14 The natural history of infection
Key points
• Symptomatic infection is a rare outcome when human beings and micro-organisms meet. None the less, some organisms, known as obligate pathogens, must cause disease to survive; this applies to most viral pathogens. Many bacterial pathogens appear to derive little benefit from causing infection.
• Micro-organisms that form short- or long-term associations with humans do so in a number of recognizable forms and stages, including: entry/establishment, colonization, commensalism, spread, survival and multiplication, damage, and carriage. Where these associations lead to bacterial disease, the different stages are often associated with the virulence determinants described in Chapter 13.
• Infections can generally be recognized in one of four categories: toxin mediated, acute, subacute and chronic.
• The capacity of a particular micro-organism to cause disease is known as its virulence. Where suitable experimental models are available, we can recognize virulence quantitatively by the ability of a low number of organisms to produce infection or death in the host population. Such measurements can be useful in identifying virulence determinants and in developing vaccines.
The purpose of this chapter is to link the basic properties of micro-organisms with the patterns of infective disease experienced in public health and clinical practice, and with the tissue and organ pathology that can be observed. Although there are numerous exceptions to the general patterns described, the intention is to give an underlying structure that can be used to make sense of the many different types of organism and the diseases they cause. The term ‘natural history’ is used in two senses here: first, to denote an overall biological consideration of the life cycle of the infective agent and how this intersects with the human host and, second, to consider the process of infection from the point of the encounter between the agent and the susceptible host through to its outcome.
Meetings between human beings and micro-organisms
The vast majority of micro-organisms do not form stable associations with human beings. Clearly pathogens must do so, at least temporarily. However, it is worth briefly considering how important this association is to the micro-organism concerned. In some cases, humans constitute the only environment in which micro-organisms can survive (i.e. they are obligate parasites of human beings). Thus, humans are the reservoir and immediate source of the infections caused by this group (see Ch. 1). In other cases colonization or infection of human beings may be entirely incidental to the life cycle of the organism. These organisms may need to live in animals, as in the case of zoonoses, or in specific environmental reservoirs. Their life cycles in these habitats are critical to the epidemiology of the infections they cause. This biological perspective is difficult to avoid when considering the complex life cycles of parasitic protozoa and helminths, but such considerations are equally applicable to bacterial and viral pathogens.
In the previous chapter a division was made between primary and opportunistic pathogens. Here this division is maintained but the primary group is further subdivided.
Obligate pathogens
These organisms have to cause disease in human beings in order to continue to survive and propagate. This is true for most viruses that cause human disease and for which humans are the only natural host. The major caveat to this is that the degree to which these agents cause symptomatic infections can vary over a very wide range. Thus asymptomatic infections with smallpox were virtually unknown, whereas they are very common with polio. This contributed substantially to the eradication of smallpox as it was relatively straightforward to identify where transmission was taking place. Among bacterial pathogens, Mycobacterium tuberculosis is a prime example of an agent that has to cause symptomatic disease in order to survive and propagate. In some cases pathogenicity reflects an early stage in the development of host-parasite relations, with pathogens evolving towards a more benign association with their host. Clearly, if a parasite kills all of its potential hosts then it has destroyed its own habitat. However, this does not always hold true. Some pathogens actually become more virulent as a means of increasing their potential to survive.
Accidental or incidental pathogens
This term applies to many bacterial pathogens. Causing disease confers no obvious biological advantage on the organism and indeed may be a dead end. There are two groups of bacterial pathogen for which this is probably the case. The first group have their natural habitat in humans but cause disease in only a small minority; these include the major pathogens of bacterial pharyngitis (Streptococcus pyogenes), acute pneumonia (Streptococcus pneumoniae) and the principal agents of acute pyogenic meningitis (Streptococcus pneumoniae, Neisseria meningitidis and Haemophilus influenzae type b). The second group have a habitat (or reservoir) in nature but, if they encounter a susceptible host in a particular way, infection may ensue. For example, the agent of cholera, Vibrio cholerae, lives in brackish water and causes human disease only when ingested. Clostridium tetani, the agent of tetanus, probably propagates in animal gastrointestinal tracts and infects wounds contaminated by soil containing animal excreta.
Pathogens in the environment
Whatever the method of acquisition, the organism must survive long enough to encounter a susceptible human host if it is going to cause human disease. The dynamics of pathogen survival in various environments are relevant to the control of infection. The capacities of different pathogens to survive and propagate in food and water are of particular concern, as is survival in aerosols and through desiccation and many other common environmental stresses. These properties provide the biological basis for the transmission of infection and many opportunities for improved control of specific pathogens.
Stages of infection
Most infections can be broken down into a core series of steps:
It should be noted that the virulence determinants described in Chapter 13 were related to all but the first and last of these stages. Most pathogens can cause infection only via a limited set of routes (see above). Thus Vibrio cholerae must be ingested; it cannot cause infection if rubbed on the skin. Human immunodeficiency virus (HIV) must gain access to circulating CD4+ cells via a parenteral route, and so on. Some general points concerning the passage through alternative stages of infection are made in Figure 14.1. Note that, although the simple direct pathway (A) reflects the norm for an exogenous infection, there are intermediates between this and D, endogenous infection. A single organism may be capable of following multiple routes to infection. For example, Staphylococcus aureus may be introduced exogenously into a wound. Around 30% of individuals are colonized with this organism at any point in time but in only one-third of these (10%) does the organism appear to be a member of the normal microbiota. Both temporary and permanent relationships may provide for endogenous infections due to Staph. aureus.
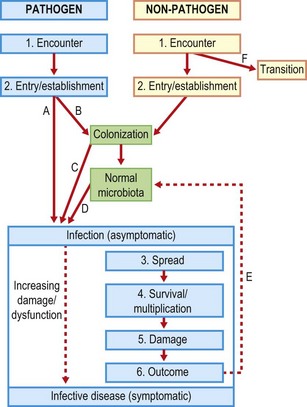
Fig. 14.1 Pathways to and stages of infection following encounter between a host and a micro-organism. The blue sector includes the possible outcomes for a pathogen, the yellow a non-pathogen, and the green either of these. An organism detected in a diagnostic laboratory might reflect one or more of the stages identified, including transition. The possibility of transition from yellow to blue zones reflects the lack of a rigid division between pathogen and non-pathogen, and one aspect of opportunism. Detection in a diagnostic laboratory is most likely following colonization or multiplication. Infection may become apparent as a result of many different pathways: A, directly, without any colonization phase; B, many pathogens colonize first then proceed to infection either C after a brief period of colonization or D after a sustained period in which they live as commensals as part of the normal microbiota (progression here is not inevitable). Note that infection becomes symptomatic only when the level of damage or dysfunction is sufficient. At the end of the symptomatic infection a small number of pathogens may enter the normal microbiota (E) and the convalescent host is then described as a carrier for the organism concerned. F, Some organisms may simply be passing through and form no stable association with the host.
Many opportunistic pathogens become part of the normal microbiota before they cause infection. They may be assisted in colonizing a new host by interventions such as repeated use of antibiotics. This appears to be the case with Pseudomonas aeruginosa, which is resistant to most routinely used antibacterial agents, and is probably also the case for methicillin-resistant Staph. aureus (MRSA) and vancomycin-resistant enterococci (VRE). Most members of the normal microbiota appear to have very little capacity to cause disease. The number and identities of readily culturable bacteria varies in different parts of the body (Table 14.1) and it is clear that a ‘healthy’ normal microbiota provides some protection against invading pathogens. Indeed the composition of the microbiome of particular individuals may prove to be critical in the balance between health and disease. As noted earlier, culture methods have only been able to reveal a minority of the bacterial species present in different samples and there is considerable excitement at the potential yield of a project to characterize all the organisms present by nucleic acid sequencing in the Human Microbiome Project.
Table 14.1 Normal human microbiotaa
| Locationb | Compositionc | Abundanced |
|---|---|---|
| Dry skin (face, forearm) | Gram-positive anaerobes (e.g. propionibacteria) | 102 |
| Moist skin (axilla, groin) | Staphylococci (esp. coagulase negative); corynebacteria; Gram-negatives rare but more frequent after prolonged hospital stay | 106–7 |
| Oropharynx | Anaerobes; streptococci; neisseria; candida | 109 |
| Small intestine | Anaerobes, lactobacilli, peptostreptocccus, porphyromonas | 105–7 |
| Large intestine | Anaerobes, clostridium, bacteroides; enterobacteria; enterococci, candida, protozoa | 109–11 |
| Vagina | Anaerobes, lactobacillus; streptococci, candida | 108–9 |
a The term microbiota is preferred to ‘flora’ as the latter refers back to a period when bacteria were classified with plants.
b These are extremely broad. In practice each micro-niche in the body constitutes a different environment colonized with different organisms; for example, the microbiota associated with the lumen and the mucosa of the gut are different.
c A very rough introductory guide. Note the predominance of anaerobes.
d Per square centimetre of surface or gram of fluid.
The survival of humankind to the present day reflects the fact that most untreated infections are not fatal. Indeed, many human genes, particularly those concerned with the immune response, clearly reflect the selection pressure provided by infection. Before the development of antibiotics and immunization at least 50% of deaths were attributable to infection (this is still the case in many resource-poor countries). Nevertheless, owing to our inherent and highly efficient defence mechanisms, many infections resolve without medical intervention. All doctors must have some skill in recognizing those infections for which an intervention is unnecessary. This is particularly important in the case of antibiotic use because of the dangers of encouraging resistance.
Pathological patterns associated with infection
All of the foregoing reflects a set of proposed mechanisms that, by and large, fit and make sense of the available facts concerning the epidemiology and detailed pathogenesis of infection. In this section the link to clinical practice is developed by describing the pattern of pathology directly observable in various infections. Most infections can be placed into one of four patterns:
1. Toxin mediated (mainly bacterial).
2. Acute (including acute viral syndromes and acute pyogenic bacterial infections).
3. Subacute (many virus and several ‘atypical’ bacterial infections).
4. Chronic (chronic viral infections, chronic granulomatous bacterial, fungal and parasitic infections).
Simply by recognizing the basic characteristics of a suspected infection against these four possibilities, the possible range of causal agents can be narrowed down substantially. Their features are summarized and exemplified from the perspective of bacterial infections in Table 14.2.
Table 14.2 Patterns in the presentation and pathology of bacterial infection
| Pattern | Examples |
|---|---|
| Toxin-mediated disease | |
| Pathology often distant from site of bacterial growth | Diphtheria, tetanus |
| Protective immunity may be mediated by anti-toxin antibodies alone | Staphylococcal food poisoning, cholera |
| Disease may be fully reproduced by administering the toxin alone | Pseudomembranous colitis |
| Acute pyogenic infection | |
| Generally rapid growing organisms | Streptococcal pharyngitis |
| Interaction with innate immune system and acute inflammation predominates | Staphylococcal abscess, bacterial meningitis, lobar pneumonia, acute cystitis |
| Where immune damage occurs, it is ‘post-infective’ | Post-streptococcal glomerulonephritis |
| Subacute infection | |
| No pattern to growth rate | Subacute bacterial endocarditis |
| Site of infection may be only partially accessible to the immune system | ‘Atypical’ pneumonia |
| Immunopathology often in parallel with direct effects of organism | |
| Chronic (granulomatous) | |
| Bacterial growth rate often moderate or slow | Tuberculosis, brucellosis |
| Organisms often survive and grow intracellularly | |
| Immune damage occurs with infection – predominantly cell mediated | (Some fungal and parasitic infections have this pattern) |
Toxin-mediated bacterial infections
This was the first recognized pathogenic mechanism in bacterial infection and resulted in early successful therapeutic and preventive measures. When a single toxin is responsible for most of the features of an infection, the dysfunction or damage is often distant from the site of bacterial multiplication, the disease may be reproduced by administration of the pure toxin alone and it can be prevented with antibodies directed against the toxin. The clostridial diseases tetanus and botulism are toxin-mediated. In the latter case, as in several other forms of food poisoning, ingestion of only the toxin is required, so many cases of botulism are not strictly infections. Once the pathogen has grown and produced toxin, the onset of disease can be very rapid.
It is often possible to abolish the biological activity of toxins without affecting their immunogenicity. Such toxoids were among the first effective immunizations against bacterial infection. Diphtheria and tetanus toxoid vaccines have controlled these infections in the UK. In life-threatening toxin-mediated disease, the administration of pre-formed antibodies can be life-saving. Antibiotics are not effective in treating established disease, but may prevent further toxin formation. In the special case of Escherichia coli O157 infections, however, some antibiotics actually stimulate further synthesis of toxin.
Acute pyogenic bacterial infections
Pyogenic means pus inducing. Pus is composed primarily of live and dead neutrophil polymorphs. The presence of pus generally reflects an acute inflammatory process and activation of the innate immune system. The inflammatory process may be localized, as in the formation of an abscess, or more disseminated through tissue planes. Anything more than a trivial acute pyogenic infection is usually accompanied by an increase in the blood neutrophil count. The acuteness of these infections is reflected in their rapid onset. Accordingly the bacteria that cause them generally grow rapidly, producing visible colonies within 24 h of inoculation. Medical intervention is most effective when given early in infection before the development of acquired immunity, which, when successful, terminates the illness. Serological evidence of acquired immunity cannot be used in the diagnosis of infection during its acute phase. Occasionally, immunopathology occurs after the causal organism is no longer detectable in the host; classic examples are poststreptococcal glomerulonephritis and rheumatic fever. Similarly, Guillain-Barré syndrome, a paralytic disease, sometimes follows acute campylobacter infection.
Many bacteria that cause acute pyogenic infections also produce toxins. Thus there may be both acute pyogenic and toxin-mediated components to the damage and dysfunction that develops. This is particularly true of staphylococcal and streptococcal infections. The complex mixture of the pathogenic processes attributable to different virulence determinants can make the most severe of these infections very difficult to treat.
Subacute bacterial infections
These have a more insidious onset than acute infections and are accompanied by less prominent signs of acute inflammation. Classically, bacterial endocarditis was described as subacute, although this is no longer considered a suitable catch-all term for this type of infection. Because such diseases have a more protracted course, the adaptive immune response often contributes to damage. Hence subacute forms of bacterial endocarditis are often accompanied by immune complex-mediated pathology, whereas Mycoplasma pneumoniae infection (a form of atypical pneumonia) may be accompanied by several different immunopathological reactions reflecting specific immune responses (see Ch. 41).
Chronic granulomatous bacterial infections
When bacterial infections persist over months or even years they tend to elicit a pathological entity known as a granuloma. Granulomas are a common form of localized cell-mediated immune response directed to antigens or other foreign bodies that appear to be refractory to elimination from tissues. An ordered accumulation of lymphocytes and macrophages occurs around a central focus in a manner that, to the experienced pathologist, can be more or less specific to the eliciting stimulus. Persisting bacterial infections, notably those due to mycobacteria (e.g. tuberculosis) and, to a lesser extent Brucella spp., produce chronic granulomatous infections. The agents concerned are generally slow growing and have the capacity to survive inside host cells, notably macrophages. Cell-mediated immunopathology (delayed-type hypersensitivity; see Ch. 9) is a prominent feature of these infections.
Timing of key events in infection
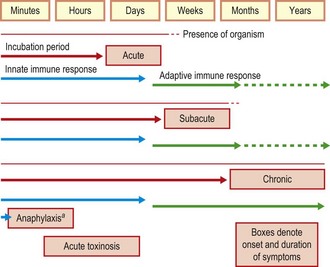
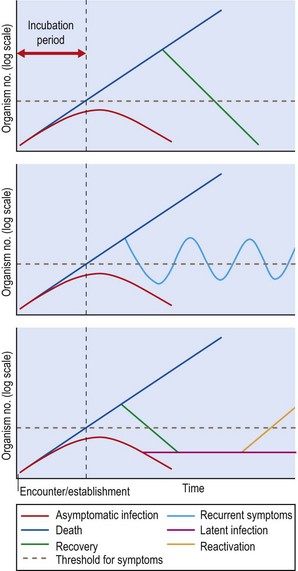
As different infections proceed at different rates, the timing of the symptoms, their relation to immune responses and the ability to detect the causal agent all vary. The incubation period, the time between the encounter with the pathogen and the onset of symptoms, is an important practical consideration in understanding and managing infection. This is characteristic for different pathogens and can be vital in determining whether an individual is still at risk of developing disease after exposure to a particular agent. Incubation periods for the four patterns of infection discussed above are illustrated in Figure 14.2, along with the time-frames over which immune responses and presence of the pathogen are expected. A more dynamic view of individual infections is shown in Figure 14.3, in which the additional concepts of recurrent, latent and reactivated infections are illustrated. Figure 14.3 introduces the notion that the progression of an infection is related to the numbers of the pathogen. Although many other factors are involved, the concept is useful because it illustrates how, in some rapidly developing infections, the interval between the onset of symptoms and death may be short. The slope of increasing pathogen numbers clearly also reflects the balance between growth of the pathogen and the efforts of the immune system to resist. Accordingly, when the immune system is suppressed, progression may be exceptionally rapid and the response must be equally so.
Virulence and infectivity
By now it should be apparent that what makes a pathogen more or less virulent is, in most cases, extremely complex. None the less, when infections can be studied in animal experimental systems, virulence can be seen as a quantifiable property. As the dose administered to a group of susceptible hosts is increased, the number acquiring infections also increases in a fairly well defined dose–response relationship (Fig. 14.4). Not only does recognition of this relationship help explain why, when a group of individuals is exposed to a pathogen only some get infected, it also provides a framework for understanding the effects of immunization and immune deficiency, as well as a systematic basis for identifying individual traits that contribute to virulence and host resistance.
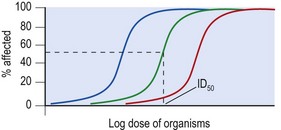
Fig. 14.4 Quantitative relationships between organism dose and outcome in experimental infection. The organism has been administered by a single route to a uniform host population. Note that the outcome (percentage affected – which could be percentage infected, percentage who died or many other endpoints) depends on the dose. This approach allows virulence to be compared between strains of a particular micro-organism. A more virulent organism shifts the curve to the left (blue curve) and a less virulent organism to the right (red curve). Lesser or greater host resistance would, respectively, have the same effect. The approach allows for the specific recognition of virulence determinants and the effects of immunization. The dose required to produce the specified endpoint in 50% of the target population is often reproducible and can be used for statistical comparisons. The 50% infected endpoint is known as the ID50.
Burnet FM. Natural History of Infectious Diseases. Cambridge: Cambridge University Press; 1970.
Ewald PW. Evolution of Infectious Disease. USA: Oxford University Press; 1996.
Mims C, Nash A, Stephen J. Mims’ Pathogenesis of Infectious Disease, ed 5. London: Academic Press; 2000.
Salyers AA, Wilson BA, Whitt DD. Bacterial Pathogenesis: A Molecular Approach, ed 3. Washington, DC: ASM Press; 2011.