41 Mycoplasmas
Respiratory and genital tract infections
Key points
• Mycoplasmas pathogenic for man include the genera Mycoplasma and Ureaplasma. They are found in the respiratory and genital tracts of man and many animal species, and sometimes invade the bloodstream to gain access to joints and other organs.
• Mycoplasmas are the smallest organisms that grow in cell-free bacteriological media. They lack a rigid cell wall containing peptidoglycan; hence they are not susceptible to β-lactam or glycopeptide antibiotics. Infections are treated principally with macrolides, tetracyclines or fluoroquinolones.
• M. pneumoniae is a pathogen for the human respiratory tract, sometimes resulting in pneumonia, especially in children.
• M. genitalium and U. urealyticum cause acute non-gonococcal urethritis in men and are also implicated in chronic disease; M. genitalium also causes balanoposthitis.
• Various mycoplasmas and ureaplasmas are associated with genital infections in women, including bacterial vaginosis, post-partum fever, cervicitis, endometritis and salpingitis.
• Mycoplasmas, most often ureaplasmas, are responsible for about two-fifths of the cases of suppurative arthritis occuring in patients with hypogammaglobulinaemia. M. fermentans has been detected in the joints of patients with chronic arthritides.
Mycoplasmas are the smallest prokaryotic organisms that can grow in cell-free culture medium. They are found in man, animals, plants, insects, soil and sewage. The first to be recognized, Mycoplasma mycoides ssp. mycoides, was isolated in 1898 from cattle with pleuropneumonia. As other pathogenic and saprophytic isolates accumulated from veterinary and human sources they became known as pleuropneumonia-like organisms (PPLO), a term long superseded by ‘mycoplasmas’. Mycoplasma (Greek: mykes, fungus; plasma, something moulded) refers to the filamentous (fungus-like) nature of the organisms of some species and to the plasticity of the outer membrane resulting in pleomorphism.
The term mycoplasma(s) is often used, as here, in a trivial fashion to refer to any member of the class Mollicutes (’soft skins’), which embraces 5 families, 8 genera and over 200 species. The family Mycoplasmataceae contains the mycoplasmas that are of importance in human medicine. It is subdivided into two genera:
• Mycoplasma, of which there are at least 120 named species, 14 of which are of human origin.
• Ureaplasma, urea-hydrolysing organisms (trivially termed ureaplasmas), of which there are seven species, two of which are of human origin.
Organisms in the genus Spiroplasma cause disease in plants and insects, and those in genus Acholeplasma are ubiquitous in nature. Several species of Haemobartonella and Eperythrozoon are also placed within the genus Mycoplasma on the basis of their 16S ribosomal RNA (rRNA) sequence similarities.
Mycoplasmas are distributed widely in nature, and various species cause economically important infections in domestic animals, other mammals, birds and reptiles as well as man. In addition, mycoplasmas are of concern to those who use cell cultures because mycoplasmal contamination may severely affect the cells.
Ureaplasmas were known originally as T strains or T mycoplasmas – ‘T’ for ‘tiny’ to indicate the small size of the colonies in comparison with those produced by other mycoplasmas. Many animals are infected by ureaplasmas. Those of avian, bovine, canine and feline origin are antigenically distinct from the human strains and have been placed in separate species. Ureaplasmas of human origin, formerly classified as Ureaplasma urealyticum, comprise at least 14 serovars and fall into two groups differentiated in several ways, most importantly by genome size. Strains of small genome size include the serovars 1, 3, 6 and 14 within the species U. parvum, whereas those of larger genome size include the other ten serovars and these remain within U. urealyticum.
Description
Reproduction
Mycoplasmas, like conventional bacteria, multiply by binary fission. However, cytoplasmic division may not always be synchronous with genome replication, resulting in the formation of multinucleate filaments and other shapes. Subsequent division of the cytoplasm by constriction of the membrane at sites between the genomes leads to chains of beads that later fragment to give single cells. Budding occurs when the cytoplasm is not divided equally between the daughter cells. The minimal reproductive unit of mycoplasmas is a roughly spherical or bottle-shaped cell about 200–250 nm in diameter. Organisms of this order initiate growth in cell-free medium and make up, with larger forms (0.5–1.0 µm diameter), the substance of the characteristic agar-embedded colonies.
Morphology
Cell morphology varies with the species, environmental conditions and the stage of the growth cycle. Light microscopy reveals pleomorphic organisms, which may range from spherical through coccoid, coccobacillary, ring and dumb-bell forms, to short and long branching (Fig. 41.1), beaded or segmented filaments.
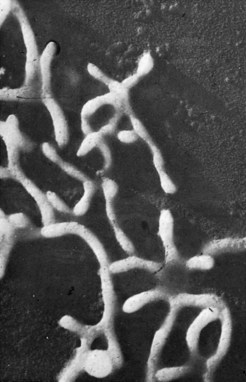
Fig. 41.1 Electron micrograph of M. mycoides ssp. mycoides (bovine origin), gold shadowed, to show branching filaments (original magnification ×28 000).
(From Rodwell AW, Abbot A 1961 The function of glycerol, cholesterol and long-chain fatty acids in the nutrition of Mycoplasma mycoides. Journal of General Microbiology 25: 201–214.)
Several mycoplasmas, including M. pneumoniae, M. genitalium (Fig. 41.2) and M. penetrans of human origin, have specialized structures at one or both ends by which they attach to respiratory or genital tract mucosal surfaces. Sections of the terminal structures of the two former mycoplasmas may exhibit a dense rod-like core when viewed by electron microscopy.
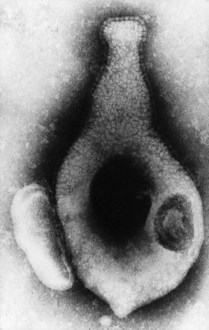
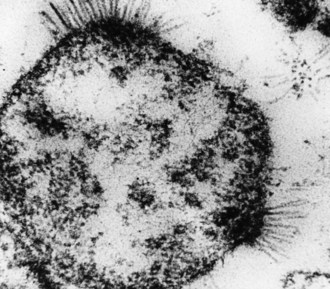
Fig. 41.2 Electron micrograph of M. genitalium (human origin), negatively stained, to show flask-shaped appearance and terminal specialized structure covered by extracellular ‘nap’ (original magnification ×120 000).
(From Tully JG, Taylor-Robinson D, Rose DL et al 1983 Mycoplasma genitalium, a new species from the human urogenital tract. International Journal of Systematic Bacteriology 33: 387–396.)
Mycoplasmas, unlike conventional bacteria, do not have a rigid cell wall containing peptidoglycan. This also differentiates them from bacterial L-forms for which the absence of the cell wall is a temporary environmental change. The mycoplasma cell is limited by a membrane, 7.5–10 nm wide, in which two electron-dense layers are separated by a translucent one (Fig. 41.3). Some species have an extramembranous layer, which, in the case of M. mycoides ssp. mycoides, for example, comprises galactan and has a dense capsular appearance. Some others, including M. genitalium and M. pneumoniae, have surface spikes (sometimes described as a ‘nap’), somewhat coarser than those seen on myxoviruses, which may contribute, through adhesin proteins, to attachment of the organisms to eukaryotic cells. M. gallisepticum (avian), M. genitalium and M. pneumoniae attach to neuraminic acid receptors. Close adherence enables the mycoplasma to insert nucleases and other enzymes into the cell and to take from it the products of enzyme activity, such as nucleotides. Adherence to erythrocytes (haemadsorption), tissue culture cells, spermatozoa and other eukaryotic cells may be demonstrated with certain mycoplasmas, including M. pneumoniae, M. genitalium and M. agalactiae (shown in Fig. 41.4). Some mycoplasmas, including M. pneumoniae, M. genitalium and M. hominis, invade eukaryotic cells.
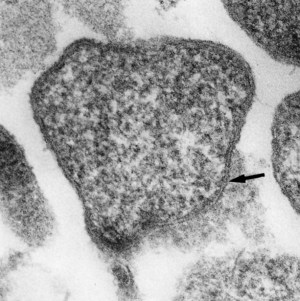
Fig. 41.3 Electron micrograph of M. pulmonis (of murine origin); thin section illustrating trilaminar membrane (arrow) (original magnification ×75 000).
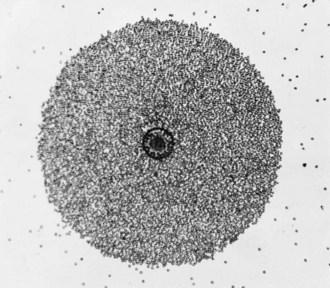
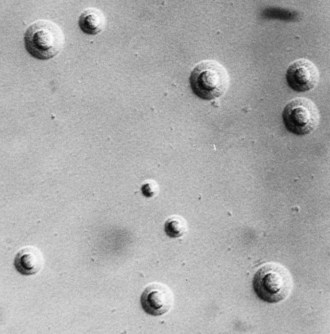
Fig. 41.4 Colony of M. agalactiae (of caprine origin) showing adherent guinea-pig erythrocytes (phenomenon of haemadsorption).
The electron-lucent part of the membrane comprises lipids with the long chains of the fatty acids arranged inwards and polar groups to the external and internal parts of the organisms; the electron-dense layers consist of protein and carbohydrate. The mycoplasma membrane is the site of many metabolic reactions involving membrane-bound enzymes and transport mechanisms. Cholesterol, or carotenoid/carotenol, is interspersed between the phospholipid molecules and plays an important part in maintaining membrane integrity in the face of varying external osmotic pressures and in the absence of the rigid cell wall found in other bacteria. However, the presence of cholesterol renders the cell membrane susceptible to damage with agents such as saponin, digitonin and some polyene antibiotics (e.g. amphotericin B) that complex with sterols. As expected, mycoplasmas are completely resistant to β-lactam and other antibiotics that influence bacterial cell wall synthesis and also to lysozyme.
Mycoplasmas are prokaryotes (p. 10) and the cytoplasm does not contain endoplasmic reticulum, but is packed with ribosomes. Ribosomal protein synthesis is inhibited by antibiotics such as tetracyclines, aminoglycosides and macrolides.
The genome of M. genitalium is the smallest known so far (580 kb). It is not much larger than that of a large poxvirus, an indication of the small amount of genetic information needed for a free-living existence and the reason why mycoplasmas have a paucity of biochemical activity and are nutritionally fastidious.
Viruses and plasmids
Fourteen viruses have been identified: six in Acholeplasma, four in Mycoplasma and four in Spiroplasma species. They are rod-shaped (Fig. 41.5) or enveloped spheres that bud from the mycoplasma membrane surface, or are polyhedral with a tail. Those that have been examined in detail contain single- or double-stranded DNA in circular or linear form. There is evidence for integration of the viral genome into the mycoplasmal chromosome, especially in spiroplasmas, and this may provide a mechanism for the promotion of genetic diversity. There is no evidence, however, that the viruses influence mycoplasmal pathogenicity. Viral release is continuous and is not accompanied by cell lysis. Plasmids, so far regarded as cryptic, have been detected in Acholeplasma, Mycoplasma and, most frequently, Spiroplasma species.
Cultivation
Mycoplasmas have limited biosynthetic abilities, so that they need a rich growth medium containing natural animal protein (usually blood serum) and, in most cases, a sterol component. Serum supplies not only cholesterol but also saturated and unsaturated fatty acids for membrane synthesis, components that the organisms cannot synthesize. A medium that has been widely used for isolation contains bovine heart infusion (PPLO broth) with fresh yeast extract and horse serum. However, these components vary in their ability to support growth and success may depend on use of different batches of the components, sera from other animal species or the addition of various supplements.
Cultivation of spiroplasmas is more difficult. A medium designated SP-4 has been helpful in their isolation and also in the isolation of fastidious mycoplasmas, such as M. pneumoniae, M. fermentans and M. genitalium, which is notoriously difficult to isolate. Although various specific formulations have been described for the isolation of ureaplasmas, most mycoplasmal media containing urea may be suitable. The sterol-independent organisms also grow readily on these media, but serum, which often promotes growth, is not usually essential.
A strategy using Vero (African green monkey) cell cultures has been described for isolating M. genitalium. The inoculated cell cultures are monitored for mycoplasmal growth by polymerase chain reaction (PCR) technology followed, when there are signs of growth, by subculture to mycoplasmal medium. However, investigators should be aware of the dangers inherent in the widespread use of cell cultures which may become contaminated by mycoplasmas.
Most mycoplasmas are facultatively anaerobic but, because organisms from primary tissue specimens often grow only under anaerobic conditions, an atmosphere of 95% nitrogen and 5% carbon dioxide is preferred for primary isolation. The optimal temperature for growth of most mycoplasmas and ureaplasmas from man or animals is 36–38°C, but is lower for acholeplasmas and spiroplasmas.
Colonial morphology
On agar, mycoplasmas often produce colonies that have a ‘fried egg’ appearance, with an opaque central zone of growth within the agar and a translucent peripheral zone on the surface (Fig. 41.6). However, some, such as M. pneumoniae on primary isolation, have a mulberry appearance without the peripheral zone. The size of the colonies varies widely: colonies of some bovine mycoplasmas and most acholeplasmas may exceed 2 mm in diameter, and are visible to the naked eye. Nevertheless, most require low-power microscopic magnification. Colonies of ureaplasmas are characteristically small (15–60 µm in diameter), mainly because they usually lack the peripheral zone of growth. However, the size and appearance of all mycoplasmal colonies depend on the constituents and degree of hydration of the medium, the agar concentration, atmospheric condition and age of the culture.
Biochemical reactions
Most mycoplasmas use glucose (or other carbohydrates) or arginine as a major source of energy; a few use both or do not metabolize either substrate. In most mycoplasmas the respiratory pathways are flavin-terminated so that the haem compounds, cytochromes and catalase are absent. The unique and distinctive biochemical feature of ureaplasmas is the conversion of urea to ammonia by urease.
Mycoplasma, Ureaplasma and certain other species depend on sterol. Thus, they fail to grow in serum-free media and are inhibited by digitonin, distinguishing them from the species that do not require sterol. In addition, most Mycoplasma species and ureaplasmas produce hydrogen peroxide, which causes some lysis of guinea-pig or other erythrocytes when these are suspended in agar over developing colonies. M. pneumoniae produces clear zones of β-haemolysis. About one-third of all Mycoplasma species display the phenomenon of haemadsorption (Fig. 41.4).
Antigenic properties
DNA analysis and sequencing has made a fundamental contribution to the taxonomy of mycoplasmas. Amplification of part of the 16S rRNA gene by PCR plays an important role in the rapid identification of some mycoplasmas and ureaplasmas, and simplifies species classification. Nevertheless, identification and speciation are often accomplished by the use of specific antisera containing antibodies that reflect their different antigenic compositions. The western blot technique is useful in assessing the importance of particular antigens, but gel diffusion and immuno-electrophoresis techniques are important for studying the antigenic structure and the relationships between them. For sero-epidemiological studies and for serological diagnosis, more sensitive tests, such as indirect haemagglutination, the enzyme-linked immunosorbent assay (ELISA) and micro-immunofluorescence, are used.
The way in which different antigens function is exemplified by considering M. pneumoniae. The glucose- and galactose-containing membrane glycolipids of the organism are haptens, which are antigenic only when bound to membrane protein. They induce antibodies that react in complement-fixation, metabolism-inhibition and growth-inhibition tests. Glycolipids of the mycoplasma have a structure fortuitously similar to that in the human brain. The cross-reactivity of the brain glycolipids with antibodies to M. pneumoniae could feasibly account for the neurological manifestations of M. pneumoniae infection (vide infra). Furthermore, the ability of the organisms to alter the I antigen on erythrocytes sufficiently to stimulate anti-I antibodies (cold agglutinins) leads to an autoimmune response and damage to erythrocytes.
M. pneumoniae has two major surface proteins, including the P1 protein involved in attachment, that are recognized by the host; antibody to them is detected in convalescent sera and respiratory secretions. Variation in the P1 protein has led to the recognition of two subtypes of strains, which may vary epidemiologically and in pathogenicity.
Variable membrane lipoproteins of several mycoplasmas form an antigenic variation system that provides a means of escaping from the host immune response; the variations are restricted to a small number of protein antigens and do not appear to alter the total cell protein profiles of the organisms.
Pathogenesis
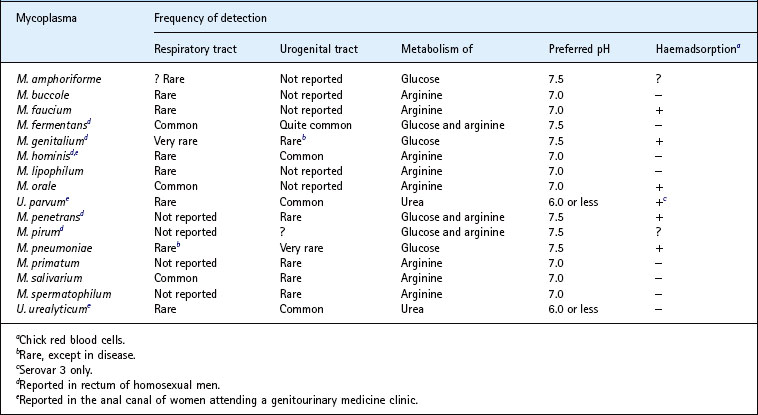
The Mycoplasma and Ureaplasma species that have been isolated from man, either from the respiratory tract (mostly the oropharynx) or from the urogenital tract, are shown Table 41.1. M. pneumoniae, M. genitalium, M. hominis, M. fermentans and U. urealyticum unequivocally cause disease or are strongly associated with disease. M. amphoriforme was recovered from the respiratory tract of patients with antibody deficiency and chronic bronchitis, but its relation to this disease or, indeed, others is not clear.
Table 41.1 Mycoplasmas/ureaplasmas of human origin: anatomical site of detection and some properties
The mechanisms whereby mycoplasmas and ureaplasmas cause disease are multifactorial and include the following:
• Adherence of mycoplasma organisms to host cells, the adhesins of M. pneumoniae and M. genitalium being proteins P1 and MgPa, respectively.
• The ability of some mycoplasmas to invade host cells.
• The production of toxins by M. pneumoniae and some other mycoplasmas.
• The stimulation of pro-inflammatory cytokines.
• Antigenic variation which enables the mycoplasma to evade the protective immune systems of the host.
• Immunological responses of the host, for example, autoimmune ones causing disease, and an exaggerated response to a repeat infection leading to worse disease.
• The ability of mycoplasmas to develop antibiotic resistance culminating sometimes in chronic disease.
Respiratory infections
Mycoplasmal pneumonia
Pneumonias not attributable to any of the common bacterial causes were labelled historically as primary atypical pneumonias. In one variety associated with the development of cold agglutinins, a filterable micro-organism, first called the Eaton agent, was isolated in embryonated eggs. Inhibition by chlortetracycline and gold salts, and cultivation on cell-free medium eventually clinched its mycoplasmal nature. The organism was named M. pneumoniae, and its importance as a cause of respiratory disease was confirmed in numerous studies.
Epidemiology
M. pneumoniae infection occurs world wide. Although endemic in most areas, there is a preponderance of infection in late summer and early autumn in temperate climates; in some countries, such as the UK, epidemic peaks have been observed about every 4 years. Spread is fostered by close contact, for example in a family. Overall, M. pneumoniae may cause only about one-sixth of all cases of pneumonia, but in certain confined populations, such as military recruits, it has been responsible for almost half of the cases of pneumonia. Children are infected more often than adults, and the consequence of infection is also influenced by age. Thus, in school-aged children and teenagers, about a quarter of infections culminate in pneumonia, whereas in young adults fewer than 10% do so. Thereafter, pneumonia is even less frequent, although the severity tends to increase with the age of the patient.
Clinical features
M. pneumoniae infections often have an insidious onset, with malaise, myalgia, sore throat or headache overshadowing and preceding chest symptoms by 1–5 days. Cough, which starts around the third day, is characteristically dry, troublesome and sometimes paroxysmal, and becomes a prominent feature. Physical signs such as râles become apparent, frequently after radiographic evidence of pneumonia. Most often this amounts to patchy opacities, usually of one of the lower or middle lobes. About 20% of patients suffer bilateral pneumonia, but pleurisy and pleural effusions are unusual. The course of the disease is variable; cough, abnormal chest signs and radiographic changes may extend over several weeks and relapse is a feature. A prolonged paroxysmal cough simulating the features of whooping cough may occur in children. Despite these signs and symptoms, most patients are not seriously ill and few warrant admission to hospital. However, very severe infections have been reported in adults, usually in those with immunodeficiency or sickle cell anaemia, although death is rare. Apart from being involved in some exacerbations of chronic bronchitis, there is an unproven suggestion that M. pneumoniae might not only exacerbate asthma, but sometimes be a primary cause of asthma in children.
Disease is limited usually to the respiratory tract. Extra-pulmonary manifestations include:
• Stevens–Johnson syndrome and other rashes
• meningitis or encephalitis (and other neurological sequelae including, rarely, Bell’s palsy)
Haemolytic anaemia with crisis is an auto-immune phenomenon brought about by cold agglutinins (anti-I antibodies). Some of the other complications, such as the neurological ones, may arise in a similar indirect way, although M. pneumoniae has been isolated from cerebrospinal fluid.
Other respiratory infections
M. hominis and ureaplasmas, in particular, have been associated with respiratory disease in the newborn, as described below.
M. hominis has produced sore throats when given orally to adult volunteers; however, it does not seem to cause naturally occurring sore throats in children or adults possibly because the dose encountered is smaller.
M. fermentans has been associated with adult respiratory distress syndrome with or without systemic disease, and with pneumonia in a few children with community-acquired disease. M. genitalium has been isolated, together with M. pneumoniae, from the respiratory secretions of a few adults but its role, if any, in respiratory disease seems small.
Urogenital infections
As listed in Table 41.1, various mycoplasmas and ureaplasmas, most frequently M. hominis and U. urealyticum, have been isolated most frequently from the urogenital tract. Use of the PCR technique has enabled some mycoplasmas, for example M. fermentans and especially M. genitalium, to be detected far more often than would otherwise be the case.
Urogenital infections in men
Although M. hominis may be isolated from about 20% of men with acute non-gonococcal urethritis (NGU), it has not been incriminated as a cause. There is increasing evidence to implicate U. urealyticum, but not U. parvum, in this acute condition; U. urealyticum is the most likely species involved in some chronic cases.
M. genitalium has been strongly implicated worldwide as a cause of about 25% of cases of acute NGU; it also causes chronic NGU, usually in those who have been treated inadequately. In addition, there is preliminary evidence that M. genitalium causes balanoposthitis and it might be involved in some cases of acute epididymitis. However, there is no evidence that this or other mycoplasmas cause acute or chronic prostatitis.
There is evidence that ureaplasmas may occasionally cause acute epididymitis, but it is very doubtful that they have any role in male infertility. The idea that M. genitalium could have a role in infertility is based on the possibility that it might be involved in causing epididymitis and its known ability to adhere to spermatozoa.
Reproductive tract disease in women
M. hominis and, to a lesser extent, ureaplasmas are found in the vagina of women who have bacterial vaginosis in much larger numbers, up to 10 000-fold in the case of M. hominis, than in healthy women. Thus, it is possible that, with various other bacteria, they may contribute to the development of the condition. However, it seems very unlikely that M. hominis causes disease if present in small numbers. M. genitalium has been associated strongly with cervicitis, despite the criteria used to define the condition being variable.
M. hominis has been isolated from the endometrium and fallopian tubes of about 10% of women with laparoscopically confirmed pelvic inflammatory disease (PID), together with a specific antibody response. This suggests a causal role, but the significance of M. hominis is difficult to judge with confidence as several other micro-organisms, including Chlamydia trachomatis (Ch. 39), may be found at the same time. Ureaplasmas have also been isolated directly from affected fallopian tubes, but the absence of antibody responses and failure to produce salpingitis in subhuman primates make them an even less likely cause of disease at this site.
The pathogenic potential of M. genitalium in PID seems greater; it has been associated significantly with endometritis, and antibody responses to the mycoplasma in some patients with the disease and produces endometritis, parametritis and salpingitis in subhuman primates. However, reports of detection of M. genitalium in laparoscopically derived fallopian tube specimens by PCR technology are few and this decisive approach needs to be taken whenever the opportunity arises.
The part that M. hominis is likely to play in infertility in women as a result of tubal damage is small. In contrast, serology has shown a strong association between M. genitalium and tubal factor infertility.
Disease associated with pregnancy and the newborn
M. hominis and ureaplasmas have been isolated from the amniotic fluid of women with severe chorio-amnionitis who had pre-term labour. Similarly, ureaplasmas have been isolated from spontaneously aborted fetuses and stillborn or premature infants more frequently than from induced abortions or normal full-term infants.
Isolation of ureaplasmas from the internal organs of aborted fetuses, together with some serological responses and an apparently diminished occurrence following antibiotic therapy, suggest that these organisms may have a role in abortion. However, bacterial vaginosis is strongly associated with pre-term labour and late miscarriage and, as M. hominis and ureaplasmas are part of the extensive microbial flora of bacterial vaginosis, they may act with a multitude of other micro-organisms and not independently. There is, so far, inadequate evidence to associate M. genitalium with pre-term outcome of pregnancy.
The same considerations relate to the role of other genital mycoplasmas, particularly ureaplasmas, in causing low birth-weight in otherwise normal full-term infants. Bacterial vaginosis has been largely ignored in defining this association. A study in which women given erythromycin in the third trimester delivered larger babies than those given a placebo has not been confirmed in later trials. However, in infants of less than 1000 g with a ureaplasmal respiratory tract infection acquired in utero or at the time of delivery, congenital pneumonia and the likelihood of death or chronic lung disease is about double that in uninfected infants of similar or greater birthweight. Inflammatory cytokines have been found in tracheal aspirates from these very low birthweight infants with respiratory ureaplasmal infection. There is considerable support for this scenario but no evidence of a reduction in the incidence of chronic disease has yet been found when preterm infants have been treated with erythromycin. Although there is uncertainty surrounding this particular issue, premature infants are prone to meningitis, with or without further neurological damage, as a result of M. hominis or ureaplasmas gaining access to the cerebrospinal fluid within the first few days of life.
After an abortion or even a normal delivery, M. hominis has been isolated, apparently in pure culture, from the blood of about 10% of febrile women, about half of them developing an antibody response, but not from afebrile women who aborted or from healthy pregnant women. Observations of this kind have also been made for ureaplasmas and it seems that both micro-organisms induce fever, possibly by causing endometritis.
Urinary infection and calculi
M. hominis has been isolated from the upper urinary tract of patients with acute pyelonephritis and may cause a small proportion (possibly about 5%) of such cases. Ureaplasmas do not seem to be involved in pyelonephritis. However, the fact that they produce urease, induce crystallization of struvite and calcium phosphates in urine in vitro, and calculi experimentally in animal models, raises the question of whether they cause calculi in the human urinary tract. In support of this, ureaplasmas are more often found in the urine and calculi of patients with infective-type stones than in those with metabolic stones.
Joint infections
Evidence that ureaplasmas are involved in the aetiology of sexually acquired reactive arthritis is based on synovial fluid mononuclear cell proliferation in response to specific ureaplasmal antigens. Ureaplasmas also have a role in suppurative arthritis in hypogammaglobulinaemic patients (vide infra). M. genitalium has been reported in the joints of patients with Reiter’s disease or rheumatoid arthritis, but the significance of this remains unclear. M. fermentans has been found by a PCR assay in the joints of about 20% of patients with rheumatoid arthritis and in those of patients with other chronic inflammatory rheumatic disorders, but not in the joints of patients with non-inflammatory arthritides. However, whether this mycoplasma is simply attracted to an inflammatory site and is behaving as an innocent bystander remains to be determined.
Infections in immunocompromised patients
M. pneumoniae pneumonia in immunodeficient patients may be severe and the organisms may persist for many months in the respiratory tract of hypogammaglobulinaemic patients, despite apparently adequate antibiotic treatment. A few hypogammaglobulinaemic patients develop suppurative arthritis and mycoplasmas are responsible for at least two-fifths of the cases. The mycoplasmas mainly involved are M. pneumoniae, M. salivarium (usually regarded as non-pathogenic), M. hominis and, particularly, ureaplasmas. In some cases involving ureaplasmas, the arthritis has been associated with subcutaneous abscesses, persistent urethritis and chronic cystitis. Although sometimes responding to antibiotic treatment, these organisms and disease may persist for many months despite concomitant use of anti-inflammatory and γ-globulin replacement therapy.
Haematogenous spread of M. hominis leading to septic arthritis, surgical wound infections and peritonitis seems to occur more often after organ transplantation and in other patients on immunosuppressive therapy. Particularly common are sternal wound infections in heart and lung transplant patients.
The suggestion that M. fermentans infection of the peripheral blood monocytes of human immunodeficiency virus (HIV)-positive subjects may lead to a more rapid development of AIDS now seems highly unlikely. Similarly, nothing has emerged to support the notion that M. penetrans is associated with HIV positivity or with Kaposi’s sarcoma.
Laboratory diagnosis
Respiratory infections
Because the clinical manifestations of M. pneumoniae-induced disease are often insufficiently distinct for definitive diagnosis, laboratory help is required. Isolation is not often attempted because of insensitivity and the time required. In the past reliance was often placed on serology, notably the complement fixation test. However, specificity is relative since cross-reactivity with M. genitalium is sometimes seen. A four-fold or greater rise in antibody titre, with a peak at about 3–4 weeks, is indicative of a recent M. pneumoniae infection, but because paired sera are not always available or because of the delay in acquiring a second serum, a single antibody titre of 64–128 has been used to institute therapy in a suggestive clinical setting.
Serology and culture have largely been superseded by PCR technology, which is specific, sensitive and rapid. If an M. pneumoniae isolate is required, a sensible approach is to test specimens by both the PCR assay and culture, and to continue the culture procedure only for those specimens that prove to be PCR positive.
The cold agglutinin test is non-specific and unreliable. Of a gamut of other tests, the enzyme immunoassay is sensitive and specific, and there are formats for testing paired sera or a single serum. The ImmunoCard® test (Meridian Diagnostics) for IgM is simple and rapid and has proved reliable, especially in children. The Remel enzyme immunoassay detects IgM and IgG simultaneously and is sensitive and specific.
Attempted isolation of M. pneumoniae (and M. fermentans) requires the use of mycoplasmal SP4 broth medium supplemented with penicillin and glucose, and phenol red as a pH indicator. After inoculation with sputum, throat washing, pharyngeal swab or other specimen, the medium is incubated at 37°C, and a colour change (red to yellow), which may take up to 3 weeks or longer, indicates fermentation of glucose by multiplying organisms. The broth is then subcultured on agar medium to await the development of colonies, which are identified specifically by immunofluorescence with a specific antiserum or by other serological methods.
Diagnostic procedures for other mycoplasmas that might be found in the respiratory tract are considered below.
Urogenital infections
Culture still has a place in the detection of some genital mycoplasmas and ureaplasmas. Material from urethral, cervical or vaginal swabs, or a centrifuged deposit from urine, is added to separate vials of liquid mycoplasmal medium containing phenol red and 0.1% glucose, arginine or urea. M. genitalium metabolizes glucose and changes the colour of the medium from red to yellow. M. fermentans also metabolizes glucose but also converts arginine to ammonia, as do M. hominis and M. primatum. Ammonia is also produced when ureaplasmal urease breaks down urea and the colour changes from yellow to red.
M. hominis multiplies in most routine blood culture media if gelatin (1% w/v) is added to overcome the inhibitory effect of sodium polyanethol sulphonate, included as an anticoagulant.
Kits designed to isolate and identify M. hominis and ureaplasmas, and to provide antimicrobial susceptibility profiles, are available commercially. They are of particular value if the need to detect these micro-organisms arises infrequently. Phylogeny-based rapid identification of urogenital mycoplasmas and ureaplasmas, based on the amplification of a part of the 16S rRNA gene by PCR, has also been described.
DNA primers specific for practically all Mycoplasma and Ureaplasma species, including M. fermentans, M. genitalium, U. urealyticum and U. parvum, have been developed and used for amplification by PCR. This technique is much more sensitive than culture for detecting the two former mycoplasmas and is the only reliable way of determining their presence in clinical specimens.
Genital mycoplasmal infections stimulate antibody responses, but the various techniques to detect them are rarely used diagnostically.
Treatment
The in-vitro sensitivity of several mycoplasmas and ureaplasmas to various antibiotics used in treatment is shown in Table 41.2. These micro-organisms are completely resistant to β-lactam or glycopeptide antibiotics.
Mycoplasmal pneumonia
M. pneumoniae is sensitive to erythromycin and moderately sensitive to the tetracyclines in vitro and these antibiotics have been used widely in clinical practice. Though they have sometimes proved less effective for treating pneumonia than in planned trials, it is still worthwhile administering a macrolide or a tetracycline to adults, and erythromycin to children and pregnant women.
Newer macrolides, such as clarithromycin and azithromycin, and the newer fluoroquinolones, are highly active in vitro and certainly have a place in treatment, although macrolide resistance may sometimes be an issue. The fluoroquinolones seem to have a cidal effect, an important attribute which may be useful as tetracyclines and macrolides only inhibit growth. Failure to kill the organisms, together with the fact that they may become intracellular, probably explains persistence in the respiratory tract long after clinical recovery, as well as clinical relapse in some patients. Furthermore, a functioning immune system has been shown to be important in eradication, some hypogammaglobulinaemic patients, for example, experiencing persistence of the organisms for months or years. A veterinary pleuromutilin antibiotic (Econor®) has been used with some success to treat mycoplasmal respiratory infections in immunocompromised subjects.
Antibiotic treatment of M. pneumoniae or other mycoplasma-induced infection should start as soon as possible, based on clinical suspicion, and a course of at least two weeks is justified. The wait for laboratory confirmation is now less pressing in view of the rapidity of PCR technology.
Urogenital infections
Treatment must take into account the fact that several different micro-organisms may be involved and that a precise microbiological diagnosis may not be attainable. Thus, patients with NGU should receive an antibiotic that is active against C. trachomatis, ureaplasmas and M. genitalium. Azithromycin, which is used increasingly for chlamydial infections, is also active against a wide range of mycoplasmas, including M. genitalium and, to a lesser extent, ureaplasmas. This may be preferable to a tetracycline, because M. genitalium is less sensitive to tetracyclines and at least 10% of ureaplasmas are resistant. Some M. genitalium strains are, or have become, resistant to azithromycin and in this circumstance moxifloxicin has been used successfully.
A broad-spectrum antibiotic should also be included for the treatment of PID to cover C. trachomatis, M. genitalium and M. hominis as well as various anaerobic bacteria. As about 20% of M. hominis strains are resistant to tetracyclines, other antibiotics such as clindamycin or fluoroquinolones may need to be considered.
Fever following abortion or childbirth often settles within a few days, but if it does not therapy with a broad-spectrum antibiotic should be started, keeping the above comments in mind.
Mycoplasmas and cell cultures
It is rare for primary cell cultures to become infected with mycoplasmas, but continuous cell lines do so frequently. Various species may be responsible. The effects of contamination include those caused by mycoplasmal enzymes and toxins, and those resulting from metabolism of cell culture media components or from changes in pH. Despite the presence of up to 108 organisms per millilitre of culture fluid, there may be little effect on viral propagation, although it may decrease the yield. Occasionally, the yield may be increased as, for example, with vaccinia virus in M. hominis-infected cells.
Mycoplasmal culture methods and an indicator cell system with staining (e.g. Hoechst DNA dye) are used conventionally to detect contamination, but PCR methods have much greater sensitivity. Numerous procedures have been described to eliminate mycoplasmas from cell cultures, but none is consistently successful. Whenever possible it is easier to discard the cultures, replace them with mycoplasma-free cells and adhere to simple guidelines to prevent contamination. If it is imperative to save cells, treatment with an antibiotic that is likely to have mycoplasmacidal activity, such as a fluoroquinolone, identification of the contaminant and use of a specific antiserum, or a combination of these methods, is most likely to be successful in eradication.
Blanchard A, Browning G. Mycoplasmas: Molecular Biology, Pathogenicity and Strategies for Control. Wymondham, UK: Horizon Scientific Press, 2005.
Furr PM, Taylor-Robinson D, Webster ADB. Mycoplasmas and ureaplasmas in patients with hypogammaglobulinaemia and their role in arthritis: microbiological observations over 20 years. Annals of the Rheumatic Diseases. 1994;53:183–187.
Maniloff J, McElhaney RN, Finch LR, Baseman JB. Mycoplasmas. Molecular Biology and Pathogenesis. Washington, DC: American Society for Microbiology, 1992.
Taylor-Robinson D. Mycoplasmas and their role in human respiratory tract disease. In: Myint S, Taylor-Robinson D. Viral and Other Infections of the Human Respiratory Tract. London: Chapman and Hall; 1996:319–339.
Taylor-Robinson D. Mycoplasma genitalium – an update. International Journal of STD & AIDS. 2002;13:145–151.
Taylor-Robinson D, Bebear C. Antibiotic susceptibilities of mycoplasmas and treatment of mycoplasmal infections. Journal of Antimicrobial Chemotherapy. 1997;40:622–630.
Taylor-Robinson D, Gilroy CB, Jensen JS. The biology of Mycoplasma genitalium. Venereology. 2000;13:119–127.
Taylor-Robinson D, Jensen JS. Genital mycoplasmas. In: Morse SA, Ballard RC, Holmes KK, Moreland AA. Atlas of Sexually Transmitted Diseases and AIDS. ed 4. London: Mosby; 2011:64–75.
Taylor-Robinson D, Jensen JS. Mycoplasma genitalium: from chrysalis to multicolored butterfly. Clinical Microbiology Reviews. 2011;24:498–514.
Totten PA, Taylor-Robinson D, Jensen JS. Genital mycoplasmas. In: Holmes KK, Sparling PF, Stamm WE, et al. Sexually Transmitted Diseases. ed 4. New York: McGraw Hill; 2008:709–736.
Tully JG, Razin S, eds. Molecular and Diagnostic Procedures in Mycoplasmology. Diagnostic Procedures, Vol. 2. London: Academic Press, 1996.
Waites KB, Taylor-Robinson D. Mycoplasma and Ureaplasma, ed 10. Murray PR, Baron EJ, Jorgensen JH, et al, eds. Manual of Clinical Microbiology, Vol. 1. Washington, DC: American Society for Microbiology. 2011:970–985.