Vitamins and Minerals
1 Explain the body’s need for vitamins and minerals.
2 Summarize and explain how vitamins are classified.
3 Name and describe the water- and fat-soluble vitamins—sources, recommended amounts, roles, deficiencies, adverse reactions, and clinical considerations.
4 Describe the sources, recommended amounts, roles, deficiencies, and toxicity of the minerals iron, zinc, and calcium.
5 Discuss the clinical considerations of vitamins and minerals and provide several examples of their relevance to dental treatment.
The vitamins (VYE-ta-mins), which are essential in small quantities for the maintenance of cell structure and metabolism, are a group of low-molecular-weight compounds. In normal quantities, a vitamin is used to replace that vitamin which is deficient. Vitamins are also used to treat problems not associated with vitamin deficiency. When used as such, they are regarded as drugs. However, few situations exist for which there is proof that vitamins are useful for the treatment of any condition except vitamin deficiency.
Vitamins are classified into two large groups: water soluble and fat soluble. The water-soluble vitamins include the B vitamins and vitamin C. The fat-soluble vitamins are vitamins A, D, E, and K. Vitamins act in three different ways: as coenzymes, antioxidants, or hormones. The water-soluble vitamins act as coenzymes, acting with a specific enzyme that catalyzes a specific reaction. Vitamins C and E act as antioxidants, and vitamin A and D act as hormones. Table 12-1 lists the common names of the vitamins and their deficiencies.
TABLE 12-1
VITAMINS AND THEIR DEFICIENCIES
FAD, Flavin adenine dinucleotide; FMN, flavin mononucleotide; NAD, nicotinamide adenine dinucleotide; NADP, NAD phosphate.
*Called vitamin D3.
†Form found in vitamin pills, vitamin D2, product of sun on skin.
MEASUREMENTS OF VITAMIN NEEDS
Since the early 1940s, the Food and Nutrition Board of the National Academy of Sciences has been reviewing research to determine dietary recommendations. The original recommended daily allowance (RDA) was designed with the goal of preventing the diseases produced by the deficiency of a certain nutrient. These values were meant to be used to make recommendations for populations (e.g., school lunches or nursing homes) rather than specific people. These RDAs are not synonymous with an individual’s requirement. Individual requirements are influenced by many factors, including but not limited to physical characteristics, dietary habits, sex, pregnancy, lactation, and age. However, the RDAs are set high enough to allow for many variations in patient needs. During the subsequent years, because no other values were available, the RDAs began to be used (inappropriately) to address specific patient needs.
Since the discovery of vitamins, dietitians have been searching the literature for evidence for the vitamin needs of individuals and populations. After the release of the last published RDAs in 1989, a discussion of inappropriate use of the former RDAs was begun. Agreement was reached that the use of this one number did not cover all the needs for nutrition information. In 1993 the Food and Nutrition Board initiated a review process, beginning with a symposium. This process of reviewing the literature to determine the appropriate recommendations reflecting the current research began and continues today. The new RDAs are designed not only to prevent deficiency diseases but also to minimize chronic diseases such as heart disease.
The original term, RDAs (old), that was used as reference for years has been divided into different, more specific recommendations. The definitions of the five newer terms appear in Box 12-1. Dietary reference intakes (DRIs) are used to develop diets for healthy people. The DRIs for individual vitamins and minerals began to be compiled and released. In 1997, the DRIs for vitamin D, magnesium, and fluoride were completed, and in 1998, the B vitamins were addressed and recommendations released. Currently, the recommendations available are divided between the RDAs, and if that is unknown, the adequate intake (AI). In general, the AI for a specific agent is less than the RDA because the RDA allows for some variation.
WATER-SOLUBLE VITAMINS
Ascorbic (a-SKOR-bik) acid, or vitamin C, chemically is a sugar acid that readily undergoes oxidation to form dehydroascorbic acid. Because of this ability, ascorbic acid is an effective reducing agent. The active isomer is l-ascorbic acid.
 SOURCE
SOURCE
Good natural sources of ascorbic acid include citrus fruits, green peppers, tomatoes, strawberries, broccoli, raw cabbage, baked potatoes, and papaya (Figure 12-1). Some food products are fortified with vitamin C. Ascorbic acid is absorbed in the ileum by a Na+-dependent carrier-mediated mechanism.

FIGURE 12-1 Good source of vitamin C includes citrus fruits such as oranges, green peppers, broccoli, and strawberries. (Copyright 2009 Jupiterimages Corporation.)
Because of its ability to be easily oxidized, ascorbic acid is readily destroyed through cooking, and as much as 50% of the ascorbic acid content of foods can be lost in this manner.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The RDA of ascorbic acid for a healthy female adult is 75 mg and 90 mg for a healthy male adult. During pregnancy and lactation, stress, or tobacco smoking, the need for this vitamin increases.
 ROLE
ROLE
The metabolic role of ascorbic acid is probably related to the fact that ascorbic acid and dehydroascorbic acid form a readily reversible oxidation-reduction system. It is thought that this vitamin plays a role in biologic oxidations and reductions in cellular respirations. Ascorbic acid also plays a definite role in connective tissue metabolism because it is required for the formation of collagen. The function of ascorbic acid can be dramatically demonstrated in the wound-healing process. Scorbutic wounds have a decrease in mature collagen fibrils associated with an accumulation of mucopolysaccharides or ground substance around a matrix of precollagenous fibers. The absence of mature collagen results in abnormal healing that reduces the tensile strength of the wound.
 DEFICIENCY
DEFICIENCY
The deficiency of ascorbic acid produces a condition termed scurvy. The manifestations of scurvy occur because of the inability of the connective tissue to produce and maintain intercellular substances such as collagen, bone matrix, dentin, cartilage, and vascular endothelium.
The functions of vitamin C include the following:
• Synthesis of epinephrine and norepinephrine
• Synthesis of carnitine, a protein that facilitates transport of fatty acids into mitochondria for β oxidation
The following are manifestations of defective connective tissue formation in vitamin C deficiency:
• Impaired wound healing resulting from a lack of collagen
• Inadequate response to infections
• Alterations in the integrity of capillary walls, manifested as hemorrhages in skin, mucous membranes, muscles, lungs, joints, and gingivae (spongy, edematous, inflamed)
• Lack of formation of bone matrix, resulting in disorganization of epiphyseal line, weakening of bones, pathologic fractures, and resorption of alveolar bone with loosening and loss of teeth
Because humans and other primates cannot synthesize vitamin C, they must obtain it daily from their diet.
Diets completely deficient in vitamin C are unusual, and there are few cases of serious vitamin C deficiency (scurvy). After a prolonged period (4 to 5 months) without vitamin C, humans have symptoms of weakness, anorexia, suppressed growth, anemia, lower resistance to infection and fever, swollen and inflamed gums, loosened teeth, swollen wrists and ankle joints, petechial hemorrhages, fracture of ribs at costochondral junctions, and hemorrhaging resulting from capillary fragility in joints, muscle, and intestines.
 ADVERSE REACTIONS
ADVERSE REACTIONS
Untoward effects have been reported with the use of megadoses of vitamin C. A daily intake of 1 gm of vitamin C may cause precipitation of oxalate stones in the urinary tract. For this reason, unwarranted use of large quantities of vitamin C is discouraged. A rebound scurvy has been reported in adults and infants who received megadoses that were then stopped abruptly.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
As long ago as 1942, the suggestion was made that vitamin C could be therapeutically beneficial in preventing the common cold. Linus Pauling reviewed the available data and indicated that vitamin C has a substantial beneficial effect in preventing and treating the common cold. Other investigators reviewed the data and concluded that little if any evidence existed to suggest the effectiveness of vitamin C in either preventing or treating the common cold. Based on current evidence, unrestricted use of ascorbic acid for these purposes cannot be advocated.
Another Pauling hypothesis suggested that large quantities of vitamin C may suppress neoplastic cellular proliferation. He indicated that vitamin C should be used in the management of all types of cancer. Other investigators have been unable to verify his claim.
Because vitamin C enhances the absorption of iron, iron is either combined with vitamin C or taken with orange juice to treat iron deficiency anemia.
B-Complex Vitamins
The water-soluble vitamins, except for vitamin C, are known as the B-complex vitamins. On a functional basis, these vitamins may be subdivided into the following three classes:
1. Those that primarily release energy from carbohydrates and fats (thiamine, pyridoxine, niacin, riboflavin, pantothenic acid, and biotin)
2. Those that among other functions catalyze the formation of red cells (folic acid, vitamin B12)
3. Those that have not been shown to be required in human nutrition (choline and inositol)
A close interrelationship among the B-complex vitamins exists. If a deficiency of one of them occurs, it will impair the utilization of others. Also, the signs and symptoms of a deficiency of individual B vitamins are similar. This is probably because a deficiency of a single member of the B complex seldom occurs. A diet deficient in one B vitamin is usually lacking in other B vitamins. Figure 12-2 shows some examples of good sources of B-complex vitamins.
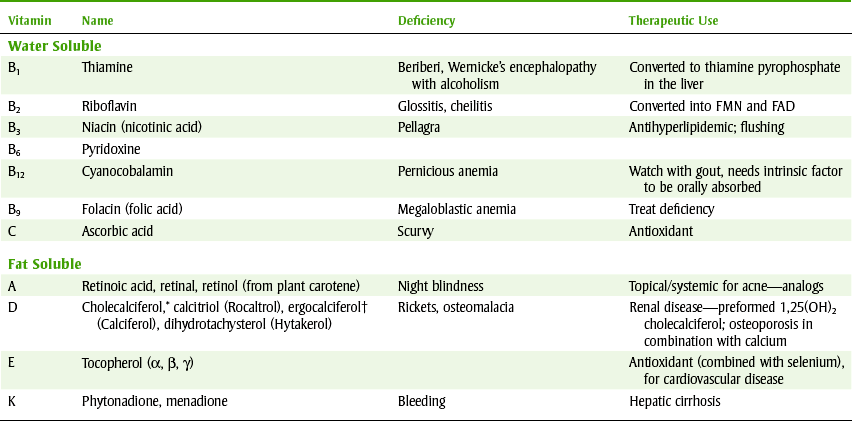
FIGURE 12-2 Examples of foods containing various B-complex vitamins. A, Whole-grain and enriched breads provide vitamins B1, B3, and B6, folic acid, pantothenic acid, and biotin. B, Peas and other legumes are good sources of thiamine (vitamin B1), niacin (vitamin B3), and pyridoxine (vitamin B6). Riboflavin (vitamin B2) is present in leafy green vegetables. C, Meat is a rich source of several vitamins, including riboflavin, niacin, pyridoxine, folic acid, cyanocobalamin, pantothenic acid, and biotin, whereas fish provides niacin (D). E, Eggs contain riboflavin, pyridoxine, cyanocobalamin, pantothenic acid, and biotin. F, Riboflavin, cyanocobalamin, and biotin are found in dairy products such as milk and cheese. (Copyright 2009 Jupiterimages Corporation.)
 THIAMINE (VITAMIN B1)
THIAMINE (VITAMIN B1)
Thiamine (THYE-a-min) (vitamin B1) is an essential water-soluble vitamin in humans. It is converted in the liver to its active coenzyme form, thiamine pyrophosphate (TPP).
Source: Thiamine is present in foods of both animal and vegetable origin. The best sources are pork, whole grain and enriched breads, cereals and pastas, seeds of legumes such as peas, dried brewer’s yeast, and wheat germ (see Figure 12-2). The vitamin tends to be destroyed if heated to about 100° C; therefore significant amounts of this vitamin may be lost if foods are cooked too long above this temperature.
Recommended Dietary Allowance: The RDA for thiamine is 1.2 mg for adult men (ages 14 to >70 years) and 1.1 mg for adult women (ages 19 to >70 years). Table 12-2 lists RDAs for other groups. Thiamine requirements parallel the caloric or carbohydrate content of the diet.
TABLE 12-2 DIETARY REFERENCE INTAKES: RECOMMENDED DIETARY ALLOWANCES AND ADEQUATE INTAKES OF SELECTED VITAMINS AND ELEMENTS Rights were not granted to include this table in electronic media. Please refer to the printed book. From Otten JJ, Hellwig JP, Meyers LD, eds: Dietary reference intakes: the essential guide to nutrient requirements, Washington, DC, 2006, National Academics Press. National Academics of Sciences
Role: TPP plays a principal role in intermediary metabolism. It is a coenzyme required for the oxidative decarboxylation of α-ketoacids. In this role, TPP is sometimes referred to as cocarboxylase.
Deficiency: The severe deficiency of thiamine leads to a condition known as beriberi. Characteristics of beriberi are peripheral neuritis, muscle weakness, paralysis of the limbs, enlargement of the heart, tachycardia, and edema (typical of wet beriberi). Gastrointestinal tract effects include loss of appetite and intestinal atony, and constipation may also be present. The symptoms of mild thiamine deficiency are less characteristic. They include fatigue and apathy, loss of appetite, moodiness and irritability, pain and paresthesias in the extremities, slight edema, decreased blood pressure, and lowered body temperature.
Possible oral manifestations of thiamine deficiency are burning tongue, ageusia (loss of taste functions of the tongue), and hyperesthesia of the oral mucosa.
The most common cause of thiamine deficiency in the United States is alcoholism. Both poor appetite and the effect of alcohol on the nerves may exacerbate this problem. Both Wernicke’s encephalopathy and Korsakoff’s psychosis can result if a severe deficiency exists.
Adverse Reactions: Thiamine is usually nontoxic, even in large parenteral doses. However, in patients who are hypersensitive to thiamine, pruritus, sweating, nausea, respiratory distress with hypotension, vascular collapse, and death have occurred.
Clinical Considerations: For treatment of a variety of manifestations of thiamine deficiencies, including beriberi and peripheral neuritis, which is also associated with pellagra, thiamine is used. In acute Wernicke’s encephalopathy, which occurs in some chronic alcoholics, thiamine is administered intravenously. It can temporarily correct certain rare genetic metabolic disorders (maple syrup urine disease and subacute necrotizing encephalomyelopathy). It has also been used as an insect repellent, but there is no evidence of its effectiveness.
 RIBOFLAVIN (VITAMIN B2)
RIBOFLAVIN (VITAMIN B2)
Riboflavin (RYE-boe-flay-vin) (vitamin B2) is a water-soluble vitamin composed of flavin and d-ribitol.
Source: Riboflavin is abundant in both plants and animals. However, dairy products and meat (especially organ meats such as liver) are the best sources of this vitamin. It is also present in green leafy vegetables and yeast. Riboflavin is relatively stable to heat, and cooking will not cause an appreciable loss. It is destroyed by ultraviolet radiation.
Recommended Dietary Allowance: The RDA for riboflavin ranges from 1.1 (adult women) to 1.2 mg (adult men). Table 12-2 lists the requirements for other groups. Requirements for riboflavin usually parallel caloric intake or metabolic body size.
Role: Riboflavin functions in the body as a component of two flavoprotein coenzymes, riboflavin phosphate (flavin mononucleotide [FMN]) and flavin adenine dinucleotide (FAD). Flavoprotein coenzymes in turn are proteins that act as electron acceptors and are involved in a variety of oxidation-reduction reactions. Riboflavin is also indirectly involved in maintaining the integrity of the erythrocytes.
Deficiency: Symptoms of riboflavin deficiency usually involve the lips, tongue, and skin. Sore throat and angular stomatitis (cheilosis) appearing as an ulceration with painful fissuring at the corners of the mouth are early and frequent findings. The lips may be either unusually red or whitish because of desquamation. Later, glossitis can occur, with the dorsum of the tongue becoming pebbly or granular. Contact with food or drink may produce pain or a burning sensation on the tongue. In some instances, the tongue may become magenta or purplish-red. Excessive salivation and enlargement of the salivary glands may occur. Skin manifestations include a greasy, scaling inflammation around the nose, cheeks, and chin. Involvement of the scrotum and the vulva is frequent. Other manifestations of a severe riboflavin deficiency are normocytic, normochromic anemia and neuropathy.
Clinical Considerations: Riboflavin deficiency is most likely to be seen in alcoholics, economically deprived individuals, or patients with severe gastrointestinal disease that causes loss of appetite, vomiting, and malabsorption syndromes. Oral contraceptives and probenecid may be associated with an increased need for riboflavin. The manifestations of riboflavin deficiency are difficult to distinguish from those of other B-vitamin deficiencies because of the similarities in syndromes. The discovery of a deficiency of riboflavin warrants the use of a multivitamin because deficiency of several vitamins often coexists. The ease of measuring riboflavin in the urine has prompted its use as a marker in drug studies to measure compliance.
 NIACIN OR NICOTINIC ACID (VITAMIN B3)
NIACIN OR NICOTINIC ACID (VITAMIN B3)
Niacin (NYE-a-sin), or nicotinic acid, is converted in the body to niacinamide or nicotinamide, its active form, to serve as a vitamin. These water-soluble organic compounds have the ability to alleviate a deficiency syndrome known as pellagra.
Source: Good sources of niacin are lean meats, fish, liver, poultry, legumes, and whole grains. Pellagra was at one time a common disease of the southeastern United States among persons subsisting on a diet exclusively of corn products because corn is extremely low in tryptophan, a precursor of this vitamin. Beans are often used in combination with corn to rectify this deficiency.
Recommended Dietary Allowance: Niacin requirement in the diet is somewhat dependent on both caloric and protein intake. Because tryptophan, an amino acid found in dietary protein, is metabolized to niacin in the body, intake of protein would reduce the amount of vitamin needed in the diet. The recommended dietary allowance for niacin is 16 mg niacin equivalents (NE) for men (1 NE is equal to 1 mg of niacin or 60 mg of tryptophan) and 14 mg NE for women. Oral doses of 15 to 20 mg of niacin daily are sufficient as a dietary supplement if patients have normal gastrointestinal absorption.
Role: Nicotinic acid, like riboflavin, plays a key role in metabolism by participating in a variety of oxidation-reduction reactions (transfer of electrons). In the body, it is converted into two active forms, the coenzymes nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP). This vitamin serves as an essential coenzyme for dehydrogenases involved in the Krebs cycle. The Krebs cycle is responsible for anaerobic carbohydrate metabolism and lipid and protein metabolism.
The clinical syndrome produced by niacin deficiency is pellagra, so named because the skin becomes rough (Latin: pelle, skin; agra, rough). Early symptoms are an erythematous cutaneous eruption on the back of the hands, glossitis, and stomatitis. In advanced stages, pellagra can be diagnosed by the classic “three Ds”: dermatitis, diarrhea, and dementia. The dermatitis consists of redness, thickening, and roughening of the skin, followed by scaling desquamation and depigmentation. Diarrhea is caused by atrophy of the gastrointestinal tract mucosal epithelium, followed by inflammation of the mucosal lining of the esophagus, stomach, and colon. The dementia results from regressive changes in the ganglion cells of the brain and tracts of the spinal cord. Death may also result.
During the course of pellagra, symptoms are evident in the oral cavity. A burning sensation occurs throughout the oral mucosa. The lip and lateral margins of the tongue are initially reddened and swollen. In the later stages, the entire dorsal surface of the tongue becomes red and swollen. In acute stages, vascular hyperemia, proliferation, hypertrophy, and atrophy occur successively in the papillae. Papillary loss may ultimately become complete, with the tongue surface becoming beefy red. Deep penetrating ulcers may appear on the tongue surface. In the gingiva, desquamative epithelial degeneration may occur, exposing the tissue to infection, inflammation, and fibrinous exudation. Gingivitis caused by pellagra is characterized by ulcers in the interdental papillae and marginal gingiva. Excessive salivary secretion with enlargement of the salivary glands also occurs.
Niacin deficiency occurs most often in poverty-stricken areas of the world because of inadequate intake. Deficiency may also arise from chronic alcoholism, gastrointestinal disturbances, pregnancy, hyperthyroidism, and infections.
Side effects that occur from ingestion of large doses of niacin include cutaneous flushing, pruritus, and gastrointestinal distress. These side effects can be reduced by administering an aspirin one-half hour before the niacin is ingested. Other adverse reactions include increased sebaceous gland action and increased gastrointestinal motility. With chronic use, dry skin, xerostomia, hyperuricemia, peptic ulcer, blurred vision, nervousness, panic, and hyperglycemia can occur. Abnormal liver function tests, prothrombin time, and hypoalbuminemia have been reported.
Clinical Considerations: Niacin or nicotinic acid and niacinamide are used as a vitamin in the treatment of pellagra. Only high-dose niacin is useful in the treatment of hyperlipidemias. It reduces plasma cholesterol, triglycerides, very-low-density lipoproteins (VLDL), low-density lipoproteins (LDL), and chylomicrons. These effects are dose-dependent. Niacin can also be used in combination with other lipid-lowering agents, so that a lower dose of each may be used.
 PYRIDOXINE (VITAMIN B6)
PYRIDOXINE (VITAMIN B6)
Pyridoxine (peer-i-DOX-een) is one of three different pyridoxine derivatives known as vitamin B6. The other two derivatives, pyridoxal and pyridoxamine, are chemically similar.
Source: Vitamin B6 is present in most foods of both plant and animal origin. Good sources of this vitamin include whole grain cereals, meat, legumes, eggs, and some vegetables. These similar foods are the sources of many of the B vitamins.
Recommended Dietary Allowance: The RDA for vitamin B6 varies from 1.0 to 1.7 mg daily for men and women ages 9 to older than 70 years (see Table 12-2).
Role: To exert physiologic activity, all three forms of vitamin B6 are converted to pyridoxal phosphate in the body. Pyridoxal phosphate is the active coenzyme form of vitamin B6 and participates in all metabolic reactions that require the vitamin. Pyridoxal phosphate acts as a coenzyme in a variety of metabolic transformations of amino acids, including transamination and decarboxylation.
Vitamin B6 deficiency is rare because of the widespread distribution of this vitamin in food. The characteristics of vitamin B6 deficiency resemble those of riboflavin, niacin, and thiamine deficiencies. These include angular cheilosis, stomatitis, dermatitis, and erythema of the nasolabial folds. The dorsal mucosa of the tongue seems to be unusually sensitive to a single deficiency or mixed deficiencies of the B vitamins. Specifically, glossitis resulting from pyridoxine deficiency has been described in which the tongue’s surface is smooth, slightly edematous, painful, and purplish.
Adverse Reactions: Pyridoxine is usually nontoxic. When it is given parenterally in large doses, peripheral neuritis may be produced.
Clinical Considerations: Vitamin B6 can interact with other therapeutically useful drugs. For example, isoniazid (INH), a drug used to treat tuberculosis, inhibits the action of vitamin B6 by blocking both the formation and the reaction involving pyridoxal phosphate, the active coenzyme. INH-induced vitamin B6 deficiency can be prevented or treated by the administration of pyridoxine. For this reason, patients taking INH usually also take vitamin B6. Long-term administration or high doses of steroids can require administration of folic acid. Because the anticonvulsants, such as carbamazepine and phenytoin, interfere with the absorption and storage of folic acid, patients taking these medications are given pyridoxine (see Table 12-4).
Vitamin B6 administration can cancel the therapeutic and side effects of levodopa, a drug used to treat Parkinson’s disease. If carbidopa, a peripheral decarboxylase inhibitor, is administered simultaneously with levodopa (the combination is Sinemet), pyridoxine may be administered concomitantly. In practice, Sinemet is currently used almost exclusively rather than levodopa alone.
Certain other drugs, such as cycloserine, hydralazine, and pyrazinamide, may produce a pyridoxine deficiency. Estrogenic steroids can produce vitamin B6 deficiency in women. About 20% of women taking oral contraceptive agents can be shown to have a biochemical pyridoxine (B6) deficiency. Usual RDAs seem to be enough to prevent this situation, and women taking birth control pills should routinely be encouraged to take supplemental pyridoxine.
The use of pyridoxine for many conditions has not been shown to be effective in well-controlled trials (premenstrual syndrome, acne, vertigo, tardive dyskinesia, asthma, or alcohol intoxication).
 FOLIC ACID
FOLIC ACID
Folic acid (FOE-lik) (pteroylglutamic acid, folacin, folate) is a form of the water-soluble vitamin B9.
Source: Significant sources of folic acid include glandular meats such as liver, some fruits and vegetables, wheat germ, and yeasts. Because availability of folic acid from foods is highly variable, a wide margin of safety is allowed in the RDA.
Recommended Dietary Allowance: The RDA for folic acid is 400 µg daily for healthy, nonpregnant, or nonlactating adults.
Role: The biologically active form of folic acid is the reduced derivative tetrahydrofolic acid, which is formed enzymatically in the body. Tetrahydrofolic acid functions primarily in the transfer and utilization of one-carbon groups.
Certain microorganisms synthesize their own folic acid from para-aminobenzoic acid (PABA). The sulfonamides exert their bacteriostatic effect by antagonizing PABA and thereby interfering with the biosynthesis of folic acid in these organisms. This antagonism has no effect on humans because they require preformed folic acid and do not synthesize their own.
Folic acid deficiency, the most common deficiency in the United States, produces megaloblastic anemia, which is indistinguishable from that caused by vitamin B12 deficiency. Other symptoms include weakness, weight loss, loss of skin pigmentation, and mental irritability. As with riboflavin deficiency, oral manifestations of folic acid deficiency include glossitis, angular cheilosis, and gingivitis. The glossitis begins with swelling and pallor of the tongue followed by desquamation of the papillae and accompanied by minute ulcers with fiery red borders.
Some causes of folic acid deficiency are inadequate diet, pregnancy, malabsorption syndrome, and chronic alcoholism. Pregnant women need supplemental synthetic folacin and should not rely solely on dietary sources. The absorption of folate decreases during pregnancy and in patients taking oral contraceptives.
Several drugs have been reported to produce folic acid deficiencies, including the anticonvulsants, oral contraceptives, and nitrofurantoin. Some drugs act as folic acid antagonists (e.g., pyrimethamine, trimethoprim) (see Table 12-4). The anticonvulsants produce a deficiency by interfering with the conversion of folate to a form of the vitamin that can penetrate the brain. Some cancer chemotherapy agents prevent the formation of tetrahydrofolic acid and interfere with DNA synthesis. Folic acid is not an antidote to an overdose from a folic acid antagonist (e.g., methotrexate); leucovorin calcium is used.
Adverse Reactions: Folic acid is relatively nontoxic. Allergic reactions have been reported rarely and include rash, itching, and respiratory difficulty.
Clinical Considerations: Although the administration of folic acid will cause remission of the hematologic effects of pernicious anemia, it will not prevent the neurologic effects caused by a deficiency of vitamin B12. Therefore folic acid can mask a vitamin B12 deficiency. For this reason the Food and Drug Administration (FDA) has limited the dose of folic acid per tablet that can be purchased without a prescription to 0.4 mg in normal vitamin supplements and 0.8 mg for pregnant or lactating women (over-the-counter [OTC] prenatal vitamins). The use of folic acid several months before conception and early in pregnancy can help prevent neural tube defects.
 CYANOCOBALAMIN (VITAMIN B12)
CYANOCOBALAMIN (VITAMIN B12)
Cyanocobalamin (sye-an-oh-koe-BAL-a-min) (vitamin B12) is a chemically complex substance that contains four extensively substituted pyrrole rings surrounding an atom of cobalt. A cyanide molecule is attached to the cobalt, thus the name cyanocobalamin. Vitamin B12 is heat stable at a neutral pH but is readily destroyed by heat at an alkaline pH.
Source: The only sources of vitamin B12 in nature are certain microorganisms that synthesize the vitamin. When vegetable produce is contaminated with these microorganisms, the produce possesses the vitamin. Animals depend on synthesis within their own intestinal tracts. Human vitamin B12, synthesized within the gastrointestinal tract, is not available for absorption.
Good sources of vitamin B12 include foods of animal origin such as liver, meat, milk, cheese, and eggs. Vegans (strict vegetarians who do not eat animal or dairy products) can become deficient because they do not eat these foods. In recent years, some vegetable products, such as soy milk, have been fortified with vitamin B12.
Vitamin B12 is absorbed from the distal ileum by a receptor-mediated process. Without the presence of intrinsic factor, a protein-binding factor that aids in the absorption of vitamin B12, it cannot be well absorbed. This system is saturated by about 3 mg of vitamin B12. With very large doses of oral vitamin B12 (1 mg) daily, the vitamin may be absorbed independent of intrinsic factor. Absorption of vitamin B12 is decreased by damage to the stomach or ileum.
Recommended Dietary Allowance: The RDA of vitamin B12 is 2.4 µg, with an additional 2.6 µg and 2.8 µg during pregnancy and lactation, respectively. Oral doses between 1 and 25 µg are adequate if the gastrointestinal absorption is normal. In pernicious anemia, a maintenance injection of vitamin B12 is recommended once a month for life.
Role: Vitamin B12 serves as a coenzyme for the hydrogen transfer and isomerization process required in the conversion of methylmalonyl-CoA to succinyl-CoA. Thus vitamin B12 is important in the metabolism of fats and carbohydrates.
The symptoms of vitamin B12 deficiency include inadequate hematopoiesis, gastrointestinal tract disturbances, inadequate myelin synthesis, and generalized debility. The lack of this vitamin affects the cells that are most actively dividing such as those in the bone marrow and gastrointestinal tract. The erythroblasts do not undergo proper division, resulting in megaloblastic anemia. Atrophic changes occur in the alimentary canal. The synthesis of abnormal fatty acids, which are then incorporated into cell membranes, may produce the neurologic manifestations of deficiency, including peripheral neuropathies and spinal cord and organic brain syndromes. The patient suffers from weakness, numbness, and difficulty in walking, which are symptoms that fluctuate with remission and relapses. The skin may have a distinctive lemon-yellow hue.
The most common cause of vitamin B12 deficiency is pernicious anemia, an autoimmune disease that prevents the production of intrinsic factor. The secretory cells in the gastric mucosa do not produce intrinsic factor, and thus vitamin B12 is very poorly absorbed. With a gastrectomy, usually for the treatment of peptic ulcer in the “old days,” intrinsic factor secretion ceases. It then takes 3 to 6 years for a vitamin B12 deficiency to develop. Other causes of vitamin B12 deficiency include inadequate dietary intake, malabsorption syndromes, and gastric bypass surgery.
Pernicious anemia results in several oral manifestations (see Color Plate 3). Recurrent attacks of soreness and burning of the tongue occur followed by glossitis, at the peak of which the tongue is extremely painful and red. Atrophy of the filiform and fungiform papillae is a common occurrence. Involvement of the circumvallate papillae may cause diminution of taste. Painful, bright red lesions may occur in the buccal and pharyngeal mucosa and undersurface of the tongue.
Adverse Reactions: Even large doses of vitamin B12 are usually nontoxic. Diarrhea, itching, urticaria, and swelling have occasionally been reported. If intrinsic factor is given with the vitamin B12, an allergy to hog protein (a source of exogenous intrinsic factor) may be exhibited.
Clinical Considerations: As mentioned previously, patients who are strict vegetarians (rarely) or who have had a gastrectomy can exhibit the symptoms of vitamin B12 deficiency. Ingestion of other agents can alter the absorption of vitamin B12. For example, vitamin C may destroy the vitamin B12 levels in food. Pregnancy and use of the sweetener sorbitol increase vitamin B12 absorption. Absorption of vitamin B12 is decreased in persons with pyridoxine deficiency, iron deficiency, or hypothyroidism. Sustained-release potassium and anticonvulsants may decrease the absorption of vitamin B12. Vitamin B12 has also been used, without any proof of efficacy, to treat trigeminal neuralgia, psychiatric disorders, and fatigue. Because intrinsic factor is not required for absorption from an intramuscular (IM) site, vitamin B12 can be administered intramuscularly (100 µg/month) in the absence of intrinsic factor. An oral dose of 1 mg daily is equivalent to the 100 µg/month intramuscularly. Because oral administration of vitamin B12 is unreliable, the IM route is preferred.
 PANTOTHENIC ACID
PANTOTHENIC ACID
Pantothenic acid is another compound required to form acetyl-CoA. The active form of pantothenic acid is a component of the more complex compound, coenzyme A.
Source: Pantothenic acid is a part of all living material. Egg yolk, bran, yeast, and beef liver are excellent sources.
Recommended Dietary Allowance: It is suggested that a daily dietary intake of 5 to 7 mg is adequate for adults with normal gastrointestinal absorption. Table 12-3 lists the estimated safe and adequate daily dietary intake (ESADDI) for pantothenic acid.

TABLE 12-3 DIETARY REFERENCE INTAKES: TOLERABLE UPPER INTAKE LEVELS FOR SELECTED VITAMINS AND ELEMENTS* Rights were not granted to include this table in electronic media. Please refer to the printed book. From Otten JJ, Hellwig JP, Meyers LD, eds: Dietary reference intakes: the essential guide to nutrient requirements, Washington, DC, 2006, National Academics Press. National Academics of Sciences
Role: The physiologically active form of pantothenic acid is incorporated into coenzyme A, which serves as a coenzyme in various metabolic reactions; some of these reactions involve the transfer of acetyl (two-carbon) groups. Pantothenic acid is required for gluconeogenesis and synthesis of fatty acids and sterols and steroid hormones. Both pantothenic acid and thiamine are required for the oxidative decarboxylation of pyruvate to produce acetyl-CoA. Pantothenic acid also functions as part of a glucose-carrier system to facilitate absorption through the intestinal mucosa. It is essential for normal epithelial function.
Deficiency: Because clinical deficiencies of pantothenic acid are extremely rare in humans, they are produced experimentally in humans only by using a pantothenic acid antagonist. Deficiency may develop in patients with liver disease or who drink excessive alcohol. The symptoms of pantothenic acid deficiency include fatigue, headache, malaise, nausea, abdominal pain, burning feeling of hands and feet, and cramping of leg muscles.
Clinical Considerations: Pantothenic acid has been used to treat gastrointestinal tract paralysis after surgery because it apparently promotes gastrointestinal motility. Although a deficiency of pantothenic acid produces gray hair in black rats, there is absolutely no evidence that taking pantothenic acid reverses gray hair in humans.
 BIOTIN
BIOTIN
Biotin was initially demonstrated to be an essential growth factor for yeast, and it was later isolated from both yeast and egg yolk.
Source: Although biotin is present in almost all foods, good sources include liver, cow’s milk, egg yolk, and yeast. It is also synthesized by the microflora in the intestinal tract, so the amount of biotin excreted in the feces can actually exceed the intake.
Recommended Dietary Allowance: Although no minimum daily requirement of biotin has been established, the suggested adequate daily dietary intake for adults is 25 to 35 mg. Table 12-3 lists the RDA for biotin.
Role: Biotin is a coenzyme required in metabolism in carbon dioxide fixation reactions, β-carboxylation, and deamination.
Deficiency: Biotin deficiency is extremely rare but can occur with long-term parenteral nutrition. A biotin deficiency can be induced by eating large quantities of raw egg white. Avidin, a component of egg white, combines with biotin in the gastrointestinal tract and prevents its absorption. If the egg white is cooked, the avidin is denatured and has no activity. When biotin deficiency is experimentally induced by concurrent administration of large amounts of raw egg white containing avidin, symptoms include loss of appetite, mental depression, hyperesthesia of the skin, nausea, malaise, and dry dermatitis.
Other B Vitamins
Vitamin B15 and vitamin B17, also known as pangamic acid and amygdalin (Laetrile), respectively, have been shown to be neither vitamins nor important in human nutrition.
Neither choline (KOE-leen) nor inositol (EYE-nos-e-tal) has been demonstrated to be required in the human diet. They serve as lipotropic agents and prevent fatty infiltration of the liver. Choline serves as a precursor to acetylcholine. In humans, no deficiency for either choline or inositol has been demonstrated. Deficiencies of choline (in rats) and inositol (in mice) have been produced.
FAT-SOLUBLE VITAMINS
Fat-soluble vitamins include vitamins A, D, E, and K. Figure 12-3 provides some examples of good sources of fat-soluble vitamins.

FIGURE 12-3 Examples of foods containing fat-soluble vitamins. A, Carrots are a rich source of carotenes, which provide the body with vitamin A, also found in milk (B) and some cheeses (C). Dairy products also contribute to the body’s absorption of vitamin D. C, Vegetable oils are the best sources of vitamin E, whereas spinach (D) is rich in vitamins A, E, and K. (Copyright 2009 Jupiterimages Corporation.)
Vitamin A
Vitamin A, which is an essential fat-soluble compound, is necessary for normal growth and for maintaining the health and integrity of certain epithelial tissues. The term vitamin A represents a group of retinoids (e.g., vitamin A1 [retinol], vitamin A2 [3-dehydroretinol]) and carotenoids. The retinoids include both naturally occurring and synthetic analogs of vitamin A. By cleavage of the carotene molecule, two molecules of vitamin A aldehyde (retinal) are formed.
 SOURCE
SOURCE
Vitamin A1 occurs naturally in saltwater fish and animal tissues. Vitamin A2 is found in freshwater fish. Preformed vitamin A is found in milk, liver, and some cheeses. Margarine can be fortified with vitamin A. However, carotenes provide the greatest source of vitamin A in most diets. Carotenes are found in various pigmented fruits, such as apricots, peaches, tomatoes, and watermelon, and in vegetables such as carrots, pumpkins, broccoli, spinach, and sweet potatoes. A dark green, yellow, or orange color indicates that a vegetable or fruit has carotene.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The adult RDA for vitamin A is 700 to 1300 retinol equivalents (RE). One RE is equal to 1 µg of retinol or 6 µg of β-carotene.
 ROLE
ROLE
Vitamin A is essential for the maintenance of the photoreceptor mechanism of the retina; the integrity of the epithelia, such as the mucous membranes of the eye, and the mucosa of the respiratory, gastrointestinal, and genitourinary tracts; and lysosome stability. Vitamin A plays a significant role in maintaining the integrity and controlling differentiation and possibly the normal permeability of the cell membrane and the membrane subcellular particles. Vitamin A deficiency decreases the activity of osteoblasts and odontoblasts, thereby reducing the growth of bones and teeth. In contrast, excessive doses of vitamin A accelerate bone and cartilage resorption and new bone formation.
 DEFICIENCY
DEFICIENCY
The human liver may store enough vitamin A to meet physiologic demands for as long as a year, and therefore a deficiency of this vitamin is rare. Deficiencies, if they do occur, generally result from inadequate intake of the vitamin; a malabsorption syndrome, especially biliary tract disease; or severe liver disease. Deficiency of the vitamin leads to impaired vision in dim light, called night blindness (nyctalopia). It also results in keratinization of mucosa and cornea. Corneal keratinization leads to impairment of vision, called xerophthalmia. Irritation and inflammation may occur on the cornea, a condition called keratomalacia. Keratinization may also occur in the oral cavity and mucosa. The normal defense mechanisms of ciliary movement and mucous production are impaired, producing irritation and inflammation of these surfaces. Loss of the senses of taste and smell also occurs in vitamin A deficiency. Deficiency of vitamin A during pregnancy and infancy contributes to the development of enamel hypoplasia and caries in primary teeth.
 TOXICITY
TOXICITY
Excessive intake of vitamin A results in a toxic condition called hypervitaminosis A. The characteristics of this toxic reaction include itching skin, desquamation, coarse or absent hair, painful subcutaneous swellings, gingivitis, hyperirritability, and limitation of motion. Hyperostosis in the bone is easily demonstrated on radiography. In infants, headache from increased intracranial pressure, gastrointestinal distress, jaundice, and hepatomegaly may occur. Because the margin of safety of vitamin A intake is large, a toxic reaction can occur only after long-term daily ingestion of more than 50,000 RE.
Acute poisoning has been reported in both infants and adults. After ingestion of lesser amounts by infants, increased intracranial pressure with bulging fontanel and vomiting was reported. When the Vikings landed in Iceland, they ingested polar bear liver, a rich source of vitamin A, and died from acute poisoning.
 PREGNANCY CONSIDERATIONS
PREGNANCY CONSIDERATIONS
The use of or exposure to excess retinoids (vitamin A or its analogs) during pregnancy can have serious teratogenic effects. Excessive doses of both vitamin A and the analogs etretinate and isotretinoin are classified as FDA pregnancy category X drugs (see discussion of FDA category drugs in Chapter 24). Other retinoids, such as adapalene (Differin) and tretinoin (Retin-A), are FDA category C drugs because the equivalent amount of vitamin A absorbed is substantially below the RDA.
 VITAMIN A ANALOGS
VITAMIN A ANALOGS
Tretinoin (TRET-i-noyn) (Retin-A), the acid form of vitamin A, is a topical product that causes skin peeling and is used to treat acne. Another indication is the treatment of wrinkles. Erythema, desquamation, and unusual sun sensitivity can occur.
Isotretinoin (eye-soe-TRET-i-noyn) (13-cis-retinoic acid, Accutane) is used orally for treatment of severe cystic acne. Side effects include corneal opacities; abnormal liver function tests; elevated plasma triglycerides; and rarely, pseudotumor cerebri. It is highly teratogenic (FDA category X) and should not be used without adequate birth control measures. Remission of acne can remain after the drug has been withdrawn.
Like all vitamin A analogs, these agents are contraindicated in anyone who might become pregnant within the next few years, and they are FDA pregnancy category X drugs. Side effects are similar to those of hypervitaminosis A and relate to the mucocutaneous, musculoskeletal, hepatic, and central nervous systems. Oral manifestations include gingival bleeding, inflammation, and xerostomia with its concomitant implications. A drug interaction with alcohol exists because they can both produce hypertriglyceridemia. Tetracyclines may increase the potential for a rare side effect, pseudotumor cerebri.
Vitamin D
Vitamin D is a collective term used to refer to both vitamin D2 and vitamin D3, two closely related sterols. Vitamin D3 (cholecalciferol [koh-lee-kal-SIF-e-role]) is produced in the skin of mammals by the action of sunlight (ultraviolet rays) on its precursor, 7-dehydrocholesterol. Cholecalciferol (vitamin D3) is also present in some foods and is added as a supplement to dairy products. Ergocalciferol (er-goe-kal-SIF-e-role) (vitamin D2), the vitamin D found in plants, is the form of vitamin D used in vitamin supplements. Vitamin D2 is produced by the commercial irradiation (by ultraviolet light) of ergosterol.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The adequate daily dietary intake of vitamin D is 5 µg/day for children and 5 to 15 µg/day for adults.
 ROLE
ROLE
Vitamin D promotes normal mineralization of bone by stimulating intestinal absorption of calcium and decreasing the excretion from the kidney.
 DEFICIENCY
DEFICIENCY
The deficiency of vitamin D produces inadequate absorption of calcium and phosphate with a decrease in plasma calcium. Parathyroid hormone secretion is stimulated, which removes calcium from the bone to restore plasma levels. In children, this deficiency results in rickets, a disease involving a decreased mineralization of newly formed bone and cartilage tissue. Children with rickets have bones that are unusually soft and easily bent, compressed, or fractured. Under the stress and strain of weight-bearing, the gross deformities of rickets, including spine curvature and bowing of the legs, become evident. Because of the excess formation of osteoids, a squared appearance of the head occurs. Collapse of the ribs and protrusion of the sternum (pigeon breast syndrome) are also seen. Bone pain and muscle weakness may be present.
Vitamin D deficiency during pregnancy or in young children may result in enamel hypoplasia, but the teeth may remain caries free. In adults, vitamin D deficiency produces a disease state called osteomalacia. In general, there is decreased bone density because of inadequate mineralization, which results in an excess of osteoid matrix. Because of the weakness of the bones, pathologic fractures and deformities of weight-bearing bones occur. This happens most often during times of increased calcium use such as pregnancy or lactation. Persons with malabsorption syndromes, alcoholics, those adhering to a low-fat diet, strict vegetarians, and those undergoing anticonvulsant therapy or using sedatives or tranquilizers are more prone to vitamin D deficiency.
 TOXICITY
TOXICITY
The symptoms of hypervitaminosis D, which may result from either long-term or short-term ingestion of excessive quantities of vitamin D, are caused by abnormal calcium metabolism. The signs and symptoms of vitamin D toxicity include weakness, fatigue, headache, nausea, vomiting, and diarrhea. With prolonged hypercalcemia, calcification of the blood vessels, heart, lung, and kidney can occur. Continued ingestion of large doses in a normal adult is likely to produce hypervitaminosis D.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
Vitamin D is used to prevent and treat rickets. It is also used to treat chronic hypocalcemia, hypophosphatemia, osteodystrophy, and osteomalacia. Dihydroxycholecalciferol (calcitriol, 1,25 [OH]2D3) and dihydroxyergocalciferol ([OH]2D2) do not require activation by the kidneys and are used for hypocalcemia in patients with chronic renal failure undergoing dialysis. Because of the need for a functioning kidney to activate vitamin D, the patient is given the preformed active vitamin D. Exogenous dihydrotachysterol (DHT), a close isomer of vitamin D, is hydroxylated in the liver to 25-hydroxy-DHT.
 OSTEOPOROSIS
OSTEOPOROSIS
Normal bone is continuously being made and broken down in response to various stimuli. Osteoporosis occurs when the equilibrium between the resorption and formation of bone becomes negative. The loss of bone mass predisposes the patient to fractures. Patients who develop osteoporosis are much more likely to sustain fractures of their bones, including vertebra or hip, which often reduces their quality of life. The thin, white or Asian woman who smokes is most likely to develop osteoporosis. Patients taking chronic corticosteroids develop osteoporosis earlier. The most common occurrence of osteoporosis is in postmenopausal women because of inadequate sex hormones. Calcium intake of the equivalent of 1200 to 1500 mg of elemental calcium is recommended for postmenopausal women. Weight-bearing exercise may modulate osteoporosis. Estrogen replacement therapy (ERT) reduces the risk of osteoporosis, and recent evidence has determined that the minimum dose of estrogen for this effect is 0.3 mg (0.625 mg was previously recommended). Some women fear the side effects of estrogens, including an increased risk of breast cancer (small increase in risk) and increased risk of uterine cancer (nullified by using progestin with the estrogen). Therefore they do not take supplemental estrogens and osteoporosis is not prevented.
Calcium supplementation is encouraged to prevent osteoporosis in postmenopausal women. Osteoporosis is more effectively prevented when adequate intake of calcium begins in their 20s and 30s.
Three new drugs are indicated for the management of osteoporosis and have been recently released. The bisphosphonates include alendronate and etidronate.
Alendronate (Fosamax), a third-generation bisphosphonate, has been shown to inhibit osteoclastic activity and reduce bone turnover. Under the influence of alendronate, bone formation is greater than bone resorption. It can produce an increase in bone density and reduces fractures for a 5-year period. It is taken on an empty stomach with plain water with instructions to refrain from lying down for 30 minutes thereafter. With higher doses, there is a potential for gastrointestinal adverse reactions. Salicylates can increase the gastrointestinal side effects.
Etidronate (Didronel) has been shown to increase bone mass and reduce the incidence of fractures for at least 2 years. It has been shown to increase the bone mass slightly.
Calcitonin (Miacalcin) is administered by intranasal inhalation. It has been shown to increase spinal bone mass in postmenopausal women with osteoporosis. Further study is needed to define the appropriate therapeutic use of these agents.
Although it is known that fluoride can stimulate bone formation, the use of sodium fluoride in the treatment of osteoporosis is controversial.
Vitamin E
There are eight naturally occurring tocopherols possessing vitamin E activity. α-Tocopherol is the most active tocopherol. α-Tocopherol is found in wheat, sunflower, cottonseed, and olive oils. Although the metabolic role of vitamin E is not understood, it is known that this vitamin functions as an antioxidant.
 SOURCE
SOURCE
The best sources of vitamin E are vegetable oils such as soybean, corn, and cottonseed oils. Other sources include fresh greens and vegetables.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
It has been estimated that a daily intake of 10 to 20 mg of vitamin E will keep the vitamin E serum level within a normal range. The Food and Nutrition Board of the National Academy of Sciences recommends between 8 and 10 α-tocopherol equivalents (TE) for adults per day (see Table 12-2).
 ROLE
ROLE
The action of vitamin E is probably exerted via its antioxidant effect. It prevents the oxidation of vitamins A and C, protects polyunsaturated fatty acids in membranes from attack by free radicals, and protects red blood cells against hemolysis. Vitamin E increases the absorption and utilization of vitamin A and protects against hypervitaminosis A.
Vitamin E is being intensely studied for its effect on clotting and on prevention of thromboses. It can be shown to interfere with clotting, but the exact mechanism is unknown (may be related to inhibition of prostaglandins). The evidence of vitamin E’s role in cardiovascular disease is strengthened because studies have shown a decrease in both myocardial infarction and stroke.
 DEFICIENCY
DEFICIENCY
Deficiency of vitamin E, produced in laboratory animals, can affect the reproductive, muscular, cardiovascular, and hematopoietic systems. Vitamin E deficiency in male rats has resulted in reproductive failure and sterility; in the pregnant female rat, it has led to fetal death and resorption.
In humans, vitamin E has been used for the treatment of sterility and habitual abortion, but there is no conclusive evidence that this vitamin provides any beneficial effect in these conditions. Although vitamin E has been used to treat several cardiovascular diseases, there is no scientific rationale for this use.
A deficiency of vitamin E can occur in malabsorption syndromes and in premature infants with impaired absorption ability. A deficiency of vitamin E has also been reported to cause anemia resulting from a decreased erythrocyte life span and abnormal hematopoiesis. Oxidizing agents can more easily hemolyze the erythrocytes from vitamin E–deficient animals.
 TOXICITY
TOXICITY
Vitamin E is generally thought to have low toxicity. Levels of vitamin E greatly in excess of the normal dietary requirements have been administered to human subjects with no apparent adverse effect. Nausea, diarrhea, fatigue, weakness, and rash have occurred rarely. Recently, there have been concerns raised about the safety of vitamin E, particularly in doses greater than 400 IU/day. Evidence suggests that regular use of high-dose vitamin E supplements may increase the risk of death.*
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
Vitamin E therapy has been recommended for treatment of a wide variety of human diseases that are similar to conditions of vitamin E deficiency. Vitamin E has been used for many indications in which documentation is poor such as intermittent claudication and protection against certain air pollutants. Other researchers have found that vitamin E supplementation had no effect on work performance, sexuality, or general well-being. At present, no therapeutic use of vitamin E has been proved by controlled scientific studies, with the exception of hemolytic anemia of the newborn.
Pharmacologic doses of vitamin E (used as a drug) have been used as an antioxidant in premature infants exposed to high concentrations of oxygen to reduce the incidence and severity of retinopathy and bronchopulmonary dysplasia. Vitamin E has been used to treat both β-thalassemia and sickle cell anemia with questionable success. The usual dose of vitamin E for its protective cardiovascular effect is 400 IU.
Vitamin K
Vitamin K was originally found to be a fat-soluble substance present in hog liver fat and alfalfa. Large quantities of the vitamin are also found in the feces of most species of animals. At least two distinct natural substances possess vitamin K activity: vitamin K1 and vitamin K2. Vitamin K2 consists of several substances, with menaquinone-4 being the most active form. Vitamin K1 (phytonadione [fye-toe-na-DYE-one], phytyl-menaquinone, phylloquinone) is found in plants. Vitamin K2 (menaquinone, multi-prenyl-menaquinone) is synthesized by gram-positive bacteria present in the gastrointestinal tract. Both Vitamins K1 and K2 require bile salts for absorption from the intestines.
 SOURCE
SOURCE
Vitamin K occurs in green vegetables, such as alfalfa, cabbage, and spinach, and in egg yolk, soybean oil, and liver. Vitamin K is synthesized by gram-positive bacteria, and the microorganisms in the intestinal flora can provide humans with some vitamin K. Synthetic vitamin K is a form of vitamin K that has activity similar to that of naturally occurring vitamin K.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The adequate daily dietary intake for vitamin K is 60 to 120 µg for males and 60 to 90 µg for females. Generally, the normal diet and intestinal bacteria provide all the necessary vitamin K.
 ROLE
ROLE
Vitamin K is essential for the hepatic synthesis of four of the clotting factors: II (prothrombin), VII, IX, and X. Without adequate clotting factors, normal blood clotting does not occur.
 DEFICIENCY
DEFICIENCY
A vitamin K deficiency can produce hypoprothrombinemia. In the absence of this vitamin, bleeding will result. With a severe deficiency of vitamin K, the smallest trauma may produce hemorrhage. The most common sites of hemorrhage are operative wounds, skin (petechial bleeding), mucous membranes in the intestinal tract, and serosal surfaces. Ecchymoses, epistaxis, and hematuria are also common.
A vitamin K deficiency is usually caused by an inadequate intake or absorption (lack of bile salts) of the vitamin or by decreased normal bacterial flora resulting from prolonged antibiotic use. The newborn can have vitamin K deficiency because the intestinal organisms have not yet been established.
 TOXICITY
TOXICITY
The naturally occurring vitamins K1 and K2 are essentially nontoxic in massive doses, and vitamin K (menadione) must be administered in large doses before toxicity can be demonstrated. It has been implicated in producing hemolytic anemia in newborns and hemolysis in persons suffering from glucose-6-phosphate dehydrogenase (G6PD) deficiency. Hypersensitivity reactions can occur.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
Anticoagulant drugs such as warfarin competitively antagonize vitamin K and interfere with the production of prothrombin (II) and factors VII, IX, and X. Vitamin K, in the form of phytonadione, is used to treat excessive hypoprothrombinemia caused by warfarin toxicity. It is ineffective in reversing the hypoprothrombinemia caused by severe liver disease.
In patients with severe hepatic disease, a deficiency of clotting factors can result in a prolonged prothrombin time without therapy. Vitamin K is administered to these patients, and their prothrombin time is measured before surgery. Other measures to help clotting include fresh frozen plasma or platelets.
SELECTED MINERALS
Table 12-3 lists the ESADDIs of selected minerals. Less information is available on minerals than on vitamins; selected minerals are discussed: iron, zinc, and calcium. Figure 12-4 provides examples of good sources of these minerals.

FIGURE 12-4 Examples of foods containing the minerals iron, zinc, and calcium. Iron is found in oysters, red meats, and egg yolks. Meat and seafood are the best sources of zinc, whereas dairy products, such as milk and cheese, and shrimp are rich in calcium. (Copyright 2009 Jupiterimages Corporation.)
Iron
Although iron (Fe) is widely distributed throughout the human body, it is principally found as hemoglobin. Approximately 80% of the iron in the body is functional or “essential” iron (e.g., in hemoglobin [70%] and myoglobin [10%]), whereas 10% to 20% remains as storage or “nonessential” iron (in ferritin and hemosiderin).
 SOURCE
SOURCE
Good sources of iron include organ meats such as liver and heart, wheat germ, brewer’s yeast, egg yolks, oysters, red meats, and dried beans. Breads, flours, and cereals are commonly enriched with iron. Cooking utensils made of iron can raise the iron content of foods if the foods prepared in them are acidic. The percentage of iron absorbed from foods varies considerably, with absorption from meats being better. Numerous factors affect absorption, such as other foods eaten concomitantly, the bulk in the diet, the size of the dose of iron, the body’s need, and the presence of achlorhydria. The H2-receptor antagonists (H2-RA) and proton pump inhibitors (PPIs) can reduce the absorption of iron. If iron supplements are taken, they should be ingested at different times than the intake of these agents that increase pH.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The body carefully conserves its iron, and there is no mechanism for its excretion. Excess ingestion of iron over a long period may produce iron toxicity. The iron level is regulated by limiting its absorption from the intestinal tract. However, because iron is contained in each cell, when body cells are lost, iron is also lost. Women must replace the extra iron lost during menstruation. The RDA for iron is about 8 mg of iron per day for males and about 8 to 27 mg of iron daily for females to replace loss. Replacement of a donated pint of blood requires an additional 0.7 mg of absorbed iron daily for 1 year. Two percent to 10% of ingested iron is absorbed, and therefore men need about 1 mg of absorbed iron and women about 2 mg of absorbed iron each day. A pregnant woman’s need for iron cannot be met by the usual American diet or the 18-mg supplement recommended for women. For this reason, 27 mg of iron is recommended for pregnant women. Of course, if they have iron deficiency anemia, the requirement is higher.
 ROLE
ROLE
The basic function of iron is to allow the movement of oxygen and carbon dioxide from one tissue to another. Iron accomplishes this task by being a part of both hemoglobin and myoglobin. Iron is also a component of enzymes involved in the uptake and release of oxygen and carbon dioxide, and therefore it is essential for protein metabolism.
 DEFICIENCY
DEFICIENCY
Because the body is so efficient in conserving iron, a deficiency can occur only with growth, blood loss, or inadequate intake during pregnancy or lactation. The requirements of younger women cannot easily be reached without a supplement. Preschool children, adolescents, and elderly persons are also often found to be deficient in iron, probably because of inadequate intake.
Iron deficiency produces microcytic and hypochromic anemia. The symptoms are nonspecific but include pallor, irritability, fatigue, decreased resistance to infection, and sore mouth. Anemia, a decrease in the quality or quantity of red blood cells, can be measured by laboratory tests.
Before treatment, patients with iron deficiency anemia should be checked for any sources of bleeding to rule out chronic problems such as colon cancer. After ruling out problems, iron deficiency is treated by the concurrent administration of adequate iron salt, usually in tablet form. Ascorbic acid, which increases the absorption of iron may be used concomitantly (e.g., take iron tablets with orange juice). When the hemoglobin becomes normal, which may require months, there is no reason to continue therapy if the diet has improved or the cause of the deficiency has been removed (e.g., by control of excessive bleeding).
Although iron has many salt forms, no product has been shown to be superior to another, but the dose of elemental iron must be calculated based on the specific salt. Because ferrous sulfate (FER-us SUL-fate) contains about 30% iron, 200-mg tablets contain about 60 mg of iron. If side effects occur, ferrous gluconate (FER-us GLOO-koe-nate) may be substituted, but the 300-mg (12% iron) tablets contain about 36 mg of iron. (Of course these will have fewer side effects because they have less iron.) Other preparations, including sustained-release products, have no advantage and in some cases may actually be less effective than ferrous sulfate. The actual amount of iron needed can be calculated based on the results of a laboratory test.
 TOXICITY
TOXICITY
Complaints of gastrointestinal distress are common, even with therapeutic doses of iron. However, with an acute overdose, bleeding into the intestine can occur, resulting in shock or even death. Poisoning of children with iron has occurred and iron products, like all medications, should be placed out of the reach of children. Treatment of an acute overdose of iron involves removing the iron by gastric lavage and introducing phosphate into the stomach to decrease the iron’s solubility. Chelating agents, such as deferoxamine, which form a complex with iron, can be used if warranted.
With prolonged administration of iron, the intestinal mucosa, which normally regulates iron absorption, can be overcome. When an excess of iron accumulates, it produces hemochromatosis, a deposition of hemosiderin, in the organs. An iron overload can also occur with frequent blood transfusions. (Hemoglobin in blood is released, and the iron is conserved within the body.) As with the treatment of acute overdose, chelating agents can be used to treat chronic toxic effects from iron. Removing a pint of blood from the patient weekly can also be used to remove iron from the body.
Zinc
Zinc has only recently been recognized as a mineral the body requires.
 SOURCE
SOURCE
The best sources of zinc are seafood and meat. Cereals and legumes also contain zinc, but it is more poorly absorbed from these foods because of the presence of phytic acid, which interferes with intestinal absorption.
 ROLE
ROLE
Zinc is required to transport carbon dioxide in the blood and eliminate it in the lungs. It is essential in the utilization of alcohol, and it rids the body of lactic acid formed during exercise. It is also a component of insulin.
At least 59 enzymes involved in digestion or metabolism contain zinc or need it to function. Zinc plays an integral part in some enzymatic reactions and is a catalyst for others.
 DEFICIENCY
DEFICIENCY
Only in 1961 was zinc deficiency recognized in humans. A delay in sexual maturity, slow healing of wounds, and slowed growth are associated with this deficiency. In zinc-deficient rats, fetuses were either resorbed or born with congenital malformations. In humans, zinc-deficient mothers gave birth to low-birth-weight infants or infants with suggested malformations of the central nervous system. Both sexes have shown retarded gonadal development, and with severe deficiency, reproduction is impossible. In view of the drop in serum zinc produced by the oral contraceptives, speculation concerning subsequent pregnancies would be natural. A deficiency of zinc also stunts growth.
Hypogeusia, anorexia, and hyposmia have been reported in conjunction with zinc deficiency. Zinc seems to be essential to the growth and differentiation of the taste buds. It may also be a part of a “growth factor” for taste buds, appropriately named gustin. It may soon become routine to measure the zinc in saliva to correlate it with body zinc levels.
 TOXICITY
TOXICITY
Long-term studies must be conducted to determine the effect of chronic zinc toxicity. Excessive intake of zinc has impaired the lymphocyte and polymorphonuclear leukocyte functions in healthy persons. Nausea, vomiting, fever, and diarrhea have been reported to follow acute ingestion.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
Although it has long been known that zinc participates in wound healing, there is no known advantage to the administration of zinc in patients who have no zinc deficiency. The use of zinc supplements to promote wound healing is currently being studied for periodontal surgery. Zinc has also been used, without documented evidence, to treat acne, arthritis, and Wilson’s disease. Zinc gluconate lozenges have been shown to reduce the duration of the common cold. Zinc lozenges are available OTC.
Calcium
Calcium, the fifth most prevalent element in the body, is present in bones, teeth, and extracellular fluids. The level of calcium in the serum must be maintained within a narrow concentration to prevent serious problems.
 SOURCE
SOURCE
Dairy products are the best source of calcium in the diet. These include milk, cheese, yogurt, and cottage cheese. Other good sources of calcium are sardines with bones, tofu, and shrimp.
 RECOMMENDED DIETARY ALLOWANCE
RECOMMENDED DIETARY ALLOWANCE
The adequate daily dietary intake of calcium is from 1000 to 1300 mg for the adult. Pregnant and nursing women require 1000 to 1300 mg daily, depending on age. The recommended intake of calcium in postmenopausal women is 1200 mg per day. Postmenopausal women who add estrogen replacement therapy reduce their chance of exhibiting osteoporosis. Weight-bearing exercise also helps to prevent osteoporosis. Adequate vitamin D intake should be evaluated. Before a patient ingests exogenous calcium, the patient’s diet should be evaluated to determine the baseline intake of calcium from food. Supplemental calcium should be taken to adjust the intake of calcium to 1500 mg. For example, if a diet analysis finds that the patient is taking 600 mg of calcium (two glasses of skim milk), then the calcium supplementation should be 900 mg for the postmenopausal woman.
 ROLE
ROLE
Calcium is essential for the function of the nervous, muscular, and skeletal systems and for cell membrane and capillary permeability. It is needed for skeletal muscle contraction, cardiac function, renal function, membrane integrity, and blood coagulation. The skeleton is a reservoir of calcium for the body. Parathyroid hormone, calcitonin, and vitamin D regulate calcium concentrations in the body.
 DEFICIENCY
DEFICIENCY
A deficiency of calcium can occur when both calcium and vitamin D are withheld. Mobilization from the bone keeps tissue levels nearly normal. If levels in the blood fall, tetany, paresthesias, muscle cramps, and convulsions can result.
 ADVERSE REACTIONS
ADVERSE REACTIONS
Oral calcium may be irritating to the gastrointestinal tract. It may cause constipation. Hypercalcemia may result if large doses of calcium are given to patients with chronic renal failure. Calcium can complex with tetracycline and the quinolones and inactivate them. Their oral administration should be separated by at least 2 hours.
 CLINICAL CONSIDERATIONS
CLINICAL CONSIDERATIONS
Calcium is used to treat a deficiency of calcium and secondary to low calcium levels. When calculating the RDA of calcium, the amount of elemental calcium, not the total weight of the calcium salt, must be used. For example, 1250 mg of calcium carbonate provides 500 mg of elemental calcium (40% Ca), whereas 500 mg of calcium gluconate provides 45 mg of elemental calcium (9%). So calcium gluconate has 90 mg of elemental calcium per gram, and calcium carbonate has 400 mg of elemental calcium per gram. Before choosing a calcium supplement, the labels of each product must be carefully compared. It is interesting that on the label of one bottle of calcium the weight of salt (1250 mg Ca [CO3]2) is printed in BIG letters and the weight of the elemental calcium (Ca) is in small letters.
Calcium may be used parenterally to elevate the serum calcium in an emergency. It is also used during cardiopulmonary resuscitation, in the treatment of hyperkalemia with secondary cardiac toxicity, and to treat hypermagnesemia.
DRUG-INDUCED VITAMIN DEFICIENCIES
Drugs from a large variety of drug groups have the ability to produce vitamin deficiency. Some actually produce a deficiency, whereas others tend to lower the levels of some vitamins. Table 12-4 lists drugs and the vitamin deficiencies they produce.
TABLE 12-4
DRUG-INDUCED VITAMIN DEFICIENCIES
| Drug(s) | Potential Deficiency |
| Methotrexate (MTX) | Folic acid |
| Isoniazid (INH) | Pyridoxine (B6) |
| Sulfasalazine | Folic acid |
| Trimethoprim | Folic acid |
| Anticonvulsants | Vitamin D |
| Phenytoin | Folic acid |
| Birth control pills (oral contraceptives) | Vitamin A; B vitamins: thiamine (B1), riboflavin (B2), pyridoxine (B6), folic acid, and vitamin B12; vitamins C and E |
| Levodopa | Pyridoxine (vitamin B6) |
| Smoking | Vitamin C |
| Colchicine | Vitamin B12 |
| Alcohol | Vitamin B12 |
INH, a drug used for management of tuberculosis, can produce a neuropathy resulting from vitamin B6 deficiency, so patients taking INH are given concomitant vitamin B6. Patients taking the anticonvulsant phenytoin (Dilantin) may exhibit vitamin D deficiency because phenytoin stimulates the liver microsomal enzymes, resulting in an increase in vitamin D metabolism and a decrease in its blood levels. It may be necessary to give vitamin D to patients taking phenytoin. Folic acid levels may fall in patients taking phenytoin, but folic acid supplementation may lower the blood level of the anticonvulsant, requiring an increase in its dose.
Certain drugs, such as oral contraceptives, tend to induce a deficiency of vitamins B1 and B2 and folic acid. The exact mechanism by which these vitamin deficiencies occur is not known, but an interference with absorption of the vitamin is a postulate. Drugs that produce a folic acid deficiency include methotrexate, sulfonamides, and triamterene. The anticonvulsants phenytoin, valproic acid, and phenobarbital are associated with folic acid deficiency.
CLINICAL SKILLS ASSESSMENT
1. What are some sources of vitamin C?
2. Are there any toxicities to vitamin C and if so, what are they?
3. What are the clinical uses of vitamin C?
5. How could vitamin C deficiency be treated?
6. Can any drugs cause a vitamin C deficiency? If so, what are they?
7. Why were the recommended dietary allowances developed?
8. Compare and contrast fat-soluble and water-soluble vitamins.
9. What are the toxicities and deficiencies of vitamin A?
10. Who is at risk for vitamin D deficiency and what are its signs and symptoms?
11. What are the sources of vitamin K and what are its signs and symptoms of deficiency?
12. What are the B vitamins and who is at risk for vitamin B deficiency?
13. What are the oral manifestations of vitamin B deficiency?
14. What are choline and inositol and what is their function?
15. What are sources of calcium and who is at risk for deficiency? Include signs and symptoms of deficiency.
16. What is zinc and what is its relevance to maintaining a healthy diet and body?
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
*Data from Miller ER III, Pastor-Barriuso R, Dalal D, et al: Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality, Ann Intern Med 142(1):37, 2004.