Chapter 29 Viral hepatitis
A clear understanding of viral hepatitis is essential for all dental practitioners, particularly in view of the serious sequelae of the disease and the potential of transmitting the infection in the dental clinic. Hepatitis can be due to a number of causes, such as infections, alcohol abuse, trauma or drug-induced toxicity. However, in global terms, viral infections are by far the single most important agent of hepatitis. These include infections with herpes simplex virus, cytomegalovirus and Epstein–Barr virus, but the vast majority of viral liver diseases are one of the following:
These may be classified into two groups depending on the viral transmission route:
Data from the World Health Organization (WHO) indicate that viral hepatitis B infection alone accounts for more than 1 million deaths worldwide. In terms of morbidity, there are around 350 million hepatitis B chronic carriers and another 100 million chronic carriers of hepatitis C.
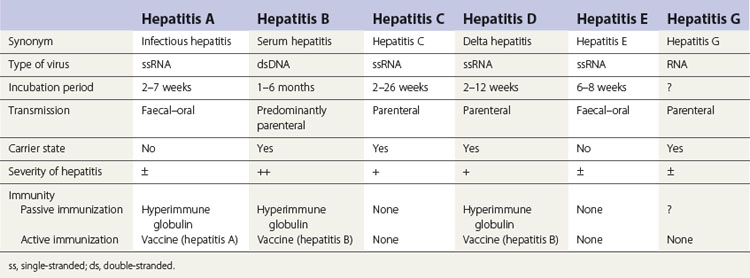
The various types of viral hepatitis differ in severity of infection, morbidity, mortality rate, presence or absence of a carrier state and frequency of long-term sequelae such as cirrhosis and cancer. The main differences between the hepatitides caused by these viruses are shown in Table 29.1.
Signs and symptoms of hepatitis
The common symptoms and signs of hepatitis include malaise, jaundice, dark urine and pale, fatty stools. These, together with results of serum and urine biochemistry and specific serology tests, facilitate the diagnosis of viral hepatitis. Investigation typically reveals abnormal liver function with raised levels of serum transaminases and bilirubin, and bilirubinuria. Specific serological tests are now available to detect hepatitis A, B, C, D and E antibodies.
Hepatitis A
The hepatitis A virus (HAV) is a small (27 nm) RNA virus belonging to the picornavirus group (which also includes poliovirus and coxsackieviruses). The virus is inactivated by ultraviolet light, exposure to water at 100°C for 5 min and by exposure to 2% glutaraldehyde for 15 min.
Epidemiology
Hepatitis A commonly occurs in developing parts of the world where sewage disposal measures and food hygiene are unsatisfactory. Only 10–13% of the population in developed countries has been exposed to the virus by the age of 20 years. It is usually contracted by the faecal–oral route from contaminated food and water. Children and young people are most often infected, and for this reason, a history of hepatitis in childhood would, in most instances, be indicative of a hepatitis A infection.
Clinical features
The mean incubation period is 30 days (range 2–7 weeks). Patients are infectious before the onset of symptoms during the prodromal phase and just before the onset of clinical disease.
Jaundice is common in adults and rare in young children. There are no chronic sequelae. Some patients continue to excrete HAV in faeces during weeks 1–3 of the illness, and HAV may also be present in saliva (100 particles per millilitre) throughout this period.
Diagnosis
Diagnosis is by demonstration of HAV antigen in faeces. Serological tests demonstrate immunoglobulin M (IgM) class anti-HAV antibodies in serum during the acute or early convalescent phase (IgG class antibodies appear later in the disease and confer enduring protection against the disease).
Unlike hepatitis B, there is no carrier state associated with the disease. This, together with its faecal–oral transmission, implies that hepatitis A transmission in the dental clinic is highly unlikely.
Prophylaxis
Passive immunization by hyperimmune globulin is effective against clinical illness, particularly when administered in the early incubation period. However, the main use of short-term, pre-exposure prophylaxis is for travellers to hepatitis A-endemic areas, such as some parts of the developing world. Several vaccines of inactivated HAV produced in human cell culture are available. Immunization (two doses: initial and a booster 6–12 months after) is safe and effective and recommended for professionals working with institutionalized patients. A combined vaccine for hepatitis A and B is now available.
Hepatitis A and dentistry
HAV is not a significant infection risk in dentistry as the route of transmission is faecal–oral. Close contact with saliva may transmit infection as saliva can contain some HAV. Rarely infection has been transmitted by needlestick injury, and there is a report of transmission from a surgeon to a patient. Standard infection control measures are adequate to prevent transmission in dental practice.
Hepatitis B
The hepatitis B virus (HBV) is a DNA hepadnavirus (hepa: liver + DNA), which is structurally and immunologically complex. Electron microscopy of HBV reveals three distinct particles (Fig. 29.1):
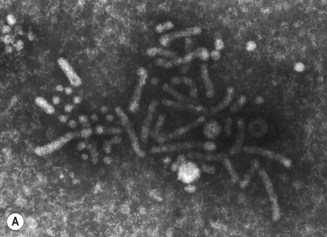
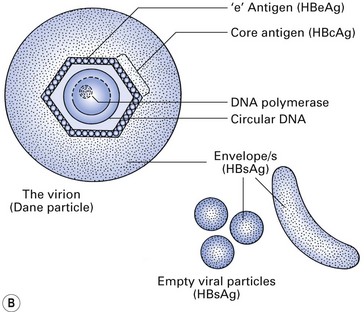
Fig. 29.1 Hepatitis B virus. (A) Scanning electron micrograph; (B) hepatitis B virus and particles. HBsAg, hepatitis B surface antigen.
Being a hepatotropic virus, HBV will reside and multiply in hepatocytes after entering the body, and cause hepatic injury and inflammation (hepatitis) to varying degrees. When it multiplies in the hepatocytes, for some unknown reason, the virus particles described above are produced in different proportions within the liver cell cytoplasm. As a result of overproduction of these non-infectious spheres and tubules, which are the surface proteins of the virus (hence called hepatitis B surface antigens or HBsAgs), they circulate freely in the serum for prolonged periods after the acute hepatitis episode.
The central core of the HBV consists of a single-stranded DNA, an enzyme (DNA polymerase) and a core antigen (HBcAg). Although this antigen is rarely found in the serum, a breakdown product of HBcAg, termed hepatitis B ‘e’ antigen (HBeAg), may be found in the serum and is a marker of active infection.
Epidemiology
The prevalence of hepatitis B varies greatly in different parts of the world: it is higher in African and Asian countries than in the Americas, Australia and western Europe (Fig. 29.2); in urban than in rural areas; and in men than in women. In developed countries, the risk of exposure to hepatitis B is high in certain categories of people, as shown in Table 29.2. Several variants of HBV are now known, and when these involve rearrangement of the surface antigens, existing vaccines may not be protective. This has come to light as a few individuals who had been successfully immunized against HBV but who were at high risk of infection nevertheless contracted hepatitis B. A variant HBV, HBV-2, has been described in West Africa, the Middle East, Spain, France, Taiwan, New Zealand and the USA, and another has been reported from Italy, Greece and the UK. Both variants are able to infect persons immunized against the usual form of HBV.
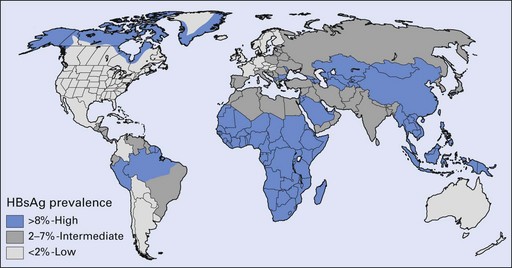
Fig. 29.2 Geographic distribution of chronic hepatitis B virus infection. HBsAg, hepatitis B surface antigen.
(Courtesy of the Centers for Disease Control and Prevention, USA.)
Table 29.2 Hepatitis B high-risk population groups
Carrier state and identification of carriers
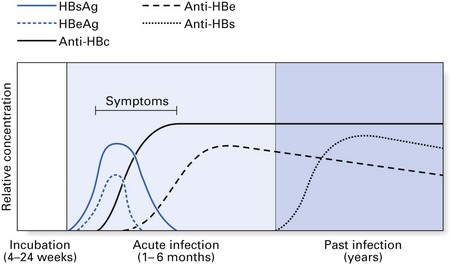
Most patients who contract hepatitis B recover within a few weeks without any sequelae (Fig. 29.3). However, serological markers of previous HBV infection are invariably present in these patients for prolonged periods. Such markers take the form of antibodies to various components of the HBV. A minority (2–5%) fail to clear HBV by 6–9 months and consequently develop a chronic carrier state. This state more frequently follows anicteric HBV infection (i.e. infection without jaundice). The converse of this is that a majority of infections that lead to jaundice resolve without a carrier state; hence, a history of jaundice in a patient in most instances indicates little or no risk in terms of hepatitis B transmission.
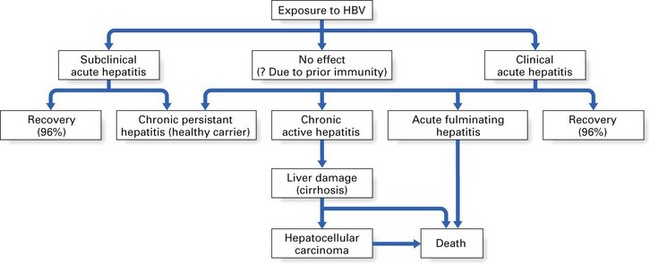
Fig. 29.3 Possible sequelae of exposure to hepatitis B virus (HBV). Values in parentheses indicate percentage recovery.
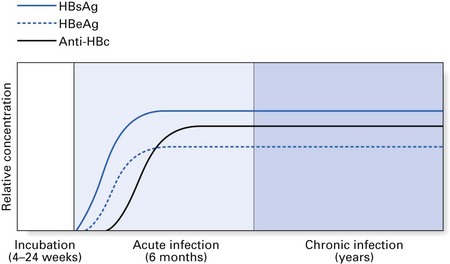
The chronic carriers of hepatitis B infection fall into two main groups: those with chronic persistent hepatitis (the so-called ‘healthy carrier’ state) and those with chronic active hepatitis (Fig. 29.3). In chronic persistent hepatitis, the patient does not develop liver damage and is generally in good health, although the liver cells persistently produce viral antigen (HBsAg) because of the integration of the viral genome into the DNA of the hepatocytes. The second group of chronic carriers are extremely infectious as they harbour the infective Dane particles in their blood. In addition, they are very susceptible to cirrhosis and hepatocellular carcinoma. Nonetheless, the chronic active hepatitis group represents a small minority of hepatitis B patients. In general, infection with HBV leads to complete recovery in most individuals, while only about 2–5% develop a carrier state. These two disease states elicit characteristic serological profiles in the affected individual during various phases of the disease, as shown in Figures 29.4 and 29.5.
Diagnosis and serological markers
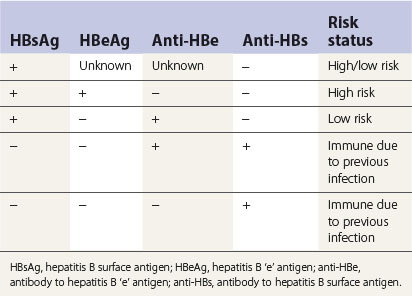
Diagnosis of HBV is complicated by the variety of serological markers and the complex sequelae of the disease itself. Table 29.3 summarizes the significance of the serological markers described below:
Hepatitis B and dentistry
More than 400 health care workers, including dental surgeons, have been infected with hepatitis B in clinical settings. Most were surgeons; in dentistry, the risk of infection is greater among oral surgeons and periodontists than among general dental practitioners. Standard infection control procedures were often lacking when transmission occurred.
The number of health care workers contracting infection reported since the introduction of the vaccine programme in 1987, especially in dentistry, has been small. However, there is an ever-present danger of hepatitis B transmission in dentistry if dental personnel are not vaccinated, or are vaccinated but with inadequate seroconversion (see below).
Although the usual mode of transmission of hepatitis B is from the patient to the dentist, there are at least eight recorded outbreaks where dentists have transmitted the disease to patients.
Intraorally, the greatest concentration of HBV is at the gingival sulcus as a result of the continuous serum exudate, which is small in healthy people but greatly increased in diseased states, e.g. periodontitis; the virus is present in mixed saliva but not in parotid or submandibular saliva (Table 29.4).
Table 29.4 Concentration of hepatitis B in body fluids
| High | Moderate | Low/undetectable |
|---|---|---|
| Blood | Mixed saliva | Urine |
| Wound exudates | Semen | Sweat, tears |
| Vaginal fluid | Breast milk | |
| Parotid/submandibular saliva |
Special precautions are not necessary when treating carriers of hepatitis B (or any other disease), as standard infection control measures, routinely employed in dentistry irrespective of the clinical status of the patient (see Chapter 36), should prevent disease transmission.
Hepatitis C
Some years ago, the term ‘non-A non-B hepatitis’ (NANBH) was used to describe a disease complex with probable infective origin, that did not belong to either hepatitis A or hepatitis B. Subsequent research demonstrated that NANBH is due to infective agents transmitted by both the parenteral and the enteric route. One such parenterally transmitted agent was named ‘hepatitis C virus’ (HCV) and another, enterically transmitted, NANBH was termed ‘hepatitis E virus’ (HEV).
Aetiology
Hepatitis C is caused by an enveloped RNA virus related to the flaviviruses. The virus has yet to be grown in culture or visualized ultrastructurally. It may exist as one of at least six different genotypes. Some patients may be infected with more than one genotype. The viral RNA can remain intact for at least 7 days at room temperature. Thus, although the infectivity of HCV is still unclear, it is essential that adherence to standard infection control is observed at all times.
Epidemiology
Hepatitis C is globally prevalent. According to WHO, about 3% of the world population has been infected with hepatitis C and there are more than 170 million chronic carriers at risk of developing liver cirrhosis or cancer. There may, however, be considerable regional and ethnic group variation (Fig. 29.6).
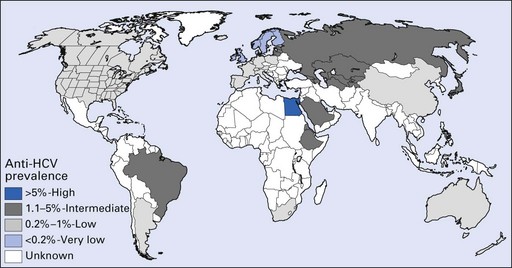
Fig. 29.6 Prevalence of hepatitis C virus (HCV) infection among blood donors.
(Courtesy of the Centers for Disease Control and Prevention, USA.)
Blood, blood products, intravenous immunoglobulins and donated organs have transmitted HCV, although newer methods of HCV detection have reduced but not entirely eradicated such risk. Injecting drug abusers, transfusion recipients and haemophiliac patients receiving blood products are other groups who are at risk. The disease occurs in 5–10% of transfusion recipients, leading to chronic hepatitis in about half of them.
Diagnosis
The diagnosis of HCV infection is serological. Assays using the enzyme-linked immunosorbent assay (ELISA) technique can detect antibodies to HCV envelope or core proteins. Polymerase chain reaction (PCR) assays are also very sensitive and specific and can detect early infection. Most HCV-infected persons are HCV-seropositive within 6 months of infection. Because of this delay in antibody response, donated blood may not be screened effectively.
Clinical features
Sequelae of chronic HCV infection
Persistent chronic infection develops in approximately 80% of infected persons and the course of infection may run for 20 or more years. Approximately 70% of those with chronic HCV will develop chronic liver disease. The virus may also cause mixed cryoglobulinaemia, thyroid disorders, diabetes mellitus and thrombocytopenia.
Treatment
Infection can be managed with interferon-alpha, aciclovir and ribavirin. These, in essence, attempt to clear the virus and the viraemia and reduce the risk or slow down the development of liver sequelae. Interferon is moderately effective, with reported success rates varying from 15% to 50%.
Prevention
At present, there is no passive or active immunization programme for HCV infection. All immunization methods appear to be unsatisfactory as re-exposure of HCV-infected patients to different strains of HCV still results in reinfection. This reflects the possible different subtypes of HCV and their rapid rate of mutation. By the same token, prophylaxis with immunoglobulins confers little, if any, immunity.
Hepatitis C and dentistry
Hepatitis D (delta hepatitis)
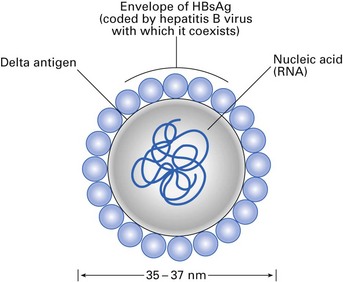
Delta hepatitis is caused by a ‘defective’ RNA virus, which coexists with HBV (Fig. 29.7). Hepatitis D virus (HDV) is the smallest animal virus known and contains a nucleoprotein, a delta antigen and an outer surface protein. The outer coat of the delta virus is ‘borrowed’ HBsAg and hence the virus cannot survive independently without the hepatitis B viral particles. Consequently, delta infection is only seen as a:
Both usually cause an episode of acute hepatitis. Co-infection usually resolves, while superinfection frequently causes chronic delta infection, leading to chronic active hepatitis (Fig. 29.8).
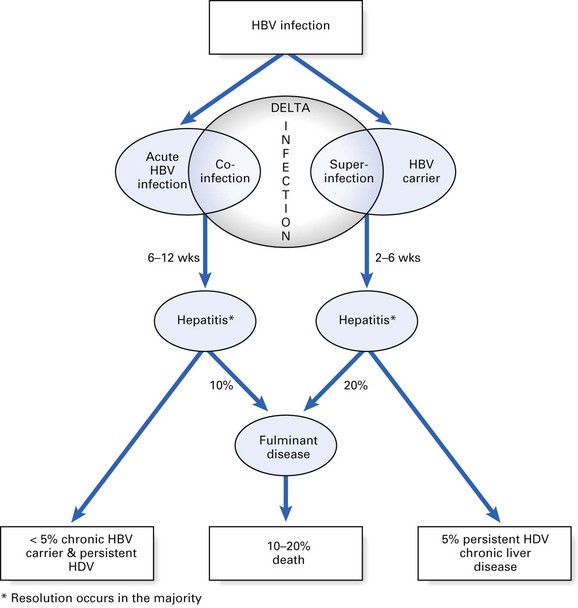
Fig. 29.8 Sequelae of hepatitis delta virus (HDV) infection. HBV, hepatitis B virus. *Resolution occurs in the majority.
Epidemiology
It has been estimated that about 15 million persons are infected with HDV worldwide, as about 5% of HBV carriers are HDV-positive. In non-endemic areas such as the USA and northern Europe, HDV is mainly confined to persons frequently exposed to blood and blood products, particularly drug addicts. Up to 4% of US blood donors have evidence of previous HDV infection. It is noteworthy that HDV infection is not common in most groups in South-East Asia. Geographic areas with a high incidence of delta hepatitis are the Amazon basin, parts of Africa, the Middle East and Arab countries, where 30–90% HBsAg carriers with liver disease are infected. Delta infection occurs rarely in the susceptible population of northern Europe and is virtually confined to parenteral drug abusers.
Routes of delta transmission appear to be similar to those of hepatitis B, the infection being most commonly seen among persons at high risk of acquiring hepatitis B infection (see Table 29.2). The transmission and epidemiology of HDV infection are much the same. In general, it is a parenterally transmitted infection, which has become a major problem in injecting drug abusers. It is also transmitted by sexual or close contact with HDV-infected persons. However, sexual transmission of HDV appears to be less common than for HBV, and HDV infection is uncommon in men who have sex with men.
Clinical features and diagnosis
The incubation period of HDV infection ranges from 2 to 12 weeks, and most infections lead to jaundice. The virus produces acute hepatitis, which usually resolves but may precipitate fulminant liver disease. The latter is 10 times more frequent in HDV infection than in HBV infection alone. Chronic hepatitis is a common sequela of HDV infection, and 70% of those affected develop cirrhosis. The role, if any, of HDV in hepatic carcinogenesis is unclear.
Diagnosis is by detection of delta antigen (using ELISA) in serum and/or by the appearance of delta antibody. Delta infection does not respond well to interferon therapy.
Prophylaxis
As the delta virus is dependent on HBV for replication, successful immunization with the hepatitis B vaccine will prevent delta infection.
Hepatitis D and dentistry
The main route of HDV transmission is parenteral, in either blood or blood products. Sexual transmission may occur sometimes within households, and perinatally if mothers are positive for HDV and HBeAg. It is unclear whether saliva is a vehicle.
There is at least one report of HDV transmission in dentistry in the USA, where up to 700 cases were recorded. At least four dentists were infected; one oral surgeon became an HBV carrier and was thought to have infected several patients.
Hepatitis E
HEV is a relatively newly described RNA virus that bears some similarities to the Caliciviridae. Transmission is via the faecal–oral route, by ingesting contaminated drinking water. Hepatitis E outbreaks are common in Africa, Asia and Latin America, especially in countries with poor sewage disposal facilities. In these geographic regions, different HEV viruses are responsible for the infection. Intrafamilial and parenteral spread is rare. In most instances, the disease follows a benign pattern like that of hepatitis A, with a low mortality rate of 1–2%. The infection is infrequently associated with fulminant hepatitis. The disease can be diagnosed by Western blot, ELISA and a PCR assay.
Due to its mode of transmission, the virus does not pose a major risk of cross infection in dentistry.
Hepatitis F?
In 1994, an investigator reported finding viral particles in the stool of post-transfusion, non-A, non-B, non-C, non-E hepatitis cases. Injection of these particles into Indian Rhesus monkeys presumably caused hepatitis, named ‘hepatitis F’. However, no other investigator has been able to confirm these findings, and the original observation is now thought to be incidental.
Thus, there is no hepatitis F virus as yet. Unfortunately, though, this alphabetic position in the viral nomenclature has been occupied.
Hepatitis G
It has become increasingly evident that there are patients with acute or chronic hepatitis who are not infected by the hepatitis viruses A–E described above (hence the designation non-A–E hepatitis). Another hepatitis agent isolated in 1967 from a surgeon (whose initials were GB) with acute hepatitis has been transmitted in tamarins. This particular virus, first termed the GB agent, was thought to be two novel RNA viruses of the Flaviviridae family, and were designated hepatitis GB virus A (GBV-A) and hepatitis GB virus B (GBV-B). To add to the confusion, other closely related viruses, hepatitis GBV-C and hepatitis G virus (HGV), have been found in humans with chronic hepatitis and recovered from patients with non-A–E hepatitis. All these viruses appear to be identical and fall under the common term ‘hepatitis G’, at least for the time being.
Infections with these viruses appear more common among injecting drug abusers and people with haemophilia. HGV does not seem to elicit a strong immune response and indeed no cases have symptoms like the other hepatitis viruses. Carrier rate (in the US) is between 2–5% in the general population.
Hepatitis G and dentistry
HGV RNA is present in whole saliva of infected individuals, but transmission through this route has not been determined. No data are available on the transmission of hepatitis G or the rate of HGV carriage in dental staff.
No vaccine is available; implementation of standard infection control measures should be adequate to prevent transmission of this virus in dentistry.
Transfusion-transmitted virus and SEN viruses
Both these viruses produce post-transfusion hepatitis. Described in 1997, transfusion-transmitted virus (TTV) is a non-enveloped, single-stranded DNA virus possibly belonging to the Parvoviridae family. It has been isolated from persons in the UK, Japan and Brazil, especially older blood donors. The most remarkable feature of TTV is the extraordinarily high prevalence of chronic viraemia in apparently healthy people – up to nearly 100% in some countries. It may be transmitted parenterally, but this route has not been confirmed.
SEN viruses (SEN D and SEN H), so called after the initials of the patient from whom the virus was isolated, are recently identified single-stranded, circular DNA viruses. They are strongly associated with transfusion-related non-A–E hepatitis. The vast majority of SEN virus-infected recipients did not develop hepatitis. No information on either TTV or SEN virus salivary carriage or transmission in dental settings is available.
Key facts
Bendinelli M., Pistello M., Maggi F., et al. Molecular properties, biology, and clinical implications of TTvirus, a recently identified widespread infectious agent of humans. Clinical Microbiology Reviews. 2001;14:98-104.
Karaylannis P., Thomas H. Hepatitis G virus: Identification, prevalence and unanswered questions. Gut. 1997;40:294-296.
Klein R.S., Freeman K., Taylor P.E., et al. Occupational risk of hepatitis C virus infection among New York City dentists. Lancet. 1999;338:1539-1542.
Scully C., Samaranayake L.P. Clinical virology in oral medicine and dentistry. Cambridge: Cambridge University Press; 1992.
Zuckerman A.J., Harrison T.J. Hepatitis viruses. In Zuckerman A.J., Banatvala J.E., Pattison J.R., editors: Principles and practice of clinical virology, 3rd ed., Chichester: John Wiley, 1994. Ch. 2
Review questions (answers on p. 354)
Please indicate which answers are true, and which are false.