CHAPTER 6 Dental physiology
Introduction
This chapter focuses on how the masticatory system manages ingested feed and prepares it for digestion. There is a large amount of published data on the effect of chewing in the ruminant and other herbivorous species.1 In the former case, fermentation within the rumen does not significantly reduce particle size, and in the horse there is no significant reduction in ingesta particle size between stomach and anus, leaving mastication as the most important determinant in feed particle size reduction.
The morphology of equid teeth and their role in effectiveness of mastication have not been widely studied. Human studies have used molar occlusal surface area (OSA) as an indicator of chewing effectiveness. The influence of this variable on a variety of nutritional parameters has been studied in red deer,2 where researchers found that animals with low OSA had a greater mean fecal particle size, but no difference in the mean retention time of gastrointestinal contents was found between groups. Additionally, animals in the low OSA group had a greater mean number of chews per gram of dry matter than high OSA animals.
Because the primary masticatory movement of herbivorous animals is a lateral translocation of the mandible, the enamel ridge perimeter distance (termed ‘length of occlusal enamel edge’, ERPD) has been used to assess mastication in some studies. This measurement has been used in studies of Nubian ibex, red deer, possums, gliders, koalas, and horses.2–7 In koalas, ERPD was found to be directly related to chewing effectiveness.6
Only recently has similar attention turned to studies of dental physiology and masticatory biomechanics of the horse. This revival has been driven by the fact that a greater number of horses are currently undergoing oral examinations on a routine basis, the findings of which are querying the etiopathogenesis of common dental malocclusions. The absence of dental malocclusions or dental abnormalities should, in theory, result in a normal masticatory cycle, which would generate the appropriate forces on the occlusal surfaces of teeth for effective mastication, as well as resulting in sufficient even wear of the entire occlusal surfaces of all teeth, thus preventing overgrowth. However, the patterns of jaw movement (masticatory biomechanics) are the result of a complex interrelationship between food consistency, particle size, and neural control of mastication.8
Functional morphology
Thomas Henry Huxley, an ardent evolutionist, gave a public lecture in New York City on September 22 1876 entitled ‘The demonstrative evidence of evolution’ in which he used, as his subject matter, the evolution of the horse beginning with Eohippus (now termed Hyracotherium).9,10 This lecture was given in support of the newly published work, On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life by Charles Darwin.
The evolution of the modern horse (Equus caballus) from its Eocene ancestors was one of the first examples recognized to support the work of Charles Darwin. Two remarkable adaptive traits allowed the horse to take advantage of increasing areas of grassland: a rapid (by paleobiological standards) change in dental morphology in just over 1.5 million years (or 0.5 million generations) and secondly, the loss of extraneous digits in the support of a single, enlarged middle toe adapted for speed.
The basic mammalian dental formula is of 3 incisors, a canine, 4 premolars and 3 molar teeth. The significance of the cheek tooth arrangement is two-fold. Firstly, premolars have a deciduous (juvenile) counterpart, and secondly, they are generally smaller and less robust than the larger, permanent molars. Thus, the importance of the molarization (or the assimilation of molar tooth characteristics) of the 2nd – 4th equine premolars should not be underestimated. In the modern horse, the first premolar is vestigial and is the only cheek tooth not to have evolved hypsodonty and a complex root system in conjunction with the remaining teeth.
The environmental drive forcing the ancestors of the horse from browsing to grazing and to develop a greater body size, selected individuals with a different dentition than previously required. Grasses are high in abrasive silicates, which, in addition to Cope’s Law (stating that within a lineage there is a tendency or trend toward increasing body size), forced an increase in body size from the fox-terrier-sized Eohippus (estimated to be 3 hands, i.e., 12 inches tall ≈5 kg) to the modern 14–16 hand (56–68 inch tall, 500–1000 kg) horse. A doubling of body mass requires an eight-fold increase in the amount of ingested feed. Simply doubling the size of teeth would result in only a 4-fold increase in the surface area of the occlusal surface of the tooth, which would be inadequate to support these evolutionary changes.
Thus, evolutionary strategies to overcome these problems included an increase in the relative size of each tooth, a change in the type and complication of the relationship between dentin and enamel ridges, and an increase in crown height (hypsodonty). There was even a lineage (clade) of late Miocene horses (Pseudohipparion) that developed hypselodont (elodont) teeth (continually growing teeth, like those of rodents), but these horses were ultimately unsuccessful.
Ancestors to Eohippus (Phenacodus) had premolar and molar teeth formed like those of the pig or bear.11 They were broad and bore many separate conical cusps on the occlusal surface that evolved to deal with a varied diet of insects, fruits or vegetables. As with the modern horse, the lower teeth were narrower than the uppers; however, the surface variations were such that both sets of teeth meshed together when the jaws were closed. Microscopic wear patterns on these teeth have suggested that the predominant chewing motion was crushing (vertical), rather than the modern shearing (side-to-side) action.12 In Hyracotherium the anterior (towards the front) premolars of the upper jaw were still shaped as cutting blades and were triangular. The complete row of cheek teeth in this animal was no more than 10 cm in length in a mesial-distal (rostrocaudal) direction, approximately the same length as two cheek teeth of the modern Equus caballus.
A later horse, Orohippus, developed a sub-triangular-shaped second premolar (from its initial cutting blade appearance), but the fourth premolar was already four-cusped and quadrate (square). Epihippus had a squarer second premolar, but both the third and fourth premolars were now quadrate. The outer crest of the tooth (the ectoloph) became W-shaped at this stage and has remained so since. Mesohippus emerges at 40 million years, and all teeth except the first premolar are now molarized with the exception of the first premolar, which (if present) remains a unicuspid tooth to the present day.
Thus molarization, the first defining moment in equine dental evolution, was complete within 20 million years. However, these teeth were still brachydont (low crowned) and not able to withstand the rigors of animals wholly committed to grazing. Hypsodonty did not make a determined appearance in the fossil record until the evolution of Parahippus (23 million years) and its descendent Merychippus, at which time the predominant chewing direction was side-to-side with a wide stroke action ensuring efficient shearing forces applied over the chewing surface.13 Interestingly, research also indicates a gradual increase in crown height from Parahippus through Merychippus into the modern equine lineage and also into the extinct Hipparion group of horses.14
Enamel crests became increasingly convoluted, with the effect of increasing the surface area (or perimeter) of the enamel. These changes were similar in both upper and lower jaws, but the changes were less extreme in the lower teeth.15 The spaces between enamel ridges are filled by a softer material (dentin) which is preferentially worn away to create craters rimmed by sharp enamel edges over which the grass is sheared. In conjunction with the infolding (pleating) of enamel ridges, cementum appeared in late Parahippus and Merychippus animals.16 This material is softer than enamel but is firm, tough and less brittle. Cementum filled the areas around the edges of the brittle enamel crests providing support, which otherwise would have resulted in breakage of the enamel crests and thus negation of the hypsodont effect. Cement is also formed within the enamel infoldings (infundibula) of the maxillary cheek teeth as the teeth develop within the dental sac. Cement is also produced around the developing roots as they are formed. In this way, a cheek tooth with prolonged eruption is formed with a crown height (including reserve height) of at least twice its width. Such a tooth usually erupts at a rate equal to the rate of the wear of the crown by attrition; however, in some cases, where there is no opposing tooth, ‘super-eruption’ may occur.
Odontological evolution in the horse was thus a rapid, sustained event encompassing the molarization of premolar teeth, the development of hypsodonty and cemental protection, as well as amalgamation of enamel crests into linear occlusal ridges to increase efficacy for shearing coarse forage. These evolutionary advantages are thought to have allowed the horse to advance from a generalized browsing animal to a specialized grazer, thus enabling it to take advantage of the increasing grassland areas of the late Eocene, Oligocene, and Miocene eras. By the time of the arrival of Merychippus and the subsequent radiation (encompassing 19 species in the late Miocene), the dental revolution was effectively at an end. Some small variations in the degree of hypsodonty continued, notably in the form of Nannohippus; however, the dental pattern observed in the modern horse (Equus caballus) is essentially unchanged for 15 million years. (See Chapter 1 for more detail.)
Anatomy
Molarization of the cheek teeth of the horse, as with most herbivores, resulted in a row of 6 cheek teeth (not including the 1st premolar), which function as a single chewing unit. The integrity of this unit is maintained due to a combination of the initial caudal angulation of the clinical crown of premolars 2 (Triadan 06) and to a variable extent of the 07 and the rostral (mesial) angulation of the clinical crowns of molars 1 to 3 (Triadan 09–11), the ‘keystone effect’ of an almost vertical eruption of the 4th premolar (08, Fig. 6.1) and the rostral (mesial) drift of cheek teeth as the horse ages.
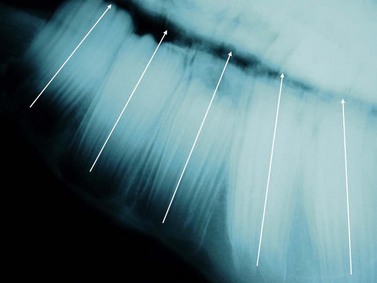
Fig. 6.1 A lateral radiograph of a young horse (4 years old) showing the relative angulation of the cheek teeth (lines).
The horse is anisognathic, i.e., the lower dental arcade is straighter and 23–30 % narrower than that of the upper arcade (Fig. 6.2), such that the maxillary teeth project laterally beyond the mandibular teeth (Fig. 6.3). There is a significant difference in morphology between maxillary and mandibular cheek teeth, with the former generally having a greater surface area and containing more enamel in the form of mesial and distal infundibula.17 Despite large differences in body weight realized by Equus callabus, measurements of total cheek tooth enamel ridge perimeter distance have been found to be only 7 cm more in a 1000 kg horse when compared with a 350 kg pony.18 More recently, a number of dental morphological variables (including total, inner, and outer enamel ridge perimeter distance as well as total, inner, and outer surface area) were measured in horses of different body size and further confirmed an absence of correlation between body size and any measured variables.7
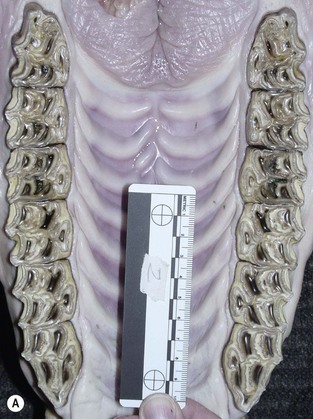
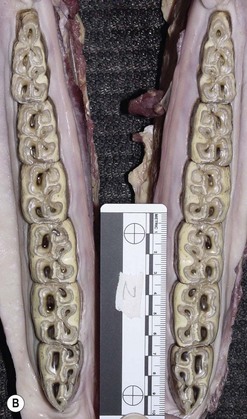
Fig. 6.2 (A) A photograph of an equine maxilla taken post mortem. Note the curvilinear arrangement of the cheek teeth and compare the width of the interdental oral cavity (IDOC) with that of the mandibular teeth. (B) A photograph of an equine mandible taken post mortem. Note the relatively straight arrangement of the cheek teeth and compare the width of the interdental oral cavity with that of the maxilla.
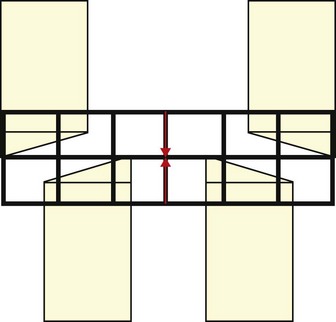
Fig. 6.3 A schematic diagram of the equine head as it pertains to the teeth. The outline of the incisors is in black and the position of the cheek teeth can be seen through them. Note that in centric occlusion (when interdental spaces 1/201 and 3/401, red arrows, are in alignment) the cheek teeth are not in occlusion. This is normal.
The mandible articulates with the maxilla at the temporomandibular joint (TMJ). This is a synovial joint formed by the condylar processes of the mandible and the articular tubercle of the temporal bone. The relative incongruity between these bones is accommodated by the presence of a fibrocartilagenous disc. The joint capsule is reinforced by the presence of rostral and caudal ligaments, the latter of which is concurrently attached to the disc. The joint is divided by the disc into a larger dorsal and a smaller ventral compartment, which communicate on the axial aspect of the joint. TMJ morphology has been described in-depth using a variety of different imaging modalities,19,20–22 (and as described in Ch. 23) and while the relative shapes and sizes of the osseus components vary with the size of the head, there is a consistent angulation of the mandibular condyle and temporal bone. The mandibular condyles are angled at approximately 15°, in a ventromedial to dorsolateral plane as well as from a mediocaudal to laterorostral direction (Fig. 6.4). These angles are mirrored by those of the articular portion of the temporal bone. Interestingly, this 15° angulation of the TMJ is also reported to be reflected in cheek tooth occlusal angles, as well as the angles of the palatine ridges.23 More recent studies do not support this association as closely. Cadaver studies suggested that not all cheek teeth within an arcade have the same angulation.24,25 Mean angulation in the former study was 10° (+/− 6 degrees SD) and was unaffected by tooth position or tooth age,24 whereas in the latter study mandibular angles ranged from 19.2 to 30° and were significantly different from their maxillary counterparts, with a range from 12.5 to 18°.25 In a study using live horses, mean molar cheek tooth occlusal angle was determined to be 9° (+/−2 degrees) and 10.6 degrees (+/− 7°) using a photographic and single tooth method, respectively.26
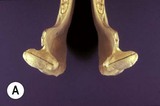
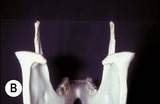
Fig. 6.4 (A) Mandibular condyles, caudal view –15° angles. (B) Mandibular condyles, dorsal view –15° angles.
Jaw closing is effected primarily by the paired masseter and temporalis muscles with a contribution from the pterygoid muscles. These muscles originate on the maxilla and cranium and insert on the mandible.
Jaw opening is effected by contraction of the anterior belly of digastricus combined with the contraction of geniohyoideus, and the inferior fibers of genioglossus coupled with the sternohyoideus and omohyoideus. All of these muscles that open and close the jaw are innervated by the fifth cranial (trigeminal) nerve.
The significant disparity in mass between jaw elevator and depressor musculature can be explained by understanding the movements of the equine jaw during prehension and mastication. The mandible is elevated (mouth closed) against resistance (gravity and feed), whereas opening is primarily effected by gravity. Prehended feed is crushed and sheared during jaw closure, which requires forces exceeding those generated simply by elevating the mass of the mandibular structures. Jaw muscles have faster contraction rates than most other striated muscles, with reported contraction rates of 333–500 cycles/min in pygmy goats.27
The muscles of the cheeks and lips include the levator and depressor labii maxillaris and mandibularis, the orbicularis oris, the incisivus mandibularis and maxillaris, the buccinator and zygomaticus muscles. These muscles are innervated by the seventh cranial (facial) nerve and control lip closure, elevation, retraction and depression, as well as the flattening of the cheeks.
The role of dentition in post-masticatory feed digestibility in the horse has been extensively studied in recent years. Three initial clinical trials examined the role of dentition in equine nutrition.28–30 The former two pronounced a benefit of dental floating on feed digestibility, whereas the latter did not. Further studies suggest that although occlusal equilibration (floating) increases the rostrocaudal mobility (RCM) of the mandible,31 and reduces the number of chews/kg DM (hay and barley) it does not alter chew rate,32 or improve feed digestibility, fecal particle size, or weight gain in the pregnant mare.31,33,34 Examination of the control animal subset similarly did not find a correlation between oral pathology score and weight gain.
There is also controversy surrounding the effect cheek tooth occlusal angle and cheek tooth occlusion percentage have on crude protein and fiber digestion,30,35 with others finding no discernable effect on the same outcome variables.26 Additionally, the study of Carmalt and Allan found no correlation between any dental morphological variable examined and feed digestibility or ingesta particle size.7
Mastication: the chewing cycle
Chewing is a repetitive, cyclical motion resulting from the contraction of musculature. Other than in man, studies of chewing cycles in mammals indicate consistent chewing patterns (individually and within the species). How the food is broken down, however, in horses, depends on the integrity of the cheek teeth. In the following description of the masticatory cycle, the start-point will be the ‘neutral position’, when the incisors are in centric occlusion (i.e., the interdental space of 1/201 and 3/401 are in vertical alignment, Fig. 6.3) and the cheek teeth are not in occlusion. Other methods of description use maximal incisor separation as the start-point.36
Herbivorous mammals have a masticatory cycle consisting of three phases: the opening stroke (O), the closing stroke (C), and the power stroke (P), which are defined by the relative displacement of the mandible.37 Using isolated frames from a video recording of equine mastication (Fig. 6.5A), a schematic diagram of mandibular motion was constructed (Fig. 6.5B). Note that Points 1–4 represent the opening stroke, including a small but integral rostral movement of the mandible,31,38 Points 5–6 represent the closing stroke, and Points 7–10 represent the power stroke. This analysis of the equine masticatory cycle suggests, in fact, the presence of a fourth stroke – a post power ‘recovery’ stroke. During the opening stroke, the mandible is displaced (laterally translated) to the point that cheek teeth occlusion occurs, at which point further displacement (translation) necessitates incisor separation (Fig. 6.6). The forces generated during the closing and power equine masticatory strokes have been reported to be 248N ± 117N (24.8kg) and 875N ± 278N (87.5 kg), respectively. Maximum chewing force recorded was 1758N (175.8 kg).39
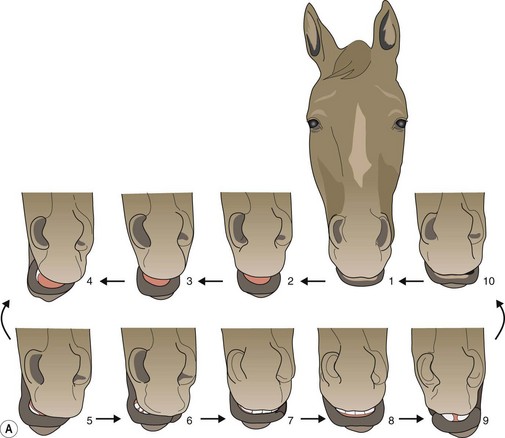
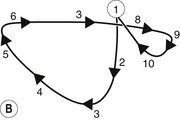
Fig. 6.5 (A) Isolated video frames during a single masticatory cycle in the horse. (B) Schematic diagram to explain mandibular movement during the masticatory (chewing) cycle. The figure is drawn from an imaginary perspective, above and immediately ahead of the horse’s head.
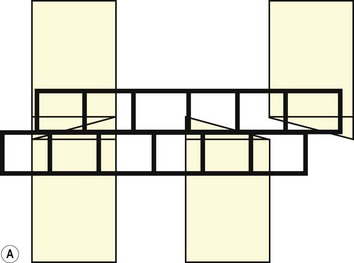
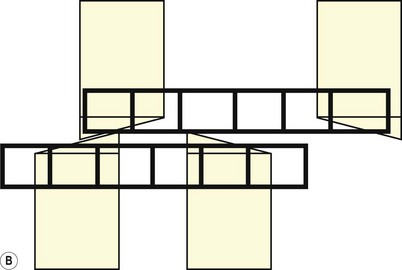
Fig. 6.6 (A) A schematic diagram of the equine head as it pertains to the teeth during the opening cycle of mastication. In this view, the mandibular movement is to the reader’s left and is arrested at the point of molar contact. To travel further laterally, incisor separation has to occur. (B) As above. Lateral movement is complete. Cheek teeth are in maximal occlusion, and the incisors are separated.
Some observers have stated that horses appear to be either right-sided or left-sided chewers. While some horses consistently demonstrate major lateral mandibular excursion to one side only, the belief that they are only applying crushing force to one side of the mouth consistently is not accurate. In a study of the masticatory movements of 400 horses, 45 horses (11 %) chewed on both sides, 163 horses (41 %) moved the mandible to the right (i.e., clockwise as viewed from the front) and 131 (32 %) chewed counterclockwise (i.e., mandibular movement to the left), while 63 horses had incomplete observations recorded.40
It has further been suggested that during the power stroke in ungulate mastication, there is only contact with one side of the arcade at a time.41,42 This may be the case in some species, but in the horse, the extent of anisognathism would suggest that there has to be some contact with both sides. Despite this anatomical constraint, major pressure would first be applied to one side and then, as the surfaces slide across each other, to the other side (frames 5–10). It might be concluded from these observations that there is a tendency for unequal dental attrition as a result of the variation in masticatory physiology. Necropsy examination of the occlusal surfaces of the cheek teeth of horses (including those with routine dental care, as well as those with a known history of no dental care) does not support this.
When eating, the horse uses its lips to prehend food material and pull it between the incisor teeth. The incisors cut or grasp the food material using a bite force of approximately 2 % of body weight.43 The rostral part of the mouth is thus filled, and mastication begins. The passage of feed across the occlusal surfaces of the cheek teeth and its subsequent movement caudally within the oral cavity can be likened to that of an auger. It is important to remember at this stage that all six cheek teeth function as a single unit and feed material is processed by each portion of each tooth only once, as the bolus is moved caudally. The cheeks keep the ingested, partially masticated feed within the intradental oral cavity (IDOC).
As the feed material is crushed, it is directed into the IDOC by the food channels on the occlusal surfaces of the cheek teeth (loph basins). There are also 18 pairs of incomplete palatine ridges, each of which is curved from caudolateral to rostro-medial, and offset in the midline (Fig. 6.2A). Food material is compacted in the IDOC, pressed against the palatine ridges by the tongue and moved caudally by the rotatory action of mastication, tongue and cheek compression. Swallowing is initiated when boluses of food collect in the oropharynx.
The auger analogy has been substantiated by descriptions of feed-bolus shapes obtained from edentate horses. Provided that feed presentation (crushed or soaked) is acceptable, edentate horses can survive and thrive, but if they are allowed access to long-stem fiber (such as grass or hay), spiral boluses of unmasticated feed combined with copius amounts of saliva are produced, which may represent a choke (esophageal obstruction) hazard.40
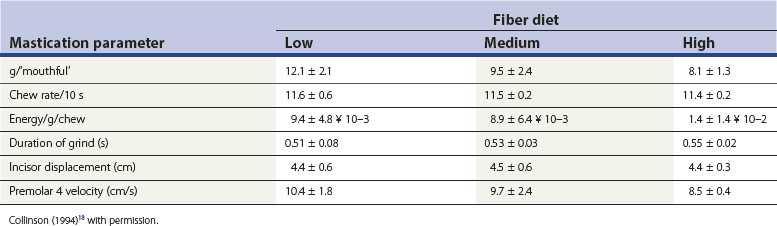
Factors influencing masticatory movements include the fiber and moisture content, and the physical structure (degree of processing) of the diet. Chew rates have been calculated from electromyographic data,37 and direct observations18,44 (Table 6.1, data recorded over 10 minutes). It was noted that horses were capable of attaining higher than 11 per second chew rates, particularly at the onset of feeding. In more recent studies, rates of 8 +/−1 chew cycles per 10 seconds (0.8 cycles per second) have been recorded.39 The degree of lateral excursion was originally documented in an innovative study that produced ‘molographs’ of the chewing pattern (the extent of lateral excursion) in horses eating different feeds.45 However, Collinson’s research18 did not confirm these earlier observations that higher fiber content and lower moisture content reduced the extent of excursion of the mandible.45 More recently, studies have documented the three-dimensional kinematics of the equine TMJ and associated movement of the equine mandible during mastication.18,46 A further study used the same recording method (using markers attached to the maxilla and mandible) to determine the relative positions of these structures while horses chewed either hay or pellets.47 From these data, the three-dimensional position, i.e., displacement and rotation (defined as yaw, pitch, and roll) of the mandible compared to the maxilla could be calculated. There was no difference in mean velocity of mandibular motion between hay and pelleted diets; however, chewing frequency was lower in hay compared to pellet diets. The study also found that there was greater lateral translation (movement) of the mandible when chewing hay than when chewing pellets. This increased lateral movement was sufficient to allow full occlusal contact between upper and lower cheek teeth rows during the chewing cycle. These scientific data lend support to clinical observations that horses managed extensively may not develop significant cheek tooth malocclusions (specifically sharp lateral edges to the maxillary cheek teeth and lingual edges to their mandibular counterparts) as frequently as pellet fed, intensively managed stabled animals.33,48
Table 6.1 Mean (±SD) values for mastication parameters for the four horses used in the food-processing experiment
Mandibular motion is controlled by the muscular forces acting upon it; however, morphological changes in dentition (occlusal pathology or malocclusions such as a ‘shear mouth’) or TMJ pathology have the potential to dramatically affect masticatory efficiency. Determining whether TMJ disease leads to alterations in the biomechanical forces during mastication that subsequently cause dental malocclusions, or whether dental malocclusions lead to altered mastication, thereby leading to TMJ pathology, is very difficult. To date, other than cases of septic or traumatic joint disease of the TMJ, there are no published data (clinical case reports or otherwise) to support a diagnosis of degenerative joint disease of the TMJ, despite the fact that it clearly occurs (see Ch. 23).
Summary
The design and function of the equine masticatory apparatus has been refined over millions of years to cope with the feedstuffs necessary for survival. The development of hypsodonty, prolonged eruption of teeth, and the balancing of eruption and attrition have been finely tuned. The masticatory cycle results in balanced dental attrition which, in the absence of dental abnormalities, serves the horse well. The process of domestication (intensive rather than extensive management styles) and the advent of processed feedstuffs significantly alter the duration and biomechanics of mastication. These factors, in combination with increased performance expectations, selective breeding that ignores the presence of inherent dental disorders, and the current longevity of horses, have resulted in the appearance of dental malocclusions and diseases of the soft tissues of the mouth upon which the science and art of equine dentistry are based.
1 Pérez-Barberia FJ, Gordon IJ. Factors affecting food comminution during chewing in ruminants: a review. Biological Journal of the Linnean Society. 1998;6(3):233–256.
2 Pérez-Barberia FJ, Gordon I. The influence of molar occlusal surface area on the voluntary intake, digestion, chewing behaviour and diet selection of red deer (Cervus elaphus). J Zool Lond. 1998;245:307–316.
3 Gross J, Demment M, Alkon P, et al. Feeding and chewing behaviors of Nubian ibex- compensation for sex-related differences in body-size. Funct Ecol. 1995;9:385–393.
4 Gipps J, Sanson G. Mastication and digestion in Pseudocheirus. In: Smith AP, Hume ID. Possums and gliders. Sydney: Australian Mammal Society; 1984:237–246.
5 Lanyon JM, Sanson GD. Koala (Phascolarctos cinereus) dentition and nutrition. I. Morphology and occlusion of cheek teeth. J Zool Lond. 1986;209:155–168.
6 Lanyon JM, Sanson GD. Koala (Phascolarctos cinereus) dentition and nutrition. II. Implications of tooth wear in nutrition. J Zool Lond. 1986;209:169–181.
7 Carmalt JL, Allen A. The relationship between cheek tooth occlusal morphology, apparent digestibility, and ingesta particle size in the horse. J Am Vet Med Assoc. 2008;223(3):452–455.
8 Hiiemae KM. Mammalian mastication: a review of the activity of the jaw muscles and the movements they produce in chewing. In: Butler PM, Joysey KA. Development, function and evolution of teeth. London: Academic Press; 1978:359–398.
9 MacFadden BJ. What’s the use? Functional morphology of feeding and locomotion (pp 229–262). In: Fossil horses. Systematics, paleobiology and evolution of the family equidae. Cambridge, UK: Cambridge University Press; 1992.
10 Anon. The theory of evolution. The New York Times. September 23rd 1876
11 Bennett D. The evolution of the horse. In: Evans JW, ed. Horse breeding and management. New York: Elsevier Science; 1992:21–29.
12 Butler PM. Some functional aspects of molar evolution. Evolution. 1972;26:474–483.
13 Rensberger JM, Forsten A, Fortelius M. Functional evolution of the cheek tooth pattern and chewing direction in Tertiary horses. Paleobiology. 1984;10:439–452.
14 Stirton RA. Observations on evolutionary rates in hypsodonty. Evolution. 1947;1:32–41.
15 Simpson GG. Horses. Oxford: Oxford University Press; 1951. pp 106–108
16 Stirton RA. Development of characters in horse teeth and the dental nomenclature. J Mamm. 1941;22:339–410.
17 Carmalt JL, Allen AL. Morphology of the occlusal surfaces of premolar andmolar teeth as an indicator of age in the horse. J Vet Dent. 2008;25(3):182–188.
18 Collinson M. Food processing and digestibility in horses (Equus caballus). Monash University: BSc Dissertation; 1994. pp 36–42
19 Weller R, Taylor S, Maierl J, et al. Ultrasonographic anatomy of the equine temporomandibular joints. Equine Vet J. 1999;31:529–532.
20 Rodriguez MJ, Agut A, Gil F, Latorre R. Anatomy of the equine temporomandibular joint: study by gross dissection, vascular injection and section. Equine Vet J. 2006;36:143–147.
21 May KA, Moll HD, Howard RD. Arthroscopic anatomy of the equine temporomandibular joint. Vet Surg. 2001;30:564–571.
22 Rodriguez MJ, Latorre R, Lopez-Albors O, et al. Computed tomographic anatomy of the temporomandibular joint in the young horse. Equine Vet J. 40, 2008. (fast track)
23 Baker GJ, Equine temporomandibular joints (TMJ): Morphology, function and clinical disease. Proceedings American Association of Equine Practitioners, 2002:442–447.
24 Carmalt JL. Observations of the cheek tooth occlusal angle in the horse. J Vet Dent. 2004;21:70–75.
25 Brown SL, Arkins S, Shaw DJ, Dixon PM. Occlusal angles of cheek teeth in normal horses and horses with dental disease. Vet Rec. 2008;162:807–810.
26 Carmalt JL, Townsend HGG, Cymbaluk NJ. The effect of cheek tooth occlusal angle and degree of occlusion on weight gain, feed digestibility and fecal particle size in horses. J Am Vet Med Assoc. 2005;227:110–113.
27 Gans C, DeVree F. Correlation of accelerometers with electromyograph in the mastication of pygmy goats (Capra hircus). Anatomical Record. 1974;306:1342–1343. (Abst)
28 Gatta D, Krusic L, Casini L, et al, Influence of corrected teeth on digestibility of two types of diets in pregnant mares. Proceedings 14th Symposium Equine Nutrition and Physiology Society, 1995:326–331.
29 Krusic L, Easley J, Pagan JD. Influence of corrected teeth on daily food consumption and glucose availability in horses. In: Proceedings 1st Symposium on horse diseases. Slovenia: Radenci; 1995:53–68.
30 Ralston SL, Foster DL, Divers T, et al. Effect of dental correction on feed digestibility in horses. Equine Vet J. 2001;33:390–393.
31 Carmalt JL, Townsend HGG, Allen AL. A preliminary study to examine the effect of dental correction on rostro-caudal mobility of the equine mandible. J Am Vet Med Assoc. 2003;223:666–669.
32 Ellis AD. The effect of dental condition on feed intake behaviour, digestibility of nutrients and faecal particle sizes in horses. In: Proceedings horse health nutrition: Second European Equine Health & Nutrition Congress, Equine Research Centre. Lelystad, Netherlands: Waiboerhoeve; 2004.
33 Carmalt JL, Allen AL. The effect of rostro-caudal mobility of the mandible on feed digestibility and fecal particle size in the horse. J Am Vet Med Assoc. 2006;229:1275–1278.
34 Carmalt JL, Townsend HGG, et al. The effect of dental floating on weight gain, body condition score, feed digestibility and fecal particle size in the pregnant mare. J Am Vet Med Assoc. 2004;225:1889–1893.
35 Rucker BA, Modified procedure for incisor reduction. Proceedings 41st Annual Meeting American Association of Equine Practitioners, 1995:41. 42–44
36 Hiiemae KM. Mammalian mastication: a review of the activity of the jaw muscles and the movements they produce in chewing. In: Butler PM, Joysey KA. Development, function and evolution of teeth. London: Academic Press; 1978:359–398.
37 Weijs WA, Dantuma R. Electromyography and mechanics of mastication in the albino rat. Journal of Morphology. 1975;146:1–34.
38 Bonin SJ. Three dimensional kinematics of the equine temporalmandibular joint. Michigan State University: MS Thesis; 2001.
39 Staszyk C, Lehmann F, Bienert A, et al. Measurement of masticatory forces in the horse. Pferdeheilkunde. 2006;22:12–16.
40 Baker GJ. Dental Physiology. In: Baker GJ, Easley J. Equine dentistry. 2nd edn. London: Elsevier; 2005:49–54.
41 Fortelius M. Ecological aspects of dental functional morphology in the Pleistocene rhinoceroses of Europe. In: Kirsten B, ed. Teeth, form, function and evolution. New York: Columbia University Press; 1982:163–181.
42 Fortelius M. Ungulate cheek teeth: developmental, functional and evolutionary interrelations. Acta Zoologica Fennica (Helsinki). 1985;180:78.
43 Hongo A, Akimoto M. The role of incisors in selective grazing by cattle and horses. J Agric Science. 2003;140:469–477.
44 Brøkner C, Nørgaard P, Søland TM. The effect of grain type and processing on equine chewing time. Pferdeheilkunde. 2006;22:453–460.
45 Leue G. Beziehungen zwischen Zahnanomalien und Verdauungsstörungen beim Pferde unter Heranziehung von Kaubildern. Hanover: Veterinary Medicine Dissertation; 1941. pp 170–174
46 Bonin SJ, Clayton HM, Lanovaz JL, Johnson TJ. Kinematics of the equine temporomandibular joint. Am J Vet Res. 2006;67:423–428.
47 Bonin SJ, Clayton HM, Lanovaz JL, Johnson TJ. Comparison of mandibular motion in horses chewing hay and pellets. Equine Vet J. 2007;39:258–262.
48 Carmalt KP, Carmalt JL. Equine dentistry: what do we really know? J Vet Dent. 2007;21:134–135.