55 Retroviruses
Acquired immune deficiency syndrome; HTLV-1
Key points
• Some retroviruses, including HTLV-I, the cause of human T-cell leukaemia, cause tumours in natural hosts.
• HIV-1 is the cause of the acquired immune deficiency syndrome (AIDS), a persistent infection leading to loss of CD4+ T cells, immunodeficiency and many opportunistic infections.
• Disease status can be measured by sequential changes in CD4 count and viral load in plasma (HIV RNA copy number).
• HIV has a global distribution; it is spread by sexual intercourse, mother-to-child transmission and via blood and blood products. The greatest incidence is in sub-Saharan Africa and South-East Asia.
• HIV replication can be inhibited by drugs that block co-receptor binding, membrane fusion, reverse transcription, integration and protein cleavage during maturation.
• Combination antiretroviral therapy (cART) reduces the appearance of drug resistance and, combined with control of opportunistic infections, substantially improves survival.
The family Retroviridae contains many viruses from widely different host species. They have been studied for many years, initially because a wide variety of tumours including leukaemias and lymphomas, sarcomas, breast and brain tumours are caused by oncogenic members of this family. Other retroviruses are associated with a plethora of neurologic, autoimmune and blood disorders. The host species include birds, mice, cattle, pigs and several primates. The first human retrovirus was isolated from T cells of patients with T cell leukaemia – human T-cell lymphotropic virus type I or HTLV-I – in 1980. The acquired immune deficiency syndrome (AIDS) is caused by a different retrovirus, also with a predilection for T cells that is referred to as human immunodeficiency virus type 1 or HIV-1. Infection with this virus has become pandemic and remains a major cause of morbidity and mortality in sub-Saharan Africa and other developing countries. HIV-2 infection is restricted largely to West Africa and is less pathogenic.
Description
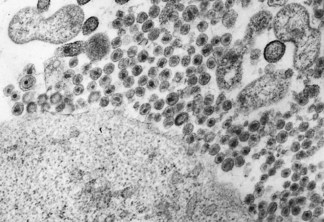
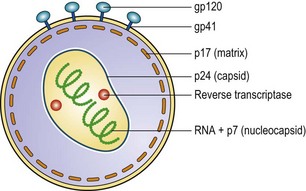
All retroviruses have an outer envelope of lipid and viral proteins; the envelope encloses the core, consisting of other viral proteins, within which lie two molecules of viral RNA (positive single-stranded) and the enzyme reverse transcriptase, an RNA-dependent DNA polymerase. The virions have a diameter of about 100 nm (Fig. 55.1) and, in thin section, characteristic differences can be seen in the appearance and position of the core (e.g. C-type and D-type particles), a feature that was previously used to classify retroviruses. The typical genome size is approximately 10 kilobases (kb) or less.
Classification
The Retroviridae family was originally divided into the subfamilies Oncovirinae, Spumavirinae and Lentivirinae, based on their biological properties and appearances in cell cultures. However, nucleotide sequencing of a large number of human and animal retroviruses has revealed that viruses cluster in two subfamilies, the Spumavirinae and Orthoretrovirinae, with the latter comprising 6 distinct genera, the Alpha-, Beta-, Delta-, Epsilon- and Gamma-retroviruses and the Lentivirus genus (Fig. 55.2).
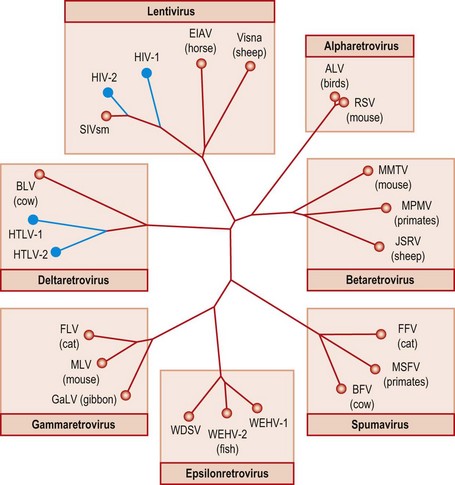
Fig. 55.2 Tree of sequences from the retroviral pol gene showing relatedness of retroviruses infecting man and a range of animal species. Sequences form seven main genera, in which human retroviruses (HTLV and HIV; shown in blue) are found in two. For animal retroviruses (red), the host species and names of familiar viruses are indicated. ALV, avian leukosis virus; BFV, Bovine foamy virus; BLV, bovine leukaemia virus; EIAV, equine infectious anaemia virus; FFV, feline foamy virus; FLV, feline leukaemia virus; GaLV, Gibbon ape leukaemia virus; JSRV, Jaagsiekte sheep retrovirus; MLV, murine leukaemia virus; MMTV, mouse mammary tumour virus; MPMV, Mason-Pfizer monkey virus; MSFV, Macaque simian foamy virus; RSV, Rous sarcoma virus; SIV, simian immunodeficiency virus; WDSV, walleye dermal sarcoma virus; WEHV, walleye epidermal hyperplasia virus.
The spumaviruses have been detected in various species, including cats, cattle and primates, but are not associated with disease. The name is derived from the foamy (vacuolated) appearance of infected cells in culture. There is no evidence for pathogenic human infection with spumaviruses.
The human retroviruses HTLV-I and HTLV-II belong to the genus Deltaretrovirus (formerly termed Oncovirus). They are related to the simian viruses STLV-I and -II, which are widely distributed in Old and New World monkeys. STLV-I shows approximately 90% similarity to HTLV-I at the sequence level. A more distantly related virus is found in cows (Bovine Leukaemia Virus). The recent identification of two new HTLV types (HTLV-III and HTLV-IV) in a few bush meat hunters in central Africa is believed to represent isolated incidents of primate-to-human virus transmission rather than an established human infection.
The other two human retroviruses HIV-1 and HIV-2 are lentiviruses, closely related to lentiviruses that infect Old World primates. HIV-2 is almost identical to simian immunodeficiency virus (SIVsm) found in sooty mangabeys. It is believed that human infection originated through cross-species transmission. Similarly, HIV-1 corresponds closely to SIVcpz variants that infect chimpanzees in Central Africa, the probable source of the human virus. The genus Lentivirus also includes feline and bovine immunodeficiency viruses and Visna-Maedi of sheep, the first lentivirus to be recognized. Lentiviruses are distinguished from Deltaretroviruses by their molecular structure and lack of oncogenic capability; however both genera are capable of establishing prolonged asymptomatic infection.
Both HIV-1 and HIV-2 show considerable sequence variability, allowing their classification into a number of subtypes with marked differences in geographical distribution and association with risk groups. HIV-1 variants are classified into three genetic groups: major (M), outlier (O) and non-M, non-O (N). Group M viruses which dominate the pandemic are further classified into subtypes A, B, C, D, F, G, H, J and K and several ‘circulating recombinant forms’ (CRF) that comprise more than one subtype, e.g. CRF01-A/E. Subtype B is most frequently found in western countries, whereas other genotypes such as C (Africa, parts of Asia), E (Thailand) and F (South America) are the main subtypes responsible for the recent epidemic spread. HIV-1 diversity is greatest in sub-Saharan African countries such as the Congo, where there is wide co-circulation of most of the current subtypes.
Human endogenous retroviruses, often replication-defective, are present in the human germ line and have not been shown to cause disease.
Genome and gene coding assignment
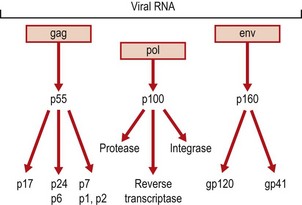
The genome organization is similar for all retroviruses as their genomes contain in the same order the genes gag, pol and env coding for three groups of structural and enzymatic proteins (Figs 55.3 & 55.4). The long terminal repeat sequences (LTR) at both ends of the genome contain promoter and enhancer sequences. However, there are important differences between the viruses in the accessory genes involved in the regulation of replication which are found only in Complex retroviruses (Δ-, ε-, lenti- and spumaviruses). Simple retroviruses (α-, β- and γ-retroviruses) encode only for gag, pol and env genes products. Studies of HTLV genes and gene products provided the basis for understanding of the functional homologues subsequently found in HIV, despite the lack of significant sequence homology between the two viruses. The HIV trans-activating gene, tat, which stimulates the synthesis of all viral proteins and rev, the gene that mediates the transport of unspliced viral mRNA from the nucleus to the cytoplasm are homologues of the HTLV genes tax and rex, respectively.
The six proteins coded for by the gag gene, of HIV, are all found in the virion (see Figs. 55.3 and 55.4). The pol gene products are the protease, reverse transcriptase and integrase enzymes; all required during replication. The env gene codes for a large protein that is glycosylated and cleaved to gp41, the transmembrane protein and gp120, present on the envelope as a trimer with many glycosylation sites.
Replication
Retroviruses replicate and produce viral RNA from a DNA copy of the virion RNA (hence their name).
Initial attachment of HIV to target cells is by the interaction of the external envelope glycoprotein gp120 with part of the CD4 molecule of T helper lymphocytes and other cells; the HIV envelope then interacts with a second (co-) receptor. These include the chemokine receptors, CCR5 and CXCR4 expressed on a wide range of lymphoid and non-lymphoid cells, whose ligands are chemotactic cytokines such as CCL3 (formerly known as macrophage inflammatory protein-1α). After this second binding step, entry of the virus occurs by fusion of the viral envelope with the cellular membrane, which requires exposure of a hydrophobic domain in gp41. Once the RNA is released into the cytoplasm, the reverse transcriptase acts to form the double-stranded DNA copy, which is transported to the nucleus and is spliced into the host cell DNA (see Ch. 7) in which state it is referred to as the provirus. Once inserted into the host DNA, infection with HIV is permanent. The virus may stay latent or enter a productive cycle. Transcription of mRNA from the provirus is by the host RNA polymerase II to produce viral mRNA and RNA. Proteins are synthesized and processed to form the virion components (see Figs 55.3 & 55.4). Virions are assembled at the cell membrane where envelope and core proteins have located. The internal structure of the virion matures as it buds from the cell; the entire replicative cycle is completed in approximately 24 hours. In the productive growth cycle the host cell is frequently destroyed.
HIV infection
Clinical features
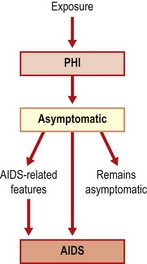
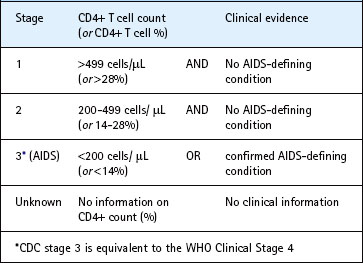
The different stages of HIV infection are summarized in Figure 55.5. These stages are generally reflected in CDC (Table 55.1) and WHO surveillance case definitions that utilize clinical and immunological evidence to classify established HIV infections. Both schemes may provide reliable information on the stage of the epidemic within populations in which combination antiretroviral therapy (cART) is not universally used, however their usefulness is somewhat limited in the cART era. Patients are assigned a stage according to the lowest CD4 positive T lymphocyte cell count (CD4+) or worst clinical stage they have ever reached.
Table 55.1 Staging of laboratory confirmed human immunodeficiency virus infection (Communicable Disease Centre, CDC)
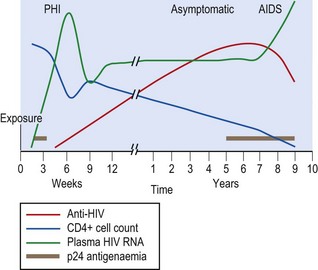
Symptomatic primary HIV infection (PHI), often referred to as acute seroconversion illness, occurs within 10–30 days of initial exposure to the virus and resolves in the majority of infected individuals within a month. Features described include fever, pharyngitis, headache, malaise, generalized lymphadenopathy and non-pruruitic maculopapular rash. Although up to 70% of patients experience some symptoms, only 5–10% show the full picture. Even fewer have a rare aseptic meningoencephalitis presentation.
Following resolution of primary infection symptoms, a prolonged, largely asymptomatic phase ensues. This may last as long as 10 years during which the virus continues to replicate resulting ultimately in significant damage to the immune system in untreated individuals. Persistent generalized lymphadenopathy is present in 30–70% of patients who are otherwise asymptomatic. The rate of progression of patients to AIDS is no greater in these patients than in those without adenopathy.
The inexorable decline in immune function eventually predisposes the patient to the development of the acquired immune deficiency syndrome (AIDS). This may present in many ways, all due to loss of the ability to respond appropriately to infectious agents and to control tumours. There are over 20 AIDS-defining conditions reflecting the specific agents involved; a diagnosis of AIDS is made if one (or more) of the conditions listed in Box 55.1 is present.
Box 55.1
AIDS defining clinical conditions
• Bacterial infections, multiple or recurrent in child aged less than 13 years
• Candidiasis of oesophagus, bronchi, or lungs
• Coccidiomycosis, disseminated
• Cryptococcosis, extrapulmonary
• Herpes simplex virus for more than 1 month; or bronchi, lung or oesophagus involved
• Histoplasmosis, extrapulmonary or disseminated
• Lymphoma, non-Hodgkin’s or primary in brain
• Lymphoid interstitial pneumonia/pulmonary lymphoid hyperplasia in child less than 13 years
• Mycobacterium tuberculosis, any site
• Mycobacterium (other species), extrapulmonary or disseminated
• Pneumocystis jirovecii pneumonia
• Progressive multifocal leuco-encephalopathy
The occurrence of an AIDS-defining illness may be preceded by non-specific features such as fever, adenopathy and weight loss and minor opportunistic infections such as reactivation of latent herpes viruses (e.g. herpes zoster), oral candidiasis and oral hairy leucoplakia (secondary to Epstein–Barr virus infection). The latter condition in which the margins of the tongue show white ridges of fronds on the epithelium appears to be unique to HIV-infected patients. Without treatment, such patients progress rapidly to AIDS.
Pneumocystis jirovecii pneumonia was the presenting illness in many of the first AIDS patients. Salient features include fever, unproductive cough and progressive shortness of breath. Radiological examination classically shows bilateral lung infiltrates radiating out of the peri-hilar region on plain chest film and/or ground-glass lung opacities on high resolution CT scan. Diagnosis is confirmed by detection of fungal cysts in deep respiratory specimens and increasingly by fungal DNA detection by PCR (see Ch. 61).
Toxoplasma gondii remains the most common cause of AIDS complications affecting the brain. Toxoplasma encephalitis (TE), which is almost always caused by a reactivation of latent toxoplasma gondii cysts, classically presents with headache, confusion and fever, developing subacutely over a few days to a month. Seizure and focal neurological signs such as hemiparesis are often present and head MRI (or CT) scans show multiple, ring enhancing brain lesions in most cases. The imaging findings are characteristic but not diagnostic of TE.
Kaposi’s sarcoma was one of the earliest diseases used to define AIDS. This previously rare tumour had been known for many years; it usually occurred at a single site and was not aggressive. In patients with AIDS the tumour arises in many sites, including the skin, mouth, gut and eye. The tumours arise from endothelial cells of blood vessels, causing bluish-purple, raised, irregular lesions. The aetiological agent is human herpesvirus 8 (see Ch. 43). The tumours were mainly seen in homosexual men, presumably reflecting sexual transmission of the causative agent; the incidence has now declined.
In developing countries, whilst many of the same infections are seen, there is also an emphasis on local problems. Mycobacterium tuberculosis infections are an enormous problem in many regions, with the development of strains of the organism resistant to many antibiotics. Many patients show profound weight loss, perhaps accompanied by chronic diarrhoea; the term ‘slim disease’ has been given to this presentation.
Paediatric patients with AIDS suffer from many of the same problems as adults. However, children infected early in life or at birth are at risk of recurring bacterial infections as they have not acquired immunity to micro-organisms. Lymphoid interstitial pneumonia and pulmonary lymphoid hyperplasia are presentations seen only in young children.
HIV associated clinical manifestations, regardless of whether they are AIDS-defining or not, are diverse and affect all body organ systems. The advent of combination antiretroviral therapy has dramatically decreased the overall incidence of opportunistic infections (OI) owing to the improvement attained in immune function. However, immune recovery may paradoxically worsen underlying OI. This phenomenon, referred to as immune reconstitution inflammatory syndrome (IRIS) is not fully understood. IRIS clinical features vary with the pathogen and the organ system involved (e.g. Cryptococcus neoformans in the brain).
Pathogenesis of HIV infection and AIDS
The major virological and immunological features of the acute and persistent stages of HIV infection are shown in Figure 55.6. The incubation period in the acute stage is 1–2 months. This is preceded by a period of intense, unrestrained viral replication, reflected in the presence of high numbers of viral RNA genomes and p24 antigen in the circulation. After entering the body, virus is taken up by cells such as dendritic cells that express viral receptors. Within 24–48 h infected cells are present in the regional lymph nodes; virus can be detected in the blood and circulating lymphocytes by 5 days; the number of circulating CD4+ T lymphocytes is decreased. As the immune system responds, both p24 antigen and RNA copy number (usually referred to as viral load) decrease, so that by 6–12 months p24 antigen is usually undetectable and the RNA load has stabilized at a lower level (referred to as the set-point); in some it may be undetectable. The HIV viral load set-point, an indirect reflection of the rate of CD4+ cell death, is a strong predictor of the rapidity of progression to AIDS. Temporary increases in viral load can be seen during intercurrent infections, immunizations and pregnancy. CD4+ cell counts recover, albeit partially, and remain more or less within the normal ranges until progression to AIDS occurs, the counts then being less than 200/µL.
In peripheral blood, lymphoid tissue and other tissues such as brain where HIV replication occurs, HIV targets CD4+ T cells and cells of the monocyte–macrophage lineage; the latter may act as a reservoir of virus. Macrophages are also important in carrying the virus into the central nervous system across the blood–brain barrier.
Destruction of CD4+ T cells is caused by:
• syncytium formation via membrane gp120 binding to cell CD4 antigen
• cytotoxic T cell lysis of infected cells
• cytotoxic T cell lysis of CD4+ cells carrying gp120 released from infected cells
• increased susceptibility of CD4+ cells (infected and un-infected) to apoptosis.
The proportion of infected CD4+ cells and the quantity of circulating virus rise as the infection progresses, until the patient becomes symptomatic.
Analysis of viral genomes from an individual patient shows that there are several different viral sequences present at any time and that these change with time. Virus isolated in culture may be different from the predominant variants in the blood. Viruses isolated in the later stages of infection have been shown to grow more rapidly, to higher titres and to form syncytia (giant cells) more readily than virus isolated in the early stages. The switch to syncytium-inducing variants is accompanied by a change in co-receptor use from CCR5 to CXCR4. Regions of the envelope glycoproteins show most variation and this could affect the ability of antibody to react with the viruses. Although this could be relevant to the progression of the infection, it also has important implications for the development of vaccines.
Disease progression
There are host genetic differences influencing the risk of disease progression; for example, human leucocyte antigen (HLA) haplotype A1B8DR3 has been linked to rapid progression to severe disease. There is also an age effect, with evidence of fast progression in some infants and in the elderly. Conversely, other host genetic factors, such as HLA B*5701 and heterozygosity for a 32-bp deletion in the chemokine receptor CCR5 (CCR5-Δ32) and immune response factors, such as effective CTL responses, are strongly linked to long-term non-progression or even resistance to HIV infection. Box 55.2 lists a number of laboratory markers that are associated with progression. The most useful marker in assessing the state of a patient’s immune system is the absolute CD4+ cell count. Although this can vary, a downward trend is indicative of progression: when the count reaches 200/µL the patient is severely compromised and the diagnosis of AIDS is made even in the absence of an AIDS-defining clinical illness (Table 55.1). The median time from infection onset to AIDS is approximately 10 years, although the use of cART has fundamentally changed the outlook for HIV-infected patients.
Paediatric infection
In most paediatric cases, infection arises from mother-to-baby transmission in the perinatal period when the child’s immune system is immature. This results in a major difference from the picture seen in older children and adults as the initial replicative phase is not limited by the immune response and high levels of viral RNA persist. RNA viral loads are often greater than 105 copies/mL at 2 months of age. About 75% of children show a steady decline thereafter; however, by age 9–16 years a third are still asymptomatic and show little impairment of immune function. The other 25% of children have high levels of viral RNA and develop early-onset disease, with death by 20–24 months without treatment. These babies may have been infected before birth by a mother with advanced disease. This group can be identified by detection within peripheral blood of proviral DNA, viral RNA (and culture if available) within 48 h of birth. Analysis of the child’s RNA may show that it differs from that of the mother, suggesting that replication has occurred by the time of sample collection or that a minor maternal variant has been transmitted to the baby.
Laboratory diagnosis
HIV infection can be diagnosed through detection of antibodies to the virus (anti-HIV), or of the virus itself (e.g. HIV p24 antigen, RNA or proviral DNA) in a peripheral blood sample.
Tests for anti-HIV
The main approach to the diagnosis of infection in patients and for screening populations (e.g. blood donors) has been by testing for anti-HIV. Many different testing formats are available, most using enzyme-linked immunoassay (EIA). All current tests use HIV antigens derived from cloned recombinant HIV gag, pol and env genes expressed in Escherichia coli, or synthetic peptides. Western or immunoblotting has been used extensively as a confirmatory assay. Most current assays can detect antibody to both HIV-1 and HIV-2 antigens. A first positive result must be confirmed by at least two other different assays with different viral antigens and a second serum sample checked to confirm that the original sample was identified correctly. At least one of the confirmatory assays should distinguish between antibodies to HIV-1 and to HIV-2. Most patients will seroconvert within 1–2 months (see Fig. 55.6). Thus there is a window before antibody tests can detect infection. Rapid tests are now available to detect antibody in blood and saliva; this format is very useful for point of care testing.
Combination assays
Reliable and highly sensitive methods to detect anti-HIV and p24 antigen in a single EIA are now available (so-called 4th generation assays); these allow improved diagnostic sensitivity of diagnosis of primary HIV infections by approximately 1 week. Although less sensitive than RT-PCR for HIV RNA, the combination test is of value for diagnosis and for large-scale screening (e.g. blood donors).
PCR
Direct detection methods are required when serological tests are inappropriate, such as during the early acute stage and in infants who still carry maternal anti-HIV, and for monitoring progression.
Both HIV RNA and DNA sequences can be detected in blood. RNA sequences are found in extracellular virus particles in plasma, and the RNA can be accurately quantified as the number of RNA copies to indicate the extent of virus replication in the patient. Measurement of plasma virus load is now essential for monitoring disease progression and the response to antiretroviral therapy. A number of commercial assays have been developed to provide accurate and standardized viral load measurements in clinical laboratories.
HIV proviral DNA is present in infected cells and can be detected in peripheral blood mononuclear cells. This method is used principally to diagnose infection in infants born to HIV-infected mothers. The standard procedure involves analysis of serial blood samples collected at birth, 6 weeks and 3 months. The absence of proviral DNA at 3 months of age (or any time later) excludes HIV infection in babies who are not being breastfed.
Treatment
Currently used multiple drug regimens (previously referred to as highly active antiretroviral therapy, or HAART; now known as combination antiretroviral therapy, or cART) have achieved remarkable success in halting the progression to AIDS, and have helped to transform HIV from a deadly infection to a treatable chronic condition in industrialized countries with access to therapy.
The aim of antiretroviral therapy is to arrest and reverse the damage to the immune system. This will in turn avert the risks of HIV-related clinical problems, reduce infectivity and prolong survival. The decision to commence treatment is dependent on specific clinical and immunological factors (Box 55.3). cART is not recommended, at the present time, in asymptomatic patients who display no clinical or laboratory features of definite or imminent immunodeficiency.
Antiretroviral drugs exert their effects by inhibiting specific steps in the virus replicative cycle that range from virus entry and reverse transcription to integration and protein processing in budding virus (see Ch. 5). To maximize effect and delay or prevent the emergence of viral resistance, three drugs, from at least two different classes, are prescribed together.
The objective of therapy is to achieve continuous suppression of plasma viral RNA to a level below the limits of detection afforded by modern molecular assays, usually around 40 copies/mL. In a patient who has not been treated before, a combination of two nucleoside/nucleotide reverse transcriptase inhibitors (NRTI, e.g. tenofovir/emtricitabine, abacavir/lamivudine, zidovudine/lamivudine) plus either a non-nucleoside reverse transcriptase inhibitor (NNRTI, e.g. efavirenz, nevirapine) or a ritonavir boosted protease inhibitor (PI/r, e.g. darunavir/ritonavir, lopinavir/ritonavir, atazanavir/ritonavir) is currently recommended. The two NRTI agents, often referred to as ‘the backbone’ are preferably given as a co-formulated preparation.
The management of HIV infection can be difficult and is ideally delivered by a specialist team with access to a clinical laboratory. Specific clinical guidelines on timing, components and monitoring of cART are available from many national bodies, including the British HIV Association (BHIVA). The World Health Organization makes similar recommendations for patients in ‘resource-limited settings’, taking account of local conditions such as the need for a cold chain to deliver the drugs to the patients and how they are administered.
A patient on combination therapy will have to adhere to a strict regimen. This may be a problem in very young and adolescent patients. The drugs chosen may have side effects in a particular patient or interactions with therapy for other conditions and infections such as hepatitis B and C viruses, herpesviruses, tuberculosis, toxoplasmosis and P. jirovecii. Therapy to prevent or treat these infections, if indicated, must be maintained.
Knowledge of previous antiretroviral therapy is also essential as drug resistance may have arisen. It is now standard practice to test the patient’s virus population for drug resistance using genotyping methods, before initiation of cART and in situations of virological failure (see below).
Monitoring progress
The CD4 count and the plasma viral load should be assayed when therapy is started and at 1-month and 3–4-month intervals thereafter. If there is a response, the RNA load will decrease within a few days, will drop by 1 log10 at 2–8 weeks and be less than 40 copies/mL by 4–6 months. If these objectives are not achieved, or the viral load increases after a time on therapy, or the clinical state deteriorates, a new combination regimen should be started (assuming patient compliance with the prescribed regimen). The resistance pattern of the patient’s virus should be tested before selecting new drugs. Genotypic resistance testing is accomplished by sequencing of viral target genes and comparing the sequences against an extensive repository of possible resistance mutations such as the Stanford University Drug Resistance Database. The locus and nature of any identified mutations will allow determination of which drug(s) within the combination regimen are failing and therefore rational drug switches/substitution(s) by the attending physician.
It is important to note that drug-related toxicity, a largely unpredictable phenomenon, and potential drug–drug interactions remain significant problems despite the progress made over the past decade in improving drug efficacy and tolerability.
The prophylaxis of perinatal infection and accidental exposure is described below.
Transmission and epidemiology
Virus is present in the blood, semen, and cervical and vaginal secretions, and these sources are important in transmission. Virus may also be present in cerebrospinal fluid, saliva, tears and urine, but at lower titres than in blood. There is no epidemiological evidence that these are significant sources for transmission. Free virus is present at high titre during the early stage of infection and increases in titre in the blood in the later stages of the disease; there is evidence of a greater risk of transmission from such patients.
To transmit, virus has to reach susceptible cells at the point of entry (e.g. Langerhan’s cells in mucous membranes) or after entering the circulation.
The three important routes of transmission of HIV are:
Sexual intercourse
Heterosexual transfer of virus is the route by which the great majority of infections are spread, accounting for 90% of the global total, mostly in the developing world. Overall the estimated risk of transmission from one unprotected exposure is 0.1–0.2% for vaginal intercourse. The probability of transfer is increased if either partner has ulcerative genital or other sexually transmitted disease. Any trauma during intercourse will also facilitate transfer, by allowing direct access of the virus to susceptible cells and the circulation. Transmission may be more likely from male to female.
AIDS was first recognized in men who have sex with men in the USA. Most early studies established that unprotected anal intercourse was a particular risk, especially to the passive, receptive partner. The estimated risk from a single exposure is 0.1–0.3%.
Transmission during oral sexual contact has been documented, but is not a major route.
Mother to child
Most transmission occurs late in pregnancy or during birth (perinatal). The most likely source is cells and virus in the cervix and vagina, as the baby passes through the birth canal. Maternal plasma viral load is the strongest predictor of the risk of virus transmission from mother to infant; the transmission rate is estimated to be 2% or less in mothers with a viral load of less than 1000 copies/mL. Clinical factors known to influence the risk of transmission include, primary or advanced HIV disease, co-infection with other sexually transmitted diseases and prolonged and difficult labour, Breast milk is another source that is responsible for as many as 40% of new HIV infections in infants occurring in the postnatal period.
Blood and blood products
All blood for transfusion and the preparation of products such as factor VIII for haemophiliacs is screened by sensitive assays. This eliminates almost all the risk, but it is important to ask donors about possible exposure to risk. Preparation of blood products from large pools of donations was a major factor in contaminating the product as even one infected donation could introduce virus to all the material. Transplanted organs have been implicated in a few cases.
Intravenous drug use is a risk factor in about one-quarter of patients with AIDS in the USA and to a varying extent elsewhere in the world. The risk rises with the volume of blood injected and the frequency of sharing contaminated equipment. The withdrawal of blood before injection increases contamination. By sharing syringes, the virus can spread very rapidly so that most IV drug users in an area become infected in a few months. Those infected in this way can spread the virus to their sexual partners or children. Drug and sexual routes merge when IV drug users support their habit by prostitution.
Occupational exposure of health-care workers to infected patients has resulted in transmission in a small number of cases. The route is via accidental penetrating injuries with needles and sharps contaminated with blood. The risk from a needlestick is 1 in 200–300; contamination of eyes and mucous membranes has a lower risk of transmission. Transmission from health-care workers to patients has been suspected in only a few cases.
General
The majority of infected individuals have a recognized exposure to a known source of infection. In some this may be difficult to establish, but there is no evidence that HIV can spread by casual contact or inhalation.
Studies of people exposed to the virus on many occasions (e.g. sex workers in sub-Saharan Africa) have shown that a few develop no evidence of infection, and remain negative for anti-HIV. How these individuals are resistant to infection is of great interest in understanding protective immunity.
Epidemiology of HIV
The extent of spread of infection can be measured by the numbers of cases identified clinically and by serological testing. Much more evidence can be obtained from seroprevalence surveys of particular groups or the general population. Surveys have been performed on patients attending hospitals, antenatal clinics, sexually transmitted disease clinics and blood donors. Specific groups such as IV drug users and commercial sex workers can be targeted; noninvasive sampling (e.g. collecting saliva or dried blood spots) may make these studies more feasible. Repeat testing over time will give an indication of the trend of infection in that population. Such studies are important in monitoring the effect of intervention strategies and forecasting the demand for health services.
HIV was isolated in the early 1980s, but the first identified cases date to the 1960s. During the 1970s, the virus began to spread widely in some populations and groups by the routes described above.
The scale of the HIV-1 epidemic is monitored by coordinated surveillance by the Joint United Nations Programme on HIV/AIDS (UNAIDS). For 2009, it is estimated that there were 33.3 million individuals living with HIV worldwide, 2.6 million newly infected individuals and 1.8 million AIDS-associated deaths, of whom 370 000 were children under 15 years of age. Although the global number of annual new infections has declined by 19% in comparison to 1999, owing to the impact of preventative efforts and the natural course of the HIV epidemic, the total number of individuals living with HIV has actually increased by 27% since 1999. This is contributed to by the significant reductions in annual AIDS-associated deaths brought about by the increased availability of antiretroviral drugs and care afforded to infected patients in developing countries. Ominously, over 7000 new HIV infections occur every day, a third of whom are young people between 15 and 24 years old. The social and economic consequences of this epidemic have been devastating, with the loss of parents and wage earners.
Frequencies of HIV infection remain highest in sub-Saharan Africa (with a mean 5% overall population prevalence) where most AIDS-related deaths have occurred. Exceptional effort is clearly required, at all levels, to curtail the ruinous effects of the epidemic in this region. In North and South America, Europe and Australia, at least 30–40% of cases are in men who have sex with men (MSM). Parenteral drug users are the other major risk group in these areas. The other cases are in the heterosexual partners of bisexual men, IV drug users, and men and women infected in other areas of the world. The numbers of infections in risk groups can change as health education programmes are introduced; however, the success of programmes varies and advice may be ignored if the perception of risk changes.
HIV was introduced into South and South-East Asia later than in the rest of the world; infection is spreading rapidly. The earliest infections were in IV drug users, but this did not lead to wide spread outside the risk group. Some 1–10% of antenatal patients may be infected. Without effective intervention large numbers of cases and deaths will occur, with all the expected human and socio-economic consequences. It is of concern that the number of new HIV infections is increasing in Eastern Europe, North Africa and the Middle East.
The existence of different subtypes of HIV, and recombinants, is important for two reasons. Firstly, assays for anti-HIV and viral nucleic acid must be able to recognize all types. Secondly, vaccine developers must take account of the various types and establish the spectrum of protection of candidate vaccines.
The Global Fund to fight AIDS (tuberculosis and malaria), a public/private partnership established following the United Nations’ pivotal Declaration of Commitment on HIV/AIDS in 2001, spearheads the efforts to mobilize and provide funds for national and regional HIV/AIDS programmes. Despite the unprecedented expansion of access to antiretroviral treatment that has occurred since 2001, up to two thirds of people in need of therapy still have no access to the drugs. The United Nations General Assembly adopted a new declaration on HIV/AIDS on 10 June 2011, reaffirming the 2001 commitment and emphasizing the urgent need to scale up significantly the efforts towards the goal of universal access to comprehensive preventative schemes, treatment, care and support. The target of the new strategy is to halt and reverse the spread of HIV by 2015.
Control
Until a vaccine is available, the emphasis in controlling the spread of infection must be on risk reduction. Antiretroviral therapy is expected to play an important part in attempts to contain the spread of HIV-1.
Sexual transmission
The emphasis is on risk reduction by avoiding unprotected penetrative intercourse with partners of unknown status. Despite knowledge of the major routes of infection, there has been only limited success in reducing sexual transmission. Globally the problem is enormous and efforts are hampered by the poverty and lack of resources of the countries worst affected. The use of condoms and vaginal antiseptics could have an impact, but they need to be available and acceptable to the local population.
In the areas of the world with low levels of infection, early efforts to encourage safe practices had an effect on the spread of the virus among MSM in the Americas, Europe and Australia, but this was not always maintained as the perception of the risks changed as a result of declining rates of infection and, more recently, as the latest therapies appeared to be succeeding and prolonging survival. In addition, it is difficult to persuade the heterosexual majority that safe practices are relevant to them.
Male circumcision has been shown to confer a protective effect against HIV in men, possibly through reduction in surface area of disrupted foreskin epithelium teeming with cells permissive for HIV infection, including CD4+ T lymphocytes. Modeling indicates that the population level impact will be greater than the individual level gain if a large proportion of men get circumcised. This may have an impact on HIV prevalence at least in endemic areas where heterosexual transmission is the dominant route.
Mother to child transmission
Overall, around 25% of HIV-infected pregnant women transmit the virus to their infants. Rational strategies, based on improved understanding of disease pathogenesis and availability of better antiretroviral drugs and reliable laboratory monitoring methods, have almost eliminated the risk of mother-to-child transmission (MTCT) in high income nations. The main components of current recommendations are universal antenatal HIV testing to identify infected mothers, use of cART to suppress plasma viral load, planning caesarian delivery when indicated and avoidance of breast feeding. Although low cost drugs have increasingly become available, the above strategy is not entirely applicable in low income countries where most MTCT occurs.
The limited therapy available in resource-poor countries such as sub-Saharan Africa can also still play a major preventive role although better approaches are urgently needed. Zidovudine alone given to the mother before delivery and to the baby for 6 weeks can reduce transmission three-fold even if the mother has advanced disease and single doses of nevirapine given to the mother at the onset of labour and to the neonate within the first 3 days after birth reduce the rate of transmission by more than 50%. However, several concerns, including recruitment of drug-resistant viral strains and increased risk of perinatal transmission or progression in infected babies, remain.
Breast-feeding is another possible route of transmission. Exclusive bottle-feeding may reduce risk of virus transmission however that may occur at the expense of increased infant mortality from diarrheal and respiratory infection in developing countries. Where alternative nutrition to breast milk is available, avoidance of breast-feeding is sensible.
Exposure to blood
Drug injectors can avoid risk by not injecting, or can reduce risk by using only clean equipment. Screening of all blood donors should eliminate almost all possibility of transmission through receipt of blood transfusion. Factor VIII and other blood products are heat treated, if possible, to inactivate HIV. All organ donors must be screened.
Occupational risk in the health-care setting can be controlled by the implementation of safe working practices to prevent accidental injury and contamination with blood and body fluids. The use of gloves, masks and eye protection is important in situations such as surgical procedures where bleeding and spattering are possible. The risk must be assessed in other situations. Safe disposal of used needles, scalpel blades and other sharps is an essential requirement. The sensitivity of HIV to heat and various disinfectants is described below.
• heat, in an autoclave or hot-air oven
• hypochlorite (10 000 ppm); 1 in 10 dilution of domestic bleach
The chemicals will inactivate at least 105 units of virus within a few minutes, but disinfectants are inactivated in the presence of organic material.
HIV can survive for up to 15 days at room temperature and for 10–15 days at 37°C. At temperatures greater than 60°C, virus is inactivated 100-fold each hour.
If an accidental exposure occurs, any wound should be washed with soap and water, or mucous membranes flushed with water. The accident must be reported so that, if necessary, post-exposure prophylaxis (PEP) can be started as soon as possible. The risk must be assessed through knowledge of the circumstances:
• The HIV status of the source patient; if unknown, can the source be tested?
• Any particular risk of infection of the source patient.
• The nature of the exposure (e.g. penetrating injury or contamination of skin or mucous membranes).
The risk of infection from splashing on to mucous membranes or skin is hard to quantify, but is certainly less than with penetrating injuries. An intact skin is an effective barrier, but abrasions and diseases such as eczema may impair this protection.
If a sharp injury is reported the nature of the injury has to be assessed.
If there is an indication of risk, PEP must be started within 1–2 h, and not later than 48–72 h. If no professional advice is available, for instance at night, prophylaxis should be started and advice obtained. A decision should made about continuing with the drugs preferably within 12–24 h. The victim should be involved in the decision, with discussion of the risks and the possible side effects of the drugs.
Zidovudine alone can reduce the transmission rate, but should now be combined with another reverse transcriptase inhibitor (e.g. lamivudine) and a protease inhibitor. Alternative triple agent combinations can be used or specifically selected with knowledge of any drug resistance in the source. Therapy should be continued for 4 weeks and the victim followed with testing for virus for the next 6 months. A few cases of transmission have been seen in cases despite appropriate PEP.
Vaccines
Much effort has been devoted to the development of a vaccine to provide protection against infection (prophylactic vaccine) or to boost the immune system of those infected (therapeutic vaccine). Major problems arise because of the antigenic variability of HIV and the difficulty of developing immunogens that elicit protective responses to all variants. In addition, HIV may be transferred by blood-borne or mucosal routes, through transfer of free virus or infected cells. To protect, therefore, it is likely that both cell-mediated and humoral responses need to be stimulated. Whether an HIV vaccine could ever induce fully protective immunity is subject to some doubt, because the immune response, although highly active during acute infection, is never capable of fully clearing infection, and life-long persistence is the norm.
Most efforts have been directed to the development of vaccines containing the viral Env proteins gp160, gp120 or gp41 prepared by recombinant DNA cloning and expression, or synthetic peptides known to be important epitopes for induction of neutralizing antibodies. To date, human trials have shown no evidence of protection from infection by sexual transmission and injecting drug use.
HTLV-I and II infection
Clinical features
Primary HTLV-I infection is not associated with a recognizable clinical syndrome or seroconversion illness. Like HIV, up to 8 weeks may elapse following initial exposure for antibody to become detectable. Seroconversion is followed by an asymptomatic period that can last from years to decades. Clinical disease may eventually develop as a direct result of cell transformation by the virus, in 1–4% of cases (adult T-cell leukaemia/lymphoma, ATL), or as a manifestation of immunological responses to it, in 1–2% (human T-cell lymphotropic virus-associated myelopathy, HAM; or tropical spastic paraparesis, TSP).
ATL was first recognized in Japan. It is an aggressive T cell proliferative malignancy; the features are leukaemia, generalized lymphadenopathy and hepatosplenomegaly, skin lesions, and metabolic disorders, especially hypercalcaemia (acute ATL). A distinct, aggressive T-cell lymphoma clinical type has also been identified (lymphoma/leukaemia ATL). The two forms have poor prognosis with median survival time of 6.2 and 10 months, respectively. Other less dramatic forms exist, including a variant that runs a slow course, associated with adenopathy and splenomegaly (chronic ATL) and an indolent form with skin lesions but no visceral involvement (smoldering ATL). Males are at greater risk than females of developing ATL. The period of latency until ATL arises lasts for many years, often decades. The T cells involved carry the CD4 antigen.
HTLV-I is also the cause of HAM/TSP, a slowly progressive myelopathy with spastic or ataxic features. Pathologically, areas of demyelination with lymphocytic inflammation and perivascular cuffing are seen.
There are several other recognized virus-associated diseases, notably a form of uveitis in otherwise asymptomatic carriers and infective dermatitis in children born to HTLV-I positive mothers.
HTLV-II has not conclusively been shown to cause a particular disease however evidence showing a link to HAM/TSP and possibly other neurologic manifestations is accumulating.
Pathogenesis
During the latent period viral proteins are expressed, as there are steady high antibody titres to various proteins, particularly the gag proteins. The virus is genetically stable and little cell-free virus is produced. However, during the latent period, virus is present as integrated provirus and is replicated with the cellular DNA as the cell divides. The tumour cells contain monoclonally integrated HTLV-I provirus at random sites. There are no transforming genes. The T cell proliferation is the result of the action of the viral tax gene, which can activate transcription of cellular genes including those for interleukin-2 and its receptor, and cause cell proliferation. It is not known what triggers this effect after the long latency in the 1–4% of those infected who develop disease. Antibody to the tax protein can block the stimulation of cell division; loss or decay of immune control may be important. Interactions with tumour-suppressor genes such as p53 and promotion of S-phase in the cell cycle are other probable mechanisms of oncogenesis.
Laboratory diagnosis
Current serological assays, incorporating recombinant or synthetic viral peptides, for the detection of antibody to HTLV-I and HTLV-II are highly sensitive and specific. As with HIV, confirmation is achieved by other assays or immunoblotting (e.g. Western blot) although interpretation can be difficult. Confirmatory tests are able to distinguish between antibodies to HTLV-I and HTLV-II. Detection of HTLV proviral sequences by PCR can also be used as a confirmatory test, and to distinguish between types.
Treatment
There is no indication for treatment of asymptomatic HTLV carriers. ATL patients are treated with conventional anti-cancer chemotherapy or, when appropriate, haematopoietic stem cell transplantation. Interferon and inhibitors of reverse transcriptase (e.g. zidovudine) and lamivudine may have a complementary role in treatment of HTLV-related diseases but further evaluation is needed.
Transmission and epidemiology
There are three lineages of HTLV-I strains, which are linked to Melanesia, Central Africa and various countries (the Cosmopolitan group). The latter includes viruses from Japan, North and West Africa and the Caribbean, which can be distinguished. HTLV-I and the simian virus, STLV-I, are closely related and it is proposed that human infection occurred many thousands of years ago in Africa and that the presence of the virus in many different parts of the world is related to the migration of ancient peoples. The slave trade may account for foci found in the West Indies and the southern USA.
The virus is endemic in certain communities. In parts of Japan, the prevalence of antibody can be as high as 27%, with a rising trend from 7–8% in the 20–39-year age group to 52% in females and 32% in males by 80 years. In the Caribbean, the rates are in the range of 5–10%, with clusters in communities and families. In other regions, infection has been found in parenteral drug users and sex workers.
The virus is cell associated in the host, so transmission will occur when infected cells are transferred. This can occur during sexual intercourse, blood transfusion and through sharing injecting equipment by IV drug users. In contrast to HIV, breast-feeding appears to be the dominant route of mother-to-child transmission; maternal retroviral load is the major predictor of transmission. HTLV-II is transmitted by the same routes. The strains found in IV drug users in different countries are related.
Pillay D, Geretti AM, Weiss RA. Human immunodeficiency viruses. In: Zuckerman AJ, Banatvala JE, Schoub BD. Principles and Practice of Clinical Virology. ed 6. Chichester: Wiley-Blackwell; 2009:897–938.
Taylor GP. The human T-lymphotropic viruses. In: Zuckerman AJ, Banatvala JE, Schoub BD. Principles and Practice of Clinical Virology. ed 6. Chichester: Wiley-Blackwell; 2009:875–896.
UK Health Departments. Guidance for Clinical Health Care Workers; Protection Against Infection with Blood-Borne Viruses. Recommendations of the Expert Advisory Group. London: HMSO; 1998.
Volberding PA, Deeks SG. Antiretroviral therapy and management of HIV infection. The Lancet. 2010;376:49–62.
British HIV Association (BHIVA). http://www.bhiva.org/ClinicalGuidelines.aspx.
Joint United Nations Programme on HIV/AIDS (UNAIDS). http://www.unaids.org/en/dataanalysis/.
Stanford University Drug Resistance Database. http://hivdb.stanford.edu/.