CHAPTER 14 Ancillary diagnostic techniques
Cytological examination and culture of exudate
Empyema of the paranasal sinuses is a common complication of periapical infection of those cheek teeth whose apices reside within a maxillary sinus and of primary infection of the upper portion of the respiratory tract. Determining the underlying cause of paranasal sinus empyema is important because the cause of empyema influences treatment of the affected horse. Although periapical infection of teeth rostral to the maxillary sinuses is readily identified during radiographic examination of the skull, periapical infection of teeth within the sinuses can be confidently recognized in only about half of affected horses.1
To help determine whether empyema is caused by dental disease or by primary infection, exudate obtained by centesis of the paranasal sinuses can be examined cytologically and cultured for bacteria. Exudate from within the paranasal sinuses can be obtained by introducing a dog urinary catheter through a small portal created in the frontal bone at a site one-third of the distance from the medial canthus of the eye to the dorsal midline, and 0.5 cm caudal to a line connecting the medial canthi.2 The portal can be created by making a stab incision in the skin and either drilling through the frontal bone with a small, trocar-tipped, Steinmann pin using a Jacob’s chuck or by driving a steel, 14- or 16-gauge, hypodermic needle through the frontal bone with a mallet. The needle should be removed before introducing a catheter into the sinuses. The catheter is passed through the portal and advanced through the underlying frontomaxillary aperture until it contacts the floor of the caudal maxillary sinus where exudate, if present, can usually be found, regardless of the site of disease causing empyema. If no exudate can be obtained, 10–20 ml of sterile, isotonic saline solution should be instilled into the sinus and aspirated (Fig. 14.1).
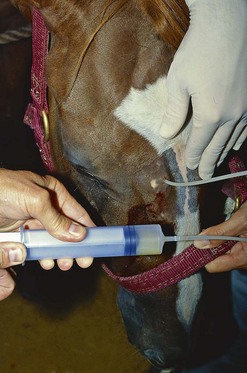
Fig. 14.1 A sample of exudate is obtained from the sinuses for bacteriological examination using a dog urinary catheter.
Identifying a single bacterial species during cytological examination of the exudate or culturing a single bacterial species from the exudate indicates that the empyema is probably caused by primary bacterial infection.3 If empyema is caused by periapical dental infection or an orosinus fistula, multiple types of bacterial colonies are identified, and plant material can sometimes be seen in the exudate.
A wide variety of bacteria, including aerobic and anaerobic, Gram-positive and Gram-negative bacteria, have been isolated from exudate obtained from the sinuses of horses affected with primary bacterial sinusitis. Hemolytic Streptococcus spp. are the most commonly isolated bacterial species.4,5 Streptococcus equi var. equi, the cause of strangles, is rarely isolated,3,6,7 but the presence of this organism may be masked by the overgrowth of other bacterial species. If a horse suffering from sinusitis has had signs typical of strangles, PCR examination of exudate obtained from the paranasal sinus should be considered to rule out infection caused by S. equi var. equi.8 The clinical significance of isolating bacterial species other than hemolytic Streptococcus spp. is largely unknown. Culture of exudate from the paranasal sinuses of horses affected by primary bacterial sinusitis can yield multiple bacterial isolates, especially if the infection is long-standing or if the horse has received prolonged administration of one or more antimicrobial drugs,4 and therefore, definitively determining the organism responsible for primary infection of the paranasal sinuses on the basis of microbial culture of the exudate is difficult. Waiting for results of bacteriological examination before proceeding with other diagnostic tests, such as sinoscopic or radiographic examination of the sinuses, may not be warranted.
Odor is seldom helpful in determining the cause of paranasal sinus empyema. Although nasal discharge produced because of primary sinusitis is usually not characterized by necrotic odor,9 primary bacterial sinus empyema can result in a necrotic odor to the horse’s breath7 if the exudate becomes inspissated, because expansion of the inspissated exudate causes necrosis of surrounding tissue. Relying on odor to distinguish primary sinusitis from sinusitis occurring secondary to other disease, such as dental infection, may result in inaccurate diagnosis.
The ventral conchal sinus is frequently the compartment of the paranasal sinuses in which exudate becomes inspissated,4 and a large volume of inspissated exudate within this compartment usually signifies that the empyema is caused by primary infection (authors’ observation).
Empyema of the paranasal sinuses caused by periapical dental disease (i.e., dental sinusitis) is frequently associated with a necrotic odor because periapical dental infection is usually accompanied by anaerobic infection of alveolar bone.10 Mycotic infection of the paranasal sinuses should also be considered if paranasal sinusitis is accompanied by a malodorous nasal discharge.11 A space-occupying lesion, such as a neoplasm or a progressive ethmoidal hematoma, may also produce a malodorous nasal charge from destruction of surrounding tissue or stagnation of mucosal secretions.
Percussion
Percussion of the paranasal sinuses is performed by rapping one’s knuckles against the facial bones overlying the sinuses and listening for a difference in resonance between right and left sinuses. Percussion may identify loss of resonance within the sinuses, especially if sinuses are completely filled with fluid or tissue, but percussion, in general, is unreliable for detecting disease within the sinuses.12 Resentment of the horse to percussion, however, may signify the presence of inflammation within the sinuses. Percussion is best performed with the horse’s mouth held open with a speculum so that sound produced by percussion is not dampened by being transferred through the mandible.
Rhinoscopy
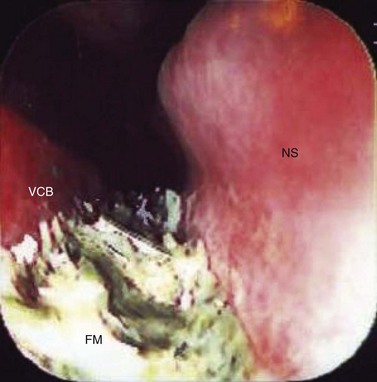
Rhinoscopy is performed with a flexible, video- or fiberoptic endoscope, with the horse standing. Rhinoscopy is indicated to determine the source of purulent exudate discharging from an external naris. The source of purulent exudate at an external naris could be the ipsilateral nasal cavity or paranasal sinuses, a guttural pouch, the nasopharynx, or the lungs. Finding exudate consistently at only one naris implicates the ipsilateral paranasal sinuses or nasal cavity as the source of the exudate. Exudate that originates within the paranasal sinuses can usually be observed, during rhinoscopy, discharging into the middle nasal meatus at the drainage angle located at the caudal aspect of the nasal cavity (Fig. 14.2).
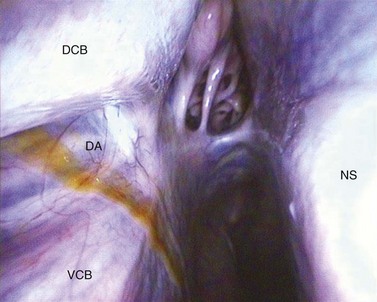
Fig. 14.2 Rhinoscopic image of the right caudal middle meatus showing exudate exiting the ‘drainage angle’ of the right paranasal sinuses. DCB, dorsal conchal bone; VCB, ventral conchal bone; DA, drainage angle; NS, nasal septum.
Rhinoscopy is also indicated to determine the cause of obstruction of the nasal cavity. Obstruction, even mild obstruction, can often be detected by placing the palms of the hands in front of the nares and comparing the volume of air exhaled from each nasal cavity.12 A nasal cavity can become obstructed from a mass originating within the nasal cavity, such as a progressive ethmoid hematoma expanding from the nasal portion of the ethmoid labyrinth, or from axial deviation of the conchae caused by an expanding mass within the paranasal sinuses, such as a neoplasm, an osteoma, a cyst, inspissated exudate, or a progressive ethmoidal hematoma whose origin is the sinusal portion of the ethmoidal labyrinth (Fig. 14.3). Partial or complete obstruction of the nasomaxillary aperture may also result in axial deviation of the conchae from accumulation of exudate. A horse that has developed distortion of the facial bones rostral to the eye should be examined endoscopically to determine if the nasal conchae have also been distorted. Axial distortion of the nasal conchae indicates that the facial distortion is caused by an expanding mass or large volume of exudate within the paranasal sinuses. Sometimes, subtle deviation of the conchae can be appreciated only by endoscopically comparing the circumference and morphology of the two nasal cavities. Distortion of the conchae is not commonly caused by dental disease.
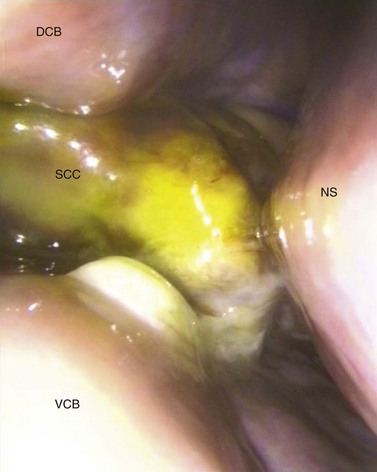
Fig. 14.3 Rhinoscopic image of the left middle meatus of a horse with a squamous cell carcinoma growing through the nasomaxillary aperture obscuring the drainage angle and middle meatus. DCB, dorsal conchal bone; VCB, ventral conchal bone; NS, nasal septum; SCC, squamous cell carcinoma.
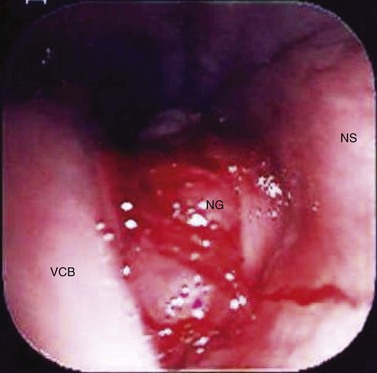
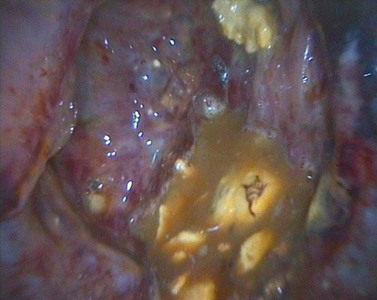
Lesions encountered during rhinoscopy associated with dental disease include oronasal or oromaxillary fistulas (Fig. 14.4), apical granulomas, and displaced teeth. An apical granuloma is visible during rhinoscopy within the rostrolateral aspect of the nasal cavity and is caused by periapical infection of the 2nd, 3rd, or 4th premolars (Triadan 06–08). If accompanied by an oro-nasal fistula, the granuloma is sometimes covered with exudate and feed. Removal of the feed reveals the granuloma (Fig. 14.5).
Sinoscopy
The primary role of sinoscopy in the investigation of dental disease is to rule out other causes of paranasal sinusitis. Sinoscopy enables the interior of the paranasal sinuses to be visualized, aiding in the diagnosis of many conditions of the paranasal sinuses, such as primary sinusitis and the presence of a mass.
Most of the structures within the paranasal sinuses can be examined endoscopically, using a flexible, video- or fiberoptic endoscope with the horse standing. Although a rigid arthroscope can also be used, it provides an inferior field of view because it is unable to navigate around structures such as the infra-orbital canal, making good visualization of some structures within the sinuses difficult.
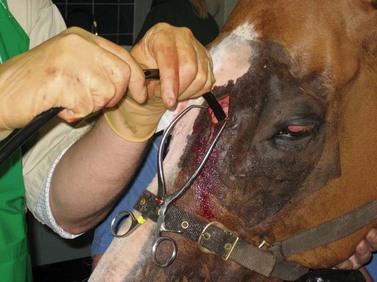
In preparation for sinoscopy, the horse is restrained in a stock and sedated, usually with detomidine (0.01–0.02 mg/kg, IV or 0.03–0.04 mg/kg, IM) or xylazine (0.5–1.0 mg/kg, IV or IM) and butorphanol tartrate (0.02–0.05 mg/kg, IV) or morphine (0.15 mg/kg, IV; see Ch. 15, Restraint and anesthesia). After the horse is sedated, its head can be supported on a stand or small table so that the site for trephination is at a comfortable level for the surgeon. The sites at which the endoscopic portals are to be created are prepared for surgery and desensitized by subcutaneous instillation of 2–3 ml of local anesthetic solution. A portal for insertion of the endoscope is created through a 2- to 3-cm, longitudinal, skin and periosteal incision. A portal through the frontal or maxillary bone created with a 3/8-inch (9.5-mm) to 5/8-inch (1.6-cm) Galt trephine or drill bit accommodates the insertion tube of most endoscopes and allows for quick retrieval of the endoscope, if necessary (Fig. 14.6). The cutaneous incision is sutured or stapled after sinoscopy has been completed. The periosteum is left unsutured.
Most of the paranasal sinuses can be examined through a portal through the frontal bone into the conchofrontal sinus (i.e., the frontal and dorsal conchal sinuses) or through a portal through the maxillary bone into the caudal maxillary sinus. A portal into the conchofrontal sinus generally provides the best visualization of the conchofrontal and caudal maxillary sinuses. The incision for a portal into the conchofrontal sinus is centered over the frontomaxillary aperture at a site one-third of the distance from the medial canthus of the eye to the dorsal midline, and 0.5 cm caudal to a plane connecting the medial canthi (Fig. 14.6).2 The incision for a portal into the caudal maxillary sinus is centered 2 cm ventral and 2 cm rostral to the medial canthus of the eye.2
To evaluate the rostral maxillary and ventral conchal sinuses from either the conchofrontal or the caudal maxillary portal, the bulla of the ventral conchal sinus must be fenestrated under endoscopic guidance, using an arthroscopic rongeur or crocodile forceps passed through the same portal as the endoscope (Figs 14.7 & 14.8).13 Hemorrhage is a minor, but not an infrequent complication of fenestrating the bulla. If hemorrhage obscures visualization, sinoscopy should be repeated when hemorrhage has ceased or been evacuated using suction.
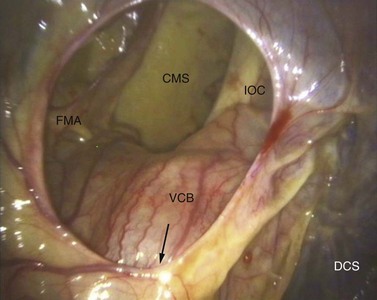
Fig. 14.7 Sinoscopic view of the right conchofrontal sinus. CMS, caudal maxillary sinus; FMA, frontomaxillary aperture; IOC, infraorbital canal; VCB, ventral conchal bulla; DCS, dorsal conchal sinus. The arrow points to the entrance of the intra-sinus, nasomaxillary aperture of the caudal maxillary sinus.
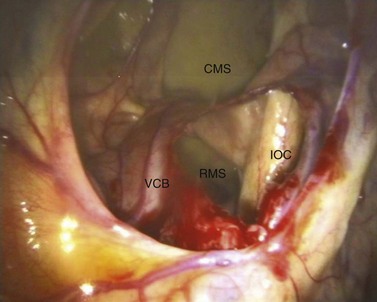
Fig. 14.8 Sinoscopic view of the right conchofrontal sinus (the same as in Fig. 14.7) after endoscopic-guided fenestration of the ventral conchal bulla. CMS, caudal maxillary sinus; IOC, infraorbital canal; VCB, ventral conchal bulla (fenestrated); RMS, rostral maxillary sinus.
The rostral maxillary sinus can also be examined through a portal created in the maxillary bone directly over the rostral or the caudal aspect of the sinus,2,5 although this procedure is not without risk of damaging a normal tooth in young horses (i.e., <5 years old), due to the small size of this sinus and its anatomic variability among horses. The skin incision for a rostral portal into the rostral maxillary sinus is oriented longitudinal to the long axis of the head and is created 3 cm caudal to the infraorbital foramen and 1 cm ventral to an imaginary line joining the infraorbital foramen and the medial canthus of the eye.2 The skin incision for a caudal portal into the rostral maxillary sinus is centered at a point midway between the rostral end of the facial crest and a point on the facial crest at the level of the medial canthus of the eye, 1 cm ventral to an imaginary line drawn between the infraorbital foramen and the medial canthus of the eye.2 Access to the ventral conchal sinus through either portal is poor, and visualization of the rostral maxillary sinus is reduced in comparison to that obtained through a portal into the conchofrontal sinus combined with fenestration of the bulla of the ventral conchal sinus.
Care should be taken when creating a portal into the rostral maxillary sinus to avoid damaging the reserve crowns of the teeth contained within. Damage to the underlying apices is more likely when creating the rostrally positioned portal into the rostral maxillary sinus than when creating a caudally positioned portal,2,14 and therefore, the rostrally positioned portal should be used only on horses aged 6 years or older.14 Damage to an apex of a cheek tooth is unlikely when using a conchofrontal or caudal maxillary approach.
The respiratory mucosa of the paranasal sinuses should be pink, and its vasculature should be visible. The sinuses should contain little or no fluid. The presence of purulent exudate, even a small amount, is abnormal. If visualizing structures within the paranasal sinuses is difficult because a large volume of exudate is present within the sinuses, the sinuses should be lavaged with normal saline solution once or twice daily for one or two days before sinoscopy is performed.
The interior of the conchofrontal and caudal maxillary sinuses can be visualized directly through the portal in the frontal bone. Structures that can be identified within the conchofrontal sinus include the scroll-like surface of the ethmoturbinates, located at the caudomedial aspect of the frontal sinus, and the frontomaxillary aperture located directly below the endoscopic portal (Fig. 14.7). Structures observed within the caudal maxillary sinus include the maxillary septum, which marks the rostral boundary of the caudal maxillary sinus; the opening of the nasomaxillary aperture, formed by floor of the dorsal conchal sinus and maxillary septum, at the rostral aspect of the frontomaxillary aperture; the apices of the 5th and 6th cheek teeth (Triadan 10 and 11); the infraorbital canal coursing caudally from the caudal aspect of the caudal maxillary sinus to the maxillary septum; and the entrance of the sphenopalatine sinus medial to the caudal aspect of the infraorbital canal (Fig. 14.9).
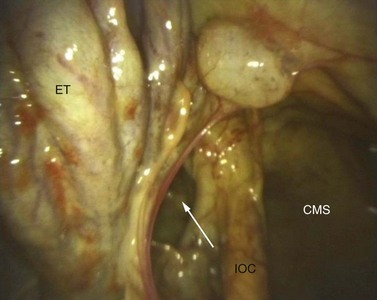
Fig. 14.9 Sinoscopic view in the left caudal maxillary sinus showing the entrance to the sphenopalatine sinus (arrow). ET, intra-sinus portion of the ethmoidal turbinates; CMS, caudal maxillary sinus; IOC, infraorbital canal.
These same structures can also be observed through the portal into the caudal maxillary sinus created in the maxillary bone ventral to the eye, but visualization of some structures in the ventral conchal sinus and the rostral maxillary sinus, such as the apices of the 3rd and 4th cheek teeth (Triadan 08 and 09) and the entrance of the sphenopalatine sinus, is more difficult.
Structures that can be observed through the portals into the rostral maxillary sinus created in the maxillary bone include the apex of the 4th cheek tooth (Triadan 09) and a portion of the 3rd and 5th cheek teeth (Triadan 08 and 10), the long, slit-like conchomaxillary aperture (i.e., the entrance to the ventral conchal sinus), and occasionally, the rostromaxillary portion of the nasomaxillary aperture. Visualization of the apices of the 3rd and 4th cheek teeth (Triadan 08 and 09) through either of the portals in the maxillary bone into the rostral maxillary sinus is comparable to that achieved through the conchofrontal portal. Because of its small size, the rostral maxillary sinus is more difficult to evaluate endoscopically than are the caudal maxillary and conchofrontal sinuses. The rostral maxillary sinus of young horses is especially difficult to evaluate because the reserve crowns of the 3rd, 4th, and 5th cheek teeth (Triadan 08–10) occupy most of the sinus. The size of the maxillary sinuses increases, and the apices of the cheek teeth become less prominent as horses age because of the perpetual extrusion and rostral migration of the reserve crowns of the cheek teeth.
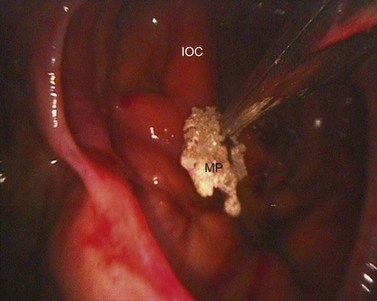
Sinoscopic findings specific to dental sinusitis include a swollen and hyperemic apex of an alveolus (Figs 14.10 & 14.11), an orosinus fistula (Fig. 14.12), food material within the sinuses (Fig. 14.13), and an apical granuloma (Fig. 14.14). Mycotic sinusitis can be primary, or it can occur secondary to chronic bacterial sinusitis, such as that caused by apical infection of a cheek tooth. It can also occur after sinonasal fenestration (Fig. 14.15).7,11 Findings during sinoscopy, such as generalized mucosal thickening and edema, petechial hemorrhages, and inspissated exudate, are frequently non-specific, and although confirming the presence of sinusitis, do not allow determination of its cause (Fig. 14.16). Other diagnostic modalities, such as radiography,1,15 scintigraphy,16 and computed tomography,17 as discussed in detail in Chapter 13, are often required to confirm a diagnosis of dental sinusitis. Because dental sinusitis is frequently chronic, the mucosa is often grossly thickened,18 preventing accurate identification of the affected tooth by sinoscopy.
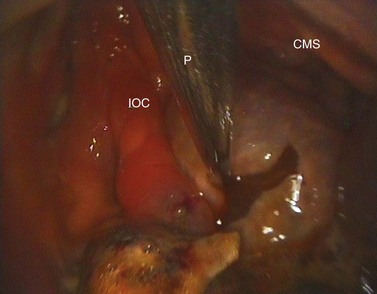
Fig. 14.10 Sinoscopic view of the left caudal maxillary sinus showing edema of the mucosa overlying an apical infection of the 5th maxillary cheek tooth (probe). CMS, caudal maxillary sinus; IOC, infraorbital canal.
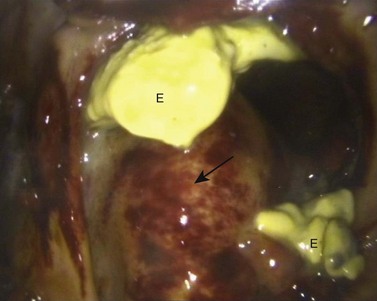
Fig. 14.11 Sinoscopic view of the rostral maxillary sinus, after fenestration of the ventral conchal bulla, showing edema of the mucosa overlying an apically infected 4th maxillary cheek tooth (arrow) and exudate (E) associated with the infection.
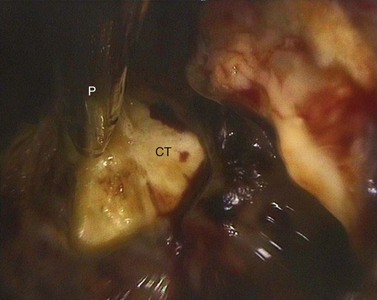
Fig. 14.12 Sinoscopic view into the rostral maxillary sinus after fenestration of the ventral conchal bulla. The probe (P) is placed on the apex of the 4th maxillary cheek tooth (CT). The overlying alveolar bone has been eroded. A chronic oro-sinus fistula (not seen in this view) provided a direct channel for food to enter the sinuses.
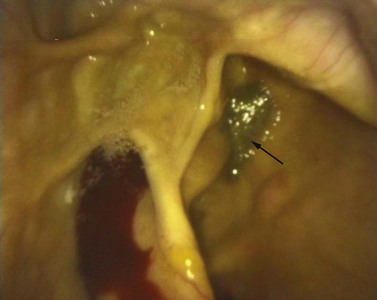
Fig. 14.13 Sinoscopic view of the rostral maxillary sinus after fenestration of the ventral conchal bulla of a horse with an oro-sinus fistula. The rostral maxillary sinus contains small amounts of food (arrow).
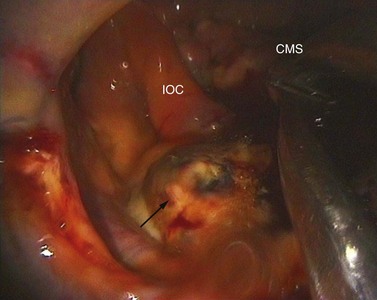
Fig. 14.14 Sinoscopic view of the left caudal maxillary sinus showing an apical granuloma overlying an apical infection of the 5th maxillary cheek tooth (arrow). CMS, caudal maxillary sinus; IOC, infraorbital canal.
1 Gibbs C, Lane JG. Radiographic investigation of the facial, nasal and paranasal sinus regions of the horse: II. Radiological findings. Equine Vet J. 1987;19:474–492.
2 Ruggles AJ, Ross MW, Freeman DE. Endoscopic examination of normal paranasal sinuses in horses. Veterinary Surgery. 1991;20:418–423.
3 Laverty S, Pascoe JR. Sinusitis. In: Robinson NE, ed. Current therapy in equine medicine. 4th edn. Philadelphia: WB Saunders; 1997:419–422.
4 Schumacher J, Honnas C, Smith B. Paranasal sinusitis complicated by inspissated exudate in the ventral conchal sinus. Veterinary Surgery. 1987;16:373–377.
5 Ruggles AJ, Ross MW, Freeman DE. Endoscopic examination and treatment of paranasal sinus disease in 16 Horses. Veterinary Surgery. 1993;22:508–514.
6 Mansmann RA, Wheat JD. The diagnosis and treatment of equine upper respiratory diseases. In: Proceedings of the 18th Annual Convention of the American Association of Equine Practitioners. Lexington, KY, 1973, pp 388–487
7 Tremaine WH, Dixon PM. A long-term study of 277 cases of equine sinonasal disease. Part 1: Details of horses, historical, clinical and ancillary diagnostic findings. Equine Vet J. 2001;33:274–282.
8 Newton JR, Verheyen K, Talbot NC, et al. Control of strangles outbreaks by isolation of guttural pouch carriers identified using PCR and culture of Streptococcus equi. Equine Vet J. 2000;32:515–526.
9 Mason BJE. Empyema of the equine paranasal sinuses. Journal of American Veterinary Medical Association. 1975;167:727–731.
10 Mackintosh ME, Colles CM. Anaerobic bacteria associated with abscesses in the horse and donkey. Equine Vet J. 1987;19:360–362.
11 McGorum BC, Dixon PM, Lawson GHK. A review of ten cases of mycotic rhinitis. Equine Veterinary Education. 1992;4:8–12.
12 Lane JG. The management of sinus disorders of horses – Part 1. Equine Veterinary Education. 1993;5:5–9.
13 Freeman DE. Sinus Disease. Veterinary clinics of North America: equine practice. 2003;19:209–243.
14 Barakzai SZ, Kane-Smyth J, Lowles J, Townsend N. Trephination of the equine rostral maxillary sinus: efficacy and safety of two trephine sites. Veterinary Surgery. 2008;37:278–282.
15 Wyn-Jones G. Interpreting radiographs 6: Radiology of the equine head (Part 2). Equine veterinary Journal. 1985;17:417–425.
16 Weller R, Livesey L, Maierl J, et al. Comparison of radiography and scintigraphy in the diagnosis of dental disorders in the horse. Equine Vet J. 2001;33:49–58.
17 Henninger W, Frame EM, Willmann M, et al. CT features of alveolitis and sinusitis in horses. Veterinary Radiology and Ultrasound. 2003;44:269–276.
18 Tremaine WH, Clarke CJ, Dixon PM. Histopathological findings in equine sinonasal disorders. Equine Vet J. 1999;31:296–303.