25 Shigella
Bacillary dysentery
Key points
• Shigella species cause bacillary dysentery.
• The infective dose is very small.
• Sh. dysenteriae type 1 produces a toxin that resembles the Verocytotoxin of certain strains of E. coli and is responsible for the most serious forms of shigellosis.
• Sh. flexneri, Sh. boydii and Sh. sonnei cause enteric disease of varying severity.
• Sonnei dysentery is the most prevalent form of shigellosis in developed countries.
• Most cases of shigellosis do not require antibiotics. Treatment with ciprofloxacin is indicated in severe cases. Ampicillin, tetracyclines and trimethoprim are suitable alternatives, but resistance is common.
Dysentery, the bloody flux of biblical times, is a clinical entity characterized by the frequent passage of bloodstained mucopurulent stools. Aetiologically it is divisible into two main categories, amoebic and bacillary. Both forms are endemic in most countries with a warm climate. Bacillary dysentery, caused by members of the genus Shigella, is also prevalent in many countries with temperate climates.
Description
The genus Shigella is subdivided on biochemical and serological grounds into four species:
Strains of Shigella spp. are typical members of the Enterobacteriaceae and are closely related to the genus Escherichia. Studies focusing on the ancestral lineage of these bacteria suggest that Sh. boydii is related only distantly to other members of the genus Shigella, which are more closely related to enteroinvasive serotypes of E. coli (see p. 285).
Microscopically and culturally shigellae are indistinguishable from other enteric Gram-negative bacilli. They are non-motile, non-capsulate and appear not to express fimbriae. Strains of Shigella spp. share lipopolysaccharide (LPS) antigens with strains of E. coli but the LPS structures are distinct and merely share common epitopes.
The distinction between strains of Shigella spp. and E. coli depends on a limited number of diagnostic tests including motility, production of lysine decarboxylase and the utilization of citrate. Sh. dysenteriae type 1 produces Shiga toxin and this differentiates it from other members of the genus.
All strains express LPS somatic antigens which form the basis for the Shigella serotyping scheme. There are 13 serotypes of Sh. dysenteriae of which type-1 is much the most important as a cause of severe bacillary dysentery. Sh. boydii can be subdivided into 18 specific serotypes. The six serotypes of Sh. flexneri can each be further subdivided into subtypes. Strains of Sh. sonnei are serologically homogeneous, and a variety of other markers such as the ability to produce specific colicines, the carriage of drug resistance or other plasmids, or lysogeny by a panel of bacteriophages are used to discriminate between strains for epidemiological purposes.
Clinical features
The incubation period is usually between 2 and 3 days, but may be as long as 8 days. The onset of symptoms is usually sudden, and frequently the initial symptom is abdominal colic. This is followed by the onset of watery diarrhoea, and in all but the mildest cases this is accompanied by fever, headache, malaise and anorexia. Many episodes resolve at this point, but others progress to abdominal cramps, tenesmus and the frequent passage of small volumes of stool, predominantly consisting of bloody mucus. The symptoms typically last about 4 days, but may continue for 14 days or more. Infection may affect the nervous system resulting in seizures and encephalitis. Shigellosis is occasionally associated with the development of Reiter’s syndrome (reactive arthritis).
The severity of the clinical illness is to some extent associated with the species involved. Infection with Sh. dysenteriae 1 is usually associated with a severe illness in which prostration is marked and, in young children, may be accompanied by febrile convulsions. Members of the Sh. flexneri and Sh. boydii groups may also cause severe illness. In contrast, dysentery associated with Sh. sonnei (Sonnei dysentery) in an otherwise healthy person may be confined to the passage of a few loose stools with vague abdominal discomfort, and the patient often continues at school or work.
Strains of Sh. dysenteriae 1 have been responsible for many cases of the haemolytic uraemic syndrome that accompanies outbreaks of dysentery in several countries. The condition, with its triad of haemolytic anaemia, thrombocytopenia and acute renal failure, can be caused by many pathogens (in particular, E. coli O157; see p. 286). It is associated with complement activation and disseminated intravascular coagulation, and in some parts of the world is one of the most common forms of acute renal failure in children.
Death from bacillary dysentery is uncommon in the developed world; it occurs mostly at the extremes of life or in individuals who are suffering from some other disease or debilitating condition.
Pathogenesis
Shigella spp. are pathogens of man and other primates, and the pathogenesis of infection with these bacteria and enteroinvasive E. coli (EIEC; see p. 285) is very similar. The infective dose is small: bacillary dysentery may follow the ingestion of as few as ten viable bacteria. The site of infection is the M cells in the Peyer’s patches of the large intestine. Strains of Shigella spp. are non-motile and it is not known how the bacteria reach and adhere to M cells.
Association with the intestinal mucosa initiates mucosal inflammation leading to apoptosis, which is thought to facilitate the invasion of the M cells, after which the bacteria are phagocytosed. The shigellae multiply within the epithelial cells and spread laterally into adjacent cells and deep into the lamina propria. The infected epithelial cells are killed, and the lamina propria and submucosa develop an inflammatory reaction with capillary thrombosis. Patches of necrotic epithelium are sloughed and ulcers form. The cellular response is mainly by polymorphonuclear leucocytes, which can be seen readily on microscopic examination of the stool, together with red cells and sloughed epithelium.
Dysentery bacilli rarely invade other tissues. Transient bacteraemia can occur but septicaemia with metastatic infection is rare.
Pathogenic mechanisms
In common with most bacteria, shigellae require a range of pathogenic mechanisms to cause disease. They are tolerant to the conditions of low pH encountered in the human stomach and the action of bile.
Pathogenic strains of shigellae, like entero-invasive E. coli, carry a plasmid of 100–140 MDa, which encodes the pathogenic mechanisms involved with eukaryotic cell invasion. Expression of the mechanisms encoded on the virulence plasmid are thermoregulated such that strains become invasive when growing at 37°C but not at 30°C, and regulation is by both plasmid and chromosomally located elements. Plasmid-encoded proteins are required for bacteria to break free from cellular endosomes and for the migration between epithelial cells.
Long-chain lipopolysaccharide plays a role in virulence by preventing the effects of serum complement. The lipid A component has been implicated in causing localized cytokine release, and the resultant inflammatory response and cellular disruption enable these bacteria to enter intestinal cells. Sh. flexneri and Sh. sonnei express an aerobactin-mediated high-affinity iron uptake system; however, as Sh. dysenteriae 1 does not express this siderophore, the role of aerobactin in the pathogenesis of the disease is unclear.
Shiga toxin
Sh. dysenteriae 1 produces a potent protein toxin (Shiga toxin) very similar to Verocytotoxin (VT)-1 expressed by strains of Verocytotoxigenic E. coli (VTEC; see p. 286); however, in contrast to VTEC, the genes encoding Shiga toxin are located on the chromosome. Expression of Shiga toxin has been shown to be iron regulated, with toxin production increasing under conditions of iron restriction.
Shiga toxin is a subunit toxin comprising an A portion and five B subunits. The A subunit possesses the biological activities of the toxin, and the B subunits mediate specific binding and receptor-mediated uptake. In common with the Verocytotoxins of E. coli, Shiga toxin binds to globotriosylceramide (Gb3) molecules present on the surface of certain eukaryotic cells. During pathogenesis, release of the inflammatory mediators tumour necrosis factor and interleukin-1 increases the number of Gb3 receptors on the surface of eukaryotic cells, enhancing the binding of toxin to these cells.
Like Verocytotoxin, Shiga toxin becomes internalized by host cells and remains active within endosomes, eventually reaching the Golgi apparatus. Within the host cell the A subunit divides to form portions A1 and A2; the A1 portion of the toxin prevents protein synthesis and causes cell death. Haemolytic uraemic syndrome is thought to be caused by the action of Shiga toxin on kidney tissues; however, Shiga toxin has also been shown to have neurotoxic properties and the role of this toxin in the pathogenesis of bacillary dysentery remains to be elucidated fully.
Laboratory diagnosis
A specimen of faeces is always preferable to a rectal swab. Rectal swabs do not allow adequate macroscopic and microscopic examination of the stool, and unless taken properly and bearing obvious faecal material may be no more than a swab of peri-anal skin. Moreover, because of drying of the swab, pathogenic species die quite rapidly, and may not survive transport to the laboratory.
The faeces are inoculated on desoxycholate citrate agar or MacConkey agar. Mucus, if present in the specimen, may be used as the inoculum. After overnight incubation, pale non-lactose-fermenting colonies are tested by standard biochemical and sugar utilization tests to differentiate them from other enterobacteria. Identity is confirmed by agglutination tests with serotype-specific rabbit antisera, unless the strain is Sh. sonnei. A DNA-array-based assay has been developed to detect the recognized serotypes of Shigella spp.
Strains of Shigella spp. can be detected with polymerase chain reaction (PCR) tests targeting the genes encoding the invasion plasmid antigen H (ipaH) although this assay will also detect certain strains of invasive E. coli. The genes for Shigella enterotoxin 2 can be detected with the ShET-2 PCR and strains with the genes encoding an aerobactin-mediated iron uptake system can be identified with a PCR using primers targeting the iuc gene complex. Plasmid pattern analysis and colicine typing can also be used to characterize strains of Sh. sonnei.
Expression of Shiga toxin can be detected by Vero and HeLa cell tests (see p. 286) and immunoassays designed for the Verocytotoxin produced by certain E. coli strains.
Patients infected with Sh. dysenteriae 1 produce serum and salivary antibodies to the lipopolysaccharide antigens, but tests for the antibodies are not available routinely.
Treatment
Most cases of shigella dysentery, especially those due to Sh. sonnei, are mild and do not require antibiotic therapy. Maintaining good nutrition is essential. Symptomatic treatment with the maintenance of hydration by use of oral rehydration salt solution (see Table 30.1, p. 318) is all that is required. As with salmonella infections, drugs that impair gut motility should be avoided.
Treatment with a suitable antibiotic is necessary in the very young, the aged or the debilitated, and in those with severe infections. The World Health Organization recommends the use of ciprofloxacin, ceftriaxone and pivmecillinam for the treatment of dysentery in children. There is no evidence that antibiotics reduce the period of excretion of the organisms, and they should not be used in the asymptomatic person, either prophylactically or in attempts to hasten clearance after recovery.
Epidemiology
During the twentieth century, infections due to Sh. dysenteriae 1 and Sh. boydii declined and strains of Sh. sonnei became dominant in the UK and other European countries. In England and Wales more than 16 000 cases of Sonnei dysentery were recorded in 1992. The incidence then declined steadily to an annual average of fewer than 1000 notified cases between 1998 and 2004 (Fig. 25.1). Infections caused by Sh. flexneri have also steadily declined to around 250 cases/year, but are still more common than those caused by Sh. boydii (50–100 cases/year) or Sh. dysenteriae (40–50 cases/year), most of which are contracted abroad. Globally, shigellosis remains a major problem particularly in tropical areas of the developing world, where shigellosis is endemic. It has been estimated that some 5 million cases require hospital treatment and about 600 000 die every year. Young children are particularly vulnerable.
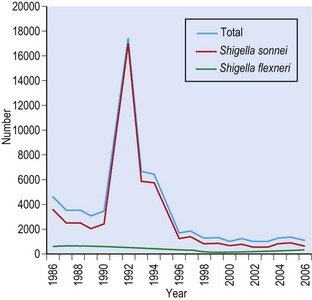
Fig. 25.1 Faecal isolates of shigellae (total), Sh. sonnei and Sh. flexneri, in England and Wales 1986–2004.
(Health Protection Agency data.)
Sources and spread
Bacillary dysentery is highly contagious and is usually spread by the faecal–oral route. The case or carrier, after contaminating his or her hands while cleansing at toilet, may contaminate the lavatory flush-handle, door knobs, washbasin taps, hand towels and other objects that, when handled by another individual, allow transfer of the dysentery bacteria to the recipient’s hands and to the mouth. The carrier may also infect bedding and, in the case of young children, may contaminate toys. Dysentery bacilli are also liberated into the air in an aerosol when an infected loose stool is flushed from the toilet and, after settling on the surfaces of toilet seats, furniture and surroundings, may survive for some days in a moist atmosphere. Infection can also occur among those who indulge in sexual practices involving anal–oral contact. Contamination of foods, particularly those that are to be consumed raw, are a major source of infection.
An important feature of the epidemiology of bacillary dysentery in the UK and other countries with good environmental sanitation is that the main patient group involved is school-aged children, particularly primary school children. Neglect of toilet hygiene by children at school undoubtedly plays a part. The disease can be endemic among adults living in residential institutions where high standards of hygiene are difficult to maintain, and has in the past been a scourge in gaols and in armies in the field. The seasonal distribution of bacillary dysentery in the UK is bimodal, with the highest incidence in spring and a second peak in October and November. The incidence is at its lowest in summer, when school children are on holiday. As this is the period when flying insects are most abundant, this suggests that in the UK insects play little, if any, part in transmission. In certain settings, where disposal of human faeces is inadequate, flies may also serve as vectors for the spread of Shigella bacteria.
Occasional epidemics of bacillary dysentery have been traced to water supplies when chlorination of the supply has not been instituted or has been defective. Such water-borne epidemics are usually spectacular in the large numbers of people simultaneously infected and in the speed with which they can be terminated when the water supply is adequately treated. Epidemic infection may also follow the contamination of milk or ice cream.
Control
The mild and often fleeting nature of the clinical illness associated with Sh. sonnei infection means that frequently the patient with bacillary dysentery remains ambulant and follows his or her daily labour and leisure pursuits, remaining in circulation as a disperser of the causal organism. The pressure on toilet facilities, particularly in schools, allows hand-to-mouth spread of the bacilli. The provision of washbasins in the same compartment as the toilet pedestal would allow some reduction in spread, especially if flushing mechanisms and washbasin taps could be operated by foot instead of by hand.
During diarrhoea, faecal soiling of the fingers can be heavy, and hand washing, although very important, will at best only reduce the numbers of bacteria present. The normal disinfecting effect of the skin fatty acids and competing skin organisms may take up to half an hour to destroy the rest. People with dysentery should stay off work and, as far as possible, out of circulation until the symptoms have subsided, especially when their work involves the preparation of food or direct contact with other people. Asymptomatic carriers are far less important in the spread of this disease and seldom, if ever, need to be excluded from any employment.
Control of an outbreak
Outbreaks of dysentery in schools and other institutions are notoriously difficult to control. In nursery school outbreaks, infection is usually widespread before the first cases have been notified, with considerable environmental contamination. Some children will be incubating the infection; others will have recovered from the diarrhoea but still be excreting the organisms. In this situation the acute case is far more important in the spread of infection than the symptomless excreter. There is little to be gained by trying to ascertain bacteriologically who is infected and who is not once the cause of the outbreak has been established, as there is little reason to exclude an asymptomatic carrier and it is pointless to seek confirmation of a clearly symptomatic case. The practical course is usually to keep children away from school.
Having determined the exclusion policy it is important to try to stop hand-to-hand spread among those who remain at school. Supervision of children using the lavatory, supervised hand washing before meals, frequent disinfection of toilets, including seats, lavatory chain and door handles, and the general use of paper towels all play a part.
Similar principles can be applied to outbreaks in residential institutions or a hospital ward. The most important single factor in all these situations is the need for adequate communication. Teachers, nurses, parents and all who are involved in trying to control the outbreak need to have explained to them exactly how the infection spreads, and the reasons for the measures taken or not taken. Finally, the temptation to use antibiotics prophylactically in an attempt to limit the spread must be resisted.
Vaccination
Infection with a given species of Shigella spp. results in high levels of mucosal IgA-class antibodies which confer protection against subsequent infection with the same species; however, there is only limited protection against infection with a heterologous species. Research has focused on two prototype Shigella vaccines, a live attenuated oral vaccine and the other a parenteral vaccine comprising Shigella LPS conjugated to a carrier protein. Achieving a useful vaccine, capable of proving broad protection, has been hampered by the large number of different epidemiologically significant serotypes of Shigella.
Ashkenasi S. Shigella infections in children: new insights. Seminars in Pediatric Infectious Diseases. 2004;15:246–252.
Kingcombe CI, Cerqueira-Compos ML, Farber JM. Molecular strategies for the detection, identification and differentiation between enteroinvasive Escherichia coli and Shigella spp. Journal of Food Protection. 2005;68:239–245.
Levine MM, Kotloff KL, Barry EM, et al. Clinical trials of Shigella vaccines: two steps forward and one step back on a long, hard road. National Revues in Microbiology. 2007;5:540–553.
Li Y, Cao B, Liu B, et al. Molecular detection of all 34 distinct O-antigen forms of Shigella. Journal of Medical Microbiology. 2009;58:69–81.
Lopez EL, Prado-Jimenez V, O’Ryan-Gallardo M, Contrini MM. Shigella and Shiga toxin-producing Escherichia coli causing bloody diarrhea in Latin America. Infectious Disease Clinics of North America. 2000;14:41–65.
Nhieu GT, Sansonetti PJ. Mechanism of Shigella entry into epithelial cells. Current Opinion in Microbiology. 1999;2:51–55.
Niyogi SK. Shigellosis. Journal of Microbiology. 2005;43:133–143.
Sansonetti PJ. Molecular and cellular mechanisms of invasion of the intestinal barrier by enteric pathogens: the paradigm of Shigella. Folia Microbiologica. 1998;43:239–246.
Sasakawa C. Molecular basis of pathogenicity of Shigella. Reviews in Medical Microbiology. 1995;6:257–266.
Vu DT, Sethabutr O, Von Seidlein L, et al. Detection of Shigella by a PCR assay targeting the ipaH gene suggests increased prevalence of shigellosis in Nha Trang, Vietnam. Journal of Clinical Microbiology. 2004;42:2031–2035.
Yang YG, Song MK, Park SJ, Kim SW. Direct detection of Shigella flexneri and Salmonella typhimurium in human feces by real-time PCR. Journal of Microbiology and Biotechnology. 2007;17:1616–1621.
Centers for Disease Control and Prevention. Shigellosis. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/shigellosis/.
Health Protection Agency. http://www.hpa.org.uk/Topics/InfectiousDiseases/InfectionsAZ/Shigella/.
International Centre for Diarrhoeal Disease Research. http://www.icddrb.org/.
World Health Organization. Shigella. http://www.who.int/topics/shigella/en/.