Concentration of the creams ranges from 2% to 5%. Viscous lidocaine (2%) is available for oral rinse to manage aphthous lesions or reduce gagging.
 LIDOCAINE AND PRILOCAINE (INJECTION-FREE LOCAL ANESTHESIA)
LIDOCAINE AND PRILOCAINE (INJECTION-FREE LOCAL ANESTHESIA)
More often than not, the fear of injection prevents many people from seeking necessary dental treatment. Either the thought of the injection or the injection itself can be painful and upsetting. The combination of lidocaine and prilocaine gel (Oraqix) applied into the periodontal pocket offers pain relief during scaling and root planing procedures. The combination of lidocaine and prilocaine, in the gel form, provides a duration of action of approximately 20 minutes. Its onset of action is approximately 30 seconds after application. Lidocaine provides rapid anesthesia, and prilocaine has a slower onset of action. The combination appears to be well tolerated. The more common side effects include pain, soreness, irritation, edema or redness at the area of application, and taste changes.
Esters
Benzocaine (BEN-zoe-kane) (Hurricaine, Anbesol, Benzodent, or Orabase-B), an ester of PABA, cannot be converted to a water-soluble form for injection. Because it is poorly soluble, it is poorly absorbed and lacks significant systemic toxicity. Local reactions reported include burning and stinging. Dermatologic reactions have included angioedema and contact dermatitis, which can occur if the operator does not wear gloves (an unacceptable practice today). Benzocaine is available in dental products and in many over-the-counter (OTC) products for teething, sunburn, hemorrhoids, or insect bites (up to 20% for many but not all). Benzocaine is used in many dental offices, although a hypersensitivity reaction is possible.
 COCAINE
COCAINE
Cocaine (koe-KANE) is a naturally occurring ester of benzoic acid that is potent and extremely toxic. Its onset of action is less than 1 minute, and its peak is within 5 minutes. Its duration of action is about 30 minutes. Although cocaine has ideal pharmacokinetics, the systemic absorption and subsequent CNS stimulation and its great potential for abuse make the use of cocaine as a local anesthetic untenable. Its CNS effects are discussed in Chapter 25. It has no dental application.
Precautions in Topical Anesthesia
Some local anesthetics are absorbed rapidly when applied topically to mucous membranes. To avoid toxic reactions from surface anesthesia, the dental health care provider should consider many factors (Box 9-7).
DOSES OF LOCAL ANESTHETIC AND VASOCONSTRICTOR
The amounts of local anesthetic and vasoconstrictor contained in a certain volume of solution can be calculated from the concentration of that solution. The local anesthetic percent, for example, 2%, may be expressed as seen in Box 9-8. Box 9-9 shows how to calculate the amount of epinephrine.
The dental health care provider should be able to determine the number of milligrams of both local anesthetic and vasoconstrictor given in any clinical situation. The maximal safe dose for each component should not be exceeded.
Each dose should be recorded in the patient’s chart as soon as possible after the injection. The information placed in the chart should include the strength of both ingredients and the volume of solution used or the number of milligrams of each given. For example, if a patient were given one cartridge of lidocaine 2% with 1 : 100,000 epinephrine, the chart would read: lidocaine 2% with epinephrine 1 : 100,000 1.8 ml, or lidocaine 36 mg with epinephrine 0.018 mg.
One reason for including this information in the chart is to minimize questions that might arise later if the patient or a future practitioner has concerns about the treatment. Because of the increasing incidence of lawsuits against dentists and hygienists, maintaining a complete chart to prevent any ambiguity is extremely important.
CLINICAL SKILLS ASSESSMENT
1. Name the properties of the ideal local anesthetic.
2. Differentiate between the two major chemical groups of local anesthetic agents.
3. Contrast the allergenicity and metabolism of the ester and amide local anesthetics.
4. List the systemic adverse reactions to the local anesthetics.
5. List five injectable local anesthetic agents and give their composition.
6. Explain the presence in a dental cartridge of agents other than the local anesthetic.
7. State the rationale for the inclusion of vasoconstricting agents in local anesthetic solution.
8. State the maximal safe dose of the two vasoconstrictors used in dentistry for both the normal patient and the cardiac patient.
9. Explain the rationale for use of Oraqix in a dental practice.
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
*pH at which half is in each form (salt and base equal).