19 Environmental mycobacteria
Opportunist disease
Key points
• Environmental or non-tuberculous mycobacteria (about 130 species) are divisible into slow and rapid growers. Human disease is often opportunistic (e.g. in AIDS and chronic pre-existing lung disease) and attributable to slow growers, notably members of the M. avium complex.
• Infection is from the environment, and transmission control methods applied to tuberculosis are ineffective.
• Presentations include localized lymphadenitis, post-inoculation (injection or trauma) skin lesions, tuberculosis-like pulmonary lesions, solitary non-pulmonary lesions and disseminated disease.
• Lymphadenitis occurs in otherwise healthy young children (<6 years of age) and in immunosuppressed persons.
• Skin diseases include swimming pool granuloma (M. marinum), Buruli ulcer (M. ulcerans) and post-inoculation abscesses caused by various rapid growers.
• Diagnosis is based on a range of simple tests or by molecular techniques including DNA probes or specific PCR primers. Repeated detection in the face of progressive disease assists in distinguishing pathogenic association from contamination or colonization.
• Therapy depends on the causative organism and is largely empirical as few clinical trials have been conducted and drug susceptibility tests are not very reliable.
In addition to the tubercle and leprosy bacilli there are more than 130 recognized species of mycobacteria in the List of Prokaryotic names with Standing in Nomenclature that normally exist as saprophytes of soil and water. Termed environmental (or non-tuberculous) mycobacteria, some of these species occasionally cause opportunist disease in animals and man. Those species described here are well-recognised human pathogens but many of the other species are rare causes of disease, especially in those with HIV disease and other causes of profound immunosuppression. Mycobacterial species are divided into photochromogens, which develop yellow or orange pigmentation in (or after exposure to) light, scotochromogens, which become pigmented in the dark, and non-chromogens. They are also divided into slow and rapid growers, with the latter species producing visible growth on Löwenstein–Jensen medium within 1 week on subculture, although growth on primary culture of clinical material often takes considerably longer. Traditionally, mycobacterial species were characterised by a range of physical and biochemical properties but molecular methods, notably determination the base sequence of the 16S ribosomal RNA, provide the ‘gold standards’ for speciation.
Description
Photochromogens
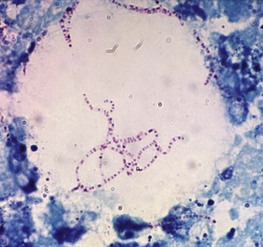
• Mycobacterium kansasii, which grows well at 37°C and is isolated principally from patients with pulmonary disease. On microscopy, it often appears elongated and distinctly beaded (Fig. 19.1).
• M. simiae, which, like M. kansasii, grows at 37°C and is occasionally involved in pulmonary disease.
• M. marinum, the cause of a warty skin infection known as swimming pool granuloma or fish tank granuloma. It grows poorly, if at all, at 37°C, and cultures from skin lesions should be incubated at 33°C. Microscopically, it resembles M. kansasii.
Scotochromogens
Most slowly growing scotochromogens isolated from sputum or urine are of no clinical significance. These include M. gordonae (formerly M. aquae), which is frequently found in water and is a common contaminant of clinical material. It is, however, a rare cause of pulmonary disease. Two species are more usually associated with disease:
Non-chromogens
The most prevalent and important opportunistic pathogens are in a group known as the M. avium complex (MAC). This group consists of:
• M. avium avium (the avian tubercle bacillus), a pathogen of birds and also a cause of lymphadenitis in pigs as well as occasional disease in various other wild and domestic animals.
• M. avium intracellulare, principally a human pathogen but also a cause of disease in animals including pigs. Many isolates formerly included in this species are now identified as M. avium hominissuis, although this term has not been formally published.
• M. avium paratuberculosis, the cause of chronic hypertrophic enteritis or Johne’s disease of cattle. There have been claims, which remain to be substantiated, that it is a cause of Crohn’s disease in man. It produces little or no mycobactin, a lipid-soluble iron-binding compound essential for growth, and this substance must be added to media used for cultivation.
• M. avium sylvaticum, a cause of tuberculosis in wood pigeons. Like M. paratuberculosis it needs mycobactin for growth.
• M. avium lepraemurium, the cause of a leprosy-like disease of rats, mice and cats.
Both M. avium hominissuis and M. avium intracellulare are causative agents of lymphadenitis and pulmonary disease. They are also a cause of disseminated disease in profoundly immunosuppressed patients, notably in those with the acquired immune deficiency syndrome (AIDS). Most clinical isolates are smooth and easy to emulsify and were previously typed by agglutination serology although RFLP and MIRU-VNTR (see Ch. 18) are now used.
• M. ulcerans, a very slowly growing species that grows in vitro only at 31–34°C. Colonies are non-pigmented or a pale lemon-yellow colour. M. ulcerans is the cause of Buruli ulcer; unlike other mycobacterial pathogens it produces a toxin that causes tissue necrosis and is involved in the pathogenesis of the disease. M. shinshuense, a very rare cause of skin ulcers in Japan and China, is a variant of M. ulcerans.
• M. xenopi, originally isolated from a xenopus toad, is a thermophile that grows well at 45°C. It is principally responsible for pulmonary lesions in man. Most reported cases have been from London and south-east England and northern France. Two phylogenetically similar species, M. celatum and M. branderi, have been described.
• M. malmoense is a cause of pulmonary disease and lymphadenitis. It grows very slowly, often taking as long as 10 weeks to appear on primary culture, and is therefore likely to be missed if cultures are discarded earlier. For unknown reasons, disease due to this pathogen is increasing in several European countries. In some regions, notably northern England, it has become one of the most frequently isolated of the environmental mycobacteria from patients with pulmonary disease.
• M. terrae (the radish bacillus), M. nonchromogenicum and M. triviale are sometimes grouped as the M. terrae complex. They are very rare causes of pulmonary disease and, in the case of M. terrae, infections of injuries acquired while farming or gardening.
• M. haemophilum, characterized by its growth requirement for haem or other sources of iron, is a rare cause of granulomatous or ulcerative skin lesions in xenograft recipients and other immunocompromised individuals, and of lymphadenitis in otherwise healthy children.
• M. genevense, a very slow growing organism occasionally isolated from patients infected with HIV or with other causes of immunosuppresion, from pet and zoo birds and, rarely, other animals.
Rapid growers
Four rapidly growing non-chromogenic species are well-recognized human pathogens: M. chelonae, M. abscessus, M. fortuitum and M. peregrinum. They occasionally cause pulmonary or disseminated disease but are principally responsible for post-injection abscesses and wound infections, including corneal ulcers.
Most of the many other rapidly growing species are pigmented and, although disease due to them is exceedingly rare, they frequently contaminate clinical specimens. They are found on the genitalia and gain access to urine samples, although, contrary to a common belief, M. smegmatis is rarely found in this site. M. flavescens, which grows more slowly than other members of this group and is therefore sometimes classified as a slow grower, is an occasional cause of post-injection abscesses.
Pathogenesis
Compared with tubercle bacilli, environmental mycobacteria are of low virulence and, although human beings are in regular contact with these organisms and therefore frequently infected, overt disease is uncommon except in those who are profoundly immunosuppressed. Five main types of opportunist mycobacterial disease have been described in man (Table 19.1):
2. skin lesions following traumatic inoculation of bacteria
3. tuberculosis-like pulmonary lesions
Table 19.1 Principal types of opportunist mycobacterial disease in man, and the usual causative agents
| Disease | Usual causative agent |
|---|---|
| Lymphadenopathy | M. aviurn complex |
| M. scrofulaceum | |
| Skin lesions | |
| Post-traumatic abscesses | M. chelonae |
| M. abscessus | |
| M. fortuitum | |
| M. peregrinum | |
| Swimming pool granuloma | M. marinum |
| Buruli ulcer | M. ulcerans |
| Pulmonary disease and solitary non-pulmonary lesions | M. aviurn complex |
| M. kansasii | |
| M. xenopi | |
| M. malmoense | |
| Disseminated disease | |
| AIDS related | M. aviurn complex |
| M. genevense | |
| Non-AIDS related | M. aviurn complex |
| M. chelonae | |
| M. abscessus |
Lymphadenitis
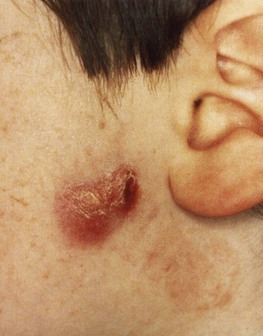
This is caused by a number of different species that vary in relative frequency from region to region. The M. avium complex is the predominant cause worldwide. Earlier reports claimed a high incidence of M. scrofulaceum, but these strains were probably misidentified members of the M. avium complex. In most cases a single node, usually tonsillar or pre-auricular, is involved, and most patients are children aged less than 5 years (Fig. 19.2). Unless contra-indicated by the risk of nerve damage, excision of the node, usually performed for diagnostic purposes, is almost always curative. This disease was very rare where children were vaccinated with BCG (Bacille Calmette–Guérin) neonatally, but the incidence increased considerably in countries where such vaccination was terminated. Lymphadenitis occasionally occurs as part of a more disseminated infection, particularly in individuals with AIDS.
Skin lesions
Three main types have been described: post-injection (and post-traumatic) abscesses, swimming pool granuloma and Buruli ulcer.
Post-injection and post-traumatic abscesses
These are usually caused by the rapidly growing pathogens M. abscessus, M. chelonae, M. fortuitum and, less frequently, M. peregrinum and M. flavescens. Abscesses occur sporadically, particularly in the tropics, or in small epidemics when these bacteria contaminate batches of injectable materials. Abscesses develop within a week or so, or for up to a year or more, after the injection. They are painful, may become quite large – up to 8 or 10 cm in diameter – and may persist for many months. Treatment is by drainage with curettage or total excision. Chemotherapy is not required unless there is local spread of disease or multiple abscesses, as may occur in insulin-injecting diabetics.
More serious lesions, also usually due to these rapidly growing pathogens, have followed surgery, particularly procedures involving insertion of prostheses such as heart valves, and corneal infections have followed ocular injuries or surgery (Fig. 19.3). Infections by M. terrae have occurred in farmers and others who have been injured while working with soil.
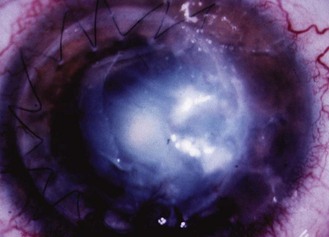
Fig. 19.3 Keratitis due to M. chelonae following corneal grafting. The cornea shows a characteristic ‘cracked windscreen’ appearance.
Reproduced by permission of Elsevier from Khooshabeh R, Grange JM, Yates MD, McCartney ACE, Casey T A 1994 A case report of Mycobacterium chelonae keratitis and a review of mycobacterial infections of the eye. Tubercle and Lung Disease 75: 377–382.
Swimming pool granuloma
This is also known as fish tank granuloma and, occasionally, fish fancier’s finger, and is caused by M. marinum. Those affected are mostly users of swimming pools, keepers of tropical fish and others involved in aquatic hobbies. The bacilli enter scratches and abrasions, and cause warty lesions similar to those seen in skin tuberculosis. The lesions, which usually occur on the knees and elbows of swimmers and on the hands of aquarium keepers, are usually localized, although secondary lesions sometimes appear along the line of the dermal lymphatics. This is termed sporotrichoid spread as a similar phenomenon occurs in the fungus infection sporotrichosis (see Ch. 61). A few cases of disseminated disease have occurred in immunosuppressed patients, including those treated with systemic steroids.
Buruli ulcer
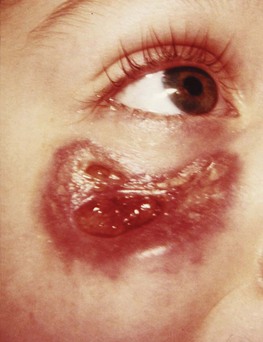
This disease, caused by M. ulcerans, was first described in Australia, although the name is derived from the Buruli district of Uganda where a large outbreak was investigated extensively in the 1960s. Buruli ulcer has been reported in over 30 countries including Ghana, Nigeria, Congo Kinshasa, Côte d’Ivoire, Togo, Mexico, Malaysia, Papua New Guinea and Australia, and is limited to certain localities, characteristically low-lying areas subject to periodic flooding. M. shinshuense, a rare cause of similar lesions in Japan and China, is a variant of M. ulcerans. There is strong though indirect evidence that M. ulcerans is introduced from the environment into the human dermis by minor injuries, particularly by spiky grasses and, possibly, by biting insects. It is becoming a major public health problem in central and western equatorial Africa, where the number and severity of cases has increased; in some areas, its incidence is exceeding that of tuberculosis and leprosy. The risk of the disease is higher in those infected with HIV, in whom the disease may be more aggressive, but this is not the sole cause of the increase.
The first manifestation of the disease is a hard cutaneous nodule, which may be itchy. The nodule enlarges and develops central softening and fluctuation owing to necrosis of the subcutaneous adipose tissue caused by an exotoxin, which also has immunosuppressive properties. The overlying skin becomes anoxic and breaks down, the liquefied necrotic contents of the lesion are discharged, and one or more ulcers with deeply undermined edges are thereby formed (Fig. 19.4). At this stage the lesion is teeming with acid-fast bacilli and there is no histological immunological evidence of an active cell-mediated immune response. During this anergic stage the lesion may progress to an enormous size, sometimes involving an entire limb or a major part of the trunk.
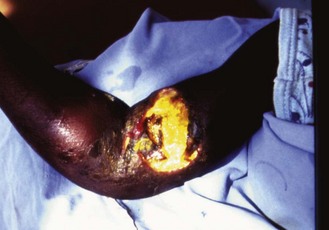
Fig. 19.4 Buruli ulcer. Undermined ulcer overlying the biceps and swelling of the surrounding tissues.
Courtesy of Dr Alan Knell, Wellcome Tropical Institute.
For unknown reasons, the anergic phase may eventually give way to an immunoreactive phase when a granulomatous response develops in the lesion, the acid-fast bacilli disappear and immune reactivity to antigens of M. ulcerans is detectable. Healing then occurs but the patient is often left with considerable disfigurement and disability caused by extensive scarring and contractures.
Pulmonary disease
This is seen most frequently in middle-aged or elderly men with lung damage caused by smoking or exposure to industrial dusts. It also occurs in individuals with congenital or acquired immune deficiencies, malignant disease and cystic fibrosis, but a substantial minority of cases occur in persons with no apparent underlying localized or generalized disorder. The disease may be caused by many species, although the most frequent are the M. avium complex and M. kansasii. Other causative organisms in the UK are M. xenopi and M. malmoense, with the former being more common in the south of the country and the latter in the north.
Localized non-pulmonary disease
Localized non-pulmonary lesions resembling those seen in tuberculosis are uncommon. Disease involving the meninges, bone (including the spine) and joints, kidney and male genitalia has been described. Mycobacterial peritonitis is a serious complication of peritoneal dialysis.
Disseminated disease
In the 1980s and early 1990s, up to a half of all persons dying from AIDS in the USA had disseminated mycobacterial disease, almost always due to the M. avium complex. This AIDS-related disease was also common in Europe but not in Africa. The introduction of highly active antiretroviral therapy (see Ch. 55) has led to a substantial decline in the incidence of this disease in developed nations.
The source of the causative organisms is uncertain. Some workers consider that disease is due to reactivation of dormant foci of infection acquired in childhood, whereas others argue that it results from recent infection from the environment; these explanations are not mutually exclusive. Acid-fast bacilli are readily isolated from bone marrow aspirates, intestinal biopsies, blood and faeces. A few cases of AIDS-related disseminated disease have been caused by other species, including M. genavense.
Disseminated disease occurs occasionally in individuals with other congenital or acquired causes of immuno-suppression, including renal transplantation. Again, the M. avium complex is the usual cause, but disseminated disease due to other infections, notably M. chelonae, has occurred in recipients of renal transplants and in other immunocompromised patients (Fig. 19.5).
Laboratory diagnosis
Most environmental mycobacteria can be detected microscopically and cultured on media suitable for M. tuberculosis (see p. 212). Great care must be taken to differentiate true disease from transient colonization or superinfection. In particular, there are no clinical or radiological characteristics that reliably differentiate pulmonary disease caused by opportunist mycobacteria from tuberculosis, and the diagnosis is therefore made by isolation and identification of the pathogen. As a general rule, a diagnosis of opportunist mycobacterial disease may be made when a heavy growth of the pathogen is repeatedly isolated from the sputum of a patient with compatible clinical and radiological features, and in whom other causes of these features have been carefully excluded. Fibreoptic bronchoscopy is a useful aid to diagnosis as lesions may be directly biopsied.
Identification is usually undertaken by specialist reference laboratories. There is no universally recognized identification scheme, although reliance is usually placed on cultural characteristics (rate and temperature of growth and pigmentation), various biochemical reactions and resistance to antimicrobial agents. Some reference centres use HPLC to delineate the chromatographic profile of cell wall mycolic acids and some use nucleic acid-based methods including commercially available PCR primers and DNA probes and sequencing of the 16S ribosomal DNA.
Treatment
Most slowly growing environmental mycobacteria are resistant to many antituberculosis drugs in vitro, although infections often respond to various combinations of these drugs. Few controlled trials of therapy for pulmonary disease caused by the M. avium complex and other slowly growing species including M. xenopi, M. kansasii and M. malmoense have been conducted, but the available data indicate that a two-year regimen of rifampicin and ethambutol with addition of either clarithromycin or ciprofloxacin, or both if there is no clinical response after one year, is highly effective.
Similar drug regimens based on a macrolide (clarithromycin or azithromycin) and ethambutol are effective against AIDS-associated disease caused by the M. avium complex. There is, however, evidence that reduction of the viral load by use of antiretroviral agents contributes more to remission of such disease than antibacterial therapy.
Pulmonary and non-pulmonary disease due to the rapidly growing species M. abscessus, M. chelonae, M. fortuitum and M. peregrinum, have been treated successfully by various combinations of erythromycin, newer macrolides, sulphonamides, trimethoprim, amikacin, gentamicin, imipenem, extended-spectrum cephalosporins and fluoroquinolones. In-vitro susceptibility tests are not always a reliable guide of clinical response and some infections, notably keratitis due to M. chelonae, frequently relapse and require surgical intervention, despite in-vitro drug susceptibility.
Mycobacterium marinum disease is usually self-limiting, although chemotherapy with minocycline, co-trimoxazole or rifampicin with ethambutol hastens its resolution and surgical debridement is required if the disease involves joints or tendons. More aggressive antimicrobial therapy, usually based on rifampicin, ethambutol and clarithromycin is required for extensive and disseminated disease.
The early, pre-ulcerative, lesions of Buruli ulcer are easily treatable by excision and primary closure by suture. Ulcerated lesions require excision and skin grafting. After disappointing early results, antibiotic therapy has been shown to be effective in the majority of treated patients. The World Health Organization recommends 4 weeks of treatment with rifampicin and streptomycin, then surgery followed by 8 more weeks of antibiotic therapy, depending on clinical response.
Epidemiology
Mycobacteria are widely distributed in the environment and are particularly abundant in wet soil, marshland, streams, rivers and estuaries. Some species, such as M. terrae, are found in soil whereas others, including M. marinum and M. gordonae, prefer free water. Many species, including potential pathogens such as the M. avium complex, M. kansasii and M. xenopi, are able to colonize piped water supplies. Human beings are therefore regularly exposed to mycobacteria as a result of drinking, washing, showering and inhalation of natural aerosols. Such repeated subclinical infection may induce sensitization to tuberculin and other mycobacterial skin-testing reagents. There is also evidence that contact with environmental mycobacteria profoundly affects the subsequent ability of BCG vaccine to induce protective immunity, thereby explaining the great regional differences in the efficacy of this vaccine (see p. 220).
The number of cases of disease due to environmental mycobacteria relative to cases of tuberculosis increases in regions where the latter is uncommon and declining in incidence. In addition, the absolute incidence is increasing as a result of the growing number of immunocompromised individuals, notably patients with AIDS.
A few ‘epidemics’ of falsely diagnosed mycobacterial pulmonary disease and urinary tract infection have resulted from the collection of sputum and urine specimens in containers rinsed out with water from taps colonized by mycobacteria. Inadequate cleaning of endoscopes has also led to mycobacterial contamination of clinical specimens and diagnostic confusion. Likewise, false-positive sputum smear examinations for acid-fast bacilli have occurred when staining reagents were prepared from contaminated water.
Control
The incidence and type of disease in any region are determined by the species and numbers of mycobacteria in the environment and the opportunities for human transmission. Unlike tuberculosis, person-to-person transmission of opportunist mycobacterial disease rarely, if ever, occurs. Thus the incidence of such disease is independent of that of tuberculosis and unaffected by public health measures designed to control the latter.
Asiedu K, Scherpbier R, Raviglione M. Buruli Ulcer. Mycobacterium ulcerans Infection. Geneva: World Health Organization Global Buruli Ulcer Initiative; 2000.
Banks J, Campbell IA. Environmental mycobacteria. In: Davies PDO, Barnes PF, Gordon SB. Clinical Tuberculosis. ed 4. London: Hodder Arnold; 2008:509–518.
Daley CL, Heifets L. Other mycobacteria causing human disease. In: Schaaf HS, Zumla AI. Tuberculosis. A Comprehensive Clinical Reference. London: Saunders Elsevier; 2009:60–74.
Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. American Journal of Respiratory and Critical Care Medicine. 2007;175:367–416.
Jenkins PA, Campbell IA, Banks J, et al. Clarithromycin vs ciprofloxacin as adjuncts to rifampicin and ethambutol in treating opportunist mycobacterial lung diseases and an assessment of Mycobacterium vaccae immunotherapy. Thorax. 2008;63:627–634.
Pfyffer GE, Inderlied CB. Mycobacteria. In: Cohen J, Opal SM, Powderley WG. Infectious Diseases. ed 3. London: Mosby Elsevier; 2010:1777–1800.
van der Werf TS, Stienstra Y Johnson RC, et al. Mycobacterium ulcerans disease. Bulletin – World Health Organization. 2005;83:785–791.
World Health Organization. Buruli ulcer: progress report, 2004–2008. Weekly Epidemiological Record. 2008;83:145–154.
Chest Medicine On-line. Environmental mycobacteria. http://priory.com/cmol/envmyco1.htm.
List of Prokaryotic names with Standing in Nomenclature. http://www.bacterio.cict.fr/.
World Health Organization. Buruli ulcer. http://www.who.int/buruli/en/.