18 Mycobacterium
Tuberculosis; leprosy
Key points
• The mycobacteria are characterized by thick lipid-rich cell walls and the ‘acid-fast’ staining property.
• Most of the 130 or so named mycobacterial species are environmental saprophytes, although some occasionally cause disease, notably in immunosuppressed persons. M. tuberculosis and M. leprae are obligate pathogens.
• Tuberculosis is a chronic intracellular infection characterized by granuloma formation. Most individuals (about 90%) do not develop symptomatic disease and some remain latently infected.
• The lung is the usual site of initial infection and disease, but non-pulmonary forms occur. Post-primary tuberculosis is characterized by gross tissue necrosis leading to pulmonary cavity formation and aerogenous infectivity
• One-third of the world’s population has been infected by the tubercle bacillus and about 100 million individuals are newly infected annually.
• Diagnosis is made by clinical and radiological examination, tuberculin testing and detection of the tubercle bacillus by acid-fast staining, culture and nucleic acid-based techniques. Therapy is based on a regimen of several drugs, usually for a period of 6 months. BCG is an attenuated live vaccine; its protective efficacy varies greatly between geographical regions and its overall impact on tuberculosis control is limited.
• Multi- and extensively drug resistant tuberculosis is encountered worldwide and requires the use of an extended range of anti-tuberculosis agents for its treatment.
• M. leprae has never been cultivated in vitro. In leprosy, the nerves and skin are the principal sites of disease, resulting in deformities and visible lesions.
• Transmission is probably by the aerogenous route rather than by touch.
• Tuberculoid leprosy is characterized by excessive granuloma formation in the presence of a very small (paucibacillary) bacterial load, whereas lepromatous leprosy is characterized by a huge (multibacillary) bacterial load and little or no immune reactivity.
• Diagnosis is by clinical examination, microscopic detection or PCR of bacilli in skin or nasal smears and biopsies. Multidrug treatment regimens are highly effective and BCG vaccination appears to offer some degree of protection.
The name of the genus Mycobacterium (fungus-bacterium) is an allusion to the mould-like pellicles formed when members of this genus are grown in liquid media. This hydrophobic property is due to their possession of thick, complex, lipid-rich, waxy cell walls. A further important characteristic, also due to their waxy cell walls, is acid-fastness, or resistance to decolourization by a dilute mineral acid (or acid with alcohol) after staining with hot carbol fuchsin or other arylmethane dyes.
There are over 130 named species of mycobacteria, the great majority of which are environmental saprophytes, although some cause opportunist disease in humans and animals (see Ch. 19). There are, however, two human mycobacterial diseases caused by obligate pathogens – tuberculosis and leprosy. The former is caused by members of the Mycobacterium tuberculosis complex, strictly speaking one species, and the latter by the leprosy bacillus, Mycobacterium leprae, which has never convincingly been grown in vitro.
Mycobacterium tuberculosis complex
The ‘M. tuberculosis complex’ refers to a group of genetically very closely related variants of what is strictly speaking a single species. All cause tuberculosis, a chronic granulomatous disease affecting humans and many other mammals. The complex includes:
• M. tuberculosis, the predominant cause of human tuberculosis.
• M. bovis, the principal cause of tuberculosis in cattle and many other mammals including humans.
• M. africanum, which appears to be intermediate in form between the human and bovine types. It causes human tuberculosis and is found mainly in equatorial Africa. Type 1 is more common in West Africa and has several features in common with M. bovis; type 2 is mainly of east African origin and more closely resembles M. tuberculosis.
• M. caprae, isolated from goats and the cause of a few cases of tuberculosis in veterinary surgeons.
• M. pinnipedi, an uncommon pathogen of seals and a very rare cause of tuberculosis in those occupationally exposed to seals.
• M. microti, a rarely encountered pathogen of voles and other small mammals, but which is attenuated in humans.
• M. canetti, a very rare and genetically primitive member of the complex.
Description
The entire genomes of M. tuberculosis and M. bovis have been sequenced. On the basis of sequencing, there is evidence that the members of the M. tuberculosis complex (tubercle bacilli) listed above devolved from a common progenitor by the successive loss of short chromosomal segments known as Regions of Difference (RD). The variants closest to the progenitor form are M. canetti and certain strains common in South India. Contrary to earlier opinion, M. bovis appears to have devolved from M. tuberculosis. A number of families or lineages are distinguishable within each species, notably M. tuberculosis, and one particular family, the Beijing or W/Beijing family, is spreading worldwide, is of high virulence and readily mutates to drug resistance while retaining high virulence.
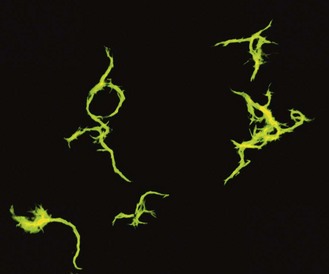
Tubercle bacilli are non-motile, non-sporing, non-capsulate, straight or slightly curved rods about 3 × 0.3 µm in size. In sputum and other clinical specimens they may occur singly or in small clumps, and in liquid cultures they often grow as twisted rope-like colonies termed serpentine cords (Fig. 18.1). They are able to grow on a wide range of enriched culture media, but Löwenstein–Jensen (LJ) medium is the most widely used in clinical practice. This is an egg–glycerol-based medium to which malachite green dye is added to inhibit the growth of some contaminating bacteria and to provide a contrasting colour against which colonies of mycobacteria are easily seen. Strains of M. bovis grow poorly, or not at all, on standard LJ medium but grow much better on media containing sodium pyruvate in place of glycerol. Agar-based media or broths enriched with bovine serum albumin are also used, particularly in automated culture systems.
On subculture, human tubercle bacilli usually produce visible growth on LJ medium in 2–4 weeks, although on primary isolation from clinical material colonies may take up to 8 weeks to appear. Colonies are of an off-white (buff) colour and (except for the very rarely encountered M. canetti which has smooth colonies) usually have a dry breadcrumb-like appearance. Growth is characteristically heaped up and luxuriant or ‘eugonic’, in contrast to the small, flat ‘dysgonic’ colonies of M. bovis.
The optimal growth temperature of tubercle bacilli is 35–37°C, but they fail to grow at 25°C or 41°C. Most other mycobacteria grow at one or other, or both, of these temperatures.
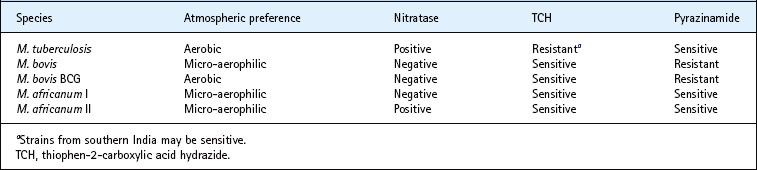
All mycobacteria are obligate aerobes, but M. bovis grows better in conditions of reduced oxygen tension. Thus, when incorporated in soft agar media, M. tuberculosis grows on the surface whereas M. bovis grows as a band a few millimetres below the surface. This provides a useful differentiating test. Various other differential characteristics of human tubercle bacilli are shown in Table 18.1.
Table 18.1 Some differential characteristics of the principal tubercle bacilli causing human disease
Tubercle bacilli survive in milk and other organic materials and on pasture land so long as they are not exposed to ultraviolet light, to which they are very sensitive. They are also heat sensitive and are destroyed by pasteurization. Mycobacteria are susceptible to alcohol, formaldehyde, glutaraldehyde and, to a lesser extent, hypochlorites and phenolic disinfectants. They are considerably more resistant than other bacteria to acids, alkalis and quaternary ammonium compounds.
Pathogenesis
The tubercle bacillus owes its virulence to its ability to survive within the macrophage rather than to the production of a toxic substance. The mechanism of virulence is poorly understood and is almost certainly multifactorial, resulting in inappropriate patterns of immune reactivity. The immune response to the bacillus is of the cell-mediated type, which, if mediated by T helper (TH) cells and associated type 1 cytokines, leads to protective immunity, although the presence of TH2 cells and associated cytokines facilitates tissue-destroying hypersensitivity reactions and progression of the disease process. The nature of the immune responses following infection changes with time, so that human tuberculosis is divisible into primary and post-primary forms with quite different pathological features.
Primary tuberculosis
The site of the initial infection is usually the lung, following the inhalation of bacilli. These bacilli are engulfed by alveolar macrophages in which they replicate to form the initial lesion. Some bacilli are carried in phagocytic cells to the hilar lymph nodes where additional foci of infection develop. The initial focus of infection together with the enlarged hilar lymph nodes forms the primary complex. In addition, bacilli are seeded by further lymphatic and haematogenous dissemination in many organs and tissues, including other parts of the lung. When the bacilli enter the mouth, as when drinking milk containing M. bovis, the primary complexes involve the tonsil and cervical nodes (scrofula; Fig. 18.2) or the intestine, often the ileocaecal region, and the mesenteric lymph nodes. Likewise, the primary focus may be in the skin, with involvement of the regional lymph nodes. This uncommon type of tuberculosis, formerly termed prosector’s wart, is principally an occupational disease of anatomists, pathologists and meat handlers.
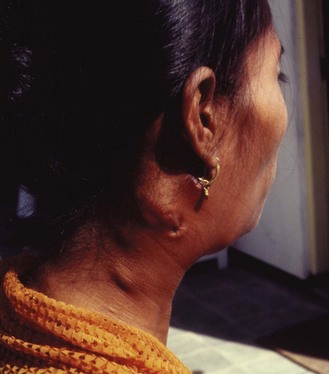
Fig. 18.2 Tuberculous cervical lymphadenitis (scrofula) with sinus formation in an Indonesian woman.
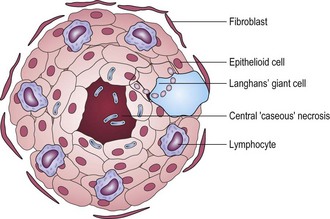
During infection, antigens of M. tuberculosis are processed by antigen-presenting cells, activated by bacterial components termed adjuvants, and presented to antigen-specific T lymphocytes which undergo clonal proliferation. The activated T cells release cytokines, notably interferon-γ, which, together with calcitriol, activate macrophages (Fig. 18.3) and cause them to form a compact cluster, or granuloma, around the foci of infection (Fig. 18.4). These activated macrophages are termed epithelioid cells because of their microscopical resemblance to epithelial cells. Some of them fuse to form multinucleate giant cells. The centre of the granuloma contains a mixture of necrotic tissue and dead macrophages, and, because of its cheese-like appearance and consistency, is referred to as caseation.
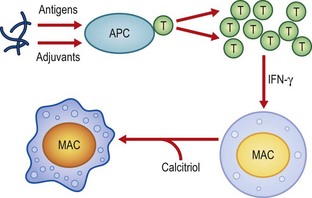
Fig. 18.3 Antigens of M. tuberculosis are, after priming by adjuvants, processed by the antigen-presenting cell (APC) and presented to T helper lymphocytes (T), which are activated and proliferate to form a clone. T cell-produced interferon (IFN)-γ and calcitriol activate the resting macrophages (MAC).
Activated human macrophages inhibit the replication of the tubercle bacilli, but their ability to kill ingested bacilli is limited. Being metabolically very active, the macrophages in the granuloma consume oxygen, and the resulting anoxia and acidosis in the centre of the lesion probably kills most of the bacilli. Granuloma formation is usually sufficient to limit the primary infection: the lesions become quiescent and surrounding fibroblasts produce dense scar tissue, which may become calcified. Programmed cell death (apoptosis) of bacteria-laden macrophages by cytotoxic T cells and natural killer (NK) cells contributes to protective immunity by generating a metabolic burst that kills tubercle bacilli.
In a minority of cases one of the infective foci progresses and gives rise to the serious manifestations of primary disease, including progressive local lesions (particularly in infants; Table 18.2), meningitis, pleurisy and disease of the kidneys, spine (Pott’s disease) and other bones and joints. If a focus ruptures into a blood vessel, bacilli are disseminated throughout the body with the formation of numerous granulomata. This, from the millet seed-like appearance of the lesions, is known as miliary tuberculosis.
Table 18.2 Stages of primary tuberculosis in childhood
| Stage | Time (from onset) | Characteristics |
|---|---|---|
| 1 | 3–8 weeks | Primary complex develops and tuberculin conversion occurs |
| 2 | 2–6 months | Progressive healing of primary complex Possibility of pleural effusion |
| 3 | 6–12 months | Possibility of miliary or meningeal tuberculosis |
| 4 | 1–3 years | Possibility of bone or joint tuberculosis |
| 5 | 3–5 years or more | Possibility of genito-urinary or chronic skin tuberculosis |
Adapted from Miller FJW 1982 Tuberculosis in Children. Churchill Livingstone, Edinburgh.
Tuberculin reactivity
About 6–8 weeks after the initial infection, the phenomenon of tuberculin conversion occurs. This altered reactivity was discovered by Robert Koch while attempting to develop a remedy for tuberculosis based on old tuberculin – a heat-concentrated filtrate of a broth in which tubercle bacilli had been grown. Although Koch’s tuberculin proved unsuccessful as a therapeutic agent, it formed the basis of the widely used tuberculin test (see below).
Latent tuberculosis
In most infected individuals, the primary infection resolves but some residual tubercle bacilli enter a poorly understood stage of latency or dormancy. It was previously assumed that ‘once infected, always infected’, but as the risk of developing active tuberculosis after initial infection is highest within the first and second year, around 1% and 0.3% respectively, and much lower in subsequent years, it seems likely that tubercle bacilli are totally eliminated in many infected individuals. This is also suggested by the finding that the risk of a HIV-infected person developing tuberculosis is much higher if they are newly infected by tubercle bacilli than if they had been infected before acquiring HIV. The use of molecular fingerprinting has established that exogenous reinfection occurs far more readily than was previously thought. Long term latency may, however, occur as small numbers of reactivation tuberculosis due to M. bovis are encountered for several decades after effective eradication of cattle tuberculosis in a given region.
Post-primary tuberculosis
Reactivation of dormant foci of tubercle bacilli or exogenous reinfection leads to post-primary tuberculosis, which differs in several respects from primary disease (Table 18.3). For unknown reasons, reactivation or reinfection tuberculosis tends to develop in the upper lobes of the lungs. The same process of granuloma formation occurs, but the necrotic element of the reaction causes extensive tissue destruction and the formation of large areas of caseation termed tuberculomas. Proteases liberated by activated macrophages soften and liquefy the caseous material, and an excess of tumour necrosis factor and other immunological mediators causes the wasting and fevers characteristic of the disease.
Table 18.3 Main differences between primary and post-primary tuberculosis in non-immunocompromised patients
| Characteristic | Primary | Post-primary |
|---|---|---|
| Local lesion | Small | Large |
| Lymphatic involvement | Yes | Minimal |
| Cavity formation | Rare | Frequent |
| Tuberculin reactivity | Negative (initially) | Positive |
| Infectivitya | Uncommon | Usual |
| Site | Any part of lung | Apical region |
| Local spread | Uncommon | Frequent |
The interior of the tuberculoma is acidic and anoxic, and contains few viable tubercle bacilli. Eventually, however, the expanding lesion erodes through the wall of a bronchus, the liquefied contents are discharged and a well aerated cavity is formed. The atmosphere of the lung, with a high carbon dioxide level, is ideal for supporting the growth of the bacilli, and huge numbers of these are found in the cavity walls. For this reason, closure of the cavities by collapsing the lung, either by artificial pneumothorax or by excising large portions of the chest wall, was a standard treatment for tuberculosis in the pre-chemotherapy era.
Once the cavity is formed, large numbers of bacilli gain access to the sputum, and the patient becomes an open or infectious case. This is a good example of the transmissibility of a pathogen being dependent upon the host’s immune response to infection. Surprisingly, about 20% of cases of open cavitating tuberculosis in the pre-therapy era resolved without treatment.
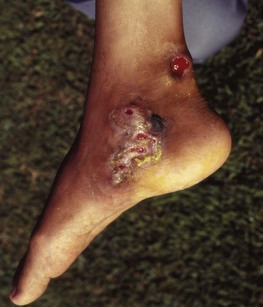
In post-primary tuberculosis, dissemination of bacilli to lymph nodes and other organs is unusual. Instead, spread of infection occurs through the bronchial tree so that secondary lesions develop in the lower lobes of the lung and, occasionally, in the trachea, larynx and mouth. Bacilli in swallowed sputum cause intestinal lesions. Secondary lesions may also develop in the bladder and epididymis in cases of renal tuberculosis. Post-primary cutaneous tuberculosis (lupus vulgaris) usually affects the face and neck. Untreated, it is a chronic condition leading to gross scarring and deformity. Most cases of lupus vulgaris were caused by M. bovis and this condition is now rarely seen in developed countries where transmission of disease from cattle rarely if ever occurs. Some cases of cutaneous tuberculosis are secondary to sinus formation between tuberculous lymph nodes and the skin (scrofuloderma) and other structures including bones and joints (Fig. 18.5).
Immunocompromised individuals
People with congenital or acquired causes of immunosuppression, including the very young, the elderly and recipients of organ transplants, are at a much higher risk of developing tuberculosis due to reactivation of latent disease or following infection or reinfection. HIV infection has emerged as the major predisposing factor to active tuberculosis worldwide (see below). Tuberculosis acts synergistically with HIV to lower the patient’s immunity and it is a defining condition for the acquired immune deficiency syndrome (AIDS). As a result, even when tuberculosis is treated effectively in HIV-positive patients, the mortality rate from other AIDS-related conditions is high, with many dying within 2 years. Cavity formation is unusual in the more profoundly immunocompromised patients, emphasizing the importance of the immune response in this pathological process. Instead, diffuse infiltrates develop in any part of the lung. In contrast to post-primary disease in non-immunocompromised individuals, lymphatic and haematogenous dissemination are common. Sometimes there are numerous minute lesions teeming with tubercle bacilli throughout the body – a rapidly fatal condition termed cryptic disseminated tuberculosis. The interval between infection and development of disease is considerably shortened in immunocompromised persons.
The tuberculin test and other immunological tests
Although Robert Koch’s attempts to use old tuberculin as a remedy for tuberculosis failed, an Austrian physician, Clemens von Pirquet, used the Koch phenomenon as an indication of bacterial ‘allergy’ resulting from previous infection. Individuals with active tuberculosis were usually tuberculin positive, but many of those with disseminated and rapidly progressive disease were negative. This led to the widespread but erroneous belief that tuberculin reactivity is an indicator of immunity to tuberculosis.
Old tuberculin caused non-specific reactions and has been replaced by purified protein derivative (PPD). This is given by:
• intracutaneous injection (Mantoux method)
• a spring-loaded gun which fires six prongs into the skin through a drop of PPD (Heaf method)
• single-test disposable devices with PPD dried on to prongs (tine tests).
The biological activity of tuberculin is compared with international standards and activity expressed in international units (IU). In the UK, solutions of PPD for Mantoux testing are supplied as dilutions of 1 in 10 000, 1 in 1000 and 1 in 100, which correspond to 1, 10 and 100 IU in the injected dose of 0.1 mL. The standard test dose is 10 IU, but those suspected of having tuberculosis, and who are therefore likely to react strongly, may first be tested with 1 IU. Undiluted PPD is supplied for use with the Heaf gun.
The tuberculin test is widely used as a diagnostic test, although its usefulness is limited by its failure to distinguish active disease from quiescent infections and past Bacille Calmette–Guérin (BCG) vaccination and, in some regions, sensitization by environmental mycobacteria. Numerous attempts have therefore been made to develop alternative immunological tests for the disease, though with many setbacks. One promising approach, though not without problems of specificity, is the detection of interferon-γ-producing peripheral blood T cells that respond to antigens present in virulent tubercle bacilli but not environmental mycobacteria or BCG.
Laboratory diagnosis
The definitive diagnosis of tuberculosis is based on detection of the causative organism in clinical specimens by microscopy, cultural techniques or the polymerase chain reaction (PCR) and its various derivatives.
Specimens
The most usual specimen for diagnosis of pulmonary tuberculosis is sputum but, if none is produced, bronchial washings, brushings or biopsies and early-morning gastric aspirates (to harvest any bacilli swallowed overnight) may be examined. Tissue biopsies are homogenized by grinding for microscopy and culture. Cerebrospinal fluid, pleural fluid, urine and other fluids are centrifuged and the deposits examined.
Microscopy
Use is made of the acid-fast property of mycobacteria to detect them in sputum and other clinical material. In the Ziehl–Neelsen (ZN) staining technique, heat-fixed smears of the specimens are flooded with a solution of carbol fuchsin (a mixture of basic fuchsin and phenol) and heated until steam rises. After washing with water, the slide is flooded with a dilute mineral acid (e.g. 3% hydrochloric acid) and, after further washing, a green or blue counterstain is applied. Red bacilli are seen against the contrasting background colour. In some methods, the acid is diluted in 95% ethanol rather than water. This gives a cleaner background but, contrary to a common belief, it does not enable tubercle bacilli to be distinguished from other mycobacteria. Fluorescence microscopy, based on the same principle of acid-fastness, is increasingly used and is much less tiring for the microscopist. Modifications of the various staining techniques are used to examine tissue sections.
Cultural methods
As sputum and certain other specimens frequently contain many bacteria and fungi that would rapidly overgrow any mycobacteria on the culture media, these must be destroyed. Decontamination methods make use of the relatively high resistance of mycobacteria to acids, alkalis and certain disinfectants. In the widely used Petroff method, sputum is mixed well with 4% sodium hydroxide for 15–30 min, neutralized with potassium dihydrogen orthophosphate and centrifuged. The deposit is used to inoculate LJ or similar media. Specimens such as cerebrospinal fluid and tissue biopsies, which are unlikely to be contaminated, are inoculated directly on to culture media. As an alternative to chemical decontamination, mixtures of antibiotics that kill fungi and all bacteria other than mycobacteria may be added to the culture media. These are used principally in the automated culture systems described below.
Inoculated media are incubated at 35–37°C and inspected weekly for at least 8 weeks. Cultures of material from skin lesions should also be incubated at 33°C. Any bacterial growth is stained by the ZN method and, if acid-fast, subcultured for further identification.
A more rapid bacteriological diagnosis is achievable by use of commercially available automated systems. Systems that detect colour changes in dyes induced by the release of carbon dioxide, or the unquenching of fluorescent dyes on the consumption of oxygen by metabolizing bacilli, have replaced the earlier radiometric method.
The first step in identification is to determine whether an isolate is a member of the M. tuberculosis complex. These organisms:
• do not produce yellow pigment
• fail to grow at 25°C and 41°C
• do not grow on egg media containing p-nitrobenzoic acid (500 mg/L).
Strains differing in any of these properties belong to other species.
Nucleic acid technology
Amplification of specific nucleic acid sequences in specimens is achievable by PCR and related techniques. Initial problems of low sensitivity, ‘false-positive’ reactions and cross-contamination have been largely overcome by the introduction of closed-system, isothermal techniques for amplification of species-specific 16S ribosomal ribonucleic acid (RNA). User-friendly automated systems with high sensitivity and specificity are commercially available.
Molecular techniques for fingerprinting members of the M. tuberculosis complex for epidemiological purposes have been developed. An early method, restriction length polymorphism (RFLP) analysis, was based on the determination of the numbers and position on electrophoretic gels and of the insertion sequence IS6110, 1 to 20 copies of which are present in most strains. Preferred methods now include spoligotyping, based on the detection of spacer oligonucleotides, short DNA sequences found around the sites of the insertion sequences, and analysis of genetic units termed mycobacterial interspersed repetitive units (MIRU), each of which each has sequential, or tandem, repeats which vary in number from species to species, proving a highly discriminative typing system for strains with few or no IS6110 insertion sequences.
Drug susceptibility testing
Several methods have been described.
• Assessment of growth inhibition on solid media containing various dilutions of the drug, in comparison with test strains. As the methods depend on observation of visible growth, results are not available until several weeks after isolation of the organism.
• Automated systems, based on the activation or quenching of dyes by metabolic activity of the bacilli, provide results more quickly.
• Nucleic acid technology, which is even more rapid, though relatively expensive. Reverse hybridization assays for the detection of resistance to rifampicin and isoniazid are commercially available and ‘in-house’ assay methods for pyrazinamide and ethambutol have been described.
Treatment
The antituberculosis drugs are divisible into three groups (Table 18.4):
1. Bactericidal drugs that effectively sterilize tuberculous lesions.
2. Bactericidal drugs that kill tubercle bacilli only in certain situations. Streptomycin is effective in neutral or alkaline environments, such as the cavity wall; pyrazinamide is active only in acidic environments, such as within macrophages and in dense inflammatory tissue; and isoniazid only kills actively replicating bacilli.
3. Bacteristatic drugs, which are of limited usefulness and are only used when drug resistance renders the agents in the two groups above ineffective.
Table 18.4 In-vivo activity of antituberculosis drugs
| Sterilizing | Bactericidal | Bacteristatic |
|---|---|---|
| Rifampicin | Isoniazid | Ethionamide |
| Pyrazinamide | Streptomycin | Prothionamide |
| Ethambutola | Thiacetazone | |
| Quinolones | p-Aminosalicylic acid | |
| Macrolides | Cycloserine |
Mutation to drug resistance occurs at a rate of about one mutation every 108 cell divisions. Successful therapy requires the prevention of the emergence of drug-resistant strains by the simultaneous use of at least two drugs to which the organism is sensitive.
The current World Health Organization recommendations are that all new patients with tuberculosis, irrespective of site or severity of disease, and in the absence of evidence of drug resistance, should receive a 6-month course of therapy, consisting of a 2-month intensive phase of rifampicin, isoniazid, pyrazinamide and ethambutol followed by 4-month phase of rifampicin and isoniazid. Ideally, the drugs are given daily, but they may be given thrice weekly during the continuation phase or throughout, provided that all doses are given under careful supervision and that the patient is not HIV seropositive.
The response to therapy of drug-susceptible tuberculosis is divisible into three phases:
1. During the first week or two, the large numbers of actively replicating bacilli in cavity walls are killed, principally by isoniazid, but also by rifampicin and ethambutol. The patient rapidly ceases to be infectious, and prolonged hospitalization with barrier nursing is now rarely necessary.
2. In the following few weeks the less active bacilli within macrophages, caseous material and dense, acidic, inflammatory lesions are killed by pyrazinamide and rifampicin.
3. In the continuation phase any remaining dormant bacilli are killed by rifampicin during their short bursts of metabolic activity. Any rifampicin-resistant mutants that arise and start to replicate are killed by isoniazid.
Short-course regimens are usually well tolerated by the patient. Isoniazid, rifampicin and pyrazinamide are all potentially hepatotoxic, and rifampicin may cause an influenza-like syndrome, which, paradoxically, is more likely to occur when the drug is given intermittently. Rifampicin antagonizes the action of several drugs and also oral contraceptives – an important point to be considered when treating women. There is also mutual antagonism between rifampicin and the antiretroviral drugs used for HIV infection (see Ch. 55). Isoniazid may cause mild psychiatric disturbances and peripheral neuropathy, particularly in alcoholics, but these are usually preventable by prescribing pyridoxine (vitamin B6). Ethambutol is toxic to the eye and, although this is rare with standard dosages, care is required; the patient should be informed of this possibility.
The emergence of drug-resistant strains is a major barrier to the control of tuberculosis. According to World Health Organization there were, in 2009, an estimated 250 000 patients (range, 230 000–270 000) with multidrug resistant tuberculosis worldwide, but only 12% were diagnosed and notified, indicating a serious need to strengthen services to address this emerging problem.
Resistance may develop during therapy (acquired resistance) with poor quality drugs or inadequate supervision, or patients may be infected with resistant strains (initial or primary resistance). Multidrug-resistant strains are defined as those resistant to isoniazid and rifampicin, with or without resistance to additional drugs. The newer category of extensively drug resistant tuberculosis (XDR-TB) is defined as resistance to, at least, isoniazid, rifampicin, any fluroquinolone and any injectable agent. The exact incidence of XDR-TB is unknown as susceptibility testing to a wide range of drugs is not widely available but in June 2008, 49 countries reported confirmed cases.
Wherever possible, drug susceptibility tests, at least for isoniazid and rifampicin and ideally by rapid molecular methods, should be conducted on all patients who relapse after a course of standard therapy. The categories of agents for treating drug-resistant tuberculosis are listed in Table 18.5 and their use depends on local guidelines based on known resistance trends. Group 1 agents are the most effective and best tolerated; Group 2 (injectable) and Group 3 (fluroquinolone) agents are added unless resistance to them is has been demonstrated. Group 4 agents are less effective and often poorly tolerated and, like Group 5 agents, are used in cases of extensive resistance in which the previous groups would prove ineffective. With careful and caring supervision, the majority of patients with drug resistance can be cured, although HIV seropositivity is a poor prognostic factor.
Table 18.5 WHO groups of antituberculosis agents for the treatment of multi- and extensive drug resistant tuberculosis
| Group | Antituberculosis agent |
|---|---|
| 1: First-line oral agents | |
| 2: Injectable agents | |
| 3: Fluoroquinolones | |
| 4: Oral bacteriostatic second-line agents | |
| 5: Agents with unclear roles in the treatment of drug resistant tuberculosis |
Epidemiology
In 2009 there were, according to World Health Organization estimates, 9.4 million new cases of tuberculosis (range, 8.9 million–9.9 million) and 14 million prevalent cases (range, 12 million–16 million) worldwide. Most cases were in the South-East Asia (35%), African (30%) and Western Pacific regions (20%). There were an estimated 1.3 million deaths due to tuberculosis among HIV-negative people (range, 1.2 million–1.5 million) and 0.38 million deaths among HIV-positive people (range, 0.32 million–0.45 million).
Human tuberculosis is transmitted principally by inhalation of bacilli in moist droplets coughed out by individuals with open pulmonary tuberculosis. Dried bacilli in dust appear to be much less of a health hazard. The sputum of patients with positive findings on microscopy contains at least 5000 tubercle bacilli per millilitre; these patients are considerably more infectious than those who have negative findings on microscopy. Most transmission of the disease occurs within households or other environments where individuals are close together for long periods.
For epidemiological purposes, the incidence of infection by the tubercle bacillus in a community is calculated from the number of tuberculin-positive individuals in different age groups, provided they have not received BCG vaccination. The annual infection rate gives an indirect measure of the number of open or infectious individuals in the community. Although subject to many variables, a 1% annual infection rate indicates that there are around 50 infectious cases in every 100 000 members of the community. The prevalence of tuberculosis in a region is not the same as the annual infection rate, because only a minority of infected individuals develop overt disease and they often become ill several years after the initial infection.
Tuberculosis is relatively uncommon in the industrially developed nations, with an annual infection rate of 0.3–0.1% or less, but the previous decline has halted and in several countries there has been an increase in notifications since around 1990. The disease is often concentrated in certain ‘hot spots’, particularly deprived inner city areas. In the developing countries the annual infection rates are usually between 2% and 5%.
In many parts of the world, implementation of the World Health Organization control strategies have led to substantial reductions in the incidence of tuberculosis but, overall these gains have been offset by the impact of the HIV/AIDS pandemic. In 2009, 11–12% of all cases of tuberculosis were HIV related, with 80% of cases occurring in sub-Saharan Africa.
A person infected by the tubercle bacillus before acquiring HIV has an 8% annual risk of developing active tuberculosis but the risk of an HIV-infected person developing tuberculosis after infection or reinfection is much higher, approaching 100%. The interval between infection and overt disease is considerably shortened, resulting in explosive mini-epidemics in institutions where HIV-positive persons congregate. Some such mini-epidemics have involved multidrug-resistant strains, notably in the USA.
Bovine tuberculosis is spread from animal to animal, and sometimes to human attendants, in moist cough spray. About 1% of infected cows develop lesions in the udder, and bacilli excreted in the milk can then infect people who drink it. Heat treatment, or pasteurization, prevents milk-borne disease. In most developed countries, the incidence of bovine tuberculosis has been reduced drastically by regular tuberculin testing of herds and the slaughter of reactors, but the total eradication has been prevented by infection of cattle from wild animals, notably badgers in the UK and possums in New Zealand. Human disease due to M. bovis is very rare in developed countries and is usually the result of reactivation of old lesions. Cattle have occasionally been infected by farm workers with open tuberculosis due to M. bovis. The prevalence of the disease in cattle in the developing nations is poorly documented. Person-to-person spread of M. bovis leading to active disease is uncommon, although a few instances of such spread among HIV-positive persons have been reported. Occupational exposure to goats and seals has resulted in a few cases of tuberculosis due, respectively, to M. caprae and M. pinnipedi.
Control
Human tuberculosis is preventable:
• by the early detection and effective therapy of the open or infectious individuals in a community
• by lowering the chance of infection by reducing overcrowding
Active case finding involves a deliberate search, often on a house-to-house basis or in workplaces, for suspects with a chronic cough of a month or more in duration. Merely waiting for patients with symptoms to seek medical attention is much less effective, even when supported by education programmes. Regular chest examination by mass miniature radiography detects fewer than 15% of individuals with tuberculosis and its use is now restricted to certain high-risk situations.
The most important factors affecting the incidence of tuberculosis are socio-economic ones, particularly those leading to a reduction of overcrowding in homes and workplaces. In developing countries it is estimated that each patient with open tuberculosis infects about 20 contacts annually, whereas in Europe the corresponding figure is two or three.
Vaccination
Bacille Calmette–Guérin (BCG) is a living attenuated vaccine derived from a strain of M. bovis by repeated subculture between the years 1908 and 1921. This species was selected rather than M. tuberculosis as the vaccine was initially intended for veterinary use. Although originally administered orally, BCG is now given by intracutaneous injection.
The protective efficacy of BCG varies enormously from country to country. Early trials in the UK indicated that administration of BCG to schoolchildren afforded 78% protection, but a major trial in south India involving individuals of all ages found no protection (Table 18.6). Several explanations have been advanced for this difference, the most likely one being prior exposure of the human population to environmental mycobacteria, which, in some regions, confer some protection, but in others induce inappropriate immune reactions that antagonize protection. For this reason, neonatal vaccination is recommended.
Table 18.6 Variations in the protective efficacy of BCG vaccinations in nine major trials
| Country or population | Age range of vaccinees (years) | Protection (%) |
|---|---|---|
| North American Indian | 0–20 | 80 |
| UK | 14–15 | 78 |
| Chicago, Illinois, USA | Neonates | 75 |
| Puerto Rico | 1–18 | 31 |
| South India (Bangalore) | All ages | 30 |
| Georgia and Alabama, USA | >5 | 14 |
| Georgia, USA | 6–17 | 0 |
| Illinois, USA | Young adults | 0 |
| South India (Chingleput) | All ages | 0a |
a Some protection demonstrated in those vaccinated neonatally on 15-year follow-up.
When BCG vaccination is introduced into a region it has an immediate impact on the incidence of the serious but non-infectious forms of childhood tuberculosis such as meningitis, but has little impact on the annual infection rate in the community as the smear-positive source cases arise mostly from among the older, unvaccinated, tuberculin-positive members of the community. Vaccination has not therefore proved to be an effective control measure.
Being a living vaccine, serious infections and even disseminated disease may occur in immunocompromised persons. Thus, BCG should never be given to persons known to be HIV-positive. Many attempts are currently being made to develop alternative vaccines, particularly non-viable subunit ones.
Prophylactic chemotherapy
In true prophylactic chemotherapy, drugs are administered to uninfected individuals who are in unavoidable contact with a patient with open tuberculosis. The main example is an infant born to a mother with the disease. More usually, it refers to therapy, principally with isoniazid alone for 6–12 months, given to individuals who have been infected but show no signs of active disease. A shorter, 3-month, regimen of isoniazid and rifampicin is as effective as a 12-month course of isoniazid and with less hepatic toxicity and is advocated in the UK. An even shorter, 2-month, regimen of isoniazid, rifampicin and pyrazinamide was equally effective but was withdrawn due to severe and sometimes fatal adverse effects in HIV-positive patients. Persons co-infected with M. tuberculosis and HIV who are positive on tuberculin testing or the interferon-γ test are much more responsive to preventive therapy than those who are negative, and the WHO thus recommends prophylactic therapy only for the former. Other indications are for children positive on tuberculin testing or the interferon-γ test, recently converting adults exposed to a source case or with an immune suppressing condition, and those with fibrotic lung lesions suggesting untreated but healed tuberculosis.
Mycobacterium leprae
Leprosy is a particularly tragic affliction as the nature of the infection often causes severe disfigurement and deformity which, throughout history, have led to social ostracism or even total banishment from society of those suffering from the disease. The disease was long endemic in the British Isles; Robert the Bruce of Scotland was one of its victims. The last British patient to acquire the disease in this country died in the Shetland Islands in 1798. In Norway the disease persisted into the twentieth century; Armauer Hansen first described the causative organism in that country in 1873.
The current situation is cause for optimism as the number of registered patients on treatment declined from more than 10 million in 1982 to 514 718 in 2003 and 244 796 in 2009. The decline has been slower in recent years, but this may reflect a higher case detection rate. Leprosy has been eliminated from 119 out of the 122 countries in which the disease was regarded as a public health problem in 1985 and, in stark contrast to tuberculosis, resistance to therapy has not proved to be a barrier to treatment. The World Health Organization has therefore adopted a ‘final push’ strategy with the goal of eliminating this disease.
Leprosy is often cited as a disease of great antiquity but literary and skeletal evidence of this very characteristic disease go back no further than 500 BC. Biblical leprosy was almost certainly not the same as the disease that now bears this name. This raises the intriguing question of the ancestry of this bacillus which has no widespread animal reservoirs. Natural environmental reservoirs for a progenitor of M. leprae, such as amoebe, have been postulated but not convincingly demonstrated. Likewise, it has been postulated that M. leprae can survive in the environment, possibly in amoebe, and infect human beings.
Description
M. leprae has never convincingly been cultivated in vitro; this has been attributed to a loss or disruption of many of the genes required for metabolism. The genome of M. leprae is smaller than that of cultivable mycobacteria, and about half of its genes are defective counterparts of functional ones found in other mycobacteria. In the 1970s it was shown that armadillos experimentally infected with M. leprae often developed extensive disease, with up to 1010 bacilli in each gram of diseased tissue. This animal has therefore provided sufficient bacilli for research projects and for the production of a skin test reagent, leprosin-A. Limited replication, yielding 106 bacilli after 6–8 months, also occurs in the mouse footpad, and this has been used for testing the sensitivity of bacilli to antileprosy drugs.
Leprosy bacilli resemble tubercle bacilli in their general morphology, but they are not so strongly acid-fast. In clinical material from lepromatous patients, the bacilli are typically found within macrophages in dense clumps. A characteristic surface lipid, peptidoglycolipid-1 (PGL-1), has been extracted from M. leprae, and its unique carbohydrate antigenic determinant has been synthesized. Specific PCR primers for diagnostic purposes and for seeking the bacillus or its progenitor in the natural environment have been synthesized.
Pathogenesis
The principal target cell for the leprosy bacillus is the Schwann cell. The resulting nerve damage is responsible for the main clinical features of leprosy: anaesthesia and muscle paralysis. Repeated injuries to, and infection of, the anaesthetic extremities leads to their gradual destruction (Fig. 18.6). Infiltration of the skin and cutaneous nerves by bacilli leads to the formation of visible lesions, often with pigmentary changes.
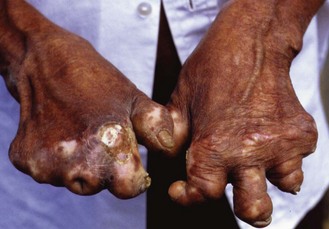
Fig. 18.6 Borderline tuberculoid leprosy. Trophic changes in the hands secondary to nerve damage, vasculitis and anaesthesia.
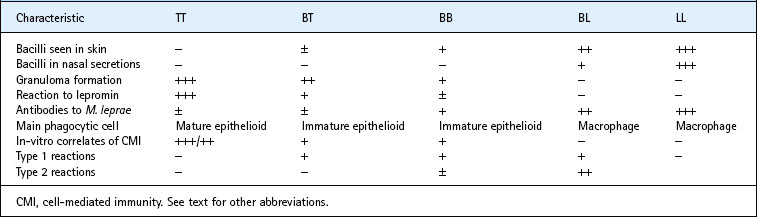
The first sign of leprosy is a non-specific or indeterminate skin lesion, which often heals spontaneously. If the disease progresses, its clinical manifestation is determined by the specific immune responsiveness of the patient to the bacillus, and there is a distinct immunological spectrum of the disease (Table 18.7). The points on the spectrum are:
• Hyper-reactive tuberculoid (TT) leprosy, with small numbers of localized skin lesions containing so few bacilli that they are not seen on microscopy and an inappropriately intense granulomatous response that often damages major nerve trunks.
• Anergic lepromatous (LL) leprosy, in which the skin lesions are numerous or confluent and contain huge numbers of bacilli, usually seen as clusters or globi within monocytes. Cooler parts of the body, such as the ear lobes, are particularly heavily infiltrated by bacilli (Fig. 18.7). There is no histological evidence of an immune response.
• Intermediate forms classified as borderline tuberculoid (BT), mid-borderline (BB) or borderline lepromatous (BL).
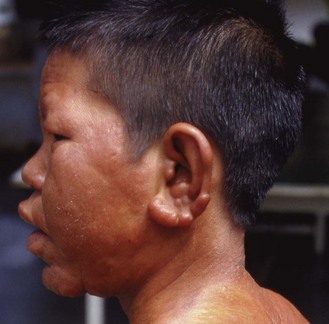
Fig. 18.7 Lepromatous leprosy. Nodular swelling of face, enlargement of ear lobes and loss of eyebrows.
Destruction of the nasal bones may lead to collapse of the nose (Fig. 18.8). In addition, large numbers of leprosy bacilli are discharged in nasal secretions in multibacillary disease. The eye is frequently damaged by direct bacillary invasion, uveitis or corneal infection secondary to paralysis of the eyelids (Fig. 18.9). Blindness is a common and tragic complication of untreated leprosy.

Fig. 18.8 Treated lepromatous leprosy. The nodularity of the skin has resolved on treatment but the absence of eyebrows and the nasal collapse remain.

Fig. 18.9 Tuberculoid leprosy. The only feature on presentation was paralysis of the left facial nerve, resulting in loss of the left nasolabial fold and an inability to close the eye, predisposing to corneal damage.
Additional tissue damage in leprosy is caused by immune reactions resulting from delayed hypersensitivity (Jopling type 1 reactions) or a vasculitis associated with the deposition of antigen–antibody complexes (Jopling type 2 reactions, erythema nodosum leprosum) (Table 18.8). The former, which occurs in borderline cases (BT, BB and BL), may rapidly cause severe and permanent nerve damage, and requires urgent treatment with anti-inflammatory agents and, sometimes, surgical decompression of a greatly swollen nerve. The latter occurs at the multibacillary pole of the spectrum (BL and LL) and is principally due to deposition of antigen-antibody complexes.
Table 18.8 Main characteristics of the reactions in leprosy
| Characteristic | Type 1 (reversal reaction) | Type 2 (erythema nodosum leprosum) |
|---|---|---|
| Immunological basis | Cell mediated | Vasculitis with antigen-antibody complex deposition |
| Type of patient | BT, BB, BL | BL, LL |
| Systemic disturbance | No (or mild) | Yes |
| Haematological changes | No | Yes |
| Proteinuria | No | Frequently |
| Relation to therapy | Usually within first 6 months | Rare during first 6 months |
See text for explanation of abbreviations.
Laboratory diagnosis
The clinical diagnosis may be confirmed by histological examination of skin biopsies and by the detection of acid-fast bacilli in nasal discharges, scrapings from the nasal mucosa and slit-skin smears. The latter are prepared by making superficial incisions in the skin, scraping out some tissue fluid and cells, and making smears on glass slides. Smears are obtained from obvious lesions, the ear lobes and apparently unaffected skin. Secretions and skin smears are stained by the ZN method, and the number of bacilli seen in each high-power field may be recorded as the bacillary index; however, for usual practical purposes, patients with clinically active leprosy but in whom no bacilli are seen on slit-skin smear examination are described as having paucibacillary disease, and those who are positive at any site are described as having multibacillary disease. This is an important distinction for the selection of treatment (see below).
It is widely assumed, but unproven, that leprosy bacilli that stain strongly and evenly are viable whereas those that stain weakly and irregularly are dead. The percentage of the former gives the morphological index, which declines during chemotherapy. An increase in the morphological index is a useful indication of non-compliance of the patient and the emergence of drug resistance. Where facilities exist, PCR may be used to detect M. leprae in clinical specimens.
Treatment
Multidrug therapy based on dapsone, rifampicin and clofazimine is highly effective. The choice of regimen is determined by whether the patient has paucibacillary or multibacillary disease (Table 18.9). Clofazimine causes skin discoloration, particularly in fair-skinned people. If this results in the patient refusing the drug, a combination of rifampicin, ofloxacin and minocycline, administered monthly for 24 months for multibacillary disease, may be used instead. A single dose of rifampicin (600 mg), ofloxacin (400 mg) and minocycline (100 mg) has been advocated for the treatment of adults with single-lesion paucibacillary leprosy, although some authorities consider this to be inadequte.
The treatment of leprosy demands far more than the administration of antimicrobial agents. It is often necessary to:
Epidemiology
Once thought to be restricted to humans, leprosy has been reported in chimpanzees and sooty mangabey monkeys in Africa, and in free-living armadillos in Louisiana, USA. It was also thought that leprosy was transmitted by skin-to-skin contact but it now appears more likely that the bacilli are disseminated from the nasal secretions of patients with lepromatous leprosy. In addition, the blood of patients with lepromatous leprosy contains enough bacilli to render transmission by blood-sucking insects a definite, though unproven, possibility.
As in the case of tuberculosis, transmission of bacilli from patients with multibacillary leprosy to their contacts occurs readily, but only a minority of those infected develop overt disease. The infectivity of patients with paucibacillary leprosy is much lower. Leprosy often commences during childhood or early adult life but, as the incubation period is usually 3–5 years, it is rare in children aged less than 5 years.
Epidemiological studies on the prevalence and transmission of leprosy have been aided by skin test reagents, of which there are two types:
1. Lepromins, which are prepared from boiled bacilli-rich lepromatous lesions.
2. Leprosins, which are ultrasonicates of tissue-free bacilli extracted from lesions (the suffixes -H and -A are used to denote human and armadillo origins, respectively).
These reagents elicit two types of reaction:
1. The Fernandez reaction is analogous to tuberculin reactivity and appears in sensitized subjects 48 h after skin testing. It is best seen with leprosins.
2. The Mitsuda reaction is a granulomatous swelling that appears about 3 weeks after testing with lepromin. This reaction is indicative of the host’s ability to give a granulomatous response to antigens of M. leprae, and is positive at or near the TT pole.
Skin testing is of limited diagnostic value, but is useful in epidemiological studies to establish the extent of infection in contacts and in the community, and in classifying patients according to the immunological spectrum.
Control
The most effective control measure in leprosy, as in tuberculosis, is the early detection and treatment of infectious cases. This requires that patients should attend for therapy as soon as signs of the disease appear. Unfortunately, owing to the stigma associated with the disease, many patients delay seeking treatment until they have infected many contacts and developed irreversible disfigurement and handicap. Women, in particular, are likely to conceal their disease.
No living attenuated vaccines have been prepared for M. leprae, but BCG vaccine seems to protect against leprosy in regions where it protects against tuberculosis, strongly suggesting that protection is induced by common mycobacterial antigens.
Britton WJ. Leprosy. In: Cohen J, Powderly WG, Opal SM. Infectious Diseases. ed 3. London: Mosby Elsevier; 2010:1099–1105.
Brown T, Nikolayevskyy V, Velji P, Drobniewski F. Associations between Mycobacterium tuberculosis strains and phenotypes. Emerging Infectious Diseases. 2010;16:272–280.
Cole ST, Brosch R, Parkhill J, et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature. 1998;393:537–544.
Davies PDO, Barnes PF, Gordon SB. Clinical Tuberculosis, ed 4, London: Hodder Arnold, 2008.
Ridley DS. Pathogenesis of Leprosy and Related Diseases. London: Wright; 1988.
Scollard DM, Adams LB, Gillis TP, et al. The continuing challenges of leprosy. Clinical Microbiology Review. 2009;19:338–381.
Withington SG. Leprosy. In: Cook GC, Zumla AI. Manson’s Tropical Diseases. ed 22. London: Saunders Elsevier; 2009:1053–1074.
World Health Organization. Global Tuberculosis Control 2010. Geneva: WHO; 2010.
World Health Organization. Multidrug and extensively drug-resistant TB (M/XDR-TB): 2010 global report on surveillance and response. Geneva: WHO; 2010.
World Health Organization. Treatment of Tuberculosis: Guidelines, ed 4. Geneva: WHO; 2009.
Zumla A, Schaaf S. Tuberculosis – a Comprehensive Clinical Reference. London: Elsevier, 2009.
Centers for Disease Control and Prevention. Division of Tuberculosis Elimination. http://www.cdc.gov/tb/.
International Federation of Anti-Leprosy Associations. http://www.ilep.org.uk/.
International Leprosy Association. http://www.leprosy-ila.org/.
International Union Against Tuberculosis and Lung Disease. http://www.theunion.org/.
Montefiore Medical Center, New York. http://www.tuberculosis.net/.
New Jersey Medical School Global Tuberculosis Institute. http://www.umdnj.edu/globaltb/home.htm.
TB Alert. http://www.tbalert.org/.
Tuberculosis Structural Genetics Consortium. http://www.webtb.org/.
World Health Organization. A World Free of TB. http://www.who.int/tb/en/.
World Health Organization. Leprosy elimination. http://www.who.int/lep/.
World Health Organization. Stop TB Partnership. http://www.stoptb.org/.
World Health Organization. Tuberculosis. http://www.who.int/topics/tuberculosis/en/.