CHAPTER 11 Oral and dental tumors
Introduction
Any tissue can develop neoplastic changes, and the structures of the mouth, including the soft tissues, the bone, and the teeth, are no exception. In general, however, this is an area of equine clinical oncology that has been largely ignored in spite of its importance to eating and its usually high visibility. Moreover, most published reports of specific oncologic conditions involve single or few cases, or are broad reviews based on these.1–4
In common with other neoplastic disease, primary oral and dental tumors are usually classified according to their tissue of origin as (Fig. 11.1):
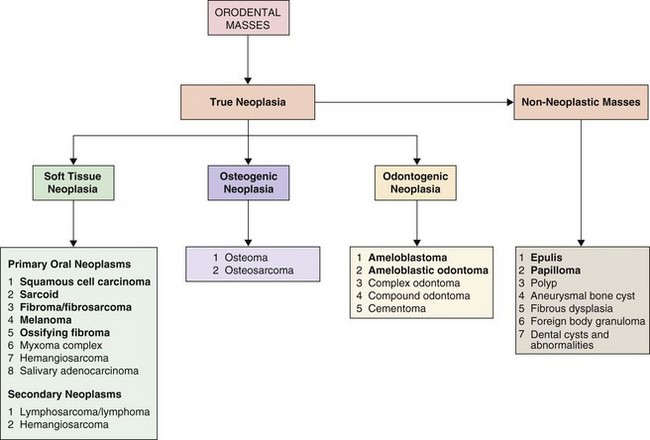
Fig. 11.1 Classification of orodental masses in the horse. The most common lesions are shown in bold font.
and according to their clinical behavior and pathologic features as:
As with almost all equine cancerous conditions, there is no meaningful information on any tumor staging of any of the orodental tumors of horses. The tissue of origin, the location of the tumor, the extent of secondary tissue involvement, and the clinical and pathologic tumor behavior all inevitably have a profound effect on the feasibility and choice of treatment, as well as the prognosis for the horse.
When orodental neoplasms occur, they are often clinically important and can in many cases be life-threatening.5 Whilst some such tumors are recognized rapidly, the majority are not; often the secondary changes, such as weight loss or difficulty with eating (dysmasesis) are the main reason for presentation. Even the most astute owners may not notice oral lesions in their early stages, and so, many tumors are in an advanced state when first presented. This makes the general diagnosis of neoplasia relatively straightforward, but rather disappointing in most circumstances, since treatment options are then extremely limited.6 Additionally, since the gross appearance of many neoplastic masses in their early and advanced states can be similar, the definitive diagnosis inevitably depends on histological examination.7
An added complication is that some non-neoplastic oral conditions, such as epulis, gingival hyperplasia, granulation tissue, and hamartoma can give the clinical suspicion of neoplasia. Indeed, some masses have histological features that support a diagnosis of neoplasia but are not, in fact, cancerous.8–10 For example, fibrous metaplasia of the nasal region and hard palate have been described,11,12 and benign neoplastic growths are seen occasionally in association with abnormal germinal tissue of tooth apices13 (Fig.11.1). These include:
There are also some cystic dentigerous disorders that may easily be mistaken for neoplasia.
Histological confirmation of the exact nature of oral tumors may prove difficult for a number of reasons. Both soft tissue and osetogenic and odontogenic tumors may be complicated by concurrent, long-standing infection or granulation tissue proliferation that may mask the true nature of the underlying lesion. Secondly, some of the hard tissue oral tumors are extremely difficult to biopsy and then to process for histopathologic examination. The rather variable classification of oral lesions also makes initial assessment of tumors difficult. Some tumors fall into the undifferentiated or unclassifiable myxoma/spindle-cell tumor group,14 which have ill-defined histological characteristics and variable clinical features. The variation in classification of equine tumors makes the specific diagnosis of many clinically obviously neoplastic diseases difficult and is further affected by the variable interpretation of different pathologists. It has to be recognized, however, that oral and dental tumors are relatively uncommon. This means that individual pathologists are unlikely to have an extensive database of experience of neoplasms at this anatomical site. It also means that careful consideration and appropriate sample collection are essential.
Although there have been some advances in the management of many neoplastic conditions in the horse, the low incidence of oral tumors makes it difficult to define the best approach to any particular tumor, and there is a lack of comparative evidence-based efficacy studies for the various therapeutic options. Treatment options may also be affected by the delayed detection of tumors. Many have a benign character, but their size may make them impossible to treat by currently available means. Clinicians frequently have to make compromises from the ideal treatment options. The early diagnosis of an untreatable condition may not always be in the horse’s best interests, since euthanasia may be performed before it is strictly necessary on welfare grounds, thus depriving the horse of some additional quality life and the owner of enjoyment. Often insurance and financial considerations take priority over the welfare issues.
Considering that most cases are presented late in the course of disease, determining the prognosis for a particular case is frequently the primary objective of the clinician, rather than providing any realistic treatment. Owners are generally more concerned with the prognosis than with the disease itself, but some expect treatment to be successful in every case. As most of these conditions are rare (or very rare), a realistic and objective prognosis, with or without treatment, may be difficult to provide. The course of most orodental tumors is unpredictable, and so the prognosis frequently becomes very subjective. Further, it is unfair to expect a pathologist to provide an accurate prognosis when there are few recorded cases of individual tumor type, and extrapolation from other species is usually not justified. However, more frequent reporting has improved the understanding of most equine neoplastic disease from both clinical and pathological perspectives.
Some oral neoplasms are very destructive, and so there may be extensive secondary changes that are more obvious than the underlying condition. Also, neoplastic tissue is more susceptible to infection and trauma, and so the clinical appearance may be more severe than the tumor alone warrants. These factors have a considerable bearing on the ability to diagnose the oral lesions simply from clinical supposition and experience.
There are few pathognomonic signs for any particular type of tumor apart from the distinctive visual appearance of some conditions. The general presenting signs encountered, which are often subtle in the early stages, include:
1. Gross appearance of an abnormal mass of tissue, or secondary anatomic alterations due to proliferation or destruction of tissue within the oral cavity or adjacent structures, such as the cheeks, nasal cavity, and paranasal sinuses
2. Oral bleeding (manifest as hemosalivation or melena)
3. Dysprehension / dysmasesis (dysmastication) / dysphagia
For example, oral carcinoma can result in loss of buccal sensation, and so the horse may suffer from significant self-trauma to the soft tissues of the mouth. A destructive oral carcinoma involving the palate may produce an oronasal fistula that might be recognized first by the presence of a nasal discharge, with or without overt food material. A space-occupying mass in the mouth may simply present with anatomic distortion and some functional deficits.
Investigation of a suspected oral mass should always begin with a detailed clinical history. A physical examination should be performed to establish both the clinical nature and extent of the neoplasm, to identify the structures involved, and to try to assess whether these changes are primary or secondary. A relatively small lesion in the mouth or adjacent structures may be secondary to a much more extensive lesion elsewhere. A good example of this is lymphosarcoma – the oral or pharyngeal lesions may be clinically insignificant, yet there can be extensive internal organ involvement.
Biopsy of any suspected neoplastic lesion is the mainstay of investigation. While biopsy of a suspect mass is frequently performed, it should be preceded by investigations that help to establish the extent and possible nature of the condition. For example, it may be very important to know if bone or other structures are involved, and this may influence both the site of biopsy and the method required to obtain diagnostic material. In many cases, it is useful to consult with a pathologist before performing a biopsy to ensure that the best diagnostic specimens are obtained and also so that the pathologist can orientate the specimens correctly in the context of precise anatomic location.
The prognosis varies markedly with the specific characteristics of the tumor. In some cases, these may not be the same as the classical description in other species. For example histopathology may suggest high malignancy, but the tumor may show no clinical evidence of this behavior. The converse situation can also arise.
Diagnostic procedures for suspected neoplastic disease
1. Radiography is the standard imaging method in the practice situation (see Ch. 13). These two-dimensional images do, however, create some interpretative difficulties. Where radiography is the only available imaging modality, carefully positioned images can be a major diagnostic help and a satisfactory diagnosis can be achieved or at least assisted in many cases. Unless experienced, it is useful to have a reference book available on normal radiographic variation because interpreting radiographs of the equine head can be difficult. Oblique projections can be helpful and fluoroscopy can be a significant aid in both diagnosis and treatment of head tumors. Contrast angiography can be a useful aid to surgical and possibly medical therapy, especially in aggressive tumors with large blood supplies.
2. Computed tomography (CT) is becoming increasingly available in veterinary practice and can now be performed on the standing sedated horse. It provides a three-dimensional radiograph and is particularly valuable for those conditions that are difficult or impossible to palpate or inspect (such as tumors within the paranasal sinuses or those associated with the teeth). CT images also help to establish the presence or absence of any secondary changes in the bone and other structures. This gives a greatly improved appreciation of the extent and nature of the challenges and complications likely to be faced by the surgeons in particular (see Ch. 13).
3. Magnetic resonance imaging (MRI) is an ideal modality for imaging soft tissues, and whilst the facilities for this are currently limited, they are increasing. MRI imaging suffers from the need for immobility and takes a considerable time to perform; therefore, general anesthesia is almost compulsory. Again, a three-dimensional image can be generated, and this greatly helps in dealing with soft tissue tumors and other masses within sites that preclude full inspection. The full extent of the tumor and its anatomic relationships can be revealed (Fig. 11.2).
6. Direct examination and endoscopy:
7. Hematology and biochemistry:
8. Biopsy is inevitably the most reliable method of establishing a definitive diagnosis. Accessibility is not usually a major obstacle with orodental tumors, but specialist approaches including trephination of sinuses and ultrasound or fluoroscopic guidance may be helpful. Biopsy of a suspected neoplastic mass can be achieved in several ways, including:
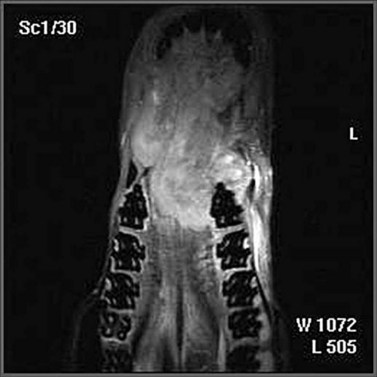
Fig. 11.2 This oro-nasal palatine carcinoma was subjected to MRI scanning, and the 3-dimensional images significantly assisted the assessment of the options available to the clinician.
(Courtesy A.J. van den Belt.)
The best fine needle aspiration technique involves the use of a small-gauge needle (21-g or less) and a small syringe (2-ml ideally; Fig.11.3). Larger needles may seem an attractive option but usually they will harvest blood and gross tissue from the vicinity of the tumor and this is not helpful diagnostically – a few good cells, properly collected are far more useful than a few cells submerged in a large amount of necrotic tissue and blood. The needle should be inserted into the margins of the lesion – not the center, as many tumors have a necrotic center, and so, at that site, tumor cells may be degenerate, or non-specific inflammatory cells may be abundant. The best technique involves only 3–5 suction efforts with a maximum vacuum volume of 2 ml. Greater vaccuum is not achieved with a bigger syringe! Furthermore, prolonged or marked vacuum pressures may damage cells significantly! The needle is then withdrawn, and the point of the needle is directed at a clean, grease-free, glass microscope slide and the cells ‘jetted’ onto it by repeated ejection of air from the syringe. Several samples can be taken, and some can be spread onto the slide, and others simply left in situ. Thick preparations should be smeared immediately before being rapidly air-dried and fixed according to the requirements of the pathologist. An important practical consideration for cytology is to ensure that smears are not exposed to formalin fumes since the latter spoil cellular preparations for subsequent staining with Romanowsky cytological stains. It is best to identify the slides by writing on the frosted area with a pencil before the smears are made; markings made with a pencil will remain during alcohol fixing, and the pathologist will know which is the correct side of the slide to look at, even if there are only a few cells!
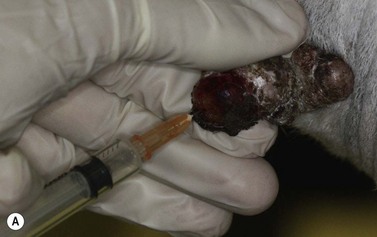

Fig. 11.3 (A) Fine needle aspiration requires no specialized equipment. It is best performed with a fine needle 23-g and a small 2-ml syringe. A fine needle is inserted into the margin of the lesion, and suction is applied via a 2-ml syringe 3–4 times. (B) The collected tissue should be jetted on to a clean slide, and air dried rapidly. The slide is identified with a pencil and sent directly to a cytologist. Alcohol fixation may be required.
Interpretive cytology is best performed by a skilled cytopathologist because low numbers of tumor cells may not be easily recognized among normal cells.
Specimens that are obtained via endoscopic biopsy instruments have limited value since they are so small, and because they are liable to artefactual distortion during collection; a reliable diagnosis may not be obtained. Often it is only possible to obtain a biopsy from the most superficial parts of the tumor and the interpretation may be confused by the presence of granulation tissue, necrosis, superficial infection, and inflammation around the tumor.
Useful information can sometimes also be derived from impression smears made from ulcerated tumors. The same principles apply here – it is far better to have several slides with a few cells than one with a thick cellular accumulation. Impression smears can be improved by gently blotting the ulcerated surface of the tumor with clean paper towel, to remove extraneous cells and excess fluid, and if possible, by gentle squeezing of the tumor itself.
Notwithstanding the specific tests that can be applied in the investigation of oral and dental tumors, most of the commoner tumors are fairly distinctive, and a tentative diagnosis can usually be made by intuitive supposition. Problems may, however, arise with rarer tumors and those with prominent secondary inflammation and necrosis.
General principles of differential diagnosis
Oral tumors are conveniently divided into:
1. Primary tumors (of dental, soft tissue, or bone origin)
2. Secondary tumors (of non-orodental tissues)
3. Tumors of adjacent structures and associated organs invading into the oral cavity or its associated structures.
Secondary tumors with primary lesions elsewhere and tumors invading the mouth from adjacent structures such as the skin, the paranasal sinuses, and the nasal cavity must be considered when investigating an oral mass since identifying the origin might be diagnostically helpful and may have therapeutic implications. For example, a destructive nasal adenocarcinoma might invade the hard palate, creating an oronasal fistula and loosening some of the teeth. It would then be pointless to attempt to deal with the oral aspect of the tumor alone (even if there was some method to do this).
The lack of reported series of individual oral tumors and tumor-like masses testifies to the fact that most of these conditions are uncommon16–19 and that no significant attempt has been made to classify them and to quantify their prevalence through multicenter studies. The specific difficulties that are presented by the tumors and their profound effects (whether benign or malignant) mean, however, that veterinarians are expected to make prognostic decisions that are inevitably based on limited experience rather than sound, evidence-based principles. Recommendations for treatment of rare conditions cannot be made with any certainty, and pathologists are often expected to provide information that simply does not exist. The reported satisfactory or unsatisfactory treatment of a single case does not entitle pathologists or clinicians to refer to ‘common’ treatment or ‘usual’ tumor behavior.
General principles of management for neoplastic disease
Ideally, of course, any diagnosis of a neoplastic disease would be followed by a timely and specific curative therapy. However, the nature of the condition, the almost inevitably high cost of treatment and the owner’s attitude have a significant bearing on the choice of treatment. Often, treatment is not attempted because of one or more of these factors. Progress is being made in the management of tumors in many veterinary species but the improvements in equine oncology have been disappointingly slow. This may reflect the fact that, on most occasions, a diagnosis is made very late in the course of the condition, and so at initial presentation the prognosis is sufficiently poor to warrant euthanasia. Also, anti-cancer medications are expensive and particularly so for large horses. There is a general opinion that horses do not tolerate systemic chemotherapy well. The secondary (unwanted or side-) effects of most systemic anti-cancer therapy mean that few owners and veterinarians are willing to subject a horse to their side-effects. Surgical options are necessarily limited by facilities, access to the tumor, and the possibility of unacceptable functional problems after the surgery.
An additional and severely limiting aspect is, of course, whether the tumor has already metastasized. Where this has occurred, palliative treatment can still be carried out to improve the short-term quality of life, but the prognosis is by then very poor. Even when some tumors have spread beyond the oral cavity, the horse might not warrant immediate destruction since, in some circumstances, the metastases may have few effects and may be slow-growing. It is clear that some procedures, such as hemimandibulectomy, and removal of part of the tongue can be well tolerated by many horses. All treatment modalities have inherent limitations, and these simply have been accepted as part of the overall case management.
Surgery
The limitations of all treatment modalities, and particularly surgery, are well recognized. Limitations relate to accessibility and the associated problems of defining the margins of a tumor to ensure its total removal. Often the margins cannot be defined, and the constraints of the oral cavity mean that there is less scope for removal of extra tissue to achieve a safer margin, and so recurrence almost inevitably occurs. When surgery of any type is performed, all of the tissues removed should be submitted for histological examination, and the risk of failure to achieve an adequate margin should not deter the surgeon from submitting the tissues. If the pathological report states that safe margins have not been achieved, the owner should be informed immediately and decisions made on the next sensible stage of the treatment. Combinations of treatments using different surgical techniques, or surgery plus other modalities, such as immunotherapy, radiation, or chemotherapy, improve the chances of a good outcome.
Sharp surgery
Sharp surgical excision of a tumor is clearly the fastest and most convenient method of treatment in most cases. Localized, benign or early localized, malignant tumors may be amenable to surgical excision. For example a squamous cell carcinoma of the tip of the tongue, a gingival fibrosarcoma, or a buccal sarcoid may be treated effectively by surgical excision.
Laser surgery
The increased availability and relatively low price of diode lasers now makes this a realistic option in most practice circumstances. It is still a surgical method and suffers from the same limitations as sharp surgery (Fig. 11.4). The advantages of laser surgery are its accuracy, the relatively bloodless surgical field, and the fact that some extra ‘die-back’ occurs when tissues are cut with a laser; the latter is also something of a disadvantage in that healing is slower, and sutured wounds may break down. Laser surgery also minimizes the risks of tumor seeding into the operative site. This is particularly important in the treatment of sarcoid tumors.
Cryosurgery
Small, focal, and superficial tumors can be treated by cryonecrosis. Systems using liquid nitrogen are the only ones that are effective in horses; however, the crude application of liquid nitrogen to a tumor mass to induce an uncontrolled freeze is unacceptable. The advantage of cryosurgery is that it is possible to destroy a wider area of tissue and allow a natural demarcation to develop over the following weeks. Cryosurgery is not suitable on its own for larger aggressive tumors since it is almost impossible to define the margin of the ‘freeze’ without complicated use of thermocouples, and in any case, the oral cavity appears to be much more difficult to freeze effectively than normal skin.
Chemotherapy
The options for chemotherapy in horses are limited to topical and intralesional medication. Usually, the materials are specific antimitotic or cytotoxic drugs, such as cisplatin (in stable emulsion, wax, or biodegradable bead forms), 5-fluorouracil (in solution, bead, or sponge forms), bleomycin, or mitomycin C in injectable solution. Although these are attractive options because they are logistically simpler and invariably cheaper than other methods, there is little information regarding their use in equine oral neoplasms in particular. Broadly, they have applications in the management of sarcoid, melanoma, and squamous cell carcinoma,20 but more information is being published about their use and applications for other tumor types. As might be expected, the position and size of the specific tumor being treated limits the opportunities for its use, and where tumors are presented late, the scope for effective treatment remains limited. Electrochemotherapy using electrical energy to increase the permeability of tumor cells to cisplatin (and possibly) 5-fluorouracil has recently been described for the treatment of buccal21 sarcoid and there are anecdotal reports of the benefits in squamous cell carcinoma and melanoma treatment also. The advantages of this system include the fact that the current can be restricted to the tumor location, but this method requires repeated general anesthesia. The biodegradable sponge or bead systems appear to be very logical and these may become the preferred practical approach in many circumstances. However, there are no comparative evidence-based studies on these methods to date.
Immunotherapy
Whilst various forms of immunotherapy, ranging from ‘autogenous vaccines’ (for melanoma) to intralesional BCG protein injections (for sarcoid, in particular), have been suggested to treat equine neoplasms, assessment of their value is limited by the lack of comparative clinical studies. Sarcoid seems to be the most prevalent tumor type that is subjected to this therapy, and there are some reports of its positive effects in certain types of sarcoid.
Radiation therapy
Radiation therapy is the gold standard therapy for most cutaneous and deep-seated malignant tumors in the horse. Radiation is used to eradicate the tumor cells, preferably without affecting the architecture and cellular elements of the adjacent normal tissues. Both gamma and beta radiation are used therapeutically through plesiotherapy, brachytherapy, and teletherapy. Radiation brachytherapy causes no material systemic toxicity and in contrast to surgical methods of treatment, has no anatomical constraints. However, it is seldom available for equine therapy for cost and logistical reasons. The likelihood of a successful outcome with radiation therapy is inversely proportional to the size of the tumor; this is a common constraint given the late presentation of most cases of equine oral tumors. The prognosis also depends on the tumor type, and its particular growth characteristics and susceptibility to radiation; slowly expanding tumors tend to respond more slowly and less favorably than rapidly dividing ones. For example, squamous cell carcinoma is probably more susceptible to gamma radiation than the fibroblastic sarcoid, and melanomas tend to respond poorly to all types of radiation. However, the same tumor type may respond differently in two different anatomic sites and in different horses, and so variations in ‘effective’ doses are almost infinite.
Disappointingly, there are few facilities that offer any sort of radiation therapy for horses. This reflects a totally unacceptable lack of interest in cancer medicine in a species that makes an enormous contribution to mankind! However, radiation can be used, and there are cases where interstitial brachytherapy or teletherapy has made a significant difference for oral tumors in particular.
Brachytherapy
Interstitial brachytherapy has considerable advantages in that high doses of radiation can be delivered precisely, safely, and conveniently over a short time without significant risks to the other parts of the body. Radioactive sources are implanted into tissues directly and are left in situ until a precalculated overall dose of radiation is delivered. The dose necessarily varies for the various types of tumor, but little is established about the best options for oral masses. Therapeutic radiation ionizes the DNA in cells within the therapeutic range of the sources but is not discriminatory for tumor cells alone. This means that susceptible normal cells are usually destroyed as well. The most susceptible normal cells in the skin are melanocytes, and so pigmentary changes are common. Iridium-192, gold-198, and iodine-125 are the most common isotopes used in this way. The procedures are all highly specialized, requiring careful dosimetry and specialist facilities both for insertion and hospitalization. The results obtained in 12 cases of oral neoplasia22 suggest that this is a potentially very satisfactory method of treatment. However, there are obvious difficulties relating to the availability and costs of such treatment. Since one of the major constraints on the outcome is the size of the tumor, the costs and the dose required can be reduced significantly by prior surgical debulking of large tumors.
Teletherapy
Teletherapy uses a generated beam of radiation (high energy beta or gamma rays) focused into the tumor mass. The main advantages are that no operator risks are incurred, the dose can be focused accurately, and several sub-lethal rays can be focused into a deep tumor without causing significant damage to the surrounding tissues. Where the beams meet, a radiation ‘hot-spot’ is produced that receives a lethal radiation dose. The problem is that this method, whilst being the true gold standard, is not available to horses at this time. General anesthesia would be required to allow treatment of most equine oral tumors, and no quantified dosimetry has been calculated for any equine tumor. A few cases of sarcoid, lymphoma and squamous cell carcinoma of the face and head have been treated successfully in this way.
Other ‘treatments’
Cancer always warrants a proper investigation and sensible treatment that has a prospect of helping, and where treatment is impossible an honest and direct opinion should be given to the owner. In spite of the availability of a variety of appropriate treatment options there are still many occasions when useless or even dangerous treatments are inflicted upon horses. Whilst homeopathy, for example, cannot possibly do any good, its main danger lies in the failure to provide effective and timely treatment and in causing unnecessary delays before proper therapy is instigated. This also means that the prognosis is far worse when proper treatment is finally requested and when that fails, the poor outcome is usually taken as indication of the inadequacy of the conventional methods! Since homeopathy has a positive explanation for any of the possible outcomes ranging from success to failure it must be viewed with a considerable degree of skepticism by any scientific mind. In spite of the considerable cost and the lack of any evidence of any efficacy whatsoever, these methods continue to be peddled by people who exploit the ignorant, the vulnerable, the gullible, and the disillusioned!
Tumors of dental-tissue origin (odontogenic tumors)
Tumors in this category are rare, although it has been suggested that they are more common in horses than in other species.17 Odontogenic tumors are classified according to the inductive effect of one dental tissue on the others.18,23 These tumors can be benign or non- metastasizing malignant, with the latter often locally invasive and aggressive in their clinical behavior. As a general rule, dental tumors are best treated by wide surgical removal (to ensure complete ablation of tumor and abnormal tissue) at an early stage in their development when such surgery has a chance of success. In most cases, however, the masses are not recognized sufficiently early, and so local recurrences are common in spite of attempts at wide surgical excision.19 Most oral bone and dental tumors are benign but can cause serious secondary effects, such as nasal obstruction and dental and facial deformity, resulting in dysmasesis and weight loss. There are some similar clinical conditions that resemble neoplasia that are in fact simply abnormal tooth formation (Fig. 11.5). An important diagnostic aspect in these cases is that the condition is present from the time of formation of the tooth. However, on presentation they may be very difficult to tell apart. Since the advent of equine dental medicine as a specialty, early recognition of abnormalities and deformities as well as neoplastic dental disorders, has become much more frequent.
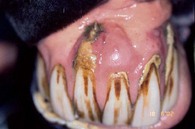
Fig. 11.5 This tumor-like mass developed in the incisive gingiva of a 12-year-old gelding. Biopsy was inconclusive, and only after full surgical removal was a diagnosis of an inflammatory reaction possible. Even then there remained some doubt, but as the condition did not recur, the matter was not pursued.
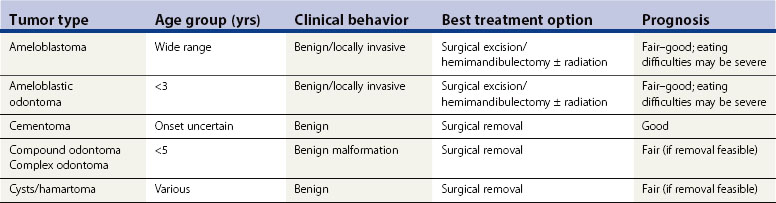
Odontogenic tumors are of variable histological appearance and are categorized currently on their morphologic basis23 (Table 11.1). Their features are summarized in Table 11.2.
Table 11.1 Equine dental tumors derived from odontogenic epithelium (E) or mesenchyme (M)
| Histologic designation | Synonyms |
|---|---|
| Ameloblastoma (E) | Keratinizing ameloblastoma adamantinoma |
| Cementoma (M) | |
| Complex odontoma (E)/(M) | |
| Cementifying fibroma (M) | |
| Ameloblastic fibroma (E)/(M) | Ameloblastic fibro-odontoma |
Ameloblastoma
Definition
These tumors are derived from odontogenic epithelium. True ameloblastoma produce no inductive changes in the connective tissue and so lack dentin and enamel.
Occurrence
These are most commonly found in the mandibular region (including medulla) of older horses24–29 but can involve the maxilla. Several cases have also been reported in young foals.30,31
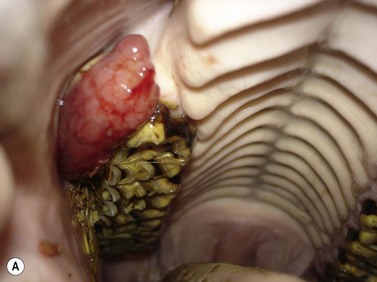
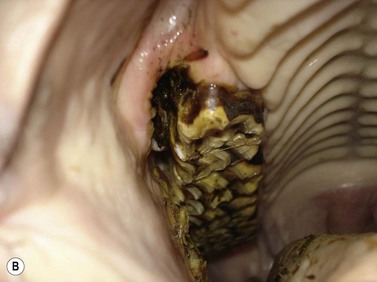
Clinical features
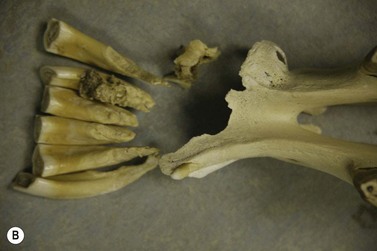
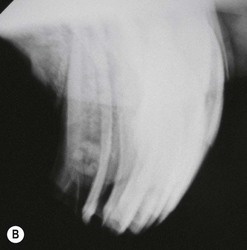
They may be overtly tooth-like, or contain little or no obvious dental tissue. They often develop a central, cystic region and cause bony/solid swellings and abnormalities in the associated dental arcade (Fig. 11.6). Occasionally, they can present with a discharging sinus on the side of the face. Late presentation shows an advanced and aggressively destructive epithelial tumor with extensive bone loss (Fig. 11.7).
Fig. 11.6 (A) Facial swelling caused by an ameloblastoma. The tissue contained no obvious dental tissue remnants. This differentiates it from an ameloblastic odontoma. (B) Radiographic appearance of an ameloblastoma in a 2-year-old Thoroughbred colt showing the characteristic multiloculated nature with radiodense fragments throughout the mass.
Reproduced with the permission of Dr Bruce Bladon.

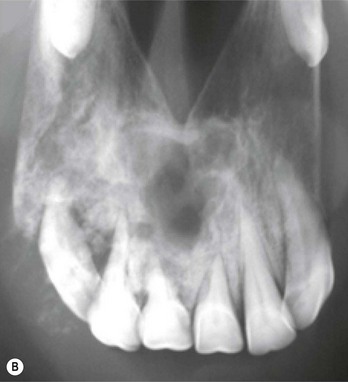
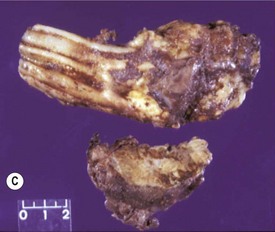
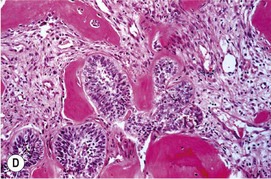
(C) An ameloblastoma excised from a 2-year-old Thoroughbred colt showing the relationship to the tooth and the expansive mass at and around its root. (D) Histologic section of an ameloblastoma showing clusters of orderly ameloblasts separated by connective tissue and spicules of hard dental material.
Differential diagnosis
Ossifying fibroma and other tumors of the jaw, such as invasive squamous cell carcinoma and myxomatous tumors, should also be considered (although the latter tend to be destructive rather than proliferative). Osteosarcoma is singularly rare in the horse. Infections of tooth apices and adjacent bone can be similar, but are associated with extensive necrosis and typical radiographic features often complicated by maxillary reactive bone proliferation with obvious facial swelling. Jaw fractures and other dental abnormalities, including malerupting and supernumerary cheek teeth, should also be considered.
Diagnostic confirmation
Biopsy and radiographic findings are typical but can be similar to other tumor masses. Ameloblastomas usually have a rubbery consistency and have a roughly spherical or multilocular shape with a cystic radiographic appearance (Fig. 11.6B). Odontomas are radiolucent or partially mineralized, with foci of calcified tissue mixed throughout. Even when there is extensive ulceration, there should be little confusion between ameloblastomas and carcinomas or sarcomas, which tend to be much more destructive than tumors of dental origin.
Pathology
Ameloblastoma is characterized grossly by swelling of the affected jaw and osteolytic changes within the jaw (Fig. 11.6C). They can be solid or cystic and are usually discrete. The major characteristic histological feature is the presence of odontogenic epithelium (Fig. 11.6D). If there is marked epithelial keratin formation, the lesion is termed keratinizing ameloblastoma. The lesion may be well circumscribed, or there may be local infiltration by odontogenic epithelium.
Treatment
Surgical removal can be curative if treatment is initiated early and wide excision can be performed. Horses seem to cope well with rostral hemimandibulectomy and especially so if the mandibular symphysis remains intact. Rostral mandibulectomy can also be successful, but special measures are required to ensure adequate nutritional intake. Radiation therapy is probably the best option and has been used successfully.31
However, suitable teletherapy facilities are not generally available, as noted, and so other options are usually sought. Topical chemotherapy is singularly unsuccessful.
Prognosis
The expansile nature of these tumors and their late recognition (particularly in foals and young horses) make the outlook poor. Many horses are euthanased soon after they are diagnosed with the tumor, although the rate of growth may be slow and some useful quality of life may be possible even if surgery is not feasible.
Cementoma
Definition
Cementoma is a rare, benign or reactive tumor derived from mesenchymal tissue and so does not contain epithelial components. It typically occurs in the apical region of the developing tooth. There are few published reports of this tumor, but one such lesion affected an incisor tooth (DCK, unpublished). It is possible that some of the features of this condition could be found in abnormal or supernumerary cheek teeth where extensive distortion of the dental structures by reactive cement deposition is encountered. However, it is often impossible to confirm the diagnosis of cementoma or to differentiate them from dental abnormalities.
Clinical features
The location of these tumors (at the apex of the tooth) makes their early recognition and diagnosis unlikely, and they are only recognized when there is overt jaw swelling (Fig. 11.8). Radiographically, they have a distinctive, very radiodense appearance, and the tissue contains sheets of cementum-like material. Secondary alveolar changes involving either infection or reactive bone proliferation may, however, make them harder to recognize. Alterations in the crown are unusual but make the condition more recognizable clinically.
Pathology
This lesion presents as a mass in the jaw or as a mass that involves the nasal cavity or maxillary sinus. It may be secondary to traumatic tooth fracture, dental impaction, or periodontitis. It is characterized histologically by the presence of mosaic-like, basophilic cement lines, with anchoring of Sharpey’s fibers into the cement matrix. With reactive cementoma, there is additional inflammation and fibrosis. Cementifying fibroma is a rare lesion that is analogous to ossifying fibroma, but the tumor matrix includes the complex basophilic lines of typical cementum.
Treatment
Removal of the tooth in its entirety is feasible but may be hindered by the large, cylindrical aggregation of hard tissue at the tooth apex. In some circumstances, it might even be better to accept the condition since it is generally very benign, and its slow onset may enable the horse to adapt well to it.
Prognosis
The lesion is benign and removal is curative. Some horses remain unaffected for many years with the condition being identified incidentally or at post-mortem examination. However, where clinically significant secondary changes occur, the prognosis depends on the possibility of removal of the affected tooth.
Complex/compound odontoma
Definition
A complex (compound) odontoma is an irregular, tumor-like mass of dental tissues in a well-differentiated form. Complex odontoma contains all the elements of a normal tooth, but the structure is chaotic. A compound odontoma is similar, except that the tissue is organized into recognizable, tooth-like structures (denticles), although they may be grossly distorted. There is some justification for considering some compound odontomas to be hamartomas of dental tissue, rather than true tumors.
Occurrence
Both young and older horses may be affected, with a greater prevalence in younger animals.
Clinical features
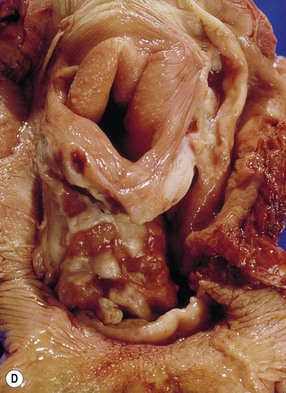
Many cases are identified incidentally. Firm, painless swellings over the apical regions of the maxillary cheek teeth or the premaxilla are characteristic.33 Swelling may not be obvious if the more caudal maxillary cheek teeth are involved as the expansion is contained in the maxillary sinuses (Fig. 11.9). Secondary sinusitis seems to be a rare complication.
Differential diagnosis
Dental infection with new bone formation and lysis. Sinus cysts may be associated with these in some cases.
Diagnostic confirmation
The radiographic appearance is characteristic – multiple, small lobulated masses within a well-defined cyst-like structure at the apex of a maxillary tooth are typical. Radiographic interpretation of the lesion becomes difficult when secondary changes occur in the adjacent teeth and sinuses.
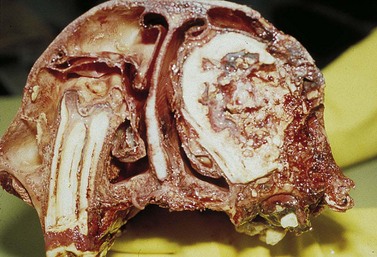
Pathology
Complex odontoma presents as a radiodense calcified lesion within the jaw of young horses. Grossly, they are very hard and difficult to prepare for histological examination. Cut surfaces reveal variegated cementum, dentin, and mineralized enamel (Fig. 11.10).34 The gross features are confirmed histologically, and there can be variable amounts of odontogenic epithelium. In the horse, there is plentiful cementum.
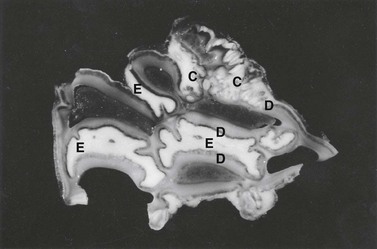
Fig. 11.10 Undecalcified thick section of a complex odontoma in a 2-year-old Morgan colt. Well-differentiated but disorganized components of tooth formation are shown. D, dentin; C, cementum; E, enamel.
(Reproduced with the kind permission of Dr R.R. Dubielzig and Iowa State Press.)
Compound odontoma presents as a similar lesion but radiographically shows several abnormal tooth-like structures (denticles) within the mass. Histological features are reminiscent of normal tooth development. In older lesions, epithelial tissue may be sparse.
Incidental tumor-like dental masses
This group includes temporal teratoma – a rare curiosity in the horse, in which dental tissue (which may be instantly recognizable as such) is located at sites away from the jaws (Fig. 11.11). The most common site is in the temporal region where a sinus tract discharges viscous, milky material from a discrete opening on the leading edge of the pinna. The cystic structure may be situated some way from the ear itself but sometimes there is an obvious dental structure located against or attached to the temporal bone of the calvarium. Sometimes the structure has no obvious dental tissue and comprises a smooth, cystic lining lying below the ear. Radiographs are used to establish the presence or absence and the location of any dental tissue.
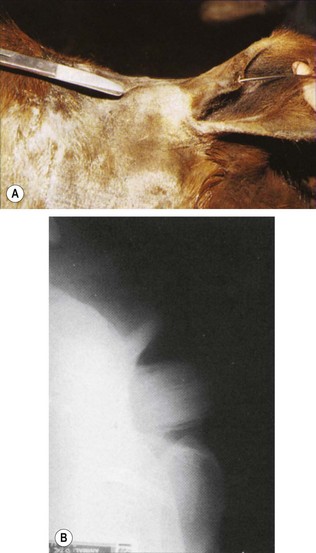
Fig. 11.11 (A) A discharging sinus had been present on the anterior margin of the pinna (where probe is inserted) for 2 years. The tract leads to an obvious solid non-painful mass just rostral to the base of the ear. This is the typical clinical appearance of a dentigerous cyst. (B) Oblique radiograph of the temporal region of the same horse as in (A). An obvious tooth-like structure with an associated ‘alveolus’ is present and is typical of many cases of dentigerous cysts. In the absence of obvious tooth-like structure, contrast radiography will identify a distinct or occasionally a poorly defined cystic structure at this or a neighboring site.
Pathology
These rare lesions are lined by stratified squamous epithelium and often contain abnormal dental structures.35–38 The surgical treatment of temporal teratoma can carry a good prognosis, particularly where the lesion is simply a cystic structure, although every part of the cystic lining must be removed to avoid recurrence. However, where aberrant tooth material is enclosed within a false alveolus protruding into the calvarium (see Fig.11.11B), surgery becomes much more of a challenge. Such cases must be fully assessed by CT scan, if possible, or at least by radiography (possibly with contrast material introduced via the sinus tract). In some cases, the cyst and the tooth-like structures are not obviously connected. Some cases can justifiably be left alone since the discharging tract is usually a cosmetic and managemental nuisance, rather than being clinically significant.
Tumors of bone (osteogenic tumors)
Osteosarcoma, osteoblastoma, chondrosarcoma, and fibrosarcoma have been described as arising in bone in horses. They are all very rare tumors, but there have been several reports involving the jaws and the mandible in particular, which indicates that this may be a predilection site.40 The histological characteristics of bone-derived tumors have been described,41 and the classification of this group of tumors is based upon these features.
Osteoma
Osteomas, which are extremely rare lesions, are slow-growing, solitary, well-differentiated masses of bone enclosing marrow and fat, and many pathologists regard them as a developmental anomaly or hamartoma, rather than neoplasms. They are reported to occur in all ages of horse, with most being located in the head region, including the mandible, maxillae, and paranasal sinuses. The osteoma may reach a large size and have a distinctive, discrete, radiodense outline. They are benign, but their growth may compromise adjacent tissues, causing disfigurement, obstruction of the nasal passages or interference with mastication and swallowing.
Macroscopically, they consist of dense bone. The histological features are of orderly cancellous bone; the intertrabecular fibrous connective tissue may include adipocytes and hemopoietic cells.
Surgical removal may be feasible, but most are a significant surgical challenge, and the prognosis is very guarded.
Osteosarcoma
Definition
Osteosarcoma is a malignant mesenchymal tumor of bone affecting horses of any age, in which the neoplastic cells produce modified or distinctive osteoid or bone matrix in a haphazard arrangement.
Occurrence
Osteosarcoma is an extremely rare tumor in the horse at any site. However, over 80 % of reported osteosarcomas involve the head, and the majority are reported in the mandible.42,43 There is a report of an osteosarcoma in the mandible of a 6-month-old Quarterhorse colt, which suggests that age is probably not a significant factor,44 although, typically, younger horses appear to be more prone to oral or dental neoplasia in general than older ones. Trauma is implicated as a risk factor for later osteosarcoma in other species such as the cat, but there is no convincing evidence for this in horses.
Clinical features
The condition is usually presented as a painful, hot, progressive swelling of the mandible with a characteristic ‘sun-burst’ radiographic appearance of bone lysis and irregular deposition of trabecular reactive new bone44 (Fig. 11.12). Pathological fractures can occur in affected bones.
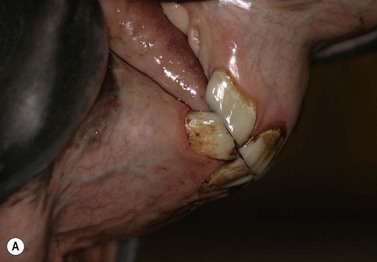
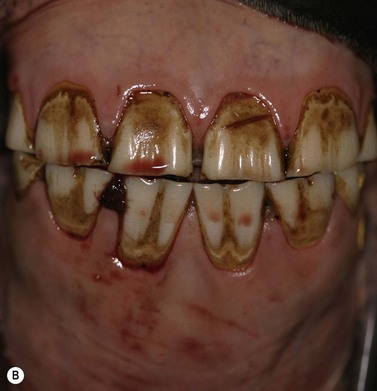
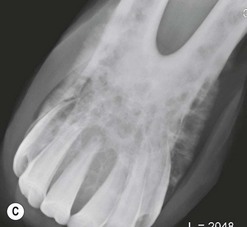
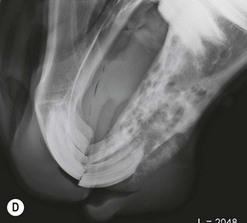
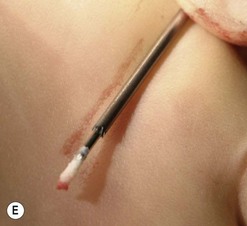
Fig. 11.12 (A) & (B) This 5-year-old Irish Draught mare was presented with a 4-week history of a mandibular swelling, gingival bleeding, weight loss and dysmasesis. The mandibular incisors were palpably loose and the mandible was warm to the touch and mildly painful on palpation. (C) & (D) Lateral and occlusive radiographs gave the suspicion of a neoplastic lesion. (E) A bone biopsy was taken from several sites, and the horse was diagnosed with osteosarcoma.
Differential diagnosis
Infection resulting in osteitis or osteomyelitis (particularly with Actinobacillus spp.) can be very destructive, and appear similar clinically and radiographically to osteosarcoma. Various cystic structures, such as ameloblastoma, ossifying fibroma, and fibrous dysplasia can be clinically similar but usually have characteristic radiographic differences.
Diagnostic confirmation
Their radiographic appearance is highly suggestive, but biopsy provides the only definitive diagnosis. There is a characteristic combination of cortical bone destruction and periosteal new bone formation giving the area a ‘sunburst’ radiographic appearance.
Bone biopsies of osteosarcoma are sometimes easy to obtain, since the bone is usually softer than normal and the medullary cavity is filled with diagnostically significant friable pink to white material containing variable amounts of cancellous bone (Fig. 11.12E). It is easy to miss tumor tissue in small bone biopsies, and florid, non-neoplastic reactive bone or fracture callus can easily be mistaken histologically for neoplasia. Multiple biopsies should, therefore, be collected from sites identified by radiography or CT or MR imaging methods, but this is not an easy procedure.
Pathology
Several histological types of osteosarcoma are recognized in other species where its incidence is higher, but this tumor is so rare in horses that it is probably unwise to extrapolate from these findings. The tumor tissue is, however, usually not densely cellular with formation of fibrillar stroma, bone or osteoid tissue. The cells have a high mitotic index and an atypical irregular morphology.
Since this tumor is so rare in the horse, the expected histological appearance is a speculative one based on experience of the lesion in other species. Osteosarcomas are characterized by painful bony swellings with variable degrees of bone lysis, tumor bone formation and reactive periosteal bone proliferation. The histological features are of neoplastic osteoblasts with variable numbers of osteoclasts. The extent of formation of tumor osteoid and/or bone is variable. The tumor bone may, therefore, be hard or soft and hemorrhagic.
Treatment
Radiation offers the only hope of success, but the tumors are likely to be locally malignant, and so treatment is usually not contemplated. Euthanasia is the only realistic option.
Prognosis
There are insufficient data for a reliable prognosis. Although metastasis is seldom reported in osteosarcoma at any site in the horse, it is impossible to predict anything about these very rare tumors. Some may progress relatively slowly and are, therefore, at least tolerable for limited periods. However, the highly aggressive nature and rapid course in most cases justify a hopeless prognosis.
Tumors of soft-tissue origin
Soft tissue tumors of the mouth are far more common than those affecting the teeth and facial bones. A wide variety of tumors have been reported, but only a few occur with any regularity. There is generally more information concerning the diagnosis, management, and prognosis for these equine soft tissue tumors than for calcified tumors. Many individual veterinarians have preferred treatments for most of the common soft tissue tumors, and some report good results while others are less successful with the same methods.
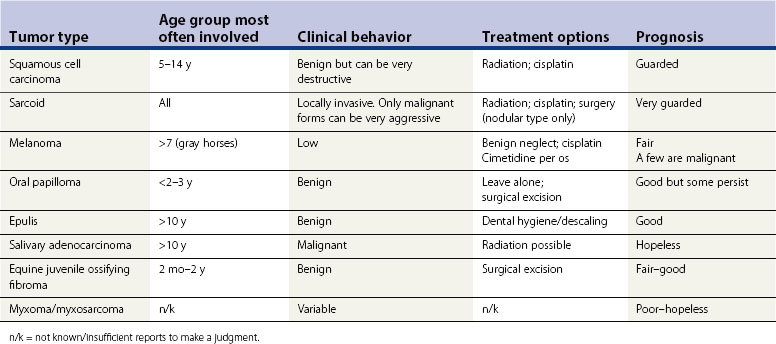
The clinical features of the main equine oral, soft-tissue tumors are summarized in Table 11.3.
Squamous cell carcinoma
Definition
A squamous cell carcinoma is a malignant neoplasm of stratified squamous epithelium that appears to have a predilection for mucosal junctions.
Occurrence
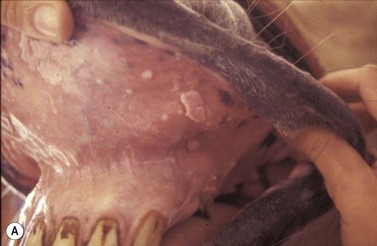
Squamous cell carcinoma (SCC) is probably the commonest oral neoplasm. Although mucocutaneous junctions are commonly affected with SCC outside the mouth, where there is an apparent correlation with non-pigmented skin and possibly with high levels of ultraviolet light, many of the most severe and aggressive SSC tumors occur within the mouth.46 The role of ultraviolet light in the pathogenesis of facial and lip carcinoma is uncertain, but the Clydesdale breed and horses with non-pigmented skin of the face and lips are more often affected than other breeds and colors. Putative carcinogens include chronic irritation, such as epulis, foreign body reactions, chronic wounds, and possibly dietary factors. Older horses are more likely to be affected.
There is often a suggestion that the primary tumor develops in the paranasal sinuses or nasal cavity and the destructive tissue involves the hard palate, but it may be difficult to identify whether the primary lesion is in the sinus or the hard palate.47 SCC possibly arises in chronically irritated hyperplastic alveolar epithelium in cases of chronic periodontitis.
Clinical features
SSC tumors are characteristically slow growing but occasionally have a more rapid course. They can be proliferative at least initially, but usually become very destructive, ulcerative and infiltrate widely into local tissues of the mouth including the lips, buccal mucosa, hard palate and tongue.48 Early lesions may simply resemble a non-healing wound but more advanced, destructive carcinoma lesions have a characteristic foul odour.
Metastases to local lymph nodes can occur, although the general sentiment is that they do not do so commonly.49 In theory, they may disseminate to the lungs and elsewhere. However, this behavior is rare in oral forms of SCC.50
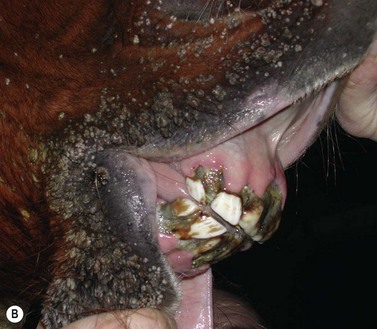
Oral SCC may involve the lips (Fig.11.13A), hard palate51 (Figs 11.13B,C), tongue51,52 (Figs 11.13D,E) or oral mucosa (Fig. 11.13F). It is also quite common for oral SCC to invade the nasal cavity and the paranasal sinuses (often to the point of gross distortion or obstruction to airflow).53 Some SCCs involve the base of the tongue (Fig. 11.13E) and pharynx (Fig. 11.13G) and can physically affect their function. Where the nasal cavity and paranasal sinuses are involved, there may be altered airflow (or even complete obstruction of the ipsilateral nostril). Horses with pharyngeal and lingual SCCs may present with dysphagia of progressive, insidious onset. In all cases, weight loss and poor general health are common.
Fig. 11.13 (A) A destructive carcinoma on the lip of a 23-year-old pony gelding. Note the extensive tissue destruction and the loss of the maxillary teeth. Treatment with intralesional cisplatin and topical 5-fluorouracil was not effective. (B) Facial distortion due to nasopalatine squamous cell carcinoma in a 12-year-old pony gelding. (C) A histologic section of the endonasal carcinoma shown in (B). Solid cords of squamous carcinoma are surrounded by fibrous stroma and trabeculae of non-neoplastic reactive bone (arrows). (D) Squamous cell carcinoma of the pharynx, which was identified some 3 months after a lesion had been detected in the hard palate. It is possible that this developed independently or that it was an extension of the earlier lesion. (E) Carcinoma of the free portion of the tongue. The local lymph node was enlarged (see J); slide courtesy of Dr R.R. Pascoe. (F) This highly destructive oral carcinoma developed in a 4-year-old Warmblood. It involved the maxillary bone and resulted in dental problems also.
(Courtesy of Chris Louf.)
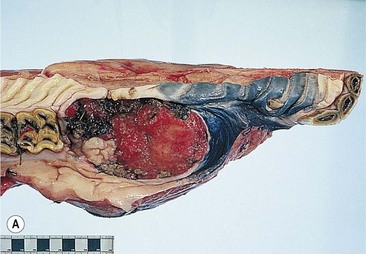
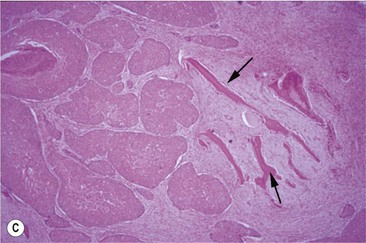
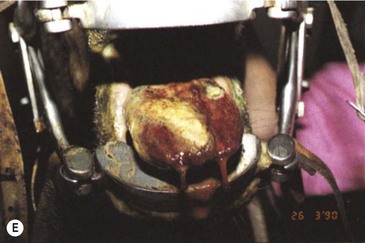
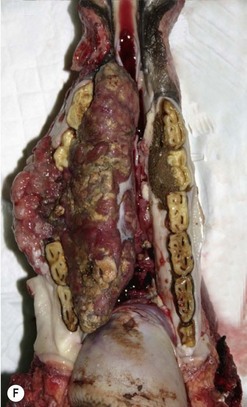
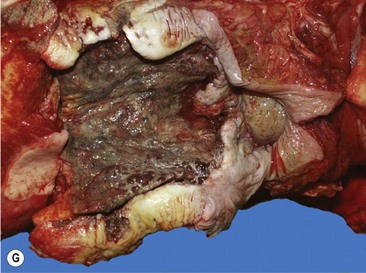
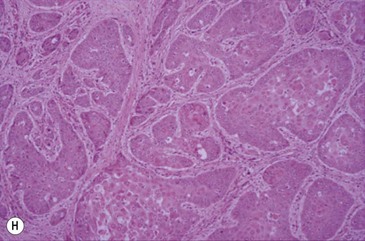
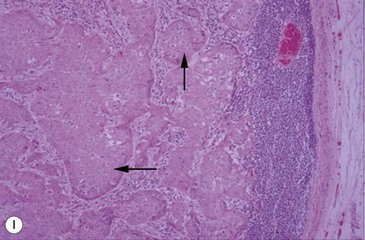
(G) This horse was presented with weight loss and dysphagia. The highly destructive oral and invasive carcinoma was not visible endoscopically from the pharynx and was only seen with difficulty during an oral examination. (H) Photomicrograph of cords of solid squamous cell carcinoma with deep invasion of the tongue. (I) Metastasis of squamous cell carcinoma to regional lymph node showing cords of tumor cells (arrows) beneath cortical lymphoid tissue.
More extensive spread may involve the orbit and the cranial cavity with secondary involvement, respectively, of the eye or even rostral brain when the cribriform plate is eroded by tumor. It is also quite common for nasal SCC to invade the hard palate, forming an oronasal fistula. In all cases, there may be extensive soft-tissue disruption and consequent loosening/shedding of the teeth.
The location of SCC means that tumors are frequently detected late when a large invasive mass may already be present projecting from the gum or hard palate as a grayish, ulcerated and bleeding mass. Where the tumor surrounds a tooth, this may become dislodged, and in almost all cases there is a fetid odor from the mouth. Involvement of the lips and gums is usually clinically obvious; early tumors are often identified incidentally during routine dental procedures.
Differential diagnosis
The differential diagnosis includes other proliferative and invasive soft tissue lesions of the lips, including equine sarcoid, hemangiosarcoma, basal cell carcinoma, and myxomatous tumors. Non-healing oral wounds may be confused with early cases, and carcinomatous changes may be a cause of non-healing.
Diagnostic confirmation
Biopsy is characteristic (Figs 11.13H,I). The pathologist should try to classify the degree of differentiation of the cells; highly differentiated carcinomas are far less dangerous than the undifferentiated ones. Biopsy sites should be carefully chosen to reflect the main carcinoma, with inclusion of a marginal zone as well. Punch or wedge biopsy sites should not be scrubbed or even washed before collection, and the biopsy site should not be sutured afterwards since wound dehiscence is almost certain. Fine needle aspirates and impression smears can be used but may be misleading, since they often consist mainly of stroma and inflammatory cells.
Radiographic examinations can be used to identify masses in the sinuses and the extent of bone destruction produced by invading carcinoma.
Pathology
Histologically, the tumor has distinctive characteristics with irregular cords of downward-invading neoplastic keratinocytes. (Figs 11.13H,I). A SCC characteristically has large amounts of non-neoplastic fibrous stroma in which inflammatory cells are plentiful. The abundant stroma results in a lesion that is characteristically tough or scirrhous when palpated or excised. The accumulation of variable amounts of keratin produces ‘keratin pearls’ in well differentiated tumors that can be used to define the likely malignancy of the lesion. Poorly differentiated carcinoma shows a more anaplastic cell structure, higher rates of division (high mitotic index), and much less keratin and, therefore, can closely resemble aggressive myxosarcoma. SCC is one of the better defined equine oral neoplasms.
Treatment
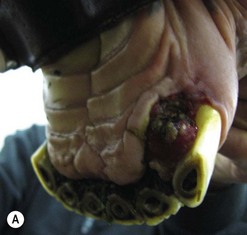
While surgical excision of oral SCC lesions has been reported to be successful,54 this treatment can be very difficult and in some sites is clearly impossible. There is a very high rate of recurrence following such surgery. Small discrete tumors may, however, be amenable to surgical removal if detected early (Fig. 11.14), and extensive excision involving hemimandibulectomy, such as has been described for other tumors of the jaw, may also possibly remove the tumor but may leave unacceptable cosmetic or functional deficits.
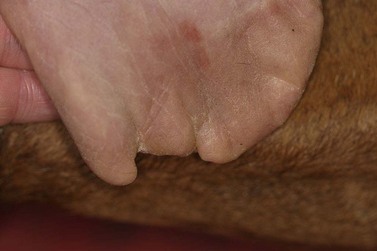
Fig. 11.14 A small hemorrhagic and destructive lesion was noted on the tongue of this 22-year-old gelding. Biopsy confirmed it to be squamous cell carcinoma, and surgical excision was performed with a safe margin of excision.
Squamous cell carcinoma appears to be relatively sensitive to gamma radiation, and this therapy offers the best prognosis, with a reasonably high success rate (DCK, unpublished observations). Teletherapy is logical and can be finely controlled, but repeated fractionated doses need to be used, and the horse, therefore, needs repeated general anesthesia. The number of centers where this can be performed is very small, and the procedure is necessarily very expensive. The much simpler 192-iridium interstitial brachytherapy using linear platinum-sheathed sources has been used to good effect (DCK, unpublished observations). There are serious logistic and human health risks involved with the procedure, however, and limitations on the size and location of the tumors that can be treated. Placement of the radiation source within the highly mobile tissues of the mouth carries serious dangers if the horse were to dislodge the wires and swallow them.
Recently, cisplatin, 5-fluorouracil, and mitomycin C have been used in various intralesional or topical formulations for treatment of cutaneous and other equine carcinomas. Some SCCs respond well to intralesional cisplatin,* either in water-soluble form with frequently repeated injections, as an emulsion of the solution, containing at least 1 mg/ml with an equal volume of sesame or almond oil,55 or in biodegradable bead or pellet forms. The use of the above drugs in treating oral SCC has apparently not been reported, but use of all these materials carries operator safety risks. And as such, their use should probably be restricted to specialist oncology centers. There is no justification in taking human health risks through their use by untrained personnel.
The response of equine SCCs to immunomodulation using mycobacterial protein materials, such as Bacillus Calmette-Guérin (BCG), is disappointing in horses when compared to treatment of squamous cell carcinoma in other species, such as cattle (DCK, unpublished observations). Treatment of labial SCC with 5 % fluorouracil cream applied topically has been shown to resolve some cases and improve others.57 It is, however, a very useful adjunct to other forms of treatment and may be particularly applicable to small, ulcerated, buccal or lip lesions.
Prognosis
The tumors are always locally invasive but usually slow to metastasize, so while the clinical prognosis is inevitably poor, many cases can survive long periods even with quite extensive oral involvement. Oral SCC is a low-grade invasive tumor that tends not to metastasize beyond the local lymph node. However, it is probably unwise to assume that this will be the case in all affected patients. Secondary complications such as facial or oral distortion and destruction, dysphagia, loosening of teeth, and nasal obstruction inevitably suggest a poorer prognosis.
Sarcoid
The equine sarcoid is the commonest cutaneous fibroblastic tumor of horses. It commonly involves the cheeks and lips.58,59 The term sarcoid is used clinically to describe a spectrum of cutaneous tumors that variously involve connective tissue and epithelium with a range of clinical behavior.60 There are usually two distinct forms that affect the mouth itself (as opposed to the skin of the lips and cheeks). The nodular form remains subcutaneous, and is most often located at the angle (commissure) of the mouth or the cheeks and may extend into the mouth (Fig.11.15A). The verrucose form often involves the perioral skin. In the perioral skin and the tissues of the cheeks, in particular, it often has a highly invasive behavior, then being classified as malignant sarcoid;60 it can also ulcerate into the mouth.
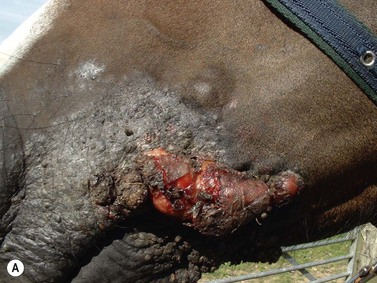

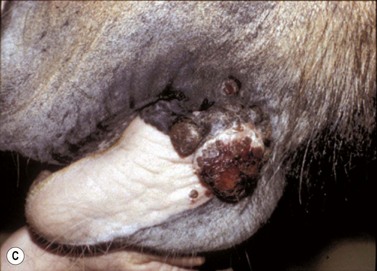
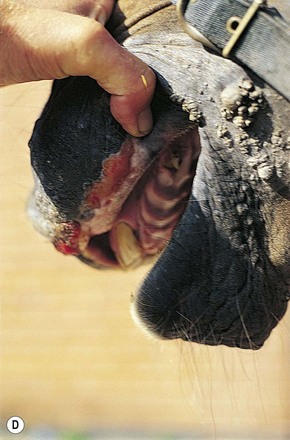
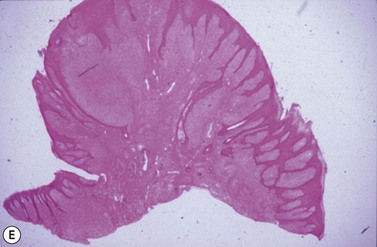
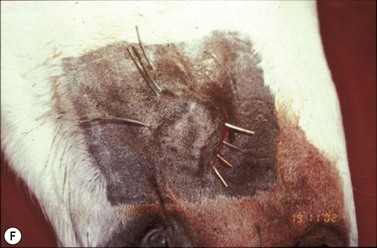
Fig. 11.15 (A) This locally invasive sarcoid also involved the buccal surface of the cheek. (B) A localized ulcerated nodular sarcoid. (C) A mixed sarcoid with verrucose and ulcerated nodular components. (D) A locally invasive malignant sarcoid in the cheek with extensive deep involvement of the muscles and oral mucosa. (E) Histologic section of verrucose sarcoid showing a bulging exophytic lesion with pseudoepitheliomatous hyperplasia of the epidermis and diffuse subepithelial fibroblastic proliferation. (F) Linear iridium-192 interstitial brachytherapy was successfully used to treat an invasive sarcoid in the cheek.
Clinical features
Intracutaneous or subcutaneous nodules on the cheek or within the lips having an ulcerated surface are the commonest oral manifestation of sarcoid (Figs 11.15B,C). The nodules frequently ulcerate either on the cutaneous surface or into the mouth. The verrucose form is also a common type in the perioral skin but does not often involve the oral mucosa; however, where the lesions are mixed, a deeper component can be expected and should be sought carefully. Combinations of nodules within the skin and cutaneous involvement of verrucose sarcoid are also common. Nodules may extend through the cheek musculature into the oral mucosa. The fibroblastic forms also occur in the perioral tissues and appear as a fleshy, ulcerated mass of friable and easily traumatized tissue that is very similar to granulation tissue; this form often develops following trauma or failed/ partial treatment attempts. The malignant form usually comprises various combinations of the sarcoid types but is highly invasive and nodules may be linked by cords of sarcoid tissue (Fig. 11.15D).
Primary sarcoid has not been reported on the tongue, gingiva or palate.
Differential diagnosis
The equine sarcoid can resemble some forms of viral papilloma, and the nodular forms may be also mistaken clinically for melanoma and mastocytoma. In addition, the fibroblastic and nodular forms may resemble oral fibroma, inflammatory nodules (e.g., foreign body and parasitic granuloma) and granulation tissue arising from any cause.
Diagnosis
Usually an intuitive tentative clinical diagnosis can be made. Horses with a single sarcoid lesion located in the mouth without any other lesion are very unusual. Horses that have the characteristic features and show lesions at other sites can usually be assumed to have sarcoid lesions.
Biopsy is not usually recommended because there are recognized dangers with biopsy of sarcoids.62 However, the histological features are characteristic and provided that a suitable contingency plan for treatment is ready prior to the results of biopsy, then it can be a logical diagnostic step.
Pathology
The clinical term ‘sarcoid’ encompasses a histological spectrum of fibroblastic tumors that may be accompanied by a variable epithelial component. The fibromatous variant is grossly well circumscribed, solitary or multiple, with a tough, pale, fibrous cut surface. Adjacent epidermis is often attenuated and may be intact or ulcerated. Most of the lesion consists of randomly arranged, well-differentiated fibroblasts with plentiful collagen. The malignant variant has ill-defined margins; adjacent epithelium may be ulcerated or intact. Histological features are of randomly arranged, activated fibroblasts that form interlacing bundles and whorls. Individual tumor cells have degrees of anisokaryosis, and mitoses may be plentiful. At the histological level, it may be difficult to discern the limits of the tumor, especially in small tissue samples. The verrucose sarcoid has histological features similar to those in the malignant form and is usually associated with marked pseudoepitheliomatous epidermal hyperplasia (Fig. 11.15E).
Treatment
The options are limited. In some cases, the lesions can justifiably be left alone in view of the risks of exacerbation by incomplete excision. However, inadvertent trauma can also result in severe deterioration, and so early treatment may be strongly recommended in most cases.
Treatment of buccal forms of the disease is notoriously difficult, with radiation, cryosurgery, hyperthermia, laser excision, and intralesional cisplatin carrying some chance of success. Referral to a specialist center is probably justified simply on the grounds that failure of a treatment may result in significant exacerbation of the lesion.
The best treatment is undoubtedly with radiation either as brachytherapy using interstitial linear or pelleted radioisotopes with a gamma emission capability. Radiation has a cure rate of over 95 %, and the cosmetic results of this method are impressive. The most frequent isotopes used include iridium-192 and gold-198. The former is presented in linear sources, sheathed with platinum that renders the isotope effectively a total gamma emitter. Gold-198 is used as pellets and has a very short half-life (48 hours) and so this is logistically easier to handle (the sources do not need to be removed) but clearly this method carries much higher operator risks than the lower emissions over a longer period characteristic of iridium. Linear iridium sources are left in situ for the calculated period to deliver the required radiation dose and are then removed (Fig.11.15F). During the treatment time, the horse must be confined within an approved radiation unit. Complications involving wire displacement and injury or colic during the treatment period can add considerably to the logistic problems. Teletherapy is an ideal method of treatment, but there currently are very few facilities for this treatment.
Other treatments all carry a worse prognosis, with surgery being the most difficult. Intralesional cisplatin using stable emulsions with sesame oil has recently gained some reputation,63 but the method carries very serious carcinogenic risks for operators and handlers alike. Biodegradable beads containing cisplatin (Matrix II, Royer Inc, USA) or sponges with 5-fluorouracil have been used to generally good effect but correct placement is critical, and retention of beads or sponges is sometimes a problem. Cisplatin use should be restricted to specialist institutes where facilities for fecal and urine disposal ensure that risks to people are minimized. The risks with the bead systems are markedly reduced.
Topical cytotoxic chemicals, such as 5-fluorouracil, imiquimod (Aldara, Graceway Pharmaceuticals, LLC Bristol, UK), and Xxterra (20% zinc chloride and Sanguinaria canadensis root extract paste) have also been used with variable results. They all require repeated applications, and penetration of lesions is difficult, so failures are common. Scarring is a major hazard particularly if the cheeks are involved and functional difficulties can arise.
Intralesional immune ‘stimulants’, such as mycobacterial cell wall extracts or BCG, can be effective in some nodular or fibroblastic forms, but the prognosis is far worse than the corresponding results from treatment of periocular sarcoids of the same type.63 Treatment is tumor-volume related, and so large lesions require more BCG material and, of course, each individual lesion requires its own injections. Repeated injections are invariably required, and each one carries the risks of causing anaphylaxis. The possibility of anaphylactic reactions can be reduced (but probably not eliminated) by premedication with flunixin meglumine and dexamethasone intravenously some 15–30 minutes before the procedure is carried out. Ensuring that true intralesional injection has occurred can also reduce these risks.
Prognosis
The prognosis for any sarcoid treatment is very guarded. Recurrences are common, and new lesions can also develop in many sites. While the malignant form is less common than the other variants, it carries a very poor prognosis. The cheek area seems prone to the malignant form. The prognosis for oral or facial sarcoids is also related to the loss of effective work use as a result of interference with tack. Lesions and scarring as a direct result of treatments at the angle of the mouth or in the cheeks can adversely affect the use of bits and harness. Repeated trauma from harness results in continued exacerbation, and so the tumor and the horse become increasingly difficult to manage.
Melanoma
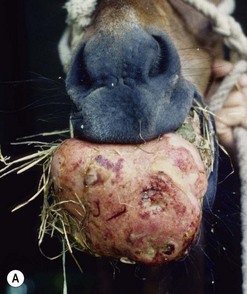
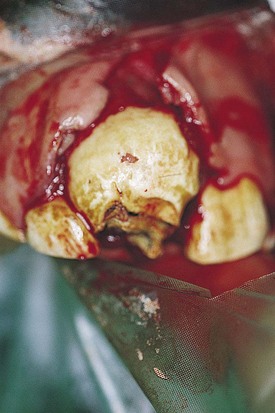
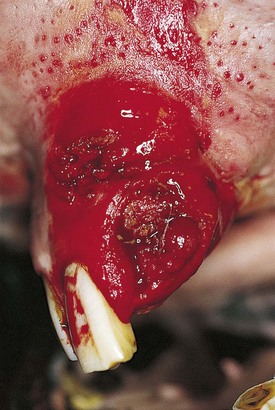
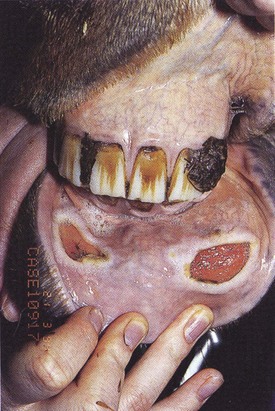
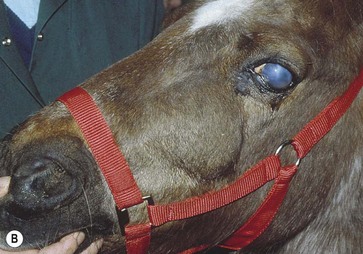
These are tumors of melanocytes occurring in the skin and in other organs (including the mouth and cheeks). The nomenclature of melanocytic masses in the horse is confused and contradictory. There is a spectrum of benign and malignant tumors involving melanocytic cells that are predominantly encountered in gray horses – indeed most gray horses over 5–8 years old have melanomas at some site. Rarely, horses of other colors are also affected. The lips are a relatively common site, but tumors in the cheeks (masseter muscles), gingivae, and tongue may occur.
There is a strong tendency for melanoma to develop in the parotid salivary glands and associated lymph nodes. Tumor development in these sites is usually obvious on clinical inspection. It is not easy to characterize the degree of malignancy in melanomas without resort to biopsy, and even histology may not always provide a firm prognosis. The large majority of melanomas are benign, but some have an aggressive appearance and aggressive growth rate; these usually have a correspondingly aggressive histopathological appearance. Generally it is accepted that small, early lesions are benign, but that, with time, most become more malignant (whether or not they invade locally or metastasize to remote sites).
Clinical features
Oral melanomas are usually benign and expand slowly but even histologically benign variants can reach considerable size. Surprisingly, tumors on the lips (Fig.11.16A) and gingivae are often only noticed when they are large. They usually do not cause systemic effects (unrelated melanomas may, however, develop simultaneously in other organs). Extensive lesions can develop in the parotid and pharyngeal lymph nodes and may extend into the parotid salivary gland either directly or by contiguous spread (Fig.11.16B).
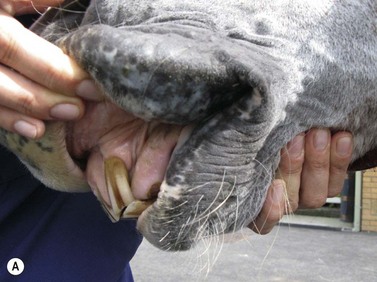

Fig. 11.16 (A) A large melanoma in the lip of an aged gray horse. The lesions expanded slowly but the horse remained symptom-free for years in spite of superficial ulceration. (B) Salivary gland melanoma.
Melanomas affecting the masseter muscle and those that ulcerate into the mouth usually affect mastication, even causing weight loss and dymasesis. In spite of the large size of some of these lesions, the clinical effects are usually minimal and relate simply to their space-occupying nature (Fig. 11.17).
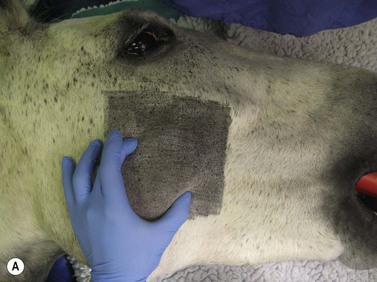
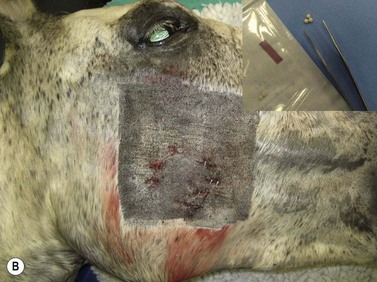
Fig. 11.17 (A) This 9-year-old gelding was presented with weight loss and dysmasesis. The mass in the right masseter muscle, and a similar mass in the left masseter muscle were palpably obvious. (B) Cisplatin biodegradable beads (Matrix II Royer Inc USA) were implanted into the lesion with moderate improvement over 6 months. A second implantation was performed with only a modest further improvement. Insert: biodegradable cisplatin impregnated beads.
Differential diagnosis
Equine sarcoid and mast cell tumors should be considered in the differential diagnosis of lesions that develop in haired skin adjacent to the lips. Parasitic and foreign body granulomas and oral penetration-induced abscesses or granuloma have a similar clinical appearance.
Diagnostic confirmation
The diagnosis of melanoma is easy to establish from clinical features and, if necessary, by use of a fine-needle aspiration or hollow-needle biopsy.
Pathology
Melanomas are bulging, well-circumscribed, gray-black masses that compress adjacent soft tissues, sometimes producing ulceration of the compromised surfaces. Cut surfaces of the tumor are usually glistening, firm, and uniformly black. Occasional lesions may be less homogeneous, less pigmented, or even amelanotic.
Most equine melanomas are characterized histologically by the presence of myriad round-to-oval cells with plentiful, densely pigmented cytoplasm. Two distinct types of cells constitute the tumor:
1. Melanin-producing cells (melanocytes)
2. Macrophages that contain phagocytosed melanin (melanophages).
Pigmented tumor cells may extend into adjacent soft tissues but this is not a reliable histological criterion of malignant behavior. Most tumor sections have to be bleached so that the underlying cytological features can be assessed. Nuclei are usually solitary with a large nucleolus and coarse nuclear chromatin. Mitoses are usually sparse.
There are occasional melanomas with clinical features of malignancy (invasion and metastatic spread); these have corresponding histological features of anisocytosis and anisokaryosis, with plentiful mitoses and variable cytoplasmic pigmentation. The diagnosis of such less well-differentiated melanoma may be clarified by the use of immunostaining against cell markers, such as Melan A and S100.65
Treatment
Many melanomas are left alone without any significant problem apart from the cosmetic aspects. For lip melanomas, surgical excision is sometimes feasible, especially in early cases, and is then usually effective, provided that a surgical margin can be achieved. Regrowth at the site can be a problem because during surgery minute tumors may be seen in the locality, and it is almost inevitable that some of these will be left to develop later. Often, however, by the time functional defects are present, surgical treatment options are very limited. This is complicated by the long-term ‘conventional wisdom’ that melanomas should be left alone and that there is a risk of metastasic dissemination if surgery is performed. The reality is that pathologic descriptions confirm that very small lesions are very benign and so are a reasonable surgical option. In contrast, advanced lesions often have a malignant histological appearance, and so, at this stage, the risks are far higher. There is some justification, therefore, in the concept that small lesions in accessible sites should be removed as soon as feasible so that at least these particular ones will not become a significant clinical problem and will not become malignant!
Prolonged daily oral administration of cimetidine at doses of 7.5–15.0 mg/kg bodyweight has been suggested as being effective, but the results are not convincing in many cases.66,67 Treatment of single or few oral tumors on their own probably does not warrant this approach.
The use of repeated intralesional cisplatin either in stable emulsion with sesame oil and water, or more sensibly in biodegradable beads (Matrix II, Royer Inc, USA), may bring some improvements in some lesions (Figs. 11.17A,B), but complete cures are most unlikely. Radiation therapy is not usually effective against melanoma.
Prognosis
The prognosis necessarily depends on the location, the particular pathological characteristics and the extent of secondary effects. The prognosis is usually relatively favorable as the majority have no metastatic tendency, but occasionally tumors are locally invasive, and others are very aggressive with extensive metastatic spread and serious secondary effects. As might be expected, older lesions tend to be more malignant, at least in histological character and possibly clinical behavior.
Oral papilloma
Viral papilloma is a relatively common occurrence on the skin of the mouth and lips and, in some cases, they can extend into the oral cavity. Host-specific equine papilloma viruses are the causative virus and usually affect horses in their first year or two of life. Less commonly, they affect older horses and particularly those that have an immunocompromising disorder, such as pituitary pars intermedia dysfunction (PPID/ equine Cushing’s disease).
Clinical features
Typically, papillomata appear as single or multiple, discrete or coalescing, verrucose, gray-pink papules around the mouth and, occasionally, on the mucosa of the mouth (Fig.11.18A). They seldom ulcerate unless they are traumatized. A few cases have lesions restricted to the oral mucosa, and it is not known if these reflect a different manifestation of the same infection. In a few cases, there may be a severe oral infection, and in these cases it is likely that some immunocompromising state exists (Fig.11.18B).
Differential diagnosis
Viral papilloma can be mistaken for some forms of the equine sarcoid, but as the latter seldom occur in the verrucous form in the mouth, differentiation should be simple. ‘Skin tags’ also occasionally develop in the mouth.
Diagnosis
The diagnosis is usually simply based on epidemiology and clinical appearance. Biopsy is characteristic, and most lesions resolve with age, although they may be very persistent in older horses.
Pathology
Papillomas are characteristic exophytic verrucose lesions that may be superficially ulcerated and inflamed. Histological examination reveals filiform fronds of hyperplastic epithelium on fibrovascular cores that often contain plentiful lymphocytes and plasma cells. Intranuclear inclusions may be plentiful or sparse.
Treatment
Most papillomas resolve spontaneously over some months, but individual lesions may persist, often for many years. Therapeutic measures used have included autogenous vaccines prepared from surgically excised lesions and various topical chemicals, including podophyllin and formalin gels. Many of these treatments are impractical on intra-oral lesions and, in those circumstances, troublesome papillomata can be removed surgically or with cryonecrosis.
Epulis
The term epulis is a clinical description of a smooth gingival nodule and can encompass different types of lesion, such as hyperplasia, granuloma, or even neoplasia. These tumor-like masses develop from the fibrous tissue of the gingiva and are often, but not always, associated with the accumulation of dental calculus caused by chronic local irritation due to persistent periodontal infection. In horses, they are much less common than in some other species, such as the dog and cat, and seldom reach significant proportions (Fig.11.19A).
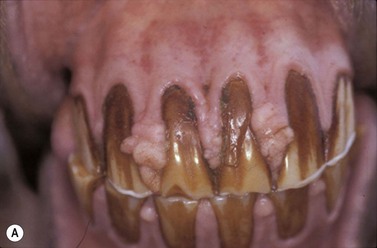
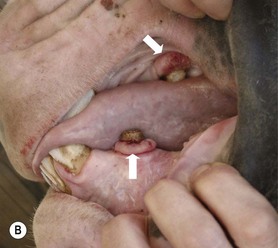
Fig. 11.19 (A) Benign, gingival hyperplasia epulis in an aged horse. The lesions were noted incidentally and were symptom-free. (B) Small epulis-like gingival proliferations are present around the base of the upper and lower canine teeth (arrows). After removal of the calculus these inflammatory lesions disappeared.
Clinical features
Usually epulis appears as a benign expansion of the gingival epithelium resulting in thickening and prominence of the gingiva particularly evident at the gingival margin where it may be focally raised (Fig.11.19B). A few cases develop ulcerated proliferating masses at the site that can be much more like a true neoplasm, and these may closely resemble oral fibroma.
Differential diagnosis
Viral papilloma, sarcoid, and the proliferative forms of squamous cell carcinoma are the main differential diagnoses; all are easily identified histologically after surgical removal.
Diagnosis
The condition is distinctive clinically, but refinement of the clinical diagnosis depends on histological examination of resected epulides or a biopsy. Biopsy may, however, be misleading if granulation tissue is present, and in any case they may still be very similar histologically. The main differential diagnoses are, however, usually easily identified histologically.
Pathology
There is poor histological characterization of the epulides, probably because they are easily recognized and are seldom treated: few are examined histologically.
Treatment
Removal of the causative factors usually results in complete resolution. Many cases resolve spontaneously after any dental calculus or damaged dental or adjacent soft-tissue structures are removed. Recurrence of both calculus and epulis should encourage a careful assessment of the whole horse in case there is any systemic disease present.
Prognosis
The prognosis is excellent. There are no reports that they are precursors to more malignant tumors, such as squamous cell carcinoma, but one case developed a locally aggressive carcinoma some years after a benign epulis was removed from the base of a canine tooth (DCK, unpublished observation). It is probably unwise to draw any causative inference from this single case.
Oral fibroma
Definition
This is a well defined exophytic fibroma that occurs with some regularity in the horse. The clinical similarity of this tumor with sarcoid and epulis can be diagnostically confusing. Possible causes include persistent local inflammation due to periodontal infection or calculus build-up, or foreign body reactions (similar to epulis). However, many of these lesions occur in otherwise normal mouths. Most are slow-growing and symptomless; almost all early cases are detected incidentally during routine dental examinations. The benign forms may progress to invasive fibrosarcoma.
Occurrence
There are few reported cases of oral fibrous tumors in horses in spite of their relatively common occurrence. Older or mature horses appear to be more often affected. The buccal gingivae of the maxillary cheek teeth are the commonest site, but they occasionally occur on the lingual margin and in the mandibular arcades.
Clinical features
A localized, fleshy, ulcerated and hemorrhagic mass is easily visualized lying usually alongside the maxillary cheek teeth (Fig. 11.4A,B). There may be some external distortion of the cheek outline with larger growths. Dysmasesis or hemoptyalism may be noticed by an astute owner.
Diagnostic confirmation
Biopsy is essential to differentiate it from squamous cell carcinoma, sarcoid, and epulis, in particular.
Pathology
There are two forms of the oral fibroma. The first is a result of irritationally induced changes and is not a genuine neoplasm (see epulis above), and the second is a genuine (usually benign) neoplastic change in the fibroblasts. Pathologically, the latter are usually firm nodules that develop as aggregated protuberant tumors within a firm indurated plaque. They often ulcerate. Histologically they are very cellular with well differentiated fibroblasts arranged radially within a sparse extracellular matrix. Mitotoses are usually evident but are not as common as in the more aggressive forms of fibrosarcoma. There may be deep extension into the subcutaneous fat. They are histologically distinctive and cannot really be confused with sarcoid.
Treatment
Treatment options are limited to surgical excision. Laser surgical excision or diathermy are effective and produce a relatively bloodless surgical field. It is important to clean/descale adjacent teeth of calculus at the same time in case the calculus acts as a focus for growth of a new mass.
Ossifying fibroma
Definition
An ossifying fibroma is a poorly defined proliferative, fibro-osseous, tumor-like, solitary lesion, that typically develops in the rostral mandible, of younger horses in particular.
Occurrence
Most cases are reported in horses less than 1 year of age. They may, however, not be observed until the horse is handled at a later age, by which time there may be significant ulceration of the buccal mucosa and distortion of the underlying tissues.68
Clinical features
The majority of these tumors occur unilaterally in the rostral mandible. They rarely develop in the maxilla and in very rare cases can be bilateral. They often reach considerable size and, initially, are covered by a domed, smooth/normal oral mucosa. Gross distortion of the lip and the associated teeth is likely. Later some ulceration is common (Figs 11.20A,B). There is some variation in the clinical presentation, with some lesions being flatter and fleshier in appearance; they can then resemble other soft tissue tumors. The expanding lesion also causes loosening of the teeth and consequent difficulty with prehension.
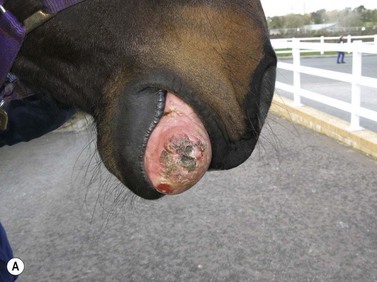
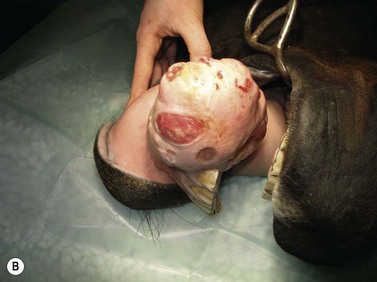
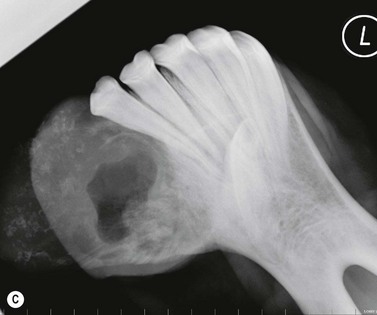
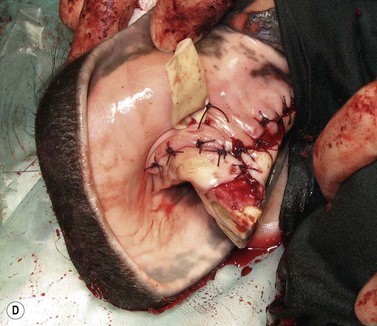
Fig. 11.20 (A, B) This ossifying fibroma was presented in a 3-year-old mare. (C) Radiographically the mass was found to involve the incisor teeth with loss of 203 and it had an obvious radiolucent center. A hemimandibulectomy (D) was performed with a good outcome.
The tumor can predispose the mandible to pathological fracture. Although the lesions may frequently be clinically obvious, they may only be identified late in their course, probably because there is little indication to examine the mouth of many young horses.
Diagnostic confirmation
Radiographically the dense tissue is obvious with only some lesions showing the calcification more commonly encountered in other species (Fig. 11.20C). More ulcerated and secondarily infected lesions may resemble other soft-tissue tumors.
Pathology
Ossifying fibromas may develop as alterations in the growth characteristics of the periodontal membrane or the developing teeth. The masses are reported to arise from a sessile base on the surface of the bone and expand to replace and displace normal structures with dense fibrous or fibro-osseous tissue. The lesion has a dense, tough, well-circumscribed appearance; it may be extensively mineralized and difficult to cut. There is a characteristically abrupt histological transition from fibroblastic stroma to osteoblasts, which form spicules of osteoid. The dense gritty nature of the mass sometimes makes biopsy difficult.
Treatment
Surgical excision is curative provided that sufficient attention is paid to identifying the true extent of the abnormal tissue. Extensive surgical debulking followed by cobalt-60 teletherapy radiation and treatment using cobalt-60 teletherapy alone in a standing sedated horse have also been used successfully.69 Hemimandibulectomy or hemimaxillectomy is also an effective option, particularly if the mandibular symphysis can be retained (Fig. 11.20D). Limited disabilities and acceptable cosmetic effects can be expected following treatment of more localized lesions. Cases subjected to this surgery should recover well and will usually cope well and lead active normal lives.69 However, if extensive excision is required, it may leave unacceptable cosmetic and functional deficits.
Prognosis
Regrowth of the tumor is common because of the difficulty of identifying the margins of the abnormal tissue. All resected tissue should, therefore, be submitted for histological examination, with particular attention paid to examination of the surgical margins, which need to be identified by the surgeon for the pathologist.
Myxomatous tumors of the jaw and gingivae
Definition
These are very rare tumors derived from embryonal connective tissue. The tumors are identified by their characteristic histological appearance.
Occurrence
In the few reported cases, older or mature horses appear to be more often affected. The incisor and molar dental arches of the maxilla are the most common sites.
Clinical features
These tumors are characteristically destructive (Fig. 11.21). The combined destruction and proliferation of tumor tissue creates obvious distortion of the maxilla, with secondary nasal and sinus obstruction. Loosening of teeth and infection of alveolar bone may later result, but in the early stages there is usually little bone destruction (Figs. 11.22).
Diagnostic confirmation
Biopsy is essential. Radiographically, there is an aggressive lytic appearance of these lesions, with a diffuse mixture of bone and soft tissues, often in a partially loculated form. The cardinal radiographic signs of the more malignant forms, however, are the combined destruction of normal bone and bizarre irregular new bone formation in random arrangement. The radiographic appearance can be very similar to osteosarcoma (see above) and squamous cell carcinoma.
Pathology
This group of tumors includes a spectrum that extends from benign myxoma to malignant myxosarcoma. The tumors have a soft gelatinous gross appearance, and may be highly infiltrative with a tendency to metastasize. Cut surfaces of the tumor may be lobulated and slimy. Histologically, the lesion contains characteristic stellate cells with abundant, amorphous extracellular matrix. These tumors can also resemble severely ulcerated juvenile ossifying fibroma, but the latter are usually slow growing and expansive rather than destructive. Additionally, ossifying fibroma has a characteristically different anatomic site, usually involving the rostral mandible.
Treatment
Treatment options are very limited – the margins of the tumor and their usual anatomical site make surgical excision virtually impossible. There are no definitive reports of metastatic spread of the malignant forms of these tumors, but this may reflect the short clinical duration, which inevitably results in euthanasia before secondary tumors could develop elsewhere.
Oral hemangiosarcoma
This is a malignant neoplasm of endothelial cells that can arise in any part of the body. They are reported to metastasize early, and so tumors in any locality may be primary or secondary. There are few reports of this tumor in the mouth.70,71 Aged horses are more likely to develop them, and there may be concurrent tumors at other sites, such as around the eye (DCK, unpublished observation).
Clinical features
A red or purple ulcerated mass in the oral mucosa (on the sides of the tongue or the gingiva is typical). The mass is likely to be slow-growing and, being subject to repeated trauma, there may be oral hemorrhage. The lesions may be identified incidentally during a clinical examination, but where periocular lesions occur, the clinician should carefully examine all the visible mucous membranes, including the mouth, for other evidence of the tumor.
Differential diagnosis
Foreign-body reactions and ulceration arising from the attachments of Gastrophilus sp. may be similar.
Pathology
Histological features are of a solid, soft-tissue sarcoma with marked anisocytosis, anisokaryosis, and myriad mitoses. There is variable formation of large or small vascular channels lined with neoplastic endothelial cells. In histologically equivocal cases, immunohistochemical staining for Factor VIII-related antigen helps in identifying neoplastic endothelium. Tissue margins of resected lesions should be assessed carefully to ensure that excision is complete, although the latter may be difficult for tumors of the gingiva.
Salivary adenocarcinoma
Salivary gland neoplasia is uncommon in horses. For example, in one study of 687 necropsies and 635 biopsies only three salivary tumors were described; two were carcinomas and one was a cystadenoma.4 Another review of 1148 equine tumors included only two salivary adenocarcinomas.72 Six of 14 documented equine salivary gland tumors were adenocarcinomas; where recorded, the sites of involvement were the parotid salivary gland (three cases) and the submandibular salivary gland (one case). The age range was 7–18 years.73 Recognition is important because they are reported to be highly malignant, and up to 33 % may develop pulmonary metastases.
Clinical features
Recognition of salivary tumors is difficult, but, clinically, the affected gland becomes enlarged. Palpation is uncomfortable but not painful. Only one gland is involved, and so the enlargement is asymmetrical, which differentiates it from the more usual very benign and transient parotid salivary gland swelling commonly seen in grazing horses.
Differential diagnosis
Benign swelling of parotid salivary glands occurs frequently in grazing horses. This idiopathic condition is invariably transient and self-resolving. Obstruction of the salivary ducts by sialoliths (usually in the parotid gland) occurs within the gland substance or in the more rostral part of the duct. This results in a solid obstructive lesion with generalized glandular swelling. This remains hot and painful for some days before it subsides naturally as atrophy of the gland follows.
Diagnosis
Diagnosis is reliant upon biopsy, but so few cases have been described that a characteristic histological appearance is not established. Post-mortem histological features are described.15
Disorders of the jaws and teeth resembling neoplasms
Mandibular aneurysmal bone cyst
Although localized (usually very small) bone cysts are relatively common in long bones and in particular in their epiphyseal regions, mandibular cysts are very rare in horses. In man, they probably arise from circulatory disturbances within the bone structure74 or traumatic alteration of the blood supply to a small area of bone.75 While such structures might be expected in young horses, they may also occur in older horses, possibly as a result of trauma.10 There is no evidence to suggest that these lesions are truly neoplastic, but diagnosis based on radiographic examination alone is probably unwise. While there are no reports of concurrent neoplasia in the horse, complexes of tumors and cysts are reported in other species.
Clinical features
These bone cysts present as sterile, firm, expanding swellings usually on the horizontal ramus of the mandible (Fig. 11.23 A,B). The mandible becomes thickened from progressive destruction of cortical bone accompanied by reactive periosteal new bone formation. Some pain associated with mastication may develop, and teeth may be shed as their alveoli are destroyed. The region may be warmer than the normal contralateral mandibular ramus. The rate of expansion may be sufficient to cause disruption and bleeding into the surrounding tissues and may easily suggest the presence of a neoplastic lesion. As the destruction becomes more aggressive, the mandible may suffer a pathological fracture.

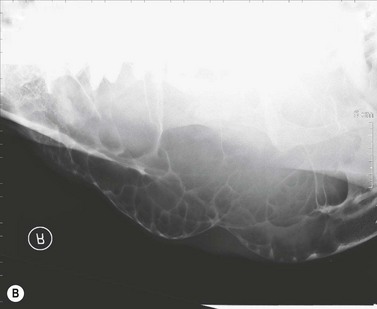
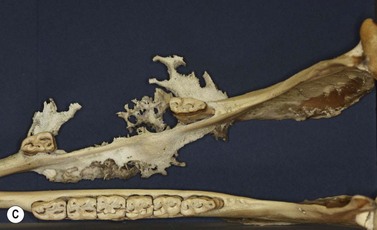
Fig. 11.23 (A) This 19-year-old pony mare developed a non-painful thickening of the horizontal ramus of the mandible. Gross distension of the proximal portion of the parotid duct and outward displacement of the masseter muscle were obvious. After some 12 months, she became painful and ate slowly. (B) Radiographs showed a multilocular cyst-like condition, and aspiration produced around 250 ml of a clear yellowish fluid with low cell count. (C) Gross destruction of a mandible caused by an aneurismal bone cyst.
In advanced cases, the extent of bone destruction can be considerable (Fig. 11.23C), and pathologic fractures are now likely. Although parotid duct obstruction can occur, this is an insignificant clinical event.
Differential diagnosis
Ossifying fibroma and destructive ameloblastoma are the main differentials. Paranasal sinus cysts have a similar radiographic appearance but occur in a very different location.
Diagnosis
The radiographic appearance reveals a complex, loculated, cyst-like structure containing bone fragments and soft tissue, usually with a fine rim of thin bone around the periphery (Fig. 11.23B). The structure closely resembles a complex paranasal sinus cyst, which may also arise congenitally or develop at later ages. Aspiration of the affected mandible reveals small volumes of yellow or red-orange fluid.
Pathology
Histologically the cystic lesion contains bone fragments, granular debris, siderophages, multinucleated giant cells, and fibrovascular tissue with areas of organizing and free blood. Histological examination of curetted fragments of cyst contents may often correlate poorly with the radiographic appearance.
Treatment
The only effective treatment is deep and aggressive curettage of all abnormal tissue. The defect may be filled with cancellous bone grafts collected from a remote site. Repair may be slow, and the site may be cosmetically compromised, but some cases can be cured effectively. However, in most cases, treatment is not justified, and palliative care should be provided until euthanasia can be justified.
Prognosis
The prognosis is poor unless the lesions are detected early and treated with aggressive surgical ablation and suitable reconstructive measures. However, the chances of early diagnosis are low, and so the prognosis for these lesions is usually bleak. Complications including tooth loss and pathological bone fractures reduce the prognosis considerably.
Non-aneurysmal cystic lesions of bone
Non-aneurysmal cystic lesions of the jaws also present as bony swellings that may affect mastication. Radiographically the cysts differ from the multiloculated destructive aneurysmal cysts in being rounded with a smooth bony lining and radiolucent center. The generic term, odontogenic cyst, is appropriate for these non-inflammatory cysts lined by epithelium.23 Radicular cysts occur adjacent to tooth roots. They are reactive lesions associated with oral inflammatory disease. Histologically they are lined by stratified squamous epithelium with associated inflammatory cells.
Fibrous dysplasia
Fibrous dysplasia of the bones of the skull has been reported in man,76 and a clinically similar condition is recognized in horses as a smooth contoured bone deformity arising from loss of bone structure with extensive formation of fibro-osseous matrix (Fig.11.24). The lesion is probably not a true neoplasm, and its major effects in the jaw/face region are due to the expansive space-occupying nature of the slowly expanding mass. The changes are easily recognized histologically but may be confused with neoplastic lesions both radiographically and clinically. Suspicious masses should be subjected to the full range of diagnostic tests, including radiography, gamma scintigraphy, biopsy and, where feasible, computed tomography.
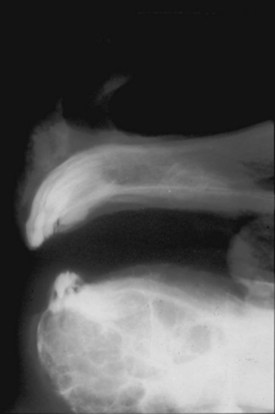
Fig. 11.24 A radiograph of fibrous dysplasia of the rostral portion of the mandible of a young horse.
Other tumors that may affect the mouth and jaws
Oral tumors arising by metastatic spread from remote organs and by invasion of the oral cavity from neighboring areas, such as the nasal cavity and paranasal sinuses can occur in horses. Tumors remote from the mouth and oral structures may have a serious influence on the mouth through metastatic spread (such as in hemangiosarcoma and lymphosarcoma). Secondary involvement of the mouth is, however, a very rare event, even in highly malignant tumors and in any case, the oral signs may be trivial compared to the other systemic involvements. The mouth may be secondarily diseased through systemic effects from functional tumors such as the pituitary adenoma-related Cushing’s disease and renal tumors.
Lymphosarcoma
Multicentric (generalized) and cutaneous histiocytic lymphosarcoma may have oral manifestations (Fig. 11.25). Usually the clinical appearance is of ill-defined nodular lesions of variable size embedded within and below the mucosal surface, which probably reflects the involvement of the normally-diffuse lymphoid tissue of the nasopharynx. The gingival mucosa is often affected. A similar nodular appearance is often present in the pharynx and in the sublingual tonsillar tissue where larger swellings may cause difficulty on swallowing. The lesions may ulcerate or become infected. Simultaneous submandibular lymphadenopathy, and other secondary effects, such as anemia, lethargy, and weight loss may signify serious systemic involvement.
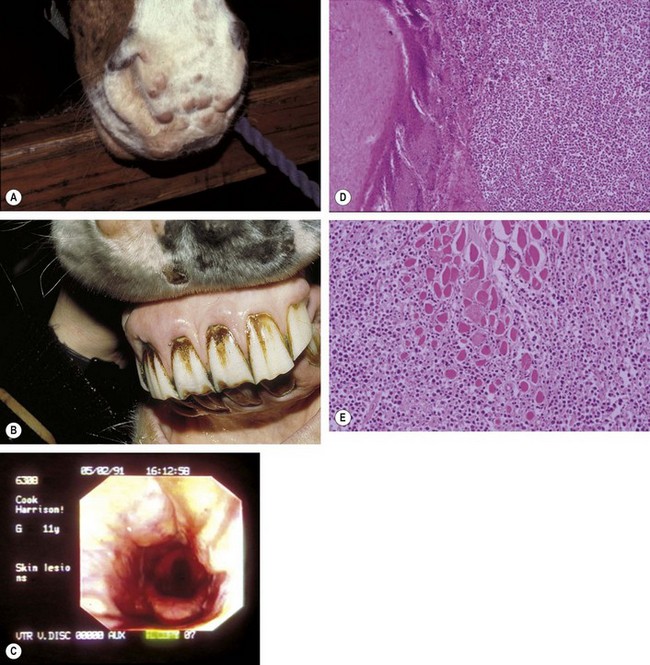
Fig. 11.25 (A) Multiple discreet dermal nodules in a pony with histiocytic lymphosarcoma. (B) Cutaneous histiocytic lymphosarcoma with involvement of the gingival tissues. The ill-defined swellings of the gingiva were due to aggregations of abnormal lymphocytes. These lesions might easily be overlooked but are a regular finding in these cases. (C) Pharyngeal lymphosarcoma lesions that can sometimes be identified endoscopically. These lesions were symptom-free but larger ones can cause dysphagia. (D) Photomicrograph of the tongue from an aged horse with oral lymphosarcoma showing epithelium and diffuse subepithelial accumulation of neoplastic lymphoid cells. (E) Lymphosarcoma of the tongue showing neoplastic cells around and between muscle fibers.
Pituitary pars intermedia dysfunction (PPIDD / equine Cushing’s disease)
Hyperplasia of the pituitary pars intermedia is responsible for secondary oral disease, such as extensive non-healing oral ulcers (Fig. 11.26), and dental and periodontal infections. The condition is regarded by some as a neoplasm of the pars intermedia, and is very common in older horses (usually > 14–17 years). Extensive dental and alveolar disease (including periodontal disease and periapical sepsis) commonly develops. This, with advancing, age-related shortening of the residual crown of older horses, predisposes the affected animals to secondary sinusitis, and there may be tertiary consequences from this, including early shedding of teeth, oronasal and oromaxillary fistulae, oral ulceration and anemia.
Other distant tumors
Paraneoplastic changes, such as hypercalcemia and oral ulceration, can develop as a result of neoplastic disease in other tissues (usually myeloproliferative neoplasia).77 It is important to remember that the mouth may be one of the early sites where clinical evidence of neoplastic disease, such as anemia, icterus, and azotemia, may be manifested. The lesions seen in the mouth may not be readily attributable to neoplastic disease either at a local or remote site.
1 Jackson C. The incidence and pathology of tumors of domestic animals in South Africa: a study of the Onderstepoort collection of neoplasms with special reference to their histopathology. Onderstepoort Journal of Veterinary Science and Animal Industries. 1936;6:1–460.
2 Hagdoost IS, Zakarian B. Neoplasms of Equidae in Iran. Equine Vet J. 1985;17:237–239.
3 Pirie RS, Tremaine WH. Neoplasia of the mouth and surrounding structures. In: Robinson NE, ed. Current therapy in equine medicine. Philadelphia: WB Saunders; 1997:153–155.
4 Sundberg JP, Burnstein T, Page EH, et al. Neoplasms of equidae. J Am Vet Med Assoc. 1997;170:150–152.
5 Smith P. Large animal internal medicine. Philadelphia: Mosby; 1996. 696
6 Baker GJ. Oral examination and diagnosis: Management of Oral Diseases. In: Harvey CE, ed. Veterinary dentistry. Philadelphia: WB Saunders; 1999:217–220.
7 Richardson DW, Evans LH, Tulleners EP. Rostral mandibulectomy in five horses. J Am Vet MedAssoc. 1991;199:1179–1182.
8 Purdy CM. Mandibular aneurysmal bone cyst in a horse. Equine Practice. 1985;7:22–24.
9 Verstraete FJM, Ligthelm AJ. Excessive granulation tissue of periodontal origin in a horse. Equine Vet J. 1988;20:380–382.
10 Lamb CR, Schelling SH. Congenital aneurysmal bone cyst in the mandible of a foal. Equine Vet J. 1989;21:130–132.
11 Gibbs C. The equine skull; its radiographic investigation. Journal of the American Veterinary Radiology Society. 1974;15:70–78.
12 Wyn-Jones G. Interpreting radiographs 6: Radiology of the equine head part 2. Equine Vet J. 1985;17:417–425.
13 McIlwraith CW. Equine digestive system. In: Jennings PB, ed. The practice of large animal surgery. Philadelphia: WB Saunders; 1984:579.
14 Moulton JE. Tumors in domestic animals, 3rd edn. Berkeley: University of California Press; 1990. 167–168
15 Head KW. Tumors of the alimentary tract. In: Moulton JE, ed. Tumors in domestic animals. 3rd edn. Berkeley: University of California Press; 1990:347–374.
16 Baker JR, Leyland A. Histological survey of tumors of the horse, with particular reference to those of the skin. Vet Rec. 1975;96:419–422.
17 Cotchin E. A general survey of tumors of the horse. Equine Vet J. 1977;9:16–21.
18 Gorlin RJ, Meskin LH, Brodey R. Odontogenic tumors in man and animals; Pathological classification and clinical behavior – a review. Annals of the New York Academy of Science. 1963;108:722–771.
19 Turrel JM. Oncology. In: Kobluk CN, Ames TR, Geor RJ. The horse: diseases and clinical management. Philadelphia: WB Saunders; 1995:1128–1130.
20 Theon AP. Intralesional and topical chemotherapy and immunotherapy. Veterinary Clinics of North America Equine Practice. 1998;14:659–671.
21 Rols MP, Tamzali Y, Teissie J. Electrochemotherapy of horses: a preliminary report. Bioelectrochemistry. 2002;55:101–105.
22 Theon AP, Radiation therapy in the horse. Veterinary clinics of North America equine practice, 1998:673–688.
23 Dubielzig RR. Tumors of the alimentary tract. In: Meuten DJ, ed. Tumors in domestic animals. 4th edn. Ames, Iowa: Iowa State Press; 2002:402–410.
24 Wahl P. Adamantinoma polycysticum ossificans am Unterkiefer eines Pferdes. Deutsche Tierartztliche Wochenschrift. 1936;46:113–117.
25 Peter CP, Myers VS, Ramsey FK. Ameloblastomic odontoma in a pony. American Journal of Veterinary Research. 1968;29:1495.
26 Vaughan JT, Bartels JE. Equine mandibular adamantinoma. J Am Vet Med Assoc. 1968;153:454–457.
27 Hanselka DW, Roberts RE, Thompson RB. Adamantinoma of the equine mandible. Veterinary Medicine for the Small Animal Clinician. 1974;69:157–160.
28 Jones SL, Brumbaugh GW. Ameloblastoma in mandible of horse. What is your diagnosis? J Am Vet Med Assoc. 1991;199:630–631.
29 Summers PM, Wells KE, Adkins KF. Ossifying ameloblastoma in a horse. Australian Veterinary Journal. 1979;55:498–500.
30 Lingard DR, Crawford TB. Congenital ameloblastoma in a foal. American Journal of Veterinary Research. 1970;31:801.
31 Roberts MC, Groenendyk S, Kelly WR. Ameloblastomic odontoma in a foal. Equine Vet J. 1978;10:91–93.
32 French DA, Fretz PB, Davis GD. Mandibular adamantinoma in a horse; radical surgical treatment. Veterinary Surgery. 1984;13:165–171.
33 Dillehay DL, Schoeb TR. Complex odontoma in a horse. Vet Pathol. 1986;23:341–342.
34 Dubielzig RR, Beck KA, Levine S, Wilson JW. Complex odontoma in a stallion. Vet Pathol. 1986;23:633–635.
35 Mason BJE. Temporal teratomata in the horse. Vet Rec. 1974;95:226–228.
36 Fessler JF. Heterotopic polyodontia in horses: nine cases 1969–1986. J Am Vet Med Assoc. 1988;192:535–538.
37 Lindshaw WA, Bech KA. Temporal teratoma in a horse. Compendium of Continuing Education for Practicing Veterinarians. 1986;8:168–171.
38 Kramer IRH, Pindborg JJ, Shear M. Histologic typing of odontogenic tumors, 2nd edn. Berlin: Springer-Verlag; 1993. 151–156
39 Pindborg JJ, Kramer IRH, Torlini H. Histologic typing of odontogenic tumors, jaw cysts and allied lesions. Geneva, Switzerland: World Health Organisation; 1971. 145–154
40 Livesey MA, Wilkie IW. Focal and multifocal osteosarcoma in two foals. Equine Vet J. 1986;18:407–410.
41 Slayter MV, Boosinger TR, Pool RR, et al, Histological classification of bone and joint tumours of domestic animals. 2nd series, Volume 1, Armed Forces Institute of Pathology, Washington DC, 1994. 9–45
42 Thorp F, Graham R. A large osteosarcoma of the mandible. J Am Vet Med Assoc. 1934;84:118–119.
43 Jacobson SA. Parosteal osteoma juxtacortical osteogenic sarcoma in animals. American Journal of Pathology. 1969;58:85a.
44 Livesey MA. Wilkie IW. Focal and multifocal osteosarcoma in two foals. Equine Vet J. 1986;18:410–412.
45 Gorlin RJ, Meskin LH, Brodey RS. Odontogenic tumors in man and animals; pathological classification and clinical behavior – a review. Annals of the New York Academy of Science. 1963;108:722–771.
46 Strafuss AC. Squamous cell carcinoma in horses. J Am Vet Med Assoc. 1976;168:61–62.
47 Howie F, Munroe G, Thompson H, Murphy D. Palatine squamous cell carcinoma involving the maxillary sinus in two horses. Equine Veterinary Education. 1992;4:3–7.
48 Schuh JCL. Squamous cell carcinoma of the oral, pharyngeal and nasal mucosa in the horse. Vet Pathol. 1986;23:205–210.
49 Johnson PJ. Dermatologic tumors excluding sarcoids. Veterinary Clinics of North America Equine Practice. 1998;14:625–658.
50 Barker IK, van Dreumel AA, Palmer N, Jubb KVF, Kennedy PC, Palmer N, eds. Pathology of domestic animals, 4th edn, Volume 2. New York: Academic Press, 1993;27.
51 Knottenbelt DC, Pascoe RR, Diseases and disorders of the horse. Wolfe, London, 1994, 303
52 Henson WR. Carcinoma of the tongue in a horse. J Am Vet Med Assoc. 1936;94:124.
53 Leyland A, Baker JR. Lesions of the nasal cavity and paranasal sinuses of the horse causing dyspnoea. British Veterinary Journal. 1975;131:339–346.
54 Orsini JA, Nunamaker DM, Jones CJ, Acland HM. Excision of oral squamous cell carcinoma in a horse. Veterinary Surgery. 1991;20:264–266.
55 Theon AP. Cisplatin treatment for cutaneous tumors. In: Robinson NE, ed. Current therapy in equine medicine. 4th edn. Philadelphia: WB Saunders; 1997:372–377.
56 Paterson S. Treatment of superficial ulcerative squamous cell carcinoma in three horses with topical 5-fluorouracil. Vet Rec. 1997;141:626–628.
57 Pulley LT, Stannard AA. Tumors of the skin and soft tissues. In: Moulton JE, ed. Tumors in domestic animals. 3rd edn. Berkeley: University of California Press; 1990:23–87.
58 Knottenbelt DC, Edwards SER, Daniel EA. The diagnosis and treatment of the equine sarcoid. Practice. 1995;17:123–129.
59 Knottenbelt DC. A suggested clinical classification for the equine sarcoid. Diagnostic Techniques in Equine Medicine. 2005;3:278–295.
60 Pascoe RR, Knottenbelt DC. Neoplastic conditions. In: Manual of equine dermatology. Philadelphia: WB Saunders; 1999:244–252.
61 Brostrom H, Equine sarcoids: a clinical, epidemiological and immunological study. PhD Thesis, University of Uppsala, Sweden, 1995. 9–70
62 Theon AP. Cisplatin treatment for cutaneous tumors. In: Robinson NE, ed. Current therapy in equine medicine. 4th edn. Philadelphia: WB Saunders; 1997:372–377.
63 Knottenbelt DC, Kelly DF. The diagnosis and treatment of periorbital sarcoid in the horse: 445 cases from 1974 to 1999. Veterinary Ophthalmology. 2000;32:169–191.
64 Kelly DF. Diagnostic problems in nasopharyngeal amelanotic melanomas. Equine Veterinary Education. 2003;15:25–26.
65 Goetz TE, Ogilvie GK, Keegan KG, Johnson PJ. Cimetidine for the treatment of melanoma in 3 horses. J Am Vet Med Assoc. 1990;196:449–452.
66 Goetz TE, Long MT. Treatment of melanoma in horses. Compendium of Continuing Education for the Practicing Veterinarian. 1993;15:608–610.
67 Morse CC, Saik JE, Richardson DW. Equine juvenile mandibular ossifying fibroma. Vet Pathol. 1988;25:415–421.
68 Robbins SC, Arighi M, Ottewell G. The use of megavoltage radiation to treat juvenile mandibular ossifying fibroma in a horse. Canadian Veterinary Journal. 1996;37:683–684.
69 Kawcak CE, Stashak TS, Norrdin RW. Treatment of ossifying fibroma in a horse by hemimaxillectomy. Equine Practice. 1996;18:22–25.
70 Fry FL, Knight HD, Brown S. Hemangiosarcoma in a horse. J Am Vet Med Assoc. 1983;182:287–289.
71 Sweigard KD, Hattell AL. Oral hemangiosarcoma in a horse. Equine Practice. 1993;15:10–13.
72 Stackhouse LL, Moore JJ, Hylton WE. Salivary gland adenocarcinoma in a mare. J Am Vet Med Assoc. 1978;172:271–273.
73 Head KW. Salivary gland disease in domestic animals. In: de Burgh P, Norman JE, McGurk M. Salivary glands. Color atlas and text. Philadelphia: Mosby-Wolfe; 1995:367–389.
74 Biesecker JL, Marcove RC, Huvos AG, Mike V. Aneurysmal bone cysts; a clinicopathological study of 66 cases. Cancer. 1970;26:616–625.
75 Dabezies EJ, D’Ambrosia RD, Chuinard RG, Ferguson AB. Aneurysmal bone cyst after fracture. Journal of Bone and Joint Surgery. 1982;64:617–621.
76 Jacobson SA. The comparative pathology of the tumors of bone. Springfield, Illinois: Charles C Thomas; 1971.
77 Williams MA, Dowling PM, Angarano DW, et al. Paraneoplastic bullous stomatitis in a horse. J Am Vet Med Assoc. 1995;207:331–334.
* cis-diamminedichloroplatinum, or cis-DDP, cis-platinum.