12 Immunity in bacterial infections
Key points
• Phagocytosis and complement activation are key innate defences against bacterial infections associated with acute inflammation.
• Antibodies are notably effective against bacterial cell surface and extracellular virulence factors such as toxins. Cell-mediated immunity is essential for defence against intracellular bacteria.
• Intracellular survival and growth provides one means of immune evasion for bacteria.
• Bacterial lipoplysaccharide and a number of other cell wall components can elicit uncontrolled innate immune responses that are potentially fatal to the host in the form of septic shock.
Modern medical science has managed to subdue many of the classical infectious diseases, but has helped to create new ones that result from interference with normal host defence mechanisms, consequent upon medical and surgical procedures such as chemotherapy, catheterization, immunosuppression and irradiation. Infections that develop in this way are known as iatrogenic (physician-induced) diseases.
It is important to differentiate infection from disease. A host may be infected with a particular micro-organism and be unaware of its presence. If the microbe reproduces itself to such an extent that toxic products or sheer numbers of organisms begin to harm the host, a disease process has developed. Potentially pathogenic bacteria such as pneumococci, streptococci and salmonellae are found in the nose, throat or bowel; this is known as the carrier state and is a source of infection to other individuals. These bacteria may also cause disease in the carrier if they enter a vulnerable tissue.
Host defences
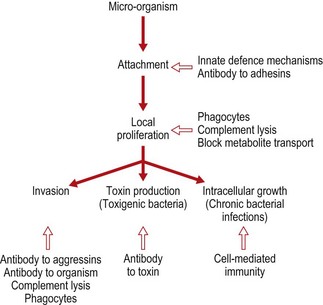
Few organisms can penetrate intact skin, and the various other innate defence mechanisms are extremely efficient at keeping bacteria at bay. When bacteria do gain access to the tissues, the ability of the host to limit damage and eliminate the microbe depends on the generation of an effective immune response against microbial antigens. In most cases the host defences are directed against external components and secreted molecules. Bacteria are surrounded by a cytoplasmic membrane and a peptidoglycan cell wall. Associated with these basic structures there can be a variety of other components such as proteins, capsules, lipopolysaccharide or teichoic acids. There are also structures involved in motility or adherence to the cells of the host (see Ch. 2). These are some of the components to which the immune system directs its response. In general, peptidoglycan is attacked by lysosomal enzymes, and the outer lipid layer of Gram-negative bacteria by cationic proteins and complement. Specific antibodies can bind to flagella or fimbriae, affecting their ability to function properly, and can inactivate various bacterial enzymes and toxins. Antibodies therefore interfere with many important bacterial processes but, ultimately, phagocytes are needed to destroy and remove the bacteria (Fig. 12.1). In some situations cell-mediated responses are required.
Inflammation
Having successfully avoided the innate immune mechanisms that protect the individual (mechanical barriers, antibacterial substances and phagocytosis, described in Chapter 9), a bacterium starts to proliferate in the tissues. The presence of bacteria-specific molecules is recognized by pattern recognition receptors, leading to cytokine release. These cytokines, along with tissue-damaging toxic bacterial products, trigger an inflammatory reaction. The resulting increase in vascular permeability leads to an exudation of serum proteins, including complement components, antibodies and clotting factors, as well as phagocytic cells. The phagocytes are attracted to the site of inflammation by chemotactic factors. Anaphylatoxins generated by complement activation further increase vascular permeability and encourage exudation of fluid and cells at the site of inflammation. Many of these mediators also cause vasodilatation, thereby increasing blood flow to the area.
Many types of micro-organism (e.g. staphylococci and streptococci) are dealt with effectively by the phagocytes. The intensity and duration of the inflammatory process that is stimulated depends on the degree of success with which the micro-organism initially establishes itself. This, in turn, depends on the extent of the injury, the amount of associated tissue damage and the number and type of micro-organisms introduced. A localized abscess may arise at the site of infection.
If bacteria are not eliminated at the site of entry and continue to proliferate, they pass via the tissue fluids and lymphatics to the draining lymph node, where a specific acquired immune response is generated. The antibody and effector cells generated will leave the node to return to the area of infection to eliminate the bacteria. Some capsulate micro-organisms, such as pneumococci, are able to resist phagocytosis and are not dealt with effectively until large amounts of antibody have been made. These ‘mop up’ the released capsular polysaccharide, and phagocytosis occurs. Other micro-organisms produce exotoxins, and effective immunity to exotoxins requires the development of specific antibodies against the toxin (i.e. antitoxin).
The types of infection described above are usually referred to as acute infections, and contrast with the protracted or chronic infections usually induced by bacteria that have adapted to survive within the cells of the host. Included among these are tuberculosis and leprosy, brucella infections and listeriosis. In these infections, cell-mediated immunity plays a predominant part in the final elimination of the micro-organism.
Humoral immunity
The attachment of a micro-organism to an epithelial surface is a prerequisite for the development of an infectious process (see p. 159). A first line of attack by antibody could be to inhibit colonization by stopping attachment.
The immunoglobulin IgA can stop colonization of the mucosal surface if it interferes with the attachment molecules (adhesins) present on the bacterial surface. IgA does not activate complement very efficiently; therefore, an inflammatory reaction is not stimulated. Damage to the gut wall during an inflammatory reaction would allow the entry of many potential pathogens into vulnerable tissues.
Many micro-organisms owe their pathogenic abilities to the production of exotoxins. Among diseases dependent on this type of mechanism are diphtheria, cholera, tetanus and botulism. Antibodies acquired by either immunization or previous infection, or given passively as antiserum, are able to neutralize bacterial toxins.
Many bacterial exotoxins are enzymes, and protective antibody can prevent interaction of the enzyme with its substrate. The antibody can bind directly to the active site of the enzyme or to adjacent residues and inhibit by steric hindrance. Antibody may also act by stopping activation of a zymogen into an active enzyme, interfere with the interaction between the toxin and its target cell, or bind to a site on the molecule, causing a conformational change that destroys the enzymatic activity.
The direct binding of antibody to a bacterium can interfere with normal bacterial functioning in numerous ways. Antibody can kill bacteria on its own or in conjunction with host factors and cells. To survive and multiply, bacteria must ingest nutrients and ions mainly by specific transport systems. Antibodies that affect the activity of specific transport systems will deprive the bacteria of their energy supply and other essential chemicals. Some bacteria are invasive, moving into the tissues aided by enzymes that they produce. Invasion can also be inhibited by antibody that attaches to the flagella of the micro-organism in such a way as to affect its motility. Antibodies can agglutinate bacteria, and formation of the aggregate will impede spread of the organism.
In addition, the formation of an immune complex of bacteria and antibody will stimulate phagocytosis and complement activation.
When a particle is coated with antibody, a large number of Fc portions are exposed to the outside. This increases the chance that the particle will be held in contact with the phagocyte long enough to stimulate phagocytosis. The interaction of multiple ligands increases the overall affinity of the binding and, if antibody and complement components are present on the same particle, the binding is even stronger.
The bacteria are internalized and attacked by the oxygen-dependent and oxygen-independent killing mechanisms within the phagocyte. Phagocytes are also responsible for the removal and digestion of bacteria that have been killed extracellularly. Bacteria are susceptible to the lytic action of complement, which may be activated by bacterial components. The presence of antibody on the bacterial cell surface further stimulates the activation of complement. In certain circumstances, antibodies in conjunction with other bactericidal molecules lead to more efficient bacterial destruction. Gram-negative organisms are normally resistant to the action of lysozyme, probably because of the lipopolysaccharide component of the cell. The action of antibody and complement is thought to expose the underlying cell wall, which is then attacked by the lysozyme.
Cell-mediated immunity
Ultimately, all bacteria will be engulfed by a phagocyte, either to be killed or removed after extracellular killing. The host defence mechanisms of macrophages and monocytes can be enhanced by various activating stimuli, including microbial products such as the muramyl dipeptide found in many cell walls and trehalose dimycolate from Mycobacterium tuberculosis. The chemotactic formylmethionyl peptides have been shown to increase the activities of various macrophage functions. The linkage of chemotaxis to activation has the advantage that the cell being attracted to the site of tissue injury will be better equipped to deal with the insult. Endotoxins present in the cell wall of Gram-negative bacteria and various carbohydrate polymers, such as β-glucans, are potent macrophage activators.
The immune system is also active in the production of macrophage-activating factors. In particular, lymphokines, produced by T lymphocytes, are often required to potentiate bacterial clearance both by attracting phagocytes to the site of infection and by activating them. The most important activator is γ-interferon, although tumour necrosis factor and colony-stimulating factors have also been implicated (see Ch. 9). All T lymphocytes produce some lymphokines when stimulated, and the balance of the different factors produced dictates the effect on surrounding cells. The overall effect of the lymphokines is to increase the effectiveness of host defence mechanisms, but if these molecules are produced in excess or to an inappropriate signal then a type IV hypersensitivity reaction can occur.
Evasion
Once a micro-organism becomes established in the tissues, having escaped the innate defence mechanisms, it can often make use of a number of evasion strategies that protect it from the immune reactions of the host. Pathogenic bacteria produce a rather ill-defined group of bacterial products called aggressins and impedins, possession of which is associated with virulence (see Ch. 13). If antibody to such substances is present, the pathogenicity of the micro-organism is likely to be reduced.
Intracellular bacteria
Bacteria that can survive and replicate within phagocytic cells are at an advantage as they are protected from host defence mechanisms. Some micro-organisms reside intracellularly only transiently, whereas others spend most, if not all, of their life inside cells. An intracellular lifestyle demands potent evasion mechanisms to survive this hostile environment. Some bacteria, such as Mycobacterium leprae, have become so accustomed to their intracellular environment that they can no longer live in the extracellular space.
Listeria monocytogenes, the causative agent of listeriosis, can survive and multiply in normal macrophages, but is killed within macrophages activated by lymphokines released from T lymphocytes. Listeriosis is most commonly seen in immunocompromised patients, pregnant women and neonates, in whom a lack of adequate T cell-derived macrophage-activating factors is probably the critical factor. Salmonella spp. and Brucella spp. can also survive intracellularly. Mycobacteria have a waxy cell wall that is very hydrophobic. This external surface is very resistant to lysosomal enzymes and persists for a long time even when the bacteria have been killed. In addition, these micro-organisms have evolved other strategies to evade destruction. M. tuberculosis secretes molecules that inhibit lysosome/phagosome fusion, and M. leprae can escape from the phagosome and grow in the cytoplasm. The cell wall of both of these bacteria contains lipoarabinomannan, which blocks the effects of γ-interferon on macrophages.
T lymphocytes represent the major host defence against intracellular pathogens. In many cases the bacteria themselves do not directly harm the host: the pathogenesis is caused by the immune response. After invasion of the host, intracellular bacteria are taken up by macrophages, evade intracellular killing and multiply. During intracellular replication some microbial molecules are processed and presented on the surface of the infected cell in association with major histocompatibility complex (MHC) gene products. The exact nature of the microbial molecules and the processing mechanism are not known. The processing appears to produce mainly antigen fragments in association with MHC class II molecules. This complex on the cell surface is recognized by specific CD4+ T cells, which are stimulated to release lymphokines. These molecules in turn activate the macrophage so that the intracellular bacteria are killed.
It has also been proposed that peptides derived from the bacteria can become associated with MHC class I molecules. This complex leads to destruction of the infected cell by CD8+ T cells. These cells also produce lymphokines that can aid in the elimination of the infection. The lymphokines produced attract blood monocytes to the site and activate them. If these newly recruited cells take up the released mycobacteria they are more likely to destroy them as they will be in an activated state. The accumulation of macrophages also causes the formation of a granuloma, which will prevent dissemination of the bacteria to other sites in the body. As conditions for the survival of the pathogens become less suitable, they stop replicating and die. Some intracellular bacteria infect cells that are unable to destroy them. In this situation a more aggressive immune response may be needed to remove the pathogen and this can cause pathogenic damage unless kept under control (see below).
Immunopathology
The immune response to an organism leads to some tissue damage through inflammation, lymph node swelling and cell infiltration. Sometimes the damage caused by the immune system is very severe, leading to serious disease and death. Rheumatic fever can follow group A streptococcal infections of the throat and is believed to be due to antibodies formed against a streptococcal cell wall component cross-reacting with cardiac muscle or heart valve. Myocarditis develops a few weeks after the throat infection and can be restimulated if the patient is reinfected with different streptococci.
Immune complex disease (type III hypersensitivity) is frequently associated with bacterial infection. Infective endocarditis caused by staphylococci and streptococci is associated with circulating complexes of antibody and bacterial antigen. Detection of these complexes can be helpful in diagnosis, but they may lead to joint and kidney lesions, vasculitis and skin rashes. Immune complexes may play a role in the pathogenesis of leprosy, typhoid fever and gonorrhoea.
Effects of endotoxin
Endotoxin interacts with cells and molecules of inflammation, immunity and haemostasis.
• Fever is induced by interleukin-1, produced by the liver in response to endotoxin, acting on the temperature-regulating hypothalamus.
• The action of lipopolysaccharide on platelets and activation of Hageman’s factor causes disseminated intravascular coagulation with ensuing ischaemic tissue damage to various organs.
• Septic shock occurs during severe infections with Gram-negative organisms when bacteria or lipopolysaccharide enter the bloodstream.
Endotoxin acts on neutrophils, platelets and complement to produce, both directly and through mast cell degranulation, vasoactive amines that cause hypotension. The mortality rate is very high.
Recognition of endotoxin through Toll-like receptor 4 (see p. 111–112) causes macrophages to produce large quantities of potent cytokines such as interleukin-1, tumour necrosis factor and colony-stimulating factors. Endotoxin also causes polyclonal activation of B cells and can stimulate natural killer cells and other cell types to produce γ-interferon. A substantial part of the pathogenesis of endotoxin shock is probably due to the production of these molecules by cells of the immune system. In small amounts, endotoxin may actually be beneficial to the host, but when present in excess the results can be disastrous.
Mycobacterial disease
Activated macrophages secrete a variety of biologically active molecules, including:
Tissue destruction is an inevitable side effect of this important mechanism of resistance. In the acute phase of a response this is likely to be tolerated; however, in the case of resistant organisms such as mycobacteria, the process may become chronic and the tissue destruction extensive. Mycobacterial components are still able to stimulate a response after the bacterium has been killed, because they persist for a long time, adding to the tissue damage.
There is evidence that lysis of infected cells may also occur. At first sight this may appear beneficial. Such a direct effect may be particularly relevant in the case of obligate intracellular pathogens such as M. leprae. Release into the hypoxic centre of a productive granuloma may also be fatal for M. tuberculosis, which is highly sensitive to low oxygen pressures. The same cytolytic event may also result in microbial discharge from the granuloma into surrounding capillaries or alveoli, and hence facilitate dissemination to other parts of the body or to other individuals. Lysis of infected cells causes tissue destruction, the severity of the effects depending on the importance of the tissue involved. M. leprae infects the Schwann cell, an irreplaceable component of the peripheral nervous system. Although the presence of M. leprae does not appear to affect the host cell to any extent, the presence of activated macrophages releasing toxic molecules or direct lysis by cytotoxic cells constitutes a major pathological mechanism in leprosy.
If the leprosy bacillus is released from a lysed non-phagocytic cell to be engulfed by an activated macrophage, the bacterium will be eliminated. Therefore, macrophage activation and target cell lysis can be beneficial as well as detrimental to the host.
The cell-mediated response that has the potential to eliminate these infections will give rise to type IV hypersensitivity reactions if the antigen is not removed efficiently. Chronic production of lymphokines causes granuloma formation, which with time can lead to fibrosis and loss of organ function. This type of response is particularly prevalent in patients with tuberculosis.